Spruce Biosciences Appoints Libbie Mansell, Ph.D., M.B.A., R.A.C., as Chief Regulatory and Quality Officer
April 11 2022 - 7:00AM
Business Wire
Spruce Biosciences, Inc. (Nasdaq: SPRB), a late-stage
biopharmaceutical company focused on developing and commercializing
novel therapies for rare endocrine disorders with significant unmet
medical need, today announced that Libbie Mansell, Ph.D., M.B.A.,
R.A.C., has been appointed Chief Regulatory and Quality Officer.
Dr. Mansell will be responsible for leading the company’s global
regulatory affairs and quality strategy.
“Dr. Mansell brings extensive regulatory affairs and quality
expertise at precisely the right time for Spruce, as we progress
tildacerfont through clinical development for adults and children
with classic congenital adrenal hyperplasia and women with
polycystic ovary syndrome,” said Javier Szwarcberg, M.D., M.P.H.,
Chief Executive Officer of Spruce Biosciences. “With more than 30
years of life sciences industry experience and a successful track
record of managing complex drug development programs for numerous
biopharmaceutical companies, Dr. Mansell is a strong addition to
our executive team. We look forward to her leadership and guidance
as we continue to advance our pipeline of novel treatments for
patients with rare endocrine disorders.”
Dr. Mansell is a seasoned regulatory affairs, development
strategy and program management professional, with over 30 years of
industry experience in serious and rare diseases. She joins Spruce
from Asklepios BioPharmaceutical (AskBio), where she served as
Senior Vice President of Regulatory Affairs. Prior to AskBio, Dr.
Mansell was Managing Director and Founder of White Oak BioPharma
Solutions, a global regulatory strategy and operations consulting
firm she established in 2006 to serve executive teams at a full
range of companies, from startups to large companies. Prior to
consulting, she held positions of increasing responsibility in
regulatory affairs, pharmacovigilance, quality affairs and
chemistry, manufacturing and controls with several biotechnology
and pharmaceutical companies, including Curis, Sigma-Tau Research,
Genzyme, CombinatoRx, Millennium Pharmaceuticals and Boehringer
Ingelheim Pharmaceuticals. Dr. Mansell earned a Ph.D. in
pharmacokinetics and biopharmaceutics with a graduate minor in
applied statistics from Oregon State University and an M.B.A. in
finance and international business from New York University.
"With multiple late-stage global clinical studies and additional
pipeline programs in development for patients with significant
unmet medical needs, Spruce is at an important stage in its
development and I look forward to contributing to the company’s
evolution,” said Libbie Mansell, Ph.D., M.B.A., R.A.C., Chief
Regulatory and Quality Officer of Spruce Biosciences. “By
leveraging my global regulatory affairs, quality and product
development expertise, I am eager to help make Spruce’s vision – to
transform the lives of those living with rare endocrine disorders –
a reality for the patients and families we serve.”
About Spruce Biosciences
Spruce Biosciences is a late-stage biopharmaceutical company
focused on developing and commercializing novel therapies for rare
endocrine disorders with significant unmet medical need. Spruce is
initially developing its wholly-owned product candidate,
tildacerfont, as the potential first non-steroidal therapy for
patients suffering from classic congenital adrenal hyperplasia
(CAH). Classic CAH is a serious and life-threatening disease with
no known novel therapies approved in approximately 50 years. Spruce
is also developing tildacerfont for women suffering from polycystic
ovary syndrome (PCOS) with primary adrenal androgen excess. To
learn more, visit www.sprucebiosciences.com and follow us on
Twitter @Spruce_Bio, LinkedIn, Facebook and YouTube.
Forward-Looking Statements
Statements contained in this press release regarding matters
that are not historical facts are “forward-looking statements”
within the meaning of the Private Securities Litigation Reform Act
of 1995. Such forward-looking statements include statements
regarding, among other things, the impact of new management hires
and promotions, the fulfillment of Spruce’s strategic business
objectives, and the advancement of Spruce’s drug development
pipeline. Because such statements are subject to risks and
uncertainties, actual results may differ materially from those
expressed or implied by such forward-looking statements. Words such
as “will,” “potential” and similar expressions are intended to
identify forward-looking statements. These forward-looking
statements are based upon Spruce’s current expectations and involve
assumptions that may never materialize or may prove to be
incorrect. Actual results could differ materially from those
anticipated in such forward-looking statements as a result of
various risks and uncertainties, which include, without limitation,
risks and uncertainties associated with Spruce’s business in
general, the impact of the COVID-19 pandemic, and the other risks
described in Spruce’s filings with the U.S. Securities and Exchange
Commission. All forward-looking statements contained in this press
release speak only as of the date on which they were made and are
based on management’s assumptions and estimates as of such date.
Spruce undertakes no obligation to update such statements to
reflect events that occur or circumstances that exist after the
date on which they were made, except as required by law.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220411005057/en/
Media Contact Will Zasadny Canale Communications (619)
961-8848 will.zasadny@canalecomm.com media@sprucebiosciences.com
Investors Xuan Yang Solebury Trout (415) 971-9412
xyang@soleburytrout.com investors@sprucebiosciences.com
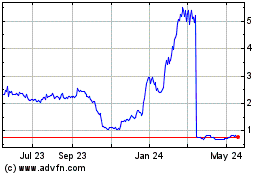
Spruce Biosciences (NASDAQ:SPRB)
Historical Stock Chart
From Apr 2024 to May 2024
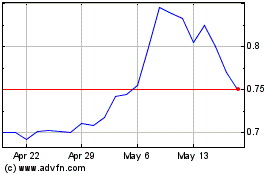
Spruce Biosciences (NASDAQ:SPRB)
Historical Stock Chart
From May 2023 to May 2024