UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
|
þ
|
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15 (d) OF THE SECURITIES
EXCHANGE ACT OF 1934
|
For the fiscal year ended March 31, 2012
|
¨
|
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15 (d) OF THE SECURITIES EXCHANGE ACT OF 1934
|
Commission File Number 001-15975
REMEDENT, INC.
(Name of small business issuer as specified
in its charter)
|
Nevada
|
|
86-0837251
|
|
(State or other jurisdiction of
|
|
(I.R.S. Employer
|
|
incorporation or organization)
|
|
Identification Number)
|
|
|
|
|
|
Zuiderlaan 1-3, bus 8, 9000 Gent, Belgium
|
|
N/A
|
|
(Address of principal executive offices)
|
|
(Zip code)
|
011-329-241-58-80
(Issuer’s telephone number, including
area code)
Securities registered pursuant to Section
12(b) of the Act:
None
Securities registered pursuant to Section
12(g) of the Act:
Common Stock, no par value per share
(Title of Class)
Indicate by check mark if the registrant is a well-known seasoned
issuer, as defined in Rule 405 of the Securities Act.
Yes
¨
No
þ
Indicate by check mark if the registrant is not required to
file reports pursuant to Section 13 or Section 15(d) of the Act.
Yes
¨
No
þ
Indicate by check mark whether the registrant (1) has filed
all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months
(or for such shorter period that the issuer was required to file such reports), and (2) has been subject to such filing requirements
for the past 90 days.
Yes
þ
No
¨
Indicate by check mark whether the registrant has submitted
electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant
to Rule 405 of Regulation S-T ( § 232.405 of this chapter) during the preceding 12 months (or for such shorter period that
the registrant was required to submit and post such files). Yes
þ
No
¨
Indicate by check mark if disclosure of delinquent filers pursuant
to Item 405 of Regulation S-K (§ 229.405) is not contained herein, and will not be contained, to the best of registrant’s
knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment
to this Form 10-K.
¨
Indicate by check mark whether the registrant is a large accelerated
filer, an accelerated filer, a non-accelerated filer or a smaller reporting company. See the definitions of “large
accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange
Act. (Check one):
|
|
Large accelerated filer
|
¨
|
|
Accelerated filer
|
¨
|
|
|
Non-accelerated filer
|
¨
|
|
Smaller reporting company
|
þ
|
|
|
(Do not check if a smaller reporting company)
|
|
|
|
Indicate by check mark whether the registrant is a shell company
(as defined in Rule 12b-2 of the Exchange Act).
Yes
¨
No
þ
The aggregate market value of voting stock held by non-affiliates
computed by reference to the price at which the common equity was last sold as of the last business day of the registrant’s
most recently completed second fiscal quarter, September 30, 2011, was approximately $2,938,238.24. For purposes of
this computation, it has been assumed that the shares beneficially held by directors and officers of registrant were “held
by affiliates” and this assumption is not to be deemed to be an admission by such persons that they are affiliates of registrant.
The number of shares of registrant’s common stock outstanding
as of June 25, 2012 was 19,995,969.
Documents incorporated by reference:
None.
Transitional Small Business Disclosure
Format
(Check one): Yes
¨
No
þ
FORM 10-K INDEX
|
PART I
|
|
ITEM 1.
|
BUSINESS
|
|
1
|
|
ITEM 1A.
|
RISK FACTORS
|
|
14
|
|
ITEM 1B.
|
UNRESOLVED STAFF COMMENTS
|
|
24
|
|
ITEM 2.
|
PROPERTIES
|
|
25
|
|
ITEM 3.
|
LEGAL PROCEEDINGS
|
|
25
|
|
ITEM 4.
|
[RESERVED AND REMOVED]
|
|
25
|
|
PART II
|
|
ITEM 5.
|
MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
|
|
25
|
|
ITEM 6.
|
SELECTED FINANCIAL DATA
|
|
27
|
|
ITEM 7.
|
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
|
|
27
|
|
ITEM 7A.
|
QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
|
|
40
|
|
ITEM 8.
|
FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
|
|
40
|
|
ITEM 9.
|
CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
|
|
40
|
|
ITEM 9A
|
CONTROLS AND PROCEDURES.
|
|
40
|
|
ITEM 9B.
|
OTHER INFORMATION
|
|
41
|
|
PART III
|
|
ITEM 10.
|
DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
|
|
41
|
|
ITEM 11.
|
EXECUTIVE COMPENSATION
|
|
45
|
|
ITEM 12.
|
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
|
|
46
|
|
ITEM 13.
|
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS AND DIRECTOR INDEPENDENCE
|
|
48
|
|
ITEM 14.
|
PRINCIPAL ACCOUNTANT FEES AND SERVICES
|
|
50
|
|
PART IV
|
|
ITEM 15.
|
EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
|
|
51
|
|
SIGNATURES
|
|
|
52
|
Forward-Looking Statement
In addition to historical information, this Annual Report
on Form 10-K
(“Annual Report”) for Remedent, Inc. (“Remedent” the “Company,” “we,”
“our” or
“us”) contains “forward-looking” statements within the meaning of Section
21E of the Securities
Exchange Act of 1934, as amended (the “Exchange Act”), including statements
regarding
the growth of product lines, optimism regarding the business,
expanding sales and other statements. Words such as expects,
anticipates,
intends, plans, believes, sees, estimates and variations of such words and
similar expressions are intended
to identify such forward-looking statements.
These statements are not guarantees of future performance and involve certain
risks and uncertainties that are difficult to predict. Actual results could
vary materially from the description contained
herein due to many factors
including continued market acceptance of our products. In addition, actual
results could
vary materially based on changes or slower growth in the oral
care and cosmetic dentistry products market; the potential
inability to realize
expected benefits and synergies; domestic and international business and
economic conditions;
changes in the dental industry; unexpected difficulties in
penetrating the oral care and cosmetic dentistry products market;
changes in
customer demand or ordering patterns; changes in the competitive environment
including pricing pressures
or technological changes; technological advances;
shortages of manufacturing capacity; future production variables impacting
excess inventory and other risk factors listed in the section of this Annual
Report entitled “Risk Factors”
and from time to time in our Securities and
Exchange Commission filings under “risk factors” and elsewhere.
Each forward-looking statement should
be read in context with, and with an
understanding of, the various disclosures concerning our business made
elsewhere
in this Annual Report, as well as other public reports filed by us
with the United States Securities and Exchange Commission.
Readers should not
place undue reliance on any forward-looking statement as a prediction of actual
results of developments.
Except as required by applicable law or regulation, we
undertake no obligation to update or revise any forward-looking statement
contained in this Annual Report.
PART I
ITEM 1 — BUSINESS
Overview
We specialize in the research, development,
and manufacturing of oral care and cosmetic dentistry products. We are one of the leading manufacturers of cosmetic
dentistry products in Europe. Leveraging our knowledge of regulatory requirements regarding dental products and management’s
experience in the needs of the professional dental community, we design, develop, manufacture and distribute our cosmetic dentistry
products, including a full line of professional dental products that are distributed in Europe, Asia and the United States.
In 2006 we developed a revolutionary system
for manufacturing and installing dental veneers which we branded as GlamSmile ® veneers revolutionize the traditional one-at-a-time
method of applying porcelain dental veneers. GlamSmile veneers are attached to the front of the patient’s teeth
using a patent pending single motion placement tray which replaces the traditional one at a time trial and error method of applying
porcelain veneers, making the application less traumatic for the patient, much easier for the dentist and perhaps most important,
far less costly than traditional dental veneers. Currently, the GlamSmile veneers are our primary products in the professional
oral care and cosmetic dentistry product. Our veneers are supported by a line of professional veneer whitening
and teeth sensitivity solutions. Our products are sold to professionals by distributors and sold directly to consumers
by our GlamSmile Studios.
Corporate History
We were originally incorporated under the
laws of Arizona in September 1996 under the name Remedent USA, Inc. In October 1998, we were acquired by Resort World
Enterprises, Inc., a Nevada corporation in a share exchange, and we immediately changed our name to Remedent USA, Inc. and later
to Remedent, Inc.
In the latter part of 2008, our Board of
Directors approved a strategic plan to separate our OTC business from our professional business, allowing us to focus on the development,
marketing and distribution of our products for the professional dental market. In December 2008, we completed a restructuring
in the form of a management-led buyout of 50% of our OTC retail business (“2008 Restructuring”). The buyout
was led by Mr. Robin List, our former director and Chief Executive Officer, with financing provided by a non-affiliated foreign
investment fund. In connection with the strategic plan, we effected our OTC restructuring through a series of transactions
involving subsidiary formations, contributions of subsidiary(ies) interests and sales of stock interests through subsidiary transactions. As
a result of the series of transactions related to the sale, Remedent Inc. currently owns 50% of Remedent OTC BV,
a Dutch company (“Remedent OTC”) with Mr. List owning the other 50%. Remedent OTC owns and holds a 75% ownership interest
in Sylphar Holding BV, a Dutch holding company which owns and holds the OTC operating subsidiaries (“Sylphar
Holding”), together with Concordia Fund B.V. who owns 25% of Sylphar Holding. As a result of Remedent, Inc.’s ownership
interest in Remedent OTC, Remedent, Inc. holds an ownership interest equal to 37.5% interest in Sylphar Holding. As
a result of the sale, all of the OTC business previously directly operated by us is now operated and held by Sylphar Holding. In
addition, following the restructuring we have focused our business primarily on the marketing and distribution of our GlamSmile
Veneers.
In January, 2010 we formed a joint venture
with Gallant Network Limited (“Gallant”) to formalize our GlamSmile operations in China. In order
to sell our products into the Chinese market, an approval by Chinese authorities is required, in the form of licenses. Since GlamSmile
Asia Ltd., a private Hong Kong company (“GlamSmile Asia”), was already the owner of such licenses prior to the acquisition,
this was an important advantage. We obtained control of GlamSmile Asia through the acquisition of a 50.98% interest
in exchange for (i) 325,000 Euro (US $466,725), of which 50,000 Euro was payable as of March 31, 2010, (ii) 250,000 shares
of common stock was originally agreed to be issued during the fiscal year ended March 31, 2011 and then subsequently amended to
be issued during fiscal year ended March 31, 2013, and (iii) option grants of up to 300,000 shares of our common stock,
of which 200,000 shares were granted as of March 31, 2011.
On January 28, 2012, we entered into a Preference
A Shares and Preference A-1 Shares Purchase Agreement (“Share Purchase Agreement”) with Glamsmile Dental Technology
Ltd., a Cayman Islands company and a subsidiary of Company (“Glamsmile Dental”), Glamsmile (Asia) Limited, a company
organized and existing under the laws of Hong Kong and a substantially owned subsidiary of Glamsmile Dental, Beijing Glamsmile
Technology Development Ltd., Beijing Glamsmile Trading Co., Ltd., Beijing Glamsmile Dental Clinic Co., Ltd., and Shanghai Glamsmile
Dental Clinic Co., Ltd., Gallant Network Limited, a shareholder of Glamsmile Dental (“Gallant”), and IDG-Accel China
Growth Fund III L.P. (“IDG Growth”), IDG-Accel China III Investors L.P.(“IDG Investors”) and Crown Link
Group Limited (“Crown”)(“IDG Growth, IDG Investors and Crown collectively referred to as the “Investors”),
pursuant to which the Investors agreed to (i) purchase from the Company an aggregate of 2,857,143 shares of Preference A-1 Shares
of Glamsmile Dental, which represents all of the issued and outstanding Preference A-1 Shares of Glamsmile Dental, for an aggregate
purchase price of $2,000,000, and (ii) purchase from Glamsmile Dental an aggregate of 5,000,000 shares of Preference A Shares for
an aggregate purchase price of $5,000,000.
On February 10, 2012, the sale of the Preference
A-1 Shares and the Preference A Shares was completed. As a result of the closing, the equity ownership of Glamsmile Dental, on
an as converted basis, is as follows: 31.4% by the Investors, 39.2 % by Gallant, and 29.4% by the Company. Mr. De Vreese, our chairman,
will remain as a director of Glamsmile Dental along with Mr. David Lok, who is the Chief Executive Officer and director of Glamsmile
Dental and principal of Gallant. In addition, at the closing, the Investors have a right to appoint one director of Glamsmile Dental,
and as such it is contemplated that after the closing the Board of Directors of Glamsmile Dental will consists of Mr. De Vreese,
Mr. Lok and a director appointed by the Investors.
Under the terms of the Share Purchase Agreement,
we agreed to transfer 500,000 shares of Glamsmile Dental owned by the Company to the Investors in the event of breach of certain
covenants by the Company. In connection with the Share Purchase Agreement, we also entered into an Investor’s Rights Agreement,
Right of First Refusal and Co-Sale Agreement, and Voting Agreement with the parties. In addition, in connection with the contemplated
transactions in the Share Purchase Agreement on January 20, 2012, we entered into a Distribution, License and Manufacturing Agreement
with Glamsmile Dental pursuant to which we appointed Glamsmile Dental as the exclusive distributor and licensee of Glamsmile Veneer
Products bearing the “Glamsmile” name and mark in the B2C Market in the People’s Republic of China (including
Hong Kong and Macau) and Republic of China (Taiwan) and granted related manufacturing rights and licenses in exchange for the original
issuance of 2,857,143 shares of Preference A-1 Shares of Glamsmile Dental and $250,000 (the receipt of which was acknowledged as
an off set to payment of certain invoices of Glamsmile (Asia) Limited).
We have the following wholly owned subsidiaries: (1)
Remedent N.V., a Belgium corporation; (2) Remedent Professional Holdings, Inc., a California corporation; (3) Remedent Professional,
Inc., a California corporation (a subsidiary of Remedent Professional Holdings, Inc.), and (4) Glamtech-USA, Inc., a Delaware corporation. In
addition, Remedent N.V., our wholly-owned subsidiary, has (A) a 51% ownership interest in GlamSmile Deutschland GmbH,
a German private company, (B) a 100% ownership interest in GlamSmile Rome SRL (an Italian private Company). Remedent Inc. also
has a 50 % ownership in Remedent OTC BV (a Dutch company which holds a 75% ownership interest in Sylphar Holding)
and (D) a 37.5% ownership interest in Sylphar Holding (a Dutch holding company which wholly owns: Sylphar Holding: (i) Sylphar
N.V., a Belgium corporation; (ii) Sylphar USA, Inc., a Nevada corporation; and (iii) Sylphar Asia Pte Ltd. (formerly
Remedent Asia Pte Ltd.), a Singapore company. In addition, Remedent, Inc. owns a 29.4% ownership interest in Glamsmile Dental Technology
Ltd., a Cayman Islands company (“Glamsmile Dental”) and its subsidiaries, Glamsmile (Asia) Limited, a company organized
and existing under the laws of Hong Kong and a substantially 100 % owned subsidiary of Glamsmile Dental, Beijing Glamsmile Technology
Development Ltd., a 100 % owned subsidiary or GlamSmile Asia, its 80% owned subsidiary Beijing Glamsmile Trading Co., Ltd. and
its 98% owned subsidiary Beijing Glamsmile Dental Clinic Co., Ltd., including its 100% Shanghai Glamsmile Dental Clinic Co., Ltd.,
and its 50 % owned Whenzhou GlamSmile Dental Clinic Ltd.
Past Product Development
We have been a manufacturer and distributor
of cosmetic dentistry products, including a full line of professional dental and retail OTC tooth whitening products which are
distributed in Europe, Asia and the United States. We have distributed our products using both our own internal sales
force and through the use of third party distributors. Prior to our 2008 Restructuring, our products were generally
classified into the following categories: professional dental products and OTC tooth whitening products. Our OTC division included
products targeted for retail such as iWhite, Cleverwhite and Remesense. However as a result of the 2008 Restructuring
and sale, all of our prior OTC operations, including the marketing and distribution of the OTC products are being conducted
by Sylphar Holding and its wholly owned subsidiaries. We developed the following products for our professional dental
segment:
|
|
·
|
Remewhite in Office Whitening System
. One of our first
dental products that we developed for the professional dental community was the RemeCure™ plasma curing light (described
below). Leveraging on our early success with the RemeCure light, we introduced the RemeWhite™ In Office Whitening
System. Based upon the initial RemeCure light, a new light, called the RemeCure CL-15, was developed featuring new enhancements
to the hardware and software enabling this light to be fully automated thereby eliminating the need for the dentist to hold the
light during whitening treatments. In addition, a proprietary gel was formulated to be used with the system as well
as a time saving method to apply the gel.
|
|
|
·
|
Remewhite Home Maintenance Kit
. In 2004,
the RemeWhite Home Maintenance Kit was introduced and sold by dentists to their patients, featuring 16 pre-filled trays with a
level of whitening agent safe for home use yet stronger than most OTC products.
|
|
|
·
|
Metatray
. In August 2005, we introduced MetaTray®,
our next generation of products targeted for the professional dentist market. The MetaTray kit consists of a proprietary,
reusable mouthpiece that has embedded in the mouthpiece both a heating element and an electroluminescent mesh that are powered
by a rechargeable 9 volt power source providing heat and light similar to that which is delivered to the teeth by conventional
dental lights. The system also introduced a proprietary foam strip that is unique in the manner in which it releases peroxide to
the tooth surface without dripping or running.
|
|
|
·
|
RemeCure.
The RemeCure plasma curing light uses plasma
arc technology instead of LED and laser technology which provides high-energy power over the complete spectrum.
|
|
|
·
|
FirstFit.
We developed the FirstFit™ System, a proprietary, patent-pending system for the creation and placement of dental bridges and crowns. Effective as of March 29, 2010 the intellectual property used and related to FirstFit product was sold to Den-Mat Holdings LLC (“Den-Mat”).
|
Current Business Strategy
Our current business strategy
is to focus our resources on vertically integrating our development, manufacturing and marketing resources on selling our GlamSmile
veneers directly to consumers by using all forms of direct response media including the internet, print, radio, television and
social network media, to expand our presence in Asia, with a primary focus on China, and Europe, as well as to establish
a direct to consumer presence in United States.
Current Products
We currently have two primary products:
GlamSmile Veneers, and Whitening Products. In addition, we are currently developing a ‘prefab’ veneer which would be
a ‘ready-to-use’ veneer, called ‘River 8’ which we anticipate we will offer at a lower price than traditional
veneers. It is anticipated that the launch will be during the first six months of the next fiscal year.
Whitening Products
We sell our whitening products directly
throughout the world. We continue to focus our sales efforts on our whitening products.
We initially started in the whitening business,
by developing and marketing whitening products in combination with the worldwide appreciated high speed curing lamp RemeCure. The
RemeCure plasma curing light uses plasma arc technology instead of LED and laser technology which provides high-energy power over
the complete spectrum. This allows RemeCure plasma curing light to be used in various applications such as: (1) curing
dental composite materials in only seconds; and (2) for single appointment, in-office whitening in less than forty minutes.
|
|
·
|
Remewhite Formulation+
by GlamSmile.
Formulation+
was the first available in-office power whitening gel featuring Remedent’s proprietary CRM-Technology.
The Continuous Release Matrix assures a prolonged and continuous release of Hydrogen Peroxide throughout the entire session, thus
improving the exposure time of the whitening agent to the enamel. Still today Formulation+ for in-office use by dentists is one
of the top products giving consumers whitening results up to 10 shades whiter without sensitivity.
|
|
|
·
|
WHITE Boost & WHITE Finishing
. Other than Formulation+, Remedent created and sells the White Boost & White Finishing through dentists for home use. It is an oxygen-induced whitening system using a revolutionary and comfortable tray and 2 thin, flexible foam strips impregnated with super tooth whitening gel. The foam strips’ unique formula guarantees a steady release of oxygen that first triggers and then speeds up the whitening process. Our ergonomic delivery tray means ease-of-use and superb whitening results in just 10 minutes.
|
GlamSmile Veneers
In connection with the 2008 Restructuring,
we shifted our focus to professional products targeted for the professional sector. Our key product in the professional oral care
and cosmetic dentistry product is the GlamSmile veneer.
In 2006 we
developed a revolutionary system for manufacturing and installing dental veneers which we branded as GlamSmile. GlamSmile veneers
revolutionize the traditional one-at-a-time method of applying porcelain dental veneers. GlamSmile veneers are attached
to the front of the patient’s teeth using a patent pending single motion placement tray which replaces the traditional one
at a time trial and error method of applying porcelain veneers, making the application less traumatic for the patient, much easier
for the dentist and perhaps most important, far less costly than traditional dental veneers. The entire process is painless and
takes only about an hour of the patient’s and the dentist’s time. GlamSmile veneers are so thin that the dentist does
not need to remove healthy tooth structure which results in a process that is reversible.
In
the fall of 2006, we opened our initial GlamSmile Lab in Ghent.
Our GlamSmile involves a proprietary veneer
fabrication technique and a patented single-motion veneer placement tray which are both guided by a proprietary computer imaging,
design and digital preview system. The unique tray delivery system lets dentists expertly seat 10 ultra-thin, custom veneers in
less than an hour while preserving tooth structure. All the features of GlamSmile, together with the CAD/CAM technology, digital
preview for dentists to evaluate the design and a unique full arch tray delivery system used in conjunction with minimally or no
preparation ultra thin veneers, have revolutionized the art of veneering.
Our GlamSmile veneers are ultra thin claddings
made from a mixture of a hybrid composite and porcelain materials which are attached to the front of the patient’s teeth. GlamSmile
veneers are ultra-thin and can best be compared to contact lenses in terms of thickness. Because GlamSmile veneers are so thin,
the dentist does not need to remove healthy tooth structure, Leaving the patient’s healthy tooth structure intact
results in several important benefits:
|
|
•
|
no local anesthesia is required to prepare the teeth;
|
|
|
•
|
reduced (if any) tooth sensitivity post-procedure; and
|
|
|
•
|
the process is reversible.
|
Our veneers are custom-made for each individual’s
personal features, taking into account numerous factors including the shape of a person’s face, the shape of their lips and
more. At the initial doctor visit, an impression is made of the patient’s teeth. During the second visit, the
hybrid composite veneers, which are computer generated as a single unit, are then ready to be installed. The single-unit
feature enables dentists with minimal training to apply up to ten teeth in one 30 – 45 minute visit. This minimizes
the risk of failure and allows more dentists to offer GlamSmile veneers as part of their dental practice. With traditional bonding,
a dentist adheres a composite material directly on the tooth which lasts about 3 to 6 years and tends to discolor. Porcelain
veneers, though a more lasting solution (ten years or more), require a significantly more invasive procedure to install, which
is irreversible, requires a very high level of training and skill from the dentist and can cost from $700 to $2,000 per tooth.
Marketing and Distribution
We market our products to the dental professional
using our business to business strategies (“B2B”), and we also market our products directly to the consumers in Asia,
Europe and the Middle East using our direct to consumer model (“B2C”). Our products are sold
to dental professionals in 28 countries through distributors. We previously sold our GlamSmile product in the United States
and throughout the world with the exception of certain excluded territories and certain B2C markets pursuant to a distribution
agreement. However, on March 27, 2012, the distribution agreements with Den-Mat were terminated pursuant to a certain Termination
and Distribution Agreement with Den-Mat (“Den-Mat Distribution Agreement”). Pursuant to the Den-Mat Distribution Agreement,
we granted Den-Mat a non-exclusive, irrevocable, perpetual, royalty free, license to use within certain territory, which among
other territories excludes China, Macau, Hong Kong, and Taiwan, the intellectual property that was the subject of the license to
Den-Mat under the Amended and Restated Distribution, License and Manufacturing Agreement dated June 3, 2009, as amended from time
to time (“Prior Agreements”), as such intellectual property relates the products which was the subject of the Prior
Agreements. In connection with the termination of the Prior Agreements, under the Den-Mat Distribution Agreement, Den-Mat agreed
to pay us $200,000. We currently sell our products in Asia, Europe and the Middle East directly to consumers using our
direct to consumer model, which includes our GlamSmile Smile Design-Virtual Studio, and GlamSmile Studios.
B2C Market and Distribution
In 2008 we opened, through a third party,
our first GlamSmile center in Beijing China, marketing GlamSmile directly to consumers. In 2009 we began direct to consumer
tests in Belgium using internet advertising to acquire potential leads and our own dedicated “Smile Consultants” to
manage the sales process from lead acquisition through final sale with successful results. Our direct to consumer model, has been
developed around a one to one relationship with our Smile Consultants. This process also results in the dentists being relieved
of the sales responsibilities allowing them to better focus on patient satisfaction. In both China and Belgium, with the aid of
our own “Smile Consultant” working direct with the customer throughout the entire sales process, we have seen positive
results in our partner retail centers.
Our Smile Consultancy Program is predominantly
marketed on the internet through our website, GlamSmile Smile Design. We focus on intensive campaign and advertisement
aimed to generate large traffic to our website that promotes GlamSmile Whitening, Veneers and Free Smile Advice. Visitors can apply
for a free personalized Smile Consultation by a Smile Consultant. The latter guides the consumer to the right GlamSmile Studio
or with one of our GlamSmile partner dentists and to the solution that meets best his or her Smile expectations. The
Smile Consultancy Program requires us to develop close partnerships with dedicated GlamSmile dentists and the establishment of
GlamSmile Studios. The GlamSmile Studio is a concept studio with a focus on aesthetic and cosmetic dentistry. Unlike a traditional
dentist office, our GlamSmile Studio are designed and managed as a dental spa.
We have begun to market our products through
our Smile Consultancy Concept and through the establishment of our GlamSmile Studios in Asia, with a primary focus on China, and
Europe. We currently have an ownership interest in the following Glamsmile Studios:
|
|
·
|
Beijing Glamsmile Studio
. Through Glamsmile Asia and its subsidiaries we opened a GlamSmile clinic in Beijing, China, during the third calendar quarter of 2009. The Beijing GlamSmile clinic was the first dental spa to offer pain free cosmetic dentistry in Beijing.
|
|
|
|
|
|
|
·
|
Hong Kong Dental Spa
. In April 2010, through GlamSmile Asia Ltd. we expanded our business to consumer model in the Asian market by opening a dental spa in Hong Kong.
|
|
|
|
|
|
|
·
|
Munich, Germany Glamsmile Studio
. In October 2010, through GlamSmile Deutschland GmbH a studio in Munich, Germany was opened.
|
|
|
|
|
|
|
·
|
Shanghai Studio
. In February 2011, we opened the Shanghai Studio through GlamSmile Asia.
|
|
|
|
|
|
|
·
|
Rome, Italian Glamsmile Studio
. In June 2011, through GlamSmile Rome SRL a studio in Rome, Italy was opened.
|
|
|
|
|
|
|
·
|
Wenzhou Studio
. In February 2012, we opened the Wenzhou Studio through GlamSmile Asia.
|
In connection with the contemplated transactions
in the Share Purchase Agreement on January 20, 2012, we entered into a Distribution, License and Manufacturing Agreement with Glamsmile
Dental pursuant to which we appointed Glamsmile Dental as the exclusive distributor and licensee of Glamsmile Veneer Products bearing
the “Glamsmile” name and mark in the B2C Market in the People’s Republic of China (including Hong Kong and Macau)
and Republic of China (Taiwan) and granted related manufacturing rights and licenses in exchange for the original issuance of 2,857,143
shares of Preference A-1 Shares of Glamsmile Dental and $250,000 (the receipt of which was acknowledged as an off set to payment
of certain invoices of Glamsmile (Asia) Limited).
Cosmetic Dentistry Industry
The cosmetic dental industry has expanded
into a multi-billion industry as a result of increased awareness of the importance of oral health, high aesthetics, improved dental
treatments, and reduced patient discomfort. An increasingly aging population and rising disposable income have also
positively impacted the growth of cosmetic dentistry. Demand for dental products and services are forecasted to remain healthy
due to growing incidences of cosmetic treatments and dental implants. According to a report published by
Koncepts Analytics, the dental industry worldwide was estimated at about $18.8 billion in 2008, dominated by the US, Europe and
Japan, which collectively accounted for more than 84% of the global revenue in 2007. The United States Dental Market was nearly
$7.6 billion in 2007, projected to grow to almost $8 billion in 2008 and nearly $10 billion by 2013, representing a compound annual
growth rate of 4.7%. The American Academy of Cosmetic Dentistry estimates that Americans spend about $2.75 billion each year on
cosmetic dentistry. The growth of this market is expected to be highest in the United States and EU where the generation of aging
baby boomers can afford these quality but expensive dental procedures. Also expected to be a catalyst for the growth and popularity
of cosmetic treatments and implants is the younger generation. Further, emerging technologies will reduce the overall turnaround
time for dental procedures while improving efficiency of the dental practitioners. For example, introduction of CAD/CAM has reduced
designing time and 3D imaging techniques have improved patient diagnosis and procedure planning. Changing consumer needs and a
shift towards cosmetic dentistry will drive the market for high end dental solutions.
In China and other parts of Asia there has
been a rapid growth in living standards. China’s young, emerging middle class is beginning to equate accumulation of possessions
and leisure opportunities with quality of life. An estimated 415,000 Chinese had more than $1 million in disposable assets in 2007,
more than any other country, according to the Merrill-Lynch Asia-Pacific Wealth Report. Up to 170 million people, or 13% of the
population, can afford luxury brands and the number will reach 250 million next year, according to the China Association of Branding
Strategy. These regions have a huge potential for growth in cosmetic dentistry due to low market penetration. Consequently, these
countries are exhibiting high demand for modern and sophisticated technology and equipment in the dental market. Overall demand
for dental products in China is expected to climb to 11% annually through 2012. In a report published by Millennium Research Group
with regard to Chinese markets for Dental Implants, a similar target market as that for our GlamSmile products, finds that this
emerging market is growing quickly at a compound annual growth rate of more than 35%. A strong driver of this growth is the deregulation
of dental services in China. Dental services in China are generally provided in government-managed facilities; however, ongoing
deregulation of dental services is resulting in the emergence of an increased number of private dental practices and increasing
accessibility to dental services. Another major driver in the Chinese market is the frequency of teeth stained by Tetracycline.
For decades, Tetracycline was one of the most prescribed antibiotics in China causing many individuals to suffer from stained teeth.
Excessive use of fluoride in drinking water causes a similar problem. When tetracycline exposure occurs while teeth are forming,
it creates a permanent gray or brown stain, causing either uniform discoloration of the entire tooth or forming horizontal bands
of stain of varying intensity that can range from mild to very dark. Veneers are the treatment of choice for this condition. In
2008, the Asia-Pacific market for dental prosthetics (crowns, bridges and dentures) was valued at over $6 billion. The Asia-Pacific
market includes Australia, Japan and South Korea. The aging population and greater demand for aesthetic dentistry are driving forces
in the prosthetics market. The global economic crisis, which began in 2008, has led to a slowdown in the overall market; however,
the market is expected to grow over the forecast period, reaching $7.2 billion by 2015.
We believe that our GlamSmile products which
are affordable in comparison to traditional veneers, pain free, easy to apply, and provide instant results make us uniquely positioned
to capitalize on the market trends in Asia, Europe and the United States.
Growth Strategy
Today, our strategic plan is to focus our
vertically integrated development, manufacturing and marketing resources on selling our GlamSmile veneers direct to consumers by
using all forms of direct response media including the internet, print, radio, television and social network media, to expand our
presence in China and Europe. In our marketing efforts we intend to emphasize the ease, convenience, affordability and
dramatic, instant results as demonstrated by before and after photos that are attained as a result of GlamSmile veneers. We will
also feature our “Until You Smile” satisfaction guarantee. Using the success formula we experienced in China and Belgium
using a "Smile Consultant" to help maintain control of the sales process and close the sale, our distribution will be
through both owned and operated Glamsmile Studios as well as affiliations with existing dental practices and partner retail centers
in Asia, Europe.
Our current strategic marketing and distribution
plan includes a combination of owned and licensed GlamSmile centers depending upon the size and location of the market, with us
managing the marketing efforts, patient communications and sales process. We established two geographic divisions, Asia
and Europe , each of which will promote GlamSmile veneer treatments in their respective territories. We plan to establish
three types of GlamSmile Centers depending upon market factors and government regulation.
|
|
·
|
Owned Centers.
These are centers in which the Company
will own, control and/or manage all aspects of the operation including the facilities, equipment, personnel, marketing, insurance
risk and other operating costs and will either employ or contract with dentists to perform the necessary dental services. In China,
we will continue to principally rely on our owned and operated dental GlamSmile clinics or centers.
|
|
|
·
|
Licensed Centers
.
In many markets we will
seek to identify and recruit cosmetic dentists that have existing practices and who endorse the GlamSmile veneer products. In these
markets, we will contract with dental practices and the Company will recognize revenue through the sale of veneer trays plus marketing
and other service fees to be charged to the dentist for services performed by the Company.
|
|
|
·
|
Distributors
. In markets where we lack the expertise with respect to managing marketing and where local regulation and/or custom may make it impractical to deploy an owned or licensed center approach we will look to appoint distributors who will be granted exclusive rights to market and distribute our GlamSmile products directly to consumers subject to minimum performance criteria and/or initial territory fees. In this model the distributor will be expected to invest in all marketing and sales conversion costs in their market. Our revenues will be derived principally from sales of our GlamSmile veneer products to the distributor.
|
In order to support and facilitate our growth
strategy, it is our intention to restructure our subsidiary companies to better manage our GlamSmile related operations. In conjunction
with this restructuring, we intend to have the intellectual property and other assets related to GlamSmile contributed to a new
entity to be formed to be called GlamSmile Worldwide. New entities would also be created called GlamSmile Asia and GlamSmile Europe,
each with licensed rights to use and exploit the GlamSmile technology in their respective territories.
