0001553643
false
0001553643
2023-09-20
2023-09-20
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM
8-K
CURRENT
REPORT
Pursuant
to Section 13 or 15(d) of the Securities Exchange Act of 1934
Date
of Report (Date of earliest event reported): September 20, 2023
RELMADA THERAPEUTICS, INC.
(Exact
name of registrant as specified in its charter)
| Nevada |
|
001-39082 |
|
45-5401931 |
(State
or other jurisdiction
of incorporation) |
|
(Commission
File Number) |
|
(IRS
Employer
Identification No.) |
2222 Ponce de Leon Blvd., Floor 3
Coral Gables, FL |
|
33134 |
| (Address
of principal executive offices) |
|
(Zip
Code) |
Registrant’s
telephone number, including area code: (786) 629 1376
| |
| (Former
name or former address, if changed since last report) |
Check
the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under
any of the following provisions (see General Instruction A.2. below):
| ☐ | Written
communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ☐ | Soliciting
material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ☐ | Pre-commencement
communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ☐ | Pre-commencement
communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities
registered pursuant to Section 12(b) of the Act:
| Title
of each class |
|
Trading Symbol |
|
Name
of each exchange on which registered |
| Common stock, $0.001 par value per share |
|
RLMD |
|
The
NASDAQ Global Select Market |
Indicate
by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405
of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging
growth company ☐
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item
7.01 Regulation FD Disclosure.
On
September 20, 2023, Relmada Therapeutics, Inc. (the “Company”), issued a press release that provided information regarding
efficacy and safety results from the Company’s Phase 3 RELIANCE – OLS (Long-term, Open-label) registrational study for REL-1017
as a treatment for major depressive disorder. Pursuant to Regulation FD, the press release is furnished with this Current
Report as Exhibit 99.1.
The
information set forth in Item 7.01 of this Current Report on Form 8-K and in the attached Exhibit 99.1 is deemed to be “furnished”
and shall not be deemed to be “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the
“Exchange Act”), or otherwise subject to the liabilities of that Section. The information set forth in Item 7.01 of this
Current Report on Form 8-K, including Exhibit 99.1, shall not be deemed incorporated by reference into any filing under the Exchange
Act or the Securities Act of 1933, as amended (the “Securities Act”), regardless of any general incorporation language in
such filing.
Item 9.01 Financial
Statements and Exhibits.
(d)
Exhibits.
| * | This
Exhibit attached to this Form 8-K shall not be deemed “filed” for purposes of Section 18 of the Exchange Act or otherwise
subject to liability under that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act or the
Exchange Act, except as expressly set forth by specific reference in such filing. |
SIGNATURES
Pursuant
to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by
the undersigned hereunto duly authorized.
| Dated:
September 20, 2023 |
RELMADA
THERAPEUTICS, INC. |
| |
|
|
| |
By: |
/s/
Sergio Traversa |
| |
Name: |
Sergio
Traversa |
| |
Title: |
Chief
Executive Officer |
2
Exhibit
99.1
September
20, 2023
Relmada
Therapeutics Announces Efficacy and Safety Results from the Phase 3 Long-Term Study of REL-1017 in Major Depressive Disorder
| ● | Patients
newly treated with REL-1017 for up to one year experienced rapid, clinically meaningful, and sustained improvements in depressive symptoms
and associated functional impairment |
| ● | Long-term
dosing with REL-1017 was well-tolerated, with low rates of adverse events and discontinuations due to adverse events, and no new safety
signals were detected |
CORAL
GABLES, Fla., Sept. 20, 2023 /PRNewswire/ -- Relmada Therapeutics, Inc. (Nasdaq: RLMD), a late-stage biotechnology company addressing
diseases of the central nervous system (CNS), today announced efficacy results for the de novo (or new to treatment) patients (204 patients)
and safety results for all subjects (627 patients) from the Phase 3, long-term, open-label, registrational trial (Study 310) of REL-1017
in patients with Major Depressive Disorder (MDD). Detailed efficacy data for rollover population in the study will be presented when
available.
Patients
treated daily with REL-1017 for up to one year experienced rapid, clinically meaningful, and sustained improvements in depressive symptoms
and associated functional impairment. REL-1017 was well-tolerated with long-term dosing, showing low rates of adverse events and discontinuations
due to adverse events. No new safety signals were detected.
“These
efficacy and safety results represent real-world potential outcomes for MDD patients when treated with REL-1017,” said Cedric
O’Gorman, MD, Chief Medical Officer of Relmada. “The rapid and sustained therapeutic effects achieved with REL-1017
suggest the significant therapeutic potential of this promising late-stage product candidate as a mechanistically novel and
differentiated treatment for MDD. The early magnitude and trajectory of clinical improvement remain consistent across all trials
conducted to date. The long-term sustained clinical improvement, coupled with an extremely well-tolerated profile, adds to our
enthusiasm for this agent as a potential therapeutic option for patients and prescribers.”
