Exagen Inc. Announces Clinical Trial Conducted at Major Academic Center in Distinguishing Type 1 and Type 2 Lupus
June 03 2020 - 7:25AM

Exagen Inc. (Nasdaq: XGN), an organization dedicated to
transforming the care continuum for patients suffering from
autoimmune diseases, announced today a clinical trial in
collaboration with Duke University in Durham, NC. The Duke Lupus
Clinic is focused on diagnosing, treating, and providing expert
care to patients living with lupus.
This prospective study will evaluate biomarkers, including the
AVISE panel and Cell-Bound Complement Activation Products
(CB-CAPs), that distinguish between changes in subsets of lupus
patients with Type 1 and Type 2 systemic lupus erythematosus (SLE)
activity. The Type 1 and Type 2 SLE model was developed at Duke by
Megan E.B. Clowse, MD, David Pisetsky, MD, PhD, Jennifer L. Rogers,
MD, and colleagues as a means to incorporate the full spectrum of
lupus symptoms into the clinical assessment and advance
personalized medicine for lupus patients. The model combines
patient- and physician- reported measures to group patients into
two main categories. Type 1 manifestations are the classical lupus
signs and symptoms due to inflammatory processes, and include
butterfly rash, joint inflammation, nephritis or vasculitis.
Patients categorized as Type 2 have a general feeling of being
unwell and may present with fatigue, depression, anxiety,
widespread pain, or difficulty sleeping.
“We’re pleased to be partnering with Duke to study Exagen’s
proprietary CB-CAPs biomarkers in patients with lupus,” said Ron
Rocca, President and CEO of Exagen. “A better understanding of the
presentation and severity of lupus patients’ symptoms will help
further the classification of disease
severity.”
“This collaboration will be the first to apply CB-CAPs to the
Type 1 and Type 2 SLE model,” added Jennifer Rogers, Director of
Duke Lupus Clinic. “When combined with clinical phenotyping, this
study will determine whether the use of these biomarkers could lead
to a more precision-medicine approach to lupus care and,
potentially, improvements in patient outcomes.”
About Exagen Inc.
Exagen is dedicated to transforming the care
continuum for patients suffering from debilitating and chronic
autoimmune diseases by enabling timely differential diagnosis and
optimizing therapeutic intervention. Exagen has developed
and is commercializing a portfolio of innovative testing products
under its AVISE® brand. Several of these products are based on our
proprietary Cell-Bound Complement Activation Products (CB-CAPs),
technology. CB-CAPs assess the activation of the complement system,
a biological pathway implicated in systemic lupus erythematosus
(SLE). Exagen’s goal is to enable rheumatologists to improve care
for patients through the differential diagnosis, prognosis and
monitoring of complex autoimmune and autoimmune-related diseases,
including SLE and rheumatoid arthritis, or RA. Exagen’s model of
integrating testing products and therapeutics
positions Exagen to offer targeted solutions to
rheumatologists and, ultimately, better serve patients. For
more information, please visit www.Exagen.com
Forward Looking Statements
Exagen cautions you that statements contained in this press
release regarding matters that are not historical facts are
forward-looking statements. These statements are based on the
company’s current beliefs and expectations. Such forward-looking
statements include, but are not limited to, express or implied
statements regarding the collaborative clinical trial and potential
advantages of applying our CB-CAPs technology in Type 1 and Type 2
SLE. The inclusion of forward-looking statements should not
be regarded as a representation by Exagen that any of its
plans will be achieved. Actual results may differ from those set
forth in this press release due to the risks and uncertainties
inherent in Exagen’s business, including, without limitation: the
COVID-19 pandemic may continue to adversely affect our business,
financial condition and results of operations, including as a
result of shutdowns of our facilities and operations as well as
those of our suppliers, courier services and our
collaborators, disruptions to the supply chain of material
needed for our tests and our ability to receive specimens and
perform or deliver the results from our tests, and delays in the
planned clinical trial with Duke; and other risks described in the
company’s prior press releases and the company’s filings with
the Securities and Exchange Commission (SEC), including
under the heading “Risk Factors” in the company’s Annual Report on
Form 10-K and any subsequent filings with the SEC. You are
cautioned not to place undue reliance on these forward-looking
statements, which speak only as of the date hereof,
and Exagen undertakes no obligation to update such
statements to reflect events that occur or circumstances that exist
after the date hereof. All forward-looking statements are qualified
in their entirety by this cautionary statement, which is made under
the safe harbor provisions of the Private Securities Litigation
Reform Act of 1995.
Investors Westwicke Partners Mike Cavanaugh
Mike.Cavanaugh@westwicke.com 646.677.1838
Company Exagen Inc. Kamal Adawi, Chief
Financial Officer kadawi@exagen.com 760.477.5514
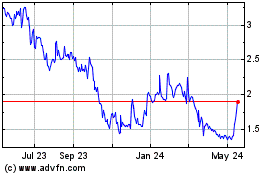
Exagen (NASDAQ:XGN)
Historical Stock Chart
From Apr 2024 to May 2024
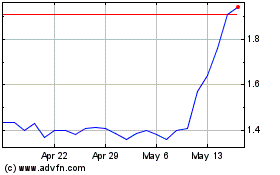
Exagen (NASDAQ:XGN)
Historical Stock Chart
From May 2023 to May 2024