B2B Market and Distribution
Starting in Belgium and the Netherlands,
our products have been introduced utilizing our Distributor Assisted Marketing programs. We implement our program by
first identifying an established dealer in each market with a well-developed sales force familiar with sales of capital equipment
to the professional dentist community. Second, we develop aggressive lead generation programs and other marketing techniques
which served as a blue print for the dealers to implement. The combination of a well-trained dealer force and dealer-assisted
marketing and lead generation programs has proven to be far more effective than utilizing a direct sales approach, which is much
slower and more costly to establish. This process has been repeated for both the professional dentist and retail, over-the-counter
markets in each country. As a result of this approach, we have been able to establish dealers in 28 countries encompassing,
Europe, Asia, Latin America, the Pacific Rim and the Middle East.
We previously sold our GlamSmile product
in the United States and throughout the world with the exception of certain excluded territories and certain B2C markets pursuant
to a distribution agreement. However, on March 27, 2012, the distribution agreements with Den-Mat were terminated pursuant to a
certain Termination and Distribution Agreement with Den-Mat (“Den-Mat Distribution Agreement”). Pursuant to the Den-Mat
Distribution Agreement, we granted Den-Mat a non-exclusive, irrevocable, perpetual, royalty free, license to use within certain
territory, which among other territories excludes China, Macau, Hong Kong, and Taiwan, the intellectual property that was the subject
of the license to Den-Mat under the Amended and Restated Distribution, License and Manufacturing Agreement dated June 3, 2009,
as amended from time to time (“Prior Agreements”), as such intellectual property relates the products which was the
subject of the Prior Agreements. In connection with the termination of the Prior Agreements, under the Den-Mat Distribution Agreement,
Den-Mat agreed to pay us $200,000. We currently sell our products in Asia, Europe and the Middle East directly to consumers
using our direct to consumer model, which includes our GlamSmile Smile Design-Virtual Studio, and GlamSmile Studios.
In September
2010, we entered into a license agreement with Excelsior Medical (“EM”) (the “EM license agreement”).
Under the EM license agreement, we granted EM an exclusive license to certain Asian territories in exchange for $500,000
which was received during the year ended March 31, 2011. The Company received a further $500,000 from EM as an advance
payment for veneers. The $500,000 advance, less taxes withheld, was recorded as deferred revenue of $475,250 as of
March 31, 2011. Effective as of January 11, 2012, the Company entered into a Rescission Agreement with EM and Asia Best
Healthcare Co., Ltd. Under the Rescission Agreement, the Company agreed to repay a total of $1,000,000 received under the
Distribution Agreement, plus a simple interest rate of 5%, beginning on June 30, 2012, according to the following payment
schedule: (i) $250,000 to be paid no later than June 30, 2012, (ii) $250,000 plus interest on June 30, 2012, (iii)
$250,000 plus interest on December 31, 2012, and (iv) $250,000 plus interest on June 30, 2013. The Company also agreed to
secure such obligations owed to EM with certain collateral of the Company
Locations
The Company leases
an office facility of 5,187 square feet in Gent, Belgium from an unrelated party pursuant to a nine year lease commencing September
1, 2008. at a base rent of €5,712 per month for the total location ($7,624 per month at March 31, 2012).
Secondly, the Company
leases an office facility of 1,991 square feet in Rome, Italy to support the sales and marketing division of our veneer business,
from an unrelated party pursuant a six year lease commencing July 1, 2011, at a base rent of € 6,500 per month for the total
location ($8,675 per month at March 31, 2012).
Manufacturing
Prior to 2003, all of the manufacturing
related to our dental products were conducted through third party manufacturers under our supervision thereby minimizing demands
on capital resources. Beginning in 2003, parts of the manufacturing and the majority of the final assembly of our products
were brought in-house, thereby improving control over product quality while significantly reducing product costs. These
efforts were expanded significantly during the fiscal year ended March 31, 2006, in particular with regard to the expansion of
in-house manufacturing capabilities for our gel products and foam strips. The Company still manufactures many of its products in
its facility in Deurle, Belgium, as well as through outsourced manufacturing in China and France. In December 2005,
our manufacturing facility became ISO 9001:2000 certified and ISO 13485:2003 certified which includes the certification for the
manufacture of medical devices.
Research and Development
Our research and development expenses decreased
$119,305 to $ 309,939 for the year ended March 31, 2012 as compared to $429,244 for the year ended March 31, 2011, an increase
of 27.79%. Research and development expenses have decreased primarily because of the completion of the research and
development of the RemeScar and the launch of the final RemeScar product, reducing our research and development expense significantly.
Secondly, the deconsolidation of our Sylphar division end of September 2011, converted to the equity method, contributed also to
the decrease in Research and Development expenses.
Although we are currently developing a ‘prefab’
veneer which would be a ‘ready-to-use’ veneer, called ‘River 8’ which we plan to offer at a lower price
than traditional veneers, we anticipate that our research and development costs will decrease over the next fiscal year since GlamSmile
Asia was deconsolidated at the end of January 2012 along with the deconsolidation of the Sylphar Division at the end of September
2011, reducing as such our consolidated research and development costs.
Intellectual Property
GlamSmile.
We have filed
a patent application related to the design and manufacturing process of the GlamSmile product in the European Union on March 2,
2003 and the United States on December 20, 2007. The patent applications are pending as of March 31, 2012. We
also have international trademark registration for GlamSmile in the United States until August 16, 2017 and in the European
Union until April 6, 2017. In addition, we have secured the domain name www.glamsmile.com as well as other related internet domains
in our targeted markets. We also have ongoing research and development efforts to improve and expand our current technology and
to develop new dental products. We intend to continue to apply for patents when we believe it is in our interest
to do so and as advised by patent counsel. We rely and will continue to rely on trade secrets, know-how and other unpatented
proprietary information in our business. Certain of our key employees and consultants are required to enter into confidentiality
and/or non-competition agreements to protect our confidential information.
Teeth Whitening Patents.
In
October 2004, the Company acquired from the inventor the exclusive, perpetual license to two issued United States patents which
are applicable to several teeth whitening products, which consists of a patent related to a heated dental tray which expires on
February 8, 2019 and a patent related to device for heating the teeth and uses therefor which expires on February 23,
2021. Pursuant to the terms of the license agreement, the Company was granted an exclusive, perpetual license to manufacture, market,
distribute and sell the products on the US market contemplated by the patents subject to the payment of $65,000 as reimbursement
to the patent holder for legal and other costs associated with obtaining the patents, which was paid in October 2004, and royalties
for each unit sold subject to an annual minimum royalty of $100,000 per year. The Company is amortizing the initial cost of $65,000
for these patents over a ten year period. The license agreement contemplated the launch of the product in
the United States and provided for payment of royalties on those sales. The launch of the product in the United States
was unsuccessful and as a result, the product was taken from the United States market. The Company failed to make payments
to the Licensor of at least $100,000 USD per year, consequently all rights to the patents reverted to the licensor effective March
31, 2006. Accordingly, the patents have been written off as of March 31, 2011.
Universal Applicator Patent.
In
September 2004, we entered into an agreement with Lident N.V. (“Lident”), a company controlled by Mr. De Vreese, the
Company’s Chairman, to obtain an option, exercisable through December 31, 2005, to license an international patent (excluding
the US) and worldwide manufacturing and distribution rights for a potential new product which Lident had been assigned certain
rights by the inventors of the products, who are unrelated parties, prior to Mr. De Vreese association with the Company. The patent
is an Italian patent which relates to a single use universal applicator for dental pastes, salves, creams, powders, liquids and
other substances where manual application could be relevant. We have filed to have the patent approved throughout Europe. The agreement
required us to advance to the inventors through Lident a fully refundable deposit of €100,000 subject to our due diligence
regarding the enforceability of the patent and marketability of the product, which, if viable, would be assigned to us for additional
consideration to the inventors of €100,000 and an ongoing royalty from sales of products related to the patent equal to 3%
of net sales and, if not viable, the deposit would be repaid in full by Lident. The consideration we agreed to pay Lident upon
the exercise of the option is the same as the consideration Lident is obligated to pay the original inventors. Consequently, Lident
would not have profited from the exercise of the option. Furthermore, at a meeting of our Board of Directors on July 13, 2005,
the Board accepted Lident’s offer to facilitate an assignment of Lident’s intellectual property rights to the technology
to us in exchange for the reimbursement of Lident’s actual costs incurred relating to the intellectual property. Consequently,
when we exercise the option, all future payments, other than the reimbursement of costs would be paid directly to the original
inventors and not to Lident.
On December 12, 2005, the Company exercised
the option and the Company and the patent holder agreed to revise the assignment agreement whereby the Company agreed to pay €50,000
additional compensation in the form of prepaid royalties instead of the €100,000 previously agreed, €25,000 of which
was paid by the Company in September 2005 and the remaining €25,000 is to be paid upon the Company’s first shipment
of a product covered by the patent. As of March 31, 2011 the Company has not yet received the final Product and therefore, $35,433
(€25,000) has been written off during the year ended March 31, 2011. Further, the patent has been completely amortized over
five (5) years and accordingly, the Company has recorded $118,840 of accumulated amortization for this patent as of March 31, 2011.
Major Customers
At March 31, 2012 five customers accounted
for a total of 65% of the Company’s trade accounts receivable and one of those customers accounted for 26.7 % of total accounts
receivable. At March 31, 2011 five customers accounted for a total of 61% of the Company’s trade accounts receivable
and one of those customers accounted for 24.6% of total accounts receivable. The Company performs ongoing credit evaluations
of its customers and normally does not require collateral to support accounts receivable.
Competition
GlamSmile
. Our competition
consists of both alternative procedures that can be performed to achieve in part the results that would be achieved through a GlamSmile
procedure, as well as competition from dentists not within the GlamSmile network who provide veneer procedures. With regard to
alternative procedures, options available to the consumer include various whitening procedures, dental implants, dental bonding
and dental caps. With the exception of whitening procedures, which for the most part cannot address many of the dental issues solved
by GlamSmile veneers, the remaining alternatives all involve more cost, more patient discomfort and more time to complete. There
are many dental practitioners that perform traditional veneer procedures. In most cases, traditional veneers will also be significantly
more costly then GlamSmile veneers and require the dentist to remove more of the existing tooth material as well as requiring multiple
patient visits to complete. That said, there will be existing practitioners that believe they can attain more customized results
with the individual veneer approach as opposed to the GlamSmile tray approach and may be reluctant to offer our less costly procedure.
To the best of our knowledge, GlamSmile will be “first to market” with respect to a direct to consumer advertising
and promotion campaign for veneers anywhere which should enable us to capture market share in what we believe will be a rapidly
growing market. Further, we have filed for patents on our proprietary tray delivery systems and have developed years of knowhow
relating to treating patients with the multiple veneer approach. However, new technologies are continue to be developed and new
processes could be designed that would not violate our patents and result in similar solutions that could compete with GlamSmile
products. Because we are uniquely positioned to have the ability to control the entire process from manufacturing to marketing
to distribution, we believe it is feasible for us to have complete control and flexibility to maximize margins and respond
aggressively to any competitive situation.
Teeth Whitening
. International
markets including Europe, Asia and Latin America have followed the United States’ lead in expanding offerings in the areas
of teeth whitening. Leading the way in both the professional dentist segment has been United States based companies
seeking to expand their distribution. Impeding these efforts has been the inability of many of these companies to fully
understand the differences from both a distribution and a regulatory standpoint that apply in each of the European and Asian markets. Notwithstanding
the formation of the European Union and its efforts to standardize regulatory and business practices throughout Europe, these practices
in reality vary widely from country to country. Competition in the professional dentistry product lines comes primarily
from the larger United States based competitors including Brite-Smile, Rembrandt (now a subsidiary of Gillette Company, Inc.),
Discuss Dental, Inc. and Zoom. All of these companies offer light and whitening solutions to the professional dentist
community. Despite our competition’s advantage with respect to size, resources and name recognition, we have continued
to maintain market share in this highly competitive segment for the following reasons:
|
|
•
|
Better combined pricing strategy than the competition when considering
net cost for whitening materials and initial cost of light.
|
|
|
•
|
Dual purpose light to maximize value of initial investment.
|
|
|
•
|
Ease of use from automated functionality of light, speed and
gel application method.
|
|
|
•
|
Superior gel formulation which maximizes performance while minimizing
sensitivity.
|
|
|
•
|
Home maintenance kit for improved patient satisfaction.
|
Regulatory Issues
Medical Device
As we market dental products which are legally
defined to be medical devices, we are considered to be a medical device manufacturer and as such we are subject to the regulations
of, among other governmental entities, the United States Food and Drug Administration (the “FDA”) and the corresponding
agencies of the states and foreign countries in which we sell our products. These regulations govern the introduction
of new medical devices, the observance of certain standards with respect to the manufacture and labeling of medical devices, the
maintenance of certain records and the reporting of potential product problems and other matters. A failure to comply
with such regulations could have material adverse effects on our business.
The Federal Food, Drug and Cosmetic Act
(“FDC Act”) regulates medical devices in the United States by classifying them into one of three classes based on the
extent of regulation believed necessary to ensure safety and effectiveness. Class I devices are those devices for which
safety and effectiveness can reasonably be ensured through general controls, such as device listing, adequate labeling, pre-market
notification and adherence to the Quality System Regulation (“QSR”) as well as medical device reporting, labeling and
other regulatory requirements. Some Class I medical devices are exempt from the requirement of pre-market approval or
clearance. Class II devices are those devices for which safety and effectiveness can reasonably be ensured through the
use of special controls, such as performance standards, post-market surveillance and patient registries, as well as adherence to
the general controls provisions applicable to Class I devices. Class III devices are devices that generally must receive
pre-market approval by the FDA pursuant to a pre-market approval application (“PMA”) to ensure their safety and effectiveness. Generally,
Class III devices are limited to life sustaining, life supporting or implantable devices; however, this classification can also
apply to novel technology or new intended uses or applications for existing devices.
Before most medical devices can be marketed
in the United States, they are required by the FDA to secure either clearance of a pre-market notification pursuant to Section
510(k) of the FDC Act (a “510(k) Clearance”) or approval of a PMA. Obtaining approval of a PMA can take
several years. In contrast, the process of obtaining 510(k) Clearance generally requires a submission of substantially
less data and generally involves a shorter review period. Most Class I and Class II devices enter the market via the
510(k) Clearance procedure, while new Class III devices ordinarily enter the market via the more rigorous PMA procedure. In
general, approval of a 510(k) Clearance may be obtained if a manufacturer or seller of medical devices can establish that a new
device is “substantially equivalent” to a predicate device other than one that has an approved PMA. The
claim for substantial equivalence may have to be supported by various types of information, including clinical data, indicating
that the device is as safe and effective for its intended use as its legally marketed equivalent device. The 510(k)
Clearance is required to be filed and cleared by the FDA prior to introducing a device into commercial distribution. Market
clearance for a 510(k) Notification submission may take 3 to 12 months or longer. If the FDA finds that the device is
not substantially equivalent to a predicate device, the device is deemed a Class III device, and a manufacturer or seller is required
to file a PMA. Approval of a PMA for a new medical device usually requires, among other things, extensive clinical data
on the safety and effectiveness of the device. PMA applications may take years to be approved after they are filed. In
addition to requiring clearance or approval for new medical devices, FDA rules also require a new 510(k) filing and review period
prior to marketing a changed or modified version of an existing legally marketed device if such changes or modifications could
significantly affect the safety or effectiveness of that device. The FDA prohibits the advertisement or promotion of
any approved or cleared device for uses other than those that are stated in the device’s approved or cleared application.
We believe that the GlamSmile products will not require a 510(k) submission because the products fall within an exemption under
the 510(k) regulation.
International sales of medical devices are
also subject to the regulatory requirements of each country. In Europe, the regulations of the European Union require
that a device have a CE Mark, a mark that indicates conformance with European Union laws and regulations before it can be sold
in that market. In China, the State Food and Drug Administration (“SFDA”) is the agency primarily
responsible for regulating medical devices. The regulatory international review process varies from country to country. We
previously relied upon our distributors and sales representatives in the foreign countries in which we market our products to ensure
we comply with the regulatory laws of such countries; however, during the year ended March 31, 2006 we expanded our own Research
and Development personnel to enable us to provide greater assistance and play a more proactive role in obtaining local regulatory
approvals, especially in Europe. We currently have an in-office regulatory affairs representative who is responsible
for coordinating local and international approvals as well as our ISO:9001 and ISO:13485 (medical device). In China,
we continue to rely on our distributors and strategic partners to ensure compliance with regulatory laws of China.
Fee Splitting and Arrangements with Health Professionals
Many states in the United States and countries
worldwide have laws that prohibit business corporations like us from practicing medicine, employing dentists to practice medicine,
exercising control over medical decisions by dentists, or engaging in certain arrangements, such as fee splitting, with dentists.
In light of these restrictions, in certain markets where permissible we intend to operate by maintaining management contracts with
dentists owned corporations or other business entities that employ or contract with dentists to provide the GlamSmile and other
dental services. Under these arrangements we will perform under contract only nonmedical administrative services, will not offer
medical services and will not exercise influence or control over the practice of medicine by the dentists employed by such business
entities. In markets where fee splitting with a business corporation is prohibited, the fees that will be received by us will have
been established on a basis that we believe complies with the applicable laws. However, regulatory authorities or other parties
may assert that, despite these arrangements, we are engaged in the corporate practice of medicine or that the contractual arrangements
with the affiliated professional contractors constitute unlawful fee splitting, in which case we could be subject to civil or criminal
penalties, the contracts could be found legally invalid and unenforceable (in whole or in part) or we could be required to restructure
our contractual arrangements.
Dental Practice
Dental practices are subject to local and
national regulations worldwide. Although the laws and regulations for operating a dental practice and engaging in dental
services vary from country to country, in general our dental studios require a health license and a business license. In
addition, the dentists providing services in the dental studios are also required to be licensed to practice.
While the Company believes it is in substantial
compliance with the laws and regulations which regulate its business, and that it possesses all the licenses required in the conduct
of its business, the failure to comply with any of those laws or regulations, or the imposition of new laws or regulations could
negatively impact the Company’s business .
Costs and Effects of Compliance with Environmental Laws and
Regulations
We are not in a business that involves the
use of materials in a manufacturing stage where such materials are likely to result in the violation of any existing environmental
rules and/or regulations. Further, we do not own any real property that could lead to liability as a landowner. Therefore,
we do not anticipate that there will be any substantial costs associated with the compliance of environmental laws and regulations.
Employees
We currently retain 16 full-time employees
in Belgium. We currently have one employee, Mr. Stephen Ross, our Chief Financial Officer, located in the
United States. Our subsidiary, Remedent, N.V., has an employment agreement with Mr. Philippe Van Acker our Chief Accounting
Officer. We have no other employment agreements with our executive officers.
Financial Information About Geographic Areas
Refer to Note 20, “Segment Reporting,”
to our Consolidated Financial Statements included under Item 8, “Financial Statements and Supplementary Data”
for financial information about our geographic areas.
Other Information
Our Internet address is
www.remedent.com.
We make available free of charge on our website our annual reports on Form 10-K, quarterly reports on Form 10-Q, current
reports on Form 8-K, and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange
Act as soon as reasonably practicable after we electronically file such material with, or furnish it to, the Securities and Exchange
Commission (“SEC”). Other than the information expressly set forth in this annual report, the information contained,
or referred to, on our website is not part of this annual report.
The public may also read and copy any materials
we file with the SEC at the SEC’s Public Reference Room at 100 F Street, NE, Washington, DC 20549. The public may
obtain information on the operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330. The SEC also maintains a
website at
www.sec.gov
that contains reports, proxy and information statements, and other information regarding issuers,
such as us, that file electronically with the SEC.
ITEM 1A — RISK FACTORS
RISK FACTORS
Investment in our common stock involves
risk. You should carefully
consider the risks we describe below before deciding to invest. The market
price of our common stock could decline due to any of these risks, in which
case you could lose all or part of your investment. In
assessing these risks,
you should also refer to the other information included in this Annual Report,
including our
consolidated financial statements and the accompanying notes. You
should pay particular attention to the fact
that we are a holding company with
substantial operations in Belgium and China and are subject to legal and regulatory
environments that in many respects differ from that of the United States. Our
business, financial condition
or results of operations could be affected
materially and adversely by any of the risks discussed below and any others not
foreseen. This discussion contains forward-looking statements.
Risks Relating To Our Business
We have a history of losses and we could suffer losses in
the future.
With the exception of a small profit of
$16,149 on revenue of $5,234,855 for the fiscal year ended March 31, 2004 and a profit of $490,483 on revenue of $9,687,292 for
the fiscal year ended March 31, 2012, we have had a history of substantial losses. Our losses were $1,547,175 on revenues
of $12,581,708 for the year ended March 31, 2011 and $2,349,915 on revenues of $8,247,960 for the year ended March 31, 2010 We
expect to continue to incur increasing cost of revenues, sales and marketing and general and administrative expenses in connection
with our business strategy. However, despite our efforts, there is no assurance that we will be able to achieve or sustain
profitability.
Our cash and cash equivalents are insufficient to support
our operations and there is substantial doubt exists about our ability to continue as a going concern.
Our cash and cash equivalents of $203,584
as of March 31, 2012 are not sufficient to support our operations through our current fiscal year and we need additional financing.
During the year ended March 31, 2012, we have been unable to generate cash flows sufficient to support our operations and have
been dependent on loans and strategic partnerships. We remain dependent on outside sources of funding until our results of operations
provide positive cash flows. During our current fiscal year, we expect to continue to incur operating losses. With our current
level of funding, substantial doubt exists about our ability to continue as a going concern.
The financial statements contained in this
report do not include any adjustments relating to the recoverability and classification of recorded asset amounts or amounts and
classification of liabilities that might be necessary should we be unable to continue in existence. Our ability to continue as
a going concern is dependent upon our ability to generate sufficient cash flows to meet our obligations on a timely basis, to obtain
additional financing as may be required, and ultimately to attain profitable operations. However, there is no assurance that profitable
operations or sufficient cash flows will occur in the future.
We have supported current operations by
raising additional operating cash through loans and strategic partnerships. This has provided us with the cash inflows to continue
our business plan, but has not resulted in significant improvement in our financial position. We are considering alternatives to
address our cash flow situation that include raising capital through additional sale of our common stock and/or debentures.
This could result in substantial dilution
of existing stockholders. There can be no assurance that our current financial position can be improved, that we will be able to
locate a strategic partner, that we can raise additional working capital, or that we can achieve positive cash flows from operations.
Our long-term viability as a going concern is dependent upon our ability to (i) locate sources of debt or equity funding to meet
current commitments and near-term future requirements and (ii) achieve profitability and ultimately generate sufficient cash flow
from operations to sustain our continuing operations.
Substantially all of our assets are secured under a credit
facility with BNP Paribas Fortis
Bank, a bank located outside of the United States, and in the event of default
under
the credit facility we may lose all of our assets.
The Company has a mixed-use line of credit
facility with BNP Paribas Fortis, a Belgian bank (the “Facility”). The Facility is secured by a first lien
on the assets of Remedent N.V. On January 3, 2008, an amendment was made decreasing the mixed-use line of credit
to €2,050,000, to be used by Remedent NV and/or Sylphar NV. Each line of credit carries its own interest rates and fees as
provided in the Facility and varies from the current prevailing bank rate
The latest amendment to the Facility, dated
June 7, 2010, amended and split the line of credit to €1,250,000, to be used by Remedent N.V. and € 1,000,000 to be used
Sylphar NV. Each line of credit carries its own interest rates and fees as provided in the Facility and vary from the current prevailing
bank rate of approximately 3.27%, for draws on the credit line, to 9.9% for advances on accounts receivable concerning Remedent
N.V. and similar for Sylphar N.V. Remedent N.V. is currently only utilizing two lines of credit, advances based on account receivables
and the straight loan. As of March 31, 2012 and March 31, 2011, Remedent N.V. had in aggregate, $1,079,263 and $1,452,024 in advances
outstanding, respectively, under the mixed-use line of credit facilities. Although we are current in our obligations under this
Facility, in the event of a default under this Facility we may lose our assets. In addition, our Facility currently
provide the Company with cash to accelerate our expansion plan to open new GlamSmile Studios. Accordingly, without
the availability of funds from our Facility, or alternative source of funding, it would take us longer to open new Studios.
We depend on strategic relationships with third parties for
sales and marketing performance and revenues in the People’s Republic of China and certain territories in North America,
and failure to maintain these relationships, poor performance by these companies or disputes with these companies could negatively
impact our business.
We rely on significant strategic relationships
with third parties, for our sales and marketing performance in certain territories. These include collaborations with strategic
partners in China for our dental studios. Reliance on collaborative relationships poses a number of risks, including
the risk that:
|
|
•
|
We are unable to control the resources our corporate partners
devote to our programs or products;
|
|
|
•
|
disagreements with our corporate partners could cause delays
in, or termination of, the research, development or commercialization of product candidates or result in litigation or arbitration;
|
|
|
•
|
contracts with our corporate partners may fail to provide significant
protection or may fail to be effectively enforced if one of these partners fails to perform;
|
|
|
•
|
our corporate partners with marketing rights may choose to pursue
competing technologies or to devote fewer resources to the marketing of our products; and
|
|
|
•
|
our distributors and our corporate partners may be unable to pay us, particularly in light of current economic conditions.
|
Given these risks, there is a great deal
of uncertainty regarding the success of our current and future collaborative efforts. If these efforts fail, our product development
or commercialization of new products could be delayed or revenues from products will most likely decline.
We may not have access to capital in the future as a result
of disruptions in capital and credit markets.
Although we currently have additional credit
available under our Facility with BNP Paribas Fortis Bank, we may not be able to access our funds in the future. Our
access to the funds under our current credit facility with BNP Paribas Fortis Bank is dependent on the ability of the financial
institution that is party to the facility to meet its funding commitments. BNP Paribas Fortis Bank may not be able to meet its
funding commitments if it experiences shortages of capital and liquidity or if it experiences excessive volumes of borrowing requests
within a short period of time. Moreover, longer term volatility and continued disruptions in the capital and credit markets as
a result of uncertainty, changing or increased regulation of financial institutions, reduced alternatives or failures of significant
financial institutions could affect adversely our access to the liquidity needed for our business in the longer term. Such disruptions
could require us to take measures to conserve cash until the markets stabilize or until alternative credit arrangements or other
funding for our business needs can be arranged. The disruptions in the capital and credit markets have also resulted in higher
interest rates on publicly issued debt securities and increased costs under credit facilities. The continuation of these disruptions
would increase our interest expense and capital costs and could affect adversely our results of operations and financial position
including our ability to grow our business through acquisitions.
We have obtained loans from third parties recently and anticipate
that we will need additional capital during the next twelve months. In addition, if we are unable to secure additional
financing to meet our future capital
this will have adverse effects on our financial condition.
We obtained on June 3, 2011, a long term
loan for $1,000,000 from an unrelated party in order to support and finance the expansion of new GlamSmile Studios in Mainland
China. This loan is secured by certain assets we own and in the event of default, the lenders will have a right to the collateral
granted to them under the loan agreements and we will lose our ownership interest in the assets. A loss of our collateral
will have material adverse effect on our Asia operations, our business and financial condition.
Effective January 11,
2012, the Company entered into a Rescission Agreement with EM and Asia Best Healthcare Co., Ltd. Under the Rescission Agreement,
the Company agreed to repay a total of $1,000,000 received under the Distribution Agreement, plus a simple interest rate of 5%,
beginning on June 30, 2012, according to the following payment schedule: (i) $250,000 to be paid no later than June 30, 2012, (ii)
$250,000 plus interest on June 30, 2012, (iii) $250,000 plus interest on December 31, 2012, and (iv) $250,000 plus interest
on June 30, 2013. The Company also agreed to secure such obligations owed to EM with certain collateral of the Company.
In addition, we anticipate needing significant
capital to introduce new products, further develop our existing products, increase awareness of our brand names and expand our
operating and management infrastructure as we grow sales in Europe, Asia and South America and launch sales and distribution
activities in the United States. We may use capital more rapidly than currently anticipated and incur higher operating
expenses and generate lower revenue than currently expected, and we may be required to depend on external financing to satisfy
our operating and capital needs. We may need new or additional financing in the future to conduct our operations or
expand our business. Any sustained weakness in the general economic conditions and/or financial markets in the United States or
globally could affect adversely our ability to raise capital on favorable terms or at all. From time to time we have relied, and
may also rely in the future, on access to financial markets as a source of liquidity to satisfy working capital requirements and
for general corporate purposes. We may be unable to secure additional debt or equity financing on terms acceptable to us, or at
all, at the time when we need such funding. If we do raise funds by issuing additional equity or convertible debt securities,
the ownership percentages of existing stockholders would be reduced, and the securities that we issue may have rights, preferences
or privileges senior to those of the holders of our common stock or may be issued at a discount to the market price of our common
stock which would result in dilution to our existing stockholders. If we raise additional funds by issuing debt, we
may be subject to debt covenants, such as the debt covenants under our secured credit facility, which could place limitations on
our operations including our ability to declare and pay dividends. Our inability to raise additional funds on a timely
basis would make it difficult for us to achieve our business objectives and would have a negative impact on our business, financial
condition and results of operations.
Our results of operations may be adversely impacted by currency
fluctuations.
We currently have operations in Belgium
and have product sales in Europe, the Middle East, South America and Asia. A significant portion of our revenue is in
currencies other than United States dollars, including Euros, Hong Kong dollar and the Chinese Renminbi (“RMB”). Because
our financial statements are reported in United States dollars, fluctuations in Euros, Hong Kong dollar and RMB against the United
States dollar may cause us to recognize foreign currency transaction gains and losses, which may be material to our operations
and impact our reported financial condition and results of operations
Substantially all of our assets and our operations are located
outside of the United States,
a significant number of sales are generated outside of the United States subjecting
us to risks associated with international operations.
Our operations are primarily in Europe
and Asia and 88% of our sales for the fiscal year ended March 31, 2012 were generated from customers outside of the United States,
compared to 82% of our sales for the fiscal year ended March 31, 2011. The international nature of our business subjects us to
the laws and regulations of the jurisdictions in which we operate and sell our products. In addition, we are subject
to risks inherent in international business activities, including:
|
|
•
|
difficulties in collecting accounts receivable and longer collection periods,
|
|
|
|
|
|
|
•
|
Changes in overseas economic conditions,
|
|
|
|
|
|
|
•
|
fluctuations in currency exchange rates,
|
|
|
|
|
|
|
•
|
potentially weaker intellectual property protections,
|
|
|
|
|
|
|
•
|
changing and conflicting local laws and other regulatory requirements,
|
|
|
|
|
|
|
•
|
Political and economic instability,
|
|
|
•
|
war, acts of terrorism or other hostilities,
|
|
|
|
|
|
|
•
|
potentially adverse tax consequences,
|
|
|
|
|
|
|
•
|
difficulties in staffing and managing foreign operations, or
|
|
|
|
|
|
|
•
|
tariffs or other trade regulations and restrictions.
|
Our quarterly sales and operating results have fluctuated
and may continue to
fluctuate in future periods which may cause the price of our common stock to
decline.
Our quarterly sales and operating results
have fluctuated and are likely to continue to vary from quarter to quarter due to a number of factors, many of which are not within
our control. Factors that might cause quarterly fluctuations in our sales and operating results include, but are not
limited by the following:
|
|
•
|
Variation in demand for our products, including variation due to seasonality;
|
|
|
|
|
|
|
•
|
Our ability to research, develop, introduce, market and gain market acceptance of new products and product enhancements in a timely manner;
|
|
|
|
|
|
|
•
|
Our ability to control costs;
|
|
|
|
|
|
|
•
|
The size, timing, rescheduling or cancellation of orders from distributors;
|
|
|
|
|
|
|
•
|
The introduction of new products by competitors;
|
|
|
|
|
|
|
•
|
Long sales cycles and fluctuations in sales cycles;
|
|
|
|
|
|
|
•
|
The availability and reliability of components used to manufacture our products;
|
|
|
|
|
|
|
•
|
Changes in our pricing policies or those of our suppliers and competitors, as well as increased price competition in general;
|
|
|
|
|
|
|
•
|
The risks and uncertainties associated with our international business;
|
|
|
|
|
|
|
•
|
Costs associated with any future acquisitions of technologies and businesses;
|
|
|
|
|
|
|
•
|
Developments concerning the protection of our proprietary rights; and
|
|
|
|
|
|
|
•
|
General global economic, political, international conflicts, and acts of terrorism.
|
We are economically sensitive to general economic conditions,
including continued weakening of the economy, therefore the sale of our products could be adversely affected.
Our industry is sensitive to recessions
in the general economy and future economic outlook. Our results may be dependent on a number of factors impacting consumer spending,
including general economic and business conditions; and consumer confidence. The demand for our dental products may decline during
recessionary periods and at other times when disposable income is lower. A downturn or an uncertain outlook in the economy may
materially adversely affect our business.
An unsuccessful material strategic transaction or relationship
could result in operating difficulties and other harmful consequences to our business.
We have evaluated, and expect to continue
to evaluate, a wide array of potential strategic transactions and relationships with third parties. From time to time,
we may engage in discussions regarding potential acquisitions or joint ventures. Any of these transactions could be material to
our financial condition and results of operations, and the failure of any of these material relationships and transactions may
have a negative financial impact on our business.
Our products may be subject to government regulation and
failure to comply with
applicable regulations could result in fines, suspensions, seizure actions,
product recalls,
injunctions and criminal prosecutions.
