Filed Pursuant to Rule 424(b)(3)
Registrations No. 333-262493
PROSPECTUS

40,000,000
Shares of Common Stock
This
prospectus relates to the offer and resale of up to 40,000,000 shares of our common stock, par value $0.001 per share by Dutchess Capital
Growth Fund LP (“Dutchess” or the “Selling Security Holder”) consisting of 40,000,000 shares of the Company’s
common stock that may be purchased from us pursuant to the Common Stock Purchase Agreement that we entered into with Dutchess on November
30, 2021(the “Purchase Agreement”).
The
Selling Security Holder may sell all or a portion of the shares being offered pursuant to this prospectus at fixed prices and prevailing
market prices at the time of sale, at varying prices, or at negotiated prices.
We
will not receive any proceeds from the sale of securities under the Purchase Agreement sold by Dutchess. However, we will receive proceeds
from our initial sale of shares to Dutchess pursuant to the Purchase Agreement. Pursuant to the terms of Purchase Agreement, we will
sell shares to purchase at a price equal to 92% of the lowest closing price of our common stock during the five (5) business days prior
to the Closing Date. Closing Date shall mean the five (5) business days after the Clearing Date. Clearing Date shall mean the first business
day that the Selling Security Holder holds the Draw Down Amount in its brokerage account and is eligible to trade the shares.
Dutchess
may sell the shares of common stock described in this Prospectus in a number of different ways and at varying prices. See “Plan
of Distribution” for more information about how the Selling Security Holder may sell the shares of common stock being registered
pursuant to this Prospectus.
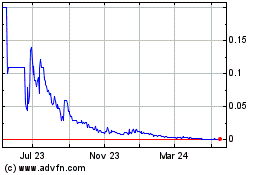
Our
Common Stock is quoted for trading on the OTCQB Marketplace (OTCQB) under the symbol “PPCB”. As of February 2, 2022,
the closing bid price for our Common Stock as reported on the OTCQB was $0.0198 per share.
This
prospectus provides a general description of the securities being offered. You should read this prospectus and the registration statement
of which it forms a part before you invest in any securities.
Investing
in our Common Stock should be considered speculative and involves a high degree of risk, including the risk of losing your entire investment.
See “Risk Factors” to read about the risks you should consider before buying shares of our Common Stock.
You
should rely only on the information contained in this prospectus or any prospectus supplement or amendment hereto. We have not authorized
anyone to provide you with different information.
Our
auditors have issued a going concern opinion. For more information please see the going concern opinion on page F-2 and the risk
factors herein.
The
Selling Security Holder is an “underwriter” within the meaning of the Securities Act of 1933. The Selling Security Holder
is offering these shares of common stock. The Selling Security Holder may sell all or a portion of these shares from time to time in
market transactions through any market on which our common stock is then traded, in negotiated transactions or otherwise, and at prices
and on terms that will be determined by the then prevailing market price or at negotiated prices directly or through a broker or brokers,
who may act as agent or as principal or by a combination of such methods of sale. The Selling Security Holder will receive all proceeds
from the sale of the common stock. For additional information on the methods of sale, you should refer to the section entitled “Plan
of Distribution.”
Neither
the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined
if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The
date of this prospectus is February 11, 2022
TABLE
OF CONTENTS
You
may only rely on the information contained in this prospectus or that we have referred you to. We have not authorized anyone to provide
you with different information. This prospectus does not constitute an offer to sell or a solicitation of an offer to buy any securities
other than the Common Stock offered by this prospectus. This prospectus does not constitute an offer to sell or a solicitation of an
offer to buy any Common Stock in any circumstances in which such offer or solicitation is unlawful. Neither the delivery of this prospectus
nor any sale made in connection with this prospectus shall, under any circumstances, create any implication that there has been no change
in our affairs since the date of this prospectus is correct as of any time after its date.
ABOUT
THIS PROSPECTUS
You
should rely only on the information that we have provided in this prospectus and any applicable prospectus supplement. We have not authorized
anyone to provide you with different information. No dealer, salesperson or other person is authorized to give any information or to
represent anything not contained in this prospectus and any applicable prospectus supplement. You must not rely on any unauthorized information
or representation. This prospectus is an offer to sell only the securities offered hereby, and only under circumstances and in jurisdictions
where it is lawful to do so. You should assume that the information in this prospectus, and any applicable prospectus supplement, is
accurate only as of the date on the front of the document, regardless of the time of delivery of this prospectus, any applicable prospectus
supplement, or any sale of a security. Our business, financial conditions, results of operations and prospects may have changed since
that date.
References
to “Management” in this Prospectus mean the senior officers of the Company; See “Director, Executive Officers and Key
Employees.” Any statements in this Prospectus made by or on behalf of Management are made in such persons’ capacities as
officers of the Company, and not in their personal capacities.
Unless
otherwise indicated or the context requires otherwise, the words “we,” “us,” “our”, the “Company”
or “our Company” refer to Propanc Biopharma, Inc., a Delaware corporation, unless the context indicates otherwise.
PROSPECTUS
SUMMARY
The
following summary highlights material information contained in this Prospectus. This summary does not contain all of the information
you should consider before investing in the securities. Before making an investment decision, you should read the entire Prospectus carefully,
including the risk factors section, the financial statements and the notes to the financial statements. You should also review the other
available information referred to in the section entitled “Where You Can Find More Information” in this Prospectus and any
amendment or supplement hereto.
The
Offering
On
November 30, 2021, we entered into an Common Stock Purchase Agreement (the “Purchase Agreement”) with Dutchess. Although
we are not mandated to sell shares under the Purchase Agreement, the Purchase Agreement gives us the option to sell to Dutchess, up to
$5,000,000 worth of our common stock over the period ending thirty-six (36) months after the execution date of the Purchase Agreement.
In consideration for Dutchess’ execution and performance under the Purchase Agreement, the Company issued 1,000,000 restricted
shares of the Company’s common stock to Dutchess.
On
November 30, 2021, we also entered into a registration rights agreement with Dutchess whereby we are obligated to (i) file with the Commission
the Registration Statement; and (ii) use its best efforts to have the Registration Statement declared effective by the Commission at
the earliest possible date.
Following
effectiveness of the Registration Statement, and subject to certain limitations and conditions set forth in the Purchase Agreement, the
Company shall have the discretion to deliver put notices to Dutchess and Dutchess will be obligated to purchase shares of the Company’s
Common Stock based on the investment amount specified in each put notice. The maximum amount that the Company shall be entitled to put
to Dutchess in each put notice shall not exceed the lesser of (i) 300% of the average daily share volume of the Common
Stock in the five (5) trading days immediately preceding the Draw Down Notice or (ii) an aggregate value of $250,000. Pursuant to the
Purchase Agreement, Dutchess and its affiliates will not be permitted to purchase and the Company may not put shares of the Company’s
Common Stock to Dutchess that would result in Dutchess’ beneficial ownership of the Company’s outstanding Common Stock exceeding
9.99%. The price of each put share shall be equal to ninety two percent (92%) of the Market Price (as defined in the Purchase Agreement).
Puts may be delivered by the Company to Dutchess until the earlier of (i) the date on which Dutchess has purchased an aggregate of $5,000,000
worth of Common Stock under the terms of the Purchase Agreement; (ii) the period ending thirty-six (36) months after the execution date
of the Purchase Agreement; or (iii) written notice of termination delivered by the Company to Dutchess, subject to certain equity conditions
set forth in the Purchase Agreement.
There
is no assurance the market price of our common stock will increase in the future. The number of common shares that remain issuable may
not be sufficient, dependent upon the share price, to allow us to access the full amount contemplated under the Purchase Agreement. If
the bid/ask spread remains the same we will not be able to place a put for the full commitment under the Equity Purchase Agreement. Based
on the lowest closing price of our common stock during the five (5) consecutive trading day period preceding the filing date of this
registration statement of $0.0198, the registration statement covers the offer and possible sale of approximately $728,640
worth of our shares (a discounted price of $0.0182) which is below $5,000,000 (the full amount of the Purchase Agreement).
Dutchess
is not permitted to engage in short sales involving our common stock during the term of the commitment period. In accordance with Regulation
SHO, however, sales of our common stock by Dutchess after delivery of a put notice of such number of shares reasonably expected to be
purchased by Dutchess under a put will not be deemed a short sale.
In
addition, we must deliver the other required documents, instruments and writings required. Dutchess is not required to purchase the put
shares unless:
| |
● |
Our
registration statement with respect to the resale of the shares of common stock delivered in connection with the applicable put shall
have been declared effective; |
| |
|
|
| |
● |
We
shall have obtained all material permits and qualifications required by any applicable state for the offer and sale of the registrable
securities; and |
| |
|
|
| |
● |
We
shall have filed all requisite reports, notices, and other documents with the SEC in a timely manner. |
As
we draw down on the equity line of credit, shares of our common stock may be sold into the market by Dutchess. The sale of these shares
could cause our stock price to decline. In turn, if our stock price declines and we issue more puts, more shares will come into the market,
which could cause a further drop in our stock price. You should be aware that there is an inverse relationship between the market price
of our common stock and the number of shares to be issued under the equity line of credit. If our stock price declines, we will be required
to issue a greater number of shares under the equity line of credit. We have no obligation to utilize the full amount available under
the equity line of credit.
We
may require Dutchess to suspend the sales of the shares of our common stock being offered pursuant to this prospectus upon the occurrence
of any event that makes any statement in this prospectus or the related registration statement untrue in any material respect or that
requires the changing of statements in those documents in order to make statements in those documents not misleading.
Neither
the Purchase Agreement nor any of our rights or Dutchess’ rights thereunder may be assigned to any other person.
BUSINESS
History
We
were originally incorporated in Melbourne, Victoria Australia on October 15, 2007 as Propanc PTY LTD and continue to be based in Camberwell,
Victoria Australia. Since our inception, substantially all of our operations have been focused on the development of new cancer treatments
targeting high-risk patients, particularly cancer survivors, who need a follow-up, non-toxic, long-term therapy designed to prevent the
cancer from returning and spreading. We anticipate establishing global markets for our products.
On
November 23, 2010, our Company was incorporated in the state of Delaware as Propanc Health Group Corporation. In January 2011, to reorganize
our Company, we acquired all of the outstanding shares of Propanc PTY LTD on a one-for-one basis and Propanc PPY LTD became our wholly-owned
subsidiary. Effective April 20, 2017, we changed our name to “Propanc Biopharma, Inc.” to better reflect our stage of operations
and development. On the same date, we also effected a 1-for-250 reverse stock split whereby we (i) decreased the number of authorized
shares of our common stock to 100,000,000 (ii) decreased the number of authorized shares of our preferred stock to 1,500,005 and (iii)
decreased, by a ratio of 1-for-250 the number of retroactively issued and outstanding shares of our common stock.
On
January 23, 2018, we filed a Certificate of Amendment to our Certificate of Incorporation to increase the number of authorized shares
of our common stock from 100,000,000 to 400,000,000. On September 21, 2018, we filed a Certificate of Amendment to our Certificate of
Incorporation to increase the number of authorized shares of our common stock from 400,000,000 to 4,000,000,000.
On June 11, 2019, we
filed a Certificate of Amendment, as amended, to our Certificate of Incorporation to decrease the number of authorized shares of our
common stock from 4,000,000,000 to 100,000,000 in connection with the 1-for-500 reverse stock split that occurred on June 24, 2019.
On
March 13, 2020, we filed a Certificate of Amendment, as amended, to our Certificate of Incorporation to increase the number of
authorized shares of our common stock from 100,000,000 to 1,000,000,000.
On
November 17, 2020, we filed a Certificate of Amendment to our Certificate of Incorporation to effect a 1-for-1,000 Reverse Stock Split
of the Company’s shares of common stock.
Overview
Propanc
Biopharma is a biopharmaceutical company developing a novel approach to prevent recurrence and metastasis from solid tumors by using
pancreatic proenzymes that target and eradicate cancer stem cells in patients suffering from pancreatic, ovarian and colorectal cancers.
Our novel proenzyme therapy is based on the science that enzymes stimulate biological reactions in the body, especially enzymes secreted
by the pancreas. These pancreatic enzymes could represent the body’s primary defense against cancer.
Our
lead product candidate, PRP, is a variation upon our novel formulation and involves proenzymes, the inactive precursors of enzymes. As
a result of positive early indications of the anti-cancer effects of our technology, we have conducted successful pre-clinical studies
on PRP and also commenced preparation for a clinical study in advanced cancer patients. Subject to us receiving sufficient financing,
we plan to begin our Investigational Medicinal Product Dossier, study proposal and Investigator’s Brochure in the 2021 calendar
year. Our plan is to then commence our study preparation process with the contract research organization, analytical lab and trial site(s)
selection and to begin our clinical trial application for PRP (“CTA”) compilation in the first calendar quarter of 2022 and
complete the CTA compilation and submit the CTA in the first half of 2022. In the second quarter of 2022, we plan to begin the preparation
of logistics and trial site initiation visits. Subject to raising additional sufficient capital, we subsequently plan to commence a First-In-Human
(FIH), Phase Ib study in patients with advanced solid tumors, evaluating the safety, pharmacokinetics and anti-tumor efficacy of PRP
in the second half of 2022 calendar year, which study we hope to complete within twelve months thereafter. We intend to develop our PRP
to treat early-stage cancer and pre-cancerous diseases and as a preventative measure for patients at risk of developing cancer based
on genetic screening.
PRP
is an intravenous injection proenzyme treatment designed as a therapeutic option in cancer treatment and prevention. PRP is a combination
of the pancreatic proenzymes, trypsinogen and chymotrypsinogen. PRP produces multiple effects on cancerous cells intended to inhibit
tumor growth and potentially stop a tumor from spreading through the body.
We
received notification from the U.S. Food and Drug Administration (FDA) that PRP had been conferred Orphan Drug Designation for the treatment
of pancreatic cancer. This special status is granted when a rare disease or condition is implicated and a potential treatment qualifies
under the Orphan Drug Act and applicable FDA regulations.
A
Certificate for Advance Overseas Finding was received from the Board of Innovation and Science Australia to receive up to a 43.5% “cash
back” benefit from overseas R&D expenses. The finding relates to the planned Phase 1 clinical trial – Multiple Ascending
Dose Studies of proteolytic proenzymes for the treatment of advanced cancer patients suffering from solid tumors planned to be conducted
at the Peter MacCallum Center, Melbourne, Australia. Overseas activities to be undertaken include the development of an analytical assay
for the quantification of active pharmaceutical ingredients in the Company’s lead product candidate, PRP, and its manufacture of
the finished product for the Phase 1 clinical trial.
Our
POP1 joint research and drug discovery program is designed to produce a backup clinical compound to the lead product candidate, PRP.
With the aim of producing large quantities of trypsinogen and chymotrypsinogen for commercial use, exhibiting minimal variation between
lots and without sourcing the proenzymes from animals, Propanc Biopharma is undertaking a challenging research project in collaboration
with the Universities of Jaén and Granada. We entered into a second two-year joint research and collaboration agreement with the
University of Jaén who are undertaking the research activities for the POP1 program.
Our
Focus
Cancer
occurs when cells in the body start to divide quickly and uncontrollably with an ability to migrate from one location and spread to distant
sites. A cell becomes cancerous when it becomes undifferentiated. The cell forgets to do its job and invests all its energy to proliferating.
Unlike normal cells, cancer cells multiply, but do not differentiate.
Common
cancer therapies take advantage of the uncontrolled proliferation of the cancer cells and kill these cells by targeting the cell division
machinery. These therapies are effective but affect healthy cells as well, particularly those with a high rate of cell turnover, inducing
undesirable side effects.
Our
goal is to stop cancer not by targeting tumor cell death, but inducing cell differentiation. This is known as differentiation therapy.
The key focus is to convince the malignant cells to stop proliferating and return to do their work as a specific cell type. Differentiation
therapy does not target cell death, so healthy cells within the patient will not be compromised, unlike chemotherapeutic drugs or gamma
irradiation.
Differentiation
therapy induces the cancer cells into the pathway of terminal differentiation and eventual senescence (i.e., a non-proliferative state).
Differentiation therapy acts not only against cancer cells, but interestingly can turn cancer stem cells (undifferentiated cells) towards
completely differentiated (i.e., normal) cells.
There
are natural elements within our body that could help us fight against cancer. Enzymes are natural proteins that stimulate and accelerate
biological reactions in the body. Particularly enzymes secreted by the exocrine pancreas that are essential for the digestion of proteins
and fats. More than one hundred years ago, Professor John Beard first proposed that pancreatic enzymes represent the body’s primary
defense against cancer and would be useful as a cancer treatment. Since then, several scientists have endorsed Beard´s hypothesis
with encouraging data from patient treatment.
We
are developing a long-term therapy based on a pancreatic proenzyme formulation to prevent tumor recurrence and metastasis, the main cause
of patient death from cancer. PRP is a novel, patented, formulation consisting of two proenzymes mixed in a synergetic ratio.
After
extensive laboratory research and a limited amount of human data, we have evidence that PRP:
| |
● |
Reduces
cancer cell growth via promotion of cell differentiation; |
| |
● |
Enhances
cell adhesion and may suppress metastasis progression; |
| |
● |
Exhibited
no observable serious side effects and improves patient survival. |
PRP
PRP
is a mixture of two proenzymes, trypsinogen and chymotrypsinogen from bovine pancreas administered by intravenous injection. A synergistic
ratio of 1:6 inhibits growth of most tumor cells. Examples include kidney, ovarian, breast, brain, prostate, colorectal, lung liver,
uterine and skin cancers.
Mechanism
Of Action
Metastasis
occurs because a program inside the cell, called the Epithelial-Mesenchymal Transition (EMT) is activated, which causes epithelial cancer
cells to become invasive and stem cell-like, features which then allow these cancer cells to spread and metastasize. PRP reverses the
conversion from an epithelial to a mesenchymal phenotype and, as such, may reduce the metastatic potential of the tumor cells. PRP also
promotes the acquisition of a less malignant phenotype, in addition to a decrease in proliferation due to lineage (i.e., direct descent)
specific cellular differentiation.
Selectivity
PRP
treatment affects the TGFβ pathway, a significant tumor promoter in late-stage cancer. The likely molecular targets are proteinase-activated-receptors
(PARs) type 1 and 2, which are over frequently overexpressed in many types of cancers. Trypsinogen and chymotrypsinogen are activated
by proteases in the extracellular matrix of tumor cells. In turn, trypsin (activated trypsinogen) has a preference to activate PAR-2,
whilst Chymotrypsin (activated chymotrypsinogen) mainly activates PAR-1.
Effects
Against Cancer Stem Cells
Cancer
Stem Cells are resistant to standard treatments because they remain dormant for long periods, then migrate to other organs, and trigger
explosive tumor growth, causing the patient to relapse. Approximately eighty percent of cancers are from solid tumors and metastasis
is the main cause of patient death. Our unique patented approach is designed to target and eradicate cancer stem cells not killed by
radiation or chemotherapy.
PRP
is designed to target and eradicate cancer stem cells not killed by radiation or chemotherapy. Traditional cancer therapies act on tumor
replicating cells, but not cancer stem cells, so they can rebuild the tumor mass and can migrate to start a new tumor in another organ.
PRP stops cancer stem cells so that a tumor loses the ability to generate new cells and therefore the tumor disappears with no option
to form a metastatic tumor elsewhere.
PRP
treatment regulates up to four relevant pathways related to cancer spread and metastasis of cancer stem cells. PRP acts on TGFβ,
Hippo, Wnt and Notch pathways. It promotes the up-regulation of RAC1b which avoids the hyper-activation of the p38 pathway induced by
the TGFβ pathway, leading to the phosphorylation of YAP, which sequesters B-catenin in the cytoplasm, blocking the canonical Wnt
pathway and inhibiting the Notch pathway. That cascade of reactions implies the disruption of the cancer stem cell phenotype and the
reversal of the malignant epithelial to mesenchymal transition process that leads to tumor invasion.
PRP
Impairs Niche Formation and Tumor Initiation
The
proenzyme treatment inhibits the expression of genes related to the cancer stem cell phenotype, changing these malignant cells toward
a more differentiated and less dangerous cellular condition. PRP interferes with the signals that the primary tumor sends to other tissues
to prepare the pre-metastatic niche.
In
Vivo Efficacy of PRP In Pancreatic and Ovarian Tumors
The
effect of the pro-enzyme formulation PRP at different doses on tumor weight in orthotopically implanted pancreatic and ovary tumors was
evaluated. In the pancreatic tumor model, there was significant (*P < 0.05) reduction in mean tumor weight in animals treated for
26 days with trypsinogen/chymotrypsinogen at 83.3/500 mg/kg (30.2 mg; 85.9% inhibition) compared with control (PBS; 214.8 mg). Furthermore,
ovary tumor-bearing mice showed a significant (*P < 0.05) reduction in mean tumor weight in animals treated for 21 days with two different
doses of trypsinogen/chymotrypsinogen, 9.1/54 mg/kg and 27.5/165 mg/kg, compared with control (PBS). The mean weight of control group
tumors was 2062.2 mg while the treated groups presented a mean tumor weight of 1074.2 mg and 957.3 respectively, ranging in a 50% tumor
inhibition (52–46%).
Overview
Of Clinical Studies
The
clinical efficacy of a suppository formulation containing bovine pancreatic pro-enzymes trypsinogen and chymotrypsinogen was evaluated
in the context of a UK Pharmaceuticals Special Scheme and the results were published in a peer reviewed journal, Scientific Reports.
Clinical effects were studied in 46 patients with advanced metastatic cancers of different origin (prostate, breast, ovarian, pancreatic,
colorectal, stomach, non-small cell lung, bowel cancer and melanoma) after treatment with a rectal formulation of both pancreatic pro-enzymes.
No
severe or serious adverse events related to the rectal administration were observed. Patients did not experience any hematological side
effects as typically seen with classical chemotherapy regimens. No allergic reactions after rectal administration of suppositories were
observed.
In
order to assess the therapeutic activity of rectal administration, overall survival of patients under treatment was compared to the life
expectancy assigned to a patient prior to treatment start. Nineteen from 46 patients (41.3%) with advanced malignant diseases, most of
them suffering from metastases, had a survival time significantly longer than their expected, in fact, for the whole set of cancer types,
mean survival (9.0 months) was significantly higher than mean life expectation (5.6 months). Although the number of patients per cancer
indication is naturally quite low, 3 out of 8 patients with prostate cancer and 5 out of 11 patients with gastrointestinal cancers appear
to particularly benefit from the treatment with the proenzyme suppositories.
PRP
proves to be an in vivo effective and non-toxic anti-tumor treatment, able to inhibit angiogenesis and tumor growth, cancer cell migration
and invasiveness. Furthermore, a suppository formulation containing both pancreatic proenzymes increased the life expectancy of advanced
cancer patients. Consequently, PRP could have relevant oncological clinical applications for the treatment of solid tumors like advanced
pancreatic adenocarcinoma and advanced epithelial ovarian cancer.
| Cancer
Type |
|
Life
Expectation
(months) |
|
Survival
*
(months) |
| Pancreatic
carcinoma (n = 4) |
|
2 |
|
8 |
| |
4 |
|
* |
| |
<3 |
|
7 |
| |
<3 |
|
4 |
| Ovarian
Cancer (n = 7) |
|
4 |
|
11 |
| |
6 |
|
12 |
| |
6 |
|
11 |
| |
<12 |
|
38 |
| |
<1 |
|
1 |
| |
4 |
|
* |
| |
3 |
|
* |
| Breast
Cancer (n = 6) |
|
6 |
|
9 |
| |
6 |
|
* |
| |
2 |
|
* |
| |
12 |
|
* |
| |
<12 |
|
* |
| |
12 |
|
* |
| Colon
Rectal Cancer (n = 5) |
|
6 |
|
* |
| |
6 |
|
* |
| |
12 |
|
* |
| |
6 |
|
40 |
| |
12 |
|
* |
| Gastric
Cancer (n = 2) |
|
2 |
|
8 |
| |
<3 |
|
7 |
| Prostate
Cancer (n = 8) |
|
4 |
|
* |
| |
1 |
|
5 |
| |
4 |
|
* |
| |
<12 |
|
* |
| |
12 |
|
14 |
| |
12 |
|
* |
| |
12 |
|
* |
| |
12 |
|
* |
| Non-Hodgkin
Lymphoma (n = 1) |
|
2 |
|
9 |
| Mesothelioma
(n = 1) |
|
3 |
|
9 |
| Melanoma
(n = 2) |
|
6 |
|
* |
| |
<3 |
|
4 |
| Neuro-endocrine
Tumor (n = 1) |
|
10 |
|
24 |
| Bladder
(n = 2) |
|
<3 |
|
* |
| |
12 |
|
* |
| NSCLS
(n = 2) |
|
3 |
|
5 |
| |
6 |
|
* |
| Bowel
(n = 2) |
|
<12 |
|
* |
| |
<3 |
|
3 |
| Small
Cell Carcinoma (n = 1) |
|
<12 |
|
* |
| Renal
Cancer (n = 1) |
|
<3 |
|
* |
| Abdomen
unknown primary (n = 1) |
|
<12 |
|
* |
Overview
of clinical studies. Patients who met prognosis of life expectation (*). For the whole set of cancer types, mean survival (9.0 months)
was statistically significantly higher than mean life expectation (5.6 months). One way ANOVA (α = 0.05, P < 0.05).
POP1
JOINT RESEARCH AND DRUG DISCOVERY PROGRAM
To
date, both proenzymes were synthesized and purified in the laboratory. Once purified, the proenzymes were lyophilized (freeze dried)
and each formed a stable, dry white powder. The sequence of proteins of each proenzyme were then determined by mass spectrometry. Larger
quantities of the proenzymes were produced with the objective of establishing their combined anti-cancer effects against pancreatic and
colorectal cancers. In addition, research activities were transferred to the MEDINA Foundation Research Center to investigate the potential
to scale up production of the recombinant proenzymes from the expression of the novel expression system, which is currently ongoing.
MEDINA is a Non-Profit Research Organization established in 2008 through a public-private alliance between the Regional Government of
Andalusia, Spain, the pharmaceutical company Merck Sharp & Dohme de España S.A. (MSD), and the University of Granada. Medina’s
scientific platforms support the development of multidisciplinary research programs in Microbiology, Natural Product Chemistry and Screening
& Target Validation.
We
are elucidating the molecular pathways involved in the proenzymes anti-tumor efficacy and study how proenzymes interact with the pre-metastatic
tumor niche, focusing on the interaction and suppression of tumor associated cells, like cancer-associated fibroblasts and macrophages.
A pre-metastatic tumor niche is an environment in a secondary organ conducive to the metastasis of a primary tumor. Such a niche provides
favorable conditions for growth, and eventually metastasis, in an otherwise foreign and hostile environment for the primary tumor cells.
Metastasis remains the main cause of patient death from solid tumors for cancer sufferers.
To
achieve this, we are using integrated tumor models in a microfluidics chip by obtaining 3-dimensional bio-impression samples from patients
with advanced solid tumors, developed at the Centre for Biomedical Research, University of Granada, Granada, Spain. As well as explaining
the mechanism of action by which proenzymes exert their anti-cancer effects, it also confirms whether proenzymes penetrate into the tumor
microenvironment and exert their effects. At the same time, it confirms the selectivity of the drug on solid tumors, by targeting cancer
cells and leaving healthy cells alone.
To
date, our investigation has confirmed that proenzyme therapy is effective against cancer stem cells, which are the cells responsible
for the formation of secondary tumors through metastasis. They were also observed to be effective against cancer cells within the primary
tumor, whilst also leaving non-tumor cells alone. The final part of the investigation is to determine the effects of the proenzymes against
both these cancerous cell types within the tumor microenvironment.
Our
Joint Research Team successfully published data confirming the anti-tumor potential of a mixture of trypsinogen and chymotrypsinogen.
Treatment with proenzymes sensitizes cancer stem cells, which may allow standard treatment approaches like chemotherapy and radiotherapy
to be more effective.
Our
vision is to produce a backup product candidate to PRP which can further stabilize and enhance the effects of the proenzymes when administered
to patients. Our scientific researchers are in the process of optimizing conditions to achieve high titers of recombinant trypsinogen
and chymotrypsinogen with this expression system.
PRP
TARGET INDICATIONS
The
management of cancer differs widely, with a multitude of factors impacting the choice of treatment strategy. Some of those factors include:
| |
● |
the
type of tumor, usually defined by the tissue in the body from which it originated; |
| |
● |
the
extent to which it has spread beyond its original location; |
| |
● |
the
availability of treatments, driven by multiple factors including cost, drugs approved, local availability of suitable facilities,
etc.; |
| |
● |
regional
and geographic differences; |
| |
● |
whether
the primary tumor is amenable to surgery, either as a potentially curative procedure, or as a palliative one; and |
| |
● |
the
balance between potential risks and potential benefits from the various treatments and, probably most importantly, the patient’s
wishes. |
For
many patients with solid cancers, such as breast, ovarian, colorectal, lung and pancreatic cancer, surgery is frequently the first treatment
option, often followed by first line chemotherapy with or without radiotherapy. While hopefully such procedures are curative, in many
instances the tumor returns, and second line treatment strategies are chosen in an effort to achieve a degree of control over the tumor.
In most instances, the benefit is temporary, and eventually the point is reached where the patient’s tumor either fails to adequately
respond to treatment, or the treatment has unacceptable toxicity which severely limits its usefulness.
Should
the planned Phase I, II and III clinical trials confirm the efficacy of PRP, along with the favorable safety and tolerability profile
suggested by pre-clinical studies conducted to date, we believe our product will have utility in a number of clinical situations including:
| |
1. |
In
the early-stage management of solid tumors, most likely as part of a multi-pronged treatment strategy in combination with existing
therapeutic interventions; |
| |
2. |
As
a product that can be administered long term for patients following standard treatment approaches, such as surgery, or chemotherapy,
in order to prevent or delay recurrence; and |
| |
3. |
As
a preventative measure for patients at risk of developing cancer based on genetic screening. |
In
the near term as part of our planned Phase I, II and III clinical trials, we plan to target patients with solid tumors, most likely ovarian
and pancreatic, for whom other treatment options have been exhausted. This is a common approach by which most new drugs for cancer are
initially tested. Once efficacy and safety has been demonstrated in this patient population, exploration of the potential utility of
the drug in earlier stage disease can be undertaken, together with investigation of the drug’s utility in other types of cancers,
such as gastro-esophageal tumors, colon or rectal carcinoma might be conducted. A Phase II study in a back-up indication, such as advanced
therapy refractant prostate cancer will also be considered. This indication is based on positive preclinical pharmacology studies.
Pancreatic
Cancer
Pancreatic
cancer is one of the most lethal malignancies with a median survival of less than 6 months and a 5-year survival rate of less than 5%.
The lethal nature of this disease stems from its propensity to rapidly disseminate to the lymphatic system and distant organs. This aggressive
biology and resistance to conventional and targeted therapeutic agents leads to a typical clinical presentation of incurable disease
at the time of diagnosis.
Pancreatic
cancer has claimed notoriety over the last decades by proving to be one of the most recalcitrant solid tumors. As an indicator of its
lethality, pancreatic cancer accounts for less than 3% of new cancers diagnosed annually in developed countries, yet it is the third
leading cause of cancer related mortality.
Since
pancreatic cancer is an essentially fatal condition, disease duration is roughly equivalent with survival time. The median time of survival
of patients with pancreatic cancer depends on the extend of disease at the time of diagnosis and ranges from 11-20 months for patients
who qualified for surgical resection (Stage I/II), to 6-11 months for patients with locally advanced disease (Stage III), and only 2-6
months for patients with metastatic disease (Stage IV) (Amikura 1995, Richter 2003). Taking these low survival times into consideration,
the yearly incidence rates for pancreatic cancer are considered the more relevant measure for this disease.
Each
year the American Cancer Society estimates the numbers of new cancer cases and deaths that will occur in the United States in the current
year and compiles the most recent data on cancer incidence, mortality, and survival. Incidence data are collected by the National Cancer
Institute (NCI), the Centers for Disease Control and Prevention (CDC), and the North American Association of Central Cancer Registries
(NAACCR). In 2015, a total of more than 1,500,000 new cancer cases and more than 500,000 cancer deaths will occur in the United States.
Amongst these, a total of almost 50.000 new cases of pancreatic cancer (3.33% of new cancer cases) have been estimated, which will result
in more than 40,000 deaths (8% of cancer deaths). This means only 20% survival rate of patients diagnosed with pancreatic cancer.
Ovarian
Cancer
Ovarian
cancer is a generic term that can be used for any cancer involving the ovaries, arising from one of the several different cell types
of ovaries, including germ cells, specialized gonadal stromal cells and epithelial cells. Epithelial ovarian cancer accounts for 90 percent
of ovarian cancers and is responsible for most ovarian cancer related deaths. Furthermore, several subtypes of ovarian cancer have been
described according to different risk factors, different genetic mutations, different biological behaviors and different prognoses. This
heterogeneity of the disease has impeded progress in the prevention, early detection, treatment and management of ovarian cancer.
Ovarian
cancer is the seventh most commonly diagnosed cancer among women in the world and accounts for an estimated 239,000 new cases and 152,000
deaths worldwide annually, of which 21,290 new cases and 14,180 related deaths are estimated to occur in the USA alone. The disease typically
presents at late stage when the 5-year relative survival rate is only 29%. Few cases (15%) are diagnosed with localized tumor (stage
1), when the 5-year survival rate is 92%. Strikingly, the overall 5-year relative survival rate generally ranges between 30%–40%
across the globe and has seen only very modest increases since 1995.
PRP
DEVELOPMENT STRATEGY
Our
goal is to undertake early-stage clinical development of PRP through to a significant value inflection point, where the commercial attractiveness
of a drug in development, together with a greater likelihood of achieving market authorization, may attract potential interest from licensees
seeking to acquire new products. Such value inflection points in the context of cancer drugs are typically at the point where formal,
controlled clinical trials have demonstrated either ‘efficacy’ or ‘proof of concept’ – typically meaning
that there is controlled clinical trial evidence that the drug is effective in the proposed target patient population, has an acceptable
safety profile, and is suitable for further development. From a ‘big picture’ perspective, it is our intention to progress
the development of our technology through the completion of our planned Phase IIa clinical trials and then to seek a licensee for further
development beyond that point.
As
part of that commercial strategy, we will:
| |
● |
continue
research and development to build our existing intellectual property portfolio, and to seek new, patentable discoveries; |
| |
● |
seek
to ensure all product development is undertaken in a manner that makes its products approvable in the major pharmaceutical markets,
including the U.S., Europe, the UK, Australia and Japan; |
| |
● |
aggressively
pursue the protection of our technology through all means possible, including patents in all major jurisdictions, and potentially
trade secrets; and |
| |
● |
make
strategic acquisitions to acquire new companies that have intellectual property or products that complement our future goals. |
PRP
DEVELOPMENT PLAN AND MILESTONES
We
plan to progress PRP down a conventional early-stage clinical development pathway for:
| |
● |
regulatory
and/or ethics approval to conduct a Phase Ib study; and |
| |
● |
Phase
IIa multiple escalating dose studies to investigate the safety, tolerability, and pharmacokinetics of PRP administered intravenously
to patients. |
Preclinical
development has been completed, including pharmacology and safety toxicology studies, process development activities and bioanalytical
method development. The full-scale GMP (Good Manufacturing Practice) finished product manufacture of PRP will be completed in preparation
for the FIH Phase Ib study. Validation of the bioanalytical method will also be completed prior to lodging our first clinical trial application
(CTA) which we plan to undertake at the Peter Mac Cancer Center in Melbourne, Victoria, Australia’s biggest cancer hospital. Propanc
Biopharma is collaborating with contract research organizations, manufacturing partners and consultants to complete activities prior
to preparing the CTA for the Phase Ib study.
The
Company has received expressions of interest to evaluate proenzyme therapy as a method to prevent recurrence and metastasis of solid
tumors in pancreatic and ovarian cancers. The letters of interest were confirmed by medical oncologists specializing in pancreatic and
ovarian cancers, from the University Hospital of Jaén, in Granada, Spain. The evaluation will most likely be conducted as separate
Phase IIa proof of concept (POC), multi-trial center studies for each target indication. The expressions of interest were confirmed after
their evaluation of Propanc’s scientific literature supporting the use of proenzymes in pancreatic and ovarian cancers. The Phase
IIa POC studies will be conducted after the Phase Ib dose escalation study investigating the tolerability and activity of proenzyme therapy
in patients with advanced solid tumors is completed at the Peter Mac Cancer Center.
In
Australia, we receive up to 43.5% “cash-back” benefit in the form of a refund of their qualified research and development
costs and expenses. The Company received a refund of $151,767 AUD ($113,415 USD) and $199,834 AUD ($134,728 USD) for the years ended
June 30, 2021 and 2020 respectively. We are continuing to evaluate all options to conduct our planned clinical trials in the most cost-efficient
manner, while striving to minimize dilution to our stockholders.
We
anticipate reaching the Phase IIa proof of concept milestone in approximately three to four years, subject to regulatory approval in
Europe, and the results from our research and development and licensing activities.
Our
overhead and expenses are likely to increase from its current level as PRP progresses down the development pathway. This increase will
be driven by the need to increase our internal resources in order to effectively manage our research and development activities.
Anticipated
timelines
In
second quarter of 2022 calendar year, we anticipate the submission of the Clinical Trial Application for PRP. We anticipate receiving
approval of such application in the first half of 2022. Following the clinical trial application, we plan to commence our Study Preparation,
including CRO Selection and Contracts, Analytical Lab Selection Contracts and Trial Sites Selection and Contracts. In connection with
the Clinical Trial Application, this product will be part of our Investigation Medicinal Product Dossier, Study Protocol and Investigator’s
Brochure. In the second half of 2022 calendar year, we hope to complete the Study Preparation together with the Preparation of Logistics
and Trial Sites Initiation Visits and complete our clinical trial application review. Commencing in the second half of 2022 calendar
year, we intend to initiate a Phase Ib study in advanced cancer patients with solid tumors and the anticipated costs will be approximately
$6.5 million. We will need to raise additional financing to fund our planned Phase I, II and III clinical trials and for working capital.
| Research Activity |
|
Timeline |
| Clinical Trial Application (CTA) |
|
|
| Investigational Medicinal Product Dossier |
|
November 2021 - April 2022 |
| Phase Ib Clinical Study Protocol |
|
|
| Investigator’s Brochure |
|
|
| CTA Compilation |
|
March - May 2022 |
| CTA submission |
|
May 2022 |
| CTA Approval |
|
June 2022 |
| CTA Review |
|
June 2022 – July 2022 |
| Contract Research Organization & Contracts |
|
|
| Analytical Laboratory Selection & Contracts |
|
January – May 2022 |
| Trial Site Selection & Contracts |
|
|
| Preparation of Logistics |
|
|
| Trial Site Initiation Visits |
|
May – September 2022 |
| First Patient/First Visit |
|
September 2022 |
POP1
JOINT RESEARCH AND DRUG DISCOVERY PROGRAM
As
outlined previously, a joint research and drug discovery program has been established with our collaborators at the Universities of Jaén
and Granada to investigate the changes in genetic and protein expression that occur in cancer cells as a consequence of being exposed
to our proenzyme formulation. The objective of this work is to understand at the molecular level the targets of our proenzyme formulation,
thereby providing the opportunity for new, patentable drugs which can be developed further. We plan to commence a targeted drug discovery
program utilizing the identified molecular target to search for novel anticancer agents.
One
specific objective of the project is to synthesize both proenzymes by an in vivo system to produce crystalized proteins that could be
maintained for long periods without suffering degradation, even in absence of refrigeration. This will be particularly useful for a longer
shelf life as well as global distribution of the drug product, particularly in warmer climates and developing regions where refrigeration
may not be available.
The
POP1 joint research and drug discovery program has produced synthetic recombinant versions of the two proenzymes, trypsinogen and chymotrypsinogen.
Propanc Biopharma’s joint scientific researchers are developing a novel expression system and are also in the process of optimizing
conditions to achieve high titers of recombinant trypsinogen and chymotrypsinogen. Further, the anticancer effects of the synthetic versions
will be tested against the naturally derived proenzymes from bovine origin.
FINANCIAL
OBJECTIVES
Multiple
factors, many of which are outside of our control, can impact our ability to achieve our target objectives within the planned time and
budgetary constraints. Subject to these caveats, our objective is to complete our planned Phase IIa study for PRP within the proposed
budget.
We
primarily outsource services, skills and expertise to third parties as required to achieve our scientific and corporate objectives. As
the business grows and gains more personnel, outsourcing will continue to be the preferred model, where fixed and variable costs are
carefully managed on a project-by-project basis. This means our research and development activities are carried out by third parties.
Additional third parties with specific expertise in research, compound screening and manufacturing (including raw material suppliers)
have been contracted as required.
CORPORATE
STRATEGY
Our
initial focus is to organize, coordinate and finance the various parts of our drug development pipeline. New personnel will be carefully
introduced into our Company over a period of time as our research and development activities expand. They will have specific expertise
in product development, manufacture and formulation, regulatory affairs, toxicology, clinical operations and business development (including
intellectual property management, licensing and other corporate activities). In the first instance, additional clinical management and
development expertise is likely to be required for our lead product. Therefore, we anticipate an increase in employees in order to effectively
manage our contractors as the projects progress down the development pathway.
This
outsourcing strategy is common in the biotechnology sector and is an efficient way to obtain access to the necessary skills required
to progress a project, in particular as the required skills change as the project progresses from discovery, through manufacturing and
non-clinical development and into clinical trials. We anticipate that we will continue to use this model, thereby retaining the flexibility
to contract in the appropriate resource as and when required.
We
intend to seek and identify potential licensing partners for our product candidates as they progress through the various development
stages, reaching certain milestones and value inflection points. If a suitable licensee is identified, a potential licensing deal could
consist of payments for certain milestones, plus royalties from future sales if the product is able to receive approval from the relevant
regulatory authorities where future product sales are targeted. We intend to seek and identify potential licensees based on the initial
efficacy data from Phase II clinical trials. To accomplish this objective, we have commenced discussions with potential partners in our
current preclinical phase of development.
As
part of our overall expansion strategy, from time to time, we investigate potential intellectual property acquisition opportunities to
expand our product portfolio. While our initial focus is on the development of PRP as the lead product candidate, potential product candidates
may also be considered for future preclinical and clinical development. These potential opportunities have arisen from other research
and development organizations, which either own existing intellectual property or are currently developing new intellectual property,
which may be of interest to us. These opportunities are possible new cancer treatments that are potentially less toxic than existing
treatment approaches and are able to fill an existing gap in the treatment process, such as a systemic de-bulking method which could
reduce the size and threat of metastases to a more manageable level for late-stage cancer patients.
We
believe these potential treatment approaches will be complementary to existing treatment regimens and our existing product candidate,
PRP. No formal approaches have been made at this stage and it is unknown whether we will engage in this discussion in the near future.
However, as PRP progresses further down the development pathway, we intend to assess future opportunities that may arise to use the expertise
of our management and scientific personnel for future prospective research and development projects.
CURRENT
OPERATIONS
We
are at a pre-revenue stage. We do not know when, if ever, we will be able to commercialize our products and begin generating revenue.
We are focusing our efforts on organizing, coordinating and financing the various aspects of the drug research and development program
outlined earlier in this document. In order to commercialize our products, we must complete preclinical development, Phase Ib, IIa and
IIb clinical trials in Europe, the U.S., United Kingdom, Australia or elsewhere, and satisfy the applicable regulatory authority that
PRP is safe and effective. If the results from the Phase II trials are convincing, we will seek conditional approval from the regulatory
authorities sooner. Therefore, from the time we commence clinical trials, we estimate that this will take approximately three to four
years if we seek conditional approval upon completion of Phase II trials. As described previously, when we advance our development projects
sufficiently down the development pathway and achieve a major increase in value, such as obtaining interim efficacy data from Phase II
clinical trials, we will seek a suitable licensing partner to complete the remaining development activities, obtain regulatory approval
and market the product.
CURRENT
THERAPIES
We
are developing a therapeutic solution for the treatment of patients with advanced stages of cancer targeting solid tumors, which is cancer
that originates in organs or tissues other than bone marrow or the lymph system. Common cancer types classified as solid tumors include
lung, colorectal, ovarian cancer, pancreatic cancer and liver cancers. In each of these indications, there is a large market opportunity
to capitalize on the limitations of current therapies.
Current
therapeutic options for the treatment of cancer offer, at most, a few months of extra life or tumor stabilization. Some experts believe
that drugs that kill most tumor cells do not affect cancer stem cells, which can regenerate the tumor (e.g., chemotherapy). Studies are
revealing the genetic changes in cells that cause cancer and spur its growth. This research is providing scientific researchers with
many potential targets for drugs. Tumor cells, however, can develop resistance to drugs.
Limitations
of Current Therapies
PRP
was developed because of the limitation of current cancer therapies. While surgery is often safe and effective for early-stage cancer,
many standard therapies for late-stage cancer urgently need improvement; current treatments generally provide modest benefits, and frequently
cause significant adverse effects. Our focus is to provide oncologists and their patients with therapies for metastatic cancer which
are more effective than current therapies, and which have a substantially reduced side effect profile.
While
progress has been made within the oncology sector in developing new treatments, the overall cancer death rate has only improved by 7%
over the last 30 years.
Most
of these new treatments have some limitations, such as:
| |
1. |
significant
toxic effects; |
| |
2. |
expense;
and |
| |
3. |
limited
survival benefits. |
We
believe that our treatment will provide a competitive advantage over the following treatments:
| |
● |
Chemotherapeutics:
Side effects from chemotherapy can include pain, diarrhea, constipation, mouth sores, hair loss, nausea and vomiting, as well as
blood-related side effects, which may include a low cell count of infection fighting white blood cells (neutropenia), low red blood
cell count (anemia), and low platelet count (thrombocytopenia). Our goal is to demonstrate that our treatment will be more effective
than chemotherapeutic and hormonal therapies with fewer side effects. |
| |
● |
Targeted
therapies: The most common type is multi-targeted kinase inhibitors (molecules which inhibit a specific class of enzymes called kinases).
Common side effects include fatigue, rash, hand–foot reaction, diarrhea, hypertension and dyspnea (shortness of breath). Further,
tyrosine kinases inhibited by these drugs appear to develop resistance to inhibitors. While the clinical findings with PRP are early
and subject to confirmation in future clinical trials, no evidence has yet been observed of the development of resistance by the
cancer to PRP. |
| |
● |
Monoclonal
antibodies: Development of monoclonal antibodies is often difficult due to safety concerns. Side effects that are most common include
skin and gastro-intestinal toxicities. For example, several serious side effects from Avastin, an anti-angiogenic cancer drug, include
gastrointestinal perforation and dehiscence (e.g., rupture of the bowel), severe hypertension (often requiring emergency treatment)
and nephrotic syndrome (protein leakage into the urine). Antibody therapy can be applied to various cancer types, but can also be
limited to certain genetic sub populations in many instances. |
| |
● |
Immunotherapy:
There is a long history of attempts to develop therapeutic cancer vaccines to stimulate the body’s own immune system to attack
cancer cells. While these products generally do not have the poor safety profile of standard therapeutic approaches, only a small
number of them are FDA-approved and available compared to the number of patients diagnosed with cancer. Furthermore, only a relatively
small number of the patient population is eligible to receive and subsequently respond to treatment, as defined by preventing tumor
growth. |
MARKET
OPPORTUNITY
The
global metastatic cancer treatment market is predicted to reach $111 Billion by 2027 by Emergen Research. Demand for new cancer products
can largely be attributed to a combination of a rapidly aging population in western countries and changing environmental factors, which
together are resulting in rising cancer incidence rates. Worldwide, the World Health Organization estimated 19.3 million new cancer cases
and almost 10.0 million cancer deaths occurred in 2020. As such, global demand for new cancer treatments which are effective, safe and
easy to administer is rapidly increasing. Our treatment will potentially target many aggressive tumor types for which little or few treatment
options exist.
We
plan to target patients with solid tumors, most likely pancreatic and ovarian tumors, for whom other treatment options have been exhausted.
Globally these cancers resulted in over 673,255 deaths in 2020, according to the World Health Organization. With such a high mortality
rate, a substantial unmet medical need exists for new treatments. Once the efficacy and safety of PRP has been demonstrated in late-stage
patient populations, we plan to undertake exploration of the utility of the drug in earlier stage disease, together with investigation
of the drug’s utility in other types of cancer.
Anticipated
Market Potential
It
is difficult to estimate the size of the market opportunity for this specific type of product as a clinically proven, pro-enzyme formulated
suppository marketed to oncologists across global territories for specific cancer indications, to the best of management’s knowledge,
has not been previously available. However, the markets for potential market for pancreatic and ovarian cancers may be characterized
as follows:
| |
● |
The
world market for pancreatic cancer drugs is projected to grow to $4.2 billion by the year 2025, according to Grandview Research.
Major players operating in the pancreatic cancer therapy market include Eli Lilly and Company, F. Hoffmann-La Roche AG, Celgene Corporation,
Amgen Inc., Novartis AG, Pharmacyte Biotech Inc., Clovis Oncology, Teva Pharmaceutical Industries Ltd., Pfizer Inc., Merck &
Co., Inc. among others. For instance, in May 2018, Eli Lilly and Company acquired AMRO BioSciences. AMRO BioSciences is engaged into
number of drugs for cancer. The clinical trial explores a drug (pegilodecakin) which is ongoing for the pancreatic cancer. The developments
performed by the companies are helping the market to grow in the coming years. |
| |
● |
The
global market for ovarian cancer drugs expected to reach $10.1 billion by 2027, according to iHealthcareAnalyst. This will be driven
by continued uptake and expected launches of the approved PARP (poly adenosine diphosphate-ribose polymerase) inhibitors. Major competitors
operating in the global ovarian cancer treatment market include AbbVie, Inc., AstraZeneca plc (Acerta Pharma), Boehringer Ingelheim
GmbH, Chugai Pharmaceutical Co., Ltd., Clovis Oncology, Five Prime Therapeutics, Inc., GlaxoSmithKline plc (Tesaro), Gradalis, Inc.,
Incyte Corporation, MacroGenics, Inc., Mateon Therapeutics, Inc., Merck & Co., Inc., Novartis AG, Novogen Limited, Oasmia Pharmaceuticals,
Inc., Pfizer, Inc., PharmaMar S.A, and Roche Holding AG. |
New
products can be defined as addition-in-class, advance-in-class, or first-in-class, depending on their degree of innovation. Addition-in-class
products, defined as new Active Pharmaceutical Ingredients (API) with established mechanisms of action, are often clinically important
and highly commercially successful. Advance-in-class product innovation, defined as significantly differentiated and innovative new APIs,
albeit with established mechanisms of action, remains a highly attractive strategy. However, first-in-class innovation, defined as products
with a molecular target and/or mechanism of action not found in any approved products globally, remains the key product development strategy
in terms of providing the greatest degree of differentiation, extending to a first-mover advantage and potentially the capture of significant
market share.
Based
on the current situation for these two markets, we believe there is an attractive opportunity in both the pancreatic and ovarian cancer
market sectors for the introduction of a first-in-class, clinically proven product which can achieve new benefits for patients in terms
of survival and quality of life. The current concentration of products suggests oncologists may be willing to try newly approved products,
particularly if they can exhibit a favorable safety profile, although substantive R&D activities will be necessary to both obtain
regulatory approval, and to generate the clinical safety and efficacy data needed to convince clinicians to use a new product.
LICENSE
AGREEMENTS
University
of Bath
We
previously sponsored a collaborative research project at University of Bath to investigate the cellular and molecular mechanisms underlying
the potential clinical approach of our proprietary proenzyme formulation. As a result of this undertaking, we entered into a Commercialization
Agreement with University of Bath (UK), dated November 12, 2009 (the “Commercialization Agreement”), where, initially, we
held an exclusive license with Bath University, and where we and University of Bath co-owned the intellectual property relating to our
proenzyme formulations. The Commercialization Agreement originally provided for University of Bath to assign the Patents (as defined
therein) to Propanc in certain specified circumstances, such as successful completion of a clinical trial and commencement of a Phase
II (Proof of Concept) clinical trial.
On
June 14, 2012, Propanc and University of Bath agreed to an earlier assignment to us of the patents pursuant to an Assignment and Amendment
Deed, on the provision that Bath University retains certain rights arising from the Commercialization Agreement, as follows:
| |
● |
University
of Bath reserves for itself (and its employees and students and permitted academic sub-licensees with respect to research use) the
non-exclusive, irrevocable, worldwide, royalty free right to use the patents for research use; |
| |
● |
The
publication rights of University of Bath specified in the contract relating to the original research made between the parties with
an effective date of July 18, 2008 shall continue in force; |
| |
● |
Propanc
shall pay to University of Bath a royalty of two percent of any and all net revenues; |
| |
● |
Propanc
shall use all reasonable endeavors to develop and commercially exploit the patents for the mutual benefit of University of Bath and
Propanc to the maximum extent throughout the covered territory and in any additional territory and to obtain, maintain and/or renew
any licenses or authorizations that are necessary to enable such development and commercial exploitation. Without prejudice to the
generality of the foregoing, Propanc shall comply with all relevant regulatory requirements in respect of its sponsoring and/or performing
clinical trials in humans involving the administration of a product or materials within a claim of the patents; and |
| |
● |
Propanc
shall take out with a reputable insurance company and maintain liability insurance coverage prior to the first human trials. |
In
consideration of such assignment, we agreed to pay royalties of 2% of net revenues to University of Bath. Additionally, we agreed to
pay 5% of each and every license agreement subscribed for. The contract is cancellable at any time by either party. To date, no amounts
are owed under the agreement.
University
of Jaén
We
have established a collaboration with the University of Jaén to carry out a Research Project aimed at the synthetic development
of PRP and its subsequent validation. The University of Jaén is providing scientific research activities the Department of Health
Sciences, which provides the necessary technical and human resources in order to carry out the programmed works. A Collaboration Agreement
(the “Collaboration Agreement”) according was established, dated October 1, 2020, with the main objective for the synthetic
development of PRP and its subsequent validation. To that end, there shall be established a pre-clinical protocol of safety evaluation
and antitumor efficacy on cancer stem cells and in orthotopic xenotransplantations derived from cancer stem cells isolated from tumor
cell lines, of a newly developed synthetic formulation based on the two pancreatic zymogens.
The
ownership of potential intellectual property rights that may arise as a result of the knowledge obtained through the project will belong
to Propanc. In consideration for payment of the compensation, the University of Jaén hereby assigns and agrees to do all things
reasonably required to assign to the contracting entity all industrial property rights arising from the Project.
In
return for ownership of the entire right and title in all industrial property rights arising from the Project, Propanc agrees to pay
the University of Jaén two percent (2%) of the net sales of any products sold by the contracting entity which fall within the
scope of the protection of such industrial property rights.
Future
Agreements
We
continue to learn the properties of proenzymes with the long-term aim of screening new compounds for development. We anticipate engaging
in future discussions with several technology companies who are progressing new developments in the oncology field as potential additions
to our product line. Initially targeting the oncology sector, our focus is to identify and develop novel treatments that are highly effective
targeted therapies, with few side effects as a result of toxicity to healthy cells.
INTELLECTUAL
PROPERTY
The
Company has filed multiple patent applications relating to its lead product, PRP. The Company’s lead patent application has been
granted and remains in force in the United States, Belgium, Czech Republic, Denmark, France, Germany, Ireland, Italy, Netherlands, Portugal,
Spain, Sweden, Switzerland, Liechtenstein, Turkey, United Kingdom, Australia, China, Japan, Indonesia, Israel, New Zealand, Singapore,
Malaysia, South Africa, Mexico, Republic of Korea, India and Brazil. In Canada, the patent application remains under examination.
In
2016 and early 2017, we filed other patent applications. Three applications were filed under the Patent Cooperation Treaty (the “PCT”).
The PCT assists applicants in seeking patent protection by filing one international patent application under the PCT, applicants can
simultaneously seek protection for an invention in over 150 countries. Once filed, the application is placed under the control of the
national or regional patent offices, as applicable, in what is called the national phase. One of the PCT applications filed in November
2016, entered national phase in July 2018 and another PCT application entered national phase in August 2018. A third PCT application
entered national phase in October 2018.
Presently,
there are 35 granted patents and 30 patents under examination in key global jurisdictions relating to the use of proenzymes against solid
tumors, covering the lead product candidate PRP.
Further
patent applications are expected to be filed to capture and protect additional patentable subject matter based on the Company’s
field of technology relating to pharmaceutical compositions of proenzymes for treating cancer.
REGULATORY
MATTERS
United
States
Government
oversight of the pharmaceutical industry is usually classified into pre-approval and post-approval categories. Most of the therapeutically
significant innovative products marketed today are the subject of New Drug Applications (“NDA”). Preapproval activities,
based on these detailed applications, are used to assure the product is safe and effective before marketing. In the United States, The
Center for Drug Evaluation and Research (“CDER”), is the FDA organization responsible for over-the- counter and prescription
drugs, including most biological therapeutics, and generic drugs.
Before
approval, the FDA may inspect and audit the development facilities, planned production facilities, clinical trials, institutional review
boards and laboratory facilities in which the product was tested in animals. After the product is approved and marketed, the FDA uses
different mechanisms for assuring that firms adhere to the terms and conditions of approval described in the application and that the
product is manufactured in a consistent and controlled manner. This is done by periodic unannounced inspections of production and quality
control facilities by FDA’s field investigators and analysts.
Federal
Food, Drug and Cosmetic Act and Public Health Service Act
Prescription
drug and biologic products are subject to extensive pre- and post-market regulation by the FDA, including regulations that govern the
testing, manufacturing, safety, efficacy, labelling, storage, record keeping, advertising and promotion of such products under the Federal
Food, Drug and Cosmetic Act, the Public Health Service Act, and their implementing regulations. The process of obtaining FDA approval
and achieving and maintaining compliance with applicable laws and regulations requires the expenditure of substantial time and financial
resources. Failure to comply with applicable FDA or other requirements may result in refusal to approve pending applications, a clinical
hold, warning letters, civil or criminal penalties, recall or seizure of products, partial or total suspension of production or withdrawal
of the product from the market. FDA approval is required before any new drug or biologic, including a new use of a previously approved
drug, can be marketed in the United States. All applications for FDA approval must contain, among other things, information relating
to safety and efficacy, stability, manufacturing, processing, packaging, labelling and quality control.
New
Drug Applications (“NDAs”)
The
FDA’s NDA approval process generally involves:
| |
● |
Completion
of preclinical laboratory and animal testing in compliance with the FDA’s good laboratory practice, or GLP, regulations; |
| |
● |
Submission
to the FDA of an investigational new drug (“IND”) application for human clinical testing, which must become effective
before human clinical trials may begin in the United States; |
| |
● |
Performance
of adequate and well-controlled human clinical trials to establish the safety, purity and potency of the proposed product for each
intended use; |
| |
● |
Satisfactory
completion of an FDA pre-approval inspection of the facility or facilities at which the product is manufactured to assess compliance
with the FDA’s “current good manufacturing practice” (“CGMP”) regulations; and |
| |
● |
Submission
to and approval by the FDA of an NDA. |
The
preclinical and clinical testing and approval process requires substantial time, effort and financial resources, and we cannot guarantee
that any approvals for our product candidates will be granted on a timely basis, if at all. Preclinical tests include laboratory evaluation
of toxicity and immunogenicity in animals. The results of preclinical tests, together with manufacturing information and analytical data,
are submitted as part of an IND application to the FDA. The IND automatically becomes effective 30 days after receipt by the FDA, unless
the FDA raises concerns or questions about the conduct of the clinical trial, including concerns that human research subjects will be
exposed to unreasonable health risks. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before clinical
trials can begin. Our submission of an IND may not result in FDA authorization to commence clinical trials. A separate submission to
an existing IND must also be made for each successive clinical trial conducted during product development. Further, an independent institutional
review board (“IRB”) covering each medical center proposing to conduct clinical trials must review and approve the plan for
any clinical trial before it commences at that center and it must monitor the study until completed. The FDA, the IRB or the sponsor
may suspend a clinical trial at any time on various grounds, including a finding that the subjects or patients are being exposed to an
unacceptable health risk. Clinical testing also must satisfy extensive “good clinical practice” (“GCP”) regulations,
which include requirements that all research subjects provide informed consent and that all clinical studies be conducted under the supervision
of one or more qualified investigators.
For
purposes of an NDA submission and approval, human clinical trials are typically conducted in the following sequential phases, which may
overlap:
| |
● |
Phase
I: Initially conducted in a limited population to test the product candidate for safety and dose tolerance; |
| |
● |
Phase
II: Generally conducted in a limited patient population to identify possible adverse effects and safety risks, to determine the initial
efficacy of the product for specific targeted indications and to determine optimal dosage. A Phase IIa trial is a non-pivotal, exploratory
study that assesses biological activity as its primary endpoint. A Phase IIb trial is designed as a definite dose finding study with
efficacy as the primary endpoint. Multiple Phase II clinical trials may be conducted by the sponsor to obtain information prior to
beginning larger and more extensive Phase III clinical trials; |
| |
● |
Phase
III: Commonly referred to as pivotal studies. When Phase II evaluations demonstrate that a dose range of the product is effective
and has an acceptable safety profile, Phase III clinical trials are undertaken in large patient populations to further evaluate dosage,
to provide substantial evidence of clinical efficacy and to further test for safety in an expanded and diverse patient population
at multiple, geographically-dispersed clinical trial sites. Generally, replicate evidence of safety and effectiveness needs to be
demonstrated in two adequate and well-controlled Phase III clinical trials of a product candidate for a specific indication. These
studies are intended to establish the overall risk/benefit ratio of the product and provide adequate basis for product labelling;
and |
| |
● |
Phase
IV: In some cases, the FDA may condition approval of a NDA on the sponsor’s agreement to conduct additional clinical trials
to further assess the product’s safety, purity and potency after NDA approval. Such post-approval trials are typically referred
to as Phase IV clinical trials. |
Progress
reports detailing the results of the clinical studies must be submitted at least annually to the FDA and safety reports must be submitted
to the FDA and the investigators for serious and unexpected adverse events. Concurrent with clinical studies, sponsors usually complete
additional animal studies and must also develop additional information about the product and finalize a process for manufacturing the
product in commercial quantities in accordance with CGMP requirements. The manufacturing process must be capable of consistently producing
quality batches of the product candidate and, among other things, the manufacturer must develop methods for testing the identity, strength,
quality and purity of the final product. Moreover, appropriate packaging must be selected and tested, and stability studies must be conducted
to demonstrate that the product candidate does not undergo unacceptable deterioration over its shelf life.
The
results of product development, preclinical studies and clinical trials, along with the aforementioned manufacturing information, are
submitted to the FDA as part of a NDA. NDA’s must also contain extensive manufacturing information. Under the Prescription Drug
User Fee Act (“PDUFA”), the FDA agrees to specific goals for NDA review time through a two-tiered classification system,
Standard Review and Priority Review. Standard Review is applied to products that offer at most, only minor improvement over existing
marketed therapies. Standard Review NDAs have a goal of being completed within a ten-month timeframe, although a review can take significantly
longer. A Priority Review designation is given to products that offer major advances in treatment, or provide a treatment where no adequate
therapy exists. A Priority Review takes the FDA six months to review a NDA. It is likely that our product candidates will be granted
Standard Reviews. The review process is often significantly extended by FDA requests for additional information or clarification. The
FDA may refer the application to an advisory committee for review, evaluation and recommendation as to whether the application should
be approved. The FDA is not bound by the recommendation of an advisory committee, but it generally follows such recommendations.
The
FDA may deny approval of a NDA if the applicable regulatory criteria are not satisfied, or it may require additional clinical data or
additional pivotal Phase III clinical trials. Even if such data is submitted, the FDA may ultimately decide that the NDA does not satisfy
the criteria for approval. Data from clinical trials is not always conclusive and the FDA may interpret data differently than Propanc.
Once issued, product approval may be withdrawn by the FDA if ongoing regulatory requirements are not met or if safety problems occur
after the product reaches the market. In addition, the FDA may require testing, including Phase IV clinical trials, Risk Evaluation and
Mitigation Strategies (“REMS”), and surveillance programs to monitor the effect of approved products that have been commercialized,
and the FDA has the power to prevent or limit further marketing of a product based on the results of these post-marketing programs. Products
may be marketed only for the approved indications and in accordance with the provisions of the approved label. Further, if there are
any modifications to the drug, including changes in indications, labelling or manufacturing processes or facilities, approval of a new
or supplemental NDA may be required, which may involve conducting additional preclinical studies and clinical trials.
Other
U.S. Regulatory Requirements
After
approval, products are subject to extensive continuing regulation by the FDA, which include company obligations to manufacture products
in accordance with GMP, maintain and provide to the FDA updated safety and efficacy information, report adverse experiences with the
product, keep certain records, submit periodic reports, obtain FDA approval of certain manufacturing or labeling changes and comply with
FDA promotion and advertising requirements and restrictions. Failure to meet these obligations can result in various adverse consequences,
both voluntary and FDA-imposed, including product recalls, withdrawal of approval, restrictions on marketing and the imposition of civil
fines and criminal penalties. In addition, later discovery of previously unknown safety or efficacy issues may result in restrictions
on the product, manufacturer or NDA holder.
Propanc,
and any manufacturers of our products, are required to comply with applicable FDA manufacturing requirements contained in the FDA’s
GMP regulations. GMP regulations require, among other things, quality control and quality assurance as well as the corresponding maintenance
of records and documentation. The manufacturing facilities for our products must meet GMP requirements to the satisfaction of the FDA
pursuant to a pre-approval inspection before Propanc can use them to manufacture products. Propanc and any third-party manufacturers
are also subject to periodic inspections of facilities by the FDA and other authorities, including procedures and operations used in
the testing and manufacture of our products to assess our compliance with applicable regulations.
