0001270073
false
0001270073
2023-09-26
2023-09-26
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of
the Securities
Exchange Act of 1934
Date
of Report (Date of earliest event reported): September 26, 2023
Intercept
Pharmaceuticals, Inc.
(Exact name of registrant as specified in its charter)
| Delaware | |
001-35668 | |
22-3868459 |
| (State or other Jurisdiction of | |
(Commission File No.) | |
(I.R.S. Employer Identification |
| Incorporation) | |
| |
No.) |
305 Madison Avenue
Morristown, NJ 07960
| |
(Address of principal executive offices, including Zip Code) |
|
(646)
747-1000
(Registrant’s telephone number, including area code)
Not
applicable
| |
(Former
Name or Former Address, if Changed Since Last Report) |
|
Check
the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under
any of the following provisions (see General Instruction A.2):
| ¨ | Written
communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ¨ | Soliciting
material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ¨ | Pre-commencement
communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ¨ | Pre-commencement
communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities
registered pursuant to Section 12(b) of the Act:
| Title of each class |
Trading
Symbol(s) |
Name of each exchange on which
registered |
| Common Stock, par value $0.001 per share |
ICPT |
Nasdaq Global Select Market |
Indicate by check mark whether the registrant is an emerging growth
company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange
Act of 1934 (§240.12b-2 of this chapter).
Emerging
growth company ¨
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for
complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Item 1.01 Entry
into a Material Definitive Agreement.
Agreement and Plan of Merger
On September 26, 2023, Intercept Pharmaceuticals,
Inc. (the “Company”) entered into an Agreement and Plan of Merger (the “Merger Agreement”) with Alfasigma S.p.A.
(“Alfasigma”) and Interstellar Acquisition Inc., a wholly owned subsidiary of Alfasigma (“Purchaser”).
Pursuant to the Merger Agreement, Purchaser
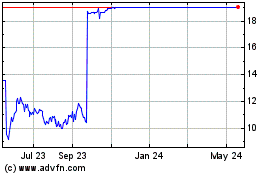
will commence a tender offer (the “Offer”) to acquire all of the outstanding shares of the Company’s common stock (the
“Shares”) at an offer price of $19.00 per Share, net to the seller in cash, without interest (the “Offer Price”),
subject to any applicable withholding taxes.
The obligation of Purchaser to purchase Shares tendered in the
Offer is subject to the satisfaction of the conditions set forth in Annex I to the Merger Agreement, including (i) there shall have
been validly tendered and not validly withdrawn that number of Shares that, considered together with all other Shares (if any)
beneficially owned by Alfasigma and its affiliates, represents one more Share than 50% of the Shares outstanding at the time of the
expiration of the Offer; (ii) the Merger Agreement shall not have been validly terminated in accordance with its terms; (iii) the
expiration or termination of the waiting period under the Hart-Scott-Rodino Antitrust Improvements Act of 1976, as amended; and (iv) those other conditions set forth in Annex I to the Merger Agreement.
Following the consummation of the Offer, Purchaser
will merge with and into the Company, with the Company surviving as a wholly owned subsidiary of Alfasigma (the “Merger”).
In the Merger, each Share issued and outstanding immediately prior to the effective time (the “Effective Time”) of the Merger
(other than certain excluded Shares as described in the Merger Agreement) will automatically be converted into the right to receive the
Offer Price.
Except as otherwise agreed by the Company, Alfasigma and Purchaser, at the Effective Time, each option to purchase
Shares under the Company’s equity plans (each, a “Company Option”) that is outstanding and unexercised as of immediately
prior to the Effective Time, whether or not vested, and that has a per share exercise price that is less than the Offer Price, will be
cancelled and converted into the right to receive a cash payment equal to the product of (i) the excess of the Offer Price over the exercise
price payable per Share of such Company Option and (ii) the total number of Shares subject to such Company Option immediately prior to
the Effective Time. At the Effective Time, each Company Option other than those described in the preceding sentence that is then outstanding
and unexercised as of immediately prior to the Effective Time, whether or not vested, will be cancelled with no consideration payable
in respect thereof.
At the Effective Time, each restricted stock
unit with respect to Shares subject to performance vesting conditions granted under the Company’s equity plans (each, a “Company
PSU”) that is outstanding as of immediately prior to the Effective Time will be cancelled and the holder thereof will be entitled
to receive a cash payment (without interest) equal to the product of the Offer Price and the number of Shares subject to such Company
PSU as of immediately prior to the Effective Time based upon the actual level of performance determined in accordance with the terms of
the applicable Company PSU award agreement.
At the Effective Time, each restricted stock
unit with respect to Shares granted under the Company’s equity plans, other than the Company PSUs, (each, a “Company RSU”) that is outstanding as
of immediately prior to the Effective Time will be converted into the contingent right to receive a cash payment (without interest) equal
to the product of the Offer Price and the number of Shares subject to such Company RSU as of immediately prior to the Effective Time.
Subject to the applicable holder’s continued service with Alfasigma or its affiliates after the consummation of the transaction,
such contingent right to receive a cash payment in respect of Company RSUs will vest and become payable in accordance with the vesting
schedule (including any vesting acceleration provisions) that applied to the corresponding portion of the applicable Company RSUs immediately
prior to the Effective Time. Such contingent right to receive cash payment in respect of Company RSUs will otherwise remain subject to
the same terms and conditions that are applicable to the underlying Company RSUs immediately prior to the Effective Time (except for terms rendered inoperative by reason of the transactions contemplated by the Merger Agreement).
The Merger Agreement includes representations,
warranties and covenants of the parties customary for a transaction of this nature. Among other things, until the earlier of the termination
of the Merger Agreement or the Effective Time, the Company has agreed to operate its business in the ordinary course consistent with past
practice and has agreed to certain other operating covenants, as set forth fully in the Merger Agreement. The Merger Agreement also prohibits
the Company’s solicitation of proposals relating to alternative transactions and restricts the Company’s ability to furnish
information to, or participate in any discussions or negotiations with, any third party with respect to any such transaction, subject
to certain limited exceptions.
Either the Company or Alfasigma may terminate
the Merger Agreement in certain circumstances, including if (i) the Offer is not completed on or prior to midnight Eastern Time on January
26, 2024, subject to automatic extension for a period of 90 days in the event that antitrust approval has not been obtained or a governmental
authority of competent jurisdiction in a jurisdiction where either party operates their respective business or owns any material assets
has issued a judgment or order preventing the consummation of the Offer or the Merger or any applicable law by such a governmental authority
makes consummation of the Offer or the Merger illegal (the “Outside Date”), (ii) a governmental authority of competent jurisdiction
in a jurisdiction where either party operates their respective business or owns any material assets has issued a final, non-appealable
judgment or order permanently preventing the consummation of the Offer or the Merger or any applicable law by such a governmental authority
makes consummation of the Offer or the Merger illegal, or (iii) prior to the Offer Acceptance Time (as defined in the Merger Agreement)
the other party breaches its representations, warranties or covenants in the Merger Agreement in a way that would cause certain conditions
of the Offer not to be satisfied, subject to the right of the breaching party to cure the breach as set forth in the Merger Agreement.
In addition, the Company may terminate the Merger Agreement, subject to compliance with specified process and notice requirements, in
order to enter into an agreement with a third party who has made a “Superior Offer” (as defined in the Merger Agreement) (provided
that the Company complied in all material respects with its non-solicitation obligations with respect to such Superior Offer), and Alfasigma
may terminate the Merger Agreement if the Company’s board of directors has changed its recommendation in favor of the Offer and
the Merger, approved or recommended a third party “Acquisition Proposal” (as defined in the Merger Agreement), failed to include its recommendation in the Schedule 14D-9, failed to publicly reaffirm its recommendation within three business
days when requested by Alfasigma or has taken certain other specified actions described in the Merger Agreement (each a “Company
Adverse Recommendation Change”).
In the event of a termination of the Merger
Agreement under certain specified circumstances, including (i) termination by the Company to enter into an agreement providing for a Superior
Offer, (ii) termination by Alfasigma following a Company Adverse Recommendation Change, or (iii) termination by either the Company or
Alfasigma if the closing of the transactions contemplated by the Merger Agreement has not occurred by the Outside Date or termination
by Alfasigma prior to the Offer Acceptance Time if the Company breaches its representations, warranties or covenants in the Merger Agreement
in a way that would cause certain conditions of the Offer not to be satisfied (subject to the Company’s right to cure the breach
as set forth in the Merger Agreement prior to such time of termination), and (A) a bona fide “Acquisition Proposal” (as defined
in the Merger Agreement) has been publicly disclosed after the date of the Merger Agreement and such Acquisition Proposal has not been
publicly withdrawn prior to such termination and (B) within twelve months following such termination, the Company enters into a definitive
agreement for an Acquisition Proposal or consummates an Acquisition Proposal, in each case of the foregoing clauses (i)-(iii), the Company
is required to pay Alfasigma a termination fee equal to $34,000,000.
This summary is qualified in its entirety
by reference to the Merger Agreement, which is filed as Exhibit 2.1 hereto and incorporated herein by reference.
Item 8.01 Other Events.
On September 26, 2023, the Company issued a press release announcing
the execution of the Merger Agreement and certain other information. A copy of the press release is attached to this Current Report as
Exhibit 99.1 and is incorporated herein by reference.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits
* Schedules to the Agreement and Plan of Merger have been omitted pursuant
to Item 601(a)(5) of Regulation S-K. The registrant will furnish copies of any such schedules to the U.S. Securities and Exchange Commission
upon request.
* * *
Additional Information
The foregoing description of the Merger Agreement
is not complete and is qualified in its entirety by reference to the Merger Agreement, which is attached as Exhibit 2.1 to this report
and is incorporated herein by reference.
The Merger Agreement and the foregoing description
have been included to provide investors and stockholders with information regarding the terms of the Merger Agreement. They are not intended
to provide any other factual information about the Company. The representations, warranties and covenants contained in the Merger Agreement
were or will be made only as of specified dates for the purposes of the Merger Agreement, were (except as expressly set forth therein)
solely for the benefit of the parties to the Merger Agreement and may be subject to qualifications and limitations agreed upon by such
parties. In particular, in reviewing the representations, warranties and covenants contained in the Merger Agreement and discussed in
the foregoing descriptions, it is important to bear in mind that such representations, warranties and covenants were negotiated with the
principal purpose of allocating risk between the parties, rather than establishing matters as facts. Such representations, warranties
and covenants may also be subject to a contractual standard of materiality different from those generally applicable to stockholders and
reports and documents filed with the U.S. Securities and Exchange Commission (the “SEC”). Investors and stockholders
should not rely on such representations, warranties and covenants as characterizations of the actual state of facts or circumstances described
therein. Information concerning the subject matter of such representations, warranties and covenants may change after the date of the
agreements, which subsequent information may or may not be fully reflected in the parties’ public disclosures.
Notice to Investors and Security Holders
The tender offer described in this Current Report on Form 8-K has not
yet commenced. This Current Report on Form 8-K is for informational purposes only and is neither an offer to purchase nor a solicitation
of an offer to sell shares of Intercept Pharmaceuticals, Inc. (the “Company”), nor is it a substitute for any tender offer
materials that the Company or Alfasigma S.p.A. (together with its subsidiaries, “Alfasigma”) will file with the SEC. A solicitation
and an offer to buy shares of the Company will be made only pursuant to an offer to purchase and related materials that Alfasigma intends
to file with the SEC. At the time the tender offer is commenced, Alfasigma will file a Tender Offer Statement on Schedule TO with the
SEC, and the Company will file a Solicitation/Recommendation Statement on Schedule 14D-9 with the SEC with respect to the tender offer.
THE COMPANY’S STOCKHOLDERS AND OTHER INVESTORS ARE URGED TO READ THE TENDER OFFER MATERIALS (INCLUDING AN OFFER TO PURCHASE, A RELATED
LETTER OF TRANSMITTAL AND CERTAIN OTHER TENDER OFFER DOCUMENTS) AND THE SOLICITATION/RECOMMENDATION STATEMENT BECAUSE THEY WILL CONTAIN
IMPORTANT INFORMATION WHICH SHOULD BE READ CAREFULLY BEFORE ANY DECISION IS MADE WITH RESPECT TO THE TENDER OFFER. The Offer to Purchase,
the related Letter of Transmittal and certain other tender offer documents, as well as the Solicitation/Recommendation Statement, will
be sent to all stockholders of the Company at no expense to them. The Tender Offer Statement and the Solicitation/Recommendation Statement
will be made available for free at the SEC’s website at www.sec.gov. Additional copies may be obtained for free by contacting Alfasigma
or the Company. Free copies of these materials and certain other offering documents will be made available by the Company by mail to Intercept
Pharmaceuticals, Inc., 305 Madison Avenue, Morristown, NJ 07960, Attention: Corporate Secretary, by email at investors@interceptpharma.com,
or by directing requests for such materials to the information agent for the offer, which will be named in the tender offer materials.
Copies of the documents filed with the SEC by the Company will be available free of charge under the “Investors & Media”
section of the Company’s internet website at https://ir.interceptpharma.com/investor-relations.
In addition to the Offer to Purchase, the
related Letter of Transmittal and certain other tender offer documents, as well as the Solicitation/Recommendation Statement, the Company
files periodic reports and other information with the SEC. The Company’s filings with the SEC are also available for free to the
public from commercial document-retrieval services and at the website maintained by the SEC at www.sec.gov.
Forward Looking Statements
This Current Report on Form 8-K contains forward-looking statements
related to the Company, Alfasigma and the proposed acquisition of the Company by Alfasigma (the “Transaction”) that involve
substantial risks and uncertainties. Forward-looking statements include any statements containing the words “anticipate,”
“believe,” “estimate,” “expect,” “intend,” “goal,” “may,” “might,”
“plan,” “predict,” “project,” “seek,” “target,” “potential,” “will,”
“would,” “could,” “should,” “continue” and similar expressions. In this Current Report
on Form 8-K, the Company’s forward-looking statements include statements about the parties’ ability to satisfy the conditions
to the consummation of the tender offer and the other conditions to the consummation of the Transaction; statements about the expected
timetable for completing the Transaction; the Company’s plans, objectives, expectations and intentions; the financial condition,
results of operations and business of the Company and Alfasigma; the ability to successfully commercialize the Company’s product
and product candidates and generate future revenues with respect to the Company’s product candidates; and the anticipated timing
of the closing of the Transaction.
Forward-looking statements are subject to certain risks, uncertainties,
or other factors that are difficult to predict and could cause actual events or results to differ materially from those indicated in any
such statements due to a number of risks and uncertainties. Those risks and uncertainties that could cause the actual results to differ
from expectations contemplated by forward-looking statements include, among other things: uncertainties as to the timing of the tender
offer and merger; uncertainties as to how many of the Company’s stockholders will tender their stock in the offer; the possibility
that competing offers will be made; the possibility that various closing conditions for the Transaction may not be satisfied or waived,
including that a governmental entity may prohibit, delay or refuse to grant approval for the consummation of the Transaction; the effects
of the Transaction on relationships with employees, other business partners or governmental entities; the difficulty of predicting the
timing or outcome of FDA approvals or actions, if any; the impact of competitive products and pricing; that Alfasigma may not realize
the potential benefits of the Transaction; other business effects, including the effects of industry, economic or political conditions
outside of the companies’ control; Transaction costs; actual or contingent liabilities; and other risks listed under the heading
“Risk Factors” in the Company’s periodic reports filed with the U.S. Securities and Exchange Commission, including current
reports on Form 8-K, quarterly reports on Form 10-Q, annual reports on Form 10-K, as well as the Schedule 14D-9 to be filed by the Company
and the Schedule TO and related tender offer documents to be filed by Alfasigma and Interstellar Acquisition Inc., a wholly owned subsidiary
of Alfasigma. You should not place undue reliance on these statements. All forward-looking statements are based on information currently
available to the Company and Alfasigma, and the Company and Alfasigma disclaim any obligation to update the information contained in this
Current Report on Form 8-K as new information becomes available.
SIGNATURES
Pursuant to the requirements
of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto
duly authorized.
| |
Intercept Pharmaceuticals, Inc. |
| |
|
| |
|
| |
By: |
/s/ Jerome Durso |
| |
|
Name: |
Jerome Durso |
| |
|
Title: |
President and Chief Executive Officer |
| |
|
|
Dated: September 26, 2023
Exhibit 2.1
EXECUTION VERSION
AGREEMENT AND PLAN OF MERGER
among:
Intercept
Pharmaceuticals, Inc.,
a Delaware corporation;
Alfasigma
S.p.A.,
an Italian società per azioni; and
Interstellar
Acquisition Inc.,
a Delaware corporation
Dated as of September 26, 2023
Table of Contents
| |
|
|
| |
Section 1 |
|
| |
|
|
|
THE OFFER |
|
| |
|
|
| 1.1 |
The Offer |
2 |
| 1.2 |
Company Actions |
5 |
| |
|
|
| |
Section 2 |
|
| |
|
|
|
MERGER TRANSACTION |
|
| |
|
|
| 2.1 |
Merger
of Purchaser into the Company |
6 |
| 2.2 |
Effect of the Merger |
7 |
| 2.3 |
Closing; Effective Time |
7 |
| 2.4 |
Certificate of Incorporation and Bylaws; Directors and Officers |
7 |
| 2.5 |
Conversion of Shares |
8 |
| 2.6 |
Surrender of Certificates; Stock Transfer Books |
9 |
| 2.7 |
Dissenters’ Rights |
11 |
| 2.8 |
Treatment of Company Equity Awards |
12 |
| 2.9 |
Further Action |
13 |
| |
|
|
| |
Section 3 |
|
| |
|
|
| |
REPRESENTATIONS AND WARRANTIES OF THE COMPANY |
|
| |
|
|
| 3.1 |
Due Organization; Subsidiaries, Etc. |
14 |
| 3.2 |
Certificate of Incorporation and Bylaws |
14 |
| 3.3 |
Capitalization, Etc. |
14 |
| 3.4 |
SEC Filings; Financial Statements |
16 |
| 3.5 |
Absence of Changes; No Material Adverse Effect |
19 |
| 3.6 |
Title to Assets |
19 |
| 3.7 |
Real Property |
19 |
| 3.8 |
Intellectual Property |
20 |
| 3.9 |
Contracts |
22 |
| 3.10 |
Liabilities |
25 |
| 3.11 |
Compliance with Legal Requirements |
26 |
| 3.12 |
Regulatory Matters |
26 |
| 3.13 |
Certain Business Practices |
28 |
| 3.14 |
Governmental Authorizations |
28 |
| 3.15 |
Tax Matters |
29 |
| 3.16 |
Employee Matters |
31 |
| 3.17 |
Benefit Plans |
32 |
| 3.18 |
Environmental Matters |
34 |
| 3.19 |
Insurance |
34 |
| 3.20 |
Legal Proceedings; Orders |
34 |
| 3.21 |
Authority; Binding Nature of Agreement |
35 |
| 3.22 |
Takeover Laws and CFIUS Matters |
35 |
| 3.23 |
Non-Contravention; Consents |
36 |
| 3.24 |
Opinion of Financial Advisors |
37 |
| 3.25 |
Brokers and Other Advisors |
37 |
| 3.26 |
Transactions with Related Persons |
37 |
| 3.27 |
Standstill |
37 |
| |
|
|
| |
Section 4 |
|
| |
|
|
| |
REPRESENTATIONS AND WARRANTIES OF PARENT AND PURCHASER |
|
| |
|
|
| 4.1 |
Due Organization |
37 |
| 4.2 |
Purchaser |
38 |
| 4.3 |
Authority; Binding Nature of Agreement |
38 |
| 4.4 |
Non-Contravention; Consents |
38 |
| 4.5 |
Disclosure |
39 |
| 4.6 |
Absence of Litigation |
39 |
| 4.7 |
Funds |
39 |
| 4.8 |
Ownership of Shares |
39 |
| 4.9 |
Acknowledgement by Parent and Purchaser |
40 |
| 4.10 |
Brokers and Other Advisors |
40 |
| |
|
|
| |
Section 5 |
|
| |
|
|
| |
CERTAIN COVENANTS OF THE COMPANY |
|
| |
|
|
| 5.1 |
Access and Investigation |
40 |
| 5.2 |
Operation of the Acquired Corporations’ Business |
41 |
| 5.3 |
No Solicitation |
46 |
| |
|
|
| |
Section 6 |
|
| |
|
|
| |
ADDITIONAL COVENANTS OF THE PARTIES |
|
| |
|
|
| 6.1 |
Company Board Recommendation |
48 |
| 6.2 |
Filings, Consents and Approvals |
50 |
| 6.3 |
Employee Benefits |
52 |
| 6.4 |
Indemnification of Officers and Directors |
53 |
| 6.5 |
Stockholder Litigation |
55 |
| 6.6 |
Additional Agreements |
55 |
| 6.7 |
Disclosure |
56 |
| 6.8 |
Takeover Laws |
56 |
| 6.9 |
Section 16 Matters |
56 |
| 6.10 |
Rule 14d-10 Matters |
57 |
| 6.11 |
Stock Exchange Delisting; Deregistration |
57 |
| 6.12 |
Convertible Notes |
57 |
| 6.13 |
UK Tax Ruling |
57 |
| |
|
|
| |
Section 7 |
|
| |
|
|
| |
CONDITIONS PRECEDENT TO THE MERGER |
|
| |
|
|
| 7.1 |
No Restraints |
58 |
| 7.2 |
Consummation of Offer |
58 |
| |
|
|
| |
Section 8 |
|
| |
|
|
| |
TERMINATION |
|
| |
|
|
| 8.1 |
Termination |
58 |
| 8.2 |
Effect of Termination |
60 |
| 8.3 |
Expenses; Termination Fees |
60 |
| |
|
|
| |
Section 9 |
|
| |
|
|
| |
MISCELLANEOUS PROVISIONS |
|
| |
|
|
| 9.1 |
Amendment |
62 |
| 9.2 |
Waiver |
62 |
| 9.3 |
No Survival of Representations and Warranties |
62 |
| 9.4 |
Entire Agreement; Counterparts |
62 |
| 9.5 |
Applicable Legal Requirements; Jurisdiction; Specific Performance; Remedies |
63 |
| 9.6 |
Assignability |
64 |
| 9.7 |
No Third Party Beneficiaries |
64 |
| 9.8 |
Transfer Taxes |
64 |
| 9.9 |
Notices |
65 |
| 9.10 |
Severability |
66 |
| 9.11 |
Obligation of Parent |
66 |
| 9.12 |
Construction |
66 |
| Exhibits |
|
| |
|
| Exhibit A |
Certain Definitions |
| |
|
| Annexes |
|
| |
|
| Annex I |
Conditions to the Offer |
| Annex II |
Form of Certificate of Incorporation of the Surviving Corporation |
AGREEMENT AND PLAN OF MERGER
THIS
AGREEMENT AND PLAN OF MERGER (“Agreement”) is made and entered into as of September 26, 2023, by and
among: Alfasigma S.p.A., an Italian società per azioni (“Parent”); Interstellar Acquisition Inc., a Delaware corporation and a
wholly owned subsidiary of Parent (“Purchaser”); and Intercept Pharmaceuticals, Inc., a Delaware corporation
(the “Company”). Certain capitalized terms used in this Agreement are defined in Exhibit A.
INTRODUCTION
On the terms and subject to the conditions of
this Agreement, Parent will cause Purchaser to commence a tender offer (as it may be amended from time to time as permitted under this
Agreement, the “Offer”) to acquire all of the outstanding shares of Company Common Stock (the “Shares”),
other than the Excluded Shares, for $19.00 per share, net to the seller in cash, without interest (such amount, or any higher amount
per Share paid pursuant to the Offer, and as any such amount may be adjusted in accordance with Section 1.1(g), being the
“Offer Price”), and subject to any withholding of Taxes, and as soon as practicable following the consummation of
the Offer, Purchaser will be merged with and into the Company in the manner provided by Section 251(h) of the DGCL (the “Merger”),
with the Company continuing as the surviving corporation in the Merger (the “Surviving Corporation”), whereupon (i) each
issued and outstanding Share as of the Effective Time (other than Excluded Shares and Dissenting Shares) shall be converted into the
right to receive the Offer Price, in cash, without interest, and subject to any withholding of Taxes, and (ii) the Company shall
become a wholly owned Subsidiary of Parent as a result of the Merger.
The board of directors of the Company (the “Board
of Directors”) has, by a vote of members participating in the decision, unanimously (i) determined that this Agreement
and the Transactions, including the Offer and the Merger, are fair to, and in the best interest of, the Company and its stockholders,
(ii) declared it advisable to enter into this Agreement, (iii) approved the execution, delivery and performance by the Company
of this Agreement and the consummation of the Transactions, including the Offer and the Merger, (iv) resolved that the Merger shall
be effected under Section 251(h) of the DGCL, and (v) resolved to recommend that the stockholders of the Company accept
the Offer and tender their Shares to Purchaser pursuant to the Offer (the “Company Board Recommendation”), in each
case, on the terms and subject to the conditions of this Agreement.
The board of directors of each of Parent and Purchaser
have approved this Agreement and declared it advisable for Parent and Purchaser, respectively, to enter into this Agreement.
AGREEMENT
The Parties to this Agreement, intending to be
legally bound, agree as follows:
Section 1
THE
OFFER
1.1 The
Offer.
(a) Commencement
of the Offer. Provided that this Agreement shall not have been terminated in accordance with Section 8, as promptly as
practicable after the date of this Agreement but in no event more than ten (10) business days after the date of this Agreement (subject
to (i) the Company having timely provided any information required to be provided by it pursuant to Sections 1.1(e) and
1.2(b) and (ii) the Company being prepared, in accordance with Section 1.2(a), to file with the SEC, and
to disseminate to holders of Shares, the Schedule 14D-9 on the Offer Commencement Date), Purchaser shall commence (within the meaning
of Rule 14d-2 under the Exchange Act) the Offer to purchase all of the outstanding Shares (other than Shares to be cancelled pursuant
to Sections 2.5(a)(i) and 2.5(a)(ii) (collectively, the “Excluded Shares”)), at a price per
Share equal to the Offer Price, net to the seller in cash, without interest, and subject to any withholding of Taxes in accordance with
Section 2.6(e).
(b) Terms
and Conditions of the Offer. The obligations of Purchaser to accept for payment, and pay for, any Shares validly tendered pursuant
to the Offer are subject only to the terms and conditions set forth in this Agreement, including the Minimum Condition, the Termination
Condition and the other conditions set forth in Annex I (collectively, the “Offer Conditions”). The Offer shall
be made by means of an offer to purchase (the “Offer to Purchase”) that contains the Offer Conditions and the other
terms set forth in this Agreement. Purchaser expressly reserves the right, to the extent permitted by applicable Legal Requirements,
to (i) increase the Offer Price, (ii) waive any Offer Condition and (iii) make any other changes in the terms and conditions
of the Offer not inconsistent with the terms of this Agreement; provided, however, notwithstanding anything to the contrary contained
in this Agreement, without the prior written consent of the Company, Parent and Purchaser shall not (A) decrease the Offer Price
(other than in a manner required by Section 1.1(g)), (B) change the form of consideration payable in the Offer, (C) decrease
the maximum number of Shares sought to be purchased in the Offer, (D) impose conditions or requirements to the Offer in addition
to the Offer Conditions, (E) amend, modify or waive the Minimum Condition, Termination Condition or the conditions set forth in
clause (f) or (g) of Annex I, (F) otherwise amend or modify any of the other terms of the Offer in
a manner that would adversely affect any holder of Shares in its capacity as such (provided that Purchaser expressly reserves the right
but shall not be obligated to waive any of the Offer Conditions to the extent permitted hereunder), (G) except as provided in Sections
1.1(c) or 1.1(d), terminate the Offer or accelerate, extend or otherwise change the Expiration Date or (H) provide
any “subsequent offering period” (or any extension thereof) within the meaning of Rule 14d-11 promulgated under the
Exchange Act. The Offer may not be withdrawn prior to the Expiration Date (or any rescheduled Expiration Date) of the Offer, unless this
Agreement is terminated in accordance with Section 8.
(c) Expiration
and Extension of the Offer. The Offer shall initially be scheduled to expire at one minute after 11:59 p.m. Eastern Time on
the date that is twenty (20) business days (determined as set forth in Rule 14d-1(g)(3) and Rule 14e-1(a) under the
Exchange Act) from the Offer Commencement Date (unless otherwise agreed to in writing by Parent and the Company) (the “Initial
Expiration Date”, and such date or such subsequent date to which the Initial Expiration Date of the Offer is extended in accordance
with the terms of this Agreement, the “Expiration Date”). Notwithstanding anything to the contrary contained in this
Agreement, but subject to the Parties’ respective termination rights under Section 8: (i) if, as of the then-scheduled
Expiration Date, any Offer Condition is not satisfied and has not been waived by Purchaser or Parent (to the extent waivable by Purchaser
or Parent), Purchaser may, in its discretion (and without the consent of the Company or any other Person), extend the Offer on one or
more occasions, for an additional period of up to ten (10) business days per extension, to permit such Offer Condition to be satisfied;
(ii) Purchaser shall extend the Offer from time to time for: (A) any period required by any Legal Requirement, any interpretation
or position of the SEC, the staff thereof or Nasdaq applicable to the Offer; and (B) periods of up to ten (10) business days
per extension, until any waiting period (and any extension thereof) applicable to the consummation of the Offer under the HSR Act and
any foreign Antitrust Laws shall have expired or been terminated; (iii) if, as of the then-scheduled Expiration Date, any Offer
Condition (other than the Minimum Condition) is not satisfied and has not been waived (to the extent waivable by Purchaser or Parent),
at the request of the Company, Purchaser shall extend the Offer on one or more occasions for an additional period of up to ten (10) business
days per extension, to permit such Offer Condition to be satisfied; and (iv) if, as of the then-scheduled Expiration Date, all Offer
Conditions have been satisfied or waived (if permitted hereunder, and other than any such Offer Conditions that by their nature are to
be satisfied at the expiration of the Offer (provided such conditions would be satisfied were the expiration of the Offer to occur at
such time)), except that the Minimum Condition has not been satisfied as of the then-scheduled Expiration Date, then at the request of
the Company, Purchaser shall, and in any event Purchaser in its sole discretion may, extend the Offer on one or more occasions for an
additional period of up to ten (10) business days per extension, to permit such Minimum Condition to be satisfied, it being understood
and agreed that the Purchaser shall not be required to extend the Offer pursuant to this clause (iv) on more than two (2) occasions,
but may, in its sole and absolute discretion elect to do so; provided, however, that in no event shall Purchaser: (1) be
required to extend the Offer beyond the earlier to occur of (x) the termination of this Agreement in compliance with Section 8
and (y) the End Date (such earlier occurrence, the “Extension Deadline”); or (2) be permitted to extend
the Offer beyond the Extension Deadline without the prior written consent of the Company. Purchaser shall not terminate the Offer, or
permit the Offer to expire, prior to the Extension Deadline without the prior written consent of the Company.
(d) Termination
of Offer. Nothing in this Section 1.1 shall be deemed to impair, limit or otherwise restrict in any manner the right
of the Company, Parent or Purchaser to terminate this Agreement pursuant to Section 8. In the event that this Agreement is
terminated pursuant to Section 8, Purchaser shall immediately, irrevocably and unconditionally terminate the Offer and shall
not acquire any Shares pursuant to the Offer. If the Offer is terminated or withdrawn by Purchaser in accordance with the terms of this
Agreement, Purchaser shall immediately return, and shall cause any depository acting on behalf of Purchaser to return, in accordance
with applicable Legal Requirements, all tendered Shares to the registered holders thereof.
(e) Offer
Documents. As promptly as practicable on the Offer Commencement Date (subject to (i) the Company having timely provided any
information required to be provided by it pursuant to Sections 1.1(e) and 1.2(b) and (ii) the Company being
prepared, in accordance with Section 1.2(a), to file with the SEC, and to disseminate to holders of Shares, the Schedule
14D-9 promptly thereafter on the Offer Commencement Date), Parent and Purchaser shall (i) file with the SEC a tender offer statement
on Schedule TO with respect to the Offer (together with any exhibits, amendments or supplements thereto, the “Offer Documents”)
that will contain or incorporate by reference the Offer to Purchase and form of the related letter of transmittal and (ii) cause
the Offer to Purchase and related documents to be disseminated to holders of Shares as and to the extent required by applicable Legal
Requirements. Parent and Purchaser agree that they shall cause the Offer Documents filed by either Parent or Purchaser with the SEC (x) to
comply in all material respects with the Exchange Act and other applicable Legal Requirements and (y) to not contain any untrue
statement of a material fact or omit to state any material fact required to be stated therein or necessary in order to make the statements
therein, in light of the circumstances under which they were made, not misleading; provided, however, that no covenant
is made by Parent or Purchaser with respect to information supplied by or on behalf of the Acquired Corporations for inclusion or incorporation
by reference in the Offer Documents. Each of Parent, Purchaser and the Company agrees to respond promptly to any comments (including
oral comments) of the SEC or its staff and to promptly correct any information provided by it for use in the Offer Documents if and to
the extent that such information shall have become false or misleading in any material respect, and Parent and Purchaser further agree
to take all steps necessary to cause the Offer Documents as so corrected to be filed with the SEC and to be disseminated to holders of
Shares, in each case as and to the extent required by applicable Legal Requirements. The Company consents to the inclusion of the Company
Board Recommendation in the Offer Documents. The Company shall promptly furnish or otherwise make available to Parent and Purchaser or
Parent’s legal counsel all information concerning the Acquired Corporations and the Company’s stockholders that may be required
or reasonably requested in connection with any action contemplated by this Section 1.1(e). The Company and its counsel shall
be given reasonable opportunity to review and comment on the Offer Documents (including any response to any comments (including oral
comments) of the SEC or its staff with respect thereto) prior to the filing thereof with the SEC, and Parent and Purchaser shall give
reasonable and good faith consideration to any such comments made by the Company or its counsel. Parent and Purchaser agree to provide
the Company and its counsel with any comments (including oral comments) Parent, Purchaser or their counsel may receive from the SEC or
its staff with respect to the Offer Documents promptly after receipt of those comments (including oral comments).
(f) Funds.
Without limiting the generality of Section 9.11, Parent shall cause to be provided to Purchaser, on a timely basis, all of
the funds necessary to purchase all Shares that Purchaser becomes obligated to purchase pursuant to the Offer, and shall cause Purchaser
to perform, on a timely basis, all of Purchaser’s obligations under this Agreement. Parent and Purchaser shall, and each of Parent
and Purchaser shall ensure that all of their respective Affiliates shall, tender any Shares held by them into the Offer.
