DA-1726 Also Exhibited Superior Glucose
Lowering Compared to Survodutide
Lipid-Lowering Effect of DA-1726 Shown to be
Superior Compared to Tirzepatide
Data Presented at the ADA 84th
Scientific Sessions
CAMBRIDGE, Mass., June 22,
2024 /PRNewswire/ -- NeuroBo Pharmaceuticals,
Inc. (Nasdaq: NRBO), a clinical-stage biotechnology
company focused on the transformation of cardiometabolic diseases,
today announced pre-clinical data which indicates that DA-1726, a
novel, dual oxyntomodulin (OXM) analog agonist that functions as a
glucagon-like peptide-1 receptor (GLP1R) and glucagon receptor
(GCGR), demonstrated superiority in weight
loss, retention of lean body mass, and lipid-lowering effects
compared to survodutide, in pre-clinical models. Tae-Hyoung Kim, Lead research scientist, Dong-A
ST Research Center, will present the data today, in a poster at the
American Diabetes Association (ADA) 84th Scientific
Sessions, taking place June 21-24, in
Orlando, Florida.
"The data being presented at the ADA further differentiates
DA-1726 from obesity drugs in the same class, potentially due to
its GLP-1 and glucagon receptor activity ratio," stated
Hyung Heon Kim, President and Chief
Executive Officer of NeuroBo. "DA-1726, in obese mouse models,
significantly lowered cholesterol levels and induced
superior weight loss, compared to survodutide, a drug with the
same mechanism of action, while also exhibiting superior glucose
lowering. Most notably, DA-1726 demonstrated superior weight loss
and retention of relative lean body mass preservation compared to
survodutide. Further, as previously disclosed pre-clinical
studies showed, DA-1726 resulted in similar weight reduction while
consuming more food compared to tirzepatide and this
additional data, being presented at the ADA, in a
hypercholesterolemia rat model, confirmed that DA-1726 is more
effective than tirzepatide in suppressing cholesterol levels, due
to its glucagon action, alongside its GLP-1 effect, while also
inhibiting weight gain. We believe these distinguishing factors can
potentially position DA-1726 as a best-in-class obesity drug with
superior efficacy and better tolerability profile. The Phase 1
trial of DA-1726 is progressing well, and we expect to both dose
the first patient in the multiple ascending dose (MAD) Part 2 and
read-out top-line data from the single ascending dose (SAD) Part 1
in the third quarter of this year, with top-line data from the MAD
Part 2 expected in the first quarter of 2025."
- Abstract Title: DA-1726, a GLP1R/GCGR Dual Agonist, A
Promising Approach in Obesity Treatment and Lipid
Management
- Presenter Name: Tae-Hyoung
Kim, Lead research scientist, Dong-A ST Research Center
- Authors: Tae-Hyoung Kim,
Il-Hun Jung, Su Jin Lee,
Hyung Heon Kim, Mi-Kyung Kim, Yuna
Chae
- Abstract Number: 2024-LB-5728
- Poster Presentation Number: 2058-LB
- Session: 23-A Obesity-Animal
- Session Date: Saturday, June 22,
2024
- Session Time: 12:30 pm –
1:30 pm ET
DA-1726 is currently in a Phase 1 clinical trial. This study
focuses on the pharmacological effects of this novel oxyntomodulin
analogue, which has been effective at improving lipid profiles and
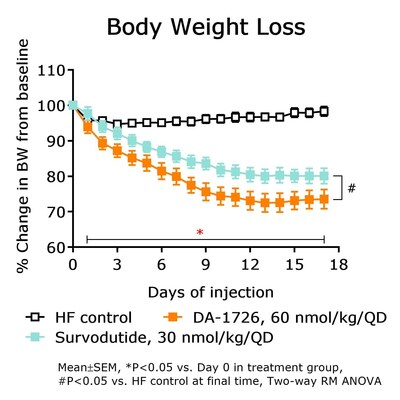
reducing weight in rodent models. In an obese mouse model, DA-1726
showed superior weight loss compared to survodutide (-24.7%,
-18.2%; P<0.05 vs. control) while demonstrating superior body
fat mass reduction and relative lean body mass preservation versus
survodutide (body fat change -31.4%, -15.1% vs. -8.7% control).
DA-1726 also significantly lowered T-CHO (-67.7%, -49.6%; P<0.05
vs. control) and TG (-49.5%, -41.2%; P<0.05 vs. control) while
showing superior glucose lowering compared to survodutide (-54.7%,
-30.4% vs. control; P<0.05). Interestingly, despite the same
mechanism of action, DA-1726-treated mice showed superior weight
loss, fat mass reduction and glucose lowering efficacy. This effect
might stem from DA-1726's GLP-1 and glucagon receptor activity
ratio. DA-1726's glucagon action could further enhance energy
expenditure (EE), and it is believed to have significantly
increased the expression of EE-related genes in brown adipose
tissue.
![DA-1726 [W0], DA-1726 [W2], Survodutide [W0], Survodutide [W2] DA-1726 [W0], DA-1726 [W2], Survodutide [W0], Survodutide [W2]](https://mma.prnewswire.com/media/2445098/pie_chart.jpg)
In a prior study, DA-1726 showed a difference in improving lipid
levels, despite similar weight loss to tirzepatide. Therefore, the
direct lipid-regulating effect of DA-1726 was assessed in a
hypercholesterolemia rat model compared with tirzepatide. As a
result, DA-1726 was more effective than tirzepatide in suppressing
the elevation of T-CHO (-33.5%, -25.5% vs. control; P<0.05) and
LDL-C (-53.2%, -41.5% vs. control; P<0.05) despite the two
groups of rats consuming the same amount of food. This
differentiated impact is thought to arise from DA-1726's glucagon
action, alongside its GLP-1 effect. The study evaluated whether
these differential effects could also be distinguished from drugs
of the same class.
