false
0001873875
0001873875
2024-02-28
2024-02-28
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM
8-K
CURRENT
REPORT
Pursuant
to Section 13 or 15(d)
of
the Securities Exchange Act of 1934
Date
of Report February 28, 2024
Incannex
Healthcare Inc.
(Exact
name of registrant as specified in its charter)
| Delaware |
|
001-41106 |
|
93-2403210 |
(State or other Jurisdiction
of Incorporation) |
|
(Commission File Number) |
|
(IRS Employer
Identification No.) |
Suite
105, 8 Century Circuit Norwest,
NSW
2153 Australia |
|
Not applicable |
| (Address of Principal
Executive Offices) |
|
(Zip Code) |
Registrant’s
Telephone Number, including Area Code: +61 409 840 786
(Former
Name or Former Address, if Changed Since Last Report): Not Applicable
Check
the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under
any of the following provisions:
| ☐ |
Written communications pursuant to Rule 425 under the
Securities Act (17 CFR 230.425) |
| ☐ |
Soliciting material pursuant to
Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ☐ |
Pre-commencement communications
pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ☐ |
Pre-commencement communications
pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities
registered pursuant to Section 12(b) of the Act:
| Title
of each class |
|
Trading
Symbol |
|
Name
of exchange on which registered |
| Common
Stock, $0.0001 par value per share |
|
IXHL |
|
The
Nasdaq Stock Market LLC |
Indicate
by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405
of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter)
Emerging
growth company ☒
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act.
Item
8.01
On
February 28, 2024, Incannex Healthcare Inc. announced that they obtained positive topline results from its Phase 2 Psi-GAD1 clinical
trial of psilocybin in generalised anxiety disorder. Further information is included in the press release attached as Exhibit 99.1 hereto,
which is incorporated by reference into this Item 8.01.
Item
9.01 Financial Statements and Exhibits.
(d) Exhibits
SIGNATURES
Pursuant
to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by
the undersigned hereunto duly authorized.
| |
Incannex Healthcare Inc. |
| |
|
|
| Date: February 28, 2024 |
|
/s/ Joel Latham |
| |
Name: |
Joel Latham |
| |
Title: |
Chief Executive Officer and President |
2
Exhibit 99.1

Date: February 28, 2024
Public Announcement (NASDAQ: IXHL)
Incannex Announces Positive Topline Results
from Phase 2 Psi-GAD1 Clinical Trial of Psilocybin in Generalised Anxiety Disorder
Highlights:
| - | PsiGAD1 trial achieves primary endpoint; PsiGAD psilocybin-assisted therapy demonstrated a statistically significant HAM-A reduction
of 12.8 points from baseline, representing a 9.2-point improvement over psychotherapy with placebo (p <0.0001), exceeding the company’s
expectations. |
| - | 44% of patients in the psilocybin group demonstrated at least 50% reduction in anxiety score and 27% of patients showed disease
remission– a remission rate more than 5 times higher than that of therapy with placebo. |
| - | Newly developed and formulated PSX-001 psilocybin drug product has been finalised - cGMP manufacture for clinical trial supply
underway. |
| - | Incannex to submit an Investigational New Drug (IND) application with U.S. Food and Drug Administration (FDA) to proceed to a multi-site
Phase 2B trial. |
MELBOURNE, Australia and NEW YORK, USA, Feb.
28, 2024: Incannex Healthcare Inc. (Nasdaq: IXHL), (Incannex or the Company), a leading cannabinoid and psychedelic medicine biotechnology
company, is pleased to announce positive topline results from its Phase 2 Psi-GAD1 clinical trial of psilocybin in generalised anxiety
disorder (GAD). The trial met its primary endpoint, demonstrating a large clinical effect in the psilocybin treatment group over the placebo
group.
The trial protocol and treatment design were developed
in partnership with the Clinical Psychedelic Lab at Monash University, led by Dr Paul Liknaitzky.
The reduction in HAM-A score from baseline in
the psilocybin group was 12.8 points, from 29.5 at baseline to 16.8 at week 11 (6 weeks following the final dosing session), representing
a decrease of 9.2 points over the placebo group (-12.8 psilocybin vs. -3.6 placebo; p<0.0001). 44% of patients in the psilocybin group
showed a clinically meaningful improvement of at least 50% reduction in anxiety score from baseline; a ‘response rate’ more
than four times higher than that of the placebo group. 27% of patients in the psilocybin group achieved full disease remission; a rate
more than five times higher than that of psychotherapy with placebo.
Psilocybin within the context of PsiGAD psychotherapy
was observed to be well-tolerated, with only mild and moderate adverse events (AEs) reported. The reported AEs were consistent with the
known effects of the drug. No serious or severe adverse events were observed. Only one person of the 73 participants withdrew from the
trial during the 7-week treatment program.
Incannex Healthcare Inc.
Level 39, Rialto South Tower, 525 Collins
Street, Melbourne VIC 3000
Email: admin@incannex.com.au

