TIDMDEST
RNS Number : 5053V
Destiny Pharma PLC
09 April 2019
Destiny Pharma plc
("Destiny Pharma" or "the Company")
Audited results for the year ended 31 December 2018
Well-funded and on track
Brighton, United Kingdom - 9 April 2019 - Destiny Pharma (AIM:
DEST), a clinical stage biotechnology company focused on the
development of novel anti-microbial drugs which address the global
problem of anti-microbial resistance (AMR), announces its audited
financial results for the year ended 31 December 2018.
Financial highlights
-- Strong cash position with cash and term deposits at 31
December 2018 of GBP12.1 million (2017: GBP16.7 million)
-- Increase in R&D expenditure to GBP3.5 million (2017:
GBP0.8 million) due to planned clinical development costs
Operational highlights
XF-73 for prevention of post-surgical infections
-- Phase 2b protocol finalised and 200 patient study now starting April 2019
-- Results due around year-end 2019
-- US market research report confirms clinical need and
attractive target product profile of XF-73 nasal - clear unmet need
and payer support for proposed pricing supports large market
potential
-- US Investigational New Drug (IND) application opened for lead
clinical programme XF-73 nasal - a novel anti-microbial being
developed as a preventative treatment
-- US Food and Drug Administration (FDA) Fast Track designation granted for XF-73
-- Clarification, through dialogue with the FDA, of the Phase 1
and Phase 2b clinical trials programme
-- Successful completion of required Phase 1 dermal safety studies to enable start of Phase 2b
Earlier pipeline and research grants
-- New dermal infection clinical programme initiated with XF-73
targeting diabetic foot ulcer infections
-- Award of two research grants in collaborations with Aston and Southampton Universities
-- Patent portfolio expanded with the grant of Canadian XF-biofilm patent
Corporate highlights
-- Board and executive management team strengthened with the
appointment of Jesus Gonzales MD as Chief Medical Officer and Shaun
Claydon as Chief Financial Officer
Post-period highlights
-- Award of UK China AMR grant of up to GBP1.6 million to
examine XF compounds potential against dermal and ocular
infections
-- UK government announces new 5 and 20 year plans to address
AMR and to support novel drug development addressing AMR and
improved financial incentives for companies bringing such new drugs
to market
-- Nick Rodgers appointed as Chairman replacing Sir Nigel Rudd
who stepped down after 15 years with the Company
Neil Clark, Chief Executive Officer of Destiny Pharma,
commented:
"We have made significant progress in the first full year
following our IPO in September 2017, delivering on key targets set
out at the time, including a number of clinical development
objectives, and the Company remains well funded to H2 2020.
"Our lead clinical candidate, XF-73 nasal is being developed as
a preventative treatment reducing the carriage of Staphylococcus
aureus with the intention of preventing post-surgical hospital
infections; a $1 billion peak sales market opportunity. During
April 2019 we will initiate the important Phase 2b clinical trial
in this setting and will complete recruitment later this year.
"Whilst our main focus is on our lead asset we are also
continuing to progress our earlier XF pipeline having won three
grants to support this workstream. There is continuing
international support for the development of novel anti-infective
drugs that address the issue of anti-microbial resistance and
Destiny Pharma's unique platform is well-positioned to meet this
global need."
This announcement has been released by Neil Clark, CEO, on
behalf of the Company
For further information, please contact:
Destiny Pharma plc
Neil Clark, CEO
Shaun Claydon, CFO
pressoffice@destinypharma.com
+44 (0)1273 704 440
FTI Consulting
Simon Conway / Victoria Foster Mitchell
destinypharma@fticonsulting.com
+44 (0) 20 3727 1000
Cantor Fitzgerald Europe (Nominated Adviser and Joint
Broker)
Philip Davies / Will Goode, Corporate Finance
Andrew Keith, Healthcare Equity Sales
+44 (0)20 7894 7000
finnCap Ltd (Joint Broker)
Geoff Nash /Kate Bannatyne, Corporate Finance
Alice Lane, Corporate Broking
+44 (0)20 7220 0500
About Destiny Pharma
Destiny Pharma is an established, clinical stage, innovative
biotechnology company focused on the development of novel medicines
that represent a new approach to the treatment of infectious
disease. These potential new medicines are being developed to
address the need for new drugs for the prevention and treatment of
life-threatening infections caused by antibiotic-resistant
bacteria, often referred to as "superbugs". Tackling anti-microbial
resistance has become a global imperative recognised by the World
Health Organisation (WHO) and the United Nations, as well as the G7
and the G20 countries. For further information, please visit
https://www.destinypharma.com
Forward looking statements
Certain information contained in this announcement, including
any information as to the Group's strategy, plans or future
financial or operating performance, constitutes "forward-looking
statements". These forward looking statements may be identified by
the use of forward-looking terminology, including the terms
"believes", "estimates", "anticipates", "projects", "expects",
"intends", "aims", "plans", "predicts", "may", "will", "seeks"
"could" "targets" "assumes" "positioned" or "should" or, in each
case, their negative or other variations or comparable terminology,
or by discussions of strategy, plans, objectives, goals, future
events or intentions. These forward-looking statements include all
matters that are not historical facts. They appear in a number of
places throughout this announcement and include statements
regarding the intentions, beliefs or current expectations of the
Directors concerning, among other things, the Group's results of
operations, financial condition, prospects, growth, strategies and
the industries in which the Group operates. The directors of the
company believe that the expectations reflected in these statements
are reasonable, but may be affected by a number of variables which
could cause actual results or trends to differ materially. Each
forward-looking statement speaks only as of the date of the
particular statement. By their nature, forward-looking statements
involve risks and uncertainties because they relate to events and
depend on circumstances that may or may not occur in the future or
are beyond the Group's control. Forward looking statements are not
guarantees of future performance. Even if the Group's actual
results of operations, financial condition and the development of
the industries in which the Group operates are consistent with the
forward-looking statements contained in this document, those
results or developments may not be indicative of results or
developments in subsequent periods.
