
EXHIBIT 10.1 EXECUTION VERSION CERTAIN INFORMATION CONTAINED IN THIS DOCUMENT, MARKED BY BRACKETS, HAS BEEN OMITTED BECAUSE THE INFORMATION IS (I) NOT MATERIAL AND (II) TREATED AS PRIVATE OR CONFIDENTIAL BY THE COMPANY. 1 AMENDED AND RESTATED LICENSE AGREEMENT This Amended and Restated License Agreement (“Agreement”), made as of December 11, 2023 (the “Amended Effective Date”), is by and between Arrowhead Pharmaceuticals, Inc., a Delaware corporation with a place of business at 177 East Colorado Boulevard, Suite 700, Pasadena, California 91105, USA (“Arrowhead”), and GlaxoSmithKline Intellectual Property (No. 3) Limited, a company existing under the laws of England, with offices at 980 Great West Road, Brentford, Middlesex, TW8 9GS (“GSK”). Arrowhead and GSK are at times referred to herein individually as a “Party” and collectively as the “Parties”. RECITALS WHEREAS, Arrowhead possesses certain information, materials, and intellectual property rights relating to oligonucleotides and oligonucleotide constructs, including the Licensed Construct in clinical development known as ARO-HBV, which inhibits expression of the hepatitis B virus (“HBV”); WHEREAS, GSK, directly and through certain of its Affiliates, has extensive experience and expertise in the development and commercialization of pharmaceutical and biologic products, and owns or otherwise controls information, materials and intellectual property rights useful to Develop and Commercialize pharmaceutical and biologic products in the Field; WHEREAS, Arrowhead and Janssen Pharmaceuticals, Inc. (“Janssen”) entered into that certain License Agreement, dated as of October 3, 2018 (the “Original Execution Date”), as amended pursuant to (a) that certain Amendment No. 1 to License Agreement, by and between Arrowhead and Janssen, dated as of December 18, 2018; and (b) that certain Amendment No. 2 to License Agreement, by and between Arrowhead and Janssen, dated as of February 4, 2019 (collectively, the “Original Agreement”), pursuant to which, among other things, Arrowhead granted to Janssen an exclusive license under Arrowhead Intellectual Property to Develop and Commercialize Licensed Constructs and Licensed Products as set forth therein; WHEREAS, GSK and Janssen have entered into that certain Asset Purchase Agreement, dated as of October 30, 2023 (the “APA”), pursuant to which, among other things, effective as of the Closing (as defined in the APA), Janssen, with Arrowhead’s consent, has assigned all of its rights, title and interest in, under and to the Original Agreement and certain other Purchased Assets (as defined in the APA) to GSK; and WHEREAS, in accordance with Section 17.6 of the Original Agreement, the Parties now desire to amend and restate the Original Agreement into this Agreement, effective as of the Amended Effective Date upon the terms and subject to the conditions set forth herein. NOW, THEREFORE, in consideration of the mutual promises and covenants set forth below and other good and valuable consideration, the receipt and sufficiency of which is hereby acknowledged, the Parties agree as follows:

2 ARTICLE I: DEFINITIONS Unless the context otherwise requires, the terms in this Agreement with initial letters capitalized shall have the meanings described below or the meaning as designated in the indicated places throughout this Agreement. 1.1 “Access Territory” means those countries set forth in Schedule 1.1. 1.2 “Accounting Standards” means GAAP or IFRS, as appropriate, as generally and consistently applied in compliance with Applicable Laws throughout the relevant company’s organization at the relevant time. 1.3 “Acquired Business” has the meaning set forth in Section 12.3. 1.4 “Acquired General Arrowhead Patent Rights” has the meaning as set forth in Section 10.2.3. 1.5 “Acquired Specific Arrowhead Patent Rights” has the meaning as set forth in Section 10.2.3. 1.6 “Acquirer” means, collectively, the Third Party referenced in the definition of Change of Control and such Third Party’s Affiliates, other than the applicable Party in the definition of Change of Control and such Party’s Affiliates, determined as of immediately prior to the closing of such Change of Control. 1.7 “Action” means any claim, action, cause of action or suit (whether in contract or tort or otherwise), litigation (whether at law or in equity, whether civil or criminal), controversy, assessment, arbitration, investigation, hearing, charge, complaint, demand, notice or proceeding of, to, from, by or before any Governmental Authority. 1.8 “Active Ingredient” means a clinically-active material that provides a pharmacological activity in a pharmaceutical or biologic product (excluding formulation components, such as coatings, stabilizers, excipients or solvents, adjuvants or controlled release technologies). 1.9 “Affiliate” means, with respect to a designated Party or entity, any entity controlling, controlled by, or under common control with such Party or entity. For purposes of this definition only, “control” means: (a) where the entity is a corporate entity, direct or indirect ownership of 50% or more of the stock or shares having the right to vote for the election of directors of such entity; and (b) where the entity is other than a corporate entity, the possession, directly or indirectly, of the power to direct, or cause the direction of, the management or policies of such entity, whether through the ownership of voting securities, by contract or otherwise. For clarity, an entity may be or become an Affiliate of a Party or another entity and may cease to be an Affiliate of such Party or other entity, in each case, during the Term. The Parties acknowledge that, for the purposes of this Agreement, Visirna Therapeutics, Inc. shall not be an Affiliate of Arrowhead. 1.10 “Agreement” has the meaning set forth in the preamble above.

3 1.11 “Agreement IP” means, collectively, the Agreement Know-How and the Agreement Patent Rights. 1.12 “Agreement Know-How” means any and all Know-How that is first discovered, developed, generated, invented, derived, created, conceived or reduced to practice during the Term by or on behalf of one (1) or more Personnel of a Party (or any of its Affiliates, licensees, sublicensees or subcontractors), either alone or jointly with one (1) or more Personnel of the other Party (or any of its Affiliates, licensees, sublicensees or subcontractors), in each case, in the performance of activities under this Agreement, including the Existing Inventions and the New Arising Know-How. For clarity, for purposes of this definition solely, where “Party” is GSK it shall include Janssen as predecessor-in-interest to GSK. 1.13 “Agreement Patent Rights” means any and all Patent Rights that (a) have a priority date after the Original Effective Date, and (b) claim or otherwise disclose any Agreement Know-How. 1.14 “Alliance Manager” has the meaning set forth in Section 3.13. 1.15 “Amended Effective Date” has the meaning set forth in the preamble above. 1.16 “Anti-Corruption Laws” means the FCPA and related regulations in the United States, and equivalent anti-bribery laws and regulations under Applicable Laws in other jurisdictions. 1.17 “APA” has the meaning set forth in the recitals above. 1.18 “Applicable Laws” means the applicable provisions of any national, supranational, regional, state and local laws, treaties, statutes, rules, regulations, administrative codes, guidance, ordinances, judgments, decrees, directives, injunctions, orders, permits, of or from any court, arbitrator, Regulatory Authority, or Governmental Authority having jurisdiction over or related to the subject item, including GCP, GLP and GMP, as applicable. 1.19 “ARO-HBV” has the meaning set forth in Section 1.123(a). 1.20 “Arrowhead” has the meaning set forth in the preamble above. 1.21 “Arrowhead Agreement IP” means, collectively, the Arrowhead Agreement Patent Rights and the Arrowhead Agreement Know-How. 1.22 “Arrowhead Agreement Know-How” has the meaning set forth in Section 8.2.3(c). 1.23 “Arrowhead Agreement Patent Rights” has the meaning set forth in Section 8.2.3(d). 1.24 “Arrowhead Excluded Know-How” means, collectively, any and all Know-How that Arrowhead or any of its Affiliates comes to Control after the Amended Effective Date during the Term (including any New Arising Know-How that is Arrowhead Agreement

4 Know-How), in each case, that (a) relates to CMC for the Manufacture of the Licensed Constructs or Licensed Products but is not necessary for the Manufacture of the Licensed Constructs or Licensed Products; or (b) (i) embodies RNAi molecule sequence selection and compound design process, and (ii) is not disclosed by Arrowhead to GSK under Section 3.4 (provided that this clause (b) shall not include any Know-How that is necessary for the clinical development, Manufacture or Commercialization of the Licensed Constructs or Licensed Products) but, in each case ((a) or (b)), excluding any Know-How that was solely or jointly invented by or on behalf of one (1) or more Personnel of GSK (or its Affiliates, licensees, sublicensees or subcontractors) (including any Joint Agreement Know-How or any such New [***] Know-How). 1.25 “Arrowhead Excluded Patent Rights” means any Patent Rights that claim or otherwise disclose any Arrowhead Excluded Know-How but, in each case, excluding any Patent Rights that also claim or otherwise disclose (a) any Arrowhead Know-How or (b) any Joint Agreement Know-How. 1.26 “Arrowhead Intellectual Property” means, collectively, (a) the Arrowhead Patent Rights; (b) the Arrowhead Know-How; and (c) Arrowhead’s interest in the Joint Agreement IP. 1.27 “Arrowhead Invention” has the meaning set forth in Section 8.1. 1.28 “Arrowhead Know-How” means any Know-How Controlled by Arrowhead or any of its Affiliates at any time that is necessary or reasonably useful to Exploit any Licensed Constructs or Licensed Products (including (a) all CMC Know-How; and (b) all Arrowhead Agreement Know-How) but, in each case, excluding (i) any Arrowhead Excluded Know-How or (ii) any Joint Agreement Know-How. 1.29 “Arrowhead Patent Rights” means any Patent Rights Controlled by Arrowhead or any of its Affiliates at any time that (a) claim or otherwise disclose any Arrowhead Know-How; or (b) otherwise Covers any Licensed Constructs or Licensed Products, or the Exploitation of any Licensed Constructs or Licensed Products (including (i) all General Arrowhead Patent Rights, (ii) all Specific Arrowhead Patent Rights and (iii) all Arrowhead Agreement Patent Rights) but, in each case ((a) or (b)), excluding (A) any Arrowhead Excluded Patent Rights or (B) any Joint Agreement Patent Rights. A list of certain Arrowhead Patent Rights that (1) as of the Original Execution Date, Cover the Exploitation of ARO-HBV, are attached to the Original Agreement as Exhibit B to the Original Agreement, or (2) as of the Amended Effective Date, Cover the Exploitation of ARO-HBV, is attached hereto as Schedule 1.29. For clarity, the General Arrowhead Patent Rights and Specific Arrowhead Patent Rights are as indicated (x) as of the Original Execution Date, on Exhibit B to the Original Agreement, or (y) as of the Amended Effective Date, on Schedule 1.29, as applicable. 1.30 “Arrowhead Platform Technology” means targeted RNAi molecule technology Controlled by Arrowhead or any of its Affiliates as of the Original Execution Date or during the Term utilizing targeting ligand-mediated delivery of RNAi designated by Arrowhead as its TRiMTM platform.

5 1.31 “Arrowhead Third Party Agreement” has the meaning set forth in Section 6.5.3(a). 1.32 “Audited Party” has the meaning set forth in Section 7.5.2. 1.33 “Audited Site” means any site or facility of a Party or any of its Affiliates, Third Party sublicensees, or Third Party contractors or subcontractors hereunder, as the case may be, on which any Clinical Trial or Manufacturing of Licensed Products for human use is conducted, and which is undergoing an inspection or audit by a Regulatory Authority or a Party as provided hereunder. 1.34 “Auditing Party” has the meaning set forth in Section 7.5.2. 1.35 “Bankrupt Party” has the meaning set forth in Section 13.3.1. 1.36 “Bankruptcy” means, with respect to a Party, that: (a) the Party has been declared insolvent or bankrupt by a court of competent jurisdiction; (b) a voluntary or involuntary petition for protection under any bankruptcy or insolvency laws (including under the Bankruptcy Code) is filed in any court of competent jurisdiction against the Party and such petition has not discharged or dismissed within forty (40) Business Days after filing; (c) the Party has made or executed an assignment of substantially all of its assets for the benefit of creditors; (d) proposes a written agreement of composition or extension of its debts; (e) proposes or is a party to any dissolution or liquidation of such Party; or (f) admits in writing its inability generally to meet its obligations as they fall due in the general course. 1.37 “Bankruptcy Code” means Title 11 of the United States Code, as may be amended or superseded from time to time. 1.38 “Bankruptcy Commencement Date” has the meaning set forth in Section 13.3.2. 1.39 “Breaching Party” has the meaning set forth in Section 13.2.1. 1.40 “Business Day” shall mean a day other than a Saturday, Sunday or public holiday in United States and England when banks in United States and England are open for normal banking business and excluding the period from 24 December to 2 January in which the corporate offices of GSK are closed for business. 1.41 “Calendar Quarter” means each three (3) month period commencing January 1, April 1, July 1 or October 1 of any Calendar Year; provided, however, that (a) the first Calendar Quarter of the Term shall extend from the Amended Effective Date to the end of the first full Calendar Quarter thereafter; and (b) the last Calendar Quarter of the Term shall end upon the expiration or termination of this Agreement. 1.42 “Calendar Year” means the period beginning on January 1 and ending on December 31 of the same year; provided, however, that (a) the first Calendar Year of the Term shall commence on the Amended Effective Date and end on December 31 of the same year; and (b) the last Calendar Year of the Term shall commence on January 1 of the Calendar Year in which this Agreement terminates or expires and end on the effective date of termination or expiration of this Agreement.

6 1.43 “CAPA” means a written recovery plan or proposal of corrective and preventative actions. 1.44 “Change of Control” means, with respect to a Party, (a) a merger or consolidation of such Party with a Third Party that results in the voting securities of such Party outstanding immediately prior thereto, or any securities into which such voting securities have been converted or exchanged, ceasing to represent at least fifty percent (50%) of the combined voting power of the surviving entity or the parent of the surviving entity immediately after such merger or consolidation, (b) a transaction or series of related transactions in which a Third Party, together with its Affiliates, becomes the direct or indirect beneficial owner of fifty percent (50%) or more of the combined voting power of the outstanding securities of such Party, or (c) the sale or other transfer to a Third Party of all or substantially all of such Party’s and its controlled Affiliates’ assets. Notwithstanding the foregoing, any transaction or series of transactions effected for the primary purpose of financing the operations of the applicable Party or changing the form or jurisdiction of organization of such Party will not be deemed a “Change of Control” for purposes of this Agreement. 1.45 “Clinical Investigation Laws” means Applicable Laws relating to human clinical investigations, such as 21 C.F.R. Parts 50, 54, 56 and 312 and then-current Good Clinical Practice, each as in effect and as amended from time to time. 1.46 “Clinical Trial” means a clinical trial in human subjects that has been approved by an institutional review board or ethics committee, as applicable, and is designed to measure the safety or efficacy of a therapeutic product, including any Phase 1 Clinical Trial, Phase 2 Clinical Trial, Phase 3 Clinical Trial, Post-Marketing Studies or any such clinical trial incorporating more than one (1) of these phases. 1.47 “CMC” means, chemistry, manufacturing and controls with respect to a product, which includes (a) manufacturing and process development records for such product; and (b) all chemistry, manufacturing and control procedures necessary or reasonably useful for the manufacture of such product. 1.48 “CMC Know-How” means the Arrowhead Know-How relating to the CMC of ARO- HBV, any Licensed Construct or any Licensed Product, including data, procedures, techniques, and information resulting from any test method development and stability testing, process development, process validation, process scale-up, formulation development, delivery system development, quality assurance and quality control development, and other related activities. 1.49 “Collaboration Activities” means the Parties’ activities (performed directly or, as may be permitted hereunder, on their behalf through their Affiliates, Third Party sublicensees or Third Party contractors or subcontractors) performed under this Agreement. 1.50 “Combination Product” means (a) a single pharmaceutical formulation containing as its Active Ingredients (i) one or more Licensed Constructs, and (ii) one or more Active Ingredients other than a Licensed Construct; (b) a bundle of products comprised of (i) one or more single pharmaceutical formulations comprising at least one Licensed Construct, and (ii) one or more other therapeutically effective or Prophylactically Active Products,

7 priced and sold in a single package containing such multiple products or packaged separately but sold together for a single price; or (c) a Licensed Product that is defined as a “combination product” by the FDA pursuant to 21 C.F.R. §3.2(e) or its foreign equivalent (but excluding devices, drug delivery vehicles, adjuvants, solubilizers and excipients), in each case ((a), (b) or (c)), in all dosage forms, formulations, presentations, line extensions and package configurations thereof. 1.51 “Commercialization” or “Commercialize” means, with respect to any product, any and all activities undertaken before and after Regulatory Approval of any Drug Application for such product and that are directed to marketing, promoting, importing or exporting for sale, using, offering for sale, or selling a product, including commercial manufacturing, launching product, conducting any Post-Marketing Studies, market access activities, price setting and price negotiation activities, managed care contract sales, medical affairs activities, and distribution and importation activities in support thereof. 1.52 “Commercially Reasonable Efforts” means, with respect to the efforts to be expended by any Party with respect to any objective, those reasonable, diligent, good faith efforts to accomplish such objective that a similarly situated pharmaceutical or biotechnology company in the exercise of its reasonable business discretion would normally use to accomplish a similar objective under similar circumstances. With respect to any objective relating to the research, Development, Manufacture or Commercialization of a Licensed Construct or Licensed Product by any Party, Commercially Reasonable Efforts shall mean those efforts and resources normally used by a similarly situated pharmaceutical or biotechnology company in the exercise of its reasonable business discretion with respect to a product owned or controlled by such Party, or to which such Party has similar rights, which product has similar product characteristics, is of similar market potential and is at a similar stage in its development or life as is such Licensed Product, taking into account all Relevant Factors. 1.53 “Competing Product” has the meaning set forth in Section 12.1. 1.54 “Confidential Information” has the meaning set forth in Section 9.1.1. 1.55 “Construct” means any Targeting Ligand linked to at least one strand of an RNAi Trigger. 1.56 “Control” means with respect to any Patent Rights, Know-How, Regulatory Approvals, Regulatory Filings, or materials, possession by a Party or one of its Affiliates (whether directly by ownership (either sole or joint) or license or sublicense from a Third Party, or indirectly through an Affiliate having ownership or license or sublicense from a Third Party (other than, in each case of such Party or such Affiliate, by a license, sublicense or other right granted (but not assignment) pursuant to this Agreement)) of the ability to grant to the other Party a license, sublicense, right of access, or other right to or under such Patent Rights, Know-How, Regulatory Approvals, Regulatory Filings, or materials, without (a) violating the terms of any agreement or other arrangement with any Third Party in existence as of the time such Party or its Affiliate would be required hereunder to grant such license, sublicense, right of access or other right thereto or thereunder, and (b) incurring any additional payment obligations to a Third Party that are not subject to an

8 allocation agreed between the Parties pursuant to this Agreement or otherwise in writing (including pursuant to Section 6.5.3), and with such agreement or other arrangement existing (i) with respect to Arrowhead, as of (A) the Original Effective Date or (B) subsequent to the Original Effective Date if (in the case of this clause (B)) Arrowhead first acquired rights to such Patent Rights, Know-How, Regulatory Approvals, Regulatory Filings or materials pursuant to such agreement or other arrangement; or (ii) with respect to GSK, as of (A) the Amended Effective Date or (B) subsequent to the Amended Effective Date if (in the case of this clause (B)) GSK first acquired rights to such Patent Rights, Know-How, Regulatory Approvals, Regulatory Filings or materials pursuant to such agreement or other arrangement. Notwithstanding anything in this Agreement to the contrary, a Party or its Affiliates will be deemed not to Control any Patent Rights, Know-How, Regulatory Approvals, Regulatory Filings or materials that are owned or in-licensed by an Acquirer except (1) if such Patent Rights, Know-How, Regulatory Approvals, Regulatory Filings or materials owned or in-licensed by the Acquirer were generated from participation by employees or consultants of such Acquirer in furtherance of Development, Manufacturing or Commercialization activities with respect to the Licensed Constructs or any Licensed Products under this Agreement after such Change of Control, (2) for any Patent Rights, Know-How, Regulatory Approvals, Regulatory Filings or materials owned or in-licensed by such Acquirer not used in the performance of Development, Manufacturing or Commercialization activities with respect to the Licensed Constructs or any Licensed Products under this Agreement prior to the consummation of such Change of Control that, after the consummation of such Change of Control, are used by such acquired Party or any of its Affiliates in the performance of Development, Manufacturing or Commercialization activities with respect to the Licensed Constructs or any Licensed Products under this Agreement, or (3) if, prior to the consummation of such Change of Control, such acquired Party or any of its Affiliates also Controlled such Patent Rights, Know-How, Regulatory Approvals, Regulatory Filings or materials owned or in-licensed by such Acquirer, in each of which cases ((1)–(3)), such Patent Rights, Know-How, Regulatory Approvals, Regulatory Filings or materials owned or in-licensed by such Acquirer will be deemed Controlled by the acquired Party or its Affiliates for purposes of this Agreement. 1.57 “Cover” means, with respect to a given chemical composition of matter, compound, construct or product (including any Licensed Construct or Licensed Product) and a given claim of a Patent Right in a particular country or other jurisdiction, that the making, offering for sale, selling, importing or using of such chemical composition of matter, compound, construct or product would, but for the ownership of, or a license granted under, such Patent Right, infringe any claim of such Patent Right in such country or other jurisdiction in which that activity occurs. 1.58 “CPR Mediation Procedure” has the meaning set forth in Section 14.2. 1.59 “CPR Rules” has the meaning set forth in Section 14.3. 1.60 “Cure Period” has the meaning set forth in Section 13.2.1.

9 1.61 “Develop” means any and all pre-clinical, clinical, and other activities to study a drug candidate or product and develop it toward Regulatory Approval (including any such activities conducted after such Regulatory Approval other than Post-Marketing Studies) for Commercialization, including toxicology and ADME tests, analytical method development, stability testing, process development and improvement, process validation, process scale-up prior to first Regulatory Approval, formulation development, delivery system development, quality assurance and quality control development, statistical analysis, pre- and post-approval Clinical Trials, regulatory affairs, regulatory activities and manufacturing activities in support thereof. For clarity, the definition of “Development” shall include all activities under the Development Plan but exclude all Commercialization activities. “Developing”, “Development” and “Development activities” shall each have a correlative meaning. 1.62 “Development Plan” means the plan describing the Development of the Licensed Product(s) as of the Amended Effective Date, which is attached hereto as Schedule 3.2. 1.63 “Disclosing Party” has the meaning set forth in Section 9.1.1. 1.64 “Dispute” means any dispute, claim, or controversy arising from or regarding this Agreement, including the interpretation, application, breach, termination, or validity of any provision hereof. 1.65 “Drug Application” means an NDA, MAA, or equivalent application, submitted to a Regulatory Authority in a particular jurisdiction, for marketing approval of a pharmaceutical or drug product. 1.66 “Drug Regulation Laws” means Applicable Laws regulating drugs and pharmaceutical products, such as the Federal Food, Drug, and Cosmetic Act, 21 U.S.C. § 301 et. seq., the Prescription Drug Marketing Act of 1987, the Controlled Substances Act, 21 U.S.C. § 801 et. seq., and policies issued by the FDA, each as in effect and as amended from time to time. 1.67 “EMA” means the European Medicines Agency or any successor agency for the EU. 1.68 “European Union” or “EU” means the countries of the European Economic Area, as it is constituted on the Amended Effective Date and as it may be modified from time to time after the Amended Effective Date. 1.69 “Exclusivity Term” has the meaning set forth in Section 12.1. 1.70 “Executive Officers” means (a) for Arrowhead, the Chief Executive Officer of Arrowhead or any executive officer of Arrowhead delegated authority by the Chief Executive Officer with respect to any particular matter; and (b) for GSK, the Chief Scientific Officer of GSK, or any executive officer of GSK delegated authority by the Chief Scientific Officer of GSK with respect to any particular matter. In the event that the position of any of the Executive Officers identified in this Section 1.70 no longer exists due to a corporate reorganization, corporate restructuring or the like that results in the elimination or modification of the identified position, the applicable Executive Officer shall be replaced with another senior

10 officer with responsibilities and seniority comparable to the eliminated or modified position. 1.71 “Existing Clinical Data” has the meaning set forth in Section 8.2.2. 1.72 “Existing Confidentiality Agreement” means (a) that certain Confidential Disclosure Agreement, dated as of June 21, 2023, by and between GlaxoSmithKline LLC and Arrowhead; and (b) that certain Joinder Agreement to Confidential Disclosure Agreement, by and among GlaxoSmithKline LLC, Janssen, and Arrowhead, dated as of July 19, 2023. 1.73 “Existing Invention” has the meaning set forth in Section 8.2.2. 1.74 “Existing Joint Agreement Patent Rights” means any Agreement Patent Rights that (a) have a priority date prior to the Amended Effective Date; and (b) claim or otherwise disclose any Existing Invention that was jointly invented by or on behalf of (i) on the one hand, Arrowhead (or any of its Affiliates) and (ii) on the other hand, GSK (as the permitted assigned of Janssen) (whether or not they solely relate to the Registered Starting Material ([***])), including those Patent Rights set forth on Schedule 1.74. 1.75 “Exploit” means to make, have made, import, use, sell or offer for sale, including to research, develop, commercialize, register, manufacture, have manufactured, hold or keep (whether for disposal or otherwise), have used, export, transport, distribute or have distributed by others, promote, market or have sold or otherwise dispose of, or have offered for sale, and convey or grant end-users use rights. “Exploiting” and “Exploitation” shall each have a correlative meaning. 1.76 “FCPA” means the U.S. Foreign Corrupt Practices Act (15 U.S.C. § 78dd-1 et. seq.), as may be amended at the relevant time. 1.77 “FDA” means the United States Food and Drug Administration or any successor agency thereto for the United States. 1.78 “Field” means all therapeutic, prophylactic and diagnostic uses in humans or animals. 1.79 “First Commercial Sale” means, on a Licensed Product-by-Licensed Product and country-by-country basis, with respect to a given Licensed Product in a given country, the first commercial sale for monetary value in an arms-length transaction of such Licensed Product to a Third Party purchaser by or on behalf of a Selling Party in such country following receipt of applicable Regulatory Approval for such Licensed Product in such country; provided, however, that First Commercial Sale shall not include any transfer of a Licensed Product (a) between or among GSK or any other Selling Party or any of its or their respective Third Party subcontractors (such as contract manufacturers, suppliers, or distributors for consignment, where such transfer is not a transfer to a wholesaler or retailer) or (b) for purposes of patient assistance, charitable or promotional purposes, for use in a Clinical Trial or for use in any other tests or studies reasonably necessary to comply with any Applicable Law or request by a Regulatory Authority; provided, further, that solely for purposes of defining the Royalty Term as set forth in Section 1.184, the transfers

11 set forth in clauses (a) or (b) above shall only be excluded from the definition of First Commercial Sale hereunder if such transfer are made without consideration. 1.80 “FTE” means the equivalent of the work of one qualified employee or agent for the applicable activities, full time, for one year (constituting one thousand eight hundred (1,800) working hours). For clarity, no more than one thousand eight hundred (1,800) hours per year (or equivalent pro-rata portion thereof for a period less than twelve (12) months) may be charged for an individual contributing work factoring into any reimbursable FTE Costs hereunder, regardless of how much additional work time is contributed by such individual during such period. An individual contributing work for less than one thousand eight hundred (1,800) hours per year shall be deemed a fraction of an FTE on a pro-rata basis. 1.81 “FTE Costs” means the FTE Rate times the number of FTEs expended during the applicable financial period. The FTE Costs shall be determined based on time (as calculated in pro-rated FTEs) actually spent performing the applicable activities under this Agreement, unless another basis is expressly specified herein or otherwise agreed in advance by the Parties in writing. 1.82 “FTE Rate” means, for the period commencing on the Amended Effective Date until such time as the Parties agree otherwise, [***] per year, subject to annual increases beginning on January 1, 2025 to reflect percentage increase in the Consumer Price Index for the US City Average, calculated by the Bureau of Labor Statistics during the immediately preceding Calendar Year and similarly calculated year to year increases each subsequent Calendar Year. 1.83 “G5 Countries” means France, Germany, Italy, Spain and the United Kingdom. 1.84 “GAAP” means United States generally accepted accounting principles applied on a consistent basis. 1.85 “General Arrowhead Patent Rights” has the meaning as set forth in Section 8.3.3(b). 1.86 “Generic Competition” has the meaning as set forth in Section 6.4.5(c). 1.87 “Generic Version” means, with respect to a particular Licensed Product in a particular country, a product on the market in such country commercialized by any Third Party that is not a sublicensee of GSK and that did not purchase such product in a chain of distribution that included GSK or any of its respective Selling Parties, that has been approved in such country by the applicable Regulatory Authority in reliance, in whole or in part, on the prior approval (or on safety or efficacy data submitted in support of the prior approval) of such Licensed Product as determined by the applicable Regulatory Authority, including any product that is authorized for sale (a) in the U.S. pursuant to Section 505(j) of the US Federal Food, Drug, and Cosmetic Act (21 USC Section 355(j)); (b) in the European Union pursuant to a provision of Articles 10, 10a or 10b of Parliament and Council Directive 2001/83/EC as amended (including an application under Article 6.1 of Parliament and Council Regulation (EC) No 726/2004 that relies for its content on any such provision); or (c) any foreign equivalent thereof or successors thereto.

12 1.88 “Good Clinical Practice” or “GCP” means the current standards for Clinical Trials for pharmaceutical and biologic products, as set forth in the ICH guidelines and applicable regulations promulgated thereunder, as amended from time to time, and such standards of good clinical practice as are required by the European Union and other Governmental Authorities in countries in which a Licensed Product is intended to be sold to the extent such standards are not less stringent than United States Good Clinical Practice. 1.89 “Good Laboratory Practice” or “GLP” means the current standards for laboratory activities for pharmaceutical and biologic products, as set forth in the FDA’s Good Laboratory Practice regulations or the Good Laboratory Practice principles of the Organization for Economic Co-Operation and Development, as amended from time to time, and such standards of good laboratory practice as are required by the European Union and other Governmental Authorities in countries in which a Licensed Product is intended to be sold, to the extent such standards are not less stringent than United States Good Laboratory Practice. 1.90 “Good Manufacturing Practice” or “GMP” means the current quality assurance standards that ensure that pharmaceutical and biologic products are consistently produced and controlled in accordance with the quality standards appropriate to their intended use as defined in 21 C.F.R. § 210 and 211, European Directive 2003/94/EC, Eudralex Volume 4 and applicable United States, European Union, Canadian and ICH guidance or equivalent laws in other jurisdictions to the extent no less stringent. 1.91 “Government Health Care Programs” means the US Medicare program (Title XVIII of the Social Security Act), the US Medicaid program (Title XIX of the Social Security Act), the TRICARE program, the US Federal employee health benefits program, and other foreign, federal, state and local governmental health care plans and programs. 1.92 “Government Order” means any order, writ, judgment, injunction, decree, stipulation, ruling, determination or award entered by or with any Governmental Authority. 1.93 “Governmental Authority” means any United States federal, state or local government or any government other than the United States government, or political subdivision thereof, or any multinational organization or authority to the extent empowered to act on behalf of or in the stead of a government, or any authority, agency, or commission entitled to exercise any administrative, executive, judicial, legislative, police, regulatory, pricing or taxing authority or power, any court or tribunal (or any department, bureau or division thereof), or any governmental arbitrator or government empowered arbitral body. 1.94 “GSK” has the meaning set forth in the preamble above. 1.95 “GSK Agreement IP” means, collectively, the GSK Agreement Know-How and the GSK Agreement Patent Rights. 1.96 “GSK Agreement Know-How” has the meaning set forth in Section 8.2.4(a). 1.97 “GSK Agreement LC/LP IP” means, collectively, the GSK Agreement LC/LP Know- How and the GSK Agreement LC/LP Patent Rights.

