MaxCyte and Wugen Sign Strategic Platform License to Expedite Scale-Up of Clinical and Commercial Manufacturing of Wugen’s Investigational Allogeneic, Off-the-Shelf Cell Therapies for Cancers
January 30 2024 - 7:05AM
UK Regulatory
ROCKVILLE, Md. and ST. LOUIS, Jan. 30, 2024 (GLOBE
NEWSWIRE) -- MaxCyte, Inc., (Nasdaq: MXCT; LSE: MXCT), a leading
cell-engineering focused company providing enabling platform
technologies to advance the discovery, development and
commercialization of next-generation cell-based therapeutics and
innovative bioprocessing applications, and Wugen, Inc., a
clinical-stage biotechnology company developing allogeneic,
off-the-shelf cell therapies to treat a broad range of
hematological and solid tumor malignancies, today announced the
signing of a strategic platform license (SPL).
Under the terms of the agreement, Wugen gains non-exclusive
clinical and commercial rights to use MaxCyte’s Flow
Electroporation® technology and ExPERT™ platform. In
return, MaxCyte will receive annual license fees and
program-related revenue.
Wugen is developing the next generation of off-the-shelf memory
natural killer (NK) and CAR-T cell therapies for cancer. The
company’s investigational cell therapies originate from healthy
donors and are further engineered to enhance their function of
eliminating cancer cells. Wugen’s lead program is evaluating
WU-CART-007, a CD7 targeted CAR-T cell therapy, in a global Phase
1/2 clinical trial for the treatment of relapsed or refractory
(R/R) T-cell acute lymphoblastic leukemia (T-ALL)/lymphoblastic
lymphoma (LBL) in adolescent and adult patients. WU-CART-007 has
received Orphan Drug, Fast Track, and Rare Pediatric Disease
Designations from the U.S. Food and Drug Administration for the
treatment of R/R T-ALL/LBL. Additional information is available on
clinicaltrials.gov, identifier NCT# 04984356.
“As WU-CART-007 is transferred onto the ExPERT platform and
continues to progress in the clinic, we are able to support
efficient and seamless expansion to large-scale manufacturing,”
said Maher Masoud, President and CEO of MaxCyte.
“By working with MaxCyte, Wugen can maximize the number of doses in
each manufacturing run for later stage clinical development and
potential commercial phase.”
“Our partnership with MaxCyte is an exciting step in advancing
our groundbreaking therapies for difficult-to-treat cancers,”
said Kumar Srinivasan, PhD, MBA, President and CEO of
Wugen. “This collaboration brings us even closer to
delivering life-saving treatments to patients with limited options.
With MaxCyte's expertise, we're in a position to expedite progress
through crucial development stages and make a significant impact on
patients’ lives.”
MaxCyte’s ExPERT™ instrument portfolio represents the next
generation of clinically validated electroporation technology for
complex and scalable cell engineering. Offering high transfection
efficiency, seamless scalability, and enhanced functionality, the
ExPERT™ platform is crucial for enabling the next wave of
biological and cellular therapeutics. Wugen is MaxCyte’s
26th clinical/commercial partnership overall, each of
which generates pre-commercial milestone revenue, the vast majority
of which includes sales-based payments.
About MaxCyte
At MaxCyte, we pursue cell engineering excellence to maximize
the potential of cells to improve patients’ lives. We have spent
more than 20 years honing our expertise by building best-in-class
platforms, perfecting the art of the transfection workflow, and
venturing beyond today’s processes to innovate tomorrow’s
solutions. Our ExPERT™ platform, which is based on our Flow
Electroporation® technology, has been
designed to support the rapidly expanding cell therapy market and
can be utilized across the continuum of the high-growth cell
therapy sector, from discovery and development through
commercialization of next-generation, cell-based medicines. The
ExPERT family of products includes: four instruments, the ATx™,
STx™, GTx™ and VLx ™; a portfolio of proprietary related processing
assemblies or disposables; and software protocols, all supported by
a robust worldwide intellectual property portfolio. By providing
our partners with the right technology platform, as well as
scientific, technical and regulatory support, we aim to guide them
on their journey to transform human health. Learn more at
maxcyte.com and follow us on X (formerly Twitter) and LinkedIn.
About Wugen
Wugen, Inc. is a clinical-stage biotechnology company developing
the next generation of off-the-shelf memory natural killer (NK) and
CAR-T cell therapies for cancer. Wugen is leveraging its
proprietary Moneta™ platform and deep genomic engineering
expertise to pioneer a new class of memory NK cell therapies to
treat hematological and solid tumor malignancies. For more
information, please visit https://wugen.com/
MaxCyte Contacts:
US IR Adviser
Gilmartin Group
David Deuchler, CFA
+1 415-937-5400
ir@maxcyte.com
US Media Relations
Spectrum Seismic Collaborative
Valerie Enes
+1 408-497-8568
venes@spectrumscience.com
Nominated Adviser and Joint Corporate
Broker
Panmure Gordon
Emma Earl / Freddy Crossley
Corporate Broking
Rupert Dearden
+44 (0)20 7886 2500
UK IR Adviser
ICR Consilium
Mary-Jane Elliott
Chris Welsh
+44 (0)203 709 5700
maxcyte@consilium-comms.com
Wugen Contacts:
Investor Contact
Mark Lewis
+1 314-501-1968
Mlewis@wugen.com
Media Contact
Christine Fanelle
Scient PR
+1 215-595-5211
christine@scientpr.com


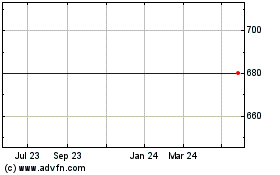
Maxcyte (LSE:MXCN)
Historical Stock Chart
From Aug 2024 to Sep 2024
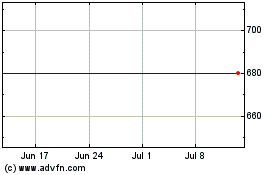
Maxcyte (LSE:MXCN)
Historical Stock Chart
From Sep 2023 to Sep 2024