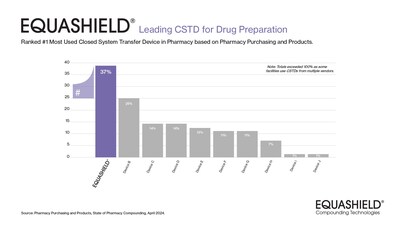
Pharmacy Purchasing & Products identified EQUASHIELD
as Top Choice for Drug Preparation of Hazardous Drugs
PORT
WASHINGTON, N.Y., May 13, 2024
/PRNewswire/ -- EQUASHIELD®, a global leader in the
development and distribution of Closed System Transfer Devices
(CSTDs) and pharmacy automation solutions, marks its 15th year of
innovation and excellence. This anniversary is highlighted by the
brand's recognition as the "Most Used CSTD for Drug Preparation" by
Pharmacy Purchasing and Products1 for an
impressive sixth consecutive year. Demonstrating its commitment to
healthcare safety and innovation, EQUASHIELD® is also
the first choice among facilities considering CSTDs.
Since its inception, EQUASHIELD® has been a
forerunner in driving advancements in safety and efficiency for
healthcare workers handling hazardous drugs. The brand's dedication
to superior engineering and manufacturing of Closed System Transfer
Devices (CSTDs) has not just set standards, but redefined them,
providing oncology workers with innovative solutions that
significantly mitigate exposure risks associated with hazardous
drug procedures. By integrating meticulous research, development,
and a profound understanding of end-user requirements,
EQUASHIELD® has introduced streamlined and user-centric
systems that prioritize safety and ease of use, setting a new
benchmark in the industry.
EQUASHIELD®'s Closed System Transfer Device is
uniquely designed to provide unparalleled safety for oncology
pharmacists and nurses involved in the compounding and
administration of hazardous drugs. The system's state-of-the-art
products are both a design and engineering feat, with pressure
equalization technology fueling everything they do. Central to
EQUASHIELD®'s product portfolio are its proprietary CSTD
features, which include an integrated barrier-type pressure
equalization system, a leak-preventing dry disconnection mechanism,
and an encapsulated closed-back syringe with a metal plunger rod to
mitigate hazardous drug contamination, preventing vapor escape and
plunger contamination. Central to EQUASHIELD®'s product
portfolio are its proprietary CSTD features, which include
peer-reviewed clinical studies2. EQUASHIELD's Syringe
Unit has also received FDA clearance reconfirming the freedom to
utilize the entire volume of the Syringe Unit up to its maximal
nominal volume3.
Since EQUASHIELD® was first recognized as the Most
Used CSTD for Drug Preparation in 2019, there has been a
significant uptick in adoption, with a 32% increase attributed to
its exceptional product benefits. Building on this legacy,
EQUASHIELD® unveiled the Mundus Mini HD, an innovative
automated compounding solution for hazardous drugs. This new
addition is poised to transform drug compounding by enhancing
safety and efficiency while reducing the potential for medication
errors and increasing patient safety. The Mundus Mini HD and
EQUASHIELD®'s comprehensive range of CSTDs underscore
the brand's "Safe, Simple, Closed" design ethos.
Jason Dutcher, Director of
Strategic Sales at EQUASHIELD®, expresses pride in the
company's achievements: "The recognition as the most used CSTD in
pharmacy, along with our top ranking among facilities ready to
adopt CSTDs, showcases our relentless pursuit of innovation,
safety, and efficiency. The Mundus Mini HD represents our
forward-thinking approach to healthcare safety, heralding a new era
of innovation."
As EQUASHIELD® celebrates this momentous anniversary,
it remains dedicated to its mission of improving healthcare safety
and efficiency through innovative solutions. With a robust history
of achievements and a future focused on continued innovation,
EQUASHIELD® is poised to maintain its leadership
position in the healthcare industry for years to come.
About EQUASHIELD®
Established in 2009, EQUASHIELD Compounding Technologies is a
globally recognized leading provider of manual and automated
solutions for compounding and administration, committed to
safeguarding healthcare professionals from exposure to hazardous
drugs while increasing efficiency. EQUASHIELD's product suite
includes its flagship CSTD and the Mundus Mini HD, a groundbreaker
in safe automated compounding of hazardous drugs. EQUASHIELD CSTD
is CE marked and FDA cleared for preventing microbial ingress for
up to seven days and for full volume use of the Syringe Unit.
Together the EQUASHIELD CSTD and Mundus Mini HD redefine handling
hazardous drugs, reducing the risk of exposure to hazardous drugs
and simplifying workflows.
For more information about EQUASHIELD® and its
solutions, visit [www.equashield.com]
For Media Inquiries:
[info@equashield.com]
References:
-
https://www.pppmag.com/digitalmag/Main.php?MagNo=377&PageNo=44#page/44
- https://www.equashield.com/sections/clinical-studies/
- https://www.accessdata.fda.gov/cdrh_docs/pdf22/K221513.pdf
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/equashield-marks-15-years-of-innovation-in-healthcare-safety-remaining-the-top-choice-cstd-for-hazardous-drug-preparation-for-six-consecutive-years-302142644.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/equashield-marks-15-years-of-innovation-in-healthcare-safety-remaining-the-top-choice-cstd-for-hazardous-drug-preparation-for-six-consecutive-years-302142644.html
SOURCE Equashield LLC