- Poster presentation of "A Randomized, Double-Blind Controlled
Comparison of NRX-101 (D-cycloserine/lurasidone) to Lurasidone for
Adults with Bipolar Depression and Subacute Suicidal Ideation or
Behavior"
- NRX-101 demonstrated a similar antidepressant effect (MADRS
reduction ~50%) compared to lurasidone, the Standard of Care
drug
- NRX-101 demonstrated a 58% relative reduction in time to
sustained remission from suicidality (P=.05) compared to
lurasidone
- NRX-101 demonstrated a 76% reduction in symptoms of akathisia
(p=0.03), a side effect linked to suicide, compared to
lurasidone
- This represents the second trial of NRX-101 demonstrating
reduction in suicidality and akathisia associated with NRX-101
compared to lurasidone
RADNOR,
Pa., May 28, 2024 /PRNewswire/ -- NRx
Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", "NRx",
the "Company"), a clinical stage pharmaceutical company, today
announced presentation of its Phase 2b/3 trial of NRX-101, entitled "A
Randomized, Double-Blind Controlled Comparison of NRX-101
(D-cycloserine/lurasidone) to Lurasidone for Adults with Bipolar
Depression and Subacute Suicidal Ideation or Behavior" at the
American Society of Clinical Psychopharmacology (ASCP) in
Miami Beach, FL. The lead author
is Prof. Andrew Nierenberg,
Director, Dauten Family Center for Bipolar Treatment
Innovation, Massachusetts General Hospital and Professor of
Psychiatry, Harvard Medical School.
"Presentation of these data at this highly respected conference
is another important step toward bringing a life-saving product to
patients in tremendous need," said Dr. Jonathan Javitt, Chairman and Chief Scientist of
NRx. "We believe that NRX-101 may offer a paradigm changing
approach to treatment of Bipolar Depression, with a product highly
effective in both treating depression and reducing suicidality and
associated side effects. We will continue working to bring
hope to life with life-saving medications."
The presentation will be held at 11:15
AM, Wednesday May 29,
2024.
W89 A Randomized, Double-Blind Controlled Comparison
of NRX-101 (D-cycloserine/lurasidone) to Lurasidone for Adults with
Bipolar Depression and Subacute Suicidal Ideation or Behavior
CONCLUSIONS of the Poster are:
- NRX-101 and lurasidone both demonstrated > 50% response for
treating bipolar depression with no difference seen on primary
efficacy endpoint (MADRS)
- A clinically meaningful difference was observed on both primary
and secondary safety endpoints favoring NRX-101
- NRX-101 was associated with 58% relative reduction in time to
sustained remission from suicidality as measured by the Columbia
Suicide Severity Rating Scale (C-SSRS) when stratified by sex, mood
stabilizer use, antipsychotic use, lifetime suicide event
(P=0.05).
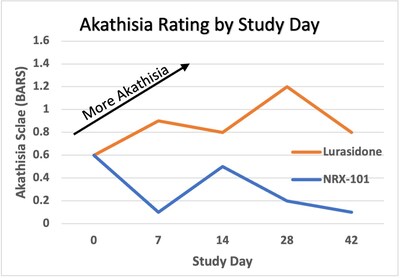
- NRX-101 was associated with a relative 76% reduction in
symptoms of akathisia compared to lurasidone that was sustained
over 42 days (Effect Size 0.37; P=0.03) on the Barnes Akathisia
Rating Scale
- Akathisia was seen in 2% of participants treated with NRX-101
vs. 11% treated with lurasidone.
- NRX-101 showed superiority to lurasidone in akathisia starting
at day 7 and continuing through day 42/ET.
- No treatment-related serious adverse event was observed in
either group. No safety issues were detected except for MedDRA
General disorders: NRX-101 - 18.2% vs lurasidone - 0%
(p=0.002).
Based on these findings, together with the earlier STABIL-B
trial, the Company believes that NRX-101 has potential to become a
standard of care drug for treating bipolar depression, an
addressable population of 7 million patients in the US and many
times that around the world.
This study represents the second trial conducted under
FDA Good Clinical Practices guidelines to demonstrate large and
meaningful advantages of NRX-101 vs lurasidone on akathisia and
suicidality and clears the path for a registration trial of NRX-101
vs. placebo to treat bipolar depression together with earlier
accelerated approval for those with akathisia. An additional
academic trial conducted by Chen and co-workers similarly
demonstrated a statistically-significant reduction in suicidality
associated with D-cycloserine, the active ingredient in NRX-101,
compared to various standard of care antidepressants.
