– First Automated Insulin Delivery System in
the World to Feature Dexcom’s Most Advanced CGM Technology –
Tandem Diabetes Care, Inc. (NASDAQ: TNDM), a leading insulin
delivery and diabetes technology company, today announced the
launch of updated t:slim X2™ insulin pump software with Dexcom G7
Continuous Glucose Monitoring (CGM) integration in the United
States. Tandem’s #1-rated automated insulin delivery (AID) system*
with Control-IQ technology is the only AID option in the world
today to use Dexcom’s latest CGM technology.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20231206939624/en/
t:slim X2 Insulin Pump from Tandem
Diabetes Care with Dexcom G7 CGM Integration (Photo: Business
Wire)
Tandem will email instructions to all in-warranty t:slim X2
users in the United States to offer the option to add the new
feature free of charge via remote software update. t:slim X2 pumps
pre-loaded with the updated software are now shipping to new
customers.
“With this launch we are offering more than 300,000 current
t:slim X2 users the ability to integrate with Dexcom’s most
advanced CGM technology, demonstrating our commitment to continued
leadership in advancing AID systems,” said John Sheridan, president
and chief executive officer of Tandem Diabetes Care. “As we
celebrate our 10-year partnership with Dexcom, we remain committed
to sustaining our rapid pace of innovation to further our mission
of helping improve the lives of people with diabetes.”
With the integration of Dexcom G7, t:slim X2 insulin pump users
can now:
- Spend more time in closed loop with little to no wait time
between Dexcom G7 CGM sensor sessions.**
- Use either a Dexcom G6 or Dexcom G7 CGM sensor, allowing even
more choice in their diabetes management.
Dexcom G7, which is 60 percent smaller than its Dexcom G6
predecessor, offers a range of new features, including:
- Improved accuracy***
- A 30-minute sensor warmup time, compared to 2 hours
previously
- A 12-hour grace period to replace finished sensors for a more
seamless transition between sessions and flexibility for when to
change sensors
“The Dexcom G7 and t:slim X2 insulin pump integration is the
culmination of years of partnership and a shared commitment to
improve the lives of people with diabetes,” said Jake Leach,
executive vice president and chief operating officer at Dexcom.
“We’re excited to bring our newest technology to Tandem users so
they can experience the benefits of the smallest, most accurate,
easy to use CGM connected to AID systems.”
For additional product and safety information, or to begin the
order process for a new Tandem pump with Dexcom G7 integration,
visit tandemdiabetes.com/products.
Information about the software update process for existing
in-warranty pump users, including system requirements, is available
at tandemdiabetes.com/G7update.
The t:slim X2 pump with Dexcom G7 integration is expected to
launch in additional countries outside the U.S. in early 2024.
* dQ&A US Q1 2023 Diabetes Connections Patient Panel Report
** No wait time claim based on user inserting a new, unpaired
Dexcom G7 at least 30 minutes before the old, paired Dexcom G7
sensor expires. See User Guide for more details on the sensor start
process. *** Dexcom, data on file, 2022.
About Tandem Diabetes Care, Inc.
Tandem Diabetes Care, Inc., a global insulin delivery and
diabetes technology company based in San Diego, California, creates
new possibilities for people living with diabetes, their loved
ones, and healthcare providers through a positively different
experience. The company’s human-centered approach to design,
development, and support delivers innovative products and services
for people who use insulin. Tandem manufactures and sells the
t:slim X2 insulin pump with Control-IQ technology. For more
information, visit tandemdiabetes.com.
Follow Tandem Diabetes Care on Twitter @tandemdiabetes; use
#tslimX2 and #TandemDiabetes. Follow Tandem Diabetes Care on
Facebook at facebook.com/TandemDiabetes. Follow Tandem Diabetes
Care on LinkedIn at linkedin.com/company/tandemdiabetes.
Forward-looking Statements
This press release contains “forward-looking statements” within
the meaning of Section 27A of the Securities Act of 1933, as
amended, and Section 21E of the Securities Exchange Act of 1934, as
amended. These forward-looking statements relate to, among other
things, the anticipated timing for the international availability
of the t:slim X2 pump with Dexcom G7 integration as well as our
ability to provide the software update for current t:slim X2 pump
users and the t:slim X2 pumps pre-loaded with Dexcom G7 integration
software. These statements are subject to numerous risks and
uncertainties, including our ability to start commercial scale
manufacturing of the t:slim X2 pumps pre-loaded with Dexcom G7
integration software, our ability to operate and maintain a system
to facilitate online training for existing t:slim X2 pump customers
upgrading their existing devices, and the risk that we may
encounter other challenges that may delay the availability of
t:slim X2 pumps pre-loaded with Dexcom G7 integration software.
