Caelum Biosciences Announces Collaborations with the Amyloidosis Advocacy Community and Support of Amyloidosis Awareness Mont...
March 01 2021 - 7:00AM

Caelum Biosciences, Inc. (“Caelum”), a clinical-stage biotechnology
company developing treatments for rare and life-threatening
diseases, today announced its collaboration with and support of the
amyloidosis advocacy community as Amyloidosis Awareness Month
begins. The campaign was created to spread awareness about this
rare disease and its symptoms to help prompt earlier diagnosis for
patients.
In honor of the month and Rare Disease Day, Caelum has made
several commitments to the amyloidosis community that seek to raise
awareness, educate, support and empower patients diagnosed with
amyloidosis and their families. Caelum has provided an unrestricted
donation to the Amyloidosis Support Groups; provided support for
the Amyloidosis Foundation patient travel grant program, which
provides travel expense assistance to medical appointments for
newly diagnosed patients and their caregivers; and made a donation
to the Cardiac Amyloidosis in Athletes Foundation.
In addition, throughout the month of March, Caelum will be
recognizing the “Light the Night for Amyloidosis” campaign that
drives awareness about amyloidosis by encouraging participants to
light up their entryways with red bulbs during the month.
Amyloidosis represents several different types of diseases where
an abnormal protein, amyloid, is produced. Amyloid protein fibers
can cause significant health problems as they attach and deposit
into organs, tissues, nerves and other places in the body. The most
common type, AL amyloidosis, is a rare systemic disorder caused by
an abnormality of plasma cells in the bone marrow. Symptoms of AL
amyloidosis vary by patient, depending on which organs are affected
by the amyloid deposits, but can include impairment of many organs,
nerves and soft tissues, among them the kidneys, heart, digestive
system and nervous system. Chronic kidney disease, arrhythmia,
nausea, diarrhea, constipation, peripheral neuropathy and autonomic
neuropathy are potential symptoms of the disease.
“Caelum is proud to work with the amyloidosis community to
increase awareness of this devastating set of diseases,”
said Michael Spector, President and Chief Executive Officer of
Caelum. “We are grateful for the patients who participate in our
clinical trials and their families, as it is with their ongoing
commitment that we hope to improve the current standard of care by
advancing a treatment that directly addresses the organ dysfunction
caused by amyloid deposition.”
Caelum’s Cardiac Amyloid Reaching for Extended Survival (CARES)
clinical program is evaluating CAEL-101, a first-in-class
monoclonal antibody designed to improve organ function by reducing
or eliminating amyloid deposits in the tissues and organs of
patients with AL amyloidosis. Enrollment is ongoing in two parallel
Phase 3 studies – one in patients with Mayo stage IIIa disease and
one in patients with Mayo stage IIIb disease (ClinicalTrials.gov
Identifier: NCT04512235 and NCT04504825), both of which provide a
travel stipend for participating patients. The company also has a
Phase 2 clinical study that is evaluating the safety and
tolerability of CAEL-101 in patients with AL amyloidosis
(ClinicalTrials.gov Identifier: NCT04304144).
About CAEL-101CAEL-101 is a first-in-class
monoclonal antibody (mAb) designed to improve organ function by
reducing or eliminating amyloid deposits in the tissues and organs
of patients with AL amyloidosis. The antibody is designed to bind
to misfolded light chain protein and amyloid and shows binding to
both kappa and lambda subtypes. In a Phase 1a/1b study, CAEL-101
demonstrated improved organ function, including cardiac and renal
function, in 27 patients with relapsed and refractory AL
amyloidosis who had previously not had an organ response to
standard of care therapy. CAEL-101 has received Orphan Drug
Designation from both the U.S. Food and Drug Administration and
European Medicine Agency as a therapy for patients with AL
amyloidosis.
About AL AmyloidosisAL amyloidosis is a rare
systemic disorder caused by an abnormality of plasma cells in the
bone marrow. Misfolded immunoglobulin light chains produced by
plasma cells aggregate and form fibrils that deposit in tissues and
organs. This deposition can cause widespread and progressive organ
damage and high mortality rates, with death most frequently
occurring as a result of cardiac failure. Current standard of care
includes plasma cell directed chemotherapy and autologous stem cell
transplant, but these therapies do not address the organ
dysfunction caused by amyloid deposition, and up to 80 percent of
patients are ineligible for transplant. AL amyloidosis is a rare
disease but is the most common form of amyloidosis. There are
approximately 22,000 patients across the United States, France,
Germany, Italy, Spain and the United Kingdom. AL amyloidosis has a
one-year mortality rate of 47 percent, 76 percent of which is
caused by cardiac amyloidosis.
About Caelum BiosciencesCaelum Biosciences,
Inc. (“Caelum”) is a clinical-stage biotechnology company
developing treatments for rare and life-threatening diseases.
Caelum’s lead asset, CAEL-101, is a novel antibody for the
treatment of patients with amyloid light chain (“AL”) amyloidosis.
In 2019, Caelum entered a collaboration agreement with Alexion
Pharmaceuticals, Inc. (“Alexion”) under which Alexion acquired a
minority equity interest in Caelum and an exclusive option to
acquire the remaining equity in the company. Caelum was founded by
Fortress Biotech, Inc. (NASDAQ: FBIO). For more information,
visit www.caelumbio.com.Company
Contacts:Michael Spector, President & Chief Executive
Officermspector@caelumbio.com
Jaclyn Jaffe and William BegienInvestor Relations(781)
652-4500info@caelumbio.com
Media Relations Contact:Tony Plohoros6
Degrees(908) 591-2839tplohoros@6degreespr.com
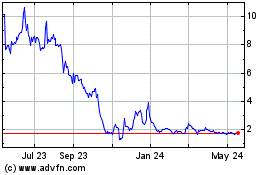
Fortress Biotech (NASDAQ:FBIO)
Historical Stock Chart
From Apr 2024 to May 2024
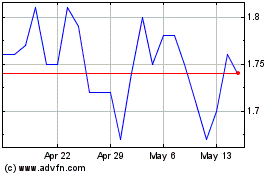
Fortress Biotech (NASDAQ:FBIO)
Historical Stock Chart
From May 2023 to May 2024