Filed Pursuant to Rule 424(b)(5)
File No. 333-218158
PROSPECTUS SUPPLEMENT
(to Prospectus dated May 22, 2017)

2,735,000
SHARES OF COMMON STOCK
AND
WARRANTS
TO PURCHASE 2,735,000 SHARES OF COMMON STOCK
We are offering 5,470,000 shares of our
common stock (including 2,735,000 shares of common stock underlying the Purchase Warrants, as defined herein) and warrants to
purchase one share of our common stock for each share purchased for cash in this offering (the “Purchase Warrants”)
directly to the institutional investors in this offering. The shares of common stock and the Purchase Warrants are being offered
pursuant to this prospectus supplement and accompanying prospectus. The Purchase Warrants will be issued separately but must be
purchased together with the common stock. The combined purchase price for each share of common stock and accompanying Purchase
Warrant is $1.75. The Purchase Warrants will be exercisable beginning on the date of issuance (the “Initial Exercise Date”),
at an exercise price of $2.00 per share and will expire on the five-year anniversary of the Initial Exercise Date. The Purchase
Warrants include an adjustment provision that, subject to certain exceptions, reduces their exercise price if the Company issues
Common Stock or Common Stock equivalents at a price lower than the then-current exercise price of the Purchase Warrants, subject
to a minimum exercise price of $0.44 per share.
The aggregate market value of our outstanding
shares of common stock held by non-affiliates was $64,295,293 based on 27,377,057 shares of common stock outstanding
as of December 14, 2017, of which 23,990,781 shares are held by non-affiliates, and a per share price of $ 2.68
based on the closing sale price of our common stock on December 18, 2017. Pursuant to General Instruction I.B.6 of Form S-3, in
no event will we sell securities in a public primary offering with a value exceeding more than one-third of our public float in
any 12-month period so long as our public float remains below $75.0 million. During the 12-month period prior to and including
the date of this prospectus supplement, we did not offer any securities pursuant to General Instruction I.B.6 of Form S-3.
We have engaged Maxim Group LLC as our sole
placement agent for this offering, or the placement agent. The placement agent is not purchasing or selling any shares offered
by this prospectus supplement and the accompanying base prospectus but will use its reasonable best efforts to arrange for the
sale of the securities offered. See “Plan of Distribution.” This offering is expected to close on or about December
22, 2017, subject to customary closing conditions, without further notice to you. We have not arranged to place the funds from
the investors in an escrow, trust or similar account.
Our common stock is listed on The NASDAQ
Capital Market under the symbol “APDN.” The Purchase Warrants being issued are not listed on any securities exchange,
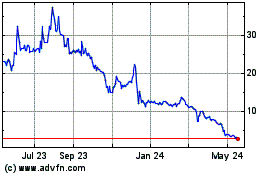
and we do not expect to list the Purchase Warrants. On December 19, 2017, the last reported sales price of our common stock
on The NASDAQ Capital Market was $2.22 per share.
The purchase of the securities offered
through this prospectus involves a high degree of risk. You should consider carefully the risk factors beginning on page S-14 of
this prospectus supplement, on page 6 of the accompanying base prospectus, and in the documents incorporated by reference into
this prospectus supplement before purchasing any of the securities offered by this prospectus.
Neither the Securities and Exchange Commission
nor any state securities commission has approved or disapproved of these securities or passed upon the adequacy or accuracy of
this prospectus. Any representation to the contrary is a criminal offense.
|
|
|
Per Share of Common
Stock and Purchase
Warrant
|
|
|
Total
|
|
|
Public offering price
|
|
$
|
1.75
|
|
|
$
|
4,786,250
|
|
|
Placement agent fee
(1)
|
|
$
|
0.11
|
|
|
$
|
311,106
|
|
|
Proceeds to us, before expenses
(2)
|
|
$
|
1.64
|
|
|
$
|
4,475,144
|
|
|
|
(1)
|
We have agreed to pay the placement agent an aggregate cash placement fee equal to 6.5% of the gross proceeds in this offering
from sales arranged by the placement agent. We also have agreed to pay certain expenses and advances of the placement agent, as
discussed under “Plan of Distribution” in this prospectus supplement.
|
|
|
(2)
|
Does not include additional compensation received by the placement agent, including reimbursement of legal fees and out of
pocket expenses, as further discussed under the heading “Plan of Distribution” herein. The amount of the offering proceeds
to us presented in this table does not give effect to any exercise of the Purchase Warrants.
|
Sole Placement Agent
Maxim Group LLC
Prospectus Supplement dated December 21 ,
2017
Table of
Contents
ABOUT
THIS PROSPECTUS SUPPLEMENT
This prospectus supplement is part of a
registration statement that we filed with the Securities and Exchange Commission (SEC) utilizing a “shelf” registration
process. Under this shelf registration statement process, we may from time to time offer to sell up to $25,000,000 of our common
stock, preferred stock, debt securities, warrants to purchase common stock, preferred stock and/or debt securities, rights to purchase
common stock, preferred stock or warrants and units consisting of shares of common stock, preferred stock, warrants, rights or
debt securities or any combination of these securities in one or more transactions.
We provide information to you about this
offering of our common stock and Purchase Warrants in two separate documents that are bound together: (1) this prospectus supplement,
which describes the specific details regarding this offering of shares of common stock and Purchase Warrants; and (2) the accompanying
base prospectus dated May 22, 2017, and that was declared effective on May 26, 2017 and is included in our registration statement
on Form S-3 (SEC File No.
333-218158
) (the “Registration Statement”), which provides
general information regarding our shares of common stock, shares of preferred stock, debt securities, warrants to purchase common
stock, preferred stock and/or debt securities, rights to purchase common stock, preferred stock or warrants and units consisting
of shares of common stock, shares of preferred stock, warrants, rights or debt securities, or any combination of these securities
and other information some of which may not apply to this offering. If information in this prospectus supplement is inconsistent
with the accompanying base prospectus, you should rely on this prospectus supplement. If any statement in one of these documents
is inconsistent with a statement in another document having a later date, for example, a document incorporated by reference in
this prospectus supplement, the statement in the document having the later date modifies or supersedes the earlier statement as
our business, financial condition, results of operations and prospects may have changed since the earlier dates.
You should read this prospectus supplement,
together with the accompanying base prospectus, the documents incorporated by reference in this prospectus supplement and the base
prospectus and any free writing prospectus that we have authorized for use in connection with this offering before making an investment
decision. You should also read and consider the information in the documents referred to in the sections of this prospectus supplement
and the accompanying base prospectus entitled “Where You Can Find More Information” and “Information Incorporated
by Reference.” When we refer to this “prospectus”, we are referring to both this prospectus supplement and the
base prospectus combined.
You should rely only on the information
contained or incorporated by reference in this prospectus supplement, the base prospectus, or in any free writing prospectus that
we have authorized for use in connection with this offering. We and the placement agent have not authorized anyone to provide you
with different information. If anyone provides you with different or inconsistent information, you should not rely on it.
This prospectus supplement and the accompanying
prospectus contain summaries of certain provisions contained in some of the documents described herein, but reference is made to
the actual documents for complete information. All of the summaries are qualified in their entirety by the actual documents. Copies
of some of the documents referred to herein have been filed, will be filed or will be incorporated herein by reference as exhibits
to the registration statement, and you may obtain copies of those documents as described below under the section entitled “
Where
You Can Find More Information
.”
We further note that the representations,
warranties and covenants made by us in any agreement that is filed as an exhibit to any document that is incorporated by reference
herein were made solely for the benefit of the parties to such agreement, including, in some cases, for the purpose of allocating
risk among the parties to such agreements, and should not be deemed to be a representation, warranty or covenant to you. Moreover,
such representations, warranties or covenants were accurate only as of the date when made. Accordingly, such representations, warranties
and covenants should not be relied on as accurately representing the current state of our affairs.
We are not making an offer to sell the securities
covered by this prospectus supplement in any jurisdiction in which an offer or solicitation is not permitted or in which the person
making the offer or solicitation is not qualified to do so or to anyone to whom it is unlawful to make an offer or solicitation.
The information appearing in this prospectus
supplement, the documents incorporated by reference in this prospectus supplement, the base prospectus, and in any free writing
prospectus that we have authorized for use in connection with this offering is accurate only as of its respective date, regardless
of the time of delivery of the respective document or of any sale of securities covered by this prospectus. You should not assume
that the information contained in or incorporated by reference in this prospectus supplement, the base prospectus, or in any free
writing prospectus that we have authorized for use in connection with this offering, is accurate as of any date other than the
respective dates thereof.
Our trademarks in the United States include
Applied DNA Sciences
®
, SigNature
®
molecular tags, SigNature
®
T molecular tags, fiberTyping
®
,
DNAnet
®
, digitalDNA
®
, SigNify
®
, BackTrac
®
, Beacon
®
and
CertainT
®
All trademarks, service marks and trade names included or incorporated by reference in this prospectus
supplement are the property of their respective owners, including, without limitation, the PimaCott
®
, HomeGrown
®
LoneStar™ and HomeGrown Acala™ marks owned by Himatsingka America, Inc. and/or its affiliates. All other trademarks,
service marks and trade names included or incorporated by reference in this prospectus, if any, are the property of their respective
owners.
In this prospectus supplement “Applied
DNA,” “we,” “us,” the “Company,” and “our” refer to Applied DNA Sciences,
Inc. and its subsidiaries.
FORWARD-LOOKING
STATEMENTS
This prospectus supplement and the documents
incorporated by reference herein contain “forward-looking statements” within the meaning of Section 27A of the Securities
Act of 1933, as amended (the “Securities Act”) and Section 21E of the Securities Exchange Act of 1934, as amended (the
“Exchange Act”), that are intended to qualify for the “safe harbor” created by those sections. In addition,
we may make forward-looking statements in other documents filed with or furnished to the SEC, and our management and other representatives
may make forward-looking statements orally or in writing to analysts, investors, representatives of the media and others.
Forward-looking statements can generally
be identified by the fact that they do not relate strictly to historical or current facts and include, but are not limited to,
statements using terminology such as “can”, “may”, “could”, “should”, “assume”,
“forecasts”, “believe”, “designated to”, “will”, “expect”, “plan”,
“anticipate”, “estimate”, “potential”, “position”, “predicts”, “strategy”,
“guidance”, “intend”, “seek”, “budget”, “project” or “continue”,
or the negative thereof or other comparable terminology regarding beliefs, plans, expectations or intentions regarding the future.
You should read statements that contain these words carefully because they:
|
|
·
|
discuss our future expectations;
|
|
|
·
|
contain projections of our future results of operations or of our financial condition; and
|
|
|
·
|
state other “forward-looking” information.
|
We believe it is important to communicate
our expectations. However, forward-looking statements involve risks and uncertainties and our actual results and the timing of
certain events could differ materially from those discussed in forward-looking statements as a result of certain factors, including
those set forth under “Risk Factors” and “Prospectus Supplement Summary – Our Company” set forth
in this prospectus supplement and the documents incorporated herein by reference.
Accordingly, our actual results and the
timing of certain events may differ materially from those expressed or implied in such forward-looking statements due to a variety
of factors and risks, including, but not limited to, those set forth in this prospectus supplement under “Risk Factors”
and those set forth from time to time in our other filings with the SEC.
All forward-looking statements and risk
factors included in this prospectus supplement and the documents incorporated herein by reference are made as of the date hereof,
based on information available to us as of such date, and we assume no obligations to update any forward-looking statement or risk
factor, unless we are required to do so by law. If we do update one or more forward-looking statements, no inference should be
drawn that we will make updates with respect to other forward-looking statements or that we will make any further updates to those
forward-looking statements at any future time.
Forward-looking statements may include our
plans and objectives for future operations, including plans and objectives relating to our products and our future economic performance,
projections, business strategy and timing and likelihood of success. Assumptions relating to the foregoing involve judgments with
respect to, among other things, future economic, competitive and market conditions, future business decisions, and the time and
money required to successfully complete development and commercialization of our technologies, all of which are difficult or impossible
to predict accurately and many of which are beyond our control.
Any of the assumptions underlying the forward-looking
statements contained in this prospectus supplement could prove inaccurate and, therefore, we cannot assure you that the results
contemplated in any of such forward-looking statements will be realized. Based on the significant uncertainties inherent in these
forward-looking statements, the inclusion of any such statement should not be regarded as a representation or as a guarantee by
us that our objectives or plans will be achieved, and we caution you against relying on any of the forward-looking statements contained
herein.
SUMMARY
OF RISKS
Before you invest in our common stock and
Purchase Warrants, you should carefully consider all the information in this prospectus supplement, including matters set forth
in the “Risk Factors” section beginning on page S-14 of this prospectus supplement. We believe that the following are
some of the major risks and uncertainties that may affect us:
|
|
·
|
the inclusion in this prospectus summary of preliminary, unaudited fiscal year end and fourth quarter information still subject
to completion of our financial closing procedures;
|
|
|
·
|
our lack of significant revenues;
|
|
|
·
|
our history of net losses, which may continue, and our potential inability to achieve profitability;
|
|
|
·
|
the possibility that we may require additional financing, which may involve the issuance of additional shares of common stock
or securities exercisable for common stock and dilute the percentage of ownership held by our current stockholders;
|
|
|
·
|
difficulty in
obtaining or inability to obtain additional financing if such financing becomes necessary;
|
|
|
·
|
volatility in the price and/or trading volume of our common stock;
|
|
|
·
|
future short selling and/or manipulation of the price of our common stock;
|
|
|
·
|
our inability to implement our short and long-term strategies;
|
|
|
·
|
competition from products and services provided by other companies;
|
|
|
·
|
potential difficulties and failures in manufacturing our products;
|
|
|
·
|
loss of strategic relationships;
|
|
|
·
|
dependence on a limited number of key customers;
|
|
|
·
|
lack of acceptance of our products and services by potential customers;
|
|
|
·
|
potential failure to introduce new products and services;
|
|
|
·
|
difficulty or failure in expanding/and or maintaining our sales, marketing and support organizations and our distribution arrangements
necessary to enable us to reach our goals with respect to increasing market acceptance of our products and services;
|
|
|
·
|
seasonality in revenues related to our cotton customer contracts;
|
|
|
·
|
inability to continue to retain the services of Dr. Hayward, our Chief Executive Officer;
|
|
|
·
|
inability to compete effectively in the industries in which we operate;
|
|
|
·
|
lack of success in our research and development efforts for new products;
|
|
|
·
|
failure to manage our growth in operations and acquisitions of new technologies and businesses;
|
|
|
·
|
inability to protect our intellectual property rights;
|
|
|
·
|
intellectual property litigation against us or other legal actions or proceedings in which we may become involved;
|
|
|
·
|
unauthorized disclosure of sensitive or confidential data (including customer data) and cybersecurity breaches; and
|
|
|
·
|
adverse changes in worldwide or domestic economic, political or business conditions.
|
WHERE
YOU CAN FIND MORE INFORMATION
We have filed with the SEC a registration
statement on Form S-3 (File No. 333-218158), of which this prospectus supplement and the accompanying base prospectus are a part,
under the Securities Act, to register the shares of common stock and Purchase Warrants offered by this prospectus supplement. However,
this prospectus supplement and the accompanying base prospectus do not contain all of the information contained in the Registration
Statement. We have omitted from this prospectus supplement some parts of the Registration Statement as permitted by the rules and
regulations of the SEC. Statements in this prospectus supplement concerning any document we have filed as an exhibit to the Registration
Statement or that we otherwise filed with the SEC are not intended to be comprehensive and are qualified in their entirety by reference
to these filings. In addition, we file annual, quarterly and current reports, proxy statements and other information with the SEC.
You may read and copy any documents that we have filed with the SEC at the SEC’s Public Reference Room at 100 F Street, N.E.,
Room 1580, Washington, D.C. 20549. Please call the SEC at 1-800-SEC-0330 for further information about the operation of the Public
Reference Room. The SEC also maintains an Internet site that contains reports, proxy and information statements and other information
that registrants file electronically with the SEC, including us. The SEC’s Internet site can be found at http://www.sec.gov.
In addition, we make available on or through our Internet site copies of these reports as soon as reasonably practicable after
we electronically file or furnished them to the SEC. Our Internet site can be found at http:www.adnas.com. Our website is not a
part of this prospectus supplement.
INFORMATION
INCORPORATED BY REFERENCE
We have elected to incorporate certain information
by reference into this prospectus supplement. By incorporating by reference, we can disclose important information to you by referring
you to other documents we have filed or will file with the SEC. The information incorporated by reference is deemed to be part
of this prospectus supplement, except for information incorporated by reference that is superseded by information contained in
this prospectus supplement. This means that you must look at all of the SEC filings that we incorporate by reference to determine
if any statements in the prospectus supplement or any document previously incorporated by reference have been modified or superseded.
This prospectus supplement incorporates by reference the documents set forth below that we have previously filed with the SEC,
except in each case the information contained in such document to the extent “furnished” and not “filed”:
|
|
·
|
Our Annual Report on Form 10-K for the year ended September 30, 2016, as filed with the SEC on December 6, 2016;
|
|
|
·
|
Our Quarterly Reports on Form 10-Q for the period ended December 31, 2016, as filed with the SEC on February 9, 2017; for the
period ended March 31, 2017, as filed with the SEC on May 11, 2017; and for the period ended June 30, 2017, as filed with the SEC
on August 10, 2017;
|
|
|
·
|
Our Current Reports on Form 8-K filed with the SEC on October 14, 2016, November 3, 2016, December 6, 2016, February 9, 2017;
March 10, 2017, May 11, 2017; June 27, 2017; June 28, 2017; August 10, 2017, August 29, 2017, December 15, 2017 and December 20,
2017; and
|
|
|
·
|
The description
of our Common Stock and listed warrants contained in our Registration Statement
on Form 8-A filed on November 13, 2014, including any amendment or report filed for the
purpose of updating such description.
|
All reports and other documents subsequently
filed by us pursuant to Sections 13(a), 13(c), 14 and 15(d) of the Exchange Act after the date of the registration statement to
which this prospectus forms a part of or after the date of this prospectus supplement and prior to the termination or completion
of the offering of Securities under this prospectus supplement shall be deemed to be incorporated by reference in this prospectus
supplement and to be a part hereof from the date of filing such reports and other documents.
You may obtain copies of these documents
on the website maintained by the SEC at
http://www.sec.gov
. We will furnish to you, upon written or oral request, a copy
of any or all of the documents that have been incorporated by reference, including exhibits to these documents. You may request
a copy of those filings at no cost by writing or telephoning us at Corporate Secretary, Applied DNA Sciences, Inc., 50 Health Sciences
Drive, Stony Brook, New York 11790, (631) 240-8800 or visiting our website at http://www.adnas.com. No information contained on
our website is intended to be included as part of, or incorporated by reference into, this prospectus supplement.
PROSPECTUS
SUPPLEMENT SUMMARY
This summary highlights certain information
about us, this offering and information appearing elsewhere in this prospectus supplement, the accompanying base prospectus and
in the documents we incorporate by reference in this prospectus supplement. This summary is not complete and does not contain all
of the information that you should consider before investing in our securities. After you carefully read this summary, to fully
understand our company and this offering and its consequences to you, you should read this entire prospectus supplement, the accompanying
base prospectus, and any related free writing prospectus authorized by us, including the information referred to under the heading
“Risk Factors” in this prospectus supplement beginning on page S-14, and any related free writing prospectus, as well
as the other documents that we incorporate by reference into this prospectus supplement, including our financial statements and
the notes to those financial statements, which are incorporated herein by reference from our Annual Report on Form 10-K for the
year ended September 30, 2016, filed on December 6, 2016 and our Quarterly Reports on Form 10-Q for the quarter ended December
31, 2016, filed on February 9, 2017, the quarter ended March 31, 2017, filed on May 11, 2017 and the quarter ended June 30, 2017,
filed on August 10, 2017. Please read “Where You Can Find More Information” on page S-4 of this prospectus supplement.
Our Company
Overview
Using biotechnology as a forensic foundation,
we create unique security solutions addressing the challenges of modern commerce. Whether for supply chain security, brand protection
or law enforcement applications, it is our goal to help establish secure flourishing environments that foster quality, integrity
and success. With secure taggants, high-resolution DNA authentication, and comprehensive reporting, our SigNature molecular tag
technologies are designed to deliver what we believe to be the greatest levels of security, deterrence and legal recourse strength.
We are also engaged in the large-scale production of specific DNA sequences using the polymerase chain reaction (“
PCR
”)
method.
SigNature
®
molecular tags, SigNature
®
T molecular tags, fiberTyping
®
, DNAnet
®
, digitalDNA
®
, SigNify
®
, BackTrac
®
, Beacon
®
and CertainT
®
comprise our principal technology platform. The large-scale production of specific DNA sequences is used in the diagnostics and
reagent industries.
SigNature molecular tags, the core of our
technology platform, are what we believe to be nature’s ultimate means of authentication and supply chain security. Our precision-engineered
molecular tags have not and, we believe, cannot be broken. Additional layers of protection and complexity are added to the mark
in a proprietary manner. SigNature molecular tags in various carriers have proven highly resistant to UV radiation, heat, cold,
vibration, abrasion and other extreme environments and conditions. We work closely with our customers to develop solutions that
will be optimized to their specifications to deliver maximum impact. Our products and technology are protected by what we believe
to be a robust portfolio of patents and trademarks.
Using our products and technology, manufacturers,
brands, and other stakeholders can ensure authenticity and protect against diversion throughout a product’s journey from
manufacturer to use.
The core technologies of our business allow
us to use molecular tags to mark objects in a unique manner that we believe cannot be replicated, and then identify these objects
by detecting the absence or presence of the molecular tag. We believe that our disruptive platform offers broad commercial relevance
across many industry verticals. Our underlying strategy is to become a solutions provider for supply chains of process industries
in which contracts for our products and services are larger and of longer duration as compared to our historic norms, where the
benefits to customers and consumers are more significant, and where our forensic security and traceability offer a unique and protected
value. Consumers, governments and companies are demanding details about the systems and sources that deliver their goods. They
worry about quality, safety, ethics, and the environmental impact. Farsighted organizations are directly addressing new threats
and opportunities presented by this question: Where do these goods come from? These are the questions and concerns we are beginning
to address for a growing number of companies. We supply key building blocks for creating secure supply chains with traceability
of goods, which in turn can help ensure integrity in supply, honest sales and marketing claims, and ethical and sustainable sourcing.
Our products and services are offered in
the United States, Europe and Asia. At the present time, we are focusing our efforts on textile and apparel, microcircuits and
other electronics, pharmaceuticals, bulk DNA production (for therapeutics, diagnostics and vaccines), cash-in-transit, consumer
asset marking, printing and packaging businesses and agrochemicals. In the future, we plan to expand our focus to include additional
consumer products, food and beverage and industrial materials. To date, the substantial portion of our revenues has been generated
from sales of our SigNature and SigNature T molecular tags, our principal supply chain security and product authentication solutions.
We expect to continue to grow revenues from sales of our SigNature molecular tags, SigNature T molecular tags, DNAnet, BackTrac,
digitalDNA, Beacon, SigNify and CertainT offerings as we work with companies and governments to secure supply chains and restore
confidence to products and product labeling throughout the world. In addition, we expect to continue to grow revenues from the
large-scale production of specific DNA sequences using our Triathlon™ PCR systems which have multiple applications including
as a diagnostic and reagent and for gene therapy, DNA vaccines and diagnostics. We also expect to see new revenue in the pharmaceutical
market.
