Ironwood Announces U.S. Availability of ZURAMPIC® (lesinurad) 200 mg Tablets for Patients with Uncontrolled Gout
October 03 2016 - 6:30AM
Business Wire
- In clinical studies, ZURAMPIC in combination
with allopurinol nearly doubled the number of patients who achieved
target serum uric acid (sUA) levels at month 6 compared to patients
who failed to achieve target levels on allopurinol alone -
Ironwood Pharmaceuticals, Inc. (NASDAQ: IRWD) today announced
that ZURAMPIC® (lesinurad) is now available in pharmacies
throughout the United States. ZURAMPIC is approved as a once-daily
oral tablet to be taken in combination with a xanthine oxidase
inhibitor (XOI) for the treatment of hyperuricemia – high serum
uric acid (sUA) levels in the blood – associated with gout in
patients who have not achieved target sUA levels with an XOI alone.
ZURAMPIC is not recommended for the treatment of asymptomatic
hyperuricemia and should not be used as monotherapy.
This Smart News Release features multimedia.
View the full release here:
http://www.businesswire.com/news/home/20161003005125/en/
ZURAMPIC® (lesinurad) is now available in
pharmacies throughout the United States. (Photo: Business Wire)
The safety and efficacy of ZURAMPIC were established in three
Phase III, multicenter, randomized, double-blind, 12-month,
placebo-controlled clinical studies in adult patients with
hyperuricemia and gout. In two of the trials, ZURAMPIC was studied
in combination with allopurinol and in a third trial was studied in
combination with febuxostat. Allopurinol and febuxostat are each
XOIs. In clinical trials, when added to allopurinol in patients
with gout who failed to achieve target sUA levels with allopurinol
alone, ZURAMPIC nearly doubled the number of patients who achieved
sUA target of <6 mg/dL at month 6, reduced the mean sUA to <6
mg/dL by month 1 and maintained that level through month 12. When
added to febuxostat 80 mg in clinical trials, ZURAMPIC helped a
greater, but not statistically significant, proportion of patients
with tophaceous gout achieve sUA <5 mg/dL at month 6, and
maintained that level through month 12. Acute renal failure has
occurred with ZURAMPIC and was more common when ZURAMPIC was given
alone. ZURAMPIC should be used in combination with an XOI. Most
common adverse reactions with ZURAMPIC in the clinical trials were
headache, influenza, blood creatinine increased, and
gastroesophageal reflux disease.
About Hyperuricemia and GoutGout is a highly symptomatic
and painful form of inflammatory arthritis affecting an estimated
eight million people in the U.S. It is caused by an underlying
metabolic disorder, hyperuricemia – high levels of uric acid in the
blood – and can lead to painful flares, characterized by
excruciating pain, inflammation, swelling and tenderness in one or
more joints. Gout is commonly hereditary and not only a lifestyle
disease. While diet and lifestyle changes are important in managing
gout and its comorbidities, they are often not enough to get
patient serum uric acid (sUA) levels to target.
Approximately four million patients are treated with a xanthine
oxidase inhibitor (XOI), either allopurinol or febuxostat, for gout
in the U.S. Of these, an estimated two million patients are
uncontrolled and are not achieving target serum uric acid (sUA)
levels <6 mg/dL as recommended by the American College of
Rheumatology, despite treatment with an XOI alone. These patients
continue to suffer from flares despite treatment with an XOI alone,
and may face serious long-term consequences that can result from
having uncontrolled sUA levels.
About ZURAMPIC® (lesinurad) 200 mg
tablets
ZURAMPIC® (lesinurad) is a URAT1 inhibitor approved by the FDA
for use in combination with a xanthine oxidase inhibitor (XOI) for
the treatment of hyperuricemia associated with gout in patients who
have not achieved target serum uric acid levels (sUA) with an XOI
alone. ZURAMPIC is not recommended for the treatment of
asymptomatic hyperuricemia and should not be used as a monotherapy.
XOIs reduce the production of uric acid; ZURAMPIC increases renal
excretion of uric acid by selectively inhibiting the action of
URAT1, the UA transporter responsible for the majority of renal UA
reabsorption. The dual-mechanism combination of ZURAMPIC plus an
XOI (allopurinol or febuxostat) can address both inefficient
excretion and overproduction of UA, thereby lowering sUA levels.