Chief Executive Officer's Statement
Operational review
Destiny Pharma is an established, clinical stage, innovative
biotechnology company focused on the development of novel medicines
that represent a new approach to the treatment of infectious
disease. These potential new medicines are being developed to
address the need for new drugs for the prevention and treatment of
life-threatening infections caused by anti-microbial resistance
(AMR). AMR poses a threat to public health and is of serious
concern to the WHO. Lord O'Neill's Independent Review on AMR,
published in May 2016, predicts ten million deaths and an estimated
$100 trillion cost by 2050. The UK government's recent update on
their 5 and 20 year AMR plans also confirmed yet again the
importance of addressing AMR and innovation in the development of
novel anti-infectives.
The Company's lead asset, XF-73, has been developed from Destiny
Pharma's novel, antimicrobial "XF" drug platform. Unlike most
antibiotics, XF drugs have not been seen to generate bacterial
resistance in industry-standard microbiology tests to date and
therefore have significant potential to address the global threat
of AMR. XF-73 has been shown to kill bacteria very rapidly and
therefore may be an effective new treatment in the reduction of
bacterial infections in hospital patients, including those caused
by methicillin resistant Staphylococcus aureus (MRSA). XF-73 is
administered topically as a nasal gel whereby it reduces the nasal
carriage of the bacteria Staphylococcus aureus, which is the source
of many post-surgical bacterial infections. Approximately a third
of all patients across the world have this nasal carriage as they
enter surgery and it has the potential to be a very valuable market
due to the millions of surgical procedures carried out each
year.
We believe XF-73 is clearly differentiated from traditional
antibiotics and many current anti-infective drugs in development in
that the XF approach is prophylactic and follows the
well-established medical truth that "prevention is better than
cure". The XF's target product profile also addresses the key issue
of AMR. This belief is supported by feedback from our market
research targeting physicians, pharmacists and payers in the US who
are responsible for managing hospital infections and the associated
cost implications. This research also supports our proposed pricing
strategies for XF-73 nasal gel as a new hospital product.
Executive management team strengthened, new Chairman
appointed
In 2018, Jesús M González Moreno, M.D was appointed as Chief
Medical Officer and Shaun Claydon joined as Chief Finance Officer.
Jesus is an infectious disease expert with more than 11 years'
experience of working within global pharmaceutical and
biotechnology companies to design, coordinate and execute clinical
development plans for anti-infective drug candidates. His
experience spans from early and late clinical development to
medical affairs and preparation for marketing authorisation
submissions. Shaun is an accomplished corporate financier and
Chartered Accountant with over 16 years board level experience
within the biotechnology sector.
Nick Rodgers, a Non-Executive Director, became Chairman
following Sir Nigel Rudd's departure on 31 December 2018 after 15
years of service. Nick has considerable Board experience in both
public and private growth companies, particularly those in the life
science sector, as well as a background as a successful corporate
financier and investment banker.
Phase 2b clinical trial starting Q2 in 200 patients undergoing
surgical procedures
During 2018, Destiny Pharma continued to progress its clinical
pipeline and has finalised the Phase 2b clinical development plans
for its lead asset, XF-73, for the prevention of post-surgical
infections such as MRSA. Destiny Pharma is now starting the Phase
2b with patient recruitment expected to be completed by the end of
2019. The trial is a multi-centre, randomised, blinded,
placebo-controlled study of multiple applications of a single
concentration of XF-73 nasal gel to assess the microbiological
effect of XF-73 on commensal Staphylococcal aureus nasal carriage
in patients scheduled for surgical procedures deemed to be at high
risk of post-operative Staphylococcal aureus infection. The study
is larger than originally planned and reflects the expert advice
taken on statistical parameters, microbiological end-points and
delivering the most complete study possible. The larger study also
has the advantage of exposing even more sites and patients in the
hospital setting to the XF-73 nasal gel.