(g) Adjustments.
If, between the date of this Agreement and the Offer Acceptance Time, the outstanding Shares are changed into a different number or class
of shares by reason of any stock split, division or subdivision of shares, stock dividend, reverse stock split, consolidation of shares,
reclassification, recapitalization or other similar transaction, then the Offer Price shall be ratably and appropriately adjusted; provided,
that nothing in this Section 1.1(g) shall be construed to permit the Acquired Corporations or any other Person to take
any action that is otherwise prohibited by the terms of the Agreement.
(h) Acceptance.
Subject only to the satisfaction or, to the extent waivable by Purchaser or Parent, waiver by Purchaser or Parent of each of the Offer
Conditions, Purchaser shall (i) immediately after the expiration of the Offer accept for payment all Shares tendered (and not validly
withdrawn) pursuant to the Offer (the time of such acceptance, the “Offer Acceptance Time”) and (ii) promptly
after the Offer Acceptance Time pay for such Shares.
(i) Transfer
Taxes. If the payment of the Offer Price is to be made to a Person other than the Person in whose name the tendered Shares are registered
on the stock transfer books of the Company, it shall be a condition of payment that the Person requesting such payment shall have paid
all transfer and other similar Taxes required by reason of the payment of the Offer Price to a Person other than the registered holder
of the Shares tendered, or shall have established to the satisfaction of the Purchaser that such Taxes either have been paid or are not
applicable. None of Parent, Purchaser or the Surviving Corporation shall have any liability for the transfer and other similar Taxes
described in this Section 1.1(i) under any circumstance.
1.2 Company
Actions.
(a) Schedule
14D-9. As promptly as practicable on the Offer Commencement Date, following the filing of the Offer Documents, the Company shall
(i) file with the SEC a Tender Offer Solicitation/Recommendation Statement on Schedule 14D-9 (together with any exhibits, amendments
or supplements thereto, the “Schedule 14D-9”) that, subject to Section 6.1(b), shall reflect the Company
Board Recommendation and include the notice and other information required by Section 262(d)(2) of the DGCL and (ii) cause
the Schedule 14D-9 and related documents to be disseminated to holders of Shares as and to the extent required by applicable Legal Requirements,
including by setting the Stockholder List Date as the record date for purposes of receiving the notice required by Section 262(d)(2) of
the DGCL. The Company agrees that it shall cause the Schedule 14D-9 (x) to comply in all material respects with the Exchange Act
and other applicable Legal Requirements and (y) to not contain any untrue statement of a material fact or omit to state any material
fact required to be stated therein or necessary in order to make the statements therein, in light of the circumstances under which they
were made, not misleading; provided, however, that no covenant is made by the Company with respect to information supplied by
or on behalf of Parent or Purchaser for inclusion or incorporation by reference in the Schedule 14D-9. Each of Parent, Purchaser and
the Company agrees to respond promptly to any comments (including oral comments) of the SEC or its staff and to promptly correct any
information provided by it for use in the Schedule 14D-9 if and to the extent that such information shall have become false or misleading
in any material respect, and the Company further agrees to take all steps necessary to cause the Schedule 14D-9 as so corrected to be
filed with the SEC and to be disseminated to holders of Shares, in each case as and to the extent required by applicable Legal Requirements.
Parent and Purchaser shall promptly furnish or otherwise make available to the Company or the Company’s legal counsel all information
concerning Parent or Purchaser that may be required or reasonably requested in connection with any action contemplated by this Section 1.2(a).
Parent and its counsel shall be given reasonable opportunity to review and comment on the Schedule 14D-9 (including any response to any
comments (including oral comments) of the SEC or its staff with respect thereto) prior to the filing thereof with the SEC, and the Company
shall give reasonable and good faith consideration to any such comments made by Parent or its counsel. The Company agrees to provide
Parent and its counsel with any comments (including oral comments) the Company or its counsel may receive from the SEC or its staff with
respect to the Schedule 14D-9 promptly after receipt of those comments (including oral comments).
(b) Stockholder
Lists. The Company shall promptly furnish Parent with a list of its stockholders, mailing labels and any available listing or computer
file containing the names and addresses of all record holders of Shares and lists of securities positions of Shares held in stock depositories,
in each case accurate and complete as of the most recent practicable date, and shall provide to Parent such additional information (including
updated lists of stockholders, mailing labels and lists of securities positions) and such other assistance as Parent may reasonably request
in connection with the Offer and the Merger (the date of the list used to determine the Persons to whom the Offer Documents and the Schedule
14D-9 are first disseminated, which date shall not be more than ten (10) calendar days prior to the date the Offer Documents and
the Schedule 14D-9 are first disseminated, the “Stockholder List Date”). Except for such steps as are necessary to
disseminate the Offer Documents and any other documents necessary to consummate the Transactions, Parent and Purchaser and their Representatives
shall hold in confidence the information contained in any such labels, listings and files, shall use such information only in connection
with the Offer and the Merger and, if this Agreement shall be terminated, shall, upon request by the Company, deliver, and shall use
their reasonable best efforts to cause their agents to deliver, to the Company (or destroy) all copies and any extracts or summaries
from such information then in their possession or control, and, if requested by the Company, promptly certify to the Company in writing
that all such material has been returned or destroyed.
(c) Share
Registry. The Company shall register (and shall instruct its transfer agent to register) the transfer of the Shares accepted for
payment by Purchaser in the Offer effective immediately after the Offer Acceptance Time.
Section 2
MERGER
TRANSACTION
2.1 Merger
of Purchaser into the Company. Upon the terms and subject to the conditions set forth in this
Agreement and in accordance with Section 251(h) of the DGCL, at the Effective Time, the Company and Parent shall consummate
the Merger, whereby Purchaser shall be merged with and into the Company, the separate existence of Purchaser shall cease, and the Company
will continue as the Surviving Corporation.
2.2 Effect
of the Merger. The Merger shall have the effects set forth in this Agreement and in the applicable
provisions of the DGCL. Without limiting the generality of the foregoing, at the Effective Time, all of the property, rights, privileges,
immunities, powers and franchises of the Company and Purchaser shall vest in the Surviving Corporation, and all of the debts, liabilities
and duties of the Company and Purchaser shall become the debts, liabilities and duties of the Surviving Corporation.
2.3 Closing;
Effective Time.
(a) Unless
this Agreement shall have been terminated pursuant to Section 8, and unless otherwise mutually agreed in writing among the
Company, Parent and Purchaser, the consummation of the Merger (the “Closing”) shall take place via the electronic
exchange of documents by the Parties, as soon as practicable (and in no event later than one business day) following the Offer Acceptance
Time except if the conditions set forth in Section 7.1 shall not be satisfied or, to the extent permissible by applicable
Legal Requirements, waived as of such date, in which case on the first business day on which all conditions set forth in Section 7.1
are satisfied or, to the extent permissible by applicable Legal Requirements, waived, unless another date or place is agreed to in
writing by the Company and Parent. The date on which the Closing occurs is referred to in this Agreement as the “Closing Date”.
(b) Subject
to the provisions of this Agreement (including Section 1.2(c)), as soon as practicable on the Closing Date, the Company and
Purchaser shall file or cause to be filed a certificate of merger with the Secretary of State of the State of Delaware with respect to
the Merger, in such form reasonably agreed upon between the Parties and as required by, and executed and acknowledged in accordance with,
the relevant provisions of the DGCL, and the Parties shall take all such further actions as may be required by applicable Legal Requirements
to make the Merger effective. The Merger shall become effective upon the date and time of the filing of that certificate of merger with
the Secretary of State of the State of Delaware or such later date and time as is agreed upon in writing by the Parties and specified
in the certificate of merger (such date and time the Merger is effective, the “Effective Time”).
2.4 Certificate
of Incorporation and Bylaws; Directors and Officers.
(a) As
of the Effective Time, the certificate of incorporation of the Company shall by virtue of the Merger and without any further action,
be amended and restated to read in its entirety as set forth on Annex II and, as so amended and restated, shall be the certificate
of incorporation of the Surviving Corporation until thereafter changed or amended as provided therein or by applicable Legal Requirements,
subject to Section 6.4(a).
(b) As
of the Effective Time, the bylaws of the Surviving Corporation shall be amended and restated to conform to the bylaws of Purchaser as
in effect immediately prior to the Effective Time, until thereafter changed or amended as provided therein or by applicable Legal Requirements,
subject to Section 6.4(a), except that references to the name of Purchaser shall be replaced by references to the name of
the Surviving Corporation.
(c) As
of the Effective Time, the directors and officers of the Surviving Corporation shall be the respective individuals who served as the
directors and officers of Purchaser as of immediately prior to the Effective Time, until their respective successors are duly elected
and qualified, or their earlier death, resignation or removal.
2.5 Conversion
of Shares.
(a) At
the Effective Time, by virtue of the Merger and without any further action on the part of Parent, Purchaser, the Company or any stockholder
of the Company:
(i) any
Shares held immediately prior to the Effective Time by the Company (or held in the Company’s treasury) shall be cancelled and retired
and shall cease to exist, and no consideration shall be delivered in exchange therefor;
(ii) any
Shares held immediately prior to the Effective Time by Parent, Purchaser or any other direct or indirect wholly owned Subsidiary of Parent
or Purchaser (including, for the avoidance of doubt, any shares acquired by Purchaser in the Offer) shall be cancelled and retired and
shall cease to exist, and no consideration shall be delivered in exchange therefor;
(iii) except
as provided in clauses (i) and (ii) above and subject to Section 2.5(b), each Share outstanding immediately
prior to the Effective Time (other than any Dissenting Shares, which shall have only those rights set forth in Section 2.7)
shall be converted into the right to receive the Offer Price (the “Merger Consideration”), without any interest thereon
and subject to any withholding of Taxes in accordance with Section 2.6(e); and
(iv) each
share of the common stock of Purchaser then outstanding shall be converted into one share of common stock of the Surviving Corporation.
From and after the Effective Time, subject to this Section 2.5(a),
all Shares shall no longer be outstanding and shall automatically be cancelled and shall cease to exist, and each applicable holder of
such Shares (other than Dissenting Shares) shall cease to have any rights with respect thereto, except the right to receive the Merger
Consideration therefor upon the surrender of such Shares in accordance with Section 2.6, or, in the case of Dissenting Shares,
the rights set forth in Section 2.7.
(b) If,
between the date of this Agreement and the Effective Time, the outstanding Shares are changed into a different number or class of shares
by reason of any stock split, division or subdivision of shares, stock dividend, reverse stock split, consolidation of shares, reclassification,
recapitalization or other similar transaction, then the Merger Consideration shall be ratably and appropriately adjusted; provided,
that nothing in this Section 2.5(b) shall be construed to permit the Acquired Corporations or any other Person to take
any action that is otherwise prohibited by the terms of the Agreement.
2.6 Surrender
of Certificates; Stock Transfer Books.
(a) Prior
to the commencement of the Offer, Parent shall designate a bank or trust company reasonably acceptable to the Company to act as agent
(the “Depository Agent”) for the holders of Shares to receive the aggregate Offer Price to which holders of such Shares
shall become entitled pursuant to Section 1.1(b) and to act as agent (the “Paying Agent”) for the
holders of Shares to receive the aggregate Merger Consideration to which holders of such shares shall become entitled pursuant to Section 2.5.
Promptly after the Offer Acceptance Time, Parent shall deposit, or shall cause to be deposited, with the Depository Agent cash sufficient
to make the payment of the aggregate Offer Price payable pursuant to Section 1.1(h). Parent shall deposit, or shall cause
to be deposited, with the Paying Agent cash sufficient to pay the aggregate Merger Consideration payable pursuant to Section 2.5
(together with the amount deposited pursuant the immediately preceding sentence, the “Payment Fund”). The Payment
Fund shall not be used for any purpose other than to pay the aggregate Offer Price in the Offer and the aggregate Merger Consideration
in the Merger. The Payment Fund shall be invested by the Depository Agent or the Paying Agent as directed by Parent; provided
that (i) no such investment or losses thereon shall relieve Parent from making the payments required by Section 1.1(h) and
Section 2.5 and (ii) no such investment shall have maturities exceeding three (3) months. To the extent Parent
becomes aware that (i) there are any losses with respect to any such investments or (ii) the Payment Fund has diminished for
any reason below the level required for the Depository Agent or Paying Agent to make prompt cash payment pursuant to Section 1.1(h) and
Section 2.5, as applicable, Parent or the Surviving Corporation shall promptly replace or restore the cash in the Payment
Fund so as to ensure that the Payment Fund is, at all times during the duration of the Payment Fund, maintained at a level sufficient
for the Depository Agent and the Paying Agent to promptly make such payments pursuant to Section 1.1(h) and Section 2.5,
as applicable.
(b) Promptly
after the Effective Time (but in no event later than three (3) business days thereafter), the Surviving Corporation shall cause
to be delivered to each Person who was, at the Effective Time, a holder of record of (i) Shares represented by a certificate evidencing
such Shares (“Certificated Shares”) or (ii) Book-Entry Shares, who, in each case of (i) and (ii), was entitled
to receive the Merger Consideration pursuant to Section 2.5, (A) a form of letter of transmittal, which shall be in
reasonable and customary form and shall specify that delivery shall be effected, and risk of loss and title to the Certificated Shares
shall pass, only upon proper delivery of the certificates evidencing such Certificated Shares (the “Certificates”)
(or affidavits of loss in lieu thereof in accordance with Section 2.6(f), if applicable) to the Paying Agent, or a customary
agent’s message with respect to Book-Entry Shares, and (B) instructions for use in effecting the surrender of the Certificates
or Book-Entry Shares in exchange for the Merger Consideration issuable and payable in respect of such Shares pursuant to Section 2.5.
Upon surrender to the Paying Agent of Certificates (or affidavits of loss in lieu thereof in accordance with Section 2.6(f),
if applicable) or Book-Entry Shares, together with such letter of transmittal in the case of Certificates, duly completed and validly
executed in accordance with the instructions thereto, and such other documents as may be required pursuant to the instructions, the holder
of such Certificated Shares or Book-Entry Shares shall be entitled to receive in exchange therefor the Merger Consideration for each
Certificated Share or Book-Entry Share, and such Certificates and Book-Entry Shares shall then be cancelled. No interest shall accrue
or be paid on the Merger Consideration payable upon the surrender of any Certificates or Book-Entry Shares for the benefit of the holder
thereof. If the payment of any Merger Consideration is to be made to a Person other than the Person in whose name the surrendered Certificates
formerly evidencing the Certificated Shares is registered on the stock transfer books of the Company, it shall be a condition of payment
that the Certificate so surrendered shall be endorsed properly or otherwise be in proper form for transfer and that the Person requesting
such payment shall have paid all transfer and other similar Taxes required by reason of the payment of the Merger Consideration to a
Person other than the registered holder of the Certificate surrendered, or shall have established to the satisfaction of the Surviving
Corporation that such transfer or other Taxes either have been paid or are not applicable. None of Parent, Purchaser or the Surviving
Corporation shall have any liability for the transfer and other similar Taxes described in this Section 2.6(b) under
any circumstance. Payment of the applicable Merger Consideration with respect to Book-Entry Shares shall only be made to the Person in
whose name such Book-Entry Shares are registered. Until surrendered as contemplated by this Section 2.6, each Certificated
Share and Book-Entry Share (in each case, other than Dissenting Shares) shall be deemed at any time after the Effective Time to represent
only the right to receive the applicable Merger Consideration as contemplated by Section 2.5.
(c) At
any time following twelve (12) months after the Effective Time, Parent shall be entitled to require the Paying Agent to deliver to it
any funds (with respect to the aggregate Merger Consideration to which holders of Shares shall become entitled pursuant to Section 2.5)
which had been made available to the Paying Agent and not disbursed to holders of Certificated Shares or Book-Entry Shares (including
all interest and other income received by the Paying Agent in respect of all funds made available to it), and, thereafter, such holders
shall be entitled to look to the Surviving Corporation (subject to abandoned property, escheat and other similar Legal Requirements)
only as general creditors thereof with respect to the Merger Consideration that may be payable upon due surrender of the Certificates
or Book-Entry Shares held by them, without any interest thereon. Notwithstanding the foregoing, neither the Surviving Corporation nor
the Paying Agent shall be liable to any holder of Certificated Shares or Book-Entry Shares for the Merger Consideration delivered in
respect of such Share to a public official pursuant to any abandoned property, escheat or other similar Legal Requirements. Any amounts
remaining unclaimed by such holders at such time at which such amounts would otherwise escheat to or become property of any Governmental
Body shall become, to the extent permitted by applicable Legal Requirements, the property of the Surviving Corporation or its designee,
free and clear of all claims or interest of any Person previously entitled thereto.
(d) At
the close of business on the day of the Effective Time, the stock transfer books of the Company with respect to the Shares shall be closed
and thereafter there shall be no further registration of transfers of Shares on the records of the Company. From and after the Effective
Time, the holders of the Shares outstanding immediately prior to the Effective Time shall cease to have any rights with respect to such
Shares except as otherwise provided herein or by applicable Legal Requirements. If, after the Effective Time, Certificates or Book-Entry
Shares are presented to the Surviving Corporation for any reason, they shall be cancelled and exchanged as provided in this Agreement.
(e) Each
of the Company, the Surviving Corporation, Parent and Purchaser, and their Affiliates, shall be entitled to deduct and withhold (or cause
the Paying Agent or the Depository Agent to deduct and withhold) from the Offer Price, the Merger Consideration payable to any holder
of the Shares or any holder of Company Options, Company PSUs, Company RSUs or any other consideration otherwise payable pursuant to this
Agreement such amounts as it is required by any Legal Requirement to deduct and withhold with respect to Taxes. Each such withholding
agent shall use commercially reasonable efforts to reduce or eliminate any such withholding. Each such withholding agent shall take all
action that may be necessary to ensure that any such amounts so withheld are timely and properly remitted to the appropriate Governmental
Body. To the extent that amounts are so withheld and timely and properly remitted to the appropriate Governmental Body, such withheld
amounts shall be treated for all purposes of this Agreement as having been paid to the holder of the Shares, holder of Company Options,
Company PSUs, Company RSUs or other recipient of consideration hereunder in respect of which such deduction and withholding was made.
(f) If
any Certificate shall have been lost, stolen or destroyed, upon the making of an affidavit of that fact by the holder of the Certificated
Shares formerly represented by that Certificate, or by a representative of that holder, claiming that Certificate to be lost, stolen
or destroyed and, if required by the Surviving Corporation, the posting by that holder of a bond, in such reasonable amount as Parent
may direct, as indemnity against any claim that may be made against it with respect to such Certificate (which amount shall not exceed
the Merger Consideration payable with respect to such Certificated Shares), the Paying Agent will pay (less any amounts entitled to be
deducted or withheld pursuant to Section 2.6(e)), in exchange for such lost, stolen or destroyed Certificate, the applicable
Merger Consideration to be paid in respect of the Certificated Shares formerly represented by such Certificate, as contemplated by this
Section 2.
2.7 Dissenters’
Rights. Notwithstanding anything to the contrary in this Agreement, any Shares outstanding immediately
prior to the Effective Time which are held by holders who are entitled to appraisal rights under Section 262 of the DGCL and have
properly exercised and perfected their respective demands for appraisal of such Shares in the time and manner provided in Section 262
of the DGCL and, as of the Effective Time, have neither effectively withdrawn nor lost their rights to such appraisal and payment under
the DGCL (the “Dissenting Shares”), shall not be converted into the right to receive the Merger Consideration, but
shall, by virtue of the Merger, be automatically cancelled and no longer outstanding, shall cease to exist and shall be entitled to only
such consideration as shall be determined pursuant to Section 262 of the DGCL; provided that if any such holder shall have
failed to perfect or shall have effectively withdrawn or lost such holder’s right to appraisal and payment under the DGCL, such
holder’s Shares shall be deemed to have been converted as of the Effective Time into the right to receive the Merger Consideration
(less any amounts entitled to be deducted or withheld pursuant to Section 2.6(e)), and such Shares shall no longer be deemed
to be Dissenting Shares. The Company shall give prompt notice to Parent and Purchaser of any demands received by the Company for appraisal
of any Dissenting Shares, withdrawals of such demands and any other instruments served pursuant to Section 262 of the DGCL, in each
case prior to the Effective Time, shall give Parent the opportunity to review any written document to be given to any third party or
Governmental Body in connection therewith, and shall consider in good faith all comments provided by Parent. Parent and Purchaser shall
have the right to direct and participate in all negotiations and proceedings with respect to such demands, and the Company shall not,
without the prior written consent of Parent and Purchaser, settle or offer to settle, or make any payment with respect to, any such demands,
or agree or commit to do any of the foregoing.
2.8 Treatment
of Company Equity Awards.
(a) Except as otherwise agreed by the Parties, at
the Effective Time, each Company Option that is outstanding and unexercised as of immediately prior to the Effective Time, whether or
not vested, and which has a per share exercise price that is less than the Merger Consideration (each, an “In the Money Option”),
shall be cancelled and converted into the right to receive a cash payment equal to (A) the excess of (x) the Merger Consideration
over (y) the exercise price payable per Share of such In the Money Option, multiplied by (B) the total number of Shares subject
to such In the Money Option immediately prior to the Effective Time, less applicable Taxes required to be withheld with respect to such
payment.
(b) At
the Effective Time, each Company Option other than an In the Money Option that is outstanding and unexercised as of immediately prior
to the Effective Time, whether or not vested, shall be cancelled with no consideration payable in respect thereof.
(c) At
the Effective Time, each Company PSU that is outstanding as of immediately prior to the Effective Time shall be cancelled and the holder
thereof shall be entitled to receive a cash payment (without interest) equal to the product of (x) the Merger Consideration and
(y) the number of Shares subject to such Company PSU as of immediately prior to the Effective Time based upon the actual level of
performance determined in accordance with the terms of the applicable Company
PSU award agreement, less applicable Taxes required to be withheld with respect to such payment.
(d) At
the Effective Time, each Company RSU that is outstanding as of immediately prior to the Effective Time shall be converted into the contingent
right (as described in Section 2.8(f)) to receive a cash payment (without interest) equal to the product of (x) the
Merger Consideration and (y) the number of Shares subject to such Company RSU as of immediately prior to the Effective Time, less
applicable Taxes required to be withheld with respect to such payment (the “Company RSU Consideration”).
(e) Payment
of In the Money Options; Company PSUs. As soon as reasonably practicable after the Effective Time (but no later than the second payroll
date after the Effective Time), the Surviving Corporation or its Affiliate, as applicable, shall pay the aggregate consideration payable
in respect of In the Money Options and Company PSUs pursuant to Sections 2.8(a) and 2.8(c) through, to the extent
applicable, the Surviving Corporation’s or its Affiliate’s payroll to the holders of In the Money Options and Company PSUs.
(f) Payment
of Company RSU Consideration. Subject to the applicable holder’s continued service with Parent and its Affiliates (including
the Surviving Corporation and its Subsidiaries), the Company RSU Consideration shall vest and become payable in accordance with vesting
schedule (including any vesting acceleration provisions) that applied to the corresponding portion of the applicable Company RSUs immediately
prior to the Effective Time. The Company RSU Consideration shall otherwise remain subject to the same terms and conditions that were
applicable to the underlying Company RSUs immediately prior to the Effective Time (except for terms rendered inoperative by reason of
the transactions contemplated by this Agreement).
(g) Prior
to the Effective Time, the Company may take all actions that it determines to be appropriate or necessary to (i) effect the transactions
described in this Section 2.8 and (ii) cause the Company Equity Plans (other than the agreements underlying, and the
terms of the Company Equity Plans applicable to, the Company RSU Consideration solely to the extent relevant to the terms and conditions
of this Section 2.8) to terminate at or prior to the Effective Time. The Company shall take all actions necessary to ensure
that from and after the Effective Time none of Parent or the Surviving Corporation shall be required to deliver Shares or other share
capital of the Company to any Person pursuant to or in settlement of Company Equity Awards.
(h) To
the extent a payment made pursuant to the timing set forth in this Section 2.8 would trigger a Tax or penalty under Section 409A
of the Code, such payment shall be made on the earliest date that payment would not trigger such Tax or penalty.
2.9 Further
Action. The Parties agree to take all necessary action to cause the Merger to become effective
in accordance with this Section 2 as soon as practicable following the consummation of the Offer without a meeting of the
Company’s stockholders, as provided in Section 251(h) of the DGCL. If, at any time after the Effective Time, any further
action is reasonably determined by Parent to be necessary or desirable to carry out the purposes of this Agreement or to vest the Surviving
Corporation with full right, title and possession of and to all rights and property of Purchaser and the Company, the officers and directors
of the Surviving Corporation and Parent shall be fully authorized (in the name of Purchaser, in the name of the Company and otherwise)
to take such action.
Section 3
REPRESENTATIONS
AND WARRANTIES OF THE COMPANY
The Company hereby represents and warrants to
Parent and Purchaser as of the date hereof and as of the Closing Date as follows (it being understood that each representation and warranty
contained in this Section 3 is subject to (a) exceptions and disclosures set forth in the section or subsection of the
Company Disclosure Schedule corresponding to the particular section or subsection in this Section 3; (b) any exception
or disclosure set forth in any other section or subsection of the Company Disclosure Schedule to the extent it is reasonably apparent
on its face that such exception or disclosure is applicable to qualify such other section or subsection; and (c) except with respect
to Sections 3.1 (Due Organization; Subsidiaries, Etc.), 3.2 (Certificate of Incorporation and Bylaws), 3.3 (Capitalization,
Etc.), 3.21 (Authority; Binding Nature of Agreement), 3.22 (Takeover Laws and CFIUS Matters), 3.23 (Non-Contravention;
Consents), 3.24 (Opinion of Financial Advisors) or 3.25 (Brokers and Other Advisors), disclosure in the Company SEC Documents
filed on or after January 1, 2022 and publicly available at least three (3) business days prior to the date of this Agreement
(other than any information in the “Risk Factors” or “Forward-Looking Statements” sections of such Company SEC
Documents or other predictive, non-specific, cautionary or forward-looking statements in any other sections of such Company SEC Documents)):
3.1 Due
Organization; Subsidiaries, Etc.
(a) The
Company is a corporation duly organized, validly existing and in good standing under the laws of the State of Delaware, and the Company’s
only Subsidiaries are set forth on Section 3.1(a) of the Company Disclosure Schedule (the Company and each such Subsidiary,
an “Acquired Corporation” and collectively, the “Acquired Corporations”). Each Acquired Corporation
has all necessary power and authority: (i) to conduct its business in the manner in which its business is currently being conducted;
and (ii) to own and use its assets in the manner in which its assets are currently owned and used. Each Acquired Corporation is
qualified or licensed to do business as a foreign corporation, and is in good standing, in each jurisdiction where the nature of its
business requires such qualification or licensing, except where such failure does not have, and would not reasonably be expected to have,
individually or in the aggregate, a Material Adverse Effect.
(b) The
Company owns beneficially and of record all of the outstanding shares of capital stock or ordinary shares of the other Acquired Corporations,
free and clear of all Encumbrances and transfer restrictions, except for Encumbrances or transfer restrictions of general applicability
as may be provided under the Securities Act or applicable securities laws. Except for the shares of capital stock or ordinary shares
of the other Acquired Corporations held by the Company, no Acquired Corporation owns, directly or indirectly, any capital stock or equity
interests in, or subscriptions, options, calls, warrants or rights (whether or not currently exercisable) to acquire, or other securities
convertible into or exchangeable or exercisable for, any capital stock or equity interests of any Entity.
3.2 Certificate
of Incorporation and Bylaws. The Company has delivered or made available to Parent accurate
and complete copies of the certificate of incorporation, bylaws and other charter and organizational documents of each Acquired Corporation,
including all amendments thereto, as in effect on the date hereof. No Acquired Corporation is in material violation of any of the
provisions of its respective certificate of incorporation, bylaws or other charter and organizational documents, as applicable.
3.3 Capitalization,
Etc.
(a) The
authorized capital stock of the Company consists of: (i) 90,000,000 Shares, of which 41,808,235 Shares had been issued and were
outstanding as of the close of business on September 25, 2023 (the “Capitalization Date”); and (ii) 5,000,000
shares of Company Preferred Stock, of which no shares are outstanding. All of the outstanding shares of the capital stock of the Company
have been duly authorized and validly issued, and are fully paid and nonassessable.
(b) All
of the outstanding shares of the capital stock or ordinary shares of the Company’s Subsidiaries have been duly authorized and validly
issued, and are fully paid and nonassessable. The Company owns all of the authorized and outstanding capital stock of the Company’s
Subsidiaries.
(c) (i) None
of the outstanding shares of capital stock of the Acquired Corporations are entitled or subject to any preemptive right, right of repurchase
or forfeiture, right of participation, right of maintenance or any similar right; (ii) none of the outstanding shares of capital
stock of the Acquired Corporations are subject to any right of first refusal in favor of any Acquired Corporation; (iii) there are
no outstanding bonds, debentures, notes or other indebtedness of any Acquired Corporation having a right to vote on any matters on which
the stockholders of the Acquired Corporations have a right to vote; and (iv) there is no Contract relating to the voting or registration
of, or restricting any Person from purchasing, selling, pledging or otherwise disposing of (or from granting any option or similar right
with respect to), any shares of capital stock of the Acquired Corporations. No Acquired Corporation is under any obligation, or bound
by any Contract pursuant to which it may become obligated, to repurchase, redeem or otherwise acquire any outstanding shares of capital
stock of the Acquired Corporations. The Shares constitute the only outstanding class of securities of the Company registered under the
Securities Act. There are no voting trusts or other Contracts with respect to the voting of any Shares.
(d) As
of the close of business on the Capitalization Date: (i) 2,427,133 Shares were subject to issuance pursuant to Company Options granted
and outstanding under the Company Equity Plans, (ii) 2,028,991 Shares were subject to issuance pursuant to Company RSUs granted
and outstanding under the Company Equity Plans, (iii) 511,800 Shares were subject to issuance pursuant to Company PSUs granted and
outstanding under the Company Equity Plans (assuming attainment of performance conditions measured at the target level of performance),
(iii) 3,545,974 Shares were reserved for future issuance under Company Equity Plans and (iv) 8,016,812 Shares were reserved
for future issuance in connection with any conversions of the Convertible Notes. Other than as set forth in this Section 3.3(d),
there are no issued, reserved for issuance, outstanding or authorized stock option, stock appreciation, phantom stock, profit participation
or similar rights or equity-based awards with respect the Company.
(e) Each
Company Option (i) was granted in compliance with all applicable Legal Requirements and all of the terms and conditions of the Company
Equity Plan pursuant to which it was issued, (ii) has an exercise price per Share equal to or greater than the fair market value
of a Share on the date of such grant, (iii) has a grant date identical to the date on which the Board of Directors or compensation
committee thereof actually awarded such Company Option and (iv) does not trigger any liability for the holder thereof under Section 409A
of the Code.
(f) There
are no issued, reserved for issuance, outstanding or authorized stock option, stock appreciation, phantom stock, profit participation
or similar rights or equity-based awards with respect to any Subsidiary of the Company.
(g) Except
(y) as set forth in this Section 3.3 and (z) for Company Options, Company PSUs, Company RSUs and Convertible Notes
outstanding as of the date of this Agreement, there are no: (i) outstanding shares of capital stock of or other securities of any
Acquired Corporation; (ii) outstanding subscriptions, options, calls, warrants or rights (whether or not currently exercisable)
to acquire any shares of the capital stock, restricted stock unit, stock-based performance unit or any other right that is linked to,
or the value of which is in any way based on or derived from the value of any shares of capital stock or other securities of any Acquired
Corporation, in each case other than derivative securities not issued by an Acquired Corporation; (iii) outstanding securities,
instruments, bonds, debentures, notes or obligations that are or may become convertible into or exchangeable for any shares of the capital
stock or other securities of any Acquired Corporation; or (iv) stockholder rights plans (or similar plans commonly referred to as
a “poison pill”) or Contracts under which any Acquired Corporation is or may become obligated to sell or otherwise issue
any shares of its capital stock or any other securities.
(h) Section 3.3(h) of
the Company Disclosure Schedule sets forth a listing of all Persons (either by name or employee identification number) who hold outstanding
Company Equity Awards as of the close of business on September 21, 2023, indicating, with respect to each Company Equity Award,
the number of Shares subject thereto (assuming, in the case of Company PSUs, the target level of performance), the date of grant, the
vesting schedule, the per Share exercise price (if applicable), the expiration date and, with respect to Company incentive stock option
(within the meaning of Section 422 of the Code) (the “Company Equity Awards Schedule”). The Company shall provide
Parent with an updated Company Equity Award Schedule within three (3) business days prior to the anticipated Closing Date to reflect
any changes occurring between the date of this Agreement and the applicable date of delivery. All Company Equity Awards are evidenced
by award agreements in the forms that have been provided to Parent.
(i) All
Convertible Notes were issued pursuant to, and all terms and conditions of the Convertible Notes are evidenced by, the Convertible Notes
Indentures.
3.4 SEC
Filings; Financial Statements.
(a) Since
January 1, 2021 (the “Applicable Date”), the Company has filed or furnished on a timely basis all reports, schedules,
forms, statements and other documents (including exhibits and all other information incorporated therein) required to be filed or furnished
by the Company with the SEC (as supplemented, modified or amended since the time of filing, the “Company SEC Documents”).
As of their respective dates, or, if amended prior to the date of this Agreement, as of the date of (and giving effect to) the last such
amendment (and, in the case of registration statements and proxy statements, on the date of effectiveness and the dates of the relevant
meetings, respectively), the Company SEC Documents complied in all material respects with the requirements of the Securities Act, the
Exchange Act or the Sarbanes-Oxley Act of 2002, as amended (the “Sarbanes-Oxley Act”), as the case may be, and the
rules and regulations of the SEC promulgated thereunder applicable to those Company SEC Documents, and, except to the extent that
information contained in such Company SEC Document has been revised, amended, modified or superseded (prior to the date of this Agreement)
by a later filed Company SEC Document, none of the Company SEC Documents when filed or furnished contained any untrue statement of a
material fact or omitted to state a material fact required to be stated therein or necessary in order to make the statements therein,
in light of the circumstances under which they were made, not misleading.
