false 0001662774 0001662774 2023-07-21 2023-07-21 0001662774 us-gaap:CommonStockMember 2023-07-21 2023-07-21 0001662774 us-gaap:SeriesAPreferredStockMember 2023-07-21 2023-07-21
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): July 21, 2023
QUINCE THERAPEUTICS, INC.
(Exact name of registrant as specified in its charter)
|
|
|
|
|
| Delaware |
|
001-38890 |
|
90-1024039 |
| (State or other jurisdiction of incorporation or organization) |
|
(Commission File Number) |
|
(I.R.S. Employer Identification No.) |
|
|
|
| 601 Gateway Boulevard, Suite 1250 South San Francisco, California |
|
94080 |
| (Address of principal executive offices) |
|
(Zip Code) |
Registrant’s telephone number, including area code: (415) 910-5717
Not Applicable
(Former name or former address, if changed since last report.)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
| ☐ |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ☐ |
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ☐ |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ☐ |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13d-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
|
|
|
|
|
| Title of each class |
|
Trading
Symbol(s) |
|
Name of each exchange on which registered |
| Common Stock, par value $0.001 per share |
|
QNCX |
|
The Nasdaq Stock Market LLC |
| Series A Junior Participating Preferred Purchase Rights |
|
N/A |
|
The Nasdaq Stock Market LLC |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
| Item 1.01. |
Entry into a Material Definitive Agreement |
On July 21, 2023 (the “Agreement Date”), Quince Therapeutics, Inc., a Delaware corporation (the “Company”), EryDel Italy, Inc., a Delaware corporation and wholly owned indirect subsidiary of the Company, EryDel S.p.A, a company with shares incorporated under the laws of Italy, (“EryDel”), holders of EryDel capital stock and the managers of EryDel (the “EryDel Shareholders”) and Shareholder Representative Services LLC, a Colorado limited liability company solely in its capacity as the representative, agent and attorney-in-fact of the EryDel Shareholders, entered into a Stock Purchase Agreement (the “Purchase Agreement”), pursuant to which, and subject to the terms and conditions set forth in the Purchase Agreement, the Company will indirectly acquire all of the outstanding equity interests of EryDel (the “EryDel Acquisition”). Upon completion of the EryDel Acquisition, (i) the Company will issue up to 7,250,352 shares of common stock of the Company to the EryDel Shareholders, subject to downward adjustment (the “Company Common Stock”), resulting in the EryDel Shareholders owning a maximum of approximately 16.7% of the outstanding common stock of the Company, and (ii) the EryDel Shareholders will have a contingent right to receive up to an aggregate of $485,000,000 in potential cash payments, comprised of up to $5,000,000 upon the achievement of a specified development milestone, $25,000,000 at NDA acceptance, up to $60,000,000 upon the achievement of specified approval milestones, and up to $395,000,000 upon the achievement of specified on market and sales milestones, with no royalties paid to EryDel. The Company will also assume in the transaction EryDel’s $13 million (€10 million in principal) European Investment Bank (EIB) loan with scheduled payments beginning in the second half of 2026.
The completion of the EryDel Acquisition is subject to customary conditions, including: (a) the absence of any legal proceeding that has the effect of enjoining or otherwise making illegal the consummation of the EryDel Acquisition; (b) the approval for listing on the Nasdaq Stock Market shares of Company Common Stock issuable pursuant to the Purchase Agreement; (c) subject to certain exceptions, the accuracy of the representations and warranties of the other party and performance by each party in all material respects of its obligations under the Purchase Agreement; (d) the absence of a material adverse effect on the Company or EryDel; and (e) retention of certain key employees.
The Company and EryDel are permitted under certain circumstances to terminate the Purchase Agreement.
The Purchase Agreement includes customary representations, warranties and covenants of the Company and EryDel. In addition, the EryDel Shareholders have agreed to indemnify the Company for losses arising out of, among other things, inaccuracies in, or breaches of, the representations, warranties and covenants of EryDel, pre-closing taxes of EryDel and matters relating to claims by holders or former holders of EryDel Stock, subject to certain caps, deductibles and other limitations and obligations.
The Company and EryDel intend, for U.S. federal income tax purposes, that the Company and its affiliates shall be entitled to file an election under Section 338(g) of the Internal Revenue Code of 1986 and any corresponding or local election with respect to the EryDel Acquisition.
The Company Board of Directors also approved the appointment of Luca Benatti to serve as a director of the Company, contingent and effective upon the closing of the EryDel Acquisition.
A copy of the Purchase Agreement is attached as Exhibit 2.1 to this Current Report on Form 8-K and is incorporated herein by reference. The foregoing description of the Purchase Agreement does not purport to be complete and is qualified in its entirety by reference to the Purchase Agreement. The representations, warranties and covenants set forth in the Purchase Agreement have been made only for the purposes of the Purchase Agreement and solely for the benefit of the parties to the Purchase Agreement, may be subject to limitations agreed upon by the contracting parties, including being qualified by confidential disclosures made for the purposes of allocating contractual risk between the parties to the Purchase Agreement instead of establishing these matters as facts, as well as by information contained in the Company’s most recent Annual Report on Form 10-K and Quarterly Report on Form 10-Q, and may be subject to standards of materiality applicable to the contracting parties that differ from those applicable to investors. Accordingly, the Purchase Agreement is included with this filing only to provide investors with information regarding the terms of the Purchase Agreement and not to provide investors with any other factual information regarding the parties or their respective businesses.
| Item 2.02 |
Results of Operations and Financial Condition. |
On July 24, 2023, the Company announced that upon completion of the EryDel Acquisition, it expects to have capital resources of approximately $87.6 million in cash, cash equivalents and short term investments, as of June 30, 2023. This amount is preliminary and is subject to change upon completion of the quarterly closing procedures and any adjustments that may result from the completion of the review of the financial statements. As a result, this preliminary estimate may differ from the actual results that will be reflected in the financial statements when they are completed and publicly disclosed. Additional information and disclosures would be required for a more complete understanding of the Company’s financial position and results of operations as of and for the three months ended June 30, 2023. The Company’s independent registered public accounting firm has not audited, reviewed, compiled, or performed any procedures with respect to such data, and, accordingly, does not express an opinion or any other form of assurance about it.
The information in Item 2.02 of this Current Report on Form 8-K shall not be deemed to be “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be incorporated by reference in any filing under the Securities Act of 1933, as amended, or the Exchange Act, except as shall be expressly set forth by specific reference in any such filing, regardless of any general incorporation language in such filing.
| Item 2.03 |
Creation of a Direct Financial Obligation or an Obligation under an Off-Balance Sheet Arrangement of a Registrant. |
In connection with and contingent upon the consummation of the EryDel Acquisition, the Company will assume EryDel’s $13 million (€10 million in principal) European Investment Bank (EIB) loan with scheduled payments beginning in the second half of 2026.
| Item 3.02 |
Unregistered Sales of Equity Securities. |
The disclosure set forth above in Item 1.01 of this Current Report on Form 8-K is incorporated by reference herein. The shares of the Company Common Stock that may be issued to EryDel Shareholders pursuant to the Purchase Agreement are exempt from registration in reliance on exemption provided for under Section 4(a)(2) of the Securities Act.
| Item 5.02 |
Departure of Directors or Certain Officers; Election of Directors; Appointment of Certain Officers; Compensatory Arrangements of Certain Officers. |
Contingent Appointment of Director
On July 21, 2023, the Company Board of Directors approved the appointment of Luca Benatti to serve as a director of the Company, contingent and effective upon the closing of the EryDel Acquisition.
Departure of Chief Medical Officer
On July 24, 2023, the Company announced that Karen Smith, Chief Medical Officer of the Company, would be leaving the Company, effective September 1, 2023. Ms. Smith’s departure is not the result of any disagreement with the Company on any matter relating to the Company’s financial operations, policies or practices.
| Item 7.01 |
Regulation FD Disclosure. |
On July 24, 2023, the Company posted an investor presentation relating to the EryDel Acquisition to the Company’s website. A copy of the investor presentation is attached hereto as Exhibit 99.1 and incorporated herein by reference.
The information in this Item 7.01 is furnished and shall not be deemed “filed” for purposes of Section 18 of the Exchange Act, or otherwise subject to the liabilities of that section and shall not deemed incorporated by reference in any filing under the Exchange Act or the Securities Act of 1933, as amended, except as shall be expressly set forth by specific reference in such filing.
On July 24, 2023, the Company issued a press release announcing the execution of the Purchase Agreement. A copy of the press release is attached as Exhibit 99.2 to this Current Report on Form 8-K and is incorporated herein by reference.
Forward-Looking Statements
Certain Statements in this Current Report on Form 8-K contain “forward-looking statements” that are subject to substantial risks and uncertainties. Forward-looking statements contained herein may be identified by the use of words such as “anticipate,” “expect,” “believe,” “plan,” “intend,” “will,” “may,” “should,” “estimate,” “project,” “outlook,” “runway,” “forecast,” “potential” or other similar words. Examples of forward-looking statements include, among others, the ability of the parties to consummate the proposed EryDel Acquisition and the timing thereof, the ability of the Company to timely and successfully achieve the anticipated benefits of the EryDel Acquisition, including the continued current and future clinical development and potential expansion of EryDel assets, related platform, and related timing and costs; the strategic development path for EryDex; planned FDA and EMA submissions and clinical trials and timeline, prospects, and milestone expectations; the timing and success of the clinical trials and related data, including plans and the ability to initiate, fund, conduct and/or complete current and additional studies; the potential therapeutic benefits, safety, and efficacy of EryDex; statements about its ability to obtain, and the timing relating to, further development of EryDex, regulatory submissions and interactions with regulators; therapeutic and commercial potential; the integration of EryDel’s business, operations, and employees into the Company; the Company’s future development plans and related timing; its cash position and projected cash runway; the Company’s focus, objectives, plans, and strategies; and the ability to execute on any strategic transactions. Forward-looking statements are based on the Company’s current expectations and are subject to inherent uncertainties, risks, and assumptions that are difficult to predict and could cause actual results to differ materially from what the Company expects. Further, certain forward-looking statements are based on assumptions as to future events that may not prove to be accurate. Factors that could cause actual results to differ include, but are not limited to, the risks and uncertainties described the section titled “Risk Factors” in the Company’s Quarterly Report on Form 10-Q filed with the SEC on May 15, 2023, and other reports as filed with the SEC. Forward-looking statements contained in this Current Report on Form 8-K are made as of this date, and the Company undertakes no duty to update such information except as required under applicable law.
| Item 9.01. |
Financial Statements and Exhibits. |
(d) Exhibits
|
|
|
| Exhibit |
|
Description |
|
|
| 2.1* |
|
Stock Purchase Agreement, dated as of July 21, 2023, by and among Quince Therapeutics, Inc., EryDel Italy, Inc., EryDel S.p.A., certain holders and the managers set forth on Schedule II thereto, and Shareholder Representative Services LLC, as the stockholder representative. |
|
|
| 99.1 |
|
Investor Presentation, dated as of July 24, 2023. |
|
|
| 99.2 |
|
Press Release, dated as of July 24, 2023. |
|
|
| 104 |
|
Cover Page Interactive Data File (embedded within the Inline XBRL document) |
| * |
Certain exhibits and schedules have been omitted pursuant to Item 601(b)(2) of Regulation S-K. The registrant agrees to furnish supplementally to the SEC a copy of any omitted exhibits or schedules upon request. |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
|
|
|
|
|
|
|
|
|
|
|
QUINCE THERAPEUTICS, INC. |
|
|
|
|
|
|
|
|
By: |
|
/s/ Dirk Thye |
| Date: July 24, 2023 |
|
|
|
Name: |
|
Dirk Thye |
|
|
|
|
Title: |
|
Chief Executive Officer |
Exhibit 2.1
STOCK PURCHASE AGREEMENT
AMONG
QUINCE THERAPEUTICS, INC.,
ERYDEL ITALY, INC.,
ERYDEL S.P.A.,
THE SHAREHOLDERS AND THE MANAGERS
SET FORTH ON SCHEDULE II HERETO
AND
SHAREHOLDER REPRESENTATIVE SERVICES LLC, AS HOLDER
REPRESENTATIVE FOR THE SHAREHOLDERS
TABLE OF CONTENTS
Page
|
|
|
|
|
|
|
| ARTICLE I CERTAIN DEFINITIONS |
|
|
2 |
|
|
|
| ARTICLE II THE TRANSACTIONS |
|
|
17 |
|
|
|
|
| 2.1 |
|
Sale and Purchase of Company Stock |
|
|
17 |
|
| 2.2 |
|
Closing Transaction Consideration |
|
|
18 |
|
| 2.3 |
|
Closing Activities and Disbursement of Closing Transaction Consideration |
|
|
18 |
|
| 2.4 |
|
Closing Calculations |
|
|
19 |
|
| 2.5 |
|
Company Securities |
|
|
19 |
|
| 2.6 |
|
Transfer Restrictions; Stop-Transfer Instructions |
|
|
19 |
|
| 2.7 |
|
Earnout |
|
|
20 |
|
| 2.8 |
|
Reversion |
|
|
23 |
|
| 2.9 |
|
Further Assurances |
|
|
24 |
|
| 2.10 |
|
Withholding |
|
|
24 |
|
|
|
| ARTICLE III REPRESENTATIONS AND WARRANTIES OF COMPANY |
|
|
24 |
|
|
|
|
| 3.1 |
|
Organization and Good Standing |
|
|
24 |
|
| 3.2 |
|
Subsidiaries |
|
|
24 |
|
| 3.3 |
|
Power, Authorization and Validity |
|
|
25 |
|
| 3.4 |
|
Capitalization of Company |
|
|
25 |
|
| 3.5 |
|
Consents; Conflict |
|
|
26 |
|
| 3.6 |
|
Litigation |
|
|
26 |
|
| 3.7 |
|
Taxes |
|
|
26 |
|
| 3.8 |
|
Financial Statements; Books and Records |
|
|
29 |
|
| 3.9 |
|
Assets and Properties |
|
|
30 |
|
| 3.10 |
|
Absence of Certain Changes |
|
|
30 |
|
| 3.11 |
|
Contracts, Agreements, Arrangements, Commitments and Undertakings |
|
|
30 |
|
| 3.12 |
|
Intellectual Property |
|
|
32 |
|
| 3.13 |
|
Regulatory Matters |
|
|
36 |
|
| 3.14 |
|
Compliance with Laws and Permits |
|
|
38 |
|
| 3.15 |
|
Certain Business Practices |
|
|
38 |
|
| 3.16 |
|
Certain Transactions and Agreements |
|
|
39 |
|
| 3.17 |
|
Employees, ERISA and Other Compliance |
|
|
39 |
|
| 3.18 |
|
Transaction Expenses |
|
|
42 |
|
| 3.19 |
|
Insurance |
|
|
42 |
|
-i-
TABLE OF CONTENTS
(continued)
Page
|
|
|
|
|
|
|
| 3.20 |
|
Environmental Matters |
|
|
43 |
|
| 3.21 |
|
Export Control Laws |
|
|
43 |
|
| 3.22 |
|
Accuracy of Information Supplied |
|
|
43 |
|
| 3.23 |
|
Receivables |
|
|
43 |
|
| 3.24 |
|
Related Party Contracts |
|
|
43 |
|
| 3.25 |
|
Anti-Corruption |
|
|
44 |
|
| 3.26 |
|
Extraordinary Transactions |
|
|
44 |
|
|
|
| ARTICLE IV REPRESENTATIONS AND WARRANTIES OF SHAREHOLDERS |
|
|
44 |
|
|
|
|
| 4.1 |
|
Power, Authorization and Validity |
|
|
44 |
|
| 4.2 |
|
Title to Company Stock |
|
|
45 |
|
| 4.3 |
|
Brokerage |
|
|
45 |
|
| 4.4 |
|
Non-U.S. Status |
|
|
45 |
|
| 4.5 |
|
Transaction Securities |
|
|
45 |
|
|
|
| ARTICLE V REPRESENTATIONS AND WARRANTIES OF BUYER |
|
|
47 |
|
|
|
|
| 5.1 |
|
Organization and Good Standing |
|
|
48 |
|
| 5.2 |
|
Subsidiaries |
|
|
48 |
|
| 5.3 |
|
Power, Authorization and Validity |
|
|
48 |
|
| 5.4 |
|
No Conflict |
|
|
49 |
|
| 5.5 |
|
Litigation |
|
|
49 |
|
| 5.6 |
|
Capitalization |
|
|
49 |
|
| 5.7 |
|
SEC Filings and Buyer Financials |
|
|
50 |
|
| 5.8 |
|
Absence of Certain Changes |
|
|
51 |
|
| 5.9 |
|
Insurance |
|
|
51 |
|
| 5.10 |
|
Litigation; Orders |
|
|
51 |
|
| 5.11 |
|
Finders and Brokers |
|
|
51 |
|
| 5.12 |
|
Issuance of Buyer Shares |
|
|
52 |
|
|
|
| ARTICLE VI SURVIVAL; INDEMNIFICATION |
|
|
52 |
|
|
|
|
| 6.1 |
|
Survival |
|
|
52 |
|
| 6.2 |
|
Indemnification |
|
|
52 |
|
| 6.3 |
|
Limitations on Indemnification |
|
|
54 |
|
| 6.4 |
|
Holdback Shares Release |
|
|
55 |
|
| 6.5 |
|
Claims for Indemnification |
|
|
55 |
|
-ii-
TABLE OF CONTENTS
(continued)
Page
|
|
|
|
|
|
|
| 6.6 |
|
Objections to and Payment of Claims |
|
|
56 |
|
| 6.7 |
|
Resolution of Objections to Claims |
|
|
56 |
|
| 6.8 |
|
Third-Party Claims |
|
|
57 |
|
| 6.9 |
|
Holder Representative |
|
|
58 |
|
|
|
| ARTICLE VII COMPANY PARTY COVENANTS |
|
|
59 |
|
|
|
|
| 7.1 |
|
Advise of Changes |
|
|
59 |
|
| 7.2 |
|
Business Activities |
|
|
60 |
|
| 7.3 |
|
Conduct of the Company Group Business |
|
|
60 |
|
| 7.4 |
|
Necessary Consents |
|
|
62 |
|
| 7.5 |
|
No Other Negotiations |
|
|
62 |
|
| 7.6 |
|
Access to Information |
|
|
63 |
|
| 7.7 |
|
Satisfaction of Conditions Precedent |
|
|
63 |
|
| 7.8 |
|
Cooperation with SEC Filings |
|
|
63 |
|
| 7.9 |
|
Regulatory Approvals |
|
|
63 |
|
| 7.10 |
|
Release Agreements |
|
|
64 |
|
| 7.11 |
|
Company Options |
|
|
64 |
|
| 7.12 |
|
By-Laws Amendment |
|
|
64 |
|
|
|
| ARTICLE VIII BUYER COVENANTS |
|
|
64 |
|
|
|
|
| 8.1 |
|
Regulatory Approvals |
|
|
64 |
|
| 8.2 |
|
Satisfaction of Conditions Precedent |
|
|
65 |
|
| 8.3 |
|
Documents and Information |
|
|
65 |
|
| 8.4 |
|
Post-Closing Board of Directors |
|
|
65 |
|
| 8.5 |
|
Employee Matters |
|
|
65 |
|
| 8.6 |
|
Conduct of Buyer Business |
|
|
66 |
|
| 8.7 |
|
Access to Information |
|
|
66 |
|
|
|
| ARTICLE IX CONDITIONS TO CLOSING |
|
|
67 |
|
|
|
|
| 9.1 |
|
Conditions to Each Party’s Obligation to Effect the Closing |
|
|
67 |
|
| 9.2 |
|
Additional Conditions to Obligations of Buyer |
|
|
67 |
|
| 9.3 |
|
Additional Conditions to Obligations of Shareholders |
|
|
70 |
|
|
|
| ARTICLE X TERMINATION OF AGREEMENT |
|
|
70 |
|
|
|
|
| 10.1 |
|
Termination by Mutual Consent |
|
|
70 |
|
| 10.2 |
|
Unilateral Termination |
|
|
70 |
|
| 10.3 |
|
Effect of Termination |
|
|
71 |
|
-iii-
TABLE OF CONTENTS
(continued)
Page
|
|
|
|
|
|
|
| ARTICLE XI TAX MATTERS |
|
|
71 |
|
|
|
|
| 11.1 |
|
Preparation of Tax Returns |
|
|
71 |
|
| 11.2 |
|
Cooperation |
|
|
72 |
|
| 11.3 |
|
Transfer Taxes |
|
|
72 |
|
| 11.4 |
|
Section 338(g) Election |
|
|
72 |
|
|
|
| ARTICLE XII MISCELLANEOUS |
|
|
72 |
|
|
|
|
| 12.1 |
|
Public Announcements |
|
|
72 |
|
| 12.2 |
|
Governing Law; Exclusive Jurisdiction; Waiver of Jury Trial |
|
|
73 |
|
| 12.3 |
|
Assignment; Binding Upon Successors and Assigns |
|
|
73 |
|
| 12.4 |
|
Severability |
|
|
74 |
|
| 12.5 |
|
Counterparts |
|
|
74 |
|
| 12.6 |
|
Other Remedies |
|
|
74 |
|
| 12.7 |
|
Amendments and Waivers |
|
|
74 |
|
| 12.8 |
|
Expenses |
|
|
74 |
|
| 12.9 |
|
Notices |
|
|
74 |
|
| 12.10 |
|
Interpretation; Rules of Construction |
|
|
75 |
|
| 12.11 |
|
Third Party Beneficiary Rights |
|
|
76 |
|
| 12.12 |
|
Release |
|
|
76 |
|
| 12.13 |
|
Shareholder Acknowledgement |
|
|
77 |
|
| 12.14 |
|
Entire Agreement |
|
|
77 |
|
-iv-
LIST OF SCHEDULES
|
|
|
| Schedule I |
|
Milestones and Milestone Payments |
| Schedule II |
|
Shareholders |
LIST OF EXHIBITS
|
|
|
| Exhibit B |
|
Sample NWC Spreadsheet |
| Exhibit C
Exhibit D |
|
Accredited Investor Questionnaire Settlement
Agreement |
STOCK PURCHASE AGREEMENT
This Stock Purchase Agreement (this “Agreement”) is made and entered into as of the date the last acceptance has been
received (the “Agreement Date”) by and among (i) Quince Therapeutics, Inc., a Delaware corporation (“Buyer”), (ii) : EryDel Italy, Inc., a Delaware corporation and an indirect, wholly owned subsidiary of Buyer
(“IT Buyer”), (iii) EryDel S.p.A., a company with shares (società per azioni) incorporated under the laws of Italy, with registered office in Bresso (MI), Via Antonio Meucci no. 3, registered with the
Companies’ Register of Milano Monza Brianza Lodi (enrolled in the special section as an innovative small-medium-enterprise), Italian tax code and VAT no. 02290380415, REA number MI – 1993330 (“Company”), (iv) the holders
of Company Stock (as such term is defined below) and the managers set forth on Schedule II hereto (the “Shareholders” and, together with the Company, the “Company Parties”) and (v) Shareholder Representative
Services LLC, a Colorado limited liability company solely in its capacity as the representative, agent and attorney-in-fact of the Shareholders (the “Holder
Representative”). Buyer and the Company Parties are sometimes referred to herein individually as a “Party” and, collectively, as the “Parties” and references to Schedules and Exhibits refer to the Schedules
and Exhibits attached to this Agreement, unless otherwise noted. Certain capitalized terms used herein are defined in Article I.
RECITALS
A. The Parties
desire that, upon the terms and subject to the conditions set forth in this Agreement, (i) the Company sell, Transfer, assign, convey and deliver to Buyer all of the US Subsidiary Stock, and Buyer purchase, acquire and accept the same and
(ii) immediately following the US Sale (as defined herein), the Shareholders sell, Transfer, assign, convey and deliver to IT Buyer all of the Company Stock, and IT Buyer purchase, acquire and accept the same.
B. The board of directors of Buyer and IT Buyer have (i) declared this Agreement and the Transactions, including the issuance of shares
of Buyer Common Stock in connection therewith, upon the terms and subject to the conditions set forth herein, advisable, fair to and in the best interests of Buyer and IT Buyer and (ii) approved this Agreement and the Transactions, including
the issuance of shares of Buyer Common Stock in connection therewith, upon the terms and subject to the conditions set forth herein.
C.
Concurrently with the execution of this Agreement and as a material inducement to the willingness of Buyer to enter into this Agreement, (i) each Key Employee is executing and delivering to Buyer an employment offer letter and Buyer’s
other customary employment documents (the “Key Employee Agreements”), to be effective at the Closing, and (ii) each Shareholder has executed and delivered to Buyer an Accredited Investor Questionnaire, certifying that such
Shareholder is an Accredited Investor.
D. Concurrently with the execution of this Agreement and as material inducement to the willingness
of Buyer to enter into this Agreement, each Optionholder is executing an option cancellation and release agreement with respect all Company Options held by such Optionholder.
E. Concurrently with the execution of this Agreement and as material inducement to the willingness of Buyer to enter into this Agreement, each
investor entitled to issuance of warrants is executing a warrant cancellation and release agreement with respect to all rights to any warrants held by such investor.
F. On July 11, 2023, Buyer has submitted, jointly with the Company, a pre-filing pursuant to
Article 2 quater, par. 2, of Italian Law Decree No. 21 dated 15 March 2012 converted in law no. 56 dated 11 May 2012, as amended, and Article 7 of Prime Ministerial Decree No. 133 dated 1 August 2022 (the “Golden
Power Pre-Filing”), in connection with the acquisition of 100% of the Company’s share capital so as to anticipate the review of said acquisition and the obtainment of the Golden Power Clearance
(as defined below).
G. Buyer and the Company Parties desire to make certain representations, warranties,
covenants and agreements in connection with the Transactions (as defined below) and to prescribe various conditions to the Transactions.
Now, therefore, in consideration of the foregoing and the mutual promises, covenants and conditions contained herein, the parties hereby agree
as follows:
ARTICLE I
CERTAIN DEFINITIONS
As
used in this Agreement, the following terms shall have the meanings set forth below. Unless indicated otherwise, all mathematical calculations contemplated hereby shall be made to the fifth decimal place.
“Accounting Principles” means the accounting methods, policies, principles, practices and procedures, with consistent
classifications, judgments and estimation methodologies, as provided by section 2423 and subsequent of the Civil Code, as integrated and interpreted by the accounting principles prepared by the “Consiglio Nazionale dei Dottori Commercialisti
e dei Ragionieri” and/or by the “OIC Organismo Italiano di Contabilità”, that were used in the preparation of the Company Financial Statements.
“Affiliate” means, with respect to any specified Person, any Person that, directly or indirectly through one or more
entities, controls or is controlled by, or is under common control with, such specified Person. The term “control” (including the terms “controlled by” and “under common control with”) means the possession, directly or
indirectly, of the power to direct or cause the direction or the management and policies of a Person, whether through the ownership of voting securities, by Contract or otherwise.
“Agreement” has the meaning set forth in the Preamble.
“Agreement Date” has the meaning set forth in the Preamble.
“Alternative Transaction” means, with respect to any Person, (i) any acquisition or purchase by any Person or
“group” (as defined under Section 13(d) of the Exchange Act and the rules and regulations promulgated thereunder) of any outstanding shares of its capital stock on such date of acquisition (other than as provided for or permitted by
this Agreement), or any merger, consolidation, business combination or similar transaction involving such Person or its controlled affiliates pursuant to which the stockholders of such Person immediately preceding such transaction, would cease to
hold, directly or indirectly, 100% of the equity or voting securities of the surviving or resulting entity following such transaction or (ii) any sale or disposition of any assets of such Person or its Subsidiaries.
“Ancillary Agreements” means the Company Ancillary Agreements and the Buyer Ancillary Agreement.
“Antitrust Authority” means the Federal Trade Commission, the Antitrust Division of the United States Department of Justice,
the attorneys general of the several states of the United States and any other Governmental Entity having jurisdiction with respect to the transactions contemplated hereby pursuant to applicable Antitrust Law(s).
2
“Antitrust Law” means, the Sherman Antitrust Act of 1890, the Clayton
Antitrust Act of 1914, the HSR Act, the Federal Trade Commission Act of 1914, Council Regulation 139/2004 of the European Union and all other national, federal or state, domestic or foreign, if any, statutes, rules, regulations, orders, decrees,
administrative and judicial doctrines and other Laws that are designed or intended to prohibit, restrict or regulate actions having the purpose or effect of monopolization or restraint of trade, affecting competition or market conditions through
merger, acquisition or other transaction or effectuating foreign investment.
“Applicable Law” means, collectively
(including for any country where the Company Group conducts the Company Business, including Italy and the United States of America), the federal, state, regional, provincial, territorial, local, municipal or other subdivision’s laws, statutes,
ordinances, regulations, rules, any supranational laws and all orders, writs, injunctions, awards, judgments and decrees applicable to the assets, properties and business (and any regulations promulgated thereunder) of the applicable Person that is
or has been issued, enacted, adopted, passed, approved, promulgated, made, implemented or otherwise put into effect by or under the authority of any Governmental Authority and which is directly or indirectly applicable to the Company and to its
activities.
“Approval” means, with respect to any Company Product or Service in any regulatory jurisdiction, clearance
or approval from the applicable Regulatory Authority sufficient to develop, manufacture, commercialize or otherwise exploit, any Company Product or Service in such regulatory jurisdiction in accordance with Applicable Laws including receipt of
pricing and reimbursement approvals, where applicable.
“Balance Sheet Date” means December 31, 2022.
“Base Share Consideration” means 7,250,352 shares of Buyer Common Stock.
“Business Day” means a day other than Saturday, Sunday or any day on which banks located in San Francisco, CA or Milan, Italy
are authorized or obligated to close.
“Buyer” has the meaning set forth in the Preamble.
“Buyer Ancillary Agreement” means, collectively, the certificate to be delivered on behalf of Buyer by an officer or officers
of Buyer at the Closing pursuant to Section 9.3(e).
“Buyer Certificate of Incorporation” means
Buyer’s Amended and Restated Certificate of Incorporation as filed with the Secretary of State of the State of Delaware on May 13, 2019, as amended on August 1, 2022.
“Buyer Common Stock” means Buyer’s common stock, par value $0.001 per share.
“Buyer Common Stock Price” shall mean the volume weighted average price, rounded to four decimal points, of a share of Buyer
Common Stock on Nasdaq calculated from the closing price and daily trading volumes reported by The Wall Street Journal for the period of ten (10) full trading days ending and including the trading day immediately prior to fifth trading
day immediately prior to the Closing.
“Buyer Disclosure Schedule” means the disclosure schedule dated as of the
Agreement Date and delivered by Buyer to the Company Parties on the Agreement Date arranged in sections corresponding to the numbered and letter sections contained in Article V of this Agreement.
“Buyer Financial Statements” has the meaning set forth in Section 5.7(c).
3
“Buyer Organizational Documents” means the Buyer Certificate of
Incorporation and the bylaws of Buyer, each as amended to date.
“Buyer Parties” means Buyer and its Affiliates
(including after the Closing, the Company and the US Subsidiary for so long as such entities remain Affiliates of Buyer).
“Buyer Permitted Encumbrances” means, with respect to Buyer (a) Encumbrances for Taxes or assessments and similar
governmental charges or levies that are either (i) not yet due and payable or (ii) being contested in good faith (and for which adequate accruals or reserves have been established on the Buyer Financial Statements in accordance with GAAP);
(b) Encumbrances to secure obligations to landlords, lessors or renters under leases or rental agreements; (c) deposits or pledges made in connection with, or to secure payment of, workers’ compensation, unemployment insurance or
similar programs mandated by Applicable Law; (d) Encumbrances disclosed on the Buyer Financial Statements; (e) materialmen’s, mechanics’, carriers’, workmen’s and repairmen’s Encumbrances and other similar
Encumbrances arising in the Ordinary Course of Business securing obligations; (f) Encumbrances which do not materially detract from the value or materially interfere with any present or intended use of such property or assets; (g) any
other Encumbrances which individually or in the aggregate would not materially adversely affect the use of the property or asset subject thereto.
“Buyer Preferred Stock” means Buyer’s preferred stock, par value $0.001 per share.
“Buyer Prepared Return” has the meaning set forth in Section 11.1(b).
“Buyer Securities” means the Buyer Common Stock and the Buyer Preferred Stock, collectively.
“Buyer Stockholders” means the record holders of Buyer Securities.
“Clinical Trial” means any human clinical trial of any Company Product or Service.
“Closing” means the consummation of the IT Sale.
“Closing Adjustment Shares” means the number of shares of Buyer Common Stock, rounded up to the nearest whole share, equal to
(a) the Closing Adjustment Value, divided by (b) Buyer Common Stock Price.
“Closing Adjustment Value”
means an amount equal to (i) Closing Debt, plus (ii) the Closing Company Transaction Expenses, plus (iii) the Closing NWC Adjustment Amount; provided that if the foregoing sum results in an amount that is a
negative number, the Closing Adjustment Value shall equal zero.
“Closing Cash” means, as of the Reference Time, the
aggregate sum of (i) the Company Group’s unrestricted cash and cash equivalents, wherever held anywhere in the world, term deposits, guaranteed investment certificates and similar readily liquid instruments and deposits in transit
(including checks received by the Company Group prior to the Reference Time), but not taking into consideration any issued but uncleared checks, plus (ii) an amount equal to the amount of value-added Taxes with respect to which, prior to
the Closing Date, the Company has filed a reimbursement request with the government of Italy that the Parties expect will result in a repayment of such value-added Taxes to the Company within ninety (90) days following the Closing Date minus
(iii) the amount of any proceeds from any Debt of the Company Group other than the EIB Loan.
4
“Closing Company Shares” means the shares of Company Stock issued and
outstanding immediately prior to the Closing.
“Closing Company Transaction Expenses” means all Transaction Expenses of
the Company Group which are unpaid as of the Reference Time and which are either (a) in excess of $4,203,000 in the aggregate and were not approved in writing by Buyer prior to their occurrence or (b) with respect to any Transaction
Expense listed in Section 9.2(d)(xvii), clauses (x)-(z), is in excess of the applicable Transaction Expense Threshold.
“Closing Date” means a time and date on which the Closing shall occur, which shall be no later than the second Business Day
after the satisfaction or waiver of the conditions set forth in Article IX, or at such other time, date and location as the Parties agree in writing.
“Closing Debt” means all Debt of the Company Group as of the Reference Time.
“Closing Pro Rata Portion” means, with respect to a Shareholder, the percentage set forth opposite such Shareholder’s
name on Schedule 1 of the Company Shareholder Letter.
“Closing NWC Adjustment Amount” means the amount by which the
Company Net Working Capital is less than the Closing NWC Target.
“Closing NWC Target” means negative €850,000,
which number shall decrease by €16,667 for each day after September 1, 2023 until and including the Closing Date, provided, that, the Company has operated in the Ordinary Course of Business and in compliance with
Section 7.3(s) and Section 7.7 from the date of this Agreement through such day. For example, if the Closing occurs on September 16, 2023, the Closing NWC Target would be negative
€1,100,005. For the avoidance of doubt, the Closing NWC Target shall not include any of the US Subsidiary Consideration.
“Closing Shares” means the number of shares of Buyer Common Stock equal to (i) the Base Share Consideration minus
(ii) the Closing Adjustment Shares.
“Code” means the U.S. Internal Revenue Code of 1986, as amended.
“Company” has the meaning set forth in the Preamble.
“Company Ancillary Agreements” means, collectively, each certificate to be delivered on behalf of Company by an officer or
officers of Company at the Closing pursuant to Article IX and each agreement or document (other than this Agreement) that Company or a Shareholder is to enter into as a party thereto pursuant to this Agreement.
“Company Balance Sheet” means the Company Group’s consolidated pro forma unaudited balance sheet as of the Balance Sheet
Date included in the Company Financial Statements.
“Company Benefit Arrangements” has the meaning set forth in
Section 3.17(d).
“Company Business” means the business of Company and the US Subsidiary as
presently conducted.
“Company Charter Documents” has the meaning set forth in Section 3.8(b).
5
“Company Disclosure Schedule” means the disclosure schedule dated as of the
Agreement Date and delivered by Company to Buyer on the Agreement Date arranged in sections corresponding to the numbered and letter sections contained in Article III of this Agreement.
“Company Financial Statements” means (a) the Company Group’s consolidated pro forma audited balance sheet as of
December 31, 2022; (b) the Company Group’s consolidated pro forma audited statement of income for the twelve-months ended December 31, 2022; (c) the Company Group’s consolidated pro forma audited statement of cash flows for
the twelve-months ended December 31, 2022; and (d) the Company Group’s unaudited balance sheet as of May 31, 2023 and the unaudited statements of income and cash flows for the month then ended..
“Company Fundamental Representations” means those representations and warranties set forth in
Section 3.1 (Organization and Good Standing), Sections 3.2 (Subsidiaries), Section 3.3 (Power, Authorization and Validity), Section 3.4
(Capitalization of Company), Section 3.7 (Taxes), Section 3.16 (Certain Transactions and Agreements), Section 3.18(a) (Transaction Expenses),
Section 4.1 (Power, Authorization and Validity), Section 4.2 (Title to Company Stock), Section 4.3 (Brokerage) and
Section 4.5 (Transaction Securities).
“Company Group” means Company and the US
Subsidiary, taken as a whole, and each individually is sometimes referred to herein as a “Company Group Member”.
“Company Material Contract” means any Contract required to be listed on the Company Disclosure Schedule pursuant to
Section 3.11.
“Company Net Working Capital” means the sum of the current assets of the Company
less the sum of the current liabilities of the Company, each determined in accordance with the Accounting Principles, measured as of the Reference Time. For purposes of calculating Company Net Working Capital: (a) the Company’s current
assets will include Closing Cash and exclude the VAT Credit, the R&D Tax Credit, and any other Tax assets and (b) the Company’s current liabilities will exclude all Closing Debt and all Closing Company Transaction Expenses. For the
avoidance of doubt, the Company Net Working Capital shall not include any amounts received in respect of the of US Subsidiary Consideration.
“Company Option” means an option to purchase a share of Company Stock issued pursuant to the Company Stock Plan or otherwise.
“Company Owned IP” means all Intellectual Property in which any Company Group Member has or purports to have an
ownership interest (whether exclusively or jointly with any other Person(s)), or that is exclusively licensed to the Company Group by any other Person.
“Company Permitted Encumbrances” means, with respect to the Company, (a) Encumbrances for Taxes or assessments
and similar governmental charges or levies that are either (i) not yet due and payable or (ii) being contested in good faith (and for which adequate accruals or reserves have been established on the Company Financial Statements in
accordance with the Accounting Principles); (b) Encumbrances to secure obligations to landlords, lessors or renters under leases or rental agreements and Encumbrances created by others upon properties over which there are easements, rights-of-way, licenses or other rights of user in favor of the leased premises of the Company Group; (c) deposits or pledges made in connection with, or to secure
payment of, workers’ compensation, unemployment insurance or similar programs mandated by Applicable Law; (d) Encumbrances disclosed on the Company Financial Statements; (e) materialmen’s, mechanics’, carriers’,
workmen’s and repairmen’s Encumbrances and other similar Encumbrances arising in the Ordinary Course of Business securing obligations; (f) Encumbrances which do not materially detract from the value or materially interfere with any
present or intended use of such property or assets; and (g) any other Encumbrances which individually or in the aggregate would not materially adversely affect the use of the property or asset subject thereto.
6
“Company Prepared Return” has the meaning set forth in
Section 11.1(a).
“Company Product or Service” means each of the current products and services
being designed, developed, produced, manufactured, assembled, sold, leased, licensed, marketed, distributed or otherwise exploited by the Company Group as of the Agreement Date that are material to the Company Business.
“Company Registered IP” means each item of Company Owned IP that is Registered IP.
“Company Shareholder Letter” means certain schedules dated as of the Agreement Date and delivered by Company to Buyer on the
Agreement Date with respect to the Shareholders.
“Company Scientific Material” means, collectively, all material
research, development, clinical, regulatory or other scientific data, information or documentation (including databases, data packages, reports and filings and any Patent data, records, lab notebooks and other similar documentation or materials), in
each case, solely to the extent reduced to writing or stored electronically, whether created or otherwise generated by, or for or otherwise on behalf of, the Company Group.
“Company Stock” means the entire outstanding capital stock of the Company, including the Class A Shares, Class B
Shares, Class C Shares, Class D Shares, Class E Shares, and Class F Shares.
“Company Stock Plan”
means the Company Stock Option Plan 2018 – 2028, regulated by the board of directors of the Company on October 30, 2018, on the basis of the guidelines approved by the extraordinary shareholders’ meeting on April 9, 2018, as
amended from time to time.
“Confidentiality Agreement” has the meaning set forth in
Section 7.6.
“Contract” means any written or oral legally binding contract, agreement,
instrument, arrangement, commitment or undertaking (including leases, licenses, mortgages, notes, guarantees, sublicenses, subcontracts and purchase orders).
“Convertible Notes” means the (i) notes issued pursuant to that certain investment agreement, by and among the Company
and the other parties thereto, dated as of June 30, 2021, as amended on December 31, 2021, March 31, 2022, September 14, 2022 and December 29, 2022 and (ii) the notes issued pursuant to that certain investment
agreement, by and among the Company and the other parties thereto, dated as of February 17, 2023.
“Convertible
Noteholders” shall mean each holder of the Convertible Notes.
“Debt” means the outstanding principal amount of,
and all interest, fees, prepayment premiums, expenses, breakage costs, indemnities, penalties or other amounts accrued in respect of and all amounts otherwise owing or payable at retirement of, (a) any indebtedness for borrowed money of the
Company, (b) any obligation of the Company evidenced by bonds, debentures, notes, mortgages, security arrangements, indentures or other similar instruments, (c) any reimbursement obligation of the Company with respect to letters of credit
(including standby letters of credit to the extent drawn upon), bankers’ acceptances or similar facilities issued for the account of the Company, (d) all obligations of the Company as lessee under leases that are required to be recorded as
capital leases in accordance with GAAP or Italian GAAP, (e) all liabilities or obligations of the Company under any interest rate, currency, swap or other hedging agreements, (f) all liabilities or obligations of the Company for the
deferred purchase price of
7
property or services (including all earn-out or similar obligations), (g) all Unpaid Pre-Closing Taxes, and
(h) any obligation of the type referred to in clauses (a) through (g) of another Person, the payment of which the Company has guaranteed or for which the Company is responsible or liable, directly or indirectly, jointly or severally, as
obligor or guarantor; provided, however that the EIB Loan shall be excluded from Debt for purposes of this Agreement, up to €10,000,000 in principal (plus interest accrued thereon prior to the Closing Date); provided, further, that any amount
drawn on the EIB Loan in excess of €10,000,000 shall constitute Debt for purposes of this Agreement. For clarity, Debt shall not include any (i) Transaction Expenses or (ii) liabilities included in Company Net Working Capital.
“DGCL” means the Delaware General Corporation Law.
“Divestiture” or “Divest” means any transaction in which any EryDex Product are divested or transferred by
Buyer to a Third Party after the Closing by any means, including by way of merger, consolidation, asset acquisition or sale, license, sublicense, purchase, sale or assignment.
“EIB Loan” means that certain line of credit in the amount of €30,000,000, pursuant to the Finance Contract between the
Company and the European Investment Bank dated as of July 24, 2020, of which the Company represents that €10,000,000 has been drawn as of the date of this Agreement.
“EMA” means the European Medicines Agency or any successor agency having substantially the same functions or, if the mutual
recognition procedure is used for the Company Product or Service in the European Union, any governmental authority having the authority to regulate the sale of medicinal or pharmaceutical products in any country in the European Union.
“Encumbrance” means, with respect to any asset, any mortgage, deed of trust, lien, pledge, charge, security interest, title
retention device, collateral assignment, adverse claim, restriction or other encumbrance of any kind in respect of such asset (including any restriction on the voting of any security, any restriction on the Transfer of any security or other asset,
any restriction on the receipt of any income derived from any asset, any restriction on the use of any asset and any restriction on the possession, exercise or Transfer of any other attribute of ownership of any asset). For purposes of
clarification only, an inability to sell a security without registering such security for sale under the Securities Act or other securities laws of any jurisdiction or other restrictions arising from applicable securities laws of any jurisdiction
shall not represent an Encumbrance.
“Enforceability Exceptions” has the meaning set forth in
Section 3.3(b).
“Environmental Law” has the meaning set forth in
Section 3.20(b).
“ERISA” means the U.S. Employee Retirement Income Security Act of 1974, as
amended.
“EryDex Product” means those products developed by the Company or the US Subsidiary prior to the Agreement Date
that incorporate one or more of the inventions patented as USPN 10,849,858 or USPN 9,089,640 and (A) a multi-use red cell loader device (RCL), (B) an EryKit (single use) disposable device,
(C) dexamethasone pro-drug (DSP solution), or (D) RBC encapsulation.
“Estimated
Closing Statement” has the meaning set forth in Section 2.4(a).
“European Commission”
means the European Union’s executive arm responsible for drawing up proposals for new European Union legislation, and implementation of the decisions of the European Parliament and the Council of the European Union.
8
“Exchange Act” means the U.S. Securities Exchange Act of 1934, as amended.
“Expiration Date” has the meaning set forth in Section 6.1(a).
“Export Control Laws” means (i) all U.S. import and export laws (including those laws under the authority of U.S.
Departments of Commerce (Bureau of Industry and Security) codified at 15 C.F.R., Parts 700-799; Homeland Security (Customs and Border Protection) codified at 19 C.F.R., Parts 1-199; State (Directorate of Defense Trade Controls) codified at
22 C.F.R., Parts 103, 120-130; and Treasury (Office of Foreign Assets Control) codified at 31 C.F.R., Parts 500-599) and (ii) all comparable Applicable Laws outside the United States.
“FDA” means the United States Food and Drug Administration, or any successor agency thereto.
“FDCA” means the Federal Food, Drug and Cosmetic Act, as amended, and all related rules, regulations and guidelines.
“Fraud” shall mean fraud as defined under Delaware law.
“GAAP” means U.S. generally accepted accounting principles.
“Golden Power Authority” means the presidency of the Italian Council of Ministries (Presidenza del Consiglio dei
Ministri) and/or any other office, department or branch of the Italian Government which is competent, under the Golden Power Law, to issue and release the Golden Power Clearance under the Golden Power Law.
“Golden Power Clearance” means the unconditional clearance, whether express or implied (due to expiry of the applicable
review period, to the extent that it equals to a clearance pursuant to the Golden Power Law), from the Golden Power Authority under the Golden Power Law to complete the steps and elements of the acquisition of 100% of the Company’s share
capital that might be relevant under the Golden Power Law, as applicable, it being comprised (but not limited to) by way of a resolution by the Golden Power Authority not to exercise its special powers, or, alternatively, the adoption of a
resolution by the Golden Power Authority excluding the application of the Golden Power Law, adopted either at the end of the procedure set forth in the Golden Power Pre-Filing or in the Golden Power Filing.
“Golden Power Filing” means the application for the Golden Power Clearance provided for by Article 2, par. 5, of Italian
Law Decree No. 21 dated 15 March 2012 converted in law no. 56 dated 11 May 2012, as amended.
“Golden Power
Law” means, in aggregate, Italian Law Decree No. 21 dated 15 March 2012 converted in law no. 56 dated 11 May 2012, Law Decree No. 105 dated 21 September 2019 converted in Law No. 133 dated 18 November
2019, as subsequently amended and supplemented, including (without limitation) by Law Decree No. 23 dated 8 April 2020, converted with amendments by Law of 5 July no. 40, and by Law Decree No. 21 of 21 March 2022, converted
with amendments by Law of 20 May 2022, no. 51, Regulation (EU) 2019/452, together with all connected or subordinated implementing decrees and regulations, including (without limitation) Prime Ministerial Decree No. 133 dated 1 August
2022.
“Good Clinical Practices” means the then current standards for clinical trials for pharmaceuticals and medical
devices, as applicable, as set forth in the FDCA and applicable regulations promulgated thereunder (including, for example, 21 C.F.R. Parts 50, 54, and 56), as amended from time to time, and such standards of good clinical practice (including all
applicable requirements relating to protection of human subjects) as are required by any other applicable organization or Governmental Authority.
9
“Good Laboratory Practices” means the then current standards for
laboratories developing pharmaceuticals and medical devices, as applicable, as set forth in the FDCA and applicable regulations promulgated thereunder, as amended from time to time, including applicable requirements contained in 21 C.F.R. Part 58,
and such standards of good laboratory practices as are required by any other applicable organization or Governmental Authority.
“Good Manufacturing Practices” means, the requirements for current good manufacturing practices as set forth in the FDCA and
applicable regulations promulgated thereunder, as amended from time to time, including the applicable requirements contained in 21 C.F.R. Parts 210, 211, 820, and 1271, as applicable, and such standards of current good manufacturing practices as are
required by any other applicable organization or Governmental Authority.
“Governmental Authority” means any court or
tribunal, supranational or national governmental or regulatory body, administrative agency, ministries, commission or other governmental authority, including the Golden Power Authority and the Antitrust Authority or Notified Body.
“Governmental Permits” has the meaning set forth in Section 3.14(b).
“Healthcare Laws” means all Applicable Laws relating to healthcare regulatory matters or the development, testing, research
(including nonclinical and clinical research or studies), manufacture, production, analysis, distribution, approval, importation, exportation, use, handling, quality, packaging, labeling, sale or promotion of any device, drug or biological product
(including any ingredient or component of the foregoing products), including the FDCA, the Public Health Service Act (42 U.S.C. §§ 201 et seq.) or comparable foreign laws, all laws relating to any federal health care program (as such term
is defined in 42 U.S.C. § 1320a-7b(f)), including the federal Anti-Kickback Statute (42 U.S.C. § 1320a-7b(b)), the Physician Self-Referral Law (42 U.S.C.
§ 1395nn) or comparable foreign laws, the civil False Claims Act (31 U.S.C. § 3729 et seq.), the administrative False Claims Law (42 U.S.C. § 1320a-7b(a)), Sections 1320a-7, 1320a-7a, and 1320a-7b of Title 42 of the United States Code and any comparable self-referral or fraud and abuse laws
promulgated by any Governmental Authority, the 21st Century Cures Act (Pub. L. 114-255), the health care fraud criminal provisions under the Health Insurance Portability and Accountability Act of 1996
(“HIPAA”) (42 U.S.C. § 1320d et seq.), HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009 (42 U.S.C. § 17921 et seq.), and any state or federal or comparable foreign law the
purpose of which is to protect the privacy of individually identifiable patient information, Medicare (Title XVIII of the Social Security Act) and Medicaid (Title XIX of the Social Security Act), the Affordable Care Act, as amended by the Health
Care and Education Affordability Reconciliation Act of 2010, TRICARE (10 U.S.C. Section 1071 et seq.) or comparable foreign law, the U.S. Physician Payments Sunshine Act (42 U.S.C. § 1320a-7h) and
