true
0001938046
S-1/A
0001938046
2023-01-01
2023-12-31
0001938046
dei:BusinessContactMember
2023-01-01
2023-12-31
0001938046
2023-12-31
0001938046
2022-12-31
0001938046
us-gaap:RelatedPartyMember
2023-12-31
0001938046
us-gaap:RelatedPartyMember
2022-12-31
0001938046
us-gaap:NonrelatedPartyMember
2023-12-31
0001938046
us-gaap:NonrelatedPartyMember
2022-12-31
0001938046
2022-01-01
2022-12-31
0001938046
us-gaap:CommonStockMember
2021-12-31
0001938046
us-gaap:AdditionalPaidInCapitalMember
2021-12-31
0001938046
us-gaap:RetainedEarningsMember
2021-12-31
0001938046
2021-12-31
0001938046
us-gaap:CommonStockMember
2022-12-31
0001938046
us-gaap:AdditionalPaidInCapitalMember
2022-12-31
0001938046
us-gaap:RetainedEarningsMember
2022-12-31
0001938046
us-gaap:CommonStockMember
2022-01-01
2022-12-31
0001938046
us-gaap:AdditionalPaidInCapitalMember
2022-01-01
2022-12-31
0001938046
us-gaap:RetainedEarningsMember
2022-01-01
2022-12-31
0001938046
us-gaap:CommonStockMember
2023-01-01
2023-12-31
0001938046
us-gaap:AdditionalPaidInCapitalMember
2023-01-01
2023-12-31
0001938046
us-gaap:RetainedEarningsMember
2023-01-01
2023-12-31
0001938046
us-gaap:CommonStockMember
2023-12-31
0001938046
us-gaap:AdditionalPaidInCapitalMember
2023-12-31
0001938046
us-gaap:RetainedEarningsMember
2023-12-31
0001938046
us-gaap:IPOMember
2023-02-28
2023-03-31
0001938046
us-gaap:IPOMember
2023-03-31
0001938046
MGRX:ResaleProspectusMember
2023-02-28
2023-03-31
0001938046
MGRX:ResaleProspectusMember
2023-03-31
0001938046
MGRX:OptionsMember
2023-01-01
2023-12-31
0001938046
us-gaap:WarrantMember
2023-01-01
2023-12-31
0001938046
MGRX:OptionsMember
2022-01-01
2022-12-31
0001938046
us-gaap:WarrantMember
2022-01-01
2022-12-31
0001938046
srt:MinimumMember
2023-12-31
0001938046
srt:MaximumMember
2023-12-31
0001938046
us-gaap:FairValueInputsLevel1Member
2023-12-31
0001938046
us-gaap:FairValueInputsLevel2Member
2023-12-31
0001938046
us-gaap:FairValueInputsLevel3Member
2023-12-31
0001938046
us-gaap:FairValueInputsLevel1Member
2022-12-31
0001938046
us-gaap:FairValueInputsLevel2Member
2022-12-31
0001938046
us-gaap:FairValueInputsLevel3Member
2022-12-31
0001938046
2022-10-01
0001938046
us-gaap:ComputerEquipmentMember
2023-12-31
0001938046
us-gaap:ComputerEquipmentMember
2022-12-31
0001938046
us-gaap:EquipmentMember
2023-12-31
0001938046
us-gaap:EquipmentMember
2022-12-31
0001938046
MGRX:StockPurchaseAgreementMember
2021-12-09
2021-12-10
0001938046
MGRX:StockPurchaseAgreementMember
2022-03-17
2022-03-18
0001938046
MGRX:StockPurchaseAgreementMember
2023-01-01
2023-12-31
0001938046
MGRX:CohenEnterprisesIncMember
2022-06-15
2022-06-16
0001938046
MGRX:CohenEnterprisesIncMember
2022-06-16
0001938046
MGRX:StockPurchaseAgreementMember
2022-06-15
2022-06-16
0001938046
MGRX:CohenEnterprisesMember
2022-06-28
2022-06-29
0001938046
MGRX:CohenEnterprisesIncMember
2022-08-17
2022-08-18
0001938046
MGRX:CohenEnterprisesIncMember
2022-01-01
2022-12-31
0001938046
MGRX:CohenEnterprisesIncMember
2023-01-01
2023-12-31
0001938046
MGRX:ZipDoctorIncMember
2021-12-09
2021-12-10
0001938046
MGRX:ZipDoctorIncMember
2023-01-01
2023-12-31
0001938046
MGRX:ZipDoctorIncMember
2022-01-01
2022-12-31
0001938046
2022-11-18
0001938046
2023-01-01
2023-03-01
0001938046
2023-04-01
2023-04-01
0001938046
2023-03-23
2023-03-23
0001938046
MGRX:DojoLabsMember
us-gaap:RestrictedStockMember
2023-01-03
2023-01-03
0001938046
MGRX:DojoLabsMember
us-gaap:RestrictedStockMember
2023-01-03
0001938046
MGRX:BethorLtdMember
us-gaap:RestrictedStockMember
2023-01-06
0001938046
MGRX:BethorLtdMember
us-gaap:RestrictedStockMember
2023-01-06
2023-01-06
0001938046
MGRX:RudmanMember
us-gaap:RestrictedStockMember
2023-01-06
2023-01-06
0001938046
MGRX:RudmanMember
us-gaap:RestrictedStockMember
2023-01-06
0001938046
MGRX:BoonMember
us-gaap:RestrictedStockMember
2023-01-06
2023-01-06
0001938046
MGRX:BoonMember
us-gaap:RestrictedStockMember
2023-01-06
0001938046
us-gaap:RestrictedStockMember
2023-01-24
2023-01-24
0001938046
MGRX:ConsultingAgreementsMember
2023-01-24
0001938046
MGRX:ConsultingAgreementsMember
2023-01-24
2023-01-24
0001938046
us-gaap:CommonStockMember
2023-03-22
2023-03-22
0001938046
us-gaap:CommonStockMember
2023-03-22
0001938046
MGRX:PrivatePlacementWarrantOneMember
2023-04-24
0001938046
MGRX:PrivatePlacementWarrantOneMember
2023-04-24
2023-04-24
0001938046
MGRX:PrivatePlacementWarrantTwoMember
2023-04-25
0001938046
MGRX:PrivatePlacementWarrantTwoMember
2023-04-25
2023-04-25
0001938046
MGRX:PrivatePlacementWarrantThreeMember
2023-04-25
0001938046
MGRX:PrivatePlacementWarrantThreeMember
2023-04-25
2023-04-25
0001938046
MGRX:PrivatePlacementWarrantFourMember
2023-04-25
0001938046
MGRX:PrivatePlacementWarrantFourMember
2023-04-25
2023-04-25
0001938046
MGRX:PrivatePlacementWarrantFiveMember
2023-04-25
0001938046
MGRX:PrivatePlacementWarrantFiveMember
2023-04-25
2023-04-25
0001938046
MGRX:PrivatePlacementWarrantSixMember
2023-04-26
0001938046
MGRX:PrivatePlacementWarrantSixMember
2023-04-26
2023-04-26
0001938046
MGRX:PrivatePlacementWarrantSevenMember
2023-05-01
0001938046
MGRX:PrivatePlacementWarrantSevenMember
2023-05-01
2023-05-01
0001938046
MGRX:HammerMember
2023-05-01
2023-05-01
0001938046
MGRX:HammerMember
2023-05-01
0001938046
MGRX:PrivatePlacementWarrantMember
us-gaap:RestrictedStockMember
2023-05-01
2023-05-01
0001938046
MGRX:PrivatePlacementWarrantMember
us-gaap:RestrictedStockMember
2023-05-01
0001938046
MGRX:MrAndrewMember
us-gaap:RestrictedStockMember
MGRX:TwentyTwentyTwoPlanMember
2023-05-25
2023-05-25
0001938046
MGRX:MrAndrewMember
us-gaap:RestrictedStockMember
2023-05-25
0001938046
MGRX:MrAndrewMember
us-gaap:RestrictedStockMember
2023-05-25
2023-05-25
0001938046
MGRX:MajorDodgeMember
us-gaap:RestrictedStockMember
MGRX:TwentyTwentyTwoPlanMember
2023-06-01
2023-06-01
0001938046
MGRX:MajorDodgeMember
us-gaap:RestrictedStockMember
2023-06-01
0001938046
MGRX:MajorDodgeMember
us-gaap:RestrictedStockMember
2023-06-01
2023-06-01
0001938046
MGRX:StreetGroupLLCMember
us-gaap:RestrictedStockMember
2023-06-01
2023-06-01
0001938046
MGRX:StreetGroupLLCMember
us-gaap:RestrictedStockMember
2023-06-01
0001938046
MGRX:PrivatePlacementWarrantMember
2023-06-05
0001938046
MGRX:PrivatePlacementWarrantMember
2023-06-05
2023-06-05
0001938046
MGRX:PrivatePlacementWarrantOneMember
2023-06-06
0001938046
MGRX:PrivatePlacementWarrantOneMember
2023-06-06
2023-06-06
0001938046
MGRX:PrivatePlacementWarrantTwoMember
2023-06-07
0001938046
MGRX:PrivatePlacementWarrantTwoMember
2023-06-07
2023-06-07
0001938046
MGRX:PrivatePlacementWarrantThreeMember
2023-06-08
0001938046
MGRX:PrivatePlacementWarrantThreeMember
2023-06-08
2023-06-08
0001938046
MGRX:PrivatePlacementWarrantFourMember
2023-06-21
0001938046
MGRX:PrivatePlacementWarrantFourMember
2023-06-21
2023-06-21
0001938046
MGRX:PrivatePlacementWarrantFiveMember
2023-06-22
0001938046
MGRX:PrivatePlacementWarrantFiveMember
2023-06-22
2023-06-22
0001938046
MGRX:PrivatePlacementWarrantSixMember
2023-06-27
0001938046
MGRX:PrivatePlacementWarrantSixMember
2023-06-27
2023-06-27
0001938046
us-gaap:RestrictedStockMember
MGRX:GreentreeFinancialGroupMember
MGRX:ServiceAgreementMember
2023-01-01
2023-12-31
0001938046
MGRX:ServiceAgreementMember
MGRX:GreentreeFinancialGroupMember
2023-12-31
0001938046
MGRX:ServiceAgreementMember
MGRX:GreentreeFinancialGroupMember
2023-01-01
2023-12-31
0001938046
MGRX:ConsultingAgreementMember
2023-10-01
2023-10-01
0001938046
MGRX:ConsultingAgreementMember
MGRX:LucaConsultingLlcMember
2023-10-10
2023-10-10
0001938046
MGRX:LucaConsultingLlcMember
MGRX:ServiceAgreementMember
2023-10-10
2023-10-10
0001938046
MGRX:ConsultingAgreementMember
MGRX:LucaConsultingLlcMember
2023-10-10
0001938046
MGRX:JasonSzkupMember
us-gaap:RestrictedStockMember
2023-11-01
2023-11-01
0001938046
MGRX:JasonSzkupMember
us-gaap:RestrictedStockMember
2023-11-01
0001938046
MGRX:DrDouglasChristiansonMember
us-gaap:RestrictedStockMember
2023-11-01
2023-11-01
0001938046
MGRX:MrAndrewMember
us-gaap:RestrictedStockMember
2023-11-01
0001938046
MGRX:PHXGlobalLLCMember
us-gaap:RestrictedStockMember
2023-11-15
2023-11-15
0001938046
MGRX:PHXGlobalLLCMember
us-gaap:RestrictedStockMember
2023-11-15
0001938046
MGRX:MariusPharmaceuticalsMember
us-gaap:RestrictedStockMember
2023-12-11
2023-12-11
0001938046
MGRX:MariusPharmaceuticalsMember
us-gaap:RestrictedStockMember
2023-12-11
0001938046
MGRX:MariusPharmaceuticalsMember
us-gaap:RestrictedStockMember
2023-12-11
2023-12-11
0001938046
us-gaap:CommonStockMember
2023-12-19
2023-12-19
0001938046
us-gaap:CommonStockMember
2023-12-19
0001938046
us-gaap:CommonStockMember
MGRX:TwentyTwentyTwoPlanMember
2022-12-31
0001938046
us-gaap:CommonStockMember
srt:ChiefExecutiveOfficerMember
MGRX:TwentyTwentyTwoPlanMember
2022-01-01
2022-12-31
0001938046
us-gaap:CommonStockMember
srt:ChiefOperatingOfficerMember
MGRX:TwentyTwentyTwoPlanMember
2022-01-01
2022-12-31
0001938046
us-gaap:CommonStockMember
srt:ChiefExecutiveOfficerMember
2022-12-31
0001938046
us-gaap:CommonStockMember
MGRX:TwentyTwentyTwoPlanMember
2023-05-01
0001938046
us-gaap:CommonStockMember
2023-05-01
0001938046
us-gaap:CommonStockMember
MGRX:TwentyTwentyTwoPlanMember
2023-12-28
0001938046
us-gaap:CommonStockMember
2023-12-28
0001938046
us-gaap:CommonStockMember
us-gaap:IPOMember
2023-03-20
0001938046
2023-03-20
2023-03-20
0001938046
us-gaap:CommonStockMember
us-gaap:IPOMember
2023-12-19
0001938046
2023-12-19
2023-12-19
0001938046
us-gaap:InvestorMember
2023-12-31
0001938046
us-gaap:InvestorMember
2022-12-31
0001938046
us-gaap:WarrantMember
2023-12-31
0001938046
us-gaap:WarrantMember
2023-01-01
2023-12-31
0001938046
us-gaap:WarrantMember
2023-01-01
2023-12-31
0001938046
MGRX:RangeOneMember
2023-12-31
0001938046
MGRX:RangeOneMember
2023-01-01
2023-12-31
0001938046
MGRX:RangeTwoMember
2023-12-31
0001938046
MGRX:RangeTwoMember
2023-01-01
2023-12-31
0001938046
srt:MaximumMember
2023-01-01
2023-12-31
0001938046
srt:MinimumMember
2023-01-01
2023-12-31
0001938046
us-gaap:WarrantMember
srt:MinimumMember
2023-12-31
0001938046
us-gaap:WarrantMember
srt:MaximumMember
2023-12-31
0001938046
us-gaap:WarrantMember
srt:MinimumMember
2023-01-01
2023-12-31
0001938046
us-gaap:WarrantMember
srt:MaximumMember
2023-01-01
2023-12-31
0001938046
us-gaap:WarrantMember
2021-12-31
0001938046
us-gaap:WarrantMember
2022-01-01
2022-12-31
0001938046
us-gaap:WarrantMember
2022-12-31
0001938046
2022-09-28
0001938046
2022-09-28
2022-09-28
0001938046
MGRX:LeaseAgreementMember
2022-09-28
2022-09-28
0001938046
srt:MaximumMember
2017-12-22
2017-12-22
0001938046
srt:MinimumMember
2017-12-22
2017-12-22
0001938046
us-gaap:SubsequentEventMember
MGRX:ConsultingAgreementMember
MGRX:GAndPGeneralConsultingMember
2024-01-02
2024-01-02
0001938046
us-gaap:SubsequentEventMember
MGRX:ConsultingAgreementMember
MGRX:GAndPGeneralConsultingMember
2024-01-02
0001938046
us-gaap:SubsequentEventMember
MGRX:ConsultingAgreementMember
MGRX:LucaConsultingLlcMember
2024-01-10
2024-01-10
0001938046
us-gaap:SubsequentEventMember
MGRX:ConsultingAgreementMember
MGRX:LucaConsultingLlcMember
2024-01-10
0001938046
us-gaap:SubsequentEventMember
MGRX:ConsultingAgreementMember
MGRX:FirstLevelCapitalMember
2024-01-11
2024-01-11
0001938046
us-gaap:SubsequentEventMember
MGRX:ConsultingAgreementMember
MGRX:FirstLevelCapitalMember
2024-01-11
0001938046
us-gaap:SubsequentEventMember
MGRX:UnderwritersMember
2024-01-18
2024-01-18
0001938046
us-gaap:SubsequentEventMember
MGRX:UnderwritersMember
us-gaap:OverAllotmentOptionMember
2024-01-18
2024-01-18
0001938046
us-gaap:SubsequentEventMember
MGRX:UnderwritingAgreementMember
us-gaap:WarrantMember
2024-01-22
0001938046
us-gaap:SubsequentEventMember
MGRX:UnderwritingAgreementMember
2024-01-22
0001938046
us-gaap:SubsequentEventMember
MGRX:ConsultingAgreementMember
MGRX:LucaConsultingLlcMember
2024-03-21
2024-03-21
0001938046
us-gaap:SubsequentEventMember
MGRX:ConsultingAgreementMember
MGRX:LucaConsultingLlcMember
2024-03-21
0001938046
us-gaap:SubsequentEventMember
MGRX:ConsultingAgreementMember
MGRX:ZvonimirMoricConsultingMember
2024-03-21
2024-03-21
0001938046
us-gaap:SubsequentEventMember
MGRX:ConsultingAgreementMember
MGRX:ZvonimirMoricConsultingMember
2024-03-21
0001938046
us-gaap:SubsequentEventMember
MGRX:RoninEquityPartnersMember
2024-04-01
0001938046
us-gaap:SubsequentEventMember
MGRX:CohenEnterprisesIncMember
2024-03-18
0001938046
us-gaap:SubsequentEventMember
MGRX:TwoThouandTwentyTwoPlanMember
2024-03-25
2024-03-25
0001938046
us-gaap:SubsequentEventMember
MGRX:TwoThouandTwentyTwoPlanMember
2024-03-25
0001938046
us-gaap:SubsequentEventMember
MGRX:TwoThouandTwentyTwoPlanMember
us-gaap:CommonStockMember
2024-03-25
0001938046
us-gaap:SubsequentEventMember
MGRX:TwoThouandTwentyTwoPlanMember
us-gaap:CommonStockMember
2024-03-25
2024-03-25
0001938046
us-gaap:SubsequentEventMember
2024-03-25
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
utr:sqft
xbrli:pure
As
filed with the Securities and Exchange Commission on May 6, 2024
Registration
No. 333-278888
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
AMENDMENT
NO. 1
FORM
S-1
REGISTRATION
STATEMENT
UNDER
THE
SECURITIES ACT OF 1933
Mangoceuticals,
Inc.
(Exact
Name of Registrant as Specified in Its Charter)
| Texas |
|
8099 |
|
87-3841292 |
(State
or other jurisdiction of
incorporation
or organization) |
|
(Primary
Standard Industrial
Classification
Code Number) |
|
(I.R.S.
Employer
Identification
Number) |
15110
N. Dallas Parkway, Suite 600
Dallas,
Texas 75248
(214)
242-9619
(Address,
including zip code, and telephone number, including area code, of registrant’s principal executive office)
Jacob
D. Cohen
Chief
Executive Officer
Mangoceuticals,
Inc.
15110
N. Dallas Parkway, Suite 600
Dallas,
Texas 75248
(214)
242-9619
(Name,
address, including zip code, and telephone number, including area code, of agent for service)
Copies
to:
Joseph
M. Lucosky, Esq.
Steven
Lipstein, Esq.
Lucosky
Brookman LLP
101
Wood Avenue South, 5th Floor
Woodbridge,
NJ 08830
(732)
395-4400
Approximate
date of commencement of proposed sale to the public: As soon as practicable after the effective date of this registration statement.
If
any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the
Securities Act of 1933, check the following box. ☒
If
this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, check the following
box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering.
☐
If
this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the
Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If
this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the
Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
Indicate
by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting
company or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer”,
“smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large
accelerated filer ☐ |
Accelerated
filer ☐ |
| Non-accelerated
filer ☒ |
Smaller
reporting company ☒ |
| |
Emerging
growth company ☒ |
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided pursuant to Section 7(a)(2)(B) of the Securities Act.
The
Registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the
Registrant shall file a further amendment which specifically states that this registration statement shall thereafter become effective
in accordance with Section 8(a) of the Securities Act of 1933, as amended, or until the registration statement shall become effective
on such date as the Commission, acting pursuant to such Section 8(a), may determine.
The
information in this preliminary prospectus is not complete and may be changed. These securities may not be sold until the registration
statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these securities
and we are not soliciting an offer to buy these securities in any jurisdiction where the offer or sale is not permitted.
| PROSPECTUS |
SUBJECT
TO COMPLETION, DATED MAY 6, 2024 |

Mangoceuticals,
Inc.
30,014,286
Shares of Common Stock
We
are registering the shares of common stock to be issued to permit the resale of these shares of common stock, after they are issued,
by the holders of the common stock from time to time after the date of this prospectus. We will not receive any of the proceeds from
the sale by the selling stockholders of the shares of common stock. Any proceeds received by the Company from the sale of shares of common
stock pursuant to the ELOC and from the exercise of the Warrants will be used for general working capital.
The
selling stockholders named in this prospectus may offer and sell, from time to time, in one or more offerings, up to an aggregate of
30,014,286 shares of our common stock, par value $0.0001 per share consisting of (i) one million (1,000,000) shares issued as a commitment
fee in connection with the Company and the selling stockholder’s entrance into the ELOC (as defined on page 18 and
described in the section entitled “Recent Events”); (ii) ten million (10,000,000) shares issuable under the ELOC using
an adjusted price of $0.26 for the per share purchase price (based on a $2.6 million total purchase price divided by the average closing
price of the common stock on the Nasdaq Capital Market for the three trading days ending on April 19, 2024 ($0.29) minus a ten percent
(10%) discount for the purchase price of $0.26), (iii) three million three hundred thousand (3,300,000) shares issuable upon exercise
of warrants issued at an exercise price of $0.26 per share (the “Warrants”), and (iv) 15,714,286 shares issuable upon conversion
of 500 shares of Series B Convertible Preferred Stock (the “Series B Preferred Stock”) with each share having a stated
value of $1,100 and having an assumed conversion price of the floor price of $0.035 per share. Pursuant to the Securities Purchase Agreement
(“SPA”) entered into, the sale of the Series B Preferred Stock and Warrants is to take place over up to three closings as
indicated in the agreement. The registration statement of which prospectus forms a part only registers the shares of common stock underlying
the initial closing of 500 shares of Series B Preferred Stock. The Company has reserved from its duly authorized capital stock 50,000,000
of shares of Common Stock issuable upon exercise of the Warrants and conversion of the Series B Preferred Stock.
We
will receive proceeds from any sale of shares of common stock pursuant to the ELOC and from any exercise of the Warrants, however,
there is no guarantee that any shares will be sold or any of the Warrants will be exercised. The shares of our common stock may be sold
publicly or through private transactions by the selling stockholders at prevailing market prices or at negotiated prices at the times
of sale. The shares of common stock may be offered by the selling stockholders to or through underwriters, dealers or other agents, directly
to investors or through any other manner permitted by law, on a continued or delayed basis. We provide more information about how the
selling stockholders may sell or otherwise dispose of the shares of common stock in the section entitled “Plan of Distribution”
beginning on page 129 of this prospectus.
We
are not selling any shares of common stock in this offering, and we will not receive any proceeds from the sale of shares by the selling
stockholders. The registration of the securities covered by this prospectus does not necessarily mean that any of these securities will
be offered or sold by the selling stockholders. The timing and amount of any sale is within the respective selling stockholders’
sole discretion, subject to certain restrictions. To the extent that any selling stockholder resells any securities, the selling stockholder
may be required to provide you with this prospectus identifying and containing specific information about the selling stockholder and
the terms of the securities being offered.
To
the extent required by the Securities Act and the rules and regulations thereunder, the selling stockholders and any broker-dealer participating
in the distribution of the shares of common stock may be deemed to be “underwriters” within the meaning of the Securities
Act, and any commission paid, or any discounts or concessions allowed to, any such broker-dealer may be deemed to be underwriting commissions
or discounts under the Securities Act.
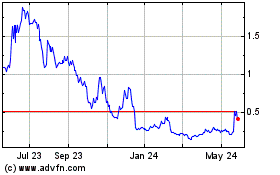
Shares
of our common stock are listed on the Nasdaq Capital Market (“Nasdaq”) under the symbol “MGRX”. On May 3,
2024, the last sale price per share of our common stock as reported on Nasdaq was $0.264.
We
are an “emerging growth company” as defined in the federal securities laws and, as a result, have elected to comply with
certain reduced public company disclosure and reporting requirements.
Investing
in our common stock involves risks that are described in the “Risk Factors” section in any other annual, periodic or current
report.
Neither
the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined
if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The
date of this prospectus is May 6, 2024
TABLE
OF CONTENTS
ABOUT
THIS PROSPECTUS
This
prospectus is part of a registration statement that we filed with the Securities and Exchange Commission (the “SEC”), using
a “shelf” registration process. Under this shelf registration process, the selling stockholders may, from time to time, offer
and sell shares of common stock offered under this prospectus. We will not receive any proceeds from the sale by the selling stockholders
of the common stock offered by them described in this prospectus.
We
and the selling stockholders have not authorized anyone to provide any information or make any representations other than those contained
in this prospectus. We and the selling stockholders take no responsibility for, and can provide no assurance as to the reliability of,
any other information that others may give you. This prospectus is an offer to sell only the securities offered hereby and only under
circumstances and in jurisdictions where it is lawful to do so. No dealer, salesperson, or other person is authorized to give any information
or to represent anything not contained in this prospectus. This prospectus is not an offer to sell securities, and it is not soliciting
an offer to buy securities, in any jurisdiction where the offer or sale is not permitted. The information in this prospectus is current
only as of its date. Our business, financial condition, results of operations, and prospects may have changed since its date.
This
prospectus contains summaries of certain provisions contained in some of the documents described herein, but reference is made to the
actual documents for complete information. All of the summaries are qualified in their entirety by the actual documents. Copies of some
of the documents referred to herein have been filed or will be filed as exhibits to the registration statement of which this prospectus
is a part, and you may obtain copies of those documents as described in the section entitled “Where You Can Find More Information.”
The
selling stockholders are offering to sell, and seeking offers to buy, shares of our common stock only in jurisdictions where offers and
sales are permitted. The information contained in this prospectus is accurate only as of the date of this prospectus regardless of the
time of delivery of this prospectus or of any sale of common stock. Neither the delivery of this prospectus, nor any sale made hereunder,
will under any circumstances create any implication that there has been no change in our affairs since the date hereof or that the information
contained herein is correct as of any time subsequent to the date of such information.
For
investors outside the United States: Neither we nor the selling stockholders have done anything that would permit this offering or possession
or distribution of this prospectus in connection with this offering in any jurisdiction, other than the United States, where action for
that purpose is required. Persons outside the United States who come into possession of this prospectus must inform themselves about,
and observe any restrictions relating to, the offering of our common stock and the distribution of this prospectus outside the United
States and in their jurisdiction.
Unless
otherwise indicated or the context otherwise requires, all references in this prospectus to “Mangoceuticals” or the “Company,”
“we,” “our,” “ours,” “us” or similar terms refer to Mangoceuticals, Inc., together with
its consolidated subsidiaries.
CAUTIONARY
STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
This
prospectus contains statements that constitute forward-looking statements. Many of the forward-looking statements contained in this prospectus
can be identified by the use of forward-looking words such as “anticipate,” “believe,” “could,” “expect,”
“should,” “plan,” “intend,” “may,” “predict,” “continue,” “estimate,”
and “potential,” or the negative of these terms or other similar expressions.
Forward-looking
statements appear in a number of places in this prospectus and include, but are not limited to, statements regarding our intent, beliefs
or current expectations. These forward-looking statements include information about possible or assumed future results of our business,
financial condition, results of operations, liquidity, plans, and objectives. Forward-looking statements are based on our management’s
beliefs and assumptions and on information currently available to our management. Such statements are subject to risks and uncertainties,
and actual results may differ materially from those expressed or implied in the forward-looking statements due to various factors, including,
but not limited to, those identified described in the section “Risk Factors” in any other annual, periodic, or current report.
The statements we make regarding the following matters are forward-looking by their nature:
| |
● |
our
ability to obtain additional funding, the terms of such funding, and dilution caused thereby; |
| |
|
|
| |
● |
the
effect of pandemics on our operations, sales, and the market for our products; |
| |
|
|
| |
● |
our
ability to build and maintain our brand; |
| |
|
|
| |
● |
cybersecurity,
information systems and fraud risks and problems with our websites; |
| |
|
|
| |
● |
our
ability to expand and grow our operations, and successfully market our products; |
| |
|
|
| |
● |
changes
in, and our compliance with, rules and regulations affecting our operations, sales, and/or our products; |
| |
|
|
| |
● |
shipping,
production or manufacturing delays; |
| |
|
|
| |
● |
our
ability to increase sales; |
| |
|
|
| |
● |
regulations
we are required to comply with in connection with our operations, manufacturing, labeling and shipping; |
| |
|
|
| |
● |
competition
from existing competitors or new competitors or products that may emerge; |
| |
|
|
| |
● |
our
dependency on third-parties to prescribe and compound our erectile dysfunction (ED) product; |
| |
|
|
| |
● |
our
ability to establish or maintain relations and/or relationships with third-parties; |
| |
|
|
| |
● |
potential
safety risks associated with our Mango ED and Mango GROW products, including the use of ingredients, combination of such ingredients
and the dosages thereof; |
| |
|
|
| |
● |
the
effects of high inflation, increasing interest rates and economic downturns, including potential recessions, as well as macroeconomic,
geopolitical, health and industry trends, pandemics, acts of war (including the ongoing Ukraine/Russian conflict and ongoing conflict
in and around Israel) and other large-scale crises; |
| |
|
|
| |
● |
our
ability to protect intellectual property rights; |
| |
|
|
| |
● |
our
ability to adequately support future growth; |
| |
|
|
| |
● |
our
ability to attract and retain key personnel to manage our business effectively; and |
| |
|
|
| |
● |
other
risk factors included under “Risk Factors” below. |
Further
information on risks, uncertainties, and other factors that could affect our financial results are included in our filings with the Securities
and Exchange Commission (the “SEC”) from time to time, including in the section entitled “Risk Factors” in any
other annual, periodic or current report. You should not rely on these forward-looking statements, as actual outcomes and results may
differ materially from those expressed or implied in the forward-looking statements as a result of such risks and uncertainties. All
forward-looking statements in this prospectus are based on management’s beliefs and assumptions and on information currently available
to us, and we do not assume any obligation to update the forward-looking statements provided to reflect events that occur or circumstances
that exist after the date on which they were made.
PROSPECTUS
SUMMARY
This
summary highlights certain information contained elsewhere in this prospectus. This summary is not complete and does not contain all
the information that may be important to you. We urge you to read this entire prospectus carefully, including the sections entitled “Risk
Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and our
consolidated financial statements and notes thereto included herein, before making an investment decision. Some of the statements made
in this prospectus discuss future events and developments, including our future strategy and our ability to generate revenue, income
and cash flow. These forward-looking statements involve risks and uncertainties which could cause actual results to differ materially
from those contemplated in these forward-looking statements. See “Cautionary Statement Regarding Forward-Looking Statements”.
Company
Overview
We
connect consumers to licensed healthcare professionals through our website at www.MangoRX.com, for the provision of care via telehealth
on our customer portal. We also provide access for customers to a licensed pharmacy for online fulfillment and distribution of certain
medications that may be prescribed as part of telehealth consultations, including our Mango ED and Mango GROW products, as further defined
below.
We
have identified men’s wellness telemedicine services and products as a growing sector in recent years and especially related to
the areas of erectile dysfunction (“ED”), hair growth products and hormone therapies.
Our
Products
Mango
ED
We
have developed and are marketing and selling a new brand of ED product under the brand name “Mango.” This product is produced
at a compounding pharmacy and is available to patients on the determination of a prescribing physician that the compounded drug is necessary
for the individual patient. This product currently includes the following three ingredients: Either Sildenafil (the active ingredient
in Viagra) or Tadalafil (the active ingredient in Cialis), and Oxytocin, all of which are used in U.S. Food and Drug Administration (“FDA”)
approved drugs, as well as L-Arginine, an amino acid that is available as a dietary supplement. However, the fact that Tadalafil, Sildenafil
and Oxytocin are used in FDA approved drugs, and L-arginine is available as a dietary supplement, does not mean that these ingredients
will prove safe when combined into a single formulation to treat ED. We currently offer two dosage levels of our Mango ED product and
anticipate doctors prescribing a dosage based on the needs and medical history of the patient. Our Mango ED product currently includes
the following amounts of the three ingredients: (1) either Sildenafil (50 milligrams (mg)) or Tadalafil (10 (mg)), Oxytocin (100 International
units (IU)) and L-Arginine (50mg); and (2) either Sildenafil (100 milligrams (mg)) or Tadalafil (20mg), Oxytocin (100IU) and L-Arginine
(50mg). Our Mango ED product has not been, and will not be, approved by the FDA and instead we produce and sell our Mango ED product
and plan to produce and sell future pharmaceutical products, under an exemption provided by Section 503A of the Federal Food, Drug, and
Cosmetic Act.
We
are not aware of any clinical studies involving the administration of Tadalafil or Sildenafil sublingually at the doses we provide patients,
or the compounding of Tadalafil or Sildenafil, Oxytocin and L-arginine to treat ED, as is contemplated by our ED product. We are, however,
aware of other companies that are currently selling oral disintegrating tablets for ED, including those using a combination of Tadalafil
and Sildenafil. Additionally, because our Mango ED product is being specially compounded for the customer by a pharmacist with a physician’s
prescription and because the ingredients for our Mango ED product are publicly disclosed, this product formula can be replicated by other
companies.
Mango
Hair Growth Product - ‘GROW’ by MangoRx
We
have developed, and since November 16, 2022 are marketing and selling, a new brand of hair growth product under the brand name ‘GROW’
by MangoRx (“Mango GROW”). This product is produced at our related party compounding pharmacy and is available to patients
on the determination of a prescribing physician that the compounded drug is necessary for the individual patient. Mango GROW currently
includes the following four ingredients - (1) Minoxidil (the active ingredient in Rogaine®) and (2) Finasteride (the active ingredient
in Propecia), each of which is used in FDA approved drugs, as well as (3) Vitamin D3 and (4) Biotin, which are available as dietary supplements.
However, the fact that Minoxidil and Finasteride are used in FDA approved drugs, and that Vitamin D3 and Biotin, are available as a dietary
supplement, does not mean that these ingredients will prove safe when combined into a single formulation to attempt to treat hair growth.
Mango GROW is encapsulated in convenient chewable, mint-flavored rapid dissolve tablets (“RDT”).
We
currently offer one dosage level of our Mango GROW product and anticipate doctors prescribing Mango GROW based on the needs and medical
history of the patient. Our Mango GROW product currently includes the following amounts of the four ingredients: (1) Minoxidil (2.5mg),
(2) Finasteride (1mg), (3) Vitamin D3 (2000IU) and (4) Biotin (1mg). Our Mango GROW product has not been, and will not be, approved by
the FDA and instead we produce and sell our Mango GROW product and plan to produce and sell future pharmaceutical products, under an
exemption provided by Section 503A of the Federal Food, Drug, and Cosmetic Act.
We
are not aware of any clinical studies involving the administration of Minoxidil and Finasteride sublingually at the dose we provide patients,
or the compounding of Minoxidil, Finasteride, Vitamin D3 and Biotin to treat hair growth. We are, however, aware of other companies that
are currently selling oral tablets for hair growth, including those using a combination of Minoxidil and Finasteride. Additionally, because
our Mango GROW product is being specially compounded for the customer by a pharmacist with a physician’s prescription and because
the ingredients for our Mango GROW product are publicly disclosed, this product formula can be replicated by other companies.
Additional
Information Regarding Mango ED and Mango GROW
Because
our Mango ED and Mango GROW products have not been, and will not be, approved by the FDA, our products have not had the benefit of the
FDA’s clinical trial protocol which seeks to prevent the possibility of serious patient injury and death. If this were to occur,
we could be subject to litigation and governmental action, which could result in costly litigation, significant fines, judgments or penalties.
We
currently anticipate using a substantial portion of the net proceeds of this offering to finance marketing and general operational expenses
associated with the sale of our Mango ED and Mango GROW products. We launched our website in mid-November 2022. To date, we have sold
only a small amount of products and generated only minimal revenues.
Mango
ED and Mango GROW have been formulated as RDT using a sublingual (applied under the tongue) delivery system to bypass the stomach and
liver. It is a generally established principle that sublingual drug absorption through the oral mucosa is generally faster than drug
absorption through the gastrointestinal tract. This is because sublingual drugs that are absorbed through the oral mucosa directly enter
the systemic circulation, bypassing the gastrointestinal tract and first-pass metabolism in the liver (see H. Zhang et al., Oral mucosal
drug delivery: clinical pharmacokinetics and therapeutic applications, 41 Clin Pharmacokinet
661, 662 (2002). Though the active ingredients that comprise our Mango ED product are meant to treat ED - an issue that according
to a 2018 study published in The Journal of Sexual Medicine has been estimated to affect over one-third of today’s men’s
population (with prevalence increasing with age) - we are also aiming to brand ourselves as a lifestyle company marketed to men seeking
enhanced sexual vitality, performance, and overall mood and confidence, together with our Mango GROW hair growth product.
Our
Mango products are sold exclusively online via our website at www.MangoRX.com.
Our
Customer Portal
Our
customer platform connects consumers to licensed healthcare professionals through our website at www.MangoRX.com for the
provision of care via telehealth and also provides access for customers to a licensed pharmacy for online fulfillment and
distribution of certain medications that may be prescribed as part of a telehealth consultation. Additional features to this backend
technology solution allow for the creation and management of customer accounts whereby customers have the ability to login, view and
make changes to their respective accounts, including reviewing order history, tracking order shipments, requesting and ordering
product refills and making other profile changes such as shipping address and payment changes. Our portal is not unique to the
industry and is not anticipated to be difficult or costly to replicate or replace.
The
backend technology solution also houses and manages all customer data providing us with additional key functionality, including to provide
customer service and support and data analytics for various marketing initiatives and reporting functions.
We
do not anticipate selling any third-party products via our portal.
Our
Growth Strategy
Our
growth strategy includes the following key initiatives:
Utilize
a variety of marketing channels using data analytics to attract customers
We
market and advertise our Mango products on a variety of advertising mediums including social media, online search websites, television,
radio, out-of-home, and other media channels, to the extent we are legally able to, and in compliance with applicable FDA rules and requirements;
however, due to such rules and requirements, we are extremely limited in the content of the claims and promotional statements that we
are able to make regarding our products under applicable FDA regulations. We believe advertising in a diversified set of media channels
is important to prevent overreliance on any single channel and to maximize the exposure of our brand to our desired customers. We also
seek to reach our customers through our own social media accounts, press coverage and public relations, internally developed educational
and lifestyle content, and through engagement of social media influencers, hired and paid celebrities and talent, and physical brand
advertising campaigns and in each case subject to applicable rules and regulations, which are expected to significantly limit the content
of such marketing materials. We believe that this overall strategy will drive customer traffic to our platform, including direct type-in
traffic and organic online search traffic.
We
also utilize a marketing strategy focused on analytics and data. We have designed our internal systems to measure consumer behavior,
including which types of consumers generate more revenue in their first purchase, generate more revenue over time, generate more gross
profit from their purchases, and which types of consumers are most valuable over their lifetime. We also seek to measure the effectiveness
of our marketing budgets and the rate of return we generate from our marketing campaigns. We also use outside marketing and advertising
firms to assist management in identifying marketing and advertising campaigns, media purchases and mediums, and seeking to drive a sufficient
rate of return from our marketing and advertising budgets.
Invest
in our telemedicine platform to enable sales throughout the United States
We
utilize both a synchronous and asynchronous approach through our telemedicine platform, connecting customers through our platform and
contracted physicians and pharmacy. An asynchronous visit allows a physician to verify the patient’s identity, demographics and
collect the medical history online without needing to physically see or speak to the patient while a synchronous visit requires the doctor
to either speak directly to the patient and/or see the patient either via video conference or in person. As discussed above, we focus
our sales in the District of Columbia and the 47 states where our related party pharmacy is licensed, with the goal of eventually undertaking
sales across all 50 states, pending licensing approvals of our related party pharmacy.
Provide
subscription plans for recurring revenue and introduction of new products
We
provide our customers with an option to purchase our Mango products on a subscription basis. Subscription plans provide an easy and convenient
way for customers to get ongoing treatment while simultaneously providing us with predictability through a recurring revenue stream.
For
subscription plans, customers are able to select a desired timeframe in which to receive products, which ranges from once every month
to once every six months, depending on several factors. The customer is then billed on a recurring basis based on the selected timeframe
and specified quantity of product, which is shipped after each billing from our contracted pharmacy (Epiq Scripts (defined and discussed
below)). Customers are able to cancel subscriptions in between billing periods to stop receiving additional products and reactivate subscriptions.
Our integrated technology platform allows us to efficiently serve customers from customer discovery, through the purchase of products
on our website, to connecting customers with medical providers for telehealth consultations (through our contracted physician network
(which is contracted through Epiq Scripts (defined and discussed below)), to the fulfillment and delivery of orders (through our contracted
pharmacy), and finally through ongoing management by medical providers (also through our contracted physician network). We believe that
our platform provides us cost advantages and efficiencies to offer customers affordable prices and generate increased revenues over time.
We
intend to launch new products over time and offer additional subscription-based offerings which we hope will result in growth in revenue
through recurring revenue streams.
Market
Overview
The
Market for ED Products
According
to a January 2022 report published by Verified Market Research, the Global Erectile Dysfunction Drugs Market size was valued at $3.63
billion in 2020, mainly due to the increase in patient awareness and the early adoption of sedentary lifestyle. Verified Market Research
also projects that the total Global Dysfunction Drugs Market size will contract to $2.95 billion in 2028. The expected reason for this
contraction is poor patient compliance with erectile dysfunction drugs and the future availability of cost-effective imitation medicines,
as well as side effects of ED drugs. We do not anticipate our Mango ED drug suffering from these limitations, as we believe our product
is easy to use and that we have priced our product competitively. Separately, Grand View Research, in a July 2022 report, projects that
the U.S. market (where we initially plan to market our ED product) for erectile dysfunction drugs which is estimated at approximately
$1.1 billion as of 2021, will increase at a 7.4% compound annual growth rate though 2030.
Further,
it is estimated that nearly 3-in-5 men questioned in the U.S. have suffered from erectile dysfunction, according to a survey reported
in February 2022 by LetsGetChecked, a leading at-home health screening and insights company (based on research carried out by Opinium
Research among 2,006 men in the U.S., 1,178 of whom had previously experienced erectile dysfunction, from February 7-10, 2020). According
to that study, age isn’t that big a factor either, with 56% of men 18 to 34 years old being affected, compared to 63% of those
over the age of 55. The study also determined that most men blame psychological factors for ED - with 41% blaming stress, 34% blaming
having “too much on their mind,” and 31% believing it is performance anxiety.
The
Market for Mango GROW
According
to the website of the American Hair Loss Association, (a) two-thirds of American men will experience some degree of hair loss by the
age of 35, (b) by age 50, around 85 percent of men have significantly thinning hair; and (c) for around 25% of men, the start of male
pattern baldness can begin before the age of 21. Additionally, and contrary to societal belief, we believe that most men who suffer from
male pattern baldness are unhappy with their situation and would take steps to change that. In our experience, hair loss affects every
aspect of the hair loss sufferer’s life including interpersonal relationships as well as the professional lives of those suffering.
According
to a May 2022 market study entitled, “Hair Loss Prevention Products Market Forecast to 2028 - COVID-19 Impact and Global Analysis
- by Product Type (Shampoos and Conditioners, Oils, Serums, and Others), Category (Natural & Organic, and Conventional), End User
(Men, Women, and Unisex), and Distribution Channel (Supermarkets and Hypermarkets, Convenience Stores, Online Retail, and Others)”,
by The Insight Partners, the hair loss prevention products market size was valued at $23.6 billion in 2021 and is projected to reach
$31.5 billion by 2028, growing at a projected compound annual growth rate of 4.2% from 2021 to 2028.
Mordor
Intelligence LLP believes that the major factors driving the hair loss prevention market are changing lifestyle patterns, adoption of
a hectic schedule that increases stress levels, which in turn results in frequent hair loss at an earlier stage among the young population,
growing disposable income, and increased emphasis on appearances.
Competition
and Competitive Advantages
We
mainly compete with other companies offering men’s wellness products, including Hims & Hers Health, Inc. and Roman. With our
Mango ED products, we compete against much larger pharmaceutical companies which offer ED branded drugs like Viagra (Pfizer) and Cialis
(marketed by Lilly ICOS LLC, a joint venture between Eli Lilly and Company and ICOS Corporation) and their generic forms. With our Mango
GROW product, we compete against the much larger pharmaceutical company Merck & Co., which offers the branded hair loss product Propecia
and Johnson & Johnson, the owner of Rogaine® - a branded form of Minoxidil. These companies have much greater resources than
we do and well-known brand names.
Our
future men’s wellness products will also likely need to compete against other traditional healthcare providers, pharmacies, and
large retailers that sell non-prescription products.
Furthermore,
we compete with other companies, which have greater resources and a greater advertising budget, and which are also selling ED related
products with either or both Tadalafil and Sildenafil (or similar products) in an oral disintegrating tablet and who are selling compounded
Minoxidil and Finasteride in both topical form (e.g., gels, foams, liquid solutions) and in oral capsule, tablet or pill form. For example,
we are aware of other companies that are currently selling oral disintegrating tablets for ED, including those using a combination of
Tadalafil and Sildenafil (the active ingredient in Viagra). However, we are not aware of any companies that are selling a compound consisting
of Minoxidil and Finasteride in an oral disintegrating tablet form.
We
intend to compete against these competitors based on our branding, advertising, unique compounding, and delivery system (i.e., our Mango
product has been designed to be taken sublingually, rather than in pill form).
Relative
to other online direct to consumer telemedicine companies that are selling both generic ED medication and generic hair loss medications,
we believe we have priced both our Mango ED products and Mango GROW product at a premium, due to the cost of compounding the product
and the use of multiple ingredients. We are currently aware of a handful of other direct to consumer companies that are also selling
compounded hair loss and ED medications and who are selling their products at a higher price than Mango’s current price. When comparing
the current market for various pharmaceutical related hair loss and ED products, we have attempted to position our pricing to be slightly
above average as we anticipate marketing or Mango ED and Mango GROW products to a demographic that we expect will pay a premium for what
we believe to be a premium product relative to the competition for the treatment of hair loss and erectile dysfunction.
Regulatory
Environment
We
produce and sell our Mango ED and Mango GROW product and plan to produce and sell our future pharmaceutical products, under an exemption
provided by Section 503A of the Federal Food, Drug, and Cosmetic Act (“FFDCA Act”). Section 503A describes the conditions
under which compounded human drug products are exempt from the FFDCA Act sections on FDA approval, current good manufacturing practice
(“cGMP”) requirements, and labeling with adequate directions for use. One of these conditions is that the drugs must be compounded
based on the receipt of valid patient-specific prescriptions; another condition limits “copying” of FDA-approved products,
which restricts compounding drugs that have the same active ingredients and route of administration as FDA-approved products that are
commercially available. The FDA also prohibits any marketing or promotional statements that are “false or misleading in any particular,”
including making any unsupported superiority claims against other products or the failure to disclose a material fact.
Notwithstanding
the above, under relevant FDA guidance, the FDA generally does not consider a compounded drug to be “essentially a copy”
of a commercially available drug if the compounded drug has a different route of administration as compared with the approved alternative,
and our Mango ED and Mango GROW products are for a different route of administration (e.g., sublingual). In addition, the FDA does not
consider a compounded drug to be “essentially a copy” of a commercially available drug if the approved product cannot be
used for the prescribed route of administration, which is available in the compounded version (which we believe it cannot, as discussed
below). Finally, we do not expect that we will be deemed to have engaged in such “copying”, because our Mango ED and Mango
GROW products are based on a prescriber’s determination for each patient that the change associated with the compounded product
(our Mango ED or Mango GROW product) produces for the patient a significant difference as compared with the commercially available drug
product. Under relevant FDA guidance, the FDA does not consider a compounded drug “essentially a copy” if a prescriber determines
that there is a change, made for an identified individual patient, which produces for that patient a significant difference from the
commercially available product.
Under
Section 503A of the FFDCA Act, it is the prescribing practitioner who determines if a compounded drug is necessary for the identified
patient and whether the change associated with the compounded product produces for the patient a significant difference as compared with
the commercially available drug product. FDA’s guidance states that the FDA generally does not intend to question prescriber determinations
that are appropriately documented. Our Mango ED and Mango GROW compounded products have been formulated as a Rapid Dissolve Tablet using
a sublingual (applied under the tongue) delivery system to bypass the stomach and liver. We believe this offers a significant difference
based on the fact that the approved versions are not available in the same route of administration (i.e., sublingual). A sublingual formulation
may be able to meet the clinical needs of a particular patient who desires a more rapid onset of action compared to an FDA-approved oral
formulation. In addition, because the prevalence of ED generally increases with age, older patients who may have difficulty swallowing
an FDA-approved oral formulation may benefit from a sublingual formulation that dissolves under the tongue.
Compounded
drugs, like our Mango ED and Mango GROW products, are not FDA-approved. This means that the FDA does not verify the safety or effectiveness
of such drugs. Instead, consumers rely on the determination of a prescribing physician that the compounded drug is necessary for the
individual patient. Compounded drugs also lack an FDA finding of manufacturing quality before such drugs are marketed.
The
FDA has the authority to impose significant restrictions on products through regulations on advertising, promotional and distribution
activities. In particular, the FDA will object to any promotional activity (including through testimonials and surrogates) that is “false
or misleading in any particular,” including the failure to disclose material facts. For example, the FDA will expect adequate substantiation
for an efficacy claim, which would require substantial evidence derived from adequate and well-controlled clinical trials. We believe
we can conduct truthful and non-misleading promotional activities, including activities involving the use of testimonials and surrogates,
with limited claims that do not require substantial evidence derived from adequate and well-controlled clinical trials and which do not
include efficacy claims.
We
are also aware of data in the scientific literature supporting how the proposed combination of the compounds which make up our Mango
ED product (i.e., Tadalafil or Sildenafil, Oxytocin, and L-arginine) might be expected to perform in ED patients. Previous clinical studies
(none of which we have paid for or undertaken ourselves) have suggested that either Sildenafil or Tadalafil and L-arginine in combination
for treatment of ED may be more effective than either compound alone-This is because L-arginine may increase nitric oxide, that in turn
may increase cyclic guanosine monophosphate, which has relaxation and vasodilation (dilatation of blood vessels) effects on smooth muscle
to assist in the treatment of ED. Furthermore, Oxytocin is a neurotransmitter linked to increased levels of social interaction, well-being,
and anti-stress effects and clinical studies suggest administration of Oxytocin may stimulate certain aspects of social interaction,
and may cause anti-anxiety and anti-stress effects.
Furthermore,
we are aware of data in the scientific literature supporting the efficacy of Minoxidil as an oral treatment (as discussed below), as
opposed to topical treatments that have been more traditionally used and marketed for hair growth to date. Topical Minoxidil and oral
Finasteride are current the standard first-line treatments for androgenetic alopecia (AGA)(male pattern baldness). Minoxidil in an oral
formulation has been previously used for the treatment of severe and uncontrolled hypertension at a dose of 10-40 mg. Unintentionally,
the early trials of oral minoxidil as an antihypertensive drug documented side effects such as hypertrichosis (excessive hair growth
anywhere on the body) and hirsutism (excess hair most often noticeable around the mouth and chin) with chronic use. A study conducted
by Ratchathorn Panchaprateep & Suparuj Lueangarun, and published in the September 24, 2020 edition of Dermatology and Therapy, found
that oral minoxidil at a dose of 5 mg taken once daily, significantly increased hair growth in men with AGA after 12 and 24 weeks of
treatment (Panchaprateep, R., Lueangarun, S. Efficacy and Safety of Oral Minoxidil 5 mg Once Daily in the Treatment of Male Patients
with Androgenetic Alopecia: An Open-Label and Global Photographic Assessment. Dermatol Ther (Heidelb) 10, 1345-1357 (2020)).
Separately,
Finasteride taken orally in the amount of 1 mg per day has shown to promote scalp hair growth and prevent further hair loss in a significant
proportion of men with male pattern hair loss (McClellan, K.J., Markham, A. Finasteride. Drugs 57, 111-126 (1999).
Neither
we, nor our representatives have had any conversations with the FDA staff regarding whether our Mango ED or Mango GROW product can be
sold pursuant to Section 503A of the FFDCA Act and future conversations with the FDA may result in the FDA staff raising issues with
such sales pursuant to Section 503A of the FFDCA, requiring certain pre-requisites or changes to our current business plan, which may
be costly or time consuming, and/or may result in us being prohibited from selling our Mango ED or Mango GROW product pursuant to Section
503A of the FFDCA Act.
Our
Contracted Telehealth Provider
In
many states, including Texas where our principal business operations are located, the corporate practice of medicine doctrine prohibits
corporations from practicing medicine and from employing physicians to provide professional medical services. Many states that recognize
this doctrine also prohibit physicians from agreeing to share the fees they receive for professional services with unlicensed entities
or individuals, a practice that is commonly known as “fee splitting.” The requirements for compliance with any applicable
corporate practice of medicine and fee splitting restrictions vary among the states. In Texas, for example, there is no statute that
expressly prohibits fee splitting, but the corporate practice of medicine doctrine has been interpreted to prohibit physicians from ceding
control over their fee structures to corporate entities or giving a substantial portion of the fees received to corporate entities.
In
order to comply with the corporate practice of medicine and fee splitting restrictions, we do not employ or directly contract with individual
physicians or physician groups, nor do we control their medical decision-making or charges. Rather, on August 1, 2022, we entered into
a Physician Services Agreement (the “Physicians Agreement”) with BrighterMD, LLC doing business as Doctegrity (“Doctegrity”),
as discussed in further detail below under, which has agreed to make available to us, healthcare professionals, to allow them to provide
clinical services directly to our future customers via telehealth. We have integrated these healthcare professionals to allow for telehealth
consultations and related services on our Mangoceuticals platform. This platform is the backbone of our business as it connects consumers
with both the medical provider and the pharmacy for fulfillment. It is also the system that we use to create marketing funnels for outgoing
marketing, customer management and support, and analytics for future sales.
Physician
Services Agreement with Doctegrity
Pursuant
to the Physicians Agreement, Doctegrity, which provides online telemedicine technology services and provides access to independently
contracted licensed physicians and providers, agreed to (a) arrange for the services of a physician or, where appropriate, a mid-level
practitioner with delegated authority from a physician, licensed in the appropriate state the practice of medicine will take place, who
will establish a physician/patient relationship with patients associated with our platform in accordance with the laws and regulations
of the appropriate state(s) and also provide physician review and assessment and quality control of our or related brands’ advertising
of services, medical questionnaires and related prescription requests; and (b) provide an asynchronous telehealth platform (and in certain
cases, synchronous capabilities in certain U.S. states where and when available and applicable) which provides patient access to licensed
physicians in the state from which the patient, who is participating under our platform, resides.
We
chose to contract with Doctegrity after reviewing and comparing the fees and services offered by similar telehealth platform companies
that facilitate visits between health care professionals and patients.
After
a patient visits our website and submits a request for a consultation with a health care professional, Doctegrity will communicate the
patient’s information to one of its affiliated physicians. Doctegrity and the physicians are responsible for conducting the telehealth
consultation and any ongoing communication with the patient in accordance with applicable laws. The physicians make a determination,
in their sole discretion, as to whether or not to prescribe our products (currently our Mango ED and Mango GROW products) to potential
customers. If the physicians prescribe our Mango ED or Mango GROW product, then the customers will pay us for our products. In turn,
Epiq Scripts, LLC, pursuant to the Master Services Agreement discussed below, is provided information on the customer and compounding
of our product, compound the product, and ship the product to customers using packaging and shipping materials which we supply.
We
pay Doctegrity for each physician visit conducted in response to request made by a patient on our website, regardless of whether the
physician prescribes our product to the patient. The fee we pay Doctegrity is fixed, set in advance and was negotiated at arms’
length after comparing the prices offered by similar services. We are not a party to any contracts between Doctegrity and any health
professionals or physician groups and do not control how Doctegrity reimburses these providers.
Although
our arrangement with Doctegrity, as summarized above, is structured to comply with applicable laws, including those restricting the corporate
practice of medicine and fee splitting, there may be a risk that a state agency, now or in the future as these laws (and interpretations
of them) evolve, would conclude that the arrangement and fee structure between Doctegrity and its contracted physicians and/or our agreement
with Doctegrity violates the corporate practice of medicine doctrine and fee splitting restrictions in Texas or in another state where
a patient who uses our Mangoceuticals platform is located.
The
Physicians Agreement has a term of one year subject to automatic one-year renewals unless and until terminated in accordance with the
Physicians Agreement, including by either party with 90 days’ prior written notice with or without cause and for cause with ten
days’ written notice.
Relationship
with Epiq Scripts
Master
Services Agreement with Epiq Scripts
On
September 1, 2022, and effective on August 30, 2022, we entered into a Master Services Agreement (the “MSA”) with Epiq Scripts,
LLC (“Epiq Scripts”), which at the time was 51%-owned by American International Holdings Corp (“American International”).
Mr. Cohen, our Chairman and Chief Executive Officer, served as the Chief Executive Officer and a director of, and had voting control
over, American International at the time of the entry into the Master Services Agreement. As discussed under “Company Information
and Formation,” our company was wholly-owned by American International until April 16, 2022, when control of our company was sold
to Cohen Enterprises, Inc. (“Cohen Enterprises”), which is owned by Mr. Cohen. Epiq Scripts was formed in January 2022, and
only began compounding drugs for patients in November 2022. On February 15, 2023, the 51% of Epiq Scripts then owned by American International
was transferred to Mr. Cohen as part of an exchange transaction, whereby Mr. Cohen agreed to cancel his preferred stock of American International,
which provided him voting control over American International, in exchange for among other assets, American International’s ownership
of Epiq Scripts. As a result, Epiq Scripts is currently 51% owned by Mr. Cohen, our Chairman and Chief Executive Officer. Additionally,
Mr. Cohen has served as the co-Manager of Epiq Scripts since January 2022.
Pursuant
to the Master Services Agreement and a related statement of work (“SOW”), Epiq Scripts agreed to provide pharmacy and related
services to us, we agreed to exclusively use Epiq Scripts as the provider of the Services (defined below) during the term of the agreement,
so long as Epiq Scripts complies with the terms of the Master Services Agreement. The agreement also includes a 30-day right of first
refusal for Epiq Scripts to provide pharmacy services for any new product that Mango may introduce during the term of the agreement.
Pursuant
to the SOW, Epiq Scripts agreed to provide for the online fulfillment, specialty compounding, packaging, shipping, dispensing and distribution
(collectively, the “Services”) of products sold exclusively via our website that may be prescribed as part of a telehealth
consultation on our platform. Epiq Scripts also agreed to provide mail service pharmacy services to us on an exclusive basis during the
term of the SOW.
We
agreed to provide Epiq Scripts with all custom packaging materials, including but not limited to, individual sachet and/or blister packaging
materials, outer box packaging, and any custom inserts and/or marketing information to accompany the prescription shipment, if any and
to provide Epiq Scripts with quarterly sales forecasts to ensure Epiq Scripts has enough packaging materials on hand to cover a 90-day
period. We agreed to pay for all direct shipping, delivery and related courier costs and to provide Epiq Scripts with direct access to
any online accounts to access and generate shipping labels for the fulfillment and delivery of our products.
The
SOW has a term through December 31, 2025, automatically renewable thereafter for successive one-year terms unless either party terminates
the agreement at least 90 days before renewal thereof and the SOW is subject to the same termination rights of the parties as set forth
in the Master Services Agreement (discussed below).
Pursuant
to the SOW, we agreed to pay Epiq Scripts certain fixed rate fees for prescription fulfillment, processing and packaging (per prescription)
and drug compounding (per pill), provided the per pill rate is reduced upon us exceeding 3,500 product packages per month.
Under
the Master Services Agreement, we are solely responsible for billing and collecting funds from our customers and Epiq Scripts is paid
out of funds that we actually collect.
We
paid Epiq Scripts a total of $60,000 upon our entry into the Master Services Agreement, comprising $45,000 as a one-time non-refundable
technology systems setup and implementation fee and $15,000 as an upfront retainer to be credited towards the future provision of pharmacy
and related services as outlined and detailed in the Master Services Agreement and SOW, of which $11,745 remained outstanding as of December
31, 2022 and $84,382 remained outstanding as of September 30, 2023. All costs related to the pharmacy services provided by Epiq Scripts
are listed as related party costs of revenues on our statement of operations.
The
Master Services Agreement has a term of five years, automatically renewable to additional one-year terms thereafter unless either party
provides the other notice of termination at least 90 days prior to the date of automatic renewal. The Master Services Agreement can be
terminated (i) upon breach of the agreement by the other party, subject to a 90-day cure right, (ii) if a party enters into bankruptcy
or fails to pay its debts as they become due, or (iii) if Epiq Scripts becomes unable to perform the services covered by the Master Services
Agreement and any statements of work associated therewith.
Epiq
Scripts is located in Texas, and has filed with the Utilization Review Accreditation Commission (“URAC”) to obtain its pharmacy
accreditation and obtained its first state license in the State of Texas in February 2022. Epiq Scripts currently has State Board of
Pharmacy (or its equivalent) licenses to operate in the District of Columbia and 47 states: Alaska, Arizona, Arkansas, Colorado, Connecticut,
Delaware, Florida, Georgia, Hawaii, Idaho, Illinois, Indiana, Iowa, Kansas, Kentucky, Louisiana, Maine, Maryland, Massachusetts, Michigan,
Minnesota, Mississippi, Missouri, Montana, Nebraska, Nevada, New Hampshire, New Jersey, New Mexico, New York, North Carolina, North Dakota,
Ohio, Oklahoma, Oregon, Pennsylvania, Rhode Island, South Dakota, Tennessee, Texas, Utah, Vermont, Virginia, Washington, West Virginia,
Wisconsin, and Wyoming and plans to eventually obtain licenses in all 50 states by the end of 2023, with some state licenses easier to
obtain and quicker to obtain than others. Although Epiq Scripts is physically located in Texas, it can ship products to customers in
each state in which it holds a license.
As
a result of the above, Epiq Scripts can currently only provide the Services to us in the District of Columbia and the 47 states described
above, and we are unable to sell products to any customers in any states other than those named above, until Epiq Scripts is able to
obtain licenses in other states and will thereafter be limited to selling products to customers only in the states in which Epiq Scripts
holds licenses.
Consulting
Agreement With Epiq Scripts
On
September 15, 2023, we entered into a Consulting Agreement (the “Consulting Agreement”) with Epiq Scripts. Pursuant to the
Consulting Agreement, Epiq Scripts agreed to provide pharmacy consulting services in connection with the Company’s global expansion
efforts, and as reasonably requested by the Company, during the term of the agreement, which is for five years, unless otherwise earlier
terminated (a) due to breach of the agreement by either party and the failure to cure such breach 30 days after written notice thereof;
(b) the mutual agreement of the parties; or (c) the date that Epiq Scripts provides the Company written notice of termination, which
may be at any time and for any reason.
In
consideration for agreeing to provide the services under the agreement, the Company agreed to pay Epiq Scripts (1) a one-time payment
of $65,000, payable within ten days of the entry into the agreement; and (2) a set fee, payable for each prescription drug pill sold
by the Company for cash, to the extent such pill must be prescribed by a medical doctor, or sold through retail pharmacies over the counter,
in jurisdictions where a doctor’s prescription is not required for the sale of such drugs, and sold in a Territory (defined below),
which consideration per pill decreases each year that the agreement is in effect, and is only payable for the first five years of the
agreement.
The
Consulting Agreement further provides that no payments are due for the sale of any prescription pills until the First Sale.
Under
the Consulting Agreement, (a) “Territory” means worldwide, except for the United States, including its territories and possessions
and the District of Columbia; and (b) “First Sale” means the date that the first commercial sale of prescription pills occurs
in the Territory.
Future
payments are also required to be offset equitably for any prescription pill sold which is later refunded, charged back, returned, or
reimbursed to a purchaser.
The
agreement includes customary representations of the parties, confidentiality and non-solicitation provisions, rights of Epiq Scripts
to audit the sales of prescription pills, subject to certain limitations and requirements, and the requirement that the Company reimburse
certain expenses of Epiq Scripts, subject to certain limitations and pre-approvals.
First
Amendment to MSA
On
September 15, 2023, we entered into a First Addendum to Master Services Agreement with Epiq Scripts (the “First Amendment”).
Pursuant
to the First Amendment, the parties agreed to amend the MSA to include certain Right of first negotiation rights and right of first refusal
rights (each as discussed below). Additionally, the First Amendment provides for certain rights to Epiq Scripts in the event that the
Company seeks to obtain pharmaceutical services in connection with certain Company products (collectively, “Pharmaceutical Services”)
in jurisdictions other than the United States, including, without limitation, Mexico and the United Kingdom, where Epiq Scripts does
not currently maintain licenses or permits (“Future Jurisdictions”, which shall also include, to the extent applicable, any
state in the United States in which Epiq Scripts does not then hold required permits or licenses for the provision of the Pharmaceutical
Services) and/or to terminate Epiq Scripts’ rights to provide exclusive Pharmaceutical Services in any current state of the United
States or Future Jurisdiction where Epiq Scripts may then be providing Pharmaceutical Services to the Company (each a “Current
Jurisdiction”).
Specifically,
the parties agreed in the First Amendment that should the Company decide to transfer any services provided by Epiq Scripts in a Current
Jurisdiction to another pharmaceutical service provider (“Transferred Services”), the Company will be required to pay Epiq
Scripts a fee of 1% of the total gross sales of all Prescription Products (defined below) by the Company resulting from the Transferred
Services in the Current Jurisdiction, for a period of the lesser of (a) five (5) years from the date the Company transferred the Transferred
Services; and (b) through the end of the term of the MSA (including where applicable, any renewal term)(the “Non-Use Fee”).
The Non-Use Fee is payable monthly in arrears, for calendar quarters, by the 15th day following the end of each calendar quarter. “Prescription
Products” means Products (as defined in the MSA) sold by the Company which must be prescribed by a medical doctor.
Notwithstanding
the above, the Non-Use Fee shall not apply, and the Company shall not be obligated to pay any Non-Use Fee (a) in the event that the Transferred
Services are provided directly by the Company or a majority-owned subsidiary of the Company; (b) in the event the Company decides to
enter into an agreement with another pharmaceutical service provider to provide Pharmaceutical Services in a Future Jurisdiction; or
(c) in connection with any services provided by any parties in any Future Jurisdictions.
The
First Amendment also provides that until the fifth anniversary of the First Amendment, the Company shall notify Epiq Scripts in writing
of any plans to (a) expand its need for pharmacy services outside of those contemplated by the MSA; (b) expand its need for pharmacy
services into a new jurisdiction which Epiq Scripts does not then operate in (including, but not limited to new countries); or (c) begin
providing pharmacy services internally (either through organic growth or acquisition). Thereafter Epiq Scripts has the right to provide
the Company written notice of its intention to provide such services (as described in (a) or (b) above, whereafter the Company is required
to discuss and negotiate such services in good faith with Epiq Scripts for a period of not less than 15 days). Otherwise, in the event
of the occurrence of an event discussed in (c) above, the Company is required to discuss the possibility of Epiq Scripts either co-operating
the pharmacy or providing management services to the Company in good faith for 15 days. In the event after such 15 day period, the Company
and Epiq Scripts cannot come to a mutually agreeable agreement, the Company is under no further obligation regarding the matter set forth
in the notice provided to Epiq Scripts.
Finally,
the First Amendment includes a requirement whereby if Epiq Scripts receives notice of any proposed fundamental transaction involving
Epiq Scripts or its assets, including any agreement, arrangement, offer or proposal (including a letter of intent, term sheet, form of
definitive agreement or definitive agreement) for an asset sale or acquisition, merger, acquisition or sale of securities, or redemption
or repurchase of securities, Epiq Scripts must provide the Company notice of such offer within three days, after which receipt the Company
will have the right of first refusal for 30 days to become the purchaser in connection with the notified transaction, on the terms, and
subject to the conditions, set forth in such notified offer and pursuant to the conditions of the First Amendment.
Recent
Events
Patent Purchase
Agreement
Effective
on April 24, 2024, the Company entered into a Patent Purchase Agreement (the “IP Purchase Agreement”), with Intramont
Technologies, Inc. (“Intramont”). Pursuant to the IP Purchase Agreement, we purchased certain patents and patent applications
owned by Intramont, related to prevention of infections, including the common cold, respiratory diseases, and orally transmitted diseases
such as human papillomavirus (HPV) (the “Patents”), in consideration for $20,000,000, which is payable to Intramont
by (a) the issuance of 980,000 shares of the Company’s newly designated 6% Series C Convertible Preferred Stock (the “Series
C Preferred Stock”), with a face value of $20.00 per share, for a total value of $19,600,000 (the “Series C Shares”);
and (b) $400,000 in cash, (i) with $200,000 payable on or before June 30, 2024, (ii) $100,000 payable on or before August 31, 2024, and
(iii) $100,000 payable on or before November 30, 2024 (collectively, the “Cash Payments”).
In
the event any of the Cash Payments have not been made on or before the due date thereof as provided above, we have 30 days to cure such
non-payment, and if not paid by the end of such 30-day period, we have the option of paying Intramont $15,000 for a thirty day extension
period for each Cash Payment.
The
IP Purchase Agreement, and the purchase of the Patents, closed on April 24, 2024, upon the parties entry into the IP Purchase Agreement,
and the Series C Shares were also issued on April 24, 2024. The IP Purchase Agreement included standard representations and warranties
and confidentiality and indemnification obligations of the parties, for a transaction of that type and size. The Company purchased the
Patents through its newly formed wholly-owned subsidiary, MangoRx IP Holdings, LLC, a Texas limited liability company.
The
IP Purchase Agreement also included a grant back license, whereby the Company provided Intramont, an irrevocable, co-exclusive, non-transferable
and non-assignable (except in the event of a change of control), non-sublicensable, worldwide, license to use the Patents for the lives
thereof (the “Grant Back-License”). The Grant Back-License is subject to Intramont paying the Company a royalty of
ten percent (10%) of gross worldwide sales of products sold by Intramont which utilize the Patents, beginning on April 24, 2025, and
continuing until the end of the life of the last Patent (the “Royalty Payments”). The Royalty Payments are to be paid
to the Company on an annual basis, within 30 days after the end of the calendar year.
Finally,
the IP Purchase Agreement granted Intramont a right of first refusal, which provides that, if at any time prior to April 24, 2027, if
we receive an offer to purchase the Patents and determine to accept such offer, or we determine to sell the Patents to a third party,
we are required to provide Intramont the right of first refusal to either match such offer, or negotiate different purchase terms for
the Patents.
The
Company intends to utilize the Patents by commencing research, development, clinical trial studies and efficacy testing on a variety
of oral applications including, but not limited to, an oral dissolvable tablet (ODT), lozenge, toothpaste and/or mouthwash.
Description
of the 6% Series C Convertible Cumulative Preferred Stock
The
Company submitted for filing to the Secretary of State of Texas, a Certificate of Designations
of Mangoceuticals, Inc. Establishing the Designations, Preferences, Limitations and Relative Rights of Its 6% Series C Convertible Cumulative
Preferred Stock (the “Series C Designation”), which was filed with the Secretary of State of Texas on April 23, 2024,
effective as of April 19, 2024 and which designated 6,250,000 shares of Series C Preferred Stock.
Subsequent
to the filing of the Series C Designation, the Company became aware of certain errors associated with section cross references and numbering
as set forth therein. As a result, on April 29, 2024, the Company filed a Certificate of Correction with the Secretary of State of Texas
correcting the prior Series C Designation, which was accepted by Texas effective as of the same date, but effective as of the original
date of filing of the Series C Designation for all purposes.
Dividend
Rights: The Series C Preferred Stock (“Series C Shares”) created under the IP Purchase Agreement include a dividend under
which each share of Series C Preferred Stock is entitled to receive, when, as and if authorized and declared by the Board of Directors
of the Company, out of any funds legally available therefor, cumulative dividends in an amount equal to (i) the 6% per annum on the stated
value (initially $20 per share)(the “Stated Value”) as of the record date for such dividend (as described in the Series
C Designation), and (ii) on an as-converted basis, any dividend or other distribution, whether paid in cash, in-kind or in other property,
authorized and declared by the Board of Directors on the issued and outstanding shares of common stock in an amount determined by assuming
that the number of shares of common stock into which such shares of Series C Preferred Stock could be converted on the applicable record
date for such dividend or distribution. Dividends payable pursuant to (i) above are payable quarterly in arrears, if, as and when authorized
and declared by the Board of Directors, or any duly authorized committee thereof, to the extent not prohibited by law, on March 31, June
30, September 30 and December 31 of each year (unless any such day is not a business day, in which event such dividends are payable on
the next succeeding business day, without accrual of interest thereon to the actual payment date), commencing on June 30, 2024. Accrued
dividends may be settled in cash, subject to applicable law, shares of common stock (valued at the closing price on the date the dividend
is due) or in-kind, by increasing the Stated Value by the amount of the quarterly dividend.
Liquidation
Preference: Upon any liquidation, dissolution or winding-up of the Company, whether voluntary or involuntary (a “Liquidation”),
the holders of the Series C Preferred Stock are entitled to receive out of the assets, whether capital or surplus, of the Company an
amount equal to the Stated Value (the “Liquidation Preference”), for each share of Series C Preferred Stock, before
any distribution or payment is made to the holders of any junior securities, but after the payment of any liquidation preference of any
holder of senior securities, including the Series B Convertible Preferred Stock, which has a preferential right to payments in liquidation,
and if the assets of the Company are insufficient to pay in full such amounts, then the entire assets to be distributed to the holders
of the Series C Preferred Stock are to be ratably distributed among the holders of the Series C Preferred Stock in accordance with the
respective amounts that would be payable on such shares if all amounts payable thereon were paid in full.
Conversion
Rights: Each holder of Series C Preferred Stock may, at its option, convert its shares of Series C Preferred Stock into that number of
shares of common stock equal to the Stated Value of such share of Series C Preferred Stock, divided by the conversion price of $10.00
per share (i.e., initially a 2-for-1 conversion ratio) (the “Conversion Price”), subject to adjustment for stock splits
and stock dividends, with any fractional shares rounded up to the nearest whole share.
The
Series C Designation includes a conversion limitation prohibiting any holder and their affiliates from converting the Series C Preferred
Stock into common stock in the event that upon such conversion their beneficial ownership of the Company’s common stock would exceed
4.999% (which can be increased as to any holder, to up to 9.999%, with 61 days prior written notice by such holder). The Series C Designation
also includes a general restriction prohibiting the issuance of more than 19.99% of the Company’s outstanding shares as of the
date of entry into the IP Purchase Agreement, without the Company’s stockholders approving such issuance(s) under the rules of
the Nasdaq Capital Market.
Voting
Rights: The Series C Preferred Stock have no voting rights, except in connection with the protective provisions as discussed within.
Protective
Provisions. So long as any shares of Series
C Preferred Stock are outstanding, the Company cannot without first obtaining the approval of the holders of a majority of the then outstanding
shares of Series C Preferred Stock, voting together as a class:
(a)
Amend any provision of the Series C Designation;
(b)
Increase or decrease (other than by redemption or conversion) the total number of authorized shares of Series C Convertible Preferred
Stock;
(c)
Amend the Certificate of Formation of the Company (including by designating additional series of Preferred Stock) in a manner which adversely
affects the rights, preferences and privileges of the Series C Preferred Stock;
(d)
Effect an exchange, or create a right of exchange, cancel, or create a right to cancel, of all or any part of the shares of another class
of shares into shares of Series C Preferred Stock; or
(e)
Alter or change the rights, preferences or privileges of the shares of Series C Preferred Stock so as to affect adversely the shares
of such series.
Redemption Rights: The Company may redeem the outstanding Series C Preferred Stock shares, from time to time, in whole or in part, at
any time after April 24, 2025, and continuing indefinitely thereafter, at the option of the Company, for cash, at the aggregate Liquidation
Preference of the shares redeemed.
Securities
Purchase Agreement
Effective on April 5, 2024 (the “Initial Closing Date”),
we agreed to definitive terms on a Securities Purchase Agreement dated April 4, 2024 (the “SPA”), with an institutional accredited
investor (the “Purchaser”), pursuant to which the Company agreed to sell to the Purchaser, and the Purchaser agreed to purchase
from the Company, 1,500 shares of Series B Preferred Stock of the Company for $1,650,000, and warrants (the “Warrants”, and
the shares of Common Stock issuable upon exercise thereof, the “Warrant Shares”), to purchase up to 3,300,000 shares of common
stock, par value $0.0001 per share of the Company (the “Common Stock”), for an aggregate purchase price of $1,500,000.
Pursuant
to the SPA, the sale of the Series B Preferred Stock and Warrants is to take place over up to three closings as follows:
| | |
Initial Stated
Value
of
Preferred
Stock
to
be
issued
by installment | | |
Warrants to
be issued | | |
Closing Date | |
Aggregate
Purchase Price | |
| Initial Closing | |
$ | 550,000 | | |
| 3,300,000 | | |
Initial Closing Date | |
$ | 500,000 | |
| Second Closing | |
$ | 275,000 | | |
| | | |
The earlier of (a) the date mutually approved by the Company and the Purchaser; and (b) three business days after the Stockholder Approval and Effectiveness | |
$ | 250,000 | |
| Third Closing | |
$ | 825,000 | | |
| | | |
The Stockholder Approval and Effectiveness | |
$ | 750,000 | |
| Total | |
$ | 1,650,000 | | |
| | | |
| |
$ | 1,500,000 | |
On
the Initial Closing Date, the Company sold the Purchaser 500 shares of Series B Preferred Stock (the “Initial Closing Shares”)
and the Warrants, for an aggregate of $500,000. As described in the table above, the sale of an additional 250 shares of Series B Preferred
Stock for $250,000, is to occur upon the earlier of the mutual approval of the Company and the Purchaser, and three business days after
the Stockholder Approval and Effectiveness (as defined below); and the sale of an additional 750 shares of Series B Preferred Stock is
expected to occur upon Stockholder Approval and Effectiveness.
As
discussed below, the Series B Preferred Stock each have an initial stated value of $1,100 per share, and as a result, the effective purchase
price of the Series B Preferred Stock shares sold, and agreed to be sold to the Purchaser, without taking into account the Warrants,
is a 10% discount to the stated value thereof.
The
consummation of the Second Closing and Third Closing contemplated by the SPA, are subject to various customary closing conditions as
well as Stockholder Approval and Effectiveness (as hereinafter defined).
Pursuant
to the SPA, the Company agreed that until Stockholder Approval (as defined below) is obtained, the Company would not issue any shares
of Common Stock upon conversion of the Series B Preferred Stock or upon exercise of the Warrants, to the extent that after giving effect
thereto, the aggregate number of shares of Common Stock that would be issued pursuant to the SPA and the ELOC (as defined below), and
the other transaction documents entered into in connection therewith would exceed 4,721,538 shares of Common Stock (representing 19.99%
of the number of shares of Common Stock issued and outstanding immediately prior to the execution of the SPA) (such maximum number of
shares, the “Exchange Cap”).
The
SPA requires us, as soon as practicable after the Initial Closing Date, but in any event no later than 30 days thereafter (the “Stockholder
Meeting Deadline”), to hold a meeting of stockholders to seek approval of a waiver of the Exchange Cap and, if needed, an increase
in the authorized number of shares of Common Stock (approval of all such proposals, the “Stockholder Approval”), by
providing each stockholder of the Company a proxy statement. If, despite the Company’s best efforts, the Stockholder Approval is
not obtained on or prior to the Stockholder Meeting Deadline, we are required to hold additional stockholder meetings, at least semi-annually
until such Stockholder Approval is obtained. “Effectiveness” means the Securities and Exchange Commission (SEC) declaring
effective the registration statement required to be filed pursuant to the Registration Rights Agreement, discussed below.
From
(a) the Initial Closing Date until 30 days after the effective date of the registration statement registering for resale all of the Warrant
Shares and shares of common stock issuable upon conversion of the Series B Preferred Stock which may be sold at the Initial Closing,
Second Closing and Third Closing (and if such Option Closing has occurred as of such date, the Option Conversion Shares)(subject to certain
cutback rights described in the Registration Rights Agreement), the Company is prohibited from (i) issuing, entering into any agreement
to issue or announcing the issuance or proposed issuance of any shares of Common Stock, Common Stock equivalents, preferred stock or
preferred stock equivalents or (ii) filing any registration statement or amendment or supplement thereto, other than the filing a registration
statement on Form S-8 in connection with any employee benefit plan; and (b) from the Initial Closing Date until 180 days after the Initial
Closing Date, the Company is prohibited from effecting or entering into an agreement to effect any issuance by the Company or any of
its subsidiaries of Common Stock, Common Stock equivalents, preferred stock or preferred stock equivalents (or a combination of units
thereof) involving a Variable Rate Transaction (as defined in the SPA), except for an equity line of credit.
The
SPA also provides the Purchaser the option (the “Option”) to purchase an additional $1,100,000 (based on the stated
value of the Series B Preferred Stock) of Series B Preferred Stock (the “Option Shares”, and the shares of common
stock issuable upon conversion of the Option Shares, the “Option Conversion Shares”). The purchase price to be paid
for the Option Shares is $1,000,000 (the “Option Price”), and the Option can be exercised at any time on or before
the date that is six months following the date of the Third Closing.
Finally,
the SPA provides that until the 18th month anniversary of the Closing Date, the Purchaser has the right to participate in
any issuance by the Company or any of its subsidiaries of Common Stock or Common Stock equivalents or any offering of debt or any other
type of financing, or a combination thereof (other certain customary exempt issuances)(each a “Subsequent Financing”),
in an amount not to exceed the amount of the Purchaser’s subscription, on the same terms, conditions and price provided for in
the Subsequent Financing.
The
Company has reserved from its duly authorized capital stock 50,000,000 of shares of Common Stock issuable upon exercise of the Warrants
and conversion of the Series B Preferred Stock.
Registration
Rights Agreement
In
connection with the SPA, the Company entered into a registration rights agreement (the “Registration Rights Agreement”)
with the Purchaser. Pursuant to the Registration Rights Agreement, the Company is required to file a resale registration statement (the
“Registration Statement”) with the SEC to register for resale of the shares of the Company’s Common Stock issuable
upon conversion of all shares of Series B Preferred Stock which may be sold at the Initial Closing, Second Closing and Third Closing
(and if such Option Closing has occurred as of such date, the Option Conversion Shares), shares of Common Stock issuable in lieu of cash
dividends which could accrue on the Series B preferred Stock for a period of two years, and the Warrant Shares, within 30 days of the
Closing Date, and to have such Registration Statement declared effective within 5 trading days after the date notified by the SEC that
the SEC is not reviewing the Registration Statement, in the event the Registration Statement is not reviewed by the SEC, or 60 days of
the Closing Date in the event the Registration Statement is reviewed by the SEC. The Company will be obligated to pay certain liquidated
damages to the Purchaser if the Company fails to file the Registration Statement when required, fails to cause the Registration Statement
to be declared effective by the SEC when required, of if the Company fails to maintain the effectiveness of the Registration Statement.
The
registration statement of which this prospectus forms a part was filed in compliance with the Company’s obligations pursuant to
the Registration Rights Agreement.
The
Company has agreed, among other things, to indemnify the Purchaser and its affiliates with respect to certain liabilities and to pay
all fees and expenses incident to the Company’s obligations under the Registration Rights Agreement.
The
SPA and the Registration Rights Agreement contain customary representations, warranties and covenants by the Company, customary conditions
to closing, indemnification obligations of the Company and the Purchaser, other obligations of the parties and termination provisions.
Description
of the Series B Convertible Preferred Stock
On
March 28, 2024, the Company submitted for filing to the Secretary of State of Texas, a Certificate of Designations, Preferences and Rights
of Series B Convertible Preferred Stock of Mangoceuticals, Inc. (the “Series B Designation”), which was filed with
the Secretary of State of Texas on April 4, 2024, effective as of March 28, 2024.
The
Series B Designation provides for the Series B Preferred Stock to have the following terms:
Series
B Preferred Stock
The
Series B Designation provides for the Series B Preferred Stock to have the following rights:
Dividend
Rights. From and after the issuance date of the Series B Preferred Stock (including in connection with the 500 shares of Series
B Preferred Stock issued on the Initial Closing Date, the Initial Closing Shares), each share of Series B Preferred Stock is entitled
to receive, when, as and if authorized and declared by the Board of Directors of the Company, out of any funds legally available therefor,
cumulative dividends in an amount equal to (i) the 10% per annum on the stated value (initially $1,100 per share)(the “Stated
Value”) as of the record date for such dividend (as described in the Series B Designation), and (ii) on an as-converted basis,
any dividend or other distribution, whether paid in cash, in-kind or in other property, authorized and declared by the Board of Directors
on the issued and outstanding Common Shares in an amount determined by assuming that the number of shares of Common Stock into which
such shares of Series B Preferred Stock could be converted on the applicable record date for such dividend or distribution.
Dividends
payable pursuant to (i) above are payable quarterly in arrears, if, as and when authorized and declared by the Board of Directors, or
any duly authorized committee thereof, to the extent not prohibited by law, on March 31, June 30, September 30 and December 31 of each
year (unless any such day is not a business day, in which event such dividends are payable on the next succeeding business day, without
accrual of interest thereon to the actual payment date), commencing on June 30, 2024.
Accrued
dividends may be settled in cash, subject to applicable law, shares of Common Stock (valued at the closing price on the date the dividend
is due) or in-kind, by increasing the stated value by the amount of the quarterly dividend.
Liquidation
Preference. Upon any liquidation, dissolution or winding-up of the Company, whether voluntary or involuntary (a “Liquidation”),
the holders of the Series B Preferred Stock are entitled to receive out of the assets, whether capital or surplus, of the Company an
amount equal to the Stated Value, plus any accrued and unpaid dividends thereon and any other fees or liquidated damages then due and
owing, for each share of Series B Preferred Stock, before any distribution or payment shall be made to the holders of any junior securities,
and if the assets of the Company shall be insufficient to pay in full such amounts, then the entire assets to be distributed to the holders
of the Series B Preferred Stock shall be ratably distributed among the holders of the Series B Preferred Stock in accordance with the
respective amounts that would be payable on such shares if all amounts payable thereon were paid in full. A Fundamental Transaction or
Change of Control Transaction (each as described in the Series B Destination) are not deemed a Liquidation.
Conversion
Rights. Each holder of Series B Preferred Stock may, at its option, convert its shares of Series B Preferred Stock (each a “Series
B Conversion”) into that number of shares of Common Stock equal to the Stated Value of such share of Series B Preferred Stock,
divided by the lesser of (x) $0.40, or (y) 90% of the average of the three lowest volume weighted average prices (“VWAPs”)
during the ten trading days preceding and ending on and including the conversion date subject to adjustment as provided in the designation
(the “Set Price” or the “Conversion Price”). Further, in no event shall the Conversion Price be
less than $0.035, subject to adjustment in the designation or the mutual agreement of the holder and the Company (the “Floor
Price”).
In
the event the Company doesn’t comply with the terms of the designation and timely issue shares of Common Stock upon conversion
to the holder, the Company is liable for damages in cash, as liquidated damages and not as a penalty, for each $5,000 of Stated Value
of preferred shares being converted, $50 per trading day (increasing to $100 per trading day on the fifth trading day and increasing
to $200 per trading day on the tenth trading day after such damages begin to accrue) for each trading day after the date due that the
shares are delivered. The designation also provides for customary buy-in rights to the holders for failure of the Company to timely deliver
conversion shares.
We
agreed to reserve not less than 50 million shares to allow for conversion of the Series B Preferred Stock, which shares have been reserved.
The
Series B Designation includes a conversion limitation prohibiting any holder and their affiliates from converting the Series B Preferred
Stock into Common Stock in the event that upon such conversion their beneficial ownership of the Company’s Common Stock would exceed
4.99%. The Series B Designation also includes a general restriction prohibiting the issuance of more than 19.99% of the Company’s
outstanding shares under certain agreements whereby the Series B Preferred Stock is expected to be issued, without the Company’s
stockholders approving such issuance(s) under Nasdaq Rule 5635(b).
The
Conversion Price is subject to anti-dilutive rights in the event that the Company issues any shares of Common Stock or Common Stock equivalents
with a value less than the then conversion price, subject to certain customary exceptions for equity plan issuances, securities already
outstanding, and certain strategic acquisitions, subject to the Floor Price.
Voting
Rights. The Series B Preferred Stock have no voting rights, except in connection with the protective provisions discussed below.
Redemption
Rights. The Series B Preferred Stock has no redemption rights.
Provisions.
So long as any shares of Series B Preferred Stock are outstanding, the Company cannot without first obtaining the approval of the holders
of a majority of the then outstanding shares of Series B Preferred Stock, voting together as a class:
(a)
Amend any provision of the Series B Designation;
(b)
Increase or decrease (other than by redemption or conversion) the total number of authorized shares of Series B Preferred
Stock;
(c)
Amend the Certificate of Formation of the Company (including by designating additional series of Preferred Stock) in a manner which adversely
affects the rights, preferences and privileges of the Series B Preferred Stock;
(d)
Effect an exchange, or create a right of exchange, cancel, or create a right to cancel, of all or any part of the shares of another class
of shares into shares of Series B Preferred Stock; or
(e)
Alter or change the rights, preferences or privileges of the shares of Series B Preferred Stock so as to affect adversely the shares
of such series.
Additionally,
so long as any Series B Preferred Stock shares remain outstanding, neither the Company nor any subsidiary thereof shall redeem, purchase
or otherwise acquire, directly or indirectly, any junior securities; pay any dividends (other than on Series B Preferred Stock), or enter
into any variable rate transaction.
Events
of Default. An “Event of Default” under the Series B Designation include the occurrence of any of the events
described below:
(a)
if at any time the Common Stock is no longer DWAC eligible;
(b)
a registration statement of the Company is not filed within sixty (60) days of the date Series B Preferred Stock is first issued;
(c)
the Company fails to obtain stockholder approval of the issuance of more than 20% of the Company’s outstanding Common Stock in
connection with the sale of certain securities within one hundred twenty (120) days of the first sale thereof;
(d)
the Company shall fail to deliver shares issuable upon a conversion prior to the fifth trading day after such shares are required to
be delivered;
(e)
the Company shall fail to have available a sufficient number of authorized and unreserved Common Stock shares to issue to any holder
upon a conversion completed under the Series B Designation;
(f)
the Company shall fail to observe or perform any other covenant, agreement or warranty contained in, or otherwise commit any breach of
any documents entered into in connection with the sale of Series B Preferred Stock, and such failure or breach shall not, if subject
to the possibility of a cure by the Company, have been cured within 10 business days after the date on which written notice of such failure
or breach shall have been delivered;
(g)
the Company shall redeem junior securities or pari passu securities;
(h)
the Company shall be party to a Change of Control Transaction (as defined in the designation);
(i)
the Company shall enter bankruptcy;
(j)
any monetary judgment, writ or similar final process shall be entered or filed against the Company, any subsidiary or any of their respective
property or other assets for more than $500,000 (provided that amounts covered by the Company’s insurance policies are not counted
toward this $500,000 threshold), and such judgment, writ or similar final process shall remain unvacated, unbonded or unstayed for a
period of thirty (30) trading days;
(k)
the electronic transfer by the Company of Common Stock shares through the Depository Trust Company is no longer available or is subject
to a “freeze” and/or “chill”, which continues for a period of five trading days; or
(l)
the common shares shall cease trading on an approved trading market, and such failure shall continue for a period of five trading days.
Following
an Event of Default, (a) the dividend rate for any dividends to be issued automatically increases to 18% per annum beginning on the date
of the Event of Default; (b) the Stated Value increases automatically by an amount equal to 17.5% of the Stated Value as of the date
of the Event of Default; and (c) the conversion price of the Series B Preferred Stock is adjusted to the lesser of (i) the then applicable
conversion price and (ii) a price per share equal to sixty five percent (65%) of the average of the three lowest trading prices for the
Company’s Common Stock during the twenty (20) trading days preceding the relevant conversion, subject to the Floor Price.
Negative
Covenants: As long as any shares of Series B Preferred Stock are outstanding, unless a simple majority of holders of the Series B
Preferred Stock have otherwise given prior written consent, the Company shall not, and shall not permit any of the subsidiaries to, directly
or indirectly:
(a)
amend its charter documents, including, without limitation, its certificate of incorporation and bylaws, in any manner that materially
and adversely affects any rights of any holder;
(b)
repay, repurchase or offer to repay, repurchase or otherwise acquire more than a de minimis number of Common Stock, Common Stock equivalents
or junior securities, other than as to (i) certain pre-approved purchases agreed to by the holders of the Series B Preferred Stock and
(ii) the repurchase common shares or common share equivalents of departing officers and directors of the Company, provided that such
repurchases shall not exceed an aggregate of $100,000 for all officers and directors for so long as the Series Preferred Stock are outstanding;
(c)
pay cash dividends or distributions on junior securities of the Company;
(d)
enter into any transaction with any affiliate of the Company which would be required to be disclosed in any public filing with the SEC,
unless such transaction is made on an arm’s-length basis and expressly approved by a majority of the disinterested directors of
the Company (even if less than a quorum otherwise required for board approval);
(e)
redeem any junior securities or pay any dividends (other than on the Series B Preferred Stock); or
(f)
enter into any agreement with respect to any of the foregoing.
Redemption
Rights. At any time while the Series B Preferred Stock are outstanding, and on any date following Stockholder Approval of the issuance
of more than 20% of the Company’s Common Stock upon conversion of the Series B Preferred Stock, the Company has the right to redeem
fifty (50%) of the Stated Value then outstanding, and an additional fifty (50%) percent of the Stated Value then outstanding upon the
written consent of the holders of the Series B Preferred Stock (each, the “Company Optional Redemption Amount”) on
the Company Optional Redemption Date (each as defined below) (a “Company Optional Redemption”). If redeemed within
ninety (90) calendar days from the date of issuance, the Series B Preferred Stock shares subject to redemption shall be redeemed by the
Company in cash at a price (the “Company Optional Redemption Price”) equal to 110% of the Stated Value being redeemed
as of the Company Optional Redemption Date, plus all accrued but unpaid dividends and all other amounts due to a holder, if any. If redeemed
within ninety-one (91) calendar days after the date of issuance, but no later than one hundred twenty (120) calendar days from the date
of issuance, the Series B Preferred Stock subject to redemption shall be redeemed by the Company in cash at a Company Optional Redemption
Price equal to 115% of the Stated Value being redeemed as of the Company Optional Redemption Date, plus all accrued but unpaid dividends
and all other amounts due to holders, if any. If redeemed after one hundred twenty (120) calendar days from the date of issuance, the
Series B Preferred Stock subject to redemption shall be redeemed by the Company in cash at a Company Optional Redemption Price equal
to 120% of the Stated Value being redeemed as of the Company Optional Redemption Date, plus all accrued but unpaid dividends and all
other amounts due to any holder, if any. The Company may deliver only one Company Optional Redemption Notice and such Company Optional
Redemption Notice shall be irrevocable.
The
Company may not deliver a Company Optional Redemption Notice, and any Company Optional Redemption Notice delivered by the Company shall
not be effective, unless all of the Equity Conditions have been met on each trading day during the period beginning on the date notice
of the redemption is provided and ending on the redemption date, which cannot be less than 10 nor more than 20 days.
“Equity
Conditions” means, during the period in question: (a) the Company shall have duly honored all conversions scheduled to occur
or occurring by virtue of one or more notices of conversion of the applicable holder on or prior to the dates so requested or required,
if any; (b) the Company shall have paid all liquidated damages and other amounts owing to the applicable holder in respect of the preferred
shares; (c) (i) there is an effective registration statement or Rule 144 can be relied upon pursuant to which either: (A) the Company
may issue conversion shares except in the case of a redemption, where only the shares being redeemed are subject to this requirement;
or (B) the holders are permitted to utilize the prospectus thereunder to resell all of the common shares issuable pursuant to certain
transaction documents (and the Company believes, in good faith, that such effectiveness will continue uninterrupted for the foreseeable
future); or (ii) all of the conversion shares issuable pursuant to the applicable transaction documents may be resold pursuant to Rule
144 without volume or manner-of-sale restrictions or current public information requirements as determined by the counsel to the Company
as set forth in a written opinion letter to such effect, addressed and acceptable to the transfer agent and the affected holders; or
(iii) all of the conversion shares may be issued to the holder pursuant to Section 3(a)(9) of the Securities Act and immediately resold
without restriction; (d) the common shares are trading on a trading market and all of the common shares issuable pursuant to the applicable
transaction documents are listed or quoted for trading on such trading market (and the Company believes, in good faith, that trading
of the common shares on a trading market will continue uninterrupted for the foreseeable future); (e) there is a sufficient number of
authorized, but unissued and otherwise unreserved, common shares for the issuance of all of the shares then issuable pursuant to the
applicable transaction documents; (f) the issuance of the common shares in question to the applicable holder would not violate the beneficial
ownership limitation set forth in the designation; (g) there has been no public announcement of a pending or proposed Fundamental Transaction
(as defined in the designation) or Change of Control Transaction (as defined in the designation) that has not been consummated; (h) the
applicable holder is not in possession of any information provided by the Company, any of its subsidiaries, or any of their officers,
directors, employees, agents or affiliates, that constitutes, or may constitute, material non-public information.