Before most medical devices can be marketed
in the United States, they are required by the United States Food and Drug Administration (“FDA”) to secure either
clearance of a pre-market notification pursuant to Section 510(k) of the Federal Food, Drug and Cosmetic Act (“FDC Act”)
(a “510(k) Clearance”) or approval of a pre-market approval application (“PMA”). Obtaining approval
of a PMA application can take several years. In contrast, the process of obtaining 510(k) Clearance generally requires
a submission of substantially less data and generally involves a shorter review period. As discussed more specifically
under the subsection title “Regulatory Issue,” most Class I and Class II devices enter the market via the 510(k) Clearance
procedure, while new Class III devices ordinarily enter the market via the more rigorous PMA procedure. Approval of
a PMA application for a new medical device usually requires, among other things, extensive clinical data on the safety and effectiveness
of the device. PMA applications may take years to be approved after they are filed. In addition to requiring
clearance or approval for new medical devices, FDA rules also require a new 510(k) filing and review period, prior to marketing
a changed or modified version of an existing legally marketed device, if such changes or modifications could significantly affect
the safety or effectiveness of that device. The FDA prohibits the advertisement or promotion of any approved or cleared
device for uses other than those that are stated in the device’s approved or cleared application.
We have received approval from the FDA to
market our RemeCure dental curing lamp in the United States. We submitted our application for approval on FDA Form 510(k)
on October 30, 2002 and received FDA approval for this product on January 9, 2003. None of our other products have FDA
approval for marketing in the United States. However, we believe that our products, including for example, GlamSmile,
do not require a 510(k) submission because the products fall within an exemption under the 510(k) regulation.
International sales of medical devices are
also subject to the regulatory requirements of each country. In Europe, the regulations of the European Union require that a device
have a CE Mark, a mark that indicates conformance with European Union laws and regulations before it can be sold in that market.
In China, the SFDA requires registration of medical devices. The regulatory international review process varies from country to
country. We rely upon our distributors, sales representatives and strategic partners in the foreign countries in which we market
our products to ensure we comply with the regulatory laws of such countries. Failure to comply with the laws of such country will
have a material adverse effect on our operations and, at the very least, could prevent us from continuing to sell products in such
countries.
We may not have effective internal controls if we fail to
remedy any
deficiencies we may identify in our system of internal controls.
In connection with Section 404 of the
Sarbanes-Oxley Act of 2002, we need to assess the adequacy of our internal control, remediate any weaknesses that may be identified,
validate that controls are functioning as documented and implement a continuous reporting and improvement process for internal
controls. We may discover deficiencies that require us to improve our procedures, processes and systems in order to ensure that
our internal controls are adequate and effective and that we are in compliance with the requirements of Section 404 of the Sarbanes-Oxley
Act. If the deficiencies are not adequately addressed, or if we are unable to complete all of our testing and any remediation in
time for compliance with the requirements of Section 404 of the Sarbanes-Oxley Act and the SEC rules under it, we would be unable
to conclude that our internal controls over financial reporting are designed and operating effectively, which could adversely affect
our investor confidence in our internal controls over financial reporting.
The loss of or a substantial reduction in, or change in the
size or timing of,
orders from distributors could harm our business
.
Our international sales are principally
comprised of sales through independent distributors, although we sell products in certain European countries through direct sales
representatives. A significant amount of our sales may consist of sales through distributors. The loss of
a substantial number of our distributors or a substantial reduction in, cancellation of or change in the size or timing of orders
from our current distributors could harm our business, financial condition and results of operations. The loss of a
key distributor would affect our operating results due to the potential length of time that might be required to locate and qualify
a new distributor or to retain direct sales representatives for the territory.
We do not have long term commitments from our suppliers and
manufacturers.
We may experience shortages of supplies
and inventory because we do not have long-term agreements with our suppliers or manufacturers. Our success is dependent
on our ability to provide our customers with our products. Although we manufacture most of our products, we are dependent
on our suppliers for component parts which are necessary for our manufacturing operations. In addition, certain of our
present and future products and product components are (or will be) manufactured by third party manufacturers. Since
we have no long-term contracts or other contractual assurances with these manufacturers for continued supply, pricing or access
to component parts, no assurance can be given that such manufacturers will continue to supply us with adequate quantities of products
at acceptable levels of quality and price. While we believe that we have good relationships with our suppliers and our
manufacturers, if we are unable to extend or secure manufacturing services or to obtain component parts or finished products from
one or more manufacturers on a timely basis and on acceptable terms, our results of operations could be adversely affected.
We face intense competition, and many of our competitors
have substantially
greater resources than we do.
We operate in a highly competitive environment.
There are numerous well-established companies and smaller entrepreneurial companies with significant resources who are
developing and marketing products and services that will compete with our products. In addition, many of our current
and potential competitors have greater financial, technical, operational and marketing resources. These resources may
make it difficult for us to compete with them in the development and marketing of our products, which could harm our business.
Our success will depend on our ability to update our technology
to remain
competitive.
The dental device and supply industry is
subject to technological change. As technological changes occur in the marketplace, we may have to modify our products
in order to become or remain competitive. While we are continuing our research and development in new products in efforts
to strengthen our competitive advantage, no assurances can be given that we will successfully implement technological improvements
to our products on a timely basis, or at all. If we fail to anticipate or respond in a cost-effective and timely manner
to government requirements, market trends or customer demands, or if there are any significant delays in product development or
introduction, our revenues and profit margins may decline which could adversely affect our cash flows, liquidity and operating
results.
We depend on market acceptance of the products of our customers. If
our
products do not gain market acceptance, our ability to compete will be
adversely affected.
Our success will depend in large part on
our ability to successfully market our line of products. Although we intend to differentiate our products from our competitors
by targeting different channels of distribution, no assurances can be given that we will be able to successfully market our products
or achieve consumer acceptance. Moreover, failure to successfully develop, manufacture and commercialize our products
on a timely and cost-effective basis will have a material adverse effect on our ability to compete in our targeted market segments. In
addition, medical and dental insurance policies generally do not cover teeth whitening or other cosmetic dental procedures, including
our products, which may have an adverse impact upon the market acceptance of our products.
We may be exposed to liabilities under the Foreign Corrupt
Practices Act, and any determination that we violated the Foreign Corrupt Practices Act could have a material adverse effect on
our business.
To the extent that we operate outside
the United States, we are subject to the Foreign Corrupt Practices Act (the “FCPA”) which generally prohibits U.S. companies
and their intermediaries from bribing foreign officials for the purpose of obtaining or keeping business or otherwise obtaining
favorable treatment. In particular, we may be held liable for actions taken by our strategic or local partners even though such
partners are foreign companies that are not subject to the FCPA. Any determination that we violated the FCPA could result in sanctions
that could have a material adverse effect on our business.
Failure to meet customers’ expectations or deliver
expected performance of our
products could result in losses and negative publicity, which will harm our
business
.
If our products fail to perform in
the manner expected by our customers, then our revenues may be delayed or lost due to adverse customer reaction, negative publicity
about us and our products, which could adversely affect our ability to attract or retain customers. Furthermore, disappointed
customers may initiate claims for substantial damages against us, regardless of our responsibility for such failure.
If product liability lawsuits are successfully brought against
us, we may incur
substantial liabilities and may be required to limit commercialization of our
products.
Although we have not been a party to any
product liability lawsuits and are currently not aware of any anticipated product liability claims with respect to our products,
the nature of our business exposes us to product liability lawsuits arising out of the commercialization of our products. In
the future, an individual may bring a liability claim against us if one of our products causes, or merely appears to have caused,
an injury. If we cannot successfully defend ourselves against the product liability claim, we may incur substantial
liabilities. Regardless of merit or eventual outcome, liability claims may result in:
|
|
•
|
decreased demand for our products;
|
|
|
|
|
|
|
•
|
injury to our reputation;
|
|
|
|
|
|
|
•
|
costs of related litigation;
|
|
|
|
|
|
|
•
|
substantial monetary awards to customers;
|
|
|
|
|
|
|
•
|
product recalls;
|
|
|
|
|
|
|
•
|
loss of revenue; and
|
|
|
|
|
|
|
•
|
the inability to commercialize our products.
|
We may have difficulty managing our growth.
We have been experiencing significant growth
in the scope of our operations, including the launch of our retail direct to consumer business model in Asia through strategic
partnerships. This growth has placed significant demands on our management as well as our financial and operational
resources. In order to achieve our business objectives, we anticipate that we will need to continue to grow. If
this growth occurs, it will continue to place additional significant demands on our management and our financial and operational
resources, and will require that we continue to develop and improve our operational, financial and other internal controls. We
have been distributing our products primarily in Europe and we have recently launched sales and distribution in the United States
and Asia, this expansion could further increase the challenges involved in implementing appropriate operational and financial systems,
expanding manufacturing capacity and scaling up production, expanding our sales and marketing infrastructure and capabilities and
providing adequate training and supervision to maintain high quality standards. The main challenge associated with our
growth has been, and we believe will continue to be, our ability to recruit and integrate skilled sales, manufacturing and management
personnel. Our inability to scale our business appropriately or otherwise adapt to growth would cause our business,
financial condition and results of operations to suffer.
It may be difficult to enforce a United States judgment against
us, our
officers and directors, or to assert United States securities laws claims in
Belgium and to serve process
on substantially all of our directors and
officers and these experts.
A majority of our directors and our chief
executive officer are nonresidents of the United States. A substantial portion of our assets and all or a substantial
portion of the assets of these officers and directors and experts are located outside of the United States. As a result,
it may be difficult to effect service of process within the United States with respect to matters arising under the United States
securities laws or to enforce, in the United States courts, judgments predicated upon civil liability under the United States securities
laws. It also may be difficult to enforce in Belgium, in original actions or in actions for enforcement of judgment
of United States courts, civil liabilities predicated upon United States securities laws.
If we are unable to protect our intellectual property rights
or our
intellectual property rights are inadequate, our competitive position could be
harmed or we could be required
to incur expenses to enforce our rights
.
Our future success will depend, in
part, on our ability to obtain and maintain patent protection for our products and technology, to preserve our trade secrets and
to operate without infringing the intellectual property of others. In part, we rely on patents to establish and maintain
proprietary rights in our technology and products. While we hold licenses to a number of issued patents and have other
patent applications pending on our products and technology, we cannot assure you that any additional patents will be issued, that
the scope of any patent protection will be effective in helping us address our competition or that any of our patents will be held
valid if subsequently challenged. Other companies also may independently develop similar products, duplicate our products
or design products that circumvent our patents.
In addition, if our intellectual property
rights are inadequate, we may be exposed to third-party infringement claims against us. Although we have not been a
party to any infringement claims and are currently not aware of any anticipated infringement claim, we cannot predict whether third
parties will assert claims of infringement against us, or whether any future claims will prevent us from operating our business
as planned. If we are forced to defend against third-party infringement claims, whether they are with or without merit
or are determined in our favor, we could face expensive and time-consuming litigation. If an infringement claim is determined
against us, we may be required to pay monetary damages or ongoing royalties. In addition, if a third party successfully
asserts an infringement claim against us and we are unable to develop suitable non-infringing alternatives or license the infringed
or similar intellectual property on reasonable terms on a timely basis, then our business could suffer.
If we are unable to meet customer demand or comply with quality
regulations,
our sales will suffer
.
We manufacture many of our products
at our Deurle, Belgium production facilities. In order to achieve our business objectives, we will need to significantly
expand our manufacturing capabilities to produce the systems and accessories necessary to meet demand. We may encounter
difficulties in scaling-up production of our products, including problems involving production capacity and yields, quality control
and assurance, component supply and shortages of qualified personnel. In addition, our manufacturing facilities are
subject to periodic inspections by foreign regulatory agencies. Our success will depend in part upon our ability to
manufacture our products in compliance with regulatory requirements. Our business will suffer if we do not succeed in
manufacturing our products on a timely basis and with acceptable manufacturing costs while at the same time maintaining good quality
control and complying with applicable regulatory requirements.
We are dependent on Guy De Vreese, our Chairman and Chief
Executive Officer, and any loss of such key personnel could result in the loss
of a significant portion of our business.
Our success is highly dependent upon
the key business relations and expertise of Guy De Vreese, our Chairman and Chief Executive Officer. Unlike larger companies,
we rely heavily on a small number of officers to conduct a large portion of our business. The loss of service of our
Chairman and Chief Executive Officer along with the loss of his numerous contacts and relationships in the industry would have
a material adverse effect on our business. We do not have an employment agreement with Guy De Vreese.
If we cannot build and maintain strong brand loyalty our
business may suffer.
We believe that the importance of brand
recognition will increase as more companies produce competing products. Development and awareness of our brands will
depend largely on our ability to advertise and market successfully. If we are unsuccessful, our brands may not be able
to gain widespread acceptance among consumers. Our failure to develop our brands sufficiently would have a material
adverse effect on our business, results of operations and financial condition.
Risks Relating To Our Common Stock
There is a limited public trading market for our common stock.
Our Common Stock presently trades on
the Over-the-Counter Bulletin Board under the symbol “REMI.” We cannot assure you, however, that such market will continue
or that you will be able to liquidate your shares acquired in this offering at the price you paid or otherwise. We also
cannot assure you that any other market will be established in the future. The price of our common stock may be highly
volatile and your liquidity may be adversely affected in the future.
Our common stock is thinly traded, so you may be unable to
sell at or near ask prices or at all if you need to sell your shares to raise money or otherwise desire to liquidate your shares.
There is limited market activity in our
stock and we are too small to attract the interest of many brokerage firms and analysts. We cannot give you any assurance that
a broader or more active public trading market for our common stock will develop or be sustained. While we are trading on the Over-The-Counter
Bulletin Board, our trading volume may be limited by the fact that many major institutional investment funds, including mutual
funds, as well as individual investors follow a policy of not investing in Over- the–Counter Bulletin Board stocks and certain
major brokerage firms restrict their brokers from recommending Over-the-Counter Bulletin Board stocks because they are considered
speculative, volatile, thinly traded and the market price of the common stock may not accurately reflect our underlying value.
The market price of our common stock could be subject to wide fluctuations in response to quarterly variations in our revenues
and operating expenses, announcements of new products or services by us, significant sales of our common stock, the operating and
stock price performance of other companies that investors may deem comparable to us, and news reports relating to trends in our
markets or general economic conditions.
The ownership of our stock is highly concentrated in our
management.
As of June 25, 2012, our present
directors and executive officers, and their respective affiliates beneficially owned approximately 29% of our outstanding common
stock, including underlying options that were exercisable or which would become exercisable within 60 days. As a result
of their ownership, our directors and executive officers and their respective affiliates collectively are able to significantly
influence all matters requiring stockholder approval, including the election of directors and approval of significant corporate
transactions. This concentration of ownership may also have the effect of delaying or preventing a change in control.
We have a substantial number of shares authorized but not
yet issued.
Our Articles of Incorporation authorize
the issuance of up to 50,000,000 shares of common stock and 10,000,000 shares of preferred stock. Our Board of Directors
has the authority to issue additional shares of common stock and preferred stock and to issue options and warrants to purchase
shares of our common stock and preferred stock without stockholder approval. Future issuance of common stock and preferred
stock could be at values substantially below current market prices and therefore could represent further substantial dilution to
our stockholders. In addition, the Board could issue large blocks of voting stock to fend off unwanted tender offers
or hostile takeovers without further stockholder approval.
We have historically not paid dividends and do not intend
to pay dividends.
We have historically not paid dividends
to our stockholders and management does not anticipate paying any cash dividends on our common stock to our stockholders for the
foreseeable future. We intend to retain future earnings, if any, for use in the operation and expansion of our business.
Our stock may be governed by the “penny stock rules,”
which impose additional
requirements on broker-dealers who make transactions in our stock.
SEC rules require a broker-dealer to provide
certain information to purchasers of securities traded at less than $5.00, which are not traded on a national securities exchange. Since
our common stock is not currently traded on an exchange, our common stock is considered a “penny stock,” and trading
in our common stock is subject to the requirements of Rules 15g-1 through 15g-9 under the Securities Exchange Act of 1934 (the
“Penny Stock Rules”). The Penny Stock Rules require a broker-dealer to deliver a standardized risk disclosure
document prepared by the SEC that provides information about penny stocks and the nature and level of risks in the penny stock
market. The broker-dealer must also give bid and offer quotations and broker and salesperson compensation information
to the prospective investor orally or in writing before or with the confirmation of the transaction. In addition, the
Penny Stock Rules require a broker-dealer to make a special written determination that the penny stock is a suitable investment
for the purchaser and receive the purchaser’s written agreement to the transaction before a transaction in a penny stock. These
requirements may severely limit the liquidity of securities in the secondary market because few broker-dealers may be likely to
undertake these compliance activities. Therefore, the disclosure requirements under the Penny Stock Rules may have the
effect of reducing trading activity in our common stock, which may make it more difficult for investors to sell their shares.
ITEM 1B — UNRESOLVED STAFF COMMENTS
Not Applicable.
ITEM 2 — PROPERTIES
The Company leases
an office facility of 5,187 square feet in Gent, Belgium from an unrelated party pursuant to a nine year lease commencing September
1, 2008. at a base rent of €5,712 per month for the total location ($7,624 per month at March 31, 2012).
The Company leases
an office facility of 1,991 square feet in Rome, Italy to support the sales and marketing division of our veneer business, from
an unrelated party pursuant a six year lease commencing July 1, 2011, at a base rent of € 6,500 per month for the total location
($8,675 per month at March 31, 2012).
ITEM 3 — LEGAL PROCEEDINGS
To the best knowledge of management,
there are no material legal proceedings pending against the Company.
ITEM 4 — [RESERVED AND REMOVED]
PART II
ITEM 5 — MARKET FOR REGISTRANT’S COMMON EQUITY,
RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Market Information
Our common stock is quoted on the Over
the Counter Bulletin Board under the symbol “REMI.” The following table shows the range of the high and
low bid for our common stock as reported by the Over-The-Counter Bulletin Board for the time periods indicated:
|
|
|
Bid Prices
|
|
|
|
|
High
|
|
|
Low
|
|
|
Quarter ended June 30, 2010
|
|
$
|
0.40
|
|
|
$
|
0.30
|
|
|
Quarter ended September 30, 2010
|
|
$
|
0.40
|
|
|
$
|
0.24
|
|
|
Quarter ended December 31, 2010
|
|
$
|
0.60
|
|
|
$
|
0.25
|
|
|
Quarter ended March 31, 2011
|
|
$
|
0.75
|
|
|
$
|
0.31
|
|
|
Quarter ended June 30, 2011
|
|
$
|
0.47
|
|
|
$
|
0.32
|
|
|
Quarter ended September 30, 2011
|
|
$
|
0.45
|
|
|
$
|
0.19
|
|
|
Quarter ended December 31, 2011
|
|
$
|
0.30
|
|
|
$
|
0.18
|
|
|
Quarter ended March 31, 2012
|
|
$
|
0.50
|
|
|
$
|
0.16
|
|
Bid quotations represent interdealer
prices without adjustment for retail markup, markdown and/or commissions and may not necessarily represent actual transactions.
Stockholders
As of June 25, 2012, the number of
stockholders of record was approximately 188, not including beneficial owners whose shares are held by banks, brokers and other
nominees.
Dividends
We have not paid any dividends on our
common stock, and we do not anticipate paying any dividends in the foreseeable future. Our Board of Directors intends to follow
a policy of retaining earnings, if any, to finance the growth of the company. The declaration and payment of dividends in the future
will be determined by our Board of Directors in light of conditions then existing, including our earnings, financial
condition, capital requirements and other factors.
Securities Authorized for Issuance under Equity Compensation
Plans
As of March 31, 2012, we had three equity
compensation plans approved by our stockholders (1) our Incentive and Nonstatutory Stock Option Plan enacted in 2001 (the “2001
Plan”), (2) our 2004 Incentive and Nonstatutory Stock Option Plan (the “2004 Plan”); and (3) our 2007 Equity
Incentive Plan (the “2007 Plan”). Our stockholders approved the 2001 Plan reserving 250,000 shares of common stock
of the Company pursuant an Information Statement on Schedule 14C filed with the Commission on August 15, 2001. In addition, our
stockholders approved the 2004 Plan reserving 800,000 shares of common stock of the Company pursuant to an Information Statement
on Schedule 14C filed with the Commission on May 9, 2005. Finally, our stockholders approved the 2007 Plan reserving
1,000,000 shares of common stock of the Company pursuant to a Definitive Proxy Statement on Schedule 14A filed with the Commission
on October 2, 2007.
In addition to the equity compensation plans
approved by our stockholders, we have issued options and warrants to individuals pursuant to individual compensation plans not
approved by our stockholders. These options and warrants have been issued in exchange for services or goods received
by us.
The following table
provides aggregate information as of March 31, 2012 and March 31, 2011 with respect to all compensation plans (including individual
compensation arrangements) under which equity securities are authorized for issuance.
|
|
|
2001 Plan
|
|
|
2004 Plan
|
|
|
2007 Plan
|
|
|
Other
|
|
|
|
|
Outstanding
Options
|
|
|
Weighted
Average
Exercise
Price
|
|
|
Outstanding
Options
|
|
|
Weighted
Average
Exercise
Price
|
|
|
Outstanding
Options
|
|
|
Weighted
Average
Exercise
Price
|
|
|
Outstanding
Options
|
|
|
Weighted
Average
Exercise
Price
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Options outstanding, March 31, 2010
|
|
|
250,000
|
|
|
|
1.20
|
|
|
$
|
668,166
|
|
|
|
0.89
|
|
|
$
|
1,000,000
|
|
|
|
1.15
|
|
|
$
|
350,000
|
|
|
$
|
0.97
|
|
|
Cancelled or expired
|
|
|
—
|
|
|
|
—
|
|
|
|
(110,666
|
)
|
|
|
4.00
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Options outstanding, March 31, 2011
|
|
|
250,000
|
|
|
|
1.20
|
|
|
|
557,500
|
|
|
|
0.89
|
|
|
|
1,000,000
|
|
|
|
1.15
|
|
|
|
350,000
|
|
|
|
0.97
|
|
|
Cancelled or expired
|
|
|
(237,500
|
)
|
|
|
—
|
|
|
|
(25,000
|
)
|
|
|
4.00
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Options outstanding, March 31, 2012
|
|
|
12,500
|
|
|
|
1.19
|
|
|
|
532,500
|
|
|
|
0.96
|
|
|
|
1,000,000
|
|
|
|
1.15
|
|
|
|
350,000
|
|
|
|
0.97
|
|
|
Options exercisable March 31, 2012
|
|
|
12,500
|
|
|
|
1.19
|
|
|
|
532,500
|
|
|
|
0.96
|
|
|
|
1,000,000
|
|
|
|
1.15
|
|
|
|
350,000
|
|
|
|
0.97
|
|
|
Exercise price range
|
|
$
|
2.00
|
|
|
|
|
|
|
$
|
0.50 - $2.46
|
|
|
|
|
|
|
$
|
0.50 - $1.75
|
|
|
|
|
|
|
$
|
.39 - 1.75
|
|
|
|
|
|
|
Weighted average remaining life
|
|
|
2 years
|
|
|
|
|
|
|
|
5.28 years
|
|
|
|
|
|
|
|
6.1 years
|
|
|
|
|
|
|
|
4.6 years
|
|
|
|
|
|
A summary of the Company’s equity compensation plans approved
and not approved by shareholders is as follows:
|
Plan Category
|
|
Number of
securities to
be
issued upon
exercise of
of
outstanding
options,
warrants
and rights
|
|
|
Weighted-average
exercise price of
outstanding
options
warrants and
rights
|
|
|
Number of
securities
remaining
available for
future
issuance
under
equity
compensation
plans
(excluding
securities
reflected
in column (a))
|
|
|
Equity Compensation Plans approved by security holders
|
|
|
1,545,000
|
|
|
$
|
1.13
|
|
|
|
505,000
|
|
|
Equity Compensation Plans not approved by security holders
|
|
|
820,000
|
|
|
$
|
.97
|
|
|
|
NA
|
|
|
Total
|
|
|
2,365,000
|
|
|
$
|
1.08
|
|
|
|
505,000
|
|
For the year ended March 31, 2012, the
Company recognized $50,387 (2011 — $113,682) in compensation expense in the consolidated statement of operations.
ITEM 6 — SELECTED FINANCIAL DATA
Not Applicable.
ITEM 7 — MANAGEMENT’S DISCUSSION AND ANALYSIS
OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
In addition to historical information,
this section contains
“forward-looking” statements, including statements regarding the growth of
product
lines, optimism regarding the business, expanding sales and other
statements. Words such as expects, anticipates, intends,
plans, believes, sees,
estimates and variations of such words and similar expressions are intended to
identify such
forward-looking statements. These statements are not guarantees
of future performance and involve certain risks and uncertainties
that are
difficult to predict. Actual results could vary materially from the description
contained herein due to
many factors including continued market acceptance of
our products. In addition, actual results could vary materially based
on
changes or slower growth in the oral care and cosmetic dentistry products
market; the potential inability to realize
expected benefits and synergies;
domestic and international business and economic conditions; changes in the
dental
industry; unexpected difficulties in penetrating the oral care and
cosmetic dentistry products market; changes in customer
demand or ordering
patterns; changes in the competitive environment including pricing pressures or
technological
changes; technological advances; shortages of manufacturing
capacity; future production variables impacting excess inventory
and other risk
factors listed in the section of this Annual Report entitled “Risk Factors” and from time to
time in our Securities and Exchange Commission
filings under “risk factors” and elsewhere.
Each forward-looking statement should
be read in context with, and with an
understanding of, the various disclosures concerning our business made
elsewhere
in this Annual Report, as well as other public reports filed by us
with the Securities and Exchange Commission. Readers
should not place undue
reliance on any forward-looking statement as a prediction of actual results of
developments.
Except as required by applicable law or regulation, we undertake
no obligation to update or revise any forward-looking statement
contained in
this Annual Report. This section should be read in conjunction with our
consolidated financial statements.
Overview
We design, develop, manufacture and distribute
cosmetic dentistry products. Leveraging our knowledge of regulatory requirements regarding dental products and management’s
experience in the needs of the professional dental community, we have developed a line of professional veneers as well as a family
of teeth whitening products for both professional and “Over-The-Counter” (“OTC”) use, that are distributed
in Europe, Asia and the United States. We manufacture many of our products in our facility in Deurle, Belgium as well
as outsourced manufacturing in China and France. We distribute our products using both our own internal sales force and through
the use of third party distributors. We have established dealers in 28 countries encompassing, Europe, Asia, Latin America,
the Pacific Rim and the Middle East.
Recent Developments
On June 3, 2011, the Company obtained a
loan in the principal amount of $1,000,000 (the “Loan”) from an unrelated private company, Excelsior Medical (HK) (“EM”).
In connection with the Loan, the Company issued a promissory note, with a simple interest rate of 5% per annum, secured by certain
assets of the Company (the “Note”). The maturity date of the Loan is June 3, 2014. Interest of $50,000 per annum is
payable in cash on an annual basis. In September 2010, we entered into a license agreement with EM (the “EM license agreement”).
Under the EM license agreement, we granted EM an exclusive license to certain Asian territories in exchange for $500,000 which
was received during the year ended March 31, 2011. The Company received a further $500,000 from EM as an advance payment for veneers. The
$500,000 advance, less taxes withheld, was recorded as deferred revenue of $475,250 as of March 31, 2011. Effective as of
January 11, 2012, the Company entered into a Rescission Agreement with EM and Asia Best Healthcare Co., Ltd. Under the Rescission
Agreement, the Company agreed to repay a total of $1,000,000 received under the Distribution Agreement, plus a simple interest
rate of 5%, beginning on June 30, 2012, according to the following payment schedule: (i) $250,000 to be paid no later than June
30, 2012, (ii) $250,000 plus interest on June 30, 2012, (iii) $250,000 plus interest on December 31, 2012, and (iv) $250,000
plus interest on June 30, 2013. The Company also agreed to secure such obligations owed to EM with certain collateral of the Company
On January 28, 2012, we entered into a Preference
A Shares and Preference A-1 Shares Purchase Agreement (“Share Purchase Agreement”) with Glamsmile Dental Technology
Ltd., a Cayman Islands company and a subsidiary of Company (“Glamsmile Dental”), Glamsmile (Asia) Limited, a company
organized and existing under the laws of Hong Kong and a substantially owned subsidiary of Glamsmile Dental, Beijing Glamsmile
Technology Development Ltd., Beijing Glamsmile Trading Co., Ltd., Beijing Glamsmile Dental Clinic Co., Ltd., and Shanghai Glamsmile
Dental Clinic Co., Ltd., Gallant Network Limited, a shareholder of Glamsmile Dental (“Gallant”), and IDG-Accel China
Growth Fund III L.P. (“IDG Growth”), IDG-Accel China III Investors L.P.(“IDG Investors”) and Crown Link
Group Limited (“Crown”)(“IDG Growth, IDG Investors and Crown collectively referred to as the “Investors”),
pursuant to which the Investors agreed to (i) purchase from the Company an aggregate of 2,857,143 shares of Preference A-1 Shares
of Glamsmile Dental, which represents all of the issued and outstanding Preference A-1 Shares of Glamsmile Dental, for an aggregate
purchase price of $2,000,000, and (ii) purchase from Glamsmile Dental an aggregate of 5,000,000 shares of Preference A Shares for
an aggregate purchase price of $5,000,000.
On February 10, 2012, the sale of the Preference
A-1 Shares and the Preference A Shares was completed. As a result of the closing, the equity ownership of Glamsmile Dental, on
an as converted basis, is as follows: 31.4% by the Investors, 39.2 % by Gallant, and 29.4% by the Company. Mr. De Vreese, our chairman,
will remain as a director of Glamsmile Dental along with Mr. David Lok, who is the Chief Executive Officer and director of Glamsmile
Dental and principal of Gallant. In addition, at the closing, the Investors have a right to appoint one director of Glamsmile Dental,
and as such it is contemplated that after the closing the Board of Directors of Glamsmile Dental will consists of Mr. De Vreese,
Mr. Lok and a director appointed by the Investors.
Under
the terms of the Share Purchase Agreement, we agreed to transfer 500,000 shares of Glamsmile Dental owned by the Company to the
Investors in the event of breach of certain covenants by the Company. In connection with the Share Purchase Agreement, we also
entered into an Investor’s Rights Agreement, Right of First Refusal and Co-Sale Agreement, and Voting Agreement with the
parties. In addition, in connection with the contemplated transactions in the Share Purchase Agreement on January 20, 2012, we
entered into a Distribution, License and Manufacturing Agreement with Glamsmile Dental pursuant to which we appointed Glamsmile
Dental as the exclusive distributor and licensee of Glamsmile Veneer Products bearing the “Glamsmile” name and mark
in the B2C Market in the People’s Republic of China (including Hong Kong and Macau) and Republic of China (Taiwan) and granted
related manufacturing rights and licenses in exchange for the original issuance of 2,857,143 shares of Preference A-1 Shares of
Glamsmile Dental and $250,000 (the receipt of which was acknowledged as an off set to payment of certain invoices of Glamsmile
(Asia) Limited).
During the fiscal year ended March 31, 2012,
we opened a new Glamsmile Studio in Rome, Italy. In addition, we are developing a ‘prefab’ veneer which would be a
‘ready-to-use’ veneer, called ‘River 8’ offered at a lower price than traditional veneers. It is anticipated
that the launch will be during the first six months of the next fiscal year.
Financial Results and Trends
Revenue decreased by approximately 23% to
$9,687,292 in the year ended March 31, 2012 as compared to $12,581,708 in the year ended March 31, 2011. In fiscal 2012,
we experienced a decline in revenues as a result of the deconsolidation of our OTC division Sylphar at the quarter ending September
2011 and secondly, the deconsolidation of our Asian Division at the end of January 2012 resulting in decreased revenue for both
entities. The equity method is currently being used in calculating the monthly results, whereas, in fiscal 2011, the reported revenue
stands for a full years revenue for all divisions.
Our net income/( loss) attributable to our
stockholders was $0.02 for fiscal 2012 and $(0.08) for fiscal 2011. Our net income for fiscal 2012 was positively impacted by a
gain of $2,085,669 on the sale of our Asian division as explained in the heading “Recent Developments” above and the
sale of assets and IP by Remedent N.V. to its Asian Subsidiary for $ 601,391. We also recognized $173,337 in equity income from
our investments in the year ended March 31, 2012, namely our investment in the OTC Division and our Asian Veneer Division.
Our net income for fiscal 2011 was negatively
impacted by an impairment loss of $750,000 for Soca Networks Singapore (“Soca”), an investment made in a production
facility to produce our GlamSmile Veneers.
Critical Accounting Estimates
Basis for Presentation
Our financial statements have been prepared
on an accrual basis of accounting, in conformity with accounting principles generally accepted in the United States of America.
Pervasiveness of Estimates
The preparation of financial statements
in conformity with accounting principles generally accepted in the United States requires the Company to make estimates and assumptions
that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the
financial statements and the reported amounts of revenues and expenses during the reporting period. On an on-going basis, the Company
evaluates estimates and judgments, including those related to revenue, bad debts, inventories, fixed assets, intangible assets,
stock based compensation, income taxes, and contingencies. Estimates are based on historical experience and on various other assumptions
that the Company believes reasonable in the circumstances. The results form the basis for making judgments about the carrying values
of assets and liabilities that are not readily apparent from other sources. Actual results could differ from those estimates.
Revenue Recognition
The Company recognizes revenue from product
sales when persuasive evidence of a sale exists: that is, a product is shipped under an agreement with a customer; risk of loss
and title has passed to the customer; the fee is fixed or determinable; and collection of the resulting receivable is reasonably
assured. Sales allowances are estimated based upon historical experience of sales returns.
Revenues from product sales are recognized
when the product is shipped and title and risk of loss has passed to the customer, typically upon delivery and when the quantity
and price is fixed and determinable, and when collectability is reasonable assured.
Upfront fees are recognized upon the date
of the agreement (i.e. point of sale) because they relate solely to the sale of territories (that are sold in perpetuity), are
non-refundable, and are not contingent upon additional deliverables.