(b) The
financial statements (including any related notes and schedules) contained or incorporated by reference in the Company SEC Documents:
(i) complied as to form in all material respects with the published rules and regulations of the SEC applicable thereto; (ii) were
prepared in accordance with United States generally accepted accounting principles (“GAAP”) applied on a consistent
basis throughout the periods covered (except as may be indicated in the notes to such financial statements or, in the case of unaudited
interim financial statements, as may be permitted by the SEC on Form 10-Q, Form 8-K or any successor form under the Exchange
Act); and (iii) fairly presented, in all material respects, the financial position of the Company as of the respective dates thereof
and the results of operations and cash flows of the Company for the periods covered thereby (subject, in the case of the interim financial
statements, to the absence of notes and to normal and recurring year-end adjustments that are not individually or in the aggregate material).
(c) The
Company maintains, and at all times since the Applicable Date, has maintained, a system of internal control over financial reporting
(as defined in Rule 13a-15 under the Exchange Act) which is designed to provide reasonable assurance regarding the reliability of
financial reporting and the preparation of financial statements for external purposes in accordance with GAAP, and includes those policies
and procedures that: (i) pertain to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions
and dispositions of the assets of the Company; (ii) provide reasonable assurance that transactions are recorded as necessary to
permit preparation of financial statements in conformity with GAAP and that receipts and expenditures are being made only in accordance
with authorizations of management and the Board of Directors; and (iii) provide reasonable assurance regarding prevention or timely
detection of unauthorized acquisition, use or disposition of the assets of the Company that could have a material effect on the financial
statements. The Company’s management has completed an assessment of the effectiveness of the Company’s system of internal
control over financial reporting in compliance with the requirements of Section 404 of the Sarbanes-Oxley Act for the fiscal year
ended December 31, 2022, and, except as set forth in the Company SEC Documents filed prior to the date of this Agreement, that assessment
concluded that those controls were effective. To the knowledge of the Company, since January 1, 2023, neither the Company nor the
Company’s independent registered accountant has identified or been made aware of: (1) any significant deficiency or material
weakness in the design or operation of the internal control over financial reporting utilized by the Company, which is reasonably likely
to adversely affect the Company’s ability to record, process, summarize and report financial information; or (2) any illegal
act or fraud, whether or not material, that involves the management or other employees of the Company who have a significant role in
the Company’s internal control over financial reporting; or (3) any claim or allegation regarding any of the foregoing.
(d) The
Company maintains, and at all times since the Applicable Date has maintained, disclosure controls and procedures as defined in and required
by Rule 13a-15 or 15d-15 under the Exchange Act that are reasonably designed to ensure that all information required to be disclosed
in the Company’s reports that it files or submits under the Exchange Act is recorded, processed, summarized and reported within
the time periods specified in the rules and forms of the SEC and that all such information is accumulated and communicated to the
Company’s management as appropriate to allow timely decisions regarding required disclosure and to enable the principal executive
officer of the Company and the principal financial officer of the Company to make the certifications required under the Exchange Act
with respect to such reports. The Company is, and at all times since the Applicable Date has been, in compliance in all material respects
with all current listing and corporate governance requirements of Nasdaq. None of the Acquired Corporations has outstanding, or has arranged
any outstanding, “extensions of credit” to directors or executive officers within the meaning of Section 402 of the
Sarbanes-Oxley Act.
(e) None
of the Acquired Corporations is a party to, nor does any Acquired Corporation have any obligation or other commitment to become a party
to, “off-balance sheet arrangements” (as defined in Item 303(a) of Regulation S-K under the Exchange Act) where the
result, purpose or intended effect of such Contract is to avoid disclosure of any material transaction involving, or material liabilities
of, such Acquired Corporation in the Company SEC Documents.
(f) As
of the date of this Agreement, there are no outstanding or unresolved comments in comment letters received from the SEC with respect
to the Company SEC Documents. None of the Company SEC Documents is the subject of ongoing SEC review and there are no inquiries or investigations
by the SEC or any internal investigations pending or threatened, in each case regarding any accounting practices of the Company.
(g) Each
document required to be filed by the Company with the SEC in connection with the Offer, including the Schedule 14D-9 (the “Company
Disclosure Documents”), and any amendments or supplements thereto, when filed, distributed or otherwise disseminated to the
Company’s stockholders, as applicable, will comply as to form in all material respects with the applicable requirements of the
Exchange Act. The Company Disclosure Documents, at the time of the filing of such Company Disclosure Documents or any supplement or amendment
thereto with the SEC and at the time such Company Disclosure Documents or any supplements or amendments thereto are first distributed
or otherwise disseminated to the Company’s stockholders, will not contain any untrue statement of a material fact or omit to state
any material fact required to be stated therein or necessary in order to make the statements made therein, in light of the circumstances
under which they were made, not misleading.
(i) The
information with respect to the Acquired Corporations that the Company furnishes to Parent or Purchaser specifically for use in the Offer
Documents, at the time of the filing of and at the time of any distribution or dissemination of the Offer Documents, will not contain
any untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary in order to make
the statements made therein, in light of the circumstances under which they were made, not misleading.
(ii) Notwithstanding
the foregoing, the Company makes no representation with respect to statements made or incorporated by reference therein based on information
supplied by or on behalf of Parent or Purchaser for inclusion or incorporation by reference in the Company Disclosure Documents.
3.5 Absence
of Changes; No Material Adverse Effect. Except as expressly contemplated by this Agreement,
from January 1, 2023 with respect to clauses (a), (b) and (c) below and from June 30, 2023 with respect to clause
(d) below, in each case, through the date of this Agreement:
(a) except
for discussions, negotiations and activities related to this Agreement or other potential strategic transactions, the Acquired Corporations
have operated in all material respects in the ordinary course of business consistent with past practice;
(b) there
has not occurred any event, occurrence, circumstance, change or effect that, individually or in the aggregate, has had or would reasonably
be expected to have, a Material Adverse Effect; and
(c) there
has not been any action taken by any of the Acquired Corporations that, if taken during the Pre-Closing Period, would constitute a breach
of, or require the consent of Parent under, clauses (i), (ii), (v), (ix), (xii), (xiii), (xiv), (xv), (xvi), (xviii), (xix), (xx), (xxi) and
(xxii) (solely as relates to the foregoing clauses) of Section 5.2(b).
(d) there
has not been any action taken by any of the Acquired Corporations that, if taken during the Pre-Closing Period, would constitute a breach
of, or require the consent of Parent under, clauses (iii), (iv), (vi), (vii), (viii), (x), (xi), (xvii), and (xxii) (solely as relates
to the foregoing clauses) of Section 5.2(b).
3.6 Title
to Assets. Each Acquired Corporation has good and valid title to all assets (excluding Intellectual
Property Rights) owned by it as of the date of this Agreement, and such assets are, in all material respects, sufficient to carry on
the business of each Acquired Corporation as currently conducted and are owned by such Acquired Corporation free and clear of any Encumbrances
(other than Permitted Encumbrances).
3.7 Real
Property.
(a) The
Acquired Corporations do not own any real property.
(b) The
Acquired Corporations hold valid and existing leasehold interests in the real property that is leased or subleased by the Acquired Corporations
from another Person (the “Leased Real Property”), free and clear of all Encumbrances other than Permitted Encumbrances.
Since the Applicable Date, no Acquired Corporation has received any notice regarding (i) any material violation or breach or default
under any lease related to the Leased Real Property that has not since been cured; (ii) pending or threatened condemnation of any
portion of the Leased Real Property; or (iii) building, fire or zoning code violations with respect to the Leased Real Property.
3.8 Intellectual
Property.
(a) Section 3.8(a) of
the Company Disclosure Schedule sets forth a complete and accurate list of all U.S. and foreign: (i) issued Patents and Patent applications;
(ii) Trademark registrations and applications (including Internet domain name registrations); and (iii) Copyright registrations
and applications, in each case of the foregoing clauses (i), (ii) and (iii), that is Company IP as of the date
hereof (the “Registered Company IP”), indicating for each item, as applicable, (1) the jurisdiction of filing
or in the case of domain names, the registrar through such domain name is registered, (2) all registration, issuance, and application
numbers, (3) all filing, registration and issue dates and (4) the legal owner. The Company IP is subsisting and the issued
and granted items included therein are valid and, to the knowledge of the Company, enforceable. All issuance, renewal, maintenance and
other similar payments to the United States Patent and Trademark Office, United States Copyright Office, domain name registrars or foreign
equivalents of the foregoing that have become finally due with respect to any Company IP and, to the knowledge of the Company, with respect
to any other Company IP for which the Company is responsible for prosecution and maintenance, in each case, have been timely paid in
full. Each Acquired Corporation has complied with their duty of candor and disclosure to the United States Patent and Trademark Office
and any relevant foreign office and have not committed any inequitable conduct in their prosecution, maintenance, enforcement and defense
of all Company IP for which it controls such activities. Each of the Patents included in the Company IP properly identifies each inventor
of the claims thereof as determined in accordance with the applicable Legal Requirements of the jurisdiction in which such Patent is
issued or is pending. No Legal Proceeding or other written claim (other than routine examination proceedings with respect to pending
applications) is pending or, to the knowledge of the Company, threatened against any Acquired Corporation, in which the scope, validity,
ownership, inventorship or enforceability, as applicable, of any Company IP is being contested or challenged, including, with respect
to Patents, any inventorship challenges, post-grant review proceedings, inter partes review proceedings, derivation proceedings, interferences,
reexaminations and pre- and post-grant oppositions and invalidity challenges, and, with respect to Trademarks, invalidity, nullity, opposition,
cancellation and expungement or other similar proceedings. The Acquired Corporations have not received written notice from any third
party stating that the Company does not have clear title and/or exclusive rights, as applicable, to the Company IP owned or purported
to be owned by the Company, or that would affect the patentability or enforceability of any Company IP.
(b) The
Acquired Corporations are the sole and exclusive owners of all right, title and interest in and to all Company IP owned or purported
to be owned by the Acquired Corporations, free and clear of all Encumbrances other than Permitted Encumbrances, except as would not reasonably
be expected to be, individually or in the aggregate, material to the businesses of the Acquired Corporations. The Acquired Corporations
own or have sufficient right to use pursuant to written licenses all Intellectual Property Rights material to, and used or held for use,
or otherwise necessary for the operation of the Acquired Corporations’ business; provided, that the foregoing is not a representation
or warranty with respect to infringement, misappropriation or other violation of Intellectual Property Rights.
(c) No
funding, facilities, personnel or other resources of any Governmental Body or any university, college, research institute or other educational
institution is being or has been used to create, in whole or in part, Company IP owned by the Acquired Corporations, or to the knowledge
of the Company, any other Company IP, except for any such funding or use of facilities, personnel or other resources that have not resulted
in such Governmental Body or institution obtaining ownership rights in, or other material rights to use or exploit, such Company IP (excluding,
for the avoidance of doubt, any retained non-exclusive rights by such Governmental Body or institution solely for the purposes of conducting
academic research).
(d) The
Acquired Corporations have taken reasonable steps to maintain the confidentiality of and otherwise protect and enforce their rights in
all Trade Secrets in the possession of or used by an Acquired Corporation and, to the knowledge of the Company, no such Trade Secrets
have been disclosed to or have been discovered by any Person, other than pursuant to a written, valid and enforceable agreement restricting
the disclosure and use thereof, which agreement has not been breached.
(e) Neither
the operation of the Acquired Corporations’ business nor the making, use, import, sale, offer for sale or other disposition of
any product or service of the Acquired Corporations, in each case, as currently conducted, infringes, misappropriates or otherwise violates
or has since the Applicable Date infringed, misappropriated or otherwise violated any Intellectual Property Rights owned by any other
Person, except as would not reasonably be expected to be, individually or in the aggregate, material to the businesses of the Acquired
Corporations. Except as would not reasonably be expected to be, individually or in the aggregate, material to the businesses of the Acquired
Corporations, since the Applicable Date, no Legal Proceeding or other written claim (including any invitation to take a license or cease
and desist letter) is or has been pending (or, to the knowledge of the Company, threatened) against an Acquired Corporation alleging
any such infringement, misappropriation or other violation.
(f) To
the knowledge of the Company, no Person is infringing, misappropriating or otherwise violating, and since the Applicable Date has not
infringed, misappropriated or otherwise violated any Company IP. Since the Applicable Date, no Legal Proceeding or other written claim
(including any invitation to take a license or cease and desist letter) is pending or threatened by an Acquired Corporation alleging
any such infringement, misappropriation or other violation.
(g) Each
Acquired Corporation has complied in all material respects with any and all obligations pursuant to the Patent and Trademark Law Amendments
Act, 35 U.S.C. §200 et seq., the Bayh-Dole Act, or other similar applicable obligations under the Legal Requirements of any applicable
jurisdiction, including with respect to any Patents that are part of the Company IP.
(h) Each
Person who is or was an employee, officer, director, consultant or contractor of an Acquired Corporation and has contributed (or is reasonably
expected to contribute) to the creation or development of any Intellectual Property Rights material to the operation of the Acquired
Corporation’s business has executed and delivered to the Acquired Corporation, a valid and enforceable written Contract containing
an irrevocable present assignment to the Acquired Corporation of all of such Person’s right, title and interest in and to any such
Intellectual Property Rights.
(i) The
Acquired Corporations own or otherwise have sufficient and valid right to use all IT Systems necessary for, or used in and material to,
the operation of the Acquired Corporations’ business. Such IT Systems (i) operate and perform in all material respects in
accordance with their documentation and functional specifications and otherwise as required by the Acquired Corporations in connection
with the operation of the Acquired Corporations’ business (ii) are free from material bugs, material malicious codes or other
material defects and (iii) have not malfunctioned or failed since the Applicable Date in a manner that has had a material impact
on the operation of the Acquired Corporations’ business. Except as would not reasonably be expected to be, individually or in the
aggregate, material to the businesses of the Acquired Corporations, no Person has gained unauthorized access to the IT Systems since
the Applicable Date. The Acquired Corporations have implemented reasonable backup and disaster recovery technology processes consistent
with industry standards.
(j) The
Acquired Corporations are and have been since the Applicable Date in compliance in all material respects with all applicable Privacy
Laws and Company Security Policies, and no circumstance has arisen in which Privacy Laws required the Acquired Corporations to notify
a Governmental Body or any affected individual of any actual or suspected unauthorized Processing of any Personal Information. Since
the Applicable Date, the Acquired Corporations have not received any written notice, order, inquiry, investigation, complaint or other
communication alleging material non-compliance with any Privacy Laws or Company Security Policies.
(k) Since
the Applicable Date, the Acquired Corporations have implemented and maintained a written information security program that complies with
Privacy Laws and Company Security Policies and contains commercially reasonable organizational, physical, administrative and technical
measures, consistent in all material respects with industry standards, and have otherwise taken steps reasonably necessary, to ensure
that Personal Information Processed by or on behalf of the Acquired Corporations is protected against loss, theft, misuse, or unauthorized
Processing. There has been no material loss, theft, misuse, or unauthorized Processing of any Personal Information Processed by or on
behalf of the Acquired Corporations. The Acquired Corporations require, pursuant to valid, written Contracts, any Person who receives
or Processes Personal Information from or on behalf of the Acquired Corporations to comply with all applicable Privacy Laws and Company
Security Policies.
3.9 Contracts.
(a) Section 3.9(a) of
the Company Disclosure Schedule identifies each Contract to which any Acquired Corporation is a party, or by which it is bound, that
constitutes a Material Contract as of the date of this Agreement. For purposes of this Agreement, each of the following (excluding any
Employee Plan) to which any Acquired Corporation is a party or by which it is bound as of the date of this Agreement constitutes a “Material
Contract”:
(i) any
Contract that (A) is a settlement, conciliation or similar agreement between any Acquired Corporation and any Governmental Body
and (x) pursuant to which an Acquired Corporation will be required after the date of this Agreement to pay any monetary obligations
or (y) that contains material obligations or limitations on such Acquired Corporation’s conduct or (B) is with any Governmental
Body under which payments in excess of $500,000 were received by any Acquired Corporation in the most recently completed fiscal year;
(ii) any
Contract between any Acquired Corporation and any third Person (A) materially limiting the freedom or right of any Acquired Corporation
to engage in any line of business or to compete with any other Person in any location or line of business, (B) containing any “most
favored nations” terms and conditions (including with respect to pricing) granted by any Acquired Corporation, or (C) containing
exclusivity obligations or otherwise materially limiting the freedom or right of any Acquired Corporation to sell, distribute or manufacture
any products or services for any other Person;
(iii) any
Contract that requires by its terms or is reasonably expected to require the payment or delivery of cash or other consideration to any
Acquired Corporation in an amount having an expected value in excess of $500,000 in the fiscal year ending December 31, 2023 or
by any Acquired Corporation in an amount having an expected value in excess of $1,000,000 in the fiscal year ending December 31,
2023 and in each case (A) which cannot be cancelled by such Acquired Corporation without penalty or further payment without more
than ninety (90) days’ notice and (B) excluding commercially available off-the-shelf software licenses and Software-as-a-Service
offerings and generally available patent license agreements not included in paragraph (xiii) below (in each case, entered into in
the ordinary course of business);
(iv) any
Contract relating to Indebtedness of any Acquired Corporation in an aggregate principal amount in excess of $500,000 (whether incurred,
assumed, guaranteed or secured by any asset);
(v) any
Contract that relates to the acquisition or disposition of any material business, a material amount of stock or assets of any Person
or any real property (whether by merger, sale of stock, sale of assets, exclusive license or otherwise), in each case that contains a
right of first negotiation, right of first refusal, ongoing royalty or milestone payment obligations, earnout or similar contingent payments,
ongoing indemnification obligations or other similar rights;
(vi) any
Contract to which an Acquired Corporation has obligations to make (A) milestone or similar payments upon the achievement of regulatory
or commercial milestones, or (B) payment of royalties or other amounts calculated based upon any revenues or income of any Acquired
Corporation, in each case that could result in payments in excess of $500,000;
(vii) any
Contract between any Acquired Corporation and any third Person constituting a joint venture, collaboration, partnership or similar revenue
sharing arrangement;
(viii) any
Contract that by its express terms requires an Acquired Corporation, or any successor to, or acquirer of, an Acquired Corporation, to
make any payment to another Person as a result of the execution, delivery and performance of this Agreement (a “Change of Control
Payment”) or gives another Person a right to receive or elect to receive a Change of Control Payment;
(ix) any
Contract creating or governing a lease of any Leased Real Property;
(x) any
management or employment Contract (other than employment offer letters entered into in the ordinary course) between any Acquired Corporation
and any individual service provider of any Acquired Corporation with total annual fees that equals or exceeds $500,000;
(xi) any
severance, retention, transaction bonus, change in control or other similar Contract between any Acquired Corporation and any Company
Associate;
(xii) any
Contract that prohibits the declaration or payment of dividends or distributions in respect of the capital stock of an Acquired Corporation,
the pledging of the capital stock or other equity interests of an Acquired Corporation or the issuance of any guaranty by an Acquired
Corporation;
(xiii) any
Contract pursuant to which (a) an Acquired Corporation is granted a license or other right under any Intellectual Property Right
owned by any third party that is material to the operation of the Acquired Corporations’ business or (b) an Acquired Corporation
grants a third party a license or other right to any Company IP material to the operation of the Acquired Corporations’ business
(including any settlement agreements with respect to Company IP), in each case of the foregoing clauses (a) and (b),
other than nondisclosure agreements, third party service provider agreements, and commercially available, non-exclusive, non-customized,
off-the-shelf software agreements and Contracts in which grants of rights to use Intellectual Property Rights are incidental to and not
material to performance under the Contract, in each case, entered into in the ordinary course of business;
(xiv) any
other Contract that is currently in effect and has been filed (or is required to be filed) by the Company as an exhibit pursuant to Item
601(b)(10) of Regulation S-K under the Securities Act or that would be required to be disclosed under Item 404 of Regulation S-K
under the Securities Act;
(xv) any
Contract with any Affiliate (other than another Acquired Corporation), director, executive officer (as such term is defined in the Exchange
Act), Person holding 5% or more of the Shares, or, to the knowledge of the Company, any Affiliate (other than another Acquired Corporation)
or immediate family member of any of the foregoing;
(xvi) any
Contract relating to the research, development, distribution, marketing, supply, license, collaboration, co-promotion or the manufacturing
of the Product Candidates which, in each case, (A) is reasonably expected to require the payment or delivery of cash or other consideration
by any Acquired Corporation in an amount having an expected value in excess of $1,000,000 in the fiscal year ending December 31,
2023 or (B) if terminated or not renewed, would reasonably be expected to have a material and adverse effect on the Product Candidates;
(xvii) any
indemnification or other similar Contract between an Acquired Corporation and a current or former director or officer of any Acquired
Corporation that provides rights to indemnification, advancement of expenses and exculpation from liabilities for acts or omissions occurring
at or prior to the Effective Time (whether asserted or claimed prior to, at or after the Effective Time) in favor of such current or
former director or officer; and
(xviii) except
for the Contracts referred to in clause (xvii) of this section, any Contract, the primary purpose of which is to provide
for indemnification or guarantee of the obligations of any other Person that would be material to the Company, other than any such Contracts
entered into in the ordinary course of business.
(b) As
of the date of this Agreement, the Company has either delivered or made available to Parent a copy of each Material Contract or has publicly
made available such Material Contract in the Electronic Data Gathering, Analysis and Retrieval (EDGAR) database of the SEC. No Acquired
Corporation nor, to the knowledge of the Company, any other party thereto is in material breach of, or material default under, any Material
Contract and no Acquired Corporation, or, to the knowledge of the Company, any other party to a Material Contract, has taken or failed
to take any action that with or without notice, lapse of time or both would constitute a material breach of or material default under
any Material Contract. Each Acquired Corporation has timely delivered all notices to the relevant recipients pursuant to, and in compliance
with, the settlement agreements with respect to Company IP to which it is a party on the date hereof. Each Material Contract is, with
respect to the Acquired Corporations and, to the knowledge of the Company, each other party thereto, a valid and binding agreement in
full force and effect, enforceable in accordance with its terms, except as such enforcement may be subject to bankruptcy, insolvency,
fraudulent transfer, reorganization, moratorium and other similar laws of general applicability relating to or affecting creditors’
rights, and by general equitable principles. Since the Applicable Date, the Acquired Corporations have not received any written notice
regarding any material violation or breach or default under any Material Contract that has not since been cured. No Acquired Corporation
has waived in writing any material rights under any Material Contract.
3.10 Liabilities.
The Acquired Corporations do not have any liabilities (whether accrued, absolute, contingent or otherwise) which would be required to
be reflected or reserved against on a consolidated balance sheet of the Company prepared in accordance with GAAP or the notes thereto,
except for: (i) liabilities reflected or reserved against in the financial statements or notes thereto included in the Company SEC
Documents filed prior to the date of this Agreement; (ii) liabilities or obligations incurred pursuant to the terms of this Agreement;
(iii) liabilities for performance of obligations under Contracts binding upon the Acquired Corporations (other than resulting from
any breach thereof) either delivered or made available to Parent prior to the date of this Agreement; and (iv) liabilities incurred
in the ordinary course of business consistent with past practice since June 30, 2023. Section 3.10 of the Company Disclosure
Schedule sets forth a true and complete list of all outstanding Indebtedness of the Acquired Corporations as of the date of this Agreement.
3.11 Compliance
with Legal Requirements.
(a) The
Acquired Corporations are, and since the Applicable Date have been, in compliance with all applicable Legal Requirements and, since the
Applicable Date, no Acquired Corporation has been given written notice of, or been charged with, any violation of, any applicable Legal
Requirement, except as would not be, individually or in the aggregate, material to the Acquired Corporations taken as a whole.
(b) For
each of the past five (5) years, each Acquired Corporation has been in compliance with all applicable Global Trade Laws. No Acquired
Corporation nor any Acquired Corporation’s Representative is a Sanctioned Person or is acting on behalf of a Sanctioned Person.
In the past five (5) years, no Acquired Corporation has done business, directly or indirectly, with a Sanctioned Person or any Sanctioned
Territory. Since the Applicable Date, no Acquired Corporation has had any direct or indirect transactions with or engaged in an activity
involving a benefit provided to or received from Russia, Belarus, or the Crimea and the so-called Donetsk People’s Republic and
Luhansk People’s Republic regions of Ukraine, including from (i) the territory of, (ii) Persons located in, or (iii) Persons
incorporated, headquartered, or whose principal place of business is in those jurisdictions or regions. In the past five (5) years,
no Acquired Corporation has, in connection with or relating to the business of any Acquired Corporation, (A) received from any Governmental
Body any notice or (B) been subject to an investigation or made any disclosure to a Governmental Body, in either case concerning
any actual or potential violations concerning applicable Global Trade Laws.
3.12 Regulatory
Matters.
(a) Each
Acquired Corporation is, and since the Applicable Date has been, in compliance in all material respects with all Healthcare Laws, in
each case as applicable to the operation of its business. Since the Applicable Date, no Acquired Corporation, nor, to the knowledge of
the Company, any third parties that have performed or are performing any development, collaboration, manufacturing, testing, quality
control, batch release, distribution or shipment activities on behalf of an Acquired Corporation or with respect to a Product Candidate,
including any contract manufacturing organization, CRO, Clinical Trial site or investigator, while acting in such capacity (each, a “Collaboration
Partner”) has been subject to any enforcement, regulatory or administrative proceedings against such Acquired Corporation alleging
material non-compliance with any Healthcare Laws, and no Acquired Corporation has received any written, oral or other notification or
threat or communication from the FDA or any other Governmental Body performing functions similar to those performed by the FDA (each,
a “Healthcare Regulatory Authority”) alleging any material non-compliance with any Healthcare Law, including any (i) FDA
Form 483 or warning letter or (ii) “Notice of Adverse Findings” from the FDA or similar notice from any other Healthcare
Regulatory Authority.
(b) Since
the Applicable Date, the Acquired Corporations have held all Regulatory Permits required for their business as then conducted, and each
Regulatory Permit as currently conducted, and each such Regulatory Permit is valid and in in full force and effect. The Acquired Corporations
are in compliance in all material respects with the terms and requirements of such Regulatory Permits. Since the Applicable Date, no
deficiencies have been asserted in writing by any Healthcare Regulatory Authority with respect to any Regulatory Permits of the Acquired
Corporations. None of the Acquired Corporations has received written notice that any material Regulatory Permit will not or is likely
not to be issued, renewed or extended.
(c) To
the knowledge of the Company, all preclinical and clinical investigations, studies, or trials (“Clinical Trials”)
sponsored or conducted by or on behalf of the Acquired Corporations have been and are being conducted in material compliance with applicable
Healthcare Laws, including Good Clinical Practices requirements and federal and state laws, rules, regulations and binding guidances
restricting the use and disclosure of individually identifiable health information. No Acquired Corporation, nor, to the knowledge of
the Company, any of their Collaboration Partners or other Persons acting directly on their behalf, has received any written notice or
other correspondence from any Healthcare Regulatory Authority with respect to any ongoing Clinical Trial requiring or recommending a
clinical hold, or the termination, suspension or material modification of such Clinical Trial.
(d) The
Acquired Corporations have filed with the FDA and any other applicable Healthcare Regulatory Authorities all required material filings,
declarations, listings, registrations, reports or submissions, including, but not limited to, adverse event reports, and any required
updates, corrections or modifications to each of the foregoing. All such filings, declarations, listings, registrations, reports or submissions
were in material compliance with applicable Healthcare Laws when filed, and no deficiencies have been asserted in writing by any applicable
Healthcare Regulatory Authority with respect to any such filings, declarations, listings, registrations, reports or submissions.
(e) Since
the Applicable Date, the Acquired Corporations have not (i) voluntarily or involuntarily initiated, conducted or issued, or caused
to be initiated, conducted or issued any recall, field corrective action, market withdrawal or replacement, safety alert, warning, “dear
doctor” letter, investigator notice, or other notice or action to wholesalers, distributors, retailers, healthcare professionals
or patients relating to an alleged lack of safety, efficacy or regulatory compliance of any product (a “Recall”) or
been required to do so, or (ii) received any written notice from a Healthcare Regulatory Authority regarding (A) any Recall
of any product, or (B) a change in the marketing status or classification, or a material change in the labeling, of any product.
(f) To
the knowledge of the Company, no Acquired Corporation, nor any Collaboration Partner or other Person acting directly on its behalf, has
(i) made an untrue statement of a material fact or fraudulent statement to any Healthcare Regulatory Authority, (ii) failed
to disclose a material fact required to be disclosed to any Healthcare Regulatory Authority or (iii) committed any other act, made
any statement or failed to make any statement, that (in any such case) establishes a reasonable basis for the FDA to invoke its Fraud,
Untrue Statements of Material Facts, Bribery, and Illegal Gratuities Final Policy, or for any other Healthcare Regulatory Authority to
invoke any similar policy. No Acquired Corporation is the subject of any pending or, to the knowledge of the Company, threatened investigation
by the FDA pursuant to its Fraud, Untrue Statements of Material Facts, Bribery and Illegal Gratuities Final Policy. No Acquired Corporation
nor, to the knowledge of the Company, any Collaboration Partner, officer, employee, agent or other Person acting on behalf of any Acquired
Corporation has been debarred or disqualified, excluded from participation in federal healthcare programs or convicted of any crime or
engaged in any conduct that would reasonably be expected to result in (A) debarment under 21 U.S.C. § 335a or any similar Legal
Requirement, (B) exclusion under 42 U.S.C. § 1320a-7 or any similar Legal Requirement, or (C) disqualification pursuant
to 21 CFR Part 312.70 or any similar Legal Requirement.
(g) Since
the Applicable Date, the Acquired Corporations have not received any written notice from a Governmental Body that any of their products
are misbranded as defined in 21 U.S.C. § 352 or adulterated as defined in 21 U.S.C. § 351, as amended, and the rules and
regulations promulgated thereunder, or as defined in comparable Legal Requirements in any jurisdiction.
(h) Except
as has not been, and would not reasonably be expected to be, material to the business of the Acquired Corporations, taken as a whole,
the Acquired Corporations and, to the knowledge of the Company, their Collaboration Partners, or other Persons acting directly on their
behalf have prepared, submitted and implemented timely responses and, as applicable, any corrective action plans required to be prepared
and submitted in response to all internal or third-party audits, inspections, investigations or examinations of the Product Candidates
or the Acquired Corporation’s business.
(i) The
statements set forth in Section 3.12(i) of the Company Disclosure Schedule are true, accurate and complete in all material
respects.
3.13 Certain
Business Practices. No Acquired Corporation nor, to the knowledge of the Company, any of its
Representatives (in each case, acting in the capacity of a Representative of such Acquired Corporation) has (i) used any funds (whether
of an Acquired Corporation or otherwise) for unlawful contributions, gifts, entertainment or other unlawful expenses relating to political
activity, (ii) made or offered to make any unlawful payment to foreign or domestic government officials or employees or to foreign
or domestic political parties or campaigns or (iii) accepted any unlawful payments, or (iv) violated any provision of any Anti-Corruption
Laws or any rules or regulations promulgated thereunder, anti-money laundering laws or any rules or regulations promulgated
thereunder or any applicable Legal Requirement of similar effect. Since the Applicable Date, no Acquired Corporation has received any
written communication from a Governmental Body that alleges or gives notice of any of the foregoing. The Acquired Corporations have instituted
and maintain policies and procedures reasonably designed to ensure compliance with Anti-Corruption Laws. As of the date of this Agreement,
no officer, director or employee of any Acquired Corporation is a government official.
3.14 Governmental
Authorizations. The Acquired Corporations hold all material Governmental Authorizations necessary
to enable the Acquired Corporations to conduct their business in the manner in which such business is currently being conducted. The
material Governmental Authorizations held by the Acquired Corporations are valid and in full force and effect. The Acquired Corporations
are in compliance with the terms and requirements of such Governmental Authorizations, except as would not be material to the Acquired
Corporations taken as a whole.
3.15 Tax
Matters
(a) (i) Each
of the material Tax Returns required to be filed by or on behalf of an Acquired Corporation with any Governmental Body (the “Company
Returns”) have been filed on or before the applicable due date (including any extensions of such due date), and have been prepared
in accordance with all applicable Legal Requirements and are accurate and complete, in each case in all material respects, and (ii) all
material Taxes due and payable by an Acquired Corporation (whether or not shown on the Company Returns) have been paid, and all material
Taxes required to be withheld have been withheld and paid, in each case, to the relevant Governmental Body.
(b) (i) There
are no pending examinations or audits of any Company Return in progress involving material Taxes and (ii) no unresolved written
claim has been received by any Acquired Corporation from any Governmental Body in any jurisdiction where each Acquired Corporation, as
applicable, does not file Tax Returns that such Acquired Corporation is or may be subject to Taxes in that jurisdiction. No extension
or waiver of the statute of limitation period applicable to any material Company Returns has been granted and is currently in effect
other than automatic extensions or waivers obtained in the ordinary course of business.
(c) No
Legal Proceeding involving the IRS or any other Governmental Body is pending or has been threatened in writing against or with respect
to any Acquired Corporation in respect of any material Tax, and no deficiency of material Taxes has been asserted in writing as a result
of any audit or examination by any Governmental Body that has not been paid, accrued for or been contested in good faith and in accordance
with applicable Legal Requirements.
(d) For
taxable years for which the applicable statute of limitations for an assessment of Taxes has not expired, no Acquired Corporation (i) has
been a member of an affiliated group (within the meaning of Section 1504(a) of the Code) filing a consolidated federal income
Tax Return (other than a group the common parent of which is or was the Company), or (ii) has any material liability for the Taxes
of any other Person (other than another Acquired Corporation) under Section 1.1502-6 of the Treasury Regulations (or any similar
provision of state, local or non-U.S. law), or as a transferee or successor or otherwise (other than pursuant to agreements not primarily
related to Taxes and entered into in the ordinary course of business).
(e) During
the two (2)-year period ending on the date hereof, none of the Acquired Corporations has been either a “distributing corporation”
or a “controlled corporation” in a distribution of stock intended to qualify for tax-free treatment under Section 355
of the Code.
(f) No
Acquired Corporation has participated in any “listed transaction” within the meaning of Treasury Regulations Section 1.6011-4(b)(2) or
any comparable provision of state, local or non-U.S. law (and has not been required to make a report under European Council Directive
2011/16 (“DAC 6”) or any domestic implementation of DAC 6). Each Acquired Corporation has disclosed on its U.S. federal
income Tax Returns all positions taken therein that could give rise to a substantial understatement of U.S. federal income Tax within
the meaning of Section 6662 of the Code (or any corresponding or similar provision of state, local or non-U.S. law).