With
respect to post-market product advertising and promotion, the FDA imposes complex regulations on entities that advertise and promote
pharmaceuticals, which include, among others, standards for direct-to-consumer advertising, promoting products for uses or in patient
populations that are not described in the product’s approved labeling (known as “off-label use”), industry-sponsored
scientific and educational activities and promotional activities involving the Internet. Failure to comply with FDA requirements can
have negative consequences, including adverse publicity, enforcement letters from the FDA, mandated corrective advertising or communications
with doctors and civil or criminal penalties. Although physicians may prescribe legally available drugs for off-label uses, manufacturers
may not market or promote such off-label uses.
Changes
to some of the conditions established in an approved application, including changes in indications, labeling, or manufacturing processes
or facilities, require submission and FDA approval of a new NDA or NDA supplement before the change can be implemented. A NDA supplement
for a new indication typically requires clinical data similar to that in the original application, and the FDA uses the same procedures
and actions in reviewing NDA supplements as it does in reviewing a NDA.
Adverse
event reporting and submission of periodic reports is required following FDA approval of a NDA. The FDA also may require post-marketing
testing, known as Phase IV testing, risk mitigation strategies and surveillance to monitor the effects of an approved product or to place
conditions on an approval that could restrict the distribution or use of the product.
In
June 2017, we were notified by the FDA that PRP had been granted orphan drug designation for the treatment of pancreatic cancer. Orphan
drug designation may be granted by the FDA when a rare disease or condition is implicated and a potential treatment qualifies under the
Orphan Drug Act and applicable FDA regulations. This qualifies us for various developmental incentives, including protocol assistance,
the potential for research grants, the waiver of future application fees, and tax credits for clinical testing if we choose to host future
clinical trials in the United States.
In
October 2017, we submitted a request for a second orphan drug designation for PRP, this time for ovarian cancer.
On
November 2, 2017, we were notified by the FDA that our request was not granted. The Office of Orphan Products Development (“OOPD”)
stated that complete prevalence is used as a measure of disease in ovarian cancer, as this reflects the number of women who have been
diagnosed with disease and may be eligible for treatment with the proposed therapy. Therefore, on the date of the submission of our application,
the OOPD estimated that the prevalence of ovarian cancer was 228,110 cases. Since the prevalence exceeds the threshold of 200,000 to
qualify for orphan drug designation, they could not grant our request. We may consider resubmitting our application if we can identify
a suitable sub population in ovarian cancer, which may meet the target threshold.
European
Union
In
addition to regulations in the United States, we will be subject to a variety of foreign regulations governing clinical trials, commercial
sales and distribution of our products. Whether or not we obtain FDA approval for a product, we must obtain approval of a product by
the comparable regulatory authorities of foreign countries before we can commence clinical trials or market our product in those countries.
The approval process varies from country to country and the time may differ than that required for FDA approval. The requirements governing
the conduct of clinical trials, product licensing, pricing and reimbursement vary greatly from country to country. Despite these differences,
the clinical trials will be conducted according to international standards such as Good Clinical Practice (GCP), Good Manufacturing Practice
(GMP) and Good Laboratory Practice (GLP), which is recognized by each foreign country under the International Conference of Harmonization
(ICH) Guidelines. We will conduct our trials in each foreign jurisdiction according to these standards, undertaking a First-In-Human
(FIH) Phase I study in patients with advanced solid tumors, evaluating the safety, pharmacokinetics, and anti-tumor efficacy of PRP.
This will be followed by two Phase II studies evaluating the efficacy and safety of PRP. To ensure harmonization between the jurisdictions,
we intend to conduct regulatory meetings in the country where trials are conducted, as well as the FDA and European Medicines Agency.
A pre-IND (Investigational New Drug) meeting will be held with the FDA once initial patient data has been collected from the FIH study
to ensure acceptability of future planned Phase II trials.
Under
European Union regulatory systems, we must submit and obtain authorization for a clinical trial application in each member state in which
we intend to conduct a clinical trial. After we have completed clinical trials, we must obtain marketing authorization before it can
market its product. We must submit applications for marketing authorizations for oncology products under a centralized procedure. The
centralized procedure provides for the grant of a single marketing authorization that is valid for all European Union member states.
The European Medicines Agency (the “EMA”) is the agency responsible for the scientific evaluation of medicines that are to
be assessed via the centralized procedure.
On
June 23, 2016, the UK government held a referendum to gauge voters’ support to remain or leave the European Union. The referendum
resulted in 51.9% of UK voters in favor of leaving the European Union, commonly referred to as “Brexit.” On March 29, 2017,
the UK invoked Article 50 of Lisbon Treaty to initiate complete withdrawal from the European Union, which was effectuated on January
31, 2020. The center for the EMA was based in London but the European Union has relocated the center to The Netherlands.
The
impact of Brexit on the drug approval process in the UK is uncertain. Companies based in the UK and operating in the drug industry are
urging the European Union and the UK to reach an agreement to harmonize the regulatory process once the UK officially exits the European
Union.
Australia
In
Australia, the relevant regulatory body responsible for the pharmaceutical industry is the Therapeutics Goods Administration (the “TGA”).
Prescription medicines are regulated under the Therapeutic Goods Act 1989. Under the Therapeutic Goods Act, the Therapeutic Goods Administration
evaluates new products for quality, safety and efficacy before being approved for market authorization, according to similar standards
employed by the FDA and EMA in the United States and European Union, respectively. However, receiving market authorization in one or
two regions does not guarantee approval in another.
Third-Party
Payor Coverage and Reimbursement
Although
none of our product candidates have been commercialized for any indication, if they are approved for marketing, commercial success of
our product candidates will depend, in part, upon the availability of coverage and reimbursement from third-party payors at the federal,
state and private levels. In addition, in many countries outside the United States, a drug must be approved for reimbursement before
it can be approved for sale in that country.
Eligibility
for reimbursement does not imply that any drug will be paid for in all cases or at a rate that covers our costs, including research,
development, manufacture, sale and distribution. Interim reimbursement levels for new drugs, if applicable, may also not be sufficient
to cover costs and may not be made permanent. Reimbursement rates may vary according to the use of the drug and the clinical setting
in which it is used, may be based on reimbursement levels already set for lower cost drugs and may be incorporated into existing payments
for other services. Net prices for drugs may be reduced by mandatory discounts or rebates required by government healthcare programs
or private payors and by any future relaxation of laws that presently restrict imports of drugs from countries where they may be sold
at lower prices than in the United States. Third-party payors often rely upon Medicare coverage policy and payment limitations in setting
their own reimbursement policies.
In
many countries outside the United States, a drug must be approved for reimbursement before it can be approved for sale in that country.
Approval by the FDA does not ensure approval by regulatory authorities in other countries or jurisdictions, and approval by one foreign
regulatory authority does not ensure approval by regulatory authorities in other foreign countries or by the FDA. The foreign regulatory
approval process may include all of the risks associated with obtaining FDA approval. We may not obtain foreign regulatory approvals
on a timely basis, if at all. We may not be able to file for regulatory approvals and may not receive necessary approvals to commercialize
our products in any foreign market.
The
regulations that govern marketing approvals, pricing and reimbursement for new drug products vary widely from country to country. In
the United States, recently passed legislation may significantly change the approval requirements in ways that could involve additional
costs and cause delays in obtaining approvals. Some countries require approval of the sale price of a drug before it can be marketed.
In many countries, the pricing review period begins after marketing or product licensing approval is granted. In some foreign markets,
prescription pharmaceutical pricing remains subject to continuing governmental control even after initial approval is granted.
Government
authorities and third-party payors, such as private health insurers and health maintenance organizations, decide which medications they
will pay for and establish reimbursement levels. A primary trend in the U.S. healthcare industry and elsewhere is cost containment. Government
authorities and third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular
medications. Increasingly, third-party payors are requiring that drug companies provide them with predetermined discounts from list prices
and are challenging the prices charged for medical products.
Other
Regulations
We
are also subject to numerous federal, state and local laws relating to such matters as safe working conditions, manufacturing practices,
environmental protection, fire hazard control, and disposal of hazardous or potentially hazardous substances. We may incur significant
costs to comply with such laws and regulations now or in the future.
COMPETITION
The
biotechnology and pharmaceutical industries are characterized by continuing technological advancement and significant competition. While
we believe that our technology platforms, product candidates, know-how, experience and scientific resources provide us with competitive
advantages, we face competition from major pharmaceutical and biotechnology companies, academic institutions, governmental agencies and
public and private research institutions, among others. Any product candidates that we successfully develop and commercialize will compete
with existing therapies and new therapies that may become available in the future. Key product features that would affect our ability
to effectively compete with other therapeutics include the efficacy, safety and convenience of our products. The level of generic competition
and the availability of reimbursement from government and other third-party payers will also significantly impact the pricing and competitiveness
of our products. Our competitors also may obtain FDA or other regulatory approval for their products more rapidly than we may obtain
approval for ours, which could result in our competitors establishing a strong market position before we are able to enter the market.
Many
of our competitors have significantly greater financial resources and expertise in research and development, manufacturing, preclinical
testing, conducting clinical trials, obtaining regulatory approvals and marketing approved products than we do. Smaller or early-stage
companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies.
These competitors also compete with us in recruiting and retaining qualified scientific and management personnel and establishing clinical
trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our
programs.
EMPLOYEES
As
of February 2, 2022, we have one full-time and one part-time employee. In addition to our employees, we engage key consultants
and utilize the services of independent contractors to perform various services on our behalf. Some of our executive officers and directors
are engaged in outside business activities that we do not believe conflict with our business. Over time, we may be required to hire additional
employees or engage independent contractors to execute various projects that are necessary to grow and develop our business. These decisions
will be made by our officers and directors, if and when appropriate.
CORPORATE
INFORMATION
Our
principal executive office is located at 302, 6 Butler Street, Camberwell, VIC, 3124 Australia. Our telephone number is 61 03 9882 0780.
Our website is www.propanc.com. We can be contacted by email at www.propanc.com/contact. Our website’s information is not, and
will not be deemed, a part of this Registration Statement or incorporated into any other filings we make with the SEC.
RECENT
DEVELOPMENTS
On
November 26, 2021, the Company entered into a securities purchase agreement (the “Purchase Agreement”) with Sixth Street
Lending, LLC (“Sixth Street”), pursuant to which Sixth Street purchased a convertible promissory note from the Company in
the aggregate principal amount of $53,750, such principal and the interest thereon convertible into shares of the Company’s common
stock at the option of Sixth Street. The transaction contemplated by the Purchase Agreement closed on or about December 2, 2021. The
Company intends to use the net proceeds ($50,000) from the Note for general working capital purposes.
On
November 30, 2021, the Company entered into a Common Stock Purchase Agreement (the “Purchase Agreement”) with Dutchess Capital
Growth Fund LP, a Delaware limited partnership, (“Dutchess”), providing for an equity financing facility (the “Equity
Line”). The Purchase Agreement provides that upon the terms and subject to the conditions in the Purchase Agreement, Dutchess is
committed to purchase up to Five Million Dollars ($5,000,000) of shares of common stock, $0.001 par value per share (the “Common
Stock”), over the 36 month term of the Purchase Agreement (the “Total Commitment”).
Under
the terms of the Purchase Agreement, Dutchess will not be obligated to purchase shares of Common Stock unless and until certain conditions
are met, including but not limited to a Registration Statement on Form S-1 (the “Registration Statement”) becoming effective
which registers Dutchess’ resale of any Common Stock purchased by Dutchess under the Equity Line. From time to time over the 36-month
term of the Purchase Agreement, commencing on the trading day immediately following the date on which the Registration Statement becomes
effective, the Company, in our sole discretion, may provide Dutchess with a draw down notice (each, a “Draw Down Notice”),
to purchase a specified number of shares of Common Stock (each, a “Draw Down Amount Requested”), subject to the limitations
discussed below. The actual amount of proceeds the Company will receive pursuant to each Draw Down Notice (each, a “Draw Down Amount”)
is to be determined by multiplying the Draw Down Amount Requested by the applicable purchase price. The purchase price of each share
of Common Stock equals 92% of the lowest trading price of the Common Stock during the five (5) business days prior to the Closing Date.
Closing Date shall mean the five (5) business days after the Clearing Date. Clearing Date shall mean the first business day that the
Selling Security Holder holds the Draw Down Amount in its brokerage account and is eligible to trade the shares.
The
maximum number of shares of Common Stock requested to be purchased pursuant to any single Draw Down Notice cannot exceed the lesser of
(i) 300% of the average daily share volume of the Common Stock in the five (5) trading days immediately preceding the Draw Down Notice
or (ii) an aggregate value of $250,000.
The
Company agreed to pay to Dutchess a commitment fee for entering into the Purchase Agreement of 1,000,000 restricted shares of our common
stock. The shares were issued December 10, 2021.
On
December 7, 2021, the Company entered into a securities purchase agreement (the “Purchase Agreement”) with ONE44 Capital
LLC, (“ONE44”), pursuant to which ONE44 purchased a convertible promissory note the “Note”) from the Company
in the aggregate principal amount of $170,000, such principal and the interest thereon convertible into shares of the Company’s
common stock at the option of ONE44. The transaction contemplated by the Purchase Agreement closed on or about December 13, 2021. The
Company intends to use the net proceeds ($153,000) from the Note for general working capital purposes. The Note contains an original
issue discount amount of $17,000.
On
January 4, 2022, the Company) entered into a securities purchase agreement (the “Purchase Agreement”) with Sixth Street Lending,
LLC (“Sixth Street”), pursuant to which Sixth Street purchased a convertible promissory note from the Company in the aggregate
principal amount of $63,750, such principal and the interest thereon convertible into shares of the Company’s common stock at the
option of Sixth Street. The transaction contemplated by the Purchase Agreement closed on January 6, 2022. The Company intends to use
the net proceeds ($60,000) from the Note for general working capital purposes.
SUMMARY
OF FINANCIAL INFORMATION
The
following summary consolidated statements of operations data for the fiscal years ended June 30, 2021 and 2020 have been derived from
our audited consolidated financial statements included elsewhere in this prospectus. The historical financial data presented below is
not necessarily indicative of our financial results in future periods, and the results for the year ended June 30, 2021 are not necessarily
indicative of our operating results to be expected for the full fiscal year ending June 30, 2022 or any other period. You should read
the summary consolidated financial data in conjunction with those financial statements and the accompanying notes and “Management’s
Discussion and Analysis of Financial Condition and Results of Operations.” Our consolidated interim financial statements
are prepared and presented in accordance with United States generally accepted accounting principles, or U.S. GAAP. Our consolidated
financial statements have been prepared on a basis consistent with our audited financial statements and include all adjustments, consisting
of normal and recurring adjustments that we consider necessary for a fair presentation of the financial position and results of operations
as of and for such periods.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
CONDENSED
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE INCOME (LOSS)
(Unaudited)
| | |
Three Months Ended September 30, | |
| | |
2021 | | |
2020 | |
| | |
| | |
| |
| REVENUE | |
| | | |
| | |
| Revenue | |
$ | - | | |
$ | - | |
| | |
| | | |
| | |
| OPERATING EXPENSES | |
| | | |
| | |
| Administration expenses | |
| 431,740 | | |
| 323,111 | |
| Occupancy expenses | |
| 7,736 | | |
| 9,204 | |
| Research and development | |
| 46,554 | | |
| 50,846 | |
| TOTAL OPERATING EXPENSES | |
| 486,030 | | |
| 383,161 | |
| | |
| | | |
| | |
| LOSS FROM OPERATIONS | |
| (486,030 | ) | |
| (383,161 | ) |
| | |
| | | |
| | |
| OTHER INCOME (EXPENSE) | |
| | | |
| | |
| Interest expense | |
| (109,853 | ) | |
| (159,281 | ) |
| Change in fair value of derivative liabilities | |
| (3,904 | ) | |
| 64,952 | |
| Gain on extinguishment of debt, net | |
| - | | |
| 49,985 | |
| Foreign currency transaction gain | |
| 109,129 | | |
| 1,960 | |
| TOTAL OTHER EXPENSE, NET | |
| (4,628 | ) | |
| (42,384 | ) |
| | |
| | | |
| | |
| LOSS BEFORE TAXES | |
| (490,658 | ) | |
| (425,545 | ) |
| | |
| | | |
| | |
| Tax benefit | |
| - | | |
| - | |
| | |
| | | |
| | |
| NET LOSS | |
| (490,658 | ) | |
| (425,545 | ) |
| | |
| | | |
| | |
| Deemed Dividend | |
| (114,844 | ) | |
| - | |
| | |
| | | |
| | |
| NET LOSS AVAILABLE TO COMMON STOCKHOLDERS | |
$ | (605,502 | ) | |
$ | (425,545 | ) |
| | |
| | | |
| | |
| BASIC AND DILUTED NET LOSS PER SHARE | |
$ | (0.02 | ) | |
$ | (0.71 | ) |
| | |
| | | |
| | |
| BASIC AND DILUTED WEIGHTED AVERAGE SHARES OUTSTANDING | |
| 27,142,519 | | |
| 597,314 | |
| | |
| | | |
| | |
| NET LOSS AVAILABLE TO COMMON STOCKHOLDERS | |
$ | (605,502 | ) | |
$ | (425,545 | ) |
| | |
| | | |
| | |
| OTHER COMPREHENSIVE INCOME (LOSS) | |
| | | |
| | |
| Unrealized foreign currency translation gain (loss) | |
| 64,193 | | |
| (75,755 | ) |
| | |
| | | |
| | |
| TOTAL OTHER COMPREHENSIVE INCOME (LOSS) | |
| 64,193 | | |
| (75,755 | ) |
| | |
| | | |
| | |
| TOTAL COMPREHENSIVE LOSS | |
$ | (541,309 | ) | |
$ | (501,300 | ) |
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
CONSOLIDATED
STATEMENTS OF OPERATIONS AND COMPREHENSIVE INCOME (LOSS)
| | |
Years Ended June 30, | |
| | |
2021 | | |
2020 | |
| | |
| | |
| |
| REVENUE | |
| | | |
| | |
| Revenue | |
$ | - | | |
$ | - | |
| | |
| | | |
| | |
| OPERATING EXPENSES | |
| | | |
| | |
| Administration expenses | |
| 1,553,075 | | |
| 3,281,464 | |
| Occupancy expenses | |
| 28,112 | | |
| 32,809 | |
| Research and development | |
| 230,956 | | |
| 179,987 | |
| TOTAL OPERATING EXPENSES | |
| 1,812,143 | | |
| 3,494,260 | |
| | |
| | | |
| | |
| LOSS FROM OPERATIONS | |
| (1,812,143 | ) | |
| (3,494,260 | ) |
| | |
| | | |
| | |
| OTHER INCOME (EXPENSE) | |
| | | |
| | |
| Interest expense | |
| (449,457 | ) | |
| (1,748,381 | ) |
| Interest income | |
| 1 | | |
| 946 | |
| Other income | |
| - | | |
| 57,636 | |
| Change in fair value of derivative liabilities | |
| (8,186 | ) | |
| 385,293 | |
| Gain from settlement of debt, net | |
| 49,319 | | |
| - | |
| Gain on extinguishment of debt, net | |
| 50,607 | | |
| 67,123 | |
| Foreign currency transaction gain (loss) | |
| 30,497 | | |
| (143,808 | ) |
| TOTAL OTHER EXPENSE, NET | |
| (327,219 | ) | |
| (1,381,191 | ) |
| | |
| | | |
| | |
| LOSS BEFORE TAXES | |
| (2,139,362 | ) | |
| (4,875,451 | ) |
| | |
| | | |
| | |
| Tax benefit | |
| 113,415 | | |
| 134,728 | |
| | |
| | | |
| | |
| NET LOSS | |
| (2,025,947 | ) | |
| (4,740,723 | ) |
| | |
| | | |
| | |
| Deemed dividend | |
| (391,749 | ) | |
| - | |
| | |
| | | |
| | |
| NET LOSS AVAILABLE TO COMMON STOCKHOLDERS | |
$ | (2,417,696 | ) | |
$ | (4,740,723 | ) |
| | |
| | | |
| | |
| BASIC AND DILUTED NET LOSS PER SHARE AVAILABLE TO COMMON STOCKHOLDERS | |
$ | (0.80 | ) | |
$ | (192.45 | ) |
| | |
| | | |
| | |
| BASIC AND DILUTED WEIGHTED AVERAGE SHARES OUTSTANDING | |
| 3,032,612 | | |
| 24,634 | |
| | |
| | | |
| | |
| NET LOSS AVAILABLE TO COMMON STOCKHOLDERS | |
$ | (2,417,696 | ) | |
$ | (4,740,723 | ) |
| | |
| | | |
| | |
| OTHER COMPREHENSIVE INCOME (LOSS) | |
| | | |
| | |
| Unrealized foreign currency translation gain (loss) | |
| (182,467 | ) | |
| 200,673 | |
| | |
| | | |
| | |
| TOTAL OTHER COMPREHENSIVE INCOME (LOSS) | |
| (182,467 | ) | |
| 200,673 | |
| | |
| | | |
| | |
| TOTAL COMPREHENSIVE LOSS | |
$ | (2,600,163 | ) | |
$ | (4,540,050 | ) |
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
CONDENSED
CONSOLIDATED BALANCE SHEETS
| | |
September 30, 2021 | | |
June 30, 2021 | |
| | |
(Unaudited) | | |
| |
| ASSETS | |
| | | |
| | |
| | |
| | | |
| | |
| CURRENT ASSETS: | |
| | | |
| | |
| Cash | |
$ | 45,817 | | |
$ | 2,255 | |
| GST tax receivable | |
| 2,238 | | |
| 4,341 | |
| Prepaid expenses and other current assets | |
| 8,353 | | |
| - | |
| | |
| | | |
| | |
| TOTAL CURRENT ASSETS | |
| 56,408 | | |
| 6,596 | |
| | |
| | | |
| | |
| Security deposit - related party | |
| 2,164 | | |
| 2,250 | |
| Property and equipment, net | |
| 3,593 | | |
| 4,255 | |
| | |
| | | |
| | |
| TOTAL ASSETS | |
$ | 62,165 | | |
$ | 13,101 | |
| | |
| | | |
| | |
| LIABILITIES AND STOCKHOLDERS’ DEFICIT | |
| | | |
| | |
| | |
| | | |
| | |
| CURRENT LIABILITIES: | |
| | | |
| | |
| Accounts payable | |
$ | 826,184 | | |
$ | 1,002,335 | |
| Accrued expenses and other payables | |
| 407,775 | | |
| 892,151 | |
| Convertible notes and related accrued interest, net of discounts and premiums | |
| 584,608 | | |
| 624,583 | |
| Embedded conversion option liabilities | |
| 58,124 | | |
| 54,220 | |
| Due to former director - related party | |
| 32,076 | | |
| 33,347 | |
| Loan from former director - related party | |
| 53,384 | | |
| 55,500 | |
| Employee benefit liability | |
| 406,644 | | |
| 418,538 | |
| | |
| | | |
| | |
| TOTAL CURRENT LIABILITIES | |
| 2,368,795 | | |
| 3,080,674 | |
| | |
| | | |
| | |
| TOTAL LIABILITIES | |
$ | 2,368,795 | | |
$ | 3,080,674 | |
| | |
| | | |
| | |
| Commitments and Contingencies | |
| | | |
| | |
| | |
| | | |
| | |
| STOCKHOLDERS’ DEFICIT: | |
| | | |
| | |
| Preferred stock, 1,500,005 shares authorized, $0.01 par value: | |
| | | |
| | |
| Series A preferred stock, $0.01 par value; 500,000 shares authorized; 500,000 shares issued and outstanding as of September 30, 2021 and June 30, 2021 | |
$ | 5,000 | | |
$ | 5,000 | |
| Series B preferred stock, $0.01 par value; 5 shares authorized; 1 share issued and outstanding as of September 30, 2021 and June 30, 2021 | |
| - | | |
| - | |
| Common stock, $0.001 par value; 1,000,000,000 shares authorized;43,841,644 and 14,055,393 shares issued and outstanding as of September 30, 2021 and June 30, 2021, respectively | |
| 43,842 | | |
| 14,056 | |
| Common stock issuable (2,002,549 and 59 shares as of September 30, 2021 and June 30, 2021, respectively) | |
| 2,002 | | |
| - | |
| Additional paid-in capital | |
| 55,444,574 | | |
| 54,074,110 | |
| Subscription receivable | |
| (100,000 | ) | |
| - | |
| Accumulated other comprehensive income | |
| 1,149,397 | | |
| 1,085,204 | |
| Accumulated deficit | |
| (58,804,968 | ) | |
| (58,199,466 | ) |
| Treasury stock (1 share) | |
| (46,477 | ) | |
| (46,477 | ) |
| | |
| | | |
| | |
| TOTAL STOCKHOLDERS’ DEFICIT | |
| (2,306,630 | ) | |
| (3,067,573 | ) |
| | |
| | | |
| | |
| TOTAL LIABILITIES AND STOCKHOLDERS’ DEFICIT | |
$ | 62,165 | | |
$ | 13,101 | |
CAUTIONARY
NOTE REGARDING FORWARD-LOOKING STATEMENTS
This
prospectus contains forward-looking statements. Forward-looking statements give our current expectations or forecasts of future events.
You can identify these statements by the fact that they do not relate strictly to historical or current facts. Forward-looking statements
involve risks and uncertainties and include statements regarding, among other things, our projected revenue growth and profitability,
our growth strategies and opportunity, anticipated trends in our market and our anticipated needs for working capital. They are generally
identifiable by use of the words “may,” “will,” “should,” “anticipate,” “estimate,”
“plans,” “potential,” “projects,” “continuing,” “ongoing,” “expects,”
“management believes,” “we believe,” “we intend” or the negative of these words or other variations
on these words or comparable terminology. These statements may be found under the sections entitled “Management’s Discussion
and Analysis of Financial Condition and Results of Operations” and “Business,” as well as in this prospectus generally.
In particular, these include statements relating to future actions, prospective products, market acceptance, future performance or results
of current and anticipated products, sales efforts, expenses, and the outcome of contingencies such as legal proceedings and financial
results.
Examples
of forward-looking statements in this prospectus include, but are not limited to, our expectations regarding our business strategy, business
prospects, operating results, operating expenses, working capital, liquidity and capital expenditure requirements. Important assumptions
relating to the forward-looking statements include, among others, assumptions regarding demand for our products, the cost, terms and
availability of components, pricing levels, the timing and cost of capital expenditures, competitive conditions and general economic
conditions. These statements are based on our management’s expectations, beliefs and assumptions concerning future events affecting
us, which in turn are based on currently available information. These assumptions could prove inaccurate. Although we believe that the
estimates and projections reflected in the forward-looking statements are reasonable, our expectations may prove to be incorrect.
SUMMARY
OF RISKS
Our
business is subject to a number of risks and uncertainties that you should understand before making an investment decision. For example,
we have no commercial product, a history of net losses, we expect to continue to incur net losses, we will require significant additional
funding and we may not achieve or maintain profitability. Furthermore, we have no cash flow from operations to sustain our operations.
We have historically relied upon the issuance of equity and/or convertible debt to fund our operations, which debt we are currently unable
to repay in cash. Our ability to ever generate revenues will depend solely on the commercial success of PRP, our only prospective product,
which depends upon its approval by applicable regulatory authorities and then market acceptance by purchasers in the pharmaceutical market
and the future market demand and medical need for products and research utilizing PRP. At present, PRP has only been used for research
and clinical trial purposes in animals, and there is no commercially approved drug product or drug product submitted in a pending marketing
application that incorporates PRP as an ingredient. As a result, no marketing authority has reviewed our drug master file (DMF) for PRP
as a product ingredient or inspected our Company. As of September 30, 2021, we have an accumulated deficit of $58,804,968 since inception.
We have incurred substantial net losses since our inception, including net loss of $2,417,696 and $4,740,723 for the fiscal years ended
June 30, 2021 and June 30, 2020, respectively. We expect to incur additional losses as we continue to invest in our research and development
programs and move forward with our human clinical trials application, clinical trials and commercialization activities. Additional risks
are discussed more fully in the section entitled “Risk Factors” following this prospectus summary. These risks include, but
are not limited to, the following:
| |
● |
Our
ability to continue as a going concern absent obtaining adequate new debt and/or equity financings. |
| |
● |
We
face risks related to Novel Coronavirus (COVID-19) which could significantly disrupt our research and development, operations, sales,
and financial results. |
| |
|
|
| |
● |
We
have incurred significant losses since our inception, and we expect to incur significant losses for the foreseeable future and may
never achieve or maintain profitability. |
| |
● |
We
will continue to need substantial additional funding and raise capital when needed to initiate and continue our product development
programs and commercialization efforts. |
| |
|
|
| |
● |
As
an early stage company, it may be difficult for you to evaluate the success of our business to date and to assess our future viability. |
| |
|
|
| |
● |
We
currently rely, and may continue to rely for the foreseeable future, on substantial debt financing that we are not able to repay
in cash. |
| |
|
|
| |
● |
Raising
additional capital is highly likely to cause dilution to our stockholders, restrict our operations or require us to relinquish rights
to our technologies or product candidate. |
| |
|
|
| |
● |
The
conversion of some or all of our currently outstanding convertible notes in shares of our common stock will dilute the ownership
interests of existing stockholders. |
| |
|
|
| |
● |
It
may be difficult for you to evaluate the success of our business to date and to assess our future viability. |
| |
|
|
| |
● |
Our
only product candidate, PRP, remains in the early stages of development and may never become commercially viable, and therefore,
you may lose your investment. |
| |
|
|
| |
● |
PRP
may cause undesirable side effects that could negatively impact its clinical trial results or limit its use, hindering further development,
subject us to possible product liability claims, and make it more difficult to commercialize PRP. |
| |
|
|
| |
● |
Our
ability to successfully initiate and complete our clinical trials of PRP. |
| |
|
|
| |
● |
Our
ability to obtain regulatory approval in jurisdictions in the United States and outside the United States to be able to market PRP
in those jurisdictions. |
| |
|
|
| |
● |
Our
ability in the future to establish sales and marketing capabilities or enter into agreements with third parties to sell and market
PRP. |
| |
|
|
| |
● |
We
face substantial competition, which may result in others discovering, developing or commercializing products before or more successfully
than we do. |
| |
|
|
| |
● |
Our
ability to seek approval for reimbursement for PRP before it can be marketed, assuming successful commercialization, and us being
then subject to unfavorable pricing regulations, third-party reimbursement practices or healthcare reform initiatives. |
| |
|
|
| |
● |
We
may depend on collaborations with third parties for the development and commercialization of PRP, and these collaborations may be
unsuccessful. |
| |
|
|
| |
● |
Our
third party manufacturers of PRP performing satisfactorily or at all, and our reliance on any third-party for the supply of PRP. |
| |
● |
Our
ability to comply with our obligations under any intellectual property licenses with third parties. |
| |
|
|
| |
● |
Our
ability to protect our intellectual property rights. |
| |
|
|
| |
● |
Our
ability to obtain, or if there are delays in obtaining, required regulatory approvals, to commercialize PRP, and our ability to generate
revenue. |
| |
|
|
| |
● |
Our
ability to obtain marketing approval in international jurisdictions to market PRP in international jurisdictions. |
| |
|
|
| |
● |
Our
ability to obtain marketing approval of and commercialize PRP and affect the prices we may obtain. |
| |
● |
PRP
or any other product candidate for which we obtain marketing approval could be subject to restrictions or withdrawal from the market
and our ability to comply with applicable regulatory requirements. |
| |
|
|
| |
● |
We
rely on the significant experience and specialized expertise of the Chief Executive Officer and the Chief Financial Officer who works
on a part-time bases, we do not currently have any other members of a management team. |
| |
|
|
| |
● |
We
have identified material weaknesses in our internal control over financial reporting that, if not properly remediated, could result
in material misstatements in our consolidated financial statements in future periods. |
| |
|
|
| |
● |
We
do not have any independent directors, which represents a potential conflict of interest, and helps create a material weakness in
our disclosure controls and procedures as well as our internal control over financial reporting. |
| |
|
|
| |
● |
Our
ability to implement and maintain an effective system of internal control over financial reporting, and accordingly, our ability
to accurately report our financial results or prevent fraud. |
| |
|
|
| |
● |
The
market price of our common stock may continue to be highly volatile, and you may not be able to resell your shares at or above the
public offering price and therefore, you could lose all or part of your investment. |
| |
|
|
| |
● |
Our
shares of common stock are thinly traded and there may not be an active, liquid trading market for our common shares. |
| |
|
|
| |
● |
Our
Chief Executive Officer is our controlling shareholder and will continue to control our Company for the foreseeable future due to
his ownership of super-voting shares, and therefore, it is not likely that you will be able to elect directors or have any say in
the policies of our Company. |
| |
|
|
| |
● |
Future
sales and issuances of our capital stock or rights to purchase capital stock will result in additional dilution of the percentage
ownership of our stockholders and could cause our stock price to decline. |
| |
|
|
| |
● |
We
are a smaller reporting company, and therefore, we are subject to scaled disclosure requirements that may make it more challenging
for investors to analyze our results of operations and financial prospects. |
| |
|
|
| |
● |
PRP
or any other product candidate for which we obtain marketing approval could be subject to restrictions or withdrawal from the market
and our ability to comply with applicable regulatory requirements. |
| |
|
|
| |
● |
We
rely on the significant experience and specialized expertise of the Chief Executive Officer and the Chief Financial Officer who works
on a part-time bases, we do not currently have any other members of a management team. |
| |
|
|
| |
● |
We
have identified material weaknesses in our internal control over financial reporting that, if not properly remediated, could result
in material misstatements in our consolidated financial statements in future periods. |
| |
|
|
| |
● |
We
do not have any independent directors, which represents a potential conflict of interest, and helps create a material weakness in
our disclosure controls and procedures as well as our internal control over financial reporting. |
| |
|
|
| |
● |
Our
ability to implement and maintain an effective system of internal control over financial reporting, and accordingly, our ability
to accurately report our financial results or prevent fraud. |
| |
● |
The
market price of our common stock may continue to be highly volatile, and you may not be able to resell your shares at or above the
public offering price and therefore, you could lose all or part of your investment. |
| |
|
|
| |
● |
Our
shares of common stock are thinly traded and there may not be an active, liquid trading market for our common shares. |
| |
|
|
| |
● |
Our
Chief Executive Officer is our controlling shareholder and will continue to control our Company for the foreseeable future due to
his ownership of super-voting shares, and therefore, it is not likely that you will be able to elect directors or have any say in
the policies of our Company. |
| |
|
|
| |
● |
Future
sales and issuances of our capital stock or rights to purchase capital stock will result in additional dilution of the percentage
ownership of our stockholders and could cause our stock price to decline. |
| |
|
|
| |
● |
We
are a smaller reporting company, and therefore, we are subject to scaled disclosure requirements that may make it more challenging
for investors to analyze our results of operations and financial prospects. |
| |
|
|
| |
● |
other
risks, including those described in the “Risk Factors” discussion of this prospectus. |
We
operate in a very competitive and rapidly changing environment. New risks emerge from time to time. It is not possible for us to predict
all of those risks, nor can we assess the impact of all of those risks on our business or the extent to which any factor may cause actual
results to differ materially from those contained in any forward-looking statement. The forward-looking statements in this prospectus
are based on assumptions management believes are reasonable. However, due to the uncertainties associated with forward-looking statements,
you should not place undue reliance on any forward-looking statements. Further, forward-looking statements speak only as of the date
they are made, and unless required by law, we expressly disclaim any obligation or undertaking to publicly update any of them in light
of new information, future events, or otherwise.
RISK
FACTORS
An
investment in our Common Stock involves a high degree of risk. Before deciding whether to invest in our Common Stock, you should consider
carefully the risks described below, together with all of the other information set forth in this prospectus and the documents incorporated
by reference herein, and in any free writing prospectus that we have authorized for use in connection with this offering. If any of these
risks actually occurs, our business, financial condition, results of operations or cash flow could be harmed. This could cause the trading
price of our Common Stock to decline, resulting in a loss of all or part of your investment. The risks described below and in the documents
referenced above are not the only ones that we face. Additional risks not presently known to us or that we currently deem immaterial
may also affect our business.
COVID-19
At
Propanc, our highest priority remains the safety, health and well-being of our employees, their families and our communities. The COVID-19
pandemic is a highly fluid situation and it is not currently possible for us to reasonably estimate the impact it may have on our financial
and operating results. We will continue to evaluate the impact of the COVID-19 pandemic on our business as we learn more and the impact
of COVID-19 on our industry becomes clearer. We are complying health guidelines regarding safety procedures, including, but are not limited
to, social distancing, remote working, and teleconferencing. The extent of the future impact of the COVID-19 pandemic on our business
is uncertain and difficult to predict. Adverse global economic and market conditions as a result of COVID-19 could also adversely affect
our business. If the pandemic continues to cause significant negative impacts to economic conditions, our results of operations, financial
condition and liquidity could be adversely impacted.
RISKS
RELATED TO OUR FINANCIAL CONDITION AND OUR NEED FOR ADDITIONAL CAPITAL
Our
ability to continue as a going concern is in substantial doubt absent obtaining adequate new debt or equity financings.
We
have concerns about our ability to continue as a going concern based on the absence of revenues, recurring losses from operations and
our need for additional financing to fund all of our operations. Working capital limitations continue to impinge on our day-to-day operations,
thus contributing to continued operating losses. For the fiscal years ended June 30, 2021 and June 30, 2020, we had net losses of $2,025,947
and $4,740,723, respectively. Further, as of September 30, 2021, we had $45,817 in cash and had an accumulated deficit of $58,804,968.
Based
upon our current business plan, we will need considerable cash investments to have the opportunity to be successful. Our capital requirements
and cash needs are significant and continuing. We can provide no assurance that we will be able to generate a sufficient amount of revenue,
if any, from our business in order to achieve profitability. It is not possible at this time for us to predict with assurance the potential
success of our business. The revenue and income potential of our proposed business and operations are unknown. If we cannot continue
as a viable entity, we may be unable to continue our operations and you may lose some or all of your investment in our common stock.
We
have incurred significant losses since our inception. We expect to incur significant losses for the foreseeable future and may never
achieve or maintain profitability.
Since
inception, we have incurred significant operating losses. Our net loss was $2,025,947 and $4,740,723, respectively, for the fiscal years
ended June 30, 2021 and June 30, 2020, respectively. As of September 30, 2021, we had a deficit accumulated of $58,804,968. To date,
we have not generated any revenues and have financed most of our operations with funds obtained from private financings.
Since
October 2007, we have devoted substantially all of our efforts to research and development of our product candidates, particularly PRP,
and efforts to protect our intellectual property. From January-February 2016, and October 2016-April 2017, we contracted
with third parties to perform a number of laboratory studies and dose range finding studies designed to examine the anti-cancer effects
of PRP and prepare for human clinical trials. Since mid-2017, we developed a suitable manufacturing process for each active drug substance
in the PRP formulation, capable of producing a full scale GMP manufacture of PRP for human trials. We were granted Orphan Drug Designation
status from the FDA for PRP for the treatment of pancreatic cancer. In March 2018, a scientific advice meeting was conducted with the
MHRA (Medicines and Healthcare Products Regulatory Agency) UK, to assist with preparation of our first Clinical Trial Application (CTA).
We expect that it will be many years, if ever, before we have a product candidate ready for commercialization. We expect to incur significant
expenses and increasing operating losses for the foreseeable future if and as we progress PRP into clinical trials, continue our research
and development, seek regulatory approvals, establish or contract for a sales and marketing infrastructure, maintain and expand our intellectual
property portfolio, and add personnel.
To
become profitable, we must develop and eventually commercialize PRP or some other product with significant market potential. This will
require us to successfully complete clinical trials, obtain market approval and market and sell PRP or whatever other product that we
obtain approval for. We might not succeed in any one or a number of these activities, and even if we do, we may never generate revenues
that are significant enough to achieve profitability. Our failure to become and remain profitable would decrease our value and could
impair our ability to raise capital, maintain our research and development efforts, expand our business or continue our operations.
As
an early-stage company, it may be difficult for you to evaluate the success of our business to date and to assess our future viability.
Despite
having been founded in 2007, we remain an early-stage company. We commenced active operations in the second half of 2010. Our operations
to date have been mainly limited to establishing our research programs, particularly PRP, building our intellectual property portfolio
and deepening our scientific understanding of our product development. We have not yet initiated, let alone demonstrated any ability
to successfully complete, any clinical trials, including large-scale, pivotal clinical trials, obtain marketing approvals, manufacture
a commercial scale product, or arrange for a third party to do so on our behalf, or conduct sales and marketing activities necessary
for successful product commercialization. It will take a number of years for PRP to be made available for the treatment of cancer, if
it ever is. Given our relatively short operating history compared to the timeline required to fully develop a new drug, you are cautioned
about making any predictions on our future success or viability based on our activities or results to date. In addition, we may encounter
unforeseen expenses, difficulties, complications, delays and other known and unknown factors. We will eventually need to transition from
a company with a research focus to a company capable of supporting commercial activities. We may not be successful in such a transition.
We
currently rely, and may continue to rely for the foreseeable future, on substantial debt financing that we are not able to repay in cash.
In
order to maintain our operations, including our research and development efforts and our preclinical development of PRP, we have over
the last few years entered into a number of securities purchase agreements pursuant to which we issued convertible debt in return for
cash. We are not currently able to repay either the current principal or interest on this debt in cash. Our lenders, therefore, can convert
their debt into shares of our common stock, at a discount to current market prices and then attempt to sell these shares on the open
market in order to pay down their loans and receive a return on their investment. These financings pose the risk that as these debts
are converted, our stock price will reflect the reduced prices our lenders are willing to sell their shares at, given the discount they
have received. These financings contain no floor on the price our lenders can convert their debt into shares of our common stock and
they could conceivably reduce the price our common stock to near zero. These types of financings negatively impact our balance sheet
and the appeal of our common stock as an investment. While we are actively exploring various alternatives to reduce if not eliminate
this debt, for the foreseeable future we will continue to carry it on our balance sheet, and we may have to enter into additional such
financings in order to sustain our operations. As a result, the price of our common stock and our market capitalization are subject to
significant declines until our convertible debt is either refinanced on a favorable basis or is eliminated.
As
of September 30, 2021, the total amount of debt net of discounts outstanding under convertible notes, including interest, is $584,608
(not including redemption premium). Please see the section captioned “Management’s Discussion of Financial Condition and
Results of Operations - Recent Developments” for further information
We
will continue to need substantial additional funding. If we are unable to raise capital when needed, we would be forced to delay, reduce
or eliminate our product development programs or commercialization efforts.
We
expect our expenses to significantly increase in connection with our ongoing activities, particularly if we initiate clinical trials
of, and ultimately seek marketing approval for, PRP. In addition, even if we ultimately obtain marketing approval for PRP or any other
product candidate, we expect to incur significant commercialization expenses related to product sales, marketing, manufacturing and distribution.
We also hope to continue and expand our research and development activities. Accordingly, we will need to obtain substantial additional
funding in connection with our continuing operations. If we are unable to raise capital when needed or on attractive terms, we would
be forced to delay, reduce or eliminate our future commercialization efforts or any research and development programs.
Our
future capital requirements will depend on many factors, including, among others, the scope, progress and results of our potential future
clinical trials, the costs, timing and outcome of regulatory review of PRP, the costs of any future commercialization activities, and
the costs of preparing and filing future patent applications, if any. Accordingly, we will continue to rely on additional financing to
achieve our business objectives. Adequate additional financing may not be available to us on acceptable terms, or at all. Even if we
are able to enter into financing agreements, we may be forced to pay higher interest rates, accept default provisions in financing agreements
that we believe are overly punitive, make balloon payments as required, and, as noted below, if we issue convertible debt the price of
our common stock may well be negatively affected and our existing stockholders may suffer dilution.
Raising
additional capital will cause dilution to our stockholders, restrict our operations or require us to relinquish rights to our technologies
or product candidates.
Until
such time, if ever, as we can generate substantial product revenues, we expect to continue to finance our cash needs through a combination
of equity offerings and additional debt financings, and possibly also through future collaborations, strategic alliances and licensing
arrangements. To the extent that we raise additional capital through the sale of equity or debt securities, including convertible debt
securities, the ownership interest of our existing stockholders will be diluted upon conversion, and the terms of these securities may
include liquidation or other preferences that adversely affect the rights of our existing stockholders.
Debt
financing, if available, may also involve agreements that include restrictive covenants limiting or restricting our ability to take specific
actions, such as merging with other companies or consummating certain changes of control, acquiring other companies, engaging in new
lines of business, incurring additional debt, making capital expenditures, making certain investments, paying dividends, transferring
or disposing of assets, amending certain material agreements, incurring additional indebtedness or enter into various specified transactions.
We therefore may not be able to engage in any of the foregoing transactions unless we obtain the consent of the lender or terminate such
debt agreements. Our debt agreements may also contain certain financial covenants, including achieving certain milestones and may be
secured by substantially all of our assets. In the event we enter into such debt agreements, there is no guarantee that we will be able
to generate sufficient cash flow or sales to pay the principal and interest under our debt agreements or to satisfy all of the financial
covenants.
If
we raise additional funds through collaborations, strategic alliances or licensing arrangements with third parties, we may have to relinquish
valuable rights to our technologies, future revenue streams, research programs or product candidates or to grant licenses on terms that
may not be favorable to us. If we are unable to raise additional funds through equity or debt financings when needed, we may be required
to delay, limit, reduce or terminate our product development or future commercialization efforts or grant rights to develop and market
product candidates that we would otherwise prefer to develop and market ourselves.
The
conversion of some or all of our currently outstanding convertible notes in shares of our common stock will dilute the ownership interests
of existing stockholders.
The
conversion of some or all of our currently outstanding convertible notes into shares of our common stock will dilute the ownership interests
of existing stockholders. As of September 30, 2021, we had 5 outstanding notes convertible into approximately 23,293,971 shares of our
common stock (based on then applicable conversion prices). Each holder of the notes has agreed to a 4.99% beneficial ownership conversion
limitation (subject to certain noteholders’ ability to increase such limitation to 9.99% upon 60 days’ notice to us), and
each note may not be converted during the first six-month period from the date of issuance. Any sales in the public market of the common
stock issuable upon such conversion or any anticipated conversion of our convertible notes into shares of our common stock could adversely
affect prevailing market prices of our common stock.
The
accounting method for convertible debt securities that may be settled in cash could have a material adverse effect on our reported financial
results.
Under
Financial Accounting Standards Board Accounting Standards Codification 470-20, Debt with Conversion and Other Options (“ASC 470-20”),
we are required to separately account for the liability and equity components of our convertible notes because they may be settled entirely
or partially in cash upon conversion in a manner that reflects our economic interest cost. The effect of ASC 470-20 on the accounting
for our convertible notes is that the equity component is required to be included in the additional paid-in capital section of stockholders’
deficit on our consolidated balance sheet, and the value of the equity component would be treated as a discount for purposes of accounting
for the debt component of our convertible notes. As a result, we will be required to record a greater amount of non-cash interest expense
in current periods presented as a result of the amortization of the discounted carrying value of our convertible debt or notes to their
face amount over the terms. We will report higher net loss in our financial results in part because ASC 470-20 will require interest
to include both the current period’s amortization of the debt discount and the instrument’s coupon interest, which could
adversely affect our reported or future financial results, the trading price of our common stock and the trading price of our convertible
notes.
In
addition, because our convertible notes may be settled entirely or partly in cash, under certain circumstances, these are currently accounted
for utilizing the treasury stock method, the effect of which is that the shares issuable upon conversion are not included in the calculation
of diluted earnings per share except to the extent that the conversion value exceeds their principal amount. Under the treasury stock
method, for diluted earnings per share purposes, the transaction is accounted for as if the number of shares of common stock that would
be necessary to settle such excess, if we elected to settle such excess in shares, are issued. We cannot be sure that the accounting
standards in the future will continue to permit the use of the treasury stock method. If we are unable to use the treasury stock method
in accounting for the shares issuable upon conversion of our convertible notes, then our diluted earnings per share would be adversely
affected.
We
maintain our cash in Australian financial institutions that are not insured.
The
Company maintains its cash in banks and financial institutions in Australia. Bank deposits in Australian banks are uninsured. The Company
has not experienced any losses in such accounts through to date.
RISKS
RELATED TO THE DISCOVERY, DEVELOPMENT AND COMMERCIALIZATION OF OUR PRODUCT CANDIDATES
Because
PRP remains in the early stages of development and may never become commercially viable, you may lose your investment.
At
present, our only product candidate, PRP, is still in preclinical development. While we are hopeful that the preclinical testing we have
completed will lead to our initiating human clinical trials in 2022 as noted elsewhere we expect that it will be several years, at least,
before PRP can be commercialized. Further, if clinical trials for PRP fail to produce statistically significant results, we would likely
be forced to either spend several more years in development attempting to correct whatever flaws were identified in the trials, or we
would have to abandon PRP altogether. Either of those contingencies, and especially the latter, would dramatically increase the amount
of time before we would be able to generate any product-related revenue, and we may well be forced to cease operations. Under such circumstances,
you may lose at least a portion of, and perhaps your entire, investment.
PRP
may cause undesirable side effects that could negatively impact its clinical trial results or limit its use, hindering further development,
subject us to possible product liability claims, and make it more difficult to commercialize PRP.
In
addition to the possibility that the clinical trials we hope to initiate for PRP could demonstrate a lack of efficacy, if we alternatively
identify adverse and undesirable side effects caused by it this will likely interrupt, delay or even halt our further development, or
possibly limit our planned therapeutic uses for it, and may even result in adverse regulatory action by the FDA or other regulatory authorities.
Moreover,
this may subject us to product liability claims by the individuals enrolled in our clinical trials; while we intend to obtain product
liability insurance in connection with our clinical trials, it is possible that the potential liability of any claims against us could
exceed the maximum amount of this coverage, or at least increase our premiums. Either would result in an increase in our operating expenses,
in turn making it more difficult to complete our clinical development, or in the suspension or termination of the clinical trial. Any
negative information concerning PRP, however unrelated to its composition or method of use, could also damage our chances to obtain regulatory
approval.
Even
if we are able to complete PRP’s development and receive regulatory approvals, undesirable side effects could prevent us from achieving
or maintaining market acceptance of the product or substantially increase the costs and expenses of commercializing it.
Because
successful development of our products is uncertain, our results of operations may be materially harmed.
Our
development of PRP and future product candidates is subject to the risks of failure inherent in the development of new pharmaceutical
products that are based on new technologies, including but not limited to delays in product development, clinical testing or manufacturing;
unplanned and higher expenditures; adverse findings relating to safety or efficacy; failure to receive regulatory approvals; the emergence
of superior or equivalent products; an inability by us or one of our collaborators to manufacture our product candidates on a commercial
scale on our own, or in collaboration with third parties; and, ultimately, a failure to achieve market acceptance.
Because
of these risks, our development efforts may not result in PRP, or any other product we attempt to develop, becoming commercially viable.
If even one aspect of these development efforts is not successfully completed, required regulatory approvals will not be obtained, or
if any approved products are not commercialized successfully, our business, financial condition and results of operations will be materially
harmed.
A
variety of factors, either alone or in concert with each other, could result in our clinical trials of PRP being delayed or unsuccessful.
While
we have conducted a variety of preclinical studies, which we have concluded provide evidence to support the potential therapeutic utility
of PRP, comprehensive human clinical trials in order to demonstrate the product’s safety, tolerability and efficacy will now need
to be completed. Clinical testing is expensive, difficult to design and implement, can take many years to complete and is uncertain as
to outcome. A failure of one or more clinical trials can occur at any stage of testing. The outcome of preclinical testing and even early
clinical trials may not be predictive of the success of later clinical trials, and interim results of a clinical trial do not necessarily
predict final results. Moreover, preclinical and clinical data are often susceptible to varying interpretations and analyses, and many
companies that have believed their product candidates performed satisfactorily in preclinical studies and clinical trials have nonetheless
failed to obtain marketing approval of their products.
Among
the numerous unforeseen events that may occur during, or as a result of, clinical trials that alone or in concert with each other could
either delay or prevent our ability to receive marketing approval or commercialize PRP are the following:
| |
● |
regulators
or institutional review boards may not authorize us or our investigators to commence a clinical trial or conduct a clinical trial
at a prospective trial site; |
| |
|
|
| |
● |
we
may have delays in reaching or fail to reach an agreement on acceptable clinical trial contracts or clinical trial protocols with
prospective trial sites; |
| |
|
|
| |
● |
as
noted previously, clinical trials of PRP may produce negative or inconclusive results, and we may decide, or regulators may require
us, to conduct additional clinical trials or abandon product development altogether; |
| |
|
|
| |
● |
the
number of patients required for clinical trials may be larger than we anticipate, enrollment in these clinical trials may be slower
than we anticipate, or participants may drop out of these clinical trials at a higher rate than we anticipate; |
| |
|
|
| |
● |
our
third-party contractors may fail to comply with regulatory requirements or fail to meet their contractual obligations to us in a
timely manner, or at all; |
| |
|
|
| |
● |
regulators
or institutional review boards may require that we or our investigators suspend or terminate clinical research for various reasons,
including noncompliance with regulatory requirements or a finding that the participants are being exposed to unacceptable health
risks; |
| |
|
|
| |
● |
the
cost of clinical trials may be greater than we anticipate; |
| |
|
|
| |
● |
the
supply or quality of PRP or other materials necessary to conduct its clinical trials may be insufficient or inadequate; and |
| |
|
|
| |
● |
PRP
may, as also noted above, have undesirable side effects or other unexpected characteristics, causing us or our investigators, regulators
or institutional review boards to suspend or terminate the trials. |
If
we are required to conduct additional clinical trials or other testing of PRP beyond those that we currently contemplate, if we are unable
to successfully complete clinical trials of PRP or other testing, if the results of these trials or tests are not positive or are only
modestly positive or if there are safety concerns, we may:
| |
● |
be
delayed in obtaining marketing approval; |
| |
|
|
| |
● |
not
obtain marketing approval at all; |
| |
|
|
| |
● |
obtain
approval for indications or patient populations that are not as broad as intended or desired; |
| |
● |
obtain
approval with labeling that includes significant use or distribution restrictions or safety warnings, including boxed warnings; |
| |
|
|
| |
● |
be
subject to additional post-marketing testing requirements; or |
| |
|
|
| |
● |
fail
to obtain that degree of market acceptance necessary for commercial success. |
Any
delay in, or termination of, our clinical trials may result in increased development costs, which would very likely cause the market
price of our shares to decline and severely limit our ability to obtain additional financing and, ultimately, our ability to commercialize
our products and generate product revenues. This in turn would likely materially harm our business, financial condition and operating
results, and possibly lead us to cease operations.
If
we fail to obtain regulatory approval in jurisdictions outside the United States, we will not be able to market PRP in those jurisdictions.
We
intend to seek regulatory approval for PRP in the United Kingdom, Europe, Australia and/or other countries outside of the United States
and expect that these countries will be important markets for our product, if approved. Marketing our product in these countries will
require separate regulatory approvals in each market and compliance with numerous and varying regulatory requirements. The regulations
that apply to the conduct of clinical trials and approval procedures vary from country to country and may require additional testing.
Moreover, the time required to obtain approval may differ from that required to obtain FDA approval.
If,
in the future, we are unable to establish sales and marketing capabilities or enter into agreements with third parties to sell and market
PRP, we may not be successful in commercializing our product candidates if and when they are approved.
We
do not have a sales or marketing infrastructure and have no experience in the sale, marketing or distribution of pharmaceutical products.
To achieve commercial success for PRP or any other approved product, we must either develop a sales and marketing organization or outsource
these functions to third parties. In the future, we may choose to build a focused sales and marketing infrastructure to market or co-promote
some of our product candidates if and when they are approved.
There
are risks involved with both establishing our own sales and marketing capabilities and entering into arrangements with third parties
to perform these services. For example, recruiting and training a sales force is expensive and time consuming and could delay any product
launch. If the commercial launch of a product candidate for which we recruit a sales force and establish marketing capabilities is delayed
or does not occur for any reason, we would have prematurely or unnecessarily incurred these commercialization expenses. This may be costly,
and our investment would be lost if we cannot retain or reposition our sales and marketing personnel.
Factors
that may inhibit our efforts to commercialize our products on our own include:
| |
● |
our
inability to recruit and retain adequate numbers of effective sales and marketing personnel; |
| |
|
|
| |
● |
the
inability of sales personnel to obtain access to physicians or persuade an adequate number of physicians to prescribe any future
products; |
| |
|
|
| |
● |
the
lack of complementary products to be offered by sales personnel, which may put us at a competitive disadvantage relative to companies
with more extensive product lines; and |
| |
|
|
| |
● |
unforeseen
costs and expenses associated with creating an independent sales and marketing organization. |
If
we enter into arrangements with third parties to perform sales, marketing and distribution services, our product revenues or the profitability
of these product revenues to us are likely to be lower than if we were to market and sell any products that we develop ourselves. In
addition, we may not be successful in entering into arrangements with third parties to sell and market our product candidates or may
be unable to do so on terms that are favorable to us. We likely will have little control over such third parties, and any of them may
fail to devote the necessary resources and attention to sell and market our products effectively. If we do not establish sales and marketing
capabilities successfully, either on our own or in collaboration with third parties, we will not be successful in commercializing PRP.
We
face substantial competition, which may result in others discovering, developing or commercializing products before or more successfully
than we do.
The
development and commercialization of new drug products is highly competitive. We face competition with respect to our current product
candidate and will face competition with respect to any product candidates that we may seek to develop or commercialize in the future
from major pharmaceutical companies, specialty pharmaceutical companies and biotechnology companies worldwide. There are a number of
large pharmaceutical and biotechnology companies that currently market and sell products or are pursuing the development of products
for the treatment of the disease indications for which we are developing our product candidates. Some of these competitive products and
therapies are based on scientific approaches that target and eradicate cancer stem cells to treat metastatic cancer. Potential competitors
also include academic institutions, government agencies and other public and private research organizations that conduct research, seek
patent protection and establish collaborative arrangements for research, development, manufacturing and commercialization.
We
are developing PRP for the treatment of pancreatic, ovarian and colorectal cancer. There are a variety of available therapies marketed
for cancer. In many cases, these drugs are administered in combination to enhance efficacy. Some of these drugs are branded and subject
to patent protection, and others are available on a generic basis. Many of these approved drugs are well-established therapies and are
widely accepted by physicians, patients and third-party payors. Insurers and other third-party payors may also encourage the use of generic
products. We expect that if our product candidate is approved, it will be priced at a significant premium over competitive generic products.
This may make it difficult for us to achieve our business strategy of using PRP in combination with existing therapies or replacing existing
therapies with PRP.
There
are also a number of products in clinical development by other parties to treat and prevent metastatic cancer. Our competitors may develop
products that are more effective, safer, more convenient or less costly than any that we are developing or that would render our product
candidate obsolete or non-competitive. In addition, our competitors may discover biomarkers that more efficiently measure their effectiveness
to treat and prevent metastatic cancer, which may give them a competitive advantage in developing potential products. Our competitors
may also obtain marketing approval from the FDA or other regulatory authorities for their products more rapidly than we may obtain approval
for ours, which could result in our competitors establishing a strong market position before we are able to enter the market.
Most
of our competitors have significantly greater financial resources and expertise in research and development, manufacturing, preclinical
testing, conducting clinical trials, obtaining regulatory approvals and marketing approved products than we do. Mergers and acquisitions
in the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of our
competitors. Smaller and other early-stage companies may also prove to be significant competitors, particularly through collaborative
arrangements with large and established companies. These third parties compete with us in recruiting and retaining qualified scientific
and management personnel, establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies
complementary to, or necessary for, our programs. In addition, to the extent that product or product candidates of our competitors demonstrate
serious adverse side effects or are determined to be ineffective in clinical trials, the development of our product candidates could
be negatively impacted.
Even
if we are able to commercialize PRP, we will need to seek approval for reimbursement before it can be marketed, and it may become subject
to unfavorable pricing regulations, third-party reimbursement practices or healthcare reform initiatives, which would harm our business.
The
regulations that govern marketing approvals, pricing and reimbursement for new drug products vary widely from country to country. In
the United States, recently passed legislation may significantly change the approval requirements in ways that could involve additional
costs and cause delays in obtaining approvals. Some countries require approval of the sale price of a drug before it can be marketed.
In many countries, the pricing review period begins after marketing or product licensing approval is granted. In some foreign markets,
prescription pharmaceutical pricing remains subject to continuing governmental control even after initial approval is granted. As a result,
we might obtain marketing approval for PRP in a particular country, but then be subject to price regulations that delay our commercial
launch of it, possibly for lengthy time periods, and negatively impact the revenues we are able to generate from the sale of PRP in that
country. Adverse pricing limitations may hinder our ability to recoup our investment in PRP, even after it has obtained marketing approval.
Our
ability to commercialize PRP successfully also will depend in part on the extent to which reimbursement for it will be available from
government health administration authorities, private health insurers and other organizations. Government authorities and third-party
payors, such as private health insurers and health maintenance organizations, decide which medications they will pay for and establish
reimbursement levels. A primary trend in the U.S. healthcare industry and elsewhere is cost containment. Government authorities and third-party
payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular medications. Increasingly,
third-party payors are requiring that drug companies provide them with predetermined discounts from list prices and are challenging the
prices charged for medical products. We cannot be sure that reimbursement will be available for PRP that we commercialize and, if reimbursement
is available, the level of reimbursement. Reimbursement may impact the demand for, or the price of, PRP. Obtaining reimbursement for
it may be particularly difficult because of the higher prices often associated with drugs administered under the supervision of a physician.
If reimbursement is not available or is available only to limited levels, we may not be able to successfully commercialize PRP.
There
may be significant delays in obtaining reimbursement for newly approved drugs, and coverage may be more limited than the purposes for
which the drug is approved by the FDA or similar regulatory authorities outside the United States. Moreover, eligibility for reimbursement
does not imply that any drug will be paid for in all cases or at a rate that covers our costs, including research, development, manufacture,
sale and distribution. Interim reimbursement levels for new drugs, if applicable, may also not be sufficient to cover our costs and may
not be made permanent. Reimbursement rates may vary according to the use of the drug and the clinical setting in which it is used, may
be based on reimbursement levels already set for lower cost drugs and may be incorporated into existing payments for other services.
Net prices for drugs may be reduced by mandatory discounts or rebates required by government healthcare programs or private payors and
by any future relaxation of laws that presently restrict imports of drugs from countries where they may be sold at lower prices than
in the United States. Third-party payors often rely upon Medicare coverage policy and payment limitations in setting their own reimbursement
policies. Our inability to promptly obtain coverage and profitable payment rates from both government-funded and private payors for any
approved products that we develop could have a material adverse effect on our operating results, our ability to raise capital needed
to commercialize products and our overall financial condition.
RISKS
RELATED TO OUR DEPENDENCE ON THIRD PARTIES
We
will depend on collaborations with third parties for the development and commercialization of PRP and other product candidates, and these
collaborations may be unsuccessful.
We
currently seek third-party collaborators for the development and commercialization of PRP, contract manufacturers (CMOs), contract research
organizations (CROs), regulatory and development consultants, and hospitals for clinical trial sites. We intend to continue to rely on
third-party collaborators for current and future product candidates for the foreseeable future. Our likely collaborators for any collaboration
arrangements include large and mid-size pharmaceutical companies, regional and national pharmaceutical companies and biotechnology companies.
If we do enter into any such arrangements with any third parties, we will likely have limited control over the amount and timing of resources
that our collaborators dedicate to the development or commercialization of our product candidates. Our ability to generate revenues from
these arrangements will depend on our collaborators’ abilities to successfully perform the functions assigned to them in these
arrangements.
Collaborations
involving our product candidates would pose the following risks to us:
| |
● |
collaborators
have significant discretion in determining the efforts and resources that they will apply to these collaborations; |
| |
|
|
| |
● |
collaborators
may not pursue development and commercialization of our product candidates or may elect not to continue or renew development or commercialization
programs based on clinical trial results, changes in the collaborator’s strategic focus or available funding or external factors
such as an acquisition that diverts resources or creates competing priorities; |
| |
|
|
| |
● |
collaborators
may delay clinical trials, provide insufficient funding for a clinical trial program, stop a clinical trial or abandon a product
candidate, repeat or conduct new clinical trials or require a new formulation of a product candidate for clinical testing; |
| |
|
|
| |
● |
collaborators
could independently develop, or develop with third parties, products that compete directly or indirectly with our products or product
candidates if the collaborators believe that competitive products are more likely to be successfully developed or can be commercialized
under terms that are more economically attractive than ours; |
| |
|
|
| |
● |
collaborators
with marketing and distribution rights to one or more products may not commit sufficient resources to the marketing and distribution
of such product or products; |
| |
|
|
| |
● |
collaborators
may not properly maintain or defend our intellectual property rights or may use our proprietary information in such a way as to invite
litigation that could jeopardize or invalidate our proprietary information or expose us to potential litigation; |
| |
|
|
| |
● |
disputes
may arise between the collaborators and us that result in the delay or termination of the research, development or commercialization
of our products or product candidates or that result in costly litigation or arbitration that diverts management attention and resources;
and |
| |
|
|
| |
● |
collaborations
may be terminated and, if terminated, may result in a need for additional capital to pursue further development or commercialization
of the applicable product candidates. |
Collaboration
agreements may not lead to development or commercialization of product candidates in the most efficient manner or at all. If a present
or future collaborator of ours were to be involved in a business combination, the continued pursuit and emphasis on our product development
or commercialization program could be delayed, diminished or terminated.
If
we are not able to establish collaborations, we may have to alter our development and commercialization plans.
Our
potential commercialization of PRP will require substantial additional cash to fund clinical trial and other expenses. As noted above,
we may decide to collaborate with other pharmaceutical and biotechnology companies for the development and potential commercialization
of PRP and perhaps future product candidates as well.