We have evaluated all deliverables in our
contracts (per ASC 605-25-5) ((a) territory & (b) manufacturing/marketing training & development fees) and determined that
they are separate, as follows:
|
|
·
|
Both (a) & (b) have value to our customers on a standalone basis and can be sold by our customers separately.
|
|
|
·
|
Delivery or performance of the undelivered item or items is considered probable and substantially in our control.
|
Our development fees/milestone payments
are recognized in accordance with the Milestone Method pursuant to FASB ASC 605. Revenues from milestones related to
an arrangement under which we have continuing performance obligations i.e. specifically scheduled training and development activities,
if deemed substantive, are recognized as revenue upon achievement of the milestone. Milestones are considered substantive if all
of the following conditions are met: (a) the milestone is non-refundable; (b) achievement of the milestone was not reasonably assured
at the inception of the arrangement; (c) substantive effort is involved to achieve the milestone; and (d) the amount of the milestone
appears reasonable in relation to the effort expended. If any of these conditions is not met, the milestone payment is deferred
and recognized as revenue as we complete our performance obligations.
We receive royalty revenues under license
agreements with third parties that sell products based on technology developed by us or to which we have rights. The license agreements
provide for the payment of royalties to us based on sales of the licensed product. We record these revenues as earned monthly,
based on reports from our licensees.
Goodwill
Goodwill is not amortized, but is tested
for impairment on an annual basis as of March 31, or whenever events or circumstances indicate that the carrying amount may not
be recoverable. These impairment tests are based upon a comparison of the fair value of the reporting units to their respective
carrying amount. If the carrying amount of the reporting unit exceeds its fair value, the goodwill impairment loss is measured
as the excess of the carrying amount of goodwill over its implied fair value. As of March 31, 2012 the goodwill is no longer recorded
because the Asia branch is no longer fully consolidated. Furthermore the goodwill is retained in the gain on sale calculation .
On April 1, 2010, the Company adopted the
new accounting guidance for business combinations according to FASB Codification ASC 805,
Business Combinations
. This guidance
establishes principles and requirements for the reporting entity in a business combination, including recognition and measurement
in the financial statements of the identifiable assets acquired, the liabilities assumed, goodwill, and any noncontrolling interest
in the acquiree, as well as disclosure requirements to enable financial statement users to evaluate the nature and financial effects
of the business combination. Additionally, it provides guidance for identifying a business combination, measuring the acquisition
date, and defining the measurement period for adjusting provisional amounts recorded. The implementation of this standard did not
have an impact on the Company’s consolidated financial statements.
Cash and Cash Equivalents
The Company considers all highly liquid
investments with maturities of three months or less at the time of purchase to be cash or cash equivalents.
Non-Controlling Interest
The Company adopted ASC Topic 810
Noncontrolling
Interests in Consolidated Financial Statements
— an Amendment of Accounting Research Bulletin No. 51
as of
April 1, 2009. SFAS 160 establishes accounting and reporting standards for ownership interests in subsidiaries held by parties
other than the parent, the amount of consolidated net income attributable to the parent and to the noncontrolling interest, changes
in a parent’s ownership interest and the valuation of retained noncontrolling equity investments when a subsidiary is deconsolidated.
ASC Topic 810 also establishes reporting requirements that provide sufficient disclosures that clearly identify and distinguish
between the interest of the parent and the interests of the noncontrolling owner. The adoption of ASC Topic 810 impacted the presentation
of our consolidated financial position, results of operations and cash flows.
Fair Value of Financial Instruments
The Company’s financial instruments
consist of cash and cash equivalents, accounts receivable, accounts payable, accrued liabilities, line of credit, short term and
long-term debt. The carrying amounts of cash and cash equivalents, accounts receivable, accounts payable and accrued liabilities
approximate their respective fair values because of the short maturities of those instruments. The Company’s long-term debt
consists of its revolving credit facility and long-term capital lease obligations. The carrying value of the revolving credit facility
approximates fair value because of its variable short-term interest rates. The fair value of the Company’s long-term
capital lease obligations is based on current rates for similar financing.
Accounts Receivable and Allowance for
Doubtful Accounts
The Company sells professional dental equipment to various companies,
primarily to distributors located in Western Europe and the United States of America, and China. The terms of sales vary by customer,
however, generally are 2% 10 days, net 30 days. Accounts receivable is reported at net realizable value and net of allowance for
doubtful accounts. The Company uses the allowance method to account for uncollectible accounts receivable. The Company’s
estimate is based on historical collection experience and a review of the current status of trade accounts receivable.
Inventories
The Company purchases certain of its products in components
that require assembly prior to shipment to customers. All other products are purchased as finished goods ready to ship to customers.
The Company writes down inventories for estimated obsolescence
to estimated market value based upon assumptions about future demand and market conditions. If actual market conditions are less
favorable than those projected, then additional inventory write-downs may be required. Inventory reserves for obsolescence totaled
$156,239 at March 31, 2012 and $130,407 at March 31, 2011.
Prepaid Expense
The Company’s prepaid expense consists of prepayments
to suppliers for inventory purchases and to the Belgium customs department, to obtain an exemption of direct VAT payments for imported
goods out of the European Union (“EU”). This prepayment serves as a guarantee to obtain the facility to pay VAT at
the moment of sale and not at the moment of importing goods at the border. Prepaid expenses also include VAT payments made for
goods and services in excess of VAT payments received from the sale of products as well as amounts for other prepaid operating
expenses.
Property and Equipment
Property and equipment are stated at cost. Major renewals and
improvements are charged to the asset accounts while replacements, maintenance and repairs, which do not improve or extend the
lives of the respective assets, are expensed. At the time property and equipment are retired or otherwise disposed of, the asset
and related accumulated depreciation accounts are relieved of the applicable amounts. Gains or losses from retirements or sales
are credited or charged to income.
The Company depreciates its property and equipment for financial
reporting purposes using the straight-line method based upon the following useful lives of the assets:
|
Tooling
|
3 Years
|
|
Furniture and fixtures
|
4 Years
|
|
Machinery and Equipment
|
4 Years
|
Patents
Patents consist of the costs incurred to purchase patent rights
and are reported net of accumulated amortization. Patents are amortized using the straight-line method over a period based on their
contractual lives.
Research and Development Costs
The Company expenses research and development costs as incurred.
Advertising
Costs incurred for producing and communicating advertising are
expensed when incurred and included in sales and marketing and general and administrative expenses. For the years ended March 31,
2012 and March 31, 2011, advertising expense was $590,285 and 709,885, respectively.
Income taxes
Income taxes are accounted for under the
asset and liability method as stipulated by Accounting Standards Codification (“ASC”) 740 formerly Statement of Financial
Accounting Standards (“SFAS”) No. 109, “Accounting for Income Taxes”. Deferred tax assets and liabilities
are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of
existing assets and liabilities and their respective tax bases and operating loss and tax credit carry forwards. Deferred tax assets
and liabilities are measured using enacted tax rates expected to apply to taxable income in the year in which those temporary differences
are expected to be recovered or settled. Under ASC 740, the effect on deferred tax assets and liabilities or a change in tax rate
is recognized in income in the period that includes the enactment date. Deferred tax assets are reduced to estimated amounts to
be realized by the use of a valuation allowance. A valuation allowance is applied when in management’s view it is more likely
than not (50%) that such deferred tax will not be utilized.
Effective February 1, 2008, the Company
adopted certain provisions under ASC Topic 740, Income Taxes, (“ASC 740”), which provide interpretative guidance for
the financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return. Effective
with the Company’s adoption of these provisions, interest related to the unrecognized tax benefits is recognized in the financial
statements as a component of income taxes. The adoption of ASC 740 did not have an impact on the Company’s financial position
and results of operations.
In the unlikely event that an uncertain
tax position exists in which the Company could incur income taxes, the Company would evaluate whether there is a probability that
the uncertain tax position taken would be sustained upon examination by the taxing authorities. Reserves for uncertain tax position
would then be recorded if the Company determined it is probable that a position would not be sustained upon examination or if a
payment would have to be made to a taxing authority and the amount is reasonably estimable. As of March 31, 2012, the Company does
not believe it has any uncertain tax positions that would result in the Company having a liability to the taxing authorities.
Warranties
The Company typically warrants its products
against defects in material and workmanship for a period of 18 months from shipment.
A tabular reconciliation of the Company’s
aggregate product warranty liability for the reporting period is as follows:
|
|
|
Year ended
March 31, 2012
|
|
|
Year ended
March 31, 2011
|
|
|
Product warranty liability:
|
|
|
|
|
|
|
|
|
|
Opening balance
|
|
$
|
21,260
|
|
|
$
|
20,238
|
|
|
Reductions for payments made
|
|
|
|
|
|
|
|
|
|
Accruals for product warranties issued in the period
|
|
|
(2,002
|
)
|
|
|
(11,620
|
)
|
|
Adjustments to liabilities for pre-existing warranties
|
|
|
761
|
|
|
|
12,642
|
|
|
Ending liability
|
|
$
|
20,019
|
|
|
$
|
21,260
|
|
Based upon historical trends and warranties
provided by the Company’s suppliers and sub-contractors, the Company has made a provision for warranty costs of $20,019 and
$21,260 as of March 31, 2012 and March 31, 2011, respectively.
Segment Reporting
“Disclosure About Segments of an Enterprise
and Related Information” requires use of the “management approach” model for segment reporting. The management
approach model is based on the way a company’s management organizes segments within the company for making operating decisions
and assessing performance. Reportable segments are based on products and services, geography, legal structure, management structure,
or any other manner in which management disaggregates a company. The Company’s management considers its business to comprise
one segment for reporting purposes.
Computation of Earnings (Loss) per Share
Basic net income (loss) per common share
is computed by dividing net income (loss) attributable to common stockholders by the weighted average number of shares of common
stock outstanding during the period. Net income (loss) per common share attributable to common stockholders assuming dilution is
computed by dividing net income by the weighted average number of shares of common stock outstanding plus the number of additional
common shares that would have been outstanding if all dilutive potential common shares had been issued.
On April 1, 2009, the Company adopted changes
issued by the FASB to the calculation of earnings per share. These changes state that unvested share-based payment awards that
contain non-forfeitable rights to dividends or dividend equivalents (whether paid or unpaid) are participating securities and shall
be included in the computation of earnings per share pursuant to the two-class method for all periods presented. The adoption of
this change had no impact on the Company’s basic or diluted net loss per share because the Company has never issued any share-based
awards that contain non-forfeitable rights.
At each of March 31, 2012 and 2011, the
Company had 19,995,969, shares of common stock issued and outstanding. At March 31, 2012 and 2011, the Company had 7,794,627
and 7,794,627 warrants outstanding, respectively and 1,895,000 and 2,157,500 options outstanding, respectively. All
outstanding warrants and options were excluded from the computation of earnings per share for the year ended March 31, 2012 because
their effect would have been anti-dilutive.
Further, pursuant to ASC 260-10-50-1(c),
if a fully diluted share calculation was computed for the years ended March 31, 2012 and 2011 respectively, it would have excluded
all warrants and all but 200,000 and 1,165,000 options respectively since the Company’s average share trading price during
the last two year period was less than the exercise price of all other warrants and options.
Conversion of Foreign Currencies
The reporting currency for the consolidated
financial statements of the Company is the U.S. dollar. The functional currency for the Company’s European subsidiaries,
Remedent N.V., Sylphar N.V., GlamSmile Rome and GlamSmile Deutschland GmbH, is the Euro, for Sylphar Asia Ptd Ltd the
Singapore Dollar and for Glamsmile Asia Ltd., and its subsidiaries, the Hong Kong dollar and the Chinese Renmimbi (“RMB”)
for Mainland China. Finally, the functional currency for Remedent Professional, Inc. is the U.S. dollar. The assets and liabilities
of companies whose functional currency is other that the U.S. dollar are included in the consolidation by translating the assets
and liabilities at the exchange rates applicable at the end of the reporting period. The statements of income of such companies
are translated at the average exchange rates during the applicable period. Translation gains or losses are accumulated as a separate
component of stockholders’ equity.
Comprehensive Income (Loss)
Comprehensive income (loss) includes all
changes in equity except those resulting from investments by owners and distributions to owners, including accumulated foreign
currency translation, and unrealized gains or losses on marketable securities.
The Company’s only component of other comprehensive income
is the accumulated foreign currency translation consisting of gains and (losses) of $(846,880) and $(184,890) for the years ended
March 31, 2012 and 2011, respectively. These amounts have been recorded as a separate component of stockholders’ equity (deficit).
Stock Based Compensation
The Company has a stock-based compensation
plan. The Company measures the compensation cost of stock options and other stock-based awards to employees and directors at fair
value at the grant date and recognizes compensation expense over the requisite service period for awards expected to vest.
Except for transactions with employees and
directors, all transactions in which goods or services are the consideration received for the issuance of equity instruments are
accounted for based on the fair value of the consideration received or the fair value of the equity instruments issued, whichever
is more reliably measurable. Additionally, the Company has determined that the dates used to value the transaction are either:
(1) The date at which a commitment for performance
by the counter party to earn the equity instruments is established; or
(2) The date at which the counter party’s
performance is complete.
Recently Issued Accounting Pronouncements
Recently Adopted
In December 2011, the FASB issued ASU 2011-11,
“
Balance Sheet (Topic 210) – Disclosures about offsetting assets and liabilities
”. ASU 2011-11 requires
entities to disclose both gross and net information about both instruments and transactions eligible for offset in the statement
of financial position and instruments and transactions subject to an agreement similar to a master netting arrangement. This scope
would include derivatives, sale and repurchase agreements and reverse sale and repurchase agreements, and securities borrowing
and securities lending arrangements. This standard is applicable for annual reporting periods beginning on or after January 1,
2013, and interim periods within those annual periods. Retrospective disclosures will be required for all periods presented. The
adoption of ASU 2011-11 is not expected to have an impact upon the Company’s consolidated financial statements.
In October 2009, the FASB issued ASU 2009-13,
“Multiple-Deliverable Revenue Arrangements, (amendments to ASC Topic 605, Revenue Recognition)”
(“ASU
2009-13”). ASU 2009-13 requires entities to allocate revenue in an arrangement using estimated selling prices of the delivered
goods and services based on a selling price hierarchy. The amendments eliminate the residual method of revenue allocation and require
revenue to be allocated using the relative selling price method. The standard also expands the disclosure requirements for multiple
deliverable revenue arrangements. ASU 2009-13 should be applied on a prospective basis for revenue arrangements entered into or
materially modified in fiscal years beginning on or after June 15, 2010, which corresponds to the Company’s fiscal year beginning
April 1, 2011. The adoption of ASU 2009-13 has had no impact upon the Company’s consolidated financial statements.
In October 2009, the FASB issued ASU 2009-14,
Certain Revenue Arrangements that Include Software Elements – a consensus of the FASB Emerging Issues Task Force
,
which changes the accounting model for revenue arrangements that include both tangible products and software elements. This new
guidance removes from the scope of the software revenue recognition guidance in ASC 985-605, Software Revenue Recognition, those
tangible products containing software components and non-software components that function together to deliver the tangible product’s
essential functionality. In addition, this guidance requires that hardware components of a tangible product containing software
components always be excluded from the software revenue recognition guidance as well as provides further guidance on determining
which software, if any, relating to the tangible product also would be excluded from the scope of software revenue recognition
guidance. The guidance further identifies specific factors in determining whether the tangible product contains software that works
together with the non-software components of the tangible product to deliver the tangible product’s essential functions.
Guidance is also provided on how a vendor should allocate arrangement consideration to deliverables in an arrangement containing
both tangible products and software. The disclosures mandated in ASU 2009-13 are also required by this new guidance. ASU 2009-14
is effective prospectively for revenue arrangements entered into or materially modified in fiscal years beginning on or after June
15, 2010, which corresponds to the Company’s fiscal year beginning April 1, 2011. The adoption of ASU 2010-14 has had no
impact upon the Company’s consolidated financial statements.
In April 2010, the FASB issued ASU 2010-17
,” Revenue Recognition – Milestone Method”.
The amendments in this Update provide guidance on the criteria
that should be met for determining whether the milestone method of revenue recognition is appropriate. A vendor can recognize consideration
that is contingent upon achievement of a milestone in its entirety as revenue in the period in which the milestone is achieved
only if the milestone meets all criteria to be considered substantive. ASU 2010-17 should be applied on a prospective basis for
revenue arrangements entered into or materially modified in fiscal years beginning on or after June 15, 2010, which corresponds
to the Company’s fiscal year beginning April 1, 2011. The adoption of ASU 2010-17 has had no impact upon the Company’s
consolidated financial statements.
In May 2011, the FASB issued ASU 2011-04
and updated the accounting guidance related to fair value measurements. The updated guidance results in a consistent definition
of fair value and common requirements for measurement of and disclosure about fair value between U.S. GAAP and International Financial
Reporting Standards (IFRS). The updated guidance is effective for the Company beginning in the fourth quarter of fiscal year 2012.
The adoption of ASU 2011-04 has not had a material effect on the Company’s results of operations, financial condition, and
cash flows.
Recent Accounting Pronouncements
Not Yet Adopted
In June 2011, the FASB issued new accounting
guidance on comprehensive income that was amended in December 2011. The objective of this accounting guidance is to improve the
comparability, consistency and transparency of financial reporting and to increase the prominence of items reported in other comprehensive
income. The guidance eliminates the option to present components of other comprehensive income as part of the statement of stockholders’
equity and requires them to be presented in the statement of comprehensive income instead. This accounting guidance, as amended,
will be effective for the Company on April 1, 2012. The Company does not expect the impact of this new accounting guidance to have
a material impact on its financial condition or results of operations.
In September 2011, the FASB issued new accounting
guidance on testing goodwill for impairment. The primary objective of this accounting guidance is to reduce complexity and costs
by allowing an entity to make a qualitative evaluation about the likelihood of goodwill impairment to determine whether it should
calculate the fair value of a reporting unit. If, after assessing qualitative factors, an entity determines that it is not more
likely than not (a likelihood of more than
50 percent
) that the fair value of a reporting unit
is less than its carrying amount, then the two-step goodwill impairment test is unnecessary. This accounting guidance is effective
for the Company in fiscal 2013 but early adoption is permitted. The Company does not expect the impact of this new accounting guidance
to have a material impact on its financial condition or results of operations.
In December 2011, the FASB issued new accounting
guidance on disclosures about offsetting assets and liabilities. The requirements for offsetting are different under U.S. GAAP
and IFRS. Therefore, the objective of this accounting guidance is to facilitate comparison between financials statements prepared
under U.S. GAAP and IFRS by enhancing disclosures of the effect or potential effect of netting arrangements on an entity’s
financial position, including the effect or potential effect of rights of setoff associated with certain assets and liabilities.
This accounting guidance will be effective for the Company on April 1, 2013. The Company does not expect the impact of this new
accounting guidance to have a material impact on its financial condition or results of operations.
RESULTS OF OPERATIONS
For the Fiscal Years Ending March 31, 2012 and 2011
Comparative details of results of operations
for the years ended March 31, 2012 and 2011 as a percentage of sales are as follows:
|
|
|
2012
|
|
|
2011
|
|
|
NET SALES
|
|
|
100.00
|
%
|
|
|
100.00
|
%
|
|
COST OF SALES
|
|
|
23.00
|
%
|
|
|
31.38
|
%
|
|
GROSS PROFIT
|
|
|
77.00
|
%
|
|
|
68.62
|
%
|
|
OPERATING EXPENSES
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
3.70
|
%
|
|
|
3.41
|
%
|
|
Sales and marketing
|
|
|
22.40
|
%
|
|
|
17.65
|
%
|
|
General and administrative
|
|
|
41.08
|
%
|
|
|
39.53
|
%
|
|
Depreciation and amortization
|
|
|
5.84
|
%
|
|
|
6.20
|
%
|
|
TOTAL OPERATING EXPENSES
|
|
|
99.32
|
%
|
|
|
66.79
|
%
|
|
(LOSS) INCOME FROM OPERATIONS
|
|
|
(5.19
|
)%
|
|
|
1.84
|
%
|
|
Other income (expense)
|
|
|
21.84
|
%
|
|
|
(6.76
|
)%
|
|
INCOME (LOSS) BEFORE INCOME TAXES & NON-CONTROLLING INTEREST
|
|
|
16.66
|
%
|
|
|
(4.93
|
)%
|
|
Income tax expense
|
|
|
(3.25
|
)%
|
|
|
(1.75
|
)%
|
|
NET INCOME (LOSS) BEFORE NON-CONTROLLING INTEREST
|
|
|
13.40
|
%
|
|
|
(6.68
|
)%
|
|
NON-CONTROLLING INTEREST
|
|
|
8.34
|
%
|
|
|
5.62
|
%
|
|
NET INCOME (LOSS)
|
|
|
5.06
|
%
|
|
|
(12.30
|
)%
|
*Note that above figures are not comparable due to the equity
method used because:
A. Remedent OTC BV and its subsidiaries
are accounted for using the equity method since September 30, 2011, while previously the financials of Remedent OTC BV and its
subsidiaries were fully included in the financial statements of Remedent Inc., and
B. GlamSmile Asia and its subsidiaries are accounted for using
the equity method since February 2012, while previously the financials of GlamSmile Asia and its subsidiaries were fully included
in the financial statement of Remedent Inc.
Net Sales
Net sales decreased by approximately 23%
to $9,687,292 in the year ended March 31, 2012 as compared to $12,581,708 in the year ended March 31, 2011. The decrease
in sales is primarily due to the deconsolidation of our OTC division at the quarter ending September 2011 and secondly, the deconsolidation
of our Asian Division at the end of January 2012, resulting in decreased revenue. For both entities, the equity method is currently
being used in calculating the monthly results, whereas, in fiscal 2011, we reported a full year of revenue for all divisions.
Cost of Sales
Cost of sales decreased approximately 43.6%
to $2,227,891 in the year ended March 31, 2012 as compared to $3,947,699 in the year ended March 31, 2011. Cost of sales
as a percentage of sales has decreased because of the deconsolidation of our OTC division at the quarter ending September 2011,
known for its higher costs in reference to the cost of goods, compared to the costs of goods for the veneer product sold by our
GlamSmile Divisions, whereas for the year ended March 2011, we reported a full year of cost of goods for all divisions.
Gross Profit
Our gross profit decreased by $1,174,608
or 13.6%, to $7,459,401 for the fiscal year ended March 31, 2012 as compared to $8,634,009 for the year ended March 31, 2011 as
a result of the deconsolidation of our OTC division at the quarter ending September 2011 and secondly, the deconsolidation of our
Asian Division at the end of January 2012, resulting in decreased gross profit. For both entities, the equity method is currently
being used in calculating the monthly results, whereas, in fiscal 2011 we reported Gross Profit for all divisions
Operating Expenses
Research and Development
. Our
research and development expenses decreased $119,305 to $309,939 for the year ended March 31, 2012 as compared to $429,244 for
the year ended March 31, 2011, a decrease of 27.8%. Research and development expenses have decreased primarily because
of completion of RemeScar product and the launch of the final RemeScar product, reducing our research and development expense significantly.
Secondly, the deconsolidation of our Sylphar division at the end of September 2011, also contributed to a decrease in Research
and Development expenses.
Sales and marketing costs
. Our
sales and marketing costs increased $196,126 to $2,416,812 or 8.8% for the year ended March 31, 2012 as compared to $2,220,686
for the year ended March 31, 2011. The increase is largely due to our new Asian GlamSmile Sales Facilities.
General and administrative
costs
. Our general and administrative costs for the year ended March 31, 2012 and 2011 were $4,686,347 and
$4,972,982 respectively, representing a decrease of $286,635 or 5.8%. The decrease in general and administrative
costs as compared to the prior year primarily as a result of the deconsolidation of our OTC division at the quarter ending
September 2011 and secondly, the deconsolidation of our Asian Division at the end of January 2012, resulting in a decreased
General and administrative cost. For both entities, the equity method is currently applicable, whereas, in fiscal 2011, we
reported general and administrative costs for a full year for all divisions.
Depreciation and amortization
. Our
depreciation and amortization decreased $231,221 or 29.6%, to $548,662 for the year ended March 31, 2012 as compared to $779,883
for the year ended March 31, 2011. The decrease is largely due to the deconsolidation of both entities, as described
above.
Net interest expense.
Our net interest expense was $387,292 for the year ended March 31, 2012 as compared to $260,235 for the year ended March 31, 2011,
an increase of $127,057 or 48.8%. Interest expense has increased primarily because of increased utilization of our available
bank credit line and increased debt
Liquidity and Capital Resources
Cash and Cash Equivalents
Our balance sheet at March 31, 2012 reflects
cash and cash equivalents of $203,584 as compared to $1,662,520 as of March 31, 2011, a decrease of $1,458,936. Net
cash used by operations was $150,552 for the year ended March 31, 2012 as compared to net cash used by operations of $135,226 for
the year ended March 31, 2011, an increase year over year of $15,326 in cash used by operations.
As of March 31, 2012, there has been no
indication of a trend of increased doubtful accounts. As a result, at this time, we do not anticipate increased reserves.
Investing Activities
Net cash used by investing activities was
$3,840,048 for the year ended March 31, 2012 as compared to net cash used by investing activities of $533,001 for the year ended
March 31, 2011. Cash used in investing activities in the year ended March 31, 2012 was primarily cash divested as a result of the
deconsolidation of the GlamSmile Asia division as well as of the OTC division.
Financing Activities
Net cash provided by financing activities
totaled $2,524,412 for the year ended March 31, 2012 as compared to net cash provided by financing activities of $1,702,939 for
the year ended March 31, 2011. Net cash provided from financing activities in the year ended March 31, 2012 was higher than in
the year ended March 31, 2011 primarily because of the partial sale of our investment in Asia and because we received a short term
loan offset by net repayments against our line of credit in the amount of $414,061. We did not complete any private
placements in either 2012 or 2011.
The Company has a mixed-use line of credit
facility with BNP Paribas Fortis Bank, a Belgian bank (the “Facility”). The Facility is secured by a first
lien on the assets of Remedent N.V.. On January 3, 2008, an amendment was made decreasing the mixed-use line of credit
to €2,050,000, to be used by Remedent NV and/or Sylphar NV. Each line of credit carries its own interest rates and fees as
provided in the Facility and varies from the current prevailing bank rate
The latest amendment to the Facility, dated
June 7, 2010, amended and split the line of credit to €1,250,000, to be used by Remedent NV and €1,000,000 to be used
Sylphar NV. Each line of credit carries its own interest rates and fees as provided in the Facility and vary from the current prevailing
bank rate of approximately 3.27%, for draws on the credit line, to 9.9% for advances on accounts receivable concerning Remedent
N.V. and similar for Sylphar N.V. Remedent N.V is currently only utilizing two lines of credit, advances based on account receivables
and the straight loan. As of March 31, 2012 and March 31, 2011, Remedent N.V. had in aggregate, $1,079,263 and $1,452,024 in advances
outstanding, respectively, under the mixed-use line of credit facilities.
During the years ended March 31, 2012 and
March 31, 2011, we recognized an increase in cash and cash equivalents of $7,252 and $14,342, respectively, from the effect of
exchange rates between the Euro and the US Dollar.
Internal and External Sources of Liquidity
As of March 31, 2012, we had current assets
of $3,268,854 compared to $7,354,170 at March 31, 2011. This decrease of $4,085,316 was due to a decrease in cash of $1,458,936,
a decrease in accounts receivable of $1,926,411, and a decrease in inventories of $1,086,280, offset by an increase
in prepaid expenses of $386,311.
Current liabilities at March 31, 2012 of
$3,607,361 were $2,708,997 less than current liabilities as at March 31, 2011 of $6,316,358. The decrease was primarily
a result of the decrease in our line of credit by $1,081,411, a decrease in short term debt of $400,000, a decrease in accounts
payable of $ 735,077, a decrease in accrued liabilities of $719,817, a decrease in deferred revenue of $417,996 and a decrease
in our due to related parties of $33,518, offset by an increase in the current portion of our long term debt of $ 678,822 respectively.
The main reason for the decrease overall is the deconsolidation of our OTC division and our Asian investment, which are accounted
for using the equity method at the end of March 31, 2012.
Our cash and cash equivalents of $203,584
as of March 31, 2012 are not sufficient to support our operations through our current fiscal year and we need additional financing.
During the year ended March 31, 2012, we have been unable to generate cash flows sufficient to support our operations and have
been dependent on loans and strategic partnerships. We remain dependent on outside sources of funding until our results of operations
provide positive cash flows. During our current fiscal year, we expect to continue to incur operating losses. With our current
level of funding, substantial doubt exists about our ability to continue as a going concern.
The financial statements contained in this
report do not include any adjustments relating to the recoverability and classification of recorded asset amounts or amounts and
classification of liabilities that might be necessary should we be unable to continue in existence. Our ability to continue as
a going concern is dependent upon our ability to generate sufficient cash flows to meet our obligations on a timely basis, to obtain
additional financing as may be required, and ultimately to attain profitable operations. However, there is no assurance that profitable
operations or sufficient cash flows will occur in the future.
We have supported current operations by
raising additional operating cash through loans and strategic partnerships. This has provided us with the cash inflows to continue
our business plan, but has not resulted in significant improvement in our financial position. We are considering alternatives to
address our cash flow situation that include raising capital through additional sale of our common stock and/or debentures. This
could result in substantial dilution of existing stockholders.
As discussed in this Report, we anticipate
that we will need to raise additional funds to satisfy our working capital requirements and implement our business strategy
to expand our direct to consumer business model. We intend to continue to look for opportunities to expand the number
of GlamSmile Studios in China. As such, the Company is in discussions to sell its 50 % interest in the OTC Division for 1,000,000
Euros (at closing rate of March 31, 2012, $1,334,600). The sale is planned because management wants to focus completely on its
core business within the professional dental community and the veneer market.
However, there can be no assurance that
we will be successfully in the implementation of our plan, our current financial position can be improved, that we will be able
to locate a strategic partner, that we can raise additional working capital, or that we can achieve positive cash flows from operations.
Our long-term viability as a going concern is dependent upon our ability to (i) locate sources of debt or equity funding to meet
current commitments and near-term future requirements and (ii) achieve profitability and ultimately generate sufficient cash flow
from operations to sustain our continuing operations. In the event we are unable to raise additional funds, we may draw funds from
the balance remaining on our credit facility or may have to postpone our expansion plans to establish additional dental studios
in Europe and China.
Off-Balance Sheet Arrangements
At March 31, 2012, we were not a party to
any transactions, obligations or relationships that could be considered off-balance sheet arrangements.
ITEM 7A — QUANTITATIVE AND QUALITATIVE DISCLOSURES
ABOUT MARKET RISK
Not Applicable
ITEM 8 — FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
The Financial Statements that constitute
Item 8 are included at the end of this report beginning on Page F-1.
ITEM 9 — CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS
ON ACCOUNTING AND
FINANCIAL DISCLOSURE
None.
ITEM 9A - CONTROLS AND PROCEDURES
(a) Evaluation
of Disclosure Controls and Procedures
Under the supervision and with the participation
of our management, including our principal executive officer and principal financial officer, as of the end of the period covered
by this report, we conducted an evaluation of the effectiveness of the design and operation of our disclosure controls and procedures,
as defined in Rules 13a-15(e) and 15d-15(e) under the Securities Act of 1934. Our disclosure controls and procedures are designed
to provide reasonable assurance that the information required to be included in our Securities and Exchange Commission (“SEC”)
reports is recorded, processed, summarized and reported within the time periods specified in SEC rules and forms, relating to the
Company, including our consolidated subsidiaries, and was made known to them by others within those entities, particularly during
the period when this report was being prepared. Based on this evaluation, our principal executive officer and principal financial
officer concluded that, as of March 31, 2012, our disclosure controls and procedures are effective at these reasonable assurance
levels.
(b) Management’s
Report on Internal Control over Financial Reporting
The management of the Company is responsible
for establishing and maintaining adequate internal control over financial reporting. The Company’s internal control over
financial reporting is a process designed under the supervision of the Company’s principal executive officer and principal
financial officer to provide reasonable assurance regarding the reliability of financial reporting and the preparation of the Company’s
financial statements for external purposes in accordance with generally accepted accounting principles. Because of its inherent
limitations, internal control over financial reporting may not prevent or detect misstatements. All internal control systems, no
matter how well designed, have inherent limitations. Therefore, even those systems determined to be effective can provide only
reasonable assurances with respect to financial statement preparation and presentation. Additionally, projections of any evaluation
of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions,
or that the degree of compliance with the policies or procedures may deteriorate.
As of March 31, 2012, management assessed
the effectiveness of the Company’s internal control over financial reporting based on the criteria for effective internal
control over financial reporting established in “Internal Control — Integrated Framework,” issued by the Committee
of Sponsoring Organizations of the Treadway Commission (the COSO criteria). Based on the assessment, management determined that
the Company maintained effective internal control over financial reporting as of March 31, 2012 based on the COSO criteria.
This annual report does not include an attestation
report of the Company’s independent registered public accounting firm regarding internal control over financial reporting.
Management’s report was not subject to attestation by the Company’s independent registered public accounting firm pursuant
to permanent rules of the Securities and Exchange Commission that permit the Company to provide only management’s
report in this annual report.
Change in Internal Control Over Financial Reporting
There have been no changes in the Company’s
internal controls over financial reporting during the quarter ended March 31, 2012 that have materially affected, or are reasonably
likely to materially affect, the Company’s internal control over financial reporting.
ITEM 9B— OTHER INFORMATION
None.
