As filed with the Securities
and Exchange Commission on January 27, 2017
Registration No. 333-
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM S-3
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
NEUROMETRIX, INC.
(Exact name of registrant as specified in
its charter)
|
Delaware
|
04-3308180
|
|
(State or other jurisdiction of
|
(I.R.S. Employer
|
|
incorporation or organization)
|
Identification Number)
|
1000 Winter Street
Waltham, MA 02451
(781) 890-9989
(Address, including zip code, and telephone
number, including area code, of registrant’s principal executive offices)
Shai N. Gozani, M.D., Ph.D.
President and Chief Executive Officer
NeuroMetrix, Inc.
1000 Winter Street
Waltham, MA 02451
(781) 890-9989
(Name, address, including zip code, and telephone
number, including area code, of agent for service)
With a copy to:
Megan N. Gates, Esq.
Mintz, Levin, Cohn, Ferris, Glovsky and Popeo,
P.C.
One Financial Center
Boston, MA 02111
Telephone: (617) 542-6000
Fax: (617) 542-2241
Approximate date of commencement of proposed sale to the public:
From time to time after this Registration Statement becomes effective.
If the only securities being registered on this Form are being offered
pursuant to dividend or interest reinvestment plans, please check the following box.
¨
If any of the securities being registered on this Form are to be
offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, other than securities offered
only in connection with dividend or interest reinvestment plans, check the following box.
x
If this Form is filed to register additional securities for an offering
pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities Act registration statement
number of the earlier effective registration statement for the same offering.
¨
If this Form is a post-effective amendment filed pursuant to Rule
462(c) under the Securities Act, please check the following box and list the Securities Act registration statement number of the
earlier effective registration statement for the same offering.
¨
If this Form is a registration statement pursuant to General Instruction
I.D. or a post-effective amendment thereto that shall become effective upon filing with the Commission pursuant to Rule 462(e)
under the Securities Act, check the following box.
¨
If this Form is a post-effective amendment to a registration statement
filed pursuant to General Instruction I.D. filed to register additional securities or additional classes of securities pursuant
to Rule 413(b) under the Securities Act, check the following box.
¨
Indicate by check mark whether the registrant is a large accelerated
filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated
filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
|
Large accelerated filer
|
¨
|
Accelerated filer
|
¨
|
|
|
|
|
|
|
Non-accelerated filer
|
¨
(Do not check if a smaller reporting company)
|
Smaller reporting company
|
x
|
CALCULATION OF REGISTRATION FEE
Title of each Class of
Securities to be Registered
|
|
Amount
to be
Registered (1)
|
|
|
Proposed
Maximum
Offering Price
Per Share
|
|
|
Proposed
Maximum
Aggregate
Offering Price
|
|
|
Amount of
Registration Fee
|
|
|
Common Stock, $0.0001 par value per share
|
|
|
20,750,000
|
(3)
|
|
$
|
0.71
|
(4)
|
|
$
|
14,732,500
|
|
|
$
|
1,707.50
|
|
|
Rights to purchase Series A Junior Participating Cumulative Preferred Stock, $0.001 par value(2)
|
|
|
|
(2)
|
|
|
|
(2)
|
|
|
|
(2)
|
|
|
|
(2)
|
|
|
(1)
|
Pursuant to Rule 416
under the U.S. Securities Act of 1933, as amended (the “Securities Act”), this Registration Statement shall also cover
any additional shares of common stock which become issuable by reason of any stock dividend, stock split or other similar transaction
that results in an increase in the number of the outstanding shares of common stock of the Registrant.
|
|
|
(2)
|
Pursuant to a shareholder
rights agreement, dated as of March 7, 2007, between the Company and American Stock Transfer & Trust Company, as amended,
each share of common stock has an attached right to purchase our Series A Junior Cumulative Preferred Stock, which rights are
not currently exercisable.
|
|
|
(3)
|
The number of shares of common
stock includes 10,000,000 shares of common stock issuable upon conversion of the Company’s outstanding shares of Series
E Convertible Preferred Stock (the “Series E Preferred Stock”) and 10,750,000 shares of common stock issuable upon
exercise of certain of the Company’s warrants (the “Warrants”).
|
|
|
(4)
|
In accordance with Rule 457(c)
under the Securities Act, the aggregate offering price of the common stock is estimated solely for the calculation of the registration
fees due for this filing. This estimate was based on the average of the high and low sales price of our stock reported by The
NASDAQ Capital Market on January 23, 2017.
|
The Registrant hereby amends this Registration Statement on such
date or dates as may be necessary to delay its effective date until the Registrant shall file a further amendment which specifically
states that this Registration Statement shall thereafter become effective in accordance with Section 8(a) of the Securities
Act of 1933 or until the Registration Statement shall become effective on such date as the Commission, acting pursuant to said
Section 8(a), may determine.
THE INFORMATION IN THIS PROSPECTUS IS NOT COMPLETE
AND MAY BE CHANGED. WE MAY NOT SELL THESE SECURITIES UNTIL THE REGISTRATION STATEMENT FILED WITH THE SECURITIES AND EXCHANGE COMMISSION
IS EFFECTIVE. THIS PROSPECTUS IS NOT AN OFFER TO SELL THESE SECURITIES, AND WE ARE NOT SOLICITING OFFERS TO BUY THESE SECURITIES
IN ANY STATE WHERE THE OFFER OR SALE IS NOT PERMITTED.
Subject to Completion, Dated
January 27, 2017
PROSPECTUS
NEUROMETRIX, INC.
20,750,000 Shares of Common Stock
This prospectus relates to the resale of up
to 20,750,000 shares of our common stock, including 10,000,000 shares of our common stock issuable upon conversion of outstanding
shares of Series E Preferred Stock and 10,750,000 shares of our common stock issuable upon exercise of certain outstanding Warrants.
These shares will be resold from time to time
by the entities listed in the section titled “Selling Security holders” beginning on page 25, which we refer to as
the selling security holders or Selling Stockholders. The shares of common stock offered under this prospectus by the selling security
holders have been issued, or are issuable upon conversion or exercise of securities issued, pursuant to the Securities Purchase
Agreement by and among NeuroMetrix, Inc. and certain of the selling security holders, dated as of December 28, 2016 (the “Purchase
Agreement”),and pursuant to an engagement letter, dated as of June 2, 2016, as amended, with H.C. Wainwright & Co., LLC,
the placement agent for the offering made pursuant to the Purchase Agreement (the “Engagement Letter”). We are not
selling any securities under this prospectus and will not receive any of the proceeds from the sale of securities by the selling
security holders.
The selling security holders may sell the shares
of common stock described in this prospectus in a number of different ways and at varying prices. We provide more information about
how a selling security holder may sell its shares of common stock in the section titled “Plan of Distribution” on page
29. We will pay the expenses incurred in registering the securities covered by the prospectus, including legal and accounting
fees.
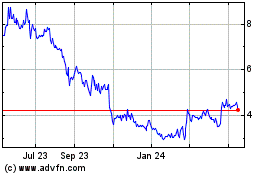
Our common stock is quoted on The NASDAQ Capital
Market, or NASDAQ, under the symbol “NURO.” On January 26, 2017, the last reported sale price of our common stock was
$0.78 per share.
Investing in our securities involves risks. See “
Risk
Factors
” beginning on page 8 of this prospectus.
Neither the Securities and Exchange Commission nor any state
securities commission has approved or disapproved of these securities or passed upon the adequacy or accuracy of this prospectus.
Any representation to the contrary is a criminal offense.
THE DATE OF THIS PROSPECTUS IS FEBRUARY __,
2017.
TABLE OF CONTENTS
INFORMATION CONTAINED IN THIS PROSPECTUS
You should rely only on the information contained
or incorporated by reference into this prospectus. We have not, and the selling security holders have not, authorized anyone to
provide you with additional or different information. These securities are not being offered in any jurisdiction where the offer
is not permitted. You should assume that the information in this prospectus is accurate only as of the date on the front of the
document and that any information we have incorporated by reference is accurate only as of the date of the documents incorporated
by reference, regardless of the time of delivery of this prospectus or of any sale of our common stock. Unless the context otherwise
requires, references to “we,” “our,” “us,” “NeuroMetrix ,” or the “Company”
in this prospectus mean NeuroMetrix, Inc.
PROSPECTUS SUMMARY
The following is only a summary. We urge
you to read the entire prospectus, including the more detailed financial statements, notes to the financial statements and other
information included herein or incorporated by reference from our other filings with the U.S. Securities and Exchange Commission
(SEC). Investing in our securities involves risks. Therefore, please carefully consider the information provided under the heading
“Risk Factors” beginning on page 8.
Our Business – An Overview
NeuroMetrix is a commercial stage, innovation
driven healthcare company combining bioelectrical and digital medicine to address chronic health conditions including chronic pain,
sleep disorders, and diabetes. Our business is fully integrated with in-house capabilities spanning product development, manufacturing,
regulatory affairs and compliance, sales and marketing, and customer support. We derive revenues from the sale of medical devices
and after-market consumable products and accessories. Our products are sold in the United States and selected overseas markets,
and are cleared by the U.S. Food and Drug Administration, or FDA, and regulators in foreign jurisdictions where appropriate. We
have two principal product lines:
|
|
·
|
Wearable neuro-stimulation
therapeutic devices
|
|
|
·
|
Point-of-care neuropathy diagnostic tests
|
Our core expertise in biomedical engineering
has been refined over nearly two decades of designing, building and marketing medical devices that stimulate nerves and analyze
nerve response for diagnostic and therapeutic purposes. We created the market for point-of-care nerve testing and were first to
market with sophisticated, wearable technology for management of chronic pain. We also have an experienced management team and
Board of Directors.
Chronic pain is a significant public health problem.
It is defined by the National Institutes of Health as any pain lasting more than 12 weeks in contrast to acute pain which is a
normal bodily response to injury or trauma. Chronic pain conditions include painful diabetic neuropathy, or PDN, arthritis, fibromyalgia,
sciatica, musculoskeletal pain, cancer pain and many others. Chronic pain may be triggered by an injury or there may be an ongoing
cause such as disease or illness. There may also be no clear cause. Pain signals continue to be transmitted in the nervous system
over extended periods of time often leading to other health problems. These can include fatigue, sleep disturbance, decreased appetite,
and mood changes which cause difficulty in carrying out important activities and contributing to disability and despair. In general,
chronic pain cannot be cured. Treatment of chronic pain is focused on reducing pain and improving function. The goal is effective
pain management.
Chronic pain is widespread. It affects over 100
million adults in the United States and more than 1.5 billion people worldwide. The global market for pain management drugs and
devices alone was valued at $35 billion in 2012. The estimated incremental impact of chronic pain on health care costs in the United
States is over $250 billion per year and lost productivity is estimated to exceed $300 billion per year. Estimated out-of-pocket
spending in the United States on chronic pain is $20 billion per year.
The most common approach to chronic pain is pain
medication. This includes over-the-counter drugs (such as Advil and Motrin), and prescription drugs including anti-convulsants
(such as Lyrica and Neurontin) and anti-depressants (such as Cymbalta and Elavil). Topical creams may also be used (such as Zostrix
and Bengay). With severe pain, narcotic pain medications may be prescribed (such as codeine, fentanyl, morphine, and oxycodone).
The approach to treatment is individualized, drug combinations may be employed, and the results are often hit or miss. Side effects
and the potential for addiction are real and the risks are substantial.
Reflecting the difficulty in treating chronic
pain, we believe that inadequate relief leads 25% to 50% of pain sufferers to turn to the over-the-counter market for supplements
or alternatives to prescription pain medications. These include non-prescription medications, topical creams, lotions, electrical
stimulators, dietary products, braces, sleeves, pads and other items. In total they account for over $4 billion in annual spending
in the United States on pain relief products.
High frequency nerve stimulation is an established
treatment for chronic pain supported by numerous clinical studies demonstrating efficacy. In simplified outline, the mechanism
of action involves intensive nerve stimulation to activate the body’s central pain inhibition system resulting in widespread
analgesia, or pain relief. The nerve stimulation activates brainstem pain centers leading to the release of endogenous opioids
that act primarily through the delta opioid receptor to reduce pain signal transmission through the central nervous system. This
therapeutic approach is available through deep brain stimulation and through implantable spinal cord stimulation, both of which
require surgery and have attendant risks. Non-invasive approaches to neuro-stimulation (transcutaneous electrical nerve stimulation,
or TENS) have achieved limited efficacy in practice due to device limitations, ineffective dosing and low patient compliance.
Our Strategy
There are large and important unmet medical
needs in chronic pain treatment. Prescription pain medications and over-the-counter therapies are often inadequate and can lead
to other health issues. We believe that controlled, personalized, neuro-stimulation to suppress pain provides an important complement
to pain medications. As a medical device company with unique experience in designing devices to manage and alter peripheral nerve
function, we believe we are well positioned to make neuro-stimulation widely available to chronic pain sufferers. We have direct
experience with neuro-stimulation through our prescription SENSUS wearable pain management device which has been on the market
for the past three years and Quell, our over-the-counter, or OTC, wearable device for pain relief which was launched in the second
quarter of 2015 and builds upon the core SENSUS neuro-stimulation technology.
Our primary objective is revenue growth. We
expect this to be led by the successful market adoption of Quell. We also expect an important contribution to revenue from DPNCheck,
our rapid, accurate diagnostic test for diabetic peripheral neuropathy.
Our key business strategies include:
Driving Commercial Adoption of Key Proprietary
Products.
|
|
·
|
Quell
, our OTC
wearable device for pain relief, was made commercially available in the United States during the second quarter of 2015. Following
commercial launch through the end of the third quarter of 2016, approximately 45,000 Quell devices plus electrodes and accessories
were shipped to consumers. Quell revenues for the year ended December 31, 2015 and for the nine-months ended September 30, 2016
were approximately $2.1 million and $4.9 million, respectively. Quell utilizes OptiTherapy, our proprietary non-invasive neuro-stimulation
technology to provide relief from chronic intractable pain, such as nerve pain due to diabetes, fibromyalgia, arthritic pain,
and lower back and leg pain. This advanced wearable device is lightweight and can be worn during the day while active, and at
night while sleeping. It has been cleared by the FDA for treatment of chronic intractable pain without a doctor’s prescription.
Users of the device have the option of using their smartphones to control pain therapy and to track sleep and therapy parameters.
