FDA Probes Cause of Failed Johnson & Johnson Covid-19 Vaccine Batch
April 01 2021 - 6:56PM
Dow Jones News
By Peter Loftus and Thomas M. Burton
The Food and Drug Administration is investigating what caused a
batch of the active ingredient for Johnson & Johnson's Covid-19
vaccine to be scrapped for failing to meet quality standards at a
contract manufacturing plant, according to a person familiar with
the matter.
The FDA may send an inspection team to assess the situation at
the Baltimore plant operated by contractor Emergent BioSolutions
Inc., the person said.
The regulatory scrutiny follows J&J's disclosure Wednesday
that a batch of the main ingredient for its Covid-19 vaccine
manufactured at the Emergent plant didn't meet standards. The batch
didn't reach the vial-filling and finishing stage, and no doses
from it were distributed.
J&J says the quality lapse didn't affect vaccine doses that
have been distributed in the U.S. since the vaccine was authorized
in late February, and the company still has enough supply to meet
near-term commitments. J&J also makes the main ingredient for
the vaccine at its own plant in the Netherlands.
Emergent BioSolutions, of Gaithersburg, Md., said in a statement
Thursday that it isolated the batch of vaccine ingredients because
it didn't meet specifications and quality standards. Emergent said
it would dispose of the batch properly.
The company said that discarding a batch is disappointing but
occasionally happens during the complex vaccine manufacturing
process.
The White House said Thursday it didn't expect the issue to
affect the promised U.S. supply of J&J's vaccine. J&J
expects to deliver about 100 million doses for use in the U.S. by
midyear, under the terms of a $1 billion purchase agreement with
the federal government.
"We have been assured that they expect to meet those deadlines,"
White House press secretary Jen Psaki said at a briefing.
Ms. Psaki said "the issue was identified as part of rigorous
quality control system checks," and the Department of Health and
Human Services notified the White House late last week.
Asked if the White House should have revealed the problem
publicly earlier, Ms. Psaki said the plant hasn't yet been
FDA-approved. "This is probably the process of working as it
should," she said. Drug plants can manufacture ingredients for a
drug or vaccine but finished products can't be released until the
FDA authorizes them.
J&J's vaccine is the third to be authorized in the U.S.,
after shots from Pfizer Inc. with its partner BioNTech SE, and
Moderna Inc. Health authorities have been counting on the arrival
of the single-shot J&J vaccine to boost the overall supply of
shots and to simplify vaccinations because it doesn't require a
second dose.
Emergent said that it was confident in its ability to meet FDA
requirements.
Emergent makes the main ingredient for AstraZeneca PLC's
Covid-19 vaccine at the same plant, though that vaccine isn't
authorized for use in the U.S. An AstraZeneca spokeswoman said the
company was "aware of the reports regarding the facility and we
understand Emergent is investigating the matter."
Emergent Chief Executive Robert Kramer told CNBC Thursday "it
wasn't the case where an ingredient from one vaccine contaminated
or impacted the other." He said the J&J batch didn't meet
quality specifications but he didn't elaborate on what
happened.
Vaccine manufacturing experts say the issues at the Emergent
plant have to be addressed, but the detection of the problem before
doses from the batch were released was an encouraging sign.
"It's definitely alarming but it also shows that this is a
system that eventually works," said Tinglong Dai, associate
professor of operations management and business analytics at Johns
Hopkins University's Carey Business School, who has researched the
vaccine supply chain. "I have high confidence in the error
detection part."
J&J said it was sending additional experts in manufacturing,
technical operations and quality to be on-site at Emergent to
oversee all manufacturing of the J&J vaccine there.
Tarini Parti contributed to this article.
Write to Peter Loftus at peter.loftus@wsj.com and Thomas M.
Burton at tom.burton@wsj.com
(END) Dow Jones Newswires
April 01, 2021 19:41 ET (23:41 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
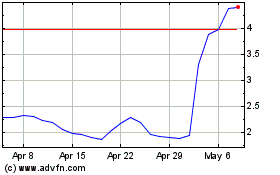
Emergent Biosolutions (NYSE:EBS)
Historical Stock Chart
From Apr 2024 to May 2024
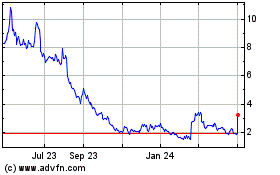
Emergent Biosolutions (NYSE:EBS)
Historical Stock Chart
From May 2023 to May 2024