Filed Pursuant to Rule 424(b)(2)
Registration No. 333-194944
PROSPECTUS SUPPLEMENT
(To Prospectus dated March 31, 2014)
XTANT MEDICAL HOLDINGS, INC.
843,289 Shares of Common Stock, $0.5692 per share
We are offering 843,289 shares of our common stock directly to certain holders of our convertible promissory
notes (the “Indenture Notes”). Our common stock is listed on the NYSE MKT exchange and traded under the symbol “XTNT.”
On January 12, 2017, the last reported sales price of our common stock was $0.59 per share. As of January 12, 2017, 2,674,397 shares
of our common stock were held by affiliates and 14,574,918 shares were held by non-affiliates. The aggregate market value of our
outstanding voting and non-voting common equity held by non-affiliates on January 12, 2017, was $8,599,202 based on $0.59, the
price at which our common stock was last sold on January 12, 2017. During the twelve calendar months prior to and including the
date hereof, we have not sold any securities pursuant to General Instruction I.B.6. of Form S-3.
We are not paying underwriting discounts
or commissions. We estimate the total expenses of this offering will be approximately $75,000, and we will pay those expenses from
cash on hand. We are issuing the common stock to offset interest owed by us under the Indenture Notes
to the purchasers of the common stock in the amount of $480,000. We will not receive any cash proceeds in this offering.
Investing in our securities
involves a high degree of risk. See “Risk Factors” beginning on page S-6 of this prospectus supplement and page 2
of the accompanying prospectus to read about factors you should consider before investing in the securities.
We anticipate
that delivery of the shares will be made on or about January 17, 2017.
Neither the Securities and Exchange
Commission nor any other regulatory body has approved or disapproved of these securities or passed upon the adequacy or accuracy
of this prospectus. Any representation to the contrary is a criminal offense.
The date of this prospectus supplement is January 17, 2017.
Table of Contents
Prospectus Supplement
Accompanying Prospectus
ABOUT THIS PROSPECTUS
SUPPLEMENT
This prospectus supplement and the accompanying
prospectus are part of a registration statement that we filed with the Securities and Exchange Commission, or SEC, utilizing a
“shelf” registration process. Under this shelf registration process, we may sell, either separately or together, common
stock, preferred stock, warrants, and units from time to time in one or more offerings up to an aggregate initial offering price
of $50,000,000, subject to the limitations imposed by General Instruction I.B.6 of Form S-3.
The accompanying prospectus provides you with a general description of the securities we may offer. In
this prospectus supplement, we provide you with specific information about this offering of 843,289 shares of our common stock,
$0.000001 par value per share. Both this prospectus supplement and the accompanying prospectus, including the documents incorporated
by reference in this prospectus supplement and the accompanying prospectus, include important information about us, our common
stock and other information you should know before investing. Any statement contained in this prospectus supplement, the accompanying
prospectus or in a document incorporated by reference shall be deemed to be modified or superseded to the extent that a statement
contained herein or in any subsequently filed document which is incorporated by reference modifies or supersedes such earlier statement.
Any statement so modified or superseded shall not be deemed, except as so modified or superseded, to constitute a part of this
prospectus supplement or the accompanying prospectus. You should read both this prospectus supplement and the accompanying prospectus
as well as the documents incorporated herein and therein before investing in our securities.
You should rely only on the information
incorporated by reference or presented in this prospectus supplement and the accompanying prospectus. Neither we nor the placement
agent have authorized anyone else to provide you with different information. You should not assume that the information in this
prospectus supplement, the accompanying prospectus or in any document incorporated herein or therein by reference is accurate as
of any date other than the dates on the front of those documents, regardless of the time of delivery of this prospectus supplement
or any sale of our securities.
As used in this prospectus, unless the
context otherwise requires, references to “we,” “us,” “our,” “Xtant” and “the
Company” refer to Xtant Medical Holdings, Inc. and its subsidiaries.
CAUTIONARY NOTE REGARDING
FORWARD-LOOKING STATEMENTS
The statements set forth and incorporated
by reference in this prospectus that are not purely historical are forward-looking statements within the meaning of applicable
securities laws. Our forward-looking statements include, but are not limited to, statements regarding our “expectations,”
“hopes,” “beliefs,” “intentions” or “strategies” regarding the future. In addition,
any statements that refer to projections, forecasts, or other characterizations of future events or circumstances, including any
underlying assumptions, are forward-looking statements. The words “anticipate,” “believe,” “continue,”
“could,” “estimate,” “expect,” “intend,” “may,” “might,”
“plan,” “possible,” “potential,” “predict,” “project,” “should”
and “would,” as well as similar expressions, may identify forward-looking statements, but the absence of these words
does not mean that a statement is not forward looking. Forward-looking statements set forth and incorporated by reference in this
prospectus may include, for example, statements about:
|
|
·
|
our ability to comply with the covenants in our senior credit facility and to make the interest
payment deferred to January 31, 2017;
|
|
|
·
|
our ability to maintain sufficient liquidity to fund our operations;
|
|
|
·
|
our ability to remain listed on NYSE MKT;
|
|
|
·
|
our ability to obtain financing on reasonable terms;
|
|
|
·
|
our ability to generate enough cash flow to meet our debt obligations;
|
|
|
·
|
our ability to remain solvent and avoid filing for bankruptcy;
|
|
|
·
|
our ability to increase revenue;
|
|
|
·
|
the ability of our sales force to achieve expected results;
|
|
|
·
|
our ability to remain competitive;
|
|
|
·
|
government regulations;
|
|
|
·
|
our ability to innovate and develop new products;
|
|
|
·
|
our ability to obtain donor cadavers for our products;
|
|
|
·
|
our ability to engage and retain qualified technical personnel and members of our management team;
|
|
|
·
|
the availability of our facilities;
|
|
|
·
|
government and third-party coverage and reimbursement for our products;
|
|
|
·
|
our ability to obtain regulatory approvals;
|
|
|
·
|
our ability to successfully integrate recent and future business combinations or acquisitions;
|
|
|
·
|
our ability to use our net operating loss carry-forwards to offset future taxable income;
|
|
|
·
|
our ability to deduct all or a portion of the interest payments on the notes for U.S. federal income
tax purposes;
|
|
|
·
|
our ability to service our debt;
|
|
|
·
|
product liability claims and other litigation to which we may be subjected;
|
|
|
·
|
product recalls and defects;
|
|
|
·
|
timing and results of clinical studies;
|
|
|
·
|
our ability to obtain and protect our intellectual property and proprietary rights;
|
|
|
·
|
infringement and ownership of intellectual property;
|
|
|
·
|
our ability to remain accredited with the American Association of Tissue Banks;
|
|
|
·
|
influence by our management;
|
|
|
·
|
our ability to pay dividends; and
|
|
|
·
|
our ability to issue preferred stock.
|
The forward-looking statements set forth
and incorporated by reference in this prospectus are based on our current expectations and beliefs concerning future developments
and their potential effects on us. There can be no assurance that future developments affecting us will be those that we have anticipated.
These forward-looking statements involve a number of risks, uncertainties or assumptions, many of which are beyond our control,
which may cause actual results or performance to be materially different from those expressed or implied by these forward-looking
statements. These risks and uncertainties include, but are not limited to, those factors described in the “Risk Factors”
section of this prospectus and the documents incorporated by reference. Should one or more of these risks or uncertainties materialize,
or should any of our assumptions prove incorrect, actual results may vary in material respects from those projected in these forward-looking
statements. We undertake no obligation to update or revise any forward-looking statements, whether as a result of new information,
future events or otherwise, except as may be required under applicable securities laws.
You should also read carefully the factors
described in the “Risk Factors” in our annual report on Form 10-K filed with the SEC on March 24, 2016, to better understand
significant risks and uncertainties inherent in our business and underlying any forward-looking statements. As a result of these
factors, actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements
in this report and you should not place undue reliance on any forward-looking statements.
PROSPECTUS SUPPLEMENT
SUMMARY
This summary highlights certain information
about us, this offering and information appearing elsewhere in this prospectus supplement, in the accompanying prospectus and in
the documents incorporated by reference. This summary is not complete and does not contain all of the information that you should
consider before investing in our securities. To fully understand this offering and its consequences to you, you should read this
entire prospectus supplement and the accompanying prospectus carefully, including the information referred to under the heading
“Risk Factors” in this prospectus supplement beginning on page S-6, the financial statements and other information
incorporated by reference in this prospectus supplement and the accompanying prospectus when making an investment decision. This
is only a summary and may not contain all the information that is important to you.
About Xtant Medical Holdings, Inc.
We operate through our subsidiaries Bacterin
International, Inc. (“Bacterin”) and X-spine Systems, Inc. (“X-spine”). Through Bacterin, we develop, manufacture
and market biologics products to domestic and international markets. Our bone graft products are used in a variety of applications
including enhancing fusion in spine surgery, relief of back pain through facet joint stabilization, promotion of bone growth in
foot and ankle surgery, promotion of skull healing following neurosurgery and subchondral bone repair in knee and other joint surgeries.
Our acellular dermis scaffolds are utilized in wound care and plastic and reconstructive procedures. Bacterin also develops custom
surgical instruments for use with our allografts, and we produce and distribute OsteoSelect
®
DBM putty, an osteoinductive
product used by surgeons as a bone void filler in the extremities and pelvis. X-spine is a global developer, manufacturer and marketer
of implants and instruments for surgery of the spine and sacroiliac joint. X-spine’s product emphasis is the minimally invasive
approach to the treatment of degenerative spine disorders. X-spine’s global strategy is to advance minimally invasive technologies
for the treatment of degenerative spinal disorders, while supporting established spinal fusion markets.
We are a Delaware corporation. Our principal
executive offices are located at 664 Cruiser Lane, Belgrade, Montana 59714. Our telephone number is (406) 388-0480 and our website
address is www.xtantmedical.com. Information contained in, or that can be accessed through, our website is not part of this prospectus.
The following description of our business
should be read in conjunction with the section titled “Business” in Item 1, Part I of our Annual Report on Form 10-K
for the year ended December 31, 2015, which is incorporated by reference into this prospectus.
Overview of Our Business
Xtant believes the following competitive
strengths will be key drivers of future growth of Xtant:
Portfolio of Proprietary
Technologies
: Xtant has developed a comprehensive portfolio of products that address a broad array of spinal pathologies, anatomies
and surgical approaches in the complex spine and minimally invasive surgery (“MIS”) markets. To protect company innovative
technologies and techniques, Xtant maintains and continues to grow its intellectual property portfolio, with over 100 issued patents
globally and over 40 patent applications pending.
Customer Focus
: Responding
quickly and efficiently to the needs of patients, surgeons and hospitals is central to corporate culture and critical to success.
Our supply chain and customer service teams make sure that the right product and instrumentation is in the right place at the right
time. Through such vertically integrated processes, we are able to meet the changing needs of our customers.
Multi-channel Distribution
Network
: Xtant has built a hybrid sales and distribution function calling on Orthopedic Surgeons, Neuro Surgeons, their staff
and the hospital administrators that support them. Approximately 300 field agents and distributors in the United States represent
some or all of Xtant’s products. The distribution channel consists of multiple sub-channels including direct sales, consignment
agents, reseller distributors, and private label distributors and technology licensees.
Our Offices
Our headquarter office and manufacturing
facility are located at 664 Cruiser Lane, Belgrade, Montana 59714. Our telephone number is (406) 388-0480 and our fax number is
(406) 388-1354. We also have two other facilities on the Montana campus, located at 600 Cruiser Lane, Belgrade, Montana 59714,
and at 732 Cruiser Lane, Belgrade, Montana 59714, a Colorado office located at 363 Centennial Parkway, Louisville, Colorado 80112,
and one Ohio facility at 452 Alexandersville Road, Miamisburg, Ohio 45342. All our properties are leased.