The Phase 2b study design is closely related to the successful
2016 clinical trial, which was funded by the National Institute of
Allergy and Infectious Disease (part of the US National Institute
of Health) and demonstrated the clinical efficacy of XF-73 versus
placebo in reducing nasal Staphylococcus aureus carriage in healthy
volunteers.
In advance of finalising the Phase 2b design, Destiny Pharma
opened a US IND application for XF-73, which is a key regulatory
prerequisite for conducting clinical trials in the US. This was
followed by the FDA granting Fast Track designation for XF-73, for
the prevention of post-surgical staphylococcal infections in March
2018. The clinical programme for XF-73 was further refined
following discussions with the FDA and the required Phase 1 dermal
safety study, looking at potential skin irritation of XF-73 gel
formulation was completed successfully with XF-73 having a very
benign profile with low cumulative irritancy scores similar to a
"non-irritant" such as water in our studies. This excellent dermal
safety data was also very useful in clarifying the pathway for our
new dermal infection programme.
In parallel with the clinical work, good progress has made with
improving the efficiency of the synthesis pathway and scale up of
XF-73 to improve further the costs of goods. Work is also
progressing on possible final product presentations to enhance the
ease of use in the hospital setting. The Company's plan is to build
a Phase 3 ready package consisting of the agreed clinical studies
for the XF-73 nasal gel formulation.
XF-73 dermal - new programme in multi-billion-dollar dermal
infection market
Following an extensive review of dermal infection indications
that XF-73 could potentially address, the Company will initially
focus on developing XF-73 as a new treatment for diabetic foot
ulcer infections (DFUs). Driven by the growing number of diabetics
and associated complications such as infected DFUs, this represents
a significant market opportunity for XF-73. As with all
anti-infectives, AMR is also a concern within this market. There is
no dominant treatment for DFUs and specialist physicians are
therefore working to find better treatment options, including
topical formulations. In addition, the target product profile of
XF-73 tested favourably with dermal clinicians looking for better
treatments for the smaller market for burns/wound infections. The
Phase 1 skin irritation studies completed in the period were the
first data supporting the use of XF-73 on damaged skin and as
stated above the "non-irritant" profile is very promising.
Destiny Pharma is now assessing new formulations of XF-73 for
infections in DFUs and burns wounds, which is estimated to be a
$0.5 billion global opportunity for the Company based on the
incidence of such infections, the costs of the associated medical
care and a realistic product pricing of XF-73 in this new
market.
Research collaborations and expansion of patent portfolio
Work on earlier programmes such as ventilator associated
pneumonia (VAP), biofilms and other indications will be as research
projects, including academic or commercial collaborations and grant
funded programmes.
In line with this strategy, Destiny Pharma signed a research
collaboration agreement with Aston University in July 2018 to
examine novel compounds from the XF-platform and assess their
potential to prevent, control and eradicate dangerous bacteria in
biofilms. Serious infections are frequently caused and exacerbated
by biofilms where bacteria can hide and be protected from
traditional anti-infective agents. XF compounds have already shown
efficacy in biofilm models and this research project will explore
the potential further, including looking at the
mechanisms-of-action.
A second grant was awarded in November 2018 with Southampton
University. The project is examining the use of the Company's novel
XF compounds to prevent, control and eradicate chronic clinical
infections with underlying biofilm involvement, such as those in
diabetic foot ulcers and cystic fibrosis. The NBIC funded
collaboration plans to expand on this data using laboratory and
clinical microbial biofilm models and the expertise of the team at
the University of Southampton's Faculty of Environmental and Life
Sciences, who have established ex vivo biofilm model systems and
access to clinical infection samples that will be utilised in the
collaboration.
A third grant was awarded in early 2019, with funding of up to
GBP1.6 million from a collaboration established under the UK-China
AMR grant fund set up by Innovate UK and the Department of Health
and Social Care with the Chinese Ministry of Science and
Technology. The two-year project will examine the use of the
Company's novel XF drugs (XF-73, XF-70 and DPD-207) to prevent,
control, and eradicate life threatening bacteria or "superbugs"
without generating resistance especially in the treatment of dermal
and ocular infections. The research work will be carried out by
Destiny Pharma's team in collaboration with expert groups at
Cardiff University's School of Dentistry and College of Biomedical
and Life Sciences, led by Professor David Williams, and a team at
Tianjin Medical University, China.
All three of these grant funded projects are up and running and
we are looking forward to their progress and the potential to
identify new product opportunities for the XF platform. Destiny
Pharma also continued to strengthen its patent estate, with the
grant of the XF biofilm patent in Canada in February 2018, bringing
the total number of XF platform granted patents to 95.