Quell is distributed in North America via e-commerce, including the Company’s website (www.quellrelief.com) and Amazon,
via direct response television including QVC, via retail merchandisers including Target, CVS and Walgreens, and via health care
professionals such as pain management physician practices and podiatry practices. Distribution is supported by television promotion
to expand product awareness. We believe there are significant opportunities to market Quell outside of the United States, particularly
in Western Europe, Japan and China. In June 2016, we filed with the European Medicines Agency for regulatory approval to market
Quell in the European Union and, assuming we receive such approval, we plan to initiate marketing during 2017.
|
|
|
·
|
DPNCheck
, our diagnostic test for peripheral neuropathies, was made commercially available in the fourth quarter of 2011. DPNCheck revenues for the years ended December 31, 2015 and 2014 were approximately $2.3 million and $1.8 million, respectively. DPNCheck revenues for the nine-months ended September 30, 2016 were approximately $1.7 million. Our US sales efforts focus on Medicare Advantage providers who assume financial responsibility and the associated risks for the health care costs of their patients. We believe that DPNCheck presents an attractive clinical case with early detection of neuropathy allowing for earlier clinical intervention to help mitigate the effects of neuropathy on both patient quality of life and cost of care. Also, the diagnosis and documentation of neuropathy provided by DPNCheck helps clarify the patient health profile which, in turn, may have a direct, positive effect on the Medicare Advantage premium received by the provider. We believe that attractive growth opportunities exist outside the United States, including Japan where we launched DPNCheck with our distribution partner Omron Healthcare in the third quarter of 2014; in China where we recently received regulatory approval and initiated sales in the fourth quarter of 2016 with Omron Healthcare after receiving regulatory approval in the first quarter of 2016; and in Mexico where our distributor Scienta Farma received regulatory approval and initiated sales in the fourth quarter of 2015.
|
Maintaining a High Level of Research and Development
Productivity.
Our research and development, or R&D, team successfully delivered Quell, an FDA cleared, technologically
sophisticated, smart phone integrated product with electrodes and other accessories. We believe that there are no comparable products
on the market. Our R&D team is now charged with maintaining and expanding this Quell competitive technological advantage, addressing
opportunities to reduce Quell cost of goods sold, and enhancing our intellectual property position, through continuing innovation.
We expect innovation to take the form of device and software enhancements to improve the user experience, expanded smart phone
applications, and new electrode features to optimize therapy. Technological innovation will continue to be one of our top priorities.
Our Business Model
Our products consist of a medical device used
in conjunction with a consumable electrode or biosensor. Other accessories and consumables are also available to customers. Our
goal for these devices is to build an installed base of active customer accounts and distributors that regularly order aftermarket
products to meet their needs. We successfully implemented this model when we started our business with the NC-stat system and applied
it to subsequent product generations including the ADVANCE system. Our recently developed products, Quell, SENSUS and DPNCheck,
conform to this model.
Marketed Products
Quell
Quell is a wearable device for relief of chronic
intractable pain, such as nerve pain due to diabetes and lower back problems. It incorporates our OptiTherapy technology, a collection
of proprietary approaches designed to optimize the clinical efficacy of nerve stimulation. These include high power electrical
stimulation hardware with precise control, algorithms that automatically determine therapeutic stimulation intensity and compensate
for nerve desensitization, and automated detection of user sleep and appropriate adjustment of stimulation level. Quell is comprised
of (1) an electronic device carried in a neoprene band that is worn on the upper calf and (2) an electrode that attaches to the
device and is the interface between the device and the skin. The device is lightweight and can be worn during the day while active,
and at night while sleeping. It has been cleared by the FDA for treatment of chronic intractable pain and is available OTC. Users
of the device have the option of using their smartphones to control pain therapy and to track sleep and therapy parameters. The
device was made commercially available in June 2015. In an independent post-market clinical study of Quell initiated by NeuroMetrix,
81% of subjects reported an improvement in management of their chronic pain and health, and 67% reported a reduction in their use
of pain medications. To encourage persons with chronic pain to try Quell, we offer a 60-day trial period during which the product
can be returned for a full refund. To date, product returns have averaged 28%. We estimate, over time, we will see product returns
in the range of 20% to 25%, as indicated by the results of the post-market clinical study. The addressable market opportunity for
Quell in the United States is estimated to be 19 million chronic pain sufferers. Quell is available via e-commerce on our product
website (quellrelief.com) and on Amazon, via direct response television including QVC, via retail merchandisers including Target,
CVS and Walgreens, and via select health care professionals. Distribution is supported by television promotion designed to expand
product awareness. Following commercial launch through the third quarter of 2016 approximately 45,000 devices and accessories were
shipped to customers with a total invoiced value of $10.2 million prior to the impact of product returns.
SENSUS
The SENSUS pain therapy device, the technological
predecessor to Quell, is a prescription neuro-stimulation device based on TENS for relief of chronic, intractable pain. SENSUS,
which was commercially launched in the first quarter of 2013, is a convenient and wearable device that offers physicians and their
patients a non-narcotic pain relief option as a complement to medications. SENSUS is comprised of: (1) an electronic device with
a strap that is worn on the upper calf and (2) an electrode which attaches to the device. We provide prescribing physicians with
PC-based software that links to the device via a USB connection, thereby allowing them to download a record of the patient’s
use of the device. The SENSUS device and electrodes were cleared by the FDA for commercial distribution. When medically indicated
and supported by proper documentation, TENS devices are generally reimbursed by Medicare and many commercial insurance companies
under the DME benefit. SENSUS customers have purchased approximately 10,300 devices through September 30, 2016. We believe that
the launch of Quell and contraction of the DME distribution channel due to Medicare competitive bidding will significantly reduce
future opportunities for SENSUS sales. Accordingly, we believe SENSUS will have a limited impact on future revenues.
DPNCheck
DPNCheck is a fast, accurate, and quantitative
nerve conduction test that is used to evaluate systemic neuropathies such as diabetic peripheral neuropathy, or DPN. It is designed
to be used by primary care physicians, endocrinologists, podiatrists and other clinicians at the point-of-care to objectively detect,
stage, and monitor DPN. The device measures nerve conduction velocity and response amplitude of the sural nerve, a nerve in the
lower leg and ankle. These parameters are widely recognized as sensitive and specific biomarkers of DPN. DPNCheck is comprised
of: (1) an electronic hand-held device and (2) a single patient use biosensor. In addition, we provide users with PC-based software
that links to the device via a USB connection. This PC software allows physicians to generate reports and manage their sural nerve
conduction data.
DPNCheck is a modified version of our previously
marketed NC-stat nerve testing device that has the same clinical indications with respect to DPN. The modified device which costs
less than the original device, has the same functionality with respect to sural nerve testing. More than 2.4 million patient studies
have been performed using our NC-stat technology and there have been approximately 7.0 million nerve tests. It has been the subject
of many published studies, including several studies specifically addressing the accuracy and clinical utility of the device in
assessment of DPN. DPNCheck shipments commenced in late 2011 and approximately 3,200 devices had been placed with customers through
September 30, 2016.
ADVANCE System
Our legacy neurodiagnostics business is based
on the ADVANCE NCS/EMG System, or the ADVANCE System, which is a comprehensive platform for the performance of traditional nerve
conduction studies. The ADVANCE System is comprised of: (1) the ADVANCE device and related modules, (2) various types of electrodes
and needles, and (3) a communication hub that enables the physician’s office to network their device to their personal computers
and our servers for data archiving, report generation, and other network services. The ADVANCE System is most commonly used with
proprietary nerve specific electrode arrays. These electrode arrays combine multiple individual electrodes and embedded microelectronic
components into a single patient-use disposable unit. We currently market seven different nerve specific electrode arrays but do
not actively market the ADVANCE device.
Historically, the ADVANCE System was marketed to a broad range of
physician specialties including neurologists, orthopedic surgeons, primary care physicians, and endocrinologists, and utilized
for a variety of different clinical indications including assessment of carpal tunnel syndrome, or CTS, low back and leg pain,
and DPN. It is most commonly used in the assessment of CTS. Numerous papers have been published on the use of this technology in
this clinical application. More than 2.4 million patient studies have been performed using our NC stat technology and there have
been approximately 7.0 million nerve tests, including 1.3 million sural nerve tests. As of September 30, 2016, we had an installed
base of approximately 400 active customers using our ADVANCE System.
Legacy Neurodiagnostics Business
We were founded in 1996 as a science-based
health care company. Our focus had been the development of innovative products for the detection, diagnosis, and monitoring of
peripheral nerve and spinal cord disorders, such as those associated with carpal tunnel syndrome, lumbosacral disc disease and
spinal stenosis, and diabetes. Our NC-stat System for the performance of nerve conduction studies at the point-of-care was commercially
launched in 1999. The second generation NC-stat was released in 2002. In 2008, we brought to market the more sophisticated ADVANCE
System for nerve conduction testing and performance of invasive needle electromyography. These systems were general purpose with
broad application in evaluating and diagnosing nerve disorders. Numerous studies demonstrating the clinical accuracy and utility
of these devices have been conducted and published in high quality peer-reviewed journals. Furthermore, these devices have been
used in FDA sanctioned clinical trials for pharmacological agents and large scale epidemiological studies sponsored by the NIH,
Center for Disease Control, or CDC, and other governmental agencies. The products have been cleared by the FDA, field tested for
over a decade and highly regarded for their ease of use, accuracy and reproducibility of results.
Following launch of NC-stat in 1999, we experienced
rapid revenue growth, which led to our initial public offering in 2004. The health market, particularly the physician office segment,
embraced the opportunity to perform nerve conduction tests which previously had always required referral to specialists. Point-of-care
nerve testing was seen to provide a combination of improved patient care and patient convenience. The success of point-of-care
nerve testing, a market which we created, was met with resistance in some sectors of the medical community, particularly by neurologists
and physical medicine and rehabilitation physicians, both of which had traditionally provided nerve testing services. As a consequence
of successful lobbying by these specialists, physicians using our technology experienced increased denials of coverage by third
party payers resulting in their discontinuing usage and our difficulty in accruing new customer accounts. In late 2009 CMS included
in the Physician Fee Schedule a new Category I CPT Code, CPT 95905, for nerve conduction studies performed using preconfigured
electrode such as those employed with our products. During 2010 most Medicare fiscal intermediaries assumed coverage for CPT 95905
for some clinical indications; however, the health care environment was such that we were unable to secure broad coverage among
private payers, which is essential to the success of our ADVANCE System product. This experience was reflected in our revenues
for the legacy Neurodiagnostics business, which peaked in 2006 at $55.3 million. We reported revenue for our legacy Neurodiagnostics
business of $2.3 million, $2.8 million and $3.8 million in 2015, 2014 and 2013, respectively. Revenues for our legacy Neurodiagnostics
business for the nine-months ended September 30, 2016 were approximately $1.6 million. We currently manage this business to optimize
cash flow.
Risks Affecting Us
Our business is subject to
numerous risks, as discussed more fully in the section entitled “Risk Factors” immediately following this
prospectus summary. At September 30, 2016 we had an accumulated deficit of $175.7 million and held cash and cash equivalents
of $7.6 million. We believe that these resources, the cash to be generated from expected product sales and, assuming we sell
the securities registered under this registration statement, the net proceeds from this offering will be sufficient to meet
our projected operating requirements into the fourth quarter of 2017. However, the amount of our future product sales
is difficult to predict, especially in light of the limited nature of the recent commercialization of Quell, and actual
sales may not be in line with our forecasts. We continue to face significant challenges and uncertainties and, as a result,
our available capital resources may be consumed more rapidly than currently expected. Accordingly, we will need to
raise additional funds along with the securities registered under this registration statement to support our operating and
capital needs for the fourth quarter of 2017 and beyond. However, we may not be able to secure such financing in a timely
manner or on favorable terms, if at all. Without additional funds, we may be forced to delay, scale back or eliminate some of
our sales and marketing efforts, research and development activities, or other operations and potentially delay product
development in an effort to provide sufficient funds to continue our operations.
Our Corporate Information
Our President and Chief Executive Officer,
Shai N. Gozani, M.D., Ph.D. founded NeuroMetrix in June 1996. We are incorporated in Delaware. Our common stock is listed on The
NASDAQ Capital Market under the ticker symbol “NURO.” Our principal offices are now located at 1000 Winter Street,
Waltham, Massachusetts 02451. Our telephone number is (781) 890-9989. Our web site is
www.neurometrix.com
. We have not incorporated
by reference into this prospectus the information on our website, and you should not consider it to be a part of this document.
Our web site address is included in this document as an inactive textual reference only. The NeuroMetrix name and logo and the
names of products and services offered by NeuroMetrix are trademarks, registered trademarks, service marks or registered service
marks of NeuroMetrix.
Offering of Preferred Stock and Warrants
On December 28, 2016, we entered into a securities
purchase agreement, or the Purchase Agreement, with Sabby Capital Management, LLC and its affiliates, or Sabby, pursuant to which
we agreed to issue (i) up to 7,000 shares of our Series E convertible preferred stock, or the Series E Preferred Stock, at a purchase
price of $1,000 per share and (ii) warrants to purchase up to 10,000,000 shares of our common stock, or the Offering Warrants,
in two tranches, or the Offering. The initial closing of the Offering occurred on January 5, 2017, or the Initial Closing, and
included the issuance of 4,000 shares of Series E Preferred Stock and Offering Warrants to purchase 5,714,286 shares of our common
stock for an aggregate purchase price of $4.0 million. The issuance and sale of Series E Preferred Stock and Warrants was exempt
from registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended, or the Securities Act. In addition, pursuant
to the terms of the Engagement Letter, we will issue up to 750,000 warrants to purchase shares of our common stock to H.C. Wainwright
& Co., LLC and related persons, or the HCW Warrants and together with the Offering Warrants, the Warrants. In connection with
the Initial Closing, we issued HCW Warrants to purchase 428,571 shares of our common stock.
Pursuant to the terms of the Purchase Agreement,
we are seeking stockholder approval to allow us to issue an additional (i) 3,000 shares of Series E Preferred Stock and (ii) Offering
Warrants to purchase 4,285,714 shares of our common stock to Sabby, or the Second Closing. NASDAQ Marketplace Rules require us
to obtain stockholder approval prior to the issuance or potential issuance by us of shares of our common stock (or securities convertible
into or exercisable for shares of our common stock) at a price less than the greater of book or market value if such issuance would
represent 20% or more of our common stock or our outstanding voting power prior to the issuance. The Second Closing will occur
within five (5) business days after the Registration Statement covering the resale of the shares underlying the Series E Preferred
Stock and Warrants, of which this prospectus is a part, becomes effective. Additionally, in connection with the Second Closing,
we will issue additional HCW Warrants to purchase an additional 321,429 shares of our common stock.
Each share of Series E Preferred Stock has
a stated value of $1,000 and is convertible, at any time at the option of the holder thereof, into a number of shares of our common
stock determined by dividing the stated value by the initial conversion price of $0.70, subject to the 4.99% ownership limitation
described below. The Series E Preferred Stock has no dividend rights, liquidation preference or other preferences over our common
stock and has no voting rights except as provided in the Certificates of Designation of Preferences, Rights and Limitations for
the Convertible Preferred Stock, as filed with the Secretary of State of the State of Delaware, or as otherwise required by law.
Shares of common stock underlying the Series E Preferred Stock are being registered for resale by the selling security holders
pursuant to the Registration Statement of which this prospectus forms a part.
