Bruker Corporation and Lawrence Berkeley National Laboratory
(Berkeley Lab) today announced a collaboration to develop and
distribute new structural biology methods and tools to integrate
Small-Angle X-ray Scattering (SAXS) with Nuclear Magnetic Resonance
(NMR). The goal of this collaboration is to develop a set of
integrated SAXS and NMR data analysis algorithms for determining
the structures of larger multi-domain proteins and protein
complexes with DNA, RNA or other proteins. Such multi-modality
approaches based on complementary analytical technologies play a
key role in helping researchers answer increasingly complex
questions in structural biology and drug development, and hold the
potential for advancements in clinical research applications.
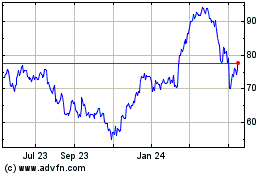
The SAXS Pair Distance Function (PDF) of
Urate Oxidase from Aspergillus Flavus (PDB 1R56) describes the
radial distribution of density in the molecule which provides
global constraints on the overall size and shape of the molecule
for the local NMR coordinate measurements. (Photo: Business
Wire)
Traditional NMR three-dimensional (3D) atomic structure
determination of the individual protein domains will be combined
and integrated with the determination of overall size, shape and
envelope constraints provided by SAXS. This approach will yield
more accurate structures of larger multi-domain proteins and
complexes under near-native solution conditions than what can be
solved currently by NMR alone. Importantly, the integrated NMR and
SAXS approach has been shown to help in the elucidation of
important functional information about intrinsically flexible,
unstructured, or partially unfolded domains.
“Hybrid methods are going to be essential for solving structures
of larger biomolecules and biomolecular complexes,” stated Dr. John
Markley, Steenbock Professor of Biomolecular Structure, and Head of
the National Magnetic Resonance Facility at Madison (NMRFAM) at the
University of Wisconsin-Madison.
Protein structure determination is crucial for a broad range of
applications from fundamental biological research to
next-generation drug development. High magnetic field NMR is unique
in its capability to study the detailed structure and dynamics of
proteins in solution, the native environment of many proteins. This
allows NMR to elucidate the structures of proteins with flexible
domains or multiple configurations, features that are often not
directly accessible with static techniques such as crystallography
or electron microscopy. However, typical NMR structures are not as
accurate as the best protein structures from X-ray crystallography,
and currently the use of NMR for protein structure determination in
solution has its upper size limits typically near 50–70 kDa
proteins.
Approaching this limit already requires the use of all modern
NMR techniques, such as ultra-high field 800-1000 MHz magnets,
isotopic labeling schemes, advanced pulse sequences and NMR
electronics, and highest sensitivity CryoProbes™. Even with all
these capabilities, solution NMR often lacks the ability to
determine the exact global structure of larger molecular assemblies
or multi-domain proteins. NMR has significant advantages in that it
can study proteins in solution near native physiological
conditions, can obtain dynamic information for different regions
and domains of a protein, and allow access to functionally very
important information on protein flexibility, intrinsically
unstructured regions, partial protein folding, and in some cases
multiple accessible protein conformations.
Solution-structure determination by NMR uses interatomic
distances determined from the Nuclear Overhauser Effect (NOE) and
torsion angles determined from Residual Dipolar Coupling (RDC). In
theory, a complete set of NMR measurements can be used to uniquely
determine the structures of labeled proteins. However, in practice
many NMR measurements provide sparse RDC datasets that cannot be
used to uniquely determine a structure, especially for larger
proteins. SAXS is an ideal complementary technique that can
efficiently and effectively compensate incomplete NMR datasets of
biological macromolecules. It has been shown by a number of
researchers that SAXS data can improve the quality and accuracy of
NMR structures and also potentially extends the capability of NMR
to larger macromolecules (e.g., J. Wang et al., “Determination of
multi-component protein structures in solution using global
orientation and shape restraints*”).
