Gov. Hogan, Novavax’ Erck, & BIO’s Dr. McMurry-Heath Toured Novavax Labs and Discussed Progress on COVID-19 Vaccine
September 24 2020 - 3:29PM
Business Wire
Event Highlighted Leadership in Vaccine
Development and Regional Growth in Biotechnology Innovation
Today, Maryland Governor Larry Hogan and Dr. Michelle
McMurry-Heath, President & CEO of Biotechnology Innovation
Organization (BIO), joined Novavax President & CEO Stanley Erck
to discuss the importance of the biotechnology sector in Maryland,
the industry’s efforts to address COVID-19, and progress being made
by Novavax to develop a vaccine for the deadly virus. A video of
the remarks is now available.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20200924005894/en/
Left to right, Novavax CEO, Stanley Erck,
MD Gov Larry Hogan, BIO CEO Dr. Michelle McMurry-Heath (Photo:
Business Wire)
"When the COVID-19 crisis began, even before we had our first
confirmed cases, I said that our state was home to some of the top
health research facilities in the world, and I hoped that we might
be a part of developing treatments and perhaps even a vaccine for
this deadly virus," said Governor Hogan. "Novavax took that
challenge and immediately hit the ground running. The State of
Maryland is proud to support the efforts of Novavax and our entire
hub of life sciences companies, which is at the forefront in the
fight against COVID-19."
Dr. Michelle McMurry-Heath spoke about the unprecedented
response by the biotechnology industry to fight COVID-19. “Novavax
is a shining example of that response, that galvanized intellectual
capacity, and the ability to put the power of science to healing
the world,” she said. “We at the Biotechnology Innovation
Organization couldn’t be prouder of how our companies, our
scientists, our entrepreneurs, have really mobilized to change the
world.”
“We are about to enter three different phase 3 trials, including
a trial that is starting imminently in the UK in 10,000 people, a
trial that is starting in the U.S. in a few weeks that will be
30,000 people and a third phase 3 trial in India. We have a global
presence. This is a global disease. I don’t think it’s sufficient
that we have a vaccine that is just used for the United States. To
protect the United States, you need to protect the world. It is a
global economy. And that’s what we’re doing,” Erck said. “We feel a
tremendous responsibility in our role at the forefront of vaccine
development and in our mission to deliver a safe and effective
vaccine as rapidly as possible to help combat the COVID-19
pandemic. As a science-based organization dedicated to improving
health, it has been gratifying to see many like-minded stakeholders
in our region come together to that end.”
The remarks were made on-site at Novavax in Gaithersburg,
Maryland. Full video of the remarks is available here.
About BIO
BIO is the world's largest trade association representing
biotechnology companies, academic institutions, state biotechnology
centers and related organizations across the United States and in
more than 30 other nations. BIO members are involved in the
research and development of innovative healthcare, agricultural,
industrial and environmental biotechnology products. BIO also
produces the BIO International Convention, the world’s largest
gathering of the biotechnology industry, along with
industry-leading investor and partnering meetings held around the
world.
About Novavax
Novavax, Inc. (Nasdaq:NVAX) is a late-stage biotechnology
company that promotes improved health globally through the
discovery, development, and commercialization of innovative
vaccines to prevent serious infectious diseases. Novavax is
undergoing clinical trials for NVX-CoV2373, its vaccine candidate
against SARS-CoV-2, the virus that causes COVID-19. NanoFlu™, its
quadrivalent influenza nanoparticle vaccine, met all primary
objectives in its pivotal Phase 3 clinical trial in older adults.
Both vaccine candidates incorporate Novavax’ proprietary
saponin-based Matrix-M™ adjuvant in order to enhance the immune
response and stimulate high levels of neutralizing antibodies.
Novavax is a leading innovator of recombinant vaccines; its
proprietary recombinant technology platform combines the power and
speed of genetic engineering to efficiently produce highly
immunogenic nanoparticles in order to address urgent global health
needs.
About NVX-CoV2373
NVX‑CoV2373 is a vaccine candidate engineered from the genetic
sequence of SARS‑CoV‑2, the virus that causes COVID-19 disease.
NVX‑CoV2373 was created using Novavax’ recombinant nanoparticle
technology to generate antigen derived from the coronavirus spike
(S) protein and contains Novavax’ patented saponin-based Matrix-M™
adjuvant to enhance the immune response and stimulate high levels
of neutralizing antibodies. NVX-CoV2373 contains purified protein
antigens and cannot replicate, nor can it cause COVID-19. In
preclinical trials, NVX-CoV2373 demonstrated indication of
antibodies that block binding of spike protein to receptors
targeted by the virus, a critical aspect for effective vaccine
protection. In its the Phase 1 portion of its Phase 1/2 clinical
trial, NVX‑CoV2373 was generally well-tolerated and elicited robust
antibody responses numerically superior to that seen in human
convalescent sera. NVX-CoV2373 is also being evaluated in two
ongoing Phase 2 studies, which began in August; a Phase 2b trial in
South Africa, and a Phase 1/2 continuation in the U.S. and
Australia. Novavax has secured $2 billion in funding for its global
coronavirus vaccine program, including up to $388 million in
funding from the Coalition for Epidemic Preparedness Innovations
(CEPI).
About Matrix-M™
Novavax’ patented saponin-based Matrix-M™ adjuvant has
demonstrated a potent and well-tolerated effect by stimulating the
entry of antigen-presenting cells into the injection site and
enhancing antigen presentation in local lymph nodes, boosting
immune response.
Novavax Forward-Looking Statements
Statements herein relating to the future of Novavax and the
ongoing development of its vaccine and adjuvant products are
forward-looking statements. Novavax cautions that these
forward-looking statements are subject to numerous risks and
uncertainties, which could cause actual results to differ
materially from those expressed or implied by such statements.
These risks and uncertainties include those identified under the
heading “Risk Factors” in the Novavax Annual Report on Form 10-K
for the year ended December 31, 2019, and Quarterly Report on Form
8-K for the period ended June 30, 2020, as filed with the
Securities and Exchange Commission (SEC). We caution investors not
to place considerable reliance on forward-looking statements
contained in this press release. You are encouraged to read our
filings with the SEC, available at sec.gov, for a discussion of
these and other risks and uncertainties. The forward-looking
statements in this press release speak only as of the date of this
document, and we undertake no obligation to update or revise any of
the statements. Our business is subject to substantial risks and
uncertainties, including those referenced above. Investors,
potential investors, and others should give careful consideration
to these risks and uncertainties.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200924005894/en/
BIO Media Theresa Brady
tbrady@bio.org
For more information visit: https://www.bio.org/ Follow us on
Twitter @IAmBiotech Find us on LinkedIn
Novavax Investors Silvia Taylor and
Erika Trahan ir@novavax.com 240-268-2022
Media Brandzone/KOGS Communication
Edna Kaplan kaplan@kogspr.com 617-974-8659
For more information, visit www.novavax.com and connect with us
on Twitter and LinkedIn.
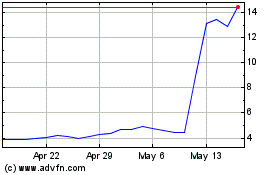
Novavax (NASDAQ:NVAX)
Historical Stock Chart
From Apr 2024 to May 2024
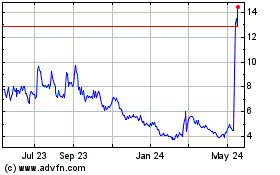
Novavax (NASDAQ:NVAX)
Historical Stock Chart
From May 2023 to May 2024