MedMira Announces Update on Development of Point-of-Care Antibody Test
April 15 2020 - 7:00PM

MedMira Inc. (MedMira) (TSXV: MIR), announced today that it has
completed the development of the serological Point-of-Care (POC)
testing solution with immediate result. Based on MedMira’s unique
and patented Rapid Vertical Flow® (RVF) technology, the newly
developed REVEALCOVID-19TM delivers immediate results without the
need for any additional equipment such as a reader or a timer. The
product is subject to obtaining regulatory approvals to sell in the
selected markets. MedMira’s aim is to provide a reliable screening
tool to relieve overburdened healthcare providers allowing for
faster turn-around times with a rapid and reliable testing
solution.
Based on it’s proven and highly flexible RVF
technology, MedMira’s in-house R&D team has developed and
validated its REVEALCOVID-19TM, a serological total antibody test.
The product is ready to submit to the relevant regulators,
such as the FDA, through its Notification/Emergency Use
Authorization process as announced on March 16, 2020 by the
FDA. This test has been listed on the COVID-19 Diagnostic Device
Applications by Health Canada.
The test can be completed under 3 minutes (from
the blood drawn until the testing is completed) and provides an
instant result. It can be used with whole blood, serum or plasma
and is also ideal for batch testing in laboratory settings, meeting
the needs of all possible users across a broad range of testing
environments.
The REVEALCOVID-19 TM is designed to detect all
antibodies found in individuals who have been exposed to SARS-CoV-2
virus, the virus that causes COVID-19 disease. The test, in
combination with molecular diagnostic testing, will aid health care
professionals to make the most accurate assessment of the status of
patients. Rapid diagnostic tools such as MedMira’s test are
urgently needed to assess and help to control the spread of the
pandemic. This in-house developed test has been validated by
MedMira’s scientific research team with over 20 years experience in
In Vitro diagnostics using blood specimens obtained from RT-PCR
confirmed patients.
“MedMira’s patented technology has been proven
to provide the fastest and highest-quality testing solutions for
HIV, Syphilis, Hepatitis B and C and has obtained regulatory
approvals for selected products in many parts of the world. The
latest member of our REVEAL family, REVEALCOVID-19 TM, is expected
to bring the much needed protection to both healthcare providers
and their patients,” said Hermes Chan, CEO of MedMira Inc. “This
provides a vital, realistic, timely and effective testing solution
that showcases the capability of our RVF technology. It is
paramount to identify those who are infected with, or who have been
exposed to the virus and also to monitor those who are at high
risk. Our existing business is the development and sales of
antibody tests for HIV and Hepatitis, and we have internal lab and
manufacturing facilities that are FDA certified and staffs who have
been engaged in similar R&D for over 20 years.”
MedMira’s CSO has approved the scientific
disclosure contained in the press release.
About
MedMira
MedMira is a leading developer and manufacturer
of vertical flow rapid diagnostics. The Company’s tests provide
hospitals, labs, clinics and individuals with instant diagnosis for
diseases such as HIV and Hepatitis C in just three easy steps. The
Company’s tests are sold globally under the Reveal®, Multiplo® and
Miriad® brands. Based on its patented Rapid Vertical Flow
Technology™, MedMira’s rapid HIV test is the only one in the world
to achieve regulatory approvals in Canada, the United States, China
and the European Union. MedMira’s corporate offices and
manufacturing facilities are located in Halifax, Nova Scotia,
Canada. For more information visit medmira.com. Follow us on
Twitter and LinkedIn.
This news release contains forward‐looking
statements, which involve risk and uncertainties and reflect the
Company’s current expectation regarding future events, including
statements regarding possible regulatory approval and launch of the
REVEALCOVID-19TM test, future growth, and new business
opportunities. Actual events could materially differ from those
projected herein and depend on a number of factors including, but
not limited to, changing market conditions, successful and timely
completion of clinical studies, uncertainties related to the
regulatory approval process, establishment of corporate alliances
and other risks detailed from time to time in the company quarterly
filings.
Neither TSX Venture Exchange nor its Regulation
Services Provider (as that term is defined in the policies of the
TSX Venture Exchange) accepts responsibility for the adequacy or
accuracy of this release.
MedMira
Contact:
Markus MeileChief Financial Officer, MedMira
Inc.ir@medmira.com
Photos accompanying this announcement are available
at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/6348c97a-9ebb-42e2-b3c2-d7fbdc5b3b93
https://www.globenewswire.com/NewsRoom/AttachmentNg/8ffe70f8-e99a-4efa-a35d-2664b5ba29a1
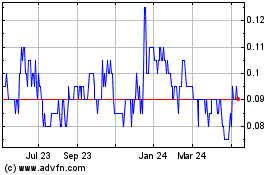
MedMira (TSXV:MIR)
Historical Stock Chart
From Jun 2024 to Jul 2024
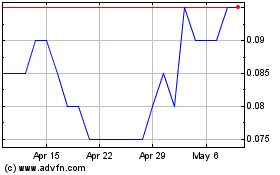
MedMira (TSXV:MIR)
Historical Stock Chart
From Jul 2023 to Jul 2024