The Warrants to purchase an aggregate of 10,750,000
shares of common stock are exercisable at any time after the six month anniversary of their issuance. The Warrants include the
Offering Warrants to purchase 10,000,000 shares of common stock, which expire five years from their initial date of issuance. All
of the Warrants issued in the Initial Closing have an exercise price of $0.92 per share, which will be reset to $0.70 per share
upon receipt of stockholder approval of the Second Closing. Shares of common stock underlying the aggregate of 10,750,000 Warrants
are being registered for resale by the selling security holders pursuant to the Registration Statement of which this prospectus
forms a part. The Warrants are subject to certain ownership limitations as described below.
The Series E Preferred Stock and the Warrants
contain limitations that prevent the holder of any Series E Preferred Stock or Warrants from acquiring shares upon conversion of
the Series E Preferred Stock, or exercise of a Warrant, that would result in the number of shares beneficially owned by it and
its affiliates exceeding 4.99% of the total number of shares of our common stock then issued and outstanding. In addition, upon
certain changes in control of NeuroMetrix, the holder of shares of Series E Preferred Stock or Warrants can elect to receive, subject
to certain limitations and assumptions, securities in a successor entity equal to the value of the Series E Preferred Stock or
Warrants or if holders of common stock are given a choice of cash or property, then cash or property equal to the value of the
outstanding Series E Preferred Stock or Warrants. The Series E Preferred Stock and Warrants were issued pursuant to the Purchase
Agreement and the Engagement Letter (as both are defined on the cover of this prospectus).
RISK FACTORS
Investing in our securities involves a high
degree of risk. You should carefully review and consider the following risk factors and in the section entitled “Risk Factors”
contained in our most recent annual report on Form 10-K, which has been filed with the SEC and is incorporated by reference in
this prospectus, as well as any updates thereto contained in subsequent filings with the SEC or in any free writing prospectus
and all other information contained in this prospectus and incorporated by reference into the prospectus before purchasing our
securities. The risks and uncertainties described below are not the only ones facing us. Additional risks and uncertainties that
we are unaware of, or that we currently deem immaterial, also may become important factors that affect us. If any of the following
risks occur, our business, financial condition or results of operations could be materially and adversely affected. In that case,
the trading price of our common stock could decline, and you may lose some or all of your investment.
Risks Related to Our Business
We have incurred significant operating losses since inception
and cannot assure you that we will achieve profitability.
We have incurred significant
cumulative net losses since our inception. Our net losses for the nine months ended September 30, 2016 and the years ended
December 31, 2015, 2014 and 2013, were approximately $12.1 million, $9.2 million, $7.8 million, and $8.0 million,
respectively. At September 30, 2016, we had an accumulated deficit of $175.7 million. The extent of our future operating
income or losses is highly uncertain, and we cannot assure you that we will be able to achieve or maintain profitability.
Our future capital needs are uncertain and our independent auditor
has expressed substantial doubt about our ability to continue as a going concern. Our ability to continue as a going concern is
dependent on our ability to raise additional capital and our operations could be curtailed if we are unable to obtain the required
additional funding when needed. We may not be able to do so when necessary, and/or the terms of any financings may not be advantageous
to us.
We held cash and cash equivalents of $7.6 million
as of September 30, 2016. We believe that these resources, and the cash to be generated from future product sales and, assuming
we sell all of the securities registered under this registration statement, the net proceeds from this offering will be sufficient
to meet our projected operating requirements into the fourth quarter of 2017. However, the amount of our future product sales
is difficult to predict, especially in light of the limited nature of the recent commercialization of Quell, and actual sales may
not be in line with our forecasts.
Our financial statements have been prepared
assuming that we will continue as a going concern which contemplates the realization of assets and satisfaction of liabilities
in the normal course of business. We expect to incur further losses as we aim to further commercialize Quell and DPNCheck and the
operations of our business and will be dependent on funding our operations through additional public or private financing, collaborative
arrangements with strategic partners, or through additional credit lines or other debt financing sources to increase the funds
available to fund operations. These circumstances raise substantial doubt about our ability to continue as a going concern. As
a result of this uncertainty and the substantial doubt about our ability to continue as a going concern as of December 31, 2015,
the report of our independent registered public accounting firm in our Annual Report on Form 10-K for the year ended December 31,
2015 includes a going concern explanatory paragraph. Management’s plans include increasing revenue through the commercialization
of Quell and DPNCheck. However, no assurance can be given at this time as to whether we will be able to achieve these objectives.
Our financial statements do not include any adjustment relating to the recoverability and classification of recorded asset amounts
or the amounts and classification of liabilities that might be necessary should we be unable to continue as a going concern.
We continue to face significant
challenges and uncertainties and, as a result, our available capital resources may be consumed more rapidly than currently
expected due to (a) decreases in sales of our products and the uncertainty of future revenues from new products; (b) changes
we may make to the business that affect ongoing operating expenses; (c) changes we may make in our business strategy; (d)
regulatory developments affecting our existing products and delays in the FDA approval process for products under
development; (e) changes in our research and development spending plans; and (f) other items affecting our forecasted level
of expenditures and use of cash resources. Accordingly, we will need to raise additional funds, even if we issue all of the
securities registered under this registration statement, to support our future operating and capital needs for the fourth
quarter of 2017 and beyond. We may attempt to obtain additional funding through public or private financing, collaborative
arrangements with strategic partners, or through additional credit lines or other debt financing sources to increase the
funds available to fund operations. However, we may not be able to secure such financing in a timely manner or on favorable
terms, if at all. Furthermore, if we issue equity or debt securities to raise additional funds, our existing stockholders may
experience dilution, and the new equity or debt securities may have rights, preferences and privileges senior to those of our
existing stockholders. If we raise additional funds through collaboration, licensing or other similar arrangements, it may be
necessary to relinquish valuable rights to our potential products or proprietary technologies, or grant licenses on terms
that are not favorable to us. Without additional funds, we may be forced to delay, scale back or eliminate some of our sales
and marketing efforts, research and development activities, or other operations and potentially delay product development in
an effort to provide sufficient funds to continue our operations. If any of these events occurs, our ability to achieve our
development and commercialization goals would be adversely affected.
We are
focused on commercialization of Quell, our over-the-counter, or OTC, wearable device for chronic pain. We cannot assure you that
we will be successful in this field or that our current commercial product for peripheral neuropathy, DPNCheck, or the product
candidates or product enhancements in our development pipeline, will be successful.
We are focused on the commercialization
of Quell, our OTC wearable device for pain relief. Quell is based on our prescription product for pain relief, SENSUS. Quell
has been on the market since June 2015 and we have shipped approximately 45,000 Quell devices through September 30, 2016.
Additionally, DPNCheck, which was launched in 2011, is a quantitative nerve conduction test for systemic neuropathies, such
as DPN. We also have other product candidates and product enhancements in our development pipeline. Our future prospects are
closely tied to our success with Quell and DPNCheck, which, in turn, depend upon market acceptance and growth in future
revenues. We cannot assure you that our commercialization strategy will be successful. If our strategy is not successful, it
could materially affect our revenues and results of operations.
Our future success could be adversely affected
by a number of factors, including:
|
|
·
|
inability to create
market demand for Quell through online marketing efforts, direct response television retail merchandisers and health care professional
channels;
|
|
|
·
|
manufacturing issues with Quell or our other products;
|
|
|
·
|
inability to increase adoption of DPNCheck within the Medicare Advantage market;
|
|
|
·
|
unfavorable market response to DPNCheck in Japan, China and Mexico;
|
|
|
·
|
unfavorable changes to current Medicare, Medicare Advantage and commercial payer payment policies;
|
|
|
·
|
changes to payor policies under the Patient Protection and Affordable Care Act;
|
|
|
·
|
unfavorable experiences by patients and physicians using Quell and our other products; and,
|
|
|
·
|
physicians’ reluctance to alter their existing practices and adopt the use of our devices.
|
If we are unable to expand exposure and penetrate
the market for Quell and/or DPNCheck, our ability to increase our revenues will be limited and our business prospects will be adversely
affected.
Our current and future revenue is dependent upon commercial acceptance
of Quell by the market. The failure of such acceptance will materially and adversely affect our operations.
We anticipate that as revenue from our legacy
neurodiagnostics business, the ADVANCE System, continues to decrease, we will continue to rely heavily on revenue from sales of
Quell, our OTC wearable device. As a result, we will continue to incur operating losses until such time as sales of Quell and other
products or product candidates reach a mature level and we are able to generate sufficient revenue from their sale to meet our
operating expenses. There can be no assurance that customers will adopt our technology and products, or that prospective customers
will agree to pay for our products. In the event that we are not able to significantly increase the number of customers that purchase
our products, or if we are unable to charge the necessary prices, our financial condition and results of operations will be materially
and adversely affected.
If health care providers are unable to obtain sufficient reimbursement
or other financial incentives from third-party health care payers related to the use of our products other than Quell, their adoption
and our future product sales will be materially adversely affected.
Widespread adoption of our DPNCheck products
by the medical community is unlikely to occur without a financial incentive from third-party payers for the use of these products.
If health care providers are unable to obtain adequate reimbursement for procedures performed using these products, if managed
care organizations do not receive improved capitated payments due to more accurate patient risk assessment using our products,
and if DME suppliers are not adequately reimbursed for supplying our therapeutic products, we may be unable to sell our products
at levels that are sufficient to allow us to achieve and maintain profitability, and our business would suffer significantly. Additionally,
even if these products and procedures are adequately reimbursed by third-party payers today, adverse changes in payers’ future
policies toward payment would harm our ability to market and sell our products. Third-party payers include those governmental programs
such as Medicare and Medicaid, private health insurers, workers’ compensation programs and other organizations.
Future regulatory action by CMS or other governmental
agencies or negative clinical results may diminish reimbursement payments to physicians for performing procedures using our products.
Medicaid reimbursement differs from state to state, and some state Medicaid programs may not cover the procedures performed with
our products or pay physicians an adequate amount for performing those procedures, if at all. Additionally, some private payers
do not follow the Medicare guidelines and may reimburse for only a portion of these procedures or not at all. We are unable to
predict what changes will be made in the reimbursement methods used by private or governmental third-party payers. Importantly,
we cannot predict the effects that implementation of the Patient Protection and Affordable Care Act, or the potential repeal and
replacement of that legislation, will have on CMS, commercial insurers, health care providers, and ultimately on our business.
We are
subject to extensive regulation by the FDA which could restrict the sales and marketing of the Quell, SENSUS and DPNCheck devices
and the ADVANCE System as well as other products for which we may seek FDA clearance or approval, and could cause us to incur significant
costs.
We sell medical devices that are subject to
extensive regulation in the United States by the FDA with regard to manufacturing, labeling, sale, promotion, distribution, shipping
and ongoing monitoring and follow-up. Before a new medical device, or a new use of or claim for an existing product, can be marketed
in the United States, it must first be cleared or approved by the FDA. Medical devices may be marketed only for the indications
for which they are approved or cleared. The regulatory review process can be expensive and lengthy. The FDA’s process for
granting 510(k) clearance typically takes approximately three to six months, but it can be significantly longer. The process for
obtaining a pre-market approval, or PMA, is much more costly and onerous. By law, the time period designated for the FDA’s
review of a PMA is 180 days; however, this time is often extended and it is not uncommon for the PMA review process to take three
years or longer from the time the application is filed with the FDA.
The FDA may remove our devices from the market
or enjoin them from commercial distribution if safety or effectiveness problems develop. Further, we may not be able to obtain
additional 510(k) clearances or pre-market approvals for new products or for modifications to, or additional indications for, our
existing products in a timely fashion, or at all. Delays in obtaining future clearances or approvals would adversely affect our
ability to introduce new or enhanced products in a timely manner, which in turn would harm our revenue and future profitability.
We have made modifications to our devices in the past and may make additional modifications in the future that we believe do not
or will not require additional clearances or approvals. If the FDA disagrees, and requires new clearances or approvals for the
modifications, we may be required to recall and to stop marketing the modified devices. If any of these events occurs or if the
FDA takes other enforcement actions, we may not be able to provide our customers with the products they require on a timely basis,
our reputation could be harmed, and we could lose customers and suffer reduced revenues and increased costs.
We also are subject to numerous post-marketing
regulatory requirements, including the FDA’s quality system regulations, which relate to the design, manufacture, packaging,
labeling, storage, installation and servicing of our products, labeling regulations, medical device reporting regulations and correction
and removal reporting regulations. Our failure or the failure by any manufacturer of our products to comply with applicable regulatory
requirements could result in enforcement action by the FDA. FDA enforcement actions relating to post-marketing regulatory requirements
or other issues may include any of the following:
|
|
·
|
warning letters, untitled letters, fines, injunctions, product seizures, consent decrees and civil penalties;
|
|
|
·
|
requiring repair, replacement, refunds, customer notifications or recall of our products;
|
|
|
·
|
imposing operating restrictions, suspension or shutdown of production;
|
|
|
·
|
refusing our requests for 510(k) clearance or PMA approval of new products, new intended uses, or modifications to existing products;
|
|
|
·
|
requesting voluntary rescission of 510(k) clearances or withdrawing PMA approvals that have already been granted; and
|
If any of these events were to occur, they
could harm our reputation, our ability to generate revenues and our profitability.
Also, from time to time, legislation is introduced
into Congress that could significantly change the statutory provisions governing the approval, manufacturing and marketing of medical
devices. FDA regulations and guidance are often revised or reinterpreted by the agency in ways that may significantly affect our
business and our products. It is impossible to predict whether legislative changes will be enacted, or FDA regulations, guidance
or interpretations changed, and what the impact of such changes, if any, may be. The FDA has publicly stated that it is reevaluating
its longstanding 510(k) review program. It is not clear when or if the program will be modified and what effect the modified review
process will have on our ability to bring our product to market.
We depend on several single source manufacturers to produce components
of our products. Any material adverse changes in our relationships with these manufacturers could prevent us from delivering products
to our customers in a timely manner and may adversely impact our future revenues or costs.
We rely on third-party manufacturers to manufacture
components of our Quell, DPNCheck and SENSUS systems, and to fully manufacture devices for the ADVANCE system. In the event that
our manufacturers cease to manufacture sufficient quantities of our products or components in a timely manner and on terms acceptable
to us, we would be forced to locate alternate manufacturers. Additionally, if our manufacturers experience a failure in their production
process, are unable to obtain sufficient quantities of the components necessary to manufacture our products or otherwise fail to
meet our quality requirements, we may be forced to delay the manufacture and sale of our products or locate an alternative manufacturer.
We may be unable to locate suitable alternative manufacturers for our products or components for which the manufacturing process
is relatively specialized, on terms acceptable to us, or at all. We have a manufacturing and supply agreement with Johnson Medtech,
LLC. for the manufacture of the ADVANCE electrodes for nerve conduction testing. Katecho, Inc. manufactures biosensors for use
with our DPNCheck devices and manufactures electrodes for Quell and SENSUS. Sunburst EMS, Inc. and MC Assembly manufacture electronic
boards and other components of our products which we assemble at our Massachusetts facility to produce completed devices. Moreover,
due to the recent commercialization of Quell and the limited amount of our sales to date we do not have long-standing relationships
with our manufacturers, other than Katecho, Inc. and Sunburst EMS, Inc. and may not be able to convince suppliers to continue to
make components available to us unless there is demand for such components from their other customers. As a result, there is a
risk that certain components could be discontinued and no longer available to us.
