Intercept Drug Receives Unfavorable Votes From FDA Advisory Panel
May 19 2023 - 6:41PM
Dow Jones News
By Josh Beckerman
An Intercept Pharmaceuticals Inc. drug received two unfavorable
votes from a Food and Drug Administration advisory committee.
Intercept shares were halted all day.
The gastrointestinal drugs committee reviewed obeticholic acid
as a treatment for pre-cirrhotic fibrosis due to nonalcoholic
steatohepatitis.
Intercept said it was disappointed with the outcome. "We
continue to disagree with the FDA on certain characterizations of
OCA's efficacy and safety in pre-cirrhotic fibrosis due to NASH and
the role of non-invasive tests," the Morristown, N.J.-based company
said.
On the question "given the available efficacy and safety data,
do the benefits of OCA 25 mg outweigh the risks in NASH patients
with stage 2 or 3 fibrosis?" there were 12 no votes, two yes votes
and two abstentions, the company said.
Fifteen of 16 advisers voted to defer approval until clinical
outcome data are submitted and reviewed, at which time the
traditional approval pathway could be considered, Intercept
said.
The FDA isn't required to follow the advice of its advisory
panels but generally does so.
Intercept recently received the FDA's orphan drug designation
for a combination of OCA and bezafibrate to treat a rare bile-duct
disease.
Write to Josh Beckerman at josh.beckerman@wsj.com
(END) Dow Jones Newswires
May 19, 2023 19:26 ET (23:26 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
Intercept Pharmaceuticals (NASDAQ:ICPT)
Historical Stock Chart
From May 2024 to Jun 2024
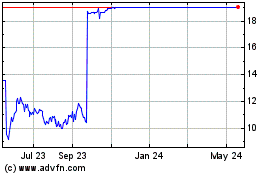
Intercept Pharmaceuticals (NASDAQ:ICPT)
Historical Stock Chart
From Jun 2023 to Jun 2024