Roche's Gantenerumab Gets FDA Breakthrough Therapy Designation for Alzheimer's Disease
October 08 2021 - 11:31AM
Dow Jones News
By Mauro Orru
The U.S. Food and Drug Administration has granted breakthrough
therapy designation to Roche Holding AG's gantenerumab for the
treatment of people living with Alzheimer's disease.
The Swiss pharmaceutical major said Friday that the FDA's
decision is based on data showing that gantenerumab, an antibody
developed for subcutaneous administration, significantly reduced
brain amyloid plaque, a pathological hallmark of Alzheimer's
disease.
The designation aims to accelerate the development and review of
medicines to treat serious or life-threatening conditions.
"For more than a decade, we've been committed to advancing the
science of Alzheimer's as well as our investigational medicine
gantenerumab, and we look forward to delivering a comprehensive and
robust data set that furthers our collective understanding of this
devastating disease," said Levi Garraway, Roche's chief medical
officer and head of global product development.
"This Breakthrough Therapy Designation reinforces our confidence
in gantenerumab, which would be the first subcutaneous medicine for
the treatment of Alzheimer's disease with the potential for at-home
administration," Mr. Garraway said.
Gantenerumab is in Phase 3 development, with trials assessing
the antibody in more than 2,000 participants for more than two
years that are expected to be completed in the second half of next
year.
Write to Mauro Orru at mauro.orru@wsj.com; @MauroOrru94
(END) Dow Jones Newswires
October 08, 2021 12:16 ET (16:16 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
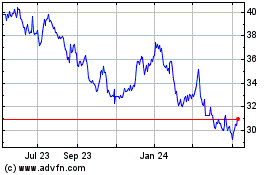
Roche (QX) (USOTC:RHHBY)
Historical Stock Chart
From Apr 2024 to May 2024
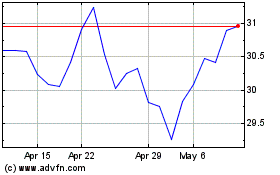
Roche (QX) (USOTC:RHHBY)
Historical Stock Chart
From May 2023 to May 2024