IN8bio, Inc. (Nasdaq: INAB), a clinical-stage biopharmaceutical
company discovering and developing innovative gamma-delta T cell
therapies that utilize its DeltEx platform, today announced it has
selected the UofL Health - Brown Cancer Center GMP Manufacturing
Facility at the University of Louisville, home of the Dunbar CAR
T-Cell Program, as its manufacturing partner for INB-400. The
collaboration provides IN8bio exclusive access to a
state-of-the-art GMP manufacturing facility and is structured to
support the Company’s anticipated INB-400 Phase 2 clinical program
in glioblastoma.
“We are proud to be partnering with the University of Louisville
(UofL), the UofL Health - Brown Cancer Center and the Dunbar CAR
T-Cell Program who have been at the forefront of clinical
development and patient access to novel cellular immunotherapies.
One of the biggest challenges facing cell therapy companies is
efficient access to high-quality manufacturing facilities with the
experience and capacity required to support multicenter clinical
trials and commercial manufacturing,” said Kate Rochlin, Ph.D.,
Chief Operating Officer of IN8bio. “The Dunbar CAR T-Cell Program
has a superb facility designed specifically to advance the
development of promising T-cell therapies. We are excited to
progress our INB-400 program towards a Company sponsored
investigational new drug (IND) application that will advance this
program into Phase 2.”
“Since their inception, the UofL Health – Brown Cancer Center
GMP Manufacturing Facility and the Dunbar CAR T-Cell Program at the
University of Louisville have been committed to bringing these
lifesaving immunotherapies to adult and pediatric cancer patients
throughout Kentucky and the region. This manufacturing agreement
with IN8bio will allow our facility to provide these groundbreaking
immunotherapies for patients throughout the United States,” said
James Cripps, Ph.D., manager of the Brown Cancer Center GMP
Manufacturing Facility.
“It is with the greatest excitement for the Evan Dunbar
Foundation CAR T-cell program that we announce that IN8bio has
joined us in this world-changing research,” said Thomas E. Dunbar.
“Along with the amazing work at the University of Louisville, I
believe that IN8bio can join with us in achieving the goals of the
Evan Dunbar Foundation CAR T-cell program. These three
organizations working together will create trials that will impact
hundreds, if not thousands of patients.
“I am so honored and pleased to see our vision of cancer
research come closer to trial and real-time treatment of patients.
This has been my lifelong dream and is the most fulfilling way to
honor my son, Evan Dunbar, my wife, Dr. Stephanie Altobellis, my
father, Wallace and the rest of my family.”
The Dunbar CAR T-Cell Program was created in 2019 thanks to a
$1-million pledge from Dunbar, a Louisville resident. It is a
state-of-the-art manufacturing facility custom built to collaborate
with the biotech industry to advance cutting-edge cell and gene
therapies. T-cell therapies utilize immune cells that are
genetically modified to fight cancer. The modified cells are
infused into the patient with the goal of fighting the cancer and
creating long-term immunity to delay or prevent recurrence. The
facility is led by and staffed with experts in cell therapy
manufacturing who will work closely with the IN8bio team to produce
the DeltEx DRI cells for INB-400.
Facility team in the of the UofL Health - Brown
Cancer Center GMP Manufacturing Facility at the University of
Louisville.
About INB-400INB-400 is IN8bio’s DeltEx
Allogeneic DRI. INB-400 expands the application of DRI gamma-delta
T cells into other solid tumor types through the development of
allogeneic or “off-the-shelf” DeltEx DRI therapies. INB-400 will be
a genetically modified DeltEx allogeneic candidate in both newly
diagnosed and relapsed refractory glioblastoma.
About IN8bioIN8bio is a clinical-stage
biopharmaceutical company focused on discovering, developing and
commercializing gamma-delta T cell product candidates for solid and
liquid tumors. Gamma-delta T cells are a specialized population of
T cells that possess unique properties, including the ability to
differentiate between healthy and diseased tissue. IN8bio’s DeltEx
platform employs allogeneic, autologous, iPSC, and genetically
modified approaches to develop cell therapies that synergize with
chemotherapy to effectively identify and eradicate tumor cells.
IN8bio is currently conducting two investigator-initiated Phase
1 clinical trials for its lead gamma-delta T cell product
candidates: INB-200 for the treatment of newly diagnosed
glioblastoma and INB-100 for the treatment of patients with
leukemia undergoing hematopoietic stem cell transplantation. IN8bio
also has a broad portfolio of preclinical programs focused on
addressing other solid tumor types. For more information about
IN8bio and its programs, please visit www.IN8bio.com.
