iRhythm Technologies Gets FDA Clearance for ZEUS Software System
July 22 2022 - 8:14AM
Dow Jones News
By Chris Wack
iRhythm Technologies Inc. said Friday it received FDA 510(k)
clearance for its Zio ECG Utilization Software System for the Zio
Watch.
The ZEUS system is produced in partnership with Verily, an
Alphabet precision health company.
The ZEUS System is the AI algorithm and solution component of
the Zio Watch, a sensor-based wearable for noninvasive, clinical
grade, long-term continuous monitoring for atrial fibrillation.
Verily also received FDA 510(k) clearance for the Zio Watch.
iRhythm plans to introduce the ZEUS System for a limited market
evaluation in 2023.
Write to Chris Wack at chris.wack@wsj.com
(END) Dow Jones Newswires
July 22, 2022 08:59 ET (12:59 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
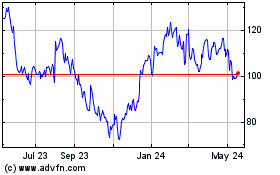
iRhythm Technologies (NASDAQ:IRTC)
Historical Stock Chart
From Apr 2024 to May 2024
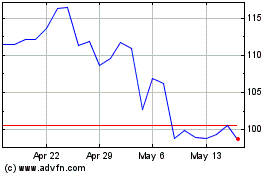
iRhythm Technologies (NASDAQ:IRTC)
Historical Stock Chart
From May 2023 to May 2024