13 1.98 “GSK Agreement LC/LP Know-How” means any GSK Agreement Know-How Controlled by GSK (or any of its Affiliates) during the Term that is (a) actually being used by GSK (or any of its Affiliates) in the Development, Manufacture or Commercialization of any Licensed Construct or Licensed Product; and (b) necessary to Exploit such Licensed Construct or Licensed Product but, in each case ((a) and (b)), excluding (i) any Know-How that is specifically related to (A) with respect to any Licensed Product that is a Combination Product, any Active Ingredient other than a Licensed Construct (or the Development, Manufacturing or Commercialization thereof) or (B) any compound, construct or product that is not a Licensed Product (or the Development, Manufacturing or Commercialization thereof), and (ii) any GSK Excluded Know-How. 1.99 “GSK Agreement LC/LP Patent Rights” means any GSK Agreement Patent Rights Controlled by GSK (or any of its Affiliates) during the Term that (a) claim any GSK Reversion Know-How; or (b) Cover any Licensed Construct or Licensed Product (or the Development, Manufacture or Commercialization thereof) but, in each case ((a) or (b)), excluding (i) any Patent Rights that also claim or otherwise disclose (A) with respect to any Licensed Product that is a Combination Product, any Active Ingredient of such Combination Product that is not a Licensed Construct (or the Development, Manufacturing or Commercialization thereof), or (B) any compound, construct or product that is not a Licensed Product (or the Development, Manufacturing or Commercialization thereof), and (ii) any GSK Excluded Patent Rights. 1.100 “GSK Agreement Patent Rights” means, collectively, (a) all Agreement Patent Rights set forth in Section 8.2.4(b), and (b) the Janssen Licensed Patent Rights. 1.101 “GSK Excluded Know-How” means [***]. 1.102 “GSK Excluded Patent Rights” means [***]. 1.103 “GSK Reversion IP” means, collectively, the GSK Reversion Know-How and the GSK Reversion Patent Rights. 1.104 “GSK Reversion Know-How” means any GSK Agreement Know-How or Joint Agreement Know-How Controlled by GSK (or any of its Affiliates) as of the effective date of any termination of this Agreement by Arrowhead pursuant to Section 13.2.2 or by GSK pursuant to Section 13.5, in each case, that, as of the effective date of such termination, is (a) actually used by GSK (or any of its Affiliates) in the Development, Manufacture or Commercialization of any Terminated Product; and (b) necessary to Exploit such Terminated Product but, in each case ((a) and (b)), excluding (i) any Know-How that is specifically related to (A) with respect to any Terminated Product that is a Combination Product, any Active Ingredient other than a Licensed Construct (or the Development, Manufacturing or Commercialization thereof) or (B) any compound, construct or product that is not a Terminated Product (or the Development, Manufacturing or Commercialization thereof), and (ii) any GSK Excluded Know-How. 1.105 “GSK Reversion Patent Rights” means any GSK Agreement Patent Rights or Joint Agreement Patent Rights Controlled by GSK (or any of its Affiliates) as of the effective

14 date of any termination of this Agreement by Arrowhead pursuant to Section 13.2.2 or by GSK pursuant to Section 13.5 that (a) claim any GSK Reversion Know-How; or (b) Cover any Terminated Product (or the Development, Manufacture or Commercialization thereof) but, in each case ((a) or (b)), excluding (i) any Patent Rights that also claim or otherwise disclose (A) with respect to any Terminated Product that is a Combination Product, any Active Ingredient of such Combination Product that is not a Licensed Construct (or the Development, Manufacturing or Commercialization thereof), or (B) any compound, construct or product that is not a Terminated Product (or the Development, Manufacturing or Commercialization thereof), and (ii) any GSK Excluded Patent Rights. 1.106 “HBV” has the meaning set forth in the recitals above. 1.107 “HDV” means the hepatitis D virus. 1.108 “Health Care Laws” means Applicable Laws relating to Government Health Care Programs, Private Health Care Plans, privacy and confidentiality of patient health information and human biological materials, including, in the United States, federal and state Applicable Laws pertaining to the federal Medicare and Medicaid programs (including the Medicaid rebate program); federal Applicable Laws pertaining to the Federal employees health benefit program and the TRICARE program; federal and state Applicable Laws applicable to health care fraud and abuse, kickbacks, physician self-referral and false claims (including 42 U.S.C. § 1320a-7a, 42 U.S.C. § 1320a-7b, 42 U.S.C. § 1395nn and the federal Civil False Claims Act, 31 U.S.C. § 3729 et. seq.); the Health Insurance Portability and Accountability Act of 1996; and 45 C.F.R. Part 46, as well as similar Applicable Laws in the Territory, each as in effect and as amended from time to time. 1.109 “Human Biological Samples” means any human biological material (including any derivative or progeny thereof), including any portion of an organ, any tissue, skin, bone, muscle, connective tissue, blood, cerebrospinal fluid, cells, gametes, or sub-cellular structures such as DNA, or any derivative of such biological material such as stem cells or cell lines; and any human biological product, including, but not limited to, hair, nail clippings, teeth, urine, faeces, breast milk, and sweat. 1.110 “ICH” means the International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use. 1.111 “IFRS” means International Financial Reporting Standards applied on a consistent basis. 1.112 “In-Licensed Arrowhead Patent Rights” has the meaning set forth in Section 10.2.3. 1.113 “IND” means an Investigational New Drug Application filed with the FDA, or a similar application filed with a Regulatory Authority outside of the United States for authorization to commence a Clinical Trial, such as a clinical trial application or a clinical trial exemption, or any related regulatory submission, license or authorization. 1.114 “Indemnified Party” has the meaning set forth in Section 11.1.

15 1.115 “Indemnified Persons” shall mean, with respect to a Party, such Party and its Affiliates, and any of its or their respective officers, directors, employees or agents. 1.116 “Indemnifying Party” has the meaning set forth in Section 11.1. 1.117 “Janssen” has the meaning set forth in the recitals above. 1.118 “Janssen Licensed Patent Rights” means, collectively, (a) the Patent Rights set forth on Schedule 1.118; and (b) any and all other Patent Rights Controlled by GSK (or its Affiliates) during the Term pursuant to a non-exclusive license granted by Janssen to GSK under the APA or an ancillary agreement thereto. 1.119 “Joint Agreement IP” means, collectively, the Joint Agreement Know-How and the Joint Agreement Patent Rights. 1.120 “Joint Agreement Know-How” has the meaning set forth in Section 8.2.5(a). 1.121 “Joint Agreement Patent Rights” has the meaning set forth in Section 8.2.5(b). 1.122 “Know-How” means any and all proprietary technical, scientific, and other know-how (whether or not patentable), data, results and other information, and materials of any type whatsoever, in any tangible or intangible form whatsoever, that are not generally known to the public, including discoveries, inventions, trade secrets, research and development data, databases, plans, practices, procedures, experimental techniques, protocols, material specifications, and assay or test protocols; biological, chemical, pharmacological, toxicological, pharmaceutical, pre-clinical, clinical, safety, and quality control data and information; methods and processes (including manufacturing methods and formulas); and molecules, chemical entities, reagents, starting materials, reaction intermediates, building blocks, synthetic products, delivery systems, excipients, ingredients, formulations, formulae, and compositions of matter; case reports forms, medical records, data analyses, reports, studies and procedures, designs for experiments and tests and results of experimentation and testing (including results of any Development activities), summaries and information contained in submissions to and information from ethical committees, or Regulatory Authorities. The fact that an item is known to the public shall not be taken to exclude the possibility that a compilation including the item, or a development relating to the item, is (and remains) not known to the public. For clarity, “Know-How” includes any rights (other than Patent Rights, but including copyright, database or design rights) protecting such Know-How. 1.123 “Licensed Construct” means (a) the chemical composition of matter known as “ARO- HBV” set forth on Schedule 1.123(a) (“ARO-HBV”); (b) [***]; and (c) any salts, hydrates, solvates, esters, metabolites, intermediates (including the Registered Starting Material ([***])), stereoisomers, polymorph, complexes, cocrystals, derivatives and formulations of any of the compositions of matter in the foregoing clause (a) or (b). 1.124 “Licensed Product” means any and all products in any dosage form and strengths, or formulation, or method of delivery, including any improvements thereto, that contains a

16 Licensed Construct as an Active Ingredient, whether as the sole Active Ingredient or as a Combination Product with one or more Active Ingredients other than a Licensed Construct. 1.125 “Losses” means damages, losses, liabilities, costs (including costs of investigation and defense), fines, penalties, or expenses (including reasonable attorneys’ and experts fees and expenses), resulting from a claim in an Action by a Third Party, and incurred by a Party (or other Indemnified Person as provided in Article XI) as a result of such Action. 1.126 “MAA” means (a) a marketing authorization application filed with (i) the EMA under the centralized EMA filing procedure or (ii) a Regulatory Authority in any country in the European Union if the centralized EMA filing procedure is not used; or (b) any other equivalent or related regulatory submission, in either case to gain approval to market a pharmaceutical or biologic product in any country in the European Union, in each case including, for the avoidance of doubt, amendments thereto and supplemental applications. 1.127 “Manufacturing” means activities performed to manufacture a product into final form for end use, including producing and manufacturing starting materials and intermediates used to manufacture such product, filling, finishing, packaging, labeling, performing quality assurance testing and release, and shipping and storing the product. 1.128 “MHLW” means the Ministry of Health, Labour and Welfare of Japan and any successor agency thereto. 1.129 “NDA” means a new drug application or biologics license application submitted to the FDA for purposes of obtaining Regulatory Approval for a new drug in the United States, for a particular indication, including, for the avoidance of doubt, amendments thereto and supplemental applications. 1.130 “Net Sales” means, with respect to a Licensed Product commencing with its First Commercial Sale, the gross invoiced sales amounts of such Licensed Product by or on behalf of GSK or any of its Affiliates or Sublicensees (each, a “Selling Party”) to a Third Party (but not including sales relating to transactions between or among any Selling Parties, other than sales to wholesalers and distributors) in an arms-length transaction, less the following deductions from such gross amounts, determined in accordance with the applicable Accounting Standards of such Selling Party consistently applied, to the extent specifically and solely allocated to the sale of such Licensed Product to such Third Party and actually taken, paid, accrued, allowed, included, or allocated based on good faith estimate in the gross sales price with respect to such sale (and consistently applied as set forth below): (a) normal and customary trade, cash or quantity discounts, allowances, wholesaler and pharmacy fees, and credits allowed or paid, in the form of deductions actually allowed or actually paid with respect to sales of such Licensed Product (to the extent not already reflected in the amount invoiced) excluding commissions for commercialization;

17 (b) non-recoverable sales taxes, VAT and Indirect Taxes, and other taxes directly linked to the sales of such Licensed Product to the extent included in the gross amount invoiced; (c) outbound freight, shipment and insurance costs to the extent included in the price and separately itemized on the invoice price; (d) compulsory payments and cash rebates imposed on sales of such Licensed Product paid to a Governmental Authority (or agent thereof) pursuant to Applicable Law by reason of any national or local health insurance program or similar program, to the extent allowed and taken, including fees levied by a Governmental Authority as a result of Applicable Law; (e) retroactive price reductions, credits or allowances actually granted upon rejections or returns of such Licensed Product, including for recalls or damaged goods and billing errors; (f) rebates, charge backs and discounts (or equivalents thereof) actually granted to managed health care organizations, pharmacy benefit managers (or equivalents thereof), federal, state/provincial, local or other Governmental Authorities, or their agencies or purchasers, reimbursers, or trade customers; (g) coupons, or discount/rebates associated with co-pay cards; and (h) any other items actually deducted from gross invoiced sales amounts as reported by such Selling Party in its financial statements in accordance with its applicable Accounting Standard, applied on a consistent basis. In no event will any particular amount identified above be deducted more than once in calculating Net Sales (i.e., no “double counting” of deductions). To the extent that any Selling Party receives consideration other than or in addition to cash upon the sale or disposition of a Licensed Product, Net Sales will be calculated based on the average price charged for such Licensed Product, as applicable, during the preceding royalty period, or in the absence of such sales, based on such Selling Party’s reasonable determination of the fair market value of the Licensed Product. The permitted deductions of Section 1.130(a) through Section 1.130(h) above will be fairly allocated to the Licensed Product and, as between the Licensed Product and other products or services of such Selling Party will not be inappropriately allocated. Such Selling Party will not attempt to reduce compensation rightly due to Arrowhead under this Agreement by shifting compensation otherwise payable to such Selling Party from a Third Party with respect to any Licensed Product to another product or service for which no royalties are payable to Arrowhead under this Agreement. For purposes of the definition of Net Sales: if any Licensed Product under this Agreement is sold in the form of a Combination Product, and such Licensed Product and any other Active Ingredient(s) contained within such Combination Product are sold separately, the Net Sales of such Combination Product for any period shall be determined by multiplying

18 the Net Sales (as defined above in this Section 1.130) of such Combination Product for such period by the fraction, A/(A+B) where A is the weighted (by sales volume) average gross sale price in a particular country of such Licensed Product during such period when sold separately in finished form and B is the weighted average gross sale price in such country during such period of such other Active Ingredient(s) are sold separately in finished form. In the event that the weighted average gross sale price of such Licensed Product for a period can be determined but the weighted average gross sale price of such other Active Ingredient(s) cannot be determined, the Net Sales of such Licensed Product for such period shall be calculated by multiplying the Net Sales of such Combination Product for such period by the fraction A/C where A is the weighted average gross sale price of such Licensed Product during such period when sold separately in finished form and C is the weighted average gross sale price of such Combination Product during such period. In the event that the weighted average gross sale price of such other Active Ingredient(s) for a period can be determined but the weighted average gross sale price of such Licensed Product for such period cannot be determined, the Net Sales of such Licensed Product for such period shall be calculated by multiplying the Net Sales of such Combination Product for such period by a fraction determined by the following formula: one (1) minus B/C where B is the weighted average gross sale price of such other Active Ingredient(s) during such period when sold separately in finished form and C is the weighted average gross sale price of such Combination Product during such period. In the event that the weighted average gross sale price of both such Licensed Product and such other Active Ingredient(s) in such Combination Product cannot be determined for a period, the Net Sales of such Licensed Product for such period shall be based upon the relative value contributed by each component. GSK (or the applicable other Selling Party) shall propose a value for the weighted average gross sale price of such Licensed Product and such other Active Ingredient(s) in such Combination Product. Within twenty (20) Business Days after GSK, on behalf of itself or the applicable other Selling Party, submits such proposal to Arrowhead, the Parties shall meet to discuss, acting reasonably and in good faith, and agree upon (which agreement shall not be unreasonably withheld) the weighted average gross sales price of such Licensed Product and such other Active Ingredient(s) in such Combination Product for such period. The weighted average gross sale price for such Licensed Product, such other Active Ingredient(s), or such Combination Product, as applicable, for such period shall be calculated once each Calendar Year and such price shall be used during all applicable reporting periods for the entire following Calendar Year. When determining the weighted average gross sale price of a Licensed Product, any other Active Ingredient(s), or any Combination Product for a period, the weighted average gross sale price shall be calculated by dividing the sales dollars (translated into US Dollars) by the units of active ingredient sold during the twelve (12) months (or the number of months sold in a partial Calendar Year) of the preceding Calendar Year for such Licensed Product, such other Active Ingredient(s), or Combination Product. In the initial Calendar Year, a forecasted weighted average gross sale price will be used for such Licensed Product, such other Active Ingredient(s), or Combination Product. Any over or under payment due to a difference between forecasted and actual weighted average gross

19 sale prices will be paid or credited in the first applicable payment of the following Calendar Year. Sales of Licensed Products among GSK and any other Selling Parties (including sales by any such Selling Party to GSK or another Selling Party) for resale by such entity to a Third Party shall not be deemed a sale for purposes of this definition of “Net Sales”; provided that the resale of such Licensed Products by such entity to a Third Party (other than a Sublicensee, but including wholesalers and distributors) shall be deemed a sale for the purposes of this definition of “Net Sales.” If GSK or any of its Affiliates or Sublicensees purchases Licensed Products from a Selling Party that is the end user of such Licensed Product, then Net Sales shall include the value of such sale, calculated at the higher of (i) the actual price paid in such sale for such Licensed Product or (ii) the fair market value of such Licensed Product at the time of such sale (as determined by the mutual agreement of the Parties, acting reasonably and in good faith). Transfers or dispositions of any Licensed Product for no monetary consideration: (A) in connection with patient assistance programs; (B) for charitable or promotional purposes; (C) for preclinical, clinical, regulatory or governmental purposes or under so-called “named patient” or other limited access programs; or (D) for use in any tests or studies, including Clinical Trials, reasonably necessary to comply with any Applicable Law, regulation or request by a Regulatory Authority shall not, in each case ((A) through (D)), be deemed sales of such Licensed Product for purposes of this definition of “Net Sales”. 1.131 “New Arising Know-How” means any and all Know-How that is discovered, developed, generated, invented, derived, created, conceived or reduced to practice during the Term, at any time on or after the Amended Effective Date, by or on behalf of one (1) or more Personnel of a Party (or any of its Affiliates, licensees, sublicensees or subcontractors), either alone or jointly with one (1) or more Personnel of the other Party (or any of its Affiliates, licensees, sublicensees or subcontractors), in each case, in the performance of activities under this Agreement. 1.132 “New [***] IP” means, collectively, the New [***] Know-How and the New [***] Patent Rights. 1.133 “New [***] Know-How” has the meaning set forth in Section 8.2.3(a). 1.134 “New [***] Patent Rights” has the meaning set forth in Section 8.2.3(b). 1.135 “Non-Bankrupt Party” has the meaning set forth in Section 13.3.1. 1.136 “Non-Breaching Party” has the meaning set forth in Section 13.2.1. 1.137 “Notice of Claim” has the meaning set forth in Section 11.2.1. 1.138 “Original Agreement” has the meaning set forth in the recitals above. 1.139 “Original Effective Date” means October 29, 2018.

20 1.140 “Original Execution Date” has the meaning set forth in the recitals above. 1.141 “Out-of-Pocket Costs” means, with respect to a Party, costs and expenses paid by such Party to any Third Party for services or materials provided by such Third Party to directly support applicable activities under this Agreement. For clarity, Out-of-Pocket Costs do not include payments for a Parties’ or its Affiliates’ internal salaries or benefits, facilities, utilities, general office or facility supplies, insurance or information technology, capital expenditures or the like. 1.142 “Owned General Arrowhead Patent Rights” has the meaning set forth in Section 10.2.3. 1.143 “Owned Specific Arrowhead Patent Rights” has the meaning set forth in Section 10.2.3. 1.144 “Party” and “Parties” have the meaning set forth in the preamble above. 1.145 “Patent Controversy” means any Dispute between the Parties to the extent that it involves an issue relating to the validity, scope, enforceability, patentability, inventorship or ownership of any Patent Rights, Know-How or other intellectual property rights hereunder, and including any such issues relevant to any Prosecution activities hereunder. 1.146 “Patent Costs” means all Out-of-Pocket Costs reasonably incurred by or on behalf of a Party (such as by a designated Affiliate) in Prosecuting applicable Patent Rights. 1.147 “Patent Office” means the United States Patent and Trademark Office, European Patent Office, or other Governmental Authority responsible for the examination of patent applications or granting of other Patent Rights in a country, region, or supra-national jurisdiction. 1.148 “Patent Rights” means, in reference to a designated invention, all original (priority establishing) patent applications claiming such invention filed anywhere in the world, including provisionals and nonprovisionals, and all related applications thereafter filed, including any continuations, continuations-in-part, divisionals, or substitute applications, any patents issued or granted from any such patent applications, and any reissues, reexaminations, renewals or extensions (including by virtue of any supplementary protection certificates) of any such patents, and any confirmation patents or registration patents or patents of addition based on any such patents, and all foreign counterparts or equivalents of any of the foregoing in any country or jurisdiction. 1.149 “Patent Term Extension” means an extension of the term of any issued patent, or a right of protection equivalent to such an extension, granted under law or regulation such as the U.S. Drug Price Competition and Patent Term Restoration Act of 1984 in the United States, the Supplementary Protection Certificate of the member states of the EU, or any other similar law or regulation in any other country or jurisdiction. For example, a pediatric extension obtained by application to or through approval of a Patent Office extending the term of any patent shall be deemed a Patent Term Extension. 1.150 “Patent Working Group” has the meaning set forth in Section 8.8.

21 1.151 “Payee Party” has the meaning set forth in Section 7.3. 1.152 “Payor Party” has the meaning set forth in Section 7.3. 1.153 “Person” means any natural person, corporation, firm, business trust, joint venture, association, organization, company, partnership or other business entity, or any government or agency or political subdivision thereof. 1.154 “Personnel” means, with respect to any Person, its officers, directors, employees, workers, contractors, advisors, consultants, agents or other representatives. 1.155 “Pharmacovigilance Agreement” has the meaning set forth in Section 3.6.4. 1.156 “Phase 1 Clinical Trial” means, in reference to a Clinical Trial of a Licensed Product, that as described in US federal regulation 21 C.F.R. § 312.21(a). 1.157 “Phase 2 Clinical Trial” means, in reference to a Clinical Trial of a Licensed Product, that as described in US federal regulation 21 C.F.R. § 312.21(b). 1.158 “Phase 3 Clinical Trial” means, in reference to a Clinical Trial of a Licensed Product, that as described in US federal regulation 21 C.F.R. § 312.21(c). 1.159 “Post-Marketing Studies” means any Clinical Trial conducted with a Licensed Product after receipt of Regulatory Approval of the Licensed Product, which are conducted voluntarily in order to enhance marketing or scientific knowledge of the Licensed Product and are not required by Regulatory Authorities or are not intended to support Regulatory Approval of a Licensed Product for a new indication or other material change to the product label. 1.160 “Pre-Existing Acquired Rights from Third Parties” means any and all agreements by and between Arrowhead and any Third Party, in effect as of the Original Execution Date, and pursuant to which the Third Party assigns (by express terms, whether or not using the word “assign”) Arrowhead any Third Party’s Patent Rights or Know-How that, in whole or in part, are necessary or useful for Developing, Manufacturing, or Commercializing any Licensed Product. As of the Amended Effective Date, the Pre-Existing Acquired Rights from Third Parties are set forth on Part III of Schedule 1.163. 1.161 “Pre-Existing Licenses from Third Parties” means any and all agreements by and between Arrowhead and any Third Party, in effect as of the Original Execution Date, and pursuant to which the Third Party grants (by express terms, whether or not using the word “license”) Arrowhead any license or sublicense (or use or other Exploitation) rights to or under any Third Party’s Patent Rights or Know-How that, in whole or in part, are necessary or useful for Developing, Manufacturing, or Commercializing any Licensed Product. As of the Amended Effective Date, the Pre-Existing Licenses from Third Parties are set forth on Part I of Schedule 1.163. 1.162 “Pre-Existing Licenses to Third Parties” means any and all agreements by and between Arrowhead and any Third Party, in effect as of the Original Execution Date, and pursuant

22 to which Arrowhead or its Affiliates grants (by express terms, whether or not using the word “license”) such Third Party any license or sublicense (or use or other Exploitation) rights to or under any Arrowhead Intellectual Property. As of the Amended Effective Date, the Pre-Existing Licenses to Third Parties are set forth on Part I of Schedule 1.163. 1.163 “Pre-Existing Third Party Agreements” means (a) Pre-Existing Licenses to Third Parties; (b) Pre-Existing Licenses from Third Parties; (c) Pre-Existing Acquired Rights from Third Parties; and (d) any other agreements between Arrowhead or its Affiliates and a Third Party in effect as of the Original Execution Date that contain any terms relating to the Development, Manufacture, or Commercialization of a Licensed Product, ARO-HBV or any Licensed Construct; provided that, as of (i) the Original Execution Date, the Pre- Existing Third Party Agreements are set forth on Exhibit E to the Original Agreement, or (ii) the Amended Effective Date, the Pre-Existing Third Party Agreements are set forth on Schedule 1.163. 1.164 “Private Health Care Plans” means non-governmental Third Party health care payors and plans, including insurance companies, health maintenance organizations and other managed care organizations, Blue Cross and Blue Shield plans, and self-funded employers. 1.165 “Product Infringement” has the meaning set forth in Section 8.4.2(a). 1.166 “Product Trademark Rights” means any Trademark Rights pertaining specifically to any Licensed Product and Controlled by a Party hereunder. 1.167 “Prophylactically Active Product” means a product that prevents any disease, condition or symptom associated with or induced by HBV infection or HDV infection in humans or animals. 1.168 “Prosecuting” means, in reference to a designated Patent Right, preparing a Patent Right in application form for filing in any Patent Office, or performing activities associated with filing, prosecuting, maintaining, defending, or correcting the Patent Right in any Patent Office proceeding or with appeal of a Patent Office decision therefrom, including with respect to any post-grant proceeding, supplemental examination, post-grant review, inter partes review, reexamination, reissue, interference, or opposition proceeding in any Patent Office. For the avoidance of doubt, Prosecuting excludes any infringement suit or other legal Action to enforce a Patent Right or declaratory judgment suit or other legal Action initiated by a Third Party to challenge in court the validity or enforceability of a Patent Right. “Prosecute” and “Prosecution” shall each have a correlative meaning. 1.169 “Prosecuting Party” means the Party with the current right to Prosecute the applicable Patent Right as set forth in Section 8.3. 1.170 “Prosecution Contact” means a Party’s designated patent attorney or agent identified in a notice to the other Party (as may be updated from time to time) as its contact for communications between the Parties regarding the Prosecuting of any Arrowhead Patent Rights. 1.171 “Receiving Party” has the meaning set forth in Section 9.1.1.

23 1.172 “Registered Starting Material ([***])” means Arrowhead’s proprietary [***]used in the Manufacture of ARO-HBV. 1.173 “Regulatory Approval” means the approval (including supplements, amendments, pre- and post-approvals), license, registration or authorization of the applicable Regulatory Authority necessary for the marketing and sale of drug product in a country or jurisdiction, including any and all pricing and reimbursement approvals that are necessary to obtain in such country or jurisdiction to launch a drug product (even if such approvals are not legally required to launch such drug product in such country or jurisdiction). For purposes of illustration, in addition to approval of a Drug Application: Regulatory Approval in France includes approval of a Drug Application and publication of the reimbursed price level in the official journal and registration on a reimbursement list by or on behalf of Comité Economique des Produits de Santé or Haute Autorité de Santé (or a successor agency); Regulatory Approval in Italy includes publication of reimbursement in the Government’s Offical Gazette (by Agenzia Italiana del Farmaco or a successor agency); Regulatory Approval in Germany includes execution of contract with the head association of sick funds (GKV-Spitzenverband, Gesetzlichen Krankenversicherung, or a successor agency); Regulatory Approval in Spain (includes authorization by La Comisión Interministerial de Precios de los Medicamentos or La Comisión Nacional para el Uso Rational de los Medicamentos, or a successor agency) for national patient access to reimbursement by or on behalf of a Governmental Authority; and Regulatory Approval in the United Kingdom includes approval by the National Institute for Health and Care Excellence (or a successor agency) to obtain mandatory funding to enable broad market access. 1.174 “Regulatory Authority” means any federal, national, multinational, state, provincial or local regulatory agency, department, bureau or other governmental entity with authority over the registration or authorization or marketing and sale of a medicinal product in a country, such as the FDA in the United States, EMA in the EU, and MHLW in Japan. 1.175 “Regulatory Exclusivity Right” means a right or protection, granted by a Regulatory Authority in a jurisdiction, providing with respect to a product in such jurisdiction marketing or data exclusivity that prevents the Regulatory Authority from accepting or approving a Drug Application (whether new or abbreviated), submitted by a Person other than GSK (or any of its Affiliates or Sublicensees), such as through new molecular entity, new use or indication exclusivity, new formulation exclusivity, non-patent related pediatric exclusivity or orphan drug exclusivity granted by the FDA, or an exclusive right to sell pursuant to the data exclusivity provisions under EC Directives 2004/27/EC and 2001/83/EC and Regulation 726/2004/EC, or marketing exclusivity granted in respect of pediatric studies under Regulation 1901/2006, or Section 505A(a) of the FD&C Act. 1.176 “Regulatory Filing” means any documentation comprising or relating to or supporting any filing or application with any Regulatory Authority with respect to a Licensed Product, or its use or potential or investigative use in humans, including any documents submitted to any Regulatory Authority and all supporting data, including INDs, supportive documents enabling a clinical program, Drug Applications, safety and adverse event reports and all correspondence with any Regulatory Authority with respect to any Licensed Product

24 (including minutes of any meetings, telephone conferences or discussions with any Regulatory Authority). 1.177 “Relevant Factors” means all relevant factors that may affect the Development, Regulatory Approval, Manufacturing or Commercialization of a Licensed Product, including (as applicable): actual and potential issues of safety, tolerability, efficacy or stability; expected and actual product profile (including product modality, category and mechanism of action), as such or in comparison with the profile of other products and regimens; stage of development or life cycle status; actual and projected Development, Regulatory Approval, Manufacturing, and Commercialization costs, timelines and budgets; any issues regarding the Manufacturing of the Licensed Product; the likelihood of obtaining Regulatory Approvals for such Licensed Product; the timing of such Regulatory Approvals; the current guidance and requirements for Regulatory Approval for such Licensed Product and similar products and the current and projected regulatory status; labeling or anticipated labeling for such Licensed Product; the then current competitive environment and the likely competitive environment at the time of projected entry into the market; past performance of such Licensed Product or similar products; present and future market potential, as such or taking into account the relevant portfolio or pipeline; present and future relevant patient population; existing or projected pricing, sales, reimbursement, return on investment and profitability; pricing or reimbursement changes in relevant countries; proprietary position, strength and duration of patent protection, anticipated exclusivity and freedom to operate hurdles; legal issues; and other relevant scientific, technical, operational, commercial or economic factors. 1.178 “Representatives” has the meaning set forth in Section 9.1.1. 1.179 “Right of Reference” has the meaning set forth for such term in 21 C.F.R. § 314.3(b) or an equivalent right of access or reference under any Applicable Law in any other jurisdiction outside the United States. 1.180 “RNAi Trigger” means an oligonucleotide comprised of a strand of RNA or chemically modified RNA designed to hybridize to the HBV genome to effect silencing through RNA interference of HBV genomic transcripts. 1.181 “[***]” has the meaning set forth in Section 1.182. 1.182 “[***]” means [***]. 1.183 “[***]” has the meaning set forth in Section. 1.184 “Royalty Term” means, on a Licensed Product-by-Licensed Product and country-by- country basis, the period from the date of the First Commercial Sale of such particular Licensed Product by or on behalf of a Selling Party in such country, until the later of (a) the expiration of the last Valid Claim of [***] which Covers [***] of such Licensed Product in such country; (b) the termination or expiration of Regulatory Exclusivity Rights protecting such Licensed Product in such country; or (c) [***] years from the date of First Commercial Sale of such Licensed Product in such country.