THE OFFERING
|
Common stock offered by us
|
843,289 shares of common stock.
|
|
|
|
|
Public offering price of a share of common stock
|
$0.5692
|
|
|
|
|
Common stock to be outstanding immediately after the offering
|
18,092,604 shares
|
|
|
|
|
Use of proceeds
|
We are issuing the common stock to offset interest owed by us under the Indenture Notes to the purchasers of the common stock in the amount of $480,000. We will not receive any cash proceeds in this offering. See “Use of Proceeds.”
|
|
|
|
|
Risk factors
|
Investing in our securities involves a high degree of risk. You should read the “Risk Factors” beginning on page S-6 of this prospectus supplement, on page 2 of the accompanying prospectus and in the documents incorporated by reference in this prospectus supplement and the accompanying prospectus, including our Annual Report on Form 10-K for the year ended December 31, 2015 filed with the SEC on March 24, 2016, for a discussion of factors to consider before deciding to purchase shares of our common stock.
|
|
|
|
|
NYSE MKT symbol
|
Our common stock is listed on the NYSE MKT under the symbol “XTNT.”
|
The number of shares of common stock to
be outstanding after this offering as reflected in the table above is based on the actual number of shares outstanding as of January
12, 2017 which was 17,249,315, and does not include, as of that date:
|
|
·
|
849,206 shares issuable upon the exercise of outstanding stock options with a weighted average
exercise price of $7.65 per share;
|
|
|
·
|
6,270,986 shares issuable upon the exercise of outstanding warrants with a weighted average exercise
price of $2.36 per share;
|
|
|
·
|
Approximately 530,000 shares available for issuance under our Amended and Restated Equity Incentive
Plan;
|
|
|
·
|
1,071,629 shares reserved for issuance pursuant to a common stock purchase agreement with Aspire
Capital Fund, LLC; and
|
|
|
·
|
25,118,805 shares reserved for issuance upon conversion of our outstanding notes.
|
Pursuant to the Indenture,
dated as of July 31, 2015, between the Company and Wilmington Trust, National Association, and the $68,000,000 of Indenture Notes
issued thereunder, the Company is required to make interest payments on January 15, 2017, to the holders of the Indenture Notes
aggregating $2,040,000. ROS Acquisition Offshore LP (“ROS”) and OrbiMed Royalty Opportunities II, LP (“OrbiMed”),
collectively own $52,000,000 of such Indenture Notes. Further, pursuant to the Securities Purchase Agreement, dated as of April
14, 2016, and the Convertible Promissory Notes (the “PIK Notes”) issued thereunder in the aggregate principal amount
of $2,238,166.45, the Company is also required to make interest payments on January 15, 2017 to OrbiMed and ROS aggregating $67,145,
who collectively own all of the PIK Notes.
Concurrently with this
offering, we are issuing new convertible promissory notes (the “New Notes”) to ROS and OrbiMed to satisfy the January
15, 2017 interest payments owed to them under their Indenture Notes and PIK Notes. The shares being issued in this offering are
being issued to satisfy the interest payment due in respect of the remaining Indenture Notes.
The New Notes will bear
interest at the rate of 6.00%, with a maturity date of August 1, 2021, and are convertible into common stock of the Company at
a 20% premium to the 20-day volume weighted average price of the common stock as of January 15, 2017.
OrbiMed and ROS have
deferred the interest due under the Company’s senior credit facility due on December 31, 2016, to January 14, 2017, and they
further deferred such interest to January 31, 2017. The Company, OrbiMed and ROS are in discussions about adding the deferred interest
to the principal amount of the senior credit facility. No assurances can be given that those discussions will be successful.
RISK FACTORS
Our business and an investment in our
securities are subject to a variety of risks. The following risk factors describe some of the most significant events, facts or
circumstances that could have a material adverse effect upon our business, financial condition, results of operations, ability
to implement our business plan and the market price for our securities. Many of these events are outside of our control. If any
of these risks actually occurs, our business, financial condition or results of operations may be materially adversely affected.
In such case, the trading price of our Common Stock could decline and investors in our Common Stock could lose all or part of their
investment.
Before making an investment decision,
you should carefully consider these risks, including those set forth below and those described in the “Risk Factors”
section of our most recent Annual Report on Form 10-K, filed with the Commission on March 24, 2016, as revised or supplemented
by our Quarterly Reports on Form 10-Q filed with the SEC since the filing of our most recent Annual Report on Form 10-K, each of
which is incorporated by reference into this prospectus, and you should also carefully consider any other information we include
or incorporate by reference in this prospectus.
See “Risk Factors” beginning
on page 2 of the accompanying prospectus.
Risks Related to this Offering
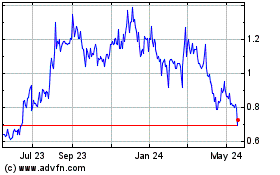
The market price of our common stock
is extremely volatile, which may affect our ability to raise capital in the future and may subject the value of your investment
to sudden decreases.
The market price for securities of biotechnology
companies, including ours, historically has been highly volatile, and the market from time to time has experienced significant
price and volume fluctuations that are unrelated to the operating performance of such companies. Fluctuations in the trading price
or liquidity of our common stock may harm the value of your investment in our securities.
Factors that may have a significant impact
on the market price and marketability of our securities include:
|
|
·
|
our ability to make the January 31, 2017 interest payment under the senior credit facility if it
is not added to the principal thereof;
|
|
|
·
|
our observance of covenants under our credit facilities, including the March 31, 2017 covenants
on minimum liquidity and consolidated senior leverage ratio;
|
|
|
·
|
our issuance of debt, equity or other securities, which we need to pursue to generate additional
funds to cover our operating expenses;
|
|
|
·
|
our quarterly operating results;
|
|
|
·
|
announcements of technological innovations or new commercial products by us, our collaborative
partners or our present or potential competitors;
|
|
|
·
|
developments or disputes concerning patent or other proprietary rights;
|
|
|
·
|
developments in our relationships with employees, suppliers or collaborative partners;
|
|
|
·
|
acquisitions or divestitures;
|
|
|
·
|
litigation and government proceedings;
|
|
|
·
|
adverse legislation, including changes in governmental regulation;
|
|
|
·
|
third-party reimbursement policies;
|
|
|
·
|
changes in securities analysts’ recommendations;
|
|
|
·
|
changes in health care policies and practices;
|
|
|
·
|
halting or suspension of trading in our common stock by the NYSE MKT;
|
|
|
·
|
economic and other external factors; and
|
|
|
·
|
general market conditions.
|
In the past, following periods of volatility in the market price
of a company’s securities, securities class action litigation has often been instituted. These lawsuits often seek unspecified
damages, and as with any litigation proceeding, one cannot predict with certainty the eventual outcome of pending litigation. Furthermore,
we may have to incur substantial expenses in connection with any such lawsuits and our management’s attention and resources
could be diverted from operating our business as we respond to any such litigation. We maintain insurance to cover these risks
for us and our directors and officers, but our insurance is subject to high deductibles to reduce premium expense, and there is
no guarantee that the insurance will cover any specific claim that we currently face or may face in the future, or that it will
be adequate to cover all potential liabilities and damages.
We will need to secure additional financing in order to
continue to finance our operations. If we are unable to secure additional financing on acceptable terms, or at all, we may be forced
to curtail or cease our operations.
We will need to secure additional sources of capital to develop
our business and product candidates as planned. We are seeking substantial additional financing through public and/or private financing,
which may include equity and/or debt financings and through other arrangements, including collaborative arrangements. If we are
unable to secure additional financing in the near term, we may be forced to:
|
|
·
|
curtail or abandon our existing business plans;
|
|
|
·
|
seek to sell some or all of our assets; and/or
|
If we are forced to take any of these steps, any investment
in our common stock may be worthless.
We may not be able to generate enough cash flow to meet
our debt obligations.
Our future cash flow may be insufficient
to meet our debt obligations and commitments. Any insufficiency could negatively impact our business. A range of economic, competitive,
business, regulatory, and industry factors will affect our future financial performance, and, as a result, our ability to generate
cash flow from operations and to pay our debt. Many of these factors, such as economic and financial conditions in our industry
and the U.S. or the global economy, or competitive initiatives of our competitors, are beyond our control.
If we do not generate enough cash flow
from operations to satisfy our debt obligations, we may have to undertake alternative financing plans, such as:
|
|
·
|
reducing or delaying capital investments;
|
|
|
·
|
raising additional capital;
|
|
|
·
|
refinancing or restructuring our debt;
|
|
|
·
|
ceasing our operations; and
|
We cannot assure you that we would be able
to implement alternative financing plans, if necessary, on commercially reasonable terms, or at all, or that implementing any such
alternative financing plans would allow us to meet our debt obligations.
If we are unable to meet our debt obligations,
we would be in default under the terms of such arrangement, permitting acceleration of the amounts due. If the amounts outstanding
under our indebtedness were to be accelerated, we could be forced to file for bankruptcy.
The Company does not have cash on hand
to satisfy its January 31, 2017 interest obligations under its senior credit facility. Failure of the Company to successfully negotiate
its interest obligations will result in the Company being in default under the senior credit facility and, due to the cross defaults
thereunder, under the Loan and Security Agreement, dated May 25, 2016, with Silicon Valley Bank.
Because we became public through
a reverse merger, we may not be able to attract the attention of major brokerage firms or certain investors.
There are coverage risks associated with our becoming public
through a reverse merger, including, among other things, security analysts of major brokerage firms may not provide coverage of
us since there is no incentive to brokerage firms to recommend the purchase of our common stock. In addition, we may not attract
the attention of major brokerage firms and certain investors due to our low stock price. We cannot assure you that brokerage firms
would want to conduct any public offerings on our behalf in the future.
If securities or industry analysts publish inaccurate
or unfavorable research or reports about our business, our stock price and trading volume could decline.
The trading market for our common stock depends, in part, on
the research and reports that securities or industry analysts publish about us or our business. We do not have any control over
these analysts. If one or more of the analysts who covers us downgrades our common stock, changes their opinion of our shares or
publishes inaccurate or unfavorable research about our business, our stock price would likely decline. If one or more of these
analysts ceases coverage of us or fails to publish reports on us regularly, demand for our common stock could decrease and we could
lose visibility in the financial markets, which could cause our stock price and trading volume to decline.
Shares of common stock are equity
securities and are subordinate to any indebtedness.
Shares of our common stock are common equity
interests. This means that our common stock will rank junior to any outstanding shares of our preferred stock that we may issue
in the future or to our current credit agreement and any future indebtedness we may incur and to all creditor claims and other
non-equity claims against us and our assets available to satisfy claims on us, including claims in a bankruptcy or similar proceeding.
Additionally, unlike indebtedness, where
principal and interest customarily are payable on specified due dates, in the case of our common stock, (i) dividends are payable
only when and if declared by our board of directors or a duly authorized committee of our board of directors, and (ii) as a corporation,
we are restricted to making dividend payments and redemption payments out of legally available assets. We have never paid a dividend
on our common stock and have no current intention to pay dividends in the future. Furthermore, our common stock places no restrictions
on our business or operations or on our ability to incur indebtedness or engage in any transactions, subject only to the voting
rights available to shareholders generally.
We do not anticipate paying dividends in the foreseeable
future; you should not buy our stock if you expect dividends.
Sales of securities in this offering may
result in substantial dilution to the interests of holders of our securities. The sale of securities in this offering, or the anticipation
of such sales, could make it more difficult for us to sell equity or equity-related securities in the future at a time and at prices
that we might otherwise wish to effect sales. Depending on market liquidity at the time, a sale of securities in this offering
at any given time could cause the trading price of our common stock to decline. Should the financing we require to sustain our
working capital needs be unavailable or prohibitively expensive when we require it, the consequences could have a material adverse
effect on our business, operating results, financial condition and prospects.
Investors in this offering will experience
immediate and substantial dilution.
If you purchase shares in this offering, you will incur immediate and substantial dilution of approximately
$(2.68) per share, representing the difference between the price per share you pay and the net tangible book value per share
of our common stock immediately after this offering. Any exercise of outstanding stock options, warrants or other equity awards
will result in further dilution. For a further description of the dilution that you will experience immediately after this offering,
see “Dilution.”
We could issue “blank check” preferred stock
without stockholder approval with the effect of diluting then current stockholder interests and impairing their voting rights,
and provisions in our charter documents and under Delaware law could discourage a takeover that stockholders may consider favorable.
Sales of securities in this offering may result in substantial
dilution to the interests of holders of our securities. The sale of securities in this offering, or the anticipation of such sales,
could make it more difficult for us to sell equity or equity-related securities in the future at a time and at prices that we might
otherwise wish to effect sales. Depending on market liquidity at the time, a sale of securities in this offering at any given time
could cause the trading price of our common stock to decline. Should the financing we require to sustain our working capital needs
be unavailable or prohibitively expensive when we require it, the consequences could have a material adverse effect on our business,
operating results, financial condition and prospects.
USE OF PROCEEDS
We are issuing the common stock to offset
interest owed by us under the Indenture Notes to the purchasers of the common stock in the amount of $480,000. We will not receive
any cash proceeds in this offering. The Company is required to make interest payments on January 15, 2017, to the holders of the
Indenture Notes aggregating $2,040,000. The remaining interest due will be satisfied by the issuance of the New Notes to
ROS and OrbiMed. All expenses of this offering will be paid from cash on hand.