These and other risks are identified and described in greater
detail under the “Risk Factors” heading of our most recent Annual
Report on Form 10-K, Quarterly Reports on Form 10-Q, and other
documents filed with the Securities and Exchange Commission.
Readers are cautioned not to place undue reliance on these
forward-looking statements, which speak only as of the date of this
release. Actual results could differ materially from those
anticipated or projected in the forward-looking statements. Tandem
undertakes no obligation to update or review any forward-looking
statement in this press release because of new information, future
events, or other factors.
Responsible use of Control-IQ technology
Control-IQ technology does not prevent all highs and lows. Users
must still bolus for meals and actively manage their diabetes.
Visit tandemdiabetes.com/safetyinfo for additional important safety
information.
Important Safety Information: RX ONLY. The t:slim X2 pump
and Control-IQ technology are intended for single patient use. The
t:slim X2 pump and Control-IQ technology are indicated for use with
NovoLog or Humalog U-100 insulin. t:slim X2
insulin pump: The t:slim X2 insulin pump with interoperable
technology is intended for the subcutaneous delivery of insulin, at
set and variable rates, for the management of diabetes mellitus in
people requiring insulin. The pump is able to reliably and securely
communicate with compatible, digitally connected devices, including
automated insulin dosing software, to receive, execute, and confirm
commands from these devices. The pump is indicated for use in
individuals 6 years of age and greater. Control-IQ technology: Control-IQ technology is
intended for use with compatible integrated continuous glucose
monitors (iCGM, sold separately) and alternate controller enabled
(ACE) pumps to automatically increase, decrease, and suspend
delivery of basal insulin based on iCGM readings and predicted
glucose values. It can also deliver correction boluses when the
glucose value is predicted to exceed a predefined threshold.
Control-IQ technology is intended for the management of Type 1
diabetes mellitus in persons 6 years of age and greater.
WARNING: Control-IQ technology should not be used by
anyone under the age of 6 years old. It should also not be used in
patients who require less than 10 units of insulin per day or who
weigh less than 55 pounds.
Control-IQ technology is not indicated for use in pregnant
women, people on dialysis, or critically ill patients. Do not use
Control-IQ technology if using hydroxyurea. Users of the t:slim X2
pump and Control-IQ technology must: use the insulin pump, CGM, and
all other system components in accordance with their respective
instructions for use; test blood glucose levels as recommended by
their healthcare provider; demonstrate adequate carb-counting
skills; maintain sufficient diabetes self-care skills; see
healthcare provider(s) regularly; and have adequate vision and/or
hearing to recognize all functions of the pump, including alerts,
alarms, and reminders. The t:slim X2 pump must be removed before
MRI, CT, or diathermy treatment. Visit
tandemdiabetes.com/safetyinfo for additional important safety
information.
© 2023 Tandem Diabetes Care, Inc. All rights reserved. Tandem
Diabetes Care, the Tandem logo, Control-IQ, and t:slim X2 are
either registered trademarks or trademarks of Tandem Diabetes Care,
Inc. in the United States and/or other countries. All third-party
marks are the property of their respective owners.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20231206939624/en/
Tandem Diabetes Care Media Contact: 858-366-6900
media@tandemdiabetes.com Tandem Diabetes Care Investor
Contact: 858-366-6900 IR@tandemdiabetes.com
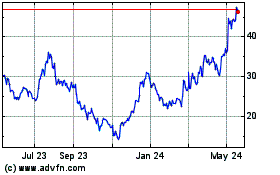
Tandem Diabetes Care (NASDAQ:TNDM)
Historical Stock Chart
From Apr 2024 to May 2024
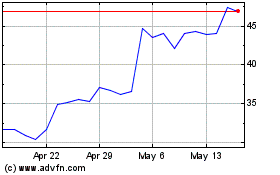
Tandem Diabetes Care (NASDAQ:TNDM)
Historical Stock Chart
From May 2023 to May 2024