Warrants
At
the Initial Closing, the Company issued the Purchaser the Warrants to purchase up to 3,300,000 shares of Common Stock. The exercise price
of the Warrants is $0.26 (the “Exercise Price”). The Exercise Price is subject to adjustment in the event of customary
stock splits, stock dividends, combinations or similar events. If at any time following the 120th day after the Initial Closing,
there is no effective registration statement registering, or the prospectus contained therein is not available for the shares of Common
Stock issuable upon exercise of the Warrants, the Warrants can be exercised on a cashless basis and the Company is subject to certain
liquidated damages and damages as described in greater detail in the Common Share Purchase Warrant entered into on the Initial Closing
Date to evidence the Warrants (the “Warrant Agreement”).
The
Warrants are exercisable on or after October 4, 2024, and for five years thereafter.
The
Warrants contain provisions that prohibit exercise if the holder, together with its affiliates, would beneficially own in excess of 4.99%
of the number of the Company’s shares of Common Stock outstanding immediately after giving effect to such exercise. The holder
of the Warrants may increase or decrease this percentage, but not in excess of 9.99%, by providing at least 61 days’ prior notice
to the Company. In the event of certain corporate transactions, the holder of the Warrants will be entitled to receive, upon exercise
of the Warrants, the kind and amount of securities, cash or other property that the holder would have received had it exercised the Warrants
immediately prior to such transaction.
If
the Company or any subsidiary at any time while the Warrants are outstanding, shall sell, enter into an agreement to sell or grant any
option to purchase, or sell or grant any right to reprice, or otherwise dispose of or issue (or announce any offer, sale, grant or any
option to purchase or other disposition) any Common Stock or Common Stock equivalents, at an effective price per share less than the
exercise price of the Warrants then in effect (such lower price, the “Base Share Price” and such issuances collectively,
a “Dilutive Issuance”) then simultaneously with the consummation (or, if earlier, the announcement) of each Dilutive
Issuance the exercise price shall be reduced and only reduced to equal the Base Share Price. No adjustment however is to be made for
certain customary Exempt Issuances (as defined in the SPA).
The
Warrants also include customary buy-in rights in the event the Company fails to timely deliver the shares of Common Stock issuable upon
exercise thereof.
If
at any time the Warrants are outstanding there occurs any share split, share dividend, share combination recapitalization or other similar
transaction involving the Common Stock (each, a “Share Combination Event”, and such date thereof, the “Share
Combination Event Date”) and the Event Market Price (defined below) is less than the then exercise price then in effect, then
on the sixth trading day immediately following such Share Combination Event Date, the exercise price then in effect on such sixth trading
day is automatically reduced (but in no event increased) to the Event Market Price. The “Event Market Price” means,
with respect to any Share Combination Event Date, the quotient determined by dividing (x) the sum of the volume weighted average price
of the Common Stock for each of the five trading days ending and including the trading day immediately preceding the sixth trading day
after such Share Combination Event Date, divided by (y) five.
ELOC
Also
on the Initial Closing Date, the Company entered into an Equity Purchase Agreement (the “ELOC”) with the Purchaser
pursuant to which the Purchaser committed to purchase up to $25,000,000 (the “Maximum Amount”) of the Company’s
Common Stock (the “Financing”). On the Initial Closing Date, the Company issued 1,000,000 shares of the Company’s
Common Stock to the Purchaser as a commitment fee (the “Commitment Shares”). In connection with the Financing, on
the Closing Date, the Company and the Purchaser also entered into a Registration Rights Agreement (the “ELOC RRA”).
Upon
filing and effectiveness of a Registration Statement on Form S-1 to register the Advance Shares (defined below) and provided other closing
conditions are met, from time to time over the term of the ELOC, the Company shall have the right, but not the obligation, to direct
the Purchaser to purchase shares of the Company’s Common Stock (the “Advance Shares”) in a maximum amount of
one hundred percent (100%) of the average daily trading volume over the five trading days preceding the applicable advance date. At any
time and from time to time during the 2-year term of the ELOC (the “Commitment Period”), the Company may deliver a
notice to Purchaser (the “Advance Notice”) and shall deliver the Advance Shares to Purchaser via DWAC (as defined
in the ELOC) on the next trading day. The purchase price (the “Purchase Price”) for the Advance Shares shall equal
90.0% of the gross proceeds received by the Purchaser for the resale of the Advance Shares during the three consecutive trading days
immediately following the date an Advance Notice is delivered (the “Valuation Period”). The closing of an Advance
Notice shall occur within two trading days following the end of the respective Valuation Period, whereby the Purchaser shall deliver
the Investment Amount (as defined below) to the Company by wire transfer of immediately available funds. The Company shall not deliver
another Advance Notice to Purchaser within one trading day of a prior closing of Advance Shares. The “Investment Amount”
means the aggregate Purchase Price for the Advance Shares purchased by the Purchaser, minus clearing costs payable to the Purchaser’s
broker or to the Company’s transfer agent for the issuance of the Advance Shares.
The
right of the Company to issue and sell the Advance Shares to the Purchaser is subject to the satisfaction of certain closing conditions,
including, but not limited to, (i) a Registration Statement on Form S-1 registering for resale by the Purchaser of the Advance Shares
and Commitment Shares being declared effective by the SEC, (ii) accuracy of the Company’s representations and warranties, (iii)
the Company’s performance under the ELOC in all material respects, (iv) no suspension of trading or delisting of Common Stock,
(v) the limitation of the Purchaser’s beneficial ownership of the Company’s Common Stock to no more than 4.99% of the Company’s
then outstanding Common Stock, (vi) the Company maintaining its DWAC-eligible status, (vii) the Company maintaining a sufficient share
reserve, and (viii) the closing price of the Company’s Common Stock on the date the Advance Notice is received must exceed $0.15.
The
ELOC terminates upon the first to occur of April 4, 2026; the date that $25,000,000 in Advance Shares have been purchased by the Purchaser;
the date that the Company terminates the ELOC, which may be terminated in the Company’s option at any time following effectiveness
of the Registration Statement registering the resale of the Advance Shares, except that the ELOC can’t be terminated at any time
the Purchaser holds any Advance Shares; and upon the Company entering into bankruptcy protection (such period of time that the ELOC is
in place, the “Commitment Period”).
Pursuant
to the ELOC, the Purchaser agreed, that neither it, nor any of its affiliates, will in any manner whatsoever, directly or indirectly,
during the period commencing on the date of the ELOC and ending on (x) earlier of the date of the delivery of the first Advance Notice
by the Company, and (y) the date that is six months from the date the ELOC was entered into (the “Lock-Up Termination Date”),
(i) offer, pledge, announce the intention to sell, sell, contract to sell, sell any option or contract to purchase, purchase any option
or contract to sell, grant any option, right or warrant to purchase, make any short sale, or otherwise transfer or dispose of, any shares
of Common Stock; (ii) enter into any transaction that is designed to, or might reasonably be expected to, result in the transfer to another
person, in whole or in part, any of the economic consequences of ownership of any shares of Common Stock (each, a “Disposition”);
or (iii) publicly disclose the intention to make any Disposition or engage in any short sale, without the prior written consent of the
Company.
Additional
conditions to the sale of any Advance Shares to the Purchaser include that such sale, together with the Commitment Shares, and any other
shares required to be aggregated therewith pursuant to the rules of Nasdaq, would exceed 4,721,538 shares of Common Stock (representing
19.99% of the number of shares of Common Stock issued and outstanding immediately prior to the execution of the ELOC); the issuance of
the Advance Shares would cause the Purchaser to beneficially own more than 4.99% of the Company’s then outstanding Common Stock;
and/or without the prior written consent of the Purchaser, the Company has entered into a variable rate transaction.
While
the Company has the obligation to maintain such share reserve while the ELOC is effective, the Company does not have the obligation to
sell any Advance Shares to the Purchaser. Additionally, neither the Purchaser, nor any affiliate of the Purchaser acting on its behalf
or pursuant to any understanding with it, will execute any short sales during the period from the date hereof to the end of the Commitment
Period.
Registration
Rights Agreement
In
connection with the ELOC, the Company entered into the ELOC RRA with the Purchaser. Pursuant to the ELOC RRA, the Company is required
to file a resale registration statement (the “ELOC Registration Statement”) with the SEC to register all Common Stock
underlying the Advance Shares, and the Commitment Shares, within 30 days of the Closing Date, and to have such ELOC Registration Statement
declared effective within 5 trading days after the date notified by the SEC that the SEC is not reviewing the ELOC Registration Statement,
in the event the Registration Statement is not reviewed by the SEC, or 60 days of the Closing Date in the event the ELOC Registration
Statement is reviewed by the SEC. The Company will be obligated to pay certain liquidated damages to the Purchaser if the Company fails
to file the ELOC Registration Statement when required, fails to cause the ELOC Registration Statement to be declared effective by the
SEC when required, of if the Company fails to maintain the effectiveness of the ELOC Registration Statement.
The
registration statement of which this prospectus forms a part was filed in compliance with the Company’s obligations pursuant to
the ELOC RRA.
The
Company has agreed, among other things, to indemnify the Purchaser and its affiliates with respect to certain liabilities and to pay
all fees and expenses incident to the Company’s obligations under the ELOC RRA.
The
ELOC and the ELOC RRA contain customary affirmative and restrictive covenants and representations and warranties, customary conditions
to closing of the sales of the Advance Shares, as well as customary indemnification obligations by each party, including for liabilities
under the Securities Act, and other obligations of the parties and termination provisions.
Boustead
Securities, LLC served as the Company’s financial advisor in connection with the SPA and ELOC and related transactions.
The
foregoing description of the SPA, Warrants, Series B Preferred Stock, Registration Rights Agreement, ELOC and the ELOC RRA, is only a
summary of the material terms of such agreements and does not purport to be complete and is qualified in its entirety by reference to
the full text of such agreements, which are filed (or incorporated by reference) as Exhibits 10.31,
4.5, 3.3,
10.33, 10.32
and 10.34, respectively,
to the registration statement of which this prospectus forms a part.
Marketing
Agreement
On
December 10, 2023, we entered into a Marketing Agreement with Marius Pharmaceuticals, LLC (“Marius”) allowing us the use
of the trademark “Kyzatrex®” oral testosterone undecanoate softgel capsules (the “Marius Marks”), for the
purposes of branding, packaging, marketing, and selling Kyzatrex® on our website, and to be sold via our telehealth platform at www.MangoRx.com (the “Marius Agreement”). Pursuant to the Marketing Agreement, Marius granted us a non-exclusive, non-transferable, royalty-free
license to use the Marius Marks in the United States, for the purpose discussed above.
The
Marius Agreement contains customary confidentiality and indemnification provisions and has an initial term of two years, automatically
renewable thereafter for successive one year terms unless otherwise terminated (a) by Marius if we do not have at least 2,500 monthly
customers of “Kyzatrex®” oral testosterone undecanoate softgel capsules (the “Minimum Subscribers”) at least
30 days prior to the end of the initial term, (b) by either party for cause in connection with a material breach that has not been cured
within 30 business days of written notice thereof provided by the non-breaching party to the breaching party, or (c) by Marius in its
sole discretion without cause by providing at least 60 days’ prior written notice to us. Marius may also terminate the agreement
with written notice to us if we have not met at least 30% of the Minimum Subscribers within six months of the product launch date on
our website, which is anticipated to commence on or before January 31, 2024.
Within
30 days of the date the Marius Agreement is terminated (or on the date of termination, which cannot occur earlier than 60 days after
notice of termination is provided, if Marius terminates the Marius Agreement for convenience), we are required to stop and cease all
use of the Marius Marks and are required to remove all references to the Marius Marks from our advertising/promotional materials, and
signage.
During
the term of the Marius Agreement and for a period of 12 months thereafter, we agreed to not create, publish or broadcast any advertisement
or otherwise promote or market any other product containing testosterone undecanoate.
Pursuant
to the Marius Agreement, and in consideration of the license granted thereunder, we issued Marius 100,000 shares of our restricted common
stock (the “Marius Shares”) which are fully earned upon entry into the agreement. The Marius Shares were valued at $0.68
per share for a total of $68,000.
Nasdaq
Non-Compliance
Our
common stock is currently listed on Nasdaq under the symbol “MGRX”. Notwithstanding such listing, there is no guarantee that
we will be able to maintain our listing on NASDAQ for any period of time. Among the conditions required for continued listing on Nasdaq,
NASDAQ requires us to maintain at least $2.5 million in stockholders’ equity, $35 million in market value of listed securities,
or $500,000 in net income over the prior two years or two of the prior three years, to have a majority of independent directors (subject
to certain “controlled company” exemptions, which we do not currently meet), to comply with certain audit committee
requirements, and to maintain a stock price over $1.00 per share. Our stockholders’ equity is currently not above NASDAQ’s
$2.5 million minimum, as discussed below, we may not generate over $500,000 of yearly net income moving forward, we may not maintain
$35 million in market value of listed securities, we may not be able to maintain independent directors (to the extent required), and
as discussed below, we do not currently have a stock price over $1.00 per share. Nasdaq’s determination that we fail to meet the
continued listing standards of NASDAQ may result in our securities being delisted from Nasdaq.
On
October 30, 2023, we received written notice from the Listing Qualifications Department of Nasdaq notifying us that we were not in compliance
with the minimum bid price requirements set forth in Nasdaq Listing Rule 5550(a)(2) for continued listing on Nasdaq. Nasdaq Listing Rule
5550(a)(2) requires listed securities to maintain a minimum bid price of $1.00 per share, and Listing Rule 5810(c)(3)(A) provides that
a failure to meet the minimum bid price requirement exists if the deficiency continues for a period of thirty (30) consecutive business
days. Based on the closing bid price of our common stock for the thirty (30) consecutive business days from September 15, 2023 to October
27, 2023, we no longer meet the minimum bid price requirement.
The
letter did not impact the listing of our common stock on Nasdaq. Instead, the letter stated that we have 180 calendar days or until April
29, 2024, to regain compliance with Nasdaq Listing Rule 5550(a)(2). To regain compliance, the bid price of our common stock must have
a closing bid price of at least $1.00 per share for a minimum of 10 consecutive business days. If we do not regain compliance by April
29, 2024, an additional 180 days may be granted to regain compliance, so long as we meet Nasdaq’s initial listing criteria (except
for the bid price requirement)(which we do not currently meet, as we do not have stockholders’ equity of at least $5 million) and
notify Nasdaq in writing of our intention to cure the deficiency during the second compliance period by effecting a reverse stock split,
if necessary. If we do not qualify for the second compliance period or fail to regain compliance during the second 180-day period, our
common stock will be subject to delisting, at which point we would have an opportunity to appeal the delisting determination to a Hearings
Panel.
We
intend to monitor the closing bid price of our common stock and may, if appropriate, consider implementing available options to regain
compliance with the minimum bid price requirement under the Nasdaq Listing Rules.
Separately,
on November 3, 2023, we received a letter from the Listing Qualifications Department of Nasdaq notifying us that our stockholders’
equity as reported in our Quarterly Report on Form 10-Q for the period ending September 30, 2023 (the “Form 10-Q”), did not
meet the minimum stockholders’ equity requirement for continued listing on Nasdaq. Nasdaq Listing Rule 5550(b)(1) requires companies
listed on Nasdaq to maintain stockholders’ equity of at least $2,500,000. In our Form 10-Q, we reported stockholders’ equity
of $1,354,821, which is below the minimum stockholders’ equity required for continued listing pursuant to Nasdaq Listing Rule 5550(b)(1).
Additionally, we do not meet the alternative Nasdaq continued listing standards under Nasdaq Listing Rules.
This
notice of noncompliance had had no immediate impact on the continued listing or trading of our common stock on Nasdaq, which continues
to be listed and traded on Nasdaq, subject to our compliance with the other continued listing requirements.
We
submitted a plan to regain compliance in a timely manner (by December 18, 2023), and, on January 24, 2024, as reported in the Current
Report on Form 8-K filed by the Company with the Commission on January 25, 2024, Nasdaq advised the Company that it determined to grant
the Company an extension to regain compliance with the Rule until April 29, 2024.
As
a result of certain transactions, the Company believes it has regained compliance with the Rule because it believes that its stockholders’
equity exceeds $2.5 million and that it also satisfies the minimum $5 million equity requirement for initial listing on The Nasdaq Capital
Market.
On
April 25, 2024, the Company submitted a request to Nasdaq for an additional 180-day extension to regain compliance with the Minimum Bid
Price Requirement.
On
April 30, 2024, the Company received a letter from Nasdaq advising that based on the April 25, 2024 Current Report on Form 8-K, Nasdaq
has determined that the Company complies with the Rule. However, Nasdaq also advised that if the Company fails to evidence compliance
with the Rule upon filing its next periodic report it may be subject to delisting. At that time, Nasdaq will provide written notification
to the Company, which may then appeal Nasdaq’s determination to a Hearings Panel.
Separately,
on April 30, 2024, the Company received a letter from Nasdaq advising that the Company had been granted a 180-day extension to October
24, 2024, to regain compliance with the Minimum Bid Price Requirement, in accordance with Nasdaq Listing Rule 5810(c)(3)(A).
The
Company will continue to monitor the closing bid price of its common stock and will, if necessary, implement a reverse stock split of
its outstanding securities, to regain compliance with the Minimum Bid Price Requirement. The stockholders of the Company, at the March
25, 2024, special meeting of stockholders previously approved an amendment to our Certificate of Formation, as amended, to effect a reverse
stock split of our issued and outstanding shares of our common stock, by a ratio of between one-for-two to one-for-fifty, inclusive,
with the exact ratio to be set at a whole number to be determined by our Board of Directors or a duly authorized committee thereof in
its discretion, at any time after approval of the amendment and prior to March 25, 2025.
If
the Company does not regain compliance within the allotted compliance period, and/or if the Company does not demonstrate compliance with
the Rule as of the filing of its next periodic report, Nasdaq will provide notice that the Company’s common stock will be subject
to delisting. The Company would then be entitled to appeal that determination to a Nasdaq hearings panel. There can be no assurance that
the Company will regain compliance with the Minimum Bid Price Requirement during this 180-day extension.
If
we fail to satisfy another Nasdaq requirement for
continued listing (including the requirement that we maintain a minimum bid price of at least $1.00 per share, which as discussed above,
the Company is not currently in compliance with), Nasdaq could provide notice that our common stock will become subject to delisting.
In such event, Nasdaq rules would permit us to appeal the decision to reject our proposed compliance plan or any delisting determination
to a Nasdaq Hearings Panel.
Even
if we demonstrate compliance with the requirements of Nasdaq as discussed above, we will have to continue to meet other objective and
subjective listing requirements to continue to be listed on Nasdaq. Delisting from Nasdaq could make trading our common stock more difficult
for investors, potentially leading to declines in our share price and liquidity. Without a Nasdaq listing, stockholders may have a difficult
time getting a quote for the sale or purchase of our stock, the sale or purchase of our stock would likely be made more difficult, and
the trading volume and liquidity of our stock could decline. Delisting from Nasdaq could also result in negative publicity and could
also make it more difficult for us to raise additional capital. The absence of such a listing may adversely affect the acceptance of
our common stock as currency or the value accorded by other parties. Further, if we are delisted, we would also incur additional costs
under state blue sky laws in connection with any sales of our securities. These requirements could severely limit the market liquidity
of our common stock and the ability of our stockholders to sell our common stock in the secondary market. If our common stock is delisted
by Nasdaq, our common stock may be eligible to trade on an over-the-counter quotation system, such as the OTCQB Market or the OTC Pink
market, where an investor may find it more difficult to sell our stock or obtain accurate quotations as to the market value of our common
stock. In the event our common stock is delisted from Nasdaq, we may not be able to list our common stock on another national securities
exchange or obtain quotation on an over-the counter quotation system.
A
delisting of our common stock from the Nasdaq could adversely affect our business, financial condition and results of operations and
our ability to attract new investors, reduce the price at which our common stock trades, decrease, investors’ ability to make transactions
in our common stock, decrease the liquidity of our outstanding shares, increase the transaction costs inherent in trading such shares,
and reduce our flexibility to raise additional capital without overall negative effects for our stockholders.
Implications
of Being an Emerging Growth Company
We
qualify as an “emerging growth company” as defined in the federal securities laws. An emerging growth company may take advantage
of specified reduced reporting and other burdens that are otherwise applicable generally to public companies in the United States. These
provisions include:
| |
● |
a
requirement to have only two years of audited financial statements and only two years of related Management’s Discussion and
Analysis of Financial Condition and Results of Operations disclosure in this prospectus; |
| |
|
|
| |
● |
reduced
executive compensation disclosure; and |
| |
|
|
| |
● |
an
exemption from the auditor attestation requirement in the assessment of our internal control over financial reporting pursuant to
the Sarbanes-Oxley Act of 2002. |
We
may choose to take advantage of some but not all of these reduced disclosure requirements. We may take advantage of these provisions
until we are no longer an emerging growth company. We will remain an emerging growth company until the earliest to occur of: (1) (a)
December 31, 2028 (the last day of the fiscal year following the fifth anniversary of the closing of the Company’s IPO on March
21, 2023), (b) the last day of the fiscal year in which our annual gross revenues are $1.235 billion or more, or (c) the date on which
we are deemed to be a “large accelerated filer,” under the rules of the SEC, which means the market value of our equity securities
that is held by non-affiliates exceeds $700 million as of the end of our second quarter, and (2) the date on which we have issued more
than $1.0 billion in non-convertible debt during the prior three-year period.
In
addition, emerging growth companies can delay adopting new or revised accounting standards until such time as those standards apply to
private companies. See “Management’s Discussion and Analysis of Financial Condition and Results of Operations - Emerging
Growth Company and Smaller Reporting Company Status” in our Annual Report on Form 10-K. We have elected to use the extended transition
period. Accordingly, our financial statements may not be comparable to the financial statements of public companies that comply with
such new or revised accounting standards.
The
Offering
This
prospectus relates to the offer and sale from time to time of up to an aggregate of 30,014,286 shares of our common stock, par value
$0.0001 per share consisting of (i) one million (1,000,000) shares issued as a commitment fee in connection with the Company and the
selling stockholder’s entrance into the ELOC; (ii) ten million (10,000,000) shares issuable under the ELOC using
an adjusted price of $0.26 for the per share purchase price (based on a $2.6 million total purchase price divided by the average closing
price of the common stock on the Nasdaq Capital Market for the three trading days ending on April 19, 2024 ($0.29) minus a ten percent
(10%) discount for the purchase price of $0.26), (iii) three million three hundred thousand (3,300,000) shares issuable upon exercise
of warrants issued at an exercise price of $0.26 per share and (iv) 15,714,286 shares issuable upon conversion of 500 shares of Series
B Preferred Stock with each share having a stated value of $1,100 and having an assumed conversion price of the floor price of $0.035
per share.
The
number of shares ultimately offered for resale by the selling stockholders depends upon how much of the Series B Preferred Stock and
Warrants the selling stockholders elect to convert and exercise, respectively, and how many shares are issuable in connection with purchase
notices pursuant to the ELOC, and the liquidity and market price of shares of our common stock.
| Issuer |
|
Mangoceuticals
Inc. |
| |
|
|
| Common
stock to be offered by the selling stockholders |
|
The
selling stockholders are offering up to an aggregate of 30,014,286 shares of the Company’s common stock, par value $0.0001
per share. |
| |
|
|
| Common
stock outstanding prior to this offering (1) |
|
24,819,500
shares of common stock. |
| |
|
|
| Common
stock to be outstanding after the offering (1) |
|
53,833,786
shares of common stock if all the Warrants are
exercised in full, the 500 shares of Series B Preferred Stock are converted in full, and the Company sells shares of common stock
for $2.6 million pursuant to the ELOC. |
| |
|
|
| Use
of proceeds |
|
We
will not receive any proceeds from the sale of common stock by the selling stockholders. All of the net proceeds from the sale of
shares of our common stock will go to the selling stockholders as described below in the sections entitled “Selling Stockholders”
and “Plan of Distribution”. We have agreed to bear the expenses relating to the registration of the shares of common
stock for the selling stockholders. Any proceeds received by the Company from the sale of shares of common stock pursuant to the
ELOC or from the exercise of the Warrants will be used for general working capital. |
| |
|
|
| Risk
factors |
|
Investing
in our securities is highly speculative and involves a high degree of risk. You should carefully consider the information set forth
in the “Risk Factors” section beginning on page 26 before deciding to invest in our securities. |
The
number of shares of common stock to be outstanding after this offering is based on 24,819,500 shares outstanding as of May
6, 2024 and excludes:
| |
● |
exercise
of outstanding options to purchase 1,158,333 shares of common stock at an exercise
price of $1.10 per share;
|
| |
● |
exercise
of outstanding options to purchase 1,250,000 shares of common stock at an exercise price of $0.32 per share; |
| |
|
|
| |
● |
exercise
of outstanding warrants to purchase 4,685,000 shares of common stock at a weighted average exercise price of $0.51
per share; and |
| |
|
|
| |
● |
future
awards under our 2022 Equity Incentive Plan. |
Summary
Financial Data
The
following table set forth, for the periods and as of the dates indicated, our summary consolidated financial information. This information
should be read in conjunction with “Management’s Discussion and Analysis of Financial Condition and Results of Operations”
and our consolidated financial statements, including the notes thereto, included herein. Our consolidated financial statements are prepared
and presented in accordance with GAAP. Our historical results do not necessarily indicate results expected for any future periods.
| Statements of Operations Data: | |
December 31,
2023 | | |
December 31,
2022 | |
| | |
| | |
| |
| Revenues | |
$ | 731,493 | | |
$ | 8,939 | |
| Cost of revenues | |
| (299,992 | )(1) | |
| (4,089 | ) |
| Gross Profit | |
| 431,501 | | |
| 4,850 | |
| General and administrative expenses | |
| 9,650,391 | | |
| 1,991,582 | |
| Imputed interest - related party | |
| (6,473 | ) | |
| 6,473 | |
| Net loss | |
| (9,212,417 | ) | |
| (1,998,055 | ) |
| Basic and diluted loss per share | |
$ | (0.57 | ) | |
$ | (0.19 | ) |
| | |
December 31, 2023 | |
| Balance Sheet Data: | |
Actual | | |
As
Adjusted (2) | |
| | |
| | |
| |
| Cash and cash equivalents | |
$ | 739,006 | | |
$ | 3,339,006 | |
| Total assets | |
| 1,050,793 | | |
| 3,650,793 | |
| Debt | |
| - | | |
| | |
| Working capital | |
| 607,382 | | |
| 3,946,388 | |
| Accumulated deficit | |
| (11,228,173 | ) | |
| (11,228,173 | ) |
| Total stockholders’ equity | |
$ | 1,050,793 | | |
$ | 3,650,793 | |
(1)
Includes $145,092 of related party cost of revenues for the year ended December 31, 2023.
(2)
The As Adjusted column gives effect to the sale of ten million (10,000,000) shares of common stock pursuant to the ELOC at an assumed
price of $0.26 per share, less estimated offering expenses payable by us.
RISK
FACTORS
Investing
in our securities involves a high degree of risk. Before making an investment decision, you should carefully consider the risk factors
described herein, together with all of the other information appearing herein, before deciding whether to purchase any of the Company’s
stock. Our business, financial condition or results of operations could be materially adversely affected by any of these risks. The trading
price of our common stock could decline due to any of these risks, and you may lose all or part of your investment. Please also read
carefully the section titled “Cautionary Statement Regarding Forward-Looking Statements” contained elsewhere in this prospectus.
Our
business is subject to numerous risks and uncertainties, including those described below and elsewhere in this prospectus. These risks
include, but are not limited to, the following:
| |
● |
Our
need for additional funding, the availability and terms of such funding, and dilution caused thereby; |
| |
|
|
| |
● |
We
have a limited operating history, have produced only a limited amount of products and have generated only limited revenues to date; |
| |
|
|
| |
● |
Our
ability to execute our growth strategy and scale our operations and risks associated with such growth, and our ability to attract
members and customers; |
| |
|
|
| |
● |
The
effect of pandemics and governmental responses thereto on our operations, those of our vendors, our customers and the economy in
general; |
| |
|
|
| |
● |
Risks
associated with our ED product which has not been, and will not be, approved by the FDA and has not had the benefit of the FDA’s
clinical trial protocol which seeks to prevent the possibility of serious patient injury and death; |
| |
|
|
| |
● |
Risks
that the FDA may determine that the compounding of our planned products does not fall within the exemption from the FFDCA Act provided
by Section 503A; |
| |
|
|
| |
● |
Our
significant reliance on related party transactions and risks associated with such related party relationships and agreements; |
| |
|
|
| |
● |
The
effect of data security breaches, malicious code and/or hackers; |
| |
|
|
| |
● |
Competition
and our ability to create a well-known brand name; |
| |
|
|
| |
● |
Changes
in consumer tastes and preferences; |
| |
|
|
| |
● |
Material
changes and/or terminations of our relationships with key parties; |
| |
● |
Significant
product returns from customers, product liability, recalls and litigation associated with tainted products or products found to cause
health issues; |
| |
|
|
| |
● |
Our
ability to innovate, expand our offerings and compete against competitors which may have greater resources; |
| |
|
|
| |
● |
Our
Chairman and Chief Executive Officer, Jacob D. Cohen, has significant voting control over the company which may deter some investors; |
| |
|
|
| |
● |
Our
ability to prevent credit card and payment fraud; |
| |
|
|
| |
● |
Risks
associated with inflation, and increases in interest rates and economic downturns, including potential recessions, as well as macroeconomic,
geopolitical, health and industry trends, pandemics, acts of war (including the ongoing Ukraine/Russian conflict and Israel/Hamas
conflict) and other large-scale crises; |
| |
|
|
| |
● |
The
risk of unauthorized access to confidential information; |
| |
|
|
| |
● |
Our
ability to protect our intellectual property and trade secrets, claims from third-parties that we have violated their intellectual
property or trade secrets and potential lawsuits in connection therewith; |
| |
|
|
| |
● |
Our
and our providers’ ability to comply with government regulations, changing regulations and laws, penalties associated with
any non-compliance (inadvertent or otherwise), the effect of new laws or regulations, and our ability to comply with such new laws
or regulations; |
| |
|
|
| |
● |
Our
reliance on our current management and the terms of their employment agreements with us; |
| |
|
|
| |
● |
The
outcome of future lawsuits, litigation, regulatory matters or claims; |
| |
|
|
| |
● |
The
fact that certain recent initial public offerings of companies with public floats comparable to the public float of the Company have
experienced extreme volatility that was seemingly unrelated to the underlying performance of the respective company; and the fact
that we may experience similar volatility, which may make it difficult for investors to assess the value of our common stock; |
| |
|
|
| |
● |
Certain
terms and provisions of our governing documents which may prevent a change of control, and which provide for indemnification of officers
and directors, limit the liability of officers or directors, and provide for the board of director’s ability to issue blank
check preferred stock; and |
| |
|
|
| |
● |
The
volatile nature of the trading price of our common stock; dilution experienced by investors in the offering; and dilution which may
be caused by future sales of securities. |
Risks
Related to our Operating History and Need for Funding
We
were recently formed, have a limited operating history and have generated only limited revenues to date and there is no assurance that
we can generate revenues or sell any commercial amount of our products in the future. We will need to raise additional funding to support
our operations in the future.
We
were only recently formed and have a limited operating history. We launched our website in mid-November 2022. To date we have sold only
a small number of products and generated only limited revenues and have not sold sufficient quantities of our Mango ED or Mango GROW
products to support our operations. There is no assurance that we can generate revenues sufficient to support our operations, and even
if additional revenues are generated, there is no assurance that we can generate sufficient net income to support our operations. As
reflected in the accompanying financials, the Company had a net loss of $9,170,435 for the year ended December 31, 2023 and an accumulated
deficit of $11,186,191 as of December 31, 2023. Additionally, the Company had a net loss of $1,998,055 for the year ended December 31,
2022 and an accumulated deficit of $2,015,756 as of December 31, 2022.
We
have experienced recurring net losses since inception. We believe that we will continue to incur substantial operating expenses in the
foreseeable future as we continue to invest to bring our Mango ED and Mango GROW products to market and to attract customers, expand
the product offerings and enhance technology and infrastructure. These efforts may prove more expensive than we anticipate, and we may
not succeed in generating commercial revenues or net income to offset these expenses. Accordingly, we may not be able to achieve profitability,
and we may incur significant losses for the foreseeable future. Our independent registered public accounting firm included an explanatory
paragraph in its report on our financial statements as of December 31, 2023, included herein. As of the date of this prospectus, our
current capital resources, combined with the net proceeds from the offering, are expected to be sufficient for us to fund operations
for the next 12 months. We will need funding in addition to the funding raised in our IPO and Follow On Offering to support our operations
in the future. We may also seek to acquire additional businesses or assets in the future, which may require us to raise funding. We currently
anticipate such funding, if required, being raised through the offering of debt or equity. Such additional financing may not be available
on favorable terms, if at all. If debt financing is available and obtained, our interest expense may increase and we may be subject to
the risk of default, depending on the terms of such financing. If equity financing is available and obtained it may result in our shareholders
experiencing significant dilution. If such financing is unavailable, we may be forced to curtail our business plan, which may cause the
value of our securities to decline in value.
Since
we have a limited operating history, it is difficult for potential investors to evaluate our business and our business is in a relatively
new consumer product segment, which is difficult to forecast.
Our
limited operating history in the health and wellness industry may hinder our ability to successfully meet our objectives and makes it
difficult for potential investors to evaluate our business or prospective operations. As an early-stage company, we are subject to all
the risks inherent in the financing, expenditures, operations, regulatory compliance, complications and delays inherent in a new business.
Accordingly, our business and success face risks from uncertainties faced by developing companies in a competitive environment. The likelihood
of our success must be considered in light of the problems, expenses, difficulties, regulatory challenges, complications and delays frequently
encountered in connection with the formation of a new business, the development of a new strategy and the competitive environment in
which we operate. There can be no assurance that our efforts will be successful or that we will ultimately be able to attain profitability.
Additionally,
our industry segment is relatively new, and is constantly evolving. As a result, there is a lack of available information with which
to forecast industry trends or patterns. There is no assurance that sustainable industry trends or preferences will develop that will
lead to predictable growth or earnings forecasts for individual companies or the industry segment as a whole. We are also unable to determine
what impact future governmental regulation may have on trends and preferences or patterns within our industry segment.
We
need additional capital which may not be available on commercially acceptable terms, if at all, and this raises questions about our ability
to continue as a going concern.
We
need additional capital to support our operations and continue to market and commercialize our current Mango ED and Mango GROW products.
We may also require additional funding in the future to support our operations, expand our product line, pay expenses, or expand or complete
acquisitions. The most likely source of future funds presently available to us will be through the sale of equity capital or debt. Any
sale of equity or convertible equity or debt will result in dilution to existing shareholders. Furthermore, we may incur debt in the
future, and may not have sufficient funds to repay our future indebtedness or may default on our future debts, jeopardizing our business
viability.
We
may not be able to borrow or raise additional capital in the future to meet our needs or to otherwise provide the capital necessary to
expand our operations and business, which might result in the value of our securities decreasing in value or becoming worthless. Additional
financing may not be available to us on terms that are acceptable. Consequently, we may not be able to proceed with our intended business
plans. Obtaining additional financing contains risks, including:
| |
● |
additional
equity financing may not be available to us on satisfactory terms and any equity or convertible equity or debt we are able to issue
could lead to dilution for current shareholders; |
| |
|
|
| |
● |
loans
or other debt instruments may have terms and/or conditions, such as interest rate, restrictive covenants and control or revocation
provisions, which are not acceptable to management or our directors; |
| |
|
|
| |
● |
the
current environment in capital markets combined with our capital constraints may prevent us from being able to obtain adequate debt
financing; and |
| |
|
|
| |
● |
if
we fail to obtain required additional financing to commercialize our products and grow our business, we would need to delay or scale
back our business plan, reduce our operating costs, or delay product launches, each of which would have a material adverse effect
on our business, future prospects, and financial condition. |
Additionally,
we may have difficulty obtaining additional funding, and we may have to accept terms that would adversely affect our shareholders. For
example, the terms of any future financings may impose restrictions on our right to declare dividends (provided that none are currently
planned) or on the manner in which we conduct our business. Additionally, lending institutions or private investors may impose restrictions
on a future decision by us to make capital expenditures, acquisitions or significant asset sales. If we are unable to raise additional
funds, we may be forced to curtail or even abandon our business plan.
Risks
Related to Our Business Activities
We
may not be able to successfully commercialize our Mango ED or Mango GROW products or any other potential future men’s wellness
products.
We
may not be able to effectively commercialize our Mango ED or Mango GROW products or any other potential future men’s wellness products.
If we are unable to successfully commercialize our Mango ED and Mango GROW products or successfully develop, produce, launch and commercialize
any other potential future men’s wellness products, our ability to generate product sales will be severely limited, which will
have a material adverse impact on our business, financial condition, and results of operations.
We
expect to face intense competition, often from companies with greater resources and experience than we have.
The
health, wellness, and telemedicine industries are highly competitive and subject to rapid change. The industries continue to expand and
evolve as an increasing number of competitors and potential competitors enter the market. Many of these competitors and potential competitors
have substantially greater financial, technological, managerial and research and development resources and experience than we have. We
mainly compete with other companies offering men’s wellness products, including Hims & Hers Health, Inc. and Roman, and with
our Mango ED products, we are also competing against much larger pharmaceutical companies who offer ED branded drugs like Viagra (Pfizer)
and Cialis (marketed by Lilly ICOS LLC, a joint venture between Eli Lilly and Company and ICOS Corporation) and their generic forms.
With our Mango GROW product, we compete against the much larger pharmaceutical company Merck & Co., which offers the branded hair
loss product Propecia, and Johnson & Johnson, the owner of Rogaine® - a branded form of Minoxidil. The majority of these competitors
and potential competitors have more experience than we have in the development of health and wellness services and products. In addition,
our planned services and products will compete with service and product offerings from large and well-established companies that have
greater marketing and sales experience and capabilities than we or the parties with which we contract have. If we are unable to compete
successfully, we may be unable to grow and sustain our revenue.
We
believe that our ability to compete depends upon many factors both within and beyond our control, including:
| |
● |
our
marketing efforts; |
| |
|
|
| |
● |
the
flexibility and variety of our product offerings relative to our competitors, and our ability to timely launch new product initiatives; |
| |
|
|
| |
● |
the
quality and price of products offered by us and our competitors; |
| |
|
|
| |
● |
our
reputation and brand strength relative to our competitors; |
| |
|
|
| |
● |
customer
satisfaction; |
| |
|
|
| |
● |
the
size and composition of our customer base; |
| |
|
|
| |
● |
the
convenience of the experience that we provide; |
| |
|
|
| |
● |
our
ability to comply with, and manage the costs of complying with, laws and regulations applicable to our business; and |
| |
|
|
| |
● |
our
ability to cost-effectively source and distribute the products we offer and to manage our operation. |
Many
competitors also have longer operating histories, and will have larger fulfillment infrastructures, greater technical capabilities, faster
shipping times, lower-cost shipping, lower operating costs, greater financial, marketing, institutional and other resources and larger
consumer bases than we do. These factors may also allow our competitors to derive greater revenue and profits from their existing consumer
bases, acquire consumers at lower costs or respond more quickly than we are able to, to new or emerging technologies and changes in product
trends and consumer shopping behavior. These competitors may engage in more extensive research and development efforts, enter or expand
their presence in any or all of the ecommerce or retail channels where we compete, undertake more far-reaching marketing campaigns, and
adopt more aggressive pricing policies, which may allow them to build larger consumer bases or generate revenue from their existing consumer
bases more effectively than we are able to. As a result, these competitors may be able to offer comparable or substitute products to
consumers at similar or lower costs. This could put pressure on us to lower our prices, resulting in lower revenue and margins or cause
us to lose market share even if we lower prices.
Furthermore,
companies with greater resources or more well-known brand names may attempt to compete with us, and as a result, we may lose current
or potential customers and may be unable to generate sufficient revenues to support our operations, any one of which could have a material
adverse effect on our ability to grow and our results of operations.
We
may not successfully compete with larger competitors that have greater financial, sales, technical and other resources. Companies with
greater resources may acquire our competitors or launch new products, and they may be able to use their resources and scale to respond
to competitive pressures and changes in consumer preferences by reducing prices or increasing promotional activities, among other things.
If
we fail to successfully provide a good customer experience, including by developing new product offerings, our ability to attract members
and customers may be materially adversely affected.
Our
ability to obtain customers and retain future customers, attract customers and increase customer engagement with us will depend in part
on our ability to successfully implement and improve our customer experience, including by continuing to create and introduce new product
offerings, improving upon and enhancing our existing product offerings and strengthening our customers interactions with our brand and
products. If new or enhanced product offerings are unsuccessful, we may be unable to attract or retain customers and our operating results
could be materially adversely affected. Furthermore, new or shifting customer demands, tastes or interests, superior competitive offerings
or a deterioration in our product offering quality or our ability to bring new or enhanced product offerings to market quickly and efficiently
could negatively affect the attractiveness of our products and the economics of our business and require us to make substantial changes
to and additional investments in our product offerings or business model.
Counterfeit
versions of our products could harm our customers and have a negative impact on our revenues, earnings, reputation and business.
Our
industry is subject to illegal counterfeiting and the presence of counterfeit products in certain of our markets and over the Internet.
Third parties may illegally distribute and sell counterfeit versions of our products, which do not meet our manufacturing and testing
standards, and which contain varying ingredients. To customers counterfeit products may be visually indistinguishable from the authentic
version. Counterfeit products pose a risk to customer health and safety because of the conditions under which they are manufactured as
well as the lack of regulation of their ingredients. The sale of counterfeit products could adversely impact our business and reputation
by impacting customer confidence in our authentic products, potentially resulting in lost sales, product recalls, and an increased threat
of litigation.
We
may expend our limited resources to pursue particular products or services and may fail to capitalize on products or services that may
be more profitable or for which there is a greater likelihood of success.
Because
we have limited financial and managerial resources, we must focus our efforts on particular service programs and products. As a result,
we may forego or delay pursuit of opportunities with other services or products that later prove to have greater commercial potential.
Our resource allocation decisions may cause us to fail to capitalize on viable commercial products or profitable market opportunities.
Any such failure could result in missed opportunities and/or our focus on products or services with low market potential, which would
harm our business and financial condition. Our current use of proceeds is specifically focused on among other things, the marketing and
selling of our current Mango ED and Mango GROW products and includes capital allocated for future products or services anticipated to
be sold in the future under the ‘Mango’ label and brand.
We
have entered into a Master Services Agreement and Statement of Work with Epiq Scripts, LLC, a related party, which entity is currently
licensed to provide pharmacy services in only 47 states and the District of Columbia.
As
described in greater detail under “Business-Material Agreements-Master Services Agreement with Epiq Scripts” and “-First
Amendment to MSA,” we have entered into a Master Services Agreement and SOW for Epiq Scripts, a related party, 51% owned and controlled
by Jacob D. Cohen, our Chairman and Chief Executive Officer, to provide us pharmacy and compounding services. Epiq Scripts has filed
with the Utilization Review Accreditation Commission (“URAC”) to obtain its pharmacy accreditation and has State Board of
Pharmacy (or its equivalent) licenses in the District of Columbia and 47 states: Alaska, Arizona, Arkansas, Colorado, Connecticut, Delaware,
Florida, Georgia, Hawaii, Idaho, Illinois, Indiana, Iowa, Kansas, Kentucky, Louisiana, Maine, Maryland, Massachusetts, Michigan, Minnesota,
Mississippi, Missouri, Montana, Nebraska, Nevada, New Hampshire, New Jersey, New Mexico, New York, North Carolina, North Dakota, Ohio,
Oklahoma, Oregon, Pennsylvania, Rhode Island, South Dakota, Tennessee, Texas, Utah, Vermont, Virginia, Washington, West Virginia, Wisconsin,
and Wyoming. It is also in the process of applying for additional state licenses and plans to eventually obtain licenses in all 50 states
by the end of the first quarter of 2024. As a result of the above, Epiq Scripts can currently only provide the Services to us in the
47 states described above and the District of Columbia, and we are unable to sell products to any customers in any states other than
those 47 states and the District of Columbia, until Epiq Scripts is able to obtain licenses in other states and is limited to selling
products to customers only in the states in which Epiq Scripts holds licenses.
The
Master Services Agreement does not address product liability claims which may result in us bringing legal claims or actions against Epiq
Scripts to attempt to seek indemnification or contribution for product liability claims.
Each
party to the Master Services Agreement agreed to indemnify, defend, and hold harmless the other and the other party’s officers,
directors, shareholders, employees, and agents from and against any and all nonparty claims, or actions for damages, liabilities (including
strict liability), penalties, costs and expenses (including reasonable legal fees, expenses and costs) to the proportionate extent caused
by (1) the negligence or willful misconduct of the indemnitor or any of its employees or agents in connection with the performance of
the agreement, or (2) any breach of any representation, warranty or covenant under the agreement by the indemnitor or any of its employees
or agents. Additionally, the parties agreed that neither party will be liable to the other for special, incidental, or exemplary damages,
subject to certain limited exceptions. The Master Services Agreement does not address product liability claims or assign any rights of
indemnification or contribution in connection therewith. As a result, in the event of product liability claims, we may be forced to bring
legal claims or actions against Epiq Scripts to attempt to seek indemnification or contribution for product liability claims, to the
extent that we are sued in connection with such claims and Epiq Scripts isn’t sued or that we are found primarily liable for such
claims. Such claims may be costly, time consuming, and may not ultimately result in a favorable outcome to us, all of which may have
an adverse effect on the value of our securities.
We
currently owe certain rights to Epic Scrips under the Management Services Agreement which may limit our future operations and/or have
a material adverse effect on our operations and cash flow.
As
described in greater detail under “Business-Material Agreements-Master Services Agreement with Epiq Scripts” and “-First
Amendment to MSA,” we have entered into a Master Services Agreement and SOW for Epiq Scripts, a related party, 51% owned and controlled
by Jacob D. Cohen, our Chairman and Chief Executive Officer, to provide us pharmacy and compounding services. Pursuant to the Master
Services Agreement and a related SOW, Epiq Scripts agreed to provide pharmacy and related services to us, we agreed to exclusively use
Epiq Scripts as the provider of online fulfillment, specialty compounding, packaging, shipping, dispensing and distribution services
relating to products sold exclusively via our website, that may be prescribed as part of a telehealth consultation on our platform, during
the term of the Master Services Agreement, so long as Epiq Scripts complies with the terms of the Master Services Agreement. The agreement
also includes a 30-day right of first refusal for Epiq Scripts to provide pharmacy services for any new product that Mango may introduce
during the term of the Master Services Agreement.
Pursuant
to the Master Services Agreement, as amended, Epiq Scripts has certain rights in the event that the Company seeks to obtain pharmaceutical
services in connection with certain Company products (collectively, “Pharmaceutical Services”) in jurisdictions other than
the United States, including, without limitation, Mexico and the United Kingdom, where Epiq Scripts does not currently maintain licenses
or permits (“Future Jurisdictions”, which shall also include, to the extent applicable, any state in the United States in
which Epiq Scripts does not then hold required permits or licenses for the provision of the Pharmaceutical Services) and/or to terminate
Epiq Scripts’ rights to provide exclusive Pharmaceutical Services in any current state of the United States or Future Jurisdiction
where Epiq Scripts may then be providing Pharmaceutical Services to the Company (each a “Current Jurisdiction”).
Specifically,
should the Company decide to transfer any services provided by Epiq Scripts in a Current Jurisdiction to another pharmaceutical service
provider (“Transferred Services”), the Company will be required to pay Epiq Scripts a fee of 1% of the total gross sales
of all Prescription Products (defined below) by the Company resulting from the Transferred Services in the Current Jurisdiction, for
a period of the lesser of (a) five (5) years from the date the Company transferred the Transferred Services; and (b) through the end
of the term of the Master Services Agreement (including where applicable, any renewal term)(the “Non-Use Fee”). The Non-Use
Fee is payable monthly in arrears, for calendar quarters, by the 15th day following the end of each calendar quarter. “Prescription
Products” means Products (as defined in the Master Services Agreement) sold by the Company which must be prescribed by a medical
doctor.
Notwithstanding
the above, the Non-Use Fee shall not apply, and the Company shall not be obligated to pay any Non-Use Fee (a) in the event that the Transferred
Services are provided directly by the Company or a majority-owned subsidiary of the Company; (b) in the event the Company decides to
enter into an agreement with another pharmaceutical service provider to provide Pharmaceutical Services in a Future Jurisdiction; or
(c) in connection with any services provided by any parties in any Future Jurisdictions.
Pursuant
to the Master Services Agreement, as amended, until September 15, 2028, the Company is required to notify Epiq Scripts in writing of
any plans to (a) expand its need for pharmacy services outside of those contemplated by the Master Services Agreement; (b) expand its
need for pharmacy services into a new jurisdiction which Epiq Scripts does not then operate in (including, but not limited to new countries);
or (c) begin providing pharmacy services internally (either through organic growth or acquisition). Thereafter Epiq Scripts has the right
to provide the Company written notice of its intention to provide such services (as described in (a) or (b) above, whereafter the Company
is required to discuss and negotiate such services in good faith with Epiq Scripts for a period of not less than 15 days). Otherwise,
in the event of the occurrence of an event discussed in (c) above, the Company is required to discuss the possibility of Epiq Scripts
either co-operating the pharmacy or providing management services to the Company in good faith for 15 days. In the event after such 15
day period, the Company and Epiq Scripts cannot come to a mutually agreeable agreement, the Company is under no further obligation regarding
the matter set forth in the notice provided to Epiq Scripts.
The
rights and obligations set forth above could have a material adverse effect on the Company, its plans for future products and expansions,
or make such future products or expansion more costly or time consuming.
We
currently exclusively rely, and continue to exclusively rely, on Epiq Scripts, a related party entity with a limited operating history,
for our pharmacy compounding services.
As
disclosed herein, we have entered into a Master Services Agreement with Epiq Scripts, a related party, 51% owned and controlled by Jacob
D. Cohen, our Chairman and Chief Executive Officer, to operate as our sole and exclusive licensed pharmacy to compound our Mango ED and
Mango GROW products to customers, assuming such Mango ED and Mango GROW products are prescribed by physicians pursuant to our agreement
with Doctegrity. Epiq Scripts was only formed in January 2022, and has only been compounding drugs for patients for a short period of
time. We currently exclusively rely, and continue to exclusively rely, on Epiq Scripts. We face risks relying on a newly formed pharmacy
with limited operations. Those risks include risks that Epiq Scripts will not be able to follow applicable regulatory guidelines relating
to, will not be able to timely or cost effectively complete, or may not correctly, fulfill, specialty compound, package, ship, dispense
and/or distribute our Mango ED and Mango GROW products. If Epiq Scripts is not able to scale its operations to meet the demand of our
operations, or is unable to undertake any of the actions described above, our business may be materially and adversely affected, we may
need to find a new partner pharmacy, which may charge us more money for its services or may not have as favorable contract terms, we
may be delayed or prevented from selling our Mango ED and Mango GROW products, and may face fines, penalties or litigation. In the event
of the occurrence of any of the above, the value of our securities may decline in value or become worthless.
The
use of social media and influencers may materially and adversely affect our reputation or subject us to fines or other penalties.
We
use third-party social media platforms as part of our marketing strategy. We also maintain relationships with social media influencers.
As existing e-commerce and social media platforms continue to rapidly evolve and new platforms develop, we expect to maintain a presence
on these existing platforms and expect them to be an important part of our marketing strategy. If we are unable to cost-effectively use
social media platforms as marketing tools, if the social media platforms we use change their policies or algorithms, or if evolving laws
and regulations limit how we can market through these channels, if at all, we may not be able to fully optimize our use of such platforms
and our ability to retain current customers and acquire new customers may suffer. Any such failure could adversely affect our reputation,
revenue, and results of operations.
In
addition, an increase in the use of social media for product promotion and marketing may increase the burden on us to monitor compliance
related thereto, and increase the risk that such materials could contain problematic product or marketing claims in violation of applicable
regulations. For example, in some cases, the Federal Trade Commission has sought enforcement action where an endorsement has failed to
clearly and conspicuously disclose a financial relationship or material connection between an influencer and an advertiser. We do not
control the content of what our influencers post on social media, and if we were held responsible for any false, misleading, or otherwise
unlawful content of their posts or their actions, we could be fined or subjected to other monetary liabilities or required to alter our
practices, which could have an adverse impact on our business, reputation, cash flows and ability to operate.
Negative
commentary regarding our business, or influencers who endorse our products and other third parties who are affiliated with or endorse
us, may also be posted on social media platforms. Influencers with whom we maintain endorsement arrangements could engage in behavior
or use their platforms to communicate with our customers in a manner that reflects poorly on our brand and may be attributed to us or
otherwise adversely affect our reputation. Any such negative commentary could impact our reputation or brand and affect our ability to
attract and retain customers, which could have a material adverse effect on our business and results of operations.
Our
business depends on our brand, and any failure to maintain, protect or enhance our brand, including as a result of events outside our
control, could materially adversely affect our business.
We
believe our future success depends on our ability to maintain and grow the value of the “Mango” brand. Maintaining, promoting
and positioning our brand and reputation will depend on, among other factors, the success of our marketing and merchandising efforts
and our ability to provide a consistent, high-quality customer experience. Any negative publicity, regardless of its accuracy, could
materially adversely affect our business. Brand value is based in large part on perceptions of subjective qualities, and any incident
that erodes the loyalty of our customers, including adverse publicity or a governmental investigation or litigation, could significantly
reduce the value of our brand and significantly damage our business.
The
value of our brand also depends on effective customer support to provide a high-quality customer experience, which requires significant
personnel expense. If not managed properly, this expense could impact our profitability. Failure to manage or train our own or outsourced
customer support representatives properly, or our inability to hire sufficient customer support representatives could result in lower-quality
customer support and/or increased customer response times, compromising our ability to handle customer complaints effectively.
Our
ability to gain and increase market acceptance and generate commercial revenues is subject to a variety of risks, many of which are out
of our control.
Our
Mango ED and Mango GROW products and any other potential future men’s wellness products may not gain or increase market acceptance
among physicians, patients, healthcare payors or the medical community. We believe that the degree of market acceptance and our ability
to generate commercial revenues from such products will depend on a number of factors, including:
| |
● |
our
ability to expand the use of our products through targeted patient and physician education; |
| |
|
|
| |
● |
competition
and timing of market introduction of competitive products; |
| |
|
|
| |
● |
quality,
safety and efficacy in the approved setting; |
| |
|
|
| |
● |
prevalence
and severity of any side effects, including those of the components of our products; |
| |
|
|
| |
● |
emergence
of previously unknown side effects, including those of the generic components of our products; |
| |
|
|
| |
● |
potential
or perceived advantages or disadvantages over alternative treatments; |
| |
|
|
| |
● |
the
convenience and ease of purchasing the product, as perceived by potential patients; |
| |
|
|
| |
● |
strength
of sales, marketing and distribution support; |
| |
|
|
| |
● |
price,
both in absolute terms and relative to alternative treatments; |
| |
|
|
| |
● |
the
effectiveness of any future collaborators’ sales and marketing strategies; |
| |
|
|
| |
● |
the
effect of current and future healthcare laws; |
| |
|
|
| |
● |
availability
of coverage and reimbursement from government and other third-party payors; |
| |
|
|
| |
● |
recommendations
for prescribing physicians to complete certain educational programs for prescribing drugs; |
| |
|
|
| |
● |
the
willingness of patients to pay out-of-pocket in the absence of government or third-party coverage; and |
| |
|
|
| |
● |
product
labeling, product insert, or new studies or trial requirements of the FDA or other regulatory authorities. |
Our
Mango ED and Mango GROW and/or future products may fail to achieve market acceptance or generate significant revenue to achieve sustainable
profitability. In addition, our efforts to educate the medical community and third-party payors on the safety and benefits of our drugs
may require significant resources and may not be successful.
We
may be unable to scale our operations fast enough to bring down our cost of sales and generate revenues sufficient to support our operations.
We
believe that in general, the faster we are able to scale up our operations, the lower our cost of sales, as a percentage of revenue,
will be, as we believe that certain economies of scale exist with our operations. If we are unable to grow our business fast enough to
take advantage of these economies of scale, our operations may suffer, and we may not be profitable.
Economic
downturns or a change in consumer preferences, perception and spending habits could limit consumer demand for our products and negatively
affect our future business.
The
products that we sell and plan to sell in the future (including our Mango ED and Mango GROW products) may be adversely affected from
time to time by economic downturns that impact consumer spending, including discretionary spending. Future economic conditions such as
employment levels, business conditions, housing starts, market volatility, interest rates, inflation rates, energy and fuel costs and
tax rates, or our actions in response to these conditions, such as price increases, could reduce consumer spending or change consumer
purchasing habits.
Our
performance depends significantly on factors that may affect the level and pattern of consumer spending in the markets in which we operate.
Such factors include consumer preference, consumer confidence, consumer income, consumer perception of the safety and quality of our
future products and shifts in the perceived value for our products relative to alternatives. A general decline in the consumption of
our future products could occur at any time as a result of change in consumer preference, perception, confidence and spending habits,
including an unwillingness to pay a premium or an inability to purchase our products due to financial hardship or increased price sensitivity,
which may be exacerbated by inflationary pressures, interest rates, and economic uncertainty. If consumer preferences shift away from
our products, our business, financial condition and results of operations could be adversely affected.
The
success of our products depends on a number of factors including our ability to accurately anticipate changes in market demand and consumer
preferences, our ability to differentiate the quality of our future products from those of our competitors, and the effectiveness of
our marketing and advertising campaigns for our products. We may not be successful in identifying trends in consumer preferences and
developing products that respond to such trends in a timely manner. We also may not be able to effectively promote our products by our
marketing and advertising campaigns and gain market acceptance. If our products fail to gain market acceptance, are restricted by regulatory
requirements or have quality problems, we may not be able to fully recover costs and expenses incurred in our operation, and our business,
financial condition, results of operations and prospects could be adversely affected.
We
rely upon independent third-party transportation providers for all of our product shipments and are subject to increased shipping costs
as well as the potential inability of our third-party transportation providers to deliver on a timely basis.
We
rely upon independent third-party transportation providers for all of our product shipments, including shipments from our related party
pharmacy to our customers. Our utilization of these third-party delivery services for shipments is subject to risks which may impact
a shipping company’s ability to provide delivery services that adequately meet our shipping needs, including risks related to employee
strikes, labor and capacity constraints, port security considerations, trade policy changes or restrictions, military conflicts, acts
of terrorism, accidents, natural disasters and inclement weather. Any interruption in service provided by our shipping companies could
cause temporary disruptions in our business, a loss of sales and profits, and other material adverse effects. In addition, we are subject
to increased shipping costs when fuel prices increase, as we use expedited means of transportation such as air freight. If we change
the shipping company we use, we could face logistical difficulties that could adversely affect deliveries, and we would incur costs and
expend resources in connection with such change.
The
failure of our physician services provider, Doctegrity, to attract and retain physicians in a competitive labor market could limit our
ability to execute our growth strategy, resulting in a slower rate of growth.
The
success of our wellness business will depend on the ability of Doctegrity and any future contracted telemedicine services provider(s)
to continue to recruit and retain a sufficient number of qualified licensed doctors. Although we believe such provider(s) will have an
effective recruitment process, there is no assurance that such provider(s) will be able to secure arrangements with sufficient numbers
of licensed doctors or retain the services of such practitioners. If Doctegrity or any provider(s) we engage in the future, experience
delays or shortages in obtaining access to qualified physicians, we would be unable to operate and may be forced to seek alternative
arrangements which could be more costly or may be forced to suspend our business operations.
If
we are unable to maintain or enter into future agreements with suppliers or our suppliers fail to supply us with our Mango ED and Mango
GROW products ingredients or any other potential future men’s wellness products, we may experience delays in selling our products.
We
may not be successful in maintaining or entering into new supply agreements on reasonable terms or at all or that we or our suppliers
will be able to obtain or maintain the necessary regulatory approvals or state and federal controlled substances registrations for current
or potential future suppliers in a timely manner or at all. If we are unable to obtain a sufficient quantity of active pharmaceutical
ingredients manufactured at a facility that is registered and listed with the FDA and required to produce products, there could be a
delay in producing products, which could adversely affect our product sales and operating results materially, which could significantly
harm our business. This has not occurred to date.
We
currently do not have any manufacturing facilities and intend to rely on third parties for the supply of our products (such as Epiq Scripts,
which is a related party), as well as for the supply of materials. However, we cannot be certain that we or our suppliers will be able
to obtain or maintain the necessary regulatory approvals or registrations for these suppliers in a timely manner or at all.
Our
business is exposed to risks associated with credit card and other online payment chargebacks and fraud.
A
majority of our revenue is, and is expected to be, processed through credit cards and other online payments. If we experience refunds
or chargebacks, our processors could require us to create reserves, increase fees or terminate contracts with us, which would have an
adverse effect on our financial condition. Our failure to limit fraudulent transactions conducted on our website, such as through the
use of stolen credit card numbers, could also subject us to liability and adversely impact our reputation. Under credit card association
rules, penalties may be imposed at the discretion of the association for inadequate fraud protection. Any such potential penalties would
be imposed on our credit card processor by the association. However, we face the risk that we may fail to maintain an adequate level
of fraud protection and that one or more credit card associations or other processors may, at any time, assess penalties against us or
terminate our ability to accept credit card payments or other form of online payments from customers, which would have a material adverse
effect on our business, financial condition and operating results.
We
could also incur significant fines or lose our ability to give customers the option of using credit cards to pay for our products if
we fail to follow payment card industry data security standards, even if there is no compromise of customer information. Although we
believe that we operate in compliance with payment card industry data security standards, it is possible that at times we may not be
in full compliance with these standards. Accordingly, we could be fined, which could impact our financial condition, or our ability to
accept credit and debit cards as payment could be suspended, which would cause us to be unable to process payments using credit cards.
If we are unable to accept credit card payments, our business, financial condition and operating results may be adversely affected.
In
addition, we could be liable if there is a breach of the payment information. Online commerce and communications depend on the secure
transmission of confidential information over public networks. We rely on encryption and authentication technology to authenticate and
secure the transmission of confidential information, including cardholder information. However, this technology may not prevent breaches
of the systems we use to protect cardholder information. In addition, some of our contracting parties may also collect or possess information
about our customers, and we may be subject to litigation or our reputation may be harmed if our contracting parties fail to protect our
customers’ information or if they use it in a manner inconsistent with our policies and practices. Data breaches can also occur
as a result of non-technical issues. Under contracts with processors, if there is unauthorized access to, or disclosure of, credit card
information we store, we could be liable to the credit card issuing banks for their cost of issuing new cards and related expenses.
Security
breaches, loss of data and other disruptions could compromise sensitive information related to our business or customers, or prevent
us from accessing critical information and expose us to liability, which could adversely affect our business and our reputation.
In
the ordinary course of our business, we collect, store, use and disclose sensitive data, including health information and other types
of personally identifiable information, or PII. We also process and store, and use additional third parties to process and store, confidential
and proprietary information such as intellectual property and other proprietary business information, including that of our customers,
providers and contracting parties.
Security
breaches of this infrastructure, including physical or electronic break-ins, computer viruses, attacks by hackers and similar breaches,
and employee or contractor error, negligence or malfeasance, can create system disruptions, shutdowns or unauthorized disclosure or modifications
of information, causing sensitive, confidential or proprietary information to be accessed or acquired without authorization or to become
publicly available. Because of the nature of the sensitive, confidential and proprietary information that we expect to collect, store,
transmit, and otherwise process, the security of our technology platform and other aspects of our services, including those provided
or facilitated by our third-party service providers, will be important to our operations and business strategy. Measures taken to protect
our systems, those of our third-party service providers, or sensitive, confidential and proprietary information that we or our third-party
service providers process or maintain, may not adequately protect us from the risks associated with the collection, storage and transmission
of such information. A security breach or privacy violation that leads to disclosure or unauthorized use or modification of, or that
prevents access to or otherwise impacts the confidentiality, security, or integrity of, sensitive, confidential, or proprietary information
we or our third-party service providers maintain or otherwise process, could harm our reputation, compel us to comply with breach notification
laws, and cause us to incur significant costs for remediation, fines, penalties, notification to individuals and governmental authorities,
implementation of measures intended to repair or replace systems or technology and to prevent future occurrences, potential increases
in insurance premiums, and forensic security audits or investigations. As a result, a security breach or privacy violation could result
in increased costs or loss of revenue.
Any
actual or suspected security breach or other compromise of our security measures or those of our third-party vendors, whether as a result
of hacking efforts, denial-of-service attacks, viruses, malicious software, break-ins, phishing attacks, social engineering or otherwise,
could harm our reputation and business, damage our brand and make it harder to retain existing customers or acquire new ones, require
us to expend significant capital and other resources to address the breach, and result in a violation of applicable laws, regulations
or other legal obligations. Our insurance policies may not cover, or may not be adequate to reimburse us for, losses caused by any such
security breach.
We
rely on email and other messaging services to connect with our existing and potential customers. Our customers may be targeted by parties
using fraudulent spoofing and phishing emails to misappropriate passwords, payment information or other personal information or to introduce
viruses through Trojan horse programs or otherwise through our customers’ computers, smartphones, tablets or other devices. Despite
our efforts to mitigate the effectiveness of such malicious email campaigns through product improvements, spoofing and phishing may damage
our brand and increase our costs. Any of these events or circumstances could materially adversely affect our business, financial condition
and operating results.
As
of the date of this filing, there have been no such data breaches or other security related issues.
We
may experience fluctuations in our tax obligations and effective tax rate, which could adversely affect our business, results of operations,
and financial condition.
We
are subject to taxes in every jurisdiction in which we operate. We record tax expense based on current tax liabilities and our estimates
of future tax liabilities, which may include reserves for estimates of probable settlements of tax audits. At any one-time, multiple
tax years are subject to audit by various taxing jurisdictions. The results of these audits and negotiations with taxing authorities
may affect the ultimate settlement of these issues. Further, our effective tax rate in a given financial statement period may be materially
impacted by changes in tax laws, changes in the mix and level of earnings by taxing jurisdictions, or changes to existing accounting
rules or regulations. Fluctuations in our tax obligations and effective tax rate could adversely affect our business, results of operations,
and financial condition.
If
we become subject to product liability claims, we may be required to pay damages that exceed our insurance coverage, if any.
Our
products are subject to risks for product liability claims due to inherent potential side effects. We may be unable to obtain or maintain
product liability coverage. A product liability claim in excess of, or excluded from, our insurance coverage which currently covers exposure
to product liability claims, both technology products and physical products, would have to be paid out of cash reserves and could have
a material adverse effect upon our business, financial condition and results of operations. Product liability insurance is expensive
even with large self-insured retentions or deductibles, difficult to maintain, and current or increased coverage may not continue to
be available on acceptable terms, if at all.
If
we cannot successfully defend ourselves against a product liability claim, we may incur substantial liabilities. Regardless of merit
or eventual outcome, liability claims may result in:
| |
● |
injury
to our reputation; |
| |
|
|
| |
● |
costs
of defending the claim and/or related litigation; |
| |
|
|
| |
● |
cost
of any potential adverse verdict; |
| |
|
|
| |
● |
substantial
monetary awards to patients or other claimants; and |
| |
|
|
| |
● |
the
inability to commercialize our products. |
Damages
awarded in a product liability action could be substantial and could have a negative impact on our financial condition. Whether or not
we were ultimately successful in product liability litigation, such litigation would consume substantial amounts of our financial and
managerial resources, and might result in adverse publicity, all of which would impair our business.
For
example, a 2014 study published in The Journal of the American Medical Association determined that Sildenafil (the active ingredient
in Viagra and one of the ingredients we alternatively use, together with Sildenafil in our Mango ED product) may be associated with a
higher risk of developing melanoma. The study evaluated data from more than 25,000 men who used Sildenafil and found that Sildenafil
use was significantly associated with an increased risk of subsequent melanoma, after considering other risk factors. It is possible
that the ingredients we use in our Mango ED and Mango GROW products or any other products we sell (including our Mango ED product, which
is made with Sildenafil as an alternative to Tadalafil), could be found to result in increases in the likelihood of developing cancer
or other diseases, which could subject us to litigation, penalties or recalls, all of which could have a material adverse effect on our
operations and cause the value of our securities to decline in value or become worthless. Furthermore, our use of Sildenafil in our products
could subject us to litigation, penalties or recalls, all of which could have a material adverse effect on our operations and cause the
value of our securities to decline in value or become worthless.
Disruptions
in our data and information systems could harm our reputation and our ability to run our business.
We
rely extensively on data and information systems for our supply chain, financial reporting, human resources and various other operations,
processes and transactions. Furthermore, a significant portion of the communications between us, our suppliers and customers depend on
information technology. Our data and information systems are subject to damage or interruption from power outages, computer and telecommunications
failures, computer viruses, security breaches (including breaches of our transaction processing or other systems that could result in
the compromise of confidential customer data), catastrophic events, data breaches and usage errors by our employees or third-party service
providers. Our data and information technology systems may also fail to perform as we anticipate, and we may encounter difficulties in
adapting these systems to changing technologies or expanding them to meet the future needs of our business. If our systems are breached,
damaged or cease to function properly, we may have to make significant investments to fix or replace them, suffer interruptions in our
operations, incur liability to our customers and others or face costly litigation, and our reputation with our customers may be harmed.
We also rely on third parties for a majority of our data and information systems, including for third-party hosting and payment processing.
If these facilities fail, or if they suffer a security breach or interruption or degradation of service, a significant amount of our
data could be lost or compromised and our ability to operate our business and deliver our product offerings could be materially impaired.
In addition, various third parties, such as our suppliers and payment processors, also rely heavily on information technology systems,
and any failure of these systems could also cause loss of sales, transactional or other data and significant interruptions to our business.
Any material interruption in the data and information technology systems we rely on, including the data or information technology systems
of third parties, could materially adversely affect our business, financial condition and operating results.
Risks
Related to Legal, Regulatory and Government
We
incur significant costs to ensure compliance with U.S. and Nasdaq reporting and corporate governance requirements.
We
incur significant costs associated with our public company reporting requirements and with applicable U.S. and Nasdaq corporate governance
requirements, including requirements under the Sarbanes-Oxley Act of 2002 and other rules implemented by the SEC and Nasdaq. We expect
all of these applicable rules and regulations to significantly increase our legal and financial compliance costs and to make some activities
more time-consuming and costly. We also expect that these applicable rules and regulations may make it more difficult and more expensive
for us to retain director and officer liability insurance and we may be required to accept reduced policy limits and coverage or incur
substantially higher costs to obtain the same or similar coverage. As a result, it may be more difficult for us to attract and retain
qualified individuals to serve on our Board of Directors or as executive officers.
If
we fail to comply with government laws and regulations it could have a materially adverse effect on our business.
The
health care industry is subject to extensive federal, state and local laws and regulations relating to licensure, conduct of operations,
ownership of facilities, addition of facilities and services, payment for services and prices for services that are extremely complex
and for which, in many instances, the industry does not have the benefit of significant regulatory or judicial interpretation. We exercise
care in structuring our arrangements with physicians and other referral sources to attempt to comply in all material respects with applicable
laws. We also take such laws into account when planning future marketing and other activities, and expect that our operations are in
compliance with applicable laws. The laws, rules and regulations described above are complex and subject to interpretation. In the event
of a determination that we are in violation of such laws, rules or regulations, or if further changes in the regulatory framework occur,
any such determination or changes could have a material adverse effect on our business. There can be no assurance however that we will
not be found in noncompliance in any particular situation.
Separately,
Federal law limits compounded drugs that are “essentially copies” of commercially available FDA approved drugs, including
those with the same route of administration. If our Mango ED and Mango GROW products, or any future products we may choose to market
in the future are deemed to be “essentially copies” of commercially available FDA approved drugs we would be prohibited from
compounding such drugs and would be unable to sell our Mango ED and Mango GROW drug or future products. If that were to occur, we would
need to change our business plan which would require substantial additional expenses and would have a material adverse effect on our
cash flows and the value of our securities.
Marketing
activities for our Mango ED and Mango GROW products are subject to strict governmental regulation which may limit our ability to market
or promote such product.
Our
business model depends on qualifying for certain statutory exemptions for drugs that are compounded by pharmacies in accordance with
applicable requirements. Pharmacy compounding is also subject to state oversight and regulation. Federal requirements include obtaining
individual prescriptions establishing that the compounded drug is necessary for each drug prescribed for each of our customers. Federal
law also limits compounded drugs that are “essentially copies” of commercially available FDA approved drugs, including those
with the same route of administration. These restrictions will limit our ability to market compounded drugs that have the same active
ingredients and route of administration as FDA-approved drugs, unless the compounded version offers a significant difference that the
prescriber determines is necessary for each individual patient.
The
FDA also has the authority to impose significant restrictions on approved products through regulations on advertising, promotional and
distribution activities. In particular, the FDA will object to any promotional activity (including through testimonials and surrogates)
that is “false or misleading in any particular,” including the failure to disclose material facts. For example, the FDA will
expect adequate substantiation for an efficacy claim, which would require substantial evidence derived from adequate and well-controlled
clinical trials. We believe we can conduct truthful and non-misleading promotional activities, including activities involving the use
of testimonials and surrogates, with limited claims that do not require substantial evidence derived from adequate and well-controlled
clinical trials and which do not include efficacy claims. If our products (including our Mango ED and Mango GROW products) are marketed
in contradiction with FDA laws and regulations, the FDA may issue warning letters that require specific remedial measures to be taken,
as well as an immediate cessation of the impermissible conduct, resulting in adverse publicity. The FDA may also require that all future
promotional materials receive prior agency review and approval before use. Certain states have also adopted regulations and reporting
requirements surrounding the promotion of pharmaceuticals. Failure by us or any of our collaborators to comply with state requirements
may affect our ability to promote or sell future products in certain states. This, in turn, could have a material adverse impact on our
financial results and financial condition and could subject us to significant liability, including civil and administrative remedies
as well as criminal sanctions.
These
restrictions may be more burdensome for compounded products as compared with FDA approved products because the latter have substantial
evidence of safety and effectiveness, which will limit our ability to compete against the sale of comparable FDA-approved products.
Evolving
government regulations and enforcement activities may require increased costs or adversely affect our results of operations.
Our
operations may be subject to direct and indirect adoption, expansion or reinterpretation of various laws and regulations. Compliance
with these evolving laws, regulations and interpretations may require us to change our practices at an undeterminable and possibly significant
initial monetary and annual expense. These additional monetary expenditures may increase future overhead, which could have a material
adverse effect on our results of operations. There could also be laws and regulations applicable to our business that we have not identified
or that, if changed, may be costly to us, and we cannot predict all the ways in which implementation of such laws and regulations may
affect us.
Additionally,
the introduction of new products may require us to comply with additional, yet undetermined, laws and regulations. Compliance may require
obtaining appropriate federal, state, or local licenses or certificates, increasing our security measures and expending additional resources
to monitor developments in applicable rules and ensure compliance. The failure to adequately comply with these future laws and regulations
may delay or possibly prevent our products from being offered to customers, which could have a material adverse effect on our business,
financial condition, and results of operations.
Failure
to comply with federal, state and foreign laws and regulations relating to privacy, data protection and consumer protection, or the expansion
of current or the enactment of new laws or regulations relating to privacy, data protection and consumer protection, could adversely
affect our business and our financial condition.
A
variety of federal, state and foreign laws and regulations govern the collection, use, retention, sharing and security of consumer data.
Laws and regulations relating to privacy, data protection and consumer protection are evolving and subject to potentially differing interpretations.
These requirements may be interpreted and applied in a manner that is inconsistent from one jurisdiction to another or may conflict with
other rules or our practices. As a result, our practices may not comply with all such laws, regulations, requirements and obligations.
Any failure, or perceived failure, by us to comply with any federal, state or foreign privacy or consumer protection-related laws, regulations,
industry self-regulatory principles, industry standards or codes of conduct, regulatory guidance, orders to which we may be subject or
other legal obligations relating to privacy or consumer protection could adversely affect our reputation, brand and business, and may
result in claims, investigations, proceedings or actions against us by governmental entities or others or other liabilities or require
us to change our operations.
We
collect, store, process, and use personal information and other customer data, and will rely on third parties that are not directly under
our control to manage certain of these operations and to collect, store, process and use payment information. Our customers’ personal
information may include names, addresses, phone numbers, email addresses, payment card data, and payment account information, as well
as other information. Due to the volume and sensitivity of the personal information and data we and these third parties manage, the security
features of our information systems are critical. If our security measures, some of which are managed by third parties, are breached
or fail, unauthorized persons may be able to access sensitive customer data, including payment card data. If we or our independent service
providers or business partners experience a breach of systems that collect, store or process our members’ and customers’
sensitive data, our brand could be harmed, sales of our products could decrease, and we could be exposed to claims, losses, administrative
fines, litigation or regulatory and governmental investigations and proceedings. Any such claim, investigation, proceeding or action
could hurt our reputation, brand and business, force us to incur significant expenses in defense of such proceedings, distract our management,
increase our costs of doing business, result in a loss of customers and suppliers and may result in the imposition of monetary penalties
and administrative fines. Depending on the nature of the information compromised, we may also have obligations to notify users, law enforcement,
or payment companies about the incident and may need to provide some form of remedy, such as refunds, for the individuals affected by
the incident.
Privacy
laws, rules, and regulations are constantly evolving in the United States and abroad and may be inconsistent from one jurisdiction to
another. We expect that new industry standards, laws and regulations will continue to be proposed regarding privacy, data protection
and information security in many jurisdictions, including privacy acts previously adopted by the states of California, Colorado, Connecticut,
Delaware, Indiana, Iowa, Montana, New Jersey, Oregon, Tennessee, Texas, Utah, and Virginia, certain of which are already effective, and
certain of which become effective during 2023, and from 2024 to 2026. We cannot yet determine the impact such future laws, regulations
and standards may have on our business. Complying with these evolving obligations is costly. For instance, expanding definitions and
interpretations of what constitutes “personal data” (or the equivalent) within the United States and elsewhere may
increase our compliance costs. Any failure to comply could give rise to unwanted media attention and other negative publicity, damage
our customer and consumer relationships and reputation, and result in lost sales, claims, administrative fines, lawsuits or regulatory
and governmental investigations and proceedings and may harm our business and results of operations.
Our
Mango ED and Mango GROW products have not been, and will not be, approved by the FDA. The use of such products may cause serious side
effects which could subject us to material litigation, damages and penalties.
Our
Mango ED and Mango GROW products have not been, and will not be, approved by the FDA. It is compounded using bulk drug substances and
as such, we believe it is exempt from specific FDA approval, provided that it is compounded in accordance with statutory requirements.
Because compounded drugs are not FDA-approved, the FDA does not verify their safety, effectiveness, or quality before they are marketed.
In addition, poor compounding practices can result in serious drug quality problems, such as contamination or a drug that contains too
much or too little active ingredient, among other possible quality deficiencies.
We
are not aware of any clinical studies involving the administration of Sildenafil or Tadalafil sublingually at the doses we intend to
provide patients, or the compounding of Sildenafil or Tadalafil, Oxytocin, and L-arginine to treat ED, as is contemplated by our Mango
ED products. We are also not aware of any clinical studies involving the administration of Minoxidil and Finasteride sublingually at
the dose we provide patients, or the compounding of Minoxidil, Finasteride, Vitamin D3 and Biotin, to attempt to treat hair loss, as
is contemplated by our Mango GROW product.
Because
our Mango ED and Mango GROW products have not been, and will not be, approved by the FDA, our products have not had the benefit of the
FDA’s clinical trial protocol which seeks to prevent the possibility of serious patient injury and death. If this were to occur,
we could be subject to litigation and governmental action, which could result in costly litigation, significant fines, judgments or penalties.
For example, in October 2012, a pharmacy in Massachusetts shipped compounded drugs that were contaminated with a fungus throughout the
country, and these drugs were injected into patients’ spines and joints. More than 750 people in 20 states developed fungal infections,
and more than 60 people died. This type of action could have a significant negative impact on our brand name, results of operations and
cash flows, and result in us having to cease selling products, curtailing our business plan, or seeking bankruptcy protection.
The
main ingredients of our Mango ED and Mango GROW products are publicly disclosed and separately our Mango ED products are being specially
compounded for the customer by a pharmacist with a physician’s prescription, and as a result, our Mango ED and Mango GROW products
formula can be replicated by other companies.
Our
Mango ED products are made up of the following three ingredients: (1) Either Sildenafil (50 milligrams (mg) or Tadalafil (10 (mg)), Oxytocin
(100 International units (IU)) and L-Arginine (50mg); and (2) either Sildenafil (100mg) or Tadalafil (20mg), Oxytocin (100IU) and L-Arginine
(50mg), an amino acid that is available as a dietary supplement. However, the fact that Sildenafil, Tadalafil and Oxytocin are used in
FDA approved drugs, and L-arginine is available as a dietary supplement, does not mean that these ingredients will prove safe when combined
into a single formulation to treat ED. Further, our Mango GROW product currently includes the following amounts of the four ingredients
discussed below: (1) Minoxidil (2.5mg), (2) Finasteride (1mg), (3) Vitamin D3 (2000IU), and (4) Biotin (1mg). However, the fact that
Minoxidil and Finasteride are used in FDA approved drugs, and Vitamin D3 and Biotin are available as a dietary supplement, does not mean
that these ingredients will prove safe when combined into a single formulation to treat hair growth.
We
currently offer two dosage levels of our Mango ED products and one dosage level of our Mango GROW product and anticipate a prescribing
doctor prescribing a dosage based on the needs and medical history of the patient. Additionally, because our Mango ED and Mango GROW
products are being specially compounded for the customer by a pharmacist with a physician’s prescription and because the ingredients
for our Mango ED and Mango GROW products are publicly disclosed, these product formulas can be replicated by other companies. As a result,
competitors, including those with greater resources, marketing, and brand recognition, may compete against us in the future using our
exact product ingredients or variations thereof. We may be unable to distinguish our Mango ED and Mango GROW products from copycat products
and may not be able to differentiate our product from competitors in the marketplace. As a result, we may fail to obtain a significant
market share, or may lose any market share we may obtain in the future, may be unable to compete with competitors, and may be forced
to abandon or curtail our business plan, which could cause the value of our shares to decline in value or become worthless.
Our
Mango ED and Mango GROW products need to be compounded by licensed pharmacists who are subject to risks regarding applicable exemptions
from the Federal Food, Drug, and Cosmetic Act.
Section
503A of the FFDCA describes the conditions under which compounded human drug products are exempt from the FFDCA sections on FDA approval
prior to marketing, current good manufacturing practice (“cGMP”) requirements, and labeling with adequate directions for
use. One of these conditions is that the drugs must be compounded based on the receipt of valid patient-specific prescriptions. Our ED
product needs to be compounded by licensed pharmacists, after being prescribed by a licensed physician. Licensed pharmacists who compound
drug products in accordance with Section 503A of the FFDCA are not required to comply with CGMP requirements and the drugs that they
compound are not required to be approved by the FDA, provided that the compounding complies with applicable requirements. Therefore,
the FDA is often not aware of potential problems with compounded drug products or compounding practices unless it receives a complaint,
such as a report of a serious adverse event or visible contamination. As such, the compounding of our products is subject to limited
FDA oversight, which could lead to such products not being compounded safely and could lead to product recalls and litigation which could
have a significant negative impact on our brand name, results of operations and cash flows, and result in us having to cease selling
products, curtailing our business plan, or seeking bankruptcy protection. Neither we, nor our representatives have had any conversations
with the FDA staff regarding whether our Mango ED or Mango GROW products can be sold pursuant to Section 503A of the FFDCA Act and future
conversations with the FDA may result in the FDA staff raising issues with such sales pursuant to Section 503A of the FFDCA, requiring
certain pre-requisites or changes to our current business plan, which may be costly or time consuming, and/or may result in us being
prohibited from selling our Mango ED and Mango GROW products pursuant to Section 503A of the FFDCA Act. We also face risks that the compounding
of our products does not fall within the exemption from the FFDCA provided by Section 503A thereof. For example, if the FDA determined
that any of our products are essentially a copy of an FDA approved product, we would be severely limited in our ability to compound such
a product. If any of the above were to apply, we may need to change our business plan or compounding activities, which could force us
to curtail our business plan or expend significant additional resources to obtain FFDCA or FDA approval for our products.
Notwithstanding
the above, under relevant FDA guidance, the FDA generally does not consider a compounded drug to be “essentially a copy”
of a commercially available drug if the compounded drug has a different route of administration as compared with the approved alternative,
and our Mango ED and Mango GROW products are for a different route of administration (e.g., sublingual). In addition, we do not expect
that we will be deemed to have engaged in such “copying”, because our Mango ED and Mango GROW products are based on a prescriber’s
determination for each patient that the change associated with the compounded product (our Mango ED and Mango GROW products) produces
for the patient a significant difference as compared with the commercially available drug product. Under relevant FDA guidance, the FDA
does not consider a compounded drug “essentially a copy” if a prescriber determines that there is a change, made for an identified
individual patient, which produces for that patient a significant difference from the commercially available product.
Health
care services, including arrangements with health care professionals, are heavily regulated at the state level, and the laws and regulations
may be changed or subject to new interpretations.
Each
state separately licenses health care professionals and determines when and under what conditions they may interact with and provide
services to patients. Telehealth consultations initiated through our platform must be offered in accordance with the laws and regulations
of the state where a patient is located, which may include laws that restrict the corporate practice of medicine and fee splitting. Each
state’s laws are subject to legislative and regulatory changes, as well as judicial interpretations, and future changes or interpretations
of state laws restricting the corporate practice of medicine and fee splitting could adversely affect the permissibility of (a) our relationship
with Doctegrity; and/or (b) Doctegrity’s relationship with its contracted physicians. If our relationship with Doctegrity and/or
Doctegrity’s relationship with its contracted physicians needed to be restructured in light of any such adverse changes or interpretations,
that restructuring could negatively affect our ability to connect consumers with medical providers in certain states, and thus those
customers’ ability to ultimately receive our products.
We
do not have a pharmacy and depend on a related party to compound our Mango product and other potential future men’s wellness products.
We
rely on a related party pharmacy for the manufacture of our Mango product and will rely on this pharmacy or others for any potential
future men’s wellness products we market and we cannot assure you that they will be successful. This subjects us to a number of
risks, including the following:
| |
● |
we
may not be able to control the commercialization of our products, including the amount, timing and quality of resources that our
contracting parties may devote to our products; |
| |
|
|
| |
● |
our
contracting parties may experience financial, regulatory or operational difficulties, which may impair their ability to fulfill their
contractual obligations; |
| |
|
|
| |
● |
business
combinations or significant changes in a contracting parties’ business strategy may adversely affect a contracting party’s
willingness or ability to perform their obligations under any arrangement; |
| |
|
|
| |
● |
legal
disputes or disagreements may occur with one or more of our contracting parties or between our contracting parties and our suppliers
or former contracting parties; and |
| |
|
|
| |
● |
a
contracting party could independently move forward with a competing product developed either independently or in collaboration with
others, including with one of our competitors. |
If
any of our contracting parties fail to fulfill their future contractual obligations, our business may be negatively affected and we may
receive limited or no revenues under our agreements with them. See also the risk factor, “The related party pharmacy we have
entered into an agreement with may not receive licenses in all of the 50 United States to provide national coverage for us to sell our
Mango ED and Mango GROW products and future products” below.
Our
use and disclosure of personally identifiable information, including health information, is subject to federal and state privacy and
security regulations, and our failure to comply with those regulations or to adequately secure the information we hold could result in
significant liability or reputational harm and, in turn, a material adverse effect on our client base and revenue.
Numerous
state and federal laws and regulations govern the collection, dissemination, use, privacy, confidentiality, security, availability and
integrity of personally identifiable information, or PII, including protected health information, or PHI. These laws and regulations
include the Health Information Portability and Accountability Act of 1996 (“HIPAA”), as amended by the Health Information
Technology for Economic and Clinical Health Act, or HITECH, and their implementing regulations (referred to collectively as “HIPAA”).
HIPAA establishes a set of basic national privacy and security standards for the protection of PHI. HIPAA requires us to develop and
maintain policies and procedures with respect to PHI that is used or disclosed, including the adoption of administrative, physical and
technical safeguards to protect such information. HIPAA imposes mandatory penalties for certain violations. Penalties for violations
of HIPAA and its implementing regulations start at $100 per violation and are not to exceed $50,000 per violation, subject to a cap of
$1.5 million for violations of the same standard in a single calendar year. However, a single breach incident can result in violations
of multiple standards. HIPAA also authorizes state attorneys general to file suit on behalf of their residents. Courts are able to award
damages, costs and attorneys’ fees related to violations of HIPAA in such cases. While HIPAA does not create a private right of
action allowing individuals to sue us in civil court for violations of HIPAA, its standards have been used as the basis for duty of care
in state civil suits such as those for negligence or recklessness in the misuse or breach of PHI. In addition, HIPAA mandates that the
Secretary of Health and Human Services, or HHS, conduct periodic compliance audits of HIPAA covered entities or business associates for
compliance with the HIPAA Privacy and Security Standards. It also tasks HHS with establishing a methodology whereby harmed individuals
who were the victims of breaches of unsecured PHI may receive a percentage of the Civil Monetary Penalty fine paid by the violator. HIPAA
further requires that patients be notified of any unauthorized acquisition, access, use or disclosure of their unsecured PHI that compromises
the privacy or security of such information, with certain exceptions related to unintentional or inadvertent use or disclosure by employees
or authorized individuals. HIPAA specifies that such notifications must be made “without unreasonable delay and in no case later
than 60 calendar days after discovery of the breach.” If a breach affects 500 patients or more, it must be reported to HHS without
unreasonable delay, and HHS will post the name of the breaching entity on its public web site. Breaches affecting 500 patients or more
in the same state or jurisdiction must also be reported to the local media. If a breach involves fewer than 500 people, the covered entity
must record it in a log and notify HHS at least annually.
Numerous
other federal and state laws protect the confidentiality, privacy, availability, integrity and security of PII, including PHI. These
laws in many cases are more restrictive than, and may not be pre-empted by, the HIPAA rules and may be subject to varying interpretations
by courts and government agencies, creating complex compliance issues for us and our clients and potentially exposing us to additional
expense, adverse publicity and liability.
Because
of the extreme sensitivity of the PII we store and transmit, the security features of our technology platform are very important. If
our security measures are breached or fail, unauthorized persons may be able to obtain access to sensitive client data, including HIPAA-regulated
PHI. As a result, our reputation could be severely damaged, adversely affecting client confidence. In addition, we could face litigation,
damages for contract breach, penalties and regulatory actions for violation of HIPAA and other applicable laws or regulations and significant
costs for remediation, notification to individuals and for measures to prevent future occurrences. Any potential security breach could
also result in increased costs associated with liability for stolen assets or information, repairing system damage that may have been
caused by such breaches, incentives offered to clients in an effort to maintain our business relationships after a breach and implementing
measures to prevent future occurrences, including organizational changes, deploying additional personnel and protection technologies,
training employees and engaging third-party experts and consultants.
Risks
Related to Related Party Relationships and Transactions and Our Management
We
depend heavily on our senior management, including our Chief Executive Officer, who may have a conflict of interest with regard to various
matters. The ability of certain key employees to devote adequate time to us is critical to the success of our business, and failure to
do so may adversely affect our revenues and as a result could materially adversely affect our business, financial condition and results
of operations.
We
must retain the services of our key employees and strategically recruit and hire new talented employees. Our future business and results
of operations depend in significant part upon the continued contributions of our senior management personnel, particularly our Chairman
and Chief Executive Officer, Jacob D. Cohen. Mr. Cohen is currently a co-Manager and 51% owner of Epiq Scripts, and as Chief Executive
Officer of Ronin Equity Partners, Inc., a private investment company, and in various positions with other entities and groups. Mr. Cohen
currently spends approximately 95% of his time on Company matters. As a result, Mr. Cohen dedicates only a portion of his professional
efforts to our business and operations, and there is no contractual obligation for him to spend a specific amount of his time with us.
Mr. Cohen may not be able to dedicate adequate time to our business and operations and we could experience an adverse effect on our operations
due to the demands placed on him from his other professional obligations. Such involvement in other businesses may therefore present
a conflict of interest regarding decisions he makes for us or with respect to the amount of time available for us. If we lose his services
or if he fails to perform in his current position, or if we are not able to attract and retain skilled personnel as needed, our business
could suffer. Significant turnover in our senior management could significantly deplete our institutional knowledge held by our existing
senior management team. We depend on the skills and abilities of these key personnel in managing our operations, product development,
marketing and sales aspects of our business, any part of which could be harmed by turnover in the future.
Moving
forward, should the services of Mr. Cohen be lost for any reason, we will incur costs associated with recruiting replacements and any
potential delays in operations which this may cause. If we are unable to replace such individual with a suitably trained alternative
individual(s), we may be forced to scale back or curtail our business plan.
Separately,
if our executive officers do not devote sufficient time towards our business, we may never be able to effectuate our business plan.
We
have engaged and in the future may engage in transactions with related parties and such transactions present possible conflicts of interest
that could have an adverse effect on us.
We
have entered, and may continue to enter, into transactions with related parties for financing, corporate, business development and operational
services. Included in such transactions is a Master Services Agreement and Statement of Work with Epiq Scripts, LLC, a related party,
51% owned and controlled by Jacob D. Cohen, our Chairman and Chief Executive Officer, as discussed in greater detail under “ Business-Material
Agreements-Master Services Agreement with Epiq Scripts” and “-First Amendment to MSA,” for pharmacy and compounding
services. Such transactions may not have been/may not be, entered into on an arm’s-length basis, and we may have achieved more
or less favorable terms because such transactions were entered into with our related parties. This could have a material effect on our
business, results of operations and financial condition. Such conflicts could cause an individual in our management to seek to advance
his or her economic interests or the economic interests of certain related parties above ours. Further, the appearance of conflicts of
interest created by related party transactions could impair the confidence of our investors.
We
are significantly reliant on related party relationships.
We
have entered into a Master Services Agreement and Statement of Work with Epiq Scripts, LLC, a related party, 51% owned and controlled
by Jacob D. Cohen, our Chairman and Chief Executive Officer, who also serves as a co-Manager of Epiq Scripts, as discussed in greater
detail under “Business-Material Agreements-Master Services Agreement with Epiq Scripts” and “-First Amendment to MSA,”
for pharmacy and compounding services. In the event that relationship is terminated, our costs may increase, and we may be unable to
effectively obtain the services currently provided by Epiq Scripts, LLC. Additionally, certain of our consultants are employed by Epiq
Scripts, LLC. We also anticipate entering into other related party relationships in the future. While we believe that all related party
agreements have been and will be on arms-length terms, such significant related party relationships may be perceived negatively by potential
shareholders or investors and/or may result in conflicts of interest. Each of our officers and directors (including those discussed above)
presently has, and any of them in the future may have, additional fiduciary or contractual obligations to other entities pursuant to
which such officer or director may be required to present a business opportunity to such entity, subject to his or her fiduciary duties
under applicable law. Additionally, such persons may have conflicts of interest in allocating their time among various business activities.
These conflicts may not be resolved in our favor. Our significant related party relationships and transactions, the terms of such relationships
and transactions, and/or the termination of any such relationships or transactions, may have a material adverse effect on our results
of operations moving forward and/or create conflicts of interest or perceived conflicts of interest which may have a material adverse
effect on the value of our securities.
The
related party pharmacy we have entered into an agreement with may not receive licenses in all of the 50 United States to provide national
coverage for us to sell our Mango ED and Mango GROW products and future products.