DILUTION
Our net negative tangible book value on
September 30, 2016 was approximately $42.1 million, or approximately $(3.47) per share of common stock. After giving effect to
the sale of shares of common stock offered by us in this offering at a price of $0.5692 per share, less the other expenses of this
offering payable by us, our pro forma as adjusted negative net tangible book value on September 30, 2016 would have been approximately
$42.2 million, or $(3.25) per share of common stock. The following table illustrates this dilution on a per share basis:
|
Public offering price per share
|
|
|
|
|
|
$
|
0.5692
|
|
|
Net tangible book value per share as of September 30, 2016
|
|
$
|
(3.47
|
)
|
|
|
|
|
|
Increase in net tangible book value per share attributable to offering
|
|
$
|
0.22
|
|
|
|
|
|
|
Pro forma net tangible book value per share after giving effect to this offering
|
|
|
|
|
|
$
|
(3.25
|
)
|
|
Dilution per share to investors in the offering
|
|
|
|
|
|
$
|
(2.68
|
)
|
The above discussion and table are based
on 17,249,315 common shares outstanding at January 12, 2017, and do not include, as of that date:
|
|
·
|
849,206 shares issuable upon the exercise of outstanding stock options with a weighted average
exercise price of $7.65 per share;
|
|
|
·
|
6,270,986 shares issuable upon the exercise of outstanding warrants with a weighted average exercise
price of $2.36 per share;
|
|
|
·
|
Approximately 530,000 shares available for issuance under our Amended and Restated Equity Incentive
Plan;
|
|
|
·
|
1,071,629 shares reserved for issuance pursuant to a common stock purchase agreement with Aspire
Capital Fund, LLC; and
|
|
|
·
|
25,118,805 shares reserved for issuance upon conversion of our outstanding notes.
|
If any shares of our
common stock are issued upon exercise of outstanding options or warrants, you will experience further dilution.
CAPITALIZATION
Our authorized capital stock consists of
95,000,000 shares of common stock, $0.000001 par value per share, and 5,000,000 shares of preferred stock, $0.000001 par value
per share. As of January 12, 2017, we had 17,249,315 outstanding shares of common stock and no outstanding shares of preferred
stock.
As of January 12, 2017, we had 849,206 shares issuable upon the exercise of outstanding stock options
with a weighted average exercise price of $7.65 per share; 6,270,986 shares issuable upon the exercise of outstanding warrants
with a weighted average exercise price of $2.36 per share; approximately 530,000 shares available for issuance under our Amended
and Restated Equity Incentive Plan; 1,071,629 shares reserved for issuance pursuant to a common stock purchase agreement with Aspire
Capital Fund, LLC; and 25,118,805 shares reserved for issuance upon conversion of our outstanding notes.
Holders of common stock are entitled to
one vote for each share held on all matters submitted to a vote of shareholders including in all elections for directors; stockholders
are not entitled to cumulative voting in the election of directors.
Holders of outstanding shares of our common
stock are entitled to those dividends declared by the Board of Directors out of legally available funds, and, in the event of our
liquidation, dissolution or winding up of our affairs, holders are entitled to receive ratably our net assets available to the
shareholders. Holders of our outstanding common stock have no preemptive, conversion or redemption rights. All of the issued and
outstanding shares of our common stock are, and all unissued shares of our common stock, when offered and sold will be, duly authorized,
validly issued, fully paid and nonassessable. To the extent that additional shares of our common stock may be issued in the future,
the relative interests of the then existing shareholders may be diluted.
Our authorized but unissued shares of common
stock are available for future issuances without shareholder approval and could be utilized for a variety of corporate purposes,
including future offerings to raise additional capital, corporate acquisitions and employee benefit plans. The existence of authorized
but unissued and unreserved common stock could render more difficult or discourage an attempt to obtain control of us by means
of a proxy contest, tender offer, merger or otherwise.
Additional information about our common
stock is provided in the accompanying prospectus under “Description of Common Stock.”
Transfer Agent
. The transfer agent
for our common stock is Corporate Stock Transfer, Inc..
Listing
. The shares of our common
stock are currently listed on the NYSE MKT under the symbol “XTNT.”
PLAN OF DISTRIBUTION
We are selling
843,289 shares
of our common stock directly to certain holders of the Indenture Notes. The common stock will be sold at a negotiated price of
$0.5692.
We will be entering into securities purchase
agreements, dated effective as of January 17, 2017, with such investors relating to the sale of these shares.
On the closing date, we will issue the
shares of common stock to the investors. The purchase price for the shares will be paid by each investor by a dollar-for-dollar
offset against all interest due to the investor as of the closing date under the Indenture Notes. Such offset shall have the same
effect as if such investor paid cash to the Company for such shares and the Company used such cash to pay to the investor the interest
so offset. We estimate that the expenses of this offering payable by us will be approximately $75,000, which we will pay from cash
on hand.
The shares are being offered directly
to the investors without a placement agent, underwriter, broker or dealer.
We currently anticipate that the closing
of the sale of such shares will take place on or about January 17, 2017.
The transfer agent for our common stock
is Corporate Stock Transfer, Inc. Our common stock is traded on the NYSE MKT exchange and traded under the symbol “XTNT.”
Foreign Regulatory Restrictions on Purchase
of the Common Stock
No action may be taken in any jurisdiction
other than the United States that would permit a public offering of the common stock or the possession, circulation or distribution
of this prospectus supplement in any jurisdiction where action for that purpose is required. Accordingly, the common stock may
not be offered or sold, directly or indirectly, and neither the prospectus supplement nor any other offering material or advertisements
in connection with the common stock may be distributed or published in or from any country or jurisdiction except under circumstances
that will result in compliance with any applicable rules and regulations of any such country or jurisdiction.
If you purchase shares of common stock
offered by this prospectus supplement, you may be required to pay stamp taxes and other charges under the laws and practices of
the country of purchase, in addition to the offering price listed on the cover page of this prospectus supplement.
INCORPORATION OF
CERTAIN INFORMATION BY REFERENCE
The SEC allows us to incorporate by reference
the information that we file with the SEC, which means that we can disclose important information to you by referring you to those
documents. The information incorporated by reference is considered to be part of the prospectus supplement. These documents may
include periodic reports, such as Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K,
as well as Proxy Statements. Any documents that we subsequently file with the SEC will automatically update and replace the information
previously filed with the SEC. Thus, for example, in the case of a conflict or inconsistency between information set forth in this
prospectus supplement and the accompanying prospectus and information incorporated by reference into this prospectus supplement,
you should rely on the information contained in the document filed later.
This prospectus supplement incorporates
by reference the documents listed below that we previously have filed with the SEC and any additional documents that we may file
with the SEC under Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act between the date of this prospectus and the termination
of the offering of the securities. These documents contain important information about us.
|
|
·
|
Our Annual Report on Form 10-K for the year ended December 31, 2015 (filed on March 24, 2016);
|
|
|
·
|
Our Quarterly Reports on Form 10-Q for the quarters ended March 31, 2016 (filed on May 11, 2016);
June 30, 2016 (filed on August 10, 2016); and September 30, 2016 (filed on November 10, 2016);
|
|
|
·
|
Our definitive proxy statement on Schedule 14A (filed on May 26, 2016);
|
|
|
·
|
Our Current Reports on Form 8-K (other than portions thereof furnished under Item 2.02 or Item 7.01 of
Form 8-K and exhibits accompanying such reports that are related to such items) filed on January 22, 2016, April 4, 2016, April
19, 2016, May 31, 2016, June 17, 2016, August 2, 2016, August 16, 2016, August 19, 2016, September 28, 2016, October 6, 2016, October
11, 2016, October 18, 2016, November 1, 2016, November 7, 2016, November 9, 2016, November 14, 2016, November 15, 2016, November
17, 2016, January 6, 2017, January 11, 2017, January 13, 2017 and January 17, 2017;
|
|
|
·
|
The description of our common stock contained in our registration statement on Form 8-A, filed
on November 5, 2010, as amended by Form 8-A/A, filed on March 4, 2011 pursuant to Section 12 of the Exchange Act and the Registration
Statement on Form 8-A filed on October 15, 2015; and
|
|
|
·
|
All documents filed by us pursuant to Section 13(a), 13(c), 14 or 15(d) of the Exchange Act after
the date of this prospectus and before the termination of the offering.
|
We are not, however, incorporating by reference
any documents, or portions of documents, whether specifically listed above or arising in the future, which are not deemed “filed”
with the SEC.
You can obtain a copy of any or all of
the documents incorporated by reference in this prospectus (other than an exhibit to a document unless that exhibit is specifically
incorporated by reference into that document) from the SEC on its website at www.sec.gov. You also can obtain these documents from
us, free of charge, by visiting our internet website www.xtantmedical.com or by writing to us or calling us at the following address
and phone number:
Xtant Medical Holdings, Inc.
664 Cruiser Lane
Belgrade, MT 59714
Attn: Corporate Secretary
(406) 388-0480
WHERE YOU CAN FIND
MORE INFORMATION
We have filed with the SEC a registration
statement under the Securities Act that registers the distribution of the securities offered under this prospectus supplement and
the accompanying prospectus. The registration statement, including the attached exhibits and schedules and the information incorporated
by reference, contains additional relevant information about our company and the securities. The rules and regulations of the SEC
allow us to omit from this prospectus supplement and the accompanying prospectus certain information included in the registration
statement. In addition, we file annual, quarterly and special reports, proxy statements and other information with the SEC. You
may read and copy this information and the registration statement at the SEC’s Public Reference Room located at 100 F Street,
N.E., Washington D.C. 20549. Please call the SEC at 1-800-SEC-0330 for more information about the operation of the public reference
room.
You may also obtain the documents that
we file electronically on the SEC’s website at hwww.sec.gov or on our website at www.xtantmedical.com. Information contained
on our website is not incorporated by reference herein and does not constitute part of this prospectus.
LEGAL MATTERS
Certain legal matters with respect to the
securities offered hereby have been passed upon our former General Counsel, Jill Gilpin, and by Ballard Spahr LLP.
EXPERTS
The financial statements incorporated
by reference into this prospectus supplement have been audited by EKS&H LLLP, independent certified public accounts, as set
forth in their report thereon appearing in our Annual Report on Form 10-K and incorporated by reference into this prospectus supplement,
and such report is included in reliance upon the authority of such firm as experts in accounting and auditing.
PROSPECTUS

UP TO $50,000,000 OF OUR
COMMON STOCK
PREFERRED STOCK
WARRANTS
From time to time, we may offer up to $50,000,000
in total of:
|
|
·
|
shares of common stock;
|
|
|
·
|
shares of preferred stock;
|
|
|
·
|
warrants to purchase shares of common
stock or preferred stock; or
|
|
|
·
|
any combination of our common stock, preferred
stock or warrants.
|
We may offer the common stock, preferred
stock, and warrants, separately or together, in amounts, at prices and on terms to be set forth in one or more supplements to this
prospectus. The preferred stock and warrants we may offer may be convertible into or exercisable or exchangeable for common or
preferred stock or other securities. When we decide to issue securities, we will provide you with the specific terms and the public
offering price of the securities in prospectus supplements. In the case of shares of preferred stock, these terms will include,
as applicable, the specific title and stated value, and any dividend, liquidation, redemption, conversion, voting and other rights.
You should read this prospectus and any applicable prospectus supplement carefully before you invest. This prospectus may not be
used to offer or sell securities unless accompanied by a prospectus supplement.
Our common stock is listed on the NYSE
MKT exchange and traded under the symbol “BONE.” None of our other securities are currently publicly traded. We may
sell these securities to or through underwriters and also to other purchasers or through agents. We will set forth the names of
any underwriters or agents in an accompanying prospectus supplement, if applicable.
The last reported sale price of our
common stock on the NYSE MKT on March 28, 2014 was $0.87 per share. As of March 28, 2014, the aggregate market value of the
voting and non-voting common equity held by non-affiliates, computed by reference to the price at which the common equity was
last sold on that date, was approximately $32,802,202, based on 54,858,458 shares of outstanding common stock, of which
approximately 37,703,681 were held by non-affiliates. Pursuant to General Instruction I.B.6 of Form S-3, in no event will we
sell securities in a public primary offering with a value exceeding more than one-third of our public float in any 12-month
period so long as our public float remains below $75.0 million. We have not offered any securities pursuant to General
Instruction I.B.6 of Form S-3 during the 12 calendar months prior to and including the date of this prospectus.
Investing in our
securities involves risks. Please see “Risk Factors” beginning on page 2 for more information. You should read
carefully this prospectus, the documents incorporated by reference in this prospectus and any prospectus supplement before you
invest.
Neither the Securities
and Exchange Commission nor any state securities commission has approved or disapproved of these securities or passed upon the
adequacy or accuracy of this prospectus. Any representation to the contrary is a criminal offense.
The date of this prospectus is
, 2014
TABLE OF CONTENTS
ABOUT THIS PROSPECTUS
This prospectus is part of a registration
statement filed with the Securities and Exchange Commission (“SEC”) using a “shelf” registration process.