Date: February 28, 2024
Public Announcement (NASDAQ: IXHL)
“We are thrilled with the results from our
initial PsiGAD trial”, said President and CEO Joel Latham. “This is the first time psilocybin has been investigated for treatment
of generalised anxiety disorder, and the reduction in HAM-A scores we have observed are far greater those reported from trials on established
medicines for treatment of anxiety. The improvement in anxiety scores in PsiGAD1 are of a similar magnitude to the change seen in studies
investigating psilocybin for treatment of depression disorders. Safety is a key component of any new therapy, and we are delighted that
no serious or severe adverse events were observed in PsiGAD patients, which is testament to the focus on safety within the PsiGAD treatment
protocol. The safety and efficacy results from PsiGAD1 implore us to continue the development of PsiGAD through large scale well-controlled
trials, because this treatment method has the potential to improve the quality of life for millions of people suffering from generalised
anxiety disorder.”
Incannex have designed the follow-up Phase 2B
clinical trial, PsiGAD2, with the assistance of Clerkenwell Health, a UK based contract research organisation specialising in psychiatry
and CNS treatments. This trial will be conducted at multiple sites in the United States (US) and United Kingdom (UK).
In parallel, Incannex has finalised the development
of formulation of its psilocybin drug product, PSX-001. Final preparations for the manufacture of the cGMP clinical trial supply of PSX-001
are underway. Documentation on the formulation development and cGMP manufacture will form the final pieces of the FDA IND application
that Incannex commenced in August of 2023. Clearance of the IND by the agency is required for the Company to conduct the PsiGAD2 study
at sites in the US.
Incannex is continuing to work with Clerkenwell
Health to select trial sites and prepare the relevant regulatory documents for submission to the Medicines and Healthcare products Regulatory
Agency (MHRA) in the UK.
About Generalised Anxiety Disorder
Generalised Anxiety Disorder (GAD) is characterised
by excessive anxiety and worry that occurs more days than not for at least 6 months and is not restricted to any particular environmental
circumstances. Symptoms are variable, including feelings of persistent and excessive worry, nervousness, restlessness, difficulty concentrating,
and a range of somatic manifestations. People with GAD find it difficult to control their worry, which may cause significant distress
and impairment in social, occupational, or other areas of functioning. GAD is a relatively common disorder (about 6-9% lifetime prevalence,
and about 3% 12-month prevalence in countries like Australia and the United States). As with other mood disorders, successful treatment
of GAD remains inadequate, with less than half of patients achieving remission following evidence-based treatment, alongside high relapse
rates, and substantial treatment side-effects or cost.
This announcement has been approved for release to NASDAQ by the
Incannex Board of Directors.
END
Incannex Healthcare Inc.
Level 39, Rialto South Tower, 525 Collins
Street, Melbourne VIC 3000
Email: admin@incannex.com.au