PART III
ITEM 10 — DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE
GOVERNANCE
Directors, Executive Officers and Significant Employees
The following table sets forth the names
and ages of our current directors and executive officers, the principal offices and positions with us held by each person and the
date such person became our director or executive officer. Our executive officers are elected annually by the Board
of Directors. Each year the stockholders elect the board of directors. The executive officers serve terms
of one year or until their death, resignation or removal by the Board of Directors. There was no arrangement or understanding
between any executive officer or director and any other person pursuant to which any person was elected as an executive officer
or director. There are no family relationships between any of our directors, executive officers, director nominees or
significant employees. Mr. Kolsteeg is independent as determined by the NASDAQ rules.
|
Person
|
|
Age
|
|
Position
|
|
Guy De Vreese
|
|
57
|
|
Chairman, Chief Executive Officer
|
|
Stephen Ross
|
|
53
|
|
Chief Financial Officer, Director, Secretary
|
|
Fred Kolsteeg
|
|
69
|
|
Director
|
|
Philippe Van Acker
|
|
47
|
|
Director, Chief Accounting Officer
|
Biographies
Guy De Vreese, Chairman
. From
April 1, 2002, Mr. De Vreese has served as our Chairman of the Board. Effective upon Mr. List’s resignation as
Chief Executive Officer, on December 10, 2008 Mr. De Vreese became our Chief Executive Officer. From June 2001 Mr. De
Vreese has also served as President of Remedent N.V. and he has served as President of DMDS, Ltd., a European subsidiary of Dental
& Medical Systems, Inc. DMDS, Ltd. developed and marketed high-tech dental equipment. In August 1996, Mr. De Vreese
founded DMD N.V., a Belgian company that was the independent European distributor for DMDS products and was its Chief Executive
Officer until DMD purchased its distribution rights in April 1998. Mr. De Vreese later worked as CEO from 1996 through
February 1999 for Lident, N.V., a Belgian company that merged with DMD and specialized in digital photography and developer of
imaging software. Mr. De Vreese also served as a consultant providing services to DMDS, Ltd. from February 1999 to June
2001. Mr. De Vreese resides in Belgium. Mr. De Vreese’s years of experience in the dental industry provides
us with invaluable industry contacts and know-how, in addition to special insight into our customers’ needs and requirements.
In addition, Mr. De Vreese’s extensive experience in our industry, commitment to research and development of innovative dental
products, and in-depth knowledge of our company gained by serving as our CEO provide valuable insights for our Board. The
Board believes that Mr. De Vreese demonstrated leadership abilities and business judgment, provide an important leadership element
to our Board
Stephen Ross, Director, Chief Financial
Officer, Secretary
. Mr. Ross has served as our director since August 2001 and as our Secretary since April 2002. He
also served as our Chief Financial Officer from August 2001 until March 2005. He was reappointed as Chief Financial
Officer, effective December 18, 2008. From February 1998 through January 2001, Mr. Ross was CFO of Dental & Medical
Diagnostic Systems, Inc., a company that developed and marketed high-tech dental equipment and declared bankruptcy in July 2001. Commencing
in 1996 and terminating February 1998, Mr. Ross served as a senior management consultant with Kibel and Green, a corporate restructuring
and management firm. Prior to working for Kibel and Green, Mr. Ross served as CFO and co-founder of a personal care
company, and as tax manager with an accounting firm. Mr. Ross resides in Los Angeles, California. Our Board believes
that Mr. Ross’ financial background, relevant expertise in our industry and knowledge of our company gained by
serving as our CFO provide special insight and perspective for our Board. Our Board believes that Mr. Ross’ leadership and
management experience at multiple companies enables him to provide our Board with critical perspectives on issues important to
our business. In addition, our Board believes that our CFO should serve on the Board of Directors to facilitate direct access and
open communication on issues relating to internal controls over financial reporting.
Fred Kolsteeg, Director
. Mr.
Kolsteeg has served as a director of the Company since April 2002. Since 1996, Mr. Kolsteeg has served as the president
of WAVE Communications, a Dutch based advertising agency. Prior to founding WAVE in 1996, he founded several other advertising
agencies such as ARA, Team and Team Saatchi. Mr. Kolsteeg has also worked at Phillips and Intermarco Publicis. Mr.
Kolsteeg resides in Holland. Mr. Kolsteeg has experience managing operations and finance for multiple businesses.
Our Board believes that this experience, adds valuable perspectives and he is an “audit committee financial expert”
as defined in SEC rules.
Philippe Van Acker, Director, Chief
Accounting Officer
. Mr. Van Acker was appointed as our Chief Financial Officer as of March 30, 2005. Effective
December 18, 2008, Mr. Van Acker resigned as Chief Financial Officer and became our Chief Accounting Officer as well as assuming
a position on the Board of Directors. From July 2001 to March 30, 2005, Mr. Van Acker has served as a director of our
subsidiary, Remedent N.V. where he has also served as financial controller. From 1999 to 2001, Mr. Van Acker served
as Director of Finance for DMDS, Ltd., a European subsidiary of Dental & Medical Diagnostic Systems, Inc., a company that developed
and marketed high-tech dental equipment. From 1992 to 1999, Mr. Van Acker held various positions with Pfizer Medical
Technology Group. Mr. Van Acker resides in Belgium. Mr. Van Acker’s executive management experience
and extensive background in finance and investment matters provide important contributions and critical insight to our Board. The
Board believes that Mr. Van Acker’s financial background and understanding of the dental industry and our business
bring perspectives beneficial to the Board as the company seeks to expand its presence in Europe and China.
Legal Proceedings
None of our directors or executive officers
were involved in a legal proceeding during the past ten years which are material to an evaluation of the ability or integrity of
any director, person nominated to become a director or executive officer of the Company and required to be disclosed under Item
401(f) of Regulation S-K.
Director Committees and Director Independence
The Company does not have a separate compensation,
nominating or audit committee. The Board of Directors has determined that Mr. Kolsteeg is “independent,”
as independence is defined in the listing standards for the Nasdaq Stock Market. Accordingly, we have one director who
is “independent.”
Board Leadership Structure and Role in Risk Oversight
The Board does not have a policy as to whether
the roles of our Chairman and Chief Executive Officer should be separate. Instead, the Board makes this determination based on
what best serves the our Company’s needs at any given time. Currently, Mr. De Vreese holds the positions
of Chairman and Chief Executive Officer of our Company, and the Board has one independent director. The Board may decide to separate
the positions of Chairman and Chief Executive Officer in the future if the Board believes it is in the best interest of the Company
and our stockholders.
The Board believes that effective board
leadership is highly dependent on the experience, skills and personal interaction between persons in leadership roles. The
Company believes that having one person, particularly Mr. De Vreese with his extensive knowledge of the industry and
executive management experience, his extensive knowledge of the operations of the Company and his own commitment to innovation
and strategic thinking, serve as both Chief Executive Officer and Chairman is the best leadership structure for the Company because
it demonstrates to our employees, customers and stockholders that the Company is under strong leadership, with a single person
setting the tone and having primary responsibility for managing the Company’s operations. Accordingly, with significant input
from our Board, Mr. De Vreese sets the strategic direction for our Company and provides daily leadership and guidance to our managements
and employees. In addition, this unity of leadership promotes strategy development and execution, timely decision-making
and effective management of Company resources. The Company believes that it has been well-served by this structure.
In its governance role, and particularly
in exercising its duty of care and diligence, the Board is responsible for ensuring that appropriate risk management policies and
procedures are in place to protect the company’s assets and business. Our Board has broad and ultimate oversight
responsibility for our risk management processes and programs and executive management is responsible for the day-to-day evaluation
and management of risks to the company.
Audit Committee Financial Expert
Our Board of Directors has not established
a separate audit committee within the meaning of Section 3(a)(58)(A) of the Securities Exchange Act of 1934, as amended (the “Exchange
Act”). Instead, our entire Board of Directors acts as the audit committee within the meaning of Section 3(a)(58)(B) of the
Exchange Act. In addition, our Board of Directors has not made a determination as to whether a director on the Board
meets the definition of an “audit committee financial expert” within the meaning of Item 407(d)(5) of Regulation S-K.
We continue to seek candidates for outside directors and for a financial expert to serve on a separate audit committee
when we establish one. Due to our small size and limited resources, it has been difficult to recruit outside directors and financial
experts, especially due to the fact that we do not have directors and officer’s liability insurance to offer suitable candidates.
In fulfilling its oversight responsibilities,
the Board has reviewed and discussed the audited financial statements with management and discussed with the independent auditors
the matters required to be discussed by SAS 61. Management is responsible for the financial statements and the reporting process,
including the system of internal controls. The independent auditors are responsible for expressing an opinion on the conformity
of those audited financial statements with generally accepted accounting principles.
The Board discussed with the independent
auditors, the auditors’ independence from the management of the Company and received written disclosures and the letter from
the independent accountants required by Independence Standards Board Standard No. 1.
After Board review and discussions, as mentioned
above, the Board recommended that the audited financial statements be included in the Company’s Annual Report on Form 10-K.
Governance Committee and Nominations to the Board of Directors
There were no material changes to the procedures
by which security holders may recommend nominees to our Board of Directors.
Code of Ethics
We have adopted a written Code of Ethics
that applies to our senior management. A copy of our Code of Ethics, executed by the Chief Executive Officer and Chief Financial
Officer, has been filed as an exhibit to our Annual Report on Form 10-K for the fiscal year ended March 31, 2003. A copy of our
Code of Ethics is available to any stockholder by addressing a request to the attention of the Secretary of the Company and mailing
such request to the Company’s corporate offices. Any amendment to the Code of Ethics or any waiver of the Code of Ethics
will be disclosed promptly following the date of such amendment or waiver pursuant to a Form 8-K filing with the Securities and
Exchange Commission.
Compliance with Section 16 of the Securities Exchange Act
of 1934
Section 16(a) of the Securities Exchange
Act of 1934, as amended, requires our executive officers and directors and persons who own more than 10% of a registered class
of our equity securities, to file with the Securities and Exchange Commission (hereinafter referred to as the “Commission”)
initial statements of beneficial ownership, reports of changes in ownership and Annual Reports concerning their ownership, of Common
Stock and other of our equity securities on Forms 3, 4, and 5, respectively. Executive officers, directors and greater than 10%
stockholders are required by Commission regulations to furnish us with copies of all Section 16(a) reports they file. Based solely
on information available to us in public filings, we believe that all reports required by Section 16(a) for transactions
in the year ended March 31, 2012, were timely filed.
ITEM 11 — EXECUTIVE COMPENSATION
Summary Compensation
Our Board of Directors has not established
a separate compensation committee nor any other committee that acts as such a committee. Instead, the entire Board of Directors
reviews and approves executive compensation policies and practices, reviews, salaries and bonuses for our officers, administers
our benefit plans, and considers other matters as may, from time to time, be referred to it. We do not currently have a Compensation
Committee Charter. Our Board continues to emphasize the important link between our performance, which ultimately benefits
all stockholders, and the compensation of our executives. Therefore, the primary goal of our executive compensation policy is to
closely align the interests of the stockholders with the interests of the executive officers. In order to achieve this goal, we
attempt to (i) offer compensation opportunities that attract and retain executives whose abilities and skills are critical to our
long-term success and reward them for their efforts in ensuring our success and (ii) encourage executives to manage from the perspective
of owners with an equity stake in the Company.
The following table sets forth information
regarding all forms of compensation received by the named executive officers during the fiscal years ended March 31, 2012 and March
31, 2011, respectively:
|
SUMMARY COMPENSATION TABLE
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Name and
Principal Position
(a)
|
|
Year
(b)
|
|
|
Salary
($)
(c)
|
|
|
Bonus
($)
(d)
|
|
|
Stock
Awards
($)
(e)
|
|
|
Option
Awards
($)
(f)
|
|
|
Non-Equity
Incentive
Plan
Compensation
($)
(g)
|
|
|
Nonqualified
Deferred
Compensation
Earnings
($)
(h)
|
|
|
All Other
Compensation
($)
(i)
|
|
|
Total
($)
(j)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Guy De Vreese, CEO and Chairman
|
|
|
2012
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
348,249
|
(1)
|
|
$
|
348,249
|
(1)
|
|
|
|
|
2011
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
333,819
|
(1)
|
|
$
|
333,819
|
(1)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Philippe Van Acker,
|
|
|
2012
|
|
|
$
|
189,403
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
189,403
|
|
|
Chief Accounting Officer, Director
|
|
|
2011
|
|
|
$
|
175,491
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
175,491
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stephen Ross
|
|
|
2012
|
|
|
$
|
120,000
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
120,000
|
|
|
CFO, Director
|
|
|
2011
|
|
|
$
|
240,000
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
$
|
240,000
|
|
|
|
(1)
|
These amounts are consulting fees, including a car allowance paid by Remedent N.V. to Lausha, N.V., a company controlled by Mr. De Vreese, pursuant to an oral consulting agreement between Lausha N.V. and Remedent N.V.
|
Outstanding Equity Awards at Fiscal Year End
The following table provides information
with respect to the named executive officers concerning unexercised stock options held by them at March 31, 2012. As
of March 31, 2012, there were no outstanding stock awards and columns (g) through (j) have been omitted.
|
OPTION AWARDS
|
Name
(a)
|
|
Number of
Securities
Underlying
Unexercised
Options
(#)
Exercisable
(b)
|
|
|
Number of
Securities
Underlying
Unexercised
Options
(#)
Unexercisable
(c)
|
|
|
Equity
Incentive
Plan
Awards:
Number of
Securities
Underlying
Unexercised
Unearned
Options
(#)
(d)
|
|
|
Option
Exercise
Price
($)
(e)
|
|
|
Option
Expiration
Date
(f)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Guy De Vreese
|
|
|
100,000
|
|
|
|
-0-
|
|
|
|
|
|
|
$
|
1.75
|
|
|
20-Sept-2017
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Philippe Van Acker
|
|
|
75,000
|
|
|
|
-0-
|
|
|
|
|
|
|
$
|
2.46
|
|
|
23-Dec-2015
|
|
Philippe Van Acker
|
|
|
50,000
|
|
|
|
-0-
|
|
|
|
|
|
|
$
|
1.75
|
|
|
20-Sept-2017
|
|
Philippe Van Acker
|
|
|
100,000
|
|
|
|
-0-
|
|
|
|
|
|
|
$
|
0.50
|
|
|
19-Mar-2019
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stephen Ross
|
|
|
12,500
|
|
|
|
-0-
|
|
|
|
|
|
|
$
|
2.00
|
|
|
8-Apr-2014
|
|
Stephen Ross
|
|
|
100,000
|
|
|
|
-0-
|
|
|
|
|
|
|
$
|
0.50
|
|
|
19-Mar-2019
|
Director Compensation Table
Generally, our directors do not receive
any cash compensation, but are entitled to reimbursement of their reasonable expenses incurred in attending directors’ meetings. However,
at the discretion of our Board of Directors, we may periodically issue stock options under our stock option plan to directors.
Our directors did not receive any compensation
for board services during the fiscal year ended March 31, 2012.
Employment Agreements
Our subsidiary, Remedent, N.V., has
an employment agreement with Mr. Philippe Van Acker, our Chief Accounting Officer. We do not currently have
any other employment agreements with our executive officers. However, we anticipate having employment contracts with executive
officers and key personnel as necessary, in the future.
Long-Term Incentive Plans-Awards in Last Fiscal Year
We do not currently have any long-term
incentive plans.
ITEM 12 — SECURITY OWNERSHIP OF CERTAIN BENEFICIAL
OWNERS AND MANAGEMENT AND
RELATED STOCKHOLDER MATTERS
The following table sets forth information
regarding the beneficial ownership of our common stock as of June 25, 2012. The information in this table
provides the ownership information for:
|
|
a.
|
each person known by us to be the beneficial owner of more than 5% of our common stock;
|
|
|
b.
|
each of our directors;
|
|
|
c.
|
each of our executive officers; and
|
|
|
d.
|
our executive officers, directors and director nominees as a group.
|
Beneficial ownership has been determined
in accordance with Rule 13d-3 of the 1934 Exchange Act and includes voting or investment power with respect to the shares. Unless
otherwise indicated, the persons named in the table below have sole voting and investment power with respect to the number of shares
indicated as beneficially owned by them. Common stock beneficially owned and percentage ownership is based on 19,995,969
shares outstanding as of June 25, 2012.
|
Name of Beneficial Owner (1)
|
|
Shares
Beneficially
Owned
|
|
|
Percentage
Beneficially
Owned
|
|
|
|
|
|
|
|
|
|
|
Guy De Vreese, CEO, Chairman (2)
|
|
|
|
|
|
|
|
|
|
Xavier de Cocklaan 42
|
|
|
|
|
|
|
|
|
|
9831 Deurle, Belgium
|
|
|
4,733,680
|
|
|
|
23.67
|
%
|
|
|
|
|
|
|
|
|
|
|
|
Philippe Van Acker, Director (3)
|
|
|
|
|
|
|
|
|
|
Xavier de Cocklaan 42
|
|
|
|
|
|
|
|
|
|
9831 Deurle, Belgium
|
|
|
225,000
|
|
|
|
1.13
|
%
|
|
|
|
|
|
|
|
|
|
|
|
Stephen Ross, CFO, Secretary, Director (4)
|
|
|
|
|
|
|
|
|
|
1921 Malcolm #101
|
|
|
|
|
|
|
|
|
|
Los Angeles, CA 90025
|
|
|
576,327
|
|
|
|
2.88
|
%
|
|
|
|
|
|
|
|
|
|
|
|
Fred Kolsteeg, Director (5)
|
|
|
|
|
|
|
|
|
|
Managelaantje 10
|
|
|
|
|
|
|
|
|
|
3062 CV Rotterdam
|
|
|
|
|
|
|
|
|
|
The Netherlands
|
|
|
295,000
|
|
|
|
1.48
|
%
|
|
|
|
|
|
|
|
|
|
|
|
All Officers and Directors as a Group (4 persons)
|
|
|
5,945,007
|
|
|
|
29.16
|
%
|
|
|
|
|
|
|
|
|
|
|
|
5% or Greater Stockholders
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Austin W. Marxe and David M. Greenhouse (6)
|
|
|
|
|
|
|
|
|
|
153 East 53rd Street, 55th Floor
|
|
|
|
|
|
|
|
|
|
New York, NY 10022
|
|
|
4,520,933
|
|
|
|
22.61
|
%
|
|
|
|
|
|
|
|
|
|
|
|
Den-Mat Holdings, LLC (7)
|
|
|
|
|
|
|
|
|
|
2727 Skyway Drive
|
|
|
|
|
|
|
|
|
|
Santa Maria, CA 93455
|
|
|
3,378,379
|
|
|
|
14.45
|
%
|
* Less than one percent
|
|
1.
|
Beneficial ownership has been determined in accordance with Rule 13d-3 under the Exchange Act. Pursuant to the rules of the Securities and Exchange Commission, shares of common stock which an individual or group has a right to acquire within 60 days pursuant to the exercise of options or warrants are deemed to be outstanding for the purpose of computing the percentage ownership of such individual or group, but are not deemed to be beneficially owned and outstanding for the purpose of computing the percentage ownership of any other person shown in the table.
|
|
|
2.
|
Guy De Vreese holds 3,304,426 shares in his own name, which such amount includes 100,000 shares of common stock underlying options which vested on September 17, 2007 and have an exercise price of $1.75 per share; 72,787 shares of common stock held in the name of Lausha N.V., a Belgian company controlled by Guy De Vreese; 6,467 shares of common stock held in the name of Lident N.V., a Belgian company controlled by Guy De Vreese; and 1,400,000 shares of common stock held in the name of Lausha HK, a Hong Kong company controlled by Guy De Vreese.
|
|
|
3.
|
Includes 75,000 shares of common stock underlying options which vested on December 2005 and have an exercise price of $2.46 per share; 50,000 shares of common stock underlying options which have vested as of September 17, 2009 and have an exercise price of $1.75 per share; and 100,000 shares of common stock underlying options which were fully vested on March 19, 2009 and have an exercise price of $0.50 per share.
|
|
|
4.
|
Includes 12,500 shares of common stock underlying options which vested on April 8, 2004 and have an exercise price of $2.00 per share; and 100,000 shares of common stock underlying options which were fully vested on March 19, 2009 and have an exercise price of $0.50 per share.
|
|
|
5.
|
Includes 200,000 shares of common stock underlying options which were fully vested on March 19, 2009 and have an exercise price of $0.50 per share.
|
|
|
6.
|
Austin W. Marxe (Marxe) and David M. Greenhouse (Greenhouse) share voting and investment control over all securities owned by Special Situations Fund III QP, L.P. (QP), Special Situations Cayman Fund, L.P. (CAY) and Special Situations Private Equity Fund, LP (PE). 541,075 shares of Common Stock are owned by QP, 960,075 shares of Common Stock are owned by CAY and 3,019,783 shares of Common Stock are owned by PE. The interest of Marxe and Greenhouse in the shares of Common Stock owned by QP, CAY and PE is limited to the extent of his pecuniary interest.
|
|
|
7..
|
Consists of warrants to purchase 3,378,379 shares of common stock.
|
ITEM 13 — CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS
AND DIRECTOR INDEPENDENCE
Related Transactions
Compensation
During the years ended March 31, 2012 and
2011 respectively, the Company incurred $664,586 and $749,310 as compensation for all directors and officers.
Sales Transactions
All related party transactions involving
provision of services or tangible assets were recorded at the exchange amount, which is the value established and agreed to by
the related parties reflecting arm’s length consideration payable for similar services or transfers.
Other Transactions
On June 3, 2009, the Company entered into
the First Fit-Crown Distribution and License Agreement (the “First Fit Distribution Agreement”) with Den-Mat. Under
the terms of the First Fit Distribution Agreement, the Company appointed Den-Mat to be the its sole and exclusive distributor to
market, license and sell certain products relating to the Company’s proprietary First Fit technology (the “First Fit
Products”), in the United States, Canada and Mexico (the “First Fit Territory”). In connection therewith,
the Company also granted Den-Mat certain non-exclusive rights to manufacture and produce the First Fit Products in the First-Fit
Territory; and a sole and exclusive transferable and sublicensable right and license to use the Company’s intellectual property
rights relating to the First Fit Products to perform its obligations as a distributor (provided the Company retains the right to
use and license related intellectual property in connection with the manufacture of the First Fit Products for sale outside of
the First Fit Territory), as the terms and transactions are further detailed in the First Fit Distribution Agreement. The
consummation of the transactions described herein and contemplated in the First Fit Distribution Agreement are subject to certain
closing conditions which includes, in addition to customary closing conditions: the completion of Den-Mat’s due diligence
with respect to the First Fit Products to its satisfaction; execution and delivery of Non-Competition Agreements by
Guy De Vreese and Evelyne Jacquemyns; and the delivery of the Development Payment and first installment of the License Payment
(the “Development Payment” and License Payment” are defined below). The First Fit Distribution Agreement
provides that the consummation of the transactions contemplated therein will occur upon the performance or waiver of such closing
conditions. Under the First Fit Distribution Agreement, the Company granted such distribution rights, licensing rights
and manufacturing rights, in consideration for the following: (i) a non-refundable development fee of Four Hundred Thousand
Dollars ($400,000) (the “Development Payment”) payable in two installments as follows: (a) Fifty Thousand Dollars ($50,000)
within seven (7) days after the effective date of the First Fit Distribution Agreement (the “Effective Date”), and
(b) Three Hundred Fifty Thousand Dollars ($350,000) within twenty one (21) days after the Effective Date ; (ii) a non-refundable
license fee of Six Hundred Thousand Dollars ($600,000) payable in three (3) equal installments of $200,000 each, with the first
installment payable on the closing date contemplated in the First Fit Distribution Agreement (the “Closing Date”),
and with the second and third installments payable on the 30th and 60th day, respectively, after the Closing Date; (iii) certain
royalty payments based on the sales of the First Fit Products by Den-Mat or its sublicensees; and (iv) certain minimum royalty
payment to maintain exclusivity, as such terms are more particularly described in the First Fit Distribution Agreement.
On March 29, 2010, a certain Amendment No.
1 to First Fit Crown Distribution and License Agreement (“First Fit Amendment”) between the Company and Den-Mat,
pursuant to which the Company agreed to sell to Den-Mat all of the intellectual property or used by Company related to the First
Fit product (“First Fit IP”) became effectuated upon Company’s receipt of Den-Mat’s countersignatures to
the First Fit Amendment. The First Fit Amendment amends the First Fit-Crown Distribution and License Agreement
dated June 3, 2009 between the Company and Den-Mat, pursuant to which Den-Mat was granted certain license and distribution rights
relating to the First Fit IP and First Fit products. The total purchase price for the First Fit IP consists of installment
payments and royalty payments. The cash component of the purchase price of the First Fit IP is $2,850,000 to be
paid in the form of cash in the following installments: (a) $50,000 upon delivery by Remedent to Den-Mat of a working prototype
of the First Fit crown, (b) $525,000 on or before March 15, 2010 (c) $700,000 on June 30, 2010, and (d) $500,000 on December
31, 2010, June 30, 2011 and December 31, 2011. In connection with the execution of the First Fit Agreement, Den-Mat also agreed
to make an advance cash payment of $75,000 to the Company towards the purchase price. In addition to the cash component,
Den-Mat agreed to pay Remedent a capital payment equal to a certain percent of Den-Mat’s net revenues generated by the sale
of the First Fit products.
In connection with our Distribution, License
& Manufacturing Agreement with Den-Mat (the “Distribution Agreement”) dated as August 2008, as amended and restated
by the parties on June 3, 2009 pursuant to the Amended and Restated Distribution, License and Manufacturing Agreement, and as consideration
for Den-Mat’s obligations under the Distribution Agreement, we agreed, among other things, to issue Den-Mat or an entity
to be designated by Den-Mat, warrants to purchase up to three million three hundred seventy-eight thousand three hundred seventy-nine
(3,378,379) shares of our common stock, par value $0.001 per share at an exercise price of $1.48 per share, exercisable for a period
of five years . The warrants were issued in the period ended September 30, 2008 and were valued at $4,323,207 based
upon the Black-Scholes option pricing model utilizing a market price on the date of grant of $1.48 per share, an annualized volatility
of 131%, a risk free interest rate of 3.07% and an expected life of five years. The expense was originally classified as
a non-operating expense however, we have subsequently reclassified the expense to operations since our agreement with Den-Mat is
in the normal course of our operations. The reclassification increased our operations expenses by $4,323,207, while reducing our
other expenses by the same amount, resulting in no net impact upon our consolidated net loss for the year ended March 31,2009.
On March 29, 2010, concurrently with the
execution of the First Fit Amendment (as described above), the Company and Den-Mat entered into Amendment No. 2 to the Amended
and Restated Distribution, License and Manufacturing Agreement (“Glamsmile Amendment”) with Den-Mat pursuant to which
certain provisions of a certain Amended and Restated Distribution, License and Manufacturing Agreement previously entered into
by the Company and Den-Mat on June 3, 2009 and subsequently amended on August 11, 2009, were amended. The Glamsmile
Amendment became effective concurrently with the effectiveness of the First Fit Amendment. Among other things, the Glamsmile
Amendment (1) permits the Company to purchase its requirements for GlamSmile Products from another party, other than Den-Mat, provided
Company pays De-Mat a royalty payment on net revenues received by Company per unit/tooth, (2) decreases the percentage of securities
to be covered in a warrant to purchase securities of B2C Market Subsidiary and the exercise price of such warrant to be issued
to Den-Mat in the event a B2C Market Subsidiary is formed under the terms set forth in such agreement, (3) expands the
definition of “Excluded Market” to include Australia, Belgium, France and United Arab Emirates, and (4) provides a
consulting fee, equal to a percentage of net revenues received by Den-Mat from the Sale of Unit/Teeth and trays, to the Company
for its services, support and certain additional consideration, (5) terminates certain provisions relating to minimum requirement
obligations and rights, and (6) amends the formula for calculation a certain exit fee in the event of a change of control.
On March 27, 2012, the distribution agreements
with Den-Mat were terminated pursuant to a certain Termination and Distribution Agreement with Den-Mat (“Den-Mat Distribution
Agreement”). Pursuant to the Den-Mat Distribution Agreement, we granted Den-Mat a non-exclusive, irrevocable, perpetual,
royalty free, license to use within certain territory, which among other territories excludes China, Macau, Hong Kong, and Taiwan,
the intellectual property that was the subject of the license to Den-Mat under the Amended and Restated Distribution, License and
Manufacturing Agreement dated June 3, 2009, as amended from time to time (“Prior Agreements”), as such intellectual
property relates the products which was the subject of the Prior Agreements. In connection with the termination of the Prior Agreements,
under the Den-Mat Distribution Agreement, Den-Mat agreed to pay us $200,000.
Guy De Vreese, our Chairman of the Board
and Chief Executive Officer, is the managing director of our subsidiary, Remedent N.V. Mr. De Vreese provides his services
as Remedent N.V.’s Managing Director through his company, Lausha, N.V. Lausha, N.V. has an oral consulting
arrangements with Remedent N.V. that provide for Mr. De Vreese’s services and is a company controlled by Mr. De
Vreese. On March 20, 2006, Lausha N.V. and Lident N.V. merged into Lausha N.V., controlled by Mr. De Vreese. Lausha
N.V. received a total of $348,249 and $333,819 as compensation for services relating to his duties as Managing Director of Remedent
N.V. on a-day-to-day base for the years ending March 31, 2012 and March 31, 2011, respectively.
Director Independence
Due the Company’s size and limited
resources, the Company does not have a separate compensation, nominating or audit committee. The Board of Directors
has determined that Mr. Kolsteeg is “independent,” as independence is defined in the listing standards for
the Nasdaq Stock Market. Accordingly, we have one director who is “independent.”
ITEM 14. PRINCIPAL ACCOUNTANT FEES AND SERVICES.
Audit Fees
. The aggregate fees paid
for the annual audit of financial statements included in our Annual Report for the year ended March 31, 2012 and the review of
our quarterly reports for such year, amounted to $78,128. The aggregate fees paid for the annual audit of financial statements
included in our Annual Report for the year ended March 31, 2012 and the review of our quarterly reports for such years amounted
to $66,719.
Audit Related Fees
. For the years
ended March 31, 2012 and March 31, 2011, we paid $ 12,074 and $20,332, respectively, to PKF for other audit related fees.
Tax Fees
. For the years ended March
31, 2012 and March 31, 2011, we paid $1,824 and $2,834,, respectively, to PKF for fees associated with tax return preparation.
The above-mentioned
fees are set forth as follows in tabular form:
|
|
|
2012
|
|
|
2011
|
|
|
Audit Fees
|
|
$
|
78,128
|
|
|
$
|
66,719
|
|
|
Audit Related Fees
|
|
$
|
12,074
|
|
|
$
|
20,332
|
|
|
Tax Fees
|
|
$
|
1,824
|
|
|
$
|
2,834
|
|
|
All Other Fees
|
|
|
-
|
|
|
|
-
|
|
The Company’s Board of Directors serves
as the Audit Committee and has unanimously approved all audit and non-audit services provided by the independent auditors. The
independent accountants and management are required to periodically report to the Board of Directors regarding the extent of services
provided by the independent accountants, and the fees for the services performed to date.
There have been no non-audit services provided
by our independent accountant for the year ended March 31, 2012.
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
|
(a)(1)
|
Financial Statements
.
|
Consolidated balance sheet as of March 31, 2012 and
March 31, 2011, and the related consolidated statements of operations, stockholders’ equity, cash flows, and comprehensive
loss for each of the years in the 2 year period ended March 31, 2012.
All schedules have been omitted because they are not
required, not applicable, or the information is otherwise set forth in the consolidated financial statements or the notes thereto.
The information required by this Item is set forth
in the section of this Annual Report entitled “EXHIBIT INDEX” and is incorporated herein by reference.
