Up to 1,526,717 Units (each Unit
contains One Share of Common Stock and One Common Warrant to purchase One Share of Common Stock)
Up to 1,526,717 Pre-funded Units
(each Pre-funded Unit contains One Pre-funded Warrant to Purchase One Share of Common Stock and One Common Warrant to purchase
One Share of Common Stock)
Shares of Common Stock Underlying the Pre-funded Warrants and
Shares of Common Stock Underlying the Common Warrants
We are offering up to 1,526,717 units (each unit consisting
of one share of our common stock and one common warrant to purchase one share of our common stock) pursuant to this prospectus.
Each common warrant contained in a unit has an exercise price of $ per share. The common warrants contained
in the units will be exercisable immediately and will expire five years from the date of issuance. We are also offering the shares
of our common stock that are issuable from time to time upon exercise of the common warrants contained in the units.
We are also offering to each purchaser whose purchase
of units in this offering would otherwise result in the purchaser, together with its affiliates and certain related parties, beneficially
owning more than 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock immediately following the
consummation of this offering, the opportunity to purchase, if the purchaser so chooses, pre-funded units (each pre-funded unit
consisting of one pre-funded warrant to purchase one share of our common stock and one common warrant to purchase one share of
our common stock) in lieu of units that would otherwise result in the purchaser’s beneficial ownership exceeding 4.99% (or,
at the election of the purchaser, 9.99%) of our outstanding common stock. Because we will issue a common warrant as part of each
unit or pre-funded unit, the number of common warrants sold in this offering will not change as a result of a change in the mix
of the units and pre-funded units sold. Each pre-funded warrant contained in a pre-funded unit will be exercisable for one share
of our common stock. The purchase price of each pre-funded unit will equal the price per unit being sold to the public in this
offering, minus $0.01, and the exercise price of each pre-funded warrant included in the pre-funded unit will be $0.01 per share.
The pre-funded warrants will be immediately exercisable and may be exercised at any time until all of the pre-funded warrants
are exercised in full. This offering also relates to the shares of common stock issuable upon exercise of any pre-funded warrants
contained in the pre-funded units sold in this offering. Each common warrant contained in a pre-funded unit has an exercise price
of $ per share. The common warrants contained in the pre-funded units will be exercisable immediately and will expire five years
from the date of issuance. We are also offering the shares of our common stock that are issuable from time to time upon exercise
of the common warrants contained in the pre-funded units.
For each pre-funded unit we sell, the number of units
we are offering will be decreased on a one-for-one basis. The units and pre-funded units will not be issued or certificated. The
shares of common stock or pre-funded warrants, as the case may be, and the common warrants included in the units or pre-funded
units, can only be purchased together in this offering, but the securities contained in the units or the pre-funded units will
be issued separately and will be immediately separable upon issuance. The final breakdown between units and pre-funded units will
be determined upon the establishment of the final public offering price and immediately prior to the execution of the underwriting
agreement.
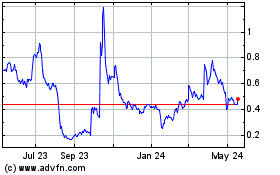
Our common stock is listed on the Nasdaq Capital Market
under the symbol “OPGN.” On October 9, 2019, the last reported sale price of our common stock on the Nasdaq Capital
Market was $6.55 per share. The public offering price per unit or pre-funded unit, as the case may be, will be determined between
us and the underwriter based on market conditions at the time of pricing, and may be at a discount to the current market price
of our common stock. Therefore, the recent market price used throughout this prospectus may not be indicative of the final offering
price.
There is no established public trading market for the
pre-funded warrants or the common warrants, and we do not expect a market to develop. In addition, we do not intend to apply for
listing of the pre-funded warrants or the common warrants on any securities exchange or other nationally recognized trading market.
Without an active trading market, the liquidity of the pre-funded warrants and the common warrants will be limited.
Following receipt of approval from stockholders at an
annual meeting of stockholders held on August 22, 2019, on August 28, 2019, we filed an amendment to our Amended and Restated
Certificate of Incorporation to effect a reverse stock split of the issued and outstanding shares of our common stock, at a ratio
of one share for twenty shares. All unit, share, per unit price and per share price in this prospectus have been adjusted to reflect
the reverse stock split.
We are an “emerging growth company” as the term
is used in the Jumpstart Our Business Startups Act of 2012 and, as such, we have elected to comply with certain reduced public
company reporting requirements for this prospectus and future filings. See “Prospectus Summary - Implications of Being an
Emerging Growth Company.”
Investing in our securities involves a high degree
of risk. See “Risk Factors” beginning on page 34.
Neither the Securities and Exchange Commission nor any
state securities commission has approved or disapproved of these securities or passed upon the accuracy or adequacy of this prospectus.
Any representation to the contrary is a criminal offense.
The offering is being underwritten on a firm commitment basis.
The underwriter has an option exercisable within 30 days from the date of this prospectus to purchase up to 229,007 additional
shares of common stock and/or common warrants to purchase up to an additional 229,007 shares of common stock from us at the public
offering price, less the underwriting discounts and commissions. If the underwriter exercises this option in full, the total underwriting
discounts and commissions payable by us will be $ , and the total proceeds to us, before expenses, will be $ , excluding potential
proceeds from the exercise of the common warrants included in such option.
We expect to deliver the securities to purchasers on or about
, 2019.
OpGen’s
Business
We are a precision medicine
company harnessing the power of molecular diagnostics and informatics to help combat infectious disease. We are developing molecular
information products and services for global healthcare settings, helping to guide clinicians with more rapid and actionable information
about life threatening infections, improve patient outcomes, and decrease the spread of infections caused by MDROs. Our proprietary
DNA tests and informatics address the rising threat of antibiotic resistance by helping physicians and other healthcare providers
optimize care decisions for patients with acute infections.
OpGen is developing high resolution
Acuitas AMR Gene Panel tests designed to determine pathogen levels in clinical specimens and the key drug resistance gene profiles
of Gram-negative organisms. Currently, the Acuitas AMR Gene Panel tests are available for sale for RUO. Following completion of
research and development efforts, and if OpGen is able to obtain the appropriate regulatory clearances, OpGen anticipates its
Acuitas AMR Gene Panel tests will be used in the clinical setting to provide pathogen and antibiotic resistance gene information
to aid in decision-making for patients at risk for cUTI, lower respiratory tract infections, blood stream infections, and for
testing of bacterial isolates. OpGen currently offers its Acuitas AMR Gene Panel (RUO) tests to CROs, pharmaceutical companies,
hospitals and other healthcare providers for RUO. OpGen offers its Acuitas Lighthouse Software to health care facilities and public
health facilities for research purposes, primarily in relation to infection control and surveillance.
Acuitas
AMR Gene Panel and Acuitas Lighthouse Software
The Acuitas AMR Gene Panel
is a development-stage, qualitative and semi-quantitative nucleic acid-based in vitro diagnostic test that is designed for simultaneous
detection and identification of multiple bacterial nucleic acids and select genetic determinants of antimicrobial resistance in
urine specimens or bacterial colonies isolated from urine and other body sites. The Acuitas AMR Gene Panel (Urine) is intended
as an aid in the diagnosis of specific agents of cUTIs for patients at risk of cUTI. The Acuitas AMR Gene Panel (Urine) employs
automated deoxyribonucleic acid, or DNA, extraction on the Qiagen® EZ1 Advanced XL and multiplex real-time PCR on the Applied
Biosystems™ QuantStudio 5 PCR System. The Acuitas AMR Gene Panel (Urine) test detects up to 47 gene targets which span 600
subtypes and convey resistance to nine classes of antibiotics directly from urine and isolated colonies, and is currently sold
as a RUO test. Gene families detected include: KPC, NDM, VIM, IMP, OXA, CTXM-1, CTXM-9, CMY, MCR, and resistance genes to fluoroquinolone
antibiotics. From urine specimens, the Acuitas AMR Gene Panel (Urine) will semi-quantitatively detect the most common bacterial
causes of cUTI (E. coli, K. pneumoniae, P. aeruginosa, P. mirabilis, E. faecalis). The Acuitas AMR Gene Panel (Urine) is
designed to provide test results in under three hours, compared with traditional microbiology methods, which can take two to three
days.
OpGen is also developing the
Acuitas AMR Gene Panel (Isolates) test for testing bacterial isolates. This test is currently available in the United States for
RUO and is being used in such capacity in connection with The New York State Infectious Disease Digital Health Initiative for
testing of bacterial isolates. The test is contributing to the initiative’s research mission by genotyping carbapenem resistant
isolates from three health systems in the New York City Metro Area. Results are subsequently analyzed by the Acuitas Lighthouse
Software (RUO) to support a series of infection control tracking capabilities that are of interest to The New York State Department
of Health and healthcare providers. On May 13, 2019, OpGen filed a 510(k) submission with the FDA for clearance for its Acuitas
AMR Gene Panel test for the detection of antimicrobial resistance genes in bacterial isolates. The FDA responded to our submission
with an AI Request in July 2018, to which we have 180 days to submit a complete response. In the meantime, our 510(k) submission
is on hold. If we are able to obtain FDA clearance of the Acuitas AMR Gene Panel (Isolates) test, we expect use of the test to
expand from current research uses to include the clinical diagnostic use of test information to support antibiotic decision making
in acute care patient management of patients with MDRO infections.
The Acuitas Lighthouse Software
(RUO) manages and evaluates data that identify the most common microbial causes of cUTI and key genetic determinants of antibiotic
drug resistance, based on the amplification data of gene targets extracted from urine specimens. Through analysis of this data,
the Acuitas Lighthouse Software can identify five bacterial species and predict resistance to up to fourteen different antibiotics
from across nine antibiotic classes including: Aminoglycosides, Carbapenems, Cephalosporins, Fluoroquinolones, Polymyxins, Penicillins,
Sulfonamides, Trimethoprim and Vancomycin. The Acuitas Lighthouse Software consists of the Acuitas Lighthouse Portal, a web application;
the Acuitas Lighthouse Prediction Engine, data analysis software; and draws from the Lighthouse Knowledgebase, a relational database
management system; and minor supporting software components. The Acuitas Lighthouse Software (RUO) was selected by The New York
State Department of Health Wadsworth Center for the genomic microbiology component of The New York State Infectious Disease Digital
Health Initiative. All components of the Acuitas Lighthouse Software are hosted in a cloud-based web application that is protected
by security measures. The input to Acuitas Lighthouse Software is a data file generated by processing the results from the Acuitas
AMR Gene Panel (Urine) test through the Acuitas AMR Gene Panel (Urine) Gene Analysis Software. This input file indicates which
gene targets were detected by the assay and is loaded into the Acuitas Lighthouse Software via an interface of the Acuitas Lighthouse
Portal, accessed by the user through a web browser. The Acuitas AMR Gene Panel (Urine) Gene Analysis Software results are retained
by the Acuitas Lighthouse Knowledgebase and are sent to the Acuitas Lighthouse Prediction Engine for analysis. The Acuitas Lighthouse
Prediction Engine contains software implementations of data models that were derived using a training panel of thousands of bacterial
isolates with detailed genotypic and phenotypic characterizations, all stored within the Acuitas Lighthouse Knowledgebase. These
models, each specific to one microbial species and antibiotic drug pairing, are used to make predictions of antibiotic resistance
by analyzing the loaded input data. The results from the Acuitas Lighthouse Prediction Engine indicate whether there is evidence
of resistance detected through the presence of specific genes, and if there is known intrinsic resistance to certain drugs. These
final results are reported to the user via a Prediction Report and the Resistance Dashboard interface in the Acuitas Lighthouse
Portal; both displays present the Acuitas Lighthouse Prediction Engine output in combination with selected input data and metadata,
as well as the semi-quantitative counts of gene copies / mL for urine specimens. Our development of the Acuitas Lighthouse Software
and the Acuitas AMR Gene Panel (Urine) test, thus far, has resulted from a comprehensive, multi-year effort, which remains ongoing,
to help address urgent clinical needs for improved rapid antibiotic decision-making capabilities.
The figure below describes
the workflow for the Acuitas AMR Gene Panel (Urine) test and the Acuitas Lighthouse Software.

In October 2018, OpGen entered
into a supply agreement with QIAGEN GmbH, or QIAGEN, to advance OpGen’s rapid diagnostics for antimicrobial resistance.
Under the agreement, OpGen will work to commercialize QIAGEN’s EZ1 Advanced XL automated nucleic acid purification instrumentation
(EZ1) and reagent kits in the United States to be used with the Acuitas AMR Gene Panel products for research purposes. Under the
terms of the agreement, OpGen will purchase EZ1 instruments and reagent kits from QIAGEN and sell or place them with customers
in the United States for use with the Acuitas AMR Gene Panel products for RUO and, if the necessary 510(k) clearances are obtained,
as diagnostic products. The EZ1 is a Class II Medical Device listed with the FDA that provides full automation with sample preparation
throughput of up to 14 samples per one-hour run. QIAGEN is the global leader for nucleic acid sample preparation with a full line
of instruments and reagents. There are thousands of EZ1 instruments currently used in laboratories worldwide.
In September 2018, OpGen announced
a collaboration with The New York State DOH and ILÚM to develop a state-of-the-art research program to detect, track, and
manage antimicrobial-resistant infections at healthcare institutions in New York State. The collaboration is called The New York
State Infectious Disease Digital Health Initiative. The first stage of the collaboration is the completion of a demonstration
project, which commenced in February 2019 and is expected to last until March 2020. We believe a successful demonstration project
will lead to a statewide program. Under the demonstration project, OpGen will work with DOH’s Wadsworth Center and ILÚM
to develop an infectious disease digital health and precision medicine platform that connects healthcare institutions to DOH and
uses genomic microbiology for statewide surveillance and control of antimicrobial resistance. The DOH, ILÚM and OpGen will
work collaboratively to build a sustainable, flexible infectious diseases reporting, tracking and surveillance tool for antimicrobial
resistance that can be applied across New York State. The goal of this research project is to improve patient outcomes and save
healthcare dollars by integrating real-time epidemiologic surveillance with rapid delivery of resistance results to care-givers
via web-based and mobile platforms. ILÚM is leading the project with the implementation of its technology platform. OpGen
is providing its Acuitas AMR Gene Panel (RUO) for rapid detection of multidrug-resistant bacterial pathogens along with its Acuitas
Lighthouse Software (RUO) for high resolution pathogen tracking. Under the agreement, OpGen will receive approximately $1.6 million
for the 12-month demonstration portion of the project, with the potential for full implementation during the next four years,
should certain milestones be achieved.
In June 2017, OpGen
entered into a supply agreement to use Thermo Fisher Scientific’s technology in the United States and Europe to support
the commercialization of its rapid molecular products for RUO. Under the terms of the agreement, OpGen provides customer
access to Thermo Fisher Scientific’s products to support the commercialization of our Acuitas QuickFISH Rapid Test and
Acuitas Lighthouse Software to combat MDROs. In January 2018, the Company entered into a second supply agreement to
incorporate Thermo Fisher Scientific’s real-time PCR technology in the Company’s Acuitas AMR Gene Panel tests.
Specific products covered under these agreements include the QuantStudio 5 Real-Time PCR System, TaqMan® Fast Advanced
Master Mix and TaqMan® MGB Probes for quick, multiplexed gene detection.
OpGen’s relationship
with Merck & Co., Inc. includes investment from Merck Global Health Innovation Fund, or MGHIF, and a research agreement with
Merck Sharp & Dohme, or MSD, to provide access to MSD’s 250,000 clinical isolate SMART bacterial surveillance archive.
In December 2017, we entered into a subcontractor agreement with ILÚM, whereby ILÚM provided services to the Company
in the performance of the Company’s CDC contract to deploy ILÚM’s commercially-available cloud- and mobile-based
software platform for infectious disease management in three medical sites in Colombia with the aim of improving antibiotic use
in resource-limited settings.
OpGen’s FDA cleared
and CE marked QuickFISH and PNA FISH products are powered by PNA technology and provide rapid pathogen identification, typically
in less than 30 minutes from a positive blood culture result.
Intellectual Property
As of December 31, 2018, we had total ownership rights
to 22 U.S. patents and applications, including six pending U.S. non-provisional patent applications, and 16 issued U.S. patents.
More specifically, as of December 31, 2018, related to our FISH products, we had ownership rights to 11 U.S. patents and patent
applications, including three pending U.S. non-provisional patent applications, and eight issued U.S. patents. These issued patents
begin to expire in November 2024 and will be fully expired by March 2032. As of December 31, 2018, related to our Acuitas products,
we had ownership rights to three pending U.S. non-provisional patent applications and no issued U.S. patents. As of December 31,
2018, related to our other products, we had ownership rights to eight issued U.S. patents. These issued patents begin to expire
in June 2026 and will be fully expired by January 2032. A majority of our issued and exclusively licensed FISH patents from Dako
Denmark A/S expired over the last six years. The remaining 17 exclusively licensed U.S. FISH patents expire between 2019 and 2024.
Curetis’
Business
Current
Diagnostic Tests and Informatics
Curetis’
automated sample-to-answer product offerings are based on the Unyvero A50 platform that has been CE-IVD-marked since 2012 and
was FDA cleared through the de novo process for the FDA cleared Unyvero LRT test. The Unyvero LRT test is designed to facilitate
diagnoses in connection with the 500,000 to 750,000 complicated pneumonia cases each year in the United States, as estimated by
pneumonia facts published by the American Thoracic Society in 2018. The test detects 19 microorganism targets and 10 antibiotic
resistance markers from endotracheal aspirates of adult hospitalized patients with suspected lower respiratory tract infections.
The Unyvero LRT test will also be available for testing bronchoalveolar lavage, or BAL, specimens of U.S. patients with lower
respiratory tract infections if our 510(k) submission, the receipt of which was acknowledged by the FDA on July 23, 2019, is cleared
for marketing in the United States. In Europe and other international markets, Curetis offers five CE-marked application cartridges
for hospitalized pneumonia, intra-abdominal infections, UTI, implant and tissue infections and blood stream infections. Curetis’
portfolio of these tests is highlighted in the figure below.

The
Unyvero Platform is a highly automated cartridge-based sample-to-answer molecular diagnostics platform based on multiplexed end-point
PCR with an array-based detection process. It integrates fully automated sample preparation, analysis and identification of disease
relevant pathogens and antibiotic resistance markers to provide timely high-quality information to its end-users. The scalable
system is designed to be either placed in laboratory settings or directly in hospital wards or intensive care units. Time-to-result
is four to five hours for the different application cartridges. The Unyvero Platform’s intuitive workflow with only minimal
hands-on time enables hospital staff to perform molecular tests at the point of need, such as in the ICU setting. Unyvero and
System Components are highlighted in the figure below. As of June 30, 2019, there were 170 Unyvero Analyzer placements globally.

Curetis’ ARESdb informatics
are currently commercialized through partnerships and an NGS service lab in Vienna, Austria. Curetis is accessing the AMR research
market through its exclusive bioinformatics collaboration with QIAGEN. Curetis has development agreements with Sandoz to reposition
antibiotics and recently introduced a specialized service laboratory offering of next-generation molecular AMR testing services
with an initial focus on infection control, AMR epidemiology and surveillance, clinical research and pharmaceutical anti-infectives
R&D. All services are based on NGS performed in Vienna, Austria and the ARESdb software.
Unyvero
A30 RQ Instrument
The Curetis Unyvero A30
RQ is a new device candidate designed to address the low to mid-plex testing market for 5-30 DNA targets. The device provides
results in 45 to 90 minutes with 2 to 5 minutes of hands on time. The Unyvero A30 RQ has a small laboratory footprint and
has an attractive cost of goods profile. Fully functional instrument system prototypes have been available since the fourth quarter
of 2018 and the first multiplex real-time PCR assays have been successfully transferred onto the Unyvero A30 RQ cartridges
and successfully benchmarked against their performance on standard PCR instruments. Curetis is aiming to have the Unyvero A30
RQ platform ready for partnering in 2020. The compact Unyvero A30 instrument and test cartridges are illustrated below.
We believe that the Unyvero A30 platform would also lend itself for the future development and regulatory approvals of certain
panels from the Acuitas and/or Unyvero side of Newco onto this novel platform.

|

|
Current
Commercial Agreements
Curetis has entered into a
number of distribution and collaboration agreements in the past few years, including:
|
|
·
|
In September 2019, Ares Genetics signed a technology evaluation agreement with an undisclosed global IVD corporation.
In the first phase of the collaboration, expected to take about 10 months, Ares Genetics expects to further enrich ARESdb with
a focus on certain pathogens relevant in a first, undisclosed infectious disease indication. Additional clinical isolates of such
pathogens will be sequenced by Ares Genetics at its recently established NGS laboratory in Vienna, Austria. Based on this enlarged
and enriched dataset, Ares Genetics expects to further optimize the algorithms for predictive antibiotic resistance testing for
drug/pathogen combinations particularly relevant in the targeted indication to enable NGS-based infectious disease diagnostics.
Under the agreement, the collaborator will fully fund Ares Genetics’ research and development activities for the genotypic
and phenotypic characterization of additional bacterial strains to augment ARESdb and the development of optimized algorithms for
predicting antibiotic resistance. Furthermore, in return for an up-front option fee, the collaborator obtained a right of first
negotiation for an exclusive, worldwide, human clinical diagnostic use license to ARESdb and the ARES Technology Platform for the
term of the technology evaluation agreement plus three months. The collaborator has the right to terminate the technology evaluation
agreement by providing limited notice to Ares Genetics.
|
|
|
·
|
In
March 2019, Curetis signed a distribution agreement with A. Menarini Diagnostics covering
the Unyvero A50 product line. Menarini is an established global healthcare company with
total revenues in 2018 of €3.6 billion, with over 1,000 employees in clinical diagnostics
and an installed base of 10,000 instruments in Europe. The collaboration initially includes
11 European countries with the option to expand into the Middle East and Africa. Including
the Menarini relationship, we anticipate that Newco will have 18 partners covering 43
countries in Europe, the Middle East, South America and Asia.
|
|
|
·
|
In
February 2019 Ares Genetics and QIAGEN GmbH
entered into a global strategic licensing agreement for ARESdb and AREStools in the area
of AMR research that aims to create a community platform for antimicrobial resistance
research. Under the terms of the agreement, QIAGEN obtained a restricted license to exclusively
develop and commercialize general bioinformatics offerings and services for AMR research
based on Ares Genetics’ database on the genetics of antimicrobial resistance. On
the signing of the agreement, Ares Genetics received a technology access fee and will
be entitled to a milestone payment at product launch, as well as industry-standard royalty
rates on net sales of products and services based on ARESdb and AREStools. Ares Genetics
retains the full rights to use ARESdb and AREStools for AMR research, customized bioinformatics
services, and the development of specific AMR assays and applications for the Curetis
Group including Ares Genetics, as well as third parties, including other diagnostics
companies or partners in the pharmaceutical industry. The license to QIAGEN excludes
any human diagnostic uses of ARESdb and/or AREStools. Under a separate agreement QIAGEN
has also provided the bulk of laboratory automation and equipment at favorable reagent
rental conditions to Ares Genetics.
|
|
|
·
|
In
September 2015, Curetis entered into an eight-year China distribution agreement with
Beijing Clear Biotech, which includes a minimum purchase commitment of Unyvero A50 systems.
The agreement also requires a minimum number of cartridge purchases. Following completion
of clinical trials, Beijing Clear Biotech Co. Ltd. has recently filed for regulatory approval
of the Unyvero A50 LRT test with the Chinese National Medical Products Administration
(NMPA; formerly Chinese Food and Drug Administration). Following a recent expert panel
hearing with the NMPA, approval is expected in 2020 with commercial launch also anticipated
in 2020.
|
Newco’s Strategy
We believe that by combining the Curetis and OpGen product
offerings and products in development, we can build and commercialize a comprehensive precision medicine solution for combatting
infectious disease with a focus on developing diagnostic tests for rapid pathogen identification and genetic profiling, antibiotic
resistance analysis and advanced informatics to store and analyze MDRO and other infectious disease data for hospitals, out-patient
settings and other healthcare providers. We believe that Newco will establish a market leadership position and will be able to
capitalize on global opportunities in infectious disease and AMR detection. Key elements of Newco’s anticipated strategy
are to:
|
|
·
|
continue to gain regulatory approvals and establish a market position for proprietary molecular
diagnostic tests and platforms;
|
|
|
·
|
capitalize on unique AMR bioinformatics solutions based on the Acuitas Lighthouse Software and
ARESdb to help differentiate Newco’s molecular diagnostic offerings and establish stand-alone product offerings directly
or through strategic partners;
|
|
|
·
|
leverage global commercial channel capabilities and partners to help accelerate growth and establish
a global footprint for Newco’s tests and informatics;
|
|
|
·
|
pursue partner relationships to help fund product development and to support commercialization
of products and services; and
|
|
|
·
|
capitalize on the financial leverage, operational and research synergies to help improve return
on capital and achieve future profitability.
|
The two core components of Newco’s strategy are the
development and commercialization of rapid diagnostic tests and leveraging AMR information services into new markets and channels.
We believe that antimicrobial resistance is an urgent
global healthcare issue. MDROs have been prioritized as an urgent national and global threat by the CDC, the executive branch
of the federal government and the World Health Organization. In March 2015, The White House issued a National Strategy for Combating
Antibiotic-Resistant Bacteria. This strategy calls for the strengthening of surveillance efforts to combat resistance, the development
and use of innovative diagnostic tests for identification and characterization of resistant bacteria and antibiotic stewardship
and development.
The CDC estimates that in the United States more than two
million people are sickened every year with antibiotic-resistant infections, with at least 23,000 dying as a result. Antibiotic-resistant
infections add considerable but often avoidable costs to the U.S. healthcare system. In most cases, these infections require prolonged
and/or costlier treatments, extended hospital stays, additional doctor visits and healthcare facilities use, and result in greater
disability and death compared with infections that are treatable with antibiotics. Estimates for the total economic cost to the
U.S. economy are difficult to calculate but the CDC has estimated such costs to be as high as $20 billion in excess direct healthcare
costs annually. As described in a December 2014 report issued by the Review on Antimicrobial Resistance commissioned by the U.K.
Prime Minister, titled “Antimicrobial Resistance: Tackling a Crisis for the Health and Wealth of Nations,” there are
estimated to be 700,000 deaths each year from antimicrobial resistance, including 50,000 deaths annually in the United States and
Europe.
|
|
·
|
Rapid
diagnostics – The two lead products for Newco’s rapid diagnostics
business are for lower respiratory infection and urinary tract infection. The LRT test
is based on the Unyvero A50 and was FDA cleared in 2018 for use with tracheal aspirates
as a sample type. In July 2019, Curetis filed for the 510(k) clearance of an LRT application
cartridge optimized for use with BAL as an additional sample type. BAL is another common
sample type for the diagnosis of lower respiratory tract infections. In response to its
July 2019 510(k) submission, Curetis received an AI request from the FDA in September
2019, effectively placing the 510(k) submission on hold until the FDA determines that
the deficiencies identified in the AI request have been resolved. Curetis has started
the response process in an interactive review format. Curetis anticipates it will be
able to respond to all information requests by the end of 2019. Curetis believes that
receipt of FDA clearance of an Unyvero LRT Application Cartridge for this additional
sample type would significantly increase the total addressable market for Unyvero in
the United States. Newco plans to continue to expand the commercial opportunity for the
Unyvero products by developing new tests, running additional clinical trials, pursuing
expanded regulatory approvals and through sales and marketing activities intended to
help increase commercial adoption and test usage. OpGen is developing OpGen-branded Acuitas
AMR Gene Panel tests for use on the Thermo Fisher Scientific Applied Biosystems™
QuantStudio™ 5 Real-Time PCR System. The first of these new tests will be for antibiotic
resistance testing of bacterial isolates. The second indication for the Acuitas AMR Gene
Panel is for management of patients with cUTI.
|
|
|
·
|
ARESdb
and Acuitas Lighthouse informatics and services – Newco plans to pursue commercial
opportunities to provide the Acuitas Lighthouse informatics and companion genomic testing
to pharmaceutical companies, CROs, health systems, third party in vitro diagnostic
companies, and government agencies. Through OpGen’s participation in The New York
State Infectious Disease Digital Health Initiative, we anticipate deploying the Acuitas
Lighthouse Software throughout the State of New York to help identify and track patients
with Superbug infections. The focus in the health system segment is on helping guide
antibiotic decision-making and supporting patient safety initiatives. Newco intends to
actively pursue government funding for development and deployment of the Acuitas Lighthouse
informatics in the United States and internationally.
|
In support of its strategy,
we anticipate that Newco will focus on:
|
|
·
|
obtaining
FDA clearance to market the Unyvero A50 LRT test for BAL specimens and expand the base
of commercial customers;
|
|
|
·
|
entering
into strategic partnering and licensing agreements to provide funding and support further
development of the Unyvero A30 platform;
|
|
|
·
|
completing
development and clinical evaluations, obtaining necessary regulatory approvals, and successfully
commercializing the Acuitas AMR Gene Panel (Urine) for cUTIs, with a goal of achieving
three-hour antibiotic resistance analysis from the time of specimen collection;
|
|
|
·
|
commercializing
the Acuitas AMR Gene Panel tests for RUO, which started in January 2018 and for which
on May 13, 2019, we filed a 510(k) submission with the FDA for clearance for the detection
of antimicrobial resistance genes in bacterial isolates;
|
|
|
·
|
making
additional FDA 510(k) submissions for the Acuitas AMR Gene Panel (Urine) test anticipated
in the first quarter of 2020, and the Acuitas Lighthouse Software (AMR Gene Panel Prediction)
anticipated in the first half of 2020;
|
|
|
·
|
successfully
completing the demonstration project of The New York State Digital Health Initiative
to support Statewide deployment in subsequent years;
|
|
|
·
|
obtaining
third-party funding to expand the ARESdb offerings in conjunction with established in
vitro diagnostic companies;
|
|
|
·
|
expanding
our business collaborations with Merck, Sandoz and other pharmaceutical companies;
|
|
|
·
|
capitalizing
on opportunities to deploy the Acuitas Lighthouse informatics and genomic testing for
pharmaceutical/CRO services;
|
|
|
·
|
growing
the ARESdb and Acuitas Lighthouse data warehouse offerings for resistance and susceptibility
data in hospital, hospital system, or broader community applications;
|
|
|
·
|
seeking
government funding to advance programs focused on identification and treatment of MDROs;
and
|
|
|
·
|
continuing
development of the Acuitas Lighthouse Software and work to install Acuitas Lighthouse
Software to customer sites in the United States and globally.
|
Risk
Factors
Our business is subject to
numerous risks and uncertainties, including those highlighted in the section entitled “Risk Factors” immediately following
this prospectus summary. These risks include, but are not limited to, the following:
|
|
·
|
we
have a history of losses and expect to incur losses for the next several years;
|
|
|
·
|
we
have not yet consummated the Newco transaction with Curetis, and need to raise additional
funds to support our business and the operations of Curetis prior to the closing under
the Implementation Agreement, and to support Newco’s business after the closing;
|
|
|
·
|
the
transactions contemplated by the Implementation Agreement may not close because of failure
to meet one of the conditions to closing;
|
|
|
·
|
many
of the material terms of the short-term, subordinated credit facility with Curetis required
by the Implementation Agreement, or the Interim Facility, including the interest rate,
term and committed amount, have not yet been negotiated, and we may be unable to negotiate
favorable terms;
|
|
|
·
|
we
may need to pursue an additional financing to fund Newco’s operations after the
closing;
|
|
|
·
|
the
process of obtaining FDA clearance and/or approval is time-consuming and expensive, and
we may not be successful in obtaining such clearances or approvals in a timely manner
or at all;
|
|
|
·
|
our
products may never achieve significant commercial market acceptance;
|
|
|
·
|
our
contracts with government agencies could be subject to uncertain future funding;
|
|
|
·
|
our
sales cycle is lengthy and variable; and
|
|
|
·
|
we
may not be able to compete successfully with the products and services sold by other
companies in our industry, who are better capitalized than we are.
|
Nasdaq
Listing Requirements and Reverse Stock Split
On May 6, 2019,
the Listing Qualifications Staff of the Nasdaq Capital Market notified us that the closing bid price of our common stock had,
for 30 consecutive business days preceding the date of such notice, been below the $1.00 per share minimum required for continued
listing on the Nasdaq Capital Market pursuant to Nasdaq Marketplace Rule 5550(a)(2), or the Minimum Bid Price Rule. In accordance
with Nasdaq Marketplace Rule 5810(c)(3)(A), we were provided 180 calendar days, or until November 4, 2019, to regain compliance.
If at any time before November 4, 2019, the closing bid price of our common stock is at least $1.00 for a minimum of ten consecutive
trading days, we will regain compliance.
On August 22,
2019, at the annual meeting of stockholders, our stockholders approved an amendment to our Amended and Restated Certificate of
Incorporation, authorizing a reverse stock split of the issued and outstanding shares of our common stock, at a ratio within a
range of not less than five-to-one and not more than twenty-five-to-one, such ratio and the implementation and timing of such
reverse stock split to be determined in the discretion of our Board of Directors. On August 22, 2019, our Board of Directors
approved a reverse stock split of one share for every twenty outstanding shares, or the 2019 Reverse Stock Split. On August 28,
2019, we filed an amendment to our Amended and Restated Certificate of Incorporation to effect the 2019 Reverse Stock Split. All
of the Company’s historic share and share prices in this prospectus have been adjusted to reflect the 2019 Reverse Stock
Split.
In implementing
the 2019 Reverse Stock Split, the number of shares of our common stock held by each stockholder was reduced by dividing the number
of shares held immediately before the 2019 Reverse Stock Split by twenty and then rounding down to the nearest whole share. We
are paying cash to each stockholder in lieu of issuing any fractional shares. The 2019 Reverse Stock Split did not affect any
stockholder’s percentage ownership interest in our Company or proportionate voting power, except to the extent that interests
in fractional shares were paid in cash.
In addition, we
have adjusted all outstanding shares of any restricted stock units, stock options and warrants entitling the holders to purchase
shares of our common stock as a result of the 2019 Reverse Stock Split, as required by the terms of these securities. In particular,
we have reduced the conversion ratio for each security, and increased the exercise price in accordance with the terms of each
security based on 2019 Reverse Stock Split ratio (i.e., the number of shares issuable under such securities has been divided by
twenty, and the exercise price per share has been multiplied by twenty). Also, we proportionately reduced the number of
shares reserved for issuance under our existing 2015 Equity Incentive Plan, or the 2015 Plan, based on the 2019 Reverse Stock
Split ratio. The 2019 Reverse Stock Split did not otherwise affect any of the rights currently accruing to holders of our common
stock, or options or warrants exercisable for our common stock.
We have regained
compliance with the Minimum Bid Price Rule as of September 13, 2019, as a result of the 2019 Reverse Stock Split. Although we
expect that the 2019 Reverse Stock Split will result in a sustained increase in the market price of our common stock, the 2019
Reverse Stock Split may not result in a permanent increase in the market price of our common stock, which is dependent on many
factors, including general economic, market and industry conditions and other factors detailed from time to time in the reports
we file with the SEC.
On August 19, 2019, OpGen, received
a written notification from The Nasdaq Stock Market LLC, or Nasdaq, notifying the Company that it has failed to comply with Nasdaq
Marketplace Rule 5550(b)(1) because the Company’s stockholders’ equity as of June 30, 2019 fell below the required
minimum of $2,500,000, and as of June 30, 2019, the Company did not meet the alternative compliance standards of market value
of listed securities or net income from continuing operations for continued listing. In accordance with Nasdaq’s listing
requirements, the Company had 45 calendar days to submit a plan to regain compliance. If the plan is accepted, Nasdaq can grant
the Company an extension of up to 180 calendar days from the date it received the notification to evidence compliance. The Company
submitted a plan to Nasdaq to regain compliance with the Nasdaq minimum stockholders’ equity standard on October 3, 2019,
which plan includes information regarding this offering. The Company believes that if this offering is successful, we will regain
compliance with the minimum stockholders’ equity standard for continued listing. However, there can be no assurance that
the Company’s plan will be accepted or that if it is, that the Company will be able to regain compliance.
THE OFFERING
|
Units
offered by us in this offering:
|
1,526,717
units, each consisting of one share of our common stock and one common warrant to purchase one share of our common stock.
|
|
Pre-funded units
offered by us in this offering:
|
We are also offering
to each purchaser whose purchase of units in this offering would otherwise result in the purchaser, together with its affiliates
and certain related parties, beneficially owning more than 4.99% (or, at the election of the purchaser, 9.99%) of our
outstanding common stock immediately following the consummation of this offering, the opportunity to purchase, if the purchaser
so chooses, pre-funded units (each pre-funded unit consisting of one pre-funded warrant to purchase one share of our common
stock and one common warrant to purchase one share of our common stock) in lieu of units that would otherwise result in the
purchaser’s beneficial ownership exceeding 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding
common stock. The purchase price of each pre-funded unit will equal the price at which the units are being sold to the public in
this offering, minus $0.01, and the exercise price of each pre-funded warrant included in each pre-funded unit will be $0.01
per share. This offering also relates to the shares of common stock issuable upon exercise of any pre-funded warrants underlying
the pre-funded units sold in this offering. For each pre-funded unit we sell, the number of units we are offering will be
decreased on a one-for-one basis. Because we will issue a common warrant as part of each unit or pre-funded unit, the number
of common warrants sold in this offering will not change as a result of a change in the mix of the units and pre-funded units
sold.
|
|
Common warrants
offered by us in the offering
|
Common warrants
to purchase an aggregate of 1,526,717 shares of our common stock. Each unit and each pre-funded unit includes a common warrant
to purchase one share of our common stock. Each common warrant will have an exercise price of $ per
share, will be immediately separable from the common stock or pre-funded warrant, as the case may be, will be immediately
exercisable and will expire on the fifth anniversary of the original issuance date. This prospectus also relates to the offering
of the shares of common stock issuable upon exercise of the common warrants.
|
|
Option to purchase additional securities:
|
The underwriter has a 30-day option to purchase up to 229,007 additional shares of our common stock and/or
warrants to purchase up to an additional 229,007 shares of our common stock from us at the public offering price, less underwriting
discounts and commissions.
|
|
Common stock outstanding prior to this offering:
|
882,268 shares of
common stock.
|
|
Common stock outstanding
after this offering:
|
2,408,985 shares
(or 2,637,992 shares if the underwriter exercises its option to purchase additional shares in full), in each case assuming
no sale of pre-funded units and no exercise of the common warrants issued in this offering.
|
|
Use of Proceeds:
|
We
currently intend to use the net proceeds of this offering for the following purposes: prior to the closing of the transactions
contemplated by the Implementation Agreement to (1) complete the business combination with Curetis; (2) provide short-term
funding to Curetis under the Interim Facility to fund the Curetis Group’s current operations; and (3) support research
and development and regulatory activities for the Company’s anticipated FDA 510(k) submissions for the Acuitas AMR Gene
Panel test and the Acuitas Lighthouse Software; and, if any proceeds remain following the closing of the transactions under
the Implementation Agreement, to: (4) commercialize Newco’s products, with a focus on the Unyvero platform and diagnostic
tests, and the Acuitas AMR Gene Panel tests; (5) support further development and commercialization of the Ares Genetics database
and Acuitas Lighthouse Software; (6) fund directed efforts to the customers and collaborators of each company to introduce
the products and services of Newco; (7) invest in manufacturing and operations infrastructure to support sales of products;
and (8) the balance, if any, for general corporate purposes. If the transactions under the Implementation Agreement do not
close, to the extent any proceeds remain, we plan to use any remaining proceeds to support OpGen’s operations as far
as possible into 2020. Many of the material terms of the Interim Facility, including the interest rate, term and committed
amount, have not yet been negotiated, and we may be unable to negotiate favorable terms. See “Use of Proceeds”
on page 64 of this prospectus and “Risk Factors” beginning on page 34 of this prospectus.
|
|
Risk Factors:
|
Investing in our securities involves a high degree of risk. See “Risk Factors” beginning on page 34 of this prospectus and the other information included or incorporated by reference in this prospectus.
|
|
Nasdaq Capital
Market symbol:
|
“OPGN.”
There is no established trading market for the common warrants or the pre-funded warrants, and we do not expect a trading
market to develop. We do not intend to list the common warrants or the pre-funded warrants on Nasdaq, any national securities
exchange or any other nationally-recognized trading system. Without an active trading market, the liquidity of the common
warrants and pre-funded warrants will be limited.
|
The number of shares of common stock to be outstanding immediately after this offering is based on 882,268 shares of our common
stock outstanding as of June 30, 2019, and excludes:
|
|
·
|
10,542 shares of common stock issuable
upon the exercise of outstanding options granted as of June 30, 2019, under our equity incentive plans at a weighted average exercise
price of $410.31 per share;
|
|
|
·
|
175,982
shares of common stock issuable upon the
exercise of outstanding warrants issued as of June 30, 2019, at a weighted average exercise
price of $288.25 per share;
|
|
|
·
|
15,663 shares of common stock issuable upon vesting of outstanding restricted stock units granted
as of June 30, 2019; and
|
|
|
·
|
4,221 shares of common stock available for future issuance under our equity incentive plans as
of June 30, 2019;
|
|
|
·
|
76,335 shares
of common stock, or 87,786 shares of common stock if the underwriter exercises its option
to purchase additional securities in full, issuable upon exercise of warrants to be issued
to the underwriter at an exercise price of 130% of the public offering price as described
in “Underwriting,”
|
|
|
·
|
1,526,717
shares of common stock issuable upon the exercise of the common warrants to be issued
to purchasers in this offering at an exercise price of $ per
share; and
|
|
|
·
|
up to 2,662,564 shares of common stock issuable pursuant to the terms of the Implementation Agreement.
|
The number of outstanding options,
restricted stock units and shares of common stock available for future issuances under our equity incentive plans does not reflect
grants of 1,500 shares of common stock issuable upon vesting of restricted stock units, the expiration of stock options to purchase
499 shares of our common stock, forfeitures of stock options to purchase 107 shares of our common stock or forfeitures of 500
shares of our common stock issuable upon vesting of restricted stock units since June 30, 2019.
Unless otherwise indicated, all information
contained in this prospectus assumes (i) that the underwriter has not exercised its option to purchase additional securities, (ii) no sale of pre-funded units in this offering, which, if sold, would reduce the number of units that we are offering
on a one-for-one basis, and (iii) no exercise of options issued under our equity incentive plans or of warrants, including the
common warrants offered in this offering and the underwriter’s warrants to be issued to the underwriter in connection with
this offering.
Company and Other Information
OpGen, Inc. was incorporated in Delaware in 2001. On July
14, 2015, the Company acquired AdvanDx, Inc., a Delaware corporation, as a wholly-owned subsidiary in a merger transaction, or
the AdvanDx Merger. On September 3, 2019, we formed Crystal GmbH, a private limited liability company organized under the laws
of the Federal Republic of Germany for the sole purpose of acquiring the Transferred Shares and transferred assets and liabilities
of the Curetis business. Our principal executive office is located at 708 Quince Orchard Road, Gaithersburg, Maryland, 20878,
and our telephone number is (240) 813-1260. The Company also has operations in Copenhagen, Denmark and Bogota, Colombia. Our website
address is www.opgen.com. We do not incorporate the information on or accessible through our website into this prospectus, and
you should not consider any information on, or that can be accessed through, our website as part of this prospectus.
Implications
of Being an Emerging Growth Company
As a company with less than
$1.07 billion in revenue during our last fiscal year, we qualify as an “emerging growth company” as defined in the
Jumpstart Our Business Startups Act, or the JOBS Act, enacted in April 2012. An “emerging growth company” may take
advantage of exemptions from some of the reporting requirements that are otherwise applicable to public companies. These exceptions
include:
|
|
·
|
being
permitted to present only two years of audited financial statements and only two years
of related Management’s Discussion and Analysis of Financial Condition and Results
of Operations in this prospectus;
|
|
|
·
|
not
being required to comply with the auditor attestation requirements of Section 404 of
the Sarbanes-Oxley Act of 2002, as amended;
|
|
|
·
|
reduced
disclosure obligations regarding executive compensation in our periodic reports, proxy
statements and registration statements; and
|
|
|
·
|
exemptions
from the requirements of holding a nonbinding advisory vote on executive compensation
and stockholder approval of any golden parachute payments not previously approved.
|
We may take advantage of these
provisions until the last day of our fiscal year following the fifth anniversary of the closing of our initial public offering
in May 2015. However, if certain events occur prior to the end of such five-year period, including if we become a “large
accelerated filer,” our annual gross revenue exceeds $1.07 billion or we issue more than $1.0 billion of non-convertible
debt in any three-year period, we will cease to be an emerging growth company prior to the end of such five-year period.
We have elected to take advantage
of certain of the reduced disclosure obligations in this prospectus and may elect to take advantage of other reduced reporting
requirements in future filings. As a result, the information that we provide to our stockholders may be different than you might
receive from other public reporting companies in which you hold equity interests.
In addition, the JOBS Act
provides that an emerging growth company can take advantage of an extended transition period for complying with new or revised
accounting standards. We have irrevocably elected not to avail ourselves of this exemption and, therefore, we will be subject
to the same new or revised accounting standards as other public companies that are not emerging growth companies.
OPGEN
SUMMARY FINANCIAL DATA
The following summary financial
data should be read together with our financial statements and related notes, and “Management’s Discussion and Analysis
of Financial Condition and Results of Operations” included in our latest Annual Report on Form 10-K and Quarterly Reports
on Form 10-Q and incorporated by reference into this prospectus. The summary statements of operations data for the years ended
December 31, 2018 and 2017 and the six months ended June 30, 2019 and 2018, and the balance sheet data as of June 30, 2019 have
been derived from our audited financial statements and unaudited interim condensed financial statements incorporated by reference
into this prospectus. Historical results are not necessarily indicative of the results that may be expected in the future.
|
|
|
Year
Ended
December
31,
|
|
Six
Months Ended
June 30,
|
|
|
|
2018
|
|
2017
|
|
2019
|
|
2018
|
|
|
|
(In thousands,
except per share data)
|
|
|
|
|
|
|
|
(Unaudited)
|
|
Statements of Operation Data:
|
|
|
|
|
|
|
|
|
|
Revenue
|
|
$
|
2,946
|
|
|
$
|
3,211
|
|
|
$
|
2,030
|
|
|
$
|
1,635
|
|
|
Operating expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of products sold
|
|
|
1,223
|
|
|
|
1,613
|
|
|
|
419
|
|
|
|
646
|
|
|
Cost of services(1)
|
|
|
626
|
|
|
|
520
|
|
|
|
396
|
|
|
|
348
|
|
|
Research and development(1)
|
|
|
5,677
|
|
|
|
6,883
|
|
|
|
2,930
|
|
|
|
2,535
|
|
|
General and administrative(1)
|
|
|
7,069
|
|
|
|
6,693
|
|
|
|
3,340
|
|
|
|
3,622
|
|
|
Sales and marketing(1)
|
|
|
1,532
|
|
|
|
2,768
|
|
|
|
766
|
|
|
|
756
|
|
|
Impairment of right-of-use
asset
|
|
|
—
|
|
|
|
—
|
|
|
|
521
|
|
|
|
—
|
|
|
Total operating expenses(1)
|
|
|
16,127
|
|
|
|
18,477
|
|
|
|
8,372
|
|
|
|
7,907
|
|
|
Operating loss
|
|
|
(13,181
|
)
|
|
|
(15,266
|
)
|
|
|
(6,342
|
)
|
|
|
(6,272
|
)
|
|
Interest and other (expense) income
|
|
|
5
|
|
|
|
(87
|
)
|
|
|
(9
|
)
|
|
|
5
|
|
|
Interest expense
|
|
|
(191
|
)
|
|
|
(233
|
)
|
|
|
(94
|
)
|
|
|
(112
|
)
|
|
Foreign currency transaction gains (losses)
|
|
|
(10
|
)
|
|
|
23
|
|
|
|
—
|
|
|
|
(10
|
)
|
|
Change in fair value of derivative financial instruments
|
|
|
8
|
|
|
|
144
|
|
|
|
—
|
|
|
|
8
|
|
|
Provision for income taxes
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Net loss
|
|
$
|
(13,369
|
)
|
|
$
|
(15,419
|
)
|
|
$
|
(6,445
|
)
|
|
$
|
(6,381
|
)
|
|
Net loss per common share, basic and diluted
|
|
$
|
(44.69
|
)
|
|
$
|
(195.95
|
)
|
|
$
|
(9.54
|
)
|
|
$
|
(25.78
|
)
|
|
Weighted average shares outstanding—basic and diluted
|
|
|
300
|
|
|
|
79
|
|
|
|
676
|
|
|
|
248
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1)
|
Includes
stock-based compensation as follows:
|
|
|
|
Year Ended
December 31,
|
|
Six Months Ended
June 30,
|
|
|
|
2018
|
|
2017
|
|
2019
|
|
2018
|
|
|
|
|
|
|
|
(Unaudited)
|
|
Cost of services
|
|
$
|
1
|
|
|
$
|
14
|
|
|
$
|
1
|
|
|
$
|
4
|
|
|
Research and development
|
|
|
241
|
|
|
|
237
|
|
|
|
35
|
|
|
|
131
|
|
|
General and administrative
|
|
|
574
|
|
|
|
604
|
|
|
|
137
|
|
|
|
292
|
|
|
Sales and marketing
|
|
|
46
|
|
|
|
57
|
|
|
|
11
|
|
|
|
25
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total stock-based compensation
|
|
$
|
862
|
|
|
$
|
912
|
|
|
$
|
184
|
|
|
$
|
452
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
As
of June 30, 2019
|
|
|
|
Actual
|
|
As
Adjusted
|
|
|
|
(In thousands)
|
|
Balance Sheet Data:
|
|
(Unaudited)
|
|
Cash and cash equivalents
|
|
$
|
3,056
|
|
|
$
|
11,950
|
|
|
Working capital (deficiency)
|
|
|
(584
|
)
|
|
|
8,310
|
|
|
Total assets
|
|
|
8,933
|
|
|
|
17,826
|
|
|
Accumulated deficit
|
|
|
(168,525
|
)
|
|
|
(168,525
|
)
|
|
Total stockholders’ equity
|
|
|
1,827
|
|
|
|
10,721
|
|
The preceding table presents
a summary of our balance sheet data as of June 30, 2019:
|
|
·
|
on
an as adjusted basis to give effect to the receipt of the estimated net proceeds from
the sale of an aggregate of 1,526,717 units and no pre-funded units in this offering
at the assumed public offering price of $6.55 per unit and the issuance of 1,526,717
shares of common stock included in the units.
|
CURETIS
BUSINESS SUMMARY FINANCIAL DATA
For purposes of the Curetis Business combined financial
statements included in this prospectus, we refer to the business of Curetis N.V., principally operated by Curetis GmbH and its
subsidiaries, or the Curetis Group, as the Curetis Business. In the Curetis Business combined financial statements included in
the prospectus, the business of Curetis N.V. is presented, which comprises the Curetis Group as well as the Curetis Convertible
Notes that are assumed by OpGen pursuant to the Implementation Agreement and certain costs related to the Curetis Business, primarily
related to the compensation of certain members of senior management and its supervisory board that were historically incurred
by Curetis N.V. but not charged to the Curetis Group.
The following summary financial data
should be read together with the combined financial statements and related notes of the Curetis Business included in this prospectus.
The combined statements of operations and other comprehensive income for the years ended December 31, 2018 and 2017, have been
derived from the audited, combined financial statements of the Curetis Business for the years ended December 31, 2018 and 2017
included in the prospectus. All Curetis financial results and measures in this prospectus, other than the Curetis Business combined
financial statements, have been translated from Euros to U.S. dollars using the translation rates listed below or, otherwise,
of $1.13667 to €1.00 as of June 30, 2019, based on Oanda.com. These translation rates are provided for convenience only,
and OpGen makes no representation that the Euro amounts could have been, or could be, converted, realized or settled in U.S. dollars
at that rate on June 30, 2019, or at any other rate.
The unaudited combined interim financial
statements of the Curetis Business for the six months ended June 30, 2019 and 2018, and the combined statement of financial position
data as of June 30, 2019, have been derived from the unaudited interim condensed combined financial statements of the Curetis
Business as of and for the six months ended June 30, 2019. The combined financial statements as of and for the years ended December
31, 2018 and 2017 were prepared in accordance with IFRS as issued by the IASB. The unaudited interim condensed combined financial
statements of the Curetis Business were prepared in accordance with IFRS as issued by the IASB applicable for interim reporting
(IAS 34). Historical results are not necessarily indicative of the results that may be expected in the future.
|
|
|
|
|
|
|
|
|
|
|
|
|
Year Ended
|
|
Six Months Ended
|
|
|
|
December
31,
|
|
June
30,
|
|
|
|
2018
|
|
2017
|
|
2019
|
|
2018
|
|
Statements of Profits or Loss:
|
|
(In thousands
of USD (1))
|
|
Revenue
|
|
$
|
1,623
|
|
|
$
|
1,422
|
|
|
$
|
1,237
|
|
|
$
|
940
|
|
|
Cost of sales
|
|
|
(1,558
|
)
|
|
|
(1,582
|
)
|
|
|
(1,525
|
)
|
|
|
(1,275
|
)
|
|
Gross loss
|
|
|
65
|
|
|
|
(160
|
)
|
|
|
(288
|
)
|
|
|
(335
|
)
|
|
Distribution costs
|
|
|
(9,318
|
)
|
|
|
(8,632
|
)
|
|
|
(3,717
|
)
|
|
|
(4,903
|
)
|
|
Administrative expenses
|
|
|
(4,092
|
)
|
|
|
(3,815
|
)
|
|
|
(1,832
|
)
|
|
|
(2,175
|
)
|
|
Research & development expenses
|
|
|
(12,085
|
)
|
|
|
(8,786
|
)
|
|
|
(4,752
|
)
|
|
|
(5,451
|
)
|
|
Other income
|
|
|
715
|
|
|
|
206
|
|
|
|
130
|
|
|
|
220
|
|
|
Operating loss
|
|
|
(24,715
|
)
|
|
|
(21,187
|
)
|
|
|
(10,459
|
)
|
|
|
(12,644
|
)
|
|
Finance income
|
|
|
39
|
|
|
|
13
|
|
|
|
8
|
|
|
|
58
|
|
|
Finance costs
|
|
|
(1,374
|
)
|
|
|
(835
|
)
|
|
|
(846
|
)
|
|
|
(575
|
)
|
|
Finance result -- net
|
|
|
(1,335
|
)
|
|
|
(822
|
)
|
|
|
(838
|
)
|
|
|
(517
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss before income taxes
|
|
|
(26,050
|
)
|
|
|
(22,009
|
)
|
|
|
(11,297
|
)
|
|
|
(13,161
|
)
|
|
Income tax expense
|
|
|
(41
|
)
|
|
|
65
|
|
|
|
(64
|
)
|
|
|
30
|
|
|
Loss for the period
|
|
$
|
(26,091
|
)
|
|
$
|
(21,944
|
)
|
|
$
|
(11,361
|
)
|
|
$
|
(13,131
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1) Convenience translation performed from Euros to U.S. Dollars
using the following exchange rate in effect as of each period end:
|
|
|
1.14379
|
|
|
|
1.19786
|
|
|
|
1.13667
|
|
|
|
1.16478
|
|
|
|
|
As
of June 30, 2019
|
|
Consolidated Statement of Financial Position
Data
|
|
|
(In
thousands of USD (1))
|
|
|
Cash and cash equivalents
|
|
|
5,432
|
|
|
Working capital
|
|
|
5,719
|
|
|
Total assets
|
|
|
26,478
|
|
|
Accumulated deficit
|
|
|
(193,632
|
)
|
|
Total equity
|
|
|
(3,347
|
)
|
|
|
|
|
|
|
|
(1) Convenience translation performed from Euros to U.S. Dollars
using the following exchange rate in effect as of June 30, 2019:
|
|
|
1.13667
|
|
UNAUDITED
PRO FORMA CONSOLIDATED COMBINED FINANCIAL INFORMATION
The following unaudited pro
forma condensed combined financial information was prepared using the acquisition method of accounting under U.S. GAAP.
For purposes of this unaudited pro forma condensed combined
financial information, we refer to the business of Curetis N.V., principally operated by Curetis GmbH and its subsidiaries as
the Curetis Business. In the Curetis Business combined financial statements included in the prospectus, the business of Curetis
N.V. is presented, which comprises the Curetis Group as well as the Curetis Convertible Notes that are assumed by OpGen pursuant
to the Implementation Agreement and certain costs related to the Curetis Business, primarily related to the compensation of certain
members of senior management and its supervisory board that were historically incurred by Curetis N.V. but not charged to the
Curetis Group.
If the business combination contemplated by the Implementation
Agreement was consummated without the need for this offering, it would be accounted for as a reverse acquisition in accordance
with U.S. GAAP. Under this method of accounting, the Curetis Business would be deemed to be the accounting acquirer for financial
reporting purposes. One of the conditions to closing the business combination under the Implementation Agreement is that OpGen
raise at least $10 million in interim financing. In presenting this unaudited pro forma consolidated combined financial information,
we have considered, among other factors, the following: (i) the number of shares to be issued to Curetis N.V. under the Implementation
Agreement; (ii) the number of units assumed to be sold in this offering; (iii) whether the percentage of voting rights held by
Curetis N.V. would continue to constitute a majority of the voting rights of OpGen after this offering and after closing under
the Implementation Agreement; (iv) the contractual right held by Curetis N.V. to designate a majority of the members of the initial
board of directors of OpGen after the closing; and (v) the change in the chief executive officer of OpGen after the closing to
be the chief executive officer of Curetis N.V. The consummation of this offering will have a substantial impact on the final determination
as to the accounting treatment of the business combination but, as of the date of this prospectus, we cannot be sure whether
the accounting treatment will change. Accordingly, we are presenting this unaudited pro forma consolidated combined financial
information as a reverse acquisition for accounting purposes. This unaudited pro forma consolidated combined financial information,
as presented, includes the number of shares to be issued to Curetis N.V. under the Implementation Agreement and the number of
units to be offered and sold in this offering, at an assumed offering price of $6.55 per unit, equal to the closing price of our
common stock on October 9, 2019. Accounting for the business combination as a reverse acquisition is the equivalent of the Curetis
Business issuing stock to acquire OpGen. As a result, as of the closing date of the business combination, the net assets of OpGen
would be recorded at their acquisition-date fair values in the financial statements of the Curetis Business and the reported operating
results prior to the business combination would be those of the Curetis Business. In addition, transaction costs incurred by Curetis
in connection with the business combination would be expensed as incurred.
We cannot assure you that the business combination will
be accounted for as a reverse acquisition. The final determination will be made after this offering is completed and once the
business combination is completed. In addition, the boards of Curetis and OpGen expect that the size of the post-closing Board
of OpGen will be up to seven members, with OpGen having input as to three of such members, and the current Chief Financial Officer
of OpGen will continue as the Chief Financial Officer of OpGen after the closing. Other than the change in Chief Executive Officer,
there is also no current intention to replace any existing executive officers of OpGen. OpGen and Curetis will re-assess the accounting
for the business combination after the consummation of this offering. However, we have assumed, for purposes of these unaudited
pro forma condensed combined financial statements, that Curetis N.V. will continue to hold a majority of the outstanding shares
of OpGen common stock at the closing, and that other indicia of control will lead to a determination that the business combination
should be accounted for as a reverse acquisition.
The unaudited pro forma condensed
combined balance sheet data assume that the business combination took place on June 30, 2019 and combines the historical
balance sheets of OpGen and the Curetis Business as of such date. The unaudited pro forma condensed combined balance sheet data
also assume the minimum capital raise, as required by the Implementation Agreement, was completed on June 30, 2019. The unaudited
pro forma condensed combined statement of operations data for the year ended December 31, 2018 and the six months ended June 30,
2019, assume that the business combination took place as of January 1, 2018, and combine the historical results of OpGen
and the Curetis Business for the year ended December 31, 2018 and the six months ended June 30, 2019, respectively.
The unaudited pro forma condensed combined financial information was prepared in accordance with the rules and regulations of
Article 11 of SEC Regulation S-X. The historical financial statements of OpGen and the Curetis Business have been adjusted
to give pro forma effect to events that are (i) directly attributable to the transaction, (ii) factually supportable,
and (iii) with respect to the unaudited pro forma condensed combined statements of operations, expected to have a continuing
impact on Newco’s results.
The combined financial statements
of the Curetis Business were prepared in accordance with International Financial Reporting Standards, or IFRS, as issued by the
International Accounting Standards Board, or IASB. The consolidated financial statements of OpGen were prepared in accordance
with U.S. GAAP. OpGen has performed a preliminary analysis and has not identified significant differences identified between IFRS
and U.S. GAAP for the purposes of presenting the unaudited pro forma condensed combined financial statements. In addition, the
unaudited condensed combined financial statements reflect reclassifications to conform the Curetis Business historical accounting
presentation to OpGen’s accounting presentation.
The consolidated financial
statements of OpGen are presented in US dollars, or USD, whereas, the financial statements of the Curetis Business are presented
in Euros. Therefore, the unaudited pro forma condensed combined financial information includes adjustments to convert the Curetis
Business’s financial information from Euros to USD.
OpGen's assets and liabilities
will be measured and recognized at their fair values as of the transaction date and combined with the assets, liabilities and
results of operations of the Curetis Business after the consummation of the business combination. The allocation of the purchase
price to acquired assets and assumed liabilities based on their underlying fair values requires the extensive use of significant
estimates and management’s judgment. The allocation of the purchase price is preliminary at this time, and will remain as
such until management completes valuations and other studies in order to finalize the valuation of the net assets acquired. These
provisional estimates will be adjusted upon the availability of further information regarding events or circumstances which exist
at the acquisition date and such adjustments may be significant. OpGen’s intangible assets have not yet been determined
and, therefore, the allocation of the purchase price in excess of OpGen’s net assets is shown entirely as goodwill.
The unaudited pro forma condensed
combined financial information is based on the assumptions and adjustments that are described in the accompanying notes. Accordingly,
the pro forma adjustments are preliminary, subject to further revision as additional information becomes available and additional
analyses are performed, including but not limited to the final assessment of the accounting acquirer, of the determination of
differences between IFRS and US GAAP, and of the application of purchase price adjustments, and have been made solely for the
purpose of providing unaudited pro forma condensed combined financial information. Differences between these preliminary estimates
and the final accounting, expected to be completed after the closing of the business combination, will occur and these differences
could have a material impact on the accompanying unaudited pro forma condensed combined financial information and the combined
company's future results of operations and financial position. In addition, depending on the number of shares issued in the interim
equity offering contemplated by this prospectus, the method for accounting of the business combination could change.
The unaudited pro forma condensed
combined financial information does not give effect to the potential impact of current financial conditions, regulatory matters,
operating efficiencies or other savings or expenses that may be associated with the integration of the business of OpGen and the
Curetis Business. The unaudited pro forma condensed combined financial information is preliminary and has been prepared for illustrative
purposes only and is not necessarily indicative of the financial position or results of operations in future periods or the results
that actually would have been realized had OpGen and the Curetis Business been a combined company during the specified periods.
The actual results reported in periods following the business combination may differ significantly from those reflected in the
unaudited pro forma condensed combined financial information presented herein for a number of reasons, including, but not limited
to, differences in actual performance compared to the assumptions used to prepare this pro forma financial information.
The unaudited pro forma condensed
combined financial information, including the notes thereto, should be read in conjunction with the separate historical financial
statements of OpGen and the Curetis Business, incorporated by reference in this prospectus or included elsewhere in this prospectus.
OpGen's audited statement of operations for the year ended December 31, 2018 is derived from OpGen’s Annual Report
on Form 10-K for the year ended December 31, 2018. OpGen's unaudited financial statements for the six months ended June 30,
2019 are derived from OpGen's Quarterly Report on Form 10-Q for the six months ended June 30, 2019.
Accounting rules require evaluation
of certain assumptions, estimates, or determination of financial statement classifications which are completed during the measurement
period as defined in current accounting standards. The accounting policies of OpGen may materially vary from those of the Curetis
Business. During preparation of the unaudited pro forma condensed combined financial information, management has performed a preliminary
analysis and is not aware of any material differences, and accordingly, this unaudited pro forma condensed combined financial
information assumes no material differences in accounting policies. Following the acquisition, management will conduct a final
review of OpGen's accounting policies in order to determine if differences in accounting policies require adjustment or reclassification
of OpGen's results of operations or reclassification of assets or liabilities to conform to the Curetis Business' accounting policies
and classifications. As a result of this review, management may identify differences that, when conformed, could have a material
impact on these unaudited pro forma condensed combined financial statements.