25 1.185 “S Trigger Target Sequence” means [***]. 1.186 “Securitization Transaction” has the meaning set forth in Section 15.1.2. 1.187 “Selling Party” has the meaning set forth in Section 1.130. 1.188 “Specific Arrowhead Patent Rights” has the meaning as set forth in Section 8.3.3(a). 1.189 “Sublicensee” means any Third Party to which GSK or any of its Affiliates has granted or grants any sublicense under any of the rights or licenses granted to GSK under Section 2.1.1 or Section 2.1.2 (and any further sublicensee of such Third Party (regardless of the number of tiers, layers or levels of sublicenses or covenants not to sue of such rights)), in each case, as permitted under this Agreement; provided that “Sublicensee” shall exclude distributors and subcontractors performing activities by or on behalf of GSK or its Affiliates, as applicable. 1.190 “Target Sequence” means the sequence of an HBV genome or HBV genomic transcript(s) to which the guide strand of an RNAi Trigger is intended to hybridize. 1.191 “Targeting Ligand” means a compound or moiety suitable for targeting the delivery of an RNAi therapeutic agent to an hepatocyte. 1.192 “Taxes” means federal, state, local or non-U.S. income, gross receipts, license, payroll, employment, excise, severance, stamp, occupation, premium, windfall profits, environmental, customs duties, capital stock, franchise, profits, withholding, social security, unemployment, disability, real property, personal property, sales, use, transfer, registration, VAT and Indirect Taxes, alternative or add-on minimum, estimated, or other tax of any kind whatsoever, including any interest, penalty, or addition thereto, whether disputed or not. 1.193 “Term” has the meaning set forth in Section 13.1. 1.194 “Terminated Product” means, in the event of any termination of this Agreement by Arrowhead pursuant to Section 13.2.2 or by GSK pursuant to Section 13.5, any Licensed Product that is actively being Developed or Commercialized by or on behalf of GSK (or any of its Affiliates) immediately prior to the effective date of such termination; provided that Terminated Product shall solely refer to the form of such Licensed Product that is being so Developed or Commercialized by or on behalf of GSK (or any of its Affiliates) immediately prior to such effective date of termination. 1.195 “Territory” means the entire world, including all of its countries and their possessions and territories. 1.196 “Third Party” or “Third-Party” means any person, entity, or other party other than a Party to this Agreement or any of its Affiliates. 1.197 “Third Party Action” has the meaning set forth in Section 8.7.1.

26 1.198 “Third Party Payments” has the meaning set forth in Section 6.4.5(d). 1.199 “Trademark Rights” means all registered and unregistered trademarks (including all common law rights thereto), service marks, trade names, brand names, logos, taglines, slogans, certification marks, internet domain names, trade dress, corporate names, business names and other indicia of origin, together with the goodwill associated with any of the foregoing, and all applications, registrations, extensions, and renewals thereof throughout the world, and all rights therein provided by international treaties and conventions. 1.200 “United States”, “US” or “U.S.” means the United States of America, including its territories and possessions. 1.201 “Valid Claim” means a claim (a) of any unexpired Patent Right issued or granted by a Patent Office that has not been revoked or held unenforceable or invalid by a decision of a court or Governmental Authority of competent jurisdiction from which no appeal can be taken, or with respect to which an appeal is not taken within the time allowed for appeal, and that has not been disclaimed or admitted to be invalid or unenforceable through reissue, disclaimer, or otherwise, or (b) of any Patent Right that is being prosecuted in good faith and has not been abandoned or finally disallowed without the possibility of appeal or re- filing of the application and has been pending for less than [***] years. 1.202 “VAT and Indirect Taxes” means any value added, sales, purchase, turnover or consumption tax as may be applicable in any relevant jurisdiction, including but not limited to value added tax chargeable under legislation implementing E.U. Council Directive 2006/112/EC on the common system of value added tax. 1.203 “X Trigger Target Sequence” means [***]. ARTICLE II: LICENSE GRANTS 2.1 Grants. 2.1.1 Development License. Subject to the terms and conditions of this Agreement, Arrowhead hereby grants to GSK an exclusive (even as to Arrowhead, except to the extent Arrowhead expressly retains or is expressly granted back rights under this Agreement), worldwide license, with the right to sublicense in accordance with Section 2.1.3, under Arrowhead Intellectual Property, to research and have researched and to Develop and have Developed Licensed Constructs and Licensed Products in the Field in the Territory, and to make and Manufacture, have made and Manufactured, use, have used, and import and have imported Licensed Constructs and Licensed Products for such purposes. The license rights granted under this Section 2.1.1 shall commence on the Amended Effective Date and run throughout the Term hereof, subject to the termination provisions under Article XIII. 2.1.2 Commercialization License. Subject to the terms and conditions of this Agreement, Arrowhead hereby grants to GSK an exclusive (even as to Arrowhead, except to the extent Arrowhead expressly retains or is expressly granted back rights

27 under this Agreement), worldwide license, with the right to sublicense in accordance with Section 2.1.3, under the Arrowhead Intellectual Property, to Commercialize and have Commercialized, offer for sale and sell, and have offered for sale and sold Licensed Products for use in the Field in the Territory, and to Manufacture, have Manufactured, use, have used, and import and have imported Licensed Constructs or Licensed Products for such purposes. The license rights granted under this Section 2.1.2 shall commence on the Amended Effective Date and continue, on a Licensed Product-by-Licensed Product and country-by-country basis, throughout the Term hereof, subject to the termination provisions under Article XIII. 2.1.3 Sublicensing. In the event that GSK grants any sublicense of the license rights granted to GSK under this Section 2.1 to any Affiliates or any Third Parties, GSK shall remain responsible for its obligations under this Agreement and shall be responsible for the performance of the relevant sublicensee and the compliance by such sublicensee with the terms and conditions of this Agreement, and Arrowhead will have the right to proceed directly against GSK without any obligation to first proceed against the Sublicensee. Any sublicense granted by GSK under this Section 2.1 to any Sublicensee shall refer to this Agreement and shall not conflict with GSK’s obligations under this Agreement, including obligations of confidentiality, non-disclosure and non-use of Confidential Information, and allocation of intellectual property rights that are at least as restrictive or protective of Confidential Information and intellectual property rights (including with respect to GSK Agreement IP, Arrowhead Agreement IP and Joint Agreement IP) as set forth in this Agreement. With respect to any such Sublicensee that includes the grant to such Third Party of exclusive rights to Develop or Commercialize any Licensed Products, GSK will, within a reasonable time period after granting such sublicense, provide a copy of the sublicensing agreement to Arrowhead, which agreement may be redacted to omit any terms not relevant to determining GSK’s and the Sublicensee’s obligations under this Agreement. 2.2 Licenses Constitute IP under Bankruptcy Code. All rights and licenses granted under or pursuant to any section of this Agreement by one Party to the other, including Section 2.1 hereof, are, and shall otherwise be deemed to be, for the purpose of Section 365(n) of the Bankruptcy Code (or comparable provisions of laws of other jurisdictions) rights to “intellectual property” as defined in Section 101(35A) of the Bankruptcy Code (or comparable provisions of laws of other jurisdictions). Each Party hereby acknowledges, on behalf of itself and its Affiliates, “embodiments” of intellectual property pursuant to the Bankruptcy Code include the following: (a) data from the research and Development of Licensed Products; (b) Licensed Constructs and Licensed Product samples and inventory; (c) Licensed Product formulations; (d) laboratory notebooks and records from either Party’s research relating to any Licensed Constructs or Licensed Product, including from the Development Plan; (e) results from Clinical Trials of Licensed Products and the Licensed Products therein; (f) Regulatory Filings and Regulatory Approvals relating to Licensed Products; and (g) marketing, advertising and promotional materials relating to Licensed Products.

28 2.3 Rights in Combination Products. Notwithstanding the terms of any license grant or covenant under this Agreement, no rights will be conveyed or granted by one Party to another hereunder to (a) an Active Ingredient of any Combination Product, whether in Development or Commercialized, where the Active Ingredient is not a Licensed Construct, (b) a product of such Combination Product that is not a Licensed Product, or (c) an Active Ingredient, other than a Licensed Construct, that is otherwise used in combination with a Licensed Product in pre-clinical research, Clinical Trials or in accordance with an approved product label. 2.4 No Other Rights. No rights other than those expressly set forth in this Agreement are granted by one Party to the other Party hereunder, and no additional rights shall be deemed granted to either Party by implication, estoppel, or otherwise, with respect to any Patent Rights, Know-How, or other intellectual property rights. ARTICLE III: DEVELOPMENT 3.1 Completion of Option Right Development Plan. The Parties acknowledge and agree that, prior to the Amended Effective Date, pursuant to the Original Agreement, (a) Janssen exercised the Option Right, and the Option Right Development Term (each, as defined under the Original Agreement) commenced, on September 15, 2021 by delivery of notice to Arrowhead in accordance with Section 4.1 of the Original Agreement; (b) Janssen subsequently terminated the activities under the Option Right Development Plan (as defined under the Original Agreement), and the Option Right Development Term ended, effective as of January 25, 2023 in accordance with Section 4.7 of the Original Agreement; and (c) GSK has as of the Amended Effective Date or shall have after the Amended Effective Date no obligations to Arrowhead (or any of its Affiliates) with respect to (i) any amounts due and payable to Arrowhead in accordance with Section 4.7 of the Original Agreement and (ii) any costs or expenses incurred by or on behalf of Arrowhead (or any of its Affiliates) in connection with the performance of any research or other Development activities under the Option Right Development Plan or otherwise in connection with the performance of any other activities under Article IV of the Original Agreement. 3.2 Development. Except as expressly provided otherwise under this Agreement, from and after the Amended Effective Date, GSK shall have the exclusive right, the sole responsibility and the sole decision-making authority (directly or through one or more of its Affiliates or Sublicensees, or any Third Party subcontractors selected by any of the foregoing) for all Development activities with respect to any Licensed Constructs or Licensed Products in the Territory, including to conduct any non-clinical studies or Clinical Trials that GSK (or any of its Affiliates or Sublicensees) believes appropriate in order to obtain Regulatory Approval for Licensed Products in the Territory. The Parties acknowledge that the Development Plan as of the Amended Effective Date that is attached to this Agreement as Schedule 3.2, is preliminary and provides high-level plans for Development activities of any Licensed Product in the Territory as of the Amended Effective Date, and is provided to Arrowhead for informational purposes only. The Parties further acknowledge and agree that (a) for clarity, the Development Plan shall not limit or otherwise restrict GSK’s (or any of its Affiliates’ or Sublicensees’) right to conduct any Development activities with respect to any Licensed Constructs or Licensed Products in

29 the Territory, and GSK shall have the sole right to make all decisions with respect to the implementation of the Development Plan or any other Development activities with respect to any Licensed Constructs or Licensed Products in the Territory in accordance with this Section 3.2. Notwithstanding the foregoing, GSK may notify Arrowhead of any update or amendment of the Development Plan; provided, however, that, subject to Section 3.7, GSK shall have no obligation to provide Arrowhead any such update or amendment to the Development Plan. 3.3 Development Diligence. 3.3.1 GSK Development Diligence. Following the Amended Effective Date, GSK shall use Commercially Reasonable Efforts to (a) conduct Development activities required in order to support an application for Regulatory Approval for a Licensed Product in the United States, the European Union, China and Japan for at least one (1) indication; and (b) in the event that such Development activities are successful and based on the available data from such Development activities, seek Regulatory Approval for such Licensed Product in the United States, the European Union, China and Japan for at least one (1) indication. Notwithstanding anything to the contrary set forth herein, Arrowhead acknowledges and agrees that GSK’s diligence obligation under this Section 3.3.1 shall not be construed in any way such as to require GSK [***]. 3.3.2 Development Compliance. Each of GSK and its Affiliates, Sublicensees, and its and their Third-Party subcontractors shall conduct its respective Development activities under this Agreement in a good scientific manner and in compliance with Applicable Law, including any Applicable Laws regarding environmental, safety, industrial hygiene, Clinical Investigation Laws, Good Laboratory Practice, Good Clinical Practice or pharmacovigilance requirements, as applicable; provided, however, that, no such Person shall be obligated to undertake or continue any Development activities with respect to any Licensed Product if such Party reasonably determines that performance of such Development activity would violate any Applicable Law or pose an unacceptable safety risk to Clinical Trial subjects. 3.4 Transfer of Know-How. As reasonably requested by GSK and as is necessary to support the Development or obtaining Regulatory Approvals for any Licensed Product, Arrowhead shall make available to GSK and shall permit GSK to make copies at GSK’s expense the Arrowhead Know-How recorded in any form (including laboratory notebook entries, database entries, monographs, reports, and slide presentations), including all clinical data resulting from the Ongoing Phase 1/2 Study (as defined in the Original Agreement). Notwithstanding any term of this Agreement to the contrary, Arrowhead shall not be obliged to provide or otherwise disclose to GSK its trade secrets related solely to trigger selection or Licensed Construct design. GSK shall reimburse [***] of Arrowhead’s FTE Costs and Out-of-Pocket Costs incurred in the performance of such activities pursuant to this Section 3.4, subject to a budget to be mutually agreed by the Parties prior to Arrowhead commencing any such activities.

30 3.5 Regulatory Approvals and Filings. 3.5.1 INDs for New Clinical Studies and other Regulatory Filings. From and after the Amended Effective Date, GSK shall have the exclusive right, the sole responsibility and the sole decision-making authority (directly or through one or more of its Affiliates or Sublicensees, or any Third Party subcontractors selected by any of the foregoing) for all regulatory matters with respect to any Licensed Products in the Territory, including (a) developing a registration strategy for any Licensed Products in the Territory; (b) filing (if applicable), maintaining, holding and owning (i) all INDs and other Regulatory Filings for any Clinical Trials of any Licensed Product initiated after the Amended Effective Date, (ii) all other Regulatory Filings for any Licensed Product following the Amended Effective Date, (iii) all approvals or authorizations resulting from the filings described in the preceding clauses (i) and (ii), including all Regulatory Approvals for any Licensed Products; and (c) all interactions with Regulatory Authorities in connection with the Development, Manufacture or Commercialization of any Licensed Product, including with respect to any Regulatory Filings or Regulatory Approvals with respect thereto. 3.5.2 Assistance. (a) Upon GSK’s reasonable request, at no additional cost to GSK, Arrowhead shall, and shall cause its Affiliates to, provide to GSK (or its designee) any documents or other materials in the possession or Control of Arrowhead or any of its Affiliates as may be reasonably necessary for GSK (or any of its Affiliates or Sublicensees) to obtain Regulatory Approvals for such Licensed Products in the Territory. (b) Without limiting Section 3.5.2(a), upon GSK’s reasonable request, Arrowhead shall provide reasonable support to GSK (or any of its Affiliates), as may be reasonably necessary, in obtaining Regulatory Approvals for any Licensed Products and in the activities in support thereof; provided that GSK shall reimburse [***] of Arrowhead’s FTE Costs and Out-of-Pocket Costs incurred in the performance of such support activities pursuant to this Section 3.5.2(b) (but, for clarity, except with respect to any provision of documents or other materials in accordance with Section 3.5.2(a)), subject to a budget to be mutually agreed by the Parties prior to Arrowhead commencing any such support activities. (c) Notwithstanding any term of this Agreement to the contrary, Arrowhead shall not be obliged to provide or otherwise disclose to GSK its trade secrets related solely to trigger selection or Licensed Construct design. 3.6 Regulatory Reporting. 3.6.1 Responsibility. Except as expressly provided otherwise herein from and after the Amended Effective Date, any reports (including adverse event reports) made to any

31 Regulatory Authority in connection with any Development activities for any Licensed Product, shall be made exclusively by GSK and in accordance with the terms and conditions of the Pharmacovigilance Agreement. In the event that information from Arrowhead is reasonably required for any such report to be submitted to any Regulatory Authority by GSK (or any of its Affiliates or Sublicensees) with respect to any Licensed Product, upon GSK’s reasonable request, Arrowhead shall, and shall cause its Affiliates to, provide to GSK (or its designee) any such information that is in the possession or Control of Arrowhead or any of its Affiliates as may be reasonably necessary for GSK (or any of its Affiliates or Sublicensees) to prepare and submit such report to the applicable Regulatory Authority in the Territory at no additional cost to GSK. Notwithstanding any term of this Agreement to the contrary, Arrowhead shall not be obliged to provide or otherwise disclose to GSK its trade secrets related solely to trigger selection or Licensed Construct design. 3.6.2 Adverse Event Reporting. Promptly after a Party becomes informed of any serious adverse event in any Clinical Trial involving a Licensed Product, it shall notify the other Party and such notifying Party shall thereafter continue to provide additional information to the other Party relevant to such serious adverse event, including to the extent necessary for such other Party to comply with all Applicable Laws (including securities laws or regulations and the applicable rules of any public stock exchange). Arrowhead acknowledges and agrees that GSK, as the Party having the right to hold any Drug Application for any Licensed Product hereunder, may be required to submit information and file reports to various Regulatory Authorities on a Licensed Product. Upon GSK’s reasonable request, Arrowhead shall, and shall cause its Affiliates to, at no additional cost to GSK, (a) provide GSK with all adverse event information and safety-related data that is in the possession or Control of Arrowhead or any of its Affiliates from any pre-clinical laboratory, animal toxicology or pharmacology studies, or Clinical Trials, as reasonably may be necessary or expected to be necessary for GSK (or any of its Affiliates or Sublicensees) to comply with all Applicable Laws pertaining to the Licensed Product; and (b) report and provide such information to GSK in such a manner and time so as to enable GSK (or any of its Affiliates or Sublicensees) to comply with all Applicable Laws. Each Party shall handle all serious adverse events information and other safety data that comes into its possession during Development and Commercialization of any Licensed Product hereunder in accordance with all Applicable Laws. 3.6.3 Global Safety Database. As between the Parties, GSK shall establish a global safety database for each Licensed Product Developed hereunder and shall, in relation to such Licensed Product, maintain in the global safety database information relating to adverse events, pregnancy reports, special situation reports, and any other information relating to other adverse events that GSK (or any of its Affiliates or Sublicensees) decides to include at its reasonable discretion, including possible safety data within the Arrowhead Know-How. GSK (and any of its Affiliates or Sublicensees) will use this database for regulatory reporting and for responding to safety queries from Regulatory Authorities. Promptly after the

32 Amended Effective Date and during the Term, Arrowhead shall, and shall cause its Affiliates and Third-Party contractors to, disclose all information relating to adverse events and pregnancy reports from clinical use of Licensed Product in its or their possession to GSK for storage into its global safety database. 3.6.4 Pharmacovigilance Agreement. In the event that, during the Term, either Party reasonably determines that it is necessary in connection with the performance of either Party’s obligations under this Agreement for the Parties to enter into a written pharmacovigilance agreement in order to define safety data exchange processes and procedures between the Parties concerning adverse events reactions with respect to Licensed Constructs and Licensed Products, then, upon such Party’s request, the Parties shall negotiate in good faith, mutually agree and enter such pharmacovigilance agreement (the “Pharmacovigilance Agreement”). The Pharmacovigilance Agreement shall govern such safety data exchange processes and, with respect to any Licensed Products, sufficient to permit each Party and its Affiliates and subcontractors or sublicensees, as the case may be, to comply with requirements of Applicable Laws pertaining to drug safety and pharmacovigilance, including, to the extent applicable, those obligations contained in Health Care Laws imposed by Regulatory Authorities. The Pharmacovigilance Agreement shall reflect that, as between the Parties, GSK shall own and maintain a comprehensive (global) safety database of adverse events, pregnancy reports, and other safety data reported anywhere in the world from human use of any Licensed Products anywhere in the Licensed Territory. 3.7 Progress Reporting. Following the Amended Effective Date, (a) (i) at least once every six (6) months until the date on which the first Licensed Product has obtained Regulatory Approval in each of [***], and (ii) after the date on which the first Licensed Product has obtained Regulatory Approval in [***] and until such time as GSK, its Affiliates and its Sublicensees cease all Development activities with respect to all Licensed Products in the Territory for a continuous period of at least twelve (12) months, once per Calendar Year, GSK will provide Arrowhead’s Alliance Manager with a reasonable high-level written summary of the activities and progress with respect to the Development of any Licensed Products that GSK and its Affiliates or Sublicensees have performed or caused to be performed since the last such summary delivered pursuant to this Section 3.7; provided, however, that, notwithstanding anything to the contrary set forth in this Agreement, in no event shall GSK have any obligation under this Agreement to disclose or otherwise provide to Arrowhead any data, material or other information with respect to any other construct, compound or product of GSK (or any of its Affiliates or Sublicensees) that is not a Licensed Construct or Licensed Product, including with respect to any other Active Ingredients that are not Licensed Constructs within any Combination Product. For the avoidance of doubt, the Parties acknowledge and agree that any high-level summaries or other information provided by GSK under this Section 3.7 shall be deemed to be the Confidential Information of GSK.

33 3.8 Auditing. If any Regulatory Authority (a) contacts Arrowhead (or any of its Affiliates) with respect to the alleged improper Development, Manufacture or Commercialization of any Licensed Construct or Licensed Product in the Territory; (b) conducts, or gives notice of its intent to conduct, an inspection at Arrowhead’s or its Affiliate’s facilities used in the Development or Manufacturing of any Licensed Construct or Licensed Product in the Territory; or (c) takes, or gives notice of its intent to take, any other regulatory action with respect to any activity of Arrowhead (or its Affiliates, licensees or sublicensees, as applicable) that could reasonably be expected to adversely affect any Development, Manufacture or Commercialization with respect to any Licensed Construct or Licensed Product in the Territory, then Arrowhead will promptly notify GSK of such contact, inspection or notice. 3.8.1 Compliance Inspections. With respect to any facility or site at which Arrowhead, any of its Affiliates or its Third Party (sub)contractors conducts any Manufacturing, clinical or regulated (e.g., under GLP, GCP, or GMP) Development activities, including Manufacturing clinical supply for use in humans, pursuant to this Agreement, GSK shall have the right, as permitted by and subject to the terms and conditions of any possible applicable agreement with a Third Party (sub)contractor or as otherwise expressly permitted by the applicable Third Party (sub)contractor, at its expense, upon reasonable written notice to Arrowhead (and if applicable, such Affiliate or Third Party (sub)contractor), and during normal business hours, to inspect such facility or site and any records relating thereto, once per year or more often with cause, to verify Arrowhead’s compliance with the terms of this Agreement and with all Applicable Laws, including GLP, GCP, and GMP, and current standards for pharmacovigilance practice. Such inspection shall be subject to the confidentiality provisions set forth in Article IX. In the event that such inspection would result in the disclosure of confidential information which is not protected by the confidentiality provisions set forth in Article IX, an appropriate confidentiality agreement shall be entered into. After any such inspection, GSK shall provide written observations to Arrowhead. In the event that non-compliance with the terms of this Agreement or with Applicable Laws were observed, Arrowhead shall promptly take or, as the case may be, use Commercially Reasonable Efforts to cause the applicable Third Party to promptly take the necessary actions to remediate such non-compliance and shall keep GSK’s Alliance Manager informed of such actions. Arrowhead agrees to use Commercially Reasonable Efforts to include in any contract or other written arrangement with its Third Party (sub)contractors, a clause permitting GSK to exercise its rights under this Section 3.8.1. 3.8.2 Regulatory Audits. Arrowhead shall cooperate in good faith in the event any Regulatory Authority inspects any site where Clinical Trials or Manufacturing of clinical supplies of Licensed Products are conducted by or on behalf of Arrowhead pursuant to this Agreement, whether such Audited Site is Arrowhead’s or its Affiliate’s or contractor’s or subcontractor’s hereunder, as permitted by and subject to the terms and conditions of any applicable agreement with a Third Party or as otherwise expressly permitted by the applicable Third Party. Arrowhead shall notify GSK within two (2) Business Days after receiving notification of any

34 Regulatory Authority inspection, which relates to or reasonably could relate to the Licensed Product or Clinical Trials for the Licensed Product, at any site where Clinical Trials or Manufacturing of clinical supplies of Licensed Products are conducted. Taking into account the timing and notice provided by the applicable Regulatory Authority, and the terms of any applicable agreements with Third Parties and Applicable Law, GSK shall be given a reasonable opportunity to assist in the preparation of the Audited Site for inspection, where appropriate, and to attend any inspection by any Regulatory Authority of the Audited Site, and the summary, or wrap-up, meeting with a Regulatory Authority at the conclusion of such inspection. If such attendance would result in the disclosure of Arrowhead’s, its Affiliate’s or a Third Party’s confidential information unrelated to the subject matter of this Agreement, an appropriate confidentiality agreement covering such unrelated subject matter shall be entered into. In the event that any Audited Site is found to be non-compliant with one or more Applicable Laws, Good Laboratory Practice, Good Clinical Practice, Good Manufacturing Practice, or current standards for pharmacovigilance practice, Arrowhead shall, promptly and in any event within twenty (20) Business Days after receiving notification of such non- compliance, submit to GSK a CAPA plan and shall use Commercially Reasonable Efforts to cause such non-compliant Audited Site to implement such CAPA plan promptly after submission. Arrowhead agrees to use Commercially Reasonable Efforts to include in any agreement or other written arrangement entered into after the Amended Effective Date with its applicable Third Party contractors or subcontractors (as the case may be), a clause permitting GSK to exercise its rights under this Section 3.8.2. 3.9 Rights of Reference and Access to Data. Arrowhead hereby grants to GSK, and GSK shall have (directly and through any of its Affiliates or Sublicensees), a Right of Reference with respect to INDs, drug master files, if any, and any other Regulatory Filings (whether made before or during the Term hereof) Controlled by Arrowhead related to any Licensed Products, for use by GSK (or any of its Affiliates or Sublicensees) in connection with the Development and Commercialization of any Licensed Product in accordance with this Agreement. Accordingly, Regulatory Authorities considering any Regulatory Filing relating to a Licensed Product that is being Developed hereunder shall be permitted to rely on and otherwise use the applicable information in such INDs or other Regulatory Filings. Arrowhead or its Affiliate shall provide a signed statement to this effect, if requested by GSK, in accordance with 21 C.F.R. § 314.50(g)(3) or the equivalent as required in any other country or region of the world, or otherwise provide appropriate notification of such right of GSK to the applicable Regulatory Authority. GSK (directly and through any of its Affiliates or Sublicensees) shall also have a right to review, access and request copies of such Regulatory Filings and any Know-How (including data) therein and use such Know- How in connection with the performance of GSK’s obligations and exercise of its rights under this Agreement, including inclusion of such Know-How in its own Regulatory Filings for Licensed Products. 3.10 Suspension of Clinical Trial for Safety Reason. Notwithstanding anything to the contrary herein, if an independent safety board determines that any Clinical Trial of a Licensed Product under the Development Plan would pose an unacceptable safety risk for

35 any subjects or patients participating in such study, the Party conducting such Clinical Trial shall not be obligated to continue such Clinical Trial. Either Party may delay or suspend any Development activities with respect to an ongoing Clinical Trial of a Licensed Product conducted by or on behalf of such Party, if such Party reasonably believes that such Clinical Trial would pose an unacceptable safety risk. 3.11 Records. 3.11.1 Maintenance of Research Records. Each of the Parties shall maintain, or cause to be maintained by its Affiliates, Sublicensees and Third Party (sub)contractors, records of its respective Collaboration Activities in material compliance with Applicable Law (including the requirements of GCP, GLP and GMP, in each case to the extent applicable), and the requirements of its corporate records retention policies consistent therewith. Such records shall be complete and accurate and shall fully and properly reflect all work done and results achieved in the performance of the Collaboration Activities in a manner appropriate for any regulatory purpose and, when applicable and permitted under this Agreement, for use in connection with the Prosecution of Patent Rights. Such records shall be retained for the longer of either: (a) such period as is required by such retaining Party’s corporate record retention policies; (b) such period as may be required by Applicable Law; or (c) the Term of this Agreement, unless a Party first offers to deliver such records to the other Party for its keeping, and delivers to such Party any records it may reasonably request, before destroying or disposing of such records. 3.11.2 Access to Records. Each Party shall have the right, at mutually agreed times during normal business hours on Business Days and upon reasonable notice, to obtain from the other Party access to and copies (at its own cost) of the records maintained by the other Party pursuant to Section 3.11.1 solely to the extent relating to any Licensed Product or any Development, Manufacturing, or Commercialization activities hereunder or any intellectual property or associated rights licensed or obtained hereunder, to the extent useful or required to (a) enable the requesting Party to conduct reasonable diligence on matters potentially giving rise to liability on the part of the requesting Party according to Applicable Law or the requirements of this Agreement, or to conduct a defense of itself with respect to any such liability, if and to the extent that a fact, circumstance or event has arisen that gives the requesting Party a reasonable basis to believe that it has or may incur such liability; (b) to meet its obligations to Regulatory Authorities or to comply with Applicable Laws with respect to a Licensed Product; (c) to Prosecute or enforce any Patent Rights hereunder; or (d) to otherwise Exploit any rights hereunder, 3.12 Conditional Subcontracting. A Party may subcontract any of its Development activities hereunder (or, with respect to GSK (directly or through one or more of its Affiliates or Sublicensees), any of its other rights or other obligations under this Article III) to any Third Party; provided that such Party executes a written agreement with such Third Party subcontractor that contains, in all material respects, the applicable obligations and covenants hereunder, including obligations of confidentiality, non-disclosure and non-use

36 of Confidential Information, and allocation of intellectual property rights that are at least as restrictive or protective of Confidential Information and intellectual property rights (including with respect to GSK Agreement IP, Arrowhead Agreement IP and Joint Agreement IP) as set forth in this Agreement. Notwithstanding the foregoing, from and after the Amended Effective Date, Arrowhead shall not (nor shall Arrowhead permit any of its Affiliates to) subcontract any of its obligations under this Article III or Section 5.2 to any Third Party, in each case, without the prior written consent of GSK. A Party engaging any Third Party subcontractor pursuant to this Section 3.12 shall remain liable for the performance of its obligations under this Agreement and be responsible for the performance of such Third Party subcontractor in connection with their performance of such Party’s obligations hereunder, including such Third Party subcontractor’s compliance with the applicable material terms of this Agreement. 3.13 Alliance Managers. Each Party shall designate a single alliance manager for coordinating interactions between the Parties regarding any activities contemplated under this Agreement (“Alliance Manager”). The Alliance Managers will be responsible for the day-to-day worldwide coordination of the Parties’ activities under this Agreement and will serve to facilitate routine communication between the Parties. The Alliance Managers shall have experience and knowledge appropriate for managers with such project management responsibilities. Each Party may change its designated Alliance Manager from time to time upon written notice to the other Party, which notice shall provide the contact details (including email address) for the Alliance Manager. ARTICLE IV: COMMERCIALIZATION 4.1 Commercial Diligence. Commencing upon GSK obtaining first Regulatory Approval for a given Licensed Product in a given country or jurisdiction in the Territory, GSK shall use Commercially Reasonable Efforts to Commercialize a Licensed Product in the country or jurisdiction where such Regulatory Approval has been obtained. 4.2 Commercialization Responsibilities. From and after the Amended Effective Date, GSK shall have the exclusive right, the sole responsibility and the sole decision-making authority (directly or through one or more of its Affiliates or Sublicensees, or any Third Party subcontractors selected by any of the foregoing) for all Commercialization activities with respect to any Licensed Products in the Territory, including booking all sales of Licensed Products in the Territory. For clarity, subject to Section 4.1, each decision whether and when to commercially launch any particular Licensed Product in any particular country or jurisdiction of the Territory shall be within the discretion of GSK (directly or through one or more Affiliates or Sublicensees). Arrowhead acknowledges that nothing herein prohibits GSK from donating reasonable and customary supplies of Licensed Products for access programs or humanitarian or charitable purposes. 4.3 Trademarks and International Nonproprietary Names. GSK (directly or through one or more of its Affiliates or Sublicensees) shall select its own trademarks under which it will Commercialize Licensed Products hereunder and will own the Trademark Rights associated therewith. GSK (directly or through one or more of its Affiliates or Sublicensees, or any Third Party subcontractors selected by any of the foregoing) shall be

37 solely responsible for the application for an international nonproprietary name in relation to any Licensed Product and for the resulting communication with the World Health Organization. 4.4 Conditional Subcontracting. GSK (directly or through one or more of its Affiliates or Sublicensees) may subcontract any of its Commercialization activities or any of its other rights or other obligations under this Article IV to any Third Party; provided that GSK executes a written agreement with such Third Party subcontractor that contains, in all material respects, the applicable obligations and covenants hereunder, including obligations of confidentiality, non-disclosure and non-use of Confidential Information, and allocation of intellectual property rights that are at least as restrictive or protective of Confidential Information and intellectual property rights (including with respect to GSK Agreement IP, Arrowhead Agreement IP and Joint Agreement IP) as set forth in this Agreement. GSK shall remain liable for the performance of its obligations under this Agreement and be responsible for the performance of such Third Party subcontractor in connection with their performance of GSK’s obligations hereunder, including such Third Party subcontractor’s compliance with the applicable material terms of this Agreement. ARTICLE V: PRODUCT MANUFACTURE AND SUPPLY 5.1 Responsibility for Manufacture. From and after the Amended Effective Date, GSK shall have the exclusive right, the sole responsibility and the sole decision-making authority (directly or through one or more of its Affiliates or Sublicensees, or any Third Party subcontractors selected by any of the foregoing) for all Manufacturing activities with respect to any Licensed Constructs or Licensed Products in the Territory, including any Manufacturing of (a) clinical supplies of Licensed Constructs or Licensed Products for use in Clinical Trials conducted by or on behalf of GSK (or any of its Affiliates or Sublicensees); or (b) Licensed Constructs or Licensed Products otherwise for its Development or Commercialization of Licensed Constructs or Licensed Products under this Agreement. 5.2 Technical Transfer. As reasonably requested by GSK, Arrowhead shall make available to GSK and shall permit GSK to make copies at GSK’s expense of any CMC Know-How that (a) is in the possession or Control of Arrowhead or any of its Affiliates as may be reasonably necessary for GSK (or any of its Affiliates or Sublicensees) to (i) Manufacture any Licensed Construct or Licensed Product or (ii) obtain Regulatory Approvals for any Licensed Product in the Territory; and (b) GSK does not already have a right to receive from Janssen under the APA. Notwithstanding any term of this Agreement to the contrary, Arrowhead shall not be obliged to disclose to GSK its trade secrets related solely to trigger selection or Licensed Construct design. GSK shall reimburse [***] of Arrowhead’s FTE Costs and Out-of-Pocket Costs incurred in the performance of such activities pursuant to this Section 5.2, subject to a budget to be mutually agreed by the Parties prior to Arrowhead commencing such activities. Arrowhead hereby represents and warrants to GSK, as of the Amended Effective Date, that it complied in all material respects with its obligations under Section 7.4 of the Original Agreement to effect the transfer of such CMC Know-How to Janssen in accordance with the terms of such Section 7.4 of the Original Agreement.