similar state or foreign laws related to the reporting of manufacturer payments or transfers of value to health care professionals, in each case including the associated rules and regulations promulgated thereunder and all of their foreign
equivalents, and any other requirements of Applicable Laws relating to the Company Business.
“Holdback Release Date” has
the meaning set forth in Section 6.4.
“Holdback Shares” means the shares of Buyer Common Stock
which shall, to the extent not forfeited or withheld in accordance with Article VI, be issued to the Shareholders in accordance with their Pro Rata Portion, in an aggregate number of shares (prior to any deduction or forfeiture in accordance
with Article VI) equal to ten percent (10%) of the Closing Shares, rounded up to the nearest whole share.
“Indemnity
Pro Rata Portion” means, with respect to a Shareholder, the portion equal to (i) the Total Consideration issued to such Shareholder as of the time of the determination of the amount of indemnifiable Losses owed by the Shareholders
under Article VI (the “Determination Time”), divided by (ii) the Total Consideration issued to all Shareholders as of that Determination Time.
10
“Intellectual Property” or “IP” means all intellectual
property, including the following items of intangible property, and all rights associated therewith in any jurisdiction and tangible embodiments thereof:
(a) Patents;
(b) trademarks,
trade names, service marks, designs, logos, trade dress, and trade styles and other indicia of origin, whether or not registered, and all registrations and pending applications for registration of the same and renewals thereof and all goodwill
associated therewith;
(c) works of authorship, copyrights, whether or not registered, and all registrations and pending applications for
registration of the same and renewals thereof and database rights;
(d) domain names and URLs;
(e) all technology, technical information, know-how and data, including, without limitation,
inventions (whether or not patentable of reduced to practice), improvements, discoveries, trade secrets, specifications, instructions, ideas, processes, methods, formulations, formulae, protocols, materials, assays, screens, algorithms, models,
databases, expertise and other technology applicable to formulations, compositions or products or to their manufacture, development, registration, use or marketing or to methods of assaying or testing them or processes for their manufacture,
formulations containing them or compositions incorporating or comprising them, and including all biological, chemical, pharmacological, biochemical, toxicological, pharmaceutical, physical and analytical, safety, quality control, manufacturing,
nonclinical, preclinical and clinical data, regulatory data and filings, instructions, processes, formulae, expertise and information, relevant to the research, development, manufacture, use, importation, offering for sale or sale of, and/or which
may be useful in studying, testing, developing, producing or formulating, products, or intermediates for the synthesis thereof and chemistry, manufacturing and control information and data, lab notebooks, Patent data and records, stability,
technology, test and other data and results; and
(f) computer programs, including, without limitation, computer programs embodied in
semiconductor chips or otherwise embodied, and related flow-charts, programmer notes, updates and data, whether in object or source code form.
“Interim Period” has the meaning set forth in Section 7.1.
“IRS” means the U.S. Internal Revenue Service.
“IT Buyer” has the meaning set forth in the Preamble.
“IT Sale” has the meaning set forth in Section 2.1(a)(ii).
“Italian Civil Code” means the Italian civil code, approved by Royal Decree No. 262 of March 16, 1942, as amended
from time to time.
“Italian GAAP” means accounting principles generally accepted in Italy.
11
“Italian Notary Public” means Andrea De Costa of Studio Marchetti, with
office in 20121 – Milan, Via Agnello 18, or any other notary public in Milan designated by the Buyer before the Closing Date.
“Key Employees” means Guenter Janhofer, Thomas Sabia and Giovanni Mambrini.
“knowledge” means, with respect to the Company and the Company Group, the actual knowledge of Luca Benatti, Guenter Janhofer,
Thomas Sabia and Giovanni Mambrini after reasonable inquiry.
“Legal Proceeding” means any action, suit, litigation,
arbitration, proceeding (including any civil, criminal, administrative, investigative or appellate proceeding), hearing, inquiry, audit, examination or investigation commenced, brought, conducted or heard by or before, or otherwise involving, any
court or other Governmental Authority or any arbitrator or arbitration panel.
“Licensee Party” means any Third Party to
whom a Buyer Party has licensed or sublicensed an EryDel Product and which Third Party still holds such license or sublicense, after Closing and before the earliest of (i) the 7th anniversary
of Closing, if Milestone 4 was not achieved prior to such anniversary, (ii) the 11th anniversary of Closing, if Milestone 4 was achieved prior to the 7th anniversary of Closing, and (iii) Reversion.
“Loss” and
“Losses” shall mean any debts, obligations and other liabilities (whether absolute or contingent, liquidated or unliquidated, due or to become due, accrued or not accrued), losses, damages, Taxes, deficiencies, judgments,
assessments, fines, fees, penalties, expenses (including amounts paid in settlement, interest, court costs, costs of investigators, fees and expenses of attorneys, accountants, financial advisors, consultants and other experts, and other expenses of
litigation), punitive damages payable to third parties, and incidental or consequential damages, in each case that may be imposed or otherwise incurred or suffered.
“made available” or “delivered” shall mean that the Company has posted such materials to the virtual data
room provided by CapLinked titled “EryDel Workspace” and the virtual data room provided by Sharepoint titled “erydel.sharepoint.com” and made available or delivered to Buyer and its Representatives during the negotiation of this
Agreement (the “Data Room”), but only if so posted and made available in the folder related to such document’s subject matter, on or prior to the date that is two (2) Business Days prior to the date of this Agreement.
“Material Adverse Effect” when used in connection with any specified Person(s) means any change, event, effect or occurrence
that has had or would be reasonably likely to, individually or in the aggregate, (a) have a material adverse effect upon the financial condition, assets (including intangible assets), liabilities, business or results of operations of such
Person and its Subsidiaries, taken as a whole, or (b) prevent or materially impair or delay the ability of such Person to consummate the Transactions or to perform its obligations under this Agreement or any Ancillary Agreement; provided,
however, that for purposes of clause (a) above, any changes, events, effects or occurrences directly or indirectly attributable to, resulting from, relating to or arising out of any of the following (by themselves or when aggregated with any
other changes, events, effects or occurrences) shall not be deemed to be, constitute or taken into account when determining whether there has or may, would or could have occurred a Material Adverse Effect: (i) the effect of any actions taken or
not taken by any Company Group Member at the direction or request of Buyer or expressly required by this Agreement; (ii) changes affecting any of the industries in which such Person or any of its Subsidiaries operates or the U.S. or worldwide
economy, financial or securities markets or political environment generally, including changes to currency exchange rates; (iii) any change in any Applicable Law, government programs, or any interpretation thereof; (iv) any change in
Italian GAAP, GAAP or other applicable accounting standards; (v) the announcement or expected completion of this
12
Agreement or the Transactions; (vi) conditions caused by acts of God, terrorism or war (whether or not declared), sabotage, natural disasters, pandemics, geological or meteorological events,
or other calamity, crisis or geopolitical event occurring after the Agreement Date or any material worsening of such conditions threatened or existing as of the Agreement Date; (vii) any failure in and of itself by such Person and its
Subsidiaries to meet any internal or published budgets, projections, forecasts or predictions of financial performance for any period (provided that the underlying cause of any such failure may be considered in determining whether a Material Adverse
Effect has occurred or would be reasonably likely to occur to the extent not included by another exception herein); and (viii) any of the matters listed in the Company Disclosure Schedule or the Buyer Disclosure Schedule; provided further,
however, that any change, event, effect or occurrence referred to in clauses (ii), (iv) or (vi) above shall be taken into account in determining whether a Material Adverse Effect has occurred or would be reasonably likely to occur to the extent
that such change, event, effect or occurrence has a disproportionate and adverse effect on such Person and its Subsidiaries, taken as a whole, compared to other participants in the industries in which such Person and its Subsidiaries primarily
conducts its business.
“Materials of Environmental Concern” has the meaning set forth in
Section 3.20(b).
“Milestone Payments” has the meaning set forth in
Section 2.7(a).
“Milestone Pro Rata Portion” means, with respect to a Shareholder, the
percentage set forth opposite such Shareholder’s name on Schedule 2 of the Company Shareholder Letter for the applicable Milestone Payment.
“Nasdaq” means the Nasdaq Global Select Market.
“Net Closing Shares” means (i) the Closing Shares minus (ii) the Holdback Shares.
“Notified Body” means an organization designated by a Governmental Authority of an EEA country to conduct conformity
assessments to relevant Applicable Laws.
“OFAC” has the meaning set forth in Section 3.15(c).
“Optionholder” means each holder of Company Options.
“Ordinary Course of Business” means the ordinary course of business consistent with past practice.
“Outside Date” has the meaning set forth in Section 10.2(b).
“Pandemic Response Laws” means the Coronavirus Aid, Relief, and Economic Security Act, the Families First Coronavirus
Response Act, the COVID-related Tax Relief Act of 2020, the Presidential Memorandum on Deferring Payroll Tax Obligations in Light of the Ongoing COVID-19 Disaster (as issued on August 8, 2020, and
including any administrative or other guidance published with respect thereto by any Governmental Authority (including IRS Notice 2020-65)), and any other similar or additional U.S. federal, state, or local or
non-U.S. Applicable Law, or administrative guidance intended to benefit taxpayers in response to the COVID-19 pandemic and associated economic downturn.
“Patents” means all patents and patent applications (including provisional applications) and patent disclosures, and
including all divisionals, continuations, substitutions, continuations-in-part, re-examinations,
re-issues, additions, renewals, extensions, confirmations, registrations, any confirmation patent or registration patent or patent of addition based on any such patent, patent term extensions, and supplemental
protection certificates or requests for continued examinations, foreign counterparts, and the like of any of the foregoing.
13
“Person” means any individual, corporation, company, limited liability
company, partnership, limited liability partnership, trust, estate, proprietorship, joint venture, association, organization, entity or Governmental Authority.
“Pre-Closing Tax Period” means any taxable period (or portion thereof) that ends on
or before the Closing Date.
“Public Certifications” has the meaning given such term in
Section 5.7(a).
“R&D Tax Credit” means the Italian corporate income tax credit for
research and development activities provided by Italian Law n. 145 of 23 December 2013, as amended and supplemented from time to time.
“Reference Time” means immediately prior to the Closing on the Closing Date (but without giving effect to the Transactions,
including any payments by Buyer hereunder to occur at the Closing) and, in the case of Taxes, as of the close of business on the Closing Date.
“Registered IP” means all Intellectual Property that is registered, filed, or issued under the authority of any Governmental
Authority, including all Patents, registered copyrights, registered mask works, and registered trademarks and all applications for any of the foregoing.
“Regulatory Authority” means any national or supranational Governmental Authority, including the FDA or the European
Commission, with responsibility for granting any license, registrations or Approvals with respect to the Company Product or Service.
“Related Party” has the meaning set out in IAS 24.
“Release Agreements” has the meaning set forth in Section 7.10.
“Representative Losses” has the meaning given such term in Section 6.9(c).
“Representatives” means, as to any Person, such Person’s Affiliates and the respective managers, directors, officers,
employees, independent contractors, consultants, advisors (including financial advisors, counsel and accountants), agents and other legal representatives of such Person or its Affiliates.
“Sale” has the meaning set forth in Section 2.1(a)(ii).
“SEC” means the U.S. Securities and Exchange Commission.
“SEC Documents” means, with respect to any Person, each report, schedule, form, statement, registration statement, prospectus
or other document filed or required to be filed or furnished with the SEC by such Person under the Securities Act or Exchange Act, together with any amendments, restatements or supplements thereto.
“SEC Reports” has the meaning given such term in Section 5.7(a).
“Securities Act” means the U.S. Securities Act of 1933, as amended.
“Share Deed of Transfer” has the meaning set forth in Section 2.2.
14
“SOX” means the U.S. Sarbanes-Oxley Act of 2002, as amended.
“Straddle Period” means any taxable period that begins on or before and ends after the Closing Date.
“Subsidiary” means with respect to any Person, a corporation, partnership, limited liability company or other business entity
in which such Person owns, directly or indirectly, at least a 50% of the total voting power of shares of stock or other equity interests thereof or that is otherwise, directly or indirectly, controlled by such Person.
“Subsidiary Charter Documents” has the meaning set forth in Section 3.8(b).
“Tax” (and, with correlative meaning, “Taxes”) means any and all (a) U.S. federal, state, and local and
non-U.S. taxes, assessments, and other governmental charges, duties (including custom duty and stamp duty), impositions, and liabilities, including net income, alternative or
add-on minimum, gross income, gross receipts, sales, use, goods and services, harmonized sales, capital, unemployed occupation, workers’ compensation, value added, ad valorem, Transfer, indirect Transfer,
franchise, profits, license, withholding, payroll, recapture, employment, escheat, social security, national health insurance, excise, severance, stamp, occupation, premium, property, environmental, windfall profit tax, or other tax, governmental
fee, or other like assessment or charge of any kind whatsoever, together with any interest or any penalty, addition to tax, or additional amount imposed by any Governmental Authority, (b) any liability for the payment of any amounts of the type
described in clause (a) as a result of being a member of an affiliated, consolidated, combined, unitary or similar group (including any arrangement for group or consortium relief or similar arrangement) for any period, and (c) any
liability for the payment of any amounts of the type described in clauses (a) or (b) as a result of any express or implied obligation to indemnify any other Person or as a result of any obligation under any agreement or arrangement with any
other Person with respect to such amounts and including any liability for taxes of a predecessor or Transferor or otherwise by operation of Applicable Law.
“Tax Representations” mean the representations and warranties set forth in Section 3.7.
“Tax Return” means any return, declaration, notice, statement, claim for refund, report, or form (including estimated Tax
returns and reports, withholding Tax returns and reports, any schedule or attachment, and information returns and reports) or other document filed or required to be filed with respect to Taxes and including all amendments or supplements thereof.
“Third Party” means, with respect to any party, any Person other than such party or an Affiliate of such party.
“Total Consideration” has the meaning set forth in Section 2.2.
“Transaction Compensation Payments” means any change of control, severance (including, without limitation, any
severance payable to Luca Benatti) or transaction bonus payments payable to any current or former employee, officer, director or independent contractor of Company or the US Subsidiary in connection with the consummation of the Transactions.
“Transaction Expenses” means the aggregate fees, costs and expenses incurred by or on behalf of the Company Group, the Holder
Representative or their respective Representatives to the extent incurred in connection with the negotiation and execution of this Agreement, any Ancillary Agreement hereto or the consummation of the Transactions, including (a) all fees, costs
and expenses of any legal counsel, accounting advisor, Tax advisor, consultant or other third party advisor for services rendered in relation to the Transactions and (b) Transaction Compensation Payments.
15
“Transaction Payroll Taxes” means the employer’s portion of any
employment, payroll, or similar Taxes with respect to any payments in respect of Company Stock (to the extent applicable), payments in respect of Company Options, any Transaction Compensation Payments, and any bonus, severance, change-in-control, or other compensatory payments in connection with the Transactions, in each case, paid at, prior to, or following the Closing Date, whether payable by
Buyer, IT Buyer any Company Group Member, or any of their respective Affiliates.
“Transaction Securities” means the
Closing Shares, to the extent issued to the Shareholders under Section 2.2.
“Transactions”
means, collectively, the transactions contemplated by this Agreement and the Ancillary Agreements.
“Transfer” means,
with respect to any Transaction Security, (a) the sale of, offer to sell, contract or agreement to sell, hypothecate, pledge, grant of any option to purchase or otherwise dispose of or agreement to dispose of, directly or indirectly, or
establishment or increase of a put equivalent position or liquidation with respect to or decrease of a call equivalent position within the meaning of Section 16 of the Exchange Act and the rules and regulations promulgated thereunder with
respect to such Transaction Security, (b) the entry into any swap or other arrangement that transfers to another, in whole or in part, any of the economic consequences of ownership of any Transaction Security, whether any such transaction is to
be settled by delivery of such Transaction Securities, in cash or otherwise, or (c) the public announcement of any intention to effect any transaction specified in clause (a) or (b).
“Transfer Taxes” means any Transfer, indirect Transfer, stamp, documentary, sales, use, registration, goods and services,
harmonized sales, recording, filing, value-added, and other similar Taxes incurred in connection with the Transactions.
“Treasury
Regulations” means the regulations promulgated under the Code.
“Unpaid
Pre-Closing Taxes” means any and all (a) Taxes of any Company Group Member relating or attributable to any Pre-Closing Tax Period (including, for the
avoidance of doubt, any such Taxes that are deferred to a taxable period (or portion thereof) beginning after the Closing Date pursuant to Pandemic Response Laws or Sections 451(c) or 481 of the Code (or any corresponding or similar provision of
state, local, or non-U.S. Applicable Law), (b) Taxes of any member of an affiliated, consolidated, combined, unitary, or similar group (including any arrangement for group or consortium relief or similar
arrangement) of which any Company Group Member (or any predecessor of such Company Group Member) is or was a member on or prior to the Closing Date, including pursuant to Treasury Regulations
Section 1.1502-6 (or any corresponding or similar provision of state, local, or non-U.S. Applicable Law), (c) Taxes of any Person imposed on any Company Group
Member as a transferee or successor, by Contract, by operation of Applicable Law, or otherwise, which Taxes relate to an event or transaction occurring before the Closing, and (d) Taxes relating or attributable to the Transactions, including
Transaction Payroll Taxes, withholding Taxes, and Transfer Taxes. For purposes of clause (a), in the case of Taxes based on income, sales, proceeds, profits, receipts, wages, compensation, or similar items and all other Taxes that are not imposed on
a periodic basis, the amount of such Taxes through the Closing Date for a Straddle Period shall be deemed to be the amount that would be payable if the taxable period ended at the end of the day on the Closing Date based on an interim closing of the
books as of the end of such day, except that exemptions, allowances, or deductions that are calculated on an annual basis (including depreciation and amortization deductions, other than with respect to property placed in service after the Closing)
shall be
16
allocated on a per diem basis; and in the case of any other Taxes that are imposed on a periodic basis for a Straddle Period, the amount of such Taxes through the Closing Date shall be the amount
of such Taxes for such taxable period (or, in the case of such Taxes determined on an arrears basis, the amount of such Taxes for the immediately preceding taxable period) multiplied by a fraction the numerator of which shall be the number of
calendar days from the beginning of such taxable period up to and including the Closing Date and the denominator of which shall be the number of calendar days in the entire Straddle Period.
“US Sale” has the meaning set forth in Section 2.1(a)(i).
“US Subsidiary” means EryDel USA Inc., a Delaware corporation.
“US Subsidiary Consideration” means $28,000.
“US Subsidiary Stock” means all of the issued and outstanding capital stock of the US Subsidiary.
“VAT” means (a) any tax imposed in compliance with the Council Directive of 28 November 2006 on the common system
of value added tax (EC Directive 2006/112) (including, in relation to Italy, value added tax imposed by Presidential Decree No. 633 of 26 October 1972 and Legislative Decree No. 331 of 30 August 1993); and (b) any other tax
of a similar nature in substitution for, or levied in addition to, such tax referred to in (a) above.
“VAT Credit”
means the excess of output VAT available to the Company to be either set-off or asked for refund.
ARTICLE II
THE
TRANSACTIONS
2.1 Sale and Purchase of Company Stock.
(a) Upon the terms and subject to the conditions set forth in this Agreement:
(i) On the Closing Date and immediately prior to the Closing, Buyer shall purchase and accept from the Company, and the Company shall sell,
Transfer and convey to Buyer, all of the US Subsidiary Stock free and clear of all Encumbrances together with all the rights, title and interest attached to the US Subsidiary Stock, such that all of the issued and outstanding shares of US Subsidiary
Stock (which represent 100% of the equity interests of the US Subsidiary as of immediately prior to Closing), shall be sold to and purchased by Buyer immediately prior to the Closing (the “US Sale”); and
(ii) At the Closing, and immediately following the US Sale, IT Buyer shall purchase and accept from the Shareholders, and each Shareholder
shall sell, Transfer, assign and convey to IT Buyer, the number of shares of Company Stock set forth opposite such Shareholder’s name on Schedule 1 of the Company Shareholder Letter free and clear of all Encumbrances together with all the
rights, title and interest attached to the Company Stock, such that all of the issued and outstanding shares of Company Stock at the Closing shall be sold to and purchased by Buyer, which, shall represent the entire share capital of the Company as
of immediately prior to the Closing (the “IT Sale” and together with the US Sale, the “Sale”).
(b) The
Parties acknowledge and agree that the purpose of this Agreement is to regulate the sale and purchase of all, and not less than all, of the US Subsidiary Stock and the Company Stock, so that, upon consummation of the Closing, Buyer shall be the sole
and exclusive shareholder holding 100% of the equity interests of the US Subsidiary, and IT Buyer shall be the sole and exclusive shareholder
17
holding the entire, and not less than the entire, share capital of the Company on a fully diluted basis, and Buyer and IT Buyer shall have full, unrestricted and marketable legal and beneficial
title to the US Subsidiary Stock and the Company Stock, respectively. The Shareholders acknowledge and agree that the above constitutes an essential assumption for Buyer and IT Buyer who otherwise would not enter into this Agreement nor undertake to
carry out the Transactions set forth herein.
2.2 Closing Transaction Consideration. Subject to and upon the terms and conditions
of this Agreement, each Shareholder shall be entitled to receive at the Closing a number of shares of Buyer Common Stock equal to (i) the Net Closing Shares multiplied by (ii) such Shareholder’s Closing Pro Rata Portion,
rounded down to the nearest whole share, without interest, upon execution of a deed of transfer – pursuant to Italian law, authenticated by the Italian Notary Public – by and among the Buyer and each of the Shareholder (the “Share
Deed of Transfer”). Additionally, each Shareholder will have the contingent right to receive its Closing Pro Rata Portion of the Holdback Shares, rounded down to the nearest whole share, and its Milestone Pro Rata Portion of the Milestone
Payments, if any (any such Holdback Shares and/or Milestone Payments actually issuable to the Shareholders, together with the Net Closing Shares and the US Subsidiary Consideration, the “Total Consideration”) after the Closing in
accordance with, and subject to, Section 2.3 and Article VI. Upon the consummation of the Closing, each Shareholder shall cease to have any direct rights in and to Company.
2.3 Closing Activities and Disbursement of Closing Transaction Consideration.
(a) Subject to the provisions of this Section 2.3, as soon as practicable after the Closing, Buyer shall issue, or
cause its transfer agent to issue, to each Shareholder, such Shareholder’s Closing Pro Rata Portion of the Net Closing Shares, rounded down to the nearest whole number, via book-entry issuance.
(b) If any portion of the Total Consideration is to be delivered or issued to a Person other than the Person in whose name the Closing Company
Shares are registered immediately prior to the Closing, it shall be a condition to such delivery that (i) the Transfer of such Company Stock shall have been permitted in accordance with the terms of the Company Charter Documents, each as in
effect immediately prior to the Closing, (ii) such Closing Company Shares shall be in proper form for transfer and (iii) the Person requesting such delivery shall pay to Buyer or Company, as applicable, any Transfer or other Taxes required
as a result of such delivery to a Person other than the registered holder of such Closing Company Shares or establish to the satisfaction of Company and Buyer that such Tax has been paid or is not payable.
(c) All Closing Shares issued to each Shareholder in accordance with the terms hereof, together with the contingent right of a Shareholder to
receive such Shareholder’s Closing Pro Rata Portion of any Holdback Shares and such Shareholder’s Milestone Pro Rata Portion of any Milestone Payments, shall be deemed to have been issued in full satisfaction of all rights pertaining to
such Shareholder’s Company Stock.
(d) Notwithstanding anything to the contrary contained herein, no fraction of a share of Buyer
Common Stock will be issued by virtue of the Transactions, and each Person who would otherwise be entitled to a fraction of a share of Buyer Common Stock (after aggregating all fractional shares of Buyer Common Stock that otherwise would be received
by such holder) shall instead have the number of shares of Buyer Common Stock issued to such Person rounded up in the aggregate to the nearest whole share of Buyer Common Stock.
(e) The Company irrevocably and unconditionally agrees to the Transfer of the US Subsidiary Stock to Buyer and so waives any right of
preemption or other limitation to the Transfer of the US Subsidiary Stock set forth in the Subsidiary Charter Documents or otherwise.
18
(f) Each Shareholder irrevocably and unconditionally agrees to the Transfer of the Company
Stock to the IT Buyer and so waives any right of preemption or other limitation to the Transfer of the Company Stock set forth in the Company Charter Documents or otherwise.
2.4 Closing Calculations.
(a) No later than three (3) Business Days prior to the Closing Date, the Company shall deliver to Buyer a statement certified by an
executive officer of Company (the “Estimated Closing Statement”) setting forth Company’s good faith estimate of (A) Closing Cash, (B) Closing Debt, (C) Closing Company Transaction Expenses and (D) Closing
NWC Adjustment Amount, calculated in accordance with the sample net working capital spreadsheet attached hereto as Exhibit B (the “Sample NWC Spreadsheet”).
(b) Not later than one (1) Business Day prior to the Closing Date, the Company shall deliver to Buyer a statement certified by an
executive officer of the Company setting forth each Shareholder’s Closing Pro Rata Portion of the Closing Shares in accordance with Schedule 1 of the Company Shareholder Letter.
2.5 Company Securities.
(a) Treatment of Convertible Notes. Prior to the Closing, in full satisfaction of the Company’s obligations under each Convertible
Note, each Convertible Note shall be cancelled and the Company shall issue to each Convertible Noteholder, in lieu of any cash payment, with respect to such Convertible Noteholder’s Convertible Note, a number of shares of Company Stock issuable
upon the occurrence of a Liquidation Event (as defined therein).
(b) Treatment of Rights to Issuance of Warrants. Prior to the
Closing, the Company and the Shareholders shall take all appropriate actions so that (i) the provisions of a certain investment agreement providing for the issuance of any warrant shall be terminated and each such investor entitled to issuance
of warrants will cease to have any rights with respect thereto, and (ii) the capital increase resolved upon by the extraordinary shareholders’ meeting of the Company on April 9, 2018, for a nominal value of up to Euro 341,227.00 and
reserved to Shareholders Sofinnova Partners SAS, Genextra S.p.A. and Innogest SGR S.p.A., shall be revoked. Each such investor shall have executed a warrant cancellation and release agreement in a form acceptable to Buyer concurrently with the
execution of this Agreement.
2.6 Transfer Restrictions; Stop-Transfer Instructions.
(a) The Transaction Securities constitute “restricted securities” under the Securities Act and may not be transferred absent valid
and effective registration under the Securities Act or an exemption therefrom, and any such transfer shall be subject to compliance with Applicable Laws. All Transaction Securities shall bear a legend or legends referencing restrictions applicable
to such shares under Applicable Law under this Agreement, which legend shall state in substance:
“The securities evidenced by this
certificate or in book-entry form have been issued and sold without registration under the United States Securities Act of 1933, as amended (the “Securities Act”), or the securities laws of any state of the United States (a
“State Act”) in reliance upon certain exemptions from registration under said acts. The securities evidenced by this certificate or in book-entry form cannot be sold, assigned or otherwise transferred within the United States
unless such sale, assignment or other transfer is (i) made pursuant to an effective registration statement under the Securities Act and in accordance with each applicable State Act or (ii) exempt from, or not subject to, the Securities Act
and each applicable State Act”.
19
In addition, the Holdback Shares shall include the following legend:
“The securities evidenced by this certificate or which are in book-entry form are subject to a holdback which restricts the rights to
transfer or otherwise dispose of these securities and subjects this share to forfeiture and cancellation by the issuer in certain circumstances. A copy of the definitive agreement governing such holdback may be obtained at the issuer’s
principal office.”
(b) In order to ensure compliance with the restrictions imposed by this Agreement, Buyer may issue appropriate
“stop-transfer” instructions to its transfer agent, and if Buyer acts as its own transfer agent, it may make appropriate notations to the same effect in its own records. Buyer will not be required (a) to transfer on its books any
Transaction Securities that have been sold or otherwise transferred in violation of any of the provisions of this Agreement, or (b) to treat as owner of such shares of Buyer Common Stock, or to accord the right to vote or receive dividends, to
any purchaser or other transferee to whom such shares of Buyer Common Stock have been so transferred. Buyer may require written assurances, in form and substance satisfactory to counsel for Buyer (which may include a requirement that the applicable
Shareholder’s counsel provide a legal opinion acceptable to Buyer), before Buyer effects any future transfers of shares of Buyer Common Stock issued in connection with this Agreement.
(c) Following the date that is six months following the Closing, and to the extent that Buyer Common Stock is eligible for resale under Rule
144(b)(1) or any successor provision, Buyer shall provide its transfer agent with the requisite opinion within two (2) Trading Days of such date to allow its transfer agent to remove any legends from the shares of Buyer Common Stock issued
under this Agreement in respect of a sale requested by a Shareholder or its broker that is compliant with Rule 144, unless such Shareholder is an Affiliate of Buyer within the meaning of the Securities Act; provided, that Buyer shall not be
obligated to remove any legends from the Holdback Shares until such shares of Buyer Common Stock are eligible to be released in accordance with Section 6.4; and; provided further, the Buyer shall, upon appropriate notice and receipt of
customary representations from a Shareholder if the Buyer Common Stock has been fully paid for and beneficially held by a Shareholder for at least one year from the Closing and is eligible for resale under Rule 144(b)(1) or any successor provision,
except with respect to any Holdback Shares, provide its transfer agent with the requisite opinion within two (2) Trading Days of such date to allow its transfer agent to remove the Securities Act restrictive legend from such Shareholder’s
Buyer Common Stock.
(d) If, after the Closing but prior to the third anniversary of the Closing Date ( unless Buyer has already caused
its transfer agent to remove any legends from the shares of Buyer Common Stock issued under this Agreement), Buyer shall (in its sole discretion) file a registration statement on Form S-3 with respect to the
resale of Buyer Common Stock on a delayed or continuous basis pursuant to Rule 415 under the Securities Act, Buyer shall use commercially reasonable efforts to include the Transaction Securities as part of such registration statement, subject to
applicable securities laws and completion and execution by each Shareholder of customary documentation reasonably required by Buyer.
2.7
Earnout.
(a) After the Closing, subject to the terms and conditions set forth in this Section 2.7, the Shareholders shall
have the contingent right to receive, and Buyer shall pay or cause to be paid, cash payments upon the achievement of certain milestone events (each, a “Milestone, and together, the “Milestones” and each such payment, a
“Milestone Payment” and together, the “Milestone Payments”)
20
in each case as set forth on Schedule I hereto; provided, however, that in no event shall Buyer be obligated to pay an aggregate amount of Milestone Payments exceeding $485,000,000.00;
provided, further, that no more than one (1) Milestone Payment shall be payable in respect of each Milestone. Notwithstanding the foregoing, in no event shall Buyer be obligated to make a Milestone Payment in respect of a Milestone that is
achieved after (i) for Milestones 1 through 6 (as set forth on Schedule I), the date that is the 7th anniversary of the Closing or (ii) for Milestones 7 through 10 (as set forth on Schedule I), the date that is the 11th anniversary of the
Closing (each date set forth (i) and (ii), a “Milestone End Date”).
(b) Upon the occurrence of any Milestone (and
in no event later than forty-five (45) days following each such occurrence), Buyer shall provide written notice to the Holder Representative (“Milestone Notice”), which Milestone Notice shall indicate the Milestone achieved,
the date of such achievement, and the Milestone Payment due on account thereof. Within thirty (30) days of such notice, Buyer will pay to each Shareholder such Shareholder’s Milestone Pro Rata Portion of the applicable Milestone Payment,
rounded down to the nearest cent, as set forth on a schedule which Holder Representative shall provide to Buyer in writing at least ten (10) days prior to such payment becoming due. Buyer shall pay interest on any Milestone Payments set forth
in the Milestone Notice that are not paid on or before the date that is 30 days after such payments are due under this Section 2.7 at a rate of ten percent (10%) per annum, calculated based on the days payment is
delinquent. The Milestone Payments to be made by or on behalf of Buyer pursuant to this Section 2.7 shall be made in U.S. dollars paid by wire transfer of immediately available funds.
(c) From and after the Closing until the earlier of the seventh anniversary of the Closing and a Reversion, Buyer shall use, and shall cause
the Buyer Parties to use, Commercially Reasonable Efforts to achieve Milestones 1-4. From and after the Closing until the earliest of (i) the seventh anniversary of the Closing, if the EryDex NDA has not
been approved by the FDA by such time, (ii) the 11th anniversary of the Closing, and (iii) a Reversion, Buyer shall use, and shall cause the Buyer Parties to use, Commercially Reasonable
Efforts to achieve Milestone 5. Without limiting the foregoing, from and after the Closing until the earlier of (x) the seventh anniversary of the Closing, if the EryDex NDA has not been approved by the FDA by such time and (y) the last
Milestone End Date, the Buyer shall not, and shall cause the Buyer Parties not to, take any action with the primary intent of not achieving any Milestone or not paying any Milestone Payment. For the purposes of this
Section 2.7(c)(ii), “Commercially Reasonable Efforts” means, with respect to the efforts to be expended by the Buyer and the Buyer Parties with respect to the applicable Milestones, good faith efforts and
reasonable use of resources to accomplish any such Milestone as is commonly dedicated in the pharmaceutical industry by a similarly situated biopharmaceutical company for a product of similar commercial potential at a similar stage of development to
the EryDex Product, in each case taking into account issues of safety and efficacy, product profile, the proprietary position, the then current competitive environment and the likely timing of market entry, the regulatory environment and status of
such product, and other relevant scientific, technical and commercial factors, but without regard to whether such actions may have an impact on the ability of the Shareholders to receive any Milestone Payments that may be owed or become payable
under this Agreement.
(d) Subject to Buyer’s obligations in Section 2.7(c), from and after the Closing,
the Buyer and its Subsidiaries, including the Company and the US Subsidiary, will be entitled to operate their respective businesses based upon the good faith business requirements of the Buyer and its Subsidiaries. Subject to the Buyer’s
obligations in Section 2.7(c), each of the Buyer and its Subsidiaries, including the Company and the US Subsidiary, will be permitted, following the Closing, to make changes at its sole discretion, based upon the good faith
business requirements of the Buyer and its Subsidiaries, to its operations, organization, personnel, accounting practices and other aspects of its business. Each Shareholder acknowledges and agrees that, subject to Buyer’s obligations in
Section 2.7(c), Buyer is entitled to conduct the business of Buyer and its Affiliates (including after Closing, the Company and US Subsidiary), including with respect to any EryDex Products, in a manner that is in the best
interests of Buyer and its shareholders, and shall have the absolute right and sole and absolute discretion to operate and otherwise make decisions with respect to the conduct of such businesses and/or the EryDex Product and to take or refrain from
taking any action with respect thereto.
21
(e) Until the last Milestone End Date, Buyer shall use Commercially Reasonable Efforts to
keep those records that are necessary to ascertain whether the Milestones have been achieved by the time of the applicable Milestone End Date. Such records shall be kept for such period of time required by Applicable Laws, but no less than three
years following the end of the Calendar Quarter to which they pertain. Until the last Milestone End Date, the Holder Representative shall not more than one (1) time each year have the right to have an external independent registered public
accounting firm acceptable to Buyer (an “Agreed Auditor”) inspect Buyer’s applicable records for the sole purpose of determining the accuracy of Milestone Payments, subject to such Agreed Auditor signing a confidentiality
agreement acceptable to Buyer. No period will be audited more than once and each audit must be reasonable in scope. Any such Agreed Auditor shall keep confidential any information obtained during such inspection and shall report to the Holder
Representative and Buyer only the amounts of Milestone Payments due and payable (if any). Such audits may only be exercised during normal business hours upon reasonable prior written notice to Buyer and subject to Buyer’s prior written consent
(not to be unreasonably withheld). The Holder Representative (on behalf of the Shareholders) shall bear the full cost of such audit unless such Agreed Auditor makes a final determination of Buyer’s failure to make a Milestone Payment due under
this Agreement which is uncontested by Buyer (or if contested by Buyer is finally determined to be due by a court of competent jurisdiction), in which case, Buyer shall bear the reasonable, out-of-pocket, documented costs of such audit.
(f) If the Holder Representative believes that
any Milestone has occurred and the Holder Representative has not timely received a Milestone Notice from Buyer, then the Holder Representative shall deliver to Buyer written notice thereof (a “Milestone Dispute Notice”), setting
forth the basis for such belief in reasonable detail. Following the delivery of a Milestone Dispute Notice, Buyer and the Holder Representative shall first attempt in good faith to resolve by negotiation and consultation between themselves, any
dispute as to whether any Milestone has occurred and whether any Milestone Payment is payable. If Buyer and the Holder Representative do not reach agreement with respect to any dispute relating to any such matter within sixty (60) days after a
Milestone Dispute Notice is delivered to Buyer by the Holder Representative, the Parties shall submit for arbitration all matters that remain in dispute to a disinterested individual who has appropriate scientific, technical and regulatory expertise
(as relevant) to resolve any disputes referred to him or her under this Section 2.7(g) (a “Scientific Expert”) who is mutually agreed to by Buyer and the Holder Representative; provided, however, that such
Scientific Expert shall not be or have been at any time within the previous ten (10) years an Affiliate, employee, consultant, officer or director of Buyer, the Holder Representative, the Company or the US Subsidiary, any Shareholder or any of
their respective Affiliates. If Buyer and the Holder Representative cannot agree on a mutually acceptable Scientific Expert within thirty (30) days after either party has determined that the Parties cannot reach agreement with respect to a
dispute, then within ten (10) Business Days after the expiration of such thirty (30) day period, each of Buyer and the Holder Representative shall appoint one Scientific Expert who shall jointly select a third Scientific Expert within ten
(10) Business Days after the last to occur of their respective appointments to arbitrate the referred matter. The Scientific Expert mutually agreed by the Parties or, if the Parties cannot agree, the third Scientific Expert selected by the
party-appointed Scientific Experts is referred to as the “Selected Scientific Expert”. Buyer and the Holder Representative shall instruct the Selected Scientific Expert to determine as promptly as practicable but in no event later
than forty-five (45) days after such person’s appointment (the “Determination Period”) whether the disputed Milestone has occurred. The Selected Scientific Expert’s determination shall be made based on the submission
of documents and evidence by the Parties (including any such documentation or evidence reasonably requested by the Selected Scientific Expert, which the Holder Representative or Buyer shall provide upon request). The Selected Scientific Expert shall
determine deadlines (which Buyer and the
22
Holder Representative shall deem to be fair and appropriate) within the Determination Period for submitting documents and dates, if any, of oral hearings. The parties agree that the arbitration
will be in accordance with the JAMS Comprehensive Arbitration Rules & Procedures (“JAMS”). Any decision rendered by the Selected Scientific Expert shall be final and binding upon the parties. The non-prevailing party (being either Buyer or the Holder Representative, on behalf of the Shareholders) shall pay the expenses of arbitration, and the fees, costs and expenses of the Selected Scientific Expert. All
proceedings conducted by the Selected Scientific Expert shall take place in Boston, Massachusetts, USA. Any underpayments of Milestone Payments shall be paid to the Shareholders within ten (10) Business Days of notification of the final
determination of the Milestone Dispute Notice in accordance with this Section 2.7.
(g) The Parties further
agree that the payment of any Milestone Payments, if any, shall be treated by the Parties for U.S. federal and applicable state and local income tax purposes as deferred contingent purchase price eligible for installment sale treatment under
Section 453 of the Code and any corresponding provision of state or local law, as appropriate (subject to imputation of interest under Section 483 or Section 1274 of the Code), except as otherwise required by Applicable Law.
(h) No Shareholder shall have the right to sell, offer to sell, contract or agree to sell, hypothecate, pledge, grant of any option to
purchase or otherwise dispose of or agreement to dispose of, directly or indirectly, any of such Shareholder’s contingent right to receive any Milestone Payment under this Section 2.7; provided however that any
assignment resulting from an operation of Law (including, in the event of liquidation of an investment fund, to successors) shall not be subject to such limitation and shall not require any other Party’s consent. The contingent right to receive
any Milestone Payment shall not be represented by any form of certificate or other instrument and does not constitute a security or an equity or ownership interest in Buyer. No Shareholder shall have any rights as a securityholders of Buyer in
respect of such Shareholder’s contingent right to receive Milestone Payments.
(i) Until the earlier of (x) the seventh
anniversary of the Closing, if the EryDex NDA has not been approved by the FDA by such time and (y) the last Milestone End Date, if Buyer or any of its Affiliates (including after Closing, the Company or the US Subsidiary for so long as they
are Affiliates of Buyer) Divests to a Third Party any EryDex Product, including the Company Owned IP (collectively, “Divested Assets” and the party receiving any Divested Assets the “Transferee”), Buyer will either
(x) make provision for the Transferee to assume and succeed to the obligations of Buyer set forth in this Section 2.7 (including Schedule I) and Section 2.8, or (y) Buyer shall remain
liable to the Shareholders for all obligations of Buyer set forth in this Section 2.7 (including Schedule I).
2.8
Reversion.(a) Subject to Section 2.7(f), in the event that Buyer fails to timely pay any Milestone Payment payable to the Shareholders in accordance with Section 2.7 (including Schedule I),
the Holder Representative shall deliver to Buyer written notice thereof (“Milestone Default Notice”). At the written election of the Holder Representative delivered to Buyer, in lieu of Buyer’s obligation to pay such Milestone
Payment and any interest on delinquent payments as set forth in Section 2.7(b), if Buyer fails to pay the applicable Milestone Payment within one hundred eighty (180) days after the Holder Representative’s
provision of the Milestone Default Notice (such date, the “Reversion Date”), Buyer shall either (x) assign and deliver the Product Program to a Person designated in writing by the Holder Representative to the Buyer on or
prior to the Reversion Date, pursuant to an assignment agreement to be negotiated in good faith by the Holder Representative (or its designee) and the Buyer within ninety (90) days following the Reversion Date, or (y) remain obligation to
make such Milestone Payment and applicable interest pursuant to Section 2.7. For purposes of this Section 2.8, “Product Program” means the following: all (1) Intellectual
Property that is solely related to the EryDex Product, including the Company Owned IP; (2) regulatory documents, submissions, and approvals related to the EryDex Product, (3) ongoing Clinical Trials of the EryDex Product and
(4) inventory of the EryDex Product (including all final product, bulk drug substance, intermediates, works-in-process, formulation materials, reference
23
standards, drug product clinical reserve samples, packaged retention samples, and the like), in each case of (1) – (4), to the extent owned, controlled or in the possession of the Buyer or
its Affiliates (including, from and after the Closing, Company and the US Subsidiary) as of the Reversion Date. Notwithstanding the foregoing, upon the occurrence of a Divestiture the provisions set forth in this
Section 2.8 shall be of no further force or effect.
2.9 Further Assurances. If, at any time before or
after the Closing, Buyer reasonably believes or is advised that any further instruments, deeds, assignments or assurances are reasonably necessary to consummate the Transactions or to carry out the purposes and intent of this Agreement at or after
the Closing, then the Company Parties and their respective officers and directors shall execute and deliver all such proper deeds, assignments, instruments and assurances and do all other things reasonably necessary to consummate the Transactions
and to carry out the purposes and intent of this Agreement.
2.10 Withholding. Notwithstanding anything to the contrary contained
in this Agreement, Buyer, IT Buyer and any Company Group Member (or any other Person required to deduct or withhold with respect to any payment made under this Agreement) shall be entitled to deduct and withhold from any consideration payable or
otherwise deliverable pursuant to this Agreement (including the Holdback Shares and the Milestone Payments) such amounts as may be required to be deducted or withheld therefrom under any provision of Applicable Laws, and to request and be provided
any necessary Tax forms, including IRS Form W-9 or the appropriate version of IRS Form W-8, as applicable, or any similar information. To the extent that amounts are so
deducted or withheld, such amounts shall be paid over to the appropriate Governmental Authority and shall be treated for all purposes of this Agreement as having been paid to the Person to whom such amounts would otherwise have been paid.
ARTICLE III
REPRESENTATIONS AND WARRANTIES OF COMPANY
Except as set forth in a numbered or lettered section of the Company Disclosure Schedule that correspond to the sections of Article III
of this Agreement and other sections of the Company Disclosure Schedule to the extent (i) such information is cross-referenced in another part of the Company Disclosure Schedule, or (ii) the relevance to such other section of Article
III of this Agreement is reasonably apparent on the face of such disclosure, Company represents and warrants to Buyer that the statements contained in this Article III are true and correct on and as of the Agreement Date:
3.1 Organization and Good Standing. Each of the Company and the US Subsidiary (i) is a corporation or other organization duly
organized, validly existing and in good standing or its jurisdictional equivalent under the laws of the jurisdiction of its incorporation, formation or organization (except, in the case of good standing or its jurisdictional equivalent, for entities
organized under the laws of any jurisdiction that does not recognize such concept); (ii) has the requisite power and authority to own, operate and lease its properties and to carry on the Company Business; (iii) is duly qualified or licensed to
