Avid Bioservices to Exhibit at 2017 BioProcess International Conference & Exhibition
September 19 2017 - 7:05AM

Avid Bioservices, Inc., a wholly owned subsidiary of Peregrine
Pharmaceuticals, Inc. (NASDAQ:PPHM) (NASDAQ:PPHMP), today announced
that the company will exhibit at the upcoming 2017 BioProcess
International Conference & Exhibition, being held September
25-28, 2017 in Boston, MA. Avid will host a corporate booth
(#918) in the conference’s exhibit hall, where it will showcase the
company’s comprehensive range of process development, analytical
development, testing and characterization services and high
quality cGMP clinical and commercial biologics manufacturing of
monoclonal antibodies and recombinant proteins. Avid
representatives attending the conference will include newly
appointed president Roger Lias, Ph.D., and Steven King, who is
stepping down from his role as president of Avid, but remaining as
president and chief executive officer of Peregrine.
At BioProcess International, the Avid team will
be able to discuss the company’s innovative processes for
generating a broad range of biopharmaceutical product types
including monoclonal antibodies, highly-glycosylated recombinant
proteins and enzymes, among others. As part of its exhibit,
the company will provide a virtual tour of the company’s 42,000
square foot state-of-the-art Myford manufacturing facility, which
is designed for late-stage clinical and commercial biologics
manufacturing. As previously announced, the company will soon be
ready to offer new larger scale capacity from the two recently
installed 2,000-liter, single-use bioreactors at the Myford
facility.
The Myford facility is designed with
cutting-edge, single-use equipment to accommodate a fully
disposable biomanufacturing process. A wide range of
innovative features are incorporated into this new facility
including monolithic modular clean rooms, dedicated support
utilities for each key processing area, and the industry’s most
advanced single-use production systems and flexible
solutions. Uni-directional process flows separate personnel
and materials and provide assurance that the design meets the most
stringent regulatory requirements for commercial biologics API
manufacturing.
For more information on the 2017 BioProcess
International Conference & Exhibition, please visit:
https://lifesciences.knect365.com/bioprocessinternational/.
Avid Bioservices was established as Peregrine’s
internal biologics manufacturing and development group, and began
formal operations in January 2002. The company has grown from
an internal support operation to a full service CDMO that
manufactures bulk drug substance for products that are approved and
marketed in over 18 countries by leading biopharma companies,
including the US and EU. Avid was recently recognized as a leading
CDMO by Life Science Leader as a recipient of multiple 2017
Contract Manufacturing Leadership Awards for Quality, Reliability,
Capabilities, Expertise and Compatibility. The company has an
outstanding regulatory inspection history, including 12 years of
commercial manufacturing and state-of-the-art cGMP manufacturing
facilities.
About Avid Bioservices, Inc.Avid Bioservices, a
wholly owned subsidiary of Peregrine Pharmaceuticals, provides a
comprehensive range of process development, high quality cGMP
clinical and commercial manufacturing services for the
biotechnology and biopharmaceutical industries. With over 20 years
of experience producing monoclonal antibodies and recombinant
proteins in batch, fed-batch and perfusion modes, including 12
years of commercial manufacturing for products sold in 18
countries, Avid's services include cGMP clinical and commercial
product manufacturing, purification, bulk packaging, lot release
and stability testing, regulatory strategy, submission and support.
The company also provides a variety of process development
activities, including cell line development and optimization, cell
culture and feed optimization, analytical methods development,
qualification/validation, process and product characterization. For
more information about Avid, please visit www.avidbio.com.
About Peregrine Pharmaceuticals,
Inc.Peregrine Pharmaceuticals, Inc. is a biopharmaceutical
company committed to improving the lives of patients by delivering
high quality pharmaceutical products through its contract
development and manufacturing organization (CDMO) services and
through advancing and licensing its investigational immunotherapy
and related products. Peregrine's in-house CDMO services,
including cGMP manufacturing and development capabilities, are
provided through its wholly-owned subsidiary Avid Bioservices, Inc.
(www.avidbio.com), which provides development and biomanufacturing
services for both Peregrine and third-party customers. The
company is also working to evaluate its lead immunotherapy
candidate, bavituximab, in combination with immune stimulating
therapies for the treatment of various cancers, and developing its
proprietary exosome technology for the detection and monitoring of
cancer. For more information, please visit
www.peregrineinc.com.
Contacts:Kelly Pisarev
Lord
Avid Bioservices, Inc. (800) 987-8256
Stephanie Diaz (Investors)Vida Strategic Partners
415-675-7401
sdiaz@vidasp.com
Tim Brons
(Media)
Vida Strategic
Partners 415-675-7402
tbrons@vidasp.com
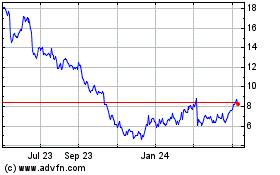
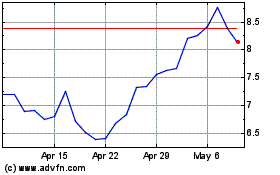
Avid Bioservices (NASDAQ:CDMO)
Historical Stock Chart
From Mar 2024 to Apr 2024
Avid Bioservices (NASDAQ:CDMO)
Historical Stock Chart
From Apr 2023 to Apr 2024