We
face significant competition in seeking appropriate collaborators. Whether we reach a definitive agreement for collaboration will depend,
among other things, upon our assessment of the collaborator’s resources and expertise, the terms and conditions of the proposed
collaboration and the proposed collaborator’s evaluation of a number of factors. Those factors may include the design or results
of clinical trials, the likelihood of approval by the FDA or similar regulatory authorities outside the United States, the potential
market for the subject product candidate, the costs and complexities of manufacturing and delivering such product candidate to patients,
the potential of competing products, the existence of uncertainty with respect to our ownership of technology, which can exist if there
is a challenge to such ownership without regard to the merits of the challenge and industry and market conditions generally. The collaborator
may also consider alternative product candidates or technologies for similar indications that may be available to collaborate on and
whether such collaboration could be more attractive than the one with us for our product candidate. Collaborations are complex and time-consuming
to negotiate and document. In addition, there have been a significant number of recent business combinations among large pharmaceutical
companies that have resulted in a reduced number of potential future collaborators.
We
may not be able to negotiate collaborations on a timely basis, on acceptable terms, or at all. If we are unable to do so, we may have
to curtail the development of such product candidate, reduce or delay its development program or one or more of our other development
programs, delay its potential commercialization or reduce the scope of any sales or marketing activities, or increase our expenditures
and undertake development or commercialization activities at our own expense. If we elect to increase our expenditures to fund development
or commercialization activities on our own, we may need to obtain additional capital, which may not be available to us on acceptable
terms or at all. If we do not have sufficient funds, we may not be able to further develop our product candidates or bring them to market
and generate product revenue.
We
currently contract with a third party for the manufacture of PRP and this third party may not perform satisfactorily or at all, and our
reliance on any third-party for the supply of PRP carries material risks.
We
do not have any manufacturing facilities or personnel. We currently obtain all of our supply of PRP for clinical development through
our Manufacturing Service Agreement (the “MSA”) with Amatsigroup, and we expect to continue to rely on Amatsigroup for the
manufacture of clinical and, if necessary, commercial quantities of PRP. We anticipate that our payments to Amatsigroup under the MSA
will range between $2.5 million and $5.0 million over three years, when the finished drug product is manufactured and released for clinical
trials. The Company has spent a total of $1,689,146 of costs to date under this contract of which $49,854 was expensed in fiscal 2019,
$701,973 in fiscal 2018 and $937,319 in fiscal 2017. The MSA shall continue for a term of three years unless extended by mutual agreement
in writing. Either party to the MSA has the right to terminate. The MSA expired in 2019 and we believe it is likely a new agreement
will be established upon mutual satisfaction from both parties.
This
reliance on a third party includes the risk that we will not have sufficient quantities of PRP on hand at any given time, which could
delay, prevent or impair our development efforts.
PRP
and any other product that we may develop may compete with other product candidates and products for access to manufacturing facilities.
Although we believe that there are several potential alternative manufacturers who could manufacture PRP, we may incur added costs and
delays in identifying and qualifying any such replacement, as well as producing the drug product. In addition, we would then have to
enter into technical transfer agreements and share our know-how with the new third-party manufacturers, which can be time-consuming and
may result in delays.
Even
if we were able to quickly establish agreements with other third-party manufacturers, our general reliance on third-party manufacturers
entails many of the same risks as our agreement with Amatsigroup, including:
| |
● |
reliance
on the third party for regulatory compliance and quality assurance; |
| |
|
|
| |
● |
the
possible breach of the manufacturing agreement by the third party, including the misappropriation of our proprietary information,
trade secrets and know-how; |
| |
|
|
| |
● |
the
possible termination or nonrenewal of the agreement by the third party at a time that is costly or inconvenient for us; and |
| |
|
|
| |
● |
disruptions
to the operations of our manufacturers or suppliers caused by conditions unrelated to our business or operations, including the bankruptcy
of the manufacturer or supplier or a catastrophic event affecting our manufacturers or suppliers. |
Our
anticipated future dependence upon others for the manufacture of PRP may adversely affect our future profit margins and our ability to
commercialize any products that receive marketing approval on a timely and competitive basis. We intend to minimize this risk by tendering
our contract agreement with several third-party manufacturers with a plan to engage in a dual supplier strategy for the contract manufacture
of PRP.
RISKS
RELATED TO OUR INTELLECTUAL PROPERTY
If
we fail to comply with our obligations under any intellectual property licenses with third parties, we could lose license rights that
are important to our business.
We
are currently a party to a joint commercialization agreement with the University of Bath and hope to enter into other license agreements
in the future. If we fail to comply with the obligations included in any future license we may enter into in the future, such licensors
may have the right to terminate these agreements, in which event we might not be able to market any product that is covered by the agreements,
or to convert the exclusive licenses to non-exclusive licenses, which could materially adversely affect the value of the product candidate
being developed under these license agreements. As a general matter, termination of license agreements or reduction or elimination of
our licensed rights may result in our having to negotiate new or reinstated licenses with less favorable terms.
If
we are unable to obtain and maintain patent protection for our technology and products, or if any licensors are unable to obtain and
maintain patent protection for the technology or products that we may license from them in the future, or if the scope of the patent
protection obtained is not sufficiently broad, our competitors could develop and commercialize technology and products similar or identical
to ours, and our ability to successfully commercialize our technology and products may be adversely affected.
We
have thirty-five (35) granted patents and have thirty (30) patent applications either pending or under examination in major global jurisdictions.
Our future success depends in large part on our and, as applicable, our licensors’, ability to obtain and maintain patent protection
in the United States and other countries with respect to our proprietary technology. We cannot be certain that patents will be issued
in those countries where our applications are still under examination.
The
patent process is expensive and time-consuming, and we may not be able to file and prosecute all necessary or desirable patent applications
at a reasonable cost or in a timely manner. It is also possible that we will fail to identify patentable aspects of our research and
development output before it is too late to obtain patent protection.
The
patent position of biotechnology and pharmaceutical companies generally is highly uncertain, involves complex legal and factual questions
and has in recent years been the subject of much litigation. As a result, the issuance, scope, validity, enforceability and commercial
value of our patent rights are uncertain. Our pending and future patent applications may not result in patents being issued which protect
our technology or products or which effectively prevent others from commercializing competitive technologies and products. Changes in
either the patent laws or interpretation of the patent laws in the United States and other countries may diminish the value of our patents
or narrow the scope of our patent protection.
The
laws of foreign countries may not protect our rights to the same extent as the laws of the United States. Publications of discoveries
in the scientific literature often lag behind the actual discoveries, and patent applications in the United States and other jurisdictions
are typically not published until 18 months after filing. Therefore, we cannot be certain that we or our licensors were the first to
make the inventions claimed in our owned or licensed patents or pending patent applications, or that we or our licensors were the first
to file for patent protection of such inventions.
Assuming
the other requirements for patentability are met, in the United States, for patents that have an effective filing date prior to March
15, 2013, the first to make the claimed invention is entitled to the patent, while outside the United States, the first to file a patent
application is entitled to the patent. In March 2013, the United States transitioned to a first inventor to file system in which, assuming
the other requirements for patentability are met, the first inventor to file a patent application will be entitled to the patent. We
may be subject to a third party preissuance submission of prior art to the U.S. Patent and Trademark Office, or become involved in opposition,
derivation, reexamination, inter parties review or interference proceedings challenging our patent rights or the patent rights of others.
An adverse determination in any such submission, proceeding or litigation could reduce the scope of, or invalidate, our patent rights,
allow third parties to commercialize our technology or products and compete directly with us, without payment to us, or result in our
inability to manufacture or commercialize products without infringing third-party patent rights.
Even
if our owned and licensed patent applications issue as patents, they may not issue in a form that will provide us with any meaningful
protection, prevent competitors from competing with us or otherwise provide us with any competitive advantage. Our competitors may be
able to circumvent our owned or licensed patents by developing similar or alternative technologies or products in a non-infringing manner.
The
issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability, and our owned and licensed patents
may be challenged in the courts or patent offices in the United States and abroad. Such challenges may result in loss of exclusivity
or freedom to operate or in patent claims being narrowed, invalidated or held unenforceable, which could limit our ability to stop others
from using or commercializing similar or identical technology and products, or limit the duration of the patent protection of our technology
and products. Given the amount of time required for the development, testing and regulatory review of new product candidates, patents
protecting such candidates might expire before or shortly after such candidates are commercialized. As a result, our owned and licensed
patent portfolio may not provide us with sufficient rights to exclude others from commercializing products similar or identical to ours.
We
may become involved in lawsuits to protect or enforce our patents, which could be expensive, time consuming and unsuccessful.
Competitors
may infringe our patents. To counter infringement or unauthorized use, we may be required to file infringement claims, which can be expensive
and time consuming. In addition, in an infringement proceeding, a court may decide that a patent of ours is invalid or unenforceable
or may refuse to stop the other party from using the technology at issue on the grounds that our patents do not cover the technology
in question. An adverse result in any litigation proceeding could put one or more of our patents at risk of being invalidated or interpreted
narrowly. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there
is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. In addition, our
licensors may have rights to file and prosecute such claims and we are reliant on them.
Third
parties may initiate legal proceedings alleging that we are infringing their intellectual property rights, the outcome of which would
be uncertain and could have a material adverse effect on the success of our business.
Our
commercial success depends upon our ability and the ability of our collaborators to develop, manufacture, market and sell PRP and any
other product candidates and use our proprietary technologies without infringing the proprietary rights of third parties. We have yet
to conduct comprehensive freedom-to-operate searches to determine whether our use of certain of the patent rights owned by or licensed
to us would infringe patents issued to third parties. We may become party to, or threatened with, future adversarial proceedings or litigation
regarding intellectual property rights with respect to our products and technology, including interference proceedings before the U.S.
Patent and Trademark Office and their European Union and global equivalents. Third parties may assert infringement claims against us
based on existing patents or patents that may be granted in the future. If we are found to infringe a third party’s intellectual
property rights, we could be required to obtain a license from such third party to continue developing and marketing our products and
technology. However, we may not be able to obtain any required license on commercially reasonable terms or at all. Even if we were able
to obtain a license, it could be non-exclusive, thereby giving our competitors access to the same technologies licensed to us. We could
be forced, including by court order, to cease commercializing the infringing technology or product. In addition, we could be found liable
for monetary damages. A finding of infringement could prevent us from commercializing our product candidates or force us to cease some
of our business operations, which could materially harm our business. Claims that we have misappropriated the confidential information
or trade secrets of third parties could have a similar negative impact on our business.
Intellectual
property litigation could cause us to spend substantial resources and distract our personnel from their normal responsibilities.
Even
if resolved in our favor, litigation or other legal proceedings relating to intellectual property claims may cause us to incur significant
expenses and could distract our personnel from their normal responsibilities. In addition, there could be public announcements of the
results of hearings, motions or other interim proceedings or developments and if securities analysts or investors perceive these results
to be negative, it could have a substantial adverse effect on the price of our common stock. Such litigation or proceedings could substantially
increase our operating losses and reduce the resources available for development activities or any future sales, marketing or distribution
activities. We may not have sufficient financial or other resources to adequately conduct such litigation or proceedings. Some of our
competitors may be able to sustain the costs of such litigation or proceedings more effectively than we can because of their greater
financial resources. Uncertainties resulting from the initiation and continuation of patent litigation or other proceedings could have
a material adverse effect on our ability to compete in the marketplace.
If
we are unable to protect the confidentiality of our trade secrets, our business and competitive position would be harmed.
In
addition to seeking patents for some of our technology and products, we also rely on trade secrets, including unpatented know-how, technology
and other proprietary information, to maintain our competitive position. We seek to protect these trade secrets, in part, by entering
into non-disclosure and confidentiality agreements with parties who have access to them, such as our employees, corporate collaborators,
outside scientific collaborators, contract manufacturers, consultants, advisors and other third parties. We also enter into confidentiality
and invention or patent assignment agreements with our employees and consultants. Despite these efforts, any of these parties may breach
the agreements and disclose our proprietary information, including our trade secrets, and we may not be able to obtain adequate remedies
for such breaches. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming,
and the outcome is unpredictable. In addition, some courts inside and outside the United States are less willing or unwilling to protect
trade secrets. If any of our trade secrets were to be lawfully obtained or independently developed by a competitor, we would have no
right to prevent them from using that technology or information to compete with us. If any of our trade secrets were to be disclosed
to or independently developed by a competitor, our competitive position would be harmed.
RISKS
RELATED TO REGULATORY APPROVAL OF OUR PRODUCT CANDIDATES AND OTHER LEGAL COMPLIANCE MATTERS
If
we are not able to obtain, or if there are delays in obtaining, required regulatory approvals, we will not be able to commercialize PRP,
and our ability to generate revenue will be materially impaired.
PRP
and the activities associated with its development and commercialization, including design, testing, manufacture, safety, efficacy, recordkeeping,
labeling, storage, approval, advertising, promotion, sale and distribution, are subject to comprehensive regulation by the FDA and other
regulatory agencies in the United States and by comparable authorities in other countries. Failure to obtain marketing approval for PRP
will prevent us from commercializing it. We have not received approval to market PRP or any other product candidate from regulatory authorities
in any jurisdiction. We have only limited experience in filing and supporting the applications necessary to gain marketing approvals
and expect to rely on third-party contract research organizations to assist us in this process. Securing FDA approval requires the submission
of extensive preclinical and clinical data and supporting information to the FDA for each therapeutic indication to establish PRP’s
safety and efficacy. Securing FDA approval also requires the submission of information about the product manufacturing process to, and
inspection of manufacturing facilities by, the FDA. PRP may not be effective, may be only moderately effective or may prove to have undesirable
or unintended side effects, toxicities or other characteristics that may preclude our obtaining marketing approval or prevent or limit
commercial use.
The
process of obtaining marketing approvals, both in the United States and abroad, is expensive, may take many years if additional clinical
trials are required, if approval is obtained at all, and can vary substantially based upon a variety of factors, including the type,
complexity and novelty of the product candidates involved. Changes in marketing approval policies during the development period, changes
in or the enactment of additional statutes or regulations, or changes in regulatory review for each submitted product application, may
cause delays in the approval or rejection of an application. The FDA has substantial discretion in the approval process and may refuse
to accept any application or may decide that our data is insufficient for approval and require additional preclinical, clinical or other
studies. In addition, varying interpretations of the data obtained from preclinical and clinical testing could delay, limit or prevent
marketing approval of a product candidate. Any marketing approval we ultimately obtain may be limited or subject to restrictions or post-approval
commitments that render the approved product not commercially viable.
If
we experience delays in obtaining approval or if we fail to obtain approval of PRP, the commercial prospects for PRP may be harmed and
our ability to generate revenues will be materially impaired.
Failure
to obtain marketing approval in international jurisdictions would prevent PRP from being marketed abroad.
We
intend to seek regulatory approval for PRP in a number of countries outside of the United States and expect that these countries will
be important markets for it, if approved. In order to market and sell our products in the European Union, the UK, Australia and many
other jurisdictions, we or our third-party collaborators must obtain separate marketing approvals and comply with numerous and varying
regulatory requirements. The approval procedure varies among countries and can involve additional testing. The time required to obtain
approval may differ substantially from that required to obtain FDA approval. The regulatory approval process outside the United States
generally includes all of the risks associated with obtaining FDA approval. In addition, in many countries outside the United States,
it is required that the product be approved for reimbursement before the product can be approved for sale in that country. We or these
third parties may not obtain approvals from regulatory authorities outside the United States on a timely basis, if at all. Approval by
the FDA does not ensure approval by regulatory authorities in other countries or jurisdictions, and approval by one regulatory authority
outside the United States does not ensure approval by regulatory authorities in other countries or jurisdictions or by the FDA. We may
not be able to file for marketing approvals and may not receive necessary approvals to commercialize our products in any market.
PRP
or any other product candidate for which we obtain marketing approval could be subject to restrictions or withdrawal from the market
and we may be subject to penalties if we fail to comply with regulatory requirements or if we experience unanticipated problems with
our products, when and if any of them are approved.
PRP,
or any other product candidate for which we obtain marketing approval, along with the manufacturing processes, post-approval clinical
data, labeling, advertising and promotional activities for such product, will be subject to continual requirements of and review by the
FDA and other regulatory authorities. These requirements include submissions of safety and other post-marketing information and reports,
registration and listing requirements, cGMP requirements relating to quality control, quality assurance and corresponding maintenance
of records and documents, requirements regarding the distribution of samples to physicians and recordkeeping. Even if marketing approval
of a product candidate is granted, the approval may be subject to limitations on the indicated uses for which the product may be marketed
or to the conditions of approval, or contain requirements for costly post-marketing testing and surveillance to monitor the safety or
efficacy of the product. The FDA closely regulates the post-approval marketing and promotion of drugs to ensure drugs are marketed only
for the approved indications and in accordance with the provisions of the approved labeling. The FDA imposes stringent restrictions on
manufacturers’ communications regarding off-label use and if we do not market our products for their approved indications, we may
be subject to enforcement action for off-label marketing.
In
addition, later discovery of previously unknown problems with our products, manufacturers or manufacturing processes, or failure to comply
with regulatory requirements, may yield various results, including:
| |
● |
restrictions
on such products, manufacturers or manufacturing processes; |
| |
● |
restrictions
on the labeling or marketing of a product; |
| |
|
|
| |
● |
restrictions
on product distribution or use; |
| |
|
|
| |
● |
requirements
to conduct post-marketing clinical trials; |
| |
|
|
| |
● |
warning
or untitled letters; |
| |
|
|
| |
● |
withdrawal
of the products from the market; |
| |
|
|
| |
● |
refusal
to approve pending applications or supplements to approved applications that we submit; |
| |
|
|
| |
● |
recall
of products; |
| |
● | fines, restitution or disgorgement of profits or revenue; |
| |
|
|
| |
● |
suspension
or withdrawal of marketing approvals; |
| |
|
|
| |
● |
refusal
to permit the import or export of our products; |
| |
|
|
| |
● |
product
seizure; or |
| |
|
|
| |
● |
injunctions
or the imposition of civil or criminal penalties. |
Our
current attempts to both expand our patent protection and seek regulatory approvals from multiple countries, as well as our future relationships
with customers and third-party payors will be subject to applicable anti-kickback, fraud and abuse and other laws and regulations, which
could expose us to criminal sanctions, civil penalties, contractual damages, reputational harm and diminished profits and future earnings.
As
we seek to obtain patent protection from multiple jurisdictions and eventually to seek marketing approval for PRP in those counties,
we are and will continue to be subject to the Foreign Corrupt Practices Act, which makes it illegal for any U.S. business, even one like
Propanc that is physically located in another country, to influence foreign officials with personal payments and rewards.
Moreover,
healthcare providers, physicians and third-party payors will play a primary role in the recommendation and prescription of PRP and any
other product candidate for which we obtain marketing approval. Our future arrangements with third-party payors and customers may expose
us to broadly applicable fraud and abuse and other healthcare laws and regulations that may constrain the business or financial arrangements
and relationships through which we market, sell and distribute our products for which we obtain marketing approval. Restrictions under
applicable federal and state healthcare laws and regulations, include the following:
| |
● |
the
federal healthcare anti-kickback statute prohibits, among other things, persons from knowingly and willfully soliciting, offering,
receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce or reward either the referral of an individual
for, or the purchase, order or recommendation of, any good or service, for which payment may be made under federal and state healthcare
programs such as Medicare and Medicaid; |
| |
|
|
| |
● |
the
federal False Claims Act imposes criminal and civil penalties, including civil whistleblower or qui tam actions, against individuals
or entities for knowingly presenting, or causing to be presented, to the federal government, claims for payment that are false or
fraudulent or making a false statement to avoid, decrease or conceal an obligation to pay money to the federal government; |
| |
|
|
| |
● |
the
federal Health Insurance Portability and Accountability Act of 1996, as amended by the Health Information Technology for Economic
and Clinical Health Act, imposes criminal and civil liability for executing a scheme to defraud any healthcare benefit program and
also imposes obligations, including mandatory contractual terms, with respect to safeguarding the privacy, security and transmission
of individually identifiable health information; |
| |
|
|
| |
● |
the
federal false statements statute prohibits knowingly and willfully falsifying, concealing or covering up a material fact or making
any materially false statement in connection with the delivery of or payment for healthcare benefits, items or services; |
| |
|
|
| |
● |
the
federal transparency requirements under the Health Care Reform Law requires manufacturers of drugs, devices, biologics and medical
supplies to report to the Department of Health and Human Services information related to physician payments and other transfers of
value and physician ownership and investment interests; and |
| |
|
|
| |
● |
analogous
state laws and regulations, such as state anti-kickback and false claims laws, may apply to sales or marketing arrangements and claims
involving healthcare items or services reimbursed by non-governmental third-party payors, including private insurers, and some state
laws require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the
relevant compliance guidance promulgated by the federal government in addition to requiring drug manufacturers to report information
related to payments to physicians and other health care providers or marketing expenditures. |
Efforts
to ensure that our business arrangements with third parties will comply with applicable healthcare laws and regulations will involve
substantial costs. It is possible that governmental authorities will conclude that our business practices may not comply with current
or future statutes, regulations or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations
are found to be in violation of any of these laws or any other governmental regulations that may apply to us, we may be subject to significant
civil, criminal and administrative penalties, damages, fines and exclusion from government funded healthcare programs, such as Medicare
and Medicaid, and the curtailment or restructuring of our operations. If any of the physicians or other providers or entities with whom
we expect to do business are found to be not in compliance with applicable laws, they may be subject to criminal, civil or administrative
sanctions, including exclusions from government funded healthcare programs.
Recently
enacted and future legislation, particularly in the United States, may increase the difficulty and cost for us to obtain marketing approval
of and commercialize PRP and affect the prices we may obtain.
In
the United States and some foreign jurisdictions there have been many legislative and regulatory changes and proposed changes regarding
the healthcare system that could prevent or delay marketing approval of our product candidates, restrict or regulate post-approval activities
and affect our ability to profitably sell any product candidates for which we obtain marketing approval.
In
the United States, the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (“Medicare Modernization Act”),
changed the way Medicare covers and pays for pharmaceutical products. The legislation expanded Medicare coverage for drug purchases by
the elderly and introduced a new reimbursement methodology based on average sales prices for physician-administered drugs. In addition,
this legislation provided authority for limiting the number of drugs that will be covered in any therapeutic class. Cost reduction initiatives
and other provisions of this legislation could decrease the coverage and price that we receive for any approved products. While the Medicare
Modernization Act applies only to drug benefits for Medicare beneficiaries, private payors often follow Medicare coverage policy and
payment limitations in setting their own reimbursement rates. Therefore, any reduction in reimbursement that results from the Medicare
Modernization Act may result in a similar reduction in payments from private payors.
In
March 2010, President Obama signed into law the Patient Protection and Affordable Care Act (“Affordable Care Act”), a sweeping
law intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud
and abuse, add new transparency requirements for health care and health insurance industries, impose new taxes and fees on the health
industry and impose additional health policy reforms. Among other things, the Affordable Care Act revised the definition of “average
manufacturer price” for reporting purposes, which could increase the amount of Medicaid drug rebates to states, and it imposed
a significant annual fee on companies that manufacture or import branded prescription drug products.
At
present, the future of the Affordable Care Act is the subject of significant debate in the U.S. Congress, with proposals to either partially
or entirely repeal it being considered and the likelihood that there will be a new law to replace it is uncertain. It is not yet possible
for us to determine the impact, if any, the enactment of any of these proposals will have on our future ability to obtain approval of
or commercialize PRP.
The
UK’s decision to leave the European Union could significantly increase regulatory burdens on obtaining approvals for PRP within
the UK.
On
March 29, 2017, the UK invoked Article 50 of Lisbon Treaty to initiate complete withdrawal from the European Union which was effectuated
on January 31, 2020, and therefore, the regulatory drug approval process in that country may be significantly different from the current
drug regulatory policies in the European Union. We currently are considering holding our clinical trials in the UK, among other countries,
and therefore this event could significantly impact our efforts to successfully bring PRP to market. It is not yet possible for us to
determine the impact of the UK’s withdrawal from the European Union, but any additional costs or delays in obtaining approvals
may hinder our ability to conduct clinical trials or market PRP in the UK.
RISKS
RELATING TO EMPLOYEE MATTERS AND MANAGING GROWTH
Our
future success depends on our ability to retain our chief executive officer and our chief scientific officer and, as we continue to develop
and grow as a company, to attract, retain and motivate qualified personnel.
We
are highly dependent on our management team, specifically Mr. James Nathanielsz, our Chief Executive Officer, Chief Financial Officer,
and Dr. Julian Kenyon, our director who also serves as our chief scientific officer in a non-executive officer capacity. While we have
a current employment agreement with Mr. Nathanielsz and a director agreement with Dr. Kenyon, both such employment agreement and director
agreement permit each of the respective parties thereto to terminate such agreements upon notice. If we lose this key employee and/or
the services of our other director, our business will suffer and we may have to cease operations.
Recruiting
and retaining qualified scientific, clinical, manufacturing and sales and marketing personnel will also be critical to our future success,
as we continue to develop PRP and attempt to grow as a company. We may not be able to attract and retain these personnel on acceptable
terms given the competition among numerous pharmaceutical and biotechnology companies for similar personnel. We also experience competition
for the hiring of scientific and clinical personnel from universities and research institutions. In addition, we rely on consultants
and advisors, including scientific and clinical advisors, to assist us in formulating our research and development and commercialization
strategy. Our consultants and advisors, including our scientific co-founders, may be employed by employers other than us and may have
commitments under consulting or advisory contracts with other entities that may limit their availability to us.
We
expect to expand our development, regulatory and future sales and marketing capabilities, and as a result, we may encounter difficulties
in managing our growth, which could disrupt our operations.
We
expect to experience significant growth in the number of our employees and the scope of our operations, particularly in the areas of
drug development, regulatory affairs and sales and marketing. To manage our anticipated future growth, we must continue to implement
and improve our managerial, operational and financial systems, expand our facilities and continue to recruit and train additional qualified
personnel. Due to our limited financial resources and the limited experience of our management team in managing a company with such anticipated
growth, we may not be able to effectively manage the expansion of our operations or recruit and train additional qualified personnel.
The physical expansion of our operations may lead to significant costs and may divert our management and business development resources.
Any inability to manage growth could delay the execution of our business plans or disrupt our operations.
We
have identified material weaknesses in our internal control over financial reporting that, if not properly remediated, could result in
material misstatements in our consolidated financial statements in future periods.
In
connection with the audits of our consolidated financial statements for the fiscal years ended June 30, 2021 and 2020, and in accordance
with management’s assessments of internal controls over financial reporting, we identified certain deficiencies relating to our
internal control over financial reporting that constitute a material weakness under the Internal Control Integrated Framework issued
by COSO in 2013. A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting
such that there is a reasonable possibility that a material misstatement of the company’s annual or interim financial statements
will not be prevented or detected on a timely basis. A deficiency in internal control exists when the design or operation of a control
does not allow management or employees, in the normal course of performing their assigned functions, to prevent or detect misstatements
on a timely basis.
The
following material weaknesses in our internal control over financial reporting continued to exist at June 30, 2021 and currently:
| |
● |
we
do not have written documentation of our internal control policies and procedures. Written documentation of key internal controls
over financial reporting is a requirement of Section 404 of the Sarbanes-Oxley Act of 2002 (the “Sarbanes-Oxley Act”); |
| |
|
|
| |
● |
we
do not have sufficient segregation of duties within accounting functions, which is a basic internal control. Due to our limited size
and early-stage nature of operations, segregation of all conflicting duties may not always be possible and may not be economically
feasible; however, to the extent possible, the initiation of transactions, the custody of assets and the recording of transactions
should be performed by separate individuals; |
| |
|
|
| |
● |
lack
of independent audit committee of our board of directors; and |
| |
|
|
| |
● |
insufficient
monitoring and review controls over the financial reporting closing process, including the lack of individuals with current knowledge
of U.S. GAAP. |
We
outsource certain functions that would normally be performed by a principal financial officer to assist us in implementing the necessary
financial controls over the financial reporting and the utilization of internal management and staff to effectuate these controls.
We
believe that these material weaknesses primarily relate, in part, to our lack of sufficient staff with appropriate training in U.S. GAAP
and U.S. Securities and Exchange Commission (the “SEC”) rules and regulations with respect to financial reporting functions,
and the lack of robust accounting systems, as well as the lack of sufficient resources to hire such staff and implement these accounting
systems.
We
plan to take a number of actions in the future to correct these material weaknesses including, but not limited to, establishing an audit
committee of our board of directors comprised of at least two independent directors, adding additional experienced accounting and financial
personnel and retaining third-party consultants to review our internal controls and recommend improvements, subject to receiving sufficient
additional capital. If we receive sufficient capital, we hope to increase the chief financial officer’s role from part-time to
full-time as the next step in building out our accounting department. We will need to take additional measures to fully mitigate these
issues, and the measures we have taken, and expect to take, to improve our internal controls may not be sufficient to (1) address the
issues identified, (2) ensure that our internal controls are effective or (3) ensure that the identified material weakness or other material
weaknesses will not result in a material misstatement of our annual or interim financial statements. In addition, other material weaknesses
may be identified in the future. If we are unable to correct deficiencies in internal controls in a timely manner, our ability to record,
process, summarize and report financial information accurately and within the time periods specified in the rules and forms of the SEC
will be adversely affected. This failure could negatively affect the market price and trading liquidity of our common stock, cause investors
to lose confidence in our reported financial information, subject us to civil and criminal investigations and penalties, and generally
materially and adversely impact our business and financial condition.
If
we fail to implement and maintain an effective system of internal control over financial reporting, we may not be able to accurately
report our financial results or prevent fraud.
Effective
internal controls over financial reporting are necessary for us to provide reliable financial reports and, together with adequate disclosure
controls and procedures, are designed to prevent fraud. Any failure to implement required new or improved controls, or difficulties encountered
in their implementation could cause us to fail to meet our reporting obligations. In addition, any testing by us conducted in connection
with Section 404 of the Sarbanes-Oxley Act, or the subsequent testing by our independent registered public accounting firm, if and when
required, may reveal additional deficiencies in our internal controls over financial reporting that are deemed to be material weaknesses
or that may require prospective or retroactive changes to our consolidated financial statements or identify other areas for further attention
or improvement. If in the future we identify other material weaknesses in our internal control over financial reporting, including at
some of our acquired companies, if we are unable to comply with the requirements of Section 404 in a timely manner or assert that our
internal control over financial reporting is effective, or if our independent registered public accounting firm is unable to express
an opinion as to the effectiveness of our internal control over financial reporting, investors may lose confidence in the accuracy and
completeness of our financial reports and the market price of our common stock could be negatively affected, and we could become subject
to investigations by the stock exchange on which our securities are then listed, the SEC, or other regulatory authorities, which could
require additional financial and management resources. Inferior internal controls could also cause investors to lose confidence in our
reported financial information, which could have a negative effect on the trading price of our common stock.
Additionally,
we currently do not have an internal audit group nor an audit committee of our board of directors, and we will eventually need to hire
additional accounting and financial staff with appropriate public company experience and technical accounting knowledge to have effective
internal controls for financial reporting.
We
will continue to incur significant increased costs as a result of operating as a public company.
As
a public company, we will continue to incur significant legal, accounting and other expenses. For example, we are subject to mandatory
reporting requirements of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), which require, among other
things, that we continue to file with the SEC annual, quarterly and current reports with respect to our business and financial condition.
We have incurred and will continue to incur costs associated with the preparation and filing of these SEC reports. In addition, the Sarbanes-Oxley
Act, as well as rules subsequently implemented by the SEC, the Dodd-Frank Wall Street Reform and Consumer Protection Act (the “Dodd-Frank
Act”) and national stock exchanges have imposed various other requirements on public companies. Stockholder activism, the current
political environment and the current high level of government intervention and regulatory reform may lead to substantial new regulations
and disclosure obligations, which may lead to additional compliance costs and impact (in ways we cannot currently anticipate) the manner
in which we operate our business. Our management and other personnel will need to devote a substantial amount of time to these compliance
initiatives. Moreover, these rules and regulations have and will continue to increase our legal and financial compliance costs and will
make some activities more time-consuming and costly. For example, we will incur additional expense to increase our director and officer
liability insurance.
In
addition, if and when we cease to be a smaller reporting company and become subject to Section 404(b) of the Sarbanes-Oxley Act, we will
be required to furnish an attestation report on internal control over financial reporting issued by our independent registered public
accounting firm. To achieve compliance with Section 404 within the prescribed time period, we will continue to be engaged in a process
to document and evaluate our internal control over financial reporting, which is both costly and challenging. In this regard, we will
need to dedicate substantially greater internal resources, potentially engage outside consultants and adopt a detailed work plan to assess
and document the adequacy of internal control over financial reporting, continue steps to improve control processes as appropriate, validate
through testing that controls are functioning as documented and implement a continuous reporting and improvement process for internal
control over financial reporting. Despite our efforts, there is a risk that our independent registered public accounting firm, when required,
will not be able to conclude within the prescribed timeframe that our internal control over financial reporting is effective as required
by Section 404. This could result in an adverse reaction in the financial markets due to a loss of confidence in the reliability of our
financial statements.
Judgments
that our stockholders obtain against us may not be enforceable.
Substantially
all of our assets are located outside of the United States. In addition, our Chief Executive Officer/Chief Financial Officer, James Nathanielsz,
and our independent director Josef Zelinger, reside in Australia and our other director, Dr. Julian Kenyon, resides in the UK. As a result,
it may be difficult for you to effect service of process within the United States upon these persons. It is uncertain whether the courts
of Australia or the UK would recognize or enforce judgments of the United States or state courts against us or such persons predicated
upon the civil liability provisions of the laws of the United States or any state.
RISKS
RELATED TO OUR COMMON STOCK
The
market price of our common stock may continue to be highly volatile, you may not be able to resell your shares at or above the public
offering price and you could lose all or part of your investment.
The
trading price of our common stock may continue to be highly volatile. Our stock price could continue to be subject to wide fluctuations
in response to a variety of factors, including the following:
| |
● |
actual
or anticipated results of our clinical trials; |
| |
|
|
| |
● |
actions
of securities analysts who initiate or maintain coverage of us, changes in financial estimates by any securities analysts who follow
our company, or our failure to meet these estimates or the expectations of investors; |
| |
|
|
| |
● |
issuance
of our equity and/or debt securities, or disclosure or announcements relating thereto; |
| |
|
|
| |
● |
additional
shares of our common stock being sold into the market by us or our existing stockholders and/or holders of convertible debt or the
anticipation of such sales; |
| |
|
|
| |
● |
stock
market valuations of companies in our industry; |
| |
|
|
| |
● |
price
and volume fluctuations in the overall stock market, including as a result of trends in the economy as a whole; |
| |
|
|
| |
● |
lawsuits
threatened or filed against us; |
| |
|
|
| |
● |
regulatory
developments in the United States and foreign countries applicable to biotech and biopharma companies; and |
| |
|
|
| |
● |
other
events or factors, including those resulting from war or incidents of terrorism, or responses to these events. |
The
stock markets in general, and the small-cap biotech market, in particular, have experienced extreme price and volume fluctuations in
recent years that have significantly affected the quoted prices of the securities of many companies, including companies in our industry.
The changes often appear to occur without regard to specific operating performance. The price of our shares of common stock could fluctuate
based upon factors that have little or nothing to do with our company and these fluctuations could materially reduce our share price.
Broad market, clinical trial results and industry factors may negatively affect the market price of our common stock, regardless of our
actual operating performance.
Currently
there is a limited public market for our common stock, and we cannot predict the future prices or the amount of liquidity of our common
stock.
Currently,
there is a limited public market for our common stock. Our common stock is quoted on the OTC QB Marketplace under the symbol “PPCB.”
However, the OTC QB is not a liquid market in contrast to the major stock exchanges. We cannot assure you as to the liquidity or the
future market prices of our common stock if a market does develop. If an active market for our common stock does not develop, the fair
market value of our common stock could be materially adversely affected. We cannot predict the future prices of our common stock.
The
designation of our common stock as a “penny stock” would limit the liquidity of our common stock.
Our
common stock may be deemed a “penny stock” (as that term is defined under Rule 3a51-1 of the Exchange Act) in any market
that may develop in the future. Generally, a “penny stock” is a common stock that is not listed on a securities exchange
and trades for less than $5.00 a share. Prices often are not available to buyers and sellers and the market may be very limited. Broker-dealers
who sell penny stocks must provide purchasers of these stocks with a standardized risk-disclosure document prepared by the SEC. The document
provides information about penny stocks and the nature and level of risks involved in investing in the penny stock market. A broker must
also provide purchasers with bid and offer quotations and information regarding broker and salesperson compensation and make a written
determination that the penny stock is a suitable investment for the purchaser and obtain the purchaser’s written agreement to the
purchase. Many brokers choose not to participate in penny stock transactions. Because of the penny stock rules, there may be less trading
activity in any market that develops for our common stock in the future and stockholders are likely to have difficulty selling their
shares.
Trading
in our common stock on the OTC QB Marketplace has been subject to wide fluctuations.
Our
common stock is currently quoted for public trading on the OTC QB Marketplace. The trading price of our common stock has been subject
to wide fluctuations. Trading prices of our common stock may fluctuate in response to a number of factors, many of which will be beyond
our control. The stock market has generally experienced extreme price and volume fluctuations that have often been unrelated or disproportionate
to the operating performance of companies with limited business operation. There can be no assurance that trading prices and price earnings
ratios previously experienced by our common stock will be matched or maintained. These broad market and industry factors may adversely
affect the market price of our common stock, regardless of our operating performance. In the past, following periods of volatility in
the market price of a company’s securities, securities class-action litigation has often been instituted. Such litigation, if instituted,
could result in substantial costs for us and a diversion of management’s attention and resources.
Our
common stock is currently quoted only on the OTC QB Marketplace, which may have an unfavorable impact on our stock price and liquidity.
Our
common stock is quoted on the OTC QB Marketplace. The OTC QB Marketplace is a significantly more limited market than the New York Stock
Exchange or the NASDAQ stock market. The quotation of our shares of common stock on the OTC QB Marketplace may result in a less liquid
market available for existing and potential stockholders to trade shares of our common stock, could depress the trading price of our
common stock and could have a long-term adverse impact on our ability to raise capital in the future.
There
can be no assurance that there will be an active market for our shares of common stock either now or in the future. Market liquidity
will depend on the perception of our operating business and any steps that our management might take to bring us to the awareness of
investors. There can be no assurance given that there will be any awareness generated. Consequently, investors may not be able to liquidate
their investment or liquidate at a price that reflects the value of the business. As a result, holders of our securities may not find
purchasers for our securities should they desire to sell them. Consequently, our securities should be purchased only by investors having
no need for liquidity in their investment and who can hold our securities for an indefinite period of time.
Because
our directors and officers currently and for the foreseeable future will continue to control our Company, it is not likely that you will
be able to elect directors or have any say in the policies of our Company.
Our
stockholders are not entitled to cumulative voting rights. Consequently, the election of directors and all other matters requiring stockholder
approval will be decided by majority vote. In addition, our chief executive officer and chief financial officer beneficially owns all
of our preferred stock, which entitles him, as a holder of Series A preferred stock, to vote on all matters submitted or required to
be submitted to a vote of the stockholders, except election and removal of directors, and each share entitles him to five hundred votes
per share of Series A preferred stock, and as a holder of Series B preferred stock, to voting power equivalent of the number of votes
equal to the total number of shares of common stock outstanding as of the record date for the determination of stockholders entitled
to vote at each meeting of our stockholders and entitled to vote on all matters submitted or required to be submitted to a vote of our
stockholders. Due to such a disproportionate voting power, new investors will not be able to affect a change in our business or management,
and therefore, stockholders would have limited recourse as a result of decisions made by management.
Moreover,
this preferred stock ownership may discourage a potential acquirer from making a tender offer or otherwise attempting to obtain control
of us, which in turn could reduce our stock price or prevent our stockholders from realizing a premium over our stock price.
Future
sales and issuances of our common stock or rights to purchase common stock could result in additional dilution of the percentage ownership
of our stockholders and could cause our stock price to decline.
We
are authorized to issue up to 1,000,000,000 shares of our common stock, $0.001 par value per share. We have the right to raise additional
capital or incur borrowings from third parties to finance our business. The board of directors has the authority, without the consent
of any of the shareholders, to cause us to issue more shares of our common stock and/or securities convertible into our common stock.
We will likely issue additional shares of our common stock and/or such securities in the future and such future sales and issuances of
our common stock or rights to purchase our common stock could result in substantial dilution to our existing stockholders. We may sell
common stock, convertible securities and other equity securities in one or more transactions at prices and in a manner as we may determine
from time to time. If we sell any such securities in subsequent transactions, our stockholders may be materially diluted. New investors
in such subsequent transactions could gain rights, preferences and privileges senior to those of holders of our common stock.
In
the future, we may issue additional preferred stock without the approval of our stockholders, which could make it more difficult for
a third party to acquire us and could depress our stock price.
We
are authorized to issue up to 1,500,005 shares of our preferred stock, par value $0.01 per share, having such rights, preferences and
privileges as are determined by our board of directors in their discretion. We have the right to raise additional capital or incur borrowings
from third parties to finance our business. The board of directors has the authority, without the consent of any of the stockholders,
to cause us to issue more shares of our preferred stock. Our board of directors may issue, and has in the past issued, without a vote
of our stockholders, one or more series of our preferred stock with such rights and preferences as it determines. This could permit our
board of directors to issue preferred stock to investors who support us and our management and permit our management to retain control
of our business. Additionally, issuance of preferred stock could block an acquisition which could result in both a drop in our stock
price and a decline in interest of our common stock.
Since
we intend to retain any earnings for development of our business for the foreseeable future, you will likely not receive any dividends
for the foreseeable future, and capital appreciation, if any, will be the source of gain for our stockholders.
We
have never declared or paid any cash dividends or distributions on our capital stock. We currently intend to retain our future earnings
to support operations and to finance expansion and therefore we do not anticipate paying any cash dividends on our common stock in the
foreseeable future. As a result, capital appreciation, if any, of our common stock will be the sole source of gain for our stockholders
for the foreseeable future.
Our
ability to use our net operating loss carryforwards and certain other tax attributes may be limited.
Section
382 (“Section 382”) of the Internal Revenue Code of 1986, as amended (the “Code”), contains rules that limit
the ability of a company that undergoes an ownership change to utilize its net operating losses (“NOLs”) and tax credits
existing as of the date of such ownership change. Under the rules, such an ownership change is generally any change in ownership of more
than 50% of a company’s stock within a rolling three-year period. The rules generally operate by focusing on changes in ownership
among stockholders considered by the rules as owning, directly or indirectly, 5% or more of the stock of a company and any change in
ownership arising from new issuances of stock by the company. As a result of this Section 382 limitation, any ownership changes as defined
by Section 382 may limit the amount of NOL carryforwards that could be utilized annually to offset future taxable income.
As
a smaller reporting company, we are subject to scaled disclosure requirements that may make it more challenging for investors to analyze
our results of operations and financial prospects.
As
a “smaller reporting company,” we (i) are able to provide simplified executive compensation disclosures in our filings, (ii)
are exempt from the provisions of Section 404(b) of the Sarbanes-Oxley Act requiring that independent registered public accounting firms
provide an attestation report on the effectiveness of internal control over financial reporting and (iii) have certain other decreased
disclosure obligations in our filings with the SEC, including being required to provide only two years of audited financial statements
in annual reports. Consequently, it may be more challenging for investors to analyze our results of operations and financial prospects.
We
will remain a smaller reporting company until the beginning of a fiscal year in which we had a public float of $250 million held by non-affiliates
as of the last business day of the second quarter of the prior fiscal year, assuming our common stock is registered under Section 12
of the Exchange Act on the applicable evaluation date. Even if we remain a smaller reporting company, if our public float exceeds $250
million and our annual revenues are greater than $100 million, we will become subject to the provisions of Section 404(b) of the Sarbanes-Oxley
Act.
RISKS
RELATED TO THE OFFERING
Our
existing stockholders may experience significant dilution from the sale of our common stock pursuant to the Purchase Agreement.
The
sale of our common stock to Dutchess in accordance with the Purchase Agreement may have a dilutive impact on our shareholders. As a result,
the market price of our common stock could decline. In addition, the lower our stock price is at the time we exercise our put options,
the more shares of our common stock we will have to issue to Dutchess in order to exercise a put under the Purchase Agreement. If our
stock price decreases, then our existing shareholders would experience greater dilution for any given dollar amount raised through the
offering.
The
perceived risk of dilution may cause our stockholders to sell their shares, which may cause a decline in the price of our common stock.
Moreover, the perceived risk of dilution and the resulting downward pressure on our stock price could encourage investors to engage in
short sales of our common stock. By increasing the number of shares offered for sale, material amounts of short selling could further
contribute to progressive price declines in our common stock.
The
issuance of shares pursuant to the Purchase Agreement may have significant a significant dilutive effect.
Depending
on the number of shares we issue pursuant to the Purchase Agreement, it could have a significant dilutive effect upon our existing shareholders.
Although the number of shares that we may issue pursuant to the Purchase Agreement will vary based on our stock price (the higher our
stock price, the less shares we have to issue), there may be a potential dilutive effect to our shareholders, based on different potential
future stock prices, if the full amount of the Purchase Agreement is realized. Dilution is based upon common stock put to Dutchess and
the stock price discounted to 92% purchase price of the lowest closing price during the pricing period.
Dutchess
will pay less than the then prevailing market price of our common stock which could cause the price of our common stock to declines.
Our
common stock to be issued under the Dutchess Purchase Agreement will be purchased at an eight percent (8%) discount, or ninety two percent
(92%) of the lowest closing price for the Company’s common stock during the five (5) consecutive trading days immediately following
the Clearing Date (as defined in the Purchase Agreement).
Dutchess
has a financial incentive to sell our shares immediately upon receiving them to realize the profit between the discounted price and the
market price. If Dutchess sells our shares, the price of our common stock may decrease. If our stock price decreases, Dutchess may have
further incentive to sell such shares. Accordingly, the discounted sales price in the Purchase Agreement may cause the price of our common
stock to decline.
We
may not have access to the full amount under the Purchase Agreement.
The
lowest closing price of the Company’s common stock during the five (5) consecutive trading day period immediately preceding the
filing of this Registration Statement was approximately $0.0198. At that price we would be able to sell shares to Dutchess under
the Purchase Agreement at the discounted price of $0.0182. At that discounted price, the 40,000,000 shares of Common Stock
to be issued in connection with the Purchase Agreement would only represent approximately $728,640, which is below $5,000,000
(the full amount of the Purchase Agreement).
***
The
risks above do not necessarily comprise of all those associated with an investment in our Company. This Registration Statement contains
forward looking statements that involve unknown risks, uncertainties and other factors that may cause our actual results, financial condition,
performance or achievements to be materially different from any future results, performance or achievements expressed or implied by such
forward looking statements. Factors that might cause such a difference include, but are not limited to, those set out above.
USE
OF PROCEEDS
We
will not receive any proceeds from the sale of the shares of our Common Stock by Dutchess (the Selling Security Holder identified in
this prospectus). However, we will receive proceeds from our initial sale of shares to Dutchess, pursuant to the Purchase Agreement.
The proceeds from the initial sale of shares will be used for the purpose of working capital or for other purposes that the Board of
Directors, in good faith deem to be in the best interest of the Company.
DILUTION
The
sale of our common stock to Dutchess in accordance with the Purchase Agreement will have a dilutive impact on our stockholders.
As
a result, our net loss per share could increase in future periods and the market price of our common stock could decline. In addition,
the lower our stock price is at the time we exercise our put option, the more shares of our common stock we will have to issue to Dutchess
in order to drawdown pursuant to the investment agreement. If our stock price decreases during the pricing period, then our existing
stockholders would experience greater dilution.
DETERMINATION
OF OFFERING PRICE
We
have not set an offering price for the shares registered hereunder, as the only shares being registered are those sold pursuant to the
Purchase Agreement. Dutchess may sell all or a portion of the shares being offered pursuant to this prospectus at fixed prices and prevailing
market prices at the time of sale, at varying prices or at negotiated prices.
SELLING
SECURITY HOLDER
The
selling stockholder may offer and sell, from time to time, any or all of shares of our common stock to be sold to Dutchess under the
investment agreement dated November 30, 2021.
The
following table sets forth certain information regarding the beneficial ownership of shares of common stock by the selling stockholder
as of February 2, 2022 and the number of shares of our common stock being offered pursuant to this prospectus. We believe that
the selling stockholder has sole voting and investment powers over its shares.
Because
the selling stockholder may offer and sell all or only some portion of the 40,000,000 shares of our common stock being offered pursuant
to this prospectus, the numbers in the table below representing the amount and percentage of these shares of our common stock that will
be held by the selling stockholder upon termination of the offering are only estimates based on the assumption that the selling stockholder
will sell all of its shares of our common stock being offered in the offering. being offered pursuant to this prospectus upon the occurrence
of any event that makes any statement in this. The selling stockholder has not had any position or office, or other material relationship
with us or any of our affiliates over the past three years. To our knowledge, the selling stockholder is not a broker-dealer or an affiliate
of a broker-dealer. We may require the selling stockholder to suspend the sales of the shares of our common stock.
| Name of Selling Stockholder | |
Shares Owned by
the Selling Stockholder
before the
Offering (1) | | |
Total Shares Offered in the
Offering | | |
Number of Shares to Be Owned by Selling Stockholder After the
Offering and Percent of Total Issued
and Outstanding Shares (1) | |
| | |
| | |
| | |
# of
Shares (3) | | |
% of
Class (2),(3) | |
| Dutchess Capital Growth Fund LP LLC (4) | |
| 1,000,000 | (5) | |
| 40,000,000 | | |
| -0- | | |
| 0 | |
| (1) |
Beneficial
ownership is determined in accordance with Securities and Exchange Commission rules and generally includes voting or investment power
with respect to shares of common stock. Shares of common stock subject to options and warrants currently exercisable, or exercisable
within 60 days, are counted as outstanding for computing the percentage of the person holding such options or warrants but are not
counted as outstanding for computing the percentage of any other person. |
| |
|
| (2) |
We
have assumed that the selling stockholder will sell all of the shares being offered in this offering. |
| |
|
| (3) |
Based
on 61,417,527 shares of our common stock issued and outstanding as of February 2, 2022. Shares of our common stock
being offered pursuant to this prospectus by a selling stockholder are counted as outstanding for computing the percentage of the
selling stockholder. |
| |
|
| (4) |
Michael
Novielli has the voting and dispositive power over the shares owned by Dutchess Capital Growth Fund LP. |
| |
|
| (5) |
As
of February 2, 2022, Dutchess held 1,000,0000 shares of our common stock issued
as a commitment fee pursuant to the terms of the Purchase Agreement. |
PLAN
OF DISTRIBUTION
The
Selling Security Holder and its pledgees, assignees and successors-in-interest may, from time to time, sell any or all of their securities
covered hereby on the principal Trading Market or any other stock exchange, market or trading facility on which the securities are traded
or in private transactions. These sales may be at fixed or negotiated prices. The Selling Security Holder may use any one or more of
the following methods when selling securities:
| |
● |
ordinary
brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
| |
|
|
| |
● |
block
trades in which the broker-dealer will attempt to sell the securities as agent but may position and resell a portion of the block
as principal to facilitate the transaction; |
| |
|
|
| |
● |
purchases
by a broker-dealer as principal and resale by the broker-dealer for its account; |
| |
|
|
| |
● |
an
exchange distribution in accordance with the rules of the applicable exchange; |
| |
|
|
| |
● |
privately
negotiated transactions; |
| |
|
|
| |
● |
settlement
of short sales; |
| |
|
|
| |
● |
in
transactions through broker-dealers that agree with the Selling Security Holder to sell a specified number of such securities at
a stipulated price per security; |
| |
|
|
| |
● |
through
the writing or settlement of options or other hedging transactions, whether through an options exchange or otherwise; |
| |
|
|
| |
● |
a
combination of any such methods of sale; or |
| |
|
|
| |
● |
any
other method permitted pursuant to applicable law. |
The
Selling Security Holder may also sell securities under Rule 144 or any other exemption from registration under the Securities Act of
1933, as amended (the “Securities Act”), if available, rather than under this prospectus.
Broker-dealers
engaged by the Selling Security Holder may arrange for other brokers-dealers to participate in sales. Broker-dealers may receive commissions
or discounts from the Selling Security Holder (or, if any broker-dealer acts as agent for the purchaser of securities, from the purchaser)
in amounts to be negotiated, but, except as set forth in a supplement to this Prospectus, in the case of an agency transaction not in
excess of a customary brokerage commission in compliance with FINRA Rule 2440; and in the case of a principal transaction a markup or
markdown in compliance with FINRA IM-2440.
Dutchess
is an underwriter within the meaning of the Securities Act and any broker-dealers or agents that are involved in selling the shares may
be deemed to be “underwriters” within the meaning of the Securities Act in connection with such sales. In such event, any
commissions received by such broker-dealers or agents and any profit on the resale of the shares purchased by them may be deemed to be
underwriting commissions or discounts under the Securities Act. Dutchess has informed us that it does not have any written or oral agreement
or understanding, directly or indirectly, with any person to distribute the common stock of our company. Pursuant to a requirement by
FINRA, the maximum commission or discount to be received by any FINRA member or independent broker-dealer may not be greater than 8%
of the gross proceeds received by us for the sale of any securities being registered pursuant to Rule 415 promulgated under the Securities
Act.
Discounts,
concessions, commissions and similar selling expenses, if any, attributable to the sale of shares will be borne by the Selling Security
Holder. The Selling Security Holder may agree to indemnify any agent, dealer, or broker-dealer that participates in transactions involving
sales of the shares if liabilities are imposed on that person under the Securities Act.
We
are required to pay certain fees and expenses incurred by us incident to the registration of the shares covered by this prospectus. We
have agreed to indemnify the Selling Security Holder against certain losses, claims, damages and liabilities, including liabilities under
the Securities Act. We will not receive any proceeds from the resale of any of the shares of our common stock by the Selling Security
Holder. We may, however, receive proceeds from the sale of our common stock under the Purchase Agreement with Dutchess. Neither the Purchase
Agreement with Dutchess nor any rights of the parties under the Purchase Agreement with Dutchess may be assigned or delegated to any
other person.
We
have entered into an agreement with Dutchess to keep this prospectus effective until Dutchess has sold all of the common shares purchased
by it under the Purchase Agreement and has no right to acquire any additional shares of common stock under the Purchase Agreement.
The
resale shares will be sold only through registered or licensed brokers or dealers if required under applicable state securities laws.
In addition, in certain states, the resale shares may not be sold unless they have been registered or qualified for sale in the applicable
state or an exemption from the registration or qualification requirement is available and is complied with.
Under
applicable rules and regulations under the Securities Exchange Act of 1934, any person engaged in the distribution of the resale shares
may not simultaneously engage in market making activities with respect to the common stock for the applicable restricted period, as defined
in Regulation M, prior to the commencement of the distribution. In addition, the Selling Security Holder will be subject to applicable
provisions of the Securities Exchange Act of 1934 and the rules and regulations thereunder, including Regulation M, which may limit the
timing of purchases and sales of shares of the common stock by the Selling Security Holder or any other person. We will make copies of
this prospectus available to the Selling Security Holder.
DESCRIPTION
OF CAPITAL STOCK
Authorized
Capital Stock
Our
authorized capital stock consists of 1,000,000,000 shares of common stock, $0.001 par value per share, and 1,500,005 shares of preferred
stock, $0.01 par value per share. As of February 2, 2022, there were (i) 61,417,527 shares of our common stock issued
and outstanding and (ii) 500,001 shares of our preferred stock issued and outstanding, consisting of 500,000 preferred shares designated
as our Series A Preferred Stock and one preferred share designated as our Series B Preferred Stock.
Common
Stock
Voting;
Holders of our common stock are entitled to one vote for each share held of record on all matters submitted to a vote of the stockholders,
including the election of directors, and do not have cumulative voting rights. Accordingly, the holders of a majority of the shares of
our common stock entitled to vote in any election of directors can elect all of the directors standing for election.
Dividend;
Subject to preferences that may be applicable to any then outstanding preferred stock, the holders of common stock are entitled to
receive dividends, if any, as may be declared from time to time by our board of directors out of legally available funds.
Liquidation;
In the event of our liquidation, dissolution or winding up, holders of our common stock will be entitled to share ratably in the
net assets legally available for distribution to stockholders after the payment of all of our debts and other liabilities, subject to
the satisfaction of any liquidation preference granted to the holders of any outstanding shares of preferred stock.
Rights
and Preferences; Holders of our common stock have no preemptive, conversion or subscription rights, and there are no redemption or
sinking fund provisions applicable to our common stock. The rights, preferences and privileges of the holders of our common stock are
subject to, and may be adversely affected by, the rights of the holders of shares of any series of our preferred stock that we may designate
and issue in the future.
Fully
Paid and Nonassessable; All of our outstanding shares of common stock are, and the shares of common stock to be issued in this offering
will be, fully paid and nonassessable.
Preferred
Stock
Our
board of directors has the authority, without further action by the stockholders, to issue up to 1,500,005 shares of preferred stock
in one or more series, to establish from time to time the number of shares to be included in each such series, to fix the rights, preferences
and privileges of the shares of each wholly unissued series and any qualifications, limitations or restrictions thereon and to increase
or decrease the number of shares of any such series, but not below the number of shares of such series then outstanding.
Our
board of directors may authorize the issuance of preferred stock with voting or conversion rights that could adversely affect the voting
power or other rights of the holders of the common stock. The issuance of preferred stock, while providing flexibility in connection
with possible acquisitions and other corporate purposes, could, among other things, have the effect of delaying, deferring or preventing
a change in our control that may otherwise benefit holders of our common stock and may adversely affect the market price of the common
stock and the voting and other rights of the holders of common stock.
As
of February 2, 2022, 500,001 shares of preferred stock were issued and outstanding, consisting of: (i) 500,000 preferred
shares designated as Series A Preferred Stock (the “Series A Preferred Stock”) pursuant to our Certificate of Designation
filed with the Secretary of State of the State of Delaware on December 9, 2014; and (ii) one preferred share designated as Series B Preferred
Stock (the “Series B Preferred Stock”) pursuant to our Certificate of Designation filed with the Secretary of State of the
State of Delaware on June 16, 2015. Up to five shares have been designated as Series B Preferred Stock.
Mr.
Nathanielsz, our Chief Executive Officer, Chairman, Secretary, Treasurer and a director, beneficially owns all of the shares of Series
A Preferred Stock via North Horizon Pty Ltd., which entitles him, as a holder of Series A Preferred Stock, to vote on all matters submitted
or required to be submitted to a vote of our stockholders, except election and removal of directors, and each share of Series A Preferred
Stock entitles him to 0.004 votes per each outstanding share of Series A Preferred Stock. North Horizon Pty Ltd. is a Nathanielsz Family
Trust. Mr. Nathanielsz has voting and investment power over these shares.
Mr.
Nathanielsz directly beneficially owns such one share of Series B Preferred Stock. Each holder of outstanding shares of Series B Preferred
Stock is entitled to voting power equivalent to the number of votes equal to the total number of shares of common stock outstanding as
of the record date for the determination of stockholders entitled to vote at each meeting of stockholders of our Company and entitled
to vote on all matters submitted or required to be submitted to a vote of the stockholders of the Company.
All
of our issued and outstanding shares of preferred stock have no rights to dividends, profit sharing or liquidation preferences.
Authorized
and Unissued Capital Stock
Delaware
law does not require stockholder approval for any issuance of authorized shares. These additional shares may be used for a variety of
corporate purposes, including future public offerings, to raise additional capital or to facilitate acquisitions.
One
of the effects of the existence of unissued and unreserved common stock or preferred stock may be to enable our board of directors to
issue shares to persons friendly to current management, which issuance could render more difficult or discourage an attempt to obtain
control of our company by means of a merger, tender offer, proxy contest or otherwise, and thereby protect the continuity of our management
and possibly deprive the stockholders of opportunities to sell their shares at prices higher than prevailing market prices.
Warrants
The
following table summarizes warrant activity for the years ended June 30, 2021 and 2020:
| | |
Number of Warrants | | |
Weighted Average
Price Per Share | |
| Outstanding at June 30, 2019 | |
| - | | |
$ | - | |
| Issued | |
| 151,170 | | |
| 150.00 | |
| Exercised | |
| - | | |
| - | |
| Forfeited | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Outstanding at June 30, 2020 | |
| 151,170 | | |
$ | 150.00 | |
| Issued | |
| - | | |
| - | |
| Exercised | |
| (29,841 | ) | |
| 26.15 | |
| Forfeited | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Outstanding at June 30, 2021 | |
| 121,329 | | |
$ | 179.63 | |
| | |
| | | |
| | |
| Exercisable at June 30, 2021 | |
| 76,955 | | |
$ | 283.21 | |
| Outstanding and Exercisable: | |
| | | |
| | |
| | |
| | | |
| | |
| Weighted average remaining contractual term | |
| 1.77 | | |
| | |
Outstanding
Warrants
As
of September 30, 2021, there were 111,932 warrants outstanding of which 76,933 warrants were exercisable with expiration dates commencing
September 2022 and continuing through August 2024, with a weighted average exercise price per share of $191.32.
Options
A
summary of the Company’s option activity during the years ended June 30, 2021 and 2020 is presented below:
| | |
Number of
Options | | |
Weighted
Average
Price Per Share | |
| Outstanding at June 30, 2019 | |
| 60 | | |
$ | 76,370 | |
| Issued | |
| - | | |
| - | |
| Exercised | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Outstanding at June 30, 2020 | |
| 60 | | |
$ | 76,370 | |
| Issued | |
| - | | |
| - | |
| Exercised | |
| - | | |
| - | |
| Expired | |
| (1 | ) | |
| 3,750,000 | |
| Outstanding at June 30, 2021 | |
| 59 | | |
$ | 13,730 | |
| | |
| | | |
| | |
| Exercisable at June 30, 2021 | |
| 40 | | |
$ | 18,193 | |
| Outstanding and Exercisable: | |
| | | |
| | |
| | |
| | | |
| | |
| Weighted average remaining contractual term | |
| 7.86 | | |
| | |
| Weighted average fair value of options granted during the period | |
$ | - | | |
| | |
| Aggregate intrinsic value | |
$ | - | | |
| | |
On
the Effective Date, the Company’s board of directors approved and adopted the Company’s 2019 Equity Incentive Plan (the “2019
Plan”), which reserves a total of 234 shares of the Company’s common stock for issuance under the 2019 Plan. Incentive awards
authorized under the 2019 Plan include, but are not limited to, incentive stock options, non-qualified stock options, restricted stock
awards and restricted stock units.
During
the year ended June 30, 2021 and 2020, the Company recognized stock-based compensation of $82,872 and $82,873 related to vested stock
options. There was $72,514 of unvested stock options expense as of June 30, 2021 that will be recognized over a remaining vesting period
of 0.87 year.
No
stock options were granted during the years ended June 30, 2021 and 2020. No stock options were granted during the three months ended
September 30, 2021.
Restricted
Stock Units
Pursuant
to employment agreements dated in May 2019, the Company granted an aggregate of 78 and 39 restricted stock unit to the Company’s
Chief Executive Officer and Chief Scientific Officer, respectively. The total 117 restricted stock units are subject to vesting terms
as defined in the employment agreements. There are 59 unvested restricted stock units which are subject to various performance conditions
which have not yet been met and such restricted stock units have not yet vested as of June 30, 2021 and 2020. Each vested restricted
stock unit shall be settled by delivery to the holder thereof of one share of our common stock and/or the cash fair market value of such
share of common stock on the date of settlement.
Delaware
Anti-Takeover Statute
We
are subject to the provisions of Section 203 of the General Corporation Law of the State of Delaware regulating corporate takeovers.
In general, Section 203 prohibits a publicly held Delaware corporation from engaging, under certain circumstances, in a business combination
with an interested stockholder for a period of three years following the date the person became an interested stockholder unless:
| |
● |
prior
to the date of the transaction, our board of directors approved either the business combination or the transaction which resulted
in the stockholder becoming an interested stockholder; |
| |
|
|
| |
● |
upon
completion of the transaction that resulted in the stockholder becoming an interested stockholder, the interested stockholder owned
at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, excluding for purposes of
determining the voting stock outstanding, but not the outstanding voting stock owned by the interested stockholder, (1) shares owned
by persons who are directors and also officers and (2) shares owned by employee stock plans in which employee participants do not
have the right to determine confidentially whether shares held subject to the plan will be tendered in a tender or exchange offer;
or |
| |
|
|
| |
● |
at
or subsequent to the date of the transaction, the business combination is approved by our board of directors and authorized at an
annual or special meeting of stockholders, and not by written consent, by the affirmative vote of at least 66 2/3% of the outstanding
voting stock which is not owned by the interested stockholder. |
Generally,
a business combination includes a merger, asset or stock sale, or other transaction resulting in a financial benefit to the interested
stockholder. An interested stockholder is a person who, together with affiliates and associates, owns or, within three years prior to
the determination of interested stockholder status, did own 15% or more of a corporation’s outstanding voting stock. We expect
the existence of this provision to have an anti-takeover effect with respect to transactions our board of directors does not approve
in advance. We also anticipate that Section 203 may discourage attempts that might result in a premium over the market price for the
shares of common stock held by stockholders.
The
provisions of Delaware law and the provisions of our Certificate of Incorporation and Bylaws could have the effect of discouraging others
from attempting hostile takeovers and, as a consequence, they might also inhibit temporary fluctuations in the market price of our common
stock that often result from actual or rumored hostile takeover attempts. These provisions might also have the effect of preventing changes
in our management. It is also possible that these provisions could make it more difficult to accomplish transactions that stockholders
might otherwise deem to be in their best interests.
Bylaws
Provisions
of our Bylaws may delay or discourage transactions involving an actual or potential change in our control or change in our management,
including transactions in which stockholders might otherwise receive a premium for their shares or transactions that our stockholders
might otherwise deem to be in their best interests. Therefore, these provisions could adversely affect the price of our common stock.