SigNature
®
molecular tags,
SigNature
®
T molecular tags, fiberTyping
®
, DNAnet
®
, digitalDNA
®
, SigNify
®
BackTrac
®
, Beacon
®
and CertainT
®
comprise our
principal technology platform. The large-scale production of specific DNA sequences is used in the diagnostics and reagent industries.
Signature Molecular Tags
SigNature Molecular Tag.
The
SigNature molecular tag is our patented molecular taggant technology, at the core of our platform. It provides forensic power and
protection for a wide array of applications. Highly secure, robust and durable, SigNature molecular tags are an ingredient that
can be used to fortify brand protection efforts; strengthen supply chain security; and mark, track and convict criminals. Through
our SigNature molecular tags, custom DNA sequences can be embedded into a wide range of host carriers including ink, varnish, thread
and metal coatings. SigNature molecular tags can be made resistant to challenging environments such as heat, cold, vibration, abrasion,
organic solvents, chemicals, UV radiation and other extreme environmental conditions, and so can be identified for numerous years
after being embedded directly, or into media applied or attached to the item to be marked. Each individual molecular tag is recorded
and stored in a secure database so that we can later detect it using a simple spot test, or the molecular tags can be forensically
analyzed in our laboratories to obtain definitive proof of the presence or absence of a specific SigNature molecular tag (e.g.,
one designed to mark a particular product). Our in-lab forensic testing capability delivers an expert witness Certificate of DNA
Authentication (“
CODA
”). Because DNA is one of the densest information carriers known, and can be amplified
with high fidelity, only minute quantities of SigNature molecular tags are necessary for successful analysis and authentication.
As a result, SigNature molecular tags can fold seamlessly into production and logistics workflows at extremely low concentrations.
SigNature molecular tags have been subjected
to rigorous testing by the Idaho National Laboratory, a U.S. National Laboratory, by CALCE (the Center for Advanced Life Cycle
Engineering), the largest electronic products and systems research center focused on electronics reliability, and by verified procedures
in our laboratories. The molecular tag has passed all tests across a broad spectrum of materials and substrates, and has met key
military stability standards. SigNature molecular tags have also passed a strenuous “red-team” vetting on behalf of
the U.S. Defense Logistics Agency.
Hundreds of millions of SigNature molecular
tags now exist on items ranging from consumer product packaging to microcircuits to cotton and synthetic fibers; to our knowledge,
none has ever been copied.
SigNature T Molecular Tags and fiberTyping
SigNature T Molecular Tags.
SigNature
T molecular tags are a unique patented tagging and authentication system specifically designed for textiles and apparel. Specially
engineered to adhere tenaciously to textile substrates, including natural and synthetic fibers, SigNature T molecular tags are
resistant to standard textile production conditions, and cannot be copied. The result: an enduring forensic level molecular tag
that remains present from the fiber stage through to the finished product.
Our SigNature T technology allows for better
quality control and assurance at any point in the supply chain. SigNature T molecular tags are currently used for brand protection
efforts and raw material source compliance programs. For example, American grown cotton fibers can be tagged at the gin in the
United States, verified as “American grown” and then traced through every step of the supply chain.
fiberTyping
. Our patented
cotton genotyping platform, known as “fiberTyping,” described below, complements our SigNature T molecular tag system.
fiberTyping is employed to identify the genus and species of the fibers before or after they are tagged with SigNature T molecular
tags. fiberTyping cannot be used to provide unique identity or traceability of a specific cotton batch through the supply chain,
a function which can only be accomplished by our SigNature T molecular tag system combined with our digital software platform.
fiberTyping is not a molecular tag, but
a genotyping test of native cotton fiber DNA, which gives a clear result that determines whether the intended “nature-made”
endogenous cotton DNA is present in fiber, yarn or fabric. Samples from the primary material are sent to our forensic labs for
DNA analysis and authentication. Cotton classification and the authentication of cotton species after cotton has left its place
of origin are issues of global significance, important to brand owners and to governments that must regulate the international
cotton trade. The use of endogenous DNA to identify the cotton fiber content of finished textiles, along with the SigNature T molecular
tag system is a significant opportunity for brand license holders to control their intellectual property, for brands to shield
themselves against legal liabilities, and for governments to improve their ability to enforce compliance with trade agreements
between nations.
In addition to the global cotton trade,
the potential markets for genotyping include biotherapeutics, nutraceuticals, natural foods, wines and fermented alcohols and other
natural textiles.
We believe that our proprietary DNA extraction
protocols and methodologies are more effective than existing forensic systems. We believe that the combination of our SigNatureT
molecular tags and fiberTyping solutions cover the forensic authentication market for textiles and that the related protocols we
have developed may be applicable to multiple industry verticals, and can mark and authenticate products at every stage of their
life cycle, from beginning to end.
DNAnet, Smart DNA and Backtrac
Recognizing that DNA-based evidence is the
cornerstone of modern-era law enforcement, we have developed what we believe to be the ultimate crime fighting tools – currently
being used in home asset and vehicle marking, as well as commercial applications.
These molecular tags can be used to definitively
link evidence and offenders to specific crime scenes. As the crime is investigated, the fluorescing molecular marker can assist
police in linking the offender and stolen items to a specific crime scene, creating a greater ability to identify and convict.
These long lasting tagging solutions contain
unique molecular tags that can help return stolen or lost property to its rightful owner.
Beacon
Beacon locked optical markers deliver secure
real-time inspection capabilities. A unique encrypted mechanism (patent-pending) creates a protected, covert screening tool that
can be easily adapted to packaging, security labels and high–value assets through inks, varnishes and coatings. When Beacon
locked optical markers are combined with SigNature molecular tags, a strong and flexible end-to-end security solution is created
where authenticity and provenance can be determined with confidence.
SigNify
Developing a secure method for real-time,
in-field screening of molecularly-tagged items has long been a priority for us. We believe that standard fluorophores, up-converting
phosphors, holograms and other more-traditional screening tools provide little to no defense against counterfeiting. We believe
that secure in-field inspection backed with forensic-level molecular tag authentication is the key to maintaining a well-defended
supply chain or asset management program.
The SigNify IF portable DNA reader provides
definitive real-time authentication of SigNature and SigNature T molecular tags in the field. With SigNify IF, Signature molecular
tags become a true, front-line solution for supply chain integrity.
Information Technology Systems
digitalDNA.
digitalDNA
is a software platform that enables customers to manage the security of company-marked goods from point of marking to point of
authentication or validation to end of life. The base platform is configurable to customer requirements which differ by vertical
market, company business process and IT environment. Basic functions offered include molecular tag inventory management, program
training and communications, a database of marked items information, associated documents and images, chain of custody and location
tracking, sample authentication processing and CODA downloads, and other administrative functions. Designed for either cloud or
local operation, the system supports mobile data capture using bar codes or other technologies. Of special note is the power of
embedding our proprietary DNA into tag ink or substrate as a covert method of forensic authentication to be recorded on the system.
The system is architected as the controller and repository for other validation and authentication devices such as our SigNify
DNA Readers, Multi-Mode Reader (prototype), DNA Transfer Systems, and other third party devices and is designed to share data with
third party applications through standard interfaces.
DNA Transfer Systems.
Our
DNA Transfer Systems are developed for DNA marking applications which are high volume with a need for monitoring and control. They
are computer based, fully automated, offer remote internet access for real-time monitoring and can be configured for application-specific
alerts and reporting online. They were used to mark cotton at eight U.S. cotton gins in the 2016 ginning season.
CertainT Supply Chain Platform
CertainT helps brands confirm their product’s
authenticity and origin with certified, trust, transparency and traceability through the seamless amalgamation of several of our
platform technologies to tag, test and track. The CertainT trademark indicates use of the CertainT tagging, testing and tracking
platform to enable proof of product claims for any material, item or product. Secure and proven, the CertainT Platform helps manufacturers,
brands or other commercial organizations deliver on their promise that customers are buying products that are ethically-sourced,
safe and authentic.
Large-scale production of specific DNA sequences using
PCR.
Our patented Triathlon™ PCR
systems allow for the large-scale production of specific DNA sequences. The systems are self-contained and modular, can work together
in mass production or can be used individually throughout the world, offering the advantage of delivering DNA locally and securely.
These DNA sequences are being used by customers as a diagnostic and reagent and provide us the opportunity to cross-sell our DNA-based
supply chain security solutions. We have the ability to manufacture longer DNA sequences valuable in gene therapy, DNA vaccines
and diagnostics, with what we believe is a distinct competitive advantage in cost, cleanliness, and time-to-market. These types
of DNA are distinct from our DNA security markers and represent a potential new entry into medical markets, where we believe there
are opportunities for our broader platform.
Recent Developments
Himatsingka.
In June 2017,
we announced that we had entered into a new licensing agreement with Himatsingka America, Inc., which is part of the Himatsingka
Group (“Himatsingka America”), a leading supplier of home textiles. This agreement terminates an earlier licensing
agreement dated March 25, 2015 between Divatex Home Fashion, Inc. (a predecessor to Himatsingka America) and the Company. Under
the terms of the Agreement, Himatsingka America will be solely responsible for promoting, marketing and selling on a worldwide
basis the Company’s technology with respect to finished and unfinished cotton products. The Agreement grants Himatsingka
America an exclusive license to use our technology in respect of cotton, subject to certain carve-outs including governmental users,
non-commercial trade associations and others. The Agreement has a term that continues until June 23, 2042, except in the case of
patents, in which case the term continues with respect to a patent until such patent is no longer in effect. The Agreement also
provides that Himatsingka America will make payments for the use of the Company’s taggant technology on a net 60 days basis.
In addition, Himatsingka America will make royalty payments on a quarterly basis in arrears in the event that our technology is
used on non-home products. Himatsingka America is responsible for the inspection and compliance within the supply chain.
Himatsingka America is generally required
to use our technology during the term of the Agreement, subject, among other things, to their customers’ requirements. We
are establishing an independent testing laboratory in Ahmedabad, India, which is required by the agreement. Finished products made
from this tagged fiber will be offered for sale under the PimaCott®, HomeGrown™, LoneStar™ and HomeGrown Acala™
content branded labels. The Agreement includes customary mutual indemnification provisions.
Office of the Secretary of Defense.
On June 6, 2017, we were awarded a two-year, approximately $1.5 million competitive-bid development contract. The award, funded
by the Office of the Secretary of Defense on behalf of the DLA, runs from June 1, 2017 to May 31, 2019, and was granted via a Rapid
Innovation Fund (RIF) that provides DLA with innovative technologies that can be rapidly inserted into acquisition programs to
meet specific defense needs. Management oversight for this RIF contract will be from DLA headquarters located in Fort Belvoir,
Virginia. This firm-fixed price contract follows our prior RIF contract, which expired during August 2016, that enabled us to develop
counterfeit mitigation technologies based upon our proprietary DNA platforms, which protect plastics, silicone elastomers, oils,
bearings, fasteners and many other high-risk commodities that are procured by DLA on behalf of DoD. This contract will extend our
authentication platform to facilitate broader use in protecting high-risk or mission-critical material purchased by DLA.
GHCL Limited.
On June 28,
2017, we signed a multi-year license agreement with GHCL Limited (“GHCL”), a global manufacturer of home textiles,
to provide CertainT platform services in connection with source-verified, polyethylene terephthalate (PET) and recycled PET (rPET
post-consumer) in select home textile products. PET is the clear plastic best known for its use in water bottles, and is the most
widely recycled plastic in the world. GHCL will use our CertainT platform in connection with PET and/or recycled PET blended bed
sheets, pillowcases, and shams products sold in-store or online in the United States. GHCL has also licensed our CertainT trademark
for use on its products, as well as for promotional, marketing and sales materials. The agreement provides for guaranteed minimum
annual revenues in order to maintain exclusivity during the renewal period, as well as trademark licensing royalties to us. GHCL
will use our CertainT platform for verifying PET and recycled PET authenticity from source to retail shelf. With this platform,
GHCL assures that any of its textile products using PET and recycled PET will contain the original source raw materials. We will
provide our patented and proprietary tagging, testing and tracking services to GHCL as a CertainT licensee. As part of the platform,
Applied DNA’s molecular tag is extruded into recycled components that create recycled PET fiber, with no impact to performance
or quality of the fiber or filament yarns. Thereafter, any piece of CertainT-tagged textiles can be forensically authenticated
by detecting the molecular tag in the recycled PET fiber, ensuring its authenticity and origin.
Loftex Home.
On July 11, 2017,
we signed a new multi-year exclusive license agreement with Loftex Home, LLC (“Loftex”), a well-respected manufacturer
of high-quality towels and home textiles. Under a prior agreement entered into during March 2017, we agreed to provide our CertainT™
platform services to Loftex to verify the authenticity and origin of rPET (post-consumer) used in bath and beach towels. This new
multi-year agreement between the two companies is now exclusive for bath and beach towels in the United States, non-exclusive for
plush throws and bath rugs, and provides for long term minimum annual revenues, in order to maintain exclusivity, as well as trademark
licensing royalties to us.
CRADA.
Effective as of August
22, 2017, we signed a Cooperative Research and Development Agreement (“CRADA”) with the U.S. Army Research, Development
and Engineering Command’s Edgewood Chemical Biological Center (“ECBC”) to study the commercialization of ECBC’s
innovative rapid, in-field DNA microarray technology for use in military and commercial supply chains. ECBC is the nation’s
primary DoD technical research organization for non-medical chemical and biological warfare defense.
Under the terms of the CRADA, a cooperative
effort under the recently-announced DLA Rapid Innovation Fund award secured by us in June 2017, ECBC’s subject matter experts
and our science team will cooperatively study the feasibility of commercializing ECBC’s in-field DNA detection technology
in varied supply chains. ECBC’s hand-held in-field DNA microarray technology allows for detection of a DNA taggant within
a few minutes. The project goal is to demonstrate the system with our taggants introduced into standard inks or varnishes or onto
other surfaces, without the need for DNA amplification or other sample preparation, thus greatly simplifying in-field DNA detection.
Videojet.
During September
2017, we entered into a strategic partnership with Videojet Technologies Inc. (“Videojet”). We have collaborated with
Videojet in the design of co-branded SigNature® molecular-tagged Videojet inks, and a co-branded printer that electronically
restricts the use of ink cartridges to only those that contain SigNature molecular tagged inks. The relationship brings the potential
to empower the tagging of countless commercial items, all of which are candidates for a CertainT licensing agreement, enabling
traceability along the entire supply chain. These solutions were jointly introduced during the last week of September 2017 at Pack
Expo 2017 in Las Vegas, NV.
Rosier S.A.
On August 8,
2017, we announced the introduction of our molecular tag to the fertilizer industry in cooperation with Rosier S.A. (“Rosier”),
a mineral fertilizer manufacturer based in Moustier, Belgium. Rosier sells high quality mineral fertilizers globally, and in Europe,
through its exclusive distributor, Borealis L.A.T. Together with Rosier we launched a pilot to DNA-tag fertilizer pellets in order
to detect the dilution of genuine fertilizer with sub-standard material within a given batch, and to be able to trace the batch
to its original manufacturing location. Since the initiation of this study, we, in partnership with Rosier, have effectively marked
fertilizer pellets and have successfully authenticated and detected the dilution of fertilizer with unmarked material in a variety
of laboratory and in-field tests over a nine-month period. A marked shipment of fertilizer has travelled through the supply chain
in West Africa and pellets have been analyzed in the field utilizing our in-field DNA detection technology (SigNify® IF) to
provide definitive real-time authentication of the SigNature DNA molecular tags, ensuring that the fertilizer had not been adulterated
with unmarked material. The pellets tested were proven to be genuine and demonstration of the technology gained further support
for the use of molecular taggants to combat counterfeiting and to aid the many countries that are affected by adulterated fertilizer.
Colorcon, Inc.
On December
14, 2017, we entered into a non-binding technology license memorandum of understanding with Colorcon, Inc. (“Colorcon”).
Under the terms of the memorandum, we will receive milestone payments as well as revenue sharing for product sales and authentication
services. The memorandum will enable us to combine our molecular taggant and authentication technology with Colorcon’s portfolio
of film coating systems, inks and color dispersions for use in solid oral dosage forms in the pharmaceutical and nutraceutical
industries. Our collaboration with Colorcon will commercialize a platform for traceability directly on dose, and is intended to
significantly reduce the risks associated with counterfeit and falsified medications entering the drug supply chain.
Preliminary Fiscal Year 2017 and Fourth Quarter Results
On December 15, 2017, the Company announced
selected preliminary unaudited financial results for the fiscal year and quarter ended September 30, 2017.
Preliminary (unaudited) fiscal 2017
financial results.
Revenues increased 13% to $4.8 million as compared to $4.2 million in the prior fiscal year. The increase
in year-over-year total revenues is attributable to increases in product revenue of approximately $1.4 million in the textile industry
primarily for protecting cotton supply chains, offset by a net decrease in service revenues from government contract awards of
approximately $800 thousand, primarily as a result of two awards which expired in July and August 2016.
Total operating expenses were $16.5 million,
compared with $15.2 million in the prior fiscal year, an increase of approximately $1.3 million or 8%. The increase is primarily
attributable to an increase in stock based compensation expense, offset by a decrease in R&D due to the expiration of the two
government contract awards.
Net loss for fiscal 2017 was $12.9 million,
or $0.49 per share, compared with a net loss of $12.2 million, or $ 0.51 per share, in the prior fiscal year.
Preliminary (unaudited) fiscal 2017
fourth quarter results.
Revenues decreased 30% for the fourth quarter of fiscal 2017 to $1.1 million, compared with $1.6
million reported in the fourth quarter of fiscal 2016, and decreased 36% from the $1.8 million reported in the third fiscal quarter
ended June 30, 2017. The year-over-year quarterly decrease in revenues resulted primarily from decreased sales to the textile industry.
The year-over-year quarterly decrease in textile industry sales was due to the fact that the shipments of DNA concentrate took
place in the third quarter of fiscal 2017 as compared to fiscal 2016 when they took place in the fourth quarter, as well as the
restructuring of payment terms under the June 23, 2017 licensing agreement with Himatsingka America. The timing of the shipments
is also the reason for the decrease in revenues for the fourth quarter of fiscal 2017 as compared to the prior fiscal quarter.
Total operating expenses remained fairly
flat at $3.7 million for the fourth fiscal quarter of 2017, compared with $3.6 million in the prior fiscal year’s quarter.
Net loss for the quarter ended September
30, 2017 was $2.9 million, or $0.10 per share, compared with a net loss of $2.4 million, or $0.10 per share, for the quarter
ended September 30, 2016 and a net loss of $2.6 million, or $0.10 per share, for the quarter ended June 30, 2017.
The preliminary fiscal year 2017 and fourth
quarter financial data is unaudited, preliminary, based upon the Company’s good faith estimates and subject to completion
of the Company's financial closing procedures. While the Company expects that its final financial results for the fiscal year and
quarter ended September 30, 2017, following the completion of its financial closing procedures, will generally be consistent with
the amounts provided herein, the Company's actual results may differ materially from these estimates as a result of the completion
of its financial closing procedures, as well as final adjustments and other developments that may arise between now and the time
that its financial results for the fiscal year and quarter ended September 30, 2017 are finalized.
The foregoing preliminary results are subject
to completion and audit of the Company’s financial statements in conjunction with the Company’s 2017 Form 10-K filing
with the Securities and Exchange Commission anticipated to occur by December 29, 2017.
Corporate History
We are a Delaware corporation, which was
initially formed in 1983 under the laws of the State of Florida as Datalink Systems, Inc. In 1998, we reincorporated in Nevada,
and in 2002, we changed our name to our current name, Applied DNA Sciences, Inc. In December 2008, we reincorporated from Nevada
to the State of Delaware.
Our corporate headquarters are located at
the Long Island High Technology Incubator at Stony Brook University in Stony Brook, New York, where we established laboratories
for the manufacture of DNA markers and product prototypes, and DNA authentication. The address of our corporate headquarters is
50 Health Sciences Drive, Stony Brook, New York 11790, and our telephone number is (631) 240-8800. We maintain a website at
www.adnas.com
where general information about us is available. The information on, or that may be accessed through, our website is not incorporated
by reference into and should not be considered a part of this registration statement.
To date, we have produced limited recurring
revenues from our products and services, have incurred expenses and have sustained losses. Consequently, our operations are subject
to all the risks inherent in the establishment of a biotechnology company.
THE
OFFERING
|
Securities offered:
|
5,470,000 shares of our common stock, including 2,735,000 shares
of common stock underlying the Purchase Warrants
Purchase Warrant s to purchase 2,735,000
shares of our common stock
|
|
|
|
|
Combined offering price per share of common stock and accompanying Purchase Warrant:
|
$1.75 per share and Purchase Warrant
|
|
|
|
|
Common stock outstanding before the offering
(1)
:
|
27,377,057 shares
|
|
|
|
|
Common stock to be outstanding after the offering
(1)(2)
:
|
30,112,057 shares
|
|
|
|
|
Use of Proceeds:
|
We intend to use the net proceeds from this offering for working capital, capital expenditures, business development and research and development expenditures and acquisitions of new technologies or businesses.
|
|
|
|
|
Listing and Symbols:
|
Our common stock is listed on The NASDAQ Capital Market under the symbol “APDN.”
|
|
|
|
|
Purchase Warrants
|
We are issuing to purchasers of shares of our common stock in this offering a Purchase Warrant to purchase one share of
our common stock for each share purchased in this offering for a combined purchase price of $1.75. The Purchase
Warrants will be exercisable beginning on the Initial Exercise Date at an exercise price of $2.00 per share and will expire
on the five year anniversary of the Initial Exercise Date. The Purchase Warrants include an adjustment provision that, subject
to certain exceptions, reduces their exercise price if the Company issues Common Stock or Common Stock equivalents at a price
lower than the then-current exercise price of the Purchase Warrants, subject to a minimum exercise price of $0.44 per share.
See “Description of Securities we are Offering—Warrants.”
|
|
|
|
|
Risk Factors:
|
Investing in our securities involves substantial risks. You should carefully review and consider the “Risk Factors” section of this prospectus supplement beginning on page S-14 and on page 6 of the accompanying base prospectus, as well as the other information in this prospectus supplement for a discussion of the factors you should consider before you decide to invest in this offering.
|
|
|
(1)
|
The number of shares of our common stock outstanding as of December
18
, 2017 excludes
5,333,227 shares of common stock issuable upon exercise of outstanding stock options, at a weighted average exercise price of $
3.70
per share and
9,540,455
shares of common stock issuable upon exercise of outstanding
warrants at a weighted average exercise price of $
3.60
per share.
|
|
|
(2)
|
The total number of shares of our common stock outstanding after
this offering is based on
27,377,057
shares
outstanding as of December
18
, 2017 and does
not give effect to any exercise of the Purchase Warrants.
|
RISK
FACTORS
Investment in our securities involves a
high degree of risk. In addition to the risks and investment considerations discussed elsewhere in this prospectus supplement or
any document incorporated by reference herein, the following factors should be carefully considered by anyone purchasing the securities
offered by this prospectus supplement. The risks and uncertainties described below are not the only ones we face. Additional risks
and uncertainties not presently known to us or that we currently deem immaterial also may impair our business operations. We also
update risk factors from time to time in our periodic reports on Forms 10-K, 10-Q and 8-K which will be incorporated by reference
in this prospectus supplement. If any of the following risks actually occur, our business could be harmed. In such case, the trading
price of our common stock could decline and investors could lose all or a part of their investment.