Forward Looking StatementsThis press release
may contain forward-looking statements made pursuant to the safe
harbor provisions of the Private Securities Litigation Reform Act
of 1995. These statements may be identified by words such as
“aims,” “anticipates,” “believes,” “could,” “estimates,” “expects,”
“forecasts,” “goal,” “intends,” “may,” “plans,” “possible,”
“potential,” “seeks,” “will” and variations of these words or
similar expressions that are intended to identify forward-looking
statements, although not all forward-looking statements contain
these words. Forward-looking statements in this press release
include, but are not limited to, statements regarding the success
of IN8bio’s partnership with UofL Health - Brown Cancer Center GMP
Manufacturing Facility at the University of Louisville; the timing
of initiation, progress and scope of clinical trials for IN8bio’s
product candidates; the potential of IN8bio’s DeltEx platform to
discover and develop innovative product candidates, including
iPSC-derived cell therapies; and IN8bio’s ability to achieve
planned milestones, including data readouts from its trials and
plans to file an IND application. IN8bio may not actually achieve
the plans, intentions or expectations disclosed in these
forward-looking statements, and you should not place undue reliance
on these forward-looking statements. Actual results or events could
differ materially from the plans, intentions and expectations
disclosed in these forward-looking statements as a result of
various factors, including: risks to site initiation, clinical
trial commencement, patient enrollment and follow-up, as well as
IN8bio’s ability to meet anticipated deadlines and milestones,
presented by the ongoing COVID-19 pandemic; uncertainties inherent
in the initiation and completion of preclinical studies and
clinical trials and clinical development of IN8bio’s product
candidates; the risk that IN8bio may not realize the intended
benefits of its DeltEx platform; availability and timing of results
from preclinical studies and clinical trials; whether the outcomes
of preclinical studies will be predictive of clinical trial
results; whether initial or interim results from a clinical trial
will be predictive of the final results of the trial or the results
of future trials; the risk that trials and studies may be delayed
and may not have satisfactory outcomes; potential adverse effects
arising from the testing or use of IN8bio’s product candidates;
expectations for regulatory approvals to conduct trials or to
market products; IN8bio’s reliance on third parties, including
licensors and clinical research organizations; and other important
factors, any of which could cause our actual results to differ from
those contained in the forward-looking statements, are described in
greater detail in the section entitled “Risk Factors” in our
Quarterly Report on Form 10-Q filed with the Securities and
Exchange Commission (SEC) on August 12, 2022, as well as in other
filings IN8bio may make with the SEC in the future. Any
forward-looking statements contained in this press release speak
only as of the date hereof, and IN8bio expressly disclaims any
obligation to update any forward-looking statements contained
herein, whether because of any new information, future events,
changed circumstances or otherwise, except as otherwise required by
law.
About the University of Louisville:Founded in
1798 as one of the nation’s first city-owned, public universities,
the University of Louisville (UofL) is a vital ecosystem that
creates thriving futures for students, our community and society.
As one of only 79 universities in the United States to earn
recognition by the Carnegie Foundation as both a Research 1 and a
Community Engaged university, we impact lives in areas of student
success and research and innovation, while our dynamic connection
with our local and global communities provides unparalleled
opportunities for students and citizens both. The university serves
as an engine that powers Metro Louisville and the commonwealth and
as a classroom for UofL’s more than 23,000 students, who benefit
from partnerships with top employers and a wide range of community
service opportunities.
Get more news from UofL delivered straight to your inbox every
Thursday evening by signing up for our UofL News Weekly Roundup
here: uoflalumni.org/uoflnews-signup.
About UofL Health: UofL Health is a fully
integrated regional academic health system with seven hospitals,
four medical centers, Brown Cancer Center, Eye Institute, nearly
200 physician practice locations, and more than 800 providers in
Louisville and the surrounding counties, including southern
Indiana. Additional access to UofL Health is provided through a
partnership with Carroll County Memorial Hospital.
With more than 12,000 team members – physicians, surgeons,
nurses, pharmacists and other highly-skilled health care
professionals, UofL Health is focused on one mission: to transform
the health of communities we serve through compassionate,
innovative, patient-centered care.
Company Contact:IN8bio, Inc.Patrick McCall+ 1
646.600.6GDT (6438)info@IN8bio.com
Investors & Media Contact: Argot
PartnersIN8bio@argotpartners.com
University of Louisville Contact:Jill
Scoggins+1 502.650.2624Jill.scoggins@louisville.edu
A photo accompanying this announcement is available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/5340530f-9020-4612-aeb7-ef243b175770
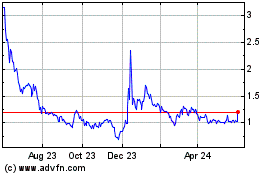
IN8bio (NASDAQ:INAB)
Historical Stock Chart
From Sep 2024 to Oct 2024
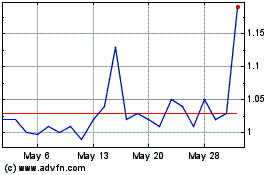
IN8bio (NASDAQ:INAB)
Historical Stock Chart
From Oct 2023 to Oct 2024