38 5.3 Quality Assurance and Compliance with Laws. Arrowhead hereby represents and warrants to GSK, as of the Amended Effective Date, that any Licensed Products or Licensed Constructs supplied by Arrowhead to Janssen (or any of its Affiliates or other designees) for human use under the Original Agreement were Manufactured and supplied in accordance with the terms of Section 7.5 of the Original Agreement. ARTICLE VI: FINANCIAL PROVISIONS 6.1 Upfront Payment. In partial consideration of Arrowhead’s grant of the rights and licenses to GSK hereunder, within ten (10) Business Days following the Amended Effective Date and GSK’s receipt of a valid invoice in accordance with Section 7.1, GSK shall pay to Arrowhead a one-time, non-refundable, non-creditable payment of [***]. 6.2 Payments Under the Original Agreement. 6.2.1 Upfront Fee. The Parties acknowledge and agree that, prior to the Amended Effective Date, Janssen paid Arrowhead a one-time non-refundable payment of one hundred seventy-five million US dollars ($175,000,000) under and in accordance with the terms of the Original Agreement. 6.2.2 Option Right Payment. The Parties acknowledge and agree that, prior to the Amended Effective Date, Janssen paid Arrowhead a one-time non-refundable payment of three million US dollars ($3,000,000) upon Janssen’s exercise of the Option Right and the signing of the Option Right Development Plan by Arrowhead and Janssen, in each case, under and in accordance with the terms of the Original Agreement and as such terms are defined in the Original Agreement. 6.2.3 Milestones Payments under Original Agreement. The Parties acknowledge and agree that, prior to the Amended Effective Date, (a) the following milestone events were achieved by Janssen (or one of its Affiliates or sublicensees); and (b) Janssen paid Arrowhead the corresponding milestone amounts for each such milestone event, in each case ((a) and (b)), under and in accordance with the terms of the Original Agreement: Milestone Event Milestone Amount (USD) Upon dosing of the fifth patient in the CAM Cohort (as defined in the Original Agreement) $25,000,000 Upon dosing of the fifth patient in the first Phase 2 Clinical Trial of a Licensed Product $25,000,000
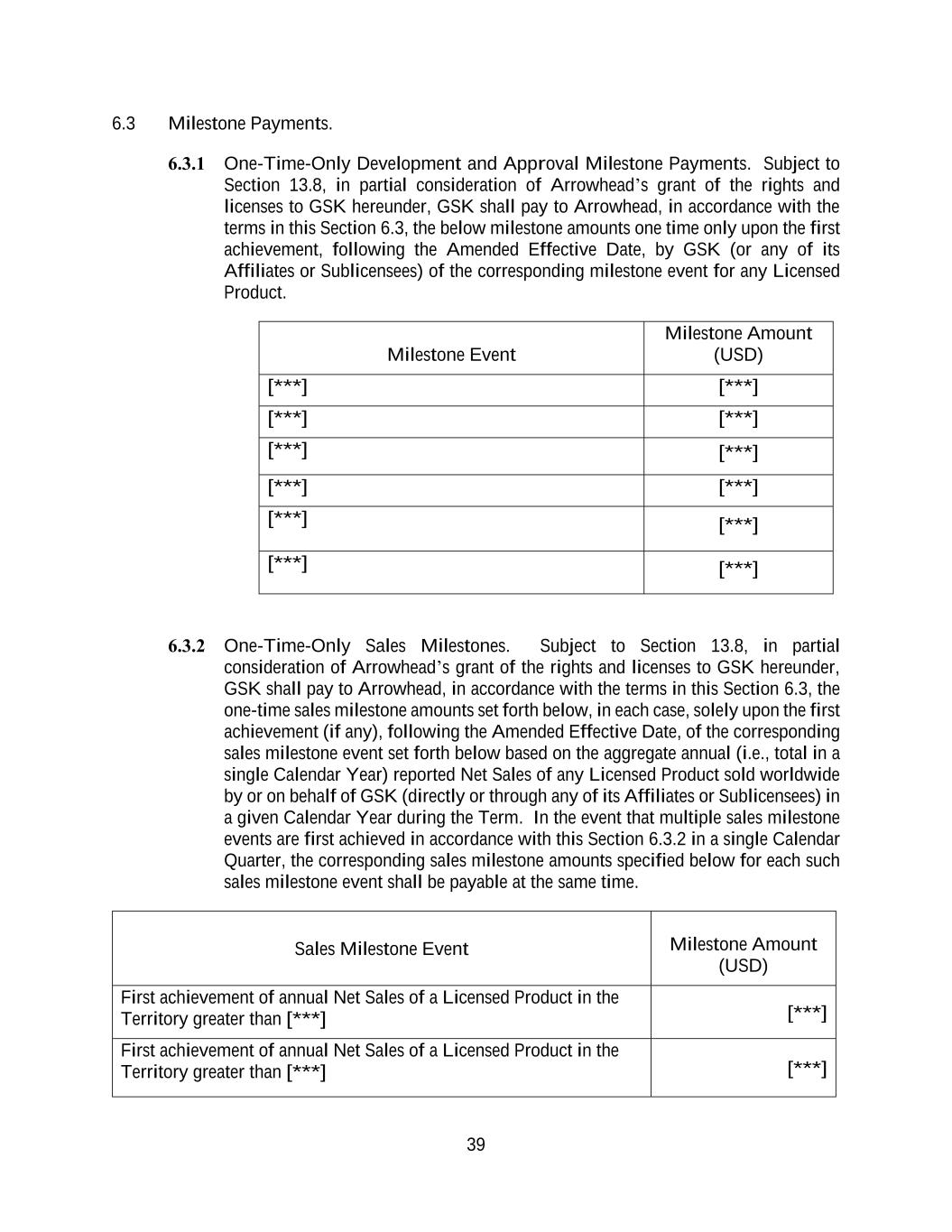
39 6.3 Milestone Payments. 6.3.1 One-Time-Only Development and Approval Milestone Payments. Subject to Section 13.8, in partial consideration of Arrowhead’s grant of the rights and licenses to GSK hereunder, GSK shall pay to Arrowhead, in accordance with the terms in this Section 6.3, the below milestone amounts one time only upon the first achievement, following the Amended Effective Date, by GSK (or any of its Affiliates or Sublicensees) of the corresponding milestone event for any Licensed Product. Milestone Event Milestone Amount (USD) [***] [***] [***] [***] [***] [***] [***] [***] [***] [***] [***] [***] 6.3.2 One-Time-Only Sales Milestones. Subject to Section 13.8, in partial consideration of Arrowhead’s grant of the rights and licenses to GSK hereunder, GSK shall pay to Arrowhead, in accordance with the terms in this Section 6.3, the one-time sales milestone amounts set forth below, in each case, solely upon the first achievement (if any), following the Amended Effective Date, of the corresponding sales milestone event set forth below based on the aggregate annual (i.e., total in a single Calendar Year) reported Net Sales of any Licensed Product sold worldwide by or on behalf of GSK (directly or through any of its Affiliates or Sublicensees) in a given Calendar Year during the Term. In the event that multiple sales milestone events are first achieved in accordance with this Section 6.3.2 in a single Calendar Quarter, the corresponding sales milestone amounts specified below for each such sales milestone event shall be payable at the same time. Sales Milestone Event Milestone Amount (USD) First achievement of annual Net Sales of a Licensed Product in the Territory greater than [***] [***] First achievement of annual Net Sales of a Licensed Product in the Territory greater than [***] [***]

40 First achievement of annual Net Sales of a Licensed Product in the Territory greater than [***] [***] First achievement of annual Net Sales of a Licensed Product in the Territory greater than [***] [***] 6.3.3 Each Milestone Amount Paid Once. In the event that a given milestone event set forth in this Section 6.3 is achieved, GSK shall pay to Arrowhead the milestone amount corresponding to each such milestone only once regardless of (a) the number of other Licensed Products to achieve such milestone event or (b) the number of times a given Licensed Product achieves such milestone event (including for additional indications). 6.3.4 Notice for Milestone Events. GSK shall inform Arrowhead in writing of the achievement of a given milestone event under this Section 6.3 as follows: (a) with respect to a given milestone event under Section 6.3.1, within [***] Business Days following the achievement of such milestone event in accordance with Section 6.3.1 by GSK or any of its Affiliates (or, in the event that such milestone event is achieved by or on behalf of a Sublicensee, within [***] Business Days following GSK’s receipt of notice from such Sublicensee with respect to the achievement of such milestone event, as applicable); or (b) with respect to a given milestone event under Section 6.3.2, within [***] days following the end of the applicable Calendar Quarter during which such milestone event is first achieved in accordance with Section 6.3.2, as applicable, which notice may be provided in connection with a royalty report delivered pursuant to Section 7.2. 6.3.5 Payment of Milestone Amounts. Following receipt of notice pursuant to Section 6.3.4 with respect to the achievement of a given milestone event in accordance with Section 6.3.1 or Section 6.3.2, as applicable, the corresponding milestone amount for such milestone event (as set forth in Section 6.3.1 or Section 6.3.2, as applicable) shall be due and payable by GSK to Arrowhead within [***] days following GSK’s receipt of a valid invoice in accordance with Section 7.1. 6.4 Royalty Payments. 6.4.1 Royalty Term. During the Term, the royalties for Licensed Products set forth in this Section 6.4 shall be paid on a Licensed-Product-by-Licensed-Product and country-by-country basis during the applicable Royalty Term for such Licensed Product in such country-of-sale. For purposes of determining whether a royalty threshold for a given Calendar Year has been attained in accordance with Section 6.4.2 or Section 6.4.3, as applicable, Net Sales that are generated by sales of a Licensed Product in a country for which the Royalty Term has expired shall be excluded from the total amount of Net Sales. Upon expiration of the Royalty Term in respect of a given Licensed Product in a given country-of-sale of the Territory, the licenses granted to GSK under Section 2.1 shall automatically convert to a fully paid-up, royalty-free, irrevocable, perpetual, non-exclusive and sublicensable (including through multiple tiers) license under the Arrowhead Intellectual

41 Property to Develop, Manufacture and Commercialize such Licensed Product in such country. 6.4.2 Royalty Rate. Subject to Section 6.4.1, Section 6.4.3, Section 6.4.4, Section 13.8 and any adjustments expressly permitted under Section 6.4.5 or Section, as applicable, in partial consideration of Arrowhead’s grant of the rights and licenses to GSK hereunder, during the applicable Royalty Term for a given Licensed Product in a given country in the Territory, GSK shall pay to Arrowhead royalties at the royalty rates on the tiers of aggregate reported Net Sales of all Licensed Products sold by GSK, its Affiliates and Sublicensees in the Territory during a given Calendar Year, as set forth in the below table. Aggregate Annual Net Sales of Licensed Products in the Territory in a Calendar Year Royalty Rate (percentage) Portion of annual Net Sales of Licensed Products for such Calendar Year up to and including [***] [***] Portion of annual Net Sales of Licensed Products for such Calendar Year greater than [***] up to and including [***] [***] Portion of annual Net Sales of Licensed Products for such Calendar Year greater than [***] up to and including [***] [***] Portion of annual Net Sales of Licensed Products for such Calendar Year greater than [***] [***] 6.4.3 Royalty Rate in Access Territory. In the event that GSK intends to Commercialize a Licensed Product in one or more countries in the Access Territory, the Parties shall timely enter into good faith negotiations to agree upon an equitable reduction in the royalty rate applicable to the aggregate reported Net Sales of such Licensed Product in such country or countries. Accordingly, and subject to any adjustments expressly permitted under Section 6.4.5 or Section 13.8, as applicable, GSK shall have the right to use the agreed upon royalty rate for the applicable aggregate reported Net Sales of Licensed Products in the Access Territory on a country-by-country basis therein. 6.4.4 Royalties Due Only Once. The obligation to pay royalties under this Agreement is imposed only once with respect to the same unit of a Licensed Product. 6.4.5 Adjustments to Royalties. (a) No Valid Claim. Subject to Section 6.4.5(e), in the event that, at any point during the applicable Royalty Term for a given Licensed Product in a given country in the Territory, the composition of matter of the Licensed Construct in such Licensed Product (or the applicable Targeting Ligand contained in the Licensed Construct in such Licensed Product) is not Covered by a Valid Claim of [***] in such country, then, the applicable royalty rates payable by GSK pursuant to Section 6.4.2 or Section 6.4.3, as

42 applicable, shall be reduced by [***] for such Licensed Product in such country. (b) Compulsory License. In the event that, at any time during the applicable Royalty Term for a given Licensed Product in a given country in the Territory, a license or other right is granted to a Third Party to use, sell (or offer for sale or contract to sell), import, export or otherwise Commercialize such Licensed Product in such country through a Government Order by a competent Governmental Authority within such country with a royalty rate lower than the royalties that otherwise would be payable by GSK pursuant to Section 6.4.2 or Section 6.4.3, as applicable, with respect to Net Sales of such Licensed Product in such country if sold by GSK (or any of its Affiliates or other Sublicensees), then, notwithstanding anything to the contrary set forth herein, the Parties will confer and in good faith negotiate an equitable reduction in the applicable royalty rate for calculating royalties payable to Arrowhead based on GSK’s and its Affiliates’ and Third Party sublicensees’ Net Sales of such Licensed Product in such country under Section 6.4.2 or Section 6.4.3, as applicable, taking into account the royalty rate payable by such Third Party under such compulsory license or other right granting such Third Party the right to use, sell (or offer for sale or contract to sell), import, export or otherwise Commercialize such Licensed Product in such country and remaining in effect for so long as such compulsory license or other right itself remains in effect. (c) Generic Competition. Subject to Section 6.4.5(e), on a Licensed Product- by-Licensed Product and country-by-country basis in the Territory, if one or more Generic Version of such Licensed Product is marketed and sold in such country by one or more Third Parties during any Calendar Quarter during the applicable Royalty Term for such Licensed Product and such Generic Version(s) of such Licensed Product have a market share of greater than [***] in such country (“Generic Competition”) (as determined based on the aggregate number of units of such Licensed Product and such Generic Version(s) sold in such country during such Calendar Quarter, as reported by a well-known reporting service agreed between the Parties acting reasonably and in good faith (e.g., IQVIA)), then, commencing in such Calendar Quarter, the applicable royalty rates under Section 6.4.2 or Section 6.4.3, as applicable, shall be reduced by [***] for such Licensed Product in such country for so long as such Generic Competition persists in such country. (d) Off-Set for Third-Party Payments. Subject to Section 6.4.5(e), (i) in the event that (A) at GSK’s discretion, a license under a Third Party’s Patent Rights or an agreement is required to resolve or prevent possible allegations that the Development or Commercialization of a Licensed Product infringes such Patent Rights; or (B) GSK elects to take a sublicense under an Arrowhead Third Party Agreement pursuant to Section 6.5.3(c) and the Parties have mutually agreed upon GSK’s pro rata share of any payment

43 obligations that would be applicable to GSK as a sublicensee with respect to the Development, Manufacture or Commercialization of any Licensed Construct or Licensed Product by or on behalf of GSK (or any of its Affiliates or Sublicensees) in accordance with Section 6.5.3(b), then, in each case ((A) or (B)), GSK shall have the right to deduct from the royalties payable to Arrowhead under this Agreement for the applicable Licensed Product concerned [***] of any royalties, milestone payments, license fees or other payments actually paid by GSK to such Third Party under such license or such agreement or to Arrowhead (or such Third Party) with respect to such Arrowhead Third Party Agreement, as applicable, and (ii) with respect to [***], as applicable ((i) or (ii), collectively, “Third Party Payments”). (e) Minimum Floor. Notwithstanding any provision to the contrary set forth in the foregoing Section 6.4.5(a), Section 6.4.5(c) and Section 6.4.5(d), the operation of Section 6.4.5(a), Section 6.4.5(c) or Section 6.4.5(d), individually or in combination (but without taking into account any reductions pursuant to Section 6.4.5(b) or Section 13.8, as applicable) shall not result in the amount of royalties payable to Arrowhead hereunder with respect to Net Sales of a given Licensed Product in a given country in a given Calendar Quarter being reduced by more than [***] of the amount of royalties that would otherwise be payable pursuant to Section 6.4.2 or Section 6.4.3, as applicable, with respect to Net Sales of such Licensed Product in such country for such Calendar Quarter [***]. 6.5 Third Party Obligations. 6.5.1 Subcontractors. A Party or its designated Affiliate, in entering into any subcontract with a Third Party for the performance of any subcontracted Collaboration Activities hereunder (including in any jurisdiction in which employees or agents of such Third Party have rights to compensation, remuneration or payments for their inventions under Applicable Laws), shall use Commercially Reasonable Efforts to obligate the Third Party subcontractor in a written subcontract agreement to be solely responsible for any compensation, remuneration or payments due to any of the Third Party’s employees or agents on account of their performance of any such activities under the subcontract agreement, including any payment obligations that may arise by operation of Applicable Law in a particular country on account of either Party’s exercise of any rights hereunder with respect to any Licensed Products that were invented, in whole or in part, by any such Third Party employees or agents in the performance of such activities. If a Party fails to include such an obligation in any of its subcontract agreements with any Third Parties, such Party shall bear any expense incurred in connection with any such payment obligations that may so arise. 6.5.2 [***]

44 6.5.3 Other Third Party Obligations. (a) During the Term, prior to Arrowhead (or any of its Affiliates) entering into an agreement with a Third Party pursuant to which Arrowhead (or its Affiliate) would in-license any Know-How or Patent Rights that would, but for requirements of Section 1.56(b), be deemed to be Arrowhead Intellectual Property (such agreement, if entered into by Arrowhead or its Affiliate in accordance with Section 6.5.3(c), an “Arrowhead Third Party Agreement”), Arrowhead will provide written notice to GSK of Arrowhead’s (or its Affiliate’s) intent to enter into such proposed Arrowhead Third Party Agreement, along with reasonably detailed information regarding the proposed financial terms, as well as any other material terms applicable to sublicensees, under such proposed Arrowhead Third Party Agreement and the relevant Know-How or Patent Rights owned or otherwise controlled by such Third Party that are proposed to be included as Arrowhead Intellectual Property if GSK elects to take a sublicense with respect to such proposed Arrowhead Third Party Agreement pursuant to Section 6.5.3(c). (b) Prior to Arrowhead executing any such proposed Arrowhead Third Party Agreement, the Parties, through the Patent Working Group, shall confer to discuss whether it is in best interest of the Parties, in respect of their respective rights to Develop, Manufacture and Commercialize the Licensed Constructs and Licensed Products in the Territory in accordance with this Agreement, for Arrowhead (or its Affiliate) to enter into such proposed Arrowhead Third Party Agreement. Arrowhead (or its Affiliate) shall use Commercially Reasonable Efforts to obtain sublicensable rights or licenses under the relevant Know-How or Patent Rights pursuant to such Arrowhead Third Party Agreement on terms substantially consistent with the rights and licenses granted to GSK under the Arrowhead Intellectual Property pursuant to Section 2.1 (but, in all cases, in any manner where the financial terms of such proposed Arrowhead Third Party Agreement do not disproportionately disadvantage any Licensed Constructs or Licensed Products vis-à-vis any other compound or product under such proposed Arrowhead Third Party Agreement); provided that, prior to Arrowhead executing any such Arrowhead Third Party Agreement, the Parties shall, acting reasonably and in good faith, negotiate and mutually agree on GSK’s pro rata share of any payment obligations under such Arrowhead Third Party Agreement that would be applicable to GSK as a sublicensee to the extent directly attributable to the Development, Manufacture or Commercialization of any Licensed Construct or Licensed Product by or on behalf of GSK (or any of its Affiliates or Sublicensees) in the event that GSK elects to take a sublicense under such Arrowhead Third Party Agreement pursuant to Section 6.5.3(c).

45 (c) If Arrowhead (or its Affiliate) is successful in obtaining such sublicensable rights or licenses under the applicable Arrowhead Third Party Agreement in accordance with Section 6.5.3(b), then, GSK shall have the right, by delivery of notice to Arrowhead, to elect to take a sublicense under such relevant Know-How or Patent Rights in-licensed by Arrowhead (or its Affiliate) under such Arrowhead Third Party Agreement, in which case GSK agrees to comply, and will cause its Affiliates and Sublicensees to comply, with any applicable obligations under such Arrowhead Third Party Agreement that apply to GSK (or its Affiliates or Sublicensees) as sublicensees thereunder and of which GSK was informed by Arrowhead in writing prior to such election by GSK pursuant to this Section 6.5.3(c), including any obligation to make GSK’s pro rata share of such payments directly attributable to the Development, Manufacture, or Commercialization of any Licensed Construct or Licensed Product as mutually agreed by the Parties pursuant to Section 6.5.3(b). ARTICLE VII: GENERAL PAYMENT TERMS 7.1 Invoices. All undisputed amounts payable by GSK to Arrowhead (or its Third Party designee under a Securitization Transaction pursuant to Section 15.1.2, as applicable) under this Agreement shall be paid in accordance with the applicable provisions of this Article VII, following receipt by GSK of a valid invoice in accordance with Schedule 7.1. 7.2 Royalty Reporting and Payments. Royalty payments due shall be payable in United States dollars [***] days after the end of each Calendar Quarter during the applicable Royalty Term. Each payment of royalties due under this Agreement will be accompanied with a royalty report setting forth, on a Licensed Product-by-Licensed Product and country- by-country basis, (a) the amount of Net Sales of Licensed Product by GSK, its Affiliates and Sublicensees in US dollars; and (b) a calculation of the aggregate amount of royalties owed based on such Net Sales, including the application of the reductions or credits, if any, made in accordance with the terms of Section 6.4.5. 7.3 Remittance. All payments made by a Party (the “Payor Party”) to the other Party (the “Payee Party”) hereunder shall be made by deposit of US Dollars in the requisite amount by electronic wire transfer of immediately available funds directly to such bank account as the Payee Party may from time to time designate by reasonable notice to the Payor Party. With respect to amounts payable hereunder not denominated in US Dollars, the Payor Party shall convert applicable amounts in foreign currency into US Dollars using its standard conversion method consistent with its applicable Accounting Standard in a manner consistent with the Payor Party’s customary and usual conversion procedures used in preparing its audited financial reports applied on a consistent basis; provided that such procedures use a widely accepted source of published exchange rates. Based on the resulting sales in US Dollars, the then-applicable royalties shall be calculated. The Parties may vary the method of payment set forth herein at any time upon mutual agreement, and any change shall be consistent with the Applicable Law at the place of payment or remittance.

