Milestone Pharmaceuticals' Etripamil Nasal Spray Application Gets FDA's Refusal to File Letter
December 26 2023 - 6:57AM
Dow Jones News
By Denny Jacob
Milestone Pharmaceuticals received a refusal to file letter from
the Food and Drug Administration over its new drug application for
its etripamil nasal spray to treat a type of abnormal heart
rhythm.
The biopharmaceutical company said the FDA determined that the
application for etripamil nasal spray as a treatment of paroxysmal
supraventricular tachycardia wasn't sufficiently complete to permit
a substantive review based on a preliminary review.
The regulator requested clarification about the time of data
recorded for adverse events in Phase 3 clinical trials, said
Milestone.
Milestone said the FDA didn't express concerns about the nature
or severity of adverse events. It will seek clarification and is
planning a meeting with the FDA.
Write to Denny Jacob at denny.jacob@wsj.com
(END) Dow Jones Newswires
December 26, 2023 07:42 ET (12:42 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
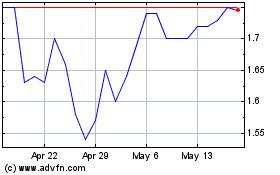
Milestone Pharmaceuticals (NASDAQ:MIST)
Historical Stock Chart
From Aug 2024 to Sep 2024
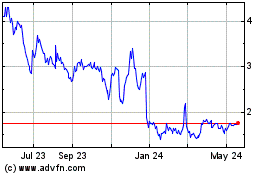
Milestone Pharmaceuticals (NASDAQ:MIST)
Historical Stock Chart
From Sep 2023 to Sep 2024