Anavex Announces Appointment of Dr. Marwan N Sabbagh, MD as Chairman of the Scientific Advisory Board
September 12 2023 - 6:30AM

Anavex Life Sciences Corp. (“Anavex” or the “Company”) (Nasdaq:
AVXL), a clinical-stage biopharmaceutical company developing
differentiated therapeutics for the treatment of neurodegenerative
and neurodevelopmental disorders including Alzheimer’s disease,
Parkinson’s disease, Rett syndrome and other central nervous system
(CNS) diseases, today announced the appointment of Prof. Dr. Marwan
N Sabbagh, MD, as Chairman of the Scientific Advisory Board.
Prof. Dr. Marwan Sabbagh, MD, is a behavioral
neurologist in the Alzheimer’s and Memory Disorders Program at
Barrow Neurological Institute. He is also a professor and the Vice
Chair of Research in the Institute’s Department of Neurology. He is
board certified in neurology by the American Board of Psychiatry
and Neurology.
Dr. Sabbagh’s expertise includes the diagnosis
and treatment of Alzheimer’s disease and other memory disorders. He
is a fellow of the American Academy of Neurology.
Dr. Sabbagh earned his medical degree from the
University of Arizona College of Medicine in Tucson and his
undergraduate degree from the University of California—Berkeley. He
completed his neurology residency at Baylor College of Medicine in
Houston, Texas, and a geriatric neurology and dementia fellowship
at the University of California San Diego School of Medicine.
Dr. Sabbagh is a leading investigator of many
prominent national Alzheimer’s prevention and treatment clinical
trials. He serves on the editorial boards of the Journal of
Alzheimer’s Disease and Alzheimer’s and Dementia: Translational
Research & Clinical Interventions. He is the editor-in-chief of
Neurology and Therapy. Dr. Sabbagh is a prolific author who has
written and edited books on Alzheimer’s disease and has contributed
to hundreds of medical and scientific articles.
“I’m excited to join Anavex’s Scientific
Advisory Board at this time of important progress of the Company,”
said Professor Dr. Sabbagh. “There is such a significant unmet
medical need around the globe caused by Alzheimer’s disease and
given the robust clinical data of blarcamesine (ANAVEX®2-73) in
Alzheimer’s disease I look forward to joining the scientific team
at Anavex to move forward a path for patients and their loved ones
to be able to provide an effective therapeutic which can be taken
orally and is generally safe.”
“We are delighted to welcome Professor Marwan
Sabbagh as Chairman of Anavex’s Scientific Advisory Board,” stated
Christopher U Missling, PhD, President and Chief Executive Officer
of Anavex. “Professor Sabbagh brings extensive clinical experience
in Alzheimer’s disease to Anavex. I look forward to his guidance
and contributions assisting our dedicated team in the process of
bringing blarcamesine (ANAVEX®2-73) to patients diagnosed with
Alzheimer’s disease.”
About Anavex Life Sciences Corp.
Anavex Life Sciences Corp. (Nasdaq: AVXL) is a
publicly traded biopharmaceutical company dedicated to the
development of novel therapeutics for the treatment of
neurodegenerative and neurodevelopmental disorders, including
Alzheimer's disease, Parkinson's disease, Rett syndrome, and other
central nervous system (CNS) diseases, pain, and various types of
cancer. Anavex's lead drug candidate, ANAVEX®2-73 (blarcamesine),
has successfully completed a Phase 2a and a Phase 2b/3 clinical
trial for Alzheimer's disease, a Phase 2 proof-of-concept study in
Parkinson's disease dementia, and both a Phase 2 and a Phase 3
study in adult patients with Rett syndrome. ANAVEX®2-73 is an
orally available drug candidate that restores cellular homeostasis
by targeting sigma-1 and muscarinic receptors. Preclinical studies
demonstrated its potential to halt and/or reverse the course of
Alzheimer's disease. ANAVEX®2-73 also exhibited anticonvulsant,
anti-amnesic, neuroprotective, and anti-depressant properties in
animal models, indicating its potential to treat additional CNS
disorders, including epilepsy. The Michael J. Fox Foundation for
Parkinson's Research previously awarded Anavex a research grant,
which fully funded a preclinical study to develop ANAVEX®2-73 for
the treatment of Parkinson's disease. ANAVEX®3-71, which targets
sigma-1 and M1 muscarinic receptors, is a promising clinical stage
drug candidate demonstrating disease-modifying activity against the
major hallmarks of Alzheimer's disease in transgenic (3xTg-AD)
mice, including cognitive deficits, amyloid, and tau pathologies.
In preclinical trials, ANAVEX®3-71 has shown beneficial effects on
mitochondrial dysfunction and neuroinflammation. Further
information is available at www.anavex.com. You can also connect
with the Company on Twitter, Facebook, Instagram, and LinkedIn.
Forward-Looking Statements
Statements in this press release that are not
strictly historical in nature are forward-looking statements. These
statements are only predictions based on current information and
expectations and involve a number of risks and uncertainties.
Actual events or results may differ materially from those projected
in any of such statements due to various factors, including the
risks set forth in the Company’s most recent Annual Report on Form
10-K filed with the SEC. Readers are cautioned not to place undue
reliance on these forward-looking statements, which speak only as
of the date hereof. All forward-looking statements are qualified in
their entirety by this cautionary statement and Anavex Life
Sciences Corp. undertakes no obligation to revise or update this
press release to reflect events or circumstances after the date
hereof.
For Further Information:Anavex
Life Sciences Corp.Research & Business DevelopmentToll-free:
1-844-689-3939Email: info@anavex.com
Investors:Andrew J.
BarwickiInvestor RelationsTel: 516-662-9461Email:
andrew@barwicki.com
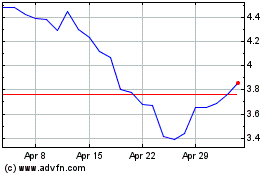
Anavex Life Sciences (NASDAQ:AVXL)
Historical Stock Chart
From Jun 2024 to Jul 2024
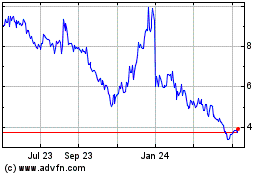
Anavex Life Sciences (NASDAQ:AVXL)
Historical Stock Chart
From Jul 2023 to Jul 2024