46 7.4 Taxes. 7.4.1 Each Party shall be solely responsible for the payment of all Taxes imposed on its share of income arising directly or indirectly from the collaborative efforts of the Parties under this Agreement. 7.4.2 If any amounts to be paid by GSK under this Agreement (including any milestone or royalty payments) are subject to any withholding or similar Tax, GSK shall provide Arrowhead advance notice (to the extent commercially reasonable such advance notice shall be at least fifteen (15) Business Days in advance of such withholding) and shall (a) pay such withholding or similar Tax to the proper taxing Governmental Authority and furnish Arrowhead with proof of such payment; and (b) remit the remaining amount of such payments to Arrowhead after deducting any such withholding or similar Tax paid by GSK. The Parties agree to cooperate with one another and use reasonable efforts to reduce or eliminate Tax withholding or similar obligations in respect of any payments or royalties paid by GSK to Arrowhead under this Agreement. Arrowhead will provide GSK with any tax forms that may be reasonably necessary in order for GSK not to withhold Tax or to withhold Tax at a reduced rate under an applicable bilateral income tax treaty. Each Party will provide the other with reasonable assistance to enable the recovery, as permitted by Applicable Law, of withholding Taxes, VAT and Indirect Taxes or similar obligations resulting from payments made under this Agreement, such recovery to be for the benefit of the Party bearing such withholding Taxes or VAT and Indirect Taxes. Notwithstanding the foregoing, if an action (including any license or sublicense of any rights or performance of any obligations under this Agreement, a change in the applicable taxing jurisdiction or any failure to comply with Applicable Law or filing or record retention requirements) by either Party or any of its Affiliates causes any new or increased withholding Tax liability that would not have been imposed in the absence of such action, such Party causing the new or increased withholding Tax liability shall indemnify and hold harmless the other Party and its Affiliates from the amount of such additional or increased withholding Tax (except to the extent that such other Party or its Affiliates is entitled to a refund of such withheld Taxes or entitled to credit such withheld Taxes against Taxes which such other Party or its Affiliates would otherwise be required to pay). Within five (5) Business Days following the Amended Effective Date of this Agreement, Arrowhead will deliver to GSK an accurate and complete Internal Revenue Service Form W-9 and such form shall be updated and renewed as required by Applicable Law. 7.4.3 All amounts payable under or in connection with this Agreement are exclusive of VAT and Indirect Taxes. Any VAT and Indirect Taxes payable on any amount paid hereunder shall be paid by GSK at the same time as the payment or provision of such consideration to which it relates, subject to the production of a VAT and Indirect Taxes valid invoice in accordance with Section 7.1. Each Party agrees that it shall provide to the other Party any information and copies of any documents within its control to the extent reasonably requested by the other Party for the purposes of (a) determining the amount of VAT and Indirect Taxes chargeable

47 under this Agreement, (b) establishing the “place of supply for VAT” purposes, or (c) complying with its VAT and Indirect Taxes reporting or accounting obligations. 7.4.4 Arrowhead warrants that Arrowhead is resident for Tax purposes in the United States and that Arrowhead is entitled to relief from United Kingdom income Tax under the terms of the double tax agreement between the UK and the United States. Arrowhead shall notify GSK promptly in writing in the event that Arrowhead ceases to be entitled to such relief. 7.5 Records and Audit Rights. 7.5.1 Maintenance of Records. Arrowhead shall keep (and shall cause its Affiliates and applicable Third Party (sub)contractors to keep) complete, true and accurate books and records in accordance with Accounting Standards in sufficient detail for GSK to determine the payments due and costs incurred under this Agreement. GSK shall keep (and shall cause its Affiliates and Sublicensees to keep) complete, true and accurate books and records in accordance with Accounting Standards in sufficient detail to permit Arrowhead to confirm the accuracy of GSK’s financial records related to the royalty calculations and calculations of Net Sales hereunder. Each Party will keep such books and records in accordance with Applicable Law and for at least three (3) years following the date of the payment to which they pertain. In the event that GSK is required to reimburse Out-of-Pocket Costs or other costs to Arrowhead, Arrowhead shall provide GSK with proof of such costs upon GSK’s request. 7.5.2 Audit Right. Upon the written request of a Party (as applicable, the “Auditing Party”), not more than once every two (2) Calendar Years, the other Party (the “Audited Party”) shall permit an independent certified public accounting firm of internationally recognized standing selected by the Auditing Party and reasonably acceptable to the Audited Party to have confidential access during normal business hours to such of the records of the Audited Party and its applicable Affiliates or Third Party sublicensees or subcontractors as may be reasonably necessary for the sole purpose of verifying the accuracy of any payments made under this Agreement for any period ending not more than three (3) years prior to the date of such request. For clarity, in the event that GSK is the Audited Party, access shall be limited to records reasonably necessary for the sole purpose of verifying the royalty and Net Sales calculations hereunder. An audit of the records relating to a particular Calendar Year may be conducted not more than once. The accounting firm shall provide the Audited Party a copy of its report prior to sharing it with the Auditing Party in order to allow the Audited Party to provide the accounting firm with justifying remarks for inclusion, at the accounting firm’s sole discretion, in the report prior to sharing the report with the Auditing Party. The accounting firm shall provide each Party, at the same time, a correct and complete copy of the final report summarizing the final results of such audit, which shall be treated as the Audited Party’s Confidential Information. The Auditing Party shall obligate its accounting firm to keep the Audited Party’s information confidential and shall, at the request

48 of the Audited Party, cause the Auditing Party’s accounting firm to execute a reasonable confidentiality agreement prior to commencing any such audit. 7.5.3 Audit Fees. The fees charged by an accounting firm engaged by a Party in accordance with Section 7.5.2 shall be paid by the Auditing Party; provided, however, that if the audit uncovers (a) with respect to GSK as the Audited Party, any underpayment of any undisputed payment obligation of GSK or (b) with respect to Arrowhead as the Audited Party, overpayment by GSK, in each case ((a) or (b)), exceeding [***]percent ([***]%) of the total amount due in accordance with this Agreement for the audited period, then, the fees of such accounting firm shall be paid by such Audited Party. Any overpayments discovered by such audit or otherwise shall be fully creditable against amounts payable by GSK in any subsequent payment periods, plus interest calculated in accordance with Section 7.6. Any underpayments discovered by such audit or otherwise will be paid promptly by GSK within [***] days following GSK’s receipt of the report of the accounting firm and a valid invoice from Arrowhead in accordance with Section 7.1, which invoice shall be delivered following the delivery of such accounting firm’s written report to the Audited Party such accounting firm’s written report, or as otherwise agreed upon by the Parties, plus interest calculated in accordance with Section 7.6. 7.6 Interest on Late Payments. The Payor Party will pay the Payee Party interest on any undisputed payments that are not paid on or before the date such payments are due under this Agreement at a rate equal to [***]per annum or, if lower, the maximum applicable legal rate under Applicable Law, calculated on the total number of days payment is delinquent; provided that, for clarity, to the extent that any such delay in payment of any amount payable by GSK under this Agreement is due to any failure by Arrowhead to deliver a valid invoice to GSK in accordance with Section 7.1 or otherwise to respond to communications from GSK regarding the interpretation or dispute of the terms of such payment, no interest will be payable by GSK with respect to such delay in remitting such payment. ARTICLE VIII: INTELLECTUAL PROPERTY MATTERS 8.1 Reporting of Arrowhead Inventions. Arrowhead shall promptly report to GSK’s Prosecution Contact, any material invention made by any of its employees or agents or its Affiliates’ or Third-Party subcontractors’ employees or agents that Covers the composition of matter of a Licensed Product or any component thereof (“Arrowhead Invention”). 8.2 Ownership of Agreement IP. 8.2.1 General. Except as otherwise provided in this Section 8.2, ownership of any Agreement IP shall follow inventorship as determined pursuant to principles of United States patent law. 8.2.2 Existing Inventions; Existing Clinical Data. The Parties acknowledge and agree that Section 10.2 of the Original Agreement shall govern the ownership of any

49 invention arising from any activities under the Original Agreement prior to the Amended Effective Date (each such invention, an “Existing Invention”) together with any Patent Rights therein or thereon; provided, however, that, notwithstanding anything to the contrary set forth in this Agreement or the Original Agreement, as between the Parties, GSK (as the permitted assignee of Janssen under this Agreement) shall solely own all rights, title and interest in and to any and all [***] but excluding any Existing Inventions that were assigned to Arrowhead in accordance with Section 10.2.2 of the Original Agreement (such Agreement Know- How, the “Existing Clinical Data”). 8.2.3 Arrowhead Agreement IP. Subject to any rights or licenses expressly granted by Arrowhead to GSK under this Agreement (including the licenses granted to GSK under Section 2.1.1 and Section 2.1.2), as between the Parties, Arrowhead shall solely own all rights, title and interest in and to any and all: (a) New Arising Know-How, regardless of inventorship, that is solely related to the Registered Starting Material ([***]) (the “New [***] Know-How”); (b) Agreement Patent Rights, regardless of inventorship, that solely claim any New [***] Know-How (the “New [***] Patent Rights”); (c) New Arising Know-How that is discovered, developed, generated, invented, derived, created, conceived or reduced to practice by or on behalf of one (1) or more Personnel of Arrowhead (or any of its Affiliates, sublicensees or subcontractors) but excluding (i) any New [***] Know- How and (ii) any Joint Agreement Know-How (such New Arising Know- How set forth in this Section 8.2.3(c), together with any New [***] Know- How and any Existing Inventions solely owned by Arrowhead (whether or not they solely relate to the Registered Starting Material ([***])), collectively, the “Arrowhead Agreement Know-How”); and (d) Agreement Patent Rights that claim (i) any Arrowhead Agreement Know- How set forth in Section 8.2.3(c) or (ii) any Existing Inventions solely owned by Arrowhead (whether or not they solely relate to the Registered Starting Material ([***])) but, in each case ((i) or (ii)), excluding (A) any Joint Agreement Patent Rights and (B) any other Agreement Patent Rights that claim or otherwise disclose (1) any Existing Clinical Data or (2) any Existing Invention solely owned by GSK (as the permitted assignee of Janssen) (such Agreement Patent Rights set forth in this Section 8.2.3(d), together with any New [***] Patent Rights, collectively, the “Arrowhead Agreement Patent Rights”). 8.2.4 GSK Agreement IP. As between the Parties, GSK shall solely own all rights, title and interest in and to any and all: (a) New Arising Know-How that is discovered, developed, generated, invented, derived, created, conceived or reduced to practice by or on behalf

50 of one (1) or more Personnel of GSK (or any of its Affiliates, licensees, sublicensees or subcontractors) excluding (i) any New [***] Know-How and (ii) any Joint Agreement Know-How (such New Arising Know-How set forth in this Section 8.2.4(a), together with any Existing Clinical Data and any other Existing Inventions solely owned by GSK (as the permitted assignee of Janssen under this Agreement), collectively, the “GSK Agreement Know-How”); and (b) Agreement Patent Rights that claim (i) any GSK Agreement Know-How set forth in Section 8.2.4(a), (ii) any Existing Clinical Data or (iii) any Existing Inventions solely owned by GSK (as the permitted assignee of Janssen under this Agreement) but, in each case ((i), (ii) or (iii)), excluding (A) any New [***] Patent Rights, (B) any Joint Agreement Patent Rights and (C) any other Agreement Patent Rights that claim or otherwise disclose any Existing Invention solely owned by Arrowhead. 8.2.5 Joint Agreement IP. Subject to any rights or licenses expressly granted by one Party to the other Party under this Agreement (including the licenses granted to GSK under Section 2.1.1 and Section 2.1.2), the Parties shall jointly own, on an equal and undivided basis, including all rights, title and interest in and to any and all: (a) New Arising Know-How that is discovered, developed, generated, invented, derived, created, conceived or reduced to practice jointly by or on behalf of (i) one (1) or more Personnel of GSK (or any of its Affiliates, licensees, sublicensees or subcontractors), on the one hand, and (ii) one (1) or more Personnel of Arrowhead (or any of its Affiliates, licensees, sublicensees or subcontractors), on the other hand, but excluding any New [***] Know-How (such New Arising Know-How set forth in this Section 8.2.5(a), together with any Existing Inventions jointly owned by Arrowhead and GSK (as the permitted assignee of Janssen under this Agreement), collectively, the “Joint Agreement Know-How”); and (b) (i) Existing Joint Agreement Patent Rights and (ii) Agreement Patent Rights that claim any Joint Agreement Know-How (the “Joint Agreement Patent Rights”). 8.2.6 Further Assurances. (a) During the Term, each Party shall promptly disclose to the other Party all invention disclosures or other similar documents relating to: (i) with respect to GSK, any New [***] IP or Joint Agreement IP that is discovered, developed, generated, invented, derived, created, conceived or reduced to practice by or on behalf of one (1) or more Personnel of GSK (or any of its Affiliates, licensees, sublicensees or subcontractors), and (ii) with respect to Arrowhead, any Arrowhead Agreement IP or Joint Agreement IP that is discovered, developed, generated, invented, derived, created, conceived or

51 reduced to practice by or on behalf of one (1) or more Personnel of Arrowhead (or any of its Affiliates, licensees, sublicensees or subcontractors), as applicable. Each Party shall also respond promptly to reasonable requests from the other Party for additional information relating to such disclosures, documents or applications; provided that (A) GSK shall only be obligated to respond to the extent such additional requested information that is included within the New [***] Know-How or the Joint Agreement Know-How, as applicable; and (B) Arrowhead shall only be obligated to respond to the extent such additional requested information is included within the Arrowhead Agreement Know-How or the Joint Agreement Know-How, as applicable. (b) In furtherance of this Section 8.2, subject to any rights or licenses expressly granted to a Party under this Agreement (including the licenses granted to GSK under Section 2.1.1 and Section 2.1.2, and the Parties’ respective rights under the remainder of this Article VIII): (i) GSK shall, and hereby does, assign to Arrowhead (A) all of its (and its Affiliates’) rights, title and interest in and to any New [***] IP that is discovered, developed, generated, invented, derived, created, conceived or reduced to practice by or on behalf of one (1) or more Personnel of GSK (or any of its Affiliates, licensees, sublicensees or subcontractors), and (B) a joint equal and undivided interest in, to and under any Joint Agreement IP and, in each case ((A) or (B)), Arrowhead hereby accepts such assignment; (ii) Arrowhead shall, and hereby does, assign to GSK (A) all of its (and its Affiliates’) rights, title and interest in, to and under any Existing Clinical Data that was discovered, developed, generated, invented, derived, created, conceived or reduced to practice by or on behalf of one (1) or more Personnel of Arrowhead (or any of its Affiliates, licensees, sublicensees or subcontractors), and (B) a joint equal and undivided interest in, to and under any Joint Agreement IP and, in each case ((A) or (B)), GSK hereby accepts such assignment; and (iii) each Party shall have the right to exploit, grant licenses under, assign and otherwise dispose of any Joint Agreement Know-How or Joint Agreement Patent Rights, without an accounting or obligation to, or consent required from, the other Party, and each Party hereby waives any right it may have under the laws of any jurisdiction to require any such consent or accounting (or, if such waiver is not valid in any jurisdiction, each Party hereby consents to the grant of any license to any Third Party without accounting to the other Party). (c) During the Term, each Party will, and will ensure that its Affiliates, licensees, sublicensees and subcontractors obtain written agreements from any and all Persons involved in or performing any Development activities

52 by or on behalf of such Party under this Agreement that (i) presently assign such Persons’ rights, title, and interests in and to any Agreement IP to the Party that is the counterparty to such agreements, in each case, prior to any such Persons performing such Development activities; (ii) require such Persons to promptly report any Agreement IP to the Party that is the counterparty to such agreements; (iii) require such Persons to cooperate in the Prosecution and enforcement of any Patent Rights by the Party that is the counterparty to such agreements; and (iv) require such Persons to perform all acts and signing, executing, acknowledging, and delivering any and all documents required for effecting the obligations and purposes of this Agreement. It is understood and agreed that such invention assignment agreement need not reference this Agreement. 8.3 Prosecution of Patent Rights. 8.3.1 Communications. Each Party shall use reasonable efforts to handle all communications between the Parties under this Section 8.3 through their Prosecution Contacts and keep such communications in strict confidence to protect their attorney-client privileged status. 8.3.2 Reporting of Filings. Subject to Section 8.3.3, a Party planning on filing any priority-establishing or original (in each case, with respect to any claims or new matter described in the patent specification) patent application within the Arrowhead Patent Rights or Joint Agreement Patent Rights hereunder shall use reasonable efforts to provide to the other Party, with reasonable advance time such as at least thirty (30) days prior to proposed Prosecution filing in a Patent Office (such as a draft application or response to an official action), and provide the other Party an opportunity to comment thereon through its Prosecution Contact. Subject to 8.3.4(g), each Party shall provide to the other, promptly after filing, a copy of each priority-establishing or original (whether provisional or nonprovisional) patent application within the Arrowhead Patent Rights, GSK Agreement LC/LP Patent Rights or Joint Agreement Patent Rights as filed in the Patent Office and each other substantive Prosecution filing (including any other patent application filed within the Arrowhead Patent Rights, GSK Agreement LC/LP Patent Rights or the Joint Agreement Patent Rights). 8.3.3 Prosecution Responsibility and Coordination of Arrowhead Patent Rights and Joint Agreement Patent Rights. (a) With respect to any Arrowhead Patent Rights that (i) claim or otherwise disclose any Licensed Construct or Licensed Product, including any Arrowhead Patent Rights that are necessary for, claim or otherwise disclose the Development, Manufacture or Commercialization of any Licensed Construct or Licensed Product, as applicable, but excluding any Arrowhead Patent Rights that specifically claim the Arrowhead Platform Technology or (ii) claim or otherwise disclose the Arrowhead Platform Technology as applied specifically to any Licensed Construct or Licensed Product,

53 including the Development, Manufacture or Commercialization thereof ((ii) or (ii), the “Specific Arrowhead Patent Rights”), as between the Parties, during the Term, GSK shall be primarily responsible for Prosecuting the Specific Arrowhead Patent Rights; provided that, during the Term, GSK shall keep Arrowhead reasonably informed of all substantive matters relating to the Prosecution of any such Specific Arrowhead Patent Rights that is Prosecuted by GSK pursuant to this Section 8.3.3(a), including as set forth in Section 8.3.2 and will consider in good faith Arrowhead’s comments, requests and suggestions with respect to strategies for the Prosecution of such Specific Arrowhead Patent Rights, including with respect to the filing of any continuation, divisional, or other continuing applications. The Specific Arrowhead Patent Rights as of the Amended Effective Date are identified in Schedule 1.29 - Part I, Part III and Part V hereto. During the Term, Arrowhead shall provide GSK prompt written notice of any changes to the Specific Arrowhead Patent Rights. (b) With respect to any Arrowhead Patent Rights other than the Specific Arrowhead Patent Rights (the “General Arrowhead Patent Rights”), as between the Parties, during the Term, Arrowhead shall be primarily responsible for Prosecuting General Arrowhead Patent Rights, which may include the use of outside patent counsel mutually acceptable to the Parties and engaged by Arrowhead, to Prosecute (or, if a Third Party has the right to control Prosecution of any General Arrowhead Patent Right under any Pre-Existing Third Party Agreements, to be represented by such Third Party in the Prosecution of) the General Arrowhead Patent Rights; provided that, during the Term, Arrowhead shall, and shall cause the applicable Third Party, if any, and subject to any restrictions or obligations in any Pre- Existing Third Party Agreements, to, keep GSK reasonably informed of all substantive matters relating to the Prosecution of any such General Arrowhead Patent Rights that is Prosecuted by Arrowhead pursuant to this Section 8.3.3(b), including as set forth in Section 8.3.2 and will consider in good faith GSK’s comments, requests and suggestions with respect to strategies for the Prosecution of such General Arrowhead Patent Rights, including with respect to the filing of any continuation, divisional, or other continuing applications. Notwithstanding the foregoing, Arrowhead shall, upon reasonable request by, and in consultation with, GSK, use Commercially Reasonable Efforts to file patent applications directed to Licensed Products with the objective of optimizing overall patent protection for Licensed Products. For clarity, the General Arrowhead Patent Rights as of the Amended Effective Date are identified in Schedule 1.29 - Part II, Part IV and Part V hereto. During the Term, Arrowhead shall provide GSK prompt written notice of any changes to the General Arrowhead Patent Rights. (c) With respect to any Joint Agreement Patent Rights, both Parties shall share primary responsibility, through outside patent counsel mutually selected and engaged by the Parties for Prosecuting such Joint Agreement Patent

54 Rights. The Existing Joint Agreement Patent Rights as of the Amended Effective Date are identified in Schedule 1.74 hereto. (d) Each Party shall provide all reasonable assistance requested by the other Party for Prosecuting any Arrowhead Patent Rights or Joint Agreement Patent Rights consistent with the terms hereof, including with respect to the timely completion of Prosecution papers to be filed in any Patent Office (including draft responses to office actions), compliance with Applicable Laws, and recording of assignments to reflect ownership consistent with the terms hereof. A Party Prosecuting any Arrowhead Patent Rights or Joint Agreement Patent Rights hereunder shall use reasonable efforts to provide the other Party with copies of all material Prosecution papers as filed in or received from any Patent Offices. The Party Prosecuting any Arrowhead Patent Rights or Joint Agreement Patent Rights hereunder shall, on an annual basis during the Term, provide the other Party with a report identifying the status of any such applicable Patent Rights for which it is primarily responsible for Prosecution; provided, however, that with respect to any Joint Agreement Patent Rights, the Parties shall cooperate to jointly prepare such status report. (e) The Parties shall discuss in good faith any Arrowhead Patent Rights or any Joint Agreement Patent Rights that will be included in the “Orange Book” maintained by the FDA or similar or equivalent patent listing or linking source, if any, in other countries in the Territory for Licensed Products, and, after considering Arrowhead’s comments in good faith, GSK will have the sole right to determine which Arrowhead Patent Rights or Joint Agreement Patent Rights will be included. Arrowhead will provide such assistance as may be reasonably requested by GSK in connection with such listing. (f) Each Party responsible to Prosecute any Arrowhead Patent Rights shall be solely responsible for all Patent Costs incurred in Prosecuting such Patent Rights. Each Party shall bear [***] of the Patent Costs incurred in Prosecuting any Joint Agreement Patent Rights. Notwithstanding the foregoing, if either Party intends to permit any particular Arrowhead Patent Right (other than any Acquired Specific Arrowhead Patent Right listed in Schedule 1.29 – Part III) or any Joint Agreement Patent Rights that is pending in any Patent Office to lapse or become abandoned (including by failure to validate an allowed multi-jurisdictional patent application, such as may be pending in the European Patent Office, in any possible country), such Party shall notify the other Party of such intention at least [***] days in advance (or, if not feasible to provide [***] days’ notice, then, within such other practicable time before the date upon which such Patent Right will lapse or become abandoned) and, solely in the case of any such Arrowhead Patent Right, to the extent not prohibited in any Pre-Existing Third Party Agreements, such other Party shall thereupon have the right, but not the obligation, to assume responsibility for the further Prosecution of such applicable Arrowhead Patent Right or Joint Agreement Patent Right

55 (and any continuing application based thereon) and all Patent Costs associated therewith, and in such event the transferring Party shall reasonably cooperate to promptly effect transfer of Prosecution of such Patent Right to the other Party. 8.3.4 Prosecution Responsibility and Coordination of GSK Agreement LC/LP Patent Rights. (a) As between the Parties, GSK shall have the sole right (but not the obligation) to pursue and direct, at its own cost and discretion, the preparation and filing of any patent applications within the GSK Agreement LC/LP Patent Rights; provided that, subject to Section 8.3.4(g), during the Term, promptly after filing, GSK shall provide Arrowhead with copies of any such priority-establishing or original (whether provisional or nonprovisional) patent application in accordance with Section 8.3.2. (b) As between the Parties, during the Term, GSK shall have the first right (but not the obligation), to pursue and direct, at its own cost and discretion, any other Prosecution of any GSK Agreement LC/LP Patent Rights (other than the preparation and filing of any patent applications within the GSK Agreement LC/LP Patent Rights for which GSK has the sole right to control pursuant to Section 8.3.4(a)); provided that, subject to Section 8.3.4(g), during the Term, GSK shall keep Arrowhead reasonably informed of all substantive matters relating to such Prosecution of any such GSK Agreement LC/LP Patent Rights that is Prosecuted by GSK pursuant to this Section 8.3.4(b) and will consider in good faith Arrowhead’s comments, requests and suggestions with respect to strategies for such Prosecution of such GSK Agreement LC/LP Patent Rights, including with respect to the filing of any continuation, divisional or other continuing applications. (c) Subject to Section 8.3.4(g), notwithstanding Section 8.3.4(b), if, during the Term, GSK intends to permit any particular GSK Agreement LC/LP Patent Right that is pending in any Patent Office to lapse or become abandoned (including by failure to validate an allowed multi-jurisdictional patent application, such as may be pending in the European Patent Office, in any possible country), GSK shall notify Arrowhead of such intention at least [***] days in advance (or, if not feasible to provide [***] days’ notice, then, within such other practicable time before the date upon which such Patent Right will lapse or become abandoned). Arrowhead shall thereupon have the right, but not the obligation, to assume responsibility for the further Prosecution of such applicable GSK Agreement LC/LP Patent Right (and any continuing application based thereon) and all Patent Costs associated therewith and, in such event, GSK shall reasonably cooperate to promptly effect transfer of Prosecution of such GSK Agreement LC/LP Patent Right to Arrowhead. Arrowhead shall keep GSK reasonably informed of all substantive matters relating to such further Prosecution of such GSK Agreement LC/LP Patent Right that is Prosecuted by Arrowhead pursuant

56 to this Section 8.3.4(c) and will consider in good faith GSK’s comments, requests and suggestions with respect to strategies for such Prosecution of such GSK Agreement LC/LP Patent Right, including with respect to the filing of any continuation, divisional or other continuing applications. For the avoidance of doubt, neither Arrowhead (nor any of its Affiliates) shall have any right to pursue or direct the preparation or filing of any patent applications within the GSK Agreement LC/LP Patent Rights, except with respect to any GSK Agreement LC/LP Patent Right for which (i) a national- stage non-provisional patent application or PCT patent application has been filed and (ii) Arrowhead has exercised its right to assume responsibility for the further Prosecution in accordance with this Section 8.3.4(c). (d) Each Party shall provide all reasonable assistance requested by the other Party for Prosecuting any GSK Agreement LC/LP Patent Rights consistent with the terms of this Section 8.3.4, including with respect to the timely completion of Prosecution papers to be filed in any Patent Office (including draft responses to office actions), and compliance with Applicable Laws. A Party Prosecuting any GSK Agreement LC/LP Patent Rights pursuant to Section 8.3.4(b) or Section 8.3.4(c), as applicable, shall use reasonable efforts to provide the other Party with copies of all material Prosecution papers as filed in or received from any Patent Offices. (e) As between the Parties, GSK shall have the sole right to determine which GSK Agreement LC/LP Patent Rights will be included in the “Orange Book” maintained by the FDA or similar or equivalent patent listing or linking source, if any, in other countries in the Territory for Licensed Products. (f) The Party responsible for Prosecuting a given GSK Agreement LC/LP Patent Right pursuant to this Section 8.3.4 shall be solely responsible for all Patent Costs incurred in Prosecuting such Patent Right. (g) Notwithstanding anything to the contrary set forth herein, the Parties acknowledge and agree that certain GSK Agreement LC/LP Patent Rights may be Controlled by GSK (or its Affiliates) during the Term pursuant to a non-exclusive license granted by Janssen to GSK under the APA and, as neither GSK nor any of its Affiliates has any rights with respect to the Prosecution, enforcement or defense of any such Patent Rights in-licensed from Janssen pursuant to the APA, the terms and conditions of this Section 8.3 and Section 10.9 shall not apply with respect to any such Patent Rights. 8.4 Patent Enforcement. 8.4.1 Notice. (a) Each Party shall provide prompt notice to the other Party of any apparent, threatened, or actual infringement by a Third Party of any Arrowhead Patent

57 Rights or Joint Agreement Patent Rights, or misappropriation of any Arrowhead Know-How or Joint Agreement Know-How, of which the Party becomes aware. The notifying Party shall promptly furnish the other Party with all known details or evidence of such infringement or misappropriation. (b) Each Party shall provide prompt notice to the other Party of any Third Party communications pertaining to any Arrowhead Patent Rights or Joint Agreement Patent Rights that the Party receives pursuant to the Drug Price Competition and Patent Term Restoration Act of 1984, including notices pursuant to §§ 101 and 103 of such act from Persons who have filed an abbreviated NDA (ANDA) or a paper NDA, or pursuant to similar such laws in the Territory. 8.4.2 Enforcement Actions. (a) As between the Parties, during the Term, GSK shall have the initial right, at its expense and in its own name (or in the name of Arrowhead as may be required under Applicable Law), to bring any infringement suit or other enforcement Action involving (i) any alleged infringement by a Third Party of (A) any Specific Arrowhead Patent Rights, or (B) any Joint Agreement Patent Rights, or (ii) any alleged misappropriation by a Third Party of any Arrowhead Know-How or Joint Agreement Know-How providing any Regulatory Exclusivity Rights for any such Licensed Product in the Territory, (each ((i) or (ii)), a “Product Infringement”), in each case, by counsel of its own choice. Arrowhead will cooperate with GSK as GSK may reasonably request in connection with any such Action initiated under this Section 8.4.2(a), including by becoming a party to such action at GSK’s cost; provided that GSK shall reimburse Arrowhead for its Out-of-Pocket Costs reasonably incurred in connection with rendering such assistance. If GSK declines to initiate such an enforcement Action against any unabated Product Infringement within [***]Business Days of receiving a notice with respect to such Product Infringement, including pursuant to Section 8.4.1, GSK shall notify Arrowhead, in which case, subject to discussion with GSK and consideration in good faith of any rationale provided by GSK as to why GSK elected not to take such action and GSK’s written consent (not to be unreasonably withheld), Arrowhead shall thereafter have the right (but not the obligation) at Arrowhead’s expense and in its own name, to initiate such Action by counsel of its choice, and GSK shall cooperate with Arrowhead as Arrowhead may reasonably request, including by becoming a party to such action at Arrowhead’s cost, and Arrowhead shall reimburse GSK for its Out-of-Pocket Costs reasonably incurred in connection with rendering such assistance. (b) As between the Parties, during the Term, Arrowhead shall have the initial right, at its expense and in its own name, to bring any infringement suit or other enforcement Action on account of any alleged infringement by a Third

58 Party of any General Arrowhead Patent Rights by counsel of its own choice. GSK will cooperate with Arrowhead as Arrowhead may reasonably request in connection with any such Action initiated under this Section 8.4.2(b), including by becoming a party to such action at Arrowhead’s cost; provided that Arrowhead shall reimburse GSK for its Out-of-Pocket Costs reasonably incurred in connection with rendering such assistance. If Arrowhead declines to initiate such an enforcement Action against any unabated infringement of such Arrowhead Patent Right within [***] Business Days of receiving a notice with respect to such alleged Third Party infringement (or such shorter period if [***] Business Days’ notice is not feasible), including pursuant to Section 8.4.1, Arrowhead shall notify GSK, in which case, subject to discussion with Arrowhead and consideration in good faith of any rationale provided by Arrowhead as to why Arrowhead elected not to take such action and Arrowhead’s written consent (not to be unreasonably withheld), GSK shall thereafter have the right (but not the obligation) at GSK’s expense and in its own name, to initiate such Action by counsel of its choice, and Arrowhead shall cooperate with GSK as GSK may reasonably request, including by becoming a party to such action at GSK’s cost, and GSK shall reimburse Arrowhead for its Out-of-Pocket Costs reasonably incurred in connection with rendering such assistance. (c) A settlement or consent judgment or other voluntary final disposition of an Action brought by a Party under this Section 8.4.2 may be entered into without the consent of the other Party; provided that such settlement, consent judgment, or other disposition does not admit the invalidity or unenforceability of any Patent Rights owned or Controlled by the other Party; provided, further, that any rights granted to a Third Party to continue any activity upon which such Action was based in such settlement, consent judgment, or other disposition shall be limited to the Third Party’s product or activity that was the subject of the Action. (d) Damages recovered and any other amounts awarded in any Actions brought by a Party under this Section 8.4.2 shall be (i) first, allocated [***] and (ii) thereafter, [***]. 8.4.3 Cooperation on Enforcement Actions. Each Party acknowledges that the outcome of any infringement suit or other enforcement Action on account of any Third-Party infringement of any Arrowhead Patent Right licensed to GSK under Section 2.1 or any Joint Agreement Patent Rights may detrimentally impact the scope, validity, or enforceability of such Patent Right with respect to potential Third Party infringements. Accordingly, the Parties shall reasonably cooperate with each other with respect to any infringement suit or other enforcement Action on account of any Third-Party infringement of any Arrowhead Patent Right or any Joint Agreement Patent Rights. For clarity, subject to Section 8.4.2, neither Arrowhead nor GSK will be required to enforce any Arrowhead Patent Right or any Joint Agreement Patent Rights against any Third Party infringement.

