Pharnext to Hold Its First Satellite Symposium "Focus on Charcot-Marie-Tooth Type 1A Disease" at the 23rd Neuromuscular Days ...
August 31 2017 - 10:45AM
Business Wire
Regulatory News:
Pharnext SA (Paris:ALPHA) (FR00111911287 - ALPHA),
a biopharmaceutical company pioneering a new approach to the
development of innovative drugs based on the combination and
repositioning of known drugs, today announced that it will hold its
first Satellite Symposium "Focus on Charcot-Marie-Tooth Type 1A
Disease" (CMT1A) on September 8th, 2017 at the 23rd Neuromuscular
Days (23èmes Journées Neuromusculaires) in Marseille, France.
Professor D. Adams, MD, PhD (Paris, France), will chair the
session. Speakers are recognized leaders in the field of
neurodenegerative diseases: Prof. S. Attarian, MD, PhD (Marseille,
France) ; Prof. Y. Péréon, MD, PhD (Nantes, France) and Prof. L.
Magy, MD, PhD (Limoges, France). Topics discussed by the faculty
during this CMT1A-focused symposium will include: pathophysiology,
clinical scales and natural history of the disease as well as new
therapeutics in development and to come.
Details are as follows:
- Date: Friday, September 08,
2017
- Time: 08:30am to 09:30am
CEST
- Location: La Major Room, 2nd
floor, Palais du Pharo, Marseille, France
For more information about the Pharnext symposium, please click
here
About CMT1ACharcot-Marie-Tooth (CMT) disease encompasses
a heterogeneous group of inherited, progressive, chronic peripheral
neuropathies. CMT type 1A (CMT1A), the most common type of CMT, is
an orphan disease affecting at least 125,000 people in Europe and
the U.S. The genetic mutation responsible for CMT1A is a
duplication of the PMP22 gene coding for a peripheral myelin
protein. Overexpression of this gene causes degradation of the
neuronal sheath (myelin) responsible for nerve dysfunction,
followed by loss of nerve conduction. As a result of peripheral
nerve degradation, patients suffer from progressive muscle atrophy
of their legs and arms causing walking, running, balance problems
and abnormal hand functioning. CMT1A patients end up in wheelchairs
in at least 5% of cases. Patients might also suffer from
mild-to-moderate sensitive disorders. First symptoms usually appear
during adolescence and progressively evolve through patients’
lives. To date, no curative or symptomatic medications have been
approved and treatment consists of supportive care such as
orthotics, leg braces, physical and occupational therapy or
surgery.
About PharnextPharnext is an advanced clinical-stage
biopharmaceutical company founded by renowned scientists and
entrepreneurs including Professor Daniel Cohen, a pioneer in modern
genomics. Pharnext has two lead products in clinical development.
PXT3003 is currently in an international Phase 3 trial for the
treatment of Charcot-Marie-Tooth disease type 1A and benefits from
orphan drug status in Europe and the United States. PXT864 has
generated positive Phase 2 results in Alzheimer’s disease. Pharnext
is the pioneer of a new drug discovery paradigm: PLEOTHERAPY™. The
Company identifies and develops synergic combinations of
repositioned drugs at new optimal lower doses. These PLEODRUG™
offer several key advantages: efficacy, safety and intellectual
property including several product or composition of matter patents
already granted. The Company is supported by a world-class
scientific team.
The company Pharnext is listed on Euronext Growth Stock Exchange
in Paris (ISIN code: FR00111911287).For more information, visit
www.pharnext.com
View source
version on businesswire.com: http://www.businesswire.com/news/home/20170831005809/en/
PharnextXavier Paoli, +33 (0)1 41 09 22 30Chief
Commercial Officercontact@pharnext.comorInvestor Relations
(Europe)MC Services AGAnne Hennecke, +49 211 529252
22anne.hennecke@mc-services.euorMedia Relations
(Europe)ALIZE RPCaroline CarmagnolMargaux Pronost, +33 (0)1 44
54 36 64pharnext@alizerp.comorInvestor Relations (U.S.)Stern
Investor Relations, Inc.Matthew Shinseki,
+1 212-361-1200matthew@sternir.comorMedia Relations
(U.S.)Russo PartnersTony RussoScott Santiamo+1 212-845-4251+1
718-344-5843tony.russo@russopartnersllc.comscott.santiamo@russopartnersllc.comorFinancial
Communication (France)New CAPEmmanuel Huynh, +33 (0)1 44 71 20
40pharnext@newcap.eu
Pharnext (EU:ALPHA)
Historical Stock Chart
From Mar 2024 to Apr 2024
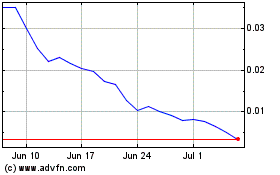
Pharnext (EU:ALPHA)
Historical Stock Chart
From Apr 2023 to Apr 2024