Emergent BioSolutions Shares Rise 16% After Cyfendus Approved for Post-Exposure Prophylaxis Use
July 21 2023 - 6:14AM
Dow Jones News
By Chris Wack
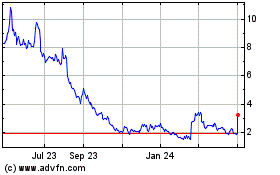
Emergent BioSolutions shares were up 16% to $8.22 in premarket
trading after the company said late Thursday that the U.S. Food and
Drug Administration has approved Cyfendus for post-exposure
prophylaxis of disease following suspected or confirmed exposure to
Bacillus anthracis in persons 18 through 65 years old.
The stock hit its 52-week low of $6.77 on July 6, and is down
78% as of Thursday's close.
The company said the efficacy of the Cyfendus vaccine for
post-exposure prophylaxis is based solely on studies in animal
models of inhalational anthrax.
Emergent BioSolutions said Cyfendus has been demonstrated that
by using an additional adjuvant, two doses administered over 14
days elicit protective levels of immune response, which can be
especially important in response to a large-scale public health
emergency involving anthrax.
In December 2018, Cyfendus was the subject of a pre-emergency
use authorization package submitted to the FDA. In 2019, the U.S.
government began procuring this product for national preparedness
efforts.
Write to Chris Wack at chris.wack@wsj.com
(END) Dow Jones Newswires
July 21, 2023 06:59 ET (10:59 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
Emergent Biosolutions (NYSE:EBS)
Historical Stock Chart
From May 2024 to Jun 2024
Emergent Biosolutions (NYSE:EBS)
Historical Stock Chart
From Jun 2023 to Jun 2024