59 8.5 Maintenance of Freedom to Operate. The Parties shall use Commercially Reasonable Efforts to avoid infringing any Third Party’s Patent Rights in conducting any Development activities under the Development Plan. Each Party shall promptly notify the Prosecution Contacts, in the event such Party becomes aware of any Third Party’s Patent Rights that may pertain to any Development activities of the Parties. 8.6 Patent Term Extensions. During the Term, upon GSK’s written request (which shall be by a notice identifying the date of the applicable Regulatory Approval of a Licensed Product and the deadline for filing a Patent Term Extension), the Prosecuting Party shall use reasonable efforts, in each country or jurisdiction where Regulatory Approval for any such Licensed Product has been obtained, and if the Applicable Law of such country or jurisdiction permits application for a Patent Term Extension, to apply, at the reasonable direction of GSK’s designated patent counsel, for a Patent Term Extension for a patent within the Arrowhead Patent Rights including a Valid Claim Covering such Licensed Product, which patent (if any) shall be selected at GSK’s reasonable judgment after considering the opinion of GSK’s patent counsel regarding its eligibility for a Patent Term Extension. GSK shall have the right to: (a) identify in any list of Patent Rights in a Drug Application the applicable Arrowhead Patent Rights as GSK reasonably believes is appropriate; (b) commence suit for any Product Infringement of any such Arrowhead Patent Rights under Applicable Law as permitted under Section 8.4.2; and (c) exercise any rights that may be exercisable by a patent owner, including applying for a Patent Term Extension, of any Arrowhead Patent Rights pertaining to an approved Licensed Product Commercialized by GSK hereunder. Arrowhead agrees to cooperate with GSK and its Affiliate and Sublicensees, upon GSK’s reasonable request in the exercise of the authorizations granted under this Section 8.6, and Arrowhead shall execute such documents and take such additional action as GSK may reasonably request in connection therewith, including, if requested by GSK, permitting Arrowhead to be joined as a party in any suit for Product Infringement brought by GSK hereunder on the terms and conditions set forth in Section 8.4.2, provided that GSK shall reimburse Arrowhead all reasonable Out-of- Pocket Costs incurred by Arrowhead in taking such action. 8.7 Defense of Third Party Actions. 8.7.1 If either Party or any of its Affiliates becomes aware of any Action initiated or threatened by a Third Party against either Party (or any of their respective Affiliates) that claims that any Licensed Construct or Licensed Product, or its Development, Manufacture or Commercialization, infringes such Third Party’s Patent Rights or misappropriates such Third Party’s Know-How (but excluding any such Action with respect to a Combination Product to the extent that the underlying claim of such Third Party relates to any other Active Ingredient (other than a Licensed Construct) with respect to such Combination Product, as applicable) (any such Action, a “Third Party Action”), such Party or such Affiliate, through the Patent Working Group, shall promptly notify the other Party of all details regarding such claim or action that is reasonably available to such Party or such Affiliate. 8.7.2 As between the Parties, subject to the respective indemnity obligations of the Parties set forth in Section 11.1, GSK shall have the sole and exclusive right and

60 authority (but not the obligation), in its sole discretion and at its own cost and expense, to defend against, and select counsel for, any Third Party Action with respect to any Licensed Product in the Territory and to compromise or settle such Third Party Action; provided that GSK shall not settle or otherwise compromise any Third Party Action in a way that imposes any costs or liability on, or involves any admission by, Arrowhead or any of its Affiliates, in each case, without Arrowhead’s prior written consent. Arrowhead and its Affiliates shall reasonably cooperate, at GSK’s request and expense, in connection with the defense of any such Third Party Action. For the avoidance of doubt, in the event that a judgment in a Third Party Action is entered against GSK or any of its Affiliates and an appeal is available, as between the Parties, GSK shall have the sole and exclusive right (but not the obligation), in its sole discretion and at its own cost and expense, to file such appeal, and if Applicable Law requires Arrowhead’s or any of its Affiliates’ involvement in an appeal, Arrowhead or such Affiliate shall be a nominal party of the appeal and shall provide reasonable cooperation to GSK and its Affiliates, at GSK’s request and expense. 8.8 Patent Working Group. The Parties shall establish a patent working group comprising an equal number of up to three representatives of each Party (“Patent Working Group”), including each Party’s respective Prosecution Contact, for the sole purposes of alignment of activities under this Article VIII including the Parties’ respective responsibilities for Prosecuting and enforcing Arrowhead Patent Rights and Joint Agreement Patent Rights, or any other Patent Rights matters as the Parties may mutually agree upon. The Patent Working Group may hold meetings as appropriate to discuss such patent matters, in each case, at such frequency, time, place and manner as may be mutually agreed by the Parties’ respective Prosecution Contacts. 8.9 Common Interest Agreement. All non-public information exchanged between the Parties or between a Party’s outside patent counsel and the other Party regarding the Prosecution, enforcement and defense of the Arrowhead Patent Rights, Joint Agreement Patent Rights or otherwise related to any Licensed Construct or Licensed Product, and all shared information regarding analyses or opinions of Third Party Patent Rights or Know- How, shall be deemed Confidential Information. The Parties agree and acknowledge that they have not waived, and nothing in this Agreement constitutes a waiver of, any legal privilege concerning any such Patent Rights, Know-How or Confidential Information, including privilege under the common interest doctrine and similar or related doctrines. In furtherance of the foregoing, if the Parties agree that a separate agreement memorializing this understanding would be advantageous, the Parties shall negotiate and enter into a common interest agreement reflecting this understanding or any other common interest agreement as the Parties may mutually agree, including with respect to any product liability for a Licensed Product. 8.10 Product Trademarks. Arrowhead hereby represents and warrants to GSK, as of the Original Effective Date (as though made then under the terms then in effect under the Original Agreement) and as of the Amended Effective Date, that it does not own or otherwise Control any Product Trademark Rights relating to Licensed Constructs, including any trademark applications or registrations or domain names. GSK shall have

61 (directly and through its Affiliates and Sublicensees Commercializing Licensed Products) the right to brand, at its discretion, the Licensed Products using trademarks and trade names selected at its discretion and to file for, obtain, and maintain at its discretion and cost Product Trademark Rights in its own name. No right, express or implied, is granted by this Agreement to either Party to use in any manner the name or any other trade name or trademark of the other Party in connection with the performance of this Agreement or otherwise. 8.11 GSK Patent Rights. For the avoidance of doubt, as between the Parties, except as otherwise expressly provided in Section 8.3.2 or Section 8.3.4 with respect to the Prosecution of GSK Agreement LC/LP Patent Rights, GSK shall have the sole right (but not the obligation) to pursue and direct, at its own cost and discretion, any Prosecution, enforcement or other Action with respect to any GSK Agreement Patent Rights or any other Patent Rights owned or otherwise Controlled (other than pursuant to any rights or licenses granted by Arrowhead under this Agreement) by GSK or any of its Affiliates and used in the performance of this Agreement. ARTICLE IX: CONFIDENTIALITY AND PUBLICITY 9.1 Confidential Information. 9.1.1 Except as otherwise expressly permitted by this Agreement, each Party agrees that during the Term and for ten (10) years thereafter, such Party shall, and shall ensure that its Affiliates and its and their respective officers, directors, employees, workers, contractors, advisors, consultants, agents or other representatives (“Representatives”), hold in confidence all non-public or confidential information relating to the business, operations, technical or scientific activities, compounds or products of the other Party or any of its Affiliates (the “Disclosing Party”), including any Know-How, that is disclosed or provided to such Party or any of its Affiliates (the “Receiving Party”) or any of its or their Representatives, or of which such Receiving Party or any of its Representatives becomes aware of, in connection with this Agreement (or the Existing Confidentiality Agreement, as applicable) (collectively, “Confidential Information”). The Receiving Party shall not disclose any of the Confidential Information of the Disclosing Party to any Third Party, except to its Representatives or any of its Sublicensees or permitted subcontractors, in each case, to the extent that such use or disclosure is for the purpose of performing the Receiving Party’s obligations, or exercising its rights, under this Agreement and who are bound by obligations of non-use and non- disclosure substantially similar to those set forth herein. The Receiving Party shall be responsible for any disclosure or use of the Confidential Information of the Disclosing Party in breach of its obligations hereunder by such Representatives. The Receiving Party shall protect the Confidential Information of the Disclosing Party using not less than the same care with which it treats its own confidential information, but at all times shall use at least reasonable care. Each Party shall (a) implement and maintain appropriate security measures to prevent unauthorized access, disclosure or use of the other Party’s Confidential Information; (b) promptly notify the other Party of any unauthorized access or disclosure of such other Party’s

62 Confidential Information; and (c) cooperate with such other Party in the investigation and remediation of any such unauthorized access or disclosure. 9.1.2 Notwithstanding anything to the contrary set forth herein, (a) [***] shall be deemed to be GSK’s Confidential Information and GSK shall be deemed to be the Disclosing Party and Arrowhead shall be deemed to be the Receiving Party with respect thereto; and (b) (i) the existence and the terms and conditions of this Agreement and (ii) [***], in each case ((b)(i) or (b)(ii)), shall be deemed to be the Party’s joint Confidential Information, with both Parties deemed to be the Receiving Party of such Confidential Information, in all cases, unless and to the extent any such information is disclosed in any press release, presentation or other form of public disclosure permitted under and in accordance with Section 9.3 or Section 9.4, as applicable. 9.1.3 The restrictions on a Receiving Party’s disclosure and use of the Disclosing Party’s Confidential Information set forth above in this Section 9.1 shall not apply to any particular Confidential Information to the extent that such Confidential Information: (a) was known by the Receiving Party prior to disclosure by the Disclosing Party hereunder (as evidenced by the Receiving Party’s written records or other competent evidence); (b) is or becomes part of the public domain through no fault of the Receiving Party in violation of this Agreement; (c) is disclosed without restriction to the Receiving Party by a Third Party having a legal right to make such disclosure without violating any confidentiality or non-use obligation that such Third Party has to the Disclosing Party; or (d) is independently developed by Personnel of the Receiving Party without reliance on or access to the Disclosing Party’s Confidential Information (as evidenced by the Receiving Party’s written records or other competent evidence). 9.2 Permitted Use and Disclosures. 9.2.1 Notwithstanding Section 9.1, subject to the remainder of this Section 9.2, a Receiving Party may, in connection with performing its obligations or exercising its rights and performing its obligations under this Agreement, disclose the Confidential Information of the Disclosing Party, including for purposes of: (a) Prosecuting Patent Rights in accordance with Article VIII; (b) prosecuting or defending litigation;

63 (c) conducting pre-clinical studies or Clinical Trials of any Licensed Product in accordance with this Agreement; (d) seeking or maintaining Regulatory Approval of any Licensed Product in accordance with this Agreement; or (e) to such Receiving Party’s actual or potential partners, acquirers, financing sources, licensors, (sub)licensees and their respective personnel, each of whom prior to disclosure must be bound by similar obligations of confidentiality and non-use at least equivalent in scope to those set forth in Section 9.1. 9.2.2 In making any disclosures set forth in Section 9.2.1(a) through Section 9.2.1(d) above, the Receiving Party shall, where reasonably practicable, give such advance notice to the Disclosing Party of such disclosure requirement as is reasonable under the circumstances and will use its reasonable efforts to cooperate with the Disclosing Party in order to secure confidential treatment of such Confidential Information required to be disclosed. In addition, in connection with any permitted filing by either Party of this Agreement with any Governmental Authority, the Receiving Party shall (a) endeavor to obtain confidential treatment of economic, trade secret information and such other information as may be requested by the Disclosing Party; (b) provide the Disclosing Party with the proposed confidential treatment request within a reasonable time for the Disclosing Party to provide comments, and the Receiving Party shall consider and incorporate such comments in good faith in connection with its submission of its confidential treatment request; and (c) submit the proposed disclosure in writing to the Disclosing Party as far in advance as reasonably practicable (and in no event less than [***] Business Days prior to the anticipated date of disclosure) so as to provide a reasonable opportunity to comment thereon and the Receiving Party shall incorporate such comments in good faith. 9.2.3 Notwithstanding Section 9.1, (a) the Receiving Party may disclose the Confidential Information of the Disclosing Party to the extent required by Applicable Law or court order; provided, however, that the Receiving Party shall first provide the Disclosing Party prior notice of such disclosure and give the Disclosing Party a reasonable opportunity to quash such order or to obtain a protective order or confidential treatment requiring that the Confidential Information and documents that are the subject of such order or required to be disclosed be held in confidence by such court or Governmental Authority or, if disclosed, be used only for the purposes for which the order was issued or such disclosure was required by Applicable Law; provided, further, that the Confidential Information disclosed in response to such order or as required by Applicable Law shall be limited to the information that is legally required to be disclosed in response to such order or by such Applicable Law; and (b) the Receiving Party may disclose Confidential Information of the Disclosing Party to the extent any such disclosure is, in the opinion of the Receiving Party’s counsel, required by Applicable Law or the rules of a stock exchange on which the securities of the Receiving Party are listed (or to

64 which an application for listing has been submitted); provided that, in the event the Receiving Party is, in the opinion of its counsel, required by Applicable Law or the rules of a stock exchange on which its securities are listed (or to which an application for listing has been submitted) to make such a public disclosure, such Receiving Party shall submit the proposed disclosure to the Disclosing Party as far in advance as reasonably practicable so as to provide a reasonable opportunity to comment thereon. 9.3 Publicity. 9.3.1 Initial Press Releases. Each Party may issue its respective press release announcing this Agreement (including certain terms thereof) attached in Schedule 9.3.1 hereto following the Amended Effective Date. 9.3.2 Further Publicity. (a) Except as permitted pursuant to Section 9.2 or as expressly provided in Section 9.3.1, this Section 9.3.2(a), Section 9.3.2(b) or Section 9.4 in accordance with the terms therein, as applicable, neither Party shall issue any press release or public statement disclosing information relating to (a) with respect to both Parties, this Agreement or the transactions contemplated hereby or the terms hereof; or (b) with respect to Arrowhead, the Development, Manufacture or Commercialization of any Licensed Construct or Licensed Product in the Territory, in each case ((a) or (b)), without the prior written consent of such other Party. If a Party desires to make any such public announcement that would require the consent of the other Party in accordance with this Section 9.3.2, either directly or indirectly (such as through an Affiliate), such Party shall provide the other Party with a draft of the proposed announcement and provide the other Party a reasonable opportunity to comment on the nature, text, and timing of such announcement, which shall be brief and factual. (b) Notwithstanding Section 9.3.2(a), GSK shall have the right to issue any press release or public statement disclosing information with respect to the Development, Manufacture or Commercialization of any Licensed Construct or Licensed Product without requiring the prior written consent of Arrowhead. (c) Notwithstanding Section 9.3.2(a), but without limiting Arrowhead’s rights under Section 9.2, Arrowhead shall have the right to issue any press release, without requiring the prior written consent of GSK but subject to the terms of this Section 9.3.2(c), solely with respect to factual statements disclosing information with respect to the Development, Manufacture or Commercialization of any Licensed Construct or Licensed Product in the Territory, in each case, solely to the extent that such information has already been made public through a press release issued by GSK or any of its Affiliates or Sublicensees (provided that such previously publicized

65 information (i) is presented without changes to the substantive information provided in such initial press release by GSK or any of its Affiliates or Sublicensees, (ii) is still considered accurate and non-misleading and (iii) has not been superseded by other subsequent information known to Arrowhead (or any of its Affiliates) or otherwise subsequently publicly disclosed by GSK or any of its Affiliates or Sublicensees); provided that, prior to issuing any such press release pursuant to this Section 9.3.2(c), Arrowhead shall submit the proposed press release in writing to GSK as far in advance as reasonably practicable (and in no event less than [***]Business Days prior to the anticipated date of release) so as to provide GSK a reasonable opportunity to review and comment thereon and, upon GSK’s request, Arrowhead shall (A) delete any Confidential Information of GSK (or any of its Affiliates) from such press release; and (B) incorporate any other reasonable comments of GSK with respect thereto in good faith, as applicable. 9.4 Publications. Following the Amended Effective Date, Arrowhead shall not have the right to make any publications regarding the Development or Commercialization of any Licensed Construct or Licensed Product in the Territory, including to publish any results of any Clinical Trial or other Development activities conducted with respect to any Licensed Construct or Licensed Product, in each case, without the prior approval of GSK. Notwithstanding anything to the contrary in this Agreement, for the avoidance of doubt, GSK shall have the right to make any publications regarding the Development or Commercialization of any Licensed Construct or Licensed Product in the Territory as it chooses, in its sole discretion, without the approval of Arrowhead, including any Confidential Information as reasonably required for GSK’ s compliance with its then- current policy on the registration and reporting of results of pharmaceutical company- sponsored Clinical Trials including disclosures made by GSK on clinicaltrials.gov; provided that (a) GSK shall submit such publication to Arrowhead at least [***] Business Days in advance of the intended submission for publication or presentation of such publication for Arrowhead’s review; (b) to the extent Arrowhead notifies GSK of any specific, reasonable objections to such publication within [***] Business Days following GSK submitting such publication to Arrowhead for review pursuant to the foregoing clause (a), based on concern regarding the specific disclosure of any Confidential Information of Arrowhead (or any of its Affiliates), as applicable, GSK will delete any such Confidential Information and, acting reasonably and in good faith, consider any other such objections, including whether it is necessary or advisable to delete any other information from such proposed publication; and (c) upon Arrowhead’s request, GSK shall delay any such publication or presentation as needed to preserve the patentability of any Confidential Information of Arrowhead (or any of its Affiliates); provided, further, that Arrowhead may elect, by delivery of written notice to GSK, to extend such publication review period for up to an additional [***] Business Days solely to the extent necessary to permit the preparation and filing of any such patent applications with respect to such Confidential Information of Arrowhead (or its Affiliates), as applicable. Arrowhead agrees to provide, and to cause its applicable subcontractors to provide, to GSK such assistance as reasonably requested in connection with fulfilling the requirements of such policy.

66 9.5 Equitable Relief. Due to the unique nature of the Confidential Information, the Parties agree that any breach or threatened breach by a Party of this Article IX with respect to the other Party’s Confidential Information will cause not only financial harm to the other Party, but also irreparable harm for which money damages will not be an adequate remedy. Therefore, the other Party shall be entitled, in addition to any other legal or equitable remedies, to seek an injunction or similar equitable relief against any such breach or threatened breach by such Party without the necessity of proving actual damages or posting any bond. ARTICLE X: REPRESENTATIONS AND WARRANTIES 10.1 Mutual Representations and Warranties. Arrowhead and GSK each hereby represents and warrants to the other Party that, (a) with respect to Arrowhead, as of the Original Execution Date and the Original Effective Date (as though made then under the terms then in effect under the Original Agreement); and (b) with respect to each Party, as of the Amended Effective Date, that: 10.1.1 It has full right, power and authority to enter into this Agreement and to perform its respective obligations under this Agreement and that it has the right to grant to the other the licenses and sublicenses granted pursuant to this Agreement. 10.1.2 Except for any approvals from Regulatory Authorities (including pricing or reimbursement approvals, Manufacturing approvals or similar approvals necessary for the Development, Manufacture or Commercialization of the Licensed Products therein), all necessary consents, approvals and authorizations of all Governmental Authorities and other Persons required to be obtained by it (a) with respect to Arrowhead, as of the Original Effective Date (under the terms then in effect under the Original Agreement); and (b) with respect to each Party, as of the Amended Effective Date, in connection with the execution, delivery and performance of this Agreement have been obtained by such date. 10.1.3 Notwithstanding anything to the contrary in this Agreement, the execution and delivery of this Agreement by such warranting Party, the performance of such Party’s obligations hereunder (as contemplated (a) with respect to Arrowhead, as of the Original Effective Date (under the terms then in effect under the Original Agreement); and (b) with respect to each Party, as of the Amended Effective Date), and the licenses and sublicenses to be granted by such Party pursuant to this Agreement (i) do not conflict with or violate any requirement of Applicable Laws existing (A) with respect to Arrowhead, as of the Original Effective Date or (B) with respect to each Party, as of the Amended Effective Date, in each case, applicable to such Party, and (ii) do not conflict with, violate, breach or constitute a default under any contractual obligations of such Party or any of its Affiliates existing (A) with respect to Arrowhead, as of the Original Effective Date or (B) with respect to each Party, as of the Amended Effective Date, as applicable. Each Party shall, and shall cause its Affiliates to, comply with all Applicable Laws pertaining to the Development, Manufacture and Commercialization of the

67 Licensed Products, including applicable Drug Regulation Laws, Clinical Investigation Laws and Health Care Laws. 10.1.4 This Agreement is a legal and valid obligation binding upon the warranting Party and is enforceable against it in accordance with its terms. 10.2 Additional Representations and Warranties of Arrowhead. Arrowhead hereby represents and warrants to GSK, (a) as of the Original Execution Date (as though made then under the terms then in effect under the Original Agreement); and (b) as of the Amended Effective Date, that: 10.2.1 Arrowhead is not aware of any claim made against it (a) asserting the invalidity, misuse, unregisterability, unenforceability or non-infringement of any of the Arrowhead Patent Rights or the Existing Joint Agreement Patent Rights or (b) challenging Arrowhead’s ownership of, or license rights in, any of the Arrowhead Patent Rights or the Existing Joint Agreement Patent Rights. 10.2.2 The Arrowhead Patent Rights and the Existing Joint Agreement Patent Rights are (and through the Amended Effective Date remained) free and clear of any liens, charges and encumbrances (other than non-exclusive licenses under General Arrowhead Patent Rights granted by Arrowhead to Third Parties, which grants did not preclude (a) as of the Original Execution Date, Janssen from Exploiting the full scope of the licenses granted to Janssen as contemplated under the Original Agreement; or (b) as of the Amended Effective Date, GSK from Exploiting the full scope of the licenses granted to GSK as contemplated under this Agreement following the Amended Effective Date). Neither Arrowhead nor any of its Affiliates or their respective current or former employees, to the best of Arrowhead’s knowledge, has misappropriated any of the Arrowhead Know-How from any Third Party, and Arrowhead is not aware of any claim by a Third Party that such misappropriation has occurred. 10.2.3 To the best of Arrowhead’s knowledge, (a) (i) Exhibit B-1 of the Original Agreement and any updates provided thereto, lists all Specific Arrowhead Patent Rights owned solely or jointly by Arrowhead as of the Original Execution Date and (ii) Schedule 1.29 – Part I lists all Specific Arrowhead Patent Rights owned solely or jointly by Arrowhead as of the Amended Effective Date (collectively, the “Owned Specific Arrowhead Patent Rights”); (b) (i) Exhibit B-2 of the Original Agreement and any updates provided thereto, lists all General Arrowhead Patent Rights owned solely or jointly by Arrowhead as of the Original Execution Date and (ii) Schedule 1.29 – Part II lists all General Arrowhead Patent Rights owned solely or jointly by Arrowhead as of the Amended Effective Date (collectively, the “Owned General Arrowhead Patent Rights”); (c) (i) Exhibit B-3 of the Original Agreement and any updates provided thereto, lists all Specific Arrowhead Patent Rights acquired by Arrowhead from Third Parties as of the Original Execution Date, (ii) Schedule 1.29 – Part III lists all Specific Arrowhead Patent Rights acquired by Arrowhead from Third Parties, as of the Amended Effective Date (collectively, the “Acquired Specific Arrowhead Patent Rights”); (d) (i) Exhibit

68 B-3 of the Original Agreement and any updates provided thereto, lists all General Arrowhead Patent Rights acquired by Arrowhead from Third Parties as of the Original Execution Date and (ii) Schedule 1.29 – Part IV lists all General Arrowhead Patent Rights acquired by Arrowhead from Third Parties, as of the Amended Effective Date (collectively, the “Acquired General Arrowhead Patent Rights”); (e) (i) Exhibit B-4 of the Original Agreement and any updates provided thereto, lists all Specific Arrowhead Patent Rights and General Arrowhead Patent Rights licensed by Arrowhead from Third Parties as of the Original Execution Date and (ii) Schedule 1.29 – Part V lists all Specific Arrowhead Patent Rights and General Arrowhead Patent Rights licensed by Arrowhead from Third Parties as of the Amended Effective Date (collectively, the “In-Licensed Arrowhead Patent Rights”); and (f) Schedule 1.74 lists all Existing Joint Agreement Patent Rights as of the Amended Effective Date. No Third Party has an equal, undivided interest in the Acquired Specific Arrowhead Patent Rights, the Acquired General Arrowhead Patent Rights or the Existing Joint Agreement Patent Rights. To the best of Arrowhead’s knowledge (A) based on all records that Arrowhead possessed or were reasonably available to Arrowhead at any time on or before the Original Execution Date, the inventorship named as of the Original Execution Date in each issued Arrowhead Patent Right is correct; and (B) based on all records that Arrowhead possessed or were reasonably available to Arrowhead at any time on or before the Amended Effective Date, as applicable, the inventorship named as of the Amended Effective Date, as applicable, in each issued Arrowhead Patent Right or Existing Joint Agreement Patent Right is correct. 10.2.4 To the best of Arrowhead’s knowledge, no written claim of infringement of the Patent Rights of any Third Party has been made nor threatened in writing, (directly or indirectly) against Arrowhead or any of its Affiliates or, to the best of Arrowhead’s knowledge, Third Party contractors under any Pre-Existing Third Party Agreements with respect to the Development, Manufacture or Commercialization of any Licensed Construct or Licensed Product. 10.2.5 To the best of Arrowhead’s knowledge, Arrowhead has disclosed to Janssen all Patent Rights of Third Parties that Cover or are related to ARO-HBV as of the Original Execution Date. 10.2.6 There are no judgments or settlements against or owed by Arrowhead or its Affiliates or to which Arrowhead or its Affiliate is a party or, to the best of Arrowhead’s knowledge, pending litigation or litigation threatened in writing, in each case relating to any Licensed Construct or Licensed Product as of the Original Execution Date. 10.2.7 Neither Arrowhead nor, to Arrowhead’s knowledge, any of its Third Party licensors or assignors of any Arrowhead Intellectual Property is, or has been a party to, any agreement with the U.S. federal government or an agency thereof pursuant to which the U.S. federal government or such agency provided funding (such as under a grant or contract) for any research or Development work relating to any Licensed Construct or Licensed Product.

69 10.2.8 As of the Original Execution Date, Arrowhead made available to Janssen for review all material information in Arrowhead’s possession and control that, to the best of Arrowhead’s knowledge, pertains to any Licensed Constructs or Licensed Product, or the Development, Manufacture or Commercialization thereof, including complete and correct copies of the following (to the extent there are any) in Arrowhead’s possession and control as of the Original Execution Date: (a) adverse event data and reports; (b) Clinical Trial reports and study data, including all de- identified data, observations, analyses, conclusions, summaries, and reports resulting from the Clinical Trial of any Licensed Constructs initiated before the Original Execution Date; and (c) Regulatory Authority inspection reports, notices of adverse findings, warning letters, Regulatory Filings and letters and other correspondence with any Regulatory Authorities relating to any Licensed Construct or Licensed Product. 10.2.9 To Arrowhead’s knowledge, all of the studies and tests (including any pre-clinical studies and Clinical Trials) of any Licensed Constructs or Licensed Products conducted prior to the Original Execution Date, or by or on behalf of Arrowhead (or any of its Affiliates) under the Original Agreement, were conducted in material compliance with Applicable Laws. 10.3 Further Representations and Warranties of Arrowhead Regarding Pre-Existing Third Party Agreements. Arrowhead hereby represents and warrants to GSK, (a) as of the Original Execution Date (as though made then under the terms then in effect under the Original Agreement); and (b) as of the Amended Effective Date, that: 10.3.1 Arrowhead has made publicly available or otherwise provided to (a) Janssen, prior to the Original Execution Date; and (b) GSK, prior to the Amended Effective Date, with complete, correct and true, reasonably redacted copies of all Pre-Existing Third-Party Agreements (including any amendments thereof) set forth on Schedule 1.163. 10.3.2 Schedule 1.163 lists all of the Pre-Existing Third-Party Agreements, including any amendments thereto. As of the Amended Effective Date, (a) Part I of Schedule 1.163 is a list of all Pre-Existing Licenses to Third Parties; (b) Part II of Schedule 1.163 is a list of all Pre-Existing Licenses from Third Parties; and (c) Part III of Schedule 1.163 is a list of all Pre-Existing Acquired Rights from Third Parties. 10.3.3 To the best of Arrowhead’s knowledge, none of the terms of any Pre-Existing Third Party Agreement would have a material adverse effect on the Development or Commercialization of any Licensed Product. All Pre-Existing Third Party Agreements listed in Schedule 1.163 remain in full force and effect as of the Original Execution Date or as of the Amended Effective Date, as applicable, except where noted otherwise in Schedule 1.163, and to its knowledge, Arrowhead and each Third-Party counterparty has been, and is, in compliance in all material respects with the terms thereof. Arrowhead covenants that it shall use Commercially Reasonable Efforts not to take or omit to take any actions that would constitute a breach of any Pre-Existing Third Party Agreement through the

70 Amended Effective Date and during the Term hereof, and Arrowhead agrees not to enter into any amendment to any Pre-Existing Third Party Agreement through the Amended Effective Date or during the Term hereof, in each case which breach or amendment would have a material adverse effect on the Development or Commercialization of any Licensed Product as contemplated hereunder. During the Term Arrowhead shall provide GSK with prompt notice of the occurrence of any such breach (or receipt of notice of an allegation of any such breach). 10.3.4 The licenses and rights granted by Arrowhead under Section 2.1.1 and Section 2.1.2 of this Agreement are not subject to the terms of any Pre-Existing Third Party Agreements. The terms of such agreements listed in Schedule 1.163 do not preclude or prevent (a) as of the Original Execution Date, Janssen; or (b) as of the Amended Effective Date, GSK, as applicable, from Exploiting the full scope of the licenses and rights granted under Section 2.1.1 and Section 2.1.2 of this Agreement. 10.3.5 To the best of Arrowhead’s knowledge and except as set forth in the second sentence of Section, through the Amended Effective Date, Arrowhead has not entered into any agreements with any Third Party by virtue of which any royalty or milestone payment or other payment would be owed by (a) Janssen under the Original Agreement or (b) GSK under this Agreement following the Amended Effective Date, in each case, to such Third Party as a result of Commercialization of any Licensed Product by or on behalf of Janssen or GSK, as applicable, as contemplated hereunder. 10.3.6 Arrowhead is under no option, right of first offer or negotiation or similar obligation to any Third Party concerning a Licensed Construct or a Licensed Product, including ARO-HBV, and, to the extent any such obligation ever existed, conditions triggering such obligation have not been met or have been fully satisfied, as the case may be, to enable Arrowhead to exclusively license ARO-HBV to (a) Janssen under the Original Agreement or (b) GSK under this Agreement following the Amended Effective Date, as applicable. 10.3.7 ARO-HBV was not developed using Patent Rights or Know-How of a Third Party [***]. 10.3.8 Arrowhead has not granted any licenses or rights to Third Parties under any Arrowhead Patent Rights, Arrowhead Know-How, Existing Clinical Data or Existing Joint Agreement Patent Rights (a) that conflict with any of the licenses or rights granted by Arrowhead under Section 2.1.1 and Section 2.1.2 of this Agreement; or (b) to offer for sale, sell, or otherwise Commercialize any Licensed Constructs or Licensed Products in any field, which license has not expired or been terminated prior to the Original Execution Date or the Amended Effective Date, as applicable. 10.4 Additional Representations, Warranties and Covenants of Arrowhead as of the Amended Effective Date. Arrowhead hereby represents and warrants to GSK, as of Amended Effective Date, and further covenants, that:

71 10.4.1 Except to the extent set forth on Schedule 10.4.1, no inventions arising from any activities under the Original Agreement (or any Patent Rights therein) were assigned by Janssen to Arrowhead pursuant to Section 10.2.2 of the Original Agreement. 10.4.2 With respect to all Arrowhead Intellectual Property, (a) Arrowhead and its Affiliates have obtained from all employees and independent contractors who participated in the invention or authorship thereof, assignments of all ownership rights of such employees and independent contractors in such Arrowhead Intellectual Property, either pursuant to written agreement or by operation of Applicable Law; (b) all of its employees, officers, contractors, and consultants have executed agreements or have existing obligations under Applicable Law requiring assignment to Arrowhead or its Affiliate, as applicable, of all rights, title, and interests in and to their inventions made during the course of and as the result of this Agreement; and (c) no officer or employee of Arrowhead or its Affiliate is subject to any agreement with any other Third Party that requires such officer or employee to assign any interest in any Arrowhead Intellectual Property to any Third Party. 10.4.3 With respect to any Human Biological Samples collected or used in connection with the conduct of any Clinical Trial of any Licensed Product by or on behalf of Arrowhead or any of its Affiliates (either prior to the Original Effective Date or during the Term of the Original Agreement prior to the Amended Effective Date), (a) any such Human Biological Samples used in the Development, Manufacture or Commercialization of any Licensed Constructs or Licensed Products conducted by or on behalf of Arrowhead or any of its Affiliates as of the Amended Effective Date have been obtained, stored, transferred, used and disposed of in accordance in all material respects with Applicable Laws and any generally accepted ethical guidelines regarding the collection, use, transport and disposal of human tissue; (b) all ethics committee approvals have been obtained to enable the use of any such Human Biological Samples obtained from patients or human subject volunteers or other donors in connection with the Development, Manufacture or Commercialization of any Licensed Constructs or Licensed Products conducted by or on behalf of Arrowhead or any of its Affiliates as of the Amended Effective Date; (c) all uses of any such Human Biological Samples in the Development, Manufacture or Commercialization of any Licensed Constructs or Licensed Products conducted by or on behalf of Arrowhead or any of its Affiliates as of the Amended Effective Date fall within the terms of the informed consent given by the donors of such Human Biological Samples; and (d) no human embryonic or fetal derived material (including cell lines) have been used in the Development, Manufacture or Commercialization of any Licensed Constructs or Licensed Products conducted by or on behalf of Arrowhead or any of its Affiliates as of the Amended Effective Date. 10.5 No Warranties. EXCEPT AS OTHERWISE EXPRESSLY SET FORTH IN THIS AGREEMENT, NEITHER PARTY MAKES ANY REPRESENTATION OR EXTENDS ANY WARRANTIES OF ANY KIND, EITHER EXPRESS OR IMPLIED, TO THE