See also the statements contained under
the heading “Forward Looking Statements.”
Risks Relating to Our Business:
The financial data included in this prospectus for
our fiscal year and fourth quarter ended September 30, 2017 is unaudited, preliminary and may change upon completion of the audit
of our financial statements in conjunction with our 2017 Form 10-K filing.
Included in this prospectus under “Preliminary Fiscal
Year 2017 and Fourth Quarter Results” is certain preliminary financial data for our fiscal year and fourth quarter ended
September 30, 2017. This financial data is unaudited, preliminary and based upon our good faith estimates and is subject to the
completion of our financial closing procedures and the audit of our financial statements that occurs in conjunction with our 2017
Form 10-K filing with the Securities and Exchange Commission anticipated to occur by December 29, 2017. While we expect that our
final financial results for the fiscal year and quarter ended September 30, 2017, following the completion of our financial closing
procedures, will generally be consistent with the financial data included in this prospectus, our actual results may differ materially
from these estimates as a result of the completion of our financial closing procedures, as well as final adjustments and other
developments that may arise between now and the time that our financial results for the fiscal year and quarter ended September
30, 2017 are finalized.
We have not produced significant revenues. This
makes it difficult to evaluate our future prospects and increases the risk that we will not be successful.
Our operations since inception have produced limited revenues,
and may not produce significant revenues in the near term, or at all, which may harm our ability to obtain additional financing
and may require us to reduce or discontinue our operations. If we create significant revenues in the future, we expect to derive
most of such revenues from the sale of supply chain security and product authentication solutions. You must consider our business
and prospects in light of the risks and difficulties we will encounter as a company operating in a rapidly evolving industry. We
may not be able to successfully address these risks and difficulties, which could significantly harm our business, operating results,
and financial condition.
We have a history of net losses which may continue,
and which may harm our ability to obtain financing and continue our operations.
We reported on a preliminary (unaudited) basis net losses of
$12.9 million and $2.9 million for the fiscal year and fourth quarter ended September 30, 2017, respectively. See “Preliminary
Fiscal Year 2017 and Fourth Quarter Results.” We incurred net losses of $12.2 million for the fiscal year ended September
30, 2016. We incurred net losses of $10.0 million for the nine months ended June 30, 2017. These net losses have principally been
the result of the various costs associated with our selling, general and administrative and research and development expenses
as we expanded operations, acquired, developed and validated technologies, and expanded marketing activities. Our operations
are subject to the risks and competition inherent in a company that moved from the development stage to an operating company.
We may not generate sufficient revenues from operations to achieve or sustain profitability on a quarterly, annual or any other
basis in the future. Our revenues and profits, if any, will depend upon various factors, including whether our existing products
and services or any new products and services we develop will achieve market acceptance. If we continue to incur losses, then
our accumulated deficit will continue to increase which may significantly impair our ability to obtain additional financing. As
a result, our business, results of operations and financial condition would be significantly harmed, and we may be required to
reduce or terminate our operations.
If we are unable to obtain additional financing
our business operations may be harmed or discontinued.
Our continuation as a going concern is dependent upon our future
revenues and our ability to commercialize more products, obtain additional capital and attain profitable operations. We may require
additional funds to complete the continued development and commercialization of our products, product manufacturing, and to fund
expected additional losses from operations, until revenues are sufficient to cover our operating expenses. If we are unsuccessful
in obtaining any necessary additional financing, we will most likely be forced to reduce or terminate our operations.
Our operating results could be adversely affected
by a reduction in business with our significant customers.
Our revenue earned from the sale of products and services for
the fiscal year ended September 30, 2016 included an aggregate of 75% of our total revenues from three customers. These three customers
accounted for approximately 20% of our total accounts receivable at September 30, 2016. At September 30, 2016, one customer accounted
for an aggregate of 78% of our total accounts receivable. Our revenues earned from sale of products and services for the fiscal
year ended September 30, 2015 included an aggregate of 79% from two customers of our total revenues. These two customers accounted
for approximately 90% of our total accounts receivable at September 30, 2015. The Company’s revenues earned from sale of
products and services for the nine month period ended June 30, 2017 included an aggregate of 15%, 18%, and 38% from three customers,
respectively. Two customers accounted for 59% and 32% of the Company’s accounts receivable at June 30, 2017.
Generally, our customers do not have an obligation to make purchases
from us and may stop ordering our products and services or may terminate existing orders or contracts at any time with little or
no financial penalty. The loss of any of our significant customers, any substantial decline in sales to these customers, or any
significant change in the timing or volume of purchases by our customers, could result in lower revenues and could harm our business,
financial condition or results of operations.
If our existing products and services are not accepted
by potential customers or if we fail to introduce new products and services, our business, results of operations and financial
condition will be harmed.
There has been limited market acceptance of our botanical DNA
based technology, encapsulation, embedment and authentication products and services to date. Some of the factors that will affect
whether we achieve market acceptance of our solutions include:
|
|
·
|
availability, quality and price relative to competitive solutions;
|
|
|
·
|
customers’ opinions of the solutions’ utility;
|
|
|
·
|
consistency with prior practices;
|
|
|
·
|
scientists’ opinions of the solutions’ usefulness; and
|
|
|
·
|
general trends in anti-counterfeit and security solutions’ research.
|
Dependence on channel partners.
Our future growth will depend to a material extent on the successful
advocacy of our technology by channel partners to their members and customers, and implementation of our technology in solutions
propagated by channel partners and provided by third parties. Our business has relied on the success of business partners. Our
continuing success is largely dependent on a new generation of business partners involved in our tagging technology.
If our channel partners are not successful in advocating and
deploying our technology, we may not be able to achieve and sustain profitable operations. If other business partners who include
our technology in their products or otherwise license our intellectual property for use in their products cease to do so, or we
fail to obtain other partners who will incorporate, embed, integrate or bundle our technology, or these partners are unsuccessful
in their efforts, expanding deployment of our technology and increasing revenues will be adversely affected. Consequently, our
ability to increase revenue could be adversely affected and we may suffer other adverse effects to our business. In addition, if
our technology does not perform according to market expectations, our future sales would suffer as customers seek and employ alternative
technologies.
Many of our business endeavors can be impeded or frustrated
by larger, more influential companies or by standard-setting bodies or institutions downplaying, minimizing or rejecting the value
or use of our technology. A negative position by such companies, bodies or institutions, could result in obstacles for us that
we would be incapable of overcoming and may block or impede the adoption of our technology. In addition, potential customers may
delay or reject initiatives that relate to deployment of our technology. Such a development would make the achievement of our business
objectives in this market difficult or impossible.
The expenses or losses associated with lack of widespread
market acceptance of our solutions may harm our business, operating results and financial condition.
Rapid technological changes and frequent new product introductions
are typical in the markets we serve. Our future success may depend in part on continuous, timely development and introduction of
new products that address evolving market requirements. We believe successful new product introductions may provide a significant
competitive advantage because customers invest their time in selecting and learning to use new products, and once invested in the
new technology, are often reluctant to switch products. To the extent we fail to introduce new and innovative products, we may
lose any market share we then have to our competitors, which will be difficult or impossible to regain. Any inability, for technological
or other reasons, to successfully develop and introduce new products could reduce our growth rate or damage our business. We may
experience delays in the development and introduction of products. We may not keep pace with the rapid rate of change in anti-counterfeiting
and security products’ research, and any new products acquired or developed by us may not meet the requirements of the marketplace
or achieve market acceptance.
We need to expand our sales, marketing and support
organizations and our distribution arrangements to increase market acceptance of our products and services.
We currently have a limited number of sales, marketing, customer
service and support personnel and will need to increase our staff to generate a greater volume of sales and to support any new
customers or the expanding needs of existing customers. The employment market for sales, marketing, customer service and support
personnel in our industry is very competitive, and we may not be able to hire the kind and number of sales, marketing, customer
service and support personnel we are targeting. Our inability to hire qualified sales, marketing, customer service and support
personnel may harm our business, operating results and financial condition. While we have entered into a limited number of agreements
with distributors, we may not be able to sufficiently build out a distribution network or enter into arrangements with qualified
distributors on acceptable terms or at all. If we are not able to develop greater distribution capacity, we may not be able to
generate sufficient revenue to support our operations.
If we are unable to continue to retain the services
of Dr. Hayward, we may not be able to continue our operations.
Our success depends to a significant extent upon the continued
service of Dr. James A. Hayward, our Chairman, Chief Executive Officer and President. On July 28, 2016, we entered into a new employment
agreement with Dr. Hayward. The initial term was from July 1, 2016 through June 30, 2017, with automatic one-year renewal periods.
As of June 30, 2017, the employment contract renewed for an additional year. Loss of the services of Dr. Hayward could significantly
harm our business, results of operations and financial condition. We do not maintain key-person insurance on the life of Dr. Hayward.
The markets for our supply chain security and product
authentication solutions are very competitive, and we may be unable to continue to compete effectively in these industries in the
future.
The principal markets for our supply chain security and product
authentication solutions are intensely competitive. Many of our competitors, both in the United States and elsewhere, are major
pharmaceutical, chemical and biotechnology companies, or have strategic alliances with such companies, and many of them have substantially
greater capital resources, marketing experience, research and development staff, and facilities than we do. Any of these companies
could succeed in developing products that are more effective than the products that we have or may develop and may be more successful
than us in producing and marketing their existing products. Some of our competitors that operate in the supply chain security and
fraud prevention markets include: 3DTL Inc., Alp Vision Sa, Authentix Inc., Brandwatch Technologies, Chromologic LLC, Collectors
Universe Inc., Collotype Labels International, Data Dot Technology, De La Rue Plc., Digimarc Corp., DNA Technologies, Inc., FractureCode
Corporation, Haelixa, ICA Bremen GmbH, Ipsidy Inc., IEHCorporation, Informium AG, Eastman Kodak Company, opSec Security Group plc.,
MicroTagTemed Ltd., Nanotech Security Corp., Nokomis, Inc., ProoftagSAS, SafeTraces Inc., Selectamark Security Systems plc, Spectra
Systems Corp., SmartWater Technology Limited, Sun Chemical Corp, TraceTag International, TruTag Technologies Inc., and YottaMark
Inc.
We expect this competition to continue and intensify in the
future. Competition in our markets is primarily driven by:
|
|
·
|
product performance, features and liability;
|
|
|
·
|
timing of product introductions;
|
|
|
·
|
ability to develop, maintain and protect proprietary products and technologies;
|
|
|
·
|
sales and distribution capabilities;
|
|
|
·
|
technical support and service;
|
|
|
·
|
applications support; and
|
|
|
·
|
breadth of product line.
|
If a competitor develops superior technology or cost-effective
alternatives to our products, our business, financial condition and results of operations could be significantly harmed.
Revenues from our customer contracts with respect
to cotton will be seasonal and may also be subject to weather conditions and other factors beyond our control, which may cause
our operating results to fluctuate significantly quarterly and annually.
A significant and growing proportion of our revenues is
expected to derive from customer contracts for tagging, authentications and other services related to cotton. The cotton
ginning season in the United States takes place between September and March each year. Therefore, revenues from our customer
contracts relating to cotton will be seasonal, which may cause our operating results to fluctuate significantly quarterly and
annually. Additionally, weather and climatic conditions, natural disasters and other factors beyond our control also affect
the production and sale of cotton and other agricultural commodities to which our customer contracts may relate, as well as
our customers’ or prospective customers’ decisions regarding purchases of our products and services, and may
cause our operating results to fluctuate significantly quarterly and annually. The seasonal fluctuations in operating results
described above may cause a decline in the price of our common stock.
Fluctuations in quarterly results.
Our revenues and profitability are difficult to predict due
to the nature of the markets in which we compete, fluctuating user demand, the uncertainty of current and future global economic
conditions, and for many other reasons, including that our operating results are highly dependent on the volume and timing of orders
received during a quarter, which are difficult to forecast. Customers generally order on an as-needed basis and we typically do
not obtain firm, long-term purchase commitments from our customers.
Our research and development efforts for new products
may be unsuccessful.
We incur research and development expenses to develop new products
and technologies in an effort to maintain our competitive position in a market characterized by rapid rates of technological advancement.
Our research and development efforts are subject to unanticipated delays, expenses and technical problems. There can be no assurance
that any of these products or technologies will be successfully developed or that, if developed, will be commercially successful.
In the event that we are unable to develop commercialized products from our research and development efforts or we are unable or
unwilling to allocate amounts beyond our currently anticipated research and development investment, we could lose our entire investment
in these new products and technologies. Any failure to translate research and development expenditures into successful new product
introduction could have an adverse effect on our business.
Failure to license new technologies could impair
sales of our existing products or any new product development we undertake in the future.
To generate broad product lines, it is advantageous to sometimes
license technologies from third parties rather than depend exclusively on the development efforts of our own employees. As a result,
we believe our ability to license new technologies from third parties may be important to our ability to offer new products. In
addition, from time to time we are notified of, or become aware of patents held by third parties that are related to technologies
we are selling or may sell in the future. After a review of these patents, we may decide to seek a license for these technologies
from these third parties. There can be no assurance that we will be able to successfully identify new technologies developed by
others. Even if we are able to identify new technologies of interest, we may not be able to negotiate a license on favorable terms,
or at all.
Our failure to manage our growth in operations and
acquisitions of new product lines and new businesses could harm our business.
The recent growth in our operations could place a significant
strain on our current management resources. To manage such growth, we may need to improve our:
|
|
·
|
operations and financial systems;
|
|
|
·
|
procedures and controls; and
|
|
|
·
|
training and management of our employees.
|
Our future growth, if any, may be attributable to acquisitions
of new product lines and new businesses. Future acquisitions, if successfully consummated, would likely create increased working
capital requirements, which would likely precede by several months any material contribution of an acquisition to our net income.
Our failure to manage growth or future acquisitions successfully could seriously harm our operating results. Also, acquisition
costs could cause our operating results to vary significantly from quarter to quarter. Furthermore, our stockholders would be diluted
if we financed the acquisitions by incurring convertible debt or issuing securities.
A percentage of our sales occur outside of the U.S.
As a result, we are subject to the economic, political, regulatory and other risks of international operations.
For the nine months ended June 30, 2017, fiscal 2016, and fiscal
2015, 28%, 24% and 14%, respectively, of our revenue was from customers located outside of the U.S. We believe that the
revenue from the sale of our products outside the U.S. will continue to grow in the near future. We intend to expand our international
operations to the extent that suitable opportunities become available. Our foreign operations and sales could be adversely affected
as a result of:
|
|
·
|
nationalization of private enterprises and assets;
|
|
|
·
|
political or economic instability in certain countries and regions;
|
|
|
·
|
differences in foreign laws, including increased difficulties in protecting intellectual property and uncertainty in enforcement
of contract rights;
|
|
|
·
|
the possibility that foreign governments may adopt regulations or take other actions that could directly or indirectly harm
our business and growth strategy;
|
|
|
·
|
tariff and tax increases;
|
|
|
·
|
export and import restrictions and restrictive regulations of foreign governments;
|
|
|
·
|
shipping products during times of crisis or wars; and
|
|
|
·
|
other risks inherent in foreign operations.
|
We are subject to numerous regulatory, legal, operational,
and other risks as a result of our international operations which could adversely impact our businesses in many ways.
As a U.S. company, we are required to comply with the economic
sanctions and embargo programs administered by Office of Foreign Assets Control and similar multi-national bodies and governmental
agencies worldwide, and the Foreign Corrupt Practices Act (“FCPA”). A violation of a sanction or embargo program or
of the FCPA or similar laws prohibiting certain payments to governmental officials, such as the U.K. Bribery Act, could subject
us, and individual employees, to a regulatory enforcement action as well as significant civil and criminal penalties which could
adversely impact our business and operations.
Failure to attract and retain qualified scientific,
production and managerial personnel could harm our business.
Recruiting and retaining qualified scientific and production
personnel to perform and manage prototype, sample, and product manufacturing and business development personnel to conduct business
development are critical to our success. In addition, our desired growth and expansion into areas and activities requiring additional
expertise, such as clinical testing, government approvals, production, sales and marketing will require the addition of new management
personnel and the development of additional expertise by existing management personnel. Because our industry is very competitive,
we face significant challenges in attracting and retaining a qualified personnel base. Although we believe we have been, and will
continue to be, able to attract and retain these personnel, we cannot assure you that we will continue to be able to successfully
attract qualified personnel in the future. The failure to attract and retain these personnel or, alternatively, to develop this
expertise internally would harm our business since our ability to conduct business development and manufacturing would be reduced
or eliminated, resulting in lower revenues. We generally do not enter into employment agreements requiring our employees to continue
in our employment for any period of time, with the exception of our Chief Executive Officer. See “—If we are unable
to continue to retain the services of Dr. Hayward, we may not be able to continue our operations” above in “Risk Factors”.
Our intellectual property rights are valuable, and
any inability to protect them could reduce the value of our products, services and brand.
Our patents, trademarks, trade secrets, copyrights and all of
our other intellectual property rights are important assets for us. There are events that are outside of our control that pose
a threat to our intellectual property rights as well as to our products and services. For example, effective intellectual property
protection may not be available in every country in which our products and services are distributed. The efforts we have taken
to protect our proprietary rights may not be sufficient or effective. Any significant impairment of our intellectual property rights
could harm our business or our ability to compete. Protecting our intellectual property rights is costly and time consuming. Any
increase in the unauthorized use of our intellectual property could make it more expensive to do business and harm our operating
results. Although we seek to obtain patent protection for our innovations, it is possible we may not be able to protect some of
these innovations. Given the costs of obtaining patent protection, we may choose not to protect certain innovations that later
turn out to be important. There is always the possibility that the scope of the protection gained from one of our issued patents
will be insufficient or deemed invalid or unenforceable. We also seek to maintain certain intellectual property as trade secrets.
The secrecy could be compromised by third parties, or intentionally or accidentally by our employees, which would cause us to lose
the competitive advantage resulting from these trade secrets.
Intellectual property litigation could harm our
business, financial condition and results of operations.
Litigation regarding patents and other intellectual property
rights is extensive in the biotechnology industry. In the event of an intellectual property dispute, we may be forced to litigate.
This litigation could involve proceedings instituted by the U.S. Patent and Trademark Office or the International Trade Commission,
as well as proceedings brought directly by affected third parties. Intellectual property litigation can be extremely expensive,
and these expenses, as well as the consequences should we not prevail, could seriously harm our business.
If a third party claims an intellectual property right to technology
we use, we might need to discontinue an important product or product line, alter our products and processes, pay license fees or
cease our affected business activities. Although we might under these circumstances attempt to obtain a license to this intellectual
property, we may not be able to do so on favorable terms, or at all. Furthermore, a third party may claim that we are using inventions
covered by the third party’s patent rights and may go to court to stop us from engaging in our normal operations and activities,
including making or selling our products. These lawsuits are costly and could affect our results of operations and divert the attention
of managerial and technical personnel. A court may decide that we are infringing the third party’s patents and would order
us to stop the activities covered by the patents. In addition, a court may order us to pay the other party damages for having violated
the other party’s patents. The biotechnology industry has produced a proliferation of patents, and it is not always clear
to industry participants, including us, which patents cover various types of products or methods of use. The coverage of patents
is subject to interpretation by the courts, and the interpretation is not always uniform. If we are sued for patent infringement,
we would need to demonstrate that our products or methods of use either do not infringe the patent claims of the relevant patent
and/or that the patent claims are invalid, and we may not be able to do this. Proving invalidity, in particular, is difficult since
it requires a showing of clear and convincing evidence to overcome the presumption of validity enjoyed by issued patents.
Because some patent applications in the United States may be
maintained in secrecy until the patents are issued, because patent applications in the United States and many foreign jurisdictions
are typically not published until eighteen months after filing, and because publications in the scientific literature often lag
behind actual discoveries, we cannot be certain that others have not filed patent applications for technology covered by our or
our licensor’s issued patents or pending applications or that we or our licensors were the first to invent the technology.
During the ordinary course of our business, we do not conduct “prior art” searches before filing a patent application.
Our competitors may have filed, and may in the future file, patent applications covering technology similar to ours. Any such patent
application may have priority over our or our licensors’ patent applications and could further require us to obtain rights
to issued patents covering such technologies. If another party has filed a United States patent application on inventions similar
to ours, we may have to participate in an interference proceeding declared by the United States Patent and Trademark Office to
determine priority of invention in the United States. The costs of these proceedings could be substantial, and it is possible that
such efforts would be unsuccessful, resulting in a loss of our United States patent position with respect to such inventions.
Some of our competitors may be able to sustain the costs of
complex patent litigation more effectively than we can because they have substantially greater resources. In addition, any uncertainties
resulting from the initiation and continuation of any litigation could have a material adverse effect on our ability to raise the
funds necessary to continue our operations.
Accidents related to hazardous materials could adversely
affect our business.
Some of our operations require the controlled use of hazardous
materials for chemical reactions and synthesis. These materials are common to molecular/biological/chemical laboratories and require
no special handling or regulation. Although we believe our safety procedures comply with the standards prescribed by federal, state,
local and foreign regulations, the risk of accidental contamination of property or injury to individuals from these materials cannot
be completely eliminated. In the event of an accident, we could be liable for any damages that result, which could seriously damage
our business and results of operations.
Potential product liability claims could affect
our earnings and financial condition.
We face a potential risk of liability claims based on our products
and services. Though we have product liability insurance coverage which we believe is adequate, we may not be able to maintain
this insurance at reasonable cost and on reasonable terms. We also cannot assure that this insurance, if obtained, will be adequate
to protect us against a product liability claim, should one arise. In the event that a product liability claim is successfully
brought against us, it could result in a significant decrease in our liquidity or assets, which could result in the reduction or
termination of our business.
Litigation generally could affect our financial
condition and results of operations.
We generally may be subject to claims made by and required to
respond to litigation brought by customers, former employees, former officers and directors, former distributors and sales representatives,
former consultants and vendors and service providers. We have faced such claims and litigation in the past and we cannot assure
you that we will not be subject to claims in the future. In the event that a claim is successfully brought against us, considering
our lack of material revenue and the losses our business has incurred for the period from our inception to September 30, 2017,
this could result in a significant decrease in our liquidity or assets, which could result in the reduction or termination of our
business.
Business disruptions could seriously harm our future
revenue and financial condition and increase our costs and expenses.
Our operations could be subject to earthquakes, power shortages,
telecommunications failures, cyber-attacks or other vulnerabilities in our computer systems, terrorism, water shortages, tsunamis,
floods, hurricanes, typhoons, fires, extreme weather conditions, medical epidemics, political or economic instability, and
other natural or manmade disasters or business interruptions. The occurrence of any of these business disruptions could seriously
harm our revenue and financial condition and increase our costs and expenses.
General economic conditions may adversely affect
our business, operating results and financial condition.
A general weakening or decline in the global economy or a period
of economic slowdown may have serious negative consequences for our business and operating results. Since our customers incorporate
our products into a variety of consumer goods, the demand for our products is subject to worldwide economic conditions and their
impact on levels of consumer spending. Some of the factors affecting consumer spending include general economic conditions, unemployment,
consumer debt, reductions in net worth, residential real estate and mortgage markets, taxation, energy prices, interest rates,
consumer confidence and other macroeconomic factors. During periods of economic weakness or uncertainty, demand for consumer goods
incorporating our products may weaken, and current or potential customers may defer purchases of our products.
