Correction to Roche Tecentriq Article on Monday
March 09 2021 - 6:17AM
Dow Jones News
Roche Holding AG said IMvigor211, the original confirmatory
study for the accelerated approval, didn't meet its primary
endpoint. Tecentriq had been granted that approval in 2016 based on
the results from the IMvigor210 study. "Roche Withdraws
Bladder-Cancer Indication for Tecentriq in US," at 0629 GMT on
Monday, incorrectly said the IMvigor210 study didn't meet its
primary endpoint.
(END) Dow Jones Newswires
March 09, 2021 07:02 ET (12:02 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
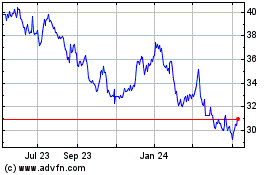
Roche (QX) (USOTC:RHHBY)
Historical Stock Chart
From Apr 2024 to May 2024
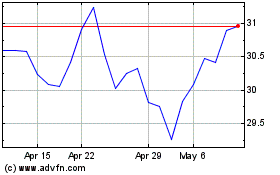
Roche (QX) (USOTC:RHHBY)
Historical Stock Chart
From May 2023 to May 2024