Under this shelf process, we may from time to time offer up to $50,000,000 in total of shares of common stock, $0.000001 par value
per share, shares of preferred stock, $0.000001 par value per share, or warrants to purchase shares of common stock or preferred
stock, each at prices and on terms to be determined at the time of sale. The common stock, preferred stock and warrants are collectively
referred to in this prospectus as “securities.” The securities offered pursuant to this prospectus may be one or more
series of issuances. The total offering price of the securities will not exceed $50,000,000.
This prospectus provides you with a general
description of the securities we may offer. Each time we sell securities, we will provide a prospectus supplement with specific
information about the terms of that offering. The prospectus supplement may also add, update or change information contained in
this prospectus. If there is any inconsistency between the information in this prospectus and the prospectus supplement, you should
rely on the information in the prospectus supplement. You should read both this prospectus and any prospectus supplement together
with the additional information described below under the heading “Where You Can Find More Information” and “Incorporation
of Certain Information by Reference.”
The registration statement that contains
this prospectus, including the exhibits to the registration statement and the information incorporated by reference, contains additional
information about the securities offered under this prospectus. The registration statement can be read at the SEC website or at
the SEC offices mentioned below under the heading “Where You Can Find More Information.”
We have not authorized anyone to provide
any information or to make any representations other than those contained or incorporated by reference in this prospectus or in
any accompanying prospectus supplement. We take no responsibility for, and can provide no assurance as to the reliability of, any
other information that others may give you.
Neither this prospectus nor any accompanying
prospectus supplement constitutes an offer to sell or the solicitation of an offer to buy any securities other than the registered
securities to which they relate, nor do this prospectus or any prospectus supplement constitute an offer to sell or the solicitation
of an offer to buy securities in any jurisdiction to any person to whom it is unlawful to make such offer or solicitation in such
jurisdiction.
You should not assume that the information
in this prospectus or any supplement to this prospectus is accurate at any date other than the date indicated on the cover page
of these documents or that any information we have incorporated by reference is correct on any date subsequent to the date of the
document incorporated by reference.
SUMMARY
This summary contains a general summary
of the information contained in this prospectus. It may not include all the information that is important to you. You should read
the entire prospectus, the prospectus supplement delivered with the prospectus, if any, and the documents incorporated by reference
before making an investment decision.
About Bacterin International Holdings,
Inc.
We develop, manufacture and market biologics
products to domestic and international markets through our biologics division. Our bone graft products are used in a variety of
applications including enhancing fusion in spine surgery, relief of back pain through facet joint stabilization, promotion of bone
growth in foot and ankle surgery, promotion of skull healing following neurosurgery and subchondral bone repair in knee and other
joint surgeries. Our acellular dermis scaffolds are utilized in wound care and plastic and reconstructive procedures.
Our medical devices division develops coatings
for medical devices and custom surgical instruments for use with allografts processed by our biologics division. Our medical devices
division also works with our biologics division to produce and distribute OsteoSelect® DBM putty, an osteoinductive product
used by surgeons as a bone void filler in the extremities and pelvis. DBM putty is considered a combination product by regulatory
agencies — both a tissue and a medical device.
We are a Delaware corporation. Our executive
offices are located at 664 Cruiser Lane, Belgrade, Montana 59714 and our telephone number is (406) 388-0480. Our website
is located at
www.bacterin.com
. The information on our website is not part of this prospectus.
Securities We are Offering
We may offer any of the following securities
from time to time:
|
|
·
|
shares of our common stock;
|
|
|
·
|
shares of our preferred stock;
|
|
|
·
|
warrants to purchase shares of our preferred stock or common stock;
or
|
|
|
·
|
any combination of our common stock, preferred stock, or warrants.
|
When we use the term “securities”
in this prospectus, we mean any of the securities we may offer with this prospectus, unless we say otherwise. The total dollar
amount of all securities that we may issue will not exceed $50,000,000. This prospectus, including the following summary, describes
the general terms that may apply to the securities. We will describe the specific terms of any particular securities that we may
offer in a separate supplement to this prospectus.
Common Stock.
We may
offer shares of our common stock. Our common stock currently is listed on the NYSE MKT under the symbol “BONE.”
Preferred Stock.
We may
offer preferred stock in one or more series. For any particular series we offer, the applicable prospectus supplement will describe
the specific designation, the aggregate number of shares offered, the rate and periods, or manner of calculating the rate and periods,
for dividends, if any, the stated value and liquidation preference amount, if any, the voting rights, if any, the terms on which
the series will be convertible into or exchangeable for other securities or property, if any, the redemption terms, if any, and
any other specific terms.
Warrants.
We may offer
warrants to purchase our common stock and preferred stock. For any particular warrants we offer, the applicable prospectus supplement
will describe the underlying security; expiration date; the exercise price or the manner of determining the exercise price; the
amount and kind, or the manner of determining the amount and kind, of any security to be delivered by us upon exercise; and any
other specific terms.
Listing.
If any securities
will be listed or quoted on a securities exchange or quotation system, the applicable prospectus supplement will say so.
RISK FACTORS
An investment in our securities
involves a high degree of risk. Before making an investment decision, you should consider carefully the risks discussed below
and under the sections captioned “Risk Factors” set forth in the documents and reports filed by us with the SEC,
that are incorporated by reference into this prospectus, including in our most recent Annual Report on Form 10-K, as revised
or supplemented by our Quarterly Reports on Form 10-Q, each of which are on file with the SEC and are incorporated herein by
reference, as well as any risks described in any applicable prospectus supplement, before deciding whether to invest in our
securities. Our business, financial condition or results of operations could be materially adversely affected by any of these
risks. The trading price of our securities could decline due to any of these risks, and you may lose all or part of your
investment.
We may not be able to meet financial
or other covenant requirements in our current credit facility, and we may not be able to successfully negotiate waivers or a new
credit agreement to cure any covenant violations.
Our debt agreements with ROS Acquisition
Offshore LP (“ROS”) contain representations, warranties, fees, affirmative and negative covenants, including a minimum
cash balance and minimum revenue amounts by quarter, and default provisions, which include departures in key management, if not
remedied within 90 days. A breach of any of these covenants could result in a default under these agreements. Upon the occurrence
of an event of default under our debt agreements, our lender could elect to declare all amounts outstanding to be immediately due
and payable and terminate all commitments to extend further credit. If our lender accelerates the repayment of borrowings, we may
not have sufficient assets to repay our indebtedness. Also, should there be an event of default, or should we need to obtain waivers
following an event of default, we may be subject to higher borrowing costs and/or more restrictive covenants in future periods.
In addition, to secure the performance of our obligations under the ROS facility, we pledged substantially all of our assets, including
our intellectual property, to ROS. Our failure to comply with the covenants under the ROS facility could result in an event of
default, the acceleration of our debt and the loss of our assets.
We are not currently profitable and
we will need to raise additional funds in the future; however, additional funds may not be available on acceptable terms, or at
all.
We have substantial operating expenses
associated with the sales and marketing of our products. The sales and marketing expenses are anticipated to be funded from operating
cash flow. There can be no assurance that we will have sufficient access to liquidity or cash flow to meet our operating expenses
and other obligations. If we do not increase our revenue or reduce our expenses, we will need to raise additional capital, which
would result in dilution to our stockholders, or seek additional loans. The incurrence of indebtedness would result in increased
debt service obligations and could require us to agree to operating and financial covenants that would restrict our operations.
Financing may not be available in amounts or on terms acceptable to us, if at all. Any failure by us to raise additional funds
on terms favorable to us, or at all, could result in our inability to pay our expenses as they come due, limit our ability to expand
our business operations, and harm our overall business prospects.
We may not be able to raise capital or,
if we can, it may not be on favorable terms. We may seek to raise additional capital through public or private equity financings,
partnerships, joint ventures, dispositions of assets, debt financings or restructurings, bank borrowings or other sources. To obtain
additional funding, we may need to enter into arrangements that require us to relinquish rights to certain technologies, products
and/or potential markets. If adequate funds are not otherwise available, we would be forced to curtail operations significantly,
including reducing our sales and marketing expenses which could negatively impact product sales and we could even be forced to
cease operations, liquidate our assets and possibly even seek bankruptcy protection.
We may not continue to satisfy the
continued listing requirements of the NYSE MKT exchange.
We are currently listed on the NYSE MKT
exchange, which imposes both objective and subjective requirements for continued listing. Continued listing criteria include
the financial condition of the company, market capitalization, shareholder equity, total assets, annual revenue, and low selling
price. Our common stock is currently trading at less than $1.00 per share, we are operating at a loss, we have negative shareholder
equity, and our market capitalization, total assets and annual revenue are all currently less than $50 million, so our continued
listing is at risk. If the NYSE MKT determines that we fail to satisfy the requirements for continued listing, we could be
de-listed from the exchange, which could result in reduced liquidity for our shareholders. There can be no assurance that
we will satisfy the continued listing requirements of the NYSE MKT or that we will continue to be listed on any exchange.
On May 13, 2013, we received a deficiency
notice from the NYSE MKT exchange notifying us that we are not in compliance with Section 1003(a)(iii) of the Company Guide with
stockholders’ equity of less than $6,000,000 and net losses in five of our most recent fiscal years and Section 1003(a)(ii)
with stockholders’ equity of less than $4,000,000 and net losses in three of our four most recent fiscal years. On June 12,
2013 we submitted a plan to regain compliance with the continued listing requirements, and on June 21, 2013 the NYSE MKT informed
us of the acceptance of our plan and gave us an extension until November 13, 2014 to regain compliance with the continued listing
standards. On November 19, 2013, we received another letter from the NYSE MKT notifying us that we are not in compliance with Section
1003(a)(i) of the Company Guide with stockholders’ equity of less than $2,000,000 as of September 30, 2013 and net losses
in two of three of our most recent fiscal years, and we submitted an amended plan to regain compliance. We will continue to be
subject to periodic review by the NYSE MKT during the extension period and failure to make progress consistent with our Plan or
to regain compliance by the end of the extension period could result in our delisting from the Exchange.
In order to regain compliance, we will
either need to increase our market capitalization or shareholders’ equity. In order to increase our shareholder’s equity,
we may need to raise substantial equity capital, which would be dilutive to existing shareholders and may require shareholder approval.
We currently have less than 20,000,000 shares available for issuance on a fully diluted basis. To raise sufficient equity capital
to achieve $6 million in shareholder equity, we may need to increase the number of authorized shares available for issuance, which
requires shareholder approval. There can be no assurance that we will obtain any necessary shareholder approval or raise sufficient
equity capital to regain compliance with the NYSE MKT continued listing standards.
The impact of United States healthcare
reform legislation remains uncertain.
In 2010 federal legislation to reform the
United States healthcare system was enacted into law. The law was upheld by a Supreme Court decision announced in June 2012. The
legislation is far-reaching and is intended to expand access to health insurance coverage, improve quality and reduce costs over
time. Among other things, the new law imposes a 2.3 percent excise tax on medical devices beginning January 2013, which applies
to United States sales of our medical device products, including our wound drains and OsteoSelect
®
DBM putty. Due
to multi-year pricing agreements and competitive pricing pressure in our industry, there can be no assurance that we will be able
to pass the cost of the device tax on to our customers. Other provisions of this legislation, including Medicare provisions aimed
at improving quality and decreasing costs, comparative effectiveness research, an independent payment advisory board, and pilot
programs to evaluate alternative payment methodologies, could meaningfully change the way healthcare is developed and delivered.
We cannot predict the impact of this legislation or other healthcare programs and regulations that may ultimately be implemented
at the federal or state level, the effect of any future legislation or regulation in the United States or internationally or whether
any changes will have the effect of lowering prices for our products or reducing medical procedure volumes.
We face risks and uncertainties relating
to an OIG subpoena.
In February 2013, we received a subpoena
from the Office of the Inspector General of the U.S. Department of Health and Human Services (“OIG”) seeking documents
in connection with an investigation into possible false or otherwise improper claims submitted to Medicare. The subpoena requested
documents related to physician referral programs operated by the Company, which we believe refers to the Company’s prior
practice of compensating physicians for performing certain educational and promotional services on behalf of the Company. This
program was discontinued in 2010. We provided an initial response to the OIG subpoena and have not received any further correspondence
or requests from the OIG. Although it does not appear that the OIG is actively pursuing the investigation at the present time,
we cannot assure you that the OIG will not resume the investigation in the future. Any further investigation by the OIG could divert
management’s attention from business demands and subject us to significant legal expenses.
Pricing pressure and cost containment
measures
could have a negative impact on our future operating results.