Following receipt of approval
from stockholders at a special meeting of stockholders held on August 22, 2019, on August 28, 2019, OpGen filed an amendment to
its Amended and Restated Certificate of Incorporation to effect a reverse stock split of the issued and outstanding shares of
our common stock, at a ratio of one share for twenty shares. All financial information in this pro forma financial information
has been adjusted to reflect the 2019 Reverse Stock Split.
|
OpGen, Inc.
|
|
Unaudited Pro Forma Condensed Combined Balance Sheet
|
|
June 30, 2019
|
|
(in thousands)
|
|
|
|
OpGen
|
|
Curetis
Business
|
|
Pro Forma Adjustments
|
|
Notes
|
|
Pro Forma Combined
|
|
Assets
|
|
|
|
|
|
|
|
|
|
|
|
Current assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
3,056
|
|
|
$
|
5,432
|
|
|
$
|
8,894
|
|
|
|
J
|
|
|
$
|
17,382
|
|
|
Accounts receivable, net
|
|
|
773
|
|
|
|
223
|
|
|
|
—
|
|
|
|
|
|
|
|
996
|
|
|
Due from parent
|
|
|
—
|
|
|
|
300
|
|
|
|
—
|
|
|
|
|
|
|
|
300
|
|
|
Inventory, net
|
|
|
567
|
|
|
|
5,359
|
|
|
|
—
|
|
|
|
|
|
|
|
5,926
|
|
|
Contractual assets
|
|
|
—
|
|
|
|
244
|
|
|
|
—
|
|
|
|
|
|
|
|
244
|
|
|
Prepaid expenses and other current assets
|
|
|
178
|
|
|
|
640
|
|
|
|
—
|
|
|
|
|
|
|
|
818
|
|
|
Total current assets
|
|
|
4,574
|
|
|
|
12,198
|
|
|
|
8,894
|
|
|
|
|
|
|
|
25,666
|
|
|
Property and equipment, net
|
|
|
198
|
|
|
|
4,249
|
|
|
|
—
|
|
|
|
|
|
|
|
4,447
|
|
|
Finance lease right-of-use assets, net
|
|
|
985
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
985
|
|
|
Operating lease right-of-use assets
|
|
|
1,382
|
|
|
|
1,475
|
|
|
|
—
|
|
|
|
|
|
|
|
2,857
|
|
|
Goodwill
|
|
|
601
|
|
|
|
—
|
|
|
|
5,058
|
|
|
|
H
|
|
|
|
5,659
|
|
|
Intangible assets, net
|
|
|
952
|
|
|
|
8,359
|
|
|
|
—
|
|
|
|
H
|
|
|
|
9,311
|
|
|
Deferred tax assets
|
|
|
—
|
|
|
|
17
|
|
|
|
—
|
|
|
|
|
|
|
|
17
|
|
|
Other noncurrent assets
|
|
|
241
|
|
|
|
180
|
|
|
|
—
|
|
|
|
|
|
|
|
421
|
|
|
Total assets
|
|
$
|
8,933
|
|
|
$
|
26,478
|
|
|
$
|
13,952
|
|
|
|
|
|
|
$
|
49,363
|
|
|
Liabilities and Stockholders’ Equity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current liabilities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable
|
|
$
|
1,259
|
|
|
$
|
831
|
|
|
$
|
—
|
|
|
|
|
|
|
$
|
2,090
|
|
|
Due to parent
|
|
|
—
|
|
|
|
211
|
|
|
|
—
|
|
|
|
|
|
|
|
211
|
|
|
Accrued compensation and benefits
|
|
|
1,191
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
1,191
|
|
|
Accrued and other current liabilities
|
|
|
821
|
|
|
|
1,232
|
|
|
|
5,236
|
|
|
|
A,B,K
|
|
|
|
7,289
|
|
|
Deferred revenue
|
|
|
10
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
10
|
|
|
Short-term notes payable
|
|
|
343
|
|
|
|
3,713
|
|
|
|
—
|
|
|
|
|
|
|
|
4,056
|
|
|
Short-term finance lease liabilities
|
|
|
576
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
576
|
|
|
Short-term operating lease liabilities
|
|
|
959
|
|
|
|
492
|
|
|
|
—
|
|
|
|
|
|
|
|
1,451
|
|
|
Total current liabilities
|
|
|
5,159
|
|
|
|
6,479
|
|
|
|
5,236
|
|
|
|
|
|
|
|
16,874
|
|
|
Notes payable
|
|
|
495
|
|
|
|
22,305
|
|
|
|
—
|
|
|
|
|
|
|
|
22,800
|
|
|
Long-term finance lease liabilities
|
|
|
380
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
380
|
|
|
Long-term operating lease liabilities
|
|
|
1,072
|
|
|
|
991
|
|
|
|
—
|
|
|
|
|
|
|
|
2,063
|
|
|
Other noncurrent liabilities
|
|
|
—
|
|
|
|
50
|
|
|
|
—
|
|
|
|
|
|
|
|
50
|
|
|
Total liabilities
|
|
|
7,106
|
|
|
|
29,825
|
|
|
|
5,236
|
|
|
|
|
|
|
|
42,167
|
|
|
Stockholders’ equity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Common stock
|
|
|
9
|
|
|
|
6,313
|
|
|
|
(6,275
|
)
|
|
|
E,F,J
|
|
|
|
47
|
|
|
Additional paid-in capital
|
|
|
170,358
|
|
|
|
184,042
|
|
|
|
(148,313
|
)
|
|
|
C,D,E,F,G,J
|
|
|
|
206,087
|
|
|
Accumulated deficit
|
|
|
(168,525
|
)
|
|
|
(193,632
|
)
|
|
|
163,289
|
|
|
|
A,B,C,K
|
|
|
|
(198,868
|
)
|
|
Accumulated other comprehensive loss
|
|
|
(15
|
)
|
|
|
(70
|
)
|
|
|
15
|
|
|
|
C
|
|
|
|
(70
|
)
|
|
Total stockholders’ equity (deficit)
|
|
|
1,827
|
|
|
|
(3,347
|
)
|
|
|
8,716
|
|
|
|
|
|
|
|
7,196
|
|
|
Total liabilities and stockholders’ equity
|
|
$
|
8,933
|
|
|
$
|
26,478
|
|
|
$
|
13,952
|
|
|
|
|
|
|
$
|
49,363
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
OpGen, Inc.
|
|
Unaudited Pro Forma Condensed
Combined Statement of Operations
|
|
For the six months ended June
30, 2019
|
|
(in thousands, except share
and per share data)
|
|
|
|
|
|
|
|
|
|
|
OpGen
|
|
|
|
Curetis
Business
|
|
|
|
Pro
Forma Adjustments (H)
|
|
|
|
Notes
|
|
|
|
Pro
Forma Combined
|
|
|
Revenue
|
|
$
|
2,030
|
|
|
$
|
1,237
|
|
|
$
|
—
|
|
|
|
|
|
|
$
|
3,267
|
|
|
Operating expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of products sold
|
|
|
419
|
|
|
|
1,525
|
|
|
|
—
|
|
|
|
|
|
|
|
1,944
|
|
|
Cost of services
|
|
|
396
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
396
|
|
|
Research and development
|
|
|
2,930
|
|
|
|
4,752
|
|
|
|
—
|
|
|
|
|
|
|
|
7,682
|
|
|
General and administrative
|
|
|
3,340
|
|
|
|
1,832
|
|
|
|
(130
|
)
|
|
|
I
|
|
|
|
5,042
|
|
|
Sales and marketing
|
|
|
766
|
|
|
|
3,717
|
|
|
|
—
|
|
|
|
|
|
|
|
4,483
|
|
|
Impairment of right-of-use
asset
|
|
|
521
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
521
|
|
|
Total operating expenses
|
|
|
8,372
|
|
|
|
11,826
|
|
|
|
(130
|
)
|
|
|
|
|
|
|
20,068
|
|
|
Operating loss
|
|
|
(6,342
|
)
|
|
|
(10,589
|
)
|
|
|
130
|
|
|
|
|
|
|
|
(16,801
|
)
|
|
Other (expense) income
|
|
|
(9
|
)
|
|
|
138
|
|
|
|
—
|
|
|
|
|
|
|
|
129
|
|
|
Interest expense
|
|
|
(94
|
)
|
|
|
(846
|
)
|
|
|
—
|
|
|
|
|
|
|
|
(940
|
)
|
|
Foreign currency transaction losses
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
—
|
|
|
Change in fair value of derivative financial instruments
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
—
|
|
|
Provision for income taxes
|
|
|
—
|
|
|
|
(64
|
)
|
|
|
—
|
|
|
|
|
|
|
|
(64
|
)
|
|
Net loss
|
|
$
|
(6,445
|
)
|
|
$
|
(11,361
|
)
|
|
$
|
130
|
|
|
|
|
|
|
$
|
(17,676
|
)
|
|
Net loss applicable to common stockholders
|
|
$
|
(6,445
|
)
|
|
$
|
(11,361
|
)
|
|
$
|
130
|
|
|
|
|
|
|
$
|
(17,676
|
)
|
|
Net loss per common share - basic and
diluted
|
|
$
|
(9.54
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
(3.92
|
)
|
|
Weighted average shares outstanding - basic and diluted
|
|
|
675,932
|
|
|
|
|
|
|
|
3,832,538
|
|
|
|
F,J
|
|
|
|
4,508,470
|
|
OpGen,
Inc.
Unaudited Pro Forma Condensed Combined Statement of Operations
For the year ended December 31, 2018
in
thousands, except share and per share data)
|
|
|
|
OpGen
|
|
|
|
Curetis
Business
|
|
|
|
Pro
Forma Adjustments (H)
|
|
|
|
Notes
|
|
|
Pro
Forma Combined
|
|
Revenue
|
|
$
|
2,946
|
|
|
$
|
1,623
|
|
|
$
|
—
|
|
|
|
|
|
|
$4,569
|
|
Operating expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of products sold
|
|
|
1,223
|
|
|
|
1,558
|
|
|
|
—
|
|
|
|
|
|
|
2,781
|
|
Cost of services
|
|
|
626
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
626
|
|
Research and development
|
|
|
5,677
|
|
|
|
12,085
|
|
|
|
—
|
|
|
|
|
|
|
17,762
|
|
General and administrative
|
|
|
7,069
|
|
|
|
4,092
|
|
|
|
—
|
|
|
|
|
|
|
11,161
|
|
Sales and marketing
|
|
|
1,532
|
|
|
|
9,318
|
|
|
|
—
|
|
|
|
|
|
|
10,850
|
|
Total operating expenses
|
|
|
16,127
|
|
|
|
27,053
|
|
|
|
—
|
|
|
|
|
|
|
43,180
|
|
Operating loss
|
|
|
(13,181
|
)
|
|
|
(25,430
|
)
|
|
|
—
|
|
|
|
|
|
|
(38,611)
|
|
Other income
|
|
|
5
|
|
|
|
754
|
|
|
|
—
|
|
|
|
|
|
|
759
|
|
Interest expense
|
|
|
(191
|
)
|
|
|
(1,374
|
)
|
|
|
—
|
|
|
|
|
|
|
(1,565)
|
|
Foreign currency transaction losses
|
|
|
(10
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
(10)
|
|
Change in fair value of derivative financial instruments
|
|
|
8
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
8
|
|
Provision for income taxes
|
|
|
—
|
|
|
|
(41
|
)
|
|
|
—
|
|
|
|
|
|
|
(41)
|
|
Net loss
|
|
$
|
(13,369
|
)
|
|
$
|
(26,091
|
)
|
|
$
|
—
|
|
|
|
|
|
|
$(39,460)
|
|
Net loss applicable to common stockholders
|
|
$
|
(13,369
|
)
|
|
$
|
(26,091
|
)
|
|
$
|
—
|
|
|
|
|
|
|
$(39,460)
|
|
Net loss per common share - basic and
diluted
|
|
$
|
(44.69
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$(9.55)
|
|
Weighted average shares outstanding - basic and diluted
for the year ended December 31, 2018
|
|
|
300,453
|
|
|
|
|
|
|
|
3,832,538
|
|
|
|
F,J
|
|
|
4,132,991
|
NOTES TO THE UNAUDITED
PRO FORMA CONDENSED COMBINED FINANCIAL INFORMATION
|
|
1.
|
Description
of Transaction
|
As announced on September
4, 2019, OpGen and Curetis N.V. entered into the Implementation Agreement. Under the Implementation Agreement, OpGen has agreed
to purchase all of the outstanding shares and acquire all of the related business assets of the Curetis Business to create a combined
business within OpGen, which we refer to as “Newco” in this prospectus.
Pursuant to the Implementation
Agreement, OpGen will acquire (i) all of the issued and outstanding capital stock, or the Transferred Shares of Curetis GmbH,
and (ii) all of the assets of Curetis N.V. that are solely and exclusively related to the business of Curetis, or the Transferred
Assets. OpGen has also agreed to assume (1) the 2016 Stock Option Plan and the outstanding awards thereunder, (2) the obligation
to issue equity to the holders of awards under the PSOP, and (3) the outstanding indebtedness of Curetis N.V. under certain Curetis
Convertible Notes, including providing for conversion of such Curetis Convertible Notes into shares of OpGen’s common stock.
OpGen will also assume all of the liabilities of Curetis N.V. solely and exclusively related to the Curetis Business.
Under the Implementation Agreement,
OpGen will issue, as the sole consideration, 2,662,564 shares of common stock, less the number of shares of common stock the issuance
of which shall be reserved by OpGen in connection with (a) its assumption of the 2016 Stock Option Plan, (b) any future issuance
of shares of common stock under the PSOP, and (c) shares of common stock reserved for future issuance upon the conversion, if
any, of the Curetis Convertible Notes, or together, the Consideration. The number of shares of common stock to be reserved for
the deductions described above are based on a conversion ratio of 0.0959, which is the ratio of the Consideration as contrasted
with the number of the Curetis N.V. ordinary shares on a fully diluted basis. The Consideration is equal to 72.5% of the common
stock of OpGen on a fully diluted basis as of the date of the Implementation Agreement.
In the Implementation Agreement,
OpGen has committed to raise at least $10,000,000 of interim equity financing to support the continuing operations of both OpGen
and the Curetis Business. The offering contemplated by this prospectus is the interim equity financing. We intend to use certain
proceeds from this offering to support the operations of each of OpGen and the Curetis Business during the period between signing
and closing of the transactions under the Implementation Agreement, and to support the combined operations of Newco after the
closing occurs. See “Use of Proceeds.”
The accompanying unaudited
pro forma condensed combined financial statements reflect, what the Company assumes would be the results and financial position
on a US GAAP basis, of the combined financial statements of the Curetis Business prepared in accordance with IFRS as issued by
the IASB. OpGen has performed a preliminary analysis and has not identified significant differences identified between IFRS and
U.S. GAAP for the purposes of presenting the unaudited pro forma condensed combined financial statements. In addition, the unaudited
condensed combined financial statements reflect reclassifications to conform the Curetis Business historical accounting presentation
to OpGen’s accounting presentation and translation from Euros to USD based on OpGen’s U.S. GAAP policies as follows.
Unaudited
Curetis Business Condensed Combined Statement of Financial Position
June 30, 2019
(in thousands)
|
|
|
Curetis
Business
(Euros)
|
|
Curetis
Business
(USD)
(1)
|
|
Assets
|
|
|
|
|
|
|
|
|
|
Current assets
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
€
|
4,779
|
|
|
$
|
5,432
|
|
|
Trade receivables
|
|
|
196
|
|
|
|
223
|
|
|
Other receivables, related party
|
|
|
264
|
|
|
|
300
|
|
|
Contractual assets
|
|
|
215
|
|
|
|
244
|
|
|
Inventories
|
|
|
4,715
|
|
|
|
5,359
|
|
|
Prepaid expenses and other current assets
|
|
|
563
|
|
|
|
640
|
|
|
Total current assets
|
|
|
10,732
|
|
|
|
12,198
|
|
|
Intangible assets
|
|
|
7,354
|
|
|
|
8,359
|
|
|
Property, plant and equipment
|
|
|
3,738
|
|
|
|
4,249
|
|
|
Right of use assets
|
|
|
1,298
|
|
|
|
1,475
|
|
|
Other non-current financial assets
|
|
|
158
|
|
|
|
180
|
|
|
Deferred tax assets
|
|
|
15
|
|
|
|
17
|
|
|
Total assets
|
|
€
|
23,295
|
|
|
$
|
26,478
|
|
|
Liabilities and
Stockholders’ Equity
|
|
|
|
|
|
|
|
|
|
Current liabilities
|
|
|
|
|
|
|
|
|
|
Trade and other payables
|
|
€
|
731
|
|
|
$
|
831
|
|
|
Other liabilities, related party
|
|
|
186
|
|
|
|
211
|
|
|
Provisions current
|
|
|
130
|
|
|
|
148
|
|
|
Tax liabilities
|
|
|
2
|
|
|
|
2
|
|
|
Other current liabilities
|
|
|
952
|
|
|
|
1,082
|
|
|
Other current financial liabilities
|
|
|
3,267
|
|
|
|
3,713
|
|
|
Current lease liabilities
|
|
|
433
|
|
|
|
492
|
|
|
Total current liabilities
|
|
|
5,701
|
|
|
|
6,479
|
|
|
Provisions non-current
|
|
|
44
|
|
|
|
50
|
|
|
Other non-current financial liabilities
|
|
|
19,623
|
|
|
|
22,305
|
|
|
Non-current lease liabilities
|
|
|
872
|
|
|
|
991
|
|
|
Total liabilities
|
|
|
26,240
|
|
|
|
29,825
|
|
|
Equity
|
|
|
|
|
|
|
|
|
|
Subscribed capital
|
|
|
5,554
|
|
|
|
6,313
|
|
|
Capital reserve
|
|
|
161,913
|
|
|
|
184,042
|
|
|
Currency translation differences
|
|
|
(62
|
)
|
|
|
(70
|
)
|
|
Retained earnings
|
|
|
(170,350
|
)
|
|
|
(193,632
|
)
|
|
Total stockholders’
equity
|
|
|
(2,945
|
)
|
|
|
(3,347
|
)
|
|
Total liabilities and
stockholders’ equity
|
|
€
|
23,295
|
|
|
$
|
26,478
|
|
|
|
|
|
|
|
|
|
|
|
|
(1) Convenience translation performed using the following exchange
rate in effect as of June 30, 2019:
|
|
|
1.13667
|
|
|
|
|
|
|
|
|
|
|
|
Unaudited Curetis Business Condensed
Combined Statement of Operations
|
|
|
For the six months ended June
30, 2019
|
|
|
(in thousands, except share and
per share data)
|
|
|
|
|
|
|
|
|
|
|
|
|
Curetis
Business
(Euros)
|
|
|
|
Curetis
Business
(USD)
(1)
|
|
|
Revenue
|
|
€
|
1,088
|
|
|
$
|
1,237
|
|
|
Cost of sales
|
|
|
(1,342
|
)
|
|
|
(1,525
|
)
|
|
Gross loss
|
|
|
(254
|
)
|
|
|
(288
|
)
|
|
Distribution costs
|
|
|
(3,270
|
)
|
|
|
(3,717
|
)
|
|
Administrative expenses
|
|
|
(1,612
|
)
|
|
|
(1,832
|
)
|
|
Research & development expenses
|
|
|
(4,181
|
)
|
|
|
(4,752
|
)
|
|
Other income
|
|
|
114
|
|
|
|
130
|
|
|
Operating loss
|
|
|
(9,203
|
)
|
|
|
(10,459
|
)
|
|
Finance income
|
|
|
7
|
|
|
|
8
|
|
|
Finance costs
|
|
|
(744
|
)
|
|
|
(846
|
)
|
|
Finance result - net
|
|
|
(737
|
)
|
|
|
(838
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Loss before income taxes
|
|
|
(9,940
|
)
|
|
|
(11,297
|
)
|
|
Income tax expense
|
|
|
(56
|
)
|
|
|
(64
|
)
|
|
Loss for the period
|
|
€
|
(9,996
|
)
|
|
$
|
(11,361
|
)
|
|
|
|
|
|
|
|
|
|
|
|
(1) Convenience translation performed using the following exchange
rate in effect as of June 30, 2019
|
|
|
1.13667
|
|
|
|
|
|
|
|
|
|
|
|
Unaudited Curetis Business Condensed
Combined Statement of Operations
|
|
|
For the year ended December 31,
2018
|
|
|
(in thousands, except share and
per share data)
|
|
|
|
|
|
|
|
|
|
|
|
Curetis
Business
(Euros)
|
|
|
|
Curetis
Business
(USD) (1)
|
|
|
Revenue
|
|
€
|
1,419
|
|
|
$
|
1,623
|
|
|
Cost of sales
|
|
|
(1,362
|
)
|
|
|
(1,558
|
)
|
|
Gross loss
|
|
|
57
|
|
|
|
65
|
|
|
Distribution costs
|
|
|
(8,147
|
)
|
|
|
(9,318
|
)
|
|
Administrative expenses
|
|
|
(3,578
|
)
|
|
|
(4,092
|
)
|
|
Research & development expenses
|
|
|
(10,566
|
)
|
|
|
(12,085
|
)
|
|
Other income
|
|
|
625
|
|
|
|
715
|
|
|
Operating loss
|
|
|
(21,609
|
)
|
|
|
(24,715
|
)
|
|
Finance income
|
|
|
34
|
|
|
|
39
|
|
|
Finance costs
|
|
|
(1,201
|
)
|
|
|
(1,374
|
)
|
|
Finance result - net
|
|
|
(1,167
|
)
|
|
|
(1,335
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Loss before income taxes
|
|
|
(22,776
|
)
|
|
|
(26,050
|
)
|
|
Income tax expense
|
|
|
(36
|
)
|
|
|
(41
|
)
|
|
Loss for the period
|
|
€
|
(22,812
|
)
|
|
$
|
(26,091
|
)
|
|
|
|
|
|
|
|
|
|
|
|
(1) Convenience translation performed using the following exchange
rate in effect as of December 31, 2018
|
|
|
1.14379
|
|
|
|
|
|
The
accompanying unaudited pro forma condensed combined financial statements reflect Curetis Business unaudited combined financial
statements prepared in accordance with IFRS mapped to OpGen’s financial statements as follows:
|
Curetis
Business Statement of Operations Descriptions
|
|
OpGen
Consolidated Statements of Operations and Comprehensive Loss Descriptions
|
|
|
Revenue
|
|
Revenue
|
|
|
Cost
of sales
|
|
Cost
of products sold
|
|
|
Distribution
costs
|
|
Sales
and marketing
|
|
|
Administrative
expenses
|
|
General
and administrative
|
|
|
Research
& development expenses
|
|
Research
and development
|
|
|
Other
income
|
|
Other
income
|
|
|
Operating
loss
|
|
Operating
loss
|
|
|
Finance
income
|
|
Other
income
|
|
|
Finance
costs
|
|
Interest
expense
|
|
|
Income
tax expense
|
|
Provision
for income taxes
|
|
|
Loss
for the period
|
|
Net
loss
|
|
|
Curetis
Business Statement of Financial Position Descriptions
|
|
OpGen
Balance Sheet Descriptions
|
|
|
Cash
and cash equivalents
|
|
Cash
and cash equivalents
|
|
|
Trade
receivables
|
|
Accounts
receivable, net
|
|
|
Other
receivables, related party
|
|
Due
from parent
|
|
|
Contractual
assets
|
|
Contractual
assets *
|
|
|
Inventories
|
|
Inventory,
net
|
|
|
Prepaid
Expenses and other current assets
|
|
Prepaid
expenses and other current assets
|
|
|
Total
current assets
|
|
Total
current assets
|
|
|
Intangible
assets
|
|
Intangible
assets, net
|
|
|
Property,
plant and equipment
|
|
Property
and equipment, net
|
|
|
Right
of use assets
|
|
Operating
lease right-of-use assets
|
|
|
Other
non-current assets
|
|
Other
noncurrent assets
|
|
|
Other
non-current financial assets
|
|
Other
noncurrent assets
|
|
|
Deferred
tax assets
|
|
Deferred
tax assets
|
|
|
Total
assets
|
|
Total
assets
|
|
|
Liabilities
and Stockholders’ Equity
|
|
Liabilities
and Stockholders’ Equity
|
|
|
Current
liabilities
|
|
Current
liabilities
|
|
|
Trade
and other payables
|
|
Accounts
payable
|
|
|
Other
liabilities, related party
|
|
Due
to parent
|
|
|
Provisions
current
|
|
Provisions
current
|
|
|
Tax
liabilities
|
|
Accrued
and other current liabilities
|
|
|
Other
current liabilities
|
|
Accrued
and other current liabilities
|
|
|
Other
current financial liabilities
|
|
Accrued
and other current liabilities
|
|
|
Current
lease liabilities
|
|
Short-term
operating lease liabilities
|
|
|
Total
current liabilities
|
|
Total
current liabilities
|
|
|
Provisions
non-current
|
|
Other
noncurrent liabilities*
|
|
|
Other
non-current financial liabilities
|
|
Notes
payable
|
|
|
Non-current
lease liabilities
|
|
Long-term
operating lease liabilities
|
|
|
Total
liabilities
|
|
Total
liabilities
|
|
|
Equity
|
|
Equity
|
|
|
Subscribed
capital
|
|
Common
stock
|
|
|
Capital
reserve
|
|
Additional
paid-in capital
|
|
|
Currency
translation differences
|
|
Accumulated
other comprehensive loss
|
|
|
Accumulated
deficit
|
|
Accumulated
deficit
|
|
|
Total
stockholders’ equity
|
|
Total
stockholders’ equity (deficit)
|
|
|
Total
liabilities and stockholders’ equity
|
|
Total
liabilities and stockholders’ equity
|
|
|
|
|
|
|
|
*
Denotes Curetis description added to pro forma balance sheet
|
|
|
|
3.
Estimated Purchase Price
The
accompanying unaudited pro forma condensed combined financial statements reflect an estimated acquisition price of approximately
$15.8 million based on the current OpGen stock price and the number of shares to be issued as Consideration. Given that the estimated
purchase price is variable depending upon the price of OpGen’s common stock, management performed a sensitivity analysis
over the change in purchase consideration based on +/– 10% fluctuation in OpGen's stock price. An increase or decrease in
the price of OpGen’s common stock by 10% would increase or decrease the purchase consideration by approximately $1.58 million.
The
total estimated purchase price and allocated purchase price is summarized as follows (in thousands, except share and per share
data):
|
Number of OpGen shares currently outstanding
|
|
|
882,268
|
|
|
Number of shares to be issued in public
offering (i)
|
|
|
1,526,717
|
|
|
Total number of shares of the combined company to be owned by
OpGen's stockholders
|
|
|
2,408,985
|
|
|
Multiplied by the fair market value per
share of OpGen's common stock(ii)
|
|
$
|
6.55
|
|
|
Total
|
|
$
|
15,779
|
|
|
|
(i)
|
1,526,717
potential units to be issued to raise gross proceeds of $10,000,000 as required by the
Implementation Agreement. The 1,526,717 potential units are based on the $6.55 per share
closing price of OpGen’s common stock on October 9, 2019. Note that each $1.00
per unit increase or decrease in the final offering price of OpGen’s equity securities
results in a 202,214 decrease or a 275,084 increase in the units issued, respectively.
Assumes no pre-funded units are issued.
|
|
|
(ii)
|
The
estimated purchase price was based on the closing price as reported on the Nasdaq Capital
Market on October 9, 2019. The final purchase price arising from the actual fair
market value of OpGen common stock outstanding immediately prior to the closing of the
business combination could result in a total purchase price different from that assumed
in this unaudited pro forma condensed combined financial information, and that difference
may be material. Therefore, the estimated consideration expected to be transferred reflected
in this unaudited pro forma condensed combined financial information does not purport
to represent what the actual consideration transferred will be when the business combination
is completed. The actual purchase price will fluctuate until the closing date of the
business combination, and the final valuation of the purchase consideration could differ
significantly from the current estimate.
|
For
purposes of this pro forma analysis, the above estimated purchase price has been allocated based on a preliminary estimate of
the fair value of assets and liabilities to be acquired.
|
Net assets as of June 30, 2019
|
|
|
$
|
10,721
|
|
|
Goodwill
|
|
|
|
5,058
|
|
|
Total
|
|
|
$
|
15,779
|
|
4.
Pro Forma Adjustments
Adjustments
included in the column under the heading "Pro forma Adjustments" are primarily based on information contained within
the Implementation Agreement. Further analysis will be performed after the completion of the business combination to confirm the
necessity of these estimates.
The
pro forma adjustments relate to the following:
|
|
A.
|
To
record OpGen's estimated transaction costs, such as legal, audit, advisory fees and transactional
fees that were not incurred as of June 30, 2019.
|
|
|
B.
|
To
record the Curetis Business's estimated transaction costs, such as legal, audit, advisory
fees and transactional fees that were not incurred as of June 30, 2019.
|
|
|
C.
|
To
eliminate OpGen’s historical paid-in-capital, accumulated other comprehensive loss,
and accumulated deficit balances.
|
|
|
D.
|
To
reflect the fair value of the number of shares of the combined company to be owned by
OpGen's stockholders, other than Curetis N.V.
|
|
|
E.
|
To
transfer the pre-business combination par value of common stock held by Curetis N.V.
from common stock to paid in capital.
|
|
|
F.
|
To
reflect shares to be issued to Curetis N.V. at closing of the business combination, excluding
potential shares the issuance of which shall be reserved by OpGen in connection with
(a) its assumption of the 2016 Stock Option Plan, (b) any future issuance of shares of
common stock under the PSOP, and (c) shares of common stock reserved for future issuance
upon the conversion, if any, of the Curetis Convertible Notes.
|
|
|
G.
|
To
reflect the par value of OpGen's remaining stock post-business combination.
|
|
|
H.
|
To
reflect the fair value of OpGen’s remaining stock post-business combination in
excess of the net assets acquired by Curetis Business. OpGen’s intangible assets
have not yet been determined and, therefore, the allocation of the purchase price in
excess of OpGen’s net assets is shown entirely as goodwill. Separately identifiable
intangible assets may be determined to exist that may require amortization expense to
be recognized in future periods.
|
|
|
I.
|
To
reflect transaction costs incurred by OpGen and the Curetis Business during the six months
ended June 30, 2019.
|
|
|
J.
|
To
record the receipt of $10,000,000 in gross proceeds, $8,894,000 estimated net proceeds, in cash for the issuance
of 1,526,717 units in a public offering (2,408,985 shares of common stock will be outstanding following such offering assuming
the underwriter does not exercise its option to purchase additional shares), based on an assumed offering price of $6.55 per unit,
equal to the closing price of OpGen’s common stock on October 9, 2019. Assuming the amount of gross proceeds to be raised
remains $10,000,000, note that a $1.00 per unit increase or decrease in the final offering price of units results in a 202,214
decrease or a 275,084 increase, respectively, in the number of units issued. The Implementation Agreement requires the completion
of an equity offering of at least $10,000,000. Note that each $500,000 increase or decrease in the final gross proceeds results
in a 76,335 increase or decrease in the number of units issued, respectively, based on an assumed offering price of $6.55 per unit,
equal to the closing price of OpGen’s common stock on October 9, 2019. Assumes that no pre-funded units are sold.
|
|
|
K.
|
To
record retention payments to be made to OpGen executives with change in control.
|
Adjustments to accrued expenses are as follows (in thousands):
|
|
|
June
30, 2019
|
|
|
OpGen’s
estimated transaction costs (A)
|
|
$
|
1,606
|
|
|
Curetis Business
estimated transaction costs (B)
|
|
|
2,841
|
|
|
Retention benefits
to be paid to OpGen executives (K)
|
|
|
789
|
|
|
Total
|
|
$
|
5,236
|
|
Adjustments to additional paid-in capital are as follows
(in thousands):
|
|
|
June
30, 2019
|
|
|
Eliminate
OpGen’s historical additional paid-in-capital balance (C)
|
|
$
|
(170,358
|
)
|
|
To reflect the
fair value of OpGen’s remaining stock post-business combination (D)
|
|
|
15,779
|
|
|
To reflect Curetis
N.V. common stock outstanding pre-business combination (E)
|
|
|
6,313
|
|
|
To reflect shares
to be issued to Curetis N.V. at closing of the business combination (F)
|
|
|
(23
|
)
|
|
To reflect the
par value of OpGen’s remaining stock post-business combination (G)
|
|
|
(9
|
)
|
|
To reflect shares
to be issued to purchasers in a $10 million public offering, net of offering costs (J)
|
|
|
(15
|
)
|
|
Total
|
|
$
|
(148,313
|
)
|
Adjustments to common stock are as follows (in thousands):
|
|
|
June
30, 2019
|
|
|
Eliminate
Curetis N.V. common stock outstanding pre-business combination (E)
|
|
$
|
(6,313
|
)
|
|
To reflect shares
to be issued to Curetis N.V. at closing of the business combination (F)
|
|
|
23
|
|
|
To reflect shares
to be issued to purchasers in a $10 million public offering, net of offering costs (J)
|
|
|
15
|
|
|
Total
|
|
$
|
(6,275
|
)
|
Adjustments to accumulated deficit are as follows (in thousands):
|
|
|
June
30, 2019
|
|
|
OpGen’s
estimated transaction costs (A)
|
|
$
|
(1,606
|
)
|
|
Curetis Business
estimated transaction costs (B)
|
|
|
(2,841
|
)
|
|
Eliminate OpGen’s
historical accumulated deficit balance (C)
|
|
|
168,525
|
|
|
Retention benefits
to be paid to OpGen executives (K)
|
|
|
(789
|
)
|
|
Total
|
|
$
|
163,289
|
|
RISK
FACTORS
Investing in our securities
involves a high degree of risk. You should consider carefully the risks and uncertainties described below, and incorporated by
reference herein, together with all of the other information in, or incorporated by reference in, this prospectus, including our
financial statements and related notes incorporated by reference herein, before making an investment decision. If any of these
risks occur, our business, financial condition, results of operations and prospects could be materially and adversely affected.
In that event, the trading price of our common stock could decline and you could lose part or all of your investment.
Risks
Related to this Offering and Our Securities
We
need to raise capital in this offering to support our operations. If the offering is not successful, our financial position will
be materially adversely impacted.
We have incurred substantial
losses since our inception, and we expect to continue to incur additional losses for the next several years. For the six months
ended June 30, 2019, we had a net loss of $6.5 million. From our inception through June 30, 2019, we had an accumulated deficit
of $168.5 million. We believe that current cash on hand will be sufficient to fund operations into October 2019. In addition,
the report of our independent registered public accounting firm on our financial statements for the years ended December 31,
2018 and 2017 contains explanatory language that substantial doubt exists about our ability to continue as a going concern. In
the event we are unable to successfully raise sufficient capital in this offering, we will not have sufficient cash flows and
liquidity to finance our business operations as currently contemplated. Accordingly, in such circumstances we would be compelled
to reduce general and administrative expenses and delay research and development projects, including the purchase of scientific
equipment and supplies, until we are able to obtain sufficient financing. We have no additional committed sources of capital and
may find it difficult to raise money on terms favorable to us or at all. The failure to obtain sufficient capital to support our
operations would have a material adverse effect on our business, financial condition and results of operations. If such sufficient
financing is not received timely, we would then need to pursue a plan to license or sell assets, seek to be acquired by another
entity, cease operations and/or seek bankruptcy protection.
We received a deficiency notice
from the Nasdaq Capital Market. If we are unable to cure this deficiency and meet the Nasdaq continued listing requirements, we
could be delisted from the Nasdaq Capital Market, which would negatively impact the trading of our common stock.
On August 19, 2019, OpGen received
a written notification from Nasdaq notifying the Company that it has failed to comply with Nasdaq Marketplace Rule 5550(b)(1)
because the Company’s stockholders’ equity as of June 30, 2019 fell below the required minimum of $2,500,000, and
as of June 30, 2019, the Company did not meet the alternative compliance standards of market value of listed securities or net
income from continuing operations for continued listing. We submitted a plan to Nasdaq to regain compliance with the Nasdaq minimum
stockholders’ equity standard on October 3, 2019, which plan included information regarding this offering. We believe that
if this offering is successful, we will regain compliance with the minimum stockholders’ equity standard for continued listing.
However, there can be no assurance that the Company’s plan will be accepted or that if it is, the Company will be able to
regain compliance.
If our common stock is delisted by Nasdaq, our common stock
may be eligible for quotation on an over-the-counter quotation system or on the pink sheets. Upon any such delisting, our common
stock would become subject to the regulations of the SEC relating to the market for penny stocks. A penny stock is any equity security
not traded on a national securities exchange that has a market price of less than $5.00 per share. The regulations applicable to
penny stocks may severely affect the market liquidity for our common stock and could limit the ability of stockholders to sell
securities in the secondary market. In such a case, an investor may find it more difficult to dispose of or obtain accurate quotations
as to the market value of our common stock, and there can be no assurance that our common stock will be eligible for trading or
quotation on any alternative exchanges or markets.
If our common stock is delisted
by Nasdaq, our common stock may be eligible for quotation on an over-the-counter quotation system or on the pink sheets. Upon
any such delisting, our common stock would become subject to the regulations of the SEC relating to the market for penny stocks.
A penny stock is any equity security not traded on a national securities exchange that has a market price of less than $5.00 per
share. The regulations applicable to penny stocks may severely affect the market liquidity for our common stock and could limit
the ability of stockholders to sell securities in the secondary market. In such a case, an investor may find it more difficult
to dispose of or obtain accurate quotations as to the market value of our common stock, and there can be no assurance that our
common stock will be eligible for trading or quotation on any alternative exchanges or markets.
Delisting from Nasdaq could
adversely affect our ability to raise additional financing through public or private sales of equity securities, would significantly
affect the ability of investors to trade our securities and would negatively affect the value and liquidity of our common stock.
Delisting could also have other negative results, including the potential loss of confidence by employees, the loss of institutional
investor interest and fewer business development opportunities.
Management
will have broad discretion as to the use of the net proceeds from this offering, and we may not use the proceeds effectively.
Our management will have broad
discretion as to the application of the net proceeds and intends to use them to support the operations of both of OpGen and the
Curetis Group during the period prior to the closing of the transaction under the Implementation Agreement, and then, if any proceeds
remain, for the operations of Newco if the transactions pursuant to the Implementation Agreement close. Our stockholders may not
agree with the manner in which our management chooses to allocate and spend the net proceeds. Moreover, our management may use
the net proceeds for corporate purposes that may not increase our results of operations or the market value of our common stock.
Our failure to apply these funds effectively could have a material adverse effect on our business, delay the development and approval
of our products and cause the price of our common stock to decline.
If
you purchase our securities sold in this offering, you will experience immediate dilution as a result of this offering.
Because the effective price per unit being offered may
be higher than the net tangible book value per share of our common stock, you will experience dilution to the extent of the difference
between the effective offering price per unit you pay in this offering and the net tangible book value per share of our common
stock immediately after this offering. Our net tangible book value as of June 30, 2019, was approximately $274 thousand, or $0.31
per share of common stock. Net tangible book value per share is equal to our total tangible assets minus total liabilities, all
divided by the number of shares of common stock outstanding. See “Dilution” on page 67 of this prospectus for a more
detailed discussion of the dilution you will incur in this offering.
If
you purchase our securities in this offering you may experience future dilution as a result of future equity offerings or other
equity issuances.
In order to raise additional capital, we believe that
we will offer and issue additional shares of our common stock or other securities convertible into or exchangeable for our common
stock in the future. We cannot assure you that we will be able to sell shares or other securities in any other offering at a price
per share that is equal to or greater than the price per unit paid by purchasers in this offering, and investors purchasing other
securities in the future could have rights superior to existing stockholders. The price per share at which we sell additional
shares of our common stock or other securities convertible into or exchangeable for our common stock in future transactions may
be higher or lower than the price per unit in this offering.
In addition, we have a significant
number of stock options, restricted stock units and warrants outstanding and anticipate that we will grant additional equity awards
following the closing of the Implementation Agreement transaction. To the extent that outstanding stock options or warrants have
been or may be exercised or other shares issued, you may experience further dilution. Further, we may choose to raise additional
capital due to market conditions or strategic considerations even if we believe we have sufficient funds for our current or future
operating plans.
The market price of
our common stock has been, and may continue to be, highly volatile, and such volatility could cause the market price of our common
stock to decrease and could cause you to lose some or all of your investment in our common stock.
During the period from
our initial public offering in May 2015 through October 9, 2019, the market price of our common stock fluctuated from a high of
$2,720.00 per share to a low of $4.20 per share, and our stock price continues to fluctuate. The market price of our common stock
may continue to fluctuate significantly in response to numerous factors, some of which are beyond our control, such as:
|
|
·
|
our
ability to grow our revenue and customer base;
|
|
|
·
|
the
announcement of new products or product enhancements by us or our competitors;
|
|
|
·
|
the
trading volume of our common stock;
|
|
|
·
|
developments
concerning regulatory oversight and approvals;
|
|
|
·
|
variations
in our and our competitors’ results of operations;
|
|
|
·
|
changes
in earnings estimates or recommendations by securities analysts, if our common stock
is covered by analysts;
|
|
|
·
|
successes
or challenges in our collaborative arrangements or alternative funding sources;
|
|
|
·
|
developments
in the health care and life science industries;
|
|
|
·
|
the
results of product liability or intellectual property lawsuits;
|
|
|
·
|
future
issuances of common stock or other securities;
|
|
|
·
|
the
addition or departure of key personnel;
|
|
|
·
|
announcements
by us or our competitors of acquisitions, investments or strategic alliances; and
|
|
|
·
|
general
market conditions and other factors, including factors unrelated to our operating performance.
|
Further, the stock market
in general, and the market for health care and life science companies in particular, has recently experienced extreme price and
volume fluctuations. The volatility of our common stock is further exacerbated due to its low trading volume. Continued market
fluctuations could result in extreme volatility in the price of our common stock, which could cause a decline in the value of
our common stock and the loss of some or all of your investment.
Trading
of our common stock is limited, and trading restrictions imposed on us by applicable regulations may further reduce trading in
our common stock, making it difficult for our stockholders to sell their shares; and future sales of common stock could reduce
our stock price.
Trading of our common stock
is currently conducted on the Nasdaq Capital Market. The liquidity of our common stock is limited, not only in terms of the number
of shares that can be bought and sold at a given price, but also as it may be adversely affected by delays in the timing of transactions
and reduction in security analysts’ and the media’s coverage of us, if any. These factors may result in different
prices for our common stock than might otherwise be obtained in a more liquid market and could also result in a larger spread
between the bid and asked prices for our common stock. In addition, without a large public float, our common stock is less liquid
than the stock of companies with broader public ownership, and, as a result, the trading prices of our common stock may be more
volatile. In the absence of an active public trading market, an investor may be unable to liquidate his investment in our common
stock. Trading of a relatively small volume of our common stock may have a greater impact on the trading price of our stock than
would be the case if our public float were larger. We cannot predict the prices at which our common stock will trade in the future,
if at all.
The
exercise of outstanding common stock purchase warrants and stock options will have a dilutive effect on the percentage ownership
of our capital stock by existing stockholders.
As of June 30, 2019, we had
outstanding warrants to acquire 175,982 shares of our common stock, and stock options to purchase 10,542 shares of our
common stock. The expiration of the terms of such options and warrants range from November 2019 to June 2028. A significant number
of such warrants are out of the money, but the holders have the right to effect a cashless exercise of such warrants. If a significant
number of such warrants and stock options are exercised by the holders, the percentage of our common stock owned by our existing
stockholders will be diluted.
We
have never paid dividends on our capital stock, and we do not anticipate paying dividends in the foreseeable future.
We have never paid dividends
on any of our capital stock and currently intend to retain any future earnings to fund the growth of our business. In addition,
an amended and restated promissory note issued in June 2017 to Merck Global Health Innovation Fund, a principal investor, or the
MGHIF Note, and the related security agreement restricts our ability to pay cash dividends on our common stock. We may also enter
into credit agreements or other borrowing arrangements in the future that will restrict our ability to declare or pay cash dividends
on our common stock. Any determination to pay dividends in the future will be at the discretion of our Board of Directors and
will depend on our financial condition, operating results, capital requirements, general business conditions and other factors
that our Board of Directors may deem relevant. As a result, capital appreciation, if any, of our common stock will be the sole
source of gain, if any, for the foreseeable future.
There is no public market
for the pre-funded warrants or the common warrants being offered by us in this offering.
There is no established public trading market for the
pre-funded warrants or the common warrants and we do not expect a market to develop. In addition, we do not intend to apply to
list the pre-funded warrants or the common warrants on any national securities exchange or other nationally recognized trading
system, including the Nasdaq Capital Market. Without an active market, the liquidity of the pre-funded warrants and the common
warrants will be limited.
The common warrants are
speculative in nature.
The common warrants do not confer any rights of common
stock ownership on its holders, such as voting rights or the right to receive dividends, but rather merely represent the right
to acquire shares of common stock at a fixed price for a limited period of time. Specifically, commencing on the date of issuance,
holders of the common warrants may exercise their right to acquire the common stock and pay an exercise price of $
per share, subject to certain adjustments, prior to five years from the date of issuance, after which date any unexercised common
warrants will expire and have no further value. Moreover, following this offering, the market value of the common warrants, if
any, is uncertain and there can be no assurance that the market value of the common warrants will equal or exceed their imputed
offering price. The common warrants will not be listed or quoted for trading on any market or exchange. There can be no assurance
that the market price of the common stock will ever equal or exceed the exercise price of our common warrants, and consequently,
whether it will ever be profitable for holders of the common warrants to exercise the common warrants.
Risks
Related to the Pending Business Combination Transaction with Curetis
We may not be
successful in consummating the proposed business combination transaction with Curetis, which failure could have a material adverse
effect on us.
The proposed combination with
Curetis is subject to the approval of our stockholders and by the shareholders and debt holders of Curetis N.V. and Curetis GmbH,
and we cannot provide any assurance that such approvals will be obtained. If the proposed transaction is not approved by our stockholders
at the special meeting called to approve the Newco transaction, we may become liable to reimburse Curetis N.V. for its expenses
up to a maximum amount of $250,000. We may also be liable to pay Curetis N.V. a termination fee of $500,000 if our board of directors
changes its recommendation to approve the proposed transaction or, if following a refusal by our stockholders to approve the proposed
transaction at the special meeting, we enter into a definitive agreement implementing an alternative transaction with a third
party. Curetis N.V. has undertaken the same obligations with respect to us if the shareholders of Curetis N.V. do not approve
the proposed transaction or if the boards of Curetis N.V. change their recommendation to approve the proposed transaction. In
case of termination of the Implementation Agreement in accordance with its terms, Curetis N.V. would also be required to repay
us under the Interim Facility, and we would need to re-focus our attention on OpGen as a stand-alone business. Any of these events
could have a material adverse impact on our financial condition.
Completion of the Newco transaction
is subject to the fulfillment or the waiver, as the case may be, of a number of conditions precedent, which may prevent, delay,
hinder or otherwise adversely affect the proposed business combination transaction.
Completion of the Newco transaction
is subject to the fulfillment or the waiver, as the case may be, of a number of conditions precedent as described in the Implementation
Agreement. These include, in addition to customary closing conditions, the necessary shareholder approvals by the requisite majority
of shareholders of Curetis N.V. and the stockholders of OpGen; the completion by OpGen of an interim equity financing of at least
$10,000,000 to support the continuing operations of both the Company and the Curetis Group; the documentation implementing the
potential for conversion of the Curetis Convertible Notes into shares of OpGen’s common stock having been agreed and executed
with the holders of such Curetis Convertible Notes; the Interim Facility having been put in place within five business days of
completion of the interim equity financing; and the receipt of the applicable consents or waivers to be received or granted by
certain debt financing providers of Curetis N.V. and Curetis GmbH. Failure to satisfy any of the conditions may result in the
Newco transaction not being closed.
We have agreed
to use a significant portion of the capital raised in this offering to support the operations of the Curetis Group in the period
prior to the closing. This reduces the proceeds invested in OpGen’s operations, which could have a negative impact on us
if the proposed business combination is not consummated, or if the approval process takes longer than anticipated.
Pursuant to the Implementation
Agreement, we have agreed to use a significant part of the proceeds of this offering to fund and support the operations, and satisfy
the current obligations, of the Curetis Group in the period prior to closing. Such amount could range between 40% and 50% of the
net proceeds of this offering. If such period extends for a longer period than anticipated or the amount loaned to Curetis is
higher than expected, such commitment could negatively impact the availability of resources to devote to the OpGen business or
to the business of Newco if the closing occurs, and we may be required to raise additional capital.
We will document this agreement by entering the Interim
Facility. We have not entered into the Interim Facility as of the date of this prospectus. The Interim Facility will be unsecured,
subordinated to the existing indebtedness of both Curetis and Curetis N.V., including the Curetis Convertible Notes, and will
provide a mechanism for us to lend funds to the Curetis Group to support its then current business and pay its short-term obligations.
Many of the material terms of the Interim Facility, including the interest rate, term and committed amount, have not yet been
negotiated, and we may be unable to negotiate favorable terms. We may also need to lend more money to Curetis than anticipated.
If that happens, we will have less money available to fund Newco, or to fund OpGen if the transactions contemplated by the Implementation
Agreement do not close. If the transactions contemplated by the Implementation Agreement do not close, we anticipate that it will
be difficult for Curetis to repay us under the Interim Facility, if at all. Any unanticipated loans under the Interim Facility,
or failure to be repaid under the Interim Facility would have a material adverse effect on our financial condition.
If the combination
with Curetis does not occur, our financial condition will be materially adversely affected.
If we or Curetis N.V. cannot
meet all of the conditions to close under the Implementation Agreement, and the business combination does not occur, we will be
in a difficult financial position. We will have lent funds to Curetis under the Interim Facility, and there is a real possibility
that Curetis would not be able to repay us some or all of such debt. In addition, we would have to refocus our attention on OpGen
as a stand-alone business and would likely need to raise additional funds to support that business going forward. We cannot assure
you that we would be able to continue OpGen as a stand-alone business or be able to raise sufficient capital to do so. If we are
unable to raise equity capital, we may need to incur debt financing, if possible, sell assets, curtail business programs, seek
bankruptcy protection or dissolve.
We will incur
significant indebtedness as a result of the combination with Curetis, which could have a material adverse effect on our financial
condition.
If the combination with Curetis closes, we will
assume the indebtedness of Curetis N.V. under the Curetis Convertible Notes. As of October 10, 2019, Curetis N.V. owed
indebtedness of $1.4 million to lenders under the Curetis Convertible Notes and Curetis owed indebtedness of $20.4 million
of principal (plus interest of $1.6 million) under a loan provided by the EIB. In addition, OpGen has secured
indebtedness to MGHIF under the MGHIF Note. Pursuant to the Implementation Agreement, OpGen will be required to assume this
indebtedness of Curetis N.V. (subject to approval of the holder of the Curetis Convertible Notes) and Curetis, and Newco will
therefore be obligated under substantially more indebtedness than OpGen currently owes. Newco may not be able to generate
sufficient cash to service all of its indebtedness and may be forced to take other actions to satisfy its obligations under
indebtedness that may not be successful. The inability in the future to repay such indebtedness when due would have a
material adverse effect on Newco.
The proposed business
combination transaction with Curetis will significantly change the business and operations of OpGen. We may face challenges integrating
the businesses.
Following the consummation of the
proposed combination with Curetis, OpGen will continue as the operating entity and both the size and geographic scope of OpGen’s
business will significantly increase. Most of the Curetis business is currently conducted in Europe, Asia and other countries
outside of the United States, and many of the Curetis employees are located outside of the United States. In addition, the
majority of the initial board of directors will consist of individuals appointed by Curetis N.V., and we expect that the focus
of Newco may shift to Curetis operations. We may face challenges integrating such geographically diverse businesses and
implementing a smooth transition of business focus and governance in a timely or efficient manner. In particular, if the
effort we devote to the integration of our businesses with that of Curetis diverts more management time or other resources from
carrying out our operations than we originally planned, our ability to maintain and increase revenues as well as manage our costs
could be impaired. Furthermore, our capacity to expand other parts of our existing businesses may be impaired. We also cannot
assure you that the combination of the OpGen and Curetis businesses will function as we anticipate, or that significant synergies
will result from the business combination. Any of the above could have a material adverse effect on our business.
Management and
the board of directors of OpGen will change upon the consummation of the Curetis transaction. We cannot assure you that this will
not have a material impact on the Newco.
The current chief executive officer of Curetis N.V.,
Oliver Schacht, Ph.D., will be the chief executive officer of Newco, and Timothy C. Dec will continue to serve as
chief financial officer. The Implementation Agreement provides that four members of the initial board of directors of
Newco following the closing will be appointed by Curetis N.V. and two by the board of directors of OpGen. The
parties currently intend to pursue a board of up to seven members post-closing. The current members of the management board
of Curetis N.V. do have experience serving on the boards of companies listed on Euronext and German prime standard companies,
but not on U.S. publicly-listed companies and this could impact the transition of Newco.
The combination
of the OpGen and Curetis businesses may not lead to the growth and success of the combined business that we believe will occur.
Although we believe the combination
of the OpGen and Curetis businesses provides a significant commercial opportunity for growth, we may not realize all of the synergies
that we anticipate and may not be successful in implementing our commercialization strategy. Our combined business will be subject
to all of the risks and uncertainties inherent in the pursuit of growth in our industry and we may not be able to successfully
sell our products, obtain the regulatory clearances and approvals we apply for or, or realize the anticipated benefits from our
distribution, collaboration and other commercial partners. If we are not able to grow the business of Newco as a commercial enterprise,
our financial condition will be negatively impacted.
Integrating the
businesses of OpGen and Curetis may disrupt or have a negative impact on Newco.
We could have difficulty integrating
the assets, personnel and business of OpGen and Curetis. The proposed transaction is complex and we will need to devote significant
time and resources to integrating the businesses. Risks that could impact us negatively include:
|
|
·
|
the
difficulty of integrating the acquired companies, and their concepts and operations;
|
|
|
·
|
the
difficulty in combining our financial operations and reporting;
|
|
|
·
|
the
potential disruption of the ongoing businesses and distraction of our management;
|
|
|
·
|
changes
in our business focus and/or management;
|
|
|
·
|
risks
related to international operations;
|
|
|
·
|
the
potential impairment of relationships with employees and partners as a result of any
integration of new management personnel; and
|
|
|
·
|
the
potential inability to manage an increased number of locations and employees.
|
If we are not successful in
addressing these risks effectively, the business of Newco could be severely impaired.
We expect our
ability to utilize our net operating loss carryforwards will be limited as a result of an “ownership change,” as defined
in Section 382 of the Internal Revenue Code triggered by consummation of the transaction with Curetis.
As of December 31, 2018, we
had approximately $178.2 million of net operating loss, or NOL, carryforwards for U.S. federal tax purposes. Under U.S. federal
income tax law, we generally can use our NOL carryforwards (and certain tax credits) to offset ordinary taxable income, thereby
reducing our U.S. federal income tax liability, for up to 20 years from the year in which the losses were generated, after which
time they will expire. State NOL carryforwards (and certain tax credits) generally may be used to offset future state taxable
income for 20 years from the year in which the losses are generated, depending on the state, after which time they will expire.
The rate at which we can utilize our NOL carryforwards is limited (which could result in NOL carryforwards expiring prior to their
use) each time we experience an “ownership change,” as determined under Section 382 of the Internal Revenue Code.
A Section 382 ownership change generally occurs if a shareholder or a group of shareholders who are deemed to own at least 5%
of our common stock increase their ownership by more than 50 percentage points over their lowest ownership percentage within a
rolling three-year period. If an ownership change occurs, Section 382 generally would impose an annual limit on the amount of
post-ownership change taxable income that may be offset with pre-ownership change NOL carryforwards equal to the product of the
total value of our outstanding equity immediately prior to the ownership change (reduced by certain items specified in Section
382) and the U.S. federal long-term tax-exempt interest rate in effect at the time of the ownership change. A number of special
and complex rules apply in calculating this Section 382 limitation. While the complexity of Section 382 makes it difficult to
determine whether and when an ownership change has occurred, and if a portion of our NOLs is subject to an annual limitation under
Section 382, we believe that an additional ownership change may occur upon the consummation of the transaction with Curetis. In
addition, our ability to use our NOL carryforwards will be limited to the extent we fail to generate enough taxable income in
the future before they expire. Existing and future Section 382 limitations and our inability to generate enough taxable income
in the future could result in a substantial portion of our NOL carryforwards expiring before they are used. In addition, under
the 2017 Tax Cut and Jobs Act, effective for losses arising in taxable years beginning after December 31, 2017, the deduction
for NOLs is limited to 80% of taxable income, NOLs can no longer be carried back, and NOLs can be carried forward indefinitely.
Current OpGen stockholders
will have a reduced ownership and voting interest after the business combination and will exercise less influence over management.
Current OpGen stockholders have the right to vote in
the election of the OpGen board of directors and on other matters affecting OpGen. Immediately after the business combination
is completed, it is estimated that then current OpGen stockholders, including purchasers in this offering, will own approximately
47.5%, and Curetis N.V. will own approximately 52.5% of the outstanding shares of Newco, in each case assuming that 1,526,717
units are sold in this offering, based on an assumed public offering price of $6.55 per unit, the closing price of our
common stock on the Nasdaq Capital Market on October 9, 2019, and assuming no sale of any pre-funded units in this offering. As
a result of the business combination, current OpGen stockholders will have less influence on the management and policies of Newco
post-closing than they currently have over OpGen.
The unaudited pro forma
financial statements included in this prospectus are presented for illustrative purposes only and the actual financial condition
and results of operations of Newco following the business combination may differ materially.
The unaudited pro forma financial
statements contained in this prospectus are presented for illustrative purposes only, are based on various adjustments, assumptions
and preliminary estimates and may not be an indication of the Newco’s financial condition or results of operations following
the business combination for several reasons. The actual financial condition and results of operations of Newco following the
business combination may not be consistent with, or evident from, these unaudited pro forma financial statements. In addition,
the assumptions used in preparing the unaudited pro forma financial information may not prove to be accurate, and other factors
may affect Newco’s financial condition or results of operations following the business combination. Any potential decline
in the combined company’s financial condition or results of operations may cause significant variations in the stock price
of the combined company.
The market price of
Newco common stock after the business combination may be affected by factors different from those affecting the shares of OpGen
currently.
Curetis’ business differs
in important respects from that of OpGen, and, accordingly, the results of operations of Newco and the market price of Newco common
stock after the completion of the business combination may be affected by factors different from those currently affecting the
results of operations of each of OpGen. For a discussion of the business of OpGen and of certain factors to consider in connection
with OpGen’s business, see “Prospectus Summary—OpGen’s Business” included elsewhere in this prospectus
and incorporated by reference herein, and the consolidated financial statements of OpGen incorporated by reference herein. For
a discussion of the business of Curetis and of certain factors to consider in connection with Curetis’ business, see “Prospectus
Summary—Curetis’ Business” and the financial statements of Curetis included elsewhere in this prospectus.
The business of Newco, like the businesses
of OpGen and Curetis, will be heavily regulated.
Both OpGen and Curetis are pursuing business strategies
that require FDA clearance, CE marking and other regulatory approvals in order to offer products and services for diagnostic purposes.
Both OpGen and Curetis are currently responding to additional information requests from the FDA. Pursuit of the necessary regulatory
clearances and approvals is time-consuming and expensive. We cannot assure you that Newco will obtain the necessary regulatory
clearances and approvals. The failure to obtain such regulatory clearances and approvals could have a material adverse effect
on Newco’s business.
Newco will be responsible for retention
and severance payments to certain OpGen executive officers, and satisfaction of PSOPs awards issued to Curetis executive
officers and directors.
Under the OpGen, Inc. Retention Plan for Executives, effective
September 21, 2018, retention payments equal to 4% of the value of the business combination transaction will be paid over time
to four executive officers of OpGen following the closing of the proposed business combination. We currently estimate the cost
of such retention payments will be approximately $789 thousand. In addition, severance payments, including an amount equal to one
times his base salary, will be paid to Evan Jones, the current Chief Executive Officer of OpGen under his Change in Control Severance
Agreement when he is terminated as Chief Executive Officer of OpGen in connection with the closing under the Implementation Agreement.
OpGen has also agreed to replace the PSOP awards granted to Curetis executive officers and directors with equity awards of OpGen.
Such payments and issuances will be obligations of Newco after the closing. Although the payments to the OpGen executives may be
payable over a two-year period, if the other OpGen executives have an eligible termination of employment under the retention plan,
such payments will be accelerated, which could have an adverse effect on Newco's financial position.
Risks Related to Our
Business
We have a history
of losses, and we expect to incur losses for the next several years. The report of our independent registered public accounting
firm on our financial statements for the years ended December 31, 2018 and 2017 contains explanatory language that substantial
doubt exists about our ability to continue as a going concern.
We have incurred substantial
losses since our inception, and we expect to continue to incur additional losses for the next several years. For the years ended
December 31, 2018 and 2017, we had net losses of $13.4 million and $15.4 million, respectively. Net loss for the six months
ended June 30, 2019 was $6.5 million. From our inception through June 30, 2019, we had an accumulated deficit of $168.5 million.
The report of our independent registered public accounting firm on our financial statements for the years ended December 31,
2018 and 2017 contains explanatory language that substantial doubt exists about our ability to continue as a going concern. We
completed a number of financings in 2019, 2018 and 2017, including offerings in March 2019, October 2018, February 2018, July
2017, and an at-the-market, or ATM, public offering commenced in September 2016 and terminated in October 2018. The net proceeds
from such financings were approximately $35.8 million.
We expect to continue to incur
significant operating expenses relating to, among other things:
|
|
·
|
developing
our Acuitas AMR Gene Panel products and services for antibiotic resistance testing;
|
|
|
·
|
commercializing
our Acuitas AMR Gene Panel tests and Acuitas Lighthouse informatics services, as RUO
products and, once cleared, as diagnostic products and services;
|
|
|
·
|
conducting
additional clinical trials as we seek regulatory approval for some of our product offerings;
|
|
|
·
|
developing,
presenting and publishing additional clinical and economic utility data intended to increase
clinician adoption of our current and future products and services;
|
|
|
·
|
expanding
our operating capabilities;
|
|
|
·
|
developing
additional collaborative arrangements;
|
|
|
·
|
maintaining,
expanding and protecting our intellectual property portfolio and trade secrets;
|
|
|
·
|
expanding
the size and geographic reach of our sales force and our marketing capabilities to commercialize
potential future products and services; and
|
|
|
·
|
recruiting
and retaining our quality assurance and compliance personnel and maintaining compliance
with regulatory requirements.
|
Even if we achieve significant
revenues, we may not become profitable, and even if we achieve profitability, we may not be able to sustain or increase profitability
on a quarterly or annual basis. Our failure to become and remain consistently profitable could adversely affect the market price
of our common stock and could significantly impair our ability to raise capital, expand our business or continue to pursue our
growth strategy. We believe that current cash on hand will be sufficient to fund operations into October 2019. In the event we
are unable to successfully raise additional capital during or before the middle of October 2019, we will not have sufficient cash
flows and liquidity to finance our business operations as currently contemplated. Accordingly, in such circumstances we would
be compelled to reduce general and administrative expenses and delay research and development projects, including the purchase
of scientific equipment and supplies, until we are able to obtain sufficient financing. We have no committed sources of capital
and may find it difficult to raise money on terms favorable to us or at all. In addition, if we are unable to complete this offering
by October 15, 2019 we or Curetis N.V. will have the right to terminate the Implementation Agreement. The failure to obtain sufficient
capital to support our operations would have an adverse effect on our business, financial condition and results of operations.
We expect to make
significant additional investment in the future related to our diagnostic products and services, which investments will require
additional financing transactions through the issuance of equity or debt. If we are unable to make such investments our business
will suffer.
We anticipate that we will
need to make significant investments in our Acuitas AMR Gene Panel tests in development and Acuitas Lighthouse Software in order
to make our business profitable. We need to expend significant investments to develop such products and services. There can be
no assurance that we can obtain sufficient resources or capital from operations or future financings to support these development
activities.
To meet our capital needs,
we are considering multiple alternatives, including, but not limited to, additional equity financings, debt financings and other
funding transactions, licensing and/or partnering arrangements and business combination transactions. We believe that additional
equity financings are the most likely source of capital. There can be no assurance that we will be able to complete any such financing
transaction on acceptable terms or otherwise.
In July 2015, in connection
with our acquisition of our subsidiary, AdvanDx, MGHIF made investments in the Company, including the $1 million MGHIF Note, secured
by a security interest in substantially all of our assets, including our intellectual property assets. The debt is due to be paid
in six semi-annual payments of $166,667 beginning on January 2, 2019 and ending on July 1, 2021. Such secured creditor rights
could negatively impact our ability to raise money in the future. If we default on payments under the MGHIF Note, MGHIF has the
rights of a secured creditor. If those rights are exercised, it could have a material adverse effect on our financial condition.
The process to
obtain and maintain FDA clearances or approvals for our products is complex and time-consuming. If we fail to obtain such clearances
or approvals, our business and results of operations will be materially adversely impacted.
The process of obtaining regulatory
clearances or approvals to market a medical device can be costly and time consuming, and we may not be able to obtain these clearances
or approvals on a timely basis, if at all. Anyone who wants to market in the United States a Class I, II, or III device intended
for human use for which a Premarket Approval application, or a PMA, is not required, must submit a 510(k) application to the FDA
unless the device is exempt from 510(k) requirements of the Federal Food, Drug, and Cosmetic Act, or the FD&C Act. The FDA
will clear marketing of a lower risk medical device through the 510(k) process if the manufacturer demonstrates that the new product
is “substantially equivalent” to an existing, or “predicate,” FDA-cleared product. A device may not be
marketed in the United States until the applicant receives a letter declaring the device substantially equivalent. If the FDA
declares a device as not substantially equivalent, the device is automatically classified as a Class III (high-risk) device
for which a PMA or de novo clearance is required. After an FDA determination that a device is not substantially equivalent,
the 510(k) applicant may: (i) resubmit another 510(k) with new data; (ii) request a Class I or II designation through the de
novo classification process; (iii) file a reclassification petition; or (iv) submit a PMA. The PMA process is more costly,
lengthy and uncertain than the 510(k) clearance process. A PMA application must be supported by extensive data, including, but
not limited to, technical, preclinical, clinical trial, manufacturing and labeling data, to demonstrate to the FDA’s satisfaction
the safety and efficacy of the device for its intended use.