(g) No
Acquired Corporation will be required to include any material item of income in, or exclude any material item of deduction from, the
computation of taxable income for any taxable period (or portion thereof) ending after the Closing Date as a result of any (i) change
in method of accounting for a taxable period ending on or prior to the Closing Date as a result of transactions or events occurring,
or accounting methods employed, prior to the Closing, (ii) “closing agreement” as described in Section 7121 of
the Code (or any corresponding or similar provision of state, local or non-U.S. income Tax law) executed prior to the Closing, (iii) installment
sale, intercompany transaction or open transaction disposition made prior to the Closing, (iv) prepaid amount received or deferred
revenue accrued prior to the Closing or (v) deferred intercompany transaction or excess loss account. None of the Acquired Corporations
has made an election under Section 965(h) of the Code or otherwise has any liability for Taxes pursuant to Section 965
of the Code that has not been fully paid to the appropriate Governmental Body.
(h) No
Acquired Corporation is party to or bound by any Tax allocation or Tax sharing , or similar agreement with any Person, other than any
commercial, employment, leasing or financing agreement not primarily related to Taxes and entered into in the ordinary course of business.
(i) There
are no material Encumbrances with respect to Taxes upon any of the assets or properties of any Acquired Corporation, other than Permitted
Encumbrances.
(j) The
jurisdiction of organization, tax residence, and classification for U.S. federal income tax purposes with respect to each Acquired Corporation
is as provided on Section 3.15(j) of the Company Disclosure Schedule. Unless otherwise noted on Section 3.15(j) of
the Company Disclosure Schedule, such classification for U.S. federal income tax purposes has been in effect at all times since formation
of the relevant entity. No Acquired Corporation is an “expatriated entity” or “surrogate foreign corporation”
under Section 7874 of the Code, nor treated as a domestic corporation by reason of Section 7874(b) of the Code. No Acquired
Corporation has any interest in any entity or arrangement treated as a partnership for U.S. federal income tax purposes, other than an
interest in another Acquired Corporation. No Acquired Corporation is, or has been, subject to Tax in any jurisdiction other than its
place of organization by virtue of having a permanent establishment (as defined by applicable Tax treaty) or other place of business
or taxable presence in that jurisdiction.
(k) Adequate
reserves have been established on the Company’s consolidated financial statements to provide for the payment of any Taxes which
are not yet due and payable with respect to any Acquired Corporation for taxable periods or portions thereof ending on or before the
date of the most recent financial statements of the Company. All Taxes of the Acquired Corporations incurred through the date of the
most recent financial statements of the Company do not exceed Taxes accrued on such financial statements, and all Taxes of the Acquired
Corporations accrued following the end of the most recent period covered by the most recent financial statements of the Company have
been accrued in the ordinary course of business.
(l) None
of the Acquired Corporations (i) applied for or received loans or payments under the CARES Act and COVID Relief Programs, (ii) applied
for or received Tax credits under the CARES Act and COVID Relief Programs and (iii) applied for or received any other credits, deferrals
(including any deferral of the employer portion of any payroll Taxes) or benefits under the CARES Act and COVID Relief Programs. None
of the Acquired Corporations has been or is currently the beneficiary of a Tax holiday (or similar benefit) from any Governmental Body.
(m) All
transactions entered into or conducted by any Acquired Corporation with any Person who is a member of the “controlled group”
(within the meaning of Treasury Regulations Section 1.482-1 or any similar or comparable provision of any state, local or non-U.S.
law) of which such Acquired Corporation is a member have been made in material compliance with Section 482 of the Code and the Treasury
Regulations thereunder. None of the Acquired Corporations is a party to any advance pricing agreement, or any similar contract or agreement,
with any Governmental Body. The Acquired Corporations have complied in all material respects with applicable transfer pricing documentation
requirements.
(n) None
of the Acquired Corporations has filed (or has had filed on its behalf) a request for (i) private letter ruling, (ii) technical
advice, or (iii) change of any method of accounting, in each case, with any Governmental Body that relates to Taxes or Tax Returns
of any of the Acquired Corporations.
(o) None
of the Acquired Corporations has (i) entered into any settlement or arrangement (including a closing agreement within the meaning
of Section 7121 of the Code or a comparable or similar provision of state, local or non-U.S. law) with any Governmental Body that
would be binding and result in a material Tax liability for any Tax period (or portion thereof) ending after the Closing Date or (ii) granted
any power of attorney with respect to any Taxes that will be in force following the Closing.
3.16 Employee
Matters
(a) Section 3.16(a) of
the Company Disclosure Schedule sets forth, as of the date of this Agreement, a true, correct and complete list of all employees of the
Company and its Subsidiaries, including for each such employee, to the extent applicable: (i) employee identification number, (ii) original
date of hire by the Company or any of its Subsidiaries, (iii) active or leave status (and, if on leave, the anticipated date of
return), (iv) title, (v) annual base salary or wage rate, (vi) target short-term bonus opportunity, (vii) target
long-term bonus opportunity and (viii) work location.
(b) Neither
the Company nor any of its Subsidiaries is a party to any collective bargaining agreement or other Contract with a labor organization
representing any of its employees, and no employees of the Company or its Subsidiaries are represented by any labor union or other labor
organization with respect to their employment with the Company or its Subsidiaries. To the knowledge of the Company, there are no activities
or proceedings by any individual or group of individuals, including representatives of any labor organizations or labor unions, to organize
any employees of the Company or any of its Subsidiaries.
(c) Since
the Applicable Date, there has not been any strike, slowdown, work stoppage, lockout or other labor dispute, or any threat thereof, by
any current or former employees of the Company or its Subsidiaries with respect to their employment with the Company or its Subsidiaries.
As of the date of this Agreement, (i) there is no slowdown, work stoppage, lockout or other labor dispute pending or, to the knowledge
of the Company, threatened, (ii) unfair labor practice charges pending before the National Labor Relations Board or any other Governmental
Body or, to the knowledge of the Company, threatened and (iii) there is no pending or, to the Company’s knowledge, threatened
arbitration or grievance, charge, complaint, audit or investigation by or before any Governmental Body with respect to any current or
former employees of the Company or any of its Subsidiaries.
(d) Neither
the Company nor any of its Subsidiaries has incurred any liability or obligation under the Worker Adjustment and Retraining Notification
Act and the regulations promulgated thereunder or any similar state or local Legal Requirement that remains unsatisfied.
(e) Since
the Applicable Date, (A) no allegations of sexual harassment, discrimination or sexual misconduct have been made to the Company
against any member of the Board of Directors or any employee of the Company or any of its Subsidiaries at the level of Vice President
or above, (B) none of the Company or any of its Subsidiaries has entered into any settlement agreement related to allegations of
sexual harassment, discrimination or sexual misconduct by any member of the Board of Directors or any employee of the Company or any
of its Subsidiaries at the level of Vice President or above and (C) there have been no, and there are no, proceedings currently
pending or, to the knowledge of the Company, threatened, related to any allegations of sexual harassment, discrimination or sexual misconduct
by any member of the Board of Directors or any employee of the Company or any of its Subsidiaries at the level of Vice President or above.
3.17 Benefit
Plans
(a) Section 3.17(a) of
the Company Disclosure Schedule sets forth a list of the material Employee Plans. To the extent applicable, the Company has either delivered
or made available to Parent prior to the execution of this Agreement with respect to each material Employee Plan copies of: (i) all
plan documents and all amendments thereto, and all related trust or other funding documents, and in the case of unwritten material Employee
Plans, written descriptions thereof, (ii) all determination letters, rulings, opinion letters, information letters or advisory opinions
issued by the IRS or the United States Department of Labor, (iii) the most recent summary plan descriptions and any material modifications
thereto and (iv) any non-routine correspondence to or from any Governmental Body received in the past three years with respect to
such material Employee Plan.
(b) Neither
the Company nor any other Person that would be or, at any relevant time, would have been considered a single employer with the Company
under the Code or ERISA has within the preceding six (6) years maintained, contributed to, or been required to contribute to a plan
subject to Section 302 or Title IV of ERISA or Code Section 412, including any “single employer” defined benefit
plan or any “multiemployer plan,” each as defined in Section 4001 of ERISA. No Employee Plan is a “multiple employer
welfare arrangement” (as defined in Section 3(40) of ERISA).
(c) Each
of the Employee Plans that is intended to be qualified under Section 401(a) of the Code has obtained a favorable determination
letter (or opinion letter, if applicable) as to its qualified status under the Code, and, to the knowledge of the Company, there are
no existing circumstances or any events that have occurred that would reasonably be expected to adversely affect the qualified status
of any such Employee Plan. Each of the Employee Plans is now and has been operated in compliance in all material respects with its terms
and all applicable Legal Requirements, including but not limited to ERISA and the Code.
(d) Except
to the extent required under Section 601 et seq. of ERISA or 4980B of the Code (or any other similar state or local Legal Requirement),
neither the Company nor any Employee Plan has any present or future obligation to provide post-employment welfare benefits to or make
any payment to, or with respect to, any present or former employee, officer or director of the Company or its Subsidiaries pursuant to
any retiree medical benefit plan or other retiree welfare plan.
(e) Except
as provided in Section 2.8, neither execution and delivery of this Agreement, shareholder or other approval of this Agreement
nor the consummation of the Transactions (including in combination with other events or circumstances) could (i) entitle any current
or former employee, director, officer, independent contractor or other service provider of the Company or its Subsidiaries to severance
pay, unemployment compensation or any other payment, (ii) accelerate the time of payment or vesting of, or materially increase the
amount of, compensation or benefits due to any such employee, director, officer, or independent contractor, (iii) directly or indirectly
cause the Company to transfer or set aside any material assets to fund any benefits under any Employee Plan, (iv) otherwise give
rise to any material liability under any Employee Plan or (v) limit or restrict the right to merge, materially amend or terminate
any Employee Plan on or following the Effective Time.
(f) Neither
the execution and delivery of this Agreement, shareholder or other approval of this Agreement nor the consummation of the Transactions
(including in combination with other events or circumstances) could result in the payment of any amount that could, individually or in
combination with any other such payment, constitute an “excess parachute payment” as defined in Section 280G(b)(1) of
the Code.
(g) Neither
the Company nor any Subsidiary has any obligation to provide, and no Employee Plan or other agreement provides any individual with the
right to, a gross up, indemnification, reimbursement or other payment for any excise or additional taxes, interest or penalties incurred
pursuant to Section 409A or Section 4999 of the Code or due to the failure of any payment to be deductible under of Section 280G
of the Code.
3.18 Environmental
Matters.
(a) The
Acquired Corporations are and since the Applicable Date have been in material compliance with all applicable Environmental Laws, which
compliance includes obtaining, maintaining or complying with all Governmental Authorizations required under Environmental Laws for the
operation of their business.
(b) As
of the date of this Agreement, there is no Legal Proceeding relating to or arising under or relating to any Environmental Law that is
pending or, to the knowledge of the Company, threatened against any Acquired Corporation or in respect of any Leased Real Property.
(c) Since
the Applicable Date through the date of this Agreement, except as would not be material to the Acquired Corporations taken as a whole,
no Acquired Corporation has received any written notice, report or other information of or entered into any legally binding agreement,
order, settlement, judgment, injunction or decree involving uncompleted, outstanding or unresolved material violations, liabilities or
requirements on the part of any Acquired Corporation relating to or arising under Environmental Laws.
(d) There
are and since the Applicable Date have been no Hazardous Materials present or Releases on, at, under or from any property or facility,
including the Leased Real Property, in a manner and concentration that would reasonably be expected to result in any material claim against
or material liability of an Acquired Corporation under any Environmental Law.
(e) No
Acquired Corporation has assumed, undertaken, or otherwise become subject to any material liability of another Person relating to Environmental
Laws.
3.19 Insurance.
The Company has delivered or made available to Parent an accurate and complete copy of all material insurance policies relating to the
business, assets and operations of the Acquired Corporations. The Acquired Corporations maintain insurance coverage in such amounts and
covering such risks as are in accordance in all material respects with normal industry practice for companies of similar size and stage
of development. To the knowledge of the Company, all such insurance policies are in full force and effect, no notice of termination,
cancellation, non-renewal, material modification or material default has been received (other than a notice in connection with ordinary
renewals) and there is no existing material default or event which, with the giving of notice or lapse of time or both, would constitute
a material default by any insured thereunder. As of the date of this Agreement, there is no claim pending under any of the Company’s
insurance policies as to which coverage has been questioned, denied or disputed by the underwriters of such policies, except as would
not be, or would not reasonably be expected to be, material to the Acquired Corporations taken as a whole.
3.20 Legal
Proceedings; Orders.
(a) There
are no Legal Proceedings pending and served (or, to the knowledge of the Company, pending or threatened and not served) against any Acquired
Corporation or, to the knowledge of the Company, against any present or former officer, director or employee of an Acquired Corporation
in such individual’s capacity as such, except as, individually or taken together (i) would not be, or would not reasonably
be expected to be, material to the Acquired Corporations taken as a whole and (ii) would not reasonably be expected to prevent,
delay, affect or prohibit the consummation of the Transactions.
(b) There
is no material order, writ, injunction or judgment to which an Acquired Corporation is subject.
(c) To
the knowledge of the Company, no material investigation or review by any Governmental Body with respect to an Acquired Corporation is
pending or is being threatened.
3.21 Authority;
Binding Nature of Agreement. The Company has the corporate power and authority to execute and
deliver and to perform its obligations under this Agreement and to consummate the Transactions. Prior to the date of this Agreement,
at a meeting duly called, convened and held in accordance with the certificate of incorporation and bylaws of the Company, the members
of the Board of Directors participating in the decision unanimously (a) determined that this Agreement and the Transactions, including
the Offer and the Merger, are fair to, and in the best interest of, the Company and its stockholders, (b) declared it advisable
to enter into this Agreement, (c) approved the execution, delivery and performance by the Company of this Agreement and the consummation
of the Transactions, including the Offer and the Merger, (d) resolved that the Merger shall be effected pursuant to Section 251(h) of
the DGCL and (e) resolved to recommend that the stockholders of the Company tender their Shares to Parent or Purchaser, as applicable,
pursuant to the Offer, in each case on the terms and subject to the conditions of this Agreement. The resolutions in the foregoing sentence,
subject to Section 6.1, have not been rescinded, withdrawn, or modified. This Agreement has been duly executed and delivered
by the Company, and assuming due authorization, execution and delivery by Parent and Purchaser, this Agreement constitutes the legal,
valid and binding obligation of the Company and is enforceable against the Company in accordance with its terms, except as such enforcement
may be subject to bankruptcy, insolvency, fraudulent transfer, reorganization, moratorium and other similar laws of general applicability
relating to or affecting creditors’ rights, and by general equitable principles. No vote of the Company’s stockholders or
any holder of Shares is necessary to authorize or adopt this Agreement or to consummate the Transactions, and, except for the filing
of the certificate of merger with the Secretary of State of the State of Delaware, no other corporate actions or proceedings on the part
of the Company are necessary to authorize the execution and delivery of and performance under this Agreement and the consummation of
the Transactions.
3.22 Takeover
Laws and CFIUS Matters.
(a) Assuming
the accuracy of the representation and warranty of Parent and Purchaser set forth in the second sentence of Section 4.8,
the Board of Directors has taken and will take all actions so that the restrictions applicable to business combinations contained in
any applicable Takeover Law (including Section 203 of the DGCL) are, and will be, to the extent such restrictions can be rendered
inapplicable by action of the Board of Directors under Legal Requirements, inapplicable to the execution, delivery and performance of
this Agreement and to the consummation of the Offer, the Merger and the other Transactions.
(b) None
of the Acquired Corporations (i) produces, designs, tests, manufactures, fabricates or develops one or more critical technologies,
as defined at 31 C.F.R. § 800.215, (ii) performs any of the functions set forth in column 2 of Appendix A to 31 C.F.R. part
800 with respect to covered investment critical infrastructure, as defined at 31 C.F.R. § 800.212, or (iii) maintains or collects,
directly or indirectly, sensitive personal data, as defined at 31 C.F.R. § 800.241, of U.S. citizens.
3.23 Non-Contravention;
Consents.
(a) Assuming
compliance with the applicable provisions of the DGCL, the HSR Act, any applicable filing, notification or approval in any foreign jurisdiction
required by Antitrust Laws (if any), and the rules and regulations of the SEC and Nasdaq, the execution and delivery of this Agreement
by the Company and the consummation of the Transactions will not: (i) cause a violation of any of the provisions of the certificate
of incorporation or bylaws (or other organizational documents) of any Acquired Corporation; (ii) cause a violation by any Acquired
Corporation of any Legal Requirement (including any applicable Privacy Laws), Company Security Policies, or order applicable to an Acquired
Corporation, or to which an Acquired Corporation is subject; (iii) require any consent or notice under, conflict with, result in
breach of, or constitute a default under (or an event that with notice or lapse of time or both would become a default), or give rise
to any right of purchase, termination, amendment, cancellation, acceleration or other adverse change of any right or obligation or the
loss of any benefit to which an Acquired Corporation is entitled under any provision of any Contract, except the rights and obligations
under the Convertible Notes Indentures as set forth in Section 3.23(a)(iii) of the Company Disclosure Schedule; or (iv) result
in an Encumbrance (other than a Permitted Encumbrance) on any of the property or assets of any Acquired Corporation or with respect to
Intellectual Property Rights, alter or impair or will alter or impair, any of the Acquired Corporations’ rights in, to and under
any Company IP or other Intellectual Property Rights material to the operation of the business of the Acquired Corporations, or the validity,
enforceability, use, right to use, ownership, registration, right to register, priority, duration, scope, or effectiveness of any such
Intellectual Property Rights or will trigger any additional royalties, license fees, honoraria or other payment obligations of the Acquired
Corporations; except, in the case of clauses (ii), (iii) and (iv), as would not reasonably be expected to have,
individually or in the aggregate, a Material Adverse Effect.
(b) Except
for the filing of the certificate of merger with the Secretary of State of the State of Delaware or as may be required by the Exchange
Act (including the filing with the SEC of the Schedule 14D-9 and such reports under the Exchange Act as may be required in connection
with this Agreement and the Transactions), the HSR Act and any applicable filing, notification or approval in any foreign jurisdiction
required by Antitrust Laws (if any) and the applicable rules and regulations of Nasdaq, the Acquired Corporations are not required
to give notice to, make any filing with, or obtain any Consent from any Governmental Body at any time prior to the Closing in connection
with the execution and delivery of this Agreement by the Company, or the consummation by the Company of the Merger or the other Transactions.
3.24 Opinion
of Financial Advisors. The Board of Directors has received (a) the oral opinion of Barclays
Capital Inc., to be subsequently confirmed by delivery of a written opinion, that, as of the date of the opinion, and based upon and
subject to the various qualifications, limitations and other matters set forth therein, the Offer Price to be offered to the holders
of Shares in the Offer is fair to such holders from a financial point of view (the “Barclays Fairness Opinion”) and
(b) the oral opinion of Centerview Partners LLC, to be subsequently confirmed by delivery of a written opinion, that, as of the
date of the opinion, and based upon and subject to the various assumptions made, procedures followed, matters considered, and qualifications
and limitations set forth therein, the Offer Price or Merger Consideration to be paid to the holders of Shares (other than Excluded Shares,
Dissenting Shares or any Shares held by any Affiliate of the Company or Parent) pursuant to this Agreement is fair, from a financial
point of view, to such holders (the “Centerview Fairness Opinion”). As of the date of this Agreement, neither the
Barclays Fairness Opinion nor the Centerview Fairness Opinion has been withdrawn, revoked, rescinded or modified in any way.
3.25 Brokers
and Other Advisors. Except for Barclays Capital Inc. and Centerview Partners LLC, no broker,
finder, investment banker, financial advisor or other Person is entitled to any brokerage, finder’s, financial advisor’s
or other similar fee or commission, or the reimbursement of expenses in connection therewith, in connection with the Transactions based
upon arrangements made by or on behalf of the Company. The Company has made available to Parent accurate copies of all agreements under
which any such fee, commission or expense reimbursement is payable and all indemnification and other agreements related to the engagement
of the persons to whom any such fee is payable.
3.26 Transactions
with Related Persons. Except (a) as set forth on Section 3.26 of the Company Disclosure
Schedule, (b) payment of compensation (whether written or unwritten) for employment to employees, (c) any Employee Plan and
(d) indemnification rights relating to an individual’s service as a director, manager or officer, no Company Related Person
is a party to any Contract or other commitment to which any Acquired Corporation is a party or by which any of its respective businesses,
assets or properties is bound.
3.27 Standstill.
None of the Acquired Corporations has taken any action prohibited by Section 5.3(b)(ii)(C) during the ninety (90) days
prior to the date of this Agreement.
Section 4
REPRESENTATIONS
AND WARRANTIES OF PARENT AND PURCHASER
Parent and Purchaser represent and warrant to
the Company as of the date hereof and as of the Closing Date as follows:
4.1 Due
Organization. Each of Parent and Purchaser is a corporation or other Entity duly organized,
validly existing and in good standing under the laws of its jurisdiction of organization and has all necessary power and authority: (a) to
conduct its business in the manner in which its business is currently being conducted; and (b) to own and use its assets in the
manner in which its assets are currently owned and used, except as would not reasonably be expected to have, individually or in the aggregate,
a Parent Material Adverse Effect.
4.2 Purchaser.
Purchaser was formed solely for the purpose of engaging in the Transactions and activities incidental thereto and has not engaged, and
prior to the Effective Time will not engage, in any business activities or conducted any operations other than as contemplated by this
Agreement in connection with the Transactions and those incident to Purchaser’s formation. Either Parent or a wholly owned Subsidiary
of Parent owns beneficially and of record all of the outstanding capital stock of Purchaser, free and clear of all Encumbrances and transfer
restrictions, except for Encumbrances or transfer restrictions of general applicability as may be provided under the Securities Act or
applicable securities laws.
4.3 Authority;
Binding Nature of Agreement. Parent and Purchaser each have the corporate power and authority
to execute and deliver and perform their respective obligations under this Agreement and to consummate the Transactions. The board of
directors of each of Parent and Purchaser have approved the execution, delivery and performance by Parent and Purchaser of this Agreement
and the consummation of the Transactions, including the Offer and the Merger. This Agreement has been duly executed and delivered by
Parent and Purchaser, and assuming due authorization, execution and delivery by the Company, this Agreement constitutes the legal, valid
and binding obligation of Parent and Purchaser and is enforceable against Parent and Purchaser in accordance with its terms, except as
such enforcement may be subject to bankruptcy, insolvency, fraudulent transfer, reorganization, moratorium and other similar laws of
general applicability relating to or affecting creditors’ rights, and by general equitable principles.
4.4 Non-Contravention;
Consents.
(a) Assuming
compliance with the applicable provisions of the DGCL, the HSR Act and any applicable filing, notification or approval in any foreign
jurisdiction required by Antitrust Laws (if any), and the rules and regulations of the SEC and Nasdaq, the execution and delivery
of this Agreement by Parent and Purchaser, and the consummation of the Transactions, will not: (i) cause a violation of any of the
provisions of the certificate of incorporation or bylaws (or other organizational documents) of Parent or Purchaser; (ii) cause
a violation by Parent or Purchaser of any Legal Requirement or order applicable to Parent or Purchaser, or to which Parent or Purchaser
are subject; or (iii) require any consent or notice under, conflict with, result in breach of, or constitute a default under (or
an event that with notice or lapse of time or both would become a default), or give rise to any right of purchase, termination, amendment,
cancellation, acceleration or other adverse change of any right or obligation or the loss of any benefit to which Parent or Purchaser
is entitled under any provision of any Contract, except in the case of clauses (ii) and (iii), as would not reasonably
be expected to have, individually or in the aggregate, a Parent Material Adverse Effect.
(b) Except
for the filing of the certificate of merger with the Secretary of State of the State of Delaware or as may be required by the Exchange
Act (including the filing with the SEC of the Offer Documents), Takeover Laws, the DGCL, the HSR Act and any applicable filing, notification
or approval in any foreign jurisdiction required by Antitrust Laws (if any) and the applicable rules and regulations of the SEC
and any national securities exchange, neither Parent nor Purchaser, nor any of Parent’s other Affiliates, is required to give notice
to, make any filing with or obtain any Consent from any Governmental Body at any time prior to the Closing in connection with the execution
and delivery of this Agreement by Parent or Purchaser, or the consummation by Parent or Purchaser of the Offer, the Merger or the other
Transactions, except those that the failure to make or obtain would not, individually or in the aggregate, reasonably be expected to
have a Parent Material Adverse Effect. No vote of Parent’s or Purchaser’s stockholders is necessary to approve this Agreement
or any of the Transactions (except in the case of Purchaser as has previously been obtained).
4.5 Disclosure.
None of the Offer Documents will contain any untrue statement of a material fact or omit to state any material fact required to be stated
therein or necessary in order to make the statements therein, in light of the circumstances under which they were made, not misleading.
None of the information with respect to Parent or Purchaser supplied or to be supplied by or on behalf of Parent or Purchaser or any
of their Subsidiaries, specifically for inclusion or incorporation by reference in the Schedule 14D-9 will, (a) at the time such
document is filed with the SEC, (b) at any time such document is amended or supplemented or (c) at the time such document is
first published, sent or given to the Company’s stockholders, contain any untrue statement of a material fact or omit to state
any material fact required to be stated therein or necessary in order to make the statements therein, in light of the circumstances under
which they were made, not misleading. For clarity, the representations and warranties in this Section 4.5 will not apply
to statements or omissions included or incorporated by reference in the Offer Documents or the Schedule 14D-9 based upon information
supplied to Parent by the Company or any of its Representatives on behalf of the Company specifically for inclusion therein.
4.6 Absence
of Litigation. As of the date of this Agreement, there is no Legal Proceeding pending and served
or, to the knowledge of Parent, pending and not served, against Parent or Purchaser, except as would not, and would not reasonably be
expected to, individually or in the aggregate, have a Parent Material Adverse Effect. To the knowledge of Parent or Purchaser, as of
the date of this Agreement, neither Parent nor Purchaser is subject to any continuing order of, consent decree, settlement agreement
or similar written agreement with, or continuing investigation by, any Governmental Body, or any order, writ, judgment, injunction, decree,
determination or award of any Governmental Body, except as would not, and would not reasonably be expected to, individually or in the
aggregate, have a Parent Material Adverse Effect.
4.7 Funds.
As of the Effective Time, Parent will have (and will make available to Purchaser in a timely manner) immediately available funds in cash
in an amount sufficient to carry out all of Parent and Purchaser’s obligations under this Agreement and to consummate the Transactions
by payment in cash of the aggregate Offer Price payable following the Offer Acceptance Time, the aggregate Merger Consideration payable
following the Effective Time and the aggregate amounts payable to holders of Company Options, Company PSUs and Company RSUs following
the Effective Time pursuant to Section 2.8.
4.8 Ownership
of Shares. Except as contemplated by this Agreement, neither Parent nor any of Parent’s
Affiliates directly or indirectly owns, and at all times for the past three (3) years, neither Parent nor any of Parent’s
controlled Affiliates has owned, beneficially or otherwise, any Shares or any securities, contracts or obligations convertible into or
exercisable or exchangeable for Shares. Neither Parent nor Purchaser is, nor for the past three (3) years has been, an “interested
stockholder” of the Company under Section 203(c) of the DGCL.
4.9 Acknowledgement
by Parent and Purchaser. Neither Parent nor Purchaser is relying and neither Parent nor Purchaser
has relied on any representations or warranties whatsoever regarding the Transactions or the subject matter of this Agreement, express
or implied, except for the representations and warranties in Section 3, as qualified by the Company Disclosure Schedule.
Such representations and warranties by the Acquired Corporations constitute the sole and exclusive representations and warranties of
the Acquired Corporations in connection with the Transactions and each of Parent and Purchaser understands, acknowledges and agrees that
all other representations and warranties of any kind or nature whether express, implied or statutory are specifically disclaimed by the
Acquired Corporations.
4.10 Brokers
and Other Advisors. Except for Persons, if any, whose fees and expenses shall be paid by Parent or
Purchaser, no broker, finder, investment banker, financial advisor or other Person is entitled to any brokerage, finder’s, financial
advisor’s or other similar fee or commission, or the reimbursement of expenses in connection therewith, in connection with the
Transactions based upon arrangements made by or on behalf of Parent, Purchaser, or any of their respective Subsidiaries.
Section 5
CERTAIN
COVENANTS OF THE COMPANY
5.1 Access
and Investigation. During the period from the execution and delivery of this Agreement until
the earlier of the Effective Time and the valid termination of this Agreement pursuant to Section 8 (the “Pre-Closing
Period”), upon reasonable advance notice to the Company, the Acquired Corporations shall, and shall cause the respective Representatives
of the Acquired Corporations to provide Parent and Parent’s Representatives with reasonable access during normal business hours
of the Company to the Company’s designated Representatives and assets and properties, and to all existing books, records, Tax Returns,
work papers, documents and information relating to the Acquired Corporations, and promptly provide Parent and Parent’s Representatives
with all reasonably requested information regarding the business of the Acquired Corporations and such additional financial, operating,
Tax and other data and information regarding the Acquired Corporations as Parent may reasonably request; provided, however, that
any such access shall be conducted at Parent’s expense, at a reasonable time, under the supervision of appropriate personnel of
the Acquired Corporations and in such a manner as not to unreasonably interfere with the normal operation of the business of the Acquired
Corporations. Nothing herein shall require any of the Acquired Corporations to disclose any information to Parent if such disclosure
would (i) jeopardize any attorney-client or other legal privilege (so long as the Acquired Corporations have reasonably cooperated
with Parent to permit such inspection of or to disclose such information on a basis that does not waive such privilege with respect thereto),
(ii) contravene any applicable Legal Requirement (so long as the Acquired Corporations have reasonably cooperated with Parent to
permit disclosure to the extent permitted by Legal Requirements) or (iii) contravene any Contract to which an Acquired Corporation
is a party or by which an Acquired Corporation is bound (so long as the Acquired Corporations have reasonably cooperated with Parent
to permit disclosure to the extent permitted by such Contract). Notwithstanding the foregoing, nothing in this Section 5.1
shall require an Acquired Corporation to disclose to Parent or Parent’s Representatives the minutes of the applicable portions
of the meetings of the Board of Directors or any committee thereof (including any portions of presentations or other materials prepared
by or for the Board of Directors or such committee thereof directly and solely related thereto) where the Board of Directors or such
committee thereof discussed (x) the Transactions, or the evaluation of strategic alternatives leading up to the Transactions, (y) any
Acquisition Proposal received after the date of this Agreement or (z) a Company Adverse Recommendation Change; provided that
in the case of any of clauses (x), (y) and (z), this Section 5.1 shall not apply to any information
required to be disclosed or furnished to Parent and Purchaser pursuant to Section 5.3 or 6.1, and the disclosure of
such information shall be governed by Section 5.3 or 6.1 respectively. With respect to the information disclosed pursuant
to this Section 5.1, Parent shall comply with, and shall instruct Parent’s Representatives to comply with, all applicable
Legal Requirements and all of its obligations under the Confidentiality Agreement dated July 10, 2023, between the Company and Parent
(the “Confidentiality Agreement”).