Pricing pressure has increased in our industry
due to continued consolidation among healthcare providers, trends toward managed care, the shift towards government becoming the
primary payer of healthcare expenses, and government laws and regulations relating to reimbursement and pricing generally. Pricing
pressure, reductions in reimbursement levels or coverage or other cost containment measures could unfavorably affect our future
operating results and financial condition.
Future regulatory action remains
uncertain.
We operate in a highly regulated environment,
and any legal or regulatory action could be time-consuming and costly. If we fail to comply with all applicable laws, standards
and regulations, action by the FDA or other regulatory agencies could result in significant restrictions, including restrictions
on the marketing or use of our products or the withdrawal of products from the market. Any such restrictions or withdrawals
could materially affect our business and operations. In addition, governmental authorities could impose fines, seize our
inventory of products, or force us to recall any product already in the market if we fail to comply with governmental regulations.
Competition from former Chief Executive
Officer
We believe our former Chief Executive Officer,
Guy Cook, has acquired an ownership interest in a tissue bank that sells competitive products. Because our former CEO has in depth
knowledge about our customers, employees, consultants, products, policies, practices and prospects, and is not bound by a non-compete
agreement, we may be adversely affected by increased competition with that business.
Many competitive products exist and
more will be developed, and we may not be able to successfully compete because we are smaller and have fewer financial resources.
Our business is in a very competitive and
evolving field. Rapid new developments in this field have occurred over the past few years, and are expected to continue
to occur. Other companies already have competing products available or may develop products to compete with ours. Many of
these products have short regulatory timeframes and our competitors, many with more substantial development resources, may be able
to develop competing products that are equal to or better than ours. This may make our products obsolete or undesirable by
comparison and reduce our revenue. Our success will depend, in large part, on our ability to maintain a competitive position
concerning our intellectual property, and to develop new technologies and new applications for our technologies. Many of
our competitors have substantially greater financial and technical resources, as well as greater production and marketing capabilities,
and our ability to compete remains uncertain.
The medical community and the general
public may perceive synthetic materials and growth factors as safer, which could have a material adverse effect on our business.
Members of the medical community and the
general public may perceive synthetic materials and growth factors as safer than our allograft-based bone tissue products. Our
products may be incapable of competing successfully with synthetic bone graft substitutes and growth factors developed and commercialized
by others, which could have a material adverse effect on our business, financial condition and results of operations.
Negative publicity concerning methods
of human tissue recovery and screening of donor tissue in the industry in which we operate may reduce demand for our allografts
and impact the supply of available donor tissue.
Media reports or other negative publicity
concerning both improper methods of tissue recovery from donors and disease transmission from donated tissue may limit widespread
acceptance of our allografts. Unfavorable reports of improper or illegal tissue recovery practices, both in the United States
and internationally, as well as incidents of improperly processed tissue leading to transmission of disease, may broadly affect
the rate of future tissue donation and market acceptance of allograft technologies. Potential patients may not be able to
distinguish our allografts, technologies and the tissue recovery and the processing procedures from those of our competitors or
others engaged in tissue recovery. In addition, families of potential donors may become reluctant to agree to donate tissue
to for-profit tissue processors.
We are highly dependent on the availability
of human donors; any disruptions could cause our customers to seek alternative providers or technologies.
We are highly dependent on our ability
to obtain donor cadavers as the raw material for many of our products. The availability of acceptable donors is relatively
limited and we compete with many other companies for this limited availability. The availability of donors is also impacted
by regulatory changes, general public opinion of the donor process and our reputation for our handling of the donor process.
In addition, due to seasonal changes in the mortality rates, some scarce tissues are at times in short supply. Any disruption
in the supply of this crucial raw material could have significant consequences for our revenue, operating results and continued
operations.
We will need to continue to innovate
and develop new products to be desirable to our customers.
The markets for our products and services
are characterized by rapid technological change, frequent new introductions, changes in customers’ demands and evolving industry
standards. Accordingly, we will need to continue to innovate and develop additional products. These efforts can be
costly, subject to long development and regulatory delays and may not result in products approved for sale. These costs may
hurt operating results and may require additional capital. If additional capital is not available, we may be forced to curtail
development activities. In addition, any failure on our behalf to react to changing market conditions could create an opportunity
for other market participants to capture a critical share of the market within a short period of time.
Our success will depend on our ability
to engage and retain qualified technical personnel who are difficult to attract.
Our success will depend on our ability
to attract and retain qualified technical personnel to assist in research and development, testing, product implementation, low-scale
production and technical support. The demand for such personnel is high and the supply of qualified technical personnel is
limited. A significant increase in the wages paid by competing employers could result in a reduction of our technical work
force and increases in the wage rates that we must pay or both. If either of these events were to occur, our cost structure
could increase and our growth potential could be impaired.
Loss of key members of our management
who we need to succeed could adversely affect our business.
We are highly dependent on the services
of key members of our management team, and the loss of any of their services could have an adverse effect on our future operations.
We do not currently maintain key-man life insurance policies insuring the life of any member of our management team.
We are highly dependent on the continued
availability of our facilities and would be harmed if they were unavailable for any prolonged period of time.
Any failure in the physical infrastructure
of our facilities or services could lead to significant costs and disruptions that could reduce our revenues and harm our business
reputation and financial results. We are highly reliant on our Belgrade, Montana facilities. Any natural or man-made
event that impacts our ability to utilize these facilities could have a significant impact on our operating results, reputation
and ability to continue operations. The regulatory process for approval of facilities is time-consuming and our ability to
rebuild facilities would take a considerable amount of time and expense and cause a significant disruption in service to our customers.
Further, the FDA or some other regulatory agency could identify deficiencies in future inspections of our facilities or our supplies
that could disrupt our business, reducing profitability.
We will be required to invest in
facilities and equipment on a continuing basis, which will put pressure on us to finance these investments.
We have invested, and intend to continue
to invest, in facilities and state-of-the-art equipment in order to increase, expand or update our capabilities and facilities.
Changes in technology or sales growth beyond currently established production capabilities, which we anticipate, will require further
investment. We currently anticipate that we will need to spend between $4 and $5 million over the next five years in order to increase,
expand or update our existing facilities to meet our expected growth over that period. However, there can be no assurance
that we will generate sufficient funds from operations to maintain our existing facilities and equipment or to finance any required
capital investments or that other sources of funding will be available. Additionally, there can be no guarantee that any future
expansion will not negatively affect earnings.
Future revenue will depend on our
ability to increase sales.
We currently sell our products through
direct sales by our employees and indirectly through distributor relationships. We incurred increased sales and marketing
expenses in building and expanding our direct sales force, and there can be no assurance that we will generate increased sales
as a result of this effort.
There may be fluctuations in our
operating results, which will impact the price of our securities.
Significant annual and quarterly fluctuations
in our results of operations may be caused by, among other factors, our volume of revenues, the timing of new product or service
announcements, releases by us and our competitors in the marketplace of new products or services, seasonality and general economic
conditions. There can be no assurance that the level of revenues achieved by us in any particular fiscal period will not
be significantly lower than in other comparable fiscal periods. Our expense levels are based, in part, on our expectations
as to future revenues. As a result, if future revenues are below expectations, net income or loss may be disproportionately
affected by a reduction in revenues, as any corresponding reduction in expenses may not be proportionate to the reduction in revenues.
We are dependent on the ability of
our licensees and development partners for obtaining regulatory approvals and market acceptance of their products, for which we
may have no control.
Our success may depend on our ability,
or that of our licensees, to obtain timely regulatory approval for products employing our technology. Moreover, our success
may also depend on whether, and how quickly, our licensees gain market acceptance of products incorporating our technology, compared
to competitors using competing technologies.
Our revenues will depend upon prompt
and adequate reimbursement from public and private insurers and national health systems.
Political, economic and regulatory influences
are subjecting the healthcare industry in the United States to fundamental change. The ability of hospitals to pay fees for
allograft bone tissue products depends in part on the extent to which reimbursement for the costs of such materials and related
treatments will continue to be available from governmental health administration authorities, private health coverage insurers
and other organizations. We may have difficulty gaining market acceptance for our products if government and third-party
payors do not provide adequate coverage and reimbursement to hospitals. Major third-party payors of hospital services and
hospital outpatient services, including Medicare, Medicaid and private healthcare insurers, annually revise their payment methodologies,
which can result in stricter standards for reimbursement of hospital charges for certain medical procedures or the elimination
of reimbursement. Further, Medicare, Medicaid and private healthcare insurer cutbacks could create downward price pressure
on our products.
Our operating results will be harmed
if we are unable to effectively manage and sustain our future growth.
We might not be able to manage our future
growth efficiently or profitably. Our business is unproven on a large scale and actual revenue and operating margins, or
revenue and margin growth, may be less than expected. If we are unable to scale our production capabilities efficiently,
we may fail to achieve expected operating margins, which would have a material and adverse effect on our operating results.
Growth may also stress our ability to adequately manage our operations, quality of products, safety and regulatory compliance.
In order to grow, we may be required to obtain additional financing, which may increase our indebtedness or result in dilution
to our stockholders. Further, there can be no assurance that we would be able to obtain any additional financing.
Future business combinations or acquisitions
may be difficult to integrate and cause our attention to be diverted.
We may pursue various business combinations
with other companies or strategic acquisitions of complementary businesses, product lines or technologies. There can be no
assurance that such acquisitions will be available at all, or on terms acceptable to us. These transactions may require additional
financing which may increase our indebtedness or outstanding shares, resulting in dilution to stockholders. The inability
to obtain such future financing may inhibit our growth and operating results. Integration of acquisitions or additional products
can be time consuming, difficult and expensive and may significantly impact operating results. Furthermore, the integration
of any acquisition may divert management’s time and resources from our core business. We may sell some or all of our
product lines to other companies or may agree to combine with another company. Selling some of our product lines may inhibit
our ability to generate positive operating results going forward.
We may be subject to future product
liability litigation that could be expensive and our insurance coverage may not be adequate in a catastrophic situation.
Although we are not currently subject to
any product liability proceedings, and we have no reserves for product liability disbursements, we may incur material liabilities
relating to product liability claims in the future, including product liability claims arising out of the usage of our products.
We currently carry product liability insurance, however, our insurance coverage and any reserves we may maintain in the future
for product related liabilities may not be adequate and our business could suffer material adverse consequences.
We may implement a product recall
or voluntary market withdrawal due to product defects or product enhancements and modifications, which would significantly increase
our costs.
The manufacturing and marketing of our
biologic products, medical devices and coating technologies involves an inherent risk that our products may prove to be defective.
In that event, we may voluntarily implement a recall or market withdrawal or may be required to do so by a regulatory authority.
A recall or withdrawal of one of our products, or a similar product manufactured by another manufacturer, could impair sales of
the products we market as a result of confusion concerning the scope of the recall or withdrawal, or as a result of the damage
to our reputation for quality and safety.
U.S. governmental regulation could
restrict the use of our products or our procurement of tissue.
In the United States, the procurement and
transplantation of allograft bone tissue is subject to federal law pursuant to the National Organ Transplant Act, or NOTA, a criminal
statute which prohibits the purchase and sale of human organs used in human transplantation, including bone and related tissue,
for “valuable consideration.” NOTA permits reasonable payments associated with the removal, transportation, processing,
preservation, quality control, implantation and storage of human bone tissue. We provide services in all of these areas in the
United States, with the exception of removal and implantation, and receive payments for all such services. We make payments to
certain of our clients and tissue banks for their services related to recovering allograft bone tissue on our behalf. If NOTA is
interpreted or enforced in a manner which prevents us from receiving payment for services we render or which prevents us from paying
tissue banks or certain of our clients for the services they render for us, our business could be materially and adversely affected.
We are engaged through our marketing employees,
independent sales agents and sales representatives in ongoing efforts designed to educate the medical community as to the benefits
of our products, and we intend to continue our educational activities. Although we believe that NOTA permits payments in connection
with these educational efforts as reasonable payments associated with the processing, transportation and implantation of our products,
payments in connection with such education efforts are not exempt from NOTA’s restrictions and our inability to make such
payments in connection with our education efforts may prevent us from paying our sales representatives for their education efforts
and could adversely affect our business and prospects. No federal agency or court has determined whether NOTA is, or will be, applicable
to every allograft bone tissue-based material which our processing technologies may generate. Assuming that NOTA applies to our
processing of allograft bone tissue, we believe that we comply with NOTA, but there can be no assurance that more restrictive interpretations
of, or amendments to, NOTA will not be adopted in the future which would call into question one or more aspects of our method of
operations.
Our business is subject to continuing
regulatory compliance by the FDA and other authorities which is costly and could result in delays in the commercialization of our
products.