We submitted a 510(k) application
seeking FDA clearance for our Acuitas AMR Gene Panel (Isolates) product in May 2019. In connection with the FDA’s Substantive
Review, we received an AI Request in July 2019 effectively placing our submission on hold until we submit a complete response,
which must be done within 180 days of the date listed on the AI Request. We plan to submit a complete response to the FDA’s
AI Request in the fourth quarter of 2019. We anticipate making an additional FDA submission in the first quarter of 2020 for our
Acuitas AMR Gene Panel (Urine) product and in the first half of 2020 for the Acuitas Lighthouse software. If we are not able to
achieve clearance of such products and services on a timely basis, or at all, we will not be able to pursue our business strategy
on the anticipated timeline and our business, results of operations and financial condition will be materially adversely impacted.
Our products and
services may never achieve significant commercial market acceptance.
Our products and services
may never gain significant acceptance in the marketplace and, therefore, may never generate substantial revenue or profits for
us. Our ability to achieve commercial market acceptance for our products will depend on several factors, including:
|
|
·
|
our
ability to convince the medical community of the clinical utility of our products and
services and their potential advantages over existing tests, including our surveillance
services offering, despite the lack of reimbursement for such services;
|
|
|
·
|
our
ability to successfully develop automated rapid pathogen identification and antibiotic
resistance testing products and services, including bioinformatics, and convince hospitals
and other healthcare providers of the patient safety, improved patient outcomes and potential
cost savings that could result;
|
|
|
·
|
our
ability to grow our microbial isolate and antibiotic resistance genes knowledgebase;
|
|
|
·
|
our
ability to convince the medical community of the accuracy and speed of our products and
services, as contrasted with the current methods available; and
|
|
|
·
|
the
willingness of hospitals and physicians to use our products and services.
|
Our future success
is dependent upon our ability to expand our customer base.
The current customers we are
targeting for our Acuitas AMR Gene Panel (RUO) and Acuitas Lighthouse Software (RUO) test products and services are hospital systems,
acute care hospitals, particularly those with advanced care units, such as intensive care units, community-based hospitals and
governmental units, such as public health facilities. If the Acuitas AMR Gene Panel and Acuitas Lighthouse Software products are
approved for diagnostic use, we will need to provide a compelling case for the savings, patient safety and recovery, reduced lengths
of stay and reduced costs, among other benefits, that come from adopting our MDRO diagnosis and antibiotic stewardship products
and services. If we are not able to successfully increase our customer base and lawfully commercialize these products for diagnostic
use, sales of our products and our margins may not meet expectations. Attracting new customers and introducing new products and
services requires substantial time and expense. Any failure to expand our existing customer base, or launch new products and services,
would adversely affect our ability to improve our operating results.
We have seen declining revenues
from our current customers for our QuickFISH products as we work to transition to Acuitas automated rapid pathogen identification
products. Continued decline without additional product offerings could materially, adversely affect our business.
We are developing
new in vitro diagnostic tests for more rapid identification of MDROs and antibiotic resistance genomic information. If we are
unable to successfully develop, receive regulatory clearance or approval for or commercialize such new products and services,
our business will be materially, adversely affected.
We are developing the Acuitas
AMR Gene Panel (Urine) as a new under-three-hour antibiotic resistance diagnostic product that we believe, if cleared for clinical
diagnostic use, could help address many of the current issues with the need for more rapid identification of infectious diseases
and testing for antibiotic resistance. Development of new diagnostic products is difficult and we cannot assure you that we will
be successful in such product development efforts, or, if successful, that we will receive the necessary regulatory clearances
to commercialize such products. We have identified up to 47 antibiotic resistance genes that, if cleared for clinical diagnostic
use, could help guide clinician antibiotic therapy decisions when test results are evaluated using the Acuitas Lighthouse Software,
if similarly cleared. Although we have demonstrated preliminary feasibility, and confirmed genotype/phenotype predictive algorithms,
such product development efforts will require us to work collaboratively with other companies, academic and government laboratories,
and healthcare providers to access sufficient numbers of microbial isolates, develop the diagnostic tests, successfully conduct
the necessary clinical trials and apply for and receive regulatory clearances or approvals for the intended use of such diagnostic
tests. In addition, we would need to successfully commercialize such products. Such product development, clearance or approval
and commercialization activities are time-consuming and expensive and there can be no assurance that we will have sufficient funds
to successfully complete such efforts. We currently plan to complete development and submit for FDA clearance to market such antibiotic
resistance diagnostic tests in the United States in late 2019. Any significant delays or failures in this process could have a
material adverse effect on our business and financial condition.
We offer these products in
development to the RUO market and for other non-clinical research uses prior to receiving clearance or approval to commercialize
these products in development for use in the clinical setting. As such, we are required to comply with the applicable laws and
regulations regarding such other uses. Failure to comply with such laws and regulations may have a significant impact on the Company.
We
may enter into agreements with U.S. or other government agencies, which could be subject to uncertain future funding.
The presence of MDROs and the
need for antibiotic stewardship activities have prompted state, federal and international government agencies to develop programs
to combat the effects of MDROs. In 2019, we have been party to a collaboration, called The New York State Infectious Disease Digital
Health Initiative, with The New York State DOH and ILÚM to develop a research program to detect, track, and manage antimicrobial-resistant
infections at healthcare institutions in New York State.
In the future, we may seek
to enter into additional agreements with governmental funding sources or contract with government healthcare organizations to
sell our products and services. Under such agreements, we would rely on the continued performance by these government agencies
of their responsibilities under these agreements, including adequate continued funding of the agencies and their programs. We
have no control over the resources and funding that government agencies may devote to these agreements, which may be subject to
annual renewal.
Government agencies may fail
to perform their responsibilities under these agreements, which may cause them to be terminated by the government agencies. In
addition, we may fail to perform our responsibilities under these agreements. Any government agreements would be subject to audits,
which may occur several years after the period to which the audit relates. If an audit identified significant unallowable costs,
we could incur a material charge to our earnings or reduction in our cash position. As a result, we may be unsuccessful entering,
or ineligible to enter, into future government agreements.
If
the utility of our current products and products in development is not supported by studies published in peer-reviewed medical
publications, the rate of adoption of our current and future products and services by clinicians and healthcare facilities may
be negatively affected.
The results of our clinical
and economic validation studies involving our Acuitas AMR Gene Panel tests and Acuitas Lighthouse Software have been presented
at major infectious disease and infection control society meetings. We need to maintain and grow a continued presence in peer-reviewed
publications to promote clinician adoption of our products. We believe that peer-reviewed journal articles that provide evidence
of the utility of our current and future products and services, and adoption by key opinion leaders in the infectious disease
market, are very important to our commercial success. Clinicians typically take a significant amount of time to adopt new products
and testing practices, partly because of perceived liability risks and the uncertainty of a favorable cost/benefit analysis. It
is critical to the success of our sales efforts that we educate a sufficient number of clinicians and administrators about our
products and demonstrate their clinical benefits. Clinicians may not adopt our current and future products and services unless
they determine, based on published peer-reviewed journal articles and the experience of other clinicians, that our products provide
accurate, reliable, useful and cost-effective information that is useful in MDRO diagnosis, screening and outbreak prevention.
If our current and future products and services or the technology underlying our products and services or our future product offerings
do not receive sufficient favorable exposure in peer-reviewed publications, the rate of clinician adoption could be negatively
affected. The publication of clinical data in peer-reviewed journals is a crucial step in commercializing our products, and our
inability to control when, if ever, results are published may delay or limit our ability to derive sufficient revenue from any
product that is the subject of a study.
Our
sales cycle for our marketed products and services is lengthy and variable, which makes it difficult for us to forecast revenue
and other operating results.
We believe the sales cycles
for our Acuitas AMR Gene Panel and Acuitas Lighthouse Software as diagnostic products will be lengthy, which will make it difficult
for us to accurately forecast revenues in a given period, and may cause revenue and operating results to vary significantly from
period to period. Potential customers for our products typically need to commit significant time and resources to evaluate our
products, and their decision to purchase our products may be further limited by budgetary constraints and numerous layers of internal
review and approval, which are beyond our control. We spend substantial time and effort assisting potential customers in evaluating
our products. Even after initial approval by appropriate decision makers, the negotiation and documentation processes for the
actual adoption of our products on a facility-wide basis can be lengthy. As a result of these factors, based on our experience
to date, our sales cycle, the time from initial contact with a prospective customer to routine commercial use of our products,
has varied and could be 12 months or longer, which has made it difficult for us to accurately project revenues and operating results.
In addition, the revenue generated from sales of our products may fluctuate from time to time due to changes in the testing volumes
of our customers. As a result, our results may fluctuate on a quarterly basis, which may adversely affect the price of our common
stock.
We
are currently party to, and may enter into additional, collaborations with third parties to develop product and services candidates.
If these collaborations are not successful, our business could be adversely affected.
We are currently party to
a few collaborations, and anticipate that we will enter into additional collaborations, related to our MDRO and informatics products
and services. Such collaborations are and may be with pharmaceutical companies, platform companies or other participants in our
industry. We have limited control over the amount and timing of resources that any such collaborators could dedicate to the development
or commercialization of the subject matter of any such collaboration. Our ability to generate revenues from these arrangements
would depend on our and our collaborators’ abilities to successfully perform the functions assigned to each of us in these
arrangements. Our relationships with collaborators may pose several risks, including the following:
|
|
·
|
collaborators
have significant discretion in determining the efforts and resources that they will apply
to these collaborations;
|
|
|
·
|
collaborators
may not perform their obligations as expected;
|
|
|
·
|
we
may not achieve any milestones, or receive any milestone payments, under our collaborations,
including milestones and/or payments that we expect to achieve or receive;
|
|
|
·
|
the
clinical trials, if any, conducted as part of these collaborations may not be successful;
|
|
|
·
|
a
collaborator might elect not to continue or renew development or commercialization programs
based on clinical trial results, changes in the collaborator’s strategic focus
or available funding or external factors, such as an acquisition, that diverts resources
or creates competing priorities;
|
|
|
·
|
we
may not have access to, or may be restricted from disclosing, certain information regarding
product or services candidates being developed or commercialized under a collaboration
and, consequently, may have limited ability to inform our stockholders about the status
of such product or services candidates;
|
|
|
·
|
collaborators
could independently develop, or develop with third parties, products that compete directly
or indirectly with our product candidates if the collaborators believe that competitive
products are more likely to be successfully developed or can be commercialized under
terms that are more economically attractive than ours;
|
|
|
·
|
product
or services candidates developed in collaboration with us may be viewed by our collaborators
as competitive with their own product or services, which may cause collaborators to cease
to devote resources to the commercialization of our product or services candidates;
|
|
|
·
|
a
collaborator with marketing and distribution rights to one or more of our product or
services candidates that achieve regulatory approval may not commit sufficient resources
to the marketing and distribution of any such product candidate;
|
|
|
·
|
disagreements
with collaborators, including disagreements over proprietary rights, contract interpretation
or the preferred course of development of any product or services candidates, may cause
delays or termination of the research, development or commercialization of such product
or services candidates, may lead to additional responsibilities for us with respect to
such product or services candidates or may result in litigation or arbitration, any of
which would be time-consuming and expensive;
|
|
|
·
|
collaborators
may not properly maintain or defend our intellectual property rights or may use our proprietary
information in such a way as to invite litigation that could jeopardize or invalidate
our intellectual property or proprietary information or expose us to potential litigation;
|
|
|
·
|
disputes
may arise with respect to the ownership of intellectual property developed pursuant to
a collaboration;
|
|
|
·
|
collaborators
may infringe the intellectual property rights of third parties, which may expose us to
litigation and potential liability; and
|
|
|
·
|
collaborations
may be terminated for the convenience of the collaborator and, if terminated, we could
be required to raise additional capital to pursue further development or commercialization
of the applicable product or services candidates.
|
If our collaborations do not
result in the successful development and commercialization of products or services, we may not receive any future research funding
or milestone or royalty payments under the collaborations. If we do not receive the funding we would expect under these agreements,
our development of product and services candidates could be delayed and we may need additional resources to develop our product
candidates.
We
may not be successful in finding strategic collaborators for continuing development of certain of our product or services candidates
or successfully commercializing or competing in the market for certain indications.
We may seek to develop strategic
partnerships for developing certain of our product or services candidates, due to capital costs required to develop the product
or services candidates or manufacturing constraints. We may not be successful in our efforts to establish such a strategic partnership
or other alternative arrangements for our product or services candidates because our research and development pipeline may be
insufficient, our product or services candidates may be deemed to be at too early of a stage of development for collaborative
effort or third parties may not view our product or services candidates as having the requisite potential to demonstrate commercial
success.
If we are unable to reach
agreements with suitable collaborators on a timely basis, on acceptable terms or at all, we may have to curtail the development
of a product or service candidate, reduce or delay our development program, delay our potential commercialization, reduce the
scope of any sales or marketing activities or increase our expenditures and undertake development or commercialization activities
at our own expense. If we elect to fund development or commercialization activities on our own, we may need to obtain additional
expertise and additional capital, which may not be available to us on acceptable terms or at all. If we fail to enter into collaborations
and do not have sufficient funds or expertise to undertake the necessary development and commercialization activities, we may
not be able to further develop our product candidates and our business, financial condition, results of operations and prospects
may be materially and adversely affected.
We
are an early commercial stage company and may never be profitable.
We rely principally on the
commercialization of our QuickFISH and Acuitas Gene Panel (RUO) test products and our Acuitas Lighthouse Software (RUO) to generate
future revenue growth. To date, our Acuitas test products and Acuitas Lighthouse services have delivered only minimal revenue,
as they have not-yet been cleared for clinical diagnostic use. We believe that our commercialization success is dependent upon
our ability to significantly increase the number of hospitals, long-term care facilities and other inpatient healthcare settings
that use our products. If demand for products does not increase as quickly as we have planned, we may be unable to increase our
revenue levels as expected. We are currently not profitable. Even if we succeed in increasing adoption of our products by our
target markets, maintaining and creating relationships with our existing and new customers and developing and commercializing
additional molecular testing products, we may not be able to generate sufficient revenue to achieve or sustain profitability.
We
have limited experience in marketing and selling our products, and if we are unable to adequately address our customers’
needs, it could negatively impact sales and market acceptance of our products and we may never generate sufficient revenue to
achieve or sustain profitability.
We sell our products through
our own direct sales force, which sells our Acuitas AMR Gene Panel (RUO) tests and Acuitas Lighthouse Software and our QuickFISH
products. All of these products and services may be offered and sold to different potential customers or involve discussions with
multiple personnel in in-patient facilities. Our future sales will depend in large part on our ability to increase our marketing
efforts and adequately address our customers’ needs. The inpatient healthcare industry is a large and diverse market. We
will need to attract and develop sales and marketing personnel with industry expertise. Competition for such employees is intense.
We may not be able to attract and retain sufficient personnel to maintain an effective sales and marketing force. If we are unable
to successfully market our products and adequately address our customers’ needs, it could negatively impact sales and market
acceptance of our products, and we may never generate sufficient revenue to achieve or sustain profitability.
If
our sole manufacturing facility becomes inoperable, our Acuitas, QuickFISH and PNA FISH products, and our business will be harmed.
We manufacture our Acuitas,
QuickFISH and PNA FISH products in our facility in Gaithersburg, Maryland. We do not have redundant facilities. Our facility and
the equipment we use manufacture our products would be costly to replace and could require substantial lead time to repair or
replace if damaged or destroyed. The facility may be harmed or rendered inoperable by natural or man-made disasters, including
flooding and power outages, which may render it difficult or impossible for us manufacture our products for some period of time.
The inability to manufacture our products may result in the loss of customers or harm our reputation, and we may be unable to
regain those customers in the future. Although we possess insurance for damage to our property and the disruption of our business,
this insurance may not be sufficient to cover all of our potential losses and may not continue to be available to us on acceptable
terms, if at all.
In order to establish a redundant
facility, we would have to spend considerable time and money securing adequate space, constructing the facility, recruiting and
training employees, and establishing the additional operational and administrative infrastructure necessary to support a second
facility. Additionally, any new manufacturing facility opened by us would be subject to FDA inspection and certification. If we
fail to maintain our FDA certification or if our FDA certification is suspended, limited or revoked, we would not be able manufacture
our products.
If demand for these products
increases beyond our current forecasts or, regulatory requirements arise, we may not be able to meet our obligations to manufacture
these products, and a backlog or reduced demand for such products could occur. If any of these issues occur, it could have a material
adverse effect on our financial condition and results of operations.
We
rely on a limited number of suppliers or, in some cases, a sole supplier, for some of our materials and may not be able to find
replacements or immediately transition to alternative suppliers.
We rely on several sole suppliers
and manufacturers, including Thermo Fisher Scientific, QIAGEN, and Fluidigm Corporation, for supplying certain reagents, raw materials,
supplies and substances that we use to manufacture our products. An interruption in our operations could occur if we encounter
delays or difficulties in securing these items or manufacturing our products, or if we are unable to obtain an acceptable substitute
in the event of such delays or difficulties. Any such interruption could significantly affect our business, financial condition,
results of operations and reputation.
If
we cannot compete successfully with our competitors, we may be unable to increase or sustain our revenue or achieve and sustain
profitability.
Our competitors include rapid
diagnostic testing and traditional microbiology companies, commercial laboratories, information technology companies, and hospital
laboratories who may internally develop testing capabilities. Principal competitive factors in our target market include: organizational
size, scale, and breadth of product offerings; rapidity of test results; quality and strength of clinical and analytical validation
data and confidence in diagnostic results; cost effectiveness; ease of use; and regulatory approval status.
Our principal competition
comes from traditional methods used by healthcare providers to diagnose and screen for MDROs and from other molecular diagnostic
companies creating screening and diagnostic products such as Cepheid, Becton-Dickinson, bioMérieux, Accelerate Diagnostics,
T2 Biosystems, GenMark, and Nanosphere.
We also face competition from
commercial laboratories, such as Bio-Reference Laboratories, Inc., Laboratory Corporation of America Holdings, Quest Diagnostics
Incorporated, Pathnostics, and EuroFins, which we believe have strong infrastructure to support the commercialization of diagnostic
laboratory services.
Competitors may develop their
own versions of competing products in countries where we do not have patents, where our patents do not cover competitor products,
or where our intellectual property rights are not recognized.
Many of our potential competitors
have widespread brand recognition and substantially greater financial, technical, research and development and selling and marketing
capabilities than we do. Others may develop products with prices lower than ours that could be viewed by hospitals, physicians
and payers as functionally equivalent to our product and service offering, or offer products at prices designed to promote market
penetration, which could force us to lower the list prices of our product and service offerings and affect our ability to achieve
profitability. If we are unable to change clinical practice in a meaningful way or compete successfully against current and future
competitors, we may be unable to increase market acceptance and sales of our products, which could prevent us from increasing
our revenue or achieving profitability and could cause our stock price to decline.
Our
products and services are not covered by reimbursement by Medicare, Medicaid and other governmental and third-party payors. If
we cannot convince our customers that the savings from use of our products and services will increase their overall reimbursement,
our business could suffer.
Our products and services do
not currently receive reimbursement from Medicare, Medicaid, other governmental payors or commercial third-party payors. Policy
and rule changes in reimbursement announced by the United States Department of Health and Human Services (HHS) Centers for Medicare
and Medicaid Services (CMS), including potential financial incentives for reductions in hospital acquired infection, and penalties
and decreased Medicare reimbursement for patients with hospital acquired infections provide us with an opportunity to establish
a business case for the purchase and use of our screening and diagnostic products and services. If we cannot convince our customers
that the savings from use of our products and services will increase or stabilize their overall profitability and improve clinical
outcomes, our business will suffer.
Failure
in our information technology, storage systems or our Acuitas Lighthouse Software could significantly disrupt our operations and
our research and development efforts, which could adversely impact our revenues, as well as our research, development and commercialization
efforts.
Our ability to execute our
business strategy depends, in part, on the continued and uninterrupted performance of our information technology systems, which
support our operations and our research and development efforts, as well as our storage systems and our analyzers. Due to the
sophisticated nature of the technology we use in our products and service offerings, including our Acuitas Lighthouse Software
services, we are substantially dependent on our information technology systems. Information technology systems are vulnerable
to damage from a variety of sources, including telecommunications or network failures, malicious human acts and natural disasters.
Moreover, despite network security and back-up measures, some of our servers are potentially vulnerable to physical or electronic
break-ins, computer viruses and similar disruptive problems. Despite the precautionary measures we have taken to prevent unanticipated
problems that could affect our information technology systems, sustained or repeated system failures that interrupt our ability
to generate and maintain data, and in particular to operate our Acuitas Lighthouse Software, could adversely affect our ability
to operate our business. Any interruption in the operation of our Acuitas Lighthouse Software, due to information technology system
failures, part failures or potential disruptions in the event we are required to relocate our instruments within our facility
or to another facility, could have an adverse effect on our operations.
Security
breaches, loss of data and other disruptions could compromise sensitive information related to our business or prevent us from
accessing critical information and expose us to liability, which could adversely affect our business and our reputation.
In the ordinary course of our
business, we collect and store sensitive data, including legally protected health information and personally identifiable information
about our customers and their patients. We also store sensitive intellectual property and other proprietary business information,
including that of our customers. We manage and maintain our applications and data utilizing a combination of on-site systems and
cloud-based data center systems. These applications and data encompass a wide variety of business critical information, including
research and development information, commercial information and business and financial information.
We face four primary risks
relative to protecting this critical information: loss of access risk, inappropriate disclosure risk, inappropriate modification
risk and the risk of our being unable to identify and audit our controls over the first three risks.
We are highly dependent on
information technology networks and systems, including the Internet, to securely process, transmit and store this critical information.
Security breaches of this infrastructure, including physical or electronic break-ins, computer viruses, attacks by hackers and
similar breaches, can create system disruptions, shutdowns or unauthorized disclosure or modification of confidential information.
The secure processing, storage, maintenance and transmission of this critical information is vital to our operations and business
strategy, and we devote significant resources to protecting such information. Although we take measures to protect sensitive information
from unauthorized access or disclosure, our information technology and infrastructure may be vulnerable to attacks by hackers
or viruses or breached due to employee error, malfeasance or other disruptions.
A security breach or privacy
violation that leads to disclosure or modification of or prevents access to consumer information (including personally identifiable
information or protected health information) could harm our reputation, compel us to comply with disparate state breach notification
laws, require us to verify the correctness of database contents and otherwise subject us to liability under laws that protect
personal data, resulting in increased costs or loss of revenue. If we are unable to prevent such security breaches or privacy
violations or implement satisfactory remedial measures, our operations could be disrupted, and we may suffer loss of reputation,
financial loss and other regulatory penalties because of lost or misappropriated information, including sensitive consumer data.
In addition, these breaches and other inappropriate access can be difficult to detect, and any delay in identifying them may lead
to increased harm of the type described above.
Any such breach or interruption
could compromise our networks, and the information stored there could be inaccessible or could be accessed by unauthorized parties,
publicly disclosed, lost or stolen. Any such interruption in access, improper access, disclosure or other loss of information
could result in legal claims or proceedings, liability under laws that protect the privacy of personal information, such as the
federal HIPAA and regulatory penalties. Unauthorized access, loss or dissemination could also disrupt our operations, including
our ability to perform tests, provide test results, bill facilities or patients, process claims and appeals, provide customer
assistance services, conduct research and development activities, collect, process and prepare Company financial information,
provide information about our current and future solutions and other patient and clinician education and outreach efforts through
our website, and manage the administrative aspects of our business and damage our reputation, any of which could adversely affect
our business. Any such breach could also result in the compromise of our trade secrets and other proprietary information, which
could adversely affect our competitive position.
In addition, the interpretation
and application of consumer, health-related, privacy and data protection laws in the United States and elsewhere are often uncertain,
contradictory and in flux. It is possible that these laws may be interpreted and applied in a manner that is inconsistent with
our practices. If so, this could result in government-imposed fines or orders requiring that we change our practices, which could
adversely affect our business. Complying with these various laws could cause us to incur substantial costs or require us to change
our business practices and compliance procedures in a manner adverse to our business.
If
we are unable to develop products to keep pace with rapid technological, medical and scientific change, our operating results
and competitive position could be harmed. New test development involves a lengthy and complex process, and we may not be successful
in our efforts to develop and commercialize our diagnostic and screening products and services. The further development and commercialization
of additional diagnostic and screening product and service offerings are key to our growth strategy.
A key element of our strategy
is to discover, develop, validate and commercialize a portfolio of additional diagnostic and screening products and services to
rapidly diagnose and effectively treat MDRO infections and reduce the associated costs to patients, inpatient facilities and the
healthcare industry. We cannot assure you that we will be able to successfully complete development of, or commercialize any of
our planned future products and services, or that they will be clinically usable. The product development process involves a high
degree of risk and may take up to several years or more. Our new product development efforts may fail for many reasons, including:
|
|
·
|
failure
of the tests at the research or development stage;
|
|
|
·
|
lack
of clinical validation data to support the effectiveness of the tests;
|
|
|
·
|
delays
resulting from the failure of third-party suppliers or contractors to meet their obligations
in a timely and cost-effective manner;
|
|
|
·
|
failure
to obtain or maintain necessary certifications, licenses, clearances or approvals to
market or perform the test; or
|
|
|
·
|
lack
of commercial acceptance by in-patient healthcare facilities.
|
Few research and development
projects result in commercial products, and success in early clinical studies often is not replicated in later studies. At any
point, we may abandon development of new products, or we may be required to expend considerable resources repeating clinical studies
or trials, which would adversely impact the timing for generating potential revenues from those new products. In addition, as
we develop new products, we will have to make additional investments in our sales and marketing operations, which may be prematurely
or unnecessarily incurred if the commercial launch of a product is abandoned or delayed.
Our
insurance policies are expensive and protect us only from some business risks, which will leave us exposed to significant uninsured
liabilities.
We do not carry insurance for
all categories of risk that our business may encounter. Some of the policies we currently maintain include general liability,
employee benefits liability, property, umbrella, business interruption, workers’ compensation, product liability, errors
and omissions and directors’ and officers’ insurance. We do not know, however, if we will be able to maintain existing
insurance with adequate levels of coverage. Any significant uninsured liability may require us to pay substantial amounts, which
would adversely affect our cash position and results of operations.
If
we use hazardous materials in a manner that causes injury, we could be liable for damages.
Our activities currently require
the use of hazardous materials and the handling of patient samples. We cannot eliminate the risk of accidental contamination or
injury to employees or third parties from the use, storage, handling or disposal of these materials. In the event of contamination
or injury, we could be held liable for any resulting damages, and any liability could exceed our resources or any applicable insurance
coverage we may have. Additionally, we are subject on an ongoing basis to federal, state and local laws and regulations governing
the use, storage, handling and disposal of these materials and specified waste products. We are, or may in the future be, subject
to compliance with additional laws and regulations relating to the protection of the environment and human health and safety,
including those relating to the handling, transportation and disposal of medical specimens, infectious and hazardous waste and
Occupational Safety and Health Administration requirements. The requirements of these laws and regulations are complex, change
frequently and could become more stringent in the future. Failure to comply with current or future environmental laws and regulations
could result in the imposition of substantial fines, suspension of production, alteration of our production processes, cessation
of operations or other actions, which could severely harm our business.
If
we are sued for product liability or errors and omissions liability, we could face substantial liabilities that exceed our resources.
The marketing, sale and use
of our products could lead to product liability claims if someone were to allege that a product failed to perform as it was designed.
We may also be subject to liability for errors in the results we provide to physicians or for a misunderstanding of, or inappropriate
reliance upon, the information we provide. For example, if we diagnosed a patient as having an MDRO but such result was a false
positive, the patient could be unnecessarily isolated in an in-patient setting or receive inappropriate treatment. We may also
be subject to similar types of claims related to products we may develop in the future. A product liability or errors and omissions
liability claim could result in substantial damages and be costly and time consuming for us to defend. Although we maintain product
liability and errors and omissions insurance, we cannot assure you that our insurance would fully protect us from the financial
impact of defending against these types of claims or any judgments, fines or settlement costs arising out of any such claims.
Any product liability or errors and omissions liability claim brought against us, with or without merit, could increase our insurance
rates or prevent us from securing insurance coverage in the future. Additionally, any product liability lawsuit could cause injury
to our reputation or cause us to suspend sales of our products and services. The occurrence of any of these events could have
an adverse effect on our business and results of operations.
We
may be adversely affected by the current economic environment and future adverse economic environments.
Our ability to attract and
retain customers, invest in and grow our business and meet our financial obligations depends on our operating and financial performance,
which, in turn, is subject to numerous factors, including the prevailing economic conditions and financial, business and other
factors beyond our control, such as the rate of unemployment, the number of uninsured persons in the United States and inflationary
pressures. We cannot anticipate all the ways in which the current economic climate and financial market conditions, and those
in the future, could adversely impact our business.
We are exposed to risks associated
with reduced profitability and the potential financial instability of our customers, many of which may be adversely affected by
volatile conditions in the financial markets. For example, unemployment and underemployment, and the resultant loss of insurance,
may decrease the demand for healthcare services and diagnostic testing. If fewer patients are seeking medical care because they
do not have insurance coverage, we may experience reductions in revenues, profitability and/or cash flow. In addition, if economic
challenges in the United States result in widespread and prolonged unemployment, either regionally or on a national basis, a substantial
number of people may become uninsured or underinsured. To the extent such economic challenges result in less demand for our proprietary
tests, our business, results of operations, financial condition and cash flows could be adversely affected.
Risks Related to Our Public
Company Status
We incur increased costs
and demands on management as a result of compliance with laws and regulations applicable to public companies, which could harm
our operating results.
As a public company, we incur
significant legal, accounting and other expenses that we did not incur as a private company, including costs associated with public
company reporting requirements. In addition, the Sarbanes-Oxley Act of 2002 and the Dodd-Frank Act of 2010, as well as rules implemented
by the SEC and the Nasdaq Stock Market, impose a number of requirements on public companies, including with respect to corporate
governance practices. Our management and other personnel need to devote a substantial amount of time to these compliance and disclosure
obligations. Moreover, compliance with these rules and regulations has increased our legal, accounting and financial compliance
costs and has made some activities more time-consuming and costly. It is also more expensive for us to obtain director and officer
liability insurance.
If we are unable to maintain
effective internal control over financial reporting, investors may lose confidence in the accuracy and completeness of our reported
financial information and the market price of our common stock may be negatively affected.
As a public company, we are
required to maintain internal control over financial reporting and to report any material weaknesses in such internal control.
Section 404 of the Sarbanes-Oxley Act of 2002 requires that we evaluate and determine the effectiveness of our internal control
over financial reporting and provide a management report on internal control over financial reporting. If we have a material weakness
in our internal control over financial reporting, we may not detect errors on a timely basis and our financial statements may
be materially misstated.
When we are no longer an emerging
growth company and a smaller reporting company, our independent registered public accounting firm will be required to issue an
attestation report on the effectiveness of our internal control over financial reporting. Even if our management concludes that
our internal control over financial reporting is effective, our independent registered public accounting firm may conclude that
there are material weaknesses with respect to our internal controls or the level at which our internal controls are documented,
designed, implemented or reviewed.
When we are no longer an emerging
growth company and a smaller reporting company, if our auditors were to express an adverse opinion on the effectiveness of our
internal control over financial reporting because we had one or more material weaknesses, investors could lose confidence in the
accuracy and completeness of our financial disclosures, which could cause the price of our common stock to decline. Internal control
deficiencies could also result in a restatement of our financial results in the future.
We are an emerging growth
company and have elected to comply with reduced public company reporting requirements applicable to emerging growth companies,
which could make our common stock less attractive to investors.
We are an emerging growth
company, as defined under the Securities Act. We will remain an emerging growth company until May 2020, although if our revenue
exceeds $1.07 billion in any fiscal year before that time, we would cease to be an emerging growth company as of the end of that
fiscal year. In addition, if the market value of our common stock that is held by non-affiliates exceeds $700 million as of the
last business day of our second fiscal quarter of any fiscal year before May 2020, we would cease to be an emerging growth company
as of December 31 of that year. As an emerging growth company, we take advantage of exemptions from various reporting requirements
applicable to certain other public companies, including not being required to comply with the auditor attestation requirements
of Section 404 of the Sarbanes-Oxley Act, reduced financial statement and financial-related disclosures, reduced disclosure obligations
regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirement of holding
a nonbinding advisory vote on executive compensation and obtaining stockholder approval of any golden parachute payments not previously
approved by our stockholders. We cannot predict whether investors will find our common stock less attractive if we choose to rely
on any of these exemptions. If some investors find our common stock less attractive as a result of any choices to reduce future
disclosure we may make, there may be a less active trading market for our common stock and our stock price may be more volatile.
Risks
Related to Regulation of Our Business
There is no guarantee
that the FDA will grant 510(k) clearance or PMA approval of our Acuitas AMR Gene Panel tests or Acuitas Lighthouse Software, or
for any other future products we may develop, and failure to obtain necessary clearances or approvals for our future products
would adversely affect our ability to grow our business.
Before we begin to label and
market our products for use as clinical diagnostics in the United States, unless an exemption applies, we are required to obtain
prior 510(k) clearance or a PMA from the FDA. We are currently in the process of review or completing and submission of three
510(k) filings with the FDA for our Acuitas AMR Gene Panel tests and Acuitas Lighthouse Software. Such process is complex, time
consuming and expensive. The FDA may not clear or approve these products for the indications that are necessary or desirable for
successful commercialization. Failure to receive, or a significant delay in receiving, a required clearance or approval for our
products would have a material adverse effect on our ability to expand our business.
Any 510(k) clearance, de
novo authorization or PMA approval we obtain for any future product would place substantial restrictions on how our device
is marketed or sold. The FDA will continue to place considerable restrictions on our products, including, but not limited to,
the obligation to comply with the Quality System Regulation, or QSR, registering manufacturing facilities, listing the products
with the FDA, and complying with labeling, marketing, complaint handling, medical device reporting requirements, and reporting
certain corrections and removals. Obtaining FDA clearance or approval for diagnostics can be expensive and uncertain, and generally
takes from several months to several years from submission, and generally requires detailed and comprehensive scientific and clinical
data, as well as compliance with FDA regulations. In addition, we have limited experience in obtaining PMA approval from the FDA
and are therefore supplementing our operational capabilities to manage the more complex processes needed to obtain and maintain
PMAs. Notwithstanding the expense, these efforts may never result in FDA approval, de novo authorizations, or 510(k) clearance.
Even if we were to obtain regulatory approval, authorization or clearance, it may not be for the uses we believe are important
or commercially attractive, in which case we would not market our product for those uses.
We may be subject to fines,
penalties, injunctions or other enforcement actions if the FDA determines that we are promoting unapproved devices or marketing
our products for unapproved or “off-label” uses.
We are currently offering
for sale our FDA-cleared QuickFISH and PNA FISH products for clinical diagnostic use and our Acuitas AMR Gene Panel tests and
Acuitas Lighthouse Software for RUO to CROs, pharmaceutical companies, hospitals and other healthcare facilities. An RUO product
may not be marketed for clinical diagnostic use and must be labeled accordingly. Products that are intended for research use only
and are properly labeled as RUO are exempt from compliance with the FDA’s pre- and post-market requirements to which traditional
devices are subject, including the requirement that the product be cleared or approved before commercialization and QSR requirements.
However, merely including the required RUO labeling will not necessarily exempt the device from the FDA's 510(k) clearance, premarket
approval, or other requirements if the circumstances surrounding the distribution of the product indicate an objective intent
to market the product for clinical diagnostic use.
According to the FDA’s
November 2013 Guidance, circumstances indicating manufacturer intent to market an in vitro device for diagnostic use may include
written or verbal marketing claims regarding a product's clinical efficacy or performance in clinical applications, instructions
for clinical interpretation, clinical information, product names, or descriptors that claim or suggest that the IVD product may
be used for any clinical diagnostic use, including a clinical investigation that is not exempt from the FDA’s investigational
device exemption regulations. Other indications include a manufacturer's provision of technical support for clinical validation
or clinical applications or solicitation of business from clinical laboratories that do not conduct research activities.
We believe that our promotional
activities for our ROU products fall within the scope of the FDA’s enforcement discretion, as described in its November
2013 Guidance, and applicable premarket exemptions. However, the FDA could disagree and require us to stop promoting our Acuitas
AMR Gene Panel tests and Acuitas Lighthouse Software as RUO devices and obtain FDA clearance or approval for such tests. We could
be subject to regulatory or enforcement actions for any of the violations described above, including, but not limited to, the
issuance of an untitled letter, a Form 483 letter, a warning letter, injunction, seizure, civil fine and criminal penalties. It
is also possible that other federal, state or foreign enforcement authorities might take action if they consider our promotional
materials to constitute promotion of an unapproved use, which could result in significant fines or penalties under other statutory
authorities, such as laws prohibiting false claims for reimbursement. In that event, our reputation could be damaged and adoption
of the products would be impaired.
A number of the rapid
diagnostic products are regulated by the FDA and non-U.S. regulatory authorities. If we or our suppliers fail to comply with ongoing
FDA, or other foreign regulatory authority, requirements, or if we experience unanticipated problems with the products, these
products could be subject to restrictions or withdrawal from the market.
We do not have significant
experience in complying with the rules and regulations of the FDA and foreign regulatory authorities. In vitro diagnostic products
are generally regulated as medical devices, and the manufacturing processes, reporting requirements, post-approval clinical data
and promotional activities for such products, are subject to continued regulatory review, oversight and periodic inspections by
the FDA and other domestic and foreign regulatory bodies. In particular, for any of our products commercialized as medical devices,
we and our suppliers are required to comply with the following regulatory requirements, among others:
|
|
·
|
the
registration and listing regulation, which requires manufacturers to register all manufacturing
facilities and list all medical devices placed into commercial distribution;
|
|
|
·
|
the
QSR, which requires manufacturers, including third party manufacturers, to follow elaborate
design, testing, production, control, supplier/contractor selection, complaint handling,
documentation and other quality assurance procedures during the manufacturing process;
|
|
|
·
|
labeling
regulations and unique device identification requirements;
|
|
|
·
|
advertising
and promotion requirements;
|
|
|
·
|
restrictions
on sale, distribution or use of a device;
|
|
|
·
|
PMA
annual reporting requirements;
|
|
|
·
|
the
FDA’s general prohibition against promoting products for unapproved or “off-label”
uses;
|
|
|
·
|
the
Medical Device Reporting, or MDR, regulation, which requires that manufacturers report
to the FDA if their device may have caused or contributed to a death or serious injury
or malfunctioned in a way that would likely cause or contribute to a death or serious
injury if it were to reoccur;
|
|
|
·
|
medical
device correction and removal reporting regulations, which require that manufacturers
report to the FDA field corrections and product recalls or removals if undertaken to
reduce a risk to health posed by the device or to remedy a violation of the FDCA that
may present a risk to health;
|
|
|
·
|
recall
requirements, including a mandatory recall if there is a reasonable probability that
the device would cause serious adverse health consequences or death;
|
|
|
·
|
an
order of repair, replacement or refund;
|
|
|
·
|
device
tracking requirements; and
|
|
|
·
|
post
approval study and post market surveillance requirements.
|
The FDA enforces the QSR and
similarly, other regulatory bodies with similar regulations enforce those regulations through periodic inspections. The failure
by us or one of our suppliers to comply with applicable statutes and regulations administered by the FDA and other regulatory
bodies, or the failure to timely and adequately respond to any adverse inspectional observations or product safety issues, could
result in, among other things, any of the following enforcement actions against us: (1) untitled letters, Form 483 observation
letters, warning letters, fines, injunctions, consent decrees and civil penalties; (2) unanticipated expenditures to address or
defend such actions; (3) customer notifications for repair, replacement and refunds; (4) recall, detention or seizure of our products;
(5) operating restrictions or partial suspension or total shutdown of production; (6) refusing or delaying our requests for 510(k)
clearance or premarket approval of new products or modified products; (7) operating restrictions; (8) withdrawing 510(k) clearances
or PMA approvals that have already been granted; (9) refusal to grant export approval for our products; or (10) criminal prosecution.
If any of these actions were
to occur it could harm our reputation and cause our product sales and profitability to suffer and may prevent us from generating
revenue. Furthermore, if any of our key component suppliers are not in compliance with all applicable regulatory requirements
we may be unable to produce our products on a timely basis and in the required quantities, if at all.
Some of the clearances obtained
are subject to limitations on the intended uses for which the product may be marketed, which can reduce our potential to successfully
commercialize the product and generate revenue from the product. If the FDA determines that our promotional materials, labeling,
training or other marketing or educational activities constitute promotion of an unapproved use, it could request that we cease
or modify our training or promotional materials or subject us to regulatory enforcement actions. It is also possible that other
federal, state or foreign enforcement authorities might take action if they consider our training or other promotional materials
to constitute promotion of an unapproved use, which could result in significant fines or penalties under other statutory authorities,
such as laws prohibiting false claims for reimbursement.
In addition, we may be required
to conduct costly post-market testing and surveillance to monitor the safety or effectiveness of our products, and we must comply
with medical device reporting requirements, including the reporting of adverse events and malfunctions related to our products.
Later discovery of previously unknown problems with our products, including unanticipated adverse events or adverse events of
unanticipated severity or frequency, manufacturing problems, or failure to comply with regulatory requirements such as QSR, may
result in changes to labeling, restrictions on such products or manufacturing processes, withdrawal of the products from the market,
voluntary or mandatory recalls, a requirement to repair, replace or refund the cost of any medical device we manufacture or distribute,
fines, suspension of regulatory approvals, product seizures, injunctions or the imposition of civil or criminal penalties which
would adversely affect our business, operating results and prospects.
If we were to lose, or have
restrictions imposed on, FDA clearances received to date, or clearances we may receive in the future, our business, operations,
financial condition and results of operations would likely be significantly adversely affected.
Modifications
to our marketed products may require new 510(k) clearances or PMA approvals, or may require us to cease marketing or recall the
modified products until clearances or approvals are obtained.
If we modify any of our FDA-cleared
products, such modifications would require additional clearances or approvals. Modifications to a 510(k)-cleared device that could
significantly affect its safety or efficacy, or that would constitute a major change in its intended use, requires a new 510(k)
clearance or, possibly, a PMA. The FDA requires every manufacturer to make this determination in the first instance, but the FDA
may review the manufacturer’s decision. The FDA may not agree with our decisions regarding whether new clearances or approvals
are necessary. If the FDA requires us to seek 510(k) clearance or file a PMA for any modification to a previously cleared product,
we may be required to cease marketing and distributing, or to recall the modified product until we obtain such clearance or approval,
and we may be subject to significant regulatory fines or penalties. Further, our products could be subject to recall if the FDA
determines, for any reason, that our products are not safe or effective. Any recall or FDA requirement that we seek additional
approvals or clearances could result in significant delays, fines, increased costs associated with modification of a product,
loss of revenue and potential operating restrictions imposed by the FDA.
Our products
have in the past been, and may in the future be, subject to product recalls and other similar actions that could harm our reputation,
business and financial results.
The FDA and similar foreign
governmental authorities have the authority to require the recall of regulated products in the event of certain health risks and/or
material deficiencies or defects in design or manufacture. Medical device recalls are typically conducted voluntarily by the manufacturer.
to correct a material product deficiency, improve device performance, or correct violations of applicable FDA regulations. When
a recall is initiated to reduce a risk to health posed by the device or to remedy a violation
of the FD&C Act caused by the device which may present a risk to health, the FDA requires that certain classifications
of recalls be reported to the FDA within 10 working days after the recall is initiated. Companies are required to maintain certain
records of recalls, even if they are not reportable to the FDA.
We have voluntarily initiated
a number of device recalls in the past. For example, on May 14, 2018, we issued a recall of certain QuickFISH products due to
a quality-control failure that occurred prior to distribution, which would have invalidated test results, and, on March 18, 2019,
we issued a recall of a batch of our PNA FISH products due to the potential for diminished performance that could result in an
invalid control result. The 2018 recall was terminated on April 8, 2019, and the 2019 recall was terminated on August 5, 2019.
We may initiate voluntary recalls
involving our products in the future that we determine do not require notification of the FDA. If the FDA disagrees with our determinations,
they could require us to report those actions as recalls. A future recall announcement could harm our reputation with customers
and negatively affect our sales. In addition, the FDA could take enforcement action for failing to report the recalls when they
were conducted. Recalls of any of our products would divert managerial and financial resources and have an adverse effect on our
financial condition and results of operations.
If our products cause
or contribute to a death or a serious injury, or malfunction in certain ways, we will be subject to medical device reporting regulations,
which can result in voluntary corrective actions or agency enforcement actions.
Under the FDA medical device
reporting regulations, medical device manufacturers are required to report to the FDA information that a device has or may have
caused or contributed to a death or serious injury or has malfunctioned in a way that would likely cause or contribute to death
or serious injury if the malfunction of the device or one of our similar devices were to recur. If we fail to report these events
to the FDA within the required timeframes, or at all, the FDA could take enforcement action against us. Any such adverse event
involving our products also could result in future voluntary corrective actions, such as recalls or customer notifications, or
agency action, such as inspection or enforcement action. Any corrective action, whether voluntary or involuntary, as well as defending
ourselves in a lawsuit, will require the dedication of our time and capital, distract management from operating our business,
and may harm our reputation and financial results.
We may generate
a larger portion of our future revenue internationally and would then be subject to increased risks relating to international
activities which could adversely affect our operating results.
We believe that a portion of
our future revenue growth will come from international sources as we implement and expand overseas operations, including South
America and Europe. Engaging in international business involves a number of difficulties and risks, including:
|
|
·
|
required
compliance with existing and changing foreign health care and other regulatory requirements
and laws, such as those relating to patient privacy;
|
|
|
·
|
required
compliance with anti-bribery laws, such as the U.S. Foreign Corrupt Practices Act, or
FCPA, and U.K. Bribery Act, data privacy requirements, labor laws and anti- competition
regulations;
|
|
|
·
|
export
or import restrictions;
|
|
|
·
|
various
reimbursement and insurance regimes;
|
|
|
·
|
laws
and business practices favoring local companies;
|
|
|
·
|
longer
payment cycles and difficulties in enforcing agreements and collecting receivables through
certain foreign legal systems;
|
|
|
·
|
political
and economic instability;
|
|
|
·
|
potentially
adverse tax consequences, tariffs, customs charges, bureaucratic requirements and other
trade barriers;
|
|
|
·
|
foreign
exchange controls;
|
|
|
·
|
difficulties
and costs of staffing and managing foreign operations; and
|
|
|
·
|
difficulties
protecting or procuring intellectual property rights.
|
As we expand internationally,
our results of operations and cash flows would become increasingly subject to fluctuations due to changes in foreign currency
exchange rates. Our expenses are generally denominated in the currencies in which our operations are located, which is in the
United States. If the value of the U.S. dollar increases relative to foreign currencies in the future, in the absence of a corresponding
change in local currency prices, our future revenue could be adversely affected as we convert future revenue from local currencies
to U.S. dollars. If we dedicate resources to our international operations and are unable to manage these risks effectively, our
business, operating results and prospects will suffer.
We
face the risk of potential liability under the FCPA for past international distributions of products and to the extent we distribute
products or otherwise operate internationally in the future.
In the past, we have distributed
certain of our products internationally, and in the future we may distribute our products internationally and possibly engage
in additional international operations. The FCPA prohibits companies such as us from engaging, directly or indirectly, in making
payments to foreign government and political officials for the purpose of obtaining or retaining business or securing any other
improper advantage, including, among other things, the distribution of products and other international business operations. Like
other U.S. companies operating abroad, we may face liability under the FCPA if we, or third parties we have used to distribute
our products or otherwise advance our international business, have violated the FCPA. Any violations of these laws, or allegations
of such violations, could disrupt our operations, involve significant management distraction, involve significant costs and expenses,
including legal fees, and could result in a material adverse effect on our business, prospects, financial condition or results
of operations. We could also suffer severe penalties, including criminal and civil penalties, disgorgement and other remedial
measures.
Risks Related to
Compliance with Healthcare Regulations
Changes in healthcare
policy, including legislation reforming the U.S. healthcare system, may have a material adverse effect on our financial condition
and operations.
In March 2010, both the Patient
Protection and Affordable Care Act, or Affordable Care Act, and the reconciliation law known as Health Care and Education Reconciliation
Act, with the Affordable Care Act, the 2010 Health Care Reform Legislation, were enacted. The constitutionality of the 2010 Health
Care Reform Legislation was confirmed twice by the Supreme Court of the United States. The 2010 Health Care Reform Legislation
has changed the existing state of the health care system by expanding coverage through voluntary state Medicaid expansion, attracting
previously uninsured persons through the new health care insurance exchanges and by modifying the methodology for reimbursing
medical services, drugs and devices. The U.S. Congress is seeking to replace the 2010 Health Care Reform Legislation. At this
time the Company is not certain as to the impact of federal health care legislation on its business.
The
2010 Health Care Reform Legislation includes the Open Payments Act (formerly referred to as the Physician Payments Sunshine Act),
which, in conjunction with its implementing regulations, requires manufacturers of certain drugs, biologics, and devices that
are reimbursed by Medicare, Medicaid and the Children’s Health Insurance Program to report annually certain payments or
“transfers of value” provided to physicians and teaching hospitals and to report annually ownership and investment
interests held by physicians and their immediate family members during the preceding calendar year. Recent amendments to the Open
Payments Act expand the categories of health care providers for which reporting is required. We are evaluating the impact of such
expansion on our business. The failure to report appropriate data accurately, timely, and completely could subject us to significant
financial penalties. Other countries and several states currently have similar laws and more may enact similar legislation.
Further,
the healthcare regulatory environment has seen significant changes in recent years and is still in flux. Legislative
initiatives to modify, limit, replace, or repeal the Healthcare Reform Law and judicial challenges continue, and may increase
in light of the current administration and legislative environment. We cannot predict the impact on our business of
future legislative and legal challenges to the Healthcare Reform Law or other changes to the current laws and regulations. The
financial impact of U.S. healthcare reform legislation over the next few years will depend on a number of factors, including the
policies reflected in implementing regulations and guidance and changes in sales volumes for therapeutics affected by the legislation.
From time to time, legislation is drafted, introduced and passed in the U.S. Congress that could significantly change the statutory
provisions governing coverage, reimbursement, and marketing of pharmaceutical products. In addition, third-party payor coverage
and reimbursement policies are often revised or interpreted in ways that may significantly affect our business and our products.
Since
taking office, President Trump has continued to support the repeal of all or portions of the Healthcare Reform Law. President
Trump has also issued an executive order in which he stated that it is his administration’s policy to seek the prompt repeal
of the Healthcare Reform Law and in which he directed executive departments and federal agencies to waive, defer, grant exemptions
from, or delay the implementation of the provisions of the Healthcare Reform Law to the maximum extent permitted by law. Congress
has enacted legislation that repeals certain portions of the Healthcare Reform Law, including but not limited to the Tax Cuts
and Jobs Act, passed in December 2017, which included a provision that eliminates the penalty under the Healthcare Reform Law’s
individual mandate, effective January 1, 2019, as well as the Bipartisan Budget Act of 2018, passed in February 2018, which, among
other things, repealed the Independent Payment Advisory Board (which was established by the Healthcare Reform Law and was intended
to reduce the rate of growth in Medicare spending). There have also been more recent examples of judicial challenges, such as
federal judges attempting to invalidate the entire Healthcare Reform Law based on the individual mandate. There is still uncertainty
with respect to the impact President Trump’s administration and the U.S. Congress may have, if any, and any changes will
likely take time to unfold.
We
cannot predict whether future healthcare initiatives will be implemented at the federal or state level or in countries outside
of the United States in which we may do business, or the effect any future legislation or regulation will have on us. The taxes
imposed by the new federal legislation and the expansion in government’s effect on the United States healthcare industry
may result in decreased profits to us, which may adversely affect our business, financial condition and results of operations.
We are subject
to potential enforcement actions involving false claims, kickbacks, physician self-referral or other federal or state fraud and
abuse laws, and we could incur significant civil and criminal sanctions, which would hurt our business.
The government has made enforcement
of the false claims, anti-kickback, physician self-referral and various other fraud and abuse laws a major priority. In many instances,
private whistleblowers also are authorized to enforce these laws even if government authorities choose not to do so. In most of
these cases, private whistleblowers brought the allegations to the attention of federal enforcement agencies. The risk of our
being found in violation of these laws and regulations is increased by the fact that some of the laws and regulations have not
been fully interpreted by the regulatory authorities or the courts, and their provisions are open to a variety of interpretations.
We could be subject to enforcement actions under the following laws:
|
|
·
|
the
federal Anti-Kickback Statute, which constrains certain marketing practices, educational
programs, pricing policies and relationships with healthcare providers or other entities
by prohibiting, among other things, soliciting, receiving, offering or paying remuneration,
directly or indirectly, to induce or in return for, the purchase or recommendation of
an item or service reimbursable under a federal healthcare program, such as the Medicare
and Medicaid programs;
|
|
|
·
|
federal
civil and criminal false claims laws and civil monetary penalty laws, which prohibit,
among other things, individuals or entities from knowingly presenting, or causing to
be presented, claims for payment from Medicare, Medicaid, or other third party payors
that are false or fraudulent;
|
|
|
·
|
federal
physician self-referral laws, such as the Stark Law, which prohibit a physician from
making a referral to a provider of certain health services with which the physician or
the physician’s family member has a financial interest, and prohibit submission
of a claim for reimbursement pursuant to a prohibited referral;
|
|
|
·
|
the
federal transparency requirements under The Patient Protection and Affordable Care Act
and the Health Care and Education Reconciliation Act, enacted into law in the United
States in March 2010 (known collectively as the “Affordable Care Act”), including
the provision commonly referred to as the Physician Payments Sunshine Act, which requires
manufacturers of drugs, biologics, devices and medical supplies for which payment is
available under Medicare, Medicaid or the Children’s Health Insurance Program to
report annually to the U.S. Department of Health and Human Services information related
to payments or other transfers of value made to physicians and teaching hospitals, as
well as ownership and investment interests held by physicians and their immediate family
members;
|
|
|
·
|
federal
consumer protection and unfair competition laws, which broadly regulate marketplace activities
and activities that potentially harm consumers; and
|
|
|
·
|
state
law equivalents of each of the above federal laws, such as anti-kickback and false claims
laws, which may apply to items or services reimbursed by any third party payor, including
commercial insurers, many of which differ from each other in significant ways and may
not have the same effect, thus complicating compliance efforts.
|
Efforts to ensure that our
business arrangements with third parties will comply with applicable healthcare laws and regulations will involve substantial
costs. It is possible that governmental authorities will conclude that our business practices do not comply with current or future
statutes, regulations or case law involving applicable fraud and abuse or other healthcare laws and regulations. If we or our
operations are found to be in violation of any of these laws and regulations, we may be subject to penalties, including civil
and criminal penalties, damages, fines, exclusion from participation in U.S. federal or state healthcare programs, such as Medicare
and Medicaid, and the curtailment or restructuring of our operations. We will monitor changes in government enforcement as we
grow and expand our business. Any action against us for violation of these laws, even if we successfully defend against it, could
cause us to incur significant legal expenses, divert our management’s attention from the operation of our business and hurt
our reputation. If we were excluded from participation in U.S. federal healthcare programs, we would not be able to receive, or
to sell our tests to other parties who receive reimbursement from Medicare, Medicaid and other federal programs, and that could
have a material adverse effect on our business.
Risks Related to
Our Intellectual Property
If we cannot license
rights to use technologies on reasonable terms, we may not be able to commercialize new products in the future.
In the future, we may license
third-party technology to develop or commercialize new products. In return for the use of a third party’s technology, we
may agree to pay the licensor royalties based on sales of our solutions. Royalties are a component of cost of services and affect
the margins on our products. We may also need to negotiate licenses to patents and patent applications after introducing a commercial
product. Our business may suffer if we are unable to enter into the necessary licenses on acceptable terms, or at all, if any
necessary licenses are subsequently terminated, if the licensors fail to abide by the terms of the license or fail to prevent
infringement by third parties, or if the licensed patents or other rights are found to be invalid or unenforceable.
If we are unable
to protect our intellectual property effectively, our business would be harmed.
We rely on patent protection
as well as trademark, copyright, trade secret and other intellectual property rights protection and contractual restrictions to
protect our proprietary technologies, all of which provide limited protection and may not adequately protect our rights or permit
us to gain or keep any competitive advantage. If we fail to protect our intellectual property, third parties may be able to compete
more effectively against us and we may incur substantial litigation costs in our attempts to recover or restrict use of our intellectual
property.
In July 2015, we issued a
senior secured promissory note, in the principal amount of $1 million to MGHIF. Such promissory note is secured by a lien on our
assets, including our intellectual property assets. If we default on our payment obligations under this secured promissory note,
MGHIF has the right to control the disposition of our assets, including our intellectual property assets. If such default occurs,
and our intellectual property assets are sold or licensed, our business could be materially adversely affected.
We apply for patents covering
our products and technologies and uses thereof, as we deem appropriate, however we may fail to apply for patents on important
products and technologies in a timely fashion or at all, or we may fail to apply for patents in potentially relevant jurisdictions.
It is possible that none of our pending patent applications will result in issued patents in a timely fashion or at all, and even
if patents are granted, they may not provide a basis for intellectual property protection of commercially viable products, may
not provide us with any competitive advantages, or may be challenged and invalidated by third parties. It is possible that others
will design around our current or future patented technologies. We may not be successful in defending any challenges made against
our patents or patent applications. Any successful third-party challenge to our patents could result in the unenforceability or
invalidity of such patents and increased competition to our business. The outcome of patent litigation can be uncertain and any
attempt by us to enforce our patent rights against others may not be successful, or, if successful, may take substantial time
and result in substantial cost, and may divert our efforts and attention from other aspects of our business.
The patent positions of life
sciences companies can be highly uncertain and involve complex legal and factual questions for which important legal principles
remain unresolved. No consistent policy regarding the breadth of claims allowed in such companies’ patents has emerged to
date in the United States or elsewhere. Courts frequently render opinions in the biotechnology field that may affect the patentability
of certain inventions or discoveries, including opinions that may affect the patentability of methods for analyzing or comparing
DNA.
In particular, the patent
positions of companies engaged in the development and commercialization of genomic diagnostic tests, like ours, are particularly
uncertain. Various courts, including the U.S. Supreme Court, have recently rendered decisions that affect the scope of patentability
of certain inventions or discoveries relating to certain diagnostic tests and related methods. These decisions state, among other
things, that patent claims that recite laws of nature (for example, the relationship between blood levels of certain metabolites
and the likelihood that a dosage of a specific drug will be ineffective or cause harm) are not themselves patentable. What constitutes
a law of nature is uncertain, and it is possible that certain aspects of genetic diagnostics tests would be considered natural
laws. Accordingly, the evolving case law in the United States may adversely affect our ability to obtain patents and may facilitate
third-party challenges to any owned and licensed patents. The laws of some foreign countries do not protect intellectual property
rights to the same extent as the laws of the United States, and we may encounter difficulties protecting and defending such rights
in foreign jurisdictions. The legal systems of many other countries do not favor the enforcement of patents and other intellectual
property protection, particularly those relating to biotechnology, which could make it difficult for us to stop the infringement
of our patents in such countries. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial
cost and divert our efforts and attention from other aspects of our business.
Changes in either the patent
laws or in interpretations of patent laws in the United States or other countries may diminish the value of our intellectual property.
We cannot predict the breadth of claims that may be allowed or enforced in our patents or in third-party patents. We may not develop
additional proprietary products, methods and technologies that are patentable.
In addition to pursuing patents
on our technology, we take steps to protect our intellectual property and proprietary technology by entering into agreements,
including confidentiality agreements, non-disclosure agreements and intellectual property assignment agreements, with our employees,
consultants, academic institutions, corporate partners and, when needed, our advisors. Such agreements may not be enforceable
or may not provide meaningful protection for our trade secrets or other proprietary information in the event of unauthorized use
or disclosure or other breaches of the agreements, and we may not be able to prevent such unauthorized disclosure. If we are required
to assert our rights against such party, it could result in significant cost and distraction.
Monitoring unauthorized disclosure
is difficult, and we do not know whether the steps we have taken to prevent such disclosure are, or will be, adequate. If we were
to enforce a claim that a third party had illegally obtained and was using our trade secrets, it would be expensive and time consuming,
and the outcome would be unpredictable. In addition, courts outside the United States may be less willing to protect trade secrets.
We may also be subject to
claims that our employees have inadvertently or otherwise used or disclosed trade secrets or other proprietary information of
third parties, or to claims that we have improperly used or obtained such trade secrets. Litigation may be necessary to defend
against these claims. If we fail in defending such claims, in addition to paying monetary damages, we may lose valuable intellectual
property rights and face increased competition to our business. A loss of key research personnel work product could hamper or
prevent our ability to commercialize potential products, which could harm our business. Even if we are successful in defending
against these claims, litigation could result in substantial costs and be a distraction to management.
Further, competitors could
attempt to replicate some or all of the competitive advantages we derive from our development efforts, willfully infringe our
intellectual property rights, design around our protected technology or develop their own competitive technologies that fall outside
of our intellectual property rights. Others may independently develop similar or alternative products and technologies or replicate
any of our products and technologies. If our intellectual property does not adequately protect us against competitors’ products
and methods, our competitive position could be adversely affected, as could our business.
We have not yet registered
certain of our trademarks in all of our potential markets. If we apply to register these trademarks, our applications may not
be allowed for registration in a timely fashion or at all, and our registered trademarks may not be maintained or enforced. In
addition, opposition or cancellation proceedings may be filed against our trademark applications and registrations, and our trademarks
may not survive such proceedings. If we do not secure registrations for our trademarks, we may encounter more difficulty in enforcing
them against third parties than we otherwise would.
To the extent our intellectual
property offers inadequate protection, or is found to be invalid or unenforceable, we would be exposed to a greater risk of direct
competition. If our intellectual property does not provide adequate coverage of our competitors’ products, our competitive
position could be adversely affected, as could our business. Both the patent application process and the process of managing patent
disputes can be time consuming and expensive.
We may be involved
in litigation related to intellectual property, which could be time-intensive and costly and may adversely affect our business,
operating results or financial condition.
We may receive notices of claims
of direct or indirect infringement or misappropriation or misuse of other parties’ proprietary rights from time to time.
Some of these claims may lead to litigation. We cannot assure you that we will prevail in such actions, or that other actions
alleging misappropriation or misuse by us of third-party trade secrets, infringement by us of third-party patents and trademarks
or other rights, or challenging the validity of our patents, trademarks or other rights, will not be asserted or prosecuted against
us.
We might not have been the
first to make the inventions covered by each of our pending patent applications and we might not have been the first to file patent
applications for these inventions. To determine the priority of these inventions, we may have to participate in interference proceedings,
derivation proceedings, or other post-grant proceedings declared by the United States Patent and Trademark Office that could result
in substantial cost to us. No assurance can be given that other patent applications will not have priority over our patent applications.
In addition, recent changes to the patent laws of the United States allow for various post-grant opposition proceedings that have
not been extensively tested, and their outcome is therefore uncertain. Furthermore, if third parties bring these proceedings against
our patents, we could experience significant costs and management distraction.
Litigation may be necessary
for us to enforce our patent and proprietary rights or to determine the scope, coverage and validity of the proprietary rights
of others. The outcome of any litigation or other proceeding is inherently uncertain and might not be favorable to us, and we
might not be able to obtain licenses to technology that we require on acceptable terms or at all. Further, we could encounter
delays in product introductions, or interruptions in product sales, as we develop alternative methods or products. In addition,
if we resort to legal proceedings to enforce our intellectual property rights or to determine the validity, scope and coverage
of the intellectual property or other proprietary rights of others, the proceedings could be burdensome and expensive, even if
we were to prevail. Any litigation that may be necessary in the future could result in substantial costs and diversion of resources
and could have a material adverse effect on our business, operating results or financial condition.
As we move into new markets
and applications for our products, incumbent participants in such markets may assert their patents and other proprietary rights
against us as a means of slowing our entry into such markets or as a means to extract substantial license and royalty payments
from us. Our competitors and others may now and, in the future, have significantly larger and more mature patent portfolios than
we currently have. In addition, future litigation may involve patent holding companies or other adverse patent owners who have
no relevant product revenue and against whom our own patents may provide little or no deterrence or protection. Therefore, our
commercial success may depend in part on our non-infringement of the patents or proprietary rights of third parties. Numerous
significant intellectual property issues have been litigated, and will likely continue to be litigated, between existing and new
participants in our existing and targeted markets and competitors may assert that our products infringe their intellectual property
rights as part of a business strategy to impede our successful entry into or growth in those markets. Third parties may assert
that we are employing their proprietary technology without authorization. In addition, our competitors and others may have patents
or may in the future obtain patents and claim that making, having made, using, selling, offering to sell or importing our products
infringes these patents. We could incur substantial costs and divert the attention of our management and technical personnel in
defending against any of these claims. Parties making claims against us may be able to obtain injunctive or other relief, which
could block our ability to develop, commercialize and sell products, and could result in the award of substantial damages against
us. In the event of a successful claim of infringement against us, we may be required to pay damages and ongoing royalties, and
obtain one or more licenses from third parties, or be prohibited from selling certain products. We may not be able to obtain these
licenses on acceptable terms, if at all. We could incur substantial costs related to royalty payments for licenses obtained from
third parties, which could negatively affect our financial results. In addition, we could encounter delays in product introductions
while we attempt to develop alternative methods or products to avoid infringing third-party patents or proprietary rights. Defense
of any lawsuit or failure to obtain any of these licenses could prevent us from commercializing products, and the prohibition
of sale of any of our products could materially affect our business and our ability to gain market acceptance for our products.