do business, and is in good standing, in each jurisdiction where the character of the properties owned, leased or operated by it or the nature of its activities makes such qualification or licensing necessary and (iv) is not subject to
bankruptcy, insolvency, reorganization, moratorium and similar laws relating to or affecting creditors generally and by the availability of equitable remedies (whether in proceedings at law or in equity) and is not in the situation provided for
under article 2446 and 2447 of the Italian Civil Code.
3.2 Subsidiaries. The US Subsidiary is the only Subsidiary of the Company
as of the Agreement Date. Except as set forth in Section 3.2 of the Company Disclosure Schedule, all the outstanding shares of capital stock of, or other equity or voting interests in, the US Subsidiary are duly authorized,
have been validly issued and are fully paid and nonassessable and are owned by Company, a wholly-owned Subsidiary of Company, or Company and another wholly-owned Subsidiary of Company, free and clear of
24
all material Encumbrances of any kind or nature whatsoever, other than Company Permitted Encumbrances, except for restrictions imposed by applicable securities laws. Except as set forth in
Section 3.2 of the Company Disclosure Schedule, other than the US Subsidiary, neither Company nor its Subsidiary owns any capital stock of, or other equity or voting interests of any nature in, or any interest convertible,
exchangeable or exercisable for, capital stock of, or other equity or voting interests of any nature in, any other Person.
3.3 Power,
Authorization and Validity.
(a) Power and Authority. Company has all requisite corporate power and corporate authority to
enter into, execute, deliver and perform its obligations under this Agreement and each of the Company Ancillary Agreements and to consummate the Transactions. The Transactions and the execution, delivery and performance by Company of this Agreement,
each of the Company Ancillary Agreements and all other agreements, transactions and actions contemplated hereby or thereby, have been duly and validly approved and authorized by all requisite corporate action on the part of the Company and no other
corporate proceedings on the part of Company are necessary to approve this Agreement and each of the Company Ancillary Agreements or to authorize or consummate the Transactions.
(b) Enforceability. This Agreement and each of the Company Ancillary Agreements are, or when executed by Company shall be, assuming the
due authorization, execution and delivery by each other party hereto or thereto, valid and binding obligations of Company, enforceable against Company in accordance with their respective terms, except as such enforceability may be limited by
applicable bankruptcy, insolvency, reorganization, moratorium and similar laws relating to or affecting creditors generally and by the availability of equitable remedies (whether in proceedings at law or in equity) (collectively, the
“Enforceability Exceptions”).
3.4 Capitalization of Company.
(a) Authorized and Outstanding Capital Stock of Company. The authorized capital stock of the Company consists solely of 1,751,985
shares of Company Stock. The number of issued and outstanding shares of Company Stock held by each Shareholder as of the Agreement Date is set forth in Section 3.4(a) of the Company Disclosure Schedule. All issued and
outstanding shares of Company Stock have been duly authorized and validly issued, are fully paid and nonassessable, were not issued in violation of any right of rescission, right of first refusal or preemptive right, and have been offered, issued,
sold and delivered by Company in compliance with all requirements of Applicable Law and all material requirements set forth in applicable Company Material Contracts. There is no liability for dividends accrued and unpaid by Company.
(b) Options. As of the Agreement Date, the Company has reserved 178,368 shares of the Company for issuance to employees, non-employee directors and consultants pursuant to the Company Stock Plan, of which 163,087 shares are subject to outstanding and unexercised Company Options, and 15,281 shares remain available for issuance
thereunder. Section 3.4(b) of the Company Disclosure Schedule sets forth, as of the Agreement Date, a correct and complete list of all Optionholders, and each Company Option, whether or not granted under the Company Stock
Plan, and which further sets forth for each such Person: (i) the number and type of shares of Company Stock subject to each Company Option, (ii) the “date of grant” of such Company Option (as defined under Treasury Regulations Section 1.409A-1(b)(5)(vi)(B)), (iii) the exercise price per share, and (iv) the particular Company stock plan from which such Company Option was granted (if any). Correct and complete copies of the
Company Stock Plan and each standard form award agreement and instrument relating to or issued under the Company Stock Plan, have been made available to Buyer, and such Company Stock Plan and Contracts have not been amended, modified or supplemented
since being made available to Buyer, and no Company Options differ in any
25
material respect from such form agreement (other than any vesting acceleration provisions contained therein as indicated in Section 3.4(b) of the Company Disclosure
Schedule). There are no agreements, understandings or commitments (written or oral) to amend, modify or supplement such Company Stock Plan or Contracts in any case from those made available to Buyer. There are no shares of Company Stock outstanding
that are not vested under the terms of any Contracts with the Company (including any stock option agreement, stock option exercise agreement or restricted stock purchase agreement).
(c) No Other Rights. Except as set forth on Section 3.4(c) of the Company Disclosure Schedule, there are no
stock appreciation rights, options, warrants, calls, rights, commitments, conversion privileges or preemptive or other rights or Contracts outstanding to purchase or otherwise acquire any shares of Company Stock, any securities or debt convertible
into or exchangeable for Company Stock, or any other securities of Company or the US Subsidiary, or obligating Company to grant, extend or enter into any such option, warrant, call, right, commitment, conversion privilege or preemptive or other
right or Contract or to redeem any shares of Company Stock or any other securities of Company or the US Subsidiary, other than the Company Charter Documents and Subsidiary Charter Documents. There are no voting agreements, registration rights,
rights of first refusal, preemptive rights, co-sale rights or other restrictions applicable to outstanding securities of Company or the US Subsidiary, other than the Company Charter Documents and the
Subsidiary Charter Documents.
3.5 Consents; Conflict.
(a) No Consents. Except for approvals and notices set forth on Section 3.5(a) of the Company Disclosure
Schedule, no consents or approvals of or notices to or filings, declarations or registrations with any Governmental Authority are necessary in connection with (i) the execution and delivery by Company of this Agreement or any of the Company
Ancillary Agreements or (ii) the consummation of the Transactions.
(b) No Conflict. Neither the execution and delivery of
this Agreement or any of the Company Ancillary Agreements by Company, nor the occurrence of the Closing, shall conflict with, result in a termination or breach or violation of (with or without notice or lapse of time, or both), or constitute a
default, or require the consent, release, waiver or approval of any Third Party, under: (i) any provision of the Company Charter Documents, each as currently in effect; (ii) except as set forth on Section 3.5(a)
of the Company Disclosure Schedule, any Applicable Law applicable to Company or any of its assets or properties; or (iii) except as set forth on Section 3.5(b) of the Company Disclosure Schedule, any Company Material
Contract.
3.6 Litigation. As of the Agreement Date, except as set forth in Section 3.6 of the Company
Disclosure Schedule, (a) there is no action, suit, arbitration, mediation, proceeding, claim or investigation pending against Company or the US Subsidiary (or against any officer, director, employee or agent of Company or the US Subsidiary in
their capacity as such) before any Governmental Authority and (b) no such action, suit, arbitration, mediation, proceeding, claim or investigation referred to in clause (a) has been threatened in writing. There is no judgement, decree,
injunction, rule or order of any Governmental Authority outstanding against Company or the US Subsidiary.
3.7 Taxes.(a)
(a) Each Company Group Member (i) has or will have timely filed, or cause to be timely filed, taking into account valid extensions
obtained in the Ordinary Course of Business, all Tax Returns required to be filed by it, and such Tax Returns are correct and complete in all material respects, and (ii) has timely paid all Taxes required to be paid by it (whether or not shown
on any Tax Return). No Company Group Member is the beneficiary of any extension of time within which to file any Tax Return (other than such extension validly obtained in the Ordinary Course of Business). No power of attorney with respect to Taxes
that is currently in force has been granted by any Company Group Member.
26
(b) Each Company Group Member has (i) properly withheld or collected with respect to
its employees, consultants, advisors, independent contractors, stockholders, and other third parties all Taxes required to be withheld or collected, (ii) timely paid over any such Taxes to the appropriate Governmental Authority in accordance
with Applicable Law, (iii) timely filed correct and complete information Tax Returns to the extent required to be filed by it with respect thereto, and (iv) otherwise complied in all material respects with all Applicable Laws relating to
the withholding and collection of Taxes.
(c) As of the Balance Sheet Date, no Company Group Member has any liabilities for unpaid Taxes
which have not been accrued or reserved on the Company Balance Sheet, whether asserted or unasserted, contingent, or otherwise. Since the Balance Sheet Date, no Company Group Member has incurred any liability for Taxes other than in the
Ordinary Course of Business (or recognized any extraordinary gain).
(d) There is no Tax deficiency outstanding, assessed, or proposed
against any Company Group Member, and no Company Group Member has executed any waiver of any statute of limitations on or extending the period for the assessment or collection of any Tax (other than extensions to file a Tax Return validly obtained
in the Ordinary Course of Business), and no request for any such waiver or extension has been filed or is currently pending.
(e) No audit
or other examination of any Tax Return of any Company Group Member is presently in progress, nor has any Company Group Member been notified of any request for such an audit or other examination. No claim has ever been made by any Governmental
Authority in a jurisdiction where any Company Group Member does not file Tax Returns that such Company Group Member is or may be subject to Tax, or required to file Tax Returns, in such jurisdiction. No adjustment relating to any Tax Return filed by
any Company Group Member has been proposed by any Governmental Authority. No Company Group Member is a party to or bound by any closing or other agreement or ruling with respect to Taxes with any Governmental Authority. There are no matters relating
to Taxes under discussion between any Company Group Member and any Governmental Authority.
(f) The Company has delivered to Buyer correct
and complete copies of (i) all income, sales and use, value added, and other material Tax Returns filed by each Company Group Member for all taxable periods remaining open under the applicable statute of limitations, (ii) all examination
reports and statements of deficiencies, adjustments, and proposed deficiencies and adjustments in respect of Taxes of each Company Group Member, and (iii) all written analyses (whether in final or draft form) prepared by, or on behalf of, each
Company Group Member regarding whether any of the Tax attribute carryforwards of any Company Group Member (including, but not limited to, net operating losses and Tax credits) are subject to limitation pursuant to Sections 382, 383, or 384 of the
Code (or any corresponding or similar provision of state, local, or non-U.S. Applicable Law). Section 3.7(f) of the Company Disclosure Schedule sets forth each jurisdiction where each
Company Group Member will be required to file a Tax Return following the Closing with respect to any Pre-Closing Tax Period, including the type of Tax Return and the type of Tax required to be paid.
(g) There are no Encumbrances on the assets of any Company Group Member relating or attributable to Taxes, other than Encumbrances for Taxes
not yet due and payable.
(h) Each Company Group Member is and always has been a C corporation for U.S. federal and applicable U.S. state
and local income Tax purposes.
27
(i) Each Company Group Member uses the accrual method of accounting for income Tax purposes.
(j) No Company Group Member (nor any predecessor of such Company Group Member) has (i) ever been a member of an affiliated,
combined, consolidated, unitary, or similar group for Tax purposes (other than a group of which the Company is the parent), (ii) ever been a party to, or bound by, any Tax sharing, indemnification, reimbursement, or allocation agreement (other than
customary provisions in commercial agreements entered into in the Ordinary Course of Business the primary purpose of which is unrelated to Tax), (iii) any potential liability for the Taxes of any Person under Treasury Regulations Section 1.1502-6 (or any corresponding or similar provision of state, local, or non-U.S. Applicable Law, including any arrangement for group or consortium relief or
similar arrangement), as a transferee or successor, by operation of Applicable Law, or by Contract (other than customary provisions in commercial agreements entered into in the Ordinary Course of Business the primary purpose of which is unrelated to
Tax), or (iv) ever been a party to any joint venture, partnership, or other arrangement or agreement that could be treated as a partnership for Tax purposes.
(k) No Company Group Member (nor any Buyer Party as a result of its acquisition of such Company Group Member, as applicable) will be required
to include any item of income or gain in, or exclude any item of deduction or loss from, taxable income for any taxable period (or portion thereof) beginning after the Closing Date as a result of (i) any adjustment under Section 481 of the
Code (or any corresponding or similar provision of state, local, or non-U.S. Applicable Law) by reason of a change in a method of accounting, or the use of an improper method of accounting, for a taxable
period that ends on or prior to the Closing Date, (ii) any “closing agreement” within the meaning of Section 7121 of the Code (or any corresponding or similar provision of state, local, or
non-U.S. Applicable Law) entered into on or prior to the Closing Date, (iii) any intercompany transaction (including any intercompany transaction subject to Sections 367 or 482 of the Code) or excess loss
account described in the Treasury Regulations promulgated pursuant to Section 1502 of the Code (or any corresponding or similar provision of state, local, or non-U.S. Applicable Law) with respect to a
transaction occurring before the Closing, (iv) any installment sale or open transaction disposition made on or prior to the Closing Date, or (v) any deferred revenue or prepaid amount received on or prior to the Closing Date.
(l) No Company Group Member has (i) participated in any “reportable transaction” within the meaning of Treasury Regulations Section 1.6011-4(b), (ii) engaged in any transaction that would reasonably be likely to require the filing of an IRS Schedule UTP, (iii) participated or engaged in any other transaction that is subject to
disclosure requirements pursuant to a corresponding or similar provision of state, local, or non-U.S. Applicable Law, or (iv) taken any position on any Tax Return that could give rise to a substantial
understatement of Tax within the meaning of Section 6662 of the Code (or any corresponding or similar provision of state, local, or non-U.S. Applicable Law).
(m) No Company Group Member has constituted either a “distributing corporation” or a “controlled corporation” in a
distribution of stock intended to qualify for Tax-free treatment under Sections 355 or 361 of the Code.
(n) All transactions and agreements entered into between any of the Company Group Members have been made on arm’s length terms. Each
Company Group Member is in compliance with all Applicable Laws relating to transfer pricing, including the execution and maintenance of contemporaneous documentation substantiating the transfer pricing practices and methodology of each Company Group
Member (as required under Section 482 of the Code and any other state, local, or non-U.S. Applicable Law). No Company Group Member has ever entered into a cost sharing arrangement to share research and
development costs and rights to any developed Company Owned IP.
28
(o) No Company Group Member is subject to Tax in any country other than its country of
formation or incorporation by virtue of having a permanent establishment, branch operations, or other place of business or having engaged in a trade or business in such other country. No Company Group owns an interest in real property in any
jurisdiction in which a Tax is imposed, or the value of the interest reassessed, on the transfer of an interest in real property and which treats the transfer of an interest in an entity that owns an interest in real property as a transfer of the
interest in real property.
(p) Section 3.7(p) of the Company Disclosure Schedule lists all Tax holidays and similar Tax
benefits which have current applicability to any Company Group Member. Such Company Group Member is currently and always has been in compliance with the requirements for all such Tax holidays and similar Tax benefits. No Tax holiday or similar Tax
benefit of any Company Group Member will terminate or be subject to recapture or clawback by reason of the Transactions.
(q) Each Company
Group Member has (i) collected, remitted, and reported to the appropriate Governmental Authority all sales, use, value added, excise, and similar Taxes required to be so collected, remitted, or reported under Applicable Law and
(ii) complied in all material respects with all Applicable Laws relating to record retention (including to the extent necessary to claim any exemption from Tax collection and maintain adequate and current resale certificates to support any such
claimed exemption).
(r) There is no material property or obligation of any Company Group Member, including uncashed checks to vendors,
customers, or Employees, non-refunded overpayments, credits, or unclaimed amounts or intangibles, that are, or may become, escheatable or reportable as unclaimed property to any Governmental Authority under
any applicable escheatment, unclaimed property, or similar Applicable Law.
(s) No Company Group Member has availed itself of any Tax
relief pursuant to any Pandemic Response Laws that could reasonably be expected to impact the Tax payment or reporting obligations of such Company Group Member after the Closing.
(t) The US Subsidiary has not been, during the applicable period provided in Section 897(c) of the Code, a “United States real
property holding corporation” within the meaning of Section 897(c)(2) of the Code.
(u) The Company (i) is not, and has
never been, a “controlled foreign corporation” as defined in Section 957 of the Code, a “passive foreign investment company” as defined in Section 1297 of the Code, or a “surrogate foreign corporation” as
defined in Section 7874(a)(2)(B) of the Code and is not (ii) subject to U.S. federal income Tax (other than U.S. withholding taxes) under any provision of the Code. The Company is not subject to net income Tax (other than withholding
taxes) in any jurisdiction other than Italy. The Company has not elected to be treated as a “domestic corporation” under Section 897(i) of the Code.
3.8 Financial Statements; Books and Records.
(a) Financial Statements. Section 3.8(a) of the Company Disclosure Schedule includes the Company Financial
Statements. The Company Financial Statements: (i) are true and correct and fairly present, in all material respects, the financial position of the Company and the US Subsidiary at the Balance Sheet Date indicated and the results of
operations and cash flows of the Company Group for the periods therein specified; and (ii) have been prepared in accordance with Italian GAAP (in the case of Company) or GAAP (in the case of the US Subsidiary) consistently applied in accordance
with Company’s or the US Subsidiary’s practices throughout the periods indicated and with each other, except for the
29
absence of footnotes. The Company Group has no material liabilities or obligations, whether accrued or fixed, absolute or contingent, determined or determinable, including those arising under any
law, action or governmental order and those arising under any Contract, of a type required to be reflected on a balance sheet prepared in accordance with Italian GAAP, except for (v) those shown on the Company Balance Sheet, (w) those
incurred after the Balance Sheet Date in the Ordinary Course of Business of the Company Group, (x) liabilities set forth on Section 3.8(a) of the Company Disclosure Schedule or included in the calculation of
Transaction Expenses, (y) other undisclosed liabilities and/or obligations, and (z) liabilities and/or obligations directly incurred under the terms of this Agreement.
(b) Charter Documents. Company has made available to Buyer correct and complete copies of Company’s Deed of Incorporation and By-Laws, each as amended to date (collectively, the “Company Charter Documents”) and the organizational and governance documents of the US Subsidiary (collectively, the “Subsidiary Charter
Documents”), and each such document is in full force and effect. Company is not in material violation of any of the provisions of the Company Charter Documents and the US Subsidiary is not in material violation of its Subsidiary Charter
Documents.
3.9 Assets and Properties. The Company Group has good and marketable title to, or a valid leasehold interest in or
right to use, all of its assets and properties (including those shown on the Company Balance Sheet) free and clear of all Encumbrances, other than (a) Company Permitted Encumbrances and (b) the rights of lessors or licensors under
leasehold or licensed interests. The assets, rights and properties of Company Group are sufficient for the operation of the Company Business as presently conducted. Neither Company nor the US Subsidiary owns any real property.
3.10 Absence of Certain Changes. Since the Balance Sheet Date, the Company has conducted its business in the ordinary course consistent
with past practice in all material respects and there has not occurred (a) any event that has had or would reasonably be expected, individually or in the aggregate, to have a Material Adverse Effect or (b) any event or action that, if
taken during the period from the Interim Period without the consent of Buyer, would constitute a breach of Section 7.3.
3.11 Contracts, Agreements, Arrangements, Commitments and Undertakings.
Section 3.11 of the Company Disclosure Schedule sets forth a list of each of the following Contracts, as of the
Agreement Date, to which Company or the US Subsidiary is a party or to which Company, the US Subsidiary or any of the Company Group’s assets or properties is bound (other than with respect to the plans set forth on
Section 3.17(d) of the Company Disclosure Schedule):
(a) any Contract having an outstanding principal amount in
excess of €50,000 evidencing indebtedness for borrowed money of any Company Group Member or the mortgaging, pledging or otherwise placing an Encumbrance on any material portion of the assets of the Company Group;
(b) any Contract under which any Company Group Member has provided or is the beneficiary of a guaranty, performance bond or similar agreement
for an amount in excess of €100,000;
(c) any Contract under which any Company Group Member is lessee of or holds or operates any
asset, owned by any other Person, except for any lease of assets or Contract pursuant to which any Company Group Member holds or operates any asset under which the annual rental payments do not exceed €50,000;
(d) any Contract under which any Company Group Member is lessor of or permits any Third Party to hold or operate any assets owned by the
Company Group, except for any lease of assets or Contract pursuant to which any Company Group Member permits any Third Party to holds or operate any asset under which the annual rental payments do not exceed €50,000;
30
(e) any material Contract pursuant to which any Company Group Member grants or receives
rights in or to use any material Intellectual Property;
(f) any Contract which, by its terms calls for aggregate payments or receipts by
the Company Group under such Contract of at least €100,000 in the aggregate during the twelve-month period ending on December 31, 2023 or is reasonably expected to involve the payment or receipt by the Company Group of more than
€100,000 in the aggregate, including any Contract for the purchase or sale of real property, raw materials, goods, commodities, utilities, equipment, supplies, products or other personal property, or for the provision or receipt of services;
(g) any Contract for the employment or engagement of any director, officer, employee, or consultant of the Company Group, or any other
type of Contract with any director, officer, employee, or consultant of the Company, that is not immediately terminable by the Company Group at-will without notice, severance, cost or liability (excluding
employment Contracts with employees located in a jurisdiction that does not recognize at-will employment and may be terminated with no more than the minimum severance and notice benefits required by Applicable
Laws); any separation agreement, or settlement agreement with any employee of any Company Group or other Person, under which the Company Group has any current actual or potential liability, as well as any settlement agreement, consent decree, or
other similar agreement with any Governmental Authority;
(h) any Contract providing for retention payments, change of control payments,
severance, accelerated vesting or any other payment or benefit that may or will become due as a result of the transactions contemplated by this Agreement;
(i) any collective bargaining agreement or any other Contract with trade or labor union, works council, employees’ association or similar
organization, or any similar Contract with Governmental Authority or employees of the Company Group;
(j) any Contract concerning the
establishment or operation of a joint venture or partnership;
(k) any Contract that materially restricts the Company Group from
(1) engaging in any aspect of the Company Group’s business, (2) participating or competing in any line of business, market or geographic area, or (3) freely setting prices for the Company Group’s products and services
(including most favored customer pricing provisions);
(l) any Contract that grants any exclusive rights, rights of refusal, or rights of
first negotiation to any Person;
(m) any Contract pursuant to which any Company Group Member has acquired a business or entity, or
substantially all of the assets of a business or entity, whether by way of merger, consolidation, purchase of stock, purchase of assets or otherwise;
(n) any Contract with respect to the conduct of Clinical Trials; and
(o) any Contract with any Third Party to develop, promote or market the Company Product or Service or that otherwise grants such Third Party
any license to develop, manufacture, supply, distribute, market, commercialize, sell, offer, import or otherwise commercialize the Company Product or Service or any other product.
31
A true and complete copy of each agreement or document, including any material amendments
thereto, required by this Section 3.11 to be listed on Section 3.11 of the Company Disclosure Schedule has been made available to Buyer. Each of the Company Material Contracts is a legal, valid and
binding obligation of Company or the US Subsidiary party thereto (assuming the due authorization, execution and delivery by the other parties thereto) and is in full force and effect and enforceable in accordance with its terms (subject to the
Enforceability Exceptions).
Company and the US Subsidiary have performed all material obligations required to be performed by it to date
under the Company Material Contracts, and there exists no default or event of default or event, occurrence, condition or act, with respect to Company or the US Subsidiary, with respect to any other party to a Company Material Contract, which, with
the giving of notice, the lapse of time or the happening of any other event or conditions, would reasonably be expected to (i) constitute a default or event of default under any Company Material Contract or (ii) give any Third Party
(1) the right to declare a default or exercise any remedy under any Company Material Contract, (2) the right to accelerate the maturity or performance of any obligation of Company under any Company Material Contract, or (3) the right
to cancel, terminate or modify any Company Material Contract. Neither Company nor the US Subsidiary has received any written notice or other written communication regarding any actual or possible violation or breach of or default under, or intention
to cancel, call a default under, or adversely modify, any Company Material Contract.
3.12 Intellectual Property.
(a) Section 3.12(a) of the Company Disclosure Schedule identifies: (x) each item of Company Registered IP; (y) the
jurisdiction in which such item of Company Registered IP has been registered or filed and the applicable registration or serial number; and (z) all owner(s), and, if one or more owner(s) is not the Company Group (including any joint owner(s)),
(i) the corresponding license agreement pursuant to which the Company Group has the right to use such Company Registered IP, and (ii) if any such Person has a joint ownership interest in such Company Registered IP, any corresponding
agreement(s) relating to such joint ownership. To the Company’s knowledge, each of the Patents included in the Company Registered IP that is solely owned or purported to be solely owned by the Company Group, and each of the jointly owned and
other Patents included in the Company Registered IP, properly identifies by name each and every inventor of the claims thereof as determined in accordance with United States patent law (and the inventors listed in each such Patent collectively
constitute the entire inventive entity, as the term ‘inventive entity’ is defined and interpreted under United States patent law). To the Company’s knowledge, the Company Group has complied in all material respects with all of its
obligations and duties to the respective patent offices, including the duty of candor and disclosure to the U.S. Patent and Trademark Office, and all Applicable Laws, with respect to all Company Registered IP. The Company has no information
regarding any information, facts or circumstances that would reasonably be expected to result in any challenge to, or otherwise adversely impact, in any material respect, the ownership, use, patentability, registrability, enforceability or validity
of any Company Registered IP, including any Intellectual Property that is the subject matter thereof. The Company Registered IP owned or purported to be owned by any Company Group, and the other Company Registered IP, is subsisting. To the
Company’s knowledge, the issued Patents included in the Company Registered IP are valid and enforceable. No Registered IP that is listed or required to be listed on Section 3.12(a) of the Company Disclosure Schedule
has been, and no Company Group Member has received a notice that any Registered IP that is listed or required to be listed on Section 3.12(a) of the Company Disclosure Schedule is, involved in any nullity, inter partes or
interference, opposition, reissue, reexamination, revocation, or equivalent proceeding, in which the inventorship, scope, validity or enforceability of any such Registered IP is being or has been contested or challenged, and no such proceeding has
been threatened with respect to any such Registered IP.
32
(b) The Company Group owns, and has good, valid, unexpired and enforceable title (free and
clear of all Encumbrances other than Company Permitted Encumbrances) to, all right, title and interest in and to all Company Owned IP that is solely owned or purported to be solely owned by a Company Group Member. Together the Company Group owns,
and has good, valid, unexpired and enforceable title (free and clear of all Encumbrances other than Company Permitted Encumbrances) to, all of the joint right, title and interest in and to all Company Owned IP that is jointly-owned or is purported
to be jointly-owned by a the Company Group Member and any other Person(s), and have been granted an exclusive license, from all other joint owners, to use and otherwise exploit, including to practice, patent, register, prosecute, maintain and
enforce, such Company Owned IP pursuant and subject to a valid and enforceable written agreement set forth in Section 3.12(b) of the Company Disclosure Schedule. The Company Group possesses adequate rights to use pursuant
to a valid and enforceable written agreement, all other Intellectual Property (i.e., all Intellectual Property to the extent it is not already within the scope of any of the foregoing representations and warranties set forth above in this
Section 3.12(b)) that is used or held for use in the conduct of its business as currently conducted or proposed to be conducted, including all Intellectual Property that is
non-exclusively licensed to a Company Group Member. No Person has any right of first refusal, option and/or other right to acquire any right, title or interest in or to, or has any other Encumbrance (other
than Company Permitted Encumbrances) with respect to, any Company Owned IP. In each case where the Company Group has acquired ownership of Registered IP from any other Person, such Company Group Member has obtained a valid and enforceable assignment
sufficient to irrevocably Transfer all rights, title and interest in and to such Registered IP to the Company Group Member, and the Company Group Member has recorded each such assignment with the United States Patent and Trademark Office and/or,
where required by Applicable Law, other applicable Governmental Authority. There is no Intellectual Property owned by any Third Party, and/or any other Intellectual Property, other than the Company Owned IP (A) that is required by the Company
Group, or is otherwise necessary, to conduct its business as currently being conducted, and/or (B) that is required by the Company Group, or is otherwise necessary, to conduct its business as currently proposed to be conducted with respect to
the Company Product or Service (other than, with respect to each of clauses (A) and (B), Intellectual Property owned by a Third Party that one or more Company Group Member have licensed or otherwise authorized to use pursuant to an Inbound
License (as defined below) listed in Section 3.12(f) of the Company Disclosure Schedule).
(c) Neither the
conduct of the business of any Company Group Member nor the Company Group (including through any consultant, employee or other Person that is or was working for a Company Group Member) has infringed upon or misappropriated or otherwise violated any
Intellectual Property rights of any other Person, and the conduct of its business as currently proposed to be conducted will not infringe upon, misappropriate or otherwise violate the Intellectual Property rights of any other Person. No Person has
asserted any written action, proceeding or other claim (or any oral claim) and, no such proceeding is pending or threatened, (i) challenging any Company Group Member’s ownership or other right, interest or title in or to, or the use,
validity, enforceability, patentability or registrability of, any of the Company Owned IP or any other Intellectual Property used or held for use in the business of any Company Group Member by or on behalf of any Company Group Member,
(ii) alleging infringement or misappropriation or other violation of any Intellectual Property rights by any Company Group Member, including any demand to license Intellectual Property of any Third Party, or (iii) involving any allegations
that a current or former employee, officer, director, consultant, contractor, service provider or advisor of any Company Group Member (each, a “Company Representative”) misappropriated, infringed or otherwise violated any
Intellectual Property rights of any Person that had previously employed or otherwise engaged such Company Representative (such Person, a “Former Employer”) or breached any agreement with its Former Employer in connection with such
Company Representative’s employment by, or other engagement with, or that otherwise relates to, any Company Group Member; and, with respect to clause (i) (solely with respect to Company Owned IP), clause (ii) and clause
(iii) immediately above, there is no reasonable basis for any such claim.
33
(d) None of the Company Group Members have assumed, or agreed to discharge or otherwise take
responsibility for, any existing or potential liability of another Person for infringement, misappropriation or other violation of any Intellectual Property rights and is not contractually obligated to do so. None of the Company Registered IP or
other Company Owned IP is subject to any pending or outstanding consent, settlement, injunction, directive, order, judgment, or other disposition of dispute that adversely impacts or restricts the use, ownership, transfer, registration or licensing
or other disposition of any such Intellectual Property by any Company Group Member, or otherwise adversely affects the validity, scope, use, registrability, or enforceability of any Company Intellectual Property.
(e) To the Company’s knowledge, no Person has infringed or misappropriated or otherwise violated, and no Person is currently infringing
or misappropriating or otherwise violating any Company Intellectual Property.
(f) Section 3.12(f) of the Company Disclosure
Schedule identifies each Contract pursuant to which any Intellectual Property is or has been licensed, sold, assigned, or otherwise conveyed or provided to any Company Group Member (other than non-exclusive
licenses to unmodified commercially available third-party software) (“Inbound Licenses”). Except with respect to the agreements listed in Section 3.12(f) of the Company Disclosure Schedule, none of the
Company Group Members are obligated under any Contract or other agreement to make any payments by way of royalties, fees, or otherwise to any owner or licensor of, or other claimant to, any Intellectual Property.
(g) Section 3.12(g) of the Company Disclosure Schedule lists each outbound license of Company Intellectual Property.
(h) Section 3.12(h) of the Company Disclosure Schedule lists all Contracts (not disclosed or required to be disclosed on
Sections 3.12(f) or 3.12(g) of the Company Disclosure Schedule) in effect as of the Agreement Date (i) relating to the Company Product or Service in any material respect, or (ii) containing any (A) restrictions,
in any material respect, on any Company Group Member’s rights to patent, register, enforce, use or otherwise exploit any Company Intellectual Property or other Intellectual Property used or held for use in the business of any Company Group
Member by or on behalf of any Company Group Member, including covenants not to sue and settlement and co-existence agreements, (B) right of first refusal, option or any other right to acquire any right,
title or interest, including any license, in or to any Company Owned IP or (C) payment due or payable in connection with any change in control of any Company Group Member or earn-out, milestone or other
contingent payments under any outbound license agreement or any Contract pursuant to which any Intellectual Property is or has been licensed, sold, assigned, or otherwise conveyed or provided to any Company Group Member.
(i) Each of the Company Group Members have taken reasonable security and other measures to protect the Company Owned IP, including measures
against unauthorized disclosure, to protect the secrecy, confidentiality, and value of its trade secrets and other technical information.
(j) All Company Representatives who have been involved in the creation or development of Intellectual Property for or otherwise on behalf of
any Company Group Member, have executed valid and enforceable written Contracts with a Company Group Member that include (i) present assignments to a Company Group Member of sole ownership of all inventions and other Intellectual Property
created for or otherwise on behalf of the Company Group, and (ii) obligations of confidentiality that require such Persons to maintain and protect the confidential information of the Company Group (an “IP Agreement”). Each of
the Company Group Members have secured written assignments from all Persons who contributed to the creation or development of any Company Intellectual Property owned or purported to be owned by a Company Group Member, of the rights to such Company
Intellectual Property arising from such contributions that may be owned by such Persons or that a Company Group Member does
34
not already own by operation of law. No Company Representative (i) has any right, title, license, claim, option or other similar interest whatsoever in or with respect to any Company
Intellectual Property owned or purported to be owned by a Company Group Member (or any other Company Intellectual Property), or (ii) is in material violation of any IP Agreement. To the Company’s knowledge, no Company Representative
or scientific advisor of a Company Group Member has excluded, in any agreement with a Company Group Member, any inventions, methods, processes, compounds, developments or other Intellectual Property that relate to the business of any Company Group
Member, including the Company Product or Service and/or Company Intellectual Property.
(k) No Governmental Authority or academic
institution has any rights in or to, ownership of, or right to royalties and/or other payments for, any Company Intellectual Property, nor has any Company Group Member used, directly or indirectly, any funding, grants, facilities, IP or personnel or
other similar resources of any such Person in connection with any research or development activities of a Company Group Member, including with respect to the Company Product or Service and/or Company Intellectual Property.
(l) The execution and delivery of this Agreement and the consummation of the transactions contemplated hereby (alone or in combination with
any other event), and the compliance with the provisions of this Agreement do not and will not conflict with, alter, or impair, any of the rights of the Company Group in or to any Company Intellectual Property or the validity, enforceability,
registrability, use, right to use, ownership, priority, duration, scope, or effectiveness of any Company Intellectual Property.
(m) Each
of the Company Group Members are and have for the past three (3) years been, in material compliance with (i) all Applicable Laws relating to the privacy, data protection and security of patient medical records and all other personal
information and data, including with respect to the processing, collection or use, including the storage, sharing, transfer, disposition, protection and processing thereof (including in connection with any preclinical or clinical trials conducted
with respect to the Company Product or Service (collectively, and together with all data and other information that is subject to any such Applicable Laws, “Personal Information”)), (ii) all publicly communicated privacy policies of
the Company Group concerning patient medical records and other Personal Information, and (iii) any contractual requirements to which any Company Group Member is subject that relate to any of the foregoing. To the Knowledge of the Company Group,
none of the Company Group Members has been subject to any security breaches (including a breach of security leading to the accidental or unlawful destruction, loss, alteration, unauthorized disclosure of, or access to, personal data transmitted,
stored or otherwise processed also on behalf of the Company Group Members) with respect to any Personal Information of the Company Group. Each of the Company Group Members have taken reasonable actions and implemented policies and procedures which
are designed to protect and maintain the security of all Personal Information of the Company Group from any unauthorized access or use and to detect any security breach. There have not been any written complaints or notices, or request for exercise
of data subjects’ rights or any audits, proceedings, investigations or claims conducted or asserted, or threatened by any Governmental Authority or other Person against any Company Group Member, and to the knowledge of the Company Group none
are pending, regarding any processing, collection, use, storage, disclosure, transfer or other disposition of any patient medical records or other Personal Information by or on behalf of any Company Group Member, including in connection with any
preclinical or clinical trials conducted with respect to the Company Product or Service, or the violation of any Applicable Laws relating to any of the foregoing.
35
(n) The software and related systems, including hardware, if any, owned, leased or licensed
by the Company Group in the conduct of its business as currently conducted (collectively, the “Company Systems”) are in good working order, and the Company Systems are backed up on a regular basis. The Company Group Members do not
own any proprietary software and all other software used by the Company Group is unmodified commercially available off-the-shelf software having a replacement cost and
annual license fee of less than €5,000 in the aggregate, and each of the Company Group Members have complied with the terms of all such software licenses. In the 18-month period preceding the Agreement Date, there have not been any security
breaches or other adverse events affecting any Company Systems.
(o) The Company Group Members have the right pursuant to a Company
Material Contract or, another valid and enforceable written agreement to, upon the Company’s request, duplicate, access and Transfer to each of the Company Group Members and/or their designees possession and control of any and all Company
Scientific Materials created or otherwise generated pursuant to such Company Material Contract or such other agreement.
3.13
Regulatory Matters.
(a) For the last seven (7) years, the Company Group Members have obtained all clearances, authorizations,
licenses and registrations required by any foreign or domestic Governmental Authorities (including, without limitation, the FDA and EMA) to permit the Company Group Members to conduct their business as currently conducted. The Company Group Members
have filed with the applicable regulatory authorities (including, without limitation, the FDA, the EMA or any other Governmental Authority performing functions similar to those performed by the FDA and EMA) all material filings, notices, responses
to notices, supplemental applications, declarations, listings, registrations, reports or submissions, including but not limited to adverse event reports required to be filed prior to the Agreement Date. All such filings, declarations, listings,
registrations, reports or submissions are, and were since filing (or were corrected in or supplemented by a subsequent filing), in compliance in all material respects with Applicable Laws and all formal filing and maintenance requirements, and no
material deficiencies have been asserted by any applicable Governmental Authority with respect to any such filings, declarations, listing, registrations, reports or submissions that have not been cured.
(b) For the last seven (7) years, all preclinical studies, and Clinical Trials and investigations sponsored or conducted by the Company
Group Members have been, and are being, conducted in material compliance with Applicable Laws, and the rules and regulations of any applicable Governmental Authority, including, as applicable, Good Laboratory Practices and Good Clinical Practices
requirements, and federal and state, national and supranational Applicable Laws, rules, regulations and guidance restricting the use, transfer and disclosure of individually identifiable health information and human subject or patient clinical
biological samples. The Company Group Members have not received any written notices or other correspondence from any institutional review board, ethics committee, safety monitoring committee or the FDA or any other supranational, foreign, federal,
state or local governmental or Regulatory Authority performing functions similar to those performed by the FDA with respect to any ongoing Clinical Trials or preclinical studies or tests requiring the termination, suspension or material modification
of such studies or tests, and to the Company’s knowledge, no such action has been threatened.
(c) The Company Group Members have not
received any written notice from any Regulatory Authority withdrawing or placing any Investigational New Drug application or equivalent application in other countries related to any Company Product or Service, on “clinical hold” or
requiring the termination or suspension or investigation of any preclinical studies or Clinical Trials conducted or sponsored by the Company Group, and to the Company’s knowledge there are no pending actions by any Regulatory Authority against
or affecting the Company Group with respect to any Company Product or Service or relating to or arising under any Applicable Laws relating to government health care programs, private health care plans or the privacy and confidentiality of patient
health information. The Company Group Members have made available to Buyer complete and correct copies of all material serious adverse event reports, non-clinical expedited safety reports and periodic adverse
event reports, and all material
36
Regulatory Authority communications and documents submitted by the Company Group Members to or received by the Company Group Members from the FDA, the European Commission, the EMA or any other
Regulatory Authority, including any material meeting minutes, scientific advice, inspection reports, warning letters and similar documents, relating to the Company Group, the conduct of the business of the Company Group or any Company Product or
Service.
(d) For the last seven (7) years, the Company Group Members have not, and to the Company’s knowledge, no Company
Representative has, (i) made an untrue statement of a material fact or fraudulent statement to the FDA or any other Governmental Authority, (ii) failed to disclose a material fact required to be disclosed to the FDA or any other
Governmental Authority, (iii) committed any other act, made any statement or failed to make any statement, that (in any such case) establishes, or would have established at the time such statement was made, a reasonable basis for the FDA to
invoke its Fraud, Untrue Statements of Material Facts, Bribery, and Illegal Gratuities Policy or any Governmental Authority to invoke any similar Applicable Law. To the Company’s knowledge, the Company Group Members are not the subject of any
pending or threatened investigation by the FDA pursuant to its Fraud, Untrue Statements of Material Facts, Bribery, and Illegal Gratuities Policy or any Governmental Authority to invoke any similar Applicable Law.
(e) The Company Group Members are in compliance and have been in compliance in all material respects with all Healthcare Laws applicable to
the operation of its business as currently conducted. The Company Group Members are not subject to any enforcement, regulatory or administrative proceedings against or affecting the Company Group relating to or arising under the FDCA or similar
Applicable Law, and no such enforcement, regulatory or administrative proceeding has been threatened in writing.
(f) The Company Group
Members have never been and none of the Company Representatives (while employed or engaged by the Company Group) has ever been, (i) debarred (under the provisions of the Generic Drug Enforcement Act of 1992, 21 U.S.C. §335a) or equivalent
foreign law, (ii) convicted of a crime for which a person can be debarred or (iii) indicted for a crime or otherwise engaged in conduct for which a person can be debarred or excluded from participating in any U.S. federal health care
programs or any equivalent foreign law.
(g) Any manufacture of a Company Product or Service used in any Clinical Trials conducted or
sponsored by or on behalf of the Company Group Members have been conducted in material compliance with the applicable requirements of current Good Manufacturing Practices and all other Applicable Laws.
(h) Except as disclosed on Section 3.13(h) of the Company Disclosure Schedule, there has not occurred (A) any
adverse event(s) in a nonclinical study or Clinical Trial or any other setting that are deemed by the study director or investigator in such nonclinical study or Clinical Trial to be at least possibly related to the Company Product or Service, or
(B) the failure of any Clinical Trial for the Company Product or Service to achieve one or more of its primary endpoints in any material respect, or (C) any failure of the Company Group or any Third Party service provider acting on behalf
of a Company Group Member to adhere to the requirements under the FDCA, the regulations and guidance documents of the FDA promulgated thereunder, the equivalent Applicable Laws and guidance of the EU, or the EU member states, or any other equivalent
foreign authority or any Governmental Permit (including the failure to possess or maintain the validity of any Governmental Permit), (x) relating to the investigational use and clinical trials of the Company Product or Service or (y) with