Study
REL-1017-310 was a long-term, open-label, non-comparative, registrational Phase 3 trial designed to evaluate the efficacy and safety
of REL-1017 administered once-daily in patients with MDD for up to one year. In total, 627 patients were enrolled, comprising 423 patients
who rolled over (rollover patients/subjects) from placebo-controlled trials with REL-1017 (Studies 301, 302 and 303), and 204 de novo
patients who had not previously participated in trials with REL-1017. The trial was concluded when at least 300 patients had been treated
for six months and approximately 100 patients had been treated for 12 months. At the time of study conclusion, 418 patients had reached
at least six months of treatment, and 118 patients had reached at least 12 months of treatment.
Efficacy
results are presented below for de novo patients only.
Rapid
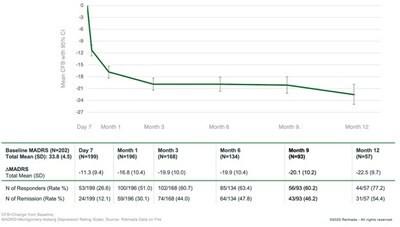
and Sustained Substantial Improvement in MADRS Total Score over Time
In
de novo patients, the mean MADRS total score was 33.8 at baseline. Treatment with REL-1017 in these patients resulted in mean
improvements from baseline in the MADRS total score of 11.3 points at Day 7, 16.8 points at Month 1, 19.9 points at Months 3 and 6,
and 22.5 points at Month 12.
High
Rates of Clinical Response, Both Rapid and Sustained
When
treated with REL-1017, 26.6% of de novo patients achieved clinical response by Day 7, 51.0% by Month 1, 60.7% by Month 3, 63.4% by Month
6, and 77.2% by Month 12. Clinical response is defined as a ≥50% improvement in MADRS total score.
Meaningful
Rates of Clinical Remission
The
virtual absence of depressive symptoms (clinical remission) was achieved by 12.1% of de novo patients at Day 7, 30.1% at Month 1, 44.0%
at Month 3, 47.8% at Month 6, and 54.4% at Month 12. Clinical remission is defined as MADRS total score ≤10.
CGI-I
Scale Also Showed Clinical Improvement
As
assessed by the Clinical Global Impression of Improvement (CGI-I) Scale, de novo treated patients showed meaningful improvements consistent
with MADRS efficacy improvements.
Significant
Reduction in Functional Impairment associated with MDD
Functional
impairment across all three separate domains of the Sheehan Disability Scale was improved with REL-1017 treatment by an average of approximately
50% in de novo patients at Month 6, with further improvement out to Month 12. The Sheehan Disability Scale (SDS) is a widely used, patient-rated
scale that measures functional impairment associated with MDD across three individual domains: work, social life, and family life.
Symptoms
of Anxiety in MDD Reduced with REL-1017 Treatment
The
mean baseline score on the Hamilton Anxiety Rating Scale (HAM-A), which measures symptoms of anxiety, was 20.6 points, reflecting moderate
anxiety. De novo patients treated with REL-1017 saw a continual decline in anxiety symptoms over time as measured by the HAM-A. Score
improvements were 7.1 points at Day 7, 9.6 points at Month 1, 11.1 points at Month 3, 11.5 points at Month 6, and 13.5 points at Month
12.
Long-Term
Safety and Tolerability Results
REL-1017
was safe and well-tolerated with a profile consistent with that observed in short- term controlled trials, with no new safety signals
detected. Safety results presented for Study REL-1017-310 are for all patients (de novo and rollover) in the trial.
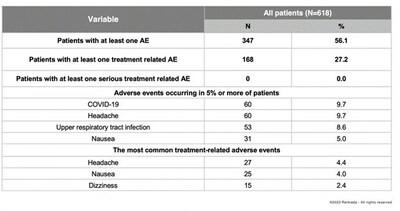
Discontinuations
due to adverse events occurred in approximately 3% of patients during the trial, with no individual adverse event discontinuation
occurring in more than 1% of patients. The most commonly reported adverse events deemed to be treatment-related all occurred at
rates less than 5% and included headache (4.4%), nausea (4.0%) and dizziness (2.4%). The most commonly reported adverse events
(occurring at a rate of ≥5%) were COVID-19 (9.7%), headache (9.7%), upper respiratory tract infection (8.6%), and nausea (5.0%).
The occurrence of these adverse events reflected the study being conducted, in part, during the SARS-CoV pandemic.
Importantly,
there was no significant safety signal for weight gain, sexual dysfunction, cardiovascular issues, dissociative effects, withdrawal phenomena
or abuse liability.
