BD Voluntarily Recalling Intraosseous Products
June 22 2022 - 4:17PM
Dow Jones News
By Stephen Nakrosis
Becton, Dickinson and Co. said Wednesday it was voluntarily
recalling certain BD Intraosseous Needle Set Kits, BD Intraosseous
Manual Driver Kits and BD Intraosseous Powered Drivers.
The company said there are no replacement products currently,
adding it will notify customers when replacement products become
available. "BD recommends that customers evaluate their clinical
needs and consider obtaining and using an alternative intraosseous
product," the company said.
The company said users of certain lots may have difficulty
separating the stylet from the intraosseous needle, which could
result in functional loss of intraosseous access. Other issues
include "needle safety mechanism on the stylet not deploying post
placement of the intraosseous needle and removal of the stylet from
the needle," and "metal discs intended to connect the needle
assembly to the magnet in the powered driver sticking unexpectedly
to the magnet, rendering the driver unable to be used," the company
said.
"These issues may result in delays in care due to limited or
non-functioning intraosseous access or could also lead to
needlestick injuries," according to the company.
The company said additional information is available on the
recall notification section of BD.com.
Write to Stephen Nakrosis at stephen.nakrosis@wsj.com
(END) Dow Jones Newswires
June 22, 2022 17:02 ET (21:02 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
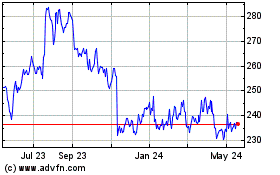
Becton Dickinson (NYSE:BDX)
Historical Stock Chart
From Apr 2024 to May 2024
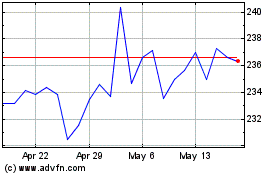
Becton Dickinson (NYSE:BDX)
Historical Stock Chart
From May 2023 to May 2024