5.2 Operation
of the Acquired Corporations’ Business. During the Pre-Closing Period, except (x) as
required or otherwise contemplated under this Agreement or as required by applicable Legal Requirements, (y) with the written consent
of Parent (which consent shall not be unreasonably withheld, conditioned or delayed), or (z) as set forth in Section 5.2 of
the Company Disclosure Schedule:
(a) the
Company shall, and shall cause each Acquired Corporation to, (i) conduct its business in the ordinary course consistent with past
practice in all material respects and (ii) use their respective reasonable best efforts to (A) preserve intact, in all material
respects, the material components of the Acquired Corporations’ current business organization, taken as a whole, including keeping
available the services of current executive officers and key employees; (B) maintain satisfactory business relationships with licensors,
licensees, suppliers, lessors, Governmental Bodies and others having material business dealings with the Acquired Corporations and (C) preserve
intact its and their material assets (including technology), properties, Contracts and licenses; and
(b) the
Acquired Corporations shall not:
(i) (1) establish
a record date for, declare, set aside or pay any dividend or make any other distribution in respect of any shares of its capital stock
(including the Shares), or (2) repurchase, redeem or otherwise reacquire any of the Shares, or any rights, warrants or options to
acquire any of the Shares, other than: (A) repurchases of Shares outstanding as of the date hereof pursuant to the Company’s
right (under written commitments in effect as of the date hereof and copies of which have been made available to Parent) to purchase
Shares held by a Company Associate only upon termination of such Person’s employment or engagement by the Company; (B) repurchases
or forfeitures of Company Options, Company PSUs or Company RSUs (or, in each case, Shares issued upon the exercise thereof) outstanding
on the date hereof (in cancellation thereof) pursuant to the terms of any such Company Option or Company RSU (as in effect as of the
date hereof) between the Company and a Company Associate; (C) in connection with withholding to satisfy the exercise price and/or
Tax obligations with respect to Company Options, Company PSUs or Company RSUs pursuant to the terms thereof (as in effect as of the date
hereof); (D) between the Company and a wholly owned Acquired Corporation or between wholly owned Acquired Corporations; or (E) settlement
of any conversions of any of the Convertible Notes pursuant to the terms of the Convertible Notes Indentures;
(ii) split,
combine, subdivide or reclassify any Shares or other equity interests;
(iii) sell,
issue, grant, deliver, pledge, transfer, encumber or authorize the sale, issuance, grant, delivery, pledge, transfer or encumbrance of
(A) any capital stock, equity interest or other security, (B) any option, call, warrant, restricted securities or right to
acquire any capital stock, equity interest or other security, or (C) any instrument convertible into or exchangeable for any capital
stock, equity interest or other security (except (x) that the Company may issue Shares as required to be issued upon the settlement
of any conversions of any Convertible Notes pursuant to the terms of the Convertible Notes Indentures or the exercise or vesting (as
the case may be) of Company Options, Company PSUs or Company RSUs outstanding as of the date of this Agreement pursuant to the terms
thereof (as in effect as of the date hereof) or (y) with respect to sales, grants, pledges, transfers or encumbrances (or authorizations
with respect any of the foregoing) (1) constituting Permitted Encumbrances under clause (g) of the definition thereof
that are in existence as of the date of this Agreement or (2) constituting Encumbrances created or incurred in connection with any
Indebtedness permitted to be established or incurred under Section 5.2(b)(x));
(iv) except
as required under any Employee Plan as in effect on the date of this Agreement or as required by applicable Legal Requirements, (A) establish,
adopt, terminate or materially amend any Employee Plan (or any plan, program, arrangement or agreement that would be an Employee Plan
if it were in existence on the date hereof), (B) amend or waive any of its material rights under any Employee Plan (or any plan,
program, arrangement or agreement that would be an Employee Plan if it were in existence on the date hereof), (C) accelerate the
vesting or lapsing of restrictions or payment, or fund or in any other way secure the payment under any provision of any of the Employee
Plans (or any plan, program, arrangement or agreement that would be an Employee Plan if it were in existence on the date hereof), (D) grant
any employee, independent contractor or director any increase in compensation, bonuses or other benefits (except that the Company may
(x) provide base salary or wage rate increases for non-executive officer employees in the ordinary course of business consistent
with past practice that do not exceed 5% individually or 2.5% in the aggregate, (y) amend any Employee Plans to the extent required
by applicable Legal Requirements, and (z) make usual and customary annual or quarterly bonus payments for completed performance
periods based on actual performance in the ordinary course of business consistent with past practice), (E) forgive any loans or
issue any loans (other than routine travel advances issued in the ordinary course of business) to any employee of the Company or its
Subsidiaries, (F) hire any employee at the level of vice president or above, (G) hire any employee or engage any independent
contractor (who is a natural person) as a result of which the personnel expenses in any given quarter would exceed the Company’s
budgeted personnel expense (including temporary, consulting or advisory costs) provided to Parent prior to the date hereof or (H) terminate
the employment of any employee of the Company or its Subsidiaries at the level of vice president or above other than for cause;
(v) amend
or permit the adoption of any amendment to its certificate of incorporation or bylaws or other charter or organizational documents;
(vi) form
any Subsidiary, acquire (including by merger, consolidation, acquisition of stock or assets or otherwise), in one transaction or any
series of related transactions, directly or indirectly, any equity interest in or businesses (or any division thereof) of, any other
Entity or enter into any joint venture, partnership or similar revenue sharing arrangement;
(vii) make,
commit to make, or authorize any capital expenditure (except that the Acquired Corporations may make capital expenditures that do not
exceed $250,000 in the aggregate);
(viii) (x) acquire,
lease, sublease, license, sublicense, pledge, sell or otherwise dispose of, divest or spin-off, abandon, surrender, cancel, waive, relinquish,
covenant not to assert, or permit to lapse or expire, transfer, assign, guarantee, exchange or swap, mortgage or subject to any other
Encumbrance (other than Permitted Encumbrances) any material right or other material asset or property or Governmental Authorizations
(other than Intellectual Property Rights, which are addressed in Section 5.2(b)(ix)) (except (A) in the ordinary course
of business consistent with past practice, (B) pursuant to dispositions of obsolete, surplus or worn out assets that are no longer
useful in the conduct of the business of the Acquired Corporations, (C) capital expenditures permitted by clause (vii) of
this Section 5.2(b), (D) transactions between the Company and another Acquired Corporation or between Acquired Corporations
or (E) with respect to pledges, sales or other dispositions (x) constituting Permitted Encumbrances under clause (g) of
the definition thereof, (y) constituting Encumbrances created or incurred in connection with any Indebtedness permitted to be established
or incurred under Section 5.2(b)(x)) or (z) acquire assets or technologies, or rights in such assets or technologies,
of the nature described in Section 3.22(b)(i);
(ix) acquire,
sell, divest, transfer, assign, license, sublicense, grant a covenant not to assert or release with respect to, cancel, abandon, let
lapse, fail to diligently prosecute or use commercially reasonable efforts to enforce, create or incur any Encumbrance (other than a
Permitted Encumbrances) on, or otherwise dispose of, any Company IP or any other Intellectual Property Right used in the operation of
the businesses of the Acquired Corporations;
(x) lend
money or make capital contributions or advances to or make investments in, any Person, or incur, assume, guarantee, endorse or otherwise
become responsible for (or amend or modify the terms of) any Indebtedness for borrowed money (except for (A) advances to directors,
employees and consultants for travel and other business related expenses in the ordinary course of business consistent with past practice
and in compliance with the Company’s policies related thereto and (B) advances of expenses as required under the Company’s
certificate of incorporation or bylaws or any indemnification agreement with the Company’s directors or officers made available
to Parent before the date of this Agreement);
(xi) except
in the ordinary course of business consistent with past practice or as otherwise permitted by this Section 5.2(b), (A) amend
or modify in any material respect, or waive, assign or release any material rights or claims under, or voluntarily terminate, any Material
Contract (or any Contract that would have been a Material Contract if such Contract was in effect as of the execution and delivery of
this Agreement) or (B) enter into any Contract which would have been a Material Contract if such Contract was in effect as of the
execution and delivery of this Agreement;
(xii) except
as required by applicable Legal Requirements or GAAP, (A) make any material change to any accounting method or accounting period
used for Tax purposes; (B) make, rescind or change any material Tax election; (C) file an amended income or other material
amended Tax Return; (D) enter into a closing agreement with any Governmental Body regarding any material Tax liability or assessment;
(E) settle, compromise or consent to any material Tax claim or assessment or surrender a right to a material Tax refund; (F) waive
or extend the statute of limitations with respect to any income or other material Tax or income or other material Tax Return, other than
automatic waivers or extensions obtained in the ordinary course of business; (G) request any private letter, technical advice or
similar Tax ruling from any Governmental Body with respect to Taxes; or (H) enter into any Tax allocation, indemnity, sharing or
similar agreement (other than any such agreements entered into in the ordinary course of business consistent with past practice and not
relating primarily to Taxes);
(xiii) settle,
release, waive or compromise any Legal Proceeding or other claim (or threatened Legal Proceeding or other claim) against any Acquired
Corporation (including with respect to Company IP), other than, without prejudice to the limitations specified in Section 5.2(b)(xiv) and
solely with respect to Legal Proceedings unrelated to Company IP, any settlement, release, waiver or compromise that (A) results
solely in monetary obligations involving only the payment of monies by the Acquired Corporations of not more than $500,000 in the aggregate
(excluding monetary obligations that are funded by an indemnity obligation to, or an insurance policy of, any Acquired Corporations)
or (B) results in no monetary or other material non-monetary obligation of and does not involve the admission of wrongdoing by any
Acquired Corporation; provided that the settlement, release, waiver or compromise of any Legal Proceeding or claim brought by
the stockholders of the Company against the Company and/or its directors relating to the Transactions or a breach of this Agreement or
any other agreements contemplated hereby shall be subject to Section 2.7 or Section 6.5, as applicable, and not
this Section 5.2(b)(xiii);
(xiv) settle,
release, waive or compromise any Legal Proceeding or other claim (or threatened Legal Proceeding or other claim) brought by any Acquired
Corporation (including with respect to Company IP) (A) specified in Section 5.2(b)(xiv) of the Company Disclosure Schedule
or (B) where the settlement of such Legal Proceeding would have a material impact on the business of the Acquired Corporations,
taken as a whole, it being understood that, and for avoidance of doubt, any settlement of patent litigation can have a material impact
on the business of the Acquired Corporations, taken as a whole;
(xv) commence
any material Legal Proceeding against any Person, other than Parent, Purchaser or their respective Affiliates;
(xvi) (A) terminate
any Clinical Trials in respect of any Product Candidate that are ongoing as of the date of the Agreement, (B) commence (alone or
with any third party) any new Clinical Trial in respect of any Product Candidate or (C) commence any new research program under
the collaboration agreements set forth in Section 3.9(a)(vii) of the Company Disclosure Schedule;
(xvii) (A) create,
implement, operate, participate in, or offer any patient assistance or patient support program that offers, provides or intends to provide
free drug product (including any Product Candidate) or any cost-sharing assistance, such as co-pay coupons or co-pay cards in relation
to a drug product, to any patient, including any federal healthcare program beneficiaries (each, a “Patient Assistance Program”);
or (B) offer, make (whether in cash or in kind) any contribution that relates to or otherwise supports any third-party Patient Assistance
Program (including any co-pay assistance foundation) or other charitable contribution;
(xviii) fail
to use commercially reasonable efforts to (A) maintain in full force and effect the existing insurance policies of the Acquired
Corporations or (B) to renew or replace such insurance policies with comparable insurance policies;
(xix) enter
into any collective bargaining agreement or other agreement with any labor union or similar organization;
(xx) adopt
or implement any stockholder rights plan or similar arrangement;
(xxi) adopt
a plan or agreement of complete or partial liquidation or dissolution, merger, consolidation, restructuring, recapitalization or other
reorganization of any of the Acquired Corporations; or
(xxii) authorize
any of, or agree or commit to take, any of the actions described in the foregoing clauses (i) through (xxi) of
this Section 5.2(b).
Notwithstanding the foregoing, nothing contained herein shall give
to Parent or Purchaser, directly or indirectly, rights to control or direct the operations of the Acquired Corporations prior to the
Offer Acceptance Time. Prior to the Effective Time, each of Parent and the Company shall exercise, consistent with the terms and conditions
hereof, complete control and supervision of its and its, if applicable, Subsidiaries’ respective operations.
(c) In
furtherance and not in limitation of any other provision of this Agreement, to the extent permitted by applicable Legal Requirements,
the Company shall keep Parent reasonably informed on a current basis of any developments, material discussions or negotiations with the
FDA or any other Healthcare Regulatory Authority relating to any Product Candidate. Without limiting the generality of the foregoing,
to the extent permitted by applicable Legal Requirements, the Company shall (i) consult with Parent and promptly inform Parent,
and provide Parent with a reasonable opportunity to review, as reasonably in advance as practicable under the circumstances, and consider
in good faith Parent’s comments to or in connection with any meetings, filings, submissions (including any supplementary new drug
applications), material correspondence (including briefing materials) or other material communications made by or on behalf of any Acquired
Corporation to, between or with the FDA or any other Healthcare Regulatory Authority; (ii) prepare or cause to be prepared, and
promptly provide to Parent copies of minutes or draft minutes of any meetings between the Acquired Corporations and their representatives,
on the one hand, and the FDA or any other Healthcare Regulatory Authority, on the other hand; (iii) prepare or cause to be prepared,
and promptly provide to Parent copies of meeting agendas and minutes or draft minutes of any meetings of a joint steering committee or
similar body, to which any Acquired Corporation participates, established by the Acquired Corporations and any third party; (iv) promptly
provide to Parent, upon finalization of such reports, reports prepared by the Company or any Collaboration Partner that have been provided
to the Company relating to chemistry, manufacturing and controls (CMC), clinical or non-clinical reports and topline results, in each
case, relating to any Clinical Trial involving any Product Candidate, provided, however, that the foregoing subclause (iv) shall
not require any of the Acquired Corporations to provide any such reports to Parent if such disclosure would contravene any Contract between
an Acquired Corporation and a Collaboration Partner (provided that the Acquired Corporations shall use reasonable efforts to seek
any Consent that may be required pursuant to such Contract to provide such reports); (v) promptly inform Parent and provide Parent
or Purchaser with a reasonable opportunity (but no fewer than five (5) business days) to comment, in each case, prior to making,
or proposing to make, any material change to any study protocol, adding any new Clinical Trial, making any material change to a manufacturing
plan or process, making any material change to a development timeline or initiating, or making any material change to, promotional or
marketing materials or activities relating to any Product Candidate; and (vi) promptly update Parent in writing with respect to
any proposal to make a change described in the foregoing clause (v) that would materially affect the timing and cost of the
development of any Product Candidate. The Acquired Corporations shall promptly submit information and data to the relevant Healthcare
Regulatory Authority in response to specific inquiries from such Healthcare Regulatory Authority, including information and data to be
submitted in connection with the matters described in Section 3.12(i) of the Company Disclosure Schedule.
5.3 No
Solicitation.
(a) For
the purposes of this Agreement, “Acceptable Confidentiality Agreement” means any customary confidentiality agreement
that (i) contains provisions that are not materially less favorable to the Company than those contained in the Confidentiality Agreement
and (ii) does not prohibit the Company from providing any information to Parent in accordance with this Agreement.
(b) Except
as permitted by this Section 5.3, during the Pre-Closing Period (i) the Company shall, and shall cause the other Acquired
Corporations and its and their respective Representatives to cease any direct or indirect solicitation, facilitation, encouragement,
discussions or negotiations with any Persons that may be ongoing with respect to an Acquisition Proposal and (ii) the Company shall
not, and shall cause the other Acquired Corporations and its and their respective directors, officers and employees not to, and shall
use reasonable best efforts to cause their other Representatives not to, (A) continue any solicitation, knowing encouragement or
facilitation (including by way of furnishing non-public information), discussions or negotiations with any Persons that may be ongoing
with respect to an Acquisition Proposal; (B) directly or indirectly (x) solicit, initiate or knowingly facilitate or encourage
(including by way of furnishing non-public information) any inquiries regarding, or the making of any proposal or offer that constitutes,
or could reasonably be expected to lead to, an Acquisition Proposal, (y) engage in, continue or otherwise participate in any discussions
or negotiations regarding, or furnish to any other Person any non-public information in connection with, or for the purpose of soliciting
or knowingly encouraging or facilitating, any Acquisition Proposal or any proposal or offer that could reasonably be expected to lead
to an Acquisition Proposal or (z) enter into any letter of intent, transaction agreement, agreement in principle or other agreement
with respect to an Acquisition Proposal or any proposal or offer that could reasonably be expected to lead to an Acquisition Proposal;
(C) waive or release any Person from, forebear in the enforcement of, or amend any standstill agreement or any standstill provisions
of any other Contract, unless in the case of this clause (C), the Board of Directors determines in good faith, after consultation
with the Company’s outside legal counsel and financial advisors, that the failure to do so would be inconsistent with the fiduciary
duties of the Board of Directors to the Company’s stockholders under applicable Delaware law, in which event the Acquired Corporations
may take the preceding actions described in this clause (C) solely to the extent necessary to permit a third party to make,
on a confidential basis, to the Board of Directors, an Acquisition Proposal; or (D) resolve to do, or agree or announce an intention
to do, any of the foregoing under the preceding clauses (A) through (C). As promptly as reasonably practicable (and
in any event within 24 hours) following the date hereof, the Company shall discontinue electronic or physical data room access granted
and request and otherwise seek the prompt return or destruction (to the extent provided for by the applicable confidentiality agreement)
of all non-public information previously furnished to any Person (other than Parent and Parent’s Representatives) within the one
(1)-year period prior to the date of this Agreement in connection with any Acquisition Proposal or in connection with the Company’s
evaluation of strategic alternatives leading up to the Transactions.
(c) Notwithstanding
anything in this Agreement to the contrary, if at any time after the execution and delivery of this Agreement and prior to the Offer
Acceptance Time any Acquired Corporation or any of their Representatives receives a bona fide written Acquisition Proposal from
any Person or group of Persons, which Acquisition Proposal was made or renewed after the execution and delivery of this Agreement, and
did not result from a breach of this Section 5.3, and the Board of Directors determines in good faith, after consultation
with its outside legal counsel and financial advisors, that such Acquisition Proposal constitutes or could reasonably be expected to
lead to a Superior Offer and the failure to take such action described in clauses (x) and (y) of this Section 5.3(c) would
be inconsistent with its fiduciary duties under Delaware law, then the Company and its Representatives may (x) furnish, pursuant
to an Acceptable Confidentiality Agreement (an executed copy of which shall be provided by the Company to Parent solely for informational
purposes within 24 hours of the execution thereof), information (including non-public information) with respect to the Acquired Corporations
to the Person or group of Persons who has made such Acquisition Proposal and the Representatives of such Person or group of Persons;
provided that the Company shall as promptly as practicable (and no later than within 24 hours) provide to Parent any non-public
information concerning the Acquired Corporations that is provided to any such Person or Representative to the extent access to such information
was not previously provided to Parent or its Representatives and (y) engage in or otherwise participate in discussions or negotiations
with any such Person or Representative with respect to such Acquisition Proposal; provided, in the case of the preceding clauses
(x) and (y), that at or prior to the first time that the Company furnishes any information to or participates in any
discussions or negotiations with any such Person on or after the date of this Agreement, the Company shall provide written notice to
Parent of such determination of the Board of Directors described in this Section 5.3(c), together with the identity of such
Person or group making such Acquisition Proposal.
(d) During
the Pre-Closing Period, the Company shall (i) promptly (and in any event within one (1) business day) notify Parent if any
inquiries, proposals or offers with respect to an Acquisition Proposal are received by any Acquired Corporation or any Representative
thereof and provide to Parent a copy of any written Acquisition Proposal (including any proposed term sheet, letter of intent, acquisition
agreement or similar agreement provided to the Acquired Corporations in connection therewith) and a summary of any material unwritten
terms and conditions thereof (and indicate the identity of such Person), and (ii) keep Parent reasonably informed of any material
developments, discussions or negotiations relating thereto on a prompt basis (and in any event within 24 hours of such material development,
discussion or negotiation).
(e) Nothing
in this Section 5.3 or elsewhere in this Agreement shall prohibit the Company from (i) disclosing to the stockholders
of the Company any “stop, look and listen” communication pursuant to Rule 14d-9(f) promulgated under the Exchange
Act, or (ii) making any disclosure to the stockholders of the Company that is required by applicable Legal Requirements.
(f) The
Company agrees that in the event any Representative of any Acquired Corporation takes any action that, if taken by any Acquired Corporation,
would constitute a breach of this Section 5.3, the Company shall be deemed to be in breach of this Section 5.3.
Section 6
ADDITIONAL
COVENANTS OF THE PARTIES
6.1 Company
Board Recommendation.
(a) Subject
to Section 6.1(b), the Company hereby consents to the inclusion of a description of the Company Board Recommendation in the
Offer Documents. During the Pre-Closing Period, subject to Section 6.1(b), neither the Board of Directors nor any committee
thereof shall (i)(A) withdraw, withhold or qualify (or modify in a manner adverse to Parent or Purchaser), or publicly propose to
withdraw, withhold or qualify (or modify in a manner adverse to Parent or Purchaser), the Company Board Recommendation, (B) approve,
adopt, endorse, recommend or declare advisable, or publicly propose to approve, adopt, endorse, recommend or declare advisable, any Acquisition
Proposal, (C) after the public announcement of an Acquisition Proposal (other than a tender offer or exchange offer), fail to publicly
affirm the Company Board Recommendation within three (3) business days after a written request by Parent to do so (or, if earlier,
by the close of business on the business day immediately preceding the scheduled date of the Offer Acceptance Time), which request may
be made once per applicable Acquisition Proposal, provided that Parent shall be entitled to make a new request each time there
is a publicly disclosed material change in such applicable Acquisition Proposal, (D) following the commencement of a tender offer
or exchange offer relating to the Shares by a Person unaffiliated with Parent, fail to publicly affirm the Company Board Recommendation
and recommend that the Company’s stockholders reject such tender offer or exchange offer within ten (10) business days after
the commencement of such tender offer or exchange offer pursuant to Rule 14e-2(a) promulgated under the Exchange Act (or, if
earlier, by the close of business on the business day immediately preceding the scheduled date of the Offer Acceptance Time), (E) fail
to include the Company Board Recommendation in the Schedule 14D-9 when filed with the SEC or disseminated to the Company’s stockholders,
or (F) other than under the circumstances addressed in the preceding subclauses (C) and (D), fail to publicly
reaffirm the Company Board Recommendation within five (5) business days after Parent so requests in writing (it being understood
that the Company will have no obligation to make such reaffirmation on more than three (3) occasions) (any action described in this
clause (i) being referred to as a “Company Adverse Recommendation Change”), or (ii) approve, adopt,
endorse, recommend or declare advisable, or propose to approve, adopt, endorse, recommend or declare advisable, or allow the Company
to execute or enter into any letter of intent, agreement in principle, acquisition agreement or other Contract with respect to, or that
is intended to or would reasonably be expected to lead to, any Acquisition Proposal, or Contract that would require, or would reasonably
be expected to cause, the Company to abandon, terminate, delay or fail to consummate, or that would otherwise materially impede, interfere
with or be inconsistent with, the Transactions (other than an Acceptable Confidentiality Agreement).
(b) Notwithstanding
anything to the contrary contained in this Agreement, at any time prior to the Offer Acceptance Time:
(i) if
at any time after the execution and delivery of this Agreement and prior to the Offer Acceptance Time, the Company has received a bona
fide written Acquisition Proposal from any Person that has not been withdrawn and, after consultation with outside legal counsel,
the Board of Directors shall have determined, in good faith, that such Acquisition Proposal is a Superior Offer, then (x) the Board
of Directors may make a Company Adverse Recommendation Change, or (y) subject to the Company’s compliance with Section 8.3(b),
the Company may terminate this Agreement pursuant to Section 8.1(e) solely if the Acquired Corporations and their Representatives
shall have complied in all material respects with the provisions of Section 5.3 and this Section 6.1 with respect
to such Superior Offer, but in each case under (x) or (y), if and only if: (A) the Board of Directors determines in good faith,
after consultation with the Company’s outside legal counsel, that the failure to do so would be inconsistent with the fiduciary
duties of the Board of Directors to the Company’s stockholders under applicable Delaware law; (B) the Company shall have given
Parent prior written notice of its intention to consider making a Company Adverse Recommendation Change or terminating this Agreement
pursuant to Section 8.1(e) at least five (5) business days prior to making any such Company Adverse Recommendation
Change or termination (a “Determination Notice”) (which notice shall not constitute a Company Adverse Recommendation
Change or termination) and, if desired by Parent, during such five (5)-business day period shall have negotiated, and shall have caused
its Representatives to negotiate, in good faith with respect to any revisions to the terms of this Agreement or another proposal, to
the extent proposed by Parent, so that such Acquisition Proposal would cease to constitute a Superior Offer; and (C) (1) the
Company shall have provided, and shall have caused its Representatives to provide, to Parent information with respect to such Acquisition
Proposal in accordance with Section 5.3(c) and Section 5.3(d), including the proposed definitive agreements
(and any related agreements) among any Acquired Corporation and any Person or group of Persons making such Acquisition Proposal, (2) the
Company shall have given Parent the five (5)-business day period after the Determination Notice to propose revisions to the terms of
this Agreement or make another proposal so that such Acquisition Proposal would cease to constitute a Superior Offer, and (3) after
giving effect to the proposals made by Parent during such period, if any, after consultation with the Company’s outside legal counsel
and financial advisors, the Board of Directors shall have determined, in good faith, that such Acquisition Proposal is a Superior Offer
and that the failure to make the Company Adverse Recommendation Change or terminate this Agreement pursuant to Section 8.1(e) would
be inconsistent with the fiduciary duties of the Board of Directors to the Company’s stockholders under Delaware law. Issuance
of any “stop, look and listen” communication by or on behalf of the Company pursuant to Rule 14d-9(f) promulgated
under the Exchange Act shall not be considered a Company Adverse Recommendation Change and shall not require the giving of a Determination
Notice or compliance with the procedures set forth in this Section 6.1. The provisions of this Section 6.1(b)(i) shall
also apply to any change to any of the material terms (including the form and amount of consideration) to any Acquisition Proposal and
shall require a new Determination Notice, except that the references to five (5) business days shall be deemed to be three (3) business
days; and
(ii) other
than in connection with any Acquisition Proposal the Board of Directors may make a Company Adverse Recommendation Change if and only
if: (A) an Intervening Event has occurred; (B) the Company shall have given Parent a Determination Notice at least five (5) business
days prior to making any such Company Adverse Recommendation Change and, if desired by Parent, during such five (5)-business day period
shall have negotiated, and shall have caused its Representatives to negotiate, in good faith with respect to any revisions to the terms
of this Agreement or another proposal to the extent proposed by Parent so that a Company Adverse Recommendation Change would no longer
be necessary; and (C) (1) the Determination Notice specifies in reasonable detail the facts and circumstances relating to such
Intervening Event that render a Company Adverse Recommendation Change necessary and includes such documents, information and data in
the Acquired Corporations’ possession as reasonably relate to such Intervening Event, (2) the Company shall have given Parent
the five (5)-business day period after the Determination Notice to propose revisions to the terms of this Agreement or make another proposal
so that a Company Adverse Recommendation Change in response to such Intervening Event would no longer be necessary, and (3) after
giving effect to the proposals made by Parent during such period, if any, after consultation with the Company’s outside legal counsel
and financial advisors, the Board of Directors shall have determined, in good faith, that the failure to make the Company Adverse Recommendation
Change would be inconsistent with the fiduciary duties of the Board of Directors to the Company’s stockholders under Delaware law.
6.2 Filings,
Consents and Approvals.
(a) The
Parties agree to use their respective reasonable best efforts to take or cause to be taken promptly any and all steps necessary to avoid
or eliminate each and every impediment under applicable Legal Requirements, including applicable Antitrust Laws, that may be asserted
by any Governmental Body or any other party, so as to enable the Closing to occur as promptly as practicable, but in no case later than
the End Date including providing as promptly as reasonably practicable all information required by any Governmental Body pursuant to
its evaluation of the Transactions under the HSR Act of other applicable Antitrust Laws (including any request for additional information
pursuant to the HSR Act). Subject to the limitations specified in the following sentence, the Parties shall defend through litigation
on the merits any claim asserted in court by any party under Antitrust Laws in order to avoid entry of, or to have vacated or terminated,
any decree, order or judgment (whether temporary, preliminary or permanent) that could restrain, delay, or prevent the Closing by the
End Date. Notwithstanding anything to the contrary contained in this Agreement, neither Parent nor Purchaser nor their respective Affiliates
shall be obligated to take any of the following actions (and, without Parent’s prior written consent, the Acquired Corporations
shall not take any of the following actions in furtherance of this Section 6.2(a)): (i) proposing, negotiating, committing
to or effecting, by consent decree, hold separate order or otherwise, the sale, divestiture, license, hold separate or other disposition
of any asset, interest or business; (ii) terminating, relinquishing, modifying, transferring, assigning, restructuring, or waiving
existing agreements, collaborations, relationships, ventures, contractual rights, obligations or other arrangements; and (iii) any
other behavioral undertakings and commitments whatsoever including but not limited to creating or consenting to create any relationships,
ventures, contractual rights, obligations, or other arrangements and, in each case, to enter, or offer to enter, into agreements and
stipulate to the entry of an order or decree or file appropriate applications with any Governmental Body in connection with any of the
foregoing. No Party shall commit to or agree with any Governmental Body to stay, toll or extend any applicable waiting period or withdraw
its filing under the HSR Act or any other applicable Antitrust Laws, or enter into any similar timing agreement, without the prior written
consent of the other Parties (such consent not to be unreasonably withheld, delayed or conditioned).
(b) Subject
to the terms and conditions of this Agreement, each of the Parties shall (and shall cause their respective Affiliates, if applicable,
to): (i) promptly, but in no event later than ten (10) business days after the date hereof, unless otherwise mutually agreed
to by the Parties, make an appropriate filing of all notification and report forms as required by the HSR Act with respect to the Transactions
and (ii) cooperate with each other in determining whether, and promptly prepare and make, any other filings, notifications or other
consents are required to be made with, or obtained from, any other Governmental Bodies in connection with the Transactions.
(c) Without
limiting the generality of anything contained in this Section 6.2, during the Pre-Closing Period, each Party shall (i) give
the other Parties prompt notice of the making or commencement of any request, inquiry, investigation, action or Legal Proceeding brought
by a Governmental Body or brought by a third party before any Governmental Body, in each case, with respect to the Transactions, (ii) keep
the other Parties reasonably informed as to the status of any such request, inquiry, investigation, action or Legal Proceeding, (iii) promptly
inform the other Parties of, and wherever practicable give the other Parties reasonable advance notice of, and the opportunity to participate
in, any communication to or from the FTC, DOJ or any other Governmental Body in connection with any such request, inquiry, investigation,
action or Legal Proceeding, (iv) promptly furnish to the other Parties, subject to an appropriate confidentiality agreement to limit
disclosure to counsel and outside consultants, copies of documents provided to or received from any Governmental Body in connection with
any such request, inquiry, investigation, action or Legal Proceeding (other than “4(c) documents” as that term is used
in the rules and regulations under the HSR Act, that contain valuation information (which can be redacted)), (v) subject to
an appropriate confidentiality agreement to limit disclosure to counsel and outside consultants, and to the extent reasonably practicable,
consult and cooperate with the other Parties and consider in good faith the views of the other Parties in connection with any analysis,
appearance, presentation, memorandum, brief, argument, opinion or proposal made or submitted in connection with any such request, inquiry,
investigation, action or Legal Proceeding, and (vi) except as may be prohibited by any Governmental Body or by any Legal Requirement,
in connection with any such request, inquiry, investigation, action or Legal Proceeding in respect of the Transactions, give the other
party reasonable advance notice of, and permit authorized Representatives of the other Party to be present at each meeting or conference
relating to such request, inquiry, investigation, action or Legal Proceeding and to have access to and be consulted in connection with
any argument, opinion or proposal made or submitted to any Governmental Body in connection with such request, inquiry, investigation,
action or Legal Proceeding. Notwithstanding anything to the contrary in this Section 6.2, the Parties may redact materials
provided to one another (A) to remove competitively sensitive information or information concerning valuation; (B) as necessary
to comply with Legal Requirements and Contracts; and (C) as necessary to address reasonable attorney-client privilege or other privilege
or confidentiality concerns.
6.3 Employee
Benefits. For a period of one (1) year following the Effective Time (the “Continuation
Period”), Parent shall provide, or cause to be provided, to each employee of the Company or its Subsidiaries who is employed
by the Company or its Subsidiaries as of immediately prior to the Effective Time and who continues to be actively employed by the Surviving
Corporation (or any Affiliate thereof, including, for the avoidance of doubt, Parent and Parent’s Affiliates) during such one (1)-year
period (each, a “Continuing Employee”) with (i) a base salary or wage rate that is no less than that provided
to such Continuing Employee by any Acquired Corporation immediately prior to the Effective Time, (ii) target short-term cash incentive
compensation opportunities that are no less favorable, in the aggregate, than those provided to such Continuing Employee by any Acquired
Corporation immediately prior to the Effective Time and (iii) other employee benefits (excluding equity incentive and long-term
incentive compensation) that in the aggregate are no less favorable than those provided to such Continuing Employee by any Acquired Corporation
immediately prior to the Effective Time. Without limiting the foregoing:
(a) Parent
shall cause all Continuing Employees to be eligible to continue to participate in the Surviving Corporation’s health and welfare
benefit plans (to the same extent such Continuing Employees were eligible to participate under the Company’s health and welfare
benefit plans immediately prior to the Effective Time); provided, however, that (i) nothing in this Section 6.3
or elsewhere in this Agreement shall limit the right of Parent or the Surviving Corporation to amend or terminate any such health or
welfare benefit plan at any time and (ii) if Parent or the Surviving Corporation terminates any such health or welfare benefit plan
then the Continuing Employees shall be eligible to participate in the Surviving Corporation’s (or an Affiliate’s, including,
for the avoidance of doubt, Parent’s and Parent’s Affiliates’) corresponding health and welfare benefit plan. To the
extent that service is relevant for eligibility or vesting under any benefit plan of Parent and/or the Surviving Corporation, then Parent
shall ensure that such benefit plan shall, for purposes of eligibility and vesting, but not for purposes of defined benefit pension accrual
or for purposes of qualifying for subsidized early retirement, credit Continuing Employees for service prior to the Effective Time with
the Company and its Affiliates or their respective predecessors to the same extent that such service was recognized prior to the Effective
Time under the corresponding benefit plan of the Company except to the extent it would result in a duplication of benefits. In addition,
Parent and/or the Surviving Corporation shall credit each Continuing Employee with paid time off equal to the accrued paid time off such
Continuing Employee had accrued with the Company that was unused as of the Effective Time. To the extent that service is relevant for
benefit levels, including severance benefits and paid time off accruals, following the Effective Time, Parent shall ensure that any employee
plan of Parent and/or the Surviving Corporation shall, for purposes of benefit levels, credit Continuing Employees for service prior
to the Effective Time with the Company to the same extent that such service was recognized prior to the Effective Time except to the
extent it would result in a duplication of benefits.
(b) Following
the Effective Time, Parent or an Affiliate of Parent shall use commercially reasonable efforts to (i) waive any preexisting condition
limitations otherwise applicable to Continuing Employees and their eligible dependents under any plan of Parent or an Affiliate that
provides health benefits in which Continuing Employees are eligible to participate following the Effective Time, other than any limitations
that were in effect with respect to such employees immediately prior to the Effective Time under the corresponding Employee Plan, (ii) honor
any deductible, co-payment and out-of-pocket maximums incurred by the Continuing Employees and their eligible dependents under the health
plans in which they participated immediately prior to transitioning into a plan of Parent or an Affiliate during the portion of the plan
year prior to such transition in satisfying any deductibles, co-payments or out-of-pocket maximums under health plans of Parent or an
Affiliate and (iii) waive any waiting period limitation or evidence of insurability requirement that would otherwise be applicable
to a Continuing Employee and his or her eligible dependents on or after the Effective Time, in each case to the extent such Continuing
Employee or eligible dependent had satisfied any similar limitation or requirement under an analogous Employee Plan prior to the Effective
Time.
(c) Parent
shall provide, or shall cause its Affiliates to provide, each Continuing Employee who experiences a qualifying termination of employment
from Parent or any of their respective Affiliates during the Continuation Period with severance benefits that are no less favorable,
in the aggregate, than those that would have been provided to such Continuing Employee by any Acquired Corporation under the applicable
severance policies set forth on Section 6.3(c) of the Company Disclosure Schedule had such termination occurred prior to the
Effective Time.
(d) Prior
to the Effective Time, if requested by Parent in writing, to the extent permitted by applicable Legal Requirements and the terms of the
applicable plan or arrangement, the Company shall cause the Company’s 401(k) Plan (the “Company 401(k) Plan”)
to be terminated effective immediately prior to the Effective Time. In the event that Parent requests that the Company 401(k) Plan
be terminated, the Company shall provide Parent with evidence that such Company 401(k) Plan has been terminated (the form and substance
of which shall be subject to review and approval by Parent) not later than the day immediately preceding the Effective Time.
(e) Prior
to making any written or oral communications to the directors, officers or employees of the Company or any of its Subsidiaries pertaining
to compensation or benefit matters that are affected by the transactions contemplated by this Agreement, the Company shall provide Parent
with a copy of the intended communication, Parent shall have a reasonable period of time to review and comment on the communication,
and the Company shall consider any such comments in good faith; provided that the Company is not required to provide any such
communication for Parent’s review if such communication is consistent with previous press releases, public disclosures or other
communications made by Parent or the Company in compliance with Section 6.7 or this Section 6.3(e).