Furthermore, because of the
substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our
confidential information could be compromised by disclosure during this type of litigation. In addition, during the course of
this kind of litigation, there could be public announcements of the results of hearings, motions or other interim proceedings
or developments. If securities analysts or investors perceive these results to be negative, it could have a substantial adverse
effect on the price of our common stock.
In addition, our agreements
with some of our customers, suppliers or other entities with whom we do business require us to defend or indemnify these parties
to the extent they become involved in infringement claims, including the types of claims described above. We could also voluntarily
agree to defend or indemnify third parties in instances where we are not obligated to do so if we determine it would be important
to our business relationships. If we are required or agree to defend or indemnify third parties in connection with any infringement
claims, we could incur significant costs and expenses that could adversely affect our business, operating results, or financial
condition.
INFORMATION
REGARDING FORWARD-LOOKING STATEMENTS
This prospectus includes forward-looking
statements. All statements other than statements of historical facts contained in this prospectus, including statements regarding
our future results of operations and financial position, strategy and plans, and our expectations for future operations, are forward-looking
statements. The words “believe,” “may,” “will,” “estimate,” “continue,”
“anticipate,” “design,” “intend,” “expect” or the negative version of these words
and similar expressions are intended to identify forward-looking statements. We have based these forward-looking statements on
our current expectations and projections about future events and trends that we believe may affect our financial condition, results
of operations, strategy, short- and long-term business operations and objectives, and financial needs. These forward-looking statements
are subject to a number of risks, uncertainties and assumptions, including those described in “Risk Factors.” In light
of these risks, uncertainties and assumptions, the forward-looking events and circumstances discussed in this prospectus may not
occur, and actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements.
Given these uncertainties, you should not place undue reliance on these forward-looking statements. Forward-looking statements
include, but are not limited to, statements about:
|
|
·
|
our
need to raise capital and the uses of such capital, including funding the Interim Facility;
|
|
|
·
|
our
ability to successfully complete and close the transaction with Curetis;
|
|
|
·
|
our
need to apply a significant amount of the proceeds of this offering to support Curetis
operations, and our ability to be repaid if the transaction does not close;
|
|
|
·
|
the
commercialization of Newco’s products;
|
|
|
·
|
the
completion of the development efforts for the Acuitas AMR Gene Panel tests and Acuitas
Lighthouse Software, and the timing of FDA 510(k) clearance filings;
|
|
|
·
|
our
ability to successfully integrate the OpGen and Curetis businesses;
|
|
|
·
|
our
liquidity and working capital requirements, including cash requirements over the next
12 months for us and Newco;
|
|
|
·
|
our
ability to regain compliance with the ongoing listing requirements for the Nasdaq Capital
Market;
|
|
|
·
|
anticipated
trends and challenges in our business and the competition that we face;
|
|
|
·
|
the
execution of Newco’s business plan and growth strategy;
|
|
|
·
|
Newco’s
expectations regarding the size of and growth in potential markets;
|
|
|
·
|
Newco’s
opportunity to successfully enter into new collaborative agreements;
|
|
|
·
|
regulations
and changes in laws or regulations applicable to our business, including regulation by
the FDA and the EU;
|
|
|
·
|
compliance
with the U.S. and international regulations applicable to our business; and
|
|
|
·
|
our
expectations regarding future revenue and expenses.
|
Although we believe that the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future
results, level of activity, performance or achievements. In addition, neither we nor any other person assumes responsibility for
the accuracy and completeness of any of these forward-looking statements. These risks should not be construed as exhaustive and
should be read in conjunction with our other disclosures, including but not limited to the risk factors described in this prospectus.
Other risks may be described from time to time in our filings made under the securities laws. New risks emerge from time to time.
It is not possible for our management to predict all risks. All forward-looking statements in this prospectus speak only as of
the date made and are based on our current beliefs and expectations. We undertake no obligation to update or revise any forward-looking
statement, whether as a result of new information, future events or otherwise.
These factors should not be
construed as exhaustive and should be read in conjunction with our other disclosures, including but not limited to the risk factors
described in this prospectus. Other risks may be described from time to time in our filings made under the securities laws. New
risks emerge from time to time. It is not possible for our management to predict all risks. All forward-looking statements in
this prospectus speak only as of the date made and are based on our current beliefs and expectations. We undertake no obligation
to update or revise any forward-looking statement, whether as a result of new information, future events or otherwise, except
to the extent required by applicable securities laws.
USE
OF PROCEEDS
We estimate the net proceeds from this offering will
be approximately $8.9 million, based on an assumed public offering price of $6.55 per unit, the closing price of our common stock
on the Nasdaq Capital Market on October 9, 2019, and assuming the sale of 1,526,717 units and no sale of any pre-funded units
in this offering, after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by
us. If the underwriter exercises its option to purchase additional securities in full, we estimate the net proceeds from this
offering will be approximately $10.4 million from the sale of our securities, based on an assumed public offering price of $6.55
per unit and assuming no sale of any pre-funded units in this offering, after deducting estimated underwriting discounts and commissions
and estimated offering expenses payable by us. These estimates exclude the proceeds, if any, from the exercise of common warrants
in this offering. The public offering price per unit will be determined between us and the underwriter based on market conditions
at the time of pricing and may be at a discount to the current market price of our common stock.
The primary programs and activities
to which we intend to devote the net proceeds of this offering are:
Prior to the closing of the
transactions under the Implementation Agreement:
|
|
·
|
for completing the business combination with
Curetis;
|
|
|
·
|
for provision of short-term funding to Curetis
under the Interim Facility to fund the Curetis Group’s operations, including
the Unyvero LRT BAL regulatory clearance-related activities, Unyvero platform
R&D activities, Ares Genetics-related R&D and business development activities
with potential collaborators and distributors; and
|
|
|
·
|
for research and development and regulatory
activities in support of the Company’s anticipated FDA 510(k) submissions for
the Acuitas AMR Gene Panel test and the Acuitas Lighthouse Software.
|
If any proceeds remain, following
the closing of the transactions under the Implementation Agreement:
|
|
·
|
commercialization
of Newco’s products, with a focus on the Unyvero platform and diagnostic tests,
and the Acuitas AMR Gene Panel tests;
|
|
|
·
|
further
development and commercialization of the Ares Genetics database and Acuitas Lighthouse
Software;
|
|
|
·
|
directed
efforts to the customers and collaborators of each company to introduce the products
and services of Newco;
|
|
|
·
|
investments
in manufacturing and operations infrastructure to support sales of products; and
|
|
|
·
|
the
balance for general corporate purposes, such as general and administrative expenses,
capital expenditures and working capital needs.
|
If the transactions under
the Implementation Agreement do not close, and to the extent any proceeds remain, we plan to use any remaining proceeds to support
OpGen’s operations as far as possible into 2020.
Many of the material terms of the Interim Facility, including
the interest rate, term and committed amount, have not yet been negotiated, and we may be unable to negotiate favorable terms.
The expected use of net proceeds
of this offering represents our current intentions based upon our present plan and business conditions. We may use a portion of
the net proceeds for the acquisitions of businesses, products, technologies or licenses that are complementary to our business,
although we have no present commitments or agreements to do so. As of the date of this prospectus, we cannot specify with certainty
all of the particular uses for the net proceeds to be received upon the completion of this offering. Our management will have
broad discretion in the application of the net proceeds, and investors will be relying on the judgment of our management regarding
the application of the proceeds of this offering.
A $1.00 increase (decrease) in the assumed public offering
price of $6.55 per unit would increase (decrease) the expected net proceeds to us from this offering by approximately $1.4 million,
assuming that the number of units offered by us, as set forth on the cover page of this prospectus, remains the same and after
deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us and excluding the
proceeds, if any, from the exercise of the common warrants issued in this offering.
Similarly, a 100,000 unit increase or decrease in the
number of units offered by us, as set forth on the cover page of this prospectus, would increase or decrease the net proceeds
to us by approximately $0.6 million, assuming the assumed public offering price of $6.55 per unit remains the same, and after
deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us and excluding the proceeds,
if any, from the exercise of the common warrants issued pursuant to this offering.
CAPITALIZATION
The following table sets forth
our cash and cash equivalents and capitalization as of June 30, 2019 as follows:
|
|
·
|
on
an actual basis; and
|
|
|
·
|
on
an as adjusted basis to give effect to the sale by us of 1,526,717 units in this offering at an assumed public offering
price of $6.55 per unit, which is the last reported sale price of our common stock on the Nasdaq Capital Market on October 9, 2019,
after deducting the estimated underwriting discounts and commissions and estimated offering expenses, and assuming no sale of any
pre-funded units in this offering and excluding the proceeds, if any, from the exercise of common warrants issued in this offering.
|
The as adjusted information
set forth below is illustrative only and will be adjusted based on the actual public offering price and other terms of this offering
determined at pricing. You should read this table in conjunction with “Management’s Discussion and Analysis of Financial
Condition and Results of Operations” and our financial statements and related notes included elsewhere in, or incorporated
by reference into, this prospectus.
|
|
|
As of June 30, 2019
|
|
|
|
Actual
|
|
As Adjusted
|
|
|
|
(In thousands, except share and per share data)
|
|
Cash and cash equivalents
|
|
$
|
3,056
|
|
|
$
|
11,950
|
|
|
Debt
|
|
$
|
838
|
|
|
$
|
838
|
|
|
Stockholders’ equity:
|
|
|
|
|
|
|
|
|
|
Common stock, par value $0.01 per share: 50,000,000 shares authorized, 882,268 shares issued and outstanding, actual; 50,000,000 shares authorized, 2,408,985 issued and outstanding, as adjusted
|
|
|
9
|
|
|
|
24
|
|
|
Preferred stock, par value $0.01 per share; 10,000,000 shares
|
|
|
—
|
|
|
|
—
|
|
|
authorized, no shares outstanding, actual and as adjusted
|
|
|
|
|
|
|
|
|
|
Additional paid-in capital
|
|
|
170,358
|
|
|
|
179,237
|
|
|
Accumulated other comprehensive loss
|
|
|
(15
|
)
|
|
|
(15
|
)
|
|
Accumulated deficit
|
|
|
(168,525
|
)
|
|
|
(168,525
|
)
|
|
Total stockholders’ equity
|
|
|
1,827
|
|
|
|
10,721
|
|
|
Total capitalization
|
|
$
|
2,665
|
|
|
$
|
11,559
|
|
A $1.00 increase (decrease) in the assumed public offering
price of $6.55 per unit would increase (decrease) the expected net proceeds to us from this offering by approximately $1.4 million,
assuming that the number of units offered by us, as set forth on the cover page of this prospectus, remains the same and after
deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us and excluding the
proceeds, if any, from the exercise of the common warrants issued pursuant to this offering.
Similarly, a 100,000 unit increase or decrease in the
number of units offered by us, as set forth on the cover page of this prospectus, would increase or decrease the net proceeds
to us by approximately $0.6 million, assuming the assumed public offering price of $6.55 per unit remains the same, and after
deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us and excluding the proceeds,
if any, from the exercise of the common warrants issued pursuant to this offering.
The number of shares of common
stock to be outstanding immediately after this offering is based on 882,268 shares of our common stock outstanding as of June
30, 2019, and excludes:
|
|
·
|
10,542
shares of common stock issuable upon the exercise
of outstanding options granted as of June 30, 2019, under our equity incentive plans
at a weighted average exercise price of $410.31 per share;
|
|
|
·
|
175,982
shares of common stock issuable upon the
exercise of outstanding warrants issued as of June 30, 2019, at a weighted average exercise
price of $288.25 per share;
|
|
|
·
|
15,663
shares of common stock issuable upon vesting of outstanding restricted stock units granted
as of June 30, 2019; and
|
|
|
·
|
4,221
shares of common stock available for future issuance under our equity incentive plans
as of June 30, 2019;
|
|
|
·
|
76,335 shares
of common stock, or 87,786 shares of common stock if the underwriter exercises its option
to purchase additional securities in full, issuable upon exercise of warrants to
be issued to the underwriter at an exercise price of 130% of the public offering price
as described in “Underwriting;”
|
|
|
·
|
1,526,717
shares of common stock issuable upon the exercise of the common warrants to be issued
to purchasers in this offering at an exercise price of $ per
share; and
|
|
|
·
|
up
to 2,662,564 shares of common stock issuable pursuant to the terms of the Implementation
Agreement.
|
The number
of outstanding options, restricted stock units and shares of common stock available for future issuances under our equity incentive
plans does not reflect grants of 1,500 shares of common stock issuable upon vesting of restricted stock units, the expiration
of stock options to purchase 499 shares of our common stock, forfeitures of stock options to purchase 107 shares of our common
stock or forfeitures of 500 shares of our common stock issuable upon vesting of restricted stock units since June 30, 2019.
Unless otherwise indicated, all information
contained in this prospectus assumes (i) that the underwriter has not exercised its option to purchase additional securities, (ii)
no sale of pre-funded units in this offering, which, if sold, would reduce the number of units that we are offering on a one-for-one
basis, and (iii) no exercise of options issued under our equity incentive plans or of warrants, including the warrants included
in the units or pre-funded units offered hereby and the underwriter’s warrants to be issued to the underwriter in connection
with this offering.
DILUTION
Our net tangible book value as of June 30, 2019 was approximately
$274 thousand, or $0.31 per share. Net tangible book value per share is determined by dividing our total tangible assets, less
total liabilities, by the number of shares of our common stock outstanding as of June 30, 2019. Dilution with respect to net tangible
book value per share represents the difference between the amount per unit paid by purchasers in this offering and the net tangible
book value per share of our common stock immediately after this offering.
After giving effect to the assumed sale of 1,526,717
units in this offering at an assumed public offering price of $6.55 per unit, based on the last reported sale price of our common
stock on the Nasdaq Capital Market on October 9, 2019, assuming no sale of any pre-funded units in this offering and after deducting
estimated underwriting fees and estimated offering expenses payable by us, our as adjusted net tangible book value as of June
30, 2019 would have been approximately $9.2 million, or $3.81 per share. This represents an immediate increase in net tangible
book value of $3.50 per share to existing stockholders and immediate dilution of $2.74 per share to purchasers purchasing our
securities in this offering at the public offering price. The following table illustrates this dilution on a per share basis:
|
Assumed public offering price per unit
|
|
|
|
|
|
$
|
6.55
|
|
|
Net tangible book value per share of as June 30, 2019
|
|
$
|
0.31
|
|
|
|
|
|
|
Increase in net tangible book value per
share attributable to this offering
|
|
$
|
3.50
|
|
|
|
|
|
|
As adjusted net tangible book value per
share as of June 30, 2019, after giving effect to this offering
|
|
|
|
|
|
$
|
3.81
|
|
|
Dilution per share to investors
in this offering
|
|
|
|
|
|
$
|
2.74
|
|
A $1.00 increase in the assumed public offering price
of $6.55 per unit, based on the last reported sale price for our common stock as reported on the Nasdaq Capital Market on October
9, 2019, would increase our as adjusted net tangible book value after this offering by $1.4 million, or $0.59 per share, and the
dilution per share to investors purchasing securities in this offering would be approximately $3.15 per share, assuming that the
number of units, as set forth on the cover page of this prospectus, remains the same and after deducting the estimated underwriting
discounts and commissions and estimated offering expenses payable by us and excluding the proceeds, if any, from exercise of the
common warrants issued in this offering. Similarly, a $1.00 decrease in the assumed public offering price of $6.55 per unit would
decrease our as adjusted net tangible book value after this offering by $1.4 million, or $0.59 per share, and the dilution per
share to investors purchasing securities in this offering would be $2.33 per share, assuming that the number of units offered
by us, as set forth on the cover page of this prospectus remains the same and after deducting the estimated underwriting discounts
and commissions and estimated offering expenses payable by us and excluding the proceeds, if any, from exercise of the common
warrants issued in this offering.
We may also increase or decrease the number of units
we are offering. An increase of 100,000 units in the number units offered by us would increase our as adjusted net tangible book
value by approximately $0.7 million, or $0.11 per unit, and decrease the dilution per share to purchasers participating in this
offering by $0.11 per share, assuming the assumed offering price per unit remains the same and after deducting the estimated underwriting
fees and estimated offering expenses payable by us and excluding the proceeds, if any, from exercise of the common warrants issued
in this offering. Similarly, a decrease of 100,000 units in the number of units offered by us would decrease our as adjusted net
tangible book value by approximately $0.7 million or $0.12 per share, and increase the dilution per share to purchasers participating
in this offering by $0.12 per share, assuming the assumed offering price per unit remains the same and after deducting the estimated
underwriting fees and estimated offering expenses payable by us and excluding the proceeds, if any, from exercise of the common
warrants issued in this offering. The information discussed above is illustrative only and will adjust based on the actual offering
price, the actual number of units we offer in this offering, and other terms of this offering determined at pricing.
If the underwriter exercises its option to purchase additional
shares of common stock and/or common warrants to purchase common stock in full, the as adjusted net tangible book value per share
after giving effect to this offering would be $4.00 per share, and the dilution in net tangible book value per share to new purchasers
in this offering would be $2.55 per share.
The number of shares of common stock to be outstanding immediately
after this offering is based on 882,268 shares of our common stock outstanding as of June 30, 2019, and excludes:
|
|
·
|
10,542
shares of common stock issuable upon the exercise
of outstanding options granted as of June 30, 2019, under our equity incentive plans
at a weighted average exercise price of $410.31 per share;
|
|
|
·
|
175,982
shares of common stock issuable upon the
exercise of outstanding warrants issued as of June 30, 2019, at a weighted average exercise
price of $288.25 per share;
|
|
|
·
|
15,663
shares of common stock issuable upon vesting of outstanding restricted stock units granted
as of June 30, 2019; and
|
|
|
·
|
4,221
shares of common stock available for future issuance under our equity incentive plans
as of June 30, 2019;
|
|
|
·
|
76,335 shares
of common stock, or 87,786 shares of common stock if the underwriter exercises its option
to purchase additional securities in full, issuable upon exercise of warrants to be issued
to the underwriter at an exercise price of 130% of the public offering price as described
in “Underwriting;”
|
|
|
·
|
1,526,717
shares of common stock issuable upon the exercise of the common warrants to be issued
to purchasers in this offering at an exercise price of $ per
share; and
|
|
|
·
|
up
to 2,662,564 shares of common stock issuable pursuant to the terms of the Implementation
Agreement.
|
The number
of outstanding options, restricted stock units and shares of common stock available for future issuances under our equity incentive
plans does not reflect grants of 1,500 shares of common stock issuable upon vesting of restricted stock units, the expiration
of stock options to purchase 499 shares of our common stock, forfeitures of stock options to purchase 107 shares of our common
stock or forfeitures of 500 shares of our common stock issuable upon vesting of restricted stock units since June 30, 2019.
Unless otherwise indicated, all information contained
in this prospectus assumes (i) that the underwriter has not exercised its option to purchase additional securities, (ii) no sale
of pre-funded units in this offering, which, if sold, would reduce the number of units that we are offering on a one-for-one basis,
and (iii) no exercise of options issued under our equity incentive plans or of warrants, including the underwriter’s warrants
to be issued to the underwriter in connection with this offering.
PRICE
RANGE FOR OUR COMMON EQUITY AND RELATED SHAREHOLDER MATTERS
Market
Information
Our common stock and warrants
we issued in our initial public offering, or the IPO Warrants, have traded on the Nasdaq Capital Market under the symbols “OPGN”
and “OPGNW,” respectively, since May 5, 2015. Prior to such time, there was no public market for our common stock
or our warrants.
Stockholder
Information
On October 16, 2019, the last reported sale price of
our common stock on the Nasdaq Capital Market was $5.99 per share. As of October 16, 2019, there were approximately 27 stockholders
of record of our common stock, which does not include stockholders that beneficially own shares held in a “nominee”
or in “street” name.
DIVIDEND
POLICY
We have never paid or declared
any cash dividends on our common stock. We do not anticipate paying any cash dividends on our common stock in the foreseeable
future, and we intend to retain all available funds and any future earnings to fund the development and expansion of our business.
Any future determination to pay dividends will be at the discretion of our board of directors and will depend upon a number of
factors, including our results of operations, financial condition, future prospects, contractual restrictions, restrictions imposed
by applicable law and other factors our board of directors deems relevant.
MARKET
AND INDUSTRY DATA
This prospectus and the
documents incorporated by reference in this prospectus contain market data and industry statistics that are based on independent
industry publications and other publicly available information. Although we believe that these sources are reliable, we do not
guarantee the accuracy or completeness of the information and we have not independently verified this information. Although we
are not aware of any misstatements regarding the market and industry data presented or incorporated by reference in this prospectus,
these estimates involve risks and uncertainties and are subject to change based on various factors, including those discussed
in the section titled “Risk Factors” or incorporated by reference herein, and any related free writing prospectus.
Accordingly, purchasers should not place undue reliance on this information.
DESCRIPTION
OF CAPITAL STOCK
Our authorized capital stock
consists of 50,000,000 shares of common stock, par value $0.01 per share, and 10,000,000 shares of preferred stock, par value
$0.01 per share, of which 7,690,572 shares are available for issuance. The following is a summary of the rights of our common
and preferred stock, and some of the provisions of our amended and restated certificate of incorporation and amended and restated
bylaws and the Delaware General Corporation Law. Because it is only a summary, it does not contain all of the information that
may be important to you. Such summary is subject to and qualified in its entirety by our amended and restated certificate of incorporation
and our amended and restated bylaws, a copy of each of which has been incorporated as an exhibit to the registration statement
of which this prospectus forms a part.
Following
receipt of approval from stockholders at a special meeting held on January 17, 2018, we filed an amendment to our Amended and
Restated Certificate of Incorporation to effect a reverse stock split, or the 2018 Reverse Stock Split, of the issued and outstanding
shares of our common stock, at a ratio of one share for each twenty-five shares outstanding, and to reduce the authorized shares
of common stock from 200,000,000 to 50,000,000 shares. In implementing the 2018 Reverse Stock Split, the number of shares of our
common stock held by each stockholder was reduced by dividing the number of shares held immediately before the 2018 Reverse Stock
Split by twenty-five and then rounding down to the nearest whole share. We paid cash to each stockholder in lieu of any fractional
interest in a share to which each stockholder would otherwise be entitled as a result of the 2018 Reverse Stock Split. The 2018
Reverse Stock Split did not affect any stockholder’s percentage ownership interest in our Company or proportionate voting
power, except to the extent that interests in fractional shares were paid in cash. At the time of the 2018 Reverse Stock Split,
we also adjusted all then outstanding shares underlying restricted stock units, stock options and warrants.
On August
22, 2019, at the annual meeting of stockholders, our stockholders approved an amendment to our Amended and Restated Certificate
of Incorporation, authorizing a further reverse stock split, or the 2019 Reverse Stock Split, of the issued and outstanding shares
of our common stock, at a ratio within a range of not less than five-to-one and not more than twenty-five-to-one, such ratio and
the implementation and timing of such reverse stock split to be determined in the discretion of our Board of Directors.
On August 22, 2019, our Board of Directors approved the 2019 Reverse Stock Split of one share for twenty outstanding shares. On
August 28, 2019, we filed an Amendment to our Amended and Restated Certificate of Incorporation to effect the 2019 Reverse Stock
Split. The 2019 Reverse Stock Split became effective on Nasdaq on August 29, 2019. In implementing the 2019 Reverse Stock Split,
the number of shares of our common stock held by each stockholder was reduced by dividing the number of shares held immediately
before the 2019 Reverse Stock Split by twenty. We paid cash to each stockholder in lieu of any fractional shares to which each
stockholder would otherwise be entitled as a result of the 2019 Reverse Stock Split. The 2019 Reverse Stock Split did not affect
any stockholder’s percentage ownership interest in our Company or proportionate voting power, other than to the extent of
fractional shares settled in cash.
In addition,
we have adjusted all outstanding shares underlying all outstanding restricted stock units, stock options and warrants entitling
the holders to purchase shares of our common stock, as required by the terms of these securities. In particular, we have reduced
the number of underlying shares, and increased the exercise price in accordance with the terms of each security based on 2019
Reverse Stock Split ratio (i.e., the number of shares issuable under such securities have been divided by twenty, and the exercise
price per share has been multiplied by twenty). Also, we reduced the number of shares reserved for issuance under our existing
2015 Equity Incentive Plan, or the 2015 Plan, based on the 2019 Reverse Stock Split ratio. The 2019 Reverse Stock Split does not
otherwise affect any of the rights currently accruing to holders of our common stock, or restricted stock units, stock options
or warrants exercisable for our common stock.
Common
Stock
As of October 16, 2019,
there were 882,268 shares outstanding, 202,581 shares of common stock reserved for the exercise of outstanding stock options,
warrants and restricted stock units, and approximately 27 record holders. The holders of our common stock are entitled to one
vote for each share held on all matters submitted to a vote of the stockholders. The holders of our common stock do not have any
cumulative voting rights. The Board of Directors are elected to a one year term; the Company does not have a staggered board.
Holders of our common stock are entitled to receive ratably any dividends declared by the Board of Directors out of funds legally
available for that purpose, subject to any preferential dividend rights of any outstanding preferred stock. Our common stock has
no preemptive rights, conversion rights or other subscription rights or redemption or sinking fund provisions.
In the event of our liquidation,
dissolution or winding up, holders of our common stock will be entitled to share ratably in all assets remaining after payment
of all debts and other liabilities and any liquidation preference of any outstanding preferred stock.
Preferred
Stock
Series
A Convertible Preferred Stock
Of the authorized preferred
stock, the Company had previously issued 2,309,428 shares of Series A Convertible Preferred Stock. The holder of the Series A
Convertible Preferred Stock converted all 2,309,428 shares of Series A Convertible Preferred Stock into shares of common stock.
All such converted shares of Series A Convertible Preferred Stock were canceled and will not be reissued. As of October 9, 2019,
no shares of the Series A Convertible Preferred Stock were outstanding.
Additional
Series of Preferred Stock
Our Board of Directors has
the authority, without further action by our stockholders, to issue from time to time 7,690,572 shares of preferred stock in one
or more series. Our Board of Directors will have the authority to establish the number of shares to be included in each series
and fix the powers, preferences and rights of the shares of each wholly unissued series and any of its qualifications, limitations
or restrictions. Our Board of Directors will also be able to increase or decrease the number of shares of any series, but not
below the number of shares of that series then outstanding, without any further vote or action by the stockholders.
The issuance of preferred
stock could decrease the amount of earnings and assets available for distribution to the holders of common stock or adversely
affect the rights and powers, including voting rights, of the holders of common stock. The issuance of preferred stock, while
providing flexibility in connection with possible acquisitions and other corporate purposes, could, among other things, have the
effect of delaying, deferring or preventing a change in control of our Company, which could depress the market price of our common
stock. We have no current plans to issue any additional shares of preferred stock.
Outstanding
Warrants
IPO
Warrants
The warrants to purchase common
stock that we issued in our initial public offering, or the IPO Warrants, entitle the registered holder to purchase one share
of common stock at a price equal to $3,300.00 per share, subject to adjustment as discussed below, immediately following the issuance
of such IPO Warrants and terminate at 5:00 p.m., New York City time, on May 8, 2020 or earlier upon the dissolution or winding
up of the Company. We have listed the IPO Warrants on the Nasdaq Capital Market, as a standalone security under the symbol “OPGNW.”
The IPO Warrants were issued
pursuant to a Warrant Agreement between us and our transfer agent as the Warrant Agent. The exercise price and number of shares
of common stock issuable upon exercise of the IPO Warrants may be adjusted in certain circumstances, including in the event of
a stock dividend or recapitalization, reorganization, merger or consolidation.
The IPO Warrants may be exercised
upon surrender of the applicable Warrant Certificate on or prior to the applicable expiration date at the offices of the Warrant
Agent, with the exercise form on the reverse side of the Warrant Certificate completed and executed as indicated, accompanied
by full payment of the exercise price, by certified or official bank check payable to us, unless such holders are willing to exercise
their IPO Warrants on a cashless basis, as further described in this Warrant Agreement, for the number of IPO Warrants being exercised.
Under the terms of the Warrant Agreement, we have agreed to use our reasonable best efforts to maintain the effectiveness of a
registration statement and prospectus relating to common stock issuable upon exercise of the IPO Warrants until the expiration
of the IPO Warrants. The IPO Warrant holders do not have the rights or privileges of holders of common stock or any voting rights
until they exercise their IPO Warrants and receive shares of common stock. After the issuance of shares of common stock upon exercise
of the IPO Warrants, each holder will be entitled to one vote for each share held of record on all matters to be voted on by stockholders.
A holder may not exercise
any portion of an IPO Warrant to the extent that the holder, together with its affiliates and any other person or entity acting
as a group, would own more than 4.99% of the outstanding common stock after exercise, as such percentage ownership is determined
in accordance with the terms of the IPO Warrant. The foregoing limitation on exercise shall not apply to any registered holder
of an IPO Warrant who, together with his, her or its affiliates, and any persons acting as a group together with such registered
holder and such registered holder’s affiliates, owns in excess of 4.99% immediately prior to the closing of this offering.
In addition, upon at least 61 days’ prior notice from the holder to us, the holder may waive such limitation.
No fractional shares of common
stock will be issued upon exercise of the IPO Warrants. If, upon exercise of the IPO Warrant, a holder would be entitled to receive
a fractional interest in a share, we will, upon exercise, round to the nearest whole number of shares of common stock to be issued
to the IPO Warrant holder. If multiple IPO Warrants are exercised by the holder at the same time, we will aggregate the number
of whole shares issuable upon exercise of all the IPO Warrants.
2016
PIPE Warrants
Pursuant to the terms of the
Amended & Restated Purchase Agreement, dated as of May 18, 2016, by and among the Company and the purchasers party thereto,
the purchasers purchased 18,107 warrants, or the PIPE Warrants, exercisable for an aggregate of 13,580 shares of common stock,
or the PIPE Warrant Shares, in the PIPE Financing. The PIPE Warrants are exercisable at an exercise price of $656.25 per share
of common stock, became exercisable 90 days after the date of issuance, and may be exercised for five years from the date of issuance.
The exercise price and the number of PIPE Warrant Shares will be adjusted to account for the subdivision or combination by the
Company of outstanding shares of common stock. The exercise price may, at any time, also be voluntarily reduced at the discretion
of the Board of Directors of the Company. The PIPE Warrants may be exercised pursuant to a cashless exercise, but only if a registration
statement covering the resale of the PIPE Warrant Shares that are the subject of an exercise notice is not available for the resale
of such PIPE Warrant Shares.
The PIPE Warrants also contain
certain provisions providing for liquidated damages to be paid by the Company in the event the Company does not timely deliver
registered shares of common stock to the holder upon exercise of a PIPE Warrant. Specifically, in addition to the PIPE Warrant
holder’s other available remedies, if the Company fails to issue and deliver (or cause to be delivered) to a holder by the
required delivery date a certificate representing the shares so delivered to the Company by such holder that is free from all
restrictive and other legends, the Company shall pay to a holder in cash, as partial liquidated damages and not as a penalty,
an amount equal to 1% of the product of (A) the aggregate number of shares of common stock not issued to the holder on a timely
basis and to which the holder is entitled and (B) the closing sale price on the trading day immediately preceding the required
delivery date of the certificate, per trading day for each trading day after such required delivery date until such securities
are delivered to the holder. In addition, if the Company fails to (i) issue and deliver (or cause to be delivered) to a holder
by the required delivery date a certificate representing the shares so delivered to the Company by such holder that is free from
all restrictive and other legends or (ii) if after the required delivery date such holder purchases (in an open market transaction
or otherwise) shares of common stock to deliver in satisfaction of a sale by such holder of all or any portion of the number of
shares of common stock, or a sale of a number of shares of common stock equal to all or any portion of the number of shares of
common stock that such holder anticipated receiving from the Company without any restrictive legend, then, the Company shall either
(y) pay cash to the holder in an amount equal to the holder’s total purchase price (including brokerage commissions and
other out-of-pocket expenses, if any) for the shares of common stock so purchased, or the Buy-In Price, at which point the Company’s
obligation to deliver such shares shall terminate, or (z) promptly honor its obligation to deliver to the holder a certificate
or certificates representing such shares and pay cash to the holder in an amount equal to the excess (if any) of the Buy-In Price
over the product of (1) such number of shares of common stock that the Company was required to deliver multiplied by (2) the lowest
closing sale price of the common stock on any trading day during the period commencing on the date of the delivery by such holder
to the Company of the applicable shares (as the case may be) and ending on the date of such delivery and payment under this clause
(z).
Warrants issued
in Bridge Financing
Pursuant to the Note Purchase
Agreement and the underlying transactions, the Company has issued warrants to purchase shares of its common stock to jVen Capital
in an amount equal to 20% of the principal of each of the two bridge financing notes issued, or the jVen Capital Warrants, and
warrants to purchase shares of its common stock to MGHIF in an amount equal to 20% of the outstanding principal and accrued interest
under the amended and restated MGHIF Note on June 28, 2017, the date of issuance. The warrants each have a five year term from
issuance, are first exercisable on the date that is six months after the date of issuance and have an exercise price equal to
110% of the closing price of the Company’s common stock on the date immediately prior to the date of issuance. The terms
of the warrants issued in connection with the Bridge Financing (other than the exercise price and the number of shares) may be
amended, in the discretion of the holder, to reflect the terms of the warrants issued in the July 2017 Public Offering.
The jVen Capital Warrants
each include a blocker provision that prevents the exercise of the jVen Capital Warrants if such exercise, when aggregated with
the other issuances contemplated under the Note Purchase Agreement, would violate Nasdaq Listing Rule 5635, unless stockholder
approval is first obtained by the Company.
Warrants issued
in the July 2017 Public Offering
The Company issued warrants
in connection with the July 2017 Public Offering. The common warrants issued in the July 2017 Public Offering entitle the registered
holder to purchase one five-hundredths of a share of common stock at an exercise price of $212.50 per share. In addition, the
Company issued warrants to the placement agent that have an exercise price of $250.00 per share of common stock. All of the warrants
issued in the July 2017 Public Offering are immediately exercisable and have a five-year term from the date of issuance.
Warrants issued
in the February 2018 Public Offering
The Company issued warrants
in connection with the February 2018 Public Offering. The common warrants issued in the February 2018 Public Offering entitle
the registered holder to purchase one-fortieth of a share of common stock at an exercise price of $65.00 per share. In addition,
the Company issued warrants to the placement agent that have an exercise price of $81.25 per share of common stock. All of the
warrants issued in the February 2018 Public Offering are immediately exercisable and have a five-year term from the date of issuance.
Registration
Rights
Investors’
Rights Agreement
Under the Third Amended and
Restated Investors’ Rights Agreement, dated as of December 18, 2013, among the Company and certain investors, or the investors’
rights agreement, we granted registration rights to the holders of shares acquired prior to our initial public offering, or their
permitted transferees. These rights are provided under the terms of the investors’ rights agreement, and include demand
registration rights, short-form registration rights and piggyback registration rights. All fees, costs and expenses of underwritten
registrations will be borne by us and all selling expenses, including underwriting discounts and selling commissions, will be
borne by the holders of the shares being registered. As of the date of this prospectus, the holders of 11,619 shares of our common
stock have registration rights under the investors’ rights agreement. The investors’ rights agreement contains customary
cross-indemnification provisions, under which we are obligated to indemnify holders of registrable shares in the event of material
misstatements or omissions in the registration statement attributable to us, and they are obligated to indemnify us for material
misstatements or omissions attributable to them. The registration rights granted under the investors’ rights agreement will
terminate at the earlier of the closing of a deemed liquidation event and when all of the holders of registrable securities are
eligible to be sold without restrictions under Rule 144 promulgated under the Securities Act within any 90-day period.
AdvanDx
Merger and MGHIF Investment
In connection with the July
2015 merger transaction among the Company, a merger sub and AdvanDx, Inc., and the related transactions in which MGHIF purchased
shares of our common stock and initially issued the MGHIF Note, the Company also entered into a registration rights agreement
with the AdvanDx stockholders receiving merger consideration and with MGHIF, pursuant to which the investors were granted certain
demand registration rights and piggyback registration rights in connection with subsequent registered offerings of the Company’s
common stock. MGHIF also received rights to participate on a pro-rata basis in future securities offerings by the Company. MGHIF
is the only holder of registrable securities under this registration rights agreement.
Bridge
Financing Registration Rights
In connection with the bridge
financing the Company entered into a registration rights agreement with jVen Capital and with MGHIF, pursuant to which the investors
were granted certain demand registration rights and piggyback registration rights in connection with subsequent registered offerings
of the Company’s common stock. The registrable securities include the shares of common stock underlying the warrants issued
to jVen Capital and to MGHIF under the terms of the bridge financing promissory notes.
DESCRIPTION
OF SECURITIES WE ARE OFFERING
We are offering (i) up to 1,526,717 units, each unit consisting of one share of our common stock and one common warrant to purchase one share of our common stock, and (ii) up to 1,526,717 pre-funded units, each pre-funded unit consisting of one pre-funded warrant to purchase one share of our common stock and one common warrant to purchase one share of our common stock. For each pre-funded unit we sell, the number of units we are offering will be decreased on a one-for-one basis. The share of common stock and accompanying common warrant included in each unit will be issued separately and will be immediately separable upon issuance, and the pre-funded warrant to purchase one share of common stock and the accompanying common warrant included in each pre-funded unit will be issued separately and will be immediately separable upon issuance. The units and pre-funded units will not be issued or certificated. We are also registering the shares of common stock included in the units and the shares of common stock issuable from time to time upon exercise of the pre-funded warrants included in pre-funded units and common warrants included in the units and the pre-funded units offered hereby.
Common Stock
The material terms and provisions
of our common stock and each other class of our securities which qualifies or limits our common stock are described under the
caption “Description of Capital Stock” in this prospectus.
Pre-Funded Warrants
The following summary of certain terms and provisions
of the pre-funded warrants included in the pre-funded units that are being offered hereby is not complete and is subject to, and
qualified in its entirety by, the provisions of the pre-funded warrant, the form of which is filed as an exhibit to the registration
statement of which this prospectus forms a part. Prospective purchasers should carefully review the terms and provisions of the
form of pre-funded warrant for a complete description of the terms and conditions of the pre-funded warrants.
Duration
and Exercise Price
Each pre-funded warrant
offered hereby will have an initial exercise price per share equal to $0.01. The pre-funded warrants will be immediately exercisable
and may be exercised at any time until the pre-funded warrants are exercised in full. The exercise price and number of shares
of common stock issuable upon exercise is subject to appropriate adjustment in the event of stock dividends, stock splits, reorganizations
or similar events affecting our common stock and the exercise price. The pre-funded warrants will be issued separately from the
accompanying common warrants included in the pre-funded units, and may be transferred separately immediately thereafter.
Exercisability
The pre-funded
warrants will be exercisable, at the option of each holder, in whole or in part, by delivering to us a duly executed exercise
notice accompanied by payment in full for the number of shares of our common stock purchased upon such exercise (except in
the case of a cashless exercise as discussed below). A holder (together with its affiliates) may not exercise any portion of
the pre-funded warrant to the extent that the holder would own more than 4.99% of the outstanding common stock immediately
after exercise, except that upon at least 61 days’ prior notice from the holder to us, the holder may increase the
amount of ownership of outstanding stock after exercising the holder’s pre-funded warrants up to 9.99% of the number of
shares of our common stock outstanding immediately after giving effect to the exercise, as such percentage ownership is
determined in accordance with the terms of the pre-funded warrants. Purchasers of pre-funded units in this offering may also
elect prior to the issuance of the pre-funded warrants to have the initial exercise limitation set at 9.99% of our
outstanding common stock.
Cashless
Exercise
If, at the time a holder exercises
its pre-funded warrants, a registration statement registering the issuance of the shares of common stock underlying the pre-funded
warrants under the Securities Act is not then effective or available for the issuance of such shares, then in lieu of making the
cash payment otherwise contemplated to be made to us upon such exercise in payment of the aggregate exercise price, the holder
may elect instead to receive upon such exercise (either in whole or in part) the net number of shares of common stock determined
according to a formula set forth in the pre-funded warrants.
Transferability
Subject to applicable laws,
a pre-funded warrant may be transferred at the option of the holder upon surrender of the pre-funded warrant to us together with
the appropriate instruments of transfer.
Fractional
Shares
No fractional shares of common
stock will be issued upon the exercise of the pre-funded warrants. Rather, the number of shares of common stock to be issued will,
at our election, either be rounded up to the nearest whole number or we will pay a cash adjustment in respect of such final fraction
in an amount equal to such fraction multiplied by the exercise price.
Trading
Market
There is no trading market
available for the pre-funded warrants on any securities exchange or nationally recognized trading system.
Right
as a Stockholder
Except as otherwise provided
in the pre-funded warrants or by virtue of such holder’s ownership of shares of our common stock, the holders of the pre-funded
warrants do not have the rights or privileges of holders of our common stock, including any voting rights, until they exercise
their pre-funded warrants.
Common Warrants
The following summary of certain terms and provisions
of common warrants included in the units and the pre-funded units that are being offered hereby is not complete and is subject
to, and qualified in its entirety by, the provisions of the common warrants, the form of which is filed as an exhibit to the registration
statement of which this prospectus forms a part. Prospective purchasers should carefully review the terms and provisions of the
form of common warrant for a complete description of the terms and conditions of the common warrants.
Duration and Exercise Price
Each common warrant included in the units and the pre-funded
units offered hereby will have an initial exercise price per share equal to $ . The common warrants will be immediately exercisable
and will expire on the fifth anniversary of the original issuance date. The exercise price and number of shares of common stock
issuable upon exercise is subject to appropriate adjustment in the event of stock dividends, stock splits, reorganizations or
similar events affecting our common stock and the exercise price. The common warrants will be issued separately from the common
stock included in the units, or the pre-funded warrants included in the pre-funded units, as the case may be, and may be transferred
separately immediately thereafter. A common warrant to purchase one share of our common stock will be included in each unit or
pre-funded unit purchased in this offering.
Cashless Exercise
If, at the time a holder exercises its common warrants,
a registration statement registering the issuance of the shares of common stock underlying the common warrants under the Securities
Act is not then effective or available for the issuance of such shares, then in lieu of making the cash payment otherwise contemplated
to be made to us upon such exercise in payment of the aggregate exercise price, the holder may elect instead to receive upon such
exercise (either in whole or in part) the net number of shares of common stock determined according to a formula set forth in
the common warrants.
Exercisability
The common warrants will be exercisable, at the option
of each holder, in whole or in part, by delivering to us a duly executed exercise notice accompanied by payment in full for the
number of shares of our common stock purchased upon such exercise (except in the case of a cashless exercise as discussed below).
A holder (together with its affiliates) may not exercise any portion of the common warrant to the extent that the holder would
own more than 4.99% of the outstanding common stock immediately after exercise, except that upon at least 61 days' prior notice
from the holder to us, the holder may increase the amount of ownership of outstanding stock after exercising the holder's common
warrants up to 9.99% of the number of shares of our common stock outstanding immediately after giving effect to the exercise,
as such percentage ownership is determined in accordance with the terms of the common warrants.
Fractional Shares
No fractional shares of common stock will be issued upon
the exercise of the common warrants. Rather, the number of shares of common stock to be issued will, at our election, either be
rounded up to the nearest whole number or we will pay a cash adjustment in respect of such final fraction in an amount equal to
such fraction multiplied by the exercise price.
Transferability
Subject to applicable laws, a common warrant may be transferred
at the option of the holder upon surrender of the common warrant to us together with the appropriate instruments of transfer.
Trading Market
There is no trading market available for the common warrants
on any securities exchange or nationally recognized trading system.
Right as a Stockholder
Except as otherwise provided in the common warrants or
by virtue of such holder’s ownership of shares of our common stock, the holders of the common warrants do not have the rights
or privileges of holders of our common stock, including any voting rights, until they exercise their common warrants.
Fundamental Transaction
In the event of a fundamental transaction which is within
our control, other than the business combination pursuant to the Implementation Agreement, the holders of the common warrants
have the right to require us or a successor entity to redeem the common warrant for cash in the amount of the Black-Scholes value
of the unexercised portion of the common warrant on the date of the consummation of the fundamental transaction. In the event
of a fundamental transaction which not in our control, including a fundamental transaction that is not approved by our Board,
the holders of the common warrants have the right to require us or a successor entity to redeem the common warrant for the consideration
paid in the fundamental transaction in the amount of the Black Scholes value of the unexercised portion of the common warrant
on the date of the consummation of the fundamental transaction.
Anti-Takeover
Effects of Our Certificate of Incorporation, Bylaws and Delaware Law
Our certificate of incorporation
and bylaws include a number of provisions that may have the effect of delaying, deferring or preventing another party from acquiring
control of us and encouraging persons considering unsolicited tender offers or other unilateral takeover proposals to negotiate
with our Board of Directors rather than pursue non-negotiated takeover attempts. These provisions include the items described
below. The following descriptions are summaries of the material terms of our Amended and Restated Certificate of Incorporation,
as amended, and Amended and Restated Bylaws. We refer in this section to our Amended and Restated Certificate of Incorporation
as our certificate of incorporation, and we refer to our Amended and Restated Bylaws as our bylaws.
Meetings
of Stockholders
Our certificate of incorporation
provides that only the Chair of the Board, the Chief Executive Officer or a majority of the members of our Board of Directors then
in office may call special meetings of stockholders and only those matters set forth in the notice of the special meeting may
be considered or acted upon at a special meeting of stockholders. Our bylaws limit the business that may be conducted at an annual
meeting of stockholders to those matters properly brought before the meeting.
Advance
Notice Requirements
Our bylaws establish advance
notice procedures with regard to stockholder proposals relating to the nomination of candidates for election as directors or new
business to be brought before meetings of our stockholders. These procedures provide that notice of stockholder proposals must
be timely given in writing to our corporate secretary prior to the meeting at which the action is to be taken. Generally, to be
timely, notice must be received at our principal executive offices not less than 90 days nor more than 120 days prior to the first
anniversary date of the annual meeting for the preceding year. Our bylaws specify the requirements as to form and content of all
stockholders’ notices. These requirements may preclude stockholders from bringing matters before the stockholders at an
annual or special meeting.
Amendment
to Certificate of Incorporation and Bylaws
Any amendment of our certificate
of incorporation must first be approved by a majority of our Board of Directors, and if required by law or our certificate of
incorporation, must thereafter be approved by a majority of the outstanding shares entitled to vote on the amendment, except that
the amendment of the provisions relating to stockholder action, board composition, limitation of liability and the amendment of
our certificate of incorporation must be approved by not less than 66 2/3% of the outstanding shares entitled to vote on the amendment,
and not less than 66 2/3% of the outstanding shares of each class entitled to vote thereon as a class. Our bylaws may be amended
by the affirmative vote of a majority of the directors then in office, subject to any limitations set forth in the bylaws; and
may also be amended by the affirmative vote of at least 66 2/3% of the outstanding shares entitled to vote on the amendment, or,
if our Board of Directors recommends that the stockholders approve the amendment, by the affirmative vote of the majority of the
outstanding shares entitled to vote on the amendment, in each case voting together as a single class.
Undesignated
Preferred Stock
Our Board of Directors has
the authority, without further action by our stockholders, to issue from time to time 7,690,572 shares of preferred stock in one
or more series. The existence of authorized but unissued shares of preferred stock may enable our Board of Directors to discourage
an attempt to obtain control of us by means of a merger, tender offer, proxy contest or otherwise. For example, if in the due
exercise of its fiduciary obligations, our Board of Directors were to determine that a takeover proposal is not in the best interests
of our stockholders, our Board of Directors could cause shares of preferred stock to be issued without stockholder approval in
one or more private offerings or other transactions that might dilute the voting or other rights of the proposed acquirer or insurgent
stockholder or stockholder group. In this regard, our certificate of incorporation grants our Board of Directors broad power to
establish the rights and preferences of authorized and unissued shares of preferred stock. The issuance of shares of preferred
stock could decrease the amount of earnings and assets available for distribution to holders of shares of common stock. The issuance
may also adversely affect the rights and powers, including voting rights, of these holders and may have the effect of delaying,
deterring or preventing a change in control of us.
Exclusive
Jurisdiction for Certain Actions
Our certificate of incorporation
provides that, once our common stock is a “covered security,” unless we consent in writing to the selection of an
alternative forum, the Court of Chancery of the State of Delaware shall be the sole and exclusive forum for (i) any derivative
action or proceeding brought on our behalf, (ii) any action asserting a claim of breach of a fiduciary duty owed by any of our
directors, officers or other employees to us or our stockholders, (iii) any action asserting a claim arising pursuant to any provision
of the Delaware General Corporation Law, our certificate of incorporation or our bylaws, or (iv) any action asserting a claim
against us governed by the internal affairs doctrine. Although we believe this provision benefits us by providing increased consistency
in the application of Delaware law in the types of lawsuits to which it applies, the provision may have the effect of discouraging
lawsuits against our directors and officers. The enforceability of similar exclusive forum provisions in other companies’
certificates of incorporation has been challenged in legal proceedings, and it is possible that a court could rule that this provision
in our certificate of incorporation is inapplicable or unenforceable.
Section
203 of the Delaware General Corporation Law
We are subject to the provisions
of Section 203 of the Delaware General Corporation Law. In general, Section 203 prohibits a publicly held Delaware corporation
from engaging in a “business combination” with an “interested stockholder” for a three-year period following
the time that this stockholder becomes an interested stockholder, unless the business combination is approved in a prescribed
manner. Under Section 203, a business combination between a corporation and an interested stockholder is prohibited unless it
satisfies one of the following conditions:
|
|
·
|
before
the stockholder became interested, our Board of Directors approved either the business
combination or the transaction which resulted in the stockholder becoming an interested
stockholder;
|
|
|
·
|
upon
consummation of the transaction which resulted in the stockholder becoming an interested
stockholder, the interested stockholder owned at least 85% of the voting stock of the
corporation outstanding at the time the transaction commenced, excluding for purposes
of determining the voting stock outstanding, shares owned by persons who are directors
and also officers, and employee stock plans, in some instances, but not the outstanding
voting stock owned by the interested stockholder; or
|
|
|
·
|
at
or after the time the stockholder became interested, the business combination was approved
by our Board of Directors and authorized at an annual or special meeting of the stockholders
by the affirmative vote of at least two-thirds of the outstanding voting stock which
is not owned by the interested stockholder.
|
Section 203 defines a business
combination to include:
|
|
·
|
any
merger or consolidation involving the corporation and the interested stockholder;
|
|
|
·
|
any
sale, transfer, lease, pledge or other disposition involving the interested stockholder
of 10% or more of the assets of the corporation;
|
|
|
·
|
subject
to exceptions, any transaction that results in the issuance or transfer by the corporation
of any stock of the corporation to the interested stockholder;
|
|
|
·
|
subject
to exceptions, any transaction involving the corporation that has the effect of increasing
the proportionate share of the stock of any class or series of the corporation beneficially
owned by the interested stockholder; and
|
|
|
·
|
the
receipt by the interested stockholder of the benefit of any loans, advances, guarantees,
pledges or other financial benefits provided by or through the corporation.
|
In general, Section 203 defines
an interested stockholder as any entity or person beneficially owning 15% or more of the outstanding voting stock of the corporation
and any entity or person affiliated with or controlling or controlled by the entity or person.
Listing
Our common stock is listed
on the Nasdaq Capital Market under the symbol “OPGN” and our IPO Warrants are listed on the Nasdaq Capital Market
under the symbol “OPGNW.”
On August 19, 2019, the Company received
a written notification from Nasdaq notifying the Company that it has failed to comply with Nasdaq Marketplace Rule 5550(b)(1)
because the Company’s stockholders’ equity as of June 30, 2019 fell below the required minimum of $2,500,000 and as
of June 30, 2019, the Company did not meet the alternative compliance standards of market value of listed securities or net income
from continuing operations for continued listing. In accordance with Nasdaq’s listing requirements, the Company had 45 calendar
days to submit a plan to regain compliance. If the plan is accepted, Nasdaq can grant the Company an extension of up to 180 calendar
days from the date it received the notification to evidence compliance. The Company submitted a plan to Nasdaq to regain compliance
with the Nasdaq minimum stockholders’ equity standard on October 3, 2019, which plan included information regarding this
offering. However, there can be no assurance that the Company’s plan will be accepted or that if it is, the Company will
be able to regain compliance.
Transfer
Agent and Registrar
The transfer agent and registrar
for our common stock is Philadelphia Stock Transfer, Inc. The transfer agent’s address is 2320 Haverford Rd., Suite 230,
Ardmore, PA 19003.
UNDERWRITING
We have entered into an
underwriting agreement dated , 2019 with H.C. Wainwright & Co., LLC, as underwriter, with respect to the securities being
offered hereby. Subject to the terms and conditions of the underwriting agreement, we have agreed to sell to the underwriter and
the underwriter has agreed to purchase from us, at the public offering price less the underwriting discounts and commissions set
forth on the cover page of this prospectus, units and pre-funded units.
A copy of the form of underwriting
agreement has been filed as an exhibit to the registration statement of which this prospectus is a part. The securities we are
offering are being offered by the underwriter subject to certain conditions specified in the underwriting agreement.
We have been advised by the underwriter that it proposes
to offer the units and pre-funded units, as the case may be, directly to the public at the public offering prices set forth on
the cover page of this prospectus. Any units and pre-funded units sold by the underwriter to securities dealers will be sold at
the public offering price less a selling concession not in excess of $ per unit or pre-funded unit.
The underwriting agreement provides that the underwriter’s
obligation to purchase the securities we are offering is subject to conditions contained in the underwriting agreement. The underwriter
is obligated to purchase and pay for all of the securities offered by this prospectus if any of these securities are purchased,
other than those shares of common stock and/or warrants covered by the option to purchase additional securities described below.
No action has been taken by us or the underwriter that
would permit a public offering of the units or pre-funded units in any jurisdiction where action for that purpose is required.
None of the securities included in this offering may be offered or sold, directly or indirectly, nor may this prospectus or any
other offering material or advertisements in connection with the offer and sales of any of the securities be distributed or published
in any jurisdiction, except under circumstances that will result in compliance with the applicable rules and regulations of that
jurisdiction. Persons who receive this prospectus are advised to inform themselves about and to observe any restrictions relating
to this offering of units and pre-funded units and the distribution of this prospectus. This prospectus is neither an offer to
sell nor a solicitation of any offer to buy the units or pre-funded units in any jurisdiction where that would not be permitted
or legal.
Underwriting Discounts,
Commissions and Expenses
The following table shows
the public offering price, underwriting discounts and commissions and proceeds, before expenses to us. These amounts are shown
assuming both no exercise and full exercise of the underwriter’s option to purchase additional shares of common stock and/or
common warrants to purchase shares of common stock.
|
|
|
Per
Unit
|
|
Per
Pre-funded Unit
|
|
Total
Without Option
|
|
Total
With Option
|
|
Public offering price
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
Underwriting discounts and commissions
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
Proceeds before expenses
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
We estimate the total expenses payable by us for this
offering, excluding the underwriting discounts and commissions, to be approximately $ , which includes (i) $50,000 non-accountable
expense allowance payable to the underwriter, (ii) reimbursement of the accountable expenses of the underwriter equal to $100,000,
including the legal fees of the underwriter and other out-of-pocket expenses being paid by us and (iii) other estimated expenses
of approximately $256,000, which include legal, accounting, printing costs and various fees associated with the registration and
listing of our securities sold in this offering.
In addition, we have agreed to issue to the underwriter
warrants to purchase up to 76,335 shares of common stock, or 87,786 shares of common stock if the underwriter exercises its option
to purchase additional securities in full, which represents 5% of the aggregate number of shares of common stock shares of common
stock (i) included within the units and (ii) issuable upon the exercise of the pre-funded warrants included within the pre-funded
units sold in this offering, at an exercise price of $ per share (representing 130% of the public offering price per unit or a
pre-funded unit to be sold in this offering). The underwriter warrants will be exercisable immediately and for five years from
the effective date of the registration statement. Pursuant to FINRA Rule 5110(g), the underwriter warrants and any shares issued
upon exercise of the underwriter warrants shall not be sold, transferred, assigned, pledged, or hypothecated, or be the subject
of any hedging, short sale, derivative, put or call transaction that would result in the effective economic disposition of the
securities by any person for a period of 180 days immediately following the date of effectiveness or commencement of sales of
this offering, except the transfer of any security: (i) by operation of law or by reason of our reorganization; (ii) to any FINRA
member firm participating in the offering and the officers or partners thereof, if all securities so transferred remain subject
to the lock-up restriction set forth above for the remainder of the time period; (iii) if the aggregate amount of our securities
held by the underwriter or related persons do not exceed 1% of the securities being offered; (iv) that is beneficially owned on
a pro-rata basis by all equity owners of an investment fund, provided that no participating member manages or otherwise directs
investments by the fund and the participating members in the aggregate do not own more than 10% of the equity in the fund; or
(v) the exercise or conversion of any security, if all securities remain subject to the lock-up restriction set forth above for
the remainder of the time period.
Option to Purchase Additional
Securities
We have granted to the underwriter an option, exercisable
not later than 30 days after the date of this prospectus, to purchase up to an additional 229,007 shares of common stock and/or
up to 229,007 additional common warrants to purchase one share of common stock less the underwriting discounts and commissions,
set forth on the cover page of this prospectus. If any additional shares of common stock and/or common warrants are purchased
pursuant to such option, the underwriter will offer these securities on the same terms as those on which the securities are being
offered hereby.
Tail Financing Payments
We have also agreed to pay
the underwriter a tail fee equal to the cash and warrant compensation in this offering, if any investor, who was contacted or
introduced to us by the underwriter during the term of the underwriter’s engagement, provides us with capital in any public
or private offering or other financing or capital raising transaction, subject to certain exceptions, during the six-month period
following expiration or termination of our engagement of the underwriter.
Lock-up Agreements
Our officers and directors
have agreed with the underwriter to be subject to a lock-up period of 90 days following the date of this prospectus. This means
that, during the applicable lock-up period, such persons may not offer for sale, contract to sell, sell, distribute, grant any
option, right or warrant to purchase, pledge, hypothecate or otherwise dispose of, directly or indirectly, any shares of our common
stock or any securities convertible into, or exercisable or exchangeable for, shares of common stock. Certain limited transfers
are permitted during the lock-up period if the transferee agrees to these lock-up restrictions. We have also agreed, in the underwriting
agreement, to similar lock-up restrictions on the issuance and sale of our common stock, or any securities convertible into, or
exercisable or exchangeable for, shares of common stock, for 90 days following the closing of this offering, subject to certain
exemptions. The underwriter may, in its sole discretion and without notice, waive the terms of any of these lock-up agreements.
Stabilization, Short
Positions and Penalty Bids
The underwriter may engage
in syndicate covering transactions, stabilizing transactions and penalty bids or purchases for the purpose of pegging, fixing
or maintaining the price of our common stock:
|
|
●
|
Syndicate covering transactions involve purchases
of securities in the open market after the distribution has been completed in order to cover syndicate short positions.
Such a naked short position would be closed out by buying securities in the open market. A naked short position is more
likely to be created if the underwriter is concerned that there could be downward pressure on the price of the securities
in the open market after pricing that could adversely affect purchasers who purchase in the offering.
|
|
|
|
|
|
|
●
|
Stabilizing transactions permit bids to purchase the underlying security so long as the
stabilizing bids do not exceed a specific maximum.
|
|
|
|
|
|
|
●
|
Penalty bids permit the underwriter to reclaim a selling concession from a syndicate member
when the securities originally sold by the syndicate member are purchased in a stabilizing or syndicate covering transaction
to cover syndicate short positions.
|
These syndicate covering transactions,
stabilizing transactions and penalty bids may have the effect of raising or maintaining the market prices of our securities or
preventing or retarding a decline in the market prices of our securities. As a result, the price of our common stock may be higher
than the price that might otherwise exist in the open market. Neither we nor the underwriter make any representation or prediction
as to the effect that the transactions described above may have on the price of our common stock. These transactions may be effected
on the Nasdaq Capital Market, in the over-the-counter market or on any other trading market and, if commenced, may be discontinued
at any time.
In connection with this offering,
the underwriter also may engage in passive market making transactions in our common stock in accordance with Regulation M during
a period before the commencement of offers or sales of our securities in this offering and extending through the completion of
the distribution. In general, a passive market maker must display its bid at a price not in excess of the highest independent
bid for that security. However, if all independent bids are lowered below the passive market maker’s bid that bid must then
be lowered when specific purchase limits are exceeded. Passive market making may stabilize the market price of the securities
at a level above that which might otherwise prevail in the open market and, if commenced, may be discontinued at any time.
Neither we nor the underwriter
make any representation or prediction as to the direction or magnitude of any effect that the transactions described above may
have on the prices of our securities. In addition, neither we nor the underwriter make any representation that the underwriter
will engage in these transactions or that any transactions, once commenced, will not be discontinued without notice.
Indemnification
We have agreed to indemnify
the underwriter against certain liabilities, including certain liabilities arising under the Securities Act of 1933, as amended,
or to contribute to payments that the underwriter may be required to make for these liabilities.
Determination of Offering
Price
The actual offering price
of the securities we are offering will be negotiated between us and the underwriter based on the trading of our common stock prior
to the offering, among other things, and may be at a discount to the current market price.
Electronic Offer, Sale
and Distribution of Securities
A prospectus in electronic
format may be made available on the websites maintained by the underwriter, if any, participating in this offering and the underwriter
may distribute prospectuses electronically. Other than the prospectus in electronic format, the information on these websites
is not part of this prospectus or the registration statement of which this prospectus form a part, has not been approved or endorsed
by us or the underwriter, and should not be relied upon by purchasers.
Other Relationships
The underwriter and its respective
affiliates have provided, and may in the future engage in, investment banking and other commercial dealings in the ordinary course
of business with us or our affiliates for which they have received and may continue to receive customary fees and commissions.
The underwriter of this offering also acted as our placement agent in connection with the registered public offerings we consummated
in July 2017 and February 2018.
Listing
Our common stock is listed on the Nasdaq
Capital Market under the symbol “OPGN.” We do not intend to apply for listing of the common warrants or pre-funded
warrants on any securities exchange or other nationally recognized trading system.
MATERIAL
U.S. FEDERAL INCOME TAX CONSEQUENCES
The following is a summary of the material U.S.
federal income tax consequences of the acquisition, ownership and disposition of our common stock, and pre-funded warrants and the acquisition, ownership, exercise, expiration or disposition of common
warrants acquired in this offering, but does not purport to be
a complete analysis of all the potential tax considerations relating thereto. Throughout this summary, all references to our
common stock are meant to include the common stock issuable upon exercise of the pre-funded warrants in the pre-funded units.
This summary is based upon the provisions of the Internal Revenue Code of 1986, as amended, or the Code, Treasury Regulations
promulgated thereunder, administrative rulings and judicial decisions, all as of the date hereof. These authorities may be
changed or subject to differing interpretations, possibly with retroactive effect, with the resulting U.S. federal income tax
consequences being different from those set forth below. We have not sought and will not seek any ruling from the Internal
Revenue Service, or the IRS, with respect to the statements made and the conclusions reached in the following summary, and
there can be no assurance that the IRS or a court will agree with such statements and conclusions.
This summary also does not
address the tax considerations arising under the laws of any U.S. state or local or any non-U.S. jurisdiction, estate or gift
tax, the 3.8% Medicare tax on net investment income or any alternative minimum tax consequences. In addition, this discussion
does not address tax considerations applicable to a holder’s particular circumstances or to a holder that may be subject
to special tax rules, including, without limitation:
|
|
·
|
banks,
insurance companies or other financial institutions;
|
|
|
·
|
tax-exempt
or government organizations;
|
|
|
·
|
brokers
or dealers in securities or currencies;
|
|
|
·
|
traders
in securities that elect to use a mark-to-market method of accounting for their securities
holdings;
|
|
|
·
|
persons
that own, or are deemed to own, more than 5.0% of our capital stock;
|
|
|
·
|
certain
U.S. expatriates, citizens or former long-term residents of the United States;
|
|
|
·
|
persons
who hold our common stock as a position in a hedging transaction, “straddle,”
“conversion transaction,” synthetic security, other integrated investment,
or other risk reduction transaction;
|
|
|
·
|
persons
who do not hold our common stock as a capital asset within the meaning of Section 1221
of the Code (generally, for investment purposes);
|
|
|
·
|
persons
deemed to sell our common stock under the constructive sale provisions of the Code;
|
|
|
·
|
partnerships,
or other entities or arrangements treated as partnerships for U.S. federal income tax
purposes, or investors in any such entities;
|
|
|
·
|
persons
for whom our stock constitutes “qualified small business stock” within the
meaning of Section 1202 of the Code;
|
|
|
·
|
integral
parts or controlled entities of foreign sovereigns;
|
|
|
·
|
passive
foreign investment companies and corporations that accumulate earnings to avoid U.S.
federal income tax; or
|
|
|
·
|
persons
that acquire our common stock as compensation for services.
|
In addition, if a partnership,
including any entity or arrangement classified as a partnership for U.S. federal income tax purposes, holds our common stock,
the tax treatment of a partner generally will depend on the status of the partner, the activities of the partnership, and certain
determinations made at the partner level. Accordingly, partnerships that hold our common stock, and partners in such partnerships,
should consult their tax advisors regarding the U.S. federal income tax consequences to them of the purchase, ownership, and disposition
of our common stock.