As a manufacturer and marketer of medical
devices, we are subject to extensive regulation by the FDA and the Center for Medicare Services of the U.S. Department of Health
and Human Services and other federal governmental agencies and, in some jurisdictions, by state and foreign governmental authorities.
These regulations govern the introduction of new medical devices, the observance of certain standards with respect to the design,
manufacture, testing, labeling, promotion and sales of the devices, the maintenance of certain records, the ability to track devices,
the reporting of potential product defects, the import and export of devices and other matters. We are facing an increasing
amount of scrutiny and compliance costs as more states are implementing regulations governing medical devices, pharmaceuticals
and/or biologics which affect many of our products.
Medical devices that incorporate coatings
technology are subject to FDA regulation and compliance. Generally, any medical device manufacturer that wishes to incorporate
our coatings technology into its products will be responsible for obtaining FDA approval for the medical devices it intends to
market though we will assist in the 510(k) filing submitted by licensees. The FDA process can take several months to several
years in the United States. The time required to obtain approval for international sales may be longer or shorter, depending
on the laws of the particular country. There can be no assurance that our licensees will be able to obtain FDA or international
approval on a timely basis. The FDA may also require the more extensive Premarket Approval Application, or PMA, process for
certain products, which requires an additional level of FDA scientific review to ensure the safety and effectiveness of such devices.
Approval or clearance may place substantial restrictions on the indications for which the product may be marketed or to whom it
may be marketed, warnings that may be required to accompany the product or additional restrictions placed on the sale and/or use
of the product. Changes in regulations or adoption of new regulations could also cause delays in obtaining product approval.
In addition, regulatory approval is subject to continuing compliance with regulatory standards, and product approval is subject
to withdrawal if a licensee fails to comply with standards, or if an unforeseen event should occur concerning a product.
Significant delays in obtaining product approval could have a significantly detrimental impact on our business.
Human tissues intended for transplantation
have been regulated by the FDA since 1993. In May 2005, three new comprehensive regulations went into effect that address manufacturing
activities associated with human cells, tissues and cellular and tissue-based products, or HCT/Ps. The first requires that companies
that produce and distribute HCT/Ps register with the FDA. The second provides criteria that must be met for donors to be eligible
to donate tissues and is referred to as the “Donor Eligibility” rule. The third rule governs the processing and distribution
of the tissues and is often referred to as the “Current Good Tissue Practices” rule. The “Current Good
Tissue Practices” rule covers all stages of allograft processing, from procurement of tissue to distribution of final allografts.
Together they are designed to ensure that sound, high quality practices are followed to reduce the risk of tissue contamination
and of communicable disease transmission to recipients. These regulations increased regulatory scrutiny within the industry
in which we operate and have lead to increased enforcement action which affects the conduct of our business. In addition, these
regulations can increase the cost of tissue recovery activities.
Other regulatory entities include state
agencies with statutes covering tissue banking. Regulations issued by Florida, New York, California and Maryland will be
particularly relevant to our business. Most states do not currently have tissue banking regulations. It is possible
that others may make allegations against us or against donor recovery groups or tissue banks about non-compliance with applicable
FDA regulations or other relevant statutes or regulations. Allegations like these could cause regulators or other authorities
to take investigative or other action, or could cause negative publicity for our business and the industry in which we operate.
Our products may be subject to regulation
in the EU as well should we enter that market. In the European Union, or EU, regulations, if applicable, differ from one
EU member state to the next. Because of the absence of a harmonized regulatory framework and the proposed regulation for
advanced therapy medicinal products in the EU, as well as for other countries, the approval process for human derived cell or tissue
based medical products may be extensive, lengthy, expensive and unpredictable. Some of our products may be subject to European
Union member states’ regulations that govern the donation, procurement, testing, coding, traceability, processing, preservation,
storage, and distribution of human tissues and cells and cellular or tissue-based products. Some EU member states have their
own tissue banking regulations.
Clinical trials can be long, expensive
and ultimately uncertain which could jeopardize our ability to obtain regulatory approval and market our products.
Clinical trials are required to develop
products, gain market acceptance and obtain 510(k) certifications from the FDA. These trials often take several years to
execute and are subject to factors within and outside of our control. The outcome of any trial is uncertain and may have a significant
impact on the success of our current and future products and future profits.
The commencement or completion of any clinical
trial may be delayed or halted for numerous reasons, including, but not limited to, a regulatory body placing clinical trials on
hold, patients not enrolling in clinical trials at the rate we expect, patients experiencing adverse side effects, third party
contractors failing to perform in accordance with our anticipated schedule or consistent with good clinical practices, inclusive
or negative interim trial results or our inability to obtain sufficient quantities of raw materials to produce our products.
Our development costs may increase if we have material delays in clinical trials or if we need to perform more or larger clinical
trials than planned. If this occurs, our financial results and the commercial prospects for our products may be harmed.
Product pricing (and, therefore,
profitability) is subject to regulatory control which could impact our revenue and financial performance.
The pricing and profitability of our products
may become subject to control by the government and other third-party payors. The continuing efforts of governmental and
other third-party payors to contain or reduce the cost of healthcare through various means may adversely affect our ability to
successfully commercialize our products. In most foreign markets, the pricing and/or profitability of certain diagnostics
and prescription pharmaceuticals are subject to governmental control. In the United States, we expect that there will continue
to be federal and state proposals to implement similar governmental control though it is unclear which proposals will ultimately
become law, if any. Changes in prices, including any mandated pricing, could impact our revenue and financial performance.
Failure to protect our intellectual
property rights could result in costly and time consuming litigation and our loss of any potential competitive advantage.
Our success will depend, to a large extent,
on our ability to successfully obtain and maintain patents, prevent misappropriation or infringement of intellectual property,
maintain trade secret protection, and conduct operations without violating or infringing on the intellectual property rights of
third parties. There can be no assurance that our patented and patent-pending technologies will provide us with a competitive
advantage, that we will be able to develop or acquire additional technology that is patentable, or that third parties will not
develop and offer technologies which are similar to ours. Moreover, we can provide no assurance that confidentiality agreements,
trade secrecy agreements or similar agreements intended to protect unpatented technology will provide the intended protection.
Intellectual property litigation is extremely expensive and time-consuming, and it is often difficult, if not impossible, to predict
the outcome of such litigation. A failure by us to protect our intellectual property could have a materially adverse effect
on our business and operating results and our ability to successfully compete in this industry.
We may not be able to obtain or protect
our proprietary rights relating to our products without resorting to costly and time consuming litigation.
We may not be able to obtain, maintain
and protect certain proprietary rights necessary for the development and commercialization of our products or product candidates.
Our commercial success will depend in part on obtaining and maintaining patent protection on our products and successfully defending
these patents against third-party challenges. Our ability to commercialize our products will also depend in part on the patent
positions of third parties, including those of our competitors. The patent positions of pharmaceutical and biotechnology
companies can be highly uncertain and involve complex legal and factual questions. Accordingly, we cannot predict with certainty
the scope and breadth of patent claims that may be afforded to other companies’ patents. We could incur substantial
costs in litigation if we are required to defend against patent suits brought by third parties, or if we initiate suits to protect
our patent rights.
In addition to the risks involved with
patent protection, we also face the risk that our competitors will infringe on our trademarks. Any infringement could
lead to a likelihood of confusion and could result in lost sales.
There can be no assurance that we will
prevail in any claims we make to protect our intellectual property.
Future protection for our proprietary
rights is uncertain which may impact our ability to successfully compete in our industry.
The degree of future protection for our
proprietary rights is uncertain. We cannot ensure that:
|
|
·
|
we were the first to make the inventions covered by each of our patent
applications;
|
|
|
·
|
we were the first to file patent applications for these inventions;
|
|
|
·
|
others will not independently develop similar or alternative technologies
or duplicate any of our technologies;
|
|
|
·
|
any of our pending patent applications will result in issued patents;
|
|
|
·
|
any of our issued patents or those of our licensors will be valid
and enforceable;
|
|
|
·
|
any patents issued to us or our collaborators will provide a basis
for commercially viable products or will provide us with any competitive advantages or will not be challenged by third parties;
|
|
|
·
|
we will develop additional proprietary technologies that are patentable;
|
|
|
·
|
the patents of others will not have a material adverse effect on our
business rights; or
|
|
|
·
|
the measures we rely on to protect the intellectual property underlying
our products will be adequate to prevent third parties from using our technology, all of which could harm our ability to compete
in the market.
|
Our success depends on our ability
to avoid infringing on the intellectual property rights of third parties which could expose us to litigation or commercially unfavorable
licensing arrangements.
Our commercial success depends in part
on our ability and the ability of our collaborators to avoid infringing patents and proprietary rights of third parties.
Third parties may accuse us or our collaborators of employing their proprietary technology in our products, or in the materials
or processes used to research or develop our products, without authorization. Any legal action against our collaborators
or us claiming damages and/or seeking to stop our commercial activities relating to the affected products, materials and processes
could, in addition to subjecting us to potential liability for damages, require our collaborators or us to obtain a license to
continue to utilize the affected materials or processes or to manufacture or market the affected products. We cannot predict
whether we or our collaborators would prevail in any of these actions or whether any license required under any of these patents
would be made available on commercially reasonable terms, if at all. If we are unable to obtain such a license, we or our
collaborators may be unable to continue to utilize the affected materials or processes or manufacture or market the affected products
or we may be obligated by a court to pay substantial royalties and/or other damages to the patent holder. Even if we are
able to obtain such a license, the terms of such a license could substantially reduce the commercial value of the affected product
or products and impair our prospects for profitability. Accordingly, we cannot predict whether or to what extent the commercial
value of the affected product or products or our prospects for profitability may be harmed as a result of any of the liabilities
discussed above. Furthermore, infringement and other intellectual property claims, with or without merit, can be expensive
and time-consuming to litigate and can divert management’s attention from our core business. We may be unable to obtain
and enforce intellectual property rights to adequately protect our products and related intellectual property.
Others may claim an ownership interest
in our intellectual property which could expose us to litigation and have a significant adverse effect on our prospects.
A third-party may claim an ownership interest
in our intellectual property. While we believe we own 100% of the right, title and interest in the patents for which we have
applied and our other intellectual property, including that which we license from third parties, we cannot guarantee that a third-party
will not, at some time, assert a claim or an interest in any of such patents or intellectual property. A successful challenge
or claim by a third party to our patents or intellectual property could have a significant adverse effect on our prospects.
Litigation may result in financial
loss and/or impact our ability to sell our products going forward.
We intend to vigorously defend any future
intellectual property litigation that may arise but there can be no assurance that we will prevail in these matters. An unfavorable
judgment may result in a financial burden on us. An unfavorable judgment may also result in restrictions on our ability to
sell certain products and therefore may impact future operating results.
Because we became public through
a reverse merger, and our stock is currently trading below $1.00 per share, we may not be able to attract the attention of major
brokerage firms or certain investors.
There are coverage risks associated with
our becoming public through a reverse merger, including, among other things, security analysts of major brokerage firms may not
provide coverage of us since there is no incentive to brokerage firms to recommend the purchase of our common stock. In addition,
we may not attract the attention of major brokerage firms and certain investors due to our low stock price. We cannot assure you
that brokerage firms would want to conduct any public offerings on our behalf in the future.
The market price of our common stock
is extremely volatile, which may affect our ability to raise capital in the future and may subject the value of your investment
to sudden decreases.
The market price for securities of biotechnology
companies, including ours, historically has been highly volatile, and the market from time to time has experienced significant
price and volume fluctuations that are unrelated to the operating performance of such companies. Fluctuations in the trading price
or liquidity of our common stock may harm the value of your investment in our securities.
Factors that may have a significant impact
on the market price and marketability of our securities include:
|
|
·
|
announcements of technological innovations or new commercial products
by us, our collaborative partners or our present or potential competitors;
|
|
|
·
|
our issuance of debt, equity or other securities, which we need to
pursue to generate additional funds to cover our operating expenses;
|
|
|
·
|
our quarterly operating results;
|
|
|
·
|
developments or disputes concerning patent or other proprietary rights;
|
|
|
·
|
developments in our relationships with employees, suppliers or collaborative
partners;
|
|
|
·
|
acquisitions or divestitures;
|
|
|
·
|
litigation and government proceedings;
|
|
|
·
|
adverse legislation, including changes in governmental regulation;
|
|
|
·
|
third-party reimbursement policies;
|
|
|
·
|
changes in securities analysts’ recommendations;
|
|
|
·
|
changes in health care policies and practices;
|
|
|
·
|
halting or suspension of trading in our common stock by the NYSE MKT;
|
|
|
·
|
economic and other external factors; and
|
|
|
·
|
general market conditions.
|
In the past, following periods of volatility
in the market price of a company’s securities, securities class action litigation has often been instituted. These lawsuits
often seek unspecified damages, and as with any litigation proceeding, one cannot predict with certainty the eventual outcome of
pending litigation. Furthermore, we may have to incur substantial expenses in connection with any such lawsuits and our management’s
attention and resources could be diverted from operating our business as we respond to any such litigation. We maintain insurance
to cover these risks for us and our directors and officers, but our insurance is subject to high deductibles to reduce premium
expense, and there is no guarantee that the insurance will cover any specific claim that we currently face or may face in the future,
or that it will be adequate to cover all potential liabilities and damages.