SIGNATURES
In accordance with Section 13 or 15(d)
of the Securities Exchange Act of 1934, as amended (the “Exchange Act”) the Registrant caused this report to be signed
on its behalf by the undersigned, thereunto duly authorized.
|
|
REMEDENT, INC.
|
|
|
|
|
Dated: July 16, 2012
|
/s/ Guy De Vreese
|
|
|
By: Guy De Vreese
|
|
|
Its: Chief Executive Officer (Principal Executive
|
|
|
Officer)
|
|
|
|
|
Dated: July 16, 2012
|
/s/ Stephen Ross
|
|
|
By: Stephen Ross
|
|
|
Its: Chief Financial Officer (Principal Financial
|
|
|
Officer and Principal Accounting Officer)
|
In accordance with
the Exchange Act, this report has been signed by the following persons on behalf of the registrant and in the capacities and on
the dates indicated.
|
Dated: July 16, 2012
|
/s/ Guy De Vreese
|
|
|
Guy De Vreese, Chairman of the Board of Directors, and
|
|
|
Chief Executive Officer (“Principal Executive Officer”)
|
|
|
|
|
Dated: July 16, 2012
|
/s/ Stephen Ross
|
|
|
Stephen Ross, Director and Chief Financial
|
|
|
Officer (Principal Financial Officer and
|
|
|
Principal Accounting Officer)
|
|
|
|
|
Dated: July 16, 2012
|
/s/ Philippe Van Acker
|
|
|
Philippe Van Acker, Director and Chief
|
|
|
Accounting Officer
|
|
|
|
|
Dated: July 16, 2012
|
/s/ Fred Kolsteeg
|
|
|
Fred Kolsteeg, Director
|
EXHIBIT INDEX
|
Exhibit No.
|
|
Description
|
|
|
|
|
|
2.1
|
|
Stock Exchange Agreement with Resort World Enterprises, Inc. (1)
|
|
|
|
|
|
3.1
|
|
Articles of Incorporation of Jofran Confectioners International, Inc., a Nevada corporation, dated July 31, 1986 (1)
|
|
|
|
|
|
3.2
|
|
Amendment to Articles of Incorporation changing name from Jofran Confectioners International, Inc., a Nevada corporation, to Cliff Typographers, Inc., a Nevada corporation, dated July 31, 1986 (1)
|
|
|
|
|
|
3.3
|
|
Amendment to Articles of Incorporation changing name from Cliff Typographers, Inc., a Nevada corporation, to Cliff Graphics International, Inc., a Nevada corporation, dated January 9, 1987 (1)
|
|
|
|
|
|
3.4
|
|
Amendment to Articles of Incorporation changing name from Cliff Graphics International, Inc., a Nevada corporation, to Global Golf Holdings, Inc., a Nevada corporation, dated March 8, 1995 (1)
|
|
|
|
|
|
3.5
|
|
Amendment to Articles of Incorporation changing name from Global Golf Holdings, Inc., a Nevada corporation, to Dino Minichiello Fashions, Inc., a Nevada corporation, dated November 20, 1997 (1)
|
|
|
|
|
|
3.6
|
|
Amendment to Articles of Incorporation changing name from Dino Minichiello Fashions, Inc., a Nevada corporation, to Resort World Enterprises, Inc., a Nevada corporation, dated August 18, 1998 (1)
|
|
|
|
|
|
3.7
|
|
Amendment to Articles of Incorporation changing name from Resort World Enterprises, Inc., a Nevada corporation, to Remedent, Inc., dated October 5, 1998 (1)
|
|
|
|
|
|
3.8
|
|
Amended and Restated Articles of Incorporation changing name from Remedent, USA, Inc. to Remedent, Inc. and to effect a one-for-twenty reverse stock split on June 3, 2005 (2)
|
|
|
|
|
|
3.9
|
|
Amended and Restated Bylaws (2)
|
|
|
|
|
|
4.1
|
|
Specimen of Stock Certificate (3)
|
|
|
|
|
|
10.1
|
|
Incentive and Nonstatutory Stock Option Plan, dated May 29, 2001 (1)
|
|
|
|
|
|
10.2
|
|
2004 Incentive and Nonstatutory Stock Option Plan (4)
|
|
|
|
|
|
10.3
|
|
Voting Agreement between Remedent, Inc., and Robin List, dated December 10, 2008 (5)
|
|
|
|
|
|
10.4
|
|
Distribution, License and Manufacturing Agreement dated as of
January 20, 2012, (the “Effective Date”) by and among Remedent, Inc., a Nevada corporation (“Remedent Nevada”),
Remedent N.V., a company incorporated under the laws of Belgium (“Remedent Belgium) and GlamSmile Dental Technology, Ltd.,
a company incorporated under the laws of the Cayman Islands *
|
|
10.5
|
|
Termination and License Agreement dated March 27, 2012*
|
|
14.1
|
|
Code of Ethics, adopted March 25, 2003 (22)
|
|
|
|
|
|
21.1
|
|
List of Subsidiaries*
|
|
|
|
|
|
|
31.1
|
|
Certifications of the Chief Executive Officer under Section 302 of the Sarbanes-Oxley Act.*
|
|
|
|
|
|
|
|
31.2
|
|
Certifications of the Chief Financial Officer under Section 302 of the Sarbanes-Oxley Act.*
|
|
|
|
|
|
|
|
32.1
|
|
Certifications of the Chief Executive Officer under Section 906 of the Sarbanes-Oxley Act.*
|
|
|
32.2
|
|
Certifications of the Chief Financial Officer under Section
906 of the Sarbanes-Oxley Act.*
|
|
|
101.INS
|
|
XBRL Instance Document
(7)
|
|
|
101.SCH
|
|
XBRL Taxonomy Extension Schema
(7)
|
|
|
101.CAL
|
|
XBRL Taxonomy Extension Calculation Linkbase
(7)
|
|
|
101.DEF
|
|
XBRL Taxonomy Extension Definition Linkbase
(7)
|
|
|
101.LAB
|
|
XBRL Taxonomy Extension Label Linkbase
(7)
|
|
|
101.FRE
|
|
XBRL Taxonomy Extension Presentation Linkbase
(7)
|
|
*
|
Filed herewith
|
|
|
|
|
|
|
(1)
|
Incorporated by reference from Registration Statement on Form SB-2 filed with the SEC on July 24, 2002.
|
|
|
|
|
|
|
(2)
|
Incorporated by reference from Form 8-K filed with the SEC on June 8, 2005.
|
|
|
|
|
|
|
(3)
|
Incorporated by reference from Form SB-2 filed with the SEC on August 4, 2005.
|
|
|
|
|
|
|
(4)
|
Incorporated by reference from Form SB-2/A filed with the SEC on October 26, 2005.
|
|
|
|
|
|
|
(5)
|
Incorporated by reference from Form 8-K filed with the SEC on December 16, 2008.
|
|
|
|
|
(6)
|
Incorporated by reference from Form 10-KSB filed with the SEC on July 15, 2003.
|
|
(7)
|
XBRL (Extensible Business Reporting Language) information is furnished and not filed or a part of a registration statement or prospectus for purposes of sections 11 or 12 of the Securities Act of 1933, as amended, is deemed not filed for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, and otherwise is not subject to liability under these sections.
|
REMEDENT, INC. AND SUBSIDIARIES
CONSOLIDATED FINANCIAL STATEMENTS
MARCH 31, 2012
Index
|
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
|
F-1
|
|
|
|
|
CONSOLIDATED BALANCE SHEETS
|
F-2
|
|
|
|
|
CONSOLIDATED STATEMENTS OF OPERATIONS
|
F-3
|
|
|
|
|
CONSOLIDATED STATEMENT OF STOCKHOLDERS’ EQUITY (DEFICIT)
|
F-4
|
|
|
|
|
CONSOLIDATED STATEMENTS OF CASH FLOWS
|
F-5
|
|
|
|
|
CONSOLIDATED STATEMENTS OF COMPREHENSIVE INCOME (LOSS)
|
F-6
|
|
|
|
|
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
|
F-7
|
INDEPENDENT AUDITORS’ REPORT
REPORT OF INDEPENDENT REGISTERED ACCOUNTING
FIRM
To the Board of Directors and Stockholders
of Remedent, Inc.
We have audited the accompanying consolidated
balance sheets of Remedent, Inc. as of March 31, 2012 and March 31, 2011 and the related consolidated statements of operations,
stockholders’ equity, cash flows, and comprehensive loss for the years then ended. These consolidated financial statements
are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidated financial
statements based on our audits.
We conducted our audits in accordance with
the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform
an audit to obtain reasonable assurance whether the financial statements are free of material misstatement. The Company is not
required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audits included
consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the
circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over
financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting
the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made
by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable
basis for our opinion.
In our opinion, the accompanying consolidated
financial statements present fairly, in all material respects, the financial position of the Company at March 31, 2012 and March
31, 2011 and the results of its operations and its cash flows for the years then ended in conformity with accounting principles
generally accepted in the United States of America.
The accompanying consolidated financial
statements have been prepared assuming Remedent, Inc. will continue as a going concern. As described in note 1, at March 31, 2012
the financial situation of the Company has been affected by the losses carried forward and the increased pressure on the operating
cash flows. This going concern assumption is appropriate on the condition that sufficient funding will be realized to support the
Company’s operations. Without devaluing the unqualified opinion expressed above, we want to draw your attention to Note 1
which, the Board of Directors comments the risks regarding the going concern perspective. The consolidated financial statements
do not include any adjustments relating to the recoverability and classification of recorded asset amounts or amounts and classification
of liabilities that might be necessary should the company be unable to continue in existence.
Brussels - Belgium, July 16, 2012
PKF bedrijfsrevisoren CVBA
Statutory Auditors
Represented by
|
/s/ Gunther Loits
|
|
|
Registered Auditor
|
|
Tel +32 (0)2 242 11 40 / Fax +32 (0)2 242 03 45 / glo@pkf.be
/ www.pkf.be
PKF bedrijfsrevisoren CVBA / burgerlijke vennootschap met handelsvorm
Metrologielaan 10, bus 15 / 1130 Brussel / BTW BE 0439 814 826
/ RPR Antwerpen
PKF bedrijfsrevisoren CVBA is a member
firm of the PKF International Limited network of legally independent firms.
REMEDENT, INC. AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
|
|
|
March 31, 2012
|
|
|
March 31, 2011
|
|
|
ASSETS
|
|
|
|
|
|
|
|
|
|
CURRENT ASSETS:
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
203,584
|
|
|
$
|
1,662,520
|
|
|
Accounts receivable, net of allowance for doubtful accounts of $12,361 at March 31, 2012 and $28,975 at March 31, 2011
|
|
|
838,240
|
|
|
|
2,764,651
|
|
|
Inventories, net
|
|
|
1,077,766
|
|
|
|
2,164,046
|
|
|
Prepaid expense
|
|
|
1,149,264
|
|
|
|
762,953
|
|
|
Total current assets
|
|
|
3,268,854
|
|
|
|
7,354,170
|
|
|
PROPERTY AND EQUIPMENT, NET
|
|
|
641,220
|
|
|
|
1,401,735
|
|
|
OTHER ASSETS
|
|
|
|
|
|
|
|
|
|
Investment in GlamSmile Asia Ltd
|
|
|
2,092,518
|
|
|
|
—
|
|
|
Investment in Remedent OTC BV (Note 3)
|
|
|
962,505
|
|
|
|
—
|
|
|
Patents, net
|
|
|
—
|
|
|
|
166,746
|
|
|
Goodwill (Note 3)
|
|
|
—
|
|
|
|
699,635
|
|
|
Total assets
|
|
$
|
6,965,097
|
|
|
$
|
9,622,286
|
|
|
LIABILITIES AND STOCKHOLDERS’ EQUITY (DEFICIT)
|
|
|
|
|
|
|
|
|
|
CURRENT LIABILITIES:
|
|
|
|
|
|
|
|
|
|
Current portion, long term debt
|
|
$
|
863,501
|
|
|
$
|
184,679
|
|
|
Line of Credit
|
|
|
1,079,263
|
|
|
|
2,160,674
|
|
|
Short term loan (Note 4)
|
|
|
—
|
|
|
|
400,000
|
|
|
Accounts payable
|
|
|
1,009,176
|
|
|
|
1,744,253
|
|
|
Accrued liabilities
|
|
|
536,331
|
|
|
|
1,256,148
|
|
|
Deferred revenue
|
|
|
57,254
|
|
|
|
475,250
|
|
|
Due to related parties (Note 3)
|
|
|
61,836
|
|
|
|
95,354
|
|
|
Total current liabilities
|
|
|
3,607,361
|
|
|
|
6,316,358
|
|
|
Long term debt less current portion
|
|
|
1,452,523
|
|
|
|
273,557
|
|
|
Total liabilities
|
|
|
5,059,884
|
|
|
|
6,589,915
|
|
|
|
|
|
|
|
|
|
|
|
|
EQUITY:
|
|
|
|
|
|
|
|
|
|
Preferred Stock $0.001 par value (10,000,000 shares authorized, none issued and outstanding)
|
|
|
—
|
|
|
|
—
|
|
|
Common stock, $0.001 par value; (50,000,000 shares authorized, 19,995,969 shares issued and outstanding at March 31, 2012 and March 31, 2011 respectively)
|
|
|
19,996
|
|
|
|
19,996
|
|
|
Treasury stock, at cost; 723,000 shares outstanding at March 31, 2012 and March 31, 2011 respectively
|
|
|
(831,450
|
)
|
|
|
(831,450
|
)
|
|
Additional paid-in capital
|
|
|
24,906,269
|
|
|
|
24,855,883
|
|
|
Accumulated deficit
|
|
|
(20,622,635
|
)
|
|
|
(21,113,118
|
)
|
|
Accumulated other comprehensive income (loss) (foreign currency translation adjustment)
|
|
|
(1,681,829
|
)
|
|
|
(834,949
|
)
|
|
Obligation to issue shares (Note 3)
|
|
|
97,500
|
|
|
|
97,500
|
|
|
Total Remedent, Inc. stockholders’ equity
|
|
|
1,887,851
|
|
|
|
2,193,862
|
|
|
Non-controlling interest
|
|
|
17,362
|
|
|
|
838,509
|
|
|
Total stockholders’ equity
|
|
|
1,905,213
|
|
|
|
3,032,371
|
|
|
Total liabilities and equity
|
|
$
|
6,965,097
|
|
|
$
|
9,622,286
|
|
COMMITMENTS (Note 21)
The accompanying notes are an integral part
of these consolidated financial statements.
REMEDENT, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF OPERATIONS
|
|
|
For the years ended
March 31,
|
|
|
|
|
2012
|
|
|
2011
|
|
|
Net sales
|
|
$
|
9,687,292
|
|
|
$
|
12,581,708
|
|
|
Cost of sales
|
|
|
2,227,891
|
|
|
|
3,947,699
|
|
|
Gross profit
|
|
|
7,459,401
|
|
|
|
8,634,009
|
|
|
Operating Expenses
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
309,939
|
|
|
|
429,244
|
|
|
Sales and marketing
|
|
|
2,416,812
|
|
|
|
2,220,686
|
|
|
General and administrative
|
|
|
4,686,347
|
|
|
|
4,972,982
|
|
|
Depreciation and amortization
|
|
|
548,662
|
|
|
|
779,883
|
|
|
TOTAL OPERATING EXPENSES
|
|
|
7,961,760
|
|
|
|
8,402,795
|
|
|
OPERATING INCOME (LOSS)
|
|
|
(502,359
|
)
|
|
|
231,214
|
|
|
NON-OPERATING (EXPENSE) INCOME
|
|
|
|
|
|
|
|
|
|
Gain on sale of asset (Note 3)
|
|
|
2,085,669
|
|
|
|
—
|
|
|
Equity income from investments
|
|
|
173,337
|
|
|
|
—
|
|
|
Impairment of long-term investments and advances (Note 11 )
|
|
|
—
|
|
|
|
(750,000
|
)
|
|
Interest expense
|
|
|
(387,292
|
)
|
|
|
(260,235
|
)
|
|
Other income
|
|
|
244,276
|
|
|
|
159,289
|
|
|
TOTAL OTHER INCOME (EXPENSES)
|
|
|
2,115,990
|
|
|
|
(850,946
|
)
|
|
PROFIT (LOSS) FROM CONTINUING OPERATIONS BEFORE INCOME TAXES
|
|
|
1,613,650
|
|
|
|
(619,732
|
)
|
|
Income tax expense
|
|
|
(315,069
|
)
|
|
|
(220,297
|
)
|
|
NET INCOME (LOSS)
|
|
|
1,298,561
|
|
|
|
(840,029
|
)
|
|
NET INCOME ATTRIBUTABLE TO NON-CONTROLLING INTERESTS
|
|
|
(808,078
|
)
|
|
|
(707,146
|
)
|
|
NET INCOME (LOSS) ATTRIBUTABLE TO REMEDENT INC. Common Stockholders
|
|
$
|
490,483
|
|
|
$
|
(1,547,175
|
)
|
|
PROFIT (LOSS) PER SHARE
|
|
|
|
|
|
|
|
|
|
Basic
|
|
$
|
0.02
|
|
|
$
|
(0.08
|
)
|
|
Fully diluted
|
|
$
|
0.02
|
|
|
$
|
(0.08
|
)
|
|
WEIGHTED AVERAGE SHARES OUTSTANDING
|
|
|
|
|
|
|
|
|
|
Basic
|
|
|
19,995,969
|
|
|
|
19,995,969
|
|
|
Fully diluted
|
|
|
20,019,969
|
|
|
|
19,995,969
|
|
The accompanying notes are an integral part
of these consolidated financial statements.
REMEDENT, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENT OF STOCKHOLDERS’
EQUITY (DEFICIT)
FOR THE YEARS ENDED MARCH 31, 2012
and 2011
|
|
|
Shares
|
|
|
Amount
|
|
|
Additional
Paid in
Capital
|
|
|
Obligation to
Issue
Shares
|
|
|
Accumulated
Deficit
|
|
|
Treasury
Stock
|
|
|
Other
Comprehensive
Income (Loss)
|
|
|
Total
|
|
|
Non-
controlling
Interest
(net of OCI)
|
|
|
Total
|
|
|
|
|
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, March 31, 2010
|
|
|
19,995,969
|
|
|
|
19,996
|
|
|
|
24,742,201
|
|
|
|
97,500
|
|
|
|
(19,565,943
|
)
|
|
|
(831,450
|
)
|
|
|
(650,059
|
)
|
|
|
3,812,245
|
|
|
|
114,002
|
|
|
|
3,926,247
|
|
|
Value of stock options issued to employees
|
|
|
—
|
|
|
|
—
|
|
|
|
113,682
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
113,682
|
|
|
|
—
|
|
|
|
113,682
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Unrealized foreign exchange gain (loss)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(184,890
|
)
|
|
|
(184,890
|
)
|
|
|
17,361
|
|
|
|
(167,529
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net (loss) income for the year
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,547,175
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,547,175
|
)
|
|
|
707,146
|
|
|
|
(840,029
|
)
|
|
Balance, March 31, 2011
|
|
|
19,995,969
|
|
|
|
19,996
|
|
|
|
24,855,883
|
|
|
|
97,500
|
|
|
|
(21,113,118
|
)
|
|
|
(831,450
|
)
|
|
|
(834,949
|
)
|
|
|
2,193,862
|
|
|
|
838,509
|
|
|
|
3,032,371
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Value of stock options issued to employees
|
|
|
—
|
|
|
|
—
|
|
|
|
50,386
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
50,386
|
|
|
|
—
|
|
|
|
50,386
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deconsolidation
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(1,629,225
|
)
|
|
|
(1,629,225
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Unrealized foreign exchange gain (loss)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(846,880
|
)
|
|
|
(846,880
|
)
|
|
|
-
|
|
|
|
(846,880
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net income for the year
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
490,483
|
|
|
|
—
|
|
|
|
—
|
|
|
|
490,483
|
|
|
|
808,078
|
|
|
|
1,298,561
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, March 31, 2012
|
|
|
19,995,969
|
|
|
|
19,996
|
|
|
|
24,906,269
|
|
|
|
97,500
|
|
|
|
(20,622,635
|
)
|
|
|
(831,450
|
)
|
|
|
(1,681,829
|
)
|
|
|
1,887,851
|
|
|
|
17,362
|
|
|
|
1,905,213
|
|
The accompanying notes are an integral part
of these consolidated financial statements.
REMEDENT, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
|
|
|
For the year ended March 31,
|
|
|
|
|
2012
|
|
|
2011
|
|
|
CASH FLOWS FROM OPERATING ACTIVITIES
|
|
|
|
|
|
|
|
|
|
Net Profit (loss)
|
|
$
|
1,298,561
|
|
|
$
|
(840,029
|
)
|
|
Adjustments to reconcile net (loss) to net cash used by operating activities:
|
|
|
|
|
|
|
|
|
|
Gain on Sale Asia (note3)
|
|
|
(1,470,776
|
)
|
|
|
—
|
|
|
Non cash revenue
|
|
|
(475,250
|
)
|
|
|
—
|
|
|
Impairment of long-term investments and advances
|
|
|
—
|
|
|
|
750,000
|
|
|
Depreciation and amortization
|
|
|
548,662
|
|
|
|
779,883
|
|
|
Inventory reserve
|
|
|
25,832
|
|
|
|
48,072
|
|
|
Allowance for doubtful accounts
|
|
|
(16,614
|
)
|
|
|
(36,870
|
)
|
|
Value of stock options issued to employees and consultants
|
|
|
50,387
|
|
|
|
113,682
|
|
|
Investment income
|
|
|
(173,337
|
)
|
|
|
—
|
|
|
Changes in operating assets and liabilities:
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
790,249
|
|
|
|
(1,957,720
|
)
|
|
Inventories
|
|
|
228,790
|
|
|
|
(2,354
|
)
|
|
Prepaid expenses
|
|
|
(804,000
|
)
|
|
|
157,534
|
|
|
Accounts payable
|
|
|
(171,692
|
)
|
|
|
(214,156
|
)
|
|
Accrued liabilities
|
|
|
(626,896
|
)
|
|
|
764,612
|
|
|
Due to related parties
|
|
|
61,836
|
|
|
|
(173,130
|
)
|
|
Accrued and unpaid interest on debt
|
|
|
42,467
|
|
|
|
—
|
|
|
Deferred revenue
|
|
|
582,004
|
|
|
|
475,250
|
|
|
Net cash used by operating activities
|
|
|
(109,777
|
)
|
|
|
(135,226
|
)
|
|
CASH FLOWS FROM INVESTING ACTIVITIES
|
|
|
|
|
|
|
|
|
|
Cash divested on deconsolidation of Remedent OTC BV
|
|
|
(2,372,445
|
)
|
|
|
—
|
|
|
Cash divested on deconsolidation of GlamSmile Asia
|
|
|
(1,044,674
|
)
|
|
|
—
|
|
|
Purchases of patent rights
|
|
|
(101,759
|
)
|
|
|
(51,233
|
)
|
|
Purchases of equipment
|
|
|
(321,170
|
)
|
|
|
(481,768
|
)
|
|
Net cash used by investing activities
|
|
|
(3,840,048
|
)
|
|
|
(533,001
|
)
|
|
CASH FLOWS FROM FINANCING ACTIVITIES
|
|
|
|
|
|
|
|
|
|
Short term loan
|
|
|
1,000,000
|
|
|
|
400,000
|
|
|
Proceeds from issuance of GlamSmile Asia preferred stock (Note 3)
|
|
|
2,000,000
|
|
|
|
—
|
|
|
Repayments of capital lease note payable
|
|
|
(61,527
|
)
|
|
|
(183,135
|
)
|
|
Advances (repayments) of line of credit
|
|
|
(414,061
|
)
|
|
|
1,486,074
|
|
|
Net cash provided by financing activities
|
|
|
2,524,412
|
|
|
|
1,702,939
|
|
|
|
|
|
|
|
|
|
|
|
|
NET INCREASE (DECREASE) IN CASH
|
|
|
(1,466,188
|
)
|
|
|
1,034,712
|
|
|
Effect of exchange rate changes on cash and cash equivalents
|
|
|
(33,523
|
)
|
|
|
14,342
|
|
|
CASH AND CASH EQUIVALENTS, BEGINNING
|
|
|
1,662,520
|
|
|
|
613,466
|
|
|
CASH AND CASH EQUIVALENTS, ENDING
|
|
$
|
203,584
|
|
|
$
|
1,662,520
|
|
|
Supplemental Cash Flow Information
:
|
|
|
|
|
|
|
|
|
|
Interest paid
|
|
$
|
252,208
|
|
|
$
|
133,397
|
|
|
Income taxes paid
|
|
$
|
—
|
|
|
$
|
—
|
|
|
SUPPLEMENTAL NON-CASH FINANCING AND INVESTING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deferred revenue converted to loan (Note 14)
|
|
$
|
500,000
|
|
|
$
|
—
|
|
The accompanying notes are an integral part
of these consolidated financial statements.
REMEDENT, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF COMPREHENSIVE
INCOME (LOSS)
|
|
|
For the year ended
March 31,
|
|
|
|
|
2012
|
|
|
2011
|
|
|
Net Profit(Loss) attributable to Remedent, Inc. common shareholders
|
|
$
|
490,483
|
|
|
$
|
(1,547,175
|
)
|
|
OTHER COMPREHENSIVE INCOME (LOSS):
|
|
|
|
|
|
|
|
|
|
Foreign currency translation adjustment
|
|
|
(846,880
|
)
|
|
|
(184,890
|
)
|
|
Total Other Comprehensive Profit (Loss)
|
|
|
(356,397
|
)
|
|
|
(1,732,065
|
)
|
|
|
|
|
|
|
|
|
|
|
|
LESS: COMPREHENSIVE INCOME ATTRIBUTABLE TO NON-CONTROLLING INTEREST
|
|
|
—
|
|
|
|
21,996
|
|
|
|
|
|
|
|
|
|
|
|
|
COMPREHENSIVE PROFIT(LOSS) ATTRIBUTABLE TO REMEDENT, INC. common shareholders
|
|
$
|
(356,397
|
)
|
|
$
|
(1,710,069
|
)
|
The accompanying notes are an integral part
of these consolidated financial statements.
REMEDENT, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
1.
DESCRIPTION OF THE COMPANY AND BASIS OF PRESENTATION
The Company is a manufacturer and distributor
of cosmetic dentistry products, including a full line of professional dental and retail “Over-The-Counter” tooth whitening
products which are distributed in Europe, Asia and the United States. The Company manufactures many of its products in its facility
in Deurle, Belgium as well as outsourced manufacturing in its facility in Beijing, China. The Company distributes its
products using both its own internal sales force and through the use of third party distributors.
The Company’s financial statements
have been prepared on an accrual basis of accounting, in conformity with accounting principles generally accepted in the United
States of America.
Despite the net profit of the current accounting
year, the accumulated losses of the past affect the financial situation of the company. Moreover there has been increased pressure
on the operating cash flows of the Company and by consequence additional funding will be required to support the Company’s
operations and the execution of the business plan. These risks, among others, are also discussed in ITEM 1A –RISK FACTORS.
Despite these matters of emphasis, the financial statements have been prepared on a going concern basis.
In these notes, the terms “Remedent”,
“Company”, “we”, “us” or “our” mean Remedent, Inc. and all of its subsidiaries,
whose operations are included in these consolidated financial statements.
The Company has conducted a subsequent
events review through the date the financial statements were issued, and has concluded that there were no subsequent events requiring
adjustments or additional disclosures to the Company's financial statements at March 31, 2012.
2.
SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Organization and Principles of Consolidation
The accompanying consolidated financial
statements include the accounts of: Remedent N.V. (incorporated in Belgium) located in Ghent , Belgium, Remedent Professional,
Inc. and Remedent Professional Holdings, Inc. (both incorporated in California and inactive), Glamtech-USA, Inc. (a Delaware corporation
acquired effective August 24, 2008), Remedent N.V.’s 51% owned subsidiary, GlamSmile Deutschland GmbH, a German
private company located in Munich and Remedent N.V.’s 100 % owned subsidiary, GlamSmile Rome, a Italian private company located
in Rome.
Remedent Inc.’s 50% owned subsidiary,
Remedent OTC B.V. (a Dutch Holding company) which owns a 75% interest in Sylphar Holding B.V. (a Dutch holding company) through
which ownership Remedent Inc. ultimately has a 37.5% ownership interest in Sylphar N.V., a 100% owned company by Sylphar Holding
BV, Sylphar USA, a 100% owned Nevada corporation by Sylphar Holding BV. and Sylphar Asia Pte Ltd. , formerly Remedent Asia Pte
Ltd. a 100% owned Singapore company owned by Sylphar Holding BV, is accounted for using the equity method after September 30, 2011(see
Note 3 – Long-term Investment).
Remedent Inc.’s 29,4 % investment in Glamsmile Dental
Technology Ltd., a Cayman Islands company (“Glamsmile Dental”) and its subsidiaries, Glamsmile (Asia) Limited, a company
organized and existing under the laws of Hong Kong and a substantially 100 % owned subsidiary of Glamsmile Dental, Beijing Glamsmile
Technology Development Ltd., a 100 % owned subsidiary or GlamSmile Asia, its 80% owned subsidiary Beijing Glamsmile Trading Co.,
Ltd. and its 98% owned subsidiary Beijing Glamsmile Dental Clinic Co., Ltd., including its 100% Shanghai Glamsmile Dental Clinic
Co., Ltd., and its 50 % owned Whenzhou GlamSmile Dental Clinic Ltd., which are accounted for using the equity method after January
31, 2012 (see Note 3 – Long-term Investment)
Remedent, Inc. is a holding company with
headquarters in Ghent, Belgium. Remedent Professional, Inc. and Remedent Professional Holdings, Inc. have been dormant since inception.
The rebranded Sylphar Asia Pte. Ltd. (formerly Remedent Asia Pte. Ltd.), commenced operations as of July 2005.
For all periods presented, all significant
inter-company accounts and transactions have been eliminated in the consolidated financial statements and corporate administrative
costs are not allocated to subsidiaries.
Pervasiveness of Estimates
The preparation of financial statements
in conformity with accounting principles generally accepted in the United States requires the Company to make estimates and assumptions
that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the
financial statements and the reported amounts of revenues and expenses during the reporting period. On an on-going basis, the Company
evaluates estimates and judgments, including those related to revenue, bad debts, inventories, fixed assets, intangible assets,
stock based compensation, income taxes, and contingencies. Estimates are based on historical experience and on various other assumptions
that the Company believes reasonable in the circumstances. The results form the basis for making judgments about the carrying values
of assets and liabilities that are not readily apparent from other sources. Actual results could differ from those estimates.
Revenue Recognition
The Company recognizes revenue from product
sales when persuasive evidence of a sale exists: that is, a product is shipped under an agreement with a customer; risk of loss
and title has passed to the customer; the fee is fixed or determinable; and collection of the resulting receivable is reasonably
assured. Sales allowances are estimated based upon historical experience of sales returns.
Revenues from product sales are recognized when the product
is shipped and title and risk of loss has passed to the customer, typically upon delivery and when the quantity and price is fixed
and determinable, and when collectability is reasonable assured.
Upfront fees are recognized upon the date of the agreement (i.e.
point of sale) because they relate solely to the sale of territories (that are sold in perpetuity), are non-refundable, and are
not contingent upon additional deliverables.
We have evaluated all deliverables in our
contracts (per ASC 605-25-5) ((a) territory & (b) manufacturing/marketing training & development fees) and determined that
they are separate, as follows:
|
|
·
|
Both (a) & (b) have value to our customers on a standalone basis and can be sold by our customers
separately.
|
|
|
·
|
Delivery or performance of the undelivered item or items is considered probable and substantially
in our control.
|
Our development fees/milestone payments
are recognized in accordance with the Milestone Method pursuant to FASB ASC 605. Revenues from milestones related to
an arrangement under which we have continuing performance obligations i.e. specifically scheduled training and development activities,
if deemed substantive, are recognized as revenue upon achievement of the milestone. Milestones are considered substantive if all
of the following conditions are met: (a) the milestone is non-refundable; (b) achievement of the milestone was not reasonably assured
at the inception of the arrangement; (c) substantive effort is involved to achieve the milestone; and (d) the amount of the milestone
appears reasonable in relation to the effort expended. If any of these conditions is not met, the milestone payment is deferred
and recognized as revenue as we complete our performance obligations.
We receive royalty revenues under license
agreements with third parties that sell products based on technology developed by us or to which we have rights. The license agreements
provide for the payment of royalties to us based on sales of the licensed product. We record these revenues as earned monthly,
based on reports from our licensees.
Shipping and Handling
Shipping and handling costs are included
as a component of cost of sales. Shipping and handling costs billed to customers are included in sales.
Impairment of Long-Lived Assets
Long-lived assets consist primarily of
patents and property and equipment. The recoverability of long-lived assets is evaluated by an analysis of operating results and
consideration of other significant events or changes in the business environment. If impairment exists, the carrying amount of
the long-lived assets is reduced to its estimated fair value, less any costs associated with the final settlement. To
date, management has not identified any impairment of property and equipment. There can be no assurance, however, that
market conditions or demands for the Company’s services will not change which could result in future long-lived asset impairment.
Goodwill
Goodwill is not amortized, but is tested
for impairment on an annual basis as of March 31, or whenever events or circumstances indicate that the carrying amount may not
be recoverable. These impairment tests are based upon a comparison of the fair value of the reporting units to their respective
carrying amount. If the carrying amount of the reporting unit exceeds its fair value, the goodwill impairment loss is measured
as the excess of the carrying amount of goodwill over its implied fair value. As of March 31, 2012 the goodwill is no longer recorded
because the Asia branch is no longer fully consolidated. Furthermore the goodwill is retained in the gain on sale calculation (see
Note 3)
On April 1, 2010, the Company adopted the
new accounting guidance for business combinations according to FASB Codification ASC 805,
Business Combinations
. This guidance
establishes principles and requirements for the reporting entity in a business combination, including recognition and measurement
in the financial statements of the identifiable assets acquired, the liabilities assumed, goodwill, and any noncontrolling interest
in the acquiree, as well as disclosure requirements to enable financial statement users to evaluate the nature and financial effects
of the business combination. Additionally, it provides guidance for identifying a business combination, measuring the acquisition
date, and defining the measurement period for adjusting provisional amounts recorded. The implementation of this standard did not
have an impact on the Company’s consolidated financial statements.
Cash and Cash Equivalents
The Company considers all highly liquid
investments with maturities of three months or less at the time of purchase to be cash or cash equivalents.
Non-Controlling Interest
The Company adopted ASC Topic 810
Noncontrolling
Interests in Consolidated Financial Statements
— an Amendment of Accounting Research Bulletin No. 51
as of
April 1, 2009. SFAS 160 establishes accounting and reporting standards for ownership interests in subsidiaries held by parties
other than the parent, the amount of consolidated net income attributable to the parent and to the noncontrolling interest, changes
in a parent’s ownership interest and the valuation of retained noncontrolling equity investments when a subsidiary is deconsolidated.
ASC Topic 810 also establishes reporting requirements that provide sufficient disclosures that clearly identify and distinguish
between the interest of the parent and the interests of the noncontrolling owner. The adoption of ASC Topic 810 impacted the presentation
of our consolidated financial position, results of operations and cash flows.
Fair Value of Financial Instruments
The Company’s financial instruments
consist of cash and cash equivalents, accounts receivable, accounts payable, accrued liabilities, line of credit, short term and
long-term debt. The carrying amounts of cash and cash equivalents, accounts receivable, accounts payable and accrued liabilities
approximate their respective fair values because of the short maturities of those instruments. The Company’s long-term debt
consists of its revolving credit facility and long-term capital lease obligations. The carrying value of the revolving credit facility
approximates fair value because of its variable short-term interest rates. The fair value of the Company’s long-term
capital lease obligations is based on current rates for similar financing.