We
have entered into a Master Services Agreement and Statement of Work with Epiq Scripts, LLC, a related party, 51% owned and controlled
by Jacob D. Cohen, our Chairman and Chief Executive Officer, as discussed in greater detail under “Business-Material Agreements-Master
Services Agreement with Epiq Scripts,” for pharmacy and compounding services. Epiq Script’s ability to provide pharmacy services
in each state is subject to, among other things, receipt of regulatory approvals and licenses in the states in which it operates. Currently
Epiq Scripts holds State Board of Pharmacy (or its equivalent) licenses to operate in the District of Columbia and 47 states: Alaska,
Arizona, Arkansas, Colorado, Connecticut, Delaware, Florida, Georgia, Hawaii, Idaho, Illinois, Indiana, Iowa, Kansas, Kentucky, Louisiana,
Maine, Maryland, Massachusetts, Michigan, Minnesota, Mississippi, Missouri, Montana, Nebraska, Nevada, New Hampshire, New Jersey, New
Mexico, New York, North Carolina, North Dakota, Ohio, Oklahoma, Oregon, Pennsylvania, Rhode Island, South Dakota, Tennessee, Texas, Utah,
Vermont, Virginia, Washington, West Virginia, Wisconsin, and Wyoming. Its failure to receive regulatory approval or licenses in the other
states in which we hope to operate, or loss of such licenses in the future, may prohibit us from selling our Mango products to customers
that reside in those states limiting our ability to grow and compete with other companies that have those capabilities. Any of the above
may have an adverse effect on our revenues, operations and cash flow and cause the value of our securities to decline in value or become
worthless. We also face related party conflicts associated with our engagement of Epiq Scripts, LLC as discussed in greater detail above.
Jacob
D. Cohen, our Chairman and Chief Executive Officer, beneficially owns a significant percentage of our outstanding common stock and as
such exercise significant voting control over us, which limits shareholders’ abilities to influence corporate matters and could
delay or prevent a change in corporate control.
Jacob
D. Cohen, our Chairman and Chief Executive Officer, beneficially owns approximately 33.6 % of the outstanding shares of our common
stock. As a result, he has significant influence on the shareholder vote. Consequently, he has the ability to influence matters affecting
our shareholders and therefore exercise significant control in determining the outcome of a number of corporate transactions or other
matters, including (i) making amendments to our certificate of formation; (ii) whether to issue additional shares of common stock and
preferred stock, including to himself; (iii) employment decisions, including compensation arrangements; (iv) whether to enter into material
transactions with related parties; (v) election of directors; and (vi) any merger or significant corporate transactions, including with
himself or other related parties. Additionally, it will be difficult if not impossible for investors to remove our current directors
(including, but not limited to Mr. Cohen), which will mean he will remain in control of who serves as officers of the Company as well
as whether any changes are made in the Board of Directors. As a potential investor in the Company, you should keep in mind that even
if you own shares of our common stock and wish to vote them at annual or special shareholder meetings, your shares will have little effect
on the outcome of corporate decisions. Because Mr. Cohen will significantly influence the vote on all shareholder matters, investors
may find it difficult to replace our management if they disagree with the way our business is being operated. The interests of Mr. Cohen
may not coincide with our interests or the interests of other shareholders.
Mr.
Cohen acquired his shares of common stock for substantially less than the price of the shares of common stock acquired in our IPO and
our Follow On Offering, and/or the current trading price of our common stock, and may have interests, with respect to their common stock,
that are different from other investors and the concentration of voting power held by Mr. Cohen may have an adverse effect on the price
of our common stock.
In
addition, this concentration of ownership might adversely affect the market price of our common stock by: (1) delaying, deferring or
preventing a change of control of our Company; (2) impeding a merger, consolidation, takeover or other business combination involving
our Company; or (3) discouraging a potential acquirer from making a tender offer or otherwise attempting to obtain control of our Company.
Potential
competition from our existing executive officers, after they leave their employment with us, and subject to the non-compete terms of
their employment agreements, could negatively impact our profitability.
Although
our Chief Executive Officer, Jacob D. Cohen, and our Chief Operating Officer, Amanda Hammer, are prohibited from competing with us while
they are employed with us and for 12 months thereafter (subject to the terms of, and exceptions set forth in, their employment agreements
with the Company), none of such individuals will be prohibited from competing with us after such 12-month period ends. Additionally,
the Federal Trade Commission recently proposed a new rule that, if it becomes effective, would ban employers from imposing non-competes
on their workers, which if effective could prohibit the Company from enforcing, or invalidate, the non-competes in our executive’s
and in certain other employee’s, employment agreements. Finally, various states have recently enacted rules banning non-competes,
including California. Accordingly, any of these individuals could be in a position to use industry experience gained while working with
us to compete with us. Such competition could distract or confuse customers, reduce the value of our intellectual property and trade
secrets, or reduce our future revenues, earnings or growth prospects.
Risks
Related to Intellectual Property
We
operate in an industry with the risk of intellectual property litigation. Claims of infringement against us may hurt our business.
We
must protect the proprietary nature of the intellectual property used in our business. There can be no assurance that trade secrets and
other intellectual property will not be challenged, invalidated, misappropriated or circumvented by third parties.
Additionally,
our success depends, in part, upon non-infringement of intellectual property rights owned by others and being able to resolve claims
of intellectual property infringement without major financial expenditures or adverse consequences. Participants that own, or claim to
own, intellectual property may aggressively assert their rights. From time to time, we may be subject to legal proceedings and claims
relating to the intellectual property rights of others. Future litigation may be necessary to defend us by determining the scope, enforceability,
and validity of third-party proprietary rights or to establish its proprietary rights. Our competitors have substantially greater resources
and are able to sustain the costs of complex intellectual property litigation to a greater degree and for longer periods of time. In
addition, patent holding companies that focus solely on extracting royalties and settlements by enforcing patent rights may target us.
Regardless of whether claims that we are infringing patents or other intellectual property rights have any merit, these claims are time-consuming
and costly to evaluate and defend and could:
| |
● |
cause
delays or stoppages in providing products; |
| |
|
|
| |
● |
divert
management’s attention and resources; |
| |
|
|
| |
● |
require
technology changes to our products that would cause our Company to incur substantial cost; |
| |
|
|
| |
● |
subject
us to significant liabilities; and |
| |
|
|
| |
● |
require
us to cease some or all of our activities. |
In
addition to liability for monetary damages, which may be tripled and may include attorneys’ fees, or, in some circumstances, damages
against clients, we may be prohibited from developing, commercializing, or continuing to provide some or all of our products unless we
obtain licenses from, and pay royalties to, the holders of the patents or other intellectual property rights, which may not be available
on commercially favorable terms, or at all.
Risks
Related to the Telehealth Operations of Our Contracting Parties
The
telehealth business of our telehealth provider could be adversely affected by ongoing legal challenges or by new state actions restricting
the ability to provide telehealth services in certain states.
We
use telehealth providers to provide telehealth consultations and related services on our Mangoceuticals platform, which connects users/customers
with third-party health care providers and Epiq Scripts, LLC, a related party pharmacy. We have entered into an agreement with Doctegrity,
pursuant to which Doctegrity provides clinical services directly to our customers via telehealth. Through these arrangements, the professionals
or professional entities are responsible for the practice of medicine and control of the clinical decision-making.
Our
ability to conduct business operations in each state is dependent upon the state’s treatment of medicine under such state’s
laws, and rules and policies governing the practice of physician supervised services, which are subject to changing political, regulatory
and other influences.
We
depend on our contracted parties to maintain appropriate telehealth licenses to be able to provide telehealth services to our potential
customers and prescribe them our products, which are required to be prescribed by licensed physicians. In the event we are not able to
maintain relationships with telehealth providers, state licensing laws make it harder, more costly or impossible to provide telehealth
services, or our customers are otherwise unable to obtain prescriptions for our products, we may be unable to sell products, which could
result in us having to curtail our business plan or cease operating.
Our
contracting parties’ telehealth business could be adversely affected by ongoing legal challenges to their business model or by
new state actions restricting their ability to provide the full range of services in certain states.
The
ability of our contracted parties’ telehealth operations in each state is dependent upon the state’s treatment of medicine
under such state’s laws, rules and policies governing the practice of physician supervised services, which are subject to changing
political, regulatory and other influences. In the event our contracted parties are unable to provide telehealth services for any reason,
it would have a material adverse effect on our ability to sell products and in turn our revenues and operating results.
Risks
Related to Our Governing Documents and Texas Law
Our
Certificate of Formation, Bylaws and Texas law provide for indemnification of officers and directors at our expense and limit the liability
of our directors, which may result in a major cost to us and hurt the interests of our shareholders because corporate resources may be
expended for the benefit of officers or directors.
Our
Certificate of Formation, Bylaws and Texas law provide for us to indemnify and hold harmless, to the fullest extent permitted by applicable
law, each person who is or was made a party or is threatened to be made a party to or is otherwise involved in any threatened, pending
or completed action, suit or proceeding by reason of the fact that he or she is or was a director or officer of the Company or, while
a director or officer of the Company, is or was serving at the request of the Company as a director, officer, employee or agent of another
corporation or of a partnership, joint venture, trust, other enterprise or nonprofit entity, including service with respect to an employee
benefit plan. Our Certificate of Formation also provides that the personal liability of our directors is eliminated to the fullest extent
permitted by the Texas Business Organizations Code, as such may be amended or supplemented. These indemnification obligations and limitations
of liability may result in a major cost to us and hurt the interests of our shareholders because corporate resources may be expended
for the benefit of officers or directors.
We
have been advised that, in the opinion of the SEC, indemnification for liabilities arising under federal securities laws is against public
policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification for liabilities
arising under federal securities laws, other than the payment by us of expenses incurred or paid by a director, officer or controlling
person in the successful defense of any action, suit or proceeding, is asserted by a director, officer or controlling person in connection
with our activities, we will (unless in the opinion of our counsel, the matter has been settled by controlling precedent) submit to a
court of appropriate jurisdiction, the question whether indemnification by us is against public policy as expressed in the Securities
Act and will be governed by the final adjudication of such issue. The legal process relating to this matter if it were to occur is likely
to be very costly and may result in us receiving negative publicity, either of which factors is likely to materially reduce the market
and price for our shares.
We
have established preferred stock which can be designated by our Board of Directors without shareholder approval.
We
have 10,000,000 shares of preferred stock authorized. The shares of our preferred stock may be issued from time to time in one or more
series, each of which shall have a distinctive designation or title as shall be determined by our Board of Directors prior to the issuance
of any shares thereof. The preferred stock shall have such voting powers, full or limited, or no voting powers, and such preferences
and relative, participating, optional or other special rights and such qualifications, limitations or restrictions thereof as adopted
by the Board of Directors. Because the Board of Directors is able to designate the powers and preferences of the preferred stock without
the vote of a majority of our shareholders, our shareholders will have no control over what designations and preferences our preferred
stock will have. The issuance of shares of preferred stock or the rights associated therewith, could cause substantial dilution to our
existing shareholders. Additionally, the dilutive effect of any preferred stock which we may issue may be exacerbated given the fact
that such preferred stock may have voting rights and/or other rights or preferences which could provide the preferred shareholders with
substantial voting control over us and/or give those holders the power to prevent or cause a change in control, even if that change in
control might benefit our shareholders. As a result, the issuance of shares of preferred stock may cause the value of our securities
to decrease.
Anti-takeover
provisions in our Certificate of Formation and our Bylaws, as well as provisions of Texas law, might discourage, delay or prevent a change
in control of our company or changes in our management and, therefore, depress the trading price of our common stock.
Our
Certificate of Formation, Bylaws and Texas law contain provisions that may discourage, delay or prevent a merger, acquisition or other
change in control that shareholders may consider favorable, including transactions in which you might otherwise receive a premium for
your shares of our common stock. These provisions may also prevent or delay attempts by our shareholders to replace or remove our management.
Our corporate governance documents include provisions:
| |
● |
requiring
advance notice of shareholder proposals for business to be conducted at meetings of our shareholders and for nominations of candidates
for election to our Board of Directors; |
| |
|
|
| |
● |
authorizing
blank check preferred stock, which could be issued with voting, liquidation, dividend and other rights superior to our common stock;
and |
| |
|
|
| |
● |
providing
indemnification to, our directors and officers. |
The
existence of the foregoing provisions and anti-takeover measures could limit the price that investors might be willing to pay in the
future for shares of our common stock. They could also deter potential acquirers of our company, thereby reducing the likelihood that
you could receive a premium for your common stock in an acquisition.
Risks
Related to Our Common Stock
If
we fail to comply with the continued minimum closing bid requirements of The Nasdaq Capital Market by October 24, 2024, or other requirements
for continued listing, including stockholder equity requirements, our common stock may be delisted and the price of our common stock
and our ability to access the capital markets could be negatively impacted.
Our
common stock is listed for trading on Nasdaq. In order to remain listed on Nasdaq, we must satisfy Nasdaq’s continued listing requirements,
including, among other things, the $1.00 minimum bid price requirement set forth in Nasdaq Listing Rule 5550(a)(2) (the “Minimum
Bid Price Requirement”).
On
October 30, 2023, the Staff of the Listing Qualifications Department of The Nasdaq Stock Market LLC (the “Staff”)
notified us that we did not comply with the Minimum Bid Price Requirement, and that we had 180 calendar days, or until April 29, 2024,
to regain compliance. On April 25, 2024, under the Nasdaq Listing Rules, we submitted a request to Nasdaq for an additional 180-day extension
to regain compliance with the Minimum Bid Price Requirement and notice of our intention to cure the deficiency, including by effecting
a reverse stock split if necessary. On April 30, 2024, Nasdaq advised us that we were provided an additional 180 calendar day compliance
period, or until October 24, 2024, to regain compliance with the Minimum Bid Price Requirement. The closing bid price of our securities
must be at least $1.00 per share for a minimum of ten consecutive business days to regain compliance. We intend to monitor the closing
bid price of our common stock and plan, if necessary, to implement a reverse stock split, to regain compliance with the Minimum Bid Price
Requirement. The stockholders of the Company, at the March 25, 2024, special meeting of stockholders previously approved an amendment
to our Certificate of Formation, as amended, to effect a reverse stock split of our issued and outstanding shares of our common stock,
by a ratio of between one-for-two to one-for-fifty, inclusive, with the exact ratio to be set at a whole number to be determined by our
Board of Directors or a duly authorized committee thereof in its discretion, at any time after approval of the amendment and prior to
March 25, 2025.
If
we seek to implement a reverse stock split in order to remain listed on Nasdaq, the announcement or implementation of such a reverse
stock split could negatively affect the price of our common stock. However, there can be no assurance that we will regain compliance
with the Minimum Bid Price Requirement prior to October 24, 2024.
If
we are unable to regain compliance with the Minimum Bid Price Requirement by October 24, 2024, or if we fail to meet any of the other
continued listing requirements, including stockholder equity requirements (which we are required to meet as of the filing of our next
periodic report with the Commission), our securities may be delisted from Nasdaq, which could reduce the liquidity of our common stock
materially and result in a corresponding material reduction in the price of our common stock. In addition, delisting could harm our ability
to raise capital through alternative financing sources on terms acceptable to us, or at all, and may result in the potential loss of
confidence by investors, employees and business development opportunities. Such a delisting likely would impair your ability to sell
or purchase our common stock when you wish to do so. Further, if we were to be delisted from Nasdaq, our common stock may no longer be
recognized as a “covered security” and we would be subject to regulation in each state in which we offer our securities.
Thus, delisting from Nasdaq could adversely affect our ability to raise additional financing through the public or private sale of equity
securities, would significantly impact the ability of investors to trade our securities and would negatively impact the value and liquidity
of our common stock.
A
reverse stock split may not increase our stock price and have the desired effect of maintaining compliance with the rules of the Nasdaq.
The
Company received stockholder approval at a special meeting of stockholders held on March 25, 2024, of an amendment to the Company’s
Second Amended and Restated Certificate of Incorporation, to effect a reverse stock split of the Company’s issued and outstanding
shares of common stock, by a ratio of between one-for-two to one-for-fifty, inclusive, with the exact ratio to be set at a whole number
to be determined by the Company’s Board of Directors or a duly authorized committee thereof in its discretion, at any time prior
to March 25, 2025.
The
Board expects that a reverse stock split of our common stock will increase the market price of our common stock so that we are able to
regain and maintain compliance with the Nasdaq minimum bid price listing standard. However, the effect of the reverse stock split upon
the market price of our common stock cannot be predicted with any certainty, and the history of similar reverse stock splits for companies
in like circumstances is varied. The price per share of our common stock after the reverse stock split may not reflect the exchange ratio
implemented by the Board of Directors and the price per share following the effective time of the reverse stock split may not be maintained
for any period of time following the reverse stock split. Accordingly, the total market capitalization of our common stock following
a reverse stock split may be lower than before the reverse stock split.
Under
applicable Nasdaq rules, to regain compliance with the $1.00 minimum closing bid price requirement and maintain our listing on the Nasdaq
Capital Market, the $1.00 closing bid price must be maintained for a minimum of ten (10) consecutive business days. Accordingly, we cannot
assure you that we will be able to maintain our Nasdaq listing after a reverse stock split is effected or that the market price per share
after a reverse stock split will exceed or remain in excess of the $1.00 minimum bid price for a sustained period of time.
It
is possible that the per share price of our common stock after a reverse stock split will not rise in proportion to the reduction in
the number of shares of our common stock outstanding resulting from the reverse stock split, and the market price per post-reverse stock
split share may not exceed or remain in excess of the $1.00 minimum bid price for a sustained period of time, and the reverse stock split
may not result in a per share price that would attract brokers and investors who do not trade in lower priced stocks. Even if we effect
the reverse stock split, the market price of our common stock may decrease due to factors unrelated to the stock split. In any case,
the market price of our common stock may also be based on other factors which may be unrelated to the number of shares outstanding, including
our future performance. If the reverse stock split is consummated and the trading price of the common stock declines, the percentage
decline as an absolute number and as a percentage of our overall market capitalization may be greater than would occur in the absence
of the reverse stock split. Even if the market price per post-reverse stock split share of our common stock remains in excess of $1.00
per share, we may be delisted due to a failure to meet other continued listing requirements, including Nasdaq requirements related to
the minimum stockholders’ equity, the minimum number of shares that must be in the public float, the minimum market value of the
public float and the minimum number of round lot holders.
A
reverse stock split may decrease the liquidity of our common stock.
The
liquidity of our common stock may be harmed by a reverse stock split given the reduced number of shares of common stock that would be
outstanding after a reverse stock split, particularly if the stock price does not increase as a result of the reverse stock split. In
addition, investors might consider the increased proportion of unissued authorized shares of common stock to issued shares to have an
anti-takeover effect under certain circumstances, because the proportion allows for dilutive issuances which could prevent certain stockholders
from changing the composition of the Board of Directors or render tender offers for a combination with another entity more difficult
to successfully complete. The Board of Directors does not intend for a reverse stock split to have any anti-takeover effects.
If
we implement a reverse stock split, the liquidity of our common stock may be adversely effected.
As
discussed above, the stockholders of the Company, at the March 25, 2024 special meeting of stockholders, our stockholders approved an
amendment to our Certificate of Formation, as amended, to effect a reverse stock split of our issued and outstanding shares of our common
stock, by a ratio of between one-for-two to one-for-fifty, inclusive, with the exact ratio to be set at a whole number to be determined
by our Board of Directors or a duly authorized committee thereof in its discretion, at any time after approval of the amendment and prior
to March 25, 2025. The Company plans to complete a reverse stock split within the stockholder approved ratio, if required to comply with
the Nasdaq Minimum Bid Price Requirement, and if deemed to be in the interests of the Company. However, there can be no assurance that
the market price per new share of our common stock after the reverse stock split will remain unchanged or increase in proportion to the
reduction in the number of old shares of our common stock outstanding before the reverse stock split. The liquidity of the shares of
our common stock may be affected adversely by any reverse stock split given the reduced number of shares of our common stock that will
be outstanding following the reverse stock split, especially if the market price of our common stock does not increase as a result of
the reverse stock split. In addition, the reverse stock split may increase the number of stockholders who own odd lots (less than 100
shares) of our common stock, creating the potential for such stockholders to experience an increase in the cost of selling their shares
and greater difficulty effecting such sales.
Following
any reverse stock split, the resulting market price of our common stock may not attract new investors and may not satisfy the investing
requirements of those investors. Although we believe that a higher market price of our common stock may help generate greater or broader
investor interest, there can be no assurance that the reverse stock split will result in a share price that will attract new investors,
including institutional investors. In addition, there can be no assurance that the market price of our common stock will satisfy the
investing requirements of those investors. As a result, the trading liquidity of our common stock may not necessarily improve.
Stockholders
may be diluted significantly through our efforts to obtain financing and satisfy obligations through the issuance of additional shares
of our common stock.
Wherever
possible, our Board of Directors will attempt to use non-cash consideration to satisfy obligations. In many instances, we believe that
the non-cash consideration will consist of restricted shares of our common stock or where shares are to be issued to our officers, directors
and applicable consultants. Our Board of Directors has authority, without action or vote of the stockholders, but subject to Nasdaq rules
and regulations (which generally require stockholder approval for any transactions which would result in the issuance of more than 20%
of our then outstanding shares of common stock or voting rights representing over 20% of our then outstanding shares of stock), to issue
all or part of the authorized but unissued shares of common stock. In addition, we may attempt to raise capital by selling shares of
our common stock, possibly at a discount to market. These actions will result in dilution of the ownership interests of existing stockholders,
which may further dilute common stock book value, and that dilution may be material. Such issuances may also serve to enhance existing
management’s ability to maintain control of the Company because the shares may be issued to parties or entities committed to supporting
existing management.
Certain
recent initial public offerings of companies with public floats comparable to the anticipated public float of the Company have experienced
extreme volatility that was seemingly unrelated to the underlying performance of the respective company. We have in the past, and may
in the future experience similar volatility, which may make it difficult for prospective investors to assess the value of our common
stock.
In
addition to the risks addressed below under the heading “- Our common stock prices have been, and may continue to be, volatile
and could decline substantially following the date of this Prospectus,” our common stock may be subject to extreme volatility
that is seemingly unrelated to the underlying performance of our business. For example, since our common stock began trading on the Nasdaq
Capital Market in connection with our IPO on March 20, 2023, the trading price of our common stock has traded as high as $4.37 and as
low as $0.143 per share. The trading price of our common stock is expected to continue to be volatile, and our common stock may be subject
to rapid and substantial price volatility. Such volatility, including any stock-run up, may be unrelated to our actual or expected operating
performance, financial condition or prospects, making it difficult for prospective investors to assess the rapidly changing value of
our common stock. There have been recent instances of extreme stock price run-ups followed by rapid price declines following public offerings,
particularly among companies with relatively smaller public floats, and we expect that such instances may continue and/or increase in
the future. Contributing to this risk of volatility are a number of factors. First, our common stock is likely to be more sporadically
and thinly traded than that of larger, more established companies. As a consequence of this lack of liquidity, the trading of relatively
small quantities of shares by our shareholders may disproportionately influence the price of those shares in either direction, which
may cause our stock price to deviate, potentially significantly, from a price that better reflects the underlying performance of our
business. The price of our shares could, for example, decline precipitously in the event that a large number of our shares are sold in
the market without commensurate demand as compared to a seasoned issuer that could better absorb those sales without an adverse impact on its
stock price. Second, we are a speculative investment due to our limited operating history, not being profitable, and not expecting to
be profitable in the near term. As a consequence of this enhanced risk, more risk-adverse investors may, under the fear of losing all
or most of their investment in the event of negative news or lack of progress, be more inclined to sell their shares on the market more
quickly and at greater discounts than would be the case with the stock of a larger, more established company that has a relatively large
public float.
Many
of these factors are beyond our control and may decrease the market price of our securities. Such volatility, including any stock run-ups,
may be unrelated or disproportionate to our actual or expected operating performance and financial condition or prospects, making it
difficult for prospective investors to assess the rapidly changing value of our shares.
Furthermore,
the stock market in general, and the market for men’s wellness product companies in particular, have experienced extreme price
and volume fluctuations that have often been unrelated or disproportionate to the operating performance of those companies. Broad market
and industry factors, as well as general economic, political and market conditions such as recessions, or changes in inflation or interest
rates, may seriously affect the market price of our securities, regardless of our actual operating performance. As a result of this volatility,
investors may experience losses on their investment in our common stock. A decline in the market price of our common stock also could
adversely affect our ability to issue additional shares of common stock or other securities and our ability to obtain additional financing
in the future. No assurance can be given that an active market in our common shares will develop or be sustained. If an active market
does not develop, holders of our common stock may be unable to readily sell the shares they hold or may not be able to sell their shares
at all.
Our
common stock prices have been, and may continue to be, volatile and could decline substantially following the date of this Prospectus.
The
market price of our common stock may be highly volatile and subject to wide fluctuations. Our financial performance, government regulatory
action, tax laws, interest rates, and market conditions in general could have a significant impact on the future market price of our
common stock.
Some
of the factors that could negatively affect or result in fluctuations in the market price of our common stock include:
| |
● |
actual
or anticipated variations in our quarterly operating results; |
| |
|
|
| |
● |
changes
in market valuations of similar companies; |
| |
|
|
| |
● |
adverse
market reaction to the level of our indebtedness; |
| |
|
|
| |
● |
additions
or departures of key personnel; |
| |
|
|
| |
● |
actions
by shareholders; |
| |
|
|
| |
● |
speculation
in the press or investment community; |
| |
|
|
| |
● |
general
market, economic, and political conditions, including an economic slowdown or dislocation in the global credit markets; |
| |
● |
announcements
by us or our competitors of significant acquisitions, strategic partnerships, joint ventures, collaborations, or capital commitments; |
| |
|
|
| |
● |
general
economic and market conditions; |
| |
|
|
| |
● |
disputes
or other developments related to our intellectual property or other proprietary rights, including litigation; |
| |
|
|
| |
● |
our
operating performance and the performance of other similar companies; |
| |
|
|
| |
● |
changes
in accounting principles; and |
| |
|
|
| |
● |
passage
of legislation or other regulatory developments that adversely affect us or our industry. |
If
our stock price fluctuates after the offering, you could lose a significant part of your investment.
The
market price of our common stock could be subject to wide fluctuations in response to, among other things, the risk factors described
in this prospectus, and other factors beyond our control, such as fluctuations in the valuation of companies perceived by investors to
be comparable to us For example, since our common stock began trading on the Nasdaq Capital Market in connection with our IPO on March
20, 2023, the trading price of our common stock has traded as high as $4.37 and as low as $0.143 per share. Furthermore, the stock markets
have experienced price and volume fluctuations that have affected and continue to affect the market prices of equity securities of many
companies. These fluctuations often have been unrelated or disproportionate to the operating performance of those companies. These broad
market and industry fluctuations, as well as general economic, political, and market conditions, such as recessions, interest rate changes
or international currency fluctuations, may negatively affect the market price of our common stock. In the past, many companies that
have experienced volatility in the market price of their stock have been subject to securities class action litigation. We may be the
target of this type of litigation in the future. Securities litigation against us could result in substantial costs and divert our management’s
attention from other business concerns, which could seriously harm our business.
If
securities or industry analysts do not publish research or reports about us, or if they adversely change their recommendations regarding
our common stock, then our stock price and trading volume could decline.
The
trading market for our common stock is influenced by the research and reports that industry or securities analysts publish about us,
our industry and our market. If no analyst elects to cover us and publish research or reports about us, the market for our common stock
could be severely limited and our stock price could be adversely affected. As a small-cap company, we are more likely than our larger
competitors to lack coverage from securities analysts. In addition, even if we receive analyst coverage, if one or more analysts ceases
coverage of us or fails to regularly publish reports on us, we could lose visibility in the financial markets, which in turn could cause
our stock price or trading volume to decline. If one or more analysts who elect to cover us issue negative reports or adversely change
their recommendations regarding our common stock, our stock price could decline.
Future
sales of our common stock, other securities convertible into our common stock, or preferred stock could cause the market value of our
common stock to decline and could result in dilution of your shares.
Our
Board of Directors is authorized, without your approval, to cause us to issue additional shares of our common stock or to raise capital
through the creation and issuance of preferred stock, other debt securities convertible into common stock, options, warrants and other
rights, on terms and for consideration as our Board of Directors in its sole discretion may determine. Additionally, pursuant to the
Resale Prospectus, we registered the resale of an aggregate of 4,765,000 shares of common stock, which shares of common stock are available
for immediate resale in the public market (which number includes 2,000,000 shares of common stock issuable upon the exercise of warrants,
of which 975,500 shares of common stock remain issuable thereunder as of the date of this prospectus). An additional (a) 87,500 shares
of common stock are issuable upon exercise of outstanding warrants to purchase shares at $5.00 per share, which were issued in connection
with the IPO; and (b) 322,000 shares of common stock are issuable upon exercise of outstanding warrants to purchase shares at $0.375
per share, which were issued in connection with the Follow On Offering. Sales of substantial amounts of our common stock or of preferred
stock could cause the market price of our common stock to decrease significantly. We cannot predict the effect, if any, of future sales
of our common stock, or the availability of our common stock for future sales, on the value of our common stock. Sales of substantial
amounts of our common stock by large shareholders, or the perception that such sales could occur, may adversely affect the market price
of our common stock.
We
have no intention of declaring dividends in the foreseeable future.
The
decision to pay cash dividends on our common stock rests with our Board of Directors and will depend on our earnings, unencumbered cash,
capital requirements and financial condition. We do not anticipate declaring any dividends in the foreseeable future, as we intend to
use any excess cash to fund our operations. Investors in our common stock should not expect to receive dividend income on their investment,
and investors will be dependent on the appreciation of our common stock to earn a return on their investment.
The
issuance and sale of common stock upon exercise of outstanding warrants may cause substantial dilution to existing shareholders and may
also depress the market price of our common stock. Outstanding warrants to purchase shares of our common stock have cashless exercise
rights.
As
of the date of this prospectus, we had a total of 4,685,000 warrants outstanding with a weighted average exercise price of $0.51
per share and term ranging from August 16, 2027 through October 4, 2029. If the holders of the warrants choose to exercise
the warrants, it may cause significant dilution to the then holders of our common stock. If exercises of the warrants and sales of such
shares issuable upon exercise thereof take place, the price of our common stock may decline. In addition, the common stock issuable upon
exercise of the warrants may represent overhang that may also adversely affect the market price of our common stock. Overhang occurs
when there is a greater supply of a company’s stock in the market than there is demand for that stock. When this happens the price
of our stock will decrease, and any additional shares which shareholders attempt to sell in the market will only further decrease the
share price. If the share volume of our common stock cannot absorb shares sold by the warrant holders, then the value of our common stock
will likely decrease.
A
total of 409,500 of the warrants discussed above (which have an exercise price of $5.00 per share (87,500) and $0.375 (322,000)) currently
allow for cashless exercise rights. In a ‘cashless exercise’, the holder reduces the number of shares of common stock issuable
upon exercise of the warrants in amount equal to the aggregate value of the exercise price of the exercised warrants. For example, if
our common stock was trading at $2.00 per share and a holder desires to exercise warrants to purchase 100 shares of common stock with
an exercise price of $1.00 per share on a cashless basis, the number of shares of common stock issuable to the holder upon such exercise
would be reduced by 50 shares, equal in value to $100 ($2.00 per share x 50 shares), and the holder would receive 50 shares of common
stock upon such exercise. We do not receive any cash upon a cashless exercise and as such, while a cashless exercise reduces the dilution
which would otherwise exist upon a warrant exercise, it is also not as beneficial to us, as it does not bring in any new investment proceeds.
Additionally, holders of warrants with cashless exercise provisions may be more likely to exercise their warrants as they do not have
to come out of pocket with any cash exercise payments.
General
Risk Factors
Our
industry and the broader U.S. economy experienced higher than expected inflationary pressures during 2022 related to continued supply
chain disruptions, labor shortages and geopolitical instability, and if these conditions persist, our business, results of operations
and cash flows could be materially and adversely affected.
2022
saw significant increases in the costs of labor and certain materials and equipment, and longer lead times for such materials and equipment,
as a result of availability constraints, supply chain disruption, increased demand, labor shortages associated with a fully employed
U.S. labor force, high inflation and other factors. Supply and demand fundamentals have been further aggravated by disruptions in global
energy supply caused by multiple geopolitical events, including the ongoing conflict between Russia and Ukraine. Recent supply chain
constraints and inflationary pressures may in the future adversely impact our operating costs, and as a result, our business, financial
condition, results of operations and cash flows could be materially and adversely affected.
We
and the health and wellness industry in general may be adversely affected during periods of high inflation, primarily because of higher
shipping and product manufacturing costs. While we plan to attempt to pass on increases in our costs through increased sales prices,
market forces may limit our ability to do so. If we are unable to raise sales prices enough to compensate for higher costs, our future
revenues, gross profit margin and revenues could be adversely affected.
Economic
uncertainty may affect our access to capital and/or increase the costs of such capital.
Global
economic conditions continue to be volatile and uncertain due to, among other things, consumer confidence in future economic conditions,
fears of recession and trade wars, the price of energy, fluctuating interest rates, the availability and cost of consumer credit, the
availability and timing of government stimulus programs, levels of unemployment, increased inflation, tax rates, and the war between
Ukraine and Russia which began in February 2022, and has continued through the date of this prospectus, as well as the current ongoing
war between Hamas and Israel, which began in October 2023, and has continued through the date of this prospectus. These conditions remain
unpredictable and create uncertainties about our ability to raise capital in the future. In the event required capital becomes unavailable
in the future, or more costly, it could have a material adverse effect on our business, future results of operations, and financial condition.
Our
business may be materially and adversely disrupted by epidemics or pandemics in the future, including COVID-19.
An
epidemic, pandemic or similar serious public health issue, and the measures undertaken by governmental authorities to address it, could
significantly disrupt or prevent us from operating our business in the ordinary course for an extended period, and thereby, and/or along
with any associated economic and/or social instability or distress, have a material adverse impact on our financial statements.
On
March 11, 2020, the World Health Organization characterized the outbreak of COVID-19 as a global pandemic and recommended containment
and mitigation measures. On March 13, 2020, the United States declared a national emergency concerning the outbreak, and several states
and municipalities have declared public health emergencies. The U.S. Congress formally ended the COVID-19 national emergency on April
10, 2023. Although COVID-19 has to date not had a material impact on our operations, should the COVID-19 public health effort re-intensify
to such an extent that we cannot operate, if there are new government restrictions on our business and our customers, and/or an extended
economic recession or significant inflation, we could be unable to produce significant revenues and cash flows sufficient to conduct
our business. Such a circumstance could, among other things, exhaust our available liquidity (and ability to access liquidity sources)
and/or trigger an acceleration to pay a significant portion or all of our then-outstanding debt obligations, which we may be unable to
do.
Our
business could be disrupted by catastrophic events and man-made problems, such as power disruptions, data security breaches, and terrorism.
Our
systems are vulnerable to damage or interruption from the occurrence of any catastrophic event, including earthquake, fire, flood, or
other weather event, power loss, telecommunications failure, software or hardware malfunction, cyber-attack, war, terrorist attack, or
incident of mass violence, which could result in lengthy interruptions in access to our systems. In addition, acts of terrorism, including
malicious internet-based activity, could cause disruptions to the internet or the economy as a whole. If our systems were to fail or
be negatively impacted as a result of a natural disaster or other event, our ability to provide products to customers would be impaired
or we could lose critical data. We do not carry business interruption insurance sufficient to compensate us for the potentially significant
losses, including the potential harm to our business, financial condition and results of operations that may result from interruptions
in access to our platform as a result of system failures.
Economic
uncertainty may affect consumer purchases of discretionary items, which may affect demand for our products.
Our
products may be considered discretionary items for consumers. Factors affecting the level of consumer spending for such discretionary
items include general economic conditions and other factors such as consumer confidence in future economic conditions, fears of recession
and trade wars, the price of energy, fluctuating interest rates, the availability and cost of consumer credit, the availability and timing
of government stimulus programs, levels of unemployment, inflation, and tax rates. As U.S. economic conditions continue to be volatile
or economic uncertainty remains, and with increasing inflation and interest rates, trends in consumer discretionary spending also remain
unpredictable and subject to reductions as a result of significant increases in employment, financial market instability, and uncertainties
about the future. Unfavorable economic conditions have led, and in the future may lead, consumers to reduce their spending on men’s
wellness products, which in turn has in the past led to a decrease in the demand for such products. Consumer demand for the Company’s
products may decline as a result of an economic downturn, or economic uncertainty. The sensitivity to economic cycles and any related
fluctuation in consumer demand may have a material adverse effect on the Company’s business, results of operations, and financial
condition.
In
February 2022, an armed conflict escalated between Russia and Ukraine. The sanctions announced by the United States and other countries
against Russia and Belarus following Russia’s invasion of Ukraine to date include restrictions on selling or importing goods, services,
or technology in or from affected regions and travel bans and asset freezes impacting connected individuals and political, military,
business, and financial organizations in Russia and Belarus. The United States and other countries could impose wider sanctions and take
other actions should the conflict further escalate. Separately, in October 2023, Israel and certain Iranian-backed Palestinian forces
began an armed conflict in Israel, the Gaza Strip, and surrounding areas. This conflict currently threatens to spread to other Middle
Eastern countries, and may ultimately result in the United States and other countries becoming involved in the conflict. Although the
Company does not, and does not plan to, do business in Russia, Belarus, Ukraine, Israel, or the Middle East, it is not possible to predict
the broader consequences of these ongoing conflicts, which could include further sanctions, embargoes, regional instability, and geopolitical
shifts. It is also not possible to predict with certainty these ongoing conflicts and additional adverse effects on existing U.S. macroeconomic
conditions, consumer spending habits, currency exchange rates, and financial markets, all of which could impact the business, financial
condition, and results of operations of the Company.
Global
economic conditions could materially adversely affect our business, results of operations, financial condition and growth.
Adverse
macroeconomic conditions, including inflation, slower growth or recession, new or increased tariffs, changes to fiscal and monetary policy,
tighter credit, higher interest rates, high unemployment and currency fluctuations could materially adversely affect our operations,
expenses, access to capital and the market for our products. In addition, consumer confidence and spending could be adversely affected
in response to financial market volatility, negative financial news, conditions in the real estate and mortgage markets, declines in
income or asset values, changes to fuel and other energy costs, labor and healthcare costs and other economic factors.
In
addition, uncertainty about, or a decline in, global or regional economic conditions could have a significant impact on our expected
funding sources, suppliers and partners. Potential effects include financial instability; inability to obtain credit to finance operations
and purchases of our products; and insolvency.
A
downturn in the economic environment could also lead to limitations on our ability to issue new debt; reduced liquidity; and declines
in the fair value of our financial instruments. These and other economic factors could materially adversely affect our business, results
of operations, financial condition and growth.
We
may become party to litigation, mediation and/or arbitration from time to time given our product focus.
We
may become party to regulatory proceedings, litigation, mediation and/or arbitration from time to time in the ordinary course of business
which could adversely affect our business. Monitoring and defending against legal actions, whether or not meritorious, can be time-consuming,
divert management’s attention and resources and cause us to incur significant expenses. In addition, legal fees and costs incurred
in connection with such activities may be significant and we could, in the future, be subject to judgments or enter into settlements
of claims for significant monetary damages. While we expect to have insurance in the future that may cover the costs and awards of certain
types of litigation, the amount of our future insurance may not be sufficient to cover any costs or awards. Substantial litigation costs
or an adverse result in any litigation may adversely impact our business, operating results or financial condition.
Higher
labor costs due to statutory and regulatory changes could materially adversely affect our business, financial condition and operating
results.
Various
federal and state labor laws, including new laws and regulations enacted in response to COVID-19, govern our relationships with our employees
and affect operating costs. These laws include employee classifications as exempt or non-exempt, minimum wage requirements, unemployment
tax rates, workers’ compensation rates, overtime, family leave, workplace health and safety standards, payroll taxes, citizenship
requirements and other wage and benefit requirements for employees classified as non-exempt. As certain of our employees are paid at
rates set at, or above but related to, the applicable minimum wage, further increases in the minimum wage could increase our labor costs.
Significant additional government regulations could materially adversely affect our business, financial condition and operating results.
Failure
to adequately manage our planned aggressive growth strategy may harm our business or increase our risk of failure.
For
the foreseeable future, we intend to pursue an aggressive growth strategy for the expansion of our operations through increased marketing.
Our ability to rapidly expand our operations will depend upon many factors, including our ability to work in a regulated environment,
establish and maintain strategic relationships with suppliers, and obtain adequate capital resources on acceptable terms. Any restrictions
on our ability to expand may have a materially adverse effect on our business, results of operations, and financial condition. Accordingly,
we may be unable to achieve our targets for sales growth, and our operations may not be successful or achieve anticipated operating results.
Additionally,
our growth may place a significant strain on our managerial, administrative, operational, and financial resources and our infrastructure.
Our future success will depend, in part, upon the ability of our senior management to manage growth effectively. This will require us
to, among other things:
| |
● |
implement
additional management information systems; |
| |
|
|
| |
● |
further
develop our operating, administrative, legal, financial, and accounting systems and controls; |
| |
|
|
| |
● |
hire
additional personnel; |
| |
|
|
| |
● |
develop
additional levels of management within our company; |
| |
|
|
| |
● |
locate
additional office space; and |
| |
|
|
| |
● |
maintain
close coordination among our operations, legal, finance, sales and marketing, and client service and support personnel. |
As
a result, we may lack the resources to deploy our services on a timely and cost-effective basis. Failure to accomplish any of these requirements
could impair our ability to deliver services in a timely fashion or attract and retain new customers.
If
we make any acquisitions, they may disrupt or have a negative impact on our business.
If
we make acquisitions in the future, we could have difficulty integrating the acquired company’s assets, personnel and operations
with our own. We do not anticipate that any acquisitions or mergers we may enter into in the future would result in a change of control
of the Company. In addition, the key personnel of the acquired business may not be willing to work for us. We cannot predict the effect
expansion may have on our core business. Regardless of whether we are successful in making an acquisition, the negotiations could disrupt
our ongoing business, distract our management and employees and increase our expenses. In addition to the risks described above, acquisitions
are accompanied by a number of inherent risks, including, without limitation, the following:
| |
● |
the
difficulty of integrating acquired products, services or operations; |
| |
|
|
| |
● |
the
potential disruption of the ongoing businesses and distraction of our management and the management of acquired companies; |
| |
|
|
| |
● |
difficulties
in maintaining uniform standards, controls, procedures and policies; |
| |
|
|
| |
● |
the
potential impairment of relationships with employees and customers as a result of any integration of new management personnel; |
| |
|
|
| |
● |
the
potential inability or failure to achieve additional sales and enhance our customer base through cross-marketing of the products
to new and existing customers; |
| |
|
|
| |
● |
the
effect of any government regulations which relate to the business acquired; |
| |
|
|
| |
● |
potential
unknown liabilities associated with acquired businesses or product lines, or the need to spend significant amounts to retool, reposition
or modify the marketing and sales of acquired products or operations, or the defense of any litigation, whether or not successful,
resulting from actions of the acquired company prior to our acquisition; and |
| |
|
|
| |
● |
potential
expenses under the labor, environmental and other laws of various jurisdictions. |
Our
business could be severely impaired if and to the extent that we are unable to succeed in addressing any of these risks or other problems
encountered in connection with an acquisition, many of which cannot be presently identified. These risks and problems could disrupt our
ongoing business, distract our management and employees, increase our expenses and adversely affect our results of operations.
Claims,
litigation, government investigations, and other proceedings may adversely affect our business and results of operations.
We
may be subject to actual and threatened claims, litigation, reviews, investigations, and other proceedings, including proceedings relating
to products offered by us and by third parties, and other matters. Any of these types of proceedings, may have an adverse effect on us
because of legal costs, disruption of our operations, diversion of management resources, negative publicity, and other factors. The outcomes
of these matters are inherently unpredictable and subject to significant uncertainties. Determining legal reserves and possible losses
from such matters involves judgment and may not reflect the full range of uncertainties and unpredictable outcomes. Until the final resolution
of such matters, we may be exposed to losses in excess of the amount recorded, and such amounts could be material. Should any of our
estimates and assumptions change or prove to have been incorrect, it could have a material effect on our business, consolidated financial
position, results of operations, or cash flows. In addition, it is possible that a resolution of one or more such proceedings, including
as a result of a settlement, could require us to make substantial future payments, prevent us from offering certain products or services,
require us to change our business practices in a manner materially adverse to our business, requiring development of non-infringing or
otherwise altered products or technologies, damaging our reputation, or otherwise having a material effect on our operations.
We
may incur indebtedness in the future which could reduce our financial flexibility, increase interest expense and adversely impact our
operations and our costs.
We
may incur significant amounts of indebtedness in the future. Our level of indebtedness could affect our operations in several ways, including
the following:
| |
● |
a
significant portion of our cash flows is required to be used to service our indebtedness; |
| |
|
|
| |
● |
a
high level of debt increases our vulnerability to general adverse economic and industry conditions; |
| |
|
|
| |
● |
covenants
contained in the agreements governing our outstanding indebtedness limit our ability to borrow additional funds and provide additional
security interests, dispose of assets, pay dividends and make certain investments; |
| |
|
|
| |
● |
a
high level of debt may place us at a competitive disadvantage compared to our competitors that are less leveraged and, therefore,
may be able to take advantage of opportunities that our indebtedness may prevent us from pursuing; and |
| |
|
|
| |
● |
debt
covenants may affect our flexibility in planning for, and reacting to, changes in the economy and in our industry. |
A
high level of indebtedness increases the risk that we may default on our debt obligations. We may not be able to generate sufficient
cash flows to pay the principal or interest on our debt, and future working capital, borrowings or equity financing may not be available
to pay or refinance such debt. If we do not have sufficient funds and are otherwise unable to arrange financing, we may have to sell
significant assets or have a portion of our assets foreclosed upon which could have a material adverse effect on our business, financial
condition and results of operations.
For
all of the foregoing reasons and others set forth herein, an investment in our securities involves a high degree of risk.
Additional
Risk Factors:
Investors
who buy shares in this offering at different times will likely pay different prices.
Investors
who purchase shares of common stock in this offering at different times will likely pay different prices, and so may experience different
levels of dilution and different outcomes in their investment results. In connection with the ELOC, we will have discretion, subject
to market demand, to vary the timing, prices, and numbers of shares of common stock sold to the Purchaser. Similarly, the Purchaser
may sell such shares of common stock at different times and at different prices. Investors may experience a decline in the value
of the shares they purchase from the Purchaser in this offering as a result of sales made by us in future transactions to the
Purchaser at prices lower than the prices they paid. Sales to the Purchaser by us could result in substantial dilution
to the interests of other holders of our common stock. Additionally, the sale of a substantial number of shares of our common stock to
the Purchaser, or the anticipation of such sales, could make it more difficult for us to sell equity or equity-related securities
in the future at a time and at a price that we might otherwise wish to effect sales, which could have a materially adverse effect on
our business and operations.
Our
management will have broad discretion over the use of the net proceeds from our sale of shares of common stock to the Purchaser,
and you may not agree with how we use the proceeds and the proceeds may not be invested successfully.
Our
management will have broad discretion with respect to the use of proceeds from the sale of any shares of our common stock to the Purchaser,
including for any of the purposes described in the section of this prospectus entitled “Use of Proceeds.” You will be relying
on the judgment of our management regarding the application of the proceeds from the sale of any shares of our common stock to the Purchaser.
The results and effectiveness of the use of proceeds are uncertain, and we could spend the proceeds in ways that you do not agree
with or that do not improve our results of operations or enhance the value of our common stock. Our failure to apply these funds effectively
could harm our business, delay the development of our pipeline product candidates and cause the price of our common stock to decline.
We
may require additional financing to sustain our operations, without which we may not be able to continue operations, and the terms of
subsequent financings may adversely impact our stockholders.
We
may direct the Purchaser to purchase up to $25,000,000 worth of shares of our common stock under the ELOC until April
4, 2026, in amounts up to $25,000,000 in shares of our common stock depending on market prices.
Our
ability to sell shares to the Purchaser and obtain funds under the ELOC is limited by the terms and conditions in the ELOC,
including restrictions on the amounts we may sell to the Purchaser at any one time, and a limitation on our ability to sell shares
to the Purchaser to the extent that it would cause the Purchaser to beneficially own more than 9.99% of our outstanding
shares of common stock. Additionally, we will only be able to sell or issue to the Purchaser (subject to certain reductions and
other adjustments pursuant to the ELOC, the “Exchange Cap”) in total under the ELOC, which is equal to 19.99%
of the aggregate number of shares outstanding prior to execution of the ELOC, unless stockholder approval is obtained to issue
in excess of such amount. Therefore, we may not in the future have access to the full amount available to us under the ELOC, depending
on the price of our common stock. In addition, any amounts we sell under the ELOC may not satisfy all of our funding needs, even
if we are able and choose to sell and issue all of our common stock currently registered.
The
extent we rely on the Purchaser as a source of funding will depend on a number of factors including the prevailing market price
of our common stock and the extent to which we are able to secure working capital from other sources. If obtaining sufficient funding
from the Purchaser were to prove unavailable or prohibitively dilutive, we will need to secure another source of funding in order
to satisfy our working capital needs. Depending on the type and the terms of any financing we pursue, stockholders’ rights and
the value of their investment in our common stock could be reduced. A financing could involve one or more types of securities including
common stock, convertible debt, or warrants to acquire common stock. These securities could be issued at or below the then prevailing
market price for our common stock. If the issuance of new securities results in diminished rights to holders of our common stock, the
market price of our common stock could be negatively impacted. Should the financing we require to sustain our working capital needs be
unavailable or prohibitively expensive when we require it, the consequences could be a material adverse effect on our business, operating
results, financial condition, and prospects.
USE
OF PROCEEDS
Any
proceeds received by the Company from the sale of shares of common stock pursuant to the ELOC and from the exercise of the Warrants
will be used for general working capital. A portion of the net proceeds from the sale of our common stock will go to the selling stockholders
as described below in the sections entitled “Selling Stockholders” and “Plan of Distribution”. We have agreed
to bear the expenses relating to the registration of the shares of common stock for the selling stockholders.
Dividend
Policy
We
have never declared or paid any cash dividends on our capital stock. We currently intend to retain earnings, if any, to finance the growth
and development of our business. We do not expect to pay any cash dividends on our common stock in the foreseeable future. Payment of
future dividends, if any, will be at the discretion of our Board of Directors and will depend on our financial condition, results of
operations, capital requirements, restrictions contained in any financing instruments, provisions of applicable law and other factors
the board deems relevant
Management’s
Discussion and Analysis of
Financial
Condition and Results of Operations
The
following discussion of the Company’s historical performance and financial condition should be read together with the
consolidated financial statements and related notes included herein. This discussion contains forward-looking statements based on
the views and beliefs of our management, as well as assumptions and estimates made by our management. These statements by their
nature are subject to risks and uncertainties, and are influenced by various factors. As a consequence, actual results may differ
materially from those in the forward-looking statements. See “I Risk Factors” included herein for
the discussion of risk factors and see “Cautionary Statement Regarding Forward-Looking Statements”
for information on the forward-looking statements included below.
The
following discussion is based upon our financial statements included elsewhere in in this prospectus, which have been prepared in accordance
with U.S. generally accepted accounting principles. The preparation of these financial statements requires us to make estimates and judgments
that affect the reported amounts of assets, liabilities, revenues and expenses, and related disclosure of contingencies.
Introduction
Our
Management’s Discussion and Analysis of Financial Condition and Results of Operations (“MD&A”) is provided in addition
to the accompanying financial statements and notes to assist readers in understanding our results of operations, financial condition,
and cash flows. MD&A is organized as follows:
| ● |
Key
Performance Indicators. Indicators describing our performance for the periods presented. |
| |
|
| ● |
Plan
of Operations. A description of our plan of operations for the next 12 months including required funding. |
| |
|
| ● |
Results
of Operations. An analysis of our financial results comparing the years ended December 31, 2023 and 2022. |
| |
|
| ● |
Liquidity
and Capital Resources. An analysis of changes in our balance sheets and cash flows and discussion of our financial condition. |
| |
|
| ● |
Critical
Accounting Policies and Estimates. Accounting estimates that we believe are important to understanding the assumptions and judgments
incorporated in our reported financial results and forecasts. |
See
also “Glossary of Industry Terms” above for information on certain of the terms used below.
Plan
of Operations
We
had working capital of $0.7 million as of December 31, 2023. With our current cash on hand, expected revenues, and based on our current
average monthly expenses, we currently anticipate the need for additional funding in order to continue our operations at their current
levels and to pay the costs associated with being a public company for the next 12 months. We may also require additional funding in
the future to expand or complete acquisitions.
Our
plan for the next 12 months is to continue using the same marketing and management strategies and continue providing a quality product
with excellent customer service while also seeking to expand our operations organically or through acquisitions as funding and opportunities
arise. As our business continues to grow, customer feedback will be integral in making small adjustments to improve products and our
overall customer experience.
We
are headquartered in Dallas, Texas and intend to grow our business both organically and through identifying acquisition targets over
the next 12 months in the technology, health and wellness space, funding permitting. Specifically, we plan to continue to make additional
and ongoing technology enhancements to our platform, further develop, market and advertise additional men’s health and wellness
related products on our telemedicine platform, and identify strategic acquisitions that complement our vision. As these opportunities
arise, we will determine the best method for financing such acquisitions and growth which may include the issuance of debt instruments,
common stock, preferred stock, or a combination thereof, all of which may result in significant dilution to existing shareholders.
We
may seek additional funding in the future through equity financings, debt financings or other capital sources, including collaborations
with other companies or other strategic transactions. We may not be able to obtain financing on acceptable terms or at all. The terms
of any financing may adversely affect the holdings or rights of our shareholders and/or create significant dilution. Although we continue
to pursue these plans, there is no assurance that we will be successful in obtaining sufficient funding on terms acceptable to us to
fund continued operations, if at all.
Results
of Operations
Comparison
of the Year Ended December 31, 2023 and 2022
Revenues
We
began generating revenues in November 2022 and had revenues of $731,493 and $8,939 for the years ended December 31, 2023 and 2022, respectively.
Cost
of Revenues
We
had cost of revenues of $154,900 and $4,089 for the years ended December 31, 2023 and 2022, respectively, relating to amounts paid to
Epiq Scripts, a related party, 51% owned and controlled by Jacob D. Cohen, our Chairman and Chief Executive Officer, which entity provides
us pharmacy and compounding services, resulting in gross profit of $431,501 and $4,850 for the years ended December 31, 2023 and 2022,
respectively. The related party cost of revenues was associated with the Master Services Agreement entered into with Epiq Scripts and
a related statement of work and the remaining cost of revenues was attributed to the amounts paid to our unrelated party doctors network
and shipping expenses.
The
Company analyzed the following factors when determining the amounts to be paid to Epiq Scripts under the Master Services Agreement and
related statement of work: a) the fairness of the terms for the Company (including fairness from a financial point of view); b) the materiality
of the transaction; c) bids / terms for a similar transaction from unrelated parties; d) the structure of the transaction; and e) the
interests of each related party in the transaction.
Operating
Expenses and Net Loss
We
had total general and administrative expenses of $9,608,409 and $1,996,432 and imputed interest expense of $0 and $6,473 (which represented
imputed interest on the related party loans which were repaid as discussed below under “Liquidity and Capital Resources”)
for the years ended December 31, 2023 and 2022, respectively, resulting in a net loss of $9,212,417 and $1,998,055, respectively, for
the years ended December 31, 2023 and 2022.
The
increase in general administration expenses for the years ended December 31, 2023 and 2022, compared to the prior period, was due primarily
to (a) stock-based compensation totaling $2,155,144 and $774,153 (including a total of $1,530,651 and $540,065 attributed to stock issued
for services and $624,463 and $234,088 attributed to stock-based compensation from issuances of options and warrants), respectively,
which increase was due to us having issued less stock for compensation during the 2022 period; (b) advertising and marketing expenses
in the amount of $2,097,505 and $352,860, for the years ended December 31, 2023 and 2022, respectively, related to us increasing our
advertising and marketing costs in the 2023 period as we ramped up our marketing efforts in connection with the expansion of our operations;
(c) legal fees of $327,055 and $231,799, for the years ended December 31, 2023 and 2022, respectively, mainly related to legal fees in
connection with our initial public offering and related matters; (d) placement agent fees of $496,000 and $160,000, for the years ended
December 2023 and 2022, respectively, relating to fees paid to our placement agent in connection with our private placement and initial
public offering; (e) salaries and benefits of $914,115 and $164,941 for the years ended December 31, 2023 and 2022, respectively, which
increased due to the engagement of new employees as we ramped up our operations in the current period; (f) accounting and auditing fees
of $121,330 and $44,500, for the years ended December 2023 and 2022, respectively, which was in connection with fees paid to our accountants
and auditors in connection with the preparation of the financial statements for our initial public offering , quarterly reviews, and
annual filing; (g) general consulting related expenses of $585,729 and $622,331, for the years ended December 31, 2023 and 2022, respectively,
related to other various consulting fees paid in connection with our operations in the current period; and (h) software development fees
of $434,490 and $72,440 for the years ended December 2023 and 2022, respectively, related to the front and backend development of our
website in the current period. Software development expenses are integral to customers accessing our ordering system and successfully
placing an order for our products. We had not yet implemented our online ordering in the first nine months of 2022.
Liquidity
and Capital Resources
As
of December 31, 2023, we had $739,006 of cash on-hand, compared to $682,860 of cash on-hand of December 31, 2022. We also had $60,953
of prepaid expenses, related party, relating to amounts funded to Epiq Scripts, which is 51% owned and controlled by Jacob D. Cohen,
our Chairman and Chief Executive Officer, $18,501 of inventory; $96,129 of property and equipment, net, consisting of computers, office
and custom product packaging equipment, $16,942 of security deposit, representing the security deposit on our leased office space and
$119,262 of right of use asset in connection with our office space lease. Cash increased mainly due to funds raised in the IPO and Follow
On Offering, offset by cash used for general operating expenses.
As
of December 31, 2023, the Company had total current liabilities of $276,039, consisting of $140,765 of accounts payable and accrued liabilities,
$6,595 of payroll tax liabilities, and $63,718 of right-of-use liability, operating lease, current portion. We also had $64,961 of right-of-use
liability, long-term.
As
of December 31, 2023, we had $1,050,793 in total assets, $276,039 in total liabilities, working capital of $0.6 million and a total accumulated
deficit of $11,228,173.
We
have mainly relied on related party loans, as well as funds raised through the sale of securities, mainly through the private placement
offering, our IPO and our Follow On Offering, each discussed below, and revenues generated from sales of our Mango ED and Mango GROW
products, to support our operations since inception. We have primarily used our available cash to pay operating expenses. We do not have
any material commitments for capital expenditures.
We
have experienced recurring net losses since inception. We believe that we will continue to incur substantial operating expenses in the
foreseeable future as we continue to invest to bring our Mango ED and Mango GROW products to market and to attract customers, expand
the product offerings and enhance technology and infrastructure. These efforts may prove more expensive than we anticipate, and we may
not succeed in generating commercial revenues or net income to offset these expenses. Accordingly, we may not be able to achieve profitability,
and we may incur significant losses for the foreseeable future. Our independent registered public accounting firm included an explanatory
paragraph in its report on our financial statements as of December 31, 2023. As of December 31, 2023, our current capital resources,
combined with the net proceeds from the offering, are not expected to be sufficient for us to fund operations for the next 12 months.
We need to raise funding in addition to the funding raised in our IPO and Follow On Offering, to support our operations in the future.
We may also seek to acquire additional businesses or assets in the future, which may require us to raise funding. We currently anticipate
such funding being raised through the offering of debt or equity. Such additional financing, if required, may not be available on favorable
terms, if at all. If debt financing is available and obtained, our interest expense may increase and we may be subject to the risk of
default, depending on the terms of such financing. If equity financing is available and obtained it may result in our shareholders experiencing
significant dilution. If such financing is unavailable, we may be forced to curtail our business plan, which may cause the value of our
securities to decline in value.
To
support our existing operations or any future expansion of business, including the ability to execute our growth strategy, we must have
sufficient capital to continue to make investments and fund operations. We have plans to pursue an aggressive growth strategy for the
expansion of operations through marketing to attract new customers for our Mango ED and Mango GROW products.
Cash
Flows
| | |
Year ended December
31, 2023 | | |
Year ended December
31, 2022 | |
| Cash provided by (used in): | |
| | | |
| | |
| Operating activities | |
$ | (6,997,375 | ) | |
$ | (1,346,518 | ) |
| Investing activities | |
| (3,519 | ) | |
| (43,102 | ) |
| Financing activities | |
| 7,057,040 | | |
| 2,047,930 | |
| Net increase in cash | |
$ | 56,146 | | |
$ | 660,310 | |
Net
cash used in operating activities was $6,997,375 for the year ended December 31, 2023, which was mainly due to $9,212,417 of net loss,
offset by $1,530,651 of common stock issued for services, $624,463 for options vested for stock-based compensation.
Net
cash used in operating activities was $1,346,518 for the year ended December 31, 2022, which was mainly due to $1,998,055 of net loss
offset by $540,065 of common stock issued for services and $234,088 for options vested for stock-based compensation.
Net
cash used in investing activities was $3,519 for the year ended December 31, 2023, compared to $43,102 for the year ended December 31,
2022, which were due to the purchase of equipment.
Net
cash provided by financing activities was $7,057,040 for the year ended December 31, 2023, which was mainly due to $6,200,000 of funds
raised in the IPO and Follow On Funding and $1,024,500 in proceeds from the exercise of warrants, offset by repayments of notes payable
of $78,260 and repayments of related party notes payable of $89,200.
Net
cash provided by financing activities was $2,049,930 for the year ended December 31, 2022, which was mainly due to $2,000,000 of proceeds
from the sale of common stock in our private offering, discussed below.
Related
Party Loans and Advances
On
December 10, 2021, the Company received an advance of $70 from ZipDoctor, Inc., a wholly-owned subsidiary of its then sole shareholder,
American International, which was used to open and establish the Company’s bank account. The advance bears no interest and is due
on demand upon the Company’s ability to repay the advance from either future revenues or investment proceeds. The amount owed to
ZipDoctor was $70 as of December 31, 2021. Imputed interest equal to 8% per annum, or $0, was recorded against the related party advance
as of December 31, 2021. The amount was paid in full on May 24, 2022 and the amount owed to ZipDoctor was $0 as of December 31, 2022.
On
December 10, 2021 and March 18, 2022, the Company received advances of $39,200 and $50,000, respectively, for a total of $89,200 from
its previous majority shareholder, American International, in order to cover various general and administrative expenses. The amount
owed to American International was $39,200 as of December 31, 2021. Imputed interest equal to 8% per annum, or $181, was recorded against
the related party advance as of December 31, 2021. Other than the imputed interest discussed above, the advances bear no interest and
are due on demand upon the Company’s ability to repay the advances from either future revenues or investment proceeds. Pursuant
to the terms of the June 16, 2022, SPA discussed below, on June 16, 2022, Cohen Enterprises also acquired the right to be repaid the
$89,200 advanced from American International to the Company.
On
June 16, 2022, American International entered into and closed the transactions contemplated by a Stock Purchase Agreement (the “SPA”),
with Cohen Enterprises, Inc. (“Cohen Enterprises”), which entity is owned by Jacob D. Cohen, the Chairman and Chief Executive
Officer of the Company, who is also the majority shareholder of the Company. Pursuant to the SPA, American International sold 8,000,000
shares of the outstanding common stock of the Company which represented 80% of the then outstanding shares of common stock of the Company,
to Cohen Enterprises in consideration for $90,000, which was approximately the same amount that had been advanced to the Company from
American International through the date of the SPA ($89,200). Cohen Enterprises also acquired the right to be repaid the $89,200 advanced
from American International to the Company, from the Company, pursuant to the terms of the SPA. As a result of the closing of the SPA,
Cohen Enterprises increased its ownership of the Company to 90% (with the remaining 10% of the Company then being owned by Mr. Arango
(or former President and Director), as discussed above), and American International completely divested its interest in the Company.
On
June 29, 2022, the Company received an advance of $25,000 from Cohen Enterprises, which is owned by Mr. Cohen, the Chairman and Chief
Executive Officer of the Company, who is also the majority shareholder of the Company, in order to cover various general and administrative
expenses. The Company repaid Cohen Enterprises $25,000 on August 18, 2022 and the remaining $89,200 on April 4, 2023, bringing the total
amount owed to Cohen Enterprises to $0 as of December 31, 2023. The Company further recorded a credit of $6,473 towards imputed interest,
as other income (previously calculated at a rate of 8% per annum) against the related party advances for the year ended December 31,
2023.
On
November 18, 2022, the Company entered into a Secured Installment Promissory Note with a vendor for the purchase of equipment in the
amount of $78,260 (the “Note Payable”). The note bears no interest unless an event of default occurs, and then it bears interest
at the rate of 10% per annum until paid in full. The Note Payable was payable in installments, requiring payments of $5,000 on each of
January 1, 2023, February 1, 2023, and March 1, 2023, with a $31,630 payment due on April 1, 2023 and a final payment due on May 1, 2023.
The January 1 and March 1, 2023 payments were timely made and on March 23, 2023, the Company elected to pay off the remaining balance
of $63,260. The outstanding balance on December 31, 2022 was $78,260 and as of December 31, 2023, was $0.
2022
Private Placement
In
August 2022, the Company initiated a private placement of up to $2 million of units to accredited investors, with each unit consisting
of one share of common stock and a warrant to purchase one share of common stock, at a price of $1.00 per unit. The warrants have a five-year
term (from each closing date that units were sold) and an exercise price of $1.00 per share. If at any time after the six-month anniversary
of the issuance date, there is no effective registration statement registering, or no current prospectus available for the resale of
the shares of common stock issuable upon exercise the warrants, the holder of the warrants may elect a cashless exercise of the warrants.
Boustead Securities, LLC, the representative of the underwriters in our IPO, served as the placement agent in connection with the private
placement. In total, we sold an aggregate of 2,000,000 units for $2,000,000 to 23 accredited investors between August 16, 2022 and December
22, 2022, the end date of the offering.
Initial
Public Offering
On
March 23, 2023, we consummated our IPO of 1,250,000 shares of common stock at a price to the public of $4.00 per share, pursuant to that
certain Underwriting Agreement, dated March 20, 2023, between the Company and Boustead Securities, LLC, as representative of several
underwriters named in the Underwriting Agreement. The Company received gross proceeds of approximately $5 million, before deducting underwriting
discounts and commissions and estimated offering expenses payable by the Company upon the sale of the shares. In connection with the
IPO, the Company also granted Boustead a 45-day option to purchase up to an additional 187,500 shares of its common stock, which expired
unexercised.
At
the same time, and as part of the same registration statement, but pursuant to a separate prospectus the Company registered the sale
of 4,765,000 shares of common stock, including 2,000,000 shares of common stock issuable upon the exercise of outstanding warrants to
purchase shares of common stock with an exercise price of $1.00 per share, of which warrants to purchase 975,500 shares of common stock
remain outstanding, and unexercised, as of the date of this prospectus.
As
additional consideration in connection with the IPO, we granted Boustead, the representative of the underwriters named in the Underwriting
Agreement for the IPO, warrants to purchase 87,500 shares of common stock with an exercise price of $5.00 per share, which are exercisable
beginning six months after the effective date of the registration statement filed in connection with the IPO (March 20, 2023) and expire
five years after such effectiveness date.
At
the same time, and as part of the same registration statement, but pursuant to a separate prospectus (the “Resale Prospectus”)
the Company registered the sale of 4,765,000 shares of common stock, including 2,000,000 shares of common stock issuable upon the exercise
of outstanding warrants to purchase shares of common stock with an exercise price of $1.00 per share, of which warrants to purchase 975,500
shares of common stock remain outstanding and unexercised.
As
additional consideration in connection with the IPO, upon the closing of the IPO, we granted Boustead, the representative of the underwriters
named in the Underwriting Agreement for the IPO, warrants to purchase 87,500 shares of common stock with an exercise price of $5.00 per
share, which are exercisable beginning six months after the effective date of the registration statement filed in connection with the
IPO (March 20, 2023) and expire five years after such effectiveness date.
Follow
On Offering
On
December 15, 2023, we entered into another underwriting agreement (the “Underwriting Agreement”) with Boustead, as representative
of the underwriters named on Schedule 1 thereto (the “Underwriters”), relating to a public offering of 4,000,000 shares of
the Company’s common stock to the Underwriters at a purchase price to the public of $0.30 per share and also granted to the Underwriters
a 45-day option to purchase up to 600,000 additional shares of its common stock, solely to cover over-allotments, if any, at the public
offering price less the underwriting discounts.
The
Follow On Offering closed on December 19, 2023. As a result, the Company sold 4,000,000 shares of its common stock for total gross proceeds
of $1.2 million.
The
net proceeds to the Company from the Offering, after deducting the underwriting discounts and commissions and offering expenses, were
approximately $1.0 million. The Company used the net proceeds from the Offering to finance the marketing and operational expenses associated
with the planned marketing of its Mango ED and GROW hair growth products, to develop and maintain software, and for working capital and
other general corporate purposes.
We
and our directors, executive officers, and shareholders holding 5% or more of our outstanding common stock previously agreed, in connection
with our IPO, subject to certain exceptions and without the approval of Boustead, not to offer, issue, sell, contract to sell, encumber,
grant any option for the sale of or otherwise dispose of any of our securities until March 20, 2024, and any directors or officers who
did not enter into a lock-up agreement in connection with our IPO entered into a lock-up agreement in connection with the Follow On Offering,
agreeing to not to offer, issue, sell, contract to sell, encumber, grant any option for the sale of or otherwise dispose of any of our
securities for a period of 90 days after December 14, 2023.
On
December 19, 2023, pursuant to the Underwriting Agreement, the Company issued a common stock purchase warrant to Boustead for the purchase
of 280,000 shares of common stock at an exercise price of $0.38, subject to adjustments. The warrant is exercisable at any time and from
time to time, in whole or in part, until December 14, 2029, and may be exercised on a cashless basis.
On
January 18, 2024, the Underwriters notified the Company that they were exercising their over-allotment option in full to purchase an
additional 600,000 shares of common stock, which sale closed on January 22, 2024. The net proceeds to the Company from the sale of the
600,000 shares of common stock, after deducting underwriting discounts and expenses, was approximately $160,000. Inclusive of the full
exercise of the over-allotment option, a total of 4,600,000 shares of common stock were issued and sold in the Offering.
On
January 22, 2024, pursuant to the Underwriting Agreement, the Company also issued a common stock purchase warrant to Boustead for the
purchase of 42,000 shares of common stock at an exercise price of $0.375, subject to adjustments. The warrant is exercisable at any time
and from time to time, in whole or in part, until December 14, 2028, and may be exercised on a cashless basis.
Need
for Future Funding
As
discussed above, our current capital resources, combined with the net proceeds from the offering, are not expected to be sufficient for
us to fund operations for the next 12 months. We believe we will need funding in addition to the funding raised in our IPO and Follow
On Offering, to support our operations in the future. We may also seek to acquire additional businesses or assets in the future, which
may require us to raise funding. We currently anticipate such funding, if required, being raised through the offering of debt or equity.
Such additional financing, if required, may not be available on favorable terms, if at all. If debt financing is available and obtained,
our interest expense may increase and we may be subject to the risk of default, depending on the terms of such financing. If equity financing
is available and obtained it may result in our shareholders experiencing significant dilution. If such financing is unavailable, we may
be forced to curtail our business plan, which may cause the value of our securities to decline in value.
Critical
Accounting Policies and Estimates
The
preparation of the Company’s financial statements in accordance with accounting principles generally accepted in the United States
of America (“GAAP”) requires management to make estimates and assumptions that affect the reported amounts of assets, liabilities
and expenses. “Note 2 - Summary of Significant Accounting Policies” to the audited financial statements included under “Index
to Financial Statements,” below describes the significant accounting policies used in the preparation of the financial statements.
Certain of these significant accounting policies and estimates have a higher degree of inherent uncertainty and require significant judgments.
Accordingly, actual results could differ from those estimates. To the extent that there are differences between our estimates and actual
results, our future financial statement presentation, financial condition, results of operations and cash flows will be affected.
A
critical accounting policy is defined as one that is both material to the presentation of our financial statements and requires management
to make difficult, subjective or complex judgments that could have a material effect on our financial condition and results of operations.
Specifically, critical accounting estimates have the following attributes: (1) we are required to make assumptions about matters that
are highly uncertain at the time of the estimate; and (2) different estimates we could reasonably have used, or changes in the estimate
that are reasonably likely to occur, would have a material effect on our financial condition or results of operations.
Estimates
and assumptions about future events and their effects cannot be determined with certainty. We base our estimates on historical experience
and on various other assumptions believed to be applicable and reasonable under the circumstances. These estimates may change as new
events occur, as additional information is obtained and as our operating environment changes. These changes have historically been minor
and have been included in the financial statements as soon as they became known. Based on a critical assessment of our accounting policies
and the underlying judgments and uncertainties affecting the application of those policies, management believes that our financial statements
are fairly stated in accordance with GAAP and present a meaningful presentation of our financial condition and results of operations.
We believe the following critical accounting policies reflect our more significant estimates and assumptions used in the preparation
of our consolidated financial statements:
Share-Based
Compensation - Stock-based compensation is accounted for based on the requirements of the Share-Based Payment Topic of Financial
Accounting Standards Board (“FASB”) Accounting Standards Codification (“ASC”) 718, which requires recognition
in the consolidated financial statements of the cost of employee and director services received in exchange for an award of equity instruments
over the shorter of period the employee or director is required to perform the services in exchange for the award or the vesting period.
ASC 718 also requires measurement of the cost of employee and director services received in exchange for an award based on the grant-date
fair value of the award. Pursuant to ASC 505-50, for share-based payments to non-employees, compensation expense is determined at the
“measurement date.” The expense is recognized over the service period of the award. Until the measurement date is reached,
the total amount of compensation expense remains uncertain. The Company initially records compensation expense based on the fair value
of the award at the reporting date. Additionally, we used this same methodology when determining the fair value of our restricted common
stock issuances to managers and other related parties.
Estimating
the Fair Value of Common Stock - We are required to estimate the fair value of the common stock underlying our stock-based awards
and warrants when performing the fair value calculations using the Black-Scholes option pricing model
Our
determination of the fair value of stock options with time-based vesting on the date of grant utilizes the Black-Scholes option pricing
model, and is impacted by our common stock price as well as other variables including, but not limited to, expected term that options
will remain outstanding, expected common stock price volatility over the term of the option awards, risk-free interest rates and expected
dividends. Estimating the fair value of equity-settled awards as of the grant date using valuation models, such as the Black-Scholes
option pricing model, is affected by assumptions regarding a number of complex variables. Changes in the assumptions can materially affect
the fair value and ultimately how much stock-based compensation expense is recognized. These inputs are subjective and generally require
significant analysis and judgment to develop.
Warrants
- In accordance with ASC 480, the Company classifies as equity any contracts that (i) require physical settlement or net-share
settlement or (ii) gives the Company a choice of net-cash settlement in its own shares. The Company classifies as liabilities any contracts
that (i) require net-cash settlement (including a requirement to net-cash settle the contract if an event occurs and if that event is
outside the control of the Company) or (ii) give the counterparty a choice of net-cash settlement or settlement in shares.
The
Company accounts for its currently issued warrants in conjunction with the Company’s ordinary shares in permanent equity. These
warrants are indexed to the Company’s stock and meet the requirements of equity classification as prescribed under ASC 815-40.
Warrants classified as equity are initially measured at fair value, and subsequent changes in fair value are not recognized so long as
the warrants continue to be classified as equity. The value of the warrant is based on accepted valuation procedures and practices that
rely substantially on the third-party professional’s use of numerous assumptions and its consideration of various factors that
are relevant to the operation of the Company.
JOBS
Act and Recent Accounting Pronouncements
The
JOBS Act provides that an “emerging growth company” can take advantage of the extended transition period provided in Section
7(a)(2)(B) of the Securities Act, for complying with new or revised accounting standards. In other words, an “emerging growth company”
can delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have elected
to take advantage of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act, for complying with new or revised
accounting standards that have different effective dates for public and private companies until the earlier of the date we (i) are no
longer an emerging growth company or (ii) affirmatively and irrevocably opt out of the extended transition period provided in Section
7(a)(2)(B) of the Securities Act.
We
have implemented all new accounting pronouncements that are in effect and may impact our financial statements and we do not believe that
there are any other new accounting pronouncements that have been issued that might have a material impact on our financial position or
results of operations.
Recently
Issued Accounting Pronouncements
From
time to time, new accounting pronouncements are issued by the Financial Accounting Standards Board (“FASB”) that are adopted
by the Company as of the specified effective date. If not discussed, management believes that the impact of recently issued standards,
which are not yet effective, will not have a material impact on the Company’s financial statements upon adoption.
In
August 2020, the FASB issued Accounting Standards Update (“ ASU”) 2020-06, “Debt - Debt with Conversion and Other
Options (Subtopic 470-20) and Derivatives and Hedging - Contracts in Entity’s Own Equity (Subtopic 815 - 40)” (“ASU
2020-06”). ASU 2020-06 simplifies the accounting for certain financial instruments with characteristics of liabilities and equity,
including convertible instruments and contracts on an entity’s own equity. The ASU is part of the FASB’s simplification initiative,
which aims to reduce unnecessary complexity in U.S. GAAP. The ASU’s amendments are effective for fiscal years beginning after December
15, 2023, and interim periods within those fiscal years. The Company is currently evaluating the impact of ASU 2020-06 on its financial
statements.
Business
Overview
We
connect consumers to licensed healthcare professionals through our website at www.MangoRX.com, for the provision of care via telehealth
on our customer portal. We also provide access for customers to a licensed pharmacy for online fulfillment and distribution of certain
medications that may be prescribed as part of telehealth consultations, including our Mango ED and Mango GROW products.
We
have identified men’s wellness telemedicine services and products as a growing sector in recent years and especially related to
the areas of erectile dysfunction (“ED”) and hair growth products.
Mango
ED
We
have developed, and are commercially marketing and selling, a new brand of ED product under the brand name “Mango.” This
product is produced at a compounding pharmacy and is available to patients on the determination of a prescribing physician that the compounded
drug is necessary for the individual patient. This product currently includes the following three ingredients: either Tadalafil (the
active ingredient in Cialis) or Sildenafil (the active ingredient in Viagra) and Oxytocin, all of which are used in FDA approved drugs,
as well as L-Arginine, an amino acid that is available as a dietary supplement. However, the fact that Tadalafil and Oxytocin are used
in FDA approved drugs, and L-arginine is available as a dietary supplement, does not mean that these ingredients will prove safe when
combined into a single formulation to treat ED. We currently offer two dosage levels of our Mango ED product and anticipate doctors prescribing
a dosage based on the needs and medical history of the patient. Our Mango ED product currently includes the following amounts of the
three ingredients: (1) either Sildenafil (50 milligrams (mg)) or Tadalafil (10 (mg)), Oxytocin (100 International units (IU)) and L-Arginine
(50mg); and (2) either Sildenafil (100 milligrams (mg)) or Tadalafil (20mg), Oxytocin (100IU) and L-Arginine (50mg). Our Mango ED product
has not been, and will not be, approved by the FDA and instead we produce and sell our products, including our Mango ED product, under
an exemption provided by Section 503A of the FFDCA Act, as discussed below. Additionally, because our Mango ED product is being specially
compounded for the customer by a pharmacist with a physician’s prescription and because the ingredients for our Mango ED product
are publicly disclosed, this product formula can be replicated by other companies.
We
are not aware of any clinical studies involving (i) administration of Tadalafil or Sildenafil sublingually at the doses we provide patients,
or (ii) compounding of Tadalafil or Sildenafil, Oxytocin, and L-arginine to treat ED, similar to our Mango ED products. We are, however,
aware of other companies that are currently selling oral disintegrating tablets for ED, including those using a combination of Tadalafil
(the active ingredient in Cialis) and Sildenafil (the active ingredient in Viagra). We believe that the potential safety risks associated
with our Mango ED products are comparable to the safety risks associated with oral formulations of Tadalafil and Sildenafil approved
by the FDA for the treatment of ED. We do not expect significant safety risks associated with L-arginine, as the FDA has recognized in
its regulations that L-arginine may be safely added as a nutrient to foods. Clinical studies of intranasal Oxytocin have also found that
Oxytocin is generally safe and well-tolerated. Notwithstanding the above, because our ED product has not been, and will not be, approved
by the FDA, our product has not had the benefit of the FDA’s clinical trial protocol which seeks to prevent the possibility of
serious patient injury and death. If this were to occur, we could be subject to litigation and governmental action, which could result
in costly litigation, significant fines, judgments or penalties.
Launch
of Mango Hair Growth Product - ‘GROW’ by MangoRx
We
have developed, since November 16, 2022 are marketing, and selling, a new brand of hair growth product under the brand name ‘GROW’
by MangoRx (“Mango GROW”). This product is produced at our related party compounding pharmacy and is available to patients
on the determination of a prescribing physician that the compounded drug is necessary for the individual patient. Mango GROW currently
includes the following four ingredients - (1) Minoxidil (the active ingredient in Rogaine®) and (2) Finasteride (the active ingredient
in Propecia), each of which is used in FDA approved drugs, as well as (3) Vitamin D3 and (4) Biotin, which are available as dietary supplements.
However, the fact that Minoxidil and Finasteride are used in FDA approved drugs, and that Vitamin D3 and Biotin, are available as a dietary
supplement, does not mean that these ingredients will prove safe when combined into a single formulation to attempt to treat hair growth.
Mango GROW is encapsulated in convenient chewable, mint-flavored RDT’s.
We
currently offer one dosage level of our Mango GROW product and anticipate doctors prescribing Mango GROW based on the needs and medical
history of the patient. Our Mango GROW product currently includes the following amounts of the four ingredients discussed above: (1)
Minoxidil (2.5mg), (2) Finasteride (1mg), (3) Vitamin D3 (2000IU), and (4) Biotin (1mg). Our Mango GROW product has not been, and will
not be, approved by the FDA and instead we produce and sell our Mango GROW product and plan to produce and sell future pharmaceutical
products, under an exemption provided by Section 503A of the Federal Food, Drug, and Cosmetic Act.
We
are not aware of any clinical studies involving the administration of Minoxidil and Finasteride sublingually at the dose we provide patients,
or the compounding of Minoxidil, Finasteride, Vitamin D3, and Biotin, to treat hair growth, as is contemplated by our Mango GROW product.
We are, however, aware of other companies that are currently selling oral tablets for hair growth, including those using a combination
of Minoxidil and Finasteride. Additionally, because our Mango GROW product is being specially compounded for the customer by a pharmacist
with a physician’s prescription and because the ingredients for our Mango GROW product are publicly disclosed, this product formula
can be replicated by other companies.
Additional
Information Regarding Mango ED and Mango GROW
Because
our Mango ED and Mango GROW products have not been, and will not be, approved by the FDA, our products have not had the benefit of the
FDA’s clinical trial protocol which seeks to prevent the possibility of serious patient injury and death. If this were to occur,
we could be subject to litigation and governmental action, which could result in costly litigation, significant fines, judgments or penalties.
We
currently anticipate using a substantial portion of the net proceeds of this offering to finance marketing and general operational expenses
associated with the sale of our Mango ED and Mango GROW products. We launched our website in mid-November 2022. To date, we have sold
only a small amount of products and generated only minimal revenues.
Mango
ED and Mango GROW have been formulated as RDT using a sublingual (applied under the tongue) delivery system to bypass the stomach and
liver. It is a generally established principle that sublingual drug absorption through the oral mucosa is generally faster than drug
absorption through the gastrointestinal tract. This is because sublingual drugs that are absorbed through the oral mucosa directly enter
the systemic circulation, bypassing the gastrointestinal tract and first-pass metabolism in the liver (see H. Zhang et al., Oral mucosal
drug delivery: clinical pharmacokinetics and therapeutic applications, 41 Clin Pharmacokinet
661, 662 (2002). Though the active ingredients that comprise our Mango ED product are meant to treat ED - an issue that according
to a 2018 study published in The Journal of Sexual Medicine has been estimated to affect over one-third of today’s men’s
population (with prevalence increasing with age) - we are also aiming to brand ourselves as a lifestyle company marketed to men seeking
enhanced sexual vitality, performance, and overall mood and confidence, together with our Mango GROW product.
Our
Mango products are sold exclusively online via our website at www.MangoRX.com.
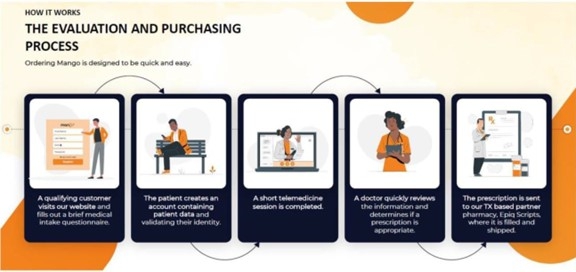
Our
Contracted Telehealth Provider
In
many states, including Texas where our principal business office is located, the corporate practice of medicine doctrine prohibits corporations
from practicing medicine and from employing physicians to provide professional medical services. Many states that recognize this doctrine
also prohibit physicians from agreeing to share the fees they receive for professional services with unlicensed entities or individuals,
a practice that is commonly known as “fee splitting.” The requirements for compliance with any applicable corporate practice
of medicine and fee splitting restrictions vary among the states. In Texas, for example, there is no statute that expressly prohibits
fee splitting, but the corporate practice of medicine doctrine has been interpreted to prohibit physicians from ceding control over their
fee structures to corporate entities or giving a substantial portion of the fees received to corporate entities.
In
order to comply with corporate practice of medicine and fee splitting restrictions, we do not employ or directly contract with individual
physicians or physician groups, nor do we control their medical decision-making or charges. Rather, on August 1, 2022, we entered into
a Physician Services Agreement (the “Physicians Agreement”) with BrighterMD, LLC doing business as Doctegrity (“Doctegrity”),
as discussed in further detail below under, which has agreed to make available to us, healthcare professionals, to allow them to provide
clinical services directly to our future customers via telehealth. We have integrated these healthcare professionals to allow for telehealth
consultations and related services on our Mangoceuticals platform which has been developed and is complete. This platform is the backbone
of our business as it connects consumers with both the medical provider and the pharmacy for fulfillment. It is also the system that
we will use to create marketing funnels for outgoing marketing, customer management and support, and analytics for future sales.
Through
our Physician Services Agreement with Doctegrity (as defined below), the healthcare professionals are responsible for the practice of
medicine and control of the clinical decision-making.
Our
Related Party Pharmacy
As
discussed in greater detail below under “-Material Agreements-Master Services Agreement with Epiq Scripts” and “-First
Amendment to MSA,” we have entered into an exclusive Master Services Agreement and statement of work with Epiq Scripts, LLC (“Epiq
Scripts”), for its specialty compounding and packaging capabilities, fulfillment, and distribution of certain prescription products
available through our platform. These prescription products include our Mango ED and Mango GROW products. Epiq Scripts is a related party
because it was 51%-owned by American International at the time of our entry into the Master Services Agreement and is currently 51% owned
by Mr. Cohen, our Chairman and Chief Executive Officer. Mr. Cohen, our Chairman and Chief Executive Officer, also served as the Chief
Executive Officer and a director of, and had voting control over, American International at the time of the entry into the Master Services
Agreement. As discussed under “Company Information and Formation,” our company was wholly-owned by American International
until April 16, 2022, when control of our company was sold to Cohen Enterprises, which is owned by Mr. Cohen. Epiq Scripts is a relatively
newly formed entity, having been formed in January 2022, and only began compounding drugs for patients in November 2022. On February
15, 2023, the 51% of Epiq Scripts then owned by American International was transferred to Mr. Cohen as part of an exchange transaction,
whereby Mr. Cohen agreed to cancel his preferred stock of American International, which provided him voting control over American International,
in exchange for among other assets, American International’s ownership of Epiq Scripts. As a result, Epiq Scripts is currently
51% owned by Mr. Cohen, our Chairman and Chief Executive Officer. Additionally, Mr. Cohen has served as the co-Manager of Epiq Scripts
since January 2022.
Epiq
Scripts is currently fully licensed with the Texas State Board of Pharmacy (“TSBP”) and further has State Board of Pharmacy
(or its equivalent) licenses from the District of Columbia and 46 other states: Alaska, Arizona, Arkansas, Colorado, Connecticut, Delaware,
Florida, Georgia, Hawaii, Idaho, Illinois, Indiana, Iowa, Kansas, Kentucky, Louisiana, Maine, Maryland, Massachusetts, Michigan, Minnesota,
Mississippi, Missouri, Montana, Nebraska, Nevada, New Hampshire, New Jersey, New Mexico, New York, North Carolina, North Dakota, Ohio,
Oklahoma, Oregon, Pennsylvania, Rhode Island, South Dakota, Tennessee, Utah, Vermont, Virginia, Washington, West Virginia, Wisconsin,
and Wyoming, with the intent of obtaining the remaining 3 state licenses by the end of 2023. Epiq Scripts has obtained its National Provider
Identifier (“NPI”) number and is now a member of the National Council for Prescription Drug Programs (“NCPDP”),
a standards development organization. Additionally, Epiq Scripts has applied for the highest level of accreditation with the Utilization
Review Accreditation Commission (“URAC”), a Washington DC-based healthcare accrediting organization that establishes quality
standards for the entire healthcare industry. Until Epiq Scripts receives licenses in the other three states, we are limited to selling
our Mango ED and Mango GROW products in only the states in which Epiq Scripts holds licenses. Although Epiq Scripts is physically located
in Texas, it can ship products to customers in each state in which it holds licenses.
As
a result of the above, Epiq Scripts can currently only provide services to the Company in the District of Columbia and those 47 states
described above and the Company will be unable to sell its products to any customers in any states other than those listed above, until
Epiq Scripts is able to obtain licenses in other states and will thereafter be limited to selling products to customers only in the states
in which Epiq Scripts holds licenses.
Our
Customer Portal
Our
customer platform connects consumers to licensed healthcare professionals through our website at www.MangoRX.com, for the provision
of care via telehealth and also provides access for customers to a licensed pharmacy for online fulfillment and distribution of certain
medications that may be prescribed as part of telehealth consultations. Additional features to this backend technology solution allow
for the creation and management of customer accounts whereby customers have the ability to login, view and make changes to their respective
accounts. These changes include, but are not limited to, reviewing order history, tracking order shipments, requesting and ordering product
refills and making other profile changes such as shipping address and payment changes. Our portal is not unique to the industry and is
not anticipated to be difficult or costly to replicate or replace.
The
backend technology solution also houses and manages all customer data allowing the Company with additional key functionality, including
but not limited to, providing customer service and support and data analytics for various marketing initiatives and reporting functions.
We
do not anticipate selling any third-party products via our portal.
Our
Growth Strategy
Utilize
a variety of marketing channels using data analytics to attract customers
We
currently market and advertise our Mango ED and Mango GROW products on a variety of advertising mediums including, but not limited to,
social media, online search websites, podcasts, television, radio, out-of-home, and other media channels, in compliance with applicable
FDA rules and requirements. However, due to such rules and requirements, we are extremely limited in the content of the claims and promotional
statements that we are able to make regarding our products under applicable FDA regulations. We believe advertising in a diversified
set of media channels is important to prevent overreliance on any single channel and to maximize the exposure of our brand to our desired
customers. We also intend to reach customers through our own social media accounts, press coverage and public relations, internally developed
educational and lifestyle content, and through engagement of social media influencers, hired and paid celebrities and talent, and physical
brand advertising campaigns, in each case funding permitting, and in each case subject to applicable rules and regulations, which are
expected to significantly limit the content of such marketing materials. We believe that this overall strategy will drive significant
customer traffic to our platform, including direct type-in traffic and organic online search traffic.
We
also intend to utilize a marketing strategy focused on analytics and data. We are designing our internal systems to measure consumer
behavior, including which types of consumers generate more revenue in their first purchase, generate more revenue over time, generate
more gross profit from their purchases, and which types of consumers are most valuable over their lifetime. We also plan on measuring
the effectiveness of our marketing budgets and the rate of return we generate from our marketing campaigns. We have retained and plan
on using an outside marketing and advertising firm to assist management in identifying marketing and advertising campaigns, media purchases
and mediums, and seeking to drive a sufficient rate of return from our marketing and advertising budgets.
Invest
in our telemedicine platform to enable sales throughout the United States
We
utilize both a synchronous and asynchronous approach through our telemedicine platform, connecting customers through our platform and
contracted physicians and pharmacy. An asynchronous visit allows a physician to verify the patient’s identity, demographics and
collect the medical history online without needing to physically see or speak to the patient. A synchronous visit requires the doctor
to either speak directly to the patient and/or see the patient either via video conference or in person. As discussed above, we initially
are focusing our sales in the District of Columbia and 47 states where our related party pharmacy is licensed, with the goal of eventually
undertaking sales across all 50 states, pending licensing approvals of our related party pharmacy.
Provide
subscription plans for recurring revenue and introduction of new products
We
provide our customers with an option to purchase our Mango ED and Mango GROW products on a subscription basis. Subscription plans provide
an easy and convenient way for customers to get ongoing treatment while simultaneously providing the Company with predictability through
a recurring revenue stream.
For
subscription plans, customers are able to select a desired timeframe in which to receive products, which range from once every month
to once every six months. The customer will then be billed on a recurring basis based on the selected timeframe and specified quantity
of product, which is shipped after each billing from our contracted pharmacy (Epiq Scripts). Customers are able to cancel subscriptions
in between billing periods to stop receiving additional products and reactivate subscriptions. Our integrated technology platform serves
customers from customer discovery, through the purchase of products on our website, to connecting customers with medical providers for
telehealth consultations (through our contracted physician network), to the fulfillment and delivery of orders (through our contracted
pharmacy), and finally through ongoing management by medical providers (also through our contracted physician network). We believe our
platform provides us cost advantages and efficiencies to offer customers affordable prices and generate increased revenues over time.
In
addition to our Mango ED and Mango GROW products, we intend to launch new products over time and offer additional subscription-based
offerings which we hope will result in growth in revenue through recurring revenue streams.
Market
Overview
The
Market for ED Products
According
to a January 2022 report published by Verified Market Research, the Global Erectile Dysfunction Drugs Market size was valued at $3.63
billion in 2020, mainly due to the increase in patient awareness and the early adoption of sedentary lifestyle. Verified Market Research
also projects that the total Global Dysfunction Drugs Market size will contract to $2.95 billion in 2028. The expected reason for this
contraction is poor patient compliance with erectile dysfunction drugs and the future availability of cost-effective imitation medicines,
as well as side effects of ED drugs. We do not anticipate our Mango ED drug suffering from these limitations, as we believe our product
is easy to use and that we have priced our product competitively. Separately, Grand View Research, in a July 2022 report, projects that
the U.S. market (where we are initially marketing our ED product) for erectile dysfunction drugs estimated at approximately $1.1 billion
as of 2021, will increase at a 7.4% compound annual growth rate though 2030.
It
is also estimated that nearly 3-in-5 men in the US have suffered from erectile dysfunction, according to a survey reported in February
2022, by LetsGetChecked, a leading at-home health screening and insights company (based on research carried out by Opinium Research among
2,006 men in the USA, 1,178 of whom had previously experienced erectile dysfunction, from February 7-10, 2020). According to that study,
age isn’t that big a factor either, with 56% of men 18 to 34 years old being affected, compared to 63% of those over the age of
55. The study also determined that most men blame psychological factors for ED - with 41% blaming stress, 34% blaming having “too
much on their mind,” and 31% believing it is performance anxiety.
The
Market for Mango GROW
According
to the website of the American Hair Loss Association, (a) two-thirds of American men will experience some degree of hair loss by the
age of 35, (b) by age 50, around 85 percent of men have significantly thinning hair; and (c) for around 25% of men, the start of male
pattern baldness can begin before the age of 21. Additionally, and contrary to societal belief, we believe that most men who suffer from
male pattern baldness are unhappy with their situation and would take steps to change that. In our experience, hair loss affects every
aspect of the hair loss sufferer’s life including interpersonal relationships as well as the professional lives of those suffering.
According
to a May 2022 market study entitled, “Hair Loss Prevention Products Market Forecast to 2028 - COVID-19 Impact and Global Analysis
- by Product Type (Shampoos and Conditioners, Oils, Serums, and Others), Category (Natural & Organic, and Conventional), End User
(Men, Women, and Unisex), and Distribution Channel (Supermarkets and Hypermarkets, Convenience Stores, Online Retail, and Others)”,
by The Insight Partners, the hair loss prevention products market size was valued at $23.6 billion in 2021 and is projected to reach
$31.5 billion by 2028, growing at a projected compound annual growth rate of 4.2% from 2021 to 2028.
Mordor
Intelligence LLP believes that the major factors driving the hair loss prevention market are changing lifestyle patterns, adoption of
a hectic schedule that increases stress levels, which in turn results in frequent hair loss at an earlier stage among the young population,
growing disposable income, and increased emphasis on appearances.