(f) The
provisions of this Section 6.3 are solely for the benefit of the Parties, and no provision of this Section 6.3
is intended to, or shall, constitute the establishment or adoption of or an amendment to any employee benefit plan for purposes of ERISA
or otherwise prevent Parent, the Surviving Corporation or any of their Affiliates from amending or terminating any of their benefit plans
or, after the Effective Time, any Employee Plan in accordance with their terms, and no current or former employee or any other individual
associated therewith shall be regarded for any purpose as a third party beneficiary of the Agreement or have the right to enforce the
provisions hereof. Nothing in this Section 6.3 or elsewhere in this Agreement shall be construed to create a right in any
Person to employment with Parent, the Surviving Corporation or any other Affiliate of the Surviving Corporation or to any compensation
or benefits and the employment of each Continuing Employee shall be “at will” employment.
6.4 Indemnification
of Officers and Directors.
(a) The
Parties agree that all rights to indemnification, advancement of expenses and exculpation from liabilities for acts or omissions occurring
at or prior to the Effective Time (whether asserted or claimed prior to, at or after the Effective Time) now existing in favor of the
current or former directors or officers of any Acquired Corporation under the certificate of incorporation, bylaws or other charter or
organizational documents of any of the Acquired Corporations, as applicable, and those agreements listed in Section 3.9(a)(xvii) of
the Company Disclosure Schedule, shall continue in full force and effect in accordance with their terms, and the Acquired Corporations
shall perform their obligations thereunder, in each case, for a period of six (6) years from the Effective Time. Without limiting
the foregoing, for a period of six (6) years from the Effective Time, Parent shall, and shall cause the Surviving Corporation to,
indemnify and hold harmless each individual who is as of the date of this Agreement, or who becomes prior to the Effective Time, a director
or officer of any Acquired Corporation or who is as of the date of this Agreement, or who thereafter commences prior to the Effective
Time, serving at the request of any Acquired Corporation as a director or officer of another Person (the “Indemnified Persons”),
against all claims, losses, liabilities, damages, judgments, inquiries, fines and reasonable fees, costs and expenses, including reasonable
and documented attorneys’ fees and disbursements, incurred in connection with any claim, action, suit or proceeding, whether civil,
criminal, administrative or investigative (including with respect to matters existing or occurring at or prior to the Effective Time,
including this Agreement and the transactions and actions contemplated hereby), arising out of or pertaining to the fact that the Indemnified
Person is or was a director or officer of any Acquired Corporation or is or was serving at the request of any Acquired Corporation as
a director or officer of another Person, whether asserted or claimed prior to, at or after the Effective Time, to the fullest extent
permitted under applicable Legal Requirements. In the event of any such claim, action, suit or proceeding, (x) each Indemnified
Person shall be entitled to the advancement of expenses incurred in defense thereof from Parent or any of its Subsidiaries; provided
that the Indemnified Person to whom expenses are advanced provides an undertaking to repay such amount if it shall ultimately be
determined that such Indemnified Person is not entitled to be indemnified pursuant to the preceding sentence and (y) the Indemnified
Persons and Parent shall, and Parent shall cause its Subsidiaries to, reasonably cooperate in the defense of any such matter.
(b) If
and only if the Company fails to purchase “tail” insurance policies of the type contemplated by the second sentence of this
Section 6.4(b), for a period of six (6) years from and after the Effective Time, Parent and the Surviving Corporation
shall either cause to be maintained in effect the current policies of directors’ and officers’ insurance maintained by or
for the benefit of the Acquired Corporations or purchase substitute policies for the Acquired Corporations and their current and former
directors and officers who are currently covered by the directors’ and officers’ insurance coverage currently maintained
by or for the benefit of the Acquired Corporations, in either case, of not less than the existing coverage and having other terms not
less favorable to the insured persons than the directors’ and officers’ insurance coverage currently maintained by or for
the benefit of the Acquired Corporations with respect to claims arising from facts or events that existed or occurred at or before the
Effective Time (with insurance carriers having at least an “A” rating by A.M. Best), except that in no event shall Parent
or the Surviving Corporation be required to pay with respect to such insurance more than 300% of the aggregate annual premium most recently
paid by the Acquired Corporations prior to the date of this Agreement (the “Maximum Amount”), and if the Surviving
Corporation is unable to obtain the coverage required by this Section 6.4(b) it shall obtain as much comparable coverage
as possible for the years within such six (6)-year period for a premium equal to the Maximum Amount. Prior to the Closing Date, the Company
shall use its reasonable best efforts to purchase “tail” directors’ and officers’ insurance for the Acquired
Corporations and their current and former directors and officers who are currently covered by the directors’ and officers’
insurance coverage currently maintained by or for the benefit of the Acquired Corporations, such “tail” insurance to provide
coverage in an amount not less than the existing coverage and to have other terms not less favorable to the insured persons than the
directors’ and officers’ insurance coverage currently maintained by or for the benefit of the Acquired Corporations, in each
case with respect to claims arising from facts or events that existed or occurred at or before the Effective Time; provided that
in no event shall the aggregate premium for any such “tail” insurance exceed the Maximum Amount. Parent shall cause the Surviving
Corporation to maintain such insurance in full force and effect and continue to honor the obligations thereunder.
(c) In
the event that any Acquired Corporation or any of its successors or assigns (i) consolidates with or merges into any other Person
and is not the continuing or surviving corporation or Entity of such consolidation or merger or (ii) transfers or conveys all or
substantially all of its properties and assets to any Person, then, and in each such case, the Acquired Corporation or such successors
and assigns, as applicable, shall cause proper provision to be made so that such successors and assigns of such Acquired Corporation
assume the obligations set forth in this Section 6.4.
(d) The
provisions of this Section 6.4 (i) shall survive the Offer Acceptance Time and the Effective Time and (ii) are
intended to be for the benefit of, and will be enforceable by, each Indemnified Person and the heirs, successors, assigns and representatives
thereof, each in their capacities as such, and (iii) are in addition to, and not in substitution for, any other rights to indemnification,
advancement of expenses, exculpation or contribution that any such Indemnified Person may have by contract or otherwise. Unless required
by applicable Legal Requirement, this Section 6.4 may not be amended, altered or repealed after the Offer Acceptance Time
in such a manner as to adversely affect the rights of any Indemnified Person or any of their successors, assigns or heirs without the
prior written consent of the affected Indemnified Person.
6.5 Stockholder
Litigation. The Company shall give Parent the right to review and comment on all material filings
or responses to be made by the Company (and shall give reasonable consideration to Parent’s comments and other advice) in connection
with, and the opportunity to participate in, any litigation against the Company and/or its directors or officers relating to the Transactions,
and the right to consult on any settlement with respect to such litigation, and no such settlement shall be agreed to or offered without
Parent’s prior written consent (such consent not to be unreasonably withheld, conditioned or delayed). The Company shall promptly
notify Parent of any such litigation and shall keep Parent reasonably and promptly informed with respect to the status thereof.
6.6 Additional
Agreements. Subject to the terms and conditions of this Agreement, including Section 6.2(a),
Parent and the Company shall use reasonable best efforts to take, or cause to be taken, all actions necessary to consummate the Offer
and the Merger and make effective the other Transactions. Without limiting the generality of the foregoing, subject to the terms and
conditions of this Agreement, each Party to this Agreement shall use reasonable best efforts to (a) make all filings (if any) and
give all notices (if any) required to be made and given by such Party pursuant to any Material Contract in connection with the Offer
and the Merger and the other Transactions, (b) seek each Consent (if any) required to be obtained pursuant to any Material Contract
by such Party in connection with the Transactions to the extent requested in writing by Parent and (c) seek to lift any restraint,
injunction or other legal bar to the Offer or the Merger brought by any third Person against such Party. The Company shall promptly deliver
to Parent a copy of each such filing made, each such notice given and each such Consent obtained by the Company during the Pre-Closing
Period.
6.7 Disclosure.
(a) The
initial press release relating to this Agreement shall be a joint press release issued by the Company and Parent and thereafter Parent
and the Company shall consult with each other before issuing any further press release(s) or otherwise making any public statement
(to the extent not previously issued or made in accordance with this Agreement) with respect to the Offer, the Merger, this Agreement
or any of the other Transactions and shall not issue any such press release or public statement without the other Party’s written
consent (such consent not to be unreasonably withheld, conditioned or delayed). Notwithstanding the foregoing, (i) each Party may,
without such consultation or consent, make any public statement in response to questions from the press, analysts, investors or those
attending industry conferences, make internal announcements to employees and make disclosures in Company SEC Documents, so long as such
statements are consistent with previous press releases, public disclosures or public statements made jointly by the Parties (or individually,
if approved by the other Party); (ii) each Party may, without the prior consent of the other Party but subject to giving advance
notice to, and giving reasonable consideration to any comments from, the other Party, issue any such press release or make any such public
announcement or statement as may be required by Legal Requirement; provided, however, that if any such press release, announcement
or statement relate to an Acquisition Proposal or an Intervening Event and do not reaffirm the Company Board Recommendation in such disclosure
or communication or have the effect of withdrawing, qualifying or modifying the Company Board Recommendation in a manner adverse to Parent,
such disclosure or communication shall constitute a Company Adverse Recommendation Change; and (iii) the Company need not consult
with Parent in connection with such portion of any press release, public statement or filing to be issued or made pursuant to Section 5.3(e) or
with respect to any Acquisition Proposal or Company Adverse Recommendation Change.
(b) The
Company will make available to Parent solely for informational purposes and on a non-reliance basis, a signed copy of each of the Barclays
Fairness Opinion and Centerview Fairness Opinion as soon as possible following the date of this Agreement.
6.8 Takeover
Laws. If any Takeover Law may become, or may purport to be, applicable to the Transactions,
the Company and the Board of Directors shall use their respective reasonable best efforts to grant such approvals and take such actions
as are necessary so that the Transactions may be consummated as promptly as practicable on the terms and conditions contemplated hereby
and otherwise act to lawfully eliminate the effect of any Takeover Law on any of the Transactions.
6.9 Section 16
Matters. The Company, and the Board of Directors, shall, to the extent necessary, take appropriate
action, prior to or as of the Offer Acceptance Time, to approve, for purposes of Section 16(b) of the Exchange Act, the disposition
and cancellation or deemed disposition and cancellation of Shares, Company Options, Company PSUs and Company RSUs in the Merger by applicable
individuals in order to cause such dispositions and/or cancellations to be exempt under Rule 16b-3 promulgated under the Exchange
Act.
6.10 Rule 14d-10
Matters. Prior to the Offer Acceptance Time and to the extent permitted by applicable Legal
Requirements, the compensation committee of the Board of Directors will approve, as an “employment compensation, severance or other
employee benefit arrangement” within the meaning of Rule 14d-10(d)(2) under the Exchange Act, each agreement, arrangement
or understanding between Purchaser, the Company or their respective Affiliates and any of the officers, directors or employees of the
Company or its Subsidiaries that are effective as of the date of this Agreement or are entered into after the date of this Agreement
and prior to the Offer Acceptance Time pursuant to which compensation is paid to such officer, director or employee and will take all
other action reasonably necessary to satisfy the requirements of the non-exclusive safe harbor set forth in Rule 14d-10(d)(2) under
the Exchange Act.
6.11 Stock
Exchange Delisting; Deregistration. Prior to the Closing Date, the Company shall cooperate with
Parent and use its reasonable best efforts to take, or cause to be taken, all actions, and do or cause to be done all things, reasonably
necessary, proper or advisable on its part under applicable laws and rules and policies of Nasdaq to enable the delisting by the
Surviving Corporation of the Shares from Nasdaq and the deregistration of the Shares under the Exchange Act as promptly as practicable
after the Effective Time.
6.12 Convertible
Notes. Within the time periods required by the terms of the Convertible Notes Indentures, the
Company shall take all actions required by the Convertible Notes Indentures to be performed by the Company at or prior to the Effective
Time as a result of the execution and delivery of this Agreement or the consummation of the Transactions, including the giving of any
notices that may be required and the delivery to the trustee, noteholders or other applicable Persons, as applicable, of any documents
or instruments required to be delivered at or prior to the Effective Time to such trustee, noteholders or other applicable Persons. In
addition, the Company shall, at Parent’s request prior to the Effective Time, provide all notices and take all other actions to
comply with all of the terms and conditions of the Convertible Notes Indentures. The Company shall provide Parent and its legal counsel
(i) copies of any notices or other communications, promptly after the receipt thereof, from the trustees or holders of notes under
the Convertible Notes Indentures, (ii) prompt updates on any Convertible Notes surrendered for conversion or repurchased by the
Company prior to the Effective Time, and (iii) with reasonable opportunity (and in any event at least two (2) business days
to the extent reasonably practicable) to review and comment on any notices, documents or instruments prior to such notices, documents
or instruments being provided to the trustee, noteholders or other applicable Persons, and Parent shall cooperate with the Company with
respect to any such notices, documents or instruments that are required to be executed or delivered on or after the Effective Time; provided,
that any notices or actions that are not required by the terms of the applicable Convertible Notes Indenture and any time periods for
conversion or repurchase that are determined by the Company pursuant to the applicable Convertible Notes Indenture, respectively, shall
be subject to the prior approval of Parent (such approval not to be unreasonably withheld, conditioned or delayed).
6.13 UK
Tax Ruling. The Company shall (and shall cause its Subsidiaries to) cooperate in good faith
and use commercially reasonable efforts to assist Parent to obtain a tax ruling from HM Revenue & Customs regarding the time
that a “change of control” occurs for UK Tax purposes with respect to the transactions contemplated by this Agreement (including
a ruling substantially to the effect that such change of control does not occur prior to the Offer Acceptance Time).
Section 7
CONDITIONS
PRECEDENT TO THE MERGER
The obligations of the Parties to effect the Merger
are subject to the satisfaction or waiver (where permissible pursuant to applicable Legal Requirements) as of the Closing of each of
the following conditions:
7.1 No
Restraints. No Governmental Body of competent jurisdiction in a jurisdiction where either Parent
and its Affiliates or the Acquired Corporations operate their respective businesses or own any material assets (a “Specified
Governmental Body”) shall have issued, promulgated, enacted or deemed applicable to the Merger or the Offer any order, injunction,
decree, ruling or other Legal Requirement (whether temporary, preliminary or permanent) or taken any other action, in each case restraining,
enjoining, making illegal or otherwise prohibiting the consummation of the Merger or the Offer, which order, injunction, decree, ruling,
or other Legal Requirement or action remains in effect.
7.2 Consummation
of Offer. Purchaser (or Parent on Purchaser’s behalf) shall have accepted for payment
all of the Shares validly tendered pursuant to the Offer and not validly withdrawn.
Section 8
TERMINATION
8.1 Termination.
This Agreement may be terminated prior to the Effective Time:
(a) by
mutual written consent of Parent and the Company at any time prior to the Offer Acceptance Time;
(b) by
either Parent or the Company, at any time prior to the Offer Acceptance Time, if the Closing shall not have occurred on or prior to midnight
Eastern Time, on January 26, 2024 (the “End Date”); provided, however, that in the case of this Section 8.1(b),
(x) if on the End Date all of the conditions set forth in Annex I, other than clause (f) or (g) set
forth in Annex I, shall have been satisfied or waived by Parent or Purchaser, to the extent waivable by Parent or Purchaser
(other than conditions that by their nature are to be satisfied at the Offer Acceptance Time, each of which is then capable of being
satisfied), then the End Date shall automatically be extended by a period of 90 days (and all references to the End Date herein and in
Annex I shall be as so extended); and (y) the right to terminate this Agreement pursuant to this Section 8.1(b) shall
not be available to any Party whose material breach of this Agreement has caused or resulted in the Offer not being consummated by such
date;
(c) by
either Parent or the Company if a Specified Governmental Body shall have issued, promulgated, enacted or deemed applicable to the Merger
or the Offer any order, injunction, decree, ruling or other Legal Requirement or taken any other action, in each case permanently restraining,
enjoining, making illegal or otherwise prohibiting the consummation of the Merger or the Offer, which order, injunction, decree, ruling
or other Legal Requirement or action shall be final and nonappealable; provided, however, that the right to terminate this Agreement
pursuant to this Section 8.1(c) shall not be available to any Party whose material breach of this Agreement has caused
or resulted in the issuance of such final and nonappealable order, injunction, decree, ruling or other Legal Requirement or action or
to any Party that has failed to use its reasonable best efforts as required by Section 6.2 to remove such order, injunction,
decree, ruling or other Legal Requirement or action;
(d) by
Parent at any time prior to the Offer Acceptance Time, if the Company or the Board of Directors shall have effected a Company Adverse
Recommendation Change;
(e) by
the Company, at any time prior to the Offer Acceptance Time, if (i) the Acquired Corporations and their Representatives shall have
complied in all material respects with the provisions of Section 5.3 and Section 6.1 with respect to the Superior
Offer described in the next clause (ii), (ii) the Company, substantially concurrently with such termination, enters into
a binding written definitive acquisition agreement providing for the consummation of a transaction which the Board of Directors shall
have determined, in good faith, constitutes a Superior Offer (a “Specified Agreement”), and (iii) the Company
pays to Parent the Termination Fee and otherwise complies with Section 8.3(b);
(f) by
Parent at any time prior to the Offer Acceptance Time, if a breach of any representation or warranty contained in this Agreement or failure
to perform any covenant or obligation in this Agreement on the part of the Company shall have occurred such that a condition set forth
in clause (b) or (c) of Annex I would not be satisfied and such breach or failure to perform either cannot
be cured by the Company by the End Date, or if capable of being cured in such time period, shall not have been cured within thirty (30)
days of the date Parent gives the Company written notice of such breach or failure to perform; provided, however, that Parent
shall not have the right to terminate this Agreement pursuant to this Section 8.1(f) if either Parent or Purchaser is
then in material breach of any representation, warranty, covenant or obligation hereunder which breach would permit the Company to terminate
this Agreement pursuant to Section 8.1(g);
(g) by
the Company at any time prior to the Offer Acceptance Time, if a breach of any representation or warranty contained in this Agreement
or failure to perform any covenant or obligation in this Agreement on the part of Parent or Purchaser shall have occurred, in each case,
if such breach or failure would reasonably be expected to prevent Parent or Purchaser from consummating the Transactions and such breach
or failure cannot be cured by Parent or Purchaser, as applicable, by the End Date, or, if capable of being cured in such time period,
shall not have been cured within thirty (30) days of the date the Company gives Parent written notice of such breach or failure to perform;
provided, however, that the Company shall not have the right to terminate this Agreement pursuant to this Section 8.1(g) if
the Company is then in material breach of any representation, warranty, covenant or obligation hereunder which breach would give rise
to a failure of a condition set forth in clause (b) or (c) of Annex I (if such condition were tested as
of the date of such breach instead of as of the Offer Acceptance Time); or
(h) by
the Company (i) if following the expiration of the Offer, Purchaser shall have failed to accept for payment all Shares validly tendered
(and not validly withdrawn) pursuant to the Offer in accordance with Section 1.1(h) or (ii) if following the Offer
Acceptance Time, Purchaser shall have failed to purchase all Shares validly tendered (and not validly withdrawn) pursuant to the Offer
in accordance with Section 1.1(h).
8.2 Effect
of Termination. In the event of the termination of this Agreement as provided in Section 8.1,
written notice thereof shall be given to the other Party or Parties, specifying the provision hereof pursuant to which such termination
is made, and this Agreement shall be of no further force or effect and there shall be no liability on the part of Parent, Purchaser or
the Company or any of their respective former, current or future officers, directors, partners, stockholders, managers, members or Affiliates
following any such termination; provided, however, that (a) the final sentence of Section 1.2(b), the final sentence
of Section 5.1, this Section 8.2, Section 8.3 and Section 9 (other than Section 9.5(b))
shall survive the termination of this Agreement and shall remain in full force and effect, (b) the Confidentiality Agreement shall
survive the termination of this Agreement and shall remain in full force and effect in accordance with its terms and (c) the termination
of this Agreement shall not relieve any Party from any liability for fraud or willful breach of this Agreement prior to termination.
For purposes of this Agreement, “willful breach” means a breach that is a consequence of an act or omission undertaken by
the breaching party with the knowledge that the taking of, or failure to take, such act would, or would reasonably be expected to, cause
or constitute a material breach of this Agreement.
8.3 Expenses;
Termination Fees.
(a) Except
as set forth in this Section 8.3, all fees and expenses incurred in connection with this Agreement and the Transactions shall
be paid by the Party incurring such expenses, whether or not the Offer and Merger are consummated.
(b) In
the event that:
(i) this
Agreement is terminated by the Company pursuant to Section 8.1(e);
(ii) this
Agreement is terminated by Parent pursuant to Section 8.1(d); or
(iii) (A) this
Agreement is terminated by Parent or the Company pursuant to Section 8.1(b) (but in the case of a termination by the
Company, only if at such time Parent would not be prohibited from terminating this Agreement pursuant to proviso (y) to Section 8.1(b))
or by Parent pursuant to Section 8.1(f), (B) any Person shall have publicly disclosed a bona fide Acquisition
Proposal after the date hereof and prior to such termination and such Acquisition Proposal has not been publicly withdrawn prior to such
termination and (C) within twelve (12) months of such termination the Company shall have entered into a definitive agreement with
respect to, or consummated, an Acquisition Proposal; provided that for purposes of this clause (C) the references
to “20%” in the definition of “Acquisition Proposal” shall be deemed to be references to “50%”;
then, in any such event under clause (i),
(ii) or (iii) of this Section 8.3(b), the Company shall pay to Parent or its designee the Termination
Fee by wire transfer of same day funds (x) in the case of Section 8.3(b)(i), on the date that the Specified Agreement
is executed (or if the Specified Agreement is executed on a day that is not a business day, the next business day), (y) in the case
of Section 8.3(b)(ii), within three (3) business days after such termination or (z) in the case of Section 8.3(b)(iii),
on the date that the Acquisition Proposal referred to in clause (C) of Section 8.3(b)(iii) is consummated
(or if the consummation occurs on a day that is not a business day, the next business day); it being understood that in no event shall
the Company be required to pay the Termination Fee on more than one occasion. As used herein, “Termination Fee” shall
mean a cash amount equal to $34,000,000. In the event of any termination described in this Section 8.3(b), (1) payment
of the Termination Fee pursuant to this Section 8.3(b) shall be deemed to be liquidated damages for any and all losses
or damages suffered or incurred by Parent, Purchaser, any of their respective Affiliates or any other Person in connection with this
Agreement (and the termination hereof), the Transactions (and the abandonment thereof) or any matter forming the basis for such termination,
and none of Parent, Purchaser or any of their respective Affiliates shall be entitled to bring or maintain any claim, action or proceeding
against the Company or any of its Affiliates arising out of or in connection with this Agreement, any of the Transactions or any matters
forming the basis for such termination; provided that none of the foregoing under this Section 8.3(b) shall relieve
any Company Related Party from any liability for willful breach of this Agreement or fraud; (2) following the Company’s payment
to Parent of the Termination Fee, payment when due and paid from the Company to Parent of the Termination Fee pursuant to this Section 8.3(b) and
any amounts, when due and paid pursuant to Section 8.3(c), shall be the sole and exclusive remedy of Parent, Purchaser or
any of their respective Affiliates against the Acquired Corporations and any of their respective former, current or future officers,
directors, partners, stockholders, managers, members or Affiliates (collectively, “Company Related Parties”) for any
loss suffered as a result of the failure of the Offer or the Merger to be consummated or for a breach or failure to perform hereunder
or otherwise; and (3) upon payment of such amount(s), none of the Company Related Parties shall have any further liability or obligation
relating to or arising out of this Agreement or the Transactions; provided, that none of the foregoing under this Section 8.3(b) shall
relieve any Company Related Party from any liability for willful breach or fraud.
(c) The
Parties acknowledge that the agreements contained in this Section 8.3 are an integral part of the Transactions and that,
without these agreements, the Parties would not enter into this Agreement; accordingly, if the Company fails to timely pay any amount
due pursuant to Section 8.3(b), and, in order to obtain the payment, Parent commences a Legal Proceeding which results in
a judgment against the Company, the Company shall pay Parent its reasonable and documented costs and expenses (including reasonable and
documented attorneys’ fees) in connection with such suit, together with interest on such amount at the prime rate as published
in the Wall Street Journal in effect on the date such payment was required to be made through the date such payment was actually received.
Section 9
MISCELLANEOUS
PROVISIONS
9.1 Amendment.
Prior to the Effective Time, this Agreement may be amended with the approval of the respective Boards of Directors of the Company, Parent
and Purchaser at any time. This Agreement may not be amended except by an instrument in writing signed on behalf of each of the Parties.
9.2 Waiver.
No failure on the part of any Party to exercise any power, right, privilege or remedy under this Agreement, and no delay on the part
of any Party in exercising any power, right, privilege or remedy under this Agreement, shall operate as a waiver of such power, right,
privilege or remedy; and no single or partial exercise of any such power, right, privilege or remedy shall preclude any other or further
exercise thereof or of any other power, right, privilege or remedy. At any time prior to the Effective Time, Parent and Purchaser, on
the one hand, and the Company, on the other hand, may, to the extent permissible under applicable Legal Requirements, (a) extend
the time for the performance of any of the obligations or other acts of the other, (b) waive any breach of the representations and
warranties of the other contained herein or in any document delivered pursuant hereto or (c) waive compliance by the other with
any of the agreements or covenants contained herein. Any such extension or waiver shall be valid only if is expressly set forth in a
written instrument duly executed and delivered on behalf of the Party or Parties to be bound thereby, but such extension or waiver or
failure to insist on strict compliance with an obligation, covenant, agreement or condition shall not operate as a waiver of, or estoppel
with respect to, any subsequent or other failure.
9.3 No
Survival of Representations and Warranties. None of the representations and warranties contained
in this Agreement, the Company Disclosure Schedule or in any certificate or schedule or other document delivered by any Person pursuant
to this Agreement shall survive the Effective Time.
9.4 Entire
Agreement; Counterparts. This Agreement (including its Exhibits, Annexes and the Company Disclosure
Schedule) and the Confidentiality Agreement constitute the entire agreement and supersede all prior agreements and understandings, both
written and oral, among or between any of the Parties and their respective Affiliates, with respect to the subject matter hereof and
thereof. This Agreement may be executed in one or more counterparts, including by facsimile or by email with .pdf attachments, all of
which shall be considered one and the same agreement, and shall become effective when one or more counterparts have been signed by each
of the parties and delivered to the other parties.
9.5 Applicable
Legal Requirements; Jurisdiction; Specific Performance; Remedies.
(a) This
Agreement shall be governed by, and construed in accordance with, the laws of the State of Delaware, regardless of the laws that might
otherwise govern under applicable principles of conflicts of laws thereof. In any action or proceeding arising out of or relating to
this Agreement or any of the Transactions: (i) each of the Parties irrevocably and unconditionally consents and submits to the exclusive
jurisdiction and venue of the Chancery Court of the State of Delaware and any state appellate court therefrom or, if (but only if) such
court lacks subject matter jurisdiction, the United States District Court sitting in New Castle County in the State of Delaware and any
appellate court therefrom (collectively, the “Delaware Courts”); and (ii) each of the Parties irrevocably consents
to service of process by first class certified mail, return receipt requested, postage prepaid, to the address at which such Party is
to receive notice in accordance with Section 9.9. Each of the Parties irrevocably and unconditionally (1) agrees not
to commence any such action or proceeding except in the Delaware Courts, (2) agrees that any claim in respect of any such action
or proceeding may be heard and determined in the Delaware Courts, (3) waives, to the fullest extent it may legally and effectively
do so, any objection that it may now or hereafter have to the jurisdiction or laying of venue of any such action or proceeding in the
Delaware Courts and (4) waives, to the fullest extent permitted by law, the defense of an inconvenient forum to the maintenance
of such action or proceeding in the Delaware Courts. The Parties agree that a final judgment in any such action or proceeding shall be
conclusive and may be enforced in other jurisdictions by suit on the judgment or in any other manner provided by applicable Legal Requirements;
provided, however, that nothing in the foregoing shall restrict any Party’s rights to seek any post-judgment relief regarding,
or any appeal from, such final trial court judgment.
(b) The
Parties agree that irreparable damage for which monetary damages, even if available, would not be an adequate remedy, would occur in
the event that the Parties do not perform their obligations under the provisions of this Agreement in accordance with its specified terms
or otherwise breach such provisions. The Parties acknowledge and agree that (i) the Parties shall be entitled to an injunction or
injunctions, specific performance, or other equitable relief, to prevent breaches of this Agreement and to enforce specifically the terms
and provisions hereof in the courts described in Section 9.5(a) without proof of monetary damages, this being in addition
to any other remedy to which they are entitled under this Agreement, and (ii) the right of specific performance is an integral part
of the Transactions and without that right, neither the Company nor Parent would have entered into this Agreement. Each of the Parties
agrees that it will not oppose the granting of an injunction, specific performance or other equitable relief on the basis that the other
Parties have an adequate remedy at law. The Parties acknowledge and agree that any Party seeking an injunction or injunctions to prevent
breaches of this Agreement and to enforce specifically the terms and provisions of this Agreement in accordance with this Section 9.5(b) shall
not be required to provide any bond or other security in connection with any such order or injunction.
(c) EACH
OF THE PARTIES IRREVOCABLY WAIVES ANY AND ALL RIGHT TO TRIAL BY JURY IN ANY LEGAL PROCEEDING BETWEEN THE PARTIES (WHETHER BASED ON CONTRACT,
TORT OR OTHERWISE), INCLUDING ANY COUNTERCLAIM, ARISING OUT OF OR RELATING TO THIS AGREEMENT OR THE TRANSACTIONS CONTEMPLATED HEREBY
OR THE ACTIONS OF ANY PARTY HERETO IN THE NEGOTIATION, ADMINISTRATION, PERFORMANCE AND ENFORCEMENT THEREOF. EACH PARTY (I) MAKES
THIS WAIVER VOLUNTARILY AND (II) ACKNOWLEDGES THAT SUCH PARTY HAS BEEN INDUCED TO ENTER INTO THIS AGREEMENT BY, AMONG OTHER THINGS,
THE MUTUAL WAIVERS CONTAINED IN THIS SECTION 9.5.
9.6 Assignability.
This Agreement shall be binding upon, and shall be enforceable by and inure solely to the benefit of, the Parties and their respective
successors and permitted assigns; provided, however, that neither this Agreement nor any of the rights hereunder may be assigned
by a Party without the prior written consent of the other Parties, and any attempted assignment of this Agreement or any of such rights
without such consent shall be void and of no effect.
9.7 No
Third Party Beneficiaries. Nothing in this Agreement, express or implied, is intended to or
shall confer upon any Person (other than the Parties) any right, benefit or remedy of any nature whatsoever under or by reason of this
Agreement; except for: (a) if the Offer Acceptance Time occurs, (i) the right of the Company’s stockholders to receive
the Offer Price or Merger Consideration, as applicable, pursuant to Section 1 or Section 2 following the Offer
Acceptance Time or the Effective Time, as applicable, in accordance with the terms of this Agreement, and (ii) the right of the
holders of Company Options, Company PSUs and Company RSUs to receive the Merger Consideration pursuant to Section 2.8 following
the Effective Time in accordance with the terms of this Agreement; (b) the provisions set forth in Section 6.4 of this
Agreement; and (c) the limitations on liability set forth in Section 8.3(b).
9.8 Transfer
Taxes. Except as otherwise provided in Sections 1.1(i) and 2.6(b), all transfer,
documentary, sales, use, stamp, registration and other similar Taxes and fees with respect to the transfer of Shares pursuant to the
Offer or the Merger shall be borne by the Company and expressly shall not be a liability of holders of Shares.
9.9 Notices.
Any notice or other communication required or permitted to be delivered to any Party under this Agreement shall be in writing and shall
be deemed properly delivered, given and received (a) upon receipt when delivered by hand, (b) two (2) business days after
being sent by registered mail or by courier or express delivery service, (c) if sent by email prior to 6:00 p.m. recipient’s
local time, upon transmission when receipt is confirmed, or (d) if sent by email after 6:00 p.m. recipient’s local time
and receipt is confirmed, the business day following the date of transmission; provided that in each case the notice or other
communication is sent to the physical address or email address set forth beneath the name of such Party below (or to such other physical
address or email address as such Party shall have specified in a written notice given to the other Parties):
if to Parent or Purchaser (or following the Effective Time,
the Surviving Corporation):
| |
Alfasigma
S.p.A. |
| |
Via
Regazzi del ’99, 5 |
| |
40133
Bologna (BO), Italy |
| |
Attention: |
Corporate General Counsel |
| |
|
| |
Email: |
michele.cera@alfasigma.com |
with a copy (which shall not constitute notice) to:
| |
Sullivan &
Cromwell LLP |
| |
125
Broad Street |
| |
New
York NY 10004 |
| |
Attention: |
Matthew
G. Hurd; |
| |
|
Oderisio
de Vito Piscicelli; |
| |
|
Luke
Carsley |
| |
|
| |
Email: |
hurdm@sullcrom.com; |
| |
|
devitopiscicellio@sullcrom.com; |
| |
|
carsleyl@sullcrom.com |
| |
|
if to the Company (prior to the Effective Time):
| |
Intercept
Pharmaceuticals, Inc. |
| |
305
Madison Avenue |
| |
Morristown,
NJ 07960 |
| |
Attention:
General Counsel |
| |
|
| |
Email:
jared.freedberg@interceptpharma.com |
| |
|
with a copy (which shall not constitute notice) to:
| |
Skadden,
Arps, Slate, Meagher & Flom LLP |
| |
500
Boylston Street, 23rd Floor |
| |
Boston,
MA 02116 |
| |
Attention: |
Graham Robinson; |
| |
|
Laura Knoll |
| |
|
| |
Email: |
graham.robinson@skadden.com; |
| |
|
laura.knoll@skadden.com |
| |
|
9.10 Severability.
Any term or provision of this Agreement that is invalid or unenforceable in any situation in any jurisdiction shall not affect the validity
or enforceability of the remaining terms and provisions of this Agreement or the validity or enforceability of the offending term or
provision in any other situation or in any other jurisdiction. If a final judgment of a court of competent jurisdiction declares that
any term or provision of this Agreement is invalid or unenforceable, the Parties agree that the court making such determination shall
have the power to limit such term or provision, to delete specific words or phrases or to replace such term or provision with a term
or provision that is valid and enforceable and that comes closest to expressing the intention of the invalid or unenforceable term or
provision, and this Agreement shall be valid and enforceable as so modified. In the event such court does not exercise the power granted
to it in the prior sentence, the Parties agree to replace such invalid or unenforceable term or provision with a valid and enforceable
term or provision that will achieve, to the extent possible, the economic, business and other purposes of such invalid or unenforceable
term or provision.
