Dr. Reddy's Laboratories Announces the Launch of Progesterone Capsules in the U.S. Market
April 21 2017 - 10:40AM
Business Wire
Dr. Reddy’s Laboratories Ltd (BSE: 500124, NSE: DRREDDY, NYSE:
RDY) announced today that it has launched Progesterone Capsules,
100 mg and 200 mg, a therapeutic equivalent generic version of
Prometrium® (Progesterone) Capsules in the United States market,
approved by the U.S. Food and Drug Administration (USFDA).
The Prometrium® brand and generic had U.S. sales of
approximately $153 million MAT for the most recent twelve months
ending in February 2017 according to IMS Health*.
Dr. Reddy’s Progesterone Capsules, 100 mg and 200 mg, are
available in bottle count size of 100.
Prometrium® is a registered trademark of ABBVIE Products
LLC.
*IMS National Sales Perspective: Retail and Non-Retail MAT Feb
2017RDY-0317-149
WARNING: CARDIOVASCULAR DISORDERS,
BREAST CANCER and PROBABLE DEMENTIA
FOR ESTROGEN PLUS PROGESTIN
THERAPY
Cardiovascular Disorders and Probable
Dementia
Estrogens plus progestin therapy should not be used for the
prevention of cardiovascular disease or dementia. The
Women’s Health Initiative (WHI) estrogen plus progestin substudy
reported increased risks of deep vein thrombosis, pulmonary
embolism, stroke and myocardial infarction in postmenopausal women
(50 to 79 years of age) during 5.6 years of treatment with daily
oral conjugated estrogens (CE) [0.625 mg] combined with
medroxyprogesterone acetate (MPA) [2.5 mg], relative to placebo.
The WHI Memory Study (WHIMS) estrogen plus progestin
ancillary study of the WHI reported an increased risk of developing
probable dementia in postmenopausal women 65 years of age or older
during 4 years of treatment with daily CE (0.625 mg) combined with
MPA (2.5 mg), relative to placebo. It is unknown whether this
finding applies to younger postmenopausal women.
Breast Cancer
The WHI estrogen plus progestin substudy also demonstrated an
increased risk of invasive breast cancer. In the absence of
comparable data, these risks should be assumed to be similar for
other doses of CE and MPA, and other combinations and dosage forms
of estrogens and progestins. Progestins with estrogens
should be prescribed at the lowest effective doses and for the
shortest duration consistent with treatment goals and risks for the
individual woman.
About Dr. Reddy’s: Dr. Reddy’s Laboratories Ltd. (BSE:
500124, NSE: DRREDDY, NYSE: RDY) is an integrated pharmaceutical
company, committed to providing affordable and innovative medicines
for healthier lives. Through its three businesses - Pharmaceutical
Services & Active Ingredients, Global Generics and Proprietary
Products – Dr. Reddy’s offers a portfolio of products and services
including APIs, custom pharmaceutical services, generics,
biosimilars and differentiated formulations. Our major therapeutic
areas of focus are gastrointestinal, cardiovascular, diabetology,
oncology, pain management and dermatology. Dr. Reddy’s operates in
markets across the globe. Our major markets include – USA, India,
Russia & CIS countries, and Europe. For more information, log
on to: www.drreddys.com
Disclaimer: This press release may include statements of
future expectations and other forward-looking statements that are
based on the management’s current views and assumptions and involve
known or unknown risks and uncertainties that could cause actual
results, performance or events to differ materially from those
expressed or implied in such statements. In addition to statements
which are forward-looking by reason of context, the words "may",
"will", "should", "expects", "plans", "intends", "anticipates",
"believes", "estimates", "predicts", "potential", or "continue" and
similar expressions identify forward-looking statements. Actual
results, performance or events may differ materially from those in
such statements due to without limitation, (i) general economic
conditions such as performance of financial markets, credit
defaults, currency exchange rates, interest rates, persistency
levels and frequency / severity of insured loss events, (ii)
mortality and morbidity levels and trends, (iii) changing levels of
competition and general competitive factors, (iv) changes in laws
and regulations and in the policies of central banks and/or
governments, (v) the impact of acquisitions or reorganization,
including related integration issues.
The company assumes no obligation to update any information
contained herein.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20170421005550/en/
Dr. Reddy's Laboratories Ltd.INVESTOR RELATIONSSaunak
Savla, +91-40-49002135saunaks@drreddys.comorMEDIA
RELATIONSCalvin Printer,
+91-40-49002121calvinprinter@drreddys.com
Dr Reddys Laboratories (NYSE:RDY)
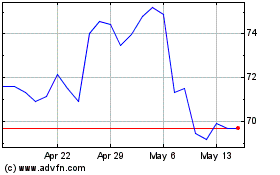
Historical Stock Chart
From Apr 2024 to May 2024
Dr Reddys Laboratories (NYSE:RDY)
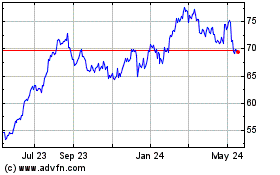
Historical Stock Chart
From May 2023 to May 2024