You are urged to consult your
tax advisor with respect to the application of the U.S. federal income tax laws to your particular situation, as well as any tax
consequences of the purchase, ownership and disposition of our common stock arising under the U.S. federal estate or gift tax
rules or under the laws of any U.S. state or local or any non-U.S. or other taxing jurisdiction or under any applicable tax treaty.
Allocation of Purchase Price and Characterization
of a Unit
Each unit and pre-funded unit should be treated for U.S.
federal income tax purposes as an investment unit consisting of one share of common stock and one warrant. For U.S. federal income
tax purposes, each holder must allocate the purchase price of the unit or pre-funded unit between that share of common stock or
pre-funded warrant, as applicable, and the warrant based on the relative fair market value of each at the time of issuance. The
price allocated to each share of common stock, pre-funded warrant and common warrant generally will be the holder’s tax
basis in such share or warrant, as the case may be.
Treatment of Pre-Funded Warrant
Although it is not entirely free from doubt, we believe a pre-funded
warrant should be treated as common stock for U.S. federal income tax purposes and a holder of pre-funded warrants should generally
be taxed in the same manner as a holder of our common stock, as described below. Accordingly, no gain or loss should be recognized
upon the exercise of a pre-funded warrant and, upon exercise, the holding period of a pre-funded warrant should carry over to
the common stock received. Similarly, the tax basis of the pre-funded warrant should carry over to the common stock received upon
exercise, increased by the exercise price of $0.01 per share. However, our characterization of a pre-funded warrant is
not binding on the IRS, and the IRS may treat our pre-funded warrants as warrants to acquire our common stock. If so, the amount
and character of your gain with respect to an investment in our pre-funded warrants could change. Accordingly, each holder should
consult his, her or its own tax advisor regarding the risks associated with the acquisition of pre-funded warranst pursuant to
this offering (including potential alternative characterizations). The balance of this discussion generally assumes that our characterization
described above is respected for U.S. federal income tax purposes.
Definition of a U.S.
Holder
For purposes of this summary, a “U.S. Holder”
is any beneficial owner of our common stock, pre-funded warrants or common warrants that is a “U.S. person,” and is
not a partnership, or an entity treated as a partnership or disregarded from its owner, each for U.S. federal income tax purposes.
A U.S. person is any person that, for U.S. federal income tax purposes, is or is treated as any of the following:
|
|
·
|
an
individual who is a citizen or resident of the United States;
|
|
|
·
|
a
corporation created or organized under the laws of the United States, any state thereof,
or the District of Columbia;
|
|
|
·
|
an
estate, the income of which is subject to U.S. federal income tax regardless of its source;
or
|
|
|
·
|
a
trust that (1) is subject to the primary supervision of a U.S. court and the control
of one or more U.S. persons (within the meaning of Section 7701(a)(30) of the Code),
or (2) has a valid election in effect to be treated as a U.S. person for U.S. federal
income tax purposes.
|
For purposes of this summary, a “Non-U.S. Holder”
is any beneficial owner of our common stock, pre-funded warrants or common warrants that is not a U.S. Holder or a partnership,
or other entity treated as a partnership or disregarded from its owner, each for U.S. federal income tax purposes.
Tax Consequences to U.S.
Holders
Exercise and Expiration of Warrants
In
general, a U.S. Holder will not recognize gain or loss for U.S. federal income tax purposes upon exercise of a warrant, except
to the extent the U.S. Holder receives a cash payment for a such fractional share that would otherwise have been issuable upon
exercise of the warrant, which will be treated as a sale subject to the rules described under “Sale or Other Disposition
of Our Common Stock, Pre-Funded Warrants or Common Warrants” below. The U.S. Holder will take a tax basis in the shares
acquired on the exercise of a warrant equal to the exercise price of the warrant. The U.S. Holder’s holding period in the
shares of our common stock acquired on exercise of the warrant will begin on the date of exercise of the warrant, and will not
include any period for which the U.S. Holder held the warrant. The lapse or expiration of a warrant will be treated as if the
U.S. Holder sold or exchanged the warrant and recognized a capital loss equal to the U.S. Holder’s tax basis in the warrant.
The deductibility of capital losses is subject to limitations.
Certain Adjustments to the Warrants
Under
Section 305 of the Code, an adjustment to the number of shares of common stock issued on the exercise of the warrants, or an adjustment
to the exercise price of the warrants, may be treated as a constructive distribution to a U.S. Holder of the warrants if, and
to the extent that, such adjustment has the effect of increasing such U.S. Holder’s proportionate interest in our “earnings
and profits” or assets, depending on the circumstances of such adjustment (for example, if such adjustment is to compensate
for a distribution of cash or other property to our stockholders). Adjustments to the exercise price of warrants made pursuant
to a bona fide reasonable adjustment formula that has the effect of preventing dilution of the interest of the holders of the
warrants generally should not be considered to result in a constructive distribution. Such constructive distribution would be
treated as a dividend, return of capital or capital gain as described under the heading “Distributions” below. Any
such constructive distribution would be taxable whether or not there is an actual distribution of cash or other property.
On
April 12, 2016, the IRS issued proposed regulations addressing the amount and timing of deemed distributions, obligations of withholding
agents and filing and notice obligations of issuers. If adopted as proposed, the regulations would generally provide that (i)
the amount of a deemed distribution is the excess of the fair market value of a warrant immediately after the number-of-shares
or exercise-price adjustment over the fair market value of the warrant without the adjustment, (ii) the deemed distribution occurs
at the earlier of the date the adjustment occurs under the terms of the warrant and the date of the actual distribution of cash
or property that results in the deemed distribution, and (iii) we are required to report the amount of any deemed distributions
on our website or to the IRS and all holders of warrants (including holders of warrants that would otherwise be exempt from reporting).
The final regulations will be effective for deemed distributions occurring on or after the date of adoption, but holders of warrants
agents may rely on them prior to that date under certain circumstances.
Distributions
As discussed above under “Dividend Policy,”
we do not currently expect to make distributions on our common stock. In the event that we do make distributions of cash or other
property, distributions paid on common stock or pre-funded warrants, other than certain pro rata distributions of common stock
or pre-funded warrants, will be treated as a dividend to the extent paid out of our current or accumulated earnings and profits,
if any, and will be includible in income by the U.S. Holder and taxable as ordinary income when received. If a distribution exceeds
our current and accumulated earnings and profits, the excess will be first treated as a tax-free return of the U.S. Holder’s
investment, up to the U.S. Holder’s tax basis in the common stock or pre-funded warrant, as applicable. Any remaining excess
will be treated as a capital gain. Subject to applicable limitations, dividends paid to certain non-corporate U.S. Holders may
be eligible for taxation as “qualified dividend income” and therefore may be taxable at rates applicable to long-term
capital gains. U.S. Holders should consult their tax advisers regarding the availability of the reduced tax rate on dividends
in their particular circumstances. Dividends received by a corporate U.S. Holder will be eligible for the dividends-received deduction
if the U.S. Holder meets certain holding period and other applicable requirements.
Sale or Other Disposition of Our Common Stock,
Pre-Funded Warrants or Common Warrants
For U.S. federal income tax purposes, gain or loss realized
on the sale or other disposition of common stock, pre-funded warrants or common warrants will be capital gain or loss, and will
be long-term capital gain or loss if the U.S. Holder held the common stock, pre-funded warrants or common warrants, as applicable,
for more than one year. The amount of the gain or loss will equal the difference between the U.S. Holder’s tax basis in
the common stock, pre-funded warrants, or common warrants disposed of and the amount realized on the disposition. Long-term capital
gains recognized by non-corporate U.S. Holders will be subject to reduced tax rates. The deductibility of capital losses is subject
to limitations.
Tax Consequences to Non-U.S. Holders
Exercise and Expiration of Warrants
In general, a Non-U.S.
Holder will not recognize gain or loss for U.S. federal income tax purposes upon exercise of a warrant, except to the extent the
Non-U.S. Holder receives a cash payment for a fractional share that would otherwise have been issuable upon exercise of the warrant,
which will be treated as a sale subject to the rules described under “Gain on Sale or Other Disposition of Our Common Stock,
Pre-Funded Warrants or Common Warrants” below. The Non-U.S. Holder will take a tax basis in the shares acquired on the exercise
of a warrant equal to the exercise price of the warrant. The Non-U.S. Holder’s holding period in the shares of our common
stock acquired on exercise of the warrant will begin on the date of exercise of the warrant, and will not include any period for
which the Non-U.S. Holder held the warrant.
The expiration of a
warrant will be treated as if the Non-U.S. Holder sold or exchanged the warrant and recognized a capital loss equal to the Non-U.S.
Holder’s tax basis in the warrant. However, a Non-U.S. Holder will not be able to utilize a loss recognized upon expiration
of a warrant against the Non-U.S. Holder’s U.S. federal income tax liability unless the loss is effectively connected with
the Non-U.S. Holder’s conduct of a trade or business within the United States (and, if an income tax treaty applies, is
attributable to a permanent establishment in the United States) or is treated as a U.S.-source loss and the Non-U.S. Holder is
present 183 days or more in the taxable year of disposition and certain other conditions are met.
Certain Adjustments to the Warrants
Under Section 305 of
the Code, an adjustment to the number of shares of common stock issued on the exercise of the warrants, or an adjustment to the
exercise price of the warrants, may be treated as a constructive distribution to a Non-U.S. Holder of the warrants if, and to
the extent that, such adjustment has the effect of increasing such Non-U.S. Holder’s proportionate interest in our “earnings
and profits” or assets, depending on the circumstances of such adjustment (for example, if such adjustment is to compensate
for a distribution of cash or other property to our shareholders). Adjustments to the exercise price of warrants made pursuant
to a bona fide reasonable adjustment formula that has the effect of preventing dilution of the interest of the holders of the
warrants generally should not be considered to result in a constructive distribution. Such constructive distribution would be
treated as a dividend, return of capital or capital gain as described under the heading “Distributions” below. Any
such constructive distribution would be taxable whether or not there is an actual distribution of cash or other property.
On April 12, 2016,
the IRS issued proposed regulations addressing the amount and timing of deemed distributions, obligations of withholding agents
and filing and notice obligations of issuers. If adopted as proposed, the regulations would generally provide that (i) the amount
of a deemed distribution is the excess of the fair market value of a warrant immediately after the number-of-shares or exercise-price
adjustment over the fair market value of the warrant without the adjustment, (ii) the deemed distribution occurs at the earlier
of the date the adjustment occurs under the terms of the warrant and the date of the actual distribution of cash or property that
results in the deemed distribution, (iii) subject to certain limited exceptions, a withholding agent is required to impose any
applicable withholding on deemed distributions to a Non-U.S. Holder and, if there is no associated cash payment, may set off its
withholding obligations against other payments to or funds of such holder and (iv) we are required to report the amount of any
deemed distributions on our website or to the IRS and all holders of warrants (including holders of warrants that would otherwise
be exempt from reporting). The final regulations will be effective for deemed distributions occurring on or after the date of
adoption, but holders of warrants and withholding agents may rely on them prior to that date under certain circumstances.
Distributions
As discussed in the section entitled “Dividend
Policy,” we do not anticipate paying any dividends on our common stock in the foreseeable future. If we make distributions
on our common stock or pre-funded warrants, those payments will constitute dividends for U.S. federal income tax purposes to the
extent we have current or accumulated earnings and profits, as determined under U.S. federal income tax principles. To the extent
those distributions exceed both our current and our accumulated earnings and profits, they will constitute a return of capital
and will first reduce a Non-U.S. Holder’s basis in our common stock or pre-funded warrants, as applicable, but not below
zero. Any excess will be treated as capital gain and will be treated as described below under the “—Gain on Sale or
Other Disposition of Common Stock, Pre-Funded Warrnts or Common Warrants” section. Any such distributions would be subject
to the discussions below regarding back-up withholding and the Foreign Account Tax Compliance Act, or FATCA.
Subject to the discussion below on effectively connected
income, any dividend paid to a Non-U.S. Holder generally will be subject to U.S. withholding tax either at a rate of 30% of the
gross amount of the dividend or such lower rate as may be specified by an applicable income tax treaty. To receive a reduced treaty
rate, a Non-U.S. Holder must provide us or our agent with an IRS Form W-8BEN (generally including a U.S. taxpayer identification
number), IRS Form W-8 BEN-E or another appropriate version of IRS Form W-8 (or a successor form), which must be updated periodically,
and which, in each case, must certify qualification for the reduced treaty rate. Non-U.S. Holders should consult their tax advisors
regarding their entitlement to benefits under any applicable income tax treaty.
Dividends paid to a Non-U.S. Holder that are effectively
connected with the Non-U.S. Holder’s conduct of a trade or business within the United States and that are not eligible for
relief from U.S. (net basis) income tax under an applicable income tax treaty, generally are exempt from the (gross basis) withholding
tax described above. To obtain this exemption from withholding tax, the Non-U.S. Holder must provide the applicable withholding
agent with an IRS Form W-8ECI or successor form or other applicable IRS Form W-8 certifying that the dividends are effectively
connected with the Non-U.S. Holder’s conduct of a trade or business within the United States. Such effectively connected
dividends, if not eligible for relief under a tax treaty, would not be subject to a withholding tax, but would be taxed at the
same graduated rates applicable to U.S. persons, net of certain deductions and credits and if, in addition, the Non-U.S. Holder
is a corporation, may also be subject to a branch profits tax at a rate of 30% (or such lower rate as may be specified by an applicable
income tax treaty).
If you are eligible for a reduced rate of withholding tax
pursuant to a tax treaty, you may be able to obtain a refund of any excess amounts withheld if you timely file an appropriate claim
for refund with the IRS.
Gain on Sale or Other Disposition of Our Common
Stock, Pre-Funded Warrants or Common Warrants
Subject to the discussion below regarding backup withholding
and FATCA, a Non-U.S. Holder generally will not be required to pay U.S. federal income tax on any gain realized upon the sale
or other disposition of our common stock, pre-funded warrants or common warrants unless:
|
|
·
|
the
gain is effectively connected with the Non-U.S. Holder’s conduct of a trade or
business within the United States and not eligible for relief under an applicable income
tax treaty, in which case the Non-U.S. Holder will be required to pay tax on the net
gain derived from the sale under regular graduated U.S. federal income tax rates, and
for a Non-U.S. Holder that is a corporation, such Non-U.S. Holder may be subject to the
branch profits tax at a 30% rate (or such lower rate as may be specified by an applicable
income tax treaty) on such effectively connected gain, as adjusted for certain items;
|
|
|
·
|
the
Non-U.S. Holder is an individual who is present in the United States for a period or
periods aggregating 183 days or more during the calendar year in which the sale or disposition
occurs and certain other conditions are met, in which case the Non-U.S. Holder will be
required to pay a flat 30% tax on the gain derived from the sale, which tax may be offset
by U.S. source capital losses (even though the Non-U.S. Holder is not considered a resident
of the United States) (subject to applicable income tax or other treaties); or
|
|
|
·
|
we
are a “U.S. real property holding corporation” for U.S. federal income
tax purposes, or a USRPHC, at any time within the shorter of the five-year period preceding
the disposition or the Non-U.S. Holder’s holding period for our common stock, pre-funded
warrants or common warrants, as applicable. We believe we are not currently and do not
anticipate becoming a USRPHC. However, because the determination of whether we are a
USRPHC depends on the fair market value of our United States real property interests
relative to the fair market value of our other business assets, there can be no assurance
that we will not become a USRPHC in the future. Even if we become a USRPHC, however,
gain arising from the sale or other taxable disposition by a Non-U.S. Holder of our common
stock, pre-funded warrants, or common warrants will not be subject to United States federal
income tax if (a) shares of our common stock are “regularly traded,” as defined
by applicable Treasury Regulations, on an established securities market, such as Nasdaq,
and (b) the Non-U.S. Holder owns or owned, actually and constructively, 5% or less of
the shares of our common stock and pre-funded warrants throughout the five-year period
ending on the date of the sale or exchange. If the foregoing exception does not apply,
such Non-U.S. Holder’s proceeds received on the disposition of shares will generally
be subject to withholding at a rate of 15% and such Non-U.S. Holder will generally be
taxed on any gain in the same manner as gain that is effectively connected with the conduct
of a U.S. trade or business, except that the branch profits tax generally will not apply.
|
Information Reporting
and Backup Withholding
Information returns may be filed with the IRS in connection
with distributions on common stock or pre-funded warrants, and the proceeds of a sale or other disposition of common stock or
pre-funded warrants. A non-exempt U.S. Holder may be subject to U.S. backup withholding on these payments if it fails to provide
its taxpayer identification number to the withholding agent and comply with certification procedures or otherwise establish an
exemption from backup withholding.
A Non-U.S. Holder may be
subject to U.S. information reporting and backup withholding on these payments unless the Non-U.S. Holder complies with certification
procedures to establish that it is not a U.S. person (within the meaning of the Code). The certification requirements generally
will be satisfied if the Non-U.S. Holder provides the applicable withholding agent with a statement on the applicable IRS Form
(or a suitable substitute or successor form), together with all appropriate attachments, signed under penalties of perjury, stating,
among other things, that such Non-U.S. Holder is not a U.S. Person. Applicable Treasury Regulations provide alternative methods
for satisfying this requirement. In addition, the amount of distributions on common stock or pre-funded warrants paid to a Non-U.S.
Holder, and the amount of any U.S. federal tax withheld therefrom, must be reported annually to the IRS and the holder. This information
may be made available by the IRS under the provisions of an applicable tax treaty or agreement to the tax authorities of the country
in which the Non-U.S. Holder resides.
Payment of the proceeds of the sale or other disposition
of common stock, pre-funded warrants or common warrants to or through a non-U.S. office of a U.S. broker or of a non-U.S. broker
with certain specified U.S. connections generally will be subject to information reporting requirements, but not backup withholding,
unless the Non-U.S. Holder certifies under penalties of perjury that it is not a U.S. person or an exemption otherwise applies.
Payments of the proceeds of a sale or other disposition of common stock, pre-funded warrants or common warrants to or through
a U.S. office of a broker generally will be subject to information reporting and backup withholding, unless the Non-U.S. Holder
certifies under penalties of perjury that it is not a U.S. person or otherwise establishes an exemption.
Backup withholding is not
an additional tax. The amount of any backup withholding from a payment generally will be allowed as a credit against the holder’s
U.S. federal income tax liability and may entitle the holder to a refund, provided that the required information is timely furnished
to the IRS.
Foreign Accounts
The Code generally imposes
a U.S. federal withholding tax of 30% on dividends and, subject to the discussion below regarding proposed regulations recently
issued by the U.S. Treasury Department, the gross proceeds of a disposition of our securities paid to a “foreign financial
institution” (as specifically defined for this purpose), unless such institution enters into an agreement with the U.S.
government to, among other things, withhold on certain payments and to collect and provide to the U.S. tax authorities substantial
information regarding U.S. account holders of such institution (which includes certain equity and debt holders of such institution,
as well as certain account holders that are foreign entities with U.S. owners) or otherwise qualifies for an exemption from these
rules. A U.S. federal withholding tax of 30% also applies to dividends and, subject to the discussion below regarding proposed
regulations recently issued by the U.S. Treasury Department, will apply to the gross proceeds of a disposition of our securities
paid to a non-financial foreign entity (as defined in the Code), unless such entity provides the withholding agent with either
a certification that it does not have any substantial direct or indirect. “United States owners”
(as defined in the Code), provides information regarding each substantial United States owners of the entity, or otherwise qualifies
for an exemption from these rules.
Under certain circumstances,
a non-U.S. holder might be eligible for refunds or credits of such taxes. An intergovernmental agreement between the United States
and an applicable foreign country may modify the requirements described in this paragraph.
The U.S. Treasury Department recently released proposed
regulations which, if finalized in their present form, would eliminate the federal withholding tax of 30% applicable to the gross
proceeds of a sale or other disposition of our common stock, pre-funded warrants and common warrants. In its preamble to such
proposed regulations, the U.S. Treasury Department stated that taxpayers may generally rely on the proposed regulations until
final regulations are issued. Prospective purchasers should consult their own tax advisors regarding the possible impact of these
rules on their investment in our common stock, pre-funded warrants and common warrants, and the possible impact of these rules
and the proposed regulations on the entities through which they hold our common stock, pre-funded warrants and common warrants,
including, without limitation, the process and deadlines for meeting the applicable requirements to prevent the imposition of
this 30% withholding tax.
EACH PROSPECTIVE PURCHASER SHOULD CONSULT ITS TAX ADVISOR
REGARDING THE PARTICULAR U.S. FEDERAL, STATE AND LOCAL AND NON-U.S. TAX CONSEQUENCES OF PURCHASING, HOLDING AND DISPOSING OF OUR
SECURITIES, INCLUDING THE CONSEQUENCES OF ANY PROPOSED CHANGE IN APPLICABLE LAWS. IN ADDITION, SIGNIFICANT CHANGES IN U.S. FEDERAL
TAX LAWS WERE RECENTLY ENACTED. PROSPECTIVE PURCHASERS SHOULD ALSO CONSULT WITH THEIR TAX ADVISORS WITH RESPECT TO SUCH CHANGES
IN U.S. TAX LAW AS WELL AS POTENTIAL CONFORMING CHANGES IN STATE TAX LAWS.
LEGAL
MATTERS
The validity of the securities
offered by this prospectus will be passed upon for us by Ballard Spahr LLP, Philadelphia, Pennsylvania. Haynes and Boone, LLP,
New York, New York is acting as counsel for the underwriter in connection with the securities offered hereby.
EXPERTS
The consolidated financial
statements of OpGen, Inc. and its subsidiaries as of December 31, 2018 and 2017, and for the years then ended, have been incorporated
by reference herein in reliance upon the report, also incorporated by reference herein, of CohnReznick LLP, an independent registered
public accounting firm, and upon the authority of said firm as experts in accounting and auditing. The audit report covering the
December 31, 2018 consolidated financial statements contains an explanatory paragraph that states that the Company has experienced
losses and negative cash flows from operations since its inception, has an accumulated deficit, and has debt obligations which
raise substantial doubt about its ability to continue as a going concern. The consolidated financial statements do not include
any adjustments that might result from the outcome of that uncertainty.
The audited historical combined
financial statements of the Curetis Business as of December 31, 2018, December 31, 2017 and January 1, 2017 and for each of the
two years in the period ended December 31, 2018 included in this Prospectus have been so included in reliance on the report (which
contains an explanatory paragraph relating to the Company's ability to continue as a going concern as described in Note 3.22 to
the financial statements) of PricewaterhouseCoopers GmbH Wirtschaftsprüfungsgesellschaft, independent accountants, given
on the authority of said firm as experts in auditing and accounting. PricewaterhouseCoopers GmbH Wirtschaftsprüfungsgesellschaft
is a member of the Chamber of Public Accountants (Wirtschaftsprüferkammer), Berlin, Germany.
WHERE
YOU CAN FIND ADDITIONAL INFORMATION
We filed with the SEC a registration
statement under the Securities Act of 1933 for the shares of common stock in this offering. This prospectus does not contain all
of the information in the registration statement and the exhibits and schedule that were filed with the registration statement.
For further information with respect to us and our common stock, we refer you to the registration statement and the exhibits and
schedule that were filed with the registration statement. Statements contained in this prospectus about the contents of any contract
or any other document that is filed as an exhibit to the registration statement are not necessarily complete, and we refer you
to the full text of the contract or other document filed as an exhibit to the registration statement. The SEC maintains a website
that contains reports, proxy and information statements, and other information regarding registrants that file electronically
with the SEC. The address of the website is www.sec.gov.
We file periodic reports under
the Securities Exchange Act of 1934, including annual, quarterly and special reports, and other information with the Securities
and Exchange Commission. These periodic reports and other information are available for inspection and copying at the SEC regional
offices, public reference facilities and on the website of the SEC referred to above.
We make available free of
charge on or through our internet website our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on
Form 8-K, and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Securities Exchange Act
of 1934 as soon as reasonably practicable after we electronically file such material with, or furnish it to, the SEC. The information
found on our website, www.opgen.com, other than as specifically incorporated by reference in this prospectus, is not part of this
prospectus.
INCORPORATION
OF CERTAIN INFORMATION BY REFERENCE
The SEC allows us to “incorporate
by reference” information from other documents that we file with it, which means that we can disclose important information
to you by referring you to those documents. The information incorporated by reference is considered to be part of this prospectus.
Information in this prospectus supersedes information incorporated by reference that we filed with the SEC prior to the date of
this prospectus, while information that we file later with the SEC will automatically update and supersede the information in
this prospectus. We incorporate by reference into this prospectus and the registration statement of which this prospectus is a
part the information or documents listed below that we have filed with the SEC (Commission File No. 001-37367):
|
|
·
|
our
Quarterly Reports on Form 10-Q for the quarters ended March
31, 2019 and June
30, 2019, filed with the SEC on May 15, 2019 and August 14, 2019, respectively;
|
|
|
·
|
our
Current Reports on Form 8-K filed with the SEC on February
25, 2019 (Items 5.02 and 9.01), March
26, 2019 (Items 1.01, 8.01 and 9.01), April
15, 2019 (Item 5.02), May
10, 2019 (Item 3.01), May
14, 2019 (Items 8.01 and 9.01), July
1, 2019 (Item 5.02), August
22, 2019 (Item 5.07), August
23, 2019 (Item 3.01), August
28, 2019 (Items 5.03 and 9.01) and September
4, 2019 (Items 1.01 and 9.01 (except for Exhibit 99.2)) and September 18, 2019
(Item 8.01).;
|
|
|
·
|
the
description of our common stock contained in the Registration Statement on Form
8-A filed on April 30, 2015 and any amendments to such Registration Statement filed
subsequently thereto, including all amendments or reports filed for the purpose of updating
such description.
|
We also incorporate by
reference any future filings (other than current reports furnished under Item 2.02 or Item 7.01 of Form 8-K and exhibits filed
on such form that are related to such items unless such Form 8-K expressly provides to the contrary) made with the SEC after the
effective date of the registration statement of which this prospectus forms a part pursuant to Sections 13(a), 13(c), 14 or 15(d)
of the Exchange Act, until the termination of the offering of the securities made by this prospectus and will become a part of
this prospectus from the date that such documents are filed with the SEC. Information in such future filings updates and supplements
the information provided in this prospectus. Any statements in any such future filings will automatically be deemed to modify
and supersede any information in any document we previously filed with the SEC that is incorporated or deemed to be incorporated
herein by reference to the extent that statements in the later filed document modify or replace such earlier statements.
We will furnish to you, on
written or oral request, a copy of any or all of the documents that have been incorporated by reference, including exhibits to
these documents. You may request a copy of these filings at no cost by writing or telephoning our Secretary at the following address
and telephone number:
OpGen,
Inc.
Attention: Timothy C. Dec, Corporate Secretary
708 Quince Orchard Road, Suite 205
Gaithersburg, MD 20878
Telephone No.: (240) 813-1260
Copies of these filings are
also available through the “Investor” section of our website at www.opgen.com. For other ways to obtain a copy of
these filings, please refer to “Where You Can Find More Information” above.
Any information in any of
the foregoing documents will automatically be deemed to be modified or superseded to the extent that information in this prospectus
modifies or replaces such information.
CURETIS
BUSINESS
COMBINED FINANCIAL STATEMENTS
Combined Financial Statements for the Years Ended December 31, 2018 and 2017
|
Combined Statement of Operations
and Other Comprehensive Loss
|
|
F-2
|
|
Combined Statement of Financial Position
|
|
F-3
|
|
Combined Statement of Cash Flows
|
|
F-4
|
|
Combined Statement of Changes in Equity
|
|
F-5
|
|
Notes to Combined Financial Statements
|
|
F-6
|
|
Report of Independent Auditors
|
|
F-48
|
Unaudited
Interim Condensed Combined Financial Statement for the Six Months Ended June 30, 2019 and 2018
|
Unaudited Interim Condensed
Combined Statement of Operations and Other Comprehensive Loss
|
|
F-50
|
|
Unaudited Interim Condensed Combined Statement
of Financial Position
|
|
F-51
|
|
Unaudited Interim Condensed Combined Statement
of Cash Flows
|
|
F-52
|
|
Unaudited Interim Condensed Combined Statement
of Changes in Equity
|
|
F-53
|
|
Notes to Unaudited Interim Condensed Combined
Financial Statements
|
|
F-54
|
|
|
|
|
CURETIS
BUSINESS
COMBINED
STATEMENT OF OPERATIONS AND OTHER COMPREHENSIVE LOSS
For
the years ended 31 December
|
in kEuro
|
|
Note
|
|
2018
|
|
2017
|
|
|
|
|
|
|
|
|
|
Revenue
|
|
|
4
|
|
|
|
1,419
|
|
|
|
1,187
|
|
|
Cost
of sales
|
|
|
5
|
|
|
|
(1,362
|
)
|
|
|
(1,321
|
)
|
|
Gross
profit (loss)
|
|
|
|
|
|
|
57
|
|
|
|
(134
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Distribution
costs
|
|
|
5,6
|
|
|
|
(8,147
|
)
|
|
|
(7,206
|
)
|
|
Administrative
expenses
|
|
|
5,7
|
|
|
|
(3,578
|
)
|
|
|
(3,185
|
)
|
|
Research
& development expenses
|
|
|
5,8
|
|
|
|
(10,566
|
)
|
|
|
(7,335
|
)
|
|
Other
income
|
|
|
|
|
|
|
625
|
|
|
|
172
|
|
|
Operating
loss
|
|
|
|
|
|
|
(21,609
|
)
|
|
|
(17,688
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Finance
income
|
|
|
|
|
|
|
34
|
|
|
|
11
|
|
|
Finance
costs
|
|
|
|
|
|
|
(1,201
|
)
|
|
|
(697
|
)
|
|
Finance
result - net
|
|
|
10
|
|
|
|
(1,167
|
)
|
|
|
(686
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net
loss before income tax
|
|
|
|
|
|
|
(22,776
|
)
|
|
|
(18,374
|
)
|
|
Income
tax expenses
|
|
|
11
|
|
|
|
(36
|
)
|
|
|
54
|
|
|
Net
loss for the period
|
|
|
|
|
|
|
(22,812
|
)
|
|
|
(18,320
|
)
|
|
Foreign
currency translation gain (loss)*
|
|
|
|
|
|
|
88
|
|
|
|
(133
|
)
|
|
Total
comprehensive loss for the period
|
|
|
|
|
|
|
(22,724
|
)
|
|
|
(18,453
|
)
|
*
Exchange differences on translation of foreign operations, which may be recycled through profit and/or loss in the future.
The
accompanying notes are an integral part of these combined financial statements.
CURETIS
BUSINESS
COMBINED
STATEMENT OF FINANCIAL POSITION
|
in kEuro
|
|
Note
|
|
31
December 2018
|
|
31
December 2017
|
|
1
January 2017
|
|
|
|
|
|
|
|
|
|
|
|
Current
assets
|
|
|
|
|
|
|
11,888
|
|
|
|
10,204
|
|
|
|
12,210
|
|
|
Cash
and cash equivalents
|
|
|
21
|
|
|
|
4,800
|
|
|
|
3,468
|
|
|
|
6,434
|
|
|
Trade
receivables
|
|
|
12,21
|
|
|
|
323
|
|
|
|
200
|
|
|
|
101
|
|
|
Other
receivables, related party
|
|
|
25
|
|
|
|
453
|
|
|
|
824
|
|
|
|
1,370
|
|
|
Inventories
|
|
|
13
|
|
|
|
6,052
|
|
|
|
5,453
|
|
|
|
4,078
|
|
|
Prepaid
expenses and other current assets
|
|
|
14
|
|
|
|
260
|
|
|
|
259
|
|
|
|
227
|
|
|
Non-current
assets
|
|
|
|
|
|
|
10,850
|
|
|
|
11,324
|
|
|
|
12,311
|
|
|
Intangible
assets
|
|
|
15
|
|
|
|
7,425
|
|
|
|
7,524
|
|
|
|
7,520
|
|
|
Property,
plant and equipment
|
|
|
16
|
|
|
|
3,196
|
|
|
|
3,566
|
|
|
|
4,466
|
|
|
Other
non-current financial assets
|
|
|
21
|
|
|
|
158
|
|
|
|
156
|
|
|
|
325
|
|
|
Deferred
tax assets
|
|
|
11
|
|
|
|
71
|
|
|
|
78
|
|
|
|
—
|
|
|
Total
assets
|
|
|
|
|
|
|
22,738
|
|
|
|
21,528
|
|
|
|
24,521
|
|
|
Current
liabilities
|
|
|
|
|
|
|
5,773
|
|
|
|
2,890
|
|
|
|
2,501
|
|
|
Trade
and other payables
|
|
|
21
|
|
|
|
921
|
|
|
|
850
|
|
|
|
672
|
|
|
Other
liabilities, related party
|
|
|
25
|
|
|
|
187
|
|
|
|
580
|
|
|
|
617
|
|
|
Provisions
current
|
|
|
|
|
|
|
65
|
|
|
|
124
|
|
|
|
51
|
|
|
Tax
liabilities
|
|
|
11
|
|
|
|
22
|
|
|
|
24
|
|
|
|
10
|
|
|
Other
current liabilities
|
|
|
18
|
|
|
|
881
|
|
|
|
838
|
|
|
|
683
|
|
|
Other
current financial liabilities
|
|
|
19,21
|
|
|
|
3,697
|
|
|
|
474
|
|
|
|
468
|
|
|
Non-current
liabilities
|
|
|
|
|
|
|
13,993
|
|
|
|
10,385
|
|
|
|
40
|
|
|
Provisions
non-current
|
|
|
|
|
|
|
44
|
|
|
|
43
|
|
|
|
40
|
|
|
Other
non-current financial liabilities
|
|
|
20,21
|
|
|
|
13,949
|
|
|
|
10,342
|
|
|
|
—
|
|
|
Total
liabilities
|
|
|
|
|
|
|
19,766
|
|
|
|
13,275
|
|
|
|
2,541
|
|
|
Equity
|
|
|
|
|
|
|
2,972
|
|
|
|
8,253
|
|
|
|
21,980
|
|
|
Subscribed
capital
|
|
|
|
|
|
|
5,554
|
|
|
|
5,554
|
|
|
|
5,554
|
|
|
Capital
reserve
|
|
|
|
|
|
|
157,847
|
|
|
|
140,402
|
|
|
|
135,675
|
|
|
Currency
translation differences
|
|
|
|
|
|
|
(75
|
)
|
|
|
(160
|
)
|
|
|
(28
|
)
|
|
Accumulated
deficit
|
|
|
|
|
|
|
(160,354
|
)
|
|
|
(137,543
|
)
|
|
|
(119,221
|
)
|
|
Total
Equity and liabilities
|
|
|
|
|
|
|
22,738
|
|
|
|
21,528
|
|
|
|
24,521
|
|
The
accompanying notes are an integral part of these combined financial statements.
CURETIS
BUSINESS
COMBINED
STATEMENT OF CASH FLOWS
For
the years ended 31 December
|
in kEuro
|
|
Note
|
|
2018
|
|
2017
|
|
Net
loss for the period
|
|
|
|
|
|
|
(22,812
|
)
|
|
|
(18,320
|
)
|
|
Adjustment
for:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-
Net finance income (costs)
|
|
|
10
|
|
|
|
1,167
|
|
|
|
686
|
|
|
-
Depreciation, amortization and impairments
|
|
|
15,16
|
|
|
|
1,257
|
|
|
|
1,327
|
|
|
-
Share based payment expense
|
|
|
22
|
|
|
|
366
|
|
|
|
528
|
|
|
-
Changes in deferred tax assets and liabilities
|
|
|
11
|
|
|
|
7
|
|
|
|
(78
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Changes
in working capital relating to:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
- Inventories
|
|
|
13
|
|
|
|
(599
|
)
|
|
|
(1,375
|
)
|
|
-
Trade receivables and other receivables
|
|
|
12,14
|
|
|
|
245
|
|
|
|
586
|
|
|
-
Trade payables and other payables
|
|
|
21,18
|
|
|
|
(422
|
)
|
|
|
153
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income
taxes received (+) / paid (-)
|
|
|
|
|
|
|
36
|
|
|
|
(54
|
)
|
|
Interest
paid (-)
|
|
|
|
|
|
|
(406
|
)
|
|
|
6
|
|
|
Net
cash flow used in operating activities
|
|
|
|
|
|
|
(21,161
|
)
|
|
|
(16,541
|
)
|
|
Payments
for intangible assets
|
|
|
|
|
|
|
(119
|
)
|
|
|
(110
|
)
|
|
Payments
for property, plant and equipment
|
|
|
|
|
|
|
(670
|
)
|
|
|
(323
|
)
|
|
Net
cash flow used in investing activities
|
|
|
|
|
|
|
(789
|
)
|
|
|
(433
|
)
|
|
Proceeds
from other non-current financial liabilities
|
|
|
|
|
|
|
3,000
|
|
|
|
10,000
|
|
|
Proceeds
from current financial liabilities (convertible notes), net of issuance cost
|
|
|
|
|
|
|
3,109
|
|
|
|
—
|
|
|
Capital
increase from shareholder
|
|
|
|
|
|
|
15,984
|
|
|
|
3,000
|
|
|
Shareholder
contributions
|
|
|
|
|
|
|
1,095
|
|
|
|
1,199
|
|
|
Net
cash flow provided by financing activities
|
|
|
|
|
|
|
23,188
|
|
|
|
14,199
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net decrease
/ increase in cash and cash equivalents
|
|
|
|
|
|
|
1,238
|
|
|
|
(2,775
|
)
|
|
Net
cash and cash equivalents at the beginning of the year
|
|
|
|
|
|
|
3,468
|
|
|
|
6,434
|
|
|
Effects
of exchange rate changes on cash and cash equivalents
|
|
|
|
|
|
|
94
|
|
|
|
(191
|
)
|
|
Net
Cash and cash equivalents at the end of the period
|
|
|
|
|
|
|
4,800
|
|
|
|
3,468
|
|
The
accompanying notes are an integral part of these combined financial statements.
CURETIS
BUSINESS
COMBINED
STATEMENT OF CHANGES IN EQUITY
For
the years ended 31 December
|
|
|
|
|
|
|
Currency
|
|
|
|
|
|
|
|
Subscribed
|
|
Capital
|
|
translation
|
|
Accumulated
|
|
TOTAL
|
|
in
kEuro
|
|
capital
|
|
Reserve
|
|
difference
|
|
deficit
|
|
equity
|
|
Balance
at 1 January 2017
|
|
|
5,554
|
|
|
|
135,675
|
|
|
|
(28
|
)
|
|
|
(119,222
|
)
|
|
|
21,979
|
|
|
Loss of the period
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(18,320
|
)
|
|
|
(18,320
|
)
|
|
Other
comprehensive income
|
|
|
|
|
|
|
|
|
|
|
(132
|
)
|
|
|
|
|
|
|
(132
|
)
|
|
Total
comprehensive income
|
|
|
0
|
|
|
|
0
|
|
|
|
(132
|
)
|
|
|
(18,320
|
)
|
|
|
(18,452
|
)
|
|
Transactions
with owners in their capacity as owners
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Capital
increase
|
|
|
|
|
|
|
3,000
|
|
|
|
|
|
|
|
|
|
|
|
3,000
|
|
|
Shareholder
contributions
|
|
|
|
|
|
|
1,199
|
|
|
|
|
|
|
|
|
|
|
|
1,199
|
|
|
Share-based
payments
|
|
|
|
|
|
|
528
|
|
|
|
|
|
|
|
|
|
|
|
528
|
|
|
Balance
as of 31 December 2017
|
|
|
5,554
|
|
|
|
140,402
|
|
|
|
(160
|
)
|
|
|
(137,542
|
)
|
|
|
8,254
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Currency
|
|
|
|
|
|
|
|
Subscribed
|
|
Capital
|
|
translation
|
|
Accumulated
|
|
TOTAL
|
|
in
kEuro
|
|
capital
|
|
Reserve
|
|
difference
|
|
deficit
|
|
equity
|
|
Balance
at 1 January 2018
|
|
|
5,554
|
|
|
|
140,402
|
|
|
|
(160
|
)
|
|
|
(137,542
|
)
|
|
|
8,254
|
|
|
Loss of the period
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(22,812
|
)
|
|
|
(22,812
|
)
|
|
Other
comprehensive income
|
|
|
|
|
|
|
|
|
|
|
85
|
|
|
|
|
|
|
|
85
|
|
|
Total
comprehensive income
|
|
|
0
|
|
|
|
0
|
|
|
|
85
|
|
|
|
(22,812
|
)
|
|
|
(22,727
|
)
|
|
Transactions
with owners in their capacity as owners
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Capital
increase
|
|
|
|
|
|
|
15,984
|
|
|
|
|
|
|
|
|
|
|
|
15,984
|
|
|
Shareholder
contributions
|
|
|
|
|
|
|
1,095
|
|
|
|
|
|
|
|
|
|
|
|
1,095
|
|
|
Share-based
payments
|
|
|
|
|
|
|
366
|
|
|
|
|
|
|
|
|
|
|
|
366
|
|
|
Balance
as of 31 December 2018
|
|
|
5,554
|
|
|
|
157,847
|
|
|
|
(75
|
)
|
|
|
(160,354
|
)
|
|
|
2,972
|
|
The
accompanying notes are an integral part of these combined financial statements.
CURETIS
BUSINESS
NOTES
TO THE COMBINED FINANCIAL STATEMENTS
|
|
1.
|
GENERAL
INFORMATION ABOUT THE COMPANY
|
These
combined financial statements comprise the business of Curetis N.V. (collectively referred to as “the Group”, “Curetis”
or the “Company”). Refer to note 2.1 for further information. The Group´s headquarters is located at Max-Eyth-Str.
42, 71088 Holzgerlingen, Germany. The Group is an early commercial-stage molecular diagnostics (MDx) company focused on rapid
infectious disease testing for hospitalized patients with the aim to improve the treatment of hospitalized, critically ill patients
with suspected microbial infections. The Curetis business is primarily operated by Curetis GmbH and its wholly owned subsidiaries.
The
first Group entity, Curetis AG, was created in Germany in 2007 and was primarily funded through equity investments from venture
capital and private equity investors. In 2015, in connection with an initial public offering (“IPO”), Curetis N.V.
was created as a parent entity to Curetis AG and in that same year the stock of Curetis N.V. was sold in an IPO on the Euronext
market. In 2016 Curetis AG was changed to Curetis GmbH. Since 2015, Curetis has been financed through contributions from Curetis
N.V. from proceeds of the initial offerings, secondary offerings, various other financing agreements Curetis N.V. has entered
into, including Convertible Notes (see notes 19 and 21), the EIB financing (see note 20) and government grants.
At
15 September 2019 the Management Board authorized the combined financial statements for issue and passed it through to the Supervisory
Board for review and authorization.
1.1.
General Information about the business and the commercial development of the Company
The
Group has developed the innovative Unyvero molecular diagnostic solution for comprehensive infectious disease testing. Curetis’
proprietary application portfolio for its Unyvero system currently consists of several CE-marked applications:
|
|
●
|
The
Unyvero HPN (Hospitalized Pneumonia) cartridge for the detection of pathogens and antibiotic
resistances to aid diagnosing pneumonia.
|
|
|
●
|
The
Unyvero ITI (Implant and tissue infections) cartridge for the detection of pathogens
and antibiotic resistance markers in diagnosis of prosthetic joint infections, surgical
site infections, infections associated with implants, infections of the deep skin and
soft tissue, burn wounds as well as diabetic foot, cellulitis and others.
|
|
|
●
|
The
Unyvero BCU (Blood culture) cartridge for the detection of pathogens (bacteria and fungi)
and antibiotic resistance markers in bloodstream infections.
|
|
|
●
|
The
Unyvero IAI (Intra-abdominal infections) cartridge for the detection targeted microorganisms
and antibiotic resistance markers.
|
|
|
●
|
The
Unyvero UTI (Urinary tract infections) cartridge for the detection of severe cases of
urinary tract infections, microorganisms and antibiotic resistance markers.
|
In
addition to the existing Unyvero A50 multiplex platform, Curetis has expanded its product portfolio with the development of a
low- and midplex analyzer, the new Unyvero A30 RQ for Unyvero integration or as a standalone operation.
Furthermore,
in Q4-2016 Curetis acquired the GEAR database from Siemens, which is the most comprehensive database on genetics of antibiotic
resistance. In 2017, Curetis established Ares Genetics GmbH, a wholly-owned subsidiary of Curetis GmbH in Vienna, Austria. Ares
Genetics is dedicated to maximize the R&D and related scientific and business opportunities of the GEAR assets for the entire
Group.
|
|
2.
|
BASIS
OF PREPARATION – COMBINED FINANCIAL STATEMENTS
|
2.1.
Basis of presentation
The
accompanying combined financial statements of Curetis have been prepared for filing with the United States Securities and Exchange
Commission (SEC) in connection with the proposed acquisition of all of the outstanding shares of Curetis GmbH by OpGen Inc. (“OpGen”),
pursuant to an agreement to combine the two companies’ businesses. Following the agreement, OpGen will acquire 100% of Curetis
GmbH’s assets and liabilities through the acquisition of all outstanding shares of Curetis GmbH, including the Curetis name,
as well as the outstanding indebtedness of Curetis N.V. under certain convertible notes, including providing that the conversion
rights of the notes may be changed from a right to convert into shares of Curetis N.V to a right to convert into shares of OpGen.
In addition, OpGen has also agreed to acquire all of the assets of Curetis N.V. that are solely and exclusively related to the
business of Curetis GmbH and assume (1) the Curetis N.V. 2016 Stock Option Plan, as amended, and the outstanding awards thereunder,
or the 2016 Stock Option Plan, and (2) the obligation to issue equity to the holders of awards under the Curetis AG Phantom Stock
Option Plan, or the PSOP. OpGen will also assume all of the liabilities of Curetis N.V. solely and exclusively related to the
business being acquired.
The
business combination is subject to a number of conditions including (i) the satisfaction of customary conditions to closing for
a transaction of this type, including the absence of a material adverse event for either party, (ii) for each OpGen and Curetis,
appropriate approvals by their respective shareholders, (iii) for Curetis, consents from certain debt financing providers, (iv)
OpGen’s Form S-4 having been declared effective by the U.S. Securities and Exchange Commission, (v) the new shares of OpGen’s
common stock to be issued (or reserved for issuance) in connection with the transaction having been approved for listing on Nasdaq
and (vi) OpGen having secured additional funding prior to Closing.
The
business of Curetis N.V. is primarily operated by Curetis GmbH and its wholly owned subsidiaries. However, certain costs related
to the Curetis business, primarily related to the compensation of certain members of senior management and its supervisory board,
have historically been incurred by Curetis N.V. but have not been recharged by Curetis N.V. to Curetis GmbH or its subsidiaries.
SEC Staff Accounting Bulletin (SAB) Topic 1.B. (“SAB 1.B”) Allocation of Expenses and Related Disclosure in Financial
Statements of Subsidiaries, Divisions or Lessor Business Components of Another Entities states that the historical income statements
of a registrant should reflect all of its costs of doing business and therefore in specific situations requires a subsidiary to
reflect certain expenses incurred by the parent on its behalf. In addition, the combined financial statements include the convertible
notes issued by Curetis N.V. as well as related expenses. The proceeds of the issuance of the convertible notes were historically
contributed to the Curetis GmbH via cash contributions to capital reserves. Accordingly, the combined financial statements of
Curetis have been prepared to combine the consolidated financial statements of Curetis GmbH together with certain costs incurred
by Curetis N.V. on behalf of Curetis GmbH. As a result, the combined financial statements of Curetis does not currently constitute
a separate group of legal entities.
The
combined financial statements and notes have been prepared in accordance with International Financial Reporting Standards as issued
by the International Accounting Standards Board (IASB). During 2017 and 2018, the costs incurred by Curetis N.V. that have been
allocated to the Company for the purposes of preparing the combined financial statements are based on a specific identification
basis where possible. Management believes that the assumptions used in determining these allocations are reasonable. However,
the financial statements may not necessarily reflect the Company’s financial position, results of operations, or cash flows
in the future, or what its financial position, results of operations, or cash flows would have been if it had been a stand-alone
entity during the periods presented.
The
Group has not published standalone financial statements in the past. As a result, these combined financial statements have been
derived from the Curetis N.V. accounting records, which were prepared in accordance with IFRS. As it is the first time that the
Group is applying IFRS for the preparation of these standalone combined financial statements, IFRS 1 (“First-time Adoption
of International Financial Reporting Standards”) was required to be applied. Additional IFRS 1 disclosures related to the
1 January 2017 combined statement of financial position are presented in the notes to the financial statements; however, the footnotes
exclude the various reconciliation disclosures otherwise required by IFRS 1 as they are not applicable.
IFRS
does not provide principles for the preparation of combined financial statements for carve-out financial statements, and accordingly
in preparing the combined financial statements certain accounting and allocation conventions commonly used in practice for the
preparation of carve-out financial statements were applied. The assets and liabilities included in the combined balance sheets
were measured at the carrying amounts recorded in the Curetis N.V. consolidated financial statements.
The
combined financial statements have been prepared on the historical cost basis, except for certain assets and liabilities as separately
stated in Note 3 “Summary of significant accounting policies”. The combined statement of operations and other comprehensive
loss has been prepared in accordance with the function of expense method. The financial statements have been prepared on a going
concern basis (see also Note 3.22 below). These combined financial statements are presented in Euro – where appropriate
– have been rounded to the nearest thousand (abbreviated kEUR). All intercompany accounts and transactions have been eliminated
in the combination. The financial year corresponds to the calendar year.
2.2.
Scope of combination
Curetis
GmbH is domiciled in Germany.
Details
of the Group´s subsidiaries at the end of the reporting period are as follows:
|
Name
|
|
Registration
No.
|
|
Country
|
|
Participation
|
|
Main
activity
|
|
Curetis
USA Inc.
|
|
|
EIN
81-3113346
|
|
|
|
USA
|
|
|
|
100.00%
|
|
|
|
Sale
of molecular diagnostic products
|
|
Curetis
UK Ltd.
|
|
|
10164457
|
|
|
|
UK
|
|
|
|
100.00%
|
|
|
|
Sale
of molecular diagnostic products
|
|
Curetis
France S.A.R.L.
|
|
|
TI
822952511
|
|
|
|
France
|
|
|
|
100.00%
|
|
|
|
Sale
of molecular diagnostic products
|
|
Curetis
BeNeLux B.V.
|
|
|
KvK66281814
|
|
|
|
Netherlands
|
|
|
|
100.00%
|
|
|
|
Sale
of molecular diagnostic products
|
|
Curetis
Schweiz GmbH
|
|
|
CHE-228.103.501
|
|
|
|
Switzerland
|
|
|
|
100.00%
|
|
|
|
Sale
of molecular diagnostic products
|
|
Ares
Genetics GmbH
|
|
|
468899h
|
|
|
|
Austria
|
|
|
|
100.00%
|
|
|
|
Maximize
R&D and related scientific opportunities with Aresdb Bio-IT platform (previously GEAR)
|
2.3.
Critical accounting judgements and key sources of estimation uncertainty
The
preparation of financial statements in conformity with IFRS requires management to make estimates and assumptions that affect
the reported amounts of assets and liabilities, the disclosure of contingent assets and liabilities at the date of the financial
statements and the reported amounts of revenue, income and expenses during the reporting periods. Significant estimates and assumptions
reflected in these combined financial statements include, but are not limited to, the useful life of intangible assets, provisions,
and inventory valuation. The Company bases its estimates on historical experience, known trends and other market-specific or other
relevant factors that it believes to be reasonable under the circumstances. On an ongoing basis, management evaluates its estimates
as there are changes in circumstances, facts and experience. Actual results may differ from those estimates or assumptions.
Preparing
these carve-out combined financial statements required management to make judgement within the identification of certain costs
incurred by Curetis N.V. on behalf of Curetis GmbH and reflected back to the combined financial statements of Curetis GmbH. Management
evaluated on historical experience the best approach by identifying such costs.
|
|
3.
|
SUMMARY
OF SIGNIFICANT ACCOUNTING POLICIES
|
The
principal accounting policies set out below have been applied consistently to all periods presented in these combined financial
statements, unless otherwise stated.
3.1.
New standards and interpretations applied for the first time
The
International Accounting Standards Board (IASB) continues to issue new standards, interpretations and amendments to existing standards.
Curetis applies these new standards as required. Curetis has not opted for early adoption for any of these standards. New standards,
amendments to standards and new or amended interpretations are effective for annual periods beginning on or after 1 January 2018,
and have been applied in preparing these financial statements.
|
Standard/Interpretation
|
|
Content
|
|
Application
mandatory from
|
|
Amendment to IFRS 9
|
|
Prepayment Features with Negative Compensation
|
|
1 January 2019
|
|
IFRS 16
|
|
Accounting of Leasing-transactions
|
|
1 January 2019
|
|
IFRIC 23
|
|
Uncertainty over Income Tax Treatments
|
|
1 January 2019
|
|
Amendments to IFRS 3, IAS 11, IAS 12, IAS 23
|
|
Amended by Annual Improvements to IFRS Standards 2015–2017
Cycle.
|
|
1 January 2019
|
|
Amendments to IAS 19
|
|
Plan Amendment, Curtailment or Settlement
|
|
1 January 2020
|
|
Amendments to IFRS 3
|
|
Clarifying the definition of “businesses”
|
|
1 January 2020
|
|
Amendments to IAS 1 and IAS 8
|
|
Clarifying the definition of “material”
|
|
1 January 2020
|
|
IFRS 17 (replaces IFRS 4)
|
|
Insurance Contract
|
|
1 January 2021
|
First
time adoption of IFRS 9 – financial Instruments
Transition
of IFRS 9
The
Group has applied the new IFRS 9 Financial Instruments standard for financial instruments since 1 January 2018, whereby
the exception granted by IFRS 9 Section 7.2.15 is applied for the transitional provisions for classification and measurement,
according to which, the adjustment of prior year figures is not required.
On
1 January 2018 (the date of initial application of IFRS 9), the Group´s management has assessed which business models apply
to the financial assets held by the Group and has classified its financial instruments into the appropriate IFRS 9 categories.
Trade Receivables and Cash and cash equivalents that would have previously been classified as “loan and receivables”
(LaR) are now classified at “amortized cost” (AC). The Group intends to hold the assets to maturity to collect contractual
cash flows and these cash flows consist solely of payments of principal and interest on the principal amount outstanding. There
was no difference between the previous carrying amount and the revised carrying amount of both classes at 1 January 2018 to be
recognized in opening Accumulated deficit.
Financial
Liabilities are classified as “Financial Liabilities at amortized Costs” (FLAC) which will be continued under IFRS
9. See note 21.
|
in
kEuro
|
|
LaR
|
|
AC
|
|
Accumulated
deficit
|
|
Closing
Balance 31.12.2017
|
|
|
3,824
|
|
|
|
—
|
|
|
|
—
|
|
|
Reclassify
trade receivables from LaR to AC
|
|
|
(200
|
)
|
|
|
200
|
|
|
|
—
|
|
|
Reclassify
cash and cash equivalents from LaR to AC
|
|
|
(3,468
|
)
|
|
|
3,468
|
|
|
|
—
|
|
|
Reclassify
rent deposits and pledged security deposits from LaR to AC
|
|
|
(156
|
)
|
|
|
156
|
|
|
|
—
|
|
|
Opening
Balance 01.01.2018
|
|
|
—
|
|
|
|
3,824
|
|
|
|
—
|
|
The
Group has two types of financial assets that are subject to IFRS 9´s new expected credit loss model:
|
|
●
|
debt
instruments at amortized cost (i.e. cash and cash equivalents and rent deposits)
|
The
Group was required to revise its impairment methodology under IFRS 9 for each of these classes of assets. The Group applies the
IFRS 9 simplified approach to measuring expected credit losses which uses a lifetime expected loss allowance for all trade receivables.
Historical losses have been very limited; therefore the expected losses on trade receivables is immaterial. Lifetime expected
credit losses do not significantly exceed the impairment under IAS 39.
Other
instruments are considered to have a low credit risk when the issuer has a strong capacity to meet its contractual cash flow obligations
in the near term. In that meaning, Cash and cash equivalents are only placed at banks with credit ratings at investment grade.
Rent deposits are trust assets that means that in case of a default of the counterparty the assets are separated from the insolvency
estate and are paid back primarily.
Due
to the immaterial expected losses on financial assets, the Group did not change the loss allowance as of 1 January 2018. The recognized
loss allowance contained only specific loss provisions which are assigned to stage 3 of the new credit deterioration model and
expected losses are immaterial.
Curetis
did not apply hedge accounting under IAS 39, therefore IFRS 9 has no impact on the recognition of hedging relationships.
First
time adoption of IFRS 15 – Revenues from Contracts with Customers
Transition
of IFRS 15
The
Group has adopted IFRS 15 Revenues from Contracts with Customers from 1 January 2018. IFRS 15 establishes a comprehensive
framework for determining whether, how much and when revenue is recognized. It replaces IAS 18 Revenue and IAS 11 Construction
Contracts and related interpretations. In accordance with the transition provision in IFRS 15, the Group has adopted the new
rules by applying the modified retrospective approach. Consequently, the information presented for 2017 has not been restated.
The impact of transition from IAS 18 to IFRS 15 did not result in a significant change to the Group’s revenue recognition
policy and therefore the impact on Accumulated deficit and on other financial statement line items was immaterial and therefore
not adjusted in the opening balance sheet.
IFRS
15 Accounting policies
Under
IFRS 15, revenue is recognized when a customer obtains control of the goods or services. Determining the timing of the transfer
of control – at a point in time or over time – requires judgement.
The
Group’s revenue consist mainly of the sale of Unyvero Application cartridges and Unyvero Systems. The sale of Unyvero Application
cartridges and the sale of Unyvero Systems represent separate performance obligations. Curetis recognizes revenues at a point
in time when the control is transferred to the customer. The control of the product transfers upon shipment to the customer or
when the product is made available to the customer, provided that the Group did not retain any significant risks of ownerships
or future obligations with respect to the product shipped.
The
Group has identified its performance obligation and noted that there are no other significant performance obligations outside
the shipment of the products as outlined above. In certain contracts Curetis has a relationship both as a supplier and as a customer.
Subject to the underlying transaction, any costs associated with services received by Curetis as a customer are recorded in operating
expenses.
Furthermore,
the Group offers Bio-IT related services via its subsidiary Ares Genetics GmbH. the Group recognizes revenues for such project
related services over the period of time in which the services are being provided, in accordance with IFRS 15.
Service
revenues also includes license fees. A license may be provide the customer (licensee) the right to use the Company’s (licensor)
intellectual property as it exists at the point in time the license is granted. For such license, revenue is recognized at a point
in time when controls transfers to the licensee (i.e., the licensee is able to use and benefit from the license) and the license
period begins. As opposed to the right to use IP, as described above, a license may provide access to the Company’s IP as
it exists throughout the license period (right to access IP), such license being a performance obligation satisfied over time
which results in revenue recognized over time accordingly, provided that all criteria in IFRS 15.
Revenue
is measured based on the consideration expected to be received. The Group also evaluated existing contracts with customers and
has determined it currently does not have any contracts or agreements with an enforceable right with regard to minimum purchase
obligations.
Payment
is generally due at the time of delivery or in line with customary payment terms. Deferred payment terms may be agreed in rare
circumstances, however; the deferral never exceeds twelve months. The transaction price is therefore not adjusted for the effects
of a significant financing component.
The
other new standards and amendments to standards noted in the table above had no effect on the combined financial statements of
the Group as of 31 December 2017 and 2018.
3.2.
Standards, interpretations, and amendments issued, but not yet applied
The
following new standards and interpretations and amendments to existing standards will become effective after 1 January 2019.
|
Standard/Interpretation
|
|
Content
|
|
Application
mandatory from
|
|
Amendment
to IFRS 9
|
|
Prepayment
Features with Negative Compensation
|
|
1
January 2019
|
|
IFRS
16
|
|
Accounting
of Leasing-transactions
|
|
1
January 2019
|
|
IFRIC
23
|
|
Uncertainty
over Income Tax Treatments
|
|
1
January 2019
|
|
Amendments
to IFRS 3, 11, I
|
|
Amended
by Annual Improvements to IFRS
|
|
1
January 2019
|
|
AS
12, IAS 23
|
|
Standards
2015–2017 Cycle.
|
|
1
January 2019
|
|
Amendments
to IAS 19
|
|
Plan
Amendment, Curtailment or Settlement Clarifying the definition of businesses
|
|
1
January 2020
|
|
Amendments
to IFRS 3
|
|
Clarifying
the definition of “material”
|
|
1
January 2020
|
|
Amendments
to IAS 1 and IAS 8 IFRS 17 (replaces IFRS 4)
|
|
Insurance
Contract
|
|
1
January 2021
|
The
Group has assessed the accounting standards effective after 1 January 2019 and determined that none are likely to have a material
impact on the combined financial statements with the exception of IFRS 16.
IFRS
16 Leases replaces IAS 17 as well as the associated interpretations. The new standard requires leases to be recorded on
the balance sheets by recording a right-of-use asset and lease liability for all leases with a term of greater than 12 months.
Leases with a term of 12 months or less may be accounted for similar to existing guidance on operating leases prior to adoption.
As
at the reporting date, the Group has operating lease commitments of kEUR 1,301. Of these commitments, an immaterial amount relate
to short-term leases and low value leases which will both be recognized on a straight-line basis as expense in profit or loss.
For
the remaining lease commitments, the Group expects the adoption to result in recognition of right-of-use assets and lease liabilities
of kEUR 1,494 on its combined balance sheets. No material impacts are expected on the combined statement of operations or
net cash flows. The impact of changes under IFRS 16 to the statement of operations would be a positive impact to operating income
(loss) due to operating lease expense being replaced by depreciation and interest expense, the latter of which is not recognized
within results from operations. Further, changes to the combined statement of cash flows would be to decrease the cash used in
operating activities and decrease net cash provided by financing activities by the same amount as repayment of the principal portion
of the lease liabilities will be classified as cash flows from financing activities.
The
Group’s activities as a lessor are not material and hence the Group does not expect any significant impact on the financial
statements.
The
Group will apply the standard from its mandatory adoption date of 1 January 2019. The Group intends to apply the simplified transition
approach and will not restate comparative amounts for the year prior to adoption.
3.3.
Segment Reporting
In
accordance with IFRS 8, Curetis is a single-segment entity. The Company manages its operations as a single segment for the purposes
of assessing performance and making operating decisions. The Company’s primary focus is on research and development activities
as well as developing sales and distribution channels and relationships to further the commercialization of its offerings. The
Management Board is the chief operating decision maker, and regularly reviews the combined operating results to make decisions
about the allocation of the Company’s resources.
3.4.
Current and non-current distinction
Curetis
presents current and non-current assets and current and non-current liabilities as separate classifications in the statement of
financial position. Curetis classifies all amounts expected to be recovered or settled within twelve months after the reporting
period as ´current´ and all other amounts as ´non-current´.
3.5.
Foreign currency translation
Items
included in the financial statements of each of the Group’s entities are measured using the currency of the primary economic
environment in which the entity operates (the functional currency). The combined financial statements are presented in Euro which
is functional and presentation currency of Curetis GmbH.
Transactions
in foreign currencies are translated into Euros at the exchange rates at the date of the transaction. Monetary assets and liabilities
denominated in foreign currencies at the reporting date are translated into Euros at the exchange rate at the reporting date.
Curetis converted amounts from each corresponding currency to the functional currency with the exchange rates indicated in the
table below.
|
1
EUR =
|
|
31
December 2018
|
|
31
December 2017
|
|
1
January 2017
|
|
|
USD
|
|
|
|
1.1450
|
|
|
|
1.1993
|
|
|
|
1.0541
|
|
|
|
CHF
|
|
|
|
1.1269
|
|
|
|
1.1702
|
|
|
|
1.0739
|
|
|
|
GBP
|
|
|
|
0.8945
|
|
|
|
0.8872
|
|
|
|
0.8561
|
|
Foreign
currency transactions are translated into the functional currency using the spot exchange rate at the transaction date. Foreign
currency monetary items are translated into the functional currency using the exchange rate as of the end of the reporting period.
Non-monetary items measured at historical cost in foreign currencies are translated into the functional currency using the exchange
rates at the transaction date. Non-monetary items measured at fair value that are denominated in foreign currencies are translated
into the functional currency using the exchange rates at the date when the fair value is measured. Exchange differences arising
from the translation or settlement are recognized in profit or loss, except for those recognized in other comprehensive loss.
On
consolidation, exchange differences arising from the translation of any net investment in foreign entities, and of borrowings
and other financial instruments designated as hedges of such investments, are recognized in other comprehensive income or loss.
When a foreign operation is sold or any borrowings forming part of the net investment are repaid, the associated exchange differences
are reclassified to profit or loss, as part of the gain or loss on sale.
3.6.
Cash flow statement
The
combined statement of cash flows has been prepared using the indirect method. The balance of cash and cash equivalents as at the
date of the financial statements disclosed in the cash flow statement is comprised of cash and cash equivalents. Cash comprises
cash on hand and demand deposits. Cash equivalents are short-term bank deposits and are not subject to any significant risk of
changes in value. Interest paid is included within the net cash flows from operating activities whereas interest received is included
within the net cash flows from investing activities.
3.7.
Revenue recognition
The
Group recognizes revenue most significantly from the sale of Unyvero-cartridges, disposables and systems, as well as other disposables.
Revenue
is measured based in the consideration to which the Group expects to be entitled in a contract with a customer and excludes amounts
collected on behalf of third parties (if applicable). The Group recognizes revenue when it transfers control of a product or service
to a customer. The impact of transition from IAS 18 to IFRS 15 did not result in a significant change in the Group’s revenue
recognition policy. Refer to note 3.1 for further information on adoption of the new standard and additional accounting policy
information. As of 31 December 2017 and 2018 the Group did not have material contract assets or liabilities.