To the Company's knowledge, this trial and its prior STABIL-B
study represent the only clinical trials in which an oral
antidepressant has been demonstrated to cause meaningful reductions
in suicidality and akathisia. All currently approved antidepressant
drugs carry FDA-mandated "black box" warnings on their labels
indicating that they may increase the risk of suicide. Similarly,
akathisia – a side effect in which patients are agitated and
frequently experience involuntary movement – is a side effect that
occurs in 10-15% of patients who take the lurasidone class of drugs
and is closely linked to suicide. As shown in the clinical trial,
those randomized to lurasidone experienced a substantial increase
in akathisia from baseline, whereas those randomized to NRX-101
demonstrated a statistically-significant reduction in akathisia
(see Figure).
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical
company developing therapeutics based on its NMDA platform for the
treatment of central nervous system disorders, specifically
suicidal bipolar depression, chronic pain and PTSD. The Company is
developing NRX-101, an FDA-designated investigational Breakthrough
Therapy for suicidal treatment-resistant bipolar depression and
chronic pain. NRx has partnered with Alvogen and Lotus around the
development and marketing of NRX-101 for the treatment of suicidal
bipolar depression. NRX-101 additionally has potential to act as a
non-opioid treatment for chronic pain, as well as a treatment for
complicated UTI.
NRx has recently announced plans to submit a New Drug
Application for HTX-100 (IV ketamine), through Hope Therapeutics,
in the treatment of suicidal depression, based on results of
well-controlled clinical trials conducted under the auspices of the
US National Institutes of Health and newly obtained data from
French health authorities, licensed under a data sharing agreement.
NRx was awarded Fast Track Designation for development of ketamine
(NRX-100) by the US FDA as part of a protocol to treat patients
with acute suicidality.
Notice Regarding Forward-Looking Statements
The information contained herein includes forward-looking
statements within the meaning of Section 21E of the Securities
Exchange Act of 1934, as amended, and Section 27A of the Securities
Act of 1933, as amended. These statements include, among others,
statements regarding the proposed public offering and the timing
and the use of the proceeds from the offering. Forward-looking
statements generally include statements that are predictive in
nature and depend upon or refer to future events or conditions, and
include words such as "may," "will," "should," "would," "expect,"
"plan," "believe," "intend," "look forward," and other similar
expressions among others. These statements relate to future events
or to the Company's future financial performance, and involve known
and unknown risks, uncertainties and other factors that may cause
the Company's actual results to be materially different from any
future results, levels of activity, performance or achievements
expressed or implied by these forward-looking statements. You
should not place undue reliance on forward-looking statements since
they involve known and unknown risks, uncertainties and other
factors which are, in some cases, beyond the Company's control and
which could, and likely will, materially affect actual results,
levels of activity, performance or achievements. Any
forward-looking statement reflects the Company's current views with
respect to future events and is subject to these and other risks,
uncertainties and assumptions relating to the Company's operations,
results of operations, growth strategy and liquidity. More detailed
information about the Company and the risk factors that may affect
the realization of forward-looking statements is set forth in the
Company's most recent Annual Report on Form 10-K and other filings
with the Securities and Exchange Commission. Investors and security
holders are urged to read these documents free of charge on the
SEC's website at http://www.sec.gov. Except as may be required
by applicable law, The Company assumes no obligation to publicly
update or revise these forward-looking statements for any reason,
or to update the reasons actual results could differ materially
from those anticipated in these forward-looking statements, whether
as a result of new information, future events or otherwise.

 View original content to download
multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-presents-landmark-trial-of-nrx-101-in-suicidal-bipolar-depression-at-the-american-society-of-clinical-psychopharmacology-annual-meeting-nrx-101-is-the-first-oral-antidepressant-demonstrated-to-re-302156419.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-presents-landmark-trial-of-nrx-101-in-suicidal-bipolar-depression-at-the-american-society-of-clinical-psychopharmacology-annual-meeting-nrx-101-is-the-first-oral-antidepressant-demonstrated-to-re-302156419.html
SOURCE NRx Pharmaceuticals, Inc.