A cybersecurity incident and other technology disruptions
could negatively affect our business and our relationships with customers.
We use technology in substantially all aspects of our business
operations. The widespread use of technology, including mobile devices, cloud computing, and the internet, give rise
to cybersecurity risks, including security breach, espionage, system disruption, theft and inadvertent release of information. Our
business involves the storage and transmission of numerous classes of sensitive and/or confidential information and intellectual
property, including information relating to customers and suppliers, private information about employees, and financial and strategic
information about us and our business partners. If we fail to effectively assess and identify cybersecurity risks associated
with the use of technology in our business operations, we may become increasingly vulnerable to such risks. Additionally,
while we have implemented measures to prevent security breaches and cyber incidents, our preventative measures and incident response
efforts may not be entirely effective. The theft, destruction, loss, misappropriation, or release of sensitive and/or
confidential information or intellectual property, or interference with our information technology systems or the technology systems
of third parties on which we rely, could result in business disruption, negative publicity, brand damage, violation of privacy
laws, loss of customers, potential liability and competitive disadvantage.
Risks Relating to Our Common Stock and Other Securities:
There are a large number of shares of common stock
underlying our outstanding options and warrants and the sale of these shares may depress the market price of our common stock and
cause immediate and substantial dilution to our existing stockholders.
As of December
19
,
2017, we had 27,377,057 shares of common stock issued and outstanding, outstanding options to purchase 5,333,227 shares
of common stock and outstanding warrants to purchase
9,540,455
shares of common stock.
The issuance of shares upon exercise of our outstanding options and warrants will cause immediate and substantial dilution to
our stockholders. In addition, under our publicly traded warrants (including additional warrants sold privately that have registration
rights), in the event of a “Fundamental Transaction” (as defined in the related warrant agreement, which generally
includes any merger with another entity, the sale, transfer or other disposition of all or substantially all of our assets to
another entity, or the acquisition by a person of more than 50% of our common stock), each warrant holder will have the right
at any time prior to the consummation of the Fundamental Transaction to require us to repurchase the warrant for a purchase price
in cash equal to the Black Scholes value (as calculated under the warrant agreement) of the then remaining unexercised portion
of such warrant on the date of such Fundamental Transaction, which may materially adversely affect our financial condition and/or
results of operations.
We may require additional financing which may in
turn require the issuance of additional shares of common stock, preferred stock or other debt or equity securities (including convertible
securities) and which would dilute the ownership held by our stockholders.
We may need to raise funds through either debt or the sale of
our shares of our common stock in order to achieve our business goals. Any additional shares issued would further dilute the percentage
ownership held by the stockholders. Furthermore, if we raise funds in equity transactions through the issuance of convertible securities
which are convertible at the time of conversion at a discount to the prevailing market price, substantial dilution is likely to
occur resulting in a material decline in the price of your shares.
We may require additional financing in the future,
which may not be available or, if available, may be on terms that cause a decline in the value of the shares of our common stock
held by stockholders.
If we raise capital in the future by issuing additional securities,
our stockholders may experience a decline in the value of the shares of our common stock they currently hold or may acquire prior
to any such financing. In addition, such securities may have rights senior to the rights of holders of our shares of common stock.
If we fail to comply with the continuing listing
standards of The NASDAQ Capital Market, our securities could be delisted.
Our common stock and publicly traded warrants are listed on
The NASDAQ Capital Market under the symbols “APDN” and “APDNW,” respectively. For our common stock and
publicly traded warrants to continue to be listed on The NASDAQ Capital Market, we must meet the current continued listing requirements.
If we were unable to meet these requirements, our common stock and warrants could be delisted from The NASDAQ Capital Market. If
our securities were to be delisted from The NASDAQ Capital Market, our securities could begin to trade on the Over-The-Counter
Bulletin Board or on one of the markets operated by OTC Markets Group, including OTC Pink (formerly known as the “pink sheets”),
as the case may be. In such event, our securities could once again be subject to the “penny stock” rules which among
other things require brokers or dealers to approve investors’ accounts, receive written agreements and determine investor
suitability for transactions and disclose risks relating to investing in the penny stock market. Any such delisting of our securities
could have an adverse effect on the market price of, and the efficiency of the trading market for our securities, not only in terms
of the number of shares that can be bought and sold at a given price, but also through delays in the timing of transactions and
less coverage of us by securities analysts, if any. Also, if in the future we were to determine that we need to seek additional
equity capital, it could have an adverse effect on our ability to raise capital in the public or private equity markets.
Any material weaknesses in our internal control
over financing reporting in the future could adversely affect investor confidence, impair the value of our common stock and increase
our cost of raising capital.
Any failure to remedy deficiencies in our internal control over
financial reporting that may be discovered or our failure to implement new or improved controls, or difficulties encountered in
the implementation of such controls, could harm our operating results, cause us to fail to meet our reporting obligations or result
in material misstatements in our financial statements. Any such failure could, in turn, affect the future ability of our management
to certify that internal control over our financial reporting is effective. Inferior internal control over financial reporting
could also subject us to the scrutiny of the SEC and other regulatory bodies which could cause investors to lose confidence in
our reported financial information and could subject us to civil or criminal penalties or stockholder litigation, which could have
an adverse effect on our results of operations and the market price of our common stock.
In addition, if we or our independent registered public accounting
firm identify deficiencies in our internal control over financial reporting, the disclosure of that fact, even if quickly remedied,
could reduce the market’s confidence in our financial statements and harm our share price. Furthermore, deficiencies could
result in future non-compliance with Section 404 of the Sarbanes-Oxley Act of 2002. Such non-compliance could subject us to a variety
of administrative sanctions, including review by the SEC or other regulatory authorities.
Short sellers of our stock may be manipulative and
may drive down the market price of our common stock.
Short selling is the practice of selling securities that the
seller does not own but rather has borrowed or intends to borrow from a third party with the intention of buying identical securities
at a later date to return to the lender. A short seller hopes to profit from a decline in the value of the securities between the
sale of the borrowed securities and the purchase of the replacement shares, as the short seller expects to pay less in that purchase
than it received in the sale. As it is therefore in the short seller’s interest for the price of the stock to decline, some
short sellers publish, or arrange for the publication of, opinions or characterizations regarding the relevant issuer, its business
prospects and similar matters calculated to or which may create negative market momentum, which may permit them to obtain profits
for themselves as a result of selling the stock short. Issuers whose securities have historically had limited trading volumes
and/or have been susceptible to relatively high volatility levels can be particularly vulnerable to such short seller attacks.
The publication of any such commentary regarding us in the future may bring about a temporary, or possibly long term, decline
in the market price of our common stock. In the past, the publication of commentary regarding us by a disclosed short seller has
been associated with the selling of shares of our common stock in the market on a large scale, resulting in a precipitous decline
in the market price per share of our common stock. No assurances can be made that similar declines in the market price of our common
stock will not occur in the future, in connection with such commentary by short sellers or otherwise.
The price of our common stock may be volatile or
may decline, and the trading volume of our common stock may fluctuate, which may make it more difficult to realize a profit on
your investment in our shares of common stock.
Our common stock is listed on The NASDAQ Capital Market. The
trading price of our common stock has been and may continue to be volatile. In addition, the trading volume of our common stock
may fluctuate and cause significant price variations to occur. Volatility in the market price of our common stock may prevent you
from being able to sell your shares of common stock at or above the price you paid for your shares of common stock, which may make
it more difficult to realize a profit on your investment. A number of factors may affect the market price of our common stock,
including, but not limited to, the following:
|
|
·
|
our operating and financial performance and prospects;
|
|
|
·
|
our quarterly or annual earnings or those of other companies in our industry or that investors deem comparable to us;
|
|
|
·
|
conditions that impact demand for our products and services;
|
|
|
·
|
public reactions to our press releases, other public announcements and filings with the SEC;
|
|
|
·
|
market and industry perception of our success, or lack thereof, in pursuing our growth strategy;
|
|
|
·
|
strategic actions by us or our competitors, such as acquisitions or restructurings;
|
|
|
·
|
changes in accounting standards, policies, guidance, interpretations or principles;
|
|
|
·
|
arrival and departure of key personnel, including management personnel;
|
|
|
·
|
changes in our capital structure;
|
|
|
·
|
changes in the price of our warrants or other securities we may issue from time to time;
|
|
|
·
|
sales of common stock by us, our directors, officers or large stockholders;
|
|
|
·
|
the expiration of any applicable contractual lock-up agreements;
|
|
|
·
|
changes in general market, economic and political conditions in the United States and global economies or financial markets,
including those resulting from natural disasters, terrorist attacks, acts of war and responses to such events;
|
|
|
·
|
announcements of new products or innovations by us or our competitors and announcements concerning our competitors or our industry
in general;
|
|
|
·
|
difficulties in commercialization and distribution of our products or lower than expected sales volume or revenues;
|
|
|
·
|
changes in our relationships with manufacturers, suppliers or collaborators, or our inability to supply enough product to meet
demand;
|
|
|
·
|
our ability to obtain additional funding;
|
|
|
·
|
changes or developments in applicable laws or regulations;
|
|
|
·
|
any intellectual property infringement actions or other litigation or legal proceeding in which we may become involved;
|
|
|
·
|
changes in financial estimates or recommendations by securities analysts, or their ceasing to publish research or reports about
our business;
|
|
|
·
|
the trading volume of our common stock; and
|
|
|
·
|
the appeal and current level of investor interest in the biotechnology/biopharmaceutical capital market sector and in companies
in general with business, research strategies and product development pipelines which are similar to us.
|
In addition, The NASDAQ Capital Market and other securities
markets have, from time to time, experienced extreme price and trading volume fluctuations. The market prices of securities of
biotechnology and other life sciences companies in a comparable stage to ours historically have been particularly volatile, and
trading volume in such securities and our common stock has often been relatively low. Moreover, the securities and financial markets
in general have experienced substantial volatility that has often been unrelated or disproportionate to the operating results of
any individual company. During certain periods, specific industry sectors, such as the biotechnology segment, may experience greater
volatility than other sectors or the securities markets as a whole. These broad market fluctuations, during which our industry
and companies at our stage may experience a stronger degree of market sensitivity, will adversely affect the market price of our
common stock.
In the past, following periods of volatility in the market price
of a company’s securities, stockholders have often instituted class action securities litigation against those companies.
Such litigation, if instituted, could result in substantial costs and diversion of management attention and resources, which could
significantly harm our reputation and materially adversely affect our business, financial condition and results of operations.
Risks Relating to this Offering:
Our management has broad discretion as to the use
of the net proceeds from this offering.
We cannot specify with certainty the particular
uses of the net proceeds we will receive from this offering, and these uses may vary from our current plans. Our management will
have broad discretion in the application of the net proceeds, including for any of the purposes described in “Use of Proceeds.”
Accordingly, you will have to rely upon the judgment of our management with respect to the use of the proceeds. Our management
may spend a portion or all of the net proceeds from this offering in ways that holders of our common stock may not desire or that
may not yield a significant return or any return at all. The failure by our management to apply these funds effectively could harm
our business. Pending their use, we may also invest the net proceeds from this offering in a manner that does not produce income
or that loses value.
You will experience immediate and substantial dilution
in the net tangible book value per share of the common stock you purchase.
Since the price per share of our common
stock being offered is substantially higher than the net tangible book value per share of our common stock, you will suffer substantial
dilution in the net tangible book value of the common stock you purchase in this offering. Based on a public offering price of
$1.75 per share and Purchase Warrant , if you purchase shares of common stock in this offering, you will suffer immediate
and substantial dilution of approximately $1.47 per share in the net tangible book value of the common stock. See the section
entitled “Dilution” in this prospectus supplement for a more detailed discussion of the dilution you will incur if
you purchase common stock in this offering.
Holders of Purchase Warrants purchased in this offering
will have no rights as common stockholders until such holders acquire our common stock upon
exercise of their Purchase Warrants.
Until holders of Purchase Warrants acquire shares of our
common stock upon exercise of the Purchase Warrants, holders of Purchase Warrants will have no rights with respect to the shares
of our common stock underlying such Purchase Warrants. Upon exercise of the Purchase Warrants, the holders will be entitled to
exercise the rights of a common stockholder only as to matters for which the record date occurs after the exercise date.
The exercise price of the Purchase Warrants offered by
this prospectus may not be adjusted for certain dilutive events.
The exercise price of the Purchase Warrants
offered by this prospectus is subject to adjustment for certain events, including, but not limited to, certain issuances and/or
distributions of capital stock, options, convertible securities and other securities. In certain situations, the adjustment of
the exercise price is subject to a floor of $0.44 per share. The exercise prices will not be adjusted for dilutive issuances
of securities considered “exempt securities” and there may be transactions or occurrences that may adversely
affect the market price of our common stock or the market value of such Purchase Warrants without resulting in an adjustment
of the exercise prices of such Purchase Warrants.
Provisions of the Purchase Warrants offered by this prospectus
could discourage an acquisition of us by a third party.
In addition to the discussion of the provisions
of our Certificate of Incorporation, as amended, certain provisions of the Purchase Warrants offered by this prospectus could
make it more difficult or expensive for a third party to acquire us. Such Purchase Warrants prohibit us from engaging in certain
transactions constituting “fundamental transactions” unless, among other things, the surviving entity assumes our
obligations under the Purchase Warrants. Further, the Purchase Warrants provide that, in the event of certain transactions
constituting “fundamental transactions,” with some exception, holders of such Purchase Warrants will have the right,
at their option, to require us to repurchase such Purchase Warrants at a price described in the Purchase Warrants. These
and other provisions of the Purchase Warrants offered by this prospectus could prevent or deter a third party from acquiring us
even where the acquisition could be beneficial to you.
USE
OF PROCEEDS
We estimate that the net proceeds from
the sale of the shares of common stock and Purchase Warrants we are offering will be approximately $4.2 million, not including
any amounts we receive upon exercise of the Purchase Warrants. Net proceeds are what we expect to receive after paying the placement
agent’s fees and other expenses of the offering.
We intend to use the net proceeds received
from this offering for working capital, capital expenditures, business development and research and development expenditures and
acquisitions of new technologies or businesses. The actual allocation of proceeds realized from this offering will depend upon
our operating revenues and cash position and our working capital requirements.
Therefore, as of the date of this prospectus
supplement, we cannot specify with certainty all of the particular uses for the net proceeds to be received upon the completion
of this offering. Accordingly, we will have broad discretion in the application of the net proceeds, and investors will be relying
on our judgment regarding the application of the proceeds of this offering.
Pending our use of the net proceeds from
this offering, we intend to invest the net proceeds in a variety of capital preservation investments, including short-term, investment-grade,
interest-bearing instruments and U.S. government securities.
We have never declared or paid any cash
dividends on our common stock. We do not anticipate paying any cash dividends to stockholders in the foreseeable future. In addition,
any future determination to pay cash dividends will be at the discretion of our board of directors and will be dependent upon our
financial condition, results of operations, capital requirements, and such other factors as our board of directors deems relevant
at such time.
DILUTION
If you purchase securities in this offering,
your interest will be immediately and substantially diluted to the extent of the difference between the public offering price per
share of our common stock in this offering and the as adjusted net tangible book value per share of our common stock after giving
effect to this offering.
Our net tangible book value as of June
30, 2017 was approximately $4,110,696, or approximately $0.15 per share of common stock. After giving effect to the sale of the
shares and Purchase Warrants in this offering at the public offering price of $1.75 per share and Purchase Warrant, and
after deducting the placement agent’s fees and other estimated offering expenses payable by us, and, excluding the proceeds
attributable from the exercise of the Purchase Warrants, our pro forma as adjusted net tangible book value at June 30, 2017
would have been approximately $8.3 million or $0.28 per share. This represents an immediate increase in net tangible book value
of approximately $0.13 per share to our existing stockholders, and an immediate dilution of $1.47 per share to investors purchasing
shares in the offering.
Dilution in net tangible book value per
share represents the difference between the amount per share paid by purchasers of our common stock in this offering and the net
tangible book value per share of our common stock immediately after this offering.
The following table illustrates the per
share dilution to investors purchasing securities in the offering:
|
Public offering price per
share and Purchase Warrant
|
|
|
|
|
$
|
1.75
|
|
Net tangible book value per share as of June 30, 2017
|
|
|
|
|
$
|
0.15
|
|
Increase in net tangible book value per share attributable to this offering
|
|
|
|
|
$
|
0.13
|
|
Adjusted net tangible book value per share
after this offering
|
|
|
|
|
$
|
0.28
|
|
Amount of dilution in net tangible book value per share to new investors in this offering
|
|
|
|
|
$
|
1.47
|
The discussion and tables above are based
on 27,377,057 shares of our common stock outstanding as of June 30, 2017, which excludes 5,276,222 shares of common stock issuable
upon exercise of outstanding options and 9,548,969 shares of common stock issuable upon exercise of outstanding warrants as of
such date. This amount also excludes 2,735,000 shares of common stock issuable upon the exercise of the Purchase Warrants
issued in the offering. To the extent that options or warrants outstanding as of June 30, 2017 have been or may be exercised,
investors purchasing our common stock in this offering may experience further dilution. In addition, we may choose to raise additional
capital due to market conditions or strategic considerations even if we believe we have sufficient funds for our current or future
operating plans. To the extent that we raise additional capital through the sale of equity or convertible debt securities, the
issuance of these securities could result in further dilution to our stockholders.
CAPITALIZATION
The following table sets forth our cash
and cash equivalents and capitalization, as of June 30, 2017:
|
|
·
|
on an actual basis; and
|
|
|
·
|
on a pro forma
basis, based on a public offering price of $1.75 per share of common stock and Purchase
Warrant, to give effect to the sale of 5,470,000 shares of common stock, after deducting
the estimated placement agent’s fee and estimated offering expenses payable by
us.
|
You should consider this table in conjunction
with “Use of Proceeds” above as well as our “Management’s Discussion and Analysis of Financial Condition
and Results of Operations” and our financial statements and the notes to those financial statements incorporated by reference
in this prospectus supplement.
|
|
|
As of June 30, 2017
|
|
|
|
|
Unaudited,
Actual
|
|
|
Unaudited,
Pro forma
|
|
|
Cash and cash equivalents
|
|
$
|
2,402,809
|
|
|
$
|
6,625,805
|
|
|
Stockholders’ Equity:
|
|
|
|
|
|
|
|
|
|
Preferred stock, par value $0.001 per share; 10,000,000 shares authorized; -0- shares issued and outstanding as of June
30, 2017
|
|
|
|
|
|
|
|
|
|
Series A Preferred stock, par value $0.001 per share; 10,000,000 shares authorized; -0- shares outstanding as of June 30, 2017
|
|
|
|
|
|
|
|
|
|
Series B Preferred stock, par value $0.001 per share; 10,000,000 shares authorized; -0- shares outstanding as of June 30, 2017
|
|
|
|
|
|
|
|
|
|
Common stock, $0.001 par value per share; 500,000,000 shares authorized; 27,377,057 shares issued and outstanding as of June 30, 2017
|
|
|
27,377
|
|
|
|
30,112
|
|
|
|
Additional paid-in capital
|
|
|
242,808,854
|
|
|
|
247,029,115
|
|
|
|
Stock subscription receivable
|
|
|
(1,000,000
|
)
|
|
|
(1,000,000
|
)
|
|
|
Accumulated deficit
|
|
|
(233,818,477
|
)
|
|
|
(233,818,477
|
)
|
|
|
Total Stockholders’ Equity
|
|
$
|
8,017,754
|
|
|
$
|
12,240,750
|
|
|
DESCRIPTION
OF SECURITIES WE ARE OFFERING
In this offering, we are offering a maximum
of 2,735,000 shares of common stock and Purchase Warrants to purchase 2,735,000 shares of common stock.
As of December 18, 2017, our authorized
capital stock consists of 500,000,000 shares of common stock, par value $0.001 per share, of which 27,377,057 shares were issued
and outstanding, and 10,000,000 shares of preferred stock, par value $0.001 per share, of which no shares were issued and outstanding.
In addition, as of December 18, 2017, there were issued and outstanding options to purchase 5,333,227 shares of common stock and
warrants to purchase 9,540,455 shares of our common stock. The authorized and unissued shares of common stock and preferred stock
are available for issuance without further action by our stockholders, unless such action is required by applicable law or the
rules of any stock exchange on which our securities may be listed. Unless approval of our stockholders is so required, our board
of directors will not seek stockholder approval for the issuance and sale of our common stock.
Common Stock
Each shareholder of our common stock is
entitled to one vote for each share issued and outstanding held on all matters to be voted upon by the shareholders. Our shares
of common stock have no preemptive, conversion, or redemption rights. Upon the sale of substantially all of our stock or assets
or dissolution, liquidation or winding up, and after all liquidation preferences payable to any series of preferred stock entitled
thereto have been satisfied, our remaining assets shall be distributed to all holders of common stock and any similarly situated
stockholders who are not entitled to any liquidation preference or, if there be an insufficient amount to pay all such stockholders,
then ratably among such holders. All of our issued and outstanding shares of common stock are fully paid and non-assessable. Our
Certificate of Incorporation, as amended (the “Certificate of Incorporation”), does not provide for cumulative voting in
the election of directors. The holders of shares of our common stock will be entitled to such cash dividends as may be declared
from time to time by our board of directors from funds available therefor.
Our common stock is listed on The NASDAQ
Capital Market under the symbol “APDN.” The transfer agent and registrar for our common stock is American Stock Transfer
& Trust Company.
Warrants
We are selling to investors in this offering
Purchase Warrants to purchase one share of our common stock for each share of common stock purchased in this offering at a combined
purchase price of $1.75.
Each Purchase Warrant will be exercisable
beginning on the Initial Exercise Date, which is the date of closing, at an exercise price of $2.00 per share, subject to adjustment.
The Purchase Warrants will be exercisable for five years from the Initial Exercise Date, but not thereafter. The Purchase Warrants
include an adjustment provision that, subject to certain exceptions, reduces their exercise price if the Company issues Common
Stock or Common Stock equivalents at a price lower than the then-current exercise price of the Purchase Warrants, subject to a
minimum exercise price of $0.44 per share.
The Company has agreed not to effect any
issuance of Common Stock or securities convertible into Common Stock involving a Variable Rate Transaction, as defined in the
Securities Purchase Agreement, while any Purchaser holds any Purchase Warrants. The Purchase Warrants are subject to a call provision
whereby the Company may, subject to certain provisions including that the volume weighted average price of the Company’s
Common Stock has exceeded $5.00 for twenty consecutive trading days, call for cancellation of all or any portion of the Purchase
Warrants not yet exercised. Subject to limited exceptions, a holder of Purchase Warrants will not have the right to exercise any
portion of its Purchase Warrants if the holder, together with its affiliates, would beneficially own in excess of 4.99% (or, at
the election of the holder, 9.99%) of the number of shares of our common stock outstanding immediately after giving effect to
such exercise, or the Beneficial Ownership Limitation; provided, however, that upon 61 days’ prior notice to the Company,
the holder may increase or decrease the Beneficial Ownership Limitation, provided that in no event shall the Beneficial Ownership
Limitation exceed 9.99%.
The Purchase Warrants contain a “cashless
exercise” feature that allows holders to exercise the warrants without a cash payment to the Company upon the terms set
forth in the Purchase Warrants, if at the time of exercise there is no effective registration statement registering, or the prospectus
contained therein is not available for the issuance of the shares to the exercising Purchase Warrant holder.