We have experienced transient inventory shortages
on new products, including Quell, during the initial production ramp-up phase. If any materially adverse changes in our relationships
with the manufacturers of our products occur, our ability to supply our customers will be severely limited until we are able to
engage an alternate manufacturer or, if applicable, resolve any quality issues with our existing manufacturer. This situation could
prevent us from delivering products to our customers in a timely manner, lead to decreased sales or increased costs, or harm our
reputation with our customers.
If our manufacturers are unable to supply us with an adequate
supply of product components as we expand our markets, we could lose customers, our potential future growth could be limited and
our business could be harmed.
In order for us to successfully expand our
business within the United States and internationally, our contract manufacturers must be able to provide us with substantial quantities
of components of our products in compliance with regulatory requirements, in accordance with agreed upon specifications, at acceptable
cost and on a timely basis. Our potential future growth could strain the ability of our manufacturers to deliver products and obtain
materials and components in sufficient quantities. Manufacturers often experience difficulties in scaling up production, including
problems with production yields and quality control and assurance. If we are unable to obtain sufficient quantities of high quality
products to meet customer demand on a timely basis, we could lose customers, our growth may be limited and our business could be
harmed.
If we or our manufacturers fail to comply with the FDA’s
quality system regulation, the manufacturing and distribution of our products could be interrupted, and our product sales and operating
results could suffer.
We and our contract manufacturers are required
to comply with the FDA’s quality system regulation, or QSR, which is a complex regulation that governs the procedures and
documentation of the design, testing, production, control, quality assurance, labeling, packaging, sterilization, storage and shipping
of our devices. The FDA enforces the QSR through periodic inspections. We cannot assure you that our facilities or the facilities
of the manufacturers of our products would pass any future inspection. If our facilities or any of the facilities of the manufacturers
of our products fail an inspection, the manufacturing or distribution of our products could be interrupted and our operations disrupted.
Failure to take adequate and timely corrective action in response to an adverse inspection could result in a suspension or shutdown
of our packaging and labeling operations and the operations of the manufacturers of our products or a recall of our products, or
other administrative or judicial sanctions. If any of these events occurs, we may not be able to provide our customers with the
quantity of products they require on a timely basis, our reputation could be harmed, and we could lose customers and suffer reduced
revenues and increased costs.
Our products may be subject to recalls, even after receiving
FDA clearance or approval, which would harm our reputation, business and financial results.
We are subject to the medical device reporting
regulations, which require us to report to the FDA if our products may have caused or contributed to a death or serious injury,
or have malfunctioned in a way that would likely cause or contribute to a death or serious injury if the malfunction were to occur.
We are also subject to the correction and removal reporting regulations, which require us to report to the FDA any field corrections
and device recalls or removals that we undertake to reduce a risk to health posed by the device or to remedy a violation of the
Federal Food, Drug and Cosmetic Act, or FDCA, caused by the device which may present a risk to health. In addition, the FDA and
similar governmental agencies in other countries have the authority to require the recall of our products if there is a reasonable
probability that the products would cause serious adverse health consequences or death. A government-mandated or voluntary recall
by us could occur as a result of manufacturing defects, labeling deficiencies, packaging defects or other failures to comply with
applicable regulations. Any recall would divert management attention and financial resources and harm our reputation with customers
and could have a material adverse effect on our financial condition and results of operations.
The success of our business depends upon our ability to advance
our pipeline products to commercialization.
We commenced commercialization of Quell in
June 2015. We have additional product candidates and enhancements of our existing products in our R&D pipeline. We expect that
advancing our pipeline products will require significant time and resources. We may not be successful in our commercialization
efforts for any of the product candidates or product enhancements currently in our pipeline and we may not be successful in developing,
acquiring, or in-licensing additional product candidates, to the extent we decide to do so. If we are not successful advancing
new products through our development pipeline, the regulatory process and commercial launch, our business, financial condition,
and results of operations will be adversely affected.
Our ability to achieve profitability depends
in part on maintaining or increasing our gross margins on product sales which we may not be able to achieve.
A number of factors may adversely impact our
gross margins on product sales and services, including:
|
|
·
|
lower than expected manufacturing yields of high cost components leading to increased manufacturing costs;
|
|
|
·
|
low production volume which will result in high levels of overhead cost per unit of production;
|
|
|
·
|
the timing of revenue recognition and revenue deferrals;
|
|
|
·
|
increased material or labor costs;
|
|
|
·
|
increased service or warranty costs or the failure to reduce service or warranty costs;
|
|
|
·
|
increased price competition;
|
|
|
·
|
variation in the margins across products in a particular period; and
|
|
|
·
|
how well we execute on our strategic and operating plans.
|
If we are unable to maintain or increase our
gross margins on product sales, our results of operations could be adversely impacted, we may not achieve profitability and our
stock price could decline.
The patent rights we rely upon to protect the intellectual property
underlying our products may not be adequate, which could enable third parties to use our technology and would harm our ability
to compete in the market.
Our success will depend in part on our ability
to develop or acquire commercially valuable patent rights and to protect these rights adequately. The risks and uncertainties that
we face with respect to our patents and other related rights include the following:
|
|
·
|
the pending patent applications we have filed or to which we have exclusive rights may not result in issued patents or may take longer than we expect to result in issued patents;
|
|
|
·
|
the claims of any patents that are issued may not provide meaningful protection;
|
|
|
·
|
we may not be able to develop additional proprietary technologies that are patentable;
|
|
|
·
|
other parties may challenge patents, patent claims or patent applications licensed or issued to us; and
|
|
|
·
|
other companies may design around technologies we have patented, licensed or developed.
|
Our issued and filed patents for our wearable
therapeutic products are recent. With regard to our legacy neurodiagnostic products, our issued design patents began to expire
in 2015, and our issued utility patents begin to expire in 2017. Although the patent protection for material aspects of these products
covered by the claims of the patents will be lost at that time, we have additional patents and patent applications directed to
other novel inventions that will have patent terms extending beyond 2017. We may not be able to protect our patent rights effectively
in some foreign countries. For a variety of reasons, we may decide not to file for patent protection in the United States or in
particular foreign countries. Our patent rights underlying our products may not be adequate, and our competitors or customers may
design around our proprietary technologies or independently develop similar or alternative technologies or products that are equal
or superior to our technology and products without infringing on any of our patent rights. In addition, the patents licensed or
issued to us may not provide a competitive advantage. If any of these events were to occur, our ability to compete in the market
would be harmed.
Other rights and measures we have taken to protect our intellectual
property may not be adequate, which would harm our ability to compete in the market.
In addition to patents, we rely on a combination
of trade secrets, copyright and trademark laws, confidentiality, nondisclosure and assignment of invention agreements and other
contractual provisions and technical measures to protect our intellectual property rights. We rely on trade secrets to protect
the technology and algorithms we use in our customer data processing and warehousing information system. While we currently require
employees, consultants and other third parties to enter into confidentiality, non-disclosure or assignment of invention agreements
or a combination thereof where appropriate, any of the following could still occur:
|
|
·
|
the agreements may be breached or not enforced in a particular jurisdiction;
|
|
|
·
|
we may have inadequate remedies for any breach;
|
|
|
·
|
trade secrets and other proprietary information could be disclosed to our competitors; or
|
|
|
·
|
others may independently develop substantially equivalent proprietary information and techniques or otherwise gain access to our trade secrets or disclose such technologies.
|
If, for any of the above reasons, our intellectual
property is disclosed or misappropriated, it would harm our ability to protect our rights and our competitive position.
We may need to initiate lawsuits to protect or enforce our patents
and other intellectual property rights, which could be expensive and, if we lose, could cause us to lose some of our intellectual
property rights, which would harm our ability to compete in the market.
We rely on patents to protect a portion of
our intellectual property and our competitive position. Patent law relating to the scope of claims in the technology fields in
which we operate is still evolving and, consequently, patent positions in the medical device industry are generally uncertain.
In order to protect or enforce our patent rights, we may initiate patent litigation against third parties, such as infringement
suits or interference proceedings. Litigation may be necessary to:
|
|
·
|
assert claims of infringement;
|
|
|
·
|
protect our trade secrets or know-how; or
|
|
|
·
|
determine the enforceability, scope and validity of the proprietary rights of others.
|
Any lawsuits that we initiate could be expensive,
take significant time and divert management’s attention from other business concerns. Litigation also puts our patents at
risk of being invalidated or interpreted narrowly and our patent applications at risk of not issuing. Additionally, we may provoke
third parties to assert claims against us. We may not prevail in any lawsuits that we initiate and the damages or other remedies
awarded, if any, may not be commercially valuable. The occurrence of any of these events could harm our business, our ability to
compete in the market or our reputation.
Claims
that our products infringe on the proprietary rights of others could adversely affect our ability to sell our products and increase
our costs.
Substantial litigation over intellectual
property rights exists in the medical device industry. We expect that our products could be increasingly subject to third-party
infringement claims as the number of competitors grows and the functionality of products and technology in different industry segments
overlap. Third parties may currently have, or may eventually be issued, patents on which our products or technologies may infringe.
Any of these third parties might make a claim of infringement against us. Any litigation regardless of its impact would likely
result in the expenditure of significant financial resources and the diversion of management’s time and resources. In addition,
litigation in which we are accused of infringement may cause negative publicity, adversely impact prospective customers, cause
product shipment delays or require us to develop non-infringing technology, make substantial payments to third parties, or enter
into royalty or license agreements, which may not be available on acceptable terms, or at all. If a successful claim of infringement
were made against us and we could not develop non-infringing technology or license the infringed or similar technology on a timely
and cost-effective basis, our revenues may decrease substantially and we could be exposed to significant liability.
We are subject to federal and state laws prohibiting “kickbacks”
and false or fraudulent claims, which, if violated, could subject us to substantial penalties. Additionally, any challenge to or
investigation into our practices under these laws could cause adverse publicity and be costly to respond to, and thus could harm
our business.
A federal law commonly known as the federal
anti-kickback law, and several similar state laws, prohibit the payment of any remuneration that is intended to induce physicians
or others either to refer patients or to acquire or arrange for or recommend the acquisition of health care products or services.
These laws constrain a medical device company’s sales, marketing and other promotional activities by limiting the kinds of
business relationships and financial arrangements, including sales programs we may have with hospitals, physicians or other potential
purchasers of medical devices. Other federal and state laws generally prohibit individuals or entities from knowingly presenting,
or causing to be presented, claims for payment to Medicare, Medicaid or other third-party payers that are false or fraudulent,
or for items or services that were not provided as claimed. From time to time, we may provide coding and billing information as
product support to purchasers of our products. Anti-kickback and false claims laws prescribe civil and criminal penalties for noncompliance,
which can be quite substantial including exclusion from participation in federal health care programs. A number of states have
enacted laws that require pharmaceutical and medical device companies to monitor and report payments, gifts and other remuneration
made to physicians and other health care professionals and health care organizations. Some state statutes, such as the one in Massachusetts,
impose an outright ban on gifts to physicians. These laws are often referred to as “gift ban” or “aggregate spend”
laws and carry substantial fines if they are violated. Similar legislation, known as the Physician Payments Sunshine Act, was enacted
by Congress during 2014. In the event that we are found to have violated these laws or determine to settle a claim that we have
done so, our business may be materially adversely affected as a result of any payments required to be made, restrictions on our
future operations or actions required to be taken, damage to our business reputation or adverse publicity in connection with such
a finding or settlement or other adverse effects relating thereto. Additionally, even an unsuccessful challenge or investigation
into our practices could cause adverse publicity, and be costly to respond to, and thus could harm our business and results of
operations.
If we are found to have violated laws protecting the confidentiality
of patient health information, we could be subject to civil or criminal penalties, which could increase our liabilities, damage
our reputation and harm our business.
There are a number of federal and state laws
protecting the confidentiality of individually identifiable patient health information, including patient records, and restricting
the use and disclosure of that protected information. In particular, the U.S. Department of Health and Human Services promulgated
patient privacy rules under the Health Insurance Portability and Accountability Act of 1996, or HIPAA. These privacy rules protect
medical records and other personal health information by limiting their use and disclosure, giving individuals the right to access,
amend and seek accounting of their own health information and limiting most use and disclosures of health information to the minimum
amount reasonably necessary to accomplish the intended purpose. We do not believe that we are subject to the HIPAA rules. However,
if we are found to be in violation of the privacy rules under HIPAA, we could be subject to civil or criminal penalties, which
could increase our liabilities and harm our reputation or our business.
The use of our products could result in product liability claims
that could be expensive, damage our reputation and harm our business.
Our business exposes us to an inherent risk
of potential product liability claims related to the manufacturing, marketing and sale of medical devices. The medical device industry
historically has been litigious, and we face financial exposure to product liability claims if the use of our products were to
cause or contribute to injury or death. Our products may be susceptible to claims of injury because their use involves the electric
stimulation of a patient’s nerves. Although we maintain product liability insurance for our products and other commercial
insurance, the coverage limits of these policies may not be adequate to cover future claims. As sales and use of our products increase,
we may be unable to maintain sufficient product liability or other commercial insurance on acceptable terms or at reasonable costs,
and this insurance may not provide us with adequate coverage against potential liabilities. A successful claim brought against
us in excess of, or outside of, our insurance coverage could have a material adverse effect on our financial condition and results
of operations. A product liability claim, regardless of its merit or eventual outcome, could result in substantial costs to us,
a substantial diversion of management attention and adverse publicity. A product liability claim could also harm our reputation
and result in a decline in revenues and an increase in expenses.
Our products are complex in design, and defects may not be discovered
prior to shipment to customers, which could result in warranty obligations or product liability or other claims, reducing our revenues
and increasing our costs and liabilities.
We depend upon third parties for the manufacture
of our products or components. Our products, particularly our electrodes, require a significant degree of technical expertise to
produce. If these manufacturers fail to produce our products to specification, or if the manufacturers use defective materials
or workmanship in the manufacturing process, the reliability and performance of our products will be compromised.
If our products contain defects that cannot
be repaired quickly, easily and inexpensively, we may experience:
|
|
·
|
loss of customer orders and delay in order fulfillment;
|
|
|
·
|
damage to our brand reputation;
|
|
|
·
|
increased cost of our warranty program due to product repair or replacement;
|
|
|
·
|
inability to attract new customers;
|
|
|
·
|
diversion of resources from our manufacturing and research and development departments into our service department; and
|
The occurrence of any one or more of the foregoing
could harm our reputation and materially reduce our revenues and increase our costs and liabilities.
If we lose any of our officers or key employees, our management
and technical expertise could be weakened significantly.