3.8.
Cost of Sales
Cost
of sales includes the costs for products sold in terms of manufacturing, obsolescence write-downs of inventories as well as delivery
costs for the products sold. Manufacturing costs for products manufactured in-house include the directly allocable individual
material and production costs, the allocable parts of the overhead costs for production including depreciation of production equipment
and changes in semi-finished and finished inventories.
3.9.
Research and development expenses
Research
expenses are defined as costs incurred for investigations undertaken with the prospect of gaining new scientific or technical
knowledge and understanding. Development expenses are defined as costs incurred for the application of research findings or other
knowledge to a plan or design for the production of new or substantially improved materials, devices, products, processes, systems
or services before the start of commercial production or use.
Research
and development costs have historically been and will continue to be expensed as incurred until the recognition criteria outlined
in IAS 38 are met. The criteria for the recognition of development costs are closely defined: an intangible asset must be recognized
if, and only if, there is reasonable certainty that the future economic benefits that are attributable to the asset will flow
to the entity; and the cost of the asset can be measured reliably. Since Curetis’ development projects are often subject
to product development risks, clinical trial risks, regulatory approval procedures and other uncertainties, the conditions for
the recognition of costs incurred before receipt of approvals are not satisfied in the ordinary course of business of Curetis.
3.10.
Leases
Leasing
transactions are classified according to the lease agreements and to the underlying risks and rewards. Curetis has entered into
agreements in which it is the lessor and other agreements in which it is the lessee. Additionally, certain arrangements are analyzed
with regard to embedded leases (IFRIC 4). If specific criteria are met, certain arrangements should be accounted for as leases
even if they do not take the legal form of a lease. The Group did not elect to adopt IFRS 16 Leases early.
Curetis
leases certain property, plant and equipment. Leasing transactions in which Curetis is the lessee are classified either as finance
leases or operating leases. Leases of property, plant and equipment where Curetis bears substantially all of the risks and rewards
of ownership are classified as finance leases. Finance leases are recognized at the lease’s commencement at the lower of
the fair value of the leased property and the present value of the minimum lease payments. Accordingly, Curetis recognizes the
asset and the associated liability in equal amounts. The leased property is depreciated over its useful economic life or, if it
is shorter, the term of the lease. The lease liability is discounted by using the interest rate implicit to the lease.
Each
lease payment is allocated between the liability and finance charges. The corresponding rental obligations, net of finance charges,
are included in other current financial liabilities and other non-current financial liabilities. The interest element of the finance
cost is charged to the statement operations and other comprehensive loss over the lease period so as to produce a constant periodic
rate of interest on the remaining balance of the liability for each period. The property, plant and equipment acquired under finance
leases are depreciated over the shorter of the useful life of the asset and lease term.
All
other transactions not classified as a finance lease in which Curetis is the lessee, if any, would be classified as operating
leases. Payments made under operating leases (net of any incentives received from the lessor) are charged to the statement operations
and other comprehensive loss on a straight-line basis over the period of the lease.
In
2017 and 2018 Curetis did not have any material agreements in which they operated as a lessor.
3.11.
Finance income and finance costs
Finance
income and finance costs are recognized in the income statement in the period as they occur. For non-current loans expenses are
recognized using the effective interest method.
3.12.
Fair value measurements
Historic
cost is generally based on the fair value of the consideration given in exchange for assets.
Fair
value is the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market
participants at the measurement date. The fair value measurement is based on the presumption that the transaction to sell the
asset or transfer the liability takes place, either in the principal market for the asset or liability, or in the absence of a
principal market, in the most advantageous market for the asset or liability. The principal or the most advantageous market must
be accessible by the Company. The fair value of an asset or a liability is measured using the assumptions that market participants
would use when pricing the asset or liability, assuming that market participants act in their economic best interest.
All
assets and liabilities for which fair value is measured or disclosed in the financial statements are categorized within the fair
value hierarchy, described as follows, based on the lowest level input that is significant to the fair value measurement as a
whole:
-
Level 1 - Quoted prices (unadjusted) in active markets for identical assets or liabilities.
-
Level 2 - Inputs other than quoted prices included within level 1 that are observable for the asset or liability, either directly
(that is, as prices) or indirectly (that is, derived from prices).
-
Level 3 - Inputs for the asset or liability that are not based on observable market data (that is, unobservable inputs).
3.13.
Inventories
Inventories
are valued at the lower of cost or net realizable value. The cost of merchandise as well as raw, auxiliary and operating materials
is determined by using the specific identification of their individual cost method. The cost of semi-finished and finished goods
is determined using the weighted average cost method. Net realizable value is the estimated selling price in the ordinary course
of business less the estimated costs of completion and the estimated costs necessary to make the sale.
If
the net realizable value of a finished good is lower than its cost, inventories are written down to their net realizable value
and the related expenses are recognized in Cost of sales.
3.14.
Intangible assets
Licenses
and patents
Separately
acquired intangible assets are initially measured at cost. Intangible assets not yet available for use are tested for impairment
at least annually or more frequently if a potential triggering event is identified. Upon being placed into service, intangible
assets are carried at cost less accumulated amortization and impairment losses.
Intangible
assets are tested annually for impairment or more frequently if events or changes in circumstances indicate that they might be
impaired, either individually or at cash-generating unit level. An impairment loss is recognized for the amount by which the asset's
carrying amount exceeds its recoverable amount. Impairments are reversed if and to the extent that the reasons for impairment
no longer exist. The recoverable amount is defined as the higher of an asset’s fair value less cost to sell and its value
in use.
Licenses
for biomarkers are amortized according to the terms of validity of the patent (up to 17.8 years) and amortized according to the
straight-line method.
3.15.
Property, plant and equipment
Property,
plant and equipment are valued at cost less depreciation and impairment losses, if any. Cost includes direct costs (e.g. materials,
direct labor and work contracted out) and directly attributable overhead costs. Maintenance and repair costs (day-to-day servicing)
are expensed as incurred.
Asset
retirement obligations are recognized at the cost of tangible fixed assets and expensed over the asset’s estimated useful
life. The estimated useful lives of the principal property, plant and equipment categories are as follows:
|
Asset
class
|
|
Depreciation
term
|
|
Land
and buildings
|
|
Max.
10 years
|
|
Machines
and technical equipment
|
|
3-13
years
|
|
Office
equipment
|
|
2-14
years
|
|
Unyvero-Platforms
|
|
3-5
years
|
Office
equipment and Unyvero-Platforms, used for internal demands, are combined into Other tangible assets (refer to note 16).
Property,
plant and equipment are depreciated using the straight-line method, based on estimated useful life, taking into account their
respective residual value. Property, plant and equipment are reviewed for impairment whenever events or changes in circumstances
indicate that the book value of the assets concerned may not be recoverable. An impairment loss is recognized for the amount by
which the asset’s book value exceeds its recoverable amount. The recoverable amount is defined as the higher of an asset’s
fair value less cost to sell and its value in use. Impairments are reversed if and to the extent that the reasons for impairment
no longer exist.
The
assets’ residual values and useful lives are reviewed at least annually and adjusted if appropriate.
3.16.
Financial instruments
Financial
instruments are contracts that give rise to a financial asset of one entity and a financial liability or equity instrument of
another entity.
The
classification of financial instruments depends on how to characterize a financial instrument into equity instruments, debt instruments
or derivatives.
A
financial instrument is an equity instrument only if (a) the instrument includes no contractual obligation to deliver cash or
another financial asset to another entity and (b) if the instrument will or may be settled in the issuer ́s own equity instruments.
It is either:
|
|
●
|
A
non-derivative that includes no contractual obligation for the issuer to deliver a variable
number of its own equity instrument; or
|
|
|
●
|
A
derivative that will be settled only by the issuer exchanging a fixed amount of cash
or another financial asset for a fixed number of its own equity instruments.
|
A
financial instrument is a debt instruments are contractual rights and obligations with defined terms for amount and timing to
pay.
A
derivative financial instrument is any contract with all three of the following:
(a)
its value changes in response to the change in a specified interest rate, financial instrument price, commodity price, foreign
exchange rate, index of prices or rates, credit rating or credit index, or other variable, provided in the case of a non-financial
variable that the variable is not specific to a party to the contract (sometimes called the ‘underlying’).
(b)
it requires no initial net investment or an initial net investment that is smaller than would be required for other types of contracts
that would be expected to have a similar response to changes in market factors.
(c)
it is settled at a future date.
Purchases
or sales of financial assets that require delivery of assets within a time frame established by regulation or convention in the
market place (regular way trades) are recognized on the trade date, i.e. the date that the Group commits to purchase or sell the
asset.
Financial
Assets
At
initial recognition, the Group measures a financial asset at its fair value plus, in the case of financial asset not at fair value
through profit or loss, transaction costs that are directly attributable to the acquisition of the financial asset. Transaction
costs of financial assets carried at fair value through profit or loss are expensed in profit or loss.
Subsequent
measurement of debt instruments depends on the Group´s business model for managing the asset and the cash flow characteristics
of the asset. The Group classifies its debt instruments into one of the following measurement categories.
Assets
that are held for collection of contractual cash flows where those cash flows represent solely payments of principal and interest
are measured at amortized cost. Interest income from these financial assets is included in finance income using the effective
interest rate method. Any gain or loss arising on the de-recognition is recorded directly in profit or loss and presented in finance
income (cost). Impairment losses are presented as separate line item in the statement of operations and other comprehensive loss.
Assets
that are held for collection of contractual cash flows and for selling the financial assets, where the assets ́ cash flows
represent solely payments of principal and interest, are measured at fair value through other comprehensive income or loss. Movements
in the carrying amount are taken through other comprehensive income or loss, except for the recognition of impairment gains or
losses, interest revenue and foreign exchange gains and losses which are recognized in profit or loss. When the financial asset
is derecognized, the cumulative gain or loss previously recognized in other comprehensive income or loss is reclassified from
equity to profit or loss and presented in finance income (cost). Interest income from the financial assets are presented in other
income (cost) and impairment expenses are presented as separate line item in the statement of operations and other comprehensive
loss.
Assets
that do not meet the criteria for amortized cost or at fair value through other comprehensive income or loss or for which the
fair value option in accordance with IFRS 9 is exercised, are measured at fair value through profit or loss. A gain or loss on
a debt investment that is subsequently measured at fair value through profit or loss is recognized in profit or loss and presented
net within finance income (cost) in the period in which it arises. Curetis does not use the fair value option.
Curetis
has elected to measure all equity instruments at fair value through profit or loss. In the current reporting period, the Group
did not hold any equity instruments.
Financial
assets with embedded derivatives are considered in their entirety when determining whether their cash flows are solely payments
of principal and interest.
Financial
Assets are derecognized when the contractual rights to the cash flows from the financial asset expire or it transfers all contractual
rights of the financial asset.
Financial
Liabilities
At
initial recognition, the Group measures a financial liability at its fair value plus, in the case of financial liability not at
fair value through profit or loss, transaction costs that are directly attributable to the acquisition of the financial liability.
Transaction costs of financial liabilities carried at fair value through profit or loss are expensed in profit or loss.
Financial
liabilities are generally classified at amortized cost. There are some exceptions, for example, financial liabilities at fair
value through profit or loss including derivatives not designated as hedging instruments.
Financial
liabilities are analyzed to determine whether they contain any embedded derivatives. Embedded derivatives not closely related
to the host contract will be separated and accounted for separately at FVTPL.
Financial
liabilities (or a part of a financial liability) are derecognized from the statement of financial position when, and only when,
it is extinguished, i.e. when the obligation specified in the contract is discharged or cancelled or expires.
Impairment
From
1 January 2018, the Group assesses on a forward looking basis the expected credit losses associated with its debt instruments
carried at amortized cost and at fair value through other comprehensive income or loss. The impairment methodology applied depends
on whether there has been a significant increase in credit risk. If, at the reporting date, the credit risk on a financial instrument
has not increased significantly since initial recognition, the Group measures the loss allowance for the financial instrument
at an amount equal to twelve-month expected losses. In case the credit risk on a financial instrument has increased significantly
since initial recognition, the Group measures the loss allowance for that financial instrument at an amount equal to the lifetime
expected credit losses. To assess whether there is a significant increase in credit risk Curetis compares the risk of a default
occurring on the asset as at the reporting date with the risk of default as at the date of initial recognition. It considers available
reasonable and supportive forward-looking information. Especially the following indicators are incorporated:
|
|
●
|
external
credit rating (as far as available)
|
|
|
●
|
actual
or expected significant adverse changes in business, financial or economic conditions
that are expected to cause a significant change to the borrower’s ability to meet
its obligations
|
|
|
●
|
significant
increases in credit risk on other financial instruments of the same borrower
|
|
|
●
|
significant
changes in the expected performance and behavior of the borrower, including changes in
the payment status of borrowers in the group and changes in the operating results of
the borrower.
|
Regardless
of the analysis above, a significant increase in credit risk is presumed if a debtor is more than 30 days past due in making a
contractual payment.
Deposits
with banks and financial institutions are considered to have low credit risk as of the reporting date as the relevant counterparties
have investment grade ratings. However, in case of an objective evidence of an impairment, Curetis analyses the respective financial
asset on an individual basis and recognizes an impairment in an amount of the lifetime expected credit losses. Impairment losses
are incurred if, and only if, there is objective evidence of impairment as a result of one or more events that occurred after
the initial recognition of the asset (an incurred “loss event”) and that loss event has an impact on the estimated
future cash flows of the financial asset that can be reliably estimated. Evidence of impairment may include indication that the
debtors or a Group of debtors is experiencing significant financial difficulty, default or delinquency in interest or principal
payments, the probability that they will enter bankruptcy or other financial reorganization and observable data indicating that
there is a measurable decrease in the estimated future cash flows, such as changes in arrears or economic conditions that correlate
with defaults. Regardless of the analysis before, a default on a financial asset is presumed to occur when the counterparty fails
to make contractual payments within 90 days of when they fall due.
For
accounts receivables, Curetis applies the simplified approach permitted by IFRS 9, which requires expected lifetime losses to
be recognized from initial recognition of the receivables. To measure the expected credit losses, all accounts receivables have
been grouped together as they share the same credit risk characteristics. A historic corporate default rate specific to the healthcare
industry adjusted for forward-looking macroeconomic factors and an appropriate recovery rate were applied to calculate the expected
credit losses. During the reporting period, there were no significant changes with regard to the calculation approach or applied
assumptions.
Accounts
receivables are written off when there is no reasonable expectation of recovery. One indicator that there is no reasonable expectation
of recovery include, amongst others, when internal or external information indicate that the Group is unlikely to receive the
outstanding contractual amount in full. Another indicator that there is no reasonable expectation of recovery is a durable failure
of the counterparty to meet its contractual obligations.
Offsetting
financial assets and financial liabilities
Curetis
currently has not recognized any financial instruments that are offset. The Group did not enter into any enforceable netting arrangements
or other derivative instruments or offsetting arrangements that meet the offsetting criteria in IAS 32.
Cash
and Cash equivalents
Cash
and cash equivalents comprise cash on hand, deposits held at call with banks, and other short-term highly liquid investments with
original maturities of three months or less.
Trade
receivables
Trade
receivables are amounts due from customers for merchandise sold or services performed in the ordinary course of business. A specific
valuation adjustment is established, when there is objective evidence that Curetis will not be able to collect all amounts due,
according to the original terms of the receivables. If collection is expected in one year or less, they are classified as current
assets. If not, they are presented as non-current assets.
Trade
and other payables
These
amounts represent liabilities for goods and services provided to the Group prior to the end of financial year which remain are
unpaid as of period end. The amounts are unsecured and are usually paid within 30 days of recognition. Trade and other payables
are presented as current liabilities unless payment is not due within 12 months after the reporting period. They are recognized
initially at their fair value and subsequently measured at amortized cost using the effective interest method.
3.17.
Provisions for other liabilities and charges
Provisions
are recognized when Curetis has a present legal or factual obligation as a result of past events; and it is probable that an outflow
of resources will be required to settle the obligation and the amount can be reliably estimated. Where the effect of the time
value of money is material, provisions are discounted using a current pre-tax rate. If discounting is used, the increase in the
provision over time is recognized as interest expense. Gains from the reversal of other current liabilities that arose originally
in previous years are recognized as other operating income.
3.18.
Current and deferred tax income
The
tax expense for the period comprises current and deferred tax. Tax is recognized in the statement of operations and other comprehensive
income or loss.
The
current income tax charge is calculated on the basis of the tax law enacted or substantively enacted at the balance sheet date
where the Company operates and generates taxable income. Management periodically evaluates positions taken in tax returns with
respect to situations in which applicable tax regulation is subject to interpretation. It establishes provisions where appropriate
on the basis of amounts expected to be paid to the tax authorities.
Deferred
income tax is recognized on temporary differences arising between the tax bases of assets and liabilities and their carrying amounts
in the financial statements, as well as for tax loss carryforward. However, deferred tax liabilities are not recognized if they
arise from the initial recognition of goodwill. In addition, deferred income tax is not accounted for if it arises from initial
recognition of an asset or liability in a transaction other than a business combination that at the time of the transaction affects
neither accounting nor taxable profit nor loss. Deferred income tax is determined applying tax rates (and laws) that have been
enacted or substantively enacted by the balance sheet date and are expected to apply when the related deferred income tax asset
is realized or the deferred income tax liability is settled.
Deferred
income tax assets are recognized only to the extent that it is probable that future taxable profit will be available against which
the temporary differences can be utilized. Deferred tax assets are only considered in the financial statements to offset deferred
tax liabilities. The Company recognizes deferred tax assets on unused losses only if it is probable that the related tax benefit
will be realized short-term.
Deferred
income tax assets and liabilities are offset when there is a legally enforceable right to offset current tax assets against current
tax liabilities and when the deferred income taxes assets and liabilities relate to income taxes levied by the same taxation authority
on either the same taxable entity or different taxable entities where there is an intention to settle the balances on a net basis.
In
accordance with IAS 1 ‘Presentation of financial statements’, the current part of deferred taxes is recognized as
non-current assets/ liabilities in the statement of financial position.
3.19.
Equity
An
equity instrument is any contract that evidences a residual interest in the assets of an entity after deducting all of its liabilities.
Equity instruments issued by the Company are recognized at the proceeds received, net of direct issue costs. Capital reserves
within Equity includes capital increases from Curetis N.V. to fund the operations of the business; shareholder contributions related
to costs incurred by Curetis N.V. primarily related to the compensation of certain members of senior management and its supervisory
board for the benefit of the business, which have historically been incurred by Curetis N.V. but have not been recharged by Curetis
N.V. to Curetis GmbH or its subsidiaries; and share-based payment transactions with Curetis GmbH employees that are based on Curetis
N.V. shares. In both the case of shareholder contributions and share-based payments, the cost for these items have been recognized
as expenses in the statement of operations and comprehensive loss.
3.20.
Share-based payments
The
Employee Stock Option Plan 2016 (“ESOP”)
In
July 2016, Curetis N.V began to grant stock options according to the Employee Stock Option Plan 2016. The terms of this ESOP were
adopted by the general meeting on 16 June 2016. The stock option plan was designed in order to grant options to ordinary shares
in the capital of Curetis N.V. to nominees. The purpose of the plan is the retention of current and the recruiting of new key
employees, managing directors and supervisory directors, to spare liquidity, diminish employee turnover, alignment of shareholders
́ interests with employees´ and directors´ interests and finally to increase interest of capital markets in the
Curetis N.V. by a shareholder value orientated compensation system.
The
fair value of the stock options were measured by using a binomial option pricing model taking into account the terms and conditions
upon which the options were granted.
The
expense resulting from the share-based payment transactions is recognized by the Group during the vesting period with a corresponding
increase to the capital reserve. Furthermore, the amount recognized is based on the best available estimate of the number of equity
instruments expected to vest and is revised, if subsequent information indicates that the number of equity instruments expected
to vest differs from previous estimates.
Valuation
model, input parameters, recognized expenses and further details are stated in Note 22.
3.21.
Use of assumptions and estimates
The
preparation of financial statements in conformity with IFRS requires management to make estimates and assumptions that affect
the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial
statements and the reported amount of income and expenses during the period. Actual results could differ from those estimates.
Significant
areas requiring the use of management estimates relate to determination of the useful lives of property, plant and equipment,
inventories valuation, provisions, discounted cash flows for impairment testing, and recognition of deferred tax assets.
The
determination of the useful economic life for intangible assets and property, plant and equipment of Curetis is subject to the
estimates made by the management.
Inventories
are valued at the lower value cost and net realizable value. The net realizable value is determined by the estimated selling price
in the ordinary course of business less the incurred plus estimated costs of completion and the estimated costs necessary to sale
the end product. The assessment of the obsolescence write-downs on inventories is considered a significant estimate with inherent
uncertainty. Given Curetis does not yet have a reliable sales-track-record; the write-downs are based on the best estimate considering
technical aging and estimated sales volumes and prices for systems.
When
accounting for provisions, management must make assumptions regarding the estimated amounts, timing, and probability of economic
outflows which form the basis for the measurement of provisions.
To
evaluate the recoverability of intangible assets with indefinite useful lives, the Company compares the carrying values of the
asset to the asset's fair value, determined using a discounted cash flow approach or other methods, if appropriate. In determining
the discounted cash flows, management must estimate future revenues and weighted average cost of capital (“WACC”),
both of which are considered significant assumptions.
The
calculation of deferred tax assets requires assumptions to be made with regard to the level of future taxable income and the timing
of recovery of deferred tax assets. These assumptions take account of forecasting operating results and the impact on earnings
of the reversal of taxable temporary differences. Since future business developments cannot be predicted with certainty and to
some extent cannot be influenced by Curetis, the measurement of deferred tax assets is subject to risk and uncertainty.
In
accordance with IFRS 2 – Share based Payment, the fair value of the options at grant date is recognized as an expense
in the statement of operations and other comprehensive loss over the vesting period of delivery of work. Subsequently, the fair
value of equity-settled stock options is not re-measured. The fair value of each option granted during the year is calculated
using the binominal valuation model. This valuation model requires the input of subjective assumptions, which are detailed in
note 22.
3.22.
Going concern
Since
inception, the Company’s activities have consisted primarily of performing research and development to advance its technologies
and more recently, establishing sales and distribution networks to commercialize its technology. Through 31 December 2018, the
Company has not yet established a stable ongoing source of revenues sufficient to cover its operating costs and has funded its
operations through proceeds from equity investments, collaboration and licensing agreements, grants and borrowings under various
agreements with funding agencies, and contributions from Curetis N.V., the ultimate holding company of Curetis GmbH as of 31 December
2017 and 2018, from the sale of Curetis N.V. stock in an Initial Public Offering, secondary offerings and various other financing
agreements. Since inception, the Company has incurred recurring losses (with the exception of 2015 due to an extraordinary gain),
including net losses of EUR 18.3 million and EUR 22.8 million for the years ended 31 December 2017 and 2018, respectively. As
of 31 December 2018, the Company had an accumulated deficit of EUR 160.4 million, EUR 4.8 million in cash and cash equivalents,
trade receivables of EUR 0.3 million, and EUR 0.5 million VAT refund receivable shown within Group Receivables.
The
Company also realized the following inflows of funds from financing during 2019.
|
|
●
|
EIB
Debt Financing Facility has funded the EUR 5 million milestone tranche in June 2019,
however, Curetis believes this was the last of the debt financing tranches that Curetis
could or would access under the current EIB facility.
|
|
|
●
|
Yorkville
Convertible Note facility withdrawal of EUR 3.5 million.
|
Despite
the cost reduction measures already implemented in Europe and the USA, the Company expects to continue to generate operating losses
in the foreseeable future, and the existing current assets, including cash, as well as the aforementioned secured external funding
sources are not sufficient to finance Curetis’ operating activities for said 12 months after the signing date of these financial
statements. Substantial doubt regarding the Group’s ability to continue as a going concern exists as of 15 September 2019,
the issuance date of these combined financial statements.
The
Group’s Management believes that if it can realize cash-inflow and funding measures, execute on strategy options, realize
liquidity planning and implement these planned measures as needed, funding of our business operations for a period of at least
12 months after the issue date of these financial statements is achievable. Curetis is in the process of evaluating and progressing
strategic and liquidity planning options to be able to raise additional capital and reduce costs, including:
|
|
●
|
The
negotiation and implementation of a strategic option and scenario that, if successful,
would allow Curetis to access the capital markets and raise additional capital again.
|
|
|
●
|
Curetis
aims at accessing cash relating to entering into one or more licensing and partnering
deal(s) around its Unyvero A30 RQ platform and Aresdb. A draft term sheet has
been received for Unyvero A30 RQ and is currently under negotiation; however,
none are currently committed or secured.
|
|
|
●
|
Potentially
putting on hold, delaying, or reducing further expenditures for certain R&D, commercialization
and operational programs.
|
The
Company has also engaged financial and other advisors to assist it in those efforts. The Company will seek additional funding
and to execute on these strategic business and commercial plans in order to reach its development and commercialization objectives.
There are no assurances the Company will be able to obtain financing on acceptable or favorable terms, or at all, and the Company
may not be able to execute on strategic business and commercial plans or to enter into collaborations or other arrangements. The
Company is primarily dependent on its parent, Curetis N.V., for financing. Further, Curetis N.V. is not an operational entity
which generates cash inflows, rather, is reliant on its shareholders and other external financing to remain funded. In the event
the Company is unable to successfully raise additional capital during or before the fourth quarter of 2019, the Company will not
have sufficient cash flows and liquidity to finance its business operations as currently contemplated. Accordingly, in such circumstances
the Company would be compelled to immediately and significantly reduce general and administrative expenses, delay research and
development projects, and product portfolio expansion or commercialization efforts until it is able to obtain sufficient financing,
which could adversely affect its business prospects. If such sufficient financing is not received on a timely basis, the Company
would then need to pursue a plan to license or sell its assets, seek to be acquired by another entity, cease operations and/or
seek bankruptcy protection.
The
accompanying combined financial statements have been prepared assuming that the Company will continue as a going concern, which
contemplates the realization of assets and the settlement of liabilities and commitments in the normal course of business. The
combined financial statements do not reflect any adjustments relating to the recoverability and classification of assets or the
amounts and classification of liabilities that might be necessary if the Company is unable to continue as a going concern.
3.23.
Government grants
Government
grants are not recognized until there is reasonable assurance that the Company will comply with the conditions attached to them
and that the grants will be received. The Group receives grants related to research projects from governmental agencies, these
are recognized at their fair value when the Group receives grants from the agency and will comply with the conditions attached
to the grants, but in no event prior to the formal grant approval. The grants are accounted for as other operating income in the
statement of operations and other comprehensive loss. When the grant relates to an expense item, it is recognized as income on
a systematic basis over the periods that the related costs, for which it is intended to compensate, are expensed.
|
in
kEUR
|
|
2018
|
|
2017
|
|
Sale
of Unyvero-Systems
|
|
|
546
|
|
|
|
448
|
|
|
Sale of cartridges
|
|
|
811
|
|
|
|
722
|
|
|
Sale
of services
|
|
|
62
|
|
|
|
17
|
|
|
Total
|
|
|
1,419
|
|
|
|
1,187
|
|
In
accordance with IFRS 8, Curetis is a single-segment entity. Revenues from external customers by territory, based on the destination
of the customers are as follows:
|
in
kEUR
|
|
2018
|
|
2017
|
|
EMEA
direct markets
|
|
|
676
|
|
|
|
274
|
|
|
USA
|
|
|
32
|
|
|
|
7
|
|
|
Asia
|
|
|
286
|
|
|
|
269
|
|
|
Rest
of the world
|
|
|
425
|
|
|
|
637
|
|
|
Total
|
|
|
1,419
|
|
|
|
1,187
|
|
All
revenues are derived from external customers, including hospitals as well as distribution partners.
|
in
kEUR
|
|
2018
|
|
2017
|
|
Personnel
expenses
|
|
|
10,780
|
|
|
|
8,614
|
|
|
Depreciation,
amortization and impairment charges
|
|
|
1,256
|
|
|
|
1,327
|
|
|
Raw
material, goods and consumables used
|
|
|
848
|
|
|
|
610
|
|
|
Facility
expenses
|
|
|
777
|
|
|
|
519
|
|
|
Disposables
for clinical trials and R&D-activities
|
|
|
832
|
|
|
|
751
|
|
|
Third
party services for clinical trials incl. US-FDA-trial
|
|
|
331
|
|
|
|
377
|
|
|
Marketing
and travel expenses
|
|
|
1,659
|
|
|
|
1,392
|
|
|
Other
consulting, advisory & third party support
|
|
|
3,276
|
|
|
|
1,780
|
|
|
Other
expenses
|
|
|
3,894
|
|
|
|
3,677
|
|
|
Total
Cost of sales, Distribution costs, Administrative expenses and Research & development expenses
|
|
|
23,653
|
|
|
|
19,047
|
|
The
Personnel expenses in 2018 include kEUR 366 (2017: kEUR 528) expenses recognized for the valuation of equity-settled share-based
payment transactions. Refer to Note 22 for additional information.
|
in
kEUR
|
|
2018
|
|
2017
|
|
Personnel
expenses
|
|
|
5,016
|
|
|
|
4,155
|
|
|
thereof
from share-based payments equity-settled
|
|
|
113
|
|
|
|
370
|
|
|
Depreciation
and Amortization
|
|
|
74
|
|
|
|
170
|
|
|
Other
operating expenses
|
|
|
3,057
|
|
|
|
2,881
|
|
|
thereof
marketing expenses
|
|
|
1,410
|
|
|
|
1,138
|
|
|
thereof
travel expenses
|
|
|
746
|
|
|
|
520
|
|
|
thereof
consulting, advisory & third party service
|
|
|
255
|
|
|
|
412
|
|
|
Total
|
|
|
8,147
|
|
|
|
7,206
|
|
Distribution
costs include all direct individual sales and marketing costs as well as overhead costs. These include all expenses for sales,
marketing, public relations, and business development such as personnel, materials, depreciation, and other related expenditures.
|
|
7.
|
ADMINISTRATIVE
EXPENSES
|
|
in
kEUR
|
|
2018
|
|
2017
|
|
Personnel
expenses
|
|
|
1,264
|
|
|
|
1,117
|
|
|
thereof
from share-based payments equity-settled
|
|
|
111
|
|
|
|
116
|
|
|
Depreciation
and Amortization
|
|
|
90
|
|
|
|
104
|
|
|
Other
expenses
|
|
|
2,224
|
|
|
|
1,964
|
|
|
thereof
for remuneration of supervisory board
|
|
|
409
|
|
|
|
359
|
|
|
thereof
consulting, advisory & third party service
|
|
|
500
|
|
|
|
291
|
|
|
Total
|
|
|
3,578
|
|
|
|
3,185
|
|
Administrative
expenses include personnel, depreciation and other costs of the central administrative areas, which are not related to production,
sales or research and development.
|
|
8.
|
RESEARCH
AND DEVELOPMENT EXPENSES
|
|
in kEUR
|
|
2018
|
|
2017
|
|
Personnel
expenses
|
|
|
4,182
|
|
|
|
3,124
|
|
|
thereof
from share-based payments equity-settled
|
|
|
141
|
|
|
|
32
|
|
|
Depreciation
and Amortization
|
|
|
861
|
|
|
|
810
|
|
|
Material
expenses
|
|
|
513
|
|
|
|
407
|
|
|
Other
expenses
|
|
|
5,010
|
|
|
|
2,994
|
|
|
thereof
IP-fees and expenses for patent lawyers
|
|
|
576
|
|
|
|
—
|
|
|
thereof
external services for clinical trial
|
|
|
351
|
|
|
|
367
|
|
|
thereof
costs for laboratory demand
|
|
|
473
|
|
|
|
303
|
|
|
thereof
consulting, advisory & third party service
|
|
|
1,999
|
|
|
|
—
|
|
|
thereof
other manufacturing expenses for cartridges used in R&D
|
|
|
736
|
|
|
|
284
|
|
|
Total
|
|
|
10,566
|
|
|
|
7,335
|
|
|
in kEUR
|
|
2018
|
|
2017
|
|
Wages
and salaries
|
|
|
8,715
|
|
|
|
6,870
|
|
|
Social
security costs
|
|
|
1,699
|
|
|
|
1,216
|
|
|
EPOSs
/ PSOs granted to management and employees
|
|
|
366
|
|
|
|
528
|
|
|
Total
|
|
|
10,780
|
|
|
|
8,614
|
|
The
employer´s contribution paid to the statutory retirement insurance (Deutsche Rentenversicherung) in Germany amounted
to kEUR 409 in 2018 (2017: kEUR 374).
|
|
10.
|
FINANCE
RESULT / COSTS NET
|
Finance
result, net loss of kEUR 1,167 (2017: loss of kEUR 686) is primarily from interest on the 13 million Euro tranche drawn from the
EIB debt facility and foreign currency exchange difference resulting from the exchange rate difference of USD vs. EUR.
|
in kEUR
|
|
2018
|
|
2017
|
|
Foreign
exchange differences
|
|
|
6
|
|
|
|
(69
|
)
|
|
Interests
for borrowings
|
|
|
(1,079
|
)
|
|
|
(621
|
)
|
|
Interest
and finance expenses for convertible notes
|
|
|
(93
|
)
|
|
|
—
|
|
|
Other
finance income / finance costs
|
|
|
(1
|
)
|
|
|
4
|
|
|
Finance
result/costs net
|
|
|
(1,167
|
)
|
|
|
(686
|
)
|
Interests
for borrowings represent interest and financing charges paid/payable for financial liabilities not at fair value through profit
or loss using the effective interest method.
Income
tax expense:
|
in kEUR
|
|
2018
|
|
2017
|
|
Current
Income taxes
|
|
|
|
|
|
|
|
|
|
Germany
|
|
|
—
|
|
|
|
—
|
|
|
other
countries
|
|
|
29
|
|
|
|
24
|
|
|
Total
current income taxes
|
|
|
29
|
|
|
|
24
|
|
|
Deferred
taxes
|
|
|
7
|
|
|
|
(78
|
)
|
|
Total
|
|
|
36
|
|
|
|
(54
|
)
|
In
Germany, Income tax consists of trade tax (´Gewerbesteuer´) and corporate income tax (´Körperschaftsteuer´).
Corporate income tax is imposed at a uniform rate of 15% and is additionally subject to a solidarity surcharge of 5.5%, resulting
in an effective tax rate of 15.825% (2017: 15.825%).
Municipalities
impose a trade tax. Each municipality set its individual local multiplier rate, so that no uniform trade tax rate exists in Germany.
In 2018, Curetis has a trade tax rate of 12.05% (2017: 12.05%).
The
Company is fully taxable in Germany with the business seat in Holzgerlingen, Germany.
The
income tax expense for the year can be reconciled to the accounting profit (loss) as follows:
|
in kEUR
|
|
2018
|
|
2017
|
|
Loss
before income tax
|
|
|
(22,776
|
)
|
|
|
(18,374
|
)
|
|
Expected
income tax at a tax rate 2018: 27.88% (2017: 27.88%)
|
|
|
6,350
|
|
|
|
5,123
|
|
|
Non-taxable
income and non-deductible expenses
|
|
|
(34
|
)
|
|
|
(37
|
)
|
|
Expenses
resulting from Equity settled stock options
|
|
|
(138
|
)
|
|
|
(232
|
)
|
|
Changes
in the recognition of deferred tax assets on tax loss carry-forwards
|
|
|
(4,087
|
)
|
|
|
(3,803
|
)
|
|
Effect
from revaluation of DTA (in context with DTL)
|
|
|
74
|
|
|
|
(79
|
)
|
|
Tax
effect from local taxes
|
|
|
(33
|
)
|
|
|
(20
|
)
|
|
Tax
effect of the application of foreign tax rates and use of foreign tax losses carried forward
|
|
|
(2,128
|
)
|
|
|
(927
|
)
|
|
Other
effects
|
|
|
(40
|
)
|
|
|
29
|
|
|
Income
tax as stated in P&L
|
|
|
(36
|
)
|
|
|
54
|
|
|
Effective
tax rate
|
|
|
0
|
%
|
|
|
0
|
%
|
Changes
in the recognition of deferred tax assets on tax loss carry-forwards of kEUR 4,087 in Germany are due to unrecognized deferred
tax assets on tax loss carryforwards for 2018.
Tax
effects of the application of foreign tax rates and use of foreign tax losses carried forward comprise mainly to unrecognized
deferred tax assets for the loss of Curetis USA Inc. as there is no reliable certainty that these losses will be usable.
Deferred
tax assets and liabilities:
|
|
|
31
December 2018
|
|
31
December 2017
|
|
1
January 2017
|
|
in
kEUR
|
|
Total
|
|
thereof
|
|
Total
|
|
thereof
|
|
Total
|
|
thereof
|
|
|
|
|
|
current
|
|
|
|
current
|
|
|
|
current
|
|
DTA
|
|
|
426
|
|
|
|
30
|
|
|
|
430
|
|
|
|
104
|
|
|
|
430
|
|
|
|
61
|
|
|
DTL
|
|
|
426
|
|
|
|
93
|
|
|
|
430
|
|
|
|
73
|
|
|
|
430
|
|
|
|
61
|
|
Deferred
taxes relate to the following statement of financial position items:
|
in
kEUR
|
|
Deferred
tax assets
|
|
Deferred
tax liabilities
|
|
|
|
31
December 2018
|
|
31
December 2017
|
|
1
January 2017
|
|
31
December 2018
|
|
31
December 2017
|
|
1
January 2017
|
|
Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Trade
and other receivables
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Inventories
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
93
|
|
|
|
73
|
|
|
|
61
|
|
|
Property,
plant and equipment
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
280
|
|
|
|
357
|
|
|
|
369
|
|
|
Receivables
unrealized currency differences
|
|
|
30
|
|
|
|
104
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Financial
liabilities
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Provisions
current
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other
current liabilities
|
|
|
15
|
|
|
|
16
|
|
|
|
8
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other
current financial liabilities
|
|
|
—
|
|
|
|
—
|
|
|
|
33
|
|
|
|
53
|
|
|
|
—
|
|
|
|
—
|
|
|
Provisions
non-current
|
|
|
2
|
|
|
|
4
|
|
|
|
5
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other
non-current financial liabilities
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Equity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated
deficit
|
|
|
379
|
|
|
|
306
|
|
|
|
384
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deferred
taxes (gross)
|
|
|
426
|
|
|
|
430
|
|
|
|
430
|
|
|
|
426
|
|
|
|
430
|
|
|
|
430
|
|
|
Offsetting
|
|
|
426
|
|
|
|
430
|
|
|
|
430
|
|
|
|
426
|
|
|
|
430
|
|
|
|
430
|
|
|
Deferred
taxes (net)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
Deferred
tax assets for losses carried forward have been recognized in the amount of existing deferred tax liabilities. Due to the uncertainty
surrounding the Group´s ability to realize taxable profits in the near future, the Company did not recognize any further
deferred tax assets. Deferred tax assets shown under the non-current assets result from the elimination of intercompany profits.
Due
to differences in the valuation of the shares in Curetis GmbH (former AG) between IFRS and national (German) tax law. While the
valuation under IFRS is based on the net asset value of Curetis GmbH (former AG), the valuation under German tax law is based
on the taxable net book value. The resulting difference is however a permanent one which does not result in a deferred tax entry.
As
of 31 December 2018, Curetis had tax loss carryforwards that were not utilizable and for which no deferred taxes were recognized.
These tax loss carryforwards amount to kEUR 96,587 for corporate tax purposes and kEUR 96,098 for trade tax purposes (31 December
2017: kEUR 82,173 for corporate tax purposes and kEUR 81,957 for trade tax purposes). The aforementioned tax loss carryforwards
exist only in Germany hence they are only in Germany available unlimited for offsetting against future taxable profits of Curetis.
Deferred tax assets have not been recognized in respect of these losses as no sufficient certainty is given, whether mid-term
such tax loss carryforwards will enable Curetis to offset its future taxable profits.
Overview
of the Group´s tax loss carryforwards:
|
in
kEUR
|
|
Curetis
GmbH
|
|
|
|
|
|
31
December 2018
|
|
31
December 2017
|
|
1
January 2017
|
|
Tax
loss carryforwards corporate tax
|
|
|
96,587
|
|
|
|
82,173
|
|
|
|
68,377
|
|
|
Tax
loss carryforwards trade tax
|
|
|
96,098
|
|
|
|
81,957
|
|
|
|
68,328
|
|
|
Non-taxable
income and non-deductible expenses
|
|
|
(34
|
)
|
|
|
(37
|
)
|
|
|
(32
|
)
|
|
Expenses
resulting from Equity settled stock options
|
|
|
(138
|
)
|
|
|
(232
|
)
|
|
|
|
|
The
carrying amounts of the trade receivables approximate to their fair values. Current trade receivables are non-interest bearing.
|
in
kEUR
|
|
31
December
|
|
31
December
|
|
1
January
|
|
|
|
2018
|
|
2017
|
|
2017
|
|
|
|
|
|
|
|
|
|
Trade
receivables, gross
|
|
|
325
|
|
|
|
202
|
|
|
|
127
|
|
|
less
loss allowance
|
|
|
(2
|
)
|
|
|
(2
|
)
|
|
|
(26
|
)
|
|
Trade
receivables, net
|
|
|
323
|
|
|
|
200
|
|
|
|
101
|
|
The
aging of the gross trade receivables at the reporting date was as follows:
|
in kEUR
|
|
31
December
|
|
31
December
|
|
1
January
|
|
|
|
2018
|
|
2017
|
|
2017
|
|
Gross
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Amounts
not due
|
|
|
242
|
|
|
|
195
|
|
|
|
103
|
|
|
Past
due 0-30 days
|
|
|
60
|
|
|
|
4
|
|
|
|
8
|
|
|
Past
due 31-60 days
|
|
|
23
|
|
|
|
3
|
|
|
|
2
|
|
|
More
than 60 days
|
|
|
—
|
|
|
|
—
|
|
|
|
14
|
|
|
Total
|
|
|
325
|
|
|
|
202
|
|
|
|
127
|
|
The
Company did not have any material amounts of past due receivables as of December 31, 2017 and 2018. As of 31 December 2018, trade
receivables of kEUR 83 (31 December 2017 kEUR 7 and 1 January 2017 kEUR 24) were past due, however no significant impairments
were identified. The aging analysis of these trade receivables is as follows:
Movements
in the Company´s allowance on trade receivables are as follows:
|
in
kEUR
|
|
2018
|
|
2017
|
|
Balance
as of 1 January
|
|
|
(2
|
)
|
|
|
(26
|
)
|
|
Net
additions (-) / reversals (+)
|
|
|
—
|
|
|
|
(1
|
)
|
|
Write-offs
|
|
|
—
|
|
|
|
25
|
|
|
Balance
as of 31 December
|
|
|
(2
|
)
|
|
|
(2
|
)
|
|
in kEUR
|
|
31
December
|
|
31
December
|
|
1
January
|
|
|
|
2018
|
|
2017
|
|
2017
|
|
Raw
materials
|
|
|
838
|
|
|
|
875
|
|
|
|
898
|
|
|
Semi-finished
goods
|
|
|
61
|
|
|
|
46
|
|
|
|
61
|
|
|
Trade
goods
|
|
|
4,987
|
|
|
|
4,419
|
|
|
|
3,040
|
|
|
Finished
goods
|
|
|
65
|
|
|
|
47
|
|
|
|
63
|
|
|
Spare
parts
|
|
|
101
|
|
|
|
66
|
|
|
|
16
|
|
|
Total
inventories, net
|
|
|
6,052
|
|
|
|
5,453
|
|
|
|
4.078
|
|
Semi-finished
goods comprise not yet completely assembled or manufactured parts of our disposables, such as reagent containers, base plates,
PCR chambers, etc. Trade goods comprise Unyvero Systems-components.
As
outlined in note 3.21 the assessment of the obsolescence write-downs on inventories is considered a significant estimate with
inherent uncertainty. Given Curetis does not yet have a reliable sales-track-record; the write-downs are based on the best estimate
considering technical aging and estimated sales volumes and prices for systems. If assumptions regarding future sales prices,
volumes, useful life or end product market potentials are not appropriate, this may lead to a further need for write-off. A change
in the estimated sales price of +/- 10% would result in a decrease or increase of obsolescence write-downs of kEUR 280, respectively.
A change in the estimated useful life of five years of the Unyvero systems by +/- 1 year would result in an decrease or increase
of obsolescence write-downs kEUR 692, respectively.
The
change of write-off to net asset value of inventories recognized as an expense and included in ´Cost of Sales´ in
2018 amounted to kEUR 244 (2017: kEUR 192).
|
|
14.
|
PREPAID
EXPENSES AND OTHER CURRENT ASSETS
|
|
in kEUR
|
|
31
December
2018
|
|
31
December
2017
|
|
1
January
2017
|
|
Prepaid
Expenses
|
|
|
177
|
|
|
|
148
|
|
|
|
80
|
|
|
Other
current assets
|
|
|
83
|
|
|
|
111
|
|
|
|
147
|
|
|
Total
|
|
|
260
|
|
|
|
259
|
|
|
|
227
|
|
Prepaid
expenses and other current assets mainly include lease payments, travel expenses, insurance fees, tax refunds and receivables,
and conference and exhibition fees.
|
|
|
|
|
Licenses
|
|
Unyvero
A30
|
|
Advance
|
|
|
|
in
kEUR
|
|
Software
|
|
&
Patents
|
|
technology
|
|
payments
|
|
Total
|
|
Cost:
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as of 1 January 2017
|
|
|
574
|
|
|
|
2,484
|
|
|
|
5,000
|
|
|
|
—
|
|
|
|
8,058
|
|
|
Additions
|
|
|
83
|
|
|
|
—
|
|
|
|
—
|
|
|
|
27
|
|
|
|
110
|
|
|
Balance
as of 31 December 2017
|
|
|
657
|
|
|
|
2,484
|
|
|
|
5,000
|
|
|
|
27
|
|
|
|
8,168
|
|
|
Accumulated
amortizations:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as of 1 January 2017
|
|
|
(509
|
)
|
|
|
(29
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(538
|
)
|
|
Amortization
|
|
|
(53
|
)
|
|
|
(53
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(106
|
)
|
|
Balance
as of 31 December 2017
|
|
|
(562
|
)
|
|
|
(82
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(644
|
)
|
|
Carrying
value as of 31 December 2017
|
|
|
95
|
|
|
|
2,402
|
|
|
|
5,000
|
|
|
|
27
|
|
|
|
7,524
|
|
|
Cost:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as of 1 January 2018
|
|
|
657
|
|
|
|
2,484
|
|
|
|
5,000
|
|
|
|
27
|
|
|
|
8,168
|
|
|
Additions
|
|
|
34
|
|
|
|
1
|
|
|
|
—
|
|
|
|
84
|
|
|
|
119
|
|
|
Balance
as of 31 December 2018
|
|
|
691
|
|
|
|
2,485
|
|
|
|
5,000
|
|
|
|
111
|
|
|
|
8,287
|
|
|
Accumulated
amortizations:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as of 1 January 2018
|
|
|
(562
|
)
|
|
|
(82
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(644
|
)
|
|
Amortization
|
|
|
(76
|
)
|
|
|
(142
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(218
|
)
|
|
Balance
as of 31 December 2018
|
|
|
(638
|
)
|
|
|
(224
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(862
|
)
|
|
Carrying
value as of 31 December 2018
|
|
|
53
|
|
|
|
2,261
|
|
|
|
5,000
|
|
|
|
111
|
|
|
|
7,425
|
|
In
2018 amortization of kEUR 0 (2017: kEUR 0) is included in ´Cost of sales´, in Distribution costs kEUR 2 (2017: kEUR
17), in R&D costs kEUR 152 (2017: kEUR 60) and kEUR 10 (2017: kEUR 29) in Administrative expenses.
The
GEAR platform, held within the Licenses & Patents, was transferred from Curetis GmbH in Q4-2017 to the wholly owned subsidiary
Ares Genetics GmbH and continues under the name Aresdb. The platform had not been amortized from its acquisition in Q4-2016 until
the transfer to Ares Genetics GmbH as it had not been available to be used. Subsequent to the transfer, the platform has been
in commercial use and is being amortized according to the runtime of the main patent (17.8 years) and the remaining amortization
period of the GEAR platform as of 31 December 2018 is 16.6 years. Curetis continues to invest further in these assets.
Intangible
assets not yet available for use (Unyvero A30 RQ) must be tested for impairment at least annually. The acquired Gyronimo-asset
has meanwhile been renamed to Unyvero A30, and will be developed by Curetis into a partnering-ready asset. The platform is still
in a development phase and the development takes place by the same team that had developed and continues to maintain the Unyvero
A50-multiplex-platform. As the Unyvero A30 RQ is not yet fully developed and ready for sale, it has no defined residual
amortization period.
Intangible
assets are tested annually for impairment or more frequently if events or changes in circumstances indicate that they might be
impaired. The recoverable amount for the Licenses and patents, most significantly Ares platform, and for Unyvero A30 is defined
by assessing the separately identifiable cash inflows, which are largely independent of the cash inflows from other assets. For
2017 and 2018 there were no indicators of potential impairment as the recoverable amounts of all intangible assets exceeded their
carrying amount, hence no impairment losses have been recognized.
|
|
16.
|
PROPERTY,
PLANT AND EQUIPMENT
|
|
in
kEUR
|
|
Land
and buildings
|
|
Machines
and technical equipment
|
|
Other
tangible assets
|
|
Assets
under construction
|
|
Total
|
|
Cost:
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as of 1 January 2017
|
|
|
72
|
|
|
|
7,853
|
|
|
|
2,413
|
|
|
|
199
|
|
|
|
10,537
|
|
|
Additions
|
|
|
—
|
|
|
|
1
|
|
|
|
232
|
|
|
|
90
|
|
|
|
323
|
|
|
Disposals
|
|
|
—
|
|
|
|
(2
|
)
|
|
|
(9
|
)
|
|
|
—
|
|
|
|
(11
|
)
|
|
Balance
as of 31 December 2017
|
|
|
72
|
|
|
|
7,852
|
|
|
|
2,636
|
|
|
|
289
|
|
|
|
10,849
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated
depreciation:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as of 1 January 2017
|
|
|
(42
|
)
|
|
|
(4,409
|
)
|
|
|
(1,619
|
)
|
|
|
—
|
|
|
|
(6,070
|
)
|
|
Disposals
|
|
|
—
|
|
|
|
1
|
|
|
|
7
|
|
|
|
—
|
|
|
|
8
|
|
|
Depreciation
|
|
|
(7
|
)
|
|
|
(835
|
)
|
|
|
(379
|
)
|
|
|
—
|
|
|
|
(1,221
|
)
|
|
Balance
as of 31 December 2017
|
|
|
(49
|
)
|
|
|
(5,243
|
)
|
|
|
(1,991
|
)
|
|
|
—
|
|
|
|
(7,283
|
)
|
|
Carrying
amount as of 31 December 2017
|
|
|
23
|
|
|
|
2,609
|
|
|
|
645
|
|
|
|
289
|
|
|
|
3,566
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as of 1 January 2018
|
|
|
72
|
|
|
|
7,852
|
|
|
|
2,636
|
|
|
|
289
|
|
|
|
10,849
|
|
|
Additions
|
|
|
—
|
|
|
|
31
|
|
|
|
215
|
|
|
|
424
|
|
|
|
670
|
|
|
Disposals
|
|
|
—
|
|
|
|
—
|
|
|
|
(81
|
)
|
|
|
—
|
|
|
|
(81
|
)
|
|
Reclassifications
|
|
|
—
|
|
|
|
417
|
|
|
|
—
|
|
|
|
(417
|
)
|
|
|
—
|
|
|
Balance
as of 31 December 2018
|
|
|
72
|
|
|
|
8,300
|
|
|
|
2,770
|
|
|
|
296
|
|
|
|
11,438
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated
depreciation:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as of 1 January 2018
|
|
|
(49
|
)
|
|
|
(5,243
|
)
|
|
|
(1,991
|
)
|
|
|
—
|
|
|
|
(7,283
|
)
|
|
Disposals
|
|
|
—
|
|
|
|
—
|
|
|
|
80
|
|
|
|
—
|
|
|
|
80
|
|
|
Depreciation
|
|
|
(8
|
)
|
|
|
(701
|
)
|
|
|
(330
|
)
|
|
|
—
|
|
|
|
(1,039
|
)
|
|
Reclassifications
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Balance
as of 31 December 2018
|
|
|
(57
|
)
|
|
|
(5,944
|
)
|
|
|
(2,341
|
)
|
|
|
—
|
|
|
|
(8,242
|
)
|
|
Carrying
amount as of 31 December 2018
|
|
|
15
|
|
|
|
2,356
|
|
|
|
529
|
|
|
|
296
|
|
|
|
3,196
|
|
Other
tangible assets comprise office equipment and Unyvero-Platforms used for internal demands.
Curetis
did not own any of these assets under any lease programs in 2017 or 2018. All property, plant and equipment are free from any
rights held by third parties. For further details, please refer to note 24.
|
|
17.
|
PHANTOM
STOCK OPTION INCENTIVE PLAN
|
Prior
to the IPO of Curetis N.V. shares, a share-based compensation plan, Curetis AG Phantom Stock Option Incentive Plan 2010 (“PSOP”),
was implemented under which the Company received services from employees and freelancers who received Phantom Stock Options (“PSO”)
as consideration.
Subsequent
to the IPO in 2015, all remaining outstanding PSOPs were contractually tied to a payment claim to be settled in a fixed number
of shares (PSOP-Roll-Over Agreements), the value of which had previously been measured at fair value and was fully expensed and
recognized in equity prior to 2017. Furthermore, all rights remain valid indefinitely; therefore, there have been no changes in
valuation and no effect to be accounted for in the statement operations and other comprehensive loss in 2017 or 2018. No PSOPs
were exercised or forfeited during 2017 or 2018.
Under
the PSOP-Roll-Over Agreements the beneficiaries are entitled to receive 659,237 new shares in the parent company, Curetis N.V.
as of 31 December 2018.
|
|
18.
|
OTHER
CURRENT LIABILITIES
|
|
in
kEUR
|
|
31
December
2018
|
|
31
December
2017
|
|
1
January
2017
|
|
Accruals
for vacation
|
|
|
244
|
|
|
|
232
|
|
|
|
172
|
|
|
Accruals
for Employee Bonuses
|
|
|
10
|
|
|
|
220
|
|
|
|
196
|
|
|
Accrual
for Severance / Restructuring
|
|
|
136
|
|
|
|
—
|
|
|
|
—
|
|
|
Accruals
for audit and preparation of financial statements
|
|
|
46
|
|
|
|
46
|
|
|
|
57
|
|
|
Other
tax liabilities
|
|
|
148
|
|
|
|
124
|
|
|
|
84
|
|
|
Other
liabilities
|
|
|
297
|
|
|
|
216
|
|
|
|
174
|
|
|
Total
|
|
|
881
|
|
|
|
838
|
|
|
|
683
|
|
|
|
19.
|
OTHER
CURRENT FINANCIAL LIABILITIES
|
Other
current financial liabilities include liabilities for outstanding invoices and finance lease.
|
in
kEUR
|
|
31
December
2018
|
|
31
December
2017
|
|
1
January
2017
|
|
Liabilities
for outstanding invoices
|
|
|
245
|
|
|
|
195
|
|
|
|
350
|
|
|
Provision
for deferred interest (refer to note 20)
|
|
|
343
|
|
|
|
279
|
|
|
|
118
|
|
|
Convertible
notes
|
|
|
3,109
|
|
|
|
—
|
|
|
|
—
|
|
|
Total
|
|
|
3,697
|
|
|
|
474
|
|
|
|
468
|
|
Convertible
notes
Key
facts of the convertible note facility
On 02 October 2018, Curetis N.V.
established a convertible note facility with Yorkville Advisors (Yorkville), a US institutional investor, consisting of several
tranches. Under the first tranche, 500 notes are available for issuance, whereby each note has a nominal value of kEUR 10 and
a maturity of one year. As of 31 December 2018, the Company had issued 350 notes from the first tranche with an issuance date
of 02 October 2018. The notes were issued at an 8% discount, due to a 4% commitment fee and a 4% subscription fee. The Company
incurred kEUR 120 in issuance costs related to due diligence and legal fees.
The holders of the outstanding notes
have the right to convert the notes in exchange for shares of Curetis N.V. at any time. The number of shares to be issued upon
conversion of a note is determined by the nominal amount of the note divided by 93% of the last 10-day lowest VWAP (volume weighted
average price) of a common share of Curetis N.V. on the conversion date. As of 31 December 2018, 20 notes had been converted to
shares of Curetis N.V.
All of the notes issued as of 31
December 2018 have a maturity date of 02 October 2019. Under the terms of the notes, the Company has the right to extend the maturity
date (up to four times) by 12 months. When extending the maturity date, the Company must pay a 5% fee on the outstanding balance
as of the extension date. Alternatively, Curetis also has the option to redeem the notes in cash at the maturity date.
The conversion rights represent a
financial liability, because ultimate settlement of the note would be based on a variable number of shares in the event the rights
are exercised. As a result, the Company classifies the entire instrument as a liability.
The Company assumed an initial fair
value of the notes, based on the Company’s share price as of the issuance date divided by 93%. The Company accounts for
the notes payable using the effective interest method, using an effective interest rate of 8% and the initial loan term of 12
months. The Company accounts for the outstanding convertible notes as a current liability, as the likelihood for executing the
extension option is remote.
As the legal issuer and obligor of
these notes, Curetis N.V. contributed the original proceeds to Curetis GmbH. As a result, for purposes of the combined financial
statements, the liability is attributed to the Company as a reduction of capital contributions in equity. Subsequent interest
expenses allocated to the Company are reflected as an increase to the liability and interest expense.
|
|
20.
|
OTHER
NON-CURRENT FINANCIAL LIABILITIES
|
In
2016 Curetis entered into a contract for an up to EUR 25 million senior, unsecured loan financing facility from the EIB (European
Investment Bank). The financing in the first growth capital loan under the European Growth Finance Facility (EGFF), launched in
November 2016. It is backed by a guarantee from the European Fund for Strategic Investment (EFSI). EFSI is an essential pillar
of the Investment Plan for Europe (IPE), under which the EIB and the European Commission are working as strategic partners to
support investments and bring back jobs and growth to Europe.
The
funding can be drawn in up to five tranches within 36 months, under the EIB amendment, each tranche is to be repaid upon maturity
five years after draw-down.
In
April 2017 Curetis drew down a first tranche of EUR 10 million from this facility. This tranche has a floating interest rate of
EURIBOR + 4% p.a. payable after each 12-month-period from the draw-down-date and another additional 6% p.a. that is deferred and
payable at maturity together with the principal. In June 2018 another tranche of EUR 3 million was drawn down. The terms and conditions
are analogous to the first one.
Other
non-current financial liabilities comprise the EIB debt facility and the accrued interest, calculated with the effective interest
method. The effective interest rate applied by the Company is 9.12% for the EUR 10 million tranche and 9.16% for the EUR 3 million
tranche.
|
in kEUR
|
|
31 December
2018
|
|
31 December
2017
|
|
1 January
2017
|
|
|
|
current
|
|
non-current
|
|
current
|
|
non-current
|
|
current
|
|
non-current
|
|
Loan
from EIB
|
|
|
—
|
|
|
|
13,000
|
|
|
|
—
|
|
|
|
10,000
|
|
|
|
—
|
|
|
|
—
|
|
|
Deferred
interest
|
|
|
343
|
|
|
|
949
|
|
|
|
279
|
|
|
|
342
|
|
|
|
—
|
|
|
|
—
|
|
|
Total
|
|
|
343
|
|
|
|
13,949
|
|
|
|
279
|
|
|
|
10,342
|
|
|
|
—
|
|
|
|
—
|
|
|
|
21.
|
FINANCIAL
INSTRUMENTS
|
For
each class of financial instrument the fair value of financial assets and liabilities, together with their carrying amounts contained
in the combined financial statements are shown in the following schedules.
|
in kEUR
|
|
31
December 2018
|
|
31
December 2017
|
|
|
|
Category
in accordance with IFRS9
|
|
Carrying
amount
|
|
Fair
Value
|
|
Fair
Value Level
|
|
Category
in accordance with IAS 39
|
|
Carrying
amount
|
|
Fair
Value
|
|
Fair
Value Level
|
|
Current
Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash
and Cash Equivalents
|
|
AC
|
|
|
4,800
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
LaR
|
|
|
3,468
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
Trade
Receivables
|
|
AC
|
|
|
323
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
LaR
|
|
|
200
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
Non-current
Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other
non-current financial assets
|
|
AC
|
|
|
158
|
|
|
|
158
|
|
|
|
2
|
|
|
LaR
|
|
|
156
|
|
|
|
156
|
|
|
|
2
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(n/a
*): For short-term financial instruments a fair value disclosure is not required as the carrying amount approximates the fair
value.
|
in kEUR
|
|
31
December 2018
|
|
31
December 2017
|
|
|
|
Category
in accordance with IFRS9
|
|
Carrying
amount
|
|
Fair
Value
|
|
Fair
Value Level
|
|
Category
in accordance with IAS 39
|
|
Carrying
amount
|
|
Fair
Value
|
|
Fair
Value Level
|
|
Current
Liabilities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Trade
and other Payables
|
|
FLAC
|
|
|
921
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
|
FLAC
|
|
|
|
850
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
Other
current financial liabilities
|
|
FLAC
|
|
|
3,154
|
(1)
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
|
FLAC
|
|
|
|
474
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
Other
current financial liabilities
|
|
FVTPL
|
|
|
543
|
(2)
|
|
|
543
|
|
|
|
3
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Non-current
Liabilities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other
non-current financial liabilities
|
|
FLAC
|
|
|
13,949
|
|
|
|
13,546
|
|
|
|
2
|
|
|
|
FLAC
|
|
|
|
10,342
|
|
|
|
10,368
|
|
|
|
2
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(n/a
*): For short-term financial instruments a fair value disclosure is not required as the carrying amount approximates the fair
value.
(1) Consists of liabilities
for outstanding invoices, Convertible notes and provision for deferred interest
(2) Consists of conversion rights related to Convertible notes
|
in kEUR
|
|
1
January 2017
|
|
|
|
Category
in accordance with IAS 39
|
|
Carrying
amount
|
|
Fair
Value
|
|
Fair
Value Level
|
|
Current
Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash
and Cash Equivalents
|
|
LaR
|
|
|
6,434
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
Trade
Receivables
|
|
LaR
|
|
|
101
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
Non-current
Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other
non-current financial assets
|
|
LaR
|
|
|
326
|
|
|
|
326
|
|
|
|
2
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(n/a
*): For short-term financial instruments a fair value disclosure is not required as the carrying amount approximates the fair
value.
|
in kEUR
|
|
1
January 2017
|
|
|
|
Category
in accordance with IAS 39
|
|
Carrying
amount
|
|
Fair
Value
|
|
Fair
Value Level
|
|
Current
Liabilities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Trade
and other Payables
|
|
FLAC
|
|
|
672
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
Other
current financial liabilities
|
|
FLAC
|
|
|
468
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
Non-current
Liabilities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other
non-current financial liabilities
|
|
FLAC
|
|
|
—
|
|
|
|
—
|
|
|
|
n/a
|
|
(n/a
*): For short-term financial instruments a fair value disclosure is not required as the carrying amount approximates the fair
value.
The
fair value is the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between
market participants at the measurement date.
The
fair value hierarchy is defined as follows:
|
Level 1
|
|
Quoted
(unadjusted) market prices in active markets for identical assets and liabilities.
|
|
Level 2
|
|
Valuation
techniques for which the lowest level input that is significant to the fair value measurement
is directly or indirectly observable.
|
|
Level 3
|
|
Valuation
techniques for which the lowest level input that is significant to the fair value measurement
is unobservable.
|
The
fair values of the Group's non-current other financial assets and the non-current financial liabilities were calculated based
on cash flows discounted using market interest rates and a credit spread. The spread included in the calculation for the financial
assets is derived by observable ratings of the counterparties (i.e. banks). The credit spread of the own credit risk is derived
from the margin included in the interest rates of the own borrowings. The fair value of non-current financial assets and liabilities
is included in level 2 of the fair value hierarchy, as the input factors for the fair value calculation are observable in the
market. The fair value of the compound embedded derivative separated from the convertible note is determined using observable
inputs (Curetis N.V. share price, own credit spread) and assumptions about the rational economic behavior of the related parties
which are not observable input parameters. These assumptions lead to the inclusion of the fair value within level 3 of the fair
value hierarchy.
Secured
liabilities and assets pledged as security
Curetis
has pledged cash on bank accounts as rent deposit for lease agreements with a total value of kEUR 64 and for credit card deposits
and bank guarantees with a total value of kEUR 94.
The
Executives and Supervisory Board as well as certain employees of the Group are included in the Employee Stock Option Plan 2016
“ESOP” of Curetis N.V.. The expenses associated with these individuals are recognized by the Group, which employ and
benefit from the employment of the individuals holding notional stocks in Curetis N.V..
Capital
reserve increased correspondingly to the expenses accounted for the share-based payment of the ESOP 2016 (see note 3.20).