In summary, it was confirmed that DA-1726 has differentiating
characteristics in the competition of obesity treatment drugs with
similar or identical mechanisms in its effect on improving
cholesterol metabolism through glucagon action.
After the presentations, the poster will be accessible within
the "Posters" section of NeuroBo's website at:
https://www.neurobopharma.com/posters.
About DA-1726
DA-1726 is a novel oxyntomodulin (OXM)
analogue functioning as a GLP1R/GCGR dual agonist for the treatment
of obesity and Metabolic Dysfunction-Associated Steatohepatitis
(MASH) that is to be administered once weekly subcutaneously.
DA-1726 acts as a dual agonist of GLP-1 receptors (GLP1R) and
glucagon receptors (GCGR), leading to weight loss through reduced
appetite and increased energy expenditure. DA-1726 has a well
understood mechanism and, in pre-clinical mice models, resulted in
improved weight loss compared to semaglutide and cotadutide
(another OXM analogue). Additionally, in pre-clinical mouse models,
DA-1726 elicited similar weight reduction, while consuming more
food, compared to tirzepatide and survodutide, while also
preserving lean body mass and demonstrating lipid-lowering effects
compared to survodutide.
About NeuroBo Pharmaceuticals
NeuroBo Pharmaceuticals,
Inc. is a clinical-stage biotechnology company focused on
transforming cardiometabolic diseases. The company is currently
developing DA-1241 for the treatment of Metabolic
Dysfunction-Associated Steatohepatitis (MASH) and is developing
DA-1726 for the treatment of obesity. DA-1241 is a novel
G-protein-coupled receptor 119 (GPR119) agonist that promotes the
release of key gut peptides GLP-1, GIP, and PYY. In pre-clinical
studies, DA-1241 demonstrated a positive effect on liver
inflammation, lipid metabolism, weight loss, and glucose
metabolism, reducing hepatic steatosis, hepatic inflammation, and
liver fibrosis, while also improving glucose control. DA-1726 is a
novel oxyntomodulin (OXM) analogue that functions as a
glucagon-like peptide-1 receptor (GLP1R) and glucagon receptor
(GCGR) dual agonist. OXM is a naturally-occurring gut hormone that
activates GLP1R and GCGR, thereby decreasing food intake while
increasing energy expenditure, thus potentially resulting in
superior body weight loss compared to selective GLP1R agonists.
For more information, please visit www.neurobopharma.com.
Forward Looking Statements
Certain statements in this
press release may be considered forward-looking statements within
the meaning of the Private Securities Litigation Reform Act of
1995. Words such as "believes", "expects", "anticipates", "may",
"will", "should", "seeks", "approximately", "intends", "projects",
"plans", "estimates" or the negative of these words or other
comparable terminology (as well as other words or expressions
referencing future events, conditions or circumstances) are
intended to identify forward-looking statements. Forward-looking
statements are predictions, projections and other statements about
future events that are based on current expectations and
assumptions and, as a result, are subject to risks and
uncertainties. Many factors could cause actual future events to
differ materially from the forward-looking statements in this press
release, including, without limitation, those risks associated with
NeuroBo's ability to execute on its commercial strategy; the
timeline for regulatory submissions; the ability to obtain
regulatory approval through the development steps of NeuroBo's
current and future product candidates, the ability to realize the
benefits of the license agreement with Dong-A ST Co. Ltd.,
including the impact on future financial and operating results of
NeuroBo; the cooperation of NeuroBo's contract manufacturers,
clinical study partners and others involved in the development of
NeuroBo's current and future product candidates; potential negative
interactions between NeuroBo's product candidates and any other
products with which they are combined for treatment; NeuroBo's
ability to initiate and complete clinical trials on a timely basis;
NeuroBo's ability to recruit subjects for its clinical trials;
whether NeuroBo receives results from NeuroBo's clinical trials
that are consistent with the results of pre-clinical and previous
clinical trials; impact of costs related to the license agreement,
known and unknown, including costs of any litigation or regulatory
actions relating to the license agreement; the effects of changes
in applicable laws or regulations; the effects of changes to
NeuroBo's stock price on the terms of the license agreement and any
future fundraising; and other risks and uncertainties described in
NeuroBo's filings with the Securities Exchange Commission,
including NeuroBo's most recent Annual Report on Form 10-K.
Forward-looking statements speak only as of the date when made.
NeuroBo does not assume any obligation to publicly update or revise
any forward-looking statements, whether as a result of new
information, future events or otherwise, except as required by
law.
Contacts:
NeuroBo Pharmaceuticals
Marshall H. Woodworth
Chief Financial Officer
+1-857-299-1033
marshall.woodworth@neurobopharma.com
Rx Communications Group
Michael Miller
+1-917-633-6086
mmiller@rxir.com
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/neurobo-pharmaceuticals-da-1726-demonstrated-superiority-in-weight-loss-retention-of-lean-body-mass-and-lipid-lowering-effects-compared-to-survodutide-in-pre-clinical-models-302179186.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/neurobo-pharmaceuticals-da-1726-demonstrated-superiority-in-weight-loss-retention-of-lean-body-mass-and-lipid-lowering-effects-compared-to-survodutide-in-pre-clinical-models-302179186.html
SOURCE NeuroBo Pharmaceuticals, Inc.