In the case of certain fundamental transactions
affecting the Company, a holder of Purchase Warrants, upon exercise of such Purchase Warrants after such fundamental transaction,
will have the right to receive, in lieu of shares of the Company’s common stock, the same amount and kind of securities,
cash or property that such holder would have been entitled to receive upon the occurrence of the fundamental transaction, had
the Purchase Warrants been exercised immediately prior to such fundamental transaction. In lieu of such consideration, a holder
of Purchase Warrants may instead elect to receive a cash payment based upon the Black-Scholes value of their Purchase Warrants.
The exercise price and number of the shares
of our common stock issuable upon the exercise of the Purchase Warrants will be subject to adjustment in the event of any stock
dividends and splits, reverse stock split, recapitalization, reorganization or similar transaction, as described in the Purchase
Warrants.
After the Initial Exercise Date, if and
only if no effective registration statement registering, or no current prospectus is available for, the resale of the Purchase
Warrants, the purchasers may exercise the Purchase Warrants by means of a “cashless exercise.”
Anti-takeover Effects of Certain Provisions of our Certificate
of Incorporation and Bylaws
Our Certificate of Incorporation
contains provisions that could make it more difficult to acquire control of our company by means of a tender offer, open market
purchases, a proxy contest or otherwise. A description of these provisions is set forth below.
Preferred Stock
We believe that the availability of the
preferred stock under our Certificate of Incorporation provides us with flexibility in addressing corporate issues
that may arise. Having these authorized shares available for issuance allows us to issue shares of preferred stock without the
expense and delay of a special stockholders’ meeting. The authorized shares of preferred stock, as well as shares of common
stock, will be available for issuance without further action by our stockholders, unless action is required by applicable law
or the rules of any stock exchange on which our securities may be listed. The board of directors has the power, subject to applicable
law, to issue series of preferred stock that could, depending on the terms of the series, impede the completion of a merger, tender
offer or other takeover attempt that some, or a majority, of the stockholders might believe to be in their best interests or in
which stockholders might receive a premium for their stock over the then prevailing market price of the stock.
Advance Notice Procedure
Our bylaws provide an advance notice procedure
for stockholders to nominate director candidates for election or to bring business before an annual meeting of stockholders. Only
persons nominated by, or at the direction of, our board of directors or by a stockholder who has given proper and timely notice
to our secretary prior to the meeting, will be eligible for election as a director. In addition, any proposed business other than
the nomination of persons for election to our board of directors must constitute a proper matter for stockholder action pursuant
to the notice of meeting delivered to us. For notice to be timely, it must be received by our secretary not less than 90 nor more
than 120 calendar days prior to the first anniversary of the previous year’s annual meeting (or if the date of the annual
meeting is advanced more than 30 calendar days or delayed by more than 60 calendar days from the anniversary date of the previous
year’s annual meeting, not earlier than the 90th calendar day prior to such meeting or the 10th calendar day after public
disclosure of the date of such meeting is first made). These advance notice provisions may have the effect of precluding the conduct
of certain business at a meeting if the proper procedures are not followed or may discourage or deter a potential acquirer from
conducting a solicitation of proxies to elect its own slate of directors or otherwise attempt to obtain control of us.
Special Meetings of Stockholders
Our bylaws provide that special meetings
of stockholders may be called only by the Chairman of the Board, the Chief Executive Officer, or the Board of Directors pursuant
to a resolution adopted by a majority of the Board.
Anti-Takeover Effects of Delaware Law
Section 203 of the Delaware General Corporation
Law (DGCL) provides that, subject to exceptions specified therein, an “interested stockholder” of a Delaware corporation
shall not engage in any “business combination,” including general mergers or consolidations or acquisitions of additional
shares of the corporation, with the corporation for a three-year period following the time that such stockholder becomes an interested
stockholder unless:
|
|
·
|
prior to such time, the board of directors of the corporation approved either the business combination or the transaction which
resulted in the stockholder becoming an interested stockholder;
|
|
|
·
|
upon consummation of the transaction which resulted in the stockholder becoming an “interested stockholder,” the
interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced
(excluding specified shares); or
|
|
|
·
|
on or subsequent to such time, the business combination is approved by the board of directors of the corporation and authorized
at an annual or special meeting of stockholders, and not by written consent, by the affirmative vote of at least 66 2/3% of the
outstanding voting stock not owned by the interested stockholder
|
Under Section 203, the restrictions described
above also do not apply to specified business combinations proposed by an interested stockholder following the announcement or
notification of one of specified transactions involving the corporation and a person who had not been an interested stockholder
during the previous three years or who became an interested stockholder with the approval of a majority of the corporation’s
directors, if such transaction is approved or not opposed by a majority of the directors who were directors prior to any person
becoming an interested stockholder during the previous three years or were recommended for election or elected to succeed such
directors by a majority of such directors. The restrictions described above also do not apply to specified business combinations
with a person who is an “interested stockholder” prior to the time when the corporation’s common stock is listed
on a national securities exchange, so these restrictions would not apply to a business combination with any person who is one of
our stockholders prior to this offering.
Except as otherwise specified in Section
203, an “interested stockholder” is defined to include:
|
|
·
|
any person that is the owner of 15% or more of the outstanding voting stock of the corporation, or is an affiliate or associate
of the corporation and was the owner of 15% or more of the outstanding voting stock of the corporation at any time within three
years immediately prior to the date of determination; and
|
|
|
·
|
the affiliates and associates of any such person
|
Under some circumstances, Section 203 makes
it more difficult for a person who is an interested stockholder to effect various business combinations with us for a three-year
period.
Listing
Our shares of common stock offered hereby
are listed on The NASDAQ Capital Market under the symbol “APDN.”
Transfer Agent and Registrar
American Stock Transfer & Trust Company,
located in Brooklyn, New York, is the transfer agent and registrar for our common stock.
MARKET
PRICE FOR OUR SECURITIES
Our common stock is listed on The NASDAQ
Capital Market under the symbol “APDN.” There is no certainty that the common stock will continue to be listed or that
any liquidity exists for our stockholders.
The following table sets forth the quarterly
quotes of high and low prices for our common stock on The NASDAQ Capital Market during the fiscal years ended September 30, 2018,
2017 and 2016.
|
|
|
Fiscal 2018
|
|
|
Fiscal 2017
|
|
|
Fiscal 2016
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
High
|
|
|
Low
|
|
|
High
|
|
|
Low
|
|
|
High
|
|
|
Low
|
|
|
Common Stock:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
First Quarter (Through December 18, 2017 for Fiscal 2018)
|
|
$
|
3.45
|
|
|
$
|
2.00
|
|
|
$
|
3.15
|
|
|
$
|
1.73
|
|
|
$
|
9.70
|
|
|
$
|
2.74
|
|
|
Second Quarter
|
|
|
|
|
|
|
|
|
|
$
|
2.05
|
|
|
$
|
1.40
|
|
|
$
|
3.74
|
|
|
$
|
2.46
|
|
|
Third Quarter
|
|
|
|
|
|
|
|
|
|
$
|
1.94
|
|
|
$
|
0.90
|
|
|
$
|
3.50
|
|
|
$
|
2.35
|
|
|
Fourth Quarter
|
|
|
|
|
|
|
|
|
|
$
|
2.88
|
|
|
$
|
1.55
|
|
|
$
|
3.61
|
|
|
$
|
2.70
|
|
INDEMNIFICATION
FOR SECURITIES ACT LIABILITIES
Our Certificate of Incorporation provides
to the fullest extent permitted by Delaware law that our directors or officers shall not be personally liable to us or our stockholders
for damages for breach of such director’s or officer’s fiduciary duty. The effect of this provision of our Certificate
of Incorporation is to eliminate our rights and our stockholders (through stockholders’ derivative suits on behalf of our
company) to recover damages against a director or officer for breach of the fiduciary duty of care as a director or officer (including
breaches resulting from negligent or grossly negligent behavior), except under certain situations defined by statute. We believe
that the indemnification provisions in our Certificate of Incorporation are necessary to attract and retain qualified persons as
directors and officers.
We have entered into an indemnification
agreement (each, an “Indemnification Agreement”) with each of our directors and executive officers. In general, the
Indemnification Agreement obligates us to indemnify a director or executive officer, to the fullest extent permitted by applicable
law, for certain expenses, including attorneys’ fees, judgments, penalties, fines and settlement amounts actually and reasonably
incurred by them in any action or proceeding arising out of their services as one of our directors or officers, or any of our subsidiaries
or any other company or enterprise to which the person provides services at our request. In addition, the Indemnification Agreement
provides for the advancement of expenses incurred by the indemnitee in connection with any covered proceeding to the fullest extent
permitted by applicable law. The rights provided by the Indemnification Agreement are in addition to any other rights to indemnification
or advancement of expenses to which the indemnitee may be entitled under applicable law, the Company’s Certificate of Incorporation
or bylaws, or otherwise.
Insofar as indemnification for liabilities
arising under the Securities Act, may be permitted to directors, officers or persons controlling us pursuant to the foregoing provisions,
or otherwise, we have been advised that in the opinion of the SEC, such indemnification is against public policy as expressed in
the Securities Act and is, therefore, unenforceable.
PLAN
OF DISTRIBUTION
Maxim Group LLC has agreed to act as our
sole placement agent in connection with this offering subject to the terms and conditions of a placement agency agreement, dated
December 20, 2017 between Maxim Group LLC and us. The placement agent is not purchasing or selling any securities offered
by this prospectus supplement and the accompanying base prospectus, but has arranged for the sale of certain of the shares and
Purchase Warrants offered hereby through a securities purchase agreement entered into between the purchasers and us. The public
offering price of the shares and Purchase Warrants offered by this prospectus supplement and the accompanying base prospectus
has been determined based upon arm’s-length negotiations between the purchasers and us.
We have entered into a securities purchase
agreement directly with the investors in this offering. A form of the securities purchase agreement was included as Exhibit 10.2
to our Current Report on Form 8-K filed with the SEC in connection with this offering. The securities purchase agreement provides
such investors with certain representations, warranties and covenants, including indemnifications, from us. Our obligation to
issue and sell the shares of common stock and the Purchase Warrants to the investors who are party to the securities purchase
agreement is subject to the closing conditions set forth in the securities purchase agreement, including the absence of
any material adverse change in our business and the receipt of certain opinions, letters and certificates from us or our counsel,
which may be waived by the respective parties. All of the securities will be sold at the prices specified in this prospectus supplement
and, we expect, at a single closing.
Commissions and Expenses
We have agreed to pay the placement agent
an aggregate cash placement fee equal to 6.5% of the gross proceeds in this offering from sales arranged for by the placement agent.
Subject to certain conditions, we also have agreed to reimburse all reasonable travel and other out-of-pocket expenses of the placement
agent in connection with this offering, including but not limited to the reasonable fees of legal counsel, not to exceed $45,000.
We currently anticipate that the delivery
of the shares will occur on or about December 22, 2017. At the closing, The Depository Trust Company will credit the shares of
common stock to the respective accounts of the purchasers or the transfer agent will issue the shares to the purchasers in book-entry
form, as elected by the purchasers in their respective purchase agreements. The Purchase Warrants will be delivered to the
investors by physical delivery following the closing.
Other Terms
Under the securities purchase agreement,
we have agreed not to enter into any agreement to issue or announce the issuance or proposed issuance of any common stock or common
stock equivalents for a period of 90 days following the closing of the offering.
If we elect to terminate this offering for
any reason even though the placement agent was prepared to proceed with the placement, if within six months following the termination,
we complete any financing of equity, equity-linked or other capital raising activity, then we will be required to pay to the placement
agent upon the closing of the financing the cash placement fee described above.
Lock-Up Agreements
In connection with this offering, each of
our executive officers, directors and certain stockholders has agreed, subject to certain exceptions set forth in the lock-up agreements,
not to sell, offer, agree to sell, contract to sell, hypothecate, pledge, grant any option to purchase, make any short sale of,
or otherwise dispose of, directly or indirectly, any shares of our common stock, or any securities convertible into or exercisable
or exchangeable for shares of our common stock, for 90 days from the date of the final prospectus supplement relating to this offering
without the prior written consent of the placement agent. Notwithstanding the foregoing, if (a) we issue an earnings release or
material news, or a material event relating to our company occurs, during the last 17 days of the lock-up period, or (b) prior
to the expiration of the lock-up period, we announce that we will release earnings results during the 16-day period beginning on
the last day of the lock-up period, the restrictions described above shall continue to apply until the expiration of the 18-day
period beginning on the issuance of the earnings releases or the occurrence of the material news or material event, unless the
placement agent waives that extension.
Indemnification
We have agreed to indemnify the placement
agent against certain liabilities, including liabilities under the Securities Act, and liabilities arising from breaches of representations
and warranties contained in the placement agency agreement, or to contribute to payments that the placement agent may be
required to make in respect of those liabilities.
Other Relationships
The placement agent and its affiliates
have engaged, and may in the future engage, in investment banking transactions and other commercial dealings in the ordinary course
of business with us or our affiliates. The placement agent has received, or may in the future receive, customary fees and commissions
for these transactions. As of the date hereof, an affiliate of the placement agent holds warrants to purchase 38,093 shares and
51,137 shares of our common stock with an exercise price of $4.0135 per share and $2.53 per share, respectively.
Electronic Distribution
This prospectus supplement and the accompanying
base prospectus may be made available in electronic format on websites or through other online services maintained by the placement
agent or by an affiliate. Other than this prospectus supplement and the accompanying base prospectus, the information on the placement
agent’s website and any information contained in any other website maintained by the placement agent is not part of this
prospectus supplement and the accompanying base prospectus or the registration statement of which this prospectus supplement and
the accompanying base prospectus forms a part, has not been approved and/or endorsed by us or the placement agent, and should not
be relied upon by investors.
The foregoing does not purport to be
a complete statement of the terms and conditions of the placement agency agreement and subscription agreements. A copy of the
placement agent agreement and the form of securities purchase agreement with the purchasers are included as exhibits to our
Current Report on Form 8-K filed with the SEC in connection with this offering and incorporated by reference into the
Registration Statement of which this prospectus supplement forms a part. See “Where You Can Find More
Information.”
EXPENSES
The following are the estimated expenses
of the issuance and distribution of our shares of common stock and Purchase Warrants in this offering, other than the placement
agent’s fees, all of which will be paid by us.
|
SEC registration fee*
|
|
$
|
2,898
|
|
|
FINRA filing fee*
|
|
$
|
4,250
|
|
|
Legal fees and expenses
|
|
$
|
195,000
|
|
|
Accounting fees and expenses
|
|
$
|
25,000
|
|
|
Miscellaneous
|
|
$
|
25,000
|
|
|
Total
|
|
$
|
252,148
|
|
*The SEC registration fee of $2,898 and
the FINRA filing fee of $4,250 covering all of the securities being offered under the registration statement on Form S-3 (File
No. 333-218158) filed with the SEC with an effective date of May 22, 2017, of which this prospectus supplement forms part, was
previously paid. We allocate the cost of these fees on an approximate pro-rata basis with each offering pursuant to such registration
statement.
LEGAL
MATTERS
The validity of the issuance of the securities
offered hereby will be passed upon for us by Pepper Hamilton LLP, New York, New York. Harter Secrest & Emery LLP, Rochester,
New York, is acting as counsel for the placement agent in this offering.
EXPERTS
Marcum LLP, independent registered public
accounting firm, has audited our consolidated financial statements included in our Annual Report on Form 10-K for the year ended
September 30, 2016, as set forth in their report, which is incorporated by reference in the prospectus and elsewhere in this registration
statement. Our consolidated financial statements are incorporated by reference in reliance on Marcum LLP’s report, given
on their authority as experts in accounting and auditing.

2,735,000 SHARES OF
COMMON STOCK
AND
WARRANTS TO PURCHASE 2,735,000 SHARES
OF COMMON STOCK
PROSPECTUS
Sole Placement Agent
Maxim Group LLC
December 21 , 2017
PROSPECTUS
$25,000,000

Common Stock
Preferred Stock
Debt Securities
Warrants
Rights
Units
We may offer and sell, from time to time in one or more offerings,
up to $25,000,000 of our common stock, preferred stock, debt securities, warrants and rights, or any combination of these securities,
and/or units consisting of one or more of these securities. We may also offer common stock or preferred stock upon conversion of
debt securities and common stock upon conversion of preferred stock. All of the securities listed above may be sold separately
or as units with other securities.
This prospectus describes some of the general terms that may
apply to these securities. When we decide to sell a particular class or series of securities, we will provide specific terms of
the offered securities in one or more prospectus supplements. We may also authorize one or more free writing prospectuses to be
provided to you in connection with these offerings.
The prospectus supplement, and any documents incorporated by
reference, may also add, update or change information contained in or incorporated by reference into this prospectus. However,
no prospectus supplement shall offer a security that is not registered and described in this prospectus at the time of its effectiveness.
You should read this prospectus and any prospectus supplement, as well as the documents incorporated by reference or deemed to
be incorporated by reference into this prospectus, and any free writing prospectus carefully before you invest. This prospectus
may not be used to offer or sell our securities unless accompanied by a prospectus supplement relating to the offered securities.
Our common stock and our warrants are listed on The NASDAQ Capital
Market under the symbols “APDN” and “APDNW,” respectively. Each prospectus supplement will contain information,
where applicable, as to our listing on any securities exchange of the securities covered by the prospectus supplement. The aggregate
market value of our outstanding common stock held by non-affiliates was approximately $33,323,045 based on 26,351,483 shares of
outstanding common stock, of which 4,852,744 shares are held by affiliates, and a price of $1.55 per share, which was the last
reported sale price of our common stock as quoted on The NASDAQ Capital Market on May 15, 2017. Pursuant to General Instruction
I.B.6 of Form S-3, in no event will we sell securities registered in a public primary offering with a value exceeding more than
one-third of our public float (the market value of our common stock held by our non-affiliates) in any 12 calendar month period
so long as our public float remains below $75.0 million. We have not offered any securities pursuant to General Instruction I.B.6
of Form S-3 during the prior 12 calendar month period that ends on and includes the date of this prospectus. As of May 15, 2017,
one-third of our public float is equal to approximately $11,107,682.
These securities may be sold by us directly to purchasers, through
dealers or agents, or to or through underwriters, or through a combination of these methods. See “Plan of Distribution”
in this prospectus. We may also describe the plan of distribution for any particular offering of our securities in a prospectus
supplement. If any agents, underwriters or dealers are involved in the sale of any securities in respect of which this prospectus
is being delivered, we will disclose their names and the nature of our arrangements with them in a prospectus supplement. The net
proceeds we expect to receive from any such sale will also be included in a prospectus supplement.
An investment in our securities involves a high degree of
risk. See the sections entitled “Risk Factors” in our most recent Annual Report on Form 10-K and in any Quarterly Report
on Form 10-Q, as well as in any prospectus supplement or free writing prospectus related to these specific offerings.
We may amend or supplement this prospectus from time to time
by filing amendments or supplements as required or related free writing prospectuses. You should read the entire prospectus and
any amendments or supplements carefully before you make your investment decision.
Neither the Securities and Exchange Commission nor any state
securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete.
Any representation to the contrary is a criminal offense.
The date of this prospectus is May 22, 2017
ABOUT
THIS PROSPECTUS
This prospectus is part of a Registration Statement that we
filed with the Securities and Exchange Commission (“
SEC
”) using a “shelf” registration process.
Under this shelf registration process, we may offer from time to time securities described in this prospectus having a maximum
aggregate offering price of $25,000,000 in one or more offerings. Each time we offer securities, we will prepare and file with
the SEC a prospectus supplement or information that is incorporated by reference into this prospectus that describes the specific
amounts, prices and terms of the securities we offer. We may also authorize one or more free writing prospectuses to be provided
to you that may contain material information relating to these offerings and securities. The prospectus supplement also may add,
update or change information contained in this prospectus or the documents incorporated herein by reference. You should read carefully
this prospectus, any applicable prospectus supplement and any related free writing prospectus together with additional information
described below under the caption “Where You Can Find More Information.”
This prospectus does not contain all the information provided
in the Registration Statement we filed with the SEC. For further information about us or our securities offered hereby, you should
refer to that Registration Statement, which you can obtain from the SEC as described below under “Where You Can Find More
Information.”
You should rely only on the information contained or incorporated
by reference in this prospectus, any prospectus supplement and any related free writing prospectus. We have not authorized any
other person to provide you with different information. If anyone provides you with different or inconsistent information, you
should not rely on it. This prospectus is not an offer to sell securities, and it is not soliciting an offer to buy securities,
in any jurisdiction where the offer or sale is not permitted. You should assume that the information appearing in this prospectus,
any prospectus supplement, any related free writing prospectus as well as information we have previously filed with the SEC and
incorporated by reference, is accurate as of the date of those documents only. Our business, financial condition, results of operations
and prospects may have changed since those dates.
We may sell securities through underwriters or dealers, through
agents, directly to purchasers or through any combination of these methods. We and our agents reserve the sole right to accept
or reject in whole or in part any proposed purchase of securities. The prospectus supplement, which we will prepare and file with
the SEC each time we offer securities, will set forth the names of any underwriters, agents or others involved in the sale of securities,
and any applicable fee, commission or discount arrangements with them. See “Plan of Distribution.”
In this prospectus, unless otherwise indicated, the “
Registrant
,”
“
our company
,” “
we
,” “
us
” or “
our
” refer to Applied
DNA Sciences, Inc., a Delaware corporation and its consolidated subsidiaries.
PROSPECTUS
SUMMARY
This prospectus summary highlights certain information
about our company and other information contained elsewhere in this prospectus or in documents incorporated by reference. This
summary does not contain all of the information that you should consider before making an investment decision. You should carefully
read the entire prospectus, any prospectus supplement, including the section entitled “Risk Factors” and the documents
incorporated by reference into this prospectus, before making an investment decision.
THE
OFFERING
This prospectus is part of a Registration Statement that we
filed with the SEC utilizing a shelf registration process. Under this shelf registration process, we may sell any combination of:
|
|
·
|
debt securities, in one or more series;
|
|
|
·
|
warrants to purchase any of the securities listed above;
|
|
|
·
|
rights to purchase common stock, preferred stock or warrants; and/or
|
|
|
·
|
units consisting of one or more of the foregoing
|
in one or more offerings up to a total dollar amount of $25,000,000.
This prospectus provides you with a general description of the securities we may offer. Each time we sell securities, we will provide
a prospectus supplement that will contain specific information about the terms of that specific offering and include a discussion
of any risk factors or other special considerations that apply to those securities. The prospectus supplement may also add, update
or change information contained in this prospectus. You should read both this prospectus and any prospectus supplement together
with the additional information described under the heading “Where You Can Find More Information.”
OUR
COMPANY
Overview
Using biotechnology as a forensic foundation, we create unique
security solutions addressing the challenges of modern commerce. Whether for supply chain security, brand protection or law enforcement
applications, it is our goal to help establish secure flourishing environments that foster quality, integrity and success. With
secure taggants, high-resolution DNA authentication, and comprehensive reporting, our SigNature DNA technologies are designed to
deliver what we believe to be the greatest levels of security, deterrence and legal recourse strength. We are also engaged in the
large-scale production of specific DNA sequences using the polymerase chain reaction (“
PCR
”).