respect to the making of untrue or fraudulent statements, the failure to disclose information, or any failure to adhere to clinical protocols or informed consent requirements, which, individually or in the aggregate as to (A), (B) and (C) above
would at the time of such of such adverse event or failure, as applicable, reasonably be expected to prevent or materially delay the filing of or require the withdrawal, suspension or termination of an Investigational New Drug application in the
United States or equivalent application in other countries covering the Company Product or Service, or prevent or materially delay the Company Product or Service from obtaining Approval.
37
3.14 Compliance with Laws and Permits.
(a) The Company Group is in compliance with all Applicable Laws; provided, however, that Company makes no representation and warranty under
this Section 3.14(a) as to any Applicable Law as to which Company specifically makes a representation and warranty elsewhere in this Article III, including such representations and warranties as are qualified by
reference to information disclosed on a Company Disclosure Schedule.
(b) The Company Group holds all material permits, authorizations,
licenses and approvals however denominated from, and have made all material filings with, Governmental Authorities (the “Governmental Permits”), that are necessary and/or legally required to be held by it to conduct the Company
Business as presently conducted without any violation of Applicable Law, and all such Governmental Permits are valid and in full force and effect. Company has not received any written notice or other written communication from any Governmental
Authority regarding (i) any violation of the terms of any Governmental Permit or any failure to comply with any term or requirement of any Governmental Permit or (ii) any revocation, withdrawal, suspension, cancellation, termination or
modification of any Governmental Permit.
3.15 Certain Business Practices.
(a) Neither any Company Group Member nor any of its Representatives while acting on its behalf, has (i) used any funds for unlawful
contributions, gifts, entertainment or other unlawful expenses relating to political activity, (ii) made any unlawful payment to foreign or domestic government officials or employees, to foreign or domestic political parties or campaigns or
violated any provision of the U.S. Foreign Corrupt Practices Act of 1977 or any similar anti-corruption Applicable Laws, (iii) made any other unlawful payment or (iv) directly or indirectly,
unlawfully given or agreed to give any gift or similar benefit in any material amount to any customer, supplier, governmental employee or other Person who is or may be in a position to help or hinder Company or assist it in connection with any
actual or proposed transaction.
(b) The operations of the Company Group are and have been conducted at all times in compliance with money
laundering statutes in all applicable jurisdictions, the rules and regulations thereunder and any related or similar rules, regulations or guidelines, issued, administered or enforced by any Governmental Authority, and no Legal Proceeding involving
the Company Group with respect to the any of the foregoing is pending or threatened.
(c) None of the Company Group Members nor any of
director or officer of any of the Company or the US Subsidiary, or any other Representative acting on behalf of the Company Group is currently identified on the Specially Designated Nationals list or otherwise currently subject to any U.S. sanctions
administered by the Office of Foreign Assets Control of the U.S. Treasury Department (“OFAC”), and the Company Group has not, directly or indirectly, used any funds, or loaned, contributed or otherwise made available such funds to
any Subsidiary, joint venture partner or other Person, in connection with any sales or operations in any country sanctioned by OFAC or for the purpose of unlawfully financing the activities of any Person currently subject to, or otherwise in
violation of, any U.S. sanctions administered by OFAC in the last five fiscal years.
38
3.16 Certain Transactions and Agreements. None of the officers or directors of
Company, and no Shareholder holding 10% or more of the outstanding shares of Company Stock, nor any immediate family member of an officer or director of Company, (a) has a direct ownership interest of more than 10% of the equity ownership of
any firm or corporation that competes with, or does a material amount of business with the Company Group or (b) is a party to any transaction with the Company Group that would be required to be disclosed pursuant to Item 404 of Regulation S-K promulgated by the SEC.
3.17 Employees, ERISA and Other Compliance.
(a) The employees of the Company Group are only those listed in Section 3.17(a) of the Company Disclosure Schedule
(the “Employees”), which also indicates their job title, country, location, employer, date of commencement of employment, pension entitlement, salary, bonus potential, and classification under the Applicable Law. There is no person
that, by reason of his/her relationship with each of the Company Group or otherwise, can successfully claim the status of employee of any of the Company Group or, with respect to Employees located outside of the United States (the “Non U.S.
Employees”), the recognition of his/her quality as employee of any of the Company Group. None of the Employees hired under a fixed term contract are entitled to validly claim the existence of an open-ended term employment relationship with
any of the Company Group.
(b) All the Employees of each of the Company Group are recorded in the appropriate books and records of the
Company Group by which they are employed, and such books and records contain all details required to be contained therein under Applicable Laws.
(c) The Employees and each of the employees of the Company Group whose employment agreement has been terminated for any reason whatsoever (the
“Former Employees”) are and have been correctly enrolled (for Non-U.S. Employees only) and have been accurately paid for the employment services carried out in the course of the respective
contracts in accordance with all provisions of Applicable Laws and employment contracts (for all Former Employees).
(d) The employment of
the Employees is governed by the provisions of the bargaining agreements set forth in Section 3.17(d) of the Company Disclosure Schedule. Except for such bargaining agreements, the Company Group are not legally bound by any
other collective bargaining agreement (including works agreements and company practices) or labour union contract applicable to Employees and no collective bargaining agreement, collective agreement or labour union contract is being negotiated by
the Company Group that would be applicable to the Employees.
(e) The Company Group currently is in material compliance, with all
Applicable Laws relating to employment, employment practices, immigration, wages, hours, and terms and conditions of employment, including employee compensation matters (including correct payment of social security contributions), and has correctly
classified employees as exempt employees and nonexempt employees if required under the U.S. Fair Labor Standards Act or any other Applicable Laws.
(f) All independent contractors (including agents and consultants) providing services to the Company Group have been properly classified as
independent contractors for purposes of U.S. federal and applicable state, provincial or non-U.S. Tax laws, laws applicable to employee benefits and other Applicable Law. No officer or executive or key
consultants of any Company Group Member have given written notice to such Company Group Member of an intention to terminate his or her employment or consulting relationship with such Company Group Member. No claims have been filed, nor is any
litigation pending and/or threatened vis-à -vis the Company Group with respect to any objections, salary differences requests (with respect to Non-U.S. Employees only), claims related to the payment of social security contributions, insurance premiums, bonuses and tax withholdings arising from the aforesaid agreements.
39
No claims have been filed and/or threatened, nor any litigation is pending and/or threatened vis-à-vis the
Company Group by the employees of the aforementioned services, outsourcing, consultancy, agency and staff leasing contractors.
(g) The
relationships with all consultants, agents, business procurement persons, whose agreement was terminated before the date hereof for any reason whatsoever are and have been established and conducted in accordance with the applicable laws and
regulations and in such a way that no such person is entitled to validly claim that he/she is an employee of the Company Group.
(h) The
Company Group has fulfilled the obligations concerning HSE matters, including any obligation provided for by Applicable Law concerning the prevention of injuries, hygiene, safety at the place of work, fire safety and ecology, and other laws
(including any applicable environmental Law).
(i) There are no Legal Proceedings against the Company Group pending or threatened to be
brought or filed, by or with any Governmental Authority or otherwise in connection with the employment or engagement of any current or former employee, director, officer, applicant, contractor, or other service provider of the Company Group,
including, without limitation, any claim relating to unfair labor practices, employment discrimination, harassment, retaliation, equal pay, wage or hours violations, unpaid wages, unpaid commissions, or wrongful termination.
(j) No Company Group Member is now subject to a union organizing effort to organize or represent any group of employees of such Company Group
Member. There are no unresolved labor disputes pending before the labour court between any Company Group Member and any Employee and /or Former Employee of such Company Group Member and/or trade union association and/or any public authorities. There
are no requests for payments notified in writing to the Company Group and issued by public authorities competent for ensuring compliance with social security, health care assistance and employment laws.
(k) As used in this Section 3.17, “Company Benefit Arrangement” means each “employee benefit
plan” as defined in Section 3(3) of ERISA (whether or not the plan is subject to ERISA) and each material plan or arrangement (written or oral) providing for insurance coverage (including any self-insured arrangements), life and health
benefits (including medical, mental health, dental, vision and hospitalization), vacation benefits, severance benefits, supplemental unemployment benefits, material fringe benefits, disability benefits, death benefits, hospitalization benefits,
retirement benefits, deferred compensation, profit-sharing, tax equalization, bonuses, stock options, stock purchase, phantom stock, stock appreciation or other forms of incentive compensation or post-retirement insurance, compensation or benefits
for employees, consultants or directors that is entered into, maintained or contributed to by Company or the US Subsidiary or pursuant to which Company has any liability. The Employees severance indemnity (“trattamento di fine
rapporto”) and any other indemnities due to Employees have been correctly reported and are accurately valued in the Company Group accounting books and records pursuant to the Applicable Laws, the applicable collective bargaining agreements
and the individual employment agreements, and have been correctly paid to former employees at the termination of the employment relationship and/or accrued by the Company Group and/or allocated in the social security funds.
(l) With respect to each Company Benefit Arrangement, the Company has delivered or made available to Buyer correct and complete copies of, if
applicable, (i) the current plan document and any trust agreements, insurance contracts or other funding vehicles (ii) the most recent summary plan description, (iii) the most recent annual report on Form 5500 filed with the Internal
Revenue Service, (iv) the most recent actuarial valuation (v) the most recent determination or opinion letter issued by the Internal Revenue Service (or applicable comparable Governmental Authority), and (vi) all material
correspondence to or from any Governmental Authority received in the last three (3) years with respect to any Company Benefit Arrangement. The Company has also delivered or made available a written description of any Company Benefit Arrangement
to the extent not set forth in a written document.
40
(m) Neither the Company nor the US Subsidiary or ERISA Affiliates sponsored, maintained,
contributed to or was required to contribute to, or has or had any liability in respect of, at any point during the six (6) year period prior to the date hereof, a multiemployer pension plan (as defined in Section 3(37) of ERISA) (a
“Multiemployer Plan”) or other pension plan, in each case, that is subject to Title IV of ERISA. No circumstance or condition exists that would reasonably be expected to result in an actual obligation of the Company to pay money to any
Multiemployer Plan or other pension plan that is subject to Title IV of ERISA and that is maintained by an ERISA Affiliate of the Company. For purposes of this Agreement, “ERISA Affiliate” means any entity (whether or not
incorporated) other than the Company that, together with the Company, is considered under common control and treated as one employer under Section 414(b), (c), (m) or (o) of the Code. No Company Benefit Arrangement is a “multiple
employer welfare arrangement” (as defined in Section 3(40) of ERISA).
(n) Neither the execution and delivery of this Agreement
by the Company nor the consummation of the transactions contemplated by this Agreement (either alone or in combination with another event) will (i) entitle any employee, director, officer or independent contractor of the Company to severance
pay or any material increase in severance pay, (ii) accelerate the time of payment or vesting, or materially increase the amount of compensation due to any such employee, director, officer or independent contractor, (iii) directly or
indirectly cause the Company to Transfer or set aside any assets to fund any material benefits under any Company Benefit Arrangement, (iv) otherwise give rise to any material liability under any Company Benefit Arrangement, (v) limit or
restrict the right to merge, materially amend, terminate or Transfer the assets of any Company Benefit Arrangement on or following the Closing, (vi) require a “gross-up,” indemnification for, or
payment to any individual for any Taxes imposed under Section 409A or Section 4999 of the Code or any other Tax or (vii) result in the payment of any amount that could, individually or in combination with any other such payment,
constitute an “excess parachute payment” as defined in Section 280G(b)(1) of the Code that would result in the imposition of an excise tax under Section 4999 of the Code.
(o) With respect to each Company Benefit Arrangement:
(i) such Company Benefit Arrangement has been established, registered, qualified, funded, invested and administered in accordance with, and
is in good standing under, all Applicable Laws that are applicable to such Company Benefit Arrangement and the terms of such Company Benefit Arrangements in all material respects; and
(ii) all contributions or premiums required to be made by the Company Group under the terms of such Company Benefit Arrangements or by
Applicable Law have been made in a timely fashion; and
(iii) had no withdrawals, applications or transfers of assets from such Company
Benefit Arrangements except in accordance with the terms of such Company Benefit Arrangements and Applicable Law.
(p) As of the Agreement
Date, no such Company Benefit Arrangement, nor any related trust or other funding medium thereunder, is subject to any pending or threatened investigation, examination or other legal proceeding, initiated by any Governmental Authority or by any
other Person (other than routine claims for benefits).
41
(q) All non-competition agreements or other
restrictive covenants executed by the Company Group and any of the Employees or Former Employees or Parties/Persons have been entered into in compliance with Applicable Law and collective agreements or collective bargaining agreements.
(r) All Transfers of business of which the Company Group are, or have been part, have been entered into and have been performed in accordance
with Applicable Law.
(s) No collective dismissal procedures have been implemented by the Company Group in the last year.
(t) All the resignations given by the Former Employees have been given/ratified in accordance with Applicable Law.
(u) All settlement agreements entered into by the Company Group and Employees, Former Employees and any other Third Parties/Persons have been
executed in accordance with the Applicable Law and all the payments (if any) provided therein have been paid by the Company Group.
(v)
Each Company Benefit Arrangement that is intended to be qualified under Section 401(a) of the Code has received a favorable determination or approval letter from the IRS with respect to such qualification, or is the subject of a favorable
advisory or opinion letter as to its qualification, issued by the IRS, or has time remaining for application to the IRS for a determination of the qualified status of such Company Benefit Arrangement for any period for which such Company Benefit
Arrangement would not otherwise be covered by an IRS determination. To the knowledge of the Company, no action or event has occurred since the date of the most recent determination or advisory or opinion letter relating to any such Company Benefit
Arrangement that would adversely affect the tax-qualified status of any such plan or related trust.
(w) Except as required by Section 4980B of the Code or any other Applicable Law, no Company Benefit Arrangement provides post-employment
medical or health benefits.
3.18 Transaction Expenses.
(a) Except as set forth on Section 3.18 of the Company Disclosure Schedule, neither Company nor any other Company
Group Member is obligated for the payment of any brokerage, finders or other similar fees or expenses of any investment banker, broker, finder or similar party in connection with the origin, negotiation or execution of this Agreement or in
connection with the Transactions.
(b) The legal and accounting advisors, independent auditors, and any other Persons, to whom the Company
Group expects as of the Agreement Date that it will owe fees and expenses that will constitute Transaction Expenses are set forth on Section 3.18(b) of the Company Disclosure Schedule.
3.19 Insurance. There is no claim pending under any insurance policies under which any Company Group Member is insured as to which
coverage has been questioned, denied or disputed by the underwriters of such policies. All premiums due and payable under all such insurance policies have been timely paid, and the Company Group is otherwise in compliance with the terms of such
policies. Company has not received any written threatened termination of, or material premium increase with respect to, any of such insurance policies. Neither Company nor the US Subsidiary has been refused any insurance with respect to any aspect
of the Company Business, nor has its coverage been limited by any insurance carrier to which it has applied for insurance or with which it has carried insurance.
42
3.20 Environmental Matters.
(a) (i) The Company Group is in compliance with all Environmental Laws (as defined below), which compliance includes the possession by the
Company Group of all material Governmental Permits required under applicable Environmental Laws and compliance with the terms and conditions thereof, (ii) the Company Group has not received any written notice or other written communication from
a Governmental Authority that alleges that the Company Group is not in compliance with any Environmental Laws, and (iii) there are no circumstances that may prevent or interfere with the compliance by Company with any Environmental Laws.
(b) For the sole purpose of this Section 3.20: (i) “Environmental Laws” means any U.S., Canadian or
Italian federal, state, regional, provincial, local, or foreign statute, decree, ordinance, law, regulation or other legal requirement relating to pollution or protection of human health or the environment (including ambient air, surface water,
ground water, land surface or subsurface strata), and/or the emission, discharge, release or threatened release of Materials of Environmental Concern or otherwise relating to the manufacture, processing, distribution, use, treatment, storage,
disposal, transport or handling of Materials of Environmental Concern; and (ii) “Materials of Environmental Concern” means chemicals, pollutants, contaminants, wastes (including the “special wastes” under the Italian
Legislative Decree no. 152/2006), toxic substances, petroleum and petroleum products and any other such substance that is currently regulated under any applicable Environmental Laws or that is otherwise a material danger to health, reproduction or
the environment.
3.21 Export Control Laws. The Company Group is in compliance in all respects with all Export Control Laws
applicable to it: (a) the Company Group has obtained all export licenses and other approvals required for its exports of products, software and technologies required by any Export Control Law and all such approvals and licenses are in full
force and effect; (b) the Company Group is in compliance with the terms of such applicable export licenses or other approvals; and (c) there are no claims pending or threatened in writing against any Company Group Member with respect to
such export licenses or other approvals.
3.22 Accuracy of Information Supplied. Company represents and warrants that none of the
information supplied by Company for inclusion or incorporation by reference: (a) in any current report on Form 8-K, and any exhibits thereto or any other report, form, registration or other filing made with any Governmental Authority with
respect to the Transactions, or (b) in the mailings or other distributions to Buyer Stockholders and/or prospective investors with respect to the consummation of the Transactions or in any amendment to any of documents identified in clauses
(a) and (b), will, when filed, made available, mailed or distributed, as the case may be, contain any untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary in order to make the
statements therein, in light of the circumstances under which they are made, not misleading, but in each case only to the extent that the untrue statement or alleged untrue statement or omission or alleged omission was made in reliance upon and in
conformity with the information provided. Notwithstanding the foregoing, Company makes no representation, warranty or covenant with respect to any information supplied by or on behalf of Buyer or its Affiliates.
3.23 Receivables. All notes and accounts receivable of the Company and of the US Subsidiary have arisen from bona fide transactions in
the Ordinary Course of Business and are reflected properly in the respective books and records of the Company and are existing, valid and collectable (esigibili) on the relevant due date for the full face value.
3.24 Related Party Contracts. Neither the Company nor the US Subsidiary have executed any agreement with a Related Party.
43
3.25 Anti-Corruption. The Company has duly adopted and implemented, and maintains in
full force and effect, corporate governance structures and risk prevention systems (including the organizational models and the supervisory body) envisaged under the Italian legislative Decree no. 231 dated June 8, 2001, as subsequently amended
(“Decree 231”) and such structures and systems adequately address the requirements and provisions set forth under the Decree 231. The Company has adopted a “231 Model” (Modello 231) and comply with the
provisions set forth under the Decree 231. The Company has not committed any of the crimes provided under the Decree 231 and, in general, has not breached any anti-bribery Laws applicable to its business and operations. The Company has not received
any written notice from any Governmental Authority of non-compliance with the Decree 231 and, in general, with anti-bribery Applicable Laws, nor is subject to any litigation, pending or threatened in writing,
with respect to any alleged non-compliance or violation thereof. No director, officer, employee, representative, or any other Person, duly authorized to act for or on behalf of the foregoing (individually and
collectively) of the Company has kept any behavior or committed any action, also by way of omission, which may result in the Company has been held liable under the Decree 231. There are no circumstances that may give rise to any proceedings or
investigations being brought for breach of the Decree 231.
3.26 Extraordinary Transactions. Neither the Company nor the US
Subsidiary are a party to any contract relating to the acquisition, or the Transfer of any business, assets (other than assets acquired in the Ordinary Course of Business in accordance with past practice) or share capital or other capital stock
shareholdings in any Person (whether by merger or sale of shares, or sale of assets or other), and did not make any extraordinary transaction.
ARTICLE IV
REPRESENTATIONS AND WARRANTIES OF SHAREHOLDERS
Each Shareholder, severally solely as to itself and not jointly and severally, represents and warrants to Buyer that the statements contained
in this Article IV are true and correct on and as of the Agreement Date and as of the Closing Date:
4.1 Power, Authorization and
Validity.
(a) Power and Authority. Such Shareholder has all requisite power and authority to enter into, execute, deliver and
perform its obligations under this Agreement and the Company Ancillary Agreements to which such Shareholder is, or will be, a party and to consummate the Transactions. If such Shareholder is not an individual, the Transactions and the execution,
delivery and performance by such Shareholder of this Agreement and the Company Ancillary Agreements to which such Shareholder is, or will be, a party, and all other agreements, transactions and actions contemplated hereby, have been duly and validly
approved and authorized by all requisite action on the part of such Shareholder.
(b) No Consents. No consent, approval, order or
authorization of, or registration, declaration or filing with, any Governmental Authority, or any other Person, governmental or otherwise, is necessary or required to be made or obtained by any Shareholder to enable such Shareholder to lawfully
execute and deliver, enter into, and perform its obligations under this Agreement or the Company Ancillary Agreements to which such Shareholder is, or will be, a party or to consummate the Transactions, except where the lack of consent, approval,
order, authorization or failure to make such registration, declaration or filing would not, individually or in the aggregate, materially impair such Shareholder’s performance of its obligations under this Agreement or such Company Ancillary
Agreements or ability to consummate the Transactions.
44
(c) Enforceability. This Agreement and the Company Ancillary Agreements to which such
Shareholder is, or will be, a party have, or when executed will have, been duly executed and delivered by such Shareholder. This Agreement and the Company Ancillary Agreements to which such Shareholder is, or will be, a party, is, or when executed
by such Shareholder shall be, assuming the due authorization, execution and delivery by each other party hereto, valid and binding obligations of such Shareholder, enforceable against such Shareholder in accordance with its terms, subject to the
Enforceability Exceptions.
4.2 Title to Company Stock. Such Shareholder owns of record and beneficially the Company Stock set
forth next to such Shareholder’s name on Schedule 1 of the Company Shareholder Letter; and such shares of Company Stock represent all of the issued and outstanding capital stock of the Company owned by such Shareholder. Such Shareholder
has good and valid title to such shares of Company Stock, free and clear of all Encumbrances, except for restrictions under the Company Charter Documents. The Company Stock has not been object of donation (donazione) or Transfer mortis
causa (successione).
4.3 Brokerage. There are no claims for brokerage commissions, finders’ fees or similar
compensation to other Persons in connection with the Transactions based on any arrangement or agreement to which such Shareholder is a party or to which such Shareholder is subject for which Buyer or the Company Group could become obligated after
the Closing.
4.4 Non-U.S. Status. Such Shareholder is not a “United States
person” within the meaning of Section 7701(a)(30) of the Code.
4.5 Transaction Securities.
(a) Accredited Investor. Such Shareholder is an “accredited investor” within the meaning of SEC Rule 501 of Regulation D
promulgated under the Securities Act, as in effect as of the date of this Agreement (an “Accredited Investor”) and has delivered to Buyer a duly executed accredited investor questionnaire in the form attached hereto as Exhibit
C, certifying such Shareholder’s status as an Accredited Investor (each, an “Accredited Investor Questionnaire”).
(b) No Registration. Such Shareholder understands that any Transaction Securities have not been registered under the Securities Act, by
reason of a specific exemption from the registration provisions of the Securities Act, the availability of which depends upon, among other things, the bona fide nature of the investment intent and the accuracy of such Shareholder’s
representations as expressed herein or otherwise made pursuant hereto.
(c) No Solicitation. Such Shareholder was not, at any time,
presented with or solicited by any publicly issued or circulated newspaper, mail, radio, television or other form of general advertising or solicitation in connection with the offer, sale, exchange, and/or purchase of the Transaction Securities,
whether by Buyer, its agents or otherwise.
(d) Investment Intent. Except for the distribution of the Transaction Securities to its
partners or members (if such Shareholder is a venture capital, private equity or other investment fund), such Shareholder is acquiring such Transaction Securities for investment for its, his or her own account, not as a nominee or agent, and not
with the view to, or for resale in connection with, any distribution thereof, and such Shareholder has no present intention of selling, granting any participation in, or otherwise distributing the same and, upon transfer of Transaction Securities to
such Shareholder, no one other than such Shareholder will have any beneficial ownership of or in any of such Transaction Securities. Such Shareholder does not have any Contract, undertaking, agreement or arrangement with any Person to sell, transfer
or grant participation to such Person or to any third Person with respect to any of the Transaction Securities it acquires in connection with the Transactions (other than, if such Shareholder is a venture capital, private equity or other investment
fund, pursuant to the terms of its limited partnership agreement, limited liability company agreement or other organizational documents, including any “side letters” thereto or similar agreements with its investors).
45
(e) Investment Experience. Such Shareholder has sufficient experience in evaluating
and investing in securities of companies and acknowledges that such Shareholder can protect its, his or her own interests. Such Shareholder has such knowledge and experience in financial and business matters so that such Shareholder is capable
of evaluating the merits and risks of owning the Transaction Securities.
(f) Speculative Nature of the Shares. Such Shareholder
understands and acknowledges that ownership of the Transaction Securities is highly speculative and involves substantial risks. Such Shareholder can bear the economic risk related to owning the Transaction Securities and is able, without
impairing such Shareholder’s financial condition, to hold the Transaction Securities for an indefinite period of time and to suffer a complete loss of the value of the Transaction Securities acquired by such Shareholder in connection with the
Transactions.
(g) Opportunity to Discuss. Such Shareholder has had an opportunity to ask questions of, and receive answers from,
Buyer and its Representatives concerning this Agreement or any Ancillary Agreement to which such Shareholder is or will be a party (each, a “Shareholder Related Agreement”), and the transactions contemplated hereby and
thereby, and such Shareholder believes that it, he or she has received all information such Shareholder considers necessary for deciding whether to accept the Transaction Securities as consideration payable by Buyer in connection with the
Transactions. Such Shareholder acknowledges that it, he or she is relying solely on its, his or her own counsel and not on any statements or representations of Buyer or the Company or their respective Representatives for legal advice with
respect to this Agreement and any Shareholder Related Agreements, and the transactions contemplated hereby and thereby, other than the representations and warranties of Buyer set forth in Article V of this Agreement. Such Shareholder further
acknowledges that Buyer has advised such Shareholder that Buyer, its Affiliates, and its and their respective directors, officers, employees, advisors, counsel, agents, and other representatives possess
non-public information regarding Buyer’s business, prospects, and actual or forecasted financial or operating performance not known to such Shareholder which such Shareholder may deem material to its
decision to accept the Transaction Securities if such Shareholder were provided with such information (such information, the “Buyer Excluded Information”).
(h) Residency. Such Shareholder has provided Buyer with its current address of residency and agrees to provide such additional
information to Buyer as is necessary to issue the Transaction Securities to such Shareholder in accordance with this Agreement and establish a book entry position with respect to such shares with Buyer’s transfer agent.
(i) Rule 144. Such Shareholder acknowledges that any Transaction Securities that such Shareholder acquires in connection with the
Transactions must be held indefinitely unless subsequently registered under the Securities Act or an exemption from such registration is available. Such Shareholder is aware of the provisions of Rule 144 promulgated under the Securities Act
which permit resale of restricted securities subject to the satisfaction of certain conditions, which may include, among other things, the availability of certain current public information about Buyer; the resale occurring not less than a specified
period after a party has acquired the security to be sold; the number of shares being sold during any three-month period not exceeding specified limitations; the sale being effected through a “brokers’ transaction,” a transaction
directly with a “market maker” or a “riskless principal transaction” (as those terms are defined in the Securities Act or the Securities Exchange Act of 1934, as amended, and the rules and regulations promulgated thereunder); and
the filing of a Form 144 notice, if applicable. Such Shareholder acknowledges and understands that Buyer may not be satisfying the current public information requirement of Rule 144 at the time such Shareholder wishes to sell the Transaction
Securities that such Shareholder acquires in connection with the Transactions, and that, in such event, such Shareholder may
46
be precluded from selling such securities under Rule 144, even if the other applicable requirements of Rule 144 have been satisfied. Such Shareholder acknowledges that, in the event the
applicable requirements of Rule 144 are not met, registration under the Securities Act or an exemption from registration will be required for disposition of any Transaction Securities that such Shareholder acquires in connection with the
Transactions. Such Shareholder understands that, although Rule 144 is not exclusive, the SEC has expressed its opinion that persons proposing to sell restricted securities received in a private offering other than in a registered offering or
pursuant to Rule 144 will have a substantial burden of proof in establishing that an exemption from registration is available for such offers or sales and that such persons and the brokers who participate in the transactions do so at their own risk.
(j) Legends . Such Shareholder understands that (i) the Transaction Securities constitute “restricted securities”
under the Securities Act, and may not be transferred absent registration under the Securities Act or an exemption therefrom, and any such transfer shall be subject to compliance with applicable state securities Laws, and (ii) all Transaction
Securities shall bear a legend or legends referencing restrictions applicable to such shares under applicable securities Laws and under this Agreement, in substantially the form set forth in Section 2.6.
(k) Material Nonpublic Information . Such Shareholder hereby confirms that it is aware, and that its Representatives have been advised,
that the United States securities laws prohibit any Person who has material nonpublic information about a company from purchasing or selling securities of such company or from communicating such information to any other Person under circumstances in
which it is reasonably foreseeable that such Person may purchase or sell such securities.
(l) Stop-Transfer Instructions. Such
Shareholder agrees that, in order to ensure compliance with the restrictions imposed by this Agreement, Buyer may issue appropriate “stop-transfer” instructions to its transfer agent, and if Buyer acts as its own transfer agent, it may
make appropriate notations to the same effect in its own records. Buyer will not be required (a) to transfer on its books any Transaction Securities that have been sold or otherwise transferred in violation of any of the provisions of this
Agreement, or (b) to treat as owner of such Transaction Securities, or to accord the right to vote or receive dividends, to any purchaser or other transferee to whom such Transaction Securities have been so transferred. Such Shareholder further
understands and agrees that Buyer may require written assurances, in form and substance satisfactory to counsel for Buyer (which may include a requirement that such Shareholder’s counsel provide a legal opinion acceptable to Buyer), before
Buyer effects any future transfers of Transaction Securities.
(m) Stock Certificates. Such Shareholder understands that
Buyer’s current policy is not to issue stock certificates representing shares of its capital stock, and all new issuances of capital stock are reflected on Buyer’s books and records in book entry only, with appropriate notations reflecting
the applicable legends.
ARTICLE V
REPRESENTATIONS AND WARRANTIES OF BUYER
Except as set forth (a) in a numbered or lettered section of the Buyer Disclosure Schedule that correspond to the sections of Article
V of this Agreement and other sections of the Buyer Disclosure Schedule to the extent (i) such information is cross-referenced in another part of the Buyer Disclosure Schedule or (ii) the relevance to such other section of Article
V of this Agreement is reasonably apparent on the face of such disclosure or (b) Buyer’s SEC Documents that are available on the SEC’s website through EDGAR prior to the Agreement Date (other than disclosures in the “Risk
Factors” or “Forward Looking Statements” sections of any SEC Document to the extent that such disclosure is predictive or forward-looking in nature), Buyer represents and warrants to the Company Parties that the statements contained
in this Article V are true and correct on and as of the Agreement Date and as of the Closing Date:
47
5.1 Organization and Good Standing. Buyer is a corporation duly organized, validly
existing and in good standing under the laws of the State of Delaware and has the requisite corporate power and authority to own, operate and lease its properties and to carry on its business as now conducted and as presently proposed to be
conducted. Buyer is duly qualified or licensed to do business, and is in good standing, in each jurisdiction where the character of the properties owned, leased or operated by it or the nature of its activities makes such qualification or licensing
necessary, except where the failure to be so qualified or licensed or to be in good standing would not have a Material Adverse Effect on Buyer. Buyer has provided to Company correct and complete copies of the Buyer Organizational Documents. Buyer is
not in violation of the Buyer Organizational Documents.
5.2 Subsidiaries. Section 7.2 of the Buyer
Disclosure Schedule sets forth each Subsidiary of Buyer as of the Agreement Date. Except as set forth in Section 7.2 of the Buyer Disclosure Schedule, all the outstanding shares of capital stock of, or other equity or
voting interests in, each Subsidiary of Buyer are duly authorized, have been validly issued and are fully paid and nonassessable and are owned by Buyer, a wholly-owned Subsidiary of Buyer, or Buyer and another wholly-owned Subsidiary of Buyer, free
and clear of all material Encumbrances of any kind or nature whatsoever, other than Buyer Permitted Encumbrances, except for restrictions imposed by applicable securities laws. Except as set forth in Section 7.2 of the
Buyer Disclosure Schedule, other than the Subsidiaries of Buyer, neither Buyer nor any of its Subsidiaries owns any capital stock of, or other equity or voting interests of any nature in, or any interest convertible, exchangeable or exercisable for,
capital stock of, or other equity or voting interests of any nature in, any other Person.
5.3 Power, Authorization and Validity.
(a) Buyer has all requisite corporate power and corporate authority to enter into, execute, deliver and perform its obligations under
this Agreement and the Buyer Ancillary Agreement and to consummate the Transactions. The Transactions and the execution, delivery and performance by Buyer of this Agreement, each of the Buyer Ancillary Agreement and all other agreements,
transactions and actions contemplated hereby or thereby have been duly and validly approved and authorized by all requisite corporate action on the part of Buyer and no other corporate proceedings on the part of Buyer are necessary to approve this
Agreement and the Buyer Ancillary Agreement or to authorize or consummate the Transactions.
(b) No Consents. No consent, approval,
order or authorization of, or registration, declaration or filing with, any Governmental Authority is necessary or required to be made or obtained by Buyer to enable Buyer to lawfully execute and deliver, enter into, and perform its obligations
under this Agreement and the Buyer Ancillary Agreement or to consummate the Transactions, except where the breach or violation or the lack of consent, approval, notice, filing, declaration or registration would not, individually or in the aggregate,
reasonably be expected to have a material adverse effect on Buyer’s ability to consummate the Transactions.
(c)
Enforceability. This Agreement has been duly executed and delivered by Buyer. This Agreement and the Buyer Ancillary Agreement is, or when executed by Buyer, shall be, assuming the due authorization, execution and delivery by each other party
hereto or thereto, valid and binding obligations of Buyer, enforceable against Buyer in accordance with their respective terms, subject to the Enforceability Exceptions.
48
5.4 No Conflict. Neither the execution and delivery of this Agreement nor the Buyer
Ancillary Agreement by Buyer, nor the occurrence of the Closing, shall conflict with, result in a termination, breach or violation of (with or without notice or lapse of time, or both), or constitute a default, or require the consent, release,
waiver or approval of any Third Party, under: (a) any provision of the Buyer Organizational Documents or; (b) any Applicable Law applicable to Buyer, or any of its assets or properties; or (c) any Contract to which Buyer is a party or
by which Buyer, or any of its material assets or properties are bound, except in each case, where such conflict, termination, breach, violation, default, consent, or lack of consent, release, waiver or approval would not, individually or in the
aggregate, reasonably be expected to have a material adverse effect on Buyer’s ability to consummate the Transactions.
5.5
Litigation. There is no action, suit, arbitration, mediation, proceeding, claim or investigation pending against Buyer before any Governmental Authority and no such action, suit, arbitration, mediation, proceeding, claim or investigation has
been threatened in writing that would, individually or in the aggregate, reasonably be expected to have a material adverse effect on Buyer’s ability to consummate the Transactions. There is no judgment, decree, injunction, rule or order of any
Governmental Authority, arbitrator or mediator outstanding against Buyer. Buyer has no action, suit, arbitration, mediation, proceeding, claim or investigation pending against any Governmental Authority or other Person.
5.6 Capitalization.
(a)
The authorized capital stock of Buyer as of the Agreement Date consists of 100,000,000 shares of Buyer Common Stock and 10,000,000 shares of Buyer Preferred Stock, 100,000 of which is designated as Series A Junior Participating Preferred Stock (as
defined in the Buyer Certificate of Incorporation). As of July 12, 2023 (the “Buyer Capitalization Date”): (i) 36,327,995 shares of Buyer Common Stock were issued and outstanding and (ii) no shares of Buyer Preferred Stock
were issued and outstanding All outstanding shares of Buyer Common Stock are duly authorized, validly issued, fully paid and non-assessable and not subject to or issued in violation of any purchase option,
right of first refusal, preemptive right, subscription right or any similar right under any provision of the DGCL, Applicable Law or the Buyer Organizational Documents or any Contract to which Buyer is a party. None of the outstanding Buyer
Securities has been issued in violation of any applicable securities laws. Other than IT Buyer, Buyer does not have any Subsidiaries or own any equity interests in any other Person.
(b) Except as set forth in Section 5.6(a) of the Buyer Disclosure Schedule and other than the shares of Buyer Common
Stock that have become outstanding after the Buyer Capitalization Date that were reserved for issuance as set forth in Section 4.2(a), as of the date hereof, there are no (i) outstanding options, warrants, puts, calls, convertible
securities, preemptive or similar rights, (ii) bonds, debentures, notes or other Debt having general voting rights or that are convertible or exchangeable into securities having such rights or (iii) subscriptions or other rights,
agreements, arrangements, Contracts or commitments of any character (other than this Agreement and the Buyer Ancillary Agreement), (A) relating to the issued or unissued shares of capital stock of Buyer or any other securities of Buyer,
(B) obligating Buyer to issue, Transfer, deliver or sell or cause to be issued, Transferred, delivered, sold or repurchased any options or shares or securities convertible into or exchangeable for such shares or any other securities of Buyer,
or (C) obligating Buyer to grant, extend or enter into any such option, warrant, call, subscription or other right, agreement, arrangement or commitment for such shares or any other securities of Buyer. Other than as expressly set forth in this
Agreement, there are no outstanding obligations of Buyer to repurchase, redeem or otherwise acquire any shares of capital stock of Buyer or any other securities of Buyer or to provide funds to make any investment (in the form of a loan, capital
contribution or otherwise) in any Person. Except as set forth in Section 5.6(b) of the Buyer Disclosure Schedule, there are no shareholders agreements, voting trusts or other agreements or understandings to which Buyer is a
party with respect to the voting of any shares of capital stock or other securities of Buyer.
49
(c) Since its respective date of formation, except as expressly contemplated by this
Agreement, Buyer has not declared or paid any distribution or dividend in respect of its shares of capital stock and has not repurchased, redeemed or otherwise acquired any of its shares of capital stock, and neither Buyer’s Board of Directors
has authorized any of the foregoing.
5.7 SEC Filings and Buyer Financials.
(a) Buyer, since its formation, has filed all SEC Documents with the SEC under the Securities Act and/or the Exchange Act, together with any
amendments, restatements or supplements thereto, and will file all such SEC Documents required to be filed subsequent to the Agreement Date. Except to the extent available on the SEC’s web site through EDGAR, Buyer has delivered to Company
copies in the form filed with the SEC of all of the following: (i) Buyer’s annual reports on Form 10-K for each fiscal year of Buyer beginning with the first year Buyer was required to file such a form; (ii) Buyer’s quarterly
reports on Form 10-Q for each fiscal quarter that Buyer filed such reports to disclose its quarterly financial results and (iii) all other forms, reports, registration statements, prospectuses and other documents (other than preliminary
materials) filed by Buyer with the SEC (the forms, reports, registration statements, prospectuses and other documents referred to in clauses (i), (ii) and (iii) above, whether or not available through EDGAR, are collectively, the “SEC
Reports”) and (iv) all certifications and statements required by (A) Rules 13a-14 or 15d-14 under the Exchange Act and (B) 18 U.S.C. §1350
(Section 906 of SOX) with respect to any report referred to in clause (i) above (collectively, the “Public Certifications”). The SEC Reports (x) were prepared in all material respects in accordance with the requirements of
the Securities Act and the Exchange Act, as the case may be, and the rules and regulations thereunder and (y) did not, as of their respective effective dates (in the case of SEC Reports that are registration statements filed pursuant to the
requirements of the Securities Act) and at the time they were filed with the SEC (in the case of all other SEC Reports) contain any untrue statement of a material fact or omit to state a material fact required to be stated therein or necessary in
order to make the statements made therein, in the light of the circumstances under which they were made, not misleading. The Public Certifications are each true as of their respective dates of filing. As used in this
Section 5.7, the term “file” shall be broadly construed to include any manner permitted by SEC rules and regulations in which a document or information is furnished, supplied or otherwise made available to the
SEC. (A) The Buyer Common Stock are listed on Nasdaq, (B) Buyer has not received any written deficiency notice from Nasdaq relating to the continued listing requirements of such Buyer Securities, (C) there are no Legal Proceedings
pending against Buyer by the Financial Industry Regulatory Authority or any other Person with respect to the continued listing of the Buyer Securities on Nasdaq, including any intention by such entity to suspend, prohibit or terminate the quoting of
such Buyer Securities on Nasdaq and (D) such Buyer Securities are in compliance with all of the applicable listing and corporate governance rules of Nasdaq.
(b) None of (a) any current report on Form 8-K, and any exhibits thereto or any other report, form, registration or other filing made
with any Governmental Authority with respect to the Transactions; or (b) the mailings or other distributions to Buyer Stockholders and/or prospective investors with respect to the consummation of the Transactions or in any amendment to any of
documents identified in (a) and (b), will, when filed, made available, mailed or distributed, as the case may be, contain any untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary in
order to make the statements therein, in light of the circumstances under which they are made, not misleading.
(c) The financial
statements and notes contained or incorporated by reference in the SEC Reports (the “Buyer Financial Statements”), fairly present in all material respects the financial position and the results of operations, changes in
shareholders’ equity, and cash flows of Buyer at the respective dates of and for the periods referred to in such financial statements, all in accordance with (i) GAAP methodologies applied on a consistent basis throughout the periods
involved and (ii) Regulation S- X or Regulation S-K promulgated by the SEC, as applicable (except as may be indicated in the notes thereto and for the omission of notes and audit adjustments in the case
of unaudited quarterly financial statements to the extent permitted by Regulation S-X or Regulation S-K, as applicable).
50
(d) Buyer has established and maintains disclosure controls and procedures and internal
control over financial reporting (as such terms are defined in paragraphs (e) and (f), respectively, of Rule 13a-15 and paragraph (e) of Rule 15d-15 under the
Exchange Act) as required by Rules 13a-15 and 15d-15 under the Exchange Act. Buyer’s disclosure controls and procedures are designed to ensure that all information
(both financial and non-financial) required to be disclosed by Buyer in the reports that it files or furnishes under the Exchange Act is recorded, processed, summarized and reported within the time periods
specified in the rules and forms of the SEC, and that all such information is accumulated and communicated to Buyer’s management as appropriate to allow timely decisions regarding required disclosure and to make the certifications required
pursuant to Sections 302 and 906 of SOX. Buyer’s management has completed an assessment of the effectiveness of Buyer’s disclosure controls and procedures and, to the extent required by Applicable Law, presented in any applicable SEC