Globally recognised issue that urgently needs addressing
International reviews and initiatives continued to take place in
support of tackling the global issue of antibiotic resistance.
These have included discussions and announcements at G7, G20 and
United Nations meetings, as well as the World Health Organisation's
GARDP and DRIVE-AB, an EU/industry partnership. Mechanisms to
support the clinical development of new anti-infectives proposed
include additional "push" grant incentives, as well as significant
"pull" market entry rewards. This was reiterated at the World
Economic Summit in Davos in January 2018 and also are highlighted
as key aims under the UK Government's recent announcement of its
new 5 and 20 year AMR plan. Destiny Pharma will continue to
contribute to policy development and will apply for appropriate
grants and other non-dilutive funding where they fit with the
Company's research and development plans.
Outlook
Destiny Pharma is well funded to develop its lead asset, XF-73,
through the proposed US clinical Phase 2b programme, delivering a
robust package for partnering and/or further development into Phase
3, the final stage of clinical development. The Company has had a
strong 2018, having opened a US IND and received Fast Track
designation for XF-73. Subsequent discussions with the FDA
clarified the clinical pathway for XF-73 and the Company completed
the required Phase 1 dermal safety studies successfully. The Phase
2b study is now about to start. Importantly, market analysis
continues to support the clinical need and commercial opportunity
for XF-73 in the prevention of post-surgery hospital infections,
such as MRSA, which is estimated to be over a $1 billion market
opportunity.
Funds, augmented by three grant awards, are also being used to
develop new clinical candidates from the Company's pre-clinical
pipeline and we also announced a new dermal infection clinical
programme. The Board is confident that the Company is well funded
to execute on its business strategy and to progress its lead and
follow-on programmes through the planned studies in 2019 and 2020.
There is continuing international support for the development of
novel anti-infective drugs that address the issue of anti-microbial
resistance and Destiny Pharma's unique platform is very
well-positioned to meet this global need.
Neil Clark
Chief Executive Officer
9 April 2019
Chief Financial Officer's Statement
Financial review
Following the company's successful listing on AIM in September
2017, we increased activity across our scientific and clinical
programmes during 2018. Funds raised at IPO were utilised to
advance our lead programme toward commencement of Phase 2b trials
and to develop our earlier programmes, resulting in a significant
increase in R&D spend over the prior year. We also increased
headcount during the year to support this increase in activity.
We were also pleased to announce research collaborations during
the year, enabling the company to further develop its earlier
programmes. Grant funding associated with these research
collaborations will be received from 2019 onwards.
Revenue
Destiny Pharma is a clinical stage research and development
company, and did not generate any revenue during the period.
Administrative expenses
Administrative expenses, which excludes the share-based payment
charge of GBP0.7 million (2017: GBP0.7 million), during the period
amounted to GBP5.3 million (2017: GBP2.5 million). Included within
this total are R&D costs totalling GBP3.5 million (2017: GBP0.8
million), which reflect the increase in activity with regard to our
scientific and clinical programmes, particularly during the second
half of the year. The remaining increase over 2017 of GBP0.6
million (ignoring one-off AIM costs of GBP0.5 million in 2017) are
due to increased staff costs associated with increases in headcount
and other operational costs, which were partly offset by foreign
exchange gains of GBP0.1 million during the year.
Taxation
The Company's research and development activities are eligible
for the UK research and development small or medium-sized
enterprise ("R&D tax credit") scheme, which provides additional
taxation relief for qualifying expenditure on R&D activities,
with an option to surrender a portion of tax losses arising from
qualifying activities in return for a cash payment from HM Revenue
& Customs ("HMRC"). The Company received a repayment of GBP0.23
million in respect of the R&D tax credit claimed in respect of
the year ended 31 December 2017. The R&D tax credit receivable
in the balance sheet of GBP0.84 million is an estimate of the cash
repayment the Company expects to qualify for in respect of
activities during the year ended 31 December 2018. However, as at
the date of this report these amounts have not yet been agreed with
HMRC.
Loss per share
Basic and diluted loss per share for the year was 11.9 pence
(2017: 8.4 pence).
Cash, cash equivalents and term deposits
The Company's cash, cash equivalents and term deposits at the
year-end totalled GBP12.1 million (2017: GBP16.7 million).
The net cash outflow from operating activities in 2018 was
GBP4.7 million against an operating loss of GBP6.0 million, with
the major reconciling items being the non-cash charge for
share-based payments of GBP0.7 million, the R&D credit received
of GBP0.2 million and other net movements in working capital of
GBP0.4 million.
Outlook
The Board believes the Company remains well funded to execute on
its business strategy and to progress its lead and follow-on
programmes in 2019 and 2020.