About
REL-1017
REL-1017,
a new chemical entity (NCE) and novel NMDA receptor (NMDAR) channel blocker that preferentially targets hyperactive channels while maintaining
physiological glutamatergic neurotransmission, is currently in late-stage development for the adjunctive treatment of major depressive
disorder (MDD). The ongoing Clinical Research Program is designed to evaluate the potential for REL-1017 as a rapid-acting, oral, once-daily
antidepressant treatment. In addition to the long-term, open-label study of REL-1017, the Phase 3 development program for REL-1017 as
an adjunctive treatment for MDD also includes the recently initiated Relight (Study 304) Phase 3, randomized, double-blind, placebo-controlled
trial and the ongoing Reliance II (Study 302) trial. Relight and Reliance II have the same key study design parameters.
About
Relmada Therapeutics, Inc.
Relmada
Therapeutics is a late-stage biotechnology company addressing diseases of the central nervous system (CNS), with a focus on major depressive
disorder (MDD). Relmada’s experienced and dedicated team is committed to making a difference in the lives of patients and their families.
Relmada’s lead program, REL-1017, is a new chemical entity (NCE) and novel NMDA receptor (NMDAR) channel blocker that preferentially
targets hyperactive channels while maintaining physiological glutamatergic neurotransmission. REL-1017 is in late-stage development as
an adjunctive treatment for MDD in adults. Learn more at www.relmada.com.
Forward-Looking
Statements
The
Private Securities Litigation Reform Act of 1995 provides a safe harbor for forward- looking statements made by us or on our behalf.
This press release contains statements which constitute “forward-looking statements” within the meaning of Section 27A of the
Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Any statement that is not historical in nature is a forward-looking
statement and may be identified by the use of words and phrases such as “expects,” “anticipates,” “believes,”
“will,” “will likely result,” “will continue,” “plans to,” “potential,” “promising,”
and similar expressions. These statements are based on management’s current expectations and beliefs and are subject to a number of risks,
uncertainties and assumptions that could cause actual results to differ materially from those described in the forward-looking statements,
including potential failure of clinical trial results to demonstrate statistically and/or clinically significant evidence of efficacy
and/or safety, failure of top-line results to accurately reflect the complete results of the trial, failure of the 310 open-label study
to accurately reflect the results of the ongoing 302 and 304 blinded, randomized and controlled studies, failure to obtain regulatory
approval of REL-1017 for the treatment of major depressive disorder, and the other risk factors described under the heading “Risk
Factors” set forth in the Company’s reports filed with the SEC from time to time. No forward-looking statement can be guaranteed,
and actual results may differ materially from those projected. Relmada undertakes no obligation to publicly update any forward-looking
statement, whether as a result of new information, future events, or otherwise. Readers are cautioned that it is not possible to predict
or identify all the risks, uncertainties and other factors that may affect future results and that the risks described herein should
not be a complete list.
Investor
Contact:
Tim
McCarthy
LifeSci Advisors
tim@lifesciadvisors.com
Media
Inquiries:
Corporate Communications
media@relmada.com

View
original content to download multimedia:https://www.prnewswire.com/news- releases/relmada-therapeutics-announces-efficacy-and-safety-results-from-the-phase-3-
long-term-study-of-rel-1017-in-major-depressive-disorder-301933333.html
SOURCE
Relmada Therapeutics, Inc.
5
v3.23.3
Cover
|
Sep. 20, 2023 |
| Cover [Abstract] |
|
| Document Type |
8-K
|
| Amendment Flag |
false
|
| Document Period End Date |
Sep. 20, 2023
|
| Entity File Number |
001-39082
|
| Entity Registrant Name |
RELMADA THERAPEUTICS, INC.
|
| Entity Central Index Key |
0001553643
|
| Entity Tax Identification Number |
45-5401931
|
| Entity Incorporation, State or Country Code |
NV
|
| Entity Address, Address Line One |
2222 Ponce de Leon Blvd.
|
| Entity Address, Address Line Two |
Floor 3
|
| Entity Address, City or Town |
Coral Gables
|
| Entity Address, State or Province |
FL
|
| Entity Address, Postal Zip Code |
33134
|
| City Area Code |
786
|
| Local Phone Number |
629 1376
|
| Written Communications |
false
|
| Soliciting Material |
false
|
| Pre-commencement Tender Offer |
false
|
| Pre-commencement Issuer Tender Offer |
false
|
| Title of 12(b) Security |
Common stock, $0.001 par value per share
|
| Trading Symbol |
RLMD
|
| Security Exchange Name |
NASDAQ
|
| Entity Emerging Growth Company |
false
|
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Relmada Therapeutics (NASDAQ:RLMD)
Historical Stock Chart
From Apr 2024 to May 2024
Relmada Therapeutics (NASDAQ:RLMD)
Historical Stock Chart
From May 2023 to May 2024