Report, or any amendment thereto, its conclusions about the effectiveness of the disclosure controls and procedures as of the end of the period covered by such report or amendment based on such evaluation. Based on Buyer’s management’s
most recently completed evaluation of Buyer’s internal control over financial reporting, Buyer had no significant deficiencies or material weaknesses in the design or operation of its internal control over financial reporting that would
reasonably be expected to adversely affect Buyer’s ability to record, process, summarize and report financial information.
(e)
Except as and to the extent reflected or reserved against in the Buyer Financial Statements, Buyer has not incurred any liabilities or obligations of the type required to be reflected on a balance sheet in accordance with GAAP that is not adequately
reflected or reserved on or provided for in the Buyer Financial Statements, other than liabilities of the type required to be reflected on a balance sheet in accordance with GAAP that have been incurred since Buyer’s formation in the Ordinary
Course of Business.
5.8 Absence of Certain Changes. Since the date of the latest balance sheet included in the Buyer Financial
Statements, there has not occurred any event that has had or would reasonably be expected, individually or in the aggregate, to have a materially adverse impact on Buyer’s ability to consummate the Transactions.
5.9 Insurance. There is no claim pending under any insurance policies under which Buyer or any of its Subsidiaries is insured as to
which coverage has been questioned, denied or disputed by the underwriters of such policies. All premiums due and payable under all such insurance policies have been timely paid, and Buyer and its Subsidiaries, as applicable, are otherwise in
compliance with the terms of such policies. Buyer has not received any written threatened termination of, or material premium increase with respect to, any of such insurance policies. Neither Buyer nor any of its Subsidiaries has been refused any
insurance with respect to any aspect of their respective business, nor has any of their coverage been limited by any insurance carrier to which it has applied for insurance or with which it has carried insurance.
5.10 Litigation; Orders. There is no pending or threatened Legal Proceeding to which Buyer is subject which would reasonably be
expected, individually or in the aggregate, to have a material adverse effect on Buyer’s ability to consummate the Transactions.
5.11 Finders and Brokers. Except for MTS Health Partners, L.P., no broker, finder or investment banker is entitled to any brokerage,
finders or other fee or commission from Buyer, or the Company Group or any of their respective Affiliates in connection with the Transactions based upon arrangements made by or on behalf of Buyer.
51
5.12 Issuance of Buyer Shares. All Transaction Securities be issued and delivered to
the Shareholders in accordance with Article II shall be, upon issuance and delivery of such consideration free and clear of all Encumbrances, other than the restrictions under applicable securities laws, the restrictions set forth in
Section 5.13 and any Encumbrances incurred by any Shareholder, and fully paid and non-assessable, and the issuance and sale of Buyer Common Stock pursuant to this Agreement will not
be subject to or give rise to any preemptive rights or rights of first refusal.
ARTICLE VI
SURVIVAL; INDEMNIFICATION
6.1 Survival.
(a)
(i) The representations and warranties made by the Company Parties in Article III and Article IV of this Agreement and the Company Ancillary Agreements will survive in full force and effect for twelve (12) months following
the Closing Date, except that (A) the Company Fundamental Representations (other than the Tax Representations) shall survive the Closing and continue in full force and effect until 11:59 p.m. (Pacific Time) on the six (6) year anniversary
of the Closing Date and (B) the Tax Representations shall survive the Closing and continue in full force and effect until 11:59 p.m. (Pacific Time) on the date that is sixty (60) days after the expiration of the statute of limitations
applicable to the subject matter of such Tax Representations, (ii) each covenant of any Company Party that by its terms is to be performed or complied with at or prior to the Closing will survive until the date that is twelve (12) months
following the Closing Date, (iii) each other covenant of any Company Party contained herein will survive until fully performed in accordance with its terms and (iv) claims for Fraud will survive until the date that is six (6) years
following the Closing Date; provided, however, that if at any time prior to the expiration of the applicable survival period (each, an “Expiration Date”), an Indemnified Person (as defined below) delivers to the Holder
Representative a Claims Notice asserting a Liability Claim, then the claim asserted in such Claims Notice will survive such applicable Expiration Date until such time as the Liability Claim is fully and finally resolved.
(b) The representations and warranties made by Buyer in Article V and in the Buyer Ancillary Agreement and the covenants of Buyer
contained herein shall not survive the Closing, and from and after the Closing, Buyer and its Representatives shall not have any further obligations with respect thereto, nor shall any claim be asserted or action be brought against Buyer with
respect thereto.
(c) The Parties expressly agree pursuant to this Section 6.1 to modify the statutes of
limitations applicable to claims and causes of action as provided in this Agreement and therein. It is the express intent of the parties that, if an applicable survival period as contemplated by this Section 6.1 is shorter
or longer than the statute of limitations that would otherwise apply, then, by contract, the applicable statute of limitations shall be reduced or extended to the survival period contemplated hereby. The Parties further acknowledge that the time
periods set forth in this Section 6.1 for the assertion of claims under this Agreement are the result of arms’-length negotiation among the parties and that they intend for the time periods to be enforced as agreed by
the Parties.
6.2 Indemnification.
(a) From and after the Closing, subject to this Section 6.2, the Shareholders (the “Indemnitors”)
will severally indemnify, in accordance with their Indemnity Pro Rata Portions, and hold harmless Buyer and its Subsidiaries (including, after the Closing, the Company Group), and their respective affiliates, officers, directors, agents, attorneys
and employees (each of the foregoing, an “Indemnified Person”) from and against any and all Losses sustained by an Indemnified Person to the extent arising out of, in connection with or resulting from the following:
52
(i) any breach of, inaccuracy in or failure of any representation or warranty or
certification made by any Company Party in Article III or Article IV of this Agreement (other than the Company Fundamental Representations) or the Company Ancillary Agreements to be true and correct on the date of this Agreement and on
the Closing Date as if made on such date (except in the case of representations and warranties which by their terms speak only as of a specific date or dates, in which case such representations and warranties shall be true and correct on and as of
such specified date or dates);
(ii) any breach of, inaccuracy in or any failure of any Company Fundamental Representation to be true and
correct on the date of this Agreement and on the Closing Date as if made on such date (except in the case of representations and warranties which by their terms speak only as of a specific date or dates, in which case such representations and
warranties shall be true and correct on and as of such specified date or dates);
(iii) any breach before the Closing of any of the
covenants or agreements made by any Company Parties in this Agreement and any breach after the Closing of any of the covenants of the Holder Representatives in this Agreement;
(iv) any claims or threatened claims by or purportedly on behalf of any holder or former holder or purported holder of shares of Company
Stock or rights to acquire Company Stock, including claims that relate to the Transactions, appraisal rights or dissenters’ rights Legal Proceedings or any Subsequent Notice to Stockholders delivered before the Closing, claims by any holder or
former holder of shares of Company Stock that relate to matters arising from such holder’s or former holder’s interest in Company Stock or in such holder’s or former holder’s capacity as a holder of shares of Company Stock as of
or prior to the Closing, claims alleging violations of fiduciary duty and any amount payable in respect of claims of appraisal rights or dissenters’ rights Legal Proceedings with respect to shares of Company Stock, except any claims to enforce
such holder or purported holder’s rights under this Agreement or any Company Ancillary Agreement;
(v) any inaccuracy or omission in
the Estimated Closing Statement, including any inaccuracy or omission in the calculation of the Net Closing Shares issuable to any Shareholder in the Estimated Closing Statement, including any amounts set forth therein that are paid to a Person in
excess of the amounts such Person is entitled to receive pursuant to this Agreement or any amounts a Person was entitled to receive pursuant to this Agreement that were omitted from the Estimated Closing Statement;
(vi) any Fraud on the part of the Company;
(vii) any Fraud on the part of an Indemnitor; provided that (except with respect to Fraud addressed by clause (vi) above) the
Indemnified Persons shall only be entitled to recover Losses from such Indemnitor and no other Indemnitor and such Indemnitor shall be liable for the full amount of such Losses;
(viii) regardless of the disclosure of any matter set forth in the Company Disclosure Schedule, any Closing Debt, Closing Company Transaction
Expenses and Closing NWC Adjustment Amount, in each case, to the extent such amounts exceed the amount set forth in the Estimated Closing Statement; and
(ix) any Representative Losses and any claim by any Indemnitor relating to any alleged action or failure to act on its behalf by, or the
authority to act of, the Representative or asserting any right to receive Holdback Shares or Milestone Payments on an accelerated basis.
53
(b) Notwithstanding anything herein to the contrary, for purposes of
Section 6.2(a)(i), Section 6.2(a)(ii) and Section 6.2(a)(iii), any inaccuracy in or breach of any representation or warranty of the Company Parties as well as the amount
of any Losses that are the subject matter of a claim for indemnification for any inaccuracy in or breach of any representation or warranty of the Company shall be determined without regard and without giving effect to the term(s)
“material” or “Material Adverse Effect” or any other similar qualifiers contained in or otherwise applicable to such representation, warranty, covenant or agreement. The rights of the Indemnified Persons to indemnification,
compensation or reimbursement, payment of Losses or any other remedy under this Agreement shall not be affected by any investigation conducted with respect to, or any knowledge acquired (or capable of being acquired) at any time, whether before or
after the execution and delivery of this Agreement or the Closing Date, with respect to the accuracy or inaccuracy of or compliance with, any representation, warranty, covenant or agreement made by the Company or any other matter. Other than with
respect to a claim for Fraud, no Indemnified Person shall be required to show reliance on any representation, warranty, certificate or other agreement in order for such Indemnified Person to be entitled to indemnification, compensation or
reimbursement hereunder. Nothing in this Agreement shall limit the right of Buyer or any other Indemnified Person to pursue remedies under any Ancillary Agreement against the parties thereto.
(c) This Article VI shall be the sole and exclusive remedy of the Indemnified Persons from and after the Closing for any claims for
monetary damages arising under this Agreement; provided, however, that (i) this Section 6.2(c) shall not be deemed a waiver by any party of any right to specific performance or injunctive relief,
(ii) nothing in this Agreement shall limit the liability of an Indemnitor (and this Article VI shall not be the sole and exclusive remedy for such Indemnified Person) in connection with a claim based on Fraud committed by such Indemnitor
and (iii) nothing in this Agreement shall limit the right of Buyer or any other Indemnified Person to pursue remedies under any Ancillary Agreement against the parties thereto.
6.3 Limitations on Indemnification.
(a) Except with respect to Liability Claims pursuant to Section 6.2(a)(ii),
Section 6.2(a)(vi), Section 6.2(a)(vii) and Section 7.2(a)(viii), the Indemnified Persons’ sole source of recovery for claims under
Section 6.2(a)(i) shall be the Holdback Shares, determined by reducing the number of issuable Holdback Shares from the total number of Holdback Shares then outstanding (if any).
(b) Except for Fraud, no Indemnitor shall be liable to the Indemnified Persons until the aggregate amount of all Losses which would otherwise
be indemnifiable under Section 6.2(a)(i) equal or exceed $200,000 (the “Basket”); provided, however, that to the extent such Losses exceed the Basket, the Indemnitors shall indemnify the Indemnified Persons
for each dollar of Losses up to and including the Basket, in accordance with their Indemnity Pro Rata Portion.
(c) Except as otherwise
required by Applicable Law, the Parties will treat any payments made under this Article VI as an adjustment to the Total Consideration as specified in Section 2.2 for accounting and applicable Tax purposes.
(d) No Indemnitor will have any right of contribution, right of indemnity or other right or remedy against Buyer or the Company or its
affiliates in connection with any indemnification obligation to which such Indemnitor may become subject pursuant to or in connection with this Agreement.
(e) No Indemnified Person’s rights pursuant to this Section 6.3(e) will be adversely affected by any
investigation conducted, or any knowledge acquired or capable of being acquired, by an Indemnified Person at any time, whether before or after the execution or delivery of this Agreement or the Closing, or by the waiver of any condition to Closing.
54
(f) The right of Buyer or any other Indemnified Persons to pursue a Legal Proceeding for any
other remedies or relief under any Ancillary Agreement against the counterparties thereto will not be limited by the rights of the Indemnified Persons under this Section 6.4; provided, that in no event will the
aggregate liability of any Indemnitor under this Agreement or any Ancillary Agreement exceed the Total Consideration actually received by such Indemnitor, except in the case of Fraud committed by or with the actual knowledge of such Indemnitor.
(g) The amount of any Losses that are subject to indemnification under this Section 6.3 shall be calculated net of
the amount of any insurance proceeds, indemnification payments or reimbursements actually received by an Indemnified Person from Third Parties (other than the Indemnitors) in respect of such Losses (net of any costs or expenses incurred in obtaining
such insurance, indemnification or reimbursement, including any increases in insurance premiums or retro-premium adjustments resulting from such recovery); provided that nothing in this Agreement shall require an Indemnified Person to seek recovery
from insurance. If any Indemnified Person receives any such insurance proceeds, indemnification payments or reimbursements from Third Parties (other than the Indemnitors) with respect to any Losses which have already been settled under this
Section 6.3 through the forfeiture of Holdback Shares, it shall issue (or, if prior to the Holdback Release Date, allocate for potential issuance) such Holdback Shares. No Indemnified Person shall be entitled to recover any
Losses under this Agreement in respect of a single set of facts and circumstances to the extent that such Indemnified Person has already recovered Losses in respect of such set of facts and circumstances pursuant to another provision of this
Agreement.
6.4 Holdback Shares Release. The Holdback Shares will be issued as soon as practicable following the date that is
twelve (12) months after the Closing Date (the “Holdback Release Date”). Notwithstanding anything contained in this Agreement to the contrary, on the Holdback Release Date, such portion of the Holdback Shares as may be
necessary in the reasonable good faith judgment of Buyer to satisfy any unresolved or unsatisfied Liability Claims specified in any Claims Notice delivered to Representative before the Holdback Release Date will remain available for issuance until
such Liability Claims have been resolved or satisfied; provided, however, that within ten (10) Business Days after Buyer determines that any portion of the Holdback Shares is no longer necessary to satisfy any unresolved or
unsatisfied Liability Claims specified in any Claims Notice delivered to Holder Representative before the applicable Expiration Date or any such Liability Claim is resolved, Buyer will issue to the Shareholders the remaining Holdback Shares no
longer necessary to satisfy such Liability Claim to the Indemnitors pursuant to Section 6.4.
6.5 Claims for
Indemnification. At any time that an Indemnified Person desires to claim Losses (a “Liability Claim”) that it believes are or may be indemnifiable under Section 6.3, Buyer will deliver written
notice of such Liability Claim (a “Claims Notice”) prior to the expiration of the applicable survival period to the Holder Representative. A Claims Notice will describe the Liability Claim in reasonable detail, to the extent
known, and indicate the amount (estimated, if necessary and to the extent feasible) of the Losses that have been or may be sustained by the Indemnified Persons. If the amount of Losses is not determinable as of the date of delivery of a Claims
Notice, then Buyer may deliver a Claims Notice stating the maximum amount of Losses that Buyer in good faith estimates or anticipates that an Indemnified Person may sustain. No delay in or failure to give a Claims Notice by Buyer to the Holder
Representative pursuant to this Section 6.5 will adversely affect any of the other rights or remedies that Buyer has under this Agreement or alter or relieve the Indemnitors of their obligations to indemnify the Indemnified
Persons pursuant to this Section 6.5, except and to the extent that such delay or failure has materially prejudiced the Indemnitors. Following the delivery of a Claims Notice, the Holder Representative and its
representatives and agents will be given access (including electronic access, to the extent available), upon reasonable notice, to the books and records of the Buyer as it may reasonably require for the purposes of investigating or resolve any
disputes relating to, or responding to, any matters or inquiries raised in the Claims Notice; provided that the foregoing shall not require Buyer to provide access to any information or documents if such access in the good faith reasonable
belief of Buyer would violate any Law or otherwise result in the waiver of Buyer’s attorney-client privilege, the work product doctrine or similar privilege or protection applicable to such information or documents.
55
6.6 Objections to and Payment of Claims.
(a) The Holder Representative may object to any Liability Claim set forth in a Claims Notice by delivering written notice to Buyer of the
Holder Representative’s objection (an “Objection Notice”). Such Objection Notice must describe the grounds for such objection in reasonable detail. If an Objection Notice is not delivered by the Holder Representative to Buyer
within thirty (30) days after delivery by Buyer to the Holder Representative of the Claims Notice, such failure to so object will be an irrevocable acknowledgment by each Party (including the Representative) that the Indemnified Persons are
entitled to indemnification under Section 6.4 for the Losses set forth in such Claims Notice in accordance with this Article VI.
(b) If the Claims Notice was delivered to the Holder Representative and no Objection Notice was delivered to Buyer within thirty
(30) days of the delivery of the Claims Notice, or an Objection Notice states that it was, or admits liability, only with respect to a portion of the Losses claimed in the Claims Notice, Buyer will be entitled to permanently withhold from
Indemnitors, and Indemnitors will forfeit and not be entitled to receive, Holdback Shares having a value equal to (1) the amount of the Losses set forth in such Claims Notice, if no Objection Notice was delivered to Buyer, or (2) the
amount of the portion of the Losses set forth in such Claims Notice to which no objection was made in an Objection Notice that was delivered to Buyer by the Holder Representative (and Buyer shall be entitled to permanently retain or cancel any
Holdback Shares so withheld), except that, to the extent that the amount of the Losses set forth in the Claims Notice (or portion thereof) is an estimate, Buyer will not be so entitled to permanently withhold payment in respect of such portions
of such estimated Losses unless and until the amount of such estimated Losses is finally determined; provided, that any Indemnitor shall have the right to pay its Indemnity Pro Rata Portion of any Losses to be recovered from the Holdback
Shares in cash and Buyer shall release or cause to be released to such Indemnitor a number of shares of Holdback Shares (but not before the Holdback Release Date) equal to the amount of such Losses any such Indemnitor paid in cash divided by
the Buyer Common Stock Price. For purposes of determining the number of Holdback Shares required to satisfy any Losses under this Section 6.6(b), each Holdback Share shall be deemed to have a value equal to the Buyer Common
Stock Price.
(c) Notwithstanding anything to the contrary in this Agreement, the Indemnitors do not have any individual right to object
to any claim made in a Claims Notice under this Section 6.6, and any and all claims made in a Claims Notice on behalf of the Indemnified Persons may be objected to only by the Holder Representative.
6.7 Resolution of Objections to Claims.
(a) If the Holder Representative delivers an Objection Notice to Buyer with respect to any Liability Claim made in any Claims Notice, the
Holder Representative and Buyer will attempt to agree upon the rights of Buyer and the Indemnitors with respect to each such claim. If the Holder Representative and Buyer should so agree, they will enter into an agreement setting forth such rights.
To the extent a Liability Claim is recoverable directly from any Indemnitors pursuant to Section 6.3 other than through the forfeiture of Holdback Shares, each Indemnitor shall, within ten (10) Business Days of such
agreement, either (1) pay to Buyer in cash (on behalf of itself and any other Indemnified Person) its Indemnity Pro Rata Portion of any such shortfall, (2) direct Buyer to cancel a number of shares of Buyer Common Stock held by such
Indemnitor equal to such Indemnitor’s Indemnity Pro Rata Portion of any such shortfall divided by the Buyer Common Stock Price or (3) any combination of clauses (1) and (2).
56
(b) If no such agreement can be reached within thirty (30) days after delivery of an
Objection Notice, either Buyer or the Holder Representative (on behalf of the Indemnitors) may bring a Legal Proceeding against the other to resolve the dispute in accordance with this Article VI.
(c) To the extent a Liability Claim is recoverable directly from any Indemnitors pursuant to Section 6.3, each such
Indemnitor will promptly, and in no event later than ten (10) Business Days after the final resolution of any dispute in accordance with this Section 6.7(c), either (1) pay to Buyer in cash (on behalf of itself
and any other Indemnified Person) its Indemnity Pro Rata Portion of the amount of Losses determined in accordance with this Section 6.7(b), (2) direct Buyer to cancel a number of shares of Buyer Common Stock held by
such Indemnitor equal to such Indemnitor’s Indemnity Pro Rata Portion of the amount of Losses determined in accordance with this Section 6.7 divided by the Buyer Common Stock Price or (3) any combination of
clauses (1) and (2), except that if the final resolution provides for the payment of the Losses in another manner, then the Indemnitors will make payment of the Losses according to the final resolution. If the amount of the Losses so determined
is an estimate, then the Indemnitors will be required to make such payment within ten (10) days of the date that the amount of the Losses is finally determined.
6.8 Third-Party Claims. If Buyer receives written notice of a third-party claim that Buyer believes may result in a Liability Claim by
or on behalf of an Indemnified Person, Buyer will notify the Holder Representative in writing of such third-party claim and will provide a copy of any notice or other documentation submitted by the Third Party making such third-party claim and, if
the Indemnitors could have any liability hereunder with respect to such third-party claim, (i) provide the Holder Representative the opportunity to participate at the Indemnitors’ cost in, but not direct or conduct, any defense of such
claim, and (ii) keep the Holder Representative reasonably informed about the progress of such third-party claim and will consider in good faith recommendations made by the Holder Representative with respect to the defense of such third party
claim. The Holder Representative will not be provided such opportunity to the extent that Buyer reasonably determines in good faith that such participation could result in the loss of any attorney-client privilege or right under the work-product
doctrine of Buyer or any Indemnified Person in respect of such claim. The Holder Representative’s participation will be subject to Buyer’s right to control such defense. Buyer will have the right in its sole discretion to settle any such
third-party claim, but if the settlement is without the written consent of the Holder Representative, the settlement will not be determinative of the amount of Losses relating to such matter or whether such Losses are indemnifiable by the
Indemnitors hereunder. If the Holder Representative consents in writing to any such settlement, then neither the Holder Representative nor any Indemnitor will have any power or authority to object to the amount or validity of any claim by or on
behalf of any Indemnified Person for indemnity with respect to such settlement. The Holder Representative will be deemed to have been given consent to a settlement if the Holder Representative has not objected within twenty (20) days after the
Holder Representative’s receipt of a written request for consent to such settlement delivered by Buyer. Notwithstanding any other provision of this Agreement, any reasonable and documented out-of-pocket costs and expenses of investigation or defense, including court costs and reasonable attorneys’ fees, incurred or suffered by the Indemnified Persons in connection with any third-party
claim alleging matters that would constitute the failure of a representation or a warranty set forth herein to be true and correct or be the basis of a claim for any other matter specified in Section 6.2, whether or not it
is ultimately determined that there was such a failure to be true and correct or basis for a claim, will constitute Losses subject to indemnification under Section 6.2 (subject to such other limitations set forth in this
Section 6.8).
57
6.9 Holder Representative.
(a) By the execution and delivery of this Agreement, each of the Shareholders on behalf of itself and its successors and assigns, hereby
irrevocably constitutes and appoints Shareholder Representative Services LLC in its capacity as the Holder Representative, as the true and lawful agent, representative, and
attorney-in-fact of the Shareholders, following the Closing, with full powers of substitution to act in the name, place and stead of thereof with respect to the
performance on behalf of such Person following the Closing under the terms and provisions of this Agreement and the Ancillary Agreements to which the Holder Representative is a party, as the same may be from time to time amended, and to do or
refrain from doing all such further acts and things, and to execute all such documents on behalf of such Person, if any, as the Holder Representative will deem necessary or appropriate in connection with any of the transactions contemplated under
this Agreement or any of the Ancillary Agreements to which the Holder Representative is a party, including, solely in each case, following the Closing: (i) making on behalf of such Person any determinations and taking all actions on their
behalf relating to their receipt of the Holdback Shares, the Milestone Payments and any disputes with respect thereto; (ii) authorize payment to any Indemnified Person in satisfaction of any Liability Claims hereunder by any Indemnified Person;
(iii) object to such payments, (iv) agree to, negotiate, enter into settlements and compromises of, and demand arbitration and comply with orders of courts and awards of arbitrators with respect to any such Liability Claims,
(v) assert, negotiate, enter into settlements and compromises of, and demand arbitration and comply with orders of courts and awards of arbitrators with respect to, any such indemnification claim by any Indemnified Person hereunder against any
Indemnitor or by any such Indemnitor against any Indemnified Person or any dispute between any such Indemnified Person and any such Indemnitor, in each case relating to this Agreement or the Transactions, and (vi) to take all other actions that
are (A) necessary or appropriate in the judgment of the Holder Representative for the accomplishment of any of the foregoing, or (B) as specifically mandated by the terms of this Agreement or any other Ancillary Agreement.
(vii) signing on behalf of such Person any releases or other documents with respect to any dispute or remedy arising under this Agreement or any Ancillary Agreement; (viii) employing and obtaining the advice of legal counsel, accountants
and other professional advisors as the Holder Representative, in its reasonable discretion, deems necessary or advisable in the performance of its duties as the Holder Representative and to rely on their advice and counsel; (ix) incurring and
paying reasonable costs and expenses, including fees of brokers, attorneys and accountants incurred pursuant to the Transactions, and any other reasonable fees and expenses allocable or in any way relating to such Transactions, incurred subsequent
to Closing; and (x) otherwise enforcing the rights and obligations of any such Persons under this Agreement and the Ancillary Agreements to which the Holder Representative is a party. All decisions and actions by the Holder Representative,
shall be binding upon each of the Shareholders and their respective successors and assigns, and they shall not have the right to object, dissent, protest or otherwise contest the same. The provisions of this Section 6.9 are
irrevocable and coupled with an interest. The Holder Representative hereby accepts its appointment and authorization as the Holder Representative under this Agreement.
(b) Any other Person, including Buyer, the Company and the Shareholders may conclusively and absolutely rely, without inquiry, upon any
actions of the Holder Representative as the acts of the Shareholders hereunder or any Ancillary Agreements to which the Holder Representative is a party. Buyer and the Company shall be entitled to rely conclusively on the instructions and decisions
of the Holder Representative as to (i) any payment instructions provided by the Holder Representative or (ii) any other actions required or permitted to be taken by the Holder Representative hereunder, and neither the Company nor any
Shareholder shall have any cause of action against Buyer or Company for any action taken by any of them in reliance upon the instructions or decisions of the Holder Representative. Buyer and Company shall not have any liability to Company or any
Shareholder for any allocation or distribution among the Shareholders by the Holder Representative of payments made to or at the direction of the Holder Representative. All notices or other communications required to be made or delivered to a
Shareholder under this Agreement or any Ancillary Agreement to which the Holder Representative is a party after the Closing shall be made to the Holder Representative for the benefit of such Shareholder, and any notices so made shall discharge in
full all notice requirements of the other parties hereto or thereto to such Shareholder with respect thereto. All notices or other communications required to be made or delivered by a Shareholder following the Closing shall be made by the Holder
Representative (except for a notice under Section 6.9(d) of the replacement of the Holder Representative).
58
(c) The Holder Representative will incur no liability in connection with its services
pursuant to this Agreement and any related agreements except to the extent resulting from its gross negligence or willful misconduct. The Holder Representative shall not be liable for any action or omission pursuant to the reasonable, good-faith
advice of counsel. The Shareholders shall indemnify the Holder Representative against any reasonable, documented, and out-of-pocket losses, liabilities and expenses
(“Representative Losses”) arising out of or in connection with this Agreement and any related agreements, in each case as such Representative Loss is suffered or incurred; provided, that in the event that any such Representative Loss is
finally adjudicated to have been caused by the gross negligence or willful misconduct of the Holder Representative the Holder Representative will reimburse the Shareholders the amount of such indemnified Representative Loss to the extent
attributable to such gross negligence or willful misconduct. Representative Losses may be recovered by the Holder Representative from (i) the funds in the Expense Fund and (ii) any other funds that become payable to the Shareholders under
this Agreement at such time as such amounts would otherwise be distributable to the Shareholders; provided, that while the Holder Representative may be paid from the aforementioned sources of funds, this does not relieve the Shareholders from their
obligation to promptly pay such Representative Losses as they are suffered or incurred. In no event will the Holder Representative be required to advance its own funds on behalf of the Shareholders or otherwise. Notwithstanding anything in this
Agreement to the contrary, any restrictions or limitations on liability or indemnification obligations of, or provisions limiting the recourse against non-parties otherwise applicable to, the Shareholders set
forth elsewhere in this Agreement are not intended to be applicable to the indemnities provided to the Holder Representative hereunder. The foregoing indemnities will survive the Closing, the resignation or removal of the Holder Representative or
the termination of this Agreement.
(d) Upon the Closing, the Company will wire US$50,000 (the “Expense Fund”) to the
Holder Representative, which will be used for any expenses incurred by the Holder Representative. The Shareholders will not receive any interest or earnings on the Expense Fund and irrevocably transfer and assign to the Holder Representative any
ownership right that they may otherwise have had in any such interest or earnings. The Holder Representative will hold these funds separate from its corporate funds and will not voluntarily make these funds available to its creditors in the event of
bankruptcy. As soon as practicable following the completion of the Holder Representative’s responsibilities, Holder Representative will deliver any remaining balance of the Expense Fund to the Paying Agent for further distribution to the
Shareholders. For tax purposes, the Expense Fund will be treated as having been received and voluntarily set aside by the Shareholders at the time of Closing.
(e) If the Holder Representative shall die, become disabled, dissolve, resign or otherwise be unable or unwilling to fulfill its
responsibilities as representative and agent of Shareholders, then the Shareholders shall, within ten (10) days after such death, disability, dissolution, resignation or other event, appoint a successor Holder Representative (by vote or written
consent of the Shareholders holding in the aggregate in excess of fifty percent (50%) of the Company Stock as of the Closing (or the date of such vote or consent, if earlier)), and promptly thereafter (but in any event within two (2) Business
Days after such appointment) notify Buyer in writing of the identity of such successor. Any such successor so appointed shall become the “Holder Representative” for purposes of this Agreement.
ARTICLE VII
COMPANY
PARTY COVENANTS
7.1 Advise of Changes.
During the time period from the Agreement Date until the earlier to occur of (a) the Closing or (b) the termination of this
Agreement in accordance with the provisions of Article X (the “Interim Period”), Company or any Shareholder, as applicable, shall promptly advise Buyer in writing of (i) any event occurring subsequent to the Agreement
Date that would render the representations or warranties of Company contained in Article III or of such Shareholder contained in
59
Article IV untrue or inaccurate such that the condition set forth in Section 9.2(a) would not be satisfied or (ii) any breach of the covenants or obligations
of Company pursuant to this Agreement or any Company Ancillary Agreement such that the condition set forth in Section 9.2(b) could reasonably be expected not to be satisfied (except to the extent such covenant or obligation
shall by its terms be complied with or satisfied at the Closing). In no event shall the giving of any such notice be deemed to cure any breach of this Agreement or to modify the Company Disclosure Schedule, in each case, except in the event that the
Closing occurs.
7.2 Business Activities. During the Interim Period, the Company shall, and Shareholders shall procure the Company
Group to, (a) conduct the Company Business in the Ordinary Course of Business of the Company Group and (b) use reasonable best efforts to maintain and preserve intact its business organization and the goodwill of those having business
relationships with it (including by using reasonable best efforts to preserve its assets and technology and relationships with its customers and suppliers) and to retain the services of its present executive officers.
7.3 Conduct of the Company Group Business. During the Interim Period, except as (i) expressly contemplated by this Agreement,
(ii) required by Applicable Law, (iii) set forth on Section 7.3 the Company Disclosure Schedule or (iv) expressly contemplated by a Company Material Contract, the Company shall, and Shareholders shall procure
the Company Group to, conduct the Company Business in the Ordinary Course of Business of the Company Group, and shall procure that the Company Group does not, without Buyer’s prior written consent:
(a) (i) make any investments in or capital contributions to, any Person or (ii) forgive or discharge in whole or in part any outstanding
material loans or advances;
(b) place or allow the creation of any Encumbrance (other than a Company Permitted Encumbrance) on any
material portion of its assets or properties;
(c) sell, lease, license, Transfer or dispose of any portion of its assets (except for
sales, leases or licenses of assets in the Ordinary Course of Business of the Company Group);
(d) except as required by Applicable Law,
by any Contract in effect as of the Agreement Date and reflected on Section 7.3(d) of the Company Disclosure Schedule, (i) grant or pay any special bonus or special remuneration or (ii) materially increase the
salary or benefits of any officer, director, employee, independent contractor, or consultant of the Company Group;
(e) except as required
by any Contract in effect as of the Agreement Date and reflected on Section 7.3(e) of the Company Disclosure Schedule, promise, make, or grant any retention, severance, change of control, termination, or other similar
payment to any officer, director, employee, independent contractor, or consultant of the Company Group or any other Person;
(f) except as
required by Applicable Law, enter in to or amend any collective bargaining agreement or any other Contract with trade or labor union, works council, employees’ association or similar organization, or any similar Contract with Governmental
Authority or employees of the Company Group;
(g) (i) hire any employee or (ii) engage any consultant or independent contractor
expected to earn annual compensation in excess of €50,000 or above; or terminate the employment or engagement of any employee, consultant or independent contractor other than terminations for cause;
60
(h) materially change any of its accounting methods other than as may be required by Italian
GAAP, GAAP, other applicable accounting standards or Applicable Laws;
(i) declare, set aside or pay any cash or stock dividend or other
distribution (whether in cash, stock or property) in respect of its capital stock, or redeem, repurchase or otherwise acquire any of its capital stock or other securities (except for the repurchase of stock from its employees, directors, consultants
or contractors in connection with the termination of their services), or pay or distribute any cash or property to any of its stockholders or securityholders or make any other cash payment to any of its stockholders or securityholders, other than
payments in respect of Debt of the Company Group;
(j) issue, sell, create or authorize any shares of its capital stock of any class or
series or any other of its securities, or issue, grant or create any warrants, obligations, subscriptions, options, convertible securities, or other commitments to issue shares of its capital stock or any securities that are potentially exchangeable
for, or convertible into, shares of its capital stock;
(k) make, change, or revoke any Tax election, adopt or change any Tax accounting
method or period, file any income or other material Tax Return (except as provided in Section 11.1(a)), file any amended Tax Return, enter into any “closing agreement” within the meaning of Section 7121 of the Code (or any
corresponding or similar provision of state, local, or non-U.S. Applicable Law), participate in any voluntary disclosure application or agreement or similar process, apply for any Tax ruling, settle or compromise any Tax claim or assessment, consent
to any extension or waiver of the limitation period applicable to any Tax claim or assessment, enter into any Tax allocation, sharing, or indemnity agreement, surrender any right to claim a Tax refund, or take any other similar action relating to
the filing of any Tax Return or the payment of any Tax if such similar action would have the effect of increasing the Tax liability of Buyer or any of its Affiliates (including the Company Group) for any taxable period (or portion thereof) ending
after the Closing Date or decreasing any Tax attribute or asset of any Company Group Member existing on the Closing Date;
(l) subdivide,
split, combine or reverse split the outstanding shares of its capital stock of any class or series or enter into any recapitalization affecting the number of outstanding shares of its capital stock of any class or series or affecting any other of
its securities;
(m) merge, consolidate or reorganize with, acquire, or enter into any other business combination with any corporation,
partnership, limited liability company or any other entity (other than Buyer), acquire a substantial portion of the assets of any such entity, or enter into any binding negotiations, discussions or agreement for such purpose;
(n) amend the Company Charter Documents or any Subsidiary Charter Documents;
(o) incur into any financial indebtedness in any form whatsoever;
(p) license any of its technology or Intellectual Property that is material to the Company Business (except for licenses made in the Ordinary
Course of Business, provided that under no circumstances shall any Company Group Member enter into any software escrow or similar agreement or arrangement), or acquire any material Intellectual Property (or any license thereto) from any Third
Party;
(q) reduce any insurance coverage (other than as contemplated in this Agreement);
(r) (i) initiate any litigation, action, suit, proceeding, claim or arbitration, except for the request by any Company Group Member of urgency
orders and/or injunctions which are deemed in good faith by the relevant Company Group Member to be necessary to avoid a material prejudice to the interests or rights of such Company Group Member or (ii) settle or agree to settle any material
litigation, action, suit, proceeding, claim or arbitration;
61
(s) other than with respect to contractual obligations binding the Company prior to the date
of this Agreement and disclosed under Section 7.3(s) of the Company Disclosure Schedule, incur any expense (or series of related expenses) in excess of €10,000; or
(t) agree to do any of the things described in the preceding clauses (a)-(s).
7.4 Necessary Consents. During the Interim Period, Company shall use its commercially reasonable efforts to obtain prior to Closing
such written consents and authorizations of Third Parties, give notices to Third Parties and take such other actions as may be necessary or appropriate in order to effect the Transactions, to enable the Company Group to carry on the Company Business
immediately after the Closing and to keep in effect and avoid the breach, violation of, termination of, or adverse change to, any Company Material Contract which are listed on Section 7.4 of the Company Disclosure Schedule;
provided, however, that (i) Company shall not be required to pay any amount to obtain any consent or authorization from a Third Party, (ii) Company shall not be required to agree to any term, condition or modification to any Contract with
respect to obtaining any such consents or authorizations, (iii) no such consent, authorization, notice or action shall be a condition to the Closing and (iv) Company shall have no liability solely for the failure to obtain any such consent
or authorization, give such notice or take such actions.
7.5 No Other Negotiations.
During the Interim Period, the Company and the Shareholders shall not, and shall not authorize, encourage or permit any Representatives of any
member of the Company Group’s or any of the Shareholders or any other Person to, directly or indirectly: (a) solicit, initiate, or knowingly encourage, facilitate or induce the making, submission or announcement of any inquiry, offer or
proposal from any Person (other than Buyer) concerning any Alternative Transaction; (b) furnish any nonpublic information regarding the Company Group to any Person (other than Buyer and its Representatives) in connection with or in response to
any inquiry, offer or proposal for or regarding any Alternative Transaction (other than to respond to such inquiry, offer or proposal by indicating that Company is subject to this Section 7.5); (c) enter into,
participate in, maintain or continue any discussions or negotiations with any Person (other than Buyer and its Representatives) with respect to any Alternative Transaction (other than to respond to such inquiry, offer or proposal by indicating that
the Company is subject to this Section 7.5); (d) execute, enter into or become bound by any letter of intent, memorandum of understanding, other Contract or understanding between Company and any Person (other than
Buyer) that is related to, provides for or concerns any Alternative Transaction; (e) approve, endorse or recommend, in each case, in writing, or publicly propose to approve, endorse or recommend, any Alternative Transaction; or (f) release
any Third Party from, or waive any provision of, any confidentiality agreement.
(b) The Company Group or the Shareholders, as applicable,
shall notify Buyer within 24 hours after receipt by any member of the Company Group or the Shareholders, as applicable, of any bona fide inquiry, offer or proposal that constitutes an Alternative Transaction, or any other bona fide notice that
any Person is considering making an Alternative Transaction, or any request for nonpublic information relating to the Company Group or for access to any of the properties, books or records of the Company Group by any Person or Persons other than
Buyer (which notice shall identify the Person or Persons making, or considering making, such inquiry, offer or proposal) in connection with a potential Alternative Transaction and shall keep Buyer fully informed of the status and details of any such
inquiry, offer or proposal and any correspondence or communications related thereto and shall provide to Buyer a correct and complete copy of such inquiry, offer or proposal, if it is in writing, or a written summary of the material terms thereof,
if it is not in writing. The Company Group and the Shareholders shall immediately cease and cause to be terminated any and all existing discussions and negotiations with any Persons (other than Buyer and its Representatives) conducted heretofore
with respect to an Alternative Transaction.
62
7.6 Access to Information. During the Interim Period, the Company shall allow Buyer
and its Representatives reasonable access during normal business hours, upon reasonable advance notice and without creating any disruption to the activities, business or operations of the Company Group, to the files, books, records, technology,
Contracts, senior personnel and offices of the Company Group, including any and all information relating to the Company Group’s Taxes, Contracts, liabilities, financial condition and real, personal and intangible property, subject to the terms
of the Confidentiality Agreement between Company and Buyer dated February 9, 2023 (the “Confidentiality Agreement”). Company shall instruct its accountants to cooperate with Buyer and Buyer’s Representatives in making
available all financial information reasonably requested by Buyer and its agents and advisors, including the right to examine all working papers pertaining to all financial statements prepared by such accountants (subject to the execution of a
customary access letter or other conditions required by any such accountant). If this Agreement is terminated, the Confidentiality Agreement shall remain in full force and effect, and all copies of documents containing confidential information of a
disclosing party shall be returned by the receiving party to the disclosing party or be destroyed, as provided in the Confidentiality Agreement. Notwithstanding anything to the contrary in this Section 7.6, Company and its
Representatives shall not be required to provide any access or cooperation (i) with respect to any information that is subject to attorney-client privilege to the extent doing so would reasonably be expected to cause such privilege to be waived
or (ii) to the extent such access or cooperation is prohibited by any Applicable Law. No information obtained by Buyer during the pendency of the Transactions in any investigation pursuant to this Section 7.6 shall
affect or be deemed to modify any representation, warranty, covenant, agreement, obligation or condition set forth herein.
7.7
Satisfaction of Conditions Precedent. The Company and each Shareholder shall use their respective reasonable best efforts to satisfy or cause to be satisfied all the conditions precedent set forth in Sections 9.1 and
9.2.
7.8 Cooperation with SEC Filings. Prior to the Closing and in connection with Buyer’s preparation of any filing
required to be made by Buyer with the SEC under the Exchange Act or any responses to any comments from the SEC relating to such required filings, Company shall use its commercially reasonable efforts to provide to Buyer, and shall cause each Company
Group Member to use its commercially reasonable efforts to provide to Buyer, and shall use its commercially reasonable efforts to cause their respective Representatives, including legal and accounting representatives, to provide all cooperation
reasonably requested by Buyer that is customary in connection with the preparation of such filings or responses to SEC comments, which may include, among other things, obtaining the consents of any auditor to the inclusion of the financial
statements of Company or the US Subsidiary in filings with the SEC.
7.9 Regulatory Approvals. The Company and Shareholders shall
join in the execution and filing of the Golden Power Filing, in conformity with the provision set forth in Section 8.1. The Company and the Shareholders shall use commercially reasonable efforts to obtain the Golden Power Clearance including by
promptly cooperating with Buyer to prepare and file all the information and documents reasonably requested and/or necessary for the purpose of: (i) making the filings with the Golden Power Authority, (ii) providing any additional
information and documentation that may be requested by the Golden Power Authority and (iii) any discussions and negotiations with such Governmental Authorities and, more in general, any relationship and dealing with such Governmental
Authorities in connection with the obtainment of the Golden Power Clearance.
63
7.10 Release Agreements. For any Person to whom a Transaction Compensation Payment is
payable or will become payable after the Closing, the Company shall obtain a release of claims from such Person in a form satisfactory to Buyer, including the Benatti Settlement Agreement (collectively, the “Release Agreements”),
and no Transaction Compensation Payment may be paid by the Company prior to Closing to a Person who has not duly executed a Release Agreement that is effective.
7.11 Company Options. Prior to Closing, the Company and the Shareholders shall procure the revocation of the Company Stock Plan,