Among other things, our Bylaws:
| |
● |
permit
our board of directors to issue up to 1,500,005 shares of our preferred stock, with any rights, preferences and privileges as they
may designate (including the right to approve an acquisition or other change in our control); |
| |
|
|
| |
● |
provide
that the authorized number of directors may be changed only by resolution of the board of directors; |
| |
|
|
| |
● |
provide
that all vacancies, including newly created directorships, may, except as otherwise required by law, be filled by the affirmative
vote of a majority of directors then in office, even if less than a quorum; and |
| |
|
|
| |
● |
do
not provide for cumulative voting rights (therefore allowing the holders of a majority of the shares of common stock entitled to
vote in any election of directors to elect all of the directors standing for election, if they should so choose). |
The
amendment of any of these provisions, with the exception of the ability of our board of directors to issue shares of preferred stock
and designate any rights, preferences and privileges thereto, would require approval by the holders of a majority of our then outstanding
common stock.
EXPERTS
AND COUNSEL
Our
consolidated financial statements as of and for the years ended June 30, 2021 and 2020, appearing in this prospectus and the registration
statement of which it is a part, have been audited by Salberg & Company, P.A., an independent registered public accounting firm,
as set forth in their report dated September 28, 2021(which contains an explanatory paragraph regarding our ability to continue as a
going concern) appearing elsewhere herein, and are included in reliance upon such report given on the authority of such firm as experts
in accounting and auditing.
Patrizio
& O’Leary LLP will render a legal opinion as to the validity of the shares of the common
stock to be registered hereby.
No
expert named in the registration statement of which this prospectus forms a part as having prepared or certified any part thereof (or
is named as having prepared or certified a report or valuation for use in connection with such registration statement) or counsel named
in this prospectus as having given an opinion upon the validity of the securities being offered pursuant to this prospectus, or upon
other legal matters in connection with the registration or offering such securities was employed for such purpose on a contingency basis.
Also at the time of such preparation, certification or opinion or at any time thereafter, through the date of effectiveness of such registration
statement or that part of such registration statement to which such preparation, certification or opinion relates, no such person had,
or is to receive, in connection with the offering, a substantial interest, as defined in Item 509 of Regulation SK, in our company or
any of its parents or subsidiaries. Nor was any such person connected with our company or any of its parents or subsidiaries as a promoter,
managing or principal underwriter or voting trustee.
DESCRIPTION
OF PROPERTY
Our
Offices
Our
principal executive office is located at 302, 6 Butler Street, Camberwell, VIC, 3124 Australia, which we lease from Horizon Pty Ltd.,
a related party, of which Mr. Nathanielsz, our Chief Executive Officer, Chief Financial Officer and a director, and his wife are owners
and directors. The lease has a one-year term commencing May 6, 2021, and we are currently obligated to pay $3,606 AUD or $2,431 USD (depending
on exchange rate), inclusive of tax, in rent per month.
LEGAL
PROCEEDINGS
We
are not currently involved in any litigation that we believe could have a material adverse effect on our financial condition or results
of operations. There is no action, suit, or proceeding by any court, public board, government agency, self-regulatory organization or
body pending or, to the knowledge of the executive officers of our Company or our subsidiary, threatened against or affecting our Company,
our common stock, our subsidiary or of our companies or our subsidiary’s officers or directors in their capacities as such, in
which an adverse decision could have a material adverse effect.
IRS
Liability
As
part of its requirement for having a foreign operating subsidiary, the Company’s parent U.S. entity is required to file an informational
Form 5471 to the Internal Revenue Service (the “IRS”), which is a form that explains the nature of the relationship between
the foreign subsidiary and the parent company. From 2012 through the 2014, the Company did not file this form in a timely manner. As
a result of the non-timely filings, the Company incurred a penalty from the IRS in the amount of $10,000 per year, or $30,000 in total,
plus accrued interest, such penalty and interest having been accrued and is included in the accrued expenses and other payable figure
in the balance sheet. The Company recorded the penalties for all three years during the year ended June 30, 2018 and is negotiating a
payment plan. The Company is current on all subsequent filings.
MARKET
PRICE OF AND DIVIDENDS ON OUR COMMON EQUITY AND RELATED STOCKHOLDER MATTERS
Market
Information
Our
common stock is quoted under the trading symbol “PPCB” on the OTC QB market place. Only a limited market exists for our common
stock. There is no assurance that a regular trading market will develop, or if developed, that it will be sustained. Therefore, a stockholder
may be unable to resell his securities in our Company.
The
following table sets forth the quarterly high and low sale prices for our common shares for the last two completed fiscal years and the
subsequent interim periods. The prices set forth below represent interdealer quotations, without retail markup, markdown or commission
and may not be reflective of actual transactions. The following table sets forth, for the periods indicated, the high and low closing
sales prices of our common stock, as adjusted for stock splits:
| | |
| | |
| | |
For the Years Ended June 30, | |
| | |
2022 | | |
2021 | | |
2020 | |
| | |
High | | |
Low | | |
High | | |
Low | | |
High | | |
Low | |
| | |
| | |
| | |
| | |
| | |
| | |
| |
| First Quarter | |
$ | 0.0580 | | |
$ | 0.0250 | | |
$ | 3.70 | | |
$ | 1.40 | | |
$ | 3,600.00 | | |
$ | 762.50 | |
| Second Quarter | |
$ | 0.0673 | | |
$ | 0.0240 | | |
$ | 2.50 | | |
$ | 0.26 | | |
$ | 980.00 | | |
$ | 112.40 | |
| Third Quarter | |
$ | 0.0429 | | |
$ | 0.0189 | | |
$ | 0.51 | | |
$ | 0.23 | | |
$ | 750.00 | | |
$ | 25.00 | |
| Fourth Quarter (a) | |
$ | - | | |
$ | - | | |
$ | 0.56 | | |
$ | 0.0595 | | |
$ | 27.50 | | |
$ | 3.90 | |
| |
(a) |
For
Q3 2022, the pricing information is through February 2, 2022. |
Transfer
Agent
Action
Stock Transfer, located at 2469 E. Fort Union Blvd, Suite 214, Salt Lake City, Utah 84121 and telephone number of (801) 274-1088 is the
registrar and transfer agent for our common stock.
Number
of Holders
As
of February 2, 2022, there were approximately 77 stockholders of record holding 61,417,527 shares of our common stock.
This number does not include an indeterminate number of stockholders whose shares are held by brokers in street name. The holders of
our common stock are entitled to one vote for each share held of record on all matters submitted to a vote of stockholders. Holders of
our common stock have no preemptive rights and no right to convert their common stock into any other securities. There are no redemption
or sinking fund provisions applicable to our common stock.
Dividend
Policy
We
have never paid any cash dividends on our common stock and do not anticipate paying any cash dividends on our common stock in the foreseeable
future. We intend to retain future earnings to fund ongoing operations and future capital requirements of our business. Any future determination
to pay cash dividends will be at the discretion of our Board and will be dependent upon our financial condition, results of operations,
capital requirements and such other factors as our Board deems relevant. Our ability to pay cash dividends is subject to limitations
imposed by state law.
Penny
Stock
The
SEC has adopted rules that regulate broker-dealer practices in connection with transactions in penny stocks. Penny stocks are generally
equity securities with a market price of less than $5.00, other than securities registered on certain national securities exchanges or
quoted on the NASDAQ system, provided that current price and volume information with respect to transactions in such securities is provided
by the exchange or system. The penny stock rules require a broker-dealer, prior to a transaction in a penny stock, to deliver a standardized
risk disclosure document prepared by the SEC, that: (a) contains a description of the nature and level of risk in the market for penny
stocks in both public offerings and secondary trading; (b) contains a description of the broker’s or dealer’s duties to the
customer and of the rights and remedies available to the customer with respect to a violation of such duties or other requirements of
the securities laws; (c) contains a brief, clear, narrative description of a dealer market, including bid and ask prices for penny stocks
and the significance of the spread between the bid and ask price; (d) contains a toll-free telephone number for inquiries on disciplinary
actions; (e) defines significant terms in the disclosure document or in the conduct of trading in penny stocks; and (f) contains such
other information and is in such form, including language, type size and format, as the SEC shall require by rule or regulation.
The
broker-dealer also must provide, prior to effecting any transaction in a penny stock, the customer with (a) bid and offer quotations
for the penny stock; (b) the compensation of the broker-dealer and its salesperson in the transaction; (c) the number of shares to which
such bid and ask prices apply, or other comparable information relating to the depth and liquidity of the market for such stock; and
(d) a monthly account statement showing the market value of each penny stock held in the customer’s account.
In
addition, the penny stock rules require that prior to a transaction in a penny stock not otherwise exempt from those rules, the broker-dealer
must make a special written determination that the penny stock is a suitable investment for the purchaser and receive the purchaser’s
written acknowledgment of the receipt of a risk disclosure statement, a written agreement as to transactions involving penny stocks,
and a signed and dated copy of a written suitability statement.
These
disclosure requirements may have the effect of reducing the trading activity for our common stock. Therefore, stockholders may have difficulty
selling our securities.
FINANCIAL STATEMENTS
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
Consolidated Financial Statements
Unaudited
Consolidated Financial Statements

Report
of Independent Registered Public Accounting Firm
To
the Stockholders’ and the Board of Directors of:
Propanc
Biopharma, Inc.
Opinion
on the Financial Statements
We
have audited the accompanying consolidated balance sheets Propanc Biopharma, Inc. and Subsidiary (the “Company”) as of June
30, 2021 and 2020, the related consolidated statements of operations and comprehensive income (loss), changes in stockholders’
deficit, and cash flows, for each of the two years in the period ended June 30, 2021, and the related notes (collectively referred to
as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all
material respects, the consolidated financial position of the Company as of June 30, 2021 and 2020, and the consolidated results of its
operations and its cash flows for each of the two years in the period ended June 30, 2021, in conformity with accounting principles generally
accepted in the United States of America.
Going
Concern
The
accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed
in Note 2 to the consolidated financial statements, the Company has a net loss of $2,025,947 and net cash used in operating activities
of $1,145,264 for the fiscal year ended June 30, 2021. The Company has a working capital deficit, stockholder’s deficit, and accumulated
deficit of $3,074,078, $3,067,573, and $58,199,466 respectively, at June 30, 2021. These matters raise substantial doubt about the Company’s
ability to continue as a going concern. Management’s Plan regarding these matters is also described in Note 2. The consolidated
financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis
for Opinion
These
consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion
on the Company’s consolidated financial statements based on our audits. We are a public accounting firm registered with the Public
Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company
in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission
and the PCAOB.
We
conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain
reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud.
The Company is not required to have, nor were we engaged to perform, an audit of internal control over financial reporting. As part of
our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing
an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our
audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether
due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence
regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles
used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements.
We believe that our audits provide a reasonable basis for our opinion.
2295
NW Corporate Blvd., Suite 240 • Boca Raton, FL 33431-7328
Phone:
(561) 995-8270 • Toll Free: (866) CPA-8500 • Fax: (561) 995-1920
www.salbergco.com
• info@salbergco.com
Member
National Association of Certified Valuation Analysts • Registered with the PCAOB
Member
CPAConnect with Affiliated Offices Worldwide • Member AICPA Center for Audit Quality
Critical
Audit Matters
The
critical audit matters communicated below are matters arising from the current period audit of the consolidated financial statements
that were communicated or required to be communicated to the audit committee and that: (1) relate to accounts or disclosures that are
material to the consolidated financial statements and (2) involved our especially challenging, subjective, or complex judgments. The
communication of critical audit matters does not alter in any way our opinion on the consolidated financial statements, taken as a whole,
and we are not, by communicating the critical audit matters below, providing separate opinions on the critical audit matters or on the
accounts or disclosures to which they relate.
Derivative
Liabilities
As
described in Footnote 12 “Derivative Financial Instruments and Fair Value Measurements” to the consolidated financial statements,
the Company recorded derivative transactions that resulted primarily in a net
derivative expense from change in fair value of conversion option liability of $8,186, and derivative liabilities of $54,220 at June
30, 2021.
We
identified the evaluation of instruments and contracts to determine whether there are derivatives to be recorded, the analysis of the
accounting treatment and presentation for derivative transactions and the valuation of derivatives as critical audit matters. Auditing
management’s analysis of the above critical audit matters was complex and involved a high degree of subjectivity.
The
primary procedures we performed to address these critical audit matters included (a) Reviewed and tested management’s conclusions
as to whether certain instruments or contracts qualified for derivative treatment by comparing management’s analysis and conclusions
to authoritative and interpretive literature, (b) Compared the accounting treatment and presentation to that described by the authoritative
and interpretive literature, (c) Tested management’s process for valuing derivatives by comparing it to generally accepted methodologies
for valuing derivatives, (d) Tested management’s valuation of the derivatives by testing assumptions and data used in the valuation
model including the term, volatility and interest rate, and (e) Recomputed the derivative valuations.
/s/
Salberg & Company, P.A.
SALBERG
& COMPANY, P.A.
We
have served as the Company’s auditor since 2011
Boca
Raton, Florida
September
28, 2021
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
CONSOLIDATED
BALANCE SHEETS
The
accompanying notes are an integral part of these consolidated financial statements.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
CONSOLIDATED
STATEMENTS OF OPERATIONS AND COMPREHENSIVE INCOME (LOSS)
The
accompanying notes are an integral part of these consolidated financial statements.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
CONSOLIDATED
STATEMENTS OF CHANGES IN STOCKHOLDERS’ DEFICIT FOR THE YEARS ENDED JUNE 30, 2021 and 2020
| |
|
No.
of Shares |
|
|
Value |
|
|
No.
of Shares |
|
|
Value |
|
|
No.
of Shares |
|
|
Value |
|
|
No.
of Shares |
|
|
Value |
|
|
Capital |
|
|
Deficit |
|
|
Income |
|
|
Stock |
|
|
Deficit |
|
| |
|
|
|
|
|
|
|
Common |
|
|
|
|
|
|
|
|
Accumulated |
|
|
|
|
|
|
|
| |
|
Preferred
Stock |
|
|
|
|
|
Stock |
|
|
Additional |
|
|
|
|
|
Other |
|
|
|
|
|
Total |
|
| |
|
Series
A |
|
|
Series
B |
|
|
Common
Stock |
|
|
Issuable |
|
|
Paid-in |
|
|
Accumulated |
|
|
Comprehensive |
|
|
Treasury |
|
|
Stockholders’ |
|
| |
|
No.
of Shares |
|
|
Value |
|
|
No.
of Shares |
|
|
Value |
|
|
No.
of Shares |
|
|
Value |
|
|
No.
of Shares |
|
|
Value |
|
|
Capital |
|
|
Deficit |
|
|
Income |
|
|
Stock |
|
|
Deficit |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Balance
at June 30, 2019 |
|
|
500,000 |
|
|
$ |
5,000 |
|
|
|
1 |
|
|
$ |
- |
|
|
|
968 |
|
|
$ |
1 |
|
|
|
- |
|
|
$ |
- |
|
|
$ |
45,714,289 |
|
- |
$ |
(51,041,047 |
) |
|
$ |
1,066,998 |
|
|
$ |
(46,477 |
) |
|
$ |
(4,301,236 |
) |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance
of common stock for cash |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
804 |
|
|
|
1 |
|
|
|
- |
|
|
|
- |
|
|
|
424,989 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
424,990 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance
of common stock for conversion of convertible debt and accrued interest |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
247,619 |
|
|
|
248 |
|
|
|
- |
|
|
|
- |
|
|
|
2,124,926 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
2,125,174 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Reversal of common stock issuable due to cancellation of conversions of convertible debt and accrued interest |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Reversal of common stock issuable due to cancellation of conversions of convertible debt and accrued interest, shares |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance of common stock for exercise of warrants |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance of common stock for exercise of warrants, shares |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance of common stock for cashless exercise of warrants |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance of common stock for cashless exercise of warrants |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Reversal of put premium upon cancellation of conversions of convertible debt |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Vested restricted stock units |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Vested restricted stock units, shares |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Fractional difference due to the reverse stock-split |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Fractional difference due to the reverse stock-split, shares |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Deemed
dividend upon cashless exercise of warrants |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance of common stock for alternate cashless exercise of warrants |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance of common stock for alternate cashless exercise of warrants, shares |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Reclassification
of put premium upon debt conversion |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
874,924 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
874,924 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance
of common stock for services |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
8,729 |
|
|
|
8 |
|
|
|
- |
|
|
|
- |
|
|
|
113,634 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
113,642 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Relative
fair value of warrants issued with convertible debt |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
375,905 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
375,905 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Stock
based compensation in connection with stock option grants and restricted stock unit grants |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
300,416 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
300,416 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Stock
based compensation in connection with fair value of warrants issued for services |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
984,810 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
984,810 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Foreign
currency translation gain |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
200,673 |
|
|
|
|
|
|
|
200,673 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Net
loss for the fiscal year ended June 30, 2020 |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
(4,740,723 |
) |
|
|
|
|
|
|
- |
|
|
|
(4,740,723 |
) |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Balance
at June 30, 2020 |
|
|
500,000 |
|
|
|
5,000 |
|
|
|
1 |
|
|
|
- |
|
|
|
258,120 |
|
|
|
258 |
|
|
|
- |
|
|
|
- |
|
|
|
50,913,893 |
|
- |
|
(55,781,770 |
) |
|
|
1,267,671 |
|
|
|
(46,477 |
) |
|
|
(3,641,425 |
) |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance
of common stock for conversion of convertible debt and accrued interest |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
8,786,113 |
|
|
|
8,787 |
|
|
|
- |
|
|
|
- |
|
|
|
1,230,288 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
1,239,075 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Reversal
of common stock issuable due to cancellation of conversions of convertible debt and accrued interest |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
(24,427 |
) |
|
|
(24 |
) |
|
|
- |
|
|
|
- |
|
|
|
(19,992 |
) |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
(20,016 |
) |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Reversal
of put premium upon cancellation of conversions of convertible debt |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
(11,785 |
) |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
(11,785 |
) |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance
of common stock for services |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
805,646 |
|
|
|
806 |
|
|
|
- |
|
|
|
- |
|
|
|
124,766 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
125,572 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance
of common stock for exercise of warrants |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
29,820 |
|
|
|
29 |
|
|
|
- |
|
|
|
- |
|
|
|
776,015 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
776,044 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance
of common stock for cashless exercise of warrants |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
4,199,979 |
|
|
|
4,200 |
|
|
|
|
|
|
|
|
|
|
|
(4,200 |
) |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Deemed dividend upon alternate cashless exercise of warrants |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
391,749 |
|
|
|
(391,749 |
) |
|
|
- |
|
|
|
- |
|
|
|
- |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Reclassification
of put premium upon debt conversion |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
590,504 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
590,504 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Stock
based compensation in connection with stock option grants |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
82,872 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
82,872 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Vested
restricted stock units |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
59 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Fractional
difference due to the reverse stock-split |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
142 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Foreign
currency translation loss |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
(182,467 |
) |
|
|
- |
|
|
|
(182,467 |
) |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Net
loss for the fiscal year ended June 30, 2021 |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
- |
|
(2,025,947 |
) |
|
|
- |
|
|
|
- |
|
|
|
(2,025,947 |
) |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Balance
at June 30, 2021 |
|
|
500,000 |
|
|
$ |
5,000 |
|
|
|
1 |
|
|
$ |
- |
|
|
|
14,055,393 |
|
|
$ |
14,056 |
|
|
|
59 |
|
|
$ |
- |
|
|
$ |
54,074,110 |
|
- |
$ |
(58,199,466 |
) |
|
$ |
1,085,204 |
|
|
$ |
(46,477 |
) |
|
$ |
(3,067,573 |
) |
The
accompanying notes are an integral part of these consolidated financial statements.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
CONSOLIDATED
STATEMENTS OF CASH FLOWS
The
accompanying notes are an integral part of these consolidated financial statements.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
NOTE
1 – NATURE OF OPERATIONS AND SUMMARY OF SIGNIFICANT ACCOUNTING AND REPORTING POLICIES
Nature
of Operations
Propanc
Biopharma, Inc. (the “Company,” “we,” “us” or “our”) was originally incorporated in Melbourne,
Victoria Australia on October 15, 2007 as Propanc PTY LTD, and continues to be based in Camberwell, Victoria Australia. Since its inception,
substantially all of the operations of the Company have been focused on the development of new cancer treatments targeting high-risk
patients, particularly cancer survivors, who need a follow-up, non-toxic, long-term therapy designed to prevent the cancer from returning
and spreading. The Company anticipates establishing global markets for its technologies. Our lead product candidate, which we refer to
as PRP, is an enhanced pro-enzyme formulation designed to enhance the anti-cancer effects of multiple enzymes acting synergistically.
It is currently in the preclinical phase of development.
On
November 23, 2010, the Company was incorporated in the state of Delaware as Propanc Health Group Corporation. In January 2011, to reorganize
the Company, we acquired all of the outstanding shares of Propanc PTY LTD on a one-for-one basis making it a wholly-owned subsidiary
of the Company.
On
July 22, 2016, the Company formed a wholly owned subsidiary, Propanc (UK) Limited under the laws of England and Wales for the purpose
of submitting an orphan drug application to the European Medicines Agency as a small and medium-sized enterprise. As of June 30, 2021,
there has been no activity within this entity.
Effective
April 20, 2017, the Company changed its name to “Propanc Biopharma, Inc.” to better reflect the Company’s stage of
operations and development.
In
July 2020, a world first patent was granted in Australia for the cancer treatment method patent family. Presently, there are 29 granted
patents and 33 patents under examination in key global jurisdictions relating to the use of proenzymes against solid tumors, covering
the lead product candidate PRP.
The
Company hopes to capture and protect additional patentable subject matter based on the Company’s field of technology relating to
pharmaceutical compositions of proenzymes for treating cancer by filing additional patent applications as it advances its lead product
candidate, PRP, through various stages of development.
On
November 17, 2020, the Company effected a one-for-one thousand (1:1,000) reverse stock split of the Company’s issued and outstanding
shares of common stock (the “Reverse Stock Split”). Proportional adjustments for the Reverse Stock Split were made to the
Company’s outstanding stock options, warrants and equity incentive plans. All share and per-share data and amounts have been retroactively
adjusted as of the earliest period presented in the consolidated financial statements to reflect the Reverse Stock Split.
Principles
of Consolidation
The
consolidated financial statements include the accounts of Propanc Biopharma, Inc., the parent entity, and its wholly-owned subsidiary,
Propanc PTY LTD. All inter-company balances and transactions have been eliminated in consolidation. Propanc (UK) Limited was an inactive
wholly-owned subsidiary through June 30, 2021.
Use
of Estimates
The
preparation of financial statements in conformity with the accounting principles generally accepted in the United States of America (“US
GAAP”) requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure
of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of revenues and expenses
during the reporting period. Actual results could differ from these estimates. Significant estimates in the accompanying consolidated
financial statements include the estimates of useful lives for depreciation, valuation of the operating lease liability and related right-of-use
asset, valuation of derivatives, valuation of beneficial conversion features on convertible debt, allowance for uncollectable receivables,
valuation of equity based instruments issued for other than cash, the valuation allowance on deferred tax assets and foreign currency
translation due to certain average exchange rates applied in lieu of spot rates on transaction dates.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
Foreign
Currency Translation and Other Comprehensive Income (Loss)
The
Company’s wholly owned subsidiary’s functional currency is the Australian dollar (AUD). For financial reporting purposes,
the Australian dollar has been translated into the Company’s reporting currency which is the United States dollar ($) and/or (USD).
Assets and liabilities are translated at the exchange rate in effect at the balance sheet date. Revenues and expenses are translated
at the average rate of exchange prevailing during the reporting period. Equity transactions are translated at each historical transaction
date spot rate. Translation adjustments arising from the use of different exchange rates from period to period are included as a component
of stockholders’ equity (deficit) as “Accumulated other comprehensive income (loss).” Gains and losses resulting from
foreign currency transactions are included in the statements of operations and comprehensive income (loss) as a component of other
comprehensive income (loss). There have been no significant fluctuations in the exchange rate for the conversion of Australian dollars
to USD after the balance sheet date.
Other
Comprehensive Income (Loss) for all periods presented includes only foreign currency translation gains (losses).
Assets
and liabilities denominated in foreign currencies are translated into the functional currency at the exchange rates prevailing at
the consolidated balance sheet date with any transaction gains and losses that arise from exchange rate fluctuations on transactions
denominated in a currency other than the functional currency included in the consolidated results of operations as incurred.
Effective fiscal year 2021, the parent company determined that these intercompany loans will not be repaid in the foreseeable future
and thus, per ASC 830-20-35-3, gains and losses from measuring the intercompany balances are recorded within cumulative translation
adjustment, a component of accumulated other comprehensive income (loss). Prior to July 1, 2020, the Company recorded the foreign
currency transaction gains and losses from measuring the intercompany balances as a component of other income (expenses) titled
foreign currency transaction gain (loss). For the year ended June 30, 2021 and 2020, the Company recognized an exchange gain
(loss) of approximately $1,005,000 and
($133,000),
on intercompany loans made by the parent to the subsidiary which have not been repaid as of June 30, 2021.
As
of June 30, 2021 and 2020, the exchange rates used to translate amounts in Australian dollars into USD for the purposes of preparing
the consolidated financial statements were as follows:
SCHEDULE OF TRANSLATION EXCHANGE RATES
| |
|
June
30, 2021 |
|
|
June
30, 2020 |
|
| Exchange
rate on balance sheet dates |
|
|
|
|
|
|
|
|
| USD
: AUD exchange rate |
|
|
0.7500 |
|
|
|
0.6891 |
|
| |
|
|
|
|
|
|
|
|
| Average
exchange rate for the period |
|
|
|
|
|
|
|
|
| USD
: AUD exchange rate |
|
|
0.7473 |
|
|
|
0.6742 |
|
Change
in Accumulated Other Comprehensive Income (Loss) by component during the years ended June 30, 2021 and 2020 were as follows:
SCHEDULE OF CHANGE IN ACCUMULATED OTHER COMPREHENSIVE INCOME (LOSS)
| |
|
Foreign
Currency Items: |
|
| Beginning
balance, June 30, 2019 |
|
$ |
1,066,998 |
|
| Foreign
currency translation gain |
|
|
200,673 |
|
| Balance,
June 30, 2020 |
|
|
1,267,671 |
|
| Foreign
currency translation gain |
|
|
182,467 |
|
| Ending
balance, June 30, 2021 |
|
$ |
1,085,204 |
|
Fair
Value of Financial Instruments and Fair Value Measurements
The
Company measures its financial assets and liabilities in accordance with US GAAP. For certain financial instruments, including cash and
cash equivalents, accounts receivable, accounts payable and accrued liabilities, the carrying amounts approximate fair value due to their
short maturities. Amounts recorded for notes payable, net of discount, and loans payable also approximate fair value because current
interest rates available for debt with similar terms and maturities are substantially the same.
The
Company follows accounting guidance for financial assets and liabilities. This standard defines fair value, provides guidance for measuring
fair value and requires certain disclosures. This standard does not require any new fair value measurements, but rather applies to all
other accounting pronouncements that require or permit fair value measurements. This guidance does not apply to measurements related
to share-based payments. This guidance discusses valuation techniques, such as the market approach (comparable market prices), the income
approach (present value of future income or cash flow), and the cost approach (cost to replace the service capacity of an asset or replacement
cost).
The
guidance utilizes a fair value hierarchy that prioritizes the inputs to valuation techniques used to measure fair value into three broad
levels. The following is a brief description of those three levels:
Level
1: Observable inputs such as quoted prices (unadjusted) in active markets for identical assets or liabilities.
Level
2: Inputs, other than quoted prices that are observable, either directly or indirectly. These include quoted prices for similar assets
or liabilities in active markets and quoted prices for identical or similar assets or liabilities in markets that are not active.
Level
3: Unobservable inputs in which little or no market data exists, therefore developed using estimates and assumptions developed by us,
which reflect those that a market participant would use.
Also
see Note 12 - Derivative Financial Instruments and Fair Value Measurements.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
Cash
and Cash Equivalents
Cash
and cash equivalents include cash on hand and at banks, short-term deposits with an original maturity of three months or less with financial
institutions, and bank overdrafts. Bank overdrafts, as applicable, are reflected as a current liability on the balance sheets.
There were no cash
equivalents as of June 30, 2021 or June 30, 2020.
Property
and Equipment
Property
and equipment are stated at cost, net of accumulated depreciation. Expenditures for maintenance and repairs are expensed as incurred;
additions, renewals, and betterments are capitalized. When property and equipment are retired or otherwise disposed of, the related cost
and accumulated depreciation are removed from the respective accounts, and any gain or loss is included in operations. Depreciation of
property and equipment is provided using the declining balance method. The depreciable amount is the cost less its residual value.
The
estimated useful lives are as follows:
SCHEDULE OF PROPERTY AND EQUIPMENT ESTIMATED USEFUL LIVES
| Machinery
and equipment |
-
5 years |
| Furniture |
-
7 years |
Patents
Patents
are stated at cost and amortized on a straight-line basis over the estimated future periods if and once the patent has been granted by
a regulatory agency. However, the Company will expense any patent costs as long as we are in the startup stage. Accordingly, as the Company’s
products are not currently approved for market, all patent costs incurred from 2013 through June 30, 2021 were expensed immediately.
This practice of expensing patent costs immediately ends when a product receives market authorization from a government regulatory agency.
Impairment
of Long-Lived Assets
In
accordance with ASC 360-10, “Long-lived assets,” which include property and equipment and intangible assets, are reviewed
for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. Recoverability
of long-lived assets to be held and used is measured by a comparison of the carrying amount of an asset to the estimated undiscounted
future cash flows expected to be generated by the asset. If the carrying amount of an asset exceeds its estimated undiscounted future
cash flows, an impairment charge is recognized by the amount by which the carrying amount of the asset exceeds the fair value of the
assets. Fair value is generally determined using the asset’s expected future discounted cash flows or market value, if readily
determinable.
Employee
Benefit/Liability
Liabilities
arising in respect of wages and salaries, accumulated annual leave, accumulated long service leave and any other employee benefits expected
to be settled within twelve months of the reporting date are measured based on the employee’s remuneration rates applicable at
the reporting date. All other employee benefit liabilities are measured at the present value of the estimated future cash outflow to
be made in respect of services provided by employees up to the reporting date. All employee liabilities are owed within the next twelve
months.
Australian
Goods and Services Tax (“GST”)
Revenues,
expenses and balance sheet items are recognized net of the amount of GST, except payable and receivable balances which are shown inclusive
of GST. The GST incurred is payable on revenues to, and recoverable on purchases from, the Australian Taxation Office.
Cash
flows are presented in the statements of cash flow on a gross basis, except for the GST component of investing and financing activities,
which are disclosed as operating cash flows.
As
of June 30, 2021 and 2020, the Company was owed $4,341 and $2,015, respectively, from the Australian Taxation Office. These amounts were
fully collected subsequent to the balance sheet reporting dates.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
Derivative
Instruments
ASC
Topic 815, Derivatives and Hedging (“ASC Topic 815”), establishes accounting and reporting standards for derivative
instruments and for hedging activities by requiring that all derivatives be recognized in the balance sheet and measured at fair value.
Gains or losses resulting from changes in the fair value of derivatives are recognized in earnings. On the date of conversion or payoff
of debt, the Company records the fair value of the conversion shares, removes the fair value of the related derivative liability, removes
any discounts and records a net gain or loss on debt extinguishment. On July 1, 2019 the Company adopted ASU 2017-11 under which down-round
Features in Financial Instruments will no longer cause derivative treatment. The Company applies the modified prospective method of adoption.
There were no cumulative effects on adoption.
Convertible
Notes With Variable Conversion Options
The
Company has entered into convertible notes, some of which contain variable conversion options, whereby the outstanding principal and
accrued interest may be converted, by the holder, into common shares at a fixed discount to the price of the common stock at or around
the time of conversion. The Company treats these convertible notes as stock settled debt under ASC 480, “Distinguishing Liabilities
from Equity” and measures the fair value of the notes at the time of issuance, which is the result of the share price discount
at the time of conversion and records the put premium as interest expense.
Income
Taxes
The
Company is governed by Australia and United States income tax laws, which are administered by the Australian Taxation Office and the
United States Internal Revenue Service, respectively. The Company follows ASC 740 “Accounting for Income Taxes,” when
accounting for income taxes, which requires an asset and liability approach to financial accounting and reporting for income taxes. Deferred
income tax assets and liabilities are computed annually for temporary differences between the financial statements and tax bases of assets
and liabilities that will result in taxable or deductible amounts in the future based on enacted tax laws and rates applicable to the
periods in which the differences are expected to affect taxable income. Valuation allowances are established when necessary to reduce
deferred tax assets to the amount expected to be realized. Income tax expense is the tax payable or refundable for the period plus or
minus the change during the period in deferred tax assets and liabilities.
The
Company follows ASC 740, Sections 25 through 60, “Accounting for Uncertainty in Income Taxes.” These sections provide
detailed guidance for the financial statement recognition, measurement and disclosure of uncertain tax positions recognized in the financial
statements. Tax positions must meet a “more-likely-than-not” recognition threshold at the effective date to be recognized
upon the adoption of ASC 740 and in subsequent periods.
Research
and Development Costs and Tax Credits
In
accordance with ASC 730-10, “Research and Development-Overall,” research and development costs are expensed when incurred.
Total research and development costs for the fiscal years ended June 30, 2021 and 2020 were $230,956 and $179,987, respectively.
The
Company may apply for research and development tax concessions with the Australian Taxation Office on an annual basis. Although the amount
is possible to estimate at year end, the Australian Taxation Office may reject or materially alter the claim amount. Accordingly, the
Company does not recognize the benefit of the claim amount until cash receipt since collectability is not certain until such time. The
tax concession is a refundable credit. If the Company has net income, then the Company can receive the credit which reduces its income
tax liability. If the Company has net losses, then the Company may still receive a cash payment for the credit, however, the Company’s
net operating loss carryforwards are reduced by the gross equivalent loss that would produce the credit amount when the income tax rate
is applied to that gross amount. The concession is recognized as tax benefit, in operations, upon receipt.
During
each of the fiscal years ended June 30, 2021 and 2020, the Company applied for, and received from the Australian Taxation Office, a research
and development tax credit in the amount of $113,415 and $134,728, respectively, which is reflected as a tax benefit in the accompanying
consolidated statements of operations and comprehensive income (loss).
Stock
Based Compensation
The
Company records stock-based compensation in accordance with ASC 718, “Stock Compensation”. ASC 718 requires the fair
value of all stock-based employee compensation awarded to employees to be recorded as an expense over the shorter of the service period
or the vesting period. The Company values employee and non-employee stock-based compensation at fair value using the Black-Scholes Option
Pricing Model.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
The
Company adopted ASU 2018-07 and accounts for non-employee share-based awards in accordance with the measurement and recognition criteria
of ASC 718 and recognizes the fair value of such awards over the service period. The Company used the modified prospective method of
adoption. There was no cumulative effect of adoption on July 1, 2019.
Revenue
Recognition
The
Company adopted and implemented on July 1, 2018, ASC 606 – Revenue from Contracts with Customers (“ASC 606”). ASC 606
did not have a material impact on the consolidated financial statements.
Upon
implementation of ASC 606, the Company recognizes revenue in accordance with that core principle by applying the following steps:
Step
1: Identify the contract(s) with a customer.
Step
2: Identify the performance obligations in the contract.
Step
3: Determine the transaction price.
Step
4: Allocate the transaction price to the performance obligations in the contract.
Step
5: Recognize revenue when (or as) the entity satisfies a performance obligation.
Subject
to these criteria, the Company intends to recognize revenue relating to royalties on product sales in the period in which the sale occurs
and the royalty term has begun.
Legal
Expenses
All
legal costs for litigation are charged to expense as incurred.
Leases
In
February 2016, the Financial Accounting Standards Board (“FASB”) issued ASU 2016-02, Leases (Topic 842). The updated
guidance requires lessees to recognize lease assets and lease liabilities for most operating leases. In addition, the updated guidance
requires that lessors separate lease and non-lease components in a contract in accordance with the new revenue guidance in ASC 606. This
guidance is effective for interim and annual reporting periods beginning after December 15, 2018. The Company adopted this guidance effective
July 1, 2019.
On
July 1, 2019, the Company adopted ASU No. 2016-02, applying the package of practical expedients to leases that commenced before the effective
date whereby the Company elected to not reassess the following: (i) whether any expired or existing contracts contain leases and; (ii)
initial direct costs for any existing leases. For contracts entered into on or after the effective date, at the inception of a contract
the Company assessed whether the contract is, or contains, a lease. The Company’s assessment is based on: (1) whether the contract
involves the use of a distinct identified asset, (2) whether we obtain the right to substantially all the economic benefit from the use
of the asset throughout the period, and (3) whether it has the right to direct the use of the asset. The Company will allocate the consideration
in the contract to each lease component based on its relative stand-alone price to determine the lease payments.
Operating
lease ROU assets represents the right to use the leased asset for the lease term and operating lease liabilities are recognized based
on the present value of future minimum lease payments over the lease term at commencement date. As most leases do not provide an implicit
rate, the Company use an incremental borrowing rate based on the information available at the adoption date in determining the present
value of future payments. Lease expense for minimum lease payments is amortized on a straight-line basis over the lease term and is included
in general and administrative expenses in the consolidated statements of operations.
Basic
and Diluted Net Loss Per Common Share
Basic
net loss per share is computed by dividing the net loss by the weighted average number of common shares outstanding during the period.
Diluted net loss per common share is computed by dividing the net loss by the weighted average number of common shares outstanding for
the period and, if dilutive, potential common shares outstanding during the period. Potentially dilutive securities consist of the incremental
common shares issuable upon exercise of common stock equivalents such as stock options, warrants and convertible debt instruments. Potentially
dilutive securities are excluded from the computation if their effect is anti-dilutive. As a result, the basic and diluted per share
amounts for all periods presented are identical. Each holder of the notes has agreed to a 4.99% beneficial ownership conversion limitation
(subject to certain noteholders’ ability to increase such limitation to 9.99% upon 60 days’ notice to the Company), and each
note may not be converted during the first six-month period from the date of issuance. Such securities are considered dilutive securities
which were excluded from the computation since the effect is anti-dilutive.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
SCHEDULE OF DILUTIVE SECURITIES EXCLUDED FROM COMPUTATION
| |
|
June
30, 2021 |
|
|
June
30, 2020 |
|
| |
|
|
|
|
|
|
| Stock
Options |
|
|
59 |
|
|
|
60 |
|
| Stock
Warrants |
|
|
121,329 |
|
|
|
151,171 |
|
| Unvested
restricted stock |
|
|
59 |
|
|
|
59 |
|
| Convertible
Debt |
|
|
12,416,972 |
|
|
|
439,113 |
|
| Total |
|
|
12,538,419 |
|
|
|
590,403 |
|
Recent
Accounting Pronouncements
We
have reviewed the FASB issued ASU accounting pronouncements and interpretations thereof that have effectiveness dates during the periods
reported and in future periods. We have carefully considered the new pronouncements that alter previous generally accepted accounting
principles and do not believe that any new or modified principles will have a material impact on the Company’s reported financial
position or operations in the near term with the exception of those disclosed below. The applicability of any standard is subject
to the formal review of the Company’s financial management.
In
August 2020, the FASB issued ASU 2020-06, “Debt – Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives
and Hedging – Contracts in Entity’s Own Equity (Subtopic 815-40)”. This ASU reduces the number of accounting models
for convertible debt instruments and convertible preferred stock. As well as amend the guidance for the derivatives scope exception for
contracts in an entity’s own equity to reduce form-over-substance-based accounting conclusions. In addition, this ASU improves
and amends the related EPS guidance. This standard is effective for us on July 1, 2022, including interim periods within those fiscal
years. Adoption is either a modified retrospective method or a fully retrospective method of transition. The Company is currently assessing
the impact the new guidance will have on our consolidated financial statements.
NOTE
2 – GOING CONCERN
The
accompanying consolidated financial statements have been prepared in conformity with US GAAP, which contemplate continuation of the Company
as a going concern. For the fiscal year ended June 30, 2021, the Company had no
revenues, had a net loss of $2,025,947
and had net cash used in operations
of $1,145,264.
Additionally, as of June 30, 2021, the Company had a working capital deficit, stockholders’ deficit and accumulated deficit of
$3,074,078,
$3,067,573,
and $58,199,466,
respectively. It is management’s opinion that these
conditions raise substantial doubt about the Company’s ability to continue as a going concern for a period of at least twelve months
from the date of this filing.
The
consolidated financial statements do not include any adjustments to reflect the possible future effect on the recoverability and classification
of assets or the amounts and classifications of liabilities that may result from the outcome of this uncertainty.
Successful
completion of the Company’s development program and, ultimately, the attainment of profitable operations are dependent upon future
events, including obtaining adequate financing to fulfill its development activities, acceptance of the Company’s patent applications,
obtaining additional sources of suitable and adequate financing and ultimately achieving a level of sales adequate to support the Company’s
cost structure and business plan. The Company’s ability to continue as a going concern is also dependent on its ability to further
develop and execute on its business plan. However, there can be no assurances that any or all of these endeavors will be successful.
In
March 2020, the outbreak of COVID-19 (coronavirus) caused by a novel strain of the coronavirus was recognized as a pandemic by the World
Health Organization, and the outbreak has become increasingly widespread in the United States, Europe and Australia, including in each
of the areas in which the Company operates. The COVID-19 (coronavirus) outbreak has had a notable impact on general economic conditions,
including but not limited to the temporary closures of many businesses, “shelter in place” and other governmental regulations,
reduced business and consumer spending due to both job losses, reduced investing activity and M&A transactions, among many other
effects attributable to the COVID-19 (coronavirus), and there continue to be many unknowns. While to date the Company has not been required
to stop operating, management is evaluating its use of its office space, virtual meetings and the like. The Company continues to monitor
the impact of the COVID-19 (coronavirus) outbreak closely. The extent to which the COVID-19 (coronavirus) outbreak will impact our operations,
ability to obtain financing or future financial results is uncertain.
NOTE
3 – PROPERTY AND EQUIPMENT
Property
and equipment consist of the following as of June 30,
SCHEDULE OF PROPERTY AND EQUIPMENT
| |
|
2021 |
|
|
2020 |
|
| |
|
|
|
|
|
|
| Office
equipment at cost |
|
$ |
28,623 |
|
|
$ |
26,299 |
|
| Less:
Accumulated depreciation |
|
|
(24,368 |
) |
|
|
(20,552 |
) |
| |
|
|
|
|
|
|
|
|
| Total
property, plant, and equipment |
|
$ |
4,255 |
|
|
$ |
5,747 |
|
Depreciation
expense for the years ended June 30, 2021 and 2020 were $1,993 and $2,473, respectively
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
NOTE
4 – DUE TO FORMER DIRECTOR - RELATED PARTY
Due
to former director - related party represents unsecured advances made primarily by a former director for operating expenses on behalf
of the Company such as intellectual property and formation expenses. The expenses were paid for on behalf of the Company and are due
upon demand. The Company is currently not being charged interest under these advances. The total amount owed the former director at June
30, 2021 and 2020 is $33,347 and $30,639, respectively. The Company plans to repay the advances as its cash resources allow (see Note
10).
NOTE
5 – LOANS AND NOTES PAYABLE
Loan
from Former Director - Related Party
Loan
from the Company’s former director at June 30, 2021 and 2020 were $55,500 and $50,993, respectively. The loan bear no interest
and is payable on demand. The Company did not repay any amount on this loan during the years ended June 30, 2021 and 2020, respectively,
(see Note 10).
NOTE
6 – CONVERTIBLE NOTES
The
Company’s convertible notes outstanding at June 30, 2021 and 2020 were as follows:
SCHEDULE OF CONVERTIBLE NOTES
| |
|
June
30, 2021 |
|
|
June
30, 2020 |
|
| Convertible
notes and debenture |
|
$ |
400,128 |
|
|
$ |
1,029,496 |
|
| Unamortized
discounts |
|
|
(6,139 |
) |
|
|
(126,667 |
) |
| Accrued
interest |
|
|
34,098 |
|
|
|
80,101 |
|
| Premium,
net |
|
|
196,496 |
|
|
|
574,804 |
|
| Convertible
notes, net |
|
$ |
624,583 |
|
|
$ |
1,557,734 |
|
Eagle
Equities Financing Agreements
December
29, 2017 Securities Purchase Agreement
The
Company entered into an executory contract on December 29, 2017, whereby the Company entered into a securities purchase agreement with
Eagle Equities, pursuant to which Eagle Equities purchased a convertible promissory note (the “December 2017 Eagle Note”)
from the Company in the aggregate principal amount of $532,435, with principal and the interest thereon convertible into shares of the
Company’s common stock at the option of Eagle Equities at any time. The December 2017 Eagle Note contained an original issue discount
of $25,354 such that the purchase price was $507,081. The maturity date of the December 2017 Eagle Note was December 29, 2018. The Company
was in discussions with Eagle Equities to extend the maturity date. The December 2017 Eagle Note bore interest at a rate of 8% per annum,
which interest were to be paid by the Company to Eagle Equities in shares of the Company’s common stock upon receipt of a notice
of conversion by the Company from Eagle Equities at any time. The Company recorded $20,065 of accrued interest for the December 2017
Eagle Note and total principal outstanding as of June 30, 2019 under the December 2017 Eagle Note was $171,965 following conversion of
$360,470 of principal and $43,535 of accrued interest during the fiscal year ended June 30, 2019. The Company recorded $0 of accrued
interest for the December 2017 Eagle Note and total principal outstanding as of June 30, 2020 under the December 2017 Eagle Note was
$0 following conversion of the remaining principal $171,965 and $24,751 of accrued interest during the fiscal year to June 30, 2020.
Accordingly, there was no outstanding balance as of June 30, 2020.
July
13, 2018 Securities Purchase Agreement
Effective
July 13, 2018, the Company entered into a securities purchase agreement with Eagle Equities, pursuant to which Eagle Equities purchased
a convertible promissory note (the “July 2018 Note”) from the Company in the aggregate principal amount of $75,000, such
principal and the interest thereon convertible into shares of the Company’s common stock at the option of Eagle Equities any time
after the six month anniversary of the July 2018 Eagle Note. The transaction closed on July 16, 2018 and on July 19, 2018 the Company
received proceeds of $71,250 as $3,750 was paid directly to legal fees. The maturity date of the July 2018 Eagle Note was July 13, 2019.
The July 2018 Eagle Note bears interest at a rate of 8% per annum, which interest shall be paid by the Company to Eagle Equities in shares
of the Company’s common stock upon receipt of a notice of conversion by the Company from Eagle Equities at any time after the six-month
anniversary of the Note. The Company recorded $5,786 of accrued interest and the total principal outstanding under the July 2018 Eagle
Note was $75,000 as of June 30, 2019. The Company recorded $0 of accrued interest and the total principal outstanding under the July
2018 Eagle Note was $0 as of June 30, 2020 following conversion of $75,000 of principal and $9,300 of accrued interest during the fiscal
year ended June 30, 2020. Accordingly, there was no outstanding balance as of June 30, 2020.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
August
29, 2018 Securities Purchase Agreement
Effective
August 29, 2018, the Company entered into a securities purchase agreement with Eagle Equities, pursuant to which Eagle Equities purchased
a convertible promissory note (the “August 2018 Eagle Note”) from the Company in the aggregate principal amount of $105,000,
such principal and the interest thereon convertible into shares of the Company’s common stock at the option of Eagle Equities any
time after the six-month anniversary of the August 2018 Eagle Note. The transactions contemplated by the agreement closed on August 30,
2018. The maturity date of the August 29, 2018 Eagle Note was August 29, 2019. The August 2018 Eagle Note bore interest at a rate of
8% per annum, which interest was paid by the Company to Eagle Equities in shares of the Company’s common stock upon receipt of
a notice of conversion by the Company from Eagle Equities at any time after the six-month anniversary of the August 2018 Eagle Note.
In April 2020, Eagle Equities agreed to waive the 24% default interest on this note. The note was fully converted to common stock in
fiscal 2021.
October
2, 2018 Securities Purchase Agreement
Effective
October 2, 2018, the Company entered into a securities purchase agreement with Eagle Equities, pursuant to which Eagle Equities purchased
a convertible promissory note (the “October 2018 Eagle Note”) from the Company in the aggregate principal amount of $210,000,
such principal and the interest thereon convertible into shares of the Company’s common stock at the option of Eagle Equities any
time after the six-month anniversary of the October 2018 Eagle Note. The transactions contemplated by the purchase agreement closed on
October 3, 2018. Pursuant to the terms of the purchase agreement, Eagle Equities deducted $10,000 from the principal payment due under
the October 2018 Eagle Note, at the time of closing, to be applied to its legal expenses. The maturity date of the October 2018 Eagle
Note was October 2, 2019. The October 2018 Eagle Note shall bore interest at a rate of 8% per annum, which interest was paid by the Company
to Eagle Equities in shares of common stock upon receipt of a notice of conversion by the Company from Eagle Equities at any time after
the six-month anniversary of the October 2018 Eagle Note. In April 2020, Eagle Equities agreed to waive the 24% default interest on this
note. The note was fully converted to common stock in fiscal 2020.
November
30, 2018 Securities Purchase Agreement
Effective
November 30, 2018, the Company entered into a securities purchase agreement with Eagle Equities, pursuant to which Eagle Equities purchased
a convertible promissory note (the “November 2018 Eagle Note”) from the Company in the aggregate principal amount of $105,000,
such principal and the interest thereon convertible into shares of the Company’s common stock at the option of Eagle Equities any
time after the six-month anniversary of the November 2018 Eagle Note. The transactions contemplated by the purchase agreement closed
on December 3, 2018. Pursuant to the terms of the purchase agreement, Eagle Equities deducted $5,000 from the principal payment due under
the November 2018 Eagle Note, at the time of closing, to be applied to its legal expenses. The maturity date of the November 2018 Eagle
Note was November 30, 2019. The November 2018 Eagle Note bore interest at a rate of 8% per annum, which interest shall be paid by the
Company to Eagle Equities in shares of common stock upon receipt of a notice of conversion by the Company from Eagle Equities at any
time after the six-month anniversary of the November 2018 Eagle Note. In April 2020, Eagle Equities agreed to waive the 24% default interest
on this note. The note was fully converted to common stock in fiscal 2020.
December
24, 2018 Securities Purchase Agreement
Effective
December 24, 2018, the Company entered into a securities purchase agreement with Eagle Equities, pursuant to which Eagle Equities purchased
a convertible promissory note (the “December 2018 Eagle Note”) from the Company in the aggregate principal amount of $126,000,
such principal and the interest thereon convertible into shares of the Company’s common stock at the option of Eagle Equities any
time after the six-month anniversary of the December 2018 Eagle Note. The transactions contemplated by the purchase agreement closed
on December 24, 2018. Pursuant to the terms of the purchase agreement, Eagle Equities deducted $6,000 from the principal payment due
under the December 2018 Eagle Note, at the time of closing, to be applied to its legal expenses. The Company used the net proceeds from
the December 2018 Eagle Note to repay an outstanding convertible promissory note before such note became convertible. The maturity date
of the December 2018 Eagle Note was December 24, 2019. The December 2018 Eagle Note bore interest at a rate of 8% per annum, which interest
was paid by the Company to Eagle Equities in shares of common stock upon receipt of a notice of conversion by the Company from Eagle
Equities at any time after the six-month anniversary of the December 2018 Eagle Note. Upon an event of default, principal and accrued
interest would become immediately due and payable under the notes. Additionally, upon an event of default, both notes would accrue interest
at a default interest rate of 24% per annum or the highest rate of interest permitted by law. In April 2020, Eagle Equities agreed to
waive the 24% default interest on this note.
Eagle
Equities had the option to convert all or any amount of the principal amount of the notes issued to Eagle Equities above, at any time,
for shares of the Company’s common stock at a price ranging from 60% to 61% of the lowest closing bid price (the “Closing
Bid Price”) of the Company’s common stock as reported on the OTC Markets Group, Inc. quotation system for the ten prior trading
days, including the day upon which the Company receives a notice of conversion from Eagle Equities (the “Conversion Price”).
However, in the event that the Company’s common stock was restricted by the Depository Trust Company for any reason, the Conversion
Price was to be lowered to from 50% to 51% of the lowest Closing Bid Price for the duration of such restriction. If the Company failed
to maintain a reserve of shares of its common stock at least two and a half times the number of shares issuable upon conversion of all
the Eagle Notes for at least 60 days after the issuance of the notes issued to Eagle Equities, the conversion discount was to be increased
by 10%. Notwithstanding the foregoing, Eagle Equities was restricted from effecting a conversion if such conversion, along with other
shares of the Company’s common stock beneficially owned by Eagle Equities and its affiliates, exceeds 4.99% of the outstanding
shares of the Company’s common stock.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
The
above notes issued to Eagle Equities were treated as stock settled debt under ASC 480 and accordingly, the Company recorded a total of
$357,688 put premium, of which $224,131 and $133,557 were released to additional paid in capital following conversion of principal during
the fiscal year to June 30, 2020 and 2021, respectively.
The
total principal amount outstanding under the above Eagle Equities financing agreements, specifically the August 29, 2018 and the December
24, 2018 agreements was $205,500 as of June 30, 2020 and accrued interest totaled $26,990. There were $0 outstanding principal and accrued
interest under the above Eagle Equities financing agreements as of June 30, 2021 as a result of the fiscal 2021 conversions.
GS
Capital Financing Agreements
October
2, 2018 Securities Purchase Agreement
Effective
October 2, 2018, the Company entered into a securities purchase agreement with GS Capital, pursuant to which GS Capital purchased two
8% unsecured convertible redeemable notes (the “October 2, 2018 GS Notes”) from the Company in the aggregate principal amount
of $212,000, such principal and the interest thereon convertible into shares of the Company’s common stock. The purchase price
of $106,000 of the first note (the “October 2018 GS Note”) was paid in cash by GS Capital on October 3, 2018. After payment
of certain legal fees and expenses, net proceeds to the Company from the October 2018 GS Note totaled $100,700. The purchase price of
$106,000 of the second note (the “October 2, 2018 GS Back End Note”) was initially paid for by GS Capital issuing to the
Company an offsetting $106,000 collateralized secured note (the “October 2, 2018 GS Secured Note”). The terms of the October
2018 GS Back End Note require cash funding prior to any conversion thereunder, and such cash funding shall occur on or before June 2,
2019.
Both
the October 2, 2018 GS Note and the October 2, 2018 GS Back End Note, which was funded on February 27, 2019, matured on October 2, 2019,
upon which any outstanding principal and interest thereon is due and payable. The amounts cash funded plus accrued interest under both
the October 2018 GS Note and the October 2018 GS Back End Note are convertibles into shares of the Company’s common stock, at any
time after April 2, 2019, at a conversion price for each share of common stock equal to 61% of the lowest closing bid price of the Company’s
common stock for the ten prior trading days including the day upon which a notice of conversion is received by the Company from GS Capital,
subject to adjustment in certain events. The maturity date of the October 2, 2018 GS Back-Note was October 2019 and the notes were fully
converted to common stock in fiscal 2020.
January
22, 2020 GS Capital Securities Purchase Agreements
Effective
January 22, 2020, the Company entered into a securities purchase agreement with GS Capital, pursuant to which GS Capital purchased a
convertible promissory note (the “January 22, 2020 GS Note”) from the Company in the aggregate principal amount of $58,000,
such principal and the interest thereon convertible into shares of the Company’s common stock at the option of GS Capital any time
after the six-month anniversary of the January 22, 2020 GS Capital Note. The January 22, 2020 GS Note contained an original discount
of $3,500. The transactions contemplated by the GS Capital Securities Purchase Agreement closed on January 22, 2020. Pursuant to the
terms of the GS Capital Securities Purchase Agreement, GS Capital deducted $2,500 from the principal payment due under the January 22,
2020 GW Note, at the time of closing, to be applied to its legal expenses and received net cash proceeds of $52,000 on January 28, 2020.
The Company used the net proceeds from the January 22, 2020 GW Note for general working capital purposes. The maturity date of the January
22, 2020 GS Capital was January 22, 2021. The January 22, 2020 GS Capital Note bore interest at a rate of 10% per annum, which interest
may be paid by the Company to GS Capital in shares of the Company’s common stock; but was not payable until the January 22, 2020
GS Capital Note became payable, whether at the maturity date or upon acceleration or by prepayment. The conversion price for the January
22, 2020 GS Capital Note was equal to a 40% discount of the lowest closing bid price (“Lowest Trading Price”) of the Common
Stock for the ten trading days immediately prior to the delivery of a Notice of Conversion, including the day upon which a Notice of
Conversion is received.
Additionally,
GS Capital had the option to convert all or any amount of the principal face amount of the January 22, 2020 GS Capital Note at any time
from the date of issuance and ending on the later of the maturity date or the date the Default Amount was paid if an event of default
occurs, which is an amount between 112% and 130% of an amount equal to the then outstanding principal amount of the January 22, 2020
GS Capital Note plus any interest accrued, for shares of the Company’s common stock at the then-applicable conversion price. The
January 22, 2020 GS Note contained certain events of default, upon which principal and accrued interest would become immediately due
and payable. In addition, upon an event of default, interest on the outstanding principal would accrue at a default interest rate of
24% per annum.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
The
notes issued to GS capital above were treated as stock settled debt under ASC 480 and accordingly the Company recorded a total of $106,438
put premium which was expensed in fiscal 2020 of which $22,901 and $38,667 was released to additional paid in capital following conversion
of principal during the year ended June 30, 2020 and 2021, respectively.
The
Company recorded $8,802 of accrued interest and the total principal outstanding under the above GS Capital financing agreements was $58,000
as of June 30, 2020 following the conversion of $141,820 of principal and $76,397 of accrued interest during the year ended June 30,
2020. The total remaining principal outstanding and accrued interest under the above GS Capital financing agreements was $0 as of June
30, 2021 following conversion of $58,000 of principal and $8,508 of accrued interest during the year ended June 30, 2021.
Convertible
Note Issued with Consulting Agreement
August
10, 2017 Consulting Agreement
On
August 10, 2017, the Company entered into a consulting agreement, retroactive to May 16, 2017, with a certain consultant, pursuant to
which the consultant agreed to provide certain consulting and business advisory services in exchange for a $310,000 junior subordinated
convertible note. The maturity date of the August 10, 2017 Convertible Note was August 2019 and is currently past due (see Note 8). The
note accrues interest at a rate of 10% per annum and is convertible into common stock at the lesser of $750 or 65% of the three lowest
trades in the ten trading days prior to the conversion. The note was fully earned upon signing the agreement and matures on August 10,
2019. The Company accrued $155,000 related to this expense at June 30, 2017 and recorded the remaining $155,000 related to this expense
in fiscal year 2018. Upon an event of default, principal and accrued interest will become immediately due and payable under the note.
Additionally, upon an event of default, at the election of the holder, the note would accrue interest at a default interest rate of 18%
per annum or the highest rate of interest permitted by law. The consulting agreement had a three-month term and expired on August 16,
2017. An aggregate total of $578,212 of this note was bifurcated with the embedded conversion option recorded as a derivative liability
at fair value. During the year ended June 30, 2018, the consultant converted $140,000 of principal and $10,764 of interest. During the
year ended June 30, 2019, the consultant converted an additional $161,000 of principal and $19,418 of interest leaving a principal balance
owed of $9,000 at June 30, 2019. During the year ended June 30, 2020, the consultant converted an additional $500 of principal and $5,248
of interest such that the remaining principal outstanding and accrued interest under this note as of June 30, 2020 was $8,500 and $22,168,
respectively.
On
March 15, 2021, the Company entered into a Settlement and Mutual Release Agreement (the “Settlement Agreement”) with the
consultant whereby both parties agreed to settle all claims and liabilities under the August 10, 2017 Convertible note for a total of
$100,000 in the form of a convertible note. All other terms of the August 10, 2017 Convertible Note shall remain in full force and effect.
Both parties agree that all future penalties under this note are waived unless the Company fails to authorize to distribute the requested
shares upon conversion. The Company has the right to pay off the balance of any remaining amounts dues under this note in cash at any
time more than 60 days after March 15, 2021. Prior to the Settlement Agreement, the Company recorded total liabilities $56,762 consisting
of remaining principal amount of $8,500, accrued interest of $23,262 and accrued expenses of $25,000. Accordingly, the Company recognized
loss from settlement of debt of $43,238 during the year ended June 30, 2021 which is included in gain from settlement of debt, net in
the accompanying consolidated statements of operations.
The
total principal outstanding after adjustment due to the above-mentioned March 15, 2021 settlement agreement and accrued
interest under the August 10, 2017 Convertible Note was $80,000
and $3,738,
respectively, as of June 30, 2021 following conversion of $20,000
of principal during the year ended
June 30, 2021.
Power
Up Lending Group Financing Agreements
November
26, 2019 Securities Purchase Agreement
Effective
November 26, 2019, the Company entered into a securities purchase agreement with Power Up Lending Group Ltd. (“Power Up”),
pursuant to which Power Up purchased a convertible promissory note (the “November 26, 2019 Power Up Note”) from the Company
in the aggregate principal amount of $43,000, such principal and the interest thereon convertible into shares of the Company’s
common stock at the option of Power Up. The transaction closed on November 22, 2019 and the Company received payment on December 3, 2019
in the amount of $40,000, net of $2,500 paid directly toward legal fees and $500 to Power Up for due diligence fees. The maturity date
of the November 26, 2019 Power Up Note was November 26, 2020. The November 26, 2019, Power Up Note bore interest at a rate of 8% per
annum, which interest was paid by the Company to Power Up in shares of the Company’s common stock but was not payable until the
November 26, 2019 Power Up Note became payable, whether at the maturity date or upon acceleration or by prepayment. The note was fully
converted in fiscal 2020.
January
7, 2020 Power Up Lending Group Securities Purchase Agreement
Effective
January 7, 2020, the Company entered into a securities purchase agreement with Power Up Lending Group Ltd. (“Power Up”),
pursuant to which Power Up purchased a convertible promissory note (the “January 7, 2020 Power Up Note”) from the Company
in the aggregate principal amount of $75,000, such principal and the interest thereon convertible into shares of the Company’s
common stock at the option of Power Up. The transaction closed on January 7, 2020 and the Company received payment on January 13, 2020
in the amount of $72,000, net of $2,500 paid directly toward legal fees and $500 to Power Up for due diligence fees. The maturity date
of the January 7, 2020 Power Up Note was January 7, 2021. The January 7, 2020, Power Up Note bore interest at a rate of 8% per annum,
which interest was paid by the Company to Power Up in shares of the Company’s common stock, but not payable until the January 7,
2020 Power Up Note became payable, whether at the maturity date or upon acceleration or by prepayment.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
March
12, 2020 Power Up Lending Group Securities Purchase Agreement
Effective
March 12, 2020, the Company entered into a securities purchase agreement with Power Up Lending Group Ltd. (“Power Up”), pursuant
to which Power Up purchased a convertible promissory note (the “March 12, 2020 Power Up Note”) from the Company in the aggregate
principal amount of $43,000, such principal and the interest thereon convertible into shares of the Company’s common stock at the
option of Power Up. The transaction closed on March 12, 2020 and the Company received payment on March 5, 2020 in the amount of $40,000,
net of $2,500 paid directly toward legal fees and $500 to Power Up for due diligence fees. The maturity date of the March 12, 2020 Power
Up Note was March 12, 2021. The March 12, 2020, Power Up Note bore interest at a rate of 8% per annum, which interest may be paid by
the Company to Power Up in shares of the Company’s common stock but was not payable until the March 12, 2020 Power Up Note became
payable, whether at the maturity date or upon acceleration or by prepayment.
All
the notes issued above to Power Up contained certain events of default, upon which principal and accrued interest would become immediately
due and payable. In addition, upon an event of default, interest on the outstanding principal would accrue at a default interest rate
of 22% per annum.
Additionally,
Power Up had the option to convert all or any amount of the principal face amount of the notes issued to Power Up, starting on certain
dates as defined in the note agreements and ending on the later of the maturity date or the date the Default Amount is paid if an event
of default occurs, which was an amount equal to 150% of an amount equal to the then outstanding principal amount of the notes plus any
interest accrued, for shares of the Company’s common stock at the then-applicable conversion price.
The
conversion price for the above Power Up notes was $3,050, subject to certain Market Price (as defined below) adjustment. If the Market
Price was greater than or equal to $5,000, the conversion price was to be the greater of 65% of the Market Price (“Variable Conversion
Price”) and $3,050. In the event Market Price was less than $5,000, the conversion price was to be the Variable Conversion Price.
As defined in the note agreements, the “Market Price” was the average of the lowest three closing bid prices during the ten
day trading period prior to and including the day the Company receives a notice of conversion from Power Up on the electronic quotation
system or applicable principal securities exchange or trading market or, if no closing bid price of such security is available in any
of the foregoing manners, the average of the closing bid prices of any market makers for such security that are listed in the “pink
sheets” during the ten prior trading days, including the day upon which the Company receives a notice of conversion from Power
Up. Notwithstanding the foregoing, Power Up was restricted from effecting a conversion if such conversion, along with other shares of
the Company’s common stock beneficially owned by Power Up and its affiliates, exceeds 4.99% of the outstanding shares of the Company’s
common stock. An aggregate initial total of $422,557 of these notes were bifurcated with the embedded conversion option recorded as derivative
liabilities at fair value.
The
total principal amount outstanding under the above Power Up financing agreements, specifically the January 7, 2020 and March 12, 2020
Power Up Notes, was $118,000 and accrued interest of $3,903 as of June 30, 2020. The total principal amount outstanding under the above
Power Up financing agreement, specifically the January 7, 2020 and March 12, 2020 Power Up Notes, was $0 and accrued interest of $0 as
of June 30, 2021 following repayment in cash of $43,000 of the principal balance and $1,816 of accrued interest and conversions into
common stock during the year ended June 30, 2021. Accordingly, there was no outstanding principal balance as of June 30, 2021.