Our success largely depends on the skills,
experience, and efforts of our executive officers, including Shai N. Gozani, M.D., Ph.D., our founder, Chairman, President and
Chief Executive Officer, Thomas T. Higgins, our Senior Vice President and Chief Financial Officer; and Francis X. McGillin, our
Senior Vice President and Chief Commercial Officer. We do not maintain key person life insurance policies covering any of our employees.
The loss of any of our executive officers could weaken our management and technical expertise significantly and harm our business.
If we are unable to recruit, hire and retain skilled and experienced
personnel, our ability to manage and expand our business will be harmed, which would impair our future revenues and profitability.
We are a small company with 44 full-time employees
as of December 31, 2016, and our ability to retain our skilled labor force and our success in attracting and hiring new skilled
employees will be a critical factor in determining our future performance. We may not be able to meet our future hiring needs or
retain existing personnel, particularly given the challenges faced by our business. We will face challenges and risks in hiring,
training, managing and retaining engineering and sales and marketing employees. Failure to attract and retain personnel, particularly
technical and sales and marketing personnel would materially harm our ability to compete effectively and grow our business.
Failure to develop or enter into relationships to sell products
other than our existing products or enhance our existing products could have an adverse effect on our business prospects.
Our future business and financial success will
depend, in part, on our ability to effectively market our products, such as Quell and DPNCheck, and enhance these products in response
to customer demand. Developing new products and upgrades to existing and future products imposes burdens on our research and development
department and our management. This process is costly, and we cannot assure you that we will be able to successfully develop new
products or enhance our current products. We also may not be able to enter into relationships with other companies to sell additional
products. In addition, as we develop the market for our products, future competitors may develop desirable product features earlier
than we do which could make our competitors’ products less expensive or more effective than our products and could render
our products obsolete or unmarketable. If our product development efforts are unsuccessful, we will have incurred significant costs
without recognizing the expected benefits and our business prospects may suffer.
If we are unable to develop new products or enhance existing
products, we may be unable to attract or retain customers.
Our success depends on the successful development,
regulatory clearance or approval (if required), introduction and commercialization of new generations of products, treatment systems,
and enhancements to and/or simplification of existing products. Quell and DPNCheck must keep pace with, among other things, the
products of our competitors. We are making significant investments in long-term growth initiatives. Such initiatives require significant
capital commitments, involvement of senior management and other investments on our part, which we may be unable to recover. Our
timeline for the development of new products or enhancements may not be achieved and price and profitability targets may not prove
feasible. Commercialization of new products may prove challenging, and we may be required to invest more time and money than expected
to successfully introduce them. Once introduced, new products may adversely impact orders and sales of our existing products, or
make them less desirable or even obsolete. Compliance with regulations, competitive alternatives, and shifting market preferences
may also impact the successful implementation of new products or enhancements.
Our ability to successfully develop and introduce
new products and product enhancements, and the revenues and costs associated with these efforts, may be affected by our ability
to:
|
|
·
|
properly identify customer needs;
|
|
|
·
|
prove feasibility of new products in a timely manner;
|
|
|
·
|
educate physicians about the use of new products and procedures;
|
|
|
·
|
comply with internal quality assurance systems and processes timely and efficiently;
|
|
|
·
|
limit the timing and cost of obtaining required regulatory approvals or clearances;
|
|
|
·
|
accurately predict and control costs associated with inventory overruns caused by phase-in of new products and phase-out of old products;
|
|
|
·
|
price new products competitively
|
|
|
·
|
manufacture and deliver our products in sufficient volumes on time, and accurately predict and control costs associated with manufacture of the products; and
|
|
|
·
|
meet our product development plan and launch timelines.
|
Even if customers accept new products or product
enhancements, the revenues from these products may not be sufficient to offset the significant costs associated with making them
available to customers.
Failure to successfully develop, obtain regulatory
approval or clearance for, manufacture or introduce new products or to complete these processes in a timely and efficient manner
could result in delays that could affect our ability to attract and retain customers, or could cause customers to delay or cancel
orders, causing our backlog, revenues and operating results to suffer.
We currently compete, and may in the future need to compete,
against other medical device and consumer companies with greater resources, more established distribution channels and other competitive
advantages, and the success of these competitors may harm our ability to generate revenues.
We currently do, and in the future may need
to, compete directly and indirectly with a number of other companies that may have competitive advantages over us. Our diagnostic
devices for nerve testing compete with companies that sell traditional nerve conduction study and electromyography equipment including
Cadwell Laboratories, Inc. and Natus Medical Incorporated. These companies enjoy significant competitive advantages, including:
|
|
·
|
greater resources for product development, sales and marketing;
|
|
|
·
|
more established distribution networks;
|
|
|
·
|
greater name recognition;
|
|
|
·
|
more established relationships with health care professionals, customers and third-party payers; and
|
|
|
·
|
additional lines of products and the ability to offer rebates or bundle products to offer discounts or incentives.
|
As we develop the market for wearable technology
for chronic pain, we will likely be faced with competition from other companies that decide and are able to enter the market, as
well as competition from other forms of treatment for chronic pain. Some or all of our future competitors in the diagnostic nerve
testing market and the consumer market for pain relief may enjoy competitive advantages such as those described above. If we are
unable to compete effectively against existing and future competitors, our sales will decline and our business will be harmed.
Security breaches and other disruptions could compromise our
information and expose us to liability, which could cause our business and reputation to suffer.
In the ordinary course of our business, we
collect and store sensitive data in our data centers, on our networks, including intellectual property, our proprietary business
information, and that of our customers, suppliers and business partners, and personally identifiable information of our employees.
The secure processing, maintenance and transmission of this information is critical to our operations. Despite our security measures,
our information technology and infrastructure may be vulnerable to attacks by hackers or breached due to employee error, malfeasance
or other disruptions. Any such breach could compromise our networks and the information stored there could be accessed, publicly
disclosed, lost or stolen. Any such access, disclosure or other loss of information could result in legal claims or proceedings,
disrupt our operations, damage our reputation, and cause a loss of confidence in our products and services, which could have a
material adverse effect on our business, financial condition, results of operations or cash flows.
If future clinical studies or other articles are published, or
physician associations or other organizations announce positions that are unfavorable to our products, our sales efforts and revenues
may be negatively affected.
Future clinical studies or other articles regarding
our existing products or any competing products may be published that either support a claim, or are perceived to support a claim,
that a competitor’s product is more accurate or effective than our products or that our products are not as accurate or effective
as we claim or previous clinical studies have concluded. Additionally, physician associations or other organizations that may be
viewed as authoritative or have an economic interest in nerve conduction studies and in related electrodiagnostic procedures or
other procedures that may be performed using our products or in neurostimulation therapies using our devices could endorse products
or methods that compete with our products or otherwise announce positions that are unfavorable to our products. Any of these events
may negatively affect our sales efforts and result in decreased revenues.
As we expand into foreign markets, we will be affected by new
business risks that may adversely impact our financial condition or results of operations.
Foreign markets represented approximately 19%
and 19% of our revenues in 2015 and 2014, respectively, and 11% of our revenues for the nine months ended September 30, 2016. We
are working to expand market penetration, particularly in Asia. Any such expansion will subject us to the possibility of new business
risks, including:
|
|
·
|
failure to fulfill foreign
regulatory requirements, if applicable, to market our products;
|
|
|
·
|
availability of, and changes in, reimbursement within prevailing foreign health care payment systems;
|
|
|
·
|
adapting to the differing business practices and laws in foreign countries;
|
|
|
·
|
difficulties in managing foreign relationships and operations, including any relationships that we establish with foreign distributors or sales or marketing agents;
|
|
|
·
|
limited protection for intellectual property rights in some countries;
|
|
|
·
|
difficulty in collecting accounts receivable and longer collection periods;
|
|
|
·
|
costs of enforcing contractual obligations in foreign jurisdictions;
|
|
|
·
|
recessions in economies outside of the United States;
|
|
|
·
|
political instability and unexpected changes in diplomatic and trade relationships;
|
|
|
·
|
currency exchange rate fluctuations; and
|
|
|
·
|
potentially adverse tax consequences.
|
If we are successful in introducing our products
into foreign markets, we will be affected by these additional business risks, which may adversely impact our financial condition
or results of operations. In addition, expansion into foreign markets imposes additional burdens on our executive and administrative
personnel, research and sales departments, and general managerial resources. Our efforts to introduce our products into foreign
markets may not be successful, in which case we may have expended significant resources without realizing the expected benefit.
Our loan and security agreement with a bank, which we refer to
as our credit facility, contains financial and operating restrictions that may limit our access to credit. If we fail to comply
with covenants in the credit facility, we may be required to repay any indebtedness thereunder, which may have an adverse effect
on our liquidity.
Although we have not borrowed any funds under
the credit facility, provisions in the credit facility impose restrictions on our ability to, among other things:
|
|
·
|
incur additional indebtedness;
|
|
|
·
|
replace certain of our executive officers;
|
|
|
·
|
enter into transactions with affiliates;
|
|
|
·
|
pay dividends or make distributions on, or repurchase, our capital stock; and
|
In addition, we are required to meet certain
financial covenants customary with this type of credit facility, including maintaining a minimum specified tangible net worth.
The credit facility also contains other customary covenants, which we may not be able to comply with in the future. Our failure
to comply with these covenants may result in the declaration of an event of default and could cause us to be unable to borrow under
the credit facility. In addition to preventing additional borrowings under the credit facility, an event of default, if not cured
or waived, may result in the acceleration of the maturity of indebtedness outstanding under the credit facility at the time of
the default, which would require us to pay all amounts outstanding. If an event of default occurs, we may not be able to cure it
within any applicable cure period, if at all. If the maturity of our indebtedness is accelerated, we may not have sufficient funds
available for repayment or we may not have the ability to borrow or obtain sufficient funds to replace the accelerated indebtedness
on terms acceptable to us, or at all. We have not borrowed any funds under this agreement; however, as of September 30, 2016, $499,481
of the amounts available under the agreement are restricted to support letters of credit issued in favor of our landlords and a
materials component supplier.
Risks Relating to Owning Our Securities
If we sell additional shares, our stock price may decline as
a result of the dilution which will occur to existing stockholders.
Until we are profitable, we will need significant
additional funds to develop our business and sustain our operations. We sold shares of convertible preferred stock and warrants
to purchase shares of our common stock in January 2017, June 2016 and December 2015, and any additional sales of shares of our
common stock or other securities exercisable into our common stock are likely to have a dilutive effect on some or all of our then
existing stockholders. Resales of newly issued shares in the open market could also have the effect of lowering our stock price,
thereby increasing the number of shares we may need to issue in the future to raise the same dollar amount and consequently further
diluting our outstanding shares.
The perceived risk associated with the possible
sale of a large number of shares could cause some of our stockholders to sell their stock, thus causing the price of our stock
to decline. In addition, actual or anticipated downward pressure on our stock price due to actual or anticipated issuances or sales
of stock could cause some institutions or individuals to engage in short sales of our common stock, which may itself cause the
price of our stock to decline.
If our stock price declines, we may be unable
to raise additional capital. A sustained inability to raise capital could force us to go out of business. Significant declines
in the price of our common stock could also impair our ability to attract and retain qualified employees, reduce the liquidity
of our common stock and result in the delisting of our common stock from The NASDAQ Stock Market LLC, or NASDAQ.
The trading price of our common stock has been volatile and is
likely to be volatile in the future.
The trading price of our common stock has been
highly volatile. For the five year period ended September 30, 2016, our stock price has fluctuated from a low of $1.35 to a high
of $49.20, as adjusted for stock splits during that time. The market price for our common stock will be affected by a number of
factors, including:
|
|
·
|
the denial or delay of regulatory clearances or approvals for our products under development or receipt of regulatory approval of competing products;
|
|
|
·
|
our ability to accomplish clinical, regulatory and other product development and commercialization milestones and to do so in accordance with our timing estimates;
|
|
|
·
|
changes in policies affecting third-party coverage and reimbursement in the United States and other countries;
|
|
|
·
|
changes in government regulations and standards affecting the medical device industry and our products;
|
|
|
·
|
ability of our products to achieve market success;
|
|
|
·
|
the performance of third-party contract manufacturers and component suppliers;
|
|
|
·
|
actual or anticipated variations in our results of operations or those of our competitors;
|
|
|
·
|
announcements of new products, technological innovations or product advancements by us or our competitors;
|
|
|
·
|
developments with respect to patents and other intellectual property rights;
|
|
|
·
|
sales of common stock or other securities by us or our stockholders in the future;
|
|
|
·
|
additions or departures of key scientific or management personnel;
|
|
|
·
|
disputes or other developments relating to proprietary rights, including patents, litigation matters and our ability to obtain patent protection for our technologies;
|
|
|
·
|
trading volume of our common stock;
|
|
|
·
|
changes in earnings estimates or recommendations by securities analysts, failure to obtain or maintain analyst coverage of our common stock or our failure to achieve analyst earnings estimates;
|
|
|
·
|
public statements by analysts or clinicians regarding their perceptions of our clinical results or the effectiveness of our products;
|
|
|
·
|
decreases in market valuations of medical device companies; and
|
|
|
·
|
general market conditions and other factors unrelated to our operating performance or the operating performance of our competitors.
|
The stock prices of many companies in the medical
device industry have experienced wide fluctuations that have often been unrelated to the operating performance of these companies.
Periods of volatility in the market price of a company’s securities can result in securities class action litigation against
a company. If class action litigation is initiated against us, we may incur substantial costs and our management’s attention
may be diverted from our operations, which could significantly harm our business.
There can be no assurance that we will be able to comply with
the continued listing standards of The NASDAQ Capital Market.
We cannot assure you that we will be able to
comply with the standards that we are required to meet in order to maintain a listing of our common stock on The NASDAQ Capital
Market. In 2015, we received two notices from the Listing Qualifications Department of the NASDAQ Stock Market, and we regained
compliance with respect to both of these notices before the end of fiscal 2015. If we fail to continue to meet all applicable NASDAQ
Capital Market requirements in the future and NASDAQ determines to delist our common stock, the delisting could substantially decrease
trading in our common stock and adversely affect the market liquidity of our common stock; adversely affect our ability to obtain
financing on acceptable terms, if at all, for the continuation of our operations; and harm our business. Additionally, the market
price of our common stock may decline further and stockholders may lose some or all of their investment.
If we fail to maintain compliance with any
NASDAQ listing requirements, we could be delisted and our stock would be considered a penny stock under regulations of the Securities
and Exchange Commission, or SEC, and would therefore be subject to rules that impose additional sales practice requirements on
broker-dealers who sell our securities. The additional burdens imposed upon broker-dealers by these requirements could discourage
broker-dealers from effecting transactions in our common stock, which could severely limit the market liquidity of our common stock
and your ability to sell our securities in the secondary market.
The low trading volume of our common stock may adversely affect
the price of our shares.
Although our common stock is listed on The
NASDAQ Capital Market, our common stock has experienced low trading volume. The 50 day average trading volume through September
30, 2016 as reported by NASDAQ was approximately 85,000 shares. Limited trading volume may subject our common stock to greater
price volatility and may make it difficult for investors to sell shares at a price that is attractive to them.
