SINTX Technologies & Iwatani Group Announce Agreement to Develop Antipathogenic Surfaces to Help Prevent Spread of the Common...
November 13 2020 - 8:00AM
Business Wire
SINTX Technologies, Inc. (NASDAQ: SINT) (“SINTX” or the
“Company”) (www.sintx.com), an original equipment manufacturer
(OEM) of silicon nitride ceramic for medical and non-medical
applications, and Iwatani Corporation of America (ICA), a
wholly-owned subsidiary of Iwatani Corporation, announced today
that they have entered into a joint development agreement.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20201113005116/en/
Photo of an example of proposed
applications. (Photo: Business Wire)
The goal of this agreement is to incorporate SINTX AP2 silicon
nitride powder into polymer materials. These materials will become
safe and effective surfaces for IT devices such as cases for cell
phones and tablets that are touched frequently. SINTX and Iwatani
aim to create surfaces that resist bacteria and viruses. The
successful development of these products may help prevent and
reduce the spread of respiratory diseases like SARS-CoV-2,
Influenza A, and H1N1 by inactivating viruses and bacteria on the
polymer surfaces.
“We are pleased to enter this joint development agreement with
Iwatani, toward developing new products that leverage the intrinsic
antipathogenic properties of silicon nitride,” said Dr. Bal. “The
collaboration with Iwatani is intended to directly apply our
material science expertise toward new product development and
revenues, a very significant and exciting move for SINTX.”
SINTX and Iwatani intend to capitalize on recent and promising
data demonstrating the potential of SINTX AP2silicon nitride powder
to inactivate the SARS-CoV-2 virus that is responsible for the
global COVID-19 pandemic. Studies showing the rapid inactivation of
SARS-CoV-2 by SINTX’s silicon nitride are the latest in a series of
papers showing similar activity of silicon nitride against a
variety of bacteria and viral strains. SINTX and Iwatani plan to
jointly fund collaborative product development activities in Salt
Lake City, Utah, and Japan with the support of external
laboratories as well.
SINTX notes they are working diligently to incorporate silicon
nitride into several products such as masks and consumer electronic
products and other surfaces that will help inactivate respiratory
pathogens. The joint development agreement between Iwatani and
SINTX will help SINTX advance their materials science, while
external partners like Iwatani develop commercial products that can
help protect the worldwide community against viral diseases.
About SINTX Technologies, Inc.
SINTX Technologies is an OEM ceramics company that develops and
commercializes silicon nitride for medical and non-medical
applications. The core strength of SINTX Technologies is the
manufacturing, research, and development of silicon nitride
ceramics for external partners. The Company presently manufactures
silicon nitride powders and components in its FDA registered,
AS9100D certified, and ISO 13485:2016 certified manufacturing
facility.
For more information on SINTX Technologies or its silicon
nitride material platform, please visit www.sintx.com.
Forward-Looking Statements
This press release contains forward-looking statements within
the meaning of the Private Securities Litigation Reform Act of 1995
(PSLRA) that are subject to a number of risks and uncertainties.
Risks and uncertainties that may cause such differences include,
among other things, SINTX’s ability to complete its obligations
under the agreement; the collaboration with Iwatani may not result
in the development of any products; that SINTX has not as yet
developed any products with antiviral properties which incorporate
the use of silicon nitride; products developed under the joint
development agreement may not be effective against the SARS-CoV-2
virus; incorporation of silicon nitride into personal protective
equipment may not be safe or effective; volatility in the price of
SINTX’s common stock; the uncertainties inherent in new product
development, including the cost and time required to commercialize
such product(s); market acceptance of our products once
commercialized; SINTX’s ability to raise additional funding and
other competitive developments. Readers are cautioned not to place
undue reliance on the forward-looking statements, which speak only
as of the date on which they are made and reflect management’s
current estimates, projections, expectations and beliefs. There can
be no assurance that any of the anticipated results will occur on a
timely basis or at all due to certain risks and uncertainties, a
discussion of which can be found in SINTX’s Risk Factors disclosure
in its Annual Report on Form 10-K, filed with the Securities and
Exchange Commission (SEC) on March 26, 2020, and in SINTX’s other
filings with the SEC. SINTX disclaims any obligation to update any
forward-looking statements. SINTX undertakes no obligation to
publicly revise or update the forward-looking statements to reflect
events or circumstances that arise after the date of this
report.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20201113005116/en/
Business Inquiries for SINTX: SINTX Technologies
801.839.3502 IR@sintx.com
Media Inquiries for SINTX: Amanda Barry Associate
Director, Content and PR The Summit Group abarry@summitslc.com
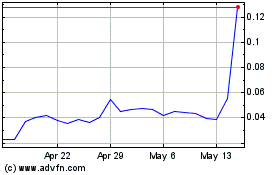
SiNtx Technologies (NASDAQ:SINT)
Historical Stock Chart
From Apr 2024 to May 2024
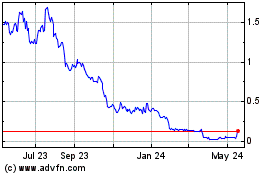
SiNtx Technologies (NASDAQ:SINT)
Historical Stock Chart
From May 2023 to May 2024