Redstart
Holdings Corp Financing Agreement
May
23, 2019 Securities Purchase Agreement
Effective
May 23, 2019, the Company issued a convertible promissory note (the “May 23 Redstart Holdings Note”) to Redstart Holdings
Corp (“Redstart Holdings”) in the aggregate principal amount of $133,000, with principal and the interest thereon convertible
into shares of the Company’s common stock at the option of Redstart Holdings any time after 180 days of issuance. At the time of
closing on May 31, 2019, Redstart Holdings deducted $3,000 from the principal payment due under the May 2019 Redstart Holdings Note to
be applied to its legal expenses, such that the Company received aggregate net proceeds of $130,000 at closing. The maturity date of
the May 2019 Redstart Holdings Note was May 23, 2020 and bore interest at a rate of 8% per annum. The total principal amount outstanding
and accrued interest under the above Redstart Holdings financing agreement, specifically the May 23, 2019 agreement at June 30, 2019
was $133,000 and $1,137 respectively and as of June 30, 2020 total principal amount outstanding and accrued interest totaled $0 and $0
respectively following conversion of $133,000 of the principal balance and $5,320 of accrued interest during the year ended June 30,
2020.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
Odyssey
Capital Financing Agreements
July
30, 2019 Securities Purchase Agreement
Effective
July 30, 2019, the Company entered into a securities purchase agreement with Odyssey Capital Funding LLC, (“Odyssey”), pursuant
to which Odyssey purchased a convertible promissory note (the “July 30, 2019 Odyssey Note”) from the Company in the aggregate
principal amount of $320,000, such principal and the interest thereon convertible into shares of the Company’s common stock at
the option of Odyssey. The July 30, 2019 Odyssey Note contains an original discount of $25,000. The transaction closed on July 30, 2019
and the Company received payment on August 1, 2019 in the amount of $295,000, of which $10,000 was paid directly toward legal fees, resulting
in net cash proceeds of $285,000. The maturity date of the July 30, 2019 Odyssey Note was July 30, 2020. The July 2019 Odyssey Note bore
interest at a rate of 10% per annum, which interest may be paid by the Company to Odyssey in shares of the Company’s common stock
but was not payable until the July 30, 2019 Odyssey Note became payable, whether at the maturity date or upon acceleration or by prepayment.
The July 30, 2019 Odyssey Note contained certain events of default, upon which principal and accrued interest would become immediately
due and payable. In addition, upon an event of default, interest on the outstanding principal would accrue at a default interest rate
of 24% per annum. The note was treated as stock settled debt under ASC 480 and accordingly the Company recorded a $172,308 put premium.
Additionally,
Odyssey had the option to convert all or any amount of the principal face amount of the July 30, 2019 Odyssey Note, starting on January
31, 2020 and ending on the later of the maturity date or the date the Default Amount was paid if an event of default occurred, which
was an amount equal to 120% of an amount equal to the then outstanding principal amount of the July 30, 2019 Odyssey Note plus any interest
accrued from July 30, 2019 at the default interest rate of 24% per annum for shares of the Company’s common stock at the then-applicable
conversion price.
The
conversion price for the July 30, 2019 Odyssey Note was equal to 65% of the lowest closing bid price of the Common Stock as reported
on the OTC Markets on which the Company’s shares were then traded or any exchange upon which the Common Stock may be traded in
the future, for the ten prior trading days including the day upon which a Notice of Conversion is received by the Company.
The
total principal amount outstanding under the above Odyssey financing agreement, specifically the July 30, 2019 Odyssey Note, was $0 and
accrued interest of $0 as of June 30, 2020 following conversion of $320,000 of the principal balance and $23,220 of accrued interest
during the year ended June 30, 2020 resulting in full repayment of the note and a full reduction of the put premium. The note was fully
converted in fiscal 2020.
Auctus
Fund Financing Agreements
August
30, 2019 Securities Purchase Agreement
Effective
August 30, 2019, the Company entered into a securities purchase agreement with Auctus Fund, LLC (“Auctus”), pursuant to which
Auctus purchased a convertible promissory note (the “August 30, 2019 Auctus Note”) from the Company in the aggregate principal
amount of $550,000, such principal and the interest thereon convertible into shares of the Company’s common stock at the option
of Auctus. The transaction closed on August 30, 2019 and the Company received payment on September 4, 2019 in the amount of $550,000,
of which $5,000 was paid directly toward legal fees and $40,000 to Auctus for due diligence fees resulting in net cash proceeds of $505,000.
The maturity date of the August 30, 2019 Auctus Note was August 30, 2020 and is currently past due. The August 30, 2019 Auctus Note bears
interest at a rate of 10% per annum, but not payable until the August 30, 2019 Auctus Note became payable, whether at the maturity date
or upon acceleration or by prepayment. The note is treated as stock settled debt under ASC 480 and accordingly the Company recorded a
$366,667 put premium. The August 30, 2019 Auctus Note may not be prepaid without the written consent of Auctus. Any amount of principal
or interest which is not paid when due shall bear interest at the rate of 24% per annum.
Additionally,
Auctus has the option to convert all or any amount of the principal face amount and accrued interest of the August 30, 2019 Auctus Note,
at any time following the issue date and ending on the later of the maturity date or the date of payment of the Default Amount if an
event of default occurs, which is an amount equal to 125% of an amount equal to the then outstanding principal amount of the August 30,
2019 Auctus Note (but not less than $15,000) plus any interest accrued from August 30, 2019 at the default interest rate of 24% per annum,
for shares of the Company’s common stock at the then-applicable conversion price. Upon the holder’s election to convert accrued
interest, default interest or any penalty amounts as stipulated, the Company may elect to pay those amounts in cash. The note may also
be prepaid by the Company at any time between the date of issuance and August 13, 2020 at 135% multiplied by the sum of (a) the then
outstanding principal amount plus (b) accrued and unpaid interest plus (c) default interests, if any.
The
conversion price for the August 30, 2019 Auctus Note shall be the Variable Conversion Price, being 60% of the Market Price on the date
of conversion. Notwithstanding the foregoing, Auctus shall be restricted from effecting a conversion if such conversion, along with other
shares of the Company’s common stock beneficially owned by Auctus and its affiliates, exceeds 4.99% of the outstanding shares of
the Company’s common stock.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
In
connection with the issuance of the August 2019 Auctus Note, the Company issued common stock purchase warrants to Auctus to purchase
450 shares of the Company’s common stock (the “First Warrant”) as a commitment fee upon the terms and subject to the
limitations and conditions set forth in such First Warrant at an “Exercise Price” of $2,250. In connection with the issuance
of the Note, the Company shall issue a common stock purchase warrant to Buyer to purchase 300 shares of the Company’s common stock
(the “Second Warrant”) as a commitment fee upon the terms and subject to the limitations and conditions set forth in such
Second Warrant at an “Exercise Price” of $3,330. In connection with the issuance of the Note, the Company shall issue a common
stock purchase warrant to Buyer to purchase 225 shares of the Company’s common stock (the “Third Warrant”) as a commitment
fee upon the terms and subject to the limitations and conditions set forth in such Third Warrant at an “Exercise Price” of
$4,500. The First Warrant, Second Warrant, and Third Warrant shall collectively be referred as the “Warrants”. The Warrants
have an “Exercise Period” of five years from the date of issuance being August 30, 2019. Under the terms of the Purchase
Agreement and the Warrants, the Selling Security Holder may not either convert the Notes nor exercise the Warrants to the extent (but
only to the extent) that the Selling Security Holder or any of its affiliates would beneficially own a number of shares of our Common
Stock which would exceed 4.99% of our outstanding shares. The Company accounted for the warrants by using the relative fair value method
and recorded debt discount from the relative fair value of the warrants of $375,905 using a simple binomial lattice model.
In
connection with the Purchase Agreement, the Company and the Purchaser entered into a Registration Rights Agreement (the “Registration
Rights Agreement”). Pursuant to the Registration Rights Agreement, the Company agreed to register the shares of Common Stock underlying
the Securities in a Registration Statement with the SEC as well as the Commitment Shares (as defined herein). The Registration Rights
Agreement contains customary representations, warranties, agreements and indemnification rights and obligations of the parties.
The
Note is subject to customary default provisions and also includes a cross-default provision which provides that a breach or default by
the Borrower of any covenant or other term or condition contained in any of the Other Agreements (as defined therein), after the passage
of all applicable notice and cure or grace periods, shall, at the option of the Holder, be considered a default under this Note and the
Other Agreements. Upon occurrence of any such event, the Holder shall be entitled (but in no event required) to apply all rights and
remedies of the Holder under the terms of this Note and the Other Agreements by reason of a default under said Other Agreements or the
Note.
The
August 30, 2019 Auctus Note contain certain events of default, upon which principal and accrued interest will become immediately due
and payable. In addition, upon an event of default, interest on the outstanding principal shall accrue at a default interest rate of
24% per annum.
The
total principal amount outstanding under the above Auctus financing agreement, specifically the August 30, 2019 Auctus Note, was $358,965
and accrued interest of $486 as of June 30, 2020 following conversion of $191,035 of the principal balance and $43,176 of accrued interest
during the year ended June 30, 2020. Accordingly, $127,356 of the put premium was released in respect of the August 30, 2019 Auctus Note
during the year ended June 30, 2020 following conversion of the principal balance.
The
total principal amount outstanding under the above Auctus financing agreement, specifically the August 30, 2019 Auctus Note, was $32,848
and accrued interest of $0 as of June 30, 2021 following conversion of $326,117 of the principal balance and $39,536 of accrued interest
during the year ended June 30, 2021. Accordingly, $217,411 of the put premium was released in respect of the August 30, 2019 Auctus Note
during the year ended June 30, 2021 following conversion of the principal balance.
GW
Holdings Securities Purchase Agreements
October
1, 2019 Securities Purchase Agreement
Effective
October 1, 2019, the Company entered into a securities purchase agreement with GW Holdings, pursuant to which GW Holdings purchased a
convertible promissory note (the “October 1, 2019 GW Note”) from the Company in the aggregate principal amount of $131,000,
such principal and the interest thereon convertible into shares of the Company’s common stock at the option of GW Holdings any
time after the six-month anniversary of the October 1, 2019 GW Holdings Note. The transactions contemplated by the GW Holdings Securities
Purchase Agreement closed on October 1, 2019. Pursuant to the terms of the GW Holdings Securities Purchase Agreement, the lender deducted
$6,000 from the principal payment due under the October 1, 2019 GW Note, at the time of closing, to be applied to its legal expenses.
The Company used the net proceeds of $125,000 from the October 1, 2019 GW Note for general working capital purposes. The maturity date
of the October 1, 2019 GW Holdings was October 1, 2020 and is currently past due. The October 1, 2019 GW Holdings Note bore interest
at a rate of 8% per annum, which interest was paid by the Company to GW Holdings in shares of the Company’s common stock; but was
not payable until the October 1, 2019 GW Holdings Note became payable, whether at the maturity date or upon acceleration or by prepayment.
December
10, 2020 Securities Purchase Agreement
Effective
December 10, 2020, the Company entered into a securities purchase agreement with GW Holdings, pursuant to which GW Holdings purchased
a convertible promissory note (the “December 10, 2020 GW Note”) from the Company in the aggregate principal amount of $131,000,
such principal and the interest thereon convertible into shares of the Company’s common stock at the option of GW Holdings anytime
from the issuance of the December 10, 2020 GW Holdings Note. The transactions contemplated by the GW Holdings Securities Purchase Agreement
closed on December 10, 2020. Pursuant to the terms of the GW Holdings Securities Purchase Agreement, the lender deducted $6,000 from
the principal payment due under the December 10, 2020 GW Note, at the time of closing, to be applied to its legal expenses. The Company
intends to use the net proceeds of $125,000 from the December 10, 2020 GW Note for general working capital purposes. The maturity date
of the December 10, 2020 GW Holdings is December 10, 2021. The December 10, 2020 GW Holdings Note bears interest at a rate of 8% per
annum, which interest may be paid by the Company to GW Holdings in shares of the Company’s common stock; but shall not be payable
until the December 10, 2020 GW Holdings Note becomes payable, whether at the maturity date or upon acceleration or by prepayment.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
The
above notes issued to GW Holdings contain certain events of default, upon which principal and accrued interest will become immediately
due and payable. In addition, upon an event of default, interest on the outstanding principal shall accrue at a default interest rate
of 24% per annum, or if such rate is usurious or not permitted by current law, then at the highest rate of interest permitted by law.
Further, certain events of default may trigger penalty and liquidated damage provisions.
Additionally,
GW Holdings has the option to convert all or any amount of the principal face amount of the notes issued to GW Holdings at any time from
the date of issuance and ending on the later of the maturity date or the date the Default Amount is paid if an event of default occurs,
which is an amount between 110% and 150% of an amount equal to the then outstanding principal amount of such notes plus any interest
accrued, for shares of the Company’s common stock at the then-applicable conversion price.
The
conversion price for the above GW Holdings notes shall be equal to a 40% discount of the lowest closing bid price (“Lowest Trading
Price”) of the Common Stock for the ten trading days immediately prior to the delivery of a Notice of Conversion, including the
day upon which a Notice of Conversion is received. Notwithstanding the foregoing, GW Holdings shall be restricted from effecting a conversion
if such conversion, along with other shares of the Company’s common stock beneficially owned by GW Holdings and its affiliates,
exceeds 4.99% of the outstanding shares of the Company’s common stock which may be increased up to 9.99% upon 60 days prior written
notice by the GW Holdings to the Company.
These
notes are treated as stock settled debt under ASC 480 and accordingly the Company recorded a total of $174,666 put premium.
The
total principal amount outstanding under the above October 1, 2019 GW Holdings financing agreement was $30,000 and accrued interest of
$1,776 as of June 30, 2020 following conversion of $101,000 of the principal balance and $5,082 of accrued interest during the year ended
June 30, 2020. The total principal amount and accrued interest outstanding under the above October 1, 2019 GW Holdings financing agreement
was $0 as of June 30, 2021 following conversion of $30,000 of the principal balance and $3,877 of accrued interest during the year ended
June 30, 2021. Accordingly, $67,333 and $20,000 of the put premium was reclassed to additional paid in capital during the year ended
June 30, 2020 and 2021, respectively, following conversion of the principal balance. This note was fully converted into common stock
in fiscal 2021.
The
total principal amount outstanding under the above December 10, 2020 GW Holdings financing agreement, was $90,000 and accrued interest
of $4,636 as of June 30, 2021 following conversion of $41,000 of the principal balance and $1,084 of accrued interest during the year
ended June 30, 2021. Accordingly, $27,333 of the put premium was reclassed to additional paid in capital in respect of the October 1,
2019 GW Holdings Note during the year ended June 30, 2021 following conversion of the principal balance.
Crown
Bridge Securities Purchase Agreements
Effective
October 3, 2019, the Company entered into a securities purchase agreement with Crown Bridge Partners, pursuant to which Crown Bridge
purchased a convertible promissory note (the “October 3, 2019 Crown Bridge Note”) from the Company in the aggregate principal
amount of $108,000, such principal and the interest thereon convertible into shares of the Company’s common stock at the option
of Crown Bridge any time from the of issuance of the of the October 3, 2019 Crown Bridge Note. The transactions contemplated by the Crown
Bridge Securities Purchase Agreement closed on October 3, 2019. Pursuant to the terms of the Crown Bridge Securities Purchase Agreement,
Crown Bridge deducted $3,000 from the principal payment due under the October 3, 2019 Crown Bridge Note, at the time of closing, to be
applied to its legal expenses, and there was a $5,000 original issuance discount resulting in $100,000 net proceeds to the Company. The
Company intends to use the net proceeds from the October 3, 2019 Crown Bridge Note for general working capital purposes. The maturity
date of the October 3, 2019 Crown Bridge was October 3, 2020 and is currently past due. The October 3, 2019 Crown Bridge Note bears interest
at a rate of 10% per annum, which interest may be paid by the Company to Crown Bridge in shares of the Company’s common stock;
but shall not be payable until the October 2019 Crown Bridge Note becomes payable, whether at the maturity date or upon acceleration
or by prepayment.
Additionally,
Crown Bridge has the option to convert all or any amount of the principal face amount of the October 3, 2019 Crown Bridge Note at any
time from the date of issuance and ending on the later of the maturity date or the date the Default Amount is paid if an event of default
occurs, which is an amount between 110% and 150% of an amount equal to the then outstanding principal amount of the October 3, 2019 Crown
Bridge Note plus any interest accrued, for shares of the Company’s common stock at the then-applicable conversion price.
The
conversion price for the October 3, 2019 Crown Bridge Note shall be equal to a 40% discount of the lowest closing bid price (“Lowest
Trading Price”) of the Common Stock for the ten trading days immediately prior to the delivery of a Notice of Conversion, including
the day upon which a Notice of Conversion is received. Notwithstanding the foregoing, Crown Bridge shall be restricted from effecting
a conversion if such conversion, along with other shares of the Company’s common stock beneficially owned by Crown Bridge and its
affiliates, exceeds 4.99% of the outstanding shares of the Company’s common stock which may be increased up to 9.99% upon 60 days
prior written notice by the Crown Bridge to the Company. The note is treated as stock settled debt under ASC 480 and accordingly the
Company recorded a $72,000 put premium.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
The
October 3, 2019 Crown Bridge Note contain certain events of default, upon which principal and accrued interest will become immediately
due and payable. In addition, upon an event of default, interest on the outstanding principal shall accrue at a default interest rate
of 15% per annum, or if such rate is usurious or not permitted by current law, then at the highest rate of interest permitted by law.
Further, certain events of default may trigger penalty and liquidated damage provisions.
The
total principal amount outstanding under the above Crown Bridge financing agreement was $65,280 and accrued interest of $7,232 as of
as of June 30, 2020 following conversion of $42,720 of the principal balance during the year ended June 30, 2020. Accordingly, $28,480
of the put premium was released in respect of the October 3, 2019 Crown Bridge Note during the year ended June 30, 2020 following conversion
of the principal balance.
The
total principal amount outstanding under the above Crown Bridge financing agreement was $65,280 and accrued interest of $16,138 as of
as of June 30, 2021.
There
were 15,000 unissued shares which were considered issuable for accounting purposes during the 1st quarter of fiscal 2021 related
to a conversion notice dated and received on September 16, 2020. In November 2020, the Company was notified by the note holder of the
cancellation of this conversion notice as a result of the reverse stock split and as such the Company reversed the effects of this transaction
thereby increasing the principal balance by $9,600 and put premium by $6,400 and a corresponding decrease in equity of $16,000.
Ader
Alef Securities Purchase Agreements
Effective
January 13, 2020, the Company entered into a securities purchase agreement with Ader Alef, pursuant to which Ader Alef purchased a convertible
promissory note (the “January 13, 2020 Ader Alef Note”) from the Company in the aggregate principal amount of $110,250, such
principal and the interest thereon convertible into shares of the Company’s common stock at the option of Ader Alef any time after
the six-month anniversary of the January 13, 2020 Ader Alef Note. The January 13, 2020 Ader Alef Note contained an original discount
of $5,250. The transactions contemplated by the Ader Alef Securities Purchase Agreement closed on January 13, 2020. Pursuant to the terms
of the Ader Alef Securities Purchase Agreement, Ader Alef deducted $5,000 from the principal payment due under the January 13, 2020 Ader
Alef Note at the time of closing, to be applied to its legal expenses and the Company received net cash proceeds of $100,000 on January
15, 2020. The Company used the net proceeds from the January 13, 2020 Ader Alef Note for general working capital purposes. The maturity
date of the January 13, 2020 Ader Alef was January 13, 2021. The January 13, 2020 Ader Alef Note bore interest at a rate of 8% per annum,
which interest may be paid by the Company to Ader Alef in shares of the Company’s common stock; but was not payable until the January
13, 2020 Ader Alef Note became payable, whether at the maturity date or upon acceleration or by prepayment.
Additionally,
Ader Alef had the option to convert all or any amount of the principal face amount of the January 13, 2020 Ader Alef Note at any time
from the date of issuance and ending on the later of the maturity date or the date the Default Amount was paid if an event of default
occurs, which was an amount between 120% and 150% of an amount equal to the then outstanding principal amount of the January 13, 2020
Ader Alef Note plus any interest accrued, for shares of the Company’s common stock at the then-applicable conversion price.
The
conversion price for the January 13, 2020 Ader Alef Note during the first 6 months the January 13, 2020 Ader Alef Note was fixed at $2.50
and thereafter would be equal to a 35% discount of the lowest closing bid price (“Lowest Trading Price”) of the Common Stock
for the ten trading days immediately prior to the delivery of a Notice of Conversion, including the day upon which a Notice of Conversion
was received. Notwithstanding the foregoing, Ader Alef was restricted from effecting a conversion if such conversion, along with other
shares of the Company’s common stock beneficially owned by Ader Alef and its affiliates, exceeds 4.99% of the outstanding shares
of the Company’s common stock which may be increased up to 9.99% upon 60 days prior written notice by the Ader Alef to the Company.
The note was treated as stock settled debt under ASC 480 and accordingly the Company recorded a $59,365 put premium.
The
January 13, 2020 Ader Alef Note contained certain events of default, upon which principal and accrued interest would become immediately
due and payable. In addition, upon an event of default, interest on the outstanding principal would accrue at a default interest rate
of 24% per annum, or if such rate was usurious or not permitted by current law, then at the highest rate of interest permitted by law.
Further, certain events of default may trigger penalty and liquidated damage provisions.
The
total principal amount outstanding under the above Ader Alef financing agreement was $110,250 and accrued interest of $4,073 as of June
30, 2020. The total principal amount outstanding and accrued interest under the above Ader Alef financing agreement was $0 as of as of
June 30, 2021 following conversion of $110,250 of the principal balance and $7,493 accrued interest during the year ended June 30, 2021.
Accordingly, $59,365 of the put premium was released in respect of the Ader Alef Note during the year ended June 30, 2021 following conversion
of the principal balance. This note was fully converted into common stock in fiscal 2021.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
LG
Capital Securities Purchase Agreements
Effective
February 19, 2020, the Company entered into a securities purchase agreement with LG Capital Funding, LLC (“LG Capital”),
pursuant to which LG Capital purchased a convertible promissory note (the “February 19, 2020 LG Capital Note”) from the Company
in the aggregate principal amount of $75,000, such principal and the interest thereon convertible into shares of the Company’s
common stock at the option of LG Capital any time after the six month anniversary of the February 19, 2020 LG Capital Note. The February
19, 2020 LG Capital Note contained an original discount of $3,750. The transactions contemplated by the LG Capital Securities Purchase
Agreement closed on March 4, 2020. Pursuant to the terms of the LG Capital Securities Purchase Agreement, LG Capital deducted $2,500
from the principal payment due under the February 19, 2020 LG Capital Note at the time of closing, to be applied to its legal expenses
and the Company received net cash proceeds of $71,250 on March 25, 2020. The Company used the net proceeds from the February 19, 2020
LG Capital Note for general working capital purposes. The maturity date of the February 19, 2020 LG Capital Note was February 19, 2021.
The February 19, 2020 LG Capital Note bore interest at a rate of 8% per annum, which interest was paid by the Company to LG Capital in
shares of the Company’s common stock; but was not payable until the February 19, 2020 LG Capital Note became payable, whether at
the maturity date or upon acceleration or by prepayment.
During
the first 60 to 180 days following the date of the note, the Company had the right to prepay the principal and accrued but unpaid interest
due under the February 19, 2020 LG Capital Note, together with any other amounts that the Company may owe the holder under the terms
of the note, at a premium ranging from 112% to 135% as defined in the note agreement. After this initial 180-day period, the Company
did not have a right to prepay the February 19, 2020 LG Capital Note.
The
conversion price for the February 19, 2020 LG Capital Note during the first 6 months the February 19, 2020 LG Capital Note was fixed
at $500 and thereafter was equal to a 35% discount of the lowest closing bid price (“Lowest Trading Price”) of the Common
Stock for the ten trading days immediately prior to the delivery of a Notice of Conversion, including the day upon which a Notice of
Conversion was received. Notwithstanding the foregoing, LG Capital was restricted from effecting a conversion if such conversion, along
with other shares of the Company’s common stock beneficially owned by LG Capital and its affiliates, exceeds 9.99% of the outstanding
shares of the Company’s common stock. The note was treated as stock settled debt under ASC 480 and accordingly the Company recorded
a $40,385 put premium.
The
February 19, 2020 LG Capital Note contained certain events of default, upon which principal and accrued interest would become immediately
due and payable. In addition, upon an event of default, interest on the outstanding principal would accrue at a default interest rate
of 24% per annum, or if such rate was usurious or not permitted by current law, then at the highest rate of interest permitted by law.
Further, certain events of default may trigger penalty and liquidated damage provisions.
The
total principal amount outstanding under the above LG Capital financing agreement was $75,000 and accrued interest of $2,164 as of June
30, 2020. The total principal amount outstanding and accrued interest under the above LG Capital financing agreement was $0 as of June
30 2021 following conversion of $75,000 of the principal balance and $5,421 accrued interest during the year ended June 30, 2021. Accordingly,
$40,385 of the put premium was released in respect of the February 19, 2020 LG Capital Note during the year ended June 30, 2021 following
conversion of the principal balance. This note was fully converted into common stock in fiscal 2021.
There
were 9,427 unissued shares which were considered issuable for accounting purposes during the first quarter of fiscal 2021 related to
a conversion notice dated and received on September 9, 2020. In November 2020, the Company was notified by the note holder of the cancellation
of this conversion notice as a result of the reverse stock split and as such the Company reversed the effects of this transaction thereby
increasing the principal balance by $10,000, accrued interest of $416 and put premium by $5,385 and a corresponding decrease in equity
of $15,801.
Geneva
Roth Remark Securities Purchase Agreements
December
2, 2020 Securities Purchase Agreement
Effective
December 2, 2020, the Company entered into a securities purchase agreement with Geneva Roth Remark Holdings, Inc.(“Geneva Roth”),
pursuant to which Geneva Roth purchased a convertible promissory note (the “December 2, 2020 Geneva Roth”) from the Company
in the aggregate principal amount of $78,000, such principal and the interest thereon convertible into shares of the Company’s
common stock at the option of Geneva Roth any time after the six month anniversary of the December 2, 2020 Geneva Roth. The December
2, 2020 Geneva Roth contains an original discount of $3,000. The Company intends to use the net proceeds from the December 2, 2020 Geneva
Roth for general working capital purposes. The maturity date of the December 2, 2020 Geneva Roth Note is December 2, 2021. The December
2, 2020 Geneva Roth Note bears interest at a rate of 8% per annum, which interest may be paid by the Company to Geneva Roth in shares
of the Company’s common stock; but shall not be payable until the December 2, 2020 Geneva Roth Note becomes payable, whether at
the maturity date or upon acceleration or by prepayment.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
January
5, 2021 Securities Purchase Agreement
Effective
January 5, 2021, the Company entered into a securities purchase agreement with Geneva Roth Remark Holdings, Inc., pursuant to which Geneva
Roth purchased a convertible promissory note (the “January 5, 2021 Geneva Roth”) from the Company in the aggregate principal
amount of $68,500, such principal and the interest thereon convertible into shares of the Company’s common stock at the option
of Geneva Roth any time after the six-month anniversary of the January 5, 2021 Geneva Roth. The January 5, 2021 Geneva Roth contains
an original issue discount of $3,500. The Company intends to use the net proceeds from the January 5, 2021 Geneva Roth for general working
capital purposes. The maturity date of the January 5, 2021 Geneva Roth Note is January 5, 2022. The January 5, 2021 Geneva Roth Note
bears interest at a rate of 8% per annum, which interest may be paid by the Company to Geneva Roth in shares of the Company’s common
stock; but shall not be payable until the January 5, 2021 Geneva Roth Note becomes payable, whether at the maturity date or upon acceleration
or by prepayment.
March
16, 2021 Securities Purchase Agreement
Effective
March 16, 2021, the Company entered into a securities purchase agreement with Geneva Roth Remark Holdings, Inc., pursuant to which Geneva
Roth purchased a convertible promissory note (the “March 16, 2021 Geneva Roth”) from the Company in the aggregate principal
amount of $63,500, such principal and the interest thereon convertible into shares of the Company’s common stock at the option
of Geneva Roth any time after the six-month anniversary of the March 16, 2021 Geneva Roth. The March 16, 2021 Geneva Roth contains an
original discount of $3,500. The Company intends to use the net proceeds from the March 16, 2021 Geneva Roth for general working capital
purposes.
The
maturity date of the March 16, 2021 Geneva Roth Note is March 16, 2022. The March 16, 2021 Geneva Roth Note bears interest at a rate
of 8% per annum, which interest may be paid by the Company to Geneva Roth in shares of the Company’s common stock; but shall not
be payable until the March 16, 2021 Geneva Roth Note becomes payable, whether at the maturity date or upon acceleration or by prepayment.
During
the first 60 to 180 days following the date of these notes, the Company has the right to prepay the principal and accrued but unpaid
interest due under the above notes issued to Geneva Roth, together with any other amounts that the Company may owe the holder under the
terms of the note, at a premium ranging from 110% to 129% as defined in the note agreement. After this initial 180-day period, the Company
does not have a right to prepay such notes.
The
conversion price for the above Geneva Roth notes shall be equal to a 35%
discount of the market price based on the average of the lowest three trading prices of the Common Stock for the ten trading days
immediately prior to the delivery of a Notice of Conversion. Notwithstanding the foregoing, Geneva Roth shall be restricted from effecting
a conversion if such conversion, along with other shares of the Company’s common stock beneficially owned by Geneva Roth and its
affiliates, exceeds 9.99%
of the outstanding shares of the Company’s common stock. These
notes are treated as stock settled debt under ASC 480 and accordingly the Company recorded a total of $113,077
put premium for the three notes.
The
above Geneva Roth notes contain certain events of default, upon which principal and accrued interest will become immediately due and
payable. In addition, upon an event of default, interest on the outstanding principal shall accrue at a default interest rate of 22%
per annum, or if such rate is usurious or not permitted by current law, then at the highest rate of interest permitted by law. Further,
certain events of default may trigger penalty and liquidated damage provisions.
The
total principal amounts outstanding under the above Geneva Roth financing agreements were $132,000 and accrued interest of $3,477 as
of June 30, 2021 following conversion of $78,000 of the principal balance and $3,120 accrued interest during the year ended June 30,
2021. Accordingly, $42,000 of the put premium was released in respect of the Geneva Roth financing agreements during the year ended June
30, 2021 following conversion of the principal balance.
Amortization
of debt discounts
The
Company recorded $211,000 and $728,904 of debt discounts related to the above note issuances during the years ended June 30, 2021 and
2020, respectively. The Company recorded $498,160 and $836,724 of put premiums related to the above note issuances during the years ended
June 30, 2021 and 2020, respectively. The debt discounts are being amortized over the term of the debt and the put premiums are expensed
on issuance of the debt with the liability released to additional paid in capital on conversion of the principal.
Amortization
of all debt discounts for the years ended June 30, 2021 and 2020 was $136,527 and $734,130, respectively.
The
Company reclassified $590,504 and $874,924 in put premiums to additional paid in capital following conversions during the year ended
June 30, 2021 and 2020, respectively.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
NOTE
7 – INCOME TAXES
The
Company follows ASC 740-10-10, under which an entity recognizes deferred tax assets and liabilities for future tax consequences or for
events that were previously recognized in the Company’s financial statements or tax returns. The measurement of deferred tax assets
and liabilities is based on enacted tax law provisions. The effects of future changes in tax laws or rates are not anticipated. Through
June 30, 2010, the Company operated exclusively in Australia. The Company was wholly subject to Australian income tax laws and regulations,
which are administered by the Australian Taxation Office for the years ended June 30, 2010 and all prior years.
On
November 23, 2010, the Company was incorporated in the state of Delaware. In January 2011, the Company acquired all of the outstanding
shares of Propanc PTY LTD on a one-for-one basis with Propanc PTY LTD becoming a wholly owned subsidiary of the Company. As a result
of these transactions, the Company is subject to the income tax laws of both the United States and Australia for the years ended June
30, 2013 through June 30, 2021.
The
reconciliation of income tax expense computed at the U.S. federal statutory rate of 21% to the income tax provision for the years ended
June 30, 2021 and 2020 is as follows:
SCHEDULE OF INCOME TAX PROVISION
| US |
|
June
30, 2021 |
|
|
June
30, 2020 |
|
| |
|
Year
Ended |
|
| US |
|
June
30, 2021 |
|
|
June
30, 2020 |
|
| Loss
before Income taxes |
|
$ |
(2,025,947 |
) |
|
$ |
(4,740,723 |
) |
| |
|
|
|
|
|
|
|
|
| Taxes
under statutory US tax rates |
|
$ |
(425,449 |
) |
|
$ |
(995,552 |
) |
| Increase
(decrease) in valuation allowance |
|
|
1,146,001 |
|
|
|
1,137,716 |
|
| Prior
period adjustment |
|
|
(1,063,710 |
) |
|
|
(14,624 |
|
| Foreign
tax rate differential |
|
|
(51,169 |
) |
|
|
(128,492 |
) |
| Income
tax rate change |
|
|
392,767 |
|
|
|
- |
|
| Other |
|
|
1,559 |
|
|
|
952 |
|
| Income
tax (expense) benefit |
|
$ |
- |
|
|
$ |
- |
|
The Company reflects a tax benefit on its consolidated
statement of operations and comprehensive income (loss) in 2021 and 2020 of $113,415 and $134,728, respectively. These amounts are research
and development tax credits and are not considered income tax.
On March 27, 2020, the Coronavirus Aid, Relief, and
Economic Security Act (CARES Act) was enacted in response to the COVID-19 pandemic. The
CARES Act, among other things, permits NOL carryovers and carrybacks to offset 100% of taxable income for taxable years beginning before
2021. In addition, the CARES Act allows NOLs incurred in 2018, 2019, and 2020 to be carried back to each of the five preceding
taxable years to generate a refund of previously paid income taxes. The Company is currently evaluating the impact of the CARES Act,
but due to sustained losses, the NOL carryback provision of the CARES Act would not yield a benefit to us.
Deferred
income taxes reflect the net tax effects of temporary differences between the carrying amount of assets and liabilities for financial
reporting purposes and amounts used for income tax purposes. Significant components of the Company’s deferred tax assets and liabilities
consist of the following:
SCHEDULE
OF DEFERRED TAX ASSETS AND LIABILITIES
| |
|
June
30, 2021 |
|
|
June
30, 2020 |
|
| |
|
Year
Ended |
|
| |
|
June
30, 2021 |
|
|
June
30, 2020 |
|
| Deferred
tax assets |
|
|
|
|
|
|
|
|
| Warrant
Derivative Liability |
|
$ |
7,403 |
|
|
$ |
7,403 |
|
| Accrued
Expenses |
|
|
342,464 |
|
|
|
297,086 |
|
| Prepaid
Investor Services |
|
|
444,411 |
|
|
|
470,050 |
|
| Non-cash
interest |
|
|
687,529 |
|
|
|
596,004 |
|
| Intangibles
(Intellectual Property and Patent Cost) |
|
|
259,743 |
|
|
|
240,428 |
|
| Deferred
Rent |
|
|
4,262 |
|
|
|
1,969 |
|
| Formation
Expense |
|
|
6,815 |
|
|
|
7,208 |
|
| Net
Operating Loss CF |
|
|
8,546,920 |
|
|
|
7,438,911 |
|
| Foreign
Exchange Loss (OCI) |
|
|
(39,379 |
) |
|
|
(39,379 |
) |
| Revalue
of derivative liability |
|
|
439,958 |
|
|
|
438,239 |
|
| Stock
Based Compensation |
|
|
51,481 |
|
|
|
51,481 |
|
| Total
Deferred tax assets |
|
$ |
10,751,607 |
|
|
$ |
9,509,400 |
|
| |
|
|
|
|
|
|
|
|
| Deferred
tax liabilities |
|
|
|
|
|
|
|
|
| R&D |
|
$ |
(197,604 |
) |
|
$ |
(177,702 |
) |
| Gain
on extinguishment of debt |
|
|
(277,614 |
) |
|
|
266,987 |
|
| Capital
Raising Costs |
|
|
(321,291 |
) |
|
|
(255,614 |
) |
| Total
deferred tax liabilities |
|
$ |
(796,509 |
) |
|
$ |
(700,303 |
) |
| |
|
|
|
|
|
|
|
|
| Net
deferred tax assets (liabilities) |
|
$ |
9,955,098 |
|
|
$ |
8,809,097 |
|
| Valuation
allowance |
|
|
(9,955,098 |
) |
|
|
(8,809,097 |
) |
| Net
deferred tax assets (liabilities) |
|
$ |
- |
|
|
$ |
- |
|
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
At
June 30, 2021, the Company had U.S. net operating loss carry forwards of approximately $9,588,164
that may be offset against future
taxable income, subject to limitation under IRC Section 382. Of the approximately $9.6 million of net operating loss carryforwards,
$7.2 million will begin to expire in 2024 and the remaining $2.4 million will not expire. The Australian tax rate changed from 27.5%
in 2020 to 26% in 2021. At June 30, 2021, the Company had Australia net operating loss carry forwards of approximately $25,128,485
million which can be carried forward
without expiration. No
tax benefit has been reported in
the June 30, 2021 and 2020 consolidated financial statements due to the uncertainty surrounding the realizability of the benefit, based
on a more likely than not criteria and in consideration of available positive and negative evidence.
The
Company applied the “more-likely-than-not” recognition threshold to all tax positions taken or expected to be taken in a
tax return, which resulted in no unrecognized tax benefits as of June 30, 2021 and 2020, respectively.
Management
has determined that the realization of the net deferred tax asset is not assured and has created a valuation allowance for the entire
amount of such benefits.
The
Company follows ASC 740-10, which provides guidance for the recognition and measurement of certain tax positions in an enterprise’s
financial statements. Recognition involves a determination whether it is more likely than not that a tax position will be sustained upon
examination with the presumption that the tax position will be examined by the appropriate taxing authority having full knowledge of
all relevant information.
The
Company applied the “more-likely-than-not” recognition threshold to all tax positions taken or expected to be taken in a
tax return, which resulted in no unrecognized tax benefits as of June 30, 2021 and 2020, respectively.
The
Company’s policy is to record interest and penalties associated with unrecognized tax benefits as additional income taxes in the
consolidated statement of operations. As of June 30, 2021, the Company had no unrecognized tax benefits. There were no changes in the
Company’s unrecognized tax benefits during the years ended June 30, 2021 and 2020. The Company did not recognize any interest or
penalties during fiscal 2021 or 2020 related to unrecognized tax benefits.
The
income tax returns filed for the tax years from inception will be subject to examination by the relevant taxing authorities.
NOTE
8 – STOCKHOLDERS’ DEFICIT
Increase
in Authorized Shares of Common Stock and Reverse Stock Split
On
February 4, 2020 the Directors resolved to increase the Common Stock of the Company from 100,000,000 authorized shares to 1,000,000,000
authorized shares and believes that such number of authorized shares of Common Stock will be in the best interests of the Corporation
and its stockholders because the Board believes that the availability of more shares of Common Stock for issuance will allow the Corporation
greater flexibility in pursuing financing from investors, meeting business needs as they arise, taking advantage of favorable opportunities
and responding to a changing corporate environment. The Company filed the necessary documents with the U.S. Securities and Exchange Commission
on February 6, 2020 and with the amendment to the authorized shares being approved by the State of Delaware on March 13, 2020.
On
November 17, 2020, the Company effected a one-for-one thousand (1:1,000) reverse stock split of the Company’s issued and outstanding
shares of common stock (the “Reverse Stock Split”). Proportional adjustments for the Reverse Stock Split were made to the
Company’s outstanding stock options, warrants and equity incentive plans. All share and per-share data and amounts have been retroactively
adjusted as of the earliest period presented in the consolidated financial statements to reflect the Reverse Stock Split.
Preferred
Stock
The
total number of shares of preferred stock that the Company is authorized to issue is 1,500,005, $0.01 par value per share. These preferred
shares have no rights to dividends, profit sharing or liquidation preferences.
Of
the total preferred shares authorized, 500,000 have been designated as Series A Preferred Stock (“Series A Preferred Stock”),
pursuant to the Certificate of Designation filed with the Secretary of State of the State of Delaware on December 9, 2014. James Nathanielsz,
the Company’s Chief Executive Officer and Chief Financial Officer, beneficially owns all of the outstanding shares of Series A
Preferred Stock via North Horizon Pty Ltd., which entitles him, as a holder of Series A Preferred Stock, to vote on all matters submitted
or required to be submitted to a vote of the Company’s stockholders, except election and removal of directors, and each share of
Series A Preferred Stock entitles him to two votes per share of Series A Preferred Stock. North Horizon Pty Ltd. is a Nathanielsz Family
Trust. Mr. James Nathanielsz, the Chief Executive Officer, Chief Financial Officer and a director of our Company, has voting and investment
power over these shares. 500,000 shares of Series A Preferred Stock are issued and outstanding as of June 30, 2021 and 2020.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
Of
the total preferred shares authorized, pursuant to the Certificate of Designation filed with the Secretary of State of the State of Delaware
on June 16, 2015, up to five shares have been designated as Series B Preferred Stock (“Series B Preferred Stock”). Each holder
of outstanding shares of Series B Preferred Stock is entitled to voting power equivalent to the number of votes equal to the total number
of shares of common stock outstanding as of the record date for the determination of stockholders entitled to vote at each meeting of
stockholders of the Company and entitled to vote on all matters submitted or required to be submitted to a vote of the stockholders of
the Company. One share of Series B Preferred Stock is issued and outstanding as of June 30, 2021 and 2020. Mr. Nathanielsz directly beneficially
owns such one share of Series B Preferred Stock.
No
additional shares of Series A Preferred Stock or Series B Preferred Stock were issued during fiscal year 2021 and 2020.
Common
Stock
Shares
Issued for Cash
April
3, 2020 Security Purchase Agreement
On
April 3, 2020, the Company closed on a transaction related to a Securities Purchase Agreement (the “Securities Purchase Agreement”)
entered into on March 30, 2020, whereby an investor (the “Investor”) purchased from the Company, 7,500 units (the “Units”),
each consisting of (i) 1.5 shares of the Company’s common stock (the “Common Stock”), or pre-funded warrants (the “Prefunded
Warrants”) upon Investor’s election due to the 4.99% blocker provision as discussed below and (ii) 1.5 warrants to purchase
one share of Common Stock (“Series A Warrants”, and collectively with the Common Stock the “Units”). In addition
to the Units, the Investor was issued 63,750 warrants to purchase one share of Common Stock (the “Series B Warrants”) and
an additional 63,750 warrants to purchase one share of Common Stock, subject to a vesting schedule (the “Series C Warrants”
and, together with the Prefunded Warrants, the Series A Warrants, and the Series B Warrants, the “Warrants”).
The
aggregate purchase price for the Units, the Series A Warrants with exercise price of $200 per share, the Series B Warrants with exercise
price of $40 per share and the Series C Warrants with exercise price of $200 per share, of $450,000 was paid at closing (the “Purchase
Price”) or $60 per unit purchase price (see Warrants below). The Company received net proceeds of $424,990, net of offering cost
of $25,010.
The
Securities Purchase Agreement contains a blocker provision whereby the Investor or any of its affiliates would not beneficially own in
excess of 4.99% of the outstanding number of shares of Common Stock (“Beneficial Ownership Limitation”). As such, the Investor
may elect to purchase Prefunded Warrants equal to the same number of shares of Common Stock that the Company would have been issued.
Due
to the Beneficial Ownership Limitation, the 11,250 shares of Common Stock underlying the Units issuable at closing of the Securities
Purchase Agreement are comprised of 804 shares of restricted Common Stock and 10,446 Prefunded Warrants with exercise price of $1 (but
can be less than par value). The Prefunded Warrants shall be exercisable immediately and shall expire when exercised in full.
The
Securities Purchase Agreement contains such representations, warranties and covenants as are typical for a transaction of this nature.
Shares
issued for conversion of convertible debt
During
the year ended June 30, 2020, the Company issued 247,619 shares of its common stock at an average contractual conversion price of $7,
ranging from $2 to $910, as a result of the conversion of principal and interest in the aggregate amount of $1,814,336 underlying certain
outstanding principal amount and accrued interest of convertible notes converted during such period, including $15,000 of conversion
fees. The total recorded to equity was $2,125,174. Notes with principal amounts totaling $254,500 and accrued interest of $15,408 contained
bifurcated embedded conversion option derivatives. Accordingly, the fair market value of the shares issued was $565,746 resulting in
a loss on extinguishment at the time of conversion of $295,838 and $362,961 of derivative fair value was recorded as a gain on extinguishment
at the time of conversion. The Company reclassified $874,924 in put premiums to additional paid in capital following conversions during
the year ended June 30, 2020.
During
the year ended June 30, 2021, the Company issued an aggregate of 8,786,113 shares of its common stock at an average contractual conversion
price of $0.13, ranging from $0.03 to $2.00, as a result of the conversion of principal of $1,018,867, interest of $103,321 and conversion
fees $16,500 underlying certain outstanding convertible notes converted during such period. The total recorded to equity was $1,239,075
prior to the reversal of unissued shares.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
There
were 24,427 unissued shares which were considered issuable for accounting purposes during the first quarter of fiscal 2021 related to
conversion notices dated and received in September 2020. In November 2020, the Company was notified by the note holder of the cancellation
of these conversion notices and as such the Company reversed the effects of these transactions thereby increasing the principal balance
by $19,600, accrued interest of $416 and put premium by $11,785 and a corresponding decrease in equity of $31,801.
Converted
notes totaling principal amount of $95,000
and accrued interest of $3,000
contained bifurcated embedded conversion
option derivatives. Accordingly, the fair market value of the shares issued was $178,368
resulting in a loss on extinguishment
at the time of conversion of $80,368
and $130,975
of derivative fair value was recorded
as a gain on extinguishment at the time of conversion.
The Company reclassified $590,504
to additional paid in capital following conversions of notes accounted for as stock settled debt during the year ended June 30,
2021.
The
Company has 197,308,116 shares of its common stock reserved for future issuances based on lender reserve requirements pursuant to underlying
financing agreements at June 30, 2021.
Shares
issued for services
On
July 19, 2019, the Company entered into an agreement with a certain consultant to provide services over a two-month period beginning
July 1, 2019 and ending September 1, 2019 in exchange for 20 shares of the Company’s common stock. On July 19, 2019, the Company
issued the 20 shares of the Company’s common stock valued at $1,990 per share; being the closing price of the stock on the date
of the agreement, to such consultant, or $39,800, which will be amortized over the term of the agreement. The Company recorded $39,800
of consulting expense with respect to such shares of its common stock during the year ended June 30, 2020.
Between
February 3, 2020 and June 26, 2020, the Company issued an aggregate of 8,709 shares of the Company’s common stock to a consultant
for services rendered pursuant to an engagement agreement dated on September 10, 2019 which agreement was later amended in February 2020.
Between February 3, 2020 and June 26, 2020, the Company issued an aggregate of 8,709 shares of the Company’s common stock valued
at an average price of $8 per share; being the closing price of the stock on the date of the agreement, to such consultant, or $73,842.
The Company recorded $73,842 of consulting expense with respect to such shares of its common stock during the year ended June 30, 2020.
On
March 22, 2021, the Company issued an aggregate of 225,037 shares of the Company’s common stock to a consultant for services rendered
from January 1, 2021 to March 22, 2021. The Company issued 225,037 shares of the Company’s common stock valued at $0.30 per share,
being the closing price of the stock on the date of grant to such consultant, or $67,511. The Company recorded $67,511 of consulting
expense with respect to such shares of its common stock during the year ended June 30, 2021.
Between
March 2021 and June 2021, the Company issued an aggregate of 580,609 shares of the Company’s common stock to a consultant for services
rendered from April 1, 2021 to June 30, 2021. The Company issued 580,609 shares of the Company’s common stock valued at $0.10 per
share, being the closing price of the stock on the date of grant to such consultant, or $58,061. The Company recorded $58,061 of consulting
expense with respect to such shares of its common stock during the year ended June 30, 2021.
Shares
issued for exercise of warrants
During
the year ended June 30, 2021, the Company received aggregate gross proceeds of $776,044
from the exercise of 10,445
prefunded warrants and 19,375
Series B Warrants resulting in
the issuance of 29,820
shares of common stock.
Additionally,
during 2021 the Company issued 4,199,979
shares of common stock from the
alternate cashless exercise of 20 Series A and 1 Series C warrants. The Company recognized the value of the effect of a down round
feature in such warrants when triggered. Upon the occurrence of the triggering event that resulted in a reduction of the strike price,
the Company measured the value of the effect of the feature as the difference between the fair value of the warrants without the down
round feature or before the strike price reduction and the fair value of the warrants with a strike price corresponding to the reduced
strike price upon the down round feature being triggered. Accordingly, the Company recognized deemed dividend of $391,749
and a corresponding reduction
of income available to common stockholders upon the alternate
cashless exercise of these warrants.
Restricted
Stock Units
Pursuant
to employment agreements dated in May 2019, the Company granted an aggregate of 78
and 39
restricted stock unit to the Company’s
Chief Executive Officer and Chief Scientific Officer, respectively. The total 117
restricted stock units are subject
to vesting terms as defined in the employment agreements. The 117 restricted stock units were valued at the fair value of $4,250
per unit or $497,240
based on the quoted trading price
on the date of grant. During the year ended June 30, 2021 and 2020, the Company recognized stock-based compensation of $0
and $217,543,
respectively, related to vested restricted stock units. There were $248,620
unrecognized restricted stock units
expense as of June 30, 2021. There are 59
unvested restricted stock units
which are subject to various performance conditions which have not yet been met and such restricted stock
units have not yet vested as of June 30, 2021 and 2020 to which the $248,620 relates.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
Options
A
summary of the Company’s option activity during the years ended June 30, 2021 and 2020 is presented below:
SCHEDULE
OF STOCK OPTION ACTIVITY
| | |
Number of | | |
Weighted Average | |
| | |
Options | | |
Price Per Share | |
| Outstanding at June 30, 2019 | |
| 60 | | |
$ | 76,370 | |
| Issued | |
| - | | |
| - | |
| Exercised | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Outstanding at June 30, 2020 | |
| 60 | | |
$ | 76,370 | |
| Issued | |
| - | | |
| - | |
| Exercised | |
| - | | |
| - | |
| Expired | |
| (1 | ) | |
| 3,750,000 | |
| Outstanding at June 30, 2021 | |
| 59 | | |
$ | 13,730 | |
| | |
| | | |
| | |
| Exercisable at June 30, 2021 | |
| 40 | | |
$ | 18,193 | |
| Outstanding and Exercisable: | |
| | | |
| | |
| | |
| | | |
| | |
| Weighted average remaining contractual term | |
| 7.86 | | |
| | |
| Weighted average fair value of options granted during the period | |
$ | - | | |
| | |
| Aggregate intrinsic value | |
$ | - | | |
| | |
On
the Effective Date, the Company’s board of directors approved and adopted the Company’s 2019 Equity Incentive Plan (the “2019
Plan”), which reserves a total of 234 shares of the Company’s common stock for issuance under the 2019 Plan. Incentive awards
authorized under the 2019 Plan include, but are not limited to, incentive stock options, non-qualified stock options, restricted stock
awards and restricted stock units.
During
the year ended June 30, 2021 and 2020, the Company recognized stock-based compensation of $82,872
and $82,873
related to vested stock options.
There was $72,514
of unvested stock options expense
as of June 30, 2021 that will be recognized over a remaining vesting period of 0.87 year.
No
stock options were granted during the years ended June 30, 2021 and 2020.
Warrants
The
following table summarizes warrant activity for the years ended June 30, 2021 and 2020:
SCHEDULE
OF WARRANT ACTIVITY
| | |
Number of | | |
Weighted Average | |
| | |
Warrants | | |
Price Per Share | |
| Outstanding at June 30, 2019 | |
| - | | |
$ | - | |
| Issued | |
| 151,170 | | |
| 150.00 | |
| Exercised | |
| - | | |
| - | |
| Forfeited | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Outstanding at June 30, 2020 | |
| 151,170 | | |
$ | 150.00 | |
| Issued | |
| - | | |
| - | |
| Exercised | |
| (29,841 | ) | |
| 26.15 | |
| Forfeited | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Outstanding at June 30, 2021 | |
| 121,329 | | |
$ | 179.63 | |
| | |
| | | |
| | |
| Exercisable at June 30, 2021 | |
| 76,955 | | |
$ | 283.21 | |
| Outstanding and Exercisable: | |
| | | |
| | |
| | |
| | | |
| | |
| Weighted average remaining contractual term | |
| 1.77 | | |
| | |
| Aggregate intrinsic value | |
$ | - | | |
| | |
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
In
connection with the issuance of the August 2019 Auctus Note, the Company issued common stock purchase warrants to Auctus to purchase
450 shares of the Company’s common stock (the “First Warrant”) as a commitment fee upon the terms and subject to the
limitations and conditions set forth in such First Warrant at an “Exercise Price” of $2,250. In connection with the issuance
of the Note, the Company shall issue a common stock purchase warrant to Buyer to purchase 300 shares of the Company’s common stock
(the “Second Warrant”) as a commitment fee upon the terms and subject to the limitations and conditions set forth in such
Second Warrant at an “Exercise Price” of $3,330. In connection with the issuance of the Note, the Company shall issue a common
stock purchase warrant to Buyer to purchase 225 shares of the Company’s common stock (the “Third Warrant”) as a commitment
fee upon the terms and subject to the limitations and conditions set forth in such Third Warrant at an “Exercise Price” of
$4,500. The First Warrant, Second Warrant, and Third Warrant shall collectively be referred as the “Warrants”. The Warrants
have an “Exercise Period” of five years from the date of issuance being August 30, 2019 (see Note 6).
On
September 10, 2019, the Company entered into an agreement with a certain consultant to provide services over a three-month period beginning
September 10, 2019 and ending December 10, 2019 in exchange for 1,000 warrants to purchase the Company’s common stock at $2,000
per share with an expiry date of September 10, 2022. The Fair Market Value of the warrants was $984,810 on the date of grant as calculated
under the Black Scholes Option Pricing model. The Company recorded $984,810 of share-based compensation expenses with respect to the
grant of such warrants during the year ended June 30, 2020.
In
connection with the issuance of shares on April 3, 2020 as discussed above, the Company closed on a transaction related to a Securities
Purchase Agreement (the “Securities Purchase Agreement”) entered into on March 30, 2020, whereby an investor purchased from
the Company, 7,500 units, each consisting of (i) 1.5 shares of the Company’s common stock, or pre-funded warrants upon Investor’s
election due to the 4.99% blocker provision and (ii) 1.5 warrants to purchase one share of Common Stock (“Series A Warrants”,
and collectively with the Common Stock the “Units”). In addition to the Units, the Investor was issued 63,750 warrants to
purchase one share of Common Stock (the “Series B Warrants”) and an additional 63,750 warrants to purchase one share of Common
Stock, subject to a vesting schedule (the “Series C Warrants” and, together with the Prefunded Warrants, the Series A Warrants,
and the Series B Warrants, the “Warrants”).
Due
to the Beneficial Ownership Limitation, the Company granted 10,445 Prefunded Warrants with exercise price of $0.10 (but can be less than
par value). The Prefunded Warrants shall be exercisable immediately and shall expire when exercised in full.
Series
A Warrants
Pursuant
to the Securities Purchase Agreement entered into March 20, 2020 as discussed above, the Investor purchased Series A Warrants to purchase
up to 11,250
shares of Common Stock, subject
to adjustment as provided therein. The Series A Warrants have a cash exercise price of $200
per share and are immediately exercisable
and expire in 3
years. The Series A Warrants contain
a provision for cashless exercise in the event there is no effective registration statement registering the shares underlying the Series
A Warrants calculated based on the difference between the exercise price of the Series A Warrant and the trading price of the stock (the
“Cashless Exercise”).Additionally, the Series A Warrants contain a provision for a cashless conversion at the Holder’s
option should the trading price of the Common Stock fall below $200 per share calculated based on the difference between the exercise
price of the Series A Warrant and 70% of the Market Price, as defined therein (the” Alternate Cashless Exercise”). See
above “Shares issued for exercise of warrants” for discussion of deemed dividend related to alternate cashless exercise.
Series
B Warrants
Pursuant
to the Securities Purchase Agreement entered into March 20, 2020 as discussed above, the Investor purchased Series B Warrants to purchase
up to 63,750 shares of Common Stock, subject to adjustment as provided therein; provided, however, commencing on the 90th day following
the effective date, the Company may reduce the number of Warrant Shares issuable upon exercise thereof by 37,500 upon 10 Trading Days’
prior written notice to the Holder provided that the Company issues to the Holder 3,750 shares of Common Stock (or, at the election of
the Holder, an equivalent number of pre-funded warrants) and Series A Warrants to purchase up to 3,750 shares of Common Stock, which
shares shall be issued pursuant to a registration statement without restrictions on resale. The Series B Warrants have a cash exercise
price of $40 per share and expire in 3 years. The Series B Warrants contain a provision for Cashless Exercise.
Series
C Warrants
Pursuant
to the Securities Purchase Agreement entered into March 20, 2020 as discussed above, the Investor purchased Series C Warrants to purchase
up to 63,750 shares of Common Stock, subject to adjustment as provided therein and expire in 3 years. The Series C Warrants have a cash
exercise price of $200 per share, subject to a vesting schedule, which is based on such Holder’s exercise of the Series B Warrants
(warrants shall be exercisable ratably upon exercise of Series B Warrants). The Series C Warrants contain provisions for Cashless Exercise
and Alternate Cashless Exercise.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
Exercise
of Warrants
During
the year ended June 30, 2021, the Company received aggregate gross proceeds of $776,044 from the exercise of 10,445 prefunded warrants
and 19,375 Series B Warrants resulting in the issuance of 29,820 shares of common stock. Additionally, the Company issued 4,199,979 shares
of common stock from the alternate cashless exercise of 20 Series A and 1 Series C warrants.
NOTE
9 – COMMITMENTS AND CONTINGENCIES
Legal
Matters
On
September 26, 2019, a complaint was filed against the Company with Supreme Court of the State of New York, County of New York, by Foley
Shechter Ablovatskiy LLP (“Foley Shechter”), our former counsel, seeking $151,031
in professional fees allegedly
owed, in addition to interest and costs of suit. The Company filed an answer, together with affirmative defenses and counterclaims. Certain
amounts related to this claim were included in accounts payable and accrued expenses in the accompanying consolidated financial statements
at June 30, 2020. On March 22, 2021, the Company entered into a settlement agreement with Foley Shechter whereby both parties
agreed to settle all claims for professional fees owed for a total of $51,032.
The Company paid the settlement amount of $51,032
on March 22, 2021. Prior to the
settlement agreement, the Company recorded total accounts payable and accrued expenses $142,660.
Accordingly, the Company recognized gain from settlement of debt of $92,556
during the year ended June
30, 2021.
Regal
Consulting, LLC (“Regal”) initiated litigation against the Company in Clark County District Court, Nevada. Regal was demanding
approximately $400,000 and 60 shares of the Company’s common stock as payment for services that Regal purports to have performed.
Regal additionally claimed that $106,500 remained due on a Convertible Note executed by the Company in May of 2017 and asserted that
it was owed in excess of $100,000 in penalties in connection with the Company’s refusal to honor certain Conversion Notices. The
Company filed an Answer and Counterclaim, denying liability and alleging that Regal procured by fraud the Company’s execution of
various consulting agreements and additionally failed to provide the consulting services contemplated by said agreements. On December
23, 2020, the parties mediated their dispute and negotiated a settlement agreement. On March 15, 2021, the Company entered into a Settlement
and Mutual Release Agreement with Regal whereby both parties agreed to settle all claims and liabilities under the August 10, 2017 Convertible
note (see Note 6) for a total of $100,000. All other terms of the August 10, 2017 Convertible Note shall remain in full force and effect.
Both parties agree that all future penalties under this convertible note are waived unless the Company fails to authorize the issuance
of the requested shares upon conversion. The Company has the right to pay off the balance of any remaining amounts dues under this convertible
note in cash at any time 61 days after March 15, 2021. Prior to the Settlement Agreement, the Company recorded total liabilities $56,762
consisting of remaining principal amount of $8,500, accrued interest of $23,262 and accrued expenses of $25,000. Accordingly, the Company
recognized loss from settlement of debt of $43,238 during the year ended June 30, 2021.
IRS
Liability
As
part of its requirement for having a foreign operating subsidiary, the Company’s parent U.S. entity is required to file an informational
Form 5471 to the Internal Revenue Service (the “IRS”), which is a form that explains the nature of the relationship between
the foreign subsidiary and the parent company. From 2012 through the 2014, the Company did not file this form in a timely manner. As
a result of the non-timely filings, the Company incurred a penalty from the IRS in the amount of $10,000
per year, or $30,000
in total, plus accrued interest,
such penalty and interest having been accrued and is included in the accrued expenses and other payable figure in the June
30, 2021 and 2020 consolidated balance sheet. The Company recorded the penalties for all three years during the year ended June 30,
2018. The Company is current on all subsequent filings. The Company’s tax advisor is awaiting a response from the IRS on this matter.
Operating
Agreements
In
November 2009, the Company entered into a commercialization agreement with the University of Bath (UK) (the “University”)
whereby the Company and the University co-owned the intellectual property relating to the Company’s pro-enzyme formulations. In
June 2012, the Company and the University entered into an assignment and amendment whereby the Company assumed full ownership of the
intellectual property while agreeing to pay royalties of 2% of net revenues to the University. Additionally, the Company agreed to pay
5% of each and every license agreement subscribed for. The contract is cancellable at any time by either party. To date, no amounts are
owed under the agreement.
Operating
Leases
On
May 5, 2016, the Company entered into a new five-year
operating lease agreement with a Horizon Pty Ltd., a related party, of which Mr. Nathanielsz, our CEO, CFO and a director, and his
wife are owners and directors, with monthly rent currently at $3,606 AUD
or $2,469 USD,
inclusive of GST (See Note 10 – Related Party Transactions). The
initial rental amount was $3,000 AUD and subject to 3% yearly escalation. In
adopting ASC Topic 842, Leases (Topic 842), the Company has elected the ‘package of practical expedients’, which permit
it not to reassess under the new standard its prior conclusions about lease identification, lease classification and initial direct
costs. In addition, the Company elected not to apply ASC Topic 842 to arrangements with lease terms of 12 month or less. On July 1,
2019, upon adoption of ASC Topic 842, the Company recorded right-of-use assets $48,662 and
total lease liabilities of $48,662 based
on an incremental borrowing rate of 6%. Such lease expired in May 2021 and was renewed for another one-year term from May 2021 to May 2022. The Company is currently obligated to pay $3,606
AUD or $2,431
USD (depending on exchange rate), inclusive of tax, in rent per month.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
ROU
is summarized below:
SCHEDULE OF RIGHT USE OF ASSET
| |
|
June
30, 2021 |
|
|
June
30, 2020 |
|
| Office
lease (24 months) |
|
$ |
48,662 |
|
|
$ |
48,662 |
|
| Less
accumulated amortization |
|
|
(48,662 |
) |
|
|
(26,980 |
) |
| Right-of-use
asset, net |
|
$ |
- |
|
|
$ |
21,682 |
|
Operating
Lease liabilities are summarized below:
SCHEDULE
OF OPERATING LEASE LIABILITY
| |
|
June
30, 2021 |
|
|
June
30, 2020 |
|
| Office
lease |
|
$ |
48,662 |
|
|
$ |
48,662 |
|
| Reduction
of lease liability |
|
|
(48,662 |
) |
|
|
(23,590 |
) |
| Long
term portion of lease liability |
|
$ |
- |
|
|
$ |
25,072 |
|
Collaboration
Agreement
On
September 13, 2018, the Company entered into a two-year collaboration agreement with the University of Jaén (the “University”)
to provide certain research services to the Company. In consideration of such services, the Company agreed to pay the University approximately
52,000 Euros ($59,508 USD) in year one and a maximum of 40,000 Euros ($45,775 USD) in year two. The Company paid 31,754 Euros ($36,117
USD) in 2019 and has accrued 28,493 Euros ($24,043 USD) as of June 30, 2021. Additionally, in exchange for full ownership of the intellectual
property the Company agreed to pay royalties of 2% of net revenues to the University. On October 1, 2020, the Company entered into another
two-year collaboration agreement with the University of Jaén to provide certain research services to the Company. In consideration
of such services, the Company agreed to pay the University approximately 30,000 Euros ($35,145 USD) which shall be paid in four installment
payment of 5,000 Euros in November 2020, 5,000 Euros ($5,858) in March 2021, 10,000 Euros ($11,715) in December 2021 and 10,000 Euros
($11,715) in September 2022. Additionally, the University shall hire and train a doctoral student for this project and as such the Company
shall pay the University 25,837 Euros ($30,268 USD). In exchange for full ownership of the intellectual property the Company agreed to
pay royalties of 2% of net revenues to the University.
NOTE
10 – RELATED PARTY TRANSACTIONS
Since
its inception, the Company has conducted transactions with its directors and entities related to such directors. These transactions have
included the following:
As
of June 30, 2021 and 2020, the Company owed its former director a total of $55,500 and $50,993, respectively, for money loaned to the
Company throughout the years. The total loans balance owed at June 30, 2021 and 2020 is not interest bearing (See Note 5 – Loans
and Notes Payable).
As
of June 30, 2021 and 2020, the Company owed its former director a total of $33,347 and $30,639, respectively, related to expenses paid
on behalf of the Company related to corporate startup costs and intellectual property (See Note 4 – Due to Former Director –
Related Party).
Effective
May 5, 2016, the Company entered into an agreement for the lease of its principal executive offices with North Horizon Pty Ltd., a related
party, of which Mr. Nathanielsz, our CEO, CFO and a director, and his wife are owners and directors. The lease has a five-year
term and provides for annual rental payments of $39,600
AUD or $28,325
USD, which includes $3,600
AUD or $2,575
USD of goods and service tax for
total payments of $198,000
AUD or $141,629
USD during the term of the lease.
