Tilray® Exports 2:100 CBD Oil to the United Kingdom
September 11 2018 - 7:00AM
Business Wire
Tilray is supplying medical cannabis to pediatric patient in
need
Tilray Inc. (NASDAQ:TLRY), a global leader in cannabis research,
production and distribution, announced today that Tilray 2:100, a
product produced at Tilray’s federally licensed, Good Manufacturing
Practice (GMP)-certified facility in Canada, was successfully
imported into the United Kingdom, to supply a pediatric patient in
need.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20180911005234/en/
Tilray 2:100 (Photo: Business Wire)
The changing regulatory measures in the United Kingdom (UK)
previously prohibited the use of medical cannabis. However, in July
2018, Home Secretary Sajid Javid announced that clinicians will now
legally be able to prescribe cannabis-derived medicines to patients
with an exceptional clinical need noting “recent cases involving
sick children made it clear to me that our position on
cannabis-related medicinal products was not satisfactory.”
With government-approved compassionate access, the UK-based
patient suffering from a rare form of epilepsy, now has reliable
access to Tilray 2:100, the product used as the primary medicine in
a clinical trial testing the efficacy and tolerability of
cannabis-derived medicine as treatment for pediatric epilepsy. The
trial was conducted in partnership with The Hospital For Sick
Children (SickKids) in Toronto, Canada and published promising
results in August.
In a press release issued by SickKids, Dr. Blathnaid McCoy,
Staff Neurologist at SickKids who conducted the clinical trial and
authored the research, said “In the participants who reached the
target treatment dose we saw a statistically significant reduction
in motor seizures, and an increase in seizure-free days compared to
those who did not reach the target dose. We observed promising
clinically beneficial effects including a reduction in seizure
frequency and improvements in certain aspects of adaptive
functioning and quality of life measures.”
The Tilray oral solution imported to the United Kingdom contains
100 mg/ml cannabidiol (CBD) and 2mg/ml tetrahydrocannabinol (THC),
the same ratio of active cannabinoids Tilray used in the recent
clinical trial led by SickKids. This ratio of the active
cannabinoids was selected after Tilray conducted extensive research
on published data of cannabinoids to treat seizures. Tilray 2:100
was deemed safe and well tolerated in pediatric patients by the
researchers.
About Tilray®Tilray is a global pioneer in the
research, cultivation, production and distribution of cannabis and
cannabinoids currently serving tens of thousands of patients in
eleven countries spanning five continents.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20180911005234/en/
For TilrayMedia:US & CanadaChrissy Roebuck,
+1-416-560-5712chrissy.roebuck@tilray.comorUKJustin Doherty, +44
(0)7768-330223jd@hemingtonconsulting.co.ukorInvestors:Katie Turner,
+1-646-277-1228Katie.turner@icrinc.com
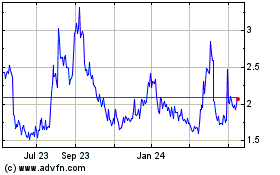
Tilray Brands (NASDAQ:TLRY)
Historical Stock Chart
From Apr 2024 to May 2024
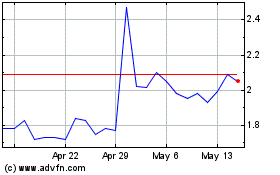
Tilray Brands (NASDAQ:TLRY)
Historical Stock Chart
From May 2023 to May 2024