Date: February 28, 2024
Public Announcement (NASDAQ: IXHL)
About Incannex Healthcare Inc.
Incannex is a clinical stage pharmaceutical development
company that is developing unique medicinal cannabis pharmaceutical products and psychedelic medicine therapies for the treatment of obstructive
sleep apnoea (OSA), traumatic brain injury (TBI) and concussion, lung inflammation (ARDS, COPD, asthma, bronchitis), rheumatoid arthritis,
inflammatory bowel disease, anxiety disorders, addiction disorders, and pain, among other indications.
U.S. FDA approval and registration, subject to
ongoing clinical success, is being pursued for each drug and therapy under development. Each indication under investigation currently
has no, or limited, existing registered pharmacotherapy (drug) treatments available to the public and represent major global economic
opportunities to Incannex and its shareholders.
Incannex has a strong patent filing strategy in
place as it develops its products and therapies in conjunction with its medical and scientific advisory board and partners. The Company
holds 19 granted patents and 30 pending patent applications. Incannex is listed on the NASDAQ as IXHL
Website: www.incannex.com.au
Investors: investors@incannex.com.au
Forward-looking statements
This press release contains “forward-looking
statements” within the meaning of the “safe harbor” provisions of the U.S. Private Securities Litigation Reform Act of
1995. These forward-looking statements are made as of the date they were first issued and were based on current expectations and estimates,
as well as the beliefs and assumptions of management. The forward-looking statements included in this press release represent Incannex’s
views as of the date of this press release. Incannex anticipates that subsequent events and developments may cause its views to change.
Incannex undertakes no intention or obligation to update or revise any forward-looking statements, whether as of a result of new information,
future events or otherwise. These forward-looking statements should not be relied upon as representing Incannex’s views as of any date
after the date of this press release.
Contact Information:
Incannex Healthcare Inc.
Mr Joel Latham
Chief Executive Officer, President and Director
admin@incannex.com.au
Investor Relations Contact – United States
Laine Yonker
Edison Group
+1 (610) 716 2868
lyonker@edisongroup.com
Incannex Healthcare Inc.
Level 39, Rialto South Tower, 525 Collins
Street, Melbourne VIC 3000
Email: admin@incannex.com.au

Date: February 28, 2024
Public Announcement (NASDAQ: IXHL)
Appendix: Comparison of PsiGAD Treatments to Existing
Registered Treatments for Anxiety
Incannex Healthcare Inc.
Level 39, Rialto South Tower, 525 Collins
Street, Melbourne VIC 3000
Email: admin@incannex.com.au

Date: February 28, 2024
Public Announcement (NASDAQ: IXHL)
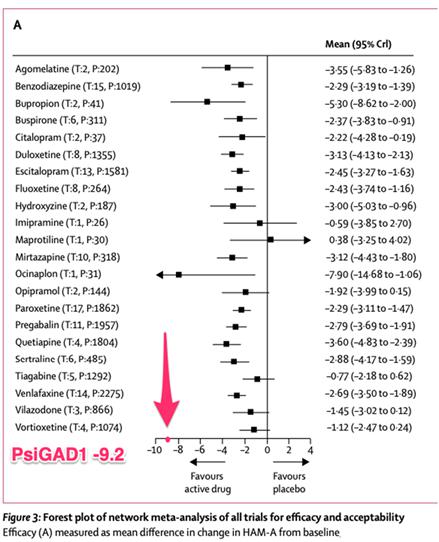
Figure: meta-analysis1
of psychotropic medications for GAD, as measured by the HAM-A; the best medication that was coded as having reliable (larger
sample size) results in this analysis, quetiapine, has a between group difference in effect of -3.60 on the HAM-A. Note, some studies
included in this meta-analysis were considered unreliable by the authors.
| 1 | Slee, A., Nazareth, I., Bondaronek, P., Liu, Y., Cheng, Z.,
& Freemantle, N. (2019). Pharmacological treatments for generalised anxiety disorder: a systematic review and network meta-analysis.
The Lancet, 393(10173), 768-777. |
Incannex Healthcare Inc.
Level 39, Rialto South Tower, 525 Collins
Street, Melbourne VIC 3000
Email: admin@incannex.com.au
Page 5 | 5
v3.24.0.1
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionISO 3166-1 alpha-2 country code.
| Name: |
dei_EntityAddressCountry |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:countryCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Incannex Healthcare (PK) (USOTC:IHLXF)
Historical Stock Chart
From Apr 2024 to May 2024

Incannex Healthcare (PK) (USOTC:IHLXF)
Historical Stock Chart
From May 2023 to May 2024
