Recro Pharma Announces Key Executive Promotions to the Financial Team
January 03 2018 - 6:00AM

Recro Pharma, Inc. (Nasdaq:REPH), a revenue generating specialty
pharmaceutical company focused on therapeutics for the hospital and
other acute care settings, today announced several key promotions
to its executive leadership team: Michael Celano has been promoted
to Chief Operating Officer, and Ryan D. Lake has been promoted
to Chief Financial Officer. Both promotions are effective January
3, 2018.
“I am pleased to announce the promotions of Mike
and Ryan, two critical members of our financial leadership team,”
said Gerri Henwood, President and Chief Executive Officer of Recro
Pharma. “With the sustained, strong performance and significant
contribution of the contract development and manufacturing (CDMO)
facility to Recro, as well as the active pre-commercialization
efforts for IV meloxicam 30mg in anticipation of launch following
the potential approval by the U.S. Food and Drug Administration
(FDA) next year, it is imperative that we have a strong and skilled
team in place capable of executing across both divisions of Recro.
I look forward to continuing to work with both Mike and Ryan in
their new roles during this transformative time for the
Company.”
Mr. Celano has over 35 years of senior financial
leadership experience in the life sciences and biopharmaceutical
industries. Mr. Celano joined Recro in July 2016 as Chief Financial
Officer. During his tenure, he has provided financial leadership
through several Phase III clinical trials, the filing of the New
Drug Application (NDA) for IV meloxicam 30mg and preparing for
commercial launch, as well as through the continued strong
performance of the Company’s manufacturing division. In his new
role as Chief Operating Officer, Mr. Celano will continue to lead
finance and administration for Recro, and he will assume
responsibility for the Company’s CDMO division. Prior to joining
Recro, Mr. Celano served as Chief Financial Officer at Kensey Nash,
a Nasdaq-listed, medical device company, and BioRexis, a
venture-funded, life sciences company. Mr. Celano serves on the
board of OraSure Technologies (Nasdaq:OSUR). Prior to entering the
biopharmaceutical industry, Mr. Celano was a Partner at Arthur
Andersen/KPMG, where he worked from 1980 until 2004, and led its
life sciences practices. Mr. Celano received a bachelor’s degree in
accounting from Saint Joseph’s University.
Mr. Lake joined Recro in June 2017 as the
Company’s Chief Accounting Officer, and he has over 15 years of
senior financial leadership experience. Prior to joining Recro, Mr.
Lake served as Chief Financial Officer and Vice President of
Finance of Aspire Bariatrics, Inc., a privately-held,
commercial-stage, medical device company. From 2012 to 2015, Mr.
Lake held executive management and senior finance positions,
including Director of the Natural Materials Division, Controller
and Senior Director of Finance, at DSM Biomedical (successor to
Kensey Nash after its acquisition in 2012), a division of Royal DSM
(listed on Euronext Amsterdam), a global science-based company
active in health, nutrition and materials. From 2002 to 2012, Mr.
Lake held various senior financial positions of increasing
responsibility, most notably Senior Director of Finance and Interim
CFO, with Kensey Nash Corporation, a Nasdaq-listed, medical device
company. Earlier in his career, Mr. Lake worked at Deloitte
& Touche, LLP. Mr. Lake is a Certified Public Accountant,
Chartered Global Management Accountant and holds a B.S. degree in
Accounting from West Chester University of Pennsylvania.
About Recro Pharma, Inc.
Recro Pharma is a specialty pharmaceutical
company that operates through two business divisions, an Acute
Care, hospital product division and a revenue-generating contract
development and manufacturing, or CDMO division, located at the
Company’s Gainesville facility. The Acute Care division is
primarily focused on developing innovative products for hospital
and other acute care settings. The Company’s lead product candidate
is a proprietary injectable form of meloxicam, a long-acting
preferential COX-2 inhibitor. IV meloxicam 30mg has
successfully completed two pivotal Phase III clinical efficacy
trials in patients following bunionectomy and abdominoplasty
surgeries, a large double blind Phase III safety trial, four Phase
II clinical trials for the management of moderate to severe
post-operative pain, as well as other safety studies. As
injectable meloxicam is in the non-opioid class of drugs, the
Company believes it will overcome many of the issues associated
with commonly prescribed opioid therapeutics, including respiratory
depression, constipation, excessive nausea and vomiting, as well as
having no addictive potential while maintaining meaningful
analgesic effects for relief of pain. The Company’s CDMO division
leverages its formulation expertise to develop and manufacture
pharmaceutical products using its proprietary delivery technologies
and other manufacturing services for commercial partners who
commercialize or plan to commercialize these products. These
collaborations can result in revenue streams including royalties,
profit sharing, research and development and manufacturing fees,
which support continued operations for its CDMO division and it
contributes non-dilutive funding for the development and
pre-commercialization activities of its Acute Care division.
Cautionary Statement Regarding Forward Looking
Statements
This press release contains forward-looking
statements that involve risks and uncertainties. Such forward
looking statements reflect Recro's expectations about its future
performance and opportunities that involve substantial risks and
uncertainties. When used herein, the words "anticipate," "believe,"
"estimate," "upcoming," "plan," "target", "intend" and "expect" and
similar expressions, as they relate to Recro or its management, are
intended to identify such forward-looking statements. These forward
looking statements are based on information available to Recro as
of the date of this press release and are subject to a number of
risks, uncertainties, and other factors that could cause Recro’s
performance to differ materially from those expressed in, or
implied by, these forward looking statements. Recro assumes no
obligation to update any such forward-looking statements. Factors
that could cause Recro’s actual performance to materially differ
from those expressed in the forward-looking statements set forth in
this press release include, without limitation: the ability to
obtain and maintain regulatory approval of injectable meloxicam and
the labeling under any such approval; regulatory developments in
the United States and foreign countries; results and timing of the
clinical trials of injectable meloxicam, the Company’s ability to
achieve its financial goals, including financial guidance; the
Company’s ability to raise future financing for continued
development, product commercialization and the payment of
milestones; the Company’s ability to pay its debt; customer product
performance and ordering patterns, the performance of third-party
suppliers and manufacturers; the Company’s ability to obtain,
maintain and successfully enforce adequate patent and other
intellectual property protection; and the successful
commercialization of injectable meloxicam. The forward looking
statements in this press release should be considered together with
the risks and uncertainties that may affect Recro’s business and
future results included in Recro’s filings with the Securities and
Exchange Commission at www.sec.gov. Recro assumes no
obligation to update any such forward looking statements.
CONTACT:
Investor Relations Contact:Argot PartnersSusan
Kim/Natalie Wildenradt(212)
600-1902susan@argotpartners.comnatalie@argotpartners.com
Recro Pharma, Inc.Michael Celano(484)
395-2413mcelano@recropharma.com
Media Contact:Argot PartnersEliza
Schleifstein(973) 361-1546 eliza@argotpartners.com
OraSure Technologies (NASDAQ:OSUR)
Historical Stock Chart
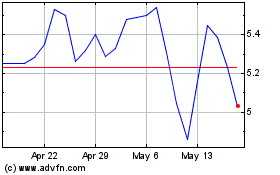
From Jun 2024 to Jul 2024
OraSure Technologies (NASDAQ:OSUR)
Historical Stock Chart
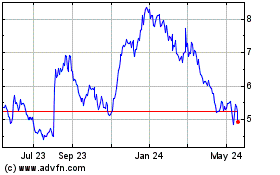
From Jul 2023 to Jul 2024