Bruker Introduces the MALDI SepsityperTM Kit for Rapid MALDI Biotyper Microbial Identification from Positive Blood Cultures
September 13 2010 - 6:00AM
Business Wire
At the 50th Interscience Conference on Antimicrobial Agents and
Chemotherapy (ICAAC), Bruker Daltonics Inc. today announced the
introduction of the new MALDI SepsityperTM kit for
the rapid identification of microorganisms from positive blood
culture bottles, using Bruker’s industry-leading MALDI Biotyper
solution. The MALDI Biotyper has obtained the IVD-CE Mark, and is
currently for Research Use Only (RUO) outside of the European
Union.
The MALDI Sepsityper kit is a set of consumables that
enables a rapid and easy identification workflow after the blood
culture bottle has tested positive. Microbial cells are harvested
from an aliquot of blood culture, are processed using the MALDI
Sepsityper kit, and are then identified by the MALDI Biotyper
workflow. The identification assay from positive blood cultures is
applicable to gram-negative bacteria, gram-positive bacteria, and
importantly also to yeasts. Only a few minutes after taking an
aliquot from the blood culture bottle, a sample can be ready for
mass spectrometry-based molecular identification by the MALDI
Biotyper. This workflow is recommended and optimized for blood
culture bottles without any charcoal supplements, because such
supplements would need to be removed first by additional sample
preparation steps.
By using the new MALDI Sepsityper product in combination
with the MALDI Biotyper, in approximately 70-90% of the tested
positive blood culture samples, a high-confidence identification
result is available much earlier than with conventional technology,
typically saving one day (or more) in time-to-result (TTR) for the
crucial identification step. The MALDI Sepsityper kit and
MALDI Biotyper identification rapid blood-culture-to-ID workflow
did not lead to any false positives during initial testing and
validation. Bruker is planning further MALDI Sepsityper kit
validation studies with an expanded number of European clinical
microbiology laboratories processing blood culture samples.
Dr. S�ren Schubert, Assistant Medical Director at the
Max-von-Pettenkofer Institute in Munich, Germany, stated: “We have
tested the MALDI Sepsityper kit now for several months in
our laboratories and we are more than pleased with the results. The
kit is easy to handle, integrated with our MALDI Biotyper approach,
and in most cases gives the identification from a positive blood
culture significantly earlier, when previously with conventional
techniques, we only had the Gram status and the morphology of the
microbes. With the MALDI Sepsityper we can expand the
advantages of the MALDI Biotyper to yet another very important
field of early microbial identification.”
Dr. Wolfgang Pusch, Vice President of Bruker Daltonics,
commented: “Recently, there have been numerous presentations by
leaders in the field of microbiology at scientific symposia
concerning the rapid direct identification of microbes isolated
from positive blood culture bottles with the MALDI Biotyper system,
demonstrating the high demand for this rapid new
blood-culture-to-ID workflow. With over 150 MALDI Biotyper systems
installed at customer sites, we believe that the new MALDI
Sepsityper kit has the potential to make the MALDI Biotyper
even more broadly applicable and useful in clinical microbiology.
The earliest possible microbial identification and faster TTR are
of particular interest for blood cultures, where conventional
techniques typically need at least one day longer for
identification.”
The MALDI Sepsityper kit is presently for research use
only. Not for use in diagnostic procedures.
For More Information:
For more information on MALDI Sepsityper, please visit
www.bdal.com
For information about Bruker Daltonics and Bruker Corporation
(NASDAQ: BRKR), please visit www.bruker.com
Bruker (NASDAQ:BRKR)
Historical Stock Chart
From Jun 2024 to Jul 2024
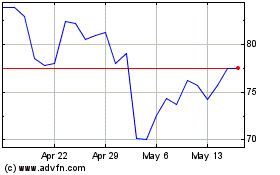
Bruker (NASDAQ:BRKR)
Historical Stock Chart
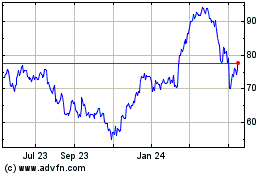
From Jul 2023 to Jul 2024