First HIFU patient treated at Allied Urological Services in New York City with the Ablatherm® Robotic HIFU device
June 14 2016 - 7:37AM

EDAP TMS SA (Nasdaq:EDAP), the global leader in therapeutic
ultrasound, today announced the first prostate ablation treatment
performed at a site managed by Allied Urological Services
(“Allied”), an affiliate of Austin, Texas based HealthTronics, Inc.
in New York, NY, using the Company’s FDA-cleared Ablatherm Robotic
HIFU. Dr. Altan Ilkay, a leading urologist at Allied with
significant experience in minimally invasive prostate treatments,
performed the procedure.
Dr. Ilkay commented: “The contemporary patient is
often not satisfied with the polar choices of active surveillance
(do nothing) and radical treatment with surgery or radiation. HIFU
is appropriate and attractive for many of the men I see in my
clinical practice. It is a versatile technology that allows for
tailoring of the procedure to fit the individual patient. The
Ablatherm device is a uniquely robotic device that allows for the
safe ablation of tissue.” He added: “The Ablatherm Robotic
HIFU provides a very user-friendly interface that allows the
physician to focus on the critical, clinical treatment of the
patient.”
The Ablatherm Robotic HIFU device used for this
procedure is owned by HealthTronics, Inc., a leading provider of
mobile medical technologies which is providing access to EDAP’s
industry-leading HIFU technology to select urologists [throughout
the United States].
Russell Newman, President of HealthTronics, said,
“We are excited to add EDAP’s Ablatherm HIFU to our best-in-class
portfolio of mobilized medical technologies and to work with our
partners at Allied as the first users of the device. Ablatherm and
HIFU are an important addition to our suite of technology offerings
as they help us meet our commitment of providing physicians with
the most innovative tools available for the treatment of their
patients.”
Marc Oczachowski, EDAP TMS Chief Executive Officer,
added: “We are very pleased that Allied, one of the country’s
largest networks of urologists, has established a HIFU program
incorporating the EDAP Ablatherm Robotic HIFU device. In
addition, this also extends our relationship with Allied, which has
been a longtime user of EDAP’s unique and exclusive Lithotripsy
technology, the Sonolith i-move.”
About EDAP TMS SAEDAP TMS SA
markets today Ablatherm® Robotic HIFU device for high-intensity
focused ultrasound (HIFU) for prostate tissue ablation in the U.S.
and for treatment of localized prostate cancer in the rest of the
world. HIFU treatment is shown to be a minimally invasive and
effective option for prostatic tissue ablation with a low
occurrence of side effects. Ablatherm-HIFU is generally recommended
for patients with localized prostate cancer (stages T1-T2) who are
not candidates for surgery or who prefer an alternative option, or
for patients who failed radiotherapy treatment. Ablatherm-HIFU is
approved for commercial distribution in Europe and some other
countries including Mexico and Canada, and has received 510(k)
clearance by the U.S. FDA. The Company also markets an innovative
robot-assisted HIFU device, the Focal One®, dedicated to focal
therapy of prostate cancer. Focal One® is CE marked but is not FDA
approved. The Company also develops its HIFU technology for the
potential treatment of certain other types of tumors. EDAP TMS SA
also produces and distributes medical equipment (the Sonolith®
lithotripters’ range) for the treatment of urinary tract stones
using extra-corporeal shockwave lithotripsy (ESWL) in most
countries including Canada and the U.S. For more information on the
Company, please visit http://www.edap-tms.com, and
http://www.hifu-planet.com.
About Allied Allied was formed in
1998 and is the management company for a urology group that has
nine AAAHC-accredited office based surgical sites located
throughout the New York metropolitan area that service 280
affiliated urologists. For more information, please visit
http://www.alliedmetromedical.com/.
About HealthTronicsHealthTronics,
Inc., established in 1989 and headquartered in Austin, Texas,
provides integrated, mobilized medical technologies and services,
as well as physician partnership opportunities. The company brings
its advanced technology and support systems to health care
providers throughout the United States. For more information
visit healthtronics.com.
Forward-Looking StatementsIn
addition to historical information, this press release may contain
forward-looking statements. Such statements are based on
management’s current expectations and are subject to a number of
risks and uncertainties, including matters not yet known to us or
not currently considered material by us, and there can be no
assurance that anticipated events will occur or that the objectives
set out will actually be achieved. Important factors that could
cause actual results to differ materially from the results
anticipated in the forward-looking statements include, among
others, the clinical status and market acceptance of our HIFU
devices and the continued market potential for our lithotripsy
device. Factors that may cause such a difference also may include,
but are not limited to, those described in the Company’s filings
with the Securities and Exchange Commission and in particular, in
the sections “Cautionary Statement on Forward-Looking Information”
and “Risk Factors” in the Company’s Annual Report on Form 20-F.
Contact:
Blandine Confort
Investor Relations / Legal Affairs
EDAP TMS SA
+33 4 72 15 31 72
bconfort@edap-tms.com
Investors:
Lee Roth
The Ruth Group
646-536-7012
lroth@theruthgroup.com
EDAP TMS (NASDAQ:EDAP)
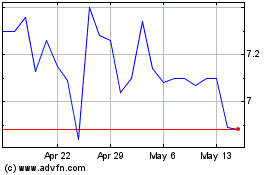
Historical Stock Chart
From Jun 2024 to Jul 2024
EDAP TMS (NASDAQ:EDAP)
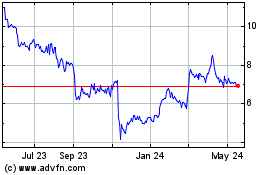
Historical Stock Chart
From Jul 2023 to Jul 2024