Shaun Claydon
Chief Financial Officer
9April 2019
Statement of comprehensive income
For the year ended 31 December 2018
Year ended Year ended
31 December 31 December
2018 2017
Notes GBP GBP
-------------------------------------------------------------------------- ----- ----------- -----------
Continuing operations
Revenue - -
Administrative expenses (5,346,170) (2,511,871)
Share option charge (737,687) (709,979)
-------------------------------------------------------------------------- ----- ----------- -----------
Operating loss (6,083,857) (3,221,850)
Finance income 4 75,999 10,459
Loss before tax (6,007,858) (3,211,391)
Taxation 5 841,144 233,908
-------------------------------------------------------------------------- ----- ----------- -----------
Loss and total comprehensive loss for the year from continuing operations (5,166,714) (2,977,483)
-------------------------------------------------------------------------- ----- ----------- -----------
Loss per share - pence
Basic 6 (11.9)p (8.4)p
Diluted 6 (11.9)p (8.4)p
-------------------------------------------------------------------------- ----- ----------- -----------
Statement of financial position
as at 31 December 2018
As at As at
31 December 31 December
2018 2017
Notes GBP GBP
------------------------------ ----- ----------- -----------
Assets
Non-current assets
Property, plant and equipment 30,421 22,313
------------------------------ ----- ----------- -----------
Non-current assets 30,421 22,313
------------------------------ ----- ----------- -----------
Current assets
Trade and other receivables 7 930,759 277,126
Cash and cash equivalents 8 7,060,821 11,724,037
Other financial assets 9 5,000,000 5,000,000
Prepayments 36,406 59,641
------------------------------ ----- ----------- -----------
Current assets 13,027,986 17,060,804
------------------------------ ----- ----------- -----------
Total assets 13,058,407 17,083,117
------------------------------ ----- ----------- -----------
Equity and liabilities
Equity
Called-up share capital 10 435,626 435,626
Share premium 17,292,284 17,292,284
Retained earnings (5,471,295) (1,042,268)
------------------------------ ----- ----------- -----------
Shareholders' equity 12,256,615 16,685,642
------------------------------ ----- ----------- -----------
Current liabilities
Trade and other payables 11 801,792 397,475
------------------------------ ----- ----------- -----------
Current liabilities 801,792 397,475
------------------------------ ----- ----------- -----------
Total equity and liabilities 13,058,407 17,083,117
------------------------------ ----- ----------- -----------
Statement of changes in equity
for the year ended 31 December 2018
Called-up Share Retained
share capital premium earnings Total
GBP GBP GBP GBP
-------------------------------- ------------- ------------ ------------ -----------
1 January 2017 638 18,335,074 (16,791,296) 1,544,416
Reduction of capital (note 12) - (18,016,532) 18,016,532 -
Bonus issue of shares (note 12) 318,542 (318,542) - -
Issue of share capital 116,446 18,165,573 - 18,282,019
Cost of share issue - (873,289) - (873,289)
Total comprehensive loss - - (2,977,483) (2,977,483)
Share option charge - - 709,979 709,979
-------------------------------- ------------- ------------ ------------ -----------
31 December 2017 435,626 17,292,284 (1,042,268) 16,685,642
Total comprehensive loss - - (5,166,714) (5,166,714)
Share option charge - - 737,687 737,687
-------------------------------- ------------- ------------ ------------ -----------
31 December 2018 435,626 17,292,284 (5,471,295) 12,256,615
-------------------------------- ------------- ------------ ------------ -----------
Statement of cash flows
for the year ended 31 December 2018
Year ended Year ended
31 December 31 December
2018 2017
GBP GBP
-------------------------------------------------------- ----------- -----------
Cash flows from operating activities
Loss before income tax (6,007,858) (3,211,391)
Depreciation charges 9,663 2,077
Share option charge 737,687 709,979
Finance income (75,999) (10,459)
Increase in trade and other receivables and prepayments (23,162) (77,935)
Increase in trade and other payables 404,317 242,736
Tax received 233,908 191,578
-------------------------------------------------------- ----------- -----------
Net cash outflow from operating activities (4,721,444) (2,153,415)
-------------------------------------------------------- ----------- -----------
Cash flows from investing activities
Purchase of tangible fixed assets (17,771) (23,230)
Purchase of other financial assets - (5,000,000)
Interest received 75,999 10,459
-------------------------------------------------------- ----------- -----------
Net cash inflow/(outflow) from investing activities 58,228 (5,012,771)
-------------------------------------------------------- ----------- -----------
Cash flows from financing activities
New shares issued net of issue costs - 17,408,730
-------------------------------------------------------- ----------- -----------
Net cash inflow from financing activities - 17,408,730
-------------------------------------------------------- ----------- -----------
Net (decrease)/increase in cash and cash equivalents (4,663,216) 10,242,544
Cash and cash equivalents at the beginning of the year 11,724,037 1,481,493
-------------------------------------------------------- ----------- -----------
Cash and cash equivalents at the end of the year 7,060,821 11,724,037
-------------------------------------------------------- ----------- -----------
Notes to the financial statements
1. Corporate information
Destiny Pharma plc (the "company") was incorporated and
domiciled in the UK on 4 March 1996 with registration number
03167025. The company's registered office is located at Unit 36,
Sussex Innovation Centre, Science Park Square, Falmer, Brighton BN1
9SB.