Accounts Receivable and Allowance for Doubtful
Accounts
The Company sells professional dental equipment
to various companies, primarily to distributors located in Western Europe and the United States of America, and China. The terms
of sales vary by customer, however, generally are 2% 10 days, net 30 days. Accounts receivable is reported at net realizable value
and net of allowance for doubtful accounts. The Company uses the allowance method to account for uncollectible accounts receivable.
The Company’s estimate is based on historical collection experience and a review of the current status of trade accounts
receivable.
Inventories
The Company purchases certain of its products
in components that require assembly prior to shipment to customers. All other products are purchased as finished goods ready to
ship to customers.
The Company writes down inventories for
estimated obsolescence to estimated market value based upon assumptions about future demand and market conditions. If actual market
conditions are less favorable than those projected, then additional inventory write-downs may be required. Inventory reserves for
obsolescence totaled $156,239 at March 31, 2012 and $130,407 at March 31, 2011.
Prepaid Expense
The Company’s prepaid expense consists
of prepayments to suppliers for inventory purchases and to the Belgium customs department, to obtain an exemption of direct VAT
payments for imported goods out of the European Union (“EU”). This prepayment serves as a guarantee to obtain the facility
to pay VAT at the moment of sale and not at the moment of importing goods at the border. Prepaid expenses also include VAT payments
made for goods and services in excess of VAT payments received from the sale of products as well as amounts for other prepaid operating
expenses.
Property and Equipment
Property and equipment are stated at cost.
Major renewals and improvements are charged to the asset accounts while replacements, maintenance and repairs, which do not improve
or extend the lives of the respective assets, are expensed. At the time property and equipment are retired or otherwise disposed
of, the asset and related accumulated depreciation accounts are relieved of the applicable amounts. Gains or losses from retirements
or sales are credited or charged to income.
The Company depreciates its property and
equipment for financial reporting purposes using the straight-line method based upon the following useful lives of the assets:
|
Tooling
|
3 Years
|
|
Furniture and fixtures
|
4 Years
|
|
Machinery and Equipment
|
4 Years
|
Patents
Patents consist of the costs incurred to
purchase patent rights and are reported net of accumulated amortization. Patents are amortized using the straight-line method over
a period based on their contractual lives.
Research and Development Costs
The Company expenses research and development
costs as incurred.
Advertising
Costs incurred for producing and communicating
advertising are expensed when incurred and included in sales and marketing and general and administrative expenses. For the years
ended March 31, 2012 and March 31, 2011, advertising expense was $590,285 and 709,885, respectively.
Income taxes
Income taxes are accounted for under the
asset and liability method as stipulated by Accounting Standards Codification (“ASC”) 740 formerly Statement of Financial
Accounting Standards (“SFAS”) No. 109, “Accounting for Income Taxes”. Deferred tax assets and liabilities
are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of
existing assets and liabilities and their respective tax bases and operating loss and tax credit carry forwards. Deferred tax assets
and liabilities are measured using enacted tax rates expected to apply to taxable income in the year in which those temporary differences
are expected to be recovered or settled. Under ASC 740, the effect on deferred tax assets and liabilities or a change in tax rate
is recognized in income in the period that includes the enactment date. Deferred tax assets are reduced to estimated amounts to
be realized by the use of a valuation allowance. A valuation allowance is applied when in management’s view it is more likely
than not (50%) that such deferred tax will not be utilized.
Effective February 1, 2008, the Company
adopted certain provisions under ASC Topic 740, Income Taxes, (“ASC 740”), which provide interpretative guidance for
the financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return. Effective
with the Company’s adoption of these provisions, interest related to the unrecognized tax benefits is recognized in the financial
statements as a component of income taxes. The adoption of ASC 740 did not have an impact on the Company’s financial position
and results of operations.
In the unlikely event that an uncertain
tax position exists in which the Company could incur income taxes, the Company would evaluate whether there is a probability that
the uncertain tax position taken would be sustained upon examination by the taxing authorities. Reserves for uncertain tax position
would then be recorded if the Company determined it is probable that a position would not be sustained upon examination or if a
payment would have to be made to a taxing authority and the amount is reasonably estimable. As of March 31, 2012, the Company does
not believe it has any uncertain tax positions that would result in the Company having a liability to the taxing authorities.
Warranties
The Company typically warrants its products
against defects in material and workmanship for a period of 18 months from shipment.
A tabular reconciliation of the Company’s
aggregate product warranty liability for the reporting period is as follows:
|
|
|
Year ended
March 31, 2012
|
|
|
Year ended
March 31, 2011
|
|
|
Product warranty liability:
|
|
|
|
|
|
|
|
|
|
Opening balance
|
|
$
|
21,260
|
|
|
$
|
20,238
|
|
|
Reductions for payments made
|
|
|
|
|
|
|
|
|
|
Accruals for product warranties issued in the period
|
|
|
(2,002
|
)
|
|
|
(11,620
|
)
|
|
Adjustments to liabilities for pre-existing warranties
|
|
|
761
|
|
|
|
12,642
|
|
|
Ending liability
|
|
$
|
20,019
|
|
|
$
|
21,260
|
|
Based upon historical trends and warranties
provided by the Company’s suppliers and sub-contractors, the Company has made a provision for warranty costs of $20,019 and
$21,260 as of March 31, 2012 and March 31, 2011, respectively.
Segment Reporting
“Disclosure About Segments of an
Enterprise and Related Information” requires use of the “management approach” model for segment reporting. The
management approach model is based on the way a company’s management organizes segments within the company for making operating
decisions and assessing performance. Reportable segments are based on products and services, geography, legal structure, management
structure, or any other manner in which management disaggregates a company. The Company’s management considers its business
to comprise one segment for reporting purposes.
Computation of Earnings (Loss) per Share
Basic net income (loss) per common share
is computed by dividing net income (loss) attributable to common stockholders by the weighted average number of shares of common
stock outstanding during the period. Net income (loss) per common share attributable to common stockholders assuming dilution is
computed by dividing net income by the weighted average number of shares of common stock outstanding plus the number of additional
common shares that would have been outstanding if all dilutive potential common shares had been issued.
On April 1, 2009, the Company adopted changes
issued by the FASB to the calculation of earnings per share. These changes state that unvested share-based payment awards that
contain non-forfeitable rights to dividends or dividend equivalents (whether paid or unpaid) are participating securities and shall
be included in the computation of earnings per share pursuant to the two-class method for all periods presented. The adoption of
this change had no impact on the Company’s basic or diluted net loss per share because the Company has never issued any share-based
awards that contain non-forfeitable rights.
At each of March 31, 2012 and 2011, the
Company had 19,995,969, shares of common stock issued and outstanding. At March 31, 2012 and 2011, the Company had 7,794,627
and 7,794,627 warrants outstanding, respectively and 1,895,000 and 2,157,500 options outstanding, respectively. All
outstanding warrants and options were excluded from the computation of earnings per share for the year ended March 31, 2012 because
their effect would have been anti-dilutive.
Further, pursuant to ASC 260-10-50-1(c),
if a fully diluted share calculation was computed for the years ended March 31, 2012 and 2011 respectively, it would have excluded
all warrants and all but 200,000 and 1,165,000 options respectively since the Company’s average share trading price during
the last two year period was less than the exercise price of all other warrants and options.
Conversion of Foreign Currencies
The reporting currency for the consolidated
financial statements of the Company is the U.S. dollar. The functional currency for the Company’s European subsidiaries,
Remedent N.V., Sylphar N.V., GlamSmile Rome and GlamSmile Deutschland GmbH, is the Euro, for Sylphar Asia Ptd Ltd the
Singapore Dollar and for Glamsmile Asia Ltd., and its subsidiaries, the Hong Kong dollar and the Chinese Renmimbi (“RMB”)
for Mainland China. Finally, the functional currency for Remedent Professional, Inc. is the U.S. dollar. The assets and liabilities
of companies whose functional currency is other that the U.S. dollar are included in the consolidation by translating the assets
and liabilities at the exchange rates applicable at the end of the reporting period. The statements of income of such companies
are translated at the average exchange rates during the applicable period. Translation gains or losses are accumulated as a separate
component of stockholders’ equity.
Comprehensive Income (Loss)
Comprehensive income (loss) includes all
changes in equity except those resulting from investments by owners and distributions to owners, including accumulated foreign
currency translation, and unrealized gains or losses on marketable securities.
The Company’s only component of other
comprehensive income is the accumulated foreign currency translation consisting of gains and (losses) of $(846,880) and $(184,890)
for the years ended March 31, 2012 and 2011, respectively. These amounts have been recorded as a separate component of stockholders’
equity (deficit).
Stock Based Compensation
The Company has a stock-based compensation
plan which is described more fully in Note 18. The Company measures the compensation cost of stock options and other stock-based
awards to employees and directors at fair value at the grant date and recognizes compensation expense over the requisite service
period for awards expected to vest.
Except for transactions with employees
and directors, all transactions in which goods or services are the consideration received for the issuance of equity instruments
are accounted for based on the fair value of the consideration received or the fair value of the equity instruments issued, whichever
is more reliably measurable. Additionally, the Company has determined that the dates used to value the transaction are either:
(1) The date at which a commitment for
performance by the counter party to earn the equity instruments is established; or
(2) The date at which the counter party’s
performance is complete.
New Accounting Pronouncements
Recently Adopted
In December 2011, the FASB issued ASU 2011-11,
“
Balance Sheet (Topic 210) – Disclosures about offsetting assets and liabilities
”. ASU 2011-11 requires
entities to disclose both gross and net information about both instruments and transactions eligible for offset in the statement
of financial position and instruments and transactions subject to an agreement similar to a master netting arrangement. This scope
would include derivatives, sale and repurchase agreements and reverse sale and repurchase agreements, and securities borrowing
and securities lending arrangements. This standard is applicable for annual reporting periods beginning on or after January 1,
2013, and interim periods within those annual periods. Retrospective disclosures will be required for all periods presented. The
adoption of ASU 2011-11 is not expected to have an impact upon the Company’s consolidated financial statements.
In October 2009, the FASB issued ASU 2009-13,
“Multiple-Deliverable Revenue Arrangements, (amendments to ASC Topic 605, Revenue Recognition)”
(“ASU
2009-13”). ASU 2009-13 requires entities to allocate revenue in an arrangement using estimated selling prices of the delivered
goods and services based on a selling price hierarchy. The amendments eliminate the residual method of revenue allocation and require
revenue to be allocated using the relative selling price method. The standard also expands the disclosure requirements for multiple
deliverable revenue arrangements. ASU 2009-13 should be applied on a prospective basis for revenue arrangements entered into or
materially modified in fiscal years beginning on or after June 15, 2010, which corresponds to the Company’s fiscal year beginning
April 1, 2011. The adoption of ASU 2009-13 has had no impact upon the Company’s consolidated financial statements.
In October 2009, the FASB issued ASU 2009-14,
Certain Revenue Arrangements that Include Software Elements – a consensus of the FASB Emerging Issues Task Force
,
which changes the accounting model for revenue arrangements that include both tangible products and software elements. This new
guidance removes from the scope of the software revenue recognition guidance in ASC 985-605, Software Revenue Recognition, those
tangible products containing software components and non-software components that function together to deliver the tangible product’s
essential functionality. In addition, this guidance requires that hardware components of a tangible product containing software
components always be excluded from the software revenue recognition guidance as well as provides further guidance on determining
which software, if any, relating to the tangible product also would be excluded from the scope of software revenue recognition
guidance. The guidance further identifies specific factors in determining whether the tangible product contains software that works
together with the non-software components of the tangible product to deliver the tangible product’s essential functions.
Guidance is also provided on how a vendor should allocate arrangement consideration to deliverables in an arrangement containing
both tangible products and software. The disclosures mandated in ASU 2009-13 are also required by this new guidance. ASU 2009-14
is effective prospectively for revenue arrangements entered into or materially modified in fiscal years beginning on or after June
15, 2010, which corresponds to the Company’s fiscal year beginning April 1, 2011. The adoption of ASU 2010-14 has had no
impact upon the Company’s consolidated financial statements.
In April 2010, the FASB issued ASU 2010-17
,” Revenue Recognition – Milestone Method”.
The amendments in this Update provide guidance on the criteria
that should be met for determining whether the milestone method of revenue recognition is appropriate. A vendor can recognize consideration
that is contingent upon achievement of a milestone in its entirety as revenue in the period in which the milestone is achieved
only if the milestone meets all criteria to be considered substantive. ASU 2010-17 should be applied on a prospective basis for
revenue arrangements entered into or materially modified in fiscal years beginning on or after June 15, 2010, which corresponds
to the Company’s fiscal year beginning April 1, 2011. The adoption of ASU 2010-17 has had no impact upon the Company’s
consolidated financial statements.
In May 2011, the FASB issued ASU 2011-04
and updated the accounting guidance related to fair value measurements. The updated guidance results in a consistent definition
of fair value and common requirements for measurement of and disclosure about fair value between U.S. GAAP and International Financial
Reporting Standards (IFRS). The updated guidance is effective for the Company beginning in the fourth quarter of fiscal year 2012.
The adoption of ASU 2011-04 has not had a material effect on the Company’s results of operations, financial condition, and
cash flows.
Recent Accounting Pronouncements
Not Yet Adopted
In June 2011, the FASB issued new accounting
guidance on comprehensive income that was amended in December 2011. The objective of this accounting guidance is to improve the
comparability, consistency and transparency of financial reporting and to increase the prominence of items reported in other comprehensive
income. The guidance eliminates the option to present components of other comprehensive income as part of the statement of stockholders’
equity and requires them to be presented in the statement of comprehensive income instead. This accounting guidance, as amended,
will be effective for the Company on April 1, 2012. The Company does not expect the impact of this new accounting guidance to have
a material impact on its financial condition or results of operations.
In September 2011, the FASB issued new
accounting guidance on testing goodwill for impairment. The primary objective of this accounting guidance is to reduce complexity
and costs by allowing an entity to make a qualitative evaluation about the likelihood of goodwill impairment to determine whether
it should calculate the fair value of a reporting unit. If, after assessing qualitative factors, an entity determines that it is
not more likely than not (a likelihood of more than
50 percent
) that the fair value of a reporting
unit is less than its carrying amount, then the two-step goodwill impairment test is unnecessary. This accounting guidance is effective
for the Company in fiscal 2013 but early adoption is permitted. The Company does not expect the impact of this new accounting guidance
to have a material impact on its financial condition or results of operations.
In December 2011, the FASB issued new accounting
guidance on disclosures about offsetting assets and liabilities. The requirements for offsetting are different under U.S. GAAP
and IFRS. Therefore, the objective of this accounting guidance is to facilitate comparison between financials statements prepared
under U.S. GAAP and IFRS by enhancing disclosures of the effect or potential effect of netting arrangements on an entity’s
financial position, including the effect or potential effect of rights of setoff associated with certain assets and liabilities.
This accounting guidance will be effective for the Company on April 1, 2013. The Company does not expect the impact of this new
accounting guidance to have a material impact on its financial condition or results of operations.
3
.
LONG-TERM INVESTMENTS
REMEDENT OTC BV
In connection with the restructuring of
the Company’s OTC business in December 2008, the Company controlled Remedent OTC BV until September 30, 2011 through its
board representations. As agreed upon in the Voting Agreement, after September 30, 2011, the Company had one board representation
and consequently no longer controlled its investment in Remedent OTC BV. As such, the financials of Remedent OTC BV are no longer
included in the consolidated Financial Statements but accounted for through the equity method. No gain or loss was recorded on
the deconsolidation. After September 30, 2011, the Company still owned 50% of Remedent OTC BV. For the six months ended March 31,
2012, the Company recorded equity income of $136,703 in “Other income” for its portion of the net income recorded by
Remedent OTC BV.
The following tables represent the summary financial information
of Remedent OTC BV as derived from its financial statements and prepared under GAAP:
|
|
|
Year ended
|
|
|
Year ended
|
|
|
Operating data:
|
|
March 31, 2012
|
|
|
March 31, 2011
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Revenues
|
|
$
|
2,465,876
|
|
|
$
|
5,840,322
|
|
|
Gross profit
|
|
|
1,617,779
|
|
|
|
3,524,959
|
|
|
Income (loss) from operations
|
|
|
93,273
|
|
|
|
590,945
|
|
|
Net income
|
|
$
|
103,998
|
|
|
$
|
548,045
|
|
GLAMSMILE ASIA LTD.
Acquisition
Effective January 1, 2010 the Company acquired
50.98% of the issued and outstanding shares of Glamsmile Asia Ltd. (“Glamsmile Asia” or “Glamsmile”), a
private Hong Kong company, with subsidiaries in Hong Kong and Mainland China, in exchange for the following consideration:
|
|
1.
|
325,000 Euro (US$466,725). As of March 31, 2011 the full amount was paid.
|
|
|
2.
|
250,000 shares of common stock to be issued during the fiscal year ended March 31, 2011($97,500
was recorded as an obligation to issue shares as at March 31, 2010). The parties have agreed that the shares will be
issued during fiscal year ended March 31, 2013.
|
|
|
3.
|
100,000 options on closing (issued);
|
|
|
4.
|
100,000 options per opened store at closing (issued);
|
|
|
5.
|
100,000 options for each additional store opened before the end of 2011 at the price of the opening
date of the store;
|
|
|
6.
|
Assumption of Glamsmile’s January 1, 2010 deficit of $73,302.; and
|
|
|
7.
|
Repayment of the founding shareholder’s original advances in the amount of $196,599. The
balance of $196,599, recorded as due to related parties at March 31, 2010, is unsecured, non-interest bearing and has no specific
terms of repayment other than it will be paid out of revenues from Glamsmile, as working capital allows. During the
year ended March 31, 2011 a total of $101,245 was paid to the founding shareholder, leaving a balance due of $95,354 on June 27,
2011. As at March 31, 2012 the full amount was paid.
|
All options reside under the Company’s
option plan and are five year options.
Also pursuant to the agreement, the Company
granted irrevocable right to Glamsmile Asia to use the Glamsmile trademark in Greater China.
The Company acquired a 50.98% interest
in GlamSmile Asia Ltd. (“GlamSmile Asia”) in order to obtain a platform in the Chinese Market to expand and introduce
our GlamSmile Asia concept into the Chinese Market. In order to sell into the Chinese Market, an approval by Chinese Authorities
is required, in the form of licenses. As GlamSmile Asia was already the owner of such licenses prior to the acquisition, this was
an important advantage. We obtained control of GlamSmile Asia through the acquisition of the 50.98% interest and the appointment
of our CEO as a Board member of GlamSmile Asia.
|
|
(a)
|
Details of the acquisition date fair value transferred, and allocation thereof are as follows:
|
|
Common shares (250,000 at $0.39/share) (rate at December 31, 2009)
|
|
$
|
97,500
|
|
|
325,000 Euro (at 1.436077)
|
|
|
466,725
|
|
|
200,000 stock options (*)
|
|
|
62,108
|
|
|
Total consideration
|
|
$
|
626,333
|
|
|
|
|
|
|
|
|
Cash
|
|
$
|
167,288
|
|
|
Accounts receivable
|
|
$
|
27,836
|
|
|
Inventory
|
|
$
|
23,347
|
|
|
Equipment, net
|
|
$
|
76,647
|
|
|
|
|
|
|
|
|
Accounts payable
|
|
$
|
145,996
|
|
|
Accruals
|
|
$
|
25,446
|
|
|
Other payables
|
|
$
|
196,978
|
|
|
|
|
|
|
|
|
Net liability (sub-consolidated financial statements of GlamSmile Asia Ltd including the Mainland China Subsidiaries)
|
|
|
73,302
|
|
|
|
|
|
|
|
|
Goodwill
|
|
$
|
699,635
|
|
* The value of the stock options was determined by using the
Black-Scholes valuation model with a stock price of $0.39/share, an option price of $0.39/share, an expected life of 5 years, a
volatility of 112%, a risk free rate of 1.30%, and a dividend rate of zero.
(b) The
acquisition date fair value of the total consideration transferred was allocated amongst assets, liabilities and noncontrolling
interest based upon their fair value at the time of acquisition. We evaluated all current assets for collectability
and because they were current and there was no history of collection problems, determined that their carrying value was equal to
their fair value. We also evaluated all current liabilities and because there was no evidence of over or under accruals,
we also determined that the carrying value of the Company’s current liabilities was equal to fair value. Lastly,
we evaluated the fair value of Glamsmile’s equipment and because it was all recently acquired, i.e. within the past year,
because there was no evidence of impairment, or non-functionality, and because the recorded value of the equipment was approximately
equal to its replacement cost, we also determined that the carrying value of the equipment was equal to its fair value.
Deconsolidation
On January 28, 2012, the Company entered
into a Preference A Shares and Preference A-1 Shares Purchase Agreement (“Share Purchase Agreement”) with Glamsmile
Dental Technology Ltd., a Cayman Islands company and a subsidiary of the Company (“Glamsmile Dental”), Glamsmile (Asia)
Limited, a company organized and existing under the laws of Hong Kong and a substantially owned subsidiary of Glamsmile Dental,
Beijing Glamsmile Technology Development Ltd., Beijing Glamsmile Trading Co., Ltd., Beijing Glamsmile Dental Clinic Co., Ltd.,
and Shanghai Glamsmile Dental Clinic Co., Ltd., Gallant Network Limited, a shareholder of Glamsmile Dental (“Gallant”),
and IDG-Accel China Growth Fund III L.P. (“IDG Growth”), IDG-Accel China III Investors L.P.(“IDG Investors”)
and Crown Link Group Limited (“Crown”)(“IDG Growth, IDG Investors and Crown collectively referred to as the “Investors”),
pursuant to which the Investors agreed to (i) purchase from the Company an aggregate of 2,857,143 shares of Preference A-1 Shares
of Glamsmile Dental, which represents all of the issued and outstanding Preference A-1 Shares of Glamsmile Dental, for an aggregate
purchase price of $2,000,000, and (ii) purchase from Glamsmile Dental an aggregate of 5,000,000 shares of Preference A Shares for
an aggregate purchase price of $5,000,000.
Under the terms of the Share Purchase Agreement,
the Company agreed (a) to indemnify the Investors and their respective affiliates for losses arising out of a breach, or inaccuracy
or misrepresentation in any representation or warranty made by the Company or a breach or violation of a covenant or agreement
made by the Company for up to $1,500,000, and (b) to transfer 500,000 shares of Glamsmile Dental owned by the Company to the Investors
in the event of breach of certain covenants by the Company. In connection with the Share Purchase Agreement, the Company also agreed
to enter into an Investor’s Rights Agreement, Right of First Refusal and Co-Sale Agreement, and Voting Agreement with the
parties.
In addition, in connection with the contemplated
transactions in the Share Purchase Agreement on January 20, 2012, the Company entered into a Distribution, License and Manufacturing
Agreement with Glamsmile Dental pursuant to which the Company appointed Glamsmile Dental as the exclusive distributor and licensee
of Glamsmile Veneer Products bearing the “Glamsmile” name and mark in the B2C Market in the People’s Republic
of China (including Hong Kong and Macau) and Republic of China (Taiwan) and granted related manufacturing rights and licenses in
exchange for the original issuance of 2,857,143 shares of Preference A-1 Shares of Glamsmile Dental and $250,000 (the receipt of
which was acknowledged as an offset to payment of certain invoices of Glamsmile (Asia) Limited).
On February 10, 2012, the sale of the Preference
A-1 Shares and the Preference A Shares was completed. As a result of the closing, the equity ownership of Glamsmile Dental, on
an as converted basis, is as follows: 31.4% by the Investors, 39.2 % by Gallant, and 29.4% by the Company. Mr. De Vreese, our chairman,
will remain as a director of Glamsmile Dental along with Mr. David Lok, who is the Chief Executive Officer and director of Glamsmile
Dental and principal of Gallant. The Investors have a right to appoint one director of Glamsmile Dental, and accordingly the Board
of Directors of Glamsmile Dental will consist of Mr. De Vreese, Mr. Lok and a director appointed by the Investors.
In conjunction with the transaction and
resulting deconsolidation of Glamsmile Dental, the Company recorded a gain of $1,470,776, calculated as follows:
|
Consideration received
|
|
$
|
2,000,000
|
|
|
Fair value of 29.4% interest
|
|
|
2,055,884
|
|
|
Carrying value of non-controlling interest
|
|
|
1,117,938
|
|
|
Less: carrying value of former subsidiary’s net assets
|
|
|
(2,002,329
|
)
|
|
Goodwill
|
|
|
(699,635
|
)
|
|
Investment China & Hong Kong
|
|
|
(1,082
|
)
|
|
Rescission agreement Excelsior (Note 14)
|
|
|
(1,000,000
|
)
|
|
|
|
$
|
1,470,776
|
|
For the period February 11, 2012 through
March 31, 2012, the Company recorded equity income of $36,634 in “Other income” for its portion of the net income recorded
by GlamSmile Dental Technology Ltd.
4. SHORT TERM LOAN
On March 25, 2011, the Company entered
into a Loan Agreement (the “Loan Agreement”) with an unrelated private individual pursuant to which the Company borrowed
$400,000 in exchange for a secured promissory note (“Promissory Note”) for a total repayment
amount of $500,000 (“Repayment Amount”) which was repaid in January 2012.
5. DISTRIBUTION AGREEMENTS
Den-Mat Distribution Agreement
On August 24, 2008, the Company entered
into a distribution agreement (the “Distribution Agreement”) with Den-Mat Holdings, LLC, a Delaware limited liability
company (“Den-Mat”). Pursuant to the Distribution Agreement, the Company appointed Den-Mat to be the
sole and exclusive distributor to market, license and sell certain products relating to the Company’s GlamSmile tray technology,
including, but not limited to, its GlamSmile veneer products and other related veneer products (the “Products”), throughout
the world, with the exception of Australia, Austria, Belgium, Brazil, France (including all French overseas territories “Dom-Tom”),
Germany, Italy, New Zealand, Oman, Poland, Qatar, Saudi Arabia, Singapore, Switzerland, Thailand, and United Arab Emirates (collectively
the “Excluded Markets”) and the China Market (the “Territory”).
As consideration for such distribution,
licensing and manufacturing rights, Den-Mat will pay the Company:
|
|
(i)
|
an initial payment of $2,425,000 (received);
|
|
|
(ii)
|
a payment of $250,000 for each of the first three contract periods in the initial Guaranty Period,
subject to certain terms and conditions;
|
|
|
(iii)
|
certain periodic payments as additional paid-up royalties in the aggregate amount of $500,000
(received);
|
|
|
(iv)
|
a payment of $1,000,000 promptly after Den-Mat manufactures a limited quantity of products at
a facility owned or leased by Den-Mat;
|
|
|
(v)
|
A payment of $1,000,000 promptly upon completion of certain training of Den-Mat’s personnel;
|
|
|
(vi)
|
a payment of $1,000,000 upon the first to occur of (a) February 1, 2009 or (b) the date
thirty (30) days after den-Mat sells GlamSmile Products incorporating twenty thousand (20,000) Units/Teeth to customers regardless
of whether Den-Mat has manufactured such Units/Teeth in a Den-Mat facility or has purchased such Units/Teeth from the Company;
|
|
|
(vii)
|
certain milestone payments; and
|
|
|
(viii)
|
certain royalty payments.
|
The Company has received $698,597 in royalty
payments and $4,000,000 in milestone payments, subsequent to the initial payment of $2,425,000.
Further, as consideration for Den-Mat’s
obligations under the Distribution Agreement, the Company agreed to, among other things:
|
|
(i)
|
issue to Den-Mat or an entity to be designated by Den-Mat, warrants to purchase up to 3,378,379 shares of the Corporation’s common stock, par value $0.001 per share (the “Warrant Shares”) at an exercise price of $1.48 per share, exercisable for a period of five years (the “Den-Mat Warrant”) (The warrants were issued in the period ended September 30, 2008 and were valued at $4,323,207 based upon the Black-Scholes option pricing model utilizing a market price on the date of grant of $1.48 per share, an annualized volatility of 131%, a risk free interest rate of 3.07% and an expected life of five years. The expense was originally classified as a non-operating expense however, we have subsequently reclassified the expense to operations since our agreement with Den-Mat is in the normal course of our operations. The reclassification increased our operations expenses by $4,323,207, while reducing our other expenses by the same amount, resulting in no net impact upon our consolidated net loss for the year ended March 31, 2009.
|
|
|
(ii)
|
execute and deliver to Den-Mat a registration rights agreement covering the registration of the Warrant Shares (the “Registration Rights Agreement”) which as of March 31, 2011 has not yet been filed (*); and
|
|
|
(iii)
|
cause its Chairman of the Board, Guy De Vreese, to execute and deliver to Den-Mat a non-competition agreement.
|
(*) In 2009, the Company and Den-Mat agreed
that the shares underlying the warrants would be registered pursuant to a registration statement upon the earlier occurrence of
(a) Den-Mat’s demand for the shares to be registered, or (b) an initiation and filing of a registration statement by other
investors or the Company. As of March 31, 2011, neither event has occurred to trigger a registration statement. As
a result, the Company is not under default under the distribution agreement and there is no impact on the distribution agreement
or the accounting for the distribution agreement.
On June 3, 2009, the Distribution Agreement
was amended and restated (the “Amended Agreement”). The Amended Agreement modifies and clarifies certain terms and
provisions which among other things includes:
(1) the expansion of the list of Excluded
Markets to include Spain, Japan, Portugal, South Korea and South Africa for a period of time;
(2) clarification that Den-Mat’s
distribution and license rights are non-exclusive to market, sell and distribute the Products directly to consumers through retail
locations (“B2C Market”) in the Territory and an undertaking to form a separate subsidiary to and to issue warrants
to Den-Mat in the subsidiary in the event that the Company decides to commercially exploit the B2C Market in North America after
January 1, 2010;
(3) subject to certain exceptions, a commitment
from the Company to use Den-Mat as its supplier to purchase all of its, and its licensee’s, GlamSmile products in the B2C
Market from Den-Mat, with reciprocal commitment from Den-Mat to sell such products;
(4) modification of certain defined terms
such as “Guaranty Period,” “Exclusivity Period” and addition of the term “Contract Period”;
and
(5) the “Guaranty Period” (as
defined therein) is no longer a three year period but has been changed to the first three “Contract Periods”. The
first Contract Period commences on the first day of the Guaranty Period (which the Parties agreed has commenced as of April 1,
2009), and continues for fifteen (15) months or such longer period that would be necessary in order for Den-Mat to purchase a certain
minimum number of Units/Teeth as agreed upon in the Amended Agreement (“Minimum Purchase Requirement”) in the event
that the Company’s manufacturing capacity falls below a certain threshold. The second and each subsequent GlamSmile
Contract Period begins on the next day following the end of the preceding “Contract Period” and continues for twelve
(12) months or such longer period that would be necessary in order for Den-Mat to meet its Minimum Purchase Requirement in the
event that the Company’s manufacturing capacity falls below a certain threshold.
In August 2009, the Distribution Agreement
was further amended (the “August Amendment”). The August Amendment expands the Company’s products covered under
the Distribution Agreement to include the Company’s new Prego System Technology (“Prego System”), also commonly
known as “Glamstrip”. Under the Amendment, the $250,000 payment which was originally due upon the expiration of the
first Contract Period (as defined in the Distribution Agreement) is now due on the earlier occurrence of (i) sixty days from August
11, 2009 or (ii) the performance of the Company’s live patient clinical demonstration of the Prego System to be performed
at Den-Mat’s reasonable satisfaction (received).
The August Amendment also provides for
(a) the royalty rate for products manufactured and sold by Den-Mat using the Prego System after the Guaranty Period (as defined
in the Distribution Agreement), (b) Den-Mat’s right to elect to manufacture or purchase from a third party manufacturer any
or all portion of the minimum purchase requirements under the Distribution Agreement provided however, that if Den-Mat fails to
purchase the minimum number of Units/Teeth as required during any month, Den-Mat may cure such default by paying the Company a
certain royalty on the difference between the minimum purchase requirement and the amount actual purchased by Den-Mat during such
month, with such royalties accruing and being due and payable upon the earlier occurrence of either (1) one hundred twenty days
from August 11, 2009 or (2) the successful performance of the Company’s live patient demonstration of the First Fit
Technology licensed to Den-Mat pursuant to the First Fit-Crown Distribution and License Agreement, to be performed at
Den-Mat’s reasonable satisfaction; and all shortfall payments thereafter being due and payable within 15 days after the end
of the month in which shortfall occurred, and (c) Den-Mat’s option to purchase a certain number of Prego Systems in lieu
of Trays during each of the first three Contract Periods pursuant to the terms, including price and conditions, set forth in the
Amendment so long as such option is exercised during the period commencing on August 11, 2009 and ending on the later of either
91 days or 31 days after the Company demonstrates to Den-Mat that it has the capacity to produce a certain number of Prego System
per Contract Period. Furthermore under the Amendment, if Den-Mat fails to purchase the required minimum Trays during any Contract
Period, such failure may be cured by payment equal to the difference between the aggregate purchase price that would have been
paid had Den-Mat purchased the required minimum and the aggregate purchase price actually paid for such Contract Year within 30
days after the end of such Contract Period. With the exception of the provisions amended by the Amendment, the Distribution Agreement
remains in full force and effect.
First Fit Distribution Agreement
On June 3, 2009, the Company entered into
the First Fit-Crown Distribution and License Agreement (the “First Fit Distribution Agreement”) with Den-Mat. Under
the terms of the First Fit Distribution Agreement, the Company appointed Den-Mat to be its sole and exclusive distributor to market,
license and sell certain products relating to the Company’s proprietary First Fit technology (the “First Fit Products”),
in the United States, Canada and Mexico (the “First Fit Territory”). In connection therewith, the Company
also granted Den-Mat certain non-exclusive rights to manufacture and produce the First Fit Products in the First-Fit Territory;
and a sole and exclusive transferable and sub-licensable right and license to use the Company’s intellectual property rights
relating to the First Fit Products to perform its obligations as a distributor (provided the Company retains the right to use and
license related intellectual property in connection with the manufacture of the First Fit Products for sale outside of the First
Fit Territory).
