First and only contact force pulsed field ablation (PFA) system
designed to transform treatment for millions with life-threatening
ventricular arrhythmias.
Preliminary data of VCAS study will be presented at HRS 2024 in
Boston, Mass.
CARDIFF-BY-THE-SEA, Calif.,
May 9, 2024 /PRNewswire/ -- Field
Medical,™ Inc., a pioneer in pulsed field cardiac catheter ablation
technology, today announced the initiation of its first-in-human
study for the FieldForce™ Ablation System at Na Homolce
Hospital in Prague, Czech
Republic. The Ventricular Catheter Ablation Study, VCAS,
will enroll up to 60 patients at up to 5 sites world-wide. The VCAS
investigational study is a critical step towards demonstrating
safety and performance of the FieldForce™ Ablation System,
developed specifically to address the limitations of existing
pulsed field ablation (PFA) and radiofrequency (RF) ablation
systems, including a more time efficient treatment protocol for the
treatment of ventricular arrhythmias. Preliminary results will be
presented on May 16, 2024, at the
Heart Rhythm Scientific Sessions in Boston, Mass.
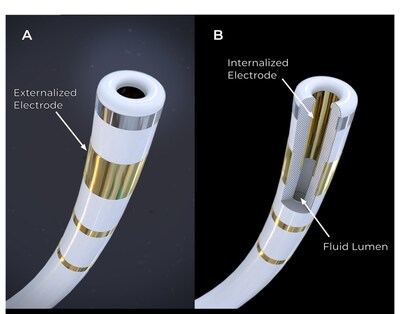
"The FieldForce Ablation System, as the first contact force PFA
system for ventricular ablation, is demonstrating significant
promise early in our trials," said Vivek
Reddy, MD, Director of Electrophysiology for the Mount Sinai
Health System. "This is my first experience with a technology
capable of achieving rapid complete transmural left ventricular
(LV) ablation, offering highly targeted treatment options and an
efficient workflow. Although these results are quite preliminary,
the potential of this system to revolutionize catheter ablation of
complex ventricular arrhythmias is profound, potentially surpassing
the current standard of care. The scientific community will benefit
from the comprehensive results from the completed study."
PFA Adoption and Transition from Atrial to Ventricular
Arrhythmias
PFA has revolutionized the treatment of atrial fibrillation (AF)
over the past decade, thanks to pioneering work by Field Medical
CEO Steven Mickelsen, MD, founder of
Farapulse which was subsequently acquired by Boston Scientific. His
development of PFA technology marked a significant shift in cardiac
electrophysiology, enhancing safety and reducing procedure times.
This innovation set a new standard in the industry, leading to
widespread adoption and the transformation of ablation practices.
With PFA's proven success in atrial settings, Field Medical is now
poised to extend these benefits to ventricular arrhythmias,
signaling a pivotal evolution in the management of complex
ventricular cardiac conditions.
Ventricular Arrhythmia Burden
It is estimated that
ventricular arrhythmia impact over 6 million patients in the US and
Europe. Ventricular arrhythmias,
including ventricular tachycardia (VT) and premature ventricular
contractions (PVCs), and pose a substantial risk, often leading to
sudden cardiac death if not adequately managed. Current ventricular
catheter ablation methods involve lengthy procedures and
significant risks associated with thermal ablation, like
conventional RF ablation. Field Medical's PFA technology aims to
address these challenges by providing the first and only contact
force PFA catheter optimized to work in the ventricle. This novel
technology may enable predictable, time efficient, and safe
treatment option. FieldBending™ takes advantage of non-intuitive
physics to deliver intense, yet brief, electric fields designed to
safely reduce procedure times dramatically.
"Our vision at Field Medical is to transform VT ablation into a
widely available, one-hour outpatient procedure with improved
safety outcomes," said Steven
Mickelsen MD, CEO of Field Medical. "With the promising
early data for our VCAS study, in much the same way that PFA
transformed AF, the FieldForce Ablation System is optimized to
transform how ventricular arrhythmias are treated, offering hope to
millions of patients worldwide. With the strong investor support,
renown physician engagement and the groundbreaking potential of our
technology, we are set to redefine the standards of cardiac
ablation once again."
About VCAS Study
Field Medical's VCAS Study is a
pivotal segment of a prospective safety and feasibility study,
assessing the innovative FieldForce Ablation System in patients
with ventricular arrhythmias. The VCAS-I group focuses on
individuals suffering from ventricular tachycardia (VT), while the
VCAS-II group targets those experiencing frequent premature
ventricular complexes (PVCs). This study is instrumental in
evaluating the performance of Pulsed Field Ablation (PFA), thereby
potentially reducing procedural time compared to traditional
Radiofrequency (RF) ablation. The trial aims to demonstrate that
PFA can safely treat and improve overall outcomes for patients with
challenging ventricular arrhythmias. For study information, visit
clinicaltrials.gov NCT: NCT06203262.
About Field Medical,™ Inc.
Founded in
2022, Field Medical is at the forefront of developing
second-generation pulsed field ablation (PFA) technologies,
designed to meet the technical and safety challenges of modern
cardiac ablation procedures. Under the leadership of Dr.
Steven Mickelsen, CEO, a globally
recognized expert in pulsed electric field technology, the company
has pioneered the proprietary FieldBending™ technology. This
innovation was developed to concentrate therapeutic effects and
reduce unwanted far-field effects, with the potential to improve
patient outcomes while simplifying the clinical experience for
physicians. Field Medical focuses on expanding the applications of
PFA beyond atrial fibrillation (AF) to address complex ventricular
arrhythmias—a condition affecting over 6 million people in
North America and Europe alone, often leading to sudden cardiac
death, heart failure and significant clinical symptoms if not
managed effectively. The company's comprehensive product portfolio,
including the FieldForce™ Catheter, FieldForce™ Ablation System,
and the FieldFlex™ Sheath, is specifically optimized to work in
complex ventricular environments. For more information,
visit www.fieldmedicalinc.com and follow us on LinkedIn
and X.
The FieldForce™ PFA Ablation System is an investigational device
and is limited by Federal (or United
States) Law to investigational use.
Photo -
https://mma.prnewswire.com/media/2408458/FieldForce_Catheter2.jpg
Logo -
https://mma.prnewswire.com/media/2408456/Field_Medical_Block_WhiteOnBlack_Logo.jpg
 View original
content:https://www.prnewswire.co.uk/news-releases/field-medical-announces-first-in-human-trials-for-groundbreaking-fieldforce-ventricular-pulsed-field-ablation-technology-302140633.html
View original
content:https://www.prnewswire.co.uk/news-releases/field-medical-announces-first-in-human-trials-for-groundbreaking-fieldforce-ventricular-pulsed-field-ablation-technology-302140633.html