The company is engaged in the discovery, development and
commercialisation of new antimicrobials that have unique properties
to improve outcomes for patients and the delivery of medical care
into the future.
2. Basis of preparation
The financial statements have been prepared in accordance with
International Financial Reporting Standards ("IFRSs") as adopted by
the European Union. The financial statements have been prepared
under the historical cost convention.
The company's financial statements have been presented in pound
sterling ("GBP"), being the functional and presentation currency of
the company.
Standards and interpretations issued but not yet applied
At the date of authorisation of the company's financial
statements, certain new standards, amendments and interpretations
to existing standards have been published by the International
Accounting Standards Board but are not yet effective and have not
been adopted early by the company. All relevant standards,
amendments and interpretations to existing standards will be
adopted in the company's accounting policies in the first period
beginning on or after the effective date of the relevant
pronouncement.
The Directors do not anticipate that the adoption of these
standards, amendments and interpretations will have a material
impact on the company's financial statements in the periods of
initial application.
3. Segment reporting
The chief operating decision-maker is considered to be the Board
of Directors of the company. The chief operating decision-maker
allocates resources and assesses performance of the business and
other activities at the operating segment level.
The chief operating decision-maker has determined that the
company has one operating segment, the development and
commercialisation of pharmaceutical formulations. All activities
take place in the United Kingdom.
4. Net finance income
31 December 31 December
2018 2017
GBP GBP
------------------------- ----------- -----------
Finance income
Deposit account interest 75,999 10,459
------------------------- ----------- -----------
5. Income tax
31 December 31 December
2018 2017
GBP GBP
-------------------------------------------------------------------------- ----------- -----------
Research and development tax credits based on costs in the financial year (841,144) (233,908)
-------------------------------------------------------------------------- ----------- -----------
Tax reconciliation
31 December 31 December
2018 2017
GBP GBP
----------------------------------------------------------------------------- ----------- -----------
Loss before tax (6,007,858) (3,221,850)
Loss before tax multiplied by the UK corporation tax rate of 19% (2017: 20%) (1,141,493) (644,370)
Effects of:
Non-deductible expenditure 148,637 99,969
R&D enhanced expenditure (622,976) (132,210)
Lower tax rate on R&D losses 261,044 38,576
Tax losses carried forward 513,644 404,127
----------------------------------------------------------------------------- ----------- -----------
Total tax credit on loss (841,144) (233,908)
----------------------------------------------------------------------------- ----------- -----------
There were no tax charges in the period. There are tax losses
available to carry forward amounting to approximately GBP13.7
million (2017: GBP12.8 million), which includes GBP0.7 million
(2017: GBP1.5 million) in respect of tax deductions on share
options. A deferred tax asset on losses is not recognised in the
accounts due to the uncertainty of future profits against which
they will be utilised.
6. Loss per ordinary share
The calculation for loss per ordinary share (basic and diluted)
for the relevant period is based on the earnings after income tax
attributable to equity shareholders for the period. As the company
made losses during the period, there are no dilutive potential
ordinary shares in issue, and therefore basic and diluted loss per
share are identical. The calculation is as follows:
31 December 31 December
2018 2017
GBP GBP
----------------------------------------------- ----------- -----------
Loss for the year attributable to shareholders (5,166,714) (2,977,483)
----------------------------------------------- ----------- -----------
Weighted average number of shares 43,562,598 35,253,765
----------------------------------------------- ----------- -----------
Loss per share - pence
- Basic and diluted (11.9)p (8.4)p
----------------------------------------------- ----------- -----------
7. Trade and other receivables
31 December 31 December
2018 2017
GBP GBP
--------------------------------------- ----------- -----------
Other debtors 89,615 43,218
Research and development tax repayment 841,144 233,908
--------------------------------------- ----------- -----------
930,759 277,126
--------------------------------------- ----------- -----------
8. Cash and cash equivalents
31 December 31 December
2018 2017
GBP GBP
----------------------- ----------- -----------
Cash and bank balances 7,060,821 11,724,037
----------------------- ----------- -----------
9. Other financial assets
31 December 31 December
2018 2017
GBP GBP
-------------------------------------------------------- ----------- -----------
Term deposits with maturities greater than three months 5,000,000 5,000,000
-------------------------------------------------------- ----------- -----------
10. Share capital
31 December 31 December
2018 2017
Ordinary shares of GBP0.01 each Number Number
------------------------------------------------------ ----------- -----------
Authorised(1) n/a n/a
Allotted and fully paid
At 1 January 43,562,598 63,836
Bonus issue of shares during the year (see note [18]) - 31,854,164
Issued for cash during the year - 11,644,598
------------------------------------------------------ ----------- -----------
At 31 December 43,562,598 43,562,598
------------------------------------------------------ ----------- -----------
(1) During the year ended 31 December 2017 the company adopted
new Articles of Association, which do not require the company to
have authorised share capital.