72 OTHER PARTY, AND EACH PARTY HEREBY DISCLAIMS ALL IMPLIED WARRANTIES OF MERCHANTABILITY AND FITNESS FOR A PARTICULAR PURPOSE. EACH PARTY HEREBY DISCLAIMS ANY REPRESENTATION OR WARRANTY THAT THE DEVELOPMENT, MANUFACTURE AND COMMERCIALIZATION OF LICENSED CONSTRUCTS OR LICENSED PRODUCTS PURSUANT TO THIS AGREEMENT WILL BE SUCCESSFUL OR THAT ANY PARTICULAR SALES LEVEL WITH RESPECT TO LICENSED PRODUCTS WILL BE ACHIEVED. 10.6 No Debarment. Each Party represents and warrants, as of the Amended Effective Date (and Arrowhead represents and warrants to GSK as of the Original Effective Date), that neither it nor any of its Affiliates has been debarred or is subject to debarment, and neither Party nor any of its Affiliates will use in any capacity, in connection with the Development, Manufacture or Commercialization of any products, any person who has been debarred pursuant to Section 306 of the United States Federal Food, Drug, and Cosmetic Act, or any foreign equivalent thereof, or who is the subject of a conviction described in such section. Each Party agrees to inform the other Party in writing immediately if it or any person who is performing activities hereunder is debarred or is the subject of a conviction described in Section 306 of the United States Federal Food, Drug, and Cosmetic Act, or any foreign equivalent thereof, or if any action, suit, claim, investigation or legal or administrative proceeding is pending or, to the best of such Party’s knowledge, is threatened, relating to the debarment or conviction of such Party or any person used in any capacity by such Party or any of its Affiliates in connection with the Development, Manufacture or Commercialization of any Licensed Construct or Licensed Products. 10.7 Compliance with Anti-Corruption Applicable Laws. Each Party shall, and shall cause each of its Affiliates and Third Party subcontractors and sublicensees conducting activities hereunder to, comply with Anti-Corruption Laws. 10.8 Maintenance of Arrowhead Intellectual Property. During the Term, Arrowhead shall not (and shall cause its Affiliates to not) (a) assign, transfer, convey, encumber (through any liens, charges, security interests, mortgages or similar actions) or dispose of, or enter into any agreement with any Third Party to assign, transfer, convey, encumber (through lien, charge, security interest, mortgage or similar action) or dispose of, any of the Arrowhead Intellectual Property to any Third Party without the prior consent of GSK; or (b) fail to maintain any of the Arrowhead Intellectual Property in the ordinary course of business, and in compliance with Applicable Law, in each case ((a) or (b)), in any manner that would conflict with, limit the scope of or adversely affect in any material respect any of the rights or licenses granted to GSK under this Agreement, including under Section 2.1.1 and Section 2.1.2. During the Term, Arrowhead covenants (on behalf of itself and its Affiliates) to ensure that any Arrowhead Intellectual Property is and remains Controlled by Arrowhead (or its Affiliates), such that Arrowhead maintains the full rights to grant the rights and licenses to the Arrowhead Intellectual Property to GSK as contemplated hereunder, including under Section 2.1.1 and Section 2.1.2. 10.9 Maintenance of GSK Agreement LC/LP IP. Subject to Section 8.3.4(g), during the Term, GSK shall not (and shall cause its Affiliates to not) (a) assign, transfer, convey,

73 encumber (through any liens, charges, security interests, mortgages or similar actions) or dispose of, or enter into any agreement with any Third Party to assign, transfer, convey, encumber (through lien, charge, security interest, mortgage or similar action) or dispose of, any of the GSK Agreement LC/LP IP to any Third Party without the prior consent of Arrowhead; or (b) fail to maintain any of the GSK Agreement LC/LP IP in the ordinary course of business, and in compliance with Applicable Law, in each case ((a) or (b)), in any manner that would conflict with, limit the scope of or adversely affect in any material respect any of the rights or licenses granted to Arrowhead under any such GSK Agreement LC/LP IP pursuant to this Agreement, including the licenses contemplated to be granted to Arrowhead under any such GSK Agreement LC/LP IP that constitute GSK Reversion IP as of the effective date of termination in accordance with Section 13.6.2, as applicable; provided, however, that nothing in this Section 10.9 shall be deemed to limit or otherwise restrict GSK’s (or any of its Affiliates’) rights (i) with respect to the Prosecution, enforcement or defense of such GSK Agreement LC/LP IP in accordance with Article VIII, (ii) to grant licenses or sublicenses (including through multiple tiers) to any Third Party under such GSK Agreement LC/LP IP during the Term, subject to Arrowhead’s rights and licenses under this Agreement, including the licenses of Section 13.6.2, or (iii) to otherwise use or practice any such GSK Agreement LC/LP IP, including in connection with the Development, Manufacture, Commercialization or other Exploitation of any Licensed Constructs or Licensed Products under this Agreement. ARTICLE XI: INDEMNIFICATION AND INSURANCE 11.1 Indemnification Obligation. Each Party (the “Indemnifying Party”) shall defend, indemnify and hold harmless the other Party and its Indemnified Persons (collectively, the “Indemnified Party”) from and against any and all Losses to which any Indemnified Party may become subject as a result of any Action brought by a Third Party against such Indemnified Party to the extent such Losses arise out of or are caused by: (a) the gross negligence or willful misconduct of the Indemnifying Party or any of its Indemnified Persons, in each case in connection with the exercise of such Indemnifying Party’s rights, or performance of such Indemnifying Party’s obligations, under this Agreement; (b) the Indemnifying Party’s breach of any of its representations and warranties, covenants, agreements or obligations under this Agreement; (c) the violation of Applicable Law by the Indemnifying Party or any of its Indemnified Persons in connection with the exercise of such Indemnifying Party’s rights, or performance of such Indemnifying Party’s obligations, under this Agreement; (d) the Development or Manufacture of any Licensed Construct or Licensed Product under this Agreement by or on behalf of the Indemnifying Party, or any of its Affiliates, Sublicensees or (sub)licensees, or any of its or their contractors; and (e) in the case of GSK as the Indemnifying Party, the Commercialization, of any Licensed Products by or on behalf of GSK or any of its Affiliates or its Sublicensees or any of its or their contractors; in each case ((a) through (e)), except to the extent that such Losses are covered by the other Party’s indemnification obligations under this Section 11.1, as applicable. For clarity, for purposes of this Section 11.1, in no event shall either Party (or any of its Affiliates or its or their respective Sublicensees, (sub)licensees or contractors) be deemed to be acting on behalf of the other Party (or any of its Affiliates or its or their respective Sublicensees, (sub)licensees or contractors).

74 11.2 Claims for Indemnification. 11.2.1 Notice. In the case of any Action for which an Indemnifying Party may be liable to an Indemnified Person under Section 11.1, the Indemnified Party shall as soon as practicable notify the Indemnifying Party in writing of such Action (a “Notice of Claim”). Failure or delay in notifying the Indemnifying Party shall not relieve the Indemnifying Party of any liability it may have to the Indemnified Party, except and only to the extent that such failure or delay causes actual harm to the Indemnifying Party with respect to such Action. The Notice of Claim shall specify in reasonable detail the Action with respect to which such Indemnified Party or any of its Indemnified Persons intends to base a request for indemnification or reimbursement under Section 11.1. Failure to provide such reasonable detail will not relieve the Indemnifying Party of any liability it may have to the Indemnified Party, except and only to the extent that such failure causes actual harm to the Indemnifying Party with respect to such Action. The Indemnified Party shall enclose with the Notice of Claim a copy of all papers served with respect to such Action, if any. The Indemnified Party shall assume the defense, settlement or other disposal of such Action, unless it provides notice within thirty (30) days from the date on which the Indemnifying Party received the Notice of Claim that it waives its right to assume the defense of such Action and any litigation resulting therefrom with counsel of its choice. Provided that the Indemnified Party has waived its right to assume the defense of an Action pursuant to this Section 11.2, then, subject to Section 11.2.3, the Indemnifying Party shall have the obligation to defend, settle and otherwise dispose of such Action. 11.2.2 Cooperation. The Parties shall act in good faith in responding to, defending against, settling or otherwise dealing with such Action pursuant to the terms hereof; provided that (a) an Indemnified Party shall not be obligated to enter into or consent to the entry of any judgment or settlement in relation to any Action as provided in Section 11.2.3, and (b) in any event, an Indemnifying Party shall not be relieved of its obligations under this Section 11.2.2 as a result of any failure of the Indemnified Party to cooperate as provided in this Section 11.2.2, except to the extent that the Indemnifying Party is actually prejudiced by such breach. The Parties shall also cooperate in any such defense by giving each other reasonable access to all non- privileged information relevant thereto to the extent permitted by Applicable Law. 11.2.3 Control by the Indemnifying Party. If the Indemnifying Party assumes control of an Action in accordance with Section 11.2.1, (a) the Indemnified Party may retain separate co-counsel at its sole cost and expense and participate in the defense of the Action, but the Indemnifying Party shall continue to control the investigation, defense and settlement thereof, and (b) the Indemnifying Party will not, without the prior written consent of the Indemnified Party, which shall not be unreasonably withheld, conditioned or delayed, consent to the entry of any judgment or enter into any settlement with respect to the Action to the extent such judgment or settlement (i) provides for equitable relief (or any other relief other than solely for money damages) against the Indemnified Party or any of its Indemnified Persons, or liability or obligation that cannot be assumed and performed by the Indemnifying

75 Party in full (without any recourse to the Indemnified Party and its Indemnified Persons), (ii) provides for any monetary relief that will not be fully discharged by the Indemnifying Party (without any recourse to the Indemnified Party and its Indemnified Persons) concurrently with the effectiveness of such judgment or settlement, (iii) does not effect a full and unconditional release of the Indemnified Party and its Indemnified Persons with respect to all claims in such Action (or the portion thereof to which the judgment or settlement relates), or (iv) that contains an admission of wrongdoing on the part of the Indemnified Party or its Indemnified Persons. 11.2.4 Interim Control. Unless and until the Indemnifying Party (if any) is determined with respect to any particular Action, the Party subject to such Action shall have the right to defend and control such Action, but shall not have the right to consent to the entry of any judgment or enter into any settlement with respect to the Action for which it would be seeking indemnification or reimbursement hereunder without the prior written consent of the other Party (which consent shall not be unreasonably withheld, conditioned or delayed). 11.2.5 Unauthorized Settlements. The Indemnified Party will not consent to the entry of any judgment or enter into any settlement with respect to any Action for which it is seeking indemnification hereunder without the prior written consent of the Indemnifying Party (which consent shall not be unreasonably withheld, conditioned or delayed), and such Indemnifying Party shall not be obligated to indemnify or reimburse the Indemnified Party hereunder for any settlement entered into, or any judgment that was consented to, by the Indemnified Party without the Indemnifying Party’s prior written consent. 11.2.6 Allocation. If, in any Action under this Article XI, the Indemnified Party incurs an amount consisting of both Losses for which the Indemnifying Party is obliged to indemnify the Indemnified Party and Losses not covered by such indemnification, then, to the extent not otherwise determined in a court of competent jurisdiction, the Parties agree to act in good faith and use their reasonable endeavours to determine a fair and reasonable allocation of such Losses. The allocation between the Parties of any such Losses, if not otherwise determined in a court of competent jurisdiction, shall, if the Parties do not reach agreement in writing on such allocation, be determined by arbitration pursuant to Section 14.3, The Parties or the arbitrator, as the case may be, shall make such allocation based on the indemnification and reimbursement principles set forth in this Article XI. Notwithstanding the foregoing, the Parties shall not be entitled to refer any Dispute with respect to Losses arising under an Action pursuant to this Section 11.2.6 to arbitration to the extent that the liability of either Party for such Losses is being contested in such Action (or any other Action that would be binding with respect to such first Action). 11.3 Mitigation. The Indemnified Party shall, and shall procure that its Indemnified Persons shall, in each instance, take reasonable steps to mitigate any Losses they suffer arising in

76 connection with any Action in respect of which they seek an indemnity from the other Party under this Agreement. 11.4 Insurance. 11.4.1 Each Party shall procure and maintain in full force and effect, at its own cost, insurance (or self-insure sufficiently to provide materially the same level and type of protection) adequate to cover its obligations and liabilities hereunder during the Term and for a period of [***] years thereafter, consistent with normal business practices of companies similarly situated. It is understood that such insurance shall not be construed to create a limit of either Party’s liability with respect to its indemnification obligations under this Agreement. 11.4.2 Prior to the initiation of any Clinical Trial or related Development activities under this Agreement, the Party responsible for the applicable activity shall secure and maintain in full force and effect, at its own cost, Clinical Trial insurance (including any self-insured arrangements) in compliance with Applicable Law in those territories where Clinical Trials are conducted. 11.4.3 The Parties have the right to elect to self-insure all or part of the limits described above. Upon written request, each Party shall provide the other with a certificate of insurance evidencing the required coverage hereunder; provided, further, that if, at any time during the Term, a Party ceases to maintain the same level of insurance coverage with respect to such Party’s obligations under this Agreement, such Party shall promptly notify the other Party thereof. Notwithstanding the foregoing, either Party’s failure to maintain adequate insurance shall not relieve that Party of its obligations set forth in this Agreement. 11.5 Limitation of Liability. NOTWITHSTANDING ANYTHING TO THE CONTRARY IN THIS AGREEMENT, TO THE MAXIMUM EXTENT PERMITTED BY LAW, EXCEPT WITH RESPECT TO (A) EACH PARTY’S RESPECTIVE INDEMNIFICATION OBLIGATIONS FOR LOSSES CAUSED BY OR ARISING OUT OF THIRD PARTY CLAIMS UNDER SECTION 11.1; (B) THE GROSS NEGLIGENCE, WILLFUL MISCONDUCT OR FRAUD OF A PARTY; OR (C) EITHER PARTY’S BREACH OF ITS CONFIDENTIALITY AND NON-USE OBLIGATIONS UNDER Article IX, IN NO EVENT SHALL EITHER PARTY OR ANY OF ITS AFFILIATES BE LIABLE TO THE OTHER PARTY OR ANY OF ITS AFFILIATES FOR ANY INDIRECT, PUNITIVE, SPECIAL, INCIDENTAL, EXEMPLARY OR CONSEQUENTIAL DAMAGES (INCLUDING FOR LOST REVENUES AND LOST PROFITS (WHETHER DIRECT OR INDIRECT)), REGARDLESS OF THE THEORY OF LIABILITY (INCLUDING CONTRACT, TORT, NEGLIGENCE, STRICT LIABILITY OR OTHERWISE), IN EACH CASE, ARISING OUT OF OR RELATING TO THIS AGREEMENT, THE TRANSACTIONS CONTEMPLATED HEREIN OR ANY BREACH HEREOF, IRRESPECTIVE OF WHETHER SUCH PARTY OR ANY REPRESENTATIVE OF SUCH PARTY HAS BEEN ADVISED OF, OR OTHERWISE MIGHT HAVE ANTICIPATED THE POSSIBILITY OF, ANY SUCH LOSS OR DAMAGE OR WHETHER SUCH LOSS OR DAMAGE WAS REASONABLY FORESEEABLE.

77 ARTICLE XII: EXCLUSIVITY 12.1 Exclusivity. During the period commencing on the Amended Effective Date and ending upon the [***] anniversary thereof (the “Exclusivity Term”), Arrowhead shall not, and shall cause its Affiliates not to (a) alone or with any Affiliates or Third Parties, Develop, Manufacture or Commercialize any double-stranded RNAi Trigger designed to hybridize to the HBV genome to effect silencing through [***] (each of the foregoing, together with any construct, compound or product that contains or comprises such RNAi Trigger or Construct, a “Competing Product”); or (b) enter into an agreement or other arrangement with any Third Party pursuant to which Arrowhead or one of its Affiliates grants such Third Party any license or other rights to Develop, Manufacture or Commercialize a Competing Product, in each case ((a) or (b)), in the Field in the Territory. 12.2 Change of Control Exception. Notwithstanding Section 12.1, in the event that, during the Exclusivity Term, a Change of Control occurs with respect to Arrowhead or any of its Affiliates, and an Acquirer owns or has any license or other rights to any Competing Product that would otherwise cause Arrowhead to be in violation of its exclusivity obligations under Section 12.1, then, such Acquirer and any of such Acquirer’s Affiliates (but excluding Arrowhead and all of its Affiliates immediately prior to such Change of Control) will not be prohibited from Developing, Manufacturing or Commercializing such Competing Product as a result of Arrowhead’s exclusivity obligations under Section 12.1 for so as long as: 12.2.1 such Acquirer (and any such Affiliates) does not use, access or reference any Joint Agreement IP, GSK Agreement IP or any other Confidential Information of GSK in connection with the Development, Manufacture or Commercialization of such Competing Product; and 12.2.2 such Acquirer institutes (with respect to itself and any such Affiliates) commercially reasonable technical and administrative safeguards to ensure the requirements set forth in Section 12.2.1 are met, including by (a) separating (i) any Personnel of such Acquirer (or any such Affiliates) conducting activities with respect to any Competing Product and (ii) any Personnel of Arrowhead (or any of its Affiliates immediately prior to such Change of Control) conducting any activities under this Agreement or who have access to any Joint Agreement IP, GSK Agreement IP or any other Confidential Information of GSK; and (b) the maintenance of separate lab notebooks and records and separate Personnel with respect to each of the activities under this Agreement and the activities with respect to such Competing Product. 12.3 Acquired Business Exception. Notwithstanding Section 12.1, in the event that, during the Exclusivity Term, Arrowhead or any of its Affiliates acquires any assets or business, whether accomplished by way of merger, business combination, asset purchase, stock purchase or otherwise (the “Acquired Business”), and such Acquired Business, immediately prior to such acquisition, owns, has or includes any license or other right to any Competing Product that would otherwise cause Arrowhead to be in violation of its exclusivity obligations under Section 12.1, then, Arrowhead will (a) notify GSK of such

78 Competing Product in writing no later than [***] days after the consummation of the acquisition of such Acquired Business; and (b) terminate the Exploitation of such Competing Product pursuant to Section 12.3.1 or divest such Competing Product pursuant to Section 12.3.2 (provided that Arrowhead shall specify which of the following it will perform in the notice provided pursuant to subsection (a) above, which decision will be final and binding on Arrowhead and its Affiliates); provided, further, that, during the pendency of any termination pursuant to Section 12.3.1 or divestiture pursuant to Section 12.3.2, as applicable, Arrowhead and its Affiliates also will comply will the requirements specified in Section 12.2.1 and Section 12.2.2 mutatis mutandis: 12.3.1 Arrowhead may elect to terminate the Exploitation of such Competing Product, in which case Arrowhead and its Affiliates will cease all Exploitation of such Competing Product as soon as reasonably practicable and in any event within [***] days after the consummation of the acquisition of such Acquired Business, giving due consideration to ethical concerns and requirements under Applicable Law and any agreements with Third Parties; provided that Arrowhead shall notify GSK in writing of such completed termination; or 12.3.2 Arrowhead may elect to divest itself (or cause its Affiliate to divest itself) such Competing Product and notify GSK in writing of such completed divestiture; provided that such divestiture shall be completed within [***] months after the consummation of the acquisition of such Acquired Business. 12.4 Non-Controlling Business Exception. For the avoidance of doubt, and notwithstanding anything in this Agreement to the contrary, nothing in Section 12.1 will be construed as prohibiting the acquisition by Arrowhead or any of its Affiliates of any non-controlling equity interest in any Person that owns or has any license or other right to any Competing Product; provided that, as part of such transaction or a related transaction, (a) Arrowhead or such Affiliate is not granted any ownership, license, option or other interest or right in, as applicable, such Competing Product; (b) Arrowhead or such Affiliate is not granted any voting board seat or any other right to direct the management of such Person or the Development, Manufacture or Commercialization of such Competing Product; and (c) such Person is not granted by Arrowhead or such Affiliate any license or right to use any Joint Agreement IP, GSK Agreement IP or any other Confidential Information of GSK in connection with the Development, Manufacture or Commercialization of such Competing Product. 12.5 No Implied Exclusivity. Except for the restrictions expressly set forth in this Agreement, including as set forth in this Article XII and the exclusive licenses granted to GSK under Section 2.1.1 or Section 2.1.2, nothing in this Agreement shall be construed to restrict the right of either Party or any of its Affiliates to engage in any business activity, investment or other opportunity anywhere in the world, including the right of Janssen or Arrowhead or any of their Affiliates to Develop and Commercialize any product that directly or indirectly competes with a Licensed Product in any field.

79 ARTICLE XIII: TERM AND TERMINATION 13.1 Agreement Term. Unless terminated earlier in accordance with this Article XIII, the term of this Agreement (the “Term”) shall commence on the Amended Effective Date and shall continue in full force and effect, on a country-by-country and Licensed Product-by- Licensed Product basis, until the expiration of the Royalty Term for the applicable Licensed Product in such country; provided that this Agreement shall expire in its entirety upon the last to expire Royalty Term for all Licensed Products under this Agreement. 13.2 Early Termination for Breach. 13.2.1 Notice of Default and Cure Period. Upon any material breach of this Agreement by a Party (the “Breaching Party”), the other Party (the “Non-Breaching Party”) shall have the right to give the Breaching Party notice specifying the nature of such material breach. If the breach of this Agreement is curable, then the Breaching Party shall have a period of [***] days from the date of receipt of the notice (the “Cure Period”) to cure such material breach in a manner that effectively remedies the harm to the Non-Breaching Party caused by the material breach. Notwithstanding the foregoing, if such breach, by its nature, is curable, but is not reasonably curable within the Cure Period, then provided that such breach is not of a payment obligation hereunder, such Cure Period shall be extended if the Breaching Party provides a written plan for curing such breach to the Non- Breaching Party and uses Commercially Reasonable Efforts to cure such breach in accordance with such written plan, provided that no such extension shall exceed [***] days (for an extended Cure Period totaling [***] days) without the consent of the Non-Breaching Party. For clarity, this provision shall not restrict in any way either Party’s right to notify the other Party of any other breach or to demand the cure of any other breach. 13.2.2 Termination Right for Default. The Non-Breaching Party shall have the right to terminate this Agreement with immediate effect by written notice to the Breaching Party: (a) in the event that the Breaching Party has not cured the material breach within the Cure Period (as may be extended pursuant to Section 13.2.1); or (b) in the event that the material breach is not curable. Notwithstanding the foregoing, if a Party in good faith raises a Dispute, pursuant to the Dispute resolution procedures under Article XIV, with respect to the existence, materiality of a breach or the sufficiency or failure of a cure of any such breach that would give rise to a right to terminate pursuant to this Section 13.2, then such termination shall be effective only upon a conclusion of the Dispute resolution procedures in accordance with Article XIV resulting in a determination that there has been an uncured material breach (or, if earlier, abandonment of the Dispute by the Breaching Party); provided, however, that the applicable Cure Period shall be tolled during the pendency of any such Dispute and shall recommence following such final resolution of such Dispute in accordance with Article XIV. For the avoidance of doubt, the exercise of a termination right under this Section 13.2 by a Non-Breaching Party shall be without prejudice to its right to seek damages or any other remedy on account of the

80 Breaching Party’s material breach that may be available at law or in equity, subject to the terms hereof. 13.3 Early Termination for Bankruptcy. 13.3.1 In the event of the Bankruptcy of a Party (or its successor in interest in the event this Agreement is assigned as permitted hereunder) (the “Bankrupt Party”), the other Party (the “Non-Bankrupt Party”) may terminate this Agreement with immediate effect by written notice to the Bankrupt Party. 13.3.2 All licenses and other rights granted pursuant to this Agreement by one Party to the other are, and will otherwise be deemed to be, for purposes of Section 365(n) of the Bankruptcy Code (or comparable provisions of laws of other jurisdictions), licenses of right to “intellectual property” as defined under Section 101 of the Bankruptcy Code (or comparable provisions of Applicable Laws of other jurisdictions). Notwithstanding anything to the contrary herein, the Parties agree that, upon commencement of a Bankruptcy proceeding by, or entry of an order for relief in connection with an Bankruptcy proceeding against, the Bankrupt Party, under the Bankruptcy Code (or comparable provisions of Applicable Laws of other jurisdictions) (collectively, the “Bankruptcy Commencement Date”), (a) in lieu of the Non-Bankrupt Party who is licensed (or sublicensed) any rights from such Bankrupt Party terminating this Agreement in its entirety as provided in Section 13.3.1, such Non-Bankrupt Party who is a licensee of such rights from the Bankrupt Party under this Agreement shall, upon such Bankrupt Party’s Bankruptcy, retain and may fully exercise all of the rights and elections under the Bankruptcy Code (or comparable Applicable Laws of other jurisdictions); or (b) the Non-Bankrupt Party, in addition to its rights under Section 13.3.1 or otherwise under this Agreement, will be entitled to a complete duplicate of, or complete access to (as the Non-Bankrupt Party deems appropriate), any such intellectual property and all embodiments of such intellectual property to which such Non-Bankrupt Party is granted license or other rights hereunder, and the same, if not already in its possession, shall be promptly delivered to the Non-Bankrupt Party (i) following any such Bankruptcy Commencement Date, within ten (10) Business Days of receiving written request by the Non-Bankrupt Party, unless the Bankrupt Party has assumed this Agreement prior to receipt of such written request by the Non- Bankrupt Party, or (ii) if not delivered under foregoing clause (i), on or before entry of an order by a competent court having jurisdiction over the matter authorizing the rejection of this Agreement. The Bankrupt Party (in any capacity, including debtor- in-possession) and its successors and assigns (including any trustee) agree not to interfere with the exercise by the Non-Bankrupt Party of its rights and licenses to such intellectual property and such embodiments of intellectual property in accordance with this Agreement. In addition, the Bankrupt Party waives to the fullest extent permitted by Applicable Law any and all rights to sell its intellectual property assets (including any Patent Rights) free and clear of the Non-Bankrupt Party’s rights and licenses in and to such intellectual property whether pursuant to section 363 of the Bankruptcy Code or pursuant to a chapter 11 plan. All rights, powers and remedies granted hereunder to a Non-Bankrupt Party as a licensee of

81 any Patent Rights, Know-How or other intellectual property rights as provided in this Agreement (including as set forth in Section 2.2 or this Section 13.3) are in addition to and not in substitution for any and all other rights, powers and remedies now or hereafter existing at law or in equity, in the event of the commencement of a Bankruptcy proceeding by or against the Bankrupt Party under Applicable Law, and the Non-Bankrupt Party, in addition to the rights, powers and remedies expressly provided herein, shall be entitled to exercise all other such rights and powers and resort to all other such remedies as may now or hereafter exist at law or in equity in such event. 13.4 Termination by GSK for Safety Concern. GSK may terminate this Agreement with immediate effect by written notice to Arrowhead in the event that GSK determines, in its good-faith judgment, that continued Development or Commercialization of a Licensed Product would be unethical or unreasonable due to a safety-related reason, such as if GSK believes, based on its good-faith assessment of relevant data, that continuation of human use of a Licensed Product has resulted in, or has a significant risk of resulting in, the occurrence of a safety or tolerability finding that would raise material concerns regarding the clinical benefit of the Licensed Product for its target population (for example, harm significantly in excess of an acceptable side-effect profile). Such termination shall be effective immediately upon GSK’s written notice to Arrowhead. 13.5 Discretionary Termination by GSK. GSK shall have the right to terminate this Agreement for convenience at any time by written notice, which termination shall be effective (a) [***] from the date of such notice in the event that notice is given prior to the First Commercial Sale of any Licensed Product; and (b) [***] from the date of such notice in the event that notice is given following the First Commercial Sale of any Licensed Product. 13.6 Consequences of Early Termination. Upon the effective date of any early termination of this Agreement, the following shall apply. 13.6.1 Termination of Rights and Obligations. Each Party’s respective rights, licenses and obligations under this Agreement, including the licenses and other rights granted by one Party to the other in Article II, shall automatically terminate and have no further force and effect as of the applicable effective date of termination; provided, however, that each Party shall retain such rights or licenses to the extent necessary to enable the grantee Party (or its Affiliates) to perform any obligations or exercise any rights that survive such termination of this Agreement as may be expressly provided in this Agreement, including pursuant to Section 13.9, this Section 13.6 or in any written agreement of the Parties, as applicable. 13.6.2 Reversion License Grant. (a) [***] effective upon the applicable effective date of such termination, GSK, on behalf of itself and its Affiliates, hereby grants (without any further subsequent action required on the part of Arrowhead) to Arrowhead and its Affiliates a [***] license under the GSK Reversion IP to Develop,

82 Commercialize and otherwise Exploit any Terminated Products in the Field in the Territory; provided, however, that the foregoing license will be [***] with respect to any Janssen Licensed Patent Rights included within such GSK Reversion IP. (b) With respect to any GSK Excluded Know-How or GSK Excluded Patent Rights, upon Arrowhead’s request, the Parties shall negotiate in good faith the terms of a [***] license under such GSK Excluded Know-How or GSK Excluded Patent Rights, which license shall include commercially reasonable financial terms payable by Arrowhead to GSK with respect thereto. 13.6.3 Patent Matters. (a) Arrowhead shall assume from GSK the sole responsibility for the Prosecution, defense and enforcement of any Arrowhead Patent Rights for which GSK was the Party responsible for Prosecution. Upon Arrowhead’s request, GSK shall reasonably cooperate in transferring to Arrowhead responsibility for the Prosecution, defense and enforcement of such Arrowhead Patent Rights, and shall provide Arrowhead with copies, at Arrowhead’s expense, of any requested documents in its possession relating thereto. (b) Solely in the event of a termination of this Agreement by (i) Arrowhead pursuant to Section 13.2.2, or (ii) GSK pursuant to Section 13.5, then, in each case ((i) or (ii)), upon Arrowhead’s request, the Parties will negotiate in good faith and agree upon terms with respect to the GSK Reversion Patent Rights, excluding any Janssen Licensed Patent Rights therein, pursuant to which (A) in the event that GSK elects not to Prosecute (or continue to Prosecute, including filing a Patent Right claiming priority to a Patent Right prior to its issuance) any such GSK Reversion Patent Right, Arrowhead will have the right to assume sole responsibility for such Prosecution thereof; and (B) in the event that GSK decides not to take any action to enforce any such GSK Reversion Patent Right in connection with a Product Infringement involving a Terminated Product or defend a Third Party Action challenging any such GSK Reversion Patent Right, Arrowhead will have the right to bring such enforcement action or defend such Third Party Action. 13.6.4 Transfer of GSK Reversion Know-How. [***] then, promptly following the effective date of such termination, at Arrowhead’s cost and in a manner and format mutually agreed by the Parties, GSK shall provide to Arrowhead copies of any GSK Reversion Know-How under which Arrowhead has a license grant pursuant to Section 13.6.2, in each case, solely to the extent that, as of the effective date of such termination, such copies are in GSK’s (or its Affiliate’s) possession and Control and in the format ready available to GSK (or its Affiliates); provided, however, that with respect to any such GSK Reversion Know-How that (a) constitutes New