As of June 30, 2021, total rent payable of $86,129
AUD ($64,597
USD) is included in accrued expenses
in the accompanying consolidated balance sheet. Such lease expired in May 2021 and was renewed for another one-year term from May 2021 to May 2022 (See Note 9 – Commitments and Contingencies).
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
Employment
and Services Agreements with Management
The
Company and Mr. Nathanielsz entered into an employment agreement as of February 25, 2015 (the “Nathanielsz Employment Agreement”)
setting forth the terms and conditions of Mr. Nathanielsz employment as the Company’s President and Chief Executive Officer. The
Nathanielsz Employment Agreement was scheduled to expire on February 25, 2019; however, the term of the Nathanielsz Employment Agreement
automatically renews for successive one-year periods unless either party provides 30 days’ prior written notice of its intent not
to renew. The Nathanielsz Employment Agreement continues in effect as of June 30, 2021 as amended May 14, 2019 (see below). The Nathanielsz
Employment Agreement provides Mr. Nathanielsz with a base salary of $25,000 AUD per month ($300,000 AUD annually or $205,680 USD) and
a monthly contribution to Mr. Nathanielsz’s pension equal to 9.5% of his monthly salary. Mr. Nathanielsz has the ability to convert
any accrued but unpaid salary into common stock at the end of each fiscal year at a conversion price to be determined by Mr. Nathanielsz
and the Company, which will in no event be lower than par value or higher than the closing bid price on the date of conversion. Pursuant
to the Nathanielsz Employment Agreement, Mr. Nathanielsz is entitled to an annual discretionary bonus in an amount up to 200% of his
annual base salary, which bonus shall be determined by the Company’s board of directors based upon the performance of the Company.
On March 16, 2018, the Company’s board of directors approved an increase of Mr. Nathanielsz’s annual base salary from $300,000
AUD ($205,680 USD) to $400,000 AUD ($274,240 USD), effective February 2018.
Mr.
Nathanielsz’s wife, Sylvia Nathanielsz, is and has been a non-executive part-time employee of the Company since October 2015. Effective
February 1, 2018, Mrs. Nathanielsz receives an annual salary of $120,000 AUD ($80,904 USD) and is entitled to customary benefits.
Pursuant
to a February 25, 2016 board resolution, James Nathanielsz shall be paid $4,481 AUD ($3,205 USD), on a monthly basis for the purpose
of acquiring and maintaining an automobile. For the year ended June 30, 2020, a total of $44,918 AUD ($30,284 USD) in payments have been
made with respect to Mr. Nathanielsz’s car allowance. For the year ended June 30, 2021, a total of $46,135 AUD ($34,476 USD) in
payments have been made with respect to Mr. Nathanielsz’s car allowance.
Pursuant
to the approval of the Company’s board of directors, on May 14, 2019, Mr. Nathanielsz was granted a $460,000
AUD ($315,376
USD) bonus for accomplishments
achieved while serving as the Company’s Chief Executive Officer during the fiscal year ended June 30, 2019 with $200,000
AUD ($137,120
USD) of such bonus payable by the
Corporation to the CEO throughout the Corporation’s 2019 fiscal year as the Corporation’s cash resources allow, with the
remaining $260,000
AUD ($178,256
USD) of such bonus to be deferred
by the CEO until a future date when the Corporation’s cash resources allow for such payment, as agreed to by the CEO. A total of
$90,000
AUD ($64,377
USD) in payments were made in the
year ended June 30, 2019. On July 13, 2020, the Board approved a bonus of $240,000
AUD being equal to 60%
of Mr. Nathanielsz base salary
which was accrued as of June 30, 2020. A total of $202,620
AUD ($136,606
USD) in payments were made against
the bonuses during the year ended June 30, 2020 which resulted to a remaining balance of $407,380
AUD ($280,726
USD) bonus payable as of June 30,
2020. On August 12, 2021, the Board approved a bonus of $177,840
USD. A total of $221,890
AUD ($166,418
USD) in payments were made against
the bonuses during the year ended June 30, 2021 resulting in a remaining balance of $422,610
AUD ($316,957
USD) bonus payable as of June 30,
2021 which is included in accrued expenses in the accompanying consolidated balance sheet. On August 12, 2021, pursuant to the Cancellation
Agreement, Mr. Nathanielsz agreed to cancel $177,840 of the bonus payable in exchange for 5,928,000
shares of the common stock of the
Company (see Note 13).
Amended
and Restated Employment Agreement - On May 14, 2019 (the “Effective Date”), the Company entered into an Amended and Restated
Employment Agreement (the “Employment Agreement”) with James Nathanielsz, the Company’s Chief Executive Officer, Chairman,
acting Chief Financial Officer and a director, for a term of three years, subject to automatic one-year renewals, at an annual salary
of $400,000 AUD. Pursuant to the Employment Agreement, Mr. Nathanielsz was granted options to purchase 39 shares of the Company’s
common stock (the “Nathanielsz Options”), with an exercise price per share of $4,675 (110% of the closing market price of
the Company’s common stock on May 14, 2019 (or $4,250), the date of approval of such grant by the Company’s board of directors),
(ii) 39 restricted stock units of the Company (the “Initial Nathanielsz RSUs”), and (iii) an additional 39 restricted stock
units of the Company (the “Additional Nathanielsz RSUs”). Such options and restricted stock units were granted pursuant to
the 2019 Plan approved by the Company’s board of directors on the Effective Date. The Nathanielsz Options have a term of 10 years
from the date of grant. 1/3rd of the Nathanielsz Options shall vest every successive one-year anniversary following the Effective Date,
provided, that on each such vesting date Mr. Nathanielsz is employed by the Company and subject to the other provisions of the Employment
Agreement. The Initial Nathanielsz RSUs shall vest on the one-year anniversary of the Effective Date, subject to Mr. Nathanielsz’s
continued employment with the Company through such vesting date. The Additional Nathanielsz RSUs will vest as follows, subject to Mr.
Nathanielsz’s continued employment with the Company through the applicable vesting date: (i) 7.80 of the Additional Nathanielsz
RSUs shall vest upon the Company submitting Clinical Trial Application (the “CTA”) for PRP, the Company’s lead product
candidate (“PRP”), for a First-In-Human study for PRP (the “Study”) in an applicable jurisdiction to be selected
by the Company, (ii) 7.80 of the Additional Nathanielsz RSUs shall vest upon the CTA being approved in an applicable jurisdiction, (iii)
7.80 of the Additional RSUs shall vest upon the Company completing an equity financing in the amount of at least $4,000,000 in gross
proceeds, (iv) 7.80 of the Additional Nathanielsz RSUs shall vest upon the shares of the Company’s Common Stock being listed on
a senior stock exchange (NYSE, NYSEMKT or NASDAQ), and (v) the remaining 7.80 of the Additional Nathanielsz RSUs shall vest upon the
Company enrolling its first patient in the Study. Each vested restricted stock unit shall be settled by delivery to Mr. Nathanielsz of
one share of the Company’s common stock and/or the fair market value of one share of common stock in cash, at the sole discretion
of the Company’s board of directors and subject to the 2019 Plan, on the first to occur of: (i) the date of a Change of Control
(as defined in the Employment Agreement), (ii) the date that is ten business days following the vesting of such restricted stock unit,
(iii) the date of Mr. Nathanielsz’s death or Disability (as defined in the Employment Agreement), and (iv) Mr. Nathanielsz’s
employment being terminated either by the Company without Cause or by Mr. Nathanielsz for Good Reason (each as defined in the Employment
Agreement). In the event of a Change of Control, any unvested portion of the Nathanielsz Options and such restricted stock units shall
vest immediately prior to such event. The 39 vested restricted stock unit are considered issuable as of June 30, 2021.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
Amended
and Restated Services Agreement - On May 14, 2019, the Company also entered into an Amended and Restated Services Agreement (the
“Services Agreement”) with Dr. Kenyon, the Company’s Chief Scientific Officer and a director, for a term of three years,
subject to automatic one-year renewals, at an annual salary of $54,000
AUD. In connection with the execution of the Services Agreement, Dr. Kenyon was designated as an executive officer of the Company
and assumed a more active executive role with the Company. Pursuant to the Services Agreement, Dr. Kenyon was granted options to
purchase 20
shares of the Company’s common stock (the “Kenyon Options”), with an exercise price per share of $4,250
(100%
of the closing market price of the Company’s common stock on May 14, 2019, the date of approval of such grant by the
Company’s board of directors), (ii) 20
restricted stock units of the Company (the “Initial Kenyon RSUs”), and (iii) an additional 20
restricted stock units of the Company (the “Additional Kenyon RSUs”). Such options and restricted stock units were
granted pursuant to the 2019 Plan approved by the Company’s board of directors on the Effective Date. The
Kenyon Options have a term of 10
years from the date of grant. 1/3rd of the Kenyon Options shall vest every successive one-year anniversary following the
Effective Date, provided, that on each such vesting date Dr. Kenyon is employed by the Company and subject to the other provisions
of the Services Agreement. The Initial Kenyon RSUs shall vest on the one-year anniversary of the Effective Date, subject to Dr.
Kenyon’s continued employment with the Company through such vesting date. The Additional Kenyon RSUs will vest as follows,
subject to Dr. Kenyon’s continued employment with the Company through the applicable vesting date: (i) 5
of the Additional Kenyon RSUs shall vest upon the Company submitting the CTA for PRP for the Study in an applicable jurisdiction to
be selected by the Company, (ii) 5 of the Additional Kenyon RSUs shall vest upon the Company completing an equity financing in the
amount of at least $4,000,000
in gross proceeds, (iii) 5 of the Additional Kenyon RSUs shall vest upon the shares of the Company’s Common Stock being
listed on a senior stock exchange (NYSE, NYSEMKT or NASDAQ), and (iv) the remaining 5 of the Additional Kenyon RSUs shall vest upon
the Company enrolling its first patient in the Study. Each vested Kenyon RSU shall be settled by delivery to Mr. Kenyon of one share
of the Company’s common stock and/or the fair market value of one share of common stock in cash, at the sole discretion of the
Company’s board of directors and subject to the Plan, on the first to occur of: (i) the date of a Change of Control (as
defined in the Services Agreement), (ii) the date that is ten business days following the vesting of such Kenyon RSU, (iii) the date
of Dr. Kenyon’s death or Disability (as defined in the Services Agreement), and (iv) Dr. Kenyon’s employment being
terminated either by the Company without Cause or by Dr. Kenyon for Good Reason (as defined in the Services Agreement). In the event
of a Change of Control (as defined in the Services Agreement), 50% of any unvested portion of the Kenyon Options and the Kenyon RSUs
shall vest immediately prior to such event. As of June 30, 2021, total accrued salaries of $135,000
AUD ($101,250 USD) is included
in accrued expenses in the accompanying consolidated balance sheet. The 20
vested restricted stock unit are considered issuable as of June 30, 2021. On August 12, 2021, pursuant to the Cancellation
Agreement, Mr. Kenyon agreed to cancel accrued salaries of $102,600
in exchange for 3,420,000
shares of the common stock of the Company (see Note 13).
Intercompany
Loans
All
Intercompany loans were made by the parent to the subsidiary, Propanc PTY LTD, which have not been repaid as of June 30, 2021. Effective
fiscal year 2021, the parent company determined that intercompany loans will not be repaid in the foreseeable future and thus, per ASC
830-20-35-3, gains and losses from measuring the intercompany balances are recorded within cumulative translation adjustment, a component
of other comprehensive income. Prior to July 1, 2020, the Company recorded the foreign currency transaction gains and losses from measuring
the intercompany balances as a component of other income (expenses) as reflected in the consolidated statements of operations.
NOTE
11 – CONCENTRATIONS AND RISKS
Concentration
of Credit Risk
The
Company maintains its cash in banks and financial institutions in Australia. Bank deposits in Australian banks are uninsured. The Company
has not experienced any losses in such accounts through June 30, 2021.
In fiscal year 2020,
the
Company currently primarily relied on funding from three convertible debt lenders. Proceeds received
during the year ended June 30, 2020 from each of the three lenders were $285,000,
$505,000,
and $227,000,
respectively, which represents approximately 19%,
34%
and 15%,
respectively of total proceeds received by the Company during fiscal year 2020.
In fiscal year 2021,
the Company primarily relied on funding from two convertible debt lenders and received net proceeds after deductions of $16,000
for original issue discounts and debt issue costs during the year ended June 30, 2021 from each of the two lenders of $125,000 and
$200,000,
respectively, which represents approximately 39%,
and 61%, respectively
of total proceeds received by the Company during fiscal year 2021.
Receivable
Concentration
As
of June 30, 2021 and 2020, the Company’s receivables were 100% related to reimbursements on GST taxes paid.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
Patent
and Patent Concentration
The
Company has filed multiple patent applications relating to its lead product, PRP. The Company’s lead patent application has been
granted and remains in force in the United States, Belgium, Czech Republic, Denmark, France, Germany, Ireland, Italy, Netherlands, Portugal,
Spain, Sweden, Switzerland, Liechtenstein, Turkey, United Kingdom, Australia, China, Japan, Indonesia, Israel, New Zealand, Singapore,
Malaysia, South Africa, Mexico, Republic of Korea, India and Brazil. In Canada, the patent application remains
under examination.
In
2016 and early 2017, we filed other patent applications. Three applications were filed under the Patent Cooperation Treaty (the “PCT”).
The PCT assists applicants in seeking patent protection by filing one international patent application under the PCT, applicants can
simultaneously seek protection for an invention in over 150 countries. Once filed, the application is placed under the control of the
national or regional patent offices, as applicable, in what is called the national phase. One of the PCT applications filed in November
2016, entered national phase in July 2018 and another PCT application is currently entering national phase in August 2018. A third PCT
application entered the national phase in October 2018.
In
July 2020, a world first patent was granted in Australia for the cancer treatment method patent family. Presently, there are 31
granted patents and 34 patents under examination in key global jurisdictions relating to the use of proenzymes against solid tumors,
covering the lead product candidate PRP.
Further
patent applications are expected to be filed to capture and protect additional patentable subject matter based on the Company’s
field of technology relating to pharmaceutical compositions of proenzymes for treating cancer.
Foreign
Operations
As
of June 30, 2021 and 2020, the Company’s operations are based in Camberwell, Australia, however the majority of research and development
is being conducted in the European Union.
On
July 22, 2016, the Company formed a wholly owned subsidiary, Propanc (UK) Limited under the laws of England and Wales for the purpose
of submitting an orphan drug application with the European Medicines Agency as a small and medium-sized enterprise. As of June 30, 2021
and 2020, there has been no activity within this entity.
NOTE
12 - DERIVATIVE FINANCIAL INSTRUMENTS AND FAIR VALUE MEASUREMENTS
Derivative
Financial Instruments:
The
Company applies the provisions of ASC 815-40, Contracts in Entity’s Own Equity, under which convertible instruments and
warrants, which contain terms that protect holders from declines in the stock price (reset provisions), may not be exempt from derivative
accounting treatment. As a result, warrants and embedded conversion options in convertible debt are recorded as a liability and are revalued
at fair value at each reporting date. If the fair value of the warrants exceeds the face value of the related debt, the excess is recorded
as change in fair value in operations on the issuance date. The Company had $80,000 (1 note) and $126,500 (3 notes) of convertible debt,
which is treated as derivative instruments outstanding at June 30, 2021 and 2020 respectively.
The
Company calculates the estimated fair values of the liabilities for derivative instruments using the Binomial Trees Method. The closing
price of the Company’s common stock at June 30, 2021, the last trading day of the fiscal year ended June 30, 2021, was $0.0517.
Volatility, expected remaining term and risk-free interest rates used to estimate the fair value of derivative liabilities at June 30,
2021 and 2020 are indicated in the table that follows. The expected term is equal to the remaining term of the warrants or convertible
instruments and the risk free rate is based upon rates for treasury securities with the same term.
SCHEDULE OF FAIR VALUE MEASUREMENTS, RECURRING AND NONRECURRING, VALUATION TECHNIQUES
Convertible
Debt
| |
|
Initial Valuations during the year ended June 30, 2021 |
|
Initial
Valuations
during
the
year ended June 30, 2020 |
|
|
June
30, 2021 |
|
|
June
30, 2020 |
|
| Volatility |
|
N/A |
|
227-279 |
% |
|
|
222 |
% |
|
|
264 |
% |
| Expected
remaining term |
|
N/A |
|
1.00 |
|
|
|
0.01 |
|
|
|
0.01
– 0.70 |
|
| Risk-free
interest rate |
|
N/A |
|
1.53
– 1.59 |
% |
|
|
0.05 |
% |
|
|
0.13
– 0.18 |
% |
| Expected
dividend yield |
|
N/A |
|
None |
|
|
|
None
|
|
|
|
None
|
|
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
Fair
Value Measurements:
The
Company measures and reports at fair value the liability for derivative instruments. The fair value liabilities for price adjustable
warrants and embedded conversion options have been recorded as determined utilizing the Binomial Trees model. The following tables summarize
the Company’s financial assets and liabilities measured at fair value on a recurring basis as of June 30, 2021:
SCHEDULE OF FAIR VALUE, ASSETS AND LIABILITIES MEASURED ON RECURRING BASIS
| |
|
Balance
at
June 30, 2021 |
|
|
Quoted
Prices
in
Active
Markets
for
Identical
Assets |
|
|
Significant
Other
Observable
Inputs |
|
|
Significant
Unobservable
Inputs |
|
| |
|
|
|
|
(Level
1) |
|
|
(Level
2) |
|
|
(Level
3) |
|
| Embedded
conversion option liabilities |
|
$ |
54,220 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
54,220 |
|
| Total |
|
$ |
54,220 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
54,220 |
|
The
following tables summarize the Company’s financial assets and liabilities measured at fair value on a recurring basis as of June
30, 2020:
| |
|
Balance
at
June 30, 2020 |
|
|
Quoted
Prices
in
Active
Markets
for
Identical
Assets |
|
|
Significant
Other
Observable
Inputs |
|
|
Significant
Unobservable
Inputs |
|
| |
|
|
|
|
(Level
1) |
|
|
(Level
2) |
|
|
(Level
3) |
|
| Embedded
conversion option liabilities |
|
$ |
177,009 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
177,009 |
|
| Total |
|
$ |
177,009 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
177,009 |
|
The
following is a roll forward for the years ended June 30, 2021 and 2020 of the fair value liability of price adjustable derivative instruments:
SCHEDULE OF DERIVATIVE LIABILITIES AT FAIR VALUE
| |
|
Fair
Value of |
|
| |
|
Liability
for |
|
| |
|
Derivative |
|
| |
|
Instruments |
|
| Balance
at June 30, 2019 |
|
$ |
698,264 |
|
| Reductions
due to conversions |
|
|
(362,962 |
) |
| Initial
fair value of embedded conversion option derivative liability recorded as debt discount |
|
|
227,000 |
|
| Initial
fair value of embedded conversion option derivative liability recorded as expense |
|
|
351,461 |
|
| Change
in fair value included in statements of operations |
|
|
(736,754 |
) |
| Balance
at June 30, 2020 |
|
|
177,009 |
|
| Gain
on debt extinguishment |
|
|
(130,975 |
) |
| Change
in fair value included in statements of operations |
|
|
8,186 |
|
| Balance
at June 30, 2021 |
|
$ |
54,220 |
|
NOTE
13 – SUBSEQUENT EVENTS
Exercise
of Warrants
From
July 9, 2021 through September 7, 2021, the Company received aggregate gross proceeds of $275,000
from the exercise of 6,875
Series B Warrants and issued
6,875 shares of common stock. Additionally, the Company issued 2,399,988
shares of common stock from the
alternate cashless exercise of 12 Series A warrants.
Note
Conversions
From
July 1, 2021 through September 20, 2021, the Company issued an aggregate of 6,702,152
shares of its common stock
at an average contractual conversion price of $0.02,
ranging from $0.01
to $0.04,
as a result of the conversion of principal of $146,348,
interest of $4,887
and conversion fees $2,250
underlying certain outstanding
convertible notes converted during such period. The Company reclassified $86,219
in put premiums to additional
paid in capital following conversions between July 2021 and September 2021.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
June
30, 2021 and 2020
Nathanielsz
Cancellation Agreement
On
August 12, 2021, the Company entered into a Cancellation Agreement with James Nathanielsz (“Nathanielsz”), Chief Executive
Officer and Director of the Company, whereby Nathanielsz agreed to cancel his cash compensation bonus award for fiscal year 2021, ended
June 30, 2021, in exchange for common stock of the Company. The Company and Nathanielsz entered into an Amended and Restated Employment
Agreement dated May 14, 2019 (the “Agreement”). Pursuant to the terms of the Agreement, Nathanielsz was eligible to earn
an annual fiscal year cash performance bonus for each fiscal year of his employment period with the Company with a target performance
bonus of 200% of his average annualized base salary during the fiscal year for which the performance bonus is earned. On July 20, 2021,
Nathanielsz was awarded a “target” bonus of 78%, or $177,840 USD (the “Debt”) for the fiscal year ended June
30, 2021, by the Company’s Board of Directors (the “Board”). Pursuant to the Cancellation Agreement, Nathanielsz agreed
to cancel this Debt in exchange for 5,928,000 shares of the common stock of the Company (the “Shares”), valued at approximately
$0.03 per share, being the closing price of the stock on the date of grant. The shares were issued on August 17, 2021.
Kenyon
Cancellation Agreement
On
August 12, 2021, the Company entered into a Cancellation Agreement with Dr. Julian Kenyon (“Kenyon”), Chief Scientific Officer
and Director of the Company, whereby Kenyon agreed to cancel of $102,600 USD of accrued salary due him as of June 30, 2021, pursuant
to that certain Amended and Restated Services Agreement by and between Kenyon and the Company, dated May 14, 2019, in exchange for 3,420,000
shares of common stock of the Company (the “Shares”), valued at approximately $0.03 per share, being the closing price of
the stock on the date of grant. The shares were issued on August 17, 2021.
Zelinger
Amended and Restated Director Agreement
On
August 12, 2021, the Company entered into an Amended and Restated Director Agreement (the “Director Agreement”) with Josef
Zelinger (“Zelinger”). Pursuant to the terms of the Director Agreement, the Company shall pay Zelinger a base salary of $250.00
AUD per month, payable on the first day of each month. In addition, the Company may compensate Zelinger additional consideration for
advisory services performed by the Director, either in the form of cash or common stock, at the discretion of the Board. The Company
issued 2,800,000 shares of common stock of the Company for accrued director services of $84,000 as of June 30, 2021. The 2,800,000 shares
of common stock was valued at approximately $0.03 per share, being the closing price of the stock on the date of grant. The shares were
issued on August 17, 2021.
Common
Stock Issued for Services
On
August 12, 2021, the Board approved the issuance of 2,800,000 shares of the Company’s common stock for bonus payable of $84,000
as of June 30, 2021 to an employee who is the wife of the CEO of the Company. The 2,800,000 shares of common stock was valued at approximately
$0.03 per share, being the closing price of the stock on the date of grant. The shares were issued on August 17, 2021.
On
August 12, 2021, the Board approved the issuance of 166,667 shares of the Company’s common stock for legal services rendered in
August 2021 worth $5,000. The 5,000 shares of common stock was valued at approximately $0.03 per share, being the closing price of the
stock on the date of grant. The shares were issued on September 3, 2021.
In
September 2021, the Company issued 2,819,712 shares of the Company’s common stock to a consultant for services rendered from July
2021 to September 2021. The Company issued 2,819,712 shares of the Company’s common stock valued at approximately $0.04 per share,
being the closing price of the stock on the date of grant to such consultant, or approximately $113,000.
August
19, 2021 and September 22, 2021 Securities Purchase Agreement
Effective
August 19, 2021, the Company entered into a securities purchase agreement with Geneva Roth Remark Holdings, Inc., pursuant to which
Geneva Roth purchased a convertible promissory note (the “August 19, 2021 Geneva Roth”) from the Company in the
aggregate principal amount of $103,750,
such principal and the interest thereon convertible into shares of the Company’s common stock at the option of Geneva Roth any
time after the six-month anniversary of the August 19, 2021 Geneva Roth. The August 19, 2021 Geneva Roth contains an original
discount of $3,750.
The Company intends to use the net proceeds from the August 19, 2021 Geneva Roth for general working capital purposes. The
maturity date of the August 19, 2021 Geneva Roth Note is August
19, 2022. The August 19, 2021 Geneva Roth Note bears interest at a rate of 8% per
annum, which interest may be paid by the Company to Geneva Roth in shares of the Company’s common stock; but shall not be
payable until the August 19, 2021 Geneva Roth Note becomes payable, whether at the maturity date or upon acceleration or by
prepayment.
Additionally, effective September 22, 2021, the
Company entered into a securities purchase agreement with Geneva Roth Remark Holdings, Inc., pursuant to which Geneva Roth purchased
a convertible promissory note (the “September 22, 2021 Geneva Roth”) from the Company in the aggregate principal amount of
$63,750, such principal and the interest thereon convertible into shares of the Company’s common stock at the option of Geneva
Roth any time after the six-month anniversary of the September 22, 2021 Geneva Roth. The September 22, 2021 Geneva Roth contains an original
discount of $3,750. The Company intends to use the net proceeds from the September 22, 2021 Geneva Roth for general working capital purposes.
The maturity date of the September 22, 2021 Geneva Roth Note is September 22, 2022. The September 22, 2021 Geneva Roth Note bears interest
at a rate of 8% per annum, which interest may be paid by the Company to Geneva Roth in shares of the Company’s common stock; but
shall not be payable until the September 22, 2021 Geneva Roth Note becomes payable, whether at the maturity date or upon acceleration
or by prepayment.
During
the first 60 to 180 days following the date of these notes, the Company has the right to prepay the principal and accrued but unpaid
interest due under the above notes issued to Geneva Roth, together with any other amounts that the Company may owe the holder under the
terms of the note, at a premium ranging from 110% to 129% as defined in the note agreement. After this initial 180-day period, the Company
does not have a right to prepay such notes.
The
conversion price for the above Geneva Roth notes shall be equal to a 35%
discount of the market price which
means the average of the lowest three trading prices of the Common Stock for the ten trading days immediately prior to the delivery of
a Notice of Conversion. Notwithstanding the foregoing, Geneva Roth shall be restricted from effecting a conversion if such conversion,
along with other shares of the Company’s common stock beneficially owned by Geneva Roth and its affiliates, exceeds 9.99%
of the outstanding shares of the
Company’s common stock. These notes are treated as stock settled debt under ASC 480 and accordingly the Company recorded a total
of $90,192
put premium.
The
above Geneva Roth notes contain certain events of default, upon which principal and accrued interest will become immediately due and
payable. In addition, upon an event of default, interest on the outstanding principal shall accrue at a default interest rate of 22%
per annum, or if such rate is usurious or not permitted by current law, then at the highest rate of interest permitted by law. Further,
certain events of default may trigger penalty and liquidated damage provisions.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
CONDENSED
CONSOLIDATED BALANCE SHEETS
The
accompanying unaudited condensed notes are an integral part of these unaudited condensed consolidated financial statements.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
CONDENSED
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE INCOME (LOSS)
(Unaudited)
The
accompanying unaudited condensed notes are an integral part of these unaudited condensed consolidated financial statements.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
CONDENSED
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ DEFICIT FOR THE THREE MONTHS ENDED SEPTEMBER 30, 2021 AND 2020
(Unaudited)
The
accompanying unaudited condensed notes are an integral part of these unaudited condensed consolidated financial statements.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
CONDENSED
CONSOLIDATED STATEMENTS OF CASH FLOWS
(Unaudited)
The
accompanying unaudited condensed notes are an integral part of these unaudited condensed consolidated financial statements.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
NOTE
1 – NATURE OF OPERATIONS AND SUMMARY OF SIGNIFICANT ACCOUNTING AND REPORTING POLICIES
Nature
of Operations
Propanc
Biopharma, Inc. (the “Company,” “we,” “us” or “our”) was originally incorporated in Melbourne,
Victoria Australia on October 15, 2007 as Propanc PTY LTD, and continues to be based in Camberwell, Victoria Australia. Since its inception,
substantially all of the operations of the Company have been focused on the development of new cancer treatments targeting high-risk
patients, particularly cancer survivors, who need a follow-up, non-toxic, long-term therapy designed to prevent the cancer from returning
and spreading. The Company anticipates establishing global markets for its technologies. Our lead product candidate, which we refer to
as PRP, is an enhanced pro-enzyme formulation designed to enhance the anti-cancer effects of multiple enzymes acting synergistically.
It is currently in the preclinical phase of development.
On
November 23, 2010, the Company was incorporated in the state of Delaware as Propanc Health Group Corporation. In January 2011, to reorganize
the Company, we acquired all of the outstanding shares of Propanc PTY LTD on a one-for-one basis making it a wholly-owned subsidiary
of the Company.
On
July 22, 2016, the Company formed a wholly owned subsidiary, Propanc (UK) Limited under the laws of England and Wales for the purpose
of submitting an orphan drug application to the European Medicines Agency as a small and medium-sized enterprise. As of September 30,
2021, there has been no activity within this entity.
Effective
April 20, 2017, the Company changed its name to “Propanc Biopharma, Inc.” to better reflect the Company’s stage of
operations and development.
In
July 2020, a world first patent was granted in Australia for the cancer treatment method patent family. Presently, there are 29 granted
patents and 33 patents under examination in key global jurisdictions relating to the use of proenzymes against solid tumors, covering
the lead product candidate PRP.
The
Company hopes to capture and protect additional patentable subject matter based on the Company’s field of technology relating to
pharmaceutical compositions of proenzymes for treating cancer by filing additional patent applications as it advances its lead product
candidate, PRP, through various stages of development.
On
November 17, 2020, the Company effected a one-for-one thousand (1:1,000) reverse stock split of the Company’s issued and outstanding
shares of common stock (the “Reverse Stock Split”). Proportional adjustments for the Reverse Stock Split were made to the
Company’s outstanding stock options, warrants and equity incentive plans. All share and per-share data and amounts have been retroactively
adjusted as of the earliest period presented in the consolidated financial statements to reflect the Reverse Stock Split.
Basis
of Presentation
The
Company’s interim unaudited condensed consolidated financial statements included in this Quarterly Report on Form 10-Q (this “Quarterly
Report”) have been prepared in accordance with accounting principles generally accepted in the United States of America (“US
GAAP”) and pursuant to the rules and regulations of the U.S. Securities and Exchange Commission (“SEC”). In the opinion
of the Company’s management, all adjustments (consisting of normal recurring adjustments and reclassifications and non-recurring
adjustments) necessary to present fairly our results of operations for the three months ended September 30, 2021 and 2020 and cash flows
for the three months ended September 30, 2021 and 2020 and our financial position at September 30, 2021 have been made. The Company’s
results of operations for the three months ended September 30, 2021 are not necessarily indicative of the operating results to be expected
for the full fiscal year ending June 30, 2022.
Certain
information and disclosures normally included in the notes to the Company’s annual audited consolidated financial statements have
been condensed or omitted from the Company’s interim unaudited condensed consolidated financial statements included in this Quarterly
Report. Accordingly, these interim unaudited condensed consolidated financial statements should be read in conjunction with the Company’s
audited consolidated financial statements and notes thereto for the fiscal year ended June 30, 2021. The June 30, 2021 balance sheet
is derived from those statements.
Principles
of Consolidation
The
unaudited condensed consolidated financial statements include the accounts of Propanc Biopharma, Inc., the parent entity, and its wholly-owned
subsidiary, Propanc PTY LTD. All inter-company balances and transactions have been eliminated in consolidation. Propanc (UK) Limited
was an inactive subsidiary at September 30, 2021.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
Use
of Estimates
The
preparation of financial statements in conformity with the accounting principles generally accepted in the United States of America (“US
GAAP”) requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure
of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of revenues and expenses
during the reporting period. Actual results could differ from these estimates. Significant estimates in the accompanying consolidated
financial statements include the estimates of useful lives for depreciation, valuation of the operating lease liability and related right-of-use
asset, valuation of derivatives, valuation of beneficial conversion features on convertible debt, allowance for uncollectable receivables,
valuation of equity based instruments issued for other than cash, the valuation allowance on deferred tax assets and foreign currency
translation due to certain average exchange rates applied in lieu of spot rates on transaction dates.
Foreign
Currency Translation and Other Comprehensive Income (Loss)
The
Company’s wholly owned subsidiary’s functional currency is the Australian dollar (AUD). For financial reporting purposes,
the Australian dollar has been translated into the Company’s reporting currency which is the United States dollar ($) and/or (USD).
Assets and liabilities are translated at the exchange rate in effect at the balance sheet date. Revenues and expenses are translated
at the average rate of exchange prevailing during the reporting period. Equity transactions are translated at each historical transaction
date spot rate. Translation adjustments arising from the use of different exchange rates from period to period are included as a component
of stockholders’ equity (deficit) as “Accumulated other comprehensive income (loss).” Gains and losses resulting from
foreign currency transactions are included in the statements of operations and comprehensive income (loss) as a component of other comprehensive
income (loss). There have been no significant fluctuations in the exchange rate for the conversion of Australian dollars to USD after
the balance sheet date.
Other
Comprehensive Income (Loss) for all periods presented includes only foreign currency translation gains (losses).
Assets
and liabilities denominated in foreign currencies are translated into the functional currency at the exchange rates prevailing at the
consolidated balance sheet date with any transaction gains and losses that arise from exchange rate fluctuations on transactions denominated
in a currency other than the functional currency included in the consolidated results of operations as incurred. Effective fiscal year
2021, the parent company determined that these intercompany loans will not be repaid in the foreseeable future and thus, per ASC 830-20-35-3,
gains and losses from measuring the intercompany balances are recorded within cumulative translation adjustment, a component of accumulated
other comprehensive income (loss). Prior to July 1, 2020, the Company recorded the foreign currency transaction gains and losses from
measuring the intercompany balances as a component of other income (expenses) titled foreign currency transaction gain (loss). For the
three months ended September 30, 2021 and 2020, the Company recognized an exchange gain (loss) of approximately $619,000 and ($583,000),
on intercompany loans made by the parent to the subsidiary which have not been repaid as of September 30, 2021.
As
of September 30, 2021 and June 30, 2021, the exchange rates used to translate amounts in Australian dollars into USD for the purposes
of preparing the consolidated financial statements were as follows:
SCHEDULE
OF TRANSLATION EXCHANGE RATES
| | |
September
30, 2021 | | |
June
30, 2021 | |
| Exchange rate on balance sheet dates | |
| | | |
| | |
| USD : AUD exchange rate | |
| 0.7214 | | |
| 0.7500 | |
| | |
| | | |
| | |
| Average exchange rate for the period | |
| | | |
| | |
| USD : AUD exchange rate | |
| 0.7350 | | |
| 0.7473 | |
The
change in Accumulated Other Comprehensive Income (Loss) by component during the three months ended September 30, 2021 was as follows:
SCHEDULE
OF CHANGE IN ACCUMULATED OTHER COMPREHENSIVE INCOME (LOSS)
| | |
Foreign
Currency Items: | |
| Balance, June 30, 2021 | |
$ | 1,085,204 | |
| Unrealized foreign currency
translation gain | |
| 64,193 | |
| Ending balance, September 30, 2021 | |
$ | 1,149,397 | |
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
Fair
Value of Financial Instruments and Fair Value Measurements
The
Company measures its financial assets and liabilities in accordance with US GAAP. For certain financial instruments, including cash and
cash equivalents, accounts receivable, accounts payable and accrued liabilities, the carrying amounts approximate fair value due to their
short maturities. Amounts recorded for notes payable, net of discount, and loans payable also approximate fair value because current
interest rates available for debt with similar terms and maturities are substantially the same.
The
Company follows accounting guidance for financial assets and liabilities. This standard defines fair value, provides guidance for measuring
fair value and requires certain disclosures. This standard does not require any new fair value measurements, but rather applies to all
other accounting pronouncements that require or permit fair value measurements. This guidance does not apply to measurements related
to share-based payments. This guidance discusses valuation techniques, such as the market approach (comparable market prices), the income
approach (present value of future income or cash flow), and the cost approach (cost to replace the service capacity of an asset or replacement
cost).
The
guidance utilizes a fair value hierarchy that prioritizes the inputs to valuation techniques used to measure fair value into three broad
levels. The following is a brief description of those three levels:
Level
1: Observable inputs such as quoted prices (unadjusted) in active markets for identical assets or liabilities.
Level
2: Inputs, other than quoted prices that are observable, either directly or indirectly. These include quoted prices for similar assets
or liabilities in active markets and quoted prices for identical or similar assets or liabilities in markets that are not active.
Level
3: Unobservable inputs in which little or no market data exists, therefore developed using estimates and assumptions developed by us,
which reflect those that a market participant would use.
Also
see Note 11 - Derivative Financial Instruments and Fair Value Measurements.
Cash
and Cash Equivalents
Cash
and cash equivalents include cash on hand and at banks, short-term deposits with an original maturity of three months or less with financial
institutions, and bank overdrafts. Bank overdrafts are reflected as a current liability on the balance sheets. There were no cash equivalents
as of September 30, 2021 or June 30, 2021.
Property
and Equipment
Property
and equipment are stated at cost, net of accumulated depreciation. Expenditures for maintenance and repairs are expensed as incurred;
additions, renewals, and betterments are capitalized. When property and equipment are retired or otherwise disposed of, the related cost
and accumulated depreciation are removed from the respective accounts, and any gain or loss is included in operations. Depreciation of
property and equipment is provided using the declining balance method. The depreciable amount is the cost less its residual value.
The
estimated useful lives are as follows:
SCHEDULE
OF PROPERTY AND EQUIPMENT ESTIMATED USEFUL LIVES
| Machinery
and equipment |
-
5 years |
| Furniture |
-
7 years |
Patents
Patents
are stated at cost and amortized on a straight-line basis over the estimated future periods if and once the patent has been granted by
a regulatory agency. However, the Company will expense any patent costs as long as we are in the startup stage. Accordingly, as the Company’s
products are not currently approved for market, all patent costs incurred from 2013 through September 30, 2021 were expensed immediately.
This practice of expensing patent costs immediately ends when a product receives market authorization from a government regulatory agency.
Impairment
of Long-Lived Assets
In
accordance with ASC 360-10, “Long-lived assets,” which include property and equipment and intangible assets, are reviewed
for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. Recoverability
of long-lived assets to be held and used is measured by a comparison of the carrying amount of an asset to the estimated undiscounted
future cash flows expected to be generated by the asset. If the carrying amount of an asset exceeds its estimated undiscounted future
cash flows, an impairment charge is recognized by the amount by which the carrying amount of the asset exceeds the fair value of the
assets. Fair value is generally determined using the asset’s expected future discounted cash flows or market value, if readily
determinable.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
Employee
Benefit/Liability
Liabilities
arising in respect of wages and salaries, accumulated annual leave, accumulated long service leave and any other employee benefits expected
to be settled within twelve months of the reporting date are measured based on the employee’s remuneration rates applicable at
the reporting date. All other employee benefit liabilities are measured at the present value of the estimated future cash outflow to
be made in respect of services provided by employees up to the reporting date. All employee liabilities are owed within the next twelve
months.
Australian
Goods and Services Tax (“GST”)
Revenues,
expenses and balance sheet items are recognized net of the amount of GST, except payable and receivable balances which are shown inclusive
of GST. The GST incurred is payable on revenues to, and recoverable on purchases from, the Australian Taxation Office.
Cash
flows are presented in the statements of cash flow on a gross basis, except for the GST component of investing and financing activities,
which are disclosed as operating cash flows.
As
of September 30, 2021, and June 30, 2021, the Company was owed $2,238 and $4,341, respectively, from the Australian Taxation Office.
These amounts were fully collected subsequent to the balance sheet reporting dates.
Derivative
Instruments
ASC
Topic 815, Derivatives and Hedging (“ASC Topic 815”), establishes accounting and reporting standards for derivative
instruments and for hedging activities by requiring that all derivatives be recognized in the balance sheet and measured at fair value.
Gains or losses resulting from changes in the fair value of derivatives are recognized in earnings. On the date of conversion or payoff
of debt, the Company records the fair value of the conversion shares, removes the fair value of the related derivative liability, removes
any discounts and records a net gain or loss on debt extinguishment. On July 1, 2019 the Company adopted ASU 2017-11 under which down-round
Features in Financial Instruments will no longer cause derivative treatment. The Company applies the modified prospective method of adoption.
There were no cumulative effects on adoption.
Convertible
Notes With Variable Conversion Options
The
Company has entered into convertible notes, some of which contain variable conversion options, whereby the outstanding principal and
accrued interest may be converted, by the holder, into common shares at a fixed discount to the price of the common stock at or around
the time of conversion. The Company treats these convertible notes as stock settled debt under ASC 480, “Distinguishing Liabilities
from Equity” and measures the fair value of the notes at the time of issuance, which is the result of the share price discount
at the time of conversion and records the put premium as interest expense.
Income
Taxes
The
Company is governed by Australia and United States income tax laws, which are administered by the Australian Taxation Office and the
United States Internal Revenue Service, respectively. The Company follows ASC 740 “Accounting for Income Taxes,” when
accounting for income taxes, which requires an asset and liability approach to financial accounting and reporting for income taxes. Deferred
income tax assets and liabilities are computed annually for temporary differences between the financial statements and tax bases of assets
and liabilities that will result in taxable or deductible amounts in the future based on enacted tax laws and rates applicable to the
periods in which the differences are expected to affect taxable income. Valuation allowances are established when necessary to reduce
deferred tax assets to the amount expected to be realized. Income tax expense is the tax payable or refundable for the period plus or
minus the change during the period in deferred tax assets and liabilities.
The
Company follows ASC 740, Sections 25 through 60, “Accounting for Uncertainty in Income Taxes.” These sections provide
detailed guidance for the financial statement recognition, measurement and disclosure of uncertain tax positions recognized in the financial
statements. Tax positions must meet a “more-likely-than-not” recognition threshold at the effective date to be recognized
upon the adoption of ASC 740 and in subsequent periods.
Research
and Development Costs and Tax Credits
In
accordance with ASC 730-10, “Research and Development-Overall,” research and development costs are expensed when incurred.
Total research and development costs for the three months ended September 30, 2021 and 2020 were $46,554 and $50,846, respectively.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
The
Company may apply for research and development tax concessions with the Australian Taxation Office on an annual basis. Although the amount
is possible to estimate at year end, the Australian Taxation Office may reject or materially alter the claim amount. Accordingly, the
Company does not recognize the benefit of the claim amount until cash receipt since collectability is not certain until such time. The
tax concession is a refundable credit. If the Company has net income, then the Company can receive the credit which reduces its income
tax liability. If the Company has net losses, then the Company may still receive a cash payment for the credit, however, the Company’s
net operating loss carryforwards are reduced by the gross equivalent loss that would produce the credit amount when the income tax rate
is applied to that gross amount. The concession is recognized as tax benefit, in operations, upon receipt.
During
each of the three months ended September 30, 2021 and 2020, the Company applied for, and received from the Australian Taxation Office,
a research and development tax credit in the amount of $0, which is reflected as a tax benefit in the accompanying unaudited condensed
consolidated statements of operations and comprehensive income (loss).
Stock
Based Compensation
The
Company records stock-based compensation in accordance with ASC 718, “Stock Compensation”. ASC 718 requires the fair
value of all stock-based employee compensation awarded to employees to be recorded as an expense over the shorter of the service period
or the vesting period. The Company values employee and non-employee stock-based compensation at fair value using the Black-Scholes Option
Pricing Model.
The
Company adopted ASU 2018-07 and accounts for non-employee share-based awards in accordance with the measurement and recognition criteria
of ASC 718 and recognizes the fair value of such awards over the service period. The Company used the modified prospective method of
adoption. There was no cumulative effect of adoption on July 1, 2019.
Revenue
Recognition
The
Company adopted and implemented on July 1, 2018, ASC 606 – Revenue from Contracts with Customers (“ASC 606”). ASC 606
did not have a material impact on the consolidated financial statements.
Upon
implementation of ASC 606, the Company recognizes revenue in accordance with that core principle by applying the following steps:
Step
1: Identify the contract(s) with a customer.
Step
2: Identify the performance obligations in the contract.
Step
3: Determine the transaction price.
Step
4: Allocate the transaction price to the performance obligations in the contract.
Step
5: Recognize revenue when (or as) the entity satisfies a performance obligation.
Subject
to these criteria, the Company intends to recognize revenue relating to royalties on product sales in the period in which the sale occurs
and the royalty term has begun.
Legal
Expenses
All
legal costs for litigation are charged to expense as incurred.
Leases
In
February 2016, the Financial Accounting Standards Board (“FASB”) issued ASU 2016-02, Leases (Topic 842). The updated
guidance requires lessees to recognize lease assets and lease liabilities for most operating leases. In addition, the updated guidance
requires that lessors separate lease and non-lease components in a contract in accordance with the new revenue guidance in ASC 606. This
guidance is effective for interim and annual reporting periods beginning after December 15, 2018. The Company adopted this guidance effective
July 1, 2019.
On
July 1, 2019, the Company adopted ASU No. 2016-02, applying the package of practical expedients to leases that commenced before the effective
date whereby the Company elected to not reassess the following: (i) whether any expired or existing contracts contain leases and; (ii)
initial direct costs for any existing leases. For contracts entered into on or after the effective date, at the inception of a contract
the Company assessed whether the contract is, or contains, a lease. The Company’s assessment is based on: (1) whether the contract
involves the use of a distinct identified asset, (2) whether we obtain the right to substantially all the economic benefit from the use
of the asset throughout the period, and (3) whether it has the right to direct the use of the asset. The Company will allocate the consideration
in the contract to each lease component based on its relative stand-alone price to determine the lease payments. In addition, the Company
elected not to apply ASC Topic 842 to arrangements with lease terms of 12 months or less.
Operating
lease ROU assets represents the right to use the leased asset for the lease term and operating lease liabilities are recognized based
on the present value of future minimum lease payments over the lease term at commencement date. As most leases do not provide an implicit
rate, the Company use an incremental borrowing rate based on the information available at the adoption date in determining the present
value of future payments. Lease expense for minimum lease payments is amortized on a straight-line basis over the lease term and will
be included in general and administrative expenses.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
Basic
and Diluted Net Loss Per Common Share
Basic
net loss per share is computed by dividing the net loss by the weighted average number of common shares outstanding during the period.
Diluted net loss per common share is computed by dividing the net loss by the weighted average number of common shares outstanding for
the period and, if dilutive, potential common shares outstanding during the period. Potentially dilutive securities consist of the incremental
common shares issuable upon exercise of common stock equivalents such as stock options, warrants and convertible debt instruments. Potentially
dilutive securities are excluded from the computation if their effect is anti-dilutive. As a result, the basic and diluted per share
amounts for all periods presented are identical. Each holder of the notes has agreed to a 4.99% beneficial ownership conversion limitation
(subject to certain noteholders’ ability to increase such limitation to 9.99% upon 60 days’ notice to the Company), and each
note may not be converted during the first six-month period from the date of issuance. The securities for the period ended September
30, 2021 and 2020 were considered dilutive securities which were excluded from the computation since the effect is anti-dilutive.
SCHEDULE
OF DILUTIVE SECURITIES EXCLUDED FROM COMPUTATION
| | |
September
30, 2021 | | |
September
30, 2020 | |
| | |
| (Unaudited) | | |
| (Unaudited) | |
| Stock Options | |
| 59 | | |
| 60 | |
| Stock Warrants | |
| 111,932 | | |
| 135,725 | |
| Unvested restricted stock | |
| 59 | | |
| 117 | |
| Convertible Debt | |
| 23,293,971 | | |
| 510,674 | |
| Total | |
| 23,406,021 | | |
| 646,576 | |
Recent
Accounting Pronouncements
We
have reviewed the FASB issued ASU accounting pronouncements and interpretations thereof that have effectiveness dates during the periods
reported and in future periods. We have carefully considered the new pronouncements that alter previous generally accepted accounting
principles and do not believe that any new or modified principles will have a material impact on the Company’s reported financial
position or operations in the near term. The applicability of any standard is subject to the formal review of the Company’s financial
management.
In
August 2020, the FASB issued ASU 2020-06, “Debt – Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives
and Hedging – Contracts in Entity’s Own Equity (Subtopic 815-40)”. This ASU reduces the number of accounting models
for convertible debt instruments and convertible preferred stock. As well as amend the guidance for the derivatives scope exception for
contracts in an entity’s own equity to reduce form-over-substance-based accounting conclusions. In addition, this ASU improves
and amends the related EPS guidance. This standard is effective for us on July 1, 2022, including interim periods within those fiscal
years. Adoption is either a modified retrospective method or a fully retrospective method of transition. The Company is currently assessing
the impact the new guidance will have on our consolidated financial statements.
NOTE
2 – GOING CONCERN
The
accompanying unaudited condensed consolidated financial statements have been prepared in conformity with US GAAP, which contemplate continuation
of the Company as a going concern. For the three months ended September 30, 2021, the Company had no revenues, had a net loss of $490,658,
and had net cash used in operations of $486,758. Additionally, as of September 30, 2021, the Company had a working capital deficit, stockholders’
deficit and accumulated deficit of $2,312,387, $2,306,630 and $58,804,968, respectively. It is management’s opinion that these
conditions raise substantial doubt about the Company’s ability to continue as a going concern for a period of at least twelve months
from the issue date of this Quarterly Report.
The
unaudited condensed consolidated financial statements do not include any adjustments to reflect the possible future effect on the recoverability
and classification of assets or the amounts and classifications of liabilities that may result from the outcome of this uncertainty.
Successful
completion of the Company’s development program and, ultimately, the attainment of profitable operations are dependent upon future
events, including obtaining adequate financing to fulfill its development activities, acceptance of the Company’s patent applications,
obtaining additional sources of suitable and adequate financing and ultimately achieving a level of sales adequate to support the Company’s
cost structure and business plan. The Company’s ability to continue as a going concern is also dependent on its ability to further
develop and execute on its business plan. However, there can be no assurances that any or all of these endeavors will be successful.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
In
March 2020, the outbreak of COVID-19 (coronavirus) caused by a novel strain of the coronavirus was recognized as a pandemic by the World
Health Organization, and the outbreak has become increasingly widespread in the United States, Europe and Australia, including in each
of the areas in which the Company operates. The COVID-19 (coronavirus) outbreak has had a notable impact on general economic conditions,
including but not limited to the temporary closures of many businesses, “shelter in place” and other governmental regulations,
reduced business and consumer spending due to both job losses, reduced investing activity and M&A transactions, among many other
effects attributable to the COVID-19 (coronavirus), and there continue to be many unknowns. While to date the Company has not been required
to stop operating, management is evaluating its use of its office space, virtual meetings and the like. The Company continues to monitor
the impact of the COVID-19 (coronavirus) outbreak closely. The extent to which the COVID-19 (coronavirus) outbreak will impact our operations,
ability to obtain financing or future financial results is uncertain.
NOTE
3 – PROPERTY AND EQUIPMENT
Property
and equipment consist of the following as of September 30, 2021 and June 30, 2021.
SCHEDULE
OF PROPERTY AND EQUIPMENT
| | |
September
30, 2021 | | |
June
30, 2021 | |
| | |
| (Unaudited) | | |
| | |
| Office equipment at cost | |
$ | 27,532 | | |
$ | 28,623 | |
| Less: Accumulated depreciation | |
| (23,939 | ) | |
| (24,368 | ) |
| | |
| | | |
| | |
| Total property, plant,
and equipment | |
$ | 3,593 | | |
$ | 4,255 | |
Depreciation
expense for the three months ended September 30, 2021 and 2020 were $509 and $438, respectively.
NOTE
4 – DUE TO FORMER DIRECTOR - RELATED PARTY
Due
to former director - related party represents unsecured advances made primarily by a former director for operating expenses on behalf
of the Company such as intellectual property and formation expenses. The expenses were paid for on behalf of the Company and are due
upon demand. The Company is currently not being charged interest under these advances. The total amount owed the former director at September
30, 2021 and June 30, 2021 were $32,076 and $33,347, respectively. The Company plans to repay the advances as its cash resources allow
(see Note 9).
NOTE
5 – LOANS AND NOTES PAYABLE
Loan
from Former Director - Related Party
Loan
from the Company’s former director at September 30, 2021 and June 30, 2021 were $53,384 and $55,500, respectively. The loan bears
no interest and is payable on demand. The Company did not repay any amount on this loan during the three months ended September 30, 2021
and 2020, respectively (see Note 9).
NOTE
6 – CONVERTIBLE NOTES
The
Company’s convertible notes outstanding at September 30, 2021 and June 30, 2021 were as follows:
SCHEDULE
OF CONVERTIBLE NOTES
| | |
September
30, 2021 | | |
June
30, 2021 | |
| | |
| (Unaudited) | | |
| | |
| Convertible notes and debenture | |
$ | 377,780 | | |
$ | 400,128 | |
| Unamortized discounts | |
| (7,565 | ) | |
| (6,139 | ) |
| Accrued interest | |
| 37,348 | | |
| 34,098 | |
| Premium, net | |
| 177,045 | | |
| 196,496 | |
| Convertible notes,
net | |
$ | 584,608 | | |
$ | 624,583 | |
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
Convertible
Note Issued with Consulting Agreement
August
10, 2017 Consulting Agreement
On
August 10, 2017, the Company entered into a consulting agreement, retroactive to May 16, 2017, with a certain consultant, pursuant to
which the consultant agreed to provide certain consulting and business advisory services in exchange for a $310,000 junior subordinated
convertible note. The maturity date of the August 10, 2017 Convertible Note was August 2019 and is currently past due (see Note 8). The
note accrues interest at a rate of 10% per annum and is convertible into common stock at the lesser of $750 or 65% of the three lowest
trades in the ten trading days prior to the conversion. The note was fully earned upon signing the agreement and matures on August 10,
2019. The Company accrued $155,000 related to this expense at June 30, 2017 and recorded the remaining $155,000 related to this expense
in fiscal year 2018. Upon an event of default, principal and accrued interest will become immediately due and payable under the note.
Additionally, upon an event of default, at the election of the holder, the note would accrue interest at a default interest rate of 18%
per annum or the highest rate of interest permitted by law. The consulting agreement had a three-month term and expired on August 16,
2017. An aggregate total of $578,212 of this note was bifurcated with the embedded conversion option recorded as a derivative liability
at fair value. During the year ended June 30, 2018, the consultant converted $140,000 of principal and $10,764 of interest. During the
year ended June 30, 2019, the consultant converted an additional $161,000 of principal and $19,418 of interest leaving a principal balance
owed of $9,000 at June 30, 2019. During the year ended June 30, 2020, the consultant converted an additional $500 of principal and $5,248
of interest such that the remaining principal outstanding and accrued interest under this note as of June 30, 2020 was $8,500 and $22,168,
respectively.
On
March 15, 2021, the Company entered into a Settlement and Mutual Release Agreement (the “Settlement Agreement”) with the
consultant whereby both parties agreed to settle all claims and liabilities under the August 10, 2017 Convertible note for a total of
$100,000 in the form of a convertible note. All other terms of the August 10, 2017 Convertible Note shall remain in full force and effect.
Both parties agree that all future penalties under this note are waived unless the Company fails to authorize to distribute the requested
shares upon conversion. The Company has the right to pay off the balance of any remaining amounts dues under this note in cash at any
time more than 60 days after March 15, 2021. Prior to the Settlement Agreement, the Company recorded total liabilities $56,762 consisting
of remaining principal amount of $8,500, accrued interest of $23,262 and accrued expenses of $25,000. Accordingly, the Company recognized
loss from settlement of debt of $43,238 during the year ended June 30, 2021.
The
total principal outstanding after adjustment due to the above-mentioned March 15, 2021 settlement agreement and accrued interest under
the August 10, 2017 Convertible Note was $80,000 and $3,738, respectively, as of June 30, 2021 following conversion of $20,000 of principal
during the year ended June 30, 2021. The total principal amount outstanding under the August 10, 2017 Convertible Note was $80,000 and
accrued interest of $7,381 as of as of September 30, 2021.
Auctus
Fund Financing Agreements
August
30, 2019 Securities Purchase Agreement
Effective
August 30, 2019, the Company entered into a securities purchase agreement with Auctus Fund, LLC (“Auctus”), pursuant to which
Auctus purchased a convertible promissory note (the “August 30, 2019 Auctus Note”) from the Company in the aggregate principal
amount of $550,000, such principal and the interest thereon convertible into shares of the Company’s common stock at the option
of Auctus. The transaction closed on August 30, 2019 and the Company received payment on September 4, 2019 in the amount of $550,000,
of which $5,000 was paid directly toward legal fees and $40,000 to Auctus for due diligence fees resulting in net cash proceeds of $505,000.
The maturity date of the August 30, 2019 Auctus Note was August 30, 2020 and was currently past due. The August 30, 2019 Auctus Note
bore interest at a rate of 10% per annum, but not payable until the August 30, 2019 Auctus Note became payable, whether at the maturity
date or upon acceleration or by prepayment. The note was treated as stock settled debt under ASC 480 and accordingly the Company recorded
a $366,667 put premium. The August 30, 2019 Auctus Note may not be prepaid without the written consent of Auctus. Any amount of principal
or interest which was not paid when due shall bear interest at the rate of 24% per annum.
Additionally,
Auctus had the option to convert all or any amount of the principal face amount and accrued interest of the August 30, 2019 Auctus Note,
at any time following the issue date and ending on the later of the maturity date or the date of payment of the Default Amount if an
event of default occurs, which was an amount equal to 125% of an amount equal to the then outstanding principal amount of the August
30, 2019 Auctus Note (but not less than $15,000) plus any interest accrued from August 30, 2019 at the default interest rate of 24% per
annum, for shares of the Company’s common stock at the then-applicable conversion price. Upon the holder’s election to convert
accrued interest, default interest or any penalty amounts as stipulated, the Company may elect to pay those amounts in cash. The note
may also be prepaid by the Company at any time between the date of issuance and August 13, 2020 at 135% multiplied by the sum of (a)
the then outstanding principal amount plus (b) accrued and unpaid interest plus (c) default interests, if any.
The
conversion price for the August 30, 2019 Auctus Note was equal to the Variable Conversion Price of 60% of the Market Price on the date
of conversion. Notwithstanding the foregoing, Auctus shall be restricted from effecting a conversion if such conversion, along with other
shares of the Company’s common stock beneficially owned by Auctus and its affiliates, exceeds 4.99% of the outstanding shares of
the Company’s common stock.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
In
connection with the issuance of the August 2019 Auctus Note, the Company issued common stock purchase warrants to Auctus to purchase
450 shares of the Company’s common stock (the “First Warrant”) as a commitment fee upon the terms and subject to the
limitations and conditions set forth in such First Warrant at an “Exercise Price” of $2,250. In connection with the issuance
of the Note, the Company issued a common stock purchase warrant to Buyer to purchase 300 shares of the Company’s common stock (the
“Second Warrant”) as a commitment fee upon the terms and subject to the limitations and conditions set forth in such Second
Warrant at an “Exercise Price” of $3,330. In connection with the issuance of the Note, the Company shall issue a common stock
purchase warrant to Buyer to purchase 225 shares of the Company’s common stock (the “Third Warrant”) as a commitment
fee upon the terms and subject to the limitations and conditions set forth in such Third Warrant at an “Exercise Price” of
$4,500. The First Warrant, Second Warrant, and Third Warrant were collectively be referred as the “Warrants”. The Warrants
have an “Exercise Period” of five years from the date of issuance being August 30, 2019. Under the terms of the Purchase
Agreement and the Warrants, the Selling Security Holder may not either convert the Notes nor exercise the Warrants to the extent (but
only to the extent) that the Selling Security Holder or any of its affiliates would beneficially own a number of shares of our Common
Stock which would exceed 4.99% of our outstanding shares. The Company accounted for the warrants by using the relative fair value method
and recorded debt discount from the relative fair value of the warrants of $375,905 using a simple binomial lattice model.
In
connection with the Purchase Agreement, the Company and the Purchaser entered into a Registration Rights Agreement (the “Registration
Rights Agreement”). Pursuant to the Registration Rights Agreement, the Company agreed to register the shares of Common Stock underlying
the Securities in a Registration Statement with the SEC as well as the Commitment Shares (as defined herein). The Registration Rights
Agreement contains customary representations, warranties, agreements and indemnification rights and obligations of the parties.
The
Note was subject to customary default provisions and also includes a cross-default provision which provides that a breach or default
by the Borrower of any covenant or other term or condition contained in any of the Other Agreements (as defined therein), after the passage
of all applicable notice and cure or grace periods, shall, at the option of the Holder, be considered a default under this Note and the
Other Agreements. Upon occurrence of any such event, the Holder was entitled (but in no event required) to apply all rights and remedies
of the Holder under the terms of this Note and the Other Agreements by reason of a default under said Other Agreements or the Note.
The
August 30, 2019 Auctus Note contained certain events of default, upon which principal and accrued interest will become immediately due
and payable. In addition, upon an event of default, interest on the outstanding principal accrued at a default interest rate of 24% per
annum.
The
total principal amount outstanding under the above Auctus financing agreement, specifically the August 30, 2019 Auctus Note, was $358,965
and accrued interest of $486 as of June 30, 2020 following conversion of $191,035 of the principal balance and $43,176 of accrued interest
during the year ended June 30, 2020. Accordingly, $127,356 of the put premium was released in respect of the August 30, 2019 Auctus Note
during the year ended June 30, 2020 following conversion of the principal balance.
The
total principal amount outstanding under the above Auctus financing agreement, specifically the August 30, 2019 Auctus Note, was $32,848
and accrued interest of $0 as of June 30, 2021 following conversion of $326,117 of the principal balance and $39,536 of accrued interest
during the year ended June 30, 2021. Accordingly, $217,411 of the put premium was released in respect of the August 30, 2019 Auctus Note
during the year ended June 30, 2021 following conversion of the principal balance.
The
total principal amount outstanding under the above Auctus financing agreement, specifically the August 30, 2019 Auctus Note, was $0 and
accrued interest of $0 as of September 30, 2021 following conversion of $32,848 of the principal balance and $716 of accrued interest
during the three months ended September 30, 2021. Accordingly, $21,899 of the put premium was released in respect of the August 30, 2019
Auctus Note during the three months ended September 30, 2021 following conversion of the principal balance. Accordingly, there was no
outstanding principal balance as of September 30, 2021.
Crown
Bridge Securities Purchase Agreements
Effective
October 3, 2019, the Company entered into a securities purchase agreement with Crown Bridge Partners, pursuant to which Crown Bridge
purchased a convertible promissory note (the “October 3, 2019 Crown Bridge Note”) from the Company in the aggregate principal
amount of $108,000, such principal and the interest thereon convertible into shares of the Company’s common stock at the option
of Crown Bridge any time from the of issuance of the of the October 3, 2019 Crown Bridge Note. The transactions contemplated by the Crown
Bridge Securities Purchase Agreement closed on October 3, 2019. Pursuant to the terms of the Crown Bridge Securities Purchase Agreement,
Crown Bridge deducted $3,000 from the principal payment due under the October 3, 2019 Crown Bridge Note, at the time of closing, to be
applied to its legal expenses, and there was a $5,000 original issuance discount resulting in $100,000 net proceeds to the Company. The
Company intends to use the net proceeds from the October 3, 2019 Crown Bridge Note for general working capital purposes. The maturity
date of the October 3, 2019 Crown Bridge was October 3, 2020 and is currently past due. The October 3, 2019 Crown Bridge Note bears interest
at a rate of 10% per annum, which interest may be paid by the Company to Crown Bridge in shares of the Company’s common stock;
but shall not be payable until the October 2019 Crown Bridge Note becomes payable, whether at the maturity date or upon acceleration
or by prepayment.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
Additionally,
Crown Bridge has the option to convert all or any amount of the principal face amount of the October 3, 2019 Crown Bridge Note at any
time from the date of issuance and ending on the later of the maturity date or the date the Default Amount is paid if an event of default
occurs, which is an amount between 110% and 150% of an amount equal to the then outstanding principal amount of the October 3, 2019 Crown
Bridge Note plus any interest accrued, for shares of the Company’s common stock at the then-applicable conversion price.
The
conversion price for the October 3, 2019 Crown Bridge Note shall be equal to a 40% discount of the lowest closing bid price (“Lowest
Trading Price”) of the Common Stock for the ten trading days immediately prior to the delivery of a Notice of Conversion, including
the day upon which a Notice of Conversion is received. Notwithstanding the foregoing, Crown Bridge shall be restricted from effecting
a conversion if such conversion, along with other shares of the Company’s common stock beneficially owned by Crown Bridge and its
affiliates, exceeds 4.99% of the outstanding shares of the Company’s common stock which may be increased up to 9.99% upon 60 days
prior written notice by the Crown Bridge to the Company. The note is treated as stock settled debt under ASC 480 and accordingly the
Company recorded a $72,000 put premium.
The
October 3, 2019 Crown Bridge Note contain certain events of default, upon which principal and accrued interest will become immediately
due and payable. In addition, upon an event of default, interest on the outstanding principal shall accrue at a default interest rate
of 15% per annum, or if such rate is usurious or not permitted by current law, then at the highest rate of interest permitted by law.
Further, certain events of default may trigger penalty and liquidated damage provisions.
The
total principal amount outstanding under the above Crown Bridge financing agreement was $65,280 and accrued interest of $7,232 as of
as of June 30, 2020 following conversion of $42,720 of the principal balance during the year ended June 30, 2020. Accordingly, $28,480
of the put premium was released in respect of the October 3, 2019 Crown Bridge Note during the year ended June 30, 2020 following conversion
of the principal balance.
There
were 15,000 unissued shares which were considered issuable for accounting purposes during the 1st quarter of fiscal 2021 related
to a conversion notice dated and received on September 16, 2020. In November 2020, the Company was notified by the note holder of the
cancellation of this conversion notice as a result of the reverse stock split and as such the Company reversed the effects of this transaction
thereby increasing the principal balance by $9,600 and put premium by $6,400 and a corresponding decrease in equity of $16,000.