31 December 31 December
2018 2017
GBP GBP
------------------------ ----------- -----------
Authorised n/a n/a
Allotted and fully paid 435,626 435,626
------------------------ ----------- -----------
31 December 31 December
2018 2017
GBP GBP
---------------------- ----------- -----------
Share premium account 17,292,284 17,292,284
---------------------- ----------- -----------
Each ordinary share ranks pari passu for voting rights,
dividends and distributions and return of capital on winding
up.
Share options
The expense arising from share-based payment transactions
recognised in the year ended 31 December 2018 was GBP737,687 (year
ended 31 December 2017: GBP709,979).
The company's share-based payment arrangements are summarised
below.
Share option schemes
As part of its strategy for executive and key employee
remuneration, the company issued share options under two schemes
established on 15 November 2000 - an Unapproved Scheme and an EMI
Scheme (the "Old Schemes"). During 2017, the company established
two new share option schemes - the LTIP Employee Scheme and the
LTIP Non-Employee Scheme, both of which were established on 18
April 2017 (the "New Schemes"). Awards under the LTIP Employee
Scheme are made to qualifying employees and in accordance with
Schedule 5 of the Income Tax (Earnings and Pensions) Act 2003 so
that, provided awards are within the qualifying limits, the awards
qualify as EMI options. Any awards under the LTIP Employee Scheme
which do not fall within the qualifying limits do not qualify as
EMI options. Awards under the LTIP Non-Employee Scheme do not
qualify as EMI options.
The principal terms of the company's share option schemes are as
follows:
Unapproved Scheme
Options are granted at the discretion of the Directors. The
price per share to be paid on exercise of an option will be the
market value as agreed with the Share Valuation Division of HM
Revenue & Customs at the time of the grant of the option and as
detailed in the option certificate. Options may be exercised three
years from the date of grant and lapse on the expiry of ten years
from the date of grant of the option.
EMI Scheme
Options granted under the EMI Scheme are on substantially the
same terms as options granted under the Unapproved Scheme, save
that the EMI Scheme rules comply with the terms of the enterprise
management incentive as set out in Schedule 14 of the Finance Act
2000.
Employee LTIP Scheme
Options are granted at the discretion of the Directors to
eligible employees in accordance with Schedule 5 of the Income Tax
(Earnings and Pensions) Act 2003 up to the limits set out therein.
The price per share to be paid on exercise of an Employee LTIP
Option will be the market value as agreed with HMRC at the time of
the grant of the option. Options lapse on the expiry of ten years
from the date of grant, the date specified in any leaver provisions
or any other lapse date specified in the relevant option
agreement.
Non-Employee LTIP Scheme
Options are granted on substantially similar terms to the
Employee LTIP Scheme except that the EMI and/or employment related
provisions and requirements do not apply. These options can be
granted to any Director of, or individual providing consultancy or
other services to, the company.
31 December 2018 31 December 2017
------------------------- ----------------------------
Weighted Weighted
Number of average Number of average
options exercise price options exercise price
------------------------------------------- --------- -------------- ------------ --------------
Balance outstanding at beginning of year 6,748,823 GBP0.062 6,079,518(1) GBP0.068
Granted during year 350,000 GBP0.334 669,305 GBP0.01
------------------------------------------- --------- -------------- ------------ --------------
Options outstanding at end of year 7,098,823 GBP0.075 6,748,823 GBP0.062
------------------------------------------- --------- -------------- ------------ --------------
Options exercisable at the end of the year 6,585,823 GBP0.035 681,000 GBP0.068
------------------------------------------- --------- -------------- ------------ --------------
(1) On 23 January 2017 the company undertook a bonus issue of
shares whereby 499 new ordinary shares were issued fully paid to
the holders of each ordinary share by way of a partial
capitalisation of share premium account. In addition, as explained
below, some existing options where modified to reduce the number of
options outstanding.
Modification of existing share option schemes
During May and June 2017, modifications were made to the Old
Schemes by issuing replacement options in the New Schemes to
participants in the Old Schemes and new awards were subsequently
made to individuals under the New Schemes.