“NMR and SAXS provide truly complementary data: detailed local
conformations are derived from NMR and global shape from SAXS.
Thus, they naturally go together,” said Dr. Angela Gronenborn, Head
of the Department of Structural Biology at the University of
Pittsburgh School of Medicine and holder of the UPMC Rosalind
Franklin Chair.
An experimental SAXS data set consists of intensities measured
at varying scattering angles. Each measured SAXS intensity
represents the mathematical transform of the protein’s
pair-distance distribution function, i.e. a set of all distances
internal to the protein. SAXS can therefore provide a complete
global snapshot of the protein in solution that can be used as a
constraint to resolve ambiguities during the determination of a
protein structure by NMR, or SAXS data can help discriminate
between similar structural conformations.
SAXS is an attractive adjunct to NMR because the experiment is
relatively fast, economical and straightforward: it requires no
sophisticated sample preparation, data can be acquired fully
automatically with only a few micrograms of unlabeled, native
protein in a monodisperse solution, and Bruker offers powerful and
highly automated SAXS laboratory systems for structural biologists
who do not have access to a dedicated synchrotron beamline. A
complete SAXS dataset can typically be acquired in hours in the
homelab, or in minutes at a beamline, without any significant
difference in ultimate data quality or information content, and
there is a growing set of powerful tools for SAXS data
interpretation, for example the ATSAS suite from the EMBL†.
Leading the Bruker-Berkeley Lab structural biology collaboration
project will be Professor John Tainer from the Scripps Research
Institute and Dr. Robert Rambo of Berkeley Lab, both
internationally recognized for their development of advanced
techniques for SAXS analysis of macromolecules at the SIBYLS
beamline at the Advanced Light Source of Berkeley Laboratory.
Professor Tainer commented: “We are excited to work with Bruker
to develop powerful new tools for the structural analysis of
biological macromolecules. The unique combination of NMR and SAXS
expertise at Bruker and LBNL will provide an ideal environment for
the development of enhanced capabilities for integrated NMR and
SAXS structural analyses.”
This work will be supported by Bruker Corporation under contract
WF008609 and the United States Department of Energy (DOE) program
Integrated Diffraction Analysis Technologies (IDAT) under contract
DE-AC02-05CH11231.
*J. Wang, X. Zuo, P. Yu, I-J, Byeon, J. Jung, X. Wang, M. Dyba,
S. Seifert, C.D. Schwieters, J. Qin, A. Gronenborn and Y-X Wang, J.
A. Chem. Soc., 2009, 131(30), 10507-10515.
**
http://www.bruker-axs.de/fileadmin/user_upload/KJR_Files/Bruker_AXS_BioSAXS_Webinar.pdf
†D. Franke , D.I Svergun, J. Appl. Cryst., 2009, 42,
342-346.
About Bruker Corporation: Bruker Corporation (NASDAQ:
BRKR) is a leading provider of high-performance scientific
instruments and solutions for molecular and materials research, as
well as for industrial and applied analysis. For more information,
please visit www.bruker.com.
About Lawrence Berkeley National Laboratory: Lawrence
Berkeley National Laboratory addresses the world’s most pressing
scientific challenges by advancing sustainable energy, protecting
human health, creating new materials, and revealing the origin and
fate of the universe. Founded in 1931, Berkeley Lab’s scientific
expertise has been recognized with 12 Nobel prizes. The University
of California manages Berkeley Lab for the U.S. Department of
Energy’s Office of Science. For more, visit
http://www.lbl.gov/.
Photos/Multimedia Gallery Available:
http://www.businesswire.com/cgi-bin/mmg.cgi?eid=50063745&lang=en
Bruker (NASDAQ:BRKR)
Historical Stock Chart
From Jun 2024 to Jul 2024
Bruker (NASDAQ:BRKR)
Historical Stock Chart
From Jul 2023 to Jul 2024