9.11 Obligation
of Parent. Parent shall ensure that Purchaser (and, following the Effective Time, the Surviving
Corporation) duly performs, satisfies and discharges on a timely basis each of the covenants, obligations and liabilities applicable
to Purchaser or the Surviving Corporation, as applicable, under this Agreement, and Parent shall be jointly and severally liable with
Purchaser or the Surviving Corporation, as applicable, for the due and timely performance and satisfaction of each of said covenants,
obligations and liabilities.
9.12 Construction.
(a) For
purposes of this Agreement, whenever the context requires: the singular number shall include the plural, and vice versa; the masculine
gender shall include the feminine and neuter genders; the feminine gender shall include the masculine and neuter genders; and the neuter
gender shall include the masculine and feminine genders.
(b) The
Parties agree that any rule of construction to the effect that ambiguities are to be resolved against the drafting Party shall not
be applied in the construction or interpretation of this Agreement.
(c) As
used in this Agreement, the word “including” and words of similar import shall mean including without limiting the generality
of any description preceding such term, unless otherwise specified.
(d) Except
as otherwise indicated, all references in this Agreement to “Sections,” “Exhibits” or “Annexes” are
intended to refer to Sections of this Agreement and Exhibits or Annexes to this Agreement.
(e) The
bold-faced headings contained in this Agreement are for convenience of reference only, shall not be deemed to be a part of this Agreement
and shall not be referred to in connection with the construction or interpretation of this Agreement.
(f) The
term “dollars” and character “$” shall mean United States dollars.
(g) Any
reference to (i) any Contract (including this Agreement) are to the Contract as amended, modified, supplemented, restated, or replaced
from time to time (in the case of any Contract, to the extent permitted by the terms thereof and, if applicable, the terms of this Agreement);
(ii) any Governmental Body includes any successor to that Governmental Body; and (iii) any applicable Legal Requirement refers
to such applicable Legal Requirement as amended, modified, supplemented, or replaced from time to time (and, in the case of statutes,
include any rule and regulation promulgated under such statute) and references to any section of any applicable Legal Requirement
includes any successor to such section (provided that, for purposes of any representation and warranty in this Agreement that
is made as of a specific date, references to any Legal Requirement or Contract shall be deemed to refer to such Legal Requirement or
Contract, as amended, and to any rule or regulation promulgated thereunder, in each case, as of such date).
[Signature page follows]
IN
WITNESS WHEREOF, the Parties have caused this Agreement to be executed as of the date first above written.
| |
Intercept Pharmaceuticals, Inc. |
| |
|
| |
By: |
/s/ Jerome Durso |
| |
|
Name: |
Jerome Durso |
| |
|
Title: |
President and Chief Executive Officer |
[Signature Page to Agreement and Plan
of Merger]
IN
WITNESS WHEREOF, the Parties have caused this Agreement to be executed as of the date first above written.
| |
Alfasigma S.p.A. |
| |
|
| |
By: |
/s/ Francesco Balestrieri |
| |
|
Name: |
Francesco Balestrieri |
| |
|
Title: |
CEO |
[Signature Page to Agreement and Plan
of Merger]
IN
WITNESS WHEREOF, the Parties have caused this Agreement to be executed as of the date first above written.
| | Interstellar
Acquisition Inc. |
| |
By: |
/s/ Francesco Balestrieri |
| |
|
Name: |
Francesco Balestrieri |
| |
|
Title: |
President |
[Signature Page to Agreement and Plan
of Merger]
EXHIBIT A
CERTAIN DEFINITIONS
For purposes of the Agreement (including this
Exhibit A and Annex I):
Acceptable
Confidentiality Agreement. “Acceptable Confidentiality Agreement” is defined in Section 5.3(a) of
the Agreement.
Acquired
Corporations. “Acquired Corporations” is defined in Section 3.1(a) of the Agreement.
Acquisition
Proposal. “Acquisition Proposal” shall mean any proposal, indication of interest or offer from any Person
(other than Parent and its Affiliates) or “group”, within the meaning of Section 13(d) of the Exchange Act, relating
to, in a single transaction or series of related transactions, any (A) direct or indirect acquisition or license of assets of the
Company equal to 20% or more of the Company’s consolidated assets or to which 20% or more of the Company’s revenues or earnings
on a consolidated basis are attributable, (B) issuance or acquisition of equity securities of the Company (or instruments convertible
into or exercisable or exchangeable for, such equity securities) representing 20% or more of the outstanding voting power of the Company
Common Stock, (C) recapitalization, tender offer or exchange offer that if consummated would result in any Person or group beneficially
owning equity securities of the Company (or instruments convertible into or exercisable or exchangeable for such equity securities) representing
20% or more of the outstanding voting power of the Company Common Stock or (D) merger, consolidation, amalgamation, share exchange,
business combination, recapitalization, liquidation, dissolution or similar transaction involving the Company that if consummated would
result in any Person or group beneficially owning 20% or more of the outstanding Company Common Stock, in each case other than the Transactions.
Affiliate.
“Affiliate” shall mean, as to any Person, any other Person that, directly or indirectly, controls, or is controlled
by, or is under common control with, such Person. For this purpose, “control” (including, with its correlative meanings,
“controlled by” and “under common control with”) shall mean the possession, directly or indirectly, of the power
to direct or cause the direction of management or policies of a Person, whether through the ownership of securities or partnership or
other ownership interests, by Contract or otherwise.
Agreement.
“Agreement” is defined in the preamble to the Agreement.
Anti-Corruption
Laws. “Anti-Corruption Laws” shall mean the Foreign Corrupt Practices Act of 1977, as amended, the Anti-Kickback
Act of 1986, as amended, the UK Bribery Act of 2010, and the Anti-Bribery Laws of the People’s Republic of China or any applicable
Legal Requirements of similar effect, and the related regulations and published interpretations thereunder.
Antitrust
Laws. “Antitrust Laws” shall mean the Sherman Act, as amended, the Clayton Act, as amended, the HSR Act,
the Federal Trade Commission Act, as amended, all applicable foreign antitrust laws and all other applicable Legal Requirements issued
by a Governmental Body that are designed or intended to prohibit, restrict or regulate actions having the purpose or effect of monopolization
or restraint of trade or lessening of competition.
Applicable
Date. “Applicable Date” is defined in Section 3.4(a) of the Agreement.
Barclays
Fairness Opinion. “Barclays Fairness Opinion” is defined in Section 3.24 of the Agreement.
Board
of Directors. “Board of Directors” is defined in the Introduction to the Agreement.
Book-Entry
Shares. “Book-Entry Shares” shall mean non-certificated Shares represented by book-entry.
business
day. “business day” shall mean a day except a Saturday, a Sunday or other day on which banks in the City
of New York are authorized or required by Legal Requirements to be closed.
Capitalization
Date. “Capitalization Date” is defined in Section 3.3(a) of the Agreement.
CARES
Act. “CARES Act” is defined in the definition of “CARES Act and COVID Relief Programs”.
CARES
Act and COVID Relief Programs. “CARES Act and COVID Relief Programs” shall mean, collectively, the Coronavirus
Aid, Relief, and Economic Security Act (H.R. 748, Pub Law 116-136) (the “CARES Act”), the Families First Coronavirus
Response Act (H.R.6201, Pub Law 116-127), Paycheck Protection Program Flexibility Act of 2020 (H.R. 7010, Pub Law 116-142), American
Rescue Plan Act (H.R. 1319, Pub Law 117-7) and all FAQs or interim final rules (to the extent having the force of a Legal Requirement)
issued by any Governmental Body related thereto, including any programs or facilities established by the Board of Governors of the Federal
Reserve System to which the U.S. Treasury Department has provided financing as contemplated by Title IV of the CARES Act, and any analogous
U.S. state, local, or non-U.S. law.
Centerview
Fairness Opinion. “Centerview Fairness Opinion” is defined in Section 3.24 of the Agreement.
Certificated
Shares. “Certificated Shares” is defined in Section 2.6(b) of the Agreement.
Certificates.
“Certificates” is defined in Section 2.6(b) of the Agreement.
Change
of Control Payment. “Change of Control Payment” is defined in Section 3.9(a)(viii) of
the Agreement.
Clinical
Trials. “Clinical Trials” is defined in Section 3.12(c) of the Agreement.
Closing.
“Closing” is defined in Section 2.3(a) of the Agreement.
Closing
Date. “Closing Date” is defined in Section 2.3(a) of the Agreement.
Code.
“Code” shall mean the Internal Revenue Code of 1986, as amended.
Collaboration
Partner. “Collaboration Partner” is defined in Section 3.12(a) of the Agreement.
Company.
“Company” is defined in the preamble to the Agreement.
Company
401(k) Plan. “Company 401(k) Plan” is defined in Section 6.3(d) of the
Agreement.
Company
Adverse Recommendation Change. “Company Adverse Recommendation Change” is defined in Section 6.1(a) of
the Agreement.
Company
Associate. “Company Associate” shall mean each officer or other employee, or individual who is an independent
contractor, consultant or director, of or to the Company or its Subsidiaries.
Company
Board Recommendation. “Company Board Recommendation” is defined in the Introduction to the Agreement.
Company
Common Stock. “Company Common Stock” shall mean the common stock, $0.001 par value per share, of the Company.
Company
Disclosure Documents. “Company Disclosure Documents” is defined in Section 3.4(g) of the
Agreement.
Company
Disclosure Schedule. “Company Disclosure Schedule” shall mean the disclosure schedule that has been prepared
by the Company in accordance with the requirements of the Agreement and that has been delivered by the Company to Parent on the date
of the Agreement.
Company
Equity Awards. “Company Equity Awards” shall mean the Company Options, Company PSUs and Company RSUs.
Company
Equity Awards Schedule. “Company Equity Awards Schedule” is defined in Section 3.3(h) of
the Agreement.
Company
Equity Plans. “Company Equity Plans” shall mean the Company’s Amended and Restated Equity Incentive
Plan and its 2023 Equity Incentive Plan, each as amended.
Company
IP. “Company IP” shall mean (a) all Intellectual Property Rights that are owned or purported to be
owned by the Acquired Corporations and (b) all third party Intellectual Property Rights exclusively licensed or purported to be
exclusively licensed to any of the Acquired Corporations.
Company
Options. “Company Options” shall mean all compensatory options to purchase Shares granted pursuant to the
Company Equity Plans.
Company
Preferred Stock. “Company Preferred Stock” shall mean the preferred stock, $0.001 par value per share,
of the Company.
Company
Related Parties. “Company Related Parties” is defined in Section 8.3(b) of the Agreement.
Company
Related Person. “Company Related Person” shall mean any Acquired Corporation’s former and current
stockholders, directors, officers, partners, members, managers, general or limited partners, other equityholders and controlling Persons.
Company
Returns. “Company Returns” is defined in Section 3.15(a) of the Agreement.
Company
PSUs. “Company PSUs” shall mean restricted stock units with respect to Shares subject to performance vesting
conditions granted pursuant to the Company Equity Plans.
Company
RSUs. “Company RSUs” shall mean restricted stock units with respect to Shares granted pursuant to the Company
Equity Plans, other than Company PSUs.
Company
RSU Consideration. “Company RSU Consideration” is defined in Section 2.8(d) of the Agreement.
Company
SEC Documents. “Company SEC Documents” is defined in Section 3.4(a) of the Agreement.
Company
Security Policies. “Company Security Policies” shall mean contractual obligations, binding commitments,
adopted policies and public statements of the Acquired Corporations with respect to privacy or data security, including the use of and
access to Personal Information.
Confidentiality
Agreement. “Confidentiality Agreement” is defined in Section 5.1 of the Agreement.
Consent.
“Consent” shall mean any approval, consent, ratification, permission, waiver or authorization.
Continuation
Period. “Continuation Period” is defined in Section 6.3 of the Agreement.
Continuing
Employee. “Continuing Employee” is defined in Section 6.3 of the Agreement.
Contract.
“Contract” shall mean any legally binding agreement.
Convertible
Notes. “Convertible Notes” shall mean the Company’s 2.00% Convertible Notes due May 15, 2026
and 3.50% Convertible Secured Notes due February 15, 2026.
Convertible
Notes Indentures. “Convertible Notes Indentures” shall mean (i) the Base Indenture, dated as of July 6,
2016, between the Company and U.S. Bank National Association (“U.S. Bank”), as trustee, as supplemented by the Second
Supplemental Indenture, dated May 14, 2019, between the Company and U.S. Bank, as trustee, with respect to the Company’s 2.00%
Convertible Notes due May 15, 2026 and (ii) the Base Indenture, dated as of August 17, 2021, between the Company and U.S.
Bank, as trustee, as supplemented by the First Supplemental Indenture, dated August 17, 2021, between the Company and U.S. Bank,
as trustee, with respect to the Company’s 3.50% Convertible Secured Notes due February 15, 2026.
Copyrights.
“Copyrights” is defined in the definition of Intellectual Property Rights.
CRO.
“CRO” shall mean a Person (including a commercial, academic, or other organization) contracted by a sponsor to perform
one or more of a sponsor’s Clinical Trial-related duties and functions, including those defined in (a) 21 C.F.R. Part 312.3(b);
(b) ICH GCP E6; and (c) foreign equivalents of the foregoing, each as may be amended from time to time.
DAC
6. “DAC 6” is defined in Section 3.15(f) of the Agreement.
Delaware
Courts. “Delaware Courts” is defined in Section 9.5(a) of the Agreement.
Depository
Agent. “Depository Agent” is defined in Section 2.6(a) of the Agreement.
Determination
Notice. “Determination Notice” is defined in Section 6.1(b)(i) of the Agreement.
DGCL.
“DGCL” shall mean the Delaware General Corporation Law, as amended.
Dissenting
Shares. “Dissenting Shares” is defined in Section 2.7 of the Agreement.
DOJ.
“DOJ” shall mean the U.S. Department of Justice.
Effective
Time. “Effective Time” is defined in Section 2.3(b) of the Agreement.
Employee
Plan. “Employee Plan” shall mean any (a) bonus, deferred compensation, incentive compensation, stock
purchase, stock option, other equity-based plan, severance pay, termination pay, death and disability benefits, hospitalization, medical,
life or other insurance, flexible benefits, supplemental unemployment benefits, profit-sharing, pension or retirement plan, policy, program,
agreement or arrangement, and (b) employment, consulting, severance or similar agreement, and each other employee benefit plan,
or arrangement (in each case whether or not funded and whether or not in writing), in each case that is (i) sponsored, maintained,
contributed to or required to be contributed to by the Company or its Affiliates for the benefit of any current or former employee of
the Company or its Subsidiaries, (ii) with respect to which the Company or any of its Subsidiaries has any liability or (iii) to
which the Company or any of its Subsidiaries is a party.
Encumbrance.
“Encumbrance” shall mean any lien, pledge, hypothecation, mortgage, security interest, encumbrance, right of first
refusal, preemptive right or similar restriction of any nature.
End
Date. “End Date” is defined in Section 8.1(b) of the Agreement.
Entity.
“Entity” shall mean any corporation (including any non-profit corporation), general partnership, limited partnership,
limited liability partnership, joint venture, estate, trust, company (including any company limited by shares, limited liability company
or joint stock company), firm, society or other enterprise, association, organization or entity.
Environmental
Law. “Environmental Law” shall mean any federal, state, local or foreign Legal Requirement relating to
pollution or protection of human health, worker health or the environment (including ambient air, surface water, ground water, land surface
or subsurface strata), including any law or regulation relating to emissions, discharges, Releases or threatened Releases of Hazardous
Materials, or otherwise relating to the manufacture, processing, distribution, use, treatment, storage, disposal, transport or handling
of Hazardous Materials.
ERISA.
“ERISA” shall mean the Employee Retirement Income Security Act of 1974, as amended.
Exchange
Act. “Exchange Act” shall mean the Securities Exchange Act of 1934, as amended, and the rules and
regulations promulgated thereunder.
Excluded
Shares. “Excluded Shares” is defined in Section 1.1(a) of the Agreement.
Expiration
Date. “Expiration Date” is defined in Section 1.1(c) of the Agreement.
Extension
Deadline. “Extension Deadline” is defined in Section 1.1(c) of the Agreement.
FDA.
“FDA” shall mean the United States Food and Drug Administration.
FDCA.
“FDCA” shall mean the Federal Food, Drug and Cosmetic Act, as amended (21 U.S.C. § 301 et seq.), and all binding
rules, regulations and guidelines promulgated thereunder.
FTC.
“FTC” shall mean the U.S. Federal Trade Commission.
GAAP.
“GAAP” is defined in Section 3.4(b) of the Agreement.
Global
Trade Laws. “Global Trade Laws” shall mean all applicable Legal Requirements governing the export, import
and provision of goods (including technical data and technology) and services in the jurisdictions in which the Acquired Corporations
operate, including (a) the applicable Legal Requirements of the United States governing embargoes, sanctions, and boycotts, under
the International Emergency Economic Powers Act (50 U.S.C. § 1701 et seq.), Trading with the Enemy Act (50 U.S.C. App. §§
1-44), and all rules, regulations and executive orders relating to any of the foregoing, including regulations promulgated by the Office
of Foreign Assets Control of the United States Department of the Treasury at 15 C.F.R. Parts 500-599 and by the U.S. Department of State;
(b) all Legal Requirements governing the export, re-export, or transfer of goods, software, technology, or technical data, including
the Export Administration Act of 1979 (50 U.S.C. App. §§ 2401-2420), the Export Control Reform Act of 2018 (Pub. L. 115-232),
the Export Administration Regulations (15 C.F.R. Parts 730-774), the Arms Export Control Act (22 U.S.C. § 2778), and the International
Traffic in Arms Regulations (22 C.F.R. § 120.1 et seq.); (c) the Foreign Trade Regulations (15 C.F.R. Part 30) administered
by the Census Bureau; (d) all applicable Legal Requirements governing the importation of products, technology, technical data, and
services, including those administered by United States Customs and Border Protection (19 C.F.R. Parts 1-199); (e) the antiboycott
laws set forth in section 999 of the Internal Revenue Code, the Department of Treasury Guidelines concerning international boycotts promulgated
thereunder, and Part 760 of the EAR; and (f) any other applicable Legal Requirements relating to the export and import activities
of the Acquired Corporations.
Good
Clinical Practices. “Good Clinical Practices” shall mean FDA’s standards for the design, conduct,
performance, monitoring, auditing, recording, analysis, and reporting of clinical trials contained in 21 C.F.R. Parts 50, 54, 56 and
312.
Governmental
Authorization. “Governmental Authorization” shall mean any: permit, license, certificate, franchise, permission,
clearance, registration, qualification or authorization issued, granted, given or otherwise made available by or under the authority
of any Governmental Body or pursuant to any Legal Requirement.
Governmental
Body. “Governmental Body” shall mean any: (a) nation, state, commonwealth, province, territory, county,
municipality, district or other jurisdiction of any nature; (b) federal, state, local, municipal, foreign or other government; or
(c) governmental or quasi-governmental authority of any nature including any governmental division, department, agency, commission,
instrumentality, official, ministry, fund, foundation, center, organization, unit or body and any court, arbitrator or other tribunal,
including the FDA.
Hazardous
Materials. “Hazardous Materials” shall mean any waste, material, or substance that is listed, regulated
or defined under any Environmental Law and includes any pollutant, chemical substance, hazardous substance, hazardous waste, special
waste, solid waste, asbestos, mold, radioactive material, polychlorinated biphenyls, petroleum or petroleum-derived substance or waste.
Healthcare
Laws. “Healthcare Laws” shall mean (a) the FDCA; (b) the federal Medicare and Medicaid statutes
(Title XVIII and Title XIX of the Social Security Act); (c) the Physician Payments Sunshine Act; (d) the federal Anti-Kickback
Statute (42 U.S.C.A § 1320a7b(b)), Stark Law (42 U.S.C.A § 1395nn), False Claims Act (31 U.S.C.A § 3729 et seq.), Civil
Monetary Penalties Law (42 U.S.C. §§ 1320a-7a and 1320a-7b); (e) state pharmaceutical licensing, disclosure and reporting
regulatory requirements; (f) the Health Insurance Portability and Accountability Act of 1996, as amended by the Health Information
and Technology for Economic and Clinical Health Act; (g) regulations promulgated pursuant to any of the statutes in clauses (a) through
(f); and (h) any comparable foreign laws.
Healthcare
Regulatory Authority. “Healthcare Regulatory Authority” is defined in Section 3.12(a) of
the Agreement.
HSR
Act. “HSR Act” shall mean the Hart-Scott-Rodino Antitrust Improvements Act of 1976, as amended.
Indebtedness.
“Indebtedness” shall mean, without duplication, (i) any indebtedness for borrowed money (including the issuance
of any debt security) to any Person, including that portion of obligations with respect to any capital leases that is classified as a
liability on a balance sheet in conformity with GAAP and any synthetic lease obligation, (ii) any obligations evidenced by notes,
bonds, debentures or similar Contracts for indebtedness for borrowed money owing to any Person other than an Acquired Corporation, (iii) any
reimbursement or other payment obligations in respect of letters of credit and bankers’ acceptances (other than obligations in
respect of letters of credit and bankers’ acceptances used in the ordinary course of business as security for leases), bank guarantees,
surety bonds and similar instruments, regardless of whether drawn upon, including the principal, interest and fees owning thereon, (iv) all
indebtedness created or arising under any conditional sale or other title retention agreement with respect to property acquired, (v) any
net obligations under swaps, options, derivatives and other hedging agreements or arrangements that would be payable upon termination
thereof (assuming they were terminated on the date of determination) or (vi) any guaranty of any such obligations described in clauses
(i) through (v) of any Person other than an Acquired Corporation (other than, in any case, accounts payable to trade
creditors and accrued expenses, in each case, arising in the ordinary course of business consistent with past practice).
Indemnified
Persons. “Indemnified Persons” is defined in Section 6.4(a) of the Agreement.
Initial
Expiration Date. “Initial Expiration Date” is defined in Section 1.1(c) of the Agreement.
Intellectual
Property Rights. “Intellectual Property Rights” shall mean all intellectual property and industrial property
rights of every kind and description throughout the world, including all U.S. and foreign (i) patents, patent applications, provisional
applications, statutory invention registrations, inventions, discoveries and invention disclosures (whether or not patented), and all
related continuations, continuations-in-part, divisionals, reissues, re-examinations, substitutions, and extensions thereof (“Patents”),
(ii) trademarks, service marks, names, corporate names, trade names, brand names, Internet domain names, logos, slogans, symbols,
URLs, trade dress, design rights, and other similar designations of source or origin, in each case whether or not registered, together
with all common law rights and goodwill associated therewith and symbolized by any of the foregoing and all applications and registrations
for the foregoing (“Trademarks”), (iii) copyrights, copyrightable subject matter, and all registrations and applications
for registrations thereof, published and unpublished works of authorship (whether or not copyrightable), together with all common law
rights and moral rights therein, including extensions, renewals, restorations, reversions, derivatives, and combinations of the foregoing
(“Copyrights”), (iv) trade secrets and other confidential or proprietary information, including ideas, know-how,
inventions, proprietary processes, formulae, models, algorithms, data, testing procedures and testing results, clinical and technical
data, and methodologies (“Trade Secrets”) and (v) other intellectual property rights and/or proprietary or industrial
rights.
Intervening
Event. “Intervening Event” shall mean any change, event or development (other than to the extent relating
to Parent or the Purchaser, or the announcement or pendency of any of the Transactions) that, individually or in the aggregate, is material
to the Acquired Corporations, taken as a whole, and (a) is not known or reasonably foreseeable to or by the Board of Directors as
of the date of the Agreement, in each case, based on facts known to the Board of Directors as of the date of the Agreement, which change,
event or development becomes known to or by the Board of Directors prior to the Offer Acceptance Time and did not arise from the operation
of the Acquired Corporations in the ordinary course of business, and (b) does not relate to any transaction of the type described
by clauses (A) through (D) of the definition of Acquisition Proposal, without giving effect to any 20% thresholds set forth
therein; provided, however, that in no event shall any of the following constitute an Intervening Event: (i) any action taken
by the Parties pursuant to or in compliance with the Agreement, (ii) any changes in Legal Requirements or the commencement, continuance
or settlement of any Legal Proceeding, (iii) changes in the market price or trading volume of the Shares in and of themselves (provided
that the underlying reasons for such changes may constitute an Intervening Event), (iv) the fact, in and of itself, that the
Acquired Corporations meet, exceed or fail to meet internal or published projections, forecasts or revenue or earnings predictions for
any period (provided that the underlying reasons for such fact may constitute an Intervening Event), or (v) any event, occurrence,
circumstance, change or effect generally affecting the industries in which the Acquired Corporations operate or the economy or other
general business, financial or market conditions generally.
In
the Money Option. “In the Money Option” is defined in Section 2.8(a) of the Agreement.
IRB.
“IRB” shall mean any independent body (a review board or a committee, institutional, regional, national, or supranational),
constituted of medical professionals and non-medical members, whose responsibility it is to ensure the protection of the rights, safety
and well-being of human subjects involved in a trial and to provide public assurance of that protection, by, among other things, reviewing
and approving / providing favorable opinion on, a trial protocol, the suitability of investigator(s), facilities, and the methods and
material to be used in obtaining and documenting informed consent of the trial subjects and includes any such entity as described in
21 C.F.R. Part 56 or foreign equivalent of the foregoing.
IRS.
“IRS” shall mean the U.S. Internal Revenue Service.
IT
Systems. “IT Systems” shall mean computers, software, servers, workstations, routers, hubs, switches, data
communications lines, and all other information technology equipment, and all associated documentation, in each case, used or held for
use by the Acquired Corporations.
knowledge.
“knowledge” with respect to an Entity shall mean with respect to any matter in question the actual knowledge, after
reasonable inquiry, of such Entity’s executive officers; provided that the foregoing shall not require any freedom to operate
or other analysis with respect to validity or non-infringement of Intellectual Property Rights.
Leased
Real Property. “Leased Real Property” is defined in Section 3.7(b) of the Agreement.
Legal
Proceeding. “Legal Proceeding” shall mean any action, suit, complaint, litigation, arbitration, proceeding
(including any civil, criminal, administrative, investigative or appellate proceeding), hearing or investigation commenced, brought,
conducted or heard by or before any Governmental Body.
Legal
Requirement. “Legal Requirement” shall mean any federal, state, local, municipal, foreign or other law,
statute, constitution, resolution, ordinance, common law, code, edict, decree, rule, regulation, ruling or requirement issued, enacted,
adopted, promulgated, implemented or otherwise put into effect by or under the authority of any Governmental Body (or under the authority
of Nasdaq or another stock exchange).
Material
Adverse Effect. “Material Adverse Effect” shall mean any event, occurrence, circumstance, change or effect
which, individually or when taken together with all other events, occurrences, circumstances, changes or effects which have occurred
in the applicable determination period for a Material Adverse Effect, has had or would reasonably be expected to have a material adverse
effect on (a) the ability of the Company to consummate the Transactions on or before the End Date, or (b) the business, assets,
financial condition or results of operations of the Acquired Corporations, taken as a whole; provided, however, that for purposes
of clause (b) of this definition only, none of the following shall be deemed to constitute or be taken into account in determining
whether there is, or would reasonably be expected to be, a Material Adverse Effect: (i) any change in the market price or trading
volume of the Company’s stock or change in the Company’s credit ratings; provided that the underlying causes of any
such change may be considered in determining whether a Material Adverse Effect has occurred to the extent not otherwise excluded by another
exception herein; (ii) any event, occurrence, circumstance, change or effect directly resulting from the announcement, pendency
or performance of the Transactions (other than for purposes of any representation or warranty contained in Section 3.23 and
the condition set forth in clause (b)(iii) of Annex I solely as such condition relates to Section 3.23);
(iii) any event, occurrence, circumstance, change or effect generally affecting the industries in which the Acquired Corporations
operate or in the economy generally or other general business, financial or market conditions; (iv) any event, occurrence, circumstance,
change or effect arising directly or indirectly from or otherwise relating to fluctuations in the value of any currency or interest rates;
(v) any event, occurrence, circumstance, change or effect arising directly or indirectly from or otherwise relating to any act of
terrorism, war, national or international calamity, natural disaster, acts of god, epidemic, pandemic or any other similar event; (vi) the
failure of the Company to meet internal or analysts’ expectations or projections; provided that the underlying causes of
such failure may be considered in determining whether a Material Adverse Effect has occurred to the extent not otherwise excluded by
another exception herein; (vii) any adverse effect arising from or otherwise relating to any action taken by an Acquired Corporation
at the written direction of Parent or any action specifically required to be taken by an Acquired Corporation under the Agreement, or
the failure of an Acquired Corporation to take any action that such Acquired Corporation is specifically prohibited by the terms of the
Agreement from taking to the extent Parent fails to give its consent thereto after a written request therefor pursuant to Section 5.2;
(viii) any event, occurrence, circumstance, change or effect resulting or arising from the identity of, or any facts or circumstances
relating to, Parent, Purchaser or any of their respective Affiliates; or (ix) any event, occurrence, circumstance, change or effect
arising directly or indirectly from or otherwise relating to any change in, or any compliance with or action taken for the purpose of
complying with any change in, any Legal Requirement or GAAP (or interpretations of any Legal Requirement or GAAP); provided that
any event, occurrence, circumstance, change or effect referred to in the foregoing clauses (iii), (iv), (v) and
(ix) may be taken into account in determining whether there is, or would be reasonably expected to be, a Material Adverse
Effect to the extent such event, occurrence, circumstance, change or effect disproportionately affects the Acquired Corporations relative
to other participants in the industries in which the Acquired Corporations operate.
Material
Contract. “Material Contract” is defined in Section 3.9(a) of the Agreement.
Maximum
Amount. “Maximum Amount” is defined in Section 6.4(b) of the Agreement.
Merger.
“Merger” is defined in the Introduction to the Agreement.
Merger
Consideration. “Merger Consideration” is defined in Section 2.5(a)(iii) of the Agreement.
Minimum
Condition. “Minimum Condition” is defined in Annex I to the Agreement.
Nasdaq.
“Nasdaq” shall mean The Nasdaq Global Select Market.
Offer.
“Offer” is defined in the Introduction to the Agreement.
Offer
Acceptance Time. “Offer Acceptance Time” is defined in Section 1.1(h) of the Agreement.
Offer
Commencement Date. “Offer Commencement Date” shall mean the date on which Purchaser commences the Offer,
within the meaning of Rule 14d-2 under the Exchange Act.
Offer
Conditions. “Offer Conditions” is defined in Section 1.1(b) of the Agreement.
Offer
Documents. “Offer Documents” is defined in Section 1.1(e) of the Agreement.
Offer
Price. “Offer Price” is defined in the Introduction to the Agreement.
Offer
to Purchase. “Offer to Purchase” is defined in Section 1.1(b) of the Agreement.
Parent.
“Parent” is defined in the preamble to the Agreement.
Parent
Material Adverse Effect. “Parent Material Adverse Effect” shall mean any effect, change, event or occurrence
that would or would reasonably be expected to, individually or in the aggregate, materially impair, prevent or materially delay Parent’s
or Purchaser’s ability to consummate the Transactions in a timely manner on the terms set forth herein.
Parties.
“Parties” shall mean Parent, Purchaser, and the Company.
Patents.
“Patents” is defined in the definition of Intellectual Property Rights.
Patient
Assistance Program. “Patient Assistance Program” is defined in Section 5.2(b)(xvii) of the Agreement.
Paying
Agent. “Paying Agent” is defined in Section 2.6(a) of the Agreement.
Payment
Fund. “Payment Fund” is defined in Section 2.6(a) of the Agreement.
Permitted
Encumbrance. “Permitted Encumbrance” shall mean (a) any Encumbrance for Taxes (i) that are not
due and payable or (ii) the validity of which is being contested in good faith by appropriate proceedings and for which provisions
have been recorded to the extent required by GAAP, (b) any Encumbrance representing the rights of customers, suppliers and subcontractors
in the ordinary course of business consistent with past practice under the terms of any Contracts to which the relevant Party is a party
or under general principles of commercial or government contract law (including mechanics’, materialmen’s, carriers’,
workmen’s, warehouseman’s, repairmen’s, landlords’ and similar liens granted or which arise in the ordinary course
of business consistent with past practice), (c) any interest or title of a lessor under leases (other than capital leases) entered
into by the Company or its Subsidiaries in the ordinary course of business and under which there is no default by the Company or its
Subsidiaries, and any Encumbrance related thereto, (d) in the case of any Contract, Encumbrances that are restrictions against the
transfer or assignment thereof that are included in the terms of such Contract, (e) non-exclusive licenses with respect to Intellectual
Property Rights entered into in the ordinary course of business, (f) in the case of real property, Encumbrances that are easements,
rights-of-way, encroachments, restrictions, conditions and other similar Encumbrances incurred or suffered in the ordinary course of
business consistent with past practice and which, individually or in the aggregate, do not and would not materially impair the use (or
contemplated use), utility or value of the applicable real property or otherwise materially impair the present or contemplated business
operations at such location, or zoning, entitlement, building and other land use regulations imposed by Governmental Bodies having jurisdiction
over such real property or that are otherwise set forth on a title report and (g) Encumbrances securing the obligations under the
3.50% Convertible Secured Notes due February 15, 2026 in accordance with the applicable Convertible Notes Indentures.
Person.
“Person” shall mean any individual, Entity or Governmental Body.
Personal
Information. “Personal Information” shall mean any information that (i) alone or in combination with
other information held by the Acquired Corporations, can be used to identify an individual person, household, device or browser, or (ii) is
otherwise protected under applicable Privacy Laws.
Pre-Closing
Period. “Pre-Closing Period” is defined in Section 5.1 of the Agreement.
Privacy
Laws. “Privacy Laws” shall mean any and all applicable Legal Requirement, guidance, decisions, determinations,
order, or associated binding judgments of any Governmental Body, regarding privacy, cybersecurity or the processing or protection of
personal information or personal data, including as applicable, the Health Insurance Portability and Accountability Act of 1996 and the
EU General Data Protection Regulation and any national laws implementing the foregoing, and all applicable breach notification or similar
Legal Requirements.
Process
or Processing. “Process” or “Processing” shall mean any operation or set of operations
performed, whether by manual or automated means, on Personal Information or on sets of Personal Information, including collection, use,
storage, disclosure, transfer, analysis, deletion or modification.
Product
Candidate. “Product Candidate” shall mean each biological, drug, combination product, compound, device
or product candidate being developed, tested, labeled, manufactured, stored or marketed by an Acquired Corporation, or regarding which
an Acquired Corporation has rights.
Purchaser.