Shares of common stock are equity
securities and are subordinate to any indebtedness.
Shares of our common stock are common equity
interests. This means that our common stock will rank junior to any outstanding shares of our preferred stock that we may issue
in the future or to our current credit agreement and any future indebtedness we may incur and to all creditor claims and other
non-equity claims against us and our assets available to satisfy claims on us, including claims in a bankruptcy or similar proceeding.
Additionally, unlike indebtedness, where
principal and interest customarily are payable on specified due dates, in the case of our common stock, (i) dividends are payable
only when and if declared by our board of directors or a duly authorized committee of our board of directors, and (ii) as a corporation,
we are restricted to making dividend payments and redemption payments out of legally available assets. We have never paid a dividend
on our common stock and have no current intention to pay dividends in the future. Furthermore, our common stock places no restrictions
on our business or operations or on our ability to incur indebtedness or engage in any transactions, subject only to the voting
rights available to shareholders generally.
Our stockholders may experience significant
dilution if future equity offerings are used to fund operations or acquire complementary businesses.
If our future operations or acquisitions
are financed through the issuance of equity securities, our stockholders could experience significant dilution. In addition, securities
issued in connection with future financing activities or potential acquisitions may have rights and preferences senior to the rights
and preferences of our common stock. We also have established an equity incentive plan for our management, consultants and employees.
We expect to grant restricted stock and options to purchase shares of our common stock to our directors, employees and consultants
and we will grant additional options in the future. The issuance of shares of our common stock upon the exercise of these options
may result in dilution to our stockholders.
We do not anticipate paying dividends
in the foreseeable future; you should not buy our stock if you expect dividends.
We currently intend to retain our future
earnings to support operations and to finance expansion and, therefore, we do not anticipate paying any cash dividends on our securities
in the foreseeable future.
We could issue “blank check”
preferred stock without stockholder approval with the effect of diluting then current stockholder interests and impairing their
voting rights, and provisions in our charter documents and under Delaware law could discourage a takeover that stockholders may
consider favorable.
Our certificate of incorporation provides
for the authorization to issue up to 5,000,000 shares of “blank check” preferred stock with designations, rights and
preferences as may be determined from time to time by our board of directors. Our board of directors is empowered, without
stockholder approval, to issue one or more series of preferred stock with dividend, liquidation, conversion, voting or other rights
which could dilute the interest of, or impair the voting power of, our common stockholders. The issuance of a series of preferred
stock could be used as a method of discouraging, delaying or preventing a change in control. For example, it would be possible
for our board of directors to issue preferred stock with voting or other rights or preferences that could impede the success of
any attempt to change control of our company. In addition, we have a staggered board of directors and advanced notice is
required prior to stockholder proposals, which might further delay a change of control.
The sale of securities in this offering
may cause dilution and could cause the price of our securities to decline.
Sales of securities in this offering may
result in substantial dilution to the interests of holders of our securities. The sale of securities in this offering, or the anticipation
of such sales, could make it more difficult for us to sell equity or equity-related securities in the future at a time and at prices
that we might otherwise wish to effect sales. Depending on market liquidity at the time, a sale of securities in this offering
at any given time could cause the trading price of our common stock to decline. Should the financing we require to sustain our
working capital needs be unavailable or prohibitively expensive when we require it, the consequences could have a material adverse
effect on our business, operating results, financial condition and prospects.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING
STATEMENTS
The statements contained in this prospectus
that are not purely historical are forward-looking statements within the meaning of applicable securities laws. Our forward-looking
statements include, but are not limited to, statements regarding our “expectations,” “hopes,” “beliefs,”
“intentions,” or “strategies” regarding the future. In addition, any statements that refer to projections,
forecasts, or other characterizations of future events or circumstances, including any underlying assumptions, are forward-looking
statements. The words “anticipate,” “believe,” “continue,” “could,” “estimate,”
“expect,” “intend,” “may,” “might,” “plan,” “possible,”
“potential,” “predict,” “project,” “should” and “would,” as well as
similar expressions, may identify forward-looking statements, but the absence of these words does not mean that a statement is
not forward looking. Forward-looking statements in this prospectus may include, for example, statements about:
|
|
·
|
our ability to obtain financing on reasonable terms;
|
|
|
·
|
our ability to increase revenue;
|
|
|
·
|
our ability to remain listed on the NYSE MKT exchange;
|
|
|
·
|
our ability to comply with the covenants in our credit facility;
|
|
|
·
|
our ability to maintain sufficient liquidity to fund our operations;
|
|
|
·
|
our ability to obtain shareholder approval to increase our authorized
shares of common stock;
|
|
|
·
|
the ability of our sales force to achieve expected results;
|
|
|
·
|
our ability to remain competitive;
|
|
|
·
|
government regulations;
|
|
|
·
|
our ability to expand our production capacity;
|
|
|
·
|
our ability to innovate and develop new products;
|
|
|
·
|
our ability to obtain donor cadavers for our products;
|
|
|
·
|
our ability to engage and retain qualified technical personnel and
members of our management team;
|
|
|
·
|
government and third-party coverage and reimbursement for our products;
|
|
|
·
|
our ability to obtain regulatory approvals;
|
|
|
·
|
our ability to successfully integrate future business combinations
or acquisitions;
|
|
|
·
|
product liability claims and other litigation to which we may be subjected;
|
|
|
·
|
product recalls and defects;
|
|
|
·
|
timing and results of clinical studies;
|
|
|
·
|
our ability to obtain and protect our intellectual property and proprietary
rights;
|
|
|
·
|
infringement and ownership of intellectual property;
|
|
|
·
|
influence by our management; and
|
|
|
·
|
our ability to issue preferred stock.
|
The forward-looking statements contained
in this prospectus are based on our current expectations and beliefs concerning future developments and their potential effects
on us. There can be no assurance that future developments affecting us will be those that we have anticipated. These
forward-looking statements involve a number of risks, uncertainties, or assumptions, many of which are beyond our control, which
may cause actual results or performance to be materially different from those expressed or implied by these forward-looking statements.
These risks and uncertainties include, but are not limited to, those factors described in the “Risk Factors” section
of this prospectus. Should one or more of these risks or uncertainties materialize, or should any of our assumptions prove
incorrect, actual results may vary in material respects from those projected in these forward-looking statements. We undertake
no obligation to update or revise any forward-looking statements, whether as a result of new information, future events, or otherwise,
except as may be required under applicable securities laws.
USE OF PROCEEDS
Except
as described in any applicable prospectus supplement in connection with a specific offering, we intend to use the net proceeds
from the sale of the securities offered under this prospectus for operating costs, working capital, and general corporate purposes.
PLAN OF DISTRIBUTION
We may sell the securities being offered
by this prospectus separately or together:
|
|
·
|
directly to purchasers;
|
|
|
·
|
to or through underwriters;
|
|
|
·
|
through a block trade in which the broker or dealer engaged to handle
the block trade will attempt to sell the securities as agent, but may position and resell a portion of the block as principal to
facilitate the transaction; or
|
|
|
·
|
through a combination of any of these methods of sale.
|
In addition, we may issue the securities
being offered by this prospectus as a dividend or distribution.
We may effect the distribution of the securities
from time to time in one or more transactions:
|
|
·
|
at a fixed price or prices, which may be changed from time to time;
|
|
|
·
|
at market prices prevailing at the times of sale;
|
|
|
·
|
at prices related to prevailing market prices; or
|
We may engage in at-the-market offerings
into an existing trading market in accordance with Rule 415(a)(4) under the Securities Act, and we may also sell securities through
a rights offering, forward contracts or similar arrangements. In any distribution of subscription rights to stockholders, if all
of the underlying securities are not subscribed for, we may then sell the unsubscribed securities directly to third parties or
may engage the services of one or more underwriters, dealers or agents, including standby underwriters, to sell the unsubscribed
securities to third parties.
Other than our common stock, which is listed
on the NYSE MKT, the securities issued and sold under this prospectus will have no established trading market. Any shares of our
common stock sold pursuant to this prospectus will be eligible for listing and trading on the NYSE MKT, subject to additional listing
approval. Any underwriters to whom securities are sold by us for public offering and sale may make a market in the securities,
but the underwriters will not be obligated to do so and may discontinue any market making at any time without notice. The securities,
other than our common stock, may or may not be listed on a national securities exchange or other trading market.
We will describe the method of distribution
of the securities in a prospectus supplement. We may directly solicit offers to purchase the securities offered by this prospectus.
Agents designated by us from time to time may solicit offers to purchase the securities. We will name any agent involved in the
offer of sale of the securities and set forth any commissions payable by us to an agent in a prospectus supplement. Unless otherwise
indicated in a prospectus supplement, any agent will be acting on a best efforts basis for the period of its appointment. Any agent
may be deemed to be an “underwriter” of the securities as that term is defined in the Securities Act of 1933, as amended
(the “Securities Act”).
We may directly solicit offers to purchase
the securities, and we may sell directly to institutional investors or others. These persons may be deemed to be underwriters within
the meaning of the Securities Act with respect to any resale of the securities. A prospectus supplement will describe the terms
of any direct sales, including the terms of any bidding or auction process.
If a dealer is used in the sale of the
securities, we or an underwriter will sell securities to the dealer, as principal. The dealer may resell the securities to the
public at varying prices to be determined by the dealer at the time of resale. A prospectus supplement will set forth the name
of the dealer and the terms of the transactions.
If we use an underwriter or underwriters
in the sale of securities, we will execute an underwriting agreement with the underwriter or underwriters at the time we reach
an agreement for sale. We will set forth in a prospectus supplement the names of the specific managing underwriter or underwriters,
as well as any other underwriters, and the terms of the transactions, including compensation of the underwriters and dealers. This
compensation may be in the form of discounts, concessions or commissions. Underwriters and others participating in any offering
of the securities may engage in transactions that stabilize, maintain or otherwise affect the price of the securities. We will
describe any of these activities in a prospectus supplement.
Agreements we enter into with agents, underwriters
and dealers may entitle them to indemnification by us against specified liabilities, including liabilities under the Securities
Act, or to contribution by us to payments they may be required to make in respect of these liabilities. A prospectus supplement
will describe the terms and conditions of indemnification or contribution.
We may authorize underwriters, dealers
and agents to solicit offers by certain institutional investors to purchase offered securities under contracts providing for payment
and delivery on a future date specified in a prospectus supplement. The prospectus supplement will describe the public offering
price for the securities and the commission payable for solicitation under any delayed delivery contract. Delayed delivery contracts
will contain definite fixed price and quantity terms.
To the extent permitted by and in accordance
with Regulation M under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), in connection with an
offering an underwriter may engage in over-allotments, stabilizing transactions, short covering transactions and penalty bids.
Over-allotments involve sales in excess of the offering size, which creates a short position. Stabilizing transactions permit bids
to purchase the underlying security so long as the stabilizing bids do not exceed a specified maximum. Short covering transactions
involve purchases of the securities in the open market after the distribution is completed to cover short positions. Penalty bids
permit the underwriters to reclaim a selling concession from a dealer when the securities originally sold by the dealer are purchased
in a covering transaction to cover short positions. Those activities may cause the price of the securities to be higher than it
would be otherwise. If commenced, the underwriters may discontinue any of the activities at any time.
The specific terms of any lock-up provisions
in respect of any given offering will be described in the applicable prospectus supplement.
In compliance with the guidelines of the
Financial Industry Regulatory Authority, Inc., or FINRA, the maximum consideration or discount to be received by any FINRA member
or independent broker-dealer may not exceed 8% of the aggregate proceeds of the offering.
The underwriters, dealers and agents may
engage in transactions with us, or perform services for us, in the ordinary course of business for which they receive compensation.
No securities may be sold under this prospectus
without delivery, in paper format, in electronic format on the internet, or both, of the applicable prospectus supplement describing
the method and terms of the offering.
DESCRIPTION OF COMMON STOCK
The following description of our common
stock, together with the additional information we include in any applicable prospectus supplements, summarizes the material terms
and provisions of the common stock that we may offer under this prospectus. For the complete terms of our common stock, please
refer to our Amended and Restated Certificate of Incorporation and Amended and Restated Bylaws that are filed as exhibits to our
reports incorporated by reference into the registration statement that includes this prospectus. The General Corporation Law of
Delaware may also affect the terms of our common stock.