Anti-takeover provisions in our organizational documents and
Delaware law, and the shareholder rights plan that we previously adopted in 2007, may discourage or prevent a change of control,
even if an acquisition would be beneficial to our stockholders, which could affect our stock price adversely and prevent attempts
by our stockholders to replace or remove our current management.
Our certificate of incorporation and bylaws
contain provisions that could delay or prevent a change of control of our Company or changes in our Board of Directors that our
stockholders might consider favorable. Some of these provisions:
|
|
·
|
authorize the issuance of preferred stock which can be created and issued by the Board of Directors without prior stockholder approval, with rights senior to those of our common stock;
|
|
|
·
|
provide for a classified Board of Directors, with each director serving a staggered three-year term;
|
|
|
·
|
prohibit our stockholders from filling board vacancies, calling special stockholder meetings, or taking action by written consent;
|
|
|
·
|
provide for the removal of a director only with cause and by the affirmative vote of the holders of 75% or more of the shares then entitled to vote at an election of our directors; and
|
|
|
·
|
require advance written notice of stockholder proposals and director nominations.
|
We have also adopted a shareholder rights plan
that could make it more difficult for a third party to acquire, or could discourage a third party from acquiring, us or a large
block of our common stock. A third party that acquires 15% or more of our common stock could suffer substantial dilution of its
ownership interest under the terms of the shareholder rights plan through the issuance of common stock to all stockholders other
than the acquiring person.
In addition, we are subject to the provisions
of Section 203 of the Delaware General Corporation Law, which may prohibit certain business combinations with stockholders owning
15% or more of our outstanding voting stock. These and other provisions in our certificate of incorporation, bylaws and Delaware
law could make it more difficult for stockholders or potential acquirers to obtain control of our Board of Directors or initiate
actions that are opposed by our then-current Board of Directors, including a merger, tender offer, or proxy contest involving our
Company. Any delay or prevention of a change of control transaction or changes in our Board of Directors could cause the market
price of our common stock to decline.
We do not intend to pay cash dividends.
We have never declared or paid cash dividends
on our capital stock. We currently intend to retain all available funds and any future earnings for use in the operation and expansion
of our business and do not anticipate paying any cash dividends in the foreseeable future. In addition, the terms of our credit
facility precludes us from paying any dividends. As a result, capital appreciation, if any, of our common stock will be our stockholders’
sole source of potential gain for the foreseeable future.
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus contains forward-looking statements.
Forward-looking statements relate to future events or our future financial performance. We generally identify forward-looking statements
by terminology such as “may,” “will,” “should,” “expects,” “plans,”
“anticipates,” “could,” “intends,” “target,” “projects,” “contemplates,”
“believes,” “estimates,” “predicts,” “potential,” “aim,” “strategy,”
“goal” or “continue” or the negative of these terms or other similar words, although not all forward-looking
statements contain these words. Forward-looking statements include, but are not limited to, statements regarding our or our management’s
expectations, hopes, beliefs, intentions or strategies regarding the future, such as our estimates regarding anticipated operating
losses, future revenues and projected expenses; our expectations for commercialization of our Quell products; our liquidity and
our expectations regarding our needs for and ability to raise additional capital; our ability to manage our expenses effectively
and raise the funds needed to continue our business; our belief that there are unmet needs in the diagnosis and treatment of diabetic
neuropathy and our expectations surrounding Quell and DPNCheck; our plans to develop and commercialize our products; our ability
to meet our proposed timelines for the commercial availability of our products; the success and timing of our studies; our ability
to obtain and maintain regulatory approval of our existing products and any future products we may develop; regulatory and legislative
developments in the United States and foreign countries; the performance of our third-party manufacturers; our ability to obtain
and maintain intellectual property protection for our products; the successful development of our sales and marketing capabilities;
the size and growth of the potential markets for our products and our ability to serve those markets; the rate and degree of market
acceptance of any future products; our reliance on key scientific management or personnel; the payment and reimbursement methods
used by private or governmental third-party payers; and other factors discussed elsewhere in this prospectus or any document incorporated
by reference herein or therein. The forward-looking statements contained in this prospectus are based on our current expectations
and beliefs concerning future developments and their potential effects on us. There can be no assurance that future developments
affecting us will be those that we have anticipated. These forward-looking statements involve a number of risks, uncertainties
(some of which are beyond our control) or other assumptions that may cause actual results or performance to be materially different
from those expressed or implied by these forward-looking statements. These risks and uncertainties include, but are not limited
to, those factors described in the section titled “Risk Factors.” Should one or more of these risks or uncertainties
materialize, or should any of our assumptions prove incorrect, actual results may vary from those projected in these forward-looking
statements. We undertake no obligation to update or revise any forward-looking statements, whether as a result of new information,
future events or otherwise, except as may be required under applicable securities laws. “Risk Factors” and “Prospectus
Summary,” as well as other sections in this prospectus or incorporated by reference into this prospectus, discuss some of
the factors that could contribute to these differences.
The forward-looking statements made in this
prospectus relate only to events as of the date on which the statements are made. We undertake no obligation to update any forward-looking
statement to reflect events or circumstances after the date on which the statement is made or to reflect the occurrence of unanticipated
events.
This prospectus also contains market data related
to our business and industry. These market data include projections that are based on a number of assumptions. While we believe
these assumptions to be reasonable and sound as of the date of this prospectus, if these assumptions turn out to be incorrect,
actual results may differ from the projections based on these assumptions. As a result, our markets may not grow at the rates projected
by these data, or at all. The failure of these markets to grow at these projected rates may have a material adverse effect on our
business, results of operations, financial condition and the market price of our common stock.
USE OF PROCEEDS
We will not receive any of the proceeds from
the sale of securities by the selling security holders pursuant to this prospectus. We may receive up to approximately $7,525,000
in aggregate gross proceeds from cash exercises of the warrants, based on the per share exercise price of the warrants. Any proceeds
we receive from the exercise of the warrants will be used for working capital and general corporate purposes.
SELLING
SECURITY HOLDERS
The shares of common stock being offered by
the selling security holders are those issuable to the selling security holders upon conversion of the Series E Preferred Stock
and those issuable upon the exercise of the Warrants. For additional information regarding the issuance of these securities, see
“Prospectus Summary —Offering of Preferred Stock and Warrants” above. We are registering the shares of common
stock in order to permit the selling security holders to offer the shares for resale from time to time. Except for the ownership
of the Series E Preferred Stock and the Warrants and the transactions contemplated pursuant to the Purchase Agreement and the Engagement
Letter, and except as otherwise disclosed below under “Relationships with Selling Security Holders,” the selling security
holders have not had any material relationship with us within the past three years.
The table below lists the selling
security holders and other information regarding the beneficial ownership (as determined under Section 13(d) of the
Securities Exchange Act of 1934, as amended, and the rules and regulations thereunder) of the shares of common stock held by
each of the selling security holders. The second column lists the percentage of shares of common stock beneficially owned by
the selling security holders, based on their respective ownership of shares of common stock, as of January 13, 2017, assuming
conversion of the Preferred Stock and exercise of the Warrants held by each such selling security holder on that date but
taking account of any limitations on exercise set forth therein. The percentage of shares beneficially owned prior to the
offering is based on 7,514,901 shares of our common stock outstanding as of January 13, 2017, which does not include, unless
specifically held by a selling security holder, (i) 5,714,286 shares of common stock issuable upon conversion, at the option
of the holder, of Series E Preferred Stock outstanding as of January 13, 2017, (ii) 8,710,554 shares of common stock issuable
upon the conversion, at the option of the holder, of 15,722.55 shares of Series D convertible preferred stock outstanding as
of January 13, 2017; (iii) 12,375 shares of common stock issuable upon the conversion, at the option of the holder, of 500
shares of Series B convertible preferred stock outstanding as of January 13, 2017; (iv) 34,349,832 shares of common stock
issuable upon the exercise of warrants outstanding as of January 13, 2017, at a weighted average exercise price of $1.81 per
share; (v) 782,364 shares of common stock issuable upon the exercise of options outstanding as of January 13, 2017, at a
weighted average exercise price of $4.57 per share; (vi) 26,420 shares of common stock available for future issuance under
our 2004 Stock Option and Incentive Plan as of January 13, 2017; (vii) 50,000 shares of common stock available for future
issuance under our 2009 Non-qualified Inducement Stock Plan as of January 13, 2017; and (viii) 93,124 shares of common stock
available for future issuance under our 2010 Employee Stock Purchase Plan as of January 13, 2017. The number of shares in the
column “Maximum Number of Shares of Common Stock to be Sold Pursuant to this Prospectus” represents all of the
shares that the selling security holder may offer under this prospectus and does not take into account any limitations
on exercise of the warrants set forth therein.
This prospectus covers the resale of
20,750,000 shares of common stock, of which 10,000,000 shares are issuable upon the conversion of shares of Series E
Preferred Stock and 10,750,000 shares are issuable upon the exercise of the Warrants. The Warrants consist of (i) 6,142,857
warrants, which are immediately exercisable six months after their initial date of issuance, or July 4, 2017 and expire five
years after their date of issuance and have an initial exercise price of $0.92 per share, which will be reset to $0.70 per
share upon receipt of stockholder approval of the Second Closing and (ii) 4,607,143 warrants issuable upon receipt of
stockholder approval of the Second Closing and within five (5) business days of the effectiveness of the Registration
Statement of which this prospectus forms a part, which will expire five years after the date of issuance. See
“Prospectus Summary —Offering of Preferred Stock and Warrants” above for a complete description of the
warrants.
The Series E Preferred Stock and the Warrants
contain limitations that prevent the holder of any Series E Preferred Stock or Warrants from acquiring shares upon conversion of
the Series E Preferred Stock, or exercise of a Warrant, that would result in the number of shares beneficially owned by it and
its affiliates exceeding 4.99% of the total number of shares of our common stock then issued and outstanding, provided, however,
that upon prior notice to us, the holder may increase this beneficial ownership limitation to up to 9.99%. The number of shares
in the second column reflects these limitations. The selling security holders may sell all, some or none of their shares in this
offering. See “Plan of Distribution.”
Name of Selling
Security Holder
|
|
Number of Shares
of
Common Stock
Beneficially
Owned
Prior to Offering
|
|
|
% of Shares
of Common
Stock
Beneficially
Owned
Prior
to Offering
|
|
|
Maximum Number
of Shares of
Common Stock
to be Sold
Pursuant to this
Prospectus
|
|
|
Number of
Shares
of
Common Stock
Owned
After Offering
|
|
|
% of Shares
of Common
Stock Owned
After
Offering
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Sabby Healthcare Master Fund, Ltd. (1)
|
|
|
38,006,111
|
(2)
|
|
|
9.99
|
%(3)
|
|
|
13,900,000
|
(4)
|
|
|
24,106,111
|
(6)
|
|
|
9.99
|
%(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Sabby Volatility Warrant Master Fund, Ltd. (1)
|
|
|
17,531,440
|
(2)
|
|
|
9.99
|
%(3)
|
|
|
6,100,000
|
(5)
|
|
|
11,431,440
|
(6)
|
|
|
9.99
|
%(6)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
H.C. Wainwright & Co., LLC
|
|
|
486,135
|
(2)
|
|
|
4.99
|
%
|
|
|
225,000
|
(7)
|
|
|
261,135
|
(12)
|
|
|
3.36
|
%(12)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Noam Rubinstein
|
|
|
507,344
|
(2)
|
|
|
4.99
|
%
|
|
|
236,250
|
(8)
|
|
|
271,094
|
(12)
|
|
|
3.48
|
%(12)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Michael Vasinkevich
|
|
|
555,663
|
(2)
|
|
|
4.99
|
%
|
|
|
258,750
|
(9)
|
|
|
296,913
|
(12)
|
|
|
3.80
|
%(12)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Mark Viklund
|
|
|
45,369
|
(2)
|
|
|
*
|
|
|
|
22,500
|
(10)
|
|
|
22,869
|
(12)
|
|
|
*
|
(12)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Charles Worthman
|
|
|
16,105
|
(2)
|
|
|
*
|
|
|
|
7,500
|
(11)
|
|
|
8,605
|
(12)
|
|
|
*
|
(12)
|
* Less than 1%.
|
(1)
|
This shareholder has indicated that Hal Mintz has voting and investment power over the shares held by it. This shareholder has indicated that Sabby Management, LLC serves as its investment manager, that Hal Mintz is the manager of Sabby Management, LLC, and that each of Sabby Management, LLC and Hal Mintz disclaim beneficial ownership over these shares except to the extent of any pecuniary interest therein.
|
|
|
|
|
(2)
|
The number of shares shown in this column reflects the aggregate number of shares of common stock beneficially owned (through the ownership of Series E Preferred Stock that is convertible into common stock and warrants to purchase common stock, as applicable) by Sabby Healthcare Master Fund, Ltd., Sabby Volatility Master Fund, Ltd., H.C. Wainwright & Co., LLC, Noam Rubinstein, Michael Vasinkevich, Mark Viklund, and Charles Worthman prior to the offering. All shares of Series E Preferred Stock and warrants exercisable for common stock are subject to beneficial ownership limitations and related warrant exercise restrictions so that the reporting persons cannot own more than 4.99%. Sabby Healthcare Master Fund, Ltd. and Sabby Volatility Master Fund, Ltd. also own securities that are subject to beneficial ownership limitations so that the reporting persons cannot own more than 9.99%.
|
|
(3)
|
Represents the aggregate combined percentage of shares beneficially owned by Sabby Healthcare Master Fund, Ltd. and Sabby Volatility Warrant Master Fund, Ltd. The exercise of certain warrants held by these entities are subject to a 9.99% ownership blocker.
|
|
|
|
|
(4)
|
This Registration Statement registers the resale by Sabby Healthcare Master Fund, Ltd. of 6,950,000 shares of common stock issuable upon the conversion of shares of Series E Preferred Stock and 6,950,000 shares of common stock issuable upon exercise of the Warrants, each as issued to Sabby Healthcare Master Fund, Ltd. in the offering, without regard for any limitations on conversion` set forth in the Series E Preferred Stock or any limitations on exercise set forth in the Warrants.
|
|
|
|
|
(5)
|
This Registration Statement registers the resale by Sabby Volatility Warrant Master Fund, Ltd. of 3,050,000 shares of common stock issuable upon the conversion of Series E Preferred Stock and 3,050,000 shares of common stock issuable upon exercise of the Warrants, each as issued to Sabby Volatility Warrant Master Fund, Ltd. in the offering, without regard for any limitations on conversion set forth in the Series E Preferred Stock or any limitations on exercise set forth in the Warrants.
|
|
|
|
|
(6)
|
The number of shares shown in this column reflect the aggregate number of shares of common stock beneficially owned (through the ownership of warrants to purchase common stock) by Sabby Healthcare Master Fund, Ltd. and Sabby Volatility Master Fund, Ltd. after the offering, assuming that all shares registered for resale by this Registration Statement are sold in the offering. The aggregate beneficial ownership of Sabby Healthcare Master Fund, Ltd. and Sabby Volatility Master Fund, Ltd. is subject to a 9.99% ownership blocker.
|
|
|
|
|
(7)
|
This Registration Statement registers the resale by H.C. Wainwright & Co., LLC of 225,000 shares of common stock issuable upon exercise of outstanding Warrants, as issued to H.C. Wainwright & Co., LLC.
|
|
|
|
|
(8)
|
This Registration Statement registers the resale by Noam Rubinstein of 236,250 shares of common stock issuable upon exercise of outstanding Warrants, as issued to Noam Rubinstein.
|
|
|
|
|
(9)
|
This Registration Statement registers the resale by Michael Vasinkevich of 258,750 shares of common stock issuable upon exercise of outstanding Warrants, as issued to Michael Vasinkevich.
|
|
|
|
|
(10)
|
This Registration Statement registers the resale by Mark Viklund of 22,500 shares of common stock issuable upon exercise of outstanding Warrants, as issued to Mark Viklund.
|
|
|
|
|
(11)
|
This Registration Statement registers the resale by Charles Worthman of 7,500 shares of common stock issuable upon exercise of outstanding Warrants, as issued to Charles Worthman.
|
|
|
|
|
(12)
|
The number of shares shown in this column assumes that all of the shares of common stock issuable upon the exercise of Warrants are sold in the offering.
|
Relationships with Selling Security Holders
Except as otherwise set forth below, we have not engaged in any
transactions with the selling security holders since January 1, 2014.