Consummation of the First Fit Distribution
Agreement is subject to: completion of Den-Mat’s due diligence; execution and delivery of Non-Competition Agreements; and
the delivery of the Development Payment and first installment of the License Payment (the “Development Payment” and
License Payment” are defined below).
Under the First Fit Distribution Agreement,
the Company granted such distribution rights, licensing rights and manufacturing rights, in consideration for the following: (i)
a non-refundable development fee of Four Hundred Thousand Dollars ($400,000) (the “Development Payment”) payable in
two installments of $50,000 each, one within seven days after the effective date of the First Fit Distribution Agreement, and another
$350,000 payment within twenty one days after the Effective Date ($400,000 received as at June 30, 2009); (ii) a non-refundable
license fee of $600,000 payable in three equal installments of $200,000 each, with the first installment payable on the Closing
Date, and with the second and third installments payable on the 30th and 60th day, respectively, after the Closing Date (received);
(iii) certain royalty payments based on the sales of the First Fit Products by Den-Mat or its sub-licensees; and (iv) certain minimum
royalty payments to maintain exclusivity.
Den-Mat’s rights
as an exclusive distributor and licensee will continue at least through the first Contract Period (defined below) and until the
termination of the First Fit Distribution Agreement. Den-Mat’s exclusivity ends at the end of any Contract Period
in which Den-Mat fails to make certain minimum royalty payments. In the event that such exclusivity is terminated, Den-Mat
has the option to either terminate the First Fit Distribution Agreement upon ninety (90) days written notice, or become a non-exclusive
distributor and licensee, in which event Den-Mat’s obligation to pay certain agreed upon royalties would continue. “Contract
Period” means the following periods: (A) the first eighteen months beginning on the first day of the month following
the month in which the Closing occurs, provided that if Den-Mat is not fully operational within sixty days after the Closing Date,
the first Contract Period will be extended by one day for each day after the sixtieth day until Den-Mat becomes fully operational;
(B) the subsequent twelve months; and (C) each subsequent twelve month period thereafter, in each case during which the First Fit
Distribution Agreement is in effect.
On March 29, 2010, a certain Amendment
No. 1 was made to the First Fit Distribution Agreement dated June 3, 2009. The terms of Amendment No. 1 are as follows:
The total purchase price for the First
Fit IP consists of installment payments and royalty payments. The cash component of the purchase price of the First Fit IP is $2,850,000
to be paid in the form of cash in the following installments: (a) $50,000 upon delivery by Remedent to Den-Mat of a working prototype
of the First Fit crown (received); (b) $525,000 on or before March 15, 2010 (received); (c) $700,000 on June 30, 2010(received);
and (d) $500,000 on December 31, 2010(received), June 30, 2011 (during the three months ended June 30, 2011 the Company agreed
to postpone receipt of the $500,000 due from Den-Mat on June 30, 2011 to be received as follows: $250,000 at the end of September,
2011 (received) and $250,000 at the end of October, 2011(received)) and December 31, 2011 (see Note 4).
Effective March 27, 2012 the Company and Den-Mat entered into
a termination and license agreement, as follows:
|
|
(a)
|
the parties have agreed to terminate the distribution, manufacturing and license agreement and any ancillary agreements (the
“Agreements”);
|
|
|
(b)
|
Den-Mat will pay the Company $200,000 cash in complete satisfaction of any and all payments due ($110,000 received and $90,000
subsequently received); and
|
|
|
(c)
|
the Company grants to Den-Mat a non-exclusive, irrevocable, perpetual, royalty free license to use within the territory of
intellectual property that was the subject of the Agreements and license with Den-Mat.
|
6. CONCENTRATION OF RISK
Financial Instruments — Financial
instruments, which potentially subject the Company to concentrations of credit risk, consist principally of trade accounts receivable.
Concentrations of credit risk with respect
to trade receivables are normally limited due to the number of customers comprising the Company’s customer base and their
dispersion across different geographic areas. At March 31, 2012 five customers accounted for a total of 65% of the Company’s
trade accounts receivable and one of those customers accounted for 26.7% of total accounts receivable. At March 31,
2011 five customers accounted for a total of 61% of the Company’s trade accounts receivable and one of those customers
accounted for 24.6% of total accounts receivable. The Company performs ongoing credit evaluations of its customers and
normally does not require collateral to support accounts receivable.
Purchases — The Company has diversified
its sources for product components and finished goods and, as a result, the loss of a supplier would not have a material impact
on the Company’s operations. For the year ended March 31, 2012, the Company had five suppliers who accounted for 34.5% of
gross purchases. For the year ended March 31, 2011, the Company had five suppliers who accounted for 37% of gross purchases.
Revenues — For the year ended March
31, 2012 the Company had five customers that accounted for 26.44% of total revenues. For the year ended March 31, 2011 the Company
had five customers that accounted for 37.14% of total revenues.
7. ACCOUNTS RECEIVABLE AND ALLOWANCE FOR
DOUBTFUL ACCOUNTS
The Company’s accounts receivable
at year end were as follows:
|
|
|
March 31, 2012
|
|
|
March 31, 2011
|
|
|
Accounts receivable, gross
|
|
$
|
850,601
|
|
|
$
|
2,793,626
|
|
|
Less: allowance for doubtful accounts
|
|
|
(12,361
|
)
|
|
|
(28,975
|
)
|
|
Accounts receivable, net
|
|
$
|
838,240
|
|
|
$
|
2,764,651
|
|
8. INVENTORIES
Inventories at year end are stated at the
lower of cost (first-in, first-out) or net realizable value and consisted of the following:
|
|
|
March 31, 2012
|
|
|
March 31, 2011
|
|
|
Raw materials
|
|
$
|
58,189
|
|
|
$
|
72,305
|
|
|
Components
|
|
|
283,572
|
|
|
|
685,553
|
|
|
Finished goods
|
|
|
892,244
|
|
|
|
1,536,595
|
|
|
|
|
|
1,234,005
|
|
|
|
2,294,453
|
|
|
Less: reserve for obsolescence
|
|
|
(156,239
|
)
|
|
|
(130,407
|
)
|
|
Net inventory
|
|
$
|
1,077,766
|
|
|
$
|
2,164,046
|
|
9. PREPAID EXPENSES
Prepaid expenses are summarized as follows:
|
|
|
March 31, 2012
|
|
|
March 31, 2011
|
|
|
Prepaid materials and components
|
|
$
|
882,998
|
|
|
$
|
469,784
|
|
|
|
|
|
|
|
|
|
|
|
|
Prepaid Belgium income taxes
|
|
|
—
|
|
|
|
8,249
|
|
|
VAT payments in excess of VAT receipts
|
|
|
60,252
|
|
|
|
153,012
|
|
|
Royalties
|
|
|
18,109
|
|
|
|
—
|
|
|
Prepaid trade show expenses
|
|
|
—
|
|
|
|
25,973
|
|
|
Prepaid rent
|
|
|
145,138
|
|
|
|
11,907
|
|
|
Other
|
|
|
42,767
|
|
|
|
94,028
|
|
|
|
|
$
|
1,149,264
|
|
|
$
|
762,953
|
|
10. PROPERTY AND EQUIPMENT
Property and equipment are summarized as
follows:
|
|
|
March 31, 2012
|
|
|
March 31, 2011
|
|
|
Furniture and Fixtures
|
|
$
|
540,104
|
|
|
$
|
985,926
|
|
|
Machinery and Equipment
|
|
|
1,499,271
|
|
|
|
2,894,888
|
|
|
Tooling
|
|
|
—
|
|
|
|
188,450
|
|
|
|
|
|
2,039,375
|
|
|
|
4,069,264
|
|
|
Accumulated depreciation
|
|
|
(1,398,155
|
)
|
|
|
(2,667,529
|
)
|
|
Property & equipment, net
|
|
$
|
641,220
|
|
|
$
|
1,401,735
|
|
11. LONG TERM INVESTMENTS AND ADVANCES
Soca Networks Singapore (“Soca”)
Pursuant to the terms of a letter of intent
dated December 17, 2007, the Company has agreed to purchase 20% of Soca for a total purchase price of $750,000. Half of the purchase
price has been advanced $375,000 to Soca as a down payment, pending completion of the agreement terms. The balance of
$375,000 cash was paid to Soca in March of 2008. Finalization of the 20% acquisition was dependent upon completion of
a Vietnamese production facility and commencement of operations. To date, the anticipated deadlines for completion of
the construction facility, and finalization of our agreement with Soca have not been met. Management believes that the
$750,000 advance will be recoverable against production costs in the future, but the timeframe for the recovery cannot be predicted
with any certainty. As a result, the Company determined that a 100% impairment allowance as at March 31, 2011 was appropriate.
As of March 31, 2012 there has been no change in this investment.
12. LICENSED PATENTS
Universal Applicator Patent
In September 2004, the Company entered
into an agreement with Lident N.V. (“Lident”), a company controlled by Mr. De Vreese, the Company’s Chairman,
to obtain an option, exercisable through December 31, 2005, to license an international patent (excluding the US) and worldwide
manufacturing and distribution rights for a potential new product which Lident had been assigned certain rights by the inventors
of the products, who are unrelated parties, prior to Mr. De Vreese association with the Company. The patent is an Italian patent
which relates to a single use universal applicator for dental pastes, salves, creams, powders, liquids and other substances where
manual application could be relevant. The Company has filed to have the patent approved throughout Europe. The agreement required
the Company to advance to the inventors through Lident a fully refundable deposit of €100,000 subject to the Company’s
due diligence regarding the enforceability of the patent and marketability of the product, which, if viable, would be assigned
to the Company for additional consideration to the inventors of €100,000 and an ongoing royalty from sales of products related
to the patent equal to 3% of net sales and, if not viable, the deposit would be repaid in full by Lident. The consideration the
Company had agreed to pay Lident upon the exercise of the option is the same as the consideration Lident is obligated to pay the
original inventors. Consequently, Lident would not have profited from the exercise of the option. Furthermore, at a meeting of
the Company’s Board of Directors on July 13, 2005, the Board accepted Lident’s offer to facilitate an assignment of
Lident’s intellectual property rights to the technology to the Company in exchange for the reimbursement of Lident’s
actual costs incurred relating to the intellectual property. Consequently, when the Company exercises the option, all future payments,
other than the reimbursement of costs would be paid directly to the original inventors and not to Lident.
On December 12, 2005, the Company exercised
the option and the Company and the patent holder agreed to revise the assignment agreement whereby the Company agreed to pay €50,000
additional compensation in the form of prepaid royalties instead of the €100,000 previously agreed, €25,000 of which
was paid by the Company in September 2005 and the remaining €25,000 is to be paid upon the Company’s first shipment
of a product covered by the patent. As of March 31, 2011 the Company has not yet received the final Product and therefore, $35,433
(€25,000) has been written off during the year ended March 31, 2011. Further, the patent has been completely amortized over
five (5) years and accordingly, the Company has recorded $118,840 of accumulated amortization for this patent.
13. LINE OF CREDIT
The Company has a mixed-use line of credit
facility with BNP Paribas Fortis Bank, a Belgian bank (the “Facility”). The Facility is secured by a first
lien on the assets of Remedent N.V. On January 3, 2008, an amendment was made decreasing the mixed-use line of credit
to €2,050,000, to be used by Remedent NV and/or Sylphar NV. Each line of credit carries its own interest rates and fees as
provided in the Facility and varies from the current prevailing bank rate.
The latest amendment to the Facility, dated
June 7, 2010, amended and split the line of credit to €1,250,000, to be used by Remedent NV and € 1,000,000 to be used
Sylphar NV. Each line of credit carries its own interest rates and fees as provided in the Facility and vary from the current prevailing
bank rate of approximately 3.27%, for draws on the credit line, to 9.9% for advances on accounts receivable concerning Remedent
N.V. and similar for Sylphar N.V. Remedent N.V is currently only utilizing two lines of credit, advances based on account receivables
and the straight loan. As of March 31, 2012 and March 31, 2011, Remedent N.V. had in aggregate, $1,079,263 and $1,452,024 (Combined
with Sylphar $ 2,160,674 at end March 31, 2011) in advances outstanding, respectively, under the mixed-use line of credit facilities.
14. LONG TERM DEBT
On August 18, 2009, the Company entered
into a capital lease agreement over a three year period for additional manufacturing equipment totaling €170,756 (US $227,891).
On January 15, 2010, the Company entered into a capital lease agreement over a 5 year period for veneer manufacturing equipment
totaling € 251,903 (US $336,190).
The leases require monthly payments of
principal and interest at between 7.43% and 9.72% and provide for buyouts at the conclusion of the lease terms of between 1% and
4% of the original value of the contract.
In most cases, management expects that
in the normal course of business, leases will be renewed or replaced by other leases.
The net book value as of March 31, 2012
and March 31, 2011 of the equipment subject to the foregoing leases are $273,557 and $458,236 respectively.
The following is a schedule by years of
future minimum lease payments under capital leases together with the present value of the net minimum lease payments as of March
31, 2012:
|
Year ending March 31:
|
|
|
|
|
2013
|
|
|
121,611
|
|
|
2014
|
|
|
82,805
|
|
|
2015
|
|
|
83,509
|
|
|
2016
|
|
|
—
|
|
|
Later years
|
|
|
—
|
|
|
Total minimum lease payments
|
|
|
287,925
|
|
|
Less: Amount representing estimated executory costs (such as taxes, maintenance, and insurance), including profit thereon, included in total minimum lease payments
|
|
|
—
|
|
|
Net minimum lease payments
|
|
|
287,925
|
|
|
Less: Amount representing interest (*)
|
|
|
14,368
|
|
|
Present value of minimum lease payments (**)
|
|
$
|
273,557
|
|
* Amount necessary to reduce net minimum lease payments
to present value calculated at the Company’s incremental borrowing rate at the inception of the leases.
** Reflected in the balance sheet as current and non-current
obligations under capital leases of $113,501 and $160,056 respectively.
Secured Debt Agreements
On June 3, 2011, the Company obtained a
loan in the principal amount of $1,000,000 (the “Loan”) from an unrelated private company, Excelsior Medical (HK) (“EM”).
In connection with the Loan, the Company issued a promissory note, with a simple interest rate of 5% per annum, secured by certain
assets of the Company (the “Note”). The maturity date of the Loan is June 3, 2014. Interest of $50,000 per annum is
payable in cash on an annual basis.
In September 2010, we entered into a license
agreement with EM (the “EM license agreement”). Under the EM license agreement, we granted EM an exclusive license
to certain Asian territories in exchange for $500,000 which was received during the year ended March 31, 2011.
The Company received a further $500,000
from EM as an advance payment for veneers. The $500,000 advance, less taxes withheld, was recorded as deferred revenue
of $475,250 as of March 31, 2011.
Effective as of January 11, 2012, the Company
entered into a Rescission Agreement with EM and Asia Best Healthcare Co., Ltd. Under the Rescission Agreement, the Company agreed
to repay a total of $1,000,000 received under the Distribution Agreement, plus a simple interest rate of 5%, beginning on June
30, 2012, according to the following payment schedule: (i) $250,000 to be paid no later than June 30, 2012, (ii) $250,000 plus
interest on June 30, 2012, (iii) $250,000 plus interest on December 31, 2012, and (iv) $250,000 plus interest on June 30,
2013. The Company also agreed to secure such obligations owed to EM with certain collateral of the Company.
15. DUE TO RELATED PARTIES AND RELATED PARTY
TRANSACTIONS
Transactions with related parties not disclosed
elsewhere in these financial statements consisted of the following:
Compensation:
During the years ended March 31, 2012 and
2011 respectively, the Company incurred $664,586 and $749,310 as compensation for all directors and officers.
Sales Transactions:
All related party transactions involving
provision of services or tangible assets were recorded at the exchange amount, which is the value established and agreed to by
the related parties reflecting arm’s length consideration payable for similar services or transfers.
16. ACCRUED LIABILITIES
Accrued liabilities are summarized as follows:
|
|
|
March 31, 2012
|
|
|
March 31, 2011
|
|
|
Accrued employee benefit taxes and payroll
|
|
$
|
358,148
|
|
|
$
|
545,815
|
|
|
Accrued travel
|
|
|
10,010
|
|
|
|
26,468
|
|
|
Advances and deposits
|
|
|
—
|
|
|
|
374,511
|
|
|
Commissions
|
|
|
687
|
|
|
|
26,553
|
|
|
Accrued audit and tax preparation fees
|
|
|
11,318
|
|
|
|
21,586
|
|
|
Reserve for warranty costs
|
|
|
20,019
|
|
|
|
21,260
|
|
|
Reserve for income taxes
|
|
|
2,936
|
|
|
|
113,588
|
|
|
Accrued interest
|
|
|
1,322
|
|
|
|
2,704
|
|
|
Accrued consulting fees
|
|
|
16,129
|
|
|
|
19,935
|
|
|
Other accrued expenses
|
|
|
115,762
|
|
|
|
103,728
|
|
|
|
|
$
|
536,331
|
|
|
$
|
1,256,148
|
|
17. INCOME TAXES
The domestic and foreign (“Belgium”,
“Singapore”, Hong Kong and China) components of income (loss) before income taxes and minority interest were comprised
of the following:
|
|
|
March 31, 2012
|
|
|
March 31, 2011
|
|
|
Domestic
|
|
$
|
(776,668
|
)
|
|
$
|
(1,896,529
|
)
|
|
Foreign
|
|
|
2,176,183
|
|
|
|
1,276,797
|
|
|
|
|
$
|
1,399,515
|
|
|
$
|
(619,732
|
)
|
The Company’s domestic and foreign
components of deferred income taxes are as follows:
|
|
|
March 31, 2012
|
|
|
March 31, 2011
|
|
|
Domestic — Net operating loss carryforward
|
|
$
|
7,805,133
|
|
|
$
|
7,533,300
|
|
|
Foreign — Net operating loss carryforward
|
|
|
244,774
|
|
|
|
691,369
|
|
|
Total
|
|
|
8,049,907
|
|
|
|
8,224,669
|
|
|
Valuation allowance
|
|
|
(8,049,907
|
)
|
|
|
(8,224,669
|
)
|
|
Net deferred tax assets
|
|
$
|
—
|
|
|
$
|
—
|
|
Because of the uncertainty surrounding
the timing of realizing the benefits of favorable tax attributes in future income tax returns, the Company has placed a valuation
allowance against its deferred income tax assets.
The principal reasons for the difference
between the income tax (benefit) and the amounts computed by applying the statutory income tax rates to the income (loss) for the
year ended March 31, 2012 and March 31, 2011 are as follows:
|
|
|
March 31, 2012
|
|
|
March 31, 2011
|
|
|
Domestic
|
|
|
|
|
|
|
|
|
|
Pre tax income (loss)
|
|
$
|
(776,668
|
)
|
|
$
|
(1,896,529
|
)
|
|
Statutory tax rate
|
|
|
35
|
%
|
|
|
35
|
%
|
|
Tax benefit based upon statutory rate
|
|
|
(271,833
|
)
|
|
|
(663,785
|
)
|
|
Valuation allowance
|
|
|
271,833
|
|
|
|
663,785
|
|
|
Net domestic income tax (benefit)
|
|
|
—
|
|
|
|
—
|
|
|
Foreign
|
|
|
|
|
|
|
|
|
|
Pre tax income (loss)
|
|
|
2,176,183
|
|
|
|
1,276,797
|
|
|
Statutory tax rate
|
|
|
35
|
%
|
|
|
35
|
%
|
|
Tax expense (benefit) based upon statutory rate
|
|
|
761,664
|
|
|
|
446,879
|
|
|
Permanent differences
|
|
|
446,595
|
|
|
|
226,582
|
|
|
|
|
|
—
|
|
|
|
—
|
|
|
Net foreign income tax expense (benefit)
|
|
|
315,069
|
|
|
|
220,297
|
|
|
Total Income tax (benefit )
|
|
$
|
315,069
|
|
|
$
|
220,297
|
|
18. EQUITY COMPENSATION PLANS
As of March 31, 2011, the Company had three
equity compensation plans approved by its stockholders (1) the 2001 Incentive and Non-statutory Stock Option Plan (the “2001
Plan”), (2) the 2004 Incentive and Non-statutory Stock Option Plan (the “2004 Plan”); and (3) the 2007 Equity
Incentive Plan (the “2007 Plan”). The Company’s stockholders approved the 2001 Plan reserving 250,000 shares
of common stock of the Company pursuant to an Information Statement on Schedule 14C filed with the Commission on August 15, 2001.
In addition, the Company’s stockholders approved the 2004 Plan reserving 800,000 shares of common stock of the Company pursuant
to an Information Statement on Schedule 14C filed with the Commission on May 9, 2005. Finally, the Company’s stockholders
approved the 2007 Plan reserving 1,000,000 shares of common stock of the Company pursuant to a Definitive Proxy Statement on Schedule
14A filed with the Commission on October 2, 2007.
In addition to the equity compensation
plans approved by the Company’s stockholders, the Company has issued options and warrants to individuals pursuant to individual
compensation plans not approved by our stockholders. These options and warrants have been issued in exchange for services
or goods received by the Company.
The following table provides aggregate
information as of March 31, 2012 and March 31, 2011 with respect to all compensation plans (including individual compensation arrangements)
under which equity securities are authorized for issuance.
|
|
|
2001
Plan
|
|
|
2004
Plan
|
|
|
2007
Plan
|
|
|
Other
|
|
|
|
|
Outstanding
Options
|
|
|
Weighted
Average
Exercise
Price
|
|
|
Outstanding
Options
|
|
|
Weighted
Average
Exercise
Price
|
|
|
Outstanding
Options
|
|
|
Weighted
Average
Exercise
Price
|
|
|
Outstanding
Options
|
|
|
Weighted
Average
Exercise
Price
|
|
|
Options outstanding, March 31, 2010
|
|
|
250,000
|
|
|
|
1.20
|
|
|
$
|
668,166
|
|
|
0.89
|
|
|
$
|
1,000,000
|
|
|
1.15
|
|
|
$
|
350,000
|
|
|
$
|
0.97
|
|
|
Cancelled or expired
|
|
|
—
|
|
|
|
—
|
|
|
(110,666)
|
|
|
4.00
|
|
|
—
|
|
|
—
|
|
|
—
|
|
|
—
|
|
|
Options outstanding,March 31, 2011
|
|
|
250,000
|
|
|
|
1.20
|
|
|
557,500
|
|
|
0.89
|
|
|
1,000,000
|
|
|
1.15
|
|
|
350,000
|
|
|
0.97
|
|
|
Cancelled or expired
|
|
|
(237,500
|
)
|
|
|
—
|
|
|
|
(25,000
|
)
|
|
|
4.00
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Options outstanding, March 31, 2012
|
|
|
12,500
|
|
|
|
1.00
|
|
|
|
532,500
|
|
|
|
0.96
|
|
|
|
1,000,000
|
|
|
|
1.15
|
|
|
|
350,000
|
|
|
|
0.97
|
|
|
Options exercisable March 31, 2012
|
|
|
12,500
|
|
|
|
1.00
|
|
|
|
532,500
|
|
|
|
0.96
|
|
|
|
1,000,000
|
|
|
|
1.15
|
|
|
|
350,000
|
|
|
|
0.97
|
|
|
Exercise price range
|
|
$
|
2.00
|
|
|
|
|
|
|
$
|
0.50
- $2.46
|
|
|
|
|
|
|
$
|
0.50
- $1.75
|
|
|
|
|
|
|
$
|
.39
- 1.75
|
|
|
|
|
|
|
Weighted average remaining life
|
|
|
2
years
|
|
|
|
|
|
|
|
5.28
years
|
|
|
|
|
|
|
|
6.1 years
|
|
|
|
|
|
|
|
4.6
years
|
|
|
|
|
|
A summary of the Company’s equity compensation plans approved
and not approved by shareholders is as follows:
|
Plan Category
|
|
Number of
securities to
be
issued upon
exercise of
of
outstanding
options,
warrants
and rights
|
|
|
Weighted-average
exercise price of
outstanding
options
warrants and
rights
|
|
|
Number of
securities
remaining
available for
future
issuance
under
equity
compensation
plans
(excluding
securities
reflected
in column (a))
|
|
|
Equity Compensation Plans approved by security holders
|
|
|
1,545,000
|
|
|
$
|
1.13
|
|
|
|
505,000
|
|
|
Equity Compensation Plans not approved by security holders
|
|
|
820,000
|
|
|
$
|
.97
|
|
|
|
NA
|
|
|
Total
|
|
|
2,365,000
|
|
|
$
|
1.08
|
|
|
|
505,000
|
|
For the year ended March 31, 2012, the
Company recognized $50,387 (2011 — $113,682) in compensation expense in the consolidated statement of operations.
19. COMMON STOCK WARRANTS AND OTHER OPTIONS
As of March 31, 2012 and March 31, 2011,
the Company had warrants to purchase the Company’s common stock outstanding that were not granted under shareholder approved
equity compensation plans as follows:
|
|
|
Outstanding
Warrants
|
|
|
Weighted
Average
Exercise
Price
|
|
|
Warrants and options outstanding, March 31, 2010
|
|
|
11,108,305
|
|
|
$
|
1.55
|
|
|
Cancelled or expired
|
|
|
(3,318,678
|
)
|
|
|
1.58
|
|
|
Warrants outstanding March 31, 2011 and March 31, 2012
|
|
|
7,794,627
|
|
|
|
1.50
|
|
|
Warrants exercisable March 31, 2012
|
|
|
7,794,627
|
|
|
$
|
1.50
|
|
|
Exercise price range
|
|
$
|
1.00 to $1.65
|
|
|
|
|
|
|
Weighted average remaining life
|
|
|
.75 Years
|
|
|
|
|
|
During the year ended March 31, 2010 as
consideration for certain services the Company granted our investor relations consultants warrants to purchase up to 1,210,000
shares of our common stock, at an exercise price of $1.00 per share, subject to certain vesting restrictions. A total
of 370,000 of the warrants vested on the July 15, 2009 grant date and an additional 100,000 vested during the period ended December 31,
2009. The balance of the warrants were cancelled effective November 30, 2009.
20. SEGMENT INFORMATION
The Company’s only operating segment
consists of dental products and oral hygiene products sold by Remedent Inc., Remedent N.V., GlamSmile Deutschland GmbH and GlamSmile
Rome SRL
|
|
|
Revenues
For the year ended
March 31, 2012
|
|
|
Long-lived Assets
As at
|
|
|
|
|
(a)
|
|
|
March 31, 2012
|
|
|
United States
|
|
$
|
1,120,437
|
|
|
$
|
3,055,023
|
|
|
Europe
|
|
|
5,010,610
|
|
|
|
641,220
|
|
|
China
|
|
|
3,556,245
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
9,687,292
|
|
|
$
|
3,696,243
|
|
|
|
|
Revenues
For the year ended
March 31, 2011
|
|
|
Long-lived Assets
As at
|
|
|
|
|
Revenues (a)
|
|
|
March 31, 2011
|
|
|
United States
|
|
$
|
2,324,102
|
|
|
$
|
700,222
|
|
|
Europe
|
|
|
7,868,285
|
|
|
|
1,183,795
|
|
|
China
|
|
|
2,388,597
|
|
|
|
383,871
|
|
|
Singapore
|
|
|
724
|
|
|
|
229
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
12,581,708
|
|
|
$
|
2,268,116
|
|
(a) Revenues are attributed to country based on location of
customer.
21. COMMITMENTS
Real Estate Lease:
The Company leases an office facility of
5,187 square feet in Gent, Belgium from an unrelated party pursuant to a nine year lease commencing September 1, 2008. at a
base rent of €5,712 per month for the total location ($7,624 per month at March 31, 2012).
Secondly, the Company leases an office
facility of 1,991 square feet in Rome, Italy to support the sales and marketing division of our veneer business, from an unrelated
party pursuant a six year lease commencing July 1, 2011, at a base rent of € 6,500 per month for the total location ($8,675
per month at March 31, 2012).
Real Estate Lease and All Other Leased
Equipment:
Minimum monthly lease payments for real
estate, and all other leased equipment are as follows based upon the conversion rate for the (Euro) at March 31, 2012:
|
March 31, 2013
|
|
$
|
458,010
|
|
|
March 31, 2014
|
|
|
407,013
|
|
|
March 31, 2015
|
|
|
349,960
|
|
|
March 31, 2016
|
|
|
254,469
|
|
|
March 31, 2016
|
|
|
215,217
|
|
|
After five years
|
|
|
94,644
|
|
|
Total:
|
|
$
|
1,779,313
|
|
22. FINANCIAL INSTRUMENTS
The FASB ASC topic 820 on fair value measurement
and disclosures establishes three levels of inputs that may be used to measure fair value: quoted prices in active markets for
identical assets or liabilities (referred to as Level 1), observable inputs other than Level 1 that are observable for the asset
or liability either directly or indirectly (referred to as Level 2), and unobservable inputs to the valuation methodology that
are significant to the measurement of fair value of assets or liabilities (referred to as Level 3).
The carrying values and fair values of our financial instruments
are as follows:
|
|
|
|
|
|
March 31, 2012
|
|
|
March 31, 2011
|
|
|
|
|
|
|
|
Carrying
|
|
|
Fair
|
|
|
Carrying
|
|
|
Fair
|
|
|
|
|
Level
|
|
|
value
|
|
|
Value
|
|
|
value
|
|
|
value
|
|
|
Cash
|
|
|
1
|
|
|
$
|
203,584
|
|
|
$
|
203,584
|
|
|
$
|
1,662,520
|
|
|
$
|
1,662,520
|
|
|
Accounts receivable
|
|
|
2
|
|
|
$
|
838,240
|
|
|
$
|
838,240
|
|
|
$
|
2,764,651
|
|
|
$
|
2,764,651
|
|
|
Long term investments and advances OTC Division
|
|
|
2
|
|
|
$
|
962,505
|
|
|
$
|
962,505
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
Long term investments and advances Soca
|
|
|
3
|
|
|
$
|
750,000
|
|
|
$
|
—
|
|
|
$
|
750,000
|
|
|
$
|
—
|
|
|
Long Term investment and advance - GlamSmile Dental Technology Asia
|
|
|
3
|
|
|
$
|
2,092,518
|
|
|
$
|
2,092,518
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
Line of credit
|
|
|
2
|
|
|
$
|
1,079,263
|
|
|
$
|
1,079,263
|
|
|
$
|
2,160,674
|
|
|
$
|
2,160,674
|
|
|
Short term debt
|
|
|
2
|
|
|
$
|
863,501
|
|
|
$
|
863,501
|
|
|
$
|
400,000
|
|
|
$
|
400,000
|
|
|
Deferred revenue
|
|
|
2
|
|
|
$
|
57,254
|
|
|
$
|
57,254
|
|
|
$
|
475,250
|
|
|
$
|
475,250
|
|
|
Accounts payable
|
|
|
2
|
|
|
$
|
1,009,176
|
|
|
$
|
1,009,176
|
|
|
$
|
1,744,252
|
|
|
$
|
1,744,252
|
|
|
Accrued liabilities
|
|
|
2
|
|
|
$
|
536,331
|
|
|
$
|
536,331
|
|
|
$
|
1,256,148
|
|
|
$
|
1,256,148
|
|
|
Long term debt
|
|
|
2
|
|
|
$
|
1,452,523
|
|
|
$
|
1,452,523
|
|
|
$
|
458,236
|
|
|
$
|
458,236
|
|
The following method was used to estimate the fair values of
our financial instruments:
The carrying amount of level 1 and level
2 financial instruments approximates fair value because of the short maturity of the instruments.
Financial assets are considered Level 3
when their fair values are determined using pricing models, discounted cash flow methodologies, or similar techniques, and at least
one significant model assumption or input is unobservable. Level 3 financial assets also include certain investment securities
for which there is limited market activity such that the determination of fair value requires significant judgment or estimation.
The Company reviews the fair value hierarchy
classification on a quarterly basis. Changes in the ability to observe valuation inputs may result in a reclassification of levels
for certain securities within the fair value hierarchy. The Company’s policy is to recognize transfers into and out of levels
within the fair value hierarchy at the end of the fiscal quarter in which the actual event or change in circumstances that caused
the transfer occurs. There were no significant transfers between Level 1, Level 2, or Level 3 during the fiscal years ended March
31, 2012 or March 31, 2011. When a determination is made to classify an asset or liability within Level 3, the determination is
based upon the significance of the unobservable inputs to the overall fair value measurement. The following table provides a reconciliation
of the beginning and ending balances of the item measured at fair value on a recurring basis in the table above that used significant
unobservable inputs (Level 3):
|
|
|
Year ended March 31,
2012
|
|
|
Year ended March 31,
2011
|
|
|
Long term investments and advances:
|
|
|
|
|
|
|
|
|
|
Beginning balance
|
|
$
|
—
|
|
|
$
|
750,000
|
|
|
Gains (losses) included in net loss
|
|
|
|
|
|
|
(750,000
|
)
|
|
Transfers in (out of level 3)
|
|
|
2,092,518
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
Ending balance
|
|
$
|
2,092,518
|
|
|
$
|
—
|
|
Elysee Development (PK) (USOTC:ASXSF)
Historical Stock Chart
From Jun 2024 to Jul 2024
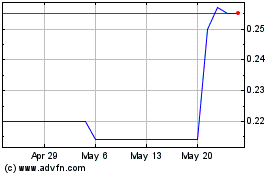
Elysee Development (PK) (USOTC:ASXSF)
Historical Stock Chart
From Jul 2023 to Jul 2024