“Purchaser” is defined in the preamble to the Agreement.
Recall.
“Recall” is defined in Section 3.12(e) of the Agreement.
Registered
Company IP. “Registered Company IP” is defined in Section 3.8(a) of the Agreement.
Regulatory
Permit. “Regulatory Permit” shall mean any Governmental Authorization required for the development, manufacturing
or marketing of a Product Candidate under applicable Healthcare Laws, including all investigational new drug applications and other authorizations
to conduct Clinical Trials, IRB approvals, new drug applications, supplemental new drug applications, abbreviated new drug applications,
biologic license applications, as defined in 21 C.F.R. § 601.2, establishment registrations, as defined in 21 C.F.R. § 207,
and product listings, as defined in 21 C.F.R. § 207, all supplements or amendments thereto, and all comparable Governmental Authorizations.
Release.
“Release” shall mean any presence, emission, spill, seepage, leak, escape, leaching, discharge, injection, pumping,
pouring, emptying, dumping, disposal, migration, or release of Hazardous Materials from any source into or upon the environment, including
the air, soil, improvements, surface water, groundwater, the sewer, septic system, storm drain, publicly owned treatment works, or waste
treatment, storage, or disposal systems.
Representatives.
“Representatives” shall mean officers, directors, employees, attorneys, accountants, investment bankers, consultants,
agents, financial advisors, other advisors and other representatives.
Sanctioned
Person. “Sanctioned Person” shall mean any Person, aircraft, or vessel that is the subject or target of
sanctions or restrictions under the Global Trade Laws, including any Person: (a) listed on any list of sanctioned persons maintained
by the United States, United Nations Security Council, including, but not limited to, (i) the “Specially Designated Nationals
and Blocked Persons” list maintained by the U.S. Office of Foreign Assets Control or (ii) the Entity List or Military End
User List maintained by the U.S. Department of Commerce’s Bureau of Industry and Security; (b) located in, resident in, or
incorporated in, a Sanctioned Territory; and (c) any Person which is 50% or more owned, directly or indirectly, individually or
in the aggregate, or otherwise controlled, by any such Person or Persons described in subclauses (a)(i) or (b) of this definition.
Sanctioned
Territory. “Sanctioned Territory” shall mean any country or territory which is itself the subject or target
of any country-wide or territory-wide comprehensive economic sanctions imposed by the United States at any point over the previous five
years, including, but not limited to, Cuba, Iran, North Korea, Syria, or the Crimea and so-called Donetsk People’s Republic
and Luhansk People’s Republic regions of Ukraine.
Sarbanes-Oxley
Act. “Sarbanes-Oxley Act” is defined in Section 3.4(a) of the Agreement.
Schedule
14D-9. “Schedule 14D-9” is defined in Section 1.2(a) of the Agreement.
SEC.
“SEC” shall mean the United States Securities and Exchange Commission.
Securities
Act. “Securities Act” shall mean the Securities Act of 1933, as amended.
Shares.
“Shares” is defined in the Introduction to the Agreement.
Specified
Agreement. “Specified Agreement” is defined in Section 8.1(e) of the Agreement.
Specified
Governmental Body. “Specified Governmental Body” is defined in Section 7.1 of the Agreement.
Stockholder
List Date. “Stockholder List Date” is defined in Section 1.2(b) of the Agreement.
Subsidiary.
An Entity shall be deemed to be a “Subsidiary” of another Person if such Person directly or indirectly owns, beneficially
or of record, (a) an amount of voting securities or other interests in such Entity that is sufficient to enable such Person to elect
at least a majority of the members of such Entity’s board of directors or other governing body, or (b) at least 50% of the
outstanding equity or financial interests of such Entity.
Superior
Offer. “Superior Offer” shall mean a bona fide written Acquisition Proposal made after the date
hereof that the Board of Directors determines, in its good faith judgment, after consultation with the Company’s outside legal
counsel and financial advisors, is reasonably likely to be consummated in accordance with its terms, taking into account all legal, regulatory
and financing aspects (including the identity of the third party making such Acquisition Proposal, anticipated timing and prospects for
consummating such Acquisition Proposal, the reliability of any debt or equity funding commitments, any termination or breakup fees, expense
reimbursement provisions and any conditions) of the proposal and the Person making the proposal, and such other aspects of the Acquisition
Proposal that the Board of Directors deems relevant, and if consummated, would result in a transaction more favorable to the Company’s
stockholders (solely in their capacity as such) from a financial point of view than the Transactions (including after giving effect to
proposals, if any, made by Parent pursuant to Section 6.1(b)(i)); provided that for purposes of the definition of
“Superior Offer,” the references to “20%” in the definition of Acquisition Proposal shall be deemed to be references
to “50%.”
Surviving
Corporation. “Surviving Corporation” is defined in the Introduction to the Agreement.
Takeover
Laws. “Takeover Laws” shall mean any “moratorium,” “control share acquisition,”
“fair price,” “supermajority,” “affiliate transactions,” or “business combination statute or
regulation” or other similar state anti-takeover laws and regulations (including, for the avoidance of doubt, Section 203
of the DGCL).
Tax.
“Tax” shall mean any federal, state, local, or foreign or other tax (including any net income tax, gross income tax,
franchise tax, capital gains tax, gross receipts tax, gross profits tax, branch profits tax, value-added tax, surtax, estimated tax,
employment tax, unemployment tax, national health insurance tax, excise tax, estimated tax, alternative or minimum tax, ad valorem tax,
transfer tax, stamp tax, sales tax, use tax, service tax, property tax, business tax, withholding tax or payroll tax), levy, assessment,
tariff, impost, imposition, duty (including any customs duty) or other tax or charge of any kind whatsoever, imposed, assessed or collected
by or under the authority of any Governmental Body, together with any interest, penalties, inflationary adjustments, additions to tax,
fines or other additional amounts imposed thereon, with respect thereto, or related thereto.
Tax
Return. “Tax Return” shall mean any return (including any information return), report, statement, declaration,
estimate, schedule, form, election, certificate or other document or information filed or required to be filed with any Governmental
Body in connection with the determination, assessment, collection or payment of any Tax and any attachments thereto or amendments thereof.
Termination
Condition. “Termination Condition” is defined in Annex I to the Agreement.
Termination
Fee. “Termination Fee” is defined in Section 8.3(b) of the Agreement.
Trademarks.
“Trademarks” is defined in the definition of Intellectual Property Rights.
Trade
Secrets. “Trade Secrets” is defined in the definition of Intellectual Property Rights.
Transactions.
“Transactions” shall mean (a) the execution and delivery of the Agreement and (b) all of the transactions
contemplated by the Agreement, including the Offer and the Merger.
U.S.
Bank. “U.S. Bank” is defined in the definition of Convertible Notes Indentures.
ANNEX I
CONDITIONS TO THE OFFER
The
obligation of Purchaser to accept for payment and pay for Shares validly tendered (and not validly withdrawn) pursuant to the Offer is
subject to the satisfaction of the conditions set forth in clauses (a) through (h) below. Notwithstanding any
other provision of the Offer or the Agreement to the contrary, Purchaser shall not be required to accept for payment or (subject to any
applicable rules and regulations of the SEC, including Rule 14e-1(c) under the Exchange Act) pay for, and may delay the
acceptance for payment of, or (subject to any such rules and regulations) the payment for, any tendered Shares, and, to the extent
permitted by the Agreement, may terminate the Offer: (i) upon valid termination of the Agreement in accordance with its terms; and
(ii) at any scheduled Expiration Date (subject to any extensions of the Offer pursuant to Section 1.1(c) of the
Agreement), if any of the conditions set forth below shall not be satisfied or waived in writing by Parent and Purchaser (to the
extent waivable by Parent and Purchaser) by one minute after 11:59 p.m. Eastern Time on the Expiration Date:
(a) there
shall have been validly tendered (and not validly withdrawn) Shares that, considered together with all other Shares (if any) beneficially
owned by Parent and its Affiliates, represent one more Share than 50% of the sum of the total number of Shares outstanding at the time
of the expiration of the Offer (the “Minimum Condition”); provided, however, that for purposes of determining
whether the Minimum Condition has been satisfied, the Parties shall exclude Shares tendered in the Offer pursuant to guaranteed delivery
procedures that have not yet been “received” (as such term is defined in Section 251(h)(6)(f) of the DGCL);
(b) (i) the
representations and warranties of the Company set forth in the first two sentences of Section 3.1(a) (Due
Organization; Subsidiaries, Etc.), the first sentence of Section 3.2 (Certificate of Incorporation and Bylaws), Section 3.21
(Authority; Binding Nature of Agreement), Section 3.22(a) (Takeover Laws), Section 3.24 (Opinion of
Financial Advisors) and Section 3.25 (Brokers and Other Advisors) of the Agreement shall be accurate in all material
respects as of the date of the Agreement and at and as of the Offer Acceptance Time as if made on and as of the Offer Acceptance
Time (except to the extent any such representation or warranty expressly relates to an earlier date or period, in which case as of
such date or period);
(ii) the
representations and warranties of the Company set forth in Sections 3.3(a) and 3.3(d) (Capitalization, Etc.)
shall be true and accurate in all respects except for de minimis inaccuracies as of the date of the Agreement and at and as of
the Offer Acceptance Time as if made on and as of the Offer Acceptance Time (except to the extent any such representation or warranty
expressly relates to an earlier date or period, in which case as of such date or period);
(iii) the
representations and warranties of the Company set forth in Section 3.5(b) (No Material Adverse Effect) of the Agreement
shall be accurate in all respects as of the date of the Agreement and at and as of the Offer Acceptance Time as if made on and as of
the Offer Acceptance Time;
(iv) the
representations and warranties of the Company set forth in the Agreement (other than those referred to in clauses (b)(i) through
(b)(iii) above) shall be true and accurate (without taking into account any “Material Adverse Effect” and “materiality”
qualifications contained in such representations and warranties) as of the date of the Agreement and at and as of the Offer Acceptance
Time as if made on and as of the Offer Acceptance Time (except to the extent any such representation or warranty expressly relates to
an earlier date or period, in which case as of such date or period), except where the failure of such representations and warranties
to be so true and accurate has not had, and would not reasonably be expected to have, individually or in the aggregate, a Material Adverse
Effect;
(c) the
Company shall have complied with or performed in all material respects the covenants and agreements it is required to comply with or
perform at or prior to the Offer Acceptance Time;
(d) since
the execution and delivery of the Agreement, there shall not have occurred any Material Adverse Effect which is continuing;
(e) Parent
and Purchaser shall have received a certificate executed on behalf of the Company by the Company’s Chief Executive Officer or Chief
Financial Officer confirming that the conditions set forth in clauses (b), (c) and (d) of this Annex
I have been satisfied;
(f) any
waiting period (or any extension thereof) applicable to the Offer under the HSR Act shall have expired or been terminated;
(g) no
Specified Governmental Body shall have issued, promulgated, enacted or deemed applicable to the Merger or the Offer any order, injunction,
decree, ruling or other Legal Requirement (whether temporary, preliminary or permanent) or taken any other action, in each case restraining,
enjoining, making illegal or otherwise prohibiting the consummation of the Merger or the Offer, which order, injunction, decree, ruling,
or other Legal Requirement or action remains in effect;
(h) the
Agreement shall not have been terminated in accordance with its terms (the “Termination Condition”); and
(i) the
Company shall have received correspondence from the FDA acknowledging that the Company’s proposed revised milestone for certain
postmarketing requirements will be December 2023, as requested in the Company’s submission to the FDA made on September 25,
2023.
The foregoing conditions are for the sole benefit of Parent and Purchaser
and (except for the Minimum Condition, the Termination Condition and the conditions set forth in clauses (f) and (g))
may be waived by Parent and Purchaser, in whole or in part at any time and from time to time, in the sole discretion of Parent and Purchaser,
to the extent permitted under applicable Legal Requirements. All capitalized terms used but not defined in this Annex I shall
have the meanings ascribed to them in the Agreement to which this Annex I is attached.
ANNEX
II
FORM OF
CERTIFICATE OF INCORPORATION OF THE SURVIVING CORPORATION
FIRST:
The name of the corporation is Intercept Pharmaceuticals, Inc. (the “Corporation”).
SECOND:
The address of the registered office of the Corporation in the State of Delaware is Corporation Service Company, 251 Little
Falls Drive, County of New Castle, Wilmington, Delaware 19808. The name of the Corporation’s registered agent at such address is
Corporation Service Company.
THIRD:
The nature of the business or purposes to be conducted or promoted is to engage in any lawful act or activity for which corporations
may be organized under the General Corporation Law of the State of Delaware, as amended (the “DGCL”).
FOURTH:
The total number of shares of all classes of stock which the Corporation shall have authority to issue is one thousand (1,000), consisting
of 1,000 shares of Common Stock, having a par value of $0.01 per share.
FIFTH:
In furtherance of and not in limitation of powers conferred by statute, it is further provided:
| 1. | The
business and affairs of the Corporation shall be managed by or under the direction of the
board of directors of the Corporation. |
| 2. | Election
of directors need not be by written ballot. |
| 3. | The
board of directors of the Corporation is expressly authorized to adopt, amend, alter or repeal
the bylaws of the Corporation. |
SIXTH:
Except to the extent that the DGCL prohibits the elimination or limitation of liability of directors for breaches of fiduciary
duty, no director of the Corporation shall be personally liable to the Corporation or its stockholders for monetary damages for breach
of fiduciary duty as a director, notwithstanding any provision of law imposing such liability; provided that this Article SIXTH
shall not eliminate or limit the liability of a director (i) for any breach of the director’s duty of loyalty to the Corporation
or its stockholders, (ii) for acts or omissions not in good faith or which involve intentional misconduct or a knowing violation
of law, (iii) under Section 174 of the DGCL, or (iv) for any transaction from which the director derived any improper
personal benefit. No amendment to or repeal of this provision shall apply to or have any effect on the liability or alleged liability
or any director of the Corporation for or with respect to any acts or omissions of such director occurring prior to such amendment or
repeal. If the DGCL is amended to authorize corporate action further eliminating or limiting the personal liability of directors, then
the liability of a director of the Corporation shall be eliminated or limited to the fullest extent permitted by the DGCL as so amended.
SEVENTH:
To the fullest extent permitted by applicable law, as amended from time to time, the Corporation is authorized to provide
indemnification for (and advancement of expenses to) directors, officers and agents of the Corporation (and any other persons to which
applicable law permits the Corporation to provide indemnification) through bylaw provisions, agreements with such agents or other persons
(including Section 6.4(a) of that certain Agreement and Plan of Merger, dated September 26, 2023, by and among Intercept
Pharmaceuticals, Inc., Alfasigma S.p.A. and Interstellar Acquisition Inc.), vote of stockholders or disinterested directors or otherwise in excess
of the indemnification and advancement otherwise permitted by such applicable law. Any repeal or modification of this provision shall
not adversely affect any right or protection hereunder of any person in respect of any act or omission occurring prior to the time of
such repeal or modification.
EIGHTH:
Subject to such limitations as may be from time to time imposed by other provisions of this Second Amended and Restated Certificate
of Incorporation, by the bylaws of the Corporation, by the DGCL or other applicable law, or by any contract or agreement to which the
Corporation is or may become a party, the Corporation reserves the right to amend, alter, change or repeal any provision contained in
this Second Amended and Restated Certificate of Incorporation, in the manner now or hereafter prescribed by statute and this Second Amended
and Restated Certificate of Incorporation, and all rights conferred upon stockholders herein are granted subject to this reservation.
Exhibit 99.1
Alfasigma to Acquire Intercept Pharmaceuticals for $19.00 per Share
in Cash, Expanding the Global Footprint of Alfasigma Via a Leader in Rare and Serious Liver Diseases
| ● | Proposed all-cash acquisition will materially expand Alfasigma’s portfolio in gastroenterology and hepatology and its presence in
the U.S. market |
| ● | Transaction price represents an 82% premium to Intercept’s closing
price on September 25, 2023 |
| ● | Alfasigma adds Ocaliva®, the only FDA approved second-line treatment for PBC, a progressive autoimmune disease affecting
the liver, which generated revenue of $152 million in 1H 2023 |
| ● | Strengthens Alfasigma’s pipeline, including the addition of
a novel fixed-dose combination with potential to establish a new treatment paradigm in PBC |
| ● | Alfasigma to commence cash tender offer to acquire all issued and
outstanding shares of Intercept for US$19.00 per share |
| ● | Transaction expected to close by the end of
2023 |
Bologna, Italy & Morristown, N.J., U.S.
– September 26, 2023 – Alfasigma S.p.A (“Alfasigma”), one of Italy's leading pharmaceutical companies, and
Intercept Pharmaceuticals, Inc. (Nasdaq: ICPT, “Intercept”), a leading biopharmaceutical company in rare and serious liver
diseases, today announced that they have entered into a definitive merger agreement under which Alfasigma has agreed to acquire Intercept
for $19.00 per share in cash,. The anticipated transaction will materially expand Alfasigma’s gastrointestinal and hepatology
portfolio and its presence in the U.S. market.
Intercept’s lead medicine is Ocaliva® (obeticholic acid), a farnesoid X receptor agonist approved in the United States and several other jurisdictions for the treatment of primary biliary cholangitis (“PBC”)
in combination with ursodeoxycholic acid (“UDCA”) in adults with an inadequate response to UDCA, or as monotherapy in adults
unable to tolerate UDCA. Ocaliva® is the only approved second-line therapy for PBC and has experienced double-digit year-over-year
growth supported by an experienced specialty sales force and strong prescriber base. Intercept also benefits from a broader clinical development
pipeline anchored by a novel fixed-dose combination of obeticholic acid and bezafibrate in phase 2 trials for PBC.
Mr. Stefano Golinelli, Chairman of Alfasigma Board, declared:
“Today’s proposed acquisition is aligned with our strategy to build presence in the U.S. market, with a focus in our
core gastroenterological area while adding another important asset to our innovation pipeline. This acquisition will contribute to the
ambitious growth strategy designed for our company.”
Mr. Francesco Balestrieri, Chief Executive Officer of Alfasigma,
said: “The acquisition of Intercept marks another important milestone in Alfasigma’s growth path, particularly with regard to
the U.S. market in which we have significant
development objectives. Intercept represents a compelling fit with Alfasigma’s core business areas
of gastroenterology and hepatology, and we believe that the transaction represents a transformational opportunity for both companies.
We are excited to welcome Intercept employees and look forward to working together as we invest in the company to realize the full potential,
to the benefit of patients.”
Mr. Jerry Durso, President
and Chief Executive Officer of Intercept, commented: “We are pleased to announce this transaction with Alfasigma,
which delivers significant value to shareholders. Importantly, it recognizes the value of
our portfolio, R&D and commercial capabilities and our talented people across
the organization. The team at Intercept is proud of the breakthrough, innovative work that we have done as a pioneer, delivering
life-saving medicine to patients with rare and serious liver diseases such as PBC.”
Transaction Terms
Under the terms of the merger agreement, Alfasigma has
agreed to commence a cash tender offer to acquire all issued and outstanding shares of Intercept common stock for US$19.00 per
share in cash. The purchase price represents a premium of 82% to Intercept’s closing stock price on September 25, 2023.
The transaction will be fully financed by Alfasigma’s existing
cash on hand and existing corporate credit facilities. The members of the Board of Directors of Intercept participating in the decision
have unanimously approved the transaction.
The closing of the tender offer will be subject to customary conditions,
including the tender of shares which represent at least a majority of the total number of Intercept’s outstanding shares of common
stock and the expiration of the waiting period under the Hart-Scott-Rodino Antitrust Improvements Act. Upon successful completion
of the tender offer, Alfasigma would acquire all shares not acquired in the tender offer through a second-step merger for the same consideration
that the tendering stockholders will receive in the tender offer.
It is anticipated the transaction will close by the
end of 2023. Upon completion of the transaction, Intercept’s common stock will no longer be publicly listed.
Advisors
Barclays and Centerview Partners are serving as financial advisors
to Intercept. Skadden, Arps, Slate, Meagher & Flom LLP are serving as legal counsel to Intercept. PJT Partners is acting as exclusive
financial advisor to Alfasigma and Sullivan & Cromwell LLP and Chiomenti Studio Legale as
legal counsel to Alfasigma.
About Alfasigma
Alfasigma is one of Italy's leading pharmaceutical
companies with a strong international position. The Group has a worldwide presence in over 100 countries where about 3000 people work
in research, development, production and distribution. In Italy, Alfasigma is a leader in the prescription products market where,
in addition to its strong focus on gastro-intestinal products, it is present in several primary care therapeutic areas. It is popular
with the consumer public for a number of nutraceuticals & food supplements that respond to different needs, and that are well known
and deeply rooted in the Italian families experience. Its historical headquarters is in Bologna, to which is added Milan, while
the production sites are: in Italy, in Pomezia (RM), Alanno (PE), Sermoneta (LT) and Trezzano Rosa (MI) and abroad in Tortosa in Spain
and in Shreveport (Louisiana) in the United States. The R&D laboratories are in Pomezia and in the Parco Scientifico Tecnologico
Kilometro Rosso in Bergamo. Alfasigma's mission is to improve people's health and quality of life by offering caregivers and healthcare
personnel therapeutic solutions according to the highest standards of quality and safety.
About Intercept
Intercept is a biopharmaceutical
company focused on the development and commercialization of novel therapeutics to treat rare and serious liver diseases, including primary
biliary cholangitis (PBC) and severe alcohol-associated hepatitis (sAH). For more information, please visit www.interceptpharma.com or
connect with the Company on Twitter and LinkedIn.
About Ocaliva® (obeticholic acid)
OCALIVA, a farnesoid X receptor (FXR) agonist, is indicated for the treatment of adult patients with primary biliary cholangitis (PBC)
| · | with compensated cirrhosis who do not have evidence of portal hypertension, either in combination with ursodeoxycholic acid (UDCA)
with an inadequate response to UDCA or as monotherapy in patients unable to tolerate UDCA. |
This indication is approved under accelerated approval based on a reduction
in alkaline phosphatase (ALP). An improvement in survival or disease-related symptoms has not been established. Continued approval for
this indication may be contingent upon verification and description of clinical benefit in confirmatory trials.
IMPORTANT SAFETY INFORMATION
WARNING: HEPATIC DECOMPENSATION AND FAILURE
IN PRIMARY BILIARY CHOLANGITIS PATIENTS WITH CIRRHOSIS
| · | Hepatic decompensation and failure, sometimes fatal or resulting in liver transplant, have been reported with OCALIVA treatment
in primary biliary cholangitis (PBC) patients with either compensated or decompensated cirrhosis. |
| · | OCALIVA is contraindicated in PBC patients with decompensated cirrhosis, a prior decompensation event, or with compensated cirrhosis
who have evidence of portal hypertension. |
| · | Permanently discontinue OCALIVA in patients who develop laboratory or clinical evidence of hepatic decompensation; have compensated
cirrhosis and develop evidence of portal hypertension, or experience clinically significant hepatic adverse reactions while on treatment. |
Contraindications
OCALIVA is contraindicated in patients with:
| · | decompensated cirrhosis (e.g., Child-Pugh Class B or C) or a prior decompensation event |
| · | compensated cirrhosis who have evidence of portal hypertension (e.g., ascites, gastroesophageal varices, persistent thrombocytopenia) |
| · | complete biliary obstruction |
Warnings and Precautions
Hepatic Decompensation and Failure in PBC Patients with Cirrhosis
Hepatic decompensation and failure, sometimes fatal or resulting in liver transplant, have been reported with OCALIVA treatment in PBC
patients with cirrhosis, either compensated or decompensated. Among post-marketing cases reporting it, median time to hepatic decompensation
(e.g., new onset ascites) was 4 months for patients with compensated cirrhosis; median time to a new decompensation event (e.g., hepatic
encephalopathy) was 2.5 months for patients with decompensated cirrhosis.
Some of these cases occurred in patients with decompensated cirrhosis
when they were treated with higher than the recommended dosage for that patient population; however, cases of hepatic decompensation and
failure have continued to be reported in patients with decompensated cirrhosis even when they received the recommended dosage.
Hepatotoxicity was observed in the OCALIVA clinical trials. A dose-response
relationship was observed for the occurrence of hepatic adverse reactions including jaundice, worsening ascites, and primary biliary cholangitis
flare with dosages of OCALIVA of 10 mg once daily to 50 mg once daily (up to 5-times the highest recommended dosage), as early as one
month after starting treatment with OCALIVA in two 3-month, placebo-controlled clinical trials in patients with primarily early stage
PBC.
Routinely monitor patients for progression of PBC, including hepatic
adverse reactions, with laboratory and clinical assessments to determine whether drug discontinuation is needed. Closely monitor patients
with compensated cirrhosis, concomitant hepatic disease (e.g., autoimmune hepatitis, alcoholic liver disease), and/or with severe intercurrent
illness for new evidence of portal hypertension (e.g., ascites, gastroesophageal varices, persistent thrombocytopenia), or increases above
the upper limit of normal in total bilirubin, direct bilirubin, or prothrombin time to determine whether drug discontinuation is needed.
Permanently discontinue OCALIVA in patients who develop laboratory or clinical evidence of hepatic decompensation (e.g., ascites, jaundice,
variceal bleeding, hepatic encephalopathy), have compensated cirrhosis and develop evidence of portal hypertension (e.g., ascites, gastroesophageal
varices, persistent thrombocytopenia), experience clinically significant hepatic adverse reactions, or develop complete biliary obstruction.
If severe intercurrent illness occurs, interrupt treatment with OCALIVA and monitor the patient’s liver function. After resolution
of the intercurrent illness, consider the potential risks and benefits of restarting OCALIVA treatment.
Severe Pruritus
Severe pruritus was reported in 23% of patients in the OCALIVA 10 mg arm, 19% of patients in the OCALIVA titration arm, and 7% of patients
in the placebo arm in a 12-month double-blind randomized controlled clinical trial of 216 patients. Severe pruritus was defined as intense
or widespread itching, interfering with activities of daily living, or causing severe sleep disturbance, or intolerable discomfort, and
typically requiring medical interventions. Consider clinical evaluation of patients with new onset or worsening severe pruritus. Management
strategies include the addition of bile acid binding resins or antihistamines, OCALIVA dosage reduction, and/or temporary interruption
of OCALIVA dosing.
Reduction in HDL-C
Patients with PBC generally exhibit hyperlipidemia characterized by a significant elevation in total cholesterol primarily due to increased
levels of high-density lipoprotein-cholesterol (HDL-C). Dose-dependent reductions from baseline in mean HDL-C levels were observed at
2 weeks in OCALIVA-treated patients, 20% and 9% in the 10 mg and titration arms, respectively, compared to 2% in the placebo arm. Monitor
patients for changes in serum lipid levels during treatment. For patients who do not respond to OCALIVA after 1 year at the highest recommended
dosage that can be tolerated (maximum of 10 mg once daily), and who experience a reduction in HDL-C, weigh the potential risks against
the benefits of continuing treatment.
Adverse Reactions
The most common adverse reactions (≥5%) are: pruritus, fatigue, abdominal pain and discomfort, rash, oropharyngeal pain, dizziness,
constipation, arthralgia, thyroid function abnormality, and eczema.
Drug Interactions
| · | Bile Acid Binding Resins
Bile acid binding resins such as cholestyramine, colestipol, or colesevelam adsorb and reduce bile acid absorption and may reduce the
absorption, systemic exposure, and efficacy of OCALIVA. If taking a bile acid binding resin, take OCALIVA at least 4 hours before or 4
hours after taking the bile acid binding resin, or at as great an interval as possible. |
| · | Warfarin
The International Normalized Ratio (INR) decreased following coadministration of warfarin and OCALIVA. Monitor INR and adjust the dose
of warfarin, as needed, to maintain the target INR range when co-administering OCALIVA and warfarin. |
| · | CYP1A2 Substrates with Narrow Therapeutic Index
Obeticholic acid may increase the exposure to concomitant drugs that are CYP1A2 substrates. Therapeutic monitoring of CYP1A2 substrates
with a narrow therapeutic index (e.g., theophylline and tizanidine) is recommended when co-administered with OCALIVA. |
| · | Inhibitors of Bile Salt Efflux Pump
Avoid concomitant use of inhibitors of the bile salt efflux pump (BSEP) such as cyclosporine. Concomitant medications that inhibit canalicular
membrane bile acid transporters such as the BSEP may exacerbate accumulation of conjugated bile salts including taurine conjugate of obeticholic
acid in the liver and result in clinical symptoms. If concomitant use is deemed necessary, monitor serum transaminases and bilirubin. |
Please click here for Full
Prescribing Information, including Boxed WARNING.
To report SUSPECTED ADVERSE REACTIONS, contact Intercept Pharmaceuticals, Inc. at 1-844-782-ICPT or FDA at 1-800-FDA-1088
or www.fda.gov/medwatch.
Additional Information and Where to Find it
The tender offer described in this communication has not yet commenced.
This communication is for informational purposes only and is neither an offer to purchase nor a solicitation of an offer to sell shares
of Intercept Pharmaceuticals, Inc. (the “Company”), nor is it a substitute for any tender offer materials that the
Company or Alfasigma S.p.A. (together with its subsidiaries, “Alfasigma”) will file with the SEC. A solicitation and an
offer to buy shares of the Company will be made only pursuant to an offer to purchase and related materials that Alfasigma intends to
file with the SEC. At the time the tender offer is commenced, Alfasigma will file a Tender Offer Statement on Schedule TO with the SEC,
and the Company will file a Solicitation/Recommendation Statement on Schedule 14D-9 with the SEC with respect to the tender offer. THE
COMPANY’S STOCKHOLDERS AND OTHER INVESTORS ARE URGED TO READ THE TENDER OFFER MATERIALS (INCLUDING AN OFFER TO PURCHASE, A RELATED
LETTER OF TRANSMITTAL AND CERTAIN OTHER TENDER OFFER DOCUMENTS) AND THE SOLICITATION/RECOMMENDATION STATEMENT BECAUSE THEY WILL CONTAIN
IMPORTANT INFORMATION WHICH SHOULD BE READ CAREFULLY BEFORE ANY DECISION IS MADE WITH RESPECT TO THE TENDER OFFER. The Offer to Purchase,
the related Letter of Transmittal and certain other tender offer documents, as well as the Solicitation/Recommendation Statement, will
be sent to all stockholders of the Company at no expense to them. The Tender Offer Statement and the Solicitation/Recommendation Statement
will be made available for free at the SEC’s website at www.sec.gov. Additional copies may be obtained for free by contacting
Alfasigma or the Company. Free copies of these materials and certain other offering documents will be made available by the Company by
mail to Intercept Pharmaceuticals, Inc., 305 Madison Avenue, Morristown, NJ 07960, Attention: Corporate Secretary, by email at investors@interceptpharma.com,
or by directing requests for such materials to the information agent for the offer, which will be named in the tender offer materials.
Copies of the documents filed with the SEC by the Company will be available free of charge under the “Investors & Media”
section of the Company’s internet website at https://ir.interceptpharma.com/investor-relations.
In addition to the Offer to Purchase, the related Letter of Transmittal
and certain other tender offer documents, as well as the Solicitation/Recommendation Statement, the Company files periodic reports and
other information with the SEC. The Company’s filings with the SEC are also available for free to the public from commercial document-retrieval
services and at the website maintained by the SEC at www.sec.gov.
Forward Looking Statements
This communication contains forward-looking statements related to
the Company, Alfasigma and the proposed acquisition of the Company by Alfasigma (the “Transaction”) that involve
substantial risks and uncertainties. Forward-looking statements include any statements containing the words
“anticipate,” “believe,” “estimate,” “expect,” “intend,”
“goal,” “may,” “might,” “plan,” “predict,” “project,”
“seek,” “target,” “potential,” “will,” “would,” “could,”
“should,” “continue” and similar expressions. In this communication, the Company’s forward-looking
statements include statements about the parties’ ability to satisfy the conditions to the consummation of the tender offer and
the other conditions to the consummation of the Transaction; statements about the expected timetable for completing the Transaction;
the Company’s plans, objectives, expectations and intentions; the financial condition, results of operations and business of
the Company and Alfasigma; the ability to successfully commercialize the Company’s product and product candidates and generate
future revenues with respect to the Company’s product candidates; and the anticipated timing of the closing of the
Transaction.
Forward-looking statements are subject to certain risks, uncertainties,
or other factors that are difficult to predict and could cause actual events or results to differ materially from those indicated in
any such statements due to a number of risks and uncertainties. Those risks and uncertainties that could cause the actual results to
differ from expectations contemplated by forward-looking statements include, among other things: uncertainties as to the timing of the
tender offer and merger; uncertainties as to how many of the Company’s stockholders will tender their stock in the offer; the possibility
that competing offers will be made; the possibility that various closing conditions for the Transaction may not be satisfied or waived,
including that a governmental entity may prohibit, delay or refuse to grant approval for the consummation of the Transaction; the effects
of the Transaction on relationships with employees, other business partners or governmental entities; the difficulty of predicting the
timing or outcome of FDA approvals or actions, if any; the impact of competitive products and pricing; that Alfasigma may not realize
the potential benefits of the Transaction; other business effects, including the effects of industry, economic or political conditions
outside of the companies’ control; Transaction costs; actual or contingent liabilities; and other risks listed under the heading
“Risk Factors” in the Company’s periodic reports filed with the U.S. Securities and Exchange Commission, including
current reports on Form 8-K, quarterly reports on Form 10-Q, annual reports on Form 10-K, as well as the Schedule 14D-9 to be filed by
the Company and the Schedule TO and related tender offer documents to be filed by Alfasigma and Interstellar
Acquisition, Inc., a wholly owned subsidiary of Alfasigma. You should not place undue reliance on these statements. All forward-looking
statements are based on information currently available to the Company and Alfasigma, and the Company and Alfasigma disclaim any obligation
to update the information contained in this communication as new information becomes available.
Contacts
Alfasigma
Corporate Communication
Simona Gelpi simona.gelpi@alfasigma.com
Gea Gardini gea.gardini@alfasigma.com
www.alfasigma.it
iCorporate
Luca Bolzoni luca.bolzoni@icorporate.it
Intercept
Investors
Nareg Sagherian, Executive Director, Global Investor Relations
investors@interceptpharma.com
Media
Michael Blash, Senior Vice President, Corporate Affairs
media@interceptpharma.com
v3.23.3
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Intercept Pharmaceuticals (NASDAQ:ICPT)
Historical Stock Chart
From May 2024 to Jun 2024
Intercept Pharmaceuticals (NASDAQ:ICPT)
Historical Stock Chart
From Jun 2023 to Jun 2024