U.S. Backs Waiver of Intellectual Property Protection for Covid-19 Vaccines -- 2nd Update
May 05 2021 - 3:51PM
Dow Jones News
By Yuka Hayashi
WASHINGTON -- The Biden administration said it would back a
proposal at the World Trade Organization to waive intellectual
property protections for Covid-19 vaccines to help speed up global
production.
Developing nations led by India and South Africa have been
pushing for the waiver, saying it is needed to stem the coronavirus
pandemic. The waiver is also supported by more than 100 members of
Congress.
Pharmaceutical companies and other business groups have opposed
the waiver, however, saying it wouldn't supply short-term supply
production problems because contract producers lack certain needed
technical knowledge.
U.S. Trade Representative Katehrine Tai said the
administration's decision was based on the extraordinary
circumstances of the pandemic.
"The administration believes strongly in intellectual property
protections, but in service of ending this pandemic, supports the
waiver of those protections for Covid-19 vaccines," Ms. Tai
said,
Member nations of the WTO are discussing a proposal supported by
over 100 developing nations calling for a temporary waiver of
intellectual property rights protection related to the "prevention,
containment, or treatment" of Covid-19.
Until now, the U.S., other wealthy nations and the European
Union have opposed the waiver, saying IP protection provides an
important incentive for innovation to develop products to fight the
current and future pandemics.
Ms. Tai said the support for the waiver is just one part of
Washington's effort to get vaccines around the world quickly.
"As our vaccine supply for the American people is secured, the
administration will continue to ramp up its efforts -- working with
the private sector and all possible partners -- to expand vaccine
manufacturing and distribution," she said. "It will also work to
increase the raw materials needed to produce those vaccines."
Pfizer Inc., which collaborated with BioNTech SE on a Covid-19
vaccine, declined to comment. Pfizer Chief Executive Albert Bourla
said in an interview Tuesday that it doesn't make sense to share
patents because it wouldn't lead to increased production of vaccine
doses.
"It is so wrong," Mr. Bourla said of forcing the patent sharing.
He said the limited supply of Covid-19 vaccines stems from how
before the pandemic, there weren't any approved products using the
new gene-based mRNA technology that is used in the Pfizer-BioNTech
vaccine.
Pfizer, he said, has been increasing its manufacturing
capabilities to close that production gap for more than a year.
Dictating the sharing of patents would discourage biotech companies
from developing products for the next pandemic, he said.
Moderna Inc. last year said it wouldn't enforce patents related
to its experimental Covid-19 vaccine while the pandemic continues
and would be willing to license the patents to others after the
pandemic.
Representatives for Moderna, AstraZeneca and Johnson &
Johnson didn't immediately respond to requests for comment, nor did
the Pharma trade groups PhRMA and BIO.
Write to Yuka Hayashi at yuka.hayashi@wsj.com
(END) Dow Jones Newswires
May 05, 2021 16:36 ET (20:36 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
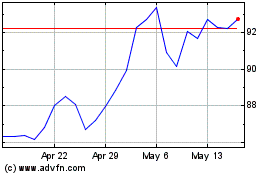
BioNTech (NASDAQ:BNTX)
Historical Stock Chart
From Apr 2024 to May 2024
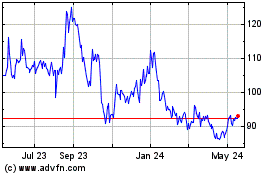
BioNTech (NASDAQ:BNTX)
Historical Stock Chart
From May 2023 to May 2024