SigNature DNA, the core of our technology platform, is nature’s
ultimate means of authentication and supply chain security. Our precision-engineered marks have not and, we believe, cannot be
broken. Additional layers of protection and complexity are added to the mark in a proprietary manner. SigNature DNA in various
carriers has proven highly resistant to UV radiation, heat, cold, vibration, abrasion and other extreme environments and conditions.
We work closely with our customers to develop a solution that will be optimized to their specifications to deliver maximum impact.
Our products and technology are protected by what we believe to be a robust portfolio of patents and trademarks.
Using our products and technology, manufacturers, brands, and
other stakeholders can ensure authenticity and protect against diversion throughout a product’s journey from manufacturer
to use.
The core technologies of our business allow us to use DNA sequences
to mark objects in a unique manner that we believe cannot be replicated, and then identify these objects by detecting the absence
or presence of the DNA. We believe that our disruptive platform offers broad commercial relevance across many industry verticals.
Our underlying strategy is to become a solutions provider in supply chains of process industries in which contracts are larger
and of longer duration, where the benefits to customers and consumers are more significant, and where our forensic security and
traceability offer a unique and protected value. Consumers, governments and companies are demanding details about the systems and
sources that deliver their goods. They worry about quality, safety, ethics, and the environmental impact. Farsighted organizations
are directly addressing new threats and opportunities presented by this question: Where do these goods come from? These are the
questions and concerns we are beginning to address for a growing number of companies. We supply key building blocks for creating
secure supply chains with traceability of goods, which in turn can help ensure integrity in supply, honest claims, and ethical
and sustainable sourcing.
Signature DNA Markers
SigNature DNA.
SigNature DNA is our patented
platform ingredient, at the core of all our security solutions. It provides forensic power and protection for a wide array of applications.
Highly secure, robust and durable, SigNature DNA markers are an ingredient that can be used to fortify brand protection efforts;
strengthen supply chain security; and mark, track and convict criminals. Custom DNA sequences can be embedded into a wide range
of host carriers including ink, varnish, thread, laminates and metal coatings. SigNature DNA markers are resistant to heat, cold,
vibration, abrasion, organic solvents, chemicals, UV radiation and other extreme environmental conditions, and so can be identified
for numerous years after being embedded directly, or into media applied or attached to the item to be marked. Each individual marker
is recorded and stored in a secure database so that we can later detect it using a simple spot test, or the marks can be forensically
analyzed to obtain definitive proof of the presence or absence of a specific type of SigNature DNA marker (e.g., one designed to
mark a particular product). Our in-lab forensic testing for authenticity results in an expert witness Certificate of DNA Authentication
(“
CODA
”). Because DNA is one of the most dense information carriers known, only minute quantities of SigNature
DNA are necessary for successful analysis and authentication. As a result, SigNature DNA can fold seamlessly into production and
logistics workflows.
SigNature DNA has been subjected to rigorous testing by the
Idaho National Laboratory, a U.S. National Laboratory, by CALCE (the Center for Advanced Life Cycle Engineering), the largest electronic
products and systems research center focused on electronics reliability, and by verified procedures in our laboratories. The forensic
marker has passed all tests across a broad spectrum of materials and has met key military stability standards. SigNature DNA has
passed a strenuous “red-team” vetting on behalf of the U.S. Defense Logistics Agency.
Hundreds of millions of SigNature DNA markers now exist on items
ranging from consumer product packaging to microcircuits to cotton and synthetic fibers; to our knowledge, none has ever been copied.
SigNature T DNA and fiberTyping
SigNature T DNA.
SigNature T DNA is a unique patent-pending
tagging and authentication system specifically designed for textiles and apparel. Specially engineered to adhere tenaciously to
any kind of textile substrate, including natural and synthetic fibers, SigNature T DNA markers are resistant to standard textile
production conditions, and cannot be copied. The result: an enduring forensic identity marker that remains present from the fiber
stage through to the finished product.
This technology allows for better quality control and assurance
at any point in the supply chain. SigNature T DNA markers are used for brand protection efforts and raw material, source compliance
programs. For example, cotton fibers can be tagged at source, verified as “American grown” and then traced through
every step of the supply chain.
Our patented cotton genotyping platform, known as “fiberTyping®,”
described below, complements tagging with SigNature T DNA. fiberTyping is employed to identify the genus and species of the fibers
before or after they are tagged with SigNature T DNA. fiberTyping cannot be used to track a specific cotton batch through the supply
chain, a function which can only be accomplished by our SigNature T DNA system.
fiberTyping
. fiberTyping is not a marker, but
a test of native cotton fiber, which gives a clear result that determines whether the intended “nature-made” endogenous
cotton DNA is present in your fiber, yarn or fabric. Samples from the primary material are sent to our forensic labs for DNA analysis
and authentication. Cotton classification and the authentication of cotton geographic origin are issues of global significance,
important to brand owners and to governments that must regulate the international cotton trade. The use of DNA to identify the
cotton fiber content of finished textiles, along with SigNature T DNA tagging is a significant opportunity for brand license holders
to control their intellectual property and for governments to improve their ability to enforce compliance with trade agreements
between nations.
In addition to the global cotton trade, the potential markets
for genotyping include biotherapeutics, nutraceuticals, natural foods, wines and fermented alcohols and other natural textiles.
We believe that our DNA extraction protocol and methodologies
are more effective than existing forensic systems. We believe that the combination of our SigNatureT DNA and fiberTyping solutions
cover the forensic authentication market for textiles and secure systems and that the related protocols we have developed may be
applicable to multiple industry verticals, and can mark and authenticate products at every stage of their life cycle, from beginning
to end.
DNAnet and Backtrac
Recognizing that DNA-based evidence is the cornerstone of modern-era
law enforcement, we have developed what we believe to be the ultimate crime fighting tools – currently being used in home
asset and vehicle marking, as well as commercial applications.
DNAnet.
DNAnet is a DNA marker that can be used
to definitively link evidence and offenders to specific crime scenes. As the crime is investigated, the fluorescing DNA marker
can assist police in linking the offender and stolen items to a specific crime scene, creating a greater ability to identify and
convict.
Backtrac.
Backtrac(TM) is a long lasting tagging
solution containing molecular signature that can help return stolen or lost property to its rightful owner.
Beacon
Beacon.
Beacon® locked optical markers deliver
secure real-time inspection capabilities. A unique encrypted mechanism (patent-pending) creates a protected, covert screening tool
that can be easily adapted to packaging, security labels and high–value assets through inks, varnishes and coatings. When
Beacon locked optical markers are combined with SigNature DNA markers, a strong and flexible end-to-end security solution is created
where authenticity and provenance can be determined with confidence.
SigNify
SigNify.
Developing a secure method for real-time,
in-field screening of DNA-marked items has long been a priority for us. We believe that standard fluorophores, up-converting phosphors,
holograms and other more-traditional screening tools provide little to no defense against counterfeiting. We believe that secure
in-field inspection backed with forensic-level DNA authentication is the key to maintaining a well-defended supply chain or asset
management program.
The SigNify IF portable DNA reader provides definitive real-time
authentication of SigNature DNA in the field– DNA becomes a true, front-line solution for supply chain integrity.
CertainT Supply Chain Platform
CertainT helps brands confirm their product’s authenticity
and origin with certified, trust, transparency and traceability. The CertainT emblem indicates use of our tagging, testing and
tracking platform to enable proof of product claims for any material, item or product. Secure and proven, the CertainT Platform
helps manufacturers, brands or other commercial organizations deliver on their promise that customers are buying products that
are ethically-sourced, safe and authentic.
Information Technology Systems
digitalDNA.
digitalDNA is a software platform
that enables customers to manage the security of company-marked goods from point of marking to point of authentication or validation
to end of life. The base platform is configurable to customer requirements which differ by vertical market, company business process
and IT environment. Basic functions offered include DNA inventory management, program training and communications, a database of
marked items information, associated documents and images, chain of custody and location tracking, sample authentication processing
and CODA downloads, and other administrative functions. Architected for either cloud or local operation, the system supports mobile
data capture using bar codes or other technologies. Of special note is the power of embedding our proprietary DNA into tag ink
or substrate as the forensic backstop for tags which can be easily copied. The system is architected as the controller and repository
for other validation and authentication devices such as our SigNify DNA Readers, Multi-Mode Reader (prototype), DNA Transfer Systems,
and other third party devices and is designed to share data with third party applications through standard interfaces.
DNA Transfer Systems.
Our DNA Transfer Systems
are developed for DNA marking applications which are high volume with a need for monitoring and control. They are computer based,
fully automated, offer remote internet access for real-time monitoring and can be configured for application-specific alerts and
reporting online. They were used to mark cotton at eight U.S. cotton gins in the 2016 ginning season.
Large-scale production of specific DNA sequences using
PCR.
Large-scale production of specific DNA sequences using
PCR.
Our patented Triathlon™ PCR systems allow for the large-scale production of specific DNA sequences. The systems
are self-contained and modular, can work together in mass production or can be used individually throughout the world, offering
the advantage of delivering DNA locally and securely. These DNA sequences are being used by customers as a diagnostic and reagent
and provide us the opportunity to cross-sell our DNA-based supply chain security solutions. A new capacity for us will be the ability
to manufacture longer DNA sequences valuable in gene therapy, DNA vaccines and diagnostics. These types of DNA are distinct from
our DNA security markers and represent a potential new entry into medical markets, where we believe there are opportunities for
our broader platform.
Corporate History
We are a Delaware corporation, which was initially formed in
1983 under the laws of the State of Florida as Datalink Systems, Inc. In 1998, we reincorporated in Nevada, and in 2002, we changed
our name to our current name, Applied DNA Sciences, Inc. In December 2008, we reincorporated from Nevada to the State of Delaware.
Our corporate headquarters are located at the Long Island High
Technology Incubator at Stony Brook University in Stony Brook, New York, where we established laboratories for the manufacture
of DNA markers and product prototypes, and DNA authentication. The address of our corporate headquarters is 50 Health Sciences
Drive, Stony Brook, New York 11790, and our telephone number is (631) 240-8800. We maintain a website at www.adnas.com where general
information about us is available. The information on, or that may be accessed through, our website is not incorporated by reference
into and should not be considered a part of this registration statement.
To date, we have had a limited operating history with our current
business model, and as a result, our operations have produced limited revenues.
RISK
FACTORS
Investing in our securities involves a high degree of risk.
Prior to making a decision about investing in our securities, you should carefully consider and evaluate the specific factors discussed
under the heading “Risk Factors” in our Annual Report on Form 10-K for the fiscal year ended September 30, 2016 filed
on December 6, 2016, with the SEC, and any updates described in subsequent Quarterly Reports on Form 10-Q, all of which are incorporated
herein by reference, and may be amended, supplemented or superseded from time to time by other reports we file with the SEC in
the future. The risks and uncertainties we have described are not the only risks that we face. Additional risks and uncertainties
not presently known to us or that we currently deem immaterial may also affect our operations. The occurrence of these known or
unknown risks might cause you to lose all or part of your investment.
See also the statements contained under the heading “Forward-Looking
Statements.”
FORWARD-LOOKING
STATEMENTS
This prospectus and the documents incorporated by reference
herein contain “forward-looking statements” within the meaning of Section 27A of the Securities Act and Section 21E
of the Securities Exchange Act of 1934, as amended (the “
Exchange Act
”), that are intended to qualify for the
“safe harbor” created by those sections. In addition, we may make forward-looking statements in other documents filed
with or furnished to the SEC, and our management and other representatives may make forward-looking statements orally or in writing
to analysts, investors, representatives of the media and others.
Forward-looking statements can generally be identified by the
fact that they do not relate strictly to historical or current facts and include, but are not limited to, statements using terminology
such as “can,” “may,” “could,” “should,” “assume,” “forecasts,”
“believe,” “designated to,” “will,” “expect,” “plan,” “anticipate,”
“estimate,” “potential,” “position,” “predicts,” “strategy,” “guidance,”
“intend,” “seek,” “budget,” “project” or “continue,” or the negative
thereof or other comparable terminology regarding beliefs, plans, expectations or intentions regarding the future. You should read
statements that contain these words carefully because they:
|
|
·
|
discuss our future expectations;
|
|
|
·
|
contain projections of our future results of operations or of our financial condition; and
|
|
|
·
|
state other “forward-looking” information.
|
We believe it is important to communicate our expectations.
However, forward-looking statements involve risks and uncertainties and our actual results and the timing of certain events could
differ materially from those discussed in forward-looking statements as a result of certain factors, including those set forth
under “Risk Factors” and “About the Company” set forth in this prospectus and the documents incorporated
herein by reference.
Accordingly, our actual results and the timing of certain events
may differ materially from those expressed or implied in such forward-looking statements due to a variety of factors and risks,
including, but not limited to, those set forth under “Risk Factors,” those set forth from time to time in our other
filings with the SEC, and the following factors and risks:
|
|
·
|
our short operating history with our current business model and lack of significant revenues;
|
|
|
·
|
our history of net losses, which may continue, and our potential inability to achieve profitability;
|
|
|
·
|
the possibility that we may require additional financing, which may involve the issuance of additional shares of common stock
or securities exercisable for common stock and dilute the percentage of ownership held by our current stockholders;
|
|
|
·
|
difficulty in obtaining or inability to obtain, additional financing if such financing becomes necessary.
|
|
|
·
|
volatility in the price and/or trading volume of our common stock;
|
|
|
·
|
future short selling and/or manipulation of the price of our common stock;
|
|
|
·
|
our inability to implement our short and long-term strategies;
|
|
|
·
|
competition from products and services provided by other companies;
|
|
|
·
|
potential difficulties and failures in manufacturing our products;
|
|
|
·
|
loss of strategic relationships;
|
|
|
·
|
dependence on a limited number of key customers;
|
|
|
·
|
lack of acceptance of our products and services by potential customers;
|
|
|
·
|
potential failure to introduce new products and services;
|
|
|
·
|
difficulty or failure in expanding/and or maintaining our sales, marketing and support organizations and our distribution arrangements
necessary to enable us to reach our goals with respect to increasing market acceptance of our products and services;
|
|
|
·
|
seasonality in revenues related to our cotton customer contracts;
|
|
|
·
|
inability to continue to retain the services of Dr. Hayward, our Chief Executive Officer;
|
|
|
·
|
inability to compete effectively in the industries in which we operate;
|
|
|
·
|
lack of success in our research and development efforts for new products;
|
|
|
·
|
failure to manage our growth in operations and acquisitions of new technologies and businesses;
|
|
|
·
|
inability to protect our intellectual property rights;
|
|
|
·
|
intellectual property litigation against us or other legal actions or proceedings in which we may become involved;
|
|
|
·
|
unauthorized disclosure of sensitive or confidential data (including customer data) and cybersecurity breaches; and
|
|
|
·
|
adverse changes in worldwide or domestic economic, political or business conditions.
|
All forward-looking statements and risk factors included in
this prospectus and the documents incorporated herein by reference are made as of the date hereof, based on information available
to us as of such date, and we assume no obligations to update any forward-looking statement or risk factor, unless we are required
to do so by law. If we do update one or more forward-looking statements, no inference should be drawn that we will make updates
with respect to other forward-looking statements or that we will make any further updates to those forward-looking statements at
any future time.
Forward-looking statements may include our plans and objectives
for future operations, including plans and objectives relating to our products and our future economic performance, projections,
business strategy and timing and likelihood of success. Assumptions relating to the foregoing involve judgments with respect to,
among other things, future economic, competitive and market conditions, future business decisions, and the time and money required
to successfully complete development and commercialization of our technologies, all of which are difficult or impossible to predict
accurately and many of which are beyond our control.
Any of the assumptions underlying the forward-looking statements
contained in this prospectus could prove inaccurate and, therefore, we cannot assure you that the results contemplated in any of
such forward-looking statements will be realized. Based on the significant uncertainties inherent in these forward-looking statements,
the inclusion of any such statement should not be regarded as a representation or as a guarantee by us that our objectives or plans
will be achieved, and we caution you against relying on any of the forward-looking statements contained herein.
USE
OF PROCEEDS
Except as otherwise provided in the applicable prospectus supplement,
we intend to use the net proceeds from the sale of the securities covered by this prospectus for general corporate purposes, which
may include, but is not limited to, working capital, capital expenditures, business development and research and development expenditures
and acquisitions of new technologies or businesses. The precise amount, use and timing of the application of such proceeds will
depend upon our funding requirements and the availability and cost of other capital. Additional information on the use of net proceeds
from an offering of securities covered by this prospectus may be set forth in the prospectus supplement relating to the specific
offering.
RATIO
OF EARNINGS TO FIXED CHARGES
The following table sets forth our ratio of earnings to fixed
charges for the periods indicated on a consolidated basis. You should read these ratios of earnings to fixed charges in connection
with our consolidated financial statements, including the notes to those statements, incorporated by reference into this prospectus.
In calculating the ratio of earnings to fixed charges, “earnings”
means the sum of income before income taxes and fixed charges exclusive of capitalized interest, and “fixed charges”
means interest expensed and capitalized, amortized premiums, discounts and capitalized expenses relating to indebtedness and an
estimate of the portion of annual rental expense on leases that represents the interest factor.
|
|
|
Three
months
ended
March 31,
|
|
|
Year ended September 30,
|
|
|
|
|
2017
|
|
|
2016
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
2012
|
|
|
Ratio of Earnings to Fixed Charges
|
|
|
—
|
(1)
|
|
|
—
|
(2)
|
|
|
—
|
(3)
|
|
|
—
|
(4)
|
|
|
—
|
(5)
|
|
|
—
|
(6)
|
|
|
1)
|
Due to the loss for the fiscal quarter ended March 31, 2017, the ratio of earning to fixed charges for that period was less
than 1.00. The deficiency of earnings to total fixed charges was approximately $3,428,856.
|
|
|
2)
|
Due to the loss for the fiscal year ended September 30, 2016, the ratio of earnings to fixed charges for that period was less
than 1.00. The deficiency of earnings to total fixed charges was approximately $12,175,979.
|
|
|
3)
|
Due to the loss for the fiscal year ended September 30, 2015, the ratio of earnings to fixed charges for that period was less
than 1.00. The deficiency of earnings to total fixed charges was approximately $11,881,137.
|
|
|
4)
|
Due to the loss for the fiscal year ended September 30, 2014, the ratio of earnings to fixed charges for that period was less
than 1.00. The deficiency of earnings to total fixed charges was approximately $13,066,661.
|
|
|
5)
|
Due to the loss for the fiscal year ended September 30, 2013, the ratio of earnings to fixed charges for that period was less
than 1.00. The deficiency of earnings to total fixed charges was approximately $17,686,472.
|
|
|
6)
|
Due to the loss for the fiscal year ended September 30, 2012, the ratio of earnings to fixed charges for that period was less
than 1.00. The deficiency of earnings to total fixed charges was approximately $7,150,712.
|
As of the date of this prospectus, we have no shares of preferred
stock outstanding and have not declared or paid any dividends on preferred stock for the periods set forth above.
DIVIDEND
POLICY
We have never declared or paid any cash dividends on our common
stock. We do not anticipate paying any cash dividends to stockholders in the foreseeable future. In addition, any future determination
to pay cash dividends will be at the discretion of our board of directors and will be dependent upon our financial condition, results
of operations, capital requirements, and such other factors as our board of directors deem relevant.
DESCRIPTIONS
OF THE SECURITIES WE MAY OFFER
The descriptions of the securities contained in this prospectus,
together with any applicable prospectus supplement, summarize all the material terms and provisions of the various types of securities
that we may offer. We will describe in the applicable prospectus supplement relating to a particular offering the specific terms
of the securities offered by that prospectus supplement. We will indicate in the applicable prospectus supplement if the terms
of the securities differ from the terms we have summarized below. We will also include in the prospectus supplement information,
where applicable, regarding material United States federal income tax considerations relating to the securities.
We may sell from time to time, in one or more offerings:
|
|
·
|
shares of our common stock;
|
|
|
·
|
shares of our preferred stock;
|
|
|
·
|
warrants to purchase any of the securities listed above;
|
|
|
·
|
rights to purchase common stock, preferred stock or warrants; and/or
|
|
|
·
|
units consisting of one or more of the foregoing.
|
This prospectus may not be used to consummate a sale of securities
unless it is accompanied by a prospectus supplement.
CAPITAL STOCK
General
The following description of common stock and preferred stock,
together with the additional information we include in any applicable prospectus supplement, summarizes the material terms and
provisions of the common stock and preferred stock that we may offer under this prospectus but is not complete. For the complete
terms of our common stock and preferred stock, please refer to our certificate of incorporation, as may be amended from time to
time (the “
Certificate of Incorporation
”), any certificates of designation for our preferred stock, that may
be authorized from time to time, and our by-laws, as amended from time to time. The Delaware General Corporation Law (“
DGCL
”)
may also affect the terms of these securities. While the terms we have summarized below will apply generally to any future common
stock or preferred stock that we may offer, we will describe the specific terms of any series of these securities in more detail
in the applicable prospectus supplement. If we so indicate in a prospectus supplement, the terms of any common stock or preferred
stock we offer under that prospectus supplement may differ from the terms we describe below.
As of May 15, 2017, our authorized capital stock consists of
500,000,000 shares of common stock, par value $0.001 per share, of which 26,351,483 shares were issued and outstanding, and 10,000,000
shares of preferred stock, par value $0.001 per share, of which no shares were issued and outstanding. In addition, as of May 15,
2017, we had options to purchase 5,277,977 shares of common stock issued and outstanding. The authorized and unissued shares of
common stock and preferred stock are available for issuance without further action by our stockholders, unless such action is required
by applicable law or the rules of any stock exchange on which our securities may be listed. Unless approval of our stockholders
is so required, our board of directors will not seek stockholder approval for the issuance and sale of our common stock.
Common Stock
Each holder of our common stock is entitled to one vote for
each share issued and outstanding held on all matters to be voted upon by the holders. Our shares of common stock have no preemptive,
conversion, or redemption rights. Upon the sale of substantially all of our stock or assets or dissolution, liquidation or winding
up, and after all liquidation preferences payable to any series of preferred stock entitled thereto have been satisfied, our remaining
assets shall be distributed to all holders of common stock and any similarly situated stockholders who are not entitled to any
liquidation preference or, if there be an insufficient amount to pay all such stockholders, then ratably among such holders. All
of our issued and outstanding shares of common stock are fully paid and non-assessable. Our Certificate of Incorporation does not
provide for cumulative voting in the election of directors. The holders of shares of our common stock will be entitled to such
cash dividends as may be declared from time to time by our board of directors from funds available therefor.
Our common stock is listed on The NASDAQ Capital Market under
the symbol “APDN.” The transfer agent and registrar for our common stock is American Stock Transfer & Trust Company.