The
total principal amount outstanding under the above Crown Bridge financing agreement was $65,280 and accrued interest of $16,138
as of June 30, 2021. The total principal amount outstanding under the above Crown Bridge financing agreement was $65,280 and accrued
interest of $18,606 as of as of September 30, 2021.
GW
Holdings Securities Purchase Agreements
December
10, 2020 Securities Purchase Agreement
Effective
December 10, 2020, the Company entered into a securities purchase agreement with GW Holdings, pursuant to which GW Holdings purchased
a convertible promissory note (the “December 10, 2020 GW Note”) from the Company in the aggregate principal amount of $131,000,
such principal and the interest thereon convertible into shares of the Company’s common stock at the option of GW Holdings anytime
from the issuance of the December 10, 2020 GW Holdings Note. The transactions contemplated by the GW Holdings Securities Purchase Agreement
closed on December 10, 2020. Pursuant to the terms of the GW Holdings Securities Purchase Agreement, the lender deducted $6,000 from
the principal payment due under the December 10, 2020 GW Note, at the time of closing, to be applied to its legal expenses. The Company
intends to use the net proceeds of $125,000 from the December 10, 2020 GW Note for general working capital purposes. The maturity date
of the December 10, 2020 GW Holdings is December 10, 2021. The December 10, 2020 GW Holdings Note bears interest at a rate of 8% per
annum, which interest may be paid by the Company to GW Holdings in shares of the Company’s common stock; but shall not be payable
until the December 10, 2020 GW Holdings Note becomes payable, whether at the maturity date or upon acceleration or by prepayment.
The
above notes issued to GW Holdings contain certain events of default, upon which principal and accrued interest will become immediately
due and payable. In addition, upon an event of default, interest on the outstanding principal shall accrue at a default interest rate
of 24% per annum, or if such rate is usurious or not permitted by current law, then at the highest rate of interest permitted by law.
Further, certain events of default may trigger penalty and liquidated damage provisions.
Additionally,
GW Holdings has the option to convert all or any amount of the principal face amount of the notes issued to GW Holdings at any time from
the date of issuance and ending on the later of the maturity date or the date the Default Amount is paid if an event of default occurs,
which is an amount between 110% and 150% of an amount equal to the then outstanding principal amount of such notes plus any interest
accrued, for shares of the Company’s common stock at the then-applicable conversion price.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
The
conversion price for the above GW Holdings notes shall be equal to a 40% discount of the lowest closing bid price (“Lowest Trading
Price”) of the Common Stock for the ten trading days immediately prior to the delivery of a Notice of Conversion, including the
day upon which a Notice of Conversion is received. Notwithstanding the foregoing, GW Holdings shall be restricted from effecting a conversion
if such conversion, along with other shares of the Company’s common stock beneficially owned by GW Holdings and its affiliates,
exceeds 4.99% of the outstanding shares of the Company’s common stock which may be increased up to 9.99% upon 60 days prior written
notice by the GW Holdings to the Company.
These
notes are treated as stock settled debt under ASC 480 and accordingly the Company recorded a total of $87,333 put premium.
The
total principal amount outstanding under the above December 10, 2020 GW Holdings financing agreement, was $90,000 and accrued interest
of $4,636 as of June 30, 2021 following conversion of $41,000 of the principal balance and $1,084 of accrued interest during the year
ended June 30, 2021. Accordingly, $27,333 of the put premium was reclassed to additional paid in capital in respect of the October 1,
2019 GW Holdings Note during the year ended June 30, 2021 following conversion of the principal balance.
The
total principal amount outstanding under the above December 10, 2020 GW Holdings financing agreement, was $65,000 and accrued interest
of $4,174 as of September 30, 2021 following conversion of $25,000 of the principal balance and $2,091 of accrued interest during the
three months ended September 30, 2021. Accordingly, $16,667 of the put premium was reclassed to additional paid in capital in respect
of the October 1, 2019 GW Holdings Note during the three months ended September 30, 2021 following conversion of the principal balance.
Geneva
Roth Remark Securities Purchase Agreements
January
5, 2021 Securities Purchase Agreement
Effective
January 5, 2021, the Company entered into a securities purchase agreement with Geneva Roth Remark Holdings, Inc., pursuant to which Geneva
Roth purchased a convertible promissory note (the “January 5, 2021 Geneva Roth”) from the Company in the aggregate principal
amount of $68,500, such principal and the interest thereon convertible into shares of the Company’s common stock at the option
of Geneva Roth any time after the six-month anniversary of the January 5, 2021 Geneva Roth. The January 5, 2021 Geneva Roth contained
an original issue discount of $3,500. The Company intended to use the net proceeds from the January 5, 2021 Geneva Roth for general working
capital purposes. The maturity date of the January 5, 2021 Geneva Roth Note was January 5, 2022. The January 5, 2021 Geneva Roth Note
bore interest at a rate of 8% per annum, which interest may be paid by the Company to Geneva Roth in shares of the Company’s common
stock; but shall not be payable until the January 5, 2021 Geneva Roth Note becomes payable, whether at the maturity date or upon acceleration
or by prepayment.
March
16, 2021 Securities Purchase Agreement
Effective
March 16, 2021, the Company entered into a securities purchase agreement with Geneva Roth Remark Holdings, Inc., pursuant to which Geneva
Roth purchased a convertible promissory note (the “March 16, 2021 Geneva Roth”) from the Company in the aggregate principal
amount of $63,500, such principal and the interest thereon convertible into shares of the Company’s common stock at the option
of Geneva Roth any time after the six-month anniversary of the March 16, 2021 Geneva Roth. The March 16, 2021 Geneva Roth contained an
original discount of $3,500. The Company intended to use the net proceeds from the March 16, 2021 Geneva Roth for general working capital
purposes. The maturity date of the March 16, 2021 Geneva Roth Note was March 16, 2022. The March 16, 2021 Geneva Roth Note bears interest
at a rate of 8% per annum, which interest may be paid by the Company to Geneva Roth in shares of the Company’s common stock; but
shall not be payable until the March 16, 2021 Geneva Roth Note becomes payable, whether at the maturity date or upon acceleration or
by prepayment.
August
19, 2021 Securities Purchase Agreement
Effective
August 19, 2021, the Company entered into a securities purchase agreement with Geneva Roth Remark Holdings, Inc., pursuant to which Geneva
Roth purchased a convertible promissory note (the “August 19, 2021 Geneva Roth”) from the Company in the aggregate principal
amount of $103,750, such principal and the interest thereon convertible into shares of the Company’s common stock at the option
of Geneva Roth any time after the six-month anniversary of the August 19, 2021 Geneva Roth. The August 19, 2021 Geneva Roth contains
an original discount of $3,750. The Company intends to use the net proceeds from the August 19, 2021 Geneva Roth for general working
capital purposes. The maturity date of the August 19, 2021 Geneva Roth Note is August 19, 2022. The August 19, 2021 Geneva Roth Note
bears interest at a rate of 8% per annum, which interest may be paid by the Company to Geneva Roth in shares of the Company’s common
stock; but shall not be payable until the August 19, 2021 Geneva Roth Note becomes payable, whether at the maturity date or upon acceleration
or by prepayment.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
September
22, 2021 Securities Purchase Agreement
Additionally,
effective September 22, 2021, the Company entered into a securities purchase agreement with Geneva Roth Remark Holdings, Inc., pursuant
to which Geneva Roth purchased a convertible promissory note (the “September 22, 2021 Geneva Roth”) from the Company in the
aggregate principal amount of $63,750, such principal and the interest thereon convertible into shares of the Company’s common
stock at the option of Geneva Roth any time after the six-month anniversary of the September 22, 2021 Geneva Roth. The September 22,
2021 Geneva Roth contains an original discount of $3,750. The Company intends to use the net proceeds from the September 22, 2021 Geneva
Roth for general working capital purposes. The maturity date of the September 22, 2021 Geneva Roth Note is September 22, 2022. The September
22, 2021 Geneva Roth Note bears interest at a rate of 8% per annum, which interest may be paid by the Company to Geneva Roth in shares
of the Company’s common stock; but shall not be payable until the September 22, 2021 Geneva Roth Note becomes payable, whether
at the maturity date or upon acceleration or by prepayment.
During
the first 60 to 180 days following the date of these notes, the Company has the right to prepay the principal and accrued but unpaid
interest due under the above notes issued to Geneva Roth, together with any other amounts that the Company may owe the holder under the
terms of the note, at a premium ranging from 110% to 129% as defined in the note agreement. After this initial 180-day period, the Company
does not have a right to prepay such notes.
The
conversion price for the above Geneva Roth notes shall be equal to a 35% discount of the market price based on the average of the lowest
three trading prices of the Common Stock for the ten trading days immediately prior to the delivery of a Notice of Conversion. Notwithstanding
the foregoing, Geneva Roth shall be restricted from effecting a conversion if such conversion, along with other shares of the Company’s
common stock beneficially owned by Geneva Roth and its affiliates, exceeds 9.99% of the outstanding shares of the Company’s common
stock. These notes are treated as stock settled debt under ASC 480 and accordingly the Company recorded a total of $161,269 put premium
for the four notes.
The
above Geneva Roth notes contain certain events of default, upon which principal and accrued interest will become immediately due and
payable. In addition, upon an event of default, interest on the outstanding principal shall accrue at a default interest rate of 22%
per annum, or if such rate is usurious or not permitted by current law, then at the highest rate of interest permitted by law. Further,
certain events of default may trigger penalty and liquidated damage provisions.
The
total principal amounts outstanding under the above Geneva Roth financing agreements were $132,000 and accrued interest of $3,477 as
of June 30, 2021 following conversion of $78,000 of the principal balance and $3,120 accrued interest during the year ended June 30,
2021. Accordingly, $42,000 of the put premium was released in respect of the Geneva Roth financing agreements during the year ended June
30, 2021 following conversion of the principal balance.
The
total principal amounts outstanding under the above Geneva Roth financing agreements were $167,500 and accrued interest of $1,081 as
of September 30, 2021 following conversion of $132,000 of the principal balance and $5,280 accrued interest during the three months ended
September 30, 2021. Accordingly, $71,077 of the put premium was released in respect of the Geneva Roth financing agreements during the
three months ended September 30, 2021 following conversion of the principal balance.
Amortization
of debt discounts
The
Company recorded $7,500 and $0 of debt discounts (including warrants, derivatives, debt issue costs and original issue discounts) related
to the above note issuances during the three months ended September 30, 2021 and 2020, respectively. The Company recorded $90,192 and
$0 of put premiums related to the above note issuances during the three months ended September 30, 2021 and 2020, respectively. The debt
discounts are being amortized over the term of the debt and the put premiums are expensed on issuance of the debt with the liability
released to additional paid in capital on conversion of the principal.
Amortization
of all debt discounts for the three months ended September 30, 2021 and 2020 was $6,074 and $121,281, respectively.
The
Company reclassified $109,643 and $204,919 in put premiums to additional paid in capital following conversions during the three months
ended September 30, 2021 and 2020, respectively.
NOTE
7 – STOCKHOLDERS’ DEFICIT
Increase
in Authorized Shares of Common Stock and Reverse Stock Split
On
February 4, 2020 the Directors resolved to increase the Common Stock of the Company from 100,000,000 authorized shares to 1,000,000,000
authorized shares and believes that such number of authorized shares of Common Stock will be in the best interests of the Corporation
and its stockholders because the Board believes that the availability of more shares of Common Stock for issuance will allow the Corporation
greater flexibility in pursuing financing from investors, meeting business needs as they arise, taking advantage of favorable opportunities
and responding to a changing corporate environment. The Company filed the necessary documents with the U.S. Securities and Exchange Commission
on February 6, 2020 and with the amendment to the authorized shares being approved by the State of Delaware on March 13, 2020.
On
November 17, 2020, the Company effected a one-for-one thousand (1:1,000) reverse stock split of the Company’s issued and outstanding
shares of common stock (the “Reverse Stock Split”). Proportional adjustments for the Reverse Stock Split were made to the
Company’s outstanding stock options, warrants and equity incentive plans. All share and per-share data and amounts have been retroactively
adjusted as of the earliest period presented in the consolidated financial statements to reflect the Reverse Stock Split.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
Preferred
Stock
The
total number of shares of preferred stock that the Company is authorized to issue is 1,500,005, $0.01 par value per share. These preferred
shares have no rights to dividends, profit sharing or liquidation preferences.
Of
the total preferred shares authorized, 500,000 have been designated as Series A Preferred Stock (“Series A Preferred Stock”),
pursuant to the Certificate of Designation filed with the Secretary of State of the State of Delaware on December 9, 2014. James Nathanielsz,
the Company’s Chief Executive Officer and Chief Financial Officer, beneficially owns all of the outstanding shares of Series A
Preferred Stock via North Horizon Pty Ltd., which entitles him, as a holder of Series A Preferred Stock, to vote on all matters submitted
or required to be submitted to a vote of the Company’s stockholders, except election and removal of directors, and each share of
Series A Preferred Stock entitles him to two votes per share of Series A Preferred Stock. North Horizon Pty Ltd. is a Nathanielsz Family
Trust. Mr. James Nathanielsz, the Chief Executive Officer, Chief Financial Officer and a director of our Company, has voting and investment
power over these shares. 500,000 shares of Series A Preferred Stock are issued and outstanding as of September 30, 2021 and June 30,
2021.
Of
the total preferred shares authorized, pursuant to the Certificate of Designation filed with the Secretary of State of the State of Delaware
on June 16, 2015, up to five shares have been designated as Series B Preferred Stock (“Series B Preferred Stock”). Each holder
of outstanding shares of Series B Preferred Stock is entitled to voting power equivalent to the number of votes equal to the total number
of shares of common stock outstanding as of the record date for the determination of stockholders entitled to vote at each meeting of
stockholders of the Company and entitled to vote on all matters submitted or required to be submitted to a vote of the stockholders of
the Company. One share of Series B Preferred Stock is issued and outstanding as of September 30, 2021 and June 30, 2021. Mr. Nathanielsz
directly beneficially owns such one share of Series B Preferred Stock.
No
additional shares of Series A Preferred Stock or Series B Preferred Stock were issued during the three months ended September 30, 2021
and fiscal year 2021.
Common
Stock:
Shares
issued for conversion of convertible debt
From
July 1, 2021 through September 30, 2021, the Company issued an aggregate of 9,445,009 shares of its common stock at an average contractual
conversion price of $0.02, ranging from $0.02 to $0.04, as a result of the conversion of principal of $189,849, interest of $8,087 and
conversion fees $2,250 underlying certain outstanding convertible notes converted during such period. The total recorded to equity was
$200,186.
The
Company reclassified $109,643, net of reversal of put premium upon cancellation of conversion notices by two lenders discussed above,
to additional paid in capital following conversions during the three months ended September 30, 2021.
The
Company has 197,308,116 shares of its common stock reserved for future issuances based on lender reserve requirements pursuant to underlying
financing agreements at September 30, 2021.
Shares
issued for services and accrued expenses
On
August 12, 2021, the Board approved the issuance of 2,800,000 shares of the Company’s common stock for bonus payable of $84,000
as of June 30, 2021 to an employee who is the wife of the CEO of the Company. The 2,800,000 shares of common stock were valued
at approximately $0.03 per share or $87,920, being the closing price of the stock on the date of grant. The shares were issued on August
17, 2021. The Company recorded stock-based compensation of $3,920 during the three months ended September 30, 2021 and reclassified bonus
payable of $84,000 to additional paid in capital upon issuance.
On
August 12, 2021, the Board approved the issuance of 166,667
shares of the Company’s common stock for
legal services rendered for the month of August 2021. The 166,667 shares of common stock were valued at approximately $0.05
per share or $7,883,
being the closing price of the stock on August 31, 2021, the date of grant. The shares were issued on September 3, 2021. The Company
recorded stock-based compensation of $7,883
during the three months ended September 30, 2021.
In
September 2021, the Company issued 2,819,712 shares of the Company’s common stock to a consultant for services rendered from July
2021 to September 2021. The Company issued 2,819,712 shares of the Company’s common stock valued at approximately $0.04 per share
or $104,611, being the closing price of the stock on the date of grant to such consultant. The Company recorded stock-based compensation
of $104,611 during the three months ended September 30, 2021.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
Nathanielsz
Cancellation Agreement
On
August 12, 2021, the Company entered into a Cancellation Agreement with James Nathanielsz (“Nathanielsz”), Chief Executive
Officer and Director of the Company, whereby Nathanielsz agreed to cancel his cash compensation bonus award for fiscal year 2021, ended
June 30, 2021, in exchange for common stock of the Company. The Company and Nathanielsz entered into an Amended and Restated Employment
Agreement dated May 14, 2019 (the “Agreement”). Pursuant to the terms of the Agreement, Nathanielsz was eligible to earn
an annual fiscal year cash performance bonus for each fiscal year of his employment period with the Company with a target performance
bonus of 200% of his average annualized base salary during the fiscal year for which the performance bonus is earned. On July 20, 2021,
Nathanielsz was awarded a “target” bonus of 78%, or $177,840 USD (the “Debt”) for the fiscal year ended June
30, 2021, by the Company’s Board of Directors (the “Board”). Pursuant to the Cancellation Agreement, Nathanielsz agreed
to cancel this Debt in exchange for 5,928,000 shares of the common stock of the Company (the “Shares”), valued at approximately
$0.03 per share or $186,139, being the closing price of the stock on the date of grant. The shares were issued on August 17, 2021. The
Company recorded stock-based compensation of $8,299 during the three months ended September 30, 2021 and reclassified bonus payable of
$177,840 to additional paid in capital upon issuance.
Kenyon
Cancellation Agreement
On
August 12, 2021, the Company entered into a Cancellation Agreement with Dr. Julian Kenyon (“Kenyon”), Chief Scientific Officer
and Director of the Company, whereby Kenyon agreed to cancel of $102,600 USD of accrued salary due him as of June 30, 2021, pursuant
to that certain Amended and Restated Services Agreement by and between Kenyon and the Company, dated May 14, 2019, in exchange for 3,420,000
shares of common stock of the Company (the “Shares”), valued at approximately $0.03 per share or $107,388, being the closing
price of the stock on the date of grant. The shares were issued on August 17, 2021. The Company recorded stock-based compensation of
$4,788 during the three months ended September 30, 2021 and reclassified accrued expenses of $102,600 to additional paid in capital upon
issuance.
Zelinger
Amended and Restated Director Agreement
On
August 12, 2021, the Company entered into an Amended and Restated Director Agreement (the “Director Agreement”) with Josef
Zelinger (“Zelinger”). Pursuant
to the terms of the Director Agreement, the Company shall pay Zelinger a base salary of $250.00 AUD ($184 USD) per month, payable
on the first day of each month. In addition, the Company may compensate Zelinger additional consideration for advisory services performed
by the Director, either in the form of cash or common stock, at the discretion of the Board. The Company issued 2,800,000 shares of common
stock of the Company for accrued director services of $84,000 as of June 30, 2021. The
2,800,000 shares of common stock were valued at approximately $0.03 per share $87,920, being the closing price of the stock on
the date of grant. The shares were issued on August 17, 2021. The shares were issued on August 17, 2021. The Company recorded stock-based
compensation of $3,920 during the three months ended September 30, 2021 and reclassified accrued expenses of $84,000 to additional paid
in capital upon issuance.
Shares
issued for exercise of warrants
From
July 9, 2021 through September 27, 2021, the Company received aggregate gross proceeds of $275,000 and subscription receivable of $100,000
from the exercise of 9,375 Series B Warrants and issued 6,875 shares of common stock and 2,500 shares of common stock issuable
as of September 30, 2021.
During
the three months ended September 30, 2021, additionally, the Company issued 2,399,988 shares of common stock and 1,999,990 shares
of common stock issuable from the alternate cashless exercise of 22 Series A warrants. The Company recognized the value of the effect
of a down round feature in such warrants when triggered. Upon the occurrence of the triggering event that resulted in a reduction of
the strike price, the Company measured the value of the effect of the feature as the difference between the fair value of the warrants
without the down round feature or before the strike price reduction and the fair value of the warrants with a strike price corresponding
to the reduced strike price upon the down round feature being triggered. Accordingly, the Company recognized deemed dividend of $114,844
and a corresponding reduction of income available to common stockholders upon the alternate cashless exercise of these warrants.
A
total of 2,002,490 common stock issuable were issued in October 2021. The Company collected the $100,000 subscription receivable in October
2021.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2021
(Unaudited)
Warrants:
The
following table summarizes warrant activity for the three months ended September 30, 2021:
SCHEDULE
OF WARRANT ACTIVITY
| | |
| | |
Weighted | |
| | |
Number of | | |
Average | |
| | |
Shares | | |
Price
Per Share | |
| Outstanding at June 30, 2021 | |
| 121,329 | | |
$ | 179.63 | |
| Issued | |
| - | | |
| - | |
| Exercised | |
| (9,397 | ) | |
| 40.37 | |
| Forfeited | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Outstanding at September 30, 2021 | |
| 111,932 | | |
$ | 191.32 | |
| | |
| | | |
| | |
| Exercisable at September 30, 2021 | |
| 76,933 | | |
$ | 278.36 | |
| Outstanding and Exercisable: | |
| | | |
| | |
| | |
| | | |
| | |
| Weighted average remaining contractual
term | |
| 1.52 | | |
| | |
Aggregate intrinsic
value
| |
$ | - | | |
| | |
No
stock warrants were granted during the three months ended September 30, 2021.
Options:
A
summary of the Company’s option activity during the three months ended September 30, 2021 is presented below:
SCHEDULE
OF STOCK OPTION ACTIVITY
| | |
| | |
Weighted | |
| | |
Number of | | |
Average Exercise | |
| | |
Shares | | |
Price
Per Share | |
| Outstanding at June 30, 2021 | |
| 59 | | |
$ | 13,730 | |
| Issued | |
| - | | |
| - | |
| Exercised | |
| - | | |
| - | |
| Forfeited | |
| - | | |
| - | |
| Expired | |
| - | | |
| - | |
| Outstanding at September 30, 2021 | |
| 59 | | |
$ | 4,533.33 | |
| | |
| | | |
| | |
| Exercisable at September 30, 2021 | |
| 39 | | |
$ | 4,530.93 | |
| Outstanding and Exercisable: | |
| | | |
| | |
| | |
| | | |
| | |
| Weighted average remaining contractual
term | |
| 7.62 | | |
| | |
| Weighted average fair value of options
granted during the period | |
$ | - | | |
| | |
| Aggregate intrinsic value | |
$ | - | | |
| | |
During
the three months ended September 30, 2021 and 2020, the Company recognized stock-based compensation of $20,718 and $20,718, respectively
related to vested stock options. There was $51,796 of unvested stock options expense as of September 30, 2021 that will be recognized
through May 2022 or 0.62 years.
No
stock options were granted during the three months ended September 30, 2021.
NOTE
8 – COMMITMENTS AND CONTINGENCIES
Legal
Matters
From
time to time, the Company may be subject to litigation and claims arising in the ordinary course of business. The Company is not currently
a party to any material legal proceedings and the Company is not aware of any pending or threatened legal proceeding against the Company
that we believe could have a material adverse effect on our business, operating results, cash flows or financial condition.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER
30, 2021
(Unaudited)
IRS
Liability
As
part of its requirement for having a foreign operating subsidiary, the Company’s parent U.S. entity is required to file an informational
Form 5471 to the Internal Revenue Service (the “IRS”), which is a form that explains the nature of the relationship between
the foreign subsidiary and the parent company. From 2012 through the 2014, the Company did not file this form in a timely manner. As
a result of the non-timely filings, the Company incurred a penalty from the IRS in the amount of $10,000 per year, or $30,000 in total,
plus accrued interest, such penalty and interest having been accrued and is included in the accrued expenses and other payable figure
in the September 30, 2021 and June 30, 2021 consolidated balance sheet. The Company recorded the penalties for all three years during
the year ended June 30, 2018. The Company is current on all subsequent filings. The Company’s tax advisor is awaiting a response
from the IRS on this matter.
Operating
Agreements
In
November 2009, the Company entered into a commercialization agreement with the University of Bath (UK) (the “University”)
whereby the Company and the University co-owned the intellectual property relating to the Company’s pro-enzyme formulations. In
June 2012, the Company and the University entered into an assignment and amendment whereby the Company assumed full ownership of the
intellectual property while agreeing to pay royalties of 2% of net revenues to the University. Additionally, the Company agreed to pay
5% of each and every license agreement subscribed for. The contract is cancellable at any time by either party. To date, no amounts are
owed under the agreement.
Collaboration
Agreement
On
September 13, 2018, the Company entered into a two-year collaboration agreement with the University of Jaén (the “University”)
to provide certain research services to the Company. In consideration of such services, the Company agreed to pay the University approximately
52,000 Euros ($59,508 USD) in year one and a maximum of 40,000 Euros ($45,775 USD) in year two. The Company paid 31,754 Euros ($36,117
USD) in 2019 and has accrued 28,493 Euros ($24,043 USD) as of June 30, 2021. Additionally, in exchange for full ownership of the intellectual
property the Company agreed to pay royalties of 2% of net revenues to the University. On October 1, 2020, the Company entered into another
two-year collaboration agreement with the University of Jaén to provide certain research services to the Company. In consideration
of such services, the Company agreed to pay the University approximately 30,000 Euros ($35,145 USD) which shall be paid in four installment
payment of 5,000 Euros in November 2020, 5,000 Euros ($5,858) in March 2021, 10,000 Euros ($11,715) in December 2021 and 10,000 Euros
($11,715) in September 2022. Additionally, the University shall hire and train a doctoral student for this project and as such the Company
shall pay the University 25,837 Euros ($30,268 USD). In exchange for full ownership of the intellectual property the Company agreed to
pay royalties of 2% of net revenues to the University.
NOTE 9 – RELATED PARTY TRANSACTIONS
Since
its inception, the Company has conducted transactions with its directors and entities related to such directors. These transactions have
included the following:
As
of September 30, 2021 and June 30, 2021, the Company owed its former director a total of $53,384 and $55,500, respectively, for money
loaned to the Company throughout the years. The total loans balance owed at September 30, 2021 and June 30, 2021 is not interest bearing
(See Note 5 – Loans and Notes Payable).
As
of September 30, 2021 and June 30, 2021, the Company owed its former director a total of $32,076 and $33,347, respectively, related to
expenses paid on behalf of the Company related to corporate startup costs and intellectual property (See Note 4 – Due to Former
Director – Related Party).
On
May 6, 2021, the Company entered into an agreement for the lease of its principal executive offices with North Horizon Pty Ltd., a related
party, of which Mr. Nathanielsz, our CEO, CFO and a director, and his wife are owners and directors. The lease has a one-year term commencing
May 6, 2021, and the Company is currently obligated to pay $3,606 AUD or $2,431 USD (depending on exchange rate), inclusive of tax, in
rent per month. During the three months ended September 30, 2021 and 2020, rent expense amounted $7,735 USD and $9,204 USD. As of September
30, 2021, total rent payable of $84,000AUD ($60,598 USD) is included in accrued expenses in the accompanying condensed consolidated balance
sheet.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER
30, 2021
(Unaudited)
Employment
and Services Agreements with Management
The
Company and Mr. Nathanielsz entered into an employment agreement as of February 25, 2015 (the “Nathanielsz Employment Agreement”)
setting forth the terms and conditions of Mr. Nathanielsz employment as the Company’s President and Chief Executive Officer. The
Nathanielsz Employment Agreement was scheduled to expire on February 25, 2019; however, the term of the Nathanielsz Employment Agreement
automatically renews for successive one-year periods unless either party provides 30 days’ prior written notice of its intent not
to renew. The Nathanielsz Employment Agreement continues in effect as of September 30, 2021 as amended May 14, 2019 (see below). The
Nathanielsz Employment Agreement provides Mr. Nathanielsz with a base salary of $25,000 AUD per month ($300,000 AUD annually or $205,680
USD) and a monthly contribution to Mr. Nathanielsz’s pension equal to 9.5% of his monthly salary. Mr. Nathanielsz has the ability
to convert any accrued but unpaid salary into common stock at the end of each fiscal year at a conversion price to be determined by Mr.
Nathanielsz and the Company, which will in no event be lower than par value or higher than the closing bid price on the date of conversion.
Pursuant to the Nathanielsz Employment Agreement, Mr. Nathanielsz is entitled to an annual discretionary bonus in an amount up to 200%
of his annual base salary, which bonus shall be determined by the Company’s board of directors based upon the performance of the
Company. On March 16, 2018, the Company’s board of directors approved an increase of Mr. Nathanielsz’s annual base salary
from $300,000 AUD ($205,680 USD) to $400,000 AUD ($274,240 USD), effective February 2018.
Mr.
Nathanielsz’s wife, Sylvia Nathanielsz, is and has been a non-executive part-time employee of the Company since October 2015. Effective
February 1, 2018, Mrs. Nathanielsz receives an annual salary of $120,000 AUD ($80,904 USD) and is entitled to customary benefits.
Pursuant
to a February 25, 2016 board resolution, James Nathanielsz shall be paid $4,481 AUD ($3,205 USD), on a monthly basis for the purpose
of acquiring and maintaining an automobile. For the three months ended September 30, 2021, a total of $$7,689 AUD ($5,651 USD) in payments
have been made with respect to Mr. Nathanielsz’s car allowance.
Pursuant to the approval of the Company’s board of directors, on May 14, 2019, Mr. Nathanielsz was granted a $460,000 AUD ($315,376 USD) bonus for accomplishments achieved while serving as the Company’s Chief Executive Officer during the fiscal year ended June 30, 2019 with $200,000 AUD ($137,120 USD) of such bonus payable by the Corporation to the CEO throughout the Corporation’s 2019 fiscal
year as the Corporation’s cash resources allow, with the remaining $260,000 AUD ($178,256 USD) of such bonus to be deferred by the CEO until a future date when the Corporation’s cash resources allow for such payment, as agreed to by the CEO. A total of $221,890
AUD ($166,418 USD) in payments were made against the bonuses during the year ended June 30, 2021 resulting in a remaining balance of $422,610 AUD ($316,957 USD) bonus payable as of June 30, 2021. On August 12, 2021, the Board approved a bonus of $177,840 USD. On August 12, 2021, pursuant to the Cancellation Agreement, Mr. Nathanielsz agreed to cancel $177,840 of the bonus payable in exchange for 5,928,000 shares of the common stock of the Company (see Note 7). A total of $$42,500 AUD ($30,660 USD) in payments were made against the bonuses during the three months ended September 30, 2021 which resulted to a remaining balance of $142,990 AUD ($103,153 USD) bonus payable as of September 30, 2021 which is included in accrued expenses in the accompanying condensed consolidated balance sheet.
Amended
and Restated Employment Agreement - On May 14, 2019 (the “Effective Date”), the Company entered into an Amended and Restated
Employment Agreement (the “Employment Agreement”) with James Nathanielsz, the Company’s Chief Executive Officer, Chairman,
acting Chief Financial Officer and a director, for a term of three years, subject to automatic one-year renewals, at an annual salary
of $400,000 AUD. Pursuant to the Employment Agreement, Mr. Nathanielsz was granted options to purchase 39 shares of the Company’s
common stock (the “Nathanielsz Options”), with an exercise price per share of $4,675 (110% of the closing market price of
the Company’s common stock on May 14, 2019 (or $4,250), the date of approval of such grant by the Company’s board of directors),
(ii) 39 restricted stock units of the Company (the “Initial Nathanielsz RSUs”), and (iii) an additional 39 restricted stock
units of the Company (the “Additional Nathanielsz RSUs”). Such options and restricted stock units were granted pursuant to
the 2019 Plan approved by the Company’s board of directors on the Effective Date. The Nathanielsz Options have a term of 10 years
from the date of grant. 1/3rd of the Nathanielsz Options shall vest every successive one-year anniversary following the Effective Date,
provided, that on each such vesting date Mr. Nathanielsz is employed by the Company and subject to the other provisions of the Employment
Agreement. The Initial Nathanielsz RSUs shall vest on the one-year anniversary of the Effective Date, subject to Mr. Nathanielsz’s
continued employment with the Company through such vesting date. The Additional Nathanielsz RSUs will vest as follows, subject to Mr.
Nathanielsz’s continued employment with the Company through the applicable vesting date: (i) 7.80 of the Additional Nathanielsz
RSUs shall vest upon the Company submitting Clinical Trial Application (the “CTA”) for PRP, the Company’s lead product
candidate (“PRP”), for a First-In-Human study for PRP (the “Study”) in an applicable jurisdiction to be selected
by the Company, (ii) 7.80 of the Additional Nathanielsz RSUs shall vest upon the CTA being approved in an applicable jurisdiction, (iii)
7.80 of the Additional RSUs shall vest upon the Company completing an equity financing in the amount of at least $4,000,000 in gross
proceeds, (iv) 7.80 of the Additional Nathanielsz RSUs shall vest upon the shares of the Company’s Common Stock being listed on
a senior stock exchange (NYSE, NYSEMKT or NASDAQ), and (v) the remaining 7.80 of the Additional Nathanielsz RSUs shall vest upon the
Company enrolling its first patient in the Study. Each vested restricted stock unit shall be settled by delivery to Mr. Nathanielsz of
one share of the Company’s common stock and/or the fair market value of one share of common stock in cash, at the sole discretion
of the Company’s board of directors and subject to the 2019 Plan, on the first to occur of: (i) the date of a Change of Control
(as defined in the Employment Agreement), (ii) the date that is ten business days following the vesting of such restricted stock unit,
(iii) the date of Mr. Nathanielsz’s death or Disability (as defined in the Employment Agreement), and (iv) Mr. Nathanielsz’s
employment being terminated either by the Company without Cause or by Mr. Nathanielsz for Good Reason (each as defined in the Employment
Agreement). In the event of a Change of Control, any unvested portion of the Nathanielsz Options and such restricted stock units shall
vest immediately prior to such event. The 39 vested restricted stock unit are considered issuable as of September 30, 2021.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER
30, 2021
(Unaudited)
Amended
and Restated Services Agreement - On May 14, 2019, the Company also entered into an Amended and Restated Services Agreement (the “Services
Agreement”) with Dr. Kenyon, the Company’s Chief Scientific Officer and a director, for a term of three years, subject to
automatic one-year renewals, at an annual salary of $54,000 AUD. In connection with the execution of the Services Agreement, Dr. Kenyon
was designated as an executive officer of the Company and assumed a more active executive role with the Company. Pursuant to the Services
Agreement, Dr. Kenyon was granted options to purchase 20 shares of the Company’s common stock (the “Kenyon Options”),
with an exercise price per share of $4,250 (100% of the closing market price of the Company’s common stock on May 14, 2019, the
date of approval of such grant by the Company’s board of directors), (ii) 20 restricted stock units of the Company (the “Initial
Kenyon RSUs”), and (iii) an additional 20 restricted stock units of the Company (the “Additional Kenyon RSUs”). Such
options and restricted stock units were granted pursuant to the 2019 Plan approved by the Company’s board of directors on the Effective
Date. The Kenyon Options have a term of 10 years from the date of grant. 1/3rd of the Kenyon Options shall vest every successive one-year
anniversary following the Effective Date, provided, that on each such vesting date Dr. Kenyon is employed by the Company and subject
to the other provisions of the Services Agreement. The Initial Kenyon RSUs shall vest on the one-year anniversary of the Effective Date,
subject to Dr. Kenyon’s continued employment with the Company through such vesting date. The Additional Kenyon RSUs will vest as
follows, subject to Dr. Kenyon’s continued employment with the Company through the applicable vesting date: (i) 5 of the Additional
Kenyon RSUs shall vest upon the Company submitting the CTA for PRP for the Study in an applicable jurisdiction to be selected by the
Company, (ii) 5 of the Additional Kenyon RSUs shall vest upon the Company completing an equity financing in the amount of at least $4,000,000
in gross proceeds, (iii) 5 of the Additional Kenyon RSUs shall vest upon the shares of the Company’s Common Stock being listed
on a senior stock exchange (NYSE, NYSEMKT or NASDAQ), and (iv) the remaining 5 of the Additional Kenyon RSUs shall vest upon the Company
enrolling its first patient in the Study. Each vested Kenyon RSU shall be settled by delivery to Mr. Kenyon of one share of the Company’s
common stock and/or the fair market value of one share of common stock in cash, at the sole discretion of the Company’s board of
directors and subject to the Plan, on the first to occur of: (i) the date of a Change of Control (as defined in the Services Agreement),
(ii) the date that is ten business days following the vesting of such Kenyon RSU, (iii) the date of Dr. Kenyon’s death or Disability
(as defined in the Services Agreement), and (iv) Dr. Kenyon’s employment being terminated either by the Company without Cause or
by Dr. Kenyon for Good Reason (as defined in the Services Agreement). In the event of a Change of Control (as defined in the Services
Agreement), 50% of any unvested portion of the Kenyon Options and the Kenyon RSUs shall vest immediately prior to such event. The 20
vested restricted stock unit are considered issuable as of September 30, 2021. As of June 30, 2021, total accrued salaries of $135,000
AUD ($101,250 USD) was included in accrued expenses. On August 12, 2021, pursuant to the Cancellation Agreement, Mr. Kenyon agreed to
cancel accrued salaries of $102,600 in exchange for 3,420,000 shares of the common stock of the Company (see Note 7). As of September
30, 2021, total accrued salaries of $13,500 AUD ($9,739 USD) was included in accrued expenses in the accompanying condensed consolidated
balance sheet.
Intercompany
Loans
All
Intercompany loans were made by the parent to the subsidiary, Propanc PTY LTD, which have not been repaid as of September 30,
2021. Effective fiscal year 2021, the parent company determined that intercompany loans will not be repaid in the foreseeable future
and thus, per ASC 830-20-35-3, gains and losses from measuring the intercompany balances are recorded within cumulative translation adjustment,
a component of other comprehensive income.
NOTE
10 – CONCENTRATIONS AND RISKS
Concentration
of Credit Risk
The
Company maintains its cash in banks and financial institutions in Australia. Bank deposits in Australian banks are uninsured. The Company
has not experienced any losses in such accounts through September 30, 2021.
The
Company primarily relied on funding from one convertible debt lender and received proceeds after deductions of $7,500 for original issue
discounts and debt issue costs during the three months ended September 30, 2021 from a lender of $160,000 which represents approximately
100% of total proceeds received by the Company during the three months ended September 30, 2021.
The
Company did not receive any funding from lenders during the three months ended September 30, 2020.
Receivable
Concentration
As
of September 30, 2021 and June 30, 2021, the Company’s receivables were 100% related to reimbursements on GST taxes paid.
Patent
and Patent Concentration
The
Company has filed multiple patent applications relating to its lead product, PRP. The Company’s lead patent application has been
granted and remains in force in the United States, Belgium, Czech Republic, Denmark, France, Germany, Ireland, Italy, Netherlands, Portugal,
Spain, Sweden, Switzerland, Liechtenstein, Turkey, United Kingdom, Australia, China, Japan, Indonesia, Israel, New Zealand, Singapore,
Malaysia, South Africa, Mexico, Republic of Korea, India and Brazil. In Canada, the patent application remains under examination.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER
30, 2021
(Unaudited)
In
2016 and early 2017, we filed other patent applications. Three applications were filed under the Patent Cooperation Treaty (the “PCT”).
The PCT assists applicants in seeking patent protection by filing one international patent application under the PCT, applicants can
simultaneously seek protection for an invention in over 150 countries. Once filed, the application is placed under the control of the
national or regional patent offices, as applicable, in what is called the national phase. One of the PCT applications filed in November
2016, entered national phase in July 2018 and another PCT application is currently entering national phase in August 2018. A third PCT
application entered the national phase in October 2018.
In
July 2020, a world first patent was granted in Australia for the cancer treatment method patent family. Presently, there are 31 granted
patents and 34 patents under examination in key global jurisdictions relating to the use of proenzymes against solid tumors, covering
the lead product candidate PRP.
Further
patent applications are expected to be filed to capture and protect additional patentable subject matter based on the Company’s
field of technology relating to pharmaceutical compositions of proenzymes for treating cancer.
Foreign
Operations
As
of September 30, 2021 and June 30, 2021, the Company’s operations are based in Camberwell, Australia, however the majority of research
and development is being conducted in the European Union.
On
July 22, 2016, the Company formed a wholly owned subsidiary, Propanc (UK) Limited under the laws of England and Wales for the purpose
of submitting an orphan drug application with the European Medicines Agency as a small and medium-sized enterprise. As of September 30,
2021 and June 30, 2021, there has been no activity within this entity.
NOTE
11 - DERIVATIVE FINANCIAL INSTRUMENTS AND FAIR VALUE MEASUREMENTS
Derivative
Financial Instruments:
The
Company applies the provisions of ASC 815-40, Contracts in Entity’s Own Equity, under which convertible instruments and
warrants, which contain terms that protect holders from declines in the stock price (reset provisions), may not be exempt from derivative
accounting treatment. As a result, warrants and embedded conversion options in convertible debt are recorded as a liability and are revalued
at fair value at each reporting date. If the fair value of the warrants exceeds the face value of the related debt, the excess is recorded
as change in fair value in operations on the issuance date. The Company had $80,000 (1 note) of convertible debt, which is treated as
derivative instruments outstanding at September 30, 2021 and June 30, 2021.
The
Company calculates the estimated fair values of the liabilities for derivative instruments using the Binomial Trees Method. The closing
price of the Company’s common stock at September 30, 2021, the last trading day of the period ended September 30, 2021, was $0.026.
The Volatility, expected remaining term and risk-free interest rates used to estimate the fair value of derivative liabilities at September
30, 2021 are indicated in the table that follows. The expected term is equal to the remaining term of the warrants or convertible instruments
and the risk-free rate is based upon rates for treasury securities with the same term.
Convertible
Debt
SCHEDULE
OF FAIR VALUE MEASUREMENTS, RECURRING AND NONRECURRING, VALUATION TECHNIQUES
| | |
Initial
Valuations
(on new derivative
instruments entered into
during the three
months ended
September 30, 2021) | | |
September
30, 2021 | |
| Volatility | |
| - | | |
| 206.00 | % |
| Expected Remaining Term (in years) | |
| - | | |
| 0.01 | |
| Risk Free Interest Rate | |
| - | | |
| 0.07 | % |
| Expected dividend yield | |
| None | | |
| None | |
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER
30, 2021
(Unaudited)
Fair
Value Measurements:
The
Company measures and reports at fair value the liability for derivative instruments. The fair value liabilities for price adjustable
warrants and embedded conversion options have been recorded as determined utilizing the Binomial Trees model. The following tables summarize
the Company’s financial assets and liabilities measured at fair value on a recurring basis as of September 30, 2021 and June 30,
2021:
SCHEDULE
OF FAIR VALUE, ASSETS AND LIABILITIES MEASURED ON RECURRING BASIS
| | |
Balance
at
September 30, 2021 | | |
Quoted
Prices in
Active Markets
for Identical
Assets | | |
Significant Other Observable
Inputs | | |
Significant Unobservable
Inputs | |
| | |
| | |
(Level 1) | | |
(Level 2) | | |
(Level 3) | |
| Embedded
conversion option liabilities | |
$ | 58,124 | | |
$ | — | | |
$ | — | | |
$ | 58,124 | |
| Total | |
$ | 58,124 | | |
$ | — | | |
$ | — | | |
$ | 58,124 | |
| | |
Balance
at
June 30, 2021 | | |
Quoted
Prices in
Active Markets
for Identical
Assets | | |
Significant Other Observable
Inputs | | |
Significant Unobservable
Inputs | |
| | |
| | |
(Level 1) | | |
(Level 2) | | |
(Level 3) | |
| Embedded
conversion option liabilities | |
$ | 54,220 | | |
$ | — | | |
$ | — | | |
$ | 54,220 | |
| Total | |
$ | 54,220 | | |
$ | — | | |
$ | — | | |
$ | 54,220 | |
The
following is a roll forward for the three months ended September 30, 2021 of the fair value liability of price adjustable derivative
instruments:
SCHEDULE
OF DERIVATIVE LIABILITIES AT FAIR VALUE
| | |
Fair Value
of | |
| | |
Liability
for | |
| | |
Derivative | |
| | |
Instruments | |
| Balance at June 30, 2021 | |
$ | 54,220 | |
| Change in fair value
included in statements of operations | |
| 3,904 | |
| Balance at September
30, 2021 | |
$ | 58,124 | |
NOTE
12 – SUBSEQUENT EVENTS
Exercise
of Warrants
In
October 2021, the Company issued 2,199,989 shares of common stock from the alternate cashless exercise of 11 Series A warrants.
Note
Conversions
In
October 2021, the Company issued an aggregate of 1,818,097 shares of its common stock at an average contractual conversion price of $0.01,
as a result of the conversion of principal of $25,000, interest of $1,726 and conversion fees $0 underlying certain outstanding convertible
notes converted during such period. The Company reclassified $16,667 in put premiums to additional paid in capital following these conversions.
Common
Stock Issuable
The
2,002,490 shares of common stock issuable as of September 30, 2021 were issued in October 2021.
Consulting
Agreement
On
October 1, 2021, the Company entered int a consulting agreement (the “Consulting Agreement”) with a consultant who will
assist in the development of the Company’s business and financing activities. The consultant will serve initially as an
independent contractor, and upon certain mutually agreed upon conditions being met, will be appointed Vice Chairman, President and
Interim CFO. The term of the Consulting Agreement shall be for three years commencing on October 1, 2021, and can be terminated by
either party upon 30 day written notice. The monthly payment per the Consulting Agreement is $7,000. The Company will also issue
shares of common stock equal to 1% of the total issued and outstanding shares at the end of each year of service.
PROPANC
BIOPHARMA, INC. AND SUBSIDIARY
NOTES
TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER
30, 2021
(Unaudited)
October
21, 2021 Securities Purchase Agreement
Effective
October 21, 2021, the Company entered into a securities purchase agreement with Sixth Street Lending LLC (“Sixth Street”),
pursuant to which Sixth Street purchased a convertible promissory note (the “October 21, 2021 Sixth Street”) from the Company
in the aggregate principal amount of $63,750, such principal and the interest thereon convertible into shares of the Company’s
common stock at the option of Sixth Street any time after the six-month anniversary of the October 21, 2021 Sixth Street. The October
21, 2021 Sixth Street contains an original discount of $3,750. The Company intends to use the net proceeds from the October 21, 2021
Sixth Street for general working capital purposes. The maturity date of the October 21, 2021 Sixth Street Note is October 21, 2022. The
October 21, 2021 Sixth Street Note bears interest at a rate of 8% per annum, which interest may be paid by the Company to Sixth Street
in shares of the Company’s common stock; but shall not be payable until the October 21, 2021 Sixth Street Note becomes payable,
whether at the maturity date or upon acceleration or by prepayment.
During
the first 60 to 180 days following the date of these notes, the Company has the right to prepay the principal and accrued but unpaid
interest due under the above notes issued to Sixth Street, together with any other amounts that the Company may owe the holder under
the terms of the note, at a premium ranging from 110% to 129% as defined in the note agreement. After this initial 180-day period, the
Company does not have a right to prepay such notes.
The
conversion price for the above Sixth Street notes shall be equal to a 35% discount of the market price which means the average of the
lowest three trading prices of the Common Stock for the ten trading days immediately prior to the delivery of a Notice of Conversion.
Notwithstanding the foregoing, Sixth Street shall be restricted from effecting a conversion if such conversion, along with other shares
of the Company’s common stock beneficially owned by Sixth Street and its affiliates, exceeds 9.99% of the outstanding shares of
the Company’s common stock. These notes are treated as stock settled debt under ASC 480 and accordingly the Company recorded a
total of $34,327 put premium.
The
above Sixth Street notes contain certain events of default, upon which principal and accrued interest will become immediately due and
payable. In addition, upon an event of default, interest on the outstanding principal shall accrue at a default interest rate of 22%
per annum, or if such rate is usurious or not permitted by current law, then at the highest rate of interest permitted by law. Further,
certain events of default may trigger penalty and liquidated damage provisions.
MANAGEMENT’S
DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The
following discussion and analysis of the results of operations and financial condition for the years ended June 30, 2021 and 2020, and
for the three month period ended September 30, 2021 should be read in conjunction with our consolidated financial statements and the
notes to those consolidated financial statements that are included elsewhere in this Registration Statement. Our discussion includes
forward-looking statements based upon current expectations that involve risks and uncertainties, such as our plans, objectives, expectations
and intentions. Actual results and the timing of events could differ materially from those anticipated in these forward-looking statements
as a result of a number of factors. See “Forward-Looking Statements.”
Management’s
discussion and analysis of results of operations and financial condition (“MD&A”) is a supplement to the accompanying
financial statements and provides additional information on Propanc Biopharma, Inc. (“Propanc” or the “Company’)
business, current developments, financial condition, cash flows and results of operations.
When
we say “we,” “us,” “our,” “Company,” or “Propanc,” we mean Propanc Biopharma,
Inc.
Overview
We
were incorporated in the state of Delaware as Propanc Health Group Corporation on November 23, 2010. In January 2011, to reorganize our
Company, we acquired all of the outstanding shares of Propanc PTY LTD, an Australian corporation, on a one-for-one basis and Propanc
PTY LTD became our wholly-owned subsidiary. Effective April 20, 2017, we changed our name to “Propanc Biopharma, Inc.” to
better reflect our current stage of operations and development.
We
are a development-stage healthcare company that is currently focused on developing new cancer treatments for patients suffering from
pancreatic, ovarian and colorectal cancer. Utilizing our scientific and oncology consultants, we have developed a rational, composite
formulation of anti-cancer compounds, which together exert a number of effects designed to control or prevent tumors from recurring and
spreading through the body. Our lead product candidate, PRP, is a variation upon our novel formulation and involves pro-enzymes, the
inactive precursors of enzymes.
Recent
Developments
On
October 1, 2020, the Company entered into a two-year collaboration agreement with the University of Jaén (the “University”)
to provide certain research services to the Company. In consideration of such services, the Company agreed to pay the University approximately
30,000 Euros ($35,145 USD) which shall be paid in four installment payments of 5,000 Euros in November 2020, 5,000 Euros ($5,858) in
March 2021, 10,000 Euros ($11,715) in December 2021 and 10,000 Euros ($11,715) in September 2022. Additionally, the University shall
hire and train a doctoral student for this project and as such the Company shall pay the University 25,837 Euros ($30,268 USD). In exchange
for full ownership of the intellectual property, the Company agreed to pay royalties of 2% of net revenues to the University.
On
May 11, 2021, the Company’s scientific researchers with the Universities of Jaén and Granada, published data in a peer reviewed
journal, Expert Opinion on Biological Therapy, confirming the anti-tumor potential of a mixture of two pancreatic proenzymes trypsinogen
and chymotrypsinogen. Treatment with proenzymes sensitizes cancer stem cells which may allow standard treatment approaches like chemotherapy
and radiotherapy to be more effective.
Between
August 2020 to November 2021, advancements were made with the Company’s patent portfolio including allowed claims describing a
pharmaceutical composition for treating cancer in Brazil and a divisional application covering additional claims describing a pharmaceutical
composition for treating cancer was granted in China (both covering the Company’s lead patent application), a first granted patent
describing a method to treat cancer stem cells in Australia, and granted patents in Australia, Indonesia and Singapore, citing higher
doses of a pharmaceutical composition. The advancements represent three of four patent families covering the Company’s lead product,
PRP.
In
November 2021, a Strategic Advisor to the Propanc Biopharma Executive team was appointed to initiate the process of establishing a wholly
owned, US based, R&D operating subsidiary, located in New Jersey. The Strategic Advisor will oversee establishment of the R&D
operating subsidiary and identify strategic partners to assist with financing and resourcing to advance PRP towards a First-In-Human
study for advanced cancer patients suffering from solid tumors.
On
November 26, 2021, the Company entered into a securities purchase agreement (the “Purchase Agreement”) with Sixth Street
Lending, LLC (“Sixth Street”), pursuant to which Sixth Street purchased a convertible promissory note (the “Note”)
from the Company in the aggregate principal amount of $53,750, such principal and the interest thereon convertible into shares of the
Company’s common stock at the option of Sixth Street. The transaction contemplated by the Purchase Agreement closed on or about
December 2, 2021. The Company intends to use the net proceeds ($50.000) from the Note for general working capital purposes.
On
November 30, 2021, the Company entered into a Common Stock Purchase Agreement (the “Purchase Agreement”) with Dutchess Capital
Growth Fund LP, a Delaware limited partnership, (“Dutchess”), providing for an equity financing facility (the “Equity
Line”). The Purchase Agreement provides that upon the terms and subject to the conditions in the Purchase Agreement, Dutchess is
committed to purchase up to Five Million Dollars ($5,000,000) of shares of common stock, $0.001 par value per share (the “Common
Stock”), over the 36 month term of the Purchase Agreement (the “Total Commitment”).
Under
the terms of the Purchase Agreement, Dutchess will not be obligated to purchase shares of Common Stock unless and until certain conditions
are met, including but not limited to a Registration Statement on Form S-1 (the “Registration Statement”) becoming effective
which registers Dutchess’ resale of any Common Stock purchased by Dutchess under the Equity Line. From time to time over the 36-month
term of the Purchase Agreement, commencing on the trading day immediately following the date on which the Registration Statement becomes
effective, the Company, in our sole discretion, may provide Dutchess with a draw down notice (each, a “Draw Down Notice”),
to purchase a specified number of shares of Common Stock (each, a “Draw Down Amount Requested”), subject to the limitations
discussed below. The actual amount of proceeds the Company will receive pursuant to each Draw Down Notice (each, a “Draw Down Amount”)
is to be determined by multiplying the Draw Down Amount Requested by the applicable purchase price. The purchase price of each share
of Common Stock equals 92% of the lowest trading price of the Common Stock during the five (5) business days prior to the Closing Date.
Closing Date shall mean the five (5) business days after the Clearing Date. Clearing Date shall mean the first business day that the
Selling Security Holder holds the Draw Down Amount in its brokerage account and is eligible to trade the shares.
The
maximum number of shares of Common Stock requested to be purchased pursuant to any single Draw Down Notice cannot exceed the lesser of
(i) 300% of the average daily share volume of the Common Stock in the five (5) trading days immediately preceding the Draw Down Notice
or (ii) an aggregate value of $250,000.
The
Company agreed to pay to Dutchess a commitment fee for entering into the Purchase Agreement of 1,000,000 restricted shares of our common
stock. The shares were issued December 10, 2021.
On
December 7, 2021, the Company entered into a securities purchase agreement (the “Purchase Agreement”) with ONE44 Capital
LLC, (“ONE44”), pursuant to which ONE44 purchased a convertible promissory note (the “Note”) from the Company
in the aggregate principal amount of $170,000, such principal and the interest thereon convertible into shares of the Company’s
common stock at the option of ONE44. The transaction contemplated by the Purchase Agreement closed on or about December 13, 2021. The
Company intends to use the net proceeds ($153,000) from the Note for general working capital purposes. The Note contains an original
issue discount amount of $17,000.
On
January 4, 2022, Propanc Biopharma, Inc. (the “Company”) entered into a securities purchase agreement (the “Purchase
Agreement”) with Sixth Street Lending, LLC (“Sixth Street”), pursuant to which Sixth Street purchased a convertible
promissory note from the Company in the aggregate principal amount of $63,750, such principal and the interest thereon convertible into
shares of the Company’s common stock at the option of Sixth Street. The transaction contemplated by the Purchase Agreement closed
on January 6, 2022. The Company intends to use the net proceeds ($60,000) from the Note for general working capital purposes.
Results
of Operations
The
following discussion should be read in conjunction with the Company’s consolidated financial statements and notes thereto
included elsewhere in this Registration Statement. The results discussed below are of the Company and its wholly-owned Australian subsidiary,
Propanc PTY LTD.
Three
months ended September 30, 2021, as compared to the Three months ended September 30, 2020
Revenue
For
the three and three months ended September 30, 2021 and 2020, we generated no revenue because we are currently undertaking research and
development activities for market approval and no sales were generated in this period.
Administration
Expense
Administration
expense increased to $431,740 for the three months ended September 30, 2021 as compared to $323,111 for the three months ended September
30, 2020. This decrease of approximately $109,000 is primarily attributable to an increase of approximately $133,000 in stock-based expenses
for services and increase in other general and administrative expenses of approximately $3,000, offset by decrease in general consulting,
legal and investor relation fees of approximately $1,000, decrease in accounting fees of approximately $13,000, and decrease in approximately
$13,000 in employee remuneration expense.
Occupancy
Expense
Occupancy
expense decreased by $1,468 to $7,736 for the three months ended September 30, 2021. The decrease primarily relates to exchange rate
movements over the period when compared to the same period in 2020.
Research
and Development Expenses
Research
and development expenses were $46,554 for the three months ended September 30, 2021, as compared to $50,846 for the three months ended
September 30, 2020, a decrease of approximately $4,000. The research and development expenses incurred are primarily attributable to
research and development expenses incurred in relation to the two-year collaboration agreement we entered with University of Jaén
in October 2020.
Interest
Expense/Income
Interest
expense decreased to $109,853 for the three months ended September 30, 2021, as compared to $159,281 for the three months ended September
30, 2020, respectively. Interest expense is primarily comprised of approximately $6,000 and $90,000 of debt discount amortization and
accretion of put premium for the three months ended September 30, 2021 and interest expense from conversion fees of $2,000 and accrual
of interest expense for approximately $11,000 for the three months ended September 30, 2021.
This
decrease is primarily attributable to a decrease in amortization of debt discount of approximately $115,000 for three months ended September
30, 2021, decrease in prepayment penalty fees of approximately $13,000 and decrease in conversion fees of $4,000 offset by increases
in accretion of put premium interest expense of approximately $90,000, and accrual of interest expense for a total of $7,000 for the
three months ended September 30, 2021.
Change
in Fair Value of Derivative Liabilities
Change
in fair value of derivative liabilities changed by $68,856, to a loss of $3,904 for the three months ended September 30, 2021, as compared
to a gain of $64,952 for the three months ended September 30, 2020. This change is primarily attributable to an increase in fair value
of the principal amount of a convertible note with bifurcated embedded conversion option derivatives during the three months ended September
30, 2021.
Gain
(loss) on Extinguishment of Debt, net
During
the three months ended September 30, 2020, notes were converted with principal amounts totaling $75,000 and accrued interest of $3,000
contained bifurcated embedded conversion option derivatives. Accordingly, the fair market value of the shares issued was $134,155 resulting
in a loss on extinguishment at the time of conversion of $56,155 and $106,140 of derivative fair value was recorded as a gain on extinguishment
at the time of conversion.
During
the three months ended September 30, 2021, there were no notes converted that contained bifurcated embedded conversion option derivatives.
Foreign
Currency Transaction Gain (Loss)
Foreign
currency transaction increased to a gain of $109,129 for the three months ended September 30, 2021 as compared with $1,960 for the three
months ended September 30, 2020.
The
foreign currency transaction decreased to a gain is partially attributable to the increase in exchange rates during the three months
ended September 30 2021, as compared to the three months ended September 30, 2020.
Net
loss
Net
loss increased to $490,658 for the three months ended September 30, 2021 as compared to a net loss of $425,545 for the three months ended
September 30, 2020. The change relates to the factors discussed above.
Deemed
dividend
The
Company recognized the value of the effect of a down round feature related to our Series A warrants when triggered. Upon the occurrence
of the triggering event that resulted in a reduction of the strike price, the Company measured the value of the effect of the feature
as the difference between the fair value of the warrants without the down round feature or before the strike price reduction and the
fair value of the warrants with a strike price corresponding to the reduced strike price upon the down round feature being triggered.
Accordingly, the Company recognized deemed dividend of $114,844 and a corresponding reduction of income available to common stockholders
upon the alternate cashless exercise of these warrants.
Net
loss available to common stockholders
Net
loss available to common stockholders increased to $605,502 for the three months ended September 30, 2021 as compared to a net loss available
to common stockholders of $425,545 for the three months ended September 30, 2020. The change relates to the factors discussed above.
Liquidity
and Capital Resources
Current
Financial Condition
As
of September 30, 2021, we had total assets of $62,165, comprised primarily of cash of $45,817, GST tax receivable of $2,238, prepaid
expenses and other current assets of $8,353, property and equipment, net, of $3,593 and security deposit of $2,164. As compared to June
30, 2021, we had total assets of $13,101, comprised primarily of cash of $2,255, GST tax receivable of $4,341, property and equipment,
net, of $4,255 and security deposit of $2,250.
We
had current liabilities of $2,368,795, primarily comprised of net convertible debt of $584,608, accounts payable and accrued expenses
of $1,233,959, employee benefit liability of $406,644, and embedded conversion option liabilities of $58,124 as of September 30, 2021.
As compared to June 30, 2021, 3,080,674, primarily comprised of net convertible debt of $624,583, accounts payable and accrued expenses
of $1,894,486, employee benefit liability of $418,538, and embedded conversion option liabilities of $54,220 as of June 30, 2021.
We
have funded our operations primarily through the issuance of equity and/or convertible securities for cash. The cash was used primarily
for payments for research and development, administration expenses, occupancy expenses, professional fees, consultants and travel.
During
the three months ended September 30, 2021 we received proceeds from exercise of warrants of $275,000 and net proceeds from issuance of
convertible notes of $160,000.
We
have substantial capital resource requirements and have incurred significant losses since inception. As of September 30, 2021, we had
$45,817 in cash. We depend upon debt and/or equity financing to fund our ongoing operations and to execute our current business plan.
Such capital requirements are in excess of what we have in available cash and for which we currently have commitments. Therefore, we
presently do not have enough available cash to meet our obligations over the next 12 months. If continued funding and capital resources
are unavailable at reasonable terms, we may curtail our plan of operations. We will be required to obtain alternative or additional financing
from financial institutions, investors or otherwise, in order to maintain and expand our existing operations. The failure by us to obtain
such financing would have a material adverse effect upon our business, financial condition and results of operations, and adversely affecting
our ability to complete ongoing activities in connection with our research and development programs.
Sources
and Uses of Cash
| | |
For the Three months ended
September 30, | |
| | |
2021 | | |
2020 | |
| Net cash used in operating activities | |
$ | (486,758 | ) | |
$ | (179,949 | ) |
| Net cash used in investing activities | |
$ | - | | |
$ | - | |
| Net cash provided by financing activities | |
$ | 435,000 | | |
$ | 158,044 | |
| Effect of exchange rate changes on cash | |
$ | 95,320 | | |
$ | 6,116 | |
Net
Cash Flow from Operating Activities
Net
cash used in operating activities was $486,758 for the three months ended September 30, 2021, due to our net loss of $490,658 offset
primarily by non-cash charges of amortization of debt discount of $6,074, stock-based compensation of $154,140 non-cash interest expense
of $2,250, accretion of put premium of $90,192, change in fair value of derivatives of $3,904 addback foreign currency transaction gain
of $109,129. Net changes in operating assets and liabilities totaled $144,040, which is primarily attributable to increase in prepaid
expense of $8,353, increase accrued interest of $11,338 offset by decrease in accounts payable of $137,927 and decrease in accrued expenses
of $15,102.
Net
cash used in operating activities was $179,949 for the three months ended September 30, 2020, due to our net loss of $425,545 offset
primarily by non-cash charges of amortization of debt discount of $121,281, stock-based compensation of $20,718, non-cash interest expense
of $6,750 addback $64,952 of change in fair value of derivatives and $49,985 gain on extinguishment of debt. Net changes in operating
assets and liabilities totaled $213,306, which is primarily attributable to increase in accounts payable of $53,576, employee benefit
liability of $10,544, accrued expenses of $133,046, and accrued interest of $16,262.
Net
Cash Flow from Financing Activities
Cash
flows provided by financing activities for the three months ended September 30, 2021 were $435,000 as compared to $158,044 for the three
months ended September 30, 2020. During the three months ended September 30, 2021 we received proceeds from the exercise of warrants
of $275,000 and net proceeds from issuance of convertible notes of $160,000. During the three months ended September 30, 2020 we received
proceeds from the exercise of warrants of $201,044 offset by repayments of convertible notes of $43,000.
Effect
of Exchange Rate
The
effect of the exchange rate on cash resulted in a $95,320 positive adjustment to cash flows in the three months ended September 30, 2021
as compared to an adjustment of $6,116 to cash flows in the three months ended September 30, 2020. The reason for the fluctuation is
due to the application of currency translation rates throughout the cash flow statement, the volume of transactions within each period
and the daily fluctuation in exchange rates
Going
Concern Qualification
We
did not generate any revenue for the three months ended September 30, 2021 and 2020 and have incurred significant losses and cash used
in operations, and such losses and use of cash are expected to continue. Our independent registered public accounting firm has included
a “Going Concern Qualification” in their audit report for each of the fiscal years ended June 30, 2021 and 2020. In addition,
we have negative working capital and convertible debt that is past maturity that we are currently negotiating with lenders in order to
amend the maturity dates. The foregoing raises substantial doubt about our ability to continue as a going concern for a period of 12
months from the issue date of this report. Our ability to continue as a going concern is dependent on our ability to execute our strategy
and on our ability to raise additional funds and/or to consummate a public offering. Management is currently seeking additional funds,
primarily through the issuance of equity and/or debt securities for cash to operate our business. No assurance can be given that any
future financing will be available or, if available, that it will be on terms that are satisfactory to us. Even if we are able to obtain
additional financing, it may contain undue restrictions on our operations, in the case of debt financing or cause substantial dilution
for our stockholders, in case of equity and/or convertible debt financing. The consolidated financial statements do not include any adjustments
that might result from the outcome of this uncertainty. The “Going Concern Qualification” might make it substantially more
difficult to raise capital.
Fiscal
Year Ended June 30, 2021, as compared to the Fiscal Year Ended June 30, 2020
Revenue
For
the fiscal years 2021 and 2020 we generated no revenue because we are currently undertaking research and development activities for market
approval and no sales were generated in this period.