including the related capital increases resolved upon by the extraordinary shareholders’ meeting on April 9, 2018, and December 19, 2018. Each such Optionholder shall have executed an option cancellation and release agreement
concurrently with the execution and delivery of this Agreement in a form acceptable to Buyer.
7.12
By-Laws Amendment. As promptly as practicable after the date hereof and no later than 10 Business Days before the Closing Date, the Company and the Shareholders shall cause a shareholders’ meeting
to validly approve an amendment of the Company’s by-laws in force as at the Agreement Date in order to: (i) delete any restriction to the transfer of Company shares; and, if necessary,
(ii) amend the liquidation preferences under article 26 of the by-laws to properly reflect the allocation of the Total Consideration set forth under Section 2.2. Any such amendment to the
Company’s by-laws shall be subject to Buyer’s prior written consent.
ARTICLE VIII
BUYER COVENANTS
8.1 Regulatory Approvals. If the Golden Power Clearance is not obtained at the end of the proceedings launched with the submission of
the Golden Power Pre-Filing and, in any event, within thirty (30) days after the submission of the Golden Power Pre-Filing, Buyer and the Company shall, by and no
later than five (5) Business Days following, alternatively, (i) the expiry of a thirty (30) day period since the submission of the Golden Power Pre-Filing, or (ii) the date of the receipt
of the notice of completion of the proceedings launched with the submission of the Golden Power Pre-Filing issued by the Golden Power Authority (whichever occurs sooner), promptly execute and file, or join in
the execution and filing of the Golden Power Filing as well as any other application, notification or other document that may be necessary in order to obtain the authorization, approval or consent of any Governmental Authority, whether foreign,
federal, state, provincial, local or municipal (including to the extent required under any laws that are designed to prohibit, restrict or regulate actions having the purpose or effect of monopolization or restraint of trade), which may be required
in connection with the Closing, at Buyer’s sole cost and expense. Buyer shall use reasonable best efforts to obtain all such authorizations, approvals and consents and shall pay any associated filing fees payable by Buyer with respect to such
authorizations, approvals and consents as promptly as reasonably practicable and cooperate in the filing of any memoranda, white papers, filings, correspondence or other written communications explaining or defending the Transactions, articulating
any regulatory or competitive argument, and/or responding to requests or objections made by any Governmental Authority. Buyer shall promptly inform the Company of any communication between Buyer and any Governmental Authority regarding any of the
Transactions and permit Company and its Representatives (including outside counsel) to review any communication given by it to, and consult with in advance of any meeting or conference with, any Governmental Authority, and to the extent permitted by
such Governmental Authority, give Representatives of Company the opportunity to attend and participate in such meetings and conferences (or, in the event any of Company’s Representatives are prohibited from participating in or attending any
such meetings or conferences, Buyer shall keep Company and its Representatives promptly and reasonably apprised with respect thereto). If Buyer or any Affiliate of Buyer receives any formal or informal request for supplemental information or
documentary material from any Governmental Authority with respect to the Transactions, then Buyer shall make, or cause to be made, as soon as reasonably practicable, a response in compliance with such request following consultation with the Company.
If any Governmental Authority requires that a hearing or meeting be held in connection with its approval of the Transactions, whether prior to the Closing or after the Closing, each Party shall arrange for
64
Representatives of such Party to be present for such hearing or meeting. If any objections are asserted with respect to the Transactions under any Applicable Law or if any Legal Proceeding is
instituted (or threatened to be instituted) by any applicable Governmental Authority or any private Person challenging any of the Transactions contemplated by this Agreement or any Ancillary Agreements as violative of any Applicable Law or which
would otherwise prevent, materially impede or materially delay the consummation of the Transactions, the Parties shall use their commercially reasonable efforts to resolve any such objections or Legal Proceedings so as to timely permit consummation
of the Transactions, including in order to resolve such objections or Legal Proceedings which, in any case if not resolved, could reasonably be expected to prevent, materially impede or materially delay the consummation of the Transactions. In the
event any Legal Proceeding is instituted (or threatened to be instituted) by a Governmental Authority or private Person challenging the Transactions, the Parties shall, and shall cause their respective Representatives to, cooperate in all respects
with each other and use their respective reasonable best efforts to contest and resist any such Legal Proceeding and to have vacated, lifted, reversed or overturned any judgment, decree, injunction, rule or order, whether temporary, preliminary or
permanent, that is in effect and that prohibits, prevents or restricts consummation of the Transactions. The Shareholders shall, and shall cause the Company to, cooperate with and provide the Buyer with the information and documents relating to the
Company or the Shareholders as may be reasonably and in good faith requested by the Buyer for the preparation of the notifications, filings or submissions with the Governmental Authorities or to satisfy any additional request by the Governmental
Authorities. The Shareholders further shall, and shall cause the Company to, provide the Governmental Authorities as soon as reasonably practicable with any additional information, data, materials and documentation that they may request directly,
while informing and cooperating with the Buyer before their submission.
8.2 Satisfaction of Conditions Precedent. Buyer shall use
its commercially reasonable efforts to satisfy or cause to be satisfied all of the conditions precedent that are set forth in Sections 9.1 and 9.3, and Buyer shall use its commercially reasonable efforts to cause the
Transactions to be consummated in accordance with the terms of this Agreement.
8.3 Documents and Information. After the Closing
Date, Buyer shall, and shall cause its Subsidiaries to, until the seventh anniversary of the Closing Date, retain all books, records and other documents pertaining to the business of the Company Group in existence on the Closing Date and make the
same available for inspection and copying by the Holder Representative during normal business hours of Buyer and its Subsidiaries, as applicable, upon reasonable request and upon reasonable notice. No such books, records or documents shall be
destroyed after the seventh anniversary of the Closing Date by Buyer or its Subsidiaries (including any Company Group Member) without first advising the Holder Representative in writing and giving the Holder Representative a reasonable opportunity
to obtain possession thereof.
8.4 Post-Closing Board of Directors. Luca Benatti shall be appointed to the board of directors of
the Buyer as of immediately after Closing.
8.5 Employee Matters. From the Closing Date until the first anniversary of the Closing
(the “Benefits Continuation Period”), Buyer shall provide, or shall cause any of its Subsidiaries to provide, for each employee of the Company Group who continues as an employee of Buyer or any of its Subsidiaries during the
Benefits Continuation Period (collectively, the “Continuing Employees”), (i) base salary or base compensation, as applicable, that is no less than the base salary or base compensation, as applicable, provided to that Continuing
Employee by the Company Group immediately prior to the Closing, (ii) retirement, health and welfare benefits that are no less favorable, in the aggregate, than (A) the retirement, health and welfare benefits that are provided to each such
Continuing Employee by the Company Group immediately prior to the Closing, (iii) severance payments and benefits to each Continuing Employee whose employment is terminated by Buyer or any of its Subsidiaries other than for cause during the
Benefits
65
Continuation Period that are no less favorable, in the aggregate, than the severance payments and benefits that are provided to similarly situated employees of Buyer at the time of such
termination and (iv) to the extent a Continuing Employee had accrued but unused holidays immediately prior to the Closing, such accrued but unused holidays shall be available to such Continuing Employee following the Closing or, to the extent
allowed under Applicable Law, Buyer shall make a payment in cash to such Continuing Employee (subject to tax withholding) in respect of such accrued but unused holidays. The provisions of this Section 8.5 are solely for the
benefit of the Company and Buyer, and (x) no provision of this Section 8.5 or otherwise in this Agreement is intended to, or shall, confer upon any employee any right to continued employment for any period or continued
receipt of any specific employee benefit, or alter or limit Buyer’s or the Company’s or their Affiliates’ ability to amend, modify or terminate any particular benefit plan, program, agreement or arrangement and (y) no current or
former employee or any other individual associated therewith shall be regarded for any purpose as a third-party beneficiary of this Section 8.5 or have the right to enforce the provisions hereof, including any signatory to
this Agreement.
8.6 Conduct of Buyer Business. During the Interim Period, except as (i) expressly contemplated by this
Agreement, (ii) required by Applicable Law, (iii) set forth on Section 8.6 of the Buyer Disclosure Schedule or (iv) expressly contemplated by any Contract or obligation reflected in Buyer’s SEC Documents
as of the Agreement Date, Buyer shall, and shall cause its Subsidiaries to, conduct its in the Ordinary Course of Business, and shall not, without the Company’s prior written consent:
(a) offer, issue, deliver, grant or sell, or authorize or propose to offer, issue, deliver, grant or sell, any capital stock of, or other
equity interest in, Buyer or any of its Subsidiaries or any securities convertible into, or any rights, warrants or options to acquire, any such capital stock or equity interest (an “Equity Issuance”), other than (1) the
issuance of Buyer Common Stock upon the vesting, settlement, exercise or lapse of any restrictions on any Buyer equity awards granted under Buyer equity plans outstanding on the date hereof; and (2) issuances of Buyer equity awards under any
Buyer equity plans to employees, directors and other service providers in the Ordinary Course of Business;
(b) directly or indirectly
acquire or agree to acquire in any transaction any majority equity interest in, or business of, any firm, corporation, partnership, company, limited liability company, or any other entity (other than the Company and the US Subsidiary), acquire a
substantial portion of any properties or assets of any such entity, or enter into any binding negotiations, discussions or agreement for such purpose;
(c) agree to do any of the things described in the foregoing clauses (a) through (b).
8.7 Access to Information. During the Interim Period, Buyer shall allow the Company and its Representatives reasonable access during
normal business hours, upon reasonable advance notice and without creating any disruption to the activities, business or operations of the Buyer, to the files, books, records, and Contracts of Buyer, subject to the terms of the Confidentiality
Agreement. If this Agreement is terminated, the Confidentiality Agreement shall remain in full force and effect, and all copies of documents containing confidential information of a disclosing party shall be returned by the receiving party to the
disclosing party or be destroyed, as provided in the Confidentiality Agreement. Notwithstanding anything to the contrary in this Section 8.7, Buyer and its Representatives shall not be required to provide any access or
cooperation (i) with respect to any information that is subject to attorney-client privilege to the extent doing so would reasonably be expected to cause such privilege to be waived or (ii) to the extent such access or cooperation is
prohibited by any Applicable Law or any Contract to which Buyer is party. No information obtained by the Company during the pendency of the Transactions in any investigation pursuant to this Section 8.7 shall affect or be
deemed to modify any representation, warranty, covenant, agreement, obligation or condition set forth herein.
66
ARTICLE IX
CONDITIONS TO CLOSING
9.1
Conditions to Each Party’s Obligation to Effect the Closing. The respective obligations of each Party to effect the Closing shall be subject to the satisfaction or written waiver (where permissible) by Company and Buyer
prior to the Closing Date of the following conditions:
(a) Governmental Approvals. All authorizations, consents, orders or
approvals of, or declarations or filings with, or expirations of waiting periods imposed by, any Governmental Authority required to consummate the Transactions that are set forth in Section 9.1(a) of the Company Disclosure
Schedule shall have been filed, expired, been terminated, occurred or been obtained, as applicable, pursuant to Applicable Law.
(b)
Golden Power Clearance. The Golden Power Clearance has been obtained pursuant to the Golden Power Law.
(c) No Injunctions or
Restraints; Illegality. No temporary restraining order, preliminary or permanent injunction or other order issued by any Governmental Authority prohibiting or preventing the Transactions shall have been issued, nor shall any proceeding brought
by a Governmental Authority seeking any of the foregoing be pending; nor shall there have been any Applicable Law enacted, entered, enforced or deemed applicable to the Transactions which makes the Transactions illegal.
(d) Appointment to the Post-Closing Buyer Board. The Post-Closing Buyer Directors shall have
been elected or appointed to the board of directors of Buyer as of the Closing.
9.2 Additional Conditions to Obligations of Buyer.
The obligations of Buyer to effect the Closing are subject to the satisfaction of each of the following conditions, any of which may be waived in writing exclusively by Buyer:
(a) Representations and Warranties.
(i) The Company Fundamental Representations (other than the Tax Representations) shall be true and correct in all respects, as of the
Agreement Date and as of the Closing Date as though made on the Closing Date (except with respect to any Company Fundamental Representations (other than any Tax Representation) that address matters only as of a particular date, the accuracy of which
shall be determined as of such date).
(ii) The representations and warranties of the Company contained in this Agreement (other than the
Company Fundamental Representations except for the Tax Representations) will be true and correct in all respects, in each case both as of the Agreement Date and as of the Closing Date, (1) other than such representations and warranties that
expressly speak only as of a specific date or time, the accuracy of which shall be determined as of such date or time, and (2) disregarding any qualifications as to “materiality” or “Material Adverse Effect” contained in
such representations and warranties, except where the failure of such representations and warranties to be true and correct at such time has not or would not, individually or in the aggregate, had or reasonably be expected to have a Material Adverse
Effect.
(iii) The representations and warranties of the Shareholders contained in this Agreement (other than such representations and
warranties which are Company Fundamental Representations) shall be true and correct in all material respects, as of the Agreement Date and as of the Closing Date as though made on the Closing Date (except with respect to any such representations
that address matters only as of a particular date, the accuracy of which shall be determined as of such date).
67
(b) Performance of Obligations of Company Parties. The Company and each Shareholder
shall have performed and complied with each of the covenants and obligations under this Agreement required to be performed and complied with by the Company or any Shareholder on or prior to the Closing.
(c) Material Adverse Effect. Since the Agreement Date, there shall not have occurred a Material Adverse Effect on the Company or the US
Subsidiary.
(d) Deliveries. The Company Parties shall have delivered or caused to be delivered to Buyer or Buyer’s counsel,
at Closing, the following:
(i) Officer’s Certificate. The Company shall have delivered to Buyer a certificate
from the Company, signed by an executive officer of the Company in such capacity, certifying as to the satisfaction of the conditions specified in Sections 9.2(a)-(c).
(ii) Closing Statement. The Estimated Closing Statement shall have been delivered in accordance with
Section 2.4.
(iii) Financials. Company shall have delivered audited financial statements of the Company
Group for the years ending December 31, 2021 and December 31, 2022.
(iv) Company Group Good Standing Certificates.
Company shall have delivered to Buyer a certificate from the applicable Governmental Authority in each jurisdiction where each Company Group Member is organized (except, in the case of good standing or its jurisdictional equivalent, for entities
organized under the laws of any jurisdiction that does not recognize such concept).
(v) Resolutions. The Company shall have
delivered to Buyer resolutions (or other documentation) reflecting all corporate proceedings of the Company necessary to approve and authorize the execution and delivery of the documentation to be executed at the Closing, accompanied by a
certificate signed on behalf of Company by an officer of Company to the effect that such resolutions (or other documentation) remain in full force and effect and have not been amended or modified in any manner.
(vi) Share Deed of Transfer. Each Shareholder and the Buyer shall have executed – before Italian Notary Public – the Share
Deed of Transfer, having as object the transfer of the Closing Company Shares in favor of the Buyer representing such Shareholder’s Company Stock.
(vii) Appointments. The Company shall have delivered the letters of resignation from the current directors of the Company, and shall
have made its best effort to deliver the letters of resignation from all the current members of the board of statutory auditors of the Company, with effect from the Closing Date, in a form reasonably acceptable to Buyer. A shareholders’ meeting
of the Company shall have been validly held on or prior the Closing Date to resolve upon the approval of the appointment of the new directors and, if necessary, of the statutory auditors of the Company, by adopting resolutions in a form reasonably
acceptable to Buyer, and such directors and (if applicable) statutory audited shall have been validly appointed.
(viii) Settlement
Agreement with Luca Benatti. The Company and Luca Benatti shall have executed a settlement agreement pursuant to section 2113, sub-section 4, of the Italian Civil Code
(so-called “accordo di conciliazione in sede protetta”), in a form substantially corresponding to the one provided under Exhibit D (the “Benatti Settlement Agreement”),
whereby Luca Benatti waives any claim he might be entitled to in connection with his employment relationship with the Company and/or termination thereof.
68
(ix) IRS Forms. Each Shareholder shall have delivered a valid, complete and duly
executed IRS Form W-9 or the appropriate version of IRS Form W-8, as applicable, for such Shareholder.
(x) FIRPTA. The Company shall have delivered a properly executed statement, in accordance with Treasury Regulations Sections
1.897-2(h) and 1.1445-2(c)(3), certifying that the US Subsidiary is not and has not been a “United States real property holding corporation” (as defined in Section 897(c)(2) of the Code) during the applicable period specified in
Section 897(c)(1)(A)(ii) of the Code, together with the required notice to the IRS and written authorization for Buyer to deliver such notice and a copy of such statement to the IRS on behalf of the US Subsidiary upon the Closing.
(xi) Data Room. No less than five (5) days prior to the Closing, the Company shall have delivered a copy of the contents of the
Data Room to the Buyer on two USB flash drives or in such other format as is agreed in writing between the Company and the Buyer.
(xii)
Company Securities. All actions contemplated by Section 2.5 shall have been completed by the Company or the Shareholders, as applicable.
(xiii) Key Employees. Each Key Employee shall remain employed by the Company or the US Subsidiary in a substantially similar role as
on the date of this Agreement and shall not have expressed an intention to terminate his or her employment with the Company or Buyer (or one of its Subsidiaries, as applicable) following the Closing, and no Key Employee shall have revoked, or
expressed an intention to revoke, such Key Employee’s Key Employment Agreement.
(xiv) Accredited Investor Questionnaires.
Each Shareholder shall have provided to Buyer, and not amended or revoked, a duly executed Accredited Investor Questionnaire showing that each such Shareholder is an Accredited Investor.
(xv) Patent Confirmatory Assignments. The Company shall have delivered confirmatory assignments from all listed inventors to the
Company for all Patents of Company Registered IP.
(xvi) Transaction Expense Letters. The Company shall have delivered written
acknowledgements from each Person to whom a Transaction Expense is paid or payable confirming (A) the total amount payable to such Person (together with wiring instructions) and (B) that, upon receipt of the amount in the foregoing clause
(A), such Person will have been paid in full and is not and will not be owed any other Transaction Expenses
(xvii) Transaction
Expenses. The Transaction Expenses in the aggregate as of immediately prior to Closing shall not exceed $4,203,000 without Buyer’s prior written consent. The Transaction Expenses payable in the aggregate to such Persons and up to the
threshold amounts set forth on Schedule 9.2(d)(xvii) (each threshold set forth on Schedule 9.2(d)(xvii) of the Company Shareholder Letter, a “Transaction Expense Threshold”).
(xviii) EIB Loan. The Company shall have delivered a consent to the Transactions from the European Investment Bank providing that the
EIB Loan shall remain outstanding and no payment thereunder shall be triggered as a result of the Transactions, in a form reasonably satisfactory to Buyer.
69
9.3 Additional Conditions to Obligations of Shareholders. The obligation of
Shareholders to effect the Closing is subject to the satisfaction of each of the following conditions, any of which may be waived, in writing, exclusively by Shareholders:
(a) Representations and Warranties. The representations and warranties of Buyer contained in this Agreement will be true and correct in
all respects, in each case both as of the Agreement Date and (except to the extent such representations and warranties speak as of an earlier date, which representations and warranties shall be true and correct as of such date) as of the Closing
Date, except for any failures to be true and correct that (without giving effect to any qualifications or limitations as to materiality or Material Adverse Effect), individually or in the aggregate, have not had and would not reasonably be expected
to have a Material Adverse Effect on Buyer’s ability to consummate the Transactions.
(b) Performance of Obligations of Buyer.
Buyer shall have performed in all material respects all covenants required to be performed by it under this Agreement at or prior to the Closing Date.
(c) Material Adverse Effect. Since the Agreement Date, there shall not have occurred a Material Adverse Effect on Buyer.
(d) Listing. The Transaction Securities shall have been approved for listing on Nasdaq, subject to official notice of issuance.
(e) Officer’s Certificate. Buyer shall have delivered a certificate from Buyer, signed by an executive officer of
Buyer in such capacity, certifying as to the satisfaction of the conditions specified in Sections 9.3(a) and 9.3(b).
ARTICLE X
TERMINATION
OF AGREEMENT
10.1 Termination by Mutual Consent.
This Agreement may be terminated at any time prior to the Closing by the mutual written consent of Buyer and Company.
10.2 Unilateral Termination.(a) Either Buyer or the Company, by giving written notice to the other, may terminate this Agreement if a
Governmental Authority shall have issued a nonappealable final order, decree or ruling or taken any other action, in each case having the effect of permanently restraining, enjoining or otherwise prohibiting the Transactions; provided, however, the
right to terminate this Agreement under this Section 10.2(a) shall not be available to a Party if the failure by such Party or its Affiliates to comply with any provision of this Agreement has been a substantial cause or,
or substantially resulted in, such order, decree, ruling or action.
(b) Either Buyer or the Company, by giving written notice to the
other, may terminate this Agreement if the Closing shall not have occurred by 11:59 p.m. (Pacific Time) on December 31, 2023 (the “Outside Date”); provided, however, that the right to terminate this Agreement pursuant to this
Section 10.2(b) shall not be available to any Party whose breach of a representation or warranty or covenant made under this Agreement by such Party results in Closing not having occurred on or before such date.
70
(c) Either Buyer or the Company, by giving written notice to the other, may terminate this
Agreement at any time prior to the Closing if the other, or, in the case of a termination by Buyer, the Company or any Shareholder, has committed a material breach of (i) any of its representations and warranties under Article III,
Article IV or Article V, as applicable, or (ii) any of its covenants under Article VII or Article VIII, as applicable, and the breaching Party has not cured such breach within twenty (20) Business Days after the
Party seeking to terminate this Agreement has given the other Party written notice of such breach and its intention to terminate this Agreement pursuant to this Section 10.2(c) (provided, however, that no such cure period
shall be available or applicable to any such breach which by its nature is incapable of being be cured) and if not cured on or prior to the Closing Date, such breach would result in the failure of any of the conditions set forth in
Section 9.2(a), Section 9.2(b), Section 9.3(a) or Section 9.3(b), as applicable, to be fulfilled or satisfied (treating the Closing Date for such
purpose as of the date of such breach); provided, however, that the right to terminate this Agreement under this Section 10.2(c) shall not be available to a Party if the Party is at that time in material breach of this
Agreement.
10.3 Effect of Termination. This Agreement may only be terminated in the circumstances described in
Section 10.1 and Section 10.2 and pursuant to a written notice delivered by the applicable Party to the other applicable Parties, which sets forth the basis for such termination, including the
provision of Section 10.1 or Section 10.2 under which such termination is made. In the event of a termination of this Agreement as provided in Section 10.1 or
Section 10.2, this Agreement shall forthwith become void and there shall be no liability or obligation on the part of Buyer, Company, or their respective Representatives; provided, however, that the provisions of this
Section 10.3 (Effect of Termination) and Article XII (Miscellaneous) shall remain in full force and effect and survive any termination of this Agreement, nothing herein shall relieve any Party from liability in
connection with any willful breach of any of such Party’s representations, warranties or covenants contained herein or any fraud of such Party, in either case, prior to the termination of this Agreement, and subject to the right to seek
injunctions, specific performance or other equitable relief in accordance with Section 12.6, the Parties’ sole right prior to the Closing with respect to any breach of any representation, warranty or covenant contained
herein by another Party or with respect to the Transactions shall be the right, if applicable, to terminate this Agreement pursuant to Section 10.2.
ARTICLE XI
TAX MATTERS
11.1 Preparation of Tax Returns
(a) The Company Group shall prepare and timely file, or cause to be prepared and timely filed, all Tax Returns that are required to be filed
by any Company Group Member (taking into account valid extensions obtained in the Ordinary Course of Business) on or before the Closing Date (each, a “Company Prepared Return”), and shall pay, or cause to be paid, all Taxes of each
Company Group Member due on or before the Closing Date. All Company Prepared Returns shall be prepared by treating items on such Tax Returns in a manner consistent with the past practices of the Company Group with respect to such items, except as
otherwise required by Applicable Law. At least twenty (20) days prior to filing a Company Prepared Return that is an income or other material Tax Return and that is required to be filed by any Company Group Member after the Agreement Date, the
Company shall submit a copy of such Tax Return to Buyer for Buyer’s review and approval (which shall not be unreasonably withheld, conditioned, or delayed) and shall make such revisions to such Tax Return as are requested by Buyer.
(b) Buyer shall prepare and file, or cause to be prepared and filed, all Tax Returns of each Company Group Member for a Pre-Closing Tax Period after the Closing Date (each, a “Buyer Prepared Return”). In the event that any Buyer Prepared Return that is an income Tax Return and shows any material Unpaid Pre-Closing Taxes that are subject to indemnification by the Shareholders pursuant to this Agreement, Buyer shall submit a copy of such Tax Return to the Holder Representative for review and comment at least twenty
(20) days (or, if such Tax Return is required to be filed within forty-five (45) days after the Agreement Date, as soon as reasonably practicable after the Agreement Date) prior to filing such Tax Return; provided that any failure or delay
in providing any such Tax Return to the Holder Representative shall not relieve the Indemnitors of any indemnification obligations with respect to such Tax Return, except to the extent the Indemnitors are actually prejudiced as a result thereof.
Buyer shall consider in good faith all reasonable comments timely received from the Holder Representative.
71
11.2 Cooperation. The Shareholders and the Holder Representative shall cooperate
fully, as and to the extent reasonably requested by any Buyer Party, in connection with the filing of any Tax Return with respect to any Company Group Member and the defense of any pending or threatened audit, Legal Proceeding, or assessment with
respect to Taxes of any Company Group Member. Such cooperation shall include the retention and (upon request therefor) the provision of records and information that are reasonably relevant to any such Tax Return, audit, Legal Proceeding, or
assessment and making employees available on a mutually convenient basis to provide additional information and explanation of any materials provided hereunder. Notwithstanding the foregoing or any other provision herein to the contrary, in no event
shall the Shareholders or the Holder Representative be entitled to review or otherwise have access to any income Tax Return, or information related thereto, of Buyer or any of its Affiliates (other than income Tax Returns of the Company Group for
taxable periods ending on or prior to the Closing Date).
11.3 Transfer Taxes. All Transfer Taxes shall be borne by the
Shareholders. The Persons required by Law shall, at the Shareholders’ expense, file all necessary Tax Returns and other documentation with respect to Transfer Taxes and, if required by Applicable Law, Buyer shall, and shall cause its Affiliates
to, join in the execution of any such Tax Returns and other documentation. The Parties shall reasonably cooperate in connection with such filing, including to reduce or eliminate Transfer Taxes.
11.4 Section 338(g) Election. Notwithstanding anything in this Agreement to the contrary, Buyer and its Affiliates shall
be entitled (but not obligated) to file an election under Section 338(g) of the Code and any corresponding state or local election with respect to the Transactions.
ARTICLE XII
MISCELLANEOUS
12.1
Public Announcements. The initial press release with respect to the execution of this Agreement shall be a joint press release agreed upon by Buyer and the Company. So long as this Agreement is in effect and has not been validly terminated
pursuant to Article X, neither Buyer, nor the Company Parties or any of their Representatives shall issue any press release or other public communications relating to the terms of this Agreement or the Transactions or use of the other Party’s
name or refer to the other Party directly or indirectly in connection with the Party’s relationship with the other Party in any media interview, advertisement, news release, press release or professional or trade publication, or in any print
media, whether or not in response to an inquiry, without the prior written approval of the other Party hereto, which consent shall not be unreasonably withheld, conditioned or delayed, unless required by Applicable Law, or the rules of any listing
authority or securities exchange (as advised by such Party’s outside legal counsel), and provided that the Party shall provide prior written notice to the other Party (to the extent legally permitted) and except as reasonably necessary for such
Party to obtain the consents and approvals of Third Parties contemplated by this Agreement. Notwithstanding anything to the contrary contained herein or in the Confidentiality Agreement, without prior consent of the other Party, each Party
(a) may communicate information that is not confidential information of any other Party and which does not disclose terms of the Transactions that have not previously been publicly disclosed by Buyer to financial analysts, investors and media
representatives in a manner consistent with its past practice in compliance with Applicable Law and (b) may disseminate the information included in a prior press release or other public announcement which was previously approved in writing for
external distribution by the other Party. Notwithstanding anything herein to the contrary, following Closing, the Holder Representative shall be permitted to disclose information as required by Applicable Law (and provided that the Holder
Representative shall provide prior written notice to Buyer to the extent legally permitted) or to advisors and representatives of the Holder Representative and to the Shareholders, in each case who have a need to know such information, provided that
such persons are subject to confidentiality obligations with respect thereto as least as restrictive as those herein.
72
12.2 Governing Law; Exclusive Jurisdiction; Waiver of Jury Trial.
(a) This Agreement and any claim, cause of action (in law or equity), controversy or dispute arising out of or related to this Agreement, any
of the Transactions, the relationship of the Parties, and/or the interpretation, performance and enforcement of the rights and duties of the Parties, whether arising in contract, tort, statutory or otherwise, shall be governed by, and construed in
accordance with, the laws of the State of Delaware (including in respect of the statute of limitations or other limitations period applicable to any such claim, controversy or dispute), in each case without giving effect to any conflicts-of-law or other principle that might require the application of the laws of any other jurisdiction.
(b) Each of the Parties irrevocably agrees that any legal action or proceeding arising out of or relating to this Agreement or for recognition
and enforcement of any judgment in respect hereof brought by any other Party or its successors or assigns may be brought and determined by the Court of Chancery of the State of Delaware or if jurisdiction is not proper in such court, in Superior
Court seated in New Castle County Delaware, or if jurisdiction is not proper in such court, a federal court sitting in Wilmington, Delaware (and in each case, any appellate courts thereof) and each of the Parties hereby irrevocably submits to the
exclusive jurisdiction of the aforesaid courts for itself and with respect to its property, generally and unconditionally, with regard to any such action or proceeding arising out of or relating to this Agreement and the transactions contemplated
hereby (and agrees not to commence any action, suit or proceeding relating thereto except in such courts). Each of the Parties further agrees to accept service of process in any manner permitted by such court. Each of the Parties hereby
irrevocably and unconditionally waives, and agrees not to assert, by way of motion or as a defense, counterclaim or otherwise, in any action or proceeding arising out of or relating to this Agreement or the transactions contemplated hereby,
(i) any claim that it is not personally subject to the jurisdiction of the above-named courts for any reason other than the failure lawfully to serve process, (ii) that it or its property is exempt or immune from jurisdiction of any such
court or from any legal process commenced in such court (whether through service of notice, attachment prior to judgment, attachment in aid of execution of judgment, execution of judgment or otherwise) and (iii) to the fullest extent permitted
by Law, that (A) the suit, action or proceeding in any such court is brought in an inconvenient forum, (B) the venue of such suit, action or proceeding is improper or (C) this Agreement, or the subject matter hereof, may not be
enforced in or by such courts.
(c) EACH OF THE PARTIES TO THIS AGREEMENT HEREBY IRREVOCABLY WAIVES ALL RIGHT TO A TRIAL BY JURY IN ANY
ACTION, PROCEEDING OR COUNTERCLAIM ARISING OUT OF OR RELATING TO THIS AGREEMENT OR THE TRANSACTIONS CONTEMPLATED HEREBY.
12.3
Assignment; Binding Upon Successors and Assigns. No Party may assign any of its rights or obligations hereunder without the prior written consent of the Company and Buyer. This Agreement shall be binding upon and inure to the benefit of the
Parties and their respective successors and permitted assigns. Any assignment in violation of this provision shall be void.
73
12.4 Severability. If any provision of this Agreement, or the application thereof,
shall for any reason and to any extent be invalid or unenforceable, then the remainder of this Agreement and the application of such provision to other persons or circumstances shall be interpreted so as reasonably to effect the intent of the
Parties. The Parties further agree to replace such void or unenforceable provision of this Agreement with a valid and enforceable provision that shall achieve, to the extent possible, the economic, business and other purposes of the void or
unenforceable provision.
12.5 Counterparts. This Agreement may be executed in any number of counterparts, each of which shall be
an original as regards any Party whose signature appears thereon and all of which together shall constitute one and the same instrument. This Agreement shall become binding when one or more counterparts hereof, individually or taken together, shall
bear the signatures of all Parties reflected hereon as signatories.
12.6 Other Remedies. Except as otherwise expressly provided
herein, any and all remedies herein expressly conferred upon a Party hereunder shall be deemed cumulative with and not exclusive of any other remedy conferred hereby or by law on such Party, and the exercise of any one remedy shall not preclude the
exercise of any other. The Parties agree that irreparable damage would occur in the event that any of the provisions of this Agreement were not performed in accordance with their specific terms or were otherwise breached. It is accordingly agreed
that the Parties shall be entitled to seek an injunction or injunctions to prevent breaches of this Agreement and to seek to enforce specifically the terms and provisions hereof, without the requirement to post any bond or other security or to prove
that money damages would be inadequate.
12.7 Amendments and Waivers. Any term or provision of this Agreement may be amended,
supplemented or modified only by execution of a written instrument signed by Buyer, Company and, to the extent such amendment, supplement or modification affects any of the rights or obligations of the Holder Representative, the Holder
Representative. The observance of any term of this Agreement may be waived (either generally or in a particular instance and either retroactively or prospectively), only by a writing signed by the Party to be bound thereby. The waiver by a Party of
any breach hereof or default in the performance hereof shall not be deemed to constitute a waiver of any other default or any succeeding breach or default. At any time prior to the Closing, the Company Parties and Buyer may, to the extent legally
allowed, (a) extend the time for the performance of any of the obligations or other acts of the other, (b) waive any inaccuracies in the representations and warranties made to it contained herein or in any document delivered pursuant
hereto, and (c) waive compliance with any of the agreements or conditions for its benefit contained herein. No such waiver or extension shall be effective unless signed in writing by the Party against whom such waiver or extension is asserted.
The failure of any Party to enforce any of the provisions hereof shall not be construed to be a waiver of the right of such Party thereafter to enforce such provisions.
12.8 Expenses. Whether or not the Transactions are consummated, Buyer shall bear its respective legal, accountants, and financial
advisory fees and other expenses incurred with respect to this Agreement and the Transactions.
12.9 Notices. All notices and other
communications required or permitted under this Agreement shall be in writing and shall be either hand delivered in person, sent by certified or registered first-class mail, postage pre-paid, sent by
nationally recognized express courier service or sent by email. Such notices and other communications shall be effective upon receipt if hand delivered or sent by email (with affirmative confirmation of receipt), three Business Days after mailing if
sent by mail, and one Business Day after dispatch if sent by express courier, to the following addresses, or such other addresses as any Party may notify the other Parties in accordance with this Section 12.9:
If to Buyer or IT Buyer:
Quince
Therapeutics, Inc.
601 Gateway Blvd, Suite 1250
74
South San Francisco CA 94080
Attention: Dirk Thye
Email: dthye@quincetx.com
with a copy to:
Cooley LLP
3175 Hanover Street
Palo Alto, CA 94304
Attention: Gordon Ho
Email: gho@cooley.com
If to the Company and/or any of the Shareholders:
EryDel S.p.A.
Via A. Meucci, 3
20091 Bresso (MI)
Italy
Attention: Luca Benatti
Email: luca.benatti@erydel.com
with a copy to:
Avv. Luca Andrea Garibaldi
Via Paolo Sarpi 12
20154 – Milan, Italy
Email: Luca@studiogaribaldi.it
Certified e-mail: eMail: LucaGaribaldi@legalmail.it
and to:
Studio Legale Associato in associazione con Clifford Chance
Via Broletto, 16
20121 – Milan, Italy
Attention: Avv. Francesca Casini, dott. Dario Carnazza
Email: francesca.casini@cliffordchance.com, dario.carnazza@cliffordchance.com
If to the Holder Representative:
Shareholder Representative Services LLC
950 17th Street, Suite 1400
Denver, CO 80202
Attention: Managing Director
Email: deals@srsacquiom.com
Telephone: (303) 648-4085
12.10 Interpretation; Rules of Construction. When a reference is made in this Agreement to Exhibits, Sections or Articles, such
reference shall be to an Exhibit to, Section of or Article of this Agreement, respectively, unless otherwise indicated. The words “include”, “includes” and “including” when used herein shall be deemed in each case to be
followed by the words “without limitation”. The
75
words “herein,” “hereto,” and “hereby” and other words of similar import in this Agreement shall be deemed in each case to refer to this Agreement as a whole and not
to any particular Section or subsection of this Agreement. The defined terms herein shall apply equally to both the singular and plural forms of the terms defined. The headings contained in this Agreement are for reference purposes only and shall
not affect in any way the meaning or interpretation of this Agreement. The Parties agree that they have been represented by legal counsel during the negotiation and execution of this Agreement and, therefore, waive the application of any law,
regulation, holding or rule of construction providing that ambiguities in an agreement or other document shall be construed against the Party drafting such agreement or document. Any dollar amount included herein shall not be used as a basis for
interpreting the terms “material,” “materially,” “materiality,” “Material Adverse Effect” or any other similar qualification in this Agreement. References to “U.S. Dollars” or “$” shall
mean the lawful currency of the United States of America. References to “Euros” or “€” shall mean the lawful currency of the European Union. For purposes of Article III of this Agreement, references to amounts in U.S.
Dollars or Euros shall include the equivalent value, as of the Agreement Date, of any applicable foreign currency, including for purposes of the thresholds in Section 3.11.
12.11 Third Party Beneficiary Rights. No provisions of this Agreement are intended, nor shall be interpreted, to provide or create any
third party beneficiary rights or any other rights of any kind in, or deemed to have been executed for the benefit of, any client, customer, employee, Affiliate, stockholder or partner of any Party or any other Person that is not a Party hereto or a
successor or permitted assign of such Party, and all provisions hereof shall be personal solely between the Parties.
12.12
Release. Effective as of the Closing, each Shareholder, on behalf of itself and any Person who may be bound by any such Shareholder (including such Shareholder’s current or former affiliates, officers, directors, employees, managers,
partners, principals, advisors, agents, stockholders, members, investors, equity holders or other representatives (including attorneys, accountants, consultants, bankers and financial advisors), heirs, beneficiaries, estates, executors,
administrators, trustees, successors or assigns), hereby unconditionally and irrevocably and forever releases and discharges the Company, Buyer, each of their subsidiaries, successors and assigns, and any present or former directors, managers,
officers, employees, agents, lenders, investors, partners, principals, members, managers, shareholders or equity holders of any of the foregoing Persons (each, a “Released Party”), of and from, and hereby unconditionally and
irrevocably waives, any and all claims, debts, losses, expenses, legal proceedings, covenants, liabilities, suits, judgments, Losses, actions and causes of action, obligations, accounts, and liabilities of any kind or character whatsoever, known or
unknown, suspected or unsuspected, in Contract, direct or indirect, at law or in equity that such party ever had, now has or ever may have or claim to have against any Released Party, for or by reason of any matter, circumstance, event, action,
inaction, omission, cause or thing whatsoever arising prior to the Closing (including in respect of the management or operation of the Company); provided, however, that this release expressly does not extend to, and shall not be interpreted to in
any way release, waive, impair or limit the right of such Shareholder to (w) the consideration set forth in Article II of this Agreement or in any offer letter or other employment-related agreement effective only at Closing entered into
with Buyer or its subsidiaries in connection with the Transactions, (x) receive compensation or other benefits owed to such Shareholder that are accrued in Company Net Working Capital, as finally determined pursuant to this Agreement,
(y) unreimbursed claims under employee health and welfare plans, consistent with terms of coverage and entitlement to continuation coverage benefits or any other similar benefits required to be provided by Applicable Law or (z) be
indemnified in accordance with any applicable indemnification agreement or directors and officers liability insurance policy. The Shareholders, on behalf of themselves and their Affiliates and Representatives, expressly waive all rights afforded by
any statute that limits the effect of a release with respect to unknown claims. Each Shareholder acknowledges and affirms that it is familiar with Section 1542 of the Civil Code of the State of California, which provides as follows: A GENERAL
RELEASE DOES NOT EXTEND TO CLAIMS WHICH THAT THE CREDITOR OR RELEASING PARTY DOES NOT KNOW OR
76
SUSPECT TO EXIST IN HIS OR HER FAVOR AT THE TIME OF EXECUTING THE RELEASE, WHICH AND THAT IF KNOWN BY HIM OR HER, WOULD HAVE MATERIALLY AFFECTED HIS OR HER SETTLEMENT WITH THE DEBTOR OR RELEASED
PARTY. Each Shareholder hereby waives and relinquishes any rights and benefits that such Shareholder may have under Section 1542 or any similar statute or common law principle of any jurisdiction to the fullest extent permitted by Applicable
Law. The Shareholders understand the significance of this release of unknown claims and waiver of statutory protection against a release of unknown claims, and acknowledge and agree that this waiver is an essential and material term of this
Agreement. Each Shareholder represents that such Shareholder has not made any assignment, conveyance or Transfer of any kind of any claim released pursuant to this Section 12.12. Each Shareholder acknowledges and intends
that this Section 12.12 shall be effective as a bar to each claim released hereby.
12.13 Shareholder
Acknowledgement. Each Shareholder agrees and acknowledges that it has conducted its own independent investigation, review and analysis of the results of operations, prospects, condition (financial or otherwise) and assets of Buyer. In entering
into this Agreement, each Shareholder has relied solely upon its own investigation and analysis and the representations and warranties of Buyer set forth in Article V, and each Shareholder acknowledges and agrees that, except for the
representations and warranties of Buyer expressly set forth in Article V, neither Buyer nor any of its respective Representatives nor any other Person acting on Buyer’s behalf makes or has made, and such Shareholder is not relying on and
has not relied on, any representation or warranty, either express or implied, as to the accuracy or completeness of any of the information provided or made available to such Shareholder or any of its Representatives, or otherwise with respect to
Buyer, its business or the Transactions. Without limiting the generality of the foregoing, each Shareholder acknowledges and agrees that neither Buyer nor any of its Representatives or any other Person has made, and such Shareholder is not relying
on and has not relied on, any representation or warranty to such Shareholder or any of its Representatives with respect to (a) any projections, estimates or budgets for Buyer or (b) any materials, documents or information relating to Buyer
or its business made available to the Company, any Shareholder or any of their respective Representatives in any “data room,” online data site, confidential memorandum, other offering materials or otherwise, except, in the case of
(a) and (b), as set forth in the representations and warranties set forth in Article V.
12.14 Entire Agreement. This
Agreement, the exhibits and schedules hereto and the Ancillary Agreements constitute the entire understanding and agreement of the Parties with respect to the subject matter hereof and supersede all prior and contemporaneous agreements or
understandings, inducements or conditions, express or implied, written or oral, between the Parties other than the Confidentiality Agreement. The express terms hereof control and supersede any course of performance or usage of the trade inconsistent
with any of the terms hereof. In furtherance of, and without limiting the foregoing, the Parties acknowledge that (i) the execution of this Agreement is the culmination of extensive negotiations between them, all of which were intended to be non-binding upon the Parties until memorialized in the executed copy of this Agreement and none of which should be construed as having created any type of oral agreement between the Parties, and (ii) except as
expressly provided in Article III (as modified by the Company Disclosure Schedule), Article IV, and Article V (as modified by the Buyer Disclosure Schedule), none of the Parties hereto has made or is making any representations
or warranties whatsoever, implied or otherwise.
77
***
|
| In sign of full and unconditional acceptance: |
|
| Erydel Italy, Inc |
|
| /s/ Dirk Thye |
| Name: Dirk Thye |
| Title: Chief Executive Officer |
***
|
| In sign of full and unconditional acceptance: |
|
| Quince Therapeutics, Inc. |
|
| /s/ Dirk Thye |
| Name: Dirk Thye |
| Title: Chief Executive Officer |
***
|
| In sign of full and unconditional acceptance: |
|
| Erydel S.p.A. |
|
| /s/ Luca Benatti |
| Name: Luca Benatti |
| Title: Chief Executive Officer |
***
Shareholder Representative Services LLC, Solely in its capacity as representative of the Shareholders
|
| /s/ Sam Riffe |
| Name: Sam Riffe |
| Title: Managing Director |
***
|
| In sign of full and unconditional acceptance: |
|
| The Shareholders and the Managers set forth on Schedule II |