Preferred Stock
Our Certificate of Incorporation provides that our board of
directors may, by resolution, designate classes of preferred stock in the future. The designated series of preferred stock shall
have such powers, designations, preferences and relative, participation or optional or other special rights and qualifications,
limitations or restrictions as shall be expressed in the resolution adopted by the board of directors. Once designated by our board
of directors, each series of preferred stock will have specific financial and other terms that will be described in a prospectus
supplement. The description of the preferred stock that is set forth in any prospectus supplement is not complete without reference
to the documents that govern the preferred stock. These include our Certificate of Incorporation and any certificates of designation
that our board of directors may adopt. Prior to the issuance of shares of each series of preferred stock, the board of directors
is required by the DGCL and our Certificate of Incorporation to adopt resolutions and file a certificate of designations with the
Secretary of State of the State of Delaware. The certificate of designations fixes for each class or series the designations, powers,
preferences, rights, qualifications, limitations and restrictions, including, but not limited to, some or all of the following:
|
|
·
|
the number of shares constituting that series and the distinctive designation of that series, which number may be increased
or decreased (but not below the number of shares then outstanding) from time to time by action of the board of directors;
|
|
|
·
|
the dividend rate and the manner and frequency of payment of dividends on the shares of that series, whether dividends will
be cumulative, and, if so, from which date;
|
|
|
·
|
whether that series will have voting rights, in addition to any voting rights provided by law, and, if so, the terms of such
voting rights;
|
|
|
·
|
whether that series will have conversion privileges, and, if so, the terms and conditions of such conversion, including provision
for adjustment of the conversion rate in such events as the board of directors may determine;
|
|
|
·
|
whether or not the shares of that series will be redeemable, and, if so, the terms and conditions of such redemption;
|
|
|
·
|
whether that series will have a sinking fund for the redemption or purchase of shares of that series, and, if so, the terms
and amount of such sinking fund;
|
|
|
·
|
whether or not the shares of the series will have priority over or be on a parity with or be junior to the shares of any other
series or class in any respect;
|
|
|
·
|
the rights of the shares of that series in the event of voluntary or involuntary liquidation, dissolution or winding up of
the corporation, and the relative rights or priority, if any, of payment of shares of that series; and
|
|
|
·
|
any other relative rights, preferences and limitations of that series.
|
All shares of preferred stock offered hereby will be, when issued,
fully paid and non-assessable, including shares of preferred stock issued upon the exercise of preferred stock warrants or subscription
rights, if any.
Although our board of directors has no intention at the present
time of doing so, it could authorize the issuance of a series of preferred stock that could, depending on the terms of such series,
impede the completion of a merger, tender offer or other takeover attempt.
Anti-Takeover
Effects of Certain Provisions of
our Certificate of Incorporation and By-laws
Our Certificate of Incorporation contains provisions that could
make it more difficult to acquire control of our company by means of a tender offer, open market purchases, a proxy contest or
otherwise. A description of these provisions is set forth below.
Preferred Stock
We believe that the availability of the preferred stock under
our Certificate of Incorporation provides us with flexibility in addressing corporate issues that may arise. Having these authorized
shares available for issuance allows us to issue shares of preferred stock without the expense and delay of a special stockholders’
meeting. The authorized shares of preferred stock, as well as shares of common stock, will be available for issuance without further
action by our stockholders, unless action is required by applicable law or the rules of any stock exchange on which our securities
may be listed. The board of directors has the power, subject to applicable law, to issue series of preferred stock that could,
depending on the terms of the series, impede the completion of a merger, tender offer or other takeover attempt that some, or a
majority, of the stockholders might believe to be in their best interests or in which stockholders might receive a premium for
their stock over the then prevailing market price of the stock.
Advance Notice Procedure
Our by-laws provide an advance notice procedure for stockholders
to nominate director candidates for election or to bring business before an annual meeting of stockholders. Only persons nominated
by, or at the direction of, our board of directors or by a stockholder who has given proper and timely notice to our secretary
prior to the meeting, will be eligible for election as a director. In addition, any proposed business other than the nomination
of persons for election to our board of directors must constitute a proper matter for stockholder action pursuant to the notice
of meeting delivered to us. For notice to be timely, it must be received by our secretary not less than 90 nor more than 120 calendar
days prior to the first anniversary of the previous year’s annual meeting (or if the date of the annual meeting is advanced
more than 30 calendar days or delayed by more than 60 calendar days from the anniversary date of the previous year’s annual
meeting, not earlier than the 90th calendar day prior to such meeting or the 10th calendar day after public disclosure of the date
of such meeting is first made). These advance notice provisions may have the effect of precluding the conduct of certain business
at a meeting if the proper procedures are not followed or may discourage or deter a potential acquirer from conducting a solicitation
of proxies to elect its own slate of directors or otherwise attempt to obtain control of our company.
Special Meetings of Stockholders
Our by-laws provide that special meetings of stockholders may
be called only by the chairman of our board of directors, the Chief Executive Officer, or our board of directors pursuant to a
resolution adopted by a majority of the Board.
Anti-Takeover Effects of Delaware Law
Section 203 of the DGCL (“
Section 203
”) provides
that, subject to exceptions specified therein, an “interested stockholder” of a Delaware corporation shall not engage
in any “business combination,” including general mergers or consolidations or acquisitions of additional shares of
the corporation, with the corporation for a three-year period following the time that such stockholder becomes an interested stockholder
unless:
|
|
·
|
prior to such time, the board of directors of the corporation approved either the business combination or the transaction which
resulted in the stockholder becoming an interested stockholder;
|
|
|
·
|
upon consummation of the transaction which resulted in the stockholder becoming an “interested stockholder,” the
interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced
(excluding specified shares); or
|
|
|
·
|
on or subsequent to such time, the business combination is approved by the board of directors of the corporation and authorized
at an annual or special meeting of stockholders, and not by written consent, by the affirmative vote of at least 66 2/3% of the
outstanding voting stock not owned by the interested stockholder.
|
Under Section 203, the restrictions described above also do
not apply to specified business combinations proposed by an interested stockholder following the announcement or notification of
one of specified transactions involving the corporation and a person who had not been an interested stockholder during the previous
three years or who became an interested stockholder with the approval of a majority of the corporation’s directors, if such
transaction is approved or not opposed by a majority of the directors who were directors prior to any person becoming an interested
stockholder during the previous three years or were recommended for election or elected to succeed such directors by a majority
of such directors. The restrictions described above also do not apply to specified business combinations with a person who is an
“interested stockholder” prior to the time when the corporation’s common stock is listed on a national securities
exchange, so these restrictions would not apply to a business combination with any person who is one of our stockholders prior
to this offering.
Except as otherwise specified in Section 203, an “interested
stockholder” is defined to include:
|
|
·
|
any person that is the owner of 15% or more of the outstanding voting stock of the corporation, or is an affiliate or associate
of the corporation and was the owner of 15% or more of the outstanding voting stock of the corporation at any time within three
years immediately prior to the date of determination; and
|
|
|
·
|
the affiliates and associates of any such person
|
Under some circumstances, Section 203 makes it more difficult
for a person who is an interested stockholder to effect various business combinations with us for a three-year period.
WARRANTS
The following description, together with the additional information
we may include in any applicable prospectus supplement or free writing prospectus, summarizes the material terms and provisions
of the warrants that we may offer under this prospectus and any related warrant agreement and warrant certificate. While the terms
summarized below will apply generally to any warrants that we may offer, we will describe the specific terms of any series of warrants
in more detail in the applicable prospectus supplement. If we indicate in the prospectus supplement, the terms of any warrants
offered under that prospectus supplement may differ from the terms described below. Specific warrant agreements will contain additional
important terms and provisions and will be incorporated by reference as an exhibit to the Registration Statement which includes
this prospectus.
General
We may issue warrants for the purchase of common stock, preferred
stock and/or debt securities in one or more series. We may issue warrants independently or together with common stock, preferred
stock and/or debt securities, and the warrants may be attached to or separate from these securities.
We will evidence each series of warrants by warrant certificates
that we may issue under a separate agreement. We may enter into a warrant agreement with a warrant agent. Each warrant agent may
be a bank that we select which has its principal office in the United States. We may also choose to act as our own warrant agent.
We will indicate the name and address of any such warrant agent in the applicable prospectus supplement relating to a particular
series of warrants.
We will describe in the applicable prospectus supplement the
terms of the series of warrants, including:
|
|
·
|
the offering price and aggregate number of warrants offered;
|
|
|
·
|
if applicable, the designation and terms of the securities with which the warrants are issued and the number of warrants issued
with each such security or each principal amount of such security;
|
|
|
·
|
if applicable, the date on and after which the warrants and the related securities will be separately transferable;
|
|
|
·
|
in the case of warrants to purchase debt securities, the principal amount of debt securities purchasable upon exercise of one
warrant and the price at, and currency in which, this principal amount of debt securities may be purchased upon such exercise;
|
|
|
·
|
in the case of warrants to purchase common stock or preferred stock, the number or amount of shares of common stock or preferred
stock, as the case may be, purchasable upon the exercise of one warrant and the price at which and currency in which these shares
may be purchased upon such exercise;
|
|
|
·
|
the manner of exercise of the warrants, including any cashless exercise rights;
|
|
|
·
|
the warrant agreement under which the warrants will be issued;
|
|
|
·
|
the effect of any merger, consolidation, sale or other disposition of our business on the warrant agreement and the warrants;
|
|
|
·
|
anti-dilution provisions of the warrants, if any;
|
|
|
·
|
the terms of any rights to redeem or call the warrants;
|
|
|
·
|
any provisions for changes to or adjustments in the exercise price or number of securities issuable upon exercise of the warrants;
|
|
|
·
|
the dates on which the right to exercise the warrants will commence and expire or, if the warrants are not continuously exercisable
during that period, the specific date or dates on which the warrants will be exercisable;
|
|
|
·
|
the manner in which the warrant agreement and warrants may be modified;
|
|
|
·
|
the identities of the warrant agent and any calculation or other agent for the warrants;
|
|
|
·
|
federal income tax consequences of holding or exercising the warrants;
|
|
|
·
|
the terms of the securities issuable upon exercise of the warrants;
|
|
|
·
|
any securities exchange or quotation system on which the warrants or any securities deliverable upon exercise of the warrants
may be listed or quoted; and
|
|
|
·
|
any other specific terms, preferences, rights or limitations of or restrictions on the warrants.
|
Before exercising their warrants, holders of warrants will not
have any of the rights of holders of the securities purchasable upon such exercise, including:
|
|
·
|
in the case of warrants to purchase debt securities, the right to receive payments of principal of, or premium, if any, or
interest on, the debt securities purchasable upon exercise or to enforce covenants in the applicable indenture; or
|
|
|
·
|
in the case of warrants to purchase common stock or preferred stock, the right to receive dividends, if any, or, payments upon
our liquidation, dissolution or winding up or to exercise voting rights, if any.
|
Exercise of Warrants
Each warrant will entitle the holder to purchase the securities
that we specify in the applicable prospectus supplement at the exercise price that we describe in the applicable prospectus supplement.
Unless we otherwise specify in the applicable prospectus supplement, holders of the warrants may exercise the warrants at any time
up to 5:00 P.M. eastern time on the expiration date that we set forth in the applicable prospectus supplement. After the close
of business on the expiration date, unexercised warrants will become void.
Holders of the warrants may exercise the warrants by delivering
the warrant certificate representing the warrants to be exercised together with specified information, and paying the required
exercise price by the methods provided in the applicable prospectus supplement. We will set forth on the reverse side of the warrant
certificate, and in the applicable prospectus supplement, the information that the holder of the warrant will be required to deliver
to the warrant agent.
Upon receipt of the required payment and the warrant certificate
properly completed and duly executed at the corporate trust office of the warrant agent or any other office indicated in the applicable
prospectus supplement, we will issue and deliver the securities purchasable upon such exercise. If fewer than all of the warrants
represented by the warrant certificate are exercised, then we will issue a new warrant certificate for the remaining amount of
warrants.
Enforceability of Rights by Holders of Warrants
Any warrant agent will act solely as our agent under the applicable
warrant agreement and will not assume any obligation or relationship of agency or trust with any holder of any warrant. A single
bank or trust company may act as warrant agent for more than one issue of warrants. A warrant agent will have no duty or responsibility
in case of any default by us under the applicable warrant agreement or warrant, including any duty or responsibility to initiate
any proceedings at law or otherwise, or to make any demand upon us. Any holder of a warrant may, without the consent of the related
warrant agent or the holder of any other warrant, enforce by appropriate legal action the holder’s right to exercise, and
receive the securities purchasable upon exercise of, its warrants in accordance with their terms.
Warrant Agreement Will Not Be Qualified Under Trust Indenture
Act
No warrant agreement will be qualified as an indenture, and
no warrant agent will be required to qualify as a trustee, under the Trust Indenture Act. Therefore, holders of warrants issued
under a warrant agreement will not have the protection of the Trust Indenture Act with respect to their warrants.
Governing Law
Each warrant agreement and any warrants issued under the warrant
agreements will be governed by New York law.
Calculation Agent
Any calculations relating to warrants may be made by a calculation
agent, an institution that we appoint as our agent for this purpose. The prospectus supplement for a particular warrant will name
the institution that we have appointed to act as the calculation agent for that warrant as of the original issue date for that
warrant, if any. We may appoint a different institution to serve as calculation agent from time to time after the original issue
date without the consent or notification of the holders. The calculation agent’s determination of any amount of money payable
or securities deliverable with respect to a warrant will be final and binding in the absence of manifest error.
Outstanding Warrants
As of May 15, 2017, we had outstanding 9,548,969 warrants to
purchase 9,548,969 shares of our common stock at a weighted average exercise price of $3.60. Of these warrants, 7,245,517 are listed
on The NASDAQ Capital Market under the symbol “APDNW.” Our Warrant Agent is American Stock Transfer & Trust Company.
Effective November 20, 2014, we consummated an underwritten
public offering of 2,800,000 shares of our common stock and 2,800,000 warrants to purchase 2,800,000 shares of our common stock.
The combined offering price of one share of common stock, together with one warrant, was $3.25. The warrants have an exercise price
of $3.50 and expire on November 20, 2019. In connection with this offering, the underwriters exercised their over-allotment option
by purchasing 52,000 shares of common stock and 416,850 warrants.
Effective April 1, 2015, we consummated an underwritten public
offering of 4,011,000 shares of our common stock and 1,604,400 warrants to purchase 1,604,400 shares of our common stock. The combined
offering price of one share of common stock, together with one warrant, was $3.00. The warrants have an exercise price of $3.50
and expire on November 20, 2019. In connection with this offering, the underwriters exercised their over-allotment option by purchasing
87,000 shares of common stock and 152,800 warrants.
Effective November 2, 2016, we consummated a private placement
of our common stock and warrants for an aggregate purchase price of $5.0 million pursuant to a securities purchase agreement with
an institutional investor. The combined offering price of one share of common stock, together with one warrant, was $2.20. In this
private placement, we sold 2,272,727 shares of our common stock and 2,272,727 warrants to purchase 2,272,727 shares of our common
stock. The warrants have the same terms as our existing publicly traded warrants. These warrants have an exercise price of $3.50
and expire on November 20, 2019.
In addition, there are 486,144 warrants outstanding which were
issued to various placement agents between 2010 and 2016.
DEBT SECURITIES
The following description, together with the additional information
we include in any applicable prospectus supplements, summarizes the material terms and provisions of the debt securities that we
may offer under this prospectus. While the terms we have summarized below will generally apply to any future debt securities we
may offer under this prospectus, we will describe the particular terms of any debt securities that we may offer in more detail
in the applicable prospectus supplement or a free writing prospectus. The terms of any debt securities we offer under a prospectus
supplement or a free writing prospectus may differ from the terms we describe below. As of the date of this prospectus, we have
no outstanding registered debt securities.
We will issue senior notes under a senior indenture, which we
will enter into with the trustee to be named in the senior indenture. We will issue subordinated notes under a subordinated indenture,
which we will enter into with the trustee to be named in the subordinated indenture. We have filed forms of these documents as
exhibits to the Registration Statement of which this prospectus is a part. We use the term “indentures” to refer to
both the senior indenture and the subordinated indenture.
The indentures will be qualified under the Trust Indenture Act
of 1939, unless an exemption from the qualification provisions is applicable. References to the Trust Indenture Act of 1939 include
all amendments thereto. We use the term “debenture trustee” to refer to either the senior trustee or the subordinated
trustee, as applicable.
The following summaries of material provisions of the senior
notes, the subordinated notes and the indentures are subject to, and qualified in their entirety by reference to, all the provisions
of the indenture applicable to a particular series of debt securities, and all supplements thereto. We urge you to read the applicable
prospectus supplements related to the debt securities that we sell under this prospectus, as well as the complete indentures that
contain the terms of the debt securities. Except as we may otherwise indicate, the terms of the senior and the subordinated indentures
are identical.
General
The terms of each series of debt securities will be established
by or pursuant to a resolution of our board of directors and set forth or determined in the manner provided in an officers’
certificate or by a supplemental indenture. Debt securities may be issued in separate series without limitation as to aggregate
principal amount. We may specify a maximum aggregate principal amount for the debt securities of any series. In addition, the particular
terms of each series of debt securities will be described in a prospectus supplement relating to such series, including any pricing
supplement. The prospectus supplement will set forth, among other things:
|
|
·
|
the principal amount being offered, and, if a series, the total amount authorized and the total amount outstanding;
|
|
|
·
|
any limit on the amount that may be issued;
|
|
|
·
|
whether or not we will issue the series of debt securities in global form and, if so, the terms and who the depositary will
be;
|
|
|
·
|
whether and under what circumstances, if any, we will pay additional amounts on any debt securities held by a person who is
not a U.S. person for tax purposes, and whether we can redeem the debt securities if we have to pay such additional amounts;
|
|
|
·
|
the annual interest rate, which may be fixed or variable, or the method for determining the rate, the date interest will begin
to accrue, the dates interest will be payable and the regular record dates for interest payment dates or the method for determining
such dates;
|
|
|
·
|
the terms of the subordination of any series of subordinated debt, if applicable;
|
|
|
·
|
the place where payments will be payable;
|
|
|
·
|
restrictions on transfer, sale or other assignment, if any;
|
|
|
·
|
our right, if any, to defer payment of interest and the maximum length of any such deferral period;
|
|
|
·
|
the date, if any, after which, the conditions upon which, and the price at which we may, at our option, redeem the series of
debt securities pursuant to any optional or provisional redemption provisions, and any other applicable terms of those redemption
provisions;
|
|
|
·
|
the date, if any, on which, and the price at which we are obligated, pursuant to any mandatory sinking fund or analogous fund
provisions or otherwise, to redeem, or at the holder’s option to purchase, the series of debt securities and the currency
or currency unit in which the debt securities are payable;
|
|
|
·
|
whether the indenture will restrict our ability and/or the ability of our subsidiaries to, among other things:
|
|
|
·
|
incur additional indebtedness;
|
|
|
·
|
issue additional securities;
|
|
|
·
|
pay dividends and make distributions in respect of our capital stock and the capital stock of our subsidiaries;
|
|
|
·
|
place restrictions on our subsidiaries’ ability to pay dividends, make distributions or transfer assets;
|
|
|
·
|
make investments or other restricted payments;
|
|
|
·
|
sell or otherwise dispose of assets;
|
|
|
·
|
enter into sale-leaseback transactions;
|
|
|
·
|
engage in transactions with stockholders and affiliates;
|
|
|
·
|
issue or sell stock of our subsidiaries; or
|
|
|
·
|
effect a consolidation or merger;
|
|
|
·
|
whether the indenture will require us to maintain any interest coverage, fixed charge, cash flow-based, asset-based or other
financial ratios;
|
|
|
·
|
information describing any book-entry features;
|
|
|
·
|
provisions for a sinking fund purchase or other analogous fund, if any;
|
|
|
·
|
whether the debt securities are to be offered at a price such that they will be deemed to be offered at an “original
issue discount” as defined in paragraph (a) of Section 1273 of the Internal Revenue Code;
|
|
|
·
|
the procedures for any auction and remarketing, if any;
|
|
|
·
|
the denominations in which we will issue the series of debt securities, if other than denominations of $1,000 and any integral
multiple thereof;
|
|
|
·
|
if other than dollars, the currency in which the series of debt securities will be denominated; and
|
|
|
·
|
any other specific terms, preferences, rights or limitations of, or restrictions on, the debt securities, including any events
of default that are in addition to those described in this prospectus or any covenants provided with respect to the debt securities
that are in addition to those described above, and any terms that may be required by us or advisable under applicable laws or regulations
or advisable in connection with the marketing of the debt securities.
|
Conversion or Exchange Rights
We will set forth in the prospectus supplement the terms on
which a series of debt securities may be convertible into or exchangeable for common stock or other securities of ours or a third
party, including the conversion or exchange rate, as applicable, or how it will be calculated, and the applicable conversion or
exchange period. We will include provisions as to whether conversion or exchange is mandatory, at the option of the holder or at
our option. We may include provisions pursuant to which the number of our securities or the securities of a third party that the
holders of the series of debt securities receive upon conversion or exchange would, under the circumstances described in those
provisions, be subject to adjustment, or pursuant to which those holders would, under those circumstances, receive other property
upon conversion or exchange, for example in the event of our merger or consolidation with another entity.
Consolidation, Merger or Sale
The indentures in the forms initially filed as exhibits to the
registration statement of which this prospectus is a part do not contain any covenant that restricts our ability to merge or consolidate,
or sell, convey, transfer or otherwise dispose of all or substantially all of our assets. However, any successor of ours or the
acquirer of such assets must assume all of our obligations under the indentures and the debt securities.
If the debt securities are convertible for our other securities,
the person with whom we consolidate or merge or to whom we sell all of our property must make provisions for the conversion of
the debt securities into securities that the holders of the debt securities would have received if they had converted the debt
securities before the consolidation, merger or sale.
Events of Default under the Indenture
The following are events of default under the indentures in
the forms initially filed as exhibits to the Registration Statement with respect to any series of debt securities that we may issue:
|
|
·
|
if we fail to pay interest when due and payable and our failure continues for 90 days and the time for payment has not been
extended or deferred;
|
|
|
·
|
if we fail to pay the principal, sinking fund payment or premium, if any, when due and payable and the time for payment has
not been extended or delayed;
|
|
|
·
|
if we fail to observe or perform any other covenant contained in the debt securities or the indentures, other than a covenant
specifically relating to another series of debt securities, and our failure continues for 90 days after we receive notice from
the debenture trustee or holders of at least 25% in aggregate principal amount of the outstanding debt securities of the applicable
series; and
|
|
|
·
|
if specified events of bankruptcy, insolvency or reorganization occur.
|
If an event of default with respect to debt securities of any
series occurs and is continuing, other than an event of default specified in the last bullet point above, the debenture trustee
or the holders of at least 25% in aggregate principal amount of the outstanding debt securities of that series, by notice to us
in writing, and to the debenture trustee if notice is given by such holders, may declare the unpaid principal of, premium, if any,
and accrued interest, if any, due and payable immediately. If an event of default specified in the last bullet point above occurs
with respect to us, the principal amount of and accrued interest, if any, of each issue of debt securities then outstanding shall
be due and payable without any notice or other action on the part of the debenture trustee or any holder.
The holders of a majority in principal amount of the outstanding
debt securities of an affected series may waive any default or event of default with respect to the series and its consequences,
except defaults or events of default regarding payment of principal, premium, if any, or interest, unless we have cured the default
or event of default in accordance with the indenture. Any waiver shall cure the default or event of default.
