~ The Global Market for Pigmented Lesion Treatment Is
Expected to Reach $3.37 Billion in
2024, and $4.69 Billion by
20291
~ Leading Experts Will Illuminate Clinical Efficacy
of UltraClear's New Indications at the Upcoming SCALE
Symposium for Aesthetic Medicine and Laser Education ~
SMITHFIELD, R.I., May 14, 2024
/PRNewswire/ -- Acclaro Medical, a medical technology company that
specializes in developing and delivering trailblazing solutions for
medical aesthetics and surgical practitioners worldwide, today
announced clearance by the U.S. Food and Drug Administration (FDA)
for a new indication of the Company's UltraClear® cold ablative
fractional 2910 nm fiber laser. The 510K approval expands the use of UltraClear's
novel technology for the treatment of benign pigmented lesions
and vascular dyschromia.
"This clearance underpins our commitment to addressing the unmet
needs of patients living with pigmentation disorders by improving
upon the current treatment paradigm of acids, peels and
passé devices," said Shlomo
Assa, Co-Founder and President, Chief Technology Officer of
Acclaro Medical. "Whether troubled by benign brown lesions or red
discoloration, patients can now experience UltraClear's proven
ability to rejuvenate their physical appearance with excellent
results, reduced discomfort, minimal downtime and very limited
risk."
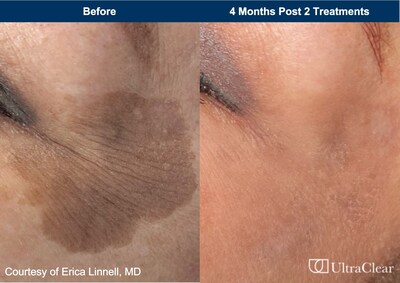
"Any change in skin tone can be concerning or upsetting," noted
Dr. Erica Linnell, Founder MD of
Linnell Dermatology and Aesthetics, Seattle, WA. "With the heightened awareness of
skin cancer checks and an aging population, we have seen a
corresponding rise in patient visits for evaluation and treatment
of their pigmented lesions. The expanded regulatory indication for
the UltraClear fiber laser now paves the way for us to resurface
and make a difference in improving pigmentation issues with just
one to two treatments."
At the upcoming SCALE meeting in Nashville, TN, on May
16-19, 2024, key opinion leaders will showcase the
effectiveness and versatility of UltraClear to treat a vast
array of aesthetic skin conditions, ranging from the early signs of
aging, facial wrinkles and crinkles around the eyes to unwanted
pigmentation, the effects of gravity and scars, including acne,
surgical and traumatic scars. By stimulating new cell growth
leading to collagen and elastin remodeling, UltraClear treatment
produces smoother and healthier-looking skin with minimal medical
downtime. Its advanced pulsing and fractionated technology, which
perfectly balances cold and thermal energy, is suitable for all
skin types and may not require topical numbing, providing a
rewarding patient experience for numerous aesthetic concerns on
patient of all skin colors.
Visit Exhibit Tabletop #808 to learn more about UltraClear at
SCALE.
PODIUM PRESENTATIONS AT SCALE:
Friday, May 17th
11:05-11:15 am
Advances in the use of the 2910nm Er: Fiber Resurfacing
Laser
Brian Biesman, MD,
FACS
Friday, May 17th
3:50-4:05 pm
The All-In-One Solution: Why I Chose UltraClear
Cheri Frey, MD
Saturday, May 18th
12:00 PM- 1:00 PM
Workshop: Live Lasers & EBDs on Stage
Michel Gold, MD
About Acclaro Medical
Acclaro Medical is a
pioneering medical technology company focused on developing
cutting-edge solutions that improve patient care and redefine
medical practices. With a relentless commitment to innovation and a
team of dedicated professionals, Acclaro Medical continues to push
the boundaries and drive positive change in the aesthetic medical
industry. For more information, please visit
http://www.ultraclearlaser.com/
1 Pigmented Lesion Treatment Market Size & Share
Analysis - Growth Trends & Forecasts (2024 - 2029)
Source:
https://www.mordorintelligence.com/industry-reports/pigmented-lesion-treatment-market
For media inquiries, contact:
Nadine Tosk
Nadine Tosk Communications
504.453.8344
nadinepr@gmail.com
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/acclaro-medical-announces-fda-clearance-of-ultraclear-cold-ablative-fractional-2910-nm-fiber-laser-for-treatment-of-benign-pigmented-lesions-and-vascular-dyschromia-302144715.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/acclaro-medical-announces-fda-clearance-of-ultraclear-cold-ablative-fractional-2910-nm-fiber-laser-for-treatment-of-benign-pigmented-lesions-and-vascular-dyschromia-302144715.html
SOURCE Acclaro Medical Corporation