AtriCure Announces First Patient Enrollment in the DEEP IDE Trial Restart
April 23 2019 - 7:00AM
Business Wire
The trial is intended to establish the safety
and effectiveness of a dual epicardial and endocardial ablation
procedure for patients with persistent or long-standing persistent
atrial fibrillation
AtriCure, Inc. (Nasdaq: ATRC), a leading innovator in surgical
treatment for atrial fibrillation (Afib) and left atrial appendage
management, today announced the restart of the Dual Epicardial and
Endocardial Procedure (DEEP) clinical trial (NCT02393885).
Following approval by the FDA to restart the trial and enroll an
additional 40 subjects, which was granted in December 2018, the
first patient was treated by a team led by Professor Mark La Meir
and Professor Carlo de Asmundis at Universitair Ziekenhuis
Brussels, Belgium.
The DEEP trial is a prospective, multicenter, single arm,
investigational device exempt (IDE) study to evaluate the safety
and efficacy of the DEEP procedure in treating persistent and
long-standing persistent atrial fibrillation. The DEEP procedure
utilizes the specialized skills of both the cardiac surgeon and
electrophysiologist (EP) for more severe cases of Afib, which have
historically been the most difficult patients to treat. Up to 220
patients will be enrolled at up to 30 hospitals. Currently, 48
patients have been treated in the trial.
The global principal investigators for this trial are Dr.
Kenneth Ellenbogen and Dr. Vigneshwar Kasirajan, from Virginia
Commonwealth University, and Dr. Ali Khoynezhad, from Memorial Care
Health & Vascular Institute at Long Beach, California.
“The DEEP trial brings electrophysiologists and cardiac surgeons
together as a team in an effort to establish a safe and effective
care pathway for patients presenting with persistent or
long-standing persistent Afib. We are excited about the restart of
enrollment in this important trial,” said Professor Mark La Meir,
Professor of Cardiothoracic surgery at UZ Brussels who along with
Professor de Asmundis treated the first patient upon restart.
“The DEEP trial reinforces AtriCure’s commitment to continue to
expand the presence in minimally invasive treatment of persistent
and long-standing persistent patients and to improve the lives of
these difficult to treat patients,” said Michael Carrel, President
and Chief Executive Officer of AtriCure. “We’ve worked very closely
with the FDA and with our investigators to do everything we can to
ensure a safe, effective and repeatable procedure.”
About AtriCure, Inc.
AtriCure, Inc. provides innovative technologies for the
treatment of Afib and related conditions. Afib affects more than 33
million people worldwide. Electrophysiologists and cardiothoracic
surgeons around the globe use AtriCure technologies for the
treatment of Afib and reduction of Afib related complications.
AtriCure’s Isolator® Synergy™ Ablation System is the first and only
medical device to receive FDA approval for the treatment of
persistent Afib. AtriCure’s AtriClip Left Atrial Appendage (LAA)
Exclusion System products are the most widely sold LAA management
devices worldwide. For more information, visit AtriCure.com or
follow us on Twitter @AtriCure.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20190423005188/en/
Valerie Storch-WillhausMedia RelationsSenior Director, Corporate
Marketing and Communications(612)
605-3311vstorch-willhaus@AtriCure.com
Andy WadeInvestor RelationsSenior Vice President and Chief
Financial Officer(513) 755-4564awade@AtriCure.com
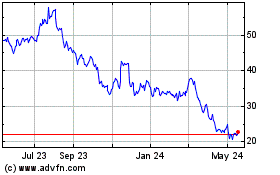
AtriCure (NASDAQ:ATRC)
Historical Stock Chart
From Apr 2024 to May 2024
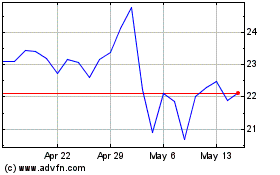
AtriCure (NASDAQ:ATRC)
Historical Stock Chart
From May 2023 to May 2024