Competition
and Competitive Advantages
We
mainly compete with other companies offering men’s wellness products, including Hims & Hers Health, Inc. and Roman, and with
our Mango ED products, we compete against much larger pharmaceutical companies who offer ED branded drugs like Viagra (Pfizer) and Cialis
(marketed by Lilly ICOS LLC, a joint venture between Eli Lilly and Company and ICOS Corporation) and their generic forms. With our Mango
GROW product, we compete against the much larger pharmaceutical company Merck & Co., which offers the branded hair loss product Propecia,
and Johnson & Johnson, the owner of Rogaine® - a branded form of Minoxidil. These companies have much greater resources than
we do and well-known brand names.
Our
future men’s wellness products will also likely need to compete against other traditional healthcare providers, pharmacies, and
large retailers that sell non-prescription products.
Furthermore,
we compete with other companies, which have greater resources and a greater advertising budget, and which are also selling ED related
products with either or both Tadalafil and Sildenafil (or similar products), in an oral disintegrating tablet and who are selling compounded
Minoxidil and Finasteride in both topical form (e.g., gels, foams, liquid solutions) and in oral capsule, tablet or pill form. For example,
we are aware of other companies that are currently selling oral disintegrating tablets for ED, including those using a combination of
Tadalafil and Sildenafil (the active ingredient in Viagra). However, we are not aware of any companies that are selling a compound consisting
of Minoxidil and Finasteride in an oral disintegrating tablet form.
We
compete against these competitors based on our branding, advertising, unique compounding, and product delivery system (i.e., our Mango
ED and Mango GROW products have been designed to be taken sublingually, rather than in pill form).
Relative
to other online direct to consumer telemedicine companies that are selling both generic ED medication and generic hair loss medications,
we believe we have priced both our Mango ED products and Mango GROW product at a premium, due to the cost of compounding the product
and the use of multiple ingredients. We are currently aware of a handful of other direct to consumer companies that are also selling
compounded hair loss and ED medications and who are selling their products at a higher price than Mango’s current price. When comparing
the current market for various pharmaceutical related hair loss and ED products, we have attempted to position our pricing to be slightly
above average as we anticipate marketing our Mango ED and Mango GROW products to a demographic that we expect will pay a premium for
what we believe to be a premium product relative to the competition for the treatment of hair loss and erectile dysfunction.
Regulatory
Environment
We
currently produce and sell our Mango ED and Mango GROW products, and plan to produce and sell future pharmaceutical products, under an
exemption provided by Section 503A of the Federal Food, Drug, and Cosmetic Act (“FFDCA Act”). Section 503A describes the
conditions under which compounded human drug products are exempt from the FFDCA Act sections on FDA approval, prior to marketing, current
good manufacturing practice (“cGMP”) requirements and labeling with adequate directions for use. One of these conditions
is that the drugs must be compounded based on the receipt of valid patient-specific prescriptions; another condition limits “copying”
of FDA-approved products, which restricts compounding drugs that have the same active ingredients and route of administration as ingredients
that are used in other FDA approved drugs which are commercially available. The FDA also prohibits any marketing or promotional statements
that are “false or misleading in any particular,” including making any unsupported superiority claims against other products
or the failure to disclose a material fact.
Notwithstanding
the above, under relevant FDA guidance, the FDA generally does not consider a compounded drug to be “essentially a copy”
of a commercially available drug if the compounded drug has a different route of administration as compared with the approved alternative,
and our Mango ED and Mango GROW products are for a different route of administration (e.g., sublingual). In addition, the FDA does not
consider a compounded drug to be “essentially a copy” of a commercially available drug if the approved product cannot be
used for the prescribed route of administration, which is available in the compounded version (which we believe it cannot, as discussed
below). Finally, we do not expect that we will be deemed to have engaged in such “copying”, because our Mango ED and Mango
GROW products are based on a prescriber’s determination for each patient that the change associated with the compounded product
(our Mango ED and Mango GROW products) produces for the patient a significant difference as compared with the commercially available
drug product. Under relevant FDA guidance, the FDA does not consider a compounded drug “essentially a copy” if a prescriber
determines that there is a change, made for an identified individual patient, which produces for that patient a significant difference
from the commercially available product.
Under
Section 503A of the FFDCA Act, it is the prescribing practitioner who determines if a compounded drug is necessary for the identified
patient and whether the change associated with the compounded product produces for the patient a significant difference as compared with
the commercially available drug product. FDA’s guidance states that FDA generally does not intend to question prescriber determinations
that are appropriately documented. Our Mango ED and Mango GROW compounded products have been formulated as a Rapid Dissolve Tablet using
a sublingual (applied under the tongue) delivery system to bypass the stomach and liver. We believe this offers a significant difference
based on the fact that the approved versions are not available in the same route of administration (i.e., sublingual). A sublingual formulation
may be able to meet the clinical needs of a particular patient who desires a more rapid onset of action compared to an FDA-approved oral
formulation. In addition, because the prevalence of ED generally increases with age, older patients who may have difficulty swallowing
an FDA-approved oral formulation may benefit from a sublingual formulation that dissolves under the tongue.
Compounded
drugs, like our Mango ED and Mango GROW products, are not FDA-approved. This means that the FDA does not verify the safety or effectiveness
of such drugs. Instead, consumers rely on the determination of a prescribing physician that the compounded drug is necessary for the
individual patient. Compounded drugs also lack an FDA finding of manufacturing quality before such drugs are marketed.
The
FDA has the authority to impose significant restrictions on products through regulations on advertising, promotional and distribution
activities. In particular, the FDA will object to any promotional activity (including through testimonials and surrogates) that is “false
or misleading in any particular,” including the failure to disclose material facts. For example, the FDA will expect adequate substantiation
for an efficacy claim, which would require substantial evidence derived from adequate and well-controlled clinical trials. We believe
we can conduct truthful and non-misleading promotional activities, including activities involving the use of testimonials and surrogates,
with limited claims that do not require substantial evidence derived from adequate and well-controlled clinical trials and which do not
include efficacy claims.
We
are also aware of data in the scientific literature supporting how the proposed combination of the compounds which make up our Mango
ED products (i.e., Tadalafil or Sildenafil, Oxytocin, and L-arginine) might be expected to perform in ED patients. Previous clinical
studies (none of which we have paid for or undertaken ourselves) have suggested that either Sildenafil Tadalafil and L-arginine in combination
for treatment of ED may be more effective than either compound alone (see L. Gallo et al., The Daily Therapy With L-Arginine 2,500
mg and Tadalafil 5 mg in Combination and in Monotherapy for the Treatment of Erectile Dysfunction: A Prospective, Randomized Multicentre
Study, 8 Sex Med 178, 184 (June 2020) - finding that in general, combination therapy
with Tadalafil and L-Arginine was superior to monotherapies for the treatment of ED; and M. Abu El-Hamd & E. Mohammed Hegazy, Comparison
of the clinical efficacy of daily use of L-arginine, tadalafil and combined L-arginine with tadalafil in the treatment of elderly patients
with erectile dysfunction, 52 Andrologia e13640, 3 (Aug. 2020) (“Hamd and Hegazy”)
- finding that the combined daily use of L-arginine with Tadalafil therapy for elderly male patients with ED could significantly increase
Sexual Health Inventory for Men (SHIM) scores and levels of total testosterone in comparison to L-arginine, or Tadalafil alone)-This
is because L-arginine may increase nitric oxide, that in turn may increase cyclic guanosine monophosphate, which has relaxation and vasodilation
(dilatation of blood vessels) effects on smooth muscle to assist in the treatment of ED (see Hamd and Hegazy paper). Furthermore, Oxytocin
is a neurotransmitter linked to increased levels of social interaction, well-being, and anti-stress effects and clinical studies suggest
administration of Oxytocin may stimulate certain aspects of social interaction, and may cause anti-anxiety and anti-stress effects (see
Hamd and Hegazy paper).
Furthermore,
we are aware of data in the scientific literature supporting the efficacy of Minoxidil as an oral treatment (as discussed below), as
opposed to topical treatments that have been more traditionally used and marketed for hair growth to date. Topical Minoxidil and oral
Finasteride are current the standard first-line treatments for androgenetic alopecia (AGA)(male pattern baldness). Minoxidil in an oral
formulation has been previously used for the treatment of severe and uncontrolled hypertension at a dose of 10-40 mg. Unintentionally,
the early trials of oral minoxidil as an antihypertensive drug documented side effects such as hypertrichosis (excessive hair growth
anywhere on the body) and hirsutism (excess hair most often noticeable around the mouth and chin) with chronic use. A study conducted
by Ratchathorn Panchaprateep & Suparuj Lueangarun, and published in the September 24, 2020 edition of Dermatology and Therapy, found
that oral minoxidil at a dose of 5 mg taken once daily, significantly increased hair growth in men with AGA after 12 and 24 weeks of
treatment (Panchaprateep, R., Lueangarun, S. Efficacy and Safety of Oral Minoxidil 5 mg Once Daily in the Treatment of Male Patients
with Androgenetic Alopecia: An Open-Label and Global Photographic Assessment. Dermatol Ther (Heidelb) 10, 1345-1357 (2020)).
Separately,
Finasteride taken orally in the amount of 1 mg per day has shown to promote scalp hair growth and prevent further hair loss in a significant
proportion of men with male pattern hair loss (McClellan, K.J., Markham, A. Finasteride. Drugs 57, 111-126 (1999).
Neither
we, nor our representatives have had any conversations with the FDA staff regarding whether our Mango ED and Mango GROW products can
be sold pursuant to Section 503A of the FFDCA Act and future conversations with the FDA may result in the FDA staff raising issues with
such sales pursuant to Section 503A of the FFDCA, requiring certain pre-requisites or changes to our current business plan, which may
be costly or time consuming, and/or may result in us being prohibited from selling our Mango ED and Mango GROW products pursuant to Section
503A of the FFDCA Act.
Government
Regulation
We,
as are many other companies, are also subject to environmental laws, rules and regulations which could affect our operations, including
those disclosed below.
Physicians
who provide professional clinical services via telehealth must, in most instances, hold a valid license to provide the applicable professional
services in the state in which the patient is located. As such, the physicians provided to us through our relationship with BrighterMD,
LLC dba Doctegrity, discussed below under “Business-Material Agreements-Master Services Agreement with Epiq Scripts” and
“-First Amendment to MSA,” are required to be licensed under applicable state law.
To
qualify for the exemptions under section 503A of the FFDCA Act, among other requirements, a drug must be compounded by a licensed pharmacist
or a licensed physician that does not compound regularly or in inordinate amounts any drug products that are essentially copies of a
commercially available drug product. As discussed below under “Business-Material Agreements-Master Services Agreement with Epiq
Scripts” and “-First Amendment to MSA,” we have entered into an agreement with Epiq Scripts, a related party, 51% owned
and controlled by Jacob D. Cohen, our Chairman and Chief Executive Officer, to provide us compounding and other pharmacy services.
Our
operations are subject to extensive government regulation, from the entry into agreements with physicians or groups of physicians to
provide telehealth services to our potential customers, to the marketing and promotion of our products, the creation of our products,
and the sale of our products through licensed pharmacists.
Some
of the rules and regulations we expect to be subject to include:
Federal
Anti-Kickback Statute
The
Federal Anti-Kickback Statute (42 U.S.C. § 1320a-7b) is broadly worded and prohibits the knowing and willful offer, payment, solicitation
or receipt of any form of remuneration in return for, or to induce, (i) the referral of a person covered by Medicare, Medicaid or other
governmental programs, (ii) the furnishing or arranging for the furnishing of items or services reimbursable under Medicare, Medicaid
or other governmental programs or (iii) the purchasing, leasing or ordering or arranging or recommending purchasing, leasing or ordering
of any item or service reimbursable under Medicare, Medicaid or other governmental programs. In addition, a person or entity does not
need to have actual knowledge of this statute or specific intent to violate it to have committed a violation. Moreover, the government
may assert that a claim including items or services resulting from a violation of the Anti-Kickback Statute constitutes a false or fraudulent
claim for purposes of the False Claims Act, we do not expect to apply to our operations as we do not plan to seek payment for our services
from the federal government. Violations of the Anti-Kickback Statute can result in exclusion from Medicare, Medicaid or other governmental
programs as well as civil and criminal penalties and fines. Imposition of any of these remedies could have a material adverse effect
on our business, financial condition and results of operations.
We
do not anticipate our current operations being subject to the Anti-Kickback Statute as we do not seek reimbursement under a federal health
care program.
U.S.
Food and Drug Administration (“FDA”) Regulation
The
FDA regulates product promotion and noncompliance and this could result in the FDA requesting that we modify our product promotion or
subject us to regulatory and/or legal enforcement actions, including the issuance of a warning letter, injunction, seizure, civil fine,
and criminal penalties. Other federal, state or foreign enforcement authorities also monitor product promotion and have the authority
to levy significant fines or penalties under other statutory authorities, such as laws prohibiting false claims for reimbursement, if
violations of applicable law or regulations occur. We also believe that the FDA will likely consider our compounded combination product
to be different from previously FDA-approved products, and that the FDA will not likely allow us to rely on any FDA-approved labeling
or prescribing information.
Risk
of Litigation
Additionally,
federal and state statutes provide for private causes of action to plaintiffs alleging misleading marketing claims, or otherwise making
allegations which are found to be in violation of such laws. As such, misleading promotional statements and practices can lead to litigation
under state consumer protection and unfair trade practices laws. To date, there has been a substantial amount of litigation under these
laws challenging the marketing and sale of compound drugs and we may face legal actions, and be subject to significant penalties, judgments
and damages, if we are found to have violated these laws.
Health
Information Privacy and Security Laws
Numerous
U.S. state and federal laws and regulations govern the collection, dissemination, use, privacy, confidentiality, security, availability,
integrity, and other processing of health information. We believe that, because of our operating processes, we are not a covered entity
or a business associate under the Health Insurance Portability and Accountability Act and the implementing regulations (“HIPAA”),
which establishes a set of national privacy and security standards for the protection of protected health information by health plans,
healthcare clearinghouses, and certain healthcare providers, referred to as covered entities, and the business associates with whom such
covered entities contract for services. Because we need to use and disclose customers’ health and personal information in order
to provide our services, we develop and maintain policies and procedures to protect that information in the future.
In
addition to HIPAA, numerous other federal, state, and foreign laws and regulations protect the confidentiality, privacy, availability,
integrity and security of health information and other types of personal information. These laws and regulations are often uncertain,
contradictory, and subject to changing or differing interpretations. Additionally, these laws may be similar to or even more protective
than, and may not be preempted by, HIPAA and other federal privacy laws. The privacy and data protection laws in many states in which
we operate are more restrictive than HIPAA and/or may apply more broadly than HIPAA. In certain cases, it may be necessary to modify
our operations and procedures to comply with these more stringent state laws. Not only may some of these state laws impose fines and
penalties upon violators, but also some, unlike HIPAA, may afford private rights of action to individuals who believe their personal
information has been misused. We expect new laws, rules and regulations regarding privacy, data protection, and information security
to be proposed and enacted in the future; as state laws are changing rapidly.
For
example, as of the date of this prospectus, eleven states-California, Colorado, Connecticut, Iowa, Indiana, Tennessee, Montana, Texas,
Utah, Virginia and Oregon-have enacted consumer data privacy laws. The data privacy laws have a number of things in common with each
other, including allowing residents of those states the right to access and delete their personal information and to opt-out of the sale
of their personal information, among others. Other provisions require commercial websites or online services to post a privacy policy
that describes the types of personal information collected, what information is shared with third parties, and how consumers can request
changes to certain information. Our compliance with these and future rules may increase our operating and expenses and our failure to
comply with these rules could subject us to fines, penalties and litigation.
In
addition to the above, proposed or new legislation and regulations could also significantly affect our business. There currently are
a number of proposals pending before federal, state, and foreign legislative and regulatory bodies.
Product
Liability
As
a distributor of men’s health and wellness products, the Company faces an inherent risk of exposure to product liability claims,
regulatory action and litigation if its future products are alleged to have caused significant loss or injury. In addition, the sale
of our products involves the risk of injury to consumers due to tampering by unauthorized third parties or product contamination. Previously
unknown adverse reactions resulting from human consumption of our products alone or in combination with other medications or substances
could occur. We may be subject to various product liability claims, including, among others, that our future products caused injury or
illness, include inadequate instructions for use or include inadequate warnings concerning possible side effects or interactions with
other substances. A product liability claim or regulatory action against the Company could result in increased costs, could adversely
affect our reputation with our clients and consumers generally, and could have a material adverse effect on our results of operations
and financial condition of the Company.
For
example, a 2014 study published in The Journal of the American Medical Association determined that Sildenafil (the active ingredient
in Viagra) may be associated with a higher risk of developing melanoma. The study evaluated data from more than 25,000 men who used Sildenafil
and found that Sildenafil use was significantly associated with an increased risk of subsequent melanoma, after considering other risk
factors. It is possible that the ingredients we use in our Mango ED and Mango GROW products or any other products we sell in the future
could be found in the future to result in increases in the likelihood of developing cancer or other diseases, which could subject us
to litigation, penalties or recalls.
Insurance
We
have an insurance policy in effect that includes customary coverage and protection for professional liability, general liability, employee
benefits and protection against claims including technology products, services and against cyber security. Our insurance policy also
covers exposure to product liability claims, including both technology product claims related to customer data breaches, copyright infringement
and/or misrepresentation and fraud and any claims made in connection with any physical products and services sold through the Company’s
website.
Material
Agreements
Physician
Services Agreement with Doctegrity
Pursuant
to the Physicians Agreement, Doctegrity, which provides online telemedicine technology services and provides access to independently
contracted licensed physicians and providers, agreed to (a) arrange for the services of a physician or, where appropriate, a mid-level
practitioner with delegated authority from a physician, licensed in the appropriate state the practice of medicine will take place, who
will establish a physician/patient relationship with patients associated with the Company’s platform in accordance with the laws
and regulations of the appropriate state(s) and also provide physician review and assessment and quality control of the Company’s
or related brands’ advertising of services, medical questionnaires and related prescription requests; and (b) provide an asynchronous
telehealth platform (and in certain cases, synchronous capabilities in certain U.S. states where and when available and applicable) which
provides patient access to licensed physicians in the state from which the patient, who is participating under our platform, resides.
We
chose to contract with Doctegrity after reviewing and comparing the fees and services offered by similar telehealth platform companies
that facilitate visits between health care professionals and patients.
After
a patient visits our website and submits a request for a consultation with a health care professional, Doctegrity will communicate the
patient’s information to one of its affiliated physicians. Doctegrity and the physicians are responsible for conducting the telehealth
consultation and any ongoing communication with the patient in accordance with applicable laws. The physicians make a determination,
in their sole discretion, as to whether or not to prescribe our products (currently our Mango ED and Mango GROW products) to potential
customers. If the physicians prescribe our Mango ED or Mango GROW products, then the customers pay us for our products. In turn, Epiq
Scripts, LLC, pursuant to the Master Services Agreement discussed below, is provided information on the customer and compounding of our
product, compound the product, and ship the product to customers using packaging and shipping materials which we supply.
We
pay Doctegrity for each physician visit conducted in response to request made by a patient on our website, regardless of whether the
physician prescribes our product to the patient. The fee we pay Doctegrity is fixed, set in advance and was negotiated at arms’
length after comparing the prices offered by similar services. We are not a party to any contracts between Doctegrity and any health
professionals or physician groups and do not control how Doctegrity reimburses these providers.
Although
our arrangement with Doctegrity, as summarized above, is structured to comply with applicable laws, including those restricting the corporate
practice of medicine and fee splitting, there may be a risk that a state agency, now or in the future as these laws (and interpretations
of them) evolve, would conclude that the arrangement and fee structure between Doctegrity and its contracted physicians and/or our agreement
with Doctegrity violates the corporate practice of medicine doctrine and fee splitting restrictions in Texas or in another state where
a patient who uses our Mangoceuticals platform is located.
The
Physicians Agreement has a term of one year subject to automatic one-year renewals unless and until terminated in accordance with the
Physicians Agreement, including by either party with 90 days’ prior written notice with or without cause and for cause with ten
days’ written notice.
The
Physicians Agreement requires us and Doctegrity to maintain certain minimum levels of insurance, and contains customary representations
and warranties, force majeure provisions and confidentiality obligations. Pursuant to the Physicians Agreement, each party is required
to indemnify and hold harmless the other party, its affiliates and representatives, from and against any third party claims, liabilities,
damages, judgments or other losses (including reasonable attorneys’ fees) imposed upon or incurred by them arising out of or as
a result of: (i) any acts or omissions by or the willful misconduct of the other party, its affiliates or representatives in connection
with the performance of any of their respective obligations under the agreement; and (ii) any material breach of the agreement by the
other party, or its affiliates or representatives; except to the extent that such losses arising pursuant to (i) and/or (ii), arise from
the bad faith, willful misconduct or gross negligence of the party seeking indemnification. The Physicians Agreement also includes customary
limitation of liability language, whereby each party waived any liability from the other for any indirect, incidental, exemplary, punitive
or consequential damages.
Doctegrity’s
physicians are tasked with determining whether patients seeking Mango ED or Mango GROW products are eligible to be prescribed our Mango
ED and Mango GROW products, respectively, with the sole purpose of the telemedicine engagement being for the determination, in the physician’s
sole judgment, of whether the patient is qualified to obtain a prescription for the Mango ED or Mango GROW products. Doctegrity’s
physicians are required to electronically send prescriptions to Epiq Scripts (the Company’s designated and accredited pharmacy
partner), which financial relationship is required to be disclosed in writing to the patient via the Terms and Conditions listed on the
Company’s website, including informed consent, and also informing the patient that the prescription is sent to the Company’s
designated pharmacy partner. Doctegrity’s physicians are only able to prescribe Mango ED or Mango GROW products to patients seeking
ED medical and/or treatment hair loss , respectively, through our customer portal.
The
agreement also includes certain covenants restricting our operations, restricting us and our owners, directors, officers, and managers,
during the term of the agreement and for 12 months thereafter from providing to or for any customer any services or products, solutions,
of the type provided by Doctegrity, using confidential information received during the term of the agreement.
Master
Services Agreement with Epiq Scripts
On
September 1, 2022, and effective on August 30, 2022, we entered into a Master Services Agreement with Epiq Scripts, which at the time
was 51%-owned by American International. Mr. Cohen, our Chairman and Chief Executive Officer, served as the Chief Executive Officer and
a director of, and had voting control over, American International at the time of the entry into the Master Services Agreement, and currently
serves on the Board of Directors of American International. As discussed above under “-Formation and Transfer of Control,”
the Company was wholly-owned by American International until June 16, 2022, when control of the Company was sold to Cohen Enterprises,
which is owned by Mr. Cohen. Epiq Scripts was formed in January 2022, and only began compounding drugs for patients in November 2022.
On February 15, 2023, the 51% of Epiq Scripts then owned by American International was transferred to Mr. Cohen as part of an exchange
transaction, whereby Mr. Cohen agreed to cancel his preferred stock of American International, which provided him voting control over
American International, in exchange for among other assets, American International’s ownership of Epiq Scripts. As a result, Epiq
Scripts is currently 51% owned by Mr. Cohen, our Chairman and Chief Executive Officer. Additionally, Mr. Cohen has served as the co-Manager
of Epiq Scripts since January 2022.
Pursuant
to the Master Services Agreement and a related statement of work (“SOW”), Epiq Scripts agreed to provide pharmacy and related
services to the Company, the Company agreed to exclusively use Epiq Scripts as the provider of the Services (defined below) during the
term of the agreement, so long as Epiq Scripts complies with the terms of the Master Services Agreement. The agreement also includes
a 30 day right of first refusal for Epiq Scripts to provide pharmacy services for any new product that Mango may introduce during the
term of the agreement.
Pursuant
to the SOW, Epiq Scripts agreed to provide for the online fulfillment, specialty compounding, packaging, shipping, dispensing and distribution
(collectively, the “Services”) of products sold exclusively via our website that may be prescribed as part of a telehealth
consultation on our platform. Epiq Scripts also agreed to provide mail service pharmacy services to us on an exclusive basis during the
term of the SOW.
We
agreed to provide Epiq Scripts with all custom packaging materials, including but not limited to, individual sachet and/or blister packaging
materials, outer box packaging, and any custom inserts and/or marketing information to accompany the prescription shipment, if any and
to provide Epiq Scripts with quarterly sales forecasts to ensure Epiq Scripts has enough packaging materials on hand to cover a 90 day
period. We agreed to pay for all direct shipping, delivery and related courier costs and to provide Epiq Scripts with direct access to
any online accounts to access and generate shipping labels for the fulfillment and delivery of our products.
The
SOW has a term through December 31, 2025, automatically renewable thereafter for successive one-year terms unless either party terminates
the agreement at least 90 days before renewal thereof and the SOW is subject to the same termination rights of the parties as set forth
in the Master Services Agreement (discussed below).
Pursuant
to the SOW, we agreed to pay Epiq Scripts certain fixed rate fees for prescription fulfillment, processing and packaging (per prescription)
and drug compounding (per pill), provided the per pill rate is reduced upon us exceeding 3,500 product packages per month.
Under
the Master Services Agreement, we are solely responsible for billing and collecting funds from our customers and Epiq Scripts is paid
out of funds that we actually collect.
We
paid Epiq Scripts a total of $60,000 upon our entry into the Master Services Agreement, comprising $45,000 as a one-time non-refundable
technology systems setup and implementation fee and $15,000 as an upfront retainer to be credited towards the future provision of pharmacy
and related services as outlined and detailed in the Master Services Agreement and SOW. All costs related to the pharmacy services provided
by Epiq Scripts are listed as related party costs of revenues on our statement of operations.
The
Master Services Agreement has a term of five years, automatically renewable to additional one-year terms thereafter unless either party
provides the other notice of termination at least 90 days prior to the date of automatic renewal. The Master Services Agreement can be
terminated (i) upon breach of the agreement by the other party, subject to a 90-day cure right, (ii) if a party enters into bankruptcy
or fails to pay its debts as they become due, or (iii) if Epiq Scripts becomes unable to perform the services covered by the Master Services
Agreement and any statements of work associated therewith.
Payments
under the Master Services Agreement are due within 15 days after the end of each month during which collections are received. The Master
Services Agreement contains customary confidentiality obligations, record retention provisions, audit rights, and representations and
warranties of the parties. Each party to the Master Services Agreement agreed to indemnify, defend, and hold harmless the other and the
other party’s officers, directors, shareholders, employees, and agents from and against any and all nonparty claims, or actions
for damages, liabilities (including strict liability), penalties, costs and expenses (including reasonable legal fees, expenses and costs)
to the proportionate extent caused by (1) the negligence or willful misconduct of the indemnitor or any of its employees or agents in
connection with the performance of the agreement, or (2) any breach of any representation, warranty or covenant under the agreement by
the indemnitor or any of its employees or agents. Additionally, the parties agreed that neither party will be liable to the other for
special, incidental, or exemplary damages, subject to certain limited exceptions. The Master Services Agreement does not address product
liability claims or assign any rights of indemnification or contribution in connection therewith.
We
paid Epiq Scripts a total of $60,000 upon our entry into the Master Services Agreement, comprising $45,000 as a one-time non-refundable
technology systems setup and implementation fee and $15,000 as an upfront retainer to be credited towards the future provision of pharmacy
and related services as outlined and detailed in the Master Services Agreement and SOW, of which $11,745 remained outstanding as of December
31, 2022 and $84,382 remained outstanding as of September 30, 2023. All costs related to the pharmacy services provided by Epiq Scripts
are listed as related party costs of revenues on our statement of operations.
Epiq
Scripts has filed with the Utilization Review Accreditation Commission (“URAC”) to obtain its pharmacy accreditation and
obtained its first state license in the State of Texas in February 2022. Epiq Scripts has State Board of Pharmacy (or its equivalent)
licenses to operate in 47 states: Alaska, Arizona, Arkansas, Colorado, Connecticut, Delaware, Florida, Georgia, Hawaii, Idaho, Illinois,
Indiana, Iowa, Kansas, Kentucky, Louisiana, Maine, Maryland, Massachusetts, Michigan, Minnesota, Mississippi, Missouri, Montana, Nebraska,
Nevada, New Hampshire, New Jersey, New Mexico, New York, North Carolina, North Dakota, Ohio, Oklahoma, Oregon, Pennsylvania, Rhode Island,
South Dakota, Tennessee, Texas, Utah, Vermont, Virginia, Washington, West Virginia, Wisconsin, and Wyoming and plans to eventually obtain
licenses in all 50 states by the end of 2023, with some state licenses easier to obtain and quicker to obtain than others.
As
a result of the above, Epiq Scripts can currently only provide the Services to the Company in the District of Columbia and 47 states
described above, and the Company will be unable to sell its products to any customers in any states other than those named above, until
Epiq Scripts is able to obtain licenses in other states and will thereafter be limited to selling products to customers only in the states
in which Epiq Scripts holds a license.
Consulting
Agreement With Epiq Scripts
On
September 15, 2023, we entered into a Consulting Agreement (the “Consulting Agreement”) with Epiq Scripts. Pursuant to the
Consulting Agreement, Epiq Scripts agreed to provide pharmacy consulting services in connection with the Company’s global expansion
efforts, and as reasonably requested by the Company, during the term of the agreement, which is for five years, unless otherwise earlier
terminated (a) due to breach of the agreement by either party and the failure to cure such breach 30 days after written notice thereof;
(b) the mutual agreement of the parties; or (c) the date that Epiq Scripts provides the Company written notice of termination, which
may be at any time and for any reason.
In
consideration for agreeing to provide the services under the agreement, the Company agreed to pay Epiq Scripts (1) a one-time payment
of $65,000, payable within ten days of the entry into the agreement; and (2) a set fee, payable for each prescription drug pill sold
by the Company for cash, to the extent such pill must be prescribed by a medical doctor, or sold through retail pharmacies over the counter,
in jurisdictions where a doctor’s prescription is not required for the sale of such drugs, and sold in a Territory (defined below),
which consideration per pill decreases each year that the agreement is in effect, and is only payable for the first five years of the
agreement.
The
Consulting Agreement further provides that no payments are due for the sale of any prescription pills until the First Sale.
Under
the Consulting Agreement, (a) “Territory” means worldwide, except for the United States, including its territories and possessions
and the District of Columbia; and (b) “First Sale” means the date that the first commercial sale of prescription pills occurs
in the Territory.
Future
payments are also required to be offset equitably for any prescription pill sold which is later refunded, charged back, returned, or
reimbursed to a purchaser.
The
agreement includes customary representations of the parties, confidentiality and non-solicitation provisions, rights of Epiq Scripts
to audit the sales of prescription pills, subject to certain limitations and requirements, and the requirement that the Company reimburse
certain expenses of Epiq Scripts, subject to certain limitations and pre-approvals.
First
Amendment to MSA
On
September 15, 2023, we entered into a First Addendum to Master Services Agreement (“MSA”) with Epiq Scripts (the “First
Amendment”).
Pursuant
to the First Amendment, the parties agreed to amend the MSA to include certain Right of first negotiation rights and right of first refusal
rights (each as discussed below). Additionally, the First Amendment provides for certain rights to Epiq Scripts in the event that the
Company seeks to obtain pharmaceutical services in connection with certain Company products (collectively, “Pharmaceutical Services”)
in jurisdictions other than the United States, including, without limitation, Mexico and the United Kingdom, where Epiq Scripts does
not currently maintain licenses or permits (“Future Jurisdictions”, which shall also include, to the extent applicable, any
state in the United States in which Epiq Scripts does not then hold required permits or licenses for the provision of the Pharmaceutical
Services) and/or to terminate Epiq Scripts’ rights to provide exclusive Pharmaceutical Services in any current state of the United
States or Future Jurisdiction where Epiq Scripts may then be providing Pharmaceutical Services to the Company (each a “Current
Jurisdiction”).
Specifically,
the parties agreed in the First Amendment that should the Company decide to transfer any services provided by Epiq Scripts in a Current
Jurisdiction to another pharmaceutical service provider (“Transferred Services”), the Company will be required to pay Epiq
Scripts a fee of 1% of the total gross sales of all Prescription Products (defined below) by the Company resulting from the Transferred
Services in the Current Jurisdiction, for a period of the lesser of (a) five (5) years from the date the Company transferred the Transferred
Services; and (b) through the end of the term of the MSA (including where applicable, any renewal term)(the “Non-Use Fee”).
The Non-Use Fee is payable monthly in arrears, for calendar quarters, by the 15th day following the end of each calendar quarter. “Prescription
Products” means Products (as defined in the MSA) sold by the Company which must be prescribed by a medical doctor.
Notwithstanding
the above, the Non-Use Fee shall not apply, and the Company shall not be obligated to pay any Non-Use Fee (a) in the event that the Transferred
Services are provided directly by the Company or a majority-owned subsidiary of the Company; (b) in the event the Company decides to
enter into an agreement with another pharmaceutical service provider to provide Pharmaceutical Services in a Future Jurisdiction; or
(c) in connection with any services provided by any parties in any Future Jurisdictions.
The
First Amendment also provides that until the fifth anniversary of the First Amendment, the Company shall notify Epiq Scripts in writing
of any plans to (a) expand its need for pharmacy services outside of those contemplated by the MSA; (b) expand its need for pharmacy
services into a new jurisdiction which Epiq Scripts does not then operate in (including, but not limited to new countries); or (c) begin
providing pharmacy services internally (either through organic growth or acquisition). Thereafter Epiq Scripts has the right to provide
the Company written notice of its intention to provide such services (as described in (a) or (b) above, whereafter the Company is required
to discuss and negotiate such services in good faith with Epiq Scripts for a period of not less than 15 days). Otherwise, in the event
of the occurrence of an event discussed in (c) above, the Company is required to discuss the possibility of Epiq Scripts either co-operating
the pharmacy or providing management services to the Company in good faith for 15 days. In the event after such 15 day period, the Company
and Epiq Scripts cannot come to a mutually agreeable agreement, the Company is under no further obligation regarding the matter set forth
in the notice provided to Epiq Scripts.
Finally,
the First Amendment includes a requirement whereby if Epiq Scripts receives notice of any proposed fundamental transaction involving
Epiq Scripts or its assets, including any agreement, arrangement, offer or proposal (including a letter of intent, term sheet, form of
definitive agreement or definitive agreement) for an asset sale or acquisition, merger, acquisition or sale of securities, or redemption
or repurchase of securities, Epiq Scripts must provide the Company notice of such offer within three days, after which receipt the Company
will have the right of first refusal for 30 days to become the purchaser in connection with the notified transaction, on the terms, and
subject to the conditions, set forth in such notified offer and pursuant to the conditions of the First Amendment.
Consulting
Agreements
On
September 6, 2022, we entered into a Consulting Agreement with PHX Global, LLC (“PHX”), which is owned by Peter “Casey”
Jensen, who was a member of the Board of Directors of American International. Pursuant to the Consulting Agreement, PHX agreed to provide
consulting and general business advisory services as reasonably requested by the Company during the term of the agreement, which was
for 12 months, unless otherwise earlier terminated due to breach of the agreement by either party, and the failure to cure such breach
30 days after written notice thereof. In consideration for agreeing to provide the services under the agreement, the Company issued PHX
50,000 shares of restricted common stock. The agreement contains customary confidentiality and non-solicitation provisions. We also agreed
to include the shares issued to PHX in the Resale Prospectus, which shares of common stock were included therein.
On
September 6, 2022, we entered into a Consulting Agreement with Ezekiel Elliott (“Elliott”), currently a professional football
player in the National Football League (NFL), to provide consulting and general business advisory services as reasonably requested by
the Company during the term of the agreement, which was for 12 months unless otherwise earlier terminated due to breach of the agreement
by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing to provide the
services under the agreement, the Company issued Elliott 100,000 shares of restricted common stock. The agreement contains customary
confidentiality and non-solicitation provisions. We also agreed to include the shares issued to Elliott in the Resale Prospectus, which
shares of common stock were included therein.
On
September 15, 2022, we entered into a Consulting Agreement with David Sandler, an individual (“Sandler”), to provide consulting
and general business advisory services as reasonably requested by the Company during the term of the agreement, which was for six months,
unless otherwise earlier terminated due to breach of the agreement by either party, and the failure to cure such breach 30 days after
written notice thereof. In consideration for agreeing to provide the services under the agreement, the Company issued Sandler 10,000
shares of restricted common stock. The agreement contains customary confidentiality and non-solicitation provisions. We also agreed to
include the shares issued to Sandler in the Resale Prospectus, which shares of common stock were included therein.
On
September 15, 2022, we entered into a Consulting Agreement with Hsiaoching Chou, an individual (“Chou”), to provide consulting
and general business advisory services as reasonably requested by the Company during the term of the agreement, which was for six months,
unless otherwise earlier terminated due to breach of the agreement by either party, and the failure to cure such breach 30 days after
written notice thereof. In consideration for agreeing to provide the services under the agreement, the Company issued Chou 5,000 shares
of restricted common stock. The agreement contains customary confidentiality and non-solicitation provisions. We also agreed to include
the shares issued to Chou in the Resale Prospectus, which shares of common stock were included therein.
On
September 22, 2022, we entered into a service agreement with Greentree Financial Group, Inc. (“Greentree” and the “Service
Agreement”). Pursuant to the Service Agreement, Greentree agreed to perform the following services: (a) bookkeeping services for
the Company for the period from October 1, 2022 through June 30, 2023; (b) advice and assistance to the Company in connection with the
conversion of its financial reporting systems, including its projected financial statements, to a format that is consistent with United
States Generally Accepted Accounting Principles (“US GAAP”); (c) assistance to the Company with compliance filings for the
quarters ended September 30, 2022, March 31, 2023, June 30, 2023 and the year ended December 31, 2022, including the consolidation structure
and entries as well as assistance with US GAAP footnotes; (d) reviewing, and providing advice to the Company on, all documents and accounting
systems relating to its finances and transactions, with the purpose of bringing such documents and systems into compliance with US GAAP
or disclosures required by the SEC; and (e) providing necessary consulting services and support as a liaison for the Company to third
party service providers, including coordination amongst the Company and its attorneys, CPAs and transfer agent. Since February 2015,
Eugene M. Johnston, our Chief Financial Officer (who was appointed October 1, 2022), has served as Audit Manager for Greentree.
The
Company agreed to issue Greentree 100,000 shares of the Company’s restricted common stock upon the parties’ entry into the
agreement, and to pay Greentree $50,000 in cash, payable as follows: (a) $12,500 on or before September 30, 2022, which has been paid;
(b) $12,500 on or before December 31, 2022, which has been paid; (c) $12,500 or before March 31, 2023; and (d) $12,500 on or before June
30, 2023. We also agreed to include the 100,000 shares of common stock issued to Greentree in the Resale Prospectus, which shares of
common stock are included therein, and to reimburse Greentree for its reasonable out-of-pocket expenses incurred in connection with Greentree’s
activities under the agreement, including the reasonable fees and travel expenses for the meetings on behalf of the Company.
The
Service Agreement continued in effect through August 14, 2023.
The
Service Agreement includes customary indemnification obligations requiring the Company to indemnify Greentree and its affiliates with
regard to certain matters.
On
November 1, 2022, we entered into a Consulting Agreement with White Unicorn, LLC (“White Unicorn”), to provide business advisory
services related to product packaging, strategic marketing, branding, advertising and future product development as reasonably requested
by the Company during the term of the agreement, which was for 12 months unless otherwise earlier terminated due to breach of the agreement
by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing to provide the
services under the agreement, the Company issued White Unicorn 100,000 shares of restricted common stock. The agreement contains customary
confidentiality and non-solicitation provisions.
On
December 21, 2022, we entered into a Consulting Agreement with Chartered Services, LLC (“Chartered Services”), to provide
strategic marketing services for advertising and consulting, product distribution, digital marketing and identifying creative and constructive
brand awareness to the Company during the term of the agreement, which was for six months unless otherwise earlier terminated due to
breach of the agreement by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for
agreeing to provide the services under the agreement, the Company agreed to pay Chartered Services $150,000 in cash (with $75,000 payable
upon entry into the agreement and $75,000 payable on January 31, 2023, which amount has been paid to date) and issued Chartered Services
250,000 shares of restricted common stock. The agreement contains customary confidentiality and non-solicitation provisions.
On
January 3, 2023, we entered into a Consulting Agreement with DojoLabs Group, Inc. (“DojoLabs”), to provide various strategic
marketing related services to the Company pursuant to a defined scope of work during the term of the agreement, which is the earlier
of a) all deliverables being received by the Company pursuant to the scope of work, or b) if terminated due to breach of the agreement
by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing to provide the
services under the agreement, the Company agreed to pay DojoLabs $100,000 in cash and issued DojoLabs 50,000 shares of restricted common
stock with registration rights (the registration of the resale of which shares were included in the Resale Prospectus) and fully vest
upon the completion of all work performed under the scope of work. The agreement contains customary confidentiality and non-solicitation
provisions.
On
January 6, 2023, we entered into a Consulting Agreement with Bethor, Ltd. (“Bethor”), to provide strategic advisory services
to the Company during the term of the agreement, which was for 12 months unless otherwise earlier terminated due to breach of the agreement
by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing to provide the
services under the agreement, the Company issued Bethor 250,000 shares of restricted common stock with registration rights (the registration
of the resale of which shares were included in the Resale Prospectus). The agreement contains customary confidentiality and non-solicitation
provisions.
On
January 6, 2023, the Company established an advisory board (the “Advisory Board”) and approved and adopted a charter (the
“Advisory Board Charter”) to govern the Advisory Board. Pursuant to the Advisory Board Charter, the Advisory Board shall
be comprised of a minimum of two (2) members, all of whom shall be appointed and subject to removal by the Board of Directors at any
time. In addition to the enumerated responsibilities of the Advisory Board in the Advisory Board Charter, the primary function of the
Advisory Board is to assist the Board of Directors in its general oversight of the Company’s development of new business ventures
and strategic planning.
In
connection with the establishment of the Advisory Board, the Board of Directors appointed Dr. Brian Rudman (“Dr. Rudman”)
and Mr. Jarrett Boon (“Mr. Boon”), both of whom are independent, non-Board members and non-Company employees, to the Advisory
Board. Dr. Rudman serves as Chairman of the Advisory Board.
In
connection with Dr. Rudman’s appointment to the Advisory Board, the Company entered into an Advisor Agreement (the “Dr. Rudman
Consulting Agreement”), dated effective January 6, 2023, with Dr. Rudman, whereby the Company agreed to issue Dr. Rudman 25,000
shares of the Company’s restricted common stock, pay Dr. Rudman $2,000 per month in cash, and reimburse Dr. Rudman for reasonable
out-of-pocket expenses, including, without limitation, travel expenses incurred by him in connection with the Company’s requests
of the performance of his duties to the Company in service on the Advisory Board.
In
connection with Mr. Boon’s appointment to the Advisory Board, the Company entered into an Advisor Agreement (the “Mr. Boon
Consulting Agreement”), dated effective January 6, 2023, with Mr. Boon, whereby the Company agreed to issue Mr. Boon 25,000 shares
of the Company’s restricted common stock and to reimburse Mr. Boon for reasonable out-of-pocket expenses, including, without limitation,
travel expenses incurred by him in connection with the Company’s requests of the performance of his duties to the Company in service
on the Advisory Board.
On
January 24, 2023, we entered into Consulting Agreements with four consultants to the Company: (1) Sultan Haroon; (2) John Helfrich; (3)
Justin Baker; and (4) Maja Matthews, each of whom is also an employee of Epiq Scripts. Pursuant to the Consulting Agreements, the Consultants
agreed to provide us services related to the research, development, packaging and marketing for additional pharmaceutical and other over-the-counter
related products during the term of the agreement, which each had a term of 18 months unless otherwise earlier terminated due to breach
of the agreement by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing
to provide the services under the agreement, the Company issued an aggregate of 350,000 shares of common stock to the consultants as
follows: (1) Sultan Haroon 150,000 shares of restricted common stock; (2) John Helfrich 25,000 shares of restricted common stock; (3)
Justin Baker 25,000 shares of restricted common stock; and (4) Maja Matthews 150,000 shares of restricted common stock. The shares issued
to Haroon and Matthews vest at the rate of 50,000 shares upon entry into the agreement, 50,000 shares upon the Company’s successful
launch of a new product category, and 50,000 shares upon the Company’s successful launch of a second and additional new product
category, in each case prior to the 18-month anniversary of the applicable agreement. The shares issued to Helfrich and Baker vest at
the rate of 10,000 shares upon entry into the agreement, 7,500 shares upon the Company’s successful launch of a new product category,
and 7,500 shares upon the Company’s successful launch of a second and additional new product category, in each case prior to the
18-month anniversary of the applicable agreement. Any shares not vested by the eighteen-month anniversary of the applicable agreement
are forfeited. The agreement contains customary confidentiality and non-solicitation provisions.
On
May 1, 2023, we entered into a Software Development Agreement with Redlime Solutions, Inc. (“Redlime”) to provide software
development services during the term of the agreement, which is for 12 months. In consideration for agreeing to provide the services
under the agreement, the Company agreed to pay Redlime $300,000 in cash and issue Redlime 180,000 shares of restricted common stock.
The shares were valued at $1.00 per share for a total of $180,000.
On
May 25, 2023, the Board of Directors appointed Mr. Aaron Andrew, an independent, non-Board member and non-Company employee, to the Advisory
Board. In connection with Mr. Andrew’s appointment to the Advisory Board, the Company entered into an Advisor Agreement (the “Andrew
Consulting Agreement”), dated effective May 25, 2023, with Mr. Andrew, whereby the Company agreed to issue Mr. Andrew 50,000 shares
of the Company’s restricted common stock under the 2022 Plan and to reimburse Mr. Andrew for reasonable out-of-pocket expenses,
including, without limitation, travel expenses incurred by him in connection with the Company’s requests of the performance of
his duties to the Company in service on the Advisory Board. The shares were valued at $1.10 per share for a total of $55,000.
On
June 1, 2023, we entered into a Consulting Agreement with Major Dodge (“Major”), to provide acting and production related
services to the Company during the term of the agreement, which is for 12 months unless otherwise earlier terminated due to breach of
the agreement by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing
to provide the services under the agreement, the Company issued Major 20,000 shares of restricted common stock under the 2022 Plan. The
agreement contains customary confidentiality and non-solicitation provisions. The shares were valued at $1.10 per share for a total of
$22,000.
On
June 1, 2023, we entered into a Production and Broadcasting Agreement with New To The Street Group, LLC (“New To The Street”),
to provide production, broadcasting and other marketing related services to the Company during the term of the agreement, which was for
3 months unless otherwise earlier terminated. In consideration for agreeing to provide the services under the agreement, the Company
issued New To The Street 50,000 shares of restricted common stock and agreed to pay New To The Street a monthly cash payment of $5,000.
The shares were valued at $1.10 per share for a total of $55,000.
On
September 1, 2023, we entered into a service agreement with Greentree. Pursuant to the Service Agreement, Greentree agreed to perform
the following services: (a) bookkeeping services for the Company for the period from October 1, 2023 through September 30, 2024; (b)
advice and assistance to the Company in connection with the conversion of its financial reporting systems, including its projected financial
statements, to a format that is consistent with US GAAP; (c) assistance to the Company with compliance filings for the quarters ended
September 30, 2023, March 31, 2024, June 30, 2024 and the year ended December 31, 2023, including the structure and entries as well as
assistance with US GAAP footnotes; (d) reviewing, and providing advice to the Company on, all documents and accounting systems relating
to its finances and transactions, with the purpose of bringing such documents and systems into compliance with US GAAP or disclosures
required by the SEC; and (e) providing necessary consulting services and support as a liaison for the Company to third party service
providers, including coordination amongst the Company and its attorneys, CPAs and transfer agent. Since February 2015, Eugene (Gene)
M. Johnston, our Chief Financial Officer (who was appointed October 1, 2022) has served as an Audit Manager for Greentree.
The
Company agreed to issue Greentree 75,000 shares of the Company’s restricted common stock upon the parties’ entry into the
agreement, and to pay Greentree $40,000 in cash, payable as follows: (a) $20,000 on or before September 30, 2023; (b) $20,000 on or before
March 31, 2024. We also agreed to reimburse Greentree for its reasonable out-of-pocket expenses incurred in connection with Greentree’s
activities under the agreement, including the reasonable fees and travel expenses for the meetings on behalf of the Company. The Service
Agreement includes customary indemnification obligations requiring the Company to indemnify Greentree and its affiliates with regard
to certain matters. The shares were valued at $1.13 per share for a total of $84,750.
On
November 1, 2023, the Board of Directors appointed Dr. Douglas Christianson (“Dr. Christianson”) an independent, non-Board
member and non-Company employee, to the Advisory Board. In connection with Dr. Christianson’s appointment to the Advisory Board,
the Company entered into an Advisor Agreement (the “Christianson Consulting Agreement”), dated effective November 1, 2023,
with Dr. Christianson, whereby the Company agreed to issue Dr. Christianson 50,000 shares of the Company’s common stock under the
2022 Plan, which vest six months from the issuance date, and to reimburse Dr. Christianson for reasonable out-of-pocket expenses, including,
without limitation, travel expenses incurred by him in connection with the Company’s requests of the performance of his duties
to the Company in his service on the Advisory Board. The agreement has a one year term, but can be terminated with written notice from
either party with 30 days’ notice. The agreement includes customary confidentiality obligations relating to Dr. Christianson and
indemnification obligations of the parties, requiring each party to indemnify and hold harmless the other against breaches of the agreement
and intentionally misconduct or gross negligence (Dr. Christianson) and the operations of the Company (the Company). The shares were
valued at $0.65 per share for a total of $32,500.
On
November 1, 2023, we entered into an Influencer Contract with Jason Szkup (“Scoop”), to provide influencer and marketing
related services to the Company during the term of the agreement, including posting social media videos. The agreement has a term of
three months, unless otherwise earlier terminated. In consideration for agreeing to provide the services under the agreement, the Company
agreed to pay Scoop $10,000 and to issue Scoop 30,000 shares of common stock under the 2022 Plan. The agreement contains customary confidentiality
and non-disclosure provisions. The shares were valued at $0.65 per share for a total of $19,500.
On
November 7, 2023, we entered into a subsequent Consulting Agreement with PHX to provide consulting and general business advisory services
as reasonably requested by the Company during the term of the agreement, which was for 12 months, unless otherwise earlier terminated
due to breach of the agreement by either party, and the failure to cure such breach 30 days after written notice thereof. In consideration
for agreeing to provide the services under the agreement, the Company paid PHX $25,000 in cash and issued PHX 200,000 shares of common
stock under the 2022 Plan. The agreement contains customary confidentiality and non-solicitation provisions.
Master
Services Agreement with Global Career Networks
On
December 1, 2022, the Company entered into a Master Services Agreement with Global Career Networks, Inc. (“GCN”). Pursuant
to the agreement, we issued GCN 100,000 shares of restricted common stock with registration rights (which shares were included in the
Resale Prospectus) and GCN agreed to assist us with a planned twitter marketing campaign. The agreement has a one year term (provided
the individual project described therein had a six month term, beginning December 1, 2022), and may be renewed thereafter for additional
one year terms with the mutual approval of the parties. Either party may terminate the agreement at any time for any reason, with at
least 60 days’ notice, or upon the occurrence of any breach or default under the agreement, which remains uncured within 30 days
of written notice thereof, or if the non-terminating party is subject to bankruptcy. The agreement contains customary confidentiality,
indemnification obligations, and limitations of liability.
Intellectual
Property
We
believe that our ability to obtain and maintain intellectual property protection for our technology platform, preserve the confidentiality
of our trade secrets, and operate without violating the intellectual property rights of others will be important to our success. We rely
on a combination of trademark, copyright, trade secret, including federal, state and common law rights in the United States and other
countries, nondisclosure agreements, and other measures to protect our intellectual property, and may seek patent protection of our intellectual
property in the future. Despite any measures taken to protect our intellectual property, unauthorized parties may attempt to copy aspects
of our products or to obtain and use information that we regard as proprietary. Our business is affected by our ability to protect against
misappropriation and infringement of our intellectual property and other proprietary rights.
Our
intellectual property includes the content of our websites, our registered domain names, our unregistered trademarks, and certain trade
secrets.
We
have been granted with the United States Patent and Trademark Office for a federal trademark for the following word mark on October 13,
2024 with Reg. No. 7,184,368:

Additionally,
the Company has been granted with the United States Patent and Trademark Office for the following federal trademarks:
-
If You Take It They Will Come April 11, 2023 Reg. No. 7,025,954
-
It Takes Two To Mango May 16, 2023 Reg. No. 7,055,400
-
Orange Is The New Blue December 19, 2023 Reg. No. 7,246,645
-
Big Mango Energy November 28, 2023 Reg. No. 7,232,305
The
Company has also applied with the United States Patent and Trademark Office for the following federal trademarks:
-
TreatMint
-
Make Every Day Hump Day
Employees
The
Company is currently operated and managed by the Founder, Chairman and Chief Executive Officer, Jacob D. Cohen, Amanda Hammer, the Chief
Operating Officer of the Company, and Eugene Johnston, the Chief Financial Officer of the Company. The Company utilizes the assistance
of various independent contractors for administrative and technology development related services. We anticipate establishing a compensation
program designed to align the compensation of our employees with performance and to provide the proper incentives to attract, retain
and motivate employees to achieve superior results in the future. The structure of our anticipated compensation program will balance
incentives earnings for both short-term and long-term performance such as incentive bonuses and flexible schedules. The Company also
intends to develop a culture of inclusion and diversity and places a high value on diversity and inclusion. Our future success will depend
partially on our ability to attract, retain and motivate qualified personnel. We are not a party to any collective bargaining agreements
and have not experienced any strikes or work stoppages. We consider our relations with our employees to be satisfactory. Mr. Cohen and
Mrs. Hammer, are currently party to employment agreements with the Company as discussed below under “Executive
and Director Compensation-Employment Agreements.”
Properties
On
September 28, 2022, and with an effective date of October 1, 2022, the Company entered into a Lease Agreement with Rox Trep Tollway,
L.P. (the “Landlord”) to lease and occupy approximately 2,201 square feet of office space located at 15110 N. Dallas Parkway,
Suite 600, Dallas, Texas 75248 to serve as the Company’s main headquarters (the “Lease Agreement”). The Lease Agreement
has a term of 38 months (through December 31, 2025) and has a monthly base rent of $0 for the second month; $5,778, or $31.50 per square
foot, for months 1 and 3-18 and increases at the rate of $1 per square foot per annum thereafter until the end of the lease term (the
“Base Rent”). In addition to the Base Rent, the Company is required to reimburse the landlord for its pro-rata share of all
real estate taxes and assessments, hazard and liability insurance and common area maintenance costs for the building at the rate of 2.45%
(the “Proportionate Rent”). Upon the execution of the Lease Agreement, the Company agreed to prepay the first full month’s
Base Rent along with a security deposit equal to $16,942. The lease includes an option to extend the lease for an additional period of
36 calendar months at market.
We
believe our facilities are sufficient to meet our current needs and that suitable space will be available as and when needed. We do not
own any real property.
Legal
Proceedings
There
are no pending or threatened legal proceedings involving our company. However, from time to time, we may become involved in various legal
proceedings that arise in the ordinary course of business. Those claims, even if lacking merit, could result in the expenditure by us
of significant financial and managerial resources. We may become involved in material legal proceedings in the future.
DETERMINATION
OF OFFERING PRICE
The
selling stockholders will offer shares of our common stock at the prevailing market prices or privately negotiated prices. The offering
price of our common stock does not necessarily bear any relationship to our book value, assets, past operating results, financial condition,
or any other established criteria of value. Our common stock may not trade at the market prices in excess of the offering prices for
common stock in any public market, will be determined in the marketplace, and may be influenced by many factors, including the depth
and liquidity of the market for our common stock.
MARKET
PRICE OF COMMON STOCK AND RELATED STOCKHOLDER MATTERS
Market
Information
Our
common stock commenced trading on the Nasdaq under the symbol “MGRX” on March 21, 2023.
Holders
As
of May 6, 2024, 24,819,500 shares of our common stock were outstanding among 34 holders of record.
Dividend
Policy
We
currently intend to retain all available funds and any future earnings, if any, to fund the development and expansion of our business
and we do not anticipate paying any cash dividends in the foreseeable future. Except for a one-time special dividend in connection with
our distribution of the Shi Loan (as defined herein), we have not paid any cash dividends. See “Certain Relationships and Related
Party Transactions.” Any future determination related to our dividend policy will be made at the discretion of our board of directors
after considering our financial condition, results of operations, capital requirements, business prospects, and other factors our board
of directors deems relevant, and subject to the restrictions contained in any future financing instruments. In addition, our ability
to pay cash dividends is currently restricted by the terms of our credit facilities. Our ability to pay cash dividends on our capital
stock in the future may also be limited by the terms of any preferred securities we may issue or agreements governing any additional
indebtedness we may incur.
SECURITY
OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The
following table sets forth certain information regarding the beneficial ownership of our common stock as of May 6, 2024 (the “Date
of Determination”) by (i) each Named Executive Officer, as such term is defined above under “Executive
Compensation,” (ii) each member of our Board of Directors, (iii) each person deemed to be the beneficial owner of more than
five percent (5%) of our common stock, and (iv) all of our executive officers and directors as a group. Unless otherwise indicated, each
person named in the following table is assumed to have sole voting power and investment power with respect to all shares of our common
stock listed as owned by such person.
The
column titled “Beneficial Ownership” is based on a total of 24,819,500 shares of our common stock outstanding as of
the Date of Determination.
Beneficial
ownership is determined in accordance with the rules of the SEC and includes voting and/or investing power with respect to securities.
These rules generally provide that shares of common stock subject to options, warrants or other convertible securities that are currently
exercisable or convertible, or exercisable or convertible within 60 days of the Date of Determination, are deemed to be outstanding and
to be beneficially owned by the person or group holding such options, warrants or other convertible securities for the purpose of computing
the percentage ownership of such person or group, but are not treated as outstanding for the purpose of computing the percentage ownership
of any other person or group.
To
our knowledge, except as indicated in the footnotes to this table and pursuant to applicable community property laws, as of the Date
of Determination, (a) the persons named in the table have sole voting and investment power with respect to all shares of common stock
shown as beneficially owned by them, subject to applicable community property laws; and (b) no person owns more than 5% of our common
stock. Unless otherwise indicated, the address for each of the officers or directors listed in the table below is 15110 N. Dallas Parkway,
Suite 600, Dallas, Texas 75248.
| Name of Beneficial Owner | |
Number of
Common Stock Shares Beneficially Owned | | |
Percent Beneficial Ownership | |
| Directors, Named Executive Officers and Executive Officers | |
| | | |
| | |
| Jacob D. Cohen | |
| 9,775,000 | (1) | |
| 39.7 | % |
| Eugene M. Johnston | |
| 200,000 | | |
| * | % |
| Amanda Hammer | |
| 75,000 | (2) | |
| * | |
| Lorraine D’Alessio | |
| 75,000 | (3) | |
| * | |
| Alex P. Hamilton | |
| 75,000 | (3) | |
| * | |
| Dr. Kenny Myers | |
| 75,000 | (4) | |
| * | |
| All executive officers and directors as a group (6 persons) | |
| 11,441,667 | (1)(2) | |
| 46.5 | % |
| | |
| | | |
| | |
| Greater than 5% Stockholders | |
| | | |
| | |
| None | |
| | | |
| | |
*
Less than 1%.
| (1) |
The
outstanding shares of common stock beneficially owned by Mr. Cohen are held in the name of The Tiger Cub Trust, which is beneficially
owned by Jacob D. Cohen, its Trustee, and which shares Mr. Cohen is deemed to beneficially own. Includes 1,250,000 shares of common
stock issuable upon exercise of options to purchase shares of common stock held by Mr. Cohen, with an exercise price of $0.32 per
share and 250,000 shares of common stock issuable upon exercise of options to purchase shares of common stock of the Company held
by Mr. Cohen, with an exercise price of $1.10 per share, and does not include options to purchase 500,000 shares of common stock
which an exercise price of $1.10 per share, which vest at the rate of 1/2 of such options on each of September 1, 2024 and 2025,
with a term of five years. |
| |
|
| (2) |
Does
not include options to purchase 150,000 shares of common stock with an exercise price of $1.10 per share, which vest at the rate
of 1/3 of such options on each of May 1, 2024, 2025 and 2026, which have not vested as of the Date of Determination. The options
have a 10 year term. |
| |
|
| (3) |
Includes
25,000 shares of restricted common stock which vest on October 14, 2024, subject to the holder’s continued service with the
Company. |
| |
|
| (4) |
Address:
15110 Dallas Parkway, Suite 600, Dallas, Texas 75248 |
Change
of Control
The
Company is not aware of any arrangements which may at a subsequent date result in a change of control of the Company.
Equity
Compensation Plan Information
The
following table provides information as of December 31, 2023 regarding the Company’s 2022 Equity Incentive Plan, as amended, under
which equity securities are authorized for issuance:
| Plan Category | |
Number of
securities
to be issued upon
exercise of
outstanding
options,
warrants and
rights | | |
Weighted-
average
exercise price
of
outstanding
options,
warrants and
rights | | |
Number of
securities
available for future
issuance under
equity
compensation plans
(excluding those in
first column) | |
| Equity compensation plans approved by the security holders (1) | |
| 2,650,000 | | |
$ | 0.73 | | |
| 923,250 | |
| Equity compensation plans not approved by the security holders | |
| - | | |
| - | | |
| - | |
| Total | |
| 2,650,000 | | |
$ | 0.73 | | |
| 518,250 | |
| (1) |
Represents
options issuable upon grants previously made under the Company’s 2022 Equity Incentive Plan, as amended, which is discussed
under “Executive Compensation-2022 Equity Incentive Plan.” |
Legal
Proceedings
Although
we may, from time to time, be involved in litigation and claims arising out of our operations in the normal course of business, we are
not currently a party to any material legal proceeding. In addition, we are not aware of any material legal or governmental proceedings
against us or contemplated to be brought against us. The impact and outcome of litigation, if any, is subject to inherent uncertainties,
and an adverse result in these or other matters may arise from time to time that may harm our business. Those claims, even if lacking
merit, could result in the expenditure by us of significant financial and managerial resources. We may become involved in material legal
proceedings in the future.
MANAGEMENT
Executive
Officers and Directors
The
following table sets forth information with respect to persons who are serving as directors and executive officers of the Company as
of May 6, 2024.
| Name |
|
Position |
|
Age |
|
Director
Since |
| Jacob
D. Cohen |
|
Chairman
and Chief Executive Officer |
|
45 |
|
October
2021 |
| Eugene
M. Johnston |
|
Chief
Financial Officer |
|
60 |
|
- |
| Amanda
Hammer |
|
Chief
Operating Officer |
|
38 |
|
- |
| Lorraine
D’Alessio |
|
Director |
|
44 |
|
October
2022 |
| Alex
P. Hamilton |
|
Director |
|
51 |
|
October
2022 |
| Dr.
Kenny Myers |
|
Director |
|
57 |
|
October
2022 |
Business
Experience
The
following is a brief description of the education and business experience of our directors and executive officers.
Jacob
D. Cohen - Chairman and Chief Executive Officer
Jacob
Cohen is a serial entrepreneur, corporate finance and executive management professional with over 20 years of investment banking and
capital markets experience having started and growing multiple companies in various industry sectors including marketing, advertising,
healthcare, IT and financial services. Prior to founding the Company, Mr. Cohen was the co-founder and managing partner of several boutique
investment bank and strategic advisory firms where he advised both early and later stage companies in raising capital in the form of
debt and/or equity and in both private and public markets.
Prior
to his experiences in investment banking, Mr. Cohen served as the Chief Financial Officer of The Renewed Group, Inc., a manufacturer,
wholesaler and retailer of eco-friendly and sustainable apparel primarily made from recycled textiles and under the brand name REUSE
JEANS from 2010 through the end of 2013. Further, Mr. Cohen served from 2008 through 2010 as Executive Vice President and Controller
of Metiscan, Inc., a publicly-traded company, and as the President and Chief Executive Officer of one of its subsidiaries, Shoreline
Employment Services, Inc. During his tenure at Metiscan, Mr. Cohen was instrumental in restructuring, reorganizing and operating the
company and its five subsidiaries, and successfully raised over $8 million in equity financing for growth capital. Mr. Cohen also spearheaded
the company’s financial audit process and managed its various filings with the SEC.
From
2007 through 2008, Mr. Cohen served as the Chief Operating Officer of Artfest International, which he assisted in taking public at the
end of 2007. Throughout his career, Mr. Cohen was involved in starting many new ventures, including The AdvertEyes Network, a digital
signage advertising company where he served as founder and CEO. Other positions include investment advisor and institutional equity research
analyst for Solomon Advisors and Huberman Financial, securities broker-dealers, from 2003 through 2005, and investment banker for Allegiance
Capital, a middle market investment bank specializing on mergers and acquisitions, from 2005-2007. Mr. Cohen holds a Bachelor of Arts
in International Economics and Finance from Brandeis University in Waltham, Massachusetts.
Mr.
Cohen has served as Chief Executive Officer of the Company since October 2021, as a director from October 2021 to present, and as Chairman
from September 2022 to present. Mr. Cohen also currently serves as a director of American International, a publicly-traded company which
was the majority owner and parent to Epiq Scripts, LLC prior to February 15, 2023, and which is the former sole owner of the Company,
having fully divested its ownership in June 2022. Mr. Cohen served as Chief Executive Officer and President of American International
from April 2019 to March 2023. Cohen also serves as Chief Executive Officer of Ronin Equity Partners, Inc., a private investment company,
which role he has held since August 2016. Mr. Cohen also serves the Chief Executive Officer of Cohen Enterprises, Inc., a private investment
company, which position he has held since November 2013. Since February 15, 2023, Mr. Cohen has owned 51% of and controlled, Epiq Scripts.
Mr. Cohen has served as the co-Manager of Epiq Scripts since January 2022.
We
believe that Mr. Cohen’s extensive background in investment banking, public company management and corporate finance makes him
well qualified to serve on the Board of Directors.
Eugene
M. Johnston - Chief Financial Officer
Mr.
Johnston has served as Chief Financial Officer of the Company since October 2022. Since February 2015, Mr. Johnston has served as Audit
Manager for Greentree Financial Group, Inc., an accounting and auditing firm. From August 1999 to September 2014, Mr. Johnston served
as Chief Executive Officer of Peoplesway.com, Inc., a skincare and nutritional products company, and from August 1999 to present, Mr.
Johnston has served as a member of the Board of Directors of Peoplesway.com, Inc. From January 1999 to July 1999, Mr. Johnston served
as Chief Executive Officer of RMC Group, Inc., a skincare and nutritional products company. Prior to that, from April 1987 to January
1989, Mr. Johnston served as Vice President of Sales Administration at WeCare Distributors, Inc., a skincare and nutritional products
company. Mr. Johnston received a Bachelor’s in Science in Business Administration from the University of North Carolina Charlotte.
Amanda
Hammer - Chief Operating Officer
Mrs.
Hammer has served as the Company’s Chief Operating Officer since May 2023 and as director of e-Commerce from October 2022 to May
2023. Prior to that, she served in various roles with D Magazine Partners, a media/publishing company, including Chief Operating Officer
(December 2021 to September 2022); Audience Development and Digital Operations Director (July 2019 to November 2021); and Audience Development
Director (August 2018 to June 2019). From February 2018 to July 2018, Mrs. Hammer served as a Sales Consultant with Liberty Mutual insurance.
From October 2014 to October 2017, Mrs. Hammer served as Director of Membership and Product Development at McKissock LLC, a professional
development / e-learning company. Prior to that, from August 2008 to September 2014, she served as Training and Membership Director at
The Institute for Luxury Home Marketing, a real estate / professional association. Mrs. Hammer obtained dual Bachelor of Arts degrees
(i) with a concentration in Graphic Design, and (ii) in Communication Studies, from the University of Iowa. She has also obtained a Negotiation
and Leadership Certificate from Harvard Law School. She is a member of the Texas Women’s Foundation and the MetroTex Young Professionals
Network.
Independent
Directors
Lorraine
D’Alessio - Director
Lorraine
D’Alessio was elected as a director of the Company effective October 14, 2022. From January 2022 to March 2023, Ms. D’Alessio
has served as a member of the Board of Directors and member of the Audit Committee of the Board of Directors of American International.
Since
2010, Ms. D’Alessio has served as CEO and Managing Partner at D’Alessio Law Group, PLC, a law firm in Beverly Hills, California
which provides immigration and entertainment law services. In that capacity, she has provided counsel to entertainment agencies, unions,
private companies, academic institutions, tech startups, entrepreneurs and enterprises including: Next Models, Food Network, SubPac,
Pepperdine University, ACTRA, New York Film Academy, Plug and Play, Expert Dojo, and 500 Startups.
Ms.
D’Alessio was named the 2017 Leader in Law by the Los Angeles Business Journal and is the recipient of the 2018 Enterprising Woman
Award. Since 2016, Ms. D’Alessio has also served on the board of directors of Artists for Change, a non-profit organization which
focuses on creating high impact film, television, and multimedia projects to inspire individuals, organizations, and communities to bring
about positive social change.
From
2005 to 2007, Ms. D’Alessio served as a policy analyst and advisor for the government of Ontario, Canada.
Ms.
D’Alessio received her Bachelor’s degree in International Relations from the University of Toronto in 2005, a Master’s
of Public Policy in Public Policy Administration from Queen’s University, in Kingston, Ontario in 2006, and a Juris Doctorate degree
from Southwestern Law School in Los Angeles, California in 2010.
The
Board of Directors believes that Ms. D’Alessio is well qualified to serve on the Board of Directors because of her legal expertise
and extensive knowledge of corporate governance and controls.
Alex
P. Hamilton - Director
Alex
P. Hamilton was elected as a director of the Company effective October 14, 2022.
In
April 2016, Mr. Hamilton founded Hamilton Laundry, a boutique laundromat that serves high-end luxury commercial companies, and has served
as its chief executive officer since then. He has also served as Chief Executive Officer of Hamilton Strategy Group, Inc., a consulting
firm, since November 2014. Mr. Hamilton is also the Co-Founder of Donald Capital LLC, a FINRA registered investment banking firm, and
has served as its president since May 2019. Since May 2021, Mr. Hamilton has served as a member of the Board of Directors, the Chairman
of the Audit Committee and member of the Corporate Governance and Nominating Committee of Addentax Group Corp. (ATXG:Nasdaq), an integrated
service provider focusing on garment manufacturing, logistics service, property management and subleasing, and epidemic prevention supplies.
From February 2017 to July 2019, Mr. Hamilton served as Chief Financial Officer of Hemp Logic, Inc. From December 2018 to February 2019,
Mr. Hamilton served as the Interim Chief Financial Officer of ChineseInvestors.com, Inc. From December 2020 to July 2021, Mr. Hamilton
served as a non-executive Board Member, Chairman of the Audit Committee and Member of the Nominating and Compensation Committee of Meiwu
Technology Co., LTD (WNW:Nasdaq). Mr. Hamilton, served as the Chief Financial Officer and Director of CBD Biotech, Inc. from November
2018 to February 2021. From January 2015 to May 2019, Mr. Hamilton served as Senior Managing Director of Consilium Global Research. From
November 2013 to November 2014, Mr. Hamilton was the president of Kei Advisors. From November 2012 to November 2013, Mr. Hamilton served
as Senior Director of FTI Consulting, a management consulting company. Prior to that, Mr. Hamilton served as managing director of Early
Bird Capital (August 2010 to September 2012) and Jesup & Lamont (July 2007 to February 2010), and as a Vice President of The Benchmark
Company (February 2006 to July 2007). Mr. Hamilton holds his Series 7, 24 and 63 licenses. Mr. Hamilton received a Batchelor’s
Degree in Economics from Brandeis University in Waltham, Massachusetts.
The
Board of Directors believes that Mr. Hamilton is well qualified to serve on the Board of Directors because of his extensive business
knowledge, public company experience and experience serving in various positions with investment management firms.
Dr.
Kenny Myers - Director
Dr.
Kenny Myers was elected as a director of the Company effective October 14, 2022. From January 2022 to March 2023, Dr. Myers has served
as a member of the Board of Directors and Audit Committee of American International.
Since
March 2020, Dr. Myers has served as VP of Business Development for Living Fit Nation, Inc., a corporate wellness provider which designs
and implements customized employee health and wellness programs for corporations around the United States. From March 2012 to February
2020, Dr. Myers worked as VP of Business Development at One Health Medical Systems, LLC, an integrated health services provider, where
he was responsible for overseeing the planning, development and execution of the organization’s marketing and advertising initiatives.
From May 1998 to March 2012, Dr. Myers was CEO of Texas Physicians Network, a healthcare management company where he was responsible
for the marketing and management of several urgent care centers, medical clinics and other related healthcare facilities.
Dr.
Myers received his Bachelor of Science degree in Microbiology from Oklahoma University in 1989, and a Doctor of Chiropractic Degree from
Parker University in Dallas, Texas in 1996.
The
Board of Directors believes that Dr. Myers is well qualified to serve on the Board of Directors because of his background in the health
services industry and his experience in business marketing and development.
Terms
of Office of Officers and Directors
In
accordance with Nasdaq corporate governance requirements, we are not required to hold an annual meeting until December 31, 2024, one
fiscal year following our listing on Nasdaq. The term of office of our directors will expire at our first annual meeting of shareholders,
subject to re-nomination and reappointment to the board by our shareholders.
Our
officers are appointed by the Board of Directors and serve at the discretion of the Board of Directors, rather than for specific terms
of office. Our Board of Directors is authorized to appoint persons to the offices set forth in our Bylaws as it deems appropriate. Our
Bylaws provide that our officers may consist of a Chairman of the Board, Chief Executive Officer, Chief Financial Officer, President,
Vice Presidents, Secretary, Treasurer, Assistant Secretaries and such other offices as may be determined by the Board of Directors.
Corporate
Governance
Family
Relationships among Directors and Officers
There
are no family relationships among our directors and executive officers.
Arrangements
between Directors and Officers
To
our knowledge, there is no arrangement or understanding between any of our officers or directors and any other person, including directors,
pursuant to which the officer was selected to serve as an officer or director.
Involvement
in Certain Legal Proceedings
None
of our executive officers or directors has been involved in any of the following events during the past ten years: (1) any bankruptcy
petition filed by or against any business of which such person was a general partner or executive officer either at the time of the bankruptcy
or within two years prior to that time; (2) any conviction in a criminal proceeding or being a named subject to a pending criminal proceeding
(excluding traffic violations and minor offenses); (3) being subject to any order, judgment, or decree, not subsequently reversed, suspended
or vacated, of any court of competent jurisdiction, permanently or temporarily enjoining, barring, suspending or otherwise limiting his
involvement in any type of business, securities or banking activities; (4) being found by a court of competent jurisdiction (in a civil
action), the SEC or the Commodities Futures Trading Commission to have violated a federal or state securities or commodities law; (5)
being the subject of, or a party to, any Federal or State judicial or administrative order, judgment, decree, or finding, not subsequently
reversed, suspended or vacated, relating to an alleged violation of (i) any Federal or State securities or commodities law or regulation;
(ii) any law or regulation respecting financial institutions or insurance companies, including, but not limited to, a temporary or permanent
injunction, order of disgorgement or restitution, civil money penalty or temporary or permanent cease-and-desist order, or removal or
prohibition order, or (iii) any law or regulation prohibiting mail or wire fraud or fraud in connection with any business entity; or
(6) being the subject of, or a party to, any sanction or order, not subsequently reversed, suspended or vacated, of any self-regulatory
organization (as defined in Section 3(a)(26) of the Exchange Act), any registered entity (as defined in Section (1a)(40) of the Commodity
Exchange Act), or any equivalent exchange, association, entity, or organization that has disciplinary authority over its members or persons
associated with a member.
Board
Leadership Structure
Our
Board of Directors has the responsibility for selecting the appropriate leadership structure for the Company. In making leadership structure
determinations, the Board of Directors considers many factors, including the specific needs of the business and what is in the best interests
of the Company’s shareholders. Our current leadership structure is comprised of a combined Chairman of the Board and Chief Executive
Officer (“CEO”), Mr. Jacob D. Cohen. The Board of Directors believes that this leadership structure is the most effective
and efficient for the Company at this time. Mr. Cohen possesses detailed and in-depth knowledge of the issues, opportunities, and challenges
facing the Company, and is thus best positioned to develop agendas that ensure that the Board of Directors’ time and attention
are focused on the most critical matters. Combining the Chairman of the Board and CEO roles promotes decisive leadership, fosters clear
accountability and enhances the Company’s ability to communicate its message and strategy clearly and consistently to our shareholders,
particularly during periods of turbulent economic and industry conditions.
Risk
Oversight
Effective
risk oversight is an important priority of the Board of Directors. Because risks are considered in virtually every business decision,
the Board of Directors discusses risk throughout the year generally or in connection with specific proposed actions. The Board of Directors’
approach to risk oversight includes understanding the critical risks in the Company’s business and strategy, evaluating the Company’s
risk management processes, allocating responsibilities for risk oversight, and fostering an appropriate culture of integrity and compliance
with legal responsibilities. The directors exercise direct oversight of strategic risks to the Company.
The
Audit Committee reviews and assesses the Company’s processes to manage business and financial risk and financial reporting risk.
It also reviews the Company’s policies for risk assessment and assesses steps management has taken to control significant risks.
Other
Directorships
No
director of the Company is also a director of an issuer with a class of securities registered under Section 12 of the Exchange Act (or
which otherwise are required to file periodic reports under the Exchange Act), except for Mr. Alex Hamilton who serves as a member of
the Board of Directors, the Chairman of the Audit Committee and member of the Corporate Governance and Nominating Committee, of Addentax
Group Corp. (NASDAQ:ATXG).
Committees
of the Board
Our
Board of Directors has three standing committees: an Audit Committee, a Compensation Committee, and a Nominating and Corporate Governance
Committee.
Board
Committee Membership
Committee
membership of the Board of Directors is as follows:
| |
|
Independent |
|
Audit
Committee |
|
Compensation
Committee |
|
Nominating
and
Corporate
Governance
Committee |
| Jacob
D. Cohen(1) |
|
|
|
|
|
|
|
|
| Jonathan
Arango(2) |
|
|
|
|
|
|
|
|
| Lorraine
D’Alessio |
|
X |
|
M |
|
M |
|
C |
| Alex
P. Hamilton |
|
X |
|
C |
|
|
|
|
| Dr.
Kenny Myers |
|
X |
|
M |
|
C |
|
M |
| |
(1) |
Chairman
of Board of Directors. |
| |
C
|
Chairman
of Committee. |
| |
M
|
Member. |
| |
(2) |
Resigned
effective March 28, 2024. |
Audit
Committee
We
have established an Audit Committee of the Board of Directors. Ms. D’Alessio, Mr. Hamilton and Dr. Meyers serve as members of our
Audit Committee, and Mr. Hamilton chairs the Audit Committee. Under the Nasdaq listing standards and applicable SEC rules, we are required
to have at least three members of the Audit Committee, all of whom must be independent. The Board of Directors has determined that each
of Ms. D’Alessio, Mr. Hamilton and Dr. Meyers meet the independent director standard under Nasdaq listing standards and under Rule
10-A-3(b)(1) of the Exchange Act.
The
Board has determined that Mr. Hamilton, is an “audit committee financial expert” (as defined in the SEC rules) because he
has the following attributes: (i) an understanding of generally accepted accounting principles in the United States of America (“GAAP”)
and financial statements; (ii) the ability to assess the general application of such principles in connection with accounting for estimates,
accruals and reserves; (iii) experience analyzing and evaluating financial statements that present a breadth and level of complexity
of accounting issues that are generally comparable to the breadth and complexity of issues that can reasonably be expected to be raised
by our financial statements; (iv) an understanding of internal control over financial reporting; and (v) an understanding of Audit Committee
functions. Mr. Hamilton has acquired these attributes as a result of his significant experience serving on the Board of Directors of
various private and public companies and the Co-Founder and president of Donald Capital LLC, a FINRA registered investment banking firm.
We
have adopted an Audit Committee Charter, which details the principal functions of the Audit Committee, including:
| |
● |
the
appointment, compensation, retention, replacement, and oversight of the work of the independent registered public accounting firm
engaged by us; |
| |
|
|
| |
● |
pre-approving
all audit and permitted non-audit services to be provided by the independent registered public accounting firm engaged by us, and
establishing pre-approval policies and procedures; |
| |
|
|
| |
● |
setting
clear hiring policies for employees or former employees of the independent registered public accounting firm, including but not limited
to, as required by applicable laws and regulations; |
| |
|
|
| |
● |
setting
clear policies for audit partner rotation in compliance with applicable laws and regulations; |
| |
|
|
| |
● |
obtaining
and reviewing a report, at least annually, from the independent registered public accounting firm describing (i) the independent
registered public accounting firm’s internal quality-control procedures, (ii) any material issues raised by the most recent
internal quality-control review, or peer review, of the audit firm, or by any inquiry or investigation by governmental or professional
authorities within the preceding five years respecting one or more independent audits carried out by the firm and any steps taken
to deal with such issues and (iii) all relationships between the independent registered public accounting firm and us to assess the
independent registered public accounting firm’s independence; |
| |
|
|
| |
● |
reviewing
and approving any related party transaction required to be disclosed pursuant to Item 404 of Regulation S-K promulgated by the SEC
prior to us entering into such transaction; and |
| |
|
|
| |
● |
reviewing
with management, the independent registered public accounting firm, and our legal advisors, as appropriate, any legal, regulatory
or compliance matters, including any correspondence with regulators or government agencies and any employee complaints or published
reports that raise material issues regarding our financial statements or accounting policies and any significant changes in accounting
standards or rules promulgated by the Financial Accounting Standards Board, the SEC or other regulatory authorities. |
The
Audit Committee also has the sole authority, at its discretion and at our expense, to retain, compensate, evaluate and terminate our
independent auditors and to review, as it deems appropriate, the scope of our annual audits, our accounting policies and reporting practices,
our system of internal controls, our compliance with policies regarding business conduct and other matters. In addition, the Audit Committee
has the authority, at its discretion and at our expense, to retain special legal, accounting or other advisors to advise the Audit Committee.
Compensation
Committee and Nominating and Corporate Governance Committee
We
have established a Compensation Committee of the Board of Directors. Ms. D’Alessio and Dr. Meyers serve as members of our Compensation
Committee. Under the Nasdaq listing standards and applicable SEC rules, we are required to have at least two members of the Compensation
Committee, all of whom must be independent. Each of Ms. D’Alessio and Dr. Meyers are independent, and Dr. Meyer’s chairs
the Compensation Committee.
We
have adopted a Compensation Committee Charter, which details the principal functions of the Compensation Committee, including:
| |
● |
reviewing
and approving on an annual basis the corporate goals and objectives relevant to our Chief Executive Officer’s compensation,
if any is paid by us, evaluating our Chief Executive Officer’s performance in light of such goals and objectives and determining
and approving the remuneration (if any) of our Chief Executive Officer based on such evaluation; |
| |
|
|
| |
● |
reviewing
and approving on an annual basis the compensation, if any is paid by us, of all of our other officers; |
| |
|
|
| |
● |
reviewing
on an annual basis our executive compensation policies and plans; |
| |
● |
implementing
and administering our incentive compensation equity-based remuneration plans; |
| |
|
|
| |
● |
assisting
management in complying with our proxy statement and annual report disclosure requirements; |
| |
|
|
| |
● |
approving
all special perquisites, special cash payments and other special compensation and benefit arrangements for our officers and employees; |
| |
|
|
| |
● |
if
required, producing a report on executive compensation to be included in our annual proxy statement; and |
| |
|
|
| |
● |
reviewing,
evaluating and recommending changes, if appropriate, to the remuneration for directors. |
The
charter also provides that the Compensation Committee may, in its sole discretion, retain or obtain the advice of a compensation consultant,
legal counsel or other adviser and will be directly responsible for the appointment, compensation and oversight of the work of any such
adviser. However, before engaging or receiving advice from a compensation consultant, external legal counsel or any other adviser, the
Compensation Committee will consider the independence of each such adviser, including the factors required by Nasdaq and the SEC.
Nominations
for Directors
We
have established a Nominating and Corporate Governance Committee. The members of our nominating and corporate governance are Ms. D’Alessio
and Dr. Meyers and Ms. D’Alessio serves as chair of the Nominating and Corporate Governance Committee.
The
primary purposes of our Nominating and Corporate Governance Committee are to assist the board in:
| |
● |
identifying,
screening and reviewing individuals qualified to serve as directors and recommending to the Board of Directors candidates for nomination
for election at the annual meeting of shareholders or to fill vacancies on the Board of Directors; |
| |
|
|
| |
● |
developing,
recommending to the Board of Directors and overseeing implementation of our corporate governance guidelines; |
| |
|
|
| |
● |
coordinating
and overseeing the annual self-evaluation of the Board of Directors, its committees, individual directors and management in the governance
of the company; and |
| |
|
|
| |
● |
reviewing
on a regular basis our overall corporate governance and recommending improvements as and when necessary. |
The
Nominating and Corporate Governance Committee is governed by a charter that complies with the rules of the Nasdaq.
Our
Nominating and Corporate Governance Committee will recommend to the Board of Directors candidates for nomination for election at the
annual meeting of the shareholders. The Board of Directors will also consider director candidates recommended for nomination by our shareholders
during such times as they are seeking proposed nominees to stand for election at the next annual meeting of shareholders (or, if applicable,
a special meeting of shareholders).
We
have not formally established any specific, minimum qualifications that must be met or skills that are necessary for directors to possess.
In general, in identifying and evaluating nominees for director, the Board of Directors considers educational background, diversity of
professional experience, knowledge of our business, integrity, professional reputation, independence, wisdom, and the ability to represent
the best interests of our shareholders.
Director
Independence
Nasdaq
listing standards require that a majority of our Board of Directors be independent. An “independent director” is defined
generally as a person other than an officer or employee of the company or its subsidiaries or any other individual having a relationship
which in the opinion of the company’s Board of Directors, would interfere with the director’s exercise of independent judgment
in carrying out the responsibilities of a director. Our Board of Directors has determined that all of our directors, other than Mr. Cohen,
are “independent directors” as defined in the Nasdaq listing standards and applicable SEC rules. Our independent directors
have regularly scheduled meetings at which only independent directors are present.
In
assessing director independence, the Board considers, among other matters, the nature and extent of any business relationships, including
transactions conducted, between the Company and each director and between the Company and any organization for which one of our directors
is a director or executive officer or with which one of our directors is otherwise affiliated.
Shareholder
Communications with the Board
A
shareholder who wishes to communicate with our Board of Directors may do so by directing a written request addressed to our Secretary,
15110 N. Dallas Parkway, Suite 600, Dallas, Texas 75248, who, upon receipt of any communication other than one that is clearly marked
“Confidential,” will note the date the communication was received, open the communication, make a copy of it for our files
and promptly forward the communication to the director(s) to whom it is addressed. Upon receipt of any communication that is clearly
marked “Confidential,” our Secretary will not open the communication, but will note the date the communication was received
and promptly forward the communication to the director(s) to whom it is addressed.
Policy
on Equity Ownership
The
Company does not have a policy on equity ownership at this time.
Policy
against Hedging
The
Company recognizes that hedging against losses in Company shares may disturb the alignment between shareholders and executives that equity
awards are intended to build; however, while ‘short sales’ are discouraged by the Company, the Company does not currently
have a policy prohibiting such transactions. We plan to implement a policy prohibiting such transactions in the future.
Compensation
Recovery
On
October 26, 2023, the Board of Directors of the Company approved the adoption of a Policy for the Recovery of Erroneously Awarded Incentive
Based Compensation (the “Clawback Policy”), with an effective date of October 2, 2023, in order to comply with the
final clawback rules adopted by the Securities and Exchange Commission under Section 10D and Rule 10D-1 of the Securities Exchange Act
of 1934, as amended (“Rule 10D-1”), and the listing standards, as set forth in the Nasdaq Listing Rule 5608 (the “Final
Clawback Rules”).
The
Clawback Policy provides for the mandatory recovery of erroneously awarded incentive-based compensation from current and former executive
officers as defined in Rule 10D-1 (“Covered Officers”) of the Company in the event that the Company is required to
prepare an accounting restatement, in accordance with the Final Clawback Rules. The recovery of such compensation applies regardless
of whether a Covered Officer engaged in misconduct or otherwise caused or contributed to the requirement of an accounting restatement.
Under the Clawback Policy, the Board of Directors may recoup from the Covered Officers erroneously awarded incentive compensation received
within a lookback period of the three completed fiscal years preceding the date on which the Company is required to prepare an accounting
restatement.
Code
of Ethics
We
have adopted a Code of Ethical Business Conduct (“Code of Ethics”) that applies to all of our directors, officers and employees.
We intend to disclose any amendments to our Code of Ethics and any waivers with respect to our Code of Ethics granted to our principal
executive officer, our principal financial officer, or any of our other employees performing similar functions in a Current Report on
Form 8-K.
There
have been no waivers granted with respect to our Code of Ethics to any such officers or employees.
Whistleblower
Protection Policy
The
Company adopted a Whistleblower Protection Policy (“Whistleblower Policy”) that applies to all of its directors, officers,
employees, consultants, contractors and agents of the Company. The Whistleblower Policy has been reviewed and approved by the Board.
Board
Diversity
While
we do not have a formal policy on diversity, our Board of Directors considers diversity to include the skill set, background, reputation,
type and length of business experience of our board members as well as a particular nominee’s contributions to that mix. Our Board
of Directors believes that diversity promotes a variety of ideas, judgments and considerations to the benefit of our Company and shareholders.
On
August 6, 2021, the Securities and Exchange Commission approved a proposed rule from Nasdaq on diversity of boards of directors of companies
listed on Nasdaq. Pursuant to the rule as approved (the “Diversity Rule”), any company newly listing on The Nasdaq Capital
Market that was not previously subject to a substantially similar requirement of another national securities exchange, is required to
have, explain why it does not have, at least two Diverse (as defined below) directors by the later of: (a) two years from the date of
listing; or (b) the date the company files its proxy statement or its information statement (or, if the company does not file a proxy,
in its Form 10-K) for the company’s second annual meeting of shareholders subsequent to the company’s listing; provided that
if the company has a board of five or fewer members it need only have, or explain why it does not have, one Diverse director. Unless
exempt from the rules as discussed below, at least one Diverse director must self-identify as female and at least one Diverse director
must self-identify as an underrepresented minority or as LGBTQ+ (unless we remain as a smaller reporting company, in which case both
Diverse directors may self-identify as female). “Diverse” means an individual who self-identifies as one or more of the following:
female, LGBTQ+, or an underrepresented individual based on national, racial, ethnic, indigenous, cultural, religious or linguistic. We
currently have one director who self-identifies as female.
Delinquent
Section 16(a) Reports
Section
16(a) of the Exchange Act requires our executive officers and directors and persons who own more than 10% of a registered class of our
equity securities to file with the SEC initial statements of beneficial ownership, reports of changes in ownership and annual reports
concerning their ownership in our common stock and other equity securities, on Form 3, 4 and 5 respectively. Executive officers, directors
and greater than 10% stockholders are required by the SEC regulations to furnish our company with copies of all Section 16(a) reports
they file.
Based
solely on our review of the copies of such reports received by us and on representations by certain of our officers and directors regarding
their compliance with the applicable reporting requirements under Section 16(a) of the Exchange Act, we believe that all filings required
to be made under Section 16(a) during the twelve months ending December 31, 2023 were timely made, except that Jacob D. Cohen, our Chief
Executive Officer and Chairman failed to timely file one Form 4 and as a result three transactions were not timely reported and Amanda
Hammer, the Company’s Chief Operating Officer, failed to timely file on Form 4 and as a result two transactions were not timely
reported.
EXECUTIVE
COMPENSATION
The
following table sets forth information concerning the compensation of (i) all individuals serving as our principal executive officer
or acting in a similar capacity for the years ended December 31, 2023 and 2022 (“PEO”), regardless of compensation level;
(ii) our two most highly compensated executive officers other than the PEO who were serving as executive officers for the years ended
December 31, 2023 and 2022, if any (subject to the limitations below); and (iii) up to two additional individuals for whom disclosure
would have been provided pursuant to (ii) but for the fact that the individual was not serving as an executive officer at December 31,
2023 or 2022 (collectively, the “Named Executive Officers”).
| Name and Principal Position | |
Fiscal Year | |
Salary ($) | | |
Bonus ($) | | |
Stock Awards ($)(1) | | |
Option Awards ($)(1) | | |
All Other Compensation ($)(2) | | |
Total ($) | |
| Jacob D. Cohen | |
2023 | |
| 260,000 | | |
| - | | |
| - | | |
| 362,238 | (9) | |
| 18,000 | (10) | |
| 598,256 | |
| CEO and Chairman | |
2022 | |
| 70,000 | | |
| - | | |
| 100,000 | | |
| 462,750 | (4) | |
| - | | |
| 632,750 | |
| | |
| |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Jonathan Arango | |
2023 | |
| 120,000 | | |
| 5,000 | | |
| - | | |
| - | | |
| - | | |
| 125,000 | |
| Former President, Secretary and Director(11) | |
2022 | |
| 50,000 | | |
| - | | |
| 100,000 | | |
| 308,500 | (5) | |
| - | | |
| 458,500 | |
| | |
| |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Eugene M. Johnston | |
2023 | |
| 14,000 | | |
| - | | |
| 42,500 | (6) | |
| - | | |
| - | | |
| 56,500 | |
| CFO(3) | |
2022 | |
| - | | |
| - | | |
| 42,000 | (7) | |
| - | | |
| - | | |
| 41,763 | |
| | |
| |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Amanda Hammer | |
2023 | |
| 105,417 | | |
| - | | |
| 75,000 | (8) | |
| 149,014 | (8) | |
| - | | |
| 329,431 | |
| COO | |
| |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| |
(1) |
In
accordance with SEC rules, the amounts included in this column are the grant date fair value for awards granted in the fiscal years
shown, computed in accordance with the stock-based compensation accounting rules that are a part of generally accepted accounting
principles in effect in the United States (as set forth in Financial Accounting Standards Board’s Accounting Standards Codification
Topic 718), but excluding the effect of any estimated forfeitures of such awards. The values in this column reflect the full grant
date fair value of all equity awards granted during the year, although the awards are subject to vesting periods based on continued
employment. |
| |
|
|
| |
(2) |
Does
not include perquisites and other personal benefits or property, unless the aggregate amount of such compensation is more than $10,000.
No executive officer earned any non-equity incentive plan compensation or nonqualified deferred compensation during the periods reported
above. No executive officer serving as a director received any compensation for services on the Board of Directors separate from
the compensation paid as an executive for the periods above. |
| |
|
|
| |
(3) |
Mr.