Exhibit 99.1 Quince Therapeutics to Acquire EryDel July 24,
2023

Forward-looking statements Statements in this presentation contain
“forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995 as contained in Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as
amended, which are subject to the “safe harbor” created by those sections. All statements, other than statements of historical facts, may be forward-looking statements. Forward-looking statements contained in this news release may be
identified by the use of words such as “believe,” “may,” “should,” “expect,” “anticipate,” “plan,” “believe,” “estimated,” “potential,”
“intend,” “will,” “can,” “seek,” or other similar words. Examples of forward-looking statements include, among others, statements relating to Quince’s acquisition of EryDel; the timing of the
closing of the transaction; the expected benefits of the transaction, including the continued current and future clinical development and potential expansion of EryDel assets, related platform, and related timing and costs; the strategic development
path for EryDex; planned FDA and EMA submissions and clinical trials and timeline, prospects, and milestone expectations; the timing and success of the clinical trials and related data, including plans and the ability to initiate, fund, conduct
and/or complete current and additional studies; the potential therapeutic benefits, safety, and efficacy of EryDex; statements about its ability to obtain, and the timing relating to, further development of EryDex, regulatory submissions and
interactions with regulators; therapeutic and commercial potential; the integration of EryDel’s business, operations, and employees into Quince; Quince’s future development plans and related timing; its cash position and projected cash
runway; the company’s focus, objectives, plans, and strategies; and the ability to execute on any strategic transactions. Forward-looking statements are based on Quince’s current expectations and are subject to inherent uncertainties,
risks, and assumptions that are difficult to predict and could cause actual results to differ materially from what the company expects. Further, certain forward- looking statements are based on assumptions as to future events that may not prove to
be accurate. Factors that could cause actual results to differ include, but are not limited to, the risks and uncertainties described in the section titled “Risk Factors” in the company’s Quarterly Report on Form 10-Q filed with
the Securities and Exchange Commission (SEC) on May 15, 2023, and other reports as filed with the SEC. Forward-looking statements contained in this news release are made as of this date, and Quince undertakes no duty to update such information
except as required under applicable law. 2