The safety and efficacy of ZURAMPIC were established in three Phase
III clinical trials that evaluated a once-daily dose of ZURAMPIC in
combination with the XOI allopurinol or febuxostat compared to XOI
alone. Visit www.zurampic.com for more information about
ZURAMPIC.
Important Safety Information
WARNING: RISK OF ACUTE RENAL FAILURE
MORE COMMON WHEN USED WITHOUT A XANTHINE OXIDASE INHIBITOR
(XOI)
-- Acute renal failure has occurred
with ZURAMPIC and was more common when ZURAMPIC was given
alone
-- ZURAMPIC should be used in
combination with an XOI
Contraindications:
- Severe renal impairment (eCLcr less
than 30 mL/min), end-stage renal disease, kidney transplant
recipients, or patients on dialysis
- Tumor lysis syndrome or Lesch-Nyhan
syndrome
Warnings and Precautions:
- Renal events: Adverse reactions
related to renal function have occurred after initiating ZURAMPIC.
A higher incidence was observed at the 400-mg dose, with the
highest incidence occurring with monotherapy use. Monitor renal
function at initiation and during therapy with ZURAMPIC,
particularly in patients with eCLcr below 60 mL/min or with serum
creatinine elevations 1.5 to 2 times the pre-treatment value, and
evaluate for signs and symptoms of acute uric acid nephropathy.
Interrupt treatment with ZURAMPIC if serum creatinine is elevated
to greater than 2 times the pre-treatment value or if there are
symptoms that may indicate acute uric acid nephropathy. ZURAMPIC
should not be restarted without another explanation for the serum
creatinine abnormalities. ZURAMPIC should not be initiated in
patients with an eCLcr less than 45 mL/min.
- Cardiovascular events: In
clinical trials, major adverse cardiovascular events (defined as
cardiovascular deaths, non-fatal myocardial infarctions, or
non-fatal strokes) were observed with ZURAMPIC. A causal
relationship has not been established.
Adverse Reactions:
- Most common adverse reactions with
ZURAMPIC (in combination with an XOI and more frequently than on an
XOI alone) were headache, influenza, blood creatinine increased,
and gastroesophageal reflux disease.
Indication and Limitations of Use for ZURAMPIC:ZURAMPIC
is a URAT1 inhibitor indicated in combination with an XOI for the
treatment of hyperuricemia associated with gout in patients who
have not achieved target serum uric acid levels with an XOI
alone.
- ZURAMPIC is not recommended for the
treatment of asymptomatic hyperuricemia
- ZURAMPIC should not be used as
monotherapy
Please see full Prescribing Information, including Boxed
WARNING, http://www.azpicentral.com/zurampic/zurampic.pdf
About Ironwood PharmaceuticalsIronwood
Pharmaceuticals (NASDAQ: IRWD) is a commercial biotechnology
company focused on creating medicines that make a difference for
patients, building value for our fellow shareholders, and
empowering our passionate team. We are advancing a pipeline of
innovative medicines in areas of significant unmet need, including
irritable bowel syndrome with constipation (IBS-C)/chronic
idiopathic constipation (CIC), uncontrolled gout, refractory
gastroesophageal reflux disease, and vascular and fibrotic
diseases. We discovered, developed and are commercializing
linaclotide, the U.S. branded prescription market leader in the
IBS-C/CIC category, and we are applying our proven R&D and
commercial capabilities to advance multiple internally-developed
and externally-accessed product opportunities. Ironwood was founded
in 1998 and is headquartered in Cambridge, Mass. For more
information, please visit
www.ironwoodpharma.com or www.twitter.com/ironwoodpharma;
information that may be important to investors will be routinely
posted in both these locations.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20161003005125/en/
Media RelationsTrista Morrison, 617-374-5095Director, Corporate
Communicationstmorrison@ironwoodpharma.comorInvestor
RelationsMeredith Kaya, 617-374-5082Director, Investor
Relationsmkaya@ironwoodpharma.com
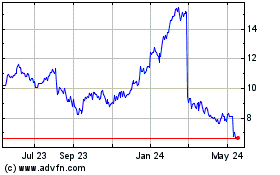
Ironwood Pharmaceuticals (NASDAQ:IRWD)
Historical Stock Chart
From Apr 2024 to May 2024
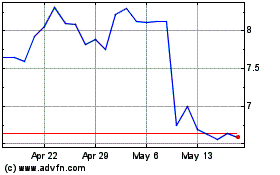
Ironwood Pharmaceuticals (NASDAQ:IRWD)
Historical Stock Chart
From May 2023 to May 2024