Gilead Sciences, Galapagos See Efficacy in Advanced Rheumatoid Arthritis Study
October 10 2019 - 6:10PM
Dow Jones News
By Kimberly Chin
Gilead Sciences Inc. (GILD) and Galapagos NV (GLPG) said an
advanced phase of rheumatoid-arthritis trials proved efficacy after
a 12-month study.
The results from Finch 1 and Finch 3 trials of filgotinib, an
oral inhibitor that treats moderate to severe active rheumatoid
arthritis, proved effective and safe in patients after a one-year
study, the companies said.
Filgotinib hasn't been approved by regulatory authorities yet.
The companies said they are awaiting approval of a marketing
application for filgotinib by the European Medicines Agency, as
well as approval of a new drug application with the Japanese
Ministry of Health. Gilead said it intends to file a new drug
application for the treatment with U.S. regulators later this
year.
Galapagos and Gilead are globally collaborating on the
development and commercialization of filgotinib. The FINCH Phase 3
studies are among several clinical trials of filgotinib for the
treatment of inflammatory diseases.
Write to Kimberly Chin at kimberly.chin@wsj.com
(END) Dow Jones Newswires
October 10, 2019 18:55 ET (22:55 GMT)
Copyright (c) 2019 Dow Jones & Company, Inc.
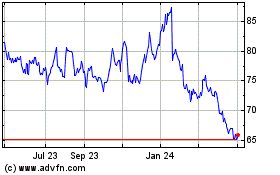
Gilead Sciences (NASDAQ:GILD)
Historical Stock Chart
From Apr 2024 to May 2024
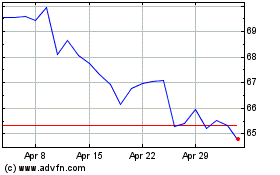
Gilead Sciences (NASDAQ:GILD)
Historical Stock Chart
From May 2023 to May 2024