The
following table illustrates the number and exercise prices of the movements in employee stock options during the year, as well
as the grant date and the remaining term of the option (Note, valuation inputs; stock price, dividend yields, volatility etc.
are related to the stock of Curetis N.V.):
|
|
|
Tranche
1
|
|
Tranche
2
|
|
Tranche
3
|
|
Tranche
4
|
|
Tranche
5
|
|
Grant
date
|
|
1
July 2016
|
|
1
October 2016
|
|
1
January 2017
|
|
1
April 2017
|
|
1
July 2017
|
|
Granted
stock options
|
|
|
170,000
|
|
|
|
45,000
|
|
|
|
42,500
|
|
|
|
5,000
|
|
|
|
20,000
|
|
|
Remaining
contractual term of the option
|
|
|
7.50
years
|
|
|
|
7.75
years
|
|
|
|
8.00
years
|
|
|
|
8.25
years
|
|
|
|
8.50
years
|
|
|
Exercise
price
|
|
|
6.45
Euro
|
|
|
|
6.41
Euro
|
|
|
|
6.42
Euro
|
|
|
|
5.81
Euro
|
|
|
|
4.93
Euro
|
|
|
Outstanding
at 1 January 2018
|
|
|
155,000
|
|
|
|
25,000
|
|
|
|
42,500
|
|
|
|
5,000
|
|
|
|
20,000
|
|
|
Granted
during the year
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Forfeited
during the year
|
|
|
22,222
|
|
|
|
2,500
|
|
|
|
1,042
|
|
|
|
—
|
|
|
|
12,222
|
|
|
Exercised
during the year
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Outstanding
at 31 December 2018
|
|
|
132,778
|
|
|
|
22,500
|
|
|
|
41,458
|
|
|
|
5,000
|
|
|
|
7,778
|
|
|
Exercisable
at 31 December 2018
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
Tranche
6
|
|
|
|
Tranche
7
|
|
|
|
Tranche
8
|
|
|
|
Tranche
9
|
|
|
|
Tranche
10
|
|
|
Grant
date
|
|
|
1
October 2017
|
|
|
|
1
January 2018
|
|
|
|
1
March 2018
|
|
|
|
1
July 2018
|
|
|
|
1
October 2018
|
|
|
Granted
stock options
|
|
|
123,500
|
|
|
|
25,000
|
|
|
|
102,000
|
|
|
|
40,500
|
|
|
|
110,000
|
|
|
Remaining
contractual term of the option
|
|
|
8.75
years
|
|
|
|
9.00
years
|
|
|
|
9.17
years
|
|
|
|
9.50
years
|
|
|
|
9.75
years
|
|
|
Exercise
price
|
|
|
4.98
Euro
|
|
|
|
3.86
EUR
|
|
|
|
6.51
EUR
|
|
|
|
4.62
EUR
|
|
|
|
3.29
EUR
|
|
|
Outstanding
at 1 January 2018
|
|
|
123,000
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Granted
during the year
|
|
|
—
|
|
|
|
25,000
|
|
|
|
102,000
|
|
|
|
40,500
|
|
|
|
110,000
|
|
|
Forfeited
during the year
|
|
|
16,667
|
|
|
|
—
|
|
|
|
5,000
|
|
|
|
3,000
|
|
|
|
—
|
|
|
Exercised
during the year
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Outstanding
at 31 December 2018
|
|
|
106,833
|
|
|
|
25,000
|
|
|
|
97,000
|
|
|
|
37,500
|
|
|
|
110,000
|
|
|
Exercisable
at 31 December 2018
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
Vesting
conditions
Each
option grant will vest over a period of three years whereby the first third of any such option grant will vest at the first anniversary
of the date of grant and the remaining two thirds of such granted options will vest in monthly increments over the following twenty-four
months.
Upon
the occurrence of a termination of employment event after the first anniversary of the date of grant, the optionee´s options
shall either be forfeited, lapse or continue to be exercisable as set forth below:
|
|
●
|
In
case of termination for cause, both the options of such optionee that have vested (to
the extent not exercised) and the options of such optionee that have not yet vested shall
be forfeited at the date of termination for cause, unless agreed otherwise by the management
board (with regard to optionees being managing directors or supervisory directors);
|
|
|
●
|
In
case of a termination without cause, the options of such optionee that have vested (to
the extent not exercised) shall not be forfeited and the remaining part of the options
of such optionee that have not yet vested shall be forfeited at the date of termination
without cause.
|
Exercise
of options
Vested
options may not be exercised prior to the third anniversary of the date of grant and may be exercised until ten years from the
date of grant or such shorter period of time remaining under the stock options plan. Options which have not been exercised prior
to the end of the exercised period shall lapse automatically without any compensation whatsoever being due to the optionee. Exercises
of options are settled in Curetis N.V. shares.
Valuation
model and input parameters
The
fair value of the stock options is measured using a binominal option pricing model taking into account the terms and conditions
upon which the options were granted. The following table lists the inputs to the model used for the options granted in 2016, 2017
and 2018 at the measurement date:
|
|
|
Tranche
1
|
|
Tranche
2
|
|
Tranche
3
|
|
Tranche
4
|
|
Tranche
5
|
|
Measurement
date
|
|
5
July 2016 1
|
|
1
October 2016
|
|
1
January 2017
|
|
1
April 2017
|
|
1
July 2017
|
|
Expected
life of the option on the grant date (years)
|
|
|
5.0
|
|
|
|
5.0
|
|
|
|
5.0
|
|
|
|
5.0
|
|
|
|
5.0
|
|
|
Share
price on the measurement date (€)
|
|
|
6.44
|
|
|
|
6.18
|
|
|
|
6.34
|
|
|
|
5.69
|
|
|
|
4.74
|
|
|
Weighted avg. exercise
price
|
|
|
6.45
|
|
|
|
6.41
|
|
|
|
6.42
|
|
|
|
5.81
|
|
|
|
4.93
|
|
|
Expected
dividend yield (%)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Risk-free
interest rate (%)
|
|
|
(0.61
|
)
|
|
|
(0.61
|
)
|
|
|
(0.49
|
)
|
|
|
(0.40
|
)
|
|
|
(0.19
|
)
|
|
Expected volatility
of the share price (%)
|
|
|
78.15
|
|
|
|
81.36
|
|
|
|
60.90
|
|
|
|
57.99
|
|
|
|
55.75
|
|
|
Option value (€)
|
|
|
3.94
|
|
|
|
3.86
|
|
|
|
3.14
|
|
|
|
2.69
|
|
|
|
2.15
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Tranche
6
|
|
Tranche
7
|
|
Tranche
8
|
|
Tranche
9
|
|
Tranche
10
|
|
Measurement
date
|
|
1
October 2017
|
|
1
January 2018
|
|
1
March 2018
|
|
1
July 2018
|
|
1
October 2018
|
|
Expected
life of the option on the grant date (years)
|
|
|
5.0
|
|
|
|
5.0
|
|
|
|
5.0
|
|
|
|
5.0
|
|
|
|
5.0
|
|
|
Share
price on the measurement date (€)
|
|
|
4.86
|
|
|
|
3.83
|
|
|
|
6.20
|
|
|
|
4.17
|
|
|
|
3.24
|
|
|
Weighted avg. exercise
price
|
|
|
4.98
|
|
|
|
3.86
|
|
|
|
6.51
|
|
|
|
4.62
|
|
|
|
3.29
|
|
|
Expected
dividend yield (%)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Risk-free
interest rate (%)
|
|
|
(0.28
|
)
|
|
|
(0.15
|
)
|
|
|
(0.01
|
)
|
|
|
(0.28
|
)
|
|
|
(0.10
|
)
|
|
Expected volatility
of the share price (%)
|
|
|
55.55
|
|
|
|
65.33
|
|
|
|
65.63
|
|
|
|
62.42
|
|
|
|
62.01
|
|
|
Option value (€)
|
|
|
2.22
|
|
|
|
2.04
|
|
|
|
3.26
|
|
|
|
2.03
|
|
|
|
1.64
|
|
1
The measurement date represents the acceptance date of the options.
For
stock option valuation the possibility of early exercise was considered in the binomial model. Management determined an estimated
early exercise is expected five years after the date of grant of the options based on considered the following factors:
The
length of the vesting period has been considered since the stock options cannot be exercised until the end of the 3-year vesting
period – i. e. the expected option life of 5 years is 2 years after the first possible exercise date.
The
Company does not have historical data points or experience from past option programs and to date no options have been exercised,
however, due to normal fluctuation as well as fluctuations triggered by the recent re-organization there have been multiple cases
of forfeited options. As a result, the Company does not have any actual data available regarding the average length of time that
similar options have remained outstanding in the past or if the employee´s level within the Company will impact the timing
of exercise.
The
risk-free interest rate is the implied yield currently available on German government issued bonds with a remaining term equal
to the term of the options.
The
future volatility for the lives of the options was estimated based on historical volatility of peer group companies.
The
expense recognized during 2017 and 2018 is shown in the following table:
|
in
kEUR
|
|
2018
|
|
2017
|
|
Expense
arising from equity-settled share-based payment transactions
|
|
|
|
|
|
|
|
|
|
Cost
of sales
|
|
|
—
|
|
|
|
11
|
|
|
Distribution
costs
|
|
|
113
|
|
|
|
370
|
|
|
Administrative
expenses
|
|
|
111
|
|
|
|
116
|
|
|
Research
& development expenses
|
|
|
141
|
|
|
|
32
|
|
|
Total
|
|
|
365
|
|
|
|
529
|
|
The
Group does not consider paying dividends as long as the result from operating activities in the combined statement of operations
and other comprehensive loss and the cash flows from operating activities are negative.
|
|
23.
|
FINANCIAL
RISK MANAGEMENT
|
23.1.
Financial risk factors
This
note explains the Group´s exposure to financial risks and how these risks could affect the Group´s future financial
performance. Current year profit and loss information has been included where relevant to add further context.
Curetis’
activities expose the Company to a variety of financial risks such as currency risks, fair value interest risks, cash flow risks,
interest rate risks and price risks. Curetis’ finance department has created controlling instruments and key metrics to
identify and evaluate such risks in close co-operation with the operating units.
a)
Market risk
Market
risk is the risk that the fair value or future cash flows of a financial instrument will fluctuate because of changes in market
prices. Curetis has a strong international business focus and therefore the Company is influenced by foreign currency exchange
rates and interest rates. However, Curetis currently does not hold any financial instruments measured at fair value and Curetis
keeps all its liquidity in immediately available money market funds.
aa)
Foreign exchange risk
Curetis
is exposed to foreign currency risks primarily through its operating activities. Curetis identifies the main currency risk in
US Dollar, because certain purchase transactions are undertaken in US Dollar (“USD”). The net exposure to exchange
differences of the monetary assets (being cash and cash equivalents) held in USD of the Group were kEUR 676 at 1 January 2017,
and kEUR 690 and kEUR 683 at 31 December 2017 and 2018, respectively.
If
the USD/EUR exchange rate were to increase/decrease by 10%, compared to year-end 2018 exchange rates, this would have a negative
impact of kEUR 62 (2017: kEUR 62) / positive impact of kEUR 76 (2017: kEUR 77). The Group considers a shift in the exchange rates
of 10% as a realistic scenario.
ab)
Interest rate risk
Curetis
is exposed to interest rate risk because entities in the Group borrow funds based a rate indexed to the EURIBOR, plus a fixed
rate of interest. The following sensitivity analysis is prepared assuming the amount of liability outstanding at reporting date
was outstanding for the whole year.
The
Group´s exposure to variable interest rates based on the EURIBOR at the end amounted to EUR 13 million as of 31 December
2018 (EUR 10 million as of 31 December 2017 and EUR 0 as of 1 January 2017).
If
the interest rates had been one per cent higher/lower and all other variables were held constant, the Group´s profit for
the year ended 31 December 2018 would decrease/increase by kEUR 130 (2017: decrease/increase by kEUR 100). This is mainly due
to the Group´s exposure to interest rates on its variable borrowings.
b)
Other market risk
Curetis
is not exposed to equity price risk or commodity price risk as it does not invest in these classes of investments.
c)
Credit risk
The
finance department works in close cooperation with the other operating departments to identify credit risks related to account
receivables balances. Curetis analyzes the credit risk of each new client before standard payment and delivery terms and conditions
are offered. Curetis has also implemented a well-organized dunning system. Curetis had immaterial write-downs on trade receivables
as of 1 January 2017 as well as during 2017 and 2018. The credit risk on the accounts receivables is limited because Curetis primarily
sells to big laboratories, pharma-companies and major public hospitals in Curetis’ direct markets in Central and Western
Europe and in the USA, all of these partners have very good credit ratings. Outside of Europe and the USA Curetis works together
with large and experienced distributors. If Curetis were to expand the business to other more credit-risky countries Curetis would
consider implementing a commercial credit insurance to cover the risks. Considering the aforementioned reasons Curetis summarizes
all trade receivables under one risk category ´common credit risk´ and impairs all trade receivables using an average
default risk of approximately 1% deducted from observable credit risk parameters of the healthcare industry. Curetis is in exchange
with different commercial credit insurers and is evaluation other credit risk mitigations periodically with the expansion of its
customer base.
In
2017 the following customer accounts each represented > 10% of total annual revenues: ATC Kuwait, Axonlab Austria, Synttergy
Consult Ltd. and in 2018 the following customers each represented > 10% of revenues: Diamed Care Germany, Axonlab Austria.
Similarly on the supplier side there is a significant concentration risk with single source suppliers of major strategic relevance
such as Zollner Elektronik for Unyvero systems, Scholz HTIK for injection molding plastics parts, as well as certain single source
suppliers of critical reagents.
Cash
and cash equivalents as well as short-term deposits which are disclosed under other financial assets are invested in EUR (with
the exemption of the amounts mentioned under aa) foreign exchange risk´ in this note. Curetis follows a decisive ´no-risk-policy´
which means that Curetis has sight deposits at banks only, and sometimes time deposits with short runtimes.
d)
Liquidity risk
Liquidity
risk is the risk that the Group will might encounter difficulties in meeting the obligations associated with its financial liabilities,
which are normally settled by delivering cash. The Group´s approach to managing liquidity is to ensure, as far as possible,
that it will always have sufficient liquidity to meet its liabilities when due.
The
Group monitors its risk of a shortage of funds using short and mid-term liquidity planning. This takes account of the expected
cash flows from all activities. The supervisory board undertakes regular reviews of the budget and forecast.
In
2018 Curetis drew down a EUR 3 million tranche from the up to EUR 25 million debt financing facility from the EIB (European Investment
Bank), in addition to the EUR 10 million already drawn down in 2017. Subsequent to obtaining an EIB waiver which waived the requirement
for the Group to meet various milestones in order to draw down additional tranches, another EUR 5 million became available for
disbursement immediately upon finalization of legal documentation for the amendment to the Finance Contract with EIB that sets
out the terms and conditions for the equity linked participation for EIB upon maturity of the EUR 5 million tranche in 2024 and
beyond. Curetis management currently believes that this EUR 5 million tranche would be the last of the debt financing tranches
that Curetis could or would access under the current EIB facility. As of year-end 2018 Curetis has cash & cash equivalents
balance of EUR 4.8 million at year-end 2018, and the EUR 0.4 million VAT refund.
Curetis'
future liquidity requirements will depend on many factors, some of which are beyond Curetis' control, including:
|
|
●
|
the
cost and timing of getting market traction in the U.S., as the USA are the most important
market for diagnostic products;
|
|
|
●
|
market
acceptance of Curetis' products;
|
|
|
●
|
the
cost and timing of establishing further distribution capabilities;
|
|
|
●
|
the
cost of Curetis' research and development activities;
|
|
|
●
|
the
ability of healthcare providers to obtain coverage and adequate reimbursement by third-party
payers for procedures using Curetis' products;
|
|
|
●
|
the
cost of goods associated with Curetis' products;
|
|
|
●
|
the
effect of competing technological and market developments; and
|
|
|
●
|
the
extent to which Curetis might decide to invest in third-party businesses, products and
technologies, including entering into licensing or collaboration arrangements for products.
|
If
Curetis were to miss its objectives or experienced material delays in one or more of these factors, additional funding would be
required which may or may not be available at all or might be available only at unfavorable terms and conditions.
The
following table depicts an analysis of the Company’s financial liabilities into relevant maturity groupings based on the
remaining term on the balance sheet date.
|
Balance
as at 31 December 2018
|
|
Up
to 1 year
|
|
1-3
years
|
|
3-5
years
|
|
More
than 5 years
|
|
Trade
and other payables
|
|
|
921
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other
financial liabilities
|
|
|
245
|
|
|
|
|
|
|
|
—
|
|
|
|
—
|
|
|
Loans
|
|
|
—
|
|
|
|
—
|
|
|
|
13,000
|
|
|
|
—
|
|
|
Convertible
note
|
|
|
3,300
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Interests
accrued
|
|
|
520
|
|
|
|
1,040
|
|
|
|
4,540
|
|
|
|
—
|
|
|
TOTAL
|
|
|
4,986
|
|
|
|
1,040
|
|
|
|
17,540
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as at 31 December 2017
|
|
|
Up
to 1 year
|
|
|
|
1-3
years
|
|
|
|
3-5
years
|
|
|
|
More
than 5 years
|
|
|
Trade
and other payables
|
|
|
850
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other
financial liabilities
|
|
|
195
|
|
|
|
|
|
|
|
—
|
|
|
|
—
|
|
|
Loans
|
|
|
—
|
|
|
|
—
|
|
|
|
10,000
|
|
|
|
—
|
|
|
Interests
accrued
|
|
|
400
|
|
|
|
800
|
|
|
|
3,800
|
|
|
|
—
|
|
|
TOTAL
|
|
|
1,445
|
|
|
|
800
|
|
|
|
13,800
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as at 1 January 2017
|
|
|
Up
to 1 year
|
|
|
|
1-3
years
|
|
|
|
3-5
years
|
|
|
|
More
than 5 years
|
|
|
Trade
and other payables
|
|
|
672
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Other
financial liabilities
|
|
|
350
|
|
|
|
|
|
|
|
—
|
|
|
|
—
|
|
|
Loans
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Interests
accrued
|
|
|
118
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
TOTAL
|
|
|
1,140
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
23.2.
Capital Management
Capital
comprises equity attributable to shareholders, cash and cash equivalents. Curetis´ policy is to maintain a strong base in
terms of equity capital and sufficient cash balance in order to maintain investor and creditors confidence and to sustain the
future development of the business. Our primary goals when managing capital are to ensure sufficient liquidity to meet our working
capital requirements, fund capital investments and purchases and to safeguard our ability to continue operating as a going concern.
See note 3.22 for further discussion of going concern.
Curetis
monitors all capital positions regularly (at least monthly) within its financial reporting, discusses the capital status frequently
within the management meetings and also within its supervisory board meetings.
|
|
24.
|
COMMITMENTS
AND CONTINGENCIES
|
Operating
lease and purchase commitments
Curetis
leases its offices, laboratories, and production facility under non-cancellable operating lease agreements. The lease term is
5 years and the agreements are renewable at the end of the lease term at market rate. The manufacturing facility in Bodelshausen
Curetis has a prolongation option.
Curetis
also leases machinery and vehicles under non-cancellable operating lease agreements. The lease term is 3 years and the agreements
are not renewable at the end of the lease term. The future aggregate minimum lease payments under non-cancellable operating leases
and existing purchase commitments are as per the table below.
|
in
kEUR
|
|
31
December
2018
|
|
31
December
2017
|
|
1
January
2017
|
|
From
lease contracts:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
No
later than 1 year
|
|
|
482
|
|
|
|
380
|
|
|
|
354
|
|
|
Later
than 1 year and no later than 5 years
|
|
|
819
|
|
|
|
629
|
|
|
|
321
|
|
|
Later
than 5 years
|
|
|
0
|
|
|
|
0
|
|
|
|
0
|
|
|
Total
from lease contracts
|
|
|
1,301
|
|
|
|
1,009
|
|
|
|
675
|
|
|
From
purchase and service agreements:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
No
later than 1 year
|
|
|
4,487
|
|
|
|
4,575
|
|
|
|
5,227
|
|
|
Later
than 1 year and no later than 5 years
|
|
|
4,331
|
|
|
|
2
|
|
|
|
813
|
|
|
Later
than 5 years
|
|
|
0
|
|
|
|
0
|
|
|
|
0
|
|
|
Total
from purchase and service agreements
|
|
|
8,818
|
|
|
|
4,577
|
|
|
|
6,040
|
|
|
Total
|
|
|
10,119
|
|
|
|
5,586
|
|
|
|
6,715
|
|
Curetis
places frame-work orders for Unyvero-Systems and for raw materials for its cartridge manufacturing to ensure availability during
commercial ramp-up-phase and also to gain volume-scale-effects with regards to purchase prices. Some of the electronic parts used
for the production of Unyvero-Systems have lead times of many months, hence it is necessary to order such systems with long-term
framework-orders to ensure the demands from market are covered.
The Company has reflected transactions
with the parent company, Curetis N.V., as related party balances within the statement of operations and other comprehensive loss
and the combined statement of financial position. Other related party transactions have been included below.
Curetis N.V. charges certain management fees for services rendered by the senior management of Curetis N.V. to Curetis GmbH and
its subsidiaries resulting in other expenses from related parties. The transactions are charged at cost. Curetis N.V. is the controlling
company for VAT purposes and receives VAT amounts due to Curetis GmbH as a controlled company, resulting in Other receivables,
related party.
Curetis has entered into arrangements
with a number of its subsidiaries, the financial impacts of which are eliminated in combination. Curetis considers transactions
with key management personnel to be related party transactions. Any transactions with such individuals are also recorded in related
party accounts.
During 2017 and 2018, the Curetis
Business received shareholder contributions from Curetis N.V. of kEUR 3,000 and kEUR 19,000 of which kEUR 3,109 are presented
as proceeds from current liabilities, net of issuance costs as that amount relates to the convertible notes that were issued by
Curetis N.V. and legally contributed to the capital reserve of Curetis GmbH. For presentation purposes (See Note 2), that amount
was presented as if the Curetis Business has issued such convertible notes.
Total compensation of key management:
Certain amounts of these totals have previously been recharged in the normal course of business. For those amounts not recharged,
they have been included in Shareholder contribution in Equity. See Note 3.19.
2018
|
in kEUR
|
|
|
|
|
|
|
|
|
|
|
|
Name
|
|
Base
salary/ consultancy fee
|
|
Annual
bonus 1
|
|
Company
Car
|
|
Share
based payments and other incentives
|
|
Total
remuneration
|
|
Johannes
Bacher
|
|
|
220
|
2
|
|
|
12
|
|
|
|
—
|
|
|
|
60
|
3
|
|
|
292
|
|
|
Dr.
Achim Plum
|
|
|
200
|
|
|
|
15
|
|
|
|
5
|
|
|
|
60
|
3
|
|
|
280
|
|
|
Oliver
Schacht
|
|
|
240
|
|
|
|
18
|
|
|
|
—
|
|
|
|
60
|
3
|
|
|
318
|
|
|
Total
|
|
|
660
|
|
|
|
45
|
|
|
|
5
|
|
|
|
180
|
|
|
|
890
|
|
1
Relates to the bonus that was paid in 2018 post FDA clearance
2
Includes holiday compensation payouts
3
Expenses recognized for granted ESOP
2017
|
in kEUR
|
|
|
|
|
|
|
|
|
|
|
|
Name
|
|
Base
salary/ consultancy fee
|
|
Annual
bonus 1
|
|
Company
Car
|
|
Share
based payments and other incentives
|
|
Total
remuneration
|
|
Johannes
Bacher
|
|
|
221
|
|
|
|
32
|
|
|
|
—
|
|
|
|
196
|
|
|
|
449
|
|
|
Dr.
Achim Plum
|
|
|
215
|
|
|
|
30
|
|
|
|
5
|
|
|
|
196
|
|
|
|
446
|
|
|
Andreas
Boos
|
|
|
128
|
|
|
|
17
|
|
|
|
|
|
|
|
23
|
|
|
|
168
|
|
|
Oliver
Schacht
|
|
|
244
|
|
|
|
45
|
|
|
|
—
|
|
|
|
196
|
|
|
|
485
|
|
|
Total
|
|
|
808
|
|
|
|
124
|
|
|
|
5
|
|
|
|
611
|
|
|
|
1,548
|
|
1
Relates to the bonus for performance year 2017 that was paid in 2018
|
in kEUR
|
|
2018
|
|
2017
|
|
Salaries
and other short-term employee benefits
|
|
|
705
|
|
|
|
965
|
|
|
Post-employment
benefits 1
|
|
|
8
|
|
|
|
—
|
|
|
Share based payments
|
|
|
180
|
|
|
|
622
|
|
|
Other
|
|
|
5
|
|
|
|
5
|
|
|
Total
|
|
|
898
|
|
|
|
1,592
|
|
1 Post-employment benefits relate
to the remuneration of a former managing director
Total compensation of Supervisory
Board: Certain amounts of these totals have previously been recharged in the normal course of business. For those amounts not
recharged, they have been included in Shareholder contribution in Equity. See note 3.19.
The
compensation of Supervisory Board is shown below:
|
in kEUR
|
|
2018
|
|
2017
|
|
William
E. Rhodes
|
|
|
105
|
|
|
|
95
|
|
|
Dr.
Werner Schäfer
|
|
|
83
|
|
|
|
75
|
|
|
Mario
Crovetto
|
|
|
64
|
|
|
|
55
|
|
|
Prabhavathi
Fernandes
|
|
|
53
|
|
|
|
45
|
|
|
Nils
Clausnitzer
|
|
|
51
|
|
|
|
31
|
|
|
Dr.
Holger Reithinger
|
|
|
(11
|
)
|
|
|
11
|
|
|
Total
|
|
|
345
|
|
|
|
312
|
|
|
thereof
from equity stock options
|
|
|
99
|
|
|
|
66
|
|
The
reason why equity stock options have been granted to the Supervisory Board Members are:
(i)
Alignment of strategic interest of Supervisory Board Members with the company and its shareholders.
(ii)
Ability to recruit, retain and incentivize Supervisory Board Members in line with what is market standard e.g. in the USA.
Curetis
does not grant any loans, advance payments and guarantees to members of the Management and Supervisory Board. There have been
no other notable related party transactions.
EVENTS
AFTER THE BALANCE SHEET DATE
Subsequent
to 31 December 2018:
|
|
●
|
Curetis
GmbH entered into an agreement to combine its business with OpGen. See Note 2.1 for further
information
|
|
|
●
|
Curetis
GmbH has drawn EUR 5 million on the existing EIB tranche.
|
|
|
●
|
Curetis
has drawn down the remaining EUR 1.5 million from the first tranche of the existing Yorkville
Convertible Notes Facility.
|
|
|
●
|
Curetis
liquidated two of their wholly owned subsidiaries, Curetis France S.A.R.L. and Curetis
BeNeLux B.V.
|
|
Holzgerlingen,
15 September 2019
Curetis GmbH
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
/s/
Oliver Schacht, PhD
|
|
/s/
Johannes Bacher
|
|
Oliver
Schacht, PhD
Chief
Executive Officer (CEO)
Managing
Director
|
|
Johannes
Bacher
Chief
Operating Officer (COO)
Managing
Director
|
|
|
|
|
|
|
|
|
|
|
|
|
|
/s/
Achim Plum
|
|
/s/
Heiko Schorr
|
|
Dr.
Achim Plum
Chief
Business Officer (CBO)
Managing
Director
|
|
Heiko
Schorr
Director
Finance
Managing
Director
|
Report
of Independent Auditors
To
the management of Curetis GmbH
We have audited the
accompanying combined financial statements of the Curetis business of Curetis N.V., which comprise the combined statements of
financial position as of December 31, 2018, December 31, 2017, and January 1, 2017 and the related combined statements of operations
and other comprehensive loss, cash flows and changes in equity for the years ended December 31, 2018 and December 31, 2017.
Management's Responsibility
for the Combined Financial Statements
Management is responsible
for the preparation and fair presentation of the combined financial statements in accordance with International Financial Reporting
Standards as issued by the International Accounting Standards Board; this includes the design, implementation, and maintenance
of internal control relevant to the preparation and fair presentation of combined financial statements that are free from material
misstatement, whether due to fraud or error.
Auditors’
Responsibility
Our responsibility is
to express an opinion on the combined financial statements based on our audits. We conducted our audits in accordance with auditing
standards generally accepted in the United States of America. Those standards require that we plan and perform the audit to obtain
reasonable assurance about whether the combined financial statements are free from material misstatement.
An audit involves performing
procedures to obtain audit evidence about the amounts and disclosures in the combined financial statements. The procedures selected
depend on our judgment, including the assessment of the risks of material misstatement of the combined financial statements, whether
due to fraud or error. In making those risk assessments, we consider internal control relevant to the Company's preparation and
fair presentation of the combined financial statements in order to design audit procedures that are appropriate in the circumstances,
but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control. Accordingly, we express
no such opinion. An audit also includes evaluating the appropriateness of accounting policies used and the reasonableness of significant
accounting estimates made by management, as well as evaluating the overall presentation of the combined financial statements.
We believe that the audit evidence we have obtained is sufficient and appropriate to provide a basis for our audit opinion.
Opinion
In our opinion, the
combined financial statements referred to above present fairly, in all material respects, the financial position of the Curetis
business of Curetis N.V. as of December 31, 2018, December 31, 2017 and January 1, 2017 and the results of its operations and
its cash flows for the years ended December 31, 2018 and December 31, 2017 in accordance with International Financial Reporting
Standards as issued by the International Accounting Standards Board.
Emphasis
of Matter
The accompanying
combined financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note
3.22 to the combined financial statements, the Company has suffered recurring losses from operations, has an accumulated deficit,
and negative cash outflows from operating activities, and has stated that substantial doubt exists about the Company’s ability
to continue as a going concern. Management's evaluation of the events and conditions and management’s plans regarding these
matters are also described in Note 3.22. The combined financial statements do not include any adjustments that might result from
the outcome of this uncertainty. Our opinion is not modified with respect to this matter.
Munich, Germany
September 15, 2019
PricewaterhouseCoopers GmbH
Wirtschaftsprüfungsgesellschaft
|
|
|
|
|
/s/ Dietmar Eglauer
|
|
/s/ ppa. Andreas Schuster
|
|
Dietmar Eglauer
|
|
ppa. Andreas Schuster
|
|
Wirtschaftsprüfer
|
|
Wirtschaftsprüfer
|
|
(German Public Auditor)
|
|
(German Public Auditor)
|
CURETIS
BUSINESS
UNAUDITED
INTERIM CONDENSED COMBINED STATEMENT OF OPERATIONS AND OTHER COMPREHENSIVE LOSS
For
the six months ended 30 June
|
in kEuro
|
|
Note
|
|
six
months ended 30 June 2019
|
|
six
months ended 30 June 2018
|
|
|
|
|
|
|
|
|
|
Revenue
|
|
|
4
|
|
|
|
1,088
|
|
|
|
807
|
|
|
Cost
of sales
|
|
|
|
|
|
|
(1,342
|
)
|
|
|
(1,095
|
)
|
|
Gross
profit (loss)
|
|
|
|
|
|
|
(254
|
)
|
|
|
(288
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Distribution
costs
|
|
|
5
|
|
|
|
(3,270
|
)
|
|
|
(4,209
|
)
|
|
Administrative
expenses
|
|
|
6
|
|
|
|
(1,612
|
)
|
|
|
(1,867
|
)
|
|
Research
& development expenses
|
|
|
7
|
|
|
|
(4,181
|
)
|
|
|
(4,680
|
)
|
|
Other
income
|
|
|
|
|
|
|
114
|
|
|
|
189
|
|
|
Operating
loss
|
|
|
|
|
|
|
(9,203
|
)
|
|
|
(10,855
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Finance
income
|
|
|
|
|
|
|
7
|
|
|
|
50
|
|
|
Finance
costs
|
|
|
|
|
|
|
(744
|
)
|
|
|
(494
|
)
|
|
Finance
result - net
|
|
|
8
|
|
|
|
(737
|
)
|
|
|
(444
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss
before income tax
|
|
|
|
|
|
|
(9,940
|
)
|
|
|
(11,299
|
)
|
|
Income
tax expenses
|
|
|
|
|
|
|
(56
|
)
|
|
|
26
|
|
|
Loss
for the period
|
|
|
|
|
|
|
(9,996
|
)
|
|
|
(11,273
|
)
|
|
Foreign
currency translation gain (loss)*
|
|
|
|
|
|
|
13
|
|
|
|
53
|
|
|
Total
comprehensive loss for the period
|
|
|
|
|
|
|
(9,983
|
)
|
|
|
(11,220
|
)
|
*Exchange
differences on translation of foreign operations, which may be recycled through profit and/or loss in the future.
The
accompanying notes are an integral part of these combined financial statements.
CURETIS
BUSINESS
UNAUDITED
INTERIM CONDENSED COMBINED STATEMENT OF FINANCIAL POSITION
As
of 30 June 2019 and 31 December 2018
|
in kEuro
|
|
Note
|
|
30
June 2019
|
|
31
December 2018
|
|
|
|
|
|
|
|
|
|
Current
assets
|
|
|
|
|
|
|
10,732
|
|
|
|
11,888
|
|
|
Cash
and cash equivalents
|
|
|
13
|
|
|
|
4,779
|
|
|
|
4,800
|
|
|
Trade
receivables
|
|
|
13
|
|
|
|
196
|
|
|
|
323
|
|
|
Other
receivables, related party
|
|
|
16
|
|
|
|
264
|
|
|
|
453
|
|
|
Contractual
assets
|
|
|
|
|
|
|
215
|
|
|
|
—
|
|
|
Inventories
|
|
|
9
|
|
|
|
4,715
|
|
|
|
6,052
|
|
|
Prepaid
Expenses and Other current assets
|
|
|
|
|
|
|
563
|
|
|
|
260
|
|
|
Non-current
assets
|
|
|
|
|
|
|
12,563
|
|
|
|
10,850
|
|
|
Intangible
assets
|
|
|
|
|
|
|
7,354
|
|
|
|
7,425
|
|
|
Property,
plant and equipment
|
|
|
10
|
|
|
|
3,738
|
|
|
|
3,196
|
|
|
Right
of use assets
|
|
|
11
|
|
|
|
1,298
|
|
|
|
—
|
|
|
Other
non-current financial assets
|
|
|
13
|
|
|
|
158
|
|
|
|
158
|
|
|
Deferred
tax assets
|
|
|
|
|
|
|
15
|
|
|
|
71
|
|
|
Total
assets
|
|
|
|
|
|
|
23,295
|
|
|
|
22,738
|
|
|
Current
liabilities
|
|
|
|
|
|
|
5,701
|
|
|
|
5,773
|
|
|
Trade
and other payables
|
|
|
13
|
|
|
|
731
|
|
|
|
921
|
|
|
Other
liabilities, related party
|
|
|
16
|
|
|
|
186
|
|
|
|
187
|
|
|
Provisions
current
|
|
|
|
|
|
|
130
|
|
|
|
65
|
|
|
Tax
liabilities
|
|
|
|
|
|
|
2
|
|
|
|
22
|
|
|
Other
current liabilities
|
|
|
|
|
|
|
952
|
|
|
|
881
|
|
|
Other
current financial liabilities
|
|
|
12,13
|
|
|
|
3,267
|
|
|
|
3,697
|
|
|
Current
lease liabilities
|
|
|
15
|
|
|
|
433
|
|
|
|
—
|
|
|
Non-current
liabilities
|
|
|
|
|
|
|
20,539
|
|
|
|
13,993
|
|
|
Provisions
non-current
|
|
|
|
|
|
|
44
|
|
|
|
44
|
|
|
Other
non-current financial liabilities
|
|
|
12,13
|
|
|
|
19,623
|
|
|
|
13,949
|
|
|
Non-current
lease liabilities
|
|
|
15
|
|
|
|
872
|
|
|
|
—
|
|
|
Total
liabilities
|
|
|
|
|
|
|
26,240
|
|
|
|
19,766
|
|
|
Equity
|
|
|
|
|
|
|
(2,945
|
)
|
|
|
2,972
|
|
|
Subscribed
capital
|
|
|
|
|
|
|
5,554
|
|
|
|
5,554
|
|
|
Capital
reserve
|
|
|
|
|
|
|
161,913
|
|
|
|
157,847
|
|
|
Currency
translation differences
|
|
|
|
|
|
|
(62
|
)
|
|
|
(75
|
)
|
|
Accumulated
deficit
|
|
|
|
|
|
|
(170,350
|
)
|
|
|
(160,354
|
)
|
|
Total
Equity and liabilities
|
|
|
|
|
|
|
23,295
|
|
|
|
22,738
|
|
The
accompanying notes are an integral part of these combined financial statements.
CURETIS
BUSINESS
UNAUDITERD
INTERIM CONDENSED COMBINED Statement of Cash Flows
For
the six months ended 30 June
|
in kEuro
|
|
|
|
six months
ended
|
|
six months
ended
|
|
|
|
Note
|
|
30
June 2019
|
|
30
June 2018
|
|
Net
loss for the period
|
|
|
|
|
|
|
(9,996
|
)
|
|
|
(11,273
|
)
|
|
Adjustment
for:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-
Net finance income (costs)
|
|
|
8
|
|
|
|
737
|
|
|
|
444
|
|
|
-
Depreciation, amortization and impairments
|
|
|
10,11
|
|
|
|
825
|
|
|
|
618
|
|
|
-
Share-based payment expense
|
|
|
14
|
|
|
|
247
|
|
|
|
251
|
|
|
-
Changes in deferred tax assets and liabilities
|
|
|
|
|
|
|
56
|
|
|
|
(45
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Changes
in working capital relating to:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-
Inventories
|
|
|
9
|
|
|
|
1,337
|
|
|
|
(215
|
)
|
|
-
Trade receivables and other receivables
|
|
|
|
|
|
|
(197
|
)
|
|
|
608
|
|
|
-
Trade payables and other payables
|
|
|
|
|
|
|
235
|
|
|
|
(409
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income
taxes received (+) / paid (-)
|
|
|
|
|
|
|
56
|
|
|
|
(26
|
)
|
|
Interest
paid (-)
|
|
|
|
|
|
|
(530
|
)
|
|
|
(406
|
)
|
|
Net
cash flow used in operating activities
|
|
|
|
|
|
|
(7,230
|
)
|
|
|
(10,453
|
)
|
|
Payments
for intangible assets
|
|
|
|
|
|
|
(31
|
)
|
|
|
(67
|
)
|
|
Payments
for property, plant and equipment
|
|
|
10
|
|
|
|
(1,049
|
)
|
|
|
(163
|
)
|
|
Net
cash flow used in investing activities
|
|
|
|
|
|
|
(1,080
|
)
|
|
|
(230
|
)
|
|
Proceeds
from other non-current financial liabilities
|
|
|
12
|
|
|
|
5,000
|
|
|
|
3,000
|
|
|
Proceeds
from current financial liabilities (convertible notes), net of issuance cost
|
|
|
|
|
|
|
1,373
|
|
|
|
—
|
|
|
Capital
increase in cash from shareholder
|
|
|
|
|
|
|
1,627
|
|
|
|
10,000
|
|
|
Shareholder
contributions
|
|
|
|
|
|
|
478
|
|
|
|
725
|
|
|
Principle
elements of leases paid
|
|
|
|
|
|
|
(203
|
)
|
|
|
—
|
|
|
Net
cash flow provided by financing activities
|
|
|
|
|
|
|
8,275
|
|
|
|
13,725
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net decrease
/ increase in cash and cash equivalents
|
|
|
|
|
|
|
(35
|
)
|
|
|
3,042
|
|
|
Net
cash and cash equivalents at the beginning of the year
|
|
|
|
|
|
|
4,800
|
|
|
|
3,468
|
|
|
Effects
of exchange rate changes on cash and cash equivalents
|
|
|
|
|
|
|
14
|
|
|
|
76
|
|
|
Net
Cash and cash equivalents at the end of the period
|
|
|
|
|
|
|
4,779
|
|
|
|
6,586
|
|
The
accompanying notes are an integral part of these combined financial statements.
CURETIS
BUSINESS
UNAUDITED
INTERIM Condensed COMBINED Statement of Changes in Equity
For
the six months ended 30 June
|
|
|
|
|
|
|
Currency
|
|
|
|
|
|
|
|
Subscribed
|
|
Capital
|
|
translation
|
|
Accumulated
|
|
TOTAL
|
|
in
kEuro
|
|
capital
|
|
reserve
|
|
difference
|
|
deficit
|
|
equity
|
|
Balance
at 1 January 2018
|
|
|
5,554
|
|
|
|
140,402
|
|
|
|
(160
|
)
|
|
|
(137,543
|
)
|
|
|
8,253
|
|
|
Loss of the period
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(11,273
|
)
|
|
|
(11,273
|
)
|
|
Other
comprehensive income
|
|
|
|
|
|
|
|
|
|
|
50
|
|
|
|
|
|
|
|
50
|
|
|
Total
comprehensive income
|
|
|
—
|
|
|
|
—
|
|
|
|
50
|
|
|
|
(11,273
|
)
|
|
|
(11,223
|
)
|
|
Transactions
with owners in their capacity as owners
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Capital
increase
|
|
|
|
|
|
|
10,000
|
|
|
|
|
|
|
|
|
|
|
|
10,000
|
|
|
Shareholder
contributions
|
|
|
|
|
|
|
725
|
|
|
|
|
|
|
|
|
|
|
|
725
|
|
|
Share-based
payments
|
|
|
|
|
|
|
251
|
|
|
|
|
|
|
|
|
|
|
|
251
|
|
|
Balance
as of 30 June 2018
|
|
|
5,554
|
|
|
|
151,378
|
|
|
|
(110
|
)
|
|
|
(148,816
|
)
|
|
|
8,006
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Currency
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Subscribed
|
|
|
|
Capital
|
|
|
|
translation
|
|
|
|
Accumulated
|
|
|
|
TOTAL
|
|
|
in
kEuro
|
|
|
capital
|
|
|
|
reserve
|
|
|
|
difference
|
|
|
|
deficit
|
|
|
|
equity
|
|
|
Balance
at 1 January 2019
|
|
|
5,554
|
|
|
|
157,847
|
|
|
|
(75
|
)
|
|
|
(160,354
|
)
|
|
|
2,972
|
|
|
Loss of the period
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(9,996
|
)
|
|
|
(9,996
|
)
|
|
Other
comprehensive income
|
|
|
|
|
|
|
|
|
|
|
13
|
|
|
|
|
|
|
|
13
|
|
|
Total
comprehensive income
|
|
|
—
|
|
|
|
—
|
|
|
|
13
|
|
|
|
(9,996
|
)
|
|
|
(9,983
|
)
|
|
Transactions
with owners in their capacity as owners
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Capital
increase
|
|
|
|
|
|
|
3,341
|
|
|
|
|
|
|
|
|
|
|
|
3,341
|
|
|
Shareholder
contribution
|
|
|
|
|
|
|
478
|
|
|
|
|
|
|
|
|
|
|
|
478
|
|
|
Share-based
payments
|
|
|
|
|
|
|
247
|
|
|
|
|
|
|
|
|
|
|
|
247
|
|
|
Balance
as of 30 June 2019
|
|
|
5,554
|
|
|
|
161,913
|
|
|
|
(62
|
)
|
|
|
(170,350
|
)
|
|
|
(2,945
|
)
|
The
accompanying notes are an integral part of these combined financial statements.
Curetis
BUSINESS
Notes
to the UNAUDTIED INTERIM CONDENDSED COMBINED financial statements
|
|
1.
|
GENERAL
INFORMATION ABOUT THE COMPANY
|
These
combined financial statements comprise the Curetis business (collectively referred to as “the Group,” “Curetis”
or the “Company”). The Group´s headquarter is located at Max-Eyth-Str. 42, 71088 Holzgerlingen, Germany. The
Group is an early commercial-stage molecular diagnostics (MDx) company focused on rapid infectious disease testing for hospitalized
patients with the aim to improve the treatment of critically ill patients with suspected microbial infections. The Curetis business
is primarily operated by Curetis GmbH and its wholly owned subsidiaries.
The
first Group entity, Curetis AG, was created in Germany in 2007 and was primarily funded through equity investments from venture
capital and private equity investors. In 2015, in connection with an initial public offering (“IPO”), Curetis N.V.
was created as a parent entity to Curetis AG and in that same year the stock of Curetis N.V. was sold in an IPO on the Euronext
market. In 2016 Curetis AG was changed to Curetis GmbH. Since 2015, the operations of Curetis have been financed through contributions
from Curetis N.V. from proceeds of the IPO, secondary offerings, and various other financing agreements Curetis N.V. has entered
into, including Convertible Notes, the EIB financing and government grants.
At
15 September 2019 the Management Board authorized the unaudited interim condensed combined financial statements for issue and
passed it through to the Supervisory Board for review and authorization.
|
|
1.1.
|
General
information about the business and the commercial development of the company.
|
The
Group has developed the innovative Unyvero molecular diagnostic solution for comprehensive infectious disease testing. Curetis´
proprietary application portfolio for its Unyvero system currently consists of several CE-marked applications:
|
|
·
|
The
Unyvero HPN (Hospitalized Pneumonia) cartridge for the detection of pathogens and antibiotic
resistances to aid diagnosing pneumonia.
|
|
|
·
|
The
Unyvero ITI (Implant and tissue infections) cartridge for the detection of pathogens
and antibiotic resistance markers in diagnosis of prosthetic joint infections, surgical
site infections, infections associated with implants, infections of the deep skin and
soft tissue, burn wounds as well as diabetic foot, cellulitis and others.
|
|
|
·
|
The
Unyvero BCU (Blood culture) cartridge for the detection of pathogens (bacteria and fungi)
and antibiotic resistance markers in bloodstream infections.
|
|
|
·
|
The
Unyvero IAI (Intra-abdominal infections) cartridge for the detection of targeted microorganisms
and antibiotic resistance markers.
|
|
|
·
|
The
Unyvero UTI (Urinary tract infections) cartridge for the detection of severe cases of
urinary tract infection targets, microorganisms and antibiotic resistance markers.
|
During
2019 Curetis began execution of the previously announced reorganization of its corporate structure. The planned measures included
the closure and liquidation of the following subsidiaries of Curetis GmbH.
|
|
·
|
Curetis
UK Ltd., London, UK (in liquidation)
|
|
|
·
|
Curetis
Schweiz GmbH, Zug, Switzerland (in liquidation)
|
|
|
·
|
Curetis
BeNeLux B.V. , Amsterdam, the Netherlands (dissolved 25.06.2019)
|
|
|
·
|
Curetis
France S.A.R.L., Strasbourg, France (dissolved 24.03.2019)
|
|
|
2.
|
BASIS
OF PREPARATION – UNAUDITED INTERIM CONDENSED COMBINED FINANCIAL STATEMENTS
|
2.1.
Basis of presentation
The
accompanying unaudited interim condensed combined financial statements of Curetis have been prepared for filing with the United
States Securities and Exchange Commission (SEC) in connection with the proposed acquisition of all of the outstanding shares of
Curetis GmbH by OpGen Inc. (“OpGen”), pursuant to an agreement to combine the two companies’ businesses. Following
the agreement, OpGen will acquire 100% of Curetis GmbH’s assets and liabilities, including the Curetis name as well as the
outstanding indebtedness of Curetis N.V. under certain convertible notes, including providing that the conversion rights of the
notes may be changed from a right to convert into shares of Curetis N.V to a right to convert into shares of OpGen. In addition,
OpGen has also agreed to acquire all of the assets of Curetis N.V. that are solely and exclusively related to the business of
Curetis GmbH and assume (1) the Curetis N.V. 2016 Stock Option Plan, as amended, and the outstanding awards thereunder, or the
2016 Stock Option Plan, and (2) the obligation to issue equity to the holders of awards under the Curetis AG Phantom Stock Option
Plan, or the PSOP. OpGen will also assume all of the liabilities of Curetis N.V. solely and exclusively related to the business
being acquired.
The
business combination is subject to a number of conditions including (i) the satisfaction of customary conditions to closing for
a transaction of this type, including the absence of a material adverse event for either party, (ii) for each OpGen and Curetis,
appropriate approvals by their respective shareholders, (iii) for Curetis, consents from certain debt financing providers, (iv)
the Form S-4 having been declared effective by the U.S. Securities and Exchange Commission, (v) the new shares of OpGen’s
common stock to be issued (or reserved for issuance) in connection with the transaction having been approved for listing on Nasdaq
and (vi) OpGen having secured additional funding prior to Closing.
The
Curetis business is primarily operated by Curetis GmbH and its wholly owned subsidiaries. However, certain costs related to the
Curetis business, primarily related to the compensation of certain members of senior management and its supervisory board, have
historically been incurred by Curetis N.V. but have not been recharged by Curetis N.V. to Curetis GmbH or its subsidiaries. SEC
Staff Accounting Bulletin (SAB) Topic 1.B. (“SAB 1.B”) Allocation of Expenses and Related Disclosure in Financial
Statements of Subsidiaries, Divisions or Lessor Business Components of Another Entities states that the historical income
statements of a registrant should reflect all of its costs of doing business and therefore in specific situations requires a subsidiary
to reflect certain expenses incurred by the parent on its behalf. In addition, the unaudited interim combined financial statements
include the convertible notes issued by Curetis N.V. as well as related expense. The proceeds of the issuance of the convertible
notes were historically contributed to the Curetis GmbH via cash contributions to capital reserves. Accordingly, the unaudited
interim condensed combined financial information of Curetis have been prepared to combine the consolidated interim financial statements
of Curetis GmbH together with certain costs incurred by Curetis N.V. on behalf of Curetis GmbH. As a result, the unaudited interim
condensed combined financial statements of Curetis do not currently constitute a separate group of legal entities.
During
the six months ended 30 June 2018 and 2019, the costs incurred by Curetis N.V. that have been allocated to the Company for the
purposes of preparing the unaudited interim condensed combined financial statements are based on a specific identification basis
where possible. Management believes that the assumptions used in determining these allocations are reasonable. However, the financial
statements may not necessarily reflect the Company’s financial position, results of operations, or cash flows in the future,
or what its financial position, results of operations, or cash flows would have been if it had been a stand-alone entity during
the periods presented.
IFRS
does not provide principles for the preparation of combined financial statements for carve-out financial statements, and accordingly
in preparing the unaudited interim condensed
combined financial statements certain accounting and allocation conventions commonly used
in practice for the preparation of carve-out financial statements were applied. The assets and liabilities included in the unaudited
interim condensed combined statement of financial position were measured at the carrying amounts recorded in the Curetis N.V.
condensed interim consolidated financial statements.
The
unaudited interim condensed combined financial statements and notes for the six months ended 30 June 2019 have been prepared in
accordance with International Accounting Standards (“IAS”) 34 Interim Financial Reporting and have been prepared
on the same basis of accounting as the audited annual combined financial statements, with the exception of the accounting of lease
agreements, which are accounted for according to IFRS 16 Leases starting 1 January 2019 (refer to note 3.1). Certain information
and footnote disclosures typically included in annual financial statements prepared in accordance with IFRS have been condensed
or omitted. Accordingly, these unaudited interim condensed combined financial statements should be read in conjunction with the
Company’s combined financial statements as of and for the year ended 31 December 2018.
The
condensed combined financial statements have been prepared on a going concern basis (see also Note 3.4 below). These condensed
combined financial statements are presented in Euro and, where appropriate, have been rounded to the nearest thousand (abbreviated
kEUR). All intercompany accounts and transactions have been eliminated in consolidation.
2.2.
Scope of combination
Curetis
GmbH is domiciled in Germany. Details of the Group´s subsidiaries at the end of the reporting period are as follows:
|
Name
|
|
Registration
No.
|
|
Country
|
|
Participation
|
|
Main
activity
|
|
Curetis
USA Inc.
|
|
|
EIN
81-3113346
|
|
|
|
USA
|
|
|
|
100.00%
|
|
|
Sale
of molecular diagnostic products
|
|
Curetis
UK Ltd.
|
|
|
10164457
|
|
|
|
UK
|
|
|
|
100.00%
|
|
|
Sale
of molecular diagnostic products
|
|
Curetis
Schweiz GmbH
|
|
|
CHE-228.103.501
|
|
|
|
Switzerland
|
|
|
|
100.00%
|
|
|
Sale
of molecular diagnostic products
|
|
Ares
Genetics GmbH
|
|
|
468899h
|
|
|
|
Austria
|
|
|
|
100.00%
|
|
|
Maximize
R&D and related scientific opportunities with Aresdb Bio-IT platform (previously GEAR)
|
2.3. Critical
accounting judgements and key sources of estimation uncertainty
The
preparation of financial statements in conformity with IFRS requires management to make estimates and assumptions that affect
the reported amounts of assets and liabilities, the disclosure of contingent assets and liabilities at the date of the financial
statements and the reported amounts of revenue, income and expenses during the reporting periods. Significant estimates and assumptions
reflected in these condensed combined financial statements include, but are not limited to, the useful life of intangible assets,
provisions, inventory valuation, and lease term. The Company bases its estimates on historical experience, known trends and other
market-specific or other relevant factors that it believes to be reasonable under the circumstances. On an ongoing basis, management
evaluates its estimates, as there are changes in circumstances, facts and experience. Actual results may differ from those estimates
or assumptions.
Preparing
these carve-out combined financial statements required management to make judgement within the identification of certain costs
incurred by Curetis N.V. on behalf of Curetis GmbH and reflected back to the combined financial statements of Curetis GmbH. Management
evaluated on historical experience the best approach by identifying such costs.
|
|
3.
|
SUMMARY
OF SIGNIFICANT ACCOUNTING POLICIES
|
The
principal accounting policies set out below have been applied consistently to all periods presented in these condensed combined
financial statements, unless otherwise stated.
3.1.
New standards and interpretations applied for the first time
The
accounting policies adopted in the preparation of the interim financial statements are consistent with those followed in the preparation
of the Group´s annual combined financial statements for the year ended 31 December 2018, except for the adoption of new
standards effective as of 1 January 2019. New standards, amendments to standards and new or amended interpretations are effective
for annual periods beginning on or after 1 January 2019, and have been applied as required in preparing these financial statements.
Curetis has not opted for early adoption for any of these standards.
|
Standard/Interpretation
|
|
Content
|
|
Application
mandatory from
|
|
Amendment
to IFRS 9
|
|
Prepayment
Features with Negative Compensation
|
|
1
January 2019
|
|
IFRS
16
|
|
Accounting
of Leasing-transactions
|
|
1
January 2019
|
|
IFRIC
23
|
|
Uncertainty
over Income Tax Treatments
|
|
1
January 2019
|
|
Amendments
to IFRS 3, IAS 11, IAS 12, IAS 23
|
|
Amended
by Annual Improvements to IFRS Standards 2015–2017 Cycle
|
|
1
January 2019
|
|
Amendments
to IAS 19
|
|
Plan
Amendment, Curtailment or Settlement
|
|
1
January 2019
|
The
Group has assessed the accounting standards effective after 1 January 2019 and determined that none have a material impact on
the unaudited condensed combined financial statements with the exception of IFRS 16, which has been applied in these interim financial
statements and as required by IAS 34, the nature and effect of these changes are disclosed below.
First
time adoption of IFRS 16 – Leases
Adopted
as of current period
In
January 2016, the IASB published the financial reporting standard IFRS 16 Leases which replaces IAS 17 Leases as
well as the associated interpretations. The new standard became effective on 1 January 2019 and sets out the principles for the
recognition, measurement, presentation and disclosure of leases. Under the new lease standard, assets leased by the Company are
being recognized as a right-of-use asset in the statements of financial position with a corresponding lease liability.
Lessor
accounting under IFRS 16 is substantially unchanged from IAS 17. Lessors will continue to classify leases as either operating
or finance leases using similar principles as in IAS 17. The Group’s activities as a lessor are not material; therefore,
IFRS 16 did not have an impact for leases where the Company is the lessor.
The
Company adopted IFRS 16 using the simplified transition approach and did not restate comparative amounts for the year prior to
first adoption.
Previously,
the Company determined at contract inception whether an arrangement was or contained a lease under IFRIC 4 “Determining
Whether an Arrangement contains a Lease”. Leases entered into before the date of initial application were not reassessed
as to whether a contract is, or contains, a lease at the date of first-time application, but the assessment previously made under
IFRIC 4 was retained.
The
Group now assesses whether a contract is or contains a lease based on the new definition of a lease. Under IFRS 16, a contract
is, or contains, a lease if the contract conveys a right to control the use of an identified asset for a period in exchange for
consideration.
Lease
terms are negotiated on an individual basis and contain a range of different terms and conditions. Lease contracts are typically
negotiated for fixed periods, but may include extension options. These terms offer the Group the greatest possible operational
flexibility. For determining the lease terms all facts and circumstances are included which offer an economic incentive to exercise
extension options. Extension options are only included in the lease term if the lease is reasonably certain to be extended.
Transition
and impact assessment on IFRS 16
The
Company elected to adopt the practical expedient related to leases of all asset classes with a lease term of less than 12 months
or for which the underlying asset is of low value and leases with a remaining lease term of less than 12 months at the transition
date. In these cases, no right-of-use asset and lease liability is recognized. Lease payments on short-term leases and leases
of low-value assets are recognized as expense on a straight-line basis over the lease term.
The
effect of the adoption of IFRS 16 to the statements of financial position as of 1 January 2019 is as follows:
|
In
kEUR
|
|
Assets
|
|
Right-of-Use
assets
|
|
|
1,494
|
|
|
Liabilities
|
|
Lease
liabilities
|
|
|
1,494
|
|
The
adoption of IFRS 16 had no impact on the Company’s sales. Lease expense has been replaced by depreciation and interest expense,
which had an immaterial impact to the statement of operations for the six months ended 30 June 2019.
In
addition, the cash flow from operating activities for the six months ended 30 June 2019 was positively impacted by approximately
kEUR 224 as, under the new standard, cash payments for the principal portion of the lease liabilities are classified in the cash
flow from financing activities rather than in the cash flow from operating activities.
The
Company foresees no impact of the adoption of IFRS 16 on compliance with debt covenants.
Leases
previously accounted for as operating leases
The
Company recognized right-of-use assets and lease liabilities for those leases previously classified as operating leases, with
the exception of short-term leases and leases of low-value assets, as discussed above. The right-of-use assets and lease liabilities
were recognized based on the present value of the remaining lease payments and discounted using the incremental borrowing rate
implicit in the lease at the date of initial application. The company applied a discount rate of 1.9% for property and discount
rate of 3.9% for all other asset classes. For these two lease categories, the company applied the practical expedient to apply
a single discount rate for a portfolio of leases with similar characteristics.
The
lease liabilities as of 1 January 2019 reconciles to the operating lease commitments as of 31 December 2018 as follows (the amounts
in the table below include lease commitments for leases with extension options determined probable to exercise upon adoption):
|
in kEUR
|
|
|
|
Operating
lease commitments as of 31 December 2018
|
|
|
1,301
|
|
|
Impact of present
value discount
|
|
|
-107
|
|
|
Short
term leases excluded
|
|
|
-94
|
|
|
Impact
of lease extensions entered into in 2019
|
|
|
394
|
|
|
IFRS
16 opening balance impact on lease liabilities as of 1 January 2019
|
|
|
1,494
|
|
Summary
of new accounting policies
Right-of-use
assets
The
Company recognizes right-of-use assets at the commencement date of the lease (i.e., the date the underlying asset is available
for use). Right-of-use assets are measured at cost, less any accumulated depreciation and impairment losses, and adjusted for
any re-measurement of lease liabilities. The cost of right-of-use assets includes the amount of lease liabilities recognized,
initial direct costs incurred, and lease payments made at or before the commencement date, less any lease incentives received.
Unless the Company is reasonably certain ownership of the leased asset will be obtained at the end of the lease term, the recognized
right-of-use assets are depreciated on a straight-line basis over the shorter of its estimated useful life and the lease term.
Right-of-use assets are subject to impairment assessment.
Lease
liabilities
At
the commencement date of the lease, the Company recognizes lease liabilities measured at the present value of lease payments to
be made over the lease term. The lease payments include, in-substance, fixed payments less any lease incentives, variable lease
payments that depend on an index or a rate, and amounts expected to be paid under residual value guarantees. The lease payments
may also include an exercise price of a purchase option reasonably certain to be exercised by the Company and payments of penalties
for terminating a lease, if the lease term reflects the company exercising the termination option.
In
calculating the present value of lease payments, the Company uses the incremental borrowing rate at the lease commencement date
if the interest rate implicit in the lease is not readily determinable. After the commencement date, the amount of lease liabilities
is reduced for the lease payments made. In addition, the carrying amount of lease liabilities is re-measured if there is a contract
modification, change in the lease term, change in the in-substance fixed lease payments, or a change in the assessment to purchase
the underlying asset.
Significant
judgement in determining the lease term of contracts with renewal options
The
Company determines the lease term as the non-cancellable term of the lease, together with any periods covered by an option to
extend the lease if it is reasonably certain to be exercised, or any periods covered by an option to terminate the lease if it
is reasonably certain not to be exercised. When determining the lease term, Curetis considers all relevant facts and circumstances
that create an economic incentive to exercise an extension option, or not to exercise a termination option.
3.2.
Standards, interpretations, and amendments issued, but not yet applied
The
following new standards and interpretations and amendments to existing standards will become effective after 1 January 2020.
|
Standard/Interpretation
|
|
Content
|
|
Application
mandatory
from
|
|
Amendments
to IFRS 3
|
|
Clarifying
the definition of “businesses”
|
|
1
January 2020
|
|
Amendments
to IAS 1 and IAS 8
|
|
Clarifying
the definition of “material”
|
|
1
January 2020
|
|
IFRS
17 (replaces IFRS 4)
|
|
Insurance
Contract
|
|
1
January 2021
|
The
Group has assessed the accounting standards effective after 1 January 2020 and determined that none are likely to have a material
impact on the combined financial statements.
3.3.
Use of assumptions and estimates
The
preparation of interim financial statements in conformity with IFRS requires management to make estimates and assumptions that
affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial
statements and the reported amount of revenue and expenses during the period. Actual results could differ from those estimates.
Except for the judgement and estimates mentioned in relation to the adoption of IFRS 16, the significant estimates and judgements
in preparing the condensed combined interim financial statements, made by management in applying the accounting policies and the
sources of estimation uncertainty, were the same as those applied to the Company´s combined financial statements for the
year ended 31 December 2018.
3.4.
Going concern
Since
inception, the Company’s activities have consisted primarily of performing research and development to advance its technologies
and more recently, establishing sales and distribution networks to commercialize its technology. Through 30 June 2019, the Company
has not yet established a stable ongoing source of revenues sufficient to cover its operating costs and has funded its operations
through proceeds from equity investments, collaboration and licensing agreements, grants and borrowings under various agreements
with funding agencies, and contributions from Curetis N.V., the ultimate holding company of Curetis GmbH as of 30 June 2018 and
2019, from the sale of Curetis N.V. stock in an Initial Public Offering, secondary offerings and various other financing agreements.
Since inception, the Company has incurred recurring losses (with the exception of 2015 due to an extraordinary gain), including
net losses of EUR 10.0 million and EUR 22.7 million for the six months ended 30 June 2019 and year ended 31 December 2018, respectively.
As of 30 June 2019, the Company had an accumulated deficit of EUR 170.4 million, EUR 4.8 million in cash and cash equivalents,
trade receivables of EUR 0.2 million.
The
Company also realized the following inflows of funds from financing during 2019.
|
|
●
|
EIB
Debt Financing Facility has funded the EUR 5 million milestone tranche in June 2019,
however, Curetis believes this was the last of the debt financing tranches that Curetis
could or would access under the current EIB facility.
|
|
|
●
|
Yorkville
Convertible Notes facility withdrawal of EUR 1.5 million.
|
|
|
●
|
Capital
contribution from Curetis N.V. of EUR
1.6 million.
|
Despite
the cost reduction measures already implemented in Europe and the USA, the Company expects to continue to generate operating losses
in the foreseeable future, the existing current assets, including cash, as well as the aforementioned secured external funding
sources are not sufficient to finance Curetis’ operating activities for said 12 months after the signing date of these financial
statements.
Substantial
doubt regarding the Group’s ability to continue as a going concern exists as of 15 September 2019, the issuance date of
these unaudited condensed combined interim financial statements. The Company’s Management believes that if it can realize
cash-inflow and funding measures, execute on strategy options, realize liquidity planning and implement these planned measures
as needed, funding of our business operations for a period of at least 12 months after the issue date of these financial statements
is achievable. Curetis is in the process of evaluating and progressing strategic and liquidity planning options to be able to
raise additional capital and reduce costs, including:
|
|
●
|
The
negotiation and implementation of a strategic option and scenario that, if successful,
would allow Curetis to access the capital markets and raise additional capital again.
|
|
|
●
|
Curetis
aims at accessing cash relating to entering into one or more licensing and partnering
deal(s) around its Unyvero A30 RQ platform and Aresdb. A draft term sheet has been received
for Unyvero A30 RQ and is currently under negotiation; however, none are currently
committed or secured.
|
|
|
●
|
Potentially
putting on hold, delaying, or reducing further expenditures for certain R&D, commercialization
and operational programs.
|
The
Company has also engaged financial and other advisors to assist it in those efforts.
The
Company will seek additional funding and to execute on these strategic business and commercial plans in order to reach its development
and commercialization objectives. There are no assurances the Company will be able to obtain financing on acceptable or favorable
terms, or at all, and the Company may not be able to execute on strategic business and commercial plans or to enter into collaborations
or other arrangements. The Company is primarily dependent on its parent, Curetis N.V., for financing. Further, Curetis N.V. is
not an operational entity that generates cash inflows, rather, is reliant on its shareholders and other external financing to
remain funded. In the event the Company is unable to successfully raise additional capital during or before the fourth quarter
of 2019, the Company will not have sufficient cash flows and liquidity to finance its business operations as currently contemplated.