Administration
Expense
Administration
expense decreased to $1,553,075 for the year ended June 30, 2021 as compared to $3,281,464 for the year ended June 30, 2020. This decrease
of approximately $1,728,000 is primarily attributable to a decrease of approximately $1,190,000 in stock-based expenses for services,
decrease of approximately $128,000 of indirect capital raising costs, decrease in general consulting, legal, and investor relation fees
of approximately $265,000, decrease in accounting fees of approximately $17,000, decrease in insurance expense of approximately $75,000,
decrease of approximately $124,000 in marketing and market research expense, decrease in travel expenses of approximately $28,000, decrease
of approximately $19,000 of other general and administrative expenses and offset by increase of approximately $118,000 in employee remuneration
expense.
Occupancy
Expense
Occupancy
expense decreased by approximately $4,700 to $28,112 for the year ended June 30, 2021. The decrease primarily relates to exchange rate
movements over the period when compared to the same period in 2020.
Research
and Development Expenses
Research
and development expenses were $230,956 for the year ended June 30, 2021, as compared to $179,987 for the year ended June 30, 2020. The
increase in research and development expenses is primarily attributable to the two-year collaboration agreement with University Jaén
which was executed in October 2020 to provide certain research services to the Company.
Interest
Expense/Income
Interest
expense decreased to $449,457 for the year ended June 30, 2021, as compared to $1,748,381 for the year ended June 30, 2020. Interest
expense is primarily comprised of approximately $337,000 of debt discount amortization and accretion of put premium for the year ended
June 30, 2021, and interest expense from conversion fees, prepayment penalty fees and accrual of interest expense for a total of approximately
$113,000 for the year ended June 30, 2021.
This
decrease of $1,298,924 is primarily attributable to a decrease in the issuance of convertible notes and was further reduced due to the
conversion and repayment of convertible notes during the year ended June 30, 2021 which resulted to a decrease in accretion of put premium
of approximately $636,000 during the year ended June 30, 2021, and the decrease in amortization of debt discount of approximately $598,000
during the year ended June 30, 2021. Additionally, interest expense from prepayment penalty fees decreased by approximately $99,000 offset
by increase in conversion fees and accrual of interest expense for a total of $34,000 during the year ended June 30, 2021.
Change
in Fair Value of Derivative Liabilities
Change
in fair value of derivative liabilities changed by $393,479, to a loss of $8,186 for the year ended June 30, 2021, as compared to a gain
of $385,293 for the year ended June 30, 2020. This change is primarily attributable to a decrease in the principal amount of convertible
notes with bifurcated embedded conversion option derivatives during the year ended June 30, 2021.
Gain
from Settlement of Debt, net
During
the year ended June 30, 2021, the Company recorded gain from settlement of debt, net of $49,319 relating to two transactions. On March
22, 2021, the Company entered into a settlement agreement with our former counsel, Foley Shechter, whereby both parties agreed to settle
all claims for professional fees owed for a total of $51,057. The Company paid the settlement amount of $51,057 on March 22, 2021. Prior
to the settlement agreement, the Company recorded total accounts payable and accrued expenses $143,614. Accordingly, the Company recognized
gain from settlement of debt of $92,557 during the year ended June 30, 2021.
Additionally,
on March 15, 2021, the Company entered into a Settlement and Mutual Release Agreement with Regal whereby both parties agreed to settle
all claims and liabilities under the August 10, 2017 Convertible note for a total of $100,000. All other terms of the August 10, 2017
Convertible Note shall remain in full force and effect. Both parties agree that all future penalties under this note are waived unless
the Company fails to authorize to distribute the requested shares upon conversion. The Company has the right to pay off the balance of
any remaining amounts dues under this note in cash at any time more than 60 days after March 15, 2021. Prior to the Settlement Agreement,
the Company recorded total liabilities $56,762 consisting of remaining principal amount of $8,500, accrued interest of $23,262 and accrued
expenses of $25,000. Accordingly, the Company recognized loss from settlement of debt of $43,238 during the year ended June 30, 2021.
Gain
(loss) on Extinguishment of Debt, net
During
the year ended June 30, 2020, notes with principal amounts totaling $254,500 and accrued interest of $15,408 contained bifurcated embedded
conversion option derivatives. Accordingly, the fair market value of the shares issued was $565,746 resulting in a loss on extinguishment
at the time of conversion of $295,838 and $362,961 of derivative fair value was recorded as a gain on extinguishment at the time of conversion.
During
the year ended June 30, 2021, notes with principal amounts totaling $95,000 and accrued interest of $3,000 contained bifurcated embedded
conversion option derivatives. Accordingly, the fair market value of the shares issued was $178,368 resulting in a loss on extinguishment
at the time of conversion of $80,368 and $130,975 of derivative fair value was recorded as a gain on extinguishment at the time of conversion,
resulting in a net gain of $50,607.
Foreign
Currency Transaction Gain (Loss)
Foreign
currency transaction changed to a gain of $30,497 for the year ended June 30, 2021 as compared with a loss of $143,808 for the year ended
June 30, 2020.
The
foreign currency transaction decreased to a gain is partially attributable to the increase in exchange rates during the year ended June
30 2021, as compared to the year ended June 30, 2020.
Benefit
(provision) for taxes
During
the year ended June 30, 2021 and 2020, the Company applied for and received from the Australian Taxation Office a research and development
tax credit in the amount of $113,415 and $134,728, respectively.
Net
loss
Net
loss decreased to $2,025,947 for the year ended June 30, 2021 as compared to a net loss of $4,740,723 for the year ended June 30, 2020.
The change relates to the factors discussed above.
Deemed
dividend
The
Company recognized the value of the effect of a down round feature related to our Series A and C warrants when triggered. Upon the occurrence
of the triggering event that resulted in a reduction of the strike price, the Company measured the value of the effect of the feature
as the difference between the fair value of the warrants without the down round feature or before the strike price reduction and the
fair value of the warrants with a strike price corresponding to the reduced strike price upon the down round feature being triggered.
Accordingly, the Company recognized deemed dividend of $391,749 and a corresponding reduction of income available to common stockholders
upon the alternate cashless exercise of these warrants.
Net
loss available to common stockholders
Net
loss available to common stockholders decreased to $2,417,696 for the year ended June 30, 2021 as compared to a net loss of $4,740,723
for the year ended June 30, 2020. The change relates to the factors discussed above.
Liquidity
and Capital Resources
Current
Financial Condition
As
of June 30, 2021, we had total assets of $13,101, comprised primarily of cash of $2,255, GST tax receivable of $4,341, property and equipment,
net, of $4,255 and security deposit of $2,250. As compared to June 30, 2020, we had total assets of $98,518, comprised primarily of cash
of $67,007, GST tax receivable of $2,015, property and equipment, net, of $5,747 and operating lease right of use asset, net, $21,682.
We
had current liabilities of $3,080,674, primarily comprised of net convertible debt of $624,583, accounts payable and accrued expenses
of $1,894,486, employee benefit liability of $418,538, and embedded conversion option liabilities of $54,220 as of June 30, 2021. As
compared to June 30, 2020, we had current liabilities of $3,739,943, primarily comprised of net convertible debt of $1,557,734, accounts
payable and accrued expenses of $1,544,387, employee benefit liability of $354,109, and embedded conversion option liabilities of $354,109.
We
have funded our operations primarily through the issuance of equity and/or convertible securities for cash. The cash was used primarily
for payments for research and development, administration expenses, occupancy expenses, professional fees, consultants and travel.
During
the year ended June 30, 2021 we received proceeds from exercise of warrants of $776,044 and proceeds from issuance of convertible notes
of $325,000.
We
have substantial capital resource requirements and have incurred significant losses since inception. As of June 30, 2021, we had $2,255
in cash. We depend upon debt and/or equity financing to fund our ongoing operations and to execute our current business plan. Such capital
requirements are in excess of what we have in available cash and for which we currently have commitments. Therefore, we presently do
not have enough available cash to meet our obligations over the next 12 months. If continued funding and capital resources are unavailable
at reasonable terms, we may curtail our plan of operations. We will be required to obtain alternative or additional financing from financial
institutions, investors or otherwise, in order to maintain and expand our existing operations. The failure by us to obtain such financing
would have a material adverse effect upon our business, financial condition and results of operations, and adversely affecting our ability
to complete ongoing activities in connection with our research and development programs.
Sources
and Uses of Cash
| | |
For the years ended June 30, | |
| | |
2021 | | |
2020 | |
| Net cash used in operating activities | |
$ | (1,145,264 | ) | |
$ | (1,849,589 | ) |
| Net cash provided by financing activities | |
$ | 1,058,044 | | |
$ | 1,890,240 | |
| Effect of exchange rate changes on cash | |
$ | 22,468 | | |
$ | 23,962 | |
Net
Cash Flow from Operating Activities
Net
cash used in operating activities was $1,145,264 for the year ended June 30, 2021, due to our net loss of $2,025,947, offset primarily
by non-cash charges of amortization of debt discount of $136,527, stock-based compensation of $208,444, non-cash interest expense of
$16,500, accretion of put premium of $200,410, change in fair value of derivatives of $8,186 addback foreign currency transaction gain
of $30,497, gain from settlement of debt of $48,390 and $50,607 gain on extinguishment of debt. Net changes in operating assets and liabilities
totaled $92,277, which is primarily attributable to increase in accounts payable of $177,382, increase in accrued expenses and other
payables of $152,861, employee benefit liability of $33,134, and accrued interest of $80,582.
Net
cash used in operating activities was $1,849,589 for the year ended June 30, 2020, due to our net loss of $4,740,723 offset primarily
by non-cash charges of amortization of debt discount of $734,130, stock-based compensation of $1,398,868, accretion of put premium of
$836,724, non-cash interest expense of $15,000, foreign currency transaction loss of $143,808 addback gain on extinguishment of debt
of $67,123 and $385,293 of change in fair value of derivatives. Net changes in operating assets and liabilities totaled $212,547, which
is primarily attributable to decrease in prepaid expenses and other assets of $83,157, increase in employee benefit liability of $35,724,
and accrued interest of $156,417 and offset by decrease in accounts payable of $29,737 and accrued expenses and other payables of $9,740.
Net
Cash Flow from Financing Activities
Cash
flows provided by financing activities for the year ended June 30, 2021 were $1,058,044 as compared to $1,890,240 for the year ended
June 30, 2020. During the year ended June 30, 2021 we received proceeds from the exercise of warrants of $776,044 and proceeds from issuance
of convertible notes of $325,000 offset by repayments of convertible notes of $43,000. During the year ended June 30, 2020, we received
proceeds from the sale of convertible promissory notes of $1,465,250 and sale of common stock of $450,000, net of $25,010 in issue costs.
Effect
of Exchange Rate
The
effect of the exchange rate on cash resulted in a $22,468 positive adjustment to cash flows in the year ended June 30, 2021 as compared
to an adjustment of $23,962 to cash flows in the year ended June 30, 2020. The reason for the fluctuation is due to the application of
currency translation rates throughout the cash flow statement, the volume of transactions within each period and the daily fluctuation
in exchange rates.
Critical
Accounting Estimates
Below
is a discussion of our more subjective accounting estimation processes for purposes of explaining (i) the methodology used in calculating
the estimates, (ii) the inherent uncertainties pertaining to such estimates, and (iii) the possible effects of a significant variance
in actual experience, from that of the estimate, on our financial condition. Estimates involve numerous assumptions that, if incorrect,
could create a material adverse impact on the Company’s results of operations and financial condition.
Reference
is frequently made herein to the Financial Accounting Standards Board (the “FASB”) Accounting Standards Codification (“ASC”).
This is the source of authoritative US GAAP recognized by the FASB to be applied to non-governmental entities. Each ASC reference in
this filing is presented with a three-digit number, which represents its Topic. As necessary for explanation and as applicable, an ASC
topic may be followed with a two-digit subtopic, a two-digit section or a two-or-three-digit paragraph.
Foreign
Currency Translation and Comprehensive Income (Loss): The Company’s wholly owned subsidiary’s functional currency is
the AUD. For financial reporting purposes, the Australian Dollar (“AUD”) has been translated into USD as the Company’s
reporting currency. Assets and liabilities are translated at the exchange rate in effect at the balance sheet date. Revenues and expenses
are translated at the average rate of exchange prevailing during the reporting period. Equity transactions are translated at each historical
transaction date spot rate. Translation adjustments arising from the use of different exchange rates from period to period are included
as a component of stockholders’ equity (deficit) as “accumulated other comprehensive income (loss).” Gains and losses
resulting from foreign currency transactions are included in the statement of operations and comprehensive loss as other income (expense).
Effective fiscal year 2021, the parent company determined that intercompany loans will not be repaid in the foreseeable future and thus,
per ASC 830-20-35-3, gains and losses from measuring the intercompany balances are recorded within cumulative translation adjustment,
a component of other comprehensive income.
Accounting
for Income Taxes: We are governed by Australian and United States income tax laws, which are administered by the Australian Taxation
Office and the United States Internal Revenue Service, respectively. We follow ASC 740, “Accounting for Income Taxes,”
which requires an asset and liability approach to financial accounting and reporting for income taxes. Deferred income tax assets
and liabilities are computed annually for temporary differences between the financial statements and tax bases of assets and liabilities
that will result in taxable or deductible amounts in the future based on enacted tax laws and rates applicable to the periods in which
the differences are expected to affect taxable income. Valuation allowances are established when necessary, to reduce deferred tax assets
to the amount expected to be realized. Income tax expense is the tax payable or refundable for the period plus or minus the change during
the period in deferred tax assets and liabilities.
The
Company adopted provisions of ASC 740, Sections 25 through 60, “Accounting for Uncertainty in Income Taxes.” These
sections provide detailed guidance for the financial statement recognition, measurement and disclosure of uncertain tax positions recognized
in the financial statements. Tax positions must meet a “more-likely-than-not” recognition threshold at the effective date
to be recognized upon the adoption of ASC 740 and in subsequent periods.
Accounting
for Stock Based Compensation: We record stock-based compensation in accordance with ASC 718, “Stock Compensation”
and Staff Accounting Bulletin No. 107 issued by the SEC in March 2005 regarding its interpretation of ASC 718. ASC 718 requires the fair
value of all stock-based employee compensation awarded to employees to be recorded as an expense over the related requisite service period.
The statement also requires the recognition of compensation expense for the fair value of any unvested stock option awards outstanding
at the date of adoption. We value any employee or non-employee stock-based compensation at fair value using the Black-Scholes Option
Pricing Model.
We
account for non-employee share-based awards in accordance with the measurement and recognition criteria of ASC 718.
Derivative
Instruments: ASC 815, “Derivatives and Hedging,” establishes accounting and reporting standards for derivative
instruments and for hedging activities by requiring that all derivatives be recognized in the balance sheet and measured at fair value.
Gains or losses resulting from changes in the fair value of derivatives are recognized in earnings. On the date of conversion, or payoff,
of debt, we record the fair value of the conversion shares, remove the fair value of the related derivative liability, remove any discounts
and record a net gain or loss on debt extinguishment.
Convertible
Notes with Variable Conversion Options: We have entered into convertible notes, some of which contain variable conversion options,
whereby the outstanding principal and accrued interest may be converted, by the holder, into common shares at or around a fixed discount
to the price of the common stock at the time of conversion. We treat these convertible notes as stock settled debt under ASC 480 and
measure the fair value of the notes at the time of issuance, which is the result of the share price discount at the time of conversion
and record the put premium as accretion to interest expense.
Research
and Development Tax Credits: We may apply for Research and Development tax concessions with the Australian Taxation Office on an
annual basis. Although the amount is possible to estimate at year end, the Australian Taxation Office may reject or materially alter
the claim amount. Accordingly, we do not recognize the benefit of the claim amount until cash receipt since collectability is not certain
until such time. The tax concession is a refundable credit. If we have net income then we can receive the credit which reduces its income
tax liability. If we have net losses, then we may still receive a cash payment for the credit, however, our net operating loss carry
forwards are reduced by the gross equivalent loss that would produce the credit amount when the income tax rate is applied to that gross
amount. The concession is recognized as an income tax benefit, in operations, upon receipt.
Leases:
In February 2016, the Financial Accounting Standards Board (“FASB”) issued ASU 2016-02, Leases (Topic 842). The
updated guidance requires lessees to recognize lease assets and lease liabilities for most operating leases. In addition, the updated
guidance requires that lessors separate lease and non-lease components in a contract in accordance with the new revenue guidance in ASC
606. This guidance is effective for interim and annual reporting periods beginning after December 15, 2018. The Company adopted this
guidance effective July 1, 2019.
On
July 1, 2019, the Company adopted ASU No. 2016-02, applying the package of practical expedients to leases that commenced before the effective
date whereby the Company elected to not reassess the following: (i) whether any expired or existing contracts contain leases and; (ii)
initial direct costs for any existing leases. For contracts entered into on or after the effective date, at the inception of a contract
the Company assessed whether the contract is, or contains, a lease. The Company’s assessment is based on: (1) whether the contract
involves the use of a distinct identified asset, (2) whether we obtain the right to substantially all the economic benefit from the use
of the asset throughout the period, and (3) whether it has the right to direct the use of the asset. The Company will allocate the consideration
in the contract to each lease component based on its relative stand-alone price to determine the lease payments.
Operating
lease ROU assets represents the right to use the leased asset for the lease term and operating lease liabilities are recognized based
on the present value of future minimum lease payments over the lease term at commencement date. As most leases do not provide an implicit
rate, the Company use an incremental borrowing rate based on the information available at the adoption date in determining the present
value of future payments. Lease expense for minimum lease payments is amortized on a straight-line basis over the lease term and is included
in general and administrative expenses in the consolidated statements of operations.
Going
Concern Qualification
The
accompanying consolidated financial statements have been prepared in conformity with US GAAP, which contemplate continuation of the Company
as a going concern. For the fiscal year ended June 30, 2021, the Company had no revenues, had a net loss of $2,025,947 and had net cash
used in operations of $1,145,264. Additionally, as of June 30, 2021, the Company had a working capital deficit, stockholders’ deficit
and accumulated deficit of $3,074,078, $3,067,573, and $58,199,466, respectively.
Our
independent registered public accounting firm has included a “Going Concern Qualification” in their audit report for each
of the fiscal years ended June 30, 2021 and 2020. In addition, we have negative working capital and convertible debt that is past maturity
that we are currently negotiating with lenders in order to amend the maturity dates. The foregoing raises substantial doubt about our
ability to continue as a going concern for a period of 12 months from the issue date of this report. Our ability to continue as a going
concern is dependent on our ability to execute our strategy and on our ability to raise additional funds and/or to consummate a public
offering. Management is currently seeking additional funds, primarily through the issuance of equity and/or debt securities for cash
to operate our business. No assurance can be given that any future financing will be available or, if available, that it will be on terms
that are satisfactory to us. Even if we are able to obtain additional financing, it may contain undue restrictions on our operations,
in the case of debt financing or cause substantial dilution for our stockholders, in case of equity and/or convertible debt financing.
The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty. The “Going
Concern Qualification” might make it substantially more difficult to raise capital.
Off-Balance
Sheet Arrangements
We
do not have any off-balance sheet arrangements that have or are reasonably likely to have a current or future effect on our financial
condition, changes in financial condition, revenues or expenses, results of operations, liquidity, capital expenditures or capital resources.
DIRECTORS,
EXECUTIVE OFFICERS AND KEY EMPLOYEES
The
following table sets forth certain information regarding our current executive officers and directors as of February 2, 2022:
| Name |
|
Age |
|
Position |
| James
Nathanielsz |
|
47 |
|
Chief
Executive Officer, Chief Financial Officer, and Director |
| Dr.
Julian Kenyon |
|
74 |
|
Director |
| Josef
Zelinger |
|
70 |
|
Independent
Director |
James
Nathanielsz has served as Chief Executive Officer and director of our Company since inception, and has served as our Chief Financial
Officer since December, 2020. He also has served as a director and Chief Executive Officer of our Australian subsidiary since October
2007. From July 2006 until October 2007, Mr. Nathanielsz served as the New Products Manager of Biota Holdings Limited, an anti- infective
drug development company in Australia.
Mr.
Nathanielsz graduated with a Bachelor of Applied Science, majoring in Biochemistry/Applied Chemistry and with a Master of Entrepreneurship
& Innovation from Swinburne University of Technology in Melbourne, Australia.
Our
board of directors has concluded that Mr. Nathanielsz is well-qualified to serve on our board of directors and has the requisite qualifications,
skills and perspectives based on, among other factors, him being a Co-Founder of our Australian company and for his experience in research
and development and manufacturing and distribution, as well as him being our controlling stockholder, and his significant business, investment,
finance and public company experience, particularly with biotech companies.
Dr.
Julian Kenyon has served as a director of our Company since inception. Dr. Kenyon co-founded our Australian subsidiary and was appointed
as a director of our Australian subsidiary on February 12, 2008. Since 2000, Dr. Kenyon has served as an integrated medical physician
and Medical Director of the Dove Clinic for Integrated Medicine in Winchester and London.
Dr.
Kenyon graduated from the University of Liverpool with a Bachelor of Medicine and Surgery and with a research degree, Doctor of Medicine.
Since 1972, he was appointed a Primary Fellow of the Royal College of Surgeons, Edinburgh.
Our
board of directors has concluded that Dr. Kenyon is well-qualified to serve on our board of directors and has the requisite qualifications,
skills and perspectives based on, among other factors, him being a Co-Founder of our Australian subsidiary and because our business is
based on his initial work at the Dove Clinic.
Josef
Zelinger has served as a director of the Company since December, 2020. He is a Certified Practicing Accountant with 45 years of experience
in tax, auditing, finance, investment and management consulting. Mr. Zelinger also has significant expertise in property management and
import/export businesses. Mr. Zelinger commenced his career as an accountant at L.M. Stanton & Partners - Chartered Accountants,
subsequently joining Caston Pty Ltd in 1980, a steel manufacturer as Chief Financial Officer, and company director, until 1983.
Since
the mid-1980’s until current date, Mr. Zelinger serves as director in several private investment companies in a range of businesses
including property portfolio manager of commercial real estate, import/export businesses and a range of commercial and financial investment
companies. Since 1980, Mr. Zelinger also operates as a sole practitioner in accountancy and tax consulting.
In
1973, Mr. Zelinger graduated in Accounting and was admitted as a Fellow of RMIT University in Business.
Our
board of directors has concluded that Mr. Zelinger is well-qualified to serve on our board of directors and has the requisite qualifications,
skills and perspectives based upon his professional experience.
Term
of Office
Our
directors are appointed for a one-year term to hold office until the next annual general meeting of our stockholders or until removed
from office in accordance with our Bylaws and the provisions of the Delaware General Corporation Law. Our directors hold office after
the expiration of his or her term until his or her successor is elected and qualified, or until his or her resignation, death or removal
in accordance with our Bylaws or the Delaware General Corporation Law.
Our
officers are appointed by our board of directors and hold office until removed by our board of directors at any time for any reason.
Family
Relationships
There
are no family relationships between or among any of our directors or executive officers or persons nominated or chosen by us to become
directors or executive officers.
Director
Independence
Our
board of directors has reviewed the independence of our directors and has determined that Josef Zelinger qualifies as an independent
director pursuant to Rule 5605(a)(2) of Nasdaq and applicable SEC rules and regulations. In making this determination, our board of directors
considered the relationships that each of our directors has with us and all other facts and circumstances our board of directors deemed
relevant in determining their independence.
Board
Committees
Our
board of directors has no separately designated committees and our board members carry out the functions of both an audit committee and
a compensation committee. We do not have an audit committee financial expert serving on our board of directors. Due to our limited financial
resources, we are not in a position to retain an independent director with the qualifications to serve as an audit committee financial
expert at this time.
Scientific
Advisory Board
We
have a Scientific Advisory Board that provides advice to our management relating to the following:
| |
● |
The
identification, assessment, evaluation, selection, conduct and management of research projects, both those which are under review
and are in progress; |
| |
● |
Intellectual
property; and |
| |
● |
Commercialization. |
The
Scientific Advisory Board may also address issues related to improving project selection, formal review processes and management procedures
within our Company. The Scientific Advisory Board will generally be composed of an advisory panel of clinicians with expertise in translational
research.
As
of February 2, 2022, the members of our Scientific Advisory Board were:
| |
● |
Professor
Klaus Kutz (also serving as our acting Chief Medical Officer); |
| |
|
|
| |
● |
Professor
Macarena Perán; |
| |
|
|
| |
● |
Professor
Juan Antonio Marchal Corrales; |
| |
|
|
| |
● |
Dr.
Maria Garcia; and |
| |
|
|
| |
● |
Dr.
Ralf Brandt. |
Each
of the members of our Scientific Advisory Board acts as an independent consultant and is compensated on an hourly basis for his or her
services. There is presently no stock based compensation for their services. In addition, we may have relationships with entities with
which the members may be associated.
Professor
Kutz is also acting as Chief Medical Officer for Propanc in a non-executive capacity. His compensation continues to be based on an hourly
rate as per his Advisory Board Agreement. Propanc intends to appoint Professor Kutz as Chief Medical Officer of Propanc in a full-time
executive officer capacity at a time that is mutually agreed upon between both parties.
Professor
Klaus Kutz has over 20 years of experience as an independent consultant in Clinical Pharmacology and Safety for pharmaceutical companies
and clinical research organizations. His specialty over the last six years is Oncology, including preparation of multiple NDAs and INDs
for small and medium sized pharmaceutical companies. He has prepared, organized and reported clinical Phase I studies in oncology and
Phase II studies in different cancer indications (prostate, gastric, ovarian, small cell lung cancer) and Non-Hodgkin Lymphomas. Professor
Kutz has more than 13 years of experience as Head of Clinical Pharmacology with world-wide responsibilities for Phase I and Clinical
Pharmacokinetics in two internationally operating pharmaceutical companies, setting up and restructuring international Clinical Pharmacology
departments. His achievements include the successful world-wide registration of multiple important Sandoz’ compounds by preparation
of multiple NDAs (New Drug Applications) and Expert reports (including Written Summary), as well as the preparation of multiple INDs
(Investigational New Drug Applications) for Sandoz Pharma Ltd and Sanofi Research. He is a specialist for Internal Medicine, Gastroenterology,
and Clinical Pharmacology and he is also Professor of Medicine at the University of Bonn, Germany.
Professor
Macarena Perán holds a B.S. in Biology and an M.S. in Biochemistry and Molecular Biology from the University of Málaga,
Spain. Dr. Perán moved to the Neuroscience Department at Durham University, UK, where she studied the Cellular Distribution and
Immobilization of GABAA Receptors on the cell membrane and graduated in 2000 with a Ph.D. She moved back to Spain and completed another
Ph.D. program in the Faculty of Medicine focused on Changes in the Behavior of Central Nervous Proteins; she completed a second Ph.D.
from Granada University. In 2005/2006, she attended Bath University, UK, Prof. David Tosh lab, and changed her research interest to the
development of new anti-cancer drugs and cell therapy for regenerative medicine. In 2011, she spent a year as a visiting scientist in
the Salk Institute for Biological Studies, California, Prof. Juan Carlos Izpisua-Belmonte lab. Currently, Dr. Perán is Reader
in Anatomy at University of Jaén in Spain and is working with the Institute for Regenerative Medicine and Pathobiology (IBIMER).
Professor
Juan Antonio Marchal Corrales is Professor of Anatomy and Embryology at the Faculty of Medicine of University of Granada. He graduated
in Medicine and Surgery in 1992, obtaining the degree “summa cum laude”. He defended his doctoral thesis in 1996. Prof. Marchal
has worked at three universities in different educational categories and is responsible for the research group “Differentiation,
Regeneration and Cancer”. He has participated in 39 research projects of national and international character, being principal
investigator in 13 of them. He has a total of 145 publications in journals, of which 125 are listed in the Journal Citation Reports.
He has spent time at the University of Sassari (Italy) and as visiting professor. He is inventor of 14 patents, 4 of them licensed. He
is a member of the Advisory Board of the International Graduate School of the University of Granada, member of the standing committee
of the Scientific Council and coordinator of Area Research in the Biosanitary Institute of Granada (ibs.GRANADA) and member of the Governing
Board at the Institute of Pathobiology and Regenerative Medicine (IBIMER). He has recently been named director of the Chair Drs. Galera
and Requena of Cancer Stem Cell Research at the University of Granada.
Dr.
Maria Garcia graduated in Biology from University of Granada (Spain) in 1997, became a Molecular Biologist working in the National
Centre of Biotechnology characterizing the mechanism of action of “Protein kinase induced by interferon: PKR”. These studies
gave rise to a PhD title awarded with an Extraordinary Thesis Award by the Autonomous University of Madrid in 2004. In 2002, Dr. García
completed a 3-months stay at the University of Wyoming with Dr. Roth. During the postdoctoral period, she got major public and private
funding to characterize new activity of the main tumor suppressor genes that are mutated in more than 50% of human cancers such as p53,
ARF and Rb. Dr. García currently has a competitive research contract from the National Health System to lead translational cancer
research, aiming at the integration of basic, clinical and epidemiological cancer research in the University Hospital Complex of Granada.
She leads a line of research involving new antitumor drugs, biological therapies, biomarkers and cancer stem cell studies. Finally, Dr.
García has more than 30 peer-reviewed publications in international journals with an average impact factor of 5 and a H-Index
of 14.
Dr.
Ralf Brandt is the co-founder of vivoPharm, a global oncology and immuno-oncology discovery services company providing a range of
preclinical services, which merged and became a part of Cancer Genetics, Inc., a Nasdaq listed company enabling precision medicine in
oncology from bench to bedside. Dr. Brandt currently serves as President of Discovery and Early Development of Cancer Genetics. Dr. Brandt
is a biochemist and cell biologist with over 15 years of experience in research programs of experimental oncology. He has immense experience
in in vivo pharmacology and anti-cancer drug profiling. Dr. Brandt received his License (BSc in Biochemistry and Animal Physiology) in
1986, and his PhD (in Biochemistry) in 1991 from the Martin-Luther University of Halle-Wittenberg, Germany. Dr. Brandt was employed at
research positions at the National Cancer Institute in Bethesda, MD, USA and at Schering AG, Germany. Since 1990, Dr. Brandt has been
active in the field of preclinical oncology. He led the Tumor Biology program at Novartis Pharma AG, Switzerland and established several
transgenic mouse lines developing tumors under the control of oncogenes. During Dr. Brandt’s long career in the pharmaceutical
industry he has acquired significant knowledge and expertise in leading business units and representation of services to the pre-clinical
research market. Dr. Brandt is also a member of the Scientific Advisory Board at Receptor Inc. in Toronto Canada.
Risk
Oversight
Our
board of directors will oversee a company-wide approach to risk management. Our board of directors will determine the appropriate risk
level for us generally, assess the specific risks faced by us and review the steps taken by management to manage those risks. While our
board of directors will have ultimate oversight responsibility for the risk management process, its committees will oversee risk in certain
specified areas.
Until
we have established our compensation committee of our board of directors, our board of directors will be responsible for overseeing the
management of risks relating to our executive compensation plans and arrangements, and the incentives created by the compensation awards
it administers. Until we have established our audit committee, our board of directors will oversee management of enterprise risks and
financial risks, as well as potential conflicts of interests. Our board of directors will be responsible for overseeing the management
of risks associated with the independence of our board of directors.
Code
of Ethics
The
Board has adopted a Code of Ethics (the “Code”) to apply to all of our directors, officers and employees. The Code is intended
to promote ethical conduct and compliance with laws and regulations, to provide guidance with respect to the handling of ethical issues,
to implement mechanisms to report unethical conduct, to foster a culture of honesty and accountability, to deter wrongdoing and to ensure
fair and accurate financial reporting. A copy of the Code is available at our website www.propanc.com.
Compensation
Committee Interlocks and Insider Participation
None
of our executive officers currently serves, or in the past three years has served, as a member of the board of directors or compensation
committee of another entity that has one or more executive officers serving on our board of directors or the compensation committee.
No member of our compensation committee has any other business relationship or affiliation with us other than his or her service as a
director.
Nominations
to the Board of Directors
General
— Our directors take a critical role in guiding our strategic direction and oversee the management of the Company. Our board
of directors’ candidates are considered based upon various criteria, such as their broad-based business and professional skills
and experiences, a global business and social perspective, concern for the long-term interests of the shareholders, diversity, and personal
integrity and judgment. In addition, directors must have time available to devote to our board of directors’ activities and to
enhance their knowledge of our business. Accordingly, we seek to attract and retain highly qualified directors who have sufficient time
to attend to their substantial duties and responsibilities to our Company.
Section
16(a) Beneficial Ownership Reporting Compliance
Under
Section 16(a) of the Exchange Act, our directors and certain of our officers, and persons holding more than 10 percent of our common
stock are required to file forms reporting their beneficial ownership of our common stock and subsequent changes in that ownership with
the United States Securities and Exchange Commission.
Based
solely upon a review of copies of such forms filed on Forms 3, 4, and 5 furnished to us, we believe that during the year ended June 30,
2021 , our executive officers, directors and greater than 10 percent beneficial owners complied on a timely basis with all Section 16(a)
filing requirements.
Legal
Proceedings
To
the best of our knowledge, none of our directors or executive officers has, during the past ten years:
| |
● |
been
convicted in a criminal proceeding or been subject to a pending criminal proceeding (excluding traffic violations and other minor
offenses); |
| |
|
|
| |
● |
had
any bankruptcy petition filed by or against the business or property of the person, or of any partnership, corporation or business
association of which he was a general partner or executive officer, either at the time of the bankruptcy filing or within two years
prior to that time; |
| |
|
|
| |
● |
been
subject to any order, judgment, or decree, not subsequently reversed, suspended or vacated, of any court of competent jurisdiction
or federal or state authority, permanently or temporarily enjoining, barring, suspending or otherwise limiting, his involvement in
any type of business, securities, futures, commodities, investment, banking, savings and loan, or insurance activities, or to be
associated with persons engaged in any such activity; |
| |
|
|
| |
● |
been
found by a court of competent jurisdiction in a civil action or by the Securities and Exchange Commission or the Commodity Futures
Trading Commission to have violated a federal or state securities or commodities law, and the judgment has not been reversed, suspended,
or vacated; |
| |
|
|
| |
● |
been
the subject of, or a party to, any federal or state judicial or administrative order, judgment, decree, or finding, not subsequently
reversed, suspended or vacated (not including any settlement of a civil proceeding among private litigants), relating to an alleged
violation of any federal or state securities or commodities law or regulation, any law or regulation respecting financial institutions
or insurance companies including, but not limited to, a temporary or permanent injunction, order of disgorgement or restitution,
civil money penalty or temporary or permanent cease-and-desist order, or removal or prohibition order, or any law or regulation prohibiting
mail or wire fraud or fraud in connection with any business entity; or |
| |
|
|
| |
● |
been
the subject of, or a party to, any sanction or order, not subsequently reversed, suspended or vacated, of any self-regulatory organization
(as defined in Section 3(a)(26) of the Exchange Act), any registered entity (as defined in Section 1(a)(29) of the Commodity Exchange
Act), or any equivalent exchange, association, entity or organization that has disciplinary authority over its members or persons
associated with a member. |
None
of our directors, officers or affiliates, or any beneficial owner of 5% or more of our common stock, or any associate of such persons,
is an adverse party in any material proceeding to, or has a material interest adverse to, us or any of our subsidiaries.
EXECUTIVE
COMPENSATION
The
following table sets forth the compensation paid or accrued by us to our Executive Officers for the fiscal years ended June 30, 2021
and 2020.
Summary
Compensation Table
| | |
Year | |
Salary
($) | | |
Bonus
($) | | |
Option
Awards
($) | | |
All Other
Compensation
($) | | |
Total
($) | |
| James Nathanielsz(1) (5) | |
2020 | |
$ | 357,988 | (2) | |
$ | 165,384 | (3) | |
$ | - | | |
$ | 66,821 | (4) | |
$ | 590,193 | |
| Chief Executive Officer | |
2021 | |
$ | 298,920 | (2) | |
$ | 177,840 | (5) | |
$ | - | | |
$ | 64,532 | (4) | |
$ | 541,292 | |
| | |
| |
| | | |
| | | |
| | | |
| | | |
| | |
| Carlo Campiciano(6) | |
2020 | |
$ | 51,222 | | |
$ | - | | |
$ | - | | |
$ | - | | |
$ | 51,222 | |
| Chief Financial Officer | |
2021 | |
$ | 19,530 | | |
$ | - | | |
$ | - | | |
$ | - | | |
$ | 19,530 | |
| | |
| |
| | | |
| | | |
| | | |
| | | |
| | |
| Julian Kenyon(7) | |
2020 | |
$ | 37,211 | | |
$ | - | | |
$ | - | | |
$ | - | | |
$ | 37,211 | |
| Chief Scientific Officer | |
2021 | |
$ | 40,534 | | |
$ | - | | |
$ | - | | |
$ | - | | |
$ | 40,534 | |
| |
(1) |
For
purposes of the information included in the table, the conversion rates as of June 30, 2021 and 2020, $0.7473 and $0.6891, respectively,
were used to convert amounts from AUD to USD. |
| |
|
|
| |
(2) |
Under
the Nathanielsz Employment Agreement (as defined below), Mr. Nathanielsz received a gross annual salary of $400,000 AUD per year
effective February 1, 2018 as approved by the board of directors. Mr. Nathanielsz has also accrued unused annual and long service
leave in the amounts of $41,110 (AUD) ($30,722 USD) and $46,187 (AUD) ($31,827 USD) for fiscal years 2021 and 2020, respectively,
which are included in the total above. |
| |
|
|
| |
(3) |
On
July 13, 2020 the Board approved a bonus of $240,000 AUD ($165,384 USD) being equal to 60% of Mr. Nathanielsz base salary. |
| |
|
|
| |
(4) |
Under
the Nathanielsz Employment Agreement, Mr. Nathanielsz receives a 9.5% contribution to a pension of which he is the beneficiary. In
addition, pursuant to the Nathanielsz Employment Agreement, we may make a monthly payment to cover the costs relating to Mr. Nathanielsz
use of a vehicle. For the fiscal years ended June 30, 2021 and 2020, $34,476 and $32,757, respectively, was paid to Mr. Nathanielsz
for use of a vehicle. |
| |
|
|
| |
(5) |
On
August 12, 2021, the Company entered into a Cancellation Agreement with James Nathanielsz (“Nathanielsz”), Chief Executive
Officer and Director of the Company, whereby Nathanielsz agreed to cancel his cash compensation bonus award for fiscal year 2021,
ended June 30, 2021, in exchange for common stock of the Company. The Company and Nathanielsz entered into an Amended and Restated
Employment Agreement dated May 14, 2019 (the “Agreement”). Pursuant to the terms of the Agreement, Nathanielsz was eligible
to earn an annual fiscal year cash performance bonus for each fiscal year of his employment period with the Company with a target
performance bonus of 200% of his average annualized base salary during the fiscal year for which the performance bonus is earned.
On July 20, 2021, Nathanielsz was awarded a “target” bonus of 78%, or $177,840 USD (the “Debt”) for the fiscal
year ended June 30, 2021, by the Company’s Board of Directors (the “Board”). Pursuant to the Cancellation Agreement,
Nathanielsz agreed to cancel this Debt in exchange for 5,928,000 shares of the common stock of the Company, valued at approximately
$0.03 per share, being the closing price of the stock on the date of grant. The shares were issued on August 17, 2021. |
| |
|
|
| |
(6) |
On
December 23, 2020, Carlo Campiciano resigned as the Chief Financial Officer and Secretary of Propanc Biopharma, Inc. (the “Company”),
and effective on that date. James Nathanielsz, the Company’s Chief Executive Officer, assumed the duties and additional position
of Chief Financial Officer. |
| |
(7) |
On
August 12, 2021, the Company entered into a Cancellation Agreement with Dr. Julian Kenyon (“Kenyon”), Chief Scientific
Officer and Director of the Company, whereby Kenyon agreed to cancel $102,600 USD of accrued salary due him as of June 30, 2021,
pursuant to that certain Amended and Restated Services Agreement by and between Kenyon and the Company, dated May 14, 2019, in exchange
for 3,420,000 shares of common stock of the Company, valued at approximately $0.03 per share, being the closing price of the stock
on the date of grant. The shares were issued on August 17, 2021. |
Narrative
to Summary Compensation Table
Employment
Agreement with James Nathanielsz
The
Company and James Nathanielsz entered into a new employment agreement as of May 14, 2019 (the “Nathanielsz Employment Agreement”)
setting forth the terms and conditions of Mr. Nathanielsz employment as the Company’s President and Chief Executive Officer. The
Nathanielsz Employment Agreement also contemplates that Mr. Nathanielsz serves as a member of the Board.
The
Nathanielsz Employment Agreement provides Mr. Nathanielsz with a base salary of $33,333 AUD per month ($400,000 AUD annually) and a monthly
contribution to Mr. Nathanielsz’s pension equal to 9.5% of his monthly salary. Mr. Nathanielsz has the ability to convert any accrued
but unpaid salary into common stock at the end of each fiscal year at a conversion price to be determined by Mr. Nathanielsz and the
Company, which will in no event be lower than par value or higher than the closing bid price on the date of conversion. The Company has
also agreed to pay Mr. Nathanielsz an annual discretionary bonus in an amount up to 200% of his annual base salary, which bonus shall
be determined by the Board and based upon the performance of the Company.
Mr.
Nathanielsz is entitled to 20 days of annual leave and 10 days of paid sick leave. Mr. Nathanielsz is also entitled to participate in
employee benefits plans, fringe benefits and perquisites maintained by the Company to the extent the Company provides similar benefits
or perquisites (or both) to similarly situated executives of the Company.
In
the event that the Company provides notice of non-renewal of the Nathanielsz Employment Agreement, the Company terminates Mr. Nathanielsz
without cause (as defined in the Nathanielsz Employment Agreement) or Mr. Nathanielsz terminates his employment for good reason (as defined
in the Nathanielsz Employment Agreement), the Company has agreed to pay Mr. Nathanielsz a severance payment in an amount equal to Mr.
Nathanielsz’s base salary for the year of termination in addition to accrued but unpaid salary, reimbursement of expenses and certain
other employee benefits as determined under the terms of the applicable plans (“Accrued Amounts”). In the event that Mr.
Nathanielsz provides notice of non-renewal of the Nathanielsz Employment Agreement, the Company terminates Mr. Nathanielsz for cause
or Mr. Nathanielsz terminates his employment without good reason, Mr. Nathanielsz is only entitled to the Accrued Amounts.
The
Company has agreed to indemnify Mr. Nathanielsz for any liabilities, costs and expenses incurred in the event that he is made a party
to a proceeding due to his roles with the Company, other than any proceeding initiated by Mr. Nathanielsz or the Company relating to
any dispute with respect to the Nathanielsz Employment Agreement or Mr. Nathanielsz’s employment.
Under
the terms of the Nathanielsz Employment Agreement, Mr. Nathanielsz is also subject to certain restrictive covenants, including a one-year
non-compete.
Employment
Agreement with Carlo Campiciano
In
connection with Mr. Campiciano’s appointment as the Company’s Chief Financial Officer and Secretary, effective as of June
24, 2019, Propanc PTY entered into an Employment Agreement with Mr. Campiciano. Pursuant to the Employment Agreement, Mr. Campiciano
will be compensated at an hourly rate based on a pro-rated annual salary for the number of hours of services to be provided to the Company.
If Mr. Campiciano’s employment is terminated by either party, he will be entitled to certain termination benefits, including payment
of accrued but untaken annual leave, salary payments pro-rated based on applicable notice period required under the Employment Agreement,
reimbursement of incurred business related expenses and such other payments as may be required by the Australian National Employment
Standards. The Employment Agreement contains covenants for the benefit of Propanc PTY relating to non-interference with Propanc PTY’s
business after termination of employment and protection of Propanc PTY’s confidential information, certain customary representations
and warranties and standard Propanc PTY indemnification obligations.
On
December 23, 2020, Carlo Campiciano resigned as the Chief Financial Officer and Secretary of Propanc Biopharma, Inc. (the “Company”),
effective on that date.
Amended
and Restated Services Agreement – Julian Kenyon
On
May 14, 2019, the Company entered into an Amended and Restated Services Agreement (the “Services Agreement”) with Dr. Kenyon,
the Company’s Chief Scientific Officer and a director, for a term of three years, subject to automatic one-year renewals, at an
annual salary of $54,000 AUD. In connection with the execution of the Services Agreement, Dr. Kenyon was designated as an executive officer
of the Company and assumed a more active executive role with the Company. Pursuant to the Services Agreement, Dr. Kenyon was granted
options to purchase 20 shares of the Company’s common stock (the “Kenyon Options”), with an exercise price per share
of $4,250 (100% of the closing market price of the Company’s common stock on May 14, 2019, the date of approval of such grant by
the Company’s board of directors), (ii) 20 restricted stock units of the Company (the “Initial Kenyon RSUs”), and (iii)
an additional 20 restricted stock units of the Company (the “Additional Kenyon RSUs”). Such options and restricted stock
units were granted pursuant to the 2019 Plan approved by the Company’s board of directors on the Effective Date. The Kenyon Options
have a term of 10 years from the date of grant. 1/3rd of the Kenyon Options shall vest every successive one-year anniversary following
the Effective Date, provided, that on each such vesting date Dr. Kenyon is employed by the Company and subject to the other provisions
of the Services Agreement. The Initial Kenyon RSUs shall vest on the one-year anniversary of the Effective Date, subject to Dr. Kenyon’s
continued employment with the Company through such vesting date. The Additional Kenyon RSUs will vest as follows, subject to Dr. Kenyon’s
continued employment with the Company through the applicable vesting date: (i) 5 of the Additional Kenyon RSUs shall vest upon the Company
submitting the CTA for PRP for the Study in an applicable jurisdiction to be selected by the Company, (ii) 5 of the Additional Kenyon
RSUs shall vest upon the Company completing an equity financing in the amount of at least $4,000,000 in gross proceeds, (iii) 5 of the
Additional Kenyon RSUs shall vest upon the shares of the Company’s Common Stock being listed on a senior stock exchange (NYSE,
NYSEMKT or NASDAQ), and (iv) the remaining 5 of the Additional Kenyon RSUs shall vest upon the Company enrolling its first patient in
the Study. Each vested Kenyon RSU shall be settled by delivery to Mr. Kenyon of one share of the Company’s common stock and/or
the fair market value of one share of common stock in cash, at the sole discretion of the Company’s board of directors and subject
to the Plan, on the first to occur of: (i) the date of a Change of Control (as defined in the Services Agreement), (ii) the date that
is ten business days following the vesting of such Kenyon RSU, (iii) the date of Dr. Kenyon’s death or Disability (as defined in
the Services Agreement), and (iv) Dr. Kenyon’s employment being terminated either by the Company without Cause or by Dr. Kenyon
for Good Reason (as defined in the Services Agreement). In the event of a Change of Control (as defined in the Services Agreement), 50%
of any unvested portion of the Kenyon Options and the Kenyon RSUs shall vest immediately prior to such event.
2019
Equity Incentive Plan
On
May 14, 2019, our board of directors adopted our 2019 Equity Incentive Plan (the “2019 Plan”), which reserves a total of
234,000 shares of our common stock for issuance under the 2019 Plan (adjusted for the planned Reverse Stock Split). As described below,
incentive awards authorized under the 2019 Plan include, but are not limited to, incentive stock options within the meaning of Section
422 of the Internal Revenue Code of 1986, as amended (the “Code”). If an incentive award granted under the 2019 Plan expires,
terminates, is unexercised or is forfeited, or if any shares are surrendered to us in connection with the exercise of an incentive award,
the shares subject to such award and the surrendered shares will become available for further awards under the 2019 Plan.
Administration
- Our board of directors will administer the 2019 Plan. Subject to the terms of the 2019 Plan, our board of directors has complete
authority and discretion to determine the terms upon which awards may be granted under the 2019 Plan.
Grants
- The 2019 Plan authorizes the grant to participants of nonqualified stock options, incentive stock options, restricted stock awards,
restricted stock units, performance grants intended to comply with Section 162(m) of the Code and stock appreciation rights, as described
below:
| |
● |
Options
granted under the 2019 Plan entitle the grantee, upon exercise, to purchase up to a specified number of shares from us at a specified
exercise price per share. The exercise price for shares of Common Stock covered by an option generally cannot be less than the fair
market value of Common Stock on the date of grant unless agreed to otherwise at the time of the grant. In addition, in the case of
an incentive stock option granted to an employee who, at the time the incentive stock option is granted, owns stock representing
more than 10% of the voting power of all classes of stock of the Company or any parent or subsidiary, the per share exercise price
will be no less than 110% of the fair market value of Common Stock on the date of grant. |
| |
|
|
| |
● |
Restricted
stock awards and restricted stock units may be awarded on terms and conditions established by the compensation committee, which may
include performance conditions for restricted stock awards and the lapse of restrictions on the achievement of one or more performance
goals for restricted stock units. |
| |
|
|
| |
● |
The
board of directors may make performance grants, each of which will contain performance goals for the award, including the performance
criteria, the target and maximum amounts payable, and other terms and conditions. |
| |
|
|
| |
● |
The
2019 Plan authorizes the granting of stock awards. The board of directors will establish the number of shares of our common stock
to be awarded (subject to the aggregate limit established under the 2019 Plan upon the number of shares of our common stock that
may be awarded or sold under the 2019 Plan) and the terms applicable to each award, including performance restrictions. |
| |
|
|
| |
● |
Stock
appreciation rights (“SARs”) entitle the participant to receive a distribution in an amount not to exceed the number
of shares of Common Stock subject to the portion of the SAR exercised multiplied by the difference between the market price of a
share of Common Stock on the date of exercise of the SAR and the market price of a share of our common Stock on the date of grant
of the SAR. |
Duration,
Amendment, and Termination - Our board of directors has the power to amend, suspend or terminate the 2019 Plan without stockholder approval
or ratification at any time or from time to time. No change may be made that increases the total number of shares of Common Stock reserved
for issuance pursuant to incentive awards or reduces the minimum exercise price for options or exchange of options for other incentive
awards, unless such change is authorized by our stockholders within one year of such change. Unless sooner terminated, the 2019 Plan
would terminate ten years after it is adopted.
No
awards or any shares of our common stock were issued during the fiscal year 2021 under the 2019 Plan.
Outstanding
Equity Awards at Fiscal Year-End
The
following table sets forth certain information with respect to grants of plan-based awards for the fiscal year ended June 30, 2021 to
the Named Executive Officer. Except as set forth below, all of the outstanding equity awards granted to our Named Executive Officer were
fully vested as of June 30, 2021.
| Option awards | |
Stock awards |
| Name | |
Number of Securities Underlying Unexercised Options (#) Exercisable | | |
Number of Securities Underlying Unexercised Options (#) Unexercisable | | |
Option Exercise
Price ($) | | |
Option Expiration Date | |
Number of Shares, Units or Other Rights That Have Not Vested
(#) | | |
Market Value or Payout Value of Unearned Shares, Units or Other Rights That Have Not Vested
($) | |
| | |
| | |
| | |
| | |
| |
| | |
| |
| James Nathanielsz (1) | |
| 26 | | |
| 13 | | |
$ | 4,675 | | |
May 13, 2029 | |
| 39 | | |
| 165,747 | |
| Julian Kenyon (2) | |
| 13 | | |
| 6 | | |
$ | 4,250 | | |
May 13, 2029 | |
| 20 | | |
| 82,873 | |
| |
(1) |
On
May 14, 2019, the Board granted Mr. Nathanielsz 39 tenure based stock options at an exercise price of $4,675 per share and 78 performance
based restricted stock units. The fair value of the 39 options and 78 restricted stock units at the grant date was $165,747 and $331,493,
respectively. With 39 of such restricted stock vested on May 14, 2020 and the balance subject to performance conditions. |
| |
|
|
| |
(2) |
On
May 14, 2019, the Board granted Mr. Kenyon 20 tenure based stock options at an exercise price of $4,250 per share and 40 performance
based restricted stock units. The fair value of the 20 options and 40 restricted stock units at the grant date was $82,873 and $165,747,
respectively. With 20 of such restricted stock vested on May 14, 2020 and the balance subject to performance conditions. |
Director
Compensation for the Fiscal Year Ended June 30, 2021
| Name | |
Fees earned
or paid in
cash ($) | | |
Option Awards ($) | | |
All Other Compensation ($) | | |
Total ($) | |
| Josef Zelinger (1) | |
$ | - | | |
$ | - | | |
| 84,000 | | |
$ | 84,000 | |
| |
(1) |
The
Company issued 2,800,000 shares of common stock of the Company for accrued director services of $84,000 as of June 30, 2021. The
2,800,000 shares of common stock was valued at approximately $0.03 per share, being the closing price of the stock on the date of
grant. The shares were issued on August 17, 2021. |
Amended
and Restated Director Agreement – Josef Zelinger
On
August 12, 2021, the Company entered into an Amended and Restated Director Agreement (the “Director Agreement”) with Josef
Zelinger (“Zelinger”). Pursuant to the terms of the Director Agreement, the Company shall pay Zelinger a base salary of $250.00
AUD per month, payable on the first day of each month. In addition, the Company may compensate Zelinger additional consideration for
advisory services performed by the Director, either in the form of cash or common stock, at the discretion of the Board. The Company
issued 2,800,000 shares of common stock of the Company for accrued director services of $84,000 as of June 30, 2021. The 2,800,000 shares
of common stock was valued at approximately $0.03 per share, being the closing price of the stock on the date of grant. The shares were
issued on August 17, 2021.
Other
Director Compensation
Directors
are reimbursed for reasonable expenses incurred in attending meetings and carrying out duties as board members.
Scientific
Advisory Board Members Compensation
The
Company has entered into Scientific Advisory Board Member Agreements with certain members of its Scientific Advisory Board (the “SAB
Agreements”). The SAB Agreements contain substantially similar terms and primarily relate to the protection of the Company’s
intellectual property. The SAB Agreements also include provisions for the members’ compensation for the services performed as a
member of the Scientific Advisory Board. Messrs. Kutz, Brandt and Smyth each are paid a monetary fee for each year of service provided.
Narrative
Disclosure of Compensation Policies and Practices as They Relate to Our Risk Management
We
believe that our compensation policies and practices for all employees and other individual service providers, including executive officers,
do not create risks that are reasonably likely to have a material adverse effect on us.
SECURITY
OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The
following sets forth information as of February 2, 2022 regarding the number of shares of our common stock beneficially owned
by (i) each person that we know beneficially owns more than 5% of our outstanding common stock, (ii) each of our directors and named
executive officer and (iii) all of our directors and named executive officers as a group.
The
amounts and percentages of our Common Stock beneficially owned are reported on the basis of SEC rules governing the determination of
beneficial ownership of securities. Under the SEC rules, a person is deemed to be a “beneficial owner” of a security if that
person has or shares “voting power,” which includes the power to vote or to direct the voting of such security, or “investment
power,” which includes the power to dispose of or to direct the disposition of such security. A person is also deemed to be a beneficial
owner of any securities of which that person has the right to acquire beneficial ownership within 60 days through the exercise of any
stock option, warrant or other right, and the conversion of preferred stock. Under these rules, more than one person may be deemed a
beneficial owner of the same securities and a person may be deemed to be a beneficial owner of securities as to which such person has
no economic interest. Unless otherwise indicated, each of the shareholders named in the table below, or his or her family members, has
sole voting and investment power with respect to such shares of our Common Stock. Except as otherwise indicated, the address of each
of the shareholders listed below is: c/o Propanc Biopharma, Inc., 302, 6 Butler Street, Camberwell, VIC, 3124 Australia.
| | |
Common Stock Beneficially Owned | | |
Series A Preferred Stock Beneficially Owned | | |
Series B Preferred Stock Beneficially Owned | |
| Name and Address of Beneficial Owner | |
Number of Shares Beneficially Owned | | |
Percentage of Class(1) | | |
Number of Shares Beneficially Owned | | |
Percentage of Class(2) | | |
Number of Shares Beneficially Owned | | |
Percentage of Class (2) | |
| | |
| | |
| | |
| | |
| | |
| | |
| |
| North Horizon Pty Ltd.(3) | |
| 5,928,004 | | |
| 9.65 | % | |
| 500,001 | | |
| 100 | % | |
| - | | |
| - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| James Nathanielsz(4) | |
| - | | |
| - | | |
| - | | |
| - | | |
| 1 | | |
| 100 | % |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Dr. Julian Kenyon(5) | |
| 3,420,005 | | |
| 5.57 | % | |
| - | | |
| - | | |
| - | | |
| - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Josef Zelinger | |
| 2,800,005 | | |
| 4.56 | % | |
| - | | |
| - | | |
| - | | |
| - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| All directors and executive officers, as a group (3 persons) | |
| 12,148,014 | | |
| 19.78 | % | |
| 500,001 | | |
| 100 | % | |
| 1 | | |
| 100 | % |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| 5% Shareholders | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Sylva International LLC (6) | |
| 5,908,291 | | |
| 9.62 | % | |
| - | | |
| - | | |
| - | | |
| - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Sylvia Nathanielsz (7) | |
| 2,800,000 | | |
| 4.73 | % | |
| - | | |
| - | | |
| - | | |
| - | |
(1)
Applicable percentages are based on 61,417,527 shares of our common stock outstanding as of February 2, 2022.
(2)
Applicable percentages are based on 500,000 shares of our Series A Preferred Stock and 1 share of our Series B Preferred Stock outstanding
as of February 2, 2022.
(3)
North Horizon Pty Ltd. is a Nathanielsz Family Trust. Mr. James Nathanielsz, the Chief Executive Officer and a director of our Company,
has investing and dispositive power of shares beneficially owned by North Horizon Pty Ltd.
(4)
Excludes 26 vested stock options, 13 unvested stock options, 39 restricted stock units that vested in May 2020 and 39 restricted stock
units that are subject to certain vesting conditions, as discussed above in the section captioned “Executive Compensation - New
Employment Agreement with James Nathanielsz,”.
(5)
Excludes 13 vested stock options, 6 unvested stock options, and 20 restricted stock units that vested in May 2020, and 20 restricted
stock units that are subject to certain vesting conditions, as discussed above in the section captioned “Executive Compensation
- New Services Agreement with Julian Kenyon”.
(6)
Ross Silver has investing and dispositive power of shares beneficially owned by Sylva International LLC.
(7)
Sylvia Nathanielsz is the wife of James Nathanielsz.
CERTAIN
RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
Related-Party
Transactions
The
following includes a summary of transactions since July 1, 2019 to which we have been a party, in which the amount involved in the transaction
exceeded $120,000, and in which any of our directors, executive officers or, to our knowledge, beneficial owners of more than 5% of our
capital stock or any member of the immediate family of any of the foregoing persons had or will have a direct or indirect material interest,
other than equity and other compensation, termination, change in control and other arrangements, which are described above under “Item
11. Executive Compensation.”
Our
principal executive office is located at 302, 6 Butler Street, Camberwell, VIC, 3124 Australia, which we lease from Horizon Pty Ltd.,
a related party, of which Mr. Nathanielsz, our Chief Executive Officer, Chief Financial Officer and a director, and his wife are owners
and directors. The lease has a one-year term commencing May 6, 2021, and we are currently obligated to pay $3,606 AUD or $2,431 USD (depending
on exchange rate), inclusive of tax, in rent per month
Mr.
Nathanielsz’s wife, Sylvia Nathanielsz, is and has been an employee of our Company since October 2015. Mrs. Nathanielsz receives
an annual salary of $120,000 AUD, or $80,904 USD, and is entitled to benefits customarily expected to be provided to employees of the
Company.
Employment
and Director Compensation Arrangements
The
relationships and related party transactions described herein are in addition to any employment and director compensation arrangements
with our executive officers and directors, which are described above under “Executive Compensation — Narrative to Summary
Compensation Table and Director Compensation.”
Indemnification
Agreements
Our
Certificate of Incorporation provides that none of our officers or directors shall be personally liable for any obligations of our Company
or for any duties or obligations arising out of any acts or conduct of said officer or director performed for or on behalf of our Company,
including without limitation, acts of negligence or contributory negligence. In addition, our Bylaws provide that we shall indemnify
and hold harmless each person and their heirs and administrators who shall serve at any time hereafter as a director or officer of our
Company from and against any and all claims, judgments and liabilities to which such persons shall become subject by reason of their
having heretofore or hereafter been a director or officer of our Company, or by reason of any action alleged to have heretofore or hereafter
taken or omitted to have been taken by him or her as such director or officer, and that we shall reimburse each such person for all legal
and other expenses reasonably incurred by him or her in connection with any such claim, judgment or liability, including our power to
defend such persons from all suits or claims as provided for under the provisions of the Delaware General Corporation Law; provided,
however, that no such persons shall be indemnified against, or be reimbursed for, any expense incurred in connection with any claim or
liability arising out of his (or her) own willful misconduct. In addition, we intend to enter into indemnification agreements with our
directors and officers and some of our executives may have certain indemnification rights arising under their employment agreements with
us. We believe that these bylaw provisions and indemnification agreements are necessary to attract and retain qualified persons as directors
and officers.
The
limitation of liability and indemnification provisions in our Certificate of Incorporation may discourage stockholders from bringing
a lawsuit against our directors for breach of their fiduciary duties. They may also reduce the likelihood of derivative litigation against
our directors and officers, even though an action, if successful, might benefit us and our stockholders. A stockholder’s investment
may be harmed to the extent we pay the costs of settlement and damage awards against directors and officers pursuant to these indemnification
provisions.
On
May 14, 2019, our board of directors approved a form of Indemnification Agreement (“Indemnification Agreement”) for each
of our officers and directors. The Indemnification Agreement requires us to indemnify our directors and officers and to advance expenses
on behalf of such directors or officers to the fullest extent permitted by applicable law and establish the procedures by which a director
or executive officer may request and receive indemnification. The Indemnification Agreement is in addition to other rights to which a
director or officer may be entitled under our Certificate of Incorporation, Bylaws and applicable law.
Director
Independence
Our
board of directors has reviewed the independence of our directors and has determined that Josef Zelinger qualifies as an independent
director pursuant to Rule 5605(a)(2) of Nasdaq and applicable SEC rules and regulations. In making this determination, our board of directors
considered the relationships that each of our directors has with us and all other facts and circumstances our board of directors deemed
relevant in determining their independence.
CHANGES
IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
There
have been no changes in our independent registered public accounting firm during the last two fiscal years, and we have not had any material
disagreements with our independent registered public accounting firm during that time.
DISCLOSURE
OF COMMISSION POSITION ON INDEMNIFICATION FOR SECURITIES ACT LIABILITIES
Our
Certificate of Incorporation contains provisions that limit the liability of our directors for monetary damages to the fullest extent
permitted by Delaware law. Consequently, our directors will not be personally liable to us or our stockholders for monetary damages for
any breach of fiduciary duties as directors, except liability for:
| |
● |
any
breach of the director’s duty of loyalty to us or our stockholders; |
| |
|
|
| |
● |
any
act or omission not in good faith or that involves intentional misconduct or a knowing violation of law; |
| |
|
|
| |
● |
unlawful
payments of dividends or unlawful stock repurchases or redemptions as provided in Section 174 of the General Corporation Law of the
State of Delaware; or |
| |
|
|
| |
● |
any
transaction from which the director derived an improper personal benefit. |
Our
Certificate of Incorporation, as amended, provides that we are required to indemnify our directors and officers, in each case to the
fullest extent permitted by Delaware law. Our Certificate of Incorporation also provides that we are obligated to advance expenses incurred
by a director or officer in advance of the final disposition of any action or proceeding, and permit us to secure insurance on behalf
of any officer, director, employee or other agent for any liability arising out of his or her actions in that capacity regardless of
whether we would otherwise be permitted to indemnify him or her under the provisions of Delaware law.
To
the extent that indemnification for liabilities arising under the Securities Act may be permitted to directors, officers or persons controlling
our company pursuant to the foregoing provisions, we have been informed that, in the opinion of the SEC, such indemnification is against
public policy as expressed in the Securities Act and is therefore unenforceable.
WHERE
YOU CAN FIND MORE INFORMATION
We
have filed a registration statement on Form S-1 with the SEC covering the shares and warrants we are offering by this prospectus. This
prospectus does not include all of the information contained in the registration statement. You should refer to the registration statement
and its exhibits for additional information. Whenever we make reference in this prospectus to any of our contracts, agreements or other
documents, the references are not necessarily complete and you should refer to the exhibits filed or documents incorporated by reference
as part of the registration statement for copies of the actual contract, agreement or other document.
We
file annual, quarterly and other periodic reports, proxy statements and other information with the SEC. You can read our SEC filings,
including this registration statement, over the Internet at the SEC’s website at www.sec.gov. You may also read and copy any document
we file with the SEC at its public reference facilities at 100 F Street NE, Washington, D.C. 20549. You may also obtain copies of these
documents at prescribed rates by writing to the Public Reference Section of the SEC at 100 F Street NE, Washington, D.C. 20549. Please
call the SEC at 1-800-SEC-0330 for further information on the operation of the public reference facilities.
Our
Internet address is www.propanc.com. There we make available free of charge, on or through the investor relations section of our website,
Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to those reports filed pursuant
to Section 13(a) or 15(d) of the Exchange Act as soon as reasonably practicable after we electronically file such material with the SEC.
The information found on our website is not part of this prospectus and investors should not rely on any such information in deciding
whether to invest.
Statements
contained in this prospectus as to the contents of any contract or other document that we have filed as an exhibit to the registration
statement are qualified in their entirety by reference to the exhibits for a complete statement of their terms and conditions.
The
representations, warranties and covenants made by us in any agreement that is filed as an exhibit to the registration statement of which
this prospectus is a part were made solely for the benefit of the parties to such agreement, including, in some cases, for the purpose
of allocating risk among the parties to such agreements, and should not be deemed to be a representation, warranty or covenant to you.
Moreover, such representations, warranties or covenants were made as of an earlier date. Accordingly, such representations, warranties
and covenants should not be deemed as accurately representing the current state of our affairs.
40,000,000
Shares of Common Stock

PROSPECTUS
February 11, 2022
Propanc Biopharma (PK) (USOTC:PPCB)
Historical Stock Chart
From Jun 2024 to Jul 2024
Propanc Biopharma (PK) (USOTC:PPCB)
Historical Stock Chart
From Jul 2023 to Jul 2024