Johnston was appointed as Chief Financial Officer of the Company effective on October 1, 2022. |
| |
(4) |
On
August 31, 2022, in consideration for agreeing to an employment agreement with the Company, Mr. Cohen received a sign-on bonus of
options to purchase 750,000 shares of common stock of the Company, with an exercise price of $1.10 per share, with options to purchase
250,000 shares vesting every 12 months that the agreement is in effect, beginning September 1, 2023. The options have a term of five
years. |
| |
|
|
| |
(5) |
On
August 31, 2022, in consideration for agreeing to an employment agreement with the Company, Mr. Arango received a sign-on bonus of
options to purchase 500,000 shares of common stock of the Company, with an exercise price of $1.10 per share, with options to purchase
166,666 shares vesting every 12 months that the agreement is in effect, beginning September 1, 2023. The options had a term of five
years, exercisable for three months following his termination of employment with the Company which occurred on March 28, 2024. |
| |
|
|
| |
(6) |
On
October 3, 2023, in consideration for agreeing to a consulting agreement with the Company, Mr. Johnston received 50,000 shares of
common stock of the Company. The shares were valued at $0.85 per share for a total of $42,500. |
| |
|
|
| |
(7) |
Effective
on October 1, 2022, the Company granted Mr. Johnston 150,000 shares of the Company’s restricted stock which vest over a 6-month
period at the rate of 25,000 shares per month with the first 25,000 shares vesting on November 1, 2022. All of the shares have been
fully vested to date. The shares were valued at $0.28 per share for a total of $42,000. |
| |
|
|
| |
(8) |
On
May 1, 2022, in consideration for agreeing to an employment agreement with the Company, Ms. Hammer received a sign-on bonus of 75,000
shares of common stock of the Company. The shares were valued at $1.00 per share for a total of $75,000. Additionally, Ms. Hammer
received options to purchase 150,000 shares of common stock of the Company, with an exercise price of $1.10 per share, with options
to purchase 50,000 shares vesting every 12 months that the agreement is in effect, beginning May 1, 2024. The options have a term
of five years. |
| |
|
|
| |
(9) |
On
December 28, 2023, in consideration for services rendered for the Company, Mr. Cohen received options to purchase 1,250,000 shares
of common stock of the Company, with an exercise price of $0.32 per share with all options being deemed vested as of the date of
grant. The options have a term of five years. |
| |
|
|
| |
(10) |
Pursuant
to Mr. Cohen’s employment agreement, Mr. Cohen is provided a car allowance of $1,500 per month for a total of $18,000. |
| |
|
|
| |
(11) |
Resigned
as an officer and director of the Company on March 28, 2024. |
Outstanding
Equity Awards at Fiscal Year-End
The
following table sets forth information as of December 31, 2023 concerning outstanding equity awards for the executive officers named
in the Summary Compensation Table.
| | |
Option Awards | | |
Stock Awards(3) | |
| Name | |
Number of securities underlying unexercised options (#) exercisable | | |
Number of securities underlying unexercised options (#) unexercisable | | |
Option Exercise price ($) | | |
Option expiration date | | |
Number of shares or units of stock that have not vested (#) | | |
Market value of shares or units of stock that have not vested ($) | |
| Jacob D. Cohen | |
| 250,000 | | |
| 500,000 | (1) | |
$ | 1.10 | | |
| 9/1/2027 | | |
| - | | |
$ | - | |
| | |
| 1,250,000 | | |
| - | | |
| 0.32 | | |
| 12/28/2028 | | |
| - | | |
| - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Jonathan Arango(4) | |
| 166,667 | | |
| 333,333 | (2) | |
$ | 1.10 | | |
| 9/1/2027 | | |
| - | | |
$ | - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Eugene M. Johnston | |
| - | | |
| - | | |
$ | - | | |
| - | | |
| - | | |
$ | - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Amanda Hammer | |
| - | | |
| 150,000 | | |
$ | 1.10 | | |
| 5/1/2033 | | |
| - | | |
$ | - | |
| |
(1) |
On
August 31, 2022, in consideration for agreeing to an employment agreement with the Company, Mr. Cohen received a sign-on bonus of
options to purchase 750,000 shares of common stock of the Company, with an exercise price of $1.10 per share, with options to purchase
250,000 shares vesting every 12 months that the agreement is in effect, beginning September 1, 2023. The options have a term of five
years. |
| |
|
|
| |
(2) |
On
August 31, 2022, in consideration for agreeing to an employment agreement with the Company, Mr. Arango received a sign-on bonus of
options to purchase 500,000 shares of common stock of the Company, with an exercise price of $1.10 per share, with options to purchase
166,666 shares vesting every 12 months that the agreement is in effect, beginning September 1, 2023. The options have a term of five
years. |
| |
|
|
| |
(3) |
Ms.
Hammer was granted options to purchase 150,000 shares of common stock of the Company in May 2023, with an exercise price of $1.10
per share, with options to purchase 50,000 shares vesting every 12 months, subject to her continued employment. |
| |
|
|
| |
(4) |
Resigned
an officer and director on March 28, 2024 and as such, all unvested options as of that date were forfieted. |
Recent
Compensation Awards
On
October 1, 2023, the Company executed a Summary of Terms and Conditions with Gene Johnston continuing his appointment as the Company’s
Chief Financial Officer on a full-time basis for a term of 12 months. Pursuant to the agreement, the Company issued Johnston 50,000 shares
of the Company’s common stock and agreed to pay him $2,000 per month. The shares were issued under, and subject to the terms of,
the Company’s 2022 Equity Incentive Plan, as amended.
On
May 1, 2023, the Company granted 150,000 options to purchase shares of common stock of the Company, under the 2022 Plan to Amanda Hammer,
the Company’s COO, related to her employment agreement. The options have an exercise price of $1.10 per share, an original life
of five years and vest at the annual renewal of their employment over three years. The options were issued under, and subject to the
terms of, the Company’s 2022 Equity Incentive Plan, as amended.
Effective
December 28, 2023, the Board of Directors, with the recommendation of the Compensation Committee of the Board of Directors, approved
the grant of stock options to purchase 1,250,000 shares of the Company’s common stock to Jacob D. Cohen, the Company’s Chief
Executive Officer and Chairman, in consideration for services rendered to the Company. The options were granted under the Company’s
2022 Equity Incentive Plan, and the options had a term of five years, subject in all cases to the terms and conditions of the 2022 Plan,
as amended, the award agreement entered into to evidence such grant, and Mr. Cohen’s continued service with the Company. The options
vested in full upon grant. The options have an exercise price of $0.32 per share, 110% of the closing sales price of the Company’s
common stock on the NASDAQ Capital market on December 28, 2023, the date the grant was approved.
Employment
Agreements
Jacob
D. Cohen, Chief Executive Officer
On
August 31, 2022, we entered into an Executive Employment Agreement with Jacob D. Cohen. The agreement, which provides for Mr. Cohen to
serve as our Chief Executive Officer, was effective September 1, 2022, and has a term extending through September 1, 2025, provided that
the agreement automatically renews for additional one-year terms thereafter in the event neither party provides the other at least 60
days prior notice of their intention not to renew the terms of the agreement.
Pursuant
to the terms of the agreement, Mr. Cohen’s annual compensation package currently includes (a) a base salary of $300,000 per year
($180,000 per year through May 1, 2023), subject to automatic annual increases of $60,000 each year the agreement is in place, and subject
to further increases as determined in the sole discretion of the Compensation Committee or the Board of Directors, and (b) a bonus payment
to be determined in the sole discretion of the Compensation Committee or the Board of Directors in an annual targeted amount of 200%
of his base salary (the “Targeted Bonus”), subject to the compliance by Mr. Cohen with performance goals that may be established
by the Compensation Committee or the Board of Directors from time to time, provided no goals have been established to date, and that
in the absence of performance goals, the amount of such bonus would be wholly determined in the discretion of the Compensation Committee
or the Board of Directors. Mr. Cohen is also paid an automobile allowance of $1,500 per month during the term of the agreement and is
eligible to participate in our stock option plan and other benefit plans.
In
consideration for agreeing to the terms of the agreement, Mr. Cohen received a sign-on bonus of options to purchase 750,000 shares of
common stock of the Company, with an exercise price of $1.10 per share, with options to purchase 250,000 shares vesting every 12 months
that the agreement is in effect. The options have a term of five years.
Mr.
Cohen’s compensation under his employment agreement may be increased from time to time, by the Compensation Committee, or the Board
of Directors (with the recommendation of the Compensation Committee), which increases do not require the entry into an amended employment
agreement. Mr. Cohen may also receive bonuses from time to time, in the discretion of the Board and/or Compensation Committee in cash,
stock, or options.
The
agreement prohibits Mr. Cohen from competing against us during the term of the agreement and for a period of 12 months after the termination
of the agreement in any state and any other geographic area in which we or our subsidiaries provide Restricted Services or Restricted
Products, directly or indirectly, during the 12 months preceding the date of the termination of the agreement. “Restricted Services”
means the or men’s wellness services and any other services and any other services that we or our subsidiaries have provided or
are researching, developing, performing and/or providing at any time during the two years immediately preceding the date of termination,
or which Mr. Cohen has obtained any trade secret or other confidential information about at any time during the two years immediately
preceding the date of termination of the agreement. “Restricted Products” branded men’s wellness products sold to consumers
via a telemedicine platform and any other product and any other product, that we or our subsidiaries have provided or are researching,
developing, manufacturing, distributing, selling and/or providing at any time during the two years immediately preceding the date the
agreement is terminated, or which Mr. Cohen obtained any trade secret or other confidential information in connection with at any time
during the two years immediately preceding the date of termination of the agreement.
We
may terminate Mr. Cohen’s employment (a) for “cause” which means (i) Mr. Cohen materially breaches any obligation,
duty, covenant or agreement under the agreement, which breach is not cured or corrected within 30 days of written notice thereof from
the Company (except for breaches of the assignment of inventions or confidentiality/non-solicitation and non-compete provisions of the
agreement, which cannot be cured and for which the Company need not give any opportunity to cure); or (ii) Mr. Cohen commits any act
of misappropriation of funds or embezzlement; or (iii) Mr. Cohen commits any act of fraud; or (iv) Mr. Cohen is convicted of, or pleads
guilty or nolo contendere with respect to, theft, fraud, a crime involving moral turpitude, or a felony under federal or applicable state
law; and, in the case of any of the above offenses, such offense casts reasonable doubt on Mr. Cohen’s ability to perform his duties
going forward; (b) in the event Mr. Cohen suffers a physical or mental disability which renders him unable to perform his duties and
obligations for either 90 consecutive days or 180 days in any 12-month period; (c) for any reason without “cause”; (d) upon
expiration of the initial term of the agreement (or any renewal) upon notice as provided above, or (e) at any time without cause. The
agreement also automatically terminates upon the death of Mr. Cohen.
Mr.
Cohen may terminate his employment (a) for “good reason” if there is (i) a material diminution in his authority, duties,
or responsibilities; (ii) a material diminution in the authority, duties, or responsibilities of the supervisor to whom Mr. Cohen is
required to report, including, if applicable, a requirement that Mr. Cohen report to an officer or employee of the Company rather than
reporting to the Board; (iii) a material breach by the Company of the agreement, or (iv) a material diminution in Mr. Cohen’s base
salary; provided, however, prior to any such termination by Mr. Cohen for “good reason,” Mr. Cohen must first advise us in
writing (within 90 days of the occurrence of such event) and provide us 30 days to cure, after which in the event we do not cure the
issue leading to such “good reason” notice, Mr. Cohen has 30 days to resign for “good reason”); (b) for any reason
without “good reason”; and (c) upon expiration of the initial term of the agreement (or any renewal) upon notice as provided
above.
If
Mr. Cohen’s employment is terminated due to his death or disability, Mr. Cohen or his estate is entitled to a lump sum cash severance
payment equal to the sum of (i) Mr. Cohen’s base salary accrued through the termination date; (ii) any unpaid cash bonus for the
prior year that would have been paid had Mr. Cohen not been terminated prior to such payment; and (iii) Mr. Cohen’s Targeted Bonus
for the year of termination multiplied by the number of days in such year preceding the termination date divided by 365. Additionally,
and notwithstanding anything to the contrary in any equity agreement, any unvested stock options or equity compensation held by Mr. Cohen
shall vest and shall be exercisable until the earlier of (x) ninety days from the date of termination and (y) the latest date upon which
such stock options or equity would have expired by their original terms under any circumstances.
If
Mr. Cohen’s employment is terminated pursuant to Mr. Cohen without “good reason” or his non-renewal of the agreement,
or by the Company with cause, Mr. Cohen is entitled to his base salary accrued through the termination date and no other benefits other
than continuation of health insurance benefits on the terms and to the extent required by COBRA, or such other similar law or regulation
as may be applicable to the Mr. Cohen or the Company with respect to the Mr. Cohen. Additionally, any unvested stock options or equity
compensation held by Mr. Cohen shall immediately terminate and be forfeited (unless otherwise provided in the applicable award) and any
previously vested stock options (or if applicable equity compensation) shall be subject to terms and conditions set forth in the applicable
equity agreement, as such may describe the rights and obligations upon termination of employment of Mr. Cohen.
If
Mr. Cohen’s employment is terminated by Mr. Cohen for “good reason” or by the Company without “cause” or
due to the Company’s non-renewal, (a) Mr. Cohen is entitled to his base salary accrued through the termination date and any unpaid
cash bonus for the prior completed calendar year that would have been paid had Mr. Cohen not been terminated prior to such payment, plus
a lump sum cash severance payment equal to the sum of (i) an amount equal to Mr. Cohen’s current annual base salary plus (ii) an
amount equal to Mr. Cohen’s Targeted Bonus for the year containing the termination date (the “Severance Payment”);
and (b) provided Mr. Cohen elects to receive continued health insurance coverage through COBRA, the Company will pay Mr. Cohen’s
monthly COBRA contributions for health insurance coverage, as may be amended from time to time (less an amount equal to the premium contribution
paid by active Company employees, if any) for 12 months following the termination date (the “Health Payment”); provided,
however, that if at any time Mr. Cohen is covered by a substantially similar level of health insurance through subsequent employment
or otherwise, the Company’s health benefit obligations shall immediately cease, and the Company shall have no further obligation
to make the Health Payment. Additionally, and notwithstanding anything to the contrary in any equity agreement, any unvested stock options
or equity compensation previously granted to the Mr. Cohen will vest immediately upon such termination and shall be exercisable by the
Mr. Cohen until the earlier of (A) ninety days from the date of termination and (B) the latest date upon which such stock options or
equity would have expired by their original terms under any circumstances.
As
a condition to Mr. Cohen’s right to receive any Severance Payment, (a) Mr. Cohen must execute and deliver to the Company a written
release in form and substance satisfactory to the Company, of any and all claims against the Company and all directors and officers of
the Company with respect to all matters arising out of Mr. Cohen’s employment, or the termination thereof (other than claims for
entitlements under the terms of the agreement or plans or programs of the Company in which Mr. Cohen has accrued a benefit); and (b)
Mr. Cohen must not have breached any of his covenants and agreements under the Agreement relating to assignment of inventions and confidentiality,
including the non-solicitation and non-compete provisions thereof, which shall continue following the Termination Date.
If
a Change of Control (as defined below) occurs during the term of the agreement, or within six months after Mr. Cohen’s termination
of employment by him for good reason or by the Company without cause or upon non-renewal, the Company is required to pay Mr. Cohen, within
60 days following the date of such Change of Control, a cash payment in a lump sum in an amount equal to (x) minus (y) where (x) equals
3.0 times the sum of (a) the current annual base salary of the Mr. Cohen; and (b) the amount of the most recent cash bonus paid to the
Mr. Cohen (collectively (a) and (b), the “Change of Control Payment”) and (y) equals the amount of any severance payment
actually paid to Mr. Cohen in connection with a non-Change of Control termination, as discussed above). In the event the Compensation
Committee has not previously made a determination regarding cash bonus or the most recent cash bonus was zero, the “amount of the
most recent cash bonus paid to the Mr. Cohen” is instead equal to “the targeted bonus for the year in which the Change in
Control occurs.” Additionally, following a change of control termination, all outstanding stock options and other equity compensation
held by Mr. Cohen are exercisable by the Mr. Cohen pursuant to the terms thereof until the earlier of (a) ninety (90) days from his termination
date and (b) the latest date upon which such stock options and other equity compensation would have expired by their original terms under
any circumstances; provided any equity awards outstanding prior to the entry into the Executive Employment Agreement continue to be governed
by the terms set forth in such award agreements.
“Change
of Control” for the purposes of the agreement means: (a) any person obtaining beneficial ownership representing more than 50% of
the total voting power represented by our then outstanding voting securities without the approval of not fewer than two-thirds of our
Board of Directors; (b) a merger or consolidation of us whether or not approved by our Board of Directors, other than a merger or consolidation
that would result in our voting securities immediately prior thereto continuing to represent at least 50% of the total voting power outstanding
immediately after such merger or consolidation, (c) our shareholders approving a plan of complete liquidation or an agreement for the
sale or disposition by us of all or substantially all of our assets, or (d) as a result of the election of members to our Board of Directors,
a majority of the Board of Directors consists of persons who are not members of the Board of Directors on September 1, 2022, except in
the event that such slate of directors is proposed by a committee of the Board of Directors.
The
agreement contains standard assignment of inventions, indemnification and confidentiality provisions. Further, Mr. Cohen is subject to
non-solicitation covenants during the term of the agreement.
Although
Mr. Cohen will be prohibited from competing with us while he is employed with us, he will only be prohibited from competing for 12 months
after his employment with us ends pursuant to the agreement. Accordingly, Mr. Cohen could be in a position to use industry experience
gained while working with us to compete with us.
Jonathan
Arango, Former President and Secretary
On
August 31, 2022, we entered into an Executive Employment Agreement with Jonathan Arango. The agreement, which provided for Mr. Arango
to serve as our President (which role he ceased serving in March 2024) and Chief Operating Officer (which role he ceased serving as in
May 2023) and Secretary, was effective September 1, 2022, and had a term extending through September 1, 2025.
Pursuant
to the terms of the agreement, Mr. Arango’s annual compensation package included (1) a base salary of $120,000 per year, subject
to annual increases of $30,000, each year the agreement is in place, and subject to further increases as determined in the sole discretion
of the Compensation Committee or the Board of Directors, and (2) a bonus payment to be determined in the sole discretion of the Compensation
Committee or the Board of Directors in an annual targeted amount of 200% of his base salary, subject to the compliance by Mr. Arango
with performance goals that may be established by the Compensation Committee or the Board of Directors from time to time, provided no
goals have been established to date, and that in the absence of performance goals, the amount of such bonus would be wholly determined
in the discretion of the Compensation Committee or the Board of Directors. Mr. Arango is also paid an automobile allowance of $1,000
per month during the term of the agreement and is eligible to participate in our stock option plan and other benefit plans.
Mr.
Arango resigned as an officer and director of the Company on March 28, 2024.
Although
Mr. Arango was prohibited from competing with us while he is employed with us, he will only be prohibited from competing for 12 months
after his employment with us ends pursuant to the agreement. Accordingly, Mr. Arango could be in a position to use industry experience
gained while working with us to compete with us.
Eugene
M. Johnston, Chief Financial Officer
On
October 1, 2022, the Company entered into an offer letter with Eugene M. Johnston (the “Offer Letter”). The Offer Letter
provided for Mr. Johnston to serve as the full-time Chief Financial Officer of the Company, reporting to the Company’s Board of
Directors and Chief Executive Officer, for a term of 12 months from October 1, 2022 to September 30, 2023. Pursuant to the Offer Letter,
the Company agreed to grant Mr. Johnston 150,000 shares of the Company’s restricted stock which vested over a 6-month period at
the rate of 25,000 shares per month with the first 25,000 shares vesting on November 1, 2022. Pursuant to the Offer Letter, Mr. Johnston
is eligible to participate in any of the Company’s future sponsored benefit plans, including but not limited to, health insurance
benefits, 401k, stock option or restricted stock grants, and other fringe benefits, once established, and no earlier than the first of
the month following 105 days of Johnston’s start date. Mr. Johnston is also eligible to receive equity incentive grants or cash
bonus awards as determined by the Company’s Board (or a committee of the Board) in their sole discretion from time to time. The
shares were valued at $0.28 per share for a total of $41,763.
On
October 1, 2023, the Company executed a Summary of Terms and Conditions with Mr. Johnston continuing his appointment as the Company’s
Chief Financial Officer on a full-time basis for a term of 12 months, through October 1, 2024. Pursuant to the agreement, the Company
issued Mr. Johnston 50,000 shares of the Company’s common stock and agreed to pay him $2,000 per month. The shares were issued
under, and subject to the terms of, the Company’s 2022 Equity Incentive Plan.
Amanda
Hammer, Chief Operating Officer
On
and effective on May 1, 2023, we entered into an Employment Agreement with Mrs. Amanda Hammer. The Employment Agreement provides for
Mrs. Hammer to serve as Chief Operating Officer of the Company for an initial three-year term extending through May 1, 2026, provided
that the agreement automatically renews for additional one-year terms thereafter in the event neither party provides the other at least
60 days prior notice of their intention not to renew the terms of the agreement.
The
agreement provides for Mrs. Hammer to receive an annual salary of $150,000 per year (the “Base Salary”). The Employment Agreement
also required the Company to grant Mrs. Hammer a sign-on bonus of (a) 75,000 shares of common stock of the Company, vested in full upon
issuance, and (b) options to purchase an additional 150,000 shares of common stock of the Company, with an exercise price of the greater
of (i) $1.10 per share; and (ii) the closing sales price of the Company’s common stock on the Nasdaq Capital Market on the date
the Employment Agreement and the grant is approved by the Board (which date was May 1, 2023), and which exercise price was $1.10 per
share, with options to purchase 50,000 shares vesting every 12 months that the Employment Agreement is in effect, subject to the terms
of the Company’s 2022 Equity Incentive Plan, as amended. The options are exercisable for a period of ten years and are documented
by a separate option agreement entered into by the Company and Mrs. Hammer (the “Option Agreement”).
Pursuant
to the terms of the Employment Agreement, Mrs. Hammer’s annual compensation package includes (1) a Base Salary (described above),
subject to increases from time to time in the determination of the Compensation Committee of the Board (or the Board with the recommendation
of the Compensation Committee), and (2) a discretionary bonus payment to be determined in the sole discretion of the Compensation Committee
or the Board of Directors in the targeted amount of 100% of her Base Salary (the “Cash Bonus”). Mrs. Hammer is also eligible
for discretionary equity bonuses and/or cash awards, from time to time in the discretion of the Compensation Committee and/or Board of
Directors.
Mrs.
Hammer’s compensation under her employment agreement may be increased from time to time, by the Compensation Committee, or the
Board of Directors (with the recommendation of the Compensation Committee), which increases do not require the entry into an amended
employment agreement.
The
Employment Agreement prohibits Mrs. Hammer from competing against us during the term of the agreement and for a period of 12 months after
the termination of the agreement in any state and any other geographic area in which we or our subsidiaries provide Restricted Services
or Restricted Products, directly or indirectly, during the 12 months preceding the date of the termination of the agreement. “Restricted
Products” means any product that the Company or any of its subsidiaries has provided or is developing, manufacturing, distributing,
selling and/or providing at any time during the term of the Agreement, or which she obtained any trade secret or other confidential information
about at any time during the term, or which she became aware of as a result of services rendered under the Employment Agreement. “Restricted
Services” means any services that the Company or any of its subsidiaries has provided or is developing, performing and/or providing
at any time during the term of the agreement, or which she obtained any trade secret or other confidential information about at any time
during the term, or which she became aware of as a result of services rendered under the Employment Agreement. The non-compete requirements
described in the paragraph above, as well as the restriction on Mrs. Hammer to refrain, for a period of 12 months from the termination
date, from soliciting customers of the Company with whom Mrs. Hammer worked during the last year of Mrs. Hammer’s employment with
the Company and from soliciting employees of the Company to leave the employment of the Company, are defined as the “Non-Compete
Provisions”.
We
may terminate Mrs. Hammer’s Employment Agreement (a) for “cause” which means (i) that Mrs. Hammer has materially breached
any obligation, duty, covenant or agreement under the agreement, which breach is not cured or corrected within 30 days of written notice
thereof from the Company (except for breaches of the assignment of inventions or confidentiality/non-solicitation and non-compete provisions
of the agreement, which cannot be cured and for which the Company need not give any opportunity to cure); (ii) Mrs. Hammer commits any
act of misappropriation of funds or embezzlement; (iii) Mrs. Hammer commits any act of fraud; or (iv) Mrs. Hammer is convicted of, or
pleads guilty or nolo contendere with respect to, theft, fraud, a crime involving moral turpitude, or a felony under federal or applicable
state law; (b) in the event Mrs. Hammer suffers a physical or mental disability which renders him unable to perform her duties and obligations
for either 90 consecutive days or 180 days in any 12-month period; (c) for any reason without “cause”; or (d) upon expiration
of the initial term of the agreement (or any renewal) upon notice as provided above. The agreement also automatically terminates upon
the death of Mrs. Hammer.
Mrs.
Hammer may terminate her employment (a) for “good reason” if there is (i) a material diminution in her authority, duties,
or responsibilities; (ii) a material diminution in the authority, duties, or responsibilities or a requirement that Mrs. Hammer report
to an officer or employee of the Company rather than reporting to the Board; (iii) a material breach by the Company of the agreement,
or (iv) a material diminution in Mrs. Hammer’s Base Salary, in each case without her prior written consent; provided, however,
prior to any such termination by Mrs. Hammer for “good reason,” Mrs. Hammer must first advise us in writing (within 30 days
of the occurrence of such event) and provide us 30 days to cure (5 days in the event the event results to a reduction in her salary),
after which in the event we do not cure the issue leading to such “good reason” notice, Mrs. Hammer has 30 days to resign
for “good reason”); (b) for any reason without “good reason”; and (c) upon expiration of the initial term of
the agreement (or any renewal) upon notice as provided above.
If
Mrs. Hammer’s employment is terminated due to her death or disability, Mrs. Hammer or her estate is entitled to a lump sum cash
severance payment equal to the sum of (i) Mrs. Hammer’s Base Salary accrued through the termination date; (ii) any unpaid Cash
Bonus for the prior year that would have been paid had Mrs. Hammer not been terminated prior to such payment; and (iii) the pro rata
amount of the current year’s targeted bonus, multiplied by the number of days in such year preceding the termination date divided
by 365. Additionally, and notwithstanding anything to the contrary in any equity agreement, any unvested stock options or equity compensation
held by Mrs. Hammer upon such termination shall vest and shall be exercisable until the earlier of (A) ninety days from the date of termination
and (B) the latest date upon which such stock options or equity would have expired by their original terms under any circumstances.
If
Mrs. Hammer’s employment is terminated by Mrs. Hammer without “good reason” or her non-renewal of the agreement, or
by non-renewal by the Company, by the Company with cause or the Company’s non-renewal of the agreement, Mrs. Hammer is entitled
to her Base Salary accrued through the termination date and no other benefits other than continuation of health insurance benefits on
the terms and to the extent required by COBRA, or such other similar law or regulation as may be applicable to Mrs. Hammer or the Company
with respect to Mrs. Hammer. Additionally, any unvested stock options or equity compensation held by Mrs. Hammer shall immediately terminate
and be forfeited (unless otherwise provided in the applicable award) and any previously vested stock options (or if applicable equity
compensation) shall be subject to terms and conditions set forth in the applicable equity agreement, as such may describe the rights
and obligations upon termination of employment of Mrs. Hammer.
If
Mrs. Hammer’s employment is terminated by Mrs. Hammer for “good reason”, or by the Company without “cause”,
(a) Mrs. Hammer is entitled to her Base Salary accrued through the termination date and any unpaid Cash Bonus for the prior completed
calendar year that would have been paid had Mrs. Hammer not been terminated prior to such payment, plus a lump sum cash severance payment
equal to (x) the sum of (i) an amount equal to her current annual Base Salary; plus (ii) an amount equal to her targeted bonus for the
year containing the termination date, multiplied by (y) a fraction, (A) the numerator of which shall equal the Severance Months (defined
below), and (B) the denominator of which is 12 (the “Severance Payment”); and (b) provided Mrs. Hammer elects to receive
continued health insurance coverage through COBRA, the Company will pay Mrs. Hammer’s monthly COBRA contributions for health insurance
coverage, as may be amended from time to time (less an amount equal to the premium contribution paid by active Company employees, if
any) for the Severance Months following the termination date (the “Health Payment”); provided, however, that if at any time
Mrs. Hammer is covered by a substantially similar level of health insurance through subsequent employment or otherwise, the Company’s
health benefit obligations shall immediately cease, and the Company shall have no further obligation to make the Health Payment. Additionally,
and notwithstanding anything to the contrary in any equity agreement, any unvested stock options or equity compensation previously granted
to Mrs. Hammer will vest immediately upon such termination and shall be exercisable by Mrs. Hammer until the earlier of (A) ninety (90)
days from the date of termination and (B) the latest date upon which such stock options or equity would have expired by their original
terms under any circumstances, provided that such provisions shall not affect any equity awards outstanding prior to the date of the
Employment Agreement.
As
a condition to Mrs. Hammer’s right to receive any Severance Payment, (A) Mrs. Hammer must execute and deliver to the Company a
written release in form and substance satisfactory to the Company, of any and all claims against the Company and all directors and officers
of the Company with respect to all matters arising out of Mrs. Hammer’s employment, or the termination thereof (other than claims
for entitlements under the terms of the agreement or plans or programs of the Company in which Mrs. Hammer has accrued a benefit), which
must be effective by the 60th day following her termination date; and (B) Mrs. Hammer must not have breached any of her covenants and
agreements under the Agreement relating to assignment of inventions and confidentiality, including the non-solicitation and non-compete
provisions thereof, which shall continue following the termination date.
“Severance
Months” means (a) three, in the event the period of time between the effective date and the termination date is less than
one year; (b) six, in the event the period of time between the effective date and the termination date is one year or more, but
less than two years; (c) nine, in the event the period of time between the effective date and the termination date is two years
or more, but less than three years; and (d) twelve, in the event the period of time between the effective date and the termination
date is more than three years.
The
Employment Agreement also contains standard assignment of inventions, indemnification and confidentiality provisions. Further, Mrs. Hammer
is subject to non-solicitation covenants during the term of the agreement.
Although
Mrs. Hammer will be prohibited from competing with us while she is employed with us, she will only be prohibited from competing for twelve
months after her employment with us ends pursuant to her employment agreement. Accordingly, Mrs. Hammer could be in a position to use
industry experience gained while working with us to compete with us.
Compensation
of Directors
The
following table sets forth compensation information with respect to our non-executive directors during our fiscal year ended December
31, 2023. The compensation of our executive directors is included above under “Executive Compensation Table.”
| Name | |
Fees Earned or
Paid in Cash ($)* | | |
Stock Awards
($) (1) (2)(3) | | |
All Other Compensation ($) | | |
Total ($) | |
| Lorraine D’Alessio | |
$ | - | | |
$ | - | | |
$ | - | | |
$ | - | |
| Alex P. Hamilton | |
$ | - | | |
$ | - | | |
$ | - | | |
$ | - | |
| Dr. Kenny Myers | |
$ | - | | |
$ | - | | |
$ | - | | |
$ | - | |
*
The table above does not include the amount of any expense reimbursements paid to the above directors. No directors received any Non-Equity
Incentive Plan Compensation or Nonqualified Deferred Compensation. Does not include perquisites and other personal benefits, or property,
unless the aggregate amount of such compensation is more than $10,000.
| |
(1) |
In
accordance with SEC rules, the amounts included in this column are the grant date fair value for awards granted in the fiscal years
shown, computed in accordance with the stock-based compensation accounting rules that are a part of generally accepted accounting
principles in effect in the United States (as set forth in Financial Accounting Standards Board’s Accounting Standards Codification
Topic 718), but excluding the effect of any estimated forfeitures of such awards. The values in this column reflect the full grant
date fair value of all equity awards granted during the year, although the awards are subject to vesting periods based on continued
employment. |
| |
(2) |
No
specific board compensation policy has been adopted to date; however, on October 14, 2022, we entered into offer letters with each
of our three independent non-executive directors, Ms. D’Alessio, Mr. Hamilton and Dr. Meyers. Pursuant to the Offer Letters,
each non-executive director agreed to serve as a member of our Board of Directors, and we agreed to grant each non-executive director
75,000 shares of restricted common stock (the “Director Shares”). The Director Shares were issued under the Company’s
2022 Equity Incentive Plan, as amended (the “Plan”), with the following vesting schedule: 1/3 of the Director Shares
vested on October 14, 2022, and the remaining Director Shares vest annually in two increments on each of October 14, 2023 (vested)
and 2024, subject to such directors continuing to provide services to the Company on such dates, and subject to the Restricted Stock
Award agreements entered into in order to evidence such grants. The shares were valued at $0.28 per share for a total of $72,039. |
| |
(3) |
The
aggregate number of unvested shares of restricted common stock held by each non-employee director listed above as of December 31,
2023 was as follows: |
| Name | |
Unvested Restricted Stock Shares (#) | |
| Lorraine D’Alessio | |
| 25,000 | |
| Alex P. Hamilton | |
| 25,000 | |
| Dr. Kenny Myers | |
| 25,000 | |
No
specific board compensation policy has been adopted to date, however, we expect that our non-executive directors will be granted equity
compensation and paid cash, from time to time, for their services on the Board of Directors.
Key
Man Insurance
Hold
key man life insurance in the aggregate amount of $2,000,000 on the life of Jacob D. Cohen, the Chief Executive Officer of the Company.
2022
Equity Incentive Plan
On
August 31, 2022, the Board of Directors and our majority shareholders adopted the Company’s 2022 Equity Incentive Plan, which was
amended by the Board of Directors on February 26, 2024, subject to stockholder approval, and ratified by the stockholders on March 25,
2024 (as amended, the “2022 Plan”).
The
2022 Plan provides an opportunity for any employee, officer, director or consultant of the Company, subject to limitations provided by
federal or state securities laws, to receive (i) incentive stock options (to eligible employees only); (ii) nonqualified stock options;
(iii) stock appreciation rights; (iv) restricted stock awards; (v) restricted stock units; (vi) shares in performance of services; (vii)
other awards of equity or equity based compensation; or (viii) any combination of the foregoing. In making such determinations, the Board
may take into account the nature of the services rendered by such person, his or her present and potential contribution to the Company’s
success, and such other factors as the Board in its discretion shall deem relevant.
Shares
Available Under the 2022 Plan; Evergreen Provision
Subject
to adjustment in connection with the payment of a stock dividend, a stock split or subdivision or combination of the shares of common
stock, or a reorganization or reclassification of the Company’s common stock, the aggregate number of shares of common stock which
may be issued pursuant to awards under the 2022 Plan is currently the sum of (i) 10,000,000, and (ii) an automatic increase on April
1st of each year for a period of nine years commencing on April 1, 2024 and ending on (and including) April 1, 2032, in an amount equal
to the lesser of (x) ten percent (10%) of the total shares of common stock of the Company outstanding on the last day of the immediately
preceding fiscal year; and (y) 2,000,000 shares of common stock; provided, however, that the Board may act prior to April 1st of a given
year to provide that the increase for such year will be a lesser number of shares of common stock. This is also known as an “evergreen”
provision. Notwithstanding the foregoing, no more than a total of 26,000,000 shares of common stock (or awards) may be issued or granted
under the 2022 Plan in aggregate, and no more than 26,000,000 shares of common stock may be issued pursuant to the exercise of Incentive
Stock Options.
If
an award granted under the 2022 Plan entitles a holder to receive or purchase shares of our common stock, then on the date of grant of
the award, the number of shares covered by the award (or to which the award relates) will be counted against the total number of shares
available for granting awards under the 2022 Plan. As a result, the shares available for granting future awards under the 2022 Plan will
be reduced as of the date of grant. However, certain shares that have been counted against the total number of shares authorized under
the 2022 Plan in connection with awards previously granted under such 2022 Plan will again be available for awards under the 2022 Plan
as follows: shares of our common stock covered by an award or to which an award relates which were not issued because the award terminated
or was paid in cash or any portion thereof that was forfeited or cancelled without the delivery of shares will again be available for
awards, including, but not limited to shares forfeited to pay any exercise price or tax obligation.
In
addition, shares of common stock related to awards that expire, are forfeited or cancelled or terminate for any reason without the issuance
of shares shall not be treated as issued pursuant to the 2022 Plan.
The
shares available for awards under the 2022 Plan will be authorized but unissued shares of our common stock or shares acquired in the
open market or otherwise.
Administration
The
Company is the issuer (manager) of the 2022 Plan. The 2022 Plan is administered by either (a) the entire Board of Directors of the Company,
or (b) the Compensation Committee; or (b) as determined from time to time by the Board of Directors (the “Administrator”).
Subject to the terms of the 2022 Plan, the Administrator may determine the recipients, the types of awards to be granted, the number
of shares of our common stock subject to or the cash value of awards, and the terms and conditions of awards granted under the 2022 Plan,
including the period of their exercisability and vesting. The Administrator also has the authority to provide for accelerated exercisability
and vesting of awards. Subject to the limitations set forth below, the Administrator also determines the fair market value applicable
to an award and the exercise or strike price of stock options and stock appreciation rights granted under the 2022 Plan.
The
Administrator may also delegate to one or more executive officers the authority to designate employees who are not executive officers
to be recipients of certain awards and the number of shares of our common stock subject to such awards. Under any such delegation, the
Administrator will specify the total number of shares of our common stock that may be subject to the awards granted by such executive
officer. The executive officer may not grant an award to himself or herself.
On
or after the date of grant of an award under the 2022 Plan, the Administrator may (i) accelerate the date on which any such award becomes
vested, exercisable or transferable, as the case may be, (ii) extend the term of any such award, including, without limitation, extending
the period following a termination of a participant’s employment during which any such award may remain outstanding, or (iii) waive
any conditions to the vesting, exercisability or transferability, as the case may be, of any such award; provided, that the Administrator
shall not have any such authority to the extent that the grant of such authority would cause any tax to become due under Section 409A
of the Internal Revenue Code (the “Code”).
Eligibility
All
of our employees (including our affiliates), non-employee directors and consultants are eligible to participate in the 2022 Plan and
may receive all types of awards other than incentive stock options. Incentive stock options may be granted under the 2022 Plan only to
our employees (including our affiliates).
No
awards are issuable by the Company under the 2022 Plan (a) in connection with services associated with the offer or sale of securities
in a capital-raising transaction; or (b) where the services directly or indirectly promote or maintain a market for the Company’s
securities.
Limit
on Non-Employee Director Compensation
The
maximum number of shares subject to awards granted during a single calendar year to any non-employee director, taken together with any
cash fees paid during the compensation year to the non-employee director, in respect of the director’s service as a member of the
Board during such year (including service as a member or chair of any committees of the Board), will not exceed $500,000, or $1,000,000
in the first year such non-employee director is appointed to the Board, or in the case of any non-employee chairperson of the Board,
in total value (calculating the value of any such awards based on the grant date fair value of such awards for financial reporting purposes).
Compensation will count towards this limit for the fiscal year in which it was granted or earned, and not later when distributed, in
the event it is deferred.
Option
Terms
Stock
options may be granted by the Administrator and may be either non-qualified (non-statutory) stock options or incentive stock options.
The Administrator, in its sole discretion, determines the exercise price of any options granted under the Plan which exercise price is
set forth in the agreement evidencing the option, provided however that at no time can the exercise price be less than the $0.0001 par
value per share of the Company’s common stock. Stock options are subject to the terms and conditions, including vesting conditions,
set by the Administrator (and incentive stock options are subject to further statutory restrictions that will be set forth in the grant
agreement for those options). The exercise price for all stock options granted under the 2022 Plan will be determined by the Administrator,
except that no stock options can be granted with an exercise price that is less than 100% of the fair market value of the Company’s
common stock on the date of grant. Further, shareholders who own greater than 10% of the Company’s voting stock will not be granted
incentive stock options that have an exercise price less than 110% of the fair market value of the Company’s common stock on the
date of grant.
The
term of all stock options granted under the 2022 Plan will be determined by the Administrator, but the term of an incentive stock option
may not exceed 10 years (five years for incentive stock options granted to shareholders who own greater than 10% of the Company’s
voting stock). Each stock option gives the grantee the right to receive a number of shares of the Company’s common stock upon exercise
of the stock option and payment of the exercise price. The exercise price may be paid in cash or if approved by the Administrator, shares
of the Company’s common stock. The Administrator may also permit other ways for a grantee to pay the exercise price.
Options
granted under the 2022 Plan may be exercisable in cumulative increments, or “vest,” as determined by the Administrator.
Incentive
stock options granted under the 2022 Plan are intended to qualify as “incentive stock options” within the meaning of Section
422 of the Internal Revenue Code of 1986, as amended, which we refer to as the Code. Nonqualified (non-statutory stock options) granted
under the 2022 Plan are not intended to qualify as incentive stock options under the Code.
The
Administrator may impose limitations on the transferability of stock options granted under the 2022 Plan in its discretion. Generally,
a participant may not transfer a stock option granted under the 2022 Plan other than by will or the laws of descent and distribution
or, subject to approval by the Administrator, pursuant to a domestic relations order. However, the Administrator may permit transfer
of a stock option in a manner that is not prohibited by applicable tax and securities laws. Options may not be transferred to a third
party financial institution for value.
Unless
the terms of an optionholder’s stock option agreement, or other written agreement between us and the optionholder, provide otherwise,
if an optionholder’s service relationship with us or any of our affiliates ceases for any reason other than disability, death,
or cause, the optionholder may generally exercise any vested options for a period of three months following the cessation of service.
This period may be extended in the event that exercise of the option is prohibited by applicable securities laws or the immediate sale
of shares acquired upon exercise of the option is prohibited by our insider trading policy. If an optionholder’s service relationship
with us or any of our affiliates ceases due to death, or an optionholder dies within a certain period following cessation of service,
the optionholder or a beneficiary may generally exercise any vested options for a period of 18 months following the date of death. If
an optionholder’s service relationship with us or any of our affiliates ceases due to disability, the optionholder may generally
exercise any vested options for a period of 12 months following the cessation of service. In the event of a termination for cause, options
generally terminate upon the termination date. In no event may an option be exercised beyond the expiration of its term. Acceptable consideration
for the purchase of common stock issued upon the exercise of a stock option will be determined by the administrator and may include (i)
cash, check, bank draft or money order; (ii) a broker-assisted cashless exercise; (iii) the tender of shares of our common stock previously
owned by the optionholder; (iv) a net exercise of the option (to the extent allowed); or (v) other legal consideration approved by the
administrator.
Except
as explicitly provided otherwise in a participant’s stock option agreement or other written agreement with us or one of our affiliates,
the term “cause” is defined in the 2022 Plan to mean any event which would qualify as cause for termination under the participant’s
employment agreement with the Company, or, if there is no such employment agreement, any of the following (i) the recipient’s dishonest
statements or acts with respect to the Company or any affiliate of the Company, or any current or prospective customers, suppliers, vendors
or other third parties with which such entity does business; (ii) the recipient’s commission of (A) a felony or (B) any misdemeanor
involving moral turpitude, deceit, dishonesty or fraud; (iii) the recipient’s failure to perform the recipient’s assigned
duties and responsibilities to the reasonable satisfaction of the Company which failure continues, in the reasonable judgment of the
Company, after written notice given to the recipient by the Company; (iv) the recipient’s gross negligence, willful misconduct
or insubordination with respect to the Company or any affiliate of the Company; or (v) the recipient’s material violation of any
provision of any agreement(s) between the recipient and the Company relating to noncompetition, non-solicitation, nondisclosure and/or
assignment of inventions.
Restricted
Stock Unit Awards
Restricted
stock unit (RSU) awards are granted under restricted stock unit award agreements adopted by the administrator. Restricted stock unit
awards may be granted in consideration for any form of legal consideration that may be acceptable to our Board of Directors and permissible
under applicable law. A restricted stock unit award may be settled by cash, delivery of stock, a combination of cash and stock as deemed
appropriate by the administrator, or in any other form of consideration set forth in the restricted stock unit award agreement. Additionally,
dividend equivalents may be credited in respect of shares covered by a restricted stock unit award. Except as otherwise provided in the
applicable award agreement, or other written agreement between us and the recipient, restricted stock unit awards that have not vested
will be forfeited once the participant’s continuous service ends for any reason.
Restricted
Stock Awards
Restricted
stock awards are granted under restricted stock award agreements adopted by the administrator. A restricted stock award may be awarded
in consideration for cash, check, bank draft or money order, past or future services to us, or any other form of legal consideration
that may be acceptable to our Board of Directors and permissible under applicable law. The administrator determines the terms and conditions
of restricted stock awards, including vesting and forfeiture terms. If a participant’s service relationship with us ends for any
reason, we may receive any or all of the shares of common stock held by the participant that have not vested as of the date the participant
terminates service with us through a forfeiture condition or a repurchase right.
Stock
Appreciation Rights
Stock
appreciation rights are granted under stock appreciation right agreements adopted by the administrator. The administrator determines
the purchase price or strike price for a stock appreciation right, which generally will not be less than 100% of the fair market value
of our common stock on the date of grant. A stock appreciation right granted under our 2022 Plan will vest at the rate specified in the
stock appreciation right agreement as determined by the administrator. Stock appreciation rights may be settled in cash or shares of
our common stock or in any other form of payment as determined by our Board of Directors and specified in the stock appreciation right
agreement.
The
administrator determines the term of stock appreciation rights granted under our 2022 Plan, up to a maximum of 10 years. If a participant’s
service relationship with us or any of our affiliates ceases for any reason other than cause, disability, or death, the participant may
generally exercise any vested stock appreciation right for a period of three months following the cessation of service. This period may
be further extended in the event that exercise of the stock appreciation right following such a termination of service is prohibited
by applicable securities laws. If a participant’s service relationship with us, or any of our affiliates, ceases due to disability
or death, or a participant dies within a certain period following cessation of service, the participant or a beneficiary may generally
exercise any vested stock appreciation right for a period of 12 months in the event of disability and 18 months in the event of death.
In the event of a termination for cause, stock appreciation rights generally terminate upon the termination date. In no event may a stock
appreciation right be exercised beyond the expiration of its term.
Performance
Awards
Our
2022 Plan permits the grant of performance awards that may be settled in stock, cash or other property. Performance awards may be structured
so that the stock or cash will be issued or paid only following the achievement of certain pre-established performance goals during a
designated performance period. Performance awards that are settled in cash or other property are not required to be valued in whole or
in part by reference to, or otherwise based on, our common stock.
The
performance goals may be based on any measure of performance selected by our Board of Directors. The performance goals may be based on
company-wide performance or performance of one or more business units, divisions, affiliates, or business segments, and may be either
absolute or relative to the performance of one or more comparable companies or the performance of one or more relevant indices. Unless
specified otherwise by our Board of Directors at the time the performance award is granted, our Board of Directors will appropriately
make adjustments in the method of calculating the attainment of performance goals as follows: (i) to exclude restructuring and/or other
nonrecurring charges; (ii) to exclude exchange rate effects; (iii) to exclude the effects of changes to generally accepted accounting
principles; (iv) to exclude the effects of any statutory adjustments to corporate tax rates; (v) to exclude the effects of items that
are “unusual” in nature or occur “infrequently” as determined under generally accepted accounting principles;
(vi) to exclude the dilutive effects of acquisitions or joint ventures; (vii) to assume that any business divested by us achieved performance
objectives at targeted levels during the balance of a performance period following such divestiture; (viii) to exclude the effect of
any change in the outstanding shares of our common stock by reason of any stock dividend or split, stock repurchase, reorganization,
recapitalization, merger, consolidation, spin-off, combination or exchange of shares or other similar corporate change, or any distributions
to common shareholders other than regular cash dividends; (ix) to exclude the effects of stock based compensation and the award of bonuses
under our bonus plans; (x) to exclude costs incurred in connection with potential acquisitions or divestitures that are required to be
expensed under generally accepted accounting principles; and (xi) to exclude the goodwill and intangible asset impairment charges that
are required to be recorded under generally accepted accounting principles.
Other
Stock Awards
The
administrator may grant other awards based in whole or in part by reference to our common stock. The administrator will set the number
of shares under the stock award (or cash equivalent) and all other terms and conditions of such awards.
Tax
Withholding Adjustments
To
the extent provided by the terms of an option or other award, or otherwise agreed to by the Administrator, a participant may satisfy
any federal, state or local tax withholding obligation relating to the exercise of such option, or award by a cash payment upon exercise,
or in the discretion of the Administrator, by authorizing our company to withhold a portion of the stock otherwise issuable to the participant,
by delivering already-owned shares of our common stock or by a combination of these means.
Changes
to Capital Structure
In
the event there is a specified type of change in our capital structure, such as a stock split, reverse stock split, or recapitalization,
appropriate adjustments will be made to (i) the class and maximum number of shares reserved for issuance under our 2022 Plan, (ii) the
class and maximum number of shares by which the share reserve may increase automatically each year, (iii) the class and maximum number
of shares that may be issued on the exercise of ISOs, and (iv) the class and number of shares and exercise price, strike price, or purchase
price, if applicable, of all outstanding stock awards.
Corporate
Transactions
In
the event of a corporate transaction (as defined in the 2022 Plan), unless otherwise provided in a participant’s stock award agreement
or other written agreement with us or one of our affiliates or unless otherwise expressly provided by the administrator at the time of
grant, any stock awards outstanding under our 2022 Plan may be assumed, continued or substituted for by any surviving or acquiring corporation
(or its parent company), and any reacquisition or repurchase rights held by us with respect to the stock award may be assigned to the
successor (or its parent company). If the surviving or acquiring corporation (or its parent company) does not assume, continue or substitute
for such stock awards, then (i) with respect to any such stock awards that are held by participants whose continuous service has not
terminated prior to the effective time of the corporate transaction, or current participants, the vesting (and exercisability, if applicable)
of such stock awards will be accelerated in full (or, in the case of performance awards with multiple vesting levels depending on the
level of performance, vesting will accelerate at 100% of the target level) to a date prior to the effective time of the corporate transaction
(contingent upon the effectiveness of the corporate transaction), and such stock awards will terminate if not exercised (if applicable)
at or prior to the effective time of the corporate transaction, and any reacquisition or repurchase rights held by us with respect to
such stock awards will lapse (contingent upon the effectiveness of the corporate transaction); and (ii) any such stock awards that are
held by persons other than current participants will terminate if not exercised (if applicable) prior to the effective time of the corporate
transaction, except that any reacquisition or repurchase rights held by us with respect to such stock awards will not terminate and may
continue to be exercised notwithstanding the corporate transaction.
In
the event a stock award will terminate if not exercised prior to the effective time of a corporate transaction, the administrator may
provide, in its sole discretion, that the holder of such stock award may not exercise such stock award but instead will receive a payment
equal in value to the excess (if any) of (i) the value of the property the participant would have received upon the exercise of the stock
award, over (ii) any per share exercise price payable by such holder, if applicable. In addition, any escrow, holdback, earn out or similar
provisions in the definitive agreement for the corporate transaction may apply to such payment to the same extent and in the same manner
as such provisions apply to the holders of our common stock.
Change
in Control
Stock
awards granted under our 2022 Plan may be subject to acceleration of vesting and exercisability upon or after a change in control (as
defined in the 2022 Plan) as may be provided in the applicable stock award agreement or in any other written agreement between us or
any affiliate and the participant, but in the absence of such provision, no such acceleration will automatically occur.
Repricing;
Cancellation and Re-Grant of Stock Options or Stock Appreciation Rights
The
Administrator has the right to effect, at any time and from time to time, subject to the consent of any participant whose award is materially
impaired by such action, (1) the reduction of the exercise price (or strike price) of any outstanding option or SAR; (2) the cancellation
of any outstanding option or SAR and the grant in substitution therefor of (A) a new option, SAR, restricted stock award, RSU award or
other award, under the 2022 Plan or another equity plan of the Company, covering the same or a different number of shares of common stock,
(B) cash and/or (C) other valuable consideration (as determined by the Board); or (3) any other action that is treated as a repricing
under generally accepted accounting principles.
Duration;
Termination of the 2022 Plan
Our
Board of Directors has the authority to amend, suspend, or terminate our 2022 Plan at any time, provided that such action does not materially
impair the existing rights of any participant without such participant’s written consent. Certain material amendments also require
the approval of our shareholders. No incentive stock options may be granted after the tenth anniversary of the date our Board of Directors
adopted our 2022 Plan. No stock awards may be granted under our 2022 Plan while it is suspended or after it is terminated.
Current
Available Shares
As
of the date of his prospectus, an aggregate of 368,250 shares are available for awards under the 2022 Plan, which allows for an aggregate
of 4,168,250 total awards thereunder.
Certain
Relationships and Related Party Transactions
Except
as discussed below or otherwise disclosed above under “Executive Compensation”, in this “Certain Relationships and
Related Transactions, and Director Independence” section, the following sets forth a summary of all transactions since January
1, 2022, or any currently proposed transaction, in which the Company was to be a participant and the amount involved exceeded or exceeds
the lesser of $120,000 or one percent of the average of the Company’s total assets at December 31, 2023 or 2022, and in which any
officer, director, or any stockholder owning greater than five percent (5%) of our outstanding voting shares, nor any member of the above
referenced individual’s immediate family, had or will have a direct or indirect material interest (other than compensation described
above under “Executive Compensation”). We believe the terms obtained or consideration that we paid or received, as applicable,
in connection with the transactions described below were comparable to terms available or the amounts that would be paid or received,
as applicable, in arm’s-length transactions.
Related
Party Transactions
Issuances
and Sales of Securities
On
April 6, 2022, the Company issued 1,000,000 shares of restricted common stock each to Mr. Cohen (the Company’s Chairman, Chief
Executive Officer, Director and majority shareholder) and Mr. Jonathan Arango (the Company’s then President, then Chief Operating
Officer, then Secretary, then Director and greater than 5% shareholder), in consideration for services rendered as the Chief Executive
Officer and President and then Chief Operating Officer, respectively, of the Company. The shares were valued at $0.10 per share or a
total of $100,000.
On
June 22, 2022, the Company issued 250,000 shares of restricted common stock to The Loev Law Firm, PC, in consideration for legal services
to be rendered, which vested upon issuance. David M. Loev, the Managing Partner, President and sole owner of The Loev Law Firm, PC, is
the brother-in-law of Jacob D. Cohen, our Chairman and Chief Executive Officer. These shares were valued at $0.10 per share or a total
of $25,000.
On
June 16, 2022, American International entered into and closed the transactions contemplated by a Stock Purchase Agreement (the “SPA”),
with Cohen Enterprises, Inc. (“Cohen Enterprises”), which entity is owned by Jacob D. Cohen, the Chairman and Chief Executive
Officer of the Company, who is also the majority shareholder of the Company. Pursuant to the SPA, American International sold 8,000,000
shares of the outstanding common stock of the Company which represented 80% of the then outstanding shares of common stock of the Company,
to Cohen Enterprises in consideration for $90,000, which was approximately the same amount that had been advanced to the Company from
American International through the date of the SPA ($89,200). Cohen Enterprises also acquired the right to be repaid the $89,200 advanced
from American International to the Company, from the Company, pursuant to the terms of the SPA. As a result of the closing of the SPA,
Cohen Enterprises increased its ownership of the Company to 90% (with the remaining 10% of the Company then being owned by Mr. Arango,
as discussed above), and American International completely divested its interest in the Company.
In
June 2022, Cohen Enterprises sold an aggregate of 600,000 shares of our restricted common stock to third parties for $0.10 per share
or $60,000 in aggregate and 40,000 shares of our restricted common stock to a third party for $0.25 per share or $10,000 in aggregate.
The shares were sold in private transactions to accredited investors.
On
June 30, 2022, Cohen Enterprises gifted 360,000 restricted shares of common stock to Isaak Cohen, the father of Jacob D. Cohen. These
shares were valued at $0.10 per share or $36,000.
On
August 31, 2022, in consideration for agreeing to an employment agreement with the Company, Mr. Cohen received a sign-on bonus of options
to purchase 750,000 shares of common stock of the Company, with an exercise price of $1.10 per share, with options to purchase 250,000
shares vesting every 12 months that the agreement is in effect, beginning September 1, 2023. The options have a term of five years. The
fair value of the 750,000 options on the grant date was $462,750 and as of December 31, 2022, the Company recognized $51,417 as stock-based
compensation.
On
August 31, 2022, in consideration for agreeing to an employment agreement with the Company, Mr. Arango received a sign-on bonus of options
to purchase 500,000 shares of common stock of the Company, with an exercise price of $1.10 per share, with options to purchase 166,666
shares vesting every 12 months that the agreement is in effect, beginning September 1, 2023. The options have a term of five years. The
fair value of the 500,000 options on the grant date was $308,500 and as of December 31, 2022, the Company recognized $30,850 as stock-based
compensation.
On
October 1, 2022, the Company agreed to grant Eugene M. Johnston, its Chief Financial Officer, 150,000 shares of the Company’s restricted
stock which vest over a 6-month period at the rate of 25,000 shares per month with the first 25,000 shares vesting on November 1, 2022.
The shares were valued at $0.28 per share for a total of $41,763.
On
October 14, 2022, the Company issued 75,000 restricted shares of common stock to each of its three independent directors, which shares
vested 1/3 on October 14, 2022, with the remaining shares vesting in one-third increments on each of October 14, 2023 and 2024, subject
to such directors continuing to provide services to the Company on such dates, and subject to the Restricted Stock Award agreements entered
into in order to evidence such grants. These shares were valued at $0.28 per share or a total of $20,881.
On
October 14, 2022, the Company issued its Project Manager, Joan Arango, 25,000 shares of restricted common stock under the Plan. The shares
were issued to Ms. Arango as a bonus for services rendered to date. Ms. Arango is the sister of the Company’s then President and
then Chief Operating Officer, then Secretary and then Director, Jonathan Arango. The shares were valued at $0.28 per share for a total
of $7,204.
Effective
May 1, 2023, the Board of Directors of the Company, with Mr. Cohen abstaining, with the recommendation of the Compensation Committee
of the Board of Directors of the Company, approved an increase in the annual salary of Mr. Jacob Cohen, the Chief Executive Officer and
Chairman of the Company, from $180,000 to $300,000 per year.
On
and effective on May 1, 2023, the Company entered into an Employment Agreement with Mrs. Amanda Hammer. The Employment Agreement provides
for Mrs. Hammer to serve as Chief Operating Officer of the Company for an initial three-year term extending through May 1, 2026, provided
that the agreement automatically renews for additional one-year terms thereafter in the event neither party provides the other at least
60 days prior notice of their intention not to renew the terms of the agreement. The agreement provides for Mrs. Hammer to receive an
annual salary of $150,000 per year. The Employment Agreement also required the Company to grant Mrs. Hammer a sign-on bonus of (a) 75,000
restricted shares of common stock of the Company, vested in full upon issuance, and (b) options to purchase an additional 150,000 shares
of common stock of the Company, under the Company’s 2022 Equity Incentive Plan, as amended, with an exercise price of the greater
of (i) $1.10 per share; and (ii) the closing sales price of the Company’s common stock on the Nasdaq Capital Market on the date
the Employment Agreement and the grant is approved by the Board (which date was May 1, 2023), and which exercise price was $1.00 per
share, with options to purchase 50,000 shares vesting every twelve months that the Employment Agreement is in effect, subject to the
terms of the 2022 Plan. The options are exercisable for a period of ten years and are documented by a separate option agreement entered
into by the Company and Mrs. Hammer.
On
October 1, 2023, the Company executed a Summary of Terms and Conditions with Gene Johnston continuing his appointment as the Company’s
Chief Financial Officer on a full-time basis for a term of 12 months. Pursuant to the agreement, the Company issued Mr. Johnston 50,000
shares of the Company’s common stock and agreed to pay him $2,000 per month. The shares were issued under, and subject to the terms
of, the Company’s 2022 Equity Incentive Plan, as amended.
Effective
December 28, 2023, the Board of Directors, with the recommendation of the Compensation Committee of the Board of Directors, approved
the grant of stock options to purchase 1,250,000 shares of the Company’s common stock to Jacob D. Cohen, the Company’s Chief
Executive Officer and Chairman, in consideration for services rendered to the Company. The options were granted under the Company’s
2022 Equity Incentive Plan, as amended, and the options had a term of five years, subject in all cases to the terms and conditions of
the 2022 Plan, the award agreement entered into to evidence such grant, and Mr. Cohen’s continued service with the Company. The
options vested in full upon grant. The options have an exercise price of $0.32 per share, 110% of the closing sales price of the Company’s
common stock on the NASDAQ Capital market on December 28, 2023, the date the grant was approved.
Related
Party Agreements
On
September 1, 2022, and effective on August 30, 2022, we entered into a Master Services Agreement with Epiq Scripts, LLC (“Epiq
Scripts”), 51% owned and controlled by Jacob D. Cohen, our Chairman and Chief Executive Officer. Pursuant to the Master Services
Agreement and a related statement of work (“SOW”), Epiq Scripts agreed to provide for the online fulfillment, specialty compounding,
packaging, shipping, dispensing and distribution (collectively, the “Services”) of products sold exclusively via our website
that may be prescribed as part of a telehealth consultation on our platform. Epiq Scripts also agreed to provide mail service pharmacy
services to us on an exclusive basis during the term of the SOW. The Master Services Agreement and SOW are described in greater detail
above under “Business-Material Agreements-Master Services Agreement with Epiq Scripts” and “-First Amendment to MSA”.
We
paid Epiq Scripts a total of $60,000 upon our entry into the Master Services Agreement, comprising $45,000 as a one-time non-refundable
technology systems setup and implementation fee and $15,000 as an upfront retainer to be credited towards the future provision of pharmacy
and related services as outlined and detailed in the Master Services Agreement and SOW, of which $11,745 remained outstanding as of December
31, 2022 and $60,953 remained outstanding as of December 31, 2023. All costs related to the pharmacy services provided by Epiq Scripts
are listed as related party costs of revenues on our statement of operations.
On
August 31, 2022, Mr. Peter “Casey” Jensen, who was then a member of the Board of Directors of American International, purchased
25,000 units in our private placement, including 25,000 shares of common stock and warrants to purchase 25,000 shares of common stock
with an exercise price of $1.00 per share, for $25,000.
On
September 6, 2022, we entered into a Consulting Agreement with PHX Global, LLC, which is owned by Mr. Jensen. The Consulting Agreement
is described in greater detail above under “Business-Material Agreements-Consulting Agreements.”
On
January 24, 2023, we entered into Consulting Agreements with four consultants to the Company: (1) Sultan Haroon; (2) John Helfrich; (3)
Justin Baker; and (4) Maja Matthews, each of whom is also an employee of Epiq Scripts. The Consulting Agreements are described in greater
detail above under “Business-Material Agreements-Consulting Agreements.”
On
February 15, 2023, the 51% of Epiq Scripts then owned by American International was transferred to Mr. Cohen as part of an exchange transaction,
whereby Mr. Cohen agreed to cancel his preferred stock of American International, which provided him voting control over American International,
in exchange for among other assets, American International’s ownership of Epiq Scripts. As a result, Epiq Scripts is currently
51% owned by Mr. Cohen, our Chairman and Chief Executive Officer. Mr. Cohen has served as the co-Manager of Epiq Scripts since January
2022.
Related
Party Loans and Advances
On
December 10, 2021 and March 18, 2022, the Company received advances of $39,200 and $50,000, respectively, for a total of $89,200 from
its previous majority shareholder, American International, in order to cover various general and administrative expenses. The amount
owed to American International was $39,200 as of December 31, 2021. Imputed interest equal to 8% per annum, or $181, was recorded against
the related party advance as of December 31, 2021. Other than the imputed interest discussed above, the advances bear no interest and
are due on demand upon the Company’s ability to repay the advances from either future revenues or investment proceeds. Pursuant
to the terms of the June 16, 2022, Securities Purchase Agreement discussed above, on June 16, 2022, Cohen Enterprises also acquired the
right to be repaid the $89,200 advanced from American International to the Company. As of December 31, 2022, the total unpaid amount
of the advance totaled $89,200 and as of September 31, 2023, the amount had been repaid in full.
On
June 29, 2022, the Company received an advance of $25,000 from Cohen Enterprises in order to cover various general and administrative
expenses. The Company repaid Cohen Enterprises $25,000 on August 18, 2022, bringing the total amount owed to Cohen Enterprises to $89,200
as of December 31, 2022. The Company paid Cohen Enterprises $89,200 on April 4, 2023, bringing the total amount owed to Cohen Enterprises
to $0 as of December 31, 2023. The Company further recorded a credit of $6,473 towards imputed interest, as other income (previously
calculated at a rate of 8% per annum) against the related party advances for the year ended December 31, 2023.
On
December 10, 2021, the Company received an advance of $70 from ZipDoctor, Inc., a wholly owned subsidiary of its then sole shareholder,
American International, which was used to open and establish the Company’s bank account. The advance bears no interest and is due
on demand upon the Company’s ability to repay the advance from either future revenues or investment proceeds. The amount owed to
ZipDoctor was $70 as of December 31, 2021. Imputed interest equal to 8% per annum, or $0, was recorded against the related party advance
as of December 31, 2021. The amount was paid in full on May 24, 2022 and the amount owed to ZipDoctor was $0 as of December 31, 2022.
The
Company’s Chairman and Chief Executive Officer, Jacob D. Cohen, has made his personal credit card available for purchases on behalf
of the Company to cover various general and administrative expenses. Mr. Cohen has been repaid a total of $746,581 as of the date of
this prospectus for Company purchases made on his personal credit card.
On
November 18, 2022, the Company entered into a Secured Installment Promissory Note with a vendor for the purchase of equipment in the
amount of $78,260. The note bears no interest unless an event of default occurs, and then it bears interest at the rate of 10% per annum
until paid in full. The Note Payable was payable in installments, requiring payments of $5,000 on each of January 1, 2023, February 1,
2023, and March 1, 2023, with a $31,630 payment due on April 1, 2023 and a final payment due on May 1, 2023. The January 1 and March
1, 2023 payments were timely made and on March 23, 2023, the Company elected to pay off the remaining balance of $63,260. The outstanding
balance on December 31, 2022 was $78,260 and as of December 31, 2023, was $0. The outstanding balance on December 31, 2022 was $78,260
and on December 31, 2023, was $0.
On
March 1, 2024, the Company borrowed $37,500 from Ronin Equity Partners, which is owned and controlled by Jacob D. Cohen, the Company’s
Chief Executive Officer and Chairman. The amount borrowed is payable on demand and does not accrue interest.
On
March 18, 2024, the Company borrowed $50,000 from Cohen Enterprises, Inc., which is owned and controlled by Jacob D. Cohen, the Company’s
Chief Executive Officer and Chairman. The amount borrowed is payable on demand and does not accrue interest.
On
April 1, 2024, the Company borrowed $100,000 from Cohen Enterprises, Inc., which is owned and controlled by Jacob D. Cohen, the Company’s
Chief Executive Officer and Chairman. The amount borrowed is payable on demand and does not accrue interest.
Review,
Approval and Ratification of Related Party Transactions
Given
our small size and limited financial resources, we have not adopted formal policies and procedures for the review, approval or ratification
of transactions, such as those described above, with our executive officers, directors and significant shareholders. However, all of
the transactions described above were approved and ratified by our directors. In connection with the approval of the transactions described
above, our directors took into account various factors, including his fiduciary duty to the Company; the relationships of the related
parties described above to the Company; the material facts underlying each transaction; the anticipated benefits to the Company and related
costs associated with such benefits; whether comparable products or services were available; and the terms the Company could receive
from an unrelated third party.
Our
Audit Committee is tasked with reviewing related party transactions to determine whether such transactions are fair to the Company and
its shareholders. The Audit Committee of the Board of Directors of the Company will also review and approve any issues relating to conflicts
of interests and all related party transactions of the Company (“Related Party Transactions”). The Audit Committee, in undertaking
such review and will analyze the following factors, in addition to any other factors the Audit Committee deems appropriate, in determining
whether to approve a Related Party Transaction: (1) the fairness of the terms for the Company (including fairness from a financial point
of view); (2) the materiality of the transaction; (3) bids / terms for such transaction from unrelated parties; (4) the structure of
the transaction; (5) the policies, rules and regulations of the U.S. federal and state securities laws; (6) the policies of the Committee;
and (7) interests of each related party in the transaction.
The
Audit Committee will only approve a Related Party Transaction if the Audit Committee determines that the terms of the Related Party Transaction
are beneficial and fair (including fair from a financial point of view) to the Company and are lawful under the laws of the United States.
In the event multiple members of the Audit Committee are deemed a related party, the Related Party Transaction will be considered by
the disinterested members of the Board of Directors in place of the Committee.
In
addition, our Code of Business Conduct and Ethics (described above under “Management-Code of Ethics”), which is applicable
to all of our employees, officers and directors, requires that all employees, officers and directors avoid any conflict, or the appearance
of a conflict, between an individual’s personal interests and our interests.
Director
Independence
Our
common stock is currently quoted on the Nasdaq Capital Market. Nasdaq requires that a majority of our Board of Directors be independent.
Our Board of Directors has determined that each of Lorraine D’Alessio , Alex P. Hamilton and Dr. Kenny Myers is an independent
director as defined under the Nasdaq rules governing members of boards of directors and as defined under Rule 10A-3 of the Exchange Act.
In
assessing director independence, the Board considers, among other matters, the nature and extent of any business relationships, including
transactions conducted, between the Company and each director and between the Company and any organization for which one of our directors
is a director or executive officer or with which one of our directors is otherwise affiliated.
Furthermore,
the Board has determined that each of the members of our Audit Committee, Compensation Committee, and Nominating and Corporate Governance
Committee, is independent within the meaning of Nasdaq director independence standards applicable to members of such committees, as currently
in effect.
The
Compensation Committee members also qualify as “non-employee directors” within the meaning of Section 16 of the Exchange
Act.
SELLING
STOCKHOLDERS
The
shares of common stock being offered by the selling stockholders consists of those previously issued to the selling stockholders and
those issuable to the selling stockholders upon conversion of the Series B Preferred Stock, the exercise of the Warrants, and the sale
of shares pursuant to the ELOC. When we refer to the “selling stockholders” in this prospectus, we mean the persons
listed in the table below, and the pledgees, donees, transferees, assignees, successors, designees and others who later come to hold
any of the selling stockholders’ interest in the common stock other than through a public sale.
The
table below lists the selling stockholders and other information regarding the beneficial ownership of the shares of common stock by
the selling stockholders. The second column lists the number of shares of Common Stock beneficially owned by the selling stockholders,
based on its ownership of the shares of common stock as of April 23, 2024.
The
third column lists the shares of common stock being offered by this prospectus by the selling stockholders.
The
fourth column assumes the sale of all of the shares offered by the selling stockholders pursuant to this prospectus.
We
cannot advise you as to whether the selling stockholders will in fact sell any or all of such shares of common stock. In addition, the
selling stockholders may sell, transfer or otherwise dispose of, at any time and from time to time, the shares of common stock in transactions
exempt from the registration requirements of the Securities Act after the date of this prospectus. For purposes of this table, we have
assumed that the selling stockholders will have sold all of the securities covered by this prospectus upon the completion of the offering.
The
selling stockholders may sell all, some or none of their shares in this offering. See “Plan of Distribution.”
| Name of Selling Stockholder | |
Number of Shares
of Common Stock
Beneficially Owned
Prior to Offering | | |
Shares of Common
Stock Offered Hereby | | |
Number of Shares
of Common Stock
Owned After Offering | | |
% | |
| Brian Freifeld (1) | |
| 1,000,000 | (2) | |
| 30,014,286 | (3) | |
| 0 | (4) | |
| * | |
| | |
| 200,000 | | |
| 200,000 | (3) | |
| 0 | (4) | |
| * | |
| |
(1) |
The
securities are held by Platinum Point Capital LLC. Platinum Point Capital LLC is managed by Brian Freifeld, who may be deemed to
have beneficial ownership.. |
| |
|
|
| |
(2) |
Consists
of (i) one million (1,000,000) shares issued as a commitment fee in connection with the Company and the selling stockholder’s
entrance into the ELOC; (ii) ten million (10,000,000) shares issuable under the ELOC using an adjusted price of $0.26
for the per share purchase price (based on a $2.6 million total purchase price divided by the average closing price of the common
stock on the Nasdaq Capital Market for the three trading days ending on April 19, 2024 ($0.29) minus a ten percent (10%) discount
for the purchase price of $0.26), (iii) three million three hundred thousand (3,300,000) shares issuable upon exercise of warrants
issued at an exercise price of $0.26 per share and (iv) 15,714,286 shares issuable upon conversion of 500 shares of Series B Preferred
Stock with each share having a stated value of $1,100 and having an assumed conversion price of the floor price of $0.035. |
Each
time a selling stockholder sells any shares of common stock offered by this prospectus, it is required to provide you with this prospectus
and the related prospectus supplement, if any, containing specific information about the selling stockholder and the terms of the shares
of common stock being offered in the manner required by the Securities Act.
No
offer or sale may occur unless the registration statement that includes this prospectus has been declared effective by the SEC and remains
effective at the time the selling stockholders offer or sell shares of common stock. We are required, under certain circumstances, to
update, supplement or amend this prospectus to reflect material developments in our business, financial position and results of operations
and may do so by an amendment to this prospectus or a prospectus supplement.
DESCRIPTION
OF CAPITAL STOCK
The
following summary is a description of the material terms of our capital stock and is not complete. You should also refer to the Mangoceuticals,
Inc. Certificate of Formation, as amended and Bylaws, which are included as exhibits to the registration statement of which this prospectus
forms a part, and the applicable provisions of the Texas Business Organizations Code.
Authorized
Capitalization
The
total number of authorized shares of our common stock is 200,000,000 shares, $0.0001 par value per share. The total number of “blank
check” authorized shares of our preferred stock is 10,000,000 shares, $0.0001 par value per share. There are 6,000 shares of Series
B Preferred Stock authorized for issuance and 650 shares of Series B Preferred Stock outstanding. There are 6,250,000 shares
of Series C Preferred Stock authorized for issuance and 980,000 shares of Series C Preferred Stock outstanding.
Common
Stock
Voting
Rights. Each share of our common stock is entitled to one vote on all shareholder matters. Shares of our common stock do not
possess any cumulative voting rights.
Except
for the election of directors, if a quorum is present, an action on a matter is approved if it receives the affirmative vote of the holders
of the majority of the shares entitled to vote on, and who voted for, against, or expressly abstained with respect to, the matter at
a shareholders’ meeting of a corporation at which a quorum is present is the act of the shareholders, unless otherwise required
by applicable law. The election of directors is determined by a plurality of the votes cast in respect of the shares present in person
or represented by proxy at the meeting and entitled to vote, meaning that the nominees with the greatest number of votes cast, even if
less than a majority, will be elected. The rights, preferences and privileges of holders of common stock are subject to, and may be impacted
by, the rights of the holders of shares of any series of preferred stock that we have designated, or may designate and issue in the future.
Dividend
Rights. Each share of our common stock is entitled to equal dividends and distributions per share with respect to the common
stock when, as and if declared by our Board of Directors, subject to any preferential or other rights of any outstanding preferred stock.
Liquidation
and Dissolution Rights. Upon liquidation, dissolution or winding up, our common stock will be entitled to receive pro rata on
a share-for-share basis, the assets available for distribution to the shareholders after payment of liabilities and payment of preferential
and other amounts, if any, payable on any outstanding preferred stock.
No
Preemptive, Conversion, or Redemption Rights. Holders of our outstanding common stock have no preemptive, conversion, or redemption
rights. Shares of our common stock are not assessable. To the extent that additional shares of our common stock may be issued in the
future, the relative interests of the then existing shareholders may be diluted.
Fully
Paid Status. All outstanding shares of the Company’s common stock are validly issued, fully paid and non-assessable.
Preferred
Stock
Our
Board of Directors has the authority to issue undesignated shares of “blank check” preferred stock in one or more series
and to fix the designation, relative powers, preferences and rights and qualifications, limitations or restrictions of all shares of
each such series, including, without limitation, dividend rates, conversion rights, voting rights, redemption and sinking fund provisions,
liquidation preferences and the number of shares constituting each such series, without any further vote or action by the stockholders.
The issuance of additional preferred stock could decrease the amount of earnings and assets available for distribution to holders of
our common stock or adversely affect the rights and powers, including voting rights, of the holders of our common stock and could, among
other things, have the effect of delaying, deferring or preventing a change in control of our company without further action by the stockholders.
There
are 6,000 shares of Series B Preferred Stock authorized for issuance and 650 shares of Series B Preferred Stock outstanding. For
a description of the Series B Preferred Stock’s rights, see the section entitled “Description of the Series B Convertible
Preferred Stock” beginning on page 15 of this prospectus. There are 6,250,000 shares of Series C Preferred Stock authorized
for issuance and 980,000 shares of Series C Preferred Stock outstanding. For a description of the Series C Preferred Stock’s rights,
see the section entitled “Description of the 6% Series C Convertible Cumulative Preferred Stock” beginning on page
12 of this prospectus.
Business
Combinations under Texas Law
A
number of provisions of Texas law, our Certificate of Formation, as amended, and Bylaws could make it more difficult for the acquisition
of our company by means of a tender offer, a proxy contest or otherwise and the removal of incumbent officers and directors. These provisions
are intended to discourage coercive takeover practices and inadequate takeover bids and to encourage persons seeking to acquire control
of our company to negotiate first with our Board of Directors.
Title
2, Chapter 21, Subchapter M of the Texas Business Organizations Code (the “Texas Business Combination Law”) provides that
a Texas corporation may not engage in specified types of business combinations, including mergers, consolidations and asset sales, with
a person, or an affiliate or associate of that person, who is an “affiliated shareholder,” for a period of three years from
the date that person became an affiliated shareholder, subject to certain exceptions (described below). An “affiliated shareholder”
is generally defined as the holder of 20% or more of the corporation’s voting shares. The law’s prohibitions do not apply
if the business combination or the acquisition of shares by the affiliated shareholder was approved by the Board of Directors of the
corporation before the affiliated shareholder became an affiliated shareholder; or the business combination was approved by the affirmative
vote of the holders of at least two-thirds of the outstanding voting shares of the corporation not beneficially owned by the affiliated
shareholder, at a meeting of shareholders called for that purpose, not less than six months after the affiliated shareholder became an
affiliated shareholder.
This
law applies to Texas corporations which have more than 100 of record shareholders and which have not affirmatively elected to not be
governed by such law, we do not have more than 100 record shareholders and are not considered an “issuing public corporation”
for purposes of this law. Separately, we have elected in our Certificate of Formation, as amended, to not be governed by the Texas Business
Corporation Law. Notwithstanding the above, the Texas Business Combination Law does not apply to the following:
| ● |
the
business combination of an issuing public corporation: where the corporation’s original charter or bylaws contain a provision
expressly electing not to be governed by the Texas Business Combination Law; or that adopts an amendment to its charter or bylaws,
by the affirmative vote of the holders, other than affiliated shareholders, of at least two-thirds of the outstanding voting shares
of the corporation, expressly electing not to be governed by the Texas Business Combination Law and so long as the amendment does
not take effect for 18 months following the date of the vote and does not apply to a business combination with an affiliated shareholder
who became affiliated on or before the effective date of the amendment; |
| |
|
| ● |
a
business combination of an issuing public corporation with an affiliated shareholder that became an affiliated shareholder inadvertently,
if the affiliated shareholder divests itself, as soon as possible, of enough shares to no longer be an affiliated shareholder and
would not at any time within the three-year period preceding the announcement of the business combination have been an affiliated
shareholder but for the inadvertent acquisition; |
| |
|
| ● |
a
business combination with an affiliated shareholder who became an affiliated shareholder through a transfer of shares by will or
intestacy and continuously was an affiliated shareholder until the announcement date of the business combination; or |
| |
|
| ● |
a
business combination of a corporation with its wholly owned Texas subsidiary if the subsidiary is not an affiliate or associate of
the affiliated shareholder other than by reason of the affiliated shareholder’s beneficial ownership of voting shares of the
corporation. |
As
discussed above, our Certificate of Formation, as amended, contains a provision expressly providing that we are not subject to the Texas
Business Combination Law.
Anti-Takeover
Provisions of Our Charter Documents
Our
Certificate of Formation, as amended, and Bylaws contain various provisions intended to promote the stability of our shareholder base
and render more difficult certain unsolicited or hostile attempts to take us over, that could disrupt us, divert the attention of our
directors, officers and employees and adversely affect the independence and integrity of our business. These provisions include:
| ● |
Special
Meetings of Shareholders - Our Bylaws provide that special meetings of the shareholders may only be called by our Chairman, our
President, or upon written notice to our Board of Directors by our shareholders holding not less than 30% of our outstanding voting
capital stock. |
| ● |
Bylaws
- Our Bylaws may be amended by our Board of Directors alone. |
| |
|
| ● |
Advance
Notice Procedures - Our Bylaws establish an advance notice procedure for shareholder proposals to be brought before an annual
meeting of our shareholders. At an annual meeting, our shareholders elect a Board of Directors and transact such other business as
may properly be brought before the meeting. By contrast, at a special meeting, our shareholders may transact only the business for
the purposes specified in the notice of the meeting. |
| |
|
| ● |
No
cumulative voting - Our Certificate of Formation, as amended, and Bylaws do not include a provision for cumulative voting in
the election of directors. |
| |
|
| ● |
Vacancies
- Our Bylaws provide that vacancies on our Board may be filled by a majority of directors in office, although less than a quorum,
and not by the shareholders. |
| |
|
| ● |
Preferred
Stock - Our Certificate of Formation, as amended, allows us to issue up to 10,000,000 shares of preferred stock. The undesignated
preferred stock may have rights senior to those of the common stock and that otherwise could adversely affect the rights and powers,
including voting rights, of the holders of common stock. In some circumstances, this issuance could have the effect of decreasing
the market price of the common stock as well as having an anti-takeover effect. |
| |
|
| ● |
Authorized
but Unissued Shares - Our Board of Directors may cause us to issue our authorized but unissued shares of common stock in the
future without shareholders’ approval. These additional shares may be utilized for a variety of corporate purposes, including
future public offerings to raise additional capital, corporate acquisitions and employee benefit plans. The existence of authorized
but unissued shares of common stock could render more difficult or discourage an attempt to obtain control of a majority of our common
stock by means of a proxy contest, tender offer, merger or otherwise. |
| |
|
| ● |
Action
by Written Consent - Any action required or permitted to be taken by our common shareholders may be effected by written consent
of the shareholders having not less than the minimum percentage of the vote required by the Texas Business Organizations Code for
the proposed corporate action. |
| |
|
| ● |
Majority
Vote - Under the Texas Business Organizations Code, the affirmative vote of the holders of at least two-thirds of the outstanding
voting shares is typically required to approve certain fundamental transactions, including amending a company’s Certificate
of Formation, approving mergers and conversions, and winding up the corporation; however, in compliance with the Texas Business Organizations
Code, our Certificate of Formation, as amended, provides that all such fundamental transactions may be approved by majority vote
of shareholders. |
Warrants
and Options
As
of the date of this offering, we have options to purchase 1,158,333 shares of common stock outstanding, which have an exercise
price of $1.10 per share and a term through September 1, 2027; warrants to purchase 975,500 shares of common stock outstanding, which
have an exercise price of $1.00 per share and terms ending between August 16, 2027 and December 22, 2027; warrants to purchase 87,500
shares of common stock with an exercise price of $5.00 per share, which are exercisable until September 20, 2028; warrants to purchase
322,000 shares of common stock with an exercise price of $0.375 per share, which are exercisable until January 20, 2029, and warrants
to purchase 3,300,000 shares of common stock with an exercise price of $0.26 per share, which are exercisable until October 4, 2029.
Limitations
on Liability and Indemnification of Officers and Directors
As
authorized by Chapter 8 of the Texas Business Organizations Code, we may indemnify our officers and directors (and our former officers
and directors) against expenses incurred by such persons in connection with any (A) threatened, pending, or completed action or other
proceeding, whether civil, criminal, administrative, arbitrative, or investigative; (B) an appeal of an action or proceeding described
by (A); and (C) an inquiry or investigation that could lead to an action or proceeding described by (A), involving such persons in their
capacities as officers and directors, if it is determined in accordance with the Texas Business Organizations Code that: (1) the person:
(A) acted in good faith; (B) reasonably believed: (i) in the case of conduct in the person’s official capacity, that the person’s
conduct was in the enterprise’s best interests; and (ii) in any other case, that the person’s conduct was not opposed to
the enterprise’s best interests; and (C) in the case of a criminal proceeding, did not have a reasonable cause to believe the person’s
conduct was unlawful; (2) with respect to expenses, the amount of expenses other than a judgment is reasonable; and (3) indemnification
should be paid.
Under
Texas law, corporations may also purchase and maintain insurance or make other financial arrangements on behalf of any person who is
or was a director or officer (or is serving at our request as a director or officer of another corporation) for any liability asserted
against such person and any expenses incurred by him in his capacity as a director or officer.
Additionally,
our Bylaws (“Bylaws”), state that we shall indemnify every (i) present or former director, advisory director or officer of
us, (ii) any person who while serving in any of the capacities referred to in clause (i) served at our request as a director, officer,
partner, venturer, proprietor, trustee, employee, agent or similar functionary of another foreign or domestic corporation, partnership,
joint venture, trust, employee benefit plan or other enterprise, and (iii) any person nominated or designated by (or pursuant to authority
granted by) the Board of Directors or any committee thereof to serve in any of the capacities referred to in clauses (i) or (ii) (each
an “Indemnitee”).
Our
Bylaws provide that we shall indemnify an Indemnitee against all judgments, penalties (including excise and similar taxes), fines, amounts
paid in settlement and reasonable expenses actually incurred by the Indemnitee in connection with any proceeding in which he was, is
or is threatened to be named as a defendant or respondent, or in which he was or is a witness without being named a defendant or respondent,
by reason, in whole or in part, of his serving or having served, or having been nominated or designated to serve, if it is determined
that the Indemnitee (a) conducted himself in good faith, (b) reasonably believed, in the case of conduct in his official capacity, that
his conduct was in our best interests and, in all other cases, that his conduct was at least not opposed to our best interests, and (c)
in the case of any criminal proceeding, had no reasonable cause to believe that his conduct was unlawful; provided, however, that in
the event that an Indemnitee is found liable to us or is found liable on the basis that personal benefit was improperly received by the
Indemnitee, the indemnification (i) is limited to reasonable expenses actually incurred by the Indemnitee in connection with the proceeding
and (ii) shall not be made in respect of any proceeding in which the Indemnitee shall have been found liable for willful or intentional
misconduct in the performance of his duty to us.
Except
as provided above, the Bylaws provide that no indemnification shall be made in respect to any proceeding in which such Indemnitee has
been (a) found liable on the basis that personal benefit was improperly received by him, whether or not the benefit resulted from an
action taken in the Indemnitee’s official capacity, or (b) found liable to us. The termination of any proceeding by judgment, order,
settlement or conviction, or on a plea of nolo contendere or its equivalent, is not of itself determinative that the Indemnitee did not
meet the requirements set forth in clauses (a) or (b) above. An Indemnitee shall be deemed to have been found liable in respect of any
claim, issue or matter only after the Indemnitee shall have been so adjudged by a court of competent jurisdiction after exhaustion of
all appeals therefrom. Reasonable expenses shall include, without limitation, all court costs and all fees and disbursements of attorneys’
fees for the Indemnitee. The indemnification provided shall be applicable whether or not negligence or gross negligence of the Indemnitee
is alleged or proven.
Neither
our Bylaws nor our Certificate of Formation include any specific indemnification provisions for our officers or directors against liability
under the Securities Act. Additionally, insofar as indemnification for liabilities arising under the Securities Act may be permitted
to directors, officers and controlling persons of the Company pursuant to the foregoing provisions, or otherwise, the Company has been
advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the
Securities Act and is, therefore, unenforceable.
Listing
Our
common stock trades on The Nasdaq Capital Market under the symbol “MGRX”. Trading of our common stock on The Nasdaq Capital
Market began on March 21, 2023.
Transfer
Agent
The
transfer agent for our common stock is ClearTrust, LLC located at 16540 Pointe Village Drive, Suite 210, Lutz, Florida, 33558.
SHARES ELIGIBLE FOR FUTURE SALE
In
general, under Rule 144, a person who has beneficially owned restricted shares for at least six months would be entitled to sell those
securities provided that (1) such person is not deemed to have been one of our affiliates at the time of, or at any time during the 90
days preceding, a sale and (2) we have been subject to the Exchange Act periodic reporting requirements for at least 90 days before the
sale and are current in filing our periodic reports. Persons who have beneficially owned restricted shares of common stock for at least
six months but who are our affiliates at the time of, or any time during the 90 days preceding, a sale, would be subject to additional
restrictions, by which such person would be entitled to sell within any three-month period only a number of securities that does not
exceed 1% of the number of shares of common stock outstanding, which will equal approximately 536,338 shares immediately after this offering,
based on the number of shares of common stock outstanding as May 6, 2024. Such sales by affiliates must also comply with the manner
of sale and notice provisions of Rule 144 and to the availability of current public information about us.
A
person (or persons whose shares are aggregated) who is deemed to be an affiliate of ours and who has beneficially owned restricted securities
within the meaning of Rule 144 for at least six months would be entitled to sell within any three-month period a number of shares that
does not exceed the greater of one percent of the then outstanding shares of our common stock or the average weekly trading volume of
our common stock on the during the four calendar weeks preceding such sale. Such sales are also subject to certain manner of sale provisions,
notice requirements, and the availability of current public information about us.
MATERIAL
U.S. FEDERAL INCOME TAX CONSEQUENCES FOR
NON-U.S. HOLDERS OF COMMON STOCK
The
following are the material U.S. federal income tax consequences of the ownership and disposition of our common stock acquired in this
offering by a “Non-U.S. Holder” that does not own, and has not owned, actually or constructively, more than 5% of common
stock . You are a Non-U.S. Holder if for U.S. federal income tax purposes you are a beneficial owner of our common stock that is:
| ● |
a
nonresident alien individual; |
| |
|
| ● |
a
foreign corporation; or |
| |
|
| ● |
a
foreign estate or trust. |
You
are not a Non-U.S. Holder if you are a nonresident alien individual present in the United States for 183 days or more in the taxable
year of disposition, or if you are a former citizen or former resident of the United States for U.S. federal income tax purposes. If
you are such a person, you should consult your tax adviser regarding the U.S. federal income tax consequences of the ownership and disposition
of our common stock.
If
a partnership or other pass-through entity (including an entity or arrangement treated as a partnership or other type of pass-through
entity for U.S. federal income tax purposes) owns our common stock, the tax treatment of a partner or beneficial owner of the entity
may depend upon the status of the owner, the activities of the entity and certain determinations made at the partner or beneficial owner
level. Partners and beneficial owners in partnerships or other pass-through entities that own our common stock should consult their own
tax advisors as to the particular U.S. federal income and estate tax consequences applicable to them.
This
discussion is based on the Internal Revenue Code of 1986, as amended to the date hereof (the “Code”), administrative pronouncements,
judicial decisions and final, temporary and proposed Treasury regulations, changes to any of which subsequent to the date of this prospectus
may affect the tax consequences described herein, possibly with retroactive effect. This discussion does not describe all of the tax
consequences that may be relevant to you in light of your particular circumstances, including alternative minimum tax and Medicare contribution
tax consequences and does not address any aspect of state, local or non-U.S. taxation, or any taxes other than income and estate taxes.
You should consult your tax adviser with regard to the application of the U.S. federal tax laws to your particular situation, as well
as any tax consequences arising under the laws of any state, local or non-U.S. taxing jurisdiction.
Dividends
As
discussed under “Dividend Policy” above, we do not anticipate paying any cash dividends in the foreseeable future. In the
event that we do make distributions of cash or other property, those distributions will constitute dividends for U.S. federal income
tax purposes to the extent paid from our current or accumulated earnings and profits, as determined under U.S. federal income tax principles.
To the extent those distributions exceed our current and accumulated earnings and profits, they will constitute a return of capital,
which will first reduce your basis in our common stock, but not below zero, and then will be treated as gain from the sale of our common
stock.
Dividends
paid to you generally will be subject to withholding tax at a 30% rate or a reduced rate specified by an applicable income tax treaty.
In order to obtain a reduced rate of withholding, you will be required to provide a properly executed applicable Internal Revenue Service
(“IRS”) Form W-8 certifying your entitlement to benefits under a treaty.
If
dividends paid to you are effectively connected with your conduct of a trade or business in the United States (and, if required by an
applicable income tax treaty, are attributable to a permanent establishment or fixed base maintained by you in the United States), you
will generally be taxed on the dividends in the same manner as a U.S. person. In this case, you will be exempt from the withholding tax
discussed in the preceding paragraph, although you will be required to provide a properly executed IRS Form W-8ECI in order to claim
an exemption from withholding. You should consult your tax adviser with respect to other U.S. tax consequences of the ownership and disposition
of our common stock, including the possible imposition of a branch profits tax at a rate of 30% (or a lower treaty rate) if you are a
corporation.
Information
Reporting and Backup Withholding
Information
returns are required to be filed with the IRS in connection with payments of dividends on our common stock. Unless you comply with certification
procedures to establish that you are not a U.S. person, information returns may also be filed with the IRS in connection with the proceeds
from a sale or other disposition of our common stock. You may be subject to backup withholding on payments on our common stock or on
the proceeds from a sale or other disposition of our common stock unless you comply with certification procedures to establish that you
are not a U.S. person or otherwise establish an exemption. Your provision of a properly executed applicable IRS Form W-8 certifying your
non-U.S. status will permit you to avoid backup withholding. Amounts withheld under the backup withholding rules are not additional taxes
and may be refunded or credited against your U.S. federal income tax liability, provided the required information is timely furnished
to the IRS.
FATCA
Withholding Taxes
Payments
to certain foreign entities of dividends on common stock of a U.S. issuer are subject to a withholding tax (separate and apart from,
but without duplication of, the withholding tax described above) at a rate of 30%, unless various U.S. information reporting and due
diligence requirements (generally relating to ownership by U.S. persons of interests in or accounts with those entities) have been satisfied
or an exemption from these rules applies. Under proposed regulations issued by the Treasury Department on December 13, 2018, which state
that taxpayers may rely on the proposed regulations until final regulations are issued, this withholding tax will not apply to the gross
proceeds from any sale or disposition of common stock. An intergovernmental agreement between the United States and an applicable foreign
country may modify these requirements. Non-U.S. holders should consult their tax advisors regarding the possible implications of this
withholding tax on dividends on common stock.
PLAN
OF DISTRIBUTION
We
are registering the shares of common stock to be issued to permit the resale of these shares of common stock, after they are issued,
by the holders of the common stock from time to time after the date of this prospectus. We will not receive any of the proceeds from
the sale by the selling stockholders of the shares of common stock. Any proceeds received by the Company from the sale of shares of common
stock pursuant to the ELOC and from the exercise of the Warrants will be used for general working capital. See section entitled
“Use of Proceeds”. We will bear all fees and expenses incident to our obligation to register the shares of common stock.
The
selling stockholders may sell all or a portion of the shares of common stock held by them and offered hereby from time to time directly
or through one or more underwriters, broker-dealers, or agents. If the shares of common stock are sold through underwriters or broker-dealers,
the selling stockholders will be responsible for underwriting discounts or commissions or agent’s commissions. The shares of common
stock may be sold in one or more transactions at fixed prices, at prevailing market prices at the time of the sale, at varying prices
determined at the time of sale or at negotiated prices. These sales may be effected in transactions, which may involve crosses or block
transactions, pursuant to one or more of the following methods:
| |
● |
on
any national securities exchange or quotation service on which the securities may be listed or quoted at the time of sale; |
| |
● |
in
the over-the-counter market; |
| |
● |
in
transactions otherwise than on these exchanges or systems or in the over-the-counter market; |
| |
● |
through
the writing or settlement of options, whether such options are listed on an options exchange or otherwise; |
| |
● |
ordinary
brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
| |
● |
block
trades in which the broker-dealer will attempt to sell the shares as agent but may position and resell a portion of the block as
principal to facilitate the transaction; |
| |
● |
purchases
by a broker-dealer as principal and resale by the broker-dealer for its account; |
| |
● |
an
exchange distribution in accordance with the rules of the applicable exchange; |
| |
● |
privately
negotiated transactions; |
| |
● |
short
sales made after the date the Registration Statement is declared effective by the SEC; |
| |
● |
agreements
between broker-dealers and the selling stockholders to sell a specified number of such shares at a stipulated price per share; |
| |
● |
a
combination of any such methods of sale; and |
| |
● |
any
other method permitted pursuant to applicable law. |
The
selling stockholders may also sell shares of common stock under Rule 144 promulgated under the Securities Act, if available, rather than
under this prospectus. In addition, the selling stockholders may transfer the shares of common stock by other means not described in
this prospectus. The ELOC and all exhibits thereto (the “Transaction Documents”) are binding upon and inure to the
benefit of the Company and the Purchaser and their respective successors. Pursuant to the terms and conditions of the ELOC,
neither any of the Transaction Documents nor any rights of the Purchaser or the Company under the ELOC may be assigned
by either party to any other person or entity. If the selling stockholders effect such transactions by selling shares of common stock
to or through underwriters, broker-dealers or agents, such underwriters, broker-dealers, or agents may receive commissions in the form
of discounts, concessions, or commissions from the selling stockholders or commissions from purchasers of the shares of common stock
for whom they may act as agent or to whom they may sell as principal (which discounts, concessions or commissions as to particular underwriters,
broker-dealers or agents may be in excess of those customary in the types of transactions involved). In connection with sales of the
shares of common stock or otherwise, the selling stockholders may enter into hedging transactions with broker-dealers, which may in turn
engage in short sales of the shares of common stock in the course of hedging in positions they assume. The selling stockholders may also
sell shares of common stock short and deliver shares of common stock covered by this prospectus to close out short positions and to return
borrowed shares in connection with such short sales. The selling stockholders may also loan or pledge shares of common stock to broker-dealers
that in turn may sell such shares.
The
selling stockholders may pledge or grant a security interest in some or all of the shares of common stock owned by them and, if they
default in the performance of their secured obligations, the pledgees or secured parties may offer and sell the shares of common stock
from time to time pursuant to this prospectus or any amendment to this prospectus under Rule 424(b)(3) or other applicable provision
of the Securities Act amending, if necessary, the list of selling stockholders to include the pledgee, transferee, or other successors
in interest as selling stockholders under this prospectus. The selling stockholders also may transfer and donate the shares of common
stock in other circumstances in which case the transferees, donees, pledgees, or other successors in interest will be the selling beneficial
owners for purposes of this prospectus.