Subject to the terms of the indentures, if an event of default
under an indenture shall occur and be continuing, the debenture trustee will be under no obligation to exercise any of its rights
or powers under such indenture at the request or direction of any of the holders of the applicable series of debt securities, unless
such holders have offered the debenture trustee reasonable indemnity. The holders of a majority in principal amount of the outstanding
debt securities of any series will have the right to direct the time, method and place of conducting any proceeding for any remedy
available to the debenture trustee, or exercising any trust or power conferred on the debenture trustee, with respect to the debt
securities of that series, provided that:
|
|
·
|
the direction so given by the holder is not in conflict with any law or the applicable indenture; and
|
|
|
·
|
subject to its duties under the Trust Indenture Act of 1939, the debenture trustee need not take any action that might involve
it in personal liability or might be unduly prejudicial to the holders not involved in the proceeding.
|
A holder of the debt securities of any series will only have
the right to institute a proceeding under the indentures or to appoint a receiver or trustee, or to seek other remedies if:
|
|
·
|
the holder has given written notice to the debenture trustee of a continuing event of default with respect to that series;
|
|
|
·
|
the holders of at least 25% in aggregate principal amount of the outstanding debt securities of that series have made written
request, and such holders have offered reasonable indemnity, to the debenture trustee to institute the proceeding as trustee; and
|
|
|
·
|
the debenture trustee does not institute the proceeding and does not receive from the holders of a majority in aggregate principal
amount of the outstanding debt securities of that series other conflicting directions within 90 days after the notice, request
and offer.
|
These limitations do not apply to a suit instituted by a holder
of debt securities if we default in the payment of the principal, premium, if any, or interest on, the debt securities.
We will periodically file statements with the debenture trustee
regarding our compliance with specified covenants in the indentures.
Modification of Indenture; Waiver
We and the debenture trustee may change an indenture without
the consent of any holders with respect to specific matters, including:
|
|
·
|
to fix any ambiguity, defect or inconsistency in the indenture;
|
|
|
·
|
to comply with the provisions described above under “—Consolidation, Merger or Sale”;
|
|
|
·
|
to comply with any requirements of the SEC in connection with the qualification of any indenture under the Trust Indenture
Act of 1939;
|
|
|
·
|
to evidence and provide for the acceptance of appointment by a successor trustee;
|
|
|
·
|
to provide for uncertificated debt securities and to make all appropriate changes for such purpose;
|
|
|
·
|
to add to, delete from, or revise the conditions, limitations and restrictions on the authorized amount, terms or purposes
of issuance, authorization and delivery of debt securities or any series, as set forth in the indenture;
|
|
|
·
|
to provide for the issuance of and establish the form and terms and conditions of the debt securities of any series as provided
under “—General” to establish the form of any certifications required to be furnished pursuant to the terms of
the indenture or any series of debt securities, or to add to the rights of the holders of any series of debt securities;
|
|
|
·
|
to add to our covenants such new covenants, restrictions, conditions or provisions for the protection of the holders, to make
the occurrence, or the occurrence and the continuance, of a default in any such additional covenants, restrictions, conditions
or provisions an event of default, or to surrender any of our rights or powers under the indenture; or
|
|
|
·
|
to change anything that does not materially adversely affect the interests of any holder of debt securities of any series.
|
In addition, under the indentures, the rights of holders of
a series of debt securities may be changed by us and the debenture trustee with the written consent of the holders of at least
a majority in aggregate principal amount of the outstanding debt securities of each series that is affected. However, we and the
debenture trustee may only make the following changes with the consent of each holder of any outstanding debt securities affected:
|
|
·
|
extending the fixed maturity of the series of debt securities;
|
|
|
·
|
reducing the principal amount, reducing the rate of or extending the time of payment of interest, or reducing any premium payable
upon the redemption of any debt securities; or
|
|
|
·
|
reducing the percentage of debt securities, the holders of which are required to consent to any amendment, supplement, modification
or waiver.
|
Discharge
Each indenture provides that we can elect to be discharged from
our obligations with respect to one or more series of debt securities, except that the following obligations, among others survive
until the maturity date or the redemption date:
|
|
·
|
register the transfer or exchange of debt securities of the series;
|
|
|
·
|
replace stolen, lost or mutilated debt securities of the series;
|
|
|
·
|
maintain paying agencies;
|
|
|
·
|
hold monies for payment in trust; and
|
|
|
·
|
appoint any successor trustee;
|
and the following obligations survive the maturity date or the
redemption date:
|
|
·
|
recover excess money held by the debenture trustee; and
|
|
|
·
|
compensate and indemnify the debenture trustee.
|
As more fully set forth in the indentures, in order to exercise
our rights to be discharged, we must either deliver for cancellation all securities of a series to the debenture trustee or must
deposit with the debenture trustee money or government obligations sufficient to pay all the principal of, any premium, if any,
and interest on, the debt securities of the series on the dates payments are due.
Form, Exchange and Transfer
We will issue the debt securities of each series only in fully
registered form without coupons and, unless we otherwise specify in the applicable prospectus supplement, in denominations of $1,000
and any integral multiple thereof. The indentures provide that we may issue debt securities of a series in temporary or permanent
global form and as book-entry securities that will be deposited with, or on behalf of, The Depository Trust Company, New York,
New York, known as DTC, or another depositary named by us and identified in a prospectus supplement with respect to that series.
At the option of the holder, subject to the terms of the indentures
and the limitations applicable to global securities described in the applicable prospectus supplement, the holder of the debt securities
of any series can exchange the debt securities for other debt securities of the same series, in any authorized denomination and
of like tenor and aggregate principal amount.
Subject to the terms of the indentures and the limitations applicable
to global securities set forth in the applicable prospectus supplement, holders of the debt securities may present the debt securities
for exchange or for registration of transfer, duly endorsed or with the form of transfer endorsed thereon duly executed if so required
by us or the security registrar, at the office of the security registrar or at the office of any transfer agent designated by us
for this purpose. Unless otherwise provided in the debt securities that the holder presents for transfer or exchange, we will make
no service charge for any registration of transfer or exchange, but we may require payment of any taxes or other governmental charges.
We will name in a board resolution the security registrar, and
any transfer agent in addition to the security registrar, that we initially designate for any debt securities. We may at any time
designate additional transfer agents or rescind the designation of any transfer agent or approve a change in the office through
which any transfer agent acts, except that we will be required to maintain a transfer agent in each place of payment for the debt
securities of each series.
If we elect to redeem the debt securities of any series, we
will not be required to:
|
|
·
|
issue, register the transfer of, or exchange any debt securities of any series being redeemed in part during a period beginning
at the opening of business 15 days before the day of mailing of a notice of redemption of any debt securities that may be selected
for redemption and ending at the close of business on the day of the mailing; or
|
|
|
·
|
register the transfer of or exchange any debt securities so selected for redemption, in whole or in part, except the unredeemed
portion of any debt securities we are redeeming in part.
|
Information Concerning the Debenture Trustee
The debenture trustee, other than during the occurrence and
continuance of an event of default under an indenture, undertakes to perform only those duties as are specifically set forth in
the applicable indenture. Upon an event of default under an indenture, the debenture trustee must use the same degree of care as
a prudent person would exercise or use in the conduct of his or her own affairs. Subject to this provision, the debenture trustee
is under no obligation to exercise any of the powers given it by the indentures at the request of any holder of debt securities
unless it is offered reasonable security and indemnity against the costs, expenses and liabilities that it might incur.
Payment and Paying Agents
Unless we otherwise indicate in the applicable prospectus supplement,
we will make payment of the interest on any debt securities on any interest payment date to the person in whose name the debt securities,
or one or more predecessor securities, are registered at the close of business on the regular record date for the interest.
We will name in the applicable board resolution any other paying
agents that we initially designate for the debt securities of a particular series. We will maintain a paying agent in each place
of payment for the debt securities of a particular series.
All money we pay to a paying agent or the debenture trustee
for the payment of the principal of or any premium or interest on any debt securities that remains unclaimed at the end of two
years after such principal, premium or interest has become due and payable will be repaid to us, and the holder of the debt security
thereafter may look only to us for payment thereof.
Governing Law
The indentures and the debt securities will be governed by and
construed in accordance with the laws of the State of New York, except to the extent that the Trust Indenture Act of 1939 is applicable.
Subordination of Subordinated Debt Securities
The subordinated debt securities will be subordinate and junior
in priority of payment to certain of our other indebtedness to the extent described in a prospectus supplement. The indentures
in the forms initially filed as exhibits to the Registration Statement of which this prospectus is a part do not limit the amount
of indebtedness that we may incur, including senior indebtedness or subordinated indebtedness, and do not limit us from issuing
any other debt, including secured debt or unsecured debt.
RIGHTS
We may issue rights to purchase common stock, preferred stock
or warrants that we may offer to our security holders in one or more series. The rights may or may not be transferable by the persons
purchasing or receiving the rights. In connection with any rights offering, we may enter into a standby underwriting or other arrangement
with one or more underwriters or other persons pursuant to which such underwriters or other persons would purchase any offered
securities remaining unsubscribed for after such rights offering. Each series of rights will be issued under a separate rights
agent agreement to be entered into between us and a bank or trust company, as rights agent, that we will name in the applicable
prospectus supplement. The rights agent will act solely as our agent in connection with the rights and will not assume any obligation
or relationship of agency or trust for or with any holders of rights certificates or beneficial owners of rights. A copy of the
form of rights agent or subscription agent agreement, including the form of rights certificate representing a series of rights,
will be filed with the SEC in connection with the offering of a particular series of rights.
The prospectus supplement relating to any rights that we offer
will include specific terms relating to the offering, including, among other matters:
|
|
·
|
the title of the rights;
|
|
|
·
|
the securities for which the rights are exercisable;
|
|
|
·
|
the date of determining the security holders entitled to the rights distribution;
|
|
|
·
|
the aggregate number of rights issued and the aggregate number of shares of common stock or preferred stock or warrants purchasable
upon exercise of the rights;
|
|
|
·
|
the extent to which the rights are transferable;
|
|
|
·
|
any provisions for changes to or adjustments in the exercise price or number of securities issuable upon exercise of the rights;
|
|
|
·
|
the conditions to completion of the rights offering;
|
|
|
·
|
any applicable federal income tax considerations;
|
|
|
·
|
if applicable, the material terms of any standby underwriting or other purchase arrangement that we may enter into in connection
with the rights offering;
|
|
|
·
|
the date on which the right to exercise the rights will commence and the date on which the rights will expire; and
|
|
|
·
|
any other terms of the rights, including terms, procedures and limitations relating to the exchange and exercise of the rights.
|
Each right would entitle the holder of the rights to purchase
for cash the amount of shares of common stock or preferred stock or warrants at the exercise price set forth in the applicable
prospectus supplement. Rights may be exercised at any time up to the close of business on the expiration date for the rights provided
in the applicable prospectus supplement. After the close of business on the expiration date, all unexercised rights will become
void.
We may determine to offer any unsubscribed securities directly
to persons other than our security holders, to or through agents, underwriters or dealers or through a combination of such methods,
including pursuant to standby arrangements, as described in the applicable prospectus supplement.
Until a holder exercises the rights to purchase shares of our
common stock or preferred stock or warrants, the holder will not have any rights as a holder of shares of our common stock or preferred
stock or warrants, as the case may be, by virtue of ownership of the rights.
UNITS
We may issue units consisting of one or more of the other securities
described in this prospectus, in any prospectus supplement or a free writing prospectus in any combination. Each unit will be issued
so that the holder of the unit is also the holder, with the rights and obligations of a holder, of each security included in the
unit. The unit agreement under which a unit is issued may provide that the securities included in the unit may not be held or transferred
separately, at any time or at any time before a specified date or upon the occurrence of a specified event or occurrence.
The applicable prospectus supplement or free writing prospectus
will describe:
|
|
·
|
the designation and terms of the units and of the securities comprising the units, including whether and under what circumstances
those securities may be held or transferred separately;
|
|
|
·
|
any unit agreement under which the units will be issued;
|
|
|
·
|
any provisions for the issuance, payment, settlement, transfer or exchange of the units or of the securities comprising the
units; and
|
|
|
·
|
whether the units will be issued in fully registered or global form.
|
PLAN
OF DISTRIBUTION
We may sell the securities offered pursuant to this prospectus
from time to time in one or more transactions, including, without limitation:
|
|
·
|
to or through underwriters;
|
|
|
·
|
through broker-dealers (acting as agent or principal);
|
|
|
·
|
directly by us to one or more purchasers (including our affiliates and stockholders), through a specific bidding or auction
process, a rights offering or otherwise;
|
|
|
·
|
through a combination of any such methods of sale; or
|
|
|
·
|
through any other methods described in a prospectus supplement.
|
The distribution of securities may be effected, from time to
time, in one or more transactions, including:
|
|
·
|
block transactions (which may involve crosses) and transactions on The Nasdaq Capital Market or any other organized market
where the securities may be traded;
|
|
|
·
|
purchases by a broker-dealer as principal and resale by the broker-dealer for its own account pursuant to a prospectus supplement;
|
|
|
·
|
ordinary brokerage transactions and transactions in which a broker-dealer solicits purchasers;
|
|
|
·
|
sales “at the market” to or through a market maker or into an existing trading market, on an exchange or otherwise;
and
|
|
|
·
|
sales in other ways not involving market makers or established trading markets, including direct sales to purchasers.
|
The applicable prospectus supplement will describe the terms
of the offering of the securities, including:
|
|
·
|
the name or names of any underwriters, if, and if required, any dealers or agents;
|
|
|
·
|
the purchase price of the securities and the proceeds we will receive from the sale;
|
|
|
·
|
any underwriting discounts and other items constituting underwriters’ compensation;
|
|
|
·
|
any discounts or concessions allowed or re-allowed or paid to dealers; and
|
|
|
·
|
any securities exchange or market on which the securities may be listed or traded.
|
We may distribute the securities from time to time in one or
more transactions at:
|
|
·
|
a fixed price or prices, which may be changed;
|
|
|
·
|
market prices prevailing at the time of sale;
|
|
|
·
|
prices related to such prevailing market prices; or
|
Only underwriters named in the prospectus supplement are underwriters
of the securities offered by the prospectus supplement.
If underwriters are used in an offering, we will execute an
underwriting agreement with such underwriters and will specify the name of each underwriter and the terms of the transaction (including
any underwriting discounts and other terms constituting compensation of the underwriters and any dealers) in a prospectus supplement.
The securities may be offered to the public either through underwriting syndicates represented by managing underwriters or directly
by one or more investment banking firms or others, as designated. If an underwriting syndicate is used, the managing underwriter(s)
will be specified on the cover of the prospectus supplement. If underwriters are used in the sale, the offered securities will
be acquired by the underwriters for their own accounts and may be resold from time to time in one or more transactions, including
negotiated transactions, at a fixed public offering price or at varying prices determined at the time of sale. Any public offering
price and any discounts or concessions allowed or re-allowed or paid to dealers may be changed from time to time. Unless otherwise
set forth in the prospectus supplement, the obligations of the underwriters to purchase the offered securities will be subject
to conditions precedent, and the underwriters will be obligated to purchase all of the offered securities, if any are purchased.
We may grant to the underwriters options to purchase additional
securities to cover over-allotments, if any, at the public offering price, with additional underwriting commissions or discounts,
as may be set forth in a related prospectus supplement. The terms of any over-allotment option will be set forth in the prospectus
supplement for those securities.
If a dealer is used in the sale of the securities, we, or an
underwriter, will sell the securities to the dealer, as principal. The dealer may then resell the securities to the public at varying
prices to be determined by the dealer at the time of resale. To the extent required, we will set forth in the prospectus supplement,
document incorporated by reference or free writing prospectus, as applicable, the name of the dealer and the terms of the transactions.
We may sell the securities directly or through agents we designate
from time to time. We will name any agent involved in the offering and sale of securities and we will describe any commissions
we will pay the agent in the prospectus supplement.
We may authorize agents or underwriters to solicit offers by
institutional investors to purchase securities from us at the public offering price set forth in the prospectus supplement pursuant
to delayed delivery contracts providing for payment and delivery on a specified date in the future. We will describe the conditions
to these contracts and the commissions we must pay for solicitation of these contracts in the prospectus supplement.
In connection with the sale of the securities, underwriters,
dealers or agents may receive compensation from us or from purchasers of the securities for whom they act as agents, in the form
of discounts, concessions or commissions. Underwriters may sell the securities to or through dealers, and those dealers may receive
compensation in the form of discounts, concessions or commissions from the underwriters or commissions from the purchasers for
whom they may act as agents. Underwriters, dealers and agents that participate in the distribution of the securities, and any institutional
investors or others that purchase securities directly for the purpose of resale or distribution, may be deemed to be underwriters,
and any discounts or commissions received by them from us and any profit on the resale of the common stock by them may be deemed
to be underwriting discounts and commissions under the Securities Act. No FINRA member firm may receive compensation in excess
of that allowable under FINRA rules, including Rule 5110, in connection with the offering of the securities.
We may provide agents, underwriters and other purchasers with
indemnification against particular civil liabilities, including liabilities under the Securities Act, or contribution with respect
to payments that the agents, underwriters or other purchasers may make with respect to such liabilities. Agents and underwriters
may engage in transactions with, or perform services for, us in the ordinary course of business.
To facilitate the public offering of a series of securities,
persons participating in the offering may engage in transactions that stabilize, maintain, or otherwise affect the market price
of the securities. This may include over-allotments or short sales of the securities, which involves the sale by persons participating
in the offering of more securities than have been sold to them by us. In addition, those persons may stabilize or maintain the
price of the securities by bidding for or purchasing securities in the open market or by imposing penalty bids, whereby selling
concessions allowed to underwriters or dealers participating in any such offering may be reclaimed if securities sold by them are
repurchased in connection with stabilization transactions. The effect of these transactions may be to stabilize or maintain the
market price of the securities at a level above that which might otherwise prevail in the open market. Such transactions, if commenced,
may be discontinued at any time. We make no representation or prediction as to the direction or magnitude of any effect that the
transactions described above, if implemented, may have on the price of our securities.
Unless otherwise specified in the applicable prospectus supplement,
any common stock sold pursuant to a prospectus supplement will be eligible for trading as listed on The NASDAQ Capital Market.
Any underwriters to whom securities are sold by us for public offering and sale may make a market in the securities, but such underwriters
will not be obligated to do so and may discontinue any market making at any time without notice.
In order to comply with the securities laws of some states,
if applicable, the securities offered pursuant to this prospectus will be sold in those states only through registered or licensed
brokers or dealers. In addition, in some states securities may not be sold unless they have been registered or qualified for sale
in the applicable state or an exemption from the registration or qualification requirement is available and complied with.
So long as the aggregate market value of our voting and non-voting
common equity held by non-affiliates is less than $75,000,000 and so long as required by the rules of the SEC, the amount of securities
we may offer hereunder will be limited such that the aggregate market value of securities sold by us during a period of 12 calendar
months cannot exceed one-third of the aggregate market value of the voting and non-voting common equity held by non-affiliates.
To the extent required, this prospectus may be amended or supplemented
from time to time to describe a specific plan of distribution.
LEGAL
MATTERS
The validity of the issuance of the offered securities will
be passed upon for us by Pepper Hamilton LLP, New York, New York.
EXPERTS
Marcum LLP, independent registered public accounting firm, has
audited our consolidated financial statements included in our Annual Report on Form 10-K for the year ended September 30, 2016,
as set forth in their report, which is incorporated by reference in the prospectus and elsewhere in this registration statement.
Our consolidated financial statements are incorporated by reference in reliance on Marcum LLP’s report, given on their authority
as experts in accounting and auditing.
WHERE
YOU CAN FIND MORE INFORMATION
This prospectus and any subsequent prospectus supplements do
not contain all of the information in the Registration Statement. We have omitted from this prospectus some parts of the Registration
Statement as permitted by the rules and regulations of the SEC. Statements in this prospectus concerning any document we have filed
as an exhibit to the Registration Statement or that we otherwise filed with the SEC are not intended to be comprehensive and are
qualified in their entirety by reference to these filings. In addition, we file annual, quarterly and current reports, proxy statements
and other information with the SEC. You may read and copy any documents that we have filed with the SEC at the SEC’s Public
Reference Room at 100 F Street, N.E., Room 1580, Washington, D.C. 20549. Please call the SEC at 1-800-SEC-0330 for further information
about the operation of the Public Reference Room. The SEC also maintains a website that contains reports, proxy and information
statements and other information that registrants file electronically with the SEC, including us. The SEC’s website can be
found at
http://www.sec.gov
. In addition, we make available on or through our website copies of these reports as soon as
reasonably practicable after we electronically file or furnished them to the SEC. Our website can be found at
http:www.adnas.com
.
Our website is not a part of this prospectus.
INFORMATION
INCORPORATED BY REFERENCE
We have elected to incorporate certain information by reference
into this prospectus. By incorporating by reference, we can disclose important information to you by referring you to other documents
we have filed or will file with the SEC. The information incorporated by reference is deemed to be part of this prospectus, except
for information incorporated by reference that is superseded by information contained in this prospectus. This means that you must
look at all of the SEC filings that we incorporate by reference to determine if any statements in the prospectus or any document
previously incorporated by reference have been modified or superseded. This prospectus incorporates by reference the documents
set forth below that we have previously filed with the SEC, except in each case the information contained in such document to the
extent “furnished” and not “filed”:
|
|
·
|
Our Annual Report on Form 10-K for the fiscal year ended September 30, 2016, filed with the SEC on December 6, 2016.
|
|
|
·
|
Our Quarterly Report on Form 10-Q for the quarterly period ended December 31, 2016, filed with the SEC on February 9, 2017.
|
|
|
·
|
Our Quarterly Report on Form 10-Q for the quarterly period ended March 31, 2017, filed with the SEC on May 11, 2017.
|
|
|
·
|
Our Current Reports on Form 8-K filed with the SEC on October 10, 2016 (except Exhibit 99.1), November 3, 2016 (except Exhibit
99.1), December 6, 2016 (except Item 2.02 and Exhibit 99.1), February 9, 2017 (except Item 2.02 and Exhibit 99.1), March 11, 2017
and May 11, 2017.
|
|
|
·
|
The description of our capital stock contained in our registration statement on Form 8-A (File No. 001-36745) filed with the
Commission on November 13, 2014, pursuant to Section 12(b) of the Exchange Act, including any amendment or report filed for the
purpose of updating such description.
|
We also incorporate by reference all documents we file in the
future pursuant to Section 13(a), 13(c), 14 or 15(d) of the Exchange Act after the date of this prospectus and prior to the sale
of all the securities covered by this prospectus (including all such documents filed with the SEC after the date of the initial
filing of the Registration Statement that contains this prospectus and prior to effectiveness of the Registration Statement or
after such effectiveness), except in each case the information contained in such document to the extent “furnished”
and not “filed.”
You may obtain copies of these documents on the website maintained
by the SEC at
http://www.sec.gov
, or from us without charge (other than exhibits to such documents, unless such exhibits
are specifically incorporated by reference into such documents) by writing us at Corporate Secretary, Applied DNA Sciences, Inc.,
50 Health Sciences Drive, Stony Brook, New York 11790 or visiting our website at
http://www.adnas.com
.
Any statement contained in a document incorporated or deemed
to be incorporated by reference in this prospectus shall be deemed to be modified or superseded for the purposes of this prospectus
to the extent that a statement contained herein, any prospectus supplement or in any other subsequently filed document which also
is or deemed to be incorporated by reference herein modifies or supersedes that statement. Any statement so modified or superseded
shall not be deemed, except as so modified or superseded, to constitute a part of this prospectus.
Applied DNA Sciences (NASDAQ:APDN)
Historical Stock Chart
From Apr 2024 to May 2024
Applied DNA Sciences (NASDAQ:APDN)
Historical Stock Chart
From May 2023 to May 2024