Halozyme Therapeutics Sees CHMP Approval for Phesgo
November 13 2020 - 9:43AM
Dow Jones News
By Chris Wack
Halozyme Therapeutics Inc. said Friday that the European
Medicines Agency's Committee for Medicinal Products for Human Use
has recommended the approval of Roche's Phesgo.
Phesgo is a fixed-dose combination of Perjeta and Herceptin,
administered by subcutaneous injection in combination with
intravenous chemotherapy, using Halozyme's Enhamze Technology for
the treatment of early and metastatic HER2-positive breast
cancer.
Based on the recommendation, a final decision regarding the
approval of Phesgo is expected from the European Commission
soon.
The CHMP recommendation is based on results from a phase III
study, which met its primary endpoint.
The U.S. Food and Drug Administration recently expedited the
approval of Phesgo for the treatment of early and metastatic
HER2-positive breast cancer.
Write to Chris Wack at chris.wack@wsj.com
(END) Dow Jones Newswires
November 13, 2020 10:28 ET (15:28 GMT)
Copyright (c) 2020 Dow Jones & Company, Inc.
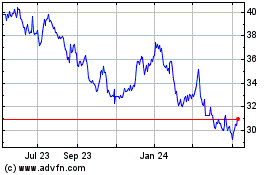
Roche (QX) (USOTC:RHHBY)
Historical Stock Chart
From Apr 2024 to May 2024
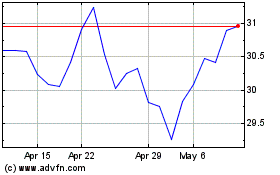
Roche (QX) (USOTC:RHHBY)
Historical Stock Chart
From May 2023 to May 2024