Options over 741,000 shares granted under the Old EMI Scheme and
over 103,000 shares granted under the Old Unapproved Scheme were
unchanged. The remaining options over 7,004,000 shares issued under
the Old Schemes were modified so that, to exercise, the holders of
such options now have the right to subscribe instead for an
aggregate of 5,235,518 shares in the company. The number of such
options and the exercise price of such options were determined by
reference to the closing fair value of the ordinary shares on the
day of modification. The modification of these options as described
had a neutral effect on the option holders immediately before and
after the amendment of the options.
After adjusting for the bonus issue on 23 January 2017,
7,848,000 share options had been issued prior to the modification
at adjusted weighted average exercise prices of between GBP0.2484
and GBP1.4522.
The estimated fair value of all share options at the
modification date was calculated by applying a Black-Scholes option
pricing model. In the absence of a liquid market for the share
capital of the company, the expected volatility of its share price
is difficult to calculate. Therefore, the Directors considered the
expected volatility used by listed entities in similar operating
environments to calculate the expected volatility. The resulting
incremental fair value was nil.
Grants of options
On 5 June 2018, 50,000 Employee LTIP EMI Options were granted to
certain senior employees at an exercise price of GBP0.01 per
ordinary share and are exercisable on or after the third
anniversary of the date of grant. On 25 October 2018, 300,000
Employee LTIP EMI Options were granted to Shaun Claydon. Of these
options, 50,000 are exercisable at GBP0.01 per ordinary share on 31
January 2019, 100,000 are exercisable at GBP0.01 on 31 January 2020
and 150,000 are exercisable at an exercise price of GBP0.765 on the
third anniversary of the date of grant.
The estimated fair value of share options granted during the
period has been calculated by applying a Black-Scholes option
pricing model. In the absence of a liquid market for the share
capital of the company the expected volatility of its share price
is difficult to calculate. Therefore, the Directors have considered
the expected volatility used by listed entities in similar
operating environments to calculate the expected volatility. The
weighted average fair value of options granted in the period was
GBP0.68 (2017: GBP1.44).
The model inputs were:
2018 2017
--------------------- ----------------- ---------
Share price GBP0.765/GBP1.115 GBP1.4522
Exercise price GBP0.01/GBP0.765 GBP0.01
Expected volatility 49% 49%
Expected option life 10 years 10 years
Risk free rate 1.5%/1.55% 1.4%
Expected dividends Nil Nil
--------------------- ----------------- ---------
11. Trade and other payables
31 December 31 December
2018 2017
GBP GBP
-------------------------------- ----------- -----------
Trade creditors 403,552 151,582
Social security and other taxes 50,874 41,110
Accrued expenses 344,275 189,251
Pension contributions payable 3,091 15,532
-------------------------------- ----------- -----------
801,792 397,475
-------------------------------- ----------- -----------
12. Bonus issue of shares and capital reduction
In January 2017, the company undertook a bonus issue of shares
whereby, in respect of each ordinary share in issue, 499 ordinary
shares were issued fully paid, resulting in a transfer of
GBP318,542 from share premium to called-up share capital.
On 26 January 2017, the company effected a reduction of capital
whereby the outstanding balance on the share premium account
amounting to GBP18,016,550 was transferred to the profit and loss
reserve.
13. Statutory accounts
The financial information set out above does not constitute the
company's statutory accounts for the years ended 31 December 2018
or 2017 but is derived from those accounts. Statutory accounts for
2017 have been delivered to the registrar of companies, and those
for 2018 will be delivered in due course. The auditor has reported
on those accounts; their reports (i) were unqualified, (ii) did not
include a reference to any matters to which the auditor drew
attention by way of emphasis without qualifying their report and
(iii) did not contain a statement under section 498 (2) or (3) of
the Companies Act 2006.
The 2018 accounts will be sent to shareholders and made
available on the Company's website www.destinypharma.com in due
course.
This information is provided by RNS, the news service of the
London Stock Exchange. RNS is approved by the Financial Conduct
Authority to act as a Primary Information Provider in the United
Kingdom. Terms and conditions relating to the use and distribution
of this information may apply. For further information, please
contact rns@lseg.com or visit www.rns.com.
END
FR UWAKRKNASRAR
(END) Dow Jones Newswires
April 09, 2019 02:00 ET (06:00 GMT)
Destiny Pharma (LSE:DEST)
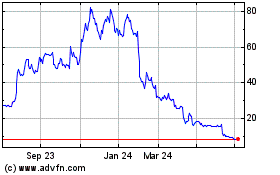
Historical Stock Chart
From Apr 2024 to May 2024
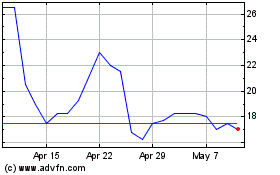
Destiny Pharma (LSE:DEST)
Historical Stock Chart
From May 2023 to May 2024