2017 Private Placement of Preferred Stock and Warrants; Resetting
of Exercise Price of Outstanding Warrants
On January 5, 2017, we completed the Initial
Closing of an offering of our securities resulting in approximately $4.0 million in proceeds in connection with the issuance to
Sabby Management, LLC and its affiliates, or Sabby, of (i) 4,000 shares of Series E Preferred Stock at a price of $1,000 per share,
and (ii) Warrants to purchase 5,714,286 shares of our common stock. We also extended the termination date of warrants to purchase
5,411,764 shares of common stock held by Sabby from January 15, 2017 to June 28, 2022 and reset the exercise prices of warrants
to purchase an aggregate of 11,685,732 shares of our common stock held by Sabby to $0.70 per share and extended the termination
dates of those warrants by an additional six months and one day. In connection with the Initial Closing we issued warrants to purchase
428,571 shares of our common stock to H.C. Wainwright & Co., LLC and certain related persons pursuant to the terms of the Engagement
Letter. We are using the proceeds from this offering for commercialization of Quell in the United States and for general working
capital purposes. See “Prospectus Summary — Offering of Preferred Stock and Warrants” for a complete description
of the offering.
2016 Private Placement of Preferred Stock and Warrants; Repurchase
of Series C Convertible Preferred Stock
In June 2016, we completed an offering of securities
resulting in approximately $21.3 million in proceeds in connection with the issuance to investors of (i) 21,300 shares of Series
D Preferred Stock at a price of $1,000 per share, and (ii) warrants to purchase up to 11,800,554 shares of our common stock.
We used $13.8 million of the proceeds from this offering to redeem all of our outstanding Series C Preferred Stock held by Sabby,
and used the remainder for general working capital purposes.
2015 Private Placement of Preferred Stock and Warrants; Repurchase
of Series B Convertible Preferred Stock
In December 2015, we completed an offering of
securities resulting in approximately $13.8 million in proceeds in connection with the issuance to the selling security holders
of (i) 13,800 shares of Series C Preferred Stock at a price of $1,000 per share, and (ii) warrants to purchase up to 10,823,528
shares of our common stock. We used $6.3 million of the proceeds from this offering to redeem our outstanding Series B convertible
preferred stock, and used the remainder for general working capital purposes. In June 2016, we repurchased all outstanding shares
of Series C Preferred Stock.
Public Offering of Preferred Stock and Warrants; Repurchase
of Series A-4 Convertible Preferred Stock and Forfeiture of Warrants
In May 2015, we completed an underwritten public
offering, or the 2015 Offering, of (i) 147,000 shares of Series B convertible preferred stock at a price of $100 per share, which
is the stated value, and (ii) five year warrants to purchase up to 3,638,250 shares of common stock with an exercise price of $5.00
per share. As part of the 2015 Offering, Sabby agreed to purchase 122,000 units at the public offering price of $100 per unit.
Simultaneous with the closing of the 2015 Offering, we repurchased from Sabby the then outstanding 3,206.357 shares of the Series
A-4 convertible preferred stock for an aggregate purchase price of $3.2 million, which we refer to as the Repurchase. Additionally,
as part of the Repurchase, Sabby agreed to forfeit warrants to purchase 392,936 shares of our common stock that were issued in
connection with the original issuance of the Series A-4 convertible preferred stock, which warrants had an exercise price of $8.16.
In December 2015, we repurchased 63,000 shares of Series B convertible preferred stock. Following this repurchase, as of December
31, 2015, there were 7,146 shares of Series B convertible preferred stock outstanding.
Private Offerings of Preferred Stock and Warrants
During June 2014 we entered into a securities
purchase agreement with Sabby, or the 2014 Offering, which provided for the issuance of (i) 166,150 shares of common stock at a
price of $8.16 per share, (ii) 2,621.859 shares of Series A-3 convertible preferred stock at a price of $1,000 per share, (iii)
4,022.357 shares of Series A-4 convertible preferred stock at a price of $1,000 per share, and (iv) five year warrants to purchase
up to 980,393 shares of common stock with an exercise price of $8.16 per share. The 2014 Offering resulted in approximately $8.0
million in gross proceeds, before deducting expenses.
In these private equity offerings, Each share
of preferred stock has or had a stated value of $1,000 and is convertible at the option of the holder into the number of shares
of common stock determined by dividing the stated value by the conversion price which is subject to adjustment as provided in each
Certificate of Designation for the preferred stock. These preferred stock have no dividend rights, liquidation preference or other
preferences over common stock and has no voting rights except as provided in each Certificate of Designation for each class of
preferred stock and as required by law.
All of the shares of Series A-3 convertible
preferred stock and Series A-4 convertible preferred stock have either been converted into shares of common stock or repurchased
by us and retired.
PLAN OF DISTRIBUTION
Each Selling Stockholder of the securities and
any of their pledgees, assignees and successors-in-interest may, from time to time, sell any or all of their securities covered
hereby on the Nasdaq Stock Market or any other stock exchange, market or trading facility on which the securities are traded or
in private transactions. These sales may be at fixed or negotiated prices. A Selling Stockholder may use any one or more of the
following methods when selling securities:
|
|
·
|
ordinary brokerage transactions
and transactions in which the broker-dealer solicits purchasers;
|
|
|
·
|
block trades in which the broker-dealer will attempt to sell the securities as agent but may position and resell a portion of the block as principal to facilitate the transaction;
|
|
|
·
|
purchases by a broker-dealer as principal and resale by the broker-dealer for its account;
|
|
|
·
|
an exchange distribution in accordance with the rules of the applicable exchange;
|
|
|
·
|
privately negotiated transactions;
|
|
|
·
|
settlement of short sales;
|
|
|
·
|
in transactions through broker-dealers that agree with the Selling Stockholders to sell a specified number of such securities at a stipulated price per security;
|
|
|
·
|
through the writing or settlement of options or other hedging transactions, whether through an options exchange or otherwise;
|
|
|
·
|
a combination of any such methods of sale; or
|
|
|
·
|
any other method permitted pursuant to applicable law.
|
The Selling Stockholders may also sell securities
under Rule 144 under the Securities Act, if available, rather than under this prospectus.
Broker-dealers engaged by the Selling Stockholders
may arrange for other brokers-dealers to participate in sales. Broker-dealers may receive commissions or discounts from the Selling
Stockholders (or, if any broker-dealer acts as agent for the purchaser of securities, from the purchaser) in amounts to be negotiated,
but, except as set forth in a supplement to this prospectus, in the case of an agency transaction not in excess of a customary
brokerage commission in compliance with FINRA Rule 2440; and in the case of a principal transaction a markup or markdown in compliance
with FINRA IM-2440.
In connection with the sale of the securities
or interests therein, the Selling Stockholders may enter into hedging transactions with broker-dealers or other financial institutions,
which may in turn engage in short sales of the securities in the course of hedging the positions they assume. The Selling Stockholders
may also sell securities short and deliver these securities to close out their short positions, or loan or pledge the securities
to broker-dealers that in turn may sell these securities. The Selling Stockholders may also enter into option or other transactions
with broker-dealers or other financial institutions or create one or more derivative securities which require the delivery to such
broker-dealer or other financial institution of securities offered by this prospectus, which securities such broker-dealer or other
financial institution may resell pursuant to this prospectus (as supplemented or amended to reflect such transaction).
The Selling Stockholders and any broker-dealers
or agents that are involved in selling the securities may be deemed to be “underwriters” within the meaning of the
Securities Act in connection with such sales. In such event, any commissions received by such broker-dealers or agents and any
profit on the resale of the securities purchased by them may be deemed to be underwriting commissions or discounts under the Securities
Act. Each Selling Stockholder has informed us that it does not have any written or oral agreement or understanding, directly or
indirectly, with any person to distribute the securities.
We are required to pay certain fees and expenses
incurred by us incident to the registration of the securities. We have agreed to indemnify the Selling Stockholders against certain
losses, claims, damages and liabilities, including liabilities under the Securities Act.
Because Selling Stockholders may be deemed
to be “underwriters” within the meaning of the Securities Act, they will be subject to the prospectus delivery requirements
of the Securities Act including Rule 172 thereunder. In addition, any securities covered by this prospectus which qualify for sale
pursuant to Rule 144 under the Securities Act may be sold under Rule 144 rather than under this prospectus. The Selling Stockholders
have advised us that there is no underwriter or coordinating broker acting in connection with the proposed sale of the resale securities
by the Selling Stockholders.
We agreed to keep this prospectus effective
until the earlier of (i) the date on which the securities may be resold by the Selling Stockholders without registration and
without regard to any volume or manner-of-sale limitations by reason of Rule 144, without the requirement for us to be in compliance
with the current public information under Rule 144 under the Securities Act or any other rule of similar effect or (ii) all
of the securities have been sold pursuant to this prospectus or Rule 144 under the Securities Act or any other rule of similar
effect. The resale securities will be sold only through registered or licensed brokers or dealers if required under applicable
state securities laws. In addition, in certain states, the resale securities covered hereby may not be sold unless they have been
registered or qualified for sale in the applicable state or an exemption from the registration or qualification requirement is
available and is complied with.
Under applicable rules and regulations under
the Exchange Act, any person engaged in the distribution of the resale securities may not simultaneously engage in market making
activities with respect to the common stock for the applicable restricted period, as defined in Regulation M, prior to the commencement
of the distribution. In addition, the Selling Stockholders will be subject to applicable provisions of the Exchange Act and the
rules and regulations thereunder, including Regulation M, which may limit the timing of purchases and sales of the common stock
by the Selling Stockholders or any other person. We will make copies of this prospectus available to the Selling Stockholders and
have informed them of the need to deliver a copy of this prospectus to each purchaser at or prior to the time of the sale (including
by compliance with Rule 172 under the Securities Act).
LEGAL MATTERS
The validity of the securities we are offering
will be passed upon for us by Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C., Boston, Massachusetts.
EXPERTS
The financial statements incorporated in this
prospectus by reference to the Annual Report on Form 10-K for the year ended December 31, 2015 have been so incorporated in reliance
on the report (which contains an explanatory paragraph relating to the Company’s ability to continue as a going concern as
described in Note 1 to the financial statements) of PricewaterhouseCoopers LLP, an independent registered public accounting firm,
given on the authority of said firm as experts in auditing and accounting.
WHERE YOU CAN FIND MORE INFORMATION
We are a public company and file annual, quarterly
and current reports, proxy statements and other information with the SEC. You may read and copy any document we file at the SEC’s
Public Reference Room at Station Place, 100 F Street, N.E., Washington, D.C. 20549. You can request copies of these documents by
writing to the SEC and paying a fee for the copying cost. Please call the SEC at 1-800-SEC-0330 for more information about the
operation of the Public Reference Room. Our SEC filings are also available to the public at the SEC’s web site at http://www.sec.gov,
and on our web site at http://www.neurometrix.com. The information contained on our web site is not included or incorporated by
reference into this prospectus. In addition, our common stock is listed for trading on The NASDAQ Capital Market under the symbol
“NURO.’’ You can read and copy reports and other information concerning us at the offices of the Financial Industry
Reporting Authority located at 1735 K Street, N.W., Washington, D.C. 20006.
This prospectus is only part of a Registration
Statement on Form S-3 that we have filed with the SEC under the Securities Act, and therefore omits certain information contained
in the Registration Statement. We have also filed exhibits and schedules with the Registration Statement that are excluded from
this prospectus, and you should refer to the applicable exhibit or schedule for a complete description of any statement referring
to any contract or other document. You may:
|
|
·
|
inspect a copy of the Registration Statement, including the exhibits and schedules, without charge at the Public Reference Room,
|
|
|
·
|
obtain a copy from the SEC upon payment of the fees prescribed by the SEC, or
|
|
|
·
|
obtain a copy from the SEC’s web site or our web site.
|
INCORPORATION OF CERTAIN DOCUMENTS BY REFERENCE
The SEC allows us to “incorporate by
reference” much of the information we file with them, which means that we can disclose important information to you by referring
you to those publicly available documents. The information that we incorporate by reference in this prospectus is considered to
be part of this prospectus. Because we are incorporating by reference future filings with the SEC, this prospectus is continually
updated and those future filings may modify or supersede some of the information included or incorporated in this prospectus. You
should refer to the registration statement, including the exhibits, for further information about us and the securities we may
offer pursuant to this prospectus. Statements in this prospectus regarding the provisions of certain documents filed with, or incorporated
by reference in, the registration statement are not necessarily complete and each statement is qualified in all respects by that
reference. We incorporate by reference into this prospectus the documents listed below and any future filings made by us with the
SEC under Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act (1) after the date of this prospectus and prior to the
time that all of the securities offered by this prospectus are sold or the earlier termination of the offering, and (2) after
the date of the initial registration statement of which this prospectus forms a part and prior to the effectiveness of the registration
statement (except in each case in which the information contained in such documents is “furnished” and not “filed”).
The documents we are incorporating by reference as of their respective dates of filing are:
|
|
·
|
Annual Report on Form 10-K for the year ended December 31, 2015, filed with the SEC on February 12, 2016;
|
|
|
·
|
the portions of our Definitive Proxy Statement on Schedule 14A that are deemed "filed" with the SEC under the Securities Exchange Act of 1934, as amended, filed on March 17, 2016;
|
|
|
·
|
Quarterly Reports on Form 10-Q filed with the SEC on April 21, 2016, July 21, 2016 and October 20, 2016;
|
|
|
·
|
Current Reports on Form 8-K filed with the SEC on May 5, 2016, June 2, 2016 and June 3, 2016, December 8, 2016 and December 29, 2016;
|
|
|
·
|
Description of our common stock contained in our Registration Statement on Form 8-A filed pursuant to Section 12(g) of the Exchange Act, filed with SEC on July 19, 2004; and
|
|
|
·
|
Description of our preferred share purchase rights contained in our Registration Statement on Form 8-A filed pursuant to Section 12(b) of the Exchange Act, filed with the SEC on March 8, 2007 (File No. 000-50856).
|
Except as set forth above, the SEC file number for each
of the documents listed above is 001-33351.
