NovaBay Pharmaceuticals Signs Agreement to Distribute COVID-19 Antibody Rapid Point-of-Care Test to U.S. Healthcare Professio...
April 20 2020 - 5:50AM
Business Wire
Company to submit the ISO 13485 and CE Mark
certified fluorecare® test kit under FDA’s EAU and 510(k) clearance
for U.S. commercial use
NovaBay® Pharmaceuticals, Inc. (NYSE American: NBY) announces an
agreement with Shenzhen Microprofit Biotech Co., Ltd. to become the
exclusive U.S. distributor of a rapid, finger prick test to
determine the presence of COVID-19 or a potential indication of
antibody immunity to COVID-19. The fluorecare® SARS-CoV-2 IgG &
IgM Antibody Combined (colloidal gold chromatographic immunoassay)
Test Kit is a point-of-care test to be administered by healthcare
professionals. The test uses a drop of blood for the detection of
COVID-19 antibodies with results available in approximately 10
minutes.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20200420005232/en/
fluorecare® SARS-CoV-2 IgG & IgM
Antibody Combined Test Kit (Photo: Business Wire)
The fluorecare test kit has been validated through widely used
RT-PCR testing to detect immunoglobulin M (IgM), which is the first
antibody produced in response to initial exposure to the COVID-19
antigen, and immunoglobulin G (IgG), which provides a potential
indication of antibody-based immunity to COVID-19. The fluorecare
test kit has been ISO 13485 and CE Mark certified.
“Public health experts and leaders across our country are citing
a critical need for mass testing and tracing procedures for those
who are infected or have been infected with COVID-19 before
reopening the nation’s economy,” said Justin Hall, NovaBay CEO.
“Nasopharyngeal (back of the nose and throat) swabs for molecular
detection are expensive and require laboratory testing that can
lead to delays in obtaining results. Through a simple finger prick,
IgG/IgM testing could provide for cost-effective detection of
COVID-19 antibodies with results available in minutes as an
important step in tracking the infection.
“We are delighted once again to work with our global health
supplier network to secure a product that can help our communities
during the COVID-19 pandemic and, subject to FDA clearance, we plan
to offer the fluorecare test kit at very competitive pricing,” he
added.
NovaBay will submit the fluorecare test kit to the U.S. Food and
Drug Administration (FDA) under Emergency Authorization Use (EAU),
which will be effective until the declaration that circumstances
exist justifying the authorization of emergency use of in vitro
diagnostic tests for detection and/or diagnosis of COVID-19 is
terminated. The Company will also submit the fluorecare test kit
for permanent FDA 510(k) clearance so that the test kit can
continue to be used once the state of emergency has been declared
over by the Federal Government. Because these test kits are one of
the first few test kits of its kind to be reviewed by the FDA,
NovaBay cannot assure a timeline for FDA review and/or clearance
for commercial marketing of the fluorecare test kit in the U.S.
under EAU or 510(k), or if clearance will be granted at all.
About NovaBay Pharmaceuticals, Inc.: Going Beyond
Antibiotics®
NovaBay Pharmaceuticals, Inc. is a biopharmaceutical company
focusing on commercializing and developing its non-antibiotic
anti-infective products to address the unmet therapeutic needs of
the global, topical anti-infective market with its two distinct
product categories: the NEUTROX® family of products and the
AGANOCIDE® compounds. The Neutrox family of products includes
AVENOVA® for the eye care market, CELLERX® for the aesthetic
dermatology market, and NEUTROPHASE® for wound care market.
Forward-Looking Statements
This release contains forward-looking statements that are based
upon management’s current expectations, assumptions, estimates,
projections and beliefs. These statements include, but are not
limited to, statements regarding our ability to obtain both short
and long term FDA approval for the test kits and the impact the
test kits may have on our future financial results. These
statements involve known and unknown risks, uncertainties and other
factors that may cause actual results or achievements to be
materially different and adverse from those expressed in or implied
by the forward-looking statements. Factors that might cause or
contribute to such differences include, but are not limited to,
risks and uncertainties relating to consumer acceptance of our new
branding, and any potential damage to our established goodwill in
the marketplace. Other risks relating to NovaBay’s business,
including risks that could cause results to differ materially from
those projected in the forward-looking statements in this press
release, are detailed in NovaBay’s latest Form 10-Q/K filings with
the Securities and Exchange Commission, especially under the
heading “Risk Factors.” The forward-looking statements in this
release speak only as of this date, and NovaBay disclaims any
intent or obligation to revise or update publicly any
forward-looking statement except as required by law.
Socialize and Stay informed on
NovaBay’s Progress Like us on Facebook Follow us on Twitter Connect with NovaBay on
LinkedIn Visit NovaBay’s
Website
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200420005232/en/
NovaBay Contact Justin Hall
CEO and General Counsel 510-899-8800 jhall@novabay.com
Investor Contact LHA
Investor Relations Jody Cain 310-691-7100 jcain@lhai.com
NovaBay Pharmaceuticals (AMEX:NBY)
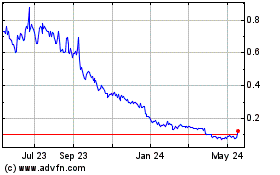
Historical Stock Chart
From Apr 2024 to May 2024
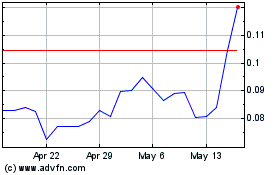
NovaBay Pharmaceuticals (AMEX:NBY)
Historical Stock Chart
From May 2023 to May 2024