Accordingly, in such circumstances the Company would be compelled to immediately and significantly reduce general and administrative
expenses, delay research and development projects, and product portfolio expansion or commercialization efforts until it is able
to obtain sufficient financing, which could adversely affect its business prospects. If such sufficient financing is not received
on a timely basis, the Company would then need to pursue a plan to license or sell its assets, seek to be acquired by another
entity, cease operations and/or seek bankruptcy protection.
The
accompanying condensed combined interim financial statements have been prepared assuming that the Company will continue as a going
concern, which contemplates the realization of assets and the settlement of liabilities and commitments in the normal course of
business. The condensed combined financial statements do not reflect any adjustments relating to the recoverability and classification
of assets or the amounts and classification of liabilities that might be necessary if the Company is unable to continue as a going
concern.
|
in kEUR
|
|
six
months ended 30 June 2019
|
|
six
months ended 30 June 2018
|
|
Sale
of Unyvero-Systems
|
|
|
146
|
|
|
|
369
|
|
|
Sale of cartridges
|
|
|
425
|
|
|
|
435
|
|
|
Sale
of services
|
|
|
517
|
|
|
|
3
|
|
|
Total
|
|
|
1,088
|
|
|
|
807
|
|
The Sale of services includes 215
kEUR of revenues recognized for services provided but not yet billed, which are recorded as a contractual asset as of 30 June
2019
In
accordance with IFRS 8, Curetis is a single-segment entity. Revenues from external customers by territory, based on the destination
of the customers are as follows:
|
in kEUR
|
|
six
months ended 30 June 2019
|
|
six
months ended 30 June 2018
|
|
EMEA
|
|
|
940
|
|
|
|
657
|
|
|
USA
|
|
|
82
|
|
|
|
32
|
|
|
Asia
|
|
|
66
|
|
|
|
118
|
|
|
Total
|
|
|
1,088
|
|
|
|
807
|
|
All
revenues are derived from external customers, including hospitals as well as distribution partners.
|
in kEUR
|
|
six
months ended 30 June 2019
|
|
six
months ended 30 June 2018
|
|
Personnel
expenses
|
|
|
1,843
|
|
|
|
2,671
|
|
|
Depreciation
and Amortization
|
|
|
46
|
|
|
|
51
|
|
|
Other
operating expenses
|
|
|
1,381
|
|
|
|
1,487
|
|
|
thereof
marketing expenses
|
|
|
534
|
|
|
|
712
|
|
|
thereof
travel expenses
|
|
|
192
|
|
|
|
365
|
|
|
thereof
consulting, advisory & 3rd party service
|
|
|
108
|
|
|
|
107
|
|
|
TOTAL
|
|
|
3,270
|
|
|
|
4,209
|
|
|
|
6.
|
ADMINISTRATIVE
EXPENSES
|
|
in kEUR
|
|
six
months ended 30 June 2019
|
|
six
months ended 30 June 2018
|
|
Personnel
expenses
|
|
|
565
|
|
|
|
646
|
|
|
Depreciation
and Amortization
|
|
|
218
|
|
|
|
44
|
|
|
Other
expenses
|
|
|
829
|
|
|
|
1,177
|
|
|
thereof
for remuneration of supervisory board
|
|
|
206
|
|
|
|
210
|
|
|
thereof
consulting, advisory & 3rd party service
|
|
|
178
|
|
|
|
302
|
|
|
TOTAL
|
|
|
1,612
|
|
|
|
1,867
|
|
|
|
7.
|
RESEARCH
AND DEVELOPMENT EXPENSES
|
|
in kEUR
|
|
six
months ended 30 June 2019
|
|
six
months ended 30 June 2018
|
|
Personnel
expenses
|
|
|
1,805
|
|
|
|
1,992
|
|
|
Depreciation
and Amortization
|
|
|
373
|
|
|
|
354
|
|
|
Material
expenses
|
|
|
274
|
|
|
|
139
|
|
|
Other
expenses
|
|
|
1,729
|
|
|
|
2,195
|
|
|
thereof
IP-fees and expenses for patent lawyers
|
|
|
121
|
|
|
|
370
|
|
|
thereof
external services for clinical trial
|
|
|
99
|
|
|
|
83
|
|
|
thereof
costs for laboratory demand
|
|
|
115
|
|
|
|
278
|
|
|
thereof
consulting, advisory & 3rd party service
|
|
|
785
|
|
|
|
890
|
|
|
thereof
other manufacturing expenses for cartridges used in R&D
|
|
|
252
|
|
|
|
120
|
|
|
TOTAL
|
|
|
4,181
|
|
|
|
4,683
|
|
|
|
8.
|
FINANCE
RESULT / COSTS NET
|
In
the six months ended 30 June 2019 the net finance loss of kEUR 640 (six months ended 30 June 2018 to a loss of kEUR 444), is primarily
from interest from the EUR 18 million cumulative tranche drawn downs from the EIB debt facility and foreign currency exchange
difference resulting from the exchange rate difference of USD vs. EUR.
|
in kEUR
|
|
six
months ended 30 June 2019
|
|
six
months ended 30 June 2018
|
|
Foreign
exchange differences
|
|
|
1
|
|
|
|
27
|
|
|
Interests
for borrowings
|
|
|
(628
|
)
|
|
|
(471
|
)
|
|
Interest
and finance expenses for convertible notes
|
|
|
(97
|
)
|
|
|
—
|
|
|
Other
finance income / finance costs
|
|
|
(13
|
)
|
|
|
—
|
|
|
Finance
result/costs net
|
|
|
-737
|
|
|
|
-444
|
|
Interests
for borrowings represent interest and financing charges paid/payable for financial liabilities not at fair value through profit
or loss using the effective interest method.
|
in kEUR
|
|
30
June
2019
|
|
31
December
2018
|
|
Raw
materials
|
|
|
759
|
|
|
|
838
|
|
|
Semi-finished
goods
|
|
|
36
|
|
|
|
61
|
|
|
Trade
goods
|
|
|
3,657
|
|
|
|
4,987
|
|
|
Finished
goods
|
|
|
125
|
|
|
|
65
|
|
|
Spare
parts
|
|
|
138
|
|
|
|
101
|
|
|
Total
inventories, net
|
|
|
4,715
|
|
|
|
6,052
|
|
The
obsolescence write-downs on inventories is considered a significant estimate with inherent uncertainty. Given Curetis does not
yet have a reliable sales-track-record, the write-downs are based on the best estimate considering technical aging and estimated
sales volumes and prices for systems. If assumptions regarding future sales prices, volumes, useful life or product market potentials
are not appropriate, this may lead to a further need for write-off. A reduction in the estimated sales price of 10% would result
in an increase of obsolescence write-downs of kEUR 310, whereas an increase in the estimated sales price of 10% would result in
a decrease of the obsolescence write-downs of kEUR 217. A reduction in the estimated useful life of five years of the Unyvero
system by one year would result in an increase of obsolescence write-downs of kEUR 630, whereas an increase in the estimated useful
life of the Unyvero systems by one year would result in a decrease of obsolescence write-downs of kEUR 592.
The
change of write-off to net asset value of inventories recognized as an expense and included in ´Cost of Sales´ in
the six months ended 30 June 2019 amounted to kEUR 556 (2018: kEUR 126).
|
|
10.
|
PROPERTY,
PLANT AND EQUIPMENT
|
|
in
kEUR
|
|
Land
and buildings
|
|
Machines
and technical installation
|
|
Other
tangible assets
|
|
Assets
under construction
|
|
Total
|
|
Cost:
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as of 1 January 2018
|
|
|
72
|
|
|
|
7,852
|
|
|
|
2,636
|
|
|
|
289
|
|
|
|
10,849
|
|
|
Additions
|
|
|
—
|
|
|
|
31
|
|
|
|
215
|
|
|
|
424
|
|
|
|
670
|
|
|
Disposals
|
|
|
—
|
|
|
|
—
|
|
|
|
(81
|
)
|
|
|
—
|
|
|
|
(81
|
)
|
|
Reclassifications
|
|
|
—
|
|
|
|
417
|
|
|
|
—
|
|
|
|
(417
|
)
|
|
|
—
|
|
|
Balance
as of 31 December 2018
|
|
|
72
|
|
|
|
8,300
|
|
|
|
2,770
|
|
|
|
296
|
|
|
|
11,438
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated
depreciation:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as of 1 January 2018
|
|
|
(49
|
)
|
|
|
(5,243
|
)
|
|
|
(1,991
|
)
|
|
|
—
|
|
|
|
(7,283
|
)
|
|
Disposals
|
|
|
—
|
|
|
|
—
|
|
|
|
80
|
|
|
|
—
|
|
|
|
80
|
|
|
Depreciation
|
|
|
(8
|
)
|
|
|
(701
|
)
|
|
|
(330
|
)
|
|
|
—
|
|
|
|
(1,039
|
)
|
|
Reclassifications
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Balance
as of 31 December 2018
|
|
|
(57
|
)
|
|
|
(5,944
|
)
|
|
|
(2,241
|
)
|
|
|
—
|
|
|
|
(8,242
|
)
|
|
Carrying
amount as of 31 December 2018
|
|
|
15
|
|
|
|
2,356
|
|
|
|
529
|
|
|
|
296
|
|
|
|
3,196
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as of 1 January 2019
|
|
|
72
|
|
|
|
8,300
|
|
|
|
2,770
|
|
|
|
296
|
|
|
|
11,438
|
|
|
Additions
|
|
|
—
|
|
|
|
50
|
|
|
|
576
|
|
|
|
425
|
|
|
|
1,051
|
|
|
Disposals
|
|
|
—
|
|
|
|
(4
|
)
|
|
|
(11
|
)
|
|
|
—
|
|
|
|
(15
|
)
|
|
Reclassifications
|
|
|
—
|
|
|
|
193
|
|
|
|
—
|
|
|
|
(193
|
)
|
|
|
—
|
|
|
Balance
as of 30 June 2019
|
|
|
72
|
|
|
|
8,539
|
|
|
|
3,335
|
|
|
|
528
|
|
|
|
12,474
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated
depreciation:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as of 1 January 2019
|
|
|
(57
|
)
|
|
|
(5,944
|
)
|
|
|
(2,241
|
)
|
|
|
—
|
|
|
|
(8,242
|
)
|
|
Disposals
|
|
|
—
|
|
|
|
3
|
|
|
|
7
|
|
|
|
—
|
|
|
|
10
|
|
|
Depreciation
|
|
|
(4
|
)
|
|
|
(343
|
)
|
|
|
(157
|
)
|
|
|
—
|
|
|
|
(504
|
)
|
|
Reclassifications
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Balance
as of 30 June 2019
|
|
|
(61
|
)
|
|
|
(6,284
|
)
|
|
|
(2,391
|
)
|
|
|
—
|
|
|
|
(8,736
|
)
|
|
Carrying
amount as of 30 June 2019
|
|
|
11
|
|
|
|
2,255
|
|
|
|
944
|
|
|
|
528
|
|
|
|
3,738
|
|
Curetis
did not own any of these assets under any lease programs in 2018 or 2019. All property, plant and equipment are free from any
rights held by third parties.
|
in
kEUR
|
|
Real
estate
|
|
IT-Equipment
|
|
car
fleet
|
|
Total
|
|
Cost:
|
|
|
|
|
|
|
|
|
|
Balance as of 1
January 2018
|
|
-
|
|
-
|
|
-
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
Cost:
|
|
|
|
|
|
|
|
|
|
Initial
recognition 01.01.2019
|
|
|
1,450
|
|
|
|
5
|
|
|
|
39
|
|
|
|
1,494
|
|
|
Additions
|
|
|
—
|
|
|
|
—
|
|
|
|
22
|
|
|
|
22
|
|
|
Balance
as of 30 June 2019
|
|
|
1,450
|
|
|
|
5
|
|
|
|
61
|
|
|
|
1,516
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated
amortization:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance
as of 1 January 2019
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Amortization:
|
|
|
(207
|
)
|
|
|
(2
|
)
|
|
|
(9
|
)
|
|
|
(218
|
)
|
|
Balance
as of 30 June 2019
|
|
|
(207
|
)
|
|
|
(2
|
)
|
|
|
(9
|
)
|
|
|
(218
|
)
|
|
Carrying
amount as of 30 June 2019
|
|
|
1,243
|
|
|
|
3
|
|
|
|
52
|
|
|
|
1,298
|
|
Refer
to Note 3.1 for additional information on right-of-use assets.
|
|
12.
|
FINANCIAL
LIABILITIES
|
Current financial liabilities
consist of the Convertible notes that were issued to Yorkville Advisors (Yorkville) on 02 October 2018 by Curetis N.V. Under the
first tranche, 500 notes are available for issuance, of which 350 notes were issued as of 02 October 2018 (subscription date)
and 150 notes were issued in June 2019. Each note has a nominal value of kEUR 10 and a maturity of one year. The notes were issued
at an 8% discount, due to a 4% commitment fee and a 4% subscription fee. The Company incurred kEUR 120 in issuance costs related
to due diligence and legal fees.
The holders of the outstanding
notes have the right to convert the notes in exchange for shares of Curetis N.V. at any time. The number of shares to be issued
upon conversion of a note is determined by the nominal amount of the note divided by 93% of the last 10-day lowest VWAP (volume
weighted average price) of a common share of Curetis N.V. on the conversion date. As of 30 June 2019, 198 notes had been converted
to shares of Curetis N.V. and subsequent to that date an additional 152 notes have been converted.
In connection with the proposed
acquisition of Curetis by OpGen, it is expected that the remaining notes are expected to be converted into shares of OpGen, rather
than of shares of Curetis N.V., subject to the consent of Yorkville. Curetis assumes that all notes will be converted within the
original maturity of one year from the date of issuance and that the likelihood of executing the extension option is remote.
In June 2019 Curetis has drawn down
a third tranche of EUR 5 million from the EIB (European Investment Bank). In line with all prior tranches, the majority of interest
is also deferred into the bullet repayment structure upon maturity. In return for EIB waiving the condition precedent of a minimum
cumulative equity capital raised of EUR 15 million to disburse this EUR 5 million tranche, the parties have agreed on a 2.1% participation
percentage interest (PPI). Upon maturity of the tranche, i.e. not before around mid-2024 (and no later than mid-2025), EIB will
be entitled to an additional payment that is equity-linked and equivalent to 2.1% of the then total valuation of Curetis. This
right constitutes an embedded derivative, which is separated and measured at fair value with changes being accounted for through
profit or loss.
Other non-current financial liabilities
comprise the EIB debt facility and the deferred taxes, calculated with the effective interest method. The effective interest rate
applied by the Company is 9.12% for the EUR 10 million tranche and 9.01% for the EUR 3 million tranche. For the EUR 5 million
tranche an effective interest rate of 9.01% is applied.
|
in kEUR
|
|
30
June 2019
|
|
31
December 2018
|
|
|
|
current
|
|
non-current
|
|
current
|
|
non-current
|
|
Loan
from EIB
|
|
|
—
|
|
|
|
18,000
|
|
|
|
—
|
|
|
|
13,000
|
|
|
Embedded
derivative
|
|
|
—
|
|
|
|
301
|
|
|
|
—
|
|
|
|
—
|
|
|
Accrued
interest
|
|
|
88
|
|
|
|
1,322
|
|
|
|
343
|
|
|
|
949
|
|
|
Total
|
|
|
88
|
|
|
|
19,623
|
|
|
|
343
|
|
|
|
13,949
|
|
|
|
13.
|
FINANCIAL
INSTRUMENTS
|
For
each class of financial instrument the fair value of financial assets and liabilities, together with their carrying amounts contained
in the condensed combined financial statements are shown in the following schedules.
|
in kEUR
|
|
30
June 2019
|
|
31
December 2018
|
|
|
|
Category
in accordance with IFRS9
|
|
Carrying
amount
|
|
Fair
Value
|
|
Fair
Value Level
|
|
Category
in accordance with IAS 39
|
|
Carrying
amount
|
|
Fair
Value
|
|
Fair
Value Level
|
|
Current
Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash
and Cash Equivalents
|
|
AC
|
|
|
4,779
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
AC
|
|
|
4,800
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
Trade
Receivables
|
|
AC
|
|
|
196
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
AC
|
|
|
323
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
Non-current
Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other
non-current financial assets
|
|
AC
|
|
|
158
|
|
|
|
158
|
|
|
|
2
|
|
|
AC
|
|
|
158
|
|
|
|
158
|
|
|
|
2
|
|
n/a*:
For short-term financial instruments a fair value disclosure is not required as the carrying amount approximates the fair value.
|
in
kEUR
|
|
30
June 2019
|
|
31
December 2018
|
|
|
|
Category
in accordance with IFRS9
|
|
Carrying
amount
|
|
Fair
Value
|
|
Fair
Value Level
|
|
Category
in accordance with IAS 39
|
|
Carrying
amount
|
|
Fair
Value
|
|
Fair
Value Level
|
|
Current
Liabilities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Trade
and other Payables
|
|
FLAC
|
|
|
731
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
FLAC
|
|
|
921
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
Other
current financial liabilities
|
|
FLAC
|
|
|
2,776
|
(1)
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
FLAC
|
|
|
3,154
|
|
|
|
n/a
*
|
|
|
|
n/a
|
|
|
Other
current financial liabilities
|
|
FVTPL
|
|
|
491
|
(2)
|
|
|
491
|
|
|
|
3
|
|
|
FVTPL
|
|
|
543
|
|
|
|
543
|
|
|
|
3
|
|
|
Non-current
Liabilities
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other
non-current financial liabilities
|
|
FLAC
|
|
|
19,623
|
|
|
|
19,084
|
|
|
|
2
|
|
|
FLAC
|
|
|
13,949
|
|
|
|
13,546
|
|
|
|
2
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
n/a*
= For short-term financial Instruments a fair value disclosure is not required as the carrying amount approximates the fair value.
|
|
(1)
|
Consists
of liabilities for outstanding invoices, Convertible notes and provision for deferred
interest.
|
|
|
(2)
|
Consists
of conversion rights related to Convertible notes
|
During
the six months ended 30 June 2019, there were no reclassifications of financial assets or financial liabilities between the classes.
The
fair value is the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between
market participants at the measurement date.
The
fair value of the embedded derivative separated from the third tranche of the EIB loan was determined using observable inputs
(Curetis N.V. share price, own credit spread) and assumptions for not observable inputs (exercise of conversion rights regarding
the convertible notes; date of requesting for PPI payment within a 12 months period from about the due date of the third tranche
of the EIB loan). These assumptions lead to the inclusion of the fair value within level 3 of the fair value hierarchy. The fair
value of the compound embedded derivative separated from the convertible note is determined using observable inputs (Curetis N.V.
share price, own credit spread) and assumptions about the rational economic behavior of the related parties which are not observable
input parameters. These assumptions lead to the inclusion of the fair value within level 3 of the fair value hierarchy.
Secured
liabilities and assets pledged as security
Curetis
has pledged cash on bank accounts as rent deposit for lease agreements with a total value of kEUR 64 and for credit card deposits
and bank guarantees with a total value of kEUR 94.
The
Executives and Supervisory Board of Curetis GmbH as well as certain employees of the Group are included in the Employee Stock
Option Plan 2016 “ESOP” of Curetis N.V.. The expenses associated with these individuals are recognized by the Group,
which employ and benefit from the employment of the individuals holding notional stocks in Curetis N.V..
Capital
reserve increase corresponding to the expenses accounted for the share-based payment of the ESOP 2016.
The
following table illustrates the number and exercise prices of the movements in employee stock options during the year, as well
as the grant date and the remaining term of the option (Note, valuation inputs; stock price, dividend yields, volatility etc.
are related to the stock of Curetis N.V.):
|
|
|
Tranche
1
|
|
Tranche
2
|
|
Tranche
3
|
|
Tranche
4
|
|
Tranche
5
|
|
Tranche
6
|
|
Grant
date
|
|
1
July 2016
|
|
1
October 2016
|
|
1
January 2017
|
|
1
April 2017
|
|
1
July 2017
|
|
1
October 2017
|
|
Granted
stock options
|
|
|
170,000
|
|
|
|
45,000
|
|
|
|
42,500
|
|
|
|
5,000
|
|
|
|
20,000
|
|
|
|
123,500
|
|
|
Remaining
contractual term of the option
|
|
|
7.00
years
|
|
|
|
7.25
years
|
|
|
|
7.50
years
|
|
|
|
7.75
years
|
|
|
|
8.00
years
|
|
|
|
8.25
years
|
|
|
Exercise price
|
|
|
6.45
Euro
|
|
|
|
6.41
Euro
|
|
|
|
6.42
Euro
|
|
|
|
5.81
Euro
|
|
|
|
4.93
Euro
|
|
|
|
4.98
Euro
|
|
|
Outstanding at 1
January 2019
|
|
|
132,778
|
|
|
|
22,500
|
|
|
|
41,458
|
|
|
|
5,000
|
|
|
|
7,778
|
|
|
|
106,833
|
|
|
Granted
during the year
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Forfeited
during the year
|
|
|
833
|
|
|
|
—
|
|
|
|
6,667
|
|
|
|
—
|
|
|
|
—
|
|
|
|
14,583
|
|
|
Exercised
during the year
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Outstanding
at 30 June 2019
|
|
|
131,945
|
|
|
|
22,500
|
|
|
|
34,791
|
|
|
|
5,000
|
|
|
|
7,778
|
|
|
|
92,250
|
|
|
Exercisable
at 30 June 2019
|
|
|
131,945
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
Tranche
7
|
|
Tranche
8
|
|
Tranche
9
|
|
Tranche
10
|
|
Tranche
11
|
|
Grant
date
|
|
1
January 2018
|
|
1
March 2018
|
|
1
July 2018
|
|
1
October 2018
|
|
1
January 2019
|
|
Granted
stock options
|
|
|
25,000
|
|
|
|
102,00
|
|
|
|
40,500
|
|
|
|
110,000
|
|
|
|
322,000
|
|
|
Remaining
contractual term of the option
|
|
|
8.50
years
|
|
|
|
8.67
years
|
|
|
|
9.00
years
|
|
|
|
9.25
years
|
|
|
|
9.50
years
|
|
|
Exercise price
|
|
|
3.86
Euro
|
|
|
|
6.51
Euro
|
|
|
|
4.62
Euro
|
|
|
|
3.29
Euro
|
|
|
|
1.40
Euro
|
|
|
Outstanding at 1
January 2019
|
|
|
25,000
|
|
|
|
97,000
|
|
|
|
37,500
|
|
|
|
110,000
|
|
|
|
—
|
|
|
Granted
during the year
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
322,000
|
|
|
Forfeited
during the year
|
|
|
16,389
|
|
|
|
21,445
|
|
|
|
22,000
|
|
|
|
—
|
|
|
|
35,000
|
|
|
Exercised
during the year
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Outstanding
at 30 June 2019
|
|
|
8,611
|
|
|
|
75,555
|
|
|
|
15,500
|
|
|
|
110,000
|
|
|
|
287,000
|
|
|
Exercisable
at 30 June 2019
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
The
Annual General Shareholder meeting (“AGM”) on 27 June 2019 approved the granting of additional stock options under
the ESOP 2016 Plan to the members of the Management Board as well as Supervisory Board, respectively. These were granted effective
1 July 2019 and had the same terms as other grants under the ESOP 2016.
Valuation
model and input parameters
The
fair value of the stock options is measured using a binominal option pricing model taking into account the terms and conditions
upon which the options were granted. The following table lists the inputs to the model used for the options granted in the first
six months of 2019 at the measurement date (all other input parameters for grants prior to 2019 remain unchanged from the combined
financial statements as of 31 December 2018:
|
|
|
Tranche
11
|
|
Measurement
date1
|
|
1
January 2019
|
|
Expected
life of the option on the grant date (years)
|
|
|
5
|
|
|
Share
price on the measurement date (€)
|
|
|
1.5
|
|
|
Weighted
avg. exercise price
|
|
|
1.4
|
|
|
Expected
dividend yield (%)
|
|
|
0
|
|
|
Risk-free
interest rate (%)
|
|
|
-0.3
|
|
|
Expected
volatility of the share price (%)
|
|
|
64.25
|
|
|
Option
value (€)
|
|
|
0.81
|
|
1
The measurement date represents the acceptance date of the option
For
stock option valuation the possibility of early exercise was considered in the binomial model. Management determined an estimated
early exercise is expected five years after the date of grant of the options based on considered the following factors:
The
length of the vesting period has been considered since the share options cannot be exercised until the end of the 3-year vesting
period – i. e. the expected option life of 5 years is 2 years after the first possible exercise date.
The
Company does not have historical data points or experience from past option programs and to date no options have been exercised,
however, due to normal fluctuation as well as fluctuations triggered by the recent re-organization there have been multiple cases
of forfeited options. As a result, the Company does not have any actual data available regarding the average length of time that
similar options have remained outstanding in the past or if the employee´s level within the Company will impact the timing
of exercise.
The
risk-free interest rate is the implied yield currently available on German government issued bonds with a remaining term equal
to the term of the options.
The
future volatility for the lives of the options was estimated based on historical volatility of peer group companies.
The
expense recognized during the six months ended 30 June 2019 and the six months ended 30 June 2018 is shown in the following table:
|
in kEUR
|
|
30
June 2019
|
|
30
June 2018
|
|
Expense
arising from equity-settled share-based payment transactions
|
|
|
|
|
|
|
|
|
|
Cost
of sales
|
|
|
6
|
|
|
|
—
|
|
|
Distribution
costs
|
|
|
132
|
|
|
|
122
|
|
|
Administrative
expenses
|
|
|
51
|
|
|
|
62
|
|
|
Research
& development expenses
|
|
|
59
|
|
|
|
67
|
|
|
Total
|
|
|
248
|
|
|
|
251
|
|
The
Group does not consider paying dividends as long as the result from operating activities in the condensed combined statement of
operations and other comprehensive loss and the cash flows from operating activities are negative.
The
group leases various cars, buildings and IT equipment. Rental contracts are typically made for fixed periods of three to five
years but may have extension options as described below. Lease terms are negotiated on an individual basis and contain a wide
range of different terms and conditions. The lease agreements do not impose any covenants, but leased assets may not be used as
security for borrowing purposes.
Extension
and termination options are included in a number of property and equipment leases across the group. These terms are used to maximize
operational flexibility in terms of managing contracts. The majority of extension and termination options held are exercisable
only by the group and not by the respective lessor. No lease payments made in 2019 were optional.
The
following table shows the maturity analysis of lease liabilities as of 30 June 2019.
|
Contractual
Maturities of Financial Liabilities
|
|
Up
to One Year
|
|
Between
One and Five Years
|
|
More
than Five Years
|
|
Total
Contractual Cash Flows
|
|
Carrying
Amount Liabilities
|
|
Lease
Liabilities
|
|
|
454
|
|
|
|
816
|
|
|
|
73
|
|
|
|
1,343
|
|
|
|
1,305
|
|
Refer
to Note 3.1 for additional information on lease liabilities.
The Company has reflected transactions
with the parent company, Curetis N.V., as related party balances within the statement of operations and other comprehensive loss
and the combined statement of financial position. Other related party transactions have been included below.
Curetis N.V. charges certain management fees for services rendered by the senior management of Curetis N.V. to Curetis GmbH and
its subsidiaries resulting in other expenses from related parties. The transactions are charged at cost. Curetis N.V. is the controlling
company for VAT purposes and receives VAT amounts due to Curetis GmbH as a controlled company, resulting in Other receivables,
related party.
Curetis has entered into arrangements
with a number of its subsidiaries, the financial impacts of which are eliminated in combination. Curetis considers transactions
with key management personnel to be related party transactions. Any transactions with such individuals are also recorded in related
party accounts.
During the six months ended 2019
and 2018, the Curetis Business received shareholder contributions from Curetis N.V. of kEUR 3,000 and kEUR 10,000 of which kEUR
1,627 are presented as proceeds from current liabilities, net of issuance costs as that amount relates to the convertible notes
that were issued by Curetis N.V. and legally contributed to the capital reserve of Curetis GmbH. For presentation purposes (See
Note 2), that amount was presented as if the Curetis Business has issued such convertible notes.
|
|
17.
|
EVENTS
AFTER THE REPORTING DATE
|
Subsequent
to 30 June 2019:
|
|
·
|
Curetis
elected share settlement for excess entitlement under first tranche of Yorkville convertible
notes
|
|
|
·
|
Curetis
GmbH entered into a definitive agreement to combine its business with OpGen. See Note
2.1 for further information.
|
|
Holzgerlingen,
15 September 2019
Curetis GmbH
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
/s/
Oliver Schacht, PhD
|
|
/s/
Johannes Bacher
|
|
Oliver
Schacht, PhD
Chief
Executive Officer (CEO)
Managing
Director
|
|
Johannes
Bacher
Chief
Operating Officer (COO)
Managing
Director
|
|
|
|
|
|
|
|
|
|
|
|
|
|
/s/Achim
Plum
|
|
/s/
Heiko Schorr
|
|
Dr.
Achim Plum
Chief
Business Officer (CBO)
Managing
Director
|
|
Heiko Schorr
Director
of Finance
Managing
Director
|

Up to 1,526,717 Units (each Unit
contains One Share of Common Stock and One Common Warrant to purchase One Share of Common Stock)
Up to 1,526,717 Pre-funded Units
(each Pre-funded Unit contains One Pre-funded Warrant to Purchase One Share of Common Stock and One Common Warrant to purchase
One Share of Common Stock)
Shares of Common Stock Underlying the Pre-funded Warrants and
Shares of Common Stock Underlying the Common Warrants
H.C.
Wainwright & Co.
,
2019
PART
II
Information
Not Required in Prospectus
Item 13. Other
Expenses of Issuance and Distribution.
The following table sets forth the fees and expenses,
other than underwriting fees and expenses, payable in connection with the registration of the securities hereunder. All amounts
are estimates except the SEC registration fee and the FINRA filing fee.
|
SEC registration fee
|
|
$
|
4,380.43
|
|
|
Legal fees and expenses
|
|
$
|
145,000.00
|
|
|
Accounting fees and expenses
|
|
$
|
99,500.00
|
|
|
FINRA filing fee
|
|
$
|
5,562.00
|
|
|
Transfer agent and registrar fees and expenses
|
|
$
|
500.00
|
|
|
Printer costs and expenses
|
|
$
|
1,057.57
|
|
|
|
|
$
|
256,000.00
|
|
Item 14. Indemnification
of Directors and Officers.
The Registrant maintains insurance
providing for indemnification of its officers and directors and certain other persons against liabilities and expenses incurred
by any of them in certain stated proceedings and under certain stated conditions.
Delaware Corporations
Section 145 of the Delaware
General Corporation Law, or DGCL, provides that a corporation may indemnify any person who was or is a party or is threatened
to be made a party to any threatened, pending or completed action, suit or proceeding whether civil, criminal, administrative
or investigative (other than an action by or in the right of the corporation) by reason of the fact that the person is or was
a director, officer, employee or agent of the corporation, or is or was serving at the request of the corporation as a director,
officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise, against expenses (including
attorneys’ fees), judgments, fines and amounts paid in settlement actually and reasonably incurred by the person in connection
with such action, suit or proceeding if the person acted in good faith and in a manner the person reasonably believed to be in
or not opposed to the best interests of the corporation, and, with respect to any criminal action or proceeding, had no reasonable
cause to believe his conduct was unlawful. Section 145 further provides that a corporation similarly may indemnify any such person
serving in any such capacity who was or is a party or is threatened to be made a party to any threatened, pending or completed
action or suit by or in the right of the corporation to procure a judgment in its favor by reason of the fact that the person
is or was a director, officer, employee or agent of the corporation or is or was serving at the request of the corporation as
a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise, against
expenses (including attorneys’ fees) actually and reasonably incurred by the person in connection with the defense or settlement
of such action or suit if the person acted in good faith and in a manner the person reasonably believed to be in or not opposed
to the best interests of the corporation and except that no indemnification shall be made in respect of any claim, issue or matter
as to which such person shall have been adjudged to be liable to the corporation unless and only to the extent that the Delaware
Court of Chancery or such other court in which such action or suit was brought shall determine upon application that, despite
the adjudication of liability but in view of all of the circumstances of the case, such person is fairly and reasonably entitled
to indemnity for such expenses which the Delaware Court of Chancery or such other court shall deem proper.
Certificate of
Incorporation and Bylaws
The Registrant’s certificate
of incorporation, as amended, provides that a director of the Registrant shall not be personally liable to the Registrant or its
stockholders for monetary damages for breach of fiduciary duty as a director, except for liability (i) for any breach of the director’s
duty of loyalty to the Registrant or its stockholders, (ii) for acts or omissions not in good faith or which involve intentional
misconduct or a knowing violation of law, (iii) under Section 174 of the DGCL, or (iv) for any transaction from which the director
derived an improper personal benefit. If the DGCL is hereafter amended to authorize the further elimination or limitation of the
liability of directors, then the liability of the directors of the Registrant, in addition to the limitation on personal liability
provided herein, shall be limited to the fullest extent permitted by the amended DGCL. Any repeal or modification of this paragraph
by the stockholders of the Registrant shall be prospective only, and shall not adversely affect any limitation on the personal
liability of a director of the corporation at the time of such repeal or modification. The Registrant’s certificate of incorporation
further provides that the Registrant’s officers and directors shall be indemnified by the Registrant as provided in the
Registrant’s bylaws.
Under the provisions of the
Registrant’s bylaws, as amended, any person who is or was a party or is threatened to be made a party of any threatened,
pending or completed action, suit or proceeding, whether civil, criminal, administrative or investigative (other than an action
by or in the right of the Registrant) by reason of the fact that he or she is or was a director, officer, employee or agent of
the Registrant or is or was serving at the Registrant’s request as a director, officer, employee or agent of another company
or other entity shall be indemnified by the Registrant against expenses (including attorneys’ fees), judgments, fines and
amounts paid in settlement actually and reasonably incurred by him or her in connection with such action, suit or proceeding if
he or she acted in good faith and in a manner he or she reasonably believed to be in, or not opposed to, the Registrant’s
best interests, and, with respect to any criminal action or proceeding, had no reasonable cause to believe his or her conduct
was unlawful. The Registrant shall further indemnify any person who was or is a party or is threatened to be made a party to any
threatened, pending or completed action or suit by or in the right of the Registrant to procure a judgment in its favor by reason
of the fact that he is or was a director, officer, employee or agent of the Registrant or is or was serving at the request of
the Registrant as a director, officer, employee or agent of another corporation or other entity against expenses (including attorneys’
fees) actually and reasonably incurred by him in connection with the defense or settlement of such action or suit if he acted
in good faith and in a manner he reasonably believed to be in or not opposed to the best interests of the Registrant. Notwithstanding
the foregoing, no indemnification shall be made in respect of any claim, issue or matter as to which such person shall have been
adjudged to be liable to the Registrant unless and only to the extent that the Delaware Court of Chancery or the court in which
such action or suit was brought shall determine upon application that, despite the adjudication of liability but in view of all
the circumstances of the case, such person is fairly and reasonably entitled to indemnity for such expenses which the Delaware
Court of Chancery or such other court shall deem proper.
The termination of any action,
suit or proceeding by judgment, order, settlement, conviction, or upon a plea of nolo contendre or its equivalent, shall not,
of itself, create a presumption that the person did not act in good faith and in a manner which he or she reasonably believed
to be in, or not opposed to, the Registrant’s best interests and, with respect to any criminal action or proceeding, had
reasonable cause to believe that his or her conduct was unlawful.
In addition, to the extent
that such a person is successful on the merits or otherwise in defense of any action, suit, or proceeding brought against him
or her by reason of the fact that he or she is the Registrant’s director, officer, employee or agent, he or she shall be
indemnified against expenses, including attorneys’ fees, actually and reasonably incurred in connection therewith.
The Registrant’s bylaws,
as amended, provide that expenses (including attorneys’ fees) incurred by a director or officer in defending a civil, criminal,
administrative, or investigative action, suit or proceeding may be paid by the Registrant in advance of the final disposition
of such action, suit or proceeding upon receipt of an undertaking by or on behalf of the director or officer to repay such amount
if it shall ultimately be determined that he or she is not entitled to be indemnified by the Registrant. Such expenses, including
attorneys’ fees, incurred by other employees and agents may be paid upon such terms and conditions as the Board of Directors
deems appropriate.
Any indemnification under
the provisions summarized above (unless ordered by a court) shall be made by the Registrant only as authorized in each specific
case upon a determination that indemnification of such person is proper under the circumstances because he or she has met the
applicable standard of conduct set forth in the applicable provision. Such determination shall be made (1) by a majority vote
of the Registrant’s directors who are not parties to the action, suit or proceeding (even though less than a quorum), (2)
if there are no such directors, or if such directors so direct, by independent legal counsel in a written opinion, or (3) by the
stockholders.
The Registrant maintains director
and officer insurance with respect to those claims described above in customary amounts.
The above discussion of the
certificate of incorporation and bylaws of the Registrant and the DGCL is not intended to be exhaustive and is qualified in its
entirety by such certificates of incorporation, bylaws and the DGCL.
The Registrant has entered
into indemnification agreements with each of our directors and executive officers. These agreements provide that we will indemnify
each of our directors, such executive officers and, at times, their affiliates to the fullest extent permitted by Delaware law.
We will advance expenses, including attorneys’ fees (but excluding judgments, fines and settlement amounts), to each indemnified
director, executive officer or affiliate in connection with any proceeding in which indemnification is available and we will indemnify
our directors and officers for any action or proceeding arising out of that person’s services as a director or officer brought
on behalf of us and/or in furtherance of our rights. Additionally, each of our directors may have certain rights to indemnification,
advancement of expenses and/or insurance provided by their affiliates, which indemnification relates to and might apply to the
same proceedings arising out of such director’s services as a director referenced herein. Nonetheless, we have agreed in
the indemnification agreements that our obligations to those same directors are primary and any obligation of the affiliates of
those directors to advance expenses or to provide indemnification for the expenses or liabilities incurred by those directors
are secondary.
We also maintain general liability
insurance which covers certain liabilities of our directors and officers arising out of claims based on acts or omissions in their
capacities as directors or officers, including liabilities under the Securities Act.
Item 15. Recent
Sales of Unregistered Securities.
The following list sets forth
information as to all securities we have sold since July 1, 2016, which were not registered under the Securities Act.
(1) On
May 31, 2017, the Company entered into a Note Purchase Agreement with jVen Capital, which Note Purchase Agreement was amended
and restated on July 10, 2017, under which jVen Capital agreed to lend bridge financing in an aggregate principal amount of up
to $1,500,000 to the Company in the form of three $500,000 secured convertible promissory notes. Pursuant to the terms of the
amended and restated Note Purchase Agreement and the underlying bridge financing notes, if a “qualified financing”
(offering of debt or equity securities with net proceeds of $5 million or more to the Company) occurs, the Company has the obligation
to repay the outstanding bridge financing notes within five business days after the closing of the qualified financing. If a qualified
financing does not occur, and an event of default occurs (the Company fails to pay any outstanding bridge financing notes when
due, seeks to liquidate the Company, enters into a bankruptcy proceeding, or fails to pay other indebtedness when due), the then-outstanding
bridge financing notes would acquire a two times liquidation preference for unpaid principal and interest. Alternatively, jVen
Capital may forego the two times liquidation preference and elect to convert the bridge financing notes to Series B Convertible
Preferred Stock, or Preferred Stock, at a ratio of one share of non-voting Preferred Stock per $1.00 of principal and interest,
which non-voting Preferred Stock would be convertible into 10 shares of voting common stock, otherwise no further preferences
are provided to the Preferred Stock. The bridge financing notes contain, and the Certificate of Designation for the Preferred
Stock will contain, a blocker provision that prevents any issuance of securities as repayment of the bridge financing notes, or
any conversion of the Preferred Stock into common stock if such conversion would violate Nasdaq Listing Rule 5635.
(2) In
connection with the bridge financing transaction described in (1) above, the Company issued warrants to purchase 11,983 shares
of common stock to jVen Capital, and warrants to purchase 13,119 shares of common stock to MGHIF.
(3) In
September 2017, the Company issued common stock with an aggregate value of $110,000 to settle a dispute related to pre-Merger
AdvanDx activities.
(4) On
June 11, 2018, the Company executed an Allonge to its Second Amended and Restated Senior Secured Promissory Note, dated June 28,
2017, with a principal amount of $1,000,000 issued to MGHIF. The Allonge provided that accrued and unpaid interest of $285,512
due as of July 14, 2018, the original maturity date, would be paid through the issuance of shares of OpGen’s common stock
in a private placement transaction. In addition, the Allonge revised and extended the maturity date for payment of the Note to
six semi-annual payments of $166,667 plus accrued and unpaid interest beginning on January 2, 2019 and ending on July 1, 2021.
On July 30, 2018, the Company issued 144,238 shares of common stock to MGHIF in a private placement transaction for $285,512 of
accrued and unpaid interest due as of July 14, 2018 under the MGHIF Note.
As of October 9, 2019,
none of the warrants issued in these transactions have been exercised.
We deemed the offers, sales
and issuances of the securities described in paragraphs (1) through (4) above to be exempt from registration under the Securities
Act, in reliance on Section 4(a)(2) of the Securities Act, including Regulation D and Rule 506 promulgated thereunder, regarding
transactions by an issuer not involving a public offering. All purchasers of securities in transactions exempt from registration
pursuant to Regulation D represented to us that they were accredited investors and were acquiring the shares for investment purposes
only and not with a view to, or for sale in connection with, any distribution thereof and that they could bear the risks of the
investment and could hold the securities for an indefinite period of time. The purchasers received written disclosures that the
securities had not been registered under the Securities Act and that any resale must be made pursuant to a registration statement
or an available exemption from such registration.
All certificates representing
the securities issued in the transactions described in this Item 15 included appropriate legends setting forth that the securities
had not been offered or sold pursuant to a registration statement and describing the applicable restrictions on transfer of the
securities. There were no underwriters employed in connection with any of the transactions set forth in this Item 15.
Item 16. Exhibits
and Financial Statement Schedules.
(a) Exhibits:
EXHIBIT INDEX
|
Exhibit
Number
|
|
Description
|
|
|
|
|
|
1.1
|
|
Form of Underwriting Agreement (incorporated by reference
to Exhibit 1.1 to the Registrant’s Registration Statement on Form S-1, Amendment No. 1, File No. 333-233775, filed on
October 15, 2019)
|
|
|
|
|
|
2.1
|
|
Implementation
Agreement, dated as of September 4, 2019, by and among Curetis N.V., as Seller, and Crystal GmbH, as Purchaser and OpGen,
Inc. (incorporated by reference to Exhibit 2.1 to the Registrant’s Current Report on Form 8-K, filed on September 4,
2019) ^
|
|
|
|
|
|
3.1
|
|
Amended
and Restated Certificate of Incorporation of the Registrant (incorporated by reference to Exhibit 3.1 of the Registrant’s
Current Report on Form 8-K, File No. 001-37367, filed on May 13, 2015) ^
|
|
|
|
|
|
3.2
|
|
Certificate
of Correction to Amended and Restated Certificate of Incorporation of the Registrant, dated June 6, 2016 (incorporated by
reference to Exhibit 3.1 of the Registrant’s Current Report on Form 8-K, filed on June 6, 2016) ^
|
|
|
|
|
|
3.3
|
|
Certificate
of Amendment to the Amended and Restated Certificate of Incorporation of the Registrant dated and filed with the Delaware
Secretary of State on January 17, 2018 (incorporated by reference to Exhibit 3.1 to the Registrant’s Current Report
on Form 8-K filed on January 17, 2018) ^
|
|
|
|
|
|
3.4
|
|
Certificate
of Amendment to the Amended and Restated Certificate of Incorporation, as amended, of the Registrant dated and filed with
the Delaware Secretary of State on August 28, 2019 (incorporated by reference to Exhibit 3.1 to the Registrant’s Current
Report on Form 8-K filed on August 28, 2019) ^
|
|
|
|
|
|
3.5
|
|
Amended
and Restated Bylaws of the Registrant (incorporated by reference to Exhibit 3.2 to the Registrant’s Form S-1, File No.
333-202478, filed on March 3, 2015)
|
|
|
|
|
|
4.1
|
|
Form
of Common Stock Certificate of the Registrant (incorporated by reference to Exhibit 4.1 to the Registrant’s Annual Report
on Form 10-K for the year ended December 31, 2018, filed on February 27, 2019) ^
|
|
|
|
|
|
4.2
|
|
Form
of Offered Warrant to Purchase Common Stock (IPO Warrant) (incorporated by reference to Exhibit 4.8 of the Registrant’s
Form S-1/A, File No. 333-202478, filed on March 20, 2015)
|
|
|
|
|
|
4.3
|
|
Form
of Warrant to Purchase Common Stock (issued to jVen Capital, LLC and Merck Global Health Innovation Fund) (incorporated by
reference to Exhibit 4.1 to the Registrant’s Current Report on Form 8-K Amendment No. 2, filed on July 10, 2017) ^
|
|
|
|
|
|
4.4
|
|
Form
of Common Stock Purchase Warrant for February 2018 Public Offering (incorporated by reference to Exhibit 4.3 to the Registrant’s
Form S-1/A, File No. 333-222140, filed on January 31, 2018)
|
|
|
|
|
|
4.5
|
|
Form
of Pre-Funded Common Stock Purchase Warrant for February 2018 Public Offering (incorporated by reference to Exhibit 4.4 to
the Registrant’s Form S-1/A, File No. 333-222140, filed on January 31, 2018)
|
|
|
|
|
|
4.6
|
|
Form
of Placement Agent Warrant for February 2018 Public Offering (incorporated by reference to Exhibit 4.5 to the Registrant’s
Form S-1/A, File No. 333-222140, filed on January 31, 2018)
|
|
|
|
|
|
4.7
|
|
Form
of Common Stock Purchase Warrant for July 2017 Public Offering (incorporated by reference to Exhibit 4.4 to the Registrant’s
Form S-1, Amendment No. 2, File No. 333-218392, filed on July 11, 2017)
|
|
|
|
|
|
4.8
|
|
Form
of Placement Agent Warrant for July 2017 Public Offering (incorporated by reference to Exhibit 4.5 to the Registrant’s
Form S-1, File No. 333-218392, filed on July 11, 2017)
|
|
|
|
|
|
4.9
|
|
Form of Pre-funded Warrant (incorporated by reference to
Exhibit 4.9 to the Registrant's Registration Statement on Form S-1, Amendment No. 1, File No. 333-233775, filed on October
15, 2019)
|
|
|
|
|
|
4.10
|
|
Form of Underwriter’s Warrant (incorporated by reference
to Exhibit 4.10 to the Registrant’s Registration Statement on Form S-1, Amendment No. 1, File No. 333-233775, filed
on October 15, 2019)
|
|
|
|
|
|
4.11
|
|
Form of Common Stock Purchase Warrant (incorporated by reference
to Exhibit 4.11 to the Registrant’s Registration Statement on Form S-1, Amendment No. 1, File No. 333-233775, filed
on October 15, 2019)
|
|
|
|
|
|
5.1
|
|
Opinion of Ballard Spahr LLP (incorporated by reference to Exhibit 5.1 to the Registrant's Registration Statement on Form S-1, Amendment No. 1, File No. 333-233775, filed on October 15, 2019)
|
|
|
|
|
|
10.1
|
|
Lease
Agreement, dated as of June 30, 2008, between the Registrant and ARE-708 Quince Orchard, LLC (the “Landlord”)
(incorporated by reference to Exhibit 10.1 of Form S-1/A, file No. 333-202478, filed March 3, 2015)
|
|
|
|
|
|
10.1.1
|
|
First
Amendment to Lease, dated as of April 4, 2011, between the Registrant and the Landlord (incorporated by reference to Exhibit
10.1.1 of Form S-1, File No. 333-202478, filed March 3, 2015)
|
|
|
|
|
|
10.1.2
|
|
Second
Amendment to Lease Agreement, dated as of August 15, 2012, between the Registrant and the Landlord (incorporated by reference
to Exhibit 10.1.2 of Form S-1, File No. 333-202478, filed March 3, 2015)
|
|
|
|
|
|
10.1.3
|
|
Third
Amendment to Lease, dated as of December 30, 2013, between the Registrant and the Landlord (incorporated by reference to Exhibit
10.1.3 of Form S-1, File No. 333-202478, filed March 3, 2015)
|
|
|
|
|
|
10.1.4
|
|
Fourth
Amendment to Lease Agreement, dated as of March 21, 2014, between the Registrant and the Landlord (incorporated by reference
to Exhibit 10.4 of Form S-1, File No. 333-202478, filed March 3, 2015)
|
|
|
|
|
|
10.1.5
|
|
Fifth
Amendment to Lease Agreement, dated as of March 20, 2015, between the Registrant and the Landlord (incorporated by reference
to Exhibit 10.1.5 of Form S-1, Amendment No. 1, File No. 333-202478, filed on March 20, 2015)
|
|
|
|
|
|
10.1.6
|
|
Sixth
Amendment to Lease Agreement (and Amendment to Reimbursement Agreement), dated as of April 30, 2015, between the Registrant
and the Landlord (incorporated by reference to Exhibit 10.1.6 of Form S-1, Amendment No. 8, File No. 333-202478, filed on
May 1, 2015)
|
|
|
|
|
|
10.1.7
|
|
Seventh
Amendment to Lease Agreement, dated as of June 30, 2015, between the Registrant and the Landlord (incorporated by reference
to Exhibit 10.1 of Current Report on Form 8-K, filed on July 7, 2015) ^
|
|
|
|
|
|
10.1.8
|
|
Eighth
Amendment to Lease Agreement, dated September 8, 2015, between the Registrant and the Landlord (incorporated by reference
to Exhibit 10.6 of Quarterly Report on Form 10-Q, filed on November 13, 2015) ^
|
|
|
|
|
|
10.2
|
|
Lease
Extension #6, dated October 14, 2016, by and between the Registrant and Cummings Properties, LLC (related to AdvanDx facility)
(incorporated by reference to Exhibit 10.2 of Quarterly Report on Form 10-Q, filed November 14, 2016) ^
|
|
|
|
|
|
10.3
|
|
Form
of Indemnification Agreement between the Registrant and each of its directors and executive officers (incorporated by reference
to Exhibit 10.2 of Form S-1, File No. 333-202478, filed on March 3, 2015)
|
|
|
|
|
|
10.4
|
|
2015
Equity Incentive Plan, as amended and restated on March 29, 2018 (incorporated by reference to Exhibit 10.4 to the Registrant’s
Annual Report on Form 10-K for the year ended December 31, 2017, filed on March 29, 2018) ^
|
|
|
|
|
|
10.5 !
|
|
Non-Employee Director Compensation Policy (incorporated by
reference to the Registrant’s Registration Statement on Form S-1, SEC File No. 333-233775, filed on September 16, 2019)
|
|
|
|
|
|
10.6
|
|
Warrant
Agreement, dated as of May 8, 2015, between the Registrant and Philadelphia Stock Transfer, Inc., as warrant agent
(incorporated by reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K, filed on May 13, 2015) ^
|
|
|
|
|
|
10.7.1 !
|
|
Form
of Stock Option Agreement under the 2015 Equity Incentive Plan for employees and consultants (incorporated by reference to
Exhibit 10.9.1 to the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2016, filed on March 24,
2017) ^
|
|
|
|
|
|
10.7.2 !
|
|
Form
of Stock Option Agreement under the 2015 Equity Incentive Plan for non-employee directors (initial grant) (incorporated by
reference to Exhibit 10.9.2 to the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2016, filed
on March 24, 2017) ^
|
|
|
|
|
|
10.7.3 !
|
|
Form
of Stock Option Agreement under the 2015 Equity Incentive Plan for non-employee directors (annual grant) (incorporated by
reference to Exhibit 10.9.3 to the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2016, filed
on March 24, 2017) ^
|
|
|
|
|
|
10.8 !
|
|
Form
of Restricted Stock Unit Award Agreement under 2015 Equity Incentive Plan (incorporated by reference to Exhibit 10.10 to the
Registrant’s Annual Report on Form 10-K for the year ended December 31, 2016, filed March 24, 2017) ^
|
|
|
|
|
|
10.9
|
|
Common
Stock and Note Purchase Agreement, dated as of July 14, 2015, between the Registrant and Merck Global Health Innovation
Fund, LLC (incorporated by reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K, filed on July
16, 2015) ^
|
|
|
|
|
|
10.10
|
|
Senior
Secured Promissory Note, dated as of July 14, 2015, between the Registrant and Merck Global Health Innovation Fund, LLC
(incorporated by reference to Exhibit 10.2 to the Registrant’s Current Report on Form 8-K, filed on July 16, 2015) ^
|
|
|
|
|
|
10.10.1
|
|
Second
Amended & Restated Senior Secured Promissory Note, dated June 28, 2017, by and between the Registrant and Merck Global
Health Innovation Fund, LLC (incorporated by reference to Exhibit 10.3 to the Registrant’s Current Report on Form 8-K,
Amendment No. 1, filed on June 28, 2017) ^
|
|
|
|
|
|
10.10.2
|
|
Allonge,
dated June 11, 2018, to the Second Amended and Restated Senior Secured Promissory Note, dated June 28, 2017, with a principal
amount of $1,000,000 issued by OpGen, Inc. to Merck Global Health Innovation Fund, LLC (incorporated by reference to Exhibit
10.1 to the Registrant’s Current Report on Form 8-K, filed on June 11, 2018) ^
|
|
|
|
|
|
10.11
|
|
Third
Amended and Restated Investors’ Rights Agreement, dated as of December 18, 2013, among the Registrant and, certain investors
(registration rights provisions) (incorporated by reference to Exhibit 4.2 to the Registrant’s Form S-1, File No. 333-202478,
filed on March 3, 2015)
|
|
|
|
|
|
10.12
|
|
Registration
Rights Agreement, dated as of July 14, 2015, among the Registrant, Merck Global Health Innovation Fund, LLC, SLS Invest AB
and LD Pensions (incorporated by reference to Exhibit 10.3 to the Registrant’s Current Report on Form 8-K, filed on
July 16, 2015) ^
|
|
|
|
|
|
10.13
|
|
Letter
Agreement, dated July 12, 2015, between the Registrant and Fluidigm Corporation (incorporated by reference to Exhibit 10.5
to the Registrant’s Quarterly Report on Form 10-Q for the quarter ended June 30, 2015, filed on August 14, 2015) ^
|
|
|
|
|
|
10.14
|
|
Securities
Purchase Agreement, dated as of May 12, 2016, by and between the Registrant and the Purchasers party thereto (incorporated
by reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K, filed on May 17, 2016) ^
|
|
|
|
|
|
10.15
|
|
Amended
and Restated Securities Purchase Agreement, dated as of May 18, 2016, by and between the Registrant and the Purchasers party
thereto (incorporated by reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K, filed on May 20,
2016) ^
|
|
|
|
|
|
10.16 !
|
|
Stock
Option Award Agreement, dated April 28, 2016, by and between the Registrant and Evan Jones (incorporated by reference to
Exhibit 10.5 to the Registrant’s Quarterly Report on Form 10-Q for the quarter ended June 30, 2016, filed on August
11, 2016) ^
|
|
|
|
|
|
10.17
|
|
Common
Stock Sales Agreement, dated September 13, 2016, by and between the Registrant and Cowen and Company, LLC (incorporated by
reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K, filed on September 14, 2016) ^
|
|
|
|
|
|
10.18
|
|
Amended
& Restated Note Purchase Agreement, dated as of July 10, 2017, by and between the Registrant and jVen Capital, LLC
(incorporated by reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K, Amendment No. 2, filed
on July 10, 2017) ^
|
|
|
|
|
|
10.19
|
|
Form
of Secured Convertible Promissory Note #1 to be issued to jVen Capital, LLC (incorporated by reference to Exhibit 10.2 to
the Registrant’s Current Report on Form 8-K/A, filed on July 10, 2017) ^
|
|
|
|
|
|
10.20
|
|
Form
of Secured Promissory Note #2 and #3 to be issued to jVen Capital, LLC (incorporated by reference to Exhibit 10.3 to the Registrant’s
Current Report on Form 8-K/A, filed on July 10, 2017) ^
|
|
|
|
|
|
10.21
|
|
Amended
and Restated Registration Rights Agreement, dated as of June 6, 2017, by and between the Registrant and the Investors party
thereto (incorporated by reference to Exhibit 10.3 to the Registrant’s Current Report on Form 8-K, filed on June 6,
2017) ^
|
|
|
|
|
|
10.22±
|
|
Supply
Agreement, dated as of June 15, 2017, by and between the Registrant and Life Technologies Corporation (incorporated by reference
to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K, filed on June 19, 2017) ^
|
|
|
|
|
|
10.23
|
|
Securities
Purchase Agreement, dated as of July 12, 2017, among the Registrant and the purchasers signatory thereto, (incorporated by
reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K, filed on July 14, 2017) ^
|
|
|
|
|
|
10.24
|
|
Engagement
Letter with H.C Wainwright & Co., dated as of June 12, 2017 (incorporated by reference to Exhibit 1.2 to the Registrant’s
Registration Statement on Form S-1, Amendment No. 2, File No. 333-218392, filed on July 11, 2017)
|
|
|
|
|
|
10.25 !
|
|
Executive
Change In Control and Severance Benefits Agreement, dated September 24, 2018 between OpGen, Inc. and Evan Jones
(incorporated by reference to Exhibit 10.2 to the Registrant’s Current Report on Form 8-K, filed on September 25,
2018) ^
|
|
|
|
|
|
10.26 !
|
|
Executive
Change In Control and Severance Benefits Agreement, dated September 24, 2018 between OpGen, Inc. and Timothy C. Dec
(incorporated by reference to Exhibit 10.3 to the Registrant’s Current Report on Form 8-K, filed on September 25,
2018) ^
|
|
|
|
|
|
10.27 !
|
|
Executive
Change In Control and Severance Benefits Agreement, dated September 24, 2018 between OpGen, Inc. and Vadim Sapiro
(incorporated by reference to Exhibit 10.4 to the Registrant’s Current Report on Form 8-K, filed on September 25,
2018). ^
|
|
|
|
|
|
10.28
|
|
Form
of Securities Purchase Agreement between the Registrant and the purchasers signatory thereto (incorporated by reference to
Exhibit 10.1 to the Registrant’s Current Report on Form 8-K, filed on February 2, 2018) ^
|
|
|
|
|
|
10.29
|
|
Engagement
Letter, dated as of December 18, 2017, between the Registrant and H.C. Wainwright & Co., LLC (incorporated by reference
to Exhibit 1.2 to the Registrant’s Registration Statement on Form S-1, Amendment No. 1, File No. 333-222140, filed on
January 31, 2018)
|
|
|
|
|
|
10.30 !
|
|
OpGen,
Inc. Retention Plan for Executives (incorporated by reference to Exhibit 10.1 to the Registrant’s Current Report on
Form 8-K, filed on September 25, 2018) ^
|
|
|
|
|
|
21.1
|
|
Subsidiaries
of the Registrant (incorporated by reference to Exhibit 21.1 to the Registrant’s Annual Report on Form 10-K for the
year ended December 31, 2018, filed on February 27, 2019) ^
|
|
|
|
|
|
23.1 *
|
|
Consent of CohnReznick LLP
|
|
|
|
|
|
23.2 *
|
|
Consent of PricewaterhouseCoopers GmbH Wirtschaftsprüfungsgesellschaft
|
|
|
|
|
|
23.3
|
|
Consent of Ballard Spahr LLP (included in Exhibit 5.1) (incorporated
by reference to Exhibit 5.1 to the Registrant’s Registration Statement on Form S-1, Amendment No. 1, File No. 333-233775,
filed on October 15, 2019)
|
|
|
|
|
|
24.1
|
|
Power of attorney (included on signature page to the Registrant’s Registration Statement on Form S-1, SEC File No. 333-233775
filed on September 16, 2019)
|
|
|
|
|
|
99.1 *
|
|
Consent of Oliver Schacht, Ph.D.
|
|
|
|
|
|
99.2 *
|
|
Consent of William Rhodes
|
|
|
|
|
_____________
* Filed
herewith
! Denotes
management compensation plan or contract
±
Confidential treatment has been requested for certain portions of this agreement pursuant to an application for confidential
treatment filed with the Securities and Exchange Commission on June 19, 2017. Such provisions have been filed separately
with the Commission.
^ SEC
File No. 001-37367
(b) Financial
Statements Schedules:
Schedules have been omitted
because the information required to be set forth therein is not applicable or is shown in the financial statements or notes thereto.
Item 17. Undertakings.
The undersigned Registrant
hereby undertakes:
(1) To
file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement:
(i) To
include any prospectus required by Section 10(a)(3) of the Securities Act;
(ii) To
reflect in the prospectus any facts or events arising after the effective date of this registration statement (or the most recent
post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set
forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if
the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high
and of the estimated maximum offering range may be reflected in the form of prospectus filed with the Securities and Exchange
Commission (the “Commission”) pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent
no more than 20% change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee”
table in the effective registration statement; and
(iii) To
include any material information with respect to the plan of distribution not previously disclosed in this registration statement
or any material change to such information in this registration statement;
provided, however,
that paragraphs (1)(i), (1)(ii) and (1)(iii) above do not apply if the information required to be included in a post-effective
amendment by those paragraphs is contained in reports filed with or furnished to the Commission by the registrant pursuant to
Section 13 or Section 15(d) of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), that are incorporated
by reference in the registration statement, or is contained in a form of prospectus filed pursuant to Rule 424(b) that is part
of the registration statement.
(2) That,
for the purpose of determining any liability under the Securities Act, each such post-effective amendment shall be deemed to be
a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall
be deemed to be the initial bona fide offering thereof.
(3) To
remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at
the termination of the offering.
(4) That,
for the purpose of determining liability of the registrant under the Securities Act to any purchaser in the initial distribution
of the securities, in a primary offering of securities of the undersigned registrant pursuant to this registration statement,
regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold to such
purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser and will
be considered to offer or sell such securities to such purchaser:
(i) any
preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule
424;
(ii) any
free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to
by the undersigned registrant;
(iii) the
portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant
or its securities provided by or on behalf of the undersigned registrant; and
(iv) any
other communication that is an offer in the offering made by the undersigned registrant to the purchaser.
The undersigned
registrant hereby undertakes that, for purposes of determining any liability under the Securities Act of 1933, each filing of
the registrant’s annual report pursuant to Section 13(a) or Section 15(d) of the Securities Exchange Act of 1934 (and, where
applicable, each filing of an employee benefit plan’s annual report pursuant to Section 15(d) of the Securities Exchange
Act of 1934) that is incorporated by reference in the registration statement shall be deemed to be a new registration statement
relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial
bona fide offering thereof.
Insofar
as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons
of the Registrant pursuant to the foregoing provisions, or otherwise, the Registrant has been advised that in the opinion of the
SEC such indemnification is against public policy as expressed in the Act and is therefore unenforceable. In the event that a
claim for indemnification against such liabilities (other than the payment by the Registrant of expenses incurred or paid by a
director, officer or controlling person of the Registrant in the successful defense of any action, suit or proceeding) is asserted
by such director, officer or controlling person in connection with the securities being registered, the Registrant will, unless
in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction
the question whether such indemnification by it is against public policy as expressed in the Act and will be governed by the final
adjudication of such issue.
The undersigned
registrant hereby undertakes that:
For purposes
of determining any liability under the Securities Act of 1933, as amended, the information omitted from a form of prospectus filed
as part of this registration statement in reliance upon Rule 430A and contained in the form of prospectus filed by the Registrant
pursuant to Rule 424(b)(1) or (4) or 497(h) under the Securities Act of 1933, as amended, shall be deemed to be part of this registration
statement as of the time it was declared effective.
For the
purpose of determining any liability under the Securities Act of 1933, as amended, each post-effective amendment that contains
a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering
of such securities at that time shall be deemed to be the initial bona fide offering thereof.
SIGNATURES
Pursuant to the requirements of the Securities Act of
1933, the registrant has duly caused this Amendment No.2 to the Registration Statement on Form S-1 to be signed on its behalf
by the undersigned, thereunto duly authorized, in the City of Gaithersburg, State of Maryland, on October 17, 2019.
|
|
OPGEN, INC.
|
|
|
|
|
|
By:
|
/s/ Evan Jones
|
|
|
|
Evan Jones
Chief Executive
Officer
|
POWER
OF ATTORNEY
Pursuant to the requirements of the Securities Act of
1933, as amended, this Amendment No.2 to the Registration Statement has been signed by the following persons in the capacities
and on the date indicated.
|
Signature
|
Title
|
Date
|
/s/ Evan Jones
Evan Jones
|
Chief
Executive Officer and Director (principal executive officer)
|
October
17, 2019
|
/s/ Timothy C. Dec
Timothy C. Dec
|
Chief
Financial Officer (principal financial officer and principal accounting officer)
|
October
17, 2019
|
*
R. Donald Elsey
|
Director
|
October
17, 2019
|
*
Tina S. Nova
|
Director
|
October
17, 2019
|
*
Misti Ushio
|
Director
|
October
17, 2019
|
|
|
|
|
|
*/s/
Timothy C. Dec
Timothy
C. Dec, as attorney in fact
|
|
October
17, 2019
|
|
|
|
|
OpGen (NASDAQ:OPGN)
Historical Stock Chart
From Aug 2024 to Sep 2024
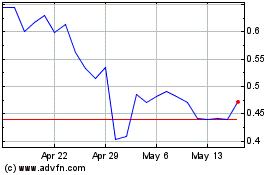
OpGen (NASDAQ:OPGN)
Historical Stock Chart
From Sep 2023 to Sep 2024