83 Arising Know-How; and (b) is specifically related to the Manufacturing of any Terminated Product, then, in lieu of providing such copies to Arrowhead directly, GSK shall have the right, in its sole discretion, to elect to provide copies of such GSK Reversion Know-How to a Third Party commercial manufacturer selected by Arrowhead (and reasonably acceptable to GSK), which Third Party commercial manufacturer may be engaged by or on behalf of Arrowhead to Manufacture such Terminated Product; provided, further, that Arrowhead shall be responsible and liable for the compliance by any such Third Party commercial manufacturer with the relevant terms and conditions of this Agreement that survive such termination and shall require that any such Third Party commercial manufacturer enter into a written agreement that complies with the requirements for Third Party subcontractors as set forth in Section 3.12 (mutatis mutandis). For clarity, [***], shall be deemed a reasonably acceptable Third Party commercial manufacturer by GSK for purposes of this Section 13.6.4 unless [***]. 13.6.5 Transfer and Assignment of Regulatory Filings and Regulatory Approvals. [***] in accordance with and to the extent permissible under Applicable Law, GSK shall promptly transfer and assign, or cause to be transferred and assigned, to Arrowhead (or its designee) (a) any and all Regulatory Approvals for a Terminated Product in the Territory; and (b) any material Regulatory Filings that specifically related to any Terminated Product in the Territory, in each case ((a) or (b)), that is owned and Controlled by GSK or its Affiliates as of the effective date of such termination and Arrowhead shall assume full responsibility for such Regulatory Filings and Regulatory Approvals; provided, however, that in no event shall GSK have any obligation to transfer or assign any Regulatory Approvals for, or any Regulatory Filings to the extent related to, (i) any Terminated Product that is a Combination Product or (ii) any compound, construct or product that is not a Terminated Product. In the event that GSK is unable to transfer and assign, or have transferred and assigned, to Arrowhead (or its designee) any such Regulatory Filing or Regulatory Approval, effective upon the effective date of termination, GSK, on behalf of itself and its Affiliates, hereby consents and grants to Arrowhead an exclusive (even as to GSK and its Affiliates), fully-paid, royalty-free, irrevocable, perpetual, sublicensable, worldwide license and right of reference under such Regulatory Filings and Regulatory Approvals (with the right to sublicense and grant further rights of reference) as necessary to Develop, Manufacture and Commercialize such Terminated Product in the Field in the Territory; provided, however, that GSK shall retain such licenses, rights of reference or other rights under such Regulatory Filings and Regulatory Approvals to the extent necessary to enable GSK (or its Affiliates) to perform any obligations or exercise any rights that survive such termination of this Agreement as may be expressly provided in this Agreement, including pursuant to Section 13.9, this Section 13.6 or in any written agreement of the Parties, as applicable. 13.6.6 Remaining Inventory. GSK (and its Affiliates and Sublicensees), with Arrowhead’s consent, which will not be unreasonably withheld, shall have the right to sell or have sold any remaining inventory of Licensed Products following the

84 effective date of termination of this Agreement subject to the applicable payment and reporting obligations under Article VI. 13.6.7 Clinical Studies. Where any Clinical Trial of any Licensed Product is ongoing upon termination, each Party shall continue, at its cost, the Clinical Trial for which it, its Affiliate, (sub)contractor or sublicensee is the regulatory sponsor, solely as deemed necessary by such Party based on reasonable medical judgment to protect the safety, health or welfare of subjects participating in the relevant Clinical Trial, until such point as the study is completed or, if earlier, such Party determines that it is ethical to terminate such study or otherwise cease supporting it. 13.6.8 Orderly Wind-Down. Upon early termination, the Parties shall coordinate in good faith to wind down Development, Manufacturing, and Commercialization activities under this Agreement relating to any Licensed Products ongoing at the effective date of such termination, including the withdrawal of any Licensed Products from the market and a final reconciliation of all payments due under this Agreement. 13.6.9 No Waiver for Termination Due to Breach. For the avoidance of doubt, an aggrieved Party that terminates this Agreement for material breach may also seek damages and other relief for such material breach and (for the avoidance of doubt) for any other breach of this Agreement. 13.7 Return of Confidential Information. Upon expiration or early termination of this Agreement, a Receiving Party shall, at the other Party’s request (and to the extent and when permitted by Applicable Law), destroy, redact, or return, and cause its Affiliates and Third Party subcontractors and sublicensees to destroy, redact, or return all records to the extent containing, and all materials constituting, the other Party’s Confidential Information in its possession and control, and, upon request, provide written certification of such destruction, redaction, or return, except that: (a) the Receiving Party may retain in strict confidence one copy of the other Party’s Confidential Information for the Receiving Party’s legal archival purposes; and (b) the foregoing requirement to destroy, redact, or return the other Party’s Confidential Information shall not apply with respect to any such Confidential Information of the Disclosing Party to the extent that this Agreement or any other written agreement between the Parties (or their respective Affiliates) expressly provides that a Party retains the right to use such Confidential Information (such as by virtue of being a joint owner, or by survival of GSK’ s license rights on a paid-up basis following expiration (without early termination) of this Agreement). 13.8 Certain Additional Remedies of GSK in Lieu of Termination. If GSK has the right to terminate this Agreement pursuant to Section 13.2.2, then in lieu of GSK terminating pursuant to Section 13.2.2, GSK may elect to have this Agreement continue in full force and effect as modified by this Section 13.8 by providing written notice to Arrowhead prior to the date that otherwise would have been the effective date of termination had GSK exercised its right to so terminate this Agreement under Section 13.2.2; provided that, if GSK so elects to continue this Agreement, then from and after such time as GSK delivers such written notice to Arrowhead, [***].

85 13.9 Survival. In the event of expiration or termination of this Agreement for any reason, the provisions of (a) Article I (Definitions) (in each case, solely with respect to defined terms that are used in surviving provisions); (b) Section 2.2 (Licenses Constitute IP under Bankruptcy Code), Section 2.3 (Rights in Combination Products) and Section 2.4 (No Other Rights); (c) Section 3.11 (Records); (d) Section 3.4 (Transfer of Know-How) and Section 5.2 (Technical Transfer) (in each case, solely with respect to any payment obligations that accrued prior to such expiration or termination of this Agreement but have not been paid); (e) Section 3.6.2 (Adverse Event Reporting) (solely with respect to GSK and a serious adverse event that arose prior to such expiration or termination of this Agreement); (f) Section 6.3 (Milestone Payments), Section 6.4 (Royalty Payments), Section 6.5 (Third Party Obligations) and Article VII (General Payment Terms) (in each case, solely with respect to any payment obligations that accrued prior to such expiration or termination of this Agreement but have not been paid); (g) Section 8.2 (Ownership of Agreement IP), Section 8.3.3(a), Section 8.3.3(c), Section 8.3.3(d) and Section 8.3.3(f) (Prosecution of Patent Rights) (in each case, solely with respect to Joint Agreement Patent Rights); (h) Section 8.9 (Common Interest Agreement); (i) Section 9.1 (Confidential Information) (solely for the term specified therein), Section 9.2 (Permitted Use and Disclosures) and Section 9.5 (Equitable Relief); (j) Section 10.5 (No Warranties); (k) Article XI (Indemnification and Insurance); (l) Section 13.3.2 (Early Termination for Bankruptcy), Section 13.6 (Consequences of Early Termination), Section 13.7 (Return of Confidential Information) and this Section 13.9 (Survival); (m) Article XIV (Dispute Resolution); and (n) Article XV (Miscellaneous), in each case, shall survive, as well as any other provisions that, as apparent from their nature and context are intended to continue or to remain (such as for interpretation purposes). Further for the avoidance of doubt, upon expiration or termination of this Agreement for any reason, neither Party shall be released from any obligation that accrued prior to the end of the Term hereof. Accordingly, termination or expiration of this Agreement, in whole or in part (including relinquishment of any license right granted hereunder) for any reason, shall be without prejudice to any obligations that accrued prior to such termination or expiration, including any payments due hereunder (regardless of when payable) and any and all damages arising from any breach. In addition, any payments accrued prior to such termination or expiration shall become payable upon the effective date of such termination or expiration or at such earlier time as otherwise provided hereunder. ARTICLE XIV: DISPUTE RESOLUTION 14.1 Referral to Executive Officers. In the event of a Dispute, except for a Patent Controversy, either Party may refer the matter to the Parties’ Executive Officers for attempted resolution. The Executive Officers, in the presence of their legal advisors, shall attempt in good faith to resolve any Dispute through negotiations. If the Executive Officers are unable to resolve a Dispute referred to them within ten (10) Business Days (or such other period as may be agreed by the Parties in writing) after such referral, and subject to any other provisions of this Agreement, such Dispute shall be resolved as provided below in this Article XIV. 14.2 Mediation. If the Executive Officers are unable to resolve a Dispute referred to them pursuant to Section 14.1 within ten (10) Business Days (or such other period as may be agreed by the Parties in writing) after such referral, the Parties shall first attempt in good

86 faith to resolve any Dispute by confidential mediation in accordance with the then-current Mediation Procedure of the International Institute for Conflict Prevention and Resolution (“CPR Mediation Procedure”) (www.cpradr.org) before initiating arbitration. The CPR Mediation Procedure shall control, except where it conflicts with these provisions, in which case these provisions control. The mediator shall be chosen pursuant to CPR Mediation Procedure. The mediation shall be held in New York, New York. Either Party may initiate mediation by written notice to the other Party. The Parties agree to select a mediator within twenty (20) days of the notice and the mediation will begin promptly after the selection. The mediation will continue until the mediator, or either Party, declares in writing, no sooner than after the conclusion of one full day of a substantive mediation conference attended on behalf of each Party by a senior business person with authority to resolve the Dispute, that the Dispute cannot be resolved by mediation. In no event, however, shall mediation continue more than sixty (60) days from the initial notice by a Party to initiate meditation unless the Parties agree in writing to extend that period. Any period of limitations that would otherwise expire between the initiation of mediation and its conclusion shall be extended until twenty (20) days after the conclusion of the mediation. No discussions between the Parties attempting to resolve a Dispute under Section 14.1 or this Section 14.2 shall be admissible in arbitration of the Dispute. 14.3 Arbitration. If the Parties fail to reach resolution pursuant to mediation in accordance with Section 14.2, and a Party desires to pursue resolution of a Dispute, then the Dispute shall be submitted by either Party for resolution in arbitration pursuant to the then current CPR Non-Administered Arbitration Rules (“CPR Rules”) (www.cpradr.org), except where they conflict with these provisions, in which case these provisions control. 14.3.1 The arbitration will be held in New York, New York. All aspects of the arbitration shall be treated as confidential. 14.3.2 The arbitrators will be chosen from the CPR Panel of Distinguished Neutrals, unless a candidate not on such panel is approved by both Parties. Each arbitrator shall be a lawyer with at least fifteen (15) years’ experience with a law firm or corporate law department of over twenty-five (25) lawyers or who was a judge of a court of general jurisdiction. To the extent that the Dispute requires special expertise, the Parties will so inform CPR prior to the beginning of the selection process. 14.3.3 The arbitration tribunal shall consist of three arbitrators, of whom each Party shall designate one in accordance with the “screened” appointment procedure provided in CPR Rule 5.4. The chair will be chosen in accordance with CPR Rule 6.4. If, however, the aggregate award sought by the Parties is less than Five Million Dollars ($5,000,000) and equitable relief is not sought, a single arbitrator shall be chosen in accordance with the CPR Rules. 14.3.4 Candidates for the arbitrator position(s) may be interviewed by representatives of the Parties in advance of their selection, provided that all Parties are represented. 14.3.5 The Parties agree to select the arbitrator(s) within forty-five (45) days of initiation of the arbitration. The hearing will be concluded within nine (9) months after

87 selection of the arbitrator(s) and the award will be rendered within sixty (60) days of the conclusion of the hearing, or of any post hearing briefing, which briefing will be completed by both sides within forty-five (45) days after the conclusion of the hearing. In the event the Parties cannot agree upon a schedule, then the arbitrator(s) shall set the schedule following the time limits set forth above as closely as practical. 14.3.6 The Parties shall have the right to conduct and enforce pre-hearing discovery in accordance with the then current Federal Rules of Civil Procedure, unless otherwise agreed by the Parties in writing. All discovery conducted pursuant to the arbitration proceedings will be subject to the then current Federal Rules of Civil Procedure, unless otherwise agreed by the Parties in writing. 14.3.7 The hearing will be concluded in ten (10) hearing days or less. Multiple hearing days will be scheduled consecutively to the greatest extent possible. A transcript of the testimony adduced at the hearing shall be made and shall be made available to each Party. 14.3.8 The arbitrator(s) shall decide the merits of any Dispute in accordance with the law governing this Agreement, without application of any principle of conflict of laws that would result in reference to a different law. The arbitrator(s) may not apply principles such as “amiable compositeur” or “natural justice and equity.” 14.3.9 The arbitrator(s) are expressly empowered to decide dispositive motions in advance of any hearing and shall endeavor to decide such motions as would a United States District Court Judge sitting in the jurisdiction whose substantive law governs. 14.3.10 The arbitrator(s) shall render a written opinion stating the reasons upon which the award is based. The Parties consent to the jurisdiction of the United States District Court for the district in which the arbitration is held for the enforcement of these provisions and the entry of judgment on any award rendered hereunder. Should such court for any reason lack jurisdiction, any court with jurisdiction may act in the same fashion. 14.3.11 Each Party has the right to seek from the appropriate court provisional remedies such as attachment, preliminary injunction, replevin, etc. to avoid irreparable harm, maintain the status quo, or preserve the subject matter of the Dispute. Rule 14 of the CPR Rules does not apply to this Agreement. 14.4 Waiver of Jury Trial. EACH PARTY HERETO WAIVES: ITS RIGHT TO TRIAL BY JURY OF ANY ISSUE UNDERLYING A DISPUTE WITHIN THE SCOPE OF SECTION 14.2 OR SECTION 14.3. 14.5 Interim or Provisional Relief. Nothing in this Agreement, including Section 11.5, shall preclude either Party from seeking interim or provisional relief in any court of competent jurisdiction, including a temporary restraining order, preliminary injunction or other interim equitable relief concerning a Dispute with the other Party, either prior to or during

88 the Dispute resolution procedures set forth in this Article XIV to protect the interests of such Party. 14.6 Consent to Jurisdiction. Each Party, for the purpose of enforcing an award under Section 14.3 or for seeking interim or provisional relief as contemplated in Section 14.5 with respect to any disputed breach of this Agreement, agrees not to raise any objection at any time to the laying or maintaining of the venue of any action, suit or proceeding for such purpose in any state or federal Court sitting in New York, irrevocably waives any claim that such action, suit or other proceeding has been brought in an inconvenient forum, and further irrevocably waives the right to object, with respect to such action, suit or other proceeding, that such Court does not have any jurisdiction over such Party. Each Party further agrees that service of any process, summons, notice or document by registered mail to such Party’s notice address provided for in this Agreement shall be effective service of process for any action, suit or proceeding in the Court with respect to any matters to which it has submitted to jurisdiction in this Section 14.6. 14.7 No Claims against Employees. Each Party undertakes to make no claim and bring no proceedings in connection with this Agreement or its subject matter against any director, officer, employee or agent of the other Party (apart from claims based on fraud or willful misconduct). This undertaking is intended to give protection to individuals: it does not prejudice any right which a Party might have to claim against another Party. 14.8 Intellectual Property Disputes. Notwithstanding any provision to the contrary set forth in this Agreement, if a Patent Controversy arises under this Agreement, then, such Patent Controversy will be submitted to a court of competent jurisdiction in the jurisdiction in which the applicable Patent Rights, Know-How or other intellectual property rights that are the subject of such Patent Controversy was granted or arose and shall not be subject to mediation under Section 14.2 or arbitration under Section 14.3. ARTICLE XV: MISCELLANEOUS 15.1 Assignment; Successors. 15.1.1 Except as expressly provided herein, neither this Agreement nor any right or obligation hereunder shall be assignable or transferable, whether voluntarily or by operation of law, without the prior written consent of the other Party (not to be unreasonably withheld or delayed). Each Party may assign or transfer this Agreement or any of its rights and obligations hereunder to any Affiliate, or to any Third Party that acquires all or substantially all of such Party’s assets or business relating to the Licensed Constructs and Licensed Product to which this Agreement relates (whether by sale of assets or stock, merger, consolidation, reorganization or otherwise), without the consent of the other Party. Each assigning Party shall give written notice to the other Party promptly following any such assignment or transfer. No assignment under this Section 15.1 shall relieve the assigning Party of any of its responsibilities or obligations hereunder and, as a condition of such assignment, the assignee shall agree in writing to be bound by all obligations of the assigning Party hereunder. This Agreement shall be binding upon the successors

89 and permitted assigns of the Parties. Any assignment or other transfer not in accordance with this Section 15.1 shall be null and void. 15.1.2 Notwithstanding anything to the contrary in Section 15.1.1 or elsewhere in this Agreement, but subject to the terms of this Section 15.1.2, at the sole cost and expense of Arrowhead, Arrowhead may assign to a Third Party its right to receive any or all of the milestone payments under Section 6.3.1 and Section 6.3.2 and any or all of the royalty payments under Section 6.4.2 and Section 6.4.3 (each such assignment, a “Securitization Transaction”); provided that (a) promptly following the closing of any such Securitization Transaction (but, in any event, at least forty (40) Business Days prior to the delivery of the first invoice for the first applicable payment to be made to such Third Party as the assignee of such payment rights), Arrowhead shall provide notice of such Securitization Transaction to GSK, which notice shall identify the applicable Third Party that is entitled to receive any such payments hereunder, including the tax domicile of such Third Party; and (b) upon GSK’s request, Arrowhead shall require such Third Party to provide to GSK (i) any information that would be required to be provided by Arrowhead with respect to such payments pursuant to Section 7.4 (mutatis mutandis) and (ii) any other information reasonably requested by GSK in order to process any such payments to be made to such Third Party. In connection with a contemplated Securitization Transaction and after the closing of any such Securitization Transaction with a Third Party, Arrowhead may disclose to such Third Party the royalty reports contemplated under Section 7.2, without the prior written consent of GSK, notwithstanding them containing GSK’s Confidential Information, to the extent reasonably necessary to enable such Third Party to evaluate the Securitization Transaction opportunity (provided that such Third Party is under obligations of confidentiality and non-use with respect to Confidential Information included in such reports and plans that are no less stringent than the terms of Article IX (but of duration customary in confidentiality agreements entered into for a similar purpose)), and to enable such Third Party to exercise its rights with respect to such Securitization Transaction, as applicable. As part of any consummated Securitization Transaction, Arrowhead may assign, without the prior written consent of GSK, its right to receive the royalty reports and to conduct audits under, respectively, Section 7.2, Section 7.5.2 and Section 7.5.3 to the Third Party in such Securitization Transaction, and to allow such Third Party to exercise its rights thereunder, in each case, in accordance with and subject to the applicable terms and conditions of this Agreement. 15.2 Waiver. The failure of any Party to assert a right hereunder or to insist upon compliance with any term or condition of this Agreement shall not constitute a waiver of that right or excuse a similar subsequent failure to perform any such term or condition by the other Party. No waiver shall be effective unless it has been given in writing and signed by the Party giving such waiver. The exercise of any right hereunder by a Party in the event of the other’s default does not constitute an election of remedies or prevent the exercise of any or all other rights (all rights and remedies being cumulative).

90 15.3 Choice of Law. This Agreement, its interpretation, construction and performance and the rights granted and obligations arising hereunder, shall be governed by, and construed in accordance with, the laws of the State of New York of the United States of America, including its statutes of limitations but exclusive of its conflicts of law rules. In the event of any conflict between U.S. and foreign Applicable Laws, U.S. Applicable Laws shall govern. The United Nations Convention on Contracts for the International Sale of Goods shall not apply to this Agreement. 15.4 Notices. All notices given under this Agreement by either Party to the other Party shall be in the English language, in writing (which shall exclude e-mail), and shall refer specifically to this Agreement and shall be delivered personally, sent by nationally-recognized overnight courier, or sent by registered or certified mail, postage prepaid, return receipt requested, to the following respective addresses (or to such other address as may be specified by notice from time to time by the relevant Party). If to Arrowhead: Arrowhead Pharmaceuticals, Inc. 177 East Colorado Boulevard, Suite 700 Pasadena, CA 91105 Attention: General Counsel With a copy (which shall not constitute notice) to: Gibson Dunn & Crutcher 555 Mission Street, Suite 3000 San Francisco, CA 94105 Attention: Ryan Murr If to GSK: GlaxoSmithKline Intellectual Property (No. 3) Limited GSK Medicines Research Centre Gunnels Wood Road Stevenage, SG1 2NY United Kingdom Attn.: Senior Vice President Business Development With a copy (which shall not constitute notice) to: GlaxoSmithKline 980 Great West Road Brentford, Middlesex TW8 9GS United Kingdom Attn: Vice President & Head of Legal Business Development & Corporate 15.4.1 Without prejudice to any earlier time at which a notice may be actually given and received, a properly addressed notice shall in any event be deemed to have been received: (a) when delivered, if personally delivered during the recipient’s normal business hours; (b) on the Business Day after dispatch, if sent by nationally- recognized overnight courier and proof of delivery is obtained; and (c) on the third (3rd) Business Day following the date of mailing, if sent by mail.

91 15.4.2 Where proceedings have been commenced in any arbitration hereunder or court of competent jurisdiction, any documents issued in the course of those proceedings will be served in accordance with the procedural rules governing the service of documents in those proceedings. 15.4.3 This Section 15.4 shall apply to notices required to be given by one Party to the other under this Agreement. Other communications between the Parties that are routine in nature, such as communications between Alliance Managers regarding their ongoing activities performed in the ordinary course of their work under this Agreement, may be made via e-mail. The official language of this Agreement and between the Parties for all notices and communications between the Parties hereunder shall be in the English language. 15.5 Severability. If the whole or any provision of this Agreement is held to be invalid, illegal or unenforceable in any jurisdiction for any reason, then, to the fullest extent permitted by Applicable Law, (a) in the case of the illegality, invalidity or unenforceability of the whole of this Agreement, it shall terminate in relation to the jurisdiction in question; and (b) in the case of illegality, invalidity or unenforceability of any provision of this Agreement, that part shall be severed from this Agreement in the jurisdiction in question (but shall remain in full force and effect in all other jurisdictions) and (i) all other provisions hereof shall remain in full force and effect in the relevant jurisdiction and shall be liberally construed in order to carry out the intent of the Parties as nearly as may be possible, and (ii) the Parties agree to use reasonable efforts to negotiate a provision, in replacement of the provision held invalid, illegal or unenforceable, that is consistent with Applicable Law in the relevant jurisdiction and accomplishes, as nearly as possible, the original intention of the Parties with respect thereto. 15.6 Integration; Amendments. The Parties hereby agree and acknowledge that (a) as of the Amended Effective Date, this Agreement amends and restates the Original Agreement in its entirety and the Original Agreement is replaced with, and superseded by, this Agreement and, from and after the Amended Effective Date, the rights and obligations of the Parties shall be governed by the terms and conditions of this Agreement; and (b) prior to the Amended Effective Date, the rights and obligations of Arrowhead and Janssen (and the rights of GSK as Janssen’s permitted assignee) shall be governed under the terms and conditions of the Original Agreement, except as otherwise expressly set forth herein (including as set forth in Section 8.2.2). Subject to the foregoing, this Agreement, together with the Schedules attached hereto, the Original Agreement (together with the Exhibits attached thereto) and the Pharmacovigilance Agreement (if any), constitutes the entire agreement, and supersedes all previous agreements (whether written or oral), between the Parties with respect to the subject matter of this Agreement, including the Existing Confidentiality Agreement. Except as specified herein, no waiver, modification or amendment of any provision of this Agreement shall be valid or effective unless made in a writing referencing this Agreement and signed by a duly authorized officer of each Party. In the event of a conflict between any terms of any Schedule or other appendix to this Agreement and the body of this Agreement, the body of this Agreement shall control.

92 15.7 Binding Effect; Independent Contractors; No Agency. As of the Amended Effective Date, this Agreement will be binding upon and inure to the benefit of the Parties and their respective permitted successors and permitted assigns. Except as expressly set forth in this Agreement, no Person other than the Parties and their respective Affiliates and permitted assignees hereunder will be deemed an intended beneficiary hereunder or have any right to enforce any obligation of this Agreement. Neither Party shall have any responsibility for the hiring, firing or compensating the other Party’s employees or agents for any employee benefits. No employee or representative of a Party shall have any authority to bind or obligate the other Party to this Agreement to pay any sum or in any manner whatsoever, or to create or impose any contractual or other liability on the other Party. Nothing in this Agreement is intended or shall be deemed, for financial, tax, legal or other purposes, to constitute a partnership, agency, joint venture or employer-employee relationship between the Parties. 15.8 Performance by Affiliates. Except as expressly prohibited hereunder, either Party may use one or more of its Affiliates to perform its obligations and duties hereunder, provided that such Party shall remain liable hereunder for the timely payment and performance of all of its obligations and duties hereunder. 15.9 Force Majeure. No Party shall be deemed to have defaulted under or breached this Agreement for failure or delay in fulfilling or performing any term of this Agreement, except for the payment of any amounts under this Agreement, when such failure or delay is caused by or results from causes beyond the reasonable control of the non-performing Party, including fires, floods, embargoes, shortages, epidemics, quarantines, war, acts of war (whether war be declared or not), terrorism, insurrections, riots, civil commotion, acts of God or acts, omissions or delays in acting by any Governmental Authority. The non- performing Party shall notify the other Party of such force majeure within five (5) Business Days after such occurrence by giving written notice to the other Party stating the nature of the event, its anticipated duration, and any action being taken to avoid or minimize its effect. The suspension of performance shall be of no greater scope and no longer duration than is necessary and the non-performing Party shall use, throughout the period of suspension of performance, Commercially Reasonable Efforts to remedy its inability to perform; provided, however, that in the event the suspension of performance continues for ninety (90) days after the date such force majeure commences, the Parties shall meet to discuss in good faith how to proceed in order to accomplish the objectives of this Agreement; and provided, further, however, that if the suspension of performance continues for more than one (1) year after the date such force majeure commences, either (a) GSK in the event that Arrowhead is the nonperforming Party, or (b) Arrowhead in the event that GSK is the non-performing Party, shall have the right to terminate this Agreement upon notice to non-performing Party. For purposes of this Agreement a force majeure shall not include a failure to commit sufficient resources, financial or otherwise, to the activities to be conducted pursuant to this Agreement or general market or economic conditions. 15.10 Construction. The headings used herein are for reference and convenience only, and will not enter into the interpretation of this Agreement. Except as otherwise explicitly specified to the contrary, (a) references to a Section, Article or Schedule means a Section or Article

93 of, or Schedule to, this Agreement and all subsections thereof, unless another agreement is specified; (b) references to a particular statute or regulation include all rules and regulations thereunder and any successor statute, rules or regulations then in effect, in each case, including any then-current amendments thereto; (c) words in the singular or plural form include the plural and singular form, respectively; (d) capitalized terms not expressly defined herein that are corollaries (such as pluralizations and changes in tense) to capitalized terms defined herein shall have the corresponding meanings (e) unless the context requires a different interpretation, the word “or” has the inclusive meaning that is typically associated with the phrase “and/or”; (f) terms “including,” “include(s),” “such as,” and “for example” as used in this Agreement mean including the generality of any description preceding such term and will be deemed to be followed by “without limitation”; (g) whenever this Agreement refers to a number of days, such number will refer to calendar days unless Business Days are specified; (h) “herein”, “hereunder”, “hereof”, and the like shall be understood to refer to this Agreement in its entirety, and not the particular provision or Section in which they appear; (i) references to a particular Person include such Person’s successors and assigns, to the extent not prohibited by this Agreement; (j) all words used in this Agreement will be construed to be of such gender or number as the circumstances require; (k) references to “persons” includes individuals, bodies corporate (wherever incorporated), unincorporated associations and partnerships; (1) the words “comprise”, “comprising”, “contain”, “containing”, “include” and “including” are used in their open, non-limiting form, and shall be understood to include the words “without limitation” even if not expressly stated; (m) all references to “dollars”, “$” or “USD” shall mean United States dollars; (n) the word “will” will be construed to have the same meaning and effect as the word “shall”; (o) any definition of or reference to any agreement, instrument or other document herein will be construed as referring to such agreement, instrument or other document as from time to time amended, supplemented or otherwise modified (subject to any restrictions on such amendments, supplements or modifications set forth herein); (p) the word “notice” will mean notice in writing (whether or not specifically stated), shall include any written instrument or communication delivered in accordance with Section 15.4, unless otherwise specified herein; (q) provisions that require that a Party or the Parties “agree,” “consent” or “approve” or words of similar import will require that such agreement, consent or approval be specific and in writing, whether by written agreement, letter, approved minutes or otherwise; and (r) any reference to a “sublicensee” of GSK under this Agreement shall be construed to include Sublicensees. The official language of this Agreement and between the Parties for all correspondence shall be the English language. Each Party represents that it has been represented by legal counsel in connection with this Agreement and acknowledges that it has participated in the drafting hereof. In interpreting and applying the terms and provisions of this Agreement, the Parties agree that no presumption will apply against the Party which drafted such terms and provisions. Unless the context otherwise requires, countries shall include territories. 15.11 Further Actions. Each Party agrees to execute, acknowledge and deliver such further instruments and to do all such other acts as may be necessary or appropriate in order to carry out the purposes and intent of this Agreement. 15.12 Expenses. Except as otherwise provided herein, all fees, costs and expenses (including any legal, accounting and banking fees) incurred in connection with the preparation,

94 negotiation, execution and delivery of this Agreement and to consummate the transactions contemplated hereby will be paid by the Party hereto incurring such fees, costs and expenses. 15.13 Execution in Counterparts; Facsimile Signatures. This Agreement may be executed in counterparts, each of which counterparts, when so executed and delivered, shall be deemed to be an original, and all of which counterparts, taken together, shall constitute one and the same instrument even if both Parties have not executed the same counterpart. Facsimile or portable document format (i.e., .pdf), execution and delivery of this Agreement by a Party constitutes a legal, valid and binding execution and delivery of this Agreement by such Party. [Remainder of this page intentionally blank]

Signature Page to Amended and Restated License Agreement IN WITNESS WHEREOF, each Party has caused this Agreement to be duly executed by its authorized representative on the respective date written herein below. Arrowhead Pharmaceuticals, Inc. By: /s/ Christopher Anzalone Name: Christopher Anzalone, Ph.D. Title: President and CEO GlaxoSmithKline Intellectual Property (No. 3) Limited By: /s/ Jill Anderson Name: Jill Anderson Title: Director

96