Transformative acquisition with value-creating clinical milestones Phase
3 lead asset EryDex targets Ataxia-Telangiectasia (A-T) with no currently approved treatments and estimated $1+ billion peak sales opportunity EryDex designed for controlled, slow release of dexamethasone over several weeks without long-term
toxicity typically associated with chronic steroid administration Plan to enroll first patient in global Phase 3 trial of EryDex in second quarter of 2024 with NDA submission targeted by end of 2025, assuming positive study results Well-capitalized
into 2026 with ability to fully fund EryDex expected through Phase 3 trial and to NDA submission, assuming positive study results 3 3 3

EryDel significant achievements Headquarters • Bresso, Italy 20+
years of work on autologous intracellular drug encapsulation (AIDE) technology platform Manufacturing $100+ million invested since founding out of University of Urbino, Italy • Medolla, Italy EryDex designated as orphan drug for A-T treatment
from FDA and EMA Leadership • Luca Benatti: CEO Special protocol assessment (SPA) in place with FDA for single Phase 3 clinical trial of EryDex – sufficient for NDA submission, assuming positive study results • Guenter Janhofer:
CMO • Giovanni Mambrini: COO EryDex efficacy and safety profile demonstrated in prior Phase 3 clinical • Thomas Sabia: CCO trial of A-T patients Employees Open label extension (OLE) and compassionate use data demonstrates up to 10+ years
of chronic steroid administration without typical safety issues • 21 4 4

EryDel acquisition transaction details Overview Stock-for-stock upfront
exchange of Quince stock and potential downstream milestone cash payments of up to $485 million Unanimously approved by both companies’ Board of Directors • Up to $5 million in development milestones • $25 million at NDA acceptance
• $60 million in approval milestones EryDel stockholders to own maximum of approximately 16.7% of combined company – subject to downward adjustment • $395 million in market and sales milestones • No royalties paid to EryDel
stockholders Governance & Leadership Structure Approvals & Closing David Lamond remains Chairperson EryDel to operate as wholly owned subsidiary Subject to certain regulatory approvals and of Quince Board of Directors of Quince with ongoing
presence in Italy other closing conditions Retain EryDel team and keep organization intact Dirk Thye remains Quince Chief Executive Officer Expected to close in third quarter 2023 and Director Assumption of $13 million (€10 million principal)
EIB loan with scheduled payments beginning in Quince Board of Directors expanded with the second half of 2026 addition of EryDel representative Luca Benatti 5
Autologous intracellular drug encapsulation (AIDE) technology Unique
drug/device combination enables automated process for autologous intracellular drug encapsulation Platform capable of expansion to other drugs or biologics, including enzyme replacement therapy 6

Fully automated autologous point-of-care treatment Patient’s blood
collected at the point-of-care for fully automated bedside procedure completed within two hours Bedside Blood Collection Erythrocytes loaded onto the Red Cell Loader using EryKit Erythrocytes swollen and pores opened via multiple steps using series
of process solutions, then dexamethasone added Intracellular Drug Encapsulation Dexamethasone loaded erythrocytes washed, isolated, and prepared and then reinfused into patient Designed for improved drug benefits including: Encapsulated Drug •
Slow, controlled release• Improved biodistribution Re-Infusion • Prolonged half-life• Mitigates steroid toxicity 7

No currently approved treatments for A-T patients A-T is an inherited
neurodegenerative and immunodeficiency disorder caused by mutations in ATM gene Approximately 10,000 A-T patients in U.S., U.K., and EU4 countries Neurological symptoms worsen until patients are wheelchair-bound, usually by adolescence Median
lifespan of approximately 25 years, with mortality due to infections and malignancy Currently no approved treatments for A-T and no known effective approaches to delay progression of disease 8

EryDex efficacy and safety profile EryDex ATTeST Trial demonstrated in
prior clinical trial Completed largest global study of A-T patients in Phase 3 ATTeST clinical trial and OLE • Double-blind, placebo-controlled study (N=175) over 6-month treatment period with 12-month OLE (N=104) • Patients randomized
1:1:1 to EryDex low dose, high dose, or placebo • Primary endpoint: mICARS – Secondary endpoints: CGI-C • QOL • VABS EryDex slowed neurological deterioration in all ages of A-T patients with statistically significant effect
in 6 to 9 year subgroup 12-month safety analysis demonstrated EryDex well-tolerated with no major adverse events typically associated with chronic steroid administration Pursuing regulatory activities related to partial clinical hold in U.S. related
to EryKit treatment consumables, in addition to activities to support potential MAA submission of EryDex • CE mark already obtained in Europe for treatment device and consumables kit 9 Note: mICARS = Modified International Cooperative Ataxia
Rating Scale • CGI-C = Clinical Global Impression – Change • QOL = Quality of Life • VABS = Vineland Adaptive Behavior Scales 9 9
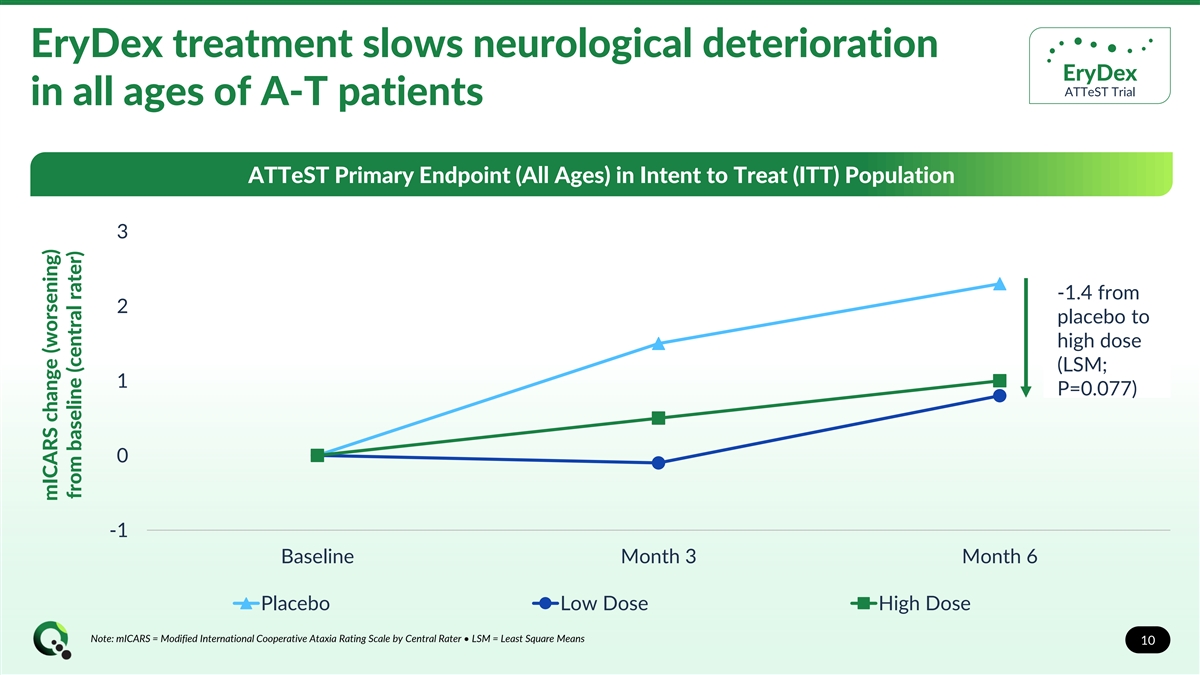
EryDex treatment slows neurological deterioration EryDex ATTeST Trial
in all ages of A-T patients ATTeST Primary Endpoint (All Ages) in Intent to Treat (ITT) Population 3 -1.4 from 2 placebo to high dose (LSM; 1 P=0.077) 0 -1 Baseline Month 3 Month 6 Placebo Low Dose High Dose Note: mICARS = Modified International
Cooperative Ataxia Rating Scale by Central Rater • LSM = Least Square Means 10 mICARS change (worsening) from baseline (central rater)

EryDex treatment showed statistically significant EryDex ATTeST Trial
effect in 6 to 9 year subgroup Primary population selected for Phase 3 NEAT clinical trial ATTeST 6-9 Year Subgroup, mICARS 6 4 -4.8 from placebo to high dose 2 (LSM; P=0.0016) 0 -2 Baseline Month 3 Month 6 Placebo Low Dose High Dose Note: mICARS =
Modified International Cooperative Ataxia Rating Scale by Local Rater • LSM = Least Square Means 11 mICARS change (worsening) from baseline (local rater)
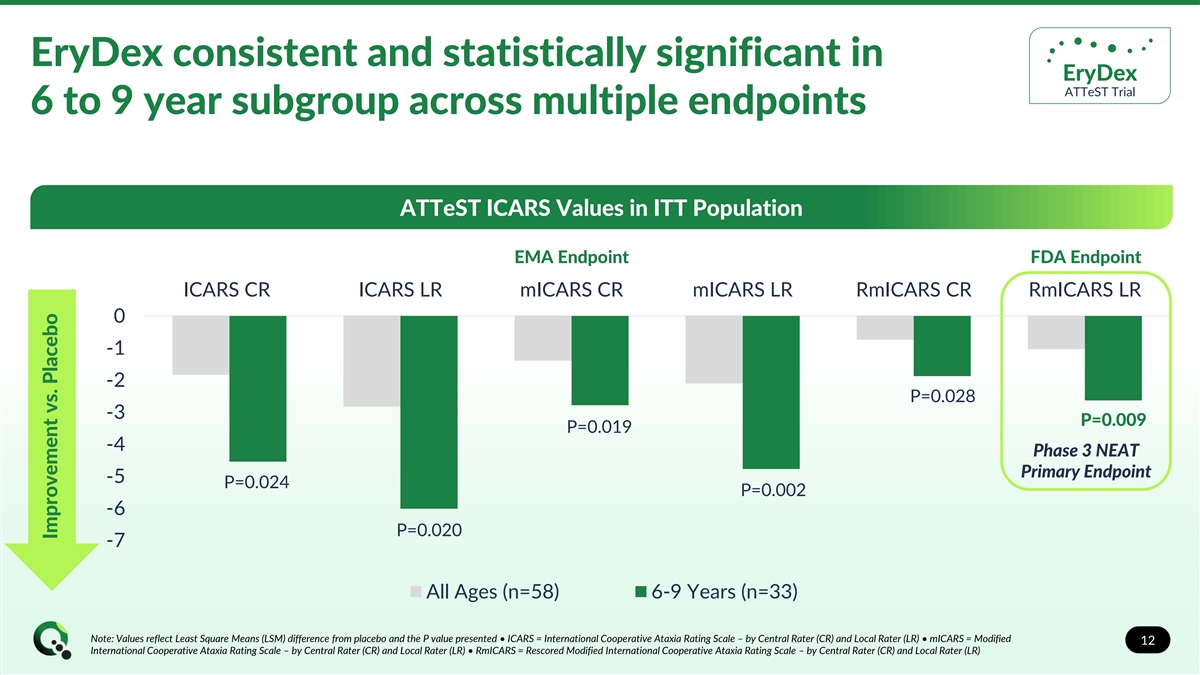
EryDex consistent and statistically significant in EryDex ATTeST Trial
6 to 9 year subgroup across multiple endpoints ATTeST ICARS Values in ITT Population EMA Endpoint FDA Endpoint ICARS CR ICARS LR mICARS CR mICARS LR RmICARS CR RmICARS LR 0 -1 -2 P=0.028 -3 P=0.009 P=0.019 -4 Phase 3 NEAT Primary Endpoint -5 P=0.024
P=0.002 -6 P=0.020 -7 All Ages (n=58) 6-9 Years (n=33) Note: Values reflect Least Square Means (LSM) difference from placebo and the P value presented • ICARS = International Cooperative Ataxia Rating Scale – by Central Rater (CR) and
Local Rater (LR) • mICARS = Modified 12 International Cooperative Ataxia Rating Scale – by Central Rater (CR) and Local Rater (LR) • RmICARS = Rescored Modified International Cooperative Ataxia Rating Scale – by Central Rater
(CR) and Local Rater (LR) Improvement vs. Placebo

No clinically meaningful adverse events with EryDex, including EryDex
ATTeST Trial those typically associated with chronic steroid treatment ATTeST: Initial Treatment Period ATTeST: Through Month 12 EDS-EP EDS-EP EDS-EP EDS-EP Non-switch Placebo Low Dose High Dose Low Dose High Dose Placebo (N=59) (N=59) (N=57) (N=59)
(N=57) (N=19) 73% 82% 73% 76% 88% 79% Patients With Any TEAE (%) Patients With Any Treatment–Related TEAE (%) 25% 37% 25% 32% 44% 26% 10% 12% 12% 14% 16% 21% Patients With Any Serious TEAE (%) 0 2% 0 2% 2% 5% Patients With Any Serious
Treatment–Related TEAE (%) Patients With Any TEAE Leading to Discontinuation (%) 0 4% 0 2% 4% 0 0 0 0 0 0 0 Patients With Any TEAE Leading to Death (%) Note: TEAE = Treatment Emergent Adverse Event • EDS-EP = EryDex System End Product
13
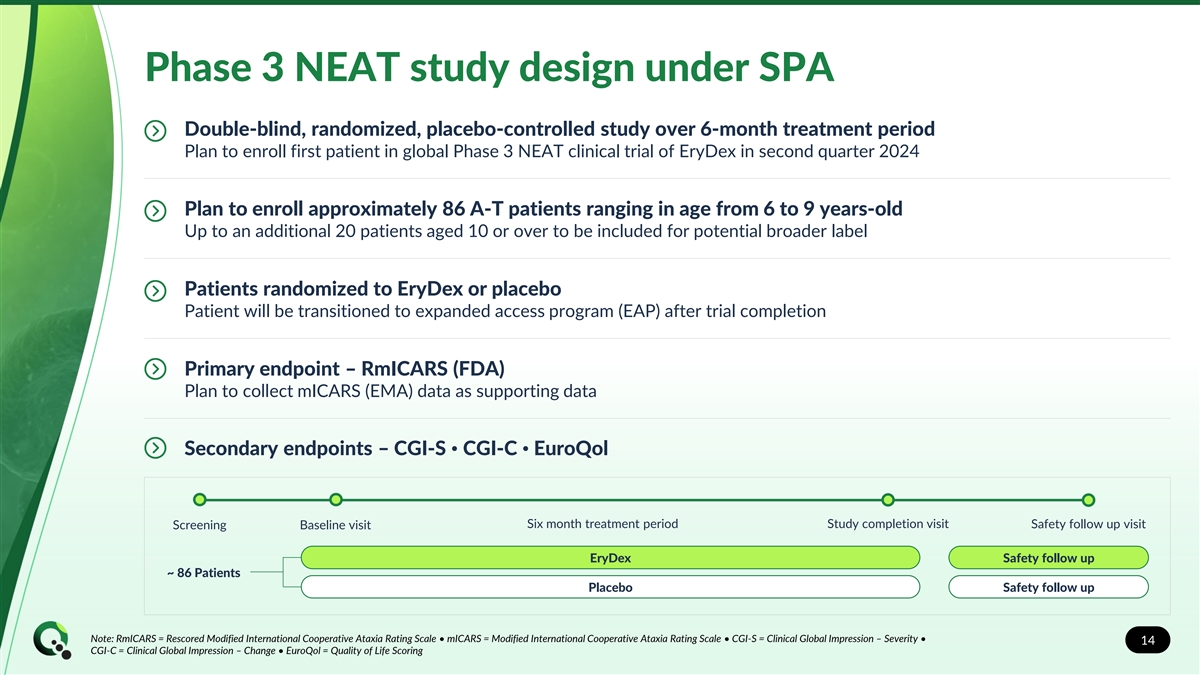
Phase 3 NEAT study design under SPA Double-blind, randomized,
placebo-controlled study over 6-month treatment period Plan to enroll first patient in global Phase 3 NEAT clinical trial of EryDex in second quarter 2024 Plan to enroll approximately 86 A-T patients ranging in age from 6 to 9 years-old Up to an
additional 20 patients aged 10 or over to be included for potential broader label Patients randomized to EryDex or placebo Patient will be transitioned to expanded access program (EAP) after trial completion Primary endpoint – RmICARS (FDA)
Plan to collect mICARS (EMA) data as supporting data Secondary endpoints – CGI-S • CGI-C • EuroQol Six month treatment period Study completion visit Safety follow up visit Screening Baseline visit EryDex Safety follow up ~ 86
Patients Placebo Safety follow up Note: RmICARS = Rescored Modified International Cooperative Ataxia Rating Scale • mICARS = Modified International Cooperative Ataxia Rating Scale • CGI-S = Clinical Global Impression – Severity
• 14 CGI-C = Clinical Global Impression – Change • EuroQol = Quality of Life Scoring

EryDex attractive commercial and rapid expansion potential EryDex for
A-T Approximately 10,000 A-T patients suffering from rare and debilitating pediatric disease in U.S., U.K., and EU4 countries with no currently approved therapies First-to-market potential with attractive pricing comparables and no known late-stage
competition $1+ billion Designated as orphan drug for A-T treatment from FDA and EMA estimated peak global sales opportunity for IP exclusivity until at least 2034 globally and at least 2035 in the U.S. A-T indication alone Rare and debilitating
disease potential expansion Target additional indications where chronic steroid treatment is the standard of care – or could be without long-term toxicity 15

Strong balance sheet with approximately $87.6 million in cash, cash
equivalents, and short term investments as of June 30, 2023 (unaudited) Well-capitalized into 2026 with ability to fully Capital efficient development plan funds: fund EryDex expected • EryDex Phase 3 NEAT study and NDA submission, assuming
positive study results through Phase 3 trial to • European regulatory activities related to potential MAA submission of EryDex NDA submission, assuming positive study Potential out-licensing of ex-U.S. results regional territories to provide
runway through approval 16 16

Seasoned leadership team Guenter Janhofer, M.D., Ph.D. Dirk Thye, M.D.
• 30+ years of healthcare industry experience in roles of increasing complexity and scope • 20+ years of experience in biotech company creation, R&D and executive leadership • Previously CMO at EryDel and CMO at BTG (acquired
by Boston Scientific for $4.2B) • Agenovir, Cidara, Cerexa and Peninsula • EryDel, BTG, Merck CEO CMO Brendan Hannah, M.B.A. Thomas Sabia, M.B.A. • 15+ years leading biotech BD, finance, and business operations • 20+ years of
drug commercialization and development experience across large, midsize, and small biotech organizations • Led BD at Agenovir (acquired by Vir for up to $290M) • Spark Therapeutics (a Roche Company), Sobi, CSL Behring, Sanofi •
Involved in $1B+ in transactions CBO CCO Giovanni Mambrini, MSc Collective experience includes 20+ regulatory • 20+ years of medical device technology experience with cross- approvals and more than $10 billion in functional expertise in
bringing complex programs to market • Co-founded EryDel with prior experience at Covidien, Dideco, aggregate transactions Livanova CTO 17

Key clinical and corporate milestones Second half of 2023 2024 2025
Close acquisition of EryDel in third Enroll first patient in Phase 3 NEAT Phase 3 NEAT clinical trial topline results quarter 2023 clinical trial in second quarter 2024 Target EryDex NDA submission Target resolution of partial clinical hold Initiate
pediatric study plan with FDA by end of 2025, assuming on improved EryKit treatment positive study results consumables in U.S. Determine additional indications for At least one Phase 2 study of EryDex EryDex and initiate R&D activities Initiate
start up activities for Phase 3 follow on indication NEAT clinical trial Initiate R&D activities for at least one Potential out-licensing of ex-U.S. Pursue European regulatory activities additional program utilizing AIDE regional territories to
provide runway related to potential MAA submission of technology platform through approval EryDex 18

Transformative acquisition with value-creating clinical milestones
Phase 3 lead asset EryDex targets Ataxia-Telangiectasia (A-T) with no currently approved treatments and estimated $1+ billion peak sales opportunity EryDex designed for controlled, slow release of dexamethasone over several weeks without long-term
toxicity typically associated with chronic steroid administration Plan to enroll first patient in global Phase 3 trial of EryDex in second quarter of 2024 with NDA submission targeted by end of 2025, assuming positive study results Well-capitalized
into 2026 with ability to fully fund EryDex expected through Phase 3 trial and to NDA submission, assuming positive study results 19 19 19
Exhibit 99.2
|
|
|
|
|
| NEWS RELEASE |
|
|
|

|
Quince Therapeutics to Acquire EryDel SpA and its Phase 3 Asset Targeting Ataxia-Telangiectasia with No
Currently Approved Treatments and Estimated $1+ Billion Peak Sales Opportunity
Well-capitalized into 2026 with ability to fully fund
lead asset EryDex expected through Phase 3 trial under special protocol assessment (SPA) and to NDA submission
EryDex utilizes
autologous intracellular drug encapsulation (AIDE) technology
designed for slow release of steroids over several weeks without
long-term toxicity
typically associated with chronic administration
Potential for rapid expansion of EryDex to other rare and debilitating disease indications
where chronic steroid treatment is or could become the standard of care
SOUTH SAN FRANCISCO, Calif. – July 24, 2023 – Quince Therapeutics, Inc. (Nasdaq: QNCX), a
biotechnology company focused on acquiring, developing, and commercializing innovative therapeutics that transform patients’ lives, today announced that the company has entered into an agreement to acquire EryDel SpA, a privately-held,
late-stage biotech company, in a stock-for-stock upfront exchange and potential downstream milestone cash payments. EryDel has developed an autologous intracellular drug
encapsulation (AIDE) technology and a Phase 3 lead asset, EryDex, targeting a rare fatal pediatric neurological disease, Ataxia-Telangiectasia (A-T), which currently has no approved treatments. Upon completion
of the transaction, EryDel stockholders will own approximately 16.7% of the combined company (subject to downward adjustment) and will be entitled to up to $485 million upon the achievement of development, regulatory, and commercial milestone
payments, with no royalties. The transaction, which has been unanimously approved by the Boards of Directors of both companies, is subject to certain regulatory approvals and other closing conditions and is expected to close in the third quarter of
2023.
Dirk Thye, M.D., Quince’s Chief Executive Officer, said, “We are highly enthusiastic and optimistic about our acquisition of this unique
drug/device combination technology platform and promising late-stage clinical asset to drive Quince’s next stage of growth. EryDel’s proprietary AIDE technology enables the autologous intracellular encapsulation and delivery of
dexamethasone in a controlled, slow-release manner that has the potential to allow chronic administration of steroids over many months or years with a favorable safety profile. This represents a tremendous opportunity to target not only A-T, but also the potential to expand into several debilitating rare diseases where chronic steroid treatment is the standard of care – or could be in the absence of long-term steroid toxicity. Upon the close
of the acquisition, we will quickly focus our considerable development expertise and financial resources toward advancing the lead asset EryDex for A-T through a single global Phase 3 clinical trial under a
SPA already in place with the FDA to an anticipated NDA submission, assuming positive study results.”
Luca Benatti, EryDel’s Chief Executive
Officer, said, “EryDel’s acquisition by Quince offers the opportunity to advance our innovative, point-of-care autologous intracellular encapsulation
technology through development to commercialization and to fulfill our mission to provide the first treatment for patients living with the devastating disease of A-T. Quince’s effort will be supported
601 Gateway Boulevard
• Suite 1250 • South San Francisco • California • 94080
Quince Therapeutics
Page
2
by the encouraging Phase 3 data generated from EryDel’s prior international study of EryDex, which demonstrated a significant delay in disease progression in
A-T patients and further supported more than 10 years of safety data. Quince is well-positioned to advance EryDel’s differentiated AIDE technology and development of our lead asset EryDex to deliver
innovative treatments to patients in need.”
Transformative Acquisition with Value-Creating Clinical Milestones
Key highlights of the EryDel acquisition include:
Well-capitalized into 2026 with ability to fully fund lead asset EryDex expected through Phase 3 clinical trial under SPA and to NDA submission
| |
• |
|
Strong balance sheet with approximately $87.6 million in cash, cash equivalents, and short term investments
(unaudited) as of June 30, 2023, to provide funding for operating requirements into 2026. |
| |
• |
|
Capital efficient development plan allows for funding of EryDex through global Phase 3 clinical trial under SPA
and, assuming positive study results, to NDA submission, in addition to pursuing European regulatory activities related to potential MAA submission. |
| |
• |
|
Potential to out-license ex-U.S.
regional territories to provide runway through regulatory approval of EryDex. |
Plan to enroll first patient in global Phase 3 trial
of EryDex in second quarter of 2024 with NDA submission targeted by end of 2025
| |
• |
|
SPA in place with FDA for a single global Phase 3 clinical trial of EryDex expected to be sufficient for NDA
submission, assuming positive study results. |
| |
• |
|
EryDex designated as orphan drug for treatment of A-T from both the FDA
and EMA. |
| |
• |
|
Phase 3 NEAT (Neurologic Effects of EryDex on Subjects with
A-T) clinical trial is a planned double blind, randomized, placebo controlled, global efficacy study in approximately 86 A-T patients aged six to nine years-old with up to an additional 20 patients aged 10 years or older included for potential broader label support. |
| |
• |
|
Primary endpoint, as agreed upon with the FDA, to measure neurological function based on rescored modified
International Cooperative Ataxia Rating Scale (RmICARS) from baseline to month six of treatment. |
| |
• |
|
Secondary endpoints to measure Clinical Global Impression scores for severity
(CGI-S) and change (CGI-C), as well as EuroQol quality of life scoring. |
| |
• |
|
Plan to enroll first patient in Phase 3 NEAT clinical trial in the second quarter of 2024. |
| |
• |
|
Commercial version of EryKit treatment consumables approved in Europe and currently under partial clinical hold
pending response to FDA query. |
| |
• |
|
Target EryDex NDA submission with the FDA by the end of 2025, assuming positive Phase 3 NEAT study results.
|
EryDex efficacy and safety profile demonstrated in prior Phase 3 clinical trial of A-T
patients
| |
• |
|
Pursuing European regulatory activities related to potential MAA submission of EryDex based on prior Phase 3
clinical trial. |
| |
• |
|
Completed largest global interventional study of A-T patients (N=175) in
Phase 3 ATTeST (Ataxia Telangiectasia Trial with the EryDex SysTem) clinical trial and open label extension (OLE) (N=104). |
Quince Therapeutics
Page
3
| |
• |
|
Primary endpoint measured modified International Cooperative Ataxia Rating Scale (mICARS) score from baseline to
month six of treatment. |
| |
• |
|
Secondary endpoints measured CGI-C, Quality of Life (QOL), and Vineland
Adaptive Behavior Scales (VABS) scores. |
| |
• |
|
EryDex high dose treatment arm demonstrated slowed neurological deterioration in
A-T disease progression as measured by mICARS in intent to treat population (ITT) with statistically significant effect in six to nine year-old subgroup across multiple
endpoints. |
| |
• |
|
12-month safety analysis demonstrated EryDex well-tolerated with no
major adverse events typically associated with chronic steroid administration. |
| |
• |
|
Sustained therapeutic effect and favorable safety profile maintained for more than three additional years in high
dose treatment arm in OLE study, in addition to no steroid related toxicity observed in patients receiving more than 10 years of treatment. |
| |
• |
|
Conformité Européene (CE) mark already obtained in Europe for drug/device combination and
commercial version of EryKit treatment consumables. |
$1+ billion estimated peak global sales opportunity for A-T indication alone with rapid expansion potential for EryDex to other rare and debilitating diseases
| |
• |
|
A-T population estimated to be approximately 10,000 patients in the U.S.,
U.K., and EU4 countries with no currently approved therapies and $1+ billion estimated peak sales opportunity globally. |
| |
• |
|
EryDex for A-T indication holds potential to be first-to-market with attractive pricing comparables and no known late-stage competition. |
| |
• |
|
EryDex designated as orphan drug for A-T treatment from the FDA and EMA.
|
| |
• |
|
Potential for rapid expansion of EryDex to other rare and debilitating disease indications where chronic steroid
administration is the standard of care – or could be in the absence of long-term steroid toxicity. |
| |
• |
|
AIDE platform capable of expansion to other drugs or biologics, including enzyme replacement therapy.
|
| |
• |
|
Multi-faceted technology protections create high barriers to entry with intellectual property exclusivity until
at least 2034 globally and at least 2035 in the U.S. |
Transaction Details
Under the terms of the acquisition transaction, EryDel will operate as a wholly owned subsidiary of Quince Therapeutics with plans to retain EryDel’s
corporate and manufacturing presence in Italy. The integrated company will be led by Dirk Thye, M.D., Quince’s Chief Executive Officer and member of the Quince Board of Directors. In addition, David Lamond remains Chairperson of Quince’s
Board of Directors, which will be expanded by one member with the addition of EryDel representative Luca Benatti following the close of the transaction.
Upon completion of the stock-for-stock upfront exchange, EryDel stockholders
will own a maximum of approximately 16.7%, or 7,250,352 shares, of the combined company (subject to downward adjustment). The transaction agreement includes up to $485 million in potential total downstream cash payments, including up to
$5 million in development milestones, $25 million at NDA acceptance, $60 million in approval milestones, and $395 million in market and sales milestones, with no royalties paid to EryDel stockholders. The transaction will include
the assumption of EryDel’s $13 million (€10 million in principal) European Investment Bank (EIB) loan with scheduled payments beginning in the second half of 2026.
Quince Therapeutics
Page
4
The transaction, which has been unanimously approved by the Boards of Directors of both companies, is subject
to certain regulatory approvals and other closing conditions and is expected to close in the third quarter of 2023.
Financial Statements
Quince has not completed preparation of its financial statements for the second quarter of 2023. The cash, cash equivalents, and short term investments
presented as of June 30, 2023, are preliminary and unaudited and are, thus, inherently uncertain and subject to change. The company is in the process of completing its customary close and review procedures for the second quarter of 2023, and
there can be no assurance that final results for this period will not differ from these preliminary, unaudited amounts. The company’s independent registered public accounting firm has not audited, reviewed, compiled, or performed any procedures
with respect to such preliminary data for the second quarter ended June 30, 2023.
Advisors
MTS Health Partners, L.P. is serving as financial advisor and Cooley LLP is serving as legal counsel to Quince. Perella Weinberg Partners is serving as
financial advisor and Goodwin Procter LLP and Clifford Chance LLP are serving as legal counsel to EryDel.
Investor Presentation Available
To learn more about the transaction, investors are encouraged to access an investor presentation provided by Quince management detailing the EryDel
acquisition, which is currently available for viewing on the company’s Investor Relations website. Please visit the Events page under the News & Events heading of Quince’s Investor Relations website at ir.quincetx.com to
access the presentation.
About Quince Therapeutics
Quince Therapeutics is a biotechnology company focused on acquiring, developing, and commercializing innovative therapeutics that transform the lives of
patients suffering from debilitating and rare diseases. For more information, visit www.quincetx.com and follow Quince Therapeutics on LinkedIn and @Quince_Tx on Twitter.
About EryDel SpA
EryDel SpA is a global late-stage
biotech company aimed at developing and commercializing therapies for the treatment of rare diseases delivered by its proprietary red blood cell technology. Its most advanced product, EryDex, is under late-stage development for the treatment of
Ataxia Telangiectasia (A-T), a rare autosomal recessive neurological disorder for which no established therapy is currently available. EryDex is an automated outpatient bedside technology to ex-vivo encapsulate dexamethasone sodium phosphate (DSP; a pro-drug) into patient’s red blood cells, which are then re-infused,
allowing for the circulation of controlled, slow release, low doses of dexamethasone (active drug) over the subsequent several weeks following treatment. EryDex has received orphan drug designation for the treatment of
A-T both from the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). An international multicenter, Phase 3 clinical trial, ATTeST, and its open label extension have been
successfully completed. In addition to EryDex, EryDel’s technology platform is capable of expansion to other drugs or biologics, including enzyme replacement therapy, and has the potential to support a wide range of therapeutic opportunities.
Quince Therapeutics
Page
5
Forward-looking Statements
Statements in this news release contain “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995 as
contained in Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, which are subject to the “safe harbor” created by those sections. All statements, other than
statements of historical facts, may be forward-looking statements. Forward-looking statements contained in this news release may be identified by the use of words such as “believe,” “may,” “should,” “expect,”
“anticipate,” “plan,” “believe,” “estimated,” “potential,” “intend,” “will,” “can,” “seek,” or other similar words. Examples of forward-looking statements
include, among others, statements relating to Quince’s acquisition of EryDel; the timing of the closing of the transaction; the expected benefits of the transaction, including the continued current and future clinical development and potential
expansion of EryDel assets, related platform, and related timing and costs; the strategic development path for EryDex; planned FDA and EMA submissions and clinical trials and timeline, prospects, and milestone expectations; the timing and success of
the clinical trials and related data, including plans and the ability to initiate, fund, conduct and/or complete current and additional studies; the potential therapeutic benefits, safety, and efficacy of EryDex; statements about its ability to
obtain, and the timing relating to, further development of EryDex, regulatory submissions and interactions with regulators; therapeutic and commercial potential; the integration of EryDel’s business, operations, and employees into Quince;
Quince’s future development plans and related timing; its cash position and projected cash runway; the company’s focus, objectives, plans, and strategies; and the ability to execute on any strategic transactions. Forward-looking statements
are based on Quince’s current expectations and are subject to inherent uncertainties, risks, and assumptions that are difficult to predict and could cause actual results to differ materially from what the company expects. Further, certain
forward-looking statements are based on assumptions as to future events that may not prove to be accurate. Factors that could cause actual results to differ include, but are not limited to, the risks and uncertainties described in the section titled
“Risk Factors” in the company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission (SEC) on May 15, 2023, and other reports as filed with the SEC.
Forward-looking statements contained in this news release are made as of this date, and Quince undertakes no duty to update such information except as required under applicable law.
|
|
|
| Quince Therapeutics Contact:
Stacy Roughan
Quince Therapeutics, Inc.
Vice President, Corporate Communications &
Investor Relations ir@quincetx.com |
|
Media:
Dan Gagnier & Riyaz Lalani
Gagnier Communications
quinceGFC@gagnierfc.com |
v3.23.2
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=us-gaap_CommonStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=us-gaap_SeriesAPreferredStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
Quince Therapeutics (NASDAQ:QNCX)
Historical Stock Chart
From Jun 2024 to Jul 2024
Quince Therapeutics (NASDAQ:QNCX)
Historical Stock Chart
From Jul 2023 to Jul 2024