Authorized and Outstanding
Common Stock
Our Amended and Restated Certificate of
Incorporation provides that we have authority to issue (i) 95,000,000 shares of common stock, par value $0.000001 per share, 54,858,458
of which are issued and outstanding as of March 28, 2014, and (ii) 5,000,000 shares of preferred stock, par value $0.000001 per
share, none of which are issued and outstanding as of the date of this prospectus. We also have outstanding warrants to purchase
approximately 11,660,603 shares of our common stock and there are 9,000,000 shares authorized for issuance under our Amended and
Restated Bacterin International Equity Incentive Plan.
Listing
Our common stock is listed on the NYSE
MKT under the symbol “BONE”.
Dividends
Our Board of Directors may authorize, and
we may make, distributions to our common stockholders, subject to any restriction in our Amended and Restated Certificate of Incorporation
and to those limitations prescribed by law. However, we have never paid cash dividends on our common stock or any other securities.
We anticipate that we will retain all of our future earnings, if any, for use in the expansion and operation of our business and
do not anticipate paying cash dividends in the foreseeable future.
Fully Paid and Non-Assessable
All shares of our outstanding common stock
are fully paid and non-assessable.
Voting Rights
Each share of our common stock is entitled
to one vote in each matter submitted to a vote at a meeting of stockholders including in all elections for directors; stockholders
are not entitled to cumulative voting in the election for directors. Our stockholders may vote either in person or by proxy.
Preemptive and Other Rights
Holders of our common stock have no preemptive
rights and have no other rights to subscribe for additional securities of the Company under Delaware law; nor does our common stock
have any conversion rights or rights of redemption. Upon liquidation, all holders of our common stock are entitled to participate
pro rata in our assets available for distribution, subject to the rights of any class of preferred stock then outstanding.
Staggered Board of Directors
Our Board of Directors is divided into
three classes, the members of each of which serve for staggered three-year terms. Our stockholders may elect only one-third of
the directors each year; therefore, it is more difficult for a third party to gain control of our Board of Directors than if our
Board was not staggered.
Transfer Agent
The transfer agent for our common stock
is Corporate Stock Transfer.
Limitations of Director Liability
Delaware law authorizes corporations to
limit or eliminate the personal liability of directors to corporations and their stockholders for monetary damages for breach of
directors’ fiduciary duty of care. Our Amended and Restated Certificate of Incorporation limits the liability of directors
to the fullest extent permitted by Delaware law.
Indemnification
Our Amended and Restated Bylaws provide
for mandatory indemnification of directors and officers to the maximum extent allowed by applicable law. In addition, we have also
entered into indemnification agreements with our directors and officers, and we must advance or reimburse directors and officers
for expenses they incur in connection with indemnifiable claims. We also maintain directors’ and officers’ liability
insurance.
DESCRIPTION OF PREFERRED STOCK
The following description of our preferred
stock, together with the additional information we include in any prospectus supplements, summarizes the material terms and provisions
of the preferred stock that we may offer under this prospectus. For the complete terms of our preferred stock, please refer to
our Amended and Restated Certificate of Incorporation and Amended and Restated Bylaws that are filed as exhibits to our reports
incorporated by reference into the registration statement that includes this prospectus. The General Corporation Law of Delaware,
as amended, may also affect the terms of our common stock.
Preferred Stock That We May
Offer and Sell
Our Amended and Restated Certificate of
Incorporation authorizes our Board of Directors, without further stockholder action, to provide for the issuance of up to 5,000,000
shares of preferred stock, in one or more classes or series and to fix the rights, preferences, privileges, and restrictions thereof,
including dividend rights, dividend rates, conversion rights, voting rights, terms of redemption, redemption prices, liquidation
preferences and the number of shares constituting any series of the designation of such series, without further vote or action
by the stockholders. As of the date of this prospectus, no shares of preferred stock are outstanding.
The particular terms of any series of preferred
stock being offered by us under this shelf registration statement will be described in the prospectus supplement and certificate
of designations relating to the applicable series of preferred stock. Those terms may include:
|
|
·
|
the title and liquidation preference per
share of the preferred stock and the number of shares offered;
|
|
|
·
|
the purchase price of the preferred stock;
|
|
|
·
|
the dividend rate (or method of calculation),
the dates on which dividends will be paid and the date from which dividends will begin to accumulate;
|
|
|
·
|
any redemption or sinking fund provisions
of the preferred stock;
|
|
|
·
|
any conversion provisions of the preferred
stock;
|
|
|
·
|
the voting rights, if any, of the preferred
stock; and
|
|
|
·
|
any additional dividend, liquidation,
redemption, sinking fund and other rights, preferences, privileges, limitations and restrictions of the preferred stock.
|
The preferred stock will be, when issued,
fully paid and non-assessable.
Voting Rights
The General Corporation Law of Delaware
provides that the holders of preferred stock will have the right to vote separately as a class on any proposal involving fundamental
changes in the rights of holders of that preferred stock. This right is in addition to any voting rights that may be provided for
in the applicable certificate of designations.
Other
Our issuance of preferred stock may have
the effect of delaying or preventing a change in control. Our issuance of preferred stock could decrease the amount of earnings
and assets available for distribution to the holders of common stock or other preferred stock or could adversely affect the rights
and powers, including voting rights, of the holders of common stock or other preferred stock. The issuance of preferred stock could
have the effect of decreasing the market price of our common stock.
Transfer Agent and Registrar
The transfer agent and registrar for the
preferred stock will be set forth in the applicable prospectus supplement.
DESCRIPTION OF WARRANTS
The following description, together with
the additional information we may include in any applicable prospectus supplements, summarizes the material terms and provisions
of the warrants that we may offer under this prospectus and the related warrant agreements and warrant certificates. While the
terms summarized below will apply generally to any warrants we may offer, we will describe the particular terms of any series of
warrants in more detail in the applicable prospectus supplement.
We may issue warrants for the purchase
of shares of our common stock or preferred stock. Warrants may be issued independently or together with the shares of common stock
or preferred stock offered by any prospectus supplement to this prospectus and may be attached to or separate from such shares.
Further terms of the warrants will be set forth in the applicable prospectus supplement.
The applicable prospectus supplement will
describe the terms of the warrants in respect of which this prospectus is being delivered, including, where applicable, the following:
|
|
·
|
the title of such warrants;
|
|
|
·
|
the aggregate number of such warrants;
|
|
|
·
|
the price or prices at which such warrants
will be issued;
|
|
|
·
|
the designation, terms and number of shares
of common stock or preferred stock purchasable upon exercise of such warrants;
|
|
|
·
|
the designation and terms of the shares
of common stock or preferred stock with which such warrants are issued and the number of such warrants issued with such shares;
|
|
|
·
|
the date on and after which such warrants
and the related common stock or preferred stock will be separately transferable, including any limitations on ownership and transfer
of such warrants;
|
|
|
·
|
the price at which each share of common
stock or preferred stock purchasable upon exercise of such warrants may be purchased;
|
|
|
·
|
the date on which the right to exercise
such warrants shall commence and the date on which such right shall expire;
|
|
|
·
|
the minimum or maximum amount of such
warrants that may be exercised at any one time;
|
|
|
·
|
information with respect to book-entry
procedures, if any;
|
|
|
·
|
a discussion of certain federal income
tax consequences; and
|
|
|
·
|
any other terms of such warrants, including
terms, procedures and limitations relating to the exchange and exercise of such warrants.
|
This summary of the warrants is not complete.
We urge you to read the warrants filed as exhibits to the registration statement that includes this prospectus and the description
of the additional terms of the warrants included in the prospectus supplement. The terms of the warrants we issue may differ materially
from warrants we have issued in the past.
Before exercising their warrants, holders
of warrants will not have any of the rights of holders of the securities purchasable upon such exercise, including the right to
receive dividends, if any, or payments upon our liquidation, dissolution or winding up or to exercise voting rights, if any.
Exercise of Warrants
Each warrant will entitle the holder thereof
to purchase the number of shares of preferred stock and the number of shares of common stock at the exercise price as shall in
each case be set forth in, or be determinable as set forth in, the applicable prospectus supplement. Warrants may be exercised
at any time up to the close of business on the expiration date set forth in the applicable prospectus supplement. After the close
of business on the expiration date, unexercised warrants will become void.
Warrants may be exercised as set forth
in the applicable prospectus supplement relating to the warrants offered thereby. Upon receipt of payment of the exercise price,
surrender of the original warrant, and submission of a properly completed and duly executed notice of exercise, we will, as soon
as practicable, forward the purchased securities. If less than all of the warrants represented by the warrant certificate are exercised,
a new warrant certificate will be issued for the remaining warrants. Holders of warrants will be required to pay any tax or governmental
charge that may be imposed in connection with transferring the underlying securities in connection with the exercise of the warrants.
INCORPORATION OF CERTAIN INFORMATION
BY REFERENCE
The SEC allows us to incorporate by reference
the information that we file with the SEC, which means that we can disclose important information to you by referring you to those
documents. The information incorporated by reference is considered to be part of the prospectus. These documents may include periodic
reports, such as Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K, as well as Proxy
Statements. Any documents that we subsequently file with the SEC will automatically update and replace the information previously
filed with the SEC. Thus, for example, in the case of a conflict or inconsistency between information set forth in this prospectus
and information incorporated by reference into this prospectus, you should rely on the information contained in the document filed
later.
This prospectus incorporates by reference
the documents listed below that we previously have filed with the SEC and any additional documents that we may file with the SEC
under Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act between the date of this prospectus and the termination of the offering
of the securities. These documents contain important information about us.
|
|
·
|
Our Annual Report on Form 10-K for the
year ended December 31, 2013;
|
|
|
·
|
Our Current Report on Form 8-K (other
than portions thereof furnished under Item 2.02 or Item 7.01 of Form 8-K and exhibits accompanying such reports that are related
to such items) filed with the SEC on March 10, 2014;
|
|
|
·
|
The description of our common stock contained
in our registration statement on Form 8-A, filed on November 5, 2010, as amended March 4, 2011, including any amendment or reports
filed for the purpose of updating such description; and
|
|
|
·
|
All documents filed by us pursuant to
Section 13(a), 13(c), 14 or 15(d) of the Exchange Act after the date of this prospectus and before the termination of the offering.
|
We are not, however, incorporating by reference
any documents, or portions of documents, whether specifically listed above or arising in the future, which are not deemed “filed”
with the SEC.
You can obtain a copy of any or all of
the documents incorporated by reference in this prospectus (other than an exhibit to a document unless that exhibit is specifically
incorporated by reference into that document) from the SEC on its website at
http://www.sec.gov.
You also can obtain these
documents from us, free of charge, by visiting our internet website
http://www.bacterin.com
or by writing to us or calling
us at the following address and phone number:
Bacterin International Holdings, Inc.
664 Cruiser Lane
Belgrade, MT 59714
Attn: Corporate Secretary
(406) 388-0480
WHERE YOU CAN FIND MORE INFORMATION
We have filed with the SEC a registration
statement under the Securities Act that registers the distribution of the securities offered under this prospectus. The registration
statement, including the attached exhibits and schedules and the information incorporated by reference, contains additional relevant
information about our company and the securities. The rules and regulations of the SEC allow us to omit from this prospectus certain
information included in the registration statement. In addition, we file annual, quarterly and special reports, proxy statements
and other information with the SEC. You may read and copy this information and the registration statement at the SEC public reference
room located at 100 F Street, N.E., Washington D.C. 20549. Please call the SEC at 1-800-SEC-0330 for more information about the
operation of the public reference room.
You may also obtain the documents that
we file electronically on the SEC’s website at
http://www.sec.gov
or on our website at
http://www.bacterin.com
.
Information contained on our website is not incorporated by reference herein and does not constitute part of this prospectus.
LEGAL MATTERS
Our General Counsel, Jill Gilpin, has passed upon certain legal
matters in connection with the securities offered hereby.
EXPERTS
The financial statements incorporated by
reference into this prospectus and registration statement on Form S−3 have been audited by EKS&H LLLP, independent certified
public accountants, as set forth in their report thereon appearing in our Annual Report on Form 10-K and incorporated by reference
into this prospectus and registration statement on Form S−3, and such report is included in reliance upon the authority of
such firm as experts in accounting and auditing.
Xtant Medical (AMEX:XTNT)
Historical Stock Chart
From Apr 2024 to May 2024
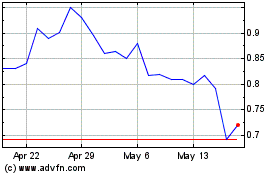
Xtant Medical (AMEX:XTNT)
Historical Stock Chart
From May 2023 to May 2024