Any statement contained
in this prospectus or in a document incorporated or deemed to be incorporated by reference into this prospectus will be deemed
to be modified or superseded for purposes of this prospectus to the extent that a statement contained in this prospectus or any
other subsequently filed document that is deemed to be incorporated by reference into this prospectus modifies or supersedes the
statement. Any statement so modified or superseded will not be deemed, except as so modified or superseded, to constitute a part
of this prospectus.
You should rely only on information contained
in, or incorporated by reference into, this prospectus and any prospectus supplement. We have not authorized anyone to provide
you with information different from that contained in this prospectus or incorporated by reference in this prospectus. We are not
making offers to sell the securities in any jurisdiction in which such an offer or solicitation is not authorized or in which the
person making such offer or solicitation is not qualified to do so or to anyone to whom it is unlawful to make such offer or solicitation
You may request a copy of the filings listed
above, at no cost, by writing or telephoning us at the following address:
NeuroMetrix, Inc.
1000 Winter Street
Waltham, Massachusetts 02451
(781) 890-9989
Attn: Investor Relations
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
|
|
Item 14.
|
Other Expenses of Issuance
and Distribution
|
The following table sets forth the Company’s estimates (other
than the SEC registration fee) of the expenses in connection with the issuance and distribution of the securities being registered.
|
Item
|
|
|
|
|
|
SEC registration fee
|
|
$
|
1,707.50
|
|
|
Legal fees and expenses
|
|
|
15,000.00
|
|
|
Accounting fees and expenses
|
|
|
10,000.00
|
|
|
Printing fees
|
|
|
2,500.00
|
|
|
Miscellaneous fees and expenses
|
|
|
2,500.00
|
|
|
Total
|
|
$
|
31,707.50
|
|
|
|
Item 15.
|
Indemnification of Directors
and Officers
|
Our restated certificate provides that we shall
indemnify, to the fullest extent authorized by the Delaware General Corporation Law, each person who is involved in any litigation
or other proceeding because such person is or was our director or officer or is or was serving as an officer or director of another
entity at our request, against all expense, loss or liability reasonably incurred or suffered in connection therewith. Our restated
certificate provides that the right to indemnification includes the right to be paid expenses incurred in defending any proceeding
in advance of its final disposition, provided, however, that such advance payment will only be made upon delivery to us of an undertaking,
by or on behalf of the director or officer, to repay all amounts so advanced if it is ultimately determined that such director
is not entitled to indemnification. If we do not pay a proper claim for indemnification in full within 10 days after we receive
a written claim for such indemnification, our restated certificate and our restated by-laws authorize the claimant to bring an
action against us and prescribe what constitutes a defense to such action.
Section 145 of the Delaware General Corporation
Law permits a corporation to indemnify any director or officer of the corporation against expenses (including attorney’s
fees), judgments, fines and amounts paid in settlement actually and reasonably incurred in connection with any action, suit or
proceeding, whether civil, criminal, administrative or investigative (other than an action by or in right of the corporation) brought
by reason of the fact that such person is or was a director or officer of the corporation, if such person acted in good faith and
in a manner that he reasonably believed to be in, or not opposed to, the best interests of the corporation, and, with respect to
any criminal action or proceeding, if he or she had no reason to believe his or her conduct was unlawful. In a derivative action,
(i.e., one brought by or on behalf of the corporation), indemnification may be provided only for expenses actually and reasonably
incurred by any director or officer in connection with the defense or settlement of such an action or suit if such person acted
in good faith and in a manner that he or she reasonably believed to be in, or not opposed to, the best interests of the corporation,
except that no indemnification shall be provided if such person shall have been adjudged to be liable to the corporation, unless
and only to the extent that the court in which the action or suit was brought shall determine that the defendant is fairly and
reasonably entitled to indemnity for such expenses despite such adjudication of liability.
Pursuant to Section 102(b)(7) of the Delaware
General Corporation Law, Article Seventh of our restated certificate eliminates the liability of a director to us or our stockholders
for monetary damages for such a breach of fiduciary duty as a director, except for liabilities arising:
|
|
·
|
from any breach of the
director’s duty of loyalty to us or our stockholders;
|
|
|
·
|
from acts or omissions not in good faith or which involve intentional misconduct or a knowing violation of law;
|
|
|
·
|
under Section 174 of the Delaware General Corporation Law; and
|
|
|
·
|
from any transaction from which the director derived an improper personal benefit.
|
As permitted by Section 145 of the Delaware
General Corporation Law, we carry insurance policies insuring our directors and officers against certain liabilities that they
may incur in their capacity as directors and officers.
Any underwriting agreements that we may enter
into will likely provide for the indemnification of us, our controlling persons, our directors and certain of our officers by the
underwriters against certain liabilities, including liabilities under the Securities Act of 1933, as amended.
The exhibits listed in the accompanying Exhibit
Index are filed or incorporated by reference as part of this Registration Statement.
(a) The undersigned Registrant hereby undertakes:
|
|
(1)
|
To file, during any period in which offers or sales are being made, a post-effective amendment to this Registration Statement:
|
|
|
(i)
|
To include any prospectus required by Section 10(a)(3) of the Securities Act of 1933;
|
|
|
(ii)
|
To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Commission pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than a 20% change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement; and
|
|
|
(iii)
|
To include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information in the registration statement;
|
Provided, however, that paragraphs (a)(1)(i), (a)(1)(ii) and (a)(1)(iii)
of this section do not apply if the registration statement is on Form S–3 (§239.13 of this chapter) or Form F–3
(§239.33 of this chapter) and the information required to be included in a post-effective amendment by those paragraphs is
contained in reports filed with or furnished to the Commission by the registrant pursuant to section 13 or section 15(d) of the
Securities Exchange Act of 1934 that are incorporated by reference in the registration statement, or is contained in a form of
prospectus filed pursuant to Rule 424(b) (§230.424(b) of this chapter) that is part of the registration statement.
|
|
(2)
|
That, for the purpose of determining any liability under the Securities Act of 1933, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
|
|
|
(3)
|
To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering.
|
|
|
(4)
|
That, for the purpose of determining liability under the Securities Act of 1933 to any purchaser:
|
|
|
(i)
|
If the registrant is relying on Rule 430B (§230.430B of this chapter):
|
|
|
(A)
|
Each prospectus filed by the registrant pursuant to Rule 424(b)(3) (§230.424(b)(3) of this chapter) shall be deemed to be part of the registration statement as of the date the filed prospectus was deemed part of and included in the registration statement; and
|
|
|
(B)
|
Each prospectus required to be filed pursuant to Rule 424(b)(2), (b)(5), or (b)(7) (§230.424(b)(2), (b)(5), or (b)(7) of this chapter) as part of a registration statement in reliance on Rule 430B relating to an offering made pursuant to Rule 415(a)(1)(i), (vii), or (x) (§230.415(a)(1)(i), (vii), or (x) of this chapter) for the purpose of providing the information required by section 10(a) of the Securities Act of 1933 shall be deemed to be part of and included in the registration statement as of the earlier of the date such form of prospectus is first used after effectiveness or the date of the first contract of sale of securities in the offering described in the prospectus. As provided in Rule 430B, for liability purposes of the issuer and any person that is at that date an underwriter, such date shall be deemed to be a new effective date of the registration statement relating to the securities in the registration statement to which that prospectus relates, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such effective date, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such effective date; or
|
|
|
(ii)
|
If the registrant is subject to Rule 430C (§230.430C of this chapter), each prospectus filed pursuant to Rule 424(b) as part of a registration statement relating to an offering, other than registration statements relying on Rule 430B or other than prospectuses filed in reliance on Rule 430A (§230.430A of this chapter), shall be deemed to be part of and included in the registration statement as of the date it is first used after effectiveness. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such date of first use.
|
|
|
(5)
|
That, for the purpose of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution of the securities:
|
The undersigned registrant undertakes that in a primary
offering of securities of the undersigned registrant pursuant to this registration statement, regardless of the underwriting method
used to sell the securities to the purchaser, if the securities are offered or sold to such purchaser by means of any of the following
communications, the undersigned registrant will be a seller to the purchaser and will be considered to offer or sell such securities
to such purchaser:
|
|
(i)
|
Any preliminary prospectus
or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424 (§230.424
of this chapter);
|
|
|
(ii)
|
Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by the undersigned registrant;
|
|
|
(iii)
|
The portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant or its securities provided by or on behalf of the undersigned registrant; and
|
|
|
(iv)
|
Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser.
|
|
|
(b)
|
The undersigned Registrant
hereby undertakes that, for purposes of determining any liability under the Securities Act of 1933, each filing of the Registrant’s
annual report pursuant to Section 13(a) or Section 15(d) of the Securities Exchange Act of 1934 that is incorporated
by reference in the registration statement shall be deemed to be a new registration statement relating to the securities offered
therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
|
|
|
(c)
|
Insofar as indemnification
for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers and controlling persons of the
Registrant pursuant to the foregoing provisions, or otherwise, the Registrant has been advised that in the opinion of the Securities
and Exchange Commission such indemnification is against public policy as expressed in the Act and is, therefore, unenforceable.
In the event that a claim for indemnification against such liabilities (other than the payment by the Registrant of expenses incurred
or paid by a director, officer or controlling person of the Registrant in the successful defense of any action, suit or proceeding)
is asserted by such director, officer or controlling person in connection with the securities being registered, the Registrant
will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate
jurisdiction the question whether such indemnification by it is against public policy as expressed in the Act and will be governed
by the final adjudication of such issue.
|
SIGNATURES
Pursuant to the requirements
of the Securities Act of 1933, as amended, the registrant certifies that it has reasonable grounds to believe that it meets all
of the requirements for filing on Form S-3 and has duly caused this Registration Statement to be signed on its behalf by the undersigned,
thereunto duly authorized, in the City of Waltham, Massachusetts on January 27, 2017.
|
|
NeuroMetrix, Inc.
|
|
|
|
|
|
|
By:
|
/s/ SHAI N. GOZANI, M.D. PH.D.
|
|
|
Name:
|
Shai N. Gozani, M.D., Ph.D.
|
|
|
Title:
|
Chairman, President and Chief Executive Officer
|
POWER OF ATTORNEY
We, the undersigned officers and directors
of NeuroMetrix, Inc. hereby severally constitute and appoint Shai N. Gozani, M.D., Ph.D. and Thomas T. Higgins, and each of them
singly, our true and lawful attorneys with full power to any of them, and to each of them singly, to sign for us and in our names
in the capacities indicated below the Registration Statement on Form S-3 filed herewith and any and all amendments to said Registration
Statement and generally to do all such things in our name and behalf in our capacities as officers and directors to enable NeuroMetrix,
Inc. to comply with the provisions of the Securities Act of 1933, as amended, and all requirements of the Securities and Exchange
Commission, hereby ratifying and confirming our signatures as they may be signed by our said attorneys, or any of them, to said
Registration Statement and any and all amendments thereto.
Pursuant to the requirements of the Securities
Act of 1933, as amended, this Registration Statement has been signed by the following persons in the capacities and on the dates
indicated.
|
Signature
|
|
Title
|
|
Date
|
|
|
|
|
|
|
|
/s/ SHAI N. GOZANI, M.D., PH.D.
|
|
Chairman, President and Chief Executive Officer
|
|
January 27, 2017
|
|
Shai N. Gozani, M.D., PH.D.
|
|
(principal executive officer)
|
|
|
|
|
|
|
|
|
|
/s/ THOMAS T. HIGGINS
|
|
Senior Vice President, Chief Financial Officer and Treasurer
|
|
January 27, 2017
|
|
Thomas T. Higgins
|
|
(principal financial and accounting officer)
|
|
|
|
|
|
|
|
|
|
/s/ DAVID E. GOODMAN, M.D.
|
|
Director
|
|
January 27, 2017
|
|
David E. Goodman, M.D.
|
|
|
|
|
|
|
|
|
|
|
|
/s/ ALLEN J. HINKLE M.D.
|
|
Director
|
|
January 27, 2017
|
|
Allen J. Hinkle M.D.
|
|
|
|
|
|
|
|
|
|
|
|
/s/ NANCY E. KATZ
|
|
Director
|
|
January 27, 2017
|
|
Nancy E. Katz
|
|
|
|
|
|
|
|
|
|
|
|
/s/ TIMOTHY R. SURGENOR
|
|
Director
|
|
January 27, 2017
|
|
Timothy R. Surgenor
|
|
|
|
|
|
|
|
|
|
|
|
/s/ DAVID VAN AVERMAETE
|
|
Director
|
|
January 27, 2017
|
|
David Van Avermaete
|
|
|
|
|
Exhibit Index
Exhibit
Number
|
|
Description
|
|
|
|
|
|
3.1
|
|
Certificate of Preferences, Rights and Limitations of Series E Convertible Preferred Stock.*
|
|
|
|
|
4.1
|
|
Form of Common Stock Purchase Warrant. *
|
|
|
|
|
|
4.2
|
|
Amendment No. 7 to Shareholder Rights Agreement.*
|
|
|
|
|
5.1
|
|
Opinion of Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C.**
|
|
|
|
|
10.1
|
|
Form of Securities Purchase Agreement dated as of December 28, 2016, by and among NeuroMetrix, Inc. and the purchasers named therein, as amended.*
|
|
|
|
|
10.2
|
|
Form of Registration Rights Agreement dated as of December 28, 2016, by and among NeuroMetrix, Inc. and the purchasers named therein.*
|
|
|
|
|
|
10.3
|
|
Amendment to Engagement Agreement with Rodman & Renshaw, as amended, dated as of January
3, 2017**
|
|
|
|
|
23.1
|
|
Consent of PricewaterhouseCoopers LLP, an independent registered public accounting firm.**
|
|
|
|
|
23.2
|
|
Consent of Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C. (included in Exhibit 5.1).
|
|
|
|
|
24.1
|
|
Power of Attorney (included in the signature pages to the Registration Statement).
|
*Previously filed with the Current Report on
Form 8-K filed on December 29, 2016.
** Filed herewith.
NeuroMetrix (NASDAQ:NURO)
Historical Stock Chart
From Apr 2024 to May 2024
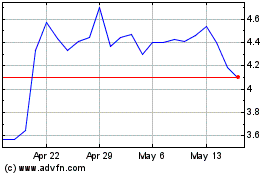
NeuroMetrix (NASDAQ:NURO)
Historical Stock Chart
From May 2023 to May 2024