To
the extent required by the Securities Act and the rules and regulations thereunder, the selling stockholders and any broker-dealer participating
in the distribution of the shares of common stock may be deemed to be “underwriters” within the meaning of the Securities
Act, and any commission paid, or any discounts or concessions allowed to, any such broker-dealer may be deemed to be underwriting commissions
or discounts under the Securities Act. At the time a particular offering of the shares of common stock is made, a prospectus supplement,
if required, will be distributed, which will set forth the aggregate amount of shares of common stock being offered and the terms of
the offering, including the name or names of any broker-dealers or agents, any discounts, commissions, and other terms constituting compensation
from the selling stockholders and any discounts, commissions, or concessions allowed or re-allowed or paid to broker-dealers.
Under
the securities laws of some states, the shares of common stock may be sold in such states only through registered or licensed brokers
or dealers. In addition, in some states the shares of common stock may not be sold unless such shares have been registered or qualified
for sale in such state or an exemption from registration or qualification is available and is complied with.
There
can be no assurance that any selling stockholder will sell any or all of the shares of common stock registered pursuant to the registration
statement, of which this prospectus forms a part.
The
selling stockholders and any other person participating in such distribution will be subject to applicable provisions of the Securities
Exchange Act of 1934, as amended, and the rules and regulations thereunder, including, without limitation, to the extent applicable,
Regulation M under the Exchange Act, which may limit the timing of purchases and sales of any of the shares of common stock by the selling
stockholders and any other participating person. To the extent applicable, Regulation M may also restrict the ability of any person engaged
in the distribution of the shares of common stock to engage in market-making activities with respect to the shares of common stock. All
of the foregoing may affect the marketability of the shares of common stock and the ability of any person or entity to engage in market-making
activities with respect to the shares of common stock.
We will pay all expenses of the registration of the shares of common
stock pursuant to the registration rights agreement, including, without limitation, SEC filing fees and expenses of compliance with state
securities or “blue sky” laws; provided, however, a selling stockholder will pay all underwriting discounts and selling commissions,
if any. We will indemnify the selling stockholders against liabilities, including some liabilities under the Securities Act, in accordance
with the registration rights agreements, or the selling stockholders will be entitled to contribution. We may be indemnified by the selling
stockholders against civil liabilities, including liabilities under the Securities Act that may arise from any written information furnished
to us by the selling stockholder specifically for use in this prospectus, in accordance with the related registration rights agreements
or we may be entitled to contribution.
Once
sold under the registration statement, of which this prospectus forms a part, the shares of common stock will be freely tradable in the
hands of persons other than our affiliates.
LEGAL
MATTERS
The
validity of the shares of our common stock offered hereby will be passed upon for us by Lucosky Brookman LLP.
EXPERTS
The
financial statements of Mangoceuticals, Inc. as of December 31, 2023 and December 31, 2022 and for the years then included in this prospectus
and the registration statement have been audited by Turner, Stone & Company, L.L.P., Dallas, Texas, independent registered public
accounting firm, and have been so included in reliance upon the report of such firm given upon their authority as experts in accounting
and auditing.
WHERE
YOU CAN FIND MORE INFORMATION
We
are a reporting company and file annual, quarterly and current reports, and other information with the SEC. Our filings are also available
to the public from the SEC’s website at www.sec.gov. The SEC’s website contains reports, proxy and information statements
and other information regarding registrants that file electronically with the SEC.
This
prospectus is part of a registration statement on Form S-1 that we filed with the SEC to register the securities offered hereby under
the Securities Act. This prospectus does not contain all of the information included in the registration statement, including certain
exhibits and schedules. For further information with respect to our company and the securities offered by this prospectus, as well as
the exhibits and schedules to the registration statement, we refer you to the registration statement and those exhibits and schedules.
You may obtain the registration statement and exhibits to the registration statement from the SEC at the address listed above or from
the SEC’s website.
INDEX
TO FINANCIAL STATEMENTS
MANGOCEUTICALS,
INC.
TABLE
OF CONTENTS TO FINANCIAL STATEMENTS

Report
of Independent Registered Public Accounting Firm
Board
of Directors and Stockholders of
Mangoceuticals, Inc.
Opinion
on the Financial Statements
We
have audited the accompanying consolidated balance sheets of Mangoceuticals, Inc. as of December 31, 2023 and 2022, and the related consolidated
statements of operations, changes in stockholders’ equity (deficit) and cash flows for each of the two years in the period ended
December 31, 2023, and the related notes (collectively referred to as the “financial statements”). In our opinion, the financial
statements present fairly, in all material respects, the financial position of Mangoceuticals, Inc. as of December 31, 2023 and 2022,
and the results of its operations and its cash flows for each of the two years in the period ended December 31, 2023, in conformity with
accounting principles generally accepted in the United States of America.
Going
Concern
The
accompanying financial statements have been prepared assuming that the entity will continue as a going concern. As discussed in Note
9 to the financial statements, the entity has suffered recurring losses from operations that raise substantial doubt about its ability
to continue as a going concern. Management’s plans in regard to these matters are also described in Note 9. The financial statements
do not include any adjustments that might result from the outcome of this uncertainty.
Basis
for Opinion
These
financial statements are the responsibility of the entity’s management. Our responsibility is to express an opinion on these financial
statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United
States) (“PCAOB”) and are required to be independent with respect to Mangoceuticals, Inc. in accordance with the U.S. federal
securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We
conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Mangoceuticals,
Inc. is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our
audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing
an opinion on the effectiveness of the entity’s internal control over financial reporting. Accordingly, we express no such opinion.
Our
audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error
or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding
the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant
estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits
provides a reasonable basis for our opinion.
/s/
Turner, Stone & Company, L.L.P.
We
have served as Mangoceuticals, Inc.’s auditor since 2023.
Dallas,
Texas
April
1, 2024

Mangoceuticals,
Inc.
Balance
Sheets
| | |
December 31, 2023 | | |
December 31, 2022 | |
| CURRENT ASSETS | |
| | | |
| | |
| Cash and cash equivalents | |
$ | 739,006 | | |
$ | 682,860 | |
| Inventory | |
| 18,501 | | |
| - | |
| Prepaid expenses - related party | |
| 60,953 | | |
| 11,745 | |
| TOTAL CURRENT ASSETS | |
| 818,460 | | |
| 694,605 | |
| | |
| | | |
| | |
| FIXED ASSETS | |
| | | |
| | |
| Property and equipment, net of accumulated depreciation of $28,752 and $3,863 | |
| 96,129 | | |
| 117,499 | |
| TOTAL FIXED ASSETS | |
| 96,129 | | |
| 117,499 | |
| | |
| | | |
| | |
| OTHER ASSETS | |
| | | |
| | |
| Deposits | |
| 16,942 | | |
| 16,942 | |
| Right of use - asset | |
| 119,262 | | |
| 174,241 | |
| TOTAL OTHER ASSETS | |
| 136,204 | | |
| 191,183 | |
| TOTAL ASSETS | |
$ | 1,050,793 | | |
$ | 1,003,287 | |
| | |
| | | |
| | |
| LIABILITIES AND STOCKHOLDERS’ EQUITY | |
| | | |
| | |
| | |
| | | |
| | |
| CURRENT LIABILITIES | |
| | | |
| | |
| Accounts payable and accrued liabilities | |
$ | 140,765 | | |
$ | 33,675 | |
| Payroll tax liabilities | |
| 6,595 | | |
| 2,717 | |
| Notes payable to related parties | |
| - | | |
| 89,200 | |
| Notes payable | |
| - | | |
| 78,260 | |
| Right-of-use liability - operating lease | |
| 63,718 | | |
| 56,725 | |
| TOTAL CURRENT LIABILITIES | |
| 211,078 | | |
| 260,577 | |
| LONG-TERM LIABILITIES | |
| | | |
| | |
| Right-of-use liability - operating lease | |
| 64,961 | | |
| 128,680 | |
| TOTAL LONG-TERM LIABILITIES | |
| 64,961 | | |
| 128,680 | |
| | |
| | | |
| | |
| TOTAL LIABILITIES | |
| 276,039 | | |
| 389,257 | |
| | |
| | | |
| | |
| COMMITMENTS AND CONTINGENCIES (SEE NOTE 10) | |
| - | | |
| - | |
| | |
| | | |
| | |
| STOCKHOLDERS’ EQUITY | |
| | | |
| | |
| Common stock (par value $0.0001, 200,000,000 shares authorized, of which 21,419,500 and 13,365,000 shares issued and outstanding as of December 31, 2023 and 2022, respectively) | |
| 2,142 | | |
| 1,337 | |
| Additional paid in capital | |
| 12,000,785 | | |
| 2,628,449 | |
| Accumulated deficit | |
| (11,228,173 | ) | |
| (2,015,756 | ) |
| TOTAL STOCKHOLDERS’ EQUITY | |
| 774,754 | | |
| 614,030 | |
| | |
| | | |
| | |
| TOTAL LIABILITIES AND STOCKHOLDERS’ EQUITY | |
$ | 1,050,793 | | |
$ | 1,003,287 | |
The
accompanying notes are an integral part of these financial statements.
Mangoceuticals,
Inc.
Statements
of Operations
| | |
December 31, 2023 | | |
December 31, 2022 | |
| | |
For The Year | | |
For The Year | |
| | |
Ended | | |
Ended | |
| | |
December 31, 2023 | | |
December 31, 2022 | |
| | |
| | |
| |
| Revenues | |
| | | |
| | |
| Revenues | |
$ | 731,493 | | |
$ | 8,939 | |
| | |
| | | |
| | |
| Cost of revenues | |
| 154,900 | | |
| 4,089 | |
| Cost of revenues - related party | |
| 145,092 | | |
| - | |
| Gross profit | |
| 431,501 | | |
| 4,850 | |
| | |
| | | |
| | |
| Operating expenses | |
| | | |
| | |
| General and administrative expenses | |
| 3,319,417 | | |
| 1,643,572 | |
| Salary and benefits | |
| 977,890 | | |
| - | |
| Advertising and marketing | |
| 2,097,505 | | |
| 352,860 | |
| Investor relations | |
| 1,100,465 | | |
| - | |
| Stock based compensation | |
| 2,155,114 | | |
| - | |
| Total operating expenses | |
| 9,650,391 | | |
| 1,996,432 | |
| | |
| | | |
| | |
| Loss from operations | |
| (9,218,890 | ) | |
| (1,991,582 | ) |
| | |
| | | |
| | |
| Other (income) expense | |
| | | |
| | |
| Other income | |
| (6,473 | ) | |
| - | |
| Imputed interest - related party | |
| - | | |
| 6,473 | |
| Total other (income) expense | |
| (6,473 | ) | |
| 6,473 | |
| | |
| | | |
| | |
| | |
| | | |
| | |
| Income taxes | |
| - | | |
| - | |
| | |
| | | |
| | |
| Net loss | |
$ | (9,212,417 | ) | |
$ | (1,998,055 | ) |
| | |
| | | |
| | |
| Basic and diluted loss per share | |
| | | |
| | |
| Basic and diluted loss per share | |
$ | (0.57 | ) | |
$ | (0.19 | ) |
| | |
| | | |
| | |
| Weighted average number of shares outstanding | |
| | | |
| | |
| Basic and diluted | |
| 16,113,029 | | |
| 10,798,083 | |
The
accompanying notes are an integral part of these financial statements.
MANGOCEUTICAL,
INC.
Statement of Changes in Stockholders’ Equity (Deficit)
For
the Years Ended December 31, 2023 and 2022
| | |
Shares | | |
Amount | | |
Capital | | |
Deficit | | |
(Deficit) | |
| | |
Common Stock | | |
Additional Paid-in | | |
Accumulated | | |
Total
Stockholders’
Equity | |
| | |
Shares | | |
Amount | | |
Capital | | |
Deficit | | |
(Deficit) | |
| | |
| | |
| | |
| | |
| | |
| |
| Balance, December 31, 2021 | |
| 8,000,000 | | |
$ | 800 | | |
$ | 181 | | |
$ | (17,701 | ) | |
$ | (16,720 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance of common stock for services | |
| 3,365,000 | | |
| 337 | | |
| 539,728 | | |
| - | | |
| 540,065 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance of units for cash | |
| 2,000,000 | | |
| 200 | | |
| 1,999,800 | | |
| - | | |
| 2,000,000 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Options and warrants vested for services | |
| - | | |
| - | | |
| 234,088 | | |
| - | | |
| 234,088 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Warrants for services cancelled | |
| - | | |
| - | | |
| (151,821 | ) | |
| - | | |
| (151,821 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Imputed interest | |
| - | | |
| - | | |
| 6,473 | | |
| - | | |
| 6,473 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Net loss | |
| - | | |
| - | | |
| - | | |
| (1,998,055 | ) | |
| (1,998,055 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Balance, December 31, 2022 | |
| 13,365,000 | | |
$ | 1,337 | | |
$ | 2,628,449 | | |
$ | (2,015,756 | ) | |
$ | 614,030 | |
| Balance | |
| 13,365,000 | | |
$ | 1,337 | | |
$ | 2,628,449 | | |
$ | (2,015,756 | ) | |
$ | 614,030 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance of common stock for services | |
| 1,780,000 | | |
| 178 | | |
| 1,530,473 | | |
| - | | |
| 1,530,651 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance of common stock for cash | |
| 5,250,000 | | |
| 525 | | |
| 6,199,475 | | |
| - | | |
| 6,200,000 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Imputed interest related party loan repayment | |
| - | | |
| - | | |
| (6,473 | ) | |
| - | | |
| (6,473 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Options and warrants vested for services | |
| - | | |
| - | | |
| 624,463 | | |
| - | | |
| 624,463 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Warrants exercised | |
| 1,024,500 | | |
| 102 | | |
| 1,024,398 | | |
| - | | |
| 1,024,500 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Net loss | |
| - | | |
| - | | |
| - | | |
| (9,212,417 | ) | |
| (9,212,417 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Balance, December 31, 2023 | |
| 21,419,500 | | |
| 2,142 | | |
| 12,000,785 | | |
| (11,228,173 | ) | |
| 774,754 | |
| Balance | |
| 21,419,500 | | |
| 2,142 | | |
| 12,000,785 | | |
| (11,228,173 | ) | |
| 774,754 | |
The
accompanying notes are an integral part of these financial statements.
MANGOCEUTICALS,
INC.
Statements
of Cash Flows
| | |
December 31, 2023 | | |
December 31, 2022 | |
| | |
For the Year Ended | | |
For the Year Ended | |
| | |
December 31, 2023 | | |
December 31, 2022 | |
| | |
| | |
| |
| CASH FLOWS FROM OPERATING ACTIVITIES: | |
| | | |
| | |
| Net loss | |
$ | (9,212,417 | ) | |
$ | (1,998,055 | ) |
| Adjustments to reconcile net loss to net cash used in operating activities: | |
| | | |
| | |
| Depreciation | |
| 24,889 | | |
| 3,863 | |
| Issuance of common stock for services | |
| 1,530,651 | | |
| 540,065 | |
| Imputed interest expense | |
| - | | |
| 6,473 | |
| Other income | |
| (6,473 | ) | |
| - | |
| Options vested for stock-based compensation | |
| 624,463 | | |
| 234,088 | |
| Warrants for service cancelled | |
| - | | |
| (151,821 | ) |
| (Increase) decrease in operating assets: | |
| | | |
| | |
| Rent Deposits | |
| - | | |
| (16,942 | ) |
| Inventory | |
| (18,501 | ) | |
| - | |
| Prepaid expenses – related party | |
| (49,208 | ) | |
| (11,745 | ) |
| Operating lease right of use asset | |
| 54,979 | | |
| (174,241 | ) |
| (Decrease) increase in operating liabilities: | |
| | | |
| | |
| Accounts payable and accrued liabilities | |
| 107,090 | | |
| 33,675 | |
| Operating lease right of use liabilities | |
| (56,726 | ) | |
| 185,405 | |
| Payroll tax liabilities | |
| 3,878 | | |
| 2,717 | |
| NET CASH USED IN OPERATING ACTIVITIES | |
| (6,997,375 | ) | |
| (1,346,518 | ) |
| | |
| | | |
| | |
| CASH FLOWS FROM INVESTING ACTIVITIES: | |
| | | |
| | |
| | |
| | | |
| | |
| Purchases of property and equipment | |
| (3,519 | ) | |
| (43,102 | ) |
| NET CASH USED IN INVESTING ACTIVITIES | |
| (3,519 | ) | |
| (43,102 | ) |
| | |
| | | |
| | |
| CASH FLOWS FROM FINANCING ACTIVITIES: | |
| | | |
| | |
| Proceeds from borrowings on notes payable to related parties | |
| - | | |
| 75,000 | |
| Repayment on notes payable | |
| (78,260 | ) | |
| - | |
| Repayment on notes payable - related parties | |
| (89,200 | ) | |
| (25,070 | ) |
| Proceeds from exercise of warrants | |
| 1,024,500 | | |
| - | |
| Proceeds from sales of common stock for cash | |
| 6,200,000 | | |
| 2,000,000 | |
| NET CASH PROVIDED BY FINANCING ACTIVITIES | |
| 7,057,040 | | |
| 2,049,930 | |
| | |
| | | |
| | |
| NET INCREASE IN CASH AND CASH EQUIVALENTS | |
| 56,146 | | |
| 660,310 | |
| | |
| | | |
| | |
| CASH AND CASH EQUIVALENTS: | |
| | | |
| | |
| Beginning of year | |
| 682,860 | | |
| 22,550 | |
| End of year | |
$ | 739,006 | | |
$ | 682,860 | |
| | |
| | | |
| | |
| Supplemental disclosure of cash flow information: | |
| | | |
| | |
| Cash paid for income taxes | |
$ | - | | |
$ | - | |
| Cash paid for interest | |
$ | - | | |
$ | - | |
The
accompanying notes are an integral part of these financial statements.
MANGOCEUTICALS,
INC.
Notes
to Financial Statements
Years
Ended December 31, 2023 and 2022
NOTE
1 – ORGANIZATION AND DESCRIPTION OF THE BUSINESS
Mangoceuticals,
Inc. (“Mangoceuticals” or the “Company”), was incorporated in the State of a Texas on October 7, 2021, with the
intent of focusing on developing, marketing, and selling a variety of men’s wellness products and services via a telemedicine platform.
To date, the Company has identified men’s wellness telemedicine services and products as a growing sector in the most recent years
and especially related to the areas of erectile dysfunction (“ED”), hair loss and testosterone replacement or enhancement
therapies. In this regard, Mangoceuticals has developed and is commercially marketing a new brand of ED products under the brand name
“Mango” and a new brand of hair loss products under the brand name “Grow.” These products are produced at a compounding
pharmacy using a proprietary combination of U.S. Food and Drug Administration (“FDA”) approved ingredients and is available
to patients on the determination of a prescribing physician that the compounded drug is necessary for the individual patient. Mangoceuticals
is currently marketing and selling these branded ED and hair loss products exclusively online via its website at www.MangoRx.com.
Initial
Public Offering. In March 2023, the Company completed an initial public offering (the “IPO”), in which the Company issued
and sold 1,250,000 shares of authorized common stock for $4.00 per share for net proceeds of $4.35 million, after deducting underwriting
discounts and commissions, and offering costs. At the same time, and as part of the same registration statement, but pursuant to a separate
prospectus (the “Resale Prospectus”) the Company registered the sale of 4,765,000 shares of common stock, including 2,000,000
shares of common stock issuable upon the exercise of outstanding warrants to purchase shares of common stock with an exercise price of
$1.00 per share.
NOTE
2 – SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis
of Presentation – The financial statements present the financial position, results of operations and cash flows of the Company
in accordance with accounting principles generally accepted in the United States of America (“US GAAP”). All dollar amounts
are rounded to the nearest thousand dollars.
Cash
Equivalents
Highly
liquid investments with original maturities of three months or less are considered cash equivalents. The Company maintains the majority
of its cash accounts at a commercial bank. The Federal Deposit Insurance Corporation (“FDIC”) insures the total cash balance
up to $250,000 per commercial bank. From time to time, cash in deposit accounts may exceed the FDIC limits, the excess would be at risk
of loss for purposes of the statement of cash flows. There are no cash equivalents at December 31, 2023 and 2022
Income
Taxes
The
Company is a taxable entity and recognizes deferred tax assets and liabilities for the future tax consequences attributable to differences
between the financial statement carrying amounts of existing assets and liabilities and their respective tax basis. Income taxes are
provided in accordance with Financial Accounting Standards Board (“FASB”) Accounting Standards Codification (“ASC”)
740, Income Taxes. Deferred tax assets and liabilities are measured using enacted tax rates expected to be in effect when the
temporary differences reverse. The effect on the deferred tax assets and liabilities of a change in tax rates is recognized in income
in the year that includes the enactment date of the rate change. A valuation allowance is used to reduce deferred tax assets to the amount
that is more likely than not to be realized.
Net
Loss Per Common Share
We
compute net loss per share in accordance with ASC 260, Earning per Share. ASC 260 requires presentation of both basic and
diluted earnings per share (“EPS”) on the face of the statement of operations. Basic EPS is computed by dividing net loss available
to common shareholders (numerator) by the weighted average number of shares outstanding (denominator) during the period. Diluted EPS
gives effect to all dilutive potential common shares outstanding during the period using the treasury stock method and convertible
preferred stock using the if-converted method. In computing Diluted EPS, the average stock price for the period is used in
determining the number of shares assumed to be purchased from the exercise of stock options or warrants. Diluted EPS excludes all
dilutive potential shares if their effect is anti-dilutive. There were 2,650,000
options, 1,343,000
warrants and no derivative securities outstanding as of December 31, 2023. There were 1,250,000
options, 2,000,000
warrants and no derivative securities outstanding as of December 31, 2022.
Use
of Estimates and Assumptions
The
preparation of financial statements in accordance with US GAAP requires the Company’s management to make estimates and assumptions
that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial
statements and the reported amounts of expenses during the reporting period. Actual results can, and in many cases will, differ from
those estimates.
Fair
Value of Financial Instruments
The
Company measures its financial and non-financial assets and liabilities, as well as makes related disclosures, in accordance with FASB
ASC 820, Fair Value Measurement (“ASC 820”), which provides guidance with respect to valuation techniques to be utilized
in the determination of fair value of assets and liabilities. Approaches include, (i) the market approach (comparable market prices),
(ii) the income approach (present value of future income or cash flow), and (iii) the cost approach (cost to replace the service capacity
of an asset or replacement cost). ASC 820 utilizes a fair value hierarchy that prioritizes the inputs to valuation techniques used to
measure fair value into three broad levels. The following is a brief description of those three levels:
Level
1: Observable inputs such as quoted prices (unadjusted) in active markets for identical assets or liabilities.
Level
2: Inputs other than quoted prices that are observable, either directly or indirectly. These include quoted prices for similar assets
or liabilities in active markets and quoted prices for identical or similar assets or liabilities in markets that are not active.
Level
3: Unobservable inputs in which little or no market data exists, therefore requiring an entity to develop its own assumptions, such as
valuations derived from valuation techniques in which one more significant inputs or significant value drivers are unobservable.
The
following tables summarize our financial instruments measured at fair value as of December 31, 2023 and 2022.
SCHEDULE
OF FINANCIAL INSTRUMENTS MEASUREMENT AT FAIR VALUE
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| | |
Fair Value Measurements at December 31, 2023 | |
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| Assets | |
| | | |
| | | |
| | |
| Cash | |
$ | 739,006 | | |
$ | - | | |
$ | - | |
| Total assets | |
| 739,006 | | |
| - | | |
| - | |
| Liabilities | |
| | | |
| | | |
| | |
| Total liabilities | |
| - | | |
| - | | |
| - | |
| Fair value, net asset
(liability) | |
$ | 739,006 | | |
$ | - | | |
$ | - | |
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| | |
Fair Value Measurements at December 31, 2022 | |
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| Assets | |
| | | |
| | | |
| | |
| Cash | |
$ | 682,860 | | |
$ | - | | |
$ | - | |
| Total assets | |
| 682,860 | | |
| - | | |
| - | |
| Liabilities | |
| | | |
| | | |
| | |
| Total liabilities | |
| - | | |
| - | | |
| - | |
| Fair value, net asset
(liability) | |
$ | 682,860 | | |
$ | - | | |
$ | - | |
Property
and Equipment
Property
and equipment are stated at cost. When retired or otherwise disposed, the related carrying value and accumulated depreciation are removed
from the respective accounts and the net difference less any amount realized from the disposition is reflected in earnings. For financial
statement purposes, property and equipment are recorded at cost and depreciated using the straight-line method over their estimated useful
lives of three (3) to five (5) years.
Concentration
and Risks
The
Company’s operations are subject to risks including financial, operational, regulatory and other risks including the potential
risk of business failure. For the years ended December 31, 2023 and 2022, the Company had no significant revenue from continuing
operations which were derived from a single or a few major customers.
Black-Scholes Option Pricing Model
The
Company uses a Black-Scholes option pricing model to determine the fair value of warrants and options issued.
Recently
Issued Accounting Pronouncements
From
time to time, new accounting pronouncements are issued by the FASB or other standard setting bodies that are adopted by the Company as
of the specified effective date. Unless otherwise discussed, the Company believes that the effect of recently issued standards that are
not yet effective will not have a material effect on its financial position or results of operations upon adoption.
In
August 2020, the FASB issued Accounting Standards Update (“ASU”) 2020-06, Debt – Debt with Conversion and
Other Options (Subtopic 470-20) and Derivatives and Hedging – Contracts in Entity’s Own Equity (Subtopic 815 – 40) (“ASU 2020-06”). ASU 2020-06 simplifies the accounting for certain financial instruments with
characteristics of liabilities and equity, including convertible instruments and contracts on an entity’s own equity. The ASU
is part of the FASB’s simplification initiative, which aims to reduce unnecessary complexity in US GAAP. The ASU’s
amendments are effective for fiscal years beginning after December 15, 2023, and interim periods within those fiscal years. The
Company is currently evaluating the impact of ASU 2020-06 on its financial statements.
Related
Parties
The
Company follows subtopic 850-10 of FASB ASC 850, Related Party Disclosures for the identification of related parties and disclosure
of related party transactions.
Pursuant
to Section 850-10-20, the related parties include a. affiliates of the Company; b. Entities for which investments in their equity securities
would be required, absent the election of the fair value option under the guidance of Fair Value Option Subsection of Section 825–10–15,
to be accounted for by the equity method by the investing entity; c. trusts for the benefit of employees, such as pension and profit-sharing
trusts that are managed by or under the trusteeship of management; d. principal owners of the Company; e. management of the Company;
f. other parties with which the Company may deal if one party controls or can significantly influence the management or operating policies
of the other to an extent that one of the transacting parties might be prevented from fully pursuing its own separate interests; and
g. other parties that can significantly influence the management or operating policies of the transacting parties or that have an ownership
interest in one of the transacting parties and can significantly influence the other to an extent that one or more of the transacting
parties might be prevented from fully pursuing its own separate interests.
The
financial statements shall include disclosures of material related party transactions, other than compensation arrangements, expense
allowances, and other similar items in the ordinary course of business. However, disclosure of transactions that are eliminated in the
preparation of financial statements is not required in those statements. The disclosures shall include: a. the
nature of the relationship(s) involved; b. a description of the transactions, including transactions to which no amounts or nominal amounts
were ascribed, for each of the periods for which income statements are presented, and such other information deemed necessary to an understanding
of the effects of the transactions on the financial statements; c. the dollar amounts of transactions for each of the periods for which
income statements are presented and the effects of any change in the method of establishing the terms from that used in the preceding
period; and d. amounts due from or to related parties as of the date of each balance sheet presented and, if not otherwise apparent,
the terms and manner of settlement. Material related party transactions have been identified in Notes 3, 6 and 8 in the notes to financial
statements.
Stock-Based
Compensation
The
Company recognizes compensation costs to employees under FASB ASC 718 Compensation - Stock Compensation (“ASC
718”). Under ASC 718, companies are required to measure the compensation costs of share-based compensation arrangements based
on the grant-date fair value and recognize the costs in the financial statements over the period during which employees are required
to provide services. Share-based compensation arrangements include stock options and warrants. As such, compensation cost is
measured on the date of grant at their fair value. Such compensation amounts, if any, are amortized over the respective vesting
periods of the option and warrant grant.
Revenue
Recognition
The Company follows the provisions of ASC 606. Revenue from Contracts with Customer for recording and recognizing
revenue from customers. The
Company generates our online revenue through the sale of products and services purchased by customers directly through our online platform.
Online revenue represents the sales of products and services on our platform, net of refunds, credits, and chargebacks, and includes
revenue recognition adjustments recorded pursuant to US GAAP. Online revenue is generated by selling directly to consumers through our
websites.
The
Company recognizes revenue when it transfers promised goods or services to customers in an amount that reflects the consideration to
which it expects to be entitled in exchange for those goods or services and has met its performance obligation. For revenue generated
through its online platform, the Company defines its customer as an individual who purchases products or services through websites. The
transaction price in the Company’s contracts with customers is the total amount of consideration to which the Company expects to
be entitled in exchange for transferring products or services to the customer.
The
Company’s contracts that contain prescription products issued as the result of a consultation include two performance obligations:
access to (i) products and (ii) consultation services. The Company’s contracts for prescription refills have a single performance
obligation. Revenue is recognized at the time the related performance obligation is satisfied by transferring the promised product to
the customer and, in contracts that contain services, by the provision of consultation services to the customer. The Company satisfies
its performance obligation for products at a point in time, which is upon delivery of the products to a third-party carrier. The Company
satisfies its performance obligation for services over the period of the consultation service, which is typically a few days. The customer
obtains control of the products and services upon the Company’s completion of its performance obligations.
The
Company has entered into a Physician Services Agreement with BrighterMD, LLC dba Doctegrity (“Doctegrity”) to provide online
telemedicine technology services to the Company. The Company accounts for service revenue as a principal in the arrangement with its
customers. This conclusion is reached because (i) the Company determines which providers provide the consultation to the customer; (ii)
the Company is primarily responsible for the satisfactory fulfillment and acceptability of the services; (iii) the Company incurs costs
for consultation services even for visits that do not result in a prescription and the sale of products; and (iv) the Company, at its
sole discretion, sets all listed prices charged on its websites for products and services.
Additionally,
the Company has entered into a Master Services Agreement and Statement of Work with Epiq Scripts, LLC (“Contracted Pharmacy”),
which is a related party, to provide pharmacy and compounding services to the Company to fulfill its promise to customers for contracts
that include sale of prescription products and to fill prescriptions that are ordered by the Company’s customers for fulfillment
through the Company’s websites. The Company accounts for prescription product revenue as a principal in the arrangement with its
customers. This conclusion is reached because (i) the Company has sole discretion in determining which Contracted Pharmacy fills a customer’s
prescription; (ii) Contracted Pharmacy fills the prescription based on fulfillment instructions provided by the Company, including using
the Company’s branded packaging for generic products; (iii) the Company is primarily responsible to the customer for the satisfactory
fulfillment and acceptability of the order, and; (iv) the Company, at its sole discretion, sets all listed prices charged on its websites
for products and services.
The
Company accounts for shipping activities, consisting of direct costs to ship products performed after the control of a product has been
transferred to the customer, in cost of revenue.
Inventories
Inventories
are stated at the lower of cost or net realizable value with cost being determined on a first-in, first-out (“FIFO”) basis. The Company writes down its
inventory for estimated obsolescence or unmarketable inventory equal to the difference between the cost of inventory and the estimated
market value based upon assumptions about future demand and market conditions. If actual market conditions are less favorable than those
projected by management, additional inventory write-downs may be required. During the years ended December 31, 2023 and 2022, there were no inventory write-downs.
Marketing
and Advertising
The
Company follows the policy of charging the costs of marketing and advertising to expense as incurred. The Company charged $2,097,505
and $352,860 towards marketing and advertising for the years ended December 31, 2023 and 2022, respectively.
Subsequent
Events
The
Company follows the guidance in Section 855-10-50 of FASB ASC 855, Subsequent Events, for the disclosure of subsequent
events. The Company will evaluate subsequent events through the date when the financial statements were issued (see Note
12).
NOTE
3 – PREPAID EXPENSES-RELATED PARTIES
During
the year ended December 31, 2023, and in association with the Master Services Agreement and Statement of Work with our related party
Contracted Pharmacy, the Company prepays the related party Contracted Pharmacy as a retainer to be credited towards future product sales.
As of December 31, 2023 and 2022, the balance was $60,953 and $11,745, respectively.
Additionally,
the Company signed a lease agreement for office space, effective October 1, 2022, which included an initial security deposit of $16,942.
NOTE
4 – INVENTORY
During
the years ended December 31, 2023 and 2022, the Company purchased inventories related to promotional merchandise intended to be sold
online. As of December 31, 2023 and 2022, the inventory balance was $18,501 and $0, respectively.
NOTE
5 – PROPERTY, PLANT AND EQUIPMENT
During
the year ended December 31, 2023, the Company acquired computers and office equipment totaling $3,519. Depreciation for the year ended
December 31, 2023 and 2022 was $24,889 and $3,863, respectively. Total net property, plant and equipment was $96,129 and $117,499, as
of December 31, 2023 and 2022, respectively.
SCHEDULE
OF PROPERTY PLANT AND EQUIPMENT
| | |
December 31, 2023 | | |
December 31, 2022 | |
| | |
| | |
| |
| Computers | |
| 5,062 | | |
| 5,062 | |
| Equipment | |
| 119,819 | | |
| 116,300 | |
| Less accumulated depreciation: | |
| (28,752 | ) | |
| (3,863 | ) |
| Property and equipment, net | |
| 96,129 | | |
| 117,499 | |
NOTE
6 – LOANS FROM RELATED PARTIES
On
December 10, 2021 and March 18, 2022, the Company received advances of $39,200 and $50,000, respectively, for a total of $89,200 from
its previous majority shareholder, American International Holdings Corp (“AMIH”), in order to cover various general and administrative
expenses. The advances bear no interest and are due on demand upon the Company’s ability to repay the advances from either future
revenues or investment proceeds. On June 16, 2022, Cohen Enterprises, Inc. (“Cohen Enterprises”), an entity owned and controlled
by Jacob D. Cohen, the Company’s Chief Executive Officer and Chairman of the Board of Directors, entered into and closed a Stock
Purchase Agreement (the “SPA”) for the purchase of 8,000,000 shares of the outstanding common stock of the Company which
were then held by AMIH, which represented 80% of the Company’s then outstanding shares of common stock, in consideration for $90,000.
Pursuant to the terms of the SPA, Cohen Enterprises also acquired the right to be repaid the $89,200 advanced from AMIH to the Company.
On
June 29, 2022, the Company received an advance of $25,000 from Cohen Enterprises in order to cover various general and administrative
expenses. The Company repaid Cohen Enterprises $25,000 on August 18, 2022 bringing the total amount owed to Cohen Enterprises to $89,200
as of December 31, 2022. This amount was paid in full on April 4, 2023 and the amount owed to Cohen Enterprises was $0 and $89,200 as
of December 31, 2023 and 2022, respectively. Previously recorded imputed interest equal to eight percent (8%) per annum, or a total of
$8,232 against the related party advances, was canceled and reversed for the year ended December 31, 2023.
On
December 10, 2021, the Company received an advance of $70 from ZipDoctor, Inc., a then wholly-owned subsidiary of its then majority shareholder,
AMIH, which was used to open and establish the Company’s bank account. The advance bears no interest and is due on demand upon
the Company’s ability to repay the advance from either future revenues or investment proceeds. The amount was paid in full on May
24, 2022 and the amount owed to ZipDoctor was $0 and $70 as of December 31, 2023 and 2022, respectively. Imputed interest at eight percent
(8%) per annum on this advance was insignificant and therefore was not calculated, recorded or paid during the time the advance was outstanding
from December 10, 2021 to May 24, 2022.
For
additional information on related party prepaid expenses see Note 3.
NOTE
7 – NOTES PAYABLE
On
November 18, 2022, the Company entered into a note payable with a vendor for the purchase of equipment in the amount of $78,260. The
note bears no interest and was due in three payments of $5,000 each January 1, 2023 through March 1, 2023, a $31,630 payment on April
1, 2023 and a final payment on May 1, 2023 for the outstanding balance. The January 1 and March 1, 2023 payments were timely made and
on March 23, 2023, the Company elected to pay off the remaining balance of $63,260. The outstanding balance as of December 31, 2023 and
2022 was $0 and 78,260, respectively.
NOTE
8 – CAPITAL STOCK
Preferred
Stock
The
Company is authorized to issue up to 10,000,000 shares of “blank check” preferred stock, $0.0001 par value. All preferred
stock were undesignated as of December 31, 2023 and 2022.
Common
Stock
The
Company is authorized to issue 200,000,000 shares of common stock, par value $0.0001 per share, of which 21,419,500 shares were issued
and outstanding at December 31, 2023 and 13,365,000 were issued and outstanding at December 31, 2022.
On
January 3, 2023, we entered into a Consulting Agreement with DojoLabs Group, Inc. (“DojoLabs”), to provide various strategic
marketing related services to the Company pursuant to a defined scope of work during the term of the agreement, which is the earlier
of a) all deliverables being received by the Company pursuant to the scope of work, or b) if terminated due to breach of the agreement
by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing to provide the
services under the agreement, the Company agreed to pay DojoLabs $100,000 in cash and issued DojoLabs 50,000 shares of restricted common
stock with registration rights and fully vest upon the completion of all work performed under the scope of work. The agreement contains
customary confidentiality and non-solicitation provisions. The shares were valued at $1.00 per share for a total of $100,000.
On
January 6, 2023, we entered into a Consulting Agreement with Bethor, Ltd. (“Bethor”), to provide strategic advisory services
to the Company during the term of the agreement, which is for 12 months unless otherwise earlier terminated due to breach of the agreement
by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing to provide the
services under the agreement, the Company issued Bethor 250,000 shares of restricted common stock with registration rights. The agreement
contains customary confidentiality and non-solicitation provisions. The shares were valued at $1.00 per share for a total of $250,000.
On
January 6, 2023, the Company established an advisory board (the “Advisory Board”) and approved and adopted a charter (the
“Advisory Board Charter”) to govern the Advisory Board. Pursuant to the Advisory Board Charter, the Advisory Board shall
be comprised of a minimum of two (2) members, all of whom shall be appointed and subject to removal by the Board of Directors at any
time. In addition to the enumerated responsibilities of the Advisory Board in the Advisory Board Charter, the primary function of the
Advisory Board is to assist the Board of Directors in its general oversight of the Company’s development of new business ventures
and strategic planning.
In
connection with the establishment of the Advisory Board, the Board of Directors appointed Dr. Brian Rudman (“Dr. Rudman”)
and Mr. Jarrett Boon (“Mr. Boon”), both of whom are independent, non-Board members and non-Company employees, to the Advisory
Board. Dr. Rudman will serve as Chairman of the Advisory Board.
In
connection with Dr. Rudman’s appointment to the Advisory Board, the Company entered into an Advisor Agreement (the “Dr. Rudman
Consulting Agreement”), dated effective January 6, 2023, with Dr. Rudman, whereby the Company agreed to issue Dr. Rudman 25,000
shares of the Company’s restricted common stock, pay Dr. Rudman $2,000 per month in cash, and reimburse Dr. Rudman for reasonable
out-of-pocket expenses, including, without limitation, travel expenses incurred by him in connection with the Company’s requests
of the performance of his duties to the Company in service on the Advisory Board. The shares were valued at $1.00 per share for a total
of $25,000.
In
connection with Mr. Boon’s appointment to the Advisory Board, the Company entered into an Advisor Agreement (the “Mr. Boon
Consulting Agreement”), dated effective January 6, 2023, with Mr. Boon, whereby the Company agreed to issue Mr. Boon 25,000 shares
of the Company’s restricted common stock and to reimburse Mr. Boon for reasonable out-of-pocket expenses, including, without limitation,
travel expenses incurred by him in connection with the Company’s requests of the performance of his duties to the Company in service
on the Advisory Board. The shares were valued at $1.00 per share for a total of $25,000.
On
January 24, 2023, we entered into Consulting Agreements with four consultants to the Company: (1) Sultan Haroon; (2) John Helfrich; (3)
Justin Baker; and (4) Maja Matthews, each of whom is also an employee of Epiq Scripts. Pursuant to the Consulting Agreements, the Consultants
agreed to provide us services related to the research, development, packaging and marketing for additional pharmaceutical and other over-the-counter
related products during the term of the agreement, which each have a term of 18 months unless otherwise earlier terminated due to breach
of the agreement by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing
to provide the services under the agreement, the Company issued an aggregate of 350,000 shares of common stock to the consultants as
follows: (1) Sultan Haroon 150,000 shares of restricted common stock; (2) John Helfrich 25,000 shares of restricted common stock; (3)
Justin Baker 25,000 shares of restricted common stock; and (4) Maja Matthews 150,000 shares of restricted common stock. The shares issued
to Haroon and Matthews vest at the rate of 50,000 shares upon entry into the agreement, 50,000 shares upon the Company’s successful
launch of a new product category, and 50,000 shares upon the Company’s successful launch of a second and additional new product
category, in each case prior to the 18-month anniversary of the applicable agreement. The shares issued to Helfrich and Baker vest at
the rate of 10,000 shares upon entry into the agreement, 7,500 shares upon the Company’s successful launch of a new product category,
and 7,500 shares upon the Company’s successful launch of a second and additional new product category, in each case prior to the
18-month anniversary of the applicable agreement. Any shares not vested by the eighteen-month anniversary of the applicable agreement
are forfeited. The agreement contains customary confidentiality and non-solicitation provisions. The shares were valued at $1.00 per
share for a total of $350,000.
On
March 22, 2023, the Company sold 1,250,000 shares of its common stock at a price of $4.00 per share to investors in connection with its
IPO for gross proceeds of $5,000,000.
On
April 24, 2023, a warrant holder exercised private placement Warrants to purchase 100,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $100,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
April 25, 2023, a warrant holder exercised private placement Warrants to purchase 100,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $100,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
April 25, 2023, a warrant holder exercised private placement Warrants to purchase 25,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $25,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
April 25, 2023, a warrant holder exercised private placement Warrants to purchase 25,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $25,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
April 25, 2023, a warrant holder exercised private placement Warrants to purchase 75,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $75,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
April 26, 2023, a warrant holder exercised private placement Warrants to purchase 100,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $100,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
May 1, 2023, a warrant holder exercised private placement Warrants to purchase 25,000 shares of common stock with an exercise price of
$1.00 per share in consideration for $25,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
and effective on May 1, 2023, the Company entered into an Employment Agreement with Mrs. Amanda Hammer (the “Employment Agreement”).
The Employment Agreement provides for Mrs. Hammer to serve as Chief Operating Officer of the Company for an initial three-year term extending
through May 1, 2026, provided that the agreement automatically renews for additional one-year terms thereafter in the event neither party
provides the other at least 60 days prior notice of their intention not to renew the terms of the agreement. The agreement provides for
Mrs. Hammer to receive an annual salary of $150,000 per year. The Employment Agreement also required the Company to grant Mrs. Hammer
a sign-on bonus of (a) 75,000 restricted shares of common stock of the Company, vested in full upon issuance, and (b) options to purchase
an additional 150,000 shares of common stock of the Company, under the Company’s 2022 Equity Incentive Plan (the “Plan”),
with an exercise price of the greater of (i) $1.10 per share; and (ii) the closing sales price of the Company’s common stock on
the Nasdaq Capital Market on the date the Employment Agreement and the grant is approved by the Board (which date was May 1, 2023), and
which exercise price was $1.00 per share, with options to purchase 50,000 shares vesting every twelve months that the Employment Agreement
is in effect, subject to the terms of the Plan. The options are exercisable for a period of ten years and are documented by a separate
option agreement entered into by the Company and Mrs. Hammer.
On
May 1, 2023, we entered into a Software Development Agreement with Redlime Solutions, Inc. (“Redlime”) to provide software
development services during the term of the agreement, which is for twelve months. In consideration for agreeing to provide the services
under the agreement, the Company agreed to pay Redlime $300,000 in cash and issue Redlime 180,000 shares of restricted common stock.
The shares were valued at $1.00 per share for a total of $180,000.
On
May 25, 2023, the Board of Directors appointed Mr. Aaron Andrew (“Mr. Andrew”), an independent, non-Board member and non-Company
employee, to the Advisory Board. In connection with Mr. Andrew’s appointment to the Advisory Board, the Company entered into an
Advisor Agreement (the “Mr. Andrew Consulting Agreement”), dated effective May 25, 2023, with Mr. Andrew, whereby the Company
agreed to issue Mr. Andrew 50,000 shares of the Company’s restricted common stock under the 2022 Plan and to reimburse Mr. Andrew
for reasonable out-of-pocket expenses, including, without limitation, travel expenses incurred by him in connection with the Company’s
requests of the performance of his duties to the Company in service on the Advisory Board. The shares were valued at $1.10 per share
for a total of $55,000.
On
June 1, 2023, we entered into a Consulting Agreement with Major Dodge (“Major”), to provide acting and production related
services to the Company during the term of the agreement, which is for 12 months unless otherwise earlier terminated due to breach of
the agreement by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing
to provide the services under the agreement, the Company issued Major 20,000 shares of restricted common stock under the 2022 Plan. The
agreement contains customary confidentiality and non-solicitation provisions. The shares were valued at $1.10 per share for a total of
$22,000.
On
June 1, 2023, we entered into a Production and Broadcasting Agreement with New To The Street Group, LLC (“New To The Street”),
to provide production, broadcasting and other marketing related services to the Company during the term of the agreement, which is for
3 months unless otherwise earlier terminated. In consideration for agreeing to provide the services under the agreement, the Company
issued New To The Street 50,000 shares of restricted common stock and agreed to pay New To The Street a monthly cash payment of $5,000.
The shares were valued at $1.10 per share for a total of $55,000.
On June 5, 2023, a warrant holder exercised private placement Warrants
to purchase 25,000 shares of common stock with an exercise price of $1.00 per share in consideration for $25,000 in cash. The shares of
common stock issuable upon exercise of the warrants were registered under the Securities Act.
On
June 6, 2023, a warrant holder exercised private placement Warrants to purchase 150,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $150,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
June 7, 2023, a warrant holder exercised private placement Warrants to purchase 75,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $75,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
June 8, 2023, a warrant holder exercised private placement Warrants to purchase 24,500 shares of common stock with an exercise price
of $1.00 per share in consideration for $24,500 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
June 21, 2023, a warrant holder exercised private placement Warrants to purchase 100,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $100,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
June 22, 2023, a warrant holder exercised private placement Warrants to purchase 100,000
shares of common stock with an exercise price
of $1.00
per share in consideration for $100,000
in cash. The shares of common stock issuable
upon exercise of the warrants were registered under the Securities Act.
On
June 27, 2023, a warrant holder exercised private placement Warrants to purchase 100,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $100,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
September 1, 2023, we entered into a service agreement with Greentree Financial Group, Inc. (“Greentree” and the “Service
Agreement”). Pursuant to the Service Agreement, Greentree agreed to perform the following services: (a) bookkeeping services for
the Company for the period from October 1, 2023 through September 30, 2024; (b) advice and assistance to the Company in connection with
the conversion of its financial reporting systems, including its projected financial statements, to a format that is consistent with
US GAAP; (c) assistance to the Company with compliance filings for the quarters ended September 30, 2023, March 31, 2024, June 30, 2024
and the year ended December 31, 2023, including the structure and entries as well as assistance with US GAAP footnotes; (d) reviewing,
and providing advice to the Company on, all documents and accounting systems relating to its finances and transactions, with the purpose
of bringing such documents and systems into compliance with US GAAP or disclosures required by the SEC; and (e) providing necessary consulting
services and support as a liaison for the Company to third party service providers, including coordination amongst the Company and its
attorneys, CPAs and transfer agent. Since February 2015, Mr. Eugene (Gene) M. Johnston, our Chief Financial Officer (who was appointed
October 1, 2022) has served as an Audit Manager for Greentree.
The
Company agreed to issue Greentree 75,000 shares of the Company’s restricted common stock upon the parties’ entry into the
agreement, and to pay Greentree $40,000 in cash, payable as follows: (a) $20,000 on or before September 30, 2023; (b) $20,000 on or before
March 31, 2024. We also agreed to reimburse Greentree for its reasonable out-of-pocket expenses incurred in connection with Greentree’s
activities under the agreement, including the reasonable fees and travel expenses for the meetings on behalf of the Company. The Service
Agreement includes customary indemnification obligations requiring the Company to indemnify Greentree and its affiliates with regard
to certain matters. The shares were valued at $1.13 per share for a total of $84,750.
On
October 1, 2023, the Company executed a Summary of Terms and Conditions (“Consulting Agreement”) with Gene Johnston (“Johnston”)
continuing his appointment as the Company’s Chief Financial Officer on a full-time basis for a term of 12 months. Pursuant to the
Consulting Agreement, the Company issued Johnston 50,000 shares of the Company’s common stock and $2,000 per month. The Consulting
Shares shall be issued under, and subject to the terms of, the Company’s 2022 Equity Incentive Plan.
On
October 10, 2023, we entered into a Consulting Agreement with Luca Consulting, LLC (“Luca”), to provide certain management
and consulting services to the Company during the term of the agreement, which is for three months unless otherwise earlier terminated
due to breach of the agreement by either party. In consideration for agreeing to provide the services under the agreement, the Company
issued 200,000 shares of the Company’s restricted common stock upon the parties’ entry into the agreement and to pay Luca
$15,000 in cash, payable as follows: (a) $5,000 on the signing of the agreement; (b) $5,000 on the tenth of each month throughout the
remainder of the agreement. The Service Agreement includes customary indemnification obligations requiring the Company to indemnify Luca
and its affiliates with regard to certain matters. The shares were valued at $0.63 per share for a total of $126,000.
On
November 1, 2023, we entered into an Influencer Agreement with Jason Szkup (“Scoop”) to promote its products or services
through social media platforms and other online channels, In consideration for agreeing to provide the services under the agreement,
the Company agreed to pay Scoop $10,000 in cash and issue 30,000 shares. The shares were valued at $0.58 per share for a total of $17,400.
The Shares shall be issued under, and subject to the terms of, the Company’s 2022 Equity Incentive Plan.
On
November 1, 2023, the Board of Directors appointed Dr. Douglas Christianson, ND (“Dr. Christianson”), an independent, non-Board
member and non-Company employee, to the Advisory Board. In connection with Dr. Christianson’s appointment to the Advisory Board,
the Company entered into an Advisor Agreement (the “Dr. Christianson Consulting Agreement”), with Dr. Christianson, whereby
the Company agreed to issue Dr. Christianson 50,000 shares. The Shares shall be issued under, and subject to the terms of, the Company’s
2022 Equity Incentive Plan. The Company will reimburse Dr. Christianson for reasonable out-of-pocket expenses, including, without limitation,
travel expenses incurred by him in connection with the Company’s requests of the performance of his duties to the Company in service
on the Advisory Board. The shares were valued at $0.58 per share for a total of $29,000.
On
November 15, 2023, we renewed a Consulting Agreement with PHX Global, LLC (“PHX”), which is owned by Peter “Casey”
Jensen, who is a member of the Board of Directors of American International. Pursuant to the Consulting Agreement, PHX agreed to provide
consulting and general business advisory services as reasonably requested by the Company during the term of the agreement, which was
for 12 months, unless otherwise earlier terminated due to breach of the agreement by either party, and the failure to cure such breach
30 days after written notice thereof. In consideration for agreeing to provide the services under the agreement, the Company issued PHX
200,000 shares of restricted common stock. The agreement contains customary confidentiality and non-solicitation provisions. The shares
were valued at $0.47 per share for a total of $94,000. PHX is a related party.
On
December 11, 2023, the Company entered into a Marketing Agreement with Marius Pharmaceuticals (“Marius”) to market and sell
KYZATREX®, an innovative FDA-approved oral Testosterone Replacement Therapy (TRT) product, under the program, ‘PRIME’
by MangoRx. During the Term, Marius grants to MangoRx a non-exclusive, non-transferable, royalty-free license to use the Marius Marks
in the United States (the “Territory”) for the sole purpose of the Permitted Purpose. The term of the initial agreement is
for two years, automatically renewable for successive one year terms, subject to certain performance targets as agreed upon each year.
As consideration for the license granted herein, MangoRx shall issue to Marius one hundred thousand (100,000) shares of the Company’s
common stock (the “Marius Shares”). The Marius Shares shall be issued to Marius upon signing of this Agreement and shall
be deemed fully earned upon signing this Agreement. The shares were valued at $0.58 per share for a total of $58,000.
On
December 19, 2023 the Company sold 4,000,000 shares of its common stock at a price of $0.30 per share to investors in connection with
a follow on offering for gross proceeds of $1,200,000.
Options:
During
the year ended December 31, 2022, the Company granted a total of 1,250,000
options to purchase shares of common stock of the Company, under the 2022 Plan, of which 750,000
were granted to Jacob Cohen, the Company’s CEO, and 500,000
were granted to Jonathan Arango, the Company’s then President and then COO, related to their respective employment agreement.
The options have an exercise price of $1.10
per share, an original life of five years and vest at the annual renewal of their employment over three
years.
On
May 1, 2023, the Company granted 150,000 options to purchase shares of common stock of the Company, under the 2022 Plan to Amanda Hammer,
the Company’s COO, related to her employment agreement. The options have an exercise price of $1.10 per share, an original life
of five years and vest at the annual renewal of their employment over three years.
On
December 28, 2023, the Company granted 1,250,000 options to purchase shares of common stock of the Company, under the 2022 Plan to Jacob
Cohen, the Company’s CEO, related to his employment agreement. The options have an exercise price of $0.32 per share, an original
life of five years and vested at the time of grant.
As
of December 31, 2023 and 2022, $624,463
and $82,267, respectively,
has been recorded and included as stock-based compensation expense on the statement of operations for the year-ended December 31,
2023 and within general and administrative expense for year-ended December 31, 2022. Mr. Cohen, Mr. Arango (former President and
Director) and Ms. Hammer are related parties.
The
following table summarizes common stock options activity: The following table summarizes common stock options activity:
SCHEDULE
OF STOCK OPTION ACTIVITY
| | |
Options | | |
Weighted Average Exercise Price | |
| December 31, 2021 | |
| - | | |
$ | - | |
| Granted | |
| 1,250,000 | | |
| 1.10 | |
| Exercised | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Outstanding, December 31, 2022 | |
| 1,250,000 | | |
$ | 1.10 | |
| Exercisable, December 31, 2022 | |
| 133,333 | | |
$ | 1.10 | |
| Outstanding, December 31, 2023 | |
| 1,250,000 | | |
$ | 1.10 | |
| | |
| | | |
| | |
| Granted | |
| 1,400,000 | | |
$ | 0.40 | |
| Exercised | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Outstanding, December 31, 2023 | |
| 2,650,000 | | |
$ | 0.73 | |
| Exercisable, December 31, 2023 | |
| 1,812,500 | | |
$ | 0.73 | |
The
weighted average exercise prices, remaining lives for options granted, and exercisable as of December 31, 2023 were as follows:
| | | |
Outstanding Options | | |
| | |
Exercisable Options | |
Options Exercise Price Per Share | | |
Shares | | |
Life (Years) | | |
Weighted Average Exercise Price | | |
Shares | | |
Weighted Average Exercise Price | |
| $ | 1.10 | | |
| 1,400,000 | | |
| 4.28 | | |
$ | 1.10 | | |
| 562,500 | | |
$ | 1.10 | |
| $ | 0.32 | | |
| 1,250,000 | | |
| 5.00 | | |
$ | 0.32 | | |
| 1,250,000 | | |
$ | 0.32 | |
As
of December 31, 2023, the fair value of options outstanding was $573,202. The aggregate initial fair value of the options measured on
the grant date of August 31, 2022, May 1, 2023 and December 28, 2023 was calculated using the Black-Scholes option pricing model based
on the following assumption:
SCHEDULE
OF FAIR VALUE ASSUMPTIONS
| Fair Value of Common Stock on measurement date | |
$ | 1.00 - 0.32 | |
| Risk free interest rate | |
| 3.83% - 3.30 | % |
| Volatility | |
| 232.05% 92.54 | % |
| Dividend Yield | |
| 0 | % |
| Expected Term | |
| 6.0 - 3.5 | |
| |
(1) |
The risk-free interest
rate was determined by management using the market yield on U.S. Treasury securities with comparable terms as of the measurement
date. |
| |
(2) |
The trading volatility
was determined by calculating the volatility of the Company’s peer group. |
| |
(3) |
The Company does not expect
to pay a dividend in the foreseeable future |
| |
(4) |
The Company, in accordance
with staff accounting bulletin (“SAB”)14-D.2, used the simplified method (plain vanilla) to determine the overall expected
term |
Warrants:
As
additional consideration in connection with the IPO, upon the closing of the IPO, we granted Boustead Securities, LLC, the representative
of the underwriters named in the Underwriting Agreement for the IPO, warrants to purchase 87,500 shares of common stock with an exercise
price of $5.00 per share, which are exercisable six months after the effective date of the registration statement filed in connection
with the IPO (March 20, 2023) and expire five years after such effectiveness date. The fair value of the warrants on the grant date was
$31,995.
As
additional consideration in connection with the follow on offering, upon the closing of the follow on offering, we granted Boustead Securities,
LLC, the representative of the underwriters named in the Underwriting Agreement for the secondary offering, warrants to purchase 280,000
shares of common stock with an exercise price of $0.38 per share, which are exercisable six months after the effective date of the registration
statement filed in connection with the follow on offering (December 19, 2023) and expire five years after such effectiveness date. The
fair value of the warrants on the grant date was $271,216.
As
of December 31, 2023 and 2022, the fair value of warrants outstanding to investors was $852,480 and $581,264, respectively.
Because the warrants vested immediately, the fair value was assessed on the grant date.
The
following table summarizes common stock warrants activity:
SCHEDULE
OF WARRANT ACTIVITY
| | |
Warrants | | |
Weighted Average Exercise Price | |
| Outstanding, December 31, 2021 | |
| - | | |
$ | - | |
| Granted | |
| 2,210,070 | | |
| 1.00 | |
| Exercised | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Cancelled | |
| (210,070 | ) | |
| 1.00 | |
| Outstanding, December 31, 2022 | |
| 2,000,000 | | |
| 1.00 | |
| Exercisable, December 31, 2022 | |
| 2,000,000 | | |
$ | 1.00 | |
| | |
| | | |
| | |
| Granted | |
| 367,500 | | |
| 1.22 | |
| Exercised | |
| (1,024,500 | ) | |
| 1.00 | |
| Expired | |
| - | | |
| - | |
| Cancelled | |
| - | | |
| - | |
| Outstanding, December 31, 2023 | |
| 1,343,000 | | |
| 1.43 | |
| Exercisable, December 31, 2023 | |
| 1,343,000 | | |
$ | 1.43 | |
The
weighted average exercise prices, remaining lives for warrants granted, and exercisable as of December 31, 2023, were as follows:
| | | |
Outstanding and Vested Warrants | |
Weighted Average Warrant
Exercise Price Per Share | | |
Shares | | |
Life (Years) | |
| $ | 1.43 | | |
| 1,343,000 | | |
| 2.07 | |
As
of December 31, 2023, warrants to purchase 1,063,000 shares of common stock are outstanding and vested, and the vested stock warrants
have a weighted average remaining life of 3.83 years.
SCHEDULE
OF FAIR VALUE ASSUMPTIONS
| Fair Value of Common Stock on measurement date | |
$ | 0.37 - $0.97 | |
| Risk-free interest rate | |
| From 2.95% to 4.00 | % |
| Volatility | |
| From 81.92% to 169.22 | % |
| Dividend Yield | |
| 0 | % |
| Expected Term | |
| 5 years | |
| |
(1) |
The risk-free interest
rate was determined by management using the market yield on U.S. Treasury securities with comparable terms as of the measurement
date. |
| |
(2) |
The trading volatility
was determined by calculating the volatility of the Company’s peer group. |
| |
(3) |
The Company does not expect
to pay a dividend in the foreseeable future. |
NOTE
9 – GOING CONCERN
These
financial statements have been prepared assuming that the Company will continue as a going concern, which contemplates the realization
of assets and the discharge of liabilities in the normal course of business for the foreseeable future. As reflected in the accompanying
financials, the Company had a net loss of $9,212,417 for the year ended December 31, 2023 and an accumulated deficit of $11,228,173 as
of December 31, 2023. The Company will need to raise additional capital to successfully execute its business plan of which there can
be no assurance. The sources of this capital are expected to be the sale of equity and debt, which may not be available on favorable
terms, if at all, and may, if sold, cause significant dilution to existing shareholders. If we are unable to access additional capital
moving forward, it may hurt our ability to grow and to generate future revenues, our financial position, and liquidity, or force us to
abandon our business plan. These factors raise substantial doubt about the ability of the Company to continue as a going concern. Unless
management is able to obtain additional financing, it is unlikely that the Company will be able to meet its funding requirements during
the next 12 months. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
NOTE
10 – COMMITMENTS AND CONTINGENCIES
In
the ordinary course of business, the Company may become a party to lawsuits involving various matters. The impact and outcome of litigation,
if any, is subject to inherent uncertainties, and an adverse result in these or other matters may arise from time to time that may harm
its business. The Company is not currently subject to any such litigation.
Operating
Leases
The
Company has a lease for an office in Dallas, Texas classified as operating leases under ASC 842.
On
September 28, 2022, and with an effective date of October 1, 2022, the Company entered into a Lease Agreement with Rox Trep Tollway,
L.P. (the “Landlord”) to lease and occupy approximately 2,201 square feet of office space located at 15110 Dallas Parkway,
Suite 600, Dallas, Texas 75248 to serve as the Company’s main headquarters (the “Lease Agreement”). The Lease
Agreement has a term of thirty-eight (38) months and has a monthly base rent of $5,777.63, or $31.50 per square foot, the from months
3-18 and increases at the rate of $1 per square foot per annum until the end of the lease term (the “Base Rent”).
In addition to the Base Rent, the Company is required to reimburse the landlord for its pro-rata share of all real estate taxes and assessments,
hazard and liability insurance and common area maintenance costs for the building at the rate of 2.45% (the “Proportionate Rent”).
Upon the execution of the Lease Agreement, the Company agreed to prepay the first full month’s Base Rent along with a security
deposit equal to $16,942.
The
Company utilizes the incremental borrowing rate in determining the present value of lease payments unless the implicit rate is readily
determinable. The Company used an estimated incremental borrowing rate of 8% to estimate the present value of the right-of-use liability.
The
Company has right-of-use assets of $119,262
and operating lease liabilities of $128,679
as of December 31, 2023. Operating lease expense
for the year ended December 31, 2023 was $65,274.
The Company has recorded $0 in
impairment charges related to right-of-use assets during the year ended December 31, 2023.
SCHEDULE
OF MATURITY OF LEASE LIABILITIES
| Maturity of Lease Liabilities at December 31, 2023 | |
Amount | |
| 2024 | |
$ | 71,716 | |
| 2025 | |
| 67,589 | |
| Later years | |
| - | |
| Total lease payments | |
| 139,305 | |
| Less: Imputed interest | |
| (10,626 | ) |
| Present value of lease liabilities | |
$ | 128,679 | |
NOTE
11 - INCOME TAXES
On
December 22, 2017, the U.S. enacted the Tax Cuts and Jobs Act (the “Act”), which significantly changed U.S. tax law. The
Act lowered the Company’s U.S. statutory federal income tax rate from 35% to 21% effective January 1, 2018, while also imposing
a deemed repatriation tax on previously deferred foreign income.
The
Act also created a new minimum tax on certain future foreign earnings. The impact of the Act increased the Company’s deferred tax
asset related to the Company’s net operating loss by approximately $9,212,417 and increased the Company’s valuation allowance
by approximately $9,212,417 resulting in no impact to the Company’s financials.
We
record tax positions as liabilities in accordance with ASC 740 and adjust these liabilities when our judgment changes as a result of
the evaluation of new information not previously available. Because of the complexity of some of these uncertainties, the ultimate resolution
may result in a payment that is materially different from our current estimate of the recognized tax benefit liabilities. These differences
will be reflected as increases or decreases to income tax expense in the period in which new information is available. As of December
31, 2023, and 2022 we have not recorded any uncertain tax positions in our financial statements.
The
effective US Federal Income Corporate Tax Rates for 2023 and 2022 are 21% and 21%, respectively.
The
Company has net operating loss carryforwards of approximately $11,228,173 at December 31, 2023 that do not expire. However, utilization
of these losses may be limited pursuant to Section 382 of the Internal Revenue Code due to subsequent stock issuances.
The
Company has a deferred tax asset as shown in the following:
SCHEDULE
OF DEFERRED TAX ASSET
| | |
Year Ending December 31, 2023 | | |
Year Ending December 31, 2022 | |
| Deferred Tax Asset | |
| 11,228,173 | | |
| 2,015,756 | |
| Valuation Allowance | |
| (11,228,173 | ) | |
| (2,015,756 | ) |
| Net Deferred Tax Asset | |
$ | — | | |
$ | — | |
NOTE
12 – SUBSEQUENT EVENTS
On
January 2, 2024, we entered into a Consulting Agreement with G&P General Consulting (“G&P”), Pursuant to the Consulting
Agreement, G&P agreed to provide consulting and general business advisory services as it relates to the expansion of the Company’s
products into additional international territory’s, including, but not limited to, the United Arab Emirates (UAE), China, Japan,
Korea, and in certain regions of Asia and additional services as reasonably requested by the Company during the Term of this Agreement
as reasonably requested by the Company during the term of the agreement, which was for 12 months, unless otherwise earlier terminated
due to breach of the agreement by either party, and the failure to cure such breach 30 days after written notice thereof. In consideration
for agreeing to provide the services under the agreement, the Company issued G&P 250,000 shares of restricted common stock. G&P
will receive an additional 500,000 shares in 90 days, if the agreement is still in place. The Consulting Shares shall be issued under,
and subject to the terms of, the Company’s 2022 Equity Incentive Plan. The agreement contains customary confidentiality and non-solicitation
provisions. The shares were valued at $0.28 per share for a total of $70,000.
On
January 10, 2024, we renewed a Consulting Agreement with Luca Consulting, LLC (“Luca”), to provide certain management and
consulting services to the Company during the term of the agreement, which is for three months unless otherwise earlier terminated due
to breach of the agreement by either party. In consideration for agreeing to provide the services under the agreement, the Company issued
200,000 shares of the Company’s restricted common stock upon the parties’ entry into the agreement and to pay Luca $15,000
in cash, payable as follows: (a) $5,000 on the signing of the agreement; (b) $5,000 on the tenth of each month throughout the remainder
of the agreement. The Service Agreement includes customary indemnification obligations requiring the Company to indemnify Luca and its
affiliates with regard to certain matters. The shares were valued at $0.28 per share for a total of $56,000.
On
January 11, 2024, we entered into a Consulting Agreement with First Level Capital (“First Level”), to provide certain management
and consulting services to the Company during the term of the agreement, which is for six months unless otherwise earlier terminated
due to breach of the agreement by either party. In consideration for agreeing to provide the services under the agreement, the Company
issued an initial 250,000 shares of the Company’s restricted common stock upon the parties’ entry into the agreement, an
additional 250,000 shares of the Company’s restricted common stock before the end of the term of the agreement and to pay First
Level $60,000 in cash, payable as follows: (a) $60,000 on the signing of the agreement; (b) $60,000 on the approval by the Company. The
Service Agreement includes customary indemnification obligations requiring the Company to indemnify First Level and its affiliates with
regard to certain matters. The initial shares were valued at $0.28 per share for a total of $70,000.
On
January 18, 2024, the Underwriters in the follow-on offering notified the Company that they were exercising their over-allotment option
in full to purchase an additional 600,000 shares of common stock, which sale closed on January 22, 2024. The net proceeds to the Company
from the sale of the 600,000 shares of common stock, after deducting underwriting discounts and expenses, was approximately $160,000.
Inclusive of the full exercise of the over-allotment option, a total of 4,600,000 shares of common stock were issued and sold in the
follow-on offering.
On
January 22, 2024, pursuant to the Underwriting Agreement, the Company also issued a common stock purchase warrant to the Representative
for the purchase of 42,000 shares of its common stock at an exercise price of $0.375, subject to adjustments (the “Warrant”).
The Warrant is exercisable at any time and from time to time, in whole or in part, until December 14, 2028, and may be exercised on a
cashless basis. The Warrant also includes customary anti-dilution provisions and immediate piggyback registration rights with respect
to the registration of the shares underlying the Warrant. The Warrant and the shares of common stock underlying the Warrant were registered
as a part of the follow-on registration statement.
On March 21, 2024, we entered into Amendment to the
of January 10, 2024 consulting agreement with Luca extending the agreement for an additional 6 months (the “Luca Amendment”).
In consideration for entering into the Luca Amendment, the Company issued 500,000 shares of the Company’s restricted common stock
upon the parties’ entry into the Luca Amendment and agreed to continue to pay Luca $5,000 in in cash on the tenth of each month
throughout the remainder of the agreement. The shares were valued at $0.1975 per share for a total of $98,750.
On March 21, 2024, we entered into a Consulting Agreement
with Zvonimir Moric (the “Zee”). Pursuant to the consulting agreement, Zee agreed to provide consulting and general business
advisory services as it relates to making introductions to strategic partners to expand the sales of the Company’s products and
additional services as reasonably requested by the Company during the Term of this Agreement as reasonably requested by the Company during
the term of the agreement, which is for 12 months, unless otherwise earlier terminated due to breach of the agreement by either party,
and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing to provide the services under
the agreement, the Company issued Zee 150,000 shares of restricted common stock. The Consulting Shares were issued under, and subject
to the terms of, the Company’s 2022 Equity Incentive Plan. The agreement contains customary confidentiality and non-solicitation
provisions. The shares were valued at $0.1975 per share for a total of $29,625.
On
March 1, 2024, the Company borrowed $37,500 from Ronin Equity Partners, which is owned and controlled by Jacob D. Cohen, the Company’s
Chief Executive Officer and Chairman. The amount borrowed is payable on demand and does not accrue interest.
On
March 18, 2024, the Company borrowed $50,000 from Cohen Enterprises, Inc. , which is owned and controlled by Jacob D. Cohen, the Company’s
Chief Executive Officer and Chairman. The amount borrowed is payable on demand and does not accrue interest.
On
March 25, 2024, at a Special Meeting of the stockholders of the Company, the
stockholders of the Company approved a First Amendment to the Mangoceuticals, Inc. 2022 Equity Incentive Plan (“First Amendment”
and the Mangoceuticals, Inc. 2022 Equity Incentive Plan, as amended by the First Amendment, the “2022 Plan”). The
First Amendment was originally approved by the Board of Directors of the Company on February 26, 2024, subject to stockholder approval
and the First Amendment became effective at the time of stockholder approval. The First Amendment increased the number of shares of common
stock available for awards under the Incentive Plan, such that currently, subject to adjustment in connection with the payment of a stock
dividend, a stock split or subdivision or combination of the shares of common stock, or a reorganization or reclassification of the Company’s
common stock, the aggregate number of shares of common stock which may be issued pursuant to awards under the 2022 Plan is currently
the sum of (i) 10,000,000, and (ii) an automatic increase on April 1st of each year for a period of nine years commencing on April 1,
2024 and ending on (and including) April 1, 2032, in an amount equal to the lesser of (x) ten percent (10%) of the total shares of common
stock of the Company outstanding on the last day of the immediately preceding fiscal year; and (y) 2,000,000 shares of common stock;
provided, however, that the Board may act prior to April 1st of a given year to provide that the increase for such year will be a lesser
number of shares of common stock. This is also known as an “evergreen” provision. Notwithstanding the foregoing, no more
than a total of 26,000,000 shares of common stock (or awards) may be issued or granted under the 2022 Plan in aggregate, and no more
than 26,000,000 shares of common stock may be issued pursuant to the exercise of Incentive Stock Options.
Also
at the Special Meeting, the stockholders approved an amendment to the Company’s Second Amended and Restated Certificate of Incorporation,
as amended, to effect a reverse stock split of the Company’s issued and outstanding shares of our common stock, par value $0.0001
per share, by a ratio of between one-for-two to one-for-fifty inclusive, with the exact ratio to be set at a whole number to be determined
by the Company’s Board of Directors or a duly authorized committee thereof in its discretion, at any time after approval of the
amendment and prior to March 25, 2025. No formal determination has been made by the Board of Directors of the Company regarding the reverse
stock split ratio, whether or not to move forward with a reverse stock split, or the timing thereof.
30,014,286
Shares of Common Stock

Mangoceuticals,
Inc.
PROSPECTUS
,
2024
PART
II
Information
Not Required in the Prospectus
Item
13. Other Expenses of Issuance and Distribution.
The following table sets forth costs and expenses payable by us in
connection with the sale and distribution of the securities being registered. All of the amounts shown are estimates, except for the SEC
registration fee:
| | |
Amount to be Paid | |
| SEC registration fee | |
$ | 1,265 | |
| Legal fees and expenses | |
| 20,000 | |
| Accounting expenses | |
| | |
| Total expenses | |
$ | | |
Item
14. Indemnification of Directors and Officers.
As
authorized by Chapter 8 of the Texas Business Organizations Code, we may indemnify our officers and directors (and our former officers
and directors) against expenses incurred by such persons in connection with any (A) threatened, pending, or completed action or other
proceeding, whether civil, criminal, administrative, arbitrative, or investigative; (B) an appeal of an action or proceeding described
by (A); and (C) an inquiry or investigation that could lead to an action or proceeding described by (A), involving such persons in their
capacities as officers and directors, if it is determined in accordance with the Texas Business Organizations Code that: (1) the person:
(A) acted in good faith; (B) reasonably believed: (i) in the case of conduct in the person’s official capacity, that the person’s
conduct was in the enterprise’s best interests; and (ii) in any other case, that the person’s conduct was not opposed to
the enterprise’s best interests; and (C) in the case of a criminal proceeding, did not have a reasonable cause to believe the person’s
conduct was unlawful; (2) with respect to expenses, the amount of expenses other than a judgment is reasonable; and (3) indemnification
should be paid.
Under
Texas law, corporations may also purchase and maintain insurance or make other financial arrangements on behalf of any person who is
or was a director or officer (or is serving at our request as a director or officer of another corporation) for any liability asserted
against such person and any expenses incurred by him in his capacity as a director or officer.
Additionally,
our Bylaws (“Bylaws”), state that we shall indemnify every (i) present or former director, advisory director or officer of
us, (ii) any person who while serving in any of the capacities referred to in clause (i) served at our request as a director, officer,
partner, venturer, proprietor, trustee, employee, agent or similar functionary of another foreign or domestic corporation, partnership,
joint venture, trust, employee benefit plan or other enterprise, and (iii) any person nominated or designated by (or pursuant to authority
granted by) the Board of Directors or any committee thereof to serve in any of the capacities referred to in clauses (i) or (ii) (each
an “Indemnitee”).
Our
Bylaws provide that we shall indemnify an Indemnitee against all judgments, penalties (including excise and similar taxes), fines, amounts
paid in settlement and reasonable expenses actually incurred by the Indemnitee in connection with any proceeding in which he was, is
or is threatened to be named as a defendant or respondent, or in which he was or is a witness without being named a defendant or respondent,
by reason, in whole or in part, of his serving or having served, or having been nominated or designated to serve, if it is determined
that the Indemnitee (a) conducted himself in good faith, (b) reasonably believed, in the case of conduct in his official capacity, that
his conduct was in our best interests and, in all other cases, that his conduct was at least not opposed to our best interests, and (c)
in the case of any criminal proceeding, had no reasonable cause to believe that his conduct was unlawful; provided, however, that in
the event that an Indemnitee is found liable to us or is found liable on the basis that personal benefit was improperly received by the
Indemnitee, the indemnification (i) is limited to reasonable expenses actually incurred by the Indemnitee in connection with the proceeding
and (ii) shall not be made in respect of any proceeding in which the Indemnitee shall have been found liable for willful or intentional
misconduct in the performance of his duty to us.
Except
as provided above, the Bylaws provide that no indemnification shall be made in respect to any proceeding in which such Indemnitee has
been (a) found liable on the basis that personal benefit was improperly received by him, whether or not the benefit resulted from an
action taken in the Indemnitee’s official capacity, or (b) found liable to us. The termination of any proceeding by judgment, order,
settlement or conviction, or on a plea of nolo contendere or its equivalent, is not of itself determinative that the Indemnitee did not
meet the requirements set forth in clauses (a) or (b) above. An Indemnitee shall be deemed to have been found liable in respect of any
claim, issue or matter only after the Indemnitee shall have been so adjudged by a court of competent jurisdiction after exhaustion of
all appeals therefrom. Reasonable expenses shall include, without limitation, all court costs and all fees and disbursements of attorneys’
fees for the Indemnitee. The indemnification provided shall be applicable whether or not negligence or gross negligence of the Indemnitee
is alleged or proven.
Neither
our Bylaws nor our Certificate of Formation, as amended, include any specific indemnification provisions for our officers or directors
against liability under the Securities Act. Additionally, insofar as indemnification for liabilities arising under the Securities Act
may be permitted to directors, officers and controlling persons of the Company pursuant to the foregoing provisions, or otherwise, the
Company has been advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy
as expressed in the Securities Act and is, therefore, unenforceable.
Item
15. Recent Sales of Unregistered Securities.
There
have been no sales of unregistered securities during the quarter ended December 31, 2023, and from the period from January 1, 2024 to
the filing date of this registration statement, except as set forth below:
On
October 10, 2023, we entered into a Consulting Agreement with Luca Consulting, LLC (“Luca”), to provide certain management
and consulting services to the Company during the term of the agreement, which is for three months unless otherwise earlier terminated
due to breach of the agreement by either party. In consideration for agreeing to provide the services under the agreement, the Company
issued 200,000 shares of the Company’s restricted common stock upon the parties’ entry into the agreement and to pay Luca
$15,000 in cash, payable as follows: (a) $5,000 on the signing of the agreement; (b) $5,000 on the tenth of each month throughout the
remainder of the agreement. The Service Agreement includes customary indemnification obligations requiring the Company to indemnify Luca
and its affiliates with regard to certain matters. The shares were valued at $0.63 per share for a total of $126,000.
On
January 10, 2024, we renewed a Consulting Agreement with Luca Consulting, LLC (“Luca”), to provide certain management and
consulting services to the Company during the term of the agreement, which is for three months unless otherwise earlier terminated due
to breach of the agreement by either party. In consideration for agreeing to provide the services under the agreement, the Company issued
200,000 shares of the Company’s restricted common stock upon the parties’ entry into the agreement and to pay Luca $15,000
in cash, payable as follows: (a) $5,000 on the signing of the agreement; (b) $5,000 on the tenth of each month throughout the remainder
of the agreement. The Service Agreement includes customary indemnification obligations requiring the Company to indemnify Luca and its
affiliates with regard to certain matters. The shares were valued at $0.28 per share for a total of $56,000.
On
January 11, 2024, we entered into a Consulting Agreement with First Level Capital (“First Level”), to provide certain management
and consulting services to the Company during the term of the agreement, which is for six months unless otherwise earlier terminated
due to breach of the agreement by either party. In consideration for agreeing to provide the services under the agreement, the Company
issued an initial 250,000 shares of the Company’s restricted common stock upon the parties’ entry into the agreement, an
additional 250,000 shares of the Company’s restricted common stock before the end of the term of the agreement and to pay First
Level $60,000 in cash, payable as follows: (a) $60,000 on the signing of the agreement; (b) $60,000 on the approval by the Company. The
Service Agreement includes customary indemnification obligations requiring the Company to indemnify First Level and its affiliates with
regard to certain matters. The initial shares were valued at $0.28 per share for a total of $70,000.
On
March 21, 2024, we entered into Amendment to the of January 10, 2024 consulting agreement with Luca Consulting, LLC (“Luca”)
extending the agreement for an additional 6 months (the “Luca Amendment”). In consideration for entering into the Luca Amendment,
the Company issued 500,000 shares of the Company’s restricted common stock upon the parties’ entry into the Luca Amendment
and agreed to continue to pay Luca $5,000 in in cash on the tenth of each month throughout the remainder of the agreement. The shares
were valued at $0.1975 per share for a total of $98,750. The issuance described above was exempt from registration pursuant to Section
4(a)(2), and/or Rule 506 of Regulation D of the Securities Act, since the foregoing issuance did not involve a public offering, the recipient
took the securities for investment and not resale, we took take appropriate measures to restrict transfer, and the recipient was (a)
an “accredited investor”; and/or (b) had access to similar documentation and information as would be required in a Registration
Statement under the Securities Act. The securities are subject to transfer restrictions, and the certificates evidencing the securities
contain an appropriate legend stating that such securities have not been registered under the Securities Act and may not be offered or
sold absent registration or pursuant to an exemption therefrom. The securities were not registered under the Securities Act and such
securities may not be offered or sold in the United States absent registration or an exemption from registration under the Securities
Act and any applicable state securities laws.
The
issuances described above were exempt from registration pursuant to Section 4(a)(2), and/or Rule 506 of Regulation D of the Securities
Act, since the foregoing issuances did not involve a public offering, the recipients took the securities for investment and not
resale, we took take appropriate measures to restrict transfer, and the recipients were (a) “accredited investors”; and/or
(b) had access to similar documentation and information as would be required in a Registration Statement under the Securities Act. The
securities are subject to transfer restrictions, and the certificates evidencing the securities contain an appropriate legend stating
that such securities have not been registered under the Securities Act and may not be offered or sold absent registration or pursuant
to an exemption therefrom. The securities were not registered under the Securities Act and such securities may not be offered or sold
in the United States absent registration or an exemption from registration under the Securities Act and any applicable state securities
laws.
Item
16. Exhibits and Financial Statement Schedules.
(a)
Exhibits.
The
following documents are filed as exhibits to this registration statement.
Exhibit
Index
| |
|
|
|
Filed/ |
|
Incorporated
by Reference |
| Exhibit |
|
Description
of |
|
Furnished |
|
|
|
|
|
Filing |
|
File |
| Number |
|
Exhibit |
|
Herewith |
|
Form |
|
Exhibit |
|
Date |
|
Number |
| 3.1 |
|
Certificate
of Formation of Mangoceuticals, Inc., filed with the Secretary of State of Texas on October 7, 2021 |
|
|
|
S-1 |
|
3.1 |
|
1/13/2023 |
|
333-269240 |
| 3.2 |
|
Certificate
of Amendment to Certificate of Formation of Mangoceuticals, Inc., filed with the Secretary of State of Texas on April 15, 2022 |
|
|
|
S-1 |
|
3.2 |
|
1/13/2023 |
|
333-269240 |
| 3.3 |
|
Certificate
of Designations, Preferences and Rights of Series B Convertible Preferred Stock of Mangoceuticals, Inc., submitted to the Secretary
of State of Texas on March 28, 2024 |
|
|
|
10-K |
|
3.3 |
|
4/1/2024 |
|
001-41615 |
| 3.4 |
|
Certificate of Correction to Certificate of Designations, Preferences and Rights of 6% Series C Convertible Preferred Stock of Mangoceuticals, Inc., filed with the Secretary of State of Texas on April 29, 2024 |
|
|
|
8-K |
|
3.1 |
|
5/2/2024 |
|
001-41615 |
| 3.5 |
|
Bylaws
of Mangoceuticals, Inc. |
|
|
|
S-1 |
|
3.3 |
|
1/13/2023 |
|
333-269240 |
| 4.1 |
|
Common
Stock Purchase Warrant granted to Boustead Securities, LLC evidencing the right to acquire 87,500 shares of common stock (dated March
23, 2023) |
|
|
|
10-Q |
|
4.1 |
|
5/10/2023 |
|
001-41615 |
| 4.2 |
|
Form
of Common Stock Purchase Warrant (Investors - 2022 Private Placement) |
|
|
|
S-1 |
|
4.2 |
|
1/13/2023 |
|
333-269240 |
| 4.3 |
|
Common
Stock Purchase Warrant issued by Mangoceuticals, Inc. to Boustead Securities, LLC on December 19, 2023 |
|
|
|
8-K |
|
4.1 |
|
12/19/2023 |
|
001-41615 |
| 4.4 |
|
Common Stock Purchase Warrant issued by Mangoceuticals, Inc. to Boustead Securities, LLC on January 22, 2024 |
|
|
|
8-K |
|
4.1 |
|
1/22/2024 |
|
001-41615 |
| 4.5 |
|
Common
Share Purchase Warrant dated April 4, 2024 (3,300,000 shares), granted to Platinum Point Capital LLC |
|
|
|
8-K |
|
4.1 |
|
4/11/2024 |
|
001-41615 |
| 5.1 |
|
Opinion
of Lucosky Brookman LLP. |
|
|
|
S-1 |
|
5.1 |
|
4/23/2024 |
|
333-278888 |
| 10.1 |
|
Physician
Services Agreement dated August 1, 2022, between Mangoceuticals, Inc. and BrighterMD, LLC dba Doctegrity |
|
|
|
S-1 |
|
10.3 |
|
1/13/2023 |
|
333-269240 |
| 10.2£ |
|
Master
Services Agreement and Statement of Work dated September 1, 2022, and effective August 31, 2022, between Epiq Scripts, LLC and Mangoceuticals,
Inc. |
|
|
|
S-1 |
|
10.4£ |
|
1/13/2023 |
|
333-269240 |
| 10.3# |
|
Executive
Employment Agreement dated August 31, 2022, between Mangoceuticals, Inc. and Jacob D. Cohen |
|
|
|
S-1 |
|
10.5# |
|
1/13/2023 |
|
333-269240 |
| 10.4# |
|
Mangoceuticals,
Inc. 2022 Equity Incentive Plan |
|
|
|
S-1 |
|
10.7# |
|
1/13/2023 |
|
333-269240 |
| 10.5# |
|
Stock
Option Agreement dated August 31, 2022 between Mangoceuticals, Inc. and Jacob D. Cohen (750,000 option shares) |
|
|
|
S-1 |
|
10.8# |
|
1/13/2023 |
|
333-269240 |
| 10.6# |
|
Stock
Option Agreement dated August 31, 2022 between Mangoceuticals, Inc. and Jonathan Arango (500,000 option shares) |
|
|
|
S-1 |
|
10.9# |
|
1/13/2023 |
|
333-269240 |
| 10.7# |
|
Offer
Letter dated October 1, 2022 entered into between Mangoceuticals, Inc. and Eugene M. Johnston |
|
|
|
S-1 |
|
10.15# |
|
1/13/2023 |
|
333-269240 |
| 10.8# |
|
Notice
of Restricted Stock Grant and Restricted Stock Grant Agreement dated October 1, 2022 between Mangoceuticals, Inc. and Eugene M. Johnston |
|
|
|
S-1 |
|
10.16# |
|
1/13/2023 |
|
333-269240 |
| 10.9# |
|
Notice
of Restricted Stock Grant and Restricted Stock Grant Agreement dated October 14, 2022 between Mangoceuticals, Inc. and Dr. Kenny
Myers |
|
|
|
S-1 |
|
10.17# |
|
1/13/2023 |
|
333-269240 |
| 10.10# |
|
October
14, 2022 Offer Letter entered into between Mangoceuticals, Inc. and Dr. Kenny Myers |
|
|
|
S-1 |
|
10.18# |
|
1/13/2023 |
|
333-269240 |
| 10.11# |
|
Notice
of Restricted Stock Grant and Restricted Stock Grant Agreement dated October 14, 2022 between Mangoceuticals, Inc. and Alex P. Hamilton |
|
|
|
S-1 |
|
10.19# |
|
1/13/2023 |
|
333-269240 |
| 10.12# |
|
October
14, 2022 Offer Letter entered into between Mangoceuticals, Inc. and Alex P. Hamilton |
|
|
|
S-1 |
|
10.20# |
|
1/13/2023 |
|
333-269240 |
| 10.13# |
|
Notice
of Restricted Stock Grant and Restricted Stock Grant Agreement dated October 14, 2022 between Mangoceuticals, Inc. and Lorraine D’Alessio |
|
|
|
S-1 |
|
10.21# |
|
1/13/2023 |
|
333-269240 |
| 10.14# |
|
October
14, 2022 Offer Letter entered into between Mangoceuticals, Inc. and Dr. Lorraine D’Alessio |
|
|
|
S-1 |
|
10.22# |
|
1/13/2023 |
|
333-269240 |
| 10.15# |
|
Consulting
Agreement dated January 24, 2023, between Mangoceuticals, Inc. and Sultan Haroon |
|
|
|
S-1/A |
|
10.31# |
|
1/26/2023 |
|
333-269240 |
| 10.16# |
|
Consulting
Agreement dated January 24, 2023, between Mangoceuticals, Inc. and John Helfrich |
|
|
|
S-1/A |
|
10.32# |
|
1/26/2023 |
|
333-269240 |
| 10.17# |
|
Consulting
Agreement dated January 24, 2023, between Mangoceuticals, Inc. and Justin Baker |
|
|
|
S-1/A |
|
10.33# |
|
1/26/2023 |
|
333-269240 |
| 10.18# |
|
Consulting
Agreement dated January 24, 2023, between Mangoceuticals, Inc. and Maja Matthews |
|
|
|
S-1/A |
|
10.34# |
|
1/26/2023 |
|
333-269240 |
| 10.19# |
|
Employment
Agreement dated and effective May 1, 2023, by and between Mangoceuticals, Inc. and Amanda Hammer |
|
|
|
8-K |
|
10.1 |
|
5/4/2023 |
|
001-41615 |
| 10.20# |
|
Stock
Option Agreement dated May 1, 2023 between Mangoceuticals, Inc. and Amanda Hammer (150,000 option shares) |
|
|
|
8-K |
|
10.2 |
|
5/4/2023 |
|
001-41615 |
| 10.21 |
|
Service
Agreement dated September 1, 2023, by and between Mangoceuticals, Inc. and Greentree Financial Group, Inc. |
|
|
|
8-K |
|
10.1 |
|
9/8/2023 |
|
001-41615 |
| 10.22£ |
|
Master
Services Agreement and Statement of Work dated September 1, 2022, and effective August 31, 2022, between Epiq Scripts, LLC and Mangoceuticals,
Inc. |
|
|
|
8-K |
|
10.2 |
|
9/21/2023 |
|
001-41615 |
| 10.23 |
|
First
Addendum to Master Services Agreement dated September 15, 2023, by and between Mangoceuticals, Inc. and Epiq Scripts, LLC |
|
|
|
8-K |
|
10.3 |
|
9/21/2023 |
|
001-41615 |
| 10.24# |
|
Consulting
Agreement dated and effective October 3, 2023, by and between Mangoceuticals, Inc. and Eugene M. Johnston |
|
|
|
8-K |
|
10.1 |
|
10/4/2023 |
|
001-41615 |
| 10.25# |
|
Advisor
Agreement dated November 1, 2023, between Mangoceuticals, Inc. and Dr. Douglas Christianson |
|
|
|
S-1 |
|
10.43 |
|
12/11/2023 |
|
333-275993 |
| 10.26# |
|
Notice
of Restricted Stock Grant and Restricted Stock Grant Agreement dated November 1, 2023 between Mangoceuticals, Inc. and Dr. Douglas
Christianson |
|
|
|
S-1 |
|
10.44 |
|
12/11/2023 |
|
333-275993 |
| 10.27 |
|
Marketing
Agreement dated December 10, 2023, by and between Mangoceuticals, Inc. and Marius Pharmaceuticals |
|
|
|
8-K |
|
10.1 |
|
12/11/2023 |
|
001-41615 |
| 10.28# |
|
Mangoceuticals, Inc. 2022 Equity Incentive Plan Stock Option Agreement dated December 28, 2023 - Jacob Cohen - 1,250,000 shares |
|
|
|
8-K |
|
10.2 |
|
01/02/2024 |
|
001-41615 |
| 10.29# |
|
First
Amendment to the Mangoceuticals, Inc. 2022 Equity Incentive Plan |
|
|
|
8-K |
|
10.1 |
|
03/26/2024 |
|
001-41615 |
| 10.30# |
|
Amended
and Restated Mangoceuticals, Inc. 2022 Equity Incentive Plan |
|
|
|
8-K |
|
10.2 |
|
03/26/2024 |
|
001-41615 |
| 10.31 |
|
Securities
Purchase Agreement dated April 4, 2024, entered into between Mangoceuticals, Inc. and Platinum Point Capital LLC |
|
|
|
8-K |
|
10.1 |
|
4/11/2024 |
|
001-41615 |
| 10.32 |
|
Equity
Purchase Agreement dated April 4, 2024, entered into between Mangoceuticals, Inc. and Platinum Point Capital LLC |
|
|
|
8-K |
|
10.2 |
|
4/11/2024 |
|
001-41615 |
| 10.33 |
|
Registration
Rights Agreement (SPA), dated April 4, 2024, entered into between Mangoceuticals, Inc. and Platinum Point Capital LLC |
|
|
|
8-K |
|
10.3 |
|
4/11/2024 |
|
001-41615 |
| 10.34 |
|
Registration
Rights Agreement (ELOC), dated April 4, 2024, entered into between Mangoceuticals, Inc. and Platinum Point Capital LLC |
|
|
|
8-K |
|
10.4 |
|
4/11/2024 |
|
001-41615 |
| 10.35 |
|
Patent
Purchase Agreement dated April 24, 2024, by and between Mangoceuticals, Inc., as purchaser
and Intramont Technologies, Inc., as seller |
|
|
|
8-K |
|
10.1 |
|
4/25/2024 |
|
001-41615 |
| 16.1 |
|
Letter
from M&K CPAS, PLLC to the U.S. Securities and Exchange Commission dated January 26, 2023, from M&K CPAS, PLLC |
|
|
|
S-1/A |
|
16.1 |
|
1/26/2023 |
|
333-269240 |
| 21.1 |
|
Subsidiaries |
|
|
|
10-K |
|
21.1 |
|
4/1/2024 |
|
001-41615 |
| 23.1 |
|
Consent of Turner, Stone & Company, L.L.P. |
|
X |
|
|
|
|
|
|
|
|
| 23.2 |
|
Consent
of Lucosky Brookman LLP (included in Exhibit 5.1) |
|
|
|
S-1 |
|
23.2 |
|
4/23/2024 |
|
333-278888 |
| 24.1 |
|
Power
of Attorney (included on the signature page) |
|
|
|
S-1 |
|
24.1 |
|
4/23/2024 |
|
333-278888 |
| 107 |
|
Filing
Fee Table |
|
|
|
S-1 |
|
107 |
|
4/23/2024 |
|
333-278888 |
#
Indicates management contract or compensatory plan or arrangement.
£
Certain portions of these Exhibits have been omitted in accordance with Regulation S-K Item 601 because they are both (i) not material
to investors and (ii) the type of information that the Registrant customarily and actually treats as private or confidential, and have
been marked with “[***]” to indicate where omissions have been made. The Registrant agrees to furnish supplementally an unredacted
copy of the Exhibit to the SEC upon its request.
Item
17. Undertakings.
| (a) |
The
undersigned registrant hereby undertakes: |
| |
|
|
|
| |
(1) |
To
file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement: |
| |
|
|
|
| |
|
(i) |
To
include any prospectus required by section 10(a)(3) of the Securities Act of 1933; |
| |
|
|
|
| |
|
(ii) |
To
reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent
post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set
forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if
the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end
of the estimated maximum offering range may be reflected in the form of prospectus filed with the Commission pursuant to Rule 424(b)
if, in the aggregate, the changes in volume and price represent no more than 20% change in the maximum aggregate offering price set
forth in the “Calculation of Registration Fee” table in the effective registration statement. |
| |
|
|
|
| |
|
(iii) |
To
include any material information with respect to the plan of distribution not previously disclosed in the registration statement
or any material change to such information in the registration statement; provided, however, that paragraphs (a)(1)(i),
(a)(1)(ii), and (a)(1)(iii) of this section do not apply if the information required to be included in a post-effective amendment
by those paragraphs is contained in reports filed with or furnished to the SEC by the registrant pursuant to section 13 or section
15(d) of the Securities Exchange Act of 1934. |
| |
|
|
|
| |
(2) |
That,
for the purpose of determining any liability under the Securities Act of 1933, each such post-effective amendment shall be deemed
to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall
be deemed to be the initial bona fide offering thereof. |
| |
|
|
| |
(3) |
To
remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the
termination of the offering. |
| |
|
|
| |
(4) |
That,
for the purpose of determining liability under the Securities Act of 1933 to any purchaser, each prospectus filed pursuant to Rule
424(b) as part of a registration statement relating to an offering, other than registration statements relying on Rule 430B or other
than prospectuses filed in reliance on Rule 430A, shall be deemed to be part of and included in the registration statement as of
the date it is first used after effectiveness. Provided, however, that no statement made in a registration statement or prospectus
that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede
or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or
made in any such document immediately prior to such date of first use. |
| |
|
|
| |
(5) |
That,
for the purpose of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution
of the securities, the undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant
to this registration statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities
are offered or sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller
to the purchaser and will be considered to offer or sell such securities to such purchaser: |
| |
|
|
|
| |
|
(i) |
Any
preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule
424; |
| |
|
|
|
| |
|
(ii) |
Any
free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by
the undersigned registrant; |
| |
|
|
|
| |
|
(iii) |
The
portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant
or its securities provided by or on behalf of the undersigned registrant; and |
| |
|
|
|
| |
|
(iv) |
Any
other communication that is an offer in the offering made by the undersigned registrant to the purchaser. |
| |
|
|
|
| (b) |
That,
insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling
persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion
of the SEC such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In
the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred
or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding)
is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will,
unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction
the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed
by the final adjudication of such issue. |
| |
|
| (c) |
The
undersigned registrant hereby undertakes: |
| |
|
|
|
| |
(1) |
That,
for the purpose of determining any liability under the Securities Act, each post-effective amendment that contains a form of prospectus
shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities
at that time shall be deemed to be the initial bona fide offering thereof. |
| |
|
|
| |
(2) |
That,
for purposes of determining any liability under the Securities Act, the information omitted from the form of prospectus filed as
part of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant
to Rule 424(b)(1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time
it was declared effective. |
SIGNATURES
Pursuant
to the requirements of the Securities Act, the registrant has duly caused this registration statement to be signed on its behalf by the
undersigned, thereunto duly authorized, in the City of Dallas, Texas, on May 6, 2024.
| MANGOCEUTICALS,
INC. |
|
| |
|
|
| By: |
/s/
Jacob D. Cohen |
|
| Name: |
Jacob
D. Cohen |
|
| Title: |
Chairman
and Chief Executive Officer |
|
Pursuant
to the requirements of the Securities Act of 1933, this registration statement has been signed by the following persons in the capacities
held on the dates indicated.
| NAME |
|
POSITION |
|
DATE |
| |
|
|
|
|
| /s/
Jacob D. Cohen |
|
Chairman
and Chief Executive Officer |
|
May
6, 2024 |
| Jacob
D. Cohen |
|
(Principal
Executive Officer) |
|
|
| |
|
|
|
|
| /s/
Eugene M. Johnston |
|
Chief
Financial Officer |
|
May
6, 2024 |
| Eugene
M. Johnston |
|
(Principal
Financial and Accounting Officer) |
|
|
| |
|
|
|
|
| * |
|
Director |
|
May
6, 2024 |
| Lorraine
D’Alessio |
|
|
|
|
| |
|
|
|
|
| * |
|
Director |
|
May
6, 2024 |
| Alex
P. Hamilton |
|
|
|
|
| |
|
|
|
|
| * |
|
Director |
|
May
6, 2024 |
| Dr.
Kenny Myers |
|
|
|
|
| |
|
|
*
By: |
/s/
Jacob D. Cohen |
|
| |
Jacob
D. Cohen
Attorney-In-Fact |
|
Exhibit
23.1
Your
Vision Our Focus

Consent
of Independent Registered Public Accounting Firm
We
hereby consent to the use in this Registration Statement (Form S-1/A) of our report dated April 1, 2024, relating to the consolidated
financial statements of Mangoceuticals, Inc. (the “Company”), as of December 31, 2023 and 2022, and for each of the two years
in the period ended December 31, 2023, which includes an explanatory paragraph relating to the Company’s ability to continue as
a going concern, and which forms part of this Registration Statement.
We
also consent to the reference to our firm under the heading “Experts” included in such Registration Statement.
/s/
Turner, Stone & Company, L.L.P.
Dallas,
Texas
May
6, 2024
| Turner, Stone & Company, L.L.P. |
|
|
| Accountants and Consultants |
|
|
12700 Park Central Drive, Suite 1400
Dallas, Texas 75251
Telephone:972-239-1660 ⁄ Facsimile: 972-239-1665
Toll Free: 877-853-4195 |
|
 |
| Web site: turnerstone.com |
|
INTERNATIONAL ASSOCIATION OF ACCOUNTANTS AND AUDITORS |
v3.24.1.u1
Cover
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Entity Addresses [Line Items] |
|
| Document Type |
S-1/A
|
| Amendment Flag |
true
|
| Amendment Description |
AMENDMENT
NO. 1
|
| Entity Registrant Name |
Mangoceuticals,
Inc.
|
| Entity Central Index Key |
0001938046
|
| Entity Tax Identification Number |
87-3841292
|
| Entity Incorporation, State or Country Code |
TX
|
| Entity Address, Address Line One |
15110
N. Dallas Parkway
|
| Entity Address, Address Line Two |
Suite 600
|
| Entity Address, City or Town |
Dallas
|
| Entity Address, State or Province |
TX
|
| Entity Address, Postal Zip Code |
75248
|
| City Area Code |
(214)
|
| Local Phone Number |
242-9619
|
| Entity Filer Category |
Non-accelerated Filer
|
| Entity Small Business |
true
|
| Entity Emerging Growth Company |
true
|
| Elected Not To Use the Extended Transition Period |
false
|
| Business Contact [Member] |
|
| Entity Addresses [Line Items] |
|
| Entity Address, Address Line One |
15110
N. Dallas Parkway
|
| Entity Address, Address Line Two |
Suite 600
|
| Entity Address, City or Town |
Dallas
|
| Entity Address, State or Province |
TX
|
| Entity Address, Postal Zip Code |
75248
|
| City Area Code |
(214)
|
| Local Phone Number |
242-9619
|
| Contact Personnel Name |
Jacob
D. Cohen
|
| X |
- DefinitionDescription of changes contained within amended document.
| Name: |
dei_AmendmentDescription |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.
| Name: |
dei_EntityAddressesLineItems |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate whether the registrant is one of the following: Large Accelerated Filer, Accelerated Filer, Non-accelerated Filer. Definitions of these categories are stated in Rule 12b-2 of the Exchange Act. This information should be based on the registrant's current or most recent filing containing the related disclosure. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityFilerCategory |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:filerCategoryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicates that the company is a Smaller Reporting Company (SRC). Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntitySmallBusiness |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Balance Sheets - USD ($)
|
Dec. 31, 2023 |
Dec. 31, 2022 |
| CURRENT ASSETS |
|
|
| Cash and cash equivalents |
$ 739,006
|
$ 682,860
|
| Inventory |
18,501
|
|
| Prepaid expenses - related party |
60,953
|
11,745
|
| TOTAL CURRENT ASSETS |
818,460
|
694,605
|
| FIXED ASSETS |
|
|
| Property and equipment, net of accumulated depreciation of $28,752 and $3,863 |
96,129
|
117,499
|
| TOTAL FIXED ASSETS |
96,129
|
117,499
|
| OTHER ASSETS |
|
|
| Deposits |
16,942
|
16,942
|
| Right of use - asset |
119,262
|
174,241
|
| TOTAL OTHER ASSETS |
136,204
|
191,183
|
| TOTAL ASSETS |
1,050,793
|
1,003,287
|
| CURRENT LIABILITIES |
|
|
| Accounts payable and accrued liabilities |
140,765
|
33,675
|
| Payroll tax liabilities |
6,595
|
2,717
|
| Right-of-use liability - operating lease |
63,718
|
56,725
|
| TOTAL CURRENT LIABILITIES |
211,078
|
260,577
|
| LONG-TERM LIABILITIES |
|
|
| Right-of-use liability - operating lease |
64,961
|
128,680
|
| TOTAL LONG-TERM LIABILITIES |
64,961
|
128,680
|
| TOTAL LIABILITIES |
276,039
|
389,257
|
| COMMITMENTS AND CONTINGENCIES (SEE NOTE 10) |
|
|
| STOCKHOLDERS’ EQUITY |
|
|
| Common stock (par value $0.0001, 200,000,000 shares authorized, of which 21,419,500 and 13,365,000 shares issued and outstanding as of December 31, 2023 and 2022, respectively) |
2,142
|
1,337
|
| Additional paid in capital |
12,000,785
|
2,628,449
|
| Accumulated deficit |
(11,228,173)
|
(2,015,756)
|
| TOTAL STOCKHOLDERS’ EQUITY |
774,754
|
614,030
|
| TOTAL LIABILITIES AND STOCKHOLDERS’ EQUITY |
1,050,793
|
1,003,287
|
| Related Party [Member] |
|
|
| CURRENT LIABILITIES |
|
|
| Notes payable |
|
89,200
|
| Nonrelated Party [Member] |
|
|
| CURRENT LIABILITIES |
|
|
| Notes payable |
|
$ 78,260
|
| X |
- DefinitionSum of the carrying values as of the balance sheet date of obligations incurred through that date and due within one year (or the operating cycle, if longer), including liabilities incurred (and for which invoices have typically been received) and payable to vendors for goods and services received, taxes, interest, rent and utilities, accrued salaries and bonuses, payroll taxes and fringe benefits. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.19,20)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_AccountsPayableAndAccruedLiabilitiesCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionCarrying value as of the balance sheet date of obligations incurred and payable for statutory payroll taxes incurred through that date and withheld from employees pertaining to services received from them, including entity's matching share of the employees FICA taxes and contributions to the state and federal unemployment insurance programs. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer). Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.20)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_AccruedPayrollTaxesCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of excess of issue price over par or stated value of stock and from other transaction involving stock or stockholder. Includes, but is not limited to, additional paid-in capital (APIC) for common and preferred stock. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(18))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(30)(a)(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_AdditionalPaidInCapital |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionSum of the carrying amounts as of the balance sheet date of all assets that are recognized. Assets are probable future economic benefits obtained or controlled by an entity as a result of past transactions or events. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (bb)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 25
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481231/810-10-45-25
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 6: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 7: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 12
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-12
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(12))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(8))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(18))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 13: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 14: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 20: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 23: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481404/852-10-50-7
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 26: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-03(11))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479853/942-210-S99-1
| Name: |
us-gaap_Assets |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionSum of the carrying amounts as of the balance sheet date of all assets that are expected to be realized in cash, sold, or consumed within one year (or the normal operating cycle, if longer). Assets are probable future economic benefits obtained or controlled by an entity as a result of past transactions or events. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (bb)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 25
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481231/810-10-45-25
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 6: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483467/210-10-45-1
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(9))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 10: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 11: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 20: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481404/852-10-50-7
| Name: |
us-gaap_AssetsCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_AssetsCurrentAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of currency on hand as well as demand deposits with banks or financial institutions. Includes other kinds of accounts that have the general characteristics of demand deposits. Also includes short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Excludes cash and cash equivalents within disposal group and discontinued operation. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483467/210-10-45-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-4
| Name: |
us-gaap_CashAndCashEquivalentsAtCarryingValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionRepresents the caption on the face of the balance sheet to indicate that the entity has entered into (1) purchase or supply arrangements that will require expending a portion of its resources to meet the terms thereof, and (2) is exposed to potential losses or, less frequently, gains, arising from (a) possible claims against a company's resources due to future performance under contract terms, and (b) possible losses or likely gains from uncertainties that will ultimately be resolved when one or more future events that are deemed likely to occur do occur or fail to occur. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(19))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(15))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 942
-SubTopic 210
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-03.17)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479853/942-210-S99-1
Reference 4: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.25)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommitmentsAndContingencies |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAggregate par or stated value of issued nonredeemable common stock (or common stock redeemable solely at the option of the issuer). This item includes treasury stock repurchased by the entity. Note: elements for number of nonredeemable common shares, par value and other disclosure concepts are in another section within stockholders' equity. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(22))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionCarrying value of amounts transferred to third parties for security purposes that are expected to be returned or applied towards payment in the future. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(17))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_DepositsAssets |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount after valuation and LIFO reserves of inventory expected to be sold, or consumed within one year or operating cycle, if longer. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483467/210-10-45-1
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(6))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_InventoryNet |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionSum of the carrying amounts as of the balance sheet date of all liabilities that are recognized. Liabilities are probable future sacrifices of economic benefits arising from present obligations of an entity to transfer assets or provide services to other entities in the future. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 25
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481231/810-10-45-25
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (bb)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 7: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 12
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-12
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(14))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 10: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 19: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481404/852-10-50-7
Reference 20: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481404/852-10-50-7
Reference 21: http://www.xbrl.org/2003/role/exampleRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 22: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.19-26)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_Liabilities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of liabilities and equity items, including the portion of equity attributable to noncontrolling interests, if any. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(25))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 3: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 4: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 5: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-03(23))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479853/942-210-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(32))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_LiabilitiesAndStockholdersEquity |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionTotal obligations incurred as part of normal operations that are expected to be paid during the following twelve months or within one business cycle, if longer. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 25
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481231/810-10-45-25
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (bb)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 7: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 5
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483467/210-10-45-5
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 10: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 19: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481404/852-10-50-7
Reference 20: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481404/852-10-50-7
Reference 21: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.21)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_LiabilitiesCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_LiabilitiesCurrentAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of obligation due after one year or beyond the normal operating cycle, if longer. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(22))
-SubTopic 10
-Topic 210
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 25
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481231/810-10-45-25
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (bb)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-3
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 9: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 18: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481404/852-10-50-7
Reference 19: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481404/852-10-50-7
Reference 20: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(23))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 21: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 201.5-02(24))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 22: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 201.5-02(25))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 23: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 201.5-02(26))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_LiabilitiesNoncurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_LiabilitiesNoncurrentAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionSum of the carrying values as of the balance sheet date of the portions of long-term notes payable due within one year or the operating cycle if longer. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.19,20)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_NotesPayableCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionPresent value of lessee's discounted obligation for lease payments from operating lease, classified as current. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479041/842-20-45-1
| Name: |
us-gaap_OperatingLeaseLiabilityCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionPresent value of lessee's discounted obligation for lease payments from operating lease, classified as noncurrent. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479041/842-20-45-1
| Name: |
us-gaap_OperatingLeaseLiabilityNoncurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of lessee's right to use underlying asset under operating lease. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479041/842-20-45-1
| Name: |
us-gaap_OperatingLeaseRightOfUseAsset |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of assets classified as other. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 12
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-12
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(10))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(17))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 4: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-03(10))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479853/942-210-S99-1
| Name: |
us-gaap_OtherAssets |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_OtherAssetsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount after accumulated depreciation, depletion and amortization of physical assets used in the normal conduct of business to produce goods and services and not intended for resale. Examples include, but are not limited to, land, buildings, machinery and equipment, office equipment, and furniture and fixtures. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-SubTopic 10
-Topic 360
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(8))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 360
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480842/942-360-50-1
| Name: |
us-gaap_PropertyPlantAndEquipmentNet |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_PropertyPlantAndEquipmentNetAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of accumulated undistributed earnings (deficit). Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 2
-Subparagraph (g)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480016/944-40-65-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 2
-Subparagraph (h)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480016/944-40-65-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480990/946-20-50-11
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(23)(a)(4))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(17))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 8: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(30)(a)(3))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_RetainedEarningsAccumulatedDeficit |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of equity (deficit) attributable to parent. Excludes temporary equity and equity attributable to noncontrolling interest. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 12
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-12
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(19))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.6-05(4))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-2
Reference 5: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(6))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(7))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 8: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 9: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 10: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 11: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 12: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(31))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 13: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(30))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 14: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 310
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SAB Topic 4.E)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480418/310-10-S99-2
| Name: |
us-gaap_StockholdersEquity |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_StockholdersEquityAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Balance Sheets (Parenthetical) - USD ($)
|
Dec. 31, 2023 |
Dec. 31, 2022 |
| Statement of Financial Position [Abstract] |
|
|
| Accumulated depreciation |
$ 28,752
|
$ 3,863
|
| Common stock, par value |
$ 0.0001
|
$ 0.0001
|
| Common stock, shares authorized |
200,000,000
|
200,000,000
|
| Common stock, shares issued |
21,419,500
|
13,365,000
|
| Common stock, shares outstanding |
21,419,500
|
13,365,000
|
| X |
- DefinitionAmount of accumulated depreciation, depletion and amortization for physical assets used in the normal conduct of business to produce goods and services. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(8)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(14))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 360
-SubTopic 10
-Section 50
-Paragraph 1
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
| Name: |
us-gaap_AccumulatedDepreciationDepletionAndAmortizationPropertyPlantAndEquipment |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionFace amount or stated value per share of common stock. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockParOrStatedValuePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe maximum number of common shares permitted to be issued by an entity's charter and bylaws. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(16)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockSharesAuthorized |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionTotal number of common shares of an entity that have been sold or granted to shareholders (includes common shares that were issued, repurchased and remain in the treasury). These shares represent capital invested by the firm's shareholders and owners, and may be all or only a portion of the number of shares authorized. Shares issued include shares outstanding and shares held in the treasury. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockSharesIssued |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of shares of common stock outstanding. Common stock represent the ownership interest in a corporation. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.6-05(4))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-2
Reference 3: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(16)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 5: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(7))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockSharesOutstanding |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_StatementOfFinancialPositionAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Statements of Operations - USD ($)
|
12 Months Ended |
Dec. 31, 2023 |
Dec. 31, 2022 |
| Revenues |
|
|
| Revenues |
$ 731,493
|
$ 8,939
|
| Cost of revenues |
154,900
|
4,089
|
| Cost of revenues - related party |
145,092
|
|
| Gross profit |
431,501
|
4,850
|
| Operating expenses |
|
|
| General and administrative expenses |
3,319,417
|
1,643,572
|
| Salary and benefits |
977,890
|
|
| Advertising and marketing |
2,097,505
|
352,860
|
| Investor relations |
1,100,465
|
|
| Stock based compensation |
2,155,114
|
|
| Total operating expenses |
9,650,391
|
1,996,432
|
| Loss from operations |
(9,218,890)
|
(1,991,582)
|
| Other (income) expense |
|
|
| Other income |
(6,473)
|
|
| Imputed interest - related party |
|
6,473
|
| Total other (income) expense |
(6,473)
|
6,473
|
| Loss before income taxes |
(9,212,417)
|
(1,998,055)
|
| Income taxes |
|
|
| Net loss |
$ (9,212,417)
|
$ (1,998,055)
|
| Basic and diluted loss per share |
|
|
| Earnings per share, basic |
$ (0.57)
|
$ (0.19)
|
| Earnings per share, diluted |
$ (0.57)
|
$ (0.19)
|
| Weighted average number of shares outstanding |
|
|
| Weighted average number of shares outstanding, Basic |
16,113,029
|
10,798,083
|
| Weighted average number of shares outstanding, Diluted |
16,113,029
|
10,798,083
|
| X |
- DefinitionThe aggregate cost of goods produced and sold and services rendered during the reporting period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 5: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 14: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
| Name: |
us-gaap_CostOfRevenue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of net income (loss) for the period per each share of common stock or unit outstanding during the reporting period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 15
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482635/260-10-55-15
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (e)(4)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-7
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-2
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 60B
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-60B
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-4
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-10
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(25))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(27))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(23))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 16: http://www.xbrl.org/2003/role/exampleRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 52
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482635/260-10-55-52
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-7
| Name: |
us-gaap_EarningsPerShareBasic |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_EarningsPerShareBasicAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_EarningsPerShareBasicOtherDisclosuresAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of net income (loss) for the period available to each share of common stock or common unit outstanding during the reporting period and to each share or unit that would have been outstanding assuming the issuance of common shares or units for all dilutive potential common shares or units outstanding during the reporting period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 15
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482635/260-10-55-15
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (e)(4)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-7
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-2
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 60B
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-60B
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-4
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(25))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(27))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(23))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 15: http://www.xbrl.org/2003/role/exampleRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 52
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482635/260-10-55-52
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-7
| Name: |
us-gaap_EarningsPerShareDiluted |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of expense for employee benefit and equity-based compensation.
| Name: |
us-gaap_EmployeeBenefitsAndShareBasedCompensation |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe aggregate total of expenses of managing and administering the affairs of an entity, including affiliates of the reporting entity, which are not directly or indirectly associated with the manufacture, sale or creation of a product or product line. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(2)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.4)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
| Name: |
us-gaap_GeneralAndAdministrativeExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAggregate revenue less cost of goods and services sold or operating expenses directly attributable to the revenue generation activity. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 6: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 17: http://www.xbrl.org/2003/role/exampleRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 31
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-31
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 19: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.1,2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
| Name: |
us-gaap_GrossProfit |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe total expense recognized in the period for promotion, public relations, and brand or product advertising. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.4)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
| Name: |
us-gaap_MarketingAndAdvertisingExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe portion of profit or loss for the period, net of income taxes, which is attributable to the parent. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-6
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-8
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 9
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-9
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-4
Reference 13: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-10
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483581/946-220-45-7
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(18))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(9))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(1)(d))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 20: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 29: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 30: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 31: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 60B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-60B
Reference 32: http://www.xbrl.org/2003/role/exampleRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 31
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-31
Reference 33: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 34: http://www.xbrl.org/2003/role/disclosureRef
-Topic 205
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483499/205-20-50-7
Reference 35: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 36: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1A
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1A
Reference 37: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1B
Reference 38: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(20))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 39: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(22))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
| Name: |
us-gaap_NetIncomeLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe aggregate amount of income or expense from ancillary business-related activities (that is to say, excluding major activities considered part of the normal operations of the business). Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.7)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
| Name: |
us-gaap_NonoperatingIncomeExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionGenerally recurring costs associated with normal operations except for the portion of these expenses which can be clearly related to production and included in cost of sales or services. Includes selling, general and administrative expense.
| Name: |
us-gaap_OperatingExpenses |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_OperatingExpensesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe net result for the period of deducting operating expenses from operating revenues. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 4: http://www.xbrl.org/2003/role/exampleRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 31
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-31
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
| Name: |
us-gaap_OperatingIncomeLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of income related to nonoperating activities, classified as other. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(7)(d))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
| Name: |
us-gaap_OtherNonoperatingIncome |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_OtherNonoperatingIncomeExpenseAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of revenue recognized from goods sold, services rendered, insurance premiums, or other activities that constitute an earning process. Includes, but is not limited to, investment and interest income before deduction of interest expense when recognized as a component of revenue, and sales and trading gain (loss). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 6: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 42
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-42
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 40
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-40
Reference 20: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 41
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-41
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 235
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-05(b)(2))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479557/942-235-S99-1
| Name: |
us-gaap_Revenues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_RevenuesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe average number of shares or units issued and outstanding that are used in calculating diluted EPS or earnings per unit (EPU), determined based on the timing of issuance of shares or units in the period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 16
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-16
| Name: |
us-gaap_WeightedAverageNumberOfDilutedSharesOutstanding |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of [basic] shares or units, after adjustment for contingently issuable shares or units and other shares or units not deemed outstanding, determined by relating the portion of time within a reporting period that common shares or units have been outstanding to the total time in that period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-10
| Name: |
us-gaap_WeightedAverageNumberOfSharesOutstandingBasic |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Statement of Changes in Stockholders' Equity (Deficit) - USD ($)
|
Common Stock [Member] |
Additional Paid-in Capital [Member] |
Retained Earnings [Member] |
Total |
| Balance at Dec. 31, 2021 |
$ 800
|
$ 181
|
$ (17,701)
|
$ (16,720)
|
| Balance, shares at Dec. 31, 2021 |
8,000,000
|
|
|
|
| Issuance of common stock for services |
$ 337
|
539,728
|
|
540,065
|
| Issuance of common stock for services, shares |
3,365,000
|
|
|
|
| Issuance of units for cash |
$ 200
|
1,999,800
|
|
2,000,000
|
| Issuance of units for cash, shares |
2,000,000
|
|
|
|
| Options and warrants vested for services |
|
234,088
|
|
234,088
|
| Warrants for services cancelled |
|
(151,821)
|
|
(151,821)
|
| Imputed interest |
|
6,473
|
|
6,473
|
| Net loss |
|
|
(1,998,055)
|
(1,998,055)
|
| Balance at Dec. 31, 2022 |
$ 1,337
|
2,628,449
|
(2,015,756)
|
614,030
|
| Balance, shares at Dec. 31, 2022 |
13,365,000
|
|
|
|
| Issuance of common stock for services |
$ 178
|
1,530,473
|
|
1,530,651
|
| Issuance of common stock for services, shares |
1,780,000
|
|
|
|
| Options and warrants vested for services |
|
624,463
|
|
624,463
|
| Net loss |
|
|
(9,212,417)
|
(9,212,417)
|
| Issuance of common stock for cash |
$ 525
|
6,199,475
|
|
6,200,000
|
| Issuance of common stock for cash, shares |
5,250,000
|
|
|
|
| Imputed interest related party loan repayment |
|
(6,473)
|
|
(6,473)
|
| Warrants exercised |
$ 102
|
1,024,398
|
|
1,024,500
|
| Warrant exercised, shares |
1,024,500
|
|
|
|
| Balance at Dec. 31, 2023 |
$ 2,142
|
$ 12,000,785
|
$ (11,228,173)
|
$ 774,754
|
| Balance, shares at Dec. 31, 2023 |
21,419,500
|
|
|
|
| X |
- DefinitionAdjustments to additional paid in capital stock imputed interest.
| Name: |
MGRX_AdjustmentsToAdditionalPaidInCapitalStockImputedInterest |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionStock issued during the period shares issuance of units for cash
| Name: |
MGRX_StockIssuedDuringThePeriodSharesIssuanceOfUnitsForCash |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionStock issued during the period shares warrant exercised.
| Name: |
MGRX_StockIssuedDuringThePeriodSharesWarrantExercised |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionStock issued during the period value issuance of units for cash.
| Name: |
MGRX_StockIssuedDuringThePeriodValueIssuanceOfUnitsForCash |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionStock issued during the period value warrant exercised.
| Name: |
MGRX_StockIssuedDuringThePeriodValueWarrantExercised |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of other increase (decrease) in additional paid in capital (APIC).
| Name: |
us-gaap_AdjustmentsToAdditionalPaidInCapitalOther |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of increase in additional paid in capital (APIC) resulting from the issuance of warrants. Includes allocation of proceeds of debt securities issued with detachable stock purchase warrants. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 470
-SubTopic 20
-Section 25
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481284/470-20-25-2
| Name: |
us-gaap_AdjustmentsToAdditionalPaidInCapitalWarrantIssued |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe portion of profit or loss for the period, net of income taxes, which is attributable to the parent. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-6
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-8
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 9
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-9
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-4
Reference 13: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-10
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483581/946-220-45-7
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(18))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(9))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(1)(d))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 20: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 29: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 30: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 31: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 60B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-60B
Reference 32: http://www.xbrl.org/2003/role/exampleRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 31
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-31
Reference 33: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 34: http://www.xbrl.org/2003/role/disclosureRef
-Topic 205
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483499/205-20-50-7
Reference 35: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 36: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1A
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1A
Reference 37: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1B
Reference 38: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(20))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 39: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(22))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
| Name: |
us-gaap_NetIncomeLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of shares issued which are neither cancelled nor held in the treasury.
| Name: |
us-gaap_SharesOutstanding |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of shares issued in lieu of cash for services contributed to the entity. Number of shares includes, but is not limited to, shares issued for services contributed by vendors and founders.
| Name: |
us-gaap_StockIssuedDuringPeriodSharesIssuedForServices |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of new stock issued during the period. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 505
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481004/946-505-50-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-03(i)(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479886/946-10-S99-3
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesNewIssues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionValue of stock issued in lieu of cash for services contributed to the entity. Value of the stock issued includes, but is not limited to, services contributed by vendors and founders.
| Name: |
us-gaap_StockIssuedDuringPeriodValueIssuedForServices |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionEquity impact of the value of new stock issued during the period. Includes shares issued in an initial public offering or a secondary public offering. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 11
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-11
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 205
-Name Accounting Standards Codification
-Section 45
-Paragraph 4
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480767/946-205-45-4
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 505
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481004/946-505-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 8: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodValueNewIssues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of equity (deficit) attributable to parent. Excludes temporary equity and equity attributable to noncontrolling interest. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 12
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-12
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(19))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.6-05(4))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-2
Reference 5: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(6))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(7))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 8: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 9: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 10: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 11: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 12: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(31))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 13: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(30))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 14: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 310
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SAB Topic 4.E)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480418/310-10-S99-2
| Name: |
us-gaap_StockholdersEquity |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
v3.24.1.u1
Statements of Cash Flows - USD ($)
|
12 Months Ended |
Dec. 31, 2023 |
Dec. 31, 2022 |
| CASH FLOWS FROM OPERATING ACTIVITIES: |
|
|
| Net loss |
$ (9,212,417)
|
$ (1,998,055)
|
| Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
| Depreciation |
24,889
|
3,863
|
| Issuance of common stock for services |
1,530,651
|
540,065
|
| Imputed interest expense |
|
6,473
|
| Other income |
(6,473)
|
|
| Options vested for stock-based compensation |
624,463
|
234,088
|
| Warrants for service cancelled |
|
(151,821)
|
| (Increase) decrease in operating assets: |
|
|
| Rent Deposits |
|
(16,942)
|
| Inventory |
(18,501)
|
|
| Prepaid expenses – related party |
(49,208)
|
(11,745)
|
| Operating lease right of use asset |
54,979
|
(174,241)
|
| (Decrease) increase in operating liabilities: |
|
|
| Accounts payable and accrued liabilities |
107,090
|
33,675
|
| Operating lease right of use liabilities |
(56,726)
|
185,405
|
| Payroll tax liabilities |
3,878
|
2,717
|
| NET CASH USED IN OPERATING ACTIVITIES |
(6,997,375)
|
(1,346,518)
|
| CASH FLOWS FROM INVESTING ACTIVITIES: |
|
|
| Purchases of property and equipment |
(3,519)
|
(43,102)
|
| NET CASH USED IN INVESTING ACTIVITIES |
(3,519)
|
(43,102)
|
| CASH FLOWS FROM FINANCING ACTIVITIES: |
|
|
| Proceeds from borrowings on notes payable to related parties |
|
75,000
|
| Repayment on notes payable |
(78,260)
|
|
| Repayment on notes payable - related parties |
(89,200)
|
(25,070)
|
| Proceeds from exercise of warrants |
1,024,500
|
|
| Proceeds from sales of common stock for cash |
6,200,000
|
2,000,000
|
| NET CASH PROVIDED BY FINANCING ACTIVITIES |
7,057,040
|
2,049,930
|
| NET INCREASE IN CASH AND CASH EQUIVALENTS |
56,146
|
660,310
|
| CASH AND CASH EQUIVALENTS: |
|
|
| Beginning of year |
682,860
|
22,550
|
| End of year |
739,006
|
682,860
|
| Supplemental disclosure of cash flow information: |
|
|
| Cash paid for income taxes |
|
|
| Cash paid for interest |
|
|
| X |
- DefinitionCash in flow from other operating activity
| Name: |
MGRX_CashInFlowFromOtherOperatingActivity |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionIncrease decrease in operating lease right of use asset.
| Name: |
MGRX_IncreaseDecreaseInOperatingLeaseRightOfUseAsset |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionIssuance of common stock for services.
| Name: |
MGRX_IssuanceOfCommonStockForServices |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionWarrants for service cancelled.
| Name: |
MGRX_WarrantsForServiceCancelled |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_AdjustmentsToReconcileNetIncomeLossToCashProvidedByUsedInOperatingActivitiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_CashAndCashEquivalentsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash and cash equivalents, and cash and cash equivalents restricted to withdrawal or usage; including, but not limited to, disposal group and discontinued operations. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits. Cash equivalents include, but are not limited to, short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482913/230-10-50-8
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 24
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-24
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-4
| Name: |
us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsIncludingDisposalGroupAndDiscontinuedOperations |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of increase (decrease) in cash, cash equivalents, and cash and cash equivalents restricted to withdrawal or usage; including effect from exchange rate change. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits. Cash equivalents include, but are not limited to, short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 24
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-24
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-SubTopic 230
-Topic 830
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481877/830-230-45-1
| Name: |
us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsPeriodIncreaseDecreaseIncludingExchangeRateEffect |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of expense recognized in the current period that reflects the allocation of the cost of tangible assets over the assets' useful lives. Includes production and non-production related depreciation. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Subparagraph (b)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 360
-SubTopic 10
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
| Name: |
us-gaap_Depreciation |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe increase (decrease) during the reporting period in moneys or securities given as security including, but not limited to, contract, escrow, or earnest money deposits, retainage (if applicable), deposits with clearing organizations and others, collateral, or margin deposits. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
| Name: |
us-gaap_IncreaseDecreaseInDepositOtherAssets |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe increase (decrease) during the reporting period in the aggregate value of all inventory held by the reporting entity, associated with underlying transactions that are classified as operating activities. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
| Name: |
us-gaap_IncreaseDecreaseInInventories |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_IncreaseDecreaseInOperatingAssetsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of increase (decrease) in obligation for operating lease. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 2: http://www.xbrl.org/2009/role/commonPracticeRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (g)(1)
-SubTopic 20
-Topic 842
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-4
| Name: |
us-gaap_IncreaseDecreaseInOperatingLeaseLiability |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_IncreaseDecreaseInOperatingLiabilitiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe increase (decrease) during the reporting period in other obligations or expenses incurred but not yet paid. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
| Name: |
us-gaap_IncreaseDecreaseInOtherAccountsPayableAndAccruedLiabilities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe increase (decrease) during the reporting period in the amount of outstanding money paid in advance for goods or services that bring economic benefits for future periods. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
| Name: |
us-gaap_IncreaseDecreaseInPrepaidExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of the cost of borrowed funds accounted for as interest expense. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-10
Reference 2: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section 45
-Paragraph 3
-Subparagraph (i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483581/946-220-45-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(3))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 835
-SubTopic 30
-Section 45
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482925/835-30-45-3
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04.9)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (210.5-03(11))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 835
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483013/835-20-50-1
| Name: |
us-gaap_InterestExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash paid for interest, including, but not limited to, capitalized interest and payment to settle zero-coupon bond attributable to accreted interest of debt discount and debt instrument with insignificant coupon interest rate in relation to effective interest rate of borrowing attributable to accreted interest of debt discount; classified as operating and investing activities. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482913/230-10-50-2
| Name: |
us-gaap_InterestPaid |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash inflow (outflow) from financing activities, including discontinued operations. Financing activity cash flows include obtaining resources from owners and providing them with a return on, and a return of, their investment; borrowing money and repaying amounts borrowed, or settling the obligation; and obtaining and paying for other resources obtained from creditors on long-term credit. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 24
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-24
| Name: |
us-gaap_NetCashProvidedByUsedInFinancingActivities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_NetCashProvidedByUsedInFinancingActivitiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash inflow (outflow) from investing activities, including discontinued operations. Investing activity cash flows include making and collecting loans and acquiring and disposing of debt or equity instruments and property, plant, and equipment and other productive assets. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 24
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-24
| Name: |
us-gaap_NetCashProvidedByUsedInInvestingActivities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_NetCashProvidedByUsedInInvestingActivitiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash inflow (outflow) from operating activities, including discontinued operations. Operating activity cash flows include transactions, adjustments, and changes in value not defined as investing or financing activities. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 24
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-24
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 25
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-25
| Name: |
us-gaap_NetCashProvidedByUsedInOperatingActivities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_NetCashProvidedByUsedInOperatingActivitiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe portion of profit or loss for the period, net of income taxes, which is attributable to the parent. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-6
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-8
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 9
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-9
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-4
Reference 13: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-10
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483581/946-220-45-7
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(18))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(9))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(1)(d))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 20: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 29: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 30: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 31: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 60B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-60B
Reference 32: http://www.xbrl.org/2003/role/exampleRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 31
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-31
Reference 33: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 34: http://www.xbrl.org/2003/role/disclosureRef
-Topic 205
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483499/205-20-50-7
Reference 35: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 36: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1A
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1A
Reference 37: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1B
Reference 38: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(20))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 39: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(22))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
| Name: |
us-gaap_NetIncomeLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash outflow associated with the acquisition of long-lived, physical assets that are used in the normal conduct of business to produce goods and services and not intended for resale; includes cash outflows to pay for construction of self-constructed assets. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 13
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-13
| Name: |
us-gaap_PaymentsToAcquirePropertyPlantAndEquipment |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash inflow from the additional capital contribution to the entity. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 14
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-14
| Name: |
us-gaap_ProceedsFromIssuanceOfCommonStock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash inflow from issuance of rights to purchase common shares at predetermined price (usually issued together with corporate debt). Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 14
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-14
| Name: |
us-gaap_ProceedsFromIssuanceOfWarrants |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash outflow for a borrowing supported by a written promise to pay an obligation. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 15
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-15
| Name: |
us-gaap_RepaymentsOfNotesPayable |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of noncash expense for share-based payment arrangement. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
| Name: |
us-gaap_ShareBasedCompensation |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
v3.24.1.u1
ORGANIZATION AND DESCRIPTION OF THE BUSINESS
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Organization, Consolidation and Presentation of Financial Statements [Abstract] |
|
| ORGANIZATION AND DESCRIPTION OF THE BUSINESS |
NOTE
1 – ORGANIZATION AND DESCRIPTION OF THE BUSINESS
Mangoceuticals,
Inc. (“Mangoceuticals” or the “Company”), was incorporated in the State of a Texas on October 7, 2021, with the
intent of focusing on developing, marketing, and selling a variety of men’s wellness products and services via a telemedicine platform.
To date, the Company has identified men’s wellness telemedicine services and products as a growing sector in the most recent years
and especially related to the areas of erectile dysfunction (“ED”), hair loss and testosterone replacement or enhancement
therapies. In this regard, Mangoceuticals has developed and is commercially marketing a new brand of ED products under the brand name
“Mango” and a new brand of hair loss products under the brand name “Grow.” These products are produced at a compounding
pharmacy using a proprietary combination of U.S. Food and Drug Administration (“FDA”) approved ingredients and is available
to patients on the determination of a prescribing physician that the compounded drug is necessary for the individual patient. Mangoceuticals
is currently marketing and selling these branded ED and hair loss products exclusively online via its website at www.MangoRx.com.
Initial
Public Offering. In March 2023, the Company completed an initial public offering (the “IPO”), in which the Company issued
and sold 1,250,000 shares of authorized common stock for $4.00 per share for net proceeds of $4.35 million, after deducting underwriting
discounts and commissions, and offering costs. At the same time, and as part of the same registration statement, but pursuant to a separate
prospectus (the “Resale Prospectus”) the Company registered the sale of 4,765,000 shares of common stock, including 2,000,000
shares of common stock issuable upon the exercise of outstanding warrants to purchase shares of common stock with an exercise price of
$1.00 per share.
|
| X |
- References
+ Details
| Name: |
us-gaap_OrganizationConsolidationAndPresentationOfFinancialStatementsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for organization, consolidation and basis of presentation of financial statements disclosure. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480424/946-10-50-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480424/946-10-50-2
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 810
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//810/tableOfContent
Reference 4: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 205
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//205/tableOfContent
| Name: |
us-gaap_OrganizationConsolidationAndPresentationOfFinancialStatementsDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Accounting Policies [Abstract] |
|
| SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES |
NOTE
2 – SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis
of Presentation – The financial statements present the financial position, results of operations and cash flows of the Company
in accordance with accounting principles generally accepted in the United States of America (“US GAAP”). All dollar amounts
are rounded to the nearest thousand dollars.
Cash
Equivalents
Highly
liquid investments with original maturities of three months or less are considered cash equivalents. The Company maintains the majority
of its cash accounts at a commercial bank. The Federal Deposit Insurance Corporation (“FDIC”) insures the total cash balance
up to $250,000 per commercial bank. From time to time, cash in deposit accounts may exceed the FDIC limits, the excess would be at risk
of loss for purposes of the statement of cash flows. There are no cash equivalents at December 31, 2023 and 2022
Income
Taxes
The
Company is a taxable entity and recognizes deferred tax assets and liabilities for the future tax consequences attributable to differences
between the financial statement carrying amounts of existing assets and liabilities and their respective tax basis. Income taxes are
provided in accordance with Financial Accounting Standards Board (“FASB”) Accounting Standards Codification (“ASC”)
740, Income Taxes. Deferred tax assets and liabilities are measured using enacted tax rates expected to be in effect when the
temporary differences reverse. The effect on the deferred tax assets and liabilities of a change in tax rates is recognized in income
in the year that includes the enactment date of the rate change. A valuation allowance is used to reduce deferred tax assets to the amount
that is more likely than not to be realized.
Net
Loss Per Common Share
We
compute net loss per share in accordance with ASC 260, Earning per Share. ASC 260 requires presentation of both basic and
diluted earnings per share (“EPS”) on the face of the statement of operations. Basic EPS is computed by dividing net loss available
to common shareholders (numerator) by the weighted average number of shares outstanding (denominator) during the period. Diluted EPS
gives effect to all dilutive potential common shares outstanding during the period using the treasury stock method and convertible
preferred stock using the if-converted method. In computing Diluted EPS, the average stock price for the period is used in
determining the number of shares assumed to be purchased from the exercise of stock options or warrants. Diluted EPS excludes all
dilutive potential shares if their effect is anti-dilutive. There were 2,650,000
options, 1,343,000
warrants and no derivative securities outstanding as of December 31, 2023. There were 1,250,000
options, 2,000,000
warrants and no derivative securities outstanding as of December 31, 2022.
Use
of Estimates and Assumptions
The
preparation of financial statements in accordance with US GAAP requires the Company’s management to make estimates and assumptions
that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial
statements and the reported amounts of expenses during the reporting period. Actual results can, and in many cases will, differ from
those estimates.
Fair
Value of Financial Instruments
The
Company measures its financial and non-financial assets and liabilities, as well as makes related disclosures, in accordance with FASB
ASC 820, Fair Value Measurement (“ASC 820”), which provides guidance with respect to valuation techniques to be utilized
in the determination of fair value of assets and liabilities. Approaches include, (i) the market approach (comparable market prices),
(ii) the income approach (present value of future income or cash flow), and (iii) the cost approach (cost to replace the service capacity
of an asset or replacement cost). ASC 820 utilizes a fair value hierarchy that prioritizes the inputs to valuation techniques used to
measure fair value into three broad levels. The following is a brief description of those three levels:
Level
1: Observable inputs such as quoted prices (unadjusted) in active markets for identical assets or liabilities.
Level
2: Inputs other than quoted prices that are observable, either directly or indirectly. These include quoted prices for similar assets
or liabilities in active markets and quoted prices for identical or similar assets or liabilities in markets that are not active.
Level
3: Unobservable inputs in which little or no market data exists, therefore requiring an entity to develop its own assumptions, such as
valuations derived from valuation techniques in which one more significant inputs or significant value drivers are unobservable.
The
following tables summarize our financial instruments measured at fair value as of December 31, 2023 and 2022.
SCHEDULE
OF FINANCIAL INSTRUMENTS MEASUREMENT AT FAIR VALUE
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| | |
Fair Value Measurements at December 31, 2023 | |
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| Assets | |
| | | |
| | | |
| | |
| Cash | |
$ | 739,006 | | |
$ | - | | |
$ | - | |
| Total assets | |
| 739,006 | | |
| - | | |
| - | |
| Liabilities | |
| | | |
| | | |
| | |
| Total liabilities | |
| - | | |
| - | | |
| - | |
| Fair value, net asset
(liability) | |
$ | 739,006 | | |
$ | - | | |
$ | - | |
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| | |
Fair Value Measurements at December 31, 2022 | |
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| Assets | |
| | | |
| | | |
| | |
| Cash | |
$ | 682,860 | | |
$ | - | | |
$ | - | |
| Total assets | |
| 682,860 | | |
| - | | |
| - | |
| Liabilities | |
| | | |
| | | |
| | |
| Total liabilities | |
| - | | |
| - | | |
| - | |
| Fair value, net asset
(liability) | |
$ | 682,860 | | |
$ | - | | |
$ | - | |
Property
and Equipment
Property
and equipment are stated at cost. When retired or otherwise disposed, the related carrying value and accumulated depreciation are removed
from the respective accounts and the net difference less any amount realized from the disposition is reflected in earnings. For financial
statement purposes, property and equipment are recorded at cost and depreciated using the straight-line method over their estimated useful
lives of three (3) to five (5) years.
Concentration
and Risks
The
Company’s operations are subject to risks including financial, operational, regulatory and other risks including the potential
risk of business failure. For the years ended December 31, 2023 and 2022, the Company had no significant revenue from continuing
operations which were derived from a single or a few major customers.
Black-Scholes Option Pricing Model
The
Company uses a Black-Scholes option pricing model to determine the fair value of warrants and options issued.
Recently
Issued Accounting Pronouncements
From
time to time, new accounting pronouncements are issued by the FASB or other standard setting bodies that are adopted by the Company as
of the specified effective date. Unless otherwise discussed, the Company believes that the effect of recently issued standards that are
not yet effective will not have a material effect on its financial position or results of operations upon adoption.
In
August 2020, the FASB issued Accounting Standards Update (“ASU”) 2020-06, Debt – Debt with Conversion and
Other Options (Subtopic 470-20) and Derivatives and Hedging – Contracts in Entity’s Own Equity (Subtopic 815 – 40) (“ASU 2020-06”). ASU 2020-06 simplifies the accounting for certain financial instruments with
characteristics of liabilities and equity, including convertible instruments and contracts on an entity’s own equity. The ASU
is part of the FASB’s simplification initiative, which aims to reduce unnecessary complexity in US GAAP. The ASU’s
amendments are effective for fiscal years beginning after December 15, 2023, and interim periods within those fiscal years. The
Company is currently evaluating the impact of ASU 2020-06 on its financial statements.
Related
Parties
The
Company follows subtopic 850-10 of FASB ASC 850, Related Party Disclosures for the identification of related parties and disclosure
of related party transactions.
Pursuant
to Section 850-10-20, the related parties include a. affiliates of the Company; b. Entities for which investments in their equity securities
would be required, absent the election of the fair value option under the guidance of Fair Value Option Subsection of Section 825–10–15,
to be accounted for by the equity method by the investing entity; c. trusts for the benefit of employees, such as pension and profit-sharing
trusts that are managed by or under the trusteeship of management; d. principal owners of the Company; e. management of the Company;
f. other parties with which the Company may deal if one party controls or can significantly influence the management or operating policies
of the other to an extent that one of the transacting parties might be prevented from fully pursuing its own separate interests; and
g. other parties that can significantly influence the management or operating policies of the transacting parties or that have an ownership
interest in one of the transacting parties and can significantly influence the other to an extent that one or more of the transacting
parties might be prevented from fully pursuing its own separate interests.
The
financial statements shall include disclosures of material related party transactions, other than compensation arrangements, expense
allowances, and other similar items in the ordinary course of business. However, disclosure of transactions that are eliminated in the
preparation of financial statements is not required in those statements. The disclosures shall include: a. the
nature of the relationship(s) involved; b. a description of the transactions, including transactions to which no amounts or nominal amounts
were ascribed, for each of the periods for which income statements are presented, and such other information deemed necessary to an understanding
of the effects of the transactions on the financial statements; c. the dollar amounts of transactions for each of the periods for which
income statements are presented and the effects of any change in the method of establishing the terms from that used in the preceding
period; and d. amounts due from or to related parties as of the date of each balance sheet presented and, if not otherwise apparent,
the terms and manner of settlement. Material related party transactions have been identified in Notes 3, 6 and 8 in the notes to financial
statements.
Stock-Based
Compensation
The
Company recognizes compensation costs to employees under FASB ASC 718 Compensation - Stock Compensation (“ASC
718”). Under ASC 718, companies are required to measure the compensation costs of share-based compensation arrangements based
on the grant-date fair value and recognize the costs in the financial statements over the period during which employees are required
to provide services. Share-based compensation arrangements include stock options and warrants. As such, compensation cost is
measured on the date of grant at their fair value. Such compensation amounts, if any, are amortized over the respective vesting
periods of the option and warrant grant.
Revenue
Recognition
The Company follows the provisions of ASC 606. Revenue from Contracts with Customer for recording and recognizing
revenue from customers. The
Company generates our online revenue through the sale of products and services purchased by customers directly through our online platform.
Online revenue represents the sales of products and services on our platform, net of refunds, credits, and chargebacks, and includes
revenue recognition adjustments recorded pursuant to US GAAP. Online revenue is generated by selling directly to consumers through our
websites.
The
Company recognizes revenue when it transfers promised goods or services to customers in an amount that reflects the consideration to
which it expects to be entitled in exchange for those goods or services and has met its performance obligation. For revenue generated
through its online platform, the Company defines its customer as an individual who purchases products or services through websites. The
transaction price in the Company’s contracts with customers is the total amount of consideration to which the Company expects to
be entitled in exchange for transferring products or services to the customer.
The
Company’s contracts that contain prescription products issued as the result of a consultation include two performance obligations:
access to (i) products and (ii) consultation services. The Company’s contracts for prescription refills have a single performance
obligation. Revenue is recognized at the time the related performance obligation is satisfied by transferring the promised product to
the customer and, in contracts that contain services, by the provision of consultation services to the customer. The Company satisfies
its performance obligation for products at a point in time, which is upon delivery of the products to a third-party carrier. The Company
satisfies its performance obligation for services over the period of the consultation service, which is typically a few days. The customer
obtains control of the products and services upon the Company’s completion of its performance obligations.
The
Company has entered into a Physician Services Agreement with BrighterMD, LLC dba Doctegrity (“Doctegrity”) to provide online
telemedicine technology services to the Company. The Company accounts for service revenue as a principal in the arrangement with its
customers. This conclusion is reached because (i) the Company determines which providers provide the consultation to the customer; (ii)
the Company is primarily responsible for the satisfactory fulfillment and acceptability of the services; (iii) the Company incurs costs
for consultation services even for visits that do not result in a prescription and the sale of products; and (iv) the Company, at its
sole discretion, sets all listed prices charged on its websites for products and services.
Additionally,
the Company has entered into a Master Services Agreement and Statement of Work with Epiq Scripts, LLC (“Contracted Pharmacy”),
which is a related party, to provide pharmacy and compounding services to the Company to fulfill its promise to customers for contracts
that include sale of prescription products and to fill prescriptions that are ordered by the Company’s customers for fulfillment
through the Company’s websites. The Company accounts for prescription product revenue as a principal in the arrangement with its
customers. This conclusion is reached because (i) the Company has sole discretion in determining which Contracted Pharmacy fills a customer’s
prescription; (ii) Contracted Pharmacy fills the prescription based on fulfillment instructions provided by the Company, including using
the Company’s branded packaging for generic products; (iii) the Company is primarily responsible to the customer for the satisfactory
fulfillment and acceptability of the order, and; (iv) the Company, at its sole discretion, sets all listed prices charged on its websites
for products and services.
The
Company accounts for shipping activities, consisting of direct costs to ship products performed after the control of a product has been
transferred to the customer, in cost of revenue.
Inventories
Inventories
are stated at the lower of cost or net realizable value with cost being determined on a first-in, first-out (“FIFO”) basis. The Company writes down its
inventory for estimated obsolescence or unmarketable inventory equal to the difference between the cost of inventory and the estimated
market value based upon assumptions about future demand and market conditions. If actual market conditions are less favorable than those
projected by management, additional inventory write-downs may be required. During the years ended December 31, 2023 and 2022, there were no inventory write-downs.
Marketing
and Advertising
The
Company follows the policy of charging the costs of marketing and advertising to expense as incurred. The Company charged $2,097,505
and $352,860 towards marketing and advertising for the years ended December 31, 2023 and 2022, respectively.
Subsequent
Events
The
Company follows the guidance in Section 855-10-50 of FASB ASC 855, Subsequent Events, for the disclosure of subsequent
events. The Company will evaluate subsequent events through the date when the financial statements were issued (see Note
12).
|
| X |
- References
+ Details
| Name: |
us-gaap_AccountingPoliciesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for all significant accounting policies of the reporting entity. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483426/235-10-50-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 235
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//235/tableOfContent
| Name: |
us-gaap_SignificantAccountingPoliciesTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
PREPAID EXPENSES-RELATED PARTIES
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Prepaid Expenses-related Parties |
|
| PREPAID EXPENSES-RELATED PARTIES |
NOTE
3 – PREPAID EXPENSES-RELATED PARTIES
During
the year ended December 31, 2023, and in association with the Master Services Agreement and Statement of Work with our related party
Contracted Pharmacy, the Company prepays the related party Contracted Pharmacy as a retainer to be credited towards future product sales.
As of December 31, 2023 and 2022, the balance was $60,953 and $11,745, respectively.
Additionally,
the Company signed a lease agreement for office space, effective October 1, 2022, which included an initial security deposit of $16,942.
|
| X |
- DefinitionPrepaid Expenses Related Parties [Text Block]
| Name: |
MGRX_PrepaidExpensesRelatedPartiesTextBlock |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
INVENTORY
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Inventory Disclosure [Abstract] |
|
| INVENTORY |
NOTE
4 – INVENTORY
During
the years ended December 31, 2023 and 2022, the Company purchased inventories related to promotional merchandise intended to be sold
online. As of December 31, 2023 and 2022, the inventory balance was $18,501 and $0, respectively.
|
| X |
- References
+ Details
| Name: |
us-gaap_InventoryDisclosureAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for inventory. Includes, but is not limited to, the basis of stating inventory, the method of determining inventory cost, the classes of inventory, and the nature of the cost elements included in inventory. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 330
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//330/tableOfContent
| Name: |
us-gaap_InventoryDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
PROPERTY, PLANT AND EQUIPMENT
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Property, Plant and Equipment [Abstract] |
|
| PROPERTY, PLANT AND EQUIPMENT |
NOTE
5 – PROPERTY, PLANT AND EQUIPMENT
During
the year ended December 31, 2023, the Company acquired computers and office equipment totaling $3,519. Depreciation for the year ended
December 31, 2023 and 2022 was $24,889 and $3,863, respectively. Total net property, plant and equipment was $96,129 and $117,499, as
of December 31, 2023 and 2022, respectively.
SCHEDULE
OF PROPERTY PLANT AND EQUIPMENT
| | |
December 31, 2023 | | |
December 31, 2022 | |
| | |
| | |
| |
| Computers | |
| 5,062 | | |
| 5,062 | |
| Equipment | |
| 119,819 | | |
| 116,300 | |
| Less accumulated depreciation: | |
| (28,752 | ) | |
| (3,863 | ) |
| Property and equipment, net | |
| 96,129 | | |
| 117,499 | |
|
| X |
- References
+ Details
| Name: |
us-gaap_PropertyPlantAndEquipmentAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for long-lived, physical asset used in normal conduct of business and not intended for resale. Includes, but is not limited to, work of art, historical treasure, and similar asset classified as collections. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 360
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//360/tableOfContent
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-SubTopic 360
-Topic 958
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480321/958-360-50-6
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (d)
-SubTopic 360
-Topic 958
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480321/958-360-50-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-SubTopic 360
-Topic 958
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480321/958-360-50-7
| Name: |
us-gaap_PropertyPlantAndEquipmentDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
LOANS FROM RELATED PARTIES
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Related Party Transactions [Abstract] |
|
| LOANS FROM RELATED PARTIES |
NOTE
6 – LOANS FROM RELATED PARTIES
On
December 10, 2021 and March 18, 2022, the Company received advances of $39,200 and $50,000, respectively, for a total of $89,200 from
its previous majority shareholder, American International Holdings Corp (“AMIH”), in order to cover various general and administrative
expenses. The advances bear no interest and are due on demand upon the Company’s ability to repay the advances from either future
revenues or investment proceeds. On June 16, 2022, Cohen Enterprises, Inc. (“Cohen Enterprises”), an entity owned and controlled
by Jacob D. Cohen, the Company’s Chief Executive Officer and Chairman of the Board of Directors, entered into and closed a Stock
Purchase Agreement (the “SPA”) for the purchase of 8,000,000 shares of the outstanding common stock of the Company which
were then held by AMIH, which represented 80% of the Company’s then outstanding shares of common stock, in consideration for $90,000.
Pursuant to the terms of the SPA, Cohen Enterprises also acquired the right to be repaid the $89,200 advanced from AMIH to the Company.
On
June 29, 2022, the Company received an advance of $25,000 from Cohen Enterprises in order to cover various general and administrative
expenses. The Company repaid Cohen Enterprises $25,000 on August 18, 2022 bringing the total amount owed to Cohen Enterprises to $89,200
as of December 31, 2022. This amount was paid in full on April 4, 2023 and the amount owed to Cohen Enterprises was $0 and $89,200 as
of December 31, 2023 and 2022, respectively. Previously recorded imputed interest equal to eight percent (8%) per annum, or a total of
$8,232 against the related party advances, was canceled and reversed for the year ended December 31, 2023.
On
December 10, 2021, the Company received an advance of $70 from ZipDoctor, Inc., a then wholly-owned subsidiary of its then majority shareholder,
AMIH, which was used to open and establish the Company’s bank account. The advance bears no interest and is due on demand upon
the Company’s ability to repay the advance from either future revenues or investment proceeds. The amount was paid in full on May
24, 2022 and the amount owed to ZipDoctor was $0 and $70 as of December 31, 2023 and 2022, respectively. Imputed interest at eight percent
(8%) per annum on this advance was insignificant and therefore was not calculated, recorded or paid during the time the advance was outstanding
from December 10, 2021 to May 24, 2022.
For
additional information on related party prepaid expenses see Note 3.
|
| X |
- DefinitionThe entire disclosure for related party transactions. Examples of related party transactions include transactions between (a) a parent company and its subsidiary; (b) subsidiaries of a common parent; (c) and entity and its principal owners; and (d) affiliates. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480990/946-20-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 5
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480990/946-20-50-5
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480990/946-20-50-6
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 235
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481062/946-235-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 235
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481062/946-235-50-2
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 850
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483326/850-10-50-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(2)(g)(3))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(2)(c))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(2)(e))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 850
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//850/tableOfContent
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 850
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483326/850-10-50-6
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 850
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483326/850-10-50-1
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 850
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483326/850-10-50-1
| Name: |
us-gaap_RelatedPartyTransactionsDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
NOTES PAYABLE
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Debt Disclosure [Abstract] |
|
| NOTES PAYABLE |
NOTE
7 – NOTES PAYABLE
On
November 18, 2022, the Company entered into a note payable with a vendor for the purchase of equipment in the amount of $78,260. The
note bears no interest and was due in three payments of $5,000 each January 1, 2023 through March 1, 2023, a $31,630 payment on April
1, 2023 and a final payment on May 1, 2023 for the outstanding balance. The January 1 and March 1, 2023 payments were timely made and
on March 23, 2023, the Company elected to pay off the remaining balance of $63,260. The outstanding balance as of December 31, 2023 and
2022 was $0 and 78,260, respectively.
|
| X |
- References
+ Details
| Name: |
us-gaap_DebtDisclosureAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for information about short-term and long-term debt arrangements, which includes amounts of borrowings under each line of credit, note payable, commercial paper issue, bonds indenture, debenture issue, own-share lending arrangements and any other contractual agreement to repay funds, and about the underlying arrangements, rationale for a classification as long-term, including repayment terms, interest rates, collateral provided, restrictions on use of assets and activities, whether or not in compliance with debt covenants, and other matters important to users of the financial statements, such as the effects of refinancing and noncompliance with debt covenants. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1B
-Subparagraph (h)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-1B
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(c))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 470
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//470/tableOfContent
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1B
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-1B
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1B
-Subparagraph (g)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-1B
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1B
-Subparagraph (i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-1B
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1C
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-1C
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1C
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-1C
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1C
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-1C
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1E
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-1E
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1I
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-1I
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1I
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-1I
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1I
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-1I
| Name: |
us-gaap_DebtDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
CAPITAL STOCK
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Equity [Abstract] |
|
| CAPITAL STOCK |
NOTE
8 – CAPITAL STOCK
Preferred
Stock
The
Company is authorized to issue up to 10,000,000 shares of “blank check” preferred stock, $0.0001 par value. All preferred
stock were undesignated as of December 31, 2023 and 2022.
Common
Stock
The
Company is authorized to issue 200,000,000 shares of common stock, par value $0.0001 per share, of which 21,419,500 shares were issued
and outstanding at December 31, 2023 and 13,365,000 were issued and outstanding at December 31, 2022.
On
January 3, 2023, we entered into a Consulting Agreement with DojoLabs Group, Inc. (“DojoLabs”), to provide various strategic
marketing related services to the Company pursuant to a defined scope of work during the term of the agreement, which is the earlier
of a) all deliverables being received by the Company pursuant to the scope of work, or b) if terminated due to breach of the agreement
by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing to provide the
services under the agreement, the Company agreed to pay DojoLabs $100,000 in cash and issued DojoLabs 50,000 shares of restricted common
stock with registration rights and fully vest upon the completion of all work performed under the scope of work. The agreement contains
customary confidentiality and non-solicitation provisions. The shares were valued at $1.00 per share for a total of $100,000.
On
January 6, 2023, we entered into a Consulting Agreement with Bethor, Ltd. (“Bethor”), to provide strategic advisory services
to the Company during the term of the agreement, which is for 12 months unless otherwise earlier terminated due to breach of the agreement
by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing to provide the
services under the agreement, the Company issued Bethor 250,000 shares of restricted common stock with registration rights. The agreement
contains customary confidentiality and non-solicitation provisions. The shares were valued at $1.00 per share for a total of $250,000.
On
January 6, 2023, the Company established an advisory board (the “Advisory Board”) and approved and adopted a charter (the
“Advisory Board Charter”) to govern the Advisory Board. Pursuant to the Advisory Board Charter, the Advisory Board shall
be comprised of a minimum of two (2) members, all of whom shall be appointed and subject to removal by the Board of Directors at any
time. In addition to the enumerated responsibilities of the Advisory Board in the Advisory Board Charter, the primary function of the
Advisory Board is to assist the Board of Directors in its general oversight of the Company’s development of new business ventures
and strategic planning.
In
connection with the establishment of the Advisory Board, the Board of Directors appointed Dr. Brian Rudman (“Dr. Rudman”)
and Mr. Jarrett Boon (“Mr. Boon”), both of whom are independent, non-Board members and non-Company employees, to the Advisory
Board. Dr. Rudman will serve as Chairman of the Advisory Board.
In
connection with Dr. Rudman’s appointment to the Advisory Board, the Company entered into an Advisor Agreement (the “Dr. Rudman
Consulting Agreement”), dated effective January 6, 2023, with Dr. Rudman, whereby the Company agreed to issue Dr. Rudman 25,000
shares of the Company’s restricted common stock, pay Dr. Rudman $2,000 per month in cash, and reimburse Dr. Rudman for reasonable
out-of-pocket expenses, including, without limitation, travel expenses incurred by him in connection with the Company’s requests
of the performance of his duties to the Company in service on the Advisory Board. The shares were valued at $1.00 per share for a total
of $25,000.
In
connection with Mr. Boon’s appointment to the Advisory Board, the Company entered into an Advisor Agreement (the “Mr. Boon
Consulting Agreement”), dated effective January 6, 2023, with Mr. Boon, whereby the Company agreed to issue Mr. Boon 25,000 shares
of the Company’s restricted common stock and to reimburse Mr. Boon for reasonable out-of-pocket expenses, including, without limitation,
travel expenses incurred by him in connection with the Company’s requests of the performance of his duties to the Company in service
on the Advisory Board. The shares were valued at $1.00 per share for a total of $25,000.
On
January 24, 2023, we entered into Consulting Agreements with four consultants to the Company: (1) Sultan Haroon; (2) John Helfrich; (3)
Justin Baker; and (4) Maja Matthews, each of whom is also an employee of Epiq Scripts. Pursuant to the Consulting Agreements, the Consultants
agreed to provide us services related to the research, development, packaging and marketing for additional pharmaceutical and other over-the-counter
related products during the term of the agreement, which each have a term of 18 months unless otherwise earlier terminated due to breach
of the agreement by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing
to provide the services under the agreement, the Company issued an aggregate of 350,000 shares of common stock to the consultants as
follows: (1) Sultan Haroon 150,000 shares of restricted common stock; (2) John Helfrich 25,000 shares of restricted common stock; (3)
Justin Baker 25,000 shares of restricted common stock; and (4) Maja Matthews 150,000 shares of restricted common stock. The shares issued
to Haroon and Matthews vest at the rate of 50,000 shares upon entry into the agreement, 50,000 shares upon the Company’s successful
launch of a new product category, and 50,000 shares upon the Company’s successful launch of a second and additional new product
category, in each case prior to the 18-month anniversary of the applicable agreement. The shares issued to Helfrich and Baker vest at
the rate of 10,000 shares upon entry into the agreement, 7,500 shares upon the Company’s successful launch of a new product category,
and 7,500 shares upon the Company’s successful launch of a second and additional new product category, in each case prior to the
18-month anniversary of the applicable agreement. Any shares not vested by the eighteen-month anniversary of the applicable agreement
are forfeited. The agreement contains customary confidentiality and non-solicitation provisions. The shares were valued at $1.00 per
share for a total of $350,000.
On
March 22, 2023, the Company sold 1,250,000 shares of its common stock at a price of $4.00 per share to investors in connection with its
IPO for gross proceeds of $5,000,000.
On
April 24, 2023, a warrant holder exercised private placement Warrants to purchase 100,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $100,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
April 25, 2023, a warrant holder exercised private placement Warrants to purchase 100,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $100,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
April 25, 2023, a warrant holder exercised private placement Warrants to purchase 25,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $25,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
April 25, 2023, a warrant holder exercised private placement Warrants to purchase 25,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $25,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
April 25, 2023, a warrant holder exercised private placement Warrants to purchase 75,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $75,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
April 26, 2023, a warrant holder exercised private placement Warrants to purchase 100,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $100,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
May 1, 2023, a warrant holder exercised private placement Warrants to purchase 25,000 shares of common stock with an exercise price of
$1.00 per share in consideration for $25,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
and effective on May 1, 2023, the Company entered into an Employment Agreement with Mrs. Amanda Hammer (the “Employment Agreement”).
The Employment Agreement provides for Mrs. Hammer to serve as Chief Operating Officer of the Company for an initial three-year term extending
through May 1, 2026, provided that the agreement automatically renews for additional one-year terms thereafter in the event neither party
provides the other at least 60 days prior notice of their intention not to renew the terms of the agreement. The agreement provides for
Mrs. Hammer to receive an annual salary of $150,000 per year. The Employment Agreement also required the Company to grant Mrs. Hammer
a sign-on bonus of (a) 75,000 restricted shares of common stock of the Company, vested in full upon issuance, and (b) options to purchase
an additional 150,000 shares of common stock of the Company, under the Company’s 2022 Equity Incentive Plan (the “Plan”),
with an exercise price of the greater of (i) $1.10 per share; and (ii) the closing sales price of the Company’s common stock on
the Nasdaq Capital Market on the date the Employment Agreement and the grant is approved by the Board (which date was May 1, 2023), and
which exercise price was $1.00 per share, with options to purchase 50,000 shares vesting every twelve months that the Employment Agreement
is in effect, subject to the terms of the Plan. The options are exercisable for a period of ten years and are documented by a separate
option agreement entered into by the Company and Mrs. Hammer.
On
May 1, 2023, we entered into a Software Development Agreement with Redlime Solutions, Inc. (“Redlime”) to provide software
development services during the term of the agreement, which is for twelve months. In consideration for agreeing to provide the services
under the agreement, the Company agreed to pay Redlime $300,000 in cash and issue Redlime 180,000 shares of restricted common stock.
The shares were valued at $1.00 per share for a total of $180,000.
On
May 25, 2023, the Board of Directors appointed Mr. Aaron Andrew (“Mr. Andrew”), an independent, non-Board member and non-Company
employee, to the Advisory Board. In connection with Mr. Andrew’s appointment to the Advisory Board, the Company entered into an
Advisor Agreement (the “Mr. Andrew Consulting Agreement”), dated effective May 25, 2023, with Mr. Andrew, whereby the Company
agreed to issue Mr. Andrew 50,000 shares of the Company’s restricted common stock under the 2022 Plan and to reimburse Mr. Andrew
for reasonable out-of-pocket expenses, including, without limitation, travel expenses incurred by him in connection with the Company’s
requests of the performance of his duties to the Company in service on the Advisory Board. The shares were valued at $1.10 per share
for a total of $55,000.
On
June 1, 2023, we entered into a Consulting Agreement with Major Dodge (“Major”), to provide acting and production related
services to the Company during the term of the agreement, which is for 12 months unless otherwise earlier terminated due to breach of
the agreement by either party and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing
to provide the services under the agreement, the Company issued Major 20,000 shares of restricted common stock under the 2022 Plan. The
agreement contains customary confidentiality and non-solicitation provisions. The shares were valued at $1.10 per share for a total of
$22,000.
On
June 1, 2023, we entered into a Production and Broadcasting Agreement with New To The Street Group, LLC (“New To The Street”),
to provide production, broadcasting and other marketing related services to the Company during the term of the agreement, which is for
3 months unless otherwise earlier terminated. In consideration for agreeing to provide the services under the agreement, the Company
issued New To The Street 50,000 shares of restricted common stock and agreed to pay New To The Street a monthly cash payment of $5,000.
The shares were valued at $1.10 per share for a total of $55,000.
On June 5, 2023, a warrant holder exercised private placement Warrants
to purchase 25,000 shares of common stock with an exercise price of $1.00 per share in consideration for $25,000 in cash. The shares of
common stock issuable upon exercise of the warrants were registered under the Securities Act.
On
June 6, 2023, a warrant holder exercised private placement Warrants to purchase 150,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $150,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
June 7, 2023, a warrant holder exercised private placement Warrants to purchase 75,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $75,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
June 8, 2023, a warrant holder exercised private placement Warrants to purchase 24,500 shares of common stock with an exercise price
of $1.00 per share in consideration for $24,500 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
June 21, 2023, a warrant holder exercised private placement Warrants to purchase 100,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $100,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
June 22, 2023, a warrant holder exercised private placement Warrants to purchase 100,000
shares of common stock with an exercise price
of $1.00
per share in consideration for $100,000
in cash. The shares of common stock issuable
upon exercise of the warrants were registered under the Securities Act.
On
June 27, 2023, a warrant holder exercised private placement Warrants to purchase 100,000 shares of common stock with an exercise price
of $1.00 per share in consideration for $100,000 in cash. The shares of common stock issuable upon exercise of the warrants were registered
under the Securities Act.
On
September 1, 2023, we entered into a service agreement with Greentree Financial Group, Inc. (“Greentree” and the “Service
Agreement”). Pursuant to the Service Agreement, Greentree agreed to perform the following services: (a) bookkeeping services for
the Company for the period from October 1, 2023 through September 30, 2024; (b) advice and assistance to the Company in connection with
the conversion of its financial reporting systems, including its projected financial statements, to a format that is consistent with
US GAAP; (c) assistance to the Company with compliance filings for the quarters ended September 30, 2023, March 31, 2024, June 30, 2024
and the year ended December 31, 2023, including the structure and entries as well as assistance with US GAAP footnotes; (d) reviewing,
and providing advice to the Company on, all documents and accounting systems relating to its finances and transactions, with the purpose
of bringing such documents and systems into compliance with US GAAP or disclosures required by the SEC; and (e) providing necessary consulting
services and support as a liaison for the Company to third party service providers, including coordination amongst the Company and its
attorneys, CPAs and transfer agent. Since February 2015, Mr. Eugene (Gene) M. Johnston, our Chief Financial Officer (who was appointed
October 1, 2022) has served as an Audit Manager for Greentree.
The
Company agreed to issue Greentree 75,000 shares of the Company’s restricted common stock upon the parties’ entry into the
agreement, and to pay Greentree $40,000 in cash, payable as follows: (a) $20,000 on or before September 30, 2023; (b) $20,000 on or before
March 31, 2024. We also agreed to reimburse Greentree for its reasonable out-of-pocket expenses incurred in connection with Greentree’s
activities under the agreement, including the reasonable fees and travel expenses for the meetings on behalf of the Company. The Service
Agreement includes customary indemnification obligations requiring the Company to indemnify Greentree and its affiliates with regard
to certain matters. The shares were valued at $1.13 per share for a total of $84,750.
On
October 1, 2023, the Company executed a Summary of Terms and Conditions (“Consulting Agreement”) with Gene Johnston (“Johnston”)
continuing his appointment as the Company’s Chief Financial Officer on a full-time basis for a term of 12 months. Pursuant to the
Consulting Agreement, the Company issued Johnston 50,000 shares of the Company’s common stock and $2,000 per month. The Consulting
Shares shall be issued under, and subject to the terms of, the Company’s 2022 Equity Incentive Plan.
On
October 10, 2023, we entered into a Consulting Agreement with Luca Consulting, LLC (“Luca”), to provide certain management
and consulting services to the Company during the term of the agreement, which is for three months unless otherwise earlier terminated
due to breach of the agreement by either party. In consideration for agreeing to provide the services under the agreement, the Company
issued 200,000 shares of the Company’s restricted common stock upon the parties’ entry into the agreement and to pay Luca
$15,000 in cash, payable as follows: (a) $5,000 on the signing of the agreement; (b) $5,000 on the tenth of each month throughout the
remainder of the agreement. The Service Agreement includes customary indemnification obligations requiring the Company to indemnify Luca
and its affiliates with regard to certain matters. The shares were valued at $0.63 per share for a total of $126,000.
On
November 1, 2023, we entered into an Influencer Agreement with Jason Szkup (“Scoop”) to promote its products or services
through social media platforms and other online channels, In consideration for agreeing to provide the services under the agreement,
the Company agreed to pay Scoop $10,000 in cash and issue 30,000 shares. The shares were valued at $0.58 per share for a total of $17,400.
The Shares shall be issued under, and subject to the terms of, the Company’s 2022 Equity Incentive Plan.
On
November 1, 2023, the Board of Directors appointed Dr. Douglas Christianson, ND (“Dr. Christianson”), an independent, non-Board
member and non-Company employee, to the Advisory Board. In connection with Dr. Christianson’s appointment to the Advisory Board,
the Company entered into an Advisor Agreement (the “Dr. Christianson Consulting Agreement”), with Dr. Christianson, whereby
the Company agreed to issue Dr. Christianson 50,000 shares. The Shares shall be issued under, and subject to the terms of, the Company’s
2022 Equity Incentive Plan. The Company will reimburse Dr. Christianson for reasonable out-of-pocket expenses, including, without limitation,
travel expenses incurred by him in connection with the Company’s requests of the performance of his duties to the Company in service
on the Advisory Board. The shares were valued at $0.58 per share for a total of $29,000.
On
November 15, 2023, we renewed a Consulting Agreement with PHX Global, LLC (“PHX”), which is owned by Peter “Casey”
Jensen, who is a member of the Board of Directors of American International. Pursuant to the Consulting Agreement, PHX agreed to provide
consulting and general business advisory services as reasonably requested by the Company during the term of the agreement, which was
for 12 months, unless otherwise earlier terminated due to breach of the agreement by either party, and the failure to cure such breach
30 days after written notice thereof. In consideration for agreeing to provide the services under the agreement, the Company issued PHX
200,000 shares of restricted common stock. The agreement contains customary confidentiality and non-solicitation provisions. The shares
were valued at $0.47 per share for a total of $94,000. PHX is a related party.
On
December 11, 2023, the Company entered into a Marketing Agreement with Marius Pharmaceuticals (“Marius”) to market and sell
KYZATREX®, an innovative FDA-approved oral Testosterone Replacement Therapy (TRT) product, under the program, ‘PRIME’
by MangoRx. During the Term, Marius grants to MangoRx a non-exclusive, non-transferable, royalty-free license to use the Marius Marks
in the United States (the “Territory”) for the sole purpose of the Permitted Purpose. The term of the initial agreement is
for two years, automatically renewable for successive one year terms, subject to certain performance targets as agreed upon each year.
As consideration for the license granted herein, MangoRx shall issue to Marius one hundred thousand (100,000) shares of the Company’s
common stock (the “Marius Shares”). The Marius Shares shall be issued to Marius upon signing of this Agreement and shall
be deemed fully earned upon signing this Agreement. The shares were valued at $0.58 per share for a total of $58,000.
On
December 19, 2023 the Company sold 4,000,000 shares of its common stock at a price of $0.30 per share to investors in connection with
a follow on offering for gross proceeds of $1,200,000.
Options:
During
the year ended December 31, 2022, the Company granted a total of 1,250,000
options to purchase shares of common stock of the Company, under the 2022 Plan, of which 750,000
were granted to Jacob Cohen, the Company’s CEO, and 500,000
were granted to Jonathan Arango, the Company’s then President and then COO, related to their respective employment agreement.
The options have an exercise price of $1.10
per share, an original life of five years and vest at the annual renewal of their employment over three
years.
On
May 1, 2023, the Company granted 150,000 options to purchase shares of common stock of the Company, under the 2022 Plan to Amanda Hammer,
the Company’s COO, related to her employment agreement. The options have an exercise price of $1.10 per share, an original life
of five years and vest at the annual renewal of their employment over three years.
On
December 28, 2023, the Company granted 1,250,000 options to purchase shares of common stock of the Company, under the 2022 Plan to Jacob
Cohen, the Company’s CEO, related to his employment agreement. The options have an exercise price of $0.32 per share, an original
life of five years and vested at the time of grant.
As
of December 31, 2023 and 2022, $624,463
and $82,267, respectively,
has been recorded and included as stock-based compensation expense on the statement of operations for the year-ended December 31,
2023 and within general and administrative expense for year-ended December 31, 2022. Mr. Cohen, Mr. Arango (former President and
Director) and Ms. Hammer are related parties.
The
following table summarizes common stock options activity: The following table summarizes common stock options activity:
SCHEDULE
OF STOCK OPTION ACTIVITY
| | |
Options | | |
Weighted Average Exercise Price | |
| December 31, 2021 | |
| - | | |
$ | - | |
| Granted | |
| 1,250,000 | | |
| 1.10 | |
| Exercised | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Outstanding, December 31, 2022 | |
| 1,250,000 | | |
$ | 1.10 | |
| Exercisable, December 31, 2022 | |
| 133,333 | | |
$ | 1.10 | |
| Outstanding, December 31, 2023 | |
| 1,250,000 | | |
$ | 1.10 | |
| | |
| | | |
| | |
| Granted | |
| 1,400,000 | | |
$ | 0.40 | |
| Exercised | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Outstanding, December 31, 2023 | |
| 2,650,000 | | |
$ | 0.73 | |
| Exercisable, December 31, 2023 | |
| 1,812,500 | | |
$ | 0.73 | |
The
weighted average exercise prices, remaining lives for options granted, and exercisable as of December 31, 2023 were as follows:
| | | |
Outstanding Options | | |
| | |
Exercisable Options | |
Options Exercise Price Per Share | | |
Shares | | |
Life (Years) | | |
Weighted Average Exercise Price | | |
Shares | | |
Weighted Average Exercise Price | |
| $ | 1.10 | | |
| 1,400,000 | | |
| 4.28 | | |
$ | 1.10 | | |
| 562,500 | | |
$ | 1.10 | |
| $ | 0.32 | | |
| 1,250,000 | | |
| 5.00 | | |
$ | 0.32 | | |
| 1,250,000 | | |
$ | 0.32 | |
As
of December 31, 2023, the fair value of options outstanding was $573,202. The aggregate initial fair value of the options measured on
the grant date of August 31, 2022, May 1, 2023 and December 28, 2023 was calculated using the Black-Scholes option pricing model based
on the following assumption:
SCHEDULE
OF FAIR VALUE ASSUMPTIONS
| Fair Value of Common Stock on measurement date | |
$ | 1.00 - 0.32 | |
| Risk free interest rate | |
| 3.83% - 3.30 | % |
| Volatility | |
| 232.05% 92.54 | % |
| Dividend Yield | |
| 0 | % |
| Expected Term | |
| 6.0 - 3.5 | |
| |
(1) |
The risk-free interest
rate was determined by management using the market yield on U.S. Treasury securities with comparable terms as of the measurement
date. |
| |
(2) |
The trading volatility
was determined by calculating the volatility of the Company’s peer group. |
| |
(3) |
The Company does not expect
to pay a dividend in the foreseeable future |
| |
(4) |
The Company, in accordance
with staff accounting bulletin (“SAB”)14-D.2, used the simplified method (plain vanilla) to determine the overall expected
term |
Warrants:
As
additional consideration in connection with the IPO, upon the closing of the IPO, we granted Boustead Securities, LLC, the representative
of the underwriters named in the Underwriting Agreement for the IPO, warrants to purchase 87,500 shares of common stock with an exercise
price of $5.00 per share, which are exercisable six months after the effective date of the registration statement filed in connection
with the IPO (March 20, 2023) and expire five years after such effectiveness date. The fair value of the warrants on the grant date was
$31,995.
As
additional consideration in connection with the follow on offering, upon the closing of the follow on offering, we granted Boustead Securities,
LLC, the representative of the underwriters named in the Underwriting Agreement for the secondary offering, warrants to purchase 280,000
shares of common stock with an exercise price of $0.38 per share, which are exercisable six months after the effective date of the registration
statement filed in connection with the follow on offering (December 19, 2023) and expire five years after such effectiveness date. The
fair value of the warrants on the grant date was $271,216.
As
of December 31, 2023 and 2022, the fair value of warrants outstanding to investors was $852,480 and $581,264, respectively.
Because the warrants vested immediately, the fair value was assessed on the grant date.
The
following table summarizes common stock warrants activity:
SCHEDULE
OF WARRANT ACTIVITY
| | |
Warrants | | |
Weighted Average Exercise Price | |
| Outstanding, December 31, 2021 | |
| - | | |
$ | - | |
| Granted | |
| 2,210,070 | | |
| 1.00 | |
| Exercised | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Cancelled | |
| (210,070 | ) | |
| 1.00 | |
| Outstanding, December 31, 2022 | |
| 2,000,000 | | |
| 1.00 | |
| Exercisable, December 31, 2022 | |
| 2,000,000 | | |
$ | 1.00 | |
| | |
| | | |
| | |
| Granted | |
| 367,500 | | |
| 1.22 | |
| Exercised | |
| (1,024,500 | ) | |
| 1.00 | |
| Expired | |
| - | | |
| - | |
| Cancelled | |
| - | | |
| - | |
| Outstanding, December 31, 2023 | |
| 1,343,000 | | |
| 1.43 | |
| Exercisable, December 31, 2023 | |
| 1,343,000 | | |
$ | 1.43 | |
The
weighted average exercise prices, remaining lives for warrants granted, and exercisable as of December 31, 2023, were as follows:
| | | |
Outstanding and Vested Warrants | |
Weighted Average Warrant
Exercise Price Per Share | | |
Shares | | |
Life (Years) | |
| $ | 1.43 | | |
| 1,343,000 | | |
| 2.07 | |
As
of December 31, 2023, warrants to purchase 1,063,000 shares of common stock are outstanding and vested, and the vested stock warrants
have a weighted average remaining life of 3.83 years.
SCHEDULE
OF FAIR VALUE ASSUMPTIONS
| Fair Value of Common Stock on measurement date | |
$ | 0.37 - $0.97 | |
| Risk-free interest rate | |
| From 2.95% to 4.00 | % |
| Volatility | |
| From 81.92% to 169.22 | % |
| Dividend Yield | |
| 0 | % |
| Expected Term | |
| 5 years | |
| |
(1) |
The risk-free interest
rate was determined by management using the market yield on U.S. Treasury securities with comparable terms as of the measurement
date. |
| |
(2) |
The trading volatility
was determined by calculating the volatility of the Company’s peer group. |
| |
(3) |
The Company does not expect
to pay a dividend in the foreseeable future. |
|
| X |
- References
+ Details
| Name: |
us-gaap_EquityAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for equity. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 13
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-13
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 13
-Subparagraph (h)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-13
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 14
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-14
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 235
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481062/946-235-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 235
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481062/946-235-50-2
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 505
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481004/946-505-50-6
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480237/815-40-50-6
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(e)(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 10: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//505/tableOfContent
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 13
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-13
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 13
-Subparagraph (g)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-13
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 13
-Subparagraph (i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-13
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 14
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-14
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 14
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-14
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 16
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-16
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 18
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-18
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 18
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-18
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 18
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-18
| Name: |
us-gaap_StockholdersEquityNoteDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
GOING CONCERN
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Organization, Consolidation and Presentation of Financial Statements [Abstract] |
|
| GOING CONCERN |
NOTE
9 – GOING CONCERN
These
financial statements have been prepared assuming that the Company will continue as a going concern, which contemplates the realization
of assets and the discharge of liabilities in the normal course of business for the foreseeable future. As reflected in the accompanying
financials, the Company had a net loss of $9,212,417 for the year ended December 31, 2023 and an accumulated deficit of $11,228,173 as
of December 31, 2023. The Company will need to raise additional capital to successfully execute its business plan of which there can
be no assurance. The sources of this capital are expected to be the sale of equity and debt, which may not be available on favorable
terms, if at all, and may, if sold, cause significant dilution to existing shareholders. If we are unable to access additional capital
moving forward, it may hurt our ability to grow and to generate future revenues, our financial position, and liquidity, or force us to
abandon our business plan. These factors raise substantial doubt about the ability of the Company to continue as a going concern. Unless
management is able to obtain additional financing, it is unlikely that the Company will be able to meet its funding requirements during
the next 12 months. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
|
| X |
- References
+ Details
| Name: |
us-gaap_OrganizationConsolidationAndPresentationOfFinancialStatementsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure when substantial doubt is raised about the ability to continue as a going concern. Includes, but is not limited to, principal conditions or events that raised substantial doubt about the ability to continue as a going concern, management's evaluation of the significance of those conditions or events in relation to the ability to meet its obligations, and management's plans that alleviated or are intended to mitigate the conditions or events that raise substantial doubt about the ability to continue as a going concern. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 205
-SubTopic 40
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//205-40/tableOfContent
| Name: |
us-gaap_SubstantialDoubtAboutGoingConcernTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
COMMITMENTS AND CONTINGENCIES
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Commitments and Contingencies Disclosure [Abstract] |
|
| COMMITMENTS AND CONTINGENCIES |
NOTE
10 – COMMITMENTS AND CONTINGENCIES
In
the ordinary course of business, the Company may become a party to lawsuits involving various matters. The impact and outcome of litigation,
if any, is subject to inherent uncertainties, and an adverse result in these or other matters may arise from time to time that may harm
its business. The Company is not currently subject to any such litigation.
Operating
Leases
The
Company has a lease for an office in Dallas, Texas classified as operating leases under ASC 842.
On
September 28, 2022, and with an effective date of October 1, 2022, the Company entered into a Lease Agreement with Rox Trep Tollway,
L.P. (the “Landlord”) to lease and occupy approximately 2,201 square feet of office space located at 15110 Dallas Parkway,
Suite 600, Dallas, Texas 75248 to serve as the Company’s main headquarters (the “Lease Agreement”). The Lease
Agreement has a term of thirty-eight (38) months and has a monthly base rent of $5,777.63, or $31.50 per square foot, the from months
3-18 and increases at the rate of $1 per square foot per annum until the end of the lease term (the “Base Rent”).
In addition to the Base Rent, the Company is required to reimburse the landlord for its pro-rata share of all real estate taxes and assessments,
hazard and liability insurance and common area maintenance costs for the building at the rate of 2.45% (the “Proportionate Rent”).
Upon the execution of the Lease Agreement, the Company agreed to prepay the first full month’s Base Rent along with a security
deposit equal to $16,942.
The
Company utilizes the incremental borrowing rate in determining the present value of lease payments unless the implicit rate is readily
determinable. The Company used an estimated incremental borrowing rate of 8% to estimate the present value of the right-of-use liability.
The
Company has right-of-use assets of $119,262
and operating lease liabilities of $128,679
as of December 31, 2023. Operating lease expense
for the year ended December 31, 2023 was $65,274.
The Company has recorded $0 in
impairment charges related to right-of-use assets during the year ended December 31, 2023.
SCHEDULE
OF MATURITY OF LEASE LIABILITIES
| Maturity of Lease Liabilities at December 31, 2023 | |
Amount | |
| 2024 | |
$ | 71,716 | |
| 2025 | |
| 67,589 | |
| Later years | |
| - | |
| Total lease payments | |
| 139,305 | |
| Less: Imputed interest | |
| (10,626 | ) |
| Present value of lease liabilities | |
$ | 128,679 | |
|
| X |
- References
+ Details
| Name: |
us-gaap_CommitmentsAndContingenciesDisclosureAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for commitments and contingencies. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 440
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482648/440-10-50-4
Reference 2: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 450
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//450/tableOfContent
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 954
-SubTopic 440
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480327/954-440-50-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 440
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482648/440-10-50-4
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 440
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//440/tableOfContent
| Name: |
us-gaap_CommitmentsAndContingenciesDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
INCOME TAXES
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Income Tax Disclosure [Abstract] |
|
| INCOME TAXES |
NOTE
11 - INCOME TAXES
On
December 22, 2017, the U.S. enacted the Tax Cuts and Jobs Act (the “Act”), which significantly changed U.S. tax law. The
Act lowered the Company’s U.S. statutory federal income tax rate from 35% to 21% effective January 1, 2018, while also imposing
a deemed repatriation tax on previously deferred foreign income.
The
Act also created a new minimum tax on certain future foreign earnings. The impact of the Act increased the Company’s deferred tax
asset related to the Company’s net operating loss by approximately $9,212,417 and increased the Company’s valuation allowance
by approximately $9,212,417 resulting in no impact to the Company’s financials.
We
record tax positions as liabilities in accordance with ASC 740 and adjust these liabilities when our judgment changes as a result of
the evaluation of new information not previously available. Because of the complexity of some of these uncertainties, the ultimate resolution
may result in a payment that is materially different from our current estimate of the recognized tax benefit liabilities. These differences
will be reflected as increases or decreases to income tax expense in the period in which new information is available. As of December
31, 2023, and 2022 we have not recorded any uncertain tax positions in our financial statements.
The
effective US Federal Income Corporate Tax Rates for 2023 and 2022 are 21% and 21%, respectively.
The
Company has net operating loss carryforwards of approximately $11,228,173 at December 31, 2023 that do not expire. However, utilization
of these losses may be limited pursuant to Section 382 of the Internal Revenue Code due to subsequent stock issuances.
The
Company has a deferred tax asset as shown in the following:
SCHEDULE
OF DEFERRED TAX ASSET
| | |
Year Ending December 31, 2023 | | |
Year Ending December 31, 2022 | |
| Deferred Tax Asset | |
| 11,228,173 | | |
| 2,015,756 | |
| Valuation Allowance | |
| (11,228,173 | ) | |
| (2,015,756 | ) |
| Net Deferred Tax Asset | |
$ | — | | |
$ | — | |
|
| X |
- DefinitionThe entire disclosure for income taxes. Disclosures may include net deferred tax liability or asset recognized in an enterprise's statement of financial position, net change during the year in the total valuation allowance, approximate tax effect of each type of temporary difference and carryforward that gives rise to a significant portion of deferred tax liabilities and deferred tax assets, utilization of a tax carryback, and tax uncertainties information. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 13
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480990/946-20-50-13
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(h)(2))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//740/tableOfContent
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 14
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-14
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 21
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-21
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 270
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482526/740-270-50-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 17
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-17
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SAB TOPIC 6.I.5.Q1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479360/740-10-S99-1
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SAB Topic 11.C)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479360/740-10-S99-2
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482603/740-30-50-2
| Name: |
us-gaap_IncomeTaxDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
SUBSEQUENT EVENTS
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Subsequent Events [Abstract] |
|
| SUBSEQUENT EVENTS |
NOTE
12 – SUBSEQUENT EVENTS
On
January 2, 2024, we entered into a Consulting Agreement with G&P General Consulting (“G&P”), Pursuant to the Consulting
Agreement, G&P agreed to provide consulting and general business advisory services as it relates to the expansion of the Company’s
products into additional international territory’s, including, but not limited to, the United Arab Emirates (UAE), China, Japan,
Korea, and in certain regions of Asia and additional services as reasonably requested by the Company during the Term of this Agreement
as reasonably requested by the Company during the term of the agreement, which was for 12 months, unless otherwise earlier terminated
due to breach of the agreement by either party, and the failure to cure such breach 30 days after written notice thereof. In consideration
for agreeing to provide the services under the agreement, the Company issued G&P 250,000 shares of restricted common stock. G&P
will receive an additional 500,000 shares in 90 days, if the agreement is still in place. The Consulting Shares shall be issued under,
and subject to the terms of, the Company’s 2022 Equity Incentive Plan. The agreement contains customary confidentiality and non-solicitation
provisions. The shares were valued at $0.28 per share for a total of $70,000.
On
January 10, 2024, we renewed a Consulting Agreement with Luca Consulting, LLC (“Luca”), to provide certain management and
consulting services to the Company during the term of the agreement, which is for three months unless otherwise earlier terminated due
to breach of the agreement by either party. In consideration for agreeing to provide the services under the agreement, the Company issued
200,000 shares of the Company’s restricted common stock upon the parties’ entry into the agreement and to pay Luca $15,000
in cash, payable as follows: (a) $5,000 on the signing of the agreement; (b) $5,000 on the tenth of each month throughout the remainder
of the agreement. The Service Agreement includes customary indemnification obligations requiring the Company to indemnify Luca and its
affiliates with regard to certain matters. The shares were valued at $0.28 per share for a total of $56,000.
On
January 11, 2024, we entered into a Consulting Agreement with First Level Capital (“First Level”), to provide certain management
and consulting services to the Company during the term of the agreement, which is for six months unless otherwise earlier terminated
due to breach of the agreement by either party. In consideration for agreeing to provide the services under the agreement, the Company
issued an initial 250,000 shares of the Company’s restricted common stock upon the parties’ entry into the agreement, an
additional 250,000 shares of the Company’s restricted common stock before the end of the term of the agreement and to pay First
Level $60,000 in cash, payable as follows: (a) $60,000 on the signing of the agreement; (b) $60,000 on the approval by the Company. The
Service Agreement includes customary indemnification obligations requiring the Company to indemnify First Level and its affiliates with
regard to certain matters. The initial shares were valued at $0.28 per share for a total of $70,000.
On
January 18, 2024, the Underwriters in the follow-on offering notified the Company that they were exercising their over-allotment option
in full to purchase an additional 600,000 shares of common stock, which sale closed on January 22, 2024. The net proceeds to the Company
from the sale of the 600,000 shares of common stock, after deducting underwriting discounts and expenses, was approximately $160,000.
Inclusive of the full exercise of the over-allotment option, a total of 4,600,000 shares of common stock were issued and sold in the
follow-on offering.
On
January 22, 2024, pursuant to the Underwriting Agreement, the Company also issued a common stock purchase warrant to the Representative
for the purchase of 42,000 shares of its common stock at an exercise price of $0.375, subject to adjustments (the “Warrant”).
The Warrant is exercisable at any time and from time to time, in whole or in part, until December 14, 2028, and may be exercised on a
cashless basis. The Warrant also includes customary anti-dilution provisions and immediate piggyback registration rights with respect
to the registration of the shares underlying the Warrant. The Warrant and the shares of common stock underlying the Warrant were registered
as a part of the follow-on registration statement.
On March 21, 2024, we entered into Amendment to the
of January 10, 2024 consulting agreement with Luca extending the agreement for an additional 6 months (the “Luca Amendment”).
In consideration for entering into the Luca Amendment, the Company issued 500,000 shares of the Company’s restricted common stock
upon the parties’ entry into the Luca Amendment and agreed to continue to pay Luca $5,000 in in cash on the tenth of each month
throughout the remainder of the agreement. The shares were valued at $0.1975 per share for a total of $98,750.
On March 21, 2024, we entered into a Consulting Agreement
with Zvonimir Moric (the “Zee”). Pursuant to the consulting agreement, Zee agreed to provide consulting and general business
advisory services as it relates to making introductions to strategic partners to expand the sales of the Company’s products and
additional services as reasonably requested by the Company during the Term of this Agreement as reasonably requested by the Company during
the term of the agreement, which is for 12 months, unless otherwise earlier terminated due to breach of the agreement by either party,
and the failure to cure such breach 30 days after written notice thereof. In consideration for agreeing to provide the services under
the agreement, the Company issued Zee 150,000 shares of restricted common stock. The Consulting Shares were issued under, and subject
to the terms of, the Company’s 2022 Equity Incentive Plan. The agreement contains customary confidentiality and non-solicitation
provisions. The shares were valued at $0.1975 per share for a total of $29,625.
On
March 1, 2024, the Company borrowed $37,500 from Ronin Equity Partners, which is owned and controlled by Jacob D. Cohen, the Company’s
Chief Executive Officer and Chairman. The amount borrowed is payable on demand and does not accrue interest.
On
March 18, 2024, the Company borrowed $50,000 from Cohen Enterprises, Inc. , which is owned and controlled by Jacob D. Cohen, the Company’s
Chief Executive Officer and Chairman. The amount borrowed is payable on demand and does not accrue interest.
On
March 25, 2024, at a Special Meeting of the stockholders of the Company, the
stockholders of the Company approved a First Amendment to the Mangoceuticals, Inc. 2022 Equity Incentive Plan (“First Amendment”
and the Mangoceuticals, Inc. 2022 Equity Incentive Plan, as amended by the First Amendment, the “2022 Plan”). The
First Amendment was originally approved by the Board of Directors of the Company on February 26, 2024, subject to stockholder approval
and the First Amendment became effective at the time of stockholder approval. The First Amendment increased the number of shares of common
stock available for awards under the Incentive Plan, such that currently, subject to adjustment in connection with the payment of a stock
dividend, a stock split or subdivision or combination of the shares of common stock, or a reorganization or reclassification of the Company’s
common stock, the aggregate number of shares of common stock which may be issued pursuant to awards under the 2022 Plan is currently
the sum of (i) 10,000,000, and (ii) an automatic increase on April 1st of each year for a period of nine years commencing on April 1,
2024 and ending on (and including) April 1, 2032, in an amount equal to the lesser of (x) ten percent (10%) of the total shares of common
stock of the Company outstanding on the last day of the immediately preceding fiscal year; and (y) 2,000,000 shares of common stock;
provided, however, that the Board may act prior to April 1st of a given year to provide that the increase for such year will be a lesser
number of shares of common stock. This is also known as an “evergreen” provision. Notwithstanding the foregoing, no more
than a total of 26,000,000 shares of common stock (or awards) may be issued or granted under the 2022 Plan in aggregate, and no more
than 26,000,000 shares of common stock may be issued pursuant to the exercise of Incentive Stock Options.
Also
at the Special Meeting, the stockholders approved an amendment to the Company’s Second Amended and Restated Certificate of Incorporation,
as amended, to effect a reverse stock split of the Company’s issued and outstanding shares of our common stock, par value $0.0001
per share, by a ratio of between one-for-two to one-for-fifty inclusive, with the exact ratio to be set at a whole number to be determined
by the Company’s Board of Directors or a duly authorized committee thereof in its discretion, at any time after approval of the
amendment and prior to March 25, 2025. No formal determination has been made by the Board of Directors of the Company regarding the reverse
stock split ratio, whether or not to move forward with a reverse stock split, or the timing thereof.
|
| X |
- References
+ Details
| Name: |
us-gaap_SubsequentEventsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for significant events or transactions that occurred after the balance sheet date through the date the financial statements were issued or the date the financial statements were available to be issued. Examples include: the sale of a capital stock issue, purchase of a business, settlement of litigation, catastrophic loss, significant foreign exchange rate changes, loans to insiders or affiliates, and transactions not in the ordinary course of business. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 855
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//855/tableOfContent
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 855
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483399/855-10-50-2
| Name: |
us-gaap_SubsequentEventsTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Policies)
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Accounting Policies [Abstract] |
|
| Basis of Presentation |
Basis
of Presentation – The financial statements present the financial position, results of operations and cash flows of the Company
in accordance with accounting principles generally accepted in the United States of America (“US GAAP”). All dollar amounts
are rounded to the nearest thousand dollars.
|
| Cash Equivalents |
Cash
Equivalents
Highly
liquid investments with original maturities of three months or less are considered cash equivalents. The Company maintains the majority
of its cash accounts at a commercial bank. The Federal Deposit Insurance Corporation (“FDIC”) insures the total cash balance
up to $250,000 per commercial bank. From time to time, cash in deposit accounts may exceed the FDIC limits, the excess would be at risk
of loss for purposes of the statement of cash flows. There are no cash equivalents at December 31, 2023 and 2022
|
| Income Taxes |
Income
Taxes
The
Company is a taxable entity and recognizes deferred tax assets and liabilities for the future tax consequences attributable to differences
between the financial statement carrying amounts of existing assets and liabilities and their respective tax basis. Income taxes are
provided in accordance with Financial Accounting Standards Board (“FASB”) Accounting Standards Codification (“ASC”)
740, Income Taxes. Deferred tax assets and liabilities are measured using enacted tax rates expected to be in effect when the
temporary differences reverse. The effect on the deferred tax assets and liabilities of a change in tax rates is recognized in income
in the year that includes the enactment date of the rate change. A valuation allowance is used to reduce deferred tax assets to the amount
that is more likely than not to be realized.
|
| Net Loss Per Common Share |
Net
Loss Per Common Share
We
compute net loss per share in accordance with ASC 260, Earning per Share. ASC 260 requires presentation of both basic and
diluted earnings per share (“EPS”) on the face of the statement of operations. Basic EPS is computed by dividing net loss available
to common shareholders (numerator) by the weighted average number of shares outstanding (denominator) during the period. Diluted EPS
gives effect to all dilutive potential common shares outstanding during the period using the treasury stock method and convertible
preferred stock using the if-converted method. In computing Diluted EPS, the average stock price for the period is used in
determining the number of shares assumed to be purchased from the exercise of stock options or warrants. Diluted EPS excludes all
dilutive potential shares if their effect is anti-dilutive. There were 2,650,000
options, 1,343,000
warrants and no derivative securities outstanding as of December 31, 2023. There were 1,250,000
options, 2,000,000
warrants and no derivative securities outstanding as of December 31, 2022.
|
| Use of Estimates and Assumptions |
Use
of Estimates and Assumptions
The
preparation of financial statements in accordance with US GAAP requires the Company’s management to make estimates and assumptions
that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial
statements and the reported amounts of expenses during the reporting period. Actual results can, and in many cases will, differ from
those estimates.
|
| Fair Value of Financial Instruments |
Fair
Value of Financial Instruments
The
Company measures its financial and non-financial assets and liabilities, as well as makes related disclosures, in accordance with FASB
ASC 820, Fair Value Measurement (“ASC 820”), which provides guidance with respect to valuation techniques to be utilized
in the determination of fair value of assets and liabilities. Approaches include, (i) the market approach (comparable market prices),
(ii) the income approach (present value of future income or cash flow), and (iii) the cost approach (cost to replace the service capacity
of an asset or replacement cost). ASC 820 utilizes a fair value hierarchy that prioritizes the inputs to valuation techniques used to
measure fair value into three broad levels. The following is a brief description of those three levels:
Level
1: Observable inputs such as quoted prices (unadjusted) in active markets for identical assets or liabilities.
Level
2: Inputs other than quoted prices that are observable, either directly or indirectly. These include quoted prices for similar assets
or liabilities in active markets and quoted prices for identical or similar assets or liabilities in markets that are not active.
Level
3: Unobservable inputs in which little or no market data exists, therefore requiring an entity to develop its own assumptions, such as
valuations derived from valuation techniques in which one more significant inputs or significant value drivers are unobservable.
The
following tables summarize our financial instruments measured at fair value as of December 31, 2023 and 2022.
SCHEDULE
OF FINANCIAL INSTRUMENTS MEASUREMENT AT FAIR VALUE
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| | |
Fair Value Measurements at December 31, 2023 | |
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| Assets | |
| | | |
| | | |
| | |
| Cash | |
$ | 739,006 | | |
$ | - | | |
$ | - | |
| Total assets | |
| 739,006 | | |
| - | | |
| - | |
| Liabilities | |
| | | |
| | | |
| | |
| Total liabilities | |
| - | | |
| - | | |
| - | |
| Fair value, net asset
(liability) | |
$ | 739,006 | | |
$ | - | | |
$ | - | |
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| | |
Fair Value Measurements at December 31, 2022 | |
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| Assets | |
| | | |
| | | |
| | |
| Cash | |
$ | 682,860 | | |
$ | - | | |
$ | - | |
| Total assets | |
| 682,860 | | |
| - | | |
| - | |
| Liabilities | |
| | | |
| | | |
| | |
| Total liabilities | |
| - | | |
| - | | |
| - | |
| Fair value, net asset
(liability) | |
$ | 682,860 | | |
$ | - | | |
$ | - | |
|
| Property and Equipment |
Property
and Equipment
Property
and equipment are stated at cost. When retired or otherwise disposed, the related carrying value and accumulated depreciation are removed
from the respective accounts and the net difference less any amount realized from the disposition is reflected in earnings. For financial
statement purposes, property and equipment are recorded at cost and depreciated using the straight-line method over their estimated useful
lives of three (3) to five (5) years.
|
| Concentration and Risks |
Concentration
and Risks
The
Company’s operations are subject to risks including financial, operational, regulatory and other risks including the potential
risk of business failure. For the years ended December 31, 2023 and 2022, the Company had no significant revenue from continuing
operations which were derived from a single or a few major customers.
|
| Black-Scholes Option Pricing Model |
Black-Scholes Option Pricing Model
The
Company uses a Black-Scholes option pricing model to determine the fair value of warrants and options issued.
|
| Recently Issued Accounting Pronouncements |
Recently
Issued Accounting Pronouncements
From
time to time, new accounting pronouncements are issued by the FASB or other standard setting bodies that are adopted by the Company as
of the specified effective date. Unless otherwise discussed, the Company believes that the effect of recently issued standards that are
not yet effective will not have a material effect on its financial position or results of operations upon adoption.
In
August 2020, the FASB issued Accounting Standards Update (“ASU”) 2020-06, Debt – Debt with Conversion and
Other Options (Subtopic 470-20) and Derivatives and Hedging – Contracts in Entity’s Own Equity (Subtopic 815 – 40) (“ASU 2020-06”). ASU 2020-06 simplifies the accounting for certain financial instruments with
characteristics of liabilities and equity, including convertible instruments and contracts on an entity’s own equity. The ASU
is part of the FASB’s simplification initiative, which aims to reduce unnecessary complexity in US GAAP. The ASU’s
amendments are effective for fiscal years beginning after December 15, 2023, and interim periods within those fiscal years. The
Company is currently evaluating the impact of ASU 2020-06 on its financial statements.
|
| Related Parties |
Related
Parties
The
Company follows subtopic 850-10 of FASB ASC 850, Related Party Disclosures for the identification of related parties and disclosure
of related party transactions.
Pursuant
to Section 850-10-20, the related parties include a. affiliates of the Company; b. Entities for which investments in their equity securities
would be required, absent the election of the fair value option under the guidance of Fair Value Option Subsection of Section 825–10–15,
to be accounted for by the equity method by the investing entity; c. trusts for the benefit of employees, such as pension and profit-sharing
trusts that are managed by or under the trusteeship of management; d. principal owners of the Company; e. management of the Company;
f. other parties with which the Company may deal if one party controls or can significantly influence the management or operating policies
of the other to an extent that one of the transacting parties might be prevented from fully pursuing its own separate interests; and
g. other parties that can significantly influence the management or operating policies of the transacting parties or that have an ownership
interest in one of the transacting parties and can significantly influence the other to an extent that one or more of the transacting
parties might be prevented from fully pursuing its own separate interests.
The
financial statements shall include disclosures of material related party transactions, other than compensation arrangements, expense
allowances, and other similar items in the ordinary course of business. However, disclosure of transactions that are eliminated in the
preparation of financial statements is not required in those statements. The disclosures shall include: a. the
nature of the relationship(s) involved; b. a description of the transactions, including transactions to which no amounts or nominal amounts
were ascribed, for each of the periods for which income statements are presented, and such other information deemed necessary to an understanding
of the effects of the transactions on the financial statements; c. the dollar amounts of transactions for each of the periods for which
income statements are presented and the effects of any change in the method of establishing the terms from that used in the preceding
period; and d. amounts due from or to related parties as of the date of each balance sheet presented and, if not otherwise apparent,
the terms and manner of settlement. Material related party transactions have been identified in Notes 3, 6 and 8 in the notes to financial
statements.
|
| Stock-Based Compensation |
Stock-Based
Compensation
The
Company recognizes compensation costs to employees under FASB ASC 718 Compensation - Stock Compensation (“ASC
718”). Under ASC 718, companies are required to measure the compensation costs of share-based compensation arrangements based
on the grant-date fair value and recognize the costs in the financial statements over the period during which employees are required
to provide services. Share-based compensation arrangements include stock options and warrants. As such, compensation cost is
measured on the date of grant at their fair value. Such compensation amounts, if any, are amortized over the respective vesting
periods of the option and warrant grant.
|
| Revenue Recognition |
Revenue
Recognition
The Company follows the provisions of ASC 606. Revenue from Contracts with Customer for recording and recognizing
revenue from customers. The
Company generates our online revenue through the sale of products and services purchased by customers directly through our online platform.
Online revenue represents the sales of products and services on our platform, net of refunds, credits, and chargebacks, and includes
revenue recognition adjustments recorded pursuant to US GAAP. Online revenue is generated by selling directly to consumers through our
websites.
The
Company recognizes revenue when it transfers promised goods or services to customers in an amount that reflects the consideration to
which it expects to be entitled in exchange for those goods or services and has met its performance obligation. For revenue generated
through its online platform, the Company defines its customer as an individual who purchases products or services through websites. The
transaction price in the Company’s contracts with customers is the total amount of consideration to which the Company expects to
be entitled in exchange for transferring products or services to the customer.
The
Company’s contracts that contain prescription products issued as the result of a consultation include two performance obligations:
access to (i) products and (ii) consultation services. The Company’s contracts for prescription refills have a single performance
obligation. Revenue is recognized at the time the related performance obligation is satisfied by transferring the promised product to
the customer and, in contracts that contain services, by the provision of consultation services to the customer. The Company satisfies
its performance obligation for products at a point in time, which is upon delivery of the products to a third-party carrier. The Company
satisfies its performance obligation for services over the period of the consultation service, which is typically a few days. The customer
obtains control of the products and services upon the Company’s completion of its performance obligations.
The
Company has entered into a Physician Services Agreement with BrighterMD, LLC dba Doctegrity (“Doctegrity”) to provide online
telemedicine technology services to the Company. The Company accounts for service revenue as a principal in the arrangement with its
customers. This conclusion is reached because (i) the Company determines which providers provide the consultation to the customer; (ii)
the Company is primarily responsible for the satisfactory fulfillment and acceptability of the services; (iii) the Company incurs costs
for consultation services even for visits that do not result in a prescription and the sale of products; and (iv) the Company, at its
sole discretion, sets all listed prices charged on its websites for products and services.
Additionally,
the Company has entered into a Master Services Agreement and Statement of Work with Epiq Scripts, LLC (“Contracted Pharmacy”),
which is a related party, to provide pharmacy and compounding services to the Company to fulfill its promise to customers for contracts
that include sale of prescription products and to fill prescriptions that are ordered by the Company’s customers for fulfillment
through the Company’s websites. The Company accounts for prescription product revenue as a principal in the arrangement with its
customers. This conclusion is reached because (i) the Company has sole discretion in determining which Contracted Pharmacy fills a customer’s
prescription; (ii) Contracted Pharmacy fills the prescription based on fulfillment instructions provided by the Company, including using
the Company’s branded packaging for generic products; (iii) the Company is primarily responsible to the customer for the satisfactory
fulfillment and acceptability of the order, and; (iv) the Company, at its sole discretion, sets all listed prices charged on its websites
for products and services.
The
Company accounts for shipping activities, consisting of direct costs to ship products performed after the control of a product has been
transferred to the customer, in cost of revenue.
|
| Inventories |
Inventories
Inventories
are stated at the lower of cost or net realizable value with cost being determined on a first-in, first-out (“FIFO”) basis. The Company writes down its
inventory for estimated obsolescence or unmarketable inventory equal to the difference between the cost of inventory and the estimated
market value based upon assumptions about future demand and market conditions. If actual market conditions are less favorable than those
projected by management, additional inventory write-downs may be required. During the years ended December 31, 2023 and 2022, there were no inventory write-downs.
|
| Marketing and Advertising |
Marketing
and Advertising
The
Company follows the policy of charging the costs of marketing and advertising to expense as incurred. The Company charged $2,097,505
and $352,860 towards marketing and advertising for the years ended December 31, 2023 and 2022, respectively.
|
| Subsequent Events |
Subsequent
Events
The
Company follows the guidance in Section 855-10-50 of FASB ASC 855, Subsequent Events, for the disclosure of subsequent
events. The Company will evaluate subsequent events through the date when the financial statements were issued (see Note
12).
|
| X |
- DefinitionRelated Parties Policy [Text Block].
| Name: |
MGRX_RelatedPartiesPolicyTextBlock |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_AccountingPoliciesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for advertising cost. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-SubTopic 35
-Topic 720
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483406/720-35-50-1
| Name: |
us-gaap_AdvertisingCostsPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for basis of accounting, or basis of presentation, used to prepare the financial statements (for example, US Generally Accepted Accounting Principles, Other Comprehensive Basis of Accounting, IFRS).
| Name: |
us-gaap_BasisOfAccountingPolicyPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for cash and cash equivalents, including the policy for determining which items are treated as cash equivalents. Other information that may be disclosed includes (1) the nature of any restrictions on the entity's use of its cash and cash equivalents, (2) whether the entity's cash and cash equivalents are insured or expose the entity to credit risk, (3) the classification of any negative balance accounts (overdrafts), and (4) the carrying basis of cash equivalents (for example, at cost) and whether the carrying amount of cash equivalents approximates fair value. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482913/230-10-50-1
| Name: |
us-gaap_CashAndCashEquivalentsPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for credit risk. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 942
-SubTopic 825
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480981/942-825-50-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (d)
-SubTopic 10
-Topic 275
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-1
| Name: |
us-gaap_ConcentrationRiskCreditRisk |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for computing basic and diluted earnings or loss per share for each class of common stock and participating security. Addresses all significant policy factors, including any antidilutive items that have been excluded from the computation and takes into account stock dividends, splits and reverse splits that occur after the balance sheet date of the latest reporting period but before the issuance of the financial statements. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 260
-SubTopic 10
-Section 50
-Paragraph 1
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 260
-SubTopic 10
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-2
| Name: |
us-gaap_EarningsPerSharePolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for fair value measurements of financial and non-financial assets, liabilities and instruments classified in shareholders' equity. Disclosures include, but are not limited to, how an entity that manages a group of financial assets and liabilities on the basis of its net exposure measures the fair value of those assets and liabilities.
| Name: |
us-gaap_FairValueMeasurementPolicyPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for determining the fair value of financial instruments. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 60
-Paragraph 1
-SubTopic 10
-Topic 820
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482053/820-10-60-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-SubTopic 10
-Topic 825
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-1
| Name: |
us-gaap_FairValueOfFinancialInstrumentsPolicy |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for income taxes, which may include its accounting policies for recognizing and measuring deferred tax assets and liabilities and related valuation allowances, recognizing investment tax credits, operating loss carryforwards, tax credit carryforwards, and other carryforwards, methodologies for determining its effective income tax rate and the characterization of interest and penalties in the financial statements. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-03(h)(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479886/946-10-S99-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 17
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-17
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 9
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-9
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 25
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482525/740-10-45-25
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482525/740-10-45-28
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 19
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-19
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-1
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 20
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-20
| Name: |
us-gaap_IncomeTaxPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of inventory accounting policy for inventory classes, including, but not limited to, basis for determining inventory amounts, methods by which amounts are added and removed from inventory classes, loss recognition on impairment of inventories, and situations in which inventories are stated above cost. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483489/210-10-50-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(6)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 3: http://www.xbrl.org/2003/role/exampleRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483426/235-10-50-4
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 912
-SubTopic 330
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482105/912-330-50-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 330
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//330/tableOfContent
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 330
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483080/330-10-50-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 330
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483080/330-10-50-4
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 6
-Subparagraph (a)
-SubTopic 10
-Topic 270
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482989/270-10-45-6
| Name: |
us-gaap_InventoryPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy pertaining to new accounting pronouncements that may impact the entity's financial reporting. Includes, but is not limited to, quantification of the expected or actual impact.
| Name: |
us-gaap_NewAccountingPronouncementsPolicyPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for long-lived, physical asset used in normal conduct of business and not intended for resale. Includes, but is not limited to, work of art, historical treasure, and similar asset classified as collections. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-SubTopic 10
-Topic 360
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(8)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-SubTopic 360
-Topic 958
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480321/958-360-50-6
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (d)
-SubTopic 360
-Topic 958
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480321/958-360-50-1
| Name: |
us-gaap_PropertyPlantAndEquipmentPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for revenue from contract with customer. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 606
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 17
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479806/606-10-50-17
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 606
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 19
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479806/606-10-50-19
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 606
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 18
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479806/606-10-50-18
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 606
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 18
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479806/606-10-50-18
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 606
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 20
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479806/606-10-50-20
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 606
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 20
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479806/606-10-50-20
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 606
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 20
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479806/606-10-50-20
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 606
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 20
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479806/606-10-50-20
Reference 9: http://www.xbrl.org/2003/role/exampleRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (e)
-SubTopic 10
-Topic 235
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483426/235-10-50-4
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Topic 606
-Publisher FASB
-URI https://asc.fasb.org//606/tableOfContent
| Name: |
us-gaap_RevenueFromContractWithCustomerPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for award under share-based payment arrangement. Includes, but is not limited to, methodology and assumption used in measuring cost. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(v)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SAB Topic 14.C.Q3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479830/718-10-S99-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SAB Topic 14.D.1.Q5)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479830/718-10-S99-1
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SAB Topic 14.D.3.Q2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479830/718-10-S99-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SAB Topic 14.D.2.Q6)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479830/718-10-S99-1
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//718/tableOfContent
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationOptionAndIncentivePlansPolicy |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for reporting subsequent events.
| Name: |
us-gaap_SubsequentEventsPolicyPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for the use of estimates in the preparation of financial statements in conformity with generally accepted accounting principles. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 275
-SubTopic 10
-Section 50
-Paragraph 9
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-9
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 275
-SubTopic 10
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-4
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)
-SubTopic 10
-Topic 275
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (c)
-SubTopic 10
-Topic 275
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-SubTopic 10
-Topic 275
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-11
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 12
-SubTopic 10
-Topic 275
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-12
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 275
-SubTopic 10
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-8
| Name: |
us-gaap_UseOfEstimates |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Tables)
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Accounting Policies [Abstract] |
|
| SCHEDULE OF FINANCIAL INSTRUMENTS MEASUREMENT AT FAIR VALUE |
SCHEDULE
OF FINANCIAL INSTRUMENTS MEASUREMENT AT FAIR VALUE
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| | |
Fair Value Measurements at December 31, 2023 | |
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| Assets | |
| | | |
| | | |
| | |
| Cash | |
$ | 739,006 | | |
$ | - | | |
$ | - | |
| Total assets | |
| 739,006 | | |
| - | | |
| - | |
| Liabilities | |
| | | |
| | | |
| | |
| Total liabilities | |
| - | | |
| - | | |
| - | |
| Fair value, net asset
(liability) | |
$ | 739,006 | | |
$ | - | | |
$ | - | |
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| | |
Fair Value Measurements at December 31, 2022 | |
| | |
Level 1 | | |
Level 2 | | |
Level 3 | |
| Assets | |
| | | |
| | | |
| | |
| Cash | |
$ | 682,860 | | |
$ | - | | |
$ | - | |
| Total assets | |
| 682,860 | | |
| - | | |
| - | |
| Liabilities | |
| | | |
| | | |
| | |
| Total liabilities | |
| - | | |
| - | | |
| - | |
| Fair value, net asset
(liability) | |
$ | 682,860 | | |
$ | - | | |
$ | - | |
|
| X |
- References
+ Details
| Name: |
us-gaap_AccountingPoliciesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure of assets and liabilities, including [financial] instruments measured at fair value that are classified in stockholders' equity, if any, that are measured at fair value on a recurring basis. The disclosures contemplated herein include the fair value measurements at the reporting date by the level within the fair value hierarchy in which the fair value measurements in their entirety fall, segregating fair value measurements using quoted prices in active markets for identical assets (Level 1), significant other observable inputs (Level 2), and significant unobservable inputs (Level 3). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 820
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482106/820-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 820
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482106/820-10-50-2
| Name: |
us-gaap_ScheduleOfFairValueAssetsAndLiabilitiesMeasuredOnRecurringBasisTableTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
PROPERTY, PLANT AND EQUIPMENT (Tables)
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Property, Plant and Equipment [Abstract] |
|
| SCHEDULE OF PROPERTY PLANT AND EQUIPMENT |
SCHEDULE
OF PROPERTY PLANT AND EQUIPMENT
| | |
December 31, 2023 | | |
December 31, 2022 | |
| | |
| | |
| |
| Computers | |
| 5,062 | | |
| 5,062 | |
| Equipment | |
| 119,819 | | |
| 116,300 | |
| Less accumulated depreciation: | |
| (28,752 | ) | |
| (3,863 | ) |
| Property and equipment, net | |
| 96,129 | | |
| 117,499 | |
|
| X |
- References
+ Details
| Name: |
us-gaap_PropertyPlantAndEquipmentAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure of physical assets used in the normal conduct of business and not intended for resale. Includes, but is not limited to, balances by class of assets, depreciation and depletion expense and method used, including composite depreciation, and accumulated deprecation. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-SubTopic 10
-Topic 360
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
| Name: |
us-gaap_PropertyPlantAndEquipmentTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
CAPITAL STOCK (Tables)
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Share-Based Compensation Arrangement by Share-Based Payment Award [Line Items] |
|
| SCHEDULE OF STOCK OPTION ACTIVITY |
The
following table summarizes common stock options activity: The following table summarizes common stock options activity:
SCHEDULE
OF STOCK OPTION ACTIVITY
| | |
Options | | |
Weighted Average Exercise Price | |
| December 31, 2021 | |
| - | | |
$ | - | |
| Granted | |
| 1,250,000 | | |
| 1.10 | |
| Exercised | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Outstanding, December 31, 2022 | |
| 1,250,000 | | |
$ | 1.10 | |
| Exercisable, December 31, 2022 | |
| 133,333 | | |
$ | 1.10 | |
| Outstanding, December 31, 2023 | |
| 1,250,000 | | |
$ | 1.10 | |
| | |
| | | |
| | |
| Granted | |
| 1,400,000 | | |
$ | 0.40 | |
| Exercised | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Outstanding, December 31, 2023 | |
| 2,650,000 | | |
$ | 0.73 | |
| Exercisable, December 31, 2023 | |
| 1,812,500 | | |
$ | 0.73 | |
The
weighted average exercise prices, remaining lives for options granted, and exercisable as of December 31, 2023 were as follows:
| | | |
Outstanding Options | | |
| | |
Exercisable Options | |
Options Exercise Price Per Share | | |
Shares | | |
Life (Years) | | |
Weighted Average Exercise Price | | |
Shares | | |
Weighted Average Exercise Price | |
| $ | 1.10 | | |
| 1,400,000 | | |
| 4.28 | | |
$ | 1.10 | | |
| 562,500 | | |
$ | 1.10 | |
| $ | 0.32 | | |
| 1,250,000 | | |
| 5.00 | | |
$ | 0.32 | | |
| 1,250,000 | | |
$ | 0.32 | |
|
| SCHEDULE OF FAIR VALUE ASSUMPTIONS |
SCHEDULE
OF FAIR VALUE ASSUMPTIONS
| Fair Value of Common Stock on measurement date | |
$ | 1.00 - 0.32 | |
| Risk free interest rate | |
| 3.83% - 3.30 | % |
| Volatility | |
| 232.05% 92.54 | % |
| Dividend Yield | |
| 0 | % |
| Expected Term | |
| 6.0 - 3.5 | |
| |
(1) |
The risk-free interest
rate was determined by management using the market yield on U.S. Treasury securities with comparable terms as of the measurement
date. |
| |
(2) |
The trading volatility
was determined by calculating the volatility of the Company’s peer group. |
| |
(3) |
The Company does not expect
to pay a dividend in the foreseeable future |
| |
(4) |
The Company, in accordance
with staff accounting bulletin (“SAB”)14-D.2, used the simplified method (plain vanilla) to determine the overall expected
term |
|
| SCHEDULE OF WARRANT ACTIVITY |
The
following table summarizes common stock warrants activity:
SCHEDULE
OF WARRANT ACTIVITY
| | |
Warrants | | |
Weighted Average Exercise Price | |
| Outstanding, December 31, 2021 | |
| - | | |
$ | - | |
| Granted | |
| 2,210,070 | | |
| 1.00 | |
| Exercised | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Cancelled | |
| (210,070 | ) | |
| 1.00 | |
| Outstanding, December 31, 2022 | |
| 2,000,000 | | |
| 1.00 | |
| Exercisable, December 31, 2022 | |
| 2,000,000 | | |
$ | 1.00 | |
| | |
| | | |
| | |
| Granted | |
| 367,500 | | |
| 1.22 | |
| Exercised | |
| (1,024,500 | ) | |
| 1.00 | |
| Expired | |
| - | | |
| - | |
| Cancelled | |
| - | | |
| - | |
| Outstanding, December 31, 2023 | |
| 1,343,000 | | |
| 1.43 | |
| Exercisable, December 31, 2023 | |
| 1,343,000 | | |
$ | 1.43 | |
The
weighted average exercise prices, remaining lives for warrants granted, and exercisable as of December 31, 2023, were as follows:
| | | |
Outstanding and Vested Warrants | |
Weighted Average Warrant
Exercise Price Per Share | | |
Shares | | |
Life (Years) | |
| $ | 1.43 | | |
| 1,343,000 | | |
| 2.07 | |
|
| Warrant [Member] |
|
| Share-Based Compensation Arrangement by Share-Based Payment Award [Line Items] |
|
| SCHEDULE OF FAIR VALUE ASSUMPTIONS |
SCHEDULE
OF FAIR VALUE ASSUMPTIONS
| Fair Value of Common Stock on measurement date | |
$ | 0.37 - $0.97 | |
| Risk-free interest rate | |
| From 2.95% to 4.00 | % |
| Volatility | |
| From 81.92% to 169.22 | % |
| Dividend Yield | |
| 0 | % |
| Expected Term | |
| 5 years | |
| |
(1) |
The risk-free interest
rate was determined by management using the market yield on U.S. Treasury securities with comparable terms as of the measurement
date. |
| |
(2) |
The trading volatility
was determined by calculating the volatility of the Company’s peer group. |
| |
(3) |
The Company does not expect
to pay a dividend in the foreseeable future. |
|
| X |
- DefinitionTabular disclosure for stock option plans. Includes, but is not limited to, outstanding awards at beginning and end of year, grants, exercises, forfeitures, and weighted-average grant date fair value. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 2
-Subparagraph (c)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)
-SubTopic 10
-Topic 718
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)
-SubTopic 10
-Topic 718
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ScheduleOfShareBasedCompensationStockOptionsActivityTableTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure of the significant assumptions used during the year to estimate the fair value of stock options, including, but not limited to: (a) expected term of share options and similar instruments, (b) expected volatility of the entity's shares, (c) expected dividends, (d) risk-free rate(s), and (e) discount for post-vesting restrictions. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 718
-SubTopic 10
-Subparagraph (f)(2)
-Name Accounting Standards Codification
-Paragraph 2
-Section 50
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ScheduleOfShareBasedPaymentAwardStockOptionsValuationAssumptionsTableTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure of warrants or rights issued. Warrants and rights outstanding are derivative securities that give the holder the right to purchase securities (usually equity) from the issuer at a specific price within a certain time frame. Warrants are often included in a new debt issue to entice investors by a higher return potential. The main difference between warrants and call options is that warrants are issued and guaranteed by the company, whereas options are exchange instruments and are not issued by the company. Also, the lifetime of a warrant is often measured in years, while the lifetime of a typical option is measured in months. Disclose the title of issue of securities called for by warrants and rights outstanding, the aggregate amount of securities called for by warrants and rights outstanding, the date from which the warrants or rights are exercisable, and the price at which the warrant or right is exercisable. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-1
| Name: |
us-gaap_ScheduleOfStockholdersEquityNoteWarrantsOrRightsTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 1D
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-1D
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 20: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(v)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_AwardTypeAxis=us-gaap_WarrantMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
| X |
- References
+ Details
| Name: |
us-gaap_CommitmentsAndContingenciesDisclosureAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure of undiscounted cash flows of lessee's operating lease liability. Includes, but is not limited to, reconciliation of undiscounted cash flows to operating lease liability recognized in statement of financial position. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-6
| Name: |
us-gaap_LesseeOperatingLeaseLiabilityMaturityTableTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
INCOME TAXES (Tables)
|
12 Months Ended |
Dec. 31, 2023 |
|---|
| Income Tax Disclosure [Abstract] |
|
| SCHEDULE OF DEFERRED TAX ASSET |
The
Company has a deferred tax asset as shown in the following:
SCHEDULE
OF DEFERRED TAX ASSET
| | |
Year Ending December 31, 2023 | | |
Year Ending December 31, 2022 | |
| Deferred Tax Asset | |
| 11,228,173 | | |
| 2,015,756 | |
| Valuation Allowance | |
| (11,228,173 | ) | |
| (2,015,756 | ) |
| Net Deferred Tax Asset | |
$ | — | | |
$ | — | |
|
| X |
- DefinitionTabular disclosure of the components of net deferred tax asset or liability recognized in an entity's statement of financial position, including the following: the total of all deferred tax liabilities, the total of all deferred tax assets, the total valuation allowance recognized for deferred tax assets. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Paragraph 2
-Section 50
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-2
| Name: |
us-gaap_ScheduleOfDeferredTaxAssetsAndLiabilitiesTableTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
ORGANIZATION AND DESCRIPTION OF THE BUSINESS (Details Narrative) - USD ($)
|
1 Months Ended |
12 Months Ended |
Mar. 31, 2023 |
Dec. 31, 2023 |
Dec. 31, 2022 |
| Subsidiary, Sale of Stock [Line Items] |
|
|
|
| Net proceeds from issuance of common stock |
|
$ 6,200,000
|
$ 2,000,000
|
| IPO [Member] |
|
|
|
| Subsidiary, Sale of Stock [Line Items] |
|
|
|
| Issuance of shares |
1,250,000
|
|
|
| Share issued price per share |
$ 4.00
|
|
|
| Net proceeds from issuance of common stock |
$ 4,350,000
|
|
|
| Resale Prospectus [Member] |
|
|
|
| Subsidiary, Sale of Stock [Line Items] |
|
|
|
| Issuance of shares |
2,000,000
|
|
|
| Share issued price per share |
$ 1.00
|
|
|
| Sale of stock, shares |
4,765,000
|
|
|
| X |
- DefinitionThe cash inflow from the additional capital contribution to the entity. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 14
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-14
| Name: |
us-gaap_ProceedsFromIssuanceOfCommonStock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of shares issued or sold by the subsidiary or equity method investee per stock transaction.
| Name: |
us-gaap_SaleOfStockNumberOfSharesIssuedInTransaction |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionPer share or per unit amount of equity securities issued.
| Name: |
us-gaap_SharesIssuedPricePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of new stock issued during the period. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 505
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481004/946-505-50-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-03(i)(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479886/946-10-S99-3
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesNewIssues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.
| Name: |
us-gaap_SubsidiarySaleOfStockLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=us-gaap_IPOMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=MGRX_ResaleProspectusMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
SCHEDULE OF FINANCIAL INSTRUMENTS MEASUREMENT AT FAIR VALUE (Details) - USD ($)
|
Dec. 31, 2023 |
Dec. 31, 2022 |
| Fair Value, Inputs, Level 1 [Member] |
|
|
| Platform Operator, Crypto-Asset [Line Items] |
|
|
| Cash |
$ 739,006
|
$ 682,860
|
| Total assets |
739,006
|
682,860
|
| Total liabilities |
|
|
| Fair value, net asset (liability) |
739,006
|
682,860
|
| Fair Value, Inputs, Level 2 [Member] |
|
|
| Platform Operator, Crypto-Asset [Line Items] |
|
|
| Cash |
|
|
| Total assets |
|
|
| Total liabilities |
|
|
| Fair value, net asset (liability) |
|
|
| Fair Value, Inputs, Level 3 [Member] |
|
|
| Platform Operator, Crypto-Asset [Line Items] |
|
|
| Cash |
|
|
| Total assets |
|
|
| Total liabilities |
|
|
| Fair value, net asset (liability) |
|
|
| X |
- DefinitionFair value portion of probable future economic benefits obtained or controlled by an entity as a result of past transactions or events. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 820
-SubTopic 10
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482106/820-10-50-2
| Name: |
us-gaap_AssetsFairValueDisclosure |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionFair value portion of currency on hand as well as demand deposits with banks or financial institutions. Includes other kinds of accounts that have the general characteristics of demand deposits. Also includes short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates.
| Name: |
us-gaap_CashAndCashEquivalentsFairValueDisclosure |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionFair value of asset after deduction of liability.
| Name: |
us-gaap_FairValueNetAssetLiability |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionFair value of financial and nonfinancial obligations. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 820
-SubTopic 10
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482106/820-10-50-2
| Name: |
us-gaap_LiabilitiesFairValueDisclosure |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
v3.24.1.u1
SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (Details Narrative) - USD ($)
|
12 Months Ended |
Dec. 31, 2023 |
Dec. 31, 2022 |
| Property, Plant and Equipment [Line Items] |
|
|
| Cash, FDIC insured amount |
$ 250,000
|
|
| Cash equivalents |
0
|
$ 0
|
| Inventory write down |
0
|
0
|
| Marketing and advertising expense |
$ 2,097,505
|
$ 352,860
|
| Minimum [Member] |
|
|
| Property, Plant and Equipment [Line Items] |
|
|
| Property plant and equipment estimated useful lives |
3 years
|
|
| Maximum [Member] |
|
|
| Property, Plant and Equipment [Line Items] |
|
|
| Property plant and equipment estimated useful lives |
5 years
|
|
| Options [Member] |
|
|
| Property, Plant and Equipment [Line Items] |
|
|
| Anti-dilutive securities |
2,650,000
|
1,250,000
|
| Warrant [Member] |
|
|
| Property, Plant and Equipment [Line Items] |
|
|
| Anti-dilutive securities |
1,343,000
|
2,000,000
|
| X |
- DefinitionSecurities (including those issuable pursuant to contingent stock agreements) that could potentially dilute basic earnings per share (EPS) or earnings per unit (EPU) in the future that were not included in the computation of diluted EPS or EPU because to do so would increase EPS or EPU amounts or decrease loss per share or unit amounts for the period presented. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
| Name: |
us-gaap_AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareAmount |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Excludes cash and cash equivalents within disposal group and discontinued operation. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(2))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 2: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CashEquivalentsAtCarryingValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of cash deposited in financial institutions as of the balance sheet date that is insured by the Federal Deposit Insurance Corporation.
| Name: |
us-gaap_CashFDICInsuredAmount |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of loss from reductions in inventory due to subsequent measurement adjustments, including, but not limited to, physical deterioration, obsolescence, or changes in price levels. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 330
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483080/330-10-50-2
| Name: |
us-gaap_InventoryWriteDown |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe total expense recognized in the period for promotion, public relations, and brand or product advertising. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.4)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
| Name: |
us-gaap_MarketingAndAdvertisingExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.
| Name: |
us-gaap_PropertyPlantAndEquipmentLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionUseful life of long lived, physical assets used in the normal conduct of business and not intended for resale, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days. Examples include, but not limited to, land, buildings, machinery and equipment, office equipment, furniture and fixtures, and computer equipment.
| Name: |
us-gaap_PropertyPlantAndEquipmentUsefulLife |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- Details
| Name: |
srt_RangeAxis=srt_MinimumMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_RangeAxis=srt_MaximumMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareByAntidilutiveSecuritiesAxis=MGRX_OptionsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareByAntidilutiveSecuritiesAxis=us-gaap_WarrantMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
| X |
- DefinitionThe aggregate of all deposit liabilities held by the entity, including foreign and domestic, interest and noninterest bearing; may include demand deposits, saving deposits, Negotiable Order of Withdrawal (NOW) and time deposits among others. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 942
-SubTopic 210
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-03.12)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479853/942-210-S99-1
| Name: |
us-gaap_Deposits |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
v3.24.1.u1
| X |
- References
+ Details
| Name: |
us-gaap_InventoryDisclosureAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount after valuation and LIFO reserves of inventory expected to be sold, or consumed within one year or operating cycle, if longer. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483467/210-10-45-1
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(6))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_InventoryNet |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
v3.24.1.u1
SCHEDULE OF PROPERTY PLANT AND EQUIPMENT (Details) - USD ($)
|
Dec. 31, 2023 |
Dec. 31, 2022 |
| Property, Plant and Equipment [Line Items] |
|
|
| Less accumulated depreciation: |
$ (28,752)
|
$ (3,863)
|
| Property and equipment, net |
96,129
|
117,499
|
| Computer Equipment [Member] |
|
|
| Property, Plant and Equipment [Line Items] |
|
|
| Equipment |
5,062
|
5,062
|
| Equipment [Member] |
|
|
| Property, Plant and Equipment [Line Items] |
|
|
| Equipment |
$ 119,819
|
$ 116,300
|
| X |
- DefinitionAmount of accumulated depreciation, depletion and amortization for physical assets used in the normal conduct of business to produce goods and services. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(8)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(14))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 360
-SubTopic 10
-Section 50
-Paragraph 1
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
| Name: |
us-gaap_AccumulatedDepreciationDepletionAndAmortizationPropertyPlantAndEquipment |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount before accumulated depreciation, depletion and amortization of physical assets used in the normal conduct of business and not intended for resale. Examples include, but are not limited to, land, buildings, machinery and equipment, office equipment, and furniture and fixtures. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(8))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(13))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 360
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
| Name: |
us-gaap_PropertyPlantAndEquipmentGross |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.
| Name: |
us-gaap_PropertyPlantAndEquipmentLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount after accumulated depreciation, depletion and amortization of physical assets used in the normal conduct of business to produce goods and services and not intended for resale. Examples include, but are not limited to, land, buildings, machinery and equipment, office equipment, and furniture and fixtures. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-SubTopic 10
-Topic 360
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(8))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 360
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480842/942-360-50-1
| Name: |
us-gaap_PropertyPlantAndEquipmentNet |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- Details
| Name: |
us-gaap_PropertyPlantAndEquipmentByTypeAxis=us-gaap_ComputerEquipmentMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_PropertyPlantAndEquipmentByTypeAxis=us-gaap_EquipmentMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
| X |
- DefinitionThe amount of expense recognized in the current period that reflects the allocation of the cost of tangible assets over the assets' useful lives. Includes production and non-production related depreciation. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Subparagraph (b)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 360
-SubTopic 10
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
| Name: |
us-gaap_Depreciation |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_PropertyPlantAndEquipmentAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount after accumulated depreciation, depletion and amortization of physical assets used in the normal conduct of business to produce goods and services and not intended for resale. Examples include, but are not limited to, land, buildings, machinery and equipment, office equipment, and furniture and fixtures. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-SubTopic 10
-Topic 360
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(8))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 360
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480842/942-360-50-1
| Name: |
us-gaap_PropertyPlantAndEquipmentNet |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
v3.24.1.u1
LOANS FROM RELATED PARTIES (Details Narrative) - USD ($)
|
|
|
|
|
|
12 Months Ended |
Aug. 18, 2022 |
Jun. 29, 2022 |
Jun. 16, 2022 |
Mar. 18, 2022 |
Dec. 10, 2021 |
Dec. 31, 2023 |
Dec. 31, 2022 |
| Related Party Transaction [Line Items] |
|
|
|
|
|
|
|
| Proceeds from issuance of common stock |
|
|
|
|
|
$ 6,200,000
|
$ 2,000,000
|
| Repayment of debt |
|
|
|
|
|
89,200
|
25,070
|
| Proceeds from related party |
|
|
|
|
|
|
75,000
|
| Cohen Enterprises [Member] |
|
|
|
|
|
|
|
| Related Party Transaction [Line Items] |
|
|
|
|
|
|
|
| Related party advances received |
|
$ 25,000
|
|
|
|
|
|
| Cohen Enterprises Inc [Member] |
|
|
|
|
|
|
|
| Related Party Transaction [Line Items] |
|
|
|
|
|
|
|
| Equity ownership interest |
|
|
80.00%
|
|
|
|
|
| Cohen Enterprises Inc [Member] |
|
|
|
|
|
|
|
| Related Party Transaction [Line Items] |
|
|
|
|
|
|
|
| Issuance of shares |
|
|
8,000,000
|
|
|
|
|
| Proceeds from issuance of common stock |
|
|
$ 90,000
|
|
|
|
|
| Repayment of debt |
$ 25,000
|
|
|
|
|
|
89,200
|
| Proceeds from related party |
|
|
|
|
|
$ 0
|
89,200
|
| Imputed interest percentage |
|
|
|
|
|
8.00%
|
|
| Related party transaction purchases |
|
|
|
|
|
$ 8,232
|
|
| Zip Doctor Inc [Member] |
|
|
|
|
|
|
|
| Related Party Transaction [Line Items] |
|
|
|
|
|
|
|
| Related party advances received |
|
|
|
|
$ 70
|
|
|
| Repayment of debt |
|
|
|
|
|
$ 0
|
$ 70
|
| Imputed interest percentage |
|
|
|
|
|
8.00%
|
|
| Stock Purchase Agreement [Member] |
|
|
|
|
|
|
|
| Related Party Transaction [Line Items] |
|
|
|
|
|
|
|
| Related party advances received |
|
|
|
$ 50,000
|
$ 39,200
|
$ 89,200
|
|
| Repayment of debt |
|
|
$ 89,200
|
|
|
|
|
| X |
- DefinitionThe percentage of ownership of common stock or equity participation in the investee accounted for under the equity method of accounting. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
| Name: |
us-gaap_EquityMethodInvestmentOwnershipPercentage |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe cash outflow from advancing money to an affiliate (an entity that is related but not strictly controlled by the entity). Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 13
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-13
| Name: |
us-gaap_PaymentsForAdvanceToAffiliate |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash inflow from the additional capital contribution to the entity. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 14
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-14
| Name: |
us-gaap_ProceedsFromIssuanceOfCommonStock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of new stock issued during the period. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 505
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481004/946-505-50-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-03(i)(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479886/946-10-S99-3
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesNewIssues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
srt_TitleOfIndividualAxis=MGRX_CohenEnterprisesMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_ScheduleOfEquityMethodInvestmentEquityMethodInvesteeNameAxis=MGRX_CohenEnterprisesIncMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_TypeOfArrangementAxis=MGRX_StockPurchaseAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
NOTES PAYABLE (Details Narrative) - USD ($)
|
|
|
2 Months Ended |
12 Months Ended |
|
Apr. 01, 2023 |
Mar. 23, 2023 |
Mar. 01, 2023 |
Dec. 31, 2023 |
Dec. 31, 2022 |
Nov. 18, 2022 |
| Debt Disclosure [Abstract] |
|
|
|
|
|
|
| Debt instrument face amount |
|
|
|
|
|
$ 78,260
|
| Debt instrument periodic payment |
$ 31,630
|
|
$ 5,000
|
|
|
|
| Repayments of notes payable |
|
$ 63,260
|
|
$ 78,260
|
|
|
| Notes payable |
|
|
|
|
|
|
| X |
- References
+ Details
| Name: |
us-gaap_DebtDisclosureAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFace (par) amount of debt instrument at time of issuance. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 835
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482900/835-30-50-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-1B
Reference 3: http://www.xbrl.org/2003/role/exampleRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 55
-Paragraph 69B
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481568/470-20-55-69B
Reference 4: http://www.xbrl.org/2003/role/exampleRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 55
-Paragraph 69C
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481568/470-20-55-69C
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 835
-SubTopic 30
-Section 45
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482925/835-30-45-2
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 835
-SubTopic 30
-Section 55
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482949/835-30-55-8
| Name: |
us-gaap_DebtInstrumentFaceAmount |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of the required periodic payments including both interest and principal payments. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.22)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 942
-SubTopic 470
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480848/942-470-50-3
| Name: |
us-gaap_DebtInstrumentPeriodicPayment |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionIncluding the current and noncurrent portions, aggregate carrying amount of all types of notes payable, as of the balance sheet date, with initial maturities beyond one year or beyond the normal operating cycle, if longer. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(22))
-SubTopic 10
-Topic 210
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 2: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(16)(a)(2))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-03(16))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479853/942-210-S99-1
| Name: |
us-gaap_NotesPayable |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe cash outflow for a borrowing supported by a written promise to pay an obligation. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 15
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-15
| Name: |
us-gaap_RepaymentsOfNotesPayable |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
v3.24.1.u1
SCHEDULE OF STOCK OPTION ACTIVITY (Details) - $ / shares
|
12 Months Ended |
Dec. 31, 2023 |
Dec. 31, 2022 |
| Share-Based Payment Arrangement, Option, Exercise Price Range [Line Items] |
|
|
| Options, Outstanding Beginning Balance |
1,250,000
|
|
| Weighted Average Exercise Price, Outstanding Beginning Balance |
$ 1.10
|
|
| Options, Granted |
1,400,000
|
1,250,000
|
| Weighted Average Exercise Price, Granted |
$ 0.40
|
$ 1.10
|
| Options, Exercised |
|
|
| Weighted Average Exercise Price, Exercised |
|
|
| Options, Expired |
|
|
| Weighted Average Exercise Price, Expired |
|
|
| Outstanding Ending Balance |
2,650,000
|
1,250,000
|
| Weighted Average Outstanding Ending Balance |
$ 0.73
|
$ 1.10
|
| Exercisable Ending Balance |
1,812,500
|
133,333
|
| Weighted Average Exercisable Price Ending Balance |
$ 0.73
|
$ 1.10
|
| Range One [Member] |
|
|
| Share-Based Payment Arrangement, Option, Exercise Price Range [Line Items] |
|
|
| Outstanding Ending Balance |
1,400,000
|
|
| Weighted Average Outstanding Ending Balance |
$ 1.10
|
|
| Exercisable Ending Balance |
562,500
|
|
| Weighted Average Exercisable Price Ending Balance |
$ 1.10
|
|
| Options Term |
4 years 3 months 10 days
|
|
| Range Two [Member] |
|
|
| Share-Based Payment Arrangement, Option, Exercise Price Range [Line Items] |
|
|
| Outstanding Ending Balance |
1,250,000
|
|
| Weighted Average Outstanding Ending Balance |
$ 0.32
|
|
| Exercisable Ending Balance |
1,250,000
|
|
| Weighted Average Exercisable Price Ending Balance |
$ 0.32
|
|
| Options Term |
5 years
|
|
| X |
- DefinitionThe number of shares into which fully or partially vested stock options outstanding as of the balance sheet date can be currently converted under the option plan. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsExercisableNumber |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe weighted-average price as of the balance sheet date at which grantees can acquire the shares reserved for issuance on vested portions of options outstanding and currently exercisable under the stock option plan. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsExercisableWeightedAverageExercisePrice |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of options or other stock instruments for which the right to exercise has lapsed under the terms of the plan agreements. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsExpirationsInPeriod |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionGross number of share options (or share units) granted during the period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsGrantsInPeriodGross |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of options outstanding, including both vested and non-vested options. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingNumber |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionWeighted average price at which grantees can acquire the shares reserved for issuance under the stock option plan. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingWeightedAverageExercisePrice |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionWeighted average price at which option holders acquired shares when converting their stock options into shares. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsExercisesInPeriodWeightedAverageExercisePrice |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionWeighted average price at which grantees could have acquired the underlying shares with respect to stock options of the plan that expired. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsExpirationsInPeriodWeightedAverageExercisePrice |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionWeighted average per share amount at which grantees can acquire shares of common stock by exercise of options. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsGrantsInPeriodWeightedAverageExercisePrice |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.
| Name: |
us-gaap_ShareBasedCompensationSharesAuthorizedUnderStockOptionPlansExercisePriceRangeLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionWeighted average remaining contractual term of outstanding stock options, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 718
-SubTopic 10
-Subparagraph (e)(1)
-Name Accounting Standards Codification
-Paragraph 2
-Section 50
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_SharebasedCompensationSharesAuthorizedUnderStockOptionPlansExercisePriceRangeOutstandingOptionsWeightedAverageRemainingContractualTerm2 |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of share options (or share units) exercised during the current period. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 4: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesStockOptionsExercised |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_ShareBasedCompensationSharesAuthorizedUnderStockOptionPlansByExercisePriceRangeAxis=MGRX_RangeOneMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_ShareBasedCompensationSharesAuthorizedUnderStockOptionPlansByExercisePriceRangeAxis=MGRX_RangeTwoMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-4
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 5
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-5
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481674/830-30-50-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 17
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-17
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 20
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-20
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 20
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-20
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 20
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-20
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 20
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-20
| Name: |
us-gaap_AccumulatedOtherComprehensiveIncomeLossLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAgreed-upon price for the exchange of the underlying asset relating to the share-based payment award.
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExercisePrice |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe estimated dividend rate (a percentage of the share price) to be paid (expected dividends) to holders of the underlying shares over the option's term. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedDividendRate |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe estimated measure of the percentage by which a share price is expected to fluctuate during a period. Volatility also may be defined as a probability-weighted measure of the dispersion of returns about the mean. The volatility of a share price is the standard deviation of the continuously compounded rates of return on the share over a specified period. That is the same as the standard deviation of the differences in the natural logarithms of the stock prices plus dividends, if any, over the period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedVolatilityRate |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe risk-free interest rate assumption that is used in valuing an option on its own shares. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsRiskFreeInterestRate |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionExpected term of award under share-based payment arrangement, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardFairValueAssumptionsExpectedTerm1 |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_StatementEquityComponentsAxis=us-gaap_WarrantMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_RangeAxis=srt_MaximumMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_RangeAxis=srt_MinimumMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
SCHEDULE OF WARRANT ACTIVITY (Details) - $ / shares
|
12 Months Ended |
Dec. 31, 2023 |
Dec. 31, 2022 |
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
| Weighted Average Exercise Price Exercised |
$ 1.43
|
|
| Warrant [Member] |
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
| Warrants Beginning Balance |
2,000,000
|
|
| Weighted Average Exercise Price Beginning Balance |
$ 1.00
|
|
| Warrants Granted |
367,500
|
2,210,070
|
| Weighted Average Exercise Price Granted |
$ 1.22
|
$ 1.00
|
| Warrants Exercised |
(1,024,500)
|
|
| Weighted Average Exercise Price Exercised |
$ 1.00
|
|
| Warrants Expired |
|
|
| Weighted Average Exercise Price Expired |
|
|
| Warrants Cancelled |
|
(210,070)
|
| Weighted Average Exercise Price Cancelled |
|
$ 1.00
|
| Warrants Outstanding, Ending balance |
1,343,000
|
2,000,000
|
| Weighted Average Exercise Price Ending Balance |
$ 1.43
|
$ 1.00
|
| Warrants Exercisable Ending balance |
1,343,000
|
2,000,000
|
| Weighted Average Exercise Price Exercisable price |
$ 1.43
|
$ 1.00
|
| Outstanding and Vested Warrants Ending balance |
1,343,000
|
|
| Outstanding and Vested Warrants Expected Term |
2 years 25 days
|
|
| X |
- DefinitionShare based compensation arrangement by share based payment award equity instruments other than options exercised in period.
| Name: |
MGRX_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsExercisedInPeriod |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionShare based payment award equity instruments other than options exercised in period weighted average grant date fair value.
| Name: |
MGRX_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsExercisedInPeriodWeightedAverageGrantDateFairValue |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionShare based compensation arrangement by share based payment award equity instruments other than options expired in period.
| Name: |
MGRX_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsExpiredInPeriod |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionShare based payment award equity instruments other than options expired in period weighted average grant date fair value.
| Name: |
MGRX_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsExpiredInPeriodWeightedAverageGrantDateFairValue |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of vested equity-based payment instruments, excluding stock (or unit) options, that validly exist and are outstanding as of the balance sheet date
| Name: |
MGRX_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsVestedNumber |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionShare based compensation arrangement by share based payment award other than options exercisable number.
| Name: |
MGRX_ShareBasedCompensationArrangementByShareBasedPaymentAwardOtherThanOptionsExercisableNumber |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionShare based payment award other than options exercisable weighted average exercise price.
| Name: |
MGRX_ShareBasedCompensationArrangementByShareBasedPaymentAwardOtherThanOptionsExercisableWeightedAverageExercisePrice |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionShare based compensation arrangement by share based payment award fair value assumptions expected term.
| Name: |
MGRX_SharebasedCompensationArrangementBySharebasedPaymentAwardFairValueAssumptionsExpectedTerm |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-4
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 5
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-5
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481674/830-30-50-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 17
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-17
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 20
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-20
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 20
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-20
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 20
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-20
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 20
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-20
| Name: |
us-gaap_AccumulatedOtherComprehensiveIncomeLossLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of equity-based payment instruments, excluding stock (or unit) options, that were forfeited during the reporting period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsForfeitedInPeriod |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionWeighted average fair value as of the grant date of equity-based award plans other than stock (unit) option plans that were not exercised or put into effect as a result of the occurrence of a terminating event. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsForfeituresWeightedAverageGrantDateFairValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of grants made during the period on other than stock (or unit) option plans (for example, phantom stock or unit plan, stock or unit appreciation rights plan, performance target plan). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsGrantsInPeriod |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe weighted average fair value at grant date for nonvested equity-based awards issued during the period on other than stock (or unit) option plans (for example, phantom stock or unit plan, stock or unit appreciation rights plan, performance target plan). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsGrantsInPeriodWeightedAverageGrantDateFairValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of non-vested equity-based payment instruments, excluding stock (or unit) options, that validly exist and are outstanding as of the balance sheet date. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedNumber |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionPer share or unit weighted-average fair value of nonvested award under share-based payment arrangement. Excludes share and unit options. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardEquityInstrumentsOtherThanOptionsNonvestedWeightedAverageGrantDateFairValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- Details
| Name: |
us-gaap_StatementEquityComponentsAxis=us-gaap_WarrantMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
CAPITAL STOCK (Details Narrative) - USD ($)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1 Months Ended |
2 Months Ended |
12 Months Ended |
|
|
Dec. 19, 2023 |
Dec. 11, 2023 |
Nov. 15, 2023 |
Nov. 01, 2023 |
Oct. 10, 2023 |
Oct. 01, 2023 |
Jun. 27, 2023 |
Jun. 22, 2023 |
Jun. 21, 2023 |
Jun. 08, 2023 |
Jun. 07, 2023 |
Jun. 06, 2023 |
Jun. 05, 2023 |
Jun. 01, 2023 |
May 25, 2023 |
May 01, 2023 |
Apr. 26, 2023 |
Apr. 25, 2023 |
Apr. 24, 2023 |
Apr. 01, 2023 |
Mar. 22, 2023 |
Mar. 20, 2023 |
Jan. 24, 2023 |
Jan. 06, 2023 |
Jan. 03, 2023 |
Mar. 31, 2023 |
Mar. 01, 2023 |
Dec. 31, 2023 |
Dec. 31, 2022 |
Dec. 28, 2023 |
Dec. 31, 2021 |
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Preferred stock, shares authorized |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10,000,000
|
10,000,000
|
|
|
| Preferred stock par value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 0.0001
|
$ 0.0001
|
|
|
| Common stock, shares authorized |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
200,000,000
|
200,000,000
|
|
|
| Common stock, par value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 0.0001
|
$ 0.0001
|
|
|
| Common stock, shares issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
21,419,500
|
13,365,000
|
|
|
| Common stock, shares outstanding |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
21,419,500
|
13,365,000
|
|
|
| Shares issued value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 6,200,000
|
|
|
|
| Debt instrument periodic payment |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 31,630
|
|
|
|
|
|
|
$ 5,000
|
|
|
|
|
| Shares issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 1,530,651
|
$ 540,065
|
|
|
| Number of options |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2,650,000
|
1,250,000
|
|
|
| Number of options granted |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,400,000
|
1,250,000
|
|
|
| Exercise price |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 0.73
|
$ 1.10
|
|
|
| Share based compensation |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 624,463
|
$ 82,267
|
|
|
| Fair value of options outstanding |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
573,202
|
|
|
|
| Fair value of warrants |
$ 271,216
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 31,995
|
|
|
|
|
|
|
|
|
|
| Investor [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Fair value of warrants outstanding |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
852,480
|
$ 581,264
|
|
|
| Private Placement Warrant One [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Warrants issued |
|
|
|
|
|
|
|
|
|
|
|
150,000
|
|
|
|
|
|
|
100,000
|
|
|
|
|
|
|
|
|
|
|
|
|
| Sale of stock price |
|
|
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
|
|
|
|
|
| Consideration paid |
|
|
|
|
|
|
|
|
|
|
|
$ 150,000
|
|
|
|
|
|
|
$ 100,000
|
|
|
|
|
|
|
|
|
|
|
|
|
| Private Placement Warrant Two [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Warrants issued |
|
|
|
|
|
|
|
|
|
|
75,000
|
|
|
|
|
|
|
100,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Sale of stock price |
|
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Consideration paid |
|
|
|
|
|
|
|
|
|
|
$ 75,000
|
|
|
|
|
|
|
$ 100,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Private Placement Warrant Three [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Warrants issued |
|
|
|
|
|
|
|
|
|
24,500
|
|
|
|
|
|
|
|
25,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Sale of stock price |
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Consideration paid |
|
|
|
|
|
|
|
|
|
$ 24,500
|
|
|
|
|
|
|
|
$ 25,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Private Placement Warrant Four [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Warrants issued |
|
|
|
|
|
|
|
|
100,000
|
|
|
|
|
|
|
|
|
25,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Sale of stock price |
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Consideration paid |
|
|
|
|
|
|
|
|
$ 100,000
|
|
|
|
|
|
|
|
|
$ 25,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Private Placement Warrant Five [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Warrants issued |
|
|
|
|
|
|
|
100,000
|
|
|
|
|
|
|
|
|
|
75,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Sale of stock price |
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Consideration paid |
|
|
|
|
|
|
|
$ 100,000
|
|
|
|
|
|
|
|
|
|
$ 75,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Private Placement Warrant Six [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Warrants issued |
|
|
|
|
|
|
100,000
|
|
|
|
|
|
|
|
|
|
100,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Sale of stock price |
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Consideration paid |
|
|
|
|
|
|
$ 100,000
|
|
|
|
|
|
|
|
|
|
$ 100,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Private Placement Warrant Seven [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Warrants issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
25,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Sale of stock price |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Consideration paid |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 25,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Private Placement Warrant [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Warrants issued |
|
|
|
|
|
|
|
|
|
|
|
|
25,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Sale of stock price |
|
|
|
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Consideration paid |
|
|
|
|
|
|
|
|
|
|
|
|
$ 25,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| IPO [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,250,000
|
|
|
|
|
|
| Share price |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 4.00
|
|
|
|
|
|
| Common Stock [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 525
|
|
|
|
| Shares issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
5,250,000
|
|
|
|
| Number of shares sold |
4,000,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,250,000
|
|
|
|
|
|
|
|
|
|
|
| Share price |
$ 0.30
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 4.00
|
|
|
|
|
|
|
|
|
|
|
| Gross proceeds |
$ 1,200,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 5,000,000
|
|
|
|
|
|
|
|
|
|
|
| Shares issued in consideration |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,780,000
|
3,365,000
|
|
|
| Shares issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 178
|
$ 337
|
|
|
| Exercise price |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 1.10
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 0.32
|
|
| Vesting term |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
3 years
|
|
|
| Common Stock [Member] | 2022 Plan [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Number of options |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
150,000
|
|
|
|
|
|
|
|
|
|
|
|
|
1,250,000
|
1,250,000
|
|
| Common Stock [Member] | IPO [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Warrants issued |
280,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
87,500
|
|
|
|
|
|
|
|
|
|
| Exercise price |
$ 0.38
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 5.00
|
|
|
|
|
|
|
|
|
|
| Warrant [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Warrants outstanding |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,063,000
|
|
|
|
| Outstanding and vested warrants expected term |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
3 years 9 months 29 days
|
|
|
|
| Consulting Agreements [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 350,000
|
|
|
|
|
|
|
|
|
| Share price |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
|
| Consulting Agreement [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
|
|
$ 2,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued |
|
|
|
|
|
50,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Mrs.Hammer [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Annual salary |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 150,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Share based compensation arrangement by share-based payment award, description |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The Employment Agreement also required the Company to grant Mrs. Hammer
a sign-on bonus of (a) 75,000 restricted shares of common stock of the Company, vested in full upon issuance, and (b) options to purchase
an additional 150,000 shares of common stock of the Company, under the Company’s 2022 Equity Incentive Plan (the “Plan”),
with an exercise price of the greater of (i) $1.10 per share; and (ii) the closing sales price of the Company’s common stock on
the Nasdaq Capital Market on the date the Employment Agreement and the grant is approved by the Board (which date was May 1, 2023), and
which exercise price was $1.00 per share, with options to purchase 50,000 shares vesting every twelve months that the Employment Agreement
is in effect, subject to the terms of the Plan. The options are exercisable for a period of ten years and are documented by a separate
option agreement entered into by the Company and Mrs. Hammer.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Exercise price |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares vesting |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
50,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Chief Executive Officer [Member] | Common Stock [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Exercise price |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 1.10
|
|
|
| Chief Executive Officer [Member] | Common Stock [Member] | 2022 Plan [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Number of options granted |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
750,000
|
|
|
| Chief Operating Officer [Member] | Common Stock [Member] | 2022 Plan [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Number of options granted |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
500,000
|
|
|
| Restricted Stock [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Settlement description |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
the Company issued an aggregate of 350,000 shares of common stock to the consultants as
follows: (1) Sultan Haroon 150,000 shares of restricted common stock; (2) John Helfrich 25,000 shares of restricted common stock; (3)
Justin Baker 25,000 shares of restricted common stock; and (4) Maja Matthews 150,000 shares of restricted common stock. The shares issued
to Haroon and Matthews vest at the rate of 50,000 shares upon entry into the agreement, 50,000 shares upon the Company’s successful
launch of a new product category, and 50,000 shares upon the Company’s successful launch of a second and additional new product
category, in each case prior to the 18-month anniversary of the applicable agreement. The shares issued to Helfrich and Baker vest at
the rate of 10,000 shares upon entry into the agreement, 7,500 shares upon the Company’s successful launch of a new product category,
and 7,500 shares upon the Company’s successful launch of a second and additional new product category, in each case prior to the
18-month anniversary of the applicable agreement.
|
|
|
|
|
|
|
|
|
| Restricted Stock [Member] | Private Placement Warrant [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 180,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
180,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Share price |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Consideration paid |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 300,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Restricted Stock [Member] | Dr.Rudman [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 25,000
|
|
|
|
|
|
|
|
| Shares issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
25,000
|
|
|
|
|
|
|
|
| Share price |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
| Compensation expense |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 2,000
|
|
|
|
|
|
|
|
| Restricted Stock [Member] | Mr.Boon [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 25,000
|
|
|
|
|
|
|
|
| Shares issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
25,000
|
|
|
|
|
|
|
|
| Share price |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
| Restricted Stock [Member] | Mr Andrew [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 55,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Share price |
|
|
|
$ 0.58
|
|
|
|
|
|
|
|
|
|
|
$ 1.10
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Restricted Stock [Member] | Mr Andrew [Member] | 2022 Plan [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
50,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Restricted Stock [Member] | Major Dodge [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 22,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Share price |
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 1.10
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Restricted Stock [Member] | Major Dodge [Member] | 2022 Plan [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
20,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Restricted Stock [Member] | Dr Douglas Christianson [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
$ 29,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued |
|
|
|
50,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Restricted Stock [Member] | Marius Pharmaceuticals [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued |
|
100,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Dojo Labs [Member] | Restricted Stock [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 100,000
|
|
|
|
|
|
|
| Shares issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
50,000
|
|
|
|
|
|
|
| Share price |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
| Bethor Ltd [Member] | Restricted Stock [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 250,000
|
|
|
|
|
|
|
|
| Share price |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 1.00
|
|
|
|
|
|
|
|
| Street Group LLC [Member] | Restricted Stock [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 55,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
50,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Share price |
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 1.10
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Debt instrument periodic payment |
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 5,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Greentree Financial Group [Member] | Service Agreement [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 84,750
|
|
|
|
| Share price |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 1.13
|
|
|
|
| Greentree Financial Group [Member] | Restricted Stock [Member] | Service Agreement [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Settlement description |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(a) $20,000 on or before September 30, 2023; (b) $20,000 on or before
March 31, 2024.
|
|
|
|
| Shares issued in consideration |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
75,000
|
|
|
|
| Shares issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ 40,000
|
|
|
|
| Luca Consulting, LLC [Member] | Service Agreement [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Settlement description |
|
|
|
|
(a) $5,000 on the signing of the agreement; (b) $5,000 on the tenth of each month throughout the
remainder of the agreement.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Luca Consulting, LLC [Member] | Consulting Agreement [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
|
$ 126,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued |
|
|
|
|
200,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Share price |
|
|
|
|
$ 0.63
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued |
|
|
|
|
$ 15,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Jason Szkup [Member] | Restricted Stock [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
|
$ 17,400
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued |
|
|
|
30,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Share price |
|
|
|
$ 0.58
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued |
|
|
|
$ 10,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| PHX Global LLC [Member] | Restricted Stock [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
|
$ 94,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued |
|
|
200,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Share price |
|
|
$ 0.47
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Marius Pharmaceuticals [Member] | Restricted Stock [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accumulated Other Comprehensive Income (Loss) [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued value |
|
$ 58,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Share price |
|
$ 0.58
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| X |
- DefinitionSharebased compensation arrangement by sharebased payment award fair value assumption expected term.
| Name: |
MGRX_SharebasedCompensationArrangementBySharebasedPaymentAwardFairValueAssumptionExpectedTerm |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-4
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 5
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-5
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481674/830-30-50-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 17
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-17
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 20
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-20
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 20
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-20
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 20
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-20
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 20
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481694/830-30-45-20
| Name: |
us-gaap_AccumulatedOtherComprehensiveIncomeLossLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of expense for award under share-based payment arrangement. Excludes amount capitalized. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SAB Topic 14.F)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479830/718-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (h)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_AllocatedShareBasedCompensationExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionExercise price per share or per unit of warrants or rights outstanding. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-3
| Name: |
us-gaap_ClassOfWarrantOrRightExercisePriceOfWarrantsOrRights1 |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of securities into which the class of warrant or right may be converted. For example, but not limited to, 500,000 warrants may be converted into 1,000,000 shares. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-3
| Name: |
us-gaap_ClassOfWarrantOrRightNumberOfSecuritiesCalledByWarrantsOrRights |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of warrants or rights outstanding.
| Name: |
us-gaap_ClassOfWarrantOrRightOutstanding |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionDescription of common stock conversion feature. Includes, but is not limited to, conversion price; conversion right; timing of right; terms, event or change in circumstance causing contingency to be met or adjustment to conversion price or number of shares; manner of settlement upon conversion; and method of settlement. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Subparagraph (a)
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-6
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Subparagraph (c)
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-6
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Subparagraph (d)
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-6
| Name: |
us-gaap_CommonStockConversionFeatures |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFace amount or stated value per share of common stock. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockParOrStatedValuePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe maximum number of common shares permitted to be issued by an entity's charter and bylaws. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(16)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockSharesAuthorized |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionTotal number of common shares of an entity that have been sold or granted to shareholders (includes common shares that were issued, repurchased and remain in the treasury). These shares represent capital invested by the firm's shareholders and owners, and may be all or only a portion of the number of shares authorized. Shares issued include shares outstanding and shares held in the treasury. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockSharesIssued |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of shares of common stock outstanding. Common stock represent the ownership interest in a corporation. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.6-05(4))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-2
Reference 3: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(16)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 5: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(7))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockSharesOutstanding |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of the required periodic payments including both interest and principal payments. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.22)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 942
-SubTopic 470
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480848/942-470-50-3
| Name: |
us-gaap_DebtInstrumentPeriodicPayment |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe compensation expense recognized during the period pertaining to the deferred compensation arrangement. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 710
-SubTopic 10
-Name Accounting Standards Codification
-Section 30
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483043/710-10-30-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-1
| Name: |
us-gaap_DeferredCompensationArrangementWithIndividualCompensationExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of expense (income) related to adjustment to fair value of warrant liability. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Subparagraph (b)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 25
-Paragraph 13
-SubTopic 10
-Topic 480
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481766/480-10-25-13
| Name: |
us-gaap_FairValueAdjustmentOfWarrants |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionPayments of cash to employees, including wages and salaries, during the current period. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 17
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-17
| Name: |
us-gaap_PaymentsToEmployees |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionFace amount or stated value per share of preferred stock nonredeemable or redeemable solely at the option of the issuer. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 13
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-13
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_PreferredStockParOrStatedValuePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe maximum number of nonredeemable preferred shares (or preferred stock redeemable solely at the option of the issuer) permitted to be issued by an entity's charter and bylaws. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(16)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_PreferredStockSharesAuthorized |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe cash inflow associated with the amount received from entity's first offering of stock to the public. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 14
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-14
| Name: |
us-gaap_ProceedsFromIssuanceInitialPublicOffering |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionCash received on stock transaction after deduction of issuance costs.
| Name: |
us-gaap_SaleOfStockConsiderationReceivedOnTransaction |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of shares issued or sold by the subsidiary or equity method investee per stock transaction.
| Name: |
us-gaap_SaleOfStockNumberOfSharesIssuedInTransaction |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionPer share amount received by subsidiary or equity investee for each share of common stock issued or sold in the stock transaction.
| Name: |
us-gaap_SaleOfStockPricePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionPeriod over which grantee's right to exercise award under share-based payment arrangement is no longer contingent on satisfaction of service or performance condition, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days. Includes, but is not limited to, combination of market, performance or service condition. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardAwardVestingPeriod1 |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDescription of terms of share-based payment arrangement. Includes, but is not limited to, type of award or grantee and reason for issuance. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardDescription |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionGross number of share options (or share units) granted during the period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsGrantsInPeriodGross |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount by which the current fair value of the underlying stock exceeds the exercise price of options outstanding. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingIntrinsicValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of options outstanding, including both vested and non-vested options. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingNumber |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionWeighted average price at which grantees can acquire the shares reserved for issuance under the stock option plan. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingWeightedAverageExercisePrice |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionPrice of a single share of a number of saleable stocks of a company.
| Name: |
us-gaap_SharePrice |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionWeighted average grant-date fair value of non-vested options outstanding.
| Name: |
us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardOptionsNonvestedWeightedAverageGrantDateFairValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of options vested.
| Name: |
us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardOptionsVestedNumberOfShares |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionPer share or per unit amount of equity securities issued.
| Name: |
us-gaap_SharesIssuedPricePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of shares issued in lieu of cash for services contributed to the entity. Number of shares includes, but is not limited to, shares issued for services contributed by vendors and founders.
| Name: |
us-gaap_StockIssuedDuringPeriodSharesIssuedForServices |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of new stock issued during the period. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 505
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481004/946-505-50-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-03(i)(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479886/946-10-S99-3
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesNewIssues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionValue of stock issued in lieu of cash for services contributed to the entity. Value of the stock issued includes, but is not limited to, services contributed by vendors and founders.
| Name: |
us-gaap_StockIssuedDuringPeriodValueIssuedForServices |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionEquity impact of the value of new stock issued during the period. Includes shares issued in an initial public offering or a secondary public offering. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 11
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-11
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 205
-Name Accounting Standards Codification
-Section 45
-Paragraph 4
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480767/946-205-45-4
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 505
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481004/946-505-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 8: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodValueNewIssues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionValue of outstanding derivative securities that permit the holder the right to purchase securities (usually equity) from the issuer at a specified price.
| Name: |
us-gaap_WarrantsAndRightsOutstanding |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=MGRX_PrivatePlacementWarrantOneMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=MGRX_PrivatePlacementWarrantTwoMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=MGRX_PrivatePlacementWarrantThreeMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=MGRX_PrivatePlacementWarrantFourMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=MGRX_PrivatePlacementWarrantFiveMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=MGRX_PrivatePlacementWarrantSixMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=MGRX_PrivatePlacementWarrantSevenMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=MGRX_PrivatePlacementWarrantMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=us-gaap_IPOMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementEquityComponentsAxis=us-gaap_CommonStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_PlanNameAxis=MGRX_TwentyTwentyTwoPlanMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementEquityComponentsAxis=us-gaap_WarrantMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_TypeOfArrangementAxis=MGRX_ConsultingAgreementsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_TypeOfArrangementAxis=MGRX_ConsultingAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_TitleOfIndividualAxis=MGRX_HammerMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_TitleOfIndividualAxis=srt_ChiefExecutiveOfficerMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_TitleOfIndividualAxis=srt_ChiefOperatingOfficerMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_AwardTypeAxis=us-gaap_RestrictedStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_TitleOfIndividualAxis=MGRX_RudmanMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_TitleOfIndividualAxis=MGRX_BoonMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_TitleOfIndividualAxis=MGRX_MrAndrewMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_TitleOfIndividualAxis=MGRX_MajorDodgeMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_TitleOfIndividualAxis=MGRX_DrDouglasChristiansonMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_TitleOfIndividualAxis=MGRX_MariusPharmaceuticalsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=MGRX_DojoLabsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=MGRX_BethorLtdMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=MGRX_StreetGroupLLCMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=MGRX_GreentreeFinancialGroupMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_TypeOfArrangementAxis=MGRX_ServiceAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=MGRX_LucaConsultingLlcMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=MGRX_JasonSzkupMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=MGRX_PHXGlobalLLCMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=MGRX_MariusPharmaceuticalsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
GOING CONCERN (Details Narrative) - USD ($)
|
12 Months Ended |
Dec. 31, 2023 |
Dec. 31, 2022 |
| Organization, Consolidation and Presentation of Financial Statements [Abstract] |
|
|
| Net loss |
$ 9,212,417
|
$ 1,998,055
|
| Accumulated deficit |
$ 11,228,173
|
$ 2,015,756
|
| X |
- DefinitionThe portion of profit or loss for the period, net of income taxes, which is attributable to the parent. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-6
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-8
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 9
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-9
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-4
Reference 13: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-10
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483581/946-220-45-7
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(18))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(9))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(1)(d))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 20: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 29: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 30: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 31: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 60B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-60B
Reference 32: http://www.xbrl.org/2003/role/exampleRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 31
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-31
Reference 33: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 34: http://www.xbrl.org/2003/role/disclosureRef
-Topic 205
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483499/205-20-50-7
Reference 35: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 36: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1A
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1A
Reference 37: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1B
Reference 38: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(20))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 39: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(22))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
| Name: |
us-gaap_NetIncomeLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_OrganizationConsolidationAndPresentationOfFinancialStatementsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of accumulated undistributed earnings (deficit). Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 2
-Subparagraph (g)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480016/944-40-65-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 2
-Subparagraph (h)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480016/944-40-65-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480990/946-20-50-11
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(23)(a)(4))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(17))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 8: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(30)(a)(3))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_RetainedEarningsAccumulatedDeficit |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
v3.24.1.u1
| X |
- DefinitionLessee operating lease liability payments due after year two.
| Name: |
MGRX_LesseeOperatingLeaseLiabilityPaymentsDueAfterYearTwo |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_CommitmentsAndContingenciesDisclosureAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of lessee's undiscounted obligation for lease payment for operating lease. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-6
| Name: |
us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of lessee's undiscounted obligation for lease payment for operating lease to be paid in next fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-6
| Name: |
us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueNextTwelveMonths |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of lessee's undiscounted obligation for lease payment for operating lease to be paid in second fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-6
| Name: |
us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueYearTwo |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of lessee's undiscounted obligation for lease payments in excess of discounted obligation for lease payments for operating lease. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-6
| Name: |
us-gaap_LesseeOperatingLeaseLiabilityUndiscountedExcessAmount |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionPresent value of lessee's discounted obligation for lease payments from operating lease. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479041/842-20-45-1
| Name: |
us-gaap_OperatingLeaseLiability |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
v3.24.1.u1
COMMITMENTS AND CONTINGENCIES (Details Narrative)
|
|
12 Months Ended |
|
|
Sep. 28, 2022
USD ($)
ft²
|
Dec. 31, 2023
USD ($)
|
Dec. 31, 2022
USD ($)
|
| Collaborative Arrangement and Arrangement Other than Collaborative [Line Items] |
|
|
|
| Area of Land | ft² |
2,201
|
|
|
| Operating lease, description |
The Lease
Agreement has a term of thirty-eight (38) months and has a monthly base rent of $5,777.63, or $31.50 per square foot, the from months
3-18 and increases at the rate of $1 per square foot per annum until the end of the lease term
|
|
|
| Lease term |
38 months
|
|
|
| Base rent |
$ 5,777.63
|
|
|
| Percentage of proportionate rent |
2.45%
|
|
|
| Percentage of incremental borrowing rate |
8.00%
|
|
|
| Operating Lease, Right-of-Use Asset |
|
$ 119,262
|
$ 174,241
|
| Operating Lease, Liability |
|
128,679
|
|
| Operating Lease, Cost |
|
65,274
|
|
| Asset Impairment Charges |
|
$ 0
|
|
| Lease Agreement [Member] |
|
|
|
| Collaborative Arrangement and Arrangement Other than Collaborative [Line Items] |
|
|
|
| Base rent |
$ 16,942
|
|
|
| X |
- DefinitionPercentage of incremental borrowing rate.
| Name: |
MGRX_PercentageOfIncrementalBorrowingRate |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionPercentage of proportionate rent.
| Name: |
MGRX_PercentageOfProportionateRent |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of write-down of assets recognized in the income statement. Includes, but is not limited to, losses from tangible assets, intangible assets and goodwill. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Subparagraph (b)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 360
-SubTopic 10
-Section 45
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482130/360-10-45-4
| Name: |
us-gaap_AssetImpairmentCharges |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 808
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479402/808-10-50-1
| Name: |
us-gaap_CollaborativeArrangementsAndNoncollaborativeArrangementTransactionsLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDescription of lessee's operating lease. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-3
| Name: |
us-gaap_LesseeOperatingLeaseDescription |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of single lease cost, calculated by allocation of remaining cost of lease over remaining lease term. Includes, but is not limited to, single lease cost, after impairment of right-of-use asset, calculated by amortization of remaining right-of-use asset and accretion of lease liability. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 55
-Paragraph 53
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479589/842-20-55-53
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-4
| Name: |
us-gaap_OperatingLeaseCost |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionPresent value of lessee's discounted obligation for lease payments from operating lease. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479041/842-20-45-1
| Name: |
us-gaap_OperatingLeaseLiability |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of lessee's right to use underlying asset under operating lease. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479041/842-20-45-1
| Name: |
us-gaap_OperatingLeaseRightOfUseAsset |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionWeighted average remaining lease term for operating lease, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 55
-Paragraph 53
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479589/842-20-55-53
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (g)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-4
| Name: |
us-gaap_OperatingLeaseWeightedAverageRemainingLeaseTerm1 |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionCash payments to lessor's for use of assets under operating leases. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 25
-Subparagraph (g)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-25
| Name: |
us-gaap_PaymentsForRent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_TypeOfArrangementAxis=MGRX_LeaseAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
SCHEDULE OF DEFERRED TAX ASSET (Details) - USD ($)
|
Dec. 31, 2023 |
Dec. 31, 2022 |
| Income Tax Disclosure [Abstract] |
|
|
| Deferred Tax Asset |
$ 11,228,173
|
$ 2,015,756
|
| Valuation Allowance |
(11,228,173)
|
(2,015,756)
|
| Net Deferred Tax Asset |
|
|
| X |
- DefinitionAmount before allocation of valuation allowances of deferred tax asset attributable to deductible temporary differences and carryforwards. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-2
| Name: |
us-gaap_DeferredTaxAssetsGross |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount after allocation of valuation allowances of deferred tax asset attributable to deductible temporary differences and carryforwards. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-2
| Name: |
us-gaap_DeferredTaxAssetsNet |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of deferred tax assets for which it is more likely than not that a tax benefit will not be realized. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-2
| Name: |
us-gaap_DeferredTaxAssetsValuationAllowance |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
v3.24.1.u1
INCOME TAXES (Details Narrative) - USD ($)
|
|
12 Months Ended |
Dec. 22, 2017 |
Dec. 31, 2023 |
Dec. 31, 2022 |
| Significant Change in Unrecognized Tax Benefits is Reasonably Possible [Line Items] |
|
|
|
| Deferred tax asset net operating loss |
|
$ 9,212,417
|
|
| Decrease in deferred tax assets, valuation allowance |
|
$ 9,212,417
|
|
| US federal income corporate tax rates |
|
21.00%
|
21.00%
|
| Net operating loss carry forwards |
|
$ 11,228,173
|
|
| Maximum [Member] |
|
|
|
| Significant Change in Unrecognized Tax Benefits is Reasonably Possible [Line Items] |
|
|
|
| Statutory federal income tax rate, percentage |
35.00%
|
|
|
| Minimum [Member] |
|
|
|
| Significant Change in Unrecognized Tax Benefits is Reasonably Possible [Line Items] |
|
|
|
| Statutory federal income tax rate, percentage |
21.00%
|
|
|
| X |
- DefinitionDecrease in deferred tax assets valuation allowance.
| Name: |
MGRX_DecreaseInDeferredTaxAssetsValuationAllowance |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount before allocation of valuation allowances of deferred tax asset attributable to deductible operating loss carryforwards. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-6
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 740
-SubTopic 10
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-8
| Name: |
us-gaap_DeferredTaxAssetsOperatingLossCarryforwards |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of operating loss carryforward, before tax effects, available to reduce future taxable income under enacted tax laws. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 740
-SubTopic 10
-Section 50
-Paragraph 3
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-3
| Name: |
us-gaap_OperatingLossCarryforwards |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.
| Name: |
us-gaap_SignificantChangeInUnrecognizedTaxBenefitsIsReasonablyPossibleLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
srt_RangeAxis=srt_MaximumMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_RangeAxis=srt_MinimumMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
SUBSEQUENT EVENTS (Details Narrative) - USD ($)
|
|
|
|
|
|
|
|
|
12 Months Ended |
|
|
|
Mar. 25, 2024 |
Mar. 21, 2024 |
Jan. 18, 2024 |
Jan. 11, 2024 |
Jan. 10, 2024 |
Jan. 02, 2024 |
Oct. 10, 2023 |
Oct. 01, 2023 |
Dec. 31, 2023 |
Dec. 31, 2022 |
Apr. 01, 2024 |
Mar. 18, 2024 |
Jan. 22, 2024 |
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Proceeds from sales of common stock for cash |
|
|
|
|
|
|
|
|
$ 6,200,000
|
$ 2,000,000
|
|
|
|
| Number of stock issued granted |
|
|
|
|
|
|
|
|
1,400,000
|
1,250,000
|
|
|
|
| Number of stock issued exercise |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Common stock, par value |
|
|
|
|
|
|
|
|
$ 0.0001
|
$ 0.0001
|
|
|
|
| Common Stock [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance of common stock for cash, shares |
|
|
|
|
|
|
|
|
5,250,000
|
|
|
|
|
| Consulting Agreement [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance of common stock for cash, shares |
|
|
|
|
|
|
|
50,000
|
|
|
|
|
|
| Consulting Agreement [Member] | Luca Consulting, LLC [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued price per share |
|
|
|
|
|
|
$ 0.63
|
|
|
|
|
|
|
| Issuance of common stock for cash, shares |
|
|
|
|
|
|
200,000
|
|
|
|
|
|
|
| Subsequent Event [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Common stock, par value |
$ 0.0001
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Member] | 2022 Plan [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Number of stock available for awards description |
The First Amendment increased the number of shares of common
stock available for awards under the Incentive Plan, such that currently, subject to adjustment in connection with the payment of a stock
dividend, a stock split or subdivision or combination of the shares of common stock, or a reorganization or reclassification of the Company’s
common stock, the aggregate number of shares of common stock which may be issued pursuant to awards under the 2022 Plan is currently
the sum of (i) 10,000,000, and (ii) an automatic increase on April 1st of each year for a period of nine years commencing on April 1,
2024 and ending on (and including) April 1, 2032, in an amount equal to the lesser of (x) ten percent (10%) of the total shares of common
stock of the Company outstanding on the last day of the immediately preceding fiscal year; and (y) 2,000,000 shares of common stock;
provided, however, that the Board may act prior to April 1st of a given year to provide that the increase for such year will be a lesser
number of shares of common stock. This is also known as an “evergreen” provision. Notwithstanding the foregoing, no more
than a total of 26,000,000 shares of common stock (or awards) may be issued or granted under the 2022 Plan in aggregate, and no more
than 26,000,000 shares of common stock may be issued pursuant to the exercise of Incentive Stock Options
|
|
|
|
|
|
|
|
|
|
|
|
|
| Number of stock issued |
10,000,000
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Member] | Common Stock [Member] | 2022 Plan [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Number of stock issued |
2,000,000
|
|
|
|
|
|
|
|
|
|
|
|
|
| Number of stock issued granted |
26,000,000
|
|
|
|
|
|
|
|
|
|
|
|
|
| Number of stock issued exercise |
26,000,000
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Member] | Underwriters [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance of common stock for cash, shares |
|
|
600,000
|
|
|
|
|
|
|
|
|
|
|
| Proceeds from sales of common stock for cash |
|
|
$ 160,000
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Member] | Underwriters [Member] | Over-Allotment Option [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance of common stock for cash, shares |
|
|
4,600,000
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Member] | Ronin Equity Partners [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Borrow amount |
|
|
|
|
|
|
|
|
|
|
$ 37,500
|
|
|
| Subsequent Event [Member] | Cohen Enterprises Inc [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Borrow amount |
|
|
|
|
|
|
|
|
|
|
|
$ 50,000
|
|
| Subsequent Event [Member] | Consulting Agreement [Member] | G&P General Consulting [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Number of restricted common shares issued |
|
|
|
|
|
250,000
|
|
|
|
|
|
|
|
| Number of additional restricted common shares issued |
|
|
|
|
|
500,000
|
|
|
|
|
|
|
|
| Shares issued price per share |
|
|
|
|
|
$ 0.28
|
|
|
|
|
|
|
|
| Number of restricted common shares issued, value |
|
|
|
|
|
$ 70,000
|
|
|
|
|
|
|
|
| Subsequent Event [Member] | Consulting Agreement [Member] | Luca Consulting, LLC [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Number of restricted common shares issued |
|
500,000
|
|
|
200,000
|
|
|
|
|
|
|
|
|
| Shares issued price per share |
|
$ 0.1975
|
|
|
$ 0.28
|
|
|
|
|
|
|
|
|
| Number of restricted common shares issued, value |
|
$ 98,750
|
|
|
$ 56,000
|
|
|
|
|
|
|
|
|
| Agreement payable in cash |
|
$ 5,000
|
|
|
$ 15,000
|
|
|
|
|
|
|
|
|
| Consulting agreement description |
|
|
|
|
(a) $5,000 on the signing of the agreement; (b) $5,000 on the tenth of each month throughout the remainder
of the agreement. The Service Agreement includes customary indemnification obligations requiring the Company to indemnify Luca and its
affiliates with regard to certain matters.
|
|
|
|
|
|
|
|
|
| Subsequent Event [Member] | Consulting Agreement [Member] | First Level Capital [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Number of restricted common shares issued |
|
|
|
250,000
|
|
|
|
|
|
|
|
|
|
| Number of additional restricted common shares issued |
|
|
|
250,000
|
|
|
|
|
|
|
|
|
|
| Shares issued price per share |
|
|
|
$ 0.28
|
|
|
|
|
|
|
|
|
|
| Number of restricted common shares issued, value |
|
|
|
$ 70,000
|
|
|
|
|
|
|
|
|
|
| Agreement payable in cash |
|
|
|
$ 60,000
|
|
|
|
|
|
|
|
|
|
| Consulting agreement description |
|
|
|
(a) $60,000 on the signing of the agreement; (b) $60,000 on the approval by the Company. The
Service Agreement includes customary indemnification obligations requiring the Company to indemnify First Level and its affiliates with
regard to certain matters.
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Member] | Consulting Agreement [Member] | Zvonimir Moric Consulting [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Number of restricted common shares issued |
|
150,000
|
|
|
|
|
|
|
|
|
|
|
|
| Shares issued price per share |
|
$ 0.1975
|
|
|
|
|
|
|
|
|
|
|
|
| Number of restricted common shares issued, value |
|
$ 29,625
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Member] | Underwriting Agreement [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Exercise price per share |
|
|
|
|
|
|
|
|
|
|
|
|
$ 0.375
|
| Subsequent Event [Member] | Underwriting Agreement [Member] | Warrant [Member] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Subsequent Event [Line Items] |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Number of warrants to purchase of common stock |
|
|
|
|
|
|
|
|
|
|
|
|
42,000
|
| X |
- DefinitionAdditional stock issued during period shares restricted stock award gross.
| Name: |
MGRX_AdditionalStockIssuedDuringPeriodSharesRestrictedStockAwardGross |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAgreement payable in cash.
| Name: |
MGRX_AgreementPayableInCash |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionConsulting agreement description.
| Name: |
MGRX_ConsultingAgreementDescription |
| Namespace Prefix: |
MGRX_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionExercise price per share or per unit of warrants or rights outstanding. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-3
| Name: |
us-gaap_ClassOfWarrantOrRightExercisePriceOfWarrantsOrRights1 |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of securities into which the class of warrant or right may be converted. For example, but not limited to, 500,000 warrants may be converted into 1,000,000 shares. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-3
| Name: |
us-gaap_ClassOfWarrantOrRightNumberOfSecuritiesCalledByWarrantsOrRights |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionFace amount or stated value per share of common stock. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockParOrStatedValuePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionAmount, after deduction of unamortized premium (discount) and debt issuance cost, of long-term debt. Excludes lease obligation. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(22))
-SubTopic 10
-Topic 210
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 55
-Paragraph 69B
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481568/470-20-55-69B
Reference 3: http://www.xbrl.org/2003/role/exampleRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 55
-Paragraph 69C
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481568/470-20-55-69C
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1D
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-1D
Reference 5: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(16)(a)(2))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-03(16))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479853/942-210-S99-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (b)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-4
| Name: |
us-gaap_LongTermDebt |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe cash inflow from the additional capital contribution to the entity. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 14
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-14
| Name: |
us-gaap_ProceedsFromIssuanceOfCommonStock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionDescription of terms of share-based payment arrangement. Includes, but is not limited to, type of award or grantee and reason for issuance. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardDescription |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionGross number of share options (or share units) granted during the period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsGrantsInPeriodGross |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of shares of stock issued as of the balance sheet date, including shares that had been issued and were previously outstanding but which are now held in the treasury. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
| Name: |
us-gaap_SharesIssued |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionPer share or per unit amount of equity securities issued.
| Name: |
us-gaap_SharesIssuedPricePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of new stock issued during the period. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 505
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481004/946-505-50-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-03(i)(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479886/946-10-S99-3
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesNewIssues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTotal number of shares issued during the period, including shares forfeited, as a result of Restricted Stock Awards. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesRestrictedStockAwardGross |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of share options (or share units) exercised during the current period. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 4: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesStockOptionsExercised |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAggregate value of stock related to Restricted Stock Awards issued during the period. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 4: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodValueRestrictedStockAwardGross |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionDetail information of subsequent event by type. User is expected to use existing line items from elsewhere in the taxonomy as the primary line items for this disclosure, which is further associated with dimension and member elements pertaining to a subsequent event. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481674/830-30-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 855
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483399/855-10-50-2
| Name: |
us-gaap_SubsequentEventLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_StatementEquityComponentsAxis=us-gaap_CommonStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_TypeOfArrangementAxis=MGRX_ConsultingAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=MGRX_LucaConsultingLlcMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsequentEventTypeAxis=us-gaap_SubsequentEventMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_PlanNameAxis=MGRX_TwoThouandTwentyTwoPlanMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_TitleOfIndividualAxis=MGRX_UnderwritersMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=us-gaap_OverAllotmentOptionMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=MGRX_RoninEquityPartnersMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=MGRX_CohenEnterprisesIncMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=MGRX_GAndPGeneralConsultingMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=MGRX_FirstLevelCapitalMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=MGRX_ZvonimirMoricConsultingMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_TypeOfArrangementAxis=MGRX_UnderwritingAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementEquityComponentsAxis=us-gaap_WarrantMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
Mangoceuticals (NASDAQ:MGRX)
Historical Stock Chart
From Aug 2024 to Sep 2024
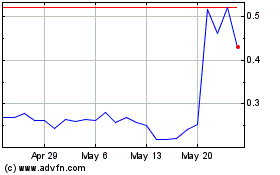
Mangoceuticals (NASDAQ